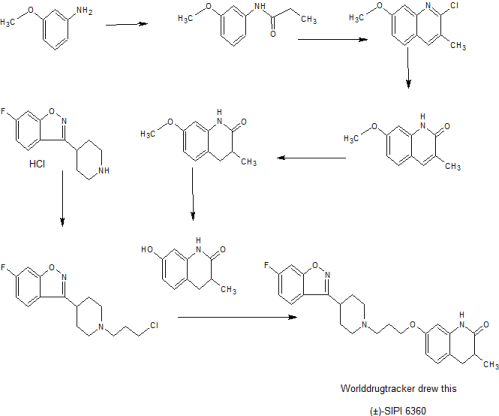
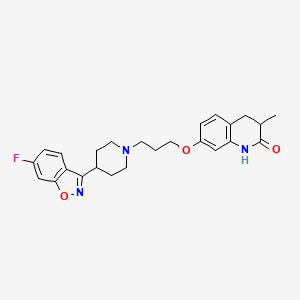
(±)-SIPI 6360
D2/5-HT2A receptor dual antagonist
7-[3-[4-(6-fluoro-1,2-benzoxazol-3-yl)piperidin-1-yl]propoxy]-3-methyl-3,4-dihydro-1H-quinolin-2-one
2(1H)-Quinolinone, 7-[3-[4-(6-fluoro-1,2-benzisoxazol-3-yl)-1-piperidinyl]propoxy]-3,4-dihydro-3-methyl-
| Molecular Formula: | C25H28FN3O3 |
|---|---|
| Molecular Weight: | 437.506523 g/mol |
Schizophrenia is a common severe mental patients, mental illness is the most serious of all, the most dangerous kind, the worldwide incidence of about I%, with the accelerate pace of social life, The incidence was significantly increased. Most schizophrenic patients due to the long treatment period, high cost, side effects and give up the treatment, often lead to more serious social consequences.
Numerous studies show that the brain monoamine neurotransmitters, especially dopamine and 5-hydroxytryptamine system is closely related to the body’s normal mental activity, these two types of system disorder can lead to a variety of neuropsychiatric diseases such as schizophrenia , neuropathic pain, mania, anxiety disorders, all kinds of depression, Parkinson’s disease and the like.
The drugs currently used clinically primarily for conventional antipsychotics (such as dopamine D2 receptor antagonists) and atypical antipsychotics (such as D2 / 5-HT2a dual antagonist), where conventional antipsychotics because it is easy leads to extrapyramidal symptoms (EPS) and gradually phased out, atypical antipsychotics variety, but no one medication to improve the overall spectrum of schizophrenia has the absolute advantage, most of the positive or negative symptoms of a a symptom improvement, or reduced side effects. So look for low toxicity, rapid onset, treatment spectral width of new anti-schizophrenia drug has been a hot topic in the world pharmaceutical industry.
In recent years, scientists have found that the dopamine D2 partial agonist can over time reduce dopamine activity in the transfer of dopamine, but not all block; the other hand, when the low dopaminergic activity is caused by stimulating effect on both positive and negative symptoms of mental illness have a significant effect. 5-HT2a receptor antagonists can improve negative symptoms, while synergies D2 EPS side effects can be reduced to about 1% level (classical antipsychotic drugs EPS incidence is about 30%), part of the 5-HTla agonism and 5-HT2a and synergy can make in therapeutic doses under observation EPS decreased to undetectable levels, therefore, has D2 ,5-HT2a, 5HTla synergy targets three new anti-drugs are currently developed Jingshenfenlie focus and an important development direction.
The present invention relates to a quinoline derivative can stabilize the brain dopaminergic, serotonergic energy system, may for a variety of neurological and psychiatric diseases have improved and treatment can be used for neuropathic pain, mania, schizophrenia, anxiety disorders, all kinds of depression, Parkinson’s disease, especially in the treatment of schizophrenia.
DETAILS COMING……….
PATENT

PATENT

Example 1
1-1
7- (3- (4- (6-fluorophenyl and [d] different dumb-3-yl) piperidin-1-yl) propoxy) -3,4-dihydro-3-carboxylic acid -one – yl quinolin -2 (1H)
1) N- (3- methoxyphenyl) propionamide
3-methoxy-aniline (0.1mol), methylene chloride (30 mL), triethylamine (0.2mol), was added to the flask lOOmL three, propionyl chloride was added dropwise under ice (0.12mol) in methylene chloride 30 mL, temperature does not exceed 5 ° C, the addition was complete, the ice bath was removed and stirred at room temperature 0.5h, the system was washed with water, dilute hydrochloric acid, saturated brine, dried over anhydrous magnesium sulfate, and evaporated to dryness to give a white powdery solid 17.01g yield 95%.
2) 2-chloro-7-methoxy-3-methylquinoline
The DMF (20mL) was added to the three 250mL flask, was added dropwise under ice-salt bath of POCl 3 (100 mL), temperature does not exceed 0 ° C, the addition was completed stirring 0.5h, was added portionwise N- (3- methoxyphenoxy yl) propanamide powder (31.0g), was slowly warmed to 50 ° C, violent reaction, to be exothermic easing slowly warmed to reflux, the reaction was kept 2h, cooled to room temperature, the system was poured into 800 g of crushed ice to sodium carbonate to adjust the pH to 7 to precipitate a yellow solid with petroleum ether – ethyl acetate to give pure product 20.86g, yield 58%.
3) 3-methyl-7-methoxy-quinolin -2 (1H) – one
2-Chloro-7-methoxy-3-methyl-quinoline (20.76g), acetic acid (150 mL) placed in 250mL one-neck flask, heated at reflux for 24h, acetic acid recovery, and the residue was recrystallized from ethanol to 95%, white needle crystalline 16.08g, yield 85%.
4) 7-methoxy-3,4-dihydro-3-methyl-quinolin -2 (1H) – one
7-Methoxy-3-methyl-quinolin -2 (1H) – one (18.92g), acetic acid (150mL), 10% Pd / C (lg) was added to the three 250mL flask, the system was replaced with nitrogen air, and then the nitrogen was replaced with hydrogen, and then the reaction was heated to 80 ° C overnight, cooled to room temperature, filtered and the filtrate evaporated to dryness to give a white powder, washed with water once, 50 ° C and dried in vacuo 4h, as a white powdery solid 18.91g yield of 98.95%.
5) 7-hydroxy-3,4-dihydro-3-methyl-quinolin -2 (1H) – one
7-Methoxy-3,4-dihydro-3-methyl-quinolin -2 (1H) – one (19.12g), 40% hydrobromic acid (150 mL) placed in 250mL one-neck flask was heated at reflux for 12h cooled to room temperature, the precipitated solid was filtered, the filter cake successively with hydrobromic acid, washed with water, 50 ° C and dried in vacuo 4h, 14.60 g as a white powdery solid, yield 82.4%.
6) 3- (1- (3-chloropropyl) piperidin-4-yl) -6-fluorophenyl and [d] oxazole different dumb
6-fluoro-3- (piperidin-4-yl) benzo [d] isoxazol dummy oxazole (22.00g), 1- bromo-3-chloropropane (40mL), anhydrous potassium carbonate (40g), acetone ( 250mL) was added to a 500mL one-neck flask was refluxed overnight, cooled to room temperature, filtered, the filter cake was washed twice with hot acetone and the combined filtrate was added dropwise a solution of anhydrous hydrogen chloride in ethanol, the precipitated white solid was filtered, the filter cake washed with acetone after washing once, it was dissolved in 200mL of water, adjusted with sodium carbonate to pH 9, and filtered to obtain a white powdery solid 15.94 g, yield 48.0%
7) 7- (3- (4- (6-fluorobenzo [d] isoxazol-3-yl dummy) piperidin-1-yl) propoxy) -3,4-dihydro-3-methyl quinolin -2 (1H) – one
3- (1- (3-chloropropyl) piperidin-4-yl) -6-fluorophenyl and [d] oxazole different dumb (lmmol), 7- hydroxy-3,4-dihydro-3-carboxylic acid yl quinolin -2 (1H) – one (1.0 mmol), anhydrous potassium carbonate (3.0mmol) were added to the lOmLDMF, 60 ° C overnight the reaction, potassium carbonate was filtered off, the mother liquor evaporated to dryness to give a pale yellow solid, the filter cake recrystallized with 95% ethanol, 50 ° C and dried in vacuo 4h, as a white powdery solid 0.30g, 69% yield.
NMR IH (of DMSO-D . 6 ): L up to .27 (D, 3H, J = 9.2Hz), 2.06-2.32 (m, 9H), 2.67-2.69 (T, 2H), 2.95 (D * D, lH, J = 3.2Hz, 12.8Hz), 3.15-3.17 ( m, 2H), 4.05 (t, 2H, J = 6Hz), 6. 37 (d, lH, J = 2.4Hz), 6.56 (d * d, lH, J = 2.4Hz, 8.0Hz), 7.05-7.11 (m, 2H), 7.25-7.29 (m, lH), 7.73-7.76 (m, lH), 7.98 (s, lH), 11.43 (brs, lH)
ESI-MS: 438 (M + 1)
Example 2
Preparation 1-1 hydrochloride
7- (3- (4- (6-fluorophenyl and [d] different dumb-3-yl) piperidin-1-yl) propoxy) -3,4-dihydro-3-methyl-quinoline morpholine -2 (1H) – one (lmmol) was dissolved with ethyl acetate (50 mL) was slowly added dropwise a solution of anhydrous hydrogen chloride in ethyl acetate (lmol / L, 5mL), stirred for 2h, the precipitated solid was filtered, the filter cake washed with ethyl acetate, 50 ° C and dried in vacuo 4h, as a white powdery solid 0.436g, yield 92%.
ESI-MS: 438 (M + 1)
Elemental analysis results:
Calcd: C, 63.35%; H, 6.17%; Cl, 7.48%; F, 4.01%; N, 8.87%; O, 10.13%
Found: C, 63.29%; H, 6.24%; CI, 7.43%; F, 4.05%; N, 8.82%; O, 10.17%
Example 3
Preparation 1-1 methanesulfonate
The 1-1 (lmmol) was dissolved with ethyl acetate (50 mL) was slowly added dropwise a solution of methanesulfonic acid in ethyl acetate (lmol / L, 5mL), stirred for 2h, the precipitated solid was filtered, the filter cake with ethyl acetate wash, 50 ° C and dried in vacuo 4h, as a white powdery solid 0.487g, yield 91.3%.
ESI-MS: 438 (M + 1, positive mode), 95 (CH 3 the SO 3 -, negative mode) Elemental analysis:
Calcd: C, 58.52%; H, 6.04%; F, 3.56%; N, 7.87%; 0, 17.99%; S, 6.01%
Found: C, 58.49%; H, 6.09%; F, 3.50%; N, 7.81%; 0, 18.02%; S, 6.09%
PATENT
Paper
Development and Kilogram-Scale Synthesis of a D2/5-HT2A Receptor Dual Antagonist (±)-SIPI 6360

The kilogram-scale synthesis of a D2/5-HT2A receptor dual antagonist (±)-SIPI 6360 was developed as an alternative treatment for schizophrenia. Specifically, three conditions were modified and optimized, including the Vilsmeier conditions, to prepare quinoline 3. In addition, the palladium-catalyzed hydrogenation was modified to synthesize dihydroquinolin-2(1H)-one 5, and the reduction of β-chloroamide was altered to form 3-chloropropanamine 8. Ultimately these improvements led to the preparation of a 1.5 kg of (±)-SIPI 6360 batch in eight steps with an overall yield of 34% and purity of 99.8%.
//////// D2/5-HT2A receptor dual antagonist (±)-SIPI 6360, 1401333-14-9
c21CC(C(Nc1cc(cc2)OCCCN3CCC(CC3)c4c5ccc(cc5on4)F)=O)C

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....