2014 in review……Dr ANTHONY’S “NEW DRUG APROVALS” BLOG


The WordPress.com stats helper monkeys prepared a 2014 annual report for this blog.
Here’s an excerpt:
The Louvre Museum has 8.5 million visitors per year. This blog was viewed about 350,000 times in 2014. If it were an exhibit at the Louvre Museum, it would take about 15 days for that many people to see it.
Important Industrial Procedures Revisited in Flow: Very Efficient Oxidation and N-Alkylation Reactions with High Atom-Economy

http://www.akademiai.com/content/u87p126856085276/?p=2f48c96a10a64882aeb5c47c657a10b7&pi=4
| Journal | Journal of Flow Chemistry |
| Publisher | Akadémiai Kiadó |
| ISSN | 2062-249X (Print) 2063-0212 (Online) |
| Subject | Flow Chemistry |
| Issue | Volume 3, Number 2/June 2013 |
| Pages | 51-58 |
| DOI | 10.1556/JFC-D-12-00025 |
Authors
1ThalesNano Zahony u. 7 1031 Budapest Hungary


Abstract
The atom economy concept is one of the earliest recognition for green and sustainable aspects of organic synthesis. Over the years, novel technologies emerged that made this important feature of reactions into practice. Continuous-flow devices increased the efficiency of the chemical transformations with novel process windows (high T, high p and heterogeneous packed catalysts etc.) and increased safety which turned the attention to reexamine old, industrial processes. Oxidation can be performed under flow catalytic conditions with molecular oxygen; alcohols can be oxidized to carbonyl compounds with high atom economy (AE = 87 %). Using O2 and 1 % Au/TiO2, alcohol oxidation in flow was achieved with complete conversion and >90 % yield. N-alkylation is another good example for achieving high atom economy. Under flow catalytic conditions (Raney Ni), amines were successfully reacted with alcohols directly (AE = 91 %) with >90 % conversion and selectivity. In both examples, the effective residence time was less than 1 min. These two examples demonstrate the significant contribution of flow technology to the realization of key principles in green and sustainable chemistry.
ThalesNano Nanotechnology Inc, GraphisoftPark. Záhony u. 7. H-1031 Budapest HUNGARY
A Method to Identify Best Available Technologies (BAT) for Hydrogenation Reactors in the Pharmaceutical Industry




J. Flow Chem. 2012, 2(3), 77–82
http://www.akademiai.com/content/8652651g3378x686/?p=ab7c1bc4cd7740e1855623297649f542&pi=3
http://www.akademiai.com/content/8652651g3378x686/fulltext.pdf
| Journal of Flow Chemistry | |
| Publisher | Akadémiai Kiadó |
| ISSN | 2062-249X (Print) 2063-0212 (Online) |
| Subject | Flow Chemistry |
| Issue | Volume 2, Number 3/September 2012 |
| Pages | 77-82 |
| DOI | 10.1556/JFC-D-12-00014 |
Authors
1CNRS, CPE Lyon University of Lyon Villeurbanne France
Abstract
A methodology that may be applied to help in the choice of a continuous reactor is proposed. In this methodology, the chemistry is first described through the use of eight simple criteria (rate, thermicity, deactivation, solubility, conversion, selectivity, viscosity, and catalyst). Then, each reactor type is also analyzed from their capability to answer each of these criteria. A final score is presented using “spider diagrams.” Lower surfaces indicate the best reactor choice. The methodology is exemplified with a model substrate nitrobenzene and a target pharmaceutical intermediate, N-methyl-4-nitrobenzenemethanesulphonamide, and for three different continuous reactors, i.e., stirred tank, fixed bed, and an advanced microstructured reactor. Comparison with the traditional batch reactor is also provided.


