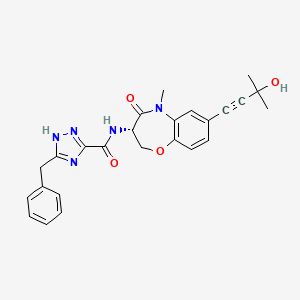
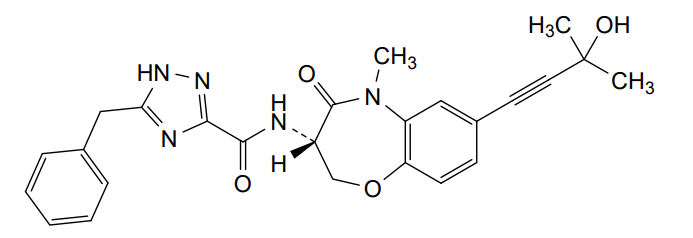
Ocadusertib
CAS 2382811-41-6
MF C25H25N5O4 MW 459.5 g/mol
5-benzyl-N-[(3S)-7-(3-hydroxy-3-methylbut-1-ynyl)-5-methyl-4-oxo-2,3-dihydro-1,5-benzoxazepin-3-yl]-1H-1,2,4-triazole-3-carboxamide
5-benzyl-N-[(3S)-7-(3-hydroxy-3-methylbut-1-yn-1-yl)-5-methyl-4-oxo-2,3,4,5-tetrahydro-1,5-benzoxazepin-3-yl]-1H-1,2,4-triazole-3-carboxamide
serine/threonine kinase inhibitor, LY3871801, R552, LY 3871801, R 552, S53J4A7ME4
- OriginatorRigel Pharmaceuticals
- DeveloperEli Lilly and Company; Rigel Pharmaceuticals
- ClassAnti-inflammatories; Antirheumatics; Small molecules
- Mechanism of ActionRIPK1 protein inhibitors
- Phase IIRheumatoid arthritis
- No development reportedUnspecified
- 28 Mar 2025No recent reports of development identified for phase-I development in Unspecified(In volunteers) in Singapore (PO, Suspension)
- 14 Nov 2024Pharmacodynamics data from preclinical trials in Rheumatoid arthritis presented at the ACR Convergence 2024 (ACR-2024)
- 14 Nov 2024Safety and pharmacokinetics data from a phase I trial in Rheumatoid arthritis presented at the ACR Convergence 2024 (ACR-2024)
Ocadusertib (LY3871801/R552) is an oral, potent, and selective small-molecule RIPK1 inhibitor developed by Rigel Pharmaceuticals and Eli Lilly for autoimmune and inflammatory diseases. It is currently in Phase 2 clinical trials for treating moderate-to-severe rheumatoid arthritis. ACR Meeting Abstracts +4
Key Aspects of Ocadusertib:
- Mechanism of Action: It inhibits receptor-interacting serine/threonine-protein kinase 1 (RIPK1), which blocks necroptotic (cell death) responses and, consequently, reduces inflammation.
- Target Indications: Primarily focused on rheumatoid arthritis, it has also been investigated for psoriasis and general inflammatory joint conditions.
- Development Status: As of late 2025, it is in Phase 2 clinical trials (NCT05848258), with previous trials evaluating its safety, tolerability, and pharmacokinetics in healthy volunteers.
- Characteristics: It is designed to be a selective inhibitor, showing no significant inhibition in a broad panel of other kinases.
ACR Meeting Abstracts +3
Ocadusertib is a small molecule drug. The usage of the INN stem ‘-sertib’ in the name indicates that Ocadusertib is a serine/threonine kinase inhibitor. Ocadusertib is under investigation in clinical trial NCT05848258 (An Adaptive Phase 2a/2b Study of LY3871801 in Adult Participants With Rheumatoid Arthritis). Ocadusertib has a monoisotopic molecular weight of 459.19 Da.
- An Adaptive Phase 2a/2b Study of LY3871801 in Adult Participants With Rheumatoid ArthritisCTID: NCT05848258Phase: Phase 2Status: RecruitingDate: 2025-12-09
- A Study of LY3871801 in Healthy Asian and Non-Asian ParticipantsCTID: NCT05960851Phase: Phase 1Status: CompletedDate: 2024-01-10
- A Drug Interaction Study of LY3871801 in Healthy ParticipantsCTID: NCT05602675Phase: Phase 1Status: CompletedDate: 2023-04-18
- A Study of LY3871801 in Healthy ParticipantsCTID: NCT05222399Phase: Phase 1Status: CompletedDate: 2022-03-18
SYN
PAT
PAT
WO 2014/125444
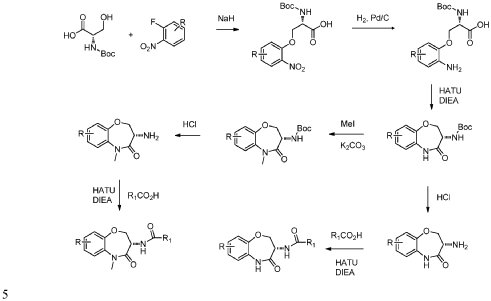
PAT
- [WO2021046407]
- https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2021046407&_cid=P20-MM02H8-46041-1
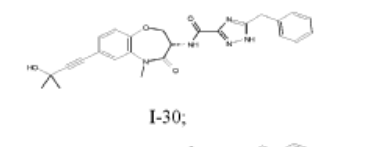
I-30: (S)-5-benzyl-N-(7-(3-hydroxy-3-methylbut-1-yn-1-yl)-5-methyl-4-oxo-2,3,4,5-tetrahydrobenzo[b][1,4]oxazepin-3-yl)-1H-1,2,4-triazole-3-carboxamide;
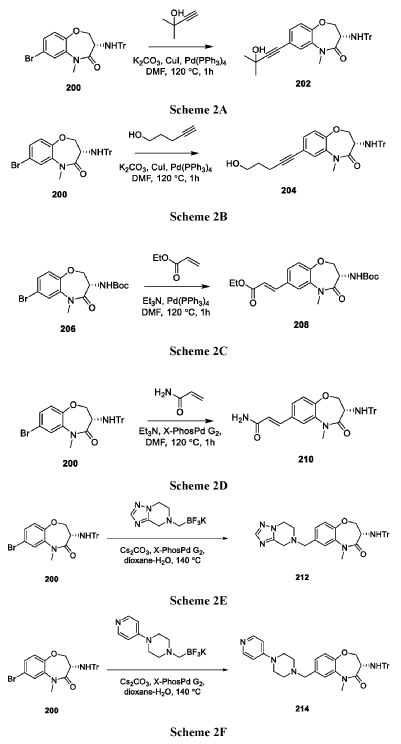
(S)-5-benzyl-N-(5-methyl-4-oxo-2,3,4,5-tetrahydrobenzo[b][1,4]oxazepin-3-yl)-4H-1,2,4-triazole-3-carboxamide (WO 2014/125444), having a structure as illustrated below, was used as a comparative compound and was examined using a similar protocol as described by WO 2014/125444. This comparison
compound exhibited 93% inhibition at a dose of 30 mg/kg according to WO 2014/125444; however, in the inventors hands, the compound inhibited only 70% at 30 mg/kg. In comparison, compound I-30 of the present disclosure achieved greater than 85% inhibition at a dose of just 5 mg/kg using the similar assay protocol described above.
PAT
- RIP1 inhibitory compounds and methods of making and using the samePublication Number: CN-112368278-BPriority Date: 2018-05-03Grant Date: 2025-06-17
- Rip1 inhibitory compounds and methods for making and using the samePublication Number: US-2021002237-A1Priority Date: 2018-05-03
- RIP1 inhibitory compounds and methods for making and using the samePublication Number: AU-2019262144-A1Priority Date: 2018-05-03
- Rip1 inhibitory compounds and methods for making and using the samePublication Number: US-2021002236-A1Priority Date: 2018-05-03
- Rip1 inhibitory compounds and methods for making and using the samePublication Number: US-2021040053-A1Priority Date: 2018-05-03
- RIP1-inhibiting compounds and methods of making and using the samePublication Number: CN-112368278-APriority Date: 2018-05-03
- Rip1 inhibitory compounds and methods for making and using the samePublication Number: US-2023113841-A1Priority Date: 2018-05-03
- RIP1 inhibitory compounds and methods for making and using the samePublication Number: US-11370765-B2Priority Date: 2018-05-03Grant Date: 2022-06-28
- RIP1 inhibitory compounds and methods for making and using the samePublication Number: US-11377428-B2Priority Date: 2018-05-03Grant Date: 2022-07-05
- Rip1 inhibitory compounds and methods for making and using the samePublication Number: US-2021238153-A2Priority Date: 2018-05-03
- Rip1 inhibitory compounds and methods for making and using the samePublication Number: EP-4248975-A2Priority Date: 2018-05-03
- RIP1 inhibitory compounds and methods for making and using the samePublication Number: US-12116354-B2Priority Date: 2018-05-03Grant Date: 2024-10-15
- RIP1 inhibitory compounds and methods for making and using the samePublication Number: US-11332451-B2Priority Date: 2018-05-03Grant Date: 2022-05-17
- RIP1 inhibitory compounds and methods for making and using the samePublication Number: US-11370764-B2Priority Date: 2018-05-03Grant Date: 2022-06-28
- RIP1 inhibitory compounds and methods of obtaining and using themPublication Number: MD-3788044-T2Priority Date: 2018-05-03
- Rip1 inhibitory compounds and methods for making and using the samePublication Number: US-2019337907-A1Priority Date: 2018-05-03
- RIP1 inhibitory compounds and methods for making and using the samePublication Number: US-10815206-B2Priority Date: 2018-05-03Grant Date: 2020-10-27
- Rip1 inhibitory compounds and methods for making and using the samePublication Number: US-2021009537-A1Priority Date: 2018-05-03
- Rip1 inhibitory compounds and methods for making and using the samePublication Number: US-2020407332-A1Priority Date: 2018-05-03
- Rip1 inhibitory compounds and methods for making and using the samePublication Number: EP-3788044-A1Priority Date: 2018-05-03



AS ON OCT2025 4.511 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

///////ocadusertib, serine/threonine kinase inhibitor, LY3871801, R552, LY 3871801, R 552, S53J4A7ME4

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
