
Rebamipide
- Molecular FormulaC19H15ClN2O4
- Average mass370.786 Da
- Monoisotopic mass370.072021 Da
OPC-12759
OPC-12759E
OPC-759
(-)-Configuration: from dimethylformamide to give colorless needles, mp 305~306 °C (decomposition). [α] D20-116.7 ° (C = 1.0, dimethylformamide).
(+)-Configuration: from dimethylformamide to give colorless needles, mp 305~306 °C (decomposition). [α] D20 + 116.9 ° (C = 1.0, dimethylformamide).
Rebamipide has been shown to inhibit the production of reactive oxygen species and to decrease cytokine release induced by H. pylori infection.
A daily oral dose of 100 mg/kg was found to be protective against the development of pyloric channel ulcers in Mongolian gerbils infected with H. pylori.
In addition to the stomach, rebamipide can also enhance secretion of mucin covering the conjunctiva and cornea, which is important for tear film adhesion.
Rebamipide, a gastroprotective drug, was developed in Japan and was proven to be superior to cetraxate, the former most prescribed drug of the same category, in 1989 in the treatment for gastric ulcers. The initially discovered basic mechanisms of action of rebamipide included its action as a prostaglandin inducer and oxygen free-radical scavenger. In the last 5 years, several basic and clinical studies have been performed for functional dyspepsia, chronic gastritis, NSAID-induced gastrointestinal injuries, gastric ulcer following eradication therapy for Helicobacter pylori, gastric ulcer after endoscopic surgery and ulcerative colitis. In addition, several molecules have been identified as therapeutic targets of rebamipide to explain its pleiotropic pharmacological actions.
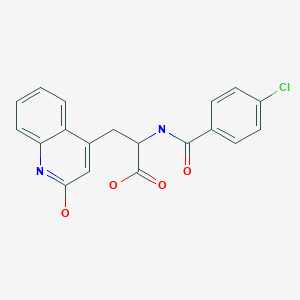
Rebamipide, an amino acid derivative of 2-(1H)-quinolinone, is used for mucosal protection, healing of gastroduodenal ulcers, and treatment of gastritis. It works by enhancing mucosal defense, scavenging free radicals, and temporarily activating genes encoding cyclooxygenase-2.
Rebamipide is used in a number of Asian countries including Japan (marketed as Mucosta), South Korea, China[1] and India (where it is marketed under the trade name Rebagen). It is also approved in Russia under the brand name Rebagit.[2] It is not approved by the Food and Drug Administration for use in the United States.
Studies have shown that rebamipide can fight the damaging effects of NSAIDs on the GIT mucosa, and more recently, the small intestine.[citation needed] It has also been studied for the treatment of Behçet’s disease.[3] It was shown to successfully treat pouchitis in a single-N study after first-line therapies for the condition were unsuccessful.[4] Some studies have shown effectiveness in presbyacusis(age-related hearing loss).[citation needed]
It has also been shown to alleviate signs and symptoms of dry eyes in a randomised controlled trial although this is not yet widely available clinically.[5]
SYN
Rebamipide (CAS NO.: 111911-87-6), with its systematic name of 4-Quinolinepropanic acid, alpha-((4-chlorobenzoyl)amino)-1,2-dihydro-2-oxo-, (+-)-, could be produced through many synthetic methods.
Following is one of the reaction routes:

4-(Bromomethyl)quinolin-2(1H)-one (I) could react with hot phosphorus oxychloride to produce a mixture of 4-(bromomethyl)-2-chloroquinoline (II) and 2-chloro-4-(chloromethyl)quinoline (III), and then the mixture without separation is ondensed with 2(S)-isopropyl-3,6-dimethoxy-2,5-dihydropyrazine (IVs) in the presence of butyllithium in hexane, affording (-)-2-chloro-4-[6(S)-isopropyl-2,5-dimethoxy-3,6-dihydropyrazin-3(R)-yl methyl]quinoline (Vr). The hydrolysis of (Vr) with HCl produces 3-(2-chloroquinolin-4-yl)-(R)-alanine methyl ester (VIr), which is treated with HCl and propylene oxide to afford 3-(2-oxo-2,3-dihydroquinolin-4-yl)-(R)-alanine (VIIr). At last, this compound is acylated with 4-chlorobenzoyl chloride (VIII) by means of K2CO3in acetone, affording (R)-OPC-12759.

Figure 2 The synthetic route of Rebamipide.

