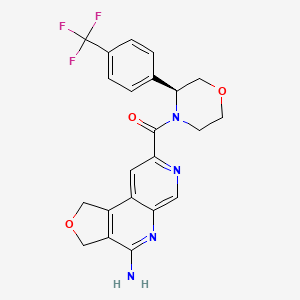
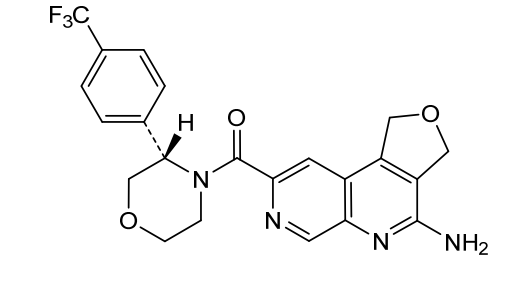
Anvumetostat
CAS 2790567-82-5
MF C22H19F3N4O3 MW444.4 g/mol
(4-amino-1,3-dihydrofuro[3,4-c][1,7]naphthyridin-8-yl)-[(3S)-3-[4-(trifluoromethyl)phenyl]morpholin-4-yl]methanone
(4-amino-1,3-dihydrofuro[3,4-c][1,7]naphthyridin-8-yl){(3S)-3-[4-(trifluoromethyl)phenyl]morpholin-4-yl}methanone
antineoplastic, AMG 193, QAT649EJ5E, PRMT5-IN-27,
Anvumetostat (also known as AMG 193) is an orally available, small-molecule inhibitor of protein arginine methyltransferase 5 (PRMT5), primarily being developed for the treatment of advanced solid tumours with MTAP-null (methylthioadenosine phosphorylase-deficient) mutations.
Mechanism of Action
- Targeting PRMT5: It is a potent and selective MTA-cooperative inhibitor of PRMT5.
- Synthetic Lethality: In cells where the MTAP gene is deleted (a common occurrence in various cancers), a metabolite called MTA (methylthioadenosine) accumulates. Anvumetostat selectively binds to the PRMT5-MTA complex, inhibiting its methyltransferase activity.
- Cellular Impact: By blocking PRMT5, the drug reduces the methylation of arginine residues in histones (H2A, H3, and H4), which can lead to decreased growth or death of cancer cells.
Clinical Development
Anvumetostat was initially developed by Amgen, Inc. and is currently in clinical trials. Institute (.gov) +1
- Current Status: As of early 2026, it is in Phase 2 of global research and development.
- Study Focus: Trials are evaluating its efficacy both as a monotherapy and in combination with other treatments for adult patients with metastatic or locally advanced MTAP-null cancers.
Key Identifiers
- Alternate Names: AMG 193, AMG-193.
- Chemical Class: Orally bioavailable small molecule.
- Genetic Biomarker: Specifically targets cancers with MTAP-null status
Anvumetostat is an orally available small molecule inhibitor of protein arginine methyltransferase 5 (PRMT5), with potential antiproliferative and antineoplastic activities. Upon oral administration, anvumetostat selectively binds to PRMT5 and inhibits its function. By inhibiting its methyltransferase activity, levels of both monomethylated and dimethylated arginine residues in histones H2A, H3 and H4 are decreased. This modulates the expression of genes involved in several cellular processes, including cellular proliferation. This may increase the expression of antiproliferative genes and/or decrease the expression of genes that promote cell proliferation, which may lead to decreased growth of rapidly proliferating cells, including cancer cells. PRMT5, a type II methyltransferase that catalyzes the formation of both omega-N monomethylarginine (MMA) and symmetric dimethylarginine (sDMA) on histones and a variety of other protein substrates involved in signal transduction and cellular transcription, is overexpressed in several neoplasms. Elevated levels are associated with decreased patient survival. Methylthioadenosine phosphorylase (MTAP) is deleted in certain cancer cells leading to an accumulation of methylthioadenosine (MTA). As MTA inhibits PRMT5, MTAP-null cancer cells are specifically sensitive to PRMT5 inhibitors.