Fanetizole

Fanetizole
Fanetizole shows immunoregulating activity.
RN: 79069-95-7
Fanetizole mesylate [USAN]
CP-48,810-27
Fanetizole mesylate
UNII-D3OG7B0G4M
Synthesis
Thioureas serve as a convenient starting material for 2-aminothiazoles.
Reaction of β-phenethylamine with ammonium isothiocyanate gives the corresponding thiourea. Treatment of that product with phenacyl bromide thus affords the thiazole product.[1]
- Lombardino, J. G.; 1981, U.S. Patent 4,307,106
| Systematic (IUPAC) name | |
|---|---|
| 4-Phenyl-N-(2-phenylethyl)-1,3-thiazol-2-amine | |
| Clinical data | |
| Legal status |
?
|
| Pharmacokinetic data | |
| Protein binding | % |
| Identifiers | |
| CAS number | 79069-94-6 |
| ATC code | ? |
| PubChem | CID 54339 |
| ChemSpider | 49083 |
| UNII | BH48F620JA |
| Chemical data | |
| Formula | C17H16N2S |
| Mol. mass | 280.39 g/mol |
………………………………………….
Journal of the Chinese Chemical Society, 2009, 56, 455-458
http://proj3.sinica.edu.tw/~chem/servxx6/files/paper_10990_1246593848.pdf
Fanetizole (3j)
mp 114-115 C (Lit.,30 116-117 C). IR (KBr) :3192, 2957, 1562, 1481, 1445, 1332, 698 cm-1;
1H NMR(CDCl3) : 2.81 (t, J = 7.4 Hz, 2H), 3.42 (dd, J = 6.8, 10.8
Hz, 2H), 6.32 (s, 1H), 6.64 (s, 1H), 7.08 (d, J = 6.8 Hz, 2H),
7.15-7.28 (m, 4H), 7.34-7.37 (m, 2H), 7.77-7.80 (m, 2H).
30=. Potewar, T. M.; Ingale, S. A.; Srinivasan, K. V. Tetrahedron
2008, 64, 5019-5022.
…………………………………………
A remarkably high-speed solution-phase combinatorial synthesis of 2-substituted-amino-4-aryl thiazoles in polar solvents in the absence of a catalyst under ambient conditions and study of their antimicrobial activities
ISRN Organic Chemistry (2011), 434613, 6 pp. Publisher: (Hindawi Publishing Corp., )
http://www.hindawi.com/journals/isrn/2011/434613/
……………………………………………
Fanetizole
Ley et al had previously developed a tube-in-tube reactor based on a semipermeable polymer membrane to enable the transfer of gases into liquid flow streams. and here, we demonstrate the scalability and throughput of this reactor when used with ammonia gas. This was made possible by a the inclusion of a titration method to assess parameters including the liquid and gas configuration, reactor temperatures, flow rates, and solvent polarity. These data were then employed in a scaling-up process affording alkyl thioureas which were ultimately used in a telescoped procedure for the preparation of anti-inflammatory agent fanetizole on a multigram scale.

Researchers at Cambridge have shown how it is possible to calibrate a ‘tube-in-tube’ reactor containing ammonia gas using a simple in-line colourimetric titration technique.
This information was then used to deliver an ammonia solution of stoichiometrically to effect the telescoped 2 stage synthesis of the anti-inflammatory agent Fanetizole.
The automated continuous flow synthesiser was able to produce drug substance at a rate of approximately 10 g per hour, isolating the product by direct precipitation from the outflow reaction stream.
Fanetizole: Scaling-up of continuous flow processes with gases using a tube-in-tube reactor: in-line titrations and fanetizole synthesis with ammonia J. Pastre, D.L. Browne, M. O’Brien and S.V. Ley, Org. Proc. Res. Dev. 2013, 17, 1183-1191.
http://pubs.acs.org/doi/full/10.1021/op400152r
………………………..
A Hantzsch synthesis of 2-aminothiazoles performed in a heated microreactor system
DOI: 10.1039/B109360F…….http://pubs.rsc.org/en/content/articlelanding/2002/lc/b109360f/unauth#!divAbstract
………………………………
Bioorganic and Medicinal Chemistry Letters, 1996 , vol. 6, 12 pg. 1409 – 1414
http://www.sciencedirect.com/science/article/pii/0960894X96002417

………………………………………
ref
Heterocycles, 2010 , vol. 81, 12 pg. 2849 – 2854
Journal of the Chinese Chemical Society, 2009 , vol. 56, 3 pg. 455 – 458
Bioorganic and Medicinal Chemistry Letters, 1996 , vol. 6, 12 pg. 1409 – 1414
Pfizer Patent: DD144055DE2922523 , 1979 ;Chem.Abstr., vol. 92, 111001
Organic Process Research and Development, 2013 , vol. 17, 9 pg. 1183 – 1191
Tetrahedron, 2007 , vol. 63, 45 pg. 11066 – 11069
Tetrahedron, 2008 , vol. 64, 22 pg. 5019 – 5022
The application of flow microreactors to the preparation of a family of casein kinase I inhibitors

|
The Application of Flow Microreactors to the Preparation of a Family of Casein Kinase I Inhibitors.
|
||
|
Org. Biomol. Chem. 2010, 8, 1798-1806.
Link: 10.1039/b925327k
|
In this article we demonstrate how a combination of enabling technologies such as flow synthesis, solid-supported reagents and scavenging resins utilised under fully automated software control can assist in typical medicinal chemistry programmes. In particular automated continuous flow methods have greatly assisted in the optimisation of reaction conditions and facilitated scale up operations involving hazardous chemical materials. Overall a collection of twenty diverse analogues of a casein kinase I inhibitor has been synthesised by changing three principle binding vectors.
DOI: 10.1039/B925327K

