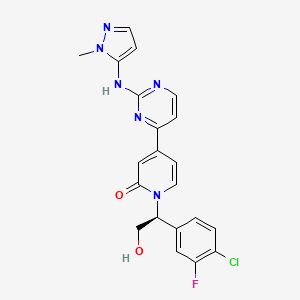
GDC 0994, Ravoxertinib
GDC 0994
GDC-0994; Ravoxertinib; 1453848-26-4; GDC0994; UNII-R6AXV96CRH; R6AXV96CRH, RG7842; RG-7842; RG 7842
CAS 1453848-26-4
1-[(1S)-1-(4-chloro-3-fluorophenyl)-2-hydroxyethyl]-4-[2-[(2-methylpyrazol-3-yl)amino]pyrimidin-4-yl]pyridin-2-one
| Molecular Formula: | C21H18ClFN6O2 |
|---|---|
| Molecular Weight: | 440.863 g/mol |
PHASE 1
Ravoxertinib also known as GDC-0994 and RG7842, is an orally available inhibitor of extracellular signal-regulated kinase (ERK), with potential antineoplastic activity. Upon oral administration, GDC-0994 inhibits both ERK phosphorylation and activation of ERK-mediated signal transduction pathways. This prevents ERK-dependent tumor cell proliferation and survival. The mitogen-activated protein kinase (MAPK)/ERK pathway is upregulated in a variety of tumor cell types and plays a key role in tumor cell proliferation, differentiation and survival.
GDC-0994 is an ERK inhibitor invented by Array under a collaboration agreement with Genentech. Array has received certain clinical milestones and is entitled to additional potential clinical and commercial milestones and royalties on product sales under the agreement. ERK is a key protein kinase in the RAS/RAF/MEK/ERK pathway, which regulates several key cellular activities including proliferation, differentiation, migration, survival and angiogenesis. Inappropriate activation of this pathway has been shown to occur in many cancers. GDC-0994 is currently advancing in a Phase 1 trial in patients with solid tumors.


| Applicants: | ARRAY BIOPHARMA INC. [US/US]; 3200 Walnut Street Boulder, Colorado 80301 (US). GENENTECH, INC. [US/US]; 1 DNA Way South San Francisco, California 94080-4990 (US) |
| Inventors: | BLAKE, James F.; (US). CHICARELLI, Mark Joseph; (US). GARREY, Rustam Ferdinand; (US). GAUDINO, John; (US). GRINA, Jonas; (US). MORENO, David A.; (US). MOHR, Peter J.; (US). REN, Li; (US). SCHWARZ, Jacob; (US). CHEN, Huifen; (US). ROBARGE, Kirk; (US). ZHOU, Aihe; (US) |
- OriginatorArray BioPharma
- DeveloperGenentech
- ClassAntineoplastics; Small molecules
- Mechanism of ActionExtracellular signal-regulated MAP kinase inhibitors; Mitogen activated protein kinase 3 inhibitors; Mitogen-activated protein kinase 1 inhibitors
- Phase ISolid tumours
Most Recent Events
- 29 Nov 2016Pharmacodynamics data from a preclinical trial in Solid tumours presented at the 28th EORTC-NCI-AACR Symposium on Molecular Targets and Cancer Therapeutics (EORTC-NCI-AACR-2016)
- 29 Nov 2016Adverse events, efficacy, pharmacokinetics and pharmacodynamics data from a phase I trial in Solid tumours presented at the 28th EORTC-NCI-AACR Symposium on Molecular Targets and Cancer Therapeutics
- 16 Jul 2016No recent reports of development identified for phase-I development in Solid-tumours(Late-stage disease, Monotherapy, Second-line therapy or greater) in USA
FREE FORM

(S)-1-(1-(4-Chloro-3-fluorophenyl)-2-hydroxyethyl)-4-(2-((1-methyl-1H-pyrazol-5-yl)amino)pyrimidin-4-yl)pyridin-2(1H)-one,
GDC-0994 benzenesulfonate salt

CAS 1817728-45-2, C21 H18 Cl F N6 O2 . C6 H6 O3 S
GDC-0994 as a light yellow solid,
mp 197.7 °C;

1H NMR (600 MHz, DMSO-d6): 9.93, (s, 1H), 8.65 (d, J = 5.2 Hz, 1H), 7.95 (d, J = 7.27 Hz, 1H), 7.63 (m, 2H), 7.62 (d, J = 1.5 Hz, 1H), 7.58 (t, J = 8.2 Hz, 1H), 7.55 (d, J = 5.2 Hz, 1H), 7.44 (dd, J = 10.6, 1.9 Hz, 1H), 7.33 (m, 3H), 7.18 (d, J = 2.0 Hz, 1H), 7.17 (d, J = 2.1 Hz, 1H), 6.90 (dd, J = 7.3, 2.1 Hz, 1H), 6.48 (d, J = 2.2 Hz, 1H), 5.99 (dd, J = 8.1, 5.5 Hz, 1H), 4.17 (dd, J = 11.9, 8.2 Hz, 1H), 4.05 (dd, J = 11.9, 5.5 Hz, 1H), 3.78 (s, 3H).

13C NMR (150 MHz, DMSO-d6): 161.60, 161.14, 160.02, 159.79, 157.02 (d, J = 245 Hz), 148.0, 146.49, 139.53 (d, J = 6.0 Hz), 139.04, 136.96, 136.39, 130.66, 128.42, 127.59, 125.38, 124.99 (d, J = 3.0 Hz), 118.72 (d, J = 18.0 Hz), 117.29, 116.05 (d, J = 22.5 Hz), 109.75, 102.79, 98.77, 60.64, 58.68, 35.29.

19F NMR (282 MHz, DMSO-d6) −115.86 (dd, J = 10.6, 7.8).
HRMS calcd for C21H18ClFN6O2 [M + H] 441.1242, found 441.1245.
PATENT
Example 39

(S)-1-(1-(4-chloro-3-fluorophenyl)-2-hydroxyethyl)-4-(2-((1-methyl-1H-pyrazol-5- yl)amino)pyrimidin-4-yl)pyridin-2(1H)-one
[00398] Step A: (S)-1-(2-(tert-Butyldimethylsiloxy)-1-(4-chloro-3-fluorophenyl)ethyl)-4-(2-(methylsulfonyl)pyrimidin-4-yl)pyridine-2(1H)-one (47 mg, 0.087 mmol), 2-methyl pyrazole-3 -amine (0.175 mmol, 2.0 equivalents) and anhydrous DMF (3.0 mL) were added to a 25 mL round bottomed flask equipped with a stirring bar. The flask was capped with a rubber septum and flushed with nitrogen. Under a blanket of nitrogen, sodium hydride (8.5 mg, 60% dispersion in mineral oil) was added in one portion. The flask was flushed with
nitrogen, capped and stirred at room temperature. The reaction progress was monitored by LCMS, and after 30 minutes, the starting material was consumed. The reaction mixture was quenched by the addition of water (0.5 mL) and ethyl acetate (15 mL). The contents of the round bottomed flask were transferred to a 125 mL separatory funnel, and the reaction flask was rinsed several times with additional ethyl acetate. Crude (S)-1-(2-((tert-butyldimethylsilyl)oxy)-1-(4-chloro-3-fluorophenyl)ethyl)-4-(2-((1-methyl-1H-pyrazol-5-yl)amino)pyrimidin-4-yl)pyridin-2(1H)-one was partitioned between ethyl acetate and water (80 mL/30 mL). The ethyl acetate layer was washed once with brine, dried (MgSO4), filtered and concentrated to give crude (S)-1-(2-((tert-butyldimethylsilyl)oxy)-1-(4-chloro-3-fluorophenyl)ethyl)-4-(2-((1-methyl-1H-pyrazol-5-yl)amino)pyrimidin-4-yl)pyridin-2(1H)-one. The crude was taken directly into the deprotection step.
[00399] Step B: Crude (S)-1-(2-((tert-butyldimethylsilyl)oxy)-1-(4-chloro-3-fluorophenyl)ethyl)-4-(2-((1-methyl-1H-pyrazol-5-yl)amino)pyrimidin-4-yl)pyridin-2(1H)-one (48 mg) was dissolved in ethyl acetate (4 mL) and treated dropwise slowly (over 2 minutes) with an ethyl acetate solution (1.0 mL, which had been saturated with HCl gas). The reaction stirred at room temperature for 15 minutes, after which time LCMS indicated complete consumption of the starting material. The reaction mixture was concentrated to an oily residue and purified by prep RP HPLC to yield (S)-1-(1-(4-chloro-3-fluorophenyl)-2-hydroxyethyl)-4-(2-((1-methyl-1H-pyrazol-5-yl)amino)pyrimidin-4-yl)pyridin-2(1H)-one (20.8 mg, 54.6% yield) as a lyophilized powder. 1H NMR (400 MHz, (CD3)2SO) δ 9.58 (s, 1H), 8.60 (d, J = 5.1 Hz, 1H), 7.91 (t, J = 9.0 Hz, 1H),7.58 (t, J = 8.1 Hz, 1H), 7.52-7.41 (m, 2H), 7.37 (d, J = 1.8 Hz, 1H), 7.14 (dd, J = 10.7,5.1 Hz 2H), 6.86 (dd, J = 7.3, 1.8 Hz, 1H), 6.27(d, J = 1.7 Hz, 1H), 5.97 (dd, J = 7.7, 5.7 Hz, 1H), 5.31(t, J = 5.2 Hz, 1H), 4.15 (m, 1H), 4.10-3.95 (m,1H), 3.69 (s, 3H); LCMS m/z 441 (M+H)+.
PAPER
Discovery of (S)-1-(1-(4-Chloro-3-fluorophenyl)-2-hydroxyethyl)-4-(2-((1-methyl-1H-pyrazol-5-yl)amino)pyrimidin-4-yl)pyridin-2(1H)-one (GDC-0994), an Extracellular Signal-Regulated Kinase 1/2 (ERK1/2) Inhibitor in Early Clinical Development

The extracellular signal-regulated kinases ERK1/2 represent an essential node within the RAS/RAF/MEK/ERK signaling cascade that is commonly activated by oncogenic mutations in BRAF or RAS or by upstream oncogenic signaling. While targeting upstream nodes with RAF and MEK inhibitors has proven effective clinically, resistance frequently develops through reactivation of the pathway. Simultaneous targeting of multiple nodes in the pathway, such as MEK and ERK, offers the prospect of enhanced efficacy as well as reduced potential for acquired resistance. Described herein is the discovery and characterization of GDC-0994 (22), an orally bioavailable small molecule inhibitor selective for ERK kinase activity.
PATENT
WO 2015154674
https://www.google.com/patents/WO2015154674A1?cl=pt
(a) contacting 4-bromo-1-chloro-2-fluorobenzene with a metallating agent in an aprotic organic solvent to afford an organomagnesium compound, which is reacted with 2-chloro-N-methoxy-N-methylacetamide to afford 2-chloro-1- (4-chloro-3-fluorophenyl) ethanone (II) ;
(b) contacting II with sodium formate and formic acid in aqueous ethanol to afford 1- (4-chloro-3-fluorophenyl) -2-hydroxyethanone (III)
(c) contacting III with a ketoreductase to afford (R) -1- (4-chloro-3-fluorophenyl) ethane-1, 2-diol (IV) ;
(d) contacting IV with a silyl chloride (Ra) 3SiCl and at least one base in a non-polar aprotic solvent to afford (V) , and subsequently adding sulfonylchloride RbS (O) 2Cl to afford VI, wherein Ra is independently in each occurrence C1-6 alkyl or phenyl and Rb is selected from C1-4 alkyl or phenyl, optionally substituted with 1 to 3 groups independently selected from C1-3 alkyl, halogen, nitro, cyano, or C1-3 alkoxy;
(e) contacting 4- (2- (methylsulfonyl) pyrimidin-4-yl) pyridin-2 (1H) -one (VII) with a strong base in an organic solvent and subsequently adding VI to afford XI;
(f) treating XI with an oxidizing agent to afford I;
(g) treating 1-methyl-1H-pyrazol-5-amine with a strong base in an aprotic solvent at reduced temperature and adding the compound of formula I to afford IX; and,
(h) contacting IX with a de-silylating agent to afford VIII.
PAPER
Development of a Practical Synthesis of ERK Inhibitor GDC-0994

The process development of a synthetic route to manufacture ERK inhibitor GDC-0994 on multikilogram scale is reported herein. The API was prepared as the corresponding benzenesulfonate salt in 7 steps and 41% overall yield. The synthetic route features a biocatalytic asymmetric ketone reduction, a regioselective pyridone SN2 reaction, and a safe and scalable tungstate-catalyzed sulfide oxidation. The end-game process involves a telescoped SNAr/desilylation/benzenesulfonate salt formation sequence. Finally, the development of the API crystallization allowed purging of process-related impurities, obtaining >99.5A% HPLC and >99% ee of the target molecule.
| Patent ID | Patent Title | Submitted Date | Granted Date |
|---|---|---|---|
| US2016136150 | COMPOUNDS AND COMPOSITIONS AS INHIBITORS OF MEK | 2015-11-13 | 2016-05-19 |
| US2016122316 | SERINE/THREONINE KINASE INHIBITORS | 2016-01-12 | 2016-05-05 |
| US2015111869 | USE OF A COMBINATION OF A MEK INHIBITOR AND AN ERK INHIBITOR FOR TREATMENT OF HYPERPROLIFERATIVE DISEASES | 2014-08-29 | 2015-04-23 |
| US2015051209 | COMPOUNDS AND COMPOSITIONS AS INHIBITORS OF MEK | 2014-08-05 | 2015-02-19 |
| US2014249127 | SERINE/THREONINE KINASE INHIBITORS | 2014-02-14 | 2014-09-04 |
| US8697715 | Serine/threonine kinase inhibitors | 2013-03-01 | 2014-04-15 |
///////////GDC 0994, Ravoxertinib, 1453848-26-4, GDC0994, UNII-R6AXV96CRH, R6AXV96CRH, RG7842, RG-7842, RG 7842, PHASE 1
CN1C(=CC=N1)NC2=NC=CC(=N2)C3=CC(=O)N(C=C3)C(CO)C4=CC(=C(C=C4)Cl)F
TROXACITABINE троксацитабин , تروكساسيتابين , 曲沙他滨 ,
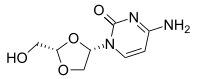
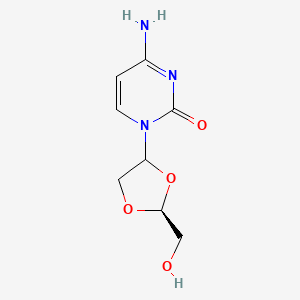
Troxacitabine
CAS 145918-75-8
- Molecular FormulaC8H11N3O4
- Average mass213.191 Da
Hmd-cytosine; NCGC00183848-01; Beta-L-Dioxolane-cytidine; 4-amino-1-[(2S)-2-(hydroxymethyl)-1,3-dioxolan-4-yl]pyrimidin-2-one; 2R(-)-cis-Hmd-cytosine, (-)-ODDC
4-amino-1-[(2S)-2-(hydroxymethyl)-1,3-dioxolan-4-yl]pyrimidin-2-one
Troxacitabine (brand name Troxatyl) is a nucleoside analogue with anticancer activity. Its use is being studied in patients with refractory lymphoproliferative diseases.[1]
Troxacitabine (brand name Troxatyl) is a nucleoside analogue with anticancer activity. Its use is being studied in patients with refractory lymphoproliferative diseases.
| Investigated for use/treatment in leukemia (myeloid). |
PATENT
https://www.google.com/patents/WO1992018517A1?cl=en
WO 9218517
| Inventors | Yung-Chi Cheng, Chung K. Chu, Hea O. Kim, Kirupathevy Shanmuganathan |
| Applicant | Yale University, The University Of Georgia Research Foundation, Inc. |
SYNTHESIS
WO 2016030335