SYN
[0163] Examples 481 and 482: (4-amino-l,3-dihydrofuro[3,4-c][l,7]naphthyridin-8-yl)(3-(4- (trifluoromethyl)phenyl)morpholino)methanone
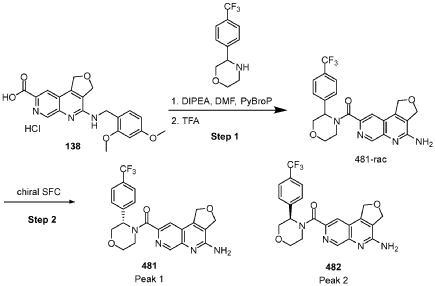
[0164] Step 1: To a solution of 3-(4-(trifluoromethyl)phenyl)morpholine (0.100 g, 0.432 mmol, Enamine), 4-((2,4-dimethoxybenzyl)amino)-l,3-dihydrofuro[3,4-c][l,7]naphthyridine-8-carboxylic acid hydrochloride (138) (0.271 g, 0.649 mmol) and l,l’-dimethyltriethylamine (0.559 g, 0.755 mL, 4.32 mmol, Sigma- Aldrich Corporation) in DMF (4 mL) was added bromotripyrrolidinophosphonium hexafluorophosphate (0.202 g, 0.432 mmol, Sigma-Aldrich Corporation) and the resulting mixture was heated at 50 °C for 30 min. The reaction was brought to rt, diluted with water, sat.NaHCCh and extracted with EtOAc (3x). The combined organics were dried over Na2SO4, filtered and concentrated. The residue was then chromatographed on silica gel using 0-50% 3:1 EtOAc/EtOH in heptane to afford (4-((2,4-dimethoxybenzyl)amino)- 1 ,3 -dihy drofuro [3 ,4-c] [ 1 ,7]naphthyridin-8-y 1) (3 – (4 -(trifluoromethyl)phenyl)morpholino)methanone (0.160 g, 0.269 mmol, 62.2% yield) as a light yellow solid, m/z (ESI): 595 (M+H)+.
[0165] To a solution of (4-((2,4-dimethoxybenzyl)amino)-l,3-dihydrofuro[3,4-c] [l,7]naphthyridin-8-yl)(3-(4-(trifluoromethyl)phenyl)morpholino)methanone (0.160 g, 0.269 mmol, 62.2 % yield) in DCM (2 mL) was added TFA (14.80 g, 10 mL, 130 mmol, Aldrich) and the resulting mixture was heated at 50 °C for 1 h. The reaction was concentrated, washed with 10% Na2CO3 and extracted with DCM. The combined organics were concentrated and chromatographed on silica gel using 0-50% 3:1 EtOAc/EtOH in heptane to afford (4-amino-l,3-dihydrofuro[3,4-c][l,7]naphthyridin-8-yl)(3-(4-(trifluoromethyl)phenyl)morpholino)methanone as the TFA salt (0.078 g, 0.140 mmol, 32.3% yield) as an off-white solid, m/z (ESI): 445 (M+H)+.
[0166] Step 2: (S)-(4-amino-l,3-dihydrofuro[3,4-c][l,7]naphthyridin-8-yl)(3-(4- (trifluoromethy l)phenyl)morpholino)methanone and (R)-(4-amino- 1 ,3 -dihy drofuro [3,4-c][l,7]naphthyridin-8-yl)(3-(4-(trifluoromethyl)phenyl)morpholino)methanone
(4-amino-l,3-dihydrofuro[3,4-c][l,7]naphthyridin-8-yl)(3-(4-(trifluoromethyl)phenyl)morpholino)methanone 2,2,2-trifluoroacetate were separated via preparative SFC using a Chiral Technologies AD column (150 x 21 mm, 5mm) with a mobile phase of 60% Liquid CO2 and 40% MeOH with 0.2% TEA using a flowrate of 80 mL/min to generate peak 1, (S)-(4-amino-l,3-dihydrofuro[3,4-c][l,7]naphthyridin-8-yl)(3-(4-(trifluoromethyl)phenyl)morpholino)methanone with an ee of >99%, and peak 2, (R)-(4-amino-l,3-dihydrofuro[3,4-c][l,7]naphthyridin-8-yl)(3-(4-(trifluoromethyl)phenyl)morpholino)methanone with an ee of 99.28%. Peak assignment determined by
SFC with AD column with 60% Liquid CO2 and 40% MeOH with 0.2% TEA and absolute
stereochemistry was arbitrarily assigned.
Peak 1: (S)-(4-amino-l,3-dihydrofuro[3,4-c][l,7]naphthyridin-8-yl)(3-(4-(trifluoromethyl)phenyl)morpholino)methanone (481) as a white solid . m/z (ESI): 445 (M+H)+. NMR 1H (400 MHz, DMSO-d6) 5 ppm 8.67 – 9.03 (m, 1 H), 7.85 (s, 1 H), 7.77 (br s, 4 H), 7.07 (br s, 2 H), 5.75 (s, 1 H), 5.37 (br s, 2 H), 5.04 (br s, 2 H), 4.46 – 4.61 (m, 1 H), 3.89 (br dd, J=12.2, 3.3 Hz, 4 H), 3.58 (br d, ./=5,8 Hz, 1 H). 19F NMR (377 MHz, DMSO-d6 ) 5 ppm -60.90 (br s, 3 F).
Peak 2: (R)-(4-amino- 1 ,3 -dihy drofuro [3 ,4-c] [ 1 ,7]naphthyridin-8-yl)(3 -(4-(trifluoromethyl)phenyl)morpholino)methanone (482) as a white solid, m/z (ESI): 445 (M+H)+. NMR 1H (400 MHz, DMSO-d6) 5 ppm 8.88 (br s, 1 H), 7.85 (s, 1 H), 7.77 (br d, J=1.7 Hz, 4 H), 7.07 (br s, 2 H),
5.69 – 5.78 (m, 1 H), 5.37 (br s, 2 H), 5.04 (br s, 2 H), 4.45 – 4.61 (m, 1 H), 3.89 (br dd, J=12.4, 3.3 Hz, 4 H), 3.51 – 3.64 (m, 1 H). 19F NMR (DMSO-d6, 377 MHz) 5 -60.90 (s, 3 F).
SYN
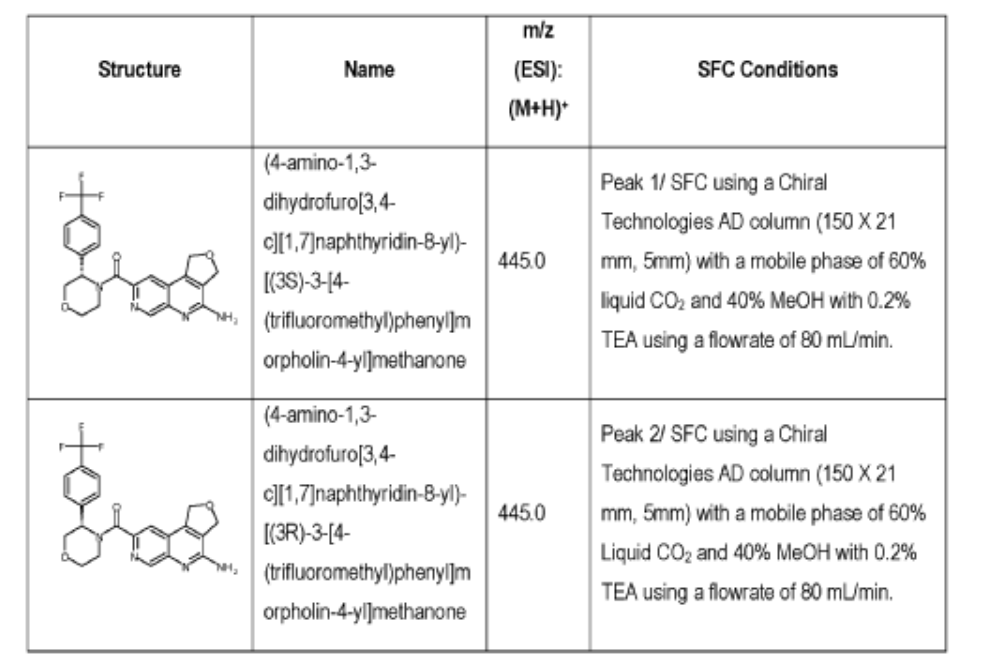
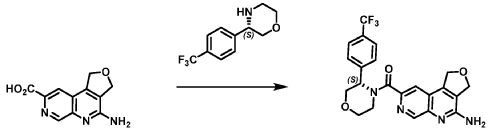
Example 4. Synthesis of Compound I – (4-amino-1 ,3-di hydrofuro[3,4-c][1 ,7]naphthyridin-8-yl)-[(3S)-3-[4-(trifluoromethyl)phenyl]morpholin-4-yl]methanone
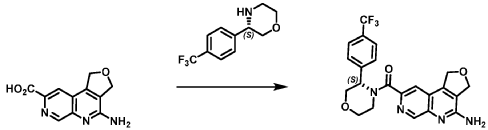
Reaction Scale 1
[0137] 4-Amino-1 ,3-dihydrofuro[3,4-c][1 ,7]naphthyridine-8-carboxylic acid (1.0 kg, 4.3 mol, 1.0 equiv), (3S)-3-[4-(trifluoromethyl)phenyl]morpholine (1.2 kg, 5.2 mmol, 1.2- equiv), and DMF, (6.6 kg, 7.0 V) were charged to a clean, dry reactor. To the mixture was added triethylamine (1.1 Kg, 13.8 mol, 2.6 equiv). The mixture was cooled to 10 ± 5 °C and O-(benzotriazol-1-yl)-N,N,N’,N’-tetramethyluronium tetrafluoroborate (TBTU) (1.67 kg, 5.2 mol, 1.2 equiv) was added slowly. Next, an additional amount of DMF (0.94 Kg, 1 V) was added. The reaction mixture was warmed to 25 ± 5 °C and stirred over 18 hours. Water (1 .0 kg, 1 V) was charged followed by MeCN (1 .6 kg, 2 V) and the reaction mass was warmed to 45 °C. Next, water (7.0 Kg, 7 V) was added over 30 min. A seed lot of 4-amino-1 ,3-dihydrofuro[3,4-c][1,7]naphthyridin-8-yl)-[(3S)-3-[4-(trifluoromethyl)phenyl]morpholin-4-yl]methanone (10 g, 22 mmol, 0.01 equiv), was charged and the mixture was stirred at 45 °C for over 2 hours before being cooled to 20 °C over 10 hours. Water (12.0 kg, 12 V) was added over 2 hours at 20 °C and further stirred for over 4 hours before being filtered. The reactor was rinsed with a mixture of 10% DMF in water (9.83 kg, 10 V) and the resulting rinse mixture was used to wash the cake. The reactor was rinsed with a mixture of water (10.0k kg, 10 V) and the resulting rinse mixture was used to wash the cake. This rinsing and washing protocol was repeated once more with water (10.0k kg, 10V). The cake was dried under vacuum with a stream of nitrogen to afford (4-amino-1 ,3-dihydrofuro[3,4-c][1 ,7]naphthyridin-8-yl)-[(3S)-3-[4-
(trifluoromethyl)phenyl]morpholin-4-yl]methanone. LCMS: 445.20 1H NMR (400 MHz, DMS0-d6 at 130 °C): 8.87 (s, 1 H), 7.80 (s, 1 H), 7.73 (d, 0=8.7 Hz, 2H), 7.71 (d, 0=8.7 Hz, 2H), 6.58 (br s, 2H), 5.72 (br s, 1 H), 5.38 (m, 2H), 5.09 (t, 0=3.5 Hz, 2H), 4.44 (br d, 0=12.3 Hz, 1 H), 4.08 (br d, 0=13.4 Hz, 1 H), 3.96 (dd, 0=12.3, 3.7 Hz, 1 H), 3.86 (br dd, 0=11 .4, 3.0 Hz, 1 H), 3.66 (td, 0=11 .4, 3.0 Hz, 1 H), 3.28 (m, 1 H).
