
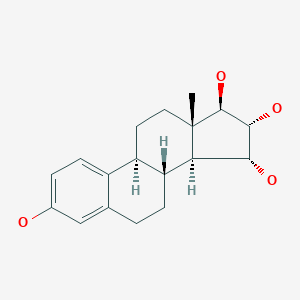
Estetrol
エステトロール;
| Formula | C18H24O4 |
|---|---|
| CAS | 15183-37-6 |
| Mol weight | 304.3808 |
FDA 4/15/2021, To prevent pregnancy, Nextstellis
New Drug Application (NDA): 214154
Company: MAYNE PHARMA
PATENT
https://patents.google.com/patent/EP1562976B1/en
Estrogenic substances are commonly used in methods of Hormone Replacement Therapy (HRT) and methods of female contraception. These estrogenic substances can be divided in natural estrogens and synthetic estrogens. Examples of natural estrogens that have found pharmaceutical application include estradiol, estrone, estriol and conjugated equine estrogens. Examples of synthetic estrogens, which offer the advantage of high oral bioavailability include ethinyl estradiol and mestranol.Recently, estetrol has been found effective as an estrogenic substance for use in HRT, disclosure of which is given in the Applicant’s co-pending application WO 02/094276 . Estetrol is a biogenic estrogen that is endogeneously produced by the fetal liver during human pregnancy. Other important applications of estetrol are in the fields of contraception, therapy of auto-immune diseases, prevention and therapy of breast and colon tumors, enhancement of libido, skin care, and wound healing as described in the Applicant’s co-pending applications WO 02/094276 , WO 02/094279 , WO 02/094278 , WO 02/094275 , EP 1511496 A1 , EP 1511498 A1 , WO 03/041718 , WO 03/018026 , EP 1526856 A1 and WO 04/0278032 .[0004]The synthesis of estetrol and derivatives thereof on a laboratory scale basis is known in the art: Fishman J., Guzik H., J. Org. Chem. 33, 3133 – 3135 (1968); Nambara T. et al., Steroids 27, 111 – 121 (1976); or Suzuki E. et al., Steroids 60, 277 – 284(1995).[0005]
Fishman J., Guzik H., J. Org. Chem. 33, 3133 – 3135 (1968) discloses a successful synthesis of estetrol from an estrone derivative (compound (III); cf. for a synthesis of compound (III) Cantrall, E.W., Littell, R., Bernstein, S. J. Org. Chem 29, 214 – 217 (1964)). In a first step, the carbonyl group at C17 of compound (III) was reduced with LiAlH4 to estra-1,3,5(10),15-tetraene-3,17-diol (compound VIa) that was isolated as the diacetate (compound VIb). Compound VIb was subjected to cis-hydroxylation of the double bond of ring D by using OsO4 which resulted into the formation of estra-1,3,5(10)-triene-3,15α,16α,17β-tetraol-3,17-diacetate (compound Ib) that under heating with K2CO3 in methanol produces estetrol (Scheme 1).

[0006]
The overall yield of this three step process is, starting from estrone derivative III, only about 7%. It is worth noting that the protected derivative 17,17-ethylenedioxyestra-1,3,5(10),15-tetraene-3-ol-3-acetate (compound IV) could be cis-hydroxylated to its 15α,16α-diol derivative (compound Va), but that thereafter the dioxolane group could not be removed (p-toluene sulfonic acid in acetone at room temperature) or that the hydrolysis (aqueous sulfuric acid in warm dioxane) of the dioxolane group resulted in a mixture containing a multitude of products (Scheme 2).

