TROFINETIDE

Trofinetide
- Molecular FormulaC13H21N3O6
- Average mass315.322 Da
Tofinetide , NNZ-256610076853400-76-7[RN]
glycyl-2-methyl-L-prolyl-L-glutamic acid
H-Gly-PMe-Glu-OHL-Glutamic acid, glycyl-2-methyl-L-prolyl-UNII-Z2ME8F52QLZ2ME8F52QLтрофинетид [Russian] [INN]تروفينيتيد [Arabic] [INN]曲非奈肽 [Chinese] [INN]
| IUPAC Condensed | H-Gly-aMePro-Glu-OH |
|---|---|
| Sequence | GXE |
| HELM | PEPTIDE1{G.[*C(=O)[C@@]1(CCCN1*)C |$_R2;;;;;;;;_R1;$|].E}$$$$ |
| IUPAC | glycyl-alpha-methyl-L-prolyl-L-glutamic acid |
An (1-3) IGF-1 analog with neuroprotective activity.
OPTICAL ROT; -52.4 ° Conc: 0.19 g/100mL; water ; 589.3 nm; Temp: 20 °C; Len: 1.0 dm…Tetrahedron 2005, V61(42), P10018-10035
EU Customs Code CN, 29339980
Harmonized Tariff Code, 293399
- L-Glutamic acid, glycyl-2-methyl-L-prolyl-
- glycyl-2-methyl-L-prolyl-L-glutamic acid
- Glycyl-L-2-methylprolyl-L-glutamic acid
FDA APPROVED 2023/3/10, Daybue
853400-76-7 CAS
トロフィネチド;
Trofinetide (NNZ-2566) is a drug developed by Neuren Pharmaceuticals that acts as an analogue of the neuropeptide (1-3) IGF-1, which is a simple tripeptide with sequence Gly–Pro–Glu formed by enzymatic cleavage of the growth factor IGF-1 within the brain. Trofinetide has anti-inflammatory properties and was originally developed as a potential treatment for stroke,[1][2] but has subsequently been developed for other applications and is now in Phase II clinical trials against Fragile X syndrome and Rett syndrome.[3][4][5]
Trofinetide (NNZ-2566), a neuroprotective analogue of glypromate, is a novel molecule that has a profile suitable for both intravenous infusion and chronic oral delivery. It is currently in development to treat traumatic brain injury.
In February 2021, Neuren is developing trofinetide (NNZ-2566, phase 2 clinical ), a small-molecule analog of the naturally occurring neuroprotectant and N-terminus IGF-1 tripeptide Glypromate (glycine-proline-glutamate), for intravenous infusion treatment of various neurological conditions, including moderate to severe traumatic brain injury (TBI), stroke, chronic neurodegenerative disorders and peripheral neuropathies. At the same time, Neuren is also investigating an oral formulation of trofinetide (phase 3 clinical) for similar neurological indications, including mild TBI.
Autism Spectrum Disorders and neurodevelopment disorders (NDDs) are becoming increasingly diagnosed. According to the fourth edition of the American Psychiatric Association’s (APA) Diagnostic and Statistical Manual oƒ Mental Disorders (DSM-4), Autism spectrum disorders (ASD) are a collection of linked developmental disorders, characterized by abnormalities in social interaction and communication, restricted interests and repetitive behaviours. Current classification of ASD according to the DSM-4 recognises five distinct forms: classical autism or Autistic Disorder, Asperger syndrome, Rett syndrome, childhood disintegrative disorder and pervasive developmental disorder not otherwise specified (PDD-NOS). A sixth syndrome, pathological demand avoidance (PDA), is a further specific pervasive developmental disorder.
More recently, the fifth edition of the American Psychiatric Association’s (APA) Diagnostic and Statistical Manual oƒ Mental Disorders (DSM-5) recognizes recognises Asperger syndrome, childhood disintegrative disorder, and pervasive developmental disorder not otherwise specified (PDD-NOS) as ASDs.
This invention applies to treatment of disorders, regardless of their classification as either DSM-4 or DSM-5.
Neurodevelopment Disorders (NDDs) include Fragile X Syndrome (FXS), Angelman Syndrome, Tuberous Sclerosis Complex, Phelan McDermid Syndrome, Rett Syndrome, CDKL5 mutations (which also are associated with Rett Syndrome and X-Linked Infantile Spasm Disorder) and others. Many but not all NDDs are caused by genetic mutations and, as such, are sometimes referred to as monogenic disorders. Some patients with NDDs exhibit behaviors and symptoms of autism.
As an example of a NDD, Fragile X Syndrome is an X-linked genetic disorder in which affected individuals are intellectually handicapped to varying degrees and display a variety of associated psychiatric symptoms. Clinically, Fragile X Syndrome is characterized by intellectual handicap, hyperactivity and attentional problems, autism spectrum symptoms, emotional lability and epilepsy (Hagerman, 1997a). The epilepsy seen in Fragile X Syndrome is most commonly present in childhood, but then gradually remits towards adulthood. Hyperactivity is present in approximately 80 percent of affected males (Hagerman, 1997b). Physical features such as prominent ears and jaw and hyper-extensibility of joints are frequently present but are not diagnostic. Intellectual handicap is the most common feature defining the phenotype. Generally, males are more severely affected than females. Early impressions that females are unaffected have been replaced by an understanding of the presence of specific learning difficulties and other neuropsychiatric features in females. The learning disability present in males becomes more defined with age, although this longitudinal effect is more likely a reflection of a flattening of developmental trajectories rather than an explicit neurodegenerative process.
The compromise of brain function seen in Fragile X Syndrome is paralleled by changes in brain structure in humans. MRI scanning studies reveal that Fragile X Syndrome is associated with larger brain volumes than would be expected in matched controls and that this change correlates with trinucleotide expansion in the FMRP promoter region (Jakala et al, 1997). At the microscopic level, humans with Fragile X Syndrome show abnormalities of neuronal dendritic structure, in particular, an abnormally high number of immature dendritic spines (Irwin et al, , 2000).
Currently available treatments for NDDs are symptomatic – focusing on the management of symptoms – and supportive, requiring a multidisciplinary approach. Educational and social skills training and therapies are implemented early to address core issues of learning delay and social impairments. Special academic, social, vocational, and support services are often required. Medication, psychotherapy or behavioral therapy may be used for management of co-occurring anxiety, ADHD, depression, maladaptive behaviors (such as aggression) and sleep issues, Antiepileptic drugs may be used to control seizures.
Patent
WO 2014085480,
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2014085480


