BMS 911543

BMS 911543
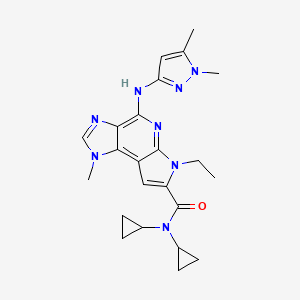
N,N-dicyclopropyl-4-((1,5-dimethyl-1H-pyrazol-3-yl)amino)-6-ethyl-1-methyl-1,6-dihydroimidazo[4,5-d]pyrrolo[2,3-b]pyridine-7-carboxamide
cas 1271022-90-2
Chemical Formula: C23H28N8O
Exact Mass: 432.23861
UNII-7N03P021J8;
N,N-dicyclopropyl-4-((1,5-dimethyl-1H-pyrazol-3-yl)amino)-6-ethyl-1-methyl-1,6-dihydroimidazo[4,5-d]pyrrolo[2,3-b]pyridine-7-carboxamide
Bristol-Myers Squibb Company innovator
BMS-911543 is an orally available small molecule targeting a subset of Janus-associated kinase (JAK) with potential antineoplastic activity. JAK2 inhibitor BMS-911543 selectively inhibits JAK2, thereby preventing the JAK/STAT (signal transducer and activator of transcription) signaling cascade, including activation of STAT3. This may lead to an induction of tumor cell apoptosis and a decrease in cellular proliferation. JAK2, often upregulated or mutated in a variety of cancer cells, mediates STAT3 activation and plays a key role in tumor cell proliferation and survival.

The JAK2 selective compound BMS911543 (WO2011028864) is in phase II clinical trials for the treatment of m elofibrosis. BMS91 1543 is shown below.

PAPER
ACS Medicinal Chemistry Letters (2015), 6(8), 850-855
Discovery of a Highly Selective JAK2 Inhibitor, BMS-911543, for the Treatment of Myeloproliferative Neoplasms

JAK2 kinase inhibitors are a promising new class of agents for the treatment of myeloproliferative neoplasms and have potential for the treatment of other diseases possessing a deregulated JAK2-STAT pathway. X-ray structure and ADME guided refinement of C-4 heterocycles to address metabolic liability present in dialkylthiazole 1 led to the discovery of a clinical candidate, BMS-911543 (11), with excellent kinome selectivity, in vivo PD activity, and safety profile

MS (ESI) m/z 434.3 (M+H). 1H NMR (CDCl3) δ: 7.96 (s, 1H), 7.65 (s, 1H), 6.83 (s, 1H), 4.67 (q, J = 7.1 Hz, 2H), 4.01 (s, 3H), 3.82 (s, 3H), 2.77 – 2.84 (m, 2H), 2.43 (s, 3H), 1.48 (t, J = 7.2 Hz, 3H), 0.79 – 0.86 (m, 4H), 0.71 – 0.77 (m, 4H).
PAPER
Journal of Organic Chemistry (2015), 80(12), 6001-601
Click to access jo5b00572_si_001.pdf
Ni-Catalyzed C–H Functionalization in the Formation of a Complex Heterocycle: Synthesis of the Potent JAK2 Inhibitor BMS-911543

BMS-911543 is a complex pyrrolopyridine investigated as a potential treatment for myeloproliferative disorders. The development of a short and efficient synthesis of this molecule is described. During the course of our studies, a Ni-mediated C–N bond formation was invented, which enabled the rapid construction of the highly substituted 2-aminopyridine core. The synthesis of this complex, nitrogen-rich heterocycle was accomplished in only eight steps starting from readily available materials.
N,N-Dicyclopropyl-4-((1,5-dimethyl-1H-pyrazol-3-yl)amino)-6-ethyl-1-methyl-1,6-dihydroimidazo[4,5-d]pyrrolo[2,3-b]pyridine-7-carboxamide, 1
PATENT
WO 2015031562
These Schemes are illustrative and are not meant to limit the possible techniques one skilled in the art may use to manufacture compounds disclosed herein.
As shown below in Scheme 1, the general preparation of compound 7 is described. Trichloroacetyl pyrrole (Compound 1) is reacted with a halogenating agent to give the C4-bromo pyrrole (Compound 2). Alcoho lysis occurs in the presence of an alcohol and base to generate ester (Compound 3), which can be selectively nitrated through contact with an appropriate nitrating agent (defined as a species that generates N02 ), yielding C5-nitro pyrrole (Compound 4). Compound 4 can be isolated as its free form, or optionally as a salt with an appropriate base. Ethylation with an appropriate alkylating agent generates the N-ethyl pyrrole (Compound 5), which in the presence of an imidazole, base, palladium and an appropriate phosphine ligand, will undergo a coupling process to form Compound 6. Reduction of the nitro-group of Compound 6 in the presence of hydrogen, a metal catalyst and optionally a base will produce Compound 7.
Scheme 1

As shown below in Scheme 2, the preparation of Compound 13 is described. Trichloroacetyl pyrrole is treated with NBS in acetonitrile to produce Compound 8. Treatment with sodium ethoxide in EtOH yields the ethyl ester Compound 9. This may be treated with a range of nitrating systems, in this example, NaNC /SCVPy, to generate nitro-pyrrole Compound 10, which can be isolated directly or as a salt form with an appropriate base, preferably dibenzylamine. Ethylation with ethyl iodide generates Compound 11 which may be isolated, or optionally telescoped directly into the arylation with Compound 32. Arylation proceeds in the presence of palladium, Xantphos, potassium pivylate and Hunig’s base to generate Compound 12. Hydrogenation presence of Pt/C followed by cyclization with NaOEt yields Compound 13.
Scheme 2

Another process of the invention is disclosed in Scheme 3 shown below. Compound 14 is prepared from Compound 3 in the presence of an alkylating agent. Treatment with a suitable diboron reagent produces Compound 15, which can then be coupled with a suitably functionalized imidazole derivative to yield Compound 16. Amino lysis with a suitable nitrogen donor produces Compound 17, which can cyclize under appropriate conditions to produce Compound 7.
Scheme 3
Step 3 Step 4 Step 5

As shown below in Scheme 4, ethylation of Compound 9 with ethyl iodide produces Compound 18. This may be directly reacted with dipinacol-diboron in the presence of Pd(OAc)2 and tricyclohexylphosphin hexafluorophosphate and
tetramethylammonium acetate to yield Compound 19. Subsequent coupling with 5-Br-imidazole derivative yields Compound 20. Treatment with hydroxylamine hydrochloride in the presence of triethylamine yields the Compound 21. Subsequent cyclization with Piv20 in the presence of PRICAT™ and hydrogen yields Compound 13.
Scheme 4
77% isolated over 2-steps%

18
Step 5 Pd(OAc)2
PPh3
78%

As shown below in Scheme 5, Compound 23 may be converted to Compound 26 by two pathways. In one option, Compound 23 can be treated with palladium, ligand and a mild base to prepare Compound 25. Reaction of Compound 25 with a metal hydroxide produces Compound 26.
Alternately, Compound 23 can be treated with palladium and ligand in the presence of a soluble hydroxide base, followed by treatment with the metal counter-ion to prepare Compound 26 directly. Once Compound 26 is formed, it can be coupled to Compound 27 to form compound I.


A solution of Compound 1 in acetonitrile (1238.0 kg, 264.9 kg after correction) was charged into a 5000 L glass-lined reactor at a temperature of 20-30 °C. The mixture was added with stirring over about 2 h and then cooled to 0 °C. NBS (221.8 kg) was charged into the mixture at intervals of 20-30 min at 0-20 °C. The mixture was cooled to 0-5 °C and reacted until the content of Compound 8was < 1.0%. Additional NBS (4.0 kg) was charged into the mixture at 0-20 °C. The mixture was reacted over 3 h until the content of Compound 8 was < 1.0%. Purified water (2650.0 kg) was added over about 1.5 – 2.5 h at 0-20 °C. The mixture was cooled to 0-5 °C and then stirred for about 1 h for crystallization. The mixture was filtered and the filter cake was rinsed with water.
Example 2

While maintaining the temperature at 20-30 °C, anhydrous ethanol (950.0 kg) was charged into a 3000 L glass-lined reactor followed by Compound 8 (342.7 kg). The mixture was cooled to 0-5 °C over about 2 h. Sodium alcoholate solution in ethanol (21%, 36.4 kg) was added dropwise over about 1-1.5 h at 0-5 °C. The reaction mixture was then heated to about 25-30 °C and tested until the content of Compounds 8/9 was < 1.0%. The reaction mixture was concentrated at a temperature < 50 °C until about 1.3-1.4 volume of Compound 8 was left. The concentrated mixture was cooled at 25-30 °C. The mixture was quenched into cooled water (3427.0 kg) over about 2 h. After addition, the mixture was stirred at 0-5 °C over about 2 h for crystallization. The mixture was filtered and the filter cake was rinsed. The solid was dried at 30-40 °C over 40-45 h to afford 234.3 kg of Compound 9 , 99.9% purity and 91.3% yield.
Example 3

9 10
A mixture of NaN03, NaHS04, and Na2S04 in CH3CN is wet-milled to constant particle size of -50 micron. To the slurry of inorganic salts is added S03 -pyridine and Compound 9. The reaction mixture is agitated at 25 °C until 90-95% conversion is achieved. The reaction is quenched with aqueous sodium hydroxide and the spent inorganic salts are removed by filtration. The filtrate is passed through a carbon pad and distilled under constant volume distillation and diluted with water to a target 15
volumes/kg of Compound 9 and a target ratio 1.0:2.0 vol/vol MeCN to water. The resulting solids are deliquored, washed, and dried to afford Compound 10.
Example 4

Toluene (10 L/Kg)
65 °C
Compound 10 (1.0 eq) and TBABr (1.0 eq) were added to a biphasic mixture of toluene (8 L/kg 10) and potassium carbonate (1.5 eq) in water (5 L/kg 10). The batch temperature was held at 25 °C. The resulting triphasic slurry was heated to 60-65 °C and diethylsulfate (1.5 eq, in a solution of toluene 2 L/kg 10) was slowly added over ~ 1 h. The reaction was aged until less than 1 RAP of Compound 10 (10:11) remained. The resulting homogeneous biphasic mixture was cooled to 20 °C and the lean aq. phase was removed. The rich organic phase was washed with water (2×7 L/kg 10) and concentrated to 6 mL/g 10. The concentrated stream was dried via azeotropic, constant volume distillation with toluene until the water content of the stream was <0.1 wt %. The resulting stream was telescoped into the subsequent direct arylation reaction.
Example 5

11 28 12
To the toluene stream of Compound 11, with potassium pivalate (1.5 equiv.) was charged, followed by DIPEA (3 eq.), Compound 28 (3 eq.) and Pd(Xantphos)Cl2 (0.04 eq.). The vessel was evacuated to < 200 torr and backfilled with nitrogen (3 X) followed by heating to 95 °C until residual Compound 11 was less than 1 RAP (11: 12). The reaction mixture was cooled to 25 °C and diluted with ethyl acetate (15 mL/g vs input pyrrole) and aq. N-acetylcysteine (0.2 eq., 5 wt % solution, 1.8 mL/g vs. input pyrrole) and heated to 50 °C for 1 h. The biphasic mixture was cooled to 25 °C. The lower aqueous layer was removed. The ethyl acetate stream was washed with water (2×7 mL/g vs. input pyrrole). The rich organic phase was polish filtered followed by a vessel/polish filter rinse with ethyl acetate (2 mL/g vs. input pyrrole). The rich organic stream was concentrated to 4 mL/g vs. input pyrrole via vacuum distillation, while maintaining the batch temperature above 50 °C. If spontaneous nucleation did not occur, Compound 12 seeds (1 wt %) were charged, followed by aging for 30 min at temperature. MTBE (5 mL/g vs. 11) was charged to the slurry over 1 hour while maintaining the batch temperature above 40 °C, followed by aging at 40 °C for 1 h. The slurry was cooled to 0 °C over 6 h and aged at 0°C for 6 h. The slurry was filtered and washed with
EtO Ac : Toluene : MTBE (1.5: 1.0: 1.5, 2 mL/g vs. input 11 ). The wet cake was dried (50 °C, 100 torr) until LOD was < 1 wt %.
Example 6

Compound 12 (1 eq., limiting reagent (LR)) is dissolved in THF/NMP (20 Vol wrt LR, 9/1 ratio) and submitted to hydrogenation using 10 wt% (wrt LR) Pt/C (5 wt%) at 25 to 40° C for 5-10 h. The reaction containing the corresponding amine is filtered. The rich organic stream is concentrated to Compound 12 Vol (wrt LR) and subjected to 0.1 eq of 21 wt% NaOEt/EtOH for 5 h at 20-25 °C, upon which Compound 13 forms. The stream is cooled to 0-10 °C, and water (5L/Kg, wrt to LR) is added and then filtered to isolate Compound 13. The product is dried at 50 °C under vacuum.
Example 7

in toluene solution
9
18
Compound 18 was prepared by treating the pyrrole with ethyl iodide and pulverized potassium carbonate in DMF at 25-30°C under inert atmosphere. After the reaction was completed, the batch mass was cooled to 15°C to 20°C and quenched by slow addition of water then MTBE. The MTBE layer was separated and washed with water. The MTBE layer was distilled to 4 Vol and solvent swapped with toluene. The toluene stream was then taken into the next step.
Example 8

18 19
Tetra-methyl ammonium acetate in toluene slurry was heated to 75-80°C to get a clear solution. The mass was cooled to below 30°C and pyrrole in toluene and bis (pinacolato) diborane were added. The reactor was inerted by nitrogen purging then the reaction was heated to 75-80°C. A freshly prepared catalyst/ligand complex (0.0 leq of palladium acetate, 0.025eq of tricyclohexyl phosphino hexafluoroborate and 0.2eq of tetra methyl ammonium acetate in toluene) was charged under nitrogen atmosphere at RT and stirred for 2h. The mass was then stirred at 75-80°C under nitrogen atmosphere. After the reaction was completed, the mixture was cooled below 30°C and quenched with aq. sodium bisulphate solution. The organic layer was polish filtered through a Celite bed and the filtrate was washed with water. The solvent swapped to ethanol until the toluene content became less than 0.5 %. The solution was cooled to 0-5°C and water was added for crystallization. The product was then isolated by filtration.
Example 9

Compound 20 was prepared by treating Compound 19 with Compound 34 in the presence of palladium acetate, triphenyl phosphine and potassium carbonate in dimethyl acetamide with the water mixture as the solvent. Dimethyl acetamide, water, potassium carbonate and the two starting materials were charged into the reactor. The mixture was made inert with nitrogen for 30 min and then charged with freshly prepared catalyst mixture (palladium acetate, triphenyl phosphine and potassium carbonate in dimethyl acetamide). The temperature was raised to 78-83 °C then the mass was stirred at this temperature. After the reaction was completed, the reaction mass was cooled to ambient temperature and purified water was added slowly into the mass for product
crystallization. The mass was stirred for a period of 3 h and filtered. The wet cake was washed with purified water and dried in VTD at 50-55 °C under vacuum.
Example 10

Compound 21 was prepared by treating Compound 20 with hydroxylamine hydrochloride and triethyl amine using ethanol as the solvent. Compound 20 was added into ethanol (15 Vol) and the reaction mass was heated to 38-40 °C. Hydroxylamine hydrochloride was charged and stirred for 10 min, then triethyl amine was added slowly at 38-40 °C over a period of lh. The above mass was stirred at 38-40 °C until Compound 20 becomes less than 5.0%, typically in about 15 h. After the reaction was completed, the above reaction mass was cooled to ambient temperature (below 30 °C) and filtered. The wet cake was washed with purified water (4 Vol) and dried under vacuum in VTD at 55-60 °C.
Example 11

Initially Compound 21 was treated with pivalic anhydride using toluene and acetic acid mixture as solvent under inert atmosphere until Compound 21 becomes less than 3.0% with respect to Compound 21, typically in about 30 min. PRICAT Nickel was then added under nitrogen atmosphere. The reaction mass was inerted with nitrogen for three cycle times and then degassed with hydrogen gas for three cycle times. Following this, 3.0 kg/cm2 hydrogen pressure was applied to the reaction mass which was stirred for about 12h. After the reaction was completed, the reaction mixture was filtered through a sparkler filter. The filtrate was distilled and the solvent exchanged with toluene until the ratio of acetic acid & toluene reaches 1 :20. At this time, n-Heptane was charged and cooled to 15°C. Then the product was filtered and the wet cake was dried in VTD at 50-55°C under vacuum.

Compound 30 was prepared by the coupling of Compound 22 with Compound 29, 3 -bromo- 1,5 -dimethyl- lH-pyrazole in the presence of
Tris(dibenzylideneacetone)dipalladium chloroform adduct, t-Brettphos and potassium phosphate in tert-amyl alcohol at 98-103 °C under inert atmosphere. After completion of the reaction (typical level of Int.9 -5% & typical reaction hrs 20 h), the mass was cooled to ambient temperature and t-amyl alcohol (4 Vol) and 20 Vol of water were charged into the reaction mass. The reaction mass was stirred for 15 min. and then phase split. The organic layer was diluted with 10 Vol of MTBE and product was extracted with 20 Vol of 1M methane sulphonic acid. The MSA stream was treated with 15 wt % charcoal to reduce the residual palladium numbers. The filtrate was cooled to below 20 °C and the pH was adjusted to 1.7-1.9 using IN NaOH for product crystallization and then iltered. The wet cake was washed with purified water (3 x 5 Vol), followed by methanol (5 Vol). The cake was vacuum dried for 3 h. then the wet cake and dimethyl sulfoxide (20 Vol) were charged into a reactor. The mass was heated to 120-125 °C to get clear solution then the mass was cooled to ambient temperature and stirred for 2 h, then filtered. The wet cake was washed with methanol (3x 4.0 Vol) and vacuum dried for 2 h. The wet cake was dried in VTD at below 55°C under vacuum.
Example 13

Compound 30 , ethanol (16.5 Vol), water and aq sodium hydroxide solution were charged into a reactor then the mass was heated to 70-75 °C and stirred until Compound 30 becomes less than 1.0%. After the reaction was completed, the mass was diluted with ethanol for complete product precipitation at 65-75 °C. Then the mass was cooled to 50 °C for a period of lh and stirred for lh at 50 °C. The mass was further cooled to 20 °C and stirred for lh at 20 °C and then filtered. The wet cake was washed with 5 Vol of 15% aqueous ethanolic solution followed by THF. The wet cake was dried under vacuum at 70-75 °C till LOD comes to less than 5.0 %, typically in about 40 h.
Example 14

In a vessel 36.5 mmol (-42.6 mL) of Compound 29 solution in 2-methyl-2-butanol was combined with 30.7g (65.1 mmol) tetrabutylammonium hydroxide (55 wt% in water), 8.01g (27.0 mmol) Compound 13 , and 10 mL 2-methyl-2-butanol. The mixture was heated at 70 °C until hydrolysis of Compound 13 was complete (full dissolution, <15 min). The solution was cooled to 60 °C and 1.12g (2.22 mmol) of tBuBippyPhos followed by 384 mg (1.028 mmol) allylpalladium chloride dimer (L:Pd = 1 :1) was added. The mixture was heated to 80 °C and was aged at this temperature for 20h before cooling to 22 °C.
Water was added and the mixture concentrated, a constant volume distillation was then performed to swap to ethanol (40-55 °C, 150 mbar). The resulting solution was passed through a 5 micron filter to remove any particulates. The solution was heated to 55 °C and 8.10 mL (40.52 mmol, 1.5 equiv) 5N NaOH (aq) was added dropwise over a 3 h period. Crystals of Compound 31 began to form, and after aging for an additional lh, the mixture was cooled to 20 °C over 3 h. After an additional 6h of aging, crystals were collected on a frit and the cake was washed with 40 mL of 90: 10 ethanol: water, followed by 48 mL acetone. After drying at 80 °C in a vacu-oven for 16 h, Compound 31 was collected as an off-white solid (8.89g, 85%).
Example 15