Reaction Scale 2
[0138] 4-Amino-1 ,3-dihydrofuro[3,4-c][1 ,7]naphthyridine-8-carboxylic acid (85.0 g, 352.2 mmol, 1.0 equiv), (3S)-3-[4-(trifluoromethyl)phenyl]morpholine (99.6 g, 422.6 mmol, 1.2- equiv), and DMF, (674 mL, 8.7 mol, 7.9 V) were charged to a clean, dry 5 L reactor. To the mixture was added 1 -methylimidazole (75.2 g, 916.2 mmol, 2.6 equiv). The mixture was cooled to 0 °C and N,N,N’,N’-tetramethylchloroformamidinium hexafluorophosphate (TCFH) (118.6 g, 422.6 mmol, 1.2 equiv) was added slowly. Next, an additional amount of DMF (170 mL, 2 V) was added at 0 °C. The reaction mixture was warmed to 25 °C and stirred overnight. Next, the reaction mass was warmed to 45 °C and 2-methyltetrahydrofuran, (169.2 mL, 2 V) was added followed by slow addition of water (850 mL, 10 V) over 30 min by addition funnel. A seed lot of 4-amino-1 ,3-dihydrofuro[3,4-c][1 ,7]naphthyridin-8-yl)-[(3S)-3-[4-(trifluoromethyl)phenyl]morpholin-4-yl]methanone (1.6 g, 3.5 mmol, 0.1 equiv), was charged as a slurry in a 1 :1 v/v of DMF and water (31 .3 mL) and the mixture was stirred at 45 °C for approximately 12 hrs. Water (510 mL, 6 V) was added over 1 h 10 min by addition funnel and the mixture was further stirred at 45°C for 30 min before being filtered. The reactor was rinsed with water (340 mL, 4 V) and the resulting rinse mixture was used to wash the cake. This rinsing and washing protocol was repeated twice more. The cake was dried under vacuum with a stream of nitrogen to afford (4-amino-1 ,3-dihydrofuro[3,4-c][1 ,7]naphthyridin-8-yl)-[(3S)-3-[4-(trifluoromethyl)phenyl]morpholin-4-yl]methanone. LCMS: 445.20 1H NMR (400 MHz, DMSO-d6 at 130 °C): 8.87 (s, 1 H), 7.80 (s, 1 H), 7.73 (d, J=8.7 Hz, 2H), 7.71 (d, J=8.7 Hz, 2H), 6.58 (br s, 2H), 5.72 (br s, 1 H), 5.38 (m, 2H), 5.09 (t, J=3.5 Hz, 2H), 4.44 (br d, J=12.3 Hz, 1 H), 4.08 (br d, J=13.4 Hz, 1 H), 3.96 (dd, J=12.3, 3.7 Hz, 1 H), 3.86 (br dd, J=11.4, 3.0 Hz, 1 H), 3.66 (td, J=11.4, 3.0 Hz, 1 H), 3.28 (m, 1 H).
Reaction Scale 3:
[0139] 4-Amino-1 ,3-dihydrofuro[3,4-c][1 ,7]naphthyridine-8-carboxylic acid (Compound A’) (20.0 g, 86.5 mmol, 1.0 equiv) was added to dimethylsulfoxide (400 mL) at 20 °C. To the mixture was added 1 ,T-carbonyldiimidazole (15.4 g, 95.2 mmol, 1.1 equiv) and the mixture was heated to 60 °C for 1 hour. A solution of (S)-3-(4-(trifluoromethyl)phenyl)morpholin-4-ium chloride (25.5 g, 95.2 mmol, 1.1 equiv) and dimethylsulfoxide (40 mL) was added, and the mixture was heated to 80 °C for 11 hours. The reaction mixture was cooled to 35 °C, then water (265 mL) was added, then the batch was cooled to 20 °C. The reaction was filtered, washed with 40% water:DMSO (80 mL), then washed with water (100 mL). The cake was dried under vacuum with a stream of nitrogen to afford (4-amino-1 ,3-dihydrofuro[3,4-c][1 ,7]naphthyridin-8-yl)-[(3S)-3-[4-(trifluoromethyl)phenyl]morpholin-4-yl]methanone (Compound I). LCMS: 445.20 1H NMR (400 MHz, DMSO-d6 at 130 °C): 8.87 (s, 1 H), 7.80 (s, 1 H), 7.73 (d, J=8.7 Hz, 2H), 7.71 (d, J=8.7 Hz, 2H), 6.58 (br s, 2H), 5.72 (br s, 1 H), 5.38 (m, 2H), 5.09 (t, >3.5 Hz, 2H), 4.44 (br d, >12.3 Hz, 1H), 4.08 (br d, >13.4 Hz, 1 H), 3.96 (dd, >12.3, 3.7 Hz, 1 H), 3.86 (br dd, >11 .4, 3.0 Hz, 1 H), 3.66 (td, >11 .4, 3.0 Hz, 1 H), 3.28 (m, 1 H).
Recrystallization of Compound I
[0140] A clean, dry 5 L reactor was charged with (4-amino-1 ,3-dihydrofuro[3,4-c][1 ,7]naphthyridin-8-yl)-[(3S)-3-[4-(trifluoromethyl)phenyl]morpholin-4-yl]methanone (279.7 g, 0.6 mol, 1.0 equiv) followed by acetone (6.2 L,
22 V). The mixture was stirred at 40 °C for 15 minutes before cooling to 25 °C. The reactor was discharged into a flask and the reactor was rinsed with acetone and the process stream was polish-filtered back into the reactor.
The reactor jacket was set to 65 °C and the reaction volume was reduced to approximately 6 V by distillation at atmospheric pressure, crystallization was observed. The reaction temperature was set to cool to 20 °C over two hours. Heptane (2.8 L, 10 V) was added over two hours. The slurry was filtered and the cake was washed twice with a 4:1 Heptane/acetone mix (750 mL, 3 V each) and dried under vacuum with a nitrogen purge to afford (4-amino-1,3-dihydrofuro[3,4-c][1,7]naphthyridin-8-yl)-[(3S)-3-[4-(trifluoromethyl)phenyl]morpholin-4-yl] methanone.



ADVT
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
#MedicinalChemistry, #DrugDiscovery, #OrganicSynthesis, #ChemicalLibrary, #BuildingBlocks, #SARStudies, #ChemistryInnovation, #medchem, #Drugdevelopment, #Biotech, #Biotechnology, #AnaxLaboratories, #Pharma



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
- Process for synthesizing naphthyridine derivatives and intermediates thereofPublication Number: EP-4396170-A1Priority Date: 2021-08-30
- PRMTS inhibitorsPublication Number: CN-116888120-APriority Date: 2020-12-16
- Prmts inhibitorsPublication Number: WO-2022132914-A1Priority Date: 2020-12-16
- Prmts inhibitorsPublication Number: EP-4263545-A1Priority Date: 2020-12-16
- Mta-cooperative prmt5 inhibitors for use in the treatment of cancerPublication Number: EP-4572760-A1Priority Date: 2022-08-15
- Cancer treatments using mta-cooperative prmt5 inhibitorsPublication Number: WO-2023196545-A1Priority Date: 2022-04-08
- Process for the synthesis of naphthyridine derivatives and intermediates thereofPublication Number: CN-117897379-APriority Date: 2021-08-30
- Process for synthesizing naphthyridine derivatives and intermediates thereofPublication Number: WO-2023034786-A1Priority Date: 2021-08-30
- Process for Synthesizing Naphthyridine Derivatives and Intermediates ThereofPublication Number: US-2024360147-A1Priority Date: 2021-08-30
- Prmt5 inhibitor for use in cancer therapyPublication Number: WO-2024170488-A1Priority Date: 2023-02-13
- Cancer treatments using a prmt5 inhibitor and a mat2a inhibitorPublication Number: WO-2024118897-A1Priority Date: 2022-11-30
- Cancer treatments using a prmt5 inhibitor and a mat2a inhibitorPublication Number: EP-4626435-A1Priority Date: 2022-11-30
- MTA synergizes with PRMT5 inhibitors for cancer treatmentPublication Number: CN-119730853-APriority Date: 2022-08-15
- Mta-cooperative prmt5 inhibitors for use in the treatment of cancerPublication Number: WO-2024038004-A1Priority Date: 2022-08-15
////////anvumetostat, ANAX LAB, antineoplastic, AMG 193, QAT649EJ5E, PRMT5-IN-27,

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