PATENT
WO 2016030335
PATENT
MACHINE TRANSLATED FROM CHINESE, BEAWARE OF FUNNY NAMES
PATENT
PATENT
CN 105503838
PAPER
In vitro optimization of non-small cell lung cancer activity with troxacitabine, L-1,3-dioxolane-cytidine, prodrugs
Journal of medicinal chemistry (2007), 50, (9), 2249-53.

l-1,3-Dioxolane-cytidine, a potent anticancer agent against leukemia, has limited efficacy against solid tumors, perhaps due to its hydrophilicity. Herein, a library of prodrugs were synthesized to optimize in vitro antitumor activity against non-small cell lung cancer. N4-Substituted fatty acid amide prodrugs of 10−16 carbon chain length demonstrated significantly improved antitumor activity over l-1,3-dioxolane-cytidine. These in vitro results suggest that the in vivo therapeutic efficacy of l-1,3-dioxolane-cytidine against solid tumors may be improved with prodrug strategies.
PAPER
- Kim, Hea O.; Schinazi, Raymond F.; Shanmuganathan, Kirupathevy; Jeong, Lak S.; Beach, J. Warren; Nampalli, Satyanarayana; Cannon, Deborah L.; Chu, Chung K.
- From Journal of Medicinal Chemistry (1993), 36(5), 519-28.
PAPER
- Jin, Haolun; Tse, Allan Tse; Evans, Colleen A.; Mansour, Tarek S.; Beels, Christopher M.; Ravenscroft, Paul; Humber, David C.; Jones, Martin F.; Payne, Jeremy J.; Ramsay, Michael V. J.
- From Tetrahedron: Asymmetry (1993), 4(2), 211-14
PAPER
- Belleau, Bernard R.; Evans, Colleen A.; Tse, H. L. Allan; Jin, Haolun; Dixit, Dilip M.; Mansour, Tarek S.
- From Tetrahedron Letters (1992), 33(46), 6949-52.
PAPER
http://pubs.acs.org/doi/pdf/10.1021/jm00089a007
J. Med. Chem. 1992,35,1987-1995 Asymmetric Synthesis of 1,3-Dioxolane-Pyrimidine Nucleosides and Their Anti-HIV Activity
 References
References
- Jump up^ Vose, Julie M.; Panwalkar, Amit; Belanger, Robert; Coiffier, Bertrand; Baccarani, Michele; Gregory, Stephanie A.; Facon, Thierry; Fanin, Renato; Caballero, Dolores; Ben-Yehuda, Dina; Giles, Francis (2007). “A phase II multicenter study of troxacitabine in relapsed or refractory lymphoproliferative neoplasms or multiple myeloma”. Leukemia & Lymphoma. 48 (1): 39–45. doi:10.1080/10428190600909578.
- Lee CK, Rowinsky EK, Li J, Giles F, Moore MJ, Hidalgo M, Capparelli E, Jolivet J, Baker SD: Population pharmacokinetics of troxacitabine, a novel dioxolane nucleoside analogue. Clin Cancer Res. 2006 Apr 1;12(7 Pt 1):2158-65. [PubMed:16609029 ]
- Quintas-Cardama A, Cortes J: Evaluation of the L-stereoisomeric nucleoside analog troxacitabine for the treatment of acute myeloid leukemia. Expert Opin Investig Drugs. 2007 Apr;16(4):547-57. [PubMed:17371201 ]
- Swords R, Giles F: Troxacitabine in acute leukemia. Hematology. 2007 Jun;12(3):219-27. [PubMed:17558697 ]
- Orsolic N, Giles FJ, Gourdeau H, Golemovic M, Beran M, Cortes J, Freireich EJ, Kantarjian H, Verstovsek S: Troxacitabine and imatinib mesylate combination therapy of chronic myeloid leukaemia: preclinical evaluation. Br J Haematol. 2004 Mar;124(6):727-38. [PubMed:15009060 ]
- Boivin AJ, Gourdeau H, Momparler RL: Action of troxacitabine on cells transduced with human cytidine deaminase cDNA. Cancer Invest. 2004;22(1):25-9. [PubMed:15069761 ]
- Kim TE, Park SY, Hsu CH, Dutschman GE, Cheng YC: Synergistic antitumor activity of troxacitabine and camptothecin in selected human cancer cell lines. Mol Pharmacol. 2004 Aug;66(2):285-92. [PubMed:15266019 ]
| Patent ID | Patent Title | Submitted Date | Granted Date |
|---|---|---|---|
| US2013011392 | METHOD FOR ASSESSING THE ABILITY OF A PATIENT TO RESPOND TO OR BE SAFELY TREATED BY A NUCLEOSIDE ANALOG BASED-CHEMOTHERAPY | 2010-11-19 | 2013-01-10 |
| US7927613 | Pharmaceutical co-crystal compositions | 2003-09-11 | 2011-04-19 |
| US7790905 | Pharmaceutical propylene glycol solvate compositions | 2003-12-29 | 2010-09-07 |
 |
|
| Identifiers | |
|---|---|
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C8H11N3O4 |
| Molar mass | 213.19 g/mol |
| 3D model (Jmol) | |
//////////////TROXACITABINE, троксацитабин , تروكساسيتابين , 曲沙他滨 , Hmd-cytosineM, NCGC00183848-01, Beta-L-Dioxolane-cytidine, 2R(-)-cis-Hmd-cytosine, (-)-ODDC
Astellas Pharma Inc. new Glucokinase Activator, ASP ? for Type 2 Diabetes
ASP ?
(2R)-2-(4-cyclopropanesulfonyl-3-cyclopropylphenyl)-N-[5-(hydroxymethyl)pyrazin-2-yl]-3-[(R)-3-oxocyclopentyl]propanamide
- Molecular Weight, 483.58
- [α]D20 −128.7 (c 1.00, MeOH);
- 1H NMR (DMSO-d6, 400 MHz) δ 11.07 (s, 1H), 9.20 (d, J = 1.4 Hz, 1H), 8.41 (d, J = 1.4 Hz, 1H), 7.79 (d, J = 8.2 Hz, 1H), 7.41 (dd, J = 8.2, 1.8 Hz, 1H), 7.15 (d, J = 1.8 Hz, 1H), 5.52 (t, J = 5.7 Hz, 1H), 4.56 (d, J = 6.0 Hz, 2H), 4.04 (t, J = 7.6 Hz, 1H), 3.03–2.97 (m, 1H), 2.79 (tt, J = 8.4, 5.1 Hz, 1H), 2.25–1.81 (m, 8H), 1.53–1.47 (m, 1H), 1.17–1.12 (m, 2H), 1.08–1.02 (m, 4H), 0.89–0.84 (m, 2H);
- 13C NMR (DMSO-d6, 101 MHz) δ 218.5, 171.8, 152.1, 147.3, 145.7, 143.2, 140.3, 138.2, 134.8, 129.0, 125.3, 125.1, 62.5, 49.9, 44.4, 38.4, 38.2, 34.8, 32.1, 29.1, 12.4, 10.8, 10.7, 5.8;
- FTIR (ATR, cm–1) 3544, 3257, 1727, 1692, 1546, 1507, 1363, 1285, 1149, 719;
- HRMS (ESI) m/z [M + Na]+ calcd for C25H29N3O5S 506.1726, found 506.1747.
- Anal. Calcd for C25H29N3O5S: C, 62.09; H, 6.04; N, 8.69. Found: C, 61.79; H, 6.19; N, 8.62.

| Inventors | Masahiko Hayakawa, Yoshiyuki Kido, Takahiro Nigawara, Mitsuaki Okumura, Akira Kanai, Keisuke Maki, Nobuaki Amino |
| Applicant | Astellas Pharma Inc. |
Synthesis

contd…………………………..

PATENT
- PAPER
A Practical and Scalable Synthesis of a Glucokinase Activator via Diastereomeric Resolution and Palladium-Catalyzed C–N Coupling Reaction

Here we describe the research and development of a process for the practical synthesis of glucokinase activator (R)-1 as a potential drug for treating type-2 diabetes. The key intermediate, chiral α-arylpropionic acid (R)-2, was synthesized in high diastereomeric excess through the diasteromeric resolution of 7 without the need for a chiral resolving agent. The counterpart 2-aminopyrazine derivative 3 was synthesized using a palladium-catalyzed C–N coupling reaction. This efficient process was demonstrated at the pilot scale and yielded 19.0 kg of (R)-1. Moreover, an epimerization process to obtain (R)-7 from the undesired (S)-7 was developed.
Hayakawa, M.; Kido, Y.; Nigawara, T.; Okumura, M.; Kanai, A.; Maki, K.; Amino, N. PCT Int. Appl. WO/2009/091014 A1 20090723,2009.
https://www.astellas.com/en/ir/library/pdf/3q2017_rd_en.pdf
///////////1174229-89-0, ASTELLAS, Glucokinase Activator, TYPE 2 DIABETES, PRECLINICAL, ASP ?, WO 2009091014, Masahiko Hayakawa, Yoshiyuki Kido, Takahiro Nigawara, Mitsuaki Okumura, Akira Kanai, Keisuke Maki, Nobuaki Amino, WO2009091014,
O=C(Nc1cnc(cn1)CO)[C@H](C[C@@H]2CC(=O)CC2)c3ccc(c(c3)C4CC4)S(=O)(=O)C5CC5
FDA approves first treatment Noctiva (Desmopressin acetate) nasal spray for frequent urination at night due to overproduction of urine

Desmopressin acetate
March 3, 2017
The U.S. Food and Drug Administration today approved Noctiva (desmopressin acetate) nasal spray for adults who awaken at least two times per night to urinate due to a condition known as nocturnal polyuria (overproduction of urine during the night). Noctiva is the first FDA-approved treatment for this condition.
“Today’s approval provides adults who overproduce urine at night with the first FDA-approved therapeutic option to help reduce the number of times a night they wake up to urinate,” said Hylton V. Joffe, M.D., M.M.Sc., director of the Division of Bone, Reproductive, and Urologic Products in the FDA’s Center for Drug Evaluation and Research. “It is important to know that Noctiva is not approved for all causes of night-time urination, so patients should discuss their symptoms with their health care provider who can determine the underlying cause of the night-time urination and whether Noctiva is right for them.”
Nocturia (wakening at night to urinate) is a symptom that can be caused by a wide variety of conditions, such as congestive heart failure, poorly controlled diabetes mellitus, medications, or diseases of the bladder or prostate. Before considering Noctiva, health care providers should evaluate each patient for possible causes for the nocturia, and optimize the treatment of underlying conditions that may be contributing to the night-time urination. Because Noctiva is approved only for adults with nocturia caused by nocturnal polyuria, health care providers should confirm overproduction of urine at night with a 24-hour urine collection, if one has not been obtained previously. Health care providers should also be mindful of underlying conditions that can cause nocturia, but that make treatment with Noctiva unsafe, such as excessive drinking of fluids or symptomatic congestive heart failure.
Noctiva is taken daily, approximately 30 minutes before going to bed. It works by increasing the absorption of water through the kidneys, which leads to less urine production.
Noctiva’s efficacy was established in two 12-week, randomized, placebo-controlled trials in 1,045 patients 50 years of age and older with nocturia due to nocturnal polyuria. Although these trials showed a small reduction in the average number of night-time urinations with Noctiva compared to placebo, more patients treated with Noctiva were able to at least halve their number of night-time urinations, and patients treated with Noctiva had more nights with one or fewer night-time urinations.
Noctiva is being approved with a boxed warning and a Medication Guide because it can cause low sodium levels in the blood (hyponatremia). Severe hyponatremia can be life-threatening if it is not promptly diagnosed and treated, leading to seizures, coma, respiratory arrest or death. Health care providers should make sure the patient’s sodium level is normal before starting Noctiva, and should check sodium levels within one week and approximately one month after starting treatment and periodically thereafter. The lower Noctiva dose is recommended as the starting dose for those who may be at risk for hyponatremia, such as the elderly. Noctiva should not be used in patients at increased risk of severe hyponatremia, such as those with excessive fluid intake, those who have illnesses that can cause fluid or electrolyte imbalances, certain patients with kidney damage, and in those using certain medicines, known as loop diuretics or glucocorticoids.
Noctiva should also not be used in patients with symptomatic congestive heart failure or uncontrolled hypertension because fluid retention can worsen these underlying conditions. Use of Noctiva should be discontinued temporarily in patients with certain nasal conditions such as colds or allergies until those conditions have resolved.
Noctiva is also not recommended for the treatment of nocturia in pregnant women. Nocturia is usually related to normal changes in pregnancy that do not require treatment with Noctiva. Noctiva should not be used in children.
The most common side effects of Noctiva in clinical trials included nasal discomfort, cold symptoms (nasopharyngitis), nasal congestion, sneezing, high or increased blood pressure, back pain, nose bleeds, bronchitis and dizziness.
Although there are other FDA-approved medications that also contain desmopressin, none of those medications are approved to treat nocturia.
Noctiva is marketed by Milford, Pennsylvania-based Renaissance Lakewood, LLC for Serenity Pharmaceuticals, LLC.
1-(3-Mercaptopropionic acid)-8-D-arginine-vasopressin monoacetate (salt).

oxopentan-2-yl]-1-[4-(2-amino-2-oxoethyl)-7-(3-amino-3-oxopropyl)-10-benzyl-13-[(4-hydroxyphenyl)methyl]-3,6,9,12,15-pentaoxo-18,19-dithia-2,5,8,11,14-pentazacycloicosane-1-carbonyl]pyrrolidine-2-carboxamide;
Synonyms: 3-MERCAPTOPROPIONYL-TYR-PHE-GLN-ASN-CYS-PRO-D-ARG-GLY-NH2 ACETATE SALT;DDAVP ACETATE;[DEAMINO-CYS1,D-ARG8]-VASOPRESSIN ACETATE SALT;DESMOPRESSIN MONOACETATE;DESMORESSIN ACETATE;Mpr-Tyr-Phe-Gln-Asn-Cys-Pro-D-Arg-Gly-NH2(S-S:1-5);DESMOPRESSIN ACETATE;DESMOPRESSIN ACETATE SALT;
The Molecular Weight of Desmopressin Acetate(62288-83-9): 1129.27




Analytica Chimica Acta (2006), 572, (2), 197-204
Abstract
A monolithic column was prepared using l-phenylalanine as template and a covalent approach through the formation of Schiff base with o-phthalaldehyde (OPA). OPA, allylmercaptan, l-phenylalanine, and triethylamine were stirred at first, then methacrylic acid, 2-vinylpyridine, ethyleneglycol dimethacrylate, α,α-azobisisobutyronitrile, and 1-propanol were added to the reaction mixture. The resulting material was introduced into a capillary column. Following thermal polymerization, the template was then extracted with a mixture of HCl and methanol. The column was employed for the capillary electrochromatographic separation of oligopeptides. A capillary column of 75 (50) cm × 75 μm ID with a mobile phase of phosphate buffer (pH 7.0, 40 mM)/methanol (5%, v/v), an applied voltage of +15 kV, and detection at 214 nm, could baseline separate angiotensin I, angiotensin II, [Sar1, Thr8] angiotensin, oxytocin, vasopressin, tocinoic acid, β-casomorphin bovine, β-casomorphin human, and FMRF amide within 20 min. The separation behavior of the templated polymer was also compared with that of the non-templated polymer. As a result, it can be concluded that the electrochromatographic separation of this set of peptides was mediated by a combination of electrophoretic migration and chromatographic retention involving hydrophobic, hydrogen bonding, electrostatic as well as the Schiff base formation with OPA in the cavity of the templated polymer.
PATENT
CN 101372504
WO 2010119450
IN 2009CH00794
CN 103102395
CN 103467574
CN 105131079
CN 104761619
Desmopressin acetate is a structural analogue of natural arginine vasopressin, which is the result of two changes in the chemical structure of natural hormones. The structure is as follows:
M $ a-Tyr-Phe-Gln-Asn-C such as -Pro-D-Arg-GIy-N
Desmopressin acetate has a good hemostatic effect and does not produce side effects of pressurization. Mainly used to treat central diabetes insipidus, hemophilia and therapeutic control of bleeding and preoperative bleeding prevention. Good results and small side effects.
In the existing synthetic method of desmopressin acetate, liquid phase synthesis to produce more waste, the reaction time is long, each coupling an amino acid need to be purified, post-processing cumbersome, low yield, is not conducive to Industrial production.
Solid phase synthesis method, Chinese Patent CN 101372505, CN103992389 using Sieber Amide Resin or Rink Amide AM Resin one by one coupling to obtain linear peptide resin, and then solid-phase oxidation resin, cleavage and purification of desmopressin acetate. Chinese Patent CN103102395, CN102863513 Using Sieber Amide Resin or Rink AM Resin, linear peptide resin was obtained by coupling one by one, and liquid desulfurization was obtained after lysis to obtain desmopressin.
| Patent ID | Patent Title | Submitted Date | Granted Date |
|---|---|---|---|
| US8765152 | Pharmaceutical or neutraceutical formulation | 2010-02-25 | 2014-07-01 |
| Cited Patent | Filing date | Publication date | Applicant | Title | |
|---|---|---|---|---|---|
| US005726287 | Title not available | ||||
| US005990273 | Title not available | ||||
| US20060276626 | May 2, 2006 | Dec 7, 2006 | Avi Tovi | Methods for the production of peptide derivatives | |
| WO2004092202A1 | Apr 5, 2004 | Oct 28, 2004 | Novetide, Ltd. | Process for production of cyclic peptides |
| Citing Patent | Filing date | Publication date | Applicant | Title |
|---|---|---|---|---|
| CN102863513A * | Sep 12, 2012 | Jan 9, 2013 | 无锡市凯利药业有限公司 | Preparation method of desmopressin acetate |
Ramizol

1,3,5-Tris[(1E)-2′-(4′′-benzoic acid)vinyl]benzene] (Ramizol™)
TSB-007
| Allan James Mckinley, Thomas V Riley, Nigel Lengkeek, Scott Stewart, Ramiz Boulos | |
| Applicant | The University Of Western Australia |
1,3,5-Tris[(1E)-2′-(4′′-benzoic acid)vinyl]benzene] (Ramizol™) is a potent and non-toxic synthetic antimicrobial agent, and we now establish that it is also a potent inhibitor of reactive oxygen species (ROS) generation, with similar antioxidant activity to α-tocopherol (Vitamin E), which is a standard antioxidantdrug.
Ramizol, useful for treating bacterial infections such as Gram positive bacterial infection. Boulos & Cooper Pharmaceuticals could be seen to have ramizol in preclinical development for treating Clostridium difficile associated diseases. preparation of ramizol that was first described by the inventor Dr Ramiz Boulos, one of the company’s founding directors and CEO, in WO2011075766 as TSB-007 (claim 3, page 71) – said family of patenting having been originally assigned to the University of Western Australia and from whom Dr Boulos is reported to have acquired the rights to said intellectual property in late 2012 (ramizol having seemingly been previously being developed by the University with the name NAL-135B for treating Gram positive bacterial infections).
Professor Ramiz Boulos with a vial of Ramizol
A scientific paper released today in the Journal of Antibiotics presents the pre-clinical development of Ramizol®, a first generation drug belonging to a new class of styrylbenzene antibiotics with a novel mechanism of action.
The research was undertaken by Australian company Boulos & Cooper Pharmaceuticals in partnership with the University of South Australia, Flinders University, Eurofins Panlabs and Micromyx LLC. The study found that over 99.9% of the drug, administered orally, stays in the gastrointestinal tract where it can reach the bacteria in the colon at high enough concentrations to yield a therapeutic effect.
Chief Executive Officer of Boulos & Cooper Pharmaceuticals, Dr Ramiz Boulos, said “this new class of antibiotics has antioxidant properties and can be manufactured for a low cost; benefits that will be felt by the end-user”.
The new antibiotic has low frequency of resistance and shows promise as a monotherapy for the treatment of Clostridium difficile associated disease. Dr Boulos stated “we are very excited about these results given the unforgiving nature of Clostridium difficile infections”. He added “In a world where there are few treatment options, we are desperate for new antibiotics to fight intractable infections”.
The company expects to start Phase I clinical trials in 2017.
1,3,5-Tris[(1E)-20 -(400-benzoic acid)vinyl]benzene……………….recrystallised from THF/H2O and dried to give the triacid as a pale brown powder.
1 H NMR (500.1 MHz, d6-DMSO): d 7.49 (m, 6H, vinyl CH), 7.76 (d, J 8.5, 6H, ArH), 7.88 (s, 3H, core ArH), 7.98 (d, J 8.5, 6H, ArH);
13C NMR (125.8 MHz, d6-DMSO): d [ppm] 125.0, 126.5, 128.4, 129.7, 129.9, 130.50, 137.6, 141.3, 167.1;
IR (KBr): n [cm1 ] 3067, 3026, 1684 (nC¼O), 1604, 1566, 1420, 1384, 1312, 1286, 1179;
HR-EIþ-MS: C33H24O6 requires 516.1573 amu, found 516.1564;
EIþ-MS: MI ¼ C33H24O6; m/z: 516.1 (100%) ¼ MIþ, 472.1 (11.3%) ¼ [MI CO2] þ.
The Synthesis of Fluorescent DNA Intercalator Precursors through Efficient Multiple Heck Reactions
Nigel A. Lengkeek A , Ramiz A. Boulos A , Allan J. McKinley A , Thomas V. Riley C , Boris Martinac B and Scott G. Stewart A D
A M313, Chemistry, School of Biomedical, Biomolecular and Chemical Science, University of Western Australia, 35 Stirling Highway, Crawley, WA, 6009, Australia.
B Victor Chang Cardiac Research Institute, Lowy Packer Building, 405 Liverpool Street, Darlinghurst, Sydney, NSW 2010, Australia.
C M502, Microbiology and Immunology, School of Biomedical, Biomolecular and Chemical Sciences, The University of Western Australia, 35 Stirling Hwy, Nedlands, WA 6009, Australia.
D Corresponding author. Email: sgs@cyllene.uwa.edu.au
Australian Journal of Chemistry 64(3) 316-323 http://dx.doi.org/10.1071/CH10374
PATENT
PATENT
WO-2017027933
Compounds with antimicrobial properties have attracted great interest in recent times as a result of an increase in the prevalence of infections caused by Gram-positive bacteria, resulting in serious or fatal diseases. Furthermore, the regular use of broad spectrum antibiotic formulas has led to the increased occurrence of bacterial strains resistant to some antimicrobial formulations.
Novel antimicrobial compounds have the potential to be highly effective against these types of treatment-resistant bacteria. The pathogens, having not previously been exposed to the antimicrobial formulation, may have little to no resistance to the treatment.
International patent application WO 2012/075766 describes a series of novel aryl compounds and their use as antimicrobials to treat bacterial infections or diseases. The chemical synthesis of a therapeutic drug has a direct effect on its cost, dosing regimens and popularity. Drugs with complicated or expensive chemical synthesis will find it challenging to reach the market, notwithstanding their efficacy. Further, syntheses amenable to application at commercial scales are highly advantageous. The development of an efficient and large-scale synthesis of a therapeutic drug is critical for its drug developmental pathway, and highly commercially advantageous.
1H NMR PREDICT


13C NMR PREDICT


REFERENCES
N. A. Lengkeek, R. A. Boulos, A. J. McKinley, T. V. Riley, B. Martinac and S. G. Stewart, Aust. J. Chem., 2011, 64, 316–323
http://pubs.rsc.org/en/content/articlehtml/2013/ra/c3ra40658j#cit11
/////////////Ramizol, PHASE 1, TSB-007
OC(=O)c4ccc(/C=C/c3cc(/C=C/c1ccc(cc1)C(=O)O)cc(/C=C/c2ccc(cc2)C(=O)O)c3)cc4
FDA approves Odactra for house dust mite allergies

March 1, 2017
Release
The U.S. Food and Drug Administration today approved Odactra, the first allergen extract to be administered under the tongue (sublingually) to treat house dust mite (HDM)-induced nasal inflammation (allergic rhinitis), with or without eye inflammation (conjunctivitis), in people 18 through 65 years of age.
“House dust mite allergic disease can negatively impact a person’s quality of life,” said Peter Marks, M.D., Ph.D., director of the FDA’s Center for Biologics Evaluation and Research. “The approval of Odactra provides patients an alternative treatment to allergy shots to help address their symptoms.”
House dust mite allergies are a reaction to tiny bugs that are commonly found in house dust. Dust mites, close relatives of ticks and spiders, are too small to be seen without a microscope. They are found in bedding, upholstered furniture and carpeting. Individuals with house dust mite allergies may experience a cough, runny nose, nasal itching, nasal congestion, sneezing, and itchy and watery eyes.
Odactra exposes patients to house dust mite allergens, gradually training the immune system in order to reduce the frequency and severity of nasal and eye allergy symptoms. It is a once-daily tablet, taken year round, that rapidly dissolves after it is placed under the tongue. The first dose is taken under the supervision of a health care professional with experience in the diagnosis and treatment of allergic diseases. The patient is to be observed for at least 30 minutes for potential adverse reactions. Provided the first dose is well tolerated, patients can then take Odactra at home. It can take about eight to 14 weeks of daily dosing after initiation of Odactra for the patient to begin to experience a noticeable benefit.
The safety and efficacy of Odactra was evaluated in studies conducted in the United States, Canada and Europe, involving approximately 2,500 people. Some participants received Odactra, while others received a placebo pill. Participants reported their symptoms and the need to use symptom-relieving allergy medications. During treatment, participants taking Odactra experienced a 16 to 18 percent reduction in symptoms and the need for additional medications compared to those who received a placebo.
The most commonly reported adverse reactions were nausea, itching in the ears and mouth, and swelling of the lips and tongue. The prescribing information includes a boxed warning that severe allergic reactions, some of which can be life-threatening, can occur. As with other FDA-approved allergen extracts administered sublingually, patients receiving Odactra should be prescribed auto-injectable epinephrine. Odactra also has a Medication Guide for distribution to the patient.
Odactra is manufactured for Merck, Sharp & Dohme Corp., (a subsidiary of Merck and Co., Inc., Whitehouse Station, N.J.) by Catalent Pharma Solutions Limited, United Kingdom.
(sublingually) to treat house dust mite (HDM)-induced nasal inflammation (allergic rhinitis), with or without eye inflammation (conjunctivitis), in people 18 through 65 years of age
/////////////Odactra, Merck, Sharp & Dohme Corp, Catalent Pharma Solutions Limited, United Kingdom, FDA 2017, approves, house dust mite allergies
Award for me, 100 Most Impactful Health care Leaders, Global listing

At award function for my award “100 Most Impactful Health care Leaders Global listing”, conferred on me at Taj lands end, Mumbai, India on 14 Feb 2014 by World Health Wellness congress and awards
FDA approves Xermelo (telotristat ethyl) for carcinoid syndrome diarrhea
Telotristat ethyl
Molecular Formula, C27-H26-Cl-F3-N6-O3,
Molecular Weight, 574.9884,
RN: 1033805-22-9
UNII: 8G388563M
LX 1032
(2S)-2-Amino-3-[4-[2-amino-6-[[(1R)-1-[4-chloro-2-(3-methylpyrazol-1-yl)phenyl]-2,2,2-trifluoroethyl]oxy]pyrimidin-4-yl]phenyl]propionic acid ethyl ester
Ethyl-4-(2-amino-6-{(1R)-1-[4-chlor-2-(3-methyl-1H-pyrazol-1-yl)phenyl]-2,2,2-trifluorethoxy}-4-pyrimidinyl)-L-phenylalaninat

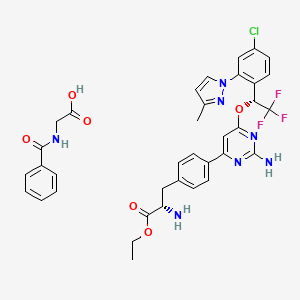
CAS: 1137608-69-5 (etiprate), LX 1606
Chemical Formula: C36H35ClF3N7O6
Molecular Weight: 754.16
- LX 1032 hippurate
- LX 1606


Carcinoid syndrome is a cluster of symptoms sometimes seen in people with carcinoid tumors. These tumors are rare, and often slow-growing. Most carcinoid tumors are found in the gastrointestinal tract. Carcinoid syndrome occurs in less than 10 percent of patients with carcinoid tumors, usually after the tumor has spread to the liver. The tumors in these patients release excess amounts of the hormone serotonin, resulting in diarrhea. Complications of uncontrolled diarrhea include weight loss, malnutrition, dehydration, and electrolyte imbalance.
“Today’s approval will provide patients whose carcinoid syndrome diarrhea is not adequately controlled with another treatment option,” said Julie Beitz, M.D., director of the Office of Drug Evaluation III in the FDA’s Center for Drug Evaluation and Research.
Xermelo, in a regimen with SSA therapy, is approved in tablet form to be taken orally three times daily with food. Xermelo inhibits the production of serotonin by carcinoid tumors and reduces the frequency of carcinoid syndrome diarrhea.
The safety and efficacy of Xermelo were established in a 12-week, double-blind, placebo-controlled trial in 90 adult participants with well-differentiated metastatic neuroendocrine tumors and carcinoid syndrome diarrhea. These patients were having between four to 12 daily bowel movements despite the use of SSA at a stable dose for at least three months. Participants remained on their SSA treatment, and were randomized to add placebo or treatment with Xermelo three times daily. Those receiving Xermelo added on to their SSA treatment experienced a greater reduction in average bowel movement frequency than those on SSA and placebo. Specifically, 33 percent of participants randomized to add Xermelo on to SSA experienced an average reduction of two bowel movements per day compared to 4 percent of patients randomized to add placebo on to SSA.
The most common side effects of Xermelo include nausea, headache, increased levels of the liver enzyme gamma-glutamyl transferase, depression, accumulation of fluid causing swelling (peripheral edema), flatulence, decreased appetite and fever. Xermelo may cause constipation, and the risk of developing constipation may be increased in patients whose bowel movement frequency is less than four bowel movements per day. Patients treated with a higher than recommended dosage of Xermelo developed severe constipation in clinical trials. One patient required hospitalization and two other patients developed complications of either intestinal perforation or intestinal obstruction. Patients should be monitored for severe constipation. If a patient experiences severe constipation or severe, persistent or worsening abdominal pain, they should discontinue Xermelo and contact their healthcare provider.
The FDA granted this application fast track designation and priority review. The drug also received orphan drug designation, which provides incentives to assist and encourage the development of drugs for rare diseases.
Xermelo is manufactured by Woodlands, Texas-based Lexicon Pharmaceuticals, Inc.
SYNTHESIS…….WO 2011100285