Compound 31 was added into dichloromethane (20 Vol) and cooled to 15-20 °C. The reaction mass was charged with DMC in DCM solution (1.4 eq of DMC in 5.0 Vol of DCM). The mixture was stirred until Compound 31 becomes less than 2.0% with respect to the corresponding acid chloride, typically in about lh. After completion of the reaction, Compound 27 (1.4 eq) and N,N-diisopropylethyleneamine (3.0 eq) were charged and the mixture was stirred. After completion of the reaction, the mass was quenched with 12 Vol of water then the layers were separated. The organic layer was washed with water and filtered through a celite bed. The filtrate was concentrated to ~6.0 vol and then the mass was cooled to 35 °C. To the resulting solution was added THF, followed by seeds of product, then stirred for 3 h. The solvent was swapped with THF until
dichloromethane becomes less than 2 wt% (wrt THF). The mass was cooled to -5 to 0 °C over a period of 2 h and stirred for 2 h. The reaction mass was then filtered under a nitrogen atmosphere. The material was slurried with pre-cooled THF (2*2 Vol) and filtered. The wet cake was dried in VTD at 60 °C under vacuum till LOD becomes < 1%, typically in about 20 h.
Example 16

DC , RT
I
To a slurry of Compound 31 (15.00 g, 40.0 mmol) in dichloromethane (300 ml) was added diphenylphosphinic chloride (12.29 g, 51.9 mmol). The mixture was stirred at room temperature for 2 h and Ν,Ν-diisopropylethylamine ( 16.53 g, 127.9 mmol) was then added and stirred for another 30 min. Compound 27 (6.94 g, 51.9 mmol) and 4-dimethylaminopyridine (0.49 g, 4.0 mmol) were subsequently added and stirred for 16 h until the reaction was completed. The reaction mixture was treated with N-acetyl-L-cysteine (3.26 g, 20.0 mmol) and citric acid (10.10 g, 48.0 mmol) in deionized water (180 ml) for 2 h. After phase split, the dichloromethane phase was washed once with 0.42 N NaOH solution (180 ml) and washed twice with deionized water (180 ml each). The final dichloromethane phase was concentrated (to 90 ml) and acetone (30 ml) was added. The solution was cooled to 35 °C and N-2 form seed of Compound 1 ( 150 mg ) was added and aged for 1 h. The resulting slurry was solvent-swapped to acetone (DCM < 10% v/v), and cooled to 0 °C. The solid was filtered and washed with cold acetone and dried to afford 14.69 g (85%) of Compound I (HPLC AP 99.8) as off-white crystals.
Patent
WO 2011028864
http://www.google.com/patents/WO2011028864A1?cl=en
Compounds of general formula I in which the R group is thiazole (as in Ial) and R1 and R2 groups are CF3 or alkyl or cycloalkyl or combine to form a saturated carbocyclic or heterocyclic ring or where R2 group is COORb could be prepared using the general method depicted in Scheme 1. Dichloro intermediate II (prepared using procedure reported in WO200612237) could be combined with a 2,4-dimethoxybenzyl and the resulting secondary amine is capped with suitable protective group (Boc) (III). The second chlorine atom could be converted into the
corresponding amine (IV) through the benzophenone imine intermediate. The amino compound could be halogenated to intermediate V. V could be subjected to transition metal mediated indole ring formation and the resulting indole nitrogen is capped with ethyl iodide to afford VI. Ester hydrolysis followed by amide bond formation and cleavage of protective groups with acid treatment would yield amine VII. Amine VII could be converted into thiourea VIII by first coupling with benzoyl isothiocyanate followed treatment with aqueous base. Formation of thiazole could be achieved by condensation with an a-bromoketone derivative (R^HBrCOR2).


a) 2,4-dimethoxybenzylamine, heat; b) NaHMDS, Boc20; c) (Ph)2=NH; d) HCl; e) NIS; f) Pd2(dba)3, ethyl pyruvate; g) Etl, Cs2C03; h) NaOH (aq); i) dicyclopropylamine HCl, HATU, DIPEA; j) TFA; k) Benzoyl isothiocyanate;
1) NaOH (aq); m) I^CHBrCOR1
Scheme 1
Compounds of general formula Ia2 in which the R1 group is CONRaRa could be made using Scheme 2. Thiourea intermediate (VIII) could be combined with Et02CCHBrCOR1 to afford the thiazole ester (IX). The ester could be hydrolyzed and the acid could be coupled with amine to afford thiazole amide derivative (la)

a) Et02CCHBrCOR1; b) NaOH (aq); c) HNRaRa, HATU, DIPEA
Scheme 2
Similarly, compounds of general formula Ia3 in which the R1 group is CONRaRa could be prepared using the general protocol depicted in Scheme 3.

a) R2CHBrCOC02Me; b) NaOH (aq); c) HNRaRa, HATU, DIPEA
Scheme 3
Compounds of general formula la in which R1 is halogen (CI, Br or I) could be prepared by condensing an a,a’-dihaloketone as depicted in Scheme 4.
a) R2COCH(Hal)2
Scheme 4
Alternatively, thiourea derivative VIII could be converted to room temperature into C-5 un-substituted thiazole XI and then directly halogenated using electrophilic halogen source or through metallation followed by quenching with an electrophilic halogenating agent (Scheme 5).

a) BrCH2COR2; b) Selectfluor or NCS or NBS or NIS or tBuLi followed Selectfluor or NBS or NCS
Scheme 5
Compounds of general formula Ia5 in which R1 is S02Rb could be synthesized using the general synthetic approach shown in Scheme 6


a) Br2-acetic acid; b) EtOH, heat
Scheme 6
Compounds with general formula la in which R1 and R2 combine to form an aromatic or heteroaromatic ring could be prepared using Scheme 7.

X = hal, -S02Me
a) Pd(0) catalyst, NaOtBu, phosphine ligand, heat
Scheme 7
Alternatively, these compounds could be made by first coupling aniline or heteroaniline (XVI) with the isothiocyanate (XV) followed by oxidative cyclization (Scheme 8).

a) 1, 1 ‘-Thiocarbonyldi-2( 1 H)-pyridone; b) NaH; c) NIS
Scheme 8
Compounds of general formula Ibl could be prepared using the general synthetic approach depicted in Scheme 9. Aniline VII could be combined with γ-dithiomethylketone compound XVII, (prepared using the procedure reported at room temperature in Synlett, p 2331 (2008)) under basic condition to afford XVIII.
Stepwise condensation of the Boc-protected hydrazine derivative would give the required pyrazole Ibl.

a) NaH, THF; b) R1N(Boc)NH2, AcOH, 35-40°C; c) HCO2H or TFA, 60°C
Scheme 9
Compounds of general formula Ibl or Ifl and If could also be prepared by coupling C-4 halo derivative (XIX) with an appropriately substituted 2-aminopyrazole derivative (XX) using a transition metal catalyzed reaction (Scheme 10).

a) isoamyl nitrite, CH2I2 or isoamyl nitrite, CH2Br2; b) Pd2(dba)3, Xanphos, Cs2C03
Scheme 10
Compounds of general formula Ib2 in which R2 group is CONRaRa could be synthesized using Scheme 11. Aniline VII could be combined with γ-dithiomethylketone derivative XXII, (prepared using the procedure from
Tetrahedron, p 2631 (2003)) to afford intermediate XXIII. Stepwise condensation of Boc-protected hydrazine derivative would give the required pyrazole aldehyde XXIV. Aldehyde could be oxidized using oxone or sodium hypochlorite to furnish carboxylic acid XXV. Coupling of acid XXV with amine would give pyrazole amide Ib2.

a) NaH, THF, heat; b) R1N(Boc)NH2, AcOH; c) TFA; d) oxone or sodium hypochlorite; e) HNRaRa, HATU, DIPEA
Scheme 11
Compounds of general formula Icl could be prepared using the general protocol as shown in Scheme 12. Aniline VII could be coupled with chloroacetyl chloride and the resulting amide could be treated with thioamide (R2CS H2) to furnish thiazole Icl .

a) chloroacetyl chloride, base; b) R2CSNH2
Scheme 12
00120] Compounds of general formula ldl could be made as per Scheme 13. Previously described isothiocyanate derivative XV could be combined with amidine XXV under dehydrating reaction conditions to give 1,2,4-thiadiazole (ldl).

Scheme 13
Compounds of general formula lei could be prepared using a synthetic approach as shown in Scheme 14. Isothiocyanate XV could be combined with azide XXVI in the presence of phosphine to yield 1,3-oxazole Iel .

Scheme 14
Compounds of general formula lgl could be prepared using a synthetic approach as shown in Scheme 15. Amine VII could be combined with acyl isothiocyanate XXVII. The acylthioureaido could be condensed with hydrazine derivative to yield the 1,2,4-triazol derivative lgl.

igi
Scheme 15
without a methyl

Preparation of 7V,7V-dicyclopropyl-6-ethyl-l-methyl-4-(5-m ethyl- lH-pyrazol-3- ylamino)-l,6-dihydroimidazo[4,5-d]pyrrolo[2,3-b]pyridine-7-carboxamide
[00437] Prepared using similar protocol as for example 72 from hydrazine.
[00438] MS (ESI) m/z 419.3 (M+H)
[00439] 1H NMR (400 MHz, CHLOROFORM-d) δ ppm 8.70 (br s, 1 H), 7.91 (br s, 1 H), 6.87 (s, 1 H), 6.09 (br s, 1 H), 4.64 (q, 2 H, J= 7.03 Hz), 4.08 (s, 3 H), 2.74 -2.95 (m, 2 H), 2.41 (s, 3 H), 1.51 (t, 3 H, J= 7.15 Hz), 0.81 – 0.95 (m, 4 H), 0.70 -0.81 (m, 4 H)
with an ethyl

7V,iV-dicyclopropyl-6-ethyl-4-(l-ethyl-5-methyl-lH-pyrazol-3-ylamino)-l-methyl- 1,6-dihydroimidazo [4,5-d] pyrrolo [2,3-b] pyridine-7-carboxamide
74A Preparation of fe/t-butyl l,3-dioxoisoindolin-2-yl(ethyl)carbamate

Diisopropyl azodicarboxylate (2.92 mL, 15.00 mmol) was added in one portion to a solution of tert-butyl l,3-dioxoisoindolin-2-ylcarbamate (2.62 g, 10 mmol, prepared following the procedure described by Nicolas Brosse et al. in Eur. J. Org. Chem. 4757-4764, 2003), triphenylphosphine (3.93 g, 15.00 mmol) and ethanol (0.691 g, 15.00 mmol) in THF (20 mL) at 0 °C and the reaction solution was stirred at room temperature for lh (monitored by TLC until completion). Solvent was evaporated and the residue was purified by flash chromatography on silica gel using an automated ISCO system (80 g column, eluting with 5-35% ethyl acetate / hexanes) to provide tert-butyl l,3-dioxoisoindolin-2-yl(ethyl)carbamate (2.6 g, 90 % yield) as a white solid which was used as it in the next step
74B Preparation of fe/t-butyl l-ethylhydrazinecarboxylate
Boc
H2N-N
\
Methylhydrazine (1.415 niL, 26.9 mmol) was added to a solution oi tert-butyl l,3-dioxoisoindolin-2-yl(ethyl)carbamate (example 74A, 5.2 g, 17.91 mmol) in THF (40 mL) at 0 °C and the reaction mixture was stirred at room temperature overnight. A white precipitate formed and was filtered off through a pad of Celite, The filtrate was concentrated in vacuo. The residue was dissolved in ethyl acetate (50 ml) and extracted with IN HC1 (3×30 ml), the acid layer was washed with ethyl acetate (50 ml) and basified to pH 10 by addition of 20% NaOH. The basic solution was then extracted with ethyl acetate (3×50 ml) and the combined organic layers were washed with brine, dried over magnesium sulfate, filtered and concentrated in vacuo to give tert-butyl 1 -ethylhydrazinecarboxylate (2.5 g, 87 % yield) as colorless oil.
XH NMR (400 MHz, CDC13) δ: 3.90 (br. s., 2H), 3.35 (q, J = 7.0 Hz, 2H), 1.42 (s, 9H), 1.07 (t, J = 7.0 Hz, 3H)
74 Preparation of N.N-dicyclopropyl-6-ethyl-4-(l-ethyl-5-methyl-lH-pyrazol-3-ylamino)-l-methyl-l ,6-dihydroimidazor4,5-d1pyrrolor2,3-b1pyridine-7-carboxamide
A mixture of (Z)-N,N-dicyclopropyl-6-ethyl- 1 -methyl-4-( 1 -(methylthio)-3-oxobut-l-enylamino)-l,6-dihydroimidazo[4,5-d]pyrrolo[2,3-b]pyridine-7-carboxamide (example 74B, 70 mg, 0.155 mmol) and tert-butyl 1-ethylhydrazinecarboxylate (49.6 mg, 0.309 mmol) in acetic acid (1 mL) wan stirred at 35 °C for 4 h (monitored by LC/MS until no starting material left). Formic acid (1 mL) was added and the reaction mixture stirred at 60 °C for 6 h. The solvent was evaporated and the crude product was purified by flash chromatography on silica gel using an automated ISCO system (12 g column, eluting with 2-10% methanol / dichloromethane). The material was further purified by preparative HPLC to afford N,N-dicyclopropyl-6-ethyl-4-( 1 -ethyl-5-methyl- lH-pyrazol-3-ylamino)- 1 -methyl- 1 ,6-dihydroimidazo[4,5-d]pyrrolo[2,3-b]pyridine-7-carboxamide (38 mg, 53.4 % yield) as an off-white solid.
MS (ESI) m/z 447.3 (Μ+Η).
XH NMR (500 MHz, CDC13) δ: 8.08 (s, 1H), 7.61 (s, 1H), 6.93 (s, 1H),
6.84 (s, 1H), 4.66 (q, J = 7.1 Hz, 2H), 4.02 (q, J = 7.2 Hz, 2H), 3.98 (s, 3H), 2.79 – 2.85 (m, 2H), 2.34 (s, 3H), 1.49 (t, J = 7.1 Hz, 3H), 1.41 (t, J = 7.2 Hz, 3H), 0.82 -0.87 (m, 4H), 0.72 – 0.78 (m, 4H).
Patent
JAK2 INHIBITORS AND THEIR USE FOR THE TREATMENT OF MYELOPROLIFERATIVE DISEASES AND CANCER [US8202881]2011-03-102012-06-19
JAK2 inhibitors and their use for the treatment of myeloproliferative diseases and cancer [US8673933]2012-04-302014-03-18
: Purandare AV, McDevitt TM, Wan H, You D, Penhallow B, Han X, Vuppugalla R, Zhang Y, Ruepp SU, Trainor GL, Lombardo L, Pedicord D, Gottardis MM, Ross-Macdonald P, de Silva H, Hosbach J, Emanuel SL, Blat Y, Fitzpatrick E, Taylor TL, McIntyre KW, Michaud E, Mulligan C, Lee FY, Woolfson A, Lasho TL, Pardanani A, Tefferi A, Lorenzi MV. Characterization of BMS-911543, a functionally selective small-molecule inhibitor of JAK2. Leukemia. 2012 Feb;26(2):280-8. doi: 10.1038/leu.2011.292. Epub 2011 Oct 21. PubMed PMID: 22015772.
Characterization of BMS-911543, a functionally selective small-molecule inhibitor of JAK2http://www.nature.com/leu/journal/vaop/ncurrent/full/leu2011292a.html
GRAPHS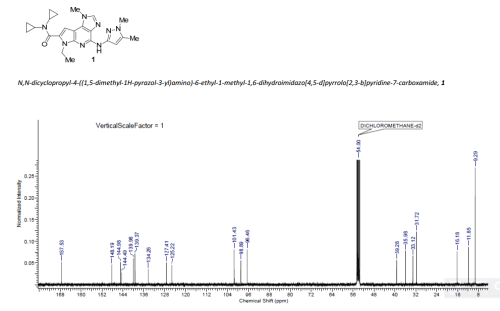
Click to access jo5b00572_si_001.pdf

//////BMS 911543, phase 2, bms,
Regorafenib, SHILPA MEDICARE LIMITED, New patent, WO 2016005874

WO2016005874, PROCESS FOR THE PREPARATION OF REGORAFENIB AND ITS CRYSTALLINE FORMS
SHILPA MEDICARE LIMITED [IN/IN]; 10/80,Second Floor,Rajendra Gunj, Raichur, ರಾಯಚೂರು , karnataka 584102 (IN)
RAMPALLI, Sriram; (IN).
UPALLA, Lav Kumar; (IN).
RAMACHANDRULA, Krishna Kumar; (IN).
PUROHIT, Prashant; (IN).
AKSHAY KANT, Chaturvedi; (IN)
The present invention relates to a process for the preparation of 4-[4-({[4-chloro-3-(trifluoromethyl)phenyl]carbamoyl}amino)-3-fluorophenoxy]-N-methylpyridine-2- carboxamide or Regorafenib (I): Formula (I). The present invention further relates to a process for the purification of 4-[4-({[4-chloro-3-(trifluoromethyl) phenyl] carbamoyl} amino)-3-fluorophenoxy]-N-methylpyridine-2- carboxamide or Regorafenib (I) to provide highly pure material. The present invention further relates to a process for the preparation stable crystalline material of 4-[4-({[4-chloro-3-(trifluoromethyl)phenyl]carbamoyl}amino)-3-fluorophenoxy]- N-methyl pyridine-2-carboxamide or Regorafenib (I) useful in the preparation of pharmaceutical compositions for the treatment of cancer.
4-[4-({[4-chloro-3-(trifluoromethyl)phenyl]carbamoyl}amino)-3-fluorophenoxy]-N-methylpyridine-2-carboxamide or Regorafenib is low molecular weight, orally available, inhibitor of multiple protein kinases, including kinases involved in tumour angiogenesis (VEGFR1, -2, -3, TIE2), oncogenesis (KIT, RET, RAF-1, BRAF, BRAFV600E), and the tumour microenvironment (PDGFR, FGFR). In preclinical studies regorafenib has demonstrated antitumour activity in a broad spectrum of tumour models including colorectal tumour models which is mediated both by its antiangiogenic and antiproliferative effects. Major human metabolites (M-2 and M-5) exhibited similar efficacies compared to Regorafenib both in vitro and in vivo models.
Regorafenib was approved by USFDA in 2012 and is marketed under the brand name Stivarga®, is an important chemotherapeutic agent useful for the treatment of adult patients with metastatic colorectal cancer (CRC) who have been previously treated with, or are not considered candidates for, available therapies. These include fluoropyrimidine-based chemotherapy, an anti-VEGF therapy and an anti-EGFR therapy.
Regorafenib is chemically known as 4-[4-({[4-chloro-3-(trifluoromethyl) phenyl] carbamoyl} amino)-3-fluorophenoxy]-N-methylpyridine-2-carboxamide (I). Regorafenib is a white to slightly pink or slightly brownish solid substance with the empirical formula C2iHi5ClF4N403 and a molecular weight of 482.82. Regorafenib is practically insoluble in water, dilute alkaline solution, dilute acid solution, n-heptane, glycerine and toluene. It is slightly soluble in acetonitrile, dichloromethane, propylene glycol, methanol, 2-propanol, ethanol and ethyl acetate. It is sparingly soluble in acetone and soluble in PEG 400 (macrogol). Regorafenib is not hygroscopic.
Regorafenib is generically disclosed in US 7351834, and specifically disclosed in US 8637553. US ‘553 disclose a process for the preparation of Regorafenib starting from 3-fluoro-4-nitrophenol. The process is as demonstrated below:

The present inventors has repeated the above process and found the following disadvantages:
Unwanted reactions are observed during the formation of Regorafenib, due to the involvement of prolonged time in process.
> Incomplete reactions were observed with excessive impurity formations due to incomplete conversion.
Removal of impurities from final product
US 2010173953 disclose Regorafenib monohydrate and crystalline Form I of Regorafenib. This patent application further discloses that crystalline Form I of Regorafenib stated in this application is obtained as per the process disclosed in WO 2005009961 A2 (Equivalent to US ‘553). The compound obtained was having a melting point of 186-206° C.
This patent publication discloses a process for the preparation of Regorafenib monohydrate comprises dissolving Regorafenib Form I obtained as per WO ‘961 in acetone
and the solution is filtered, followed by addition of water until precipitation, which was filtered and dried at room temperature
US 2010/0113533 discloses crystalline Form II of Regorafenib, comprises dissolving Regorafenib Form I obtained as per WO ‘961 in ethyl acetate, the suspension was heated to 40-45°C, addition of isocyanate solution (isocyanate in ethyl acetate) and is cooled to room temperature to yield the crystals, which was filtered, washed with ethyl acetate and dried at room temperature.
US 2010/0063112 discloses Form III of Regorafenib, process comprises of heating
Regorafenib monohydrate at 100°C or 60 min, and further 15 min at 110°C, followed by cooling to room temperature.
As polymorphism has been given importance in the recent literatures owing to its relevance to the drugs having oral dosage forms due to its apparent relation to dose preparation/suitability in composition steps/ bioavailability and other pharmaceutical profiles, stable polymorphic form of a drug has often remained the clear choice in compositions due to various reasons of handling, mixing and further processing including bioavailability and stability.
Exploring new process for these stable polymorphic forms which are amenable to scale up for pharmaceutically active / useful compounds such as 4-[4-({[4-chloro-3-(trifluoro methyl)phenyl]carbamoyl } amino)-3 -fluorophenoxy] -N-methylpyridine-2 -carboxamide or Regorafenib may thus provide an opportunity to improve the drug performance characteristics of such products.
Hence, inventors of the present application report a process for the preparation of a stable and usable form of 4-[4-({[4-chloro-3-(trifluoromethyl)phenyl]carbamoyl}amino)-3-fluoi phenoxy]-N-methylpyridine-2-carboxamide or Regorafenib, which may be industrially amenable and usable for preparing the corresponding pharmaceutical compositions. The present invention provides an improved process for the preparation of 4-[4-({[4-chloro-3-(trifluoromethyl)phenyl]carbamoyl}amino)-3-fiuorophenoxy]-N-methylpyridine-2-carboxamide or Regorafenib crystalline forms specifically for crystalline polymorphic forms Form I and Form III. Crystalline polymorphic forms of 4-[4-({[4-chloro-3-(trifluoromethyl) phenyl] carbamoyl } amino)-3 -fluorophenoxy] -N-methylpyridine-2 -carboxamide or Regorafenib obtained by the process of the present invention is non-hygroscopic and chemically stable and has good dissolution properties.
The process related impurities that appear in the impurity profile of the Regorafenib may be substantially removed by the process of the present invention resulting in the formation of highly pure material. The process of the present invention is as summarized below:

Example 1
Preparation of 4-(4-amino-3-fluorophenoxy) pyridine-2-carboxylic acid methyl amide
4-Amino-3-fiuorophenol (l lg, 0.08 moles) and of 4-Chloro-N-methyl-2-pyridinecarboxamide (8.85 g, 0.05 moles) was added to a reaction flask containing N, N-dimethylacetamide (55 ml) at 25-30°C and stirred for 15 minutes. The reaction mixture was heated to 110-115°C and then potassium tert-butoxide in tetrahydrofuran (60 ml, 0.06 moles) was added slowly over a period of 3 to 4hours. Distill off solvent at same temperature, cooled the reaction mass to 25-30°Cand water(110 ml) was added slowly over a period of 15min. and cooled the reaction mass to 0-5°C . Adjust the pH of the reaction mass in between 7 and 7.5 by using 10% aqueous hydrochloric acid (~7 ml). Stir the reaction mass for 30min at the same temperature. Filter the product, washed with water (22 mL) and Dried at 50-55 °C for 12hrs. The obtained crude material was added to the flask containing Ethyl acetate (55 mL).The reaction mass was heated to reflux to get a clear solution and stirred for 15min at reflux. Cooled to 0-5°C, stir for 2hrs at the same temperature. Filter the product, washed with Toluene (9 mL) and dried at 50-55°C for 3-5hrs.
Above recrystallized material was added to the reaction flask containing methylene dichloride (270 mL) at 25-30°C and stirred for 10-15 min. Activated carbon (1 g) and silica gel (4.4 g) was added to the reaction mass and stir for lh at the same temperature. Filter the reaction mass through hyflow bed and wash with methylene dichloride (18 mL).Distill off solvent still~l-2 volumes of methylene dichloride remains in the flask and then cooled to 25-30°C. Toluene (20 mL) was added and stirred for 30min at the same temperature. Filtered the product, washed with Toluene (9 mL) and dried at 50-55°C for 12h.
Yield: 9 gm
Chromatographic Purity (By HPLC): 98%
Example 2
Preparation of Regorafenib
4-(4-amino-3-fluorophenoxy) pyridine-2-carboxylic acid methyl amide (4g, 0.01 moles) was added in to a reaction flask containing acetone (20 ml) at 25-30°C and stirred for 15 minutes. 4-chloro-3-trifluoromethylisocyanate (6.1g, 0.02 moles) was added slowly over a period of 5 to 10 minutes and stirred the reaction mixture 3 to 4 hours. Toluene (20 n L) was added to the reaction mass and stirred for 30 min at 25-30°C.The obtained reaction mass was filtered and washed with toluene (8 mL). Dried the material still constant weight appears to yield title product a crystalline material.
Yield: 5.5 gm
Chromatographic Purity (By HPLC): 97%
Example 3
Purification of Regorafenib using acetone and toluene mixture
4- [4-( { [4-chloro-3 -(trifluoromethyl)phenyl] carbamoyl } amino)-3 -fluorophenoxy] -N-methylpyridine-2-carboxamide (I) or Regorafenib (1 g) was added slowly in to the reaction flask containing acetone (2 mL) and toluene (3 mL) at 25-30°C and stirred for 15 minutes.
The reaction mixture was heated to 50-55°C and stirred the reaction mixture for 30 minutes.
Cooled the reaction mass to 25-30°C and stirred for 1 hour. Filter the material, washed with toluene (2 mL) and suck dried for 15 min, followed by drying at 50-55°C for 10-12h to yield
Pure 4-[4-({[4-chloro-3-(trifluoromethyl)phenyl]carbamoyl}amino)-3-fluorophenoxy]-N-methyl pyridine-2-carboxamide (I) or Regorafenib.
Yield: 0.88gm
Chromatographic Purity (By HPLC): 99.3 %
Example 4
Purification of Regorafenib using acetone
4-[4-({[4-chloro-3-(trifluoromethyl) phenyl] carbamoyl} amino)-3 -fluorophenoxy] -N-methylpyridine-2-carboxamide (I) or Regorafenib (1 g) was added slowly in to the reaction flask containing acetone (5 mL) at 25-30°C and stirred for 15 minutes. The reaction mixture was heated to 50-55°C and stirred the reaction mixture for 30 minutes. Cooled the reaction mass to 0-5°C and stirred for 1 hour. Filter the material, washed with acetone (1 mL) and suck dried for 15 min. The obtained wet cake was added in to the reaction flask containing acetone (5 mL) at 25-30°C and stirred for 15 minutes. The reaction mixture was heated to 50- 55°C and stirred the reaction mixture for 30 minutes. Cooled the reaction mass to 0-5°C and stirred for 1 hour. Filter the material, washed with acetone (1 mL) and dried at 60-65°C for 12 h to yield Pure 4-[4-({[4-chloro-3-(trifluoromethyl)phenyl]carbamoyl}amino)-3-fluorophenoxy]-N-methyl pyridine -2-carboxamide (I) or Regorafenib.
Yield: 0.7 gm
Chromatographic Purity (By HPLC): 99.77%
Example 5
Double – Purification of Regorafenib using acetone and toluene mixture
4-[4-({[4-chloro-3-(trifluoromethyl) phenyl] Carbamoyl} amino)-3-fluorophenoxy]-N-methylpyridine-2-carboxamide (I) or Regorafenib (1 g) was added slowly in to the reaction flask containing acetone (2 mL) and toluene (3 mL) at 25-30°C and stirred for 15 minutes. The reaction mixture was heated to 50-55°C and stirred the reaction mixture for 30 minutes. Cooled the reaction mass to 25-30°C and stirred for 1 hour. Filter the material, washed with toluene (2 mL) and suck dried for 15 min. The obtained wet cake was added in to the reaction flask containing acetone (2 mL) and toluene (3 mL) mixture at 25-30°C and stirred for 15 minutes. The reaction mixture was heated to 50-55°C and stirred the reaction mixture for 30 minutes. Cooled the reaction mass to 25-30°C and stirred for 1 hour. Filter the material, washed with toluene (2 mL) and dry at 60-65°C for 12h.
Yield: 0.80gm
Chromatographic Purity (By HPLC): 99.79 %
Moisture content: 0.09%
Impurity-A: 0.03%
Impurity-B: Not detected
Impurity-C: 0.02%
Example 6
Preparation of Regorafenib Form I
4-(4-amino-3-fluorophenoxy) pyridine-2-carboxylic acid methyl amide (1.3 g, 0.004 moles) was added in to a reaction flask containing acetone (13 mL) at 25-30°C and stirred for 15 minutes.4-chloro-3-trifluoromethylisocyanate (6.6 g, 0.006 moles) wasadded slowly over a period of 15 to 20 minutes and stirred the reaction mixture 3 to 4 hours. The obtained reaction mass was filtered and washed with acetone. Dried the material still constant weight appears to yield title product a crystalline material.
Yield: 1.9 g
Chromatographic Purity (By HPLC): 98.4 %
XRPD was found to resemble similar to Fig-1.

Omprakash Inani – Chairman, Vishnukant C Bhutada – Managing Director, Namrata Bhutada – Non Executive Director, Ajeet Singh Karan – Independent Director, Carlton Felix Pereira – Independent Director, Pramod Kasat – Independent Director, Rajender Sunki Reddy – Independent Director, N P S Shinh – Independent Director,
 Mr. Omprakash Inani |
Mr. Omprakash Inani – CHAIRMAN
Mr. Omprakash Inani has more than 30 years of Business experience. He monitors business and functional aspects of the Company along with the operations of all the plants. Additionally, he is member of Audit and Remuneration committee of Shilpa Medicare Group of Companies. Currently he is also a council Member in “Academy of Medical Education, Dental College & V.L. College of Pharmacy”, “Taranath Shikshana Samsthe, Raichur” and a trustee in “Akhil Bhartiya Maheshwari Education Trust, Pune”. Mr. Omprakash Inani is also Managing Committee Member of “Karnataka State Cotton Assn., Hubli”. |
|
|
|
||
 Mr. Vishnukant C. Bhutada Mr. Vishnukant C. Bhutada |
Mr. Vishnukant C. Bhutada – MANAGING DIRECTOR
Mr. Vishnukant has vast and diverse Business experience of API and Intermediates and presently leads the core Business and functional teams which accelerate growth and performance by Innovating for Affordable solutions at Shilpa Medicare Group of Companies. He is the key decision maker with the teams for Shilpa Group for successful API and Generics formulation strategies. His untiring efforts have led the company to a leadership position in the Indian pharmaceutical domain and helped create a prominent presence for Oncology APIs globally. For his efforts on APIs Business, Mr. Vishnukant was awarded “Best Entrepreneur Award” by Late Dr Shankar Dayal Sharma – President of India in 1995. Subsequently, various state honours were conferred upon him -like -“Best Entrepreneur” from Karnataka State Govt. in 1996; “Excellence in Exports” from Vishweshwarayya Industrial Trade Centre, Bangalore 1996; and Export Excellence Award-2006” by FKCCI, Bangalore. Success has never stopped coming his way- as he was awarded “First runner up” at the Emerging India Awards London 2008 by CNBC TV18. Recently, his efforts in the Shilpa Group for environment sustainability, has led to “Best National Energy Conservation Award in Drugs & Pharmaceutical Sector for the year 2012” by Hon’ble President of India, Dr. Pranab Mukherjee. |
|
|
|
||
 Dr. Vimal Kumar Shrawat Dr. Vimal Kumar Shrawat |
Dr. Vimal Kumar Shrawat – CHIEF OPERATING OFFICER
Dr. Shrawat by qualification holds degrees of M.Sc (Organic Chemistry), Ph.D. (from Delhi University) and joined Shilpa Medicare in 2009. He has vast experience of more than 25 years of working in large pharma industries like Ranbaxy/ Dabur Pharma- presently known as Fresenius Kabi Oncology Ltd., spanning across activities of R&D, Pilot and Plant Productions, QA/QC, Administration, CRAMS, Project management etc. Presently, Dr. Shrawat is spearheading the entire Operations/ Control of Shilpa Medicare. His vision of team work and time bound approach always guides and motivates teams at all operational sites. His keen interest and consistent efforts for R&D has led him to become one of key contributor in large number of Patent/applications of Shilpa Medicare. |
|
|
|
||
 Dr. Pramod Kumar |
Dr. Pramod Kumar – MANAGING DIRECTOR(LOBA FEINCHEMIE GMBH AUSTRIA), SENIOR VICE-PRESIDENT (SHILPA MEDICARE LTD)
Dr. Pramod Kumar, who by qualification holds degrees of M.Pharm, Ph.D (Pharmaceutical chemistry) and a PGDBA, joined Shilpa Medicare in 1989. Since 2009 he is Managing Director of Loba FeinchemieGmBH, Austria and driving all R&D driven commercial processes. Dr. Pramod Kumar has more than 25 years of experience in Pharmaceutical industry, spanning across activities of production, QA/QC, administration, import/export, CRAMS etc. His efforts in CRAMS have led to the formation of Joint venture company RAICHEM MEDICARE Pvt LTD with Italian companies ICE SPA / P.C.A SPA. |
|
|
|
||
 Mr. Prashant Purohit |
Mr. Prashant Purohit – VICE-PRESIDENT-CRD
Mr. Prashant Purohit by qualification holds degrees of, M.Sc.(Organic Chemistry) and Diploma in Business Management and joined Shilpa Medicare in 1996. He is presently heading Chemical R&D wings of Shilpa Medicare Group. He has vast experience of handling CRAMS and Generics APIs R&D. His vast experience of nearly 35 years in R & D and production in Pharmaceutical Industry has consistently enriched the portfolio of Shilpa Medicare Group of Companies. He is one of key contributor in large number of Patent/applications of Shilpa Medicare. |
|
|
|
||
 Dr. Akshay Kant Chaturvedi |
Dr. Akshay Kant Chaturvedi – HEAD- CORPORATE IPM & LEGAL AFFAIRS
Dr. Akshay Kant by qualification holds degrees of M.Sc, Organic Chemistry (Univ. Gold Medalist), Ph.D. (Medicinal Chem), LL.B., M.B.A. and joined Shilpa Medicare in Jun 2012. Presently, Dr. Akshay is spearheading the entire IP portfolio management/ Legal Affairs of Contractual Business of Shilpa Medicare Group. His vision of innovative and creative thinking, team work and time bound approach always guide and motivate teams at all locations.His keen interest and consistent efforts for R&D has led him to become one of key contributor in large number of Patent/applications of Shilpa Medicare. |
|
|
|
||
 Dr. Seshachalam U. |
Dr. Seshachalam U. -ASSOCIATE VICEPRESIDENT- QUALITY AND RA
Dr. Seshachalam by qualification holds M.Sc (Chemistry) and Ph.D. (Chemistry) and joined Shilpa Medicare in 2008. He is presently heading Regulatory Affairs wings of Shilpa Medicare Group of Companies. He has vast experience of handling regulatory affairs related to Generics APIs. Being instrumental in Shilpa Medicare’s efforts to achieve recognition of different authorities, his key contribution in successful inspection and audit by various regulatory authorities is one of the core strength to the organization’s aims and objectives. |
|
|
|
||
 Mr. Sharath Reddy |
Mr. Sharath Reddy – VICE-PRESIDENT PROJECTS & OPERATIONS
Mr. Sharath Reddy by qualification holds M.Pharm from BITS Pilani and has overall experience of about 22 years predominately in the field of pharmaceuticals new projects and operations. His expertise of Oncology specialized equipment and Utilities designing has boosted organizations confidence to takeover new endeavors of upcoming projects with faster pace of time. His efforts have led to successfully executing Energy Saving projects of Shilpa Medicare Group of Companies and registration of the project under Clean Development Mechanism with UNFCC (Under Kyoto Protocol). |
|
|
|
||
 Mr. R K Somani |
Mr. R K Somani – VICE-PRESIDENT FORMULATION -BUSINESS DEVELOPMENT
Mr. R. K. Somani is a professional Chartered Accountant and holds a Diploma in Central Excise.He has overall business experience of more than 21 years predominately in the field of pharmaceuticals. Mr. Somani is one of the key drivers of Formulation business besides handling various key Contract Businesses of advanced oncology/ Non-Oncology APIs. He is known for successfully building formulations portfolio and spearheading the Generic sales operation. |
|
Shilpa Medicare Limited
1st Floor, 10/80,
Rajendra Gunj,
RAICHUR ರಾಯಚೂರು – 584 102.
Karnataka, India.
Telephone: +91-8532-236494
Fax: +91-8532-235876
Email: info@vbshilpa.com
RAICHUR, ರಾಯಚೂರು Karnataka, India







Historical Stone Elephants in Malayabad, Raichur Taluk …
View of Raichur city and lake Aam Talab
///Regorafenib, SHILPA MEDICARE LIMITED, new patent, WO 2016005874, raichur, ರಾಯಚೂರು , karnataka, india
FDA´s Emerging Technology Applications Program – Draft Guidance
![]()
FDA´s Emerging Technology Applications Program – Draft Guidance
The FDA recently published a draft guidance for industry on the “Advancement of Emerging Technology Applications”. The draft guidance provides recommendations to pharmaceutical companies interested in participating in a program involving the submission of CMC information containing emerging manufacturing (including testing, packaging and labeling, and quality control) technology to FDA. Find out more about the draft guidance for industry “Advancement of Emerging Technology Applications to Modernize the Pharmaceutical Manufacturing Base“..
On December 23, 2015, the FDA published a draft guidance for industry “Advancement of Emerging Technology Applications to Modernize the Pharmaceutical Manufacturing Base“. Comments and suggestions regarding this draft document should be submitted within 60 days of publication.
The draft guidance provides recommendations to pharmaceutical companies interested in participating in a program involving the submission of CMC (chemistry, manufacturing, and controls) information containing emerging manufacturing (including testing, packaging and labeling operations, and quality control) technology to FDA.
The program is open for new drug applications (INDs), original or supplemental new drug application (NDA), abbreviated new drug application (ANDA), or biologic license application (BLA). It only affects the quality section of a submission (CMC and facility-related information).
The development of emerging manufacturing technology, like, for example, aseptic manufacturing facilities with highly automated systems and isolators, may lead to improved manufacturing, and therefore improved product quality and availability throughout a product´s lifecycle.
Pharmaceutical companies can submit questions and proposals about the use of these technologies to a group within CDER (Emerging Technology Team – ETT).
The draft guidance is a follow-on to the FDA guidance for industry “PAT – A Framework for Innovative Pharmaceutical Development, Manufacturing, and Quality Assurance” which describes the concept that quality cannot be tested into products. It should be built-in or should be present by design. Through the ETT, FDA intends to encourage the adoption of innovative approaches by leveraging existing resources of FDA to facilitate regulatory reviews of submissions.
Examples of emerging technology elements include an innovative or novel:
- Product manufacturing technology, such as the dosage form;
- Manufacturing process (e.g., design, scale-up, and/or commercial scale);
- Testing technology.
Interested parties should submit a written meeting request to participate in the ETT program at least three months prior to the planned application (IND, ANDA, BLA, NDA) submission date. In addition to the items outlined in the FDA guidance “Formal Meetings Between the FDA and Sponsors or Applicants” the request should also include the following items:
- A brief description of the proposed testing, process, and/or proposed technology;
- A brief explanation why the proposed testing, process, and/or technology are substantially novel and unique;
- A description of how the proposed testing and/or technology could modernize pharmaceutical manufacturing and thus improve product safety, identity, strength, quality, or purity;
- A summary of the development plan and any perceived roadblocks to technical or regulatory implementation;
- A timeline for submission.
The request should generally not exceed five pages and FDA expects to notify companies of its decision regarding acceptance into the program within 60 days of receipt of the request. Once accepted into the program, the participant can engage with ETT and CMC in accordance with existing meeting procedures and guidances (e.g. above mentioned FDA guidance on Formal Meetings).
For further information, please find all the details in the draft guidance “Advancement of Emerging Technology Applications to Modernize the Pharmaceutical Manufacturing Base“.
Lupin Ltd, Patent, Pitavastatin, WO2014203045

Lupin Ltd, Patent, Pitavastatin, WO2014203045
A NOVEL, GREEN AND COST EFFECTIVE PROCESS FOR SYNTHESIS OF TERT-BUTYL (3R,5S)-6-OXO-3,5-DIHYDROXY-3,5-O-ISOPROPYLIDENE-HEXANOATE
ROY, Bhairabnath; (IN).
SINGH, Girij, Pal; (IN).
LATHI, Piyush, Suresh; (IN).
AGRAWAL, Manoj, Kunjabihari; (IN).
MITRA, Rangan; (IN).
TRIVEDI, Anurag; (IN).
PISE, Vijay, Sadashiv; (IN).
RUPANWAR, Manoj; (IN)
The present invention describes an eco-friendly and cost effective process for the synthesis of teri-butyl (3R,5S)-6-oxo-3,5-dihydroxy-3,5-0-isopropylidene-hexanoate [I]
 PITAVASTATIN
PITAVASTATIN

TEXT
tert-b tyl (3R,5S)-6-oxo-3,5-dihydroxy-3,5-0-isopropylidene-hexanoate [I] [CAS No. 124752-23-4] is key intermediate for the preparation of statins such as Atorvastatin (Tetrahedron 63, 2007, 8124 -8134), Cerivastatin (Journal of Labeled Compounds and Radiopharmaceuticals, 49, 2006 311-319), Fluvastatin [WO2007125547; US 4739073], Pitavastatin [WO2007/132482; US2012/22102 Al, WO2010/77062 A2; WO2012/63254 Al ; EP 304063; Tetrahedron Letters, 1993, 34, 513 – 516; Bulletin of the Chemical Society of Japan, 1995, 68, 364 – 372] and Rosuvastatin [WO2007/125547 A2; WO2011/132172 Al ; EP 521471]. Statins are used for treatment of hypercholesterolemia, which reduces the LDL cholesterol levels by inhibiting activity of HMG-CoA reductase enzyme, which is involved in the synthesis of cholesterol in liver.

[I]
Compound [I] is generally obtained by various methods of oxidation of teri-butyl 2- ((4R,65)-6-(hydroxymethyl)-2,2-dimethyl-l,3-dioxan-4-yl)acetate [compound II] and are discussed in details hereinafter. In addition, various methods for synthesis of compound [II] are also elaborated below.

[II]
[II]
A) tert-butyl2-((4«,6.S)-6-(hydroxymethyl)-2,2-dimethyl-l,3-dioxan-4-yl)acetate
[compound II]
US patent Number 5278313 describes a process for synthesis of compound [II]
(Schemel). In the said process, (5)-methyl 4-chloro-3-hydroxybutanoate has been obtained in only 70% yield through whole cell enzymatic reduction of methyl 4-chloro-3- oxobutanoate, which has a necessity of special equipment such as fermenters as well as other microbial facilities such as sterile area, autoclaves, incubator for growing seed culture, etc.
(S)-mefhyl 4-chloro-3-hydroxybutanoate upon reaction with teri-butyl acetate in presence of LiHMDS or LDA at -78°C, yielded (S)-ieri-butyl 6-chloro-5-hydroxy-3- oxohexanoate, which was further transformed to corresponding diol through syn selective reduction in presence of methoxydiethyl borane/sodium borohydride at -78°C. The diol thus obtained was converted to compound [II] .
The overall yield for this process is low and required special equipment such as fermenters, etc and in addition to that, this process is not cost effective due to use of costly reagent such as methoxydiethyl borane.
Moreover, methoxydiethylborane is highly pyrophoric (Encyclopedia for organic synthesis, editor in chief L. Paquette; 2, 5304; Published by John and Wiley Sons;
Organic Process Research & Development 2006, 10, 1292-1295) and hence safety is a major concern.

Scheme 1
EP 1282719 B l (PCT application WO 01/85975 Al ) discloses a process for synthesis of compound ( R, 5S)-tert-bv y\ 3,5,6-trihydroxyhexanoate from (S)-tert-b tyl-5,6-dihydroxy-3-oxohexanoate through a) asymmetric hydrogenation in presence of a chiral catalyst e.g. di-mu-chlorobis-[(p-cymene)chlororuthenium(II)] along with an auxiliary such as (IS, 2S)-(+)-N- (4-toluenesulfonyl)-l ,2-diphenylethylenediamine as ligand, which gave desired product only in 70% diastereomeric excess (de); b) Whole cell enzymatic reduction of (S)-tert- butyl 5,6-dihydroxy-3-oxohexanoate to obtain compound (3R, 5S)-tert-bv y\ 3,5,6-trihydroxyhexanoate in 99% de (80% yield).
It is needless to mention that it has necessity of fermenter and other microbiological equipment (Scheme 2).
Moreover, conversion of (2>R,5S)-tert-bv y\ 6-acetoxy-3,5-dihydroxyhexanoate to tert-bv yl 2-((4R,65)-6-(acetoxymethyl)-2,2-dimethyl-l ,3-dioxan-4-yl)acetate was accomplished in only 25% yield and also required the flash chromatography for isolation of desired product.
Thus, overall yield for this process is poor and process is not operation friendly especially at large scale hence cannot be considered feasible for commercial manufacturing.

Scheme 2
EP1317440 Bl (PCT Application WO 02/06266 Al) has disclosed the process for synthesis of compound [II] from 6-chloro-2,4,6-trideoxy-D-erythro-hexose (Scheme 3) .
In the said patent application 6-chloro-2,4,6-trideoxy-D-erythro-hexose was converted to (4R, 65)-4-hydroxy-6-chloromethyl-tetrahydropyran-2one with excess of bromine in presence of potassium bicarbonate, which liberates environmentally undesired gas i.e. carbon dioxide.
Moreover, starting material i.e. 6-chloro-2,4,6-trideoxy-D-erythro-hexose is not commercially available and conversion efficiency of starting material at large scale towards (4R, 65)-4-hydroxy-6-chloromethyl-tetrahydropyran-2-one is only 67%.

Scheme 3
US Patent No. 6689591 B2 has demonstrated the whole cell enzymatic reduction of teri-butyl 6-chloro-3,5-dioxohexanoate to compound [II] (Scheme 4).
In the said process, whole cell enzymatic reduction is not specific; yield for desired product is only 34% and other partially reduced products are also obtained.
Hence, further purification is required for obtaining the desired compound. Thus, this process is not suitable for commercial scale.

Scheme 4
Tatsuya et al (Tetrahedron Letters; 34, 1993,513 – 516) has reported synthesis of compound [I] from derivative of L-tartatric acid (Scheme 5).
In the said process, tartaric acid di-i‘sopropyl ester is doubly protected by tert-butyldimethylsilyl group, which was reacted with dianion of teri-butyl acetoacetate to give β, δ-diketo ester compound.
β,δ-diketo ester was reacted with 2 equivalent of diisobutylaluminium hydride (which is a pyrophoric reagent) to afford -hydroxy,8-keto ester in only 60% yield.
This process is not industrially viable as overall yield is very low and also because of use of costly and pyrophoric reagents/chemicals.

Scheme 5
US7205418 (PCT application WO03/053950A1) has described the process for synthesis of compound [II] from (S)-ieri-butyl-3,4-epoxybutanoate (Scheme 6).
The overall yield for this process is very low and moreover, it required the diastereomeric separation of teri-butyl 2-(6-(iodomethyl)-2-oxo-l,3-dioxan-4-yl)acetate by flash chromatography.
Since overall requirement of title compound is very high, any operation involving flash chromatography will tend to render the process commercially unviable.

Scheme 6
Fengali et al (Tetrahedron: Asymmetry 17; 2006; 2907-2913) has reported the process for synthesis of compound [II] from racemic epichlorohydrin (Scheme 7).
In this process, racemic epichlorohydrin was converted to corresponding nitrile intermediate through reaction with sodium cyanide; nitrile intermediate thus obtained was further resolved through lipase catalyzed stereo-selective esterification to obtain (5)-4-(benzyloxy)-3-hydroxybutanenitrile and (R)-l-(benzyloxy)-3-cyanopropan-2-yl acetate;
separation of desired product i.e. (S)-4-(benzyloxy)-3-hydroxybutanenitrile having 98% de (40% yield) was done by column chromatography.
Needless to mention a commodity chemical like compound [I] cannot be manufactured by such a laboratory method, which involved number of steps.

Scheme 7
Bode et al (Organic letters, 2002, 4, 619-621) has reported diastereomer- specific hydrolysis of 1,3-diol-acetonides (Scheme 8).
In this publication, duration of the reaction for diastereomer- specific hydrolysis of 1,3, diol-acetonides is reported to be 4 h, however, in our hand it was observed that hardly any reaction took place in 4 h, which made it non-reproducible.
In addition to that, separation of desired product is achieved by flash chromatography and it is needless to mention that any process which involved flash chromatography would render the process to be commercially unviable.
Hence, additional innovation needs to be put in for making the process industrially viable.

Scheme 8
CN 101613341A has reported the process for synthesis of compound [II] (Scheme
9).
In the same patent application tert-b tyl (S)-6-chloro-5-hydroxy-3-oxohexanoate was synthesized through Blaise condensation of (5)-4-chloro-3-hydorxy-butanenitrile with zinc enolate of tert butyl bromo acetate.
In the literature, synthesis of tert-bv yl (S)-6-chloro-5-hydroxy-3-oxohexanoate was reported through Blaise condensation of silyl protected (5)-4-chloro-3-(trimethylsilyl)oxy-butanenitrile with zinc enolate of tert butyl bromo acetate, in good yield (Synthesis 2004, 16, 2629-2632). Thus, protection of hydroxy group in (5)-4-chloro-3-hydorxy-butanenitrile is imperative.
In the said Chinese patent application, in claim 7, it was mentioned that solvent used for conversion of tert-bv yl (5)-6-chloro-5-hydroxy-3-oxohexanoate to ( R,5S)-tert-butyl 6-chloro-3,5-dihydroxyhexanoate is anyone or mixture of more than one from tetrahydrofuran, ether, methanol, ethanol, n-propanol, /so-propanol and ethylene glycol.
However, in enablement the only example using mixture of solvent was that of THF-methanol (Experimental section, Example 4: The preparation of (R,5)-6-chloro-3,5- dihydroxyhexanoate) and same outcome was expected in other individual or mixture of solvents.
Claim 8 of CN 101613341A mentioned that reduction was carried out by any one or mixture of more than one reducing agents such as sodium borohydride, potassium borohydride, lithium aluminum hydride, diethylmethoxy borane, triethyl borane and tributyl borane.
It implies that either any one of the reducing agents or a mixture of the same can be employed. From reaction mechanism it is very much clear that diethylmethoxy borane, triethyl borane and tributyl borane form the six membered complex between optically active hydroxyl and carbonyl group, which gets reduced by sodium borohydride, signifying that individually diethylmethoxy borane, triethyl borane and tributyl borane are not reducing agents
Moreover, in claims 12 and 13 (Experimental section, Example 4: The preparation of (R,S)-6-chloro-3,5-dihydroxyhexanoate), it is mentioned that reduction should be carried out in temperature range -80 °C to -60 °C, implying that reaction would not work beyond this temperature range i.e. it would work in the temperature window of -80 °C to -60 °C only.
Summarizing, the teachings of the application are not workable.

Scheme 9
Wolberg et al (Angewandte Chemie International Edition, 2000, 4306) has reported that diastereomeric excess for syn selective reduction using mixture of diethyl methoxy borane/sodium borohydride of compound [VI] gave 93% de for compound [VIII], which required further re-crystallization to obtain compound [VIII] in 99% de and 70% yield.
Thus, all the reported methods for stereo-selective hydride reduction of compound [VI] were achieved through mixture of trialkyl borane or diethyl methoxy borane & sodium borohydride in THF, at -78°C. As mentioned earlier, trialkyl borane or diethyl methoxy borane are pyrophoric in nature; in addition to that anhydrous THF is costly and moreover, reaction required large dilution.
Hence, there is need for developing efficient, environment friendly, cost effective and green process for stereo-selective reduction compound [VI].
B) The process of Oxidation of compound [II] to compound [I] has been discussed in following literature processes.
1) Swern oxidation (US4970313; Tetrahedron Letters, 1990, 2545
Synthetic Communications, 2003, 2275 – 2284).
2) Parrkh-Doering oxidation (J. Am. Chem. Soc, 1967, 89, 5505-5507)
3) TEMPO/NaOCl oxidization (EP2351762)
4) Trichloroisocyanuric acid/ TEMPO (CN 101747313A)
5) Oxidation of compound [II] to compound [I] through IBX [CN101475558A].
It would be evident that most of the reported methods are not “green” and
environmentally benign; none of the reported methods use molecular oxygen as oxidizing agent in presence of metal catalyst/co-catalyst.
Example 18: Process for synthesis of tert-butyl 2-((4R,6S)-6-formyl-2,2-dimethyl-l,3-dioxan-4-yl)acetate [I]

A reactor was charged with 1.1 g of copper (I) chloride and 10 mL of acetonitrile. 2-2′ Bipyridyl (156 mg) and TEMPO (156 mg) were added to the reactor under oxygen environment at 25°C. A solution of (6-Hydroxymethyl-2,2-dimethyl-[l,3]dioxan-4-yl)-acetic acid tert-butyl ester 2.6 g in 26 mL DCM was added dropwise over a period of 10 min into it. The reaction mass was stirred at 40°C and progress of reaction was monitored on GLC, which shows that 90% conversion for desired product.
Example 19: Process for synthesis of tert-butyl 2-((4R,6S)-6-formyl-2,2-dimethyl-l,3-dioxan-4-yl)acetate [I]
A reactor was charged with 1.1 g of copper (I) chloride and 10 mL of dichlorome thane. 2-2′ Bipyridyl (156 mg) and TEMPO (156 mg) were added to the reactor under oxygen environment at 25°C. A solution of (6-Hydroxymethyl-2,2-dimethyl-[l,3]dioxan-4-yl)-acetic acid tert-butyl ester 2.6 g in 26 mL DCM was added dropwise over a period of 10 min into it. The reaction mass was stirred at 40°C and progress of reaction was monitored on GLC, which shows that 90% conversion for desired product.
AUTHORS
///////
Lupin Ltd, New patent, Pitavastatin, WO 2016005919

Formula (1)
Lupin Ltd, New patent, Pitavastatin, WO 2016005919
MANE, Narendra, Dattatray; (IN).
NEHATE, Sagar, Purushottam; (IN).
GODBOLE, Himanshu, Madhav; (IN).
SINGH, Girij, Pal; (IN)

The present invention is directed to polymorphic forms of Pitavastatin sodium and processes for preparation of the same
Novel crystalline polymorphic forms (I and II) and an amorphous form of pitavastatin, useful for treating hyperlipidemia and mixed dyslipidemia.
Also claims a method for preparing the crystalline and amorphous forms of pitavastatin. In January 2016, Newport Premium™ reported that Lupin holds an active US DMF for pitavastatin calcium since July 2013.
Nissan Chemical Industries and licensee Kowa, with sub-licensees Sankyo, Eli Lilly, Esteve, JW Pharmaceutical, Recordati, Laboratorios Delta and Zydus-Cadila, have developed and launched pitavastatin.
WO2014203045, claiming a process for preparing an intermediate useful in the synthesis of statins (eg pitavastatin).
Pitavastatin is a cholesterol lowering agent of the class of HMG-CoA reductase inhibitor. The HMG-CoA reductase enzyme catalyzes the conversions of HMG- CoA to mevalonate. Inhibitors of HMG-CoA reductase are commonly referred to as “statins.” Statins are therapeutically effective drugs used for reducing low density lipoprotein (LDL) particle concentration in the blood stream of patients at risk for cardiovascular disease.
Pitavastatin is one of the synthetic statins which is chemically known as (3R, 5S, 6E)-7-[2-cyclopropyl-4-(4-fluorophenyl) quinoline-3-yl]-3, 5-dihydroxy-6- heptenoic acid represented by structural formula (1):

Formula (1)
Pitavastatin and its pharmaceutically acceptable salts are described in US 5,753,675 patent and US 5,856,336 patent, respectively.
Processes for the preparation of Pitavastatin are well documented in the literature. European patents, EP 0304063 and EP 1099694 and reports by Miyachi et al (Tetrahedron Letters
(1993) vol. 34, pages 8267-8270) and Takahashi et al (Bull. Chem. Soc. Japan (1995) Vol. 68, 2649-2656) describe processes for preparation of Pitavastatin.
US 5,872,130 patent discloses sodium salt of Pitavastatin. This patent, however, is silent about the solid state form of Pitavastatin Sodium.
It is generally known in the art that active pharmaceutical ingredients frequently do not exhibit the range of physical properties that makes them directly suitable for development. One of the approaches that is used to modify the characteristics of drug substances is to employ a salt form of the substance, since salts enable one to modify aqueous solubility, dissolution rate, solution pH, crystal form, hygroscopicity, chemical stability, melting point and even mechanical properties. The beneficial aspects of using salt forms of active pharmaceutical ingredients are well known and represent one of the means to increase the degree of solubility of otherwise intractable substances and to increase bioavailability.
Although the known salts of Pitavastatin like sodium, potassium, magnesium, calcium etc. and their polymorphic forms may address some of the deficiencies in terms of formulated product and its manufacturability. There remains a need for yet further improvement in these properties as well as improvements in other properties such as flowability, and solubility.
Polymorphism is a known phenomenon among pharmaceutical substances. It is commonly defined as the ability of any substance to exist in two or more crystalline phases that have a different arrangement and/or conformation of the molecules in the crystal lattice. Different polymorphic forms of the same pharmaceutically active moiety also differ in their physical properties such as melting point, solubility, chemical reactivity, etc. These properties may also appreciably influence pharmaceutical properties such as dissolution rate and bioavailability.
Further, the discovery of new polymorphic forms and solvates of an active pharmaceutical ingredient provides broader scope to a formulation scientist for formulation optimization, for example by providing a product with different properties, e.g., better processing or handling characteristics, improved dissolution profile, or improved shelf-life. For at least these reasons, there is a need for polymorphs of Pitavastatin salts such as Pitavastatin sodium.
New polymorphic forms and hydrates and/or solvates of a pharmaceutically acceptable salt of Pitavastatin can also provide an opportunity to improve the performance characteristics of a pharmaceutical product.
Therefore, there is a scope to prepare novel polymorphic forms of Pitavastatin sodium and hydrates and/or solvates.
Example-1: Preparation of Pitavastatin Sodium (Form-I)
A mixture of 40.0 gm Pitavastatin acid and 120 ml water was cooled to 15-20 °C temperature. Thereafter aqueous solution of sodium hydroxide (4.0 gm) in water (20 ml) was added to the reaction mixture. The reaction mixture was stirred for 30-45 min at 15-20 °C temperature. Ethyl acetate (80ml) was added into the reaction mixture at 15-20 °C temperature, stirred for 15-20 min and the layers were separated. The aqueous layer was filtered and acetonitrile (1200 ml) was gradually added to the aqueous layer under stirring till the precipitation was completed. The reaction mixture was cooled to 5-8 °C temperature and stirred for 2-3 hours at 5-8 °C temperature. The precipitated solid was filtered, washed with acetonitrile (40ml) and dried at 45-50 °C temperature under vacuum for 10-12 hours to afford the title compound (28.0 gm).
Yield (w/w): 0.70 (66.0%)
HPLC purity: 99.70 %
Example-2: Preparation of Pitavastatin Sodium (Form-II)
A mixture of 40.0 gm of Pitavastatin acid and 120 ml of water was cooled to 15-20°C temperature under stirring. Thereafter aqueous solution of sodium hydroxide (4.0 gm) in water (20 ml) was added to the reaction mixture. The reaction mixture was stirred for 30-45 min at 15-20 °C temperature. Ethyl acetate (80ml) was added to the reaction mixture at 15-20 °C temperature, stirred for 15-20 min and the layers were separated. The aqueous layer was filtered and acetonitrile (1200 ml) was gradually added to the aqueous layer under stirring till the precipitation was completed. The reaction mixture was cooled to 5-8 °C temperature and stirred for 2-3 hours at 5-8 °C temperature. The precipitated solid was filtered, washed with acetonitrile (40ml) and dried at 45-50 °C temperature under vacuum for 10-12 hours and kept in a petri dish at 25-30 °C and 60 ± 5 RH (relative humidity) for 18-24 hours to afford the title compound (31.6 gm).
Yield (w/w): 0.79 (65.8%)
HPLC purity: 99.70 %
Example-3: Preparation of Pitavastatin Sodium Amorphous
Pitavastatin sodium (3.0 gm) and ethanol (60 ml) were taken in a round bottomed flask at 25-30 °C temperature. The reaction mixture was filtered and the solvent was distilled off on rotatory evaporator under vacuum maintaining bath temperature at 45-50 °C temperature. Thereafter the reaction mixture was degassed under vacuum for 2-3 hours to afford the title compound (2.8gm).
HPLC purity: 99.70 %.
SEE……..https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2016005919&redirectedID=true
/////////Lupin Ltd, New patent, Pitavastatin, WO 2016005919, statins, POLYMORPH
Dr Reddy’s Laboratories Ltd, New patent, WO 2016005960, Liraglutide

!e™A!a™Trp™leu™Va!~-Arg~~GIy-~Arg~~Gly~~OH
Formula (I)
LIRAGLUTIDE

Dr Reddy’s Laboratories Ltd, New patent, WO 2016005960, Liraglutide
Process for preparation of liraglutide
Kola, Lavanya; Ramasamy, Karthik; Thakur, Rajiv Vishnukant; Katkam, Srinivas; Komaravolu, Yagna Kiran Kumar; Nandivada, Giri Babu; Gandavadi, Sunil Kumar; Nariyam Munaswamy, Sekhar; Movva, Kishore Kumar
Improved process for preparing liraglutide, by solid phase synthesis, useful for treating type 2 diabetes.
It having been developed and launched by Novo Nordisk, under license from Scios and Massachusetts General Hospital.
Liraglutide, marketed under the brand name Victoza, is a long-acting glucagon like peptide agonist developed by Novo Nordisk for the treatment of type 2 diabetes.
Liraglutide is an injectable drug that reduces the level of sugar (glucose) in the blood. It is used for treating type 2 diabetes and is similar to exenatide (Byetta). Liraglutide belongs to a class of drugs called incretin mimetics because these drugs mimic the effects of incretins. Incretins, such as human-glucagon-like peptide-1 (GLP-1 ), are hormones that are produced and released into the blood by the intestine in response to food. GLP-1 increases the secretion of insulin from the pancreas, slows absorption of glucose from the gut, and reduces the action of glucagon. (Glucagon is a hormone that increases glucose production by the liver.)
All three of these actions reduce levels of glucose in the blood. In addition, GLP-1 reduces appetite. Liraglutide is a synthetic (man-made) hormone that resembles and acts like GLP-1 . In studies, Liraglutide treated patients achieved lower blood glucose levels and experienced weight loss.
Liraglutide, an analog of human GLP-1 acts as a GLP-1 receptor agonist. The peptide precursor of Liraglutide, produced by a process that includes expression of recombinant DNA in Saccharomyces cerevisiae, has been engineered to be 97% homologous to native human GLP-1 by substituting arginine for lysine at position 34. Liraglutide is made by attaching a C-16 fatty acid (palmitic acid) with a glutamic acid spacer on the remaining lysine residue at position 26 of the peptide precursor.
The molecular formula of Liraglutide is Ci72H265N4305i and the molecular weight is 3751 .2 Daltons. It is represented by the structure of formula (I)

!e™A!a™Trp™leu™Va!~-Arg~~GIy-~Arg~~Gly~~OH
Formula (I)
U.S. Patent No. 7572884 discloses a process for preparing Liraglutide by recombinant technology followed by acylation and removal of N-terminal extension.
U.S. Patent No. 7273921 and 6451974 discloses a process for acylation of Arg-34GLP-1 (7-37) to obtain Liraglutide.
U.S. Patent No. 8445433 discloses a solid phase synthesis of Liraglutide using a fragment approach.
International Application publication No. WO2013037266A1 discloses solid phase synthesis of Liraglutide, characterized in that comprises A) the presence of the activator system, solid phase carrier and by resin Fmoc protection N end obtained by coupling of glycine (Fmoc-Gly-OH) Fmoc-Gly-resin; B) by solid phase synthesis, prepared in accordance with the sequentially advantage Liraglutide principal chain N end of the coupling with Fmoc protected amino acid side chain protection and, wherein the lysine using Fmoc-Lys (Alloc)-OH; C) Alloc getting rid of the lysine side chain protecting group; D) by solid phase synthesis, the lysine side chain coupling Palmitoyl-Glu-OtBu; E) cracking, get rid of protecting group and resin to obtain crude Liraglutide ; F) purification, freeze-dried, to obtain Liraglutide.
Even though, the above mentioned prior art discloses diverse processes for the preparation of Liraglutide, they are often not amenable on commercial scale because of expensive amino acid derivatives such as pseudo prolines used in those processes.
Hence, there remains a need to provide simple, cost effective, scalable and robust processes for the preparation of Liraglutide involving commercially viable amino acid derivatives and reagents.

EXAMPLE 1 :
Stage I Preparation of Wang resin-Gly-Arg(pbf)-Gly-Arg(pbf)-Val-Leu-Trp(Boc)-Ala-lleu-Phe-Glu(Otbu)-Lys-{Glu(OH)-NH(palmitoyl)}-Ala-Ala-Gln(trt)-Gly-OH-Glu(Otbu)-Leu-Tyr(Otbu)-Ser(Otbu)-Ser(Otbu)-Val-Asp(Otbu)-Ser(Otbu)-Thr(Otbu)-Phe-Thr(Otbu)-Gly-Glu(Otbu)-Ala-Boc-His(trt)-OH.
Wang resin (50gm) is swelled in DCM (500ml) for 1 hr in a sintered flask. DCM was filtered using Vacuum. Fmoc-Glycine (44.6 gm, 150 mmol) was dissolved in dichloromethane (250 ml). 1 -(2-mesitylene sulfonyl)-3-nitro-1 H-1 ,2,4 triazole (44.4 gm, 150 mmol) and 1 -methyl imidazole (9 ml, 1 12 mmol) was then added. The reaction mixture was added to wang resin and stirred for 3hrs at about 25° C. The resin was washed with DCM and a second lot of Fmoc-Glycine (27 gm, 90 mmol) was dissolved in dichloromethane (250 ml). 1 -(2-mesitylene sulfonyl)-3-nitro-1 H-1 ,2,4 triazole (26.6 gm, 90 mmol) and 1 -methyl imidazole (5.3 ml, 90 mmol) was then added and stirred for 3hrs. The resin was washed with DCM and a sample of resin beads were checked for UV analysis. The capping was carried out using acetic anhydride (15 ml) DCM (120 ml) and pyridine (120 ml). The resin was washed with dichloromethane and DMF. The Fmoc protecting group was removed by treatment with 20% piperidine in DMF. The
resin was washed repeatedly with DMF. The next amino acid Fmoc-Arg(pbf)-OH (52 gm, 80 mmol) dissolved in 250 ml DMF was then added. The coupling was carried out by addition of HOBt (10.8gm, 80 mmol) and DIC (6.2ml, 80 mmol) in DMF. The completion of the coupling was confirmed by a ninhydrin test. After washing the resin, the Fmoc protecting group was removed with 20% piperidine in DMF. These steps were repeated each time with the respective amino acid according to the peptide sequence. After coupling 12th amino acid Fmoc-Lys (Alloc)-OH, deprotection of alloc group is carried out with palladium tetrakis and phenyl silane in DCM. The resin was washed repeatedly with DMF. The next amino acid H-Glu(OH)-NH(palmitoyl)-Otbu (9.9 gm, 0.023 moles) dissolved in 250 ml DMF was then added. The coupling was carried out by addition of HOBt (10.8gm, 80 mmol) and DIC (6.2ml, 80 mmol) in DMF. The completion of the coupling was confirmed by a ninhydrin test. After washing the resin, the Fmoc protecting group of Lys was removed with 20% piperidine in DMF. The next amino acid Fmoc-Ala-OH (52 gm, 80 mmol) dissolved in 250 ml DMF was then added. The coupling was carried out by addition of HOBt (10.8gm, 80 mmol) and DIC (6.2ml, 80 mmol) in DMF. The completion of the coupling was confirmed by a ninhydrin test. After washing the resin, the Fmoc protecting group was removed with 20% piperidine in DMF. These steps were repeated each time with the respective amino acid according to the peptide sequence. The resin was washed repeatedly with DMF, Methanol and MTBE and dried under vacuum.
Stage II: Cleavage of Liraglutide from resin along with global deprotection
45gms of resin obtained in stage I was treated with cleavage cocktail mixture of TFA (462.5ml), TIPS (12.5ml), Water (12.5ml), and Phenol (12.5 ml), stirred at 0°C for 30 min. and at 25°C for 3hrs at 200RPM. Then the reaction mixture was filtered, repeatedly wash the resin with TFA and the filtrate was concentrated on Rotary evaporator at 30°C. Pour the concentrated solution to MTBE (2L) at 4°C slowly and stir for 1 hr. The precipitate obtained is filtered and dried in a vacuum tray drier to afford 18 gm of Liraglutide crude with a purity of 27.5%.
Stage III: Purification of crude Liraglutide using RP HPLC.
The crude Liraglutide (4 gm) of purity around 27.5% is dissolved in 10 mM Tris buffer (120ml) of pH: 8.00 and 0.5 N NaOH is further added drop wise to the solution for making the crude solid completely dissolved. The solution is further passed through 0.2 micron filter. The Reverse phase C 18 – 150 Angstrom media (C18 silica media – 10 micron particle size) is equilibrated with 10mM Tris buffer of pH: 8.0 The crude solution is loaded onto the column and the gradient elution is performed as per the below tabular column against the mobile phase B (Acetonitrile).
Table 1 : Gradient program for pre purification

The desired fractions are collected in the gradient range of and the fractions (F1 , F2, F3, F4 and F5) whose purity > 80% are pooled. The pooled fractions are then subjected to further purification.
The Pooled fractions having purity >80% are then subjected to C18 RPHPLC silica media (5 micron particle size) for further purification. The pooled fractions – Feed is diluted with purified water in the ratio of 1 :2 (one part of pooled fraction to two parts of purified water) as a part of sample preparation before loading into the column. The media C18 is first equilibrated with 0.1 % TFA for 3 column volumes (1 CV = bed volume of media). After equilibration, the sample is loaded onto the column and the gradient
elution is performed as per the below tabular column against the mobile phase B (Acetonitrile).
Table 2: Gradient program for second purification

The desired fractions are collected in the gradient range of and the fraction whose purity > 96% are pooled together and lyophilized to afford 220mg of Liraglutide trifluoro acetate salt. The pooled fractions and their purity by HPLC are listed in the below table.

The pooled fractions with the purity of average 97% are subjected further to de solvation to remove the Acetonitrile content by Rota vapor. The final solution was filtered through 0.2 micron filter and lyophilized to get Liraglutide API.
EXAMPLE 2:
Stage I Preparation of Tentagel SPHB resin-Gly-Arg(pbf)-Gly-Arg(pbf)-Val-Leu-Trp(Boc)-Ala-lleu-Phe-Glu(Otbu)-Lys-{Glu(OH)-NH(palmitoyl)}-Ala-Ala-Gln(trt)-Gly-OH-Glu(Otbu)-Leu-Tyr(Otbu)-Ser(Otbu)-Ser(Otbu)-Val-Asp(Otbu)-Ser(Otbu)-Thr(Otbu)-Phe-Thr(Otbu)-Gly-Glu(Otbu)-Ala-Boc-His(trt)-OH using Fragment approach.
Fragments used are as follows
1 . Fmoc-Arg(pbf)-Gly-OH.
2. Fmoc-Leu-Ala-Arg(pbf)-OH.
3. Fmoc-lle-Ala-Trp(boc)-OH.
4. Fmoc-Glu(Otbu)-Phe-OH.
5. Fmoc-Glu(Otbu)-Phe-OH.
6. Fmoc-Lys-Glu-Palmitic acid.
7. Fmoc-Gly-Gln(trt)-Ala-Ala-OH.
8. Fmoc-Tyr(Otbu)-Leu-Glu(Otbu)-OH.
9. Fmoc-Val-Ser(Otbu)-Ser(Otbu)-OH.
10. Fmoc-Phe-Thr(Otbu)-Ser(Otbu)-Asp(Otbu)-OH
1 1 . Fmoc-Gly-Thr(Otbu)-OH.
12. Boc-His(Trt)-Ala-Glu(Otbu)-OH.
Tentagel SPHB resin (30gm) is swelled in DCM (300ml) for 1 hr in a sintered flask. DCM was filtered using Vacuum. Fmoc-Glycine (13.8 gm, 46.8 moles) was dissolved in dichloromethane (150 ml). 1 -(2-mesitylene sulfonyl)-3-nitro-1 H-1 ,2,4 triazole (13.8 gm, 46.8 moles) and 1 -methyl imidazole (2.4 ml, 29.25 moles) was then added. The resulting solution was added to tentagel resin and stirred for 2hrs at about 25° C. The resin was washed with DCM and a second lot of Fmoc-Glycine (13.8 gm, 46.8 moles) was dissolved in dichloromethane (150 ml). 1 -(2-mesitylene sulfonyl)-3-nitro-I H-1 ,2,4 triazole (13.8 gm, 46.8 moles) and 1 -methyl imidazole (2.4 ml, 29.25 moles) was then added and stirred for 2hrs. The resin was washed with DCM and a sample of resin beads were checked for UV analysis. The Fmoc protecting group was removed by treatment with 20% piperidine in DMF. The resin was washed repeatedly
with DMF. The next amino acid fragment 1 Fmoc-Gly-Arg(pbf)-OH (8.25 gm, 1 1 .7 moles) dissolved in 150 ml DMF was then added. The coupling was carried out by addition of HOBt (2.1 gm, 1 1 .7 moles) and DIC (2.5ml, 1 1 .7 moles) in DMF for 2hrs. The completion of the coupling was confirmed by a ninhydrin test. After washing the resin, the Fmoc protecting group was removed with 20% piperidine in DMF. These steps were repeated each time with the respective amino acid fragments according to the peptide sequence. The resin was washed repeatedly with DMF, Methanol and MTBE and dried under vacuum.
Stage II: Cleavage of Liraglutide from resin along with global deprotection
58gms of resin obtained from stage I was treated with cleavage cocktail mixture of TFA (555ml), TIPS (15ml), Water (15ml), and Phenol (15 ml) and stirred at 0°C for 30 min. at 25°C for 3hrs at 200RPM. Then filter the reaction mixture, repeatedly wash the resin with TFA and concentrate on Rotary evaporator at 30°C. Pour the concentrated solution to MTBE at 4°C slowly and stirred for 1 hr. The precipitate obtained was filtered and dried in a vacuum tray drier to afford 23.12 gm of crude Liraglutide with a purity of 36.89%.
Stage III: Purification of crude Liraglutide using RP HPLC.
The crude Liraglutide (4 gm) of purity around 27.5% is dissolved in 10 mM Tris buffer (120ml) of pH: 8.00 and 0.5 N NaOH is further added drop wise to the solution for making the crude solid completely dissolved. The solution is further passed through 0.2 micron filter. The Reverse phase C 18 – 150 Angstrom media (Irregular C18 silica media – 10 micron particle size) is equilibrated with 10mM Tris buffer of pH: 8.0 The crude solution is loaded onto the column and the gradient elution is performed as per the below tabular column against the mobile phase B (Acetonitrile).
Table 1 : Gradient program for pre purification

60 40 30
55 45 30
52 48 30
51 49 60
The desired fractions are collected in the gradient range of and the fractions (F1 , F2, F3, F4 and F5) whose purity > 80% are pooled. The pooled fractions then subjected to further purification.
The Pooled fractions having purity >80% are then subjected to C18 RPHPLC silica media (5 micron particle size) for further purification. The pooled fractions – Feed is diluted with purified water in the ratio of 1 :2 (one part of pooled fraction to two parts of purified water) as a part of sample preparation before loading into the column. The media C18 is first equilibrated with 0.1 % TFA for 3 column volumes (1 CV = bed volume of media). After equilibration, the sample is loaded onto the column and the gradient elution is performed as per the below tabular column against the mobile phase B (Acetonitrile).
Table 2: Gradient program for second purification

The desired fractions are collected in the gradient range and the fraction whose purity > 96% are pooled together and Lyophilized to afford 865 mg of Liraglutide trifluoro acetate salt. The pooled fractions and their purity by HPLC are listed in the below table.

The pooled fractions with the purity of average 97% are subjected further to de solvation to remove the Acetonitrile content by Rota vapor. The final solution was filtered through 0.2 micron filter and lyophilized to get Liraglutide API.

G.V. Prasad, chairman, Dr Reddy’s Laboratories.
REFERENCE
IN2014CH3453 INDIAN PATENT
WO 2016005960, CLICK FOR PATENT
//////
Breaking and Making of Olefins Simultaneously Using Ozonolysis: Application to the Synthesis of Useful Building Blocks and Macrocyclic Core of Solomonamides

A simple and practical one-pot, two-directional approach to access olefinic esters through simultaneous breaking and making of olefins using ozonolysis of alkenyl aryl selenides is disclosed. The scope of the method with a variety of examples is demonstrated, and the end products obtained here are useful building blocks. As a direct application of the present method, the macrocyclic core of potent anti-inflammatory natural cyclic peptides, solomonamides, is synthesized.
Breaking and Making of Olefins Simultaneously Using Ozonolysis: Application to the Synthesis of Useful Building Blocks and Macrocyclic Core of Solomonamides


Dr. D. Srinivasa Reddy
AT 9283

AT9283, AT 9283
N-cyclopropyl-N’-[3-[6-(4-morpholinylmethyl)-1H-benzimidazol-2-yl]-1H-pyrazol-4-yl]urea
1-cyclopropyl-3-[(3Z)-3-[5-(morpholin-4-ylmethyl)benzimidazol-2-ylidene]-1,2-dihydropyrazol-4-yl]urea
| 896466-04-9 | |
| Molecular Weight | 381.43 |
| Molecular Formula | C19H23N7O2 |
CAS
896466-04-9, 896466-57-2 ((±)-Lactic acid), 896466-61-8 (HCl), 896466-55-0 (methanesulfonate)
MolFormulaC22H29N7O5
MolWeight471.5096
CAS 896466-76-5 L LACTATE
(2S)-2-Hydroxypropanoic acid compd. with N-cyclopropyl-N’-[3-[6-(4-morpholinylmethyl)-1H-benzimidazol-2-yl]-1H-pyrazol-4-yl]urea
Astex Therapeutics Ltd, INNOVATOR

AT-9283 is a potent AuroraA/AuroraB and multi-kinase inhibitor. AT-9283 has shown to inhibit growth and survival of multiple solid tumor cell lines and is efficacious in mouse xenograft models.
AT 9283 is a substance being studied in the treatment of some types of cancer. It is small molecule a multi-targeted c-ABL, JAK2, Aurora A and B inhibition with 4, 1.2, 1.1 ad approximate 3 nM for Bcr-Abl (T3151), Jak2 and Jak3 aurora A and B, respectively. It blocks enzymes (Aurora kinases) involved in cell division and may kill cancer cells
WO2006070195 to Astex Therapeuitcs discloses pyrazole compounds of the general structure shown below as kinase inhibitors.

The compound AT9283 is in phase II clinical trials for treating advanced or metastatic solid tumors or Non-Hodgkin’s Lymphoma. AT9283 is shown below.

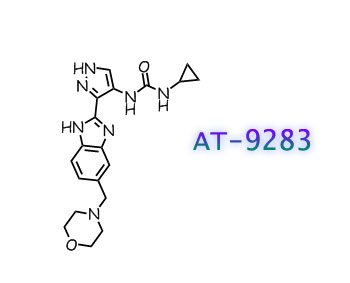
a Reagents and conditions:
(a) SOCl2, THF, DMF; (b) morpholine, THF, Et3N; ………FORMATION OOF ACID CHLORIDE AND COUPLING WITH MORPHOLINE
(c) NaBH4, BF3.OEt2, THF; …………..KETO TO CH2
(d) 10% Pd-C, H2, EtOH; TWO NITRO GPS TO TWO AMINO , REDN
(e) EDC, HOBt, DMF; (f) AcOH, reflux;COUPLING WITH 4-Nitro-lH-pyrazole-3-carboxylic acid
(g) 10%Pd-C, H2, DMF; NITRO GP TO AMINO
(h) standard amide and urea coupling methods
WO2006070195
https://www.google.co.in/patents/WO2006070195A1?cl=en
Stage 10: Synthesis of l-cvclopropyl-3-[3-(5-morpholin-4-ylmethyl-lH- beiizoimidazol-2-ylV 1 H-pyrazol-4-yli -urea.
To a mixture of 7-morpholin-4-ylmethyl-2,4-dihydro- 1 ,2,4,5a, 10- pentaaza- cyclopenta[a]fluoren-5-one (10.7 g, 32.9 mmol) in NMP (65 mL) was added cyclopropylamine (6.9 mL, 99 mmol). The mixture was heated at 100 0C for 5 h. LC/MS analysis indicated -75% conversion to product, therefore a further portion of cyclopropylamine (2.3 mL, 33 mmol) was added, the mixture heated at 100 0C for 4 h and then cooled to ambient. The mixture was diluted with water (100 mL) and extracted with EtOAc (100 niL). The organic portion was washed with sat. aq. NH4Cl (2 x 50 mL) and brine (50 rnL) and then the aqueous portions re-extracted with EtOAc (3 x 100 mL). The combined organic portions were dried over MgSO4 and reduced in vacuo to give l-cycloρropyl-3-[3-(5-morpholin-4-ylmethyl-lH- benzoimidazol-2-yl)-lH-pyrazol-4-yl]-urea as an orange glassy solid (9.10 g).
Stage 11: Synthesis of l-cvclopropyl-S-P-fS-morpholin^-ylmethyl-lH- benzoimidazol-2-yl)-lH-pyrazol-4-yll-urea, L-lactate salt
To a solution of l-cyclopropyl-3-[3-(5-morpholin-4-ylmethyl-lH-benzoimidazol-2- yl)-lH-pyrazol-4-yl]-urea (9.10 g, 24 mmol) in EtOAc-iPrOH (1 :1, 90 mL) was added L-lactic acid (2.25 g, 25 mmol). The mixture was stirred at ambient temperature for 24 h then reduced in vacuo. The residue was given consecutive slurries using toluene (100 mL) and Et2O (100 mL) and the resultant solid collected and dried (8.04 g).
This solid was purified by recrystallisation from boiling iPrOH (200 mL) to give after drying l-cyclopropyl-3-[3-(5-morpholin-4-ylmethyl-lH-benzoimidazol-2-yl)- lH-pyrazol-4-yl]-urea, L-lactate salt (5.7 g) as a beige solid.
EXAMPLE 66
Stage 1: Preparation of (3,4-dinitrophenyl)-morpholin-4-yl-methanone
3,4-Dinitrobenzoic acid (1.000Kg, 4.71mol, l.Owt), tetiuhydrofuran (10.00L5 lO.Ovol), and dimethylformamide (0.010L, O.Olvol) were charged to a flask under nitrogen. Thionyl chloride (0.450L, 6.16mol, 0.45vol) was added at 20 to 3O0C and the reaction mixture was heated to 65 to 7O0C. Reaction completion was determined by 1H NMR analysis (d6-DMSO), typically in 3 hours. The reaction mixture was cooled to 0 to 50C and triethylamine (1.25L, 8.97mol, 1.25vol) was added at 0 to 100C. Morpholine (0.62L, 7.07mol, 0.62vol) was charged to the reaction mixture at 0 to 1O0C and the slurry was stirred for 30 minutes at 0 to 1O0C. Reaction completion was determined by H NMR analysis (d6-DMSO). The reaction mixture was warmed to 15 to 2O0C and water (4.00L, 4.0vol) was added. This mixture was then charged to a 4OL flange flask containing water (21.0OL, 21.0vol) at 15 to 250C to precipitate the product. The flask contents were cooled to and aged at 0 to 50C for 1 hour and the solids were collected by filtration. The filter-cake was washed with water (4x 5.00L, 4x 5.0vol) and the pH of the final wash was found to be pH 7. The wet filter-cake was analysed by H NMR for the presence of triethylamine hydrochloride. The filter-cake was dried at 40 to 450C under vacuum until the water content by KF <0.2%w/w, to yield (3,4-dinitrophenyl)-morpholin-4-yl-methanone (1.286Kg, 97.0%, KF 0.069%w/w) as a yellow solid.
Stage 2: Preparation of 4-(3,4-dinitro-benzyl)-morpholine
C11H11N3O6 C11H13N3O5
FW:281.22 FW:267.24
(3,4-DinitiOphenyl)-morpholin-4-yl-methanone (0.750Kg, 2.67mol, l.Owt) and tetrahydrofuran (7.50L, lO.Ovol) were charged to a flask under nitrogen and cooled to 0 to 50C. Borontrifluoride etherate (0.713L, 5.63mol, 0.95vol) was added at 0 to 50C and the suspension was stirred at this temperature for 15 to 30 minutes. Sodium borohydride (0.212Kg, 5.60mol, 0.282wt) was added in 6 equal portions over 90 to 120 minutes. (A delayed exotherm was noted 10 to 15 minutes after addition of the first portion. Once this had started and the reaction mixture had been re-cooled, further portions were added at 10 to 15 minute intervals, allowing the reaction to cool between additions). The reaction mixture was stirred at 0 to 50C for 30 minutes. Reaction completion was determined by 1H NMR analysis (d6-DMSO). Methanol (6.30L, 8.4vol) was added drop wise at 0 to 1O0C to quench the reaction mixture (rapid gas evolution, some foaming). The quenched reaction mixture was stirred at 0 to 1O0C for 25 to 35 minutes then warmed to and stirred at 20 to 3O0C (exotherm, gas/ether evolution on dissolution of solid) until gas evolution had slowed. The mixture was heated to and stirred at 65 to 7O0C for 1 hour. The mixture was cooled to 30 to 4O0C and concentrated under vacuum at 40 to 450C to give crude 4-(3,4-dinitro-benzyl)-morpholine (0.702Kg, 98.4%) as a yellow/orange solid.
4-(3,4-Dinitro-benzyl)-niorpholme (2.815kg, 10.53mol, l.Owt) and methanol (12.00L, 4.3vol) were charged to a flask under nitrogen and heated to 65 to 7O0C. The temperature was maintained until complete dissolution. The mixture was then cooled to and aged at 0 to 50C for 1 hour. The solids were isolated by filtration. The filter-cake was washed with methanol (2x 1.50L, 2x 0.5vol) and dried under vacuum at 35 to 45°C to give 4-(3,4-dinitro-benzyl)-morpholine (2.353Kg, 83.5% based on input Stage 2, 82.5% overall yield based on total input Stage 1 material,) as a yellow solid.
Stage 3: Preparation of 4-morpholin-4-yl-methyl-benzene-L2-diamine
C11H13N3O5 C11H17N3O
FW:267.24 FW:207.27
4-(3,4-Dinitro-benzyl)-morρholine (0.800Kg, 2.99mol, l.Owt), and ethanol (11.20L, 14.0vol) were charged to a suitable flask and stirred at 15 to 250C and a vacuum / nitrogen purge cycle was performed three times. 10% Palladium on carbon (10%Pd/C, 50%wet paste, 0.040Kg, 0.05wt wet weight) was slurried in ethanol (0.80L, l.Ovol) and added to the reaction. The mixture was cooled to 10 to 2O0C and a vacuum / nitrogen purge cycle was performed three times. A vacuum / hydrogen purge cycle was performed three times and the reaction was stirred under a hydrogen atmosphere at 10 to 2O0C. Reaction completion was determined by 1H NMR analysis (d6-DMSO), typically 14 to 20 hours. A vacuum / nitrogen purge cycle was performed three times and the reaction mixture was filtered through glass microfibre paper under nitrogen. The filter-cake was washed with ethanol (3x 0.80L, 3x l.Ovol) and the combined filtrate and washes were concentrated to dryness under vacuum at 35 to 450C to give 4-morpholin-4-yl-methyl-benzene-l,2- diamine (0.61 IKg 98.6%) as a brown solid.
Stage 4: Preparation of 4-nitiO-lH-pyrazole-3-carboxγlic acid methyl ester
C4H3N3O4 C5H5N3O4
FW: 157.09 FW: 171.11
4-Nitro-lH-pyrazole-3-carboxylic acid (1.00kg, 6.37mol, l.Owt) and methanol (8.00L, 8.0vol) were charged to a flange flask equipped with a mechanical stirrer, condenser and thermometer. The suspension was cooled to 0 to 5°C under nitrogen and thionyl chloride (0.52L, 7.12mol, 0.52vol) was added at this temperature. The mixture was warmed to 15 to 25°C over 16 to 24 hours. Reaction completion was determined by 1H NMR analysis (d6-DMSO). The mixture was concentrated under vacuum at 35 to 45°C. Toluene (2.00L, 2.0vol) was charged to the residue and removed under vacuum at 35 to 450C. The azeotrope was repeated twice using toluene (2.00L, 2.0vol) to give 4-nitro-lH-pyrazole-3-carboxylic acid methyl ester (1.071Kg, 98.3%) as an off white solid.
Stage 5: Preparation of 4-amino-lH-pyrazole-3-carboxylic acid methyl ester. O2Me
C5H 5N3O4 C5H7N3O2 FW: 171.11 FW: 141.13
A suspension of 4-nitro-lH-pyrazole-3-carboxylic acid methyl ester (1.084Kg, 6.33mol, l.Owt) and ethanol (10.84L, lO.Ovol) was heated to and maintained at 30 to 35°C until complete dissolution occurred. 10% Palladium on carbon (10% Pd/C wet paste, 0.152Kg, 0.14wt) was charged to a separate flask under nitrogen and a vacuum / nitrogen purge cycle was performed three times. The solution of 4-nitro- lH-pyrazole-3-carboxylic acid methyl ester in ethanol was charged to the catalyst and a vacuum / nitrogen purge cycle was performed three times. A vacuum / hydrogen purge cycle was performed three times and the reaction was placed under an atmosphere of hydrogen. The reaction mixture was stirred at 28 to 30°C until deemed complete by 1H NMR analysis (d6-DMSO). The mixture was filtered under nitrogen and concentrated under vacuum at 35 to 450C to give 4-amino-lH- pyrazole-3-carboxylic acid methyl ester (0.883Kg, 98.9%) as a purple solid.
Stage 6: Preparation of 4-fert-butoxycarbonylamino-lH-pyrazole-3-carboxylic acid
C5H7N3O2 C9H13N3O4
FW: 141.13 FW:227.22
4-Amino-lH-pyrazole-3-carboxylic acid methyl ester (1.024Kg, 7.16mol, l.Owt) and dioxane (10.24L, lO.Ovol) were charged to a flange flask equipped with a mechanical stirrer, condenser and thermometer. 2M aq. Sodium hydroxide solution (4.36L, 8.72mol, 4.26vol) was charged at 15 to 250C and the mixture was heated to 45 to 550C. The temperature was maintained at 45 to 550C until reaction completion, as determined by 1H NMR analysis (d6-DMSO). Di-te/Y-butyl dicarbonate (Boc anhydride, 1.667Kg, 7.64mol, 1.628wt) was added at 45 to 55°C and the mixture was stirred for 55 to 65 minutes. 1H NMR IPC analysis (d6-DMSO) indicated the presence of 9% unreacted intermediate. Additional di-fert-butyl dicarbonate (Boc anhydride, 0.141Kg, 0.64mol, 0.14wt) was added at 55°C and the mixture was stirred for 55 to 65 minutes. Reaction completion was determined by 1H NMR analysis (d6-DMSO). The dioxane was removed under vacuum at 35 to 450C and water (17.60L, 20.0vol) was added to the residue. The pH was adjusted to pH 2 with 2M aq. hydrochloric acid (4.30L, 4.20vol) and the mixture was filtered. The filter-cake was slurried with water (10.00L3 9.7vol) for 20 to 30 minutes and the mixture was filtered. The filter-cake was washed with heptanes (4.10L, 4.0vol) and pulled dry on the pad for 16 to 20 hours. The solid was azeodried with toluene (5x 4.00L, 5x 4.6vol) then dried under vacuum at 35 to 45°C to give 4-tert- butoxycarbonylamino-lH-pyrazole-3-carboxylic acid (1.389Kg, 85.4%) as a purple solid.
Stage 7: Preparation of [3-(2-amino-4-moipholin-4-ylmetliyl-phenylcarbamoviy lH-pyrazol-4-yl]-carbamic acid tert-butyl ester
C9H13N3O4 C11H17N3O C20H28N6O4
FW: 227.22 FW: 207.27 FW: 416.48
+ regioisomer
4-førf-Butoxycarbonylamino-lH-pyrazole-3-carboxylic acid (0.750Kg, 3.30 mol, l.Owt), 4-morpholin-4yl-methyl-benzene-l,2-diamine (0.752Kg, 3.63mol, l.Owt) and N,N’-dimethylformamide (11.25L, 15.0vol) were charged under nitrogen to a flange flask equipped with a mechanical stirrer and thermometer. 1- Hydroxybenzotriazole (HOBT, 0.540Kg, 3.96mol, 0.72wt) was added at 15 to 250C. N-(3-Dimethylaminopropyl)-N’-ethylcarbodiimide (EDC, 0.759Kg, 3.96mol, 1.01 wt) was added at 15 to 250C and the mixture was stirred at this temperature for 16 to 24 hours. Reaction completion was determined by 1H NMR analysis. The reaction mixture was concentrated under vacuum at 35 to 45°C. The residue was partitioned between ethyl acetate (7.50L, lO.Ovol) and sat. aq. sodium hydrogen carbonate solution (8.03L, 10.7vol) and the layers were separated. The organic phase was washed with brine (3.75L, 5.0vol), dried over magnesium sulfate (1.00Kg, 1.33wt) and filtered. The filter-cake was washed with ethyl acetate (1.50L, 2.0vol). The combined filtrate and wash were concentrated under vacuum at 35 to 450C to give [3-(2-amino-4-morpholin-4-ylmethyl-phenylcarbamoyl)-lH-pyrazol- 4-yl]-carbamic acid tert-butyl ester (1.217Kg, 88.6%) as a dark brown solid.
Stage 8 : Preparation of 3 -f 5-morpholin-4-ylmethyl- 1 H-benzoimidazol-2-ylV 1 H- pyrazol-4-ylamme
C15H19N6O
FW: 298.35
As a mixture of two regioisomers
[3-(2-Amino-4-morpholin-4-ylmethyl-phenylcarbamoyl)-lH-pyrazol-4-yl]- carbamic acid tert-butyl ester (1.350Kg, 3.24 mol, l.Owt) and ethanol (6.75L, 5.0vol) were charged to a flange flask equipped with a mechanical stirrer, condenser and thermometer. Cone. aq. hydrochloric acid (1.10L, 13.2 mol, 0.80vol) was added at 15 to 3O0C under nitrogen and the contents were then heated to 70 to 😯0C and maintained at this temperature for 16 to 24 hours. A second portion of hydrochloric acid (0.1 IL, 1.32 mol, O.OSOvol) was added at 70 to 😯0C and the reaction was heated for a further 4 hours. Reaction completion was determined by HPLC analysis. The reaction mixture was cooled to 10 to 200C and potassium carbonate (1.355Kg, 9.08mol, l.Owt) was charged portionwise at this temperature. The suspension was stirred until gas evolution ceased and was then filtered. The filter-cake was washed with ethanol (1.35L, l.Ovol) and the filtrates retained. The filter-cake was slurried with ethanol (4.00L, 3.0vol) at 15 to 250C for 20 to 40 minutes and the mixture was filtered. The filter-cake was washed with ethanol (1.35L3 1.Ovol) and the total combined filtrates were concentrated under vacuum at 35 to 450C. Ethanol (4.00L, 3. Ovol) was charged to the residue and removed under vacuum at 35 to 450C. Tetrahydrofuran (5.90L, 4.4vol) was added to the residue and stirred for 10 to 20 minutes at 15 to 25°C. The resulting solution was filtered, the filter-cake was washed with tetrahydrofuran (1.35L, l.Ovol) and the combined filtrates were concentrated under vacuum at 35 to 450C. Tetrahydrofuran (5.40L, 4. Ovol) was charged to the concentrate and removed under vacuum at 35 to 450C. Tetrahydrofuran (5.40L, 4. Ovol) was charged to the concentrate and removed under vacuum at 35 to 45°C to give the desired product, 3-(5-morpholin-4-ylmethyl-lH- benzoimidazol-2-yl)-lH-pyrazol-4-ylamine (0.924Kg, 95.5%, 82.84% by HPLC area) as a purple foam.
Stage 9: Preparation of 7-morpholin-4-ylmethyl-2,4-dihydro- 1,2,4,5a ,10-pentaaza- cyclopentaFal fluoren-5 -one
C15H18N6O C16H16N6O2 FW: 298.35 FW: 324.34
As a mixture of two regioisomers
3-(5-Morpholin-4-ylmethyl-lH-benzoimidazol-2-yl)-lH-pyrazol-4-ylamine (0.993Kg, 3.33 mol, l.Owt) and tetrahydrofuran (14.0L, 15.0vol) were charged to a flange flask equipped with a mechanical stirrer, condenser and thermometer. The contents were stirred under nitrogen at 15 to 25°C and l,l ‘-carbonyldiimidazole (0.596Kg, 3.67 mol, O.όOwt) was added. The contents were then heated to 60 to 700C and stirred at this temperature for 16 to 24 hours. Reaction completion was determined by TLC analysis. The mixture was cooled to 15 to 200C and filtered. The filter-cake was washed with tetrahydrofuran (4.00L, 4. Ovol) and pulled dry for 15 to 30 minutes. The solid was dried under vacuum at 35 to 450C to yield 7- morpholin-4-ylmethyl-2,4-dihydro- 1 ,2,4,5a, 10-pentaaza-cyclopenta[a]fluoren-5- one (0.810Kg, 75.0%th, 92.19% by HPLC area) as a purple solid. Stage 10: Preparation of l-cvclopropyl-3-[3-(5-morpholin-4-ylmethyl-lH- benzoimidazol-2-vD- 1 H-pyrazol-4-yll -urea
C16H16N6O2 C19H23N7O2
FW: 324.34 FW: 381.44
As a mixture of two regioisomers
7-Morpholin-4-ylmethyl-254-dihydro-l,2,4,5a,10-pentaaza-cyclopenta[a]fluoren-5- one (0.797Kg, 2.46mol, l.Owt) and l-methyl-2-pyrrolidinone (2.40L, 3.0vol) were charged to a flange flask equipped with a mechanical stirrer, condenser and thermometer. Cyclopropylamine (0.279Kg, 4.88mol, 0.35 lwt) was added at 15 to 30°C under nitrogen. The contents were heated to 95 to 105°C and stirred at this temperature for 16 to 24 hours. Reaction completion was determined by 1H NMR analysis. The reaction mixture was cooled to 10 to 200C and ethyl acetate (8.00L, lO.Ovol) and sat. aq. sodium chloride (2.50L, 3.0vol) were charged, the mixture was stirred for 2 to 5 minutes and the layers separated. The organic phase was stirred with sat. aq. sodium chloride (5.00L, ό.Ovol) for 25 to 35 minutes, the mixture filtered and the filter-cake washed with ethyl acetate (0.40L, 0.5vol). The filter-cake was retained and the filtrates were transferred to a separating funnel and the layers separated. The procedure was repeated a further 3 times and the retained solids were combined with the organic phase and the mixture concentrated to dryness under vacuum at 35 to 450C. The concentrate was dissolved in propan-2-ol (8.00L, lO.Ovol) at 45 to 55°C and activated carbon (0.080Kg5 O.lwt) was charged. The mixture was stirred at 45 to 550C for 30 to 40 minutes and then hot filtered at 45 to 55°C. The filter-cake was washed with propan-2-ol (0.40L, 0.5vol). Activated carbon (0.080L, O.lwt) was charged to the combined filtrates and wash and the mixture stirred at 45 to 550C for 30 to 40 minutes. The mixture was hot filtered at 45 to 550C and the filter-cake washed with propan-2-ol (0.40L, 0.5vol). The filtrates and wash were concentrated under vacuum at 35 to 450C. Ethyl acetate (8.00, lO.Ovol) and water (2.20L, 3.0vol) were charged to the concentrate at 25 to 350C and the mixture stirred for 1 to 2 minutes. The layers were separated and the organic phase was concentrated under vacuum at 35 to 45°C. Ethyl acetate (4.00L, 5.0vol) was charged to the residue and concentrated under vacuum at 35 to 450C. Ethyl acetate (4.00L, 5.0vol) was charged to the residue and the mixture was stirred for 2 to 20 hours at 15 to 250C. The mixture was cooled to and aged at 0 to 5°C for 90 to 120 minutes and then filtered. The filter-cake was washed with ethyl acetate (0.80L, l.Ovol) and pulled dry for 15 to 30 minutes. The solid was dried under vacuum at 35 to 450C to yield l-cyclopropyl-3-[3-(5-morpholin-4-ylmethyl-lH- benzoimidazol-2-yl)-lH-pyrazol-4-yl]-urea (0.533Kg, 56.8%, 93.20% by HPLC area) as a brown solid.
Several batches of Stage 9 product were processed in this way and the details of the quantities of starting material and product for each batch are set out in Table IA.
Table IA – Yields from urea formation step – Stage 10
Stage 11 : Preparation of l-cyclopiOpyl-3-r3-(5-moipholin-4-ylmethyl-lH- benzoimidazol-2-yls)-lH-pyrazol-4-yll-urea £-lactic acid salt L-Lactic acid
acid
C19H23N7O2 C22H29N7O5
FW: 381.44 FW: 471.52 l-Cyclopropyl-3-[3-(5-morpholin-4-ylmethyl-lH-benzoimidazol-2-yl)-lH-ρyrazol- 4-yl]-urea (1.859Kg, 4.872mol, l.Owt), propan-2-ol (9.00L5 5.0vol) and ethyl acetate (8.0OL, 4.5vol) were charged to a flange flask equipped with a mechanical stirrer and thermometer. The contents were stirred under nitrogen and L-lactic acid (0.504Kg, 5.59mol, 0.269wt) was added at 15 to 25°C followed by a line rinse of ethyl acetate (0.90L, 0.5vol). The mixture was stirred at 15 to 25°C for 120 to 140 minutes. The solid was isolated by filtration, the filter-cake washed with ethyl acetate (2x 2.00L, 2x l.Ovol) and pulled dry for 20 to 40 minutes. The filter-cake was dissolved in ethanol (33.00L, 17.7vol) at 75 to 850C, cooled to 65 to 700C and the solution clarified through glass microfibre paper. The filtrates were cooled to and aged at 15 to 250C for 2 to 3 hours. The crystallised solid was isolated by filtration, the filter-cake washed with ethanol (2x 1.00L, 2x 0.5vol) and pulled dry for at least 30 minutes. The solid was dried under vacuum at 35 to 45°C to yield 1- cyclopropyl-3 – [3-(5 -morpholin-4-ylmethyl- 1 H-benzoimidazol-2-yl)- 1 H-pyrazol-4- yl]-urea l-lactic acid salt (1.386Kg, 58.7%th, 99.47% by HPLC area,) as a dark pink uniform solid.
The infra-red spectrum of the lactate salt (KBr disc method) included characteristic peaks at 3229, 2972 and 1660 cm“1.
Without wishing to be bound by any theory, it is believed that the infra red peaks can be assigned to structural components of the salt as follow:
Peak: Due to:
3229 cm“1 N-H
2972 cm“1 aliphatic C-H
1660 cm“1 urea C=O EXAMPLE 67
Synthesis of Crystalline Free Base And Crystalline Salt Forms Of l-Cyclopropyl-3-
[3-(5-Morpholin-4-ylmethyl-lH-Benzoimidazol-2-vπ-lH-Pyrazol-4-yll-Urea
A. Preparation of l-Cvclopropyl-3-[3-f5-Moφholm-4-ylmethyl-lH- Benzoimidazol-2-yl)-lH-Pyrazol-4-yll-Urea free base
A sample of crude l-cyclopropyl-3-[3-(5-morpholin-4-ylmethyl-lH-benzoimidazol- 2-yl)-lH-pyrazol-4-yl]-urea free base was prepared as outlined in Example 60 and initially purified by column chromatography on silica gel, eluting with EtOAc- MeOH (98:2 – 80:20). A sample of the free base obtained was then recrystallised from hot methanol to give crystalline material of l-cyclopropyl-3-[3-(5-morpholin- 4-ylmethyl- 1 H-benzoimidazol-2-yl)- 1 H-pyrazol-4-yl] -urea free base.
B. Preparation of l-Cyclopropyl-S-rS-fS-Morpholin^-ylmethyl-lH-Benzoimidazol- 2-yl)-lH-Pyrazol-4-yl]-Urea free base dihydrate
A sample of crude l-cyclopropyl-3-[3-(5-moφholm-4-ylmethyl-lH-benzoimidazol- 2-yl)-l H-pyrazol-4-yl] -urea free base was dissolved in THF and then concentrated in vacuo to a minimum volume (~4 volumes). To the solution was added water dropwise (2 – 4 volumes) until the solution became turbid. A small amount of THF was added to re-establish solution clarity and the mixture left to stand overnight to give a crystalline material which was air-dried to give l-cyclopropyl-3-[3-(5- morpholin-4-ylmethyl- 1 H-benzoimidazol-2-yl)- 1 H-pyrazol-4-yl] -urea free base dihydrate.
C. Preparation of l-Cyclopl^pyl-3-[3-(5-Morpholm-4-ylmethyl-lH-Benzoimidazol- 2-ylVlH-Pyrazol-4-yl]-Urea hydrochloride salt
A sample of crude l-cyclopropyl-3-[3-(5-moφholin-4-ylmethyl-lH-benzoimidazol- 2-yl)-l H-pyrazol-4-yl] -urea free base was dissolved in the minimum amount of MeOH and then diluted with EtOAc. To the solution at 0 °C was slowly added 1.1 equivalents of HCl (4M solution in dioxane). Following addition, solid precipitated from solution which was collected by filtration. To the solid was added MeOH and the mixture reduced in vacuo. To remove traces of residual MeOH the residue was evaporated from water and then dried at 60 0C/ 0.1 mbar to give the hydrochloride salt.
D. Preparation of l-Cyclopropyl-3-[3-(‘5-Morpholm-4-ylmethyl-lH- Benzoimidazol-2-yiyiH-Pyrazol-4-yl1-Urea ethanesulfonate salt
To a solution of l-cyclopropyl-3-[3-(5-morpholin-4-ylmethyl-lH-benzoimidazol-2- yl)-lH-pyrazol-4-yl]-urea free base in MeOH-EtOAc was added 1 equivalent of ethanesulfonic acid. The mixture was stirred at ambient temperature and then reduced in vacuo. The residue was taken up in MeOH and to the solution was added Et2O. Mixture left to stand for 72 h and the solid formed collected by filtration and dried to give l-cyclopropyl-3-[3-(5-morpholin-4-ylmethyl-lH- benzoimidazol-2-yl)-lH-pyrazol-4-yl]-urea ethanesulfonate salt.
E. Preparation of l-Cvclopropyl-3-[3-(‘5-Morpholm-4-ylmethyl-lH-Benzoimidazol- 2-yl)-lH-Pyrazol-4-yl]-Urea methanesulfonate salt
To a solution of l-cyclopropyl-3-[3-(5-morpholin-4-ylmethyl-lH-benzoimidazol-2- yl)-lH-pyrazol-4-yl]-urea free base (394 mg) in MeOH-EtOAc was added 1 equivalent of methanesulfonic acid (67 μl). A solid was formed which was collected by filtration, washing with EtOAc. The solid was dissolved in the minimum amount of hot MeOH, allowed to cool and then triturated with Et2O. The solid was left to stand for 72 h and then collected by filtration, washing with MeOH, to give l-cyclopropyl-3-[3-(5-morpholin-4-ylmethyl-lH-benzoimidazol-2- yl)-lH-pyrazol-4-yl]-urea methanesulfonate salt.
EXAMPLE 68
Characterisation of l-Cvclopropyl-3-[3-(5-Morpholin-4-ylmethyl-lH-
Benzoimidazol-2-yl)-lH-Pyrazol-4-yll-Urea Free Base and Salts
Various forms of l-cyclopropyl-3-[3-(5-morpholin-4-ylmethyl-lH-benzoimidazol- 2-yl)-lH-pyrazol-4-yl]-urea were characterised. The forms selected for characterisation were identified from studies which primarily investigated extent of polymorphism and salt stability. The salts selected for further characterisation were the L-lactate salt, Free base dihydrate, Esylate salt, Free base and Hydrochloride salt.
Paper
Fragment-Based Discovery of the Pyrazol-4-yl Urea (AT9283), a Multitargeted Kinase Inhibitor with Potent Aurora Kinase Activity†
†
Coordinates of the protein complexes with compounds 5, 7, 9, 10, and 16 have been deposited in the Protein Data Bank under accession codes 2w1d, 2w1f, 2w1c, 2w1e, 2w1g (Aurora A), 2w1h (CDK2), and 2w1i (JAK2).
, * To whom correspondence should be addressed. Phone: +44 (0)1223 226209. Fax: +44 (0)1223 226201. E-mail: s.howard@astex-therapeutics.com.
Abstract

Here, we describe the identification of a clinical candidate via structure-based optimization of a ligand efficient pyrazole-benzimidazole fragment. Aurora kinases play a key role in the regulation of mitosis and in recent years have become attractive targets for the treatment of cancer. X-ray crystallographic structures were generated using a novel soakable form of Aurora A and were used to drive the optimization toward potent (IC50 ≈ 3 nM) dual Aurora A/Aurora B inhibitors. These compounds inhibited growth and survival of HCT116 cells and produced the polyploid cellular phenotype typically associated with Aurora B kinase inhibition. Optimization of cellular activity and physicochemical properties ultimately led to the identification of compound16 (AT9283). In addition to Aurora A and Aurora B, compound 16 was also found to inhibit a number of other kinases including JAK2 and Abl (T315I). This compound demonstrated in vivo efficacy in mouse xenograft models and is currently under evaluation in phase I clinical trials.
1-Cyclopropyl-3-[3-(5-morpholin-4-ylmethyl-1H-benzoimidazol-2-yl)-1H-pyrazol-4-yl]urea (16), Hydrochloride Salt
///////////
C1CC1NC(=O)NC2=CNNC2=C3N=C4C=CC(=CC4=N3)CN5CCOCC5
Aliskiren

ALISKIREN
(2S,4S,5S,7S)-5-amino-N-(2-carbamoyl-2,2-dimethylethyl)-4-hydroxy-7-{[4-methoxy-3-(3-methoxypropoxy)phenyl]methyl}-8-methyl-2-(propan-2-yl)nonanamide, CAS 173334-57-1, base
CAS 173334-58-2,aliskiren hemifumarate
Aliskiren is a renin inhibitor. It was approved by the U.S. Food and Drug Administration in 2007 for the treatment of hypertension.
Tekturna contains aliskiren hemifumarate, a renin inhibitor, that is provided as tablets for oral administration. Aliskiren hemifumarate is chemically described as (2S,4S,5S,7S)-N-(2-carbamoyl-2-methylpropyl)-5-amino-4-hydroxy-2,7diisopropyl-8-[4-methoxy-3-(3-methoxypropoxy)phenyl]-octanamide hemifumarate and its structural formula is
 |
Molecular formula: C30H53N3O6 • 0.5 C4H4O4
Aliskiren hemifumarate is a white to slightly yellowish crystalline powder with a molecular weight of 609.8 (free base- 551.8). It is soluble in phosphate buffer, n-octanol, and highly soluble in water.
|
Aliskiren (INN) (trade names Tekturna, US; Rasilez, UK and elsewhere) is the first in a class of drugs called direct renin inhibitors. Its current licensed indication is essential (primary) hypertension.
Aliskiren was co-developed by the Swiss pharmaceutical companies Novartis andSpeedel.[1][2] It was approved by the US Food and Drug Administration in 2007 for the treatment of primary hypertension.[3]
In December 2011, Novartis had to halt a clinical trial of the drug after discovering increased incidence of nonfatal stroke, renal complications, hyperkalemia, and hypotension in patients with diabetes and renal impairment (ALTITUDE Trial ).[4] [5]
As a result, in April 20, 2012:
A new contraindication was added to the product label concerning the use of aliskiren with angiotensin receptor blockers (ARBs) or angiotensin-converting enzyme inhibitors (ACEIs) in patients with diabetes because of the risk of renal impairment, hypotension, and hyperkalemia.
A warning to avoid use of aliskiren with ARBs or ACEIs was also added for patients with moderate to severe renal impairment (i.e., where glomerular filtration rate is less than 60 ml/min).
Renin, the first enzyme in the renin-angiotensin-aldosterone system, plays a role in blood pressure control. It cleaves angiotensinogen to angiotensin I, which is in turn converted byangiotensin-converting enzyme (ACE) to angiotensin II. Angiotensin II has both direct and indirect effects on blood pressure. It directly causes arterial smooth muscle to contract, leading to vasoconstriction and increased blood pressure. Angiotensin II also stimulates the production of aldosterone from the adrenal cortex, which causes the tubules of the kidneys to increase reabsorption of sodium, with water following, thereby increasing plasma volume, and thus blood pressure. Aliskiren binds to the S3bp binding site of renin, essential for its activity.[6] Binding to this pocket prevents the conversion of angiotensinogen to angiotensin I. Aliskiren is also available as combination therapy withhydrochlorothiazide.[7]
Many drugs control blood pressure by interfering with angiotensin or aldosterone. However, when these drugs are used chronically, the body increases renin production, which drives blood pressure up again. Therefore, doctors have been looking for a drug to inhibit renin directly. Aliskiren is the first drug to do so.[8][9]
Aliskiren may have renoprotective effects independent of its blood pressure−lowering effect in patients with hypertension, type 2 diabetes, and nephropathy, who are receiving the recommended renoprotective treatment. According to the AVOID study, researchers found that treatment with 300 mg of aliskiren daily, as compared with placebo, reduced the mean urinary albumin-to-creatinine ratio by 20%, with a reduction of 50% or more in 24.7% of the patients who received aliskiren as compared with 12.5% of those who received placebo. Furthermore, the AVOID trial showed treatment with 300 mg of aliskiren daily reduces albuminuria in patients with hypertension, type 2 diabetes, and proteinuria, who are receiving the recommended maximal renoprotective treatment with losartan and optimal antihypertensive therapy. Therefore, direct renin inhibition will have a critical role in strategic renoprotective pharmacotherapy, in conjunction with dual blockade of the renin−angiotensin−aldosterone system with the use of ACE inhibitors and angiotensin II–receptor blockers, very high doses of angiotensin II−receptor blockers, and aldosterone blockade.[10]
Aliskiren is a minor substrate of CYP3A4 and, more important, P-glycoprotein:
- It reduces furosemide blood concentration.
- Atorvastatin may increase blood concentration, but no dose adjustment is needed.
- Due to possible interaction with ciclosporin, the concomitant use of ciclosporin and aliskiren is contraindicated.
- Caution should be exercised when aliskiren is administered with ketoconazole or other moderate P-gp inhibitors (itraconazole, clarithromycin, telithromycin, erythromycin, or amiodarone).
- Doctors should stop prescribing aliskiren-containing medicines to patients with diabetes (type 1 or type 2) or with moderate to severe kidney impairment who are also taking an ACE inhibitor or ARB, and should consider alternative antihypertensive treatment as necessary.[13]
-
Aliskiren (I) is a second generation renin inhibitor with renin-angiotensin system (RAS) as its target. It’s used clinically in the form of Aliskiren hemifumarate (Rasilez®) and was approved by FDA in May, 2007.
-
Aliskiren has the chemical name: (2S, 4S, 5S, 7S)-5-amino-N-(2-carbamoyl-2-methylpropyl)-4-hydroxy-2-isopropyl-7-[4-methoxy-3-(3-methoxypropoxy)benzyl]-8-methyloctanamide (CAS No.: 173334-57-1). Its chemical structure is illustrated with Formula I given below:
-
The method of preparation for Aliskiren and its intermediates has been reported in US7132569 , WO0208172 , US5559111 (equivalent patent toCN1266118 ), US5606078 , CN101016253 , WO2007/045421 ,EP2062874 , Helvetica ChimicaActa (2005, 3263-3273).
-
In US7132569 , WO0208172 et al., the preparation of Aliskiren (I) comprises the following steps as described in reaction scheme 1: coupling 2-(3-methoxypropoxy)-4-((R)-2-(bromomethyl)-3-methylbutyl)-1-methoxybenzene (II) with (2S, 4E)-5-chloro-2-isopropyl-4-pentenoic acid derivative (III) to obtain the compound of formula IV; halolactonization of the compound of formula IV to obtain the compound of formula V; then substituting the compound of formula V with azide to obtain the compound of formula VI; ring-opening the compound of formula VI with 3-amino-2,2-dimethylpropionamide (VII) in the presence of 2-hydroxypyridine and triethylamine to obtain the compound of formula VIII and a final catalytic hydrogenation of the compound of formula VIII to obtain Aliskiren (I). This preparation process is illustrated in Reaction Scheme 1.
-
In the patented preparation described above, chiral starting materials with the compounds of formula II and III are utilized to obtain the compound of formula IV. However, the reactions followed after the preparation of the compound of formula IV, such as the halolactonization and especially the substitutive reaction between the compound of formula V and azide, have problems of low yields and numerous by-products, which is not conducive to industrial scale production.
-
In the patented preparation described above, there are multiple reaction steps in the preparation of the compound of formula X from the compound of formula XII. The key steps, as described in Reaction Scheme 3, involve selective reduction agents such as sodium tri-tert-butoxyaluminum hydride and diisobutylaluminium hydride to prepare aldehyde and the reaction conditions need to be very well-controlled.
-
[0009]The compound of formula XI prepared by reaction scheme 2 could then be converted into Aliskiren (I) after multiple catalytic hydrogenation, protection and de-protection. In this method of preparation, a stepwise catalytic hydrogenation, azido reduction and dehydroxylation were implemented to reduce by-products during the catalytic hydrogenation. In addition, it is necessary to protect and de-protect the free hydroxyl group during the preparation. This synthetic scheme has disadvantage of multiple synthetic steps, tedious operation, lengthy overall reaction duration, low yield and particularly high production cost for the starting compound of formula X.
-
WO2007/045421 has reported an improved preparation method in which the starting material 4-bromo-1-methoxy-2-(3-methoxypropoxy)benzene (IX) firstly reacts with the compound of formula XIII via Grignard reaction to obtain the compound of formula XIV, and then followed by catalytic hydrogenation and ketone reduction to yield the compound of formula XV-A, as illustrated in Reaction Scheme 4:
-
In the above preparation, expensive reagents, such as sodium tri-tert-butoxyaluminum hydride and diisobutylaluminium hydride were eliminated, but additional synthetic steps were introduced. In addition, the preparation of the compound of formula XV-A prepared from the compound of formula XIV via ketone reduction required extended reaction time, great amount of catalyst with multiple small addition and good operation skills.
-
EP2062874A1 provides a method in preparing the compound of formula XVI. In this method, the compound of formula XVII is obtained from the compound of formula XVI via halogenation. A corresponding Grignard reagent is firstly prepared from the compound of formula IX or XVII reacting with magnesium, which is then couples with another chemical in the presence of the metal catalyst iron(III) acetylacetonate (Fe(acac)3) to obtain the compound of formula XVIII as described in Reaction Scheme 5:
-
In EP2062874A1 , the compound of formula XVIII reacts with 3-amino-2,2-dimethylpropionamide (VII). The resulted product is then through reduction of the azio group to obtain Aliskiren (I). In this patent, detailed experimental protocol was not provided although N-methylpyrrolidone was mentioned as solvent. We found: 1) it is difficult to prepare the Grgnard reagent from the compound of formula IX; 2) the compounds of formula XVII and XVIII are not quite stable in the presence of iron(III) acetylacetonate. In addition, the yield in preparing the compound of formula XVIII was extremely low.


NMR
ALISKIREN BASE

MS m/z: 552.6 (M+H)+; 1H-NMR (400 MHz, CDCl3) δ 6.88-6.75 (m, 3H), 4.08-4.04 (t, J = 6.3Hz, 2H), 3.79 (s, 3H), 3.60-3.55 (t, J = 6.3Hz, 2H), 3.30 (s, 3H), 3.30-3.25 (m, 3H), 2.69 (m, 2H), 2.49 (m, 1H), 2.27 (m, 1H), 2.04 (m, 2H), 1.78-1.35 (m, 7H), 1.10 (m, 6H), 0.90 (m, 12H) ppm.