[0007]Nambara T. et al., Steroids 27, 111 – 121 (1976) discloses another synthesis of estetrol wherein estrone is the starting material. The carbonyl group of estrone is first protected by treatment with ethylene glycol and pyridine hydrochloride followed by acetylation of the hydroxy group at C3. The next sequence of steps involved a bromination/base catalyzed dehydrobromination resulting into the formation of 17,17-ethylenedioxyestra-1,3,5(10),15-tetraene-3-ol (compound IVa). This compound IVa was subsequently acetylated which produced 17,17-ethylenedioxyestra-1,3,5(10),15-tetraene-3-ol-3-acetate (compound IVb). In a next step, the dioxolane group of compound IVb was hydrolysed by using p-toluene sulfonic acid to compound Vb, followed subsequently by reduction of the carbonyl group at C17 (compound Vc) and oxidation of the double bond of ring D thereby forming estra-1,3,5(10)-triene-3,15α,16α,17β-tetraol-3,17-diacetate (compound VIb). See Scheme 3.[0008]
Suzuki E. et al., Steroids 60, 277 – 284 (1995) also discloses the synthesis of estetrol by using compound Vb of Nambara T. et al. as starting material. The carbonyl group at C17 of this compound was first reduced followed by acetylation yielding estra-1,3,5(10),15-tetraene-3,17-diol-3,17-diacetate (compound 2b). The latter was subjected to oxidation with OsO4 which provided estra-1,3,5(10)-triene-3,15α,16α,17β-tetraol-3,17-diacetate (compound 3b) in 46% yield.

[0009]According to the Nambara T. et al. and Suzuki E. et al., the synthesis of estetrol can be performed with a yield of approximately 8%, starting from estrone.0010]
Poirier D., et al., Tetrahedron 47, 7751 – 7766 (1991) discloses the following compounds which were prepared according to methods that have been used to prepare similar compounds:

[0011]Dionne, P. et al., Steriods 62, 674 – 681 (1997) discloses the compound shown above wherein R is either methyl or t-butyldimethylsilyl.[0012]Magnus, P. et al., J. Am. Chem. Soc. 120, 12486 -12499 (1998) discloses that the main methods for the synthesis of α,β-unsaturated ketones from saturated ketones are (a) halogenation followed by dehydrohalogenation, (b) utilising sulphur or selenium derivatives, (c) DDQ and (d) utilizing palladium(II) complexes.[0013]Furthermore, it has also been found that by following the prior art methods mentioned above, estetrol of high purity was obtained only in low yield when using an acetyl group as a protecting group for the 3-hydroxy group of estra-1,3,5(10),15-tetraen-3-ol-17-one, in particular because its sensitivity to hydrolysis and solvolysis. In particular, the lability of the acetyl group lead not only to an increased formation of byproducts during the reactions, but also during chromatography and crystallisation for purification of intermediate products when protic solvents such as methanol were used. Therefore, it is difficult to isolate purified estetrol and intermediates thereof in good yield.
Example 7 3-Benzyloxy-estra-1,3,5 (10),15-tetraen-17-ol (compound 5; A = benzyl)
[0088]To a solution of 3-benzyl-dehydroestrone (compound 6; A = benzyl; 58 g, 162 mmol) in a mixture of MeOH (900 mL) and THF (200 mL) at room temperature was added CeCl3 heptahydrate (66.4 g, 178 mmol). After stirring for 1 h the mixture was cooled to 0-5°C using an ice/water bath. Then NaBH4 (12.2 g, 324 mmol) was added in small portions maintaining a temperature below 8°C. After stirring for 2 h at 0-5°C (TLC showed the reaction to be complete) 1 N NaOH (300 mL) and DCM (1 L) were added and the mixture was stirred for ½ h at room temperature. The layers were separated and the aqueous layer was extracted with DCM (200 mL). The organic layers were combined, dried (Na2SO4) and concentrated in vacuo to give an off-white solid (55.0 g, 152.8 mmol, 94%) TLC: Rf = 0.25 (heptanes/ethyl acetate = 4:1); HPLC-MS: 93% β-isomer, 2% α-isomer; DSC: Mp. 149.7°C, purity 96.6%; 1H-NMR (200 MHz, CDCl3) δ 7.48 (m, 5H), 7.27 (d, 1H, J = 8.4 Hz), 6.85 (dd, 1H, J1 = 2.8 Hz, J2 = 8.6 Hz), 6.81 (d, 1H, J = 2.4 Hz), 6.10 (d, 1H, J = 5.8 Hz), 5.79 (dd, 1H, J1 = 1.8 Hz, J2 = 3.4 Hz), 5.11 (s, 2H), 4.48 (d, 1H, J = 7.6), 2.96 (m, 2H), 2.46 – 1.64 (m, 9H), 0.93 (s, 3H) ppm.