DE 3324034; US 4578381 ABOVE
The condensation of 4-(bromomethyl)quinolin-2(1H)-one (I) with diethyl acetamidomalonate (II) by means of sodium ethoxide in refluxing ethanol gives ethyl 2-acetamido-2-(ethoxycarbonyl)-3-(2-oxo-1,2-dihydroquinolin-4yl)propionate (III), which is submitted to a decarboxylative hydrolysis with refluxing 20% HCl yielding 3-(2-oxo-1,2-dihydroquinolin-4yl)alanine (IV). Finaily this compound is acylated with 4-chlorobenzoyl chloride by means of K2CO3 in acetone water.
SYN

Chem Pharm Bull 1991,39(11),2906 ABOVE
The synthesis of (R)- and (S)-isomers of OPC-12759 has been described: These optical isomers can be obtained in three different ways: 1) The reaction of 4-(bromomethyl)quinolin-2(1H)-one (I) with hot phosphorus oxychloride gives a mixture of 4-(bromomethyl)-2-chloroquinoline (II) and 2-chloro-4-(chloromethyl)quinoline (III), which, without separation, is condensed with 2(S)-isopropyl-3,6-dimethoxy-2,5-dihydropyrazine (IVs) by means of butyllithium in hexane, yielding (-)-2-chloro-4-[6(S)-isopropyl-2,5-dimethoxy-3,6-dihydropyrazin-3(R)-yl methyl]quinoline (Vr). The hydrolysis of (Vr) with HCl affords 3-(2-chloroquinolin-4-yl)-(R)-alanine methyl ester (VIr), which is treated with HCl and propylene oxide to give 3-(2-oxo-2,3-dihydroquinolin-4-yl)-(R)-alanine (VIIr). Finally, this compound is acylated with 4-chlorobenzoyl chloride (VIII) by means of K2CO3 in acetone, affording (R)-OPC-12759.
SYN

 3) The methylation of 3-(2-oxo-1,2-dihydroquinolin-4-yl)-(R,S)-alanine (IX) with SOCl2 and methanol yields the corresponding methyl ester (X), which is submitted to optical resolution with D-(-)-mandelic acid, affording adducts (XII) and (XIII). The hydrolytic treatment of (XII) and (XIII) with HCl and propylene oxide finally yields isomers (VIIr) and (VIIs), already obtained. Racemic OPC-12759 can also be resolved into its optical isomers by treatment with brucine and fractionated crystallization.
3) The methylation of 3-(2-oxo-1,2-dihydroquinolin-4-yl)-(R,S)-alanine (IX) with SOCl2 and methanol yields the corresponding methyl ester (X), which is submitted to optical resolution with D-(-)-mandelic acid, affording adducts (XII) and (XIII). The hydrolytic treatment of (XII) and (XIII) with HCl and propylene oxide finally yields isomers (VIIr) and (VIIs), already obtained. Racemic OPC-12759 can also be resolved into its optical isomers by treatment with brucine and fractionated crystallization.
Rebamipide
-
- Synonyms:Proamipide
- ATC:A02BX
- Use:ulcer therapeutic
- Chemical name:α-[(4-chlorobenzoyl)amino]-1,2-dihydro-2-oxo-4-quinolinepropanoic acid
- Formula:C19H15ClN2O4
- MW:370.79 g/mol
- CAS-RN:90098-04-7
- LD50:572 mg/kg (M, i.v.);
700 mg/kg (R, i.v.);
>2 g/kg (dog, p.o.)
Substance Classes
Synthesis Path
Substances Referenced in Synthesis Path
| CAS-RN | Formula | Chemical Name | CAS Index Name |
|---|---|---|---|
| 39098-85-6 | C4H5ClO2 | acetoacetyl chloride | Butanoyl chloride, 3-oxo- |
| 62-53-3 | C6H7N | aniline | Benzenamine |
| 4876-10-2 | C10H8BrNO | 4-(bromomethyl)-2(1H)-quinolinone | 2(1H)-Quinolinone, 4-(bromomethyl)- |
| 128-08-5 | C4H4BrNO2 | N-bromosuccinimide | 2,5-Pyrrolidinedione, 1-bromo- |
| 122-01-0 | C7H4Cl2O | 4-chlorobenzoyl chloride | Benzoyl chloride, 4-chloro- |
| 1068-90-2 | C9H15NO5 | diethyl acetamidomalonate | Propanedioic acid, (acetylamino)-, diethyl ester |
| 4900-38-3 | C19H22N2O6 | ethyl 2-acetamido-2-(ethoxycarbonyl)-3-(2-oxo-1,2-dihydroquinolin-4-yl)propionate | Propanedioic acid, (acetylamino)[(1,2-dihydro-2-oxo-4-quinolinyl)methyl]-, diethyl ester |
| 5162-90-3 | C12H12N2O3 | 3-(2-oxo-1,2-dihydroquinolin-4-yl)alanine | 4-Quinolinepropanoic acid, α-amino-1,2-dihydro-2-oxo- |
| 102-01-2 | C10H11NO2 | 3-oxo-N-phenylbutanamide | Butanamide, 3-oxo-N-phenyl- |
Trade Names
| Country | Trade Name | Vendor | Annotation |
|---|---|---|---|
| J | Mucosta | Otsuka |
Formulations
- tabl. 100 mg
References
-
- Uchida, M. et al.: Chem. Pharm. Bull. (CPBTAL) 33, 3775 (1985).
- DOS 3 324 034 (Otsuka; appl. 7.4.1983; J-prior. 7.5.1982).
- GB 2 123 825 (Otsuka; appl. 7.5.1983; J-prior. 7.5.1982).
-
oral and parenteral formulations:
- JP 60 019 767 (Otsuka; appl. 7.11.1983).
PAPER
Magic Bullet! Rebamipide, a Superior Anti-ulcer and Ophthalmic Drug and Its Large-Scale Synthesis in a Single Organic Solvent via Process Intensification Using Krapcho Decarboxylation
https://pubs.acs.org/doi/10.1021/acs.oprd.7b00382#
Rebamipide (1) is a superior drug compared to existing drugs for use in healing of peptic ulcers, gastrointestinal bleeding, and dyspepsia. It is also useful as an ophthalmic drug for the treatment of dry eye syndrome. Process intensification for its synthesis was achieved by (i) averting uncontrollable frothing using Krapcho decarboxylation instead of conventional acid hydrolysis, where uncontrollable frothing became chaotic, (ii) minimizing organic waste generation by using a single organic solvent, and (iii) avoiding anti-foaming agents (n-octanol, acetophenone) and acetic acid. With these trifling modifications, the overall yield of active pharmaceutical ingredient (API) was ≥83% with excellent purity (≥99.89%), and the process meets the metrics of “green” chemistry with an E-factor = 11.5. The developed hassle-free commercial process is viable for multi-kilogram synthesis of Rebamipide (1) as the key step, Krapcho decarboxylation is safe to run at 130–140 °C in DMSO, and it was proved to be effective by differential scanning calorimetry thermal screening studies. The characterization data of intermediates, process-related impurities, and API are reported. The carryover and process-related impurities were controlled efficiently. The present work can enhance the scope and worldwide adoptability of Rebamipide (1), which is currently limited to Asian countries.
https://pubs.acs.org/doi/suppl/10.1021/acs.oprd.7b00382/suppl_file/op7b00382_si_001.pdf
Articles
- Arakawa T, Watanabe T, Fukuda T, Yamasaki K, Kobayashi K (1995). “Rebamipide, novel prostaglandin-inducer accelerates healing and reduces relapse of acetic acid-induced rat gastric ulcer. Comparison with cimetidine”. Dig Dis Sci. 40 (11): 2469–72. doi:10.1007/BF02063257. PMID 7587834.
- Arakawa T, Kobayashi K, Yoshikawa T, Tarnawski A (1998). “Rebamipide: overview of its mechanisms of action and efficacy in mucosal protection and ulcer healing”. Dig Dis Sci. 43 (9 Suppl): 5S–13S. PMID 9753220.
- Tarnawski AS, Chai J, Pai R, Chiou SK (2004). “Rebamipide activates genes encoding angiogenic growth factors and Cox2 and stimulates angiogenesis: a key to its ulcer healing action?”. Dig Dis Sci. 49 (2): 202–9. doi:10.1023/B:DDAS.0000017439.60943.5c. PMID 15104358.
- Takumida M, Anniko M (2009). “Radical scavengers for elderly patients with age-related hearing loss”. Acta Otolaryngol. 129 (1): 36–44. doi:10.1080/00016480802008215. PMID 18607930.
References
- Jump up^ drugs.com
- Jump up^ “Russian State Register of Medicines. Registration Sertificate: Rebagit (rebamipide) Film-Coated Tablets” (in Russian). Retrieved 10 June 2017.
- Jump up^ Matsuda T, Ohno S, Hirohata S, Miyanaga Y, Ujihara H, Inaba G, Nakamura S, Tanaka S, Kogure M, Mizushima Y (2003). “Efficacy of rebamipide as adjunctive therapy in the treatment of recurrent oral aphthous ulcers in patients with Behcet’s disease: a randomised, double-blind, placebo-controlled study”. Drugs R D. 4 (1): 19–28. doi:10.2165/00126839-200304010-00002. PMID 12568631.
- Jump up^ http://www.wjgnet.com/1007-9327/12/656.pdf Archived October 20, 2013, at the Wayback Machine.
- Jump up^ Kinoshita, S.; K. Oshiden; S. Awamura; H. Suzuki; N. Nakamichi (2013). “A randomized, multicenter phase 3 study comparing 2% rebamipide (OPC-12759) with 0.1% sodium hyaluronate in the treatment of dry eye”. Ophthalmology. 120 (6): 1158–65. doi:10.1016/j.ophtha.2012.12.022. PMID 23490326.
 |
|
| Clinical data | |
|---|---|
| Trade names | Mucosta (JP), Rebagen (KR,CN, IN), Rebagit (RU) |
| AHFS/Drugs.com | International Drug Names |
| Routes of administration |
Oral (tablets) |
| ATC code | |
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C19H15ClN2O4 |
| Molar mass | 370.786 g/mol |
| 3D model (JSmol) | |
/////////Rebamipide, UNII:LR583V32ZR, ребамипид , ريباميبيد ,瑞巴派特 , OPC-12759 , OPC-12759E , OPC-759 , OPC 12759 , OPC 12759E , OPC 759 , OTSUKA, JAPAN 1990
OC(=O)C(CC1=CC(O)=NC2=CC=CC=C12)NC(=O)C1=CC=C(Cl)C=C1

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....








[…] addition to Japan, South Korea, China and India (brand names Mucosta and Rebagen), rebamipide is approved in Russia under the brand name Rebagit. It is marketed by Otsuka Pharmaceutical, based in […]
LikeLike