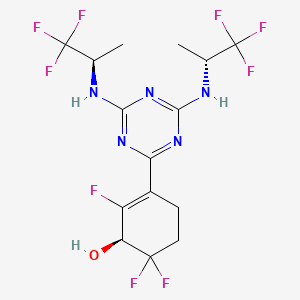
Ranosidenib
CAS 2301974-60-5
MF C15H16F9N5O MW 453.31 g/mol
(1S)-3-[4,6-bis[[(2R)-1,1,1-trifluoropropan-2-yl]amino]-1,3,5-triazin-2-yl]-2,6,6-trifluorocyclohex-2-en-1-ol
(1S)-3-(4,6-bis{[(2R)-1,1,1-trifluoropropan-2-yl]amino}-1,3,5-triazin-2-yl)-2,6,6-trifluorocyclohex-2-en-1-ol
isocitrate dehydrogenase (IDH) inhibitor, antineoplastic, [14C] HMPL-306, HMPL 306, PC64OXS2C2
- OriginatorHutchison MediPharma
- DeveloperHUTCHMED
- ClassAntineoplastics; Small molecules
- Mechanism of ActionIsocitrate dehydrogenase 1 inhibitors; Isocitrate dehydrogenase 2 inhibitors
- Phase IIIAcute myeloid leukaemia
- No development reportedHaematological malignancies; Solid tumours
- 28 Sep 2025No recent reports of development identified for phase-I development in Haematological-malignancies(Late-stage disease, Second-line therapy or greater) in Spain (PO)
- 28 Sep 2025No recent reports of development identified for phase-I development in Haematological-malignancies(Late-stage disease, Second-line therapy or greater) in USA (PO)
- 19 Sep 2025No development reported – Phase-I for Solid tumours (Late-stage disease, Metastatic disease, Second-line therapy or greater) in USA (PO)
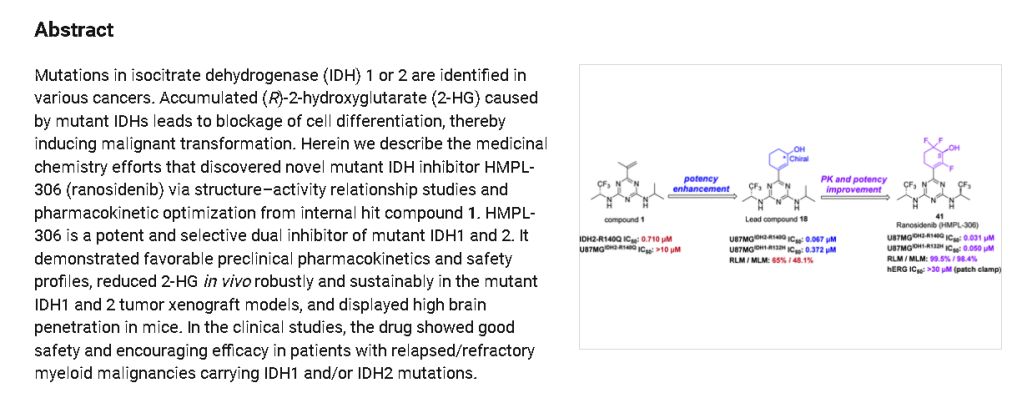
Ranosidenib is a small molecule drug. Ranosidenib is under investigation in clinical trial NCT06387069 (A Study to Evaluate HMPL-306 in Patients With IDH1- and IDH2-mutated Acute Myeloid Leukemia). Ranosidenib has a monoisotopic molecular weight of 453.12 Da.
Ranosidenib is an orally bioavailable inhibitor of mutated forms of both isocitrate dehydrogenase type 1 (IDH1, IDH1 [NADP+] soluble) in the cytoplasm and type 2 (IDH2, isocitrate dehydrogenase [NADP+], mitochondrial) in the mitochondria, with potential antineoplastic activity. Upon administration, ranosidenib specifically targets and inhibits mutant forms of IDH1 and IDH2, thereby inhibiting the formation of the oncometabolite 2-hydroxyglutarate (2HG) from alpha-ketoglutarate (a-KG). This prevents 2HG-mediated signaling and leads to both an induction of cellular differentiation and an inhibition of cellular proliferation in tumor cells expressing IDH mutations. IDH1 and 2, metabolic enzymes that catalyze the conversion of isocitrate into a-KG, play key roles in energy production and are mutated in a variety of cancer cell types. Mutant forms of IDH1 and 2 catalyze the formation of 2HG and drive cancer growth by blocking cellular differentiation and inducing cellular proliferation.
A Study of HMPL-306 in Advanced Hematological Malignancies With mIDHCTID: NCT04764474Phase: Phase 1Status: TerminatedDate: 2026-01-29
A Study of HMPL-306 in Advanced Solid Tumors With IDH MutationsCTID: NCT04762602Phase: Phase 1Status: TerminatedDate: 2025-09-16
A Study to Evaluate HMPL-306 in Patients With IDH1or IDH2-mutated Acute Myeloid LeukemiaCTID: NCT06387069Phase: Phase 3Status: RecruitingDate: 2025-08-14
Phase I Study of HMPL-306 for the Treatment of Gliomas With IDH1 and/or IDH2 MutationsCTID: NCT07025018Phase: Phase 1Status: RecruitingDate: 2025-08-01
A Study of HMPL-306 in Patients With IDH1 and/or IDH2 Mutation of Relapsed/Refractory Myeloid Leukemia/NeoplasmsCTID: NCT04272957Phase: Phase 1Status: Unknown statusDate: 2020-06-16
SYN
https://pubs.acs.org/doi/10.1021/acsmedchemlett.4c00625
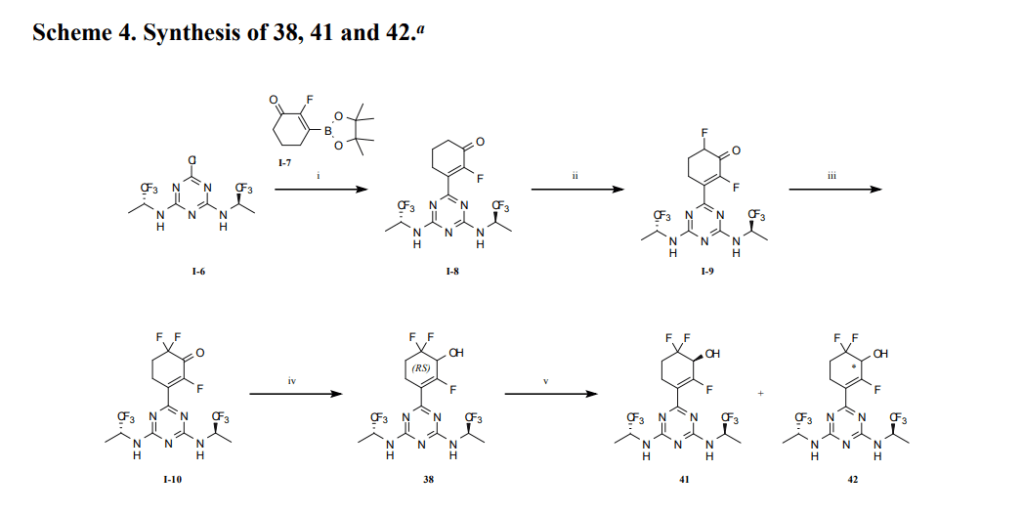
aReagents and conditions: (i) Na2PdCl4, DTBPPS, K2CO3, MeCN, H2O, 60 ℃; (ii) TBSOTf, Et3N,
DCM, 0~5 ℃; Selectfluor®, MeCN, 0~5 ℃; (iii) TBSOTf, Et3N, DCM, 0~5 ℃; Selectfluor®,
MeCN, 0~5 ℃; (iv) NaBH4, CeCl3·7H2O, EtOH, 0 ℃; (v) SFC separation.
Pat
Cycloolefin substituted heteroaromatic compounds and their use
Publication Number: US-2021363115-A2
Priority Date: 2017-09-07
PAT
Intermediate I-3
6-Chloro-N2,N4-bis((R)-1,1,1-trifluoropropan-2-yl)-1,3,5-triazine-2,4-diamine
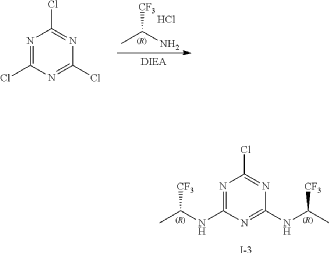
At 0° C., to a flask were added 1,4-dioxane (50 mL), 2,4,6-trichloro-1,3,5-triazine (1.84 g, 10 mmo), (R)-1,1,1-trifluoropropan-2-amine hydrochloride (2.99 g, 20 mmol) and DIEA (5.17 g, 40 mmol). The reaction was heated to 60° C. and stirred for 4 hours. After the reaction was completed, the mixture was condensed and purified by flash column chromatography (eluting with gradient water/MeOH=100:0-0:100) to give Intermediate I-3 as yellow solid (2.50 g, yield: 74%). MS (m/z): 338.0 [M+H] +
Compounds 197 and 198
3-(4,6-Bis(((R)-1,1,1-trifluoropropan-2-yl)amino)-1,3,5-triazin-2-yl)-2,6,6-trifluorocyclohex-2-en-1-ol, optically pure diastereoisomers
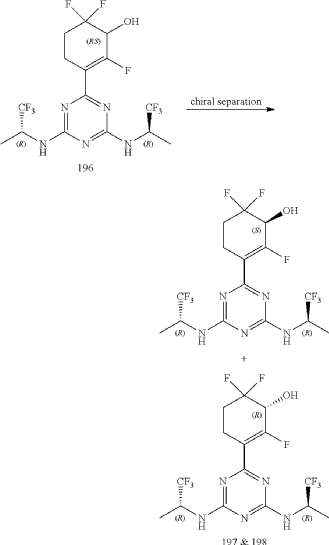
| The Compound 196 was resolved by chiral HPLC to provide a pair of optically pure diastereoisomers, Compounds 197 and 198 (Chiral HPLC conditions: Preparation instrument: Shimadzu LC-10AD vp; Column: Daicel AD-H(250 mm*30 mm, 5 um); mobile phase: n-heptane/isopropanol=90/10; flow rate: 40 mL/min; column temperature: 40° C.). The first eluent (RT=4.203 min) was concentrated and purified by flash column chromatography (eluting with gradient PE/EA=100:0-0:100) to give a compound named as Compound 197, de %=99.27%, MS (m/z): 454.1 [M+1] +. The second eluent (RT=5.906 min) was concentrated and purified by flash column chromatography (eluting with gradient PE/EA=100:0-0:100) to give a compound named as Compound 198, de %=97.82%, MS (m/z): 454.2 [M+1] +. |
| Compound 197: 1H NMR (400 MHz, CD 3OD): δ 5.00-4.86 (m, 2H), 4.36-4.17 (m, 1H), 2.80-2.65 (m, 1H), 2.58-2.42 (m, 1H), 2.25-2.05 (m, 2H), 1.37-1.31 (m, 6H). |
| Compound 198: 1H NMR (400 MHz, CD 3OD): δ 5.00-4.86 (m, 2H), 4.36-4.17 (m, 1H), 2.80-2.65 (m, 1H), 2.58-2.42 (m, 1H), 2.25-2.05 (m, 2H), 1.37-1.31 (m, 6H). |



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

/////////ranosidenib, isocitrate dehydrogenase (IDH) inhibitor, antineoplastic, [14C] HMPL-306, HMPL 306, PC64OXS2C2

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