5.67. Synthesis of (S)-2-Amino-3-[4-(2-amino-6-{R-l-[4-chloro-2-(3-methyl-pyrazol-l-yll- phenyll-2,2,2-trifluoro-ethoxy)-pyrimidin-4-yl)-phenyll-propionic acid ethyl ester

The title compound was prepared stepwise, as described below:
Step 1: Synthesis of l-(2-bromo-4-chloro-phenyl)-2,2,2-trifluoro-ethanone. To a 500 ml 2 necked RB flask containing anhydrous methanol (300 ml) was added thionyl chloride (29.2 ml, 400 mmol) dropwise at 0-5°C (ice water bath) over 10 minutes. The ice water bath was removed, and 2-bromo-4-chloro-benzoic acid (25 g, 106 mmol) was added. The mixture was heated to mild reflux for 12h. Progress of the reaction was monitored by TLC and LCMS. After completion of the reaction, the reaction mixture was concentrated. Crude product was dissolved in dichloromethane (DCM, 250 ml), washed with water (50 ml), sat. aq. NaHC03 (50 ml), brine (50 ml), dried over sodium sulfate, and concentrated to give the 2- bromo-4-chloro-benzoic acid methyl ester (26 g, 99 %), which was directly used in the following step.
2-Bromo-4-chloro-benzoic acid methyl ester (12.4 g, 50 mmol) in toluene (200 ml) was cooled to -70°C, and trifluoromethyl trimethyl silane (13 ml, 70 mmol) was added.
Tetrabutylamonium fluoride (1M, 2.5 ml) was added dropwise, and the mixture was allowed to warm to room temperature over 4h, after which it was stirred for 10 hours at room temperature. The reaction mixture was concentrated to give the crude [l-(2-bromo-4-chloro-phenyl)-2,2,2-trifluoro-l-methoxy-ethoxy]-trimethyl-silane. The crude intermediate was dissolved in methanol (100 ml) and 6N HCI (100 ml) was added. The mixture was kept at 45-50°C for 12h. Methanol was removed, and the crude was extracted with dichloromethane (200 ml). The combined DCM layer was washed with water (50 ml), NaHC03 (50 ml), brine (50 ml), and dried over sodium sulfate. Removal of solvent gave a crude product, which was purified by ISCO column chromatography, using 1-2% ethyl acetate in hexane as solvent, to afford l-(2-bromo-4-chloro-phenyl)-2,2,2-trifluoro-ethanone (10 g, 70%). !H-NMR (300 MHz, CDC ): δ (ppm) 7.50 (d,lH), 7.65(d,lH), 7.80(s,lH).
Step 2: Synthesis of R-l-(2-bromo-4-chloro-phenyl)-2,2,2-trifluoro-ethanol. To catechol borane (1M in THF 280 ml, 280 mmol) in a 2L 3-necked RB flask was added S-2-methyl-CBS oxazaborolidine (7.76 g, 28 mmol) under nitrogen, and the resulting mixture was stirred at room temperature for 20 min. The reaction mixture was cooled to -78°C (dry ice/acetone bath), and 1-(2-bromo-4-chloro-phenyl)-2,2,2-trifluoro-ethanone (40 g, 139 mmol) in THF (400 ml) was added dropwise over 2 hours. The reaction mixture was allowed to warm to -36°C, and was stirred at that temperature for 24 hours, and further stirred at -32 °C for another 24h. 3N NaOH (250 ml) was added, and the cooling bath was replaced by ice-water bath. Then 30 % hydrogen peroxide in water (250 ml) was added dropwise over 30 minutes. The ice water bath was removed, and the mixture was stirred at room temperature for 4 hours. The organic layer was separated, concentrated and re-dissolved in ether (200 ml). The aqueous layer was extracted with ether (2 x 200 ml). The combined organic layers were washed with IN aq. NaOH (4 x 100 ml), brine, and dried over sodium sulfate. Removal of solvent gave crude product which was purified by column chromatography using 2 to 5% ethyl acetate in hexane as solvent to give desired alcohol 36.2 g (90 %, e.e. >95%). The alcohol (36.2 g) was crystallized from hexane (80 ml) to obtain R-l-(2-bromo-4-chloro-phenyl)-2,2,2-trifluoro-ethanol 28.2 g (70 %; 99-100 % e.e.). !H-NMR (400 MHz, CDCIs) δ (ppm) 5.48 (m, 1H), 7.40 (d, 1H), 7.61 (d, 2H).
Step 3: Synthesis of R-l-[4-chloro-2-(3-methyl-pyrazol-l-yl)-phenyll-2.2.2-trifluoro-ethanol. R-l-(2-bromo-4-chloro-phenyl)-2,2,2-trifluoro-ethanol (15.65 g, 54.06 mmol), 3-methylpyrazole (5.33 g, 65 mmol), Cul (2.06 g, 10.8 mmol), 2CO3 (15.7 g, 113.5 mmol), (lR,2R)-N,N’-dimethyl-cyclohexane-l,2-diamine (1.54 g, 10.8 mmol) and toluene (80 ml) were combined in a 250 ml pressure tube and heated to 130°C (oil bath temperature) for 12 hours. The reaction mixture was diluted with ethyl acetate and washed with H2O (4 x 100 ml), brine, and dried over sodium sulfate. Removal of solvent gave a crude product, which was purified by ISCO column chromatography using 5-10 % ethyl acetate in hexane as solvent to get R-l-[4-chloro-2-(3-methyl-pyrazol-l-yl)-phenyl]-2,2,2-trifluoro-ethanol (13.5 g; 86 %). i-H-NMR (400 MHz, CDC ): δ (ppm) 2.30(s, 3H), 4.90(m, 1H), 6.20(s, 1H), 6.84(d, 1H), 7.20(s, 1H), 7.30(d, 1H), 7.50(d, 1H).
Step 4: Synthesis of (S)-2-Amino-3- 4-(2-amino-6-fR-l-[4-chloro-2-(3-methyl-pyrazol-l-yl)-phenyll^^^-trifluoro-ethoxyl-pyrimidin^-yll-phenvD-propionic acid ethyl ester. R-l-[4-chloro-2-(3-methyl-pyrazol-l-yl)-phenyl]-2,2,2-trifluoro-ethanol (17.78 g, 61.17 mmol), (S)-3-[4-(2-amino-6-chloro-pyrimidine-4-yl)-phenyl]-2-tert-butoxycarbonylamino-propionic acid (20.03 g, 51 mmol), 1,4-dioxane (250 ml), and CS2CO3 (79.5 g, 244 mmol) were combined in a 3-necked 500 ml RB flask and heated to 100°C (oil bath temperature) for 12-24 hours. The progress of reaction was monitored by LCMS. After the completion of the reaction, the mixture was cooled to 60°C, and water (250 ml) and THF (400 ml) were added. The organic layer was separated and washed with brine (150 ml). The solvent was removed to give crude BOC protected product, which was taken in THF (400 ml), 3N HCI (200 ml). The mixture was heated at 35-40 °C for 12 hours. THF was removed in vacuo. The remaining aqueous layer was extracted with isopropyl acetate (2x 100 ml) and concentrated separately to recover the unreacted alcohol (3.5 g). Traces of remaining organic solvent were removed from the aqueous fraction under vacuum.
To a 1L beaker equipped with a temperature controller and pH meter, was added H3PO4 (40 ml, 85 % in water) and water (300 ml) then 50 % NaOH in water to adjust pH to 6.15. The temperature was raised to 58 °C and the above acidic aqueous solution was added dropwise into the buffer with simultaneous addition of 50 % NaOH solution in water so that the pH was maintained between 6.1 to 6.3. Upon completion of addition, precipitated solid was filtered and washed with hot water (50-60°C) (2 x 200 ml) and dried to give crude (S)-2-amino-3-[4-(2-amino-6-[R-l-[4-chloro-2-(3-methyl-pyrazol-l-yl)-phenyl]-2,2,2-trifluoro-ethoxy}-pyrimidin-4-yl)-phenyl}^ propionic acid (26.8 g; 95 %). LCMS and HPLC analysis indicated the compound purity was about 96-97 %.
To anhydrous ethanol (400 ml) was added SOC (22 ml, 306 mmol) dropwise at 0-5°C.
Crude acid (26.8 ) from the above reaction was added. The ice water bath was removed, and the reaction mixture was heated at 40-45°C for 6-12 hours. After the reaction was completed, ethanol was removed in vacuo. To the residue was added ice water (300 ml), and extracted with isopropyl acetate (2 x 100 ml). The aqueous solution was neutralized with saturated Na2C03 to adjust the pH to 6.5. The solution was extracted with ethyl acetate (2 x 300 ml). The combined ethyl acetate layer was washed with brine and concentrated to give 24 g of crude ester (HPLC purity of 96-97 %). The crude ester was then purified by ISCO column chromatography using 5 % ethanol in DCM as solvent to give (S)-2-amino-3-[4-(2-amino-6-{R-l-[4-chloro-2-(3-methyl-pyrazol-l-yl)-phenyl]-2,2,2-trifluoro-ethoxy}-pyrimidin-4-yl)-phenyl}-propionic acid ethyl ester (20.5g; 70 %; HPLC purity of 98 %). LCMS M+l = 575. !H-NMR (400 MHz, CDsOD): δ (ppm) 1.10 (t, 3H), 2.25 (s, 3H), 2.85 (m, 2H), 3.65 (m, IH), 4.00 (q, 2H), 6.35 (s, IH), 6.60 (s, IH), 6.90 (m, IH), 7.18 (d, 2H), 7.45 (m, 2H), 7.70 (d, IH), 7.85 (m, 3H).
SYNTHESIS OF INTERMEDIATE
WO 2009048864

