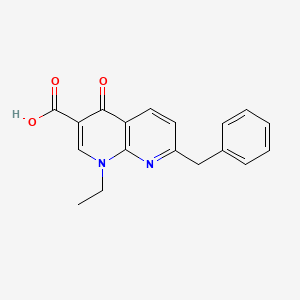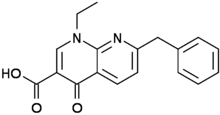
Amfonelic acid
- Molecular FormulaC18H16N2O3
- Average mass308.331 Da
Amfonelic acid (AFA; WIN 25,978) is a research chemical and dopaminergic stimulant with antibiotic properties.[1]
History
The stimulant properties of AFA were discovered serendipitously at Sterling-Winthrop in the midst of research on the antibiotic nalidixic acid.[1] In addition to behaving as antibiotics, it was found that many derivatives of nalidixic acid have either stimulant or depressant effects on the central nervous system.[2] Researchers at Sterling-Winthrop found that AFA had a higher potency and therapeutic indexthan cocaine or amphetamine and so it was singled out for further study.[1][3] A small number of clinical trials were held in the 1970s, but when it was found that AFA exacerbated psychotic symptoms in schizophrenic patients and produced undesirable stimulant properties in geriatric depressives clinical evaluation of AFA was discontinued.[1] AFA remains a widely used pharmacological tool for study of the brain’s reward system, dopamine pathways, and the dopamine transporter.[1] Since 2013 AFA has been sold on the gray market and there are numerous anecdotal reports detailing its non-medical use.[1]
Pharmacology
In studies it proved to be a potent and highly selective dopamine reuptake inhibitor (DRI) in rat brain preparations.[4][5] A study found a moderately long half-life of approximately 12 hours and a dopaminergic potency approximately 50 fold that of methylphenidate in rat brain preparations.[6] Despite lack of direct serotonin activity, rats treated with subchronic doses of amfonelic acid display subsequent decreases in 5HT and 5HIAA.[7] Amfonelic acid displays no activity in the norepinephrine system.[8]
Despite its different mechanism of action, amfonelic acid displays discriminatory substitution with 150% the stimulant potency of dextroamphetamine.[9] Amfonelic acid has been shown to be neuroprotective against methamphetamine damage to dopamine neurons.[10] It also increases the effects of the antipsychotic drugs haloperidol, trifluoperazine and spiperone.[11] Rats are shown to self-administer amfonelic acid in a dose-dependent manner.[12]
Though AFA was discovered in the course of antibiotic research, there is very little data available on the drug’s antimicrobial activity. In 1988 the biologist G.C. Crumplin wrote, “[AFA] is less active against bacteria than are many other 4-quinolones, but studies in our laboratory on selected mammalian cell lines have shown it to be markedly more toxic to these cells than are the 4-quinolones that are more active antibacterial agents. Furthermore, it can be shown that sublethal doses induced marked changes in the pattern of proteins produced by the cell, thus suggesting a possible effect of 4-quinolones on gene transcription in mammalian cells.”[13] When evaluated via broth microdilution the MIC of AFA for Escherichia coli is 125 μg/mL, a concentration thirty times higher than the MIC for nalidixic acid in the same E. coli strain.[1]
References
- ^ Jump up to:a b c d e f g Morris, Hamilton (October 2015). “Sad Pink Monkey Blues”. Harper’s Magazine. Retrieved 2015-09-19.
- Jump up^ US patent 3590036, “Naphthyridine-3-carboxylic Acids, Their Derivatives and Preparation Thereof”
- Jump up^ Aceto, M.A. (1970). “Pharmacologic properties and mechanism of action of amfonelic acid”. European Journal of Pharmacology. 10: 344–354. doi:10.1016/0014-2999(70)90206-2. PMID 4393073.