Example 8 17-Acetyloxy-3-benzyloxy-estra-1,3,5 (10),15-tetraene (compound 4; A = benzyl, C = acetyl)
[0089]A solution of 3-Benzyloxy-estra-1,3,5 (10),15-tetraen-17-ol (compound 5; A = benzyl; 55.0 g, max. 153 mmol) in pyridine (400 mL) was treated with Ac2O (50 mL, 0.53 mol) and 4-dimethylaminopyridine (1.5 g, 12.3 mmol). The mixture was stirred for 2 h at room temperature (TLC showed the reaction to be complete). It was concentrated in vacuo. The residue was dissolved in EtOAc (400 mL), washed with water (200 mL) and brine (150 mL), dried (Na2SO4) and concentrated in vacuo to yield a yellow solid (54.0 g, 49.8 mmol, 88%). The product was purified by recrystallization from heptanes/ EtOAc/ EtOH (1:0.5:1) to afford a white solid (45.0 g, 112 mmol, 73%) TLC: Rf = 0.6 (heptanes/ethyl acetate = 4/1); HPLC-MS: 98% β-isomer, 1% α-isomer, 1.3% ß-estradiol; DSC: Mp. 122.8°C, purity 99.8%; 1H-NMR (200 MHz, CDCl3) δ 7.44 (m, 5H), 7.27 (d, 1H, J = 8.4 Hz), 6.86 (dd, 1H, J1 = 2.6 Hz, J2 = 8.4 Hz), 6.80 (d, 1H, J = 2.6 Hz), 6.17 (d, 1H, J = 5.8 Hz), 5.78 (dd, 1H, J1 = 1.4 Hz, J2 = 3.2 Hz), 5.45 (m, 1H), 5.11 (s, 2H), 2.96 (m, 2H), 2.40 – 1.54 (m, 10H), 2.18 (s, 3H), 0.93 (s, 3H) ppm.
Example 9 17-Acetyl-3-Benzyl estetrol (compound 3; A = benzyl, C = acetyl)
[0090]OsO4 on PVP (9 g, ~5% w/w OsO4 on PVP, prepared according to Cainelli et al. Synthesis, 45 – 47 (1989) was added to a solution of 17-Acetyloxy-3-benzyloxy-estra-1,3,5 (10),15-tetraene (compound 4; A = benzyl, C = acetyl; 45 g, 112 mmol) in THF (450 mL) and the mixture was heated to 50°C. Trimethylamine-N-oxide dihydrate (24.9 g, 224 mmol) was added portion-wise over 2 h. After stirring for 36 h at 50°C (TLC showed the reaction to be complete) the reaction mixture was cooled to room temperature. The solids were filtered off, washed with THF (100 mL) and the filtrate was concentrated. The residue was taken up in EtOAc (250 mL) and water (250 mL) was added. The aqueous layer was acidified with 1 N HCl (ca. 10 mL). The layers were separated and the aqueous layer was extracted with EtOAc (150 mL). The organic layers were combined, dried (Na2SO4) and concentrated in vacuo. The residue was triturated with heptanes/EtOAc (1:1, 100 mL), stirred for 2 h and the resulting white precipitate was filtered off to give the product as a white solid (41 g, 94 mmol, 84%). The product was purified by recrystallization from heptanes/ ethyl acetate/ EtOH (2:1:1) three times to afford a white solid (21 g, 48.2 mmol, 43%). HPLC-MS: 99.5% βαα-isomer; DSC: Mp. 159.3°C, purity 98.7%; 1H-NMR (200 MHz, CDCl3) δ 7.49 (m, 5H), 7.27 (d, 1H, J = 8.4 Hz), 6.84 (dd, 1H, J1 = 2.6 Hz, J2 = 8.4 Hz), 6.81 (d, 1H, J = 2.4 Hz), 5.11 (s, 2H), 4.45 (d, 1H, J = 4.4), 4.11 (m, 3H), 3.12 (m, 1H) 2.95 (m, 2H), 2.46 -1.64 (m, 10H), 2.24 (s, 3H), 0.93 (s, 3H) ppm.
