The latest set of opinions from advisors to the European Medicines Agency include recommendations to approve six new medicines, including Bristol-Myers Squibb’s new hepatitis C drug and Eli Lilly’s biosimilar of the Sanofi diabetes blockbuster Lantus.
BMS 587101…….The LFA-1 receptor antagonist in preclinical for the treatment of a variety of autoimmune and inflammatory diseases such as rheumatoid arthritis and psoriasis.

- C26H20Cl2N4O4S
- mass: 555.432373 Da
read poster
http://www.cerep.fr/cerep/users/pages/news/Publications/123.pdf
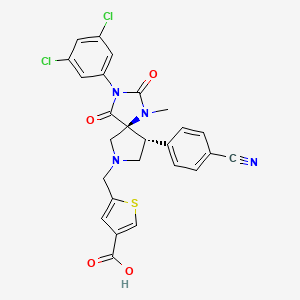
5-[(5S,9R)-9-(4-Cyanophenyl)-3-(3,5-dichlorophenyl)-1-methyl-2,4-dioxo-1,3,7-triazaspiro[4.4]non-9-ylmethyl]-thiophene-3-carboxylic Acid
3-Thiophenecarboxylic acid, 5-[[(5S,9R)-9-(4-cyanophenyl)-3-(3,5-dichlorophenyl)-1-methyl-2,4-dioxo-1,3,7-triazaspiro[4.4]non-7-yl]methyl]- [ACD/Index Name]
5-{[(5S,9R)-9-(4-Cyanophenyl)-3-(3,5-dichlorophenyl)-1-methyl-2,4-dioxo-1,3,7-triazaspiro[4.4]non-7-yl]methyl}-3-thiophenecarboxylic acid [ACD/IUPAC Name]
5-{[(5S,9R)-9-(4-Cyanphenyl)-3-(3,5-dichlorphenyl)-1-methyl-2,4-dioxo-1,3,7-triazaspiro[4.4]non-7-yl]methyl}-3-thiophencarbonsäure [German] [ACD/IUPAC Name]
Acide 5-{[(5S,9R)-9-(4-cyanophényl)-3-(3,5-dichlorophényl)-1-méthyl-2,4-dioxo-1,3,7-triazaspiro[4.4]non-7-yl]méthyl}-3-thiophènecarboxylique [French] [ACD/IUPAC Name]
2IC
BMS-587101
BMS-688521
data
Interaction between leukocyte function-associated antigen-1 (LFA-1), expressed on the surface of cytokine-stimulated cells, and intercellular adhesion molecule (I-CAM), found on the surface of both leukocytes and endothelium, plays a key function in the intercellular immune response, causing T-cell adhesion and subsequent migration through the blood vessel wall to the inflamed area.(1)
Small molecules which inhibit the LFA-1/I-CAM interaction are targeted as potential drugs for the treatment of a variety of autoimmune and inflammatory diseases such as rheumatoid arthritis and psoriasis.(2, 3) The LFA-1 receptor antagonist, BMS-587101, 1,(4, 5) was selected for clinical development, and we required a synthesis that would reliably generate kilogram quantities of API. This paper details the identification and development of a synthesis which enabled the realization of this goal.
BMS-587101 inhibits the interaction between leukocyte function-associated antigen-1 (LFA-1) and the intercellular adhesion molecule (ICAM), thereby offering a potential treatment for various autoimmune and inflammatory diseases, such as rheumatoid arthritis and psoriasis. A four-step multikilogram route to BMS-587101 (22% overall yield ) from the commercial hydantoin B features an efficient dipolar cycloaddition of an azomethine ylide generated by reaction of glycine with hexamethylenetetramine (HMTA).
………….
paper

http://pubs.acs.org/doi/abs/10.1021/op9003168
The process development and the kilogram-scale synthesis of BMS-587101 (1) are described. The synthesis features a [3 + 2] azomethine ylide cycloaddition to efficiently build the spirocyclic core in a diastereoselective fashion followed by a classical resolution which affords the desired enantiomer in >98% enantiomeric excess. The target was prepared in four steps in an overall yield of 22%.
 22.9 kg) was added until a pH of 6.5 was attained. After agitating for 15 min and holding for 30 min, the aqueous layer was discarded, and the organic layer was washed with H2O (470 kg). The solution was then polish filtered, and isopropylacetate (52.2 kg) was used to rinse the polish filter assembly. The solution was concentrated under reduced pressure (240 Torr) to a volume of 718 L at <45 °C. Seeds (500 g) were charged, and the distillation was continued until a volume of
22.9 kg) was added until a pH of 6.5 was attained. After agitating for 15 min and holding for 30 min, the aqueous layer was discarded, and the organic layer was washed with H2O (470 kg). The solution was then polish filtered, and isopropylacetate (52.2 kg) was used to rinse the polish filter assembly. The solution was concentrated under reduced pressure (240 Torr) to a volume of 718 L at <45 °C. Seeds (500 g) were charged, and the distillation was continued until a volume of  207 L was attained. Heptane (117.8 kg) was charged, the slurry was cooled to 20 °C over 1.5 h and was subsequently wet milled until d90 < 60 μm. The slurry was held for >2 h and filtered. The cake was washed with a 1:1 isopropyl acetate/heptane solution (109.7 kg) isopropyl acetate and dried in vacuum at 35−40 °C to a constant weight. Acid 1 (39.6 kg, 91.5% yield and 99.33 HPLC area % purity) was obtained as a white and sandy crystalline solid.
207 L was attained. Heptane (117.8 kg) was charged, the slurry was cooled to 20 °C over 1.5 h and was subsequently wet milled until d90 < 60 μm. The slurry was held for >2 h and filtered. The cake was washed with a 1:1 isopropyl acetate/heptane solution (109.7 kg) isopropyl acetate and dried in vacuum at 35−40 °C to a constant weight. Acid 1 (39.6 kg, 91.5% yield and 99.33 HPLC area % purity) was obtained as a white and sandy crystalline solid.
…………………………
U.S. Patent 7,381,737 B2
http://www.google.com/patents/US7381737
IIIn:
Also provided are crystalline forms of solvates and salts of the substituted spiro-hydantoin compound (IIIn).
5-[(5S,9R)-9-(4-Cyanophenyl)-3-(3,5-dichlorophenyl)-1-methyl-2,4-dioxo-1,3,7-triazaspiro[4.4]non-7-ylmethyl]-thiophene-3-carboxylic acid.
EXAMPLES
The following examples illustrate embodiments of the inventive process, and are not intended to limit the scope of the claims. For ease of reference, the following abbreviations are used herein:
ABBREVIATIONS
- DMSO=dimethyl sulfoxide
- DTTA=(+)-Di-p-toluoyl-D-tartaric acid
Preparation 13-(3,5-dichlorophenyl)-1-methylimidazolidine-2,4-dione
Triethylamine (0.78 kg, 7.75 mol) was added in 15-30 minutes with stirring to a thin suspension of sarcosine ethylene hydrochloride (1.00 kg, 6.51 mol) in dichloromethane (6.00 L). After stirring at room temperature for 1.5-2.0 hours, the mixture was filtered to remove the resulting triethylamine hydrochloride salt. The salt cake was washed with dichloromethane (2.00 L). The filtrate was cooled to 0-5° C.
A solution of 3,5-dichlorophenyl isocyanate (1.47 kg, 7.81 mol) in dichloromethane was prepared at 20-25° C. The solution was added to the above cooled filtrate slowly in 30-60 minutes. The temperature was maintained below 10° C. during the addition. After the addition, the mixture was stirred at 20-25° C. for 12-14 hours. The completeness of the reaction was followed by HPLC. Upon reaction completion, TBME (16.00 L) was added in one portion. The resulting suspension was stirred at 20-25° C. for 2-3 hours and was then filtered. The filter cake was washed with TBME (4.50 L) and dried at maximum 40° C. to a constant weight. A suspension of the above filter cake in water (17.0 L, 10 L/kg input) was prepared and stirred at 20-25° C. for at least 16 hours. The suspension was filtered and the filter cake was washed with water (3×1.36 L) and dried at maximum 40° C. to a constant weight to a constant weight. 3-(3,5-dichlorophenyl)-1-methylimidazolidine-2,4-dione (1.52 kg, 90%) was obtained as a white crystalline solid. mp=202-204° C. 1H NMR (DMSO-d6): 7.66 (1H, m), 7.51 (2H, m), 4.10 (2H, s), 3.35 (3H, s). 13C NMR (DMSO-d6): 8 Carbons (169.30, 155.00, 134.98, 134.15, 127.59, 125.30, 51.75, 29.79). Anal. Calcd for C10H8Cl2N2O2: C, 46.35; H, 3.11; N, 10.81; Cl, 27.36. Found: C, 46.43; H, 2.9; N, 10.73; Cl, 27.33.
Preparation 2(E)-4-((1-(3,5-dichlorophenyl)-3-methyl-2,5-dioxoimidazolidin-4-ylidene)methyl)benzonitrile
A mixture of 3-(3,5-dichlorophenyl)-1-methylimidazolidine-2,4-dione (1.00 kg, 3.86 mol), 4-cyanobenzaldehyde (0.70 kg, 5.79 mol) and pyrrolidone (0.27 kg, 3.86 mmol) was refluxed in EtOH (13.00 L) for 20-24 hours at a temperature of 78° C. The completeness of the reaction was followed by HPLC. Upon reaction completion, the suspension was cooled to 65° C. and THF (4.33 L) was added in 5-10 minutes. The suspension was cooled to 20-25° C. in 3-4 hours and was then filtered. The filter cake was washed with EtOH (4×2.00 L) and dried at maximum 40° C. to a constant weight. (E)-4-((1-(3,5-dichlorophenyl)-3-methyl-2,5-dioxoimidazolidin-4-ylidene)methyl)benzonitrile (1.24 kg, 86%) was obtained as a fluffy, yellowish crystalline solid. mp=239-241° C. 1H NMR (DMSO-d6): 8.07 (2H, d, J=8.3 Hz), 7.86 (2H, d, J=8.4 Hz), 7.72 (1H, m), 7.59 (2H, m), 6.72 (1H, s), 3.35 (3H, s). 13C NMR (DMSO-d6): 14 Carbons (159.80, 151.48, 137.64, 133.83, 133.70, 131.80, 130.80, 130.68, 127.71, 125.51, 118.83, 114.48, 110.32, 26.72). Anal. Calcd for C18H11Cl2N3O2: C, 58.08; H, 2.97; N, 11.29; Cl, 19.05. Found: C, 58.14; H, 2.72; N, 11.14; Cl, 19.15.
Example 14-[(5S*,9R*)-3-(3,5-Dichlorophenyl)-1-methyl-2,4-dioxo-1,3,7-triazaspiro[4.4]non-9-yl]-benzonitrile hydrochloride salt
A mixture of (E)-4-((1-(3,5-dichlorophenyl)-3-methyl-2,5-dioxoimidazolidin-4-ylidene)methyl)benzonitrile (1.00 kg, 2.69 mol), glycine (0.50 kg, 6.72 mol) and hexamethylenetetramine (0.28 kg, 2.02 mol) in 1-methyl-2-pyrrolidinone (5.00 L) and toluene (2.50 L) was heated at 140° C. for 7-8 hours. The completeness of the reaction was followed by HPLC. Upon reaction completion, the mixture was cooled to 40-50° C. and filtered. The filtered solid was washed with toluene (0.67 L). To the filtrate was added HCl (1M, 13.33 L, 13.33 mol). The resulting biphasic mixture was heated to 50-60° C. and was stirred for 10-15 minutes. The aqueous phase was separated and the organic phase was washed with HCl (1M, 1.67 L, 1.67 mol) at 60-80° C. The aqueous phases were combined and were stirred at 80° C. for 2 hours. The solution was cooled slowly in 3-4 hours to 20-25° C. with gentle stirring and seeding. Crystallization occurred and the resulting suspension was put aside at 20-25° C. for at least 16 hours with occasional stirring, cooled to 0-5° C. in 2 hours, stirred gently at 0-5° C. for 2 hours and then filtered. The filter cake was washed with ice water (2×2.50 L) and dried at maximum 40° C. to a constant weight. 4-[(5S*,9R*)-3-(3,5-Dichlorophenyl)-1-methyl-2,4-dioxo-1,3,7-triazaspiro[4.4]non-9-yl]-benzonitrile hydrochloride salt (1.09 kg, 90%) was obtained as beige crystalline solid. mp=183-185° C. 1H NMR (DMSO-d6): 7.87(2H, d, J=8.1 Hz), 7.61 (1H, m), 7.40 (2H, d, J=8.1 Hz), 6.68 (2H, m), 4.17 (1H, m), 3.85 (2H, m), 3.76 (2H, m), 3.43 (3H, s), 3.24(2H, s). 13C NMR (DMSO-d6): 14 Carbons (170.84, 152.92, 137.35, 133.94, 132.87, 132.35, 128.01, 124.50, 118.12, 111.30, 71.42, 46.57, 45.11, 25.51). Anal. Calcd for C20H17Cl3N4O2+1.3 H2O: C, 50.51; H, 3.91; N, 11.79; Cl, 22.39. Found: C, 50.56; H, 3.86; N, 11.58; Cl, 21.98; KF, 5.12.
Example 2a4-[(5S,9R)-3-(3,5-Dichlorophenyl)-1-methyl-2,4-dioxo-1,3,7-triazaspiro[4.4]non-9-yl]-benzonitrile semi (+)-DTTA salt
To a suspension of 4-[(5S*,9R*)-3-(3,5-Dichlorophenyl)-1-methyl-2,4-dioxo-1,3,7-triazaspiro[4.4]non-9-yl]-benzonitrile hydrochloric acid salt (1.00 kg, 2.21 mol) in dichloromethane (10.67 L) was added diispopropylethylamine (0.29 kg, 2.21 mol). The mixture was stirred to a clear solution, to which (+)-Di-p-toluoyl-D-tartaric acid (0.21 kg, 0.55 mol) was added. The resulting solution was warmed to 34-36° C. and seeded immediately. It was cooled to 20-25° C. in 1.5-2.0 hours. Crystallization occurred during cooling. TBME (2.75 L) was added in 0.5 hours. The suspension was stirred at 20-25° C. for 16 hours and then filtered. The filter cake was washed with dichloromethane/TBME (2/1, 1.00 L), TBME (1 L) and dried at maximum 35° C. to a constant weight. 4-[(5S,9R)-3-(3,5-Dichlorophenyl)-1-methyl-2,4-dioxo-1,3,7-triazaspiro[4.4]non-9-yl]-benzonitrile semi (+)-DTTA salt (0.47 kg, 35%) was obtained as a white crystalline solid. mp=175-177° C. 1H NMR (DMSO-d6): 7.86 (2H, d, J=8.1 Hz), 7.81 (2H, d, J=8.3 Hz), 7.61 (1H, m), 7.28 (2H, d, J=8.1 Hz), 7.22 (2H, 8.5 Hz), 6.68 (2H, m), 5.71 (1H, s), 3.81(1H, m), 3.50 (4H, m), 3.06 (3H, s), 2.34 (3H, s). 13C NMR (DMSO-d6): 24 Carbons (171.45, 169.40, 165.04, 152.88, 143.61, 138.99, 133.88, 133.08, 132.16, 129.26, 129.20, 128.76, 127.84, 126.99, 124.51, 118.25, 110.78, 72.81, 73.38, 48.15, 47.51, 46.30, 24.90, 21.14). Anal. Calcd for C30H25Cl2N4O6+0.5 H2O: C, 58.40; H, 4.17; N, 9.08; Cl, 11.49. Found C, 58.58; H, 4.06; N, 8.94; Cl, 11.38; KF, 1.59.
Example 2b4-[(5S,9R)-3-(3,5-Dichlorophenyl)-1-methyl-2,4-dioxo-1,3,7-triazaspiro[4.4]non-9-yl]-benzonitrile semi (+)-DTTA salt
A mixture of (E)-4-((1-(3,5-dichlorophenyl)-3-methyl-2,5-dioxoimidazolidin-4-ylidene)methyl)benzonitrile (10.0 g, 26.9 mmol), glycine (5.06 g, 67.4 mmol), hexamethylenetetramine (2.82 g, 20.1 mmol) in 50 mL N-methylpyrrolidinone and 25 mL of toluene under nitrogen was heated to 138° C. for approximately 12 h. Next, 25 mL toluene and 25 mL H2O were added. The aqueous and nonaqueous layers were split, and the aqueous layer was washed with 25 mL of toluene, and the nonaqueous layers were combined to form a nonaqueous mixture. The nonaqueous mixture was heated to 45-50° C. and ethylene diamine (7.0 mL) was added. The nonaqueous mixture was stirred for 3 hours and then cooled to room temperature. Next, 50 mL H2O was added, followed by the addition of 10 mL brine. The next addition was 25 mL toluene, which was followed by the addition of 125 mL CH2Cl2. The bottom layer of the mixture was removed through a filter. Next, (+)-Di-p-toluoyl-D-tartaric acid (2.59 g, 6.7 mmol) was added and the mixture was stirred for 18 h to form a slurry. Slowly 40 mL of MTBE was added to the slurry. A wash solution containing 7 mL of MTBE and 11 mL of CH2Cl2 was prepared. Filter paper was wetted with 1 mL of the wash solution. The slurry was filtered and then the filtered to form a cake. The filter, the wash reaction flask, and the cake were washed with the remaining 16 mL of the wash solution. Next, the cake was washed with 10 mL MTBE. 4-[(5S, 9R)-3-(3,5-Dichlorophenyl)-1-methyl-2,4-dioxo-1,3,7-triazaspiro[4.4]non-9-yl]-benzonitrile semi (+)-DTTA salt (4.0 g, 20% yield) was obtained as a white solid (98.7% HPLC AP and 98.3% ee).
Example 2c4-[(5S,9R)-3-3,5-Dichlorophenyl)-1-methyl-2,4-dioxo-1,3,7-triazaspiro[4,4]non-9-yl]-benzonitrile semi (+)-DTTA salt
A mixture of (E)-4-((1-(3,5)-dichlorophenyl)-3-methyl-2,5-dioxoimidazolidin-4-ylidene)methyl)benzonitrile (40.0 g, 107.5 mmol), glycine (19.76 g, 263.2 mmol), hexamethylenetetramine (9.07 g, 64.7 mmol) in 200 mL N-methyl-2-pyrrolidinone and 100 mL of toluene was heated under nitrogen to 143° C. for approximately 5.5 h. Next, the mixture was cooled to 50° C. and a solution of 25 mL of ethylenediamine in 200 mL of tetrahydrofuran was added. The mixture was maintained at a temperature of 50° C. for 30 minutes and then was cooled to room temperature. Next, 520 mL of 20 wt % NaCl aqueous solution was added. The aqueous and nonaqueous layers were separated. The nonaqueous layer was transferred to a vacuum distillation apparatus and solvent was distilled off until the temperature of the residue in the flask reached 58° C. at a pressure of 60 torr. Next, 360 mL of methylene chloride was added, followed by the additions of 20 mL of methanol and 2 mL of water. The next addition was (+)-Di-p-toluoyl-D-tartaric acid (10.38 g, 26.9 mmol), followed by 120 mL of methylene chloride and 0.200 g of seeds of 4-[(5S,9R)-3-(3,5-Dichlorophenyl)-1-methyl-2,4-dioxo-1,3,7-triazaspiro[4,4]non-9-yl]-benzonitrile semi (+)-DTTA salt. A-slurry was formed and was stirred at room temperature for 24 hours. The slurry was filtered and the cake of crystals was washed with 200 mL of methylene chloride in two portions. The washed cake was then dried at 50° C. under vacuum for 24 hours. A total amount of 20.11 g (yield 31%) of 4-[(5S,9R)-3-(3,5-Dichlorophenyl)-1-methyl-2,4-dioxo-1,3,7-triazaspiro[4,4]non-9-yl]-benzonitrile semi (+)-DTTA salt, which was of greater than 99.5% area percent purity, 98.4% potency and 99.2% ee was obtained after drying.
Example 35-[(5S,9R)-9-(4-Cyanophenyl)-3-(3,5-dichlorophenyl)-1-methyl-2,4-dioxo-1,3,7-triazaspiro[4.4]non-7-ylmethyl]-thiophene-3-carboxylic acid methyl ester hydrochloride salt
To a suspension of 4-[(5S,9R)-3-(3,5-Dichlorophenyl)-1-methyl-2,4-dioxo-1,3,7-triazaspiro[4.4]non-9-yl]-benzonitrile semi (+)-DTTA salt (7.50 kg, 12.30 mmol) and methyl 5-formylthiophene-3-carboxylate (2.2 kg, 13.10 mol) was added triethylamine (2.08 kg, 20.60 mol) at 20-25° C. The mixture was stirred to a clear solution, to which acetic acid (1.24 kg, 20.60 mol) was added. The resulting mixture was stirred at 20-25° C. for 1 hour and then cooled to 15° C. Solid sodium triacetoxyborohydride (1.31 kg, 6.17 mol) was added and the reaction mixture was stirred for 0.5 hours. The addition of sodium triacetoxyborohydride was repeated three more times. At the end, a total of 5.22 kg (24.7 mol) sodium triacetoxyborohydride was added in 2 hours. The reaction mixture was stirred at 20-25° C. for 16 hours. The completeness of the reaction was followed by HPLC. Upon reaction completion, TBME (48.1 L) was added to the resulting jelly reaction mixture. The mixture was washed with saturated sodium hydrogen carbonate solution (60.0 L×3). The combined aqueous phase was extracted with TBME (48.1 L). All organic layers were combined, washed with brine (48.1 L) and concentrated in vacuum to a volume of 10.6 L. Isopropanol (192.3 L) was added to the residue and the resulting oil precipitates were dissolved upon warming up to 70-75° C. The solvent volume was reduced to 160.0 L by distillation at 70-75° C. Concentrated HCl (1.5 L) was added at 75° C. in 10 minutes followed by the addition of seed crystals. Crystallization occurred upon cooling to 20-25° C. in 16 hours. The mixture was filtered. The cake was washed with isopropanol (9.6 L×2) and dried at maximum 40° C. to a constant weight. 5-[(5S,9R)-9-(4-Cyanophenyl)-3-(3,5-dichlorophenyl)-1-methyl-2,4-dioxo-1,3,7-triazaspiro[4.4]non-7-ylmethyl]-thiophene-3-carboxylic acid methyl ester hydrochloride salt (6.57 kg, 88.0%) was obtained as white crystalline solid. mp=204-207° C. 1H NMR (CDCl3): 14.22 (1H, b), 8.18 (1H, d, J=0.9 Hz), 7.86 (1H, m), 7.67 (2H, d, J=8.1 Hz), 7.24 (1H, m), 7.23 (2H, d, J=8.1 Hz), 6.67 (2H, m), 4.76 (2H, m), 4.46 (1H, m), 4.16 (1H, m), 4.02 (2H, m), 3.86 (3H, s), 3.75 (1H, m), 3.38 (3H, s). 13C NMR (CDCl3): 18 Carbons (171.24, 162.32, 152.98, 136.05, 135.27, 134.03, 132.83, 131.94, 130.46, 128.85, 128.56, 123.92, 117.52, 113.43, 71.13, 52.43, 52.22, 46.73). Anal. Calcd for C27H23Cl3N4O4S: C, 53.52; H, 3.83; N, 9.25; S, 5.29; Cl, 17.55. Found: C, 53.07; H, 3.69; N, 9.08; S, 5.23; Cl, 17.20.
Example 45-[(5S,9R)-9-(4-Cyanophenyl)-3-(3,5-dichlorophenyl)-1-methyl-2,4-dioxo-1,3,7-triazaspiro[4.4]non-7-ylmethyl]-thiophene-3-carboxylic acid
To a solution of 5-[(5S,9R)-9-(4-Cyanophenyl)-3-(3,5-dichlorophenyl)-1-methyl-2,4-dioxo-1,3,7-triazaspiro[4.4]non-7-ylmethyl]-thiophene-3-carboxylic acid methyl ester hydrochloride salt (20.00 g, 33.00 mmol) and 1,2-propanediol (5.0 g) in tetrahydrofuran (200 mL) and water (100 mL) was added slowly potassium hydroxide solution (0.85M, 116 mL) at 8-12° C. in 0.5 hours. The resulting biphasic mixture was stirred at 8-12° C. for 20-27 hours until the reaction was complete. The reaction mixture was washed with n-heptane (200 mL). The pH was adjusted to 6.5 with addition of water (100 mL) and acetic acid (2.5 mL). Tetrahydrofuran was removed under reduced pressure at internal temperature <40° C. The pH was adjusted to 4.5 with addition of isopropyl acetate (400 mL) and acetic acid (11 mL). After 10 minutes of stirring, the aqueous layer was separated and was extracted with isopropylacetate (200 mL). The organic layers were combined, washed with water (100 mL) and concentrated under reduced pressure to a volume of 190 mL at bath temperature <40° C. Crystallization occurred during concentration. The crystal slurry was stirred at 20-25° C. for 16 hours and was then filtered. The cake was washed with cold isopropylacetate (15 mL×3) and dried in vacuum at 35-40° C. to a constant weight.
5-[(5S,9R)-9-(4-Cyanophenyl)-3-(3,5-dichlorophenyl)-1-methyl-2,4-dioxo-1,3,7-triazaspiro[4.4]non-7-ylmethyl]-thiophene-3-carboxylic acid (14.35 g, 78.3%) was obtained as white and sandy crystalline solid.
mp=209-230° C. 1H NMR (Acetone-d6): 8.19 (1H, d, J=1.3 Hz), 7.76 (2H, d, J=8.4 Hz), 7.49 (2H, d, J=8.2 Hz), 7.43 (1H, d, J=1.0 Hz), 7.41 (1H, t, J=1.9 Hz), 6.87 (2H, d, J=1.9 Hz), 4.16 (1H, dd, J1=13.9 Hz J2=0.8 Hz), 4.10 (1H, dd, J1=11.7 Hz, J2=6.2 Hz), 3.99 (1H, d, J=14.0 Hz), 3.48(1H, d, J=10.6 Hz), 3.47 (1H, dd, J1=9.6 Hz, J2=6.2 Hz), 3.25 (3H, s), 3.24 (1H, dd, J1=9.6 Hz, J2=11.7 Hz), 3.01 (1H, d, J=11.3 Hz).
13C NMR (Acetone-d6): 22 Carbons (172.69, 163.7, 153.98, 144.55, 142.23, 135.26, 135.09, 134.41, 133.89, 132.96, 130.33, 128.27, 126.98, 125.18, 119.07, 112.44, 74.28, 59.09, 56.45, 54.33, 50.73, 25.75).
Anal. Calcd for C26H20Cl2N4O4S: C, 56.22; H, 3.62; N, 10.08; S, 5.77; Cl, 12.76. Found: C, 56.27; H, 3.20; N, 9.97; S, 5.65; Cl, 12.68.
…………………………..
paper
J. Med. Chem. 2006, 49, 6946
http://pubs.acs.org/doi/abs/10.1021/jm0610806

LFA-1 (leukocyte function-associated antigen-1), is a member of the β2-integrin family and is expressed on all leukocytes. This letter describes the discovery and preliminary SAR of spirocyclic hydantoin based LFA-1 antagonists that culminated in the identification of analog 8 as a clinical candidate. We also report the first example of the efficacy of a small molecule LFA-1 antagonist in combination with CTLA-4Ig in an animal model of transplant rejection.
http://pubs.acs.org/doi/suppl/10.1021/jm0610806/suppl_file/jm0610806si20060913_101747.pdf synthesis as compd 8
says
a white solid: Anal.RP-HPLCtR= 3.09min (method D, purity 99%);
………………….
U.S. Patent 7,199,125 B2
http://www.google.com/patents/US7199125
………………………..
.U.S. Patent 6,710,064 B2
http://www.google.com/patents/US6710064
………….
REFERENCES
-
For a discussion on the inhibition of LFA-1/ICAM-1as an approach to treating autoimmune diseases see:
Yusuf-Makagiansar, H.; Anderson, M. E.; Yakovleva, T. V.; Murray, J. S.; Siahaan, T. J. Medicinal Research Reviews 2002, 22, 146 -
2.
For a discussion of therapeutic options for treatment of psoriasis, see:
Gottlieb, A. B. J. Acad. Dermatol 2005, 53, S3Larson, R. S.; Davis, T.; Bologa, C.; Semenuk, G.; Vijayan, S.; Li, Y.; Oprea, T.; Chigaev, A.; Buranda, T.; Wagner, C. R.; Sklar, L. A. -
3.
For other small molecule LFA-1/ICAM-1 antagonists as potential drugs please see:
(a) Pei, Z.; Xin, Z.; Liu, G.; Li, Y.; Reilly, E. B.; Lubbers, N. L.; Huth, J. R.; Link, J. T.; von Geldern, T. W.; Cox, B. F.; Leitza, S.; Gao, Y.; Marsh, K. C.; DeVries, P.; Okasinski, G. F. J. Med. Chem. 2001, 44, 2913(b) Liu, G.; Huth, J. R.; Olejniczak, E. T.; Mendoza, R.; DeVries, P.; Leitza, S.; Reilly, E. B.; Olasinski, G. F.; Fesik, S. W.; von Geldern, T. W. J. Med. Chem. 2001, 44, 1202(c) Wu, J.-P.; Emeigh, J.; Gao, D. A.; Goldberg, D. R.; Kuzmich, D.; Miao, C.; Potocki, I.; Qian, K. C.; Sorcek, R. J.; Jeanfavre, D. D.; Kishimoto, K.; Mainolfi, E. A.; Nabozny, G.; Peng, C.; Reilly, P.; Rothlein, R.; Sellati, R. H.; Woska, J. R.; Chen, S.; Gunn, J. A.; O’Brien, D.; Norris, S. H.; Kelly, T. A. J. Med. Chem. 2004, 47, 5356(d) Last-Barney, K.; Davidson, W.; Cardozo, M.; Frye, L. L.; Grygon, C. a.; Hopkins, J. L.; Jeanfavre, D. D.; Pav, S.; Qian, C.; Stevenson, J. M.; Tong, L.; Zindell, R.; Kelly, T. A. J. Am. Chem. Soc. 2001, 123, 5643(e) Wang, G. T.; Wang, S.; Gentles, R.; Sowin, T.; Leitza, S.; Reilly, E. B.; von Geldern, T. W. Bioorg. Med. Chem. Lett. 2005, 15, 195(f) Wattanasin, S.; Albert, R.; Ehrhardt, C.; Roche, D.; Savio, M.; Hommel, U.; Welzenbach, K.; Weitz-Schmidt, G. Bioorg. Med. Chem. Lett. 2003, 12, 499 -
4.
The Discovery work towards this target compound BMS-587101 is described in:
Potin, D.; Launay, M.; Monatlik, F.; Malabre, P.; Fabreguettes, M.; Fouquet, A.; Maillet, M.; Nicolai, E.; Dorgeret, L.; Chevallier, F.; Besse, D.; Dufort, M.; Caussade, F.; Ahmad, S. Z.; Stetsko, D. K.; Skala, S.; Davis, P. M.; Balimane, P.; Patel, K.; Yang, Z.; Marathe, P.; Postelneck, J.; Townsend, R. M.; Goldfarb, V.; Sheriff, S.; Einspahr, H.; Kish, K.; Malley, M. F.; DiMarco, J. D.; Gougoutas, J. Z.; Kadiyala, P.; Cheney, D. L.; Tejwani, R. W.; Murphy, D. K.; Mcintyre, K. W.; Yang, X.; Chao, S.; Leith, L.; Xiao, Z.; Mathur, A.; Chen, B.-C.; Wu, D.-R.; Traeger, S. C.; McKinnon, M.; Barrish, J. C.; Robl, J. A.; Iwanowicz, E. J.; Suchard, S. J.; Dhar, M. T. G. J. Med. Chem. 2006, 49, 6946 -
5.
For additional information related to this compound see:
(a) Chen, B.-C.; DelMonte, A. J.; Dhar, T. G. M.; Fan, Y.; Gougoutas, J. Z.; Malley, M. F.; McLeod, D. D.; Waltermire, R.; Wei, C. Crystalline Forms and Process for Preparing Spiro-Hydantoin Compounds. (Bristol-Myers Squibb). U.S. Patent 7,381,737 B2 .(b) Dhar, T. G. M.; Potin, D.; Maillet, M.; Launay, M.; Nicolai, E.; Iwanowicz, E. Spiro-cyclic compounds useful as anti-inflammatory agents. Bristol-Myers Squibb and Cerep). U.S. Patent 7,199,125 B2.(c) Launay, M.; Potin, D.; Maillet, M.; Nicolai, E.; Dhar, T. G. M.; Iwanowicz, E. Hydantoin compounds useful as anti-inflammatory agents. (Bristol-Myers Squibb).U.S. Patent 6,710,064 B2.For the radiolabelled synthesis of BMS-587101 see:
Tran, S. B.; Maxwell, B. D.; Chen, S.-Y.; Bonacorsi, S. J.; Leith, L.; Ogan, M.; Rinehart, J. K.; Balasubramanian, B. J. Labelled Compd. Radiopharm. 2009, 52, 236
|
10-31-2008
|
CRYSTALLINE FORMS AND PROCESS FOR PREPARING SPIRO-HYDANTOIN COMPOUNDS
|
|
|
6-4-2008
|
Crystalline forms and process for preparing spiro-hydantoin compounds
|
|
|
3-7-2007
|
Pyridyl-substituted spiro-hydantoin compounds and use thereof
|
|
|
7-19-2006
|
Spiro-hydantoin compounds useful as anti-inflammatory agents
|
|
|
6-30-2006
|
Pyridyl-substituted spiro-hydantoin crystalline forms and process
|
|
|
12-21-2005
|
Spiro-hydantoin compounds useful as anti-inflammatory agents
|
| US8710058 * | Dec 4, 2009 | Apr 29, 2014 | Merck Patent Gmbh | Polymorphic forms of 3-(1-{3-[5-(1-methyl-piperidin-4-ylmethoxy)-pyrimidin-2-yl]-benzyl}-6-oxo-1,6-dihydro-pyridazin-3-yl)-benzonitrile hydrochloride salt and processes of manufacturing thereof |
| US20110269767 * | Dec 4, 2009 | Nov 3, 2011 | Merck Patent Gesellschaft Mit Beschrankter Haftung | Novel Polymorphic Forms of 3-(1–6-oxo-1,6-dihydro-pyridazin-3-yl)-benzonitrile Hydrochloride Salt and Processes of Manufacturing Thereof |
USPTO Guidance On Patentable Subject Matter: Impediment to Biotech Innovation?
USPTO Guidance On Patentable Subject Matter: Impediment to Biotech Innovation?
http://commercialbiotechnology.com/index.php/jcb/article/view/664
Abstract
In June 2013, the U.S. Supreme Court issued a unanimous decision upending more than three decades worth of established patent practice when it ruled that isolated gene sequences are no longer patentable subject matter under 35 U.S.C. Section 101.While many practitioners in the field believed that the USPTO would interpret the decision narrowly, the USPTO actually expanded the scope of the decision when it issued its guidelines for determining whether an invention satisfies Section 101. The guidelines were met with intense backlash with many arguing that they unnecessarily expanded the scope of the Supreme Court cases in a way that could unduly restrict the scope of patentable subject matter, weaken the U.S. patent system, and create a disincentive to innovation. By undermining patentable subject matter in this way, the guidelines may end up harming not only the companies that patent medical innovations, but also the patients who need medical care. This article examines the guidelines and their impact on various technologies.
FDA Guidance for Industry: Electronic Source Data in Clinical Investigations
FDA Guidance for Industry: Electronic Source Data in Clinical Investigations
The FDA published its new Guidance for Industry (GfI) – “Electronic Source Data in Clinical Investigations” in September 2013. The Guidance defines the expectations of the FDA concerning electronic source data generated in the context of clinical trials. Find out more about this Guidance.
|
FDA Guidance for Industry: Electronic Source Data in Clinical Investigations |
|
After more than 5 years and two draft versions, the final version of the Guidance for Industry (GfI) – “Electronic Source Data in Clinical Investigations” was published in September 2013. This new FDA Guidance defines the FDA’s expectations for sponsors, CROs, investigators and other persons involved in the capture, review and retention of electronic source data generated in the context of FDA-regulated clinical trials. In an effort to encourage the modernization and increased efficiency of processes in clinical trials, the FDA clearly supports the capture of electronic source data and emphasizes the agency’s intention to support activities aimed at ensuring the reliability, quality, integrity and traceability of this source data, from its electronic source to the electronic submission of the data in the context of an authorization procedure. The Guidance addresses aspects as data capture, data review and record retention. When the computerized systems used in clinical trials are described, the FDA recommends that the description not only focus on the intended use of the system, but also on data protection measures and the flow of data across system components and interfaces. In practice, the pharmaceutical industry needs to meet significant requirements regarding organisation, planning, specification and verification of computerized systems in the field of clinical trials. The FDA also mentions in the Guidance that it does not intend to apply 21 CFR Part 11 to electronic health records (EHR). Author: Source: |
Low levels of omega-3 fatty acids may cause memory problems
09 Mar 2012
ST. PAUL, Minn. – A diet lacking in omega-3 fatty acids, nutrients commonly found in fish, may cause your brain to age faster and lose some of its memory and thinking abilities, according to a study published in the February 28, 2012, print issue of Neurology®, the medical journal of the American Academy of Neurology. Omega-3 fatty acids include the nutrients called docosahexaenoic acid (DHA) and eicosapentaenoic acid (EPA).
View original post 225 more words
Biomarker predicts effectiveness of brain cancer treatment
Researchers at the University of California, San Diego School of Medicine have identified a new biomarker that predicts whether glioblastoma – the most common form of primary brain cancer – will respond to chemotherapy. The findings are published in the July print issue of Oncotarget.
“Every patient diagnosed with glioblastoma is treated with a chemotherapy called temozolomide. About 15 percent of these patients derive long-lasting benefit,” said Clark C. Chen, MD, PhD, vice-chairman of Academic Affairs, Division of Neurosurgery, UC San Diego School of Medicine and the study’s principal investigator. “We need to identify which patients benefit from temozolomide and which another type of treatment. All therapies involve risk and the possibility of side-effects. Patients should not undergo therapies if there’s no likelihood of benefit.”
To pinpoint which patients were most likely respond to temozolomide, the researchers studied microRNAs that control the expression of a protein called methyl-guanine-methyl-transferase…
View original post 163 more words
Amprenavir (Agenerase, GlaxoSmithKline) is a protease inhibitor…….

AMPRENAVIR
Amprenavir (Agenerase, GlaxoSmithKline) is a protease inhibitor used to treat HIV infection. It was approved by the Food and Drug Administration on April 15, 1999, for twice-a-day dosing instead of needing to be taken every eight hours. The convenient dosing came at a price, as the dose required is 1,200 mg, delivered in eight very large gel capsules.
Production of amprenavir was discontinued by the manufacturer December 31, 2004; a prodrug version (fosamprenavir) is available.
| Systematic (IUPAC) name | |
|---|---|
| (3S)-oxolan-3-yl N-[(2S,3R)-3-hydroxy-4-[N-(2-methylpropyl)(4-aminobenzene)sulfonamido]-1-phenylbutan-2-yl]carbamate | |
| Clinical data | |
| Trade names | Agenerase |
| AHFS/Drugs.com | monograph |
| MedlinePlus | a699051 |
| Licence data | EMA:Link, US FDA:link |
| Pregnancy cat. | C (US) |
| Routes | oral |
| Pharmacokinetic data | |
| Protein binding | 90% |
| Metabolism | hepatic |
| Half-life | 7.1-10.6 hours |
| Excretion | <3% renal |
| Identifiers | |
| CAS number | 161814-49-9 |
| ATC code | J05AE05 |
| PubChem | CID 65016 |
| DrugBank | DB00701 |
| ChemSpider | 58532 |
| UNII | 5S0W860XNR |
| KEGG | D00894 |
| ChEBI | CHEBI:40050 |
| ChEMBL | CHEMBL116 |
| NIAID ChemDB | 006080 |
| Chemical data | |
| Formula | C25H35N3O6S |
| Mol. mass | 505.628 g/mol |
Amprenavir (Agenerase, GlaxoSmithKline) is a protease inhibitor used to treat HIV infection. It was approved by the Food and Drug Administration on April 15, 1999, for twice-a-day dosing instead of needing to be taken every eight hours. The convenient dosing came at a price, as the dose required is 1,200 mg, delivered in eight very large gel capsules.
Production of amprenavir was discontinued by the manufacturer December 31, 2004; a prodrug version (fosamprenavir) is available
………………….
New approaches to the industrial synthesis of HIV protease inhibitors
http://pubs.rsc.org/en/content/articlelanding/2004/ob/b404071f/unauth#!divAbstract
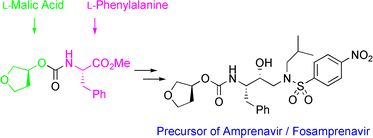
Efficient and industrially applicable synthetic processes for precursors of HIV protease inhibitors (Amprenavir, Fosamprenavir) are described. These involve a novel and economical method for the preparation of a key intermediate, (3S)-hydroxytetrahydrofuran, from L-malic acid. Three new approaches to the assembly of Amprenavir are also discussed. Of these, a synthetic route in which an (S)-tetrahydrofuranyloxy carbonyl is attached to L-phenylalanine appears to be the most promising manufacturing process, in that it offers satisfactory stereoselectivity in fewer steps.
AGENERASE (amprenavir) is an inhibitor of the human immunodeficiency virus (HIV) protease. The chemical name of amprenavir is (3S)-tetrahydro-3-furyl N-[(1S,2R)-3-(4-amino-N-isobutylbenzenesulfonamido)-1-benzyl-2-hydroxypropyl]carbamate. Amprenavir is a single stereoisomer with the (3S)(1S,2R) configuration. It has a molecular formula of C25H35N3O6S and a molecular weight of 505.64. It has the following structural formula:
 |
Amprenavir is a white to cream-colored solid with a solubility of approximately 0.04 mg/mL in water at 25°C.
AGENERASE Capsules (amprenavir capsules) are
available for oral administration. Each 50- mg capsule contains the inactive ingredients d-alpha tocopheryl polyethylene glycol 1000 succinate (TPGS), polyethylene glycol 400 (PEG 400) 246.7 mg, and propylene glycol 19 mg. The capsule shell contains the inactive ingredients d-sorbitol and sorbitans solution, gelatin, glycerin, and titanium dioxide. The soft gelatin capsules are printed with edible red ink. Each 50- mg AGENERASE Capsule contains 36.3 IU vitamin E in the form of TPGS. The total amount of vitamin E in the recommended daily adult dose of AGENERASE is 1,744 IU.
See also
- Fosamprenavir, a prodrug of amprenavir
External links
- Amprenavir bound to proteins in the PDB
Luseogliflozin, TS 071…………. strongly inhibited SGLT2 activity,

LUSEOGLIFLOZIN, CAS 898537-18-3
An antidiabetic agent that inhibits sodium-dependent glucose cotransporter 2 (SGLT2).
(1S)-1,5-Anhydro-1-[5-(4-ethoxybenzyl)-2-methoxy-4-methylphenyl]-1-thio-d-glucitol
(1S)-1,5-anhydro-1-[3-(4-ethoxybenzyl)-6-methoxy-4-methylphenyl]-1-thio-D-glucitol
Taisho Pharmaceutical Co., Ltd
Taisho (Originator), PHASE 3
Click to access 2013041801-e.pdf
TS-071
| Taisho Pharmaceutical Holdings Co. Ltd. | |
| Description | Oral sodium-glucose cotransporter 2 (SGLT2) inhibitor |

TS-071, an SGLT-2 inhibitor, is in phase III clinical development at Taisho for the oral treatment of type 1 and type 2 diabetes
In 2012, the product was licensed to Novartis and Taisho Toyama Pharmaceutical by Taisho in Japan for comarketing for the treatment of type 2 diabetes.
Diabetes is a metabolic disorder which is rapidly emerging as a global health care problem that threatens to reach pandemic levels. The number of people with diabetes worldwide is expected to rise from 285 million in 2010 to 438 million by 2030. Diabetes results from deficiency in insulin because of impaired pancreatic β-cell function or from resistance to insulin in body, thus leading to abnormally high levels of blood glucose.
Diabetes which results from complete deficiency in insulin secretion is Type 1 diabetes and the diabetes due to resistance to insulin activity together with an inadequate insulin secretion is Type 2 diabetes. Type 2 diabetes (Non insulin dependent diabetes) accounts for 90-95 % of all diabetes. An early defect in Type 2 diabetes mellitus is insulin resistance which is a state of reduced responsiveness to circulating concentrations of insulin and is often present years before clinical diagnosis of diabetes. A key component of the pathophysiology of Type 2 diabetes mellitus involves an impaired pancreatic β-cell function which eventually contributes to decreased insulin secretion in response to elevated plasma glucose. The β-cell compensates for insulin resistance by increasing the insulin secretion, eventually resulting in reduced β-cell mass. Consequently, blood glucose levels stay at abnormally high levels (hyperglycemia).
Hyperglycemia is central to both the vascular consequences of diabetes and the progressive nature of the disease itself. Chronic hyperglycemia leads to decrease in insulin secretion and further to decrease in insulin sensitivity. As a result, the blood glucose concentration is increased, leading to diabetes, which is self-exacerbated. Chronic hyperglycemia has been shown to result in higher protein glycation, cell apoptosis and increased oxidative stress; leading to complications such as cardiovascular disease, stroke, nephropathy, retinopathy (leading to visual impairment or blindness), neuropathy, hypertension, dyslipidemia, premature atherosclerosis, diabetic foot ulcer and obesity. So, when a person suffers from diabetes, it becomes important to control the blood glucose level. Normalization of plasma glucose in Type 2 diabetes patients improves insulin action and may offset the development of beta cell failure and diabetic complications in the advanced stages of the disease.
Diabetes is basically treated by diet and exercise therapies. However, when sufficient relief is not obtained by these therapies, medicament is prescribed alongwith. Various antidiabetic agents being currently used include biguanides (decrease glucose production in the liver and increase sensitivity to insulin), sulfonylureas and meglitinides (stimulate insulin production), a-glucosidase inhibitors (slow down starch absorption and glucose production) and thiazolidinediones (increase insulin sensitivity). These therapies have various side effects: biguanides cause lactic acidosis, sulfonylurea compounds cause significant hypoglycemia, a-glucosidase inhibitors cause abdominal bloating and diarrhea, and thiazolidinediones cause edema and weight gain. Recently introduced line of therapy includes inhibitors of dipeptidyl peptidase-IV (DPP-IV) enzyme, which may be useful in the treatment of diabetes, particularly in Type 2 diabetes. DPP-IV inhibitors lead to decrease in inactivation of incretins glucagon like peptide- 1 (GLP-1) and gastric inhibitory peptide (GIP), thus leading to increased production of insulin by the pancreas in a glucose dependent manner. All of these therapies discussed, have an insulin dependent mechanism.
Another mechanism which offers insulin independent means of reducing glycemic levels, is the inhibition of sodium glucose co-transporters (SGLTs). In healthy individuals, almost 99% of the plasma glucose filtered in the kidneys is reabsorbed, thus leading to only less than 1% of the total filtered glucose being excreted in urine. Two types of SGLTs, SGLT-1 and SGLT-2, enable the kidneys to recover filtered glucose. SGLT-1 is a low capacity, high-affinity transporter expressed in the gut (small intestine epithelium), heart, and kidney (S3 segment of the renal proximal tubule), whereas SGLT-2 (a 672 amino acid protein containing 14 membrane-spanning segments), is a low affinity, high capacity glucose ” transporter, located mainly in the S 1 segment of the proximal tubule of the kidney. SGLT-2 facilitates approximately 90% of glucose reabsorption and the rate of glucose filtration increases proportionally as the glycemic level increases. The inhibition of SGLT-2 should be highly selective, because non-selective inhibition leads to complications such as severe, sometimes fatal diarrhea, dehydration, peripheral insulin resistance, hypoglycemia in CNS and an impaired glucose uptake in the intestine.
Humans lacking a functional SGLT-2 gene appear to live normal lives, other than exhibiting copious glucose excretion with no adverse effects on carbohydrate metabolism. However, humans with SGLT-1 gene mutations are unable to transport glucose or galactose normally across the intestinal wall, resulting in condition known as glucose-galactose malabsorption syndrome.
Hence, competitive inhibition of SGLT-2, leading to renal excretion of glucose represents an attractive approach to normalize the high blood glucose associated with diabetes. Lower blood glucose levels would, in turn, lead to reduced rates of protein glycation, improved insulin sensitivity in liver and peripheral tissues, and improved cell function. As a consequence of progressive reduction in hepatic insulin resistance, the elevated hepatic glucose output which is characteristic of Type 2 diabetes would be expected to gradually diminish to normal values. In addition, excretion of glucose may reduce overall caloric load and lead to weight loss. Risk of hypoglycemia associated with SGLT-2 inhibition mechanism is low, because there is no interference with the normal counter regulatory mechanisms for glucose.
The first known non-selective SGLT-2 inhibitor was the natural product phlorizin
(glucose, 1 -[2-P-D-glucopyranosyloxy)-4,6-dihydroxyphenyl]-3-(4-hydroxyphenyl)- 1 – propanone). Subsequently, several other synthetic analogues were derived based on the structure of phlorizin. Optimisation of the scaffolds to achieve selective SGLT-2 inhibitors led to the discovery of several considerably different scaffolds.
C-glycoside derivatives have been disclosed, for example, in PCT publications
W.O20040131 18, WO2005085265, WO2006008038, WO2006034489, WO2006037537, WO2006010557, WO2006089872, WO2006002912, WO2006054629, WO2006064033, WO2007136116, WO2007000445, WO2007093610, WO2008069327, WO2008020011, WO2008013321, WO2008013277, WO2008042688, WO2008122014, WO2008116195, WO2008042688, WO2009026537, WO2010147430, WO2010095768, WO2010023594, WO2010022313, WO2011051864, WO201 1048148 and WO2012019496 US patents US65151 17B2, US6936590B2 and US7202350B2 and Japanese patent application JP2004359630. The compounds shown below are the SGLT-2 inhibitors which have reached advanced stages of human clinical trials: Bristol-Myers Squibb’s “Dapagliflozin” with Formula A, Mitsubishi Tanabe and Johnson & Johnson’s “Canagliflozin” with Formula B, Lexicon’s “Lx-421 1″ with Formula C, Boehringer Ingelheim and Eli Lilly’s “Empagliflozin” with Formula D, Roche and Chugai’s “Tofogliflozin” with Formula E, Taisho’s “Luseogliflozin” with Formula F, Pfizer’ s “Ertugliflozin” with Formula G and Astellas and Kotobuki’s “Ipragliflozin” with Formula H.
Formula G Formula H
In spite of all these molecules in advanced stages of human clinical trials, there is still no drug available in the market as SGLT-2 inhibitor. Out of the potential candidates entering the clinical stages, many have been discontinued, emphasizing the unmet need. Thus there is an ongoing requirement to screen more scaffolds useful as SGLT-2 inhibitors that can have advantageous potency, stability, selectivity, better half-life, and/ or better pharmacodynamic properties. In this regard, a novel class of SGLT-2 inhibitors is provided herein
………………………
SYNTHESIS

- Example 5
Synthesis of 2,3,4,6-tetra-O-benzyl-1-C-[2-methoxy-4-methyl-(4-ethoxybenzyl)phenyl]-5-thio-D-glucopyranose
-
Five drops of 1,2-dibromoethane were added to a mixture of magnesium (41 mg, 1.67 mmol), 1-bromo-3-(4-ethoxybenzyl)-6-methoxy-4-methylbenzene (0.51 g, 1.51 mmol) and tetrahydrofuran (2 mL). After heated to reflux for one hour, this mixture was allowed to stand still to room temperature to prepare a Grignard reagent. A tetrahydrofuran solution (1.40 mL) of 1.0 M i-propyl magnesium chloride and the prepared Grignard reagent were added dropwise sequentially to a tetrahydrofuran (5 mL) solution of 2,3,4,6-tetra-O-benzyl-5-thio-D-glucono-1,5-lactone (0.76 g, 1.38 mmol) while cooled on ice and the mixture was stirred for 30 minutes. After the reaction mixture was added with a saturated ammonium chloride aqueous solution and extracted with ethyl acetate, the organic phase was washed with brine and dried with anhydrous magnesium sulfate. After the desiccant was filtered off, the residue obtained by evaporating the solvent under reduced pressure was purified by silica gel column chromatography (hexane:ethyl acetate =4:1) to obtain (0.76 g, 68%) a yellow oily title compound.
1H NMR (300 MHz, CHLOROFORM-d) δ ppm 1.37 (t, J=6.92 Hz, 3 H) 2.21 (s, 3 H) 3.51 – 4.20 (m, 12 H) 3.85 – 3.89 (m, 3 H) 4.51 (s, 2 H) 4.65 (d, J=10.72 Hz, 1 H) 4.71 (d, J=5.75 Hz, 1 H) 4.78 – 4.99 (m, 3 H) 6.59 – 7.43 (m, 26 H)
Example 6
-
[0315]
Synthesis of (1S)-1,5-anhydro-2,3,4,6-tetra-O-benzyl-1-[2-methoxy-4-methyl-5-(4-ethoxybenzyl)phenyl]-1-thio-D-glucitol
-
An acetonitrile (18 mL) solution of 2,3,4,6-tetra-O-benzyl-1-C-[2-methoxy-4-methyl-5-(4-ethoxybenzyl)phenyl]-5-thio-D-glucopyranose (840 mg, 1.04 mmol) was added sequentially with Et3SiH (0.415 mL, 2.60 mmol) and BF3·Et2O (0.198 mL, 1.56 mmol) at -18°C and stirred for an hour. After the reaction mixture was added with a saturated sodium bicarbonate aqueous solution and extracted with ethyl acetate, the organic phase was washed with brine and then dried with anhydrous magnesium sulfate. After the desiccant was filtered off, the residue obtained by evaporating the solvent under reduced pressure was purified by silica gel column chromatography (hexane:ethyl acetate=4:1) to obtain the title compound (640 mg, 77%).
1H NMR (600 MHz, CHLOROFORM-d) δ ppm 1.35 (t, J=6.88 Hz, 3 H) 2.21 (s, 3 H) 3.02 – 3.21 (m, 1 H) 3.55 (t,J=9.40 Hz, 1 H) 3.71 (s, 1 H) 3.74 – 3.97 (m, 10 H) 4.01 (s, 1 H) 4.45 – 4.56 (m, 3 H) 4.60 (d, J=10.55 Hz, 2 H) 4.86 (s, 2 H) 4.90 (d, J=10.55 Hz, 1H) 6.58 – 6.76 (m, 5 H) 6.90 (d, J=7.34 Hz, 1 H) 7.09 – 7.19 (m, 5 H) 7.23 – 7.35 (m, 15 H).
ESI m/z = 812 (M+NH4).
Example 7
Synthesis of (1S)-1,5-anhydro-1-[3-(4-ethoxybenzyl)-6-methoxy-4-methylphenyl]-1-thio-D-glucitol
-
A mixture of (1S)-1,5-anhydro-2,3,4,6-tetra-O-benzyl-1-[2-methoxy-4-methyl-5-(4-ethoxybenzyl)phenyl]-1-thio-D-glucitol (630 mg, 0.792 mmol), 20% palladium hydroxide on activated carbon (650 mg) and ethyl acetate (10 mL) – ethanol (10 mL) was stirred under hydrogen atmosphere at room temperature for 66 hours. The insolubles in the reaction mixture were filtered off with celite and the filtrate was concentrated. The obtained residue was purified by silica gel column chromatography (chloroform:methanol =10:1) to obtain a colorless powdery title compound (280 mg, 81%) as 0.5 hydrate. 1H NMR (600 MHz, METHANOL- d4) δ ppm 1.35 (t, J=6.9 Hz, 3 H) 2.17 (s, 3 H) 2.92 – 3.01 (m, 1 H) 3.24 (t, J=8.71 Hz, 1 H) 3.54 – 3.60 (m, 1 H) 3.72 (dd, J=11.5, 6.4 Hz, 1 H) 3.81 (s, 3 H) 3.83 (s, 2 H) 3.94 (dd, J=11.5, 3.7 Hz, 1 H) 3.97 (q, J=6.9 Hz, 2 H) 4.33 (s, 1 H) 6.77 (d, J=8.3 Hz, 2 H) 6.76 (s, 1 H) 6.99 (d, J=8.3 Hz, 2 H) 7.10 (s, 1 H). ESI m/z = 452 (M+NH4+), 493 (M+CH3CO2-). mp 155.0-157.0°C. Anal. Calcd for C23H30O6S·0.5H2O: C, 62.28; H, 7.06. Found: C, 62.39; H, 7.10.
………………………………..
PAPER

(1S)-1,5-Anhydro-1-[5-(4-ethoxybenzyl)-2-methoxy-4-methylphenyl]-1-thio-d-glucitol (TS-071) is a Potent, Selective Sodium-Dependent Glucose Cotransporter 2 (SGLT2) Inhibitor for Type 2 Diabetes Treatment 
(Journal of Medicinal Chemistry) Saturday March 20th 2010
Author(s): ,
DOI:10.1021/jm901893x
GO TO: [Article]
http://pubs.acs.org/doi/abs/10.1021/jm901893x

(1S)-1,5-Anhydro-1-[5-(4-ethoxybenzyl)-2-methoxy-4-methylphenyl]-1-thio-d-glucitol (3p)
 3p is compd
3p is compd
| cmpds | R1 | R2 | R3 | SGLT2 (nM) mean (95% CI) | SGLT1 (nM) mean (95% CI) | T1/T2 selectivity |
|---|---|---|---|---|---|---|
| 1 | 27.8 (21.8−35.3) | 246 (162−374) | 8.8 | |||
| 3a | H | H | OEt | 73.6 (51.4−105) | 26100 (20300−33700) | 355 |
| 3b | H | OH | OEt | 283 (268−298) | 14600 (11500−18500) | 51.6 |
| 3c | H | OMe | OEt | 13.4 (11.3−15.8) | 565 (510−627) | 42.2 |
| 3d | H | F | OEt | 9.40 (5.87−15.0) | 7960 (7180−8820) | 847 |
| 3e | H | Me | OEt | 2.29 (1.76−2.99) | 671 (230−1960) | 293 |
| 3f | H | Cl | OEt | 1.77 (0.95−3.30) | 1210 (798−1840) | 684 |
| 3g | OH | H | OEt | 17.4 (15.9−19.0) | 4040 (1200−13600) | 232 |
| 3h | OMe | H | OEt | 37.9 (26.4−54.4) | 100000 (66500−151000) | 2640 |
| 3i | OMe | OMe | OEt | 10.8 (6.84−17.1) | 4270 (1560−11600) | 395 |
| 3j | H | Cl | OMe | 1.68 (1.08−2.60) | 260 (72.5−931) | 155 |
| 3k | H | Cl | Me | 1.37 (0.97−1.95) | 209 (80.2−545) | 153 |
| 3l | H | Cl | Et | 1.78 (0.88−3.63) | 602 (473−767) | 338 |
| 3m | H | Cl | iPr | 4.01 (1.75−9.17) | 8160 (4860−13700) | 2040 |
| 3n | H | Cl | tBu | 18.8 (11.0−32.1) | 35600 (31900−39800) | 1890 |
| 3o | H | Cl | SMe | 1.16 (0.73−1.85) | 391 (239−641) | 337 |
| 3p | OMe | Me | OEt | 2.26 (1.48−3.43) | 3990 (2690−5920) | 1770 |
| 3q | OMe | Me | Et | 1.71 (1.19−2.46) | 2830 (1540−5200) | 1650 |
| 3r | OMe | Me | iPr | 2.68 (2.15−3.34) | 17300 (14100−21100) | 6400 |
| 3s | OMe | Cl | Et | 1.51 (0.75−3.04) | 3340 (2710−4110) | 2210 |

| Patent | Filing date | Publication date | Applicant | Title |
|---|---|---|---|---|
| WO2004014930A1 * | Aug 8, 2003 | Feb 19, 2004 | Asanuma Hajime | PROCESS FOR SELECTIVE PRODUCTION OF ARYL 5-THIO-β-D- ALDOHEXOPYRANOSIDES |
| Reference | ||
|---|---|---|
| 1 | * | AL-MASOUDI, NAJIM A. ET AL: “Synthesis of some novel 1-(5-thio-.beta.-D-glucopyranosyl)-6-azaur acil derivatives. Thio sugar nucleosides” NUCLEOSIDES & NUCLEOTIDES , 12(7), 687-99 CODEN: NUNUD5; ISSN: 0732-8311, 1993, XP008091463 |
| 2 | * | See also references of WO2006073197A1 |
| EP2419097A1 * | Apr 16, 2010 | Feb 22, 2012 | Taisho Pharmaceutical Co., Ltd. | Pharmaceutical compositions |
| EP2455374A1 * | Oct 15, 2009 | May 23, 2012 | Janssen Pharmaceutica N.V. | Process for the Preparation of Compounds useful as inhibitors of SGLT |
| EP2601949A2 * | Apr 16, 2010 | Jun 12, 2013 | Taisho Pharmaceutical Co., Ltd. | Pharmaceutical compositions |
| EP2668953A1 * | May 15, 2009 | Dec 4, 2013 | Bristol-Myers Squibb Company | Pharmaceutical compositions comprising an SGLT2 inhibitor with a supply of carbohydrate and/or an inhibitor of uric acid synthesis |
| WO2009143020A1 | May 15, 2009 | Nov 26, 2009 | Bristol-Myers Squibb Company | Method for treating hyperuricemia employing an sglt2 inhibitor and composition containing same |
| WO2010043682A2 * | Oct 15, 2009 | Apr 22, 2010 | Janssen Pharmaceutica Nv | Process for the preparation of compounds useful as inhibitors of sglt |
| WO2010119990A1 | Apr 16, 2010 | Oct 21, 2010 | Taisho Pharmaceutical Co., Ltd. | Pharmaceutical compositions |
| WO2013152654A1 * | Mar 14, 2013 | Oct 17, 2013 | Theracos, Inc. | Process for preparation of benzylbenzene sodium-dependent glucose cotransporter 2 (sglt2) inhibitors |

-
Luseogliflozin: Phase III data 10/21/2013
Week in Review, Clinical ResultsTaisho Pharmaceutical Holdings Co. Ltd. (Tokyo:4581), Tokyo, Japan Product: Luseogliflozin (TS-071) Business: Endocrine/Metabolic Molecular target: Sodium-glucose cotransporter 2 (SGLT2) Description: Oral sodium-glucose… -
Luseogliflozin: Phase III data 10/21/2013
Week in Review, Clinical ResultsTaisho Pharmaceutical Holdings Co. Ltd. (Tokyo:4581), Tokyo, Japan Product: Luseogliflozin (TS-071) Business: Endocrine/Metabolic Molecular target: Sodium-glucose cotransporter 2 (SGLT2) Description: Oral sodium-glucose… -
Luseogliflozin regulatory update 05/13/2013
Week in Review, RegulatoryTaisho Pharmaceutical Holdings Co. Ltd. (Tokyo:4581), Tokyo, Japan Product: Luseogliflozin (TS-071) Business: Endocrine/Metabolic Last month, Taisho’s Taisho Pharmaceutical Co. Ltd. subsidiary submitted a regulatory … -
Strategy: Doubling down in diabetes 05/06/2013
Merck shoring up slowing diabetes franchise with Pfizer, Abide dealsBioCentury on BioBusiness, StrategyAs sales flatten for Merck’s sitagliptin franchise and a new class of oral diabetes drugs comes to market, the pharma has tapped Pfizer and Abide to shore up its position.
see
SEE
http://www.clinicaltrials.jp/user/showCteDetailE.jsp?japicId=JapicCTI-132352


RedHill Biopharma Ltd. Acquires Phase 2 Oncology Drug Upamostat MESUPRON From Wilex AG

Upamostat
CAS: 590368-25-5
Chemical Formula: C32H47N5O6S
Exact Mass: 629.32470
Synonym: WX 671; WX-671; WX671. Upamostat; Brand name: Mesupron.
IUPAC/Chemical name:
(S)-ethyl 4-(3-(3-(N-hydroxycarbamimidoyl)phenyl)-2-(2,4,6-triisopropylphenylsulfonamido)propanoyl)piperazine-1-carboxylate

RedHill Biopharma Ltd. , an Israeli biopharmaceutical company focused on late clinical-stage drugs for inflammatory and gastrointestinal diseases, including cancer, and WILEX AG , a biopharmaceutical company focused on oncology, based in Munich, Germany, today announced that they have signed an exclusive license agreement for the oncology drug … (more)
Upamostat, also known as Mesupron, WX-671, is an orally bioavailable, 3-amidinophenylalanine-derived, second generation serine protease inhibitor prodrug targeting the human urokinase plasminogen activator (uPA) system with potential antineoplastic and antimetastatic activities. After oral administration, serine protease inhibitor WX-671 is converted to the active Nα-(2,4,6-triisopropylphenylsulfonyl)-3-amidino-(L)-phenyla lanine-4-ethoxycarbonylpiperazide (WX-UK1), which inhibits several serine proteases, particularly uPA; inhibition of uPA may result in the inhibition of tumor growth and metastasis. uPA is a serine protease involved in degradation of the extracellular matrix and tumor cell migration and proliferation.

|
Information about this agent |
WX-671 (Mesupron) is an orally available prodrug of WX-UK1, a serine protease inhibitor that inhibits uPA as well as other serine proteases. WX-UK1 (Setyono-Han et al., Thromb Haemost 2005) and WX-671 have shown to efficiently reduce primary tumor growth and metastasis formation in a variety of animal models. The proteolytic factor uPA and its inhibitor PAI-1 belong to those biological factors which have provided the highest level of evidence (LOE1) in terms of their prognostic and predictive significance. WX-671 is currently the only drug in Phase II aiming at this target.Results: All 95 patients were accrued between Jun 2007 and Aug 2008. Efficacy is assessed by a central reader at regular intervals based on digital CT images. By end of 2009, 2 patients were still on treatment without signs of progression, 64 patients had died. Preliminary analysis of overall survival showed an increase in overall survival from 10.2 mo (gemcitabine alone) to 13.5 mo for the combination of gemcitabine and WX-671. 1-year survival increased from 37% with gemcitabine to 53% when combined with 400 mg WX- 671. Conclusions: The combination of daily oral WX-671 in combination with weekly i.v. gemcitabine was well tolerated. see asco.com’s website.

|
References |
1. Analysis of highly potent amidine containing inhibitors of serine proteases and their N-hydroxylated prodrugs (amidoximes) By Kotthaus, Joscha; Steinmetzer, Torsten; van de Locht, Andreas; Clement, Bernd From Journal of Enzyme Inhibition and Medicinal Chemistry (2011), 26(1), 115-122.
2. Combined treatment of cancer by urokinase inhibition and a cytostatic anti-cancer agent for enhancing the anti-metastatic effect By Schmalix, Wolfgang; Schneider, Anneliese; Setyono-Han, Buddy; Foekens, Johannes From U.S. Pat. Appl. Publ. (2008), US 20080226624 A1 20080918.
3. Peptides and small molecules targeting the plasminogen activation system: towards prophylactic anti-metastasis drugs for breast cancer By Tyndall, Joel D. A.; Kelso, Michael J.; Clingan, Phillip; Ranson, Marie From Recent Patents on Anti-Cancer Drug Discovery (2008), 3(1), 1-13.
4. Synthesis of hydroxyamidine and hydroxyguanidine amino acid or oligopeptide derivatives for use as urokinase plasminogen activator inhibitors for the treatment of cancer and its metastasis By Sperl, Stefan; Buergle, Markus; Schmalix, Wolfgang; Wosikowski, Katja; Clement, Bernd From U.S. Pat. Appl. Publ. (2006), US 20060142305 A1 20060629.
5. Crystalline modifications of N-α-(2,4,6-triisopropylphenylsulfonyl)-3-hydroxyamidino-(l)-phenylalanine-4-ethoxycarbonylpiperazide and/or its salts By Grunenberg, Alfons; Lenz, Jana From PCT Int. Appl. (2006), WO 2006056448 A1 20060601.
6. Synthesis of hydroxyamidine and hydroxyguanidine amino acid or oligopeptide derivatives for use as urokinase plasminogen activator inhibitors for the treatment of cancer and its metastasis By Sperl, Stefan; Burgle, Markus; Schmalix, Wolfgang; Wosikowski, Katja; Clement, Bernd From PCT Int. Appl. (2004), WO 2004103984 A1 20041202.
7. Preparation of 3-amidinophenylalanine derivatives from 3-cyanophenylalanines via reduction and hydrogenation under mild conditions By Ziegler, Hugo; Wikstroem, Peter From PCT Int. Appl. (2003), WO 2003072559 A1 20030904.
1. Buddy et al, Suppression of Rat Brest Cancer Metastasis and Reduction of Primary Tumor Growth by the Small Synthetic Urokinase Inhibitor WX-UK1. Thromb Haemost. 2005, 93:779-786.
2. Ertongur S, Lang S, Mack B, Wosikowski K, Muehlenweg B, Gires O. Inhibition of the invasion capacity of carcinoma cells by WX-UK1, a novel synthetic inhibitor of the urokinase-type plasminogen activator system. Int J Cancer. 2004, 110(6):815-24.
3. Setyono-Han B, Stürzebecher J, Schmalix WA, Muehlenweg B, Sieuwerts AM, Timmermans M, Magdolen V, Schmitt M, Klijn JG, Foekens JA. Suppression of rat breast cancer metastasis and reduction of primary tumour growth by the small synthetic urokinase inhibitor WX-UK1. Thromb Haemost. 2005, 93(4):779-86.
FDA grants orphan drug designation to Insys Therapeutics’ pharmaceutical cannabidiol


| Systematic (IUPAC) name | |
|---|---|
| 2-[(1R,6R)-6-isopropenyl-3-methylcyclohex-2-en-1-yl]-5-pentylbenzene-1,3-diol | |
| Clinical data | |
| Trade names | Epidiolex |
| AHFS/Drugs.com | International Drug Names |
| Legal status | Schedule I (US)Schedule II (Can)(THC – Schedule/Level I; THC and CBD two main chemicals in cannabis) |
| Pharmacokinetic data | |
| Bioavailability | 13-19% (oral),[1] 11-45% (mean 31%; inhaled)[2] |
| Half-life | 9 h[1] |
| Identifiers | |
| CAS number | 13956-29-1 |
| ATC code | None |
| PubChem | CID 644019 |
| ChemSpider | 24593618 |
| UNII | 19GBJ60SN5 |
| Chemical data | |
| Formula | C21H30O2 |
| Mol. mass | 314.4636 |
| Physical data | |
| Melt. point | 66 °C (151 °F) |
| Boiling point | 180 °C (356 °F) (range: 160–180 °C)[3] |
FDA grants orphan drug designation to Insys Therapeutics’ pharmaceutical cannabidiol – Pharmaceutical Technology
US-based specialty pharmaceutical company Insys Therapeutics has obtained orphan drug designation from the US Food and Drug Administration (FDA) for its pharmaceutical cannabidiol for treatment of Lennox-Gastaut Syndrome.
Insys Therapeutics president and CEO Michael Babich said: “With no cure and persistence of seizures with current antiepileptic medications, the orphan drug designation recognises the significant, unmet need that exists among children with this severe form of epilepsy and the teams who provide their care.
“We have the unique opportunity to test a controlled pharmaceutical CBD product for Lennox-Gastaut Syndrome, and our company is committed to advancing cannabinoid therapies that have the potential to provide significant medical benefits to patients across multiple indications.
“We expect to file an investigational new drug application (IND) for CBD in the second half of 2014.”
Cannabidiol (CBD) is one of at least 60 active cannabinoids identified in cannabis.[4] It is a major phytocannabinoid, accounting for up to 40% of the plant’s extract.[5] CBD is considered to have a wider scope of medical applications than tetrahydrocannabinol(THC).[5] An orally-administered liquid containing CBD has received orphan drug status in the US, for use as a treatment for dravet syndrome under the brand name, Epidiolex.[6]
Clinical applications

The bud of a Cannabis sativa flower coated with trichomes
Antimicrobial actions
CBD absorbed transcutaneously may attenuate the increased sebum production at the root of acne, according to an untested hypothesis.[7]
Neurological effects
A 2010 study found that strains of cannabis containing higher concentrations of cannabidiol did not produce short-term memory impairment vs. strains with similar concentrations of THC, but lower concentrations of CBD. The researchers attributed this attenuation of memory effects to CBD’s role as a CB1 antagonist. Transdermal CBD is neuroprotective in animals.[8]
Cannabidiol’s strong antioxidant properties have been shown to play a role in the compound’s neuroprotective and anti-ischemiceffects.[9]
- Parkinson’s disease
It has been proposed that CBD may help people with Parkinson’s disease, but promising results in animal experiments were not confirmed when CBD was trialled in humans.[10]
Psychotropic effect
CBD has anti-psychotic effects and may counteract the potential psychotomimetic effects of THC on individuals with latentschizophrenia;[5] some reports show it to be an alternative treatment for schizophrenia that is safe and well-tolerated.[11] Studies have shown CBD may reduce schizophrenic symptoms due to its apparent ability to stabilize disrupted or disabled NMDA receptor pathways in the brain, which are shared and sometimes contested by norepinephrine and GABA.[11][12] Leweke et al. performed a double blind, 4 week, explorative controlled clinical trial to compare the effects of purified cannabidiol and the atypical antipsychoticamisulpride on improving the symptoms of schizophrenia in 42 patients with acute paranoid schizophrenia. Both treatments were associated with a significant decrease of psychotic symptoms after 2 and 4 weeks as assessed by Brief Psychiatric Rating Scale andPositive and Negative Syndrome Scale. While there was no statistical difference between the two treatment groups, cannabidiol induced significantly fewer side effects (extrapyramidal symptoms, increase in prolactin, weight gain) when compared to amisulpride.[13]
Studies have shown cannabidiol decreases activity of the limbic system[14] and decreases social isolation induced by THC.[15] Cannabidiol has also been shown to reduce anxiety in social anxiety disorder.[16][17] However, chronic cannabidiol administration in rats was recently found to produce anxiogenic-like effects, indicating that prolonged treatment with cannabidiol might incite anxiogenic effects.[18]
Cannabidiol has demonstrated antidepressant-like effects in animal models of depression.[19][20][21]
Cancer
The American Cancer Society says: “There is no available scientific evidence from controlled studies in humans that cannabinoids can cure or treat cancer.”[22] Laboratory experiments have been performed on the potential use of cannabinoids for cancer therapy but as of 2013 results have been contradictory and knowledge remains poor.[23] Cannabinoids have been recommended for cancer pain but the adverse effects may make them a less than ideal treatment; two cannabinoid-based medicines have been approved as a backup remedy for nausea associated withchemotherapy.[4]
Dravet syndrome
Dravet syndrome is a rare form of epilepsy that is difficult to treat. Dravet syndrome, also known as Severe Myoclonic Epilepsy of Infancy (SMEI), is a rare and catastrophic form of intractable epilepsy that begins in infancy. Initial seizures are most often prolonged events and in the second year of life other seizure types begin to emerge.[24] While high profile and anecdotal reports have sparked interest in treatment with cannabinoids,[25] there is insufficient medical evidence to draw conclusions about their safety or efficacy.[25][26]
CBD-enhanced cannabis
Decades ago, selective breeding by growers in US dramatically lowered the CBD content of cannabis; their customers preferred varietals that were more mind-altering due to a higher THC, lower CBD content.[27] To meet the demands of medical cannabis patients, growers are currently developing more CBD-rich strains.[28]
In November 2012, Tikun Olam, an Israeli medical cannabis facility announced a new strain of the plant which has only cannabidiol as an active ingredient, and virtually no THC, providing some of the medicinal benefits of cannabis without the euphoria.[29][30] The researchers said the cannabis plant, enriched with CBD, “can be used for treating diseases like rheumatoid arthritis, colitis, liver inflammation, heart disease and diabetes”. Research on CBD enhanced cannabis began in 2009, resulting in Avidekel, a cannabis strain that contains 15.8% CBD and less than 1% THC. Raphael Mechoulam, a cannabinoid researcher, said “…Avidekel is thought to be the first CBD-enriched cannabis plant with no THC to have been developed in Israel”.[31]
Pharmacology
Pharmacodynamics
Cannabidiol has a very low affinity for CB1 and CB2 receptors but acts as an indirect antagonist of their agonists.[9] While one would assume that this would cause cannabidiol to reduce the effects of THC, it may potentiate THC’s effects by increasing CB1 receptor density or through another CB1-related mechanism.[32] It is also an inverse agonist of CB2receptors.[9][33] Recently, it was found to be an antagonist at the putative new cannabinoid receptor, GPR55, a GPCR expressed in the caudate nucleus and putamen.[34]Cannabidiol has also been shown to act as a 5-HT1A receptor agonist,[35] an action which is involved in its antidepressant,[19][36] anxiolytic,[36][37] and neuroprotective[38][39]effects. Cannabidiol is an allosteric modulator of μ and δ-opioid receptors.[40] Cannabidiol’s pharmacologial effects have also been attributed to PPAR-γ receptor agonism andintracellular calcium release.[5]
Pharmacokinetic interactions
There is some preclinical evidence to suggest that cannabidiol may reduce THC clearance, modestly increasing THC’s plasma concentrations resulting in a greater amount of THC available to receptors, increasing the effect of THC in a dose-dependent manner.[41][42] Despite this the available evidence in humans suggests no significant effect of CBD on THC plasma levels.[43]
Pharmaceutical preparations
Nabiximols (USAN, trade name Sativex) is an aerosolized mist for oral administration containing a near 1:1 ratio of CBD and THC. The drug was approved by Canadian authorities in 2005 to alleviate pain associated with multiple sclerosis.[44][45][46]
Isomerism
| 7 double bond isomers and their 30 stereoisomers | ||||||||
|---|---|---|---|---|---|---|---|---|
| Formal numbering | Terpenoid numbering | Number of stereoisomers | Natural occurrence | Convention on Psychotropic SubstancesSchedule | Structure | |||
| Short name | Chiral centers | Full name | Short name | Chiral centers | ||||
| Δ5-cannabidiol | 1 and 3 | 2-(6-isopropenyl-3-methyl-5-cyclohexen-1-yl)-5-pentyl-1,3-benzenediol | Δ4-cannabidiol | 1 and 3 | 4 | No | unscheduled |  |
| Δ4-cannabidiol | 1, 3 and 6 | 2-(6-isopropenyl-3-methyl-4-cyclohexen-1-yl)-5-pentyl-1,3-benzenediol | Δ5-cannabidiol | 1, 3 and 4 | 8 | No | unscheduled |  |
| Δ3-cannabidiol | 1 and 6 | 2-(6-isopropenyl-3-methyl-3-cyclohexen-1-yl)-5-pentyl-1,3-benzenediol | Δ6-cannabidiol | 3 and 4 | 4 | ? | unscheduled |  |
| Δ3,7-cannabidiol | 1 and 6 | 2-(6-isopropenyl-3-methylenecyclohex-1-yl)-5-pentyl-1,3-benzenediol | Δ1,7-cannabidiol | 3 and 4 | 4 | No | unscheduled |  |
| Δ2-cannabidiol | 1 and 6 | 2-(6-isopropenyl-3-methyl-2-cyclohexen-1-yl)-5-pentyl-1,3-benzenediol | Δ1-cannabidiol | 3 and 4 | 4 | Yes | unscheduled |  |
| Δ1-cannabidiol | 3 and 6 | 2-(6-isopropenyl-3-methyl-1-cyclohexen-1-yl)-5-pentyl-1,3-benzenediol | Δ2-cannabidiol | 1 and 4 | 4 | No | unscheduled |  |
| Δ6-cannabidiol | 3 | 2-(6-isopropenyl-3-methyl-6-cyclohexen-1-yl)-5-pentyl-1,3-benzenediol | Δ3-cannabidiol | 1 | 2 | No | unscheduled |  |
See also: Tetrahydrocannabinol#Isomerism, Abnormal cannabidiol.
Chemistry
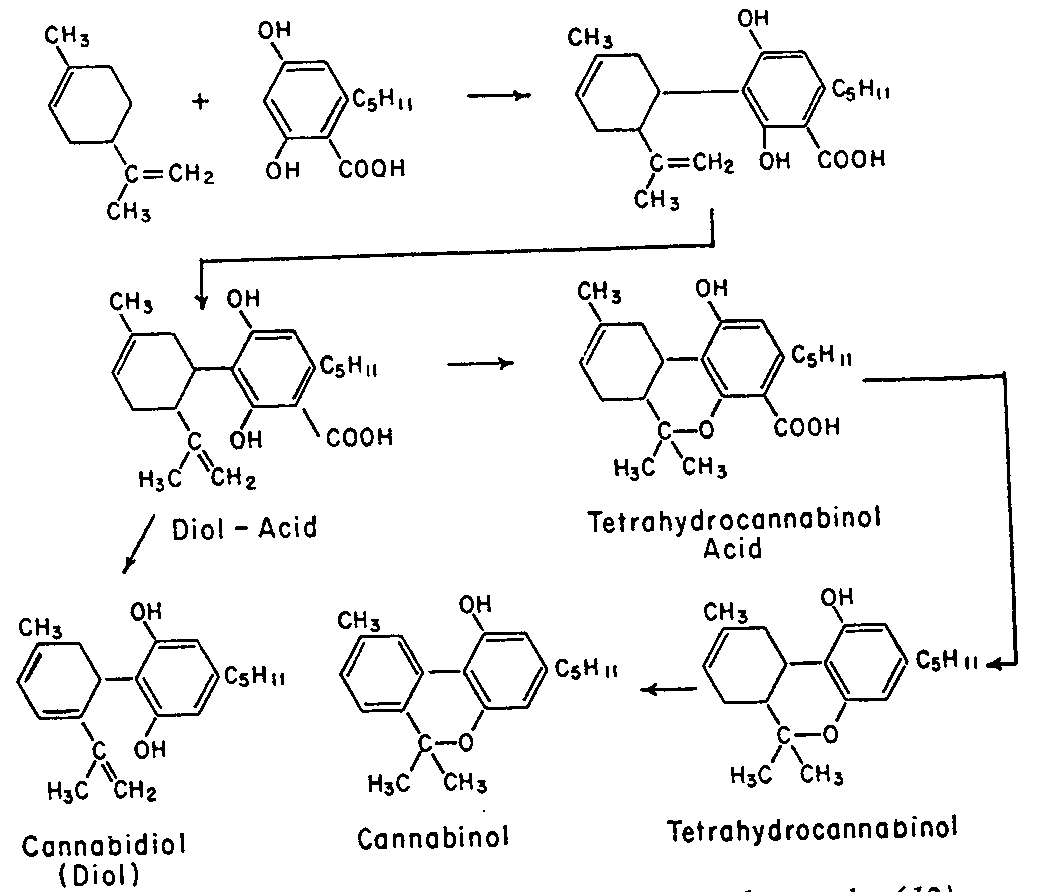
Cannabidiol is insoluble in water but soluble in organic solvents, such as pentane. At room temperature it is a colorless crystalline solid.[47] In strongly basic medium and the presence of air it is oxidized to a quinone.[48] Under acidic conditions it cyclizes to THC.[49] The synthesis of cannabidiol has been accomplished by several research groups.[50][51][52]

http://pubs.rsc.org/en/content/articlelanding/2005/ob/b416943c#!divAbstract
https://www.unodc.org/unodc/en/data-and-analysis/bulletin/bulletin_1964-01-01_4_page005.html

http://pubs.rsc.org/en/content/articlelanding/2005/ob/b416943c#!divAbstract
Biosynthesis
Cannabis produces CBD-carboxylic acid through the same metabolic pathway as THC, until the last step, where CBDA synthase performs catalysis instead of THCA synthase.[53]
Legal status
Cannabidiol is not scheduled by the Convention on Psychotropic Substances.
Cannabidiol is a Schedule II drug in Canada.[54]
Cannabidiol’s legal status in the United States:
The DEA Drug Schedule classifies synthetic THC (Tetrahydrocannabinol) as a schedule III substance (eg Marinol); while the natural marijuana plant is listed as Schedule I. Cannabidiol is not named specifically on the list.[55] However the CSA does mention all natural Phytocannabinoids in Schedule 1 Code 7372, which would include CBD.[55]
Marijuana (along with all of its cannabinoids) is defined by 21 U.S.C. §802(16), which is part of the Controlled Substances Act.[56][57][58] There is an exemption for certain Hemp products produced abroad. Under this exception, what are known as industrial hemp-finished products are legally imported into the United States each year. Hemp finished products which meet the specific definitions including hemp oil which may contain cannabidiol are legal in the United States but aren’t used for getting high.[59]
Some cannabidiol oil is derived from marijuana and therefore contains higher levels of THC.[60] This type of cannabidiol oil would be considered a Schedule I as a result of the THC present.[60]
US patent
In October 2003, U.S. patent #6630507 entitled “Cannabinoids as antioxidants and neuroprotectants” was assigned to “The United States Of America As Represented By The Department Of Health And Human Services.” The patent was filed in April 1999 and listed as the inventors: Aidan J. Hampson, Julius Axelrod, and Maurizio Grimaldi, who all held positions at the National Institute of Mental Health (NIMH) in Bethesda, MD, which is part of the National Institutes of Health (NIH), an agency of the United States Department of Health and Human Services (HHS). The patent mentions cannabidiol’s ability as an antiepileptic, to lower intraocular pressure in the treatment of glaucoma, lack of toxicity or serious side effects in large acute doses, its neuroprotectant properties, its ability to prevent neurotoxicity mediated by NMDA, AMPA, or kainate receptors; its ability to attenuate glutamate toxicity, its ability to protect against cellular damage, its ability to protect brains from ischemic damage, its anxiolytic effect, and its superior antioxidant activity which can be used in the prophylaxis and treatment of oxidation associated diseases.[61]
| “ | “Oxidative associated diseases include, without limitation, free radical associated diseases, such as ischemia, ischemic reperfusion injury, inflammatory diseases, systemic lupus erythematosus, myocardial ischemia or infarction, cerebrovascular accidents (such as a thromboembolic or hemorrhagic stroke) that can lead to ischemia or an infarct in the brain, operative ischemia, traumatic hemorrhage (for example a hypovolemic stroke that can lead to CNS hypoxia or anoxia), spinal cord trauma, Down’s syndrome, Crohn’s disease, autoimmune diseases (e.g. rheumatoid arthritis or diabetes), cataract formation, uveitis, emphysema, gastric ulcers, oxygen toxicity, neoplasia, undesired cellular apoptosis, radiation sickness, and others. The present invention is believed to be particularly beneficial in the treatment of oxidative associated diseases of the CNS, because of the ability of the cannabinoids to cross the blood brain barrier and exert their antioxidant effects in the brain. In particular embodiments, the pharmaceutical composition of the present invention is used for preventing, arresting, or treating neurological damage in Parkinson’s disease, Alzheimer’s disease and HIV dementia; autoimmune neurodegeneration of the type that can occur in encephalitis, and hypoxic or anoxic neuronal damage that can result from apnea, respiratory arrest or cardiac arrest, and anoxia caused by drowning, brain surgery or trauma (such as concussion or spinal cord shock).”[61] | ” |
On November 17, 2011, the Federal Register published that the National Institutes of Health of the United States Department of Health and Human Services was “contemplating the grant of an exclusive patent license to practice the invention embodied in U.S. Patent 6,630,507” to the company KannaLife based in New York, for the development and sale of cannabinoid and cannabidiol based therapeutics for the treatment of hepatic encephalopathy in humans.[62][63][64]
References
- Mechoulam R, Parker LA, Gallily R (November 2002). “Cannabidiol: an overview of some pharmacological aspects”. J Clin Pharmacol (Review) 42 (11 Suppl): 11S–19S.doi:10.1177/0091270002238789. PMID 12412831.
- Scuderi C, Filippis DD, Iuvone T, Blasio A, Steardo A, Esposito G (May 2009). “Cannabidiol in medicine: a review of its therapeutic potential in CNS disorders”.Phytother Res (Review) 23 (5): 597–602. doi:10.1002/ptr.2625. PMID 18844286.
- McPartland JM, Russo EB (2001). “Cannabis and cannabis extracts: greater than the sum of their parts?”. Journal of Cannabis Therapeutics 1(3/4): 103–132. doi:10.1300/J175v01n03_08.
- Borgelt LM, Franson KL, Nussbaum AM, Wang GS (February 2013). “The pharmacologic and clinical effects of medical cannabis”. Pharmacotherapy (Review) 33(2): 195–209. doi:10.1002/phar.1187. PMID 23386598.
- Campos AC, Moreira FA, Gomes FV, Del Bel EA, Guimarães FS (December 2012). “Multiple mechanisms involved in the large-spectrum therapeutic potential of cannabidiol in psychiatric disorders”. Philos. Trans. R. Soc. Lond., B, Biol. Sci.(Review) 367 (1607): 3364–78. doi:10.1098/rstb.2011.0389. PMC 3481531.PMID 23108553.
- Wilner, AN (25 March 2014). “Marijuana for Epilepsy: Weighing the Evidence”.Medscape Neurology. WebMD. Retrieved 2 April 2014.
- Russo EB (August 2011). “Taming THC: potential cannabis synergy and phytocannabinoid-terpenoid entourage effects”. Br. J. Pharmacol. (Review) 163 (7): 1344–64. doi:10.1111/j.1476-5381.2011.01238.x. PMC 3165946. PMID 21749363.
- Liput, D. J.; Hammell, D. C.; Stinchcomb, A. L.; Nixon, K (2013). “Transdermal delivery of cannabidiol attenuates binge alcohol-induced neurodegeneration in a rodent model of an alcohol use disorder”. Pharmacology Biochemistry and Behavior 111: 120–7.doi:10.1016/j.pbb.2013.08.013. PMID 24012796.
- Mechoulam R, Peters M, Murillo-Rodriguez E, Hanus LO (August 2007). “Cannabidiol–recent advances”. Chem. Biodivers. (Review) 4 (8): 1678–92.doi:10.1002/cbdv.200790147. PMID 17712814.
- Iuvone T, Esposito G, De Filippis D, Scuderi C, Steardo L (2009). “Cannabidiol: a promising drug for neurodegenerative disorders?”. CNS Neurosci Ther 15 (1): 65–75.doi:10.1111/j.1755-5949.2008.00065.x. PMID 19228180.
- Zuardi AW, Crippa JA, Hallak JE, Moreira FA, Guimarães FS (April 2006).“Cannabidiol, a Cannabis sativa constituent, as an antipsychotic drug”. Braz. J. Med. Biol. Res. (Review) 39 (4): 421–9. doi:10.1590/S0100-879X2006000400001.PMID 16612464.
- Long, L. E.; Malone, D. T.; Taylor, D. A. (2005). “Cannabidiol Reverses MK-801-Induced Disruption of Prepulse Inhibition in Mice”. Neuropsychopharmacology 31 (4): 795–803. doi:10.1038/sj.npp.1300838. PMID 16052245.
- Leweke, FM; Piomelli D, Pahlisch F, Muhl D, Gerth CW, Hoyer C, Klosterkötter J, Hellmich M and Koethe D. (2012). “Cannabidiol enhances anandamide signaling and alleviates psychotic symptoms of schizophrenia”. Translational Psychiatry 2 (3): e94–.doi:10.1038/tp.2012.15. PMC 3316151. PMID 22832859.
- José Alexandre de Souza Crippa, Antonio Waldo Zuardi, Griselda E J Garrido, Lauro Wichert-Ana, Ricardo Guarnieri, Lucas Ferrari, Paulo M Azevedo-Marques, Jaime Eduardo Cecílio Hallak, Philip K McGuire and Geraldo Filho Busatto (October 2003). “Effects of Cannabidiol (CBD) on Regional Cerebral Blood Flow”.Neuropsychopharmacology 29 (2): 417–426. doi:10.1038/sj.npp.1300340.PMID 14583744.
- Daniel Thomas Malone, Dennis Jongejana and David Alan Taylora (August 2009). “Cannabidiol reverses the reduction in social interaction produced by low dose Δ9-tetrahydrocannabinol in rats”. Pharmacology Biochemistry and Behavior 93 (2): 91–96.doi:10.1016/j.pbb.2009.04.010. PMID 19393686.
- Mateus M Bergamaschi, Regina Helena Costa Queiroz, Marcos Hortes Nisihara Chagas, Danielle Chaves Gomes de Oliveira, Bruno Spinosa De Martinis, Flávio Kapczinski, João Quevedo, Rafael Roesler, Nadja Schröder, Antonio E Nardi, Rocio Martín-Santos, Jaime Eduardo Cecílio (May 2011). “Cannabidiol Reduces the Anxiety Induced by Simulated Public Speaking in Treatment-Naïve Social Phobia Patients”.Neuropsychopharmacology 36 (6): 1219–1226. doi:10.1038/npp.2011.6.PMC 3079847. PMID 21307846.
- Crippa JA, Derenusson GN, Ferrari TB, Wichert-Ana L, Duran FL, Martin-Santos R, Simões MV, Bhattacharyya S, Fusar-Poli P, Atakan Z, Santos Filho A, Freitas-Ferrari MC, McGuire PK, Zuardi AW, Busatto GF, Hallak JE. (January 2011). “Neural basis of anxiolytic effects of cannabidiol (CBD) in generalized social anxiety disorder: a preliminary report”. J Psychopharmacol. 25 (1): 121–130.doi:10.1177/0269881110379283. PMID 20829306.
- ElBatsh, MM; Assareh, N; Marsden, CA; Kendall, DA (May 2012). “Anxiogenic-like effects of chronic cannabidiol administration in rats”. Psychopharmacology 221 (2): 239–247. doi:10.1007/s00213-011-2566-z. PMID 22083592.
- Zanelati, T; Biojone, C; Moreira, F; Guimarães, F; Joca, S (January 2010).“Antidepressant-like effects of cannabidiol in mice: possible involvement of 5-HT1A receptors”. British Journal of Pharmacology 159 (1): 122–8. doi:10.1111/j.1476-5381.2009.00521.x. PMC 2823358. PMID 20002102.
- Réus, GZ; Stringari, RB; Ribeiro, KF; Luft, T; Abelaira, HM; Fries, GR; Aguiar, BW; Kapczinski, F; Hallak, JE; Zuardi, AW; Crippa JA; Quevedo, J (October 2011). “Administration of cannabidiol and imipramine induces antidepressant-like effects in the forced swimming test and increases brain-derived neurotrophic factor levels in the rat amygdala”. Acta Neuropsychiatrica 23 (5): 241–248. doi:10.1111/j.1601-5215.2011.00579.x.
- El-Alfy, AT; Ivey, K; Robinson, K; Ahmed, S; Radwan, M; Slade, D; Khan, I; ElSohly, M; Ross, S (June 2010). “Antidepressant-like effect of Δ9-tetrahydrocannabinol and other cannabinoids isolated from Cannabis sativa L”. Pharmacology Biochemistry and Behavior 95 (4): 434–442. doi:10.1016/j.pbb.2010.03.004. PMC 2866040.PMID 20332000.
- Young, Saundra (15 Jan 2014), 3-year-old is focus of medical marijuana battle, CNN, retrieved 2014-01-16
- Cridge BJ, Rosengren RJ (2013). “Critical appraisal of the potential use of cannabinoids in cancer management”. Cancer Manag Res 5: 301–13.doi:10.2147/CMAR.S36105. PMC 3770515. PMID 24039449.
- http://www.dravetfoundation.org/dravet-syndrome/what-is-dravet-syndrome#sthash.jAC0bZ89.dpuf What is Dravet Syndrome?
- Melville, Nancy A. (14 Aug 2013), Seizure Disorders Enter Medical Marijuana Debate, Medscape Medical News, retrieved 2014-01-14
- Gloss D, Vickrey B (13 June 2012). “Cannabinoids for epilepsy”. Cochrane Database Syst Rev (Review) 6: CD009270. doi:10.1002/14651858.CD009270.pub2.PMID 22696383.
- Romney, Lee (13 September 2012). “On the frontier of medical pot to treat boy’s epilepsy”. Los Angeles Times.
- Jump up^ Good, Alastair (26 October 2010). “Growing marijuana that won’t get you high”. The Daily Telegraph (London).
- Jump up^ Sidner, Sara (8 November 2012). Medical marijuana without the high (video). CNN. “An Israeli company has cultivated a new type of medical marijuana.”
- Jump up^ Solon, Olivia (5 July 2012). “Medical Marijuana Without the High”. Wired.com
- Jump up^ Lubell, Maayan (3 July 2012). “What a drag, Israeli firm grows ‘highless’ marijuana”.Reuters. Retrieved 31 Jan 2014.
- Jump up^ Hayakawa, K.; Mishima, K.; Hazekawa, M.; Sano, K.; Irie, K.; Orito, K.; Egawa, T.; Kitamura, Y.; Uchida, N.; Nishimura, R.; Egashira, N.; Iwasaki, K.; Fujiwara, M. (2008). “Cannabidiol potentiates pharmacological effects of Δ9-tetrahydrocannabinol via CB1 receptor-dependent mechanism”. Brain Research 1188: 157–164.doi:10.1016/j.brainres.2007.09.090. PMID 18021759.
- Jump up^ Pertwee, R. G. (2008). “The diverse CB1 and CB2 receptor pharmacology of three plant cannabinoids: Δ9-tetrahydrocannabinol, cannabidiol and Δ9-tetrahydrocannabivarin”.British Journal of Pharmacology 153 (2): 199–215. doi:10.1038/sj.bjp.0707442.PMC 2219532. PMID 17828291.
- Jump up^ Ryberg E, Larsson N, Sjögren S, et al. (2007). “The orphan receptor GPR55 is a novel cannabinoid receptor”. British Journal of Pharmacology 152 (7): 1092–101.doi:10.1038/sj.bjp.0707460. PMC 2095107. PMID 17876302.
- Jump up^ Russo EB, Burnett A, Hall B, Parker KK (August 2005). “Agonistic properties of cannabidiol at 5-HT1a receptors”. Neurochemical Research 30 (8): 1037–43.doi:10.1007/s11064-005-6978-1. PMID 16258853.
- ^ Jump up to:a b Resstel LB, Tavares RF, Lisboa SF, Joca SR, Corrêa FM, Guimarães FS (January 2009). “5-HT1A receptors are involved in the cannabidiol-induced attenuation of behavioural and cardiovascular responses to acute restraint stress in rats”. British Journal of Pharmacology 156 (1): 181–8. doi:10.1111/j.1476-5381.2008.00046.x.PMC 2697769. PMID 19133999.
- Jump up^ Campos AC, Guimarães FS (August 2008). “Involvement of 5HT1A receptors in the anxiolytic-like effects of cannabidiol injected into the dorsolateral periaqueductal gray of rats”. Psychopharmacology 199 (2): 223–30. doi:10.1007/s00213-008-1168-x.PMID 18446323.
- Jump up^ Mishima K, Hayakawa K, Abe K, et al. (May 2005). “Cannabidiol prevents cerebral infarction via a serotonergic 5-hydroxytryptamine1A receptor-dependent mechanism”.Stroke; a Journal of Cerebral Circulation 36 (5): 1077–82.doi:10.1161/01.STR.0000163083.59201.34. PMID 15845890.
- Jump up^ Hayakawa K, Mishima K, Nozako M, et al. (March 2007). “Repeated treatment with cannabidiol but not Delta9-tetrahydrocannabinol has a neuroprotective effect without the development of tolerance”. Neuropharmacology 52 (4): 1079–87.doi:10.1016/j.neuropharm.2006.11.005. PMID 17320118.
- Jump up^ Kathmann, Markus; Flau, Karsten; Redmer, Agnes; Tränkle, Christian; Schlicker, Eberhard (2006). “Cannabidiol is an allosteric modulator at mu- and delta-opioid receptors”. Naunyn-Schmiedeberg’s Archives of Pharmacology 372 (5): 354–361.doi:10.1007/s00210-006-0033-x. PMID 16489449.
- Jump up^ Bornheim, LM; Kim, KY; Li, J; Perotti, BY; Benet, LZ (August 1995). “Effect of cannabidiol pretreatment on the kinetics of tetrahydrocannabinol metabolites in mouse brain”. Drug Metabolism and Disposition 23 (8): 825–831. PMID 7493549.
- Jump up^ Klein, C; Karanges, E; Spiro, A; Wong, A; Spencer, J; Huynh, T; Gunasekaran, N; Karl, T; Long, LE; Huang, XF; Liu, K; Arnold, JC; McGregor, IS (November 2011). “Cannabidiol potentiates Δ⁹-tetrahydrocannabinol (THC) behavioural effects and alters THC pharmacokinetics during acute and chronic treatment in adolescent rats”.Psychopharmacology 218 (2): 443–457. doi:10.1007/s00213-011-2342-0.PMID 21667074.
- Jump up^ Hunt, CA; Jones, RT; Herning, RI; Bachman, J (June 1981). “Evidence that Cannabidiol Does Not Significantly Alter the Pharmacokinetics of Tetrahydrocannabinol in Man”.Journal of Pharmacokinetics and Biopharmaceutics 9 (3): 245–260.doi:10.1007/BF01059266. PMID 6270295.
- Jump up^ United States Adopted Names Council: Statement on a nonproprietary name
- Jump up^ “Fact Sheet – Sativex”. Health Canada. Retrieved 16 May 2013.
- Jump up^ GWPharma- Welcome
- Jones PG, Falvello L, Kennard O, Sheldrick GM Mechoulam R (1977). “Cannabidiol”.Acta Cryst. B33 (10): 3211–3214. doi:10.1107/S0567740877010577.
- Mechoulam R, Ben-Zvi Z (1968). “Hashish—XIII On the nature of the beam test”.Tetrahedron 24 (16): 5615–5624. doi:10.1016/0040-4020(68)88159-1. PMID 5732891.
- Gaoni Y, Mechoulam R (1966). “Hashish—VII The isomerization of cannabidiol to tetrahydrocannabinols”. Tetrahedron 22 (4): 1481–1488. doi:10.1016/S0040-4020(01)99446-3.
- Petrzilka T, Haefliger W, Sikemeier C, Ohloff G, Eschenmoser A (1967). “Synthese und Chiralität des (-)-Cannabidiols”. Helv. Chim. Acta 50 (2): 719–723.doi:10.1002/hlca.19670500235. PMID 5587099.
- Gaoni Y, Mechoulam R (1985). “Boron trifluoride etherate on alumuna – a modified Lewis acid reagent. An improved synthesis of cannabidiol”. Tetrahedron Letters 26 (8): 1083–1086. doi:10.1016/S0040-4039(00)98518-6.
- Kobayashi Y, Takeuchi A, Wang YG (2006). “Synthesis of cannabidiols via alkenylation of cyclohexenyl monoacetate”. Org. Lett. 8 (13): 2699–2702.doi:10.1021/ol060692h. PMID 16774235.
- Marks, M.; Tian, L.; Wenger, J.; Omburo, S.; Soto-Fuentes, W.; He, J.; Gang, D.; Weiblen, G.; Dixon, R. (2009). “Identification of candidate genes affecting Δ9-tetrahydrocannabinol biosynthesis in Cannabis sativa”. Journal of Experimental Botany60 (13): 3715–3726. doi:10.1093/jxb/erp210. PMC 2736886. PMID 19581347.
- Controlled Drugs and Substances Act – Schedule II
- CSA Schedule, List of drugs by schedule.
- Definition of marijuana under the Controlled Substances Act.
- Title 21 US Code Controlled Substances Act, text of the CSA.
- Hemp Industries Assn., v. Drug Enforcement Admin., 9th Circuit Court of Appeals case involving industrial hemp.
- Hemp, Many definitions of common terms associated with hemp, including the history of hemp use.
- Cannabidiol: The side of marijuana you don’t know
- US patent 6630507, Hampson, Aidan J.; Axelrod, Julius; Grimaldi, Maurizio, “Cannabinoids as antioxidants and neuroprotectants”, issued 2003-10-07
- “Federal Register | Prospective Grant of Exclusive License: Development of Cannabinoid(s) and Cannabidiol(s) Based Therapeutics To Treat Hepatic Encephalopathy in Humans”. Federalregister.gov. November 17, 2011. Retrieved August 13, 2013.
- “KannaLife Sciences, Inc. Signs Exclusive License Agreement With National Institutes Of Health Office Of Technology Transfer (NIH-OTT)”. thestreet.com. Retrieved 2012-07-09.
- “KannaLife in R&D Collaboration for Cannabinoid-Based Drugs”. Genengnews.com. Retrieved 2013-04-04.
External links
- Project CBD Non-profit educational service dedicated to promoting and publicizing research into the medical utility of cannabidiol.
OLD CUT PASTE
Cannabidiol Seven Expanded Access INDs granted by FDA to U.S. physicians to treat with Epidiolex 125 children suffering from intractable epilepsy syndromes -
LONDON, Nov. 15, 2013
GW Pharmaceuticals plc (AIM: GWP, Nasdaq: GWPH, “GW”) announced today that the U.S. Food and Drug Administration (FDA) has granted orphan drug designation for Epidiolex(R), our product candidate that contains plant-derived Cannabidiol (CBD) as its active ingredient, for use in treating children with Dravet syndrome, a rare and severe form of infantile-onset, genetic, drug-resistant epilepsy syndrome. Epidiolex is an oral liquid formulation of a highly purified extract of CBD, a non-psychoactive molecule from the cannabis plant. Following receipt of this orphan designation, GW anticipates holding a pre-IND meeting with the FDA in the near future to discuss a development plan for Epidiolex in Dravet syndrome.
Dravet syndrome is a rare pediatric epilepsy syndrome with a distinctive but complex electroclinical presentation. Onset of Dravet syndrome occurs during the first year of life with clonic and tonic-clonic seizures in previously healthy and developmentally normal infants. Prognosis is poor and patients typically develop intellectual disability and life-long ongoing seizures. There are approximately 5,440 patients with Dravet in the United States and an estimated 6,710 Dravet patients in Europe. These figures may be an underestimate as this syndrome is reportedly underdiagnosed.
In addition to GW’s clinical development program for Epidiolex in Dravet syndrome, which is expected to commence in 2014, GW has also made arrangements to enable independent U.S. pediatric epilepsy specialists to treat high need pediatric epilepsy cases with Epidiolex immediately. To date in 2013, a total of seven “expanded access” INDs have been granted by the FDA to U.S. clinicians to allow treatment with Epidiolex of approximately 125 children with epilepsy. These children suffer from Dravet syndrome, Lennox-Gastaut syndrome, and other pediatric epilepsy syndromes. GW is aware of further interest from additional U.S. and ex-U.S. physicians to host similar INDs for Epidiolex. GW expects data generated under these INDs to provide useful observational data during 2014 on the effect of Epidiolex in the treatment of a range of pediatric epilepsy syndromes.
“I, together with many colleagues in the U.S. who specialize in the treatment of childhood epilepsy, very much welcome the opportunity to investigate Epidiolex in the treatment of Dravet syndrome. The FDA’s timely approval of the orphan drug designation for Epidiolex in Dravet syndrome is a key milestone that comes after many years of reported clinical cases that suggest encouraging evidence of efficacy for CBD in this intractable condition,” stated Dr. Orrin Devinsky, Professor of Neurology, Neurosurgery and Psychiatry in New York City. “With GW now making plans to advance Epidiolex through an FDA development program, we have the prospect for the first time of fully understanding the science of CBD in epilepsy with a view to making an appropriately tested and approved prescription medicine available in the future for children who suffer from this debilitating disease.”
“GW is proud to be at the forefront of this important new program to treat children with Dravet Syndrome and potentially other forms of intractable childhood epilepsy. For families in these circumstances, their lives are significantly impacted by constant and often times very severe seizures in children where all options to control these seizures have been exhausted,” stated Dr. Stephen Wright, GW’s R&D Director. “GW intends to advance a full clinical development program for Epidiolex in Dravet syndrome as quickly as possible, whilst at the same time helping families in the short term through supporting physician-led INDs to treat intractable cases. Through its efforts, GW aims to provide the necessary evidence to confirm the promise of CBD in epilepsy and ultimately enabling children to have access to an FDA-approved prescription CBD medicine.”
“This orphan program for Epidiolex in childhood epilepsy is an important corporate strategic priority for GW. Following receipt of today’s orphan designation, GW now intends to commence discussions with the FDA regarding the U.S. regulatory pathway for Epidiolex,” stated Justin Gover, GW’s Chief Executive Officer. “GW intends to pursue this development in-house and retains full commercial rights to Epidiolex.”
About Orphan Drug Designation
Under the Orphan Drug Act, the FDA may grant orphan drug designation to drugs intended to treat a rare disease or condition — generally a disease or condition that affects fewer than 200,000 individuals in the U.S. The first NDA applicant to receive FDA approval for a particular active ingredient to treat a particular disease with FDA orphan drug designation is entitled to a seven-year exclusive marketing period in the U.S. for that product, for that indication.
About GW Pharmaceuticals plc
Founded in 1998, GW is a biopharmaceutical company focused on discovering, developing and commercializing novel therapeutics from its proprietary cannabinoid product platform in a broad range of disease areas. GW commercialized the world’s first plant-derived cannabinoid prescription drug, Sativex(R), which is approved for the treatment of spasticity due to multiple sclerosis in 22 countries. Sativex is also in Phase 3 clinical development as a potential treatment of pain in people with advanced cancer. This Phase 3 program is intended to support the submission of a New Drug Application for Sativex in cancer pain with the U.S. Food and Drug Administration and in other markets around the world. GW has established a world leading position in the development of plant-derived cannabinoid therapeutics and has a deep pipeline of additional clinical-stage cannabinoid product candidates targeting epilepsy (including an orphan pediatric epilepsy program), Type 2 diabetes, ulcerative colitis, glioma and schizophrenia. For further information, please visit http://www.gwpharm.com.
Cannabidiol (CBD) is one of at least 85 cannabinoids found in cannabis.It is a major constituent of the plant, second totetrahydrocannabinol (THC), and represents up to 40% in its extracts. Compared with THC, cannabidiol is not psychoactive in healthy individuals, and is considered to have a wider scope of medical applications than THC, including to epilepsy, multiple sclerosis spasms, anxiety disorders, bipolar disorder,schizophrenia,nausea, convulsion and inflammation, as well as inhibiting cancer cell growth. There is some preclinical evidence from studies in animals that suggests CBD may modestly reduce the clearance of THC from the body by interfering with its metabolism.Cannabidiol has displayed sedative effects in animal tests. Other research indicates that CBD increases alertness. CBD has been shown to reduce growth of aggressive human breast cancer cells in vitro, and to reduce their invasiveness.