- Jump up^ Fuller, R. W.; Perry, K. W.; Bymaster, F. P.; Wong, D. T. (1978). “Comparative effects of pemoline, amfonelic acid and amphetamine on dopamine uptake and release in vitro and on brain 3,4-dihydroxyphenylacetic acid concentration in spiperone-treated rats”. Journal of Pharmacy and Pharmacology. 30 (3): 197–198. doi:10.1111/j.2042-7158.1978.tb13201.x. PMID 24701.
- Jump up^ McMillen, B. A.; Shore, P. A. (1978). “Amfonelic acid, a non-amphetamine stimulant, has marked effects on brain dopamine metabolism but not noradrenaline metabolism: Association with differences in neuronal storage systems”. Journal of Pharmacy and Pharmacology. 30 (7): 464–466. doi:10.1111/j.2042-7158.1978.tb13293.x. PMID 27622.
- Jump up^ Izenwasser, S.; Werling, L. L.; Cox, B. M. (1990). “Comparison of the effects of cocaine and other inhibitors of dopamine uptake in rat striatum, nucleus accumbens, olfactory tubercle, and medial prefrontal cortex”. Brain Research. 520 (1–2): 303–309. doi:10.1016/0006-8993(90)91719-W. PMID 2145054.
- Jump up^ McMillen, BA; Scott, SM; Williams, HL (1991). “Effects of subchronic amphetamine or amfonelic acid on rat brain dopaminergic and serotonergic function”. Journal of neural transmission. General section. 83 (1–2): 55–66. doi:10.1007/BF01244452. PMID 2018630.
- Jump up^ Agmo, A; Belzung, C; Rodríguez, C (1997). “A rat model of distractibility: Effects of drugs modifying dopaminergic, noradrenergic and GABAergic neurotransmission”. Journal of neural transmission (Vienna, Austria : 1996). 104 (1): 11–29. doi:10.1007/BF01271291. PMID 9085190.
- Jump up^ Aceto, MD; Rosecrans, JA; Young, R; Glennon, RA (1984). “Similarity between (+)-amphetamine and amfonelic acid”. Pharmacology Biochemistry and Behavior. 20 (4): 635–7. doi:10.1016/0091-3057(84)90316-2. PMID 6728880.
- Jump up^ Pu, C; Fisher, JE; Cappon, GD; Vorhees, CV (1994). “The effects of amfonelic acid, a dopamine uptake inhibitor, on methamphetamine-induced dopaminergic terminal degeneration and astrocytic response in rat striatum”. Brain Research. 649 (1–2): 217–24. doi:10.1016/0006-8993(94)91067-7. PMID 7953636.
- Jump up^ Waldmeier, PC; Huber, H; Heinrich, M; Stoecklin, K (1985). “Discrimination of neuroleptics by means of their interaction with amfonelic acid: An attempt to characterize the test”. Biochemical Pharmacology. 34 (1): 39–44. doi:10.1016/0006-2952(85)90097-8. PMID 2857083.
- Jump up^ Porrino, LJ; Goodman, NL; Sharpe, LG (1988). “Intravenous self-administration of the indirect dopaminergic agonist amfonelic acid by rats”. Pharmacology Biochemistry and Behavior. 31 (3): 623–6. doi:10.1016/0091-3057(88)90240-7. PMID 2908003.
- Jump up^ Crumplin, G.C. (1988). “Aspects of Chemistry in the Development of the 4-Quinolone Antibacterial Agents”. Reviews of Infectious Diseases. 10 Suppl 1 (10): S2–S9. doi:10.1093/clinids/10.Supplement_1.S2. PMID 3279494.
External links
 |
|
| Clinical data | |
|---|---|
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C18H16N2O3 |
| Molar mass | 308.3329 g/mol |
| 3D model (JSmol) | |
////////////Amfonelic acid, RR302AR19Y, амфонеловая кислота , حمض أمفونيليك , 安福萘酸 , アンホネル酸
CCN1C=C(C(=O)C2=C1N=C(C=C2)CC3=CC=CC=C3)C(=O)O

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....