Example 10 17-Acetyl estetrol (compound 2; C = acetyl)
[0091]To a solution of 17-acetyl-3-benzyl estetrol (compound 3; A = benzyl, C = acetyl; 21 g, 48.2 mmol) in MeOH (600 mL, HPLC-grade) was added a preformed suspension of 10% Palladium on activated carbon (2 g) in methanol (50 mL). The mixture was placed under an atmosphere of H2 at 1 atm and stirred for 24 h (TLC showed the reaction to be completed) at room temperature. It was filtered over Celite® and the filter cake was washed with MeOH (200 mL). The filtrate was concentrated in vacuo to give 17-acetyl estetrol as a white solid (15 g, 43.4 mmol, 90%). TLC: Rf = 0.2 (heptanes/ethyl acetate = 1/1); HPLC-MS: 99.2%, DSC: Mp. 212.2°C, purity 98.9%; 1H-NMR (200 MHz, CD3OD) δ 7.14 (d, 1H, J = 8.0 Hz), 6.60 (dd, 1H, J1 = 2.6 Hz, J2 = 8.8 Hz), 6.56 (d, 1H, J = 2.4 Hz), 4.81 (dd, 1H, J1 = 3.4 Hz, J2 = 6.4 Hz), 4.07 (m, 3H), 3.12 (m, 1H), 2.85 (m, 2H), 2.37 – 1.37 (m, 10H), 2.18 (s, 3H), 0.91 (s, 3H) ppm.
Example 11 Estetrol
[0092]17-Acetyl-estetrol (compound 2; C = acetyl; 15 g, 43.4 mmol) and K2CO3 (6 g, 43.4 mmol) were suspended in MeOH (500 mL, HPLC-grade) and stirred for 4 h at room temperature (TLC showed the reaction to be complete). The solvents were evaporated in vacuo. Water (200 mL) and CHCl3 (70 mL) were added and the mixture was stirred and neutralized with 0.1 N HCl (50 mL). The product was collected by filtration, washed with water (100 mL) and CHCl3 (100 mL) to give estetrol as a white solid (12.2 g, 40.1 mmol, 92.5%, overall yield from estrone 10.8%) after drying at 40°C in an air-ventilated oven. TLC: Rf = 0.05 (heptanes/ethyl acetate = 1/1); HPLC-MS: 99.1%, DSC: Mp. 243.7°C, purity 99.5%; 1H-NMR (200 MHz, CD3OD) δ 7.14 (d, 1H, J = 8.6 Hz), 6.61 (dd, 1H, J1 = 2.6 Hz, J2 = 8.4 Hz), 6.56 (d, 1H, J = 2.4 Hz), 4.83 (m, 1H), 3.93 (m, 3H), 3.50 (d, 1H, J = 5.2), 3.38 (m, 2H), 2.84 (m, 2H), 2.32 (m, 3H), 1.97 (m, 1H), 1.68 – 1.24 (m, 5H), 0.86 (s, 3H) ppm.
