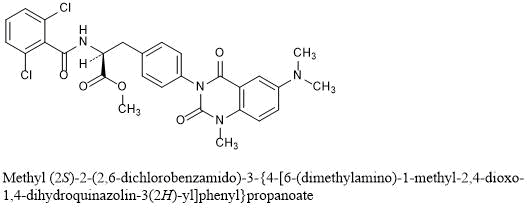
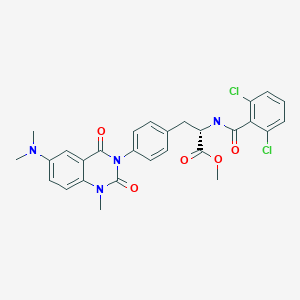
Carotegrast methyl
| Formula | C28H26Cl2N4O5 |
|---|---|
| CAS | 401905-67-7 |
| Mol weight | 569.4358 |
PMDA APROVED, CAROGRA, カロテグラストメチル
| ON 2022/3/28 |
Antiasthmatic, Integrin alpha 4 inhibitor
- An alpha4 integrin antagonist.
401905-67-7[RN]
L-Phenylalanine, N-(2,6-dichlorobenzoyl)-4-[6-(dimethylamino)-1,4-dihydro-1-methyl-2,4-dioxo-3(2H)-quinazolinyl]-, methyl ester
methyl (2S)-2-[(2,6-dichlorophenyl)formamido]-3-{4-[6-(dimethylamino)-1-methyl-2,4-dioxo-1,2,3,4-tetrahydroquinazolin-3-yl]phenyl}propanoate
Methyl N-(2,6-dichlorobenzoyl)-4-[6-(dimethylamino)-1-methyl-2,4-dioxo-1,4-dihydro-3(2H)-quinazolinyl]-L-phenylalaninate
Carotegrast Methyl

Methyl (2S)-2-(2,6-dichlorobenzamido)-3-{4-[6-(dimethylamino)-1-methyl-2,4-dioxo-1,4-dihydroquinazolin-3(2H)-yl]phenyl}propanoate
C28H26Cl2N4O5 : 569.44
[401905-67-7]
PATENT
WO 2008062859
https://patents.google.com/patent/WO2008062859A1/en
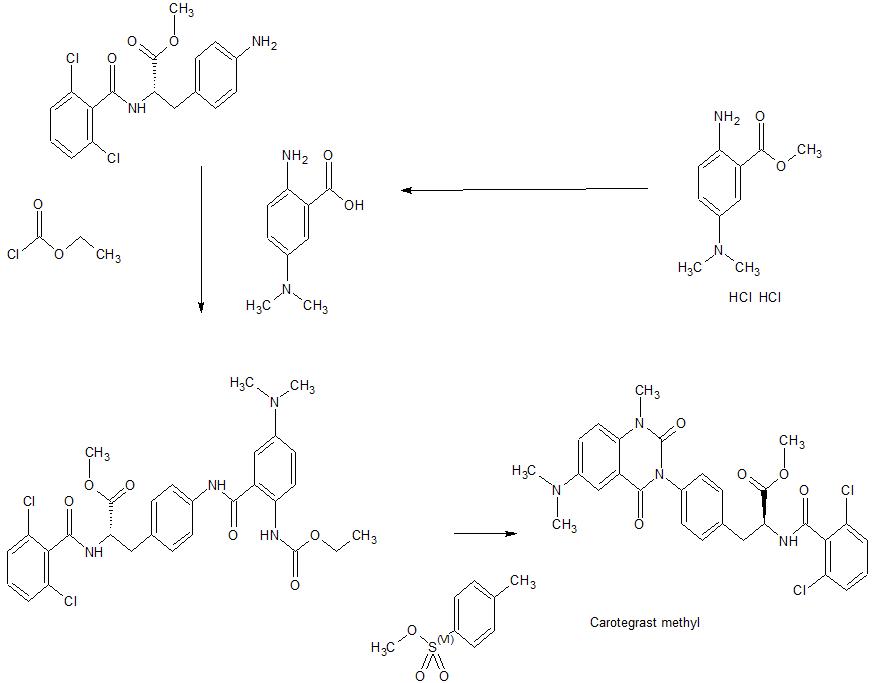
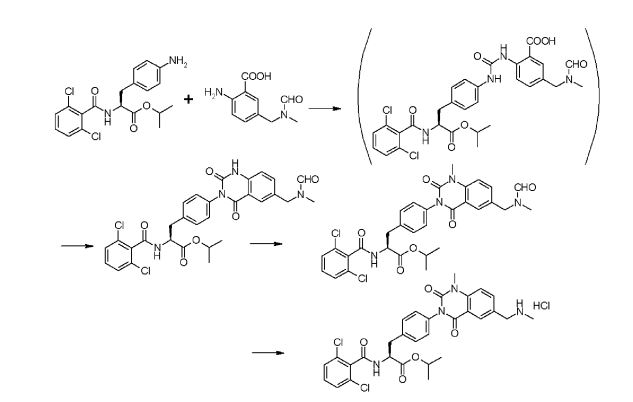
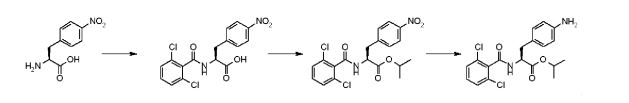
Step 1
(Method 2): The title compound was prepared starting from 2-amino-5-dimethylamino- benzoic acid methyl ester dihydrochloride through the hydrolysis under basic condition To 5.0 g of 2-amino-5-dimethylamino-benzoic acid methyl ester di-hydrochloride, there were added 15 mL of water and 15.6 mL of a 6M aqueous solution of sodium hydroxide and the resulting mixture was heated to 40°C for 2 hours. After the confirmation of the progress of the reaction according to HPLC, the reaction system was cooled to room temperature, a 6M hydrochloric acid aqueous solution was dropwise added to the reaction system to thus neutralize the same and to separate out crystals (pH 4.9) and then the reaction system was stirred at 10°C for 2 hours. The solid thus obtained was isolated through the filtration under reduced pressure, washed with 30 mL of water and then dried under reduced pressure at 60°C for 14 hours. Title compound 3.14 g was obtained as gray-colored solid. The physical properties determined were almost identical to those observed for the same compound prepared in the above-mentioned synthesis example. H-NMR (400MHz, DMSO-d6): δ 8.21 (bs, 3H), 7.10 (d, 1H, J=2.8Hz), 6.97 (dd, 1H, J=9.1, 2.8Hz), 6.70 (d, 1H, J=9.1 Hz), 2.72 (s, 6H); 13C-NMR (100MHz, DMSO-d6): δ168.89, 144.55, 141.61, 123.29, 117.90, 114.78, 110.11,41.95; MS (ESI+): m/z 181.3 (MH+), (ESI-): m/z 179.2 (M-H–).
Step 2
Step 1: Synthesis of Nα-(2,6-dichlorobenzoyl) -4-{2-ethoxycarbonylamino-5-dimethyl- amino-benzoylamino}-L-phenylalanine methyl ester To 1.96 g of 2-amino-5-dimethylaminobenzoic acid, there were added 12 mL of acetonitrile and 5.29 mL of pyridine to form a suspension and then the resulting suspension was cooled to 4°C. To this suspension there was dropwise added 4.17 mL of ethyl chloroformate over 5 minutes and then the mixture was stirred at 25°C for one hour. After confirming the disappearance of the starting material by HPLC, 0.7 mL of ethanol was added to the mixture to thus decompose the excess ethyl chloroformate and the mixture was further stirred for additional one hour. To this reaction solution there were added 4.0 g of 4-amino-Nα-(2,6-dichlorobenzoyl)-L-phenylalanine methyl ester and 12 mL of N,Ndimethylformamide, and the resulting mixture was stirred overnight. Subsequently, 48 mL of methanol was drop-wise added, the resulting mixture was stirred at 10°C overnight and then the solid separated from the mixture was isolated through filtration under reduced pressure. The solid was then washed with 8 mL of methanol and dried at 70°C for 5 hours under reduced pressure. Title compound 5.50 g was obtained as pale yellow solid. 1H-NMR (400MHz, DMSO-d6): δ 10.29 (s, 1H), 9.42 (bs, 1H), 9.24 (d, 1H, J=7.9Hz), 7.73 (bs, 1H), 7.62 (d, 2H, J=8.4Hz), 7.48-7.44 (m, 2H), 7.41 (dd, 1H, J=9.5, 6.2Hz), 7.27 (d, 2H,J=8.4Hz), 7.01 (d, 1H, J=2.7Hz), 6.93 (dd, 1H, J=9.1, 2.9Hz), 4.71 (ddd, 1H, J=9.2, 8.1, 5.7Hz), 4.05 (q, 2H, J=7.0Hz), 3.66 (s, 3H), 3.10 (dd, 1H, J=14.0, 5.6Hz), 2.96 (dd, 1H, J=14.0, 9.2Hz), 2.93 (s, 6H), 1.18 (t, 3H, J=7.2Hz); MS (ESI+): m/z 601.2 (MH+) and 623.2 (M+Na), (ESI–): m/z 599.1 (M-H–).
Step 3
Step2: Synthesis of Na-(2,6-dichlorobenzoyl)-4-{6-dimethylamino-1-methylquinazoline-2,4[1H,3H]-dion-3-yl}-L-phenylalanine methyl ester To 2.0 g of Na-(2,6-dichlorobenzoyl)-4-{2-ethoxycarbonylamino -5-dimethyl- amino-benzoylamino}-L-phenylalanine methyl ester prepared in above-mentioned step 1, were added 16 mL of N,N-dimethylfbrmamide, 0.8 mL of methanol and 0.91 g of potassium carbonate, followed by the stirring of the resulting mixture at 25°C overnight. To this reaction solution, there was added 0.75 mL of methyl p-toluenesulfonate for subjecting the methyl ester to alkylation at 25~40°C. After confirming the disappearance of the starting material by HPLC, 0.75 mL of acetic acid was added to quench the reaction, 16 mL of water was dropped and the solid was separated. Further, 8 mLof N,N-dimethylformamide/water = 1/1 mixed liquid was added to the resulting mixture, followed by the stirring of the mixture at 25°C. Then the solid thus separated was isolated through filtration under reduced pressure and then washed with 8 mL of water. Thereafter, the isolated solid was dried at 70°C for 4 hours under reduced pressure. Desired compound 1.77 g was obtained as pale yellow solid. 1H-NMR (400MHz, DMSO-d6): δ 9.28 (d, 1H, J=8.1 Hz), 7.48-7.36 (m, 6H), 7.31 (dd, 1H, J=3.0, 9.0Hz), 7.24 (d, 1H, J=3.0Hz), 7.20-7.15 (m, 2H), 4.18 (ddd, 1H, J=10.2, 8.1,4.8Hz), 3.69 (s, 3H), 3.49 (s, 3H), 3.22 (dd, 1H, J=14.1, 4.8Hz), 3.02 (dd, 1H, J=14.2, 10.5Hz), 2.94 (s, 6H); MS (ESI+): m/z 569.2 (MH+) and 591.1 (M+Na), (ESI-): m/z 567.2 (M-H–).
PATENT
https://patents.google.com/patent/WO2004074264A1/en
PATENT’ WO 2003070709
https://patents.google.com/patent/WO2003070709A1/en
PATENT
WO 2002016329
///////////

AS ON DEC2021 3,491,869 VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@amcrasto
/////////////////////////////////////////////////////////////////////////////
/////////////Carotegrast methyl, CAROGRA, カロテグラストメチル , JAPAN 2022, APPROVALS 2022,
COC(=O)[C@H](Cc1ccc(cc1)N2C(=O)N(C)c3ccc(cc3C2=O)N(C)C)NC(=O)c4c(Cl)cccc4Cl

NEW DRUG APPROVALS
ONE TIME
$10.00

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
