BRIVARACETAM
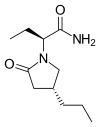
BRIVARACETAM, UCB-34714
(2S)-2-[(4R)-2-oxo-4-propylpyrrolidin-1-yl]butanamide
| Molecular Formula: | C11H20N2O2 |
|---|---|
| Molecular Weight: | 212.2887 g/mol |
UNII-U863JGG2IA
UCB; For the treatment of partial onset seizures related to epilepsy, Approved February 2016
Brivaracetam, the 4-n-propyl analog of levetiracetam, is a racetam derivative with anticonvulsant properties.[1][2] Brivaracetam is believed to act by binding to the ubiquitous synaptic vesicle glycoprotein 2A (SV2A).[3] Phase II clinical trials in adult patients with refractory partial seizures were promising. Positive preliminary results from stage III trials have been recorded,[4][5] along with evidence that it is around 10 times more potent[6] for the prevention of certain types of seizure in mouse models than levetiracetam, of which it is an analogue.
On 14 January 2016, the European Commission,[7] and on 18 February 2016, the USFDA[8] approved brivaracetam under the trade name Briviact (by UCB). The launch of this anti-epileptic is scheduled for the first quarter of that year. Currently, brivaracetam is still not approved in other countries like Australia, Canada and Switzerland.
Brivaracetam was approved by European Medicine Agency (EMA) on Jan 14, 2016 and approved by the U.S. Food and Drug Administration (FDA) on Feb 18, 2016. It was developed and marketed as Briviact® by UCB in EU/US.
Brivaracetam is a selective high-affinity synaptic vesicle protein 2A ligand, as an adjunctive therapy in the treatment of partial-onset seizures with or without secondary generalization in adult and adolescent patients from 16 years of age with epilepsy.
Briviact® is available in three formulations, including film-coated tablets, oral solution and solution for injection/infusion. And it will be available as 10 mg, 25 mg, 50 mg, 75 mg and 100 mg film-coated tablets, a 10 mg/ml oral solution, and a 10 mg/ml solution for injection/infusion. The recommended starting dose is either 25 mg twice a day or 50 mg twice a day, depending on the patient’s condition. The dose can then be adjusted according to the patient’s needs up to a maximum of 100 mg twice a day. Briviact can be given by injection or by infusion (drip) into a vein if it cannot be given by mouth.
European Patent No. 0 162 036 Bl discloses compound (S)-α-ethyl-2-oxo-l- pyrrolidine acetamide, which is known under the International Non-proprietary Name of Levetiracetam.
Levetiracetam
Levetiracetam is disclosed as a protective agent for the treatment and prevention of hypoxic and ischemic type aggressions of the central nervous system in European patent EP 0 162 036 Bl. This compound is also effective in the treatment of epilepsy.
The preparation of Levetiracetam has been disclosed in European Patent No. 0 162 036 and in British Patent No. 2 225 322.
International patent application having publication number WO 01/62726 discloses 2-oxo-l -pyrrolidine derivatives and methods for their preparation. It particularly discloses compound (2S)-2-[(4R)-2-oxo-4-propyl-pyrrolidin-l-yl] butanamide known under the international non propriety name of brivaracetam.
Brivaracetam
International patent application having publication number WO 2005/121082 describes a process of preparation of 2-oxo-l -pyrrolidine derivatives and particularly discloses a process of preparation of (2S)-2-[(4S)-4-(2,2-difluorovinyl)-2-oxo-pyrrolidin-l- yl]butanamide known under the international non propriety name of seletracetam.
Seletracetam
Kenda et al., in J. Med. Chem. 2004, 47, 530-549, describe processes of preparation of 2-oxo-l -pyrrolidine derivatives and particularly discloses compound 1-((1S)-I- carbamoyl-propyl)-2-oxo-pyrrolidone-3-carboxylic acid as a synthetic intermediate.
WO2005028435
CLIPS
Find better ways to make old and new epilepsy drugs. J. Surtees and co-inventors disclose alternative processes for making active pharmaceutical ingredients (APIs) that are used to treat epilepsy and seizures. One compound that can be prepared by their processes is the established drug levetiracetam (1, Figure 1), marketed under the trade name Keppra. Because 1 is now off-patent, there is obvious interest in new drugs.

The inventors also claim that seletracetam (2) and brivaracetam (3) (Figure 2) can be prepared by their processes. These drugs are apparently much more active than 1.
All of the drugs are used as single isomers, so a stereoselective synthesis is desirable. The inventors describe two routes for preparing the molecules; the first, shown in Figure 1, is the synthesis of 1 by the reaction between pyrrolidone (4) and chiral bromo amide 5 in the presence of a base. GC analysis showed that the conversion is 40.3% and that the product contains 51% of the (S)-enantiomer and 49% of the (R)-isomer. No details of their separation are given, although the use of chiral HPLC is discussed.
The same reaction is used to prepare derivative 6 of 1. Compound 7 is prepared from the corresponding hydroxy ester and then condensed with 4 to give 6. Chiral HPLC showed that the product is a mixture of 89.3% (S)-enantiomer 6 and 10.7% of its (R)-isomer.
The inventors do not describe the detailed preparation of 2, but they report that acid 8 is prepared in 41% yield from pyrrolidone 9 and acid 10 in the presence of NaH (Figure 2). Ammonolysis of 8 produces 2; no reaction details are provided.

In a reaction similar to the preparation of 8, acid 11 is prepared from 10 and pyrrolidone 12. The product is isolated in 77% yield and can be converted to 3 by ammonolysis. Again, no details are provided for this reaction.
The second route for preparing the substituted pyrrolidones does not start with simple pyrrolidones and is the subject of additional claims. The route involves a cyclization reaction, shown in Figure 3. The preparation of enantiomer 13 begins with the reaction of racemic salt 14 and optically pure bromo ester 15. This step produces intermediate 16, isolated as a yellow oil. The crude material is treated with 2-hydroxypyridine (2-HP) to cyclize it to 17. This ester is hydrolyzed to give acid 18. Conversion to 13 is carried out by adding ClCO2Et, followed by reaction with liquid NH3 in the presence of K2CO3. The overall yield of 13 is 32%.

This route is also used to prepare levetiracetam (1) by treating 5 with the HCl salt of amino ester 19 to give 20, recovered as its HCl salt in 49% yield. The salt is basified with Et3N and treated with 2-HP to cyclize it to 1, initially isolated as an oil. GC analysis showed 100% conversion, and chiral HPLC showed that the product contains 98.6% (S)-isomer and 1.4% (R)-isomer.
The inventors also prepared 1 and its (R)-enantiomer 21 by using a similar reaction scheme with alternative substrates to 5. Figure 4 outlines the route, which starts from protected hydroxy amide 22 and amino ester 23. When the reaction is carried out in the presence of Cs2CO3, the product is (R)-enantiomer24, which is used without purification to prepare 21 by treating it with 2-HP. Chiral HPLC showed that the product is 94% (R) and 6% (S).

When the reaction between 22 and 23 is run with K2CO3, the product is (S)-enantiomer 25. This is used to prepare 1, but the product contains only 79% (S)-isomer.
The inventors do not comment on the apparent stereoselectivity of the carbonate salts in the reaction of 22 with 23. This is an intriguing finding and worthy of investigation. (UCB S.A. [Brussels]. US Patent 8,338,621

SYNTHESIS
PATENT
WO2005028435
Example 1: Synthesis of (2S)-2-((4R)-2-oxo-4-n-propyl-l-pyrrolidinyl)butanamide 1.1 Synthesis of (2S)-2-aminobutyramide free base
1800 ml of isopropanol are introduced in a 5L reactor. 1800 g of (2S)-2- aminobutyramide tartrate are added under stirring at room temperature. 700 ml of a 25% aqueous solution of ammonium hydroxide are slowly added while maintaining the temperature below 25°C. The mixture is stirred for an additional 3 hours and then the reaction is allowed to complete at 18°C for 1 hour. The ammonium tartrate is filtered. Yield : 86%.
1.2 Synthesis of 5-hydroxy-4-n-propyl-furan-2-one
Heptane (394 ml) and morpholine (127.5 ml) are introduced in a reactor. The mixture is cooled to 0°C and glyoxylic acid (195 g, 150 ml, 50w% in water) is added. The mixture is heated at 20°C during 1 hour, and then valeraldehyde (148.8 ml) is added . The reaction mixture is heated at 43°C during 20 hours. After cooling down to 20CC, a 37 % aqueous solution of HCl (196.9 ml) is slowly added to the mixture, which is then stirred during 2 hours.
After removal of the heptane phase, the aqueous phase is washed three times with heptane. Diisopropyl ether is added to the aqueous phase. The organic phase is removed, and the aqueous phase further extracted with diisopropyl ether (2x). The diisopropyl ether phases are combined, washed with brine and then dried by azeotropic distillation. After filtration and evaporation of the solvent, 170g of 5- hydroxy-4-n-propyl-furan-2-one are obtained as a brown oil. Yield: 90.8 %
1.3 Synthesis of (2S)-2-((4R)-2-oxo-4-n-propyl-l-pyrrolidinyl)butanamide and (2S)-2-((4S)-2-oxo-4-n-propyl-l-pyrrolidinyl)butanamide
(S, R) (S, S) The (2S)-2-aιninobutyrarnide solution in isopropanol containing 250 g obtained as described here above is dried by azeotropic distillation under vacuum. To the dried (2S)-2-am obutyraιnide solution is added 5-hydroxy-4-n-propyl-furan-2-one (290 g) between 15°C and 25 °C; the mixture is heated to 30 °C and kept for at least 2 hours at that temperature. Acetic acid (1, 18 eq.), Pd/C catalyst (5 w/w%; Johnson Matthey 5% Pd on carbon – type 87L) are then added and hydrogen introduced into the system under pressure. The temperature is kept at 40 °C maximum and the H2 pressure maintained between 0,2 bar and 0,5 bar followed by stirring for at least 20 hours following the initial reaction. The solution is then cooled to between 15 °C and 25 °C and filtered to remove the catalyst. The solution of product in isopropanol is solvent switched to a solution of product in isopropyl acetate by azeotropic distillation with isopropyl acetate. The organic solution is washed with aqueous sodium bicarbonate followed by a brine wash and then filtered. After recristallisation, 349 g of (2S)-2-((4R)-2- oxo-4-n-propyl-l-pyrrolidinyl)butanamide and (2S)-2-((4S)-2-oxo-4-n-propyl-l- pyιτolidinyl)butanamide are obtained (Yield: 82.5%).
1.4 Preparation of (2S)-2-((4R)-2-oxo-4-n-propyl-l-pyrrolidinyl)butanamide The chromatographic separation of the two diastereoisomers obtained in 1.3 is performed using of (CHIRALPAK AD 20 um) chiral stationary phase and a 45/55 (volume /volume) mixture of n-heptane and ethanol as eluent at a temperature of 25 + 2°C. The crude (2S)-2-((4R)-2-oxo-4-n-propyl-l-pyrrolidinyl)butanamide thus obtained is recristallised in isopropylacetate, yielding pure (2S)-2-((4R)-2-oxo-4-n-propyl-l- pyrrolidinyl)butanamide (Overall yield: 80%) .
Example 2: Synthesis of (2S)-2-((4R)-2-oxo-4-n-propyl-l-pyrrolidinyl)butanamide
Example 1 is repeated except that in step 1.1 a solution of (2S)-2- aminoburyramide.HCl in isopropanol is used (27.72 g, 1.2 equivalent), which is neutralised with a NHs/isopropanol solution (3,4-3,7 mol/L). The resulting ainmonium chloride is removed from this solution by filtration and the solution is directly used for reaction -with 5-hydroxy-4-n-propyl-furan-2-one (23.62 g, 1.0 equivalent) without intermediate drying of the (2S)-2-aminobutyramide solution. Yield after separation of the two diastereoisomers and recristallisation: approximately 84%.

Ref ROUTE1
1. WO0162726A2.
2. WO2005028435A1 / US2007100150A1.
3. J. Med. Chem. 1988, 31, 893-897.
4. J. Org. Chem. 1981, 46, 4889-4894.
PATENT
https://www.google.com/patents/WO2007031263A1?cl=en
Example 3-Synthesis of brivaracetam (I)
3.a. Synthesis of (S) and (R) 2-((R)-2-oxo-4-propyl-pyrrolidin-l-yl)-butyric acid methyl ester fVIaa*) and (Wlab)
(VIaa) (VIab) A slurry of 60% sodium hydride suspension in mineral oil (0.94g, 23.4 mmol) in tetrahydrofuran (30 mL) is cooled at 0°C under a nitrogen atmosphere. A solution of substantially optically pure (R)-4-propyl-pyrrolidin-2-one (Ilia) (2g, 15.7 mmol) in tetrahydrofuran (2 mL) is added over a 15 minutes period. The reaction mixture is stirred 10 min at 0°C then a solution of methyl-2-bromo-butyric acid methyl ester (V) (3.69g, 20.4 mmol) in tetrahydrofuran (2mL) was added over a 20 minutes period. The reaction mixture is stirred at O0C until maximum conversion of starting material and the reaction mixture is then allowed to warm to room temperature and diluted with water (20 mL). Tetrahydrofuran is removed by evaporation and the residue is extracted with isopropyl acetate (20 ml + 10 mL). The combined organic layers are dried on anhydrous magnesium sulfate and evaporated to afford 3g (13.2 mmol, 86 %) of a mixture of epimers of compound (Via), as a mixture respectively of epimer (VIaa) and epimer (VIab). 1H NMR(400 MHz, CDCI3) of the mixture of epimers (VIaa) and (VIab) : δ = 4.68
(dd, J= 10.8, J= 5.1, 2×1 H) ; 3.71 (s, 2x3H); 3.60 (t app, J= 8.2, IH); 3.42 (t app, J= 8.7, IH); 313 (dd, J= 9.2, J = 6.8, IH); 2.95 (dd, J= 9.2, J= 6.8, IH); 2.56 (dd, J= 16.6, J = 8.7, 2xlH); 2.37 (dm, 2xlH); 2.10 (m, 2xlH); 2.00 (m, 2xlH); 1.68 (m, 2xlH); 1.46 (m, 2x2H); 1.36 (m, 2x2H); 0.92 (m, 2x6H).
13C NMR (400 MHz, CDCl3) of the mixture of epimers (VIaa) and (VIab) : δ =
175.9; 175.2; 171.9; 55.3; 52.4; 49.8; 49.5; 38.0; 37.8; 37.3; 36.9; 32.5; 32.2; 22.6; 22.4; 21.0; 14.4; 11.2; 11.1
HPLC (GRAD 90/10) of the mixture of epimers (VIaa) and (VIab): retention time= 9.84 minutes (100 %)
GC of the mixture of epimers (VIaa) and (VIab): retention time = 13.33 minutes (98.9 %)
MS of the mixture of epimers (VIaa) and (VIab) (ESI) : 228 MH+
3.b. Ammonolysis of compound of the mixture of (VIaa) and (VIab)
(VIaa) (VIab) (I) (VII)
A solution of (VIaa) and (VIab) obtained in previous reaction step (1.46g, 6.4 mmol) in aqueous ammonia 50 % w/w (18 mL) at 00C is stirred at room temperature for 5.5hours. A white precipitate that appears during the reaction, is filtered off, is washed with water and is dried to give 0.77g (3.6 mmol, yield = 56 %) of white solid which is a mixture of brivaracetam (I) and of compound (VII) in a 1 :1 ratio.
1H NMR of the mixture (I) and (VII) (400 MHz, CDCI3) : δ = 6.36 (s, broad, IH); 5.66 (s, broad, IH); 4.45 (m, IH); 3.53 (ddd, J= 28.8, J= 9.7, J= 8.1, IH); 3.02 (m, IH); 2.55 (m, IH); 2.35 (m, IH); 2.11 (m, IH); 1.96 (m, IH); 1.68 (m, IH); 1.38 (dm, 4H); 0.92 (m, 6H). 13c NMR of the mixture (I) and (VII) (400 MHz, CDCl3) : δ = 176.0; 175.9; 172.8;
172.5; 56.4; 56.3; 50.0; 49.9; 38.3; 38.1; 37.3; 37.0; 32.3; 32.2; 21.4; 21.3; 21.0; 20.9; 14.4; 10.9; 10.8
HPLC (GRAD 90/10) of the mixture of (I) and (VII) retention time= 7.67 minutes (100 %)
Melting point of the mixture of (I) and (VII) = 104.90C (heat from 400C to 1200C at 10°C/min)
Compounds (I) and (VII) are separated according to conventional techniques known to the skilled person in the art. A typical preparative separation is performed on a 11.7g scale of a 1 :1 mixture of compounds (I) and (VII) : DAICEL CHIRALPAK® AD 20 μm, 100*500 mm column at 300C with a 300 mL/minutes debit, 50 % EtOH – 50 % Heptane. The separation affords 5.28g (45 %) of compound (VII), retention time = 14 minutes and 5.2Og (44 %) of compounds (I), retention time = 23 minutes.
1H NMR of compound (I) (400 MHz, CDCl3): δ = 6.17 (s, broad, IH); 5.32 (s, broad, IH); 4.43 (dd, J= 8.6, J= 7.1, IH); 3.49 (dd, J= 9.8, J= 8.1, IH); 3.01 (dd, J= 9.8, J= 7.1, IH); 2.59 (dd, J= 16.8, J= 8.7, IH); 2.34 (m, IH); 2.08 (dd, J= 16.8, J= 7.9, IH); 1.95 (m, IH); 1.70 (m, IH); 1.47-1.28 (m, 4H); 0.91 (dt, J= 7.2, J= 2.1, 6H)
HPLC (GRAD 90/10) of compound (I) : retention time = 7.78 minutes
1H NMR of compound (VII) (400 MHz, CDCl3): δ = 6.14 (s, broad, IH); 5.27 (s, broad, IH); 4.43 (t app, J = 8.1, IH); 3.53 (t app, J = 9.1, IH); 3.01 (t app, J = 7.8, IH); 2.53 (dd, J = 16.5, J = 8.8, IH); 2.36 (m, IH); 2.14 (dd, J = 16.5, J = 8.1, IH); 1.97 (m, IH); 1.68 (m, IH); 1.43 (m, 2H); 1.34 (m, 2H); 0.92 (m, 6H)
3c. Epimerisation of compound of (2RV2-((R)-2-oxo-4-propyl-pyπOlidin-l-ylV butyramide (VID
Compound (VII) (200 mg, 0.94 mmol) is added to a solution of sodium tert- butoxide (20 mg, 10 % w/w) in isopropanol (2 mL) at room temperature. The reaction mixture is stirred at room temperature for 18h. The solvent is evaporated to afford 200 mg
(0.94 mmol, 100 %) of a white solid. Said white solid is a mixture of brivaracetam (I) and of (VII) in a ratio 49.3 / 50.7.
HPLC (ISO80): retention time= 7.45 min (49.3%) brivaracetam (I); retention time= 8.02 minutes (50.7%) compound (VII).

Reference:ROUTE 2
1. WO2007031263A1 / US2009318708A1.
PATENT
http://www.google.com/patents/WO2007065634A1?cl=en
(scheme 3).
Scheme 3
scheme 4.
5h. Synthesis of brivaracetam and (V) A suspension of (Id) and (Ie) (0.6 g, 2.3 mmol) in MIBK (10 mL) is heated at
120°C for 6 hours. The resulting solution is concentrated and separated on chromatography column (Silicagel 600.068-0.200 mm, cyclohexane/EtOAc : 10/90) to give 0.13 g of brivaracetam (0.6 mmol, 26 %, ee = 94 %) and (V).
1H NMR (400 MHz, CDCl3): δ = 6.17 (s, broad, IH); 5.32 (s, broad, IH); 4.43 (dd, J= 8.6, J= 7.1, IH); 3.49 (dd, J= 9.8, J= 8.1, IH); 3.01 (dd, J= 9.8, J= 7.1, IH); 2.59 (dd, J= 16.8, J= 8.7, IH); 2.34 (m, IH); 2.08 (dd, J= 16.8, J= 7.9, IH); 1.95 (m, IH); 1.70 (m, IH); 1.47-1.28 (m, 4H); 0.91 (dt, J= 7.2,J= 2.1, 6H).
HPLC (method 90/10) : Retention time = 7.78 minutes Chiral HPLC : Retention time = 9.66 minutes (97%) MS (ESI): 213 MH+
Route 3
Reference:1. WO2007065634A1 / US2009012313A1.
PAPER
http://pubs.acs.org/doi/abs/10.1021/acs.oprd.6b00094
A Biocatalytic Route to the Novel Antiepileptic Drug Brivaracetam


AS ON DEC2021 3,491,869 VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@amcrasto
/////////////////////////////////////////////////////////////////////////////
References
- von Rosenstiel P (Jan 2007). “Brivaracetam (UCB 34714)”. Neurotherapeutics 4 (1): 84–7. doi:10.1016/j.nurt.2006.11.004.PMID 17199019.
- Malawska B, Kulig K (Jul 2005). “Brivaracetam UCB”. Current Opinion in Investigational Drugs 6 (7): 740–746. PMID 16044671.
- Rogawski MA, Bazil CW (Jul 2008). “New molecular targets for antiepileptic drugs: alpha(2)delta, SV2A, and K(v)7/KCNQ/M potassium channels”. Current Neurology and Neuroscience Reports 8 (4): 345–352. doi:10.1007/s11910-008-0053-7. PMC 2587091.PMID 18590620.
- Clinical trial number NCT00464269 for “Double-blind, Randomized Study Evaluating the Efficacy and Safety of Brivaracetam in Adults With Partial Onset Seizures” at ClinicalTrials.gov
- Rogawski MA (Aug 2008). “Brivaracetam: a rational drug discovery success story”. British Journal of Pharmacology 154 (8): 1555–7.doi:10.1038/bjp.2008.221. PMC 2518467. PMID 18552880.
- Matagne A, Margineanu DG, Kenda B, Michel P, Klitgaard H (Aug 2008). “Anti-convulsive and anti-epileptic properties of brivaracetam (ucb 34714), a high-affinity ligand for the synaptic vesicle protein, SV2A”. British Journal of Pharmacology 154 (8): 1662.doi:10.1038/bjp.2008.198. PMID 18500360.
- http://www.ema.europa.eu/ema/index.jsp?curl=pages/medicines/human/medicines/003898/human_med_001945.jsp&mid=WC0b01ac058001d124
- http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm486827.htm
|
|
|||
| Names | |||
|---|---|---|---|
| IUPAC name
(2S)-2-[(4R)-2-oxo- 4-propylpyrrolidin-1-yl] butanamide
|
|||
| Identifiers | |||
| 357336-20-0 |
|||
| ChEMBL | ChEMBL607400 |
||
| ChemSpider | 8012964 |
||
| Jmol interactive 3D | Image | ||
| PubChem | 9837243 | ||
| UNII | U863JGG2IA |
||
| Properties | |||
| C11H20N2O2 | |||
| Molar mass | 212.15 g/mol | ||
| Pharmacology | |||
| ATC code | N03 | ||
| Legal status |
|
||
| Oral | |||
| Pharmacokinetics: | |||
| Nearly 100% | |||
| <20% | |||
| Hydrolysis, CYP2C8-mediated hydroxylation | |||
| 8 hrs | |||
| >75% renal | |||
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
|
|||
//////BRIVARACETAM, UCB, 2016 FDA, UCB-34714
CCCC1CC(=O)N(C1)C(CC)C(=O)N
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2021260721&_cid=P12-KXX1JU-33531-1
Brivaracetam is chemically known as (2S)-2-[(4R)-2-oxo-4-propyltetrahydro-1H-pyrrol-1-yl] butanamide, having the chemical structure of formula 1 as below:
Brivaracetam is basically a chemical analogue of Levetiracetam, marketed under the brand name of BRIVIACT for the treatment as adjunctive therapy in the treatment of partial-onset seizures in patients at 16 years of age and older with epilepsy. Brivaracetam has an advantage over Levetiracetam in that it gets into the brain “much more quickly,” which means that “it could be used for status epilepticus, or acute seizures than cluster, or prolonged seizures”. From the Phase III trials, the self-reported rate of irritability with Brivaracetam was 2% for both drug doses (100 mg and 200 mg) vs 1% for placebo, which compares to as much as 10% for Levetiracetam in some post-marketing studies.
With the improved safety profile and possibility to be used for wider range of epilepsy, Brivaracetam is considered as one of the most promising 3rd generation antiepileptic drugs.
Brivaracetam molecule is first disclosed in patent publication WO2001062726, which describes 2-oxo-1 -pyrrolidine derivatives and methods for their preparation. This patent publication further discloses compound (2S)-2-[(4R)-2-oxo-4-propyl-pyrrolidin-1-yl] butanamide which is known under the international non propriety name as Brivaracetam. As per Biopharmaceutics Classification System, Brivaracetam is a class I drug (high solubility and permeability).
Some prior arts US6784197 and US7629474 disclose a process for synthesizing a diastereomeric mixture of (2S)-2-[(4R)-2-oxo-4-propylpyrrolidin-1-yl]-butanamide and (2S)-2-[(4S)-2-oxo-4-propylpyrrolidin-1-yl]-butanamide (Brivaracetam) which is purified by chiral HPLC (Scheme-I & Scheme-II respectively, as provided below). This process used for chiral resolution makes it difficult for bulk manufacturing as well as it affects the overall yield making the process uneconomical.
Scheme-I
Synthesis of (2S)-2-(2-oxo-4-propyl-1-pyrrolidinyl)butanamide
[As disclosed in columns 37-38 of US 6784197 B2]
Scheme-II
1.1 Synthesis of (2S)-2-aminobutyramide-Free base
1.2 Synthesis of 5-hydroxy-4-n-propylfuran-2-one
1.3 Synthesis of (2S)-2-((4R)-2-oxo-4-n-propyl-1-pyrrolidinyl)butanamide and
(2S)-2-((4S)-2-oxo-4-n-propyl-1-pyrrolidinyl)butanamide
[As disclosed in columns 6-7 of US 7629474 B2]
Moreover, some prior arts such as US7122682B2, US8076493B2, US8338621B2 and US8957226B2 also describe processes for preparing Brivaracetam, wherein, the purifications are reportedly done by chiral HPLC methods resulting into similar shortcomings.
Kenda et al.: Journal of Medicinal Chemistry, 2004, 47, 530-549 further proposes selection of (2S)-2-[(4R)-2-oxo-4-propylpyrrolidin-1-yl]butanamide 83α (ucb 34714; Brivaracetam) as the most interesting candidate showing 10 times more potency than Levetiracetam as an antiseizure agent in audiogenic seizure-prone mice. This article further discloses methods for synthesizing the said compound Brivaracetam. However, here too these compounds are synthesized as mixtures of stereoisomers (racemic or diastereoisomeric mixtures), separated by preparative HPLC on silica gel and/or chiral phases.
All these processes for the preparation of Brivaracetam as described in the above- mentioned prior arts suffer from many disadvantages which includes difficulty in achieving desired chiral purity, tedious and cumbersome work up procedures, high temperature and longer time reaction, multiple crystallizations or isolation steps, use of excess reagents and solvents, column chromatographic separations & purifications etc. All these disadvantages affect the overall yield as well as the quality of the final product Brivaracetam and intermediates produced thereof; further, rendering such processes to be uneconomical and unsuitable for industrial scale-ups.
As a result, enantioselective synthesis of Brivaracetam was perceived to be a possible way of overcoming such problems in view of the large-scale synthesis. However, very few prior arts have been found to report successful reduction of such concept into practice.
WO2016191435A1 (also as IN201717005820A) relates to a process for a scalable synthesis of enantiomerically pure Brivaracetam from an intermediate (4R)-4- Propyldihydrofuran-2(3H)-one (compound IV):
, wherein, R is saturated or unsaturated C1-20 alkyl or C1 alkyl-unsubstituted or substituted aryl, comprising the steps of decarboxylation of
the compound of formula IV
to produce the compound of formula VI
ring-opening of the compound of formula VI to produce the compound of formula VII
, wherein Rl is saturated or unsaturated Cl-20 alkyl or Cl alkyl-unsubstituted or substituted aryl; and X is CI Br I OMs, OTs, ONs; or
the compound of formula X
reacting the compound of formula VI with (S)-2- aminobutanamide or its salt to produce the compound of formula XII in one step; or reacting the compound of formula VI with alkyl (S)-2- aminobutanoate to produce Xll-a
, wherein R in the compound of formula Xll-a is a saturated or unsaturated C1-C20 alkoxyl; and then converting Xll-a to XII that is Brivaracetam by aminolysis and amide formation reaction.
In above mentioned prior arts, the synthesis of chiral lactone which is the key starting material for making Brivaracetam involved Grignard addition, column chromatography and Krapcho decarboxylation techniques at high temperature, all of which are not at all recommendable in view of process perspective at industrial levels. Further the final step of the said reaction often involved cryogenic condition –30°C which is also difficult with respect to industrial scale up activities.
Furthermore, prior art IN201641030239A disclosed a process for the preparation of Brivaracetam of Formula (I) by means of converting enantiomerically pure compound of Formula VII to obtain enantiomerically pure compound of Formula XI:
, wherein X is each independently selected from halogen; alkyl or aryl sulfonyloxy; OR2; R2 is optionally substituted C1-C12 alkyl, aryl, alkyl aryl, aryl alkyl;
such that the said process further comprises steps of:
1) cyclizing compound of formula VII to give enantiomerically pure compound of formula IX:
, wherein R2 is optionally substituted C1-C12 alkyl, aryl, alkyl aryl or aryl alkyl;
2) converting the compound of formula IX to give a enantiomerically pure compound of formula X:
, wherein X is halogen;
3) converting compound of formula X to give a enantiomerically pure compound of formula XI:
wherein X is each independently selected from halogen; alkyl or aryl sulfonyloxy; OR2; R2 as defined above; followed by 4) treating the enantiomerically pure compound of formula XI with (S)- aminobutyramide of formula XII or its salt thereof to obtain Brivaracetam of Formula I.
However, this process suffered from drawbacks of handling acid chloride. Acid chlorides are unstable and storing a large amount of acid chloride is also not recommendable in view of safety and stability in industries. Moreover, this prior art process involves use of HBr in acetic acid, where HBr liberates Br that is hazardous and not recommendable for industrial scale-up activities in view of safety and handling.
Some other prior arts such as CN108503573A, CN105646319A, CN106588740A, CN106588831A, CN108689903B and CN108929289A report various processes of synthesizing Brivaracetam from its lactone intermediate by various ring opening techniques. Among these, in particular CN108929289A discloses a process of reacting a compound represented by the formula IV with (S) -2-aminobutyramide in order to obtain Brivaracetam. The synthetic route is as follows:
Also, CN108689903B relates to a new preparation method of Brivaracetam that comprises steps of: a) subjecting a compound of formula III and (S) -2-aminobutanamide or salt thereof to condensation reaction, in the presence of a condensing agent, in order to obtain a compound shown in a formula IV, wherein the compound has two chiral centres; b) removal of the hydroxy-protecting group R1 to obtain a compound of formula V; and c) carrying out chlorination reaction on the compound shown in the formula V using a chlorination reagent to obtain a compound shown in the formula VI; and d) carrying out substitution reaction on the compound shown in the formula VI in the presence of an
alkaline reagent, and closing a ring to obtain Brivaracetam of formula I having two chiral centres.
It has further been noted that although the above reaction goes through formation of intermediates V and/or VI; however, these intermediates are not essentially formed from the key lactone intermediate of Brivaracetam that is (4R)-4-propyldihydrofuran-2(3H)-one [or (R)-lactone].
Furthermore, CN111196771A relates to a preparation method of Brivaracetam which comprises the steps of: 1) carrying out ring-opening reaction on a compound R-4-propyldihydrofuran-2-ketone in a formula II and a compound S-2-aminobutanamide in a formula III to obtain an intermediate compound in a formula I; 2) condensing the said intermediate compound of formula I is followed by cyclization to produce Brivaracetam
However, the ring-opening reaction in step 1 of this process essentially occurs under acidic conditions, specifically in presence of Lewis acids like tetra-isopropyl titanate, anhydrous aluminium trichloride, anhydrous zinc chloride, boron trifluoride diethyl etherate etc.; and also in presence of organic solvents chosen from one or more of anhydrous tetrahydrofuran, 2-methyltetrahydrofuran, acetone, dimethyl sulfoxide and N, N-dimethylformamide; which makes this process both industrially non-scalable and environmentally unfriendly.
The prior art Org. Process Res. Dev.2016. v 20. no 9. p 1566-1575 in its scheme 4, on page 17 also discloses a scheme for synthesizing Brivaracetam from its lactone intermediate:
Nevertheless, it has been noted that the process reported in this prior art provides Brivaracetam (API) with a very poor yield of ~30% and also having an inferior chiral purity of 95.9% ee, which does not even meet the ICH-specification for the Finished Product (API).
Furthermore, a recently filed patent application WO2020148787A1 (also as IN201931002041) recites a new, improved and economical process for enantioselective synthesis and purification of a key intermediate of Brivaracetam that is the R-lactone, essentially utilizing a low chiral loading and without involving any chiral chromatographic resolution technique. Even though this prior art also discloses a process for the preparation of a chirally pure Brivaracetam of formula I utilizing the said intermediate; however, that process is mostly a conventional one.
Accordingly, there is still a need in the art for a more economical and improved process for the synthesis of Brivaracetam with better purity and yield which overcomes the drawbacks of above prior arts.
Therefore, the present inventors have developed a cost effective, novel and efficient process for the preparation of Brivaracetam which essentially avoids all the drawbacks involved in prior art as mentioned above. The currently developed process is advantageously capable of producing the key lactone intermediate with more than 80% ee applying transfer hydrogenation with a very simple operation in view of process perspective. Further, by means of using such chiral lactone with more than 80% ee, the currently developed process is also capable of delivering >99.9% chirally pure Brivaracetam with excellent yield.
EXAMPLES:
EXAMPLE 1: Synthesis of (3R)-N-[(1S)-1-carbamoylpropyl]-3-(hydroxymethyl) hexanamide [Intermediate 7 of scheme A of the present invention]
Example 1 illustrates one pot process for preparing purified (3R)-N-[(1S)-1- carbamoylpropyl]-3-(hydroxymethyl) hexanamide [Intermediate 7] from Intermediate 3 (80% ee) as developed in step 1 of scheme A of the present invention.
Procedure:
In the first step of scheme A of the present invention, a mixture of (R)/(S)-4-propyldihydrofuran-2(3H)-one (Intermediate 3, R: S isomer = 80: 20) (1 eq), (S)-2-aminobutanamide (1.1 eq), triethylamine (1.5 eq) is refluxed at a temperature of 95±5 °C for 24h. The mixture is then cooled to 60-65 °C, washed with a mixture of dichloromethane and diisopropyl ether (2.5 vol) in order to get Intermediate-7 [(3R)-N-[(1S)-1-carbamoyl-propyl]-3-(hydroxymethyl) hexanamide] (80% yield).
Results:
Formation of Intermediate 7 is confirmed further by following analytical studies: a) The 1H NMR analysis is conducted and the data as illustrated in accompanying figure 1 depicts: (400 MHz, DMSO-d6): δ 0.6 (t, J= Hz, 6H), 1.07-1.18 (m, 1H), 1.21-1.35 (m, 3H), 1.45-1.43 (m, 1H), 1.61-1.72 (m, 1H), 1.75-1.90 (m, 1H), 2.03 (dd, J=6.64 & 14.08 Hz, 1H), 2.18 (dd, J=7.0 & 14.08 Hz, 1H), 3.28 (t, J=5.36 Hz, 2H), 4.07-4.18 (m, 1H), 4.43 (t, J=5.2 Hz, 1H), 6.95 (s, 1H), 7.28 (s, 1H), 7.76 (d, J=8.08 Hz, 1H).; thus, confirming formation of Intermediate 7 of the present invention.
b) The LCMS analysis is further conducted and the data as graphically illustrated in accompanying figure 2 provides a (M+H+) value of 231.0; thus, confirming formation of Intermediate 7 of the present invention.
c) The HPLC study is also conducted and the data as graphically illustrated in accompanying figure 3 confirms formation of Intermediate 7 of the present invention with chiral purity of 97.38%
EXAMPLE 2: Preparation of (3R)-N-(1S)-1-amino-1-oxobutan-2-yl)-3-(chloromethyl) hexanamide [Intermediate 8A of scheme A of the present invention]: Example 2 illustrates a process for preparing (3R)-N-(1S)-1-Amino-1-oxobutan-2-yl)-3-(chloromethyl) hexanamide [Intermediate 8A] from Intermediate 7 of example 1 above as developed in the present invention.
Procedure:
In second step of scheme A of the present invention, the said Intermediate 7 of example 1 above that is (3R)-N-(1S)-1-Amino-1-oxobutan-2-yl)-3-(hydroxymethyl) hexanamide (~98% Chemical purity and ~97% Chiral purity) (1736.86 mmol) is dissolved in DCM (1.2 L) at RT into a RBF under N2 atm. Then the solution is cooled to 10-20 C and Oxaloyl chloride (2605.29 mmol) is added to this cooled solution at 10-20 °C. The mixture is stirred for 24 h at 25-40 °C under N2 atm. Completion of the reaction is monitored by TLC. After completion of reaction, the solvent is distilled off and the residual mass is diluted with water (6 L), stirred at 30-50 °C for 4 h. Slurry mass is then filtered and washed with water (2×400 mL) followed by MTBE (800 mL). The solid is dried under vacuum at 50-55 °C for 4-5 h to afford Intermediate 8A that is (3R)-N-(1S)-1-Amino-1-oxobutan-2-yl)-3-(chloromethyl) hexanamide as a white solid (92% yield).
Results:
Formation of Intermediate 8A is confirmed further by following analytical studies: a) The 1H NMR analysis is conducted and the data as illustrated in accompanying figure 4 depicts: 1H NMR (400 MHz, DMSO-d6) : δ 0.83 (t, J=7.44 Hz, 3H), 0.85 (t, J=6.72 Hz, 3H), 1.20-1.40 (m, 4H), 1.43-1.56 (m, 1H), 2.08-2.18 (m, 1H), 2.20-2.28 (m, 2H), 3.61 (dd, J=4.6 & 10.8 Hz, 1 H), 3.67 (dd, J=4.6 & 10.8 Hz, 1H), 4.07-4.18 (m, 1H), 6.95 (s, 1H), 7.29 (s, 1H), 7.89 (d, J=8.12 Hz, 1H); thus confirming formation of Intermediate 8A of the present invention.
b) The LCMS analysis is further conducted and the data as graphically illustrated in accompanying figure 5 (a, b) provides a (M+H+) value of 249.20; thus, confirming formation of Intermediate 8A of the present invention.
EXAMPLE 3: Process for purification of Intermediate 8A forming Intermediate 8B Example 3 illustrates a process for purifying the said Intermediate 8A of example 2 above of the present invention.
Procedure:
The Intermediate 8A as obtained in example 2 above [that is (3R)-N-(1S)-1-Amino-1-oxobutan-2-yl)-3-(chloromethyl) hexanamide] is first dissolved in a polar solvent like Acetonitrile raising the temperature to 50 to 60 °C; followed by stirring and then addition of another solvent methyl tert-butyl ether (MTBE) which is less polar in nature. The mixture is then cooled down to 0°C, the filtered mass thus obtained is dried in order to obtain a white solid of Intermediate 8B. The material thus obtained is further dissolved in THF (5 vol) at 60 °C, cooled to 20-30°C, followed by addition of heptane (5 vol), stirred at 10°C to 30 °C for 1 h. The mass obtained is filtered and washed with heptane (2×1 vol), dried under vacuum at 50-55°C in order to afford formation of purer form of Intermediate 8A that is Intermediate 8B that is (3R)-N-(1S)-1-amino-1-oxobutan-2-yl)-3-(chloromethyl) hexanamide as a white solid having a chemical purity of 99.9% along with a chiral purity of 100% (yield: 390 g).
Results:
The purification of Intermediate 8A is further confirmed by the following analytical test results:
a) Chiral HPLC: A Chiral HPLC as illustrated in accompanying figure 6 confirmed formation of purest form of Intermediate 8B having 100% chiral purity [Peak 1; RT (min) = 6.244; %Area=100%].
b) GLP-HPLC: A GLP-HPLC as illustrated in accompanying figure 7 further confirmed formation of Intermediate 8B having 99.9% chemical purity [Peak 3; BRIV8; RT = 29.278; % Area=99.90%].
EXAMPLE 4: Synthesis of (3R)-N-[(1S)-1-carbamoylpropyl]-3-(hydroxymethyl) hexanamide [Intermediate 7’ of scheme B of the present invention]
Example 4 illustrates one pot process for preparing purified (3R)-N-[(1S)-1-carbamoylpropyl]-3-(hydroxymethyl) hexanamide [Intermediate 7’] from Intermediate 6 (99.99% ee) as developed in step-1 scheme B of the present invention.
Procedure:
In another method, in the first step of scheme B of the present invention, a mixture of (R)/(S)-4-propyldihydrofuran-2(3H)-one (Intermediate 6: S isomer = 99.99% : 0.1%) (1 eq), (S)-2-aminobutanamide (1.7 eq), triethylamine (5 eq) is refluxed at a temperature between 95±5 °C for 24 h. Then, the crude reaction mass is cooled and washed with dichloromethane and diisopropyl ether mixture (2.5 vol) in order to achieve Intermediate-7’ of scheme B of the present invention [(3R)-N-[(1S)-1-carbamoylpropyl]-3-(hydroxymethyl) hexanamide] (90% yield).
Results:
The chiral purity of the formed Intermediate 7’ is analyzed by HPLC method and the data as graphically illustrated in accompanying figure 8 confirms formation of Intermediate 7’ of the present invention with chiral purity of 99.11%
EXAMPLE 5: Preparation of (3R)-N-(1S)-1-amino-1-oxobutan-2-yl)-3-(chloromethyl) hexanamide [Intermediate 8’ of scheme B of present invention]: Example 5 illustrates a process for preparing purest form of (3R)-N-(1S)-1-amino-1-oxobutan-2-yl)-3-(chloromethyl) hexanamide [Intermediate 8’] from Intermediate 7’ of example 4 as developed in scheme B (step 2) of the present invention.
Procedure:
In the second step of scheme B of the present invention, the intermediate 7’ of the above example 4 that is (3R)-N-(1S)-1-amino-1-oxobutan-2-yl)-3-(hydroxymethyl) hexanamide (98% Chemical purity and >99%Chiral purity) (1736.86 mmol) is dissolved in DCM (1.2 L) at RT in a round bottomed flask under N2 atm. Then the solution is cooled to 10-30 °C and 1-Chloro-N,N,2-trimethyl-1-propenylamine (2605.29 mmol) is added to this cooled solution at 10-30 °C. The mixture is stirred for 24 h at 25-40 °C under N2 atm. Completion of the reaction is monitored by TLC. After completion of the reaction, the solvent is distilled off and the residual mass is diluted with water (6 L), stirring at 30-50 °C for 4 h. The slurry mass thus obtained is then filtered and washed with water (2×400 mL) followed by methyl tert-buty ether (MTBE) (800 mL). The solid thus obtained is dried under vacuum at 50-55 °C for 4-5 h in order to afford formation of Intermediate 8’ that is (3R)-N-(1S)-1-amino-1-oxobutan-2-yl)-3-(chloromethyl) hexanamide as a white solid (92% yield).
Results:
Formation of Intermediate 8’ is confirmed further by following analytical studies: a) The 1H NMR analysis is conducted and the data as illustrated in accompanying figure 9 depicts: (400 MHz, DMSO-d6): 1H NMR (400 MHz, DMSO-d6) : δ 0.83 (t, J=7.44 Hz, 3H), 0.85 (t, J=6.72 Hz, 3H), 1.20-1.40 (m, 4H), 1.43-1.56 (m, 1H), 2.08-2.18 (m, 1H), 2.20-2.28 (m, 2H), 3.61 (dd, J=4.6 & 10.8 Hz, 1 H), 3.67 (dd, J=4.6 & 10.8 Hz, 1H), 4.07-4.18 (m, 1H), 6.95 (s, 1H), 7.29 (s, 1H), 7.89 (d, J=8.12 Hz, 1H); thus, confirming formation of Intermediate 8’ of the present invention.
b) The LCMS analysis is further conducted and the data as graphically illustrated in accompanying figure 10 provides a (M+H+) value of 249.1; thus, confirming formation of Intermediate 8’ of the present invention.
c) The HPLC data as illustrated in accompanying figure 11 confirms 100% chiral purity of Intermediate 8’.
EXAMPLE 6: Preparation of (2S)-2-[(4R)-2-oxo-4-propyl-pyrrolidin-1-yl] butanamide [Brivaracetam-API]:
Example 6 illustrates a process for preparing (2S)-2-[(4R)-2-oxo-4-propyl-pyrrolidin-1-yl] butanamide [Brivaracetam API] from Intermediate 8B of example 3 or from Intermediate 8’ of example 5 as developed in step 3 of scheme A or scheme B of the present invention respectively.
Procedure:
In the final step of scheme A or scheme B of the present invention, the intermediate 8B of example 3 or intermediate 8’ of example 5 above that is (3R)-N-((1S)-1-Amino-1-oxobutan-2-yl)-3-(chloromethyl) hexanamide (1608.04 mmol) is dissolved in dimethyl acetamide (0.5 vol) and isopropylacetate (2 L) into a RBF at 25-30 °C under N2 atm. Then 18-Crown-6 (160.79 mmol) is added into the solution and stirred at RT for 30 min. Reaction mixture is then cooled to 0-10 °C and t-BuOK (1.5 eq) is added portion wise to the cooled solution over 1 h maintaining the temperature from – 0-10 °C to 25 °C under N2 atm. Stirring is then continued for 2 h at -10 °C to 0 °C and then for 12 h at 15-25 °C under N2 atm. Completion of reaction is monitored by TLC. After completion of reaction, the reaction mixture is quenched with addition of 1M HCl solution (pH~6.5-7.0). The resulting mixture is extracted with i-PrOAc (2 L) and MTBE (1 L). Water (0.5 L) is added to the combined organic extract and then filtered through celite bed, washed the bed with MTBE-i-PrOAc (1:1) (400 mL). The organic part is separated and the aqueous part is re-extracted with i-PrOAc-MTBE (1:1) (2 ×0.8 L). The combined organic phases are washed with brine solution (100 mL), dried over anhydrous Na2SO4, filtered and concentrated in vacuum under a rotary evaporator to afford crude API. Distillation of dimethylacetamide solvent from the crude is then done at high vacuum pressure (0.05 mm Hg) at 70 °C. Crude product is then dissolved in isopropyl acetate (1.6 L) and treated with activated charcoal (7% w/w) to afford a tech-grade crude of Brivaracetam API as a white solid (yield: 90%) with 97.82% chemical purity.
Results:
Formation of Brivaracetam API is confirmed further by following analytical studies: a) The 1H NMR analysis is conducted and the data as illustrated in accompanying figure 12 depicts: 1H NMR (400 MHz, DMSO-d6) : δ 0.77 (t, J=7.32 Hz, 3H), 0.87 (t, J=7.2 Hz, 3H), 1.21-1.31 (m, 2H), 1.33-1.43 (m, 2H), 1.50-1.62 (m, 1H), 1.73-1.84 (m, 1H), 1.97 (dd, J=8.0 & 16.12 Hz, 1H), 2.18-2.28 (m, 1H), 2.37 (dd, J=8.4 & 16.14 Hz, 1H), 3.11 (dd, J=7.16 & 9.44 Hz, 1H), 3.36 (dd, J=9.2 & 17.5 Hz, 1H), 4.30 dd, J=5.44 & 10.28 Hz, 1H), 6.98 (s, 1H), 7.32 (s, 1H); thus, confirming formation of Brivaracetam API of the present invention.
b) The LCMS analysis is further conducted and the data as graphically illustrated in accompanying figure 13 provides a (M+H+) value of 213.0; thus, confirming formation of Brivaracetam API of the present invention.
^ Purification of Brivaracetam API:
The Brivaracetam thus formed above is further purified by means of dissolving the said material (307 g) in 30% i-PrOAc -MTBE (1 vol) at 55-60 °C, cool to 20-30°C. A mixture of Heptane and MTBE and DIPE (2:2:1) is added, stirred at 10 °C to 30°C for 1 h. The obtained mass is filtered and washed with heptane, which is subsequently dried under vacuum at 40-45 °C to afford (3R)-N-((1S)-1-amino-1-oxobutan-2-yl)-3-(chloromethyl) hexanamide as a white solid (yield: 80%, chiral purity 99.93% and chemical purity 99.94%).
Results:
a) Chiral HPLC: A Chiral HPLC as illustrated in accompanying figure 14 confirmed formation of purest form of Brivaracetam API having 99.93% chiral purity [Peak 2; RT (min) = 9.45; %Area=99.93%].
b) GLP-HPLC: A GLP-HPLC as illustrated in accompanying figure 15 further confirmed formation of Brivaracetam API having 99.9% chemical purity [Peak 2; RT = 21.138; % Area=99.94%].
PF-06747775 (Pfizer) Third generation covalent EGFR inhibitors

 .
.

PF-06747775 (Pfizer)
PF06747775; PF06747775; PF 06747775; PF6747775; PF 6747775; PF6747775. PFE-X775
N-((3R,4R)-4-fluoro-1-(6-((3-methoxy-1-methyl-1H-pyrazol-4-yl)amino)-9-methyl-9H-purin-2-yl)pyrrolidin-3-yl)acrylamide
N-((3R,4R)-4-fluoro-1-(6-((3-methoxy-1-methyl-1H-pyrazol-4-yl)amino)-9-methyl-9H-purin-2-yl)pyrrolidin-3-yl)acrylamide
CAS 1776112-90-3
Chemical Formula: C18H22FN9O2
Exact Mass: 415.188
Recruiting, Phase I/II (NTC02349633)
Epidermal growth factor receptor antagonists
Antineoplastics
Non-small cell lung cancer
Dose escalation study to evaluate safety, PK, PD and efficacy in advanced EGFRm+ NSCLC
- 02 May 2015Phase-I clinical trials in Non-small cell lung cancer (Metastatic disease, Second-line therapy or greater) in USA (PO) (NCT02349633)
- 05 Feb 2015Pfizer plans a phase I trial for Non-small cell lung cancer (Second-line therapy or greater) in USA (NCT02349633)
- 05 Jan 2015Preclinical trials in Non-small cell lung cancer in USA (PO)
PF-06747775 is an orally available inhibitor of the epidermal growth factor receptor (EGFR) mutant form T790M, with potential antineoplastic activity. EGFR T790M inhibitor PF-06747775 specifically binds to and inhibits EGFR T790M, a secondarily acquired resistance mutation, which prevents EGFR-mediated signaling and leads to cell death in EGFR T790M-expressing tumor cells. Compared to some other EGFR inhibitors, PF-06747775 may have therapeutic benefits in tumors with T790M-mediated drug resistance.
for the oral treatment of patients with locally advanced or metastatic EGFR mutant (del19 or L858R) non-small cell lung cancer
Kinetic mechanism for two-step covalent inhibition of EGFR
PATENT
Example 7
(Scheme F): Preparation of N-((3R,4R)-4-fluoro-1-(6-((3-methoxy-1-methyl-1H-pyrazol-4-yl)amino)-9-methyl-9H-purin-2-yl)pyrrolidin-3-yl)acrylamide
Step 1: Preparation of 2-fluoro-N-(3-methoxy-1-methyl-1H-pyrazol-4-yl)-9H-purin -6-amine
Step 2: Preparation of 2-fluoro-N-(3-methoxy-1-methyl-1H-pyrazol-4-yl)-9-methyl -9H-purin-6-amine
Step 3: Preparation of N-((3R,4R)-4-fluoro-1-(6-((3-methoxy-1-methyl-1H-pyrazol -4-yl)amino)-9-methyl-9H-purin-2-yl)pyrrolidin-3-yl)acrylamide
Example 7A
(Scheme F): Preparation of N-((3R,4R)-4-fluoro-1-(6-((3-methoxy-1-methyl-1H-pyrazol-4-yl)amino)-9-methyl-9H-purin-2-yl)pyrrolidin-3-yl)acrylamide
Preparation Step 1A: Preparation of (3R,4R)-1-benzyl-3,4-dihydroxypyrrolidine-2,5-dione
Preparation Step 2A: Preparation of (3S,4S)-1-benzylpyrrolidine-3,4-diol
Preparation Step 3A: Preparation of (3aR,6aS)-5-benzyl-2,2-dioxo-tetrahydro-1-oxa-2λ6-thia-3-5-diaza-pentalene-3-carboxylic acid t-butyl ester
Preparation Step 4A: Preparation of (3R,4R)-1-benzyl-4-fluoropyrrolidin-3-amine bis-tosylate
Preparation Step 5A: N-((3R,4R)-1-benzyl-4-fluoropyrrolidin-3-yl)-3-(methylsulfonyl)propanamide
Preparation Step 6A: N-((3R,4R)-4-fluoropyrrolidin-3-yl)-3-(methylsulfonyl)propanamide
Step 1: Preparation of 2-fluoro-N-(3-methoxy-1-methyl-1H-pyrazol-4-yl)-9H-purin-6-amine
Step 2: Preparation of 2-fluoro-N-(3-methoxy-1-methyl-1H-pyrazol-4-yl)-9-methyl-9H-purin-6-amine
Step 3: Preparation of N-((3R,4R)-4-fluoro-1-(6-((3-methoxy-1-methyl-1H-pyrazol-4-yl)amino)-9-methyl-9H-purin-2-yl)pyrrolidin-3-yl)acrylamide
Summary of 1st generation and 2nd generation EGFR inhibitors
REFERENCES
Planken, S.; Murray, B. W.; Lafontaine, J.; Weinrich, S.; Hemkens, M.; Kath, J. C.; Nair, S. K.; Johnson, T. O.; Cheng, H.; Sutton, S. C.; Zientek, M.; Yin, M. -J.; Solowiej, J.; Nagata, A.; Gajiwala, K. Abstracts of Papers, 249th ACS National Meeting & Exposition, Denver, CO, United States, March 22–26, 2015; MEDI-248
//////Third generation, covalent EGFR inhibitors, PF-06747775, Pfizer, PFE-X775
Compound name AND SMILES string
Rociletinib COC(C=C(N1CCN(C(C)=O)CC1)C=C2)=C2NC3=NC=C(C(F)(F)F)C(NC4=CC=CC(NC(C=C)=O)=C4)=N3
Osimertinib CN(CCN(C)C)C(C(NC(C=C)=O)=C1)=CC(OC)=C1NC2=NC=CC(C3=CN(C)C4=C3C=CC=C4)=N2
EGF816 ClC1=C2C(N=C(NC(C3=CC(C)=NC=C3)=O)N2[C@H]4CN(C(/C=C/CN(C)C)=O)CCCC4)=CC=C1
PF-06747775 CN1C2=NC(N3C[C@@H](NC(C=C)=O)[C@H](F)C3)=NC(NC4=CN(C)N=C4OC)=C2N=C1
PF-06459988 CN(N=C1)C=C1NC2=NC3=C(C(Cl)=CN3)C(OC[C@H]4CN(C(C=C)=O)C[C@@H]4OC)=N2
WZ4002 ClC1=CN=C(NC2=C(OC)C=C(N3CCN(C)CC3)C=C2)N=C1OC4=CC=CC(NC(C=C)=O)=C4
罗西替尼 роцилетиниб روسيليتينيب Rociletinib, CO-1686. Third generation covalent EGFR inhibitors

Rociletinib (CO-1686)
AVL-301,CNX-419
Celgene (Originator) , Clovis Oncology
- Molecular FormulaC27H28F3N7O3
- Average mass555.552
- HYDROBROMIDE 1446700-26-0
Molecular Weight 636.46 Formula C27H28F3N7O3 ● HBr
Cellular proliferation IC507–32 nM against EGFRm+ NSCLC cells
547 nM against A431 cell with WT EGFR
Ongoing, not currently recruiting
Phase I/II (NCT01526928)
Recruiting
Phase III (NCT02322281, TIGER-3)
Evaluate safety, PK and efficacy of previously treated NSCLC patients, Compare the efficacy of oral single agent versus single agent cytotoxic chemotherapy in patients with EGFRm+ NSCLC after failure of at least 1 previous EGFR-directed TKI and at least 1 line of platinum-containing doublet therapy. Compare the safety and efficacy of CO-1686 versus erlotinib as first line treatment of patients with EGFRm+ NSCLC
Rociletinib (CO-1686): Rociletinib is an orally administered irreversible inhibitor currently in several clinical trials targeting both the activating EGFR mutations and the acquired T790M resistance mutation while sparing WT EGFR. It is a potent inhibitor of EGFR T790M/L858R double mutant with a kinact/Ki of 2.41 × 105 M−1 s−1. It has a 22-fold selectivity over WT EGFR (kinact/Ki of 1.12 × 104 M−1 s−1). In NSCLC cell lines containing EGFR mutations, rociletinib demonstrates the following cellular pEGFR IC50: 62 nM in NCI-1975 (L858R/T790M), 187 nM in HCC827 (exon 19 deletion), 211 nM in PC9 (exon 19 deletion). In cell lines expressing WT EGFR, cellular pEGFR IC50 are: >4331 nM in A431, >2000 nM in NCI-H1299, and >2000 nM in NCI-H358.
Rociletinib displayed good oral bioavailability (65%) and long half-life when dosed at 20 mg/kg in female Nu/Nu mice. In tumor bearing mice when rociletinib was dosed orally once daily as a single agent, the compound showed dose-dependent tumor growth inhibition in various EGFR-mutant models. In NCI-H1975 as well as in patient-derived LUM 1868 lines expressing the EGFR T790M/L858R double mutation that are erlotinib-resistant models, rociletinib caused tumor regressions at 100 mg/kg/d. In the HCC827 xenograft model that expresses the del-19 activating EGFR mutation, rociletinib showed antitumor activity that was comparable with erlotinib and the second-generation EGFR TKI, afatinib. The wild-type sparing feature of rociletinib was further demonstrated through its minimal inhibition (36%) of tumor growth in the A431 xenograft model that is dependent on WT EGFR for proliferation.
In a Phase I/II study (TIGER-X), rociletinib was administered to patients with EGFR mutated NSCLC who had disease progression during treatment with a previous line of EGFR TKI therapy.The Phase I trial was a dose escalation study to assess safety, side-effect profile and pharmacokinetic properties of rociletinib, and the Phase II trial was an expansion arm to evaluate efficacy. T790M positivity was confirmed before enrollment in the Phase II portion. At the dose of 500 mg BID, the objective response rate in 243 centrally confirmed tissues from T790M positive patients was 60% and the disease control rate was 90%. The estimated overall median PFS at the time of the publication (May 2015) was 8.0 months among all centrally confirmed T790M positive patients. Rociletinib also showed activity in centrally confirmed T790M negative patients with the overall response rate being 37%. The common dose-limiting adverse event was grade 3 hyperglycemia occurring in 17% of patients at a dose of 500 mg BID. Grade 3 QTc prolongation was observed in 2.5% of the patients at the same dose. Treatment-related adverse events leading to drug discontinuation was seen in only 2.5% of patients at 500 mg BID.
Patent
WO2012061299A1
http://www.google.co.in/patents/WO2012061299A1?cl=en
EXAMPLE 1
Intermediate 1
Scheme 1
Step 1 :
In a 25 mL 3-neck RBF previously equipped with a magnetic stirrer, Thermo pocket and CaCl2 guard tube, N-Boc-l,3-diaminobenzene (0.96 g) and n-butanol (9.00 mL) were charged. Reaction mixture was cooled to 0 °C. 2,4-Dichloro-5-trifluoromethylpyrimidine (1.0 g) was added dropwise to the above reaction mixture at 0 °C. The DIPEA (0.96 mL) was dropwise added to the above reaction mixture at 0 °C and the reaction mixture was stirred for 1 hr at 0 °C to 5 °C. Finally the reaction mixture was allowed to warm to room temperature. Reaction mixture was stirred for another 4 hrs at room temperature. Completion of reaction was monitored by TLC using hexane: ethyl acetate (7: 3). The solid precipitated out was filtered off and washed with 1-butanol (2 mL). Solid was dried under reduced pressure at 40 °C for 1 hr. ^-NMR (DMSO-d6, 400 MHz) δ 1.48 (S, 9 H), 7.02 (m, 1 H), 7.26 (m, 2 H), 7.58 (S, 1 H), 8.57 (S, 1 H), 9.48 (S, 1 H), 9.55 (S, 1 H).
Step 2:
To the above crude (3.1 g) in DCM (25 mL) was added TFA (12.4 mL) slowly at 0 °C. The reaction mixture was allowed to warm to room temperature. Reaction mixture was stirred for another 10 min at room temperature. The crude was concentrated under reduced pressure.
Step 3:
The concentrated crude was dissolved in DIPEA (2.0 mL) and DCM (25 mL), and then cooled to -30 °C. To the reaction mixture was slowly added acryloyl chloride (0.76 g) at -30 °C. The reaction mass was warmed to room temperature stirred at room temperature for 1.0 hr. The reaction was monitored on TLC using hexane: ethyl acetate (7:3) as mobile phase. Reaction got completed after 1 hr. 1H-NMR (DMSO-d6, 400 MHz) δ 5.76 (dd, J = 2.0, 10.0 Hz, 1 H), 6.24 (dd, J = 2.0, 17.2 Hz, 1 H), 6.48 (m, 1 H), 7.14 (d, J = 8.8 Hz, 1 H), 7.37 (t, J = 8.0 Hz, 1 H), 7.94 (S, 1 H), 8.59 (S, 1 H), 9.60 (S, 1 H), 10.26 (S, 1 H).
EXAMPLE 3
Compound 1-4 N- henylamino)-5-
(trifluor mide)
Using 2-methoxy-4-(4-acteylpiperazinyl)aniline and intermediate 1 in Example 1, the title compound 1-4 was prepared as described in Example 2. 1H-NMR (DMSO-d6, 400 MHz) δ 10.2 (S, 1 H), 8.2 (br, 1 H), 8.30 (S, 1 H), 7.73 (br, 1 H), 7.52 (d, J = 7.8 Hz, 1 H), 7.45 (d, J = 7.8 Hz, 1 H), 7.26 (J = 8.2 Hz, 1 H), 7.14 (be, 1 H), 6.60 (S, 1 H), 6.42 (dd, J = 11.4, 16.9 Hz, 1 H), 6.24 (d, J = 16.9 Hz, 1 H), 5.75 (d, J = 11.4 Hz, 1 H), 3.76 (S, 3 H), 3.04 (br, 4 H), 2.04 (S, 3 H); calculated mass for C27H28F3N7O3 : 555.2, found: 556.2 (M+H+).
| Patent ID | Date | Patent Title |
|---|---|---|
| US2015344441 | 2015-12-03 | SALTS OF AN EPIDERMAL GROWTH FACTOR RECEPTOR KINASE INHIBITOR |
| US2015246040 | 2015-09-03 | HETEROCYCLIC COMPOUNDS AND USES THEREOF |
| US2015225422 | 2015-08-13 | HETEROARYLS AND USES THEREOF |
| US8975249 | 2015-03-10 | Heterocyclic compounds and uses thereof |
| US2013267531 | 2013-10-10 | SALTS OF AN EPIDERMAL GROWTH FACTOR RECEPTOR KINASE INHIBITOR |
| US2013267530 | 2013-10-10 | SOLID FORMS OF AN EPIDERMAL GROWTH FACTOR RECEPTOR KINASE INHIBITOR |
References
////Rociletinib, CO-1686, Clovis, Third generation, covalent EGFR inhibitors, AVL-301, CNX-419
CC(=O)N1CCN(CC1)C2=CC(=C(C=C2)NC3=NC=C(C(=N3)NC4=CC(=CC=C4)NC(=O)C=C)C(F)(F)F)OC
//////
Compound name AND SMILES string
Rociletinib COC(C=C(N1CCN(C(C)=O)CC1)C=C2)=C2NC3=NC=C(C(F)(F)F)C(NC4=CC=CC(NC(C=C)=O)=C4)=N3
Osimertinib CN(CCN(C)C)C(C(NC(C=C)=O)=C1)=CC(OC)=C1NC2=NC=CC(C3=CN(C)C4=C3C=CC=C4)=N2
EGF816 ClC1=C2C(N=C(NC(C3=CC(C)=NC=C3)=O)N2[C@H]4CN(C(/C=C/CN(C)C)=O)CCCC4)=CC=C1
PF-06747775 CN1C2=NC(N3C[C@@H](NC(C=C)=O)[C@H](F)C3)=NC(NC4=CN(C)N=C4OC)=C2N=C1
PF-06459988 CN(N=C1)C=C1NC2=NC3=C(C(Cl)=CN3)C(OC[C@H]4CN(C(C=C)=O)C[C@@H]4OC)=N2
WZ4002 ClC1=CN=C(NC2=C(OC)C=C(N3CCN(C)CC3)C=C2)N=C1OC4=CC=CC(NC(C=C)=O)=C4
EGF 816 , Nazartinib

EGF 816, Nazartinib
EGF-816; EGFRmut-TKI EGF816
Novartis Ag innovator
(R,E)-N-(7-chloro-1-(1-(4-(dimethylamino)but-2-enoyl)azepan-3-yl)-1H-benzo[d]imidazol-2-yl)-2-methylisonicotinamide
(R,E)-N-(7-chloro-l-(l-(4-(dimethylamino)but-2-enoyl)azepan-3-yl)-lH-benzo[d]imidazol-2 -yl)-2-methylisonicotinamide
NCI-H1975 (L858R/T790M): 25 nM
H3255 (L858R): 9 nM
HCC827 (Del ex19): 11 nM
| M.Wt | 495.02 | ||
|---|---|---|---|
| Formula | C26H31ClN6O2 | ||
| CAS No | 1508250-71-2 |
EGF816 is a novel covalent inhibitor of mutant-selective EGFR; overcomes T790M-mediated resistance in NSCLC.
Epidermal growth factor receptor antagonists; Protein tyrosine kinase inhibitors
- Phase IINon-small cell lung cancer
- Phase I/IISolid tumours
-
- 01 Feb 2015Phase-II clinical trials in Non-small cell lung cancer (Late-stage disease, Combination therapy) in Singapore (PO) (NCT02323126)
- 24 Nov 2014Phase-I/II clinical trials in Non-small cell lung cancer (Combination therapy, Late-stage disease) in Spain (PO) after November 2014 (EudraCT2014-000726-37)
- 24 Nov 2014Phase-I/II clinical trials in Non-small cell lung cancer (Combination therapy, Late-stage disease) in Germany (PO)
| Determine MTD, or recommended phase II dose in patients with NSCLC harboring EGFR mutations, in combination with INC280 | Recruiting Phase I/II (NCT02335944) |
| Determine MTD, or recommended phase II dose in adult patients with EGFRm+ solid malignancies | Recruiting Phase I/II (NCT02108964) |
| Determine efficacy and safety in patients with previously treated NSCLC, in combination with nivolumab | Recruiting Phase II (NCT02323126) |
In November 2015, FDA approved osimertinib (Tagrisso™) for the treatment of patients with metastatic EGFR T790M mutation-positive NSCLC, who have progressed on or after EGFR TKI therapy. Based on the clinical performance of the third generation EGFR drugs, more regulatory approvals can be expected.
Nazartinib, also known as EGF816, is an orally available, irreversible, third-generation, mutant-selective epidermal growth factor receptor (EGFR) inhibitor, with potential antineoplastic activity. EGF816 covalently binds to and inhibits the activity of mutant forms of EGFR, including the T790M EGFR mutant, thereby preventing EGFR-mediated signaling. This may both induce cell death and inhibit tumor growth in EGFR-overexpressing tumor cells. EGF816 preferentially inhibits mutated forms of EGFR including T790M, a secondarily acquired resistance mutation, and may have therapeutic benefits in tumors with T790M-mediated resistance when compared to other EGFR tyrosine kinase inhibitors
PATENT
WO 2016016822
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2016016822
PATENT
WO 2015081463
http://www.google.co.in/patents/WO2015081463A1?cl=en
PATENT
Intermediate 26
1055 (R)-tert-butyl 3-(2-amino-7-chloro- 1 H-benzo[dlimidazol- 1 -yOazepane- 1 -carboxylate

Step A: (R)-tert-butyl 3 -((2-chloro-6-nitrophenyl)amino)azepane-l -carboxylate (I-26a) was prepared following procedures analogous to 1-15, Step A, using the appropriate starting materials. JH-NMR (400MHz, CDC13): d 8.00-7.91 (m, 1H), 7.58-7.49 (m, 1H), 7.02-6.51
1060 (m, 2H), 4.31-4.03 (m, 1H), 3.84-2.98 (m, 4H), 1.98-1.60 (m, 5H), 1.46-1.39 (m, 10H); MS calculated for Ci7H25ClN304 (M+H+) 370.15, found 370.10.
Step B: A mixture of I-26a (7.5 g, 19.5 mmol) and Zn (12.8 mg, 195 mmol) in AcOH (22 mL) was stirred at room temperature for 2 h. The reaction was basified with saturated aqueous Na2C03 solution, filtered, and extracted with EtOAc (3 x 80 mL). The combined
1065 organic phase was washed with brine, dried with Na2S04 and concentrated in vacuo to afford (R)-tert-butyl 3-((2-amino-6-chlorophenyl)amino)azepane-l -carboxylate (I-26b). MS calculated for Ci7H27ClN302 (M+H+) 340.17, found 340.10. The crude was used in the next step without further purification.
Step C: The title compound (Intermediate 26) was prepared from I-26b following
1070 procedures analogous to 1-15, Step C. 1H-NMR (400MHz, CDC13): d Ί .34-126 (m, 1H),
7.04-6.97 (m, 2H), 6.05-5.85 (m, 1H), 5.84-5.72 (m, 1H), 5.50-5.37 (m, 0.5H), 5.10-4.80(m, 0.5H), 4.41-4.23(m, 1H), 4.09-3.96(m, 0.5H), 3.94-3.81 (m, 1H), 3.76-3.57 (m, 1H), 3.22-3.14 (m, 0.5H), 2.84-2.63 (m, 1H), 2.34-2.17 (m, 1H), 2.07-1.84 (m, 1H), 1.82-1.64 (m, 2H), 1.53 (s, 9H), 1.48-1.37 (m, 1H); MS calculated for C18H26CIN4O2 (M+H+) 365.17,
1075 found 365.10.
Intermediate 27
(R)-N-(l-(azepan-3-yl)-7-chloro-lH-benzo[dlimidazol-2-yl)-2-methylisonicotinamide hydrochloride

Intermediate 27
Step A
1080 Step A: A mixture of 2-methylisonicotinic acid (3.371 g, 24.6 mmol) and 2-(7-aza-lH- benzotriazole-l-yl)-l,l,3,3-tetramethyluronium hexafluorophosphate (9.345 g, 24.6 mmol) in CH2CI2 (120 ml) was treated at room temperature with NEt3 (4.1 mL, 29.4 mmol). The
reaction was stirred for 1 hour before it was slowly added into a CH2CI2 solution (45 ml) of 1-26 (5.98 g, 16.4 mmol). Ten minutes later, more NEt3 (4.1 mL, 29.4 mmol) was added and 1085 the mixture stirred for 2 h. The mixture was then diluted with CH2CI2 (240 mL), washed with H20 (2 x 80 mL), saturated aqueous NaHC03 solution (70 mL), and brine (70 mL). The organic phase was dried with Na2SC>4, and concentrated under reduced pressure. The crude material was purified by column chromatography (55% EtOAc/hexanes) to afford
(R)-tert-butyl
1090 3-(7-chloro-2-(2-methylisonicotinamido)-lH-benzo[d]imidazol-l-yl)azepane-l-carboxylate (I-27a) as a light yellow foam. 1H-NMR (400MHz, CDC13): d 12.81 (br s, 1H), 8.65-8.62 (m, 1H), 7.95-7.85 (m, 2H), 7.27-7.1 1 (m, 3H), 5.64 – 5.51 (m, 1H), 4.56-4.44 (m, 1H),
4.07-3.92 (m, 1H), 3.79-3.71 (m, 0.5H), 3.41-3.35 (m, 0.5H), 3.29-3.23 (m, 1H), 2.71-2.59 (m, 1H), 2.65 (s, 3H), 2.22-2.00 (m, 3H), 1.93-1.80 (m, 1H), 1.51-1.45 (m, 1H), 1.50 (s,
1095 3.5H), 1.41 (s, 5.5H); MS calculated for C25H3iClN503 (M+H+) 484.20, found 484.20.
Step B: A solution of I-27a (8.62 g, 16.4 mmol) in MeOH (67 mL) was treated with HC1 in dioxane (4M, 67 mL) and the mixture was stirred at room temperature for 7 h. The mixture was then concentrated under reduced pressure to afford the title compound (Intermediate 27). The product was used in the next step without further purification. A sample was treated
1 100 with 1M NaOH, extracted with EtOAc, dried with Na2SC>4 and concentrated under reduced pressure to afford 1-27 as a free base. 1H-NMR (400MHz, CD3CN): d 8.49 (d, J=5.0 Hz, 1H), 7.81 (s, 1H), 7.72 (d, J=4.8 Hz, 1H), 7.50 (br d, J=7.52 Hz, 1H), 7.16 – 7.09 (m, 2H), 5.66-5.59 (m, 1H), 3.77 (dd, J = 6.54, 14.3 Hz, 1H), 3.18 (dd, J = 5.3, 14.3 Hz, 1H), 3.05 – 2.98 (m, 1H), 2.76-2.69 (m, 1H), 2.63-2.53 (m, 1H), 2.47 (s, 3H), 2.10-2.03 (m, 1H),
1 105 1.96-1.93 (m, 2H), 1.86 – 1.75 (m, 2H), 1.61 – 1.54 (m, 2H); MS calculated for
C2oH23ClN50 (M+H+) 384.15, found 384.20.
(i?.E)-N-(7-chloro-l-(l-(4-(dimethylamino)but-2-enoyl)azepan-3-yl)-lH-benzo[dlimidazol-2
-yl)-2-methylisonicotinamide
1 1 10 
A mixture of (E)-4-(dimethylamino)but-2-enoic acid hydrochloride (58 mg, 0.35 mmol) and l-ethyl-3-(3-dimethylaminopropyl)carbodiimide hydrochloride (67 mg, 0.35 mmol) in DMF (2 mL) was treated with hydroxybenzotriazole (54 mg, 0.35 mmol) and stirred at room temperature for 1 h. The resulting mixture was added to a solution of 1-27 (100 mg, 0.22 1 1 15 mmol) in DMF (2 mL). Triethylamine (199 mg, 1.97 mmol) was then added and the mixture was stirred for 5 days. Water (2 mL) was added and the mixture was concentrated under
reduced pressure. The residue was diluted with IN NaOH (20 mL) and extracted with EtOAc (3 x 50 mL). The combined organic layers were washed with water (50 mL) and brine (2 x 50 mL), dried over Na2S04, and concentrated under reduced pressure. The crude was purified by
1 120 column chromatography (9: 1 :0.175N CH2Cl2/MeOH/NH3 in CH2C12, 0% to 100%) to afford the title compound. JH NM (400 MHz, DMSO-d6) δ 8.59 (d, J= 4.8 Hz, 1H), 7.89 (s, 1H), 7.79 (d, J = 4.8 Hz, 1H), 7.60 (d, J = 7.5 Hz, 1H), 7.30-7.22 (m, 2H), 6.71-6.65 (m, 1H), 6.57-6.54 (m, 1H), 5.54 (br. s, 1H), 4.54 (br. s, 1H), 4.20 (br s, 1H), 3.95 (br s, 1H), 3.48 (br s, 1H), 2.98 (br s, 2H), 2.72 (d, J = 12.0 Hz, 1H), 2.58 (s, 3H), 2.14 (br s, 6H), 2.05 (d, J =
1 125 6.7 Hz, 3H), 1.88 (br s, 1H), 1.46 (d, J=l 1.3 Hz, 1H); MS calculated for C26H32C1N602
(M+H+) 495.22, found 495.10. Melting point (1 14.6 °C).
WO 2015083059
https://www.google.com/patents/WO2015083059A1?cl=en
Intermediate 26
(RVtert-butyl 3-(2-amino-7-chloro-lH-benzo[dlimidazol-l-vf)azepane-l-carboxylate

Step A: (R)-tert- butyl 3-((2-chloro-6-nitrophenyl)amino)azepane-l-carboxylate (I-26a) was prepared following procedures analogous to 1-15, Step A, using the appropriate starting materials. 1H-NMR (400MHz, CDC13): d 8.00-7.91 (m, 1H), 7.58-7.49 (m, 1H), 7.02-6.51 (m, 2H), 4.31-4.03 (m, 1H), 3.84-2.98 (m, 4H), 1.98-1.60 (m, 5H), 1.46-1.39 (m, 10H); MS calculated for Ci7H25ClN304 (M+H+) 370.15, found 370.10.
Step B: A mixture of I-26a (7.5 g, 19.5 mmol) and Zn (12.8 mg, 195 mmol) in AcOH
(22 mL) was stirred at room temperature for 2 h. The reaction was basified with saturated aqueous Na2CC>3 solution, filtered, and extracted with EtOAc (3 x 80 mL). The combined organic phase was washed with brine, dried with Na2S04 and concentrated in vacuum to afford (R)-tert-butyl 3-((2-amino-6-chlorophenyl)amino)azepane-l-carboxylate (I-26b). MS calculated for C17H27CIN3O2 (M+H+) 340.17, found 340.10. The crude was used in the next step without further purification.
Step C: The title compound (Intermediate 26) was prepared from I-26b following procedures analogous to 1-15, Step C. ‘H-NMR (400MHZ, CDCI3): d 7.34-7.26 (m, 1H), 7.04-6.97 (m, 2H), 6.05-5.85 (m, 1H), 5.84-5.72 (m, 1H), 5.50-5.37 (m, 0.5H), 5.10-4.80(m, 0.5H), 4.41-4.23(m, 1H), 4.09-3.96(m, 0.5H), 3.94-3.81 (m, 1H), 3.76-3.57 (m, 1H), 3.22-3.14 (m, 0.5H), 2.84-2.63 (m, 1H), 2.34-2.17 (m, 1H), 2.07-1.84 (m, 1H), 1.82-1.64 (m, 2H), 1.53 (s, 9H), 1.48-1.37 (m, 1H); MS calculated for Ci8H26ClN402(M+H+) 365.17, found 365.10.
Intermediate 27
(R)-N-(l-(azepan-3-yl)-7-chloro-lH-benzo[dlimidazol-2-yl)-2-methylisonicotinamide hydrochloride

5-26 step A l~27a intermediate 27
Step A: A mixture of 2-methylisonicotinic acid (3.371 g, 24.6 mmol) and 2-(7-aza-lH-benzotriazole-l-yl)-l,l,3,3-tetramethyluronium hexafluorophosphate (9.345 g, 24.6 mmol) in CH2C12 (120 ml) was treated at room temperature with NEt3 (4.1 mL, 29.4 mmol). The reaction was stirred for 1 hour before it was slowly added into a CH2C12solution (45 ml) of 1-26 (5.98 g, 16.4 mmol). Ten minutes later, more NEt3 (4.1 mL, 29.4 mmol) was added and the mixture stirred for 2 h. The mixture was then diluted with CH2C12 (240 mL), washed with H20 (2 x 80 mL), saturated aqueous NaHCC solution (70 mL), and brine (70 mL). The organic phase was dried with Na2S04, and concentrated under reduced pressure. The crude material was purified by column chromatography (55% EtOAc/hexanes) to afford
(R)-tert-butyl
3-(7-chloro-2-(2-methylisonicotinamido)-lH-benzo[d]imidazol-l-yl)azepane-l-carboxylate (I-27a) as a light yellow foam. 1H-NMR (400MHz, CDCI3): d 12.81 (br s, 1H), 8.65-8.62 (m, 1H), 7.95-7.85 (m, 2H), 7.27-7.11 (m, 3H), 5.64 – 5.51 (m, 1H), 4.56-4.44 (m, 1H),
4.07-3.92 (m, 1H), 3.79-3.71 (m, 0.5H), 3.41-3.35 (m, 0.5H), 3.29-3.23 (m, 1H), 2.71-2.59 (m, 1H), 2.65 (s, 3H), 2.22-2.00 (m, 3H), 1.93-1.80 (m, 1H), 1.51-1.45 (m, 1H), 1.50 (s, 3.5H), 1.41 (s, 5.5H); MS calculated for C25H3iClN503 (M+H+) 484.20, found 484.20.
Step B: A solution of I-27a (8.62 g, 16.4 mmol) in MeOH (67 mL) was treated with HCI in dioxane (4M, 67 mL) and the mixture was stirred at room temperature for 7 h. The mixture was then concentrated under reduced pressure to afford the title compound (Intermediate 27). The product was used in the next step without further purification. A sample was treated with 1M NaOH, extracted with EtOAc, dried with Na2S04 and concentrated under reduced pressure to afford 1-27 as a free base. ‘H-NMR (400MHZ, CD3CN): d 8.49 (d, J=5.0 Hz, 1H), 7.81 (s, 1H), 7.72 (d, J=4.8 Hz, 1H), 7.50 (br d, J=7.52 Hz, 1H), 7.16 – 7.09 (m, 2H), 5.66-5.59 (m, 1H), 3.77 (dd, J = 6.54, 14.3 Hz, 1H), 3.18 (dd, J = 5.3, 14.3 Hz, 1H), 3.05 -2.98 (m, 1H), 2.76-2.69 (m, 1H), 2.63-2.53 (m, 1H), 2.47 (s, 3H), 2.10-2.03 (m, 1H), 1.96-1.93 (m, 2H), 1.86 – 1.75 (m, 2H), 1.61 – 1.54 (m, 2H); MS calculated for
C20H23CIN5O (M+H+) 384.15, found 384.20.
(i?,£,)-N-(7-chloro-l-(l-(4-(dimethylamino)but-2-enoyl)azepan-3-yl)-lH-benzo[dlimidazol-2
-νΠ-2-methylisonicotinamide

A mixture of (E)-4-(dimethylamino)but-2-enoic acid hydrochloride (58 mg, 0.35 mmol) and l -ethyl-3-(3-dimethylaminopropyl)carbodiimide hydrochloride (67 mg, 0.35 mmol) in DMF (2 mL) was treated with hydroxybenzotriazole (54 mg, 0.35 mmol) and stirred at room temperature for 1 h. The resulting mixture was added to a solution of 1-27 (100 mg, 0.22 mmol) in DMF (2 mL). Triethylamine (199 mg, 1.97 mmol) was then added and the mixture was stirred for 5 days. Water (2 mL) was added and the mixture was concentrated under reduced pressure. The residue was diluted with IN NaOH (20 mL) and extracted with EtOAc (3 x 50 mL). The combined organic layers were washed with water (50 mL) and brine (2 x 50 mL), dried over Na2S04, and concentrated under reduced pressure. The crude was purified by column chromatography (9: 1 :0.175N CH2Cl2/MeOH/NH3 in CH2C12, 0% to 100%) to afford the title compound. 1H NMR (400 MHz, DMSO-d6) δ 8.59 (d, J = 4.8 Hz, 1H), 7.89 (s, 1H), 7.79 (d, J = 4.8 Hz, 1H), 7.60 (d, J = 7.5 Hz, 1H), 7.30-7.22 (m, 2H), 6.71-6.65 (m, 1H), 6.57-6.54 (m, 1H), 5.54 (br. s, 1H), 4.54 (br. s, 1H), 4.20 (br s, 1H), 3.95 (br s, 1H), 3.48 (br s, 1H), 2.98 (br s, 2H), 2.72 (d, J = 12.0 Hz, 1H), 2.58 (s, 3H), 2.14 (br s, 6H), 2.05 (d, J = 6.7 Hz, 3H), 1.88 (br s, 1H), 1.46 (d, J=11.3 Hz, 1H); MS calculated for C26H32C1N602 (M+H+) 495.22, found 495.10. Melting point (114.6 °C).
PATENT
WO 2015112705
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2015112705
PATENT
WO 2013184757
https://www.google.com/patents/WO2013184757A1?cl=en
Intermediate 26
(R)-tert-butyl 3 -(2-amino-7-chloro- 1 H-benzo Tdlimidazol- 1 – vDazepane- 1 – carboxylate
Intermediate 26
Step A: (R)-tert-butyl 3-((2-chloro-6-nitrophenyl)amino)azepane-l-carboxylate (I- 26a) was prepared following procedures analogous to 1-15, Step A, using the appropriate starting materials. 1 H-NMR (400MHz, CDC13): d 8.00-7.91 (m, 1H), 7.58-7.49 (m, 1H), 7.02-6.51 (m, 2H), 4.31-4.03 (m, 1H), 3.84-2.98 (m, 4H), 1.98-1.60 (m, 5H), 1.46-1.39 (m, 10H); MS calculated for C17H25CIN3O4 (M+H+) 370.15, found 370.10. Step B: A mixture of I-26a (7.5 g, 19.5 mmol) and Zn (12.8 mg, 195 mmol) in AcOH (22 mL) was stirred at room temperature for 2 h. The reaction was basified with saturated aqueous Na2CC>3 solution, filtered, and extracted with EtOAc (3 x 80 mL). The combined organic phase was washed with brine, dried with Na2S04 and concentrated in vacuo to afford (R)-tert-butyl 3-((2-amino-6-chlorophenyl)amino)azepane-l-carboxylate (I-26b). MS calculated for Ci7H27ClN302 (M+H+) 340.17, found 340.10. The crude was used in the next step without further purification.
Step C: The title compound (Intermediate 26) was prepared from I-26b following procedures analogous to 1-15, Step C. ]H-NMR (400MHz, CDC13): d 7. ,34-7.26 (m, 1H), 7.04-6.97 (m, 2H), 6.05-5.85 (m, 1H), 5.84-5.72 (m, 1H), 5.50-5.37 (m, 0.5H), 5.10- 4.80(m, 0.5H), 4.41-4.23(m, 1H), 4.09-3.96(m, 0.5H), 3.94-3.81 (m, 1H), 3.76-3.57 (m, 1H), 3.22-3.14 (m, 0.5H), 2.84-2.63 (m, 1H), 2.34-2.17 (m, 1H), 2.07-1.84 (m, 1H), 1.82- 1.64 (m, 2H), 1.53 (s, 9H), 1.48-1.37 (m, 1H); MS calculated for Ci8H26ClN402 (M+H+) 365.17, found 365.10.
Intermediate 27
(R)-N-(l-(azepan-3-yl)-7-chloro-lH-benzordlimidazol-2-yl)-2-methylisonicotinamide hydrochloride
l-27a Intermediate 27
Step A: A mixture of 2-methylisonicotinic acid (3.371 g, 24.6 mmol) and 2-(7-aza- 1H- benzotriazole-l-yl)-l,l,3,3-tetramethyluronium hexafluorophosphate (9.345 g, 24.6 mmol) in CH2C12 (120 ml) was treated at room temperature with NEt3 (4.1 mL, 29.4 mmol). The reaction was stirred for 1 hour before it was slowly added into a CH2C12 solution (45 ml) of 1-26 (5.98 g, 16.4 mmol). Ten minutes later, more NEt3 (4.1 mL, 29.4 mmol) was added and the mixture stirred for 2 h. The mixture was then diluted with CH2C12 (240 mL), washed with H20 (2 x 80 mL), saturated aqueous NaHC03 solution (70 mL), and brine (70 mL). The organic phase was dried with Na2S04, and concentrated under reduced pressure. The crude material was purified by column chromatography (55% EtOAc/hexanes) to afford (R)-tert-butyl 3-(7-chloro-2-(2-methylisonicotinamido)- lH-benzo[d]imidazol-l-yl)azepane-l-carboxylate (I-27a) as a light yellow foam. ]H- NMR (400MHz, CDC13): d 12.81 (br s, IH), 8.65-8.62 (m, IH), 7.95-7.85 (m, 2H), 7.27- 7.11 (m, 3H), 5.64 – 5.51 (m, IH), 4.56-4.44 (m, IH), 4.07-3.92 (m, IH), 3.79-3.71 (m, 0.5H), 3.41-3.35 (m, 0.5H), 3.29-3.23 (m, IH), 2.71-2.59 (m, IH), 2.65 (s, 3H), 2.22-2.00 (m, 3H), 1.93-1.80 (m, IH), 1.51-1.45 (m, IH), 1.50 (s, 3.5H), 1.41 (s, 5.5H); MS calculated for C25H31CIN5O3 (M+H+) 484.20, found 484.20.
Step B: A solution of I-27a (8.62 g, 16.4 mmol) in MeOH (67 mL) was treated with HCl in dioxane (4M, 67 mL) and the mixture was stirred at room temperature for 7 h. The mixture was then concentrated under reduced pressure to afford the title compound
(Intermediate 27). The product was used in the next step without further purification. A sample was treated with 1M NaOH, extracted with EtOAc, dried with Na2S04 and concentrated under reduced pressure to afford 1-27 as a free base. ]H-NMR (400MHz, CD3CN): d 8.49 (d, J=5.0 Hz, IH), 7.81 (s, IH), 7.72 (d, J=4.8 Hz, IH), 7.50 (br d, J=7.52 Hz, IH), 7.16 – 7.09 (m, 2H), 5.66-5.59 (m, IH), 3.77 (dd, J = 6.54, 14.3 Hz, IH), 3.18 (dd, J = 5.3, 14.3 Hz, IH), 3.05 – 2.98 (m, IH), 2.76-2.69 (m, IH), 2.63-2.53 (m, IH), 2.47 (s, 3H), 2.10-2.03 (m, IH), 1.96-1.93 (m, 2H), 1.86 – 1.75 (m, 2H), 1.61 – 1.54 (m, 2H); MS calculated for C20H23CIN5O (M+H+) 384.15, found 384.20.
Example 5
(/?,£,)-N-(7-chloro-l-(l-(4-(dimethylamino)but-2-enoyl)azepan-3-yl)- lH- benzordlimidazol-2-yl)-2-methylisonicotinamide
A mixture of (E)-4-(dimethylamino)but-2-enoic acid hydrochloride (58 mg, 0.35 mmol) and l-ethyl-3-(3-dimethylaminopropyl)carbodiimide hydrochloride (67 mg, 0.35 mmol) in DMF (2 mL) was treated with hydroxybenzotriazole (54 mg, 0.35 mmol) and stirred at room temperature for 1 h. The resulting mixture was added to a solution of 1-27 (100 mg, 0.22 mmol) in DMF (2 mL). Triethylamine (199 mg, 1.97 mmol) was then added and the mixture was stirred for 5 days. Water (2 mL) was added and the mixture was concentrated under reduced pressure. The residue was diluted with IN NaOH (20 mL) and extracted with EtOAc (3 x 50 mL). The combined organic layers were washed with water (50 mL) and brine (2 x 50 mL), dried over Na2SC>4, and concentrated under reduced pressure. The crude was purified by column chromatography (9: 1 :0.175N CH2Cl2/MeOH/NH3 in CH2C12, 0% to 100%) to afford the title compound (Example 5). ]H NMR (400 MHz, DMSO-d6) δ 8.59 (d, J = 4.8 Hz, IH), 7.89 (s, IH), 7.79 (d, J = 4.8 Hz, IH), 7.60 (d, / = 7.5 Hz, IH), 7.30-7.22 (m, 2H), 6.71-6.65 (m, IH), 6.57-6.54 (m, IH), 5.54 (br. s, IH), 4.54 (br. s, IH), 4.20 (br s, IH), 3.95 (br s, IH), 3.48 (br s, IH), 2.98 (br s, 2H), 2.72 (d, / = 12.0 Hz, IH), 2.58 (s, 3H), 2.14 (br s, 6H), 2.05 (d, / = 6.7 Hz, 3H), 1.88 (br s, IH), 1.46 (d, 7=11.3 Hz, IH); MS calculated for C26H32CIN6O2 (M+H+) 495.22, found 495.10. Melting point (114.6 °C).
(/?,E)-N-(7-chloro- l-(l-(4-(dimethylamino)but-2-enoyl)azepan-3-yl)-lH- benzo[d]imidazol-2-yl)-2-methylisonicotinamide (1.0 g) was dissolved in acetone (30 mL) by heating to 55°C to form a solution. Methanesulfonic acid (325 μί) was added to acetone (50 mL), and the methanesulfonic acid/acetone (22.2 mL) was added to the solution at 0.05ml/min. Following precipitation, the resulting suspension was cooled to room temperature at 0.5 °C/min, and crystals were collected by filtration, and dried for 4 hours at 40°C under vacuum. The collected crystals (300 mg) were suspended in acetone/H20 (6 mL; v/v=95/5) by heating to 50°C. The suspension was kept slurrying for 16 hours, and cooled to room temperature at 0.5 °C/min. The crystal was collected by filtration and dried for 4 hours at 40°C under vacuum.
The structure of (7?,£‘)-N-(7-chloro-l-(l-(4-(dimethylamino)but-2-enoyl)azepan-3-yl)- lH-benzo[d]imidazol-2-yl)-2-methylisonicotinamide mesylate was confirmed by Differential Scanning Calorimetry, X-Ray Powder Diffraction, and Elemental Analyses. Melting point (170.1 °C). Theoretical calculated: C (54.8); H (5.9); N (14.2); 0 (13.5); %S (5.4); and C1 (6.0); C:N ratio: 3.86. Found: C (52.0); H (5.8); N (13.3); C1 (5.9); C:N ratio: 3.91. Stoichiometry: 1.01.
References
AACR Annual Meeting 2014; April 5-9, 2014; San Diego, CA.
nmr http://www.medchemexpress.com/product_pdf/HY-12872/EGF816-NMR-HY-12872-17795-2015.pdf
/////EGF 816, EGF816, EGFR, Covalent inhibitor, T790M, Oncogenic mutation, Lung cancer, NSCLC, SBDD, Drug resistance, EGF-816, EGFRmut-TKI EGF816, Nazartinib
O=C(NC1=NC2=CC=CC(Cl)=C2N1[C@H]3CN(C(/C=C/CN(C)C)=O)CCCC3)C4=CC=NC(C)=C4
HELP, Need one time help to pay 10 year concessional subscription to this, your favorite blog to WordPress

Just One viewer please come forward
Dear Kind Viewer’s
WordPress is kind to me and negotiated a one time 10 year concessional subscription of 260 US dollars…….https://newdrugapprovals.org/
I need one time help to pay this one time 10 year concessional subscription to our favorite blog.
This is done to keep this blog running even after my death.
Currently I am paying 99 US Dollars per annum
email me
amcrasto@gmail.com
call +919323115463
Paypal will work for me via email request to you by me, Indian govt does not allow automatic transfer via paypal buttons on the blog
email me at amcrasto@gmail.com and tell me amount, i will request you via paypal

DR ANTHONY CRASTO
LIONEL MY SON, MY MOTIVATION
.
He was only in first standard in school when I was hit by a deadly one in a million spine stroke called acute transverse mylitis, it made me 90% paralysed and bound to a wheel chair, He cried bitterly and we had never seen him so depressed
Now I keep Lionel as my source of inspiration and helping millions, thanks to millions of my readers who keep me going and help me to keep my son and family happy.
ps
The views expressed are my personal and in no-way suggest the views of the professional body or the company that I represent,
///////////
LAMIVUDINE
Lamivudine (2′,3′-dideoxy-3′-thiacytidine, commonly called 3TC) is an antiretroviral medication used to prevent and treat HIV/AIDS and used to treat chronic hepatitis B.[1]
It is of the nucleoside analog reverse transcriptase inhibitor (NRTI) class. It is marketed in the United States under the tradenames Epivir and Epivir-HBV.
It is on the World Health Organization’s List of Essential Medicines, a list of the most important medication needed in a basic health system.[2] As of 2015 the cost for a typical month of medication in the United States is more than 200 USD.[3]
Medical uses
Lamivudine has been used for treatment of chronic hepatitis B at a lower dose than for treatment of HIV/AIDS. It improves the seroconversion of e-antigen positive hepatitis B and also improves histology staging of the liver. Long term use of lamivudine leads to emergence of a resistant hepatitis B virus (YMDD) mutant. Despite this, lamivudine is still used widely as it is well tolerated.
Resistance
In HIV, high level resistance is associated with the M184V/I mutation in the reverse transcriptase gene as reported by Raymond Schinazi’s group at Emory University. GlaxoSmithKline claimed that the M184V mutation reduces “viral fitness”, because of the finding that continued lamivudine treatment causes the HIV viral load to rebound but at a much lower level, and that withdrawal of lamivudine results in a higher viral load rebound with rapid loss of the M184V mutation; GSK therefore argued that there may be benefit in continuing lamivudine treatment even in the presence of high level resistance, because the resistant virus is “less fit”. The COLATE study has suggested that there is no benefit to continuing lamivudine treatment in patients with lamivudine resistance.[4] A better explanation of the data is that lamivudine continues to have a partial anti-viral effect even in the presence of the M184V mutation.
In hepatitis B, lamivudine resistance was first described in the YMDD (tyrosine–methionine–aspartate-aspartate) locus of the HBV reverse transcriptase gene. The HBV reverse transcriptase gene is 344 amino acids long and occupies codons 349 to 692 on the viral genome. The most commonly encountered resistance mutations are M204V/I/S.[5] The change in amino acid sequence from YMDD to YIDD results in a 3.2 fold reduction in the error rate of the reverse transcriptase, which correlates with a significant growth disadvantage of the virus. Other resistance mutations are L80V/I, V173L and L180M.[6]
Mechanism of action
Lamivudine is an analogue of cytidine. It can inhibit both types (1 and 2) of HIV reverse transcriptase and also the reverse transcriptase of hepatitis B virus. It is phosphorylated to active metabolites that compete for incorporation into viral DNA. They inhibit the HIV reverse transcriptase enzyme competitively and act as a chain terminator of DNA synthesis. The lack of a 3′-OH group in the incorporated nucleoside analogue prevents the formation of the 5′ to 3′ phosphodiester linkage essential for DNA chain elongation, and therefore, the viral DNA growth is terminated.
Lamivudine is administered orally, and it is rapidly absorbed with a bio-availability of over 80%. Some research suggests that lamivudine can cross the blood–brain barrier. Lamivudine is often given in combination with zidovudine, with which it is highly synergistic. Lamivudine treatment has been shown to restore zidovudine sensitivity of previously resistant HIV. Lamivudine showed no evidence of carcinogenicity or mutagenicity in in vivo studies in mice and rats at doses from 10 to 58 times those used in humans.[7]
History
Racemic BCH-189 (the minus form is known as Lamivudine) was invented by Dr. Bernard Belleau while at work at McGill University and Dr Paul Nguyen-Ba at the Montreal-based IAF BioChem International, Inc. laboratories in 1988 and the minus enantiomer isolated in 1989. Samples were first sent to Dr. Yung-Chi Cheng of Yale University for study of its toxicity. When used in combination with AZT, he discovered that Lamivudine’s negative form reduced side effects and increased the drug’s efficiency at inhibiting reverse transcriptase.[8] The combination of Lamivudine and AZT increased the efficiency at inhibiting an enzyme HIV uses to reproduce its genetic material. As a result, Lamivudine was identified as a less toxic agent to mitochondria DNA than other retroviral drugs.[9]
Lamivudine was approved by the Food and Drug Administration (FDA) on November 17, 1995 for use with zidovudine (AZT) and again in 2002 as a once-a-day dosed medication. The fifth antiretroviral drug on the market, it was the last NRTI for three years while the approval process switched to protease inhibitors. According to the manufacturer’s 2004 annual report, its patent will expire in the United States in 2010 and in Europe in 2011.
On September 2014, Dr. Gorbee Logan, a Liberian physician, reported positive results while treating Ebola virus disease with Lamivudine. Out of 15 patients treated with the antiviral, 13 (those treated within the third to fifth day of symptoms being manifested) survived the disease and were declared virus-free; the remaining cases (treated from the fifth day or later) died.[10][11]
Presentation
- Epivir 150 mg or 300 mg tablets (GlaxoSmithKline; US and UK) for the treatment of HIV;
- Epivir-HBV 100 mg tablets (GlaxoSmithKline; US only) for the treatment of hepatitis B;
- Zeffix 100 mg tablets (GlaxoSmithKline; UK only) for the treatment of hepatitis B.
- 3TC 150 mg tablets (GlaxoSmithKline; South Africa) for the treatment of HIV;
Lamivudine is also available in fixed combinations with other HIV drugs:
- Combivir (with zidovudine);
- Epzicom/Kivexa (with abacavir);
- Trizivir (with zidovudine and abacavir)
Lamivudine (I) (CAS No. 134678-17-4) is chemically known as (-)-[2R,5S]-4T amino- 1 – [2-(hydroxymethyl)- 1 ,3 -oxathiolan-5-yl] -2( 1 H)-pyrimidin-2-one.
Formula (I)
Lamivudine is a reverse transcriptase inhibitor used alone or in combination with other classes of Anti-HIV drugs in the treatment of HIV infection. It is available commercially as a pharmaceutical composition under the brand name EPIVIR®, marketed by GlaxoSmithKline, and is covered under US 5,047,407.
This molecule has two stereo-centres, thus giving rise to four stereoisomers: (±)- Cis Lamivudine and (±)-Trans Lamivudine. The pharmaceutically active isomer however is the (-)-Cis isomer which has the absolute configuration [2R,5S] as show in Formula (I).
US 5,047,407 discloses the 1,3-oxathiolane derivatives; their geometric (cis/trans) and optical isomers. This patent describes the preparation of Lamivudine as a mixture of cis and trans isomers (shown in scheme I). The diastereomers obtained are converted into N-acetyl derivatives before separation by column chromatography using ethylacetate and methanol (99:1); however, this patent remains silent about further resolution of the cis isomer to the desired (-)- [2R,5S]-Cis-Lamivudine. Secondly, as the ethoxy group is a poor leaving group, the condensation of cytosine with compound VI gives a poor yield, i.e. 30 – 40%, of compound VII. Thirdly, chromatographic separation that has been achieved only after acetylation requires a further step of de-acetylation of the cis-(±)- isomer. Also, separation of large volumes of a compound by column chromatography makes the process undesirable on a commercial scale.
(+/-) Cis (+/-) Cis Lamivudine (VIII)
Scheme – 1 Efforts have been made in the past to overcome the shortcomings of low yield and enantiomeric enrichment, hi general, there have been two approaches to synthesize (— )-[2R,5S]-Cis-Lamivudine. One approach involves stereoselective synthesis, some examples of which are discussed below.
US 5,248,776 describes an asymmetric process for the synthesis of enantiomerically pure β-L-(-)-l,3-oxathiolone-nucleosides starting from optically pure 1,6-thioanhydro-L-gulose, which in turn can be easily prepared from L- Gulose. The condensation of the 1,3-oxathiolane derivative with the heterocyclic base is carried out in the presence of a Lewis acid, most preferably SnCl4, to give the [2R,5R] and [2R,5S] diastereomers that are then separated chromatographically.
US 5,756,706 relates a process where compound A is esterified and reduced to compound B. The hydroxy group is then converted to a leaving group (like acetyl) and the cis- and trans-2R-tetrahydrofuran derivatives are treated with a pyrimidine base, like N-acetylcytosine, in the presence trimethylsilyl triflate to give compound C in the diastereomeric ratio 4: 1 of cis and trans isomers.
A B C
Z = S5 CH
Dissolving compound C in a mixture of 3:7 ethyl acetate-hexane separates the cis isomer. The product containing predominantly the cis-2R,5S isomer and some trans-2R,5R compound is reduced with NaBH4 and subjected to column chromatography (30% MeOH-EtOAc) to yield the below compound.
US 6,175,008 describes the preparation of Lamivudine by reacting mercaptoacetaldehyde dimer with glyoxalate and further with silylated pyrimidine base to give mainly the cis-isomer by using an appropriate Lewis acid, like TMS-
I5 TMS-Tf, TiCl4 et cetera. However the stereoselectivity is not absolute and although the cis isomer is obtained in excess, this process still requires its separation from the trans isomer. The separation of the diastereomers Js done by acetylation and chromatographic separation followed by deacetylation. Further separation of the enantiomers of the cis-isomer is not mentioned.
US 6,939,965 discloses the glycosylation of 5-fluoro-cytosine with compound F (configuration: 2R and 2S)
. F
The glycosylation is carried out in the presence of TiCl3(OiPr) which is stereoselective and the cis-2R,5S-isomer is obtained in excess over the trans- 2S,5S-isomer. These diastereomers are then separated by fractional crystallization.
US 6,600,044 relates a method for converting the undesired trans-l,3-oxathiolane nucleoside to the desired cis isomer by a method of anomerizatioή or transglycosylation and the separation of the hydroxy-protected form of cis-, trans- (-)-nucleosides by fractional crystallization of their hydrochloride, hydrobromide, methanesulfonate salts. However, these cis-trans isomers already bear the [R] configuration at C2 and only differ in their configuration at C5; i.e. the isomers are [2R,5R] and [2R,5S]. Hence diastereomeric separation directly yields the desired [2R, 5S] enantiomer of Lamivudine.
In the second approach to prepare enantiomerically pure Lamivudine the resolution of racemic mixtures of nucleosides is carried out. US 5,728,575 provides one such method by using enzyme-mediated enantioselective hydrolysis of esters of the formula
wherein, ‘R’ is an acyl group and ‘Rl ‘ represents the purine or pyrimidine base.
‘R’ may be alkyl carboxylic, substituted alkyl carboxylic and preferably an acyl group that is significantly electron-withdrawing, eg. α-haloesters. After selective hydrolysis, the process involves further separation of the unhydrolyzed ester from the‘ enantiomerically pure 1,3-oxathiolane-nucleoside. Three methods are suggested in this patent, which are:
1. Separation of the more lipophilic unhydrolyzed ester by solvent extraction with one of a wide variety of nonpolar organic solvents.
2. Lyophilization followed by extraction into MeOH or EtOH. 3. Using an HPLC column designed for chiral separations.
In another of its aspects, this patent also refers to the use of the enzyme cytidine- deoxycytidine deaminase, which is enantiomer-specific, Λo catalyze the deamination of the cytosine moiety and thereby converting it to uridine. Thus, the enantiomer that remains unreacted is still basic and can be extracted by using an acidic solution.
However, the above methods suffer from the following drawbacks, (a) Enzymatic hydrolysis sets down limitations on choice of solvents: alcohol solvents cannot be used as they denature enzymes. (b) Lyophilization on an industrial scale is tedious, (c) Chiral column chromatographic separations are expensive.
WO 2006/096954 describes the separation of protected or unprotected enantiomers of the cis nucleosides of below formula by using a chiral acid to form diastereomeric salts that are isolated by filtration. Some of the acids used are R-
(-)-Camphorsulfonic acid, L-(-)-Tartaric acid, L-(-)-Malic acid, et cetera.
However, the configuration of these CIS-nucleosides are [2R,4R] and [2S,4S] as the heterocyclic base is attached at the 4 position of the oxathiolane ring and the overall stereo-structure of the molecule changes from that of the 2,5-substituted oxathiolane ring.
Thus various methods are described for the preparation of Lamivudine. However there is no mention in the prior art about the separation of an enantiomeric pair, either cis-(±) or trans-(±), from a mixture containing cis-[2R,5S], [2S,5R] and trans-[2R,5R], [2S,5S] isomers. Further, there also is a need to provide resolution of the cis-(±) isomers to yield the desired enantiomer in high optical purity.
CN 1223262 (Deng et aϊ) teaches the resolution of a certain class of compounds called Prazoles by using chiral host compounds such as dinaphthalenephenols (BINOL), diphenanthrenols or tartaric acid derivatives. The method consists of the formation of a 1:1 complex between the chiral host (BINOL) and one of the enantiomers, the guest molecule. The other enantiomer remains in solution. (S)- Omeprazole, which is pharmaceutically active as a highly potent inhibitor of gastric acid secretion, has been isolated from its racemic mixture in this manner by using S-BINOL.
BINOL is a versatile chiral ligand that has found its uses in various reactions involving asymmetric synthesis (Noyori, R. Asymmetric Catalysis in Organic
Synthesis) and optical resolution (Cram, D. J. et al J. Org. Chem. 1977, 42, 4173-
4184). Some of these reactions include BINOL-mediated oxidation and reduction reactions, C-C bond formation reactions such as Aldol reaction, Michael addition,
Mannich reaction et cetera (Brunei Chem. Rev. 2005 105, 857-897) and kinetic resolution, resolution by inclusion complexation et cetera.
BINOL, or l,l’-bi-2-Naphthol, being an atropoisomer possesses the property of chiral recognition towards appropriate compounds. One of the uses of BINOL in resolution that is known in literature is in Host-Guest complexation. In one such example, 1,1-binaphthyl derivatives have been successfully incorporated into optically active crown ethers for the enantioselective complexation of amino acid esters and chiral primary ammonium ions (Cram, D. J. Ace. Chem. Res. 1978, 11, 8-14). The chiral ‘host’ is thus able to discriminate between enantiomeric compounds by the formation of hydrogen bonds between the ether oxygen and the enantiomers. The complex formed with one of the isomers, the ‘guest’, will be less stable on steric grounds and this forms the basis for its separation.
It is evident from the literature cited that there exists a need to (a) synthesize Lamivudine by a process requiring less expensive, less hazardous and easily available reagents, and (b) achieve good yields with superior quality of product without resorting to column chromatography as a means of separation, thereby making the process of Lamivudine manufacture more acceptable industrially.
CLIP
ideally, the chemical synthesis of APIs begins from simple, inexpensive building blocks or RMs that are used for multiple purposes and are available in the fine chemicals industry, though some require uncommon RMs that contribute significantly to API manufacturing cost. RMs are converted into APIs by multi-step processes of breaking old chemical bonds and making new ones. A synthesis of 3TC is shown in . In the seven-step sequence, six steps involve breaking existing chemical bonds and creating new ones to build the molecular architecture of the API. The final recrystallization of an API is a critical step; at this stage the crystalline form of the API is determined and related substances (impurities) are removed or reduced to acceptable levels. APIs are often milled in a final step so that their particle size distribution (PSD) falls within specified limits. The crystalline form and PSD of an API must be controlled, because these properties are often critical to the formulation, dissolution, absorption and bioavailability of a drug. Bioavailability is the fraction of a drug dose that reaches systemic circulation (that is, is present in blood plasma) after administration. By definition, a drug is 100% bioavailable when administered by injection; drugs for ART are taken every day and administration by injection is not possible.
The cost of ART is absolutely critical to ensuring access in LMICs. The cost of manufacturing an API is dependent upon the cost of RMs, the cost of overheads and labour (OHL) and volume demand for the product. OHL includes the capital investment to build a manufacturing facility and operating costs, including personnel and energy, waste disposal and the eventual cost of decommissioning of the facility. Increased volume demand generally decreases the cost contribution of RM and OHL. Substantial production volumes are required to obtain full economy of scale . Producing 1–5 metric tons per year is substantially more expensive per kilogram than producing 100 metric tons of an API. There is a practical limit of approximately 50–100 metric tons/year beyond which cost reductions are modest with increased volume, but this practical limit refers to the volumes of drug manufactured in any single manufacturing plant. Exceptions to these generalizations do occur, most often when demand exceeds either the existing manufacturing capacity for a specific API or the availability of critical RMs . Exceptions that have occurred include shortages of β-thymidine for producing AZT and a squeeze on the availability and price of adenine as a starting material for TDF. Another contributor to RM and OHL costs is the efficiency of a chemical synthesis. Since operating costs for a manufacturing facility may be USD2,000/h, the number of steps or processing time for a chemical synthesis affects manufacturing cost. The efficiency of a synthesis is often quoted as an E-factor representing the kilograms of waste produced per kilogram of product manufactured. Waste management is expensive in chemical manufacturing wherever environmental guidelines are both reasonable and followed. From a slightly different perspective, increasing the overall yield of an API synthesis reduces RM use and associated cost for manufacturing.
Jinliang L, Feng LV. inventors; Shanghai Desano Pharmaceutical, assignee. A process for stereoselective synthesis of lamivudine. European Patent Application EP 2161 267 A1. 2007 June 29.

Object of the invention
Thus, one object of the present invention is to provide a process for the synthesis of_Lamivudine which is cost effective, uses less hazardous and easily available reagents, yet achieves good yields with superior quality of product without resorting to column chromatography.
A further object of the present invention is to provide an improved process for the synthesis of Lamivudine, by separating the mixture of diastereomers: Cis-[2R,5S], [2S,5R] from Trans-[2R,5R], [2S,5S] and then resolving the Cis isomers using BINOL to obtain (-)-[2R,5S]^Cis-Lamivudine with at least 99% ee.
This 1,3-oxathiolane compound VIII is further condensed with silylated cytosine in the presence of a Lewis acid such as trimethylsilyliodide to get protected 6-amino-3 – {2-hydroxymethyl- 1 ,3 -oxathiolan-5-yl} -3 -hydropyrimidine- 2-one (compound IX). OH
Cis(±)and Trans (±) racemic mixtures
Lamivudine (-)-[2
Compound (IX) is mixture of following optical isomers
SCHEME 2 The separation of the four-component diastereomeric mixture of isomers bearing the following configuration: trans-[2R,5R], [2S.5S] and cis-[2R,5S], [2S,5R] forms the next step. The separation efficiency of the benzoyl-protected compound
Example 9
Preparation of Lamivudine: (-)-[2R,5S]-4-amino-l-[2-(hydroxymethyl)-l,3- oxathiolan-5 -yl] -2(1 H)-pyrimidin-2-one
Compound I 5mL of cone. HCl was slowly added to a solution of 2Og of Lamivudine-BINOL complex in 100ml of ethylacetate and 10OmL of DM water (pH 2-2.5). The layers. were separated and a 10OmL aliquot of ethylacetate was added to the aqueous layer. The layers were separated again and the aqueous layer was neutralized using 1OmL of 10% aqueous NaOH solution. The solvent was recovered under vacuum at 40-45 0C, the product obtained was dissolved in 160 mL of methanol, filtered, the filtrate was concentrated and 32 mL of water-ethanol mixture (3:1) was added to this product, heated to get a clear solution, cooled to 5 – 10 0C and then filtered. The residue was vacuum dried at 45-50 0C. Yield: 4-5g.
Enantiomeric excess = 99.74 % m.p. = 133-135 °C [<X]D at 25°C = 98.32° (c = 5 water)
1H NMR (DMSO d6): 2.99-3.07 (dd, IH), 3.35-3.38 (dd, IH), 3.72-3.74 (m, 2H), 5.14-5.18 (t, IH), 5.32-5.38 (t, IH), 5.71-5.75 (d, IH), 6.16-6.21 (t, IH), 7.22-
7.27 (d, 2H), 7.80-7.83 (d, IH)
Moisture content: 1.67%
IR (in KBr, cm“1): 3551, 3236, 2927, 1614, 1492, 1404, 1336, 1253, 1146, 1052,
967, 786. MS: M+l =230
XRD [2Θ] (Cu – Ka1=I.54060A, Ka2=1.54443A Kβ= 1.39225A; 4OmA, 45kV):
5.08, 9.89, 10.16, 11.40, 11.65, 12.96, 13.23, 15.26, 15.82, 17.74, 18.74, 18.88,
19.67, 20.69, 22.13, 22.88, 23.71, 25.47, 26.07.
PATENT
http://www.google.com/patents/WO2013021290A1?cl=en



| EP 0382526; EP 0711771; JP 1996119967; JP 2000143662; US 5047407 |

| J Org Chem 1992,57(8),2217-9 |

Lamivudine is a nucleoside reverse transcriptase inhibitor, and is a kind of deoxycytidine analogue, which can inhibit the reproduction of Human immunodeficiency virus (HIV) and hepatitis B virus (HBV), whose chemical name is (2R-cis)-4-amino-1-(2-hydroxymethyl-1,3-oxathiolan-5-yl)-1H-pyrimidin-2-one, and structural formula is as follows:
-
In 1990, Belleau et al firstly reported Lamivudine structure, and BioChem Pharma of Canada firstly developed Lamivudine to be used to treat AIDS ( WO91/17159 ) and hepatitis B ( EP0474119 ), and found that it had distinguished therapeutic effect on hepatitis B. Since Lamivudine has two chiral centers, it has 4 stereisomers, among which the 2R,5S (2R–cis)-isomer is the most potent in anti-HIV and anti-HBV activities, and its cytotoxicity on some cells is lower than its enatiomer or racemic body.
-
In the above two schemes of this process, chirality was not controlled, and the final product was obtained by column chromatography, thus the yield was low and the requirement on the equipment was high, resulting in that the production cost was high and the operation in the production could not be controlled easily.
The specific reaction scheme is as follows:
synthetic route is preferably as follows:
. The specific reaction scheme is as follows:
The specific reaction scheme is as follows:
Example 8 The preparation of (2R,5S)-4-amino-1-(2-hydroxymethyl-1,3-oxathiolane-5-yl) -2(1H)-pyrimidone (Lamivudine)
-
The compound of Example 7 (41.0g, 0.1mol) and methanol (250ml) were added to a reaction flask, and then stirred to make the compound dissolved in methanol. The mixture was cooled to 0 °C, and then K2CO3 (41.2g, 0.3mol) was added. The mixture was further stirred at room temperature overnight and then was adjusted by 0.1N HCl to a pH of about 7. The mixture was filtered and the solvent was evaporated under reduced pressure from the filtrate, and then to the residue was added 150ml of water. The aqueous layer was extracted by 150ml of toluene (50ml X 3), and then p-nitrobenzoic acid (16.8g, 0.1mol) was added to the aqueous layer and refluxed for 30 minutes, after which, the reaction mixture was cooled and further stirred at 0-5 °C for 2 hours. Then the reaction mixture was filtered and dried to give 31.7g of a white solid.
-
The resulting salt and anhydrous ethanol (120ml) were added to a reaction flask, and warmed to 70-75 °C. Triethylamine (12ml) was added dropwise, and the reaction was conducted at that temperature for 2 hours. Then the mixture was cooled to 50 °C, at which point ethyl acetate (150ml) was added dropwsie. After the addition was complete, the mixture was cooled to 10 °C and further stirred for 4 hours. The mixture was filtered to give 15.6g of Lamivudine, and the yield was 68%. 1H-NMR (DMSO-d6) δ: 7.83(dd, 1H), 7.17∼7.23(dd, 2H), 6.21(t, 1H), 5.72 (dd, 1H), 5.29 (t, 2H), 5.16 (t, 1H), 3.70∼3.74 (m, 2H), 3.32∼3.43 (dd, 1H), 3.01∼3.05(dd, 1H); Elemental analysis: C8H11N3O3S found(%): C 41.85, H 4.88 N 18.25, S 13.94; calculated (%) C 41.91, H 4.84, N 18.33, S13.99.
PAPER

http://www.beilstein-journals.org/bjoc/single/articleFullText.htm?publicId=1860-5397-9-265
References
- 1″Lamivudine”. The American Society of Health-System Pharmacists. Retrieved 31 July 2015.
- 2
- “WHO Model List of EssentialMedicines” (PDF). World Health Organization. October 2013. Retrieved 22 April 2014.
- 3
- Hamilton, Richart (2015). Tarascon Pocket Pharmacopoeia 2015 Deluxe Lab-Coat Edition. Jones & Bartlett Learning. p. 65. ISBN 9781284057560.
- 4
- Fox Z, Dragsted UB, Gerstoft J, et al. (2006). “A randomized trial to evaluate continuation versus discontinuation of lamivudine in individuals failing a lamivudine-containing regimen: The COLATE trial”. Antiviral Therapy 11 (6): 761–70. PMID 17310820.
- 5
- http://hivdb.stanford.edu/index.html Stanford University Drug Resistance Database.
- 6
- Koziel MJ, Peters MG (2007). “Viral hepatitis in HIV infection”. N Engl J Med 356 (14): 1445–54. doi:10.1056/NEJMra065142. PMID 17409326.
- 7
- “Epivir package insert” (PDF). GlaxoSmithKline. Retrieved January 20, 2011.
- 8
- Curtis, John (June 20, 1998). “Hunting Down HIV”. Yale Medicine.
- 9
- Soderstrom, E John (July 10, 2003). “National Institutes of Health: Moving Research from the Bench to the Bedside”.
- 10
- Cohen, Elizabeth (September 29, 2014). “Doctor treats Ebola with HIV drug in Liberia — seemingly successfully”. CNN.
- 11 HIV drug may stop Ebola. Operonlabs.com, 27 September 2014
External links
- Epivir (manufacturer’s website)
 |
|
 |
|
| Systematic (IUPAC) name | |
|---|---|
|
4-amino-1-[(2R,5S)-2-(hydroxymethyl)-1,3-oxathiolan-5-yl]-1,2-dihydropyrimidin-2-one
|
|
| Clinical data | |
| Trade names | Epivir |
| AHFS/Drugs.com | monograph |
| MedlinePlus | a696011 |
| Pregnancy category |
|
| Routes of administration |
Oral |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 86% |
| Protein binding | Less than 36% |
| Biological half-life | 5 to 7 hours |
| Excretion | Renal (circa 70%) |
| Identifiers | |
| CAS Number | 134678-17-4 |
| ATC code | J05AF05 (WHO) |
| PubChem | CID 73339 |
| DrugBank | DB00709 |
| ChemSpider | 66068 |
| UNII | 2T8Q726O95 |
| KEGG | D00353 |
| ChEMBL | CHEMBL141 |
| NIAID ChemDB | 000388 |
| Synonyms | L-2′,3′-dideoxy-3′-thiacytidine |
| PDB ligand ID | 3TC (PDBe, RCSB PDB) |
| Chemical data | |
| Formula | C8H11N3O3S |
| Molar mass | 229.26 g/mol |
Trioxacarcin A

Trioxacarcin A, DC-45A
CAS No. 81552-36-5
- Molecular FormulaC42H52O20
- Average mass876.850 Da
-
17′-[(4-C-Acetyl-2,6-dideoxyhexopyranosyl)oxy]-19′-(dimethoxymethyl)-10′,13′-dihydroxy-6′-methoxy-3′-methyl-11′-oxospiro[oxirane-2,18′-[16,20,22]trioxahexacyclo[17.2.1.02,15.05,14.07,12.017,21 ]docosa[2(15),3,5(14),6,12]pentaen]-8′-yl 4-O-acetyl-2,6-dideoxy-3-C-methylhexopyranoside(1S,2R,3aS,4S,8S,10S,13aS)-13a-(4-C-Acetyl-2,6-dideoxy-alpha-L-xylo-hexopyranosyloxy)-2-(dimethoxymethyl)-10,12-dihydroxy-7-methoxy-5-methyl-11-oxo-4,8,9,10,11,13a-hexahydro-3aH-spiro[2,4-epoxyfuro[3,2-b]naphtho[2,3-h]-1-benzopyran-1,2′-oxiran]-8-yl 4-O-acetyl-2,6-dideoxy-3-C-methyl-alpha-L-xylo-hexopyranoside
- Kyowa Hakko Kirin INNOVATOR
Trioxacarcin B
Trioxacarcin B; Antibiotic DC 45B1; DC-45-B1; Trioxacarcin A, 14,17-deepoxy-14,17-dihydroxy-; AC1MJ5N1; 81534-36-3;
| Molecular Formula: | C42H54O21 |
|---|---|
| Molecular Weight: | 894.86556 g/mol |
Trioxacarcin C
(CAS NO.81781-28-4):C42H54O20
Molecular Weight: 878.8662 g/mol
Structure of Trioxacarcin C :


The trioxacarcins are polyoxygenated, structurally complex natural products that potently inhibit the growth of cultured human cancer cells
Natural products that bind and often covalently modify duplex DNA figure prominently in chemotherapy for human cancers. The trioxacarcins are a new class of DNA- modifying natural products with antiproliferative effects. The trioxacarcins were first described in 1981 by Tomita and coworkers (Tomita et al. , J. Antibiotics, 34( 12): 1520- 1524, 1981 ; Tamaoki et al., J. Antibiotics 34( 12): 1525- 1530, 1981 ; Fujimoto et al. , J. Antibiotics 36(9): 1216- 1221 , 1983). Trioxacarcin A, B, and C were isolated by Tomita and coworkers from the culture broth of Streptomyces bottropensis DO-45 and shown to possess anti-tumor activity in murine models as well as gram-positive antibiotic activity. Subsequent work led to the discovery of other members of this family. Trioxacarcin A is a powerful anticancer agent with subnanmolar IC70 values against lung (LXFL 529L, H-460), mammary (MCF-7), and CNS (SF-268) cancer cell lines. The trioxacarcins have also been shown to have antimicrobial activity {e.g., anti-bacterial and anti-malarial activity) (see, e.g. , Maskey et al., J. Antibiotics (2004) 57:771 -779).
trioxacarcin A
An X-ray crystal structure of trioxacarcin A bound to N-7 of a guanidylate residue in a duplex DNA oligonucleotide substrate has provided compelling evidence for a proposed pathyway of DNA modification that proceeds by duplex intercalation and alkylation (Pfoh et al, Nucleic Acids Research 36( 10):3508-3514, 2008).
All trioxacarcins appear to be derivatives of the aglycone, which is itself a bacterial isolate referred to in the patent literature as DC-45-A2. U.S. Patent 4,459,291 , issued July 10, 1984, describes the preparation of DC-45-A2 by fermentation. DC-45-A2 is the algycone of trioxacarcins A, B, and C and is prepared by the acid hydrolysis of the fermentation products trioxacarcins A and C or the direct isolation from the fermentation broth of Streptomyces bottropensis.
Based on the biological activity of the trioxacarcins, a fully synthetic route to these compounds would be useful in exploring the biological and chemical activity of known trioxacarcin compounds and intermediates thereto, as well as aid in the development of new trioxacarcin compounds with improved biological and/or chemical properties.
PAPER
Component-Based Syntheses of Trioxacarcin A, DC-45-A1, and Structural Analogs
T. Magauer, D. Smaltz, A. G. Myers, Nat. Chem. 2013, 5, 886–893. (Link)

Component-based syntheses of trioxacarcin A, DC-45-A1 and structural analogues
- Nature Chemistry5,886–893(2013)
- doi:10.1038/nchem.1746
PAPER

A schematic shows a trioxacarcin C molecule, whose structure was revealed for the first time through a new process developed by the Rice lab of synthetic organic chemist K.C. Nicolaou. Trioxacarcins are found in bacteria but synthetic versions are needed to study them for their potential as medications. Trioxacarcins have anti-cancer properties. Source: Nicolaou Group/Rice University
A team led by Rice University synthetic organic chemist K.C. Nicolaou has developed a new process for the synthesis of a series of potent anti-cancer agents originally found in bacteria.
The Nicolaou lab finds ways to replicate rare, naturally occurring compounds in larger amounts so they can be studied by biologists and clinicians as potential new medications. It also seeks to fine-tune the molecular structures of these compounds through analog design and synthesis to improve their disease-fighting properties and lessen their side effects.
Such is the case with their synthesis of trioxacarcins, reported this month in the Journal of the American Chemical Society.
PAPER

PATENT
http://www.google.com/patents/EP2550285A1?cl=en

(S)-9-Hvdrox v- 10-methoxy-5-(4-methoxybenzylox v)- 1 -(methoxymethox y)-3- methyl-8-oxo-5,6.7.8-tetrahvdroanthracene-2-carbaldehvde. Potassium osmate dihydrate (29 mg, 0.079 mmol, 0.05 equiv) was added to an ice -cooled mixture of (S,£)-9-hydroxy- 10- methoxy-4-(4-methoxybenzyloxy)-8-(methoxymethoxy)-6-methyl-7-(prop- l -enyl)-3,4- dihydroanthracen-l -one (780 mg, 1.58 mmol, 1 equiv), 2,6-lutidine (369 μί, 3.17 mmol, 2.0 equiv), and sodium periodate ( 1.36 g, 6.33 mmol, 4.0 equiv) in a mixture of tetrahydrofuran (20 mL) and water ( 10 mL). After 10 min, the cooling bath was removed and the reaction flask was allowed to warm to 23 °C. After 1.5 h, the reaction mixture was partitioned between water ( 100 mL) and ethyl acetate (150 mL). The layers were separated. The organic layer was washed with aqueous sodium chloride solution (50 mL) and the washed solution was dried over sodium sulfate. The dried solution was filtered and the filtrate was concentrated. The residue was purified by flash-column chromatography (20% ethyl acetate- hexanes) to provide 498 mg of the product, (5)-9-hydroxy- 10-methoxy-5-(4- methoxybenzyloxy)- l -(methoxymethoxy)-3-methyl-8-oxo-5,6,7,8-tetrahydroanthracene-2- carbaldehyde, as an orange foam (65%). Ή NMR (500 MHz, CDC13): 15.17 (s, 1 H), 10.74 (s, 1 H), 7.66 (s, 1 H), 7.27 (d, 2H, 7 = 8.5 Hz), 6.86 (d, 2H, 7 = 8.6 Hz), 5.30-5.18 (m, 3H), 4.63 (d, 1H,7= 11.1 Hz), 4.52 (d, 1H,7 = 12.0 Hz), 3.86 (s, 3H), 3.79 (s, 3H), 3.62 (s, 3H), 3.22 (m, 1H), 2.75 (s, 3H), 2.63 (m, 1H), 2.54 (m, 1H), 2.08 (m, 1H). I3C NMR (125 MHz, CDC13): 204.9, 193.2, 163.2, 161.7, 159.2, 144.4, 141.7, 137.0, 130.1, 129.4, 120.7, 117.9, 113.8, 110.0, 102.8, 70.4, 67.2, 62.9, 58.3, 55.2, 32.3, 26.3, 22.2. FTIR, cm-1 (thin film): 2936 (m), 2907 (m), 1684 (s), 1611 (s), 1377 (s), 1246 (s). HRMS (ESI): Calcd for
(C27H2808+K)+: 519.1416; Found 519.1368. TLC (20% ethyl acetate-hexanes): R,= 0.17 (CAM).
86% yield
[00457] (S)-l,9-Dihvdroxy-10-methoxy-5-(4-methoxybenzyloxy)-3-methyl-8-oxo-5,6,7,8- tetrahydroanthracene-2-carbaldehyde. A solution of B-bromocatecholborane (418 mg, 2.10 mmol, 2.0 equiv) in dichloromethane (15 mL) was added to a solution of (S)-9-hydroxy-10- methoxy-5-(4-methoxybenzyloxy)-l-(methoxymethoxy)-3-methyl-8-oxo-5,6,7,8- tetrahydroanthracene-2-carbaldehyde (490 mg, 1.05 mmol, 1 equiv) in dichloromethane (15 mL) at -78 °C. After 50 min, the reaction mixture was diluted with saturated aqueous sodium bicarbonate solution (25 mL) and dichloromethane (100 mL). The cooling bath was removed, and the partially frozen mixture was allowed to warm to 23 °C. The biphasic mixture was diluted with 0.2 M aqueous sodium hydroxide solution (100 mL). The layers were separated. The aqueous layer was extracted with dichloromethane (100 mL). The organic layers were combined. The combined solution was washed sequentially with 0.1 M aqueous hydrochloric acid solution (100 mL), water (2 x 100 mL), then saturated aqueous sodium chloride solution (100 mL) and the washed solution was dried over sodium sulfate. The dried solution was filtered and the filtrate was concentrated to provide 380 mg of the product, (S)-\ ,9- dihydroxy-10-methoxy-5-(4-methoxybenzyloxy)-3-methyl-8-oxo-5,6,7,8- tetrahydroanthracene-2-carbaldehyde, as a yellow foam (86%). Ή NMR (500 MHz, CDCI3):
15.89 (brs, 1H), 12.81 (br s, 1H), 10.51 (s, 1H), 7.27-7.26 (m, 3H), 6.86 (d, 2H, J = 9.2 Hz), 5.14 (app s, 1H),4.62 (d, \H,J= 11.0 Hz), 4.51 (d, 1H,7= 11.0 Hz), 3.85 (s, 3H), 3.80 (s, 3H), 3.21 (m, 1H), 2.73 (s, 3H), 2.62 (m, 1H), 2.54 (m, 1H), 2.07 (m, 1H). I3C NMR (125 MHz, CDCI3): 204.4, 192.7, 166.6, 164.3, 159.3, 144.4, 142.7, 137.9, 130.4, 130.2, 129.4, 114.9, 114.2, 113.9, 113.8, 109.4, 70.4, 67.1,62.8, 55.3, 31.8, 26.5. FTIR, cm-1 (thin film): 3316 (brw), 2938 (m), 1678 (m), 1610 (s), 1514 (m), 1393 (m), 1246 (s). HRMS (ESI): Calcd for (C25H2407+Na)+ 459.1414; Found 459.1354. TLC (50% ethyl acetate-hexanes): R = 0.30 (CAM).
[00458] (5)-2,2-Di-/erf-butyl-7-methoxy-8-(4-methoxybenzyloxy)-5-methyl- 1 1 -oxo- 8,9, 10, 1 1 -tetrahydroanthra[9, 1 -de \ 1 ,3,21dioxasiline-4-carbaldehyde. Όι‘-tert- butyldichlorosilane (342 μL·, 1.62 mmol, 1.8 equiv) was added to a solution of (5)-l ,9- dihydroxy- 10-methoxy-5-(4-methoxybenzyloxy)-3-methyl-8-oxo-5,6,7,8- tetrahydroanthracene-2-carbaldehyde (380 mg, 0.90 mmol, 1 equiv), hydroxybenzotriazole (60.8 mg, 0.45 mmol, 0.50 equiv) and diisopropylethylamine (786 μί, 4.50 mmol, 5.0 equiv) in dimethylformamide (30 mL). The reaction flask was heated in an oil bath at 55 °C. After 2 h, the reaction flask was allowed to cool to 23 °C. The reaction mixture was partitioned between saturated aqueous sodium bicarbonate solution (100 mL) and ethyl acetate (150 mL). The layers were separated. The organic layer was washed sequentially with water (2 x 100 mL) then saturated aqueous sodium chloride solution (100 mL) and the washed solution was dried over sodium sulfate. The dried solution was filtered and the filtrate was concentrated. The residue was purified by flash-column chromatography (10% ethyl acetate- hexanes) to provide 285 mg of the product, (S)-2,2-di-/<?ri-butyl-7-methoxy-8-(4- methoxybenzyloxy)-5-methyl- 1 1 -oxo-8,9, 10, 1 1 -tetrahydroanthra[9, 1 -de] [ 1 ,3,2]dioxasiline-4- carbaldehyde, as a yellow foam (56%). The enantiomeric compound (/?)-2,2-di-½ri-butyl-7- methoxy-8-(4-methoxybenzyloxy)-5-methyl- l 1 -oxo-8,9, 10, 1 1 -tetrahydroanthra[9, 1 – i/e][ l ,3,2]dioxasiline-4-carbaldehyde has been prepared using the same route by utilizing R- (4-methoxybenzyloxy)cyclohex-2-enone as starting material. Ή NMR (500 MHz, CDCI3): 10.84 (s, 1 H), 7.37 (s, 1 H), 7.25 (d, 2H, J = 8.8 Hz), 6.85 (d, 2H, = 8.7 Hz), 5.20 (app s, 1 H), 4.62 (d, 1 H, 7 = 10.0 Hz), 4.51 (d, 1H, J = 1 1.4 Hz), 3.88 (s, 3H), 3.78 (s, 3H), 3.03 (m, 1H), 2.73 (s, 3H), 2.57-2.53 (m, 2H), 2.07 (m, 1H), 1.16 (s, 9H), 1.14 (s, 9H). 13C NMR (125 MHz, CDCl3): 195.6, 190.9, 160.5, 159.2, 150.4, 145.7, 140.4, 134.0, 133.9, 130.3, 129.4, 1 19.5, 1 16.6, 1 15.8, 1 15.3, 1 13.8, 70.4, 67.8, 62.9, 55.2, 34.0, 26.0, 26.0, 22.5, 21.3, 21.1. FTIR, cm“1 (thin film): 2936 (m), 2862 (m), 1682 (s), 1607 (s), 1371 (s), 1244 (s) 1057 (s). HRMS (ESI): Calcd for (C33H4o07Si+H)+ 577.2616; Found 577.2584. TLC (10% ethyl acetate-hexanes): R/ = 0.19 (CAM). Alternative Routes to (4S,6S)-6-(½rt-Butyldimethylsilyloxy)-4-(4-methoxybenzyloxy) cyclohex-2-enone.
Alternative Route 1.
[00459] (25,45,55)-2,4-Bis(ferf-butyldimethylsilyloxy)-5-hvdroxycvclohexanone. Dess- Martin periodinane (6.1 1 g, 14.4 mmol, 1.1 equiv) was added to a solution of diol (5.00 g, 13.3 mmol, 1 equiv) in tetrahydrofuran (120 mL) at 23 °C (Lim, S. M.; Hill, N.; Myers, A. G. J. Am. Chem. Soc. 2009, 131, 5763-5765). After 40 min, the reaction mixture was diluted with ether (300 mL). The diluted solution was filtered through a short plug of silica gel (-5 cm) and eluted with ether (300 mL). The filtrate was concentrated. The bulk of the product was transformed as outlined in the following paragraph, without purification. Independently,
s
an analytically pure sample of the product was obtained by flash-column chromatography (20% ethyl acetate-hexanes) and was characterized by Ή NMR, l 3C NMR, IR, and HRMS. TLC: (17% ethyl acetate-hexanes) R = 0.14 (CAM); Ή NMR (500 MHz, CDCI3) δ: 4.41 (dd, 1 H, 7 = 9.8, 5.5 Hz), 4.05 (m, l H), 4.00 (m, 1H), 2.81 (ddd, 1 H, 7 = 14.0, 3.7, 0.9 Hz), 2.52 (ddd, 1 H, 7 = 14.0, 5.3, 0.9 Hz), 2.29 (br s, 1 H), 2.18 (m, 1H), 1.98 (m, 1 H), 0.91 (s, 9H), 0.89 (s, 9H), 0.13 (s, 3H), 0.1 1 (s, 3H), 0.09 (s, 3H), 0.04 (s, 3H); l 3C NMR (125 MHz, CDCI3) δ: 207.9, 73.9, 73.3, 70.5, 43.3, 39.0, 25.7, 25.6, 18.3, 17.9, -4.7, -4.8, -4.9, -5.4; FTIR (neat), cm“‘ : 3356 (br), 2954 (m), 2930 (m), 2857 (m), 1723 (m), 1472 (m). 1253 (s), 1 162 (m), 1 105 (s), 1090 (s), 1059 (s), 908 (s), 834 (s), 776 (s), 731 (s); HRMS (ESI): Calcd for (C|8H3804Si2+H)+ 375. 2381 , found 375.2381.
[00460] (4 ,6 )-4.6-Bis(fcr/-butyldimethylsilyloxy)cvclohex-2-enone. Trifluoroacetic anhydride (6.06 mL, 43.6 mmol, 3.3 equiv) was added to an ice-cooled solution of the alcohol ( 1 equiv, see paragraph above) and triethylamine ( 18.2 mL, 131 mmol, 9.9 equiv) in dichloromethane (250 mL) at 0 °C. After 20 min, the cooling bath was removed and the reaction flask was allowed to warm to 23 °C. After 18 h, the reaction flask was cooled in an ice bath at 0 °C, and the product solution was diluted with water ( 100 mL). The cooling bath was removed and the reaction flask was allowed to warm to 23 °C. The layers were separated. The aqueous layer was extracted with dichloromethane (2 x 200 mL). The organic layers were combined. The combined solution was washed with saturated aqueous sodium chloride solution ( 100 mL) and the washed solution was dried over sodium sulfate. The dried solution was filtered and the filtrate was concentrated. The residue was purified by flash- column chromatography (6% ethyl acetate-hexanes) to provide 3.02 g of the product, (4S,65)-4,6-bis(/eri-butyldimethylsilyloxy)cyclohex-2-enone, as a colorless oil (64% over two steps). TLC: (20% ethyl acetate-hexanes) R = 0.56 (CAM); Ή NMR (500 MHz, CDC13) δ: 6.76 (dd, 1 Η, / = 10.1 , 3.6 Hz), 5.88 (d, 1 H, 7 = 10.1 Hz), 4.66 (ddd, 1 H, 7 = 5.6, 4.1 , 3.6 Hz), 4.40 (dd, 1 H, 7 = 8.1 , 3.7 Hz), 2.26 (ddd, 1 H, / = 13.3, 8.0, 4.1 Hz), 2.1 1 (ddd, 1 H, J = 13.2, 5.6, 3.8 Hz), 0.91 (s, 9H), 0.89 (s, 9H), 0.12 (s, 3H), 0. 1 1 (s, 3H), 0. 10 (s, 3H), 0.10 (s, 3H); 13C NMR ( 125 MHz, CDC13) δ: 197.5, 150.3, 127.0, 71 .0, 64.8, 41.6, 25.7, 25.7, 18.3, 18.1 , -4.7, -4.8, -4.8, -5.4; FTIR (neat), cm-1 : 3038 (w), 2955 (m), 2930 (m), 1705 (m), 1472 (m), 1254 (m), 1084 (m), 835 (s), 777 (s), 675 (s); HRMS (ESI): Calcd for (C,8H3602Si2+Na)+ 379. 2095, found 379. 2080.
[00461] (4S,6S)-6-(/er/-Butyldimethylsilyloxy)-4-hydroxycvclohex-2-enone. Tetra- j- butylammonium fluoride ( 1 .0 M solution in tetrahydrofuran, 8.00 mL, 8.00 mmol, 1 .0 equiv) was added to an ice-cooled solution of the enone (2.85 g, 8.00 mmol, 1 equiv) and acetic acid (485 ί, 8.00 mmol, 1 .0 equiv) in tetrahydrofuran (80 mL) at 0 °C. After 2 h, the cooling bath was removed and the reaction flask was allowed to warm to 23 °C. After 22 h, the reaction mixture was partitioned between water ( 100 mL) and ethyl acetate (300 mL). The layers were separated. The aqueous layer was extracted with ethyl acetate (2 x 300 mL). The organic layers were combined. The combined solution was washed sequentially with saturated aqueous sodium bicarbonate solution ( 100 mL) then saturated aqueous sodium chloride solution ( 100 mL) and the washed solution was dried over sodium sulfate. The dried solution was filtered and the filtrate was concentrated. The residue was purified by flash- column chromatography (25% ethyl acetate-hexanes) to provide 760 mg of the product, (4S,6S)-6-(ferNbutyldimethylsilyloxy)-4-hydroxycyclohex-2-enone, as a white solid (39%). TLC: (20% ethyl acetate-hexanes) R/ = 0.20 (CAM); Ή NMR (500 MHz, CDC13) δ: 6.87 (dd, 1 Η, 7 = 10.2, 3.2 Hz), 5.95 (dd, 1H, J = 10.3, 0.9 Hz), 4.73 (m, 1 H), 4.35 (dd, 1 H, 7 = 7.6, 3.7 Hz), 2.39 (m, 1 H), 2. 13 (ddd, 1 H, J = 13.3, 6.2, 3.4 Hz), 1.83 (d, 1 H, J = 6.2), 0.89 (s, 9H), 0.10 (s, 3H), 0. 10 (s, 3H); 13C NMR ( 125 MHz, CDCb) δ: 197.3, 150.0, 127.5, 70.9, 64.2, 41 .0, 25.7, 18.2, -4.8, -5.4; FTIR (neat), cm“1 : 2956 (w), 293 1 (w), 2858 (w), 1694 (m); HRMS (ESI): Calcd for (C |2H2203Si+H)+ 243.141 1 , found 243. 1412.
82″:.
[00462] (45.6S)-6-(fgrf-Butyldimethylsilyloxy)-4-(4-methoxybenzyloxy)cvclohex-2- enone. Triphenylmethyl tetrafluoroborate ( 16 mg, 50 μπιοΐ, 0.050 equiv) was added to a solution of 4-methoxybenzyl-2,2,2-trichloroacetimidate (445 μΙ_, 2.5 mmol, 2.5 equiv) and alcohol (242 mg, 1 .0 mmol, 1 equiv) in ether ( 10 mL) at 23 °C. After 4 h, the reaction mixture was partitioned between saturated aqueous sodium bicarbonate solution ( 15 mL) and ethyl acetate (50 mL). The layers were separated. The aqueous layer was extracted with ethyl acetate (50 mL). The organic layers were combined. The combined solution was washed with water (2 x 20 mL) and the washed solution was dried over sodium sulfate. The dried solution was filtered and the filtrate was concentrated. The residue was purified by flash column chromatography (5% ethyl acetate-hexanes initially, grading to 10% ethyl acetate-hexanes) to provide 297 mg of the product, (4S,6S)-6-(im-butyldimethylsilyloxy)-4-(4- methoxybenzyloxy)cyclohex-2-enone, as a colorless oil (82%).
Alternative Route 2.
[00463] (5)-?erf-Butyl(4-(4-methoxybenzyloxy)cvclohexa- 1.5-dienyloxy)dimethylsilane. rerr-Butyldimethylsilyl trifluoromethanesulfonate (202 iL, 0.94 mmol, 2.0 equiv) was added to an ice-cooled solution of triethylamine (262 μί, 1.88 mmol, 4.0 equiv) and enone ( 109 mg, 0.47 mmol, 1 equiv) in dichloromethane (5.0 mL). After 30 min, the reaction mixture was partitioned between saturated aqueous sodium bicarbonate solution ( 10 mL), water (30 mL), and dichloromethane (40 mL). The layers were separated. The organic layer was washed sequentially with saturated aqueous ammonium chloride solution (20 mL) then saturated aqueous sodium chloride solution (20 mL) and the washed solution was dried over sodium sulfate. The dried solution was filtered and the filtrate was concentrated. The residue was purified by flash-column chromatography with triethylamine-treated silica gel (5% ethyl acetate-hexanes), to provide 130 mg of the product, (5)-ierr-butyl(4-(4- methoxybenzyloxy)cyclohexa- l ,5-dienyloxy)dimethylsilane, as a colorless oil (80%). Ή
NMR (500 MHz, CDC13): 7.27 (d, 2H, J = 8.7 Hz), 6.88 (d, 2H, J = 8.6 Hz), 5.96 (dd, 1 H, J = 9.9, 3.5 Hz), 5.87 (d, 1 H, 7 = 9.6 Hz), 4.94 (m, l H), 4.46 (s, 2H), 4.14 (m, 1 H), 3.81 (s, 3H), 2.49 (m, 2H), 0.93 (s, 9H), 0. 16 (s, 3H), 0.15 (s, 3H). , 3C NMR ( 125 MHz, CDC13): 159.1 , 147.5, 130.9, 129.2, 128.6, 128.1 , 1 13.8, 101.4, 70.2, 69.0, 55.3, 28.5, 25.7, 18.0, ^1.5, -4.5. FTIR, cm-1 (thin film): 2957 (m), 2931 (m), 2859 (m), 1655 (w), 1613 (w), 1515 (s), 1248 (s), 1229 (s), 1037 (m), 910 (s). HRMS (ESI): Calcd for (C2oH3o03Si+H)+ 347.2037; Found 347.1912. TLC (20% ethyl acetate-hexanes): R = 0.74 (CAM).
OP B OPMB DM 00 ,,Α,,
c Ύ’“ -ietone ii ·η- ) ‘”OH
OTBS 82 Q
[00464] (4S,6S)-6-Hvdroxy-4-(4-methoxybenzyloxy)cvclohex-2-enone. A solution of dimethyldioxirane (0.06 M solution in acetone, 2.89 mL, 0.17 mmol, 1.2 equiv) was added to an ice-cooled solution of (S)-ieri-butyl(4-(4-methoxybenzyloxy)cyclohexa- l ,5- dienyloxy)dimethylsilane (50 mg, 0.14 mmol, 1 equiv). After 10 min, the reaction mixture was partitioned between dichloromethane ( 15 mL) and 0.5 M aqueous hydrochloric acid ( 10 mL). The layers were separated. The organic layer was washed sequentially with saturated aqueous sodium bicarbonate solution ( 10 mL) then water ( 10 mL) and the washed solution was dried over sodium sulfate. The dried solution was filtered and the filtrate was concentrated. The residue was purified by flash-column chromatography to provide 30 mg of the product, (4S,6S)-6-hydroxy-4-(4-methoxybenzyloxy)cyclohex-2-enone, as a colorless oil (82%). Ή NMR (500 MHz, CDC13): 7.28 (d, 2H, J = 8.2 Hz), 6.89 (m, 3H), 6.09 (d, 1 H, J = 10.1 Hz), 4.64 (m, 2H), 4.53 (d, 1 H, 7 = 1 1 .4 Hz), 4.24 (m, 1 H), 3.81 (s, 3H), 3.39 (d, 1 H, 7 = 1.4 Hz), 2.67 (m, 1 H), 1 .95 (ddd, 1 H, 7 = 12.8, 12.8, 3.6 Hz). I 3C NMR ( 125 MHz, CDC13): 200.4, 159.5, 146.6, 129.7, 129.4, 127.8, 1 14.0, 71.6, 69.8, 68.9, 55.3, 35.1 . FTIR, cm-1 (thin film): 3474 (br), 2934 (m), 2864 (m), 1692 (s), 1613 (m), 1512 (s), 1246 (s), 1059 (s), 1032 (s). HRMS (ESI): Calcd for (C,4Hl6O4+Na)+ 271.0941 ; Found 271.0834. TLC (50% ethyl acetate-hexanes): R/ = 0.57 (CAM).
[00465] (45,65)-6-(½rt-Butyldimethylsilyloxy)-4-(4-methoxybenzyloxy)cvclohex-2- enone. rerr-Butyldimethychlorosilane (26 mg, 0.18 mmol, 1.5 equiv) was added to an ice- cooled solution of (45,65)-6-hydroxy-4-(4-methoxybenzyloxy)cyclohex-2-enone (29 mg, 0.12 mmol, 1 equiv) and imidazole (24 mg, 0.35 mmol, 3 equiv) in dimethylformamide (0.5 mL). After 45 min, the reaction mixture was partitioned between water (15 mL), saturated aqueous sodium chloride solution (15 mL), and ethyl acetate (20 mL). The layers were separated. The organic layer was washed with water (2 x 20 mL) and the washed solution was dried over sodium sulfate. The dried solution was filtered and the filtrate was concentrated. The residue was purified by flash-column chromatography to provide 29 mg of the product, (4S,6S)-6-(rm-butyldimethylsilyloxy)-4-(4-methoxybenzyloxy)cyclohex-2- enone, as a colorless oil (87%).
Glycosylation experiments
[00466] Glycosylation experiments demonstrate that the chemical process developed allows for the preparation of synthetic, glycosylated trioxacarcins. Specifically, the C4 or CI 3 hydroxyl group may be selectively glycosylated with a glycosyl donor (for example, a glycosyl acetate) and an activating agent (for example, TMSOTf), which enables preparation of a wide array of trioxacarcin analogues.
Selective Glycosylation of the C4 Hydroxyl Group
[00467] 2,3-Dichloro-5,6-dicyanobenzoquinone ( 19.9 mg, 88 μιτιοΐ, 1.1 equiv) was added to a vigorously stirring, biphasic solution of differentially protected trioxacarcin precursor (60 mg, 80 μιτιοΐ, 1 equiv) in dichloromethane ( 1.1 mL) and pH 7 phosphate buffer (220 μί) at 23 °C. The reaction flask was covered with aluminum foil to exclude light. Over the course of 3 h, the reaction mixture was observed to change from myrtle green to lemon yellow. The product solution was partitioned between water (5 mL) and dichloromethane (50 mL). The layers were separated. The organic layer was dried over sodium sulfate. The dried solution was filtered and the filtrate was concentrated. The residue was purified by preparatory HPLC (Agilent Prep-C 18 column, 10 μιτι, 30 x 150 mm, UV detection at 270 nm, gradient elution with 40→90% acetonitrile in water, flow rate: 15 mL/min) to provide 33 mg of the product as a yellow-green powder (65%).
[00468] Trimethylsilyl triflate ( 10% in dichloromethane, 28.3 μί, 16 μπιοΐ, 0.3 equiv) was added to a suspension of deprotected trioxacarcin precursor (33 mg, 52 μπιοΐ, 1 equiv), 1 -0- acetyltrioxacarcinose A ( 14.1 mg, 57 μιτιοΐ, 1.1 equiv), and powdered 4- A molecular sieves (-50 mg) in dichloromethane (1 .0 mL) at -78 °C. After 5 min, the mixture was diluted with dichloromethane containing 10% triethylamine and 10% methanol (3 mL). The reaction flask was allowed to warm to 23 °C. The mixture was filtered and partitioned between
dichloromethane (40 mL) and saturated aqueous sodium chloride solution (5 mL). The layers were separated. The organic layer was dried over sodium sulfate. The dried solution was filtered and the filtrate was concentrated. The residue was purified by preparatory HPLC (Agilent Prep-C 18 column, 10 μπι, 30 x 150 mm, UV detection at 270 nm, gradient elution with 40→90% acetonitrile in water, flow rate: 15 mL/min) to provide 20 mg of the product as a yellow-green powder (47%). TLC: (5% methanol-dichloromethane) R = 0.40 (CAM); Ή NMR (500 MHz, CDC13) δ: 7.47 (s, 1H), 5.38 (d, 1H, J = 3.6 Hz), 5.35 (app s, 1 H), 5.26 ppm (d, 1 H, 7 = 4.0 Hz), 4.84 (d, 1 H, J = 4.0 Hz), 4.78 (dd, 1 H, 7 = 12.3, 5.2 Hz), 4.75 (s, 1H), 4.71 (s, 1 H), 4.52 (q, 1H, J = 6.6 Hz), 3.86 (s, 1 H), 3.83 (s, 3H), 3.62 (s, 3H), 3.47 (s, 3H), 3.15 (d, l H, y = 5.3 Hz), 3.05 (d, 1 H, 7 = 5.3 Hz), 2.60 (s, 3H), 2.58 (m, 1H), 2.35 (m, 1 H), 2.14 (s, 3H), 1.96 (dd, 1 H, 7 = 14.6, 4.1 Hz), 1.62 (d, 1 H, 7 = 14.6 Hz), 1.26 (s, 1 H), 1.23 (d, 3H, J = 6.6 Hz), 1.08 (s, 3H), 0.95 (s, 9H), 0.24 (s, 3H), 0.16 (s, 3H); ‘3C NMR ( 125 MHz, CDC13) 6: 202.8, 170.5, 163.2, 151.8, 144.4, 142.4, 135.2, 126.6, 1 16.8, 1 15.2, 1 15.1 , 108.3, 104.0, 100.3, 98.6, 98.3, 74.6, 73.4, 69.8, 69.5, 69.5, 68.9, 69.5, 69.5, 68.9, 68.4, 62.9, 62.7, 57.2, 56.8, 50.7, 38.8, 36.8, 26.0, 25.9, 21.1 , 20.6, 18.6, 17.0, -4.2, -5.3; FTIR (neat), cm“‘ : 2953 (w), 2934 (w), 2857 (w), 1749 (w), 1622 (m), 1570 (w), 1447 (w), 1391 (m), 1321 (w), 1294 (w), 1229 (m), 1 159 (m), 1 121 (s), 1084 (s), 1071 (m), 1020 (m), 995 (s), 943 (s), 868 (m), 837 (m), 779 (m); HRMS (ESI): Calcd for (C4oH540i6Si+Na)+ 841.3073, found
841.3064.
Glycosylation of a Cycloaddition Coupling Partner
[00469] 2,3-Dichloro-5,6-dicyanobenzoquinone ( 14.3 mg, 63 μπιοΐ, 1.2 equiv) was added to a vigorously stirring, biphasic solution of differentially protected aldehyde (37 mg, 52 μιτιοΐ, 1 equiv) in dichloromethane (870 μί) and water (175 μί) at 23 °C. The reaction flask was covered with aluminum foil to exclude light. Over the course of 2 h, the reaction mixture was observed to change from myrtle green to lemon yellow. The product solution was partitioned between water (5 mL) and dichloromethane (40 mL). The layers were separated. The organic layer was dried over sodium sulfate. The dried solution was filtered and the filtrate was concentrated. The residue was purified by flash-column chromatography (5% ethyl acetate-hexanes initially, grading to 10% ethyl acetate-hexanes) to provide 28 mg of the product as a yellow powder (91 %). TLC: (20% ethyl acetate-hexanes) R/ = 0.37 (CAM); Ή NMR (500 MHz, CDC13) δ: 10.83 (s, 1H), 7.30 (s, 1 H), 5.45 (m, 1H), 4.68 (dd, 1H, / = 10.3, 4.2 Hz), 3.97 (s, 3H), 3.31 (brs, 1H), 2.72 (s, 3H), 2.51-2.45 (m, 1H), 2.41-2.37 (m, 1H), 1.15 (s, 9H), 1 , 13 (s, 9H), 0.88 (s, 9H), 0.15 (s, 3H), 0.1 1 (s, 3H); l 3C NMR (125 MHz, CDCI3) δ: 194.6, 191 , 160.5, 150.2, 146, 140.8, 135.8, 134, 1 19.6, 1 16.2, 1 15.4, 1 14.7, 72.7, 63.7, 62.4, 38.8, 29.9, 62.4, 38.8, 63.7, 62.4, 38.8, 63.7, 62.4, 38.8, 29.9, 26.2, 26.1 , 26, 22.7, 21.4; FTIR (neat), cm“1 : 3470 (br, w), 2934 (w), 2888 (w), 1684 (s), 1607 (s), 1560 (w), 1472 (m), 1445 (w), 1392 (m), 1373 (s), 1242 (s), 1 153 (s), 1 1 19 (w), 1074 (m), 1044 (s), 1013 (s), 982 (w), 934 (m), 907 (w), 870 (m), 827 (s), 795 (s), 779 (s), 733 (s), 664 (s); HRMS (ESI): Calcd for (C3iH4607Si2+H)+ 587.2855, found 587.2867.
[00470] Trimethylsilyl triflate (10% in dichloromethane, 25.9 μί, 14 μπιοΐ, 0.3 equiv) was added to a suspension of deprotected aldehyde (28 mg, 48 μηιοΐ, 1 equiv), 1-0- acetyltrioxacarcinose A (12.9 mg, 52 μπιοΐ, 1.1 equiv), and powdered 4-A molecular sieves (-50 mg) in dichloromethane ( 1.0 mL) at -78 °C. After 5 min, the mixture was diluted with dichloromethane containing 10% triethylamine and 10% methanol (3 mL). The reaction flask was allowed to warm to 23 °C. The mixture was filtered and partitioned between dichloromethane (40 mL) and saturated aqueous sodium chloride solution (5 mL). The layers were separated. The organic layer was dried over sodium sulfate. The dried solution was filtered and the filtrate was concentrated. The residue was purified by preparatory HPLC (Agilent Prep-C 18 column, 10 μπι, 30 x 150 mm, UV detection at 270 nm, gradient elution with 80→98% acetonitrile in water, flow rate: 15 mL/min) to provide 15 mg of the product as a yellow powder (41 %). TLC: (20% ethyl acetate-hexanes) R/ = 0.29 (CAM); Ή NMR (500 MHz, CDC13) δ: 10.83 (s, 1 H), 7.32 (s, 1 H), 5.43 (d, 1 H, J = 3.9 Hz), 5.32 (m, 1H), 4.74 (s, 1 H), 4.67 (dd, 1 H, J = 12.3, 5.0 Hz), 4.54 (q, 1H, J = 6.6 Hz), 3.91 (s, 1H), 3.88 (s, 3H), 2.72 (s, 3H), 2.59 (ddd, 1 H, J = 13.8, 5.0, 3.2 Hz), 2.34 (m, 1H), 2.14 (s, 3H), 1.97 (dd, 1H, J = 14.2, 4.2 Hz), 1.71 (d, 1 Η, / = 14.6 Hz), 1.22 (d, 3H, J = 6.3 Hz), 1.15 (s, 9H), 1.15 (s, 9H), 1.08 (s, 3H), 0.93 (s, 9H), 0.23 (s, 3H), 0.13 (s, 3H); 13C NMR (125 MHz, CDC13) δ: 193.9, 191.0, 170.5, 146.4, 140.9, 134.0, 132.4, 1 19.8, 1 16.8, 1 15.8, 1 15.0, 1 10.8, 99.6, 74.6, 71.5, 70.4, 68.9, 62.9, 62.7, 39.1 , 36.9, 26.2, 26.1 , 26.1 , 25.9, 24.1 , 22.7, 21.5, 21.3, 21.1 , 18.7, 16.9, -4.1 , -5.3; FTIR (neat), cm-1 : 3524 (br, w), 2934 (m), 2861 (m), 1749 (m), 1686 (s), 1607 (s), 1560 (m), 1474 (m), 1447 (m), 1424 (w), 1375 (s), 1233 (s), 1 159 (s), 1 1 17 (m), 1080 (m), 1049 (s), 1015 (s), 997 (s), 937 (m), 883 (m), 872 (m), 827 (s), 797 (m), 781 (m), 737 (w), 677 (w), 667 (m); HRMS (ESI): Calcd for (C40H60O, ,Si2+H)+773.3747, found 773.3741.
General Glycosylation Procedure of the C13 Hydroxyl Group
[00471] Crushed 4-A molecular sieves (-570 mg / 1 mmol sugar donor) was added to a stirring solution of the sugar acceptor (1 equiv.) and the sugar donor (30.0 equiv.) in dichloromethane ( 1.6 mL / 1 mmol sugar donor) and diethylether (0.228 mL / 1 mmol sugar donor) at 23 °C. The bright yellow mixture was stirred for 90 min at 23 °C and finally cooled to -78 °C. TMSOTf (10.0 equiv.) was added over the course of 10 min at -78 °C. After 4 h, a second portion of TMSOTf (5.0 equiv.) was added at -78 °C and stirring was continued for 1 h. The last portion of TMSOTf (5 equiv.) was added. After 1 h, triethylamine (20 equiv.) was added and the reaction the product mixture was filtered through a short column of silica gel deactivated with triethylamine (30% ethyl acetate-hexanes initially, grading to 50% ethyl acetate-hexanes). H NMR analysis of the residue showed minor sugar donor remainings and that the sugar acceptor had been glycosylated. The residue was purified by preparatory HPLC (Agilent Prep-C 18 column, 10 μπι, 30 x 150 mm, UV detection at 270 nm, gradient elution with 40→100% acetonitrile in water, flow rate: 15 mL/min) to provide the glycosylation product as a bright yellow oil
Three Specific Compounds Prepared by the General Glycosylation Procedure for the CI 3 Hydroxyl Group:
[00472] 10% yield; TLC: (50% ethyl acetate-hexane) R = 0.58 (UV, CAM); Ή NMR (600 MHz, CDC13) δ: 7.43 (s, 1 H), 5.84 (t, J = 3.6 Hz, 1 H), 5.29 (d, J = 4.2 Hz, 1 H), 5.19 (d, J = 4.2 Hz, 1 H), 5.01 (q, J = 6.6 Hz, 1 H), 4.75 (t, J = 3.6 Hz, 1 H), 4.73 (s, 1 H), 3.88 (s, OH), 3.77 (s, 3H), 3.63 (s, 3H), 3.47 (s, 3H), 3.03 (app q, J = 5.4 Hz, 2H), 2.84 (d, J = 6.0 Hz, 1 H), 2.77 (d, J = 6.0 Hz, 1 H), 2.72 (t, J = 6.6 Hz, 2H), 2.58 (s, 3H), 2.36 (s, 3H), 2.33 (t, J = 3.0 Hz, 2H), 2.23 (s, 3H), 2.1 1 -2.06 (m, 2H), 1.08 (d, J = 6.0 Hz, 3H). ‘
[00473] 81 % yield, TLC: (50% ethyl acetate-hexane) R = 0.30 (UV, CAM); Ή NMR (600 MHz, CDCI3) δ: 7.46 (s, 1 H), 7.28 (d, J = 9 Hz, 2H), 6.87 (d, J = 8.4 Hz, 2 H), 5.83 (dd, J = 3.6, 1.8 Hz, 1 H), 5.30 (d, J = 4.2 Hz, 1 H), 5.19 (d, J = 4.2 Hz, 1 H), 5.19 (m, 1 H), 5.00 (q, J = 6.0 Hz, 1 H), 4.96 (dd, J = 12.0, 4.8 Hz, 1 H), 4.75 (t, J = 3.6 Hz, 1 H), 4.74 (s, l H), 4.70 (d, y = 10.8 Hz, 1 H), 4.59 (d, J = 10.8 Hz, 1 H), 3.86 (s, OH), 3.83 (s, 3H), 3.80 (s, 3H), 3.63 (s, 3H), 3.47 (s, 3H), 2.81 (d, J = 6.0 Hz, 1 H), 2.73-2.68 (m, 1 H), 2.70 (d, J = 6.0 Hz, 1 H), 2.59 (s, 3H), 2.35 (s, 3H), 2.33-2.28 (m, 2H), 2.22 (s, 3H), 2.19- 2.1 3 (m, 1 H), 1 .08 (d, J = 6.0 Hz, 3H), 0.97 (s, 9H), 0.25 (s, 3H), 0.17 (s, 3H); HRMS (ESI): Calcd for (C49H62018Si+H)+ 967.3778, found 967.3795; HRMS (ESI): Calcd for (C ¾20,8Si+Na)+ 989.3598, found 989.3585.
[00474] Compound Detected by ESI Mass Spectrometry: Calculated Mass for
[C52H7| N302i Si-Hrl = 1 100.4277, Measured Mass = 1 100.4253.
PATENT
US 4511560
https://www.google.com/patents/US4511560
The physico-chemical characteristics of DC-45-A and DC-4-5-B2 according to this invention are as follows:
(1) DC-45-A
(1) Elemental analysis: H:5.74%, C:55.11%
(2) Molecular weight: 877
(3) Molecular formula: C42 H52 O20
(4) Melting point: 180° C.±3° C. (decomposed)
(5) Ultraviolet absorption spectrum: As shown in FIG. 1 (in 50% methanol)
(6) Infrared absorption spectrum: As shown in FIG. 2 (KBr tablet method)
(7) Specific rotation: [α]D 25 =-15.3° (c=1.0, ethanol)
(8) PMR spectrum (in CDC]3 ; ppm): 1.07 (3H,s); 1.10 (3H, d, J=6.8); 1.24 (3H,d, J=6.5); many peaks between 1.40-2.30; 2.14 (3H,s); 2.49 (3H,s); 2.63 (3H,s); many peaks between 2.30-2.80; 2.91 (1H,d, J=5.6); 3.00 (1H,d, J=5.6); 3.49 (3H,s); 3.63 (3H,s); 3.85 (3H, s); many peaks between 3.60-4.00; 4.18 (1H,s); 4.55 (1H,q, J=6.8); many peaks between 4.70-4.90; 5.03 (1H, q, J=6.5); 5.25 (1H,d, J=4.0); 5.39 (1H, d, J=4.0); 5.87 (1H, m); 7.52 (1H,s); 14.1 (1H,s)
(9) CMR spectrum (in CDCl3 ; ppm): 210.9; 203.8; 170.3; 162.1; 152.5; 145.2; 142.3; 135.3; 126.7; 117.0; 114.2; 108.3; 105.3; 99.7; 97.2; 93.7; 85.1; 79.0; 74.6; 71.1; 69.6; 69.3; 68.8; 67.9; 66.3; 64.0; 62.8; 57.3; 55.9; 36.5; 32.2; 28.0; 25.7; 20.9; 20.2; 17.0; 14.7
(10) Solubility: Soluble in methanol, ethanol, water and chloroform; slightly soluble in acetone and ethyl acetate, and insoluble in ether and n-hexane
(2) DC-45-B2
(1) Elemental analysis: H: 6.03%, C: 54.34%
(2) Molecular weight: 879
(3) Molecular formula: C42 H54 O20
(4) Melting point: 181°-182° C. (decomposed)
(5) Ultraviolet absorption spectrum: As shown in FIG. 5 (in 95% ethanol)
(6) Infrared absorption spectrum: As shown in FIG. 6 (KBr tablet method)
(7) Specific rotation: [α]D 25 =-10° (c=0.2, ethanol)
(8) PMR spectrum (in CDCl3 ; ppm): 1.07 (3H,s); many peaks between 1.07-1.5; many peaks between 1.50-2.80; 2.14 (3H,s); 2.61 (3H, broad s); 2.86 (1H, d, J=5.7); 2.96 (1H, d, J=5.7); 3.46 (3H,s); 3.63 (3H, s); 3.84 (3H, s); many peaks between 3.65-4.20; many peaks between 4.40-5.00; many peaks between 5.10-5.50; 5.80 (1H, broad s); 7.49 (1H, d, J=1.0); 14.1 (1H, s)
(9) CMR spectrum (in CDCl3 ; ppm): 202.8; 170.2; 163.1; 151.8; 144.8; 142.9; 135.4; 126.5; 116.8; 114.9; 107.3; 104.6; 101.5; 99.6; 98.0; 94.4; 74.4; 72.5; 71.4; 70.4; 69.1; 68.8; 68.3; 67.9; 67.5; 66.4; 62.9; 62.7; 56.8; 56.5; 48.0; 36.7; 32.3; 25.7; 20.8; 20.3; 18.2; 16.9; 15.5
(10) Solubility: Soluble in methanol, ethanol, acetone, ethyl acetate and chloroform; slightly soluble in benzene, ether and water; and insoluble in n-hexane.
//////
CC1C(C(CC(O1)OC2CC(C(=O)C3=C(C4=C5C(=C(C=C4C(=C23)OC)C)C6C7C(O5)(C8(CO8)C(O6)(O7)C(OC)OC)OC9CC(C(C(O9)C)(C(=O)C)O)O)O)O)(C)O)OC(=O)C
New FDA Guidance on Completeness Assessements for Type II API Drug Master Files
DRUG REGULATORY AFFAIRS INTERNATIONAL

Since 1st October 2012, special regulations have been applying to the US Type II Drug Master Files. This year in February, the FDA published a new Guidance for Industry. Read here what the DMF holder has to consider when submitting data about the API Drug Master File.
Since the coming into force of the “Generic Drug User Fee Act” (GDUFA) on 1st October 2012, special regulations have been applying to the submission to the FDA of a Drug Master Files for a pharmaceutical API (Type II DMF). The DMF holder must pay a one-time fee when authorising the reference of his/ her DMF in an application for a generic drug (Abbreviated New Drug Application, ANDA). Moreover, the DMF will undergo a completeness assessment through the FDA.
This year in February, the FDA published a Guidance for Industry entitled “Completeness Assessments for Type II API DMFs under GDUFA”…
View original post 350 more words
An Improved Process for the Preparation of Tenofovir Disoproxil Fumarate

Tenofovir Disoproxil Fumarate
For full details see end of page

PAPER

The current three-step manufacturing route for the preparation of tenofovir disoproxil fumarate (1) was assessed and optimized leading to a higher yielding, simpler, and greener process. Key improvements in the process route include the refinement of the second stage through the replacement of the problematic magnesium tert-butoxide (MTB) with a 1:1 ratio of a Grignard reagent and tert-butanol. The development of a virtually solvent-free approach and the establishment of a workup and purification protocol which allows the isolation of a pure diethyl phosphonate ester (8) was achieved
see………….http://pubs.acs.org/doi/abs/10.1021/acs.oprd.5b00364
An Improved Process for the Preparation of Tenofovir Disoproxil Fumarate

Department of Chemistry, Natural and Agricultural Sciences, University of Pretoria, 2 Lynnwood Road, Hatfield, 0002, Gauteng, South Africa

///////

Tenofovir Disoproxil Fumarate
5-[[(1R)-2-(6-Amino-9H-purin-9-yl)-1-methylethoxy]methyl]-2,4,6,8-tetraoxa-5-phosphanonanedioic Acid 1,9-Bis(1-methylethyl) Ester 5-Oxide (2E)-2-Butenedioate; GS 4331-05; PMPA Prodrug; Tenofovir DF; Virea; Viread;
GILEAD-4331-300
201341-05-1 – free base, (Tenofovir Disoproxil
Fumarate |
202138-50-9 |
| 113-115°C (dec.) |
| CAS No.: | 202138-50-9 |
|---|---|
| Name: | Tenofovir disoproxil fumarate |
| Molecular Structure: | |
 |
|
| Formula: | C19H30N5O10P.C4H4O4 |
| Molecular Weight: | 635.51 |
| Synonyms: | TDF;PMPA prodrug;Tenofovir Disoproxil Fumarate [USAN];9-((R)-2-((Bis(((isopropoxycarbonyl)oxy)methoxy)phosphinyl)methoxy)propyl)adenine, fumarate;201341-05-1;Bis(NeopentylOC)PMPA;Viread;GS 4331-05 (*1:1 Fumarate salt*);Viread (*1:1 Fumarate salt*);Truvada;Tenofovir DF;[[(2R)-1-(6-aminopurin-9-yl)propan-2-yl]oxymethyl-(propan-2-yloxycarbonyloxymethoxy)phosphoryl]oxymethyl propan-2-yl carbonate; |
|
|
|
|
|
Tenofovir disoproxil is an antiretroviral medication used to prevent and treat HIV/AIDS and to treat chronic hepatitis B.[1] The active substance is tenofovir, while tenofovir disoproxil is a prodrug that is used because of its better absorption in the gut.
The drug is on the World Health Organization’s List of Essential Medicines, the most important medications needed in a basic health system.[2] It is marketed by Gilead Sciences under the trade name Viread (as the fumarate, TDF).[3] As of 2015 the cost for a typical month of medication in the United States is more than 200 USD.[4]
Medical uses
- HIV-1 infection: Tenofovir is indicated in combination with other antiretroviral agents for the treatment of HIV-1 infection in adults and pediatric patients 2 years of age and older.[5] This indication is based on analyses of plasma HIV-1 RNA levels and CD4 cell counts in controlled studies of tenofovir in treatment-naive and treatment-experienced adults.
- Tenofovir is indicated for the treatment of chronic hepatitis B in adults and pediatric patients 12 years of age and older.[5][6]
HIV risk reduction
A Cochrane review examined the use of tenofovir for prevention of HIV before exposure. It found that both tenofovir alone and the tenofovir/emtricitabine combination decreased the risk of contracting HIV.[7]
The U. S. Centers for Disease Control and Prevention (CDC) conducted a study in partnership with the Thailand Ministry of Public Health to ascertain the effectiveness of providing people who inject drugs illicitly with daily doses of the antiretroviral drug tenofovir as a prevention measure. The results of the study were released in mid-June 2013 and revealed a 48.9%-reduced incidence of the virus among the group of subjects who received the drug, in comparison to the control group who received a placebo. The principal investigator of the study stated: “We now know that pre-exposure prophylaxis can be a potentially vital option for HIV prevention in people at very high risk for infection, whether through sexual transmission or injecting drug use.”[8]
Adverse effects
The most common side effects associated with tenofovir include nausea, vomiting, diarrhea, and asthenia. Less frequent side effects include hepatotoxicity, abdominal pain, and flatulence.[9] Tenofovir has also been implicated in causing renal toxicity, particularly at elevated concentrations.[10]
Tenofovir can cause acute renal failure, Fanconi syndrome, proteinuria, or tubular necrosis.[citation needed] These side effects are due to accumulation of the drug in proximal tubules.[citation needed] Tenofovir can interact with didanosine by increasing didanosine’s concentration.[citation needed] It also decreases the concentration of atazanavir sulfate.[citation needed]
Mechanism of action
Tenofovir is a defective adenosine nucleotide that selectively interferes with the action of reverse transcriptase, but only weakly interferes with mammalian DNA polymerases α, β, and mitochondrial DNA polymerase γ.[11] Tenofovir prevents the formation of the 5′ to 3′ phosphodiester linkage essential for DNA chain elongation. A phosphodiester bond cannot be formed because the tenofovir molecule lacks an —OH group on the 3′ carbon of its deoxyribose sugar.[11] Once incorporated into a growing DNA strand, tenofovir causes premature termination of DNA transcription. The drug is classified as a nucleotide analogue reverse transcriptase inhibitor (NRTI), that inhibits reverse transcriptase.[11] Reverse transcriptase is a crucial viral enzyme in retroviruses such as human immunodeficiency virus (HIV) and in hepatitis B virus infections.[5]
History
Tenofovir was initially synthesized by Antonín Holý at the Institute of Organic Chemistry and Biochemistry, Academy of Sciences of the Czech Republic in Prague. The patent[12] filed by Holý in 1984 makes no mention of the potential use of the compound for the treatment of HIV infection, which had only been discovered one year earlier.
In 1985, De Clercq and Holý described the activity of PMPA against HIV in cell culture.[13] Shortly thereafter, a collaboration with the biotechnology company Gilead Sciences led to the investigation of PMPA’s potential as a treatment for HIV infected patients. In 1997 researchers from Gilead and the University of California, San Francisco demonstrated that tenofovir exhibits anti-HIV effects in humans when dosed by subcutaneous injection.[14]
The initial form of tenofovir used in these studies had limited potential for widespread use because it was not absorbed when administered orally. A medicinal chemistry team at Gilead developed a modified version of tenofovir, tenofovir disoproxil.[15] This version of tenofovir is often referred to simply as “tenofovir”. In this version of the drug, the two negative charges of the tenofovir phosphonic acid group are masked, thus enhancing oral absorption.
Tenofovir disoproxil was approved by the U.S. FDA on October 26, 2001, for the treatment of HIV, and on August 11, 2008, for the treatment of chronic hepatitis B.[16][17]
Drug forms
Tenofovir disoproxil is a prodrug form of tenofovir. It is also marketed under the brand name Reviro by Dr. Reddy’s Laboratories. Tenofovir is also available in a fixed-dose combination with emtricitabine in a product with the brand name Truvada for once-a-day dosing. Efavirenz/emtricitabine/tenofovir disoproxil (brand name Atripla) — a fixed-dose triple combination of tenofovir, emtricitabine, and efavirenz, was approved by the FDA on 12 July 2006 and is now available, providing a single daily dose for the treatment of HIV.
Therapeutic drug monitoring
Tenofovir may be measured in plasma by liquid chromatography. Such testing is useful for monitoring therapy and to prevent drug accumulation and toxicity in people with kidney or liver problems.[18][19][20]
PATENT
http://www.google.com/patents/EP2545063A2?cl=en
Tenofovir Disoproxil is chemically known as 9-[-2-(R)-[[bis [[(isopropoxycarbonyl) oxy]methoxy] phosphinoyl]methoxy]propyl]-adenine, having the following structural formula-I.
Formula-I
Tenofovir is a highly potent antiviral agent, particularly for the therapy or prophylaxis of retroviral infections and belongs to a class of drugs called Nucleotide Reverse Transcriptase Inhibitors (NRTI) which blocks reverse transcriptase an enzyme crucial to viral production in HIV-infected people.
Tenofovir Disoproxil and its pharmaceutically acceptable salts were first disclosed in US 5,922,695. This patent discloses the preparation of Tenofovir Disoproxil by the esterification of Tenofovir with chloromethyl isopropyl carbonate using l-methyl-2- pyrrolidinone and triethylamine. In this patent Tenofovir Disoproxil is converted into its Fumarate salt without isolation. PCT Publication WO 2008007392 discloses process for the preparation of Tenofovir Disoproxil fumarate, wherein the isolated crystalline Tenofovir Disoproxil is converted into fumarate salt.
Tenofovir Disoproxil processes in the prior art are similar to process disclosed in product patent US 5,922,695. According to the prior art processes, Tenofovir Disoproxil fumarate obtained is having low yields and also show the presence of impurities such as dimers.
scheme- 1.
Tenofovir disoproxil chloromethyl isopropyl carbonate
Tenofovir disoproxil fumarate
Example 1 : Process for the preparation of Tenofovir Disoproxil fumarate
Toluene (500 ml) was added to the Tenofovir (100 gm) and stirred at room temperature. To this triethylamine (66.31 gm) was added, temperature was raised to 90° C and water was collected by azeotropic distillation at 110°C. Toluene was completely distilled under vacuum at same temperature. The reaction mixture was cooled to room temperature and to this a mixture of N-methyl pyrrolidine (300 gm), triethylamine (66.31 gm), Tetrabutyl ammonium bromide (52.8 gm) and trimethyl silyl chloride (17.8 gm) were added. The above reaction mixture was heated to 50-55 °C and was added slowly chloromethyl. isopropyl carbonate (CMIC) and maintained the reaction mixture at 50-55°C for 5 hrs. (Qualitative HPLC analysis shows about 85% product formation). The above reaction mixture was cooled to room temperature and filtered. The filtrate was added to DM water at 5-10°C and extract with dichloromethane. The combined dichloromethane layer was concentrated under vacuum and the crude was Co-distilled with cyclohexane and this crude was taken into isopropyl alcohol (1000 ml). To this fumaric acid (38 gm) was added and temperature was raised to 50° C. The reaction mixture was filtered and filtrate was cooled to 5-10° C. The obtained solid was filtered and washed with isopropyl alcohol. The compound was dried under vacuum to yield Tenofovir Disoproxil fumarate (140 gm).
Example-2 : Preparation of Tenofovir
N-methyl-2-pyrrolidone (25 gm) was taken along with toluene (150 gm) into a reaction vessel. l-(6-amino-purin-9-yl)-propan-2-ol (100 gm); toluene-4-sulfonic acid diethoxy phosphoryl methyl ester (200 gm) and magnesium ter-butoxide (71.2 gm) were also taken at’ 25-35°C. Temperature was raised to 74-75 °C and maintained for 5-6hrs. After completion of reaction, acetic acid (60 gm) was added and maintained for 1 hr. Later aq.HBr (332 gm) was taken and heated to 90-95 °C. After reaction completion, salts were filtered and filtrate was subjected to washings with water and extracted into methylene dichloride. Later pH was adjusted using CS lye below 10 °C. Tenofovir product was isolated using acetone.
Yield: 110 gm.
Example 3 : Preparation of Tenofovir disoproxil
(R)-9-[2-(phosphonomethoxy)propyl]adenine (25 gm), triethyl amine (25 ml) and cyclohexane (200 ml) were combined and heated to remove water and the solvent was distilled off under vacuum. The reaction mass was cooled to room temperature N-methyl pyrrolidinone (55 ml), triethyl amine (25 ml) and tetra butyl ammonium bromide(54 gms) were added to the reaction mixture. The reaction mass was heated to 50-60°C and chloromethyl isopropyl carbonate (65 gm) was added and maintained for 4-8 hrs at 50- 60°C and then cooled to 0°C. The reaction mass was diluted with chilled water or ice and precipitated solid product was filtered. The mother liquor was extracted with methylene chloride (150 ml). The methylene chloride layer was washed with water (200 ml). The filtered solid and the methylene chloride layer were combined and washed with water and the solvent was distilled under vacuum. Ethyl acetate was charged to the precipitated solid. The reaction mass was then cooled to 0-5 °C and maintained for 6 hrs. The solid was filtered and dried to produce Tenofovir disoproxil (45 gm).
CLIPS
The reaction of chloromethyl chloroformate (I) with isopropyl alcohol (II) by means of pyridine or triethylamine in ether gives the mixed carbonate (III), which is then condensed with (R)-PMPA (IV) by means of diisopropyl ethyl-amine in DMF.
| US 5922695; WO 9804569 |

CLIP 2
1) The protection of isobutyl D-(+)-lactate (I) with dihydropyran (DHP)/HCl in DMF gives the tetrahydropyranyloxy derivative (II), which is reduced with bis(2-methoxyethoxy)aluminum hydride in refluxing ether/ toluene yielding 2(R)-(tetrahydropyranyloxy)-1-propanol (III). The tosylation of (III) with tosyl chloride as usual affords the expected tosylate (VI), which is condensed with adenine (V) by means of Cs2CO3 in hot DMF, affording 9-[2(R)-(tetrahydropyranyloxy)propyl]adenine (VI). The deprotection of (VI) with sulfuric acid affords 9-[2(R)-hydroxypropyl]adenine (VII), which is N-benzoylated with benzoyl chloride/chlorotrimethylsilane in pyridine to give the benzamide (VIII), which is condensed with tosyl-oxymethylphosphonic acid diisopropyl ester (IX) by means of NaH in DMF to yield 9-[2(R)-(diisopropoxyphosphorylmethoxy)propyl]adenine (X). Finally, this compound is hydrolyzed by means of bromotrimethylsilane in acetonotrile.
2) The reaction of the previously described (R)-2-(2-tetrahydropyranyloxy)-1-propanol (III) with benzyl bromide (XI) by means of NaH in DMF, followed by a treatment with Dowex 50X, gives 1-benzyloxy-2(R)-propanol (XII), which is condensed with tosyloxymethylphosphonic acid diisopropyl ester (IX) by means of NaH in THF, yielding 2-benzyloxy-1(R)-methylethoxymethylphosphonic acid diisopropyl ester (XIII). The hydrogenolysis of (XIII) over Pd/C in methanol affords 2-hydroxy-1(R)-methylethoxymethylphosphonic acid diisopropyl ester (XIV), which is tosylated with tosyl chloride/dimethyl-aminopyridine in pyridine to give the expected tosylate (XV). The condensation of (XV) with adenine (VI) by means of Cs2CO3 in hot DMF yields 9-[2(R)-(diisopropoxyphosphorylmethoxy)propyl]adenine (X), which is finally hydrolyzed as before.

3) The catalytic hydrogenation of (S)-glycidol (XVI) over Pd/C gives the (R)-1,2-propanediol (XVII), which is esterified with diethyl carbonate (XVIII)/NaOEt, yielding the cyclic carbonate (XIX). The reaction of (XIX) with adenine (V) by means of NaOH in DMF affords 9-[2(R)-hydroxypropyl]adenine (VII), which is condensed with tosyloxymethylphosphonic acid diethyl ester (XX) by means of lithium tert-butoxide in THF, giving 9-[2(R)-(diethoxyphosphorylmethoxy)propyl]adenine (XXI). Finally, this compound is hydrolyzed with bromotrimethylsilane as before. Compound (XX) is obtained by reaction of diethyl phosphite (XXII) with paraformaldehyde, yielding hydroxy- methylphosphonic acid diethyl ester (XXIII), which is finally tosylated as usual.

References
- 1 “Viread”. The American Society of Health-System Pharmacists. Retrieved 31 July 2015.
- 2
- “WHO Model List of EssentialMedicines” (PDF). World Health Organization. October 2013. Retrieved 22 April 2014.
- 3
- Emau P, Jiang Y, Agy MB, et al. (2006). “Post-exposure prophylaxis for SIV revisited: Animal model for HIV infection”. AIDS Res Ther 3: 29. doi:10.1186/1742-6405-3-29. PMC 1687192. PMID 17132170.
- 4
- Hamilton, Richart (2015). Tarascon Pocket Pharmacopoeia 2015 Deluxe Lab-Coat Edition. Jones & Bartlett Learning. p. 66. ISBN 9781284057560.
- 5
- Gilead Sciences, Inc. Prescribing Information. Revised: November 2012.
- 6
- Guidelines for the prevention, care and treatment of persons with chronic hepatitis B infection, WHO, Publication details:Pages: 166 Publication date: March 2015 Languages: English ISBN 978 92 4 154905 9
- 7
- Okwundu CI, Uthman OA, Okoromah CAN (2012). “Antiretroviral pre-exposure prophylaxis (PrEP) for preventing HIV in high-risk individuals”. Cochrane Database Syst Rev 7 (7): CD007189. doi:10.1002/14651858.CD007189.pub3. PMID 22786505.
- 8
- Emma Bourke (14 June 2013). “Preventive drug could reduce HIV transmission among injecting drug users”. The Conversation Australia. The Conversation Media Group. Retrieved 17 June 2013.
- 9
- USPDI. Thompson. 2005. pp. 2741–2.
- 10
- “Viread Prescribing Guidelines” (PDF). U.S. Food and Drug Administration. March 2006. Archived from the original (PDF) on 2007-09-30. Retrieved 2007-02-12.
- 11
- Drugbank: Tenofovir
- 12
- “Patent US4808716 – 9-(phosponylmethoxyalkyl) adenines, the method of preparation and … – Google Patents”.
- 13
- A US 4724233 A, De Clercq, Erik; Antonin Holy & Ivan Rosenberg, “Therapeutical application of phosphonylmethoxyalkyl adenines”, published 1985-04-25
- 14
- Deeks SG, Barditch-Crovo P, Lietman PS, et al. (September 1998). “Safety, pharmacokinetics, and antiretroviral activity of intravenous 9-[2-(R)-(Phosphonomethoxy)propyl]adenine, a novel anti-human immunodeficiency virus (HIV) therapy, in HIV-infected adults”. Antimicrob. Agents Chemother. 42 (9): 2380–4. PMC 105837. PMID 9736567.
- 15
- “Patent US5977089 – Antiviral phosphonomethoxy nucleotide analogs having increased oral … – Google Patents”.
- 16
- FDA letter of approval (regarding treatment of hepatitis B)
- 17
- FDA Clears Viread for Hepatitis B
- 18
- Delahunty T, Bushman L, Robbins B, Fletcher CV (2009). “The simultaneous assay of tenofovir and emtricitabine in plasma using LC/MS/MS and isotopically labeled internal standards”. J. Chrom. B 877 (20–21): 1907–1914. doi:10.1016/j.jchromb.2009.05.029.
- 19
- Kearney BP, Yale K, Shah J, Zhong L, Flaherty JF (2006). “Pharmacokinetics and dosing recommendations of tenofovir disoproxil fumarate in hepatic or renal impairment”. Clin. Pharmacokinet. 45 (11): 1115–24. doi:10.2165/00003088-200645110-00005. PMID 17048975.
- 20
- R. Baselt, Disposition of Toxic Drugs and Chemicals in Man, 8th edition, Biomedical Publications, Foster City, California, 2008, pp. 1490–1492.
External links
| WO2008007392A2 | Jul 11, 2007 | Jan 17, 2008 | Matrix Lab Ltd | Process for the preparation of tenofovir |
| US5922695 | Jul 25, 1997 | Jul 13, 1999 | Gilead Sciences, Inc. | Antiviral phosphonomethyoxy nucleotide analogs having increased oral bioavarilability |
| WO2015051874A1 | Sep 22, 2014 | Apr 16, 2015 | Zentiva, K.S. | An improved process for the preparation of tenofovir disoproxil and pharmaceutically acceptable salts thereof |
| CN103360425A * | Apr 1, 2012 | Oct 23, 2013 | 安徽贝克联合制药有限公司 | Synthesis method of tenofovir disoproxil and fumarate thereof |
| CN103374038A * | Apr 11, 2012 | Oct 30, 2013 | 广州白云山制药股份有限公司广州白云山制药总厂 | Preparation method of antiviral medicine |
| CN103848868A * | Dec 4, 2012 | Jun 11, 2014 | 蚌埠丰原涂山制药有限公司 | Method for preparing tenofovir |
| CN103848869A * | Dec 4, 2012 | Jun 11, 2014 | 上海医药工业研究院 | Method for preparing tenofovir |
| CN103980319A * | Apr 24, 2014 | Aug 13, 2014 | 浙江外国语学院 | Preparation method of tenofovir |
| CN103980319B * | Apr 24, 2014 | Dec 2, 2015 | 浙江外国语学院 | 一种泰诺福韦的制备方法 |
| EP2860185A1 | Oct 9, 2013 | Apr 15, 2015 | Zentiva, k.s. | An improved process for the preparation of Tenofovir disoproxil and pharmaceutically acceptable salts thereof |
The chemical name of tenofovir disoproxil fumarate is 9-[(R)-2[[bis[[(isopropoxycarbonyl)oxy]methoxy]phosphinyl]methoxy]propyl]adenine fumarate (1:1). It has a molecular formula of C19H30N5O10P • C4H4O4 and a molecular weight of 635.52. It has the following structural formula:
 |
Tenofovir disoproxil fumarate is a white to off-white crystalline powder with a solubility of 13.4 mg/mL in distilled water at 25 °C. It has an octanol/phosphate buffer (pH 6.5) partition coefficient (log p) of 1.25 at 25 °C.
VIREAD is available as tablets or as an oral powder.
VIREAD tablets are for oral administration in strengths of 150, 200, 250, and 300 mg of tenofovir disoproxil fumarate, which are equivalent to 123, 163, 204 and 245 mg of tenofovir disoproxil, respectively. Each tablet contains the following inactive ingredients: croscarmellose sodium, lactose monohydrate, magnesium stearate, microcrystalline cellulose, and pregelatinized starch. The 300 mg tablets are coated with Opadry II Y-3010671-A, which contains FD&C blue #2 aluminum lake, hypromellose 2910, lactose monohydrate, titanium dioxide, and triacetin. The 150, 200, and 250 mg tablets are coated with Opadry II 32K-18425, which contains hypromellose 2910, lactose monohydrate, titanium dioxide, and triacetin.
VIREAD oral powder is available for oral administration as white, taste-masked, coated granules containing 40 mg of tenofovir disoproxil fumarate per gram of oral powder, which is equivalent to 33 mg of tenofovir disoproxil. The oral powder contains the following inactive ingredients: mannitol, hydroxypropyl cellulose, ethylcellulose, and silicon dioxide.
 |
|
| Systematic (IUPAC) name | |
|---|---|
|
Bis{[(isopropoxycarbonyl)oxy]methyl} ({[(2R)-1-(6-amino-9H-purin-9-yl)-2-propanyl]oxy}methyl)phosphonate
|
|
| Clinical data | |
| Trade names | Viread |
| AHFS/Drugs.com | monograph |
| Pregnancy category |
|
| Routes of administration |
Oral (tablets) |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 25% |
| Identifiers | |
| CAS Number | 201341-05-1 |
| ATC code | J05AF07 (WHO) |
| PubChem | CID 5481350 |
| ChemSpider | 4587262 |
| UNII | F4YU4LON7I |
| ChEBI | CHEBI:63717 |
| NIAID ChemDB | 080741 |
| Chemical data | |
| Formula | C19H30N5O10P |
| Molar mass | 519.443 g/mol |
 |
|
| Systematic (IUPAC) name | |
|---|---|
|
({[(2R)-1-(6-amino-9H-purin-9-yl)propan-2-yl]oxy}methyl)phosphonic acid
|
|
| Clinical data | |
| MedlinePlus | a602018 |
| Routes of administration |
In form of prodrugs |
| Pharmacokinetic data | |
| Protein binding | < 1% |
| Biological half-life | 17 hours |
| Excretion | Renal |
| Identifiers | |
| CAS Number | 147127-20-6 |
| ATC code | None |
| PubChem | CID 464205 |
| DrugBank | DB00300 |
| ChemSpider | 408154 |
| UNII | 99YXE507IL |
| KEGG | D06074 |
| ChEBI | CHEBI:63625 |
| ChEMBL | CHEMBL483 |
| Synonyms | 9-(2-Phosphonyl-methoxypropyly)adenine (PMPA) |
| Chemical data | |
| Formula | C9H14N5O4P |
| Molar mass | 287.213 g/mol |
///////

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO















































