SYN
https://www.tandfonline.com/doi/abs/10.1080/13697130802054078?journalCode=icmt20
Estetrol (E4), or oestetrol, is a weak estrogen steroid hormone, which is found in detectable levels only during pregnancy in humans.[1][2] It is produced exclusively by the fetal liver.[1] Estetrol is closely related to estriol (E3), which is also a weak estrogen that is found in high quantities only during pregnancy.[1][2] Along with estradiol (E2), estrone (E1), and E3, estetrol (E4) is a major estrogen in the body, although only during pregnancy.[1]
In addition to its role as a natural hormone, estetrol is under clinical development for use as a medication, for instance in hormonal contraception (in combination with drospirenone) and as menopausal hormone therapy; for information on estetrol as a medication, see the estetrol (medication) article.
Biological function
Estetrol is an estrogen and has estrogenic effects in various tissues.[1] Estetrol interacts with nuclear Estrogen Receptor (ERα) in a manner identical to that of the other estrogens and distinct from that observed with Selective Estrogen Receptor Modulators (SERMs).[3][4] So far the physiological function of estetrol is unknown. The possible use of estetrol as a marker for fetal well-being has been studied quite extensively. However, due to the large intra- and inter-individual variation of maternal estetrol plasma levels during pregnancy this appeared not to be feasible.[5][6][7][8][9]
Biological activity
Estetrol is an agonist of the estrogen receptors (ERs), and hence is an estrogen.[10][11] It has moderate affinity for ERα and ERβ, with Ki values of 4.9 nM and 19 nM, respectively.[10][12] As such, estetrol has 4- to 5-fold preference for the ERα over the ERβ.[10][12] The estrogen has low affinity for the ERs relative to estradiol, and both estetrol and the related estrogen estriol require substantially higher concentrations than estradiol to produce similar effects to estradiol.[10] The affinity of estetrol for the ERs is about 0.3% (rat) to 6.25% (human) of that of estradiol, and its in vivo potency in animals is about 2 to 3% of that of estradiol.[10] Estetrol shows high selectivity for the ERs.[10][12]
Biochemistry
Biosynthesis
Estetrol is synthesized during pregnancy only in the fetal liver from estradiol (E2) and estriol (E3) by the two enzymes 15α- and 16α-hydroxylase.[13][14][15] Alternatively, estetrol is synthesized with 15α-hydroxylation of 16α-hydroxy-DHEA sulfate as an intermediate step.[16] It appears in maternal urine at around week 9 of pregnancy.[2] After birth the neonatal liver rapidly loses its capacity to synthesize estetrol because these two enzymes are no longer expressed.
Estetrol reaches the maternal circulation through the placenta and was already detected at nine weeks of pregnancy in maternal urine.[17][18] During the second trimester of pregnancy high levels were found in maternal plasma, with steadily rising concentrations of unconjugated estetrol to about 1 ng/mL (>3 nM) towards the end of pregnancy.[1]
Distribution
In terms of plasma protein binding, estetrol is moderately bound to albumin, and is not bound to sex hormone-binding globulin (SHBG).[19][20]
Metabolism
Estetrol undergoes no phase I metabolism by CYP P450 enzymes.[10] It is conjugated via glucuronidation and to a lesser extent sulfation and then excreted.[10][21]
Excretion
Estetrol is excreted mostly or completely in urine.[21][10]
Chemistry
See also: List of estrogens
vteStructures of major endogenous estrogens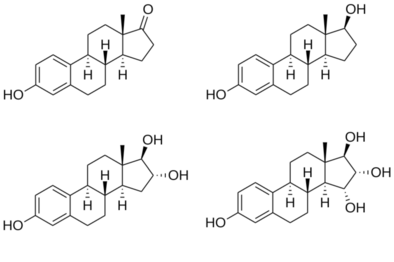 Estrone (E1)Estradiol (E2)Estriol (E3)Estetrol (E4) Estrone (E1)Estradiol (E2)Estriol (E3)Estetrol (E4) |
Estetrol, also known as 15α-hydroxyestriol or as estra-1,3,5(10)-triene-3,15α,16α,17β-tetrol, is a naturally occurring estrane steroid and derivative of estrin (estratriene).[10][11] It has four hydroxyl groups, which explains the abbreviation E4.[10][11]
Synthesis
Chemical syntheses of estetrol have been published.[22]
History
Estetrol was discovered in 1965 by Egon Diczfalusy and coworkers at the Karolinska Institute in Stockholm, Sweden, via isolation from the urine of pregnant women.[10][23]
References
- ^ Jump up to:a b c d e f Holinka CF, Diczfalusy E, Coelingh Bennink HJ (May 2008). “Estetrol: a unique steroid in human pregnancy”. J. Steroid Biochem. Mol. Biol. 110 (1–2): 138–43. doi:10.1016/j.jsbmb.2008.03.027. PMID 18462934.
- ^ Jump up to:a b c Reproductive Endocrinology: Physiology, Pathophysiology, and Clinical Management, 3rd ed., SSC Yen and RB Jaffe (eds.), pp. 936–981, Copyright Elsevier/Saunders 1991
- ^ Abot, Anne; Fontaine, Coralie; Buscato, Mélissa; Solinhac, Romain; Flouriot, Gilles; Fabre, Aurélie; Drougard, Anne; Rajan, Shyamala; Laine, Muriel; Milon, Alain; Muller, Isabelle (2014). “The uterine and vascular actions of estetrol delineate a distinctive profile of estrogen receptor α modulation, uncoupling nuclear and membrane activation”. EMBO Molecular Medicine. 6 (10): 1328–1346. doi:10.15252/emmm.201404112. ISSN 1757-4676. PMC 4287935. PMID 25214462.
- ^ Foidart, JM; et al. (2019). “30th Annual Meeting of The North America Menopause Society September 25 – 28, 2019, Chicago, IL”. Menopause. 26 (12): 1445–1481. doi:10.1097/GME.0000000000001456. ISSN 1530-0374.
- ^ J. Heikkilä, T. Luukkainen, Urinary excretion of estriol and 15a-hydroxyestriol in complicated pregnancies, Am. J. Obstet. Gynecol. 110 (1971) 509-521.
- ^ D. Tulchinsky, F.D. Frigoletto, K.J. Ryan, J. Fishman, Plasma estetrol as an index of fetal well-being, J. Clin. Endocrinol. Metab. 40 (1975) 560-567
- ^ A.D. Notation, G.E. Tagatz, Unconjugated estriol and 15a-hydroxyestriol in complicated pregnancies, Am. J. Obstet. Gynecol. 128 (1977) 747-756.
- ^ N. Kundu, M. Grant, Radioimmunoassay of 15a-hydroxyestriol (estetrol) in pregnancy serum, Steroids 27 (1976) 785-796.
- ^ N. Kundu, M. Wachs, G.B. Iverson, L.P. Petersen, Comparison of serum unconjugated estriol and estetrol in normal and complicated pregnancies, Obstet. Gynecol. 58 (1981) 276-281.
- ^ Jump up to:a b c d e f g h i j k l Coelingh Bennink HJ, Holinka CF, Diczfalusy E (2008). “Estetrol review: profile and potential clinical applications”. Climacteric. 11 Suppl 1: 47–58. doi:10.1080/13697130802073425. PMID 18464023.
- ^ Jump up to:a b c Visser M, Coelingh Bennink HJ (March 2009). “Clinical applications for estetrol” (PDF). J. Steroid Biochem. Mol. Biol. 114(1–2): 85–9. doi:10.1016/j.jsbmb.2008.12.013. PMID 19167495.
- ^ Jump up to:a b c Visser M, Foidart JM, Coelingh Bennink HJ (2008). “In vitro effects of estetrol on receptor binding, drug targets and human liver cell metabolism”. Climacteric. 11 Suppl 1: 64–8. doi:10.1080/13697130802050340. PMID 18464025.
- ^ J. Schwers, G. Eriksson, N. Wiqvist, E. Diczfalusy, 15a-hydroxylation: A new pathway of estrogen metabolism in the human fetus and newborn, Biochim. Biophys. Acta. 100 (1965) 313-316
- ^ J. Schwers, M. Govaerts-Videtsky, N. Wiqvist, E. Diczfalusy, Metabolism of oestrone sulphate by the previable human foetus, Acta Endocrinol. 50 (1965) 597-610.
- ^ S. Mancuso, G. Benagiano, S. Dell’Acqua, M. Shapiro, N. Wiqvist, E. Diczfalusy, Studies on the metabolism of C-19 steroids in the human foeto-placental unit, Acta Endocrinol. 57 (1968) 208-227.
- ^ Jerome Frank Strauss; Robert L. Barbieri (2009). Yen and Jaffe’s Reproductive Endocrinology: Physiology, Pathophysiology, and Clinical Management. Elsevier Health Sciences. pp. 262–. ISBN 1-4160-4907-X.
- ^ J. Heikkilä, H. Adlercreutz, A method for the determination of urinary 15α-hydroxyestriol and estriol, J. Steroid Biochem. 1 (1970) 243-253
- ^ J. Heikkilä, Excretion of 15α-hydroxyestriol and estriol in maternal urine during normal pregnancy, J. Steroid Biochem. 2 (1971) 83-93.
- ^ Visser M, Holinka CF, Coelingh Bennink HJ (2008). “First human exposure to exogenous single-dose oral estetrol in early postmenopausal women”. Climacteric. 11 Suppl 1: 31–40. doi:10.1080/13697130802056511. PMID 18464021.
- ^ Hammond GL, Hogeveen KN, Visser M, Coelingh Bennink HJ (2008). “Estetrol does not bind sex hormone binding globulin or increase its production by human HepG2 cells”. Climacteric. 11 Suppl 1: 41–6. doi:10.1080/13697130701851814. PMID 18464022.
- ^ Jump up to:a b Mawet M, Maillard C, Klipping C, Zimmerman Y, Foidart JM, Coelingh Bennink HJ (2015). “Unique effects on hepatic function, lipid metabolism, bone and growth endocrine parameters of estetrol in combined oral contraceptives”. Eur J Contracept Reprod Health Care. 20 (6): 463–75. doi:10.3109/13625187.2015.1068934. PMC 4699469. PMID 26212489.
- ^ Warmerdam EG, Visser M, Coelingh Bennink HJ, Groen M (2008). “A new route of synthesis of estetrol”. Climacteric. 11 Suppl 1: 59–63. doi:10.1080/13697130802054078. PMID 18464024.
- ^ Hagen AA, Barr M, Diczfalusy E (June 1965). “Metabolism of 17-beta-oestradiol-4-14-C in early infancy”. Acta Endocrinol. 49: 207–20. doi:10.1530/acta.0.0490207. PMID 14303250.
| Names | |
|---|---|
| Preferred IUPAC name(1R,2R,3R,3aS,3bR,9bS,11aS)-11a-Methyl-2,3,3a,3b,4,5,9b,10,11,11a-decahydro-1H-cyclopenta[a]phenanthrene-1,2,3,7-tetrol | |
| Other namesOestetrol; E4; 15α-Hydroxyestriol; Estra-1,3,5(10)-triene-3,15α,16α,17β-tetrol | |
| Identifiers | |
| CAS Number | 15183-37-6 |
| 3D model (JSmol) | Interactive image |
| ChEBI | CHEBI:142773 |
| ECHA InfoCard | 100.276.707 |
| KEGG | D11513 |
| PubChem CID | 27125 |
| UNII | ENB39R14VF |
| CompTox Dashboard (EPA) | DTXSID50164888 |
| showSMILES | |
| Properties | |
| Chemical formula | C18H24O4 |
| Molar mass | 304.386 g/mol |
| Solubility in water | 1.38 mg/mL |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references |
//////////estetrol, Nextstellis, fda 2021, approvals 2021

NEW DRUG APPROVALS
one time
$10.00

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