https://google.com/patents/WO2009048864A1?cl=en
6.15. Preparation of 6SV3-(4-(2-Amino-6-chloropyrimidin-4-yl)phenyl)-2- (fert-butoxycarbonylamino)propanoic Acid Using the Lithium Salt of (S)-2-(te^-butoxycarbonylamino)-3-(4-(4,4,5,5-tetramethyl-l,3,2- dioxaborolan-2-yl)phenyl)propanoic Acid
During preparation of compound 7, the isolation of the free acid can be optionally omitted. Thus, an aqueous solution of the lithium salt of compound 7 in 100 ml water, prepared from 5.0 g of Boc-Tyr-OMe (4, 17 mmol), was mixed 2-amino-4,6- dichloropyrimidine (3.3 g, 1.2 eq), potassium bicarbonate (5.0 g, 3 eq), bis(triphenylphosphine)palladium(II) dichloride (60 mg, 0.5 mol%), and 100 ml ethanol. The resulting mixture was heated at 700C for 5 hours. Additional 2-amino-4,6- dichloropyrimidine (1.1 g, 0.4 eq) was added and heating was continued at 7O0C for an additional 2 hours. HPLC analysis showed about 94% conversion. Upon cooling and filtration, the filtrate was analyzed by HPLC against a standard solution of compound 8. The assay indicated 3.9 g compound 8 was contained in the solution (59% yield from compound 4).
6.16. Alternative Procedure for Preparation of (S)-3-(4-f2-Amino-6- chloropyrimidin-4-yl)phenyl)-2-(fe^-butoxycarbonylamino)propanoic Acid Using Potassium Carbonate as Base
The boronic acid compound 11 (Ryscor Science, Inc., North Carolina, 1.0 g, 4.8 mmol) and potassium carbonate (1.32 g, 2 eq) were mixed in aqueous ethanol (15 ml ethanol and 8 ml water). Di-ter£-butyldicarbonate (1.25 g, 1.2 eq) was added in one portion. After 30 minutes agitation at room temperature, HPLC analysis showed complete consumption of the starting compound 11. The 2-amino-4,6- dichloropyrimidine (1.18 g, 1.5 eq) and the catalyst bis(triphenylphosphine)palladium(II) dichloride (34 mg, 1 mol%) were added and the resulting mixture was heated at 65-700C for 3 hours. HPLC analysis showed complete consumption of compound 12. After concentration and filtration, HPLC analysis of the resulting aqueous solution against a standard solution of compound 8 showed 1.26 g compound 8 (67% yield).
6.17. Alternative procedure for preparation of (5)-3-(4-(2-Amino-6-
The boronic acid compound 11 (10 g, 48 mmol) and potassium bicarbonate (14.4 g, 3 eq) were mixed in aqueous ethanol (250 ml ethanol and 50 ml water). Oi-tert- butyldicarbonate (12.5 g, 1.2 eq) was added in one portion. HPLC analysis indicated that the reaction was not complete after overnight stirring at room temperature. Potassium carbonate (6.6 g, 1.0 eq) and additional di-te/t-butyldicarbonate (3.1 g, 0.3 eq) were added. After 2.5 hours agitation at room temperature, HPLC analysis showed complete consumption of the starting compound 11. The 2-amino-4,6-dichloropyrimidine (11.8 g, 1.5 eq) and the catalyst bis(triphenylphosphine)-palladium(II) dichloride (0.34 g, 1 mol%” were added and the resulting mixture was heated at 75-8O0C for 2 hours. HPLC analysis showed complete consumption of compound 12. The mixture was concentrated under reduced pressure and filtered. The filtrate was washed with ethyl acetate (200 ml) and diluted with 3 : 1 THF/MTBE (120 ml). This mixture was acidified to pH about 2.4 by 6 N hydrochloric acid. The organic layer was washed with brine and concentrated under reduced pressure. The residue was precipitated in isopropanol, filtered, and dried at 500C under vacuum to give compound 8 as an off-white solid (9.0 g, 48% yield). Purity: 92.9% by HPLC analysis. Concentration of the mother liquor yielded and additional 2.2 g off-white powder (12% yield). Purity: 93.6% by HPLC analysis
PATENT
https://www.google.com/patents/WO2013059146A1?cl=en
This invention is directed to solid pharmaceutical dosage forms in which an active pharmaceutical ingredient (API) is (S)-ethyl 2-amino-3-(4-(2-amino-6-((R)-l-(4-chloro-2-(3- methyl-lH-pyrazol-l-yl)phenyl)-2,2,2-trifluoroethoxy)pyrimidin-4-yl)phenyl)propanoate
(telotristat):
or a pharmaceutically acceptable salt thereof. The compound, its salts and crystalline forms can be obtained by methods known in the art. See, e.g., U.S. patent no. 7,709,493.
PATENT
http://www.google.co.in/patents/WO2008073933A2?cl=en
6.19. Synthesis of (S)-2-Amino-3-r4-q-amino-6-{R-l-r4-chloro-2-(3-methyl- Pyrazol-l-yl)-phenyll-2,2,2-trifluoro-ethoxy}-pyrimidin-4-yl)-phenyll- propionic acid ethyl ester
The title compound was prepared stepwise, as described below: Step 1 : Synthesis of l-(2-bromo-4-chloro-phenyl)-2,2,2-trifluoro-ethanone. To a 500 ml 2 necked RB flask containing anhydrous methanol (300 ml) was added thionyl chloride (29.2 ml, 400 mmol) dropwise at 0-50C (ice water bath) over 10 min. The ice water bath was removed, and 2-bromo-4-chloro-benzoic acid (25 g, 106 mmol) was added. The mixture was heated to mild reflux for 12h. Progress of the reaction was monitored by TLC and LCMS. After completion of the reaction, the reaction mixture was concentrated. Crude product was dissolved in dichloromethane (DCM, 250 ml), washed with water (50 ml), sat. aq. NaHCO3 (50 ml), brine (50 ml), dried over sodium sulfate, and concentrated to give the 2- bromo-4-chloro-benzoic acid methyl ester (26 g, 99 %), which was directly used in the following step.
2-Bromo-4-chloro-benzoic acid methyl ester (12.4 g, 50 mmol) in toluene (200 ml) was cooled to -700C, and trifluoromethyl trimethyl silane (13 ml, 70 mmol) was added. Tetrabutylamonium fluoride (IM, 2.5 ml) was added dropwise, and the mixture was allowed to warm to room temperature over 4h, after which it was stirred for 1Oh at room temperature. The reaction mixture was concentrated to give the crude [l-(2-bromo-4-chloro-phenyl)-2,2,2- trifluoro-l-methoxy-ethoxy]-trimethyl-silane. The crude intermediate was dissolved in methanol (100 ml) and 6N HCl (100 ml) was added. The mixture was kept at 45-500C for 12h. Methanol was removed, and the crude was extracted with dichloromethane (200 ml). The combined DCM layer was washed with water (50 ml), NaHCO3 (50 ml), brine (50 ml), and dried over sodium sulfate. Removal of solvent gave a crude product, which was purified by ISCO column chromatography, using 1-2% ethyl acetate in hexane as solvent, to afford 1- (2-bromo-4-chloro-phenyl)-2,2,2-trifluoro-ethanone (10 g, 70%). 1H-NMR (300 MHz, CDCl3): δ (ppm) 7.50 (d,lH), 7.65(d,lH), 7.80(s,lH).
Step 2: Synthesis of R-l-(2-bromo-4-chloro-phenyl)-2,2,2-trifluoro-ethanol. To catechol borane (IM in THF 280 ml, 280 mmol) in a 2L 3-necked RB flask was added S-2- methyl-CBS oxazaborolidine (7.76 g, 28 mmol) under nitrogen, and the resulting mixture was stirred at room temperature for 20 min. The reaction mixture was cooled to -78°C (dry ice/acetone bath), and l-(2-bromo-4-chloro-phenyl)-2,2,2-trifluoro-ethanone (40 g, 139 mmol) in THF (400 ml) was added dropwise over 2h. The reaction mixture was allowed to warm to -36°C, and was stirred at that temperature for 24 h, and further stirred at -32°C for another 24h. 3N NaOH (250 ml) was added, and the cooling bath was replaced by ice-water bath. Then 30 % hydrogen peroxide in water (250 ml) was added dropwise over 30 minutes. The ice water bath was removed, and the mixture was stirred at room temperature for 4h. The organic layer was separated, concentrated and re-dissolved in ether (200 ml). The aqueous layer was extracted with ether (2 x 200 ml). The combined organic layers were washed with IN aq. NaOH (4 x 100 ml), brine, and dried over sodium sulfate. Removal of solvent gave crude product which was purified by column chromatography using 2 to 5% ethyl acetate in hexane as solvent to give desired alcohol 36.2 g (90 %, e.e. >95%). The alcohol (36.2 g) was crystallized from hexane (80 ml) to obtain R-l-(2-bromo-4-chloro- phenyl)-2,2,2-trifiuoro-ethanol 28.2 g (70 %; 99-100 % e.e.). 1H-NMR (400 MHz, CDCl3) δ (ppm) 5.48 (m, IH), 7.40 (d, IH), 7.61 (d, 2H). Step 3: Synthesis of R-l-r4-chloro-2-(3-methyl-pyrazol-l-vπ-phenyl1-2.2.2-trifluoro- ethanol. R-l-(2-bromo-4-chloro-phenyl)-2,2,2-trifluoro-ethanol (15.65g, 54.06 mmol), 3- methylpyrazole (5.33 g, 65 mmol), CuI (2.06 g, 10.8 mmol), K2CO3 (15.7 g, 113.5 mmol), (lR,2R)-N,N’-dimethyl-cyclohexane-l,2-diamine (1.54 g, 10.8 mmol) and toluene (80 ml) were combined in a 250 ml pressure tube and heated to 1300C (oil bath temperature) for 12 h. The reaction mixture was diluted with ethyl acetate and washed with H2O (4 x 100 ml), brine, and dried over sodium sulfate. Removal of solvent gave a crude product, which was purified by ISCO column chromatography using 5-10 % ethyl acetate in hexane as solvent to get R-I- [4-chloro-2-(3-methyl-pyrazol-l-yl)-phenyl]-2,2,2-trifluoro-ethanol (13.5 g; 86 %). 1H-NMR (400 MHz, CDCl3): δ (ppm) 2.30(s, 3H), 4.90(m, IH), 6.20(s, IH), 6.84(d, IH), 7.20(s, IH), 7.30(d, IH), 7.50(d, IH).
Step 4: Synthesis of (S)-2-Amino-3- r4-(2-amino-6- (R-I- r4-chloro-2-(3-methyl- pyrazol- 1 -ylVphenyl~|-2,2.,2-trifluoro-ethoxy| -pyrimidin-4-yl)-phenyU -propionic acid ethyl ester. R-l-[4-chloro-2-(3-methyl-pyrazol-l-yl)-phenyl]-2,2,2-trifluoro-ethanol (17.78 g, 61.17 mmol), (S)-3-[4-(2-amino-6-chloro-pyrimidine-4-yl)-phenyl]-2-tert- butoxycarbonylamino-propionic acid (20.03 g, 51 mmol), 1,4-dioxane (250 ml), and Cs2CO3 (79.5 g, 244 mmol) were combined in a 3-necked 500 ml RB flask and heated to 1000C (oil bath temperature) for 12-24 h. The progress of reaction was monitored by LCMS. After the completion of the reaction, the mixture was cooled to 600C, and water (250 ml) and THF (400 ml) were added. The organic layer was separated and washed with brine (150 ml). The solvent was removed to give crude BOC protected product, which was taken in THF (400 ml), 3N HCl (200 ml). The mixture was heated at 35-400C for 12h. THF was removed in vacuo. The remaining aqueous layer was extracted with isopropyl acetate (2x 100 ml) and concentrated separately to recover the unreacted alcohol (3.5 g). Traces of remaining organic solvent were removed from the aqueous fraction under vacuum.
To a IL beaker equipped with a temperature controller and pH meter, was added H3PO4 (40 ml, 85 % in water) and water (300 ml) then 50 % NaOH in water to adjust pH to 6.15. The temperature was raised to 58°C and the above acidic aqueous solution was added dropwise into the buffer with simultaneous addition of 50 % NaOH solution in water so that the pH was maintained between 6.1 to 6.3. Upon completion of addition, precipitated solid was filtered and washed with hot water (50-600C) (2 x 200 ml) and dried to give crude (S)-2- amino-3-[4-(2-amino-6-{R-l-[4-chloro-2-(3-methyl-pyrazol-l-yl)-phenyl]-2,2,2-trifluoro- ethoxy}-pyrimidin-4-yl)-phenyl} -propionic acid (26.8 g; 95 %). LCMS and HPLC analysis indicated the compound purity was about 96-97 %. To anhydrous ethanol (400 ml) was added SOCl2 (22 ml, 306 mmol) dropwise at 0-
5°C. Crude acid (26.8 g ) from the above reaction was added. The ice water bath was removed, and the reaction mixture was heated at 40-450C for 6-12h. After the reaction was completed, ethanol was removed in vacuo. To the residue was added ice water (300 ml), and extracted with isopropyl acetate (2 x 100 ml). The aqueous solution was neutralized with saturated Na2CO3 to adjust the pH to 6.5. The solution was extracted with ethyl acetate (2 x 300 ml). The combined ethyl acetate layer was washed with brine and concentrated to give 24 g of crude ester (HPLC purity of 96-97 %). The crude ester was then purified by ISCO column chromatography using 5 % ethanol in DCM as solvent to give (S)-2-amino-3-[4-(2- amino-6- (R- 1 -[4-chloro-2-(3-methyl-pyrazol- 1 -yl)-phenyl]-2,2,2-trifluoro-ethoxy} – pyrimidin-4-yl)-phenyl} -propionic acid ethyl ester (20.5g; 70 %; HPLC purity of 98 %). LCMS M+l = 575. 1H-NMR (400 MHz, CD3OD): δ (ppm) 1.10 (t, 3H), 2.25 (s, 3H), 2.85 (m, 2H), 3.65 (m, IH), 4.00 (q, 2H), 6.35 (s, IH), 6.60 (s, IH), 6.90 (m, IH), 7.18 (d, 2H), 7.45 (m, 2H), 7.70 (d, IH), 7.85 (m, 3H).
PATENT
WO 2011056916
https://www.google.com/patents/WO2011056916A1?cl=en
PATENT
WO 2010065333
CLIP,……..PL CHECK ERROR




REFERENCES
Kulke, M.H.; Hoersch, D.; Caplin, M.E.; et al.
Telotristat ethyl, a tryptophan hydroxylase inhibitor for the treatment of carcinoid syndrome
J Clin Oncol 2017, 35(1): 14
| WO2010056992A1 * | Nov 13, 2009 | May 20, 2010 | The Trustees Of Columbia University In The City Of New York | Methods of preventing and treating low bone mass diseases |
| US7709493 | May 20, 2009 | May 4, 2010 | Lexicon Pharmaceuticals, Inc. | 4-phenyl-6-(2,2,2-trifluoro-1-phenylethoxy)pyrimidine-based compounds and methods of their use |
| US20090088447 * | Sep 25, 2008 | Apr 2, 2009 | Bednarz Mark S | Solid forms of (s)-ethyl 2-amino-3-(4-(2-amino-6-((r)-1-(4-chloro-2-(3-methyl-1h-pyrazol-1-yl)phenyl)-2,2,2-trifluoroethoxy)-pyrimidin-4-yl)phenyl)propanoate and methods of their use |
| Citing Patent | Filing date | Publication date | Applicant | Title |
|---|---|---|---|---|
| US9199994 | Sep 5, 2014 | Dec 1, 2015 | Karos Pharmaceuticals, Inc. | Spirocyclic compounds as tryptophan hydroxylase inhibitors |
| US9512122 | Sep 1, 2015 | Dec 6, 2016 | Karos Pharmaceuticals, Inc. | Spirocyclic compounds as tryptophan hydroxylase inhibitors |
///////////telotristat ethyl, fast track designation,priority review,orphan drug designation, Xermelo , Woodlands, Texas-based, Lexicon Pharmaceuticals, Inc, fda 2017, LX 1606, LX 1032
O=C(OCC)[C@@H](N)Cc1ccc(cc1)c2cc(nc(N)n2)O[C@H](c3ccc(Cl)cc3n4ccc(C)n4)C(F)(F)F
O=C(OCC)[C@@H](N)CC1=CC=C(C2=NC(N)=NC(O[C@H](C3=CC=C(Cl)C=C3N4N=C(C)C=C4)C(F)(F)F)=C2)C=C1.O=C(O)CNC(C5=CC=CC=C5)=O
Tradipitant, традипитант , تراديبيتانت , 曲地匹坦 ,

Tradipitant
VLY-686, LY686017
- Molecular Formula C28H16ClF6N5O
- Average mass 587.903 Da
PHASE 2, Gastroparesis; Pruritus
FDA 2025, APPROVALS 2025, 12/30/2025, To treat vomiting associated with motion
pyridine-containing NK-1 receptor antagonist ie tradipitant, useful for treating anxiety, pruritus and alcoholism.
Vanda Pharmaceuticals, under license from Eli Lilly, was developing tradipitant, a NK1 antagonist, for treating anxiety disorder, pruritus and alcohol dependence. The company was also investigating the drug for treating gastroparesis. In February 2017, tradipitant was reported to be in phase 2 clinical development for treating anxiety and pruritus.
- Originator Eli Lilly
- Developer Eli Lilly; National Institute on Alcohol Abuse and Alcoholism; Vanda Pharmaceuticals
- Class Antipruritics; Anxiolytics; Chlorobenzenes; Pyridines; Small molecules; Triazoles
- Mechanism of Action Neurokinin 1 receptor antagonists; Substance P inhibitors
Highest Development Phases
- Phase II Gastroparesis; Pruritus
- Discontinued Alcoholism; Social phobia
- The drug had been in phase II clinical trials at Lilly and the National Institute on Alcohol Abuse and Alcoholism for the treatment of alcoholism; however, no recent development has been reported for this research.
- A phase II clinical trial for the treatment of social phobia has been completed by Lilly.
PATENT WO 2003091226
SYNTHESIS

Condensation of 2-chloropyridine with thiophenol in the presence of K2CO3 in DMF at 110ºC yields sulfide intermediate,
which is then oxidized by means of NaOCl in AcOH to give 2-(benzenesulfonyl)pyridine.
This is treated with (iPr)2NH and n-BuLi in THF at -60 to -70°C and subsequently couples with 2-chlorobenzaldehyde in THF at -60 to -70°C to furnish (2-(phenylsulfonyl)pyridin-3-yl)-(2-chlorophenyl)methanone.
Ketone couples with the enolate of 4-acetylpyridine (formed by treating 4-acetylpyridine (VII) with t-BuOK in DMSO) in the presence of LiOH in DMSO and subsequently is treated with PhCOOH in iPrOAc to give rise to pyridine benzoate derivative.
This finally couples with 1-azidomethyl-3,5-bistrifluoromethylbenzene (obtained by treating 3,5-bis(trifluoromethyl)benzylchloride with NaN3 ini DMSO) in the presence of K2CO3 in t-BuOH to afford the title compound Tradipitant.
Tradipitant (VLY-686 or LY686017) is an experimental drug that is a neurokinin 1 antagonist. It works by blocking substance P, a small signaling molecule. Originally, this compound was owned by Eli Lilly and named LY686017. VLY-686 was purchased by Vanda Pharmaceuticals from Eli Lilly and Company in 2012.[1] Vanda Pharmaceuticals is a U.S. pharmaceutical company that as of November 2015 only has 3 drugs in their product pipeline: tasimelteon, VLY-686, and iloperidone.[2]
Tachykinins are a family of peptides that are widely distributed in both the central and peripheral nervous systems. These peptides exert a number of biological effects through actions at tachykinin receptors. To date, three such receptors have been characterized, including the NK-1 , NK-2, and NK-3 subtypes of tachykinin receptor.
The role of the NK-1 receptor subtype in numerous disorders of the central nervous system and the periphery has been thoroughly demonstrated in the art. For instance, NK-1 receptors are believed to play a role in depression, anxiety, and central regulation of various autonomic, as well as cardiovascular and respiratory functions. NK- 1 receptors in the spinal cord are believed to play a role in pain transmission, especially the pain associated with migraine and arthritis. In the periphery, NK-1 receptor activation has been implicated in numerous disorders, including various inflammatory disorders, asthma, and disorders of the gastrointestinal and genitourinary tract.
There is an increasingly wide recognition that selective NK-1 receptor antagonists would prove useful in the treatment of many diseases of the central nervous system and the periphery. While many of these disorders are being treated by new medicines, there are still many shortcomings associated with existing treatments. For example, the newest class of anti-depressants, selective serotonin reuptake inhibitors (SSRIs), are increasingly prescribed for the treatment of depression; however, SSRIs have numerous side effects, including nausea, insomnia, anxiety, and sexual dysfunction. This could significantly affect patient compliance rate. As another example, current treatments for chemotherapy- induced nausea and emesis, such as the 5-HT3receptor antagonists, are ineffective in managing delayed emesis. The development of NK-1 receptor antagonists will therefore greatly enhance the ability to treat such disorders more effectively. Thus, the present invention provides a class of potent, non-peptide NK-1 receptor antagonists, compositions comprising these compounds, and methods of using the compounds.
Indications
Pruritus
It is being investigated by Vanda Pharmaceuticals for chronic pruritus (itchiness) in atopic dermatitis. In March 2015, Vanda announced positive results from a Phase II proof of concept study.[3] A proof of concept study is done in early stage clinical trials after there have been promising preclinical results. It provides preliminary evidence that the drug is active in humans and has some efficacy.[4]
Alcoholism
VLY-686 reduced alcohol craving in recently detoxified alcoholic patients as measured by the Alcohol Urge Questionnaire.[5] In a placebo controlled clinical trial of recently detoxified alcoholic patients, VLY-686 significantly reduced alcohol craving as measured by the Alcohol Urge Questionnaire. It also reduced the cortisol increase seen after a stress test compared to placebo. The dose given was 50 mg per day.
Social anxiety disorder
In a 12-week randomized trial of LY68017 in 189 patients with social anxiety disorder, 50 mg of LY68017 did not provide any statistically significant improvement over placebo.[6]
PATENT
WO03091226,
https://www.google.com/patents/WO2003091226A1?cl=en
PATENT
The compound {2-[l-(3,5-bis-trifluoromethyl-benzyl)-5-pyridin-4-yl-lH-[l,2,3]triazol-4-yl]- pyridin-3-yl}-(2-chlorophenyl)-methanone, depicted below as the compound of Formula I, was first described in PCT published application WO2003/091226.
(I)
Because the compound of Formula I is an antagonist of the NK-I subtype of tachykinin receptor, it is useful for the treatment of disorders associated with an excess of tachykinins. Such disorders include depression, including major depressive disorder; anxiety, including generalized anxiety disorder, panic disorder, obsessive compulsive disorder, and social phobia or social anxiety disorder; schizophrenia and other psychotic disorders, including bipolar disorder; neurodegenerative disorders such as dementia, including senile dementia of the Alzheimer’s type or Alzheimer’s disease; disorders of bladder function such as bladder detrusor hyper-reflexia and incontinence, including urge incontinence; emesis, including chemotherapy-induced nausea and acute or delayed emesis; pain or nociception; disorders associated with blood pressure, such as hypertension; disorders of blood flow caused by vasodilation and vasospastic diseases, such as angina, migraine, and Reynaud’s disease; hot flushes; acute and chronic obstructive airway diseases such as adult respiratory distress syndrome, bronchopneumonia, bronchospasm, chronic bronchitis, drivercough, and asthma; inflammatory diseases such as inflammatory bowel disease; gastrointestinal disorders or diseases associated with the neuronal control of viscera such as ulcerative colitis, Crohn’s disease, functional dyspepsia, and irritable bowel syndrome (including constipation-predominant, diarrhea- -?-
predominant, and mixed irritable bowel syndrome); and cutaneous diseases such as contact dermatitis, atopic dermatitis, urticaria, and other eczematoid dermatitis.
In PCT published application, WO2005/042515, novel crystalline forms of the compound of Formula I, identified as Form IV and Form V, are identified. Also described in WO2005/042515 is a process for preparation of the compound of Formula I, comprising reacting (2-chlorophenyl)-[2-(2- hydroxy-2-pyridin-4-yl-vinyl)pyridin-3-yl]methanone or a phosphate salt thereof with l-azidomethyl-3,5- bistrifluoromethylbenzene in the presence of a suitable base and a solvent. Use of this procedure results in several shortcomings for synthesis on a commercial scale. For example, use of the solvent DMSO, with (2- chlorophenyl)-[2-(2-hydroxy-2-pyridin-4-yl-vinyl)pyridin-3-yl]methanone phosphate, requires a complex work-up that has a propensity to emulsify. This process also requires extraction with CH2CI2, the use of which is discouraged due to its potential as an occupational carcinogen, as well as the use of MgSC>4 and acid-washed carbon, which can generate large volumes of waste on a commercial scale. Conducting the reaction with (2-chlorophenyl)-[2-(2-hydroxy-2-pyridin-4-yl-vinyl)pyridin-3-yl]methanone in isopropyl alcohol, as also described in WO2005/042515, is also undesirable due to the need to incorporate a free base step. Furthermore, variable levels of residual l-azidomethyl-3,5-bistrifluoromethylbenzene, a known mutagen, are obtained from use of the procedures described in WO2005/042515.
An improved process for preparing the compound of Formula I would control the level of 1- azidomethyl-3,5-bistrifluoromethylbenzene impurity, and improve the yield. We have discovered that use of the novel salt, (2-chlorophenyl)-[2-(2-hydroxy-2-pyridin-4-yl-vinyl)pyridin-3-yl]methanone benzoate, as well as use of tert-butanol as the reaction solvent, improves reaction times and final yield, and decreases impurities in the final product. In addition, a novel process for the preparation of (2-chlorophenyl)- [2-(2- hydroxy-2-pyridin-4-yl-vinyl)pyridin-3-yl]methanone benzoate, in which a pre-formed enolate of 4-acetyl pyridine is added to (2-phenylsulfonyl-pyridine-3-yl)-(2-chlorophenyl)methanone, results in an overall improved yield and improved purity, and is useful on a commercial scale.
EXAMPLES
Example 1 {2-[l-(3,5-bistrifluoromethylbenzyl)-5-pyridin-4-yl-lH-[l,2,3]triazol-4-yl]-pyridin-3-yl}-(2-chlorophenyl)- methanone (Form IV)
Suspend (2-chlorophenyl)-[2-(2-hydroxy-2-pyridin-4-yl-vinyl)pyridin-3-yl] methanone benzoate (204.7 g; 1.04 equiv; 445 mmoles) in t-butanol (614 mL) and treat the slurry with potassium carbonate (124.2 g; 898.6 mmoles). Heat to 7O0C with mechanical stirring for 1 hour. Add l-azidomethyl-3,5- bistrifluoromethylbenzene (115.6 g; 1.00 equiv; 429.4 mmoles) in a single portion, then heat the mixture to reflux. A circulating bath is used to maintain a condenser temperature of 3O0C. After 18 hours at reflux, HPLC reveals that the reaction is complete (<2% l-azidomethyl-3,5-bistrifluoromethylbenzene remaining). The mixture is cooled to 7O0C, isopropanol (818 mL) is added, then the mixture is stirred at 7O0C for 1 hour. The mixture is filtered, and the waste filter cake is rinsed with isopropanol (409 mL). The combined filtrate and washes are transferred to a reactor, and the mechanically stirred contents are heated to 7O0C. To the dark purple solution, water (1.84 L) is added slowly over 35 minutes. The solution is cooled to 6O0C, then stirred for 1 hour, during which time a thin precipitate forms. The mixture is slowly cooled to RT, then the solid is filtered, washed with 1 : 1 isopropanol/water (614 mL), subsequently washed with isopropanol (410 mL), then dried in vacuo at 450C to produce 200.3 g of crude {2-[l-(3,5- bistrifluoromethylbenzyl)-5-pyridin-4-yl-lH-[l,2,3]triazol-4-yl]-pyridin-3-yl}-(2-chlorophenyl)-methanone as a white solid. Crude {2-[l-(3,5-bistrifluoromethylbenzyl)-5-pyridin-4-yl-lH-[l,2,3]triazol-4-yl]-pyridin- 3-yl}-(2-chlorophenyl)-methanone (200.3 g) and isopropyl acetate (600 mL) are charged to a 5L 3-neck jacketed flask, then the contents heated to 750C. After dissolution is achieved, the vessel contents are cooled to 550C, then the solution polish filtered through a 5 micron filter, and the filter rinsed with a volume of isopropyl acetate (200 mL). After the polish filtration operation is complete, the filtrates are combined, and the vessel contents are adjusted to 5O0C. After stirring for at least 15 minutes at 5O0C, 0.21 grams of {2-[l-(3,5-bistrifluoromethylbenzyl)-5-pyridin-4-yl-lH-[l,2,3]triazol-4-yl]-pyridin-3-yl}-(2- chlorophenyl)-methanone Form IV seed (d90 = 40 microns) is added, and the mixture stirred at 5O0C for at least 2 h. Heptanes (1.90 L) are then added over at least 2 h. After the heptanes addition is completed, the slurry is stirred for an hour at 5O0C, cooled to 230C at a rate less then 2O0C per hour, then aged at 230C for an hour prior to isolation. The mixture is then filtered in portions through the bottom outlet valve in the reactor into a 600 mL filter. The resulting wetcake is washed portionwise with a solution containing heptanes (420 mL) and isopropyl acetate (180 mL), which is passed directly through the 5L crystallization vessel. The wetcake is blown dry for 5 minutes with nitrogen, then transferred to a 500 mL plastic bottle. The product is dried at 5O0C for 4 h. to produce 190.3g of pure {2-[l-(3,5- bistrifluoromethylbenzyl)-5-pyridin-4-yl-lH-[l,2,3]triazol-4-yl]-pyridin-3-yl}-(2-chlorophenyl)- methanone, Form IV in 75.0% yield with 100% purity, as determined by HPLC analysis. Particle size is reduced via pin or jet mill. 1H NMR (400 MHz, CDCl3): 5.46 (s, 2H); 7.19 (m, 5H); 7.36 (dd, IH, J = 4.9, 7.8); 7.45 (s, 2H); 7.59 (m, IH); 7.83 (s, IH); 7.93 (dd, IH, J = 1.5, 7.8); 8.56 (dd, IH, J= 1.5, 4.9); 8.70 (d, 2H, J= 5.9).
Preparation 1-A (2-chlorophenyl)-[2-(2-hydroxy-2-pyridin-4-yl-vinyl)pyridin-3-yl]methanone benzoate Charge powdered KOfBu (221.1 g, 1.93 moles, 1.40 eq.) to Reactor A, then charge DMSO (2 L) at
250C over 10 min. The KOfBu/DMSO solution is stirred for 30 min at 230C, then a solution of 4-acetyl pyridine (92 mL, 2.07 moles, 1.50 eq) in DMSO (250 mL) is prepared in reactor B. The contents of reactor B are added to Reactor A over 10 minutes, then the Reactor A enolate solution is stirred at 230C for Ih. In a separate 12-L flask (Reactor C), solid LiOH (84.26 g, 3.45 moles, 2.0 eq) is poured into a mixture of (2- phenylsulfonyl-pyridin-3-yl)-(2-chlorophenyl)methanone (500.0 g, 1.34 moles, 1.0 eq) and DMSO (2L), with stirring, at 230C. The enolate solution in reactor A is then added to Reactor C over a period of at least 15 minutes, and the red suspension warmed to 4O0C. The reaction is stirred for 3h, after which time HPLC analysis reveals less than 2% (2-phenylsulfonyl-pyridin-3-yl)-(2-chlorophenyl)methanone. Toluene (2.5 L) is charged, and the reactor temperature cooled to 3O0C. The mixture is quenched by addition of glacial acetic acid (316 mL, 5.52 moles, 4.0 eq), followed by 10 % NaCl (2.5 L). The biphasic mixture is transferred to a 22-L bottom-outlet Morton flask, and the aqueous layer is removed. The aqueous layer is then extracted with toluene (750 mL). The combined organic layers are washed with 10 % NaCl (750 mL), then concentrated to 4 volumes and transferred to a 12-L Morton flask and rinsed with isopropyl acetate (4 vol, 2 L). The opaque amber solution is warmed to 75 degrees to 750C over 40 min. Benzoic acid (171. Ig, 1.34 moles, 1.0 eq) is dissolved in hot isopropyl acetate (1.5 L), and charged to the crude free base solution over at least 30 min. The crude solution containing benzoate salt is stirred for 0.5 h at 750C then cooled to 23 0C. When solids are first observed, the cooling is stopped and the mixture is aged for an hour at the temperature at which crystals are first observed. Alternatively, if seed crystal is available, the mixture may be seeded with (2-chlorophenyl)-[2-(2-hydroxy-2-pyridin-4-yl-vinyl)pyridin-3-yl]methanone benzoate (2.25g) at 750C, followed by stirring for 0.5 h at 750C, then cooling to 230C over at least 1.5 h. The mixture is then cooled to <5 0C, then filtered through paper on a 24cm single-plate filter. The filtercake is then rinsed with cold z‘-PrOAc (750 mL) to produce granular crystals of bright orange-red color. The wet solid is dried at 550C to produce 527.3 g (83% yield) with 99.9% purity. (2-chlorophenyl)-[2-(2-hydroxy-2- pyridin-4-yl-vinyl)pyridin-3-yl]methanone benzoate. Anal. Calcd. for C26Hi9N2ClO4: C, 68.05; H, 4.17; N, 7.13. Found: C, 67.89; H, 4.15; N 6.05. HRMS: calcd for C19H13ClN2O2, 336.0666; found 336.0673.
The synthesis of(2-chlorophenyl)-[2-(2-hydroxy-2-pyridin-4-yl-vinyl)pyridin-3-yl]methanone benzoate proceeds optimally when the potassium enolate of 4-acetyl pyridine is pre-formed using KOfBu in DMSO. Pre-formation of the enolate allows the SNAR (nucleophilic aromatic substitution) reaction to be performed between room temperature and 4O0C, which minimizes the amount of degradation. Under these conditions, the SNAR is highly regioselective, resulting in a ratio of approximately 95:5 preferential C – acylation. In all cases, less polar solvents such as THF or toluene, or co-solvents of these solvents mixed with DMSO, results in a substantial increase of acylation at the oxygen in the SNAR, and leads to a lower yield of product. This is a substantial improvement over the procedures described in WO2005/042515 for synthesis of the free base or the phosphate salt, in which the SNAR is performed at 60-700C, resulting in a substantial increase in chemical impurity. Using the conditions described in WO2005/042515, when scaled to 2kg, results in maximum yields of 55%, with sub-optimal potency. In comparison, the improved conditions described herein can be run reproducibly from 0.4 to 2kg scale to give yields of 77-83%, with >99% purity. In addition, the reaction can be held overnight at 4O0C with minimal degradation, whereas holding the reaction for 1 h past completion at 60-70°C results in substantial aromatized impurity. The reaction may also be performed using sodium tert-amylate as the base, in combination with an aprotic solvent, such as DMSO or DMF.
The title compound exists as a mixture of tautomers and geometric isomers. It is understood that each of these forms is encompassed within the scope of the invention.
Preparation 1-B
(2-chlorophenyl)-[2-(2-hydroxy-2-pyridin-4-yl-vinyl)pyridin-3-yl]methanone toluate The procedure described in Preparation 1-A is followed, with the following exception. Solid toluic acid (1.0 eq) is added to the crude free base solution at 550C, then the solution cooled to 45 0C. The solution is stirred for one hour at 45 0C, then slowly cooled to 23 0C. When solids are first observed, the cooling is stopped and the mixture is aged for an hour at the temperature at which crystals are first observed. Alternatively, if seed crystal is available, the mixture may be seeded, aged for 3 h at 450C , then cooled to O0C over 4 h. The isolation slurry is filtered, and the wetcake washed with MeOH (3 volumes). The wetcake is dried at 5O0C to provide 14.0 g (76.4%) of (2-chlorophenyl)-[2-(2-hydroxy-2-pyridin-4-yl- vinyl)pyridin-3-yl]methanone toluate as a light red powder.
As with the benzoate salt, the toluate salt can also exist as a mixture of tautomers and geometric isomers, each of which is encompassed within the scope of the invention. (2-chlorophenyl)-[2-(2-hydroxy- 2-pyridin-4-yl-vinyl)pyridin-3-yl]methanone toluate . 13C NMR (125 MHz,DMS0-d6) δ 194.5, 167.8, 167.4, 155.5, 150.7 (2C), 147.4, 144.0, 143.4, 142.7, 138.6, 133.0, 130.8, 130.7, 130.5, 129.8(2C), 129.5(2C), 128.5, 128.0, 127.9, 119.9 (2C), 118.6, 92.6, 21.5.
Preparation 1-C
(2-phenylsulfonyl-pyridin-3-yl)-(2-chlorophenyl)methanone
A solution of 1.3 eq of diisopropylamine (based on 2-benzenesulfonyl pyridine) in 5 volumes of THF in a mechanically stirred 3 -necked flask is cooled to -70 to -75 0C. To this solution is added 1.05 eq of w-butyllithium (1.6M in hexanes) at such a rate as to maintain the temperature below -6O0C. The light yellow solution is stirred at -60 to -70 0C for 30 minutes. Once the temperature has cooled back down to – 60 to -650C, 1.0 eq of 2-benzene-sulfonyl pyridine, as a solution in 3 volumes of THF, is added at the fastest rate that will maintain the reaction temperature under -6O0C. A yellow suspension forms during the addition that becomes yellow-orange upon longer stirring. This mixture is stirred for 3 hours at -60 to – 750C, and then 1.06 eq of 2-chlorobenzaldehyde, as a solution in 1 volume of THF, is added dropwise at a sufficient rate to keep the temperature under -55 0C. The suspension gradually turns orange-red, thins out, and then becomes a clear red solution. The reaction mixture is allowed to stir at -60 to -7O0C for 1 hour, 3N aqueous HCl (7 volumes) is added over 20-30 minutes, and the temperature is allowed to exotherm to 0-100C. The color largely disappears, leaving a biphasic yellow solution. The solution is warmed to at least 1O0C, the layers are separated, and the aqueous layer is back-extracted with 10 volumes of ethyl acetate. The combined organic layers are washed with 10 volumes of saturated sodium bicarbonate solution and concentrated to about 2 volumes. Ethyl acetate (10 volumes) is added, and the solution is once again concentrated to 2 volumes. The thick solution is allowed to stand overnight and is taken to the next step with no purification of the crude alcohol intermediate. The crude alcohol intermediate is transferred to a 3 -necked flask with enough ethyl acetate to make the total solution about 10 volumes. The yellow solution is treated with 3.2 volumes of 10% aqueous (w/w) potassium bromide, followed by 0.07 eq of 2,2,6,6-Tetramethylpiperidine-N-oxide (TEMPO). The orange mixture is cooled to 0-50C and treated with a solution of 1.25 eq of sodium bicarbonate in 12% w/w sodium hypochlorite (9 volumes) and 5 volumes of water over 30-60 minutes while allowing the temperature to exotherm to a maximum of 2O0C. The mixture turns dark brown during the addition, but becomes yellow, and a thick precipitate forms. The biphasic light yellow mixture is allowed to stir at ambient temperature for 1-3 hours, at which time the reaction is generally completed. The biphasic mixture is cooled to 0-50C and stirred for 3 hours at that temperature. The solid is filtered off, washed with 4 volumes of cold ethyl acetate, followed by 4 volumes of water, and dried in vacuo at 450C to constant weight. Typical yield is 80-83% with a purity of greater than 98%. 1H NMR (600 MHz, CDCl3-^) δ ppm 7.38 (td, ./=7.52, 1.28 Hz, 1 H) 7.47 (dd, ./=7.80, 1.30 Hz, 1 H) 7.51 (td, ./=7.79, 1.60 Hz, 1 H) 7.51 (t, ./=7.89 Hz, 2 H) 7.50 – 7.54 (m, J=7.75, 4.63 Hz, 1 H) 7.60 (t, J=7.43 Hz, 1 H) 7.73 (dd, J=7.75, 1.60 Hz, 1 H) 7.81 (dd, J=7.79, 1.56 Hz, 1 H) 8.00 (dd, ./=8.44, 1.10 Hz, 2 H) 8.76 (dd, ./=4.63, 1.61 Hz, 1 H).
Preparation 1-D 1 -azidomethyl-3,5-bistrifluoromethyl-benzene
Sodium azide (74.3 g, 1.14 mol) is suspended in water (125 mL), then DMSO (625 mL) is added. After stirring for 30 minutes, a solution consisting of 3,5-Bis(trifluoromethyl)benzyl chloride (255.3 g, 0.97 moles) and DMSO (500 mL) is added over 30 minutes. (The 3,5-Bis(trifluoromethyl)benzyl chloride is heated to 350C to liquefy prior to dispensing (MP = 30-320C)). The benzyl chloride feed vessel is rinsed with DMSO (50 mL) into the sodium azide solution, the mixture is heated to 4O0C, and then maintained for an hour at 4O0C, then cooled to 230C.
In Process Analysis: A drop of the reaction mixture is dissolved in d6-DMSO and the relative intensities of the methylene signals are integrated (NMR verified as a 0.35% limit test for 3,5- Bis(trifluoromethyl)benzyl Chloride). Work-up: After the mixture reaches 230C , it is diluted with heptanes (1500 mL), then water (1000 mL) is added, and the mixture exotherms to 350C against a jacket setpoint of 230C. The aqueous layer is removed (-2200 mL), then the organic layer (approximately 1700 mL) is washed with water (2 X 750 mL). The combined aqueous layers (-3700 mL) are analyzed and discarded.
The solvent is then partially removed via vacuum distillation with a jacket set point of 850C, pot temperature of 60-650C and distillate head temperature of 50-550C to produce 485g (94.5% yield) of 51 Wt% solution title compound as a clear liquid. Heptanes can be either further removed by vacuum distillation or wiped film evaporation technology. 1H NMR (400 MHz, CDCl3): 4.58 (s, 2H); 7.81 (s, 2H); 7.90 (s, IH).
Preparation 1-E 2-benzene-sulfonyl pyridine Charge 2-chloropyridine (75 mL, 790 mmol), thiophenol (90 mL, 852 mmol), and DMF (450 mL) to a 2L flask. Add K2CO3 (134.6 g, 962 mmol), then heat to HO0C and stir for 18 hours. Filter the mixture, then rinse the waste cake with DMF (195 mL). The combined crude sulfide solution and rinses are transferred to a 5-L flask, and the waste filtercake is discarded. Glacial acetic acid (57 mL, 995 mmol) is added to the filtrate, then the solution is heated to 4O0C, and 13 wt % NaOCl solution (850 mL, 1.7 mol) is added over 2 hours. After the reaction is complete, water (150 mL) is added, then the pH of the mixture adjusted to 9 with 20 % (w/v) NaOH solution (250 mL). The resulting slurry is cooled to <5 0C, stirred for 1.5 h, then filtered, and the cake washed with water (3 x 200 mL). The product wetcake is dried in a 550C vacuum oven to provide 2-benzene-sulfonyl pyridine (149 g, 676 mmol) in 86 % yield: 1H NMR (500 MHz, CDCl3) δ 8.66 (d, J = 5.5 Hz, IH), 8.19 (d, J = 1.1 Hz, IH), 8.05 (m, 2H), 7.92 (ddd, J= 9.3, 7.7, 1.6 Hz, IH), 7.60 (m, IH), 7.54 (m, 2H), 7.44 (m, IH); IR (KBr) 788, 984, 1124, 1166, 1306, 1424, 1446, 1575, 3085 cm“1; MS (TOF) mlz 220.0439 (220.0427 calcd for C11H10NO2S, MH); Anal, calcd for C11H9NO2S: C, 60.26; H, 4.14; N, 6.39; S, 14.62. Found: C, 60.40; H, 4.02; N, 6.40; S, 14.76.
As noted above, use of the improved process of the present invention results in an improved habit of the crystalline Form IV compound of Formula I. The improved habit reduces surface area of the crystal, improves the filtration, and washing, and improves the efficiency of azide mutagen rejection. These improvements are described in greater detail below.
In patent application WO2005/042515, the polish filtration is carried out in 7 volumes (L/kg) of isopropanol near its boiling point (65-83 0C), a process that is difficult and hazardous to execute in commercial manufacturing because of the high risk of crystallization on the filter and/or vessel transfer lines due to supersaturation. In the preferred crystallization solvent, isopropyl acetate, the polish filtration is conducted in four volumes of isopropyl acetate at temperatures from 45 to 55 0C. This temperature range is 35 to 45 0C lower than the boiling point of isopropyl acetate, which provides a key safety advantage.
PATENT
PATENT
WO 2017031215
EXAMPLES
Example 1: Preparation of Compound (I) via Negishi Coupling Route
Example 1 provides a scheme including preparations 1A-1D, described below, for the synthesis of the compound of Formula (I) and intermediates used in the route. An overview of the scheme is as follows:

80 on ma s ale
Example 1A: Preparation of Compound (I)

Zinc dust (200 mg, 3.06 mmol) combined with 2.0 mL of dimethylformamide was treated with 0.010 mL of 1,2-dibromoethane and heated to 65°C for 3 minutes. The mixture was cooled to ambient temperature and treated with 0.010 mL of trimethylsilyl chloride. After 5 minutes, 1.26 mL of 1M zinc chloride in diethyl ether was added to the mixture followed by Compound (Ila) (600 mg, 1.20 mmol). The mixture was heated to 65°C and further treated with 0.020 mL each of 1,2-dibromoethane and trimethylsilyl chloride. After 2.5 hours, via HPLC chromatogram, the reaction showed some formation of the zincate and was allowed to stir at ambient temperature for 16 hours. At this time
tetrakis(triphenylphosphine)palladium(0) (70 mg, 0.06 mmol), Compound (Ilia) (357 mg, 1.20 mmol) were added to the reaction and the mixture heated to 65°C. HPLC analysis showed the formation of Compound (I) in the reaction.
IB: Preparation of Comp

To a solution of Compound (IV) (8.00 g, 18 mmol) in 40 mL of 1,2-dichloroethane was added a solution of iodine monochloride (10.7 g, 65.9 mmol) in 40 mL of 1,2-dichloroethane resulting in a slurry. The slurry was heated to 75°C for 4 hours then cooled to ambient temperature. The solids were collected by filtration, washed with heptane, then combined with 90 mL of ethyl acetate and 80 mL of saturated sodium thiosulfate solution. The organic phase was washed with saturated sodium chloride solution and dried with sodium sulfate. The mixture was concentrated to yield 7.80 g (87%) of Compound (Ila) as a yellow solid. The product could be further purified by silica gel chromatography. Thus 2.0 g of yellow solid was dissolved in dichloromethane and charged onto a silica gel column. The product was eluted using tert-butyl methyl ether to provide 1.87 g (93% recovery) of Compound (Ila) as a white powder. Analytical data: Iodine monochloride complex: ¾ NMR (500 MHz, DMSO-de) δ 8.80 (2 H), 8.05 (1 H), 7.77 (2 H), 7.59 (2 H), 5.86 (2 H).
Uncomplexed: ¾ NMR (500 MHz, DMSO-de) δ 8.71 (2 H), 8.03 (1 H), 7.74 (2 H), 7.44 (2 H), 5.86 (2 H).
It was observed that the iodination proceeded smoothly as a suspension in 1,2-dichloroethane with IC1 (4.0 equiv) at 75°C. An ICl-Compound (Ila) complex was initially isolated by filtration. Compound (Ila) was then obtained in approximately 85% yield by treatment of the ICl-Compound (Ila) complex with sodium thiosulfate. This protocol provided a viable means of isolation of Compound (Ila) without the use of DMF.
Example 1C: Preparation of silyl substituted triazole (Compound IV)

A mixture of Compound (V) (8.07 g, 30.0 mmol) and Compound (VI) (5.12 g, 29.2 mmol) was heated to 100°C for 18 hours. To the mixture was added 40 mL of heptane and the reaction was allowed to cool with rapid stirring. After 1 hour the solids were collected by filtration and washed with heptane then dried to 9.30 g (72%) of Compound (IV) as a tan solid. Analytical data: ¾ NMR (500 MHz, DMSO-de) δ 8.66 (2 H), 8.04 (1 H), 7.67 (2 H), 7.32 (2 H), 5.72 (2 H), 0.08 (9 H).
It was further found that combining Compound (V) and Compound (VI) (neat) and heating at 95 – 105°C afforded a 92: 8 mixture of regioisomers as shown below:

Crystallization of the mixture from heptane afforded Compound (IV) in 62-72% yield, thus obviating the need for chromatography to isolate Compound (IV).
Example ID: Preparation of starting material Compound (VI)

Zinc bromide (502 g, 2.23 mole) was added in approximately 100 g portions to 2.0 L of tetrahydrofuran cooled to between 0 and 10°C. To this cooled solution was added 4-bromopyridine hydrochloride (200 g, 1.02 mol), triphenylphosphine (54 g, 0.206 mol), and palladium (II) chloride (9.00 g, 0.0508 mol). Triethylamine (813 g, 8.03 mol) was then added at a rate to maintain the reaction temperature at less than 10°C, and finally
trimethylsilylacetylene (202 g, 2.05 mol) was added. The mixture was heated to 60°C for 4.5 hours. The reaction was cooled to -5°C and combined with 2.0 L of hexanes and treated with 2 L of 7.4 M NH4OH. Some solids were formed and were removed as much as possible with the aqueous phase. The organic phase was again washed with 2.0 L of 7.4 M NH4OH, followed by 2 washes with 500 mL of water, neutralized with 1.7 L of 3 M hydrochloric acid, dried with sodium sulfate, and concentrate to a thick slurry. The slurry was combined with 1.0 L of hexanes to give a precipitate. The precipitate was removed by filtration and the filtrate was concentrated to 209 g of dark oil. The product was purified by distillation (0.2 torr, 68°C) to give 172 g (96%) of Compound (VI) as colorless oil. Analytical data: ¾ NMR (500 MHz, DMDO-de) δ 8.57 (2 H), 7.40 (2 H), 0.23 (9 H).
EXAMPLE 2 – Preparation of Compound (Ilia)
Example 2 provides a morpholine amide route for the synthesis of Compound (Ilia). In this approach, morpholine amide (Compound VII) was prepared from 2-chlorobenzoyl chloride (Preparation 2A). Metallation of 2-bromopyridine with LDA (1.09 equiv.) in THF at -70°C followed by addition of (Compound VII) afforded Compound (Ilia) in 37% yield after crystallization from IP A/heptane (Preparation 2B). This sequence provides a direct route to Compound (Ilia), and a means to isolate Compound (Ilia) without the use of
chromatography. Compound (Ilia) may then be used to form Compound (I) as shown in Example 1A above (Preparation 2C).
Preparation 2A: Preparation of Compound (VII)

Toluene (1.5 L) was added to Compound (IX) (150 g, 0.86 mol) and cooled to 10°C. Morpholine (82 mL, 0.94 mol) was added to the clear solution over 10 minutes. The resulting white slurry was stirred for 20 minutes then pyridine (92 mL, 1.2 mol) was added dropwise over 20 minutes. The cloudy white mixture was stirred in a cold bath for 1 hour. Water (600 mL) was added in a single portion and the cold bath removed. The mixture was stirred for 20 minutes and the layers are separated. The organic layer was washed with a mixture of 1 N HC1 and water (2: 1, 500 mL:250 mL). The pH of the aqueous layer was ~ 2. The organic layer was washed with a mixture of saturated NaHCCb and water (1 : 1, 100 mL: 100 mL). The pH of the aqueous layer was ~ 9. The layers were separated. The organic layer was concentrated in vacuo to an oil. The oil was dissolved in IPA (70 mL) and heated at 60°C for 30 min. The clear solution was allowed to cool to 30°C, then heptane (700 mL, 4.7 v) was added dropwise. The resulting slurry was stirred at RT for 2 hours then cooled to 0°C for 1 hour. The slurry was filtered at RT, washed with heptane then dried under vacuum at 30°C overnight. Compound (VII) (156.2 g, 81%) was obtained as a white solid. Analytical data: ¾ NMR (500 MHz, CDCh) δ 7.42-7.40 (m, 1 H), 7.35-7.29 (m, 3 H), 3.91-3.87 (m, 1 H), 3.80-3.76 (m, 3 H), 3.71 (ddd, J= 11.5, 6.8, 3.3 Hz, 1 H), 3.60 (ddd, J = 11.2, 6.4, 3.4 Hz, 1 H), 3.28 (ddd, J= 13.4, 6.3, 3.2 Hz, 1 H), 3.22 (ddd, J= 13.7, 6.8, 3.3 Hz, 1 H); LRMS (ES+) calcd for CnHi3F6ClN02 (M+H)+ 226.1, found 225.9 m/z.
Preparation 2B: Preparation of Compound (Ilia)

THF (75 mL) was added to diisopropyl amine (4.9 mL, 34.8 mmol) and cooled to a
temperature of -70°C under N2 atmosphere. 2.5 M w-BuLi in hexanes (13.9 mL, 34.8 mmol) was added in a single portion (a 30-40°C exotherm) to the clear solution and cooled back to -70°C. Compound (VIII) (5.0 g, 31.6 mmol) was added neat to the LDA solution (a 2 to 5°C exotherm) followed by a THF (10 mL) rinse, keeping T< -65°C. This clear yellow solution was stirred at -70°C for 15 min. Compound (VII) (7.1 g, 31.6 mmol) in THF (30 mL) was added keeping T< -65°C. The resulting clear orange solution was stirred at -70°C for 3 hours. MeOH (3 mL) was added to quench reaction mixture and the cold bath was removed. 5 N HC1 (25 mL) was added to the reaction solution. MTBE (25 mL) was added, and the layers were separated. The organic layer was washed with water (25 mL X 2). The organic layer was dried over MgS04 and filtered. The organic layer was concentrated in vacuo to an orange oil. The oil was dissolved in IPA (15 mL, 3 vol) at ambient temperature. Heptane (25 mL) was added dropwise and the resulting slurry was stirred at RT for 1 hour. The slurry was cooled to 0°C for 1 hour and filtered. The filter cake was rinsed with chilled heptane (20 mL) and dried under vacuum at 30°C overnight. Compound (Ilia) (4.25 g, 45%) was obtained as a yellow solid.
Several reactions were run at different temperatures and with different addition rates of Compound (VII). If the reaction temperature was maintained below -65°C and Compound (VII) was added in <5 min, it was found that the reaction worked well. If the temperature was increased and/or the addition time of Compound (VII) was increased, then yields suffered, and the work-up was complicated by emulsions.
Preparation 2C: Preparation of Compound (I)
Compound (Ilia) may then reacted with Compound (Ila) to produce Compound (I) as shown in Preparation 1A.
EXAMPLE 3
Example 3 describes a new route for the synthesis of an intermediate free base, which may be used to form Compound (I) as described further below.
Example 3A: Preparation of starting material (Compound X) from 2-Chloronicotinonitrile

A mixture of NaH (40.0 g, 1 mol, 60% dispersion in mineral oil) and 2-chloronicotinonitrile (69.3 g, 500 mmol) in THF (1 L) was heated to reflux. A solution of 4-acetylpyridine (60.6 g, 500 mmol) in THF (400 mL) was added over a period of 40 min. The resulting dark brown mixture was stirred at reflux for ~ 2 h. The heating mantle was then removed, and AcOH (58 mL, 1 mol) was added. EtOAc (1 L) and H2O (1 L) were then added, and the layers were separated. The organic layer was concentrated to afford an oily solid. CH3CN (500 mL) was added, and the mixture was stirred for 30 min. H2O (1 L) was then added. The mixture was stirred for 1 h then filtered. The solid was rinsed with 2: 1
CH3CN-H2O (900 mL) and hexanes (400 mL) then dried under vacuum at 45°C overnight to afford 61.4 g (55% yield) of Compound (X) as yellow solid. Compound (X) exists as an approximate 95:5 enol-ketone mixture in CDCI3. Analytical data for enol: IR (CHCI3): 3024, 2973, 2229, 1631, 1597, 1579, 1550, 1497; ¾ NMR (500 MHz, CDCI3) δ 8.69 (dd, J= 4.4,
1.7 Hz, 2H), 8.55 (dd, J = 5.2, 1.8 Hz, 1H), 7.97 (dd, J= 7.9, 1.8 Hz, 1H), 7.70 (dd, J= 4.6, 1.5 Hz, 2H, 7.17 (dd, J = 7.8, 5.0 Hz, 1H), 6.59 (s, 1H); LRMS (ES+) calcd for C13H10N3O (M+H)+ 224.1, found 224.0 m/z.
Preparation 3B: Preparation of Compound (XI)
Preparation 3B(1):

(X) (XI)
Compound (XI) may be prepared using Compound (X).
Preparation 3B(2):
Alternatively, the following procedure for the conversion of nitrile into an acid which may also yield compound (XI). A mixture of Compound (X) (1 eq) and NaOH (1.5 eq) in 1 : 1 fhO-EtOH (3.5 mL/g of Compound (X)) was heated at 65°C overnight. The reaction mixture was cooled to RT then added to CH2C12 (12.5 mL/g of Compound (X)) and H20 (12.5 mL/g of Compound (X)). Cone. HC1 (2.5 mL/g of Compound (X)) was then added, and the layers were separated. The aqueous layer was extracted with CH2CI2 (10 mL/g of Compound (X)). The combined organic extracts were washed with H2O (12.5 ml/g of Compound (X)), dried (MgS04), filtered and concentrated to afford Compound (XI).
Preparation 3C
Compound Compound (XI) may then be converted into a Stage C intermediate free base, with observed 87% conversion in Grignard reaction as shown above. A complete synthesis route for Com ound (I) starting from compound Compound (XI) is depicted below.

Detailed experimental procedures for the synthesis of benzoate salt and final step are given in
International Patent Application Publication WO 2008/079600 Al .
References
- “Company Overview of Eli Lilly & Co., Worldwide License to Develop and Commercialize VLY-686”. Bloomberg Business. Retrieved 16 November 2015.
- [1]
- “Vanda Pharmaceuticals Announces Tradipitant Phase II Proof of Concept Study Results for Chronic Pruritus in Atopic Dermatitis”. PR Newswire. Retrieved 16 November 2015.
- Schmidt, B (2006). “Proof of principle studies”. Epilepsy Res. 68 (1): 48–52. doi:10.1016/j.eplepsyres.2005.09.019. PMID 16377153.
- George, DT; Gilman, J; Hersh, J; et al. (2008). “Neurokinin 1 receptor antagonism as a possible therapy for alcoholism.”. Science. 6: 1536–1539. doi:10.2147/SAR.S70350. PMC 4567173
 . PMID 26379454.
. PMID 26379454. - Tauscher, J; Kielbasa, W; Iyengar, S; et al. (2010). “Development of the 2nd generation neurokinin-1 receptor antagonist LY686017 for social anxiety disorder”. European Neuropsychopharmacology. 20 (2): 80–87. doi:10.1016/j.euroneuro.2009.10.005. PMID 20018493.
George, D.T.; Gilman, J.; Hersh, J.; Thorsell, A.; Herion, D.; Geyer, C.; Peng, X.; Kielbasa, W.; Rawlings, R.; Brandt, J.E.; Gehlert, D.R.; Tauscher, J.T.; Hunt, S.P.; Hommer, D.; Heilig, M. Neurokinin 1 receptor antagonism as a possible therapy for alcoholism, Science 2008, 319(5869): 1536
Gackenheimer, S.L.; Gehlert, D.R.In vitro and in vivo autoradiography of the NK-1 antagonist (3H)-LY686017 in guinea pig brain39th Annu Meet Soc Neurosci (October 17-21, Chicago) 2009, Abst 418.16
Tonnoscj, K.; Zopey, R.; Labus, J.S.; Naliboff, B.D.; Mayer, E.A.
The effect of chronic neurokinin-1 receptor antagonism on sympathetic nervous system activity in irritable bowel syndrome (IBS) Dig Dis Week (DDW) (May 30-June 4, Chicago) 2009, Abst T1261
Kopach, M.E.; Kobierski, M.E.; Coffey, D.S.; et al.
Process development and pilot-plant synthesis of (2-chlorophenyl)[2-(phenylsulfonyl)pyridin-3-yl]methanone
Org Process Res Dev 2010, 14(5): 1229
| Patent ID | Patent Title | Submitted Date | Granted Date |
|---|---|---|---|
| US2016060250 | NOVEL INTERMEDIATE AND PROCESS USEFUL IN THE PREPARATION OF -(2-CHLOROPHENYL)-METHANONE | 2015-11-10 | 2016-03-03 |
| US2015320866 | PHARMACEUTICAL COMPOSITION COMPRISING ANTIEMETIC COMPOUNDS AND POLYORTHOESTER | 2013-12-13 | 2015-11-12 |
| US2014206877 | NOVEL INTERMEDIATE AND PROCESS USEFUL IN THE PREPARATION OF -(2-CHLOROPHENYL)-METHANONE | 2014-03-27 | 2014-07-24 |
| US2012225904 | New 7-Phenyl-[1, 2, 4]triazolo[4, 3-a]Pyridin-3(2H)-One Derivatives | 2010-11-09 | 2012-09-06 |
| US2010056795 | NOVEL INTERMEDIATE AND PROCESS USEFUL IN THE PREPARATION OF -(2-CHLOROPHENYL)-METHANONE | 2010-03-04 | |
| US7381826 | Crystalline forms of {2-[1-(3, 5-bis-trifluoromethyl-benzyl)-5-pyridin-4-yl-1H-[1, 2, 3]triazol-4-yl]-pyridin-3-yl}-(2-chlorophenyl)-methanone | 2007-04-05 | 2008-06-03 |
| US7320994 | Triazole derivatives as tachykinin receptor antagonists | 2005-10-27 | 2008-01-22 |
 |
|
| Legal status | |
|---|---|
| Legal status |
|
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| Chemical and physical data | |
| Formula | C28H16ClF6N5O |
| Molar mass | 587.90 g/mol |
| 3D model (Jmol) | |
Tradipitant
Tradipitant is being evaluated in a Phase II study in treatment resistant pruritus in atopic dermatitis.
Tradipitant is an NK-1 receptor antagonist licensed from Eli Lilly in 2012. Tradipitant has demonstrated proof-of-concept in alcohol dependence in a study published by the NIH1. In that study tradipitant was shown to reduce alcohol cravings and voluntary alcohol consumption among patients with alcohol dependence. NK-1R antagonists have been evaluated in a number of indications including chemotherapy-induced nausea and vomiting (CINV), post-operative nausea and vomiting (PONV), alcohol dependence, anxiety, depression, and pruritus.
The NK-1R is expressed throughout different tissues of the body, with major activity found in neuronal tissue. Substance P (SP) and NK-1R interactions in neuronal tissue regulate neurogenic inflammation locally and the pain perception pathway through the central nervous system. Other tissues, including endothelial cells and immune cells, have also exhibited SP and NK-1R activity2. The activation of NK-1R by the natural ligand SP is involved in numerous physiological processes, including the perception of pain, behavioral stressors, cravings, and the processes of nausea and vomiting1,2,3. An inappropriate over-expression of SP either in nervous tissue or peripherally could result in pathological conditions such as substance dependence, anxiety, nausea/vomiting, and pruritus1,2,3,4. An NK-1R antagonist may possess the ability to reduce this over-stimulation of the NK-1R, and as a result address the underlying pathophysiology of the symptoms in these conditions.
References
- George DT, Gilman J, Hersh J, Thorsell A, Herion D, Geyer C, Peng X, Keilbasa W, Rawlings R, Brandt JE, Gehlert DR, Tauscher JT, Hunt SP, Hommer D, Heilig M. Neurokinin 1 receptor antagonism as a possible therapy for alcoholism. Science. 2008; 319(5869):1536-9
- Almeida TA, Rojo J, Nieto PM, Pinto FM, Hernandez M, et al. Tachykinins and tachykinin receptors: structure and activity relationships. Current Medicinal Chemistry. 2004;11:2045-2081.
- Hargreaves R, Ferreira JC, Hughes D, Brands J, Hale J, Mattson B, Mill S. Development of aprepitant, the first neurokinin-1 receptor antagonist for the prevention of chemotherapy-induced nausea and vomiting. Annals of the New York Academy of Sciences. 2011; 1222:40-48.
- Stander S, Weisshaar E, Luger A. Neurophysiological and neurochemical basis of modern pruritus treatment. Experimental Dermatology. 2007;17:161-69.
///////////////////tradipitant, PHASE 2, VLY-686, LY686017, традипитант , تراديبيتانت , 曲地匹坦 , VANDA, ELI LILLY, Gastroparesis Pruritus, FDA 2025, APPROVALS 2025, vomiting associated with motion

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO