MF C18H22N2O
MW: 282.17321
GDC-0919; NLG-919; RG-6078, GDC0919; GDC-0919; GDC 0919; NLG919; NLG 919; NLG-919; RG6078; RG-6078; RG 6078.
1-cyclohexyl-2-(5H-imidazo[5,1-a]isoindol-5-yl)ethanol
CAS No.1402836-58-1
GDC-0919, also known as NLG919 and RG6078, is an orally available inhibitor of indoleamine 2,3-dioxygenase 1 (IDO1), with potential immunomodulating and antineoplastic activities. Upon administration, NLG919 targets and binds to IDO1, a cytosolic enzyme responsible for the oxidation of the essential amino acid tryptophan into kynurenine. By inhibiting IDO1 and decreasing kynurenine in tumor cells, this agent increases tryptophan levels, restores the proliferation and activation of various immune cells, including dendritic cells (DCs), natural killer (NK) cells, T-lymphocytes, and causes a reduction in tumor-associated regulatory T-cells (Tregs). Activation of the immune system, which is suppressed in many cancers, may induce a cytotoxic T-lymphocyte (CTL) response against the IDO1-expressing tumor cells

- Originator Lankenau Institute for Medical Research
- Developer Genentech; NewLink Genetics Corporation
- Class Antineoplastics; Small molecules
- Mechanism of Action Immunomodulators; Indoleamine-pyrrole 2,3-dioxygenase inhibitors
Phase I Solid tumours
| Patent ID | Date | Patent Title |
|---|---|---|
| US2015210769 | 2015-07-30 | ANTIBODY MOLECULES TO PD-1 AND USES THEREOF |
| US2014066625 | 2014-03-06 | Fused Imidazole Derivatives Useful as IDO Inhibitors |
- 27 Sep 2015 Pharmacokinetics results from a phase-I clinical trial in Solid tumours presented at the European Cancer Congress 2015 (ECC-2015)
- 27 Sep 2015 Positive efficacy and safety results from a phase-I clinical trial in Solid tumours presented at the European Cancer Congress 2015 (ECC-2015)
- 31 Jul 2015 Phase-I clinical trials in Solid tumours (Combination therapy, Late-stage disease, Second-line therapy or greater) in USA (PO) (NCT02471846)
PATENT
http://www.google.com/patents/WO2012142237A1?cl=en
PATENT
Fused Imidazole Derivatives Useful as IDO Inhibitors
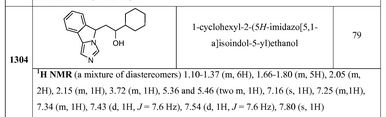
13041-cyclohexyl-2-(5H-imidazo[5,1- a]isoindol-5-yl)ethanol79 1H NMR (a mixture of diastereomers) 1.10-1.37 (m, 6H), 1.66-1.80 (m, 5H), 2.05 (m, 2H), 2.15 (m, 1H), 3.72 (m, 1H), 5.36 and 5.46 (two m, 1H), 7.16 (s, 1H), 7.25 (m, 1H), 7.34 (m, 1H), 7.43 (d, 1H, J = 7.6 Hz), 7.54 (d, 1H, J = 7.6 Hz), 7.80 (s, 1H)
| WO2011056652A1 * | Oct 27, 2010 | May 12, 2011 | Newlink Genetics | Imidazole derivatives as ido inhibitors |
| WO2012142237A1 * | Apr 12, 2012 | Oct 18, 2012 | Newlink Geneticks Corporation | Fused imidazole derivatives useful as ido inhibitors |
| WO2014159248A1 | Mar 10, 2014 | Oct 2, 2014 | Newlink Genetics Corporation | Tricyclic compounds as inhibitors of immunosuppression mediated by tryptophan metabolization |
| US8722720 | Oct 27, 2010 | May 13, 2014 | Newlink Genetics Corporation | Imidazole derivatives as IDO inhibitors |
| US9260434 | Oct 14, 2013 | Feb 16, 2016 | Newlink Genetics Corporation | Fused imidazole derivatives useful as IDO inhibitors |
| US20140066625 * | Oct 14, 2013 | Mar 6, 2014 | Newlink Genetics Corporation | Fused Imidazole Derivatives Useful as IDO Inhibitors |
| US20160002249 * | Jul 8, 2015 | Jan 7, 2016 | Newlink Genetics Corporation | Fused Imidazole Derivatives Useful as IDO Inhibitors |
REFERENCES
Nature Reviews Drug Discovery14,373(2015)doi:10.1038/nrd4658
http://www.ncbi.nlm.nih.gov/pubmed/21517759
http://www.roche.com/irp150128-annex.pdf
/////CRD1152, CRD 1152, CRD-1152, Curadev, Research Collaboration, Licensing Agreement, Develop, Cancer Immunotherapeutic, IDO1 and TDO inhibitors

OC(C1CCCCC1)CC(C2=C3C=CC=C2)N4C3=CN=C4

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....