Paper

A novel synthesis of the renin inhibitor aliskiren based on an unprecedented disconnection between C5 and C6 was developed, in which the C5 carbon acts as a nucleophile and the amino group is introduced by a Curtius rearrangement, which follows a simultaneous stereocontrolled generation of the C4 and C5 stereogenic centers by an asymmetric hydrogenation. Operational simplicity, step economy, and a good overall yield makes this synthesis amenable to manufacture on scale.
Convergent Synthesis of the Renin Inhibitor Aliskiren Based on C5–C6 Disconnection and CO2H–NH2 Equivalence










- Gradman A, Schmieder R, Lins R, Nussberger J, Chiang Y, Bedigian M (2005). “Aliskiren, a novel orally effective renin inhibitor, provides dose-dependent antihypertensive efficacy and placebo-like tolerability in hypertensive patients”. Circulation 111 (8): 1012–8.doi:10.1161/01.CIR.0000156466.02908.ED. PMID 15723979.
- Straessen JA, Li Y, and Richart T (2006). “Oral Renin Inhibitors”. Lancet 368 (9545): 1449–56. doi:10.1016/S0140-6736(06)69442-7. PMID 17055947.
- “First Hypertension Drug to Inhibit Kidney Enzyme Approved”. CBC. 2007-03-06. Retrieved 2007-03-14.[dead link]
- Healthzone.ca: Blood-pressure drug reviewed amid dangerous side effects
- Parving, Hans-Henrik; Barry M. Brenner, M.D., Ph.D., John J.V. McMurray, M.D., Dick de Zeeuw, M.D., Ph.D., Steven M. Haffner, M.D., Scott D. Solomon, M.D., Nish Chaturvedi, M.D., Frederik Persson, M.D., Akshay S. Desai, M.D., M.P.H., Maria Nicolai
- Alkylation of 3-hydroxy-4-methoxybenzyl alcohol (I) with 1-bromo-3-methoxypropane (II) gives ether (III). Subsequent conversion of benzyl alcohol (III) into bromide (IV) is carried out using bromotrimetylsilane. The chiral isovaleryloxazolidinone (V) is alkylated with bromide (IV) by means of LiHMDS to afford (VI), which is hydrolyzed to the (S)-2-aryl-2-isopropylpropionic acid (VII) by means of lithium peroxide. The reduction of acid (VII) to the corresponding alcohol with NaBH4/I2 reagent, followed by treatment with PPh3 and NBS, provides bromide (VIII). Alkylation of the chiral dimethoxydihydropyrazin (IX) with bromide (VIII) produces (X). Further hydrolysis of the pyrazine ring of (X) with HCl, followed by Boc protection of the resulting (S,S)-amino ester, yields compound (XI). Reduction of the ester group of (XI) with DIBAL gives aldehyde (XII). This compound is condensed with the Grignard reagent (XIII) to afford the diastereomeric mixture of amino alcohols (XIV). Treatment of mixture (XIV) with 2,2-dimethoxypropane (XV) and TsOH produces a mixture of oxazolidines, from which the required (S,S,S)-isomer (XVI) is isolated by flash chromatography. Hydrogenolitic deprotection of the benzyl ether of (XVI) gives alcohol (XVII).des, M.D., Alexia Richard, M.Sc., Zhihua Xiang, Ph.D., Patrick Brunel, M.D., and Marc A. Pfeffer, M.D., Ph.D. for the ALTITUDE Investigators (2012). “Cardiorenal End Points in a Trial of Aliskiren for Type 2 Diabetes”. NEJM 367 (23): 2204–13. doi:10.1056/NEJMoa1208799. PMID 23121378.
- J “Chemistry & Biology : Structure-based drug design: the discovery of novel nonpeptide orally active inhibitors of human renin”. ScienceDirect. Retrieved 2010-01-20.
- Baldwin CM, Plosker GL.[1]. doi:10.2165/00003495-200969070-00004. Drugs 2009; 69(7):833-841.
- Ingelfinger JR (June 2008). “Aliskiren and dual therapy in type 2 diabetes mellitus”. N. Engl. J. Med. 358 (23): 2503–5.doi:10.1056/NEJMe0803375. PMID 18525047.
- PharmaXChange: Direct Renin Inhibitors as Antihypertensive Drugs
- Parving HH, Persson F, Lewis JB, Lewis EJ, Hollenberg NK. “Aliskiren Combined with Losartan in Type 2 Diabetes and Nephropathy,” N Engl J Med 2008;358:2433-46.
- Drugs.com: Tekturna
- Cardiorenal end points in a trial of aliskiren for type 2 diabetes, N Engl J MED. 2012;367(23):2204-2213
- European Medicines Agency recommends new contraindications and warnings for aliskiren-containing medicines.
- Prescribing Information for Tekturna
- aliskiren at the US National Library of Medicine Medical Subject Headings (MeSH)
- Chemical synthesis
Drugs Fut2001, 26, (12): 1139
Tetrahedron Lett 2001, 42: 4819-23.
Tetrahedron Lett2000, 41, (51): 10085
EP 0678500; EP 0678503; JP 1996053434; JP 1996081430; US 5559111; US 5627182; US 5646143, WO 0109079; WO 0109083
 |
|
| Systematic (IUPAC) name | |
|---|---|
|
(2S,4S,5S,7S)-5-amino-N-(2-carbamoyl-2,2-dimethylethyl)-4-hydroxy-7-{[4-methoxy-3-(3-methoxypropoxy)phenyl]methyl}-8-methyl-2-(propan-2-yl)nonanamide
|
|
| Clinical data | |
| AHFS/Drugs.com | monograph |
| MedlinePlus | a607039 |
| Licence data | EMA:Link, US FDA:link |
| Pregnancy category |
|
| Legal status | |
| Routes of administration |
PO (oral) |
| Pharmacokinetic data | |
| Bioavailability | Low (approximately 2.5%) |
| Metabolism | Hepatic, CYP3A4-mediated |
| Biological half-life | 24 hours |
| Excretion | Renal |
| Identifiers | |
| CAS Number | 173334-57-1 |
| ATC code | C09XA02 C09XA52 (with HCT) |
| PubChem | CID: 5493444 |
| IUPHAR/BPS | 4812 |
| DrugBank | DB01258 |
| ChemSpider | 4591452 |
| UNII | 502FWN4Q32 |
| KEGG | D03208 |
| ChEBI | CHEBI:601027 |
| ChEMBL | CHEMBL1639 |
| Chemical data | |
| Formula | C30H53N3O6 |
| Molecular mass | 551.758 g/mol |

SEE……..http://www.allfordrugs.com/2013/12/17/aliskiren/
////
O=C(N)C(C)(C)CNC(=O)[C@H](C(C)C)C[C@H](O)[C@@H](N)C[C@@H](C(C)C)Cc1cc(OCCCOC)c(OC)cc1
SB 1578

SB1578
ONX 0805
(9E)-15-(2-(Pyrrolidin-1-yl)ethoxy)-7,12,25-trioxa-19,21,24-triaza-tetracyclo[18.3.1.1(2,5).1(14,18)]hexacosa-1(24),2,4,9,14(26), 15,17,20,22-nonaene
7,12,26-Trioxa-19,21,24-triazatetracyclo[18.3.1.12,5.114,18]hexacosa-1(24),2,4,9,14,16,18(25),20,22-nonaene, 15-[2-(1-pyrrolidinyl)ethoxy]-, (9E)-
Phase 1 clinical trials
C26 H30 N4 O4
CAS 937273-04-6
CITRATE 1262279-15-1
HCL 1262279-16-2
S*Bio Pte Ltd INNOVATOR
SB1578 (disclosed in WO2007058627 and in WO2011008172 as the citrate salt) is in ongoing phase I studies for the treatment of rheumatoid arthritis. SB 1578 is shown below.

SB1578, also known as ONX-0805, is a novel, orally bioavailable JAK2 inhibitor with specificity for JAK2 within the JAK family and also potent activity against FLT3 and c-Fms. SB1578 blocks the activation of these kinases and their downstream signaling in pertinent cells, leading to inhibition of pathological cellular responses. The biochemical and cellular activities of SB1578 translate into its high efficacy in two rodent models of arthritis. SB1578 not only prevents the onset of arthritis but is also potent in treating established disease in collagen-induced arthritis mice with beneficial effects on histopathological parameters of bone resorption and cartilage damage. SB1578 abrogates the inflammatory response and prevents the infiltration of macrophages and neutrophils into affected joints. It also leads to inhibition of Ag-presenting dendritic cells and inhibits the autoimmune component of the disease. In summary, SB1578 has a unique kinase spectrum, and its pharmacological profile provides a strong rationale for the ongoing clinical development in autoimmune diseases. ( J Immunol. 2012 Oct 15;189(8):4123-34)
Synonym: ONX 0805; ONX0805; ONX0805; SB1578; SB1578; SB 1578.

PATENT
WO 2011008172
http://www.google.im/patents/WO2011008172A1?cl=en
The compound 9E-15-(2-pyrrolidin-1-yl-ethoxy)-7,12,25-trioxa-19,21 ,24-triaza-tetracyclo[18.3.1.1(2,5).1(14,18)]hexacosa-1 (24),2,4,9,14,16l18(26)l20,22-nonaene (Compound I) was first described in PCT/SG2006/000352 and shows significant promise as a pharmaceutically active agent for the treatment of a number of medical conditions. Pharmaceutical development of this compound is underway based on the activity profiles demonstrated by the compound.

Compound I
In the development of a drug suitable for mass production and ultimately commercial use acceptable levels of drug activity against the target of interest is only one of the important variables that must be considered. For example, in the formulation of pharmaceutical compositions it is imperative that the pharmaceutically active substance be in a form that can be reliably reproduced in a commercial
manufacturing process and which is robust enough to withstand the conditions to which the pharmaceutically active substance is exposed.
From a manufacturing perspective, it is important that the commercial manufacturing process of a pharmaceutically active substance is such that the same material is produced when the same manufacturing conditions are used. In addition, it is desirable that the pharmaceutically active substance exists in a solid form where minor changes to the manufacturing conditions do not lead to major changes in the solid form of the pharmaceutically active substance produced. For example, it is important that the manufacturing process produces material having the same crystalline properties on a reliable basis, and also that the process produces material having the same level of hydration.
In addition, it is important that the pharmaceutically active substance be stable to degradation, hygroscopicity and subsequent changes to its solid form. This is important to facilitate the incorporation of the pharmaceutically active ingredient into pharmaceutical formulations. If the pharmaceutically active substance is hygroscopic (“sticky”) in the sense that it absorbs water over time it is almost impossible to reliably formulate the pharmaceutically active substance into a drug as the amount of substance to be added to provide the same dosage will vary greatly depending upon the degree of hydration. Furthermore, variations in hydration or solid form (“polymorphism”) can lead to changes in physico-chemical properties, such as solubility or dissolution rate, which can in turn lead to inconsistent oral absorption in a patient.
Accordingly, chemical stability, solid state stability, and “shelf life” of the pharmaceutically active agent are very important factors. In an ideal situation the pharmaceutically active agent and any compositions containing it, should be capable of being effectively stored over appreciable periods of time without exhibiting a significant change in the physico-chemical characteristics of the active component such as its activity, moisture content, solubility characteristics, solid form and the like.
In relation to 9E-15-(2-pyrrolidin-1-yl-ethoxy)-7,12,25-trioxa-19,21 ,24-triaza-tetracyclo[18.3.1.1 (2,5).1(14,18)]hexacosa-1(24),2,4,9,14,16,18(26),20,22-nonaene
initial studies were carried out on the hydrochloride salt and indicated that polymorphism was prevalent, with the compound being found to adopt more than one crystalline form depending upon the manufacturing conditions. In addition it was observed that the ratio of the polymorphs varied from batch to batch even when the manufacturing conditions remained constant. These batch-to-batch inconsistencies made the hydrochloride salt less desirable from a commercial viewpoint.
Accordingly it would be desirable to develop salts of 9E-15-(2-pyrrolidin-1-yl-ethoxy)-7, 12,25-trioxa-i 9,21 ,24-triaza-tetracyclo[18.3.1.1 (2,5).1 (14,18)]hexacosa-1(24)l2,4,9,14,16,18(26),20,22-nonaene which overcome or ameliorate one or more of the above identified problems.
Figure 22 shows a 1H NMR spectrum for Batch 4 in d6-DMSO.
Figure 23 shows a 1H NMR spectrum for Batch 4 in D2O.
List of hydrochloride and citrate salt batches used for comparative studies

Example 4 – Formation of the Citrate salt (Batch 4) in THF as solvent:
The free base of compound 1 (0.30Og, 0.648mmoles, 1.eq) was added to 12mL of THF. The solution was heated to reflux until complete dissolution was observed and maintained for 1h. A solution of citric acid (0.149g, 0.778mmoles, 1.2eq) dissolved in 12mL THF was then added slowly at reflux conditions. The mixture was refluxed for a further 15min then cooled. Crystallization was observed on gradual cooling. The crystals were stirred at room temperature for 12h and filtered under vacuum. The product was dried under vacuum to afford 250mg.
PATENT
http://www.google.im/patents/WO2007058627A1?cl=en
Representative procedure for the synthesis of compounds type (XVIIIf)
5-(2-Chloro-pyrimidin-4-yl)-furan-2-carbaldehyde (XIIIfI) 
(XIIfI) (XIIIH) .
Compound (XIIIfI) was obtained using the same procedure described for compound (XIIIeI); LC-MS (ESI positive mode) /τVz 209 ([M+H]+)
[5-(2-Chloro-pyrimidin-4-yl)-furan-2-yl]-methanol (Xlllf2)

Compound (Xlllf2) was obtained using the same procedure described for compound (XXIb); LC-MS (ESI positive mode) m/z 211 ([M+H]+).
4-(5-Allyloxymethyl-furan-2-yl)-2-chloro-pyrimidine (XVfI)

Compound (XVfI) was obtained using the same procedure described for compound (XXIIb); LC-MS (ESI positive mode) m/z 251 ([M+H]+).
^-(S-Allyloxymethyl-furan-Σ-yO-pyrimidin^-yll-IS-allyloxymethyl^^-pyrrolidin-i-yl- ethoxy)-phenyl]-amine (XVIIfI)


(XVIb2) (XVIIfI)
Compound (XVIIfI) was obtained using the same procedure described for compound (XVIIbI); LC-MS (ESI positive mode) m/z 491.
Macrocycle Example 6 (Compound 38)

(XVIIfI)

Compound (38) was obtained using the same procedure described for compound (1) HPLC purity at 254nm: 99%; LC-MS (ESI positive mode) m/z 463 ([M+H]+); 1H NMR (MeOD-d4) δ 8.90 (d, 1 H), 8.33 (d, 1 H), 7.37 (d, 1 H), 7.17 (d, 1 H), 7.14-7.11 (m, 1 H)1 7.04 (d, 1 H), 6.67 (d, 1 H), 6.04 (dt, 1 H, CH, J = 5.2Hz, Jtrans = 15.8Hz), 5.96 (dt, 1 H, CH, J = 5.0Hz, Jtrans = 15.8Hz), 4.65 (s, 2H), 4.62 (s, 2H), 4.37 (t, 2H), 4.14 (d, 2H), 4.09 (d, 2H), 3.81 (br s, 2H), 3.66 (t, 2H), 3.33 (s, 2H), 2.21-1.98 (m, 4H).
PAPER
Discovery of the Macrocycle (9E)-15-(2-(Pyrrolidin-1-yl)ethoxy)-7,12,25-trioxa-19,21,24-triaza-tetracyclo[18.3.1.1(2,5).1(14,18)]hexacosa-1(24),2,4,9,14(26),15,17,20,22-nonaene (SB1578), a Potent Inhibitor of Janus Kinase 2/Fms-LikeTyrosine Kinase-3 (JAK2/FLT3) for the Treatment of Rheumatoid Arthritis
http://pubs.acs.org/doi/abs/10.1021/jm201454n

Herein, we describe the synthesis and SAR of a series of small molecule macrocycles that selectively inhibit JAK2 kinase within the JAK family and FLT3 kinase. Following a multiparameter optimization of a key aryl ring of the previously described SB1518 (pacritinib), the highly soluble 14l was selected as the optimal compound. Oral efficacy in the murine collagen-induced arthritis (CIA) model for rheumatoid arthritis (RA) supported 14l as a potential treatment for autoimmune diseases and inflammatory disorders such as psoriasis and RA. Compound 14l (SB1578) was progressed into development and is currently undergoing phase 1 clinical trials in healthy volunteers.
(9E)-15-(2-(Pyrrolidin-1-yl)ethoxy)-7,12,25-trioxa-19,21,24-triaza-tetracyclo[18.3.1.1(2,5).1(14,18)]hexacosa-1(24),2,4,9,14(26), 15,17,20,22-nonaene (14l)
REF
Madan B, Goh KC, Hart S, William AD, Jayaraman R, Ethirajulu K, Dymock BW, Wood JM. SB1578, a novel inhibitor of JAK2, FLT3, and c-Fms for the treatment of rheumatoid arthritis. J Immunol. 2012 Oct 15;189(8):4123-34. doi: 10.4049/jimmunol.1200675. Epub 2012 Sep 7. PubMed PMID: 22962687.
2: Poulsen A, William A, Blanchard S, Lee A, Nagaraj H, Wang H, Teo E, Tan E, Goh KC, Dymock B. Structure-based design of oxygen-linked macrocyclic kinase inhibitors: discovery of SB1518 and SB1578, potent inhibitors of Janus kinase 2 (JAK2) and Fms-like tyrosine kinase-3 (FLT3). J Comput Aided Mol Des. 2012 Apr;26(4):437-50. doi: 10.1007/s10822-012-9572-z. Epub 2012 Apr 22. PubMed PMID: 22527961.
3: William AD, Lee AC, Poulsen A, Goh KC, Madan B, Hart S, Tan E, Wang H, Nagaraj H, Chen D, Lee CP, Sun ET, Jayaraman R, Pasha MK, Ethirajulu K, Wood JM, Dymock BW. Discovery of the macrocycle (9E)-15-(2-(pyrrolidin-1-yl)ethoxy)-7,12,25-trioxa-19,21,24-triaza-tetracyclo[18. 3.1.1(2,5).1(14,18)]hexacosa-1(24),2,4,9,14(26),15,17,20,22-nonaene (SB1578), a potent inhibitor of janus kinase 2/fms-like tyrosine kinase-3 (JAK2/FLT3) for the treatment of rheumatoid arthritis. J Med Chem. 2012 Mar 22;55(6):2623-40. doi: 10.1021/jm201454n. Epub 2012 Mar 6. PubMed PMID: 22339472.
| WO2007058627A1 * | 15 Nov 2006 | 24 May 2007 | S Bio Pte Ltd | Oxygen linked pyrimidine derivatives |
| SG2006000352W | Title not available |



AUTHOR’S
• Principle lead and inventor of 3 clinical stage candidates,
1) SB1518 (Pacritinib)-A selective JAK2 inhibitor for myleofibrosis into phase 2,
2) SB1317 (TG02)-A mutikinase inhibitor CDK, JAK2, FLT3, and ERK5 into phase 1 and
3) SB1578-A more selective JAK2 inhibitor than pracritinib for autoimmune diseases such as Rheumatoid Arthritis (RA) and Psoriasis into phase 1
NEXT………..

Babita Madan
DUKE NUS Graduate Medical School
Experience
Asst. Professor
Duke NUS Graduate Medical Centre
December 2011 – Present (4 years 2 months)Singapore
Scientist
S*BIO Pte Ltd
January 2010 – October 2011 (1 year 10 months)Singapore
Senior Research Fellow
University Clinics Ulm, Germany
November 2002 – December 2008 (6 years 2 months)
Education

SEE……..http://apisynthesisint.blogspot.in/2016/01/sb1578-onx-0805.html
///////
N3=C1NC(=CC=N1)c2oc(cc2)COCC=CCOCc5cc3ccc5OCCN4CCCC4
OR
C1(C2=CC=C(O2)COC/C=C/COCC3=CC(N4)=CC=C3OCCN5CCCC5)=NC4=NC=C1

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO