Adamas claims success with new and improved Parkinson’s drug
See on Scoop.it – New drug approvals
amantadine Adamas claims success with new and improved Parkinson’s drug Adamas Pharmaceuticals is claiming success with a Phase II/III study of its extended-release version of amantadine for Parkin…
Medicinal Plants Used To Treat Sexual Dysfunction- A review
See on Scoop.it – New drug approvals
Medicinal Plants Used To Treat Sexual Dysfunction: A Review by Ramandeep et al. Int J Recent Adv Pharm Res, 2013;3(1):29-35. ISSN: 2230-9306; www.ijrapronline.com. 29. Medicinal Plants Used To Tr…
Medicinal Plants Used To Treat Sexual Dysfunction- A review
See on Scoop.it – New drug approvals
Medicinal Plants Used To Treat Sexual Dysfunction: A Review by Ramandeep et al. Int J Recent Adv Pharm Res, 2013;3(1):29-35. ISSN: 2230-9306; www.ijrapronline.com. 29. Medicinal Plants Used To Tr…
Adamas claims success with new and improved Parkinson’s drug
See on Scoop.it – New drug approvals
amantadine Adamas claims success with new and improved Parkinson’s drug Adamas Pharmaceuticals is claiming success with a Phase II/III study of its extended-release version of amantadine for Parkin…
Adamas claims success with new and improved Parkinson’s drug
amantadine
Adamas claims success with new and improved Parkinson’s drug
Amantadine (trade name Symmetrel, by Endo Pharmaceuticals) is a drug that has US Food and Drug Administration approval for use both as an antiviral and an antiparkinsonian drug. It is the organic compound 1-adamantylamine or 1-aminoadamantane, meaning it consists of an adamantane backbone that has an amino group substituted at one of the four methyne positions. Rimantadine is a closely related derivative of adamantane with similar biological properties.
Apart from medical uses, this compound is useful as a building block, allowing the insertion of an adamantyl group.
According to the US Centers for Disease Control and Prevention, 100% of seasonal H3N2 and 2009 pandemic flu samples tested have shown resistance to adamantanes, and amantadine is no longer recommended for treatment of influenza in the United States. Additionally, its effectiveness as an antiparkinsonian drug is undetermined, with a 2003 Cochrane Review concluding that there was insufficient evidence in support or against its efficacy and safety.
Synthesis
Amantadine may be prepared by reacting adamantane with bromine or nitric acid to give the bromide or nitroester at position one. Reaction of either compound with acetonitrile affords the acetamide, which is hydrolyzed to give 1-adamantylamine:
New Drug Approval Process: Regulatory View
DRUG REGULATORY AFFAIRS INTERNATIONAL
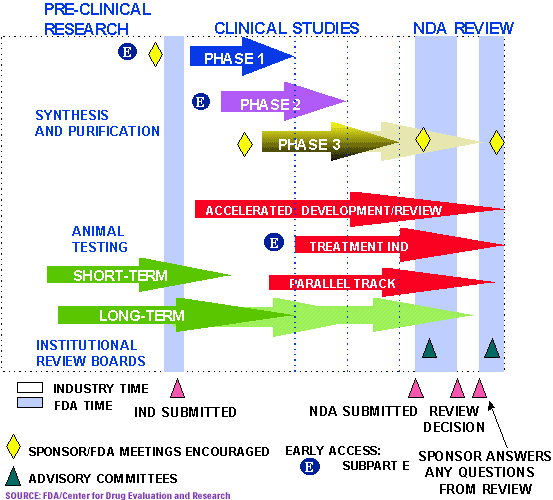
read all on
New Drug Approval Process: Regulatory View
http://www.pharmainfo.net/reviews/new-drug-approval-process-regulatory-view
Celgene saw the firm get European approval for Revlimid as a treatment for a rare form of blood disease

lenalidomide
European Commission has expanded approval on Revlimid (lenalidomide) to allow treatment of patients with transfusion-dependent anaemia due to low or intermediate-1 risk myelodysplastic syndromes (MDS) with an isolated chromosomal abnormality called deletion 5q when other options do not work. MDS is a type of cancer where the production of blood cells and platelets by the bone marrow is disrupted, which can often lead to severe anemia, infections and bleeding.
Medicinal Plants Used To Treat Sexual Dysfunction- A review
Medicinal Plants Used To Treat Sexual Dysfunction: A Review
Ramandeep et al.

Patent of Fresenius Kabi Oncology Ltd.Novel intermediates and process for the preparation of lapatinib and…….

LAPATINIB

Lapatinib (INN), used in the form of lapatinib ditosylate, (USAN) (Tykerb/Tyverb, GSK) is an orally active drug for breast cancer and other solid tumours. It is a dual tyrosine kinase inhibitor which interrupts the HER2/neu and epidermal growth factor receptor (EGFR) pathways. It is used in combination therapy for HER2-positive breast cancer. It is used for the treatment of patients with advanced or metastatic breast cancer whose tumors overexpress HER2 (ErbB2).

………………………………………………………..
Beilstein J. Org. Chem. 2013, 9, 2265–2319.
http://www.beilstein-journals.org/bjoc/single/articleFullText.htm?publicId=1860-5397-9-265
GlaxoSmithKline’s lapatinib (3.38, Tykerb) is a novel dual kinase inhibitor used in the treatment of solid tumors such as those found in breast cancer and contains a quinazoline core structure. It consists of a 2,5-disubstituted furan ring, which is directly linked to the aminoquinazoline unit (Scheme 41). The quinazoline heterocycle was prepared starting from 5-iodoanthranilic acid (3.72) via initial condensation with formamidine acetate (3.73) followed by chlorination using oxalyl chloride or phosphorous oxychloride [101]. Performing a nucleophilic aromatic substitution on the chloride 3.74 with aniline 3.75 renders the extended core of lapatinib. This intermediate (3.76) was then coupled with 5-formyl-2-furanoboronic acid (3.77) using standard Suzuki cross-coupling conditions. Finally, a reductive amination of the pendant aldehyde of 3.78 with 2-(methylsulfonyl)ethylamine (3.79) furnishes the desired product lapatinib (Scheme 41).
http://www.beilstein-journals.org/bjoc/single/articleFullText.htm?publicId=1860-5397-9-265
……………………………………………..
Fresenius Kabi Oncology Ltd.WO 2013080218
Lahiri, Saswata; Gupta, Nitin; Singh, Hemant Kumar; Handa, Vishal; Sanghani, Sunil
6 JUNE 2013, http://www.google.com/patents/WO2013080218A1?cl=en

Literature References: Reversible dual inhibitor of ErbB1 and ErbB2 tyrosine kinases. Prepn: M. C. Carter et al., WO 9935146(1999 to Glaxo); eidem, US 6727256 (2004 to SmithKline Beecham). Mechanism of action study: W. Xia et al., Oncogene 21, 6255 (2002); and crystal structure in complex with epidermal growth factor receptor (EGFR, ErbB1): E. R. Wood et al., Cancer Res. 64, 6652 (2004). In vitro antitumor activity in combination with anti-ErbB2 antibodies: W. Xia et al., Oncogene 24, 6213 (2005). Biologic effects on tumor growth: N. L. Spector et al., J. Clin. Oncol. 23, 2502 (2005). Pharmacokinetics and clinical activity in metastatic carcinomas: H. A. Burris III et al., ibid. 5305. Review of clinical development: T. E. Kim, J. R. Murren, IDrugs6, 886-893 (2003); H. A. Burris III, Oncologist 9, Suppl. 3, 10-15 (2004).

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO





![[1860-5397-9-265-i41]](https://i0.wp.com/www.beilstein-journals.org/bjoc/content/inline/1860-5397-9-265-i41.png)


