Medigen vaccine
MVC COVID-19 vaccine
- MVC-COV1901
track it https://covid19.trackvaccines.org/vaccines/24/
MVC-COV1901 is a vaccine candidate developed and commercialized by Medigen Vaccine Biologics Corporation. The vaccine candidate contains a perfusion form of the SARS-Cov2 recombinant spike protein. Medigen has combined forces with Dynavax, which offers an advanced adjuvant, CpG 1018 (also known as ISS-1018), for use with its vaccine. As of September 2020, the vaccine candidate is in Phase 1 clinical trials to assess its safety and immunogenicity (NCT04487210).
The MVC COVID-19 vaccine, designated MVC-COV1901 and also known as the Medigen COVID-19 vaccine, is a protein subunit COVID-19 vaccine developed by Medigen Vaccine Biologics Corporation [zh] in Taiwan, American company Dynavax Technologies and the U.S. National Institute of Health.[1][2]
This vaccine is made by the recombinant S-2P spike protein adjuvanted with CpG 1018 supplied by Dynavax.[3] Preliminary results from Phase I trials on 77 participants were published in June 2021, indicating what the authors described as “robust” immune system response elicited by the vaccine.[4]
The study authors have assessed the humoral immune response by measuring quantities of binding IgG to S protein, and also the cellular immune response by measuring the quantities of IFN-γ and IL-4 secreting T cells.[4]
Taiwan-based Medigen Vaccine Biologics Corporation (MVC) and Dynavax Technologies Corporation, in the US, have announced the rollout of its COVID-19 vaccine, MVC-COV1901. Approximately 600,000 people are anticipated to receive the Medigen vaccine this week.
Ryan Spencer, Chief Executive Officer of Dynavax commented, “We are pleased that Medigen’s vaccine is now available for the people of Taiwan. We are very excited for this first, of hopefully multiple, EUAs and approvals for COVID-19 vaccines that include CpG 1018 adjuvant. Considering the limitations of current vaccines and the global vaccine shortage, we believe adjuvanted vaccines can contribute significantly to current vaccination efforts.”
In July, MVC received Taiwan Emergency Use Authorization and approval for inclusion in Taiwan’s COVID-19 vaccine immunization program, MVC-COV1901.
MVC COVID-19 vaccine is indicated for adults over 20 years old and is administered in two doses 28 days apart for prevention of COVID-19.
The Advisory Committee recommended that MVC should submit safety monitoring report monthly during the declared EUA period and should submit a vaccine effectiveness report within one year after obtaining EUA approval.
(CNN)Taiwan’s President Tsai Ing-wen received her first shot of the island’s homegrown Covid-19 vaccine on Monday, a public show of support for the new drug which is central to plans for inoculation self sufficiency amid low immunization rates and struggles to obtain vaccines from overseas.Monday’s island-wide rollout of the Medigen Covid-19 vaccine, developed by Taipei-based Medigen Vaccine Biologics Corporation, comes after the drug was approved for emergency use last month by Taiwanese authorities for anyone above 20 years old, with at least 28 days between the two doses.The vaccine has yet to complete phase 3 clinical trials and no efficacy data is available. Paul Torkehagen, Medigen’s director of overseas business development, told CNN in May that the company designed a “very large” phase 2 clinical trial to ensure the vaccine’s safety and effectiveness, with 3,800 participants. Normally, a stage 2 clinical trial only involves several hundred people. Data from the trials showed that 99.8% of participants were able to form antibodies against Covid-19 after taking two doses of the vaccine, Medigen’s CEO Charles Chen said. 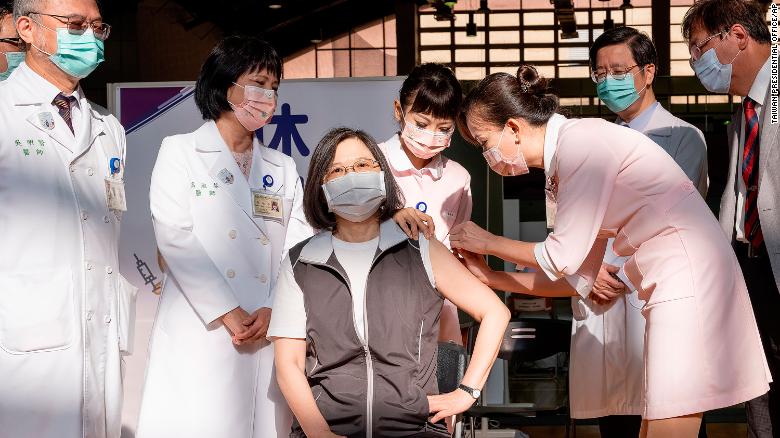
Taiwanese President Tsai Ing-wen, center, receives her first shot of the island’s first domestically developed coronavirus vaccine at the Taiwan University Hospital in Taipei, Taiwan on Monday, August 23.
Taiwan’s Centers for Disease Control said in a July 19 statement that the vaccine posed no serious health effects. Taiwan has ordered 5 million doses of the vaccine from Medigen and more than 700,000 people have already signed up to receive it, according to Reuters.In a Facebook post after receiving the vaccine at a hospital in Taipei, Tsai said she hadn’t suffered from any post-vaccination pain and thanked the health care workers who had administered the shot.”Taking the vaccine can protect yourself, your family, as well as medical staff,” Tsai wrote. “Let’s do our part in boosting Taiwan’s collective defense against the virus!”With its borders sealed to most travelers and strict measures enacted to contain local outbreaks, Taiwan has so far been largely successful in containing Covid-19, reporting fewer than 16,000 total confirmed infections and 828 deaths. But the island has struggled to vaccinate its more than 23 million population, partly due to difficulties obtaining doses from international suppliers.Taiwan’s government has only managed to import around 10 million Covid-19 vaccines, according to Reuters. In July it ordered another 36 million doses of the Moderna shot.Fewer than 5% of Taiwan’s population has received both doses of their Covid-19 vaccine, according to Reuters, as the island delays second dose vaccinations so more people can receive a first shot.On Monday, Taiwan reported four new Covid-19 cases, according to the Central Epidemic Command Center (CECC). Authorities announced on the weekend they would ease virus prevention measures to allow for larger gatherings and the opening of study centers and indoor amusement parks.But Health and Welfare Minister Chen Shih-chung said current Covid-19 restrictions — which include the closure of bars and nightclubs — would remain in place until at least September 6, with the possibility of an extension if the global outbreak continued to grow.Taiwan could become increasingly isolated if it keeps pursuing its “Covid zero” strategy, with both Australia and New Zealand hinting they might abandon the approach once vaccinations reach a certain level.In an opinion piece published on Sunday, Australian Prime Minister Scott Morrison said that while lockdowns to prevent Covid-19 transmission were “sadly necessary for now,” they may not be once vaccination rates increased to the targets of 70% and 80%.”This is what living with Covid is all about. The case numbers will likely rise when we soon begin to open up. That is inevitable,” he said.In neighboring New Zealand, which has also attempted to eliminate the virus within its borders, Covid-19 response minister Chris Hipkins told local media the highly-contagious Delta variant raised “some pretty big questions about what the long-term future of our plans are.”“At some point we will have to start to be more open in the future,” he said.
History
On 16 February 2020, Medigen Vaccine Biologics Corp. (MVC) signed a collaboration agreement with National Institutes of Health (NIH) for COVID-19 vaccine development. The partnership will allow MVC to obtain NIH’s COVID-19 vaccine and related biological materials to conduct animal studies in Taiwan.[5]
On 23 July 2020, Medigen Vaccine Biologics (MVC) announced collaboration with Dynavax Technologies to develop COVID-19 vaccine. The COVID-19 candidate vaccine will have the combination of SARS-CoV2 spike protein created by MVC and Dynavax’s vaccine adjuvant CpG 1018, which was used in a previously FDA-approved adult hepatitis B vaccine.[6][7]
Clinical trials
On 13 October 2020, Medigen Vaccine Biologics received Taiwan’s government subsidies for the initiation of Phase 1 Clinical Trial in Taiwan starting early October. The Phase 1 Clinical Trial was held at National Taiwan University Hospital with 45 participants ranging the age of 20-50.[8][9]
On 25 January 2021, Medigen Vaccine Biologics initiated Phase 2 Clinical Trial for its COVID-19 vaccine candidate MVC-COV1901 with the first participant being dosed. The Phase 2 Clinical Trial for the MVC COVID-19 vaccine was a randomized, double-blinded, and multi-center clinical trial, planned to enroll 3,700 participants of any age 20 above.[3][10][11]
On 10 June 2021, Medigen Vaccine Biologics released its COVID-19 vaccine Phase 2 interim analysis results, which demonstrates good safety profile in participants. The Phase 2 Clinical Trial in the end included 3,800 participants with all participants receiving second dose by 28 April 2021. Medigen Vaccine Biologics announced that it will request Emergency Use Authorization (EUA) with the concluding of the Phase 2 Clinical Trial.[12]
On 20 July 2021, Medigen Vaccine Biologics filed a Phase 3 Clinical Trial IND application with Paraguay’s regulatory authority, which was later approved. The Phase 3 Clinical Trial, however, was different from regular Phase 3 Clinical Trial, which uses immune-bridging trial to compare the performance of MVC COVID-19 vaccine with the Oxford-AstraZeneca COVID-19 vaccine.[13] The decision was a controversial announcement as immune-bridging trials were not fully approved or widely accepted by health authorities. In addition, the accuracy of immune-bridging trials were also been questioned for years.[citation needed]
Adolescents trial
In July 2021, Medigen commenced phase II trials for adolescents aged 12-18.[14]
Authorization
| Full authorization Emergency authorization |
See also: List of COVID-19 vaccine authorizations § Medigen
On July 19, 2021, MVC COVID-19 vaccine obtained Emergency Use Authorization (EUA) approval from the Taiwanese government after fulfilling EUA requirements set by Taiwanese authority.[15] The EUA, however, was met with controversy due to the lack of efficacy data and Phase 3 Clinical Trial. On August 23, 2021, President Tsai Ing-Wen was among the first Taiwanese to receive a dose of the vaccine. [16]
References
- ^ “Dynavax and Medigen Announce Collaboration to Develop a Novel Adjuvanted COVID-19 Vaccine Candidate”. GlobeNewswire. 23 July 2020. Retrieved 7 June 2021.
- ^ 黃驛淵 (10 June 2021). “【獨家】【國產疫苗解盲1】高端實體疫苗針劑首曝光 「每天9萬劑」生產基地直擊” (in Chinese). Mirror Media.
- ^ Jump up to:a b “Medigen Vaccine Biologics COVID-19 Vaccine Adjuvanted with Dynavax’s CpG 1018 Announces First Participant Dosed in Phase 2 Clinical Trial in Taiwan”. http://www.medigenvac.com. Retrieved 7 August 2021.
- ^ Jump up to:a b Hsieh SM, Liu WD, Huang YS, Lin YJ, Hsieh EF, Lian WC, Chen C, Janssen R, Shih SR, Huang CG, Tai IC, Chang SC (25 June 2021). “Safety and immunogenicity of a Recombinant Stabilized Prefusion SARS-CoV-2 Spike Protein Vaccine (MVCCOV1901) Adjuvanted with CpG 1018 and Aluminum Hydroxide in healthy adults: A Phase 1, dose-escalation study”. EClinicalMedicine: 100989. doi:10.1016/j.eclinm.2021.100989. ISSN 2589-5370. PMC 8233066. PMID 34222848.
- ^ “MVC and NIH Collaborate to Develop COVID-19 Vaccine”. http://www.medigenvac.com. Retrieved 7 August 2021.
- ^ “Medigen Collaborates with Dynavax to Develop Novel Adjuvanted COVID-19 Vaccine Candidate”. http://www.medigenvac.com. Retrieved 7 August 2021.
- ^ “MVC Signed an License Agreement with NIH on COVID-19 Vaccine”. Medigen. 5 May 2020. Retrieved 27 July 2021.
- ^ “Medigen’s COVID-19 Vaccine Combined with Dynavax’s CpG 1018 Adjuvant Receives Taiwan Government Subsidy with First Participant Dosed in Early October”. http://www.medigenvac.com. Retrieved 7 August 2021.
- ^ “A Study to Evaluate MVC-COV1901 Vaccine Against COVID-19 in Adult (COVID-19)”. clinicaltrials.gov. United States National Library of Medicine. Retrieved 11 March 2021.
- ^ “A Study to Evaluate the Safety and Immunogenicity of MVC-COV1901 Against COVID-19”. clinicaltrials.gov. United States National Library of Medicine. Retrieved 11 March 2021.
- ^ “A Study to Evaluate MVC-COV1901 Vaccine Against COVID-19 in Elderly Adults”. clinicaltrials.gov. United States National Library of Medicine. 28 March 2021. Retrieved 3 April 2021.
- ^ “MVC Released COVID-19 Vaccine Phase 2 Interim Analysis Result”. http://www.medigenvac.com. Retrieved 7 August 2021.
- ^ “MVC Announces Paraguay Approval of IND Application for Phase 3 Clinical Trial”. http://www.medigenvac.com. Retrieved 7 August 2021.
- ^ “A Study to Evaluate MVC-COV1901 Vaccine Against COVID-19 in Adolescents”. clinicaltrials.gov. United States National Library of Medicine. 6 July 2021. Retrieved 6 July 2021.
- ^ “MVC COVID-19 Vaccine Obtains Taiwan EUA Approval”. http://www.medigenvac.com. Retrieved 7 August 2021.
- ^ Taiwan begins contested rollout of new Medigen domestic vaccine, Nikkei Asia, Erin Hale, August 23, 2021
| Vaccine description | |
|---|---|
| Target | SARS-CoV-2 |
| Vaccine type | Protein subunit |
| Clinical data | |
| Other names | MVC-COV1901 |
| Routes of administration | Intramuscular |
| Legal status | |
| Legal status | Full and Emergency Authorizations: List of MVC COVID-19 vaccine authorizations |
| Identifiers | |
| DrugBank | DB15854 |
| Part of a series on the |
| COVID-19 pandemic |
|---|
| COVID-19 (disease)SARS-CoV-2 (virus)CasesDeaths |
| showTimeline |
| showLocations |
| showInternational response |
| showMedical response |
| showEconomic impact and recession |
| showImpacts |
| COVID-19 portal |
////////Medigen vaccine, MVC COVID-19 vaccine, SARS-CoV-2, covid 19, corona virus, taiwan, approvals 2021, iss 1018, CpG 1018, MVC-COV1901
NEW DRUG APPROVALS
one time
$10.00

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