EP 0 366 638 discloses GPE (a tri-peptide consisting of the amino acids Gly-Pro-Glu) and its di-peptide derivatives Gly-Pro and Pro-Glu. EP 0 366 638 discloses that GPE is effective as a neuromodulator and is able to affect the electrical properties of neurons.
WO95/172904 discloses that GPE has neuroprotective properties and that administration of GPE can reduce damage to the central nervous system (CNS) by the prevention or inhibition of neuronal and glial cell death.
WO 98/14202 discloses that administration of GPE can increase the effective amount of choline acetyltransferase (ChAT), glutamic acid decarboxylase (GAD), and nitric oxide synthase (NOS) in the central nervous system (CNS).
WO99/65509 discloses that increasing the effective amount of GPE in the CNS, such as by administration of GPE, can increase the effective amount of tyrosine hydroxylase (TH) in the CNS to increase TH-mediated dopamine production in the treatment of diseases such as Parkinson’s disease.
WO02/16408 discloses certain GPE analogs having amino acid substitutions and certain other modification that are capable of inducing a physiological effect equivalent to GPE within a patient. The applications of the GPE analogs include the treatment of acute brain injury and neurodegenerative diseases, including injury or disease in the CNS.
EXAMPLES
The following examples are intended to illustrate embodiments of this invention, and are not intended to limit the scope to these specific examples. Persons of ordinary skill in the art can apply the disclosures and teachings presented herein to develop other embodiments without undue experimentation and with a likelihood of success. All such embodiments are considered part of this invention.
Example 1: Synthesis of N,N-Dimethylglycyl-L-prolyl)-L-glutamic acid
The following non-limiting example illustrates the synthesis of a compound of the invention, N,N-Dimethylglycyl-L-prolyl-L-glutamic acid
All starting materials and other reagents were purchased from Aldrich; BOC=tert-butoxycarbonyl; Bn=benzyl.
BOC-L-proline-(P-benzyl)-L-glutamic acid benzyl ester
To a solution of BOC-proline [Anderson GW and McGregor AC: J. Amer. Chem. Soc: 79, 6810, 1994] (10 mmol) in dichloromethane (50 mi), cooled to 0°C, was added triethylamine (1 .39 ml, 10 mmol) and ethyl chloroformate (0.96 ml, 10 mmol). The resultant mixture was stirred at 0 °C for 30 minutes. A solution of dibenzyl-L-glutamate (10 mmol) was then added and the mixture stirred at 0° C for 2 hours then warmed to room temperature and stirred overnight. The reaction mixture was washed with aqueous sodium bicarbonate and citric acid (2 mol 1-1) then dried (MgSO4) and concentrated at reduced pressure to give BOC-L-proline-L-glutamic acid dibenzyl ester (5.0 g, 95%).
L-proline-L-glutamic acid dibenzyl ester
A solution of BOC-L-glutamyl-L-proline dibenzyl ester (3.4 g, 10 mmol), cooled to 0 °C, was treated with trifluoroacetic acid (25 ml) for 2 h. at room temperature. After removal of the volatiles at reduced pressure the residue was triturated with ether to give L-proline-L-glutamic acid dibenzyl ester.
N,N-Dimethylglycyl-L-prolyl-L-glutamic acid
A solution of dicyclohexylcarbodiimide (10.3 mmol) in dichloromethane (10 ml) was added to a stirred and cooled (0 °C) solution of L-proline-L-glutamic acid dibenzyl ester (10 mmol), N,N-dimethylglycine (10 mmol) and triethylamine ( 10.3 mmol) in dichloromethane (30 ml). The mixture was stirred at 0°C overnight and then at room temperature for 3 h. After filtration, the filtrate was evaporated at reduced pressure. The resulting crude dibenzyl ester was dissolved in a mixture of ethyl acetate (30 ml) and methanol (30 ml) containing 10% palladium on charcoal (0.5 g) then hydrogenated at room temperature and pressure until the uptake of hydrogen ceased. The filtered solution was evaporated and the residue recrystallised from ethyl acetate to yield the tripeptide derivative.
It can be appreciated that following the method of the Examples, and using alternative amino acids or their amides or esters, will yield other compounds of Formula 1.
Eample 2: Synthesis of Glycyl-L-2-Methyl-L-Prolyl-L-Glutamate
L-2-Methylproline and L-glutamic acid dibenzyl ester p-toluenesulphonate were purchased from Bachem, N-benzyloxycarbonyl-glycine from Acros Organics and bis(2-oxo-3-oxazolidinyl)phosphinic chloride (BoPCl, 97%) from Aldrich Chem. Co.
Methyl L-2-methylprolinate hydrochloride 2
Thionyl chloride (5.84 cm3, 80.1 mmol) was cautiously added dropwise to a stirred solution of (L)-2-methylproline 1 (0.43 g, 3.33 mmol) in anhydrous methanol (30 cm3) at -5 °C under an atmosphere of nitrogen. The reaction mixture was heated under reflux for 24 h, and the resultant pale yellow-coloured solution was. concentrated to dryness in vacuo. The residue was dissolved in a 1 : 1 mixture of methanol and toluene (30 cm3) then concentrated to dryness to remove residual thionyl chloride. This procedure was repeated twice more, yielding hydrochloride 2 (0.62 g, 104%) as an hygroscopic, spectroscopically pure, off-white solid: mp 127- 131 °C; [α]D -59.8 (c 0.24 in CH2Cl2); vmax (film)/cm-1 3579, 3398 br, 2885, 2717, 2681 , 2623, 2507, 1743, 1584, 1447, 1432, 1374, 1317, 1294, 1237, 1212, 1172, 1123, 981 , 894, 861 and 764; δH (300 MHz; CDCl3; Me4Si) 1.88 (3H, s, Proα-CH3), 1 .70-2.30 (3H, br m, Proβ-HAΗΒ and Proγ-H2), 2.30-2.60 (1H, br m, Proβ-HAΗΒ), 3.40-3.84 (2H, br m, Proδ-H2), 3.87 (3H, s, CO2CH3), 9.43 (1H, br s, NH) and 10.49 ( 1H, br s, HCl); δC (75 MHz; CDCl3) 21.1 (CH3, Proα-CH3), 22.4 (CH2, Proγ-C), 35.6 (CH2, Proβ-C), 45.2 (CH2, Proδ-C), 53.7 (CH3, CO2CH3), 68.4 (quat., Proα-C) and 170.7 (quat, CO); m/z (FAB+) 323.1745 [M2.H35Cl.H+: (C7H13NO2)2. H35Cl.H requires 323.1738] and 325.1718 [M2.H37Cl.H+: (C7H13NOz)2. H37Cl.H requires 325.1708],
N-Benxyloxycarbonyl-glycyl-L-2-methylproline 5
Anhydrous triethylamine (0.45 cm3, 3.23 mmol) was added dropwise to a mixture of methyl L-2-methylprolinate hydrochloride 2 (0.42 g, 2.34 mmol) and N-benzyloxycarbonyl-glycine (98.5%) 3 (0.52 g, 2.45 mmol) in methylene chloride (16 cm3), at 0 °C, under an atmosphere of nitrogen. The resultant solution was stirred for 20 min and a solution of 1 ,3-dicyclohexylcarbodiimide (0.56 g, 2.71 mmol) in methylene chloride (8 cm3) at 0 °C was added dropwise and the reaction mixture was warmed to room temperature and stirred for a further 20 h. The resultant white mixture was filtered through a Celite™ pad to partially remove 1 ,3-dicyclohexylurea, and the pad was washed with methylene chloride (50 cm3). The filtrate was washed successively with 10% aqueous hydrochloric acid (50 cm3) and saturated aqueous sodium hydrogen carbonate (50 cm3), dried (MgSO4), filtered, and concentrated to dryness in vacuo. Further purification of the residue by flash column chromatography (35 g SiO2; 30-70% ethyl acetate – hexane; gradient elution) afforded tentatively methyl N-benzyloxycarbonyl-glycyl-L-2-methylprolinate 4 (0.56 g), containing 1 ,3-dicyclohexylurea, as a white semi-solid: Rf 0.65 (EtOAc); m/z (ΕI+) 334.1534 (M+. C17H22N2O5 requires 334.1529) and 224 ( 1 ,3-dicyclohexylurea).
To a solution of impure prolinate 4 (0.56 g, ca. 1.67 mmol) in 1,4-dioxane (33 cm3) was added dropwise 1 M aqueous sodium hydroxide (10 cm3, 10 mmol) and the mixture was stirred for 19 h at room temperature. Methylene chloride ( 100 cm3) was then added and the organic layer extracted with saturated aqueous sodium hydrogen carbonate (2 x 100 cm3). The combined aqueous layers were carefully acidified with hydrochloric acid (32%), extracted with methylene chloride (2 x 100 cm3), and the combined organic layers dried (MgSO4), filtered, and
concentrated to dryness in vacuo. Purification of the ensuing residue (0.47 g) by flash column chromatography ( 17 g SiO2; 50% ethyl acetate – hexane to 30% methanol – dichloromethane; gradient elution) gave N-protected dipeptide 5 (0.45 g, 60%) as a white foam in two steps from hydrochloride 2. Dipeptide 5 was shown to be exclusively the frafw-orientated conformer by NMR analysis: Rf 0.50 (20% MeOH – CH2Cl2); [α]D -62.3 (c 0.20 in CH2Cl2); vmax (film)/cm-1 3583, 3324 br, 2980, 2942, 1722, 1649, 1529, 1454, 1432, 1373, 1337, 1251 , 1219, 1179, 1053, 1027, 965, 912, 735 and 698; δH (300 MHz; CDCl3; Me4Si) 1.59 (3H, s, Proα-CH3), 1 .89 (1H, 6 lines, J 18.8, 6.2 and 6.2, Proβ-HAHB), 2.01 (2H, dtt, J 18.7, 6.2 and 6.2, Proγ-H2), 2.25-2.40 (1H, m, Proβ-HAΗΒ), 3.54 (2H, t, J 6.6, Proδ-H2), 3.89 (1H, dd, J 17.1 and 3.9, Glyα-HAHB), 4.04 (1H, dd, J 17.2 and 5.3, Glyα-HAΗΒ), 5.11 (2H, s, OCH2Ph), 5.84 (I H, br t, J 4.2, N-H), 7.22-7.43 (5H, m, Ph) and 7.89 (1 H, br s, -COOH); δC (75 MHz; CDCl3) 21.3 (CH3, Proα-CH3), 23.8 (CH2, Proγ-C), 38.2 (CH2, Proβ-C), 43.6 (CH2, Glyα-C), 47.2 (CH2, Proδ-C), 66.7 (quat, Proα-C), 66.8 (CH2, OCH2Ph), 127.9 (CH, Ph), 127.9 (CH, Ph), 128.4, (CH, Ph), 136.4 (quat., Ph), 156.4 (quat., NCO2), 167.5 (quat., Gly-CON) and 176.7 (quat., CO); m/z (EI+) 320.1368 (M+. C16Η20Ν2Ο5 requires 320.1372).
Dibenzyl N-benzyloxycarbonyl-glycyl-L-2-methylprolyl-L-glutamate 7
Triethylamine (0.50 cm3, 3.59 mmol) was added dropwise to a solution of dipeptide 5 (0.36 g, 1.12 mmol) and L-glutamic acid dibenzyl ester /Moluenesulphonate 6 (0.73 g, 1.46 mmol) in methylene chloride (60 cm3) under nitrogen at room temperature, and the reaction mixture stirred for 10 min. Bis(2-oxo-3-oxazoIidinyl)phosphinic chloride (BoPCl, 97%) (0.37 g, 1.41 mmol) was added and the colourless solution stirred for 17 h. The methylene chloride solution was washed successively with 10% aqueous hydrochloric acid (50 cm3) and saturated aqueous sodium hydrogen carbonate (50 cm3), dried (MgSO4), filtered, and evaporated to dryness in vacuo. Purification of the resultant residue by repeated (2x) flash column chromatography (24 g SiO2; 30-70% ethyl acetate – hexane; gradient elution) yielded ƒully protected tripeptide 7 (0.63 g, 89%) as a colourless oil. Tripeptide 7 was shown to be exclusively the trans-orientated conformer by NMR analysis: Rf 0.55 (EtOAc); [α]D -41.9 (c 0.29 in CH2Cl2); vmax (film)/cm-1 3583, 3353 br, 2950, 1734, 1660, 1521, 1499, 1454, 1429, 1257, 1214, 1188, 1166, 1051, 911, 737 and 697; δH (400 MHz; CDCl3; Me4Si) 1.64 (3H, s, Proot-CH3), 1.72 (1H, dt, J 12.8, 7.6 and 7.6, Proβ-HAHB), 1.92 (2H, 5 lines, J 6.7, Proγ-H2), 2.04 (1H, 6 lines, J 7.3 Gluβ-HAHB), 2.17-2.27 (1H, m, Gluβ-HAΗΒ), 2.35-2.51 (3H, m, Proβ-HAΗΒ and Gluγ-H2), 3.37-3.57 (2H, m, Proδ-H2), 3.90 (1 H, dd, J 17.0 and 3.6, Glyα-HAHB), 4.00 (1H, dd, J 17.1 and 5.1, Glyα-HAΗΒ), 4.56 (1H, td, J 7.7 and 4.9, Glyα-H), 5.05-5.20 (6H, m, 3 x OCH2Ph), 5.66-5.72 (1H, br m, Gly-NH), 7.26-7.37 (15H, m, 3 x Ph) and 7.44 (1H, d, J 7.2, Glu-NH); δC (100 MHz; CDCl3) 21.9 (CH3, Proα-CH3), 23.4 (CH2, Proγ-C), 26.6 (CH2, Gluβ-C), 30.1 (CH2, Gluγ-C), 38.3 (CH2, Proβ-C),
43.9 (CH2, Glyα-C), 47.6 (CH2, Proδ-C), 52.2 (CH, Glua-C), 66.4 (CH2, OCH2Ph), 66.8 (CH2, OCH2Ph), 67.1 (CH2, OCH2Ph), 68.2 (quat, Proα-C), 127.9 (CH, Ph), 128.0 (CH, Ph), 128.1, (CH, Ph), 128.2, (CH, Ph), 128.2, (CH, Ph), 128.3, (CH, Ph), 128.4, (CH, Ph), 128.5, (CH, Ph), 128.5, (CH, Ph), 135.2 (quat., Ph), 135.7 (quat., Ph), 136.4 (quat, Ph), 156.1 (quat, NCO2), 167.3 (quat., Gly-CO), 171.4 (quat., CO), 172.9 (quat., CO) and 173.4 (quat., CO); m/z (FAB+) 630.2809 (MH+. C35H40N3O8 requires 630.2815).
Glycyl-L-2-methylprolyl-L-glutamic acid (G-2-MePE)
A mixture of the protected tripeptide 7 (0.63 g, 1.00 mmol) and 10 wt % palladium on activated carbon (0.32 g, 0.30 mmol) in 91 :9 methanol – water (22 cm3) was stirred under an atmosphere of hydrogen at room temperature, protected from light, for 23 h. The reaction mixture was filtered through a Celite™ pad and the pad washed with 75 :25 methanol – water (200 cm3). The filtrate was concentrated to dryness under reduced pressure and the residue triturated with anhydrous diethyl ether to afford a 38: 1 mixture of G-2-MePE and tentatively methylamine 8 (0.27 g, 86%) as an extremely hygroscopic white solid. Analytical reverse-phase HPLC studies on the mixture [Altech Econosphere C 18 Si column, 150 x 4.6 mm, 5 ☐m; 5 min flush with H2O (0.05% TFA) then steady gradient over 25 min to MeCN as eluent at flow rate of 1 ml/min; detection using diode array] indicated it was a 38: 1 mixture of two eluting peaks with retention times of 13.64 and 14.44 min at 207 and 197 nm, respectively. G-2-MePE was shown to be a 73 :27 trans:cis mixture of conformers by 1H NMR analysis (the ratio was estimated from the relative intensities of the double doublet and triplet at δ 4.18 and 3.71 , assigned to the Gluα-H protons of the major and minor conformers, respectively):
mp 144 °Cɸ;
[ α]D -52.4 (c 0.19 in H2O);
δα (300 MHz; D2O; internal MeOH) 1.52 (3H, s, Proα-CH3), 1.81-2.21 (6H, m, Proβ-H2, Proγ-H, and Gluβ-H2), 2.34 (1.46H, t, J 7.2, Gluy-H2), 2.42* (0.54H, t, 77.3, Gluγ-H2), 3.50-3.66 (2H, m, Pro6-H2), 3.71 * (0.27H, t, J 6.2, Gluoc-H), 3.85 (1H, d, J 16.6, Glyα-HAHB), 3.92 (1H, d, J 16.6, Glyα-HAΗΒ) and 4.18 (0.73H, dd, J 8.4 and 4.7, Glua-H);
δC (75 MHz; D2O; internal MeOH) 21.8 (CH3, Proα-CH3), 25.0 (CH2, Proγ-C), 27.8* (CH2: Gluβ-C), 28.8 (CH2, Gluβ-C), 32.9 (CH2, Gluγ-C), 40.8 (CH2, Proβ-C), 42.7 (CH2, Glyα-C), 49.5 (CH2, Proδ-C), 56.0* (CH, Gluα-C), 56.4 (CH, Gluα-C), 69.8 (quat, Proα-C), 166.5 (quat., Gly-CO), 177.3 (quat., Pro-CON), 179.2 (quat., Gluα-CO), 180.2* (quat., Gluγ-CO) and 180.6 (quat., Gluγ-CO);
m/z (FAB+) 3 16.1508 (MH+. C13H22N3O6 requires 316.1509).
PATENT
WO02094856
Example
The following non-limiting example illustrates the synthesis of a compound of the invention, NN-dimethylglycyl-L-prolyl-L-glutamic acid.
All starting materials and other reagents were purchased from Aldrich;
BOC = tert-butoxycarbonyl; Bn = benzyl.
BOC-(γ-benzyl)-L-prolyl-L-glutamic acid benzyl ester
To a solution of BOC-proline [Anderson GW and McGregor AC: J. Amer. Chem.
Soc: 79, 6180, 1957] (10 mmol) in dichloromethane (50 ml), cooled to 0 °C, was added triethylamine (1.39 ml, 10 mmol) and ethyl chloroformate (0.96 ml, 10 mmol). The resultant mixture was stirred at 0 °C for 30 minutes. A solution of dibenzyl L-glutamate (10 mmol) was then added and the mixture stirred at 0 °C for 2 hours then warmed to room temperature and stirred overnight. The reaction mixture was washed with aqueous sodium bicarbonate and citric acid (2 mol l“1) then dried (MgS04) and concentrated at reduced pressure to give BOC-(γ-benzyl)-L-prolyl-L-glutamic acid dibenzyl ester (5.0 g, 95%).
(7-Benzyl)-L-prolyl-L-glutamic acid dibenzyl ester
A solution of BOC-(γ-benzyl)-L-prolyl-L-glutamic acid dibenzyl ester (3.4 g, 10 mmol), cooled to 0 °C, was treated with trifluoroacetic acid (25 ml) for 2 hr at room temperature. After removal of the volatiles at reduced pressure the residue was triturated with ether to give (γ-benzyl)-L-prolyl-L-glutamic acid dibenzyl ester (I).
N,N-Dimethylglycyl-L-prolyl-L-glutamic acid
A solution of dicyclohexylcarbodiimide (10.3 mmol) in dichloromethane (10 ml) was added to a stirred and cooled (0 °C) solution of (7-benzyl)-L-prolyl-L-glutamic acid dibenzyl ester (10 mmol), TVN-dimethylglycine (10 mmol) and triethylamine
(10.3 mmol) in dichloromethane (30 ml). The mixture was stirred at 0 °C overnight and then at room temperature for 3 h. After filtration, the filtrate was evaporated at reduced pressure. The resulting crude dibenzyl ester was dissolved in a mixture of ethyl acetate (30 ml) and methanol (30 ml) containing 10% palladium on charcoal (0.5 g) then hydrogenated at room temperature and pressure until the uptake of hydrogen ceased. The filtered solution was evaporated and the residue recrystallized from ethyl acetate to yield the tri-peptide derivative.
It will be evident that following the method of the Example, and using alternative amino acids or their amides or esters, will yield other compounds of Formula 1.
Testing; Material and Methods
The following experimental protocol followed guidelines approved by the
University of Auckland animal ethics committee.
Preparation of cortical astrocyte cultures for harvest of metabolised cell culture supernatant
One cortical hemisphere from a postnatal day 1 rat was used and collected into
4ml of DMEM. Trituration was done with a 5ml glass pipette and subsequently through an 18 gauge needle. Afterwards, the cell suspension was sieved through a lOOμm cell strainer and washed in 50ml DMEM (centrifugation for 5min at 250g). The sediment was resuspended into 20ml DMEM+10% fetal calf serum. 10 Milliliters of suspension was added into each of two 25cm3 flasks and cultivated at 37°C in the presence of 10% C02, with a medium change twice weekly. After cells reached confluence, they were washed three times with PBS and adjusted to Neurobasal/B27 and incubated for another 3 days. This supernatant was frozen for transient storage until usage at -80°C.
Preparation of striatal and cortical tissue from rat E18/E19 embryos
A dam was sacrificed by C02-treatment in a chamber for up to 4 minutes and was prepared then for cesarean section. After surgery, the embryos were removed from their amniotic sacs, decapitated and the heads put on ice in DMEM/F12 medium for striatum and PBS + 0.65% D(+)-glucose for cortex.
Striatal tissue extraction procedure and preparation of cells
Whole brain was removed from the skull with the ventral side facing upside in DMEM/F12 medium. The striatum was dissected out from both hemispheres under a stereomicroscope and the striatal tissue was placed into the Falcon tube on ice.
The collected striatal tissue was triturated by using a PI 000 pipettor in 1ml of volume. The tissue was triturated by gently pipetting the solution up and down into the pipette tip about 15 times, using shearing force on alternate outflows. The tissue pieces settled to the bottom of the Falcon tube within 30 seconds, subsequently the supernatant was transferred to a new sterile Falcon tube on ice. The supernatant contained a suspension of dissociated single cells. The tissue pieces underwent a second trituration to avoid excessively damaging cells already dissociated by over triturating them. 1 Milliliter of ice-cold DMEM/F12 medium was added to the tissue pieces in the first tube and triturated as before. The tissue pieces were allowed to settle and the supernatant was removed to a new sterile Falcon tube on ice. The cells were centrifuged at 250g for 5 minutes at 4°C. The resuspended cell pellet was ready for cell counting.
Plating and cultivation of striatal cells
Striatal cells were plated into Poly-L-Lysine (O.lmg/ml) coated 96-well plates (the inner 60 wells only) at a density of 200,000 cells /cm2 in Neurobasal/B27 medium (Invitrogen). The cells were cultivated in the presence of 5% C02 at 37°C under 100% humidity. Complete medium was changed on days 1, 3 and 6.
Cortical tissue extraction procedure and preparation of cells
The two cortical hemispheres were carefully removed by a spatula from the whole brain with the ventral side facing upside into a PBS +0.65% D(+)-glucose containing petri dish. Forcips were put into the rostral part (near B. olfactorius) of the cortex for fixing the tissue and two lateral – sagittal oriented cuttings were done to remove the paraform and entorhinal cortices. The next cut involved a frontal oriented cut at the posterior end to remove the hippocampal formation. A final frontal cut was done a few millimeters away from the last cut in order to get hold of area 17/18 of the visual cortex.
The collected cortices on ice in PBS+0.65% D(+)-glucose were centrifuged at 350g for 5min. The supernatant was removed and trypsin/EDTA (0.05%/0.53mM) was added for 8min at 37°C. The reaction was stopped by adding an equal amount of DMEM+10%) fetal calf serum. The supernatant was removed by centrifugation followed by two subsequent washes in Neurobasal/B27 medium.
The cells were triturated once with a glass Pasteur pipette in 1 ml of
Neurobasal/B27 medium and subsequently twice by using a 1ml insulin syringe with a 22 gauge needle. The cell suspension was passed through a lOOμm cell strainer and subsequently rinsed by 1ml of Neurobasal B27 medium. Cells were counted and adjusted to 50,000 cells per 60μl.
Plating and cultivation of cortical cells
96-well plates were coated with 0.2mg/ml Poly-L-Lysine and subsequently coated with 2μg/ml laminin in PBS, after which 60μl of cortical astrocyte-conditioned medium was added to each well. Subsequently, 60μl of cortical cell suspension was added. The cells were cultivated in the presence of 10% C02 at 37°C under 100%) humidity. At day 1, there was a complete medium change (1:1- Neurobasal/B27 and astrocyte-conditioned medium) with addition of lμM cytosine-β-D-arabino-furanoside (mitosis inhibitor). On the second day, 2/3 of medium was changed. On day 5, 2/3 of the medium was changed again.
Cerebellar microexplants from P8 animals: preparation, cultivation and fixation
The laminated cerebellar cortices of the two hemispheres were explanted from a P8 rat, cut into small pieces in PBS + 0.65% D(+)glucose solution and triturated by a 23gauge needle and subsequently pressed through a 125 μm pore size sieve. The microexplants that were obtained were centrifuged (60 g) twice (media exchange) into serum-free BSA-supplemented START V-medium (Biochrom). Finally, the
microexplants were reconstituted in 1500 μl STARTV-medium (Biochrom). For cultivation, 40μl of cell suspension was adhered for 3 hours on a Poly-D-Lysine
(O.lmg/ml) coated cover slip placed in 35mm sized 6-well plates in the presence of 5% C02 under 100% humidity at 34°C. Subsequently, 1ml of STARTV-medium was added together with the toxins and drugs. The cultures were monitored (evaluated) after 2-3 days of cultivation in the presence of 5% C02 under 100% humidity. For cell counting analysis, the cultures were fixed in rising concentrations of paraformaldehyde (0.4%, 1.2%, 3% and 4% for 3min each) followed by a wash in PBS.
Toxin and drug administration for cerebellar, cortical and striatal cells: analysis
All toxin and drug administration experiments were designed that 1/100 parts of okadaic acid (30nM and lOOnM concentration and 0.5mM 3-nitropropionic acid for cerebellar microexplants only), GPE (InM -ImM) and G-2Methyl-PE (InM-lmM) were used respectively at 8DIV for cortical cultures and 9DIV for striatal cultures. The incubation time was 24hrs. The survival rate was determined by a colorimetric end-point MTT-assay at 595nm in a multi-well plate reader. For the cerebellar microexplants four windows (field of 0.65 mm2) with highest cell density were chosen and cells displaying neurite outgrowth were counted.
Results
The GPE analogue G-2Methyl-PE exhibited comparable neuroprotective capabilities within all three tested in vitro systems (Figures 12-15).
The cortical cultures responded to higher concentrations of GPE (Figure 12) /or
G-2Methyl-PE (lOμM, Figure 13) with 64% and 59% neuroprotection, respectively.
Whereas the other 2 types of cultures demonstrated neuroprotection at lower doses of G-2Methyl-PE (Figures 14 and 15). The striatal cells demonstrated
neuroprotection within the range of InM to ImM of G-2Methyl-PE (Figure 15) while the postnatal cerebellar microexplants demonstrated neuroprotection with G-2Methyl-PE in the dose range between InM and lOOnM (Figure 14).
While this invention has been described in terms of certain preferred embodiments, it will be apparent to a person of ordinary skill in the art having regard to that knowledge and this disclosure that equivalents of the compounds of this invention may be prepared and administered for the conditions described in this application, and all such equivalents are intended to be included within the claims of this application.
PATENT
WO-2021026066
Composition and kits comprising trofinetide and other related substances. Also claims a process for preparing trofinetide and the dosage form comprising the same. Disclosed to be useful in treating neurodegenerative conditions, autism spectrum disorders and neurodevelopmental disorders.
Trofinetide is a synthetic compound, having a similar core structure to Glycyl-Prolyl-Glutamic acid (or “GPE”). Trofinetide has been found to be useful in treating neurodegenerative conditions and recently has been found to be effective in treating Autism Spectrum disorders and Neurodevelopmental disorders.
Formula (Ila),
Example 1: Trofinetide Manufacturing Process
In general, trofinetide and related compounds can be manufactured from a precursor peptide or amino acid reacted with a silylating or persilylating agent at one or more steps. In the present invention, one can use silylating agents, such as N-trialkylsilyl amines or N-trialkylsilyl amides, not containing a cyano group.
Examples of such silylating reagents include N,O-bis(trimethylsilyl)acetamide (BSA), N,O-bis(trimethylsilyl)trifluoroacetamide, hexamethyldisilazane, N-methyl-N-(trimethylsilyl)acetamide (TMA), N-methyl-N-(trimethylsilyl)trifluoroacetamide, N-(trimethylsilyl)acetamide, N-(trimethylsilyl)diethylamine, N-(trimethylsilyl)dimethylamine, 1-(trimethylsilyl)imidazole, 3-(trimethylsilyl)-2-oxazolidone.
Step 1: Preparation of Z-Gly-OSu
Several alternative procedures can be used for this step.
Procedure 1A
One (1) eq of Z-Gly-OH and 1.1 eq of Suc-OH were solubilized in 27 eq of iPrOH and 4 eq of CH2Cl2 at 21 °C. The mixture was cooled and when the temperature reached -4 °C, 1.1 eq of EDC.HCl was added gradually, keeping the temperature below 10 °C. During the reaction a dense solid appeared. After addition of EDC.HCl, the mixture was allowed to warm to 20 °C. The suspension was cooled to 11 °C and filtered. The cake was washed with 4.9 eq of cold iPrOH and 11 eq of IPE before drying at 34 °C (Z-Gly-OSu dried product -Purity: 99.5%; NMR assay: 96%; Yield: 84%).
Procedure 1B
This Procedure is for a variant of Procedure 1A, and differs by replacing iPrOH with ACN. One (1) eq of Z-Gly-OH and 1.1 eq of Suc-OH were solubilized in 22 eq of ACN at 35 °C. The mixture was cooled in an ice bath. When the temperature reached 1 °C, 0.9 eq of DCC in 5.5 eq of ACN was added gradually to keep the temperature below 5 °C. The coupling reaction took about 20 hrs. During the reaction, DCU precipitated and was removed by filtration at the end of the coupling. After filtration, DCU was washed with ACN to recover the product. The mixture of Z-Gly-OSu was then concentrated to reach 60% by weight. iPrOH (17 eq) was added to initiate the crystallization. Quickly after iPrOH addition a dense solid appeared. An additional 17 eq of iPrOH was needed to liquify the suspension. The suspension was cooled in an ice bath and filtered. The solid was washed with 9 eq of iPrOH before drying at 45 °C (Z-Gly-OSu dried product – Purity: 99.2%; HPLC assay: 99.6%; Yield: 71%).
Step 2: Preparation of Z-Gly-MePro-OH
Several alternative procedures can be used for this step.
Procedure 2A
One (1) eq of MePro.HCl was partially solubilized in 29 eq of CH2Cl2 at 35 °C with 1.04 eq of TEA and 1.6 eq of TMA. The mixture was heated at 35 °C for 2 hrs to perform the silylation. Then 1.02 eq of Z-Gly-OSu was added to the mixture. The mixture was kept at 35 °C for 3 hrs and then 0.075 eq of butylamine was added to quench the reaction. The mixture was allowed to return to room temperature and mixed for at least 15 min. The Z-Gly-MePro-OH was extracted once with 5% w/w NaHCO3 in 186 eq of water, then three times successively with 5% w/w NaHCO3 in 62 eq of water. The aqueous layers were pooled and the pH was brought to 2.2 by addition of 34 eq of HCl as 12N HCl at room temperature. At this pH, Z-Gly-MePro-OH formed a sticky solid that was solubilized at 45 °C with approximately 33 eq of EtOAc and 2.3 eq of iButOH. Z-Gly-MePro-OH was extracted into the organic layer and washed with 62 eq of demineralized water. The organic layer was then dried by azeotropic distillation with 11.5 eq of EtOAc until the peptide began to precipitate. Cyclohexane (12 eq) was added to the mixture to complete the precipitation. The suspension was cooled at 5 °C for 2 hrs and filtered. The solid was washed with 10 eq of cyclohexane before drying at 45 °C (Z-Gly-MePro-OH dried product – Purity: 100%; HPLC assay: 100%; Yield 79%).
Procedure 2B
This Procedure is for a variant of Procedure 2A. One (1) eq of MePro.HCl was partially solubilized in 36.6 eq of CH2Cl2 at 34 °C with 1.01 eq of TEA and 0.1 eq of TMA. Then 1.05 eq of Z-Gly-OSu was added to the mixture, followed by 1.0 eq of TEA. The mixture was maintained at 35 °C for approximately 1 hr, cooled to 25 to 30 °C and 0.075 eq of DMAPA was added to stop the reaction. One hundred (100) eq of water, 8.6 eq of HCl as 12N HCl and 0.3 eq of KHSO4 were added to the mixture (no precipitation was observed, pH=1.7). Z-Gly-MePro-OH was extracted into the organic layer and washed twice with 97 eq of demineralized water with 0.3 eq of KHSO4, then 100 eq of demineralized water, respectively. EtOAc (23 eq) was added to the mixture and CH2Cl2 was removed by distillation until the peptide began to precipitate. Cyclohexane (25 eq) was added to the mixture to complete the precipitation. The suspension was cooled at -2 °C overnight and filtered. The solid was washed with 21 eq of cyclohexane before drying at 39 °C (Z-Gly-MePro-OH dried product – Purity: 98.7%; NMR assay: 98%; Yield 86%).
Procedure 2C
In reactor 1, MePro.HCl (1 eq) was suspended in EtOAc (about 7 eq). DIPEA (1 eq) and TMA (2 eq) were added, and the mixture heated to dissolve solids. After dissolution, the solution was cooled to 0 °C. In reactor 2, Z-Gly-OH (1 eq) was suspended in EtOAc (about 15 eq). DIPEA (1 eq), and pyridine (1 eq) were added. After mixing, a solution was obtained, and cooled to -5 °C. Piv-Cl (1 eq) was added to reactor 2, and the contents of reactor 1 added to reactor 2. Upon completed addition, the contents of reactor 2 were taken to room temperature. The conversion from Z-Gly-OH to Z-Gly-MePro-OH was monitored by HPLC. When the reaction was complete, the reaction mixture was quenched with DMAPA (0.1 eq), and washed with an aqueous solution comprised of KHSO4, (about 2.5 wt%), NaCl (about 4 wt%), and conc. HCl (about 6 wt%) in 100 eq H2O. The aqueous layer was re-extracted with EtOAc, and the combined organic layers washed with an aqueous solution comprised of KHSO4 (about 2.5 wt%) and NaCl (about 2.5 wt%) in 100 eq H2O, and then with water (100 eq). Residual water was removed from the organic solution of Z-Gly-MePro-OH by vacuum distillation with EtOAc. The resulting suspension was diluted with heptane (about 15 eq) and cooled to 0 °C. The product was isolated by filtration, washed with cold heptane (about 7 eq), and dried under vacuum at 45 °C. Z-Gly-MePro-OH (85% yield) was obtained.
Step 3: Preparation of Z-Gly-MePro-Glu-OH
Several alternative procedures can be used in this step.
Procedure 3A
H-Glu-OH (1.05 eq) was silylated in 2 eq of CH2Cl2 with 3.5 eq of TMA at 65 °C. Silylation was completed after 2 hrs. While the silylation was ongoing, 1.0 eq of Z-Gly-MePro-OH and 1.0 eq of Oxyma Pure were solubilized in 24 eq of CH2Cl2 and 1.0 eq of DMA at room temperature in another reactor. EDC.HCl (1.0 eq.) was added. The activation rate reached 97% after 15 min. The activated Oxyma Pure solution, was then added to silylated H-Glu-OH at 40 °C and cooled at room temperature. Coupling duration was approximately 15 min, with a coupling rate of 97%. Addition of 8.2% w/w NaHCO3 in 156 eq of water to the mixture at room temperature (with the emission of CO2) was performed to reach pH 8. Z-Gly-MePro-Glu-OH was extracted in water. The aqueous layer was washed twice with 29 eq of CH2Cl2. Residual CH2Cl2 was removed by concentration. The pH was brought to 2.5 with 2.5N HCl, followed by 1.4 eq of solid KHSO4 to precipitate Z-Gly-MePro-Glu-OH. The mixture was filtered and the solid was washed with 3 x 52 eq of water. The filtered solid was added to 311 eq of demineralized water and heated to 55-60 °C. iPrOH (29 eq) was added gradually until total solubilization of the product. The mixture was slowly cooled to 10 °C under moderate mixing during 40 min to initiate the crystallization. The peptide was filtered and washed with 2 x 52 eq of water before drying at 45 °C (Z-Gly-MePro-Glu-OH dried product – Purity: 99.5%; NMR assay: 96%; Yield 74%).
Procedure 3B
One (1) eq of Z-Gly-MePro-OH and 1.05 eq of Suc-OH were solubilized in 40 eq of ACN and 30 eq of CH2Cl2 at room temperature. The mixture was cooled in an ice bath, and when the temperature was near 0 °C, 1.05 eq of DCC dissolved in 8 eq of ACN was added gradually, keeping the temperature below 5 °C. After addition of DCC, the mixture was progressively heated from 0 °C to 5 °C over 1 hr, then to 20 °C between 1 to 2 hrs and then to 45 °C between 2 to 5 hrs. After 5 hrs, the mixture was cooled to 5 °C and maintained overnight. The activation rate reached 98% after approximately 24 hrs. DCU was removed by filtration and washed with 13.5 eq of ACN. During the activation step, 1.1 eq of H-Glu-OH was silylated in 30 eq of ACN with 2.64 eq of TMA at 65 °C. Silylation was completed after 2 hrs. Z-Gly-MePro-OSu was then added gradually to the silylated H-Glu-OH at room temperature, with 0.4 eq of TMA added to maintain the solubility of the H-Glu-OH. The mixture was heated to 45 °C and 0.7 eq of TMA was added if precipitation occurred. The coupling duration was about 24 hrs to achieve a coupling rate of approximately 91%. The reaction was quenched by addition of 0.15 eq of butylamine and 2.0 eq of TEA. Water (233 eq) was added and the mixture concentrated until gelation occurred. Z-Gly-MePro-Glu-OH was extracted in water by addition of 5% w/w NaHCO3 in 233 eq of water and 132 eq of CH2Cl2. The aqueous layer was washed twice with 44 eq of CH2Cl2. Residual CH2Cl2 was removed by distillation. The pH was brought to 2.0 with 24 eq of HCl as 12N HCl followed by 75 eq of HCl as 4N HCl. At this pH, Z-Gly-MePro-Glu-OH precipitated. The mixture was cooled in an ice bath over 1 hr and filtered. The solid was washed with 186 eq of cold water before drying at 45 °C (Z-Gly-MePro-Glu-OH dried product – HPLC Purity: 98.4%; NMR assay: 100%; Yield 55%).
Procedure 3C
This Procedure is for a variant of Procedure 3A. H-Glu-OH (1.05 eq) was silylated in 3.7 eq of CH2Cl2 with 3.5 eq of TMA at 62 °C. Silylation was completed after approximately 1.5 to 2 hrs, as evidenced by solubilization. During the silylation step, 1.0 eq of Z-Gly-MePro-OH and 1.0 eq of Oxyma Pure were solubilized in 31.5 eq of CH2Cl2 at 22 °C. One (1.06) eq of EDC.HCl was added to complete the activation. The silylated H-Glu-OH was then added to the activated Oxyma Pure solution. The temperature was controlled during the addition to stay below 45 °C. Desilylation was performed by addition of a mixture of 2.5% w/w KHSO4 in 153 eq of water and 9 eq of iPrOH to reach a pH of 1.65. Residual CH2Cl2 was removed by concentration. The mixture was cooled to 12 °C to precipitate the Z-Gly-MePro-Glu-OH. The mixture was filtered and the solid was washed with 90 eq of water before drying at 36 °C.
Procedure 3D
This Procedure is for a variant of Procedure 3A. H-Glu-OH (1.05 eq.) was silylated in 3.9 eq of CH2Cl2 with 3.5 eq of TMA at 62 °C. Silylation was completed after 2 hrs, as evidenced by Solubilization. During the silylation step, 1 eq of Z-Gly-MePro-OH and 1 eq of Oxyma Pure were solubilized in 25 eq of CH2Cl2 at 23 °C. One (1) eq of EDC.HCl was added. To complete the activation, an additional 0.07 eq of EDC. HCl was added. Silylated H-Glu-OH was then added to the activated Oxyma Pure solution. Temperature was controlled during the addition to stay below 45 °C. Desilylation was performed by addition of a mixture of 2.5% w/w KHSO4 in 160 eq of water and 9.6 eq of iPrOH to reach pH 1.63.
Residual CH2Cl2 was removed by concentration. The mixture was cooled to 20 °C to precipitate the Z-Gly-MePro-Glu-OH. The mixture was filtered and the solid was washed with 192 eq of water before drying at about 25 °C for 2.5 days. The solid was then solubilized at 64 °C by addition of 55 eq of water and 31 eq of iPrOH. After solubilization, the mixture was diluted with 275 eq of water and cooled to 10 °C for crystallization. The mixture was filtered and the solid was washed with 60 eq of water before drying at 27 °C (Z-Gly-MePro-Glu-OH dried product – Purity: 99.6%; NMR assay: 98%; Yield 74%).
Procedure 3E
In reactor 1, H-Glu-OH (1.05 eq) was suspended in ACN (about 2.2 eq). TMA (about 3.5 eq) added, and the mixture was heated to dissolve solids. After dissolution, the solution was cooled to room temperature. In reactor 2, Z-Gly-MePro-OH (1 eq) was suspended in ACN (14 eq). Oxyma Pure (1 eq) and EDC.HCl (1 eq) were added. The mixture was stirred at room temperature until the solids dissolved. The contents of reactor 2 were added to reactor 1. The conversion from Z-Gly-MePro-OH to Z-Gly-MePro-Glu-OH was monitored by HPLC. Upon completion the reaction mixture was added to an aqueous solution comprised of KHSO4 (about 2.5 wt%) dissolved in about 100 eq H2O. ACN was removed from the aqueous suspension of Z-Gly-MePro-Glu-OH by vacuum distillation with H2O. After stirring at room temperature, the product in the resulting suspension was isolated by filtration and washed with water. The solid obtained was dissolved in an aqueous solution comprised of NaHCO3 (about 5 wt%) in 110 eq H2O, and recrystallized by addition of an aqueous solution comprised of KHSO4 (about 10 wt%) in 90 eq H2O. The product was isolated by filtration, washed with water, and dried under vacuum at 45 °C. Z-Gly-MePro-Glu-OH (75% yield) was obtained.
Step 4: Deprotection and Isolation of Trofinetide
Several alternative procedures can be used in this step.
Procedure 4A
Z-Gly-MePro-Glu-OH (1 eq) was suspended in water (about 25 eq) and EtOAc (about 15 eq). Pd/C (0.025 eq by weight and containing 10% Pd by weight) was added, and the reaction mixture hydrogenated by bubbling hydrogen through the reaction mixture at room temperature. The conversion from Z-Gly-MePro-Glu-OH to trofinetide was monitored by HPLC, and upon reaction completion the catalyst was removed by filtration, and the layers separated. Residual EtOAc was removed from the aqueous solution containing trofinetide by sparging with nitrogen or washing with heptane. The aqueous solution was spray-dried to isolate the product. Trofinetide (90% yield) was obtained. Alternatively, deprotection can be accomplished using MeOH only, or a combination of iPrOH and MeOH, or by use of ethyl acetate in water.
Procedure 4B
This Procedure is for a variant of Procedure 4A, excluding EtOAc. Z-Gly-MePro-Glu-OH (1 eq) was suspended in water (about 50 eq). Pd/C (0.05 eq, 5% Pd by weight) was added, and the reaction mixture hydrogenated at room temperature with a pressure of 5 bar. The conversion from Z-Gly-MePro-Glu-OH to trofinetide was monitored by HPLC. Upon
reaction completion the catalyst was removed by filtration, and the aqueous layer washed with EtOAc (about 5 eq). Residual EtOAc was removed from the aqueous solution containing trofinetide by sparging with nitrogen or washing with heptane. The aqueous solution was spray-dried to isolate the product. Trofinetide (90% yield) was obtained.
Procedure 4C
This Procedure is for a variant of Procedure 4A, replacing EtOAc with MeOH. Z-Gly-MePro-Glu-OH (1 eq) was suspended in MeOH (100 eq) and water (12 eq). Pd/Si (0.02 eq by weight) was added and the mixture was heated at 23 °C for the hydrogenolysis. Solubilization of the peptide occurred during the deprotection. The conversion from Z-Gly-MePro-Glu-OH to trofinetide was monitored by HPLC, and upon reaction completion the catalyst was removed by filtration and the layers were washed with MeOH and iPrOH. The solvents were concentrated under vacuum at 45 °C, and trofinetide precipitated. The precipitate was filtered and dried at 45 °C to provide trofinetide.
Procedure 4D
This Procedure is for a variant of Procedure 4A, replacing Pd/C with Pd/Si. One (1.0) eq of Z-Gly-MePro-Glu-OH was partially solubilized in 105 eq of MeOH and 12 eq of water. Pd/Si (0.02 eq by weight) was added and the mixture was heated at 23 °C for the hydrogenolysis. Solubilization of the peptide occurred during the deprotection. At the end of the deprotection (conversion rate approximately 99% after 1 hr), the catalyst was filtered off and washed with 20-30 eq of MeOH. iPrOH (93 eq) was added and MeOH was replaced by iPrOH by concentration at 45 °C under vacuum. The peptide was concentrated until it began to precipitate. The peptide was filtered and dried at 45 °C (H-Gly-MePro-Glu-OH dried product: Purity: 98.1%; NMR assay: 90%; Yield 81%).
Procedure 4E
This Procedure is for a variant of Procedure 4A, removing H2O and replacing Pd/C with Pd/Si. One (1.0) eq of Z-Gly-MePro-Glu-OH was partially solubilized in 44 eq of MeOH. Pd/Si type 340 (0.02 eq by weight) was added and the mixture was kept at 20 °C for the hydrogenolysis. Solubilization of the peptide occurred during the deprotection. At the end of the deprotection (conversion rate about 99.9%, after 3-3.5 hrs), the catalyst was filtered off and washed with 8 eq of MeOH. Deprotected peptide was then precipitated in 56 eq of iPrOH. After 30 min at 5 °C, the peptide was filtered and washed with three times with 11 eq of iPrOH before drying at 25 °C (H-Gly-MePro-Glu-OH dried product: Purity: 99.4%; HPLC assay: ~98%; Yield: 81%).
Procedure 4F
This Procedure is for a variant of Procedure 4A. One (1) eq of Z-Gly-MePro-Glu-OH was partially solubilized in 14 eq of EtOAc and 25 eq of water. Pd/C (0.01 eq by weight) was added and the mixture was kept at 20 °C for the hydrogenolysis. Solubilization of the peptide occurred during the deprotection. At the end of the deprotection (conversion rate about 100%, after about 3.5 hrs), the catalyst was filtered off and washed with a mixture of 3.5 eq of EtOAc and 6 eq of water. The aqueous layer was then ready for spray-drying (Aqueous H-Gly-MePro-Glu-OH peptide solution: Purity: 98.6%; Yield: ~95%).
Procedure 4G
This Procedure is for a variant of Procedure 4A, replacing Pd/C with Pd/Si, EtOAc with MeOH, and removing H2O. Pd/Si type 340 (0.02 eq by weight) was added to 2.9 vols of MeOH for pre-reduction during 30 min. One (1.0) eq of Z-Gly-MePro-Glu-OH was partially solubilized in 34 eq of MeOH. The reduced palladium was then transferred to the peptide mixture. The mixture was kept at 20 °C for the hydrogenolysis. Solubilization of the peptide occurred during the deprotection. Pd/C type 39 (0.007 eq by weight) was added to the mixture to increase reaction kinetics. At the end of the deprotection, the catalyst was filtered off and washed with 13.6 eq of MeOH. The deprotected peptide was then precipitated in 71 eq of iPrOH. After about 40 min, the peptide was filtered and washed with 35 eq of iPrOH. The peptide was dried below 20 °C and was then ready for solubilization in water and spray-drying.
Procedure 4H
This Procedure is for a variant of Procedure 4A. One (1.0) eq of Z-Gly-MePro-Glu-OH was partially solubilized in 24.8 eq of water and 13.6 eq of EtOAc. Pd/C type 39 (0.025 eq by weight) was added to the peptide mixture. The mixture was kept at 20 °C for the hydrogenolysis. Solubilization of the peptide occurred during the deprotection. At the end of the deprotection (19 hrs), the catalyst was removed by filtration and washed with 5.3 eq of water and 2.9 eq of EtOAc. The biphasic mixture was then decanted to remove the upper organic layer. The aqueous layer was diluted with water to reach an H-Gly-MePro-Glu-OH concentration suitable for spray-drying the solution.
Example 2: Alternative Trofinetide Manufacturing Process
An alternative method for synthesis of Trofinetide is based on U.S. Patent No.
8,546,530 adapted for a tripeptide as follows.
The persilylated compounds used to synthesis Formula (Ia) (trofinetide) are obtained by silylating a corresponding peptide or amino acid by reaction with a silylating agent, optionally in an organic solvent. The persilylated peptide or amino acid can be isolated and purified if desired. One can use the persilylated peptide or amino acid in situ, e.g. by combining a solution containing persilylated peptide or amino acid with a solution containing, optionally activated, peptide or amino acid.
In step 2, the persilylated compound of an amino acid is obtained by silylating a corresponding amino acid (for example, H-MePro-OH) by reaction with a silylating agent, optionally in an organic solvent. The persilylated amino acid can be isolated and purified if desired. One can use the persilylated amino acid in situ, e.g. by combining a solution containing the persilylated amino acid with a solution containing, optionally activated, amino acid (for example, Z-Gly-OH).
In step 3, the persilylated compound of an amino acid is obtained by silylating a corresponding amino acid (for example, H-Glu-OH) by reaction with a silylating agent, optionally in an organic solvent. The persilylated amino acid or peptide can be isolated and purified if desired. It is however useful to use the persilylated amino acid or peptide in situ, e.g. by combining a solution containing the persilylated amino acid with a solution containing, optionally activated (for example, by using EDC.HCl and Oxyma Pure), peptide (for example, Z-Gly-MePro-OH).
In the present invention, it is useful to use silylating agents, such as N-trialkylsilyl amines or N-trialkylsilyl amides, not containing a cyano group. Examples of such silylating reagents include N,O-bis(trimethylsilyl)acetamide (BSA), N,O-bis(trimethylsilyl)trifluoroacetamide, hexamethyldisilazane, N-methyl-N-(trimethylsilyl)acetamide (TMA), N-methyl-N-(trimethylsilyl)trifluoroacetamide, N-(trimethylsilyl)acetamide, N-(trimethylsilyl)diethylamine, N-(trimethylsilyl)dimethylamine, 1-(trimethylsilyl)imidazole, 3-(trimethylsilyl)-2-oxazolidone.
The reaction of step 2 is generally carried out at a temperature from 0 °C to 100 °C, optionally from 10 °C to 40 °C, and optionally from 15 °C to 30 °C.
The reaction of step 3 is generally carried out at a temperature from 0 °C to 100 °C, optionally from 10 °C to 60 °C, optionally from 15 °C to 50 °C.
In the reaction of step 2, generally 0.5 to 5 equivalents, optionally 1 to 3 equivalents, optionally about 1.5 to 2.5 equivalents of silylating agent are used relative to the molar amount of functional groups to be silylated. Use of 2 to 4 equivalents of silylating agent relative to the molar amount of functional groups to be silylated is also possible. “Functional groups to be silylated” means particular groups having an active hydrogen atom that can react with the silylating agent such as amino, hydroxyl, mercapto or carboxyl groups.
In the reaction of step 3, generally 0.5 to 5 equivalents, optionally 2 to 4.5 equivalents, optionally about 3 to 4 equivalents of silylating agent are used relative to the molar amount of functional groups to be silylated. Use of 2.5 to 4.5 equivalents of silylating agent relative to the molar amount of functional groups to be silylated is also possible.
It is understood that “persilylated” means an amino acid or peptide or amino acid analogue or peptide analogue in which the groups having an active hydrogen atom that can react with the silylating agent are sufficiently silylated to ensure that a homogeneous reaction medium for a coupling step is obtained.
In the process according to the invention, the reaction between the amino acid or peptide and the persilylated amino acid or peptide is often carried out in the presence of a carboxyl group activating agent. In that case the carboxylic activating reagent is suitably selected from carbodiimides, acyl halides, phosphonium salts and uronium or guanidinium salts. More optionally, the carboxylic activating agent is an acyl halide, such as isobutyl chloroformate or pivaloyl chloride or a carbodiimide, such as EDC.HC1 or DCC.
Good results are often obtained when using additional carboxylic activating reagents which reduce side reactions and/or increase reaction efficiency. For example, phosphonium and uronium salts can, in the presence of a tertiary base, for example, N,N-diisopropylethylamine (DIPEA) and triethylamine (TEA), convert protected amino acids into activated species. Other reagents help prevent racemization by providing a protecting reagent. These reagents include carbodiimides (for example, DCC) with an added auxiliary nucleophile (for example, 1-hydroxy-benzo triazole (HOBt), 1-hydroxy-azabenzotriazole (HOAt), or Suc-OH) or derivatives thereof. Another reagent that can be utilized is TBTU. The mixed anhydride method, using isobutyl chloroformate, with or without an added auxiliary nucleophile, is also used, as is the azide method, due to the low racemization associated with it. These types of compounds can also increase the rate of carbodiimide-mediated couplings. Typical additional reagents include also bases such as N,N-diisopropylethylamine (DIPEA), triethylamine (TEA) or N-methylmorpholine (NMM).
When the silylation is carried out in the presence of a solvent, said solvent is optionally a polar organic solvent, more optionally a polar aprotic organic solvent. An amide type solvent such as N,N-dimethylformamide (DMF) or N,N-dimethylacetamide (DMAC)
can be used. In the present invention for step 2, one can use an alkyl acetate solvent, in particular ethyl acetate is more particularly optional.
In the present invention for step 3, one can use a chlorinated hydrocarbon solvent or alkyl cyanide solvent, in particular dichloromethane or acetonitrile are more particularly optional.
In another embodiment, silylation is carried out in a liquid silylation medium consisting essentially of silylating agent and amino acid or peptide.
In the present invention, amino acid or peptide is understood to denote in particular an amino acid or peptide or amino acid analogue or peptide analogue which is bonded at its N-terminus or optionally another position, to a carboxylic group of an amino protected amino acid or peptide.
Example 3: Specifications for Compositions Containing Compounds of Formula (I)
1 ICH guideline Q3C on impurities: guideline for residual solvents
Example 4: Alternative Manufacturing of Trofinetide Example 1, Step 4, Procedure 4B
This Procedure is for a variant of Step 4, Procedure 4B. Z-Gly-MePro-Glu-OH (1 eq) was added in portions to Pd/C (0.027 eq by weight and containing 5% Pd by weight) in about 50 eq of water. The reaction mixture was hydrogenated at 20 °C at a pressure of 5 bar for at least 4 cycles of 4 hrs each. Pd/C (0.0027 eq by weight) was charged between cycles, as needed, to speed up the reaction. The conversion from Z-Gly-MePro-Glu-OH to trofinetide was monitored by HPLC. Upon reaction completion the catalyst was removed by filtration, washed with water (12.5 eq) and the aqueous layer washed with EtOAc (about 14 eq). After phase separation, residual EtOAc was removed from the aqueous solution containing
trofinetide by sparging with nitrogen under vacuum at 20 °C for about 3 hrs. The aqueous solution was filtered. The final concentration of trofinetide was about 25 wt% and the solution was then ready for spray-drying to isolate the product.
Example 5: Alternative Composition of Trofinetide
A composition comprising a compound of Formula (I)
or a stereoisomer, hydrate, or pharmaceutically acceptable salt thereof, and a compound of Formula (II):
or a stereoisomer, hydrate, or pharmaceutically acceptable salt thereof, and/or a compound of Formula (III):
or a stereoisomer, hydrate, or pharmaceutically acceptable salt thereof, wherein R1, R2, R3 and R4 independently are selected from the group consisting of hydrogen and C1-4 alkyl, provided that least one of R1, R2, R3 and R4 is C1-4 alkyl, and wherein the composition comprises at least 90 wt%, such as 91 wt%, 92 wt%, 93 wt%, 94 wt%, 95 wt%, 96 wt%, or 97 wt% of the compound of Formula (I) on an anhydrous basis.
Example 6: Alternative Composition of Trofinetide
A composition comprising a compound of Formula (Ia)
or a hydrate, or pharmaceutically acceptable salt thereof, and a compound of Formula (II):
or a stereoisomer, hydrate, or pharmaceutically acceptable salt thereof, and/or a compound of Formula (III):
or a stereoisomer, hydrate, or pharmaceutically acceptable salt thereof, wherein R1, R2, R3 and R4 independently are selected from the group consisting of hydrogen and C1-4 alkyl, provided that least one of R1, R2, R3 and R4 is C1-4 alkyl, and wherein the composition comprises at least 90 wt%, such as 91 wt%, 92 wt%, 93 wt%, 94 wt%, 95 wt%, 96 wt%, or 97 wt% of the compound of Formula (Ia) on an anhydrous basis.
Example 7: A Product of Trofinetide
A product, including a kit containing a dosage form with instructions for use, comprising a compound of Formula (Ia)
or a hydrate, or pharmaceutically acceptable salt thereof, and a compound of Formula (IIa)
or a hydrate, or pharmaceutically acceptable salt thereof, wherein the product comprises between 95 wt% and 105 wt%, such as 96 wt%, 97 wt%, 98 wt%, 99 wt%, 100 wt%, 101
wt%, 102 wt%, 103 wt%, or 104 wt% of the specified amount of the compound of Formula (Ia) in the product.
Example 8: A Product of Trofinetide
A product, including a kit containing a dosage form with instructions for use, comprising a compound of Formula (Ia)
or a hydrate, or pharmaceutically acceptable salt thereof, and a compound of Formula (IIa)
or a hydrate, or pharmaceutically acceptable salt thereof, and additionally comprising one or more compounds selected from the group consisting of Formula (III), Formula (IIIa), Formula (IV), Formula (V), Formula (VI), Formula (VII), Formula (VIII), and Formula (IX), wherein the composition comprises between 95 wt% and 105 wt%, such as 96 wt%, 97 wt%, 98 wt%, 99 wt%, 100 wt%, 101 wt%, 102 wt%, 103 wt%, or 104 wt% of the specified amount of the compound of Formula (Ia) in the product.
Example 9: Analysis of Products and Compositions
The products and compositions disclosed herein may be analyzed by liquid chromatography, a suitable chromatographic method using UPLC, e.g. using materials and conditions such as Waters Acquity CSH C18, 1.7 µm, 150 x 2.1 mm column, water with 0.1 % TFA (mobile phase A), and water/ACN 70/30 + 0.1 % TFA (mobile phase B), ranging from (4% phase A/6% phase B to 100% phase B and flushed with 4% phase A/6% phase B).
Flow rate: 0.35 ml/min, Column temperature: 40 °C, autosampler temperature: 4 °C, injection volume: 4 ml (e.g. prepared by weighing about 10 mg of powder in a 10 ml volumetric flask and diluted to volume with water). Examples of detectors are UV (ultraviolet, UV 220 nm) and MS (mass spectrometry).
INDUSTRIAL APPLICABILITY
This invention finds use in the pharmaceutical, medical, and other health care fields.
PATENT
WO2014085480 ,
claiming use of trofinetide for treating autism spectrum disorders including autism, Fragile X Syndrome or Rett Syndrome.
EP 0 366 638 discloses GPE (a tri-peptide consisting of the amino acids Gly-Pro- Glu) and its di-peptide derivatives Gly-Pro and Pro-Glu. EP 0 366 638 discloses that GPE is effective as a neuromodulator and is able to affect the electrical properties of neurons.
W095/172904 discloses that GPE has neuroprotective properties and that administration of GPE can reduce damage to the central nervous system (CNS) by the prevention or inhibition of neuronal and glial cell death.
WO 98/14202 discloses that administration of GPE can increase the effective amount of choline acetyltransferase (ChAT), glutamic acid decarboxylase (GAD), and nitric oxide synthase (NOS) in the central nervous system (CNS).
WO99/65509 discloses that increasing the effective amount of GPE in the CNS, such as by administration of GPE, can increase the effective amount of tyrosine hydroxylase (TH) in the CNS for increasing TH-mediated dopamine production in the treatment of diseases such as Parkinson’s disease.
WO02/16408 discloses GPE analogs capable of inducing a physiological effect equivalent to GPE within a patient. The applications of the GPE analogs include the treatment of acute brain injury and neurodegenerative diseases, including but not limited to, injury or disease in the CNS.
Example
The following non-limiting example illustrates the synthesis of a compound of the invention, NN-dimethylglycyl-L-prolyl-L-glutamic acid.
All starting materials and other reagents were purchased from Aldrich;
BOC = tert-butoxycarbonyl; Bn = benzyl.
BOC-(γ-benzyl)-L-prolyl-L-glutamic acid benzyl ester
To a solution of BOC-proline [Anderson GW and McGregor AC: J. Amer. Chem.
Soc: 79, 6180, 1957] (10 mmol) in dichloromethane (50 ml), cooled to 0 °C, was added triethylamine (1.39 ml, 10 mmol) and ethyl chloroformate (0.96 ml, 10 mmol). The resultant mixture was stirred at 0 °C for 30 minutes. A solution of dibenzyl L-glutamate (10 mmol) was then added and the mixture stirred at 0 °C for 2 hours then warmed to room temperature and stirred overnight. The reaction mixture was washed with aqueous sodium bicarbonate and citric acid (2 mol l“1) then dried (MgS04) and concentrated at reduced pressure to give BOC-(γ-benzyl)-L-prolyl-L-glutamic acid dibenzyl ester (5.0 g, 95%).
(7-Benzyl)-L-prolyl-L-glutamic acid dibenzyl ester
A solution of BOC-(γ-benzyl)-L-prolyl-L-glutamic acid dibenzyl ester (3.4 g, 10 mmol), cooled to 0 °C, was treated with trifluoroacetic acid (25 ml) for 2 hr at room temperature. After removal of the volatiles at reduced pressure the residue was triturated with ether to give (γ-benzyl)-L-prolyl-L-glutamic acid dibenzyl ester (I).
N,N-Dimethylglycyl-L-prolyl-L-glutamic acid
A solution of dicyclohexylcarbodiimide (10.3 mmol) in dichloromethane (10 ml) was added to a stirred and cooled (0 °C) solution of (7-benzyl)-L-prolyl-L-glutamic acid dibenzyl ester (10 mmol), TVN-dimethylglycine (10 mmol) and triethylamine
(10.3 mmol) in dichloromethane (30 ml). The mixture was stirred at 0 °C overnight and then at room temperature for 3 h. After filtration, the filtrate was evaporated at reduced pressure. The resulting crude dibenzyl ester was dissolved in a mixture of ethyl acetate (30 ml) and methanol (30 ml) containing 10% palladium on charcoal (0.5 g) then hydrogenated at room temperature and pressure until the uptake of hydrogen ceased. The filtered solution was evaporated and the residue recrystallized from ethyl acetate to yield the tri-peptide derivative.
It will be evident that following the method of the Example, and using alternative amino acids or their amides or esters, will yield other compounds of Formula 1.
PAPER
Tetrahedron (2005), 61(42), 10018-10035. (CLICK HERE)
The synthesis of ten proline-modified analogues of the neuroprotective tripeptide GPE is described. Five of the analogues incorporate a proline residue with a hydrophobic group at C-2 and two further analogues have this side chain locked into a spirolactam ring system. The pyrrolidine ring was also modified by replacing the γ-CH2 group with sulfur and/or incorporation of two methyl groups at C-5.
Graphical Abstract

PAPER
Bioorganic & Medicinal Chemistry Letters (2005), 15(9), 2279-2283
A series of GPE analogues, including modifications at the Pro and/or Glu residues, was prepared and evaluated for their NMDA binding and neuroprotective effects. Main results suggest that the pyrrolidine ring puckering of the Pro residue plays a key role in the biological responses, while the preference for cis or trans rotamers around the Gly-Pro peptide bond is not important.
Graphical abstract
A series of Pro and/or Glu modified GPE analogues is described. Compounds incorporating PMe and dmP showed higher affinity for glutamate receptors than GPE and neuroprotective effects similar to those of this endogenous tripeptide in culture hippocampal neurons exposed to NMDA.

PATENT
US 20060251649
WO 2006127702
US 20070004641
US 20080145335
WO 2012102832
WO 2014085480
US 20140147491
References
- ^ Bickerdike MJ, Thomas GB, Batchelor DC, Sirimanne ES, Leong W, Lin H, et al. (March 2009). “NNZ-2566: a Gly-Pro-Glu analogue with neuroprotective efficacy in a rat model of acute focal stroke”. Journal of the Neurological Sciences. 278 (1–2): 85–90. doi:10.1016/j.jns.2008.12.003. PMID 19157421. S2CID 7789415.
- ^ Cartagena CM, Phillips KL, Williams GL, Konopko M, Tortella FC, Dave JR, Schmid KE (September 2013). “Mechanism of action for NNZ-2566 anti-inflammatory effects following PBBI involves upregulation of immunomodulator ATF3”. Neuromolecular Medicine. 15 (3): 504–14. doi:10.1007/s12017-013-8236-z. PMID 23765588. S2CID 12522580.
- ^ Deacon RM, Glass L, Snape M, Hurley MJ, Altimiras FJ, Biekofsky RR, Cogram P (March 2015). “NNZ-2566, a novel analog of (1-3) IGF-1, as a potential therapeutic agent for fragile X syndrome”. Neuromolecular Medicine. 17 (1): 71–82. doi:10.1007/s12017-015-8341-2. PMID 25613838. S2CID 11964380.
- ^ Study Details – Rett Syndrome Study
- ^ Neuren’s trofinetide successful in Phase 2 clinical trial in Fragile X
| PHASE | STATUS | PURPOSE | CONDITIONS | COUNT |
|---|---|---|---|---|
| 3 | Enrolling by Invitation | Treatment | Rett’s Syndrome | 1 |
| 3 | Recruiting | Treatment | Rett’s Syndrome | 1 |
| 2 | Completed | Supportive Care | Injuries, Brain | 1 |
| 2 | Completed | Treatment | Fragile X Syndrome (FXS) | 1 |
| 2 | Completed | Treatment | Injuries, Brain | 1 |
| 2 | Completed | Treatment | Rett’s Syndrome | 2 |
| 2 | Terminated | Treatment | Concussions | 1 |
| 1 | Completed | Treatment | Brain Injuries,Traumatic | 2 |
| Legal status | |
|---|---|
| Legal status | US: Investigational New Drug |
| Identifiers | |
| IUPAC name[show] | |
| CAS Number | 853400-76-7 |
| PubChem CID | 11318905 |
| ChemSpider | 9493869 |
| UNII | Z2ME8F52QL |
| Chemical and physical data | |
| Formula | C13H21N3O6 |
| Molar mass | 315.322 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| SMILES[hide]C[C@]1(CCCN1C(=O)CN)C(=O)N[C@@H](CCC(=O)O)C(=O)O | |
| InChI[hide]InChI=1S/C13H21N3O6/c1-13(5-2-6-16(13)9(17)7-14)12(22)15-8(11(20)21)3-4-10(18)19/h8H,2-7,14H2,1H3,(H,15,22)(H,18,19)(H,20,21)/t8-,13-/m0/s1Key:BUSXWGRAOZQTEY-SDBXPKJASA-N |
////////////Tofinetide , NNZ 2566, PHASE 2, PHASE 3. NEUREN, Amino Acids, Peptides, Proteins,
CC1(CCCN1C(=O)CN)C(=O)NC(CCC(=O)O)C(=O)O
J. Med. Chem. 2025, 68, 2147−2182
Trofinetide (Daybue). Trofinetide (8) was developed by Neuren Pharmaceuticals and Acadia Pharmaceuticals for the treatment of rare childhood neurodevelopmental disorders and
was approved by the USFDA in March 2023 for adults and pediatric patients two years of age or older with Rett syndrome. 59 In most cases, Rett syndrome is caused by loss of-function mutations in the X-linked gene that encodes methyl CpG-binding protein 2 (MeCP2). 60,61 MeCP2 is a critical transcriptional regulator required for normal neurological development. In the past, treatment of Rett syndrome has
been limited to symptom management based on knowledge from treating other conditions, 62
but new research has focused on targeting the underlying genetic cause and finding agents to
restore MeCP2 function. Trofinetide is an orally available synthetic analog of glycine-proline-glutamate (GPE), the Nterminal tripeptide metabolite of insulin like growth factor-1 (IGF-1). GPE has been shown to partially reverse Rett-like symptoms in MeCP2 deficient mouse models and trofenitide was developed to have an improved pharmakokinetic profile to GPE. 64 Its use has shown significant improvement over placebo in clinical trials.
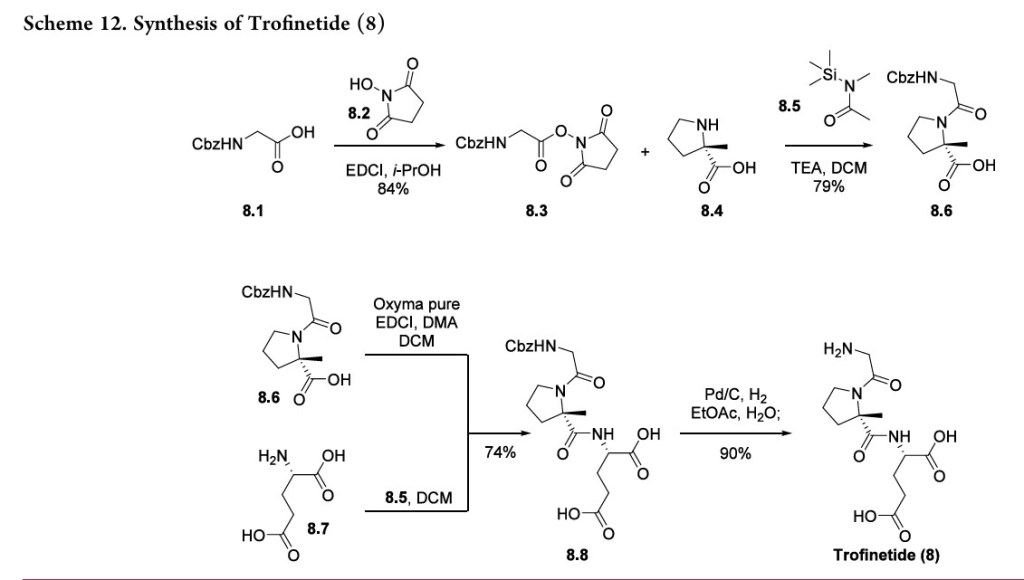
Anumberofaccountsrelated to the preparation of trofinetide have been reported with various protecting group strategies, but they are moreamenabletosmall-scaleproductionduetoreagent selection and challenging isolations requiring column chromatography.65−67 A commercially viable synthesis of the drug has been described by researchers at Neuren Pharmaceuticals and is depicted in Scheme 12.68
This synthesis takes advantage of in situ silyl protection/deprotection during its amidation steps to
avoid lengthy protecting group manipulations. Activation of Cbz protected glycine 8.1 with hydroxysuccinimide 8.2 using EDCI provided 8.3 in 84% yield as a direct drop crystallization
(isolated by crystallization directly from the reaction mixture). This activated ester was then coupled with commercially available methyl proline 8.4 69 by first silylation of the carboxylic acid of the proline analog in situ with 8.5, then adding 8.3 to couple with the amine. Deprotection of the silyl ester during the
workup provided amide 8.6 in 79% yield. In a similar sequence, 8.6 was activated with Oxyma Pure and EDCI while in a second vessel 8.7 was silyl protected using 8.5. Subsequent combination of these streams followed by workup and crystallization gave amide 8.8 in good yield. Finally, Cbz deprotection was
accomplished using Pd/C and hydrogen. Trofinetide (8) was extracted into the aqueous layer and isolated by spray drying in 90% yield.
(60) Kyle, S. M.; Vashi, N.; Justice, M. J. Rett syndrome: a
neurological disorder with metabolic components. Open Biol. 2018,
8, No. 170216.
(61) Collins, B. E.; Neul, J. L. Rett syndrome and MECP2 duplication
syndrome: disorders of MeCP2 dosage. Neuropsychiatr. Dis. Treat.
2022, 18, 2813−2835.
(62) Fu, C.; Armstrong, D.; Marsh, E.; Lieberman, D.; Motil, K.; Witt,
R.; Standridge, S.; Nues, P.; Lane, J.; Dinkel, T.; et al. Consensus
guidelines on managing Rett syndrome across the lifespan. BMJ
Paediatr. Open 2020, 4, No. e000717.
(63) Neul, J. L.; Percy, A. K.; Benke, T. A.; Berry-Kravis, E. M.; Glaze,
D.G.;Marsh,E.D.;Lin,T.;Stankovic,S.;Bishop,K. M.;Youakim,J.M.
Trofinetide for the treatment of Rett syndrome: a randomized phase 3
study. Nat. Med. 2023, 29, 1468−1475.
(64) Neul, J. L.; Percy, A. K.; Benke, T. A.; Berry-Kravis, E. M.; Glaze,
D. G.; Peters, S. U.; Jones, N. E.; Youakim, J. M. Design and outcome
measures of LAVENDER, a phase 3 study of trofinetide for Rett
syndrome. Contemp. Clin. Trials 2022, 114, No. 106704.
(65) Glass, L.; Bickerdike, M. J.; Snape, M. F. Treatment of autism
spectrum disorders using glycyl-l-2-methylprolyl-l-glutamic acid. US
20140147491, 2014.
(66) Glass, L.; Bickerdike, M. J.; Snape, M. F. Treatment of autism
spectrum disorders using glycyl L2012102832, 2012.-2-methylprolyl
L-glutamic acid. WO 2012102832, 2012
(67) Brimble, M.A.;Harris, P. W.R.;Sieg, F. Preparation of analogsof
glycyl-prolyl-glutamate as neuroprotective agents. US 20080145335,
2008
(68) Blower, C.; Peterson, M.; Shaw, J. M.; Bonnar, J. A.; Moniotte, E.
D. F. P.; Bousmanne, M. B. C.; Betti, C.; Decroos, K. W. L.; Ayoub, M.
Compositions of trofinetide. WO 2021026066, 2021.
(69) Beck, A. K.; Blank, S.; Job, K.; Seebach, D.; Sommerfeld, T.
Synthesis of (S)-2-methylproline: a general method for the preparation
of α-branched amino acids (L.-proline, 2-methyl-). Org. Synth. 1995, 72, 62−73
.
IMRECOXIB
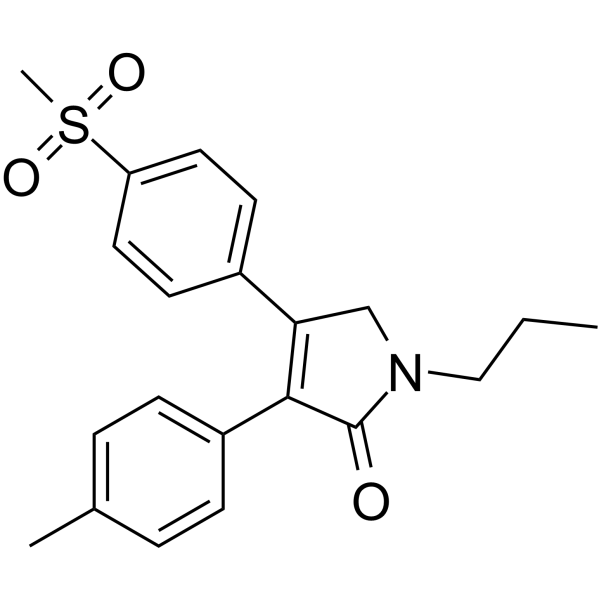
Imrecoxib (Hengyang)
CHINA 2012 osteoarthritis2H-Pyrrol-2-one, 1,5-dihydro-3-(4-methylphenyl)-4-[4-(methylsulfonyl)phenyl]-1-propyl-
3-(4-Methylphenyl)-4-[4-(methylsulfonyl)phenyl]-1-propyl-1,5-dihydro-2H-pyrrol-2-one395683-14-4[RN]
Imrecoxib was approved by China Food and Drug Administration (CFDA) on May 20, 2011. It was developed and marketed as 恒扬® by HengRui Pharmaceuticals.
Imrecoxib is a selective COX-2 inhibitor indicated for treatment of osteoarthritis.
恒扬® is available as tablet for oral use, containing 100 mg of free Imrecoxib, and the recommend dose is 100 mg twice daily.
Common name: Imrecoxib; BAP-909; BAP 909; BAP909
Trademarks: Hengyang
Molecular Formula: C21H23NO3S
CAS Registry Number: 395683-14-4
IUPAC Name: 4-(4-methane-sulfonyl-phenyl)-1-propyl-3-p-tolyl-1,5-dihydropyrrol-2-one
Molecular Weight: 369.48
SMILES: O=C1N(CCC)CC(C2=CC=C(S(=O)(C)=O)C=C2)=C1C3=CC=C(C)C=C3
Mechanism: COX-2 Inhibitor; Cyclooxygenase-2 Inhibitor
Activity: Treatment of Osteoarthritis; Analgesic; Antipyritic; Antiinflammatory Drug
Status: Launched 2011 (China)
Originator: HengRuiDrug Name:ImrecoxibResearch Code:BAP-909Trade Name:恒扬®MOA:Selective cyclooxygenase-2 (COX-2) inhibitorIndication:Osteoarthritis (OA)Status:ApprovedCompany:HengRui (Originator)Sales:ATC Code:Approved Countries or Area
| Approval Date | Approval Type | Trade Name | Indication | Dosage Form | Strength | Company | Review Classification |
|---|---|---|---|---|---|---|---|
| 2011-05-20 | Marketing approval | 恒扬 | Osteoarthritis (OA) | Tablet, Film coated | 100 mg | HengRui |
Reference:1. US7112605B2.Route 2
Reference:1. CN102206178A.
2. Chinese Chem. Lett. 2001, 12, 775-778.Route 3
Reference:1. CN104193664A.
Imrecoxib | NSAID | Treatment of Osteoarthritis | COX-2 Inhibitor
Imrecoxib [4-(4-methane-sulfonyl-phenyl)-1-propyl-3-p-tolyl-1,5-dihydropyrrol-2-one] is a novel and moderately selective cyclooxygenase-2 (COX-2) inhibitor that possesses anti-inflammatory effect by inhibition of COX-2 mRNA expression. It belongs to the family of non-steroid anti-inflammtory drugs (NSAIDs). Imrecoxib was found to inhibit COX-1 and COX-2 with IC50 value of 115 ± 28 nM and 18 ± 4 nM, respectively [1].
| Imrecoxib: 2D and 3D Structure |
Imrecoxib effectively inhibited carrageenan-induced acute inflammation at the doses of 5, 10, and 20 mg-kg-1 ig and adjuvant-induced chronic inflammation at the doses of 10 and 20 mg-kg -1·d-1 ig.
NSAIDs and Imrecoxib:
Non-steroidal anti-inflammatory drugs (NSAIDs) are used extensively for the treatment of inflammatory conditions, including pain-releasing, anti-pyretic and rheumatoid arthritis. These functions are believed to inhibit the enzyme cyclooxygenase (COX) that is involved in the biosynthesis of prostaglandins G and H from arachidonic acid. So far two isozymes of COX are known: COX-1 and COX-2. COX-1 is constitutively produced in a variety of tissues and appears to be important to the maintenance of normal physiological functions, including gastric and renal cytoprotection. The COX-2 is an inducible isozyme, which is produced in cells under the stimulation of endotoxins, cytokines, and hormones and catalyzes the production of prostaglandins which cause inflammation.
The currently therapeutic use of NSAIDs has been associated with the inhibition of both COX-1 and COX-2 and causes well-known side effects at the gastrointestinal and renal level. Therefore, the selective COX-2 inhibitors could provide anti-inflammatory agents devoid of the undesirable effects associated with classical, nonselective NSAIDs. In addition, COX-2 is over-expressed in colon cancer tissue. COX-2 inhibitors possess potential prophylactic and therapeutic application to colon cancer.
Imrecoxib is designed in a manner such that it has “moderate selectivity” for COX-2 over COX-1. This balanced inhibition to both COX-1 and COX-2 was pursued to maintain the homeostasis of the two enzymes in the body,which is presumably critical to normal functions of the cardiovascular system.
Imrecoxib was launched in China with the trade name of Hengyang for the treatment of osteoarthritis in May 2011. Hengyang is available as tablet for oral use, containing 100 mg of free Imrecoxib, and the recommend dose is 100 mg twice daily.
SYN
Imrecoxib Synthesis
Chin Chem Lett 2001, 12, 775-778 (also Ref 2. This route is quoted as industrial method in various texts)
CN104193664A (an improvement here as Br is replaced with Cl)

US7112605B2 (primary reference for synthesis routes)

Identification:

| 1H NMR (Estimated) for Imrecoxib |
Experimental: 1H-NMR (CDCl3, TMS, 400MHz) 1.008 (3H, t, J = 7.2Hz), 1.701-1.756 (2H, m), 2.376 (3H, s), 3.078 (3H, s), 3.575 (2H, t, J = 7.2Hz), 4.317 (2H, s), 7. 175 (2H, d, J = 8.0Hz), 7.294 (2H, d, J = 8.0Hz), 7.505 (2H, t, J = 6.8Hz), 7.870 (2H, t, J = 6.8Hz)
Sideeffects:
Being a mild COX-2 inhibitor, it is expected not to cause any serious cardiovascular risks. Similarly, it should not have any serious gastrointestinal problems too, as it not a good inhibitor of COX-1. None of the reports though have listed any serious adverse event reported by patients in the clinical trials.
References:
- Cheng, G. F.;et. al. Imrecoxib: A novel and selective cyclooxygenase 2 inhibitor with anti-inflammatory effect. Acta Pharmacol Sin 2004, 25(7), 927-931.
- Zhang, F.;et. al.Method for preparing imrecoxib. CN102206178A
- Chao, W.;et. al. Synthesis method of imrecoxib. CN104193664A
- Bai, A. P.;et. al. Synthesis and in vitro Evaluation of a New Class of Novel Cyclooxygenase-2 Inhibitors: 3, 4-diaryl-3-pyrrolin-2 ones.Chin Chem Lett 2001, 12, 775-7785.
- Guo, Z. Discovery of imrecoxib. Chin J New Drugs2012, 21, 223.
- Guo, Z.;et. al. Sulfonyl-containing 3,4-diaryl-3-pyrrolin-2-ones, preparation method, and medical use thereof. US7112605B2
![MS 2 spectrum of the [M þ H] þ ion (m/z 370) of imrecoxib (inset, full-scan mass spectrum).](https://www.researchgate.net/profile/Dafang_Zhong/publication/6930659/figure/fig4/AS:394580018122760@1471086616006/MS-2-spectrum-of-the-M-th-H-th-ion-m-z-370-of-imrecoxib-inset-full-scan-mass.png)
SYN
Imrecoxib (Hengyang)
Imrecoxib, a new non-steroid anti-inflammtory drug (NSAID), was launched in China with the trade name of Hengyang for the treatment of osteoarthritis in 2012. It was originally designed and synthesized by Guo and co-workers at the Institute of Materia Medica (IMM) of the Chinese Academy of Medical Sciences in collaboration with Hengrui Pharmaceuticals.88 Imrecoxib, which is a moderately selective COX-2 inhibitor (with IC50 values against COX-1 and COX-2 being 115 ± 28 and 18 ± 4 nM, respectively),89 is the subject of twwo synthetic routes reported across several publications.90–93
The most likely process-scale route to this drug is described in Scheme 15, 93 which began with 2-bromo-40 -(methylsulfonyl)-acetophenone (84) and p-tolylacetic acid (85) as starting materials. In the presence of base, a-bromoketone 84 was treated with acid 85 which resulted in lactone 86 in 72% yield across the two-step sequence. Exposure of lactone 86 with propylamine triggered a ring-opening-ring closing reaction, which resulted in imrecoxib (XIII) directly in 85% yield.93

88. Guo, Z. R. Chin. J. New Drugs 2012, 21, 223.
89. Chen, X. H.; Bai, J. Y.; Shen, F.; Bai, A. P.; Guo, Z. R.; Cheng, G. F. Acta Pharmacol. Sin. 2004, 25, 927.
90. Bai, A. P.; Guo, Z. R.; Hu, W. H.; Shen, F.; Cheng, G. F. Chin. Chem. Lett. 2001, 12, 775.
91. Guo, Z.; Cheng, G.; Chu, F.; Yang, G.; Xu, B. CN Patent 1134413 C, 2001.
92. Guo, Z.; Cheng, G.; Chu, F. US Patent 2004/0029951 A1, 2004.
93. Zhang, F. Y.; Shen, X. M.; Sun, P. Y. CN Patent 102206178 A, 2011
Patent
CN 111747879
PATENT
CN 111747874
CN 111747873
CN 110386891
CN 109553564
CN 109553563
CN 108997188
CN 108947884
CN 108912030
CN 108864003
CN 108707100
CN 107586268
CN 104193664
CN 102206178
CN 101774958
US 20040029951
PATENT
CN 109678775
https://patents.google.com/patent/CN102206178A/en
Ai Rui former times cloth (N-n-propyl-3-p-methylphenyl-4-is to methylsulfonyl phenyl-3-pyrrolidin-2-one) is the nonsteroidal anti-inflammatory drug that a kind of appropriateness suppresses COX-2; put down in writing the synthetic method of Ai Rui former times cloth in the prior art (US20040029951), may further comprise the steps:
1) is raw material with the methylsulfonyl methyl phenyl ketone, makes alpha-brominated methylsulfonyl methyl phenyl ketone through bromo;
2) sodium borohydride reduction of alpha-brominated methylsulfonyl methyl phenyl ketone obtains the Styrene oxide 98min. derivative;
3) reaction of Styrene oxide 98min. derivative and Tri N-Propyl Amine generates N-n-propyl-capable biology of beta-hydroxyphenyl ethamine;
4) tolyl-acetic acid and the reaction of excessive thionyl chloride are generated the methylbenzene Acetyl Chloride 98Min.;
5) methylbenzene Acetyl Chloride 98Min. and the capable biological respinse of N-n-propyl-beta-hydroxyphenyl ethamine are generated N-n-propyl-N-[2-hydroxyl-2-to the methylsulfonyl styroyl] phenylacetamide;
6) Jone ‘ s reagent or pyridine chromium trioxide oxidation N-n-propyl-N-[2-hydroxyl-2-are to the methylsulfonyl styroyl] phenylacetamide obtains the capable biology of oxo phenylacetamide;
7) the above-mentioned oxo phenylacetamide of condensation makes end product Ai Rui former times cloth under the alkaline medium effect.
Because existing preparation method’s route is longer, and relate to reduction, oxidation, steps such as acid amides coupling, solvent load is big, the cost height, particularly to use oxygenants such as Jone ‘ s reagent or pyridine chromium trioxide in the oxidation step, low and the product of this oxidation step productive rate is difficult for separation and purification, and is difficult to control because chromium metal residual quantity control criterion in bulk drug is extremely strict, thereby makes this preparation method be difficult to be applicable to scale operation.
Synthetic route 1
Step 1), preparation alpha-brominated methylsulfonyl methyl phenyl ketone (III)
51.0g 4-methylsulfonyl methyl phenyl ketone and 760mL acetic acid is added to has magnetic agitation, in three mouthfuls of glass flask of the 1000mL of thermometer and constant pressure funnel.Be heated to 40 ℃, beginning slowly drips 41.1g liquid bromine, after dripping, continues to stir 30 minutes at 40 ℃.Reaction solution 50 ℃ concentrate after, add entry, stir, filter, washing, oven dry obtains the thick product of 70.5g, adds ethyl acetate/normal hexane mixed solvent, reflux 1 hour, slowly be cooled to 25 ℃, filtering drying gets the alpha-brominated methylsulfonyl methyl phenyl ketone of 56.5g off-white color solid (III), yield 80.0%.
1H-NMR(CDCl 3,TMS,400MHz):3.120(3H,s),4.485(2H,s),8.101(2H,dd,J=2.0Hz),8.191(2H,dd,J=2.0Hz)
MS(M+1):279.05
Step 2), prepare 4-(4-methylsulfonyl phenyl)-3-(4-aminomethyl phenyl)-2,5-dihydrofuran-2-ketone (II)
Experiment condition A
With the alpha-brominated methylsulfonyl methyl phenyl ketone of 44.3g (III), 24.0g 4-methylphenyl acetic acid and 600mL acetonitrile are added to and have magnetic agitation, in the 500mL there-necked flask of thermometer and constant pressure funnel.Be added dropwise to the 24.0mL triethylamine by constant pressure funnel, temperature is controlled at 25 ℃, after adding, continues to stir 1 hour.Add the 36.0mL triethylamine again, reaction solution is heated to 75 ℃, stirring reaction 18 hours.Cool to 25 ℃, concentrate, add ethyl acetate, washing, organic phase concentrates the back and adds ethyl acetate and ethanol, stirs, and filters and obtains 28.0g light yellow solid compound (II), yield 53.4%.
1H-NMR(CDCl 3,TMS)2.398(3H,s),3.091(3H,s),5.192(2H,s),7.216(2H,d,J=8.0Hz),7.292(2H,d,J=8.0Hz),7.543(2H,d,J=8.0Hz),7.933(2H,d,J=8.0Hz)
MS(M+1):329.02
Similarly, compound (II) can prepare under experiment condition B, C, D.
Experiment condition B
With the alpha-brominated methylsulfonyl methyl phenyl ketone of 5.0g (III), 2.7g 4-methylphenyl acetic acid and 70mL acetonitrile are added to and have magnetic agitation, in the 100mL there-necked flask of thermometer and constant pressure funnel.Be added dropwise to the 2.3mL tetramethyl guanidine by constant pressure funnel, temperature is controlled at 20 ℃, after adding, continues to stir 1.5 hours.Add the 4.6mL tetramethyl guanidine again, 20 ℃ of stirring reactions 2 hours.Concentrate, add ethyl acetate, washing, organic phase concentrates the back and adds ethyl acetate and ethanol, stirs, and filters and obtains 2.5g light yellow solid compound (II), yield 42.0%.
Experiment condition C
With the alpha-brominated methylsulfonyl methyl phenyl ketone of 1.85g (III), 1.0g 4-methylphenyl acetic acid and 20mL ethanol are added to and have magnetic agitation, in the 50mL there-necked flask of thermometer and constant pressure funnel.Be added dropwise to the 1.0mL triethylamine by constant pressure funnel, temperature is controlled at 25 ℃, after adding, continues to stir 3 hours.Add the 2.0mL triethylamine again, 80 ℃ of stirring reactions 18 hours.Concentrate, add ethyl acetate, washing, organic phase concentrates the back and adds ethyl acetate and ethanol, stirs, and filters and obtains 0.83g light yellow solid compound (II), yield 38.1%.
Experiment condition D
With the alpha-brominated methylsulfonyl methyl phenyl ketone of 1.0g (III), 0.54g 4-methylphenyl acetic acid and 12mL acetonitrile are added to and have magnetic agitation, in the 50mL there-necked flask of thermometer and constant pressure funnel.Add 1.0g salt of wormwood, 25 ℃ were reacted 2 hours.50 ℃ of stirring reactions are 5 hours then.Concentrate, add ethyl acetate, washing, organic phase concentrates the back and adds ethyl acetate and ethanol, stirs, and filters and obtains 0.13g light yellow solid compound (II), yield 11%.
Step 3), preparation N-n-propyl-3-p-methylphenyl-4-are to methylsulfonyl phenyl-3-pyrrolidin-2-one (Ai Rui former times cloth (I))
Experiment condition A
With the 25.0mL Tri N-Propyl Amine, be added drop-wise in the 17.5mL acetic acid at 10 ℃, add the back and stir, in the Tri N-Propyl Amine acetate that generates, add 10.0g compound (II).Under the nitrogen protection, be heated to 160 ℃, stirring reaction 8 hours.Cool to 40 ℃, add methylene dichloride and water, standing demix.Organic phase concentrates in the residue of back and adds ethanol, and reflux cools to 25 ℃, filters, and oven dry obtains 8.2g white solid product compound (I), yield 72.8%.
1H-NMR(CDCl 3,TMS,400MHz)1.008(3H,t,J=7.2Hz),1.701-1.756(2H,m),2.376(3H,s),3.078(3H,s),3.575(2H,t,J=7.2Hz),4.317(2H,s),7.175(2H,d,J=8.0Hz),7.294(2H,d,J=8.0Hz),7.505(2H,t,J=6.8Hz),7.870(2H,t,J=6.8Hz)
MS(M+1):370.17
Similarly, compound (I) can prepare under experiment condition B, C, D.
Experiment condition B
2.9g Tri N-Propyl Amine hydrochloride and 1.0g compound (II) are mixed, under the nitrogen protection, be heated to 170 ℃, stirring reaction 2 hours.Cool to 40 ℃, add methylene dichloride and water, standing demix.Organic phase concentrates in the residue of back and adds ethanol, and reflux cools to 25 ℃, filters, and oven dry obtains 0.9g white solid product compound (I), yield 80.0%.
Experiment condition C
Digest compound (II) with 2.0,3 milliliters of Tri N-Propyl Amines, 1.75 gram Tri N-Propyl Amine hydrochlorides add in the tube sealing of nitrogen protections, are heated to 140 ℃, react 20 hours.Be cooled to room temperature, add methylene dichloride and water, standing demix.Organic phase concentrates in the residue of back and adds ethanol, and reflux cools to 25 ℃, filters, and oven dry obtains 1.8g white solid product compound (I), yield 80.0%.
Experiment condition D
With the 0.5mL Tri N-Propyl Amine, be added drop-wise in the 0.35mL acetic acid at 10 ℃, add the back and stir, in the Tri N-Propyl Amine acetate that generates, add 0.5g compound (II).Under the nitrogen protection, be heated to 120 ℃, stirring reaction 4 hours.Cool to 40 ℃, add methylene dichloride and water, standing demix.Obtain 0.14g compound (I) after organic phase is concentrated and purified, yield 24.2%.Publication numberPriority datePublication dateAssigneeTitleCN104072467A *2014-07-072014-10-01太仓博亿化工有限公司Synthesis method of 5-chloro-2-benzofuranyl-p-chlorophenyl-oneCN104193664A *2014-08-222014-12-10山东铂源药业有限公司Synthesis method of imrecoxibCN107586268A *2016-07-072018-01-16江苏恒瑞医药股份有限公司A kind of preparation method of imrecoxib and its intermediateCN108864003A *2018-06-152018-11-23江苏美迪克化学品有限公司A kind of preparation method of imrecoxib intermediate and imrecoxibCN108947884A *2018-06-292018-12-07江苏美迪克化学品有限公司A kind of Preparation Method And Their Intermediate of imrecoxibCN109553564A *2017-09-252019-04-02江苏恒瑞医药股份有限公司A kind of purification process of imrecoxibCN109678775A *2017-10-182019-04-26江苏恒瑞医药股份有限公司A kind of crystal form and preparation method thereof of COX-2 selective depressantCN107586268B *2016-07-072021-01-19江苏恒瑞医药股份有限公司Preparation method of dapoxib and intermediate thereofPublication numberPriority datePublication dateAssigneeTitleUS5489693A *1992-04-281996-02-06Linz; GuenterCyclic imino derivatives, pharmaceutical compositions containing these compounds and processes preparing themCN101386590A *2007-09-132009-03-18中国医学科学院药物研究所Pyrrolidone containing hydroxymethyl and carboxyl, preparation method and medicament composition and use thereofCN101497580A *2009-01-092009-08-05华南理工大学HIV-1 inhibitor 2-pyrrolidinone derivative, as well as synthesizing method and use thereof
PAPER
Chinese Chemical Letters (2001), 12(9), 775-778.
PATENT
CN 110386891,

CLIP
For Chinese drugmaker Hengrui, R&D plans pan out
Ambitious program to launch innovative drugs starts to pay off for generics producerby Jean-François TremblayJULY 17, 2017 | APPEARED IN VOLUME 95, ISSUE 29

Credit: Jean-François Tremblay/C&ENHengrui recently invested in a custom-made phage-display library screening system for its Shanghai lab.
Launching their own innovative pharmaceuticals is a common goal for managers of generic drug firms. But it remains a dream for many. Jiangsu Hengrui Medicine, one of China’s largest generic drug makers, has advanced further than most. It has already launched two of its own drugs in China and licensed rights to another to a U.S. firm.
JIANGSU HENGRUI MEDICINE AT A GLANCE
▸ Headquarters: Lianyungang, Jiangsu, China
▸ 2016 sales: $1.6 billion
▸ 2016 profits: $390 million
▸ Employees: More than 13,000, 2,000 of whom work for a Shanghai-based unit developing and commercializing innovative drugs
▸ Innovative drug R&D staff: 800
Obtaining these results required substantial resources, though. Back in 2004, Hengrui built a large R&D lab in Shanghai, hired world-class researchers to lead it, equipped the facility with the latest instruments, and staffed it with hundreds of scientists.
Initially, the project looked like a money pit. In Chinese industry circles, many doubted that it would amount to anything. But revenues from the company’s innovative drugs are starting to pour in, and R&D at Hengrui is well on its way to financial sustainability.
Over the past 10 years, China has made great strides in growing an innovative drug industry. For all the talk, cynics say, China has yet to foster a blockbuster with $1 billion or more in annual sales. But as Hengrui and other Chinese firms launch their own drugs at home and license the foreign rights to others, it is becoming clear that an innovative drug industry is taking root.
“Producing generic drugs funds our R&D,” says Weikang Tao, a Hengrui vice president who doubles as chief executive officer of Shanghai Hengrui, the company’s innovative drug subsidiary.
Overall, Hengrui invests more than 10% of its sales in R&D, “which is big by Chinese standards,” Tao says. The drug giant Pfizer by comparison spent about 15% of its sales on R&D in 2016. With sales of $1.6 billion last year, Hengrui does most of its business in China. But it also exports finished drugs to the U.S., making it one of the few Chinese firms to have the U.S. Food & Drug Administration’s okay to do so.
Hengrui was formed in 1970 as a state-owned company. It began investing in its own R&D in 2004 and has since cultivated an innovative drug subsidiary that employs 2,000 people, including more than 800 at a Shanghai lab and about 20 at a subsidiary in Princeton, N.J. Other staffers work in the usual functions found in an innovative drug firm: clinical trial management, regulatory affairs, marketing and sales, and so on.
The Shanghai subsidiary recruits in China and internationally. Tao, who joined Hengrui in 2014, is a Chinese-trained physician who earned a Ph.D. in molecular and cell biology at the University of Medicine & Dentistry of New Jersey. He focused on tumor cell biology during a postdoc at Princeton University and worked in research at Merck & Co. for 10 years. Hengrui is constantly hiring, he notes.
Hengrui’s research facilities appear to be well equipped. Earlier this year the firm opened a biologics drug lab and a pilot plant for process development in Shanghai. “We spent nearly $7 million just on equipment for the biologics lab,” Tao says.

The lab is equipped with a custom-made automated phage-display library screening system that speeds up the process of discovering antibody drugs. “The machine can do automatically in a few hours what would otherwise take days for several scientists,” says Jiakang Sun, group leader of in vivo pharmacology at Shanghai Hengrui Pharmaceutical. With the phage-display system, Sun adds, a library displaying millions of human antibodies can be screened in vitro to find antibodies that bind to a specific antigen.
However well-staffed and well-equipped, Hengrui’s labs are still smaller than those of Merck or other major drug firms. But Hengrui has made notable strides recently. In 2015, it became the first Chinese firm to license a drug candidate to a U.S. firm. Incyte agreed to pay $25 million up front, and several hundred million dollars more once certain milestones are met, for the rights outside China and Taiwan to camrelizumab, a cancer treatment in Phase III human clinical trials in China.
In China, Hengrui’s priority market, the firm launched the osteoarthritis treatment imrecoxib in 2011 and the gastric cancer drug apatinib in 2014. The two will eventually achieve combined annual sales of $160 million, Hengrui expects.
Together with the licensing deal with Incyte, this will allow the firm to nearly recoup its R&D investment. Launching a few more compounds, particularly in the U.S., would make innovative R&D at Hengrui solidly profitable. The company is making good progress in that direction. A neutropenia treatment awaits final market approval in China, and five others have reached Phase III trials. Hengrui also has drugs in Phase I clinical trials in the U.S.
“I wouldn’t say that our lab is more productive than a lab operated by a multinational drug firm,” Tao says. Merck and other major players operate excellent facilities staffed by top people, he says. “But I would say that our researchers work very hard, and our decision-making at the top is very quick.”
Unlike biotech start-ups that tend to be built around groundbreaking technology, promising drug leads, or star researchers, Hengrui at first approached innovative drug development with a conservative strategy designed to reduce the risk of failure.
Relying on developmental compounds licensed from other organizations, the company initially aimed to develop drugs with the same mechanisms of action as others already on the market. Imrecoxib, for example, is part of the well-known family of COX-2 inhibitor anti-inflammatory drugs.
Later, it sought to invent compounds offering slight improvements over existing ones. Today, Hengrui is aiming to launch pharmaceuticals that are clearly superior to the competition. The company’s ultimate goal, Tao says, is to develop groundbreaking pharmaceuticals.
“We went from me-too to me-better to now best in class, and then we will do first in class,” he says.
And as Hengrui’s research strategy has become more ambitious, its scientists have broadened the range of diseases and drugs that they work on. Six or seven years ago, Tao says, Hengrui limited itself to the development of small-molecule drugs that treat cancer. Today the company is looking at small molecules, peptides, antibodies, antibody-drug conjugates, and other drug types to treat diseases as diverse as psoriasis and diabetes. “We have expanded our focus,” Tao says.
Most research is conducted in-house, Tao says. This includes medicinal chemistry, process chemistry, biology, drug metabolism, and pharmacokinetics. But the company leans on contract research firms for certain specific tasks, such as developing animal models. “They help accelerate our R&D,” Tao says.
Although Hengrui is a pioneer in launching new drugs in China, several other Chinese firms have made progress in advancing their own drug development. For instance, in the southern city of Dongguan, the generic drug producer HEC Pharma is conducting Phase II trials of the hepatitis C drug yimitasvir.
In Beijing, the biotech firm BeiGene just sold the U.S. company Celgene rights in much of the world to one of its immuno-oncology compounds for $263 million. Celgene also agreed to inject $150 million into BeiGene.
Chinese companies increasingly have the resources required to sustain innovative drug discovery and development, China watchers say. “Hengrui has the financial resources and the commitment to become a world-class innovative drugmaker,” says George Baeder, a former pharmaceutical industry executive who is now a director of China Global Insight, a California-based think tank.
But developing drugs and selling them are two different things, Baeder warns. “It is easy to underestimate the complexity a firm faces when moving into the arena of innovative medicines,” he says. “Chinese companies typically lack the capabilities in medical affairs, marketing, and sales needed to build a successful franchise.”
For the time being, Hengrui’s innovative drug subsidiary will stay focused on developing new drugs and not worry about the fine points of marketing them. Tao expects the former will keep his firm busy. “Don’t be surprised if several of our drugs begin clinical trials in the U.S., Europe, and Australia in the next year or two,” he says.
Rich pipeline
Hengrui boasts a diverse portfolio of drugs in late-stage development.
| China approval stage | Name | Application | Mechanism or target |
|---|---|---|---|
| Phase II clinical trials | Hetrombopaga | Idiopathic thrombocytopenia | Thrombopoietin receptor agonist |
| HR7056 | Anesthesia | na | |
| Pyrotiniba | Non-small cell lung cancer | EGFR/HER2 | |
| SHR3680b | Prostate cancer | Androgen receptor | |
| Phase III | Apatinib | Liver and non-small cell lung cancer | VEGFR-2 |
| Camrelizumabc | Cancer | PD-1 blocker | |
| Pyrotinib | HER2-positive breast cancer | EGFR/HER2 | |
| Retagliptin | Type 2 diabetes | Dipeptidyl peptidase 4 | |
| SHR3824 | Type 2 diabetes | SGLT2 inhibitor | |
| New drug application (China) | Mecapegfilgrastim | Neutropenia | PEG G-CSF |
| Launched | Apatinib | Gastric cancer | VEGFR-2 |
| Imrecoxib | Osteoarthritis | COX-2 inhibitor |
a In Phase I in U.S. b In Phase I in Australia. c The U.S. firm Incyte has acquired the rights to this drug outside China. na = not available. Source: Hengrui
////////////////Imrecoxib, Hengyang, CHINA 2012, osteoarthritis
Telacebec
Telacebec
- Molecular FormulaC29H28ClF3N4O2
- Average mass557.006 Da
Telacebec, IAP6, CAS No. 1334719-95-7телацебек [Russian] [INN]تيلاسيبيك [Arabic] [INN]特雷贝克105731334719-95-7[RN]55G92WGH3X
6-Chloro-2-ethyl-N-(4-{4-[4-(trifluoromethoxy)phenyl]-1-piperidinyl}benzyl)imidazo[1,2-a]pyridine-3-carboxamide
Imidazo[1,2-a]pyridine-3-carboxamide, 6-chloro-2-ethyl-N-[[4-[4-[4-(trifluoromethoxy)phenyl]-1-piperidinyl]phenyl]methyl]-Q203Q-203T56 AN DNJ C2 HG BVM1R D- AT6NTJ DR DOXFFF
Qurient Therapeutics and Russia licensee Infectex are developing telacebec, an oral formulation which targets QcrB subunit of the cytochrome bc1 complex, for treating multi drug resistant or extensively drug resistant Mycobacterium tuberculosis infection. Qurient is also investigating telacebec for treating buruli ulcer (an infection caused by Mycobacterium ulcerans ). In January 2021, a global phase II trial was expected to begin by December 2021 for the treatment of buruli ulcer.
syn
Angewandte Chemie, International Edition, 57(4), 1108-1111; 2018

PATENT
WO-2021018387
Novel crystalline forms of telacebec , processes for their preparation and compositions comprising them are claimed. Also claimed is their use for treating bacterial infection.
Different forms of 6-chloro-2-ethyl-AT-(4-(4-(4- (trifluoromethoxy)phenvDpiperidine-i-vDbenzvDimidazolT.2-alpyridine- 3-carboxamide
The present invention relates to different forms of the compound 6-chloro-2-ethyl-lV-(4-(4-(4-(trifhioromethoxy)phenyl)piperidine-i-yl)benzyl)imidazo[i,2-a]pyridine-3-carboxamide and to methods of making such forms/compounds. The present invention furthermore relates to mono-acid addition salts thereof, to methods of making such mono-acid addition salts and to pharmaceutical compositions comprising any of the aforementioned compounds. Furthermore, the present invention relates to uses of any of these compounds.
Tuberculosis as a disease continues to result in millions of deaths each year. Inadequate use of chemotherapy has led to an increasing number of drug resistant cases. This situation is likely to worsen with the emergence of extremely resistant strains to all currently known drugs. Current chemotherapy consists of compounds that directly target Mycobacterium tuberculosis, either by neutralizing general information pathways and critical processes such as RNA polymerization and protein synthesis inhibition or by interfering with mycobacterial specific cell envelop synthesis. The most widely used dedicated anti-tubercular drugs isoniazid, ethionamide, and pyriazin amide are pro-drugs that first require activation. They are administered to a patient for a course of several months. Patients infected with multi-drug resistant strains of M. tuberculosis may have to undergo combination therapies for extended periods of time.
WO 2011/113606 describes various anti-tubercular compounds and their use in the treatment of bacterial infections, including compound“Q203” which chemically is 6-chloro-2-ethyl-!V-(4-(4-(4-(trifluoromethoxy)phenyl)piperidine-i-yl)benzyl)imidazo[i,2-a]pyridine-3-carboxamide. In a publication by Pethe et al. (Nature Medicine, 19, 1157-1160 (2013), this compound is reported to be active against tuberculosis by interfering with the bacterial energy metabolism, inhibiting cytochrome bci activity which is an essential component of the electron transport chain required for synthesis of ATP.
Whilst the compound shows promise for future therapy of tuberculosis and related infections, there continues to be a need for forms thereof that are particularly suitable for pharmaceutical administration. In particular there is a need to provide forms that are showing an improved solubility in comparison to the free base of this compound. Furthermore, there is a need in the art to provide for forms that show an improved stability.
In a first aspect the present invention relates to a compound 6-chloro-2-ethyl-N-(4-(4-(4-(trifluoromethoxy)phenyl)piperidine-i-yl)benzyl)imidazo[i,2-a]pyridine-3-carboxamide ditosylate having the structure
PATENT
WO2011113606 .
WO 2017049321
WO 2012143796
PAPER
Scientific reports (2019), 9(1), 8608.
Angewandte Chemie, International Edition (2018), 57(4), 1108-1111.
European journal of medicinal chemistry (2017), 136, 420-427.
European Journal of Medicinal Chemistry (2017), 136, 420-427.
European journal of medicinal chemistry (2017), 125, 807-815.
Nature communications (2016), 7, 12393.
Nature medicine (2013), 19(9), 1157-60
PAPER
Journal of Medicinal Chemistry (2014), 57(12), 5293-5305.
https://pubs.acs.org/doi/10.1021/jm5003606J. Med. Chem. 2014, 57, 12, 5293–5305
Publication Date:May 28, 2014
https://doi.org/10.1021/jm5003606

A critical unmet clinical need to combat the global tuberculosis epidemic is the development of potent agents capable of reducing the time of multi-drug-resistant (MDR) and extensively-drug-resistant (XDR) tuberculosis therapy. In this paper, we report on the optimization of imidazo[1,2-a]pyridine amide (IPA) lead compound 1, which led to the design and synthesis of Q203 (50). We found that the amide linker with IPA core is very important for activity against Mycobacterium tuberculosis H37Rv. Linearity and lipophilicity of the amine part in the IPA series play a critical role in improving in vitro and in vivo efficacy and pharmacokinetic profile. The optimized IPAs 49 and 50 showed not only excellent oral bioavailability (80.2% and 90.7%, respectively) with high exposure of the area under curve (AUC) but also displayed significant colony-forming unit (CFU) reduction (1.52 and 3.13 log10 reduction at 10 mg/kg dosing level, respectively) in mouse lung.
6-Chloro-2-ethyl-N-(4-{4-[4-(trifluoromethoxy)phenyl]piperidin-1-yl}benzyl)imidazo[1,2-a]pyridine-3-carboxamide (50)
Mp = 164.0 °C; 1H NMR (400 MHz, CDCl3) δ 1.37 (t, J = 7.6 Hz, 3H), 1.82–1.97 (m, 4H), 2.64–2.70 (m, 1H), 2.80–2.87 (m, 2H), 2.93 (q, J = 7.6 Hz, 2H), 3.80–3.83 (m, 2H), 4.61 (d, J = 5.2 Hz, 2H), 6.00 (br t, J = 5.2 Hz, 1H), 6.96–6.99 (m, 2H), 7.15 (d, J = 8.0 Hz, 2H), 7.24–7.30 (m, 5H), 7.52 (dd, J = 9.6, 0.8 Hz, 1H), 9.53 (dd, J = 2.0, 0.8 Hz, 1H); 13C NMR (100 MHz, CDCl3) δ 13.3, 23.6, 33.4, 42.0, 43.3, 50.4, 115.4, 117.0, 121.2, 121.6, 121.9, 126.3, 128.2, 128.3, 128.7, 128.9, 144.5, 144.7, 147.7, 151.4, 151.5, 161.2; 19F NMR (376 MHz, CDCl3) δ 58.31 (s, 3F); LC/MS (ESI) m/z 557 [M + H]+; HRESIMS calcd for C29H29ClF3N4O2 [M + H]+ 557.1926, found 557.1918.




19F NMR (376 MHz, CDCl3) δ 58.31 (s, 3F);

13C NMR (100 MHz, CDCl3) δ 13.3, 23.6, 33.4, 42.0, 43.3, 50.4, 115.4, 117.0, 121.2, 121.6, 121.9, 126.3, 128.2, 128.3, 128.7, 128.9, 144.5, 144.7, 147.7, 151.4, 151.5, 161.2;

1H NMR (400 MHz, CDCl3) δ 1.37 (t, J = 7.6 Hz, 3H), 1.82–1.97 (m, 4H), 2.64–2.70 (m, 1H), 2.80–2.87 (m, 2H), 2.93 (q, J = 7.6 Hz, 2H), 3.80–3.83 (m, 2H), 4.61 (d, J = 5.2 Hz, 2H), 6.00 (br t, J = 5.2 Hz, 1H), 6.96–6.99 (m, 2H), 7.15 (d, J = 8.0 Hz, 2H), 7.24–7.30 (m, 5H), 7.52 (dd, J = 9.6, 0.8 Hz, 1H), 9.53 (dd, J = 2.0, 0.8 Hz, 1H);
CLIP
June 3, 2019. Qurient press release:
SEONGNAM-SI, South Korea–(BUSINESS WIRE)– Qurient Co. Ltd. today announced positive results from the Phase 2a EBA (early bactericidal activity) clinical trial for telacebec (Q203), a first-in-class, orally-available antibiotic for the treatment of tuberculosis (TB). Telacebec is a selective inhibitor with high specificity for the cytochrome bc1 complex of Mycobacterium tuberculosis. This complex is a critical component of the electron transport chain, and inhibition disrupts the bacterium’s ability to generate energy.
The EBA trial assessed the pharmacokinetics, safety, and activity of telacebec in three dose strength (100 mg, 200 mg and 300 mg) in the treatment of adult patients with pulmonary TB. Telacebec met the primary objective of rate of change in the time to positivity (TTP) in sputum over days 0 to 14. Telacebec was safe and well tolerated throughout the different dose strengths. Full results from EBA trial are expected to be presented at future scientific meetings.
Phase 2. EBA began July 2018 in South Africa. As of March 2019, study is active, not enrolling.
June 2018. Q203 has a non-proprietary name assigned: telacebec. USAN: -cebec Cytochrome bc1 complex inhibitors in Mycobacterium tuberculosis.
Phase 1. Description from clinicaltrials.gov: Randomized, double-blind, placebo-controlled, dose-escalation study in healthy male and female volunteers. Subjects randomly assigned to 1 of 7 treatment cohorts (Cohorts 1 – 7) of 8 subjects each, receiving either Q203 or placebo (6 active treatment : 2 placebo) in a fasting state. Dose escalation to the next cohort may be considered when at least 6 out of 8 subjects, in a cohort, completes all procedures and none of the subjects has a clinically significant adverse event (AE) that is being followed, or at the discretion of the PI if no drug-related serious adverse events (SAEs) have occurred. A food effect cohort will be enrolled to test administration of Q203 in a fed state, at 100 mg dose level (this dose level may change based on PK analysis results). Subjects who received 100mg dose in a fasting state will return and receive the second dose, with food. Subjects will be followed up for AEs, SAE or pregnancy for 30 days postdrug administration.
Related Links
Qurient Press Release. June 2019.Kalia NP et al. 2017. Exploiting the synthetic lethality between terminal respiratory oxidases to kill M. tuberculosis and clear host infection.. PNAS.114.7426
Related Links
- Qurient website
- 2013 Pethe K–Nature Medicine–Discovery of Q203, a potent clinical candidate for the treatment of tuberculosis
- 2014 Kang S — JMC – Lead Optimization of a Novel Series of Imidazo[1,2-a]pyridine Amides Leading to a Clinical Candidate (Q203) as a Multi- and Extensively-Drug-Resistant Anti-tuberculosis Agent
//////////////Telacebec, IAP6, 1334719-95-7, PHASE 2, QURIENT, TUBERCULOSIS, телацебек , تيلاسيبيك , 特雷贝克 , Q 203
Iguratimod
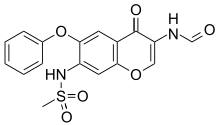
Iguratimod
- Molecular FormulaC17H14N2O6S
- Average mass374.368 Da
- UNII-4IHY34Y2NVигуратимодإيغوراتيمود艾拉莫德
123663-49-0[RN]
3-Formylamino-7-methylsulfonylamino-6-phenoxy-4H-1-benzopyran-4-one
4IHY34Y2NV8176IGU
Methanesulfonamide, N-[3-(formylamino)-4-oxo-6-phenoxy-4H-1-benzopyran-7-yl]-
N-(3-Formamido-4-oxo-6-phenoxy-4H-chromen-7-yl)methanesulfonamide
product patent US4954518
Research Code:T-614
Trade Name:Iremod® / Kolbet® / Careram®
MOA:Nuclear factor NF-κB activation inhibitor
Indication:Rheumatoid arthritis
Status:Approved
Company:Simcere (Originator) , Taisho Toyama,EisaiSales:ATC Code:
Approved Countries or Area
| Approval Date | Approval Type | Trade Name | Indication | Dosage Form | Strength | Company | Review Classification |
|---|---|---|---|---|---|---|---|
| 2012-06-29 | Marketing approval | Careram | Rheumatoid arthritis | Tablet, Film coated | 25 mg | Eisai | |
| 2012-06-29 | Marketing approval | Kolbet | Rheumatoid arthritis | Tablet, Film coated | 25 mg | Toyama Chemical, Taisho Toyama |
| Approval Date | Approval Type | Trade Name | Indication | Dosage Form | Strength | Company | Review Classification |
|---|---|---|---|---|---|---|---|
| 2011-08-15 | Marketing approval | 艾得辛/Iremod | Rheumatoid arthritis | Tablet, Film coated | 25 mg | Simcere |
Iguratimod was first approved by China Food and Drug Administration (CFDA) on August 15, 2011, then approved by Pharmaceuticals and Medicals Devices Agency of Japan (PMDA) on June 29, 2012. It was developed by Simcere and marketed as 艾得辛®/Iremod® by Simcere and as Kolbet® by Taisho Toyama and by Eisai in Japan.
Iguratimod is a nuclear factor NF-κB activation inhibitor used in the treatment of rheumatoid arthritis.
Iremod® is available as tablet for oral use, containing 25 mg of free Iguratimod, and the recommended dose is 25 mg once daily or 25 mg at a time, twice daily.
Iguratimod: (Iremod)-First approval: 2009
Iguratimod is a disease-modifying antirheumatic drug (DMARD) that was approved for use in rheumatoid arthritis (RA) patients in China and Japan in 2009
Toyama Chemical , Taisho Toyama Pharmaceutical , Eisai , Simcere and Tianjin Institute of Pharmaceutical Research have codeveloped and launched iguratimod, an inflammatory cytokine and IL-6 gene inhibiting compound that also inhibits immunoglobulin production in B cells. Iguratimod is indicated for the oral treatment of rheumatoid arthritis.
Iguratimod is an anti-inflammatory small molecule drug used for the treatment of rheumatoid arthritis, together with methotrexate in Japan and China.[1] As of 2015 the biological target was not known, but it prevents NF-κB activation and subsequently selectively inhibits COX-2 and several inflammatory cytokines.[1]
Adverse effects include elevated transaminases, nausea, vomiting, stomach pain; rashes, and itchiness.[1]
It is a derivative of 7-methanesulfonylamino-6-phenoxychromones and is a chromone with two amide groups; it was first published in 2000.[1][2] It was submitted for regulatory approval in Japan in 2003; the application was withdrawn in 2009, and it was resubmitted with additional data in 2011 and approved for marketing in Japan in 2012.[1] Eisai and Toyama Chemical market it in Japan.[3] Approval was obtained in China in 2011 by Simcere, independently of the Japanese originators.[1][4]
During discovery and development it was called T-614 and it is marketed under the names Careram and Kolbet.[5]
Syn
Indian Pat. Appl., 2014MU01507

SYN


Reference:1. US4954518.Route 2
Reference:1. Chem. Pharm. Bull. 2000, 48, 131-139.
2. Chin. J. New. Drugs. 2006, 15, 2042-2044.
3. Shanghai Chem. Ind. 2007, 32, 22-24.Route 3
Reference:1. CN1462748A.
2. Chem. Pharm. Bull. 2000, 48, 131-139.
SYN
AU 8823489; CH 679397; FR 2621585; GB 2210879; JP 1995267943; US 4954518
The reduction of 3-nitro-4-phenoxyphenol (I) with Fe, aqueous HCl gives 3-amino-4-phenoxyphenol (II), which is acylated with methanesulfonyl chloride by means of pyridine in dichloromethane affording 3-(methylsulfonamido)-4-phenoxyphenol (III). The condensation of (III) with 3-chloropropionic acid (IV) by means of NaOH in water gives 3-[3-(methylsulfonamido)-4-phenoxyphenoxy]propionic acid (V), which is cyclized by means of polyphosphoric acid at 65-70 C yielding 7-(methylsulfonamido)-6-phenoxy-3,4-dihydro-2H-1-benzopyran-4-one (VI). The bromination of (VI) with Br2 in CHCl3 affords 3-bromo-7-(methylsulfonamido)-6-phenoxy-3,4-dihydro-2H-1-benzopyran-4-one (VII), which is treated with sodium azide in DMF at 70-75 C giving 3-amino-7-(methylsulfonamido)-6-phenoxy-2H-1-benzopyran-4-one (VIII). Finally, this compound is formylated with formic acid in acetic anhydride.
SYN
Chem Pharm Bull 2000,48(1),131
A preparative-scale synthetic route for T-614 has been reported: The reaction of 4-chloro-3-nitroanisole (I) with potassium phenolate in hot DMF gives 4-phenoxy-3-nitroanisole (II), which is reduced to the corresponding 3-amino compound (III) by treatment with Fe and HCl. The reaction of (III) with mesyl chloride in pyridine affords the sulfonamide (IV), which is acylated with 2-aminoacetonitrile (V) and AlCl3 in nitrobenzene/HCl giving the 2-aminoacetophenone (VI). Formylation of (VI) at the NH2 with acetic formic anhydride yields the formamide (VII), which is demethylated with AlCl3 and NaI in acetonitrile affording the phenol (VIII). Finally, this compound is cyclized with dimethylformamide dimethylacetal in DMF.

SYN
https://www.sciencedirect.com/science/article/abs/pii/S0968089614001230
Synthetic approaches to the 2012 new drugs
Hong X. Ding, … Christopher J. O’Donnell, in Bioorganic & Medicinal Chemistry, 2014

13 Iguratimod (Careram®, Iremod®)
Iguratimod, which was discovered by Toyama Pharmaceuticals and jointly co-developed with Eisai in Japan, was approved by the PMDA (Pharmaceuticals and Medical Devices Agency) of Japan on June 29, 2012 for the treatment of rheumatoid arthritis.83 This drug was also independently developed by Simcere Pharmaceutical Group and is marketed as Iremod® in China. The drug exhibited inhibitory effects on granuloma inflammation, and was shown to be efficacious for the prevention of joint destruction in adjuvant arthritis.84,85 While several synthesis of iguratimod have been published,86 the most likely scale synthesis, which does not require chromatographic purification, is described in Scheme 14.87
The synthesis began with commercially available 3-nitro-4-chloro anisole (78) which was reacted with potassium phenoxide (generated from phenol and potassium t-butoxide at 110 °C) to provide the corresponding nitrophenyl ether which was subsequently reduced and sulfonylated to furnish sulfonamide 79. Next, this diphenyl ether was submitted to a Friedel–Crafts reaction with aminoacetonitrile hydrochloride which gave rise to aminomethylacetophenone 80 in 90% yield. This aminoketone was then formylated with formic trimethylacetic anhydride 81 at room temperature to afford formamide 82 in 91% yield, and this material was immediately subjected to O-demethylation conditions with aluminum trichloride and sodium iodide in acetonitrile to give the phenol 83 in 95% yield. Finally, treatment of the aminomethyl acetophenone phenol 83 with N,N-dimethylformamide dimethylacetal in DMF at low temperatures furnished iguratimod (XII) in 87% yield
83. Eisai and Toyama Chemical Receive Approval to Market Anti-rheumatic Agent Iguratimod in Japan, 2012, http://www.eisai.com/news/news201239.html, [Access Date: 2012-July-29].
84. Tanaka, K.; Shimotori, T.; Makino, S.; Aikawa, Y.; Inaba, T.; Yoshida, C.; Takano, S. Arzneim.-Forsch. 1992, 42, 935.
85. Tanaka, K.; Makino, S.; Shimotori, T.; Aikawa, Y.; Inaba, T.; Yoshida, C. Arzneim.-Forsch. 1992, 42, 945.
86. Takano, S.; Yoshida, C.; Inaba, T.; Tanaka, K.; Takeno, R.; Nagaki, H.; Shimotori, T.; Makino, S. US Patent 4954518 A, 1990.
87. Inaba, T.; Tanaka, K.; Takeno, R.; Nagaki, H.; Yoshida, C.; Takano, S. Chem. Pharm. Bull. 2000, 48, 131.
SYN

https://chemistry-europe.onlinelibrary.wiley.com/doi/abs/10.1002/slct.202003553A Convenient Synthesis of Iguratimod‐Amine Precursor via NHC‐Catalyzed Aldehyde‐Nitrile Cross Coupling ReactionNithya MurugeshProf. Ramasamy KarvembuDr. Seenuvasan VedachalamFirst published: 24 November 2020https://doi.org/10.1002/slct.202003553
A protocol for the synthesis of iguratimod‐amine precursor has been developed using N‐heterocyclic carbene (NHC)‐catalyzed aldehyde‐nitrile cross coupling reaction with overall atom efficiency of 71 %. The first step involves a nucleophilic aromatic substitution (SNAr) of 1‐chloro‐4‐methoxy‐2‐nitrobenzene (1) with phenol to produce 4‐methoxy‐2‐nitro‐1‐phenoxybenzene (2) which further undergoes nitro reduction followed by mesylation to produce N‐(5‐methoxy‐2‐phenoxyphenyl)methanesulfonamide (4). Furthermore, it was subjected to Vilsmeier‐Haack formylation and demethylation (using BBr3) to produce N‐(4‐formyl‐5‐hydroxy‐2‐phenoxyphenyl)methanesulfonamide (6). Subsequently, O‐alkylation followed by NHC‐catalyzed aldehyde‐nitrile cross coupling yields the amine precursor of iguratimod (8).
N-(3-Amino-4-oxo-6-phenoxy-4H-chromen-7-yl)methanesulfonamide (8):4=4. S. Vedachalam, J. Zeng, B. K. Gorityala, M. Antonio, X.-W. Liu, Org. Lett. 2010, 12, 352–355.
Compound 7 (290 mg, 0.83 mmol) and triazolium carbene catalyst (34 mg, 0.1245 mmol) were dissolved in dry CH2Cl2 under N2 atmosphere. To this, DBU (24.7 µL, 0.16 mmol) was added at room temperature, and the mixture was stirred for 12 h. After the completion of reaction, the reaction mixture was dried, and the residue was purified by column chromatography to yield compound 8. Yield: 200 mg, 70 %; m. p. 162 ℃; 1H NMR (500 MHz, DMSO-d6): δ 8.25 (s, 1H), 7.96 (s, 1H), 7.78 (s, 1H), 7.45 (t, J = 8.0 Hz, 2H), 7.25–7.21 (m, 3H), 7.16 (s, 1H), 4.91 (s, 2H), 3.23 (s, 3H); 13C NMR (125 MHz, DMSO-d6): δ 171.2, 155.0, 152.1, 150.4, 137.9, 133.0, 132.8, 131.0 (2C), 125.9, 122.4, 121.1 (2C), 117.2, 110.0, 39.0; FTIR (KBr): v 3423, 3347, 1620, 1592, 1487, 1342, 1210, 1155, 970, 757 cm-1 ; HR-MS (ESI): m/z calcd. for C16H15N2O5S 347.0702, found 347.0714 [M+H]+ …..https://chemistry-europe.onlinelibrary.wiley.com/action/downloadSupplement?doi=10.1002%2Fslct.202003553&file=slct202003553-sup-0001-misc_information.pdf
Patent
WO 2021020481
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2021020481&tab=FULLTEXT&_cid=P11-KKXGLE-06477-1The following formula (1)
[Chemical 1]
Iguratimod (chemical name: N- [3- (formylamino) -4-oxo-6-phenoxy-4H-chromen-7-yl] methanesulfonamide), which is indicated by, has excellent anti-inflammatory and antipyretic and analgesic effects. It is a very useful compound as a therapeutic agent exhibiting anti-arthritis and anti-allergic effects (see Patent Document 1).
A plurality of synthetic routes are known for the method for producing iguratimod and its derivatives (hereinafter, may be referred to as iguratimod derivatives as a term meaning both iguratimod and its derivatives), and all of them use intermediates of iguratimod derivatives. This is a stepwise manufacturing method. The present inventors have studied the stepwise production of intermediates of iguratimod derivatives of formulas (II) to (XI) by the following synthetic route to synthesize the iguratimod derivatives represented by formula (XII). doing.
[Chemical formula 2]R 1 = hydroxyl protecting group, R 2 = amino protecting group, Ar = aromatic ring group which may have a substituent, X 2 = halogen atom.[Chemical 3]
Ms = methylsulfonyl group[Chemical 4]
Production Example 1
(Production of igratimodo: Patent Documents 3 and 4)
N, N-dimethylformamide 150 mL, N, N-dimethylformamide dimethyl acetal 40.9 g (N, N-dimethylformamide dimethyl acetal ) in a 1000 mL four-necked flask equipped with two stirring blades having a diameter of 10 cm ( 343 mmol) was added, and the mixture was cooled to 10 ° C. with stirring. 8.2 g (137 mmol) of glacial acetic acid and 50.0 g (137 mmol) of formylaminomethyl (2-hydroxy-4-methylsulfonylamino-5-phenoxyphenyl) ketone were sequentially added thereto, and the temperature was raised to 20 ° C. The reaction was carried out at the same temperature for 5 hours. 250 mL of methylene chloride was added to the reaction suspension, and 500 mL of water was added dropwise to the obtained solution. After adjusting the pH to 5 with a 10% aqueous hydrochloric acid solution, the mixture was stirred at 20 ° C. for 1 hour. The obtained precipitated crystals were separated, washed successively with 50 mL of methylene chloride, 50 mL of water and 50 mL of ethanol, and then dried at 50 ° C. for 12 hours. Next, the obtained crystals were dissolved in a mixed solvent of 7.7 g (137 mmol) of potassium hydroxide, 750 mL of water and 750 mL of acetone, and then neutralized with 2N hydrochloric acid water, and the obtained precipitated crystals were separated. Then, after washing with 50 mL of water, it was dried at 50 ° C. for 12 hours to obtain 42.8 g of iguratimod (iguratimod purity: 99.72%, N-methyl compound: 0.23%).
References
- ^ Jump up to:a b c d e f Tanaka K, Yamaguchi T, Hara M (May 2015). “Iguratimod for the treatment of rheumatoid arthritis in Japan”. Expert Review of Clinical Immunology. 11 (5): 565–73. doi:10.1586/1744666X.2015.1027151. PMID 25797025. S2CID 25134255.
- ^ Inaba T, Tanaka K, Takeno R, Nagaki H, Yoshida C, Takano S (January 2000). “Synthesis and antiinflammatory activity of 7-methanesulfonylamino-6-phenoxychromones. Antiarthritic effect of the 3-formylamino compound (T-614) in chronic inflammatory disease models”. Chemical & Pharmaceutical Bulletin. 48 (1): 131–9. doi:10.1248/cpb.48.131. PMID 10705489.
- ^ Bronson J, Dhar M, Ewing W, Lonberg N (2012). “Chapter Thirty-One – To Market, To Market—2011”. Annual Reports in Medicinal Chemistry. 47: 499–569. doi:10.1016/B978-0-12-396492-2.00031-X.
- ^ “Iguratimod – Simcere”. AdisInsight. Retrieved 27 May 2018.
- ^ “Iguratimod – Toyama Chemical”. AdisInsight. Retrieved 27 May 2018.
| Clinical data | |
|---|---|
| Trade names | Careram; Kolbet |
| Other names | T-614 |
| ATC code | None |
| Identifiers | |
| IUPAC name[show] | |
| CAS Number | 123663-49-0 |
| PubChem CID | 124246 |
| ChemSpider | 110694 |
| UNII | 4IHY34Y2NV |
| ChEMBL | ChEMBL2107455 |
| CompTox Dashboard (EPA) | DTXSID0048971 |
| ECHA InfoCard | 100.236.037 |
| Chemical and physical data | |
| Formula | C17H14N2O6S |
| Molar mass | 374.37 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| SMILES[hide]O=S(=O)(Nc3c(Oc1ccccc1)cc2c(O/C=C(\C2=O)NC=O)c3)C | |
| InChI[hide]InChI=1S/C17H14N2O6S/c1-26(22,23)19-13-8-15-12(17(21)14(9-24-15)18-10-20)7-16(13)25-11-5-3-2-4-6-11/h2-10,19H,1H3,(H,18,20)Key:ANMATWQYLIFGOK-UHFFFAOYSA-N |
////////////IGURATIMOD, UNII-4IHY34Y2NV , игуратимод , إيغوراتيمود , 艾拉莫德 , T-614, T 614, Kolbet, Careram, Rheumatoid arthritis, JAPAN 2012, CHINA 2011
CS(=O)(=O)NC1=C(C=C2C(=C1)OC=C(C2=O)NC=O)OC3=CC=CC=C3
RIDINILAZOLE

RIDINILAZOLE
SMT19969
- Molecular FormulaC24H16N6
- Average mass388.424 Da
- ридинилазол [Russian] [INN]ريدينيلازول [Arabic] [INN]利地利唑 [Chinese] [INN]
- リジニラゾール;
10075
2,2′-Di(4-pyridinyl)-3H,3’H-5,5′-bibenzimidazole
308362-25-6[RN]6,6′-Bi-1H-benzimidazole, 2,2′-di-4-pyridinyl-
Summit Therapeutics (formerly Summit Corp ) is developing ridinilazole the lead compound from oral narrow-spectrum, GI-restricted antibiotics, which also include SMT-21829, for the treatment of Clostridium difficile infection and prevention of recurrent disease.
Ridinilazole (previously known as SMT19969) is an investigational small molecule antibiotic being evaluated for oral administration to treat Clostridioides difficile infection (CDI). In vitro, it is bactericidal against C. difficile and suppresses bacterial toxin production; the mechanism of action is thought to involve inhibition of cell division.[1] It has properties which are desirable for the treatment of CDI, namely that it is a narrow-spectrum antibiotic which exhibits activity against C. difficile while having little impact on other normal intestinal flora and that it is only minimally absorbed systemically after oral administration.[2] At the time ridinilazole was developed, there were only three antibiotics in use for treating CDI: vancomycin, fidaxomicin, and metronidazole.[1][2] The recurrence rate of CDI is high, which has spurred research into other treatment options with the aim to reduce the rate of recurrence.[3][4]
As of 2019, two phase II trials have been completed and two phase III trials comparing ridinilazole to vancomycin for CDI are expected to be completed in September 2021.[2][5][6] Ridinilazole was designated as a Qualified Infectious Disease Product (QIDP) and was granted Fast Track status by the U.S. FDA.[2] Fast Track status is reserved for drugs designed to treat diseases where there is currently a gap in the treatment, or a complete lack thereof.[7] The QIDP designation adds five more years of exclusivity for ridinazole upon approval.[8]


PATENT
WO-2021009514
Process for preparing ridinilazole useful for treating Clostridium difficile infection. Also claimed is the crystalline form of a compound.
The present invention relates to processes for the preparation of 2,2′-di(pyridin-4-yl)-1/-/,T/-/-5,5′-bibenzo[d]imidazole (which may also be known as 5,5’-bis[2-(4-pyridinyl)-1/-/-benzimidazole], 2,2′-bis(4-pyridyl)-3/-/,3’/-/-5,5′-bibenzimidazole or 2-pyridin-4-yl-6-(2-pyridin-4-yl-3/-/-benzimidazol-5-yl)-1/-/-benzimidazole), referenced herein by the INN name ridinilazole, and pharmaceutically acceptable derivatives, salts, hydrates, solvates, complexes, bioisosteres, metabolites or prodrugs thereof. The invention also relates to various crystalline forms of ridinilazole, to processes for their preparation and to related pharmaceutical preparations and uses thereof (including their medical use and their use in the efficient large-scale synthesis of ridinilazole).
WO2010/063996 describes various benzimidazoles, including ridinilazole, and their use as antibacterials (including in the treatment of CDAD).
WO 2011/151621 describes various benzimidazoles and their use as antibacterials
(including in the treatment of CDAD).
W02007056330, W02003105846 and W02002060879 disclose various 2-amino benzimidazoles as antibacterial agents.
W02007148093 discloses various 2-amino benzothiazoles as antibacterial agents.
W02006076009, W02004041209 and Bowser et at. (Bioorg. Med. Chem. Lett., 2007, 17, 5652-5655) disclose various substituted benzimidazole compounds useful as anti-infectives that decrease resistance, virulence, or growth of microbes. The compounds are said not to exhibit intrinsic antimicrobial activity in vitro.
US 5,824,698 discloses various dibenzimidazoles as broad-spectrum antibiotics, disclosing activity against both Gram-negative and Gram-positive bacteria, including Staphylococcus spp.and Enterococcus spp. However, this document does not disclose activity against anaerobic spore-forming bacteria and in particular does not disclose activity against any Clostridioides spp. (including C. difficile).
US 2007/0112048 A1 discloses various bi- and triarylimidazolidines and bi- and
triarylamidines as broad-spectrum antibiotics, disclosing activity against both Gram negative and Gram-positive bacteria, including Staphylococcus spp., Enterococcus spp. and Clostridioides spp. However, this document does not disclose compounds of formula (I) as described herein.
Chaudhuri et al. (2007) J.Org. Chem. 72, 1912-1923 describe various bis-2-(pyridyl)-1 H-benzimidazoles (including compounds of formula I as described herein) as DNA binding agents. This document is silent as to potential antibacterial activity.
Singh et al. (2000) Synthesis 10: 1380-1390 describe a condensation reaction for producing 2,2′-di(pyridin-4-yl)-1/-/,T/-/-5,5′-bibenzo[d]imidazole using 4-pyridine
carboxaldehyde, FeCI3, 02, in DMF at 120°C.
Bhattacharya and Chaudhuri (2007) Chemistry – An Asian Journal 2: 648-655 describe a condensation reaction for producing 2,2′-di(pyridin-4-yl)-1/-/,T/-/-5,5′-bibenzo[d]imidazole using 4-pyridine carboxaldehyde and nitrobenzene at 120°C.
WO2019/068383 describes the synthesis of ridinilazole by metal-ion catalyzed coupling of 3,4,3’,4’-tetraaminobiphenyl with 4-pyridinecarboxaldehyde in the presence of oxygen, followed by the addition of a complexing agent.
PATENT
WO2010063996
claiming antibacterial compounds. Bicyclic heteroaromatic compounds, particularly bi-benzimidazole derivatives.
WO2007056330, WO2003105846 and WO2002060879 disclose various 2-amino benzimidazoles as antibacterial agents.
WO2007148093 discloses various 2-amino benzothiazoles as antibacterial agents.
WO2006076009, WO2004041209 and Bowser et al. (Bioorg. Med. Chem. Lett., 2007, 17, 5652-5655) disclose various substituted benzimidazole compounds useful as anti-infectives that decrease resistance, virulence, or growth of microbes. The compounds are said not to exhibit intrinsic antimicrobial activity in vitro.
US 5,824,698 discloses various dibenzimidazoles as broad-spectrum antibiotics, disclosing activity against both Gram-negative and Gram-positive bacteria, including Staphylococcus spp.and Enterococcus spp. However, this document does not disclose activity against anaerobic spore-forming bacteria and in particular does not disclose activity against any Clostridium spp. (including C. difficile).
US 2007/0112048 A1 discloses various bi- and triarylimidazolidines and bi- and triarylamidines as broad-spectrum antibiotics, disclosing activity against both Gram-negative and Gram-positive bacteria, including Staphylococcus spp., Enterococcus spp.
and Clostridium spp. However, this document does not disclose compounds of general formula (I) as described herein.
Chaudhuri et al. (J.Org. Chem., 2007, 72, 1912-1923) describe various bis-2-(pyridyl)-1 H-benzimidazoles (including compounds of formula I as described herein) as DNA binding agents. This document is silent as to potential antibacterial activity.
PATENT
Product PATENT, WO2010063996 ,
protection in the EP until 2029 and expire in the US in December 2029.
PAPER
https://www.frontiersin.org/articles/10.3389/fmicb.2018.01206/full
PAPER
Synthesis (2000), (10), 1380-1390.
https://www.thieme-connect.de/products/ejournals/abstract/10.1055/s-2000-7111
PAPERT
Chemistry – An Asian Journal (2007), 2(5), 648-655.
https://onlinelibrary.wiley.com/doi/abs/10.1002/asia.200700014
Studies of double‐stranded‐DNA binding have been performed with three isomeric bis(2‐(n‐pyridyl)‐1H‐benzimidazole)s (n=2, 3, 4). Like the well‐known Hoechst 33258, which is a bisbenzimidazole compound, these three isomers bind to the minor groove of duplex DNA. DNA binding by the three isomers was investigated in the presence of the divalent metal ions Mg2+, Co2+, Ni2+, Cu2+, and Zn2+. Ligand–DNA interactions were probed with fluorescence and circular dichroism spectroscopy. These studies revealed that the binding of the 2‐pyridyl derivative to DNA is dramatically reduced in the presence of Co2+, Ni2+, and Cu2+ ions and is abolished completely at a ligand/metal‐cation ratio of 1:1. Control experiments done with the isomeric 3‐ and 4‐pyridyl derivatives showed that their binding to DNA is unaffected by the aforementioned transition‐metal ions. The ability of 2‐(2‐pyridyl)benzimidazole to chelate metal ions and the conformational changes of the ligand associated with ion chelation probably led to such unusual binding results for the ortho isomer. The addition of ethylenediaminetetraacetic acid (EDTA) reversed the effects completely.
PAPER
Journal of Organic Chemistry (2007), 72(6), 1912-1923.
https://pubs.acs.org/doi/10.1021/jo0619433

Three symmetrical positional isomers of bis-2-(n-pyridyl)-1H-benzimidazoles (n = 2, 3, 4) were synthesized and DNA binding studies were performed with these isomeric derivatives. Like bisbenzimidazole compound Hoechst 33258, these molecules also demonstrate AT-specific DNA binding. The binding affinities of 3-pyridine (m-pyben) and 4-pyridine (p-pyben) derivatized bisbenzimidazoles to double-stranded DNA were significantly higher compared to 2–pyridine derivatized benzimidazole o-pyben. This has been established by combined experimental results of isothermal fluorescence titration, circular dichroism, and thermal denaturation of DNA. To rationalize the origin of their differential binding characteristics with double-stranded DNA, computational structural analyses of the uncomplexed ligands were performed using ab initio/Density Functional Theory. The molecular conformations of the symmetric head-to-head bisbenzimidazoles have been computed. The existence of intramolecular hydrogen bonding was established in o-pyben, which confers a conformational rigidity to the molecule about the bond connecting the pyridine and benzimidazole units. This might cause reduction in its binding affinity to double-stranded DNA compared to its para and meta counterparts. Additionally, the predicted stable conformations for p-, m-, and o-pyben at the B3LYP/6-31G* and RHF/6-31G* levels were further supported by experimental pKa determination. The results provide important information on the molecular recognition process of such symmetric head to head bisbenzimidazoles toward duplex DNA.
Patent
US 8975416
PATENT
WO 2019068383
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2019068383
Clostridium difficile infection (CDI) is the leading cause of infectious healthcare-associated diarrhoea. CDI remains a challenge to treat clinically, because of a limited number of antibiotics available and unacceptably high recurrence rates. Because of this, there has been significant demand for creating innovative therapeutics, which has resulted in the development of several novel antibiotics.
Ridinilazole (SMT19969) is the INN name of 5,5’bis[2-(4-pyridinyl)-lH-benzimidazole], which is a promising non-absorbable small molecule antibiotic intended for oral use in the treatment of CDI. It has been shown to exhibit a prolonged post-antibiotic effect and treatment with ridinilazole has resulted in decreased toxin production. A phase 1 trial demonstrated that oral ridinilazole is well tolerated and specifically targets Clostridia whilst sparing other faecal bacteria.
Ridinilazole has the following chemical structure:
Bhattacharya & Chaudhuri (Chem. Asian J., 2007, No. 2, 648-655) report performing double-stranded DNA binding with three benzimidazole derivatives, including ridinilazole. The compounds have been prepared by dissolving the reactants in nitrobenzene, heating at 120°C for 8- 1 Oh and purifying the products by column chromatography over silica gel. The compounds were obtained in 65-70% yield. Singh et al., (Synthesis, 2000, No. 10, 1380-1390) describe a catalytic redox cycling approach based on Fe(III) and molecular oxygen as co-oxidant for providing access to benzimidazole and
imidazopyridine derivatives, such as ridinilazole. The reaction is performed at high temperatures of 120°C and the product is isolated in 91% yield by using silica flash chromatography.
Both processes are not optimal, for example in terms of yield, ease of handling and scalability. Thus, there is a need in the art for an efficient and scalable preparation of ridinilazole, which overcomes the problems of the prior art processes.
Example 1 : Preparation of crude ridinilazole free base
A solution of 3,4,3′,4′-tetraaminobiphenyl (3.28 g, 15.3 mmol) and isonicotinaldehyde (3.21 g, 30.0 mmol) in DMF (40 mL) was stirred at 23 °C for one hour. Then anhydrous ferric chloride (146 mg, 0.90 mmol), water (0.10 mL, 5.4 mmol) and additional DMF (2 mL) were added and fresh air was bubbled into the solution during vigorous stirring for 5 hours at room temperature. Next, water (80 mL) and EDTA (0.29 g) were added resulting in a brownish suspension, which was stirred overnight. The product was isolated by filtration, washed with water, and dried in a desiccator in vacuo as a brown powder (5.56 g; 95%). The addition of EDTA had held iron in solution and the crude ridinilazole contained significantly lower amounts of iron than comparative example 1.
Example 12: Formation of essentially pure ridinilazole free base
To a suspension von ridinilazole tritosylate (1 10 mg, 0.12 mmol) in water (35 mL) featuring a pH value of about 4.5 stirring at 70 °C sodium bicarbonate (580 mg, 6.9 mmol) were added and caused a change of color from orange to slightly tan. The mixture, now at a pH of about 8.5, was cooled down to room temperature and the solids were separated by filtration, washed with water (1 ML) and dried in vacuo providing 40 mg (85%) essentially pure ridinilazole as a brownish powder.
Spectroscopic analysis:
¾ NMR (DMSO-de, 300 MHz): δ 7.55 (d, J = 8.4 Hz, 2H), 7.70 (d, J = 8.4 Hz, 2H), 7.88 (s, 2H), 8.13 (d, J = 5.8 Hz, 4H), 8.72 (d, J = 5.8 Hz, 4H) ppm.
13C NMR (DMSO-d6, 75 MHz): δ 1 13.4 (2C), 1 16.4 (2C), 120.4 (4C), 121.8 (2C), 135.7 (2C), 138.7 (2C), 140.7 (2C), 141.4 (2C), 150.3 (4C), 151.1 (2C) ppm.
IR (neat): v 3033 (w), 1604 (s), 1429 (m), 1309 (m), 1217 (m), 1 1 15 (w), 998 (m), 964 (m), 824 (m), 791 (s), 690 (s), 502 (s) cm .
UV-Vis (MeOH): 257, 341 nm.
The sharp peaks in the ¾ NMR indicated that iron had been efficiently removed.
Comparative example 1 : Preparation of ridinilazole
A solution of 3,4,3′,4′-tetraaminobiphenyl (0.69 g, 3.2 mmol) and isonicotinaldehyde (0.64 g, 6.0 mmol) in DMF (20 mL) was stirred at 80°C for one hour. Then ferric chloride hexahydrate (49 mg, 0.18 mmol), water (0.10 mL, 5.4 mmol) and additional DMF (2 mL) were added and fresh air was bubbled into the solution during vigorous stirring for 10 hours at 120 °C. After cooling to room temperature water (50 mL) and the mixture was stirred for one hour. A black crude product was isolated by filtration and comprised ridinilazole and iron.
References
- ^ Jump up to:a b Cho JC, Crotty MP, Pardo J (March 2019). “Clostridium difficile infection”. Annals of Gastroenterology. 32 (2): 134–140. doi:10.20524/aog.2018.0336. PMC 6394264. PMID 30837785.
- ^ Jump up to:a b c d Carlson TJ, Endres BT, Bassères E, Gonzales-Luna AJ, Garey KW (April 2019). “Ridinilazole for the treatment of Clostridioides difficile infection”. Expert Opinion on Investigational Drugs. 28 (4): 303–310. doi:10.1080/13543784.2019.1582640. PMID 30767587.
- ^ Bassères E, Endres BT, Dotson KM, Alam MJ, Garey KW (January 2017). “Novel antibiotics in development to treat Clostridium difficile infection”. Current Opinion in Gastroenterology. 33 (1): 1–7. doi:10.1097/MOG.0000000000000332. PMID 28134686.
These tables highlight the increased drug development directed towards CDI due to the rise in prevalence of infections and to attempt to reduce the number of recurrent infections.
- ^ Vickers RJ, Tillotson G, Goldstein EJ, Citron DM, Garey KW, Wilcox MH (August 2016). “Ridinilazole: a novel therapy for Clostridium difficile infection”. International Journal of Antimicrobial Agents. 48 (2): 137–43. doi:10.1016/j.ijantimicag.2016.04.026. PMID 27283730.
there exists a significant unmet and increasing medical need for new therapies to treat CDI, specifically those that can reduce the rate of disease recurrence.
- ^ Clinical trial number NCT03595553 for “Ri-CoDIFy 1: Comparison of Ridinilazole Versus Vancomycin Treatment for Clostridium Difficile Infection” at ClinicalTrials.gov
- ^ Clinical trial number NCT03595566 for “Ri-CoDIFy 2: To Compare Ridinilazole Versus Vancomycin Treatment for Clostridium Difficile Infection” at ClinicalTrials.gov
- ^ “Fast Track”. U.S. Food and Drug Administration. 2018-11-03.
- ^ “”HHS spurs new antibiotic development for biodefense and common infections””. Public Health Emergency. U.S. Department of Health and Human Services. Retrieved 2020-12-04.
| Clinical data | |
|---|---|
| Other names | SMT19969 |
| ATC code | None |
| Identifiers | |
| IUPAC name[show] | |
| CAS Number | 308362-25-6 |
| PubChem CID | 16659285 |
| ChemSpider | 17592423 |
| UNII | 06DX01190R |
| KEGG | D11958 |
| Chemical and physical data | |
| Formula | C24H16N6 |
| Molar mass | 388.42 g/mol |
| 3D model (JSmol) | Interactive image |
| SMILES[hide]c6cc(c5nc4ccc(c3ccc2nc(c1ccncc1)[nH]c2c3)cc4[nH]5)ccn6 |
/////////RIDINILAZOLE, SMT19969, SMT 19969, ридинилазол , ريدينيلازول , 利地利唑 , リジニラゾール , Qualified Infectious Disease Product, QIDP, Fast Track , PHASE 3, Clostridioides difficile infection ,
OI 338

OI 338
OI338GT (NN1953)
NNC0123-0000-0338
Insulin oral (NN 1953); Insulin-338-GIPET-I; LAI 338; NN 1438; NN-1953; NNC-0123-0000-0338; NNC0123-0338; OI-338GT; Oral insulin 338 C10
- OriginatorNovo Nordisk
- ClassAntihyperglycaemics; Insulins; Pancreatic hormones
- Mechanism of ActionOrnithine decarboxylase stimulants; Phosphokinase stimulants; Protein tyrosine kinase stimulants
- Phase IIType 1 diabetes mellitus; Type 2 diabetes mellitus
- 28 Jul 2018No recent reports of development identified for phase-I development in Type-1 diabetes mellitus in Germany (SC, Injection)
- 28 Jul 2018No recent reports of development identified for phase-I development in Type-2-diabetes-mellitus in Denmark (SC, Injection)
- 11 Sep 2017Efficacy and adverse events data from a phase II trial in Type-2 diabetes mellitus presented at the 53rd Annual Meeting of the European Association for the Study of Diabetes (EASD-2017)
OI-338GT is a long-acting oral basal insulin analogue which had been in phase II clinical trials at Novo Nordisk for the treatment of patients with type 2 and type 1 diabetes. In 2016, the company discontinued the development of the product as the emergent product profile and required overall investments were not commercially viable in the increasingly challenging payer environment.
PAPERJ. Med. Chem. 2021, 64, 1, 616–628
Publication Date:December 28, 2020
https://doi.org/10.1021/acs.jmedchem.0c01576https://pubs.acs.org/doi/10.1021/acs.jmedchem.0c01576

Recently, the first basal oral insulin (OI338) was shown to provide similar treatment outcomes to insulin glargine in a phase 2a clinical trial. Here, we report the engineering of a novel class of basal oral insulin analogues of which OI338, 10, in this publication, was successfully tested in the phase 2a clinical trial. We found that the introduction of two insulin substitutions, A14E and B25H, was needed to provide increased stability toward proteolysis. Ultralong pharmacokinetic profiles were obtained by attaching an albumin-binding side chain derived from octadecanedioic (C18) or icosanedioic acid (C20) to the lysine in position B29. Crucial for obtaining the ultralong PK profile was also a significant reduction of insulin receptor affinity. Oral bioavailability in dogs indicated that C18-based analogues were superior to C20-based analogues. These studies led to the identification of the two clinical candidates OI338 and OI320 (10 and 24, respectively).
Oral insulin 338 (I338) is a long-acting, basal insulin analogue formulated in a tablet with the absorption-enhancer sodium caprate. We investigated the efficacy and safety of I338 versus subcutaneous insulin glargine (IGlar) in patients with type 2 diabetes. METHODS: This was a phase 2, 8-week, randomised, double-blind, double-dummy, active-controlled, parallel trial completed at two research institutes in Germany. Insulin-naive adult patients with type 2 diabetes, inadequately controlled on metformin monotherapy or combined with other oral antidiabetic drugs (HbA1c 7·0-10·0%; BMI 25·0-40·0 kg/m(2)), were randomly assigned (1:1) to receive once-daily I338 plus subcutaneous placebo (I338 group) or once-daily IGlar plus oral placebo (IGlar group). Randomisation occurred by interactive web response system stratified by baseline treatment with oral antidiabetic drugs. Patients and investigators were masked to treatment assignment. Weekly insulin dose titration aimed to achieve a self-measured fasting plasma glucose (FPG) concentration of 4·4-7·0 mmol/L. The recommended daily starting doses were 2700 nmol I338 or 10 U IGlar, and maximum allowed doses throughout the trial were 16 200 nmol I338 or 60 U IGlar. The primary endpoint was treatment difference in FPG concentration at 8 weeks for all randomly assigned patients receiving at least one dose of trial product (ie, the full analysis set). The trial has been completed and is registered at ClinicalTrials.gov, number NCT02470039. FINDINGS: Between June 1, 2015, and Oct 19, 2015, 82 patients were screened for eligibility and 50 patients were randomly assigned to the I338 group (n=25) or the IGlar group (n=25). Mean FPG concentration at baseline was 9·7 (SD 2·8) in the I338 group and 9·1 (1·7) in the IGlar group. Least square mean FPG concentration at 8 weeks was 7·1 mmol/L (95% CI 6·4-7·8) in the I338 group and 6·8 mmol/L (6·5-7·1) in the IGlar group, with no significant treatment difference (0·3 mmol/L [-0·5 to 1·1]; p=0·46). I338 and IGlar were well tolerated by patients. Adverse events were reported in 15 (60%) patients in the I338 group and 17 (68%) patients in the IGlar group. The most common adverse events were diarrhoea (three [12%] patients in each group) and nasopharyngitis (five [20%] in the I338 group and two [8%] in the IGlar group). Most adverse events were graded mild (47 of 68 events), and no severe adverse events were reported. One patient in the IGlar group had a treatment-emergent serious adverse event (urogenital haemorrhage of moderate intensity, assessed by the investigator as unlikely to be related to treatment; the patient recovered). Incidence of hypoglycaemia was low in both groups (n=7 events in the I338 group; n=11 in the IGlar group), with no severe episodes. INTERPRETATION: I338 can safely improve glycaemic control in insulin-naive patients with type 2 diabetes with no evidence of a difference compared with insulin glargine, a widely used subcutaneously administered basal insulin. Further development of this particular oral insulin project was discontinued because I338 doses were high and, therefore, production of the required quantities of I338 for wide public use was deemed not commercially viable. Improvement of technologies involved in the product’s development is the focus of ongoing research. FUNDING: Novo Nordisk…..Halberg, I. B.; Lyby, K.; Wassermann, K.; Heise, T.; Zijlstra, E.; Plum-Mörschel, L. Efficacy and safety of oral basal insulin versus subcutaneous insulin glargine in type 2 diabetes: a randomised, double-blind, phase 2 trial. Lancet Diabetes Endocrinol. 2019, 7, 179– 188, DOI: 10.1016/s2213-8587(18)30372-3
ral insulin 338 is a novel tablet formulation of a long-acting basal insulin. This randomised, open-label, four-period crossover trial investigated the effect of timing of food intake on the single-dose pharmacokinetic properties of oral insulin 338. Methods: After an overnight fast, 44 healthy males received single fixed doses of oral insulin 338 administered 0, 30, 60 or 360 min before consuming a standardised meal (500 kcal, 57 energy percent [E%] carbohydrate, 13 E% fat, 30 E% protein). Blood samples for pharmacokinetic assessment were taken up to 288 h post-dose. Results: Total exposure (area under the concn.-time curve from time zero to infinity [AUCIns338,0-∞]) and max. concn. (Cmax,Ins338) of insulin 338 were both significantly lower for 0 vs. 360 min post-dose fasting (ratio [95% confidence interval (CI)]: 0.36 [0.26-0.49], p < 0.001, and 0.35 [0.25-0.49], p < 0.001, resp.). There were no significant differences in AUCIns338,0-∞ and Cmax,Ins338 for 30 or 60 vs. 360 min post-dose fasting (ratio [95% CI] 30 vs. 360 min: 0.85 [0.61-1.21], p = 0.36, and 0.86 [0.59-1.26], p = 0.42; ratio [95% CI] 60 vs. 360 min: 0.96 [0.72-1.28], p = 0.77, and 0.99 [0.75-1.31], p = 0.95). The mean half-life was ∼ 55 h independent of the post-dose fasting period. Oral insulin 338 was well-tolerated with no safety issues identified during the trial. Conclusions: Oral insulin 338 pharmacokinetics are not affected by food intake from 30 min after dosing, implying that patients with diabetes mellitus do not need to wait more than 30 min after a morning dose of oral insulin 338 before having their breakfast. This is considered important for convenience and treatment compliance. ClinicalTrials.gov identifier: NCT02304627./……Halberg, I. B.; Lyby, K.; Wassermann, K.; Heise, T.; Plum-Mörschel, L.; Zijlstra, E. The effect of food intake on the pharmacokinetics of oral basal insulin: A randomised crossover trial in healthy male subjects. Clin. Pharmacokinet. 2019, 58, 1497– 1504, DOI: 10.1007/s40262-019-00772-2
///////////////OI 338, OI338GT, NN1953, NNC0123-0000-0338, Insulin oral (NN 1953), Insulin-338-GIPET-I, LAI 338, NN 1438, NN-1953, NNC-0123-0000-0338, NNC0123-0338, OI-338GT, Oral insulin 338 C10
Devimistat

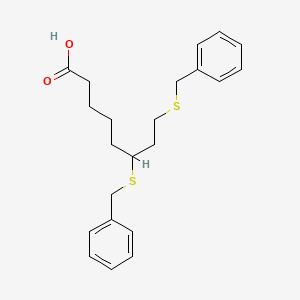
Devimistat
CPI-613
| Molecular Weight | 388.59 |
|---|---|
| Formula | C₂₂H₂₈O₂S₂ |
| CAS No. | 95809-78-2 |
| SMILES | O=C(O)CCCCC(SCC1=CC=CC=C1)CCSCC2=CC=CC=C2 |
phase III, hematological cancer
6,8-Bis(benzylsulfanyl)octanoic acid
Octanoic acid, 6,8-bis[(phenylMethyl)thio]-
Octanoic acid, 6,8-bis((phenylmethyl)thio)-
Rafael Pharmaceuticals (formerly Cornerstone Pharmaceuticals), a subsidiary of Rafael Holdings, is developing devimistat, the lead candidate from a program of thioctans and their derivatives that act as pyruvate dehydrogenase and alpha-ketoglutarate inhibitors and stimulators of pyruvate dehydrogenase kinase (PDK), using the company’s proprietary Altered Energy Metabolism Directed (AEMD) platform, for the iv treatment of hematological cancer [phase III, January 2021].
Devimistat (INN; development code CPI-613) is an experimental anti-mitochondrial drug being developed by Rafael Pharmaceuticals.[1] It is being studied for the treatment of patients with metastatic pancreatic cancer and relapsed or refractory acute myeloid leukemia (AML).
Devimistat’s mechanism of action differs from other drugs, operating on the tricarboxylic acid cycle and inhibiting enzymes involved with cancer cell energy metabolism. A lipoic acid derivative different from standard cytotoxic chemotherapy, devimistat is currently being studied in combination with modified FOLFIRINOX to treat various solid tumors and heme malignancies.
Regulation
The U.S. Food and Drug Administration (FDA) has designated devimistat as an orphan drug for the treatment of pancreatic cancer, AML, myelodysplastic syndromes (MDS), peripheral T-cell lymphoma, and Burkitt’s lymphoma, and given approval to initiate clinical trials in pancreatic cancer and AML.
Clinical trials
Clinical trials of the drug are underway including a Phase III open-label clinical trial[2] to evaluate efficacy and safety of devimistat plus modified FOLFIRINOX (mFFX) versus FOLFIRINOX (FFX) in patients with metastatic adenocarcinoma of the pancreas.
Developed as part of Rafael’s proprietary Altered Metabolism Directed (AMD) drug platform, CPI-613® was discovered at Stony Brook University. CPI-613® is designed to target the mitochondrial tricarboxylic acid (TCA) cycle, an indispensable process essential to tumor cell multiplication and survival, selectively in cancer cells.
The attacks of CPI-613® on the TCA cycle also substantially increases the sensitivity of cancer cells to a diverse range of chemotherapeutic agents. This synergy allows for combinations of CPI-613® with lower doses of these generally toxic drugs to be highly effective with lower patient side effects. Combinations with CPI-613® represent a diverse range of potential opportunities to substantially improve patient benefit in many different cancers.
The U.S. Food and Drug Administration (FDA) has given Rafael approval to initiate pivotal clinical trials in pancreatic cancer and acute myeloid leukemia (AML), and has designated CPI-613® as an orphan drug for the treatment of pancreatic cancer, AML, Myelodysplastic syndromes (MDS), peripheral T-cell lymphoma and Burkitt’s lymphoma. The EMA has granted orphan drug designation to CPI-613® for pancreatic cancer and AML.
Learn more about recent developments involving CPI-613®: CPI-613® (devimistat) Fact Sheet
he FDA granted a Fast Track designation to devimistat for the treatment of patients with acute myeloid leukemia.
The FDA has granted a Fast Track designation to devimistat (CPI-613) for the treatment of patients with acute myeloid leukemia (AML), Rafael Pharmaceuticals, announced in a press release.1
“This designation underscores the pressing need to find new ways to combat this aggressive disease,” said Jorge Cortes, MD, director of the Georgia Cancer Center at Augusta University, and principal investigator on the phase 3 clinical trial, in a statement. “It brings hope not only to clinicians, but to patients who hear that they have been diagnosed.”
The first-in-class agent devimistat targets enzymes that are involved in cancer cell energy metabolism. This therapy substantially increases the sensitivity of cancer cells to a diverse range of chemotherapies, and this synergy allows for potential combinations that could be more effective with devimistat and lower doses of drugs that are generally toxic.
“Receiving Fast Track designation, especially during a pandemic that has created significant challenges for many trials across the globe, is a testament to the dedicated work of the Rafael team,” stated Sanjeev Luther, president and CEO of Rafael Pharmaceuticals, Inc.
Devimistat combinations appear promising with a diverse range of potential opportunities to improve benefit in patients with various cancer types. Two pivotal phase 3 clinical trials, including the AVENGER 500 study in pancreatic cancer (NCT03504423) and ARMADA 2000 for AML (NCT03504410), have been approved for initiation by the FDA.
The primary end point of the multicenter, open-label, randomized ARMADA 2000 study is complete response (CR), and secondary end points include overall survival and CR plus CR with partial hematologic recovery rate. To be eligible to enroll to the study, patients must be aged ≥50 years with a documented AML diagnosis that has relapsed from or became refractory to previous standard therapy. Patients must have an ECOG performance status of 0 to 2 and an expected survival longer than 3 months.
Five hundred patients are expected to be enrolled and randomized in the study. To enroll, patients could not have received prior radiotherapy or cytotoxic chemotherapy for their current AML. Those with active central nervous system involvement, active uncontrolled bleeding, history of other malignancy, or known hypersensitivity to study drugs are ineligible to enroll to the trial as well.
This study aims to determine the safety and efficacy of devimistat in combination with high-dose cytarabine and mitoxantrone in older patients with relapsed/refractory AML compared with high-dose cytarabine and mitoxantrone therapy alone. Other control groups include patients treated with mitoxantrone, etoposide, and cytarabine and the combination of fludarabine, cytarabine, and filgrastim. The addition of devimistat is expected to improve the CR rate in patients who are aged 50 years or older with relapsed/refractory AML.
In a prior phase 1 study of devimistat plus high-dose cytarabine and mitoxantrone in patients with relapsed/refractory AML, the addition of devimistat sensitized AML cells to chemotherapy treatment.2
The objective response rate was 50% including CRs in 26 of 62 evaluable patients. Median overall survival was 6.7 months. In patients above age 60, the CR or CR with incomplete hematologic recovery rate was 47% and the median survival was 6.9 months.
This designation for this experimental anti-mitochondrial agent follows news of another Fast Track designation granted to devimistat for the treatment of patients with metastatic pancreatic cancer in November 2020, as well as an Orphan Drug designation granted in October 2020 for the treatment of patients with soft tissue sarcoma.
References
1. Rafael Pharmaceuticals Receives FDA Fast Track Designation for CPI-613® (devimistat) for the treatment of acute myeloid leukemia (AML). News Release. Rafael Pharmaceuticals, Inc. December 15, 2020. Accessed December 15, 2020. https://bit.ly/34g6YsR
2. Pardee TS, Anderson RG, Pladna KM, et al. A Phase I Study of CPI-613 in Combination with High-Dose Cytarabine and Mitoxantrone for Relapsed or Refractory Acute Myeloid Leukemia. Clin Cancer Res. 2018;24(9):2060-2073. doi:10.1158/1078-0432.CCR-17-2282 P[APERJournal of the American Chemical Society (1954), 76, 4109-12.https://pubs.acs.org/doi/abs/10.1021/ja01645a016
PAPERJournal of the American Chemical Society (1955), 77, 416-19.https://pubs.acs.org/doi/abs/10.1021/ja01607a057PAPERJustus Liebigs Annalen der Chemie (1958), 614, 66-83.https://chemistry-europe.onlinelibrary.wiley.com/doi/abs/10.1002/jlac.19586140108PATENTWO 2009123597WO 2009110859WO 2010110771PATENTCN 111362848
PATENT
WO-2021011334
Deuterated derivatives of 6,8-bis(benzylsulfanyl)octanoic acid (CPI-613 or devimistat ) or its salts for treating cancer.
CPI-613 (6,8-bis(benzylsulfanyl)octanoic acid) is a first-in-class investigational small-molecule (lipoate analog), which targets the altered energy metabolism unique to many cancer cells. CPI-613 is currently being evaluated in two phase III clinical trials, and has been granted orphan drug designation for the treatment of pancreatic cancer, acute myeloid leukemia (AML), peripheral T-cell lymphoma (PTCL), Burkitt lymphoma and myelodysplastic syndromes (MDS).
[0004] One limitation to the clinical utility of CPI-613 is its very rapid metabolism. After IV dosing the half-life of 6,8-bis(benzylsulfanyl)octanoic acid is only about 1-2 hours (Pardee,
T.S. et al, Clin Cancer Res. 2014, 20, 5255-64). The short half-life limits the patient’s overall exposure to the drug and necessitates administration of relatively high doses. For safety reasons, CPI-613 is administered via a central venous catheter as an IV infusion over 30-120 minutes, with higher doses requiring longer infusion times.
The terms“6,8-bis(benzylsulfanyl)octanoic acid” and“ 6,8-bis-benzylthio-octanoic acid” refer to the compound known as CPI-613 or devimistat, having the chemical structure
PATENT
WO2020132397
claiming the use of CPI-613 in combination with an autophagy inhibitor eg chloroquine for treating eg cancers.
CPI-613 (6,8-bis-benzylthio-octanoic acid) is a first-in-class investigational small-molecule (lipoate analog), which targets the altered energy metabolism that is common to many cancer cells. CPI-613 has been evaluated in multiple phase I, I/II, and II clinical studies, and has been granted orphan drug designation for the treatment of pancreatic cancer, acute myeloid leukemia (AML), peripheral T-cell lymphoma (PTCL), Burkitt lymphoma and myelodysplastic syndromes (MDS).
PAPER
https://pubs.acs.org/doi/10.1021/op200091t
An Efficient, Economical Synthesis of the Novel Anti-tumor Agent CPI-613
Cite this: Org. Process Res. Dev. 2011, 15, 4, 855–857
Publication Date:May 2, 2011
https://doi.org/10.1021/op200091t

An efficient and practical synthesis of the novel anti-tumor compound 6,8-dithiobenzyl octanoic acid, CPI-613 (2), was developed and executed on a practical scale. CPI-613 can be made in a single vessel from (±)-lipoic acid (1) via reductive opening of the disulfide ring followed by benzylation of the sulfhydryls with benzyl bromide. CPI-613 was isolated by simple crystallization in high yield and purity. The process is scaleable and has been demonstrated at up to 100 kg.CPI-613 (2) was isolated [4.7 kg (90%)] with an HPLC purity of 99.8 area %. Mp 66–67 °C. IR: 3050, 1710, 1400, 668 cm–1. 1H NMR (400 MHz, CDCl3) δ 7.40–7.20 (m, 10 H), 3.80–3.60 (m, 4 H), 2.60–2.50 (m, 2 H), 2.44 (t, J = 8.7, 2 H), 2.23 (t, J = 8.1, 2 H) 2.03–1.30 (m, 8 H). Anal. Calc for C22H28O2S2: C, 68.00; H, 7.26; S, 16.50. Found: C, 67.99; H, 7.31; S, 16.37.

References
- ^ “CPI-613”. Rafael Pharmaceuticals.
- ^ Philip PA, Buyse ME, Alistar AT, Rocha Lima CM, Luther S, Pardee TS, Van Cutsem E (October 2019). “A Phase III open-label trial to evaluate efficacy and safety of CPI-613 plus modified FOLFIRINOX (mFFX) versus FOLFIRINOX (FFX) in patients with metastatic adenocarcinoma of the pancreas”. Future Oncology. 15 (28): 3189–3196. doi:10.2217/fon-2019-0209. PMC 6854438. PMID 31512497.
| Clinical data | |
|---|---|
| Other names | CPI-613 |
| Legal status | |
| Legal status | Investigational |
| Identifiers | |
| IUPAC name[show] | |
| CAS Number | 95809-78-2 |
| PubChem CID | 24770514 |
| DrugBank | 12109 |
| ChemSpider | 28189062 |
| UNII | E76113IR49 |
| ChEMBL | ChEMBL3186849 |
| CompTox Dashboard(EPA) | DTXSID70914807 |
| ECHA InfoCard | 100.231.125 |
| Chemical and physical data | |
| Formula | C22H28O2S2 |
| Molar mass | 388.58 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| SMILES[hide]C1=CC=C(C=C1)CSCCC(CCCCC(=O)O)SCC2=CC=CC=C2 |
//////////devimistat, CPI-613, CPI 613, phase 3, hematological cancer , Fast Track designation, ORPHAN DRUG,
Fosnetupitant

Fosnetupitant
- Molecular FormulaC31H35F6N4O5P
- Average mass688.598 Da
[4-[5-[[2-[3,5-bis(trifluoromethyl)phenyl]-2-methylpropanoyl]-methylamino]-4-(2-methylphenyl)pyridin-2-yl]-1-methylpiperazin-1-ium-1-yl]methyl hydrogen phosphate(4-{5-[{2-[3,5-Bis(trifluoromethyl)phenyl]-2-methylpropanoyl}(methyl)amino]-4-(2-methylphenyl)-2-pyridinyl}-1-methylpiperazin-1-ium-1-yl)methyl hydrogen phosphate07-PNET10146
CAS 1703748-89-3
HCL 1643757-72-5
FDA 2014 AND EMA 2015Фоснетупитант [Russian] [INN]فوسنيتوبيتانت [Arabic] [INN]磷奈匹坦 [Chinese] [INN]
- 07-PNET
In April 2018, the U.S. Food and Drug Administration (FDA) and the Swiss company Helsinn approved the intravenous formulation of AKYNZEO® (NEPA, a fixed antiemetic combination of fosnetupitant, 235mg, and palonosetron, 0.25mg) as an alternative treatment option for patients experiencing chemotherapy-induced nausea and vomiting. Fosnetupitant is the pro-drug form of netupitant. Generally, 25% to 30% of patients with a diagnosis of cancer receive chemotherapy as a treatment modality and 70% to 80% of these patients undergoing chemotherapy treatment may experience nausea and vomiting as major side effects. Considered one of the most distressing side effects of chemotherapy, nausea and vomiting has an immense impact on the quality of life of patients receiving certain antineoplastic therapies.
In April 2018, the U.S. Food and Drug Administration (FDA) and the Swiss company Helsinn approved the intravenous formulation of AKYNZEO® (NEPA, a fixed antiemetic combination of fosnetupitant, 235mg, and palonosetron, 0.25mg) as an alternative treatment option for patients experiencing chemotherapy-induced nausea and vomiting 3. Fosnetupitant is the pro-drug form of netupitant Label.
Generally, 25% to 30% of patients with a diagnosis of cancer receive chemotherapy as a treatment modality and 70% to 80% of these patients undergoing chemotherapy treatment may experience nausea and vomiting as major side effects. Considered one of the most distressing side effects of chemotherapy, nausea and vomiting has an immense impact on the quality of life of patients receiving certain antineoplastic therapies 1.
Fosnetupitant: Fosnetupitant is a selective antagonist of human substance P/neurokinin 1 (NK-1) receptors. Upon intravenous administration, Fosnetupitant is converted by phosphatases to its active form. It competitively binds to and blocks the activity of NK-1 receptors in the central nervous system, by inhibiting binding of substance P (SP) to NK-1 receptors. This prevents delayed emesis, which is associated with SP secretion. AKYNZEO is a combination of palonosetron, a serotonin-3 receptor antagonist, and Fosnetupitant (capsules for oral use) or Fosnetupitant (injections for intravenous use). AKYNZEO for injection is indicated in combination with dexamethasone in adults for the prevention of acute and delayed nausea and vomiting associated with initial and repeat courses of highly emetogenic cancer chemotherapy.
EMA
The chemical name of fosnetupitant chloride hydrochloride is 4-(5-(2-(3,5-bis(trifluoromethyl)phenyl)- N,2-dimethylpropanamido)-4-(o-tolyl)pyridin-2-yl)-1-methyl-1-((phosphonooxy)methyl)piperazin-1- ium chloride hydrochloride is corresponding to the molecular formula C31H37Cl2N4O5P. It has a relative molecular mass of 761.53 g/mol and the following structure:

The chemical structure of fosnetupitant chloride hydrochloride was elucidated by a combination of 1H and 13C NMR spectroscopy, infrared spectroscopy, mass spectrometry and elemental analysis. The active substance is achiral. The solid state properties of the active substance were measured by gravimetric vapour sorption and x-ray powder diffraction (XRPD). It is a white to off-white to yellowish, crystalline, hygroscopic solid. Three polymorphic forms have been identified following extensive screening, requiring isolation from different solvent mixtures. Fosnetupitant chloride hydrochloride is always isolated as form I following the commercial manufacturing process. Since it is dissolved and lyophilised during finished product manufacture, particle size and polymorphic form are not considered critical quality attributes (CQAs) of the active substance and are not included in the specification.
RX
AKYNZEO (300 mg netupitant/0.5 mg palonosetron) capsules are an oral combination product of netupitant, a substance P/neurokinin 1 (NK-1) receptor antagonist, and palonosetron hydrochloride, a serotonin-3 (5-HT3) receptor antagonist. Both netupitant and palonosetron hydrochloride are anti-nausea and anti-emetic agents.
Netupitant is chemically described: 2-[3,5-bis(trifluoromethyl)phenyl]-N, 2 dimethyl-N-[4-(2methylphenyl)-6-(4-methylpiperazin-1-yl)pyridin-3-yl] propanamide. The empirical formula is C30H32F6N4O, with a molecular weight of 578.61. Netupitant exists as a single isomer and has the following structural formula:
 |
Palonosetron hydrochloride is chemically described: (3aS)-2-[(S)-1-Azabicyclo [2.2.2]oct-3-yl]2,3,3a,4,5,6-hexahydro-1-oxo-1H-benz[de]isoquinoline hydrochloride. The empirical formula is C19H24N2O.HCl, with a molecular weight of 332.87. Palonosetron hydrochloride exists as a single isomer and has the following structural formula:
 |
Netupitant is white to off-white crystalline powder. It is freely soluble in toluene and acetone, soluble in isopropanol and ethanol, and very slightly soluble in water.
Palonosetron hydrochloride is a white to off-white crystalline powder. It is freely soluble in water, soluble in propylene glycol, and slightly soluble in ethanol and 2-propanol.
Each AKYNZEO capsule is composed of one white-caramel hard gelatin capsule which contains three tablets each containing 100 mg netupitant and one gelatin capsule containing 0.5 mg palonosetron (equivalent to 0.56 mg palonosetron hydrochloride). The inactive ingredients are butylated hydroxyanisole (BHA), croscarmellose sodium, gelatin, glycerin, magnesium stearate, microcrystalline cellulose, mono-and di-glycerides of capryl/capric acid, polyglyceryl dioleate, povidone K-30, purified water, red iron oxide, silicon dioxide, sodium stearyl fumarate, sorbitol, sucrose fatty acid esters, titanium dioxide and yellow iron oxide. It may contain traces of medium-chain triglycerides, lecithin, and denatured ethanol.
AKYNZEO (235 mg fosnetupitant/0.25 mg palonosetron) for injection is a combination product of fosnetupitant, a prodrug of netupitant, which is a substance P/neurokinin 1 (NK-1) receptor antagonist, and palonosetron hydrochloride, a serotonin-3 (5-HT3) receptor antagonist.
Fosnetupitant chloride hydrochloride is chemically described as 2-(3,5-bistrifluoromethylphenyl)-N-methyl-N-[6-(4-methyl-4-O-methylene-phosphatepiperazinium-1-yl)4-o-tolyl-pyridin-3-yl]-isobutyramide chloride hydrochloride. The empirical formula is C31H36F6N4O5P•Cl•HCl, with a molecular weight of 761.53. Fosnetupitant chloride hydrochloride exists as a single isomer and has the following structural formula:
 |
Fosnetupitant chloride hydrochloride is white to off-white to yellowish solid or powder. Its solubility is pH dependent: at acidic pH (pH 2), its solubility is 1.4 mg/mL; at basic pH (pH 10), its solubility is 11.5 mg/mL.
Palonosetron hydrochloride is described above in this section.
AKYNZEO for injection is available for intravenous infusion, and is supplied as a sterile lyophilized powder in a single-dose vial. Each vial contains 235 mg of fosnetupitant (equivalent to 260 mg fosnetupitant chloride hydrochloride) and 0.25 mg of palonosetron (equivalent to 0.28 mg of palonosetron hydrochloride). The inactive ingredients are edetate disodium (6.4 mg), mannitol (760 mg), sodium hydroxide and/or hydrochloric acid (for pH adjustment).
PATENT
WO 2013082102
https://patents.google.com/patent/WO2013082102A1/un



PATENT
US 20150011510
https://patents.google.com/patent/US20150011510A1/en
Step 1:
- [0160]
13.0 g (82.5 mMol) 6-Chloro-nicotinic acid in 65 ml THF were cooled to 0° C. and 206.3 ml (206.3 mMol) o-tolylmagnesium chloride solution (1M in THF) were added over 45 minutes. The solution obtained was further stirred 3 hours at 0° C. and overnight at room temperature. It was cooled to −60° C. and 103.8 ml (1.8 Mol) acetic acid were added, followed by 35 ml THF and 44.24 g (165 mMol) manganese(III) acetate dihydrate. After 30 minutes at −60° C. and one hour at room temperature, the reaction mixture was filtered and THF removed under reduced pressure. The residue was partitioned between water and dichloromethane and extracted. The crude product was filtered on silica gel (eluent: ethyl acetate/toluene/formic acid 20:75:5) then partitioned between 200 ml aqueous half-saturated sodium carbonate solution and 100 ml dichloromethane. The organic phase was washed with 50 ml aqueous half-saturated sodium carbonate solution. The combined aqueous phases were acidified with 25 ml aqueous HCI 25% and extracted with dichloromethane. The organic extracts were dried (Na2SO4) and concentrated under reduced pressure to yield 10.4 g (51%) of 6-chloro-4-o-tolyl-nicotinic acid as a yellow foam. MS (ISN): 246 (M−H, 100), 202 (M-CO2H, 85), 166 (36).
Step 2:
- [0161]
To a solution of 8.0 g (32.3 mMol) 6-chloro-4-o-tolyl-nicotinic acid in 48.0 ml THF were added 3.1 ml (42.0 mMol) thionylchloride and 143 .mu.l (1.8 mMol) DMF. After 2 hours at 50° C., the reaction mixture was cooled to room temperature and added to a solution of 72.5 ml aqueous ammonium hydroxide 25% and 96 ml water cooled to 0° C. After 30 minutes at 0° C., THF was removed under reduced pressure and the aqueous layer was extracted with ethyl acetate. Removal of the solvent yielded 7.8 g (98%) 6-chloro-4-o-tolyl-nicotinamide as a beige crystalline foam. MS (ISP): 247 (M+H+, 100).
Step 3:
- [0162]
1.0 g (4.05 mMol) 6-Chloro-4-o-tolyl-nicotinamide in 9.0 ml 1-methyl-piperazine was heated to 100° C. for 2 hours. The excess N-methyl-piperazine was removed under high vacuum and the residue was filtered on silica gel (eluent: dichloromethane) to yield 1.2 g (95%) 6-(4-methyl-piperazin-1-yl)-4-o-tolyl-nicotinamide as a light yellow crystalline foam. - [0163]
MS (ISP): 311 (M+H+, 100), 254 (62).
Step 4:
- [0164]
A solution of 0.2 g (0.6 mMol) 6-(4-methyl-piperazin-1-yl)-4-o-tolyl-nicotinamide in 1.0 ml methanol was added to a solution of 103 mg (2.6 mMol) sodium hydroxide in 1.47 ml (3.2 mMol) NaOCl (13%) and heated for 2 hours at 70° C. After removal of methanol, the aqueous layer was extracted with ethyl acetate. The combined organic extracts were dried (Na2SO4), concentrated under reduced pressure and the residue filtered on silica gel (eluent: dichloromethane/methanol 4:1) to yield 100 mg (70%) 6-(4-methyl-piperazin-1-yl)-4-o-tolyl-pyridin-3-ylamine as a brown resin. MS (ISP): 283 (M+H+, 100), 226 (42).
Step 5:
- [0165]
2.15 mil (11.6 mMol) Sodium methoxide in methanol were added over 30 minutes to a suspension of 0.85 g (4.6 mMol) N-bromosuccinimide in 5.0 ml dichloromethane cooled to −5° C. The reaction mixture was stirred 16 hours at −5° C. Still at this temperature, a solution of 1.0 g (3.1 mMol) 6-(4-methyl-piperazin-1-yl)-4-o-tolyl-nicotinamide in 5.0 ml methanol was added over 20 minutes and stirred for 5 hours. 7.1 ml (7.1 mMol) Aqueous HCl 1N and 20 ml dichloromethane were added. The phases were separated and the organic phase was washed with deionized water. The aqueous phases were extracted with dichloromethane, brought to pH=8 with aqueous NaOH 1N and further extracted with dichloromethane. The latter organic extracts were combined, dried (Na2SO4) and concentrated to yield 1.08 g (quant.) [6-(4-methyl-piperazin-1-yl)-4-o-tolyl-pyridin-3-yl]-carbamic acid methyl ester as a grey foam. - [0166]
MS (ISP): 341 (M+H+, 100), 284 (35).
Step 6:
- [0167]
A solution of 0.5 g (1.4 mMol) [6-(4-methyl-piperazin-1-yl)-4-o-tolyl-pyridin-3-yl]-carbamic acid methyl ester in 3.0 ml dichloromethane was added over 10 minutes to a solution of 1.98 ml (6.9 mMol) Red-Al®. (70% in toluene) and 2.5 ml toluene (exothermic, cool with a water bath to avoid temperature to go >50° C.). The reaction mixture was stirred 2 hours at 50° C. in CH2Cl2, extracted with ethyl acetate and cooled to 0° C. 4 ml Aqueous NaOH 1N were carefully (exothermic) added over 15 minutes, followed by 20 ml ethyl acetate. The phases were separated and the aqueous phase was extracted with ethyl acetate. The combined organic extracts were washed with deionized water and brine, dried (Na2SO4) and concentrated under reduced pressure to yield 0.37 g (89%) methyl-[6-(4-methyl-piperazin-1-yl)-4-o-tolyl-pyridin-3-yl]-amine as an orange resin. MS (ISP): 297 (M+H+, 100).
Synthesis of 2-(3,5-bis-Trifluoromethyl-phenyl)-2-methyl-propionyl Chloride
- [0168]
- [0169]
15.0 g (50 mmol) 2-(3,5-bis-trifluoromethyl-phenyl)-2-methyl-propionic acid were dissolved in 127.5 ml dichloromethane in the presence of 0.75 ml DMF. 8.76 ml (2 eq.) Oxalyl chloride were added and after 4.5 hours, the solution was rotary evaporated to dryness. 9 ml Toluene were added and the resulting solution was again rotary evaporated, then dried under high vacuum yielding 16.25 g (quant.) of 2-(3,5-bis-trifluoromethyl-phenyl)-2-methyl-propionyl chloride as a yellow oil of 86% purity according to HPLC analysis. NMR (250 MHz, CDCl3): 7.86 (br s, 1H); 7.77, (br s, 2H, 3Harom); 1.77 (s, 6H, 2 CH3).
Synthesis of 2-(3,5-bis(trifluoromethyl)phenyl)-N,2-dimethyl-N-(6-(4-methylpiperazin-1-yl)-4-(o-tolyl)pyridin-3-yl)propanamide (Netupitant)
- [0170]
- [0171]
A solution of 20 g (67.5 mmol) methyl-[6-(4-methyl-piperazin-1-yl)-4-o-tolyl-pyridin-3-yl]-amine and 17.5 ml (101 mmol) N-ethyldiisopropylamine in 200 ml dichloromethane was cooled in an ice bath and a solution of 24 g (75 mmol)2-(3,5-bis-trifluoromethyl-phenyl)-2-methyl-propionyl chloride in 50 ml dichloromethane was added dropwise. The reaction mixture was warmed to 35-40° C. for 3 h, cooled to room temperature again and was stirred with 250 ml saturated sodium bicarbonate solution. The organic layer was separated and the aqueous phase was extracted with dichloromethane. The combined organic layers were dried (magnesium sulfate) and evaporated. The residue was purified by flash chromatography to give 31.6 g (81%) of 2-(3,5-bis(trifluoromethyl)phenyl)-N,2-dimethyl-N-(6-(4-methylpiperazin-1-yl)-4-(o-tolyl)pyridin-3-yl)propanamide as white crystals. - [0172]
M.P. 155-157° C.; MS m/e (%): 579 (M+H+, 100).
Synthesis of 5-(2-(3,5-bis(trifluoromethyl)phenyl)-N,2-dimethylpropanamido)-2-(4-methylpiperazin-1-yl)-4-(o-tolyl)pyridine 1-oxide
Step 1:
- [0174]
The solution of 6-chloropyridin-3-amine (115 g, 0.898 mol) and (Boc)2O (215.4 g, 0.988 mol) in 900 mL of dioxane was refluxed overnight. The resulting solution was poured into 1500 mL of water. The resulting solid was collected, washed with water and re-crystallized from EtOAc to afford 160 g tert-butyl (6-chloropyridin-3-yl)carbamate as a white solid (Yield: 78.2%).
Step 2:
- [0175]
To the solution of tert-butyl (6-chloropyridin-3-yl)carbamate (160 g, 0.7 mol) in 1 L of anhydrous THF was added n-BuLi (600 mL, 1.5 mol) at −78° C. under N2 atmosphere. After the addition was finished, the solution was stirred at −78° C. for 30 min, and the solution of I2 (177.68 g, 0.7 mol) in 800 mL of anhydrous THF was added. Then the solution was stirred at −78° C. for 4 hrs. TLC indicated the reaction was over. Water was added for quench, and EtOAc was added to extract twice. The combined organic phases were washed with brine, dried over Na2SO4, filtered and purified by flash chromatography to afford 80 g of tert-butyl (6-chloro-4-iodopyridin-3-yl)carbamate as a yellow solid (32.3%).
Step 3:
- [0176]
To the solution of tert-butyl (6-chloro-4-iodopyridin-3-yl)carbamate (61 g, 0.172 mol) in 300 mL of anhydrous THF was added 60% NaH (7.6 g, 0.189 mol) at 0° C. under N2 atmosphere. After the addition was finished, the solution was stirred for 30 min, and then the solution of MeI (26.92 g, 0.189 mol) in 100 mL of dry THF was added. Then the solution was stirred at 0° C. for 3 hrs. TLC indicated the reaction was over. Water was added for quench, and EtOAc was added to extract twice. The combined organic phases were washed with brine, dried over Na2SO4, filtered and concentrated to afford 63 g of crude tert-butyl (6-chloro-4-iodopyridin-3-yl)(methyl)carbamate used into the following de-protection without the further purification.
Step 4:
- [0177]
To the solution of tert-butyl (6-chloro-4-iodopyridin-3-yl)(methyl)carbamate (62.5 g, 0.172 mol) in 500 mL of anhydrous DCM was added 180 mL of TFA. Then the solution was stirred at room temperature for 4 hrs. Concentrated to remove the solvent, and purified by flash chromatography to afford 45.1 g 6-chloro-4-iodo-N-methylpyridin-3-amine as a yellow solid (Yield: 97.3%).
Step 5:
- [0178]
To the solution of 6-chloro-4-iodo-N-methylpyridin-3-amine (40.3 g, 0.15 mol) and 2-methylbenzene boric acid (24.5 g, 0.18 mol) in 600 mL of anhydrous toluene was added 400 mL of 2 N aq. Na2CO3 solution, Pd(OAc)2 (3.36 g, 15 mmol) and PPh3 (7.87 g, 0.03 mmol). The solution was stirred at 100° C. for 2 hrs. Cooled to room temperature, and diluted with water. EtOAc was added to extract twice. The combined organic phases were washed with water and brine consecutively, dried over Na2SO4, concentrated and purified by flash chromatography to afford 19 g 6-chloro-N-methyl-4-(o-tolyl)pyridin-3-amine as a white solid (Yield: 54.6%).
Step 6:
- [0179]
To the solution of 6-chloro-N-methyl-4-(o-tolyl)pyridin-3-amine (18.87 g, 81.3 mmol) and DMAP (29.8 g, 243.9 mmol) in 200 mL of anhydrous toluene was added the solution of 2-(3,5-bis-trifluoromethyl-phenyl)-2-methyl-propionyl chloride (28.5 g, 89.4 mmol) in toluene under N2 atmosphere. The solution was heated at 120° C. for 23 hrs. Cooled to room temperature, poured into 1 L of 5% aq. NaHCO3 solution, and extracted with EtOAc twice. The combined organic phases were washed by water and brine consecutively, dried over Na2SO4, filtered and purified by flash chromatography to afford 35 g 2-(3,5-bis(trifluoromethyl)phenyl)-N-(6-chloro-4-(o-tolyl)pyridin-3-yl)-N,2-dimethylpropanamide as a white solid (Yield: 83.9%).
Step 7:
- [0180]
To the solution of 2-(3,5-bis(trifluoromethyl)phenyl)-N-(6-chloro-4-(o-tolyl)pyridin-3-yl)-N,2-dimethylpropanamide (5.14 g, 10 mmol) in 60 mL of DCM was added m-CPBA (6.92 g, 40 mmol) at 0° C. under N2 atmosphere. Then the solution was stirred overnight at room temperature. 1 N aq. NaOH solution was added to wash twice for removing the excess m-CPBA and a side product. The organic phase was washed by brine, dried over Na2SO4, filtered and concentrated to afford 5.11 g of crude 5-(2-(3,5-bis(trifluoromethyl)phenyl)-N,2-dimethylpropanamido)-2-chloro-4-(o-tolyl)pyridine 1-oxide as a white solid (Yield: 96.4%).
Step 8:
- [0181]
To the solution of crude 5-(2-(3,5-bis(trifluoromethyl)phenyl)-N,2-dimethylpropanamido)-2-chloro-4-(o-tolyl)pyridine 1-oxide (5.1 g, 9.62 mmol) in 80 mL of n-BuOH was added N-methylpiperazine (7.41 g, 74.1 mmol) under N2 atmosphere. Then the solution was stirred at 80° C. overnight. Concentrated and purified by flash chromatography to afford 4.98 g 5-(2-(3,5-bis(trifluoromethyl)phenyl)-N,2-dimethylpropanamido)-2-(4-methylpiperazin-1-yl)-4-(o-tolyl)pyridine 1-oxide as a white solid (Yield: 87.2%). 1HNMR (CDCl3, 400 MHz) δ 8.15 (s, 1H), 7.93 (s, 1H), 7.78 (s, 2H), 7.38 (m, 2H), 7.28 (m, 1H), 7.17 (m, 1H), 7.07 (s, 1H), 5.50 (s, 3H), 2.72 (d, J=4.4 Hz, 4H), 2.57 (m, 3H), 2.40 (s, 3H), 2.23 (s, 3H), 1.45˜1.20 (m, 6H).
Synthesis of 4-(5-(2-(3,5-bis(trifluoromethyl)phenyl)-N,2-dimethylpropanamido)-1-oxido-4-(o-tolyl)pyridin-2-yl)-1-methylpiperazine 1-oxide
- [0182]
- [0183]
To a solution of 5-(2-(3,5-bis(trifluoromethyl)phenyl)-N,2-dimethylpropanamido)-2-(4-methylpiperazin-1-yl)-4-(o-tolyl)pyridine 1-oxide (3 g, 5.05 mmol) and NaHCO3 (0.354 g, 12.66 mmol) in 60 mL of MeOH and 15 mL of H2O were added potassium monopersulfate triple salt (1.62 g, 26.25 mmol) at room temperature during 15 min. After stirring for 4 hrs at room temperature under N2 atmosphere, the reaction mixture was concentrated in vacuo and purified by flash chromatography (eluent: MeOH). The product was dissolved into DCM, the formed solid was filtered off, and the solution was concentrated under reduced pressure to afford 1.77 g 4-(5-(2-(3,5-bis(trifluoromethyl)phenyl)-N,2-dimethylpropanamido)-1-oxido-4-(o-tolyl)pyridin-2-yl)-1-methylpiperazine 1-oxide as a white solid (Yield: 57.4%). 1HNMR (CDCl3, 400 MHz) δ 8.06 (s, 1H), 7.78 (s, 1H), 7.60 (s, 2H), 7.37˜7.20 (m, 4H), 6.81 (s, 1H), 3.89 (s, 21H), 3.74 (m, 4H), 3.31 (m, 5H), 2.48 (s, 3H), 2.18 (s, 3H), 1.36 (s, 6H).
Synthesis of 1-(5-(2-(3,5-bis(trifluoromethyl)phenyl)-N,2-dimethylpropanamido)-4-(o-tolyl)pyridin-2-yl)-4-methylpiperazine 1,4-dioxide
- [0184]
- [0185]
To the solution of 2-(3,5-bis(trifluoromethyl)phenyl)-N,2-dimethyl-N-(6-(4-methylpiperazin-1-yl)-4-(o-tolyl)pyridin-3-yl)propanamide (11.1 g, 19.2 mmol) in 75 ml of Methanol was added sodium bicarbonate (3.38 g, 40.3 mmol) dissolved in 20 ml of water. Then Oxone (14.75 g, 48.0 mmol) was added to the stirred solution at room temperature in 3-4 portions. The suspension was heated for 4 h at 50° C. After filtration of the salts (washed with 3×8 ml of methanol), the solvent has been evaporated under reduced pressure and substituted by DCM (30 ml). The organic phase was washed with water (5×30 ml), dried over Na2SO4, filtered, concentrated and purified by precipitation in toluene to afford 9.3 g 1-(5-(2-(3,5-bis(trifluoromethyl)phenyl)-N,2-dimethylpropanamido)-4-(o-tolyl)pyridin-2-yl)-4-methylpiperazine 1,4-dioxide as a white solid (Yield: 80%). 1H-NMR (CDCl3, 400 MHz, at 333K) δ 8.27 (s, 2H), 7.75 (s, 1H), 7.63 (s, 2H), 7.26˜7.19 (m, 2H), 7.14 (t, 1H, J=7.4 Hz), 7.09 (d, 1H, J=7.4 Hz), 4.93 (t, 2H, J=11.6 Hz), 4.70 (t, 2H, J=11.6 Hz), 4.12 (d, 2H, J=10.7 Hz), 3.84 (s, 3H), 3.50 (d, 2H, J=10.3 Hz), 2.47 (s, 3H), 2.12 (s, 3H), 1.40 (s, 6H).
Synthesis (A) of di-tert-butyl (chloromethyl)phosphate
- [0186]
- [0187]
Di-tert-butyl phosphohite (40.36 mmole) was combined with potassium bicarbonate (24.22 mmole) in 35 ml of water. The solution was stirred in an ice bath and potassium permanganate (28.25 mmole) was added in three equal portions over one hour’s time. The reaction as then allowed to continue at room temperature for an additional half hour. - [0188]
Decolorizing carbon (600 mg) was then incorporated as the reaction was heated to 60° C. for 15 minutes. The reaction was then vacuum filtered to remove solid magnesium dioxide. The solid was washed several times with water. The filtrate was then combined with one gram of decolorizing carbon and heated at 60° C. for an additional twenty minutes. The solution was again filtered to yield a colorless solution, which was then evaporated under vacuum to afford crude Di-tert-butyl phosphate potassium salt. Di-tert-butyl phosphate potassium salt (5 g, 20.14 mmole) was dissolved in methanol (15 g): to this solution at 0° C. a slight excess of concentrated HCl is slowly added with efficient stirring at 0° C. The addition of acid causes the precipitation of potassium chloride. The solid is then filtered and washed with methanol. The compound in the mother liquor is then converted to the ammonium form by adding an equal molar amount of tetramethylammonium hydroxide (3.65 g, 20.14 mmole) while keeping the reaction cooled by a salt/ice bath with efficient stirring. The resulting clear solution is placed under reduced pressure to give the crude product. To the tetramethylammonium di-tert-butyl-phosphate dissolved in refluxing dimethoxyethane is then added 4.3 grams of chloroiodomethane (24.16 mmole) and stirred for 1-2 hours. The reaction is then filtered and the filtrate is placed under reduced pressure to concentrate the solution in DME. The chloromethyl di-tert-butyl phosphate 12-16% in DME is used in the synthesis of 4-(5-(2-(3,5-bis(trifluoromethyl)phenyl)-N,2-dimethylpropanamido)-4-(o-tolyl)pyridin-2-yl)-1-methyl-1-((phosphonooxy)methyl)piperazin-1-ium without further purifications (60% yield): 1HNMR (CD3OD, 300 MHz) δ 1.51 (s, 12H), 5.63 (d, 2H, J=14.8). 31P-NMR (CD3OD, 300 MHz) δ −11.3 (s, 1P).
Synthesis (B) of di-tert-butyl (chloromethyl)phosphate
- [0189]
- [0190]
Di-tert-butyl phosphate potassium salt (5 g, 20.14 mmole) is dissolved in methanol (15 g): to this solution at 0° C. a slight excess of concentrated HCl is slowly added with efficient stirring at 0° C. The addition of acid causes the precipitation of potassium chloride. The solid is then filtered and washed with methanol. The compound in the mother liquor is then converted to the ammonium form by adding an equal molar amount of tetrabuthylammonium hydroxide 1 M in methanol (20.14 mmole) while keeping the reaction cooled at 0° C. with efficient stirring. The resulting clear solution is placed under reduced pressure to give the intermediate product. The tetrabuthylammonium di-tert-butyl-phosphate dissolved in acetone is then added dropwise to 53.3 grams of chloroiodomethane (302.1 mmole) and stirred at 40° C. for 1-2 hours. The solvent and excess of chloroiodomethane are distilled off, the reaction mass suspended in TBME and then filtered. The filtrate is washed by a saturated solution of sodium bicarbonate and water and then placed under reduced pressure to substitute the solvent by acetone, i.e., to remove the solvent after which it is replaced with acetone. The chloromethyl di-tert-butyl phosphate 7-20% in acetone is used in the next step without further purifications (70-80% yield): 1H-NMR (CD3OD, 300 MHz) δ 1.51 (s, 12H), 5.63 (d, 2H, J=14.8). 31P-NMR (CD3OD, 300 MHz) δ −11.3 (s, 1P).
Stability studies of 4-(5-(2-(3,5-bis(trifluoromethyl)phenyl)-N,2-dimethylpropanamido)-4-(o-tolyl)pyridin-2-yl)-1-methyl-1-((phosphonooxy)methyl)piperazin-1-ium salts
- [0191]
In order to further improve the stability and solubility of 4-(5-(2-(3,5-bis(trifluoromethyl)phenyl)-N,2-dimethylpropanamido)-4-(o-tolyl)pyridin-2-yl)-1-methyl-1-((phosphonooxy)methyl)piperazin-1-ium, a variety of its derivative salts were synthesized and tested. Their synthesis employed either a) neutralization of the dried diacid phosphate species and its corresponding base salts or b) a direct acid deprotection starting from the dried di(tert-butyl)-protected phosphate species. Neutralization was performed with L-histidine, magnesium salt, N-methyl-D-glucamine (dimeglumine), and L-lysine. Both procedures were tried in the synthesis of citric derivatives whereas with other acids the direct deprotection reaction was used. The figures below show the most relevant structures. - [0192]
When the parent acid species was not stored in dry condition it was found to undergo over 8% degradation in the first week and over 65% degradation in the first six months. When the dried parent acid species was held at 30° C. in air it underwent 0.05% degradation in the first 7 days and at total of 7.03% degradation in six months. When the dried parent acid species was held under argon at room temperature it underwent up to 0.13% degradation in the first 7 days but then was essentially stable for six months. Results for various derivative salts are shown in Table 1 below. - TABLE 1 Representative Degradation Results for Salts Purity A % Solvents Additives Yield % HPLC Comments MeOH L-Histidine, 2 eq. 26.6% 95.94% Degradation: +0.70% in 6 days (in air) +0.46% in 6 days (in argon) MeOH Mg(OH)2, 2 eq. 48.6% 94.11% Degradation: +0.81% in 6 days (in air) +0.29% in 6 days (in argon) MeOH + Citric acid, 2 eq. N.A. 94.40% From protected species. DCM, 1:1 MeOH 1. HCl dioxane, 4 eq. >90% 94.50% From protected species. 2. Ca(OH)2 MeOH H3PO4, 85%, 2 eq. >90% 98.81% From protected species and retains 0.39% of that species. MeOH HBr, 48%, 4 eq. 84.6% 96.11% From protected species. Product degrades rapidly, MeOH + CH3SO3H N.A. 61.54% From protected species. DCM, Product NOT stable: contains 1:4 32.45% decomposition species. MeOH NaH2PO4, 4 eq. N.A. n.d. Only 1.27 of parent species formed. Poor reaction. MeOH N-methyl-D- N.A. 96.88% Degradation: glucamine +0.87% in 6 days (in air) (Meglumine), 2 eq. +1.52% in 11 days (in argon) MeOH N-methyl-D- >99% 97.42% Degradation: glucamine +0.77% in 6 days (in air) (Meglumine), 1 eq. +0.83% in 7 days (in argon) MeOH+ 1. NaOH, 3 eq 96.5% 97.49% Degradation: DCM, 2. Citric acid, 1 eq. +0.09% in 2 days (in argon) 5:2 +0.59% in 89 days (in argon) MeOH+ 1. NaOH, 3 eq. 93.8% 97.46% Degradation: DCM, 2. Fumaric acid, 1 eq. +1.95% in 14 days (in air) 5:2 +1.80% in 12 days (in argon) MeOH L-lysine, 1 eq. >99% 97.62% Degradation: +0.69% in 14 days (in air) +0.48% in 12 days (in argon)
- [0194]
- [0195]
The solution of chloromethyl di-tert-butyl phosphate in DME (250 g from a 10% solution, 96.64 mmole) was evaporated under reduced pressure until the formation of pale yellow oil, dissolved then at 50° C. with 318 ml of Acetonitrile. 17.2 g (80.54 mmole) of 1,8-bis(dimethylamino)naphtalene and 46.6 g (80.54 mmole) of 2-(3,5-bis(trifluoromethyl)phenyl)-N,2-dimethyl-N-(6-(4-methylpiperazin-1-yl)-4-(o-tolyl)pyridin-3-yl)propanamide were added and the solution heated at 90° C. for at least 12 h. After the addition of 75 g of isopropylether, the precipitated crude product was cooled at room temperature, filtered and washed with acetonitrile, isopropylether/acetone, 3:1 and isopropylether, and dried under reduced pressure to afford 20-33 g of the 4-(5-{2-[3,5-bis(trifluoromethyl)phenyl]-N,2-dimethylpropanamido}-4-(o-tolyl)pyridin-2-yl)-1-methyl-1-{[(tert-butoxy)phosphoryl]oxymethyl}piperazin-1-ium as white solid (Yield: 30-50%). 1H-NMR (CD3OD, 400 MHz) δ 7.98 (s, 1H), 7.86 (s, 1H), 7.76 (s, 2H), 7.33-7.10 (m, 4H), 6.80 (s, 1H), 5.03 (d, 2H, JPH=8.5 Hz), 4.52 (s, 2H), 4.13 (m, 2H), 3.83 (m, 2H), 3.69 (m, 2H), 3.52 (m. 2H), 3.23 (s, 3H), 2.53 (s, 3H), 2.18 (s, 3H), 1.46 (s, 18H), 1.39 (s, 6H). 31P-NMR (CD3OD, 161 MHz) δ −5.01 (s, 1P). To 20 g (23.89 mmole) of the 4-(5-{2-[3,5-bis(trifluoromethyl)phenyl]-N,2-dimethylpropanamido}-4-(o-tolyl)pyridin-2-yl)-1-methyl-1-{[(tert-butoxy)phosphoryl]oxymethyl}piperazin-1-ium dissolved in 180 g of methanol and 400 g of dichloromethane was added HCl 4M in dioxane (18.8 g, 71.66 mmole) and the solution was heated for 3 h at reflux. After the addition of 200 g of dioxane, DCM and methanol were distilled under reduced pressure until precipitation of the product, which was filtered and washed with isopropylether (100 g), acetone (30 g) and pentane (2×60 g). The product was finally dried under reduced pressure at 55° C. to afford 15-17 g of 4-(5-(2-(3,5-bis(trifluoromethyl)phenyl)-N,2-dimethylpropanamido)-4-(o-tolyl)pyridin-2-yl)-1-methyl-1-((phosphonooxy)methyl)piperazin-1-ium chloride hydrochloride as white solid (Yield: 88-93%). 1H-NMR (CD3OD, 400 MHz) δ 7.02 (s, 1H), 7.87 (s, 1H), 7.74 (s, 2H), 7.33-7.40 (m, 2H), 7.27 (m, 1H), 7.21 (s, 1H), 7.16 (d, 1H, J=8.2 Hz), 5.27 (d, 2H, JPH=7.9 Hz), 4.29 (m, 2H), 4.05 (m, 2H), 3.85 (m, 2H), 3.74 (m, 2H), 3.35 (s, 3H), 2.62 (s, 3H), 2.23 (s, 3H), 1.38 (s, 6H). 31P-NMR (CD3OD, 161 MHz) δ −2.81 (t, 1P, JPH=7.9 Hz).
Synthesis (B) of 4-(5-(2-(3,5-bis(trifluoromethyl)phenyl)-N,2-dimethylpropanamido)-4-(o-tolyl)pyridin-2-yl)-1-methyl-1-((phosphonooxy)methyl)piperazin-1-ium chloride hydrochloride
- [0196]
- [0197]
To the solution of chloromethyl di-tert-butyl phosphate in Acetone (22.1 g from a 10% solution, 85.58 mmole), 15.5 g (103.24 mmole) of sodium iodide and 33.0 g (57.00 mmole) of netupitant were added and the solution heated at 50° C. for at 6-16 h. The precipitated salts were filtered off, the acetone distilled under reduced pressure and the crude product dissolved in 43.0 g of methanol and 43.0 g 1,4-dioxane. 12.6 g of HCl 4M in dioxane (113.85 mmole) were added, and then methanol is distilled off at 40° C. under reduced pressure. The solution is cooled at 5° C. and stirred at 5° C. for at least 2 h at 5° C. The product was isolated by filtration, purified by additional slurry in acetone (238 g), and filtered and washed with acetone (47 g) and pentane (2×72 g). - [0198]
The product was finally dried under reduced pressure at 60° C. to afford 22-30 g of white-yellowish solid (Yield: 50-70%) - [0199]
1H-NMR (CD3OD, 400 MHz) δ 7.02 (s, 1H), 7.87 (s, 1H), 7.74 (s, 2H), 7.33-7.40 (m, 2H), 7.27 (m, 1H), 7.21 (s, 1H), 7.16 (d, 1H, J=8.2 Hz), 5.27 (d, 2H, JPH=7.9 Hz), 4.29 (m, 2H), 4.05 (m, 2H), 3.85 (m, 2H), 3.74 (m, 2H), 3.35 (s, 3H), 2.62 (s, 3H), 2.23 (s, 3H), 1.38 (s, 6H). 31P-NMR (CD3OD, 161 MHz) δ −2.81 (t, IP, JPH=7.9 Hz).
PATENT
US 8,426,450
PATENT
US 9,403,772
SYN
https://chemistry-europe.onlinelibrary.wiley.com/doi/10.1002/chem.201901840
The synthesis of fosnetupitant (195) was developed by the Swiss company Helsinn (Scheme 34).[58] The synthesis started with the reaction of 6-chloronicotinic acid (196) with o-tolylmagnesium chloride followed by manganese(III) acetate to give acid derivative 197. This was converted to amide 198 after reaction with thionyl chloride and ammonium hydroxide. Next, reaction with N-methylpiperazine furnished intermediate 199, which was then transformed into carbamate 200 after reaction with NBS in methanol. Reduction with Red-Al followed by acylation with acyl chloride 202 afforded netupitant (203).
Finally, reaction with di-tert-butyl chloromethyl phosphate followed by the removal of the tert-butyl groups by treatment with HCl in dioxane afforded fosnetupitant (195).
L. Fadini, P. Manini, C. Pietra, C. Giuliano, E. Lovati, R. Cannella, S. Venturini, V. J. Stella, WO 082102 A1, 2013.


SYN
Fosnetupitant chloride HCl

PATENT
Fosnetupitant is a neurokynin-1 (“NK-1”) antagonist under development by Helsinn Healthcare SA, Lugano/Pazzallo Switzerland, for the treatment of chemotherapy induced nausea and vomiting. The compound is known chemically as 4-(5-(2-(3,5- bis(trifluoromethyl)phenyl)-N,2-dimethylpropanamido)-4-(o-tolyl)pyridin-2-yl)-l-methyl- 1 -((phosphonooxy)methyl)piperazin- 1 -ium, and has the following chemical structure in its acidic/free base form:

[004] The chloride monohydrochloride salt, and a method for its preparation, is described in WO 2013/082102. The chemical structure for this salt is reported as follows:

[005] The molecule can be challenging to manufacture, particularly in a highly pure crystalline form in a commercially acceptable yield. Solvents used in the manufacture of the product pose special challenges. Prior art processes have removed these solvents via evaporative techniques, which can degrade the fosnetupitant due to the excessive heat.
EXAMPLES
[089] In all the examples reported, unless otherwise reported, the starting compound was Form I of the chloride hydrochloride salt of 4-(5-(2-(3,5-bis(trifluoromethyl)phenyl)- N,2-dimethylpropanamido)-4-(o-tolyl)pyridin-2-yl)-l-methyl-l – ((phosphonooxy)methyl)piperazin-l-ium, produced substantially according to the methods described in WO 2013/082102.
EXAMPLE 1 : CHARACTERIZATION OF FOSNETUPITANT
1 . Experimental Methods
1.1 Solubility
[090] The solubility of the starting compound was determined in 25 pharmaceutically acceptable solvents (class II and III) of differing polarity. The procedure was as follows:
[091] Approximately 20 mg of material was weighed out into each glass vial.
[092] 5 volume aliquots of each solvent were added separately with stirring (i.e. 1 volume = 20 μΐ; hence, 5 volume = 100 μΐ (5 x 20 μΐ)).
[093] The mixture was stirred at RT for 5- 10 minutes. Visual checks were then made for solubility.
[094] If no solubility was achieved then steps (ii) and (iii) were repeated until either the solubility was achieved or the 50 volume aliquots of that solvent were added.
[095] Solubility was then approximated.
[096] Solubility was finally checked at the elevated temperature (40°C).
1.2 Polymorph Screen (including slurry studies)
[097] Using the information from the solubility study, the compound was slurried in the solvents outlined in Table I and two more mixtures of water/ MeOH (10:90) and water/ Acetone (1 :20) respectively with temperature cycling between 40°C and RT (4 hour periods at each temperature) over 48 hours. After the slurries the resulting solids were isolated and analyzed by Raman and XRPD (where enough material was available) for any change in physical form.
[098] The compound was also dissolved in the listed solvents and two more mixtures of water/organic solvent to yield saturated solutions, and crystallization was induced by: crash cooling (at ca. -1 8°C); evaporation (at RT); and addition of an anti-solvent. Solid materials generated were then isolated and examined by Raman and XRPD (where enough material was available).
1.3 Scale-up of any new polymorphic forms
[099] Any new potential polymorphic forms of the Form I fosnetupitant were then scaled-up to ~500mg level for further characterizations by PLM, SEM, DSC, TGA, GVS (XRPD post GVS) and NMR. Further studies of conversion between each polymorphic form were also performed. From this information, an understanding of the polymorphic space was achieved.
Synthetic Reference
Fadini, Luca; Manini, Peter; Pietra, Claudio; Giuliano, Claudio; Lovati, Emanuela; Cannella, Roberta; Venturini, Alessio; Stella, Valentino. (Assignee: Helsinn Healthcare SA, Switz). Substituted 4 – phenyl – pyridines for the treatment of nk-1 receptor related diseases. WO2013082102 (2013).
//////////Fosnetupitant, 07-PNET, Фоснетупитант , فوسنيتوبيتانت , 磷奈匹坦 , FDA 2014, EMA 2015
Centhaquine
Centhaquine
PMZ-2010
CAS 57961-90-7
2-[2-[4-(3-methylphenyl)piperazin-1-yl]ethyl]quinoline
INDIA 2020, 14.05.2020, Centhaquine citrate bulk and Centhaquine citrate injection 1.0mg/vial, Add on resuscitative agent for hypovolemic shock
- OriginatorMidwestern University; Pharmazz
- DeveloperPharmazz
- ClassAnalgesics; Antihaemorrhagics; Antihypertensives; Cardiovascular therapies; Piperazines; Quinolines; Small molecules
- Mechanism of ActionAlpha 1 adrenergic receptor antagonists; Alpha 2 adrenergic receptor agonists
- RegisteredHaemorrhagic shock
- Phase IHeart arrest; Postoperative pain
- 20 Jul 2020Pharmazz plans to launch centhaquin for Haemorrhagic shock (Adjuvant therapy) in India by the middle of September 2020
- 20 Jul 2020Efficacy data from a phase III trial in Haemorrhagic shock released by Pharmazz
- 02 Jun 2020Centhaquine is still in phase I trials for Postoperative pain in USA (Pharmazz pipeline, June 2020)
SYNCenthaquin is a compound that produces hypotension and bradycardia in higher doses and resuscitation in lower doses. It is water insoluble, and is unsuitable for intravenous use. We prepared the citrate salt of centhaquin and evaluated its cardiovascular efficacy vs. centhaquin. Centhaquin citrate was prepared and characterized; its purity was determined by HPLC. Mean arterial pressure (MAP), heart rate (HR), pulse pressure (PP), cardiac output (CO), stroke volume (SV) and stroke work (SW) following intravenous administration of centhaquin and the citrate (0.05, 0.15 and 0.45 mg.kg(-1)) were determined in anaesthetized male Sprague-Dawley rats. Centhaquin citrate was 99.8% pure and water soluble. Centhaquin (0.05, 0.15 and 0.45 mg.kg(-1)) produced a maximal decrease in MAP of 15.6, 25.2 and 28.1%, respectively; while centhaquin citrate produced a greater (p<0.001) decrease of 35.7, 47.1 and 54.3%, respectively. The decrease in PP and HR produced by the citrate was greater than centhaquin (p<0.001). At 0.45 mg.kg(-1) centhaquin produced a maximal decrease of 20.9% (p<0.01) in CO, while centhaquin citrate produced a decrease of 42.1% (p<0.001). Reduction in SV (p<0.01) and SW (p<0.001) produced by centhaquin citrate were greater than centhaquin. Centhaquin citrate has greater cardiovascular activity compared to centhaquin.https://www.semanticscholar.org/paper/Synthesis-and-characterization-of-centhaquin-and-a-Reniguntala-Lavhale/6ca3975b114b0f23753e7a47710eff2467bc2dae

PATENT
https://patents.google.com/patent/WO2014035446A1/en
Shock due to severe hemorrhage accounts for a large proportion of posttraumatic deaths, particularly during early stages of injury (Wu, Dai et al. 2009). A majority of deaths due to hemorrhage occur within the first six hours after trauma (Shackford, Mackersie et al. 1993), but many of these deaths can be prevented (Acosta, Yang et al. 1998).
[0003] Shock is accompanied by circulatory failure which is the primary cause of mortality and morbidity. Presently, the recommended fluid therapy uses large volumes of Lactated Ringer’s solution (LR), which is effective in restoring hemodynamic parameters, but presents logistic and physiologic limitations (Vincenzi, Cepeda et al. 2009). For example, resuscitation using a large volume of crystalloids, like LR, has been associated with secondary abdominal compartment syndrome, pulmonary edema, cardiac dysfunction, and paralytic ileus (Balogh, McKinley et al. 2003). Therefore, a need exists in the art for a resuscitation agent that improves survival time, and can be used with a small volume of resuscitation fluid, for resuscitation in hypovolemic shock.
[0004] Centhaquin (2-[2-(4-(3-methyphenyl)-l-piperazinyl) ethyl-quinoline) is a centrally acting antihypertensive drug. The structure of centhaquin was determined (Bajpai et al., 2000) and the conformation of centhaquin was confirmed by X-ray diffraction (Carpy and Saxena, 1991).

Structure of centhaquin (2-[2-(4-(3-methyphenyl)- 1 -piperazinyl) ethyl] -quinoline) (as free base)
[0005] Centhaquin is an active cardiovascular agent that produces a positive inotropic effect and increases ventricular contractions of isolated perfused rabbit heart (Bhatnagar, Pande et al. 1985). Centhaquin does not affect spontaneous contractions of the guinea pig right auricle, but significantly potentiates positive inotropic effect of norepinephrine (NE) (Srimal, Mason et al. 1990). The direct or indirect positive inotropic effect of centhaquin can lead to an increase in cardiac output (CO). Centhaquin produces a decrease in mean arterial pressure (MAP) and heart rate (HR) in anesthetized rats and conscious freely moving cats and rats (Srimal, Gulati et al. 1990) due to its central sympatholytic activity (Murti, Bhandari et al. 1989; Srimal, Gulati et al. 1990; Gulati, Hussain et al. 1991). When administered locally into a dog femoral artery, centhaquin (10 and 20 μg) increased blood flow, which was similar to that observed with acetylcholine and papaverine. However, the vasodilator effect of centhaquin could not be blocked by atropine or dibenamine (Srimal, Mason et al. 1990). The direct vasodilator or central sympatholytic effect of centhaquin is likely to decrease systemic vascular resistance (SVR).
[0006] It was found that centhaquin enhances the resuscitative effect of hypertonic saline (HS) (Gulati, Lavhale et al. 2012). Centhaquin significantly decreased blood lactate and increases MAP, stroke volume, and CO compared to hypertonic saline alone. It is theorized, but not relied upon, that the cardiovascular actions of hypertonic saline and centhaquin are mediated through the ventrolateral medulla in the brain (Gulati, Hussain et al. 1991 ; Cavun and Millington 2001) and centhaquin may be augmenting the effect of hypertonic saline.
[0007] A large volume of LR (i.e., about three times the volume of blood loss) is the most commonly used resuscitation fluid therapy (Chappell, Jacob et al. 2008), in part because LR does not exhibit the centrally mediated cardiovascular effects of hypertonic saline. Large volume resuscitation has been used by emergency medical personnel and surgeons to reverse hemorrhagic shock and to restore end-organ perfusion and tissue oxygenation. However, there has been a vigorous debate with respect to the optimal methods of resuscitation (Santry

ased on the molecular weight of centhaquin (free base) (MW-332) and centhaquin citrate (MW-523), for identical doses of centhaquin (as free base) and centhaquin citrate, centhaquin citrate provides only 63.5% of centhaquin free base compared to the dose of centhaquin free base, e.g., a 0.05 mg dose of centhaquin citrate contains a 0.0318 mg of centhaquin (as free base). Similarly, a dose of centhaquin citrate dihydrate (MW-559) provides 59.4% centhaquin (free base) of the same dose as centhaquin (as free base), i.e., a 0.0005 mg dose of centhaquin citrate dihydrate contains 0.030 mg of centhaquin (as free base). Surprisingly, and as demonstrated below, at the same mg/kg dose centhaquin citrate and centhaquin citrate dihydrate provides greater cardiovascular effects than centhaquin free base.