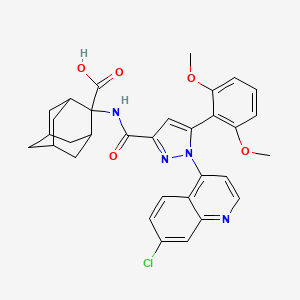
Meclinertant (SR48692)

2-[[1-(7-chloroquinolin-4-yl)-5-(2,6-dimethoxyphenyl)pyrazole-3-carbonyl]amino]adamantane-2-carboxylic acid
Meclinertant (SR-48692) is a drug which acts as a selective, non-peptide antagonist at the neurotensin receptor NTS1, and was the first non-peptide antagonist developed for this receptor.[1][2] It is used in scientific research to explore the interaction between neurotensin and other neurotransmitters in the brain,[3][4][5][6][7][8] and produces anxiolytic, anti-addictive and memory-impairing effects in animal studies.[9][10][11][12]
PatentSubmittedGranted1-(7-chloroquinolin-4-yl)pyrazole-3-carboxamide N-oxide derivatives, method of preparing them, and their pharmaceutical compositions [US5561234]1996-10-01
Substituted 1-naphthyl-3-pyrazolecarboxamides which are active on neurotensin [US5585497]1996-12-17
3-amidopyrazole derivatives, process for preparing these and pharmaceutical composites containing them [US5420141]1995-05-30
Substituted 1-naphthyl-3-pyrazolecarboxamides which are active on neurotensin, their preparation and pharmaceutical compositions containing them [US5523455]1996-06-04
3-amidopyrazole derivatives, process for preparing these and pharmaceutical compositions containing them [US5607958]1997-03-04
3-amidopyrazole derivatives, process for preparing these and pharmaceutical compositions containing them [US5616592]1997-04-01
3-amidopyrazole derivatives, process for preparing these and pharmaceutical compositions containing them [US5635526]1997-06-03
Substituted 1-phenyl-3-pyrazolecarboxamides active on neurotensin receptors, their preparation and pharmaceutical compositions containing them [US5965579]1999-10-12
| Systematic (IUPAC) name | |
|---|---|
| 2-([1-(7-Chloro-4-quinolinyl)-5-(2,6-dimethoxyphenyl)-1H-pyrazole-3-carbonyl]amino)admantane-2-carboxylic acid | |
| Clinical data | |
| Legal status |
?
|
| Identifiers | |
| CAS number | 146362-70-1 |
| ATC code | ? |
| PubChem | CID 119192 |
| IUPHAR ligand | 1582 |
| UNII | 5JBP4SI96H |
| Chemical data | |
| Formula | C32H31ClN4O5 |
| Mol. mass | 587.064 |

A Machine-Assisted Flow Synthesis of SR48692: A Probe for the Investigation of Neurotensin Receptor-1 (pages 7917–7930)
Dr. Claudio Battilocchio, Benjamin J. Deadman, Dr. Nikzad Nikbin, Dr. Matthew O. Kitching, Prof. Ian R. Baxendale and Prof. Steven V. Ley
Article first published online: 16 APR 2013 | DOI: 10.1002/chem.201300696
Flow and pharmaceuticals? An investigation into whether machine-assisted technologies can be of true help in the multistep synthesis of a potent neurotensin receptor-1 probe, Meclinertant (SR48692; see structure), is reported.
Meclinertant (SR 48692)
We developed an improved synthesis of the neurotensin antagonist biological probe SR 48692. The preparation includes an number of chemical conversions and strategies involving the use of flow chemistry platforms which helped overcome some of the limiting synthetic transformations in the original chemical route .
Meclinertant (SR 48692): The synthesis of neurotensin antagonist SR 48692 for prostate cancer research I.R. Baxendale, S. Cheung, M.O. Kitching, S.V. Ley, J.W. Shearman Bio. Org. Med. Chem. 2013, 21, 4378-4387.

A synthesis of the neurotensin 1 receptor probe Merclinertant (SR48692) has been reported using a range of continuous flow through synthesis, in-line reaction monioring and purification techniques. This strategy has been contrasted with a more conventional batch synthesis approach.
Notably the safe use of phosgene gas (generated in situ), the superheating of solvents to accelerate reaction rates, the processing of a reagent suspension under continuous flow-through conditions and the application of semi-permeable membrane technology to facilitate work-up and purification were all techniques that could be beneficially applied in the synthetic scheme.
Abstract:

Meclinertant, Reminertant, SR-48692
The condensation of 2′,6′-dimethoxyacetophenone (I) with diethyl oxalate (II) by means of sodium methoxide in refluxing methanol gives the dioxobutyrate (III), which is cyclized with 7-chloroquinoline-4-hydrazine (IV) in refluxing acetic acid yielding the pyrazole derivative (V). The hydrolysis of the ester group of (V) with KOH in refluxing methanol/water affords the corresponding carboxylic acid (VI), which is finally treated with SOCl2 in refluxing toluene and condensed with 2-aminoadamantane-2-carboxylic acid.
EP 0477049; FR 2665898; JP 1992244065; US 5420141; US 5607958; US 5616592; US 5635526; US 5744491; US 5744493
…………………………….
- Gully D, Canton M, Boigegrain R, Jeanjean F, Molimard JC, Poncelet M, Gueudet C, Heaulme M, Leyris R, Brouard A (January 1993).“Biochemical and pharmacological profile of a potent and selective nonpeptide antagonist of the neurotensin receptor”. Proceedings of the National Academy of Sciences of the United States of America 90 (1): 65–9. doi:10.1073/pnas.90.1.65. PMC 45600. PMID 8380498.
- Gully D, Jeanjean F, Poncelet M, Steinberg R, Soubrié P, Le Fur G, Maffrand JP (1995). “Neuropharmacological profile of non-peptide neurotensin antagonists”. Fundamental & Clinical Pharmacology 9 (6): 513–21. doi:10.1111/j.1472-8206.1995.tb00528.x.PMID 8808171.
- Rostene W, Azzi M, Boudin H, Lepee I, Souaze F, Mendez-Ubach M, Betancur C, Gully D (April 1997). “Use of nonpeptide antagonists to explore the physiological roles of neurotensin. Focus on brain neurotensin/dopamine interactions”. Annals of the New York Academy of Sciences 814: 125–41. doi:10.1111/j.1749-6632.1997.tb46151.x. PMID 9160965.
- Jump up^ Jolas T, Aghajanian GK (August 1997). “Neurotensin and the serotonergic system”. Progress in Neurobiology 52 (6): 455–68.doi:10.1016/S0301-0082(97)00025-7. PMID 9316156.
- Jump up^ Dobner PR, Deutch AY, Fadel J (June 2003). “Neurotensin: dual roles in psychostimulant and antipsychotic drug responses”. Life Sciences73 (6): 801–11. doi:10.1016/S0024-3205(03)00411-9. PMID 12801600.
- Jump up^ Chen L, Yung KK, Yung WH (September 2006). “Neurotensin selectively facilitates glutamatergic transmission in globus pallidus”.Neuroscience 141 (4): 1871–8. doi:10.1016/j.neuroscience.2006.05.049. PMID 16814931.
- Petkova-Kirova P, Rakovska A, Della Corte L, Zaekova G, Radomirov R, Mayer A (September 2008). “Neurotensin modulation of acetylcholine, GABA, and aspartate release from rat prefrontal cortex studied in vivo with microdialysis”. Brain Research Bulletin 77 (2–3): 129–35. doi:10.1016/j.brainresbull.2008.04.003. PMID 18721670.
- Petkova-Kirova P, Rakovska A, Zaekova G, Ballini C, Corte LD, Radomirov R, Vágvölgyi A (December 2008). “Stimulation by neurotensin of dopamine and 5-hydroxytryptamine (5-HT) release from rat prefrontal cortex: possible role of NTR1 receptors in neuropsychiatric disorders”.Neurochemistry International 53 (6–8): 355–61. doi:10.1016/j.neuint.2008.08.010. PMID 18835308.
- Griebel G, Moindrot N, Aliaga C, Simiand J, Soubrié P (December 2001). “Characterization of the profile of neurokinin-2 and neurotensin receptor antagonists in the mouse defense test battery”. Neuroscience and Biobehavioral Reviews 25 (7–8): 619–26. doi:10.1016/S0149-7634(01)00045-8. PMID 11801287.
- Tirado-Santiago G, Lázaro-Muñoz G, Rodríguez-González V, Maldonado-Vlaar CS (October 2006). “Microinfusions of neurotensin antagonist SR 48692 within the nucleus accumbens core impair spatial learning in rats”. Behavioral Neuroscience 120 (5): 1093–102. doi:10.1037/0735-7044.120.5.1093. PMID 17014260.
- Felszeghy K, Espinosa JM, Scarna H, Bérod A, Rostène W, Pélaprat D (December 2007). “Neurotensin receptor antagonist administered during cocaine withdrawal decreases locomotor sensitization and conditioned place preference”. Neuropsychopharmacology 32 (12): 2601–10. doi:10.1038/sj.npp.1301382. PMC 2992550. PMID 17356568.
- Lévesque K, Lamarche C, Rompré PP (October 2008). “Evidence for a role of endogenous neurotensin in the development of sensitization to the locomotor stimulant effect of morphine”.European Journal of Pharmacology 594 (1–3): 132–8. doi:10.1016/j.ejphar.2008.07.048. PMID 18706409.

Continuous Flow Synthesis of alpha-Halo Ketones: Building Blocks for Anti-retroviral Agents

Chiral alpha-halo ketones derived from N-protected amino acids are key building blocks for the synthesis of HIV protease inhibitors such as atazanavir used in HAART combination therapy.
Kappe and De Souza have reported a continuous flow through route to these intermediates which utilises a tube-in-tube reactor to introduce diazomethane generated on demand into the reaction stream containing mixed anhydride derivatives of N-protected amino acids. The resulting alpha-diazo ketones are then decomposed with HCl or HBr to afford the corresponding alpha-halo ketones.
This process allows the safe generation, separation and use of diazomethane in a continuous integrated multi-step synthesis of important API intermediates.

The development of a continuous flow process for the multistep synthesis of α-halo ketones starting from N-protected amino acids is described. The obtained α-halo ketones are chiral building blocks for the synthesis of HIV protease inhibitors, such as atazanavir and darunavir. The synthesis starts with the formation of a mixed anhydride in a first tubular reactor.
The anhydride is subsequently combined with anhydrous diazomethane in a tube-in-tube reactor. The tube-in-tube reactor consists of an inner tube, made from a gas-permeable, hydrophobic material, enclosed in a thick-walled, impermeable outer tube. Diazomethane is generated in the inner tube in an aqueous medium, and anhydrous diazomethane subsequently diffuses through the permeable membrane into the outer chamber.
The α-diazo ketone is produced from the mixed anhydride and diazomethane in the outer chamber, and the resulting diazo ketone is finally converted to the halo ketone with anhydrous ethereal hydrogen halide.
This method eliminates the need to store, transport, or handle diazomethane and produces α-halo ketone building blocks in a multistep system without racemization in excellent yields. A fully continuous process allowed the synthesis of 1.84 g of α-chloro ketone from the respective N-protected amino acid within ∼4.5 h (87% yield).
Arteflene





Safe Generation and Synthetic Utilization of Hydrazoic Acid in a Continuous Flow Reactor.

Safe Generation and Synthetic Utilization of Hydrazoic Acid in a Continuous Flow Reactor.
B. Gutmann, J.-P. Roduit, D. Roberge, C. O. Kappe, J. Flow Chem. 2012, 2,8-19.
http://www.akademiai.com/content/l622j82k3171t080/?p=0213e26b691f494d8eb782308d34fe77&pi=2
Authors
1Christian Doppler Laboratory for Microwave Chemistry and Institute of Chemistry, Karl-Franzens-University Graz A-8010 Heinrichstrasse 28 Graz Austria
2Microreactor Technology, Lonza AG CH-3930 Visp Switzerland
Abstract
Hydrazoic acid (HN3) was used in a safe and reliable way for the synthesis of 5-substitued-1H-tetrazoles and for the preparation of N-(2-azidoethyl)acylamides in a continuous flow format. Hydrazoic acid was generated in situ either from an aqueous feed of sodium azide upon mixing with acetic acid, or from neat trimethylsilyl azide upon mixing with methanol.
For both processes, subsequent reaction of the in situ generated hydrazoic acid with either organic nitriles (tetrazole formation) or 2-oxazolines (ring opening to β-azido-carboxamides) was performed in a coil reactor in an elevated temperature/pressure regime. Despite the explosive properties of HN3, the reactions could be performed safely at very high temperatures to yield the desired products in short reaction times and in excellent product yields.
The scalability of both protocols was demonstrated for selected examples. Employing a commercially available benchtop flow reactor, productivities of 18.9 g/h of 5-phenyltetrazole and 23.0 g/h of N-(1-azido-2-methylpropan- 2-yl)acetamide were achieved.

Keywords

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
















 Click here to see the complete report.
Click here to see the complete report.