Synthesis of Centhaquin

[0061] The synthesis of centhaquin was reported by Murthi and coworkers (Murthi et al U.S. Patent No. 3,983,121 ; Murti, Bhandari et al. 1989). In one procedure, reactants 1 and 2 were stirred at reflux for 15 hours. The resulting product was purified by evaporating the solvents to obtain an oil, which was heated in vacuo (100°C, 1 mm Hg). The remaining residue was recrystallized from ether-petroleum ether to obtain the final centhaquin product 3. The melting point reported for centhaquin was 76-77°C. In a subsequent publication (Murti, Bhandari et al. 1989), the reaction mixture was concentrated following 24 hours of reflux, diluted with water, and basified with aqueous NaOH. The basic mixture was extracted with ethyl acetate, and the ethyl acetate extracts were dried over anhydrous sodium sulfate and evaporated in vacuo to give centhaquin which was crystallized from hexane. The melting point of centhaquin (free base) obtained in this procedure was 82°C. The product obtained using either purification method is light tan in color, which is indicative of small amounts of impurities that were not completely removed using previously reported purification methods.
[0062] In accordance with the present invention, an improved purification method was found. According to the improved method, reactants 1 and 2 were stirred at reflux for 24 hours. The solvents were evaporated in vacuo and the resulting mixture was diluted with water and basified (10% NaOH). The basic mixture was extracted with ethyl acetate and the combined ethyl acetate extracts are dried over anhydrous sodium sulfate and evaporated in vacuo to obtain a residue, which was further purified with column chromatography (Si02, ethyl acetate). The resulting product can be decolorized using activated charcoal or directly crystallized from hot hexane to yield pure centhaquin. The resulting product is an off-white crystalline solid having a melting point of 94-95°C (free base). The product was
characterized using proton NMR, mass spectral, and elemental analysis and indicated high purity and superior quality.
[0063] Synthesis and characterization of centhaquin (free base): A mixture of 2- vinylquinoline (1) (5.0 g, 32.2 mmol, 98.5%) and 1 -(3-methylphenyl)piperazine (2) (5.68 g, 32.2 mmol, 99.0%) in absolute ethyl alcohol (150 ml) and glacial acetic acid (3.5 ml) was stirred at reflux for 24 hours in a round bottom flask. The reaction mixture was concentrated in vacuo, diluted with water (150 ml) and treated with 10% aqueous NaOH (150 ml). The residue was extracted with ethyl acetate (4 x 125 ml), dried with anhydrous Na2S04, and concentrated under reduced pressure to yield a crude product which was purified by column chromatography using silica gel (100-200 mesh) with ethyl acetate as an eluent. The resulting compound was recrystallized from hot hexane and filtered, to yield centhaquin as an off- white crystalline solid (7.75 g, 23.4 mmol, 73% yield); mp. 94-95°C; i? 0.30 (100% ethyl acetate); 1H NMR (300 MHz, CDC13): δ 8.07 (t, J= 7.5 Hz, 2 H), 7.78 (d, J= 7.8 Hz, 1 H), 7.70 (t, J= 7.8 Hz, 1 H), 7.50 (t, J= 7.5 Hz, 1 H), 7.36 (d, J= 8.4 Hz, 1 H), 7.16 (t, J = 7.5 Hz, 1 H), 6.77 – 6.74 (m, 2 H), 6.69 (d, J= 7.2 Hz, 1 H), 3.26- 3.21 (m, 6 H), 2.97 – 2.92 (m, 2 H), 2.76 – 2.73 (m, 4 H), 2.32 (s, 3 H); HRMS (ESI) m/z 332.2121 [M+l]+ (calcd for C22H26N3 332.2122); Anal. (C22H25N3) C, H, N.
[0064] Preparation of centhaquin citrate: Centhaquin (free base) (5.62 g, 16.98 mmol) was treated with citric acid (3.26 g, 16.98 mmol) in a suitable solvent and converted to the citrate salt obtained as an off-white solid (7.96 g, 15.2 mmol, 90%); m.p. 94-96°C ; Anal.
10065] Figs. 1(a) and 1(b) are high resolution mass spectral analyses of centhaquin free base (Fig 1(a)) and centhaquin citrate (Fig. 1(b)). Compound samples were analyzed following ionization using electrospray ionization (ESI).
[0066J For centhaquin free base in Fig 1(a), a base peak [M+l]+ was observed at m z 332.2141 (theory: 332.2121) consistent with the elemental composition of protonated centhaquin (C22H26N3).
[0067] For centhaquin citrate in Fig 1(b), the mass spectrum was identical to the mass spectrum obtained for the free base. An [M+l]+base peak was observed at m z 332.2141 (theory: 332.2121), which corresponds to the elemental composition of protonated centhaquin (C22H26N3). This result is typical of salts of organic bases to yield the [M+l]+ of the free base as observed here with centhaquin citrate.
[0068] Mass spectrometry is one of the most sensitive analytical methods, and examination of the mass spectra of Fig. 1 indicate that the samples are devoid of any extraneous peaks and are of homogeneous purity (>99.5).
PATENT
https://patents.google.com/patent/WO2014035446A1/en
////////////Centhaquine, PMZ-2010, PMZ 2010, INDIA 2020, 2020 APPROVALS
Esketamine

Esketamine
- Molecular FormulaC13H16ClNO
- Average mass237.725 Da
(+)-Ketamine(2S)-2-(2-Chlorophenyl)-2-(methylamino)cyclohexanone
(S)-Ketamine33643-46-8[RN]7884Cyclohexanone, 2-(2-chlorophenyl)-2-(methylamino)-, (2S)-Cyclohexanone, 2-(2-chlorophenyl)-2-(methylamino)-, (S)-
KetamineCAS Registry Number: 6740-88-1CAS Name: 2-(2-Chlorophenyl)-2-(methylamino)cyclohexanoneMolecular Formula: C13H16ClNOMolecular Weight: 237.73Percent Composition: C 65.68%, H 6.78%, Cl 14.91%, N 5.89%, O 6.73%Literature References: Prepn: C. L. Stevens, BE634208; idem,US3254124 (1963, 1966 both to Parke, Davis). Isoln of optical isomers: T. W. Hudyma et al.,DE2062620 (1971 to Bristol-Myers), C.A.75, 118119x (1971). Clinical pharmacology of racemate and enantiomers: P. F. White et al.,Anesthesiology52, 231 (1980). Toxicity: E. J. Goldenthal, Toxicol. Appl. Pharmacol.18, 185 (1971). Enantioselective HPLC determn in plasma: G. Geisslinger et al.,J. Chromatogr.568, 165 (1991). Comprehensive description: W. C. Sass, S. A. Fusari, Anal. Profiles Drug Subs.6, 297-322 (1977). Review of pharmacology and use in veterinary medicine: M. Wright, J. Am. Vet. Med. Assoc.180, 1462-1471 (1982). Review of pharmacology and clinical experience: D. L. Reich, G. Silvay, Can. J. Anaesth.36, 186-197 (1989); in pediatric procedures: S. M. Green, N. E. Johnson, Ann. Emerg. Med.19, 1033-1046 (1990).Properties: Crystals from pentane-ether, mp 92-93°. uv max (0.01N NaOH in 95% methanol): 301, 276, 268, 261 nm (A1%1cm 5.0, 7.0, 9.8, 10.5). pKa 7.5. pH of 10% aq soln 3.5.Melting point: mp 92-93°pKa: pKa 7.5Absorption maximum: uv max (0.01N NaOH in 95% methanol): 301, 276, 268, 261 nm (A1%1cm 5.0, 7.0, 9.8, 10.5)
Derivative Type: HydrochlorideCAS Registry Number: 1867-66-9Manufacturers’ Codes: CI-581Trademarks: Ketalar (Pfizer); Ketanest (Pfizer); Ketaset (Fort Dodge); Ketavet (Gellini); Vetalar (Bioniche)Molecular Formula: C13H16ClNO.HClMolecular Weight: 274.19Percent Composition: C 56.95%, H 6.25%, Cl 25.86%, N 5.11%, O 5.84%Properties: White crystals, mp 262-263°. Soly in water: 20 g/100 ml. LD50 in adult mice, rats (mg/kg): 224 ±4, 229 ±5 i.p. (Goldenthal).Melting point: mp 262-263°Toxicity data: LD50 in adult mice, rats (mg/kg): 224 ±4, 229 ±5 i.p. (Goldenthal)
NOTE: This is a controlled substance (depressant): 21 CFR, 1308.13.Therap-Cat: Anesthetic (intravenous).Therap-Cat-Vet: Anesthetic (intravenous).Keywords: Anesthetic (Intravenous).Esketamine hydrochloride, S enantiomer of ketamine, is in phase III clinical trials by Johnson & Johnson for the treatment of depression.Drug Name:Esketamine HydrochlorideResearchCode:JNJ-54135419MOA:Dopamine reuptake inhibitor; NMDA receptor antagonistIndication:DepressionStatus:Phase III (Active)Company:Johnson & Johnson (Originator)
| Molecular Weight | 274.19 |
| Formula | C13H16ClNO•HCl |
| CAS No. | 33643-46-8 (Esketamine); 33643-47-9 (Esketamine Hydrochloride); |
Reference:1. US6040479.
https://patents.google.com/patent/US6040479A/en
EXAMPLE 1
50 g (0.21 mol) R,S-ketamine are dissolved in 613 ml of acetone at the boiling point and subsequently mixed with 31.5 g (0.21 mol) L-(+)-tartaric acid. In order to obtain a clear solution, 40 ml of water are added thereto at the boiling point and subsequently the clear solution is filtered off while still hot. After the addition of seed crystals obtained in a small preliminary experiment, the whole is allowed to cool to ambient temperature while stirring. After standing overnight, the crystals formed are filtered off with suction and dried in a circulating air drying cabinet (first at ambient temperature and then at 50-60° C.).
Yield (tartrate): 64.8 g
m.p.: 161° C.
[α]D : +26.1° (c=2/H2 O)
Thereafter, the crystallisate is recrystallised in a mixture of 1226 ml acetone and 90 ml water. After cooling to ambient temperature and subsequently stirring for 4 hours, the crystals are filtered off with suction and dried in a circulating air drying cabinet (first at ambient temperature and then at 50-60° C). There are obtained 38.8 g of tartrate (95.29% of theory).
m.p.: 175.3° C.
[α]D : +68.9° (c=2/H2 O)
The base is liberated by taking up 38.8 g of tartrate in 420 ml of aqueous sodium hydroxide solution and stirring with 540 ml of diethyl ether. The ethereal phase is first washed with water and subsequently with a saturated solution of sodium chloride. The organic phase is dried over anhydrous sodium sulphate. After filtering, the solution is evaporated to dryness on a rotary evaporator, a crystalline, colourless product remaining behind.
Yield (crude base): 21.5 g=86.0% of theory
m.p.: 118.9° C. (literature: 120-122° C.)
[α]D : -55.8° (c=2/EtOH) (literature: [α]D : -56.35° ).
In order possibly to achieve a further purification, the base can be recrystallised from cyclohexane. For this purpose, 10.75 g of the crude base are dissolved in 43 ml cyclohexane at the boiling point. While stirring, the clear solution is slowly cooled to about 10° C. and then stirred at this temperature for about 1 hour. The crystallisate which precipitates out is filtered off with suction and dried to constant weight.
Yield (base): 10.3 g=82.4% of theory
m.p.: 120° C. (literature: 120-122° C.)
[α]D : -56.8° (c=2/EtOH) (literature: [α]D : -56.35° )
EXAMPLE 2
125 ml of water are taken and subsequently 31.5 g (0.21 mol) L-(+)-tartaric acid and 50 g (0.21 mol) R,S-ketamine added thereto. While stirring, this mixture is warmed to 50-60° C. until a clear solution results. After cooling to ambient temperature while stirring and subsequently stirring overnight, the crystals formed are filtered off with suction. Subsequently, the crystallisate is first washed with water (1-6° C.) and subsequently washed twice with, in each case, 20 ml of acetone. Drying in a circulating air drying cabinet (first at ambient temperature and then at 50-60° C.) gives 31.79 g of tartrate (78.23%) of theory).
EXAMPLE 3
150 ml of water are taken and subsequently mixed with 39.8 g (0.27 mol) L-(+)-tartaric acid and 50 g (0.21 mol) R,S-ketamine. While stirring, this mixture is warmed to 50-60° C. until a clear solution results.
After cooling to ambient temperature while stirring and subsequently stirring overnight, the crystals formed are filtered off with suction. Subsequently, the crystallisate is successively washed with 8 ml of water (1-6° C.) and thereafter twice with, in each case, 20 ml acetone.
Drying in a circulating air drying cabinet (first at ambient temperature and then at 50-60° C.) gives 32.58 g of tartrate (80.02% of theory).
EXAMPLE 4
150 ml of water and 50 ml isopropanol are taken. After the addition of 39.8 g (0.21 mol) L-(+)-tartaric acid and 50 g (0.21 mol) R,S-ketamine, the mixture is heated to reflux temperature while stirring until a solution results (possibly add water until all is dissolved).
Subsequently, while stirring, the solution is allowed to cool to ambient temperature and stirred overnight. The crystals are filtered off with suction and subsequently washed with a 1:2 mixture of 20 ml of water/isopropanol and dried in a circulating air drying cabinet (first at ambient temperature and then at 50-60° C.). There are obtained 24.45 g of tartrate (62.63% of theory).
EXAMPLE 5
50 g (0.21 mol) R,S-ketamine are dissolved at the boiling point in 300 ml acetone and subsequently mixed with 31.5 g (0.21 mol) L-(+)-tartaric acid and 100 ml of water. The whole is allowed to cool while stirring and possibly seeded.
After standing overnight, the crystals formed are filtered off with suction, then washed twice with, in each case, 20 ml acetone and dried in a circulating air drying cabinet (first at ambient temperature and then at 50-60° C.). There are obtained 30.30 g of tartrate (74.57% of theory).
EXAMPLE 6
75 ml of water and 50 ml isopropanol are taken and subsequently 39.8 g (0.27 mol) L-(+)-tartaric acid added thereto. While stirring, the mixture is heated to reflux temperature until a clear solution results. After cooling to ambient temperature while stirring and subsequently stirring overnight, the crystals formed are filtered off with suction. Subsequently, the crystallisate is washed with a 1:2 mixture of 20 ml water/isopropanol. After drying in a circulating air drying cabinet (first at ambient temperature and then at 50-60° C.), there are obtained 34.84 g of tartrate (85.74% of theory).
EXAMPLE 7
20 g of the S-(+)-tartrate obtained in Example 4 are dissolved in 100 ml of water at 30-40° C. With about 7 ml of 50% sodium hydroxide solution, an S-(-)-ketamine base is precipitated out up to about pH 13. It is filtered off with suction and washed neutral with water to pH 7-8. Subsequently, it is dried for about 24 hours at 50° C. in a circulating air drying cabinet. There are obtained 11.93 g S-(-)-ketamine (97.79% of theory).
EXAMPLE 8
5 g of the S-(-)-ketamine obtained in Example 7 are dissolved in 50 ml isopropanol at about 50° C. and possibly filtered off with suction over kieselguhr. Subsequently, gaseous hydrogen chloride is passed in at 50-60° C. until a pH value of 0-1 is reached. The reaction mixture is allowed to cool to ambient temperature, filtered off with suction and washed with about 5 ml isopropanol. The moist product is dried overnight at about 50° C. in a circulating air drying cabinet. There are obtained 5.09 g S-(+)-ketamine hydrochloride (88.06% of theory).
Route 2
Reference:1. J. Am. Chem. Soc. 2015, 137, 3205-3208.
https://pubs.acs.org/doi/10.1021/jacs.5b00229
Here we report the direct asymmetric amination of α-substituted cyclic ketones catalyzed by a chiral phosphoric acid, yielding products with a N-containing quaternary stereocenter in high yields and excellent enantioselectivities. Kinetic resolution of the starting ketone was also found to occur on some of the substrates under milder conditions, providing enantioenriched α-branched ketones, another important building block in organic synthesis. The utility of this methodology was demonstrated in the short synthesis of (S)-ketamine, the more active enantiomer of this versatile pharmaceutical.



CLIP

Initial reagent: cyclopentyl Grignard Step 0: Producing cyclopentyl Grignard Reacting cyclopentyl bromide with magnesium in solvent (ether or THF) Best results: distill solvent from Grignard under vacuum and replace with hydrocarbon solvent (e.g. benzene) Step 1: processing to (o-chlorophenyl)-cyclopentyl ketone Adding o-chlorobenzonitrile to cyclopentyl Grignard in solvent, stirring for long period of time (typically three days) Hydrolyzing reaction with mixture containing crushed ice, ammonium chloride and some ammonium hydroxide Extraction with organic solvent gives (o-chlorophenyl)-cyclopentyl ketone
Step 2: processing to alpha-bromo (o-chlorophenyl)-cyclopentyl ketone ketone processed with bromine in carbon tetrachloride at low temperature (typical T = 0°C), addition of bromine dropwise forming orange suspension Suspension washed in dilute aquerous solution of sodium bisufide and evaporated giving 1-bromocyclopentyl-(o-chlorophenyl)-ketone Note: bromoketone is unstable, immeadiate usage. Bromination carried out with NBromosuccinimide result higher yield (roughly 77%) Step 3: processing to 1-hydroxycyclopentyl-(o-chlorophenyl)-ketone-N-methylimine Dissolving bromoketone in liquid methylamine freebase (or benzene as possible solvent) After time lapse (1h): excess methylamine evaporated, residue dissolved in pentane and filtered evaporation of solvent yields 1-hydroxy-cyclopentyl-(o-chlorophenyl)-ketone N-methylimine Note: longer time span (4-5d) for evaporation of methylaminemay increase yield Step 4: processing to 2-Methylamino-2-(o-chlorophenyl)-cyclohexanone (Ketamine) Method: Thermal rearragement (qualitative yield after 30min in 180°C) N-methylimine dissolved in 15ml decalin, refluxed for 2.5h Evaporation of solvent under reduced temperature followed by extraction of residue with dilute hydrochloric acid Treatment with decolorizing charcoal (solution: acidic => basic) Recrystallization from pentane-ether Note – alternative to use of decalin: pressure bomb
racemic compound, in pharmaceutical preparation racemic more active enantiomere esketamine (S-Ketamine) available as Ketanest S, but Arketamine (R-Ketamine) never marketed for clinical use, Optical rotation: varies between salt and free base form free base form: (S)-Ketamine dextrorotation (S)-(+)-ketamine hydrochloridesalt: levorotation(S)-(-)-ketamine Reason found in molecular level: different orientation of substituents: freebase: o-chlorophenyl equatorial, methylamino axia
Sources: http://creationwiki.org/Ketamine#Synthesis http://www.lycaeum.org/rhodium/chemistry/pcp/ketamine.html https://pubchem.ncbi.nlm.nih.gov/compound/ketamine https://pubchem.ncbi.nlm.nih.gov/compound/ketamine#section=Drug-Warning http://www.rsc.org/chemistryworld/2014/02/ketamine-special-k-drugs-podcast http://drugabuse.com/library/the-effects-of-ketamine-use/ http://www.drugfreeworld.org/drugfacts/prescription/ketamine.html http://onlinelibrary.wiley.com/doi/10.1002/1615-9314(20021101)25:15/17%3C1155::AID-JSSC1155%3E3.0.CO;2-M/pdf
CLIP
Process Research and Impurity Control Strategy of Esketamine Organic Process Research & Development ( IF 3.023
) Pub Date: 2020-03-18 , DOI: 10.1021/acs.oprd.9b00553
Shenghua Gao; Xuezhi Gao; Zhezhou Yang; Fuli Zhang
An improved synthesis of ( S )-ketamine (esketamine) has been developed, which was cost-effective, and the undesired isomer could be recovered by racemization. Critical process parameters of each step were identified as well as the process-related impurities. The formation mechanisms and control strategies of most impurities were first discussed. Moreover, the ( S )-ketamine tartrate is a dihydrate, which was disclosed for the first time. The practicable racemization catalyzed by aluminum chloride was carried out in quantitative yield with 99% purity . The ICH-grade quality ( S)-ketamine hydrochloride was obtained in 51.1% overall yield (14.0% without racemization) by chiral resolution with three times recycling of the mother liquors. The robust process of esketamine could be industrially scalable.
Process Research and ketamine impurity control strategy
has been developed an improved ( S ) – ketamine (esketamine) synthesis, the high cost-effective way, the undesired isomer may be recycled by racemization. Determine the key process parameters and process-related impurities for each step. First, the formation mechanism and control strategy of most impurities are discussed. In addition, ( S )-ketamine tartrate is a dihydrate, which is the first time it has been published. The feasible racemization catalyzed by aluminum chloride proceeds in a quantitative yield with a purity of 99%. ICH grade quality ( S) 5-ketamine hydrochloride can be obtained through chiral resolution and three times the mother liquor recovery rate. The total yield is 51.1% (14.0% without racemization). The robust process of ketamine can be used in Industrial promotion.

CLIP

CLIP
https://link.springer.com/article/10.1007/s13738-018-1404-1#citeas
Taghizadeh, M.J., Gohari, S.J.A., Javidan, A. et al. A novel strategy for the asymmetric synthesis of (S)-ketamine using (S)-tert-butanesulfinamide and 1,2-cyclohexanedione. J IRAN CHEM SOC 15, 2175–2181 (2018). https://doi.org/10.1007/s13738-018-1404-1
Abstract
We present a novel asymmetric synthesis route for synthesis of (S)-ketamine using a chiral reagent according to the strategy (Scheme 1), with good enantioselectivity (85% ee) and yield. In this procedure, the (S)-tert-butanesulfinamide (TBSA) acts as a chiral auxiliary reagent to generate (S)-ketamine. A series of new intermediates were synthesized and identified for the first time in this work (2–4). The monoketal intermediate (1) easily obtained after partial conversion of one ketone functional group of 1,2-cyclohexanedione into a ketal using ethylene glycol. The sulfinylimine (2) was obtained by condensation of (S)-tert-butanesulfinamide (TBSA) with (1), 4-dioxaspiro[4.5]decan-6-one in 90% yield. The (S)-N–tert-butanesulfinyl ketamine (3) was prepared on further reaction of sulfinylimine (2) with appropriate Grignard reagent (ArMgBr) in which generated chiral center in 85% yield and with 85% diastereoselectivity. Methylation of amine afforded the product (4). Finally, the sulfinyl- and ketal-protecting groups were removed from the compound (4) by brief treatment with stoichiometric quantities of HCl in a protic solvent gave the (S)-ketamine in near quantitative yield.

Esketamine, sold under the brand name Spravato[4] among others,[6][7] is a medication used as a general anesthetic and for treatment-resistant depression.[4][1] Esketamine is used as a nasal spray or by injection into a vein.[4][1]
Esketamine acts primarily as a non-competitive N-methyl-D-aspartate (NMDA) receptor antagonist.[1][8] It also acts to some extent as a dopamine reuptake inhibitor but, unlike ketamine, does not interact with the sigma receptors.[1] The compound is the S(+) enantiomer of ketamine, which is an anesthetic and dissociative similarly.[1] It is unknown whether its antidepressant action is superior, inferior or equal to racemic ketamine and its opposite enantiomer, arketamine, which are both being investigated for the treatment of depression.
Esketamine was introduced for medical use in 1997.[1] In 2019, it was approved for use with other antidepressants, for the treatment of depression in adults in the United States.[9]
In August 2020, it was approved by the U.S. Food and Drug Administration (FDA) with the added indication for the short-term treatment of suicidal thoughts.[10]
Medical uses
Anesthesia
Esketamine is a general anesthetic and is used for similar indications as ketamine.[1] Such uses include induction of anesthesia in high-risk patients such as those with hemorrhagic shock, anaphylactic shock, septic shock, severe bronchospasm, severe hepatic insufficiency, cardiac tamponade, and constrictive pericarditis; anesthesia in caesarian section; use of multiple anesthetics in burns; and as a supplement to regional anesthesia with incomplete nerve blocks.[1]
Depression
See also: List of investigational antidepressants
Similarly to ketamine, esketamine appears to be a rapid-acting antidepressant.[8][11] It received a breakthrough designation from the FDA for treatment-resistant depression (TRD) in 2013 and major depressive disorder (MDD) with accompanying suicidal ideation in 2016.[12][11] The medication was studied for use in combination with an antidepressant in people with TRD who had been unresponsive to treatment;[12][8][11] six phase III clinical trials for this indication were conducted in 2017.[12][8][11] It is available as a nasal spray.[12][8][11]
In February 2019, an outside panel of experts recommended that the FDA approve the nasal spray version of esketamine,[13] provided that it be given in a clinical setting, with people remaining on site for at least two hours after. The reasoning for this requirement is that trial participants temporarily experienced sedation, visual disturbances, trouble speaking, confusion, numbness, and feelings of dizziness during immediately after.[14]
In January 2020, esketamine was rejected by the National Health Service of Great Britain. NHS questioned the benefits and claimed that it was too expensive. People who have been already using the medication were allowed to complete treatment if their doctors consider this necessary.[15]
Side effects
Most common side effects when used in those with treatment resistant depression include dissociation, dizziness, nausea, sleepiness, anxiety, and increased blood pressure.[16]
Pharmacology
Esketamine is approximately twice as potent as an anesthetic as racemic ketamine.[17] It is eliminated from the human body more quickly than arketamine (R(–)-ketamine) or racemic ketamine, although arketamine slows its elimination.[18]
A number of studies have suggested that esketamine has a more medically useful pharmacological action than arketamine or racemic ketamine[citation needed] but, in mice, that the rapid antidepressant effect of arketamine was greater and lasted longer than that of esketamine.[19] The usefulness of arketamine over eskatamine has been supported by other researchers.[20][21][22]
Esketamine inhibits dopamine transporters eight times more than arketamine.[23] This increases dopamine activity in the brain. At doses causing the same intensity of effects, esketamine is generally considered to be more pleasant by patients.[24][25] Patients also generally recover mental function more quickly after being treated with pure esketamine, which may be a result of the fact that it is cleared from their system more quickly.[17][26] This is however in contradiction with R-ketamine being devoid of psychotomimetic side effects.[27]
Unlike arketamine, esketamine does not bind significantly to sigma receptors. Esketamine increases glucose metabolism in frontal cortex, while arketamine decreases glucose metabolism in the brain. This difference may be responsible for the fact that esketamine generally has a more dissociative or hallucinogenic effect while arketamine is reportedly more relaxing.[26] However, another study found no difference between racemic and (S)-ketamine on the patient’s level of vigilance.[24] Interpretation of this finding is complicated by the fact that racemic ketamine is 50% (S)-ketamine.
History
Esketamine was introduced for medical use as an anesthetic in Germany in 1997, and was subsequently marketed in other countries.[1][28] In addition to its anesthetic effects, the medication showed properties of being a rapid-acting antidepressant, and was subsequently investigated for use as such.[8][12] In November 2017, it completed phase III clinical trials for treatment-resistant depression in the United States.[8][12] Johnson & Johnson filed a Food and Drug Administration (FDA) New Drug Application (NDA) for approval on September 4, 2018;[29] the application was endorsed by an FDA advisory panel on February 12, 2019, and on March 5, 2019, the FDA approved esketamine, in conjunction with an oral antidepressant, for the treatment of depression in adults.[9]
In the 1980s and ’90s, closely associated ketamine was used as a club drug known as “Special K” for its trip-inducing side effects.[30][31]
Society and culture
Names
Esketamine is the generic name of the drug and its INN and BAN, while esketamine hydrochloride is its BANM.[28] It is also known as S(+)-ketamine, (S)-ketamine, or (–)-ketamine, as well as by its developmental code name JNJ-54135419.[28][12]
Esketamine is marketed under the brand name Spravato for use as an antidepressant and the brand names Ketanest, Ketanest S, Ketanest-S, Keta-S for use as an anesthetic (veterinary), among others.[28]
Availability
Esketamine is marketed as an antidepressant in the United States;[9] and as an anesthetic in the European Union.[28]
Legal status
Esketamine is a Schedule III controlled substance in the United States.[4]
References
- ^ Jump up to:a b c d e f g h i j Himmelseher S, Pfenninger E (December 1998). “[The clinical use of S-(+)-ketamine–a determination of its place]”. Anasthesiologie, Intensivmedizin, Notfallmedizin, Schmerztherapie. 33 (12): 764–70. doi:10.1055/s-2007-994851. PMID 9893910.
- ^ “Spravato 28 mg nasal spray, solution – Summary of Product Characteristics (SmPC)”. (emc). Retrieved 24 November 2020.
- ^ “Vesierra 25 mg/ml solution for injection/infusion – Summary of Product Characteristics (SmPC)”. (emc). 21 February 2020. Retrieved 24 November2020.
- ^ Jump up to:a b c d e “Spravato- esketamine hydrochloride solution”. DailyMed. 6 August 2020. Retrieved 26 September 2020.
- ^ “Spravato EPAR”. European Medicines Agency (EMA). 16 October 2019. Retrieved 24 November 2020.
- ^ “Text search results for esketamine: Martindale: The Complete Drug Reference”. MedicinesComplete. London, UK: Pharmaceutical Press. Retrieved 20 August 2017.[dead link]
- ^ Brayfield A, ed. (9 January 2017). “Ketamine Hydrochloride”. MedicinesComplete. London, UK: Pharmaceutical Press. Retrieved 20 August2017.[dead link]
- ^ Jump up to:a b c d e f g Rakesh G, Pae CU, Masand PS (August 2017). “Beyond serotonin: newer antidepressants in the future”. Expert Review of Neurotherapeutics. 17 (8): 777–790. doi:10.1080/14737175.2017.1341310. PMID 28598698. S2CID 205823807.
- ^ Jump up to:a b c “FDA approves new nasal spray medication for treatment-resistant depression; available only at a certified doctor’s office or clinic”. U.S. Food and Drug Administration (FDA) (Press release). Retrieved 2019-03-06.
- ^ “FDA Approves A Nasal Spray To Treat Patients Who Are Suicidal”. NPR. 4 August 2020. Retrieved 27 September 2020.
- ^ Jump up to:a b c d e Lener MS, Kadriu B, Zarate CA (March 2017). “Ketamine and Beyond: Investigations into the Potential of Glutamatergic Agents to Treat Depression”. Drugs. 77 (4): 381–401. doi:10.1007/s40265-017-0702-8. PMC 5342919. PMID 28194724.
- ^ Jump up to:a b c d e f g “Esketamine – Johnson & Johnson – AdisInsight”. Retrieved 7 November 2017.
- ^ Koons C, Edney A (February 12, 2019). “First Big Depression Advance Since Prozac Nears FDA Approval”. Bloomberg News. Retrieved February 12, 2019.
- ^ Psychopharmacologic Drugs Advisory Committee (PDAC) and Drug Safety and Risk Management (DSaRM) Advisory Committee (February 12, 2019). “FDA Briefing Document” (PDF). Food and Drug Administration. Retrieved February 12, 2019.
Meeting, February 12, 2019. Agenda Topic: The committees will discuss the efficacy, safety, and risk-benefit profile of New Drug Application (NDA) 211243, esketamine 28 mg single-use nasal spray device, submitted by Janssen Pharmaceutica, for the treatment of treatment-resistant depression.
- ^ “Anti-depressant spray not recommended on NHS”. BBC News. 28 January 2020.
- ^ “Esketamine nasal spray” (PDF). U.S. Food and Drug Administration (FDA). Retrieved 21 October 2019.
- ^ Jump up to:a b Himmelseher S, Pfenninger E (December 1998). “[The clinical use of S-(+)-ketamine–a determination of its place]”. Anasthesiologie, Intensivmedizin, Notfallmedizin, Schmerztherapie (in German). 33 (12): 764–70. doi:10.1055/s-2007-994851. PMID 9893910.
- ^ Ihmsen H, Geisslinger G, Schüttler J (November 2001). “Stereoselective pharmacokinetics of ketamine: R(–)-ketamine inhibits the elimination of S(+)-ketamine”. Clinical Pharmacology and Therapeutics. 70 (5): 431–8. doi:10.1067/mcp.2001.119722. PMID 11719729.
- ^ Zhang JC, Li SX, Hashimoto K (January 2014). “R (-)-ketamine shows greater potency and longer lasting antidepressant effects than S (+)-ketamine”. Pharmacology, Biochemistry, and Behavior. 116: 137–41. doi:10.1016/j.pbb.2013.11.033. PMID 24316345. S2CID 140205448.
- ^ Muller J, Pentyala S, Dilger J, Pentyala S (June 2016). “Ketamine enantiomers in the rapid and sustained antidepressant effects”. Therapeutic Advances in Psychopharmacology. 6 (3): 185–92. doi:10.1177/2045125316631267. PMC 4910398. PMID 27354907.
- ^ Hashimoto K (November 2016). “Ketamine’s antidepressant action: beyond NMDA receptor inhibition”. Expert Opinion on Therapeutic Targets. 20 (11): 1389–1392. doi:10.1080/14728222.2016.1238899. PMID 27646666. S2CID 1244143.
- ^ Yang B, Zhang JC, Han M, Yao W, Yang C, Ren Q, Ma M, Chen QX, Hashimoto K (October 2016). “Comparison of R-ketamine and rapastinel antidepressant effects in the social defeat stress model of depression”. Psychopharmacology. 233 (19–20): 3647–57. doi:10.1007/s00213-016-4399-2. PMC 5021744. PMID 27488193.
- ^ Nishimura M, Sato K (October 1999). “Ketamine stereoselectively inhibits rat dopamine transporter”. Neuroscience Letters. 274 (2): 131–4. doi:10.1016/s0304-3940(99)00688-6. PMID 10553955. S2CID 10307361.
- ^ Jump up to:a b Doenicke A, Kugler J, Mayer M, Angster R, Hoffmann P (October 1992). “[Ketamine racemate or S-(+)-ketamine and midazolam. The effect on vigilance, efficacy and subjective findings]”. Der Anaesthesist (in German). 41 (10): 610–8. PMID 1443509.
- ^ Pfenninger E, Baier C, Claus S, Hege G (November 1994). “[Psychometric changes as well as analgesic action and cardiovascular adverse effects of ketamine racemate versus s-(+)-ketamine in subanesthetic doses]”. Der Anaesthesist (in German). 43 Suppl 2: S68-75. PMID 7840417.
- ^ Jump up to:a b Vollenweider FX, Leenders KL, Oye I, Hell D, Angst J (February 1997). “Differential psychopathology and patterns of cerebral glucose utilisation produced by (S)- and (R)-ketamine in healthy volunteers using positron emission tomography (PET)”. European Neuropsychopharmacology. 7 (1): 25–38. doi:10.1016/s0924-977x(96)00042-9. PMID 9088882. S2CID 26861697.
- ^ Yang C, Shirayama Y, Zhang JC, Ren Q, Yao W, Ma M, Dong C, Hashimoto K (September 2015). “R-ketamine: a rapid-onset and sustained antidepressant without psychotomimetic side effects”. Translational Psychiatry. 5 (9): e632. doi:10.1038/tp.2015.136. PMC 5068814. PMID 26327690.
- ^ Jump up to:a b c d e “Esketamine”. Drugs.com.
- ^ “Janssen Submits Esketamine Nasal Spray New Drug Application to U.S. FDA for Treatment-Resistant Depression”. Janssen Pharmaceuticals, Inc.
- ^ Marsa, Linda (January 2020). “A Paradigm Shift for Depression Treatment”. Discover. Kalmbach Media.
- ^ Hoffer, Lee (7 March 2019). “The FDA Approved a Ketamine-Like Nasal Spray for Hard-to-Treat Depression”. Vice. Retrieved 11 February 2020.
External links
- “Esketamine”. Drug Information Portal. U.S. National Library of Medicine.
- “Esketamine hydrochloride”. Drug Information Portal. U.S. National Library of Medicine.
/////////////Esketamine, JNJ 54135419, phase 3

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO



