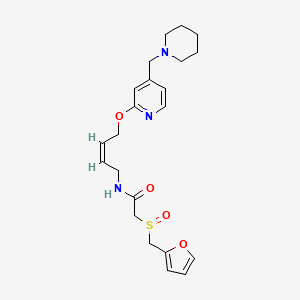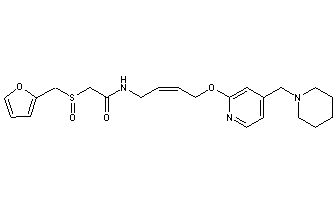


LAFUTIDINE
N-[4-[4-(Piperidin-1-ylmethyl)pyridin-2-yloxy]-(Z)-but-2-en-1-yl]-2-(furfurylsulfinyl)acetamide
-
- FRG-8813
- ATC:A02B
- Use:antisecretory, gastric H2-antagonist
- (+)-2-[(2-furanylmethyl)sulfinyl]-N-[(2Z)-4-[[4-(1-piperidinylmethyl)-2-pyridinyl]oxy]-2-butenyl]acetamide
- Formula:C22H29N3O4S
- MW:431.56 g/mol
- CAS-RN:118288-08-7
-
(±)-2-(Furfurylsulfinyl)-N-(4-(4-(piperidinomethyl)-2-pyridyl)oxy-(Z)-2-butenyl)acetamide
-
(Z)-2-((2-Furanylmethyl)sulfinyl)-N-(4-((4-(1-piperidinylmethyl)-2-pyridinyl)oxy)-2-butenyl)acetamide
-
118288-08-7
FRG‐8813、2‐(Furfurylsulfinyl)‐N‐[(Z)‐4‐[[4‐(piperidinomethyl)‐2‐pyridinyl]oxy]‐2‐butenyl]acetamide、ロクチジン、Loctidine、ラフチジン・・・
Lafutidine , also named N-[4-[4-(piperidin-1-ylmethyl)pyridin-2-yloxy]-(Z)-but-2-en-1-yl]-2-(furfurylsulfinyl)acetamide, is a histamine H2 receptor antagonist that was first produced in Japan by Taiho and UCB Japan for the oral treatment of peptic ulcers in 2000. In 2010 it was approved for the treatment of mild gastroesophageal reflux disease, and in 2012 it was approved to help improve symptoms of gastric mucosal lesions due to gastritis
Lafutidine (INN) is a second generation histamine H2 receptor antagonist having multimodal mechanism of action and used to treat gastrointestinal disorders. It is marketed in Japan and India.
Medical use
Lafutidine is used to treat gastric ulcers, duodenal ulcers, as well as wounds in the lining of the stomach associated with acute gastritis and acute exacerbation of chronic gastritis.[1][2]
Adverse effects
Adverse events observed during clinical trials included constipation, diarrhea, drug rash, nausea, vomiting and dizziness.[2]
Mechanism of action
Like other H2 receptor antagonists it prevents the secretion of gastric acid.[2] It also activates calcitonin gene-related peptide, resulting in the stimulation of nitric oxide (NO) and regulation of gastric mucosal blood flow, increases somatostatin levels also resulting in less gastric acid secretion, causes the stomach lining to generate more mucin, inhibits neutrophil activation thus preventing injury from inflammation, and blocks the attachment of Helicobacter pylori to gastric cells.[2]

Trade names
It is marketed in Japan as Stogar by UCB[1] and in India as Lafaxid by Zuventus Healthcare.[2]





N-[4-[4-(Piperidin-1-ylmethyl)pyridin-2-yloxy]-(Z)-but-2-en-1-yl]-2-(furfurylsulfinyl)acetamide 1 as a white solid (15.8 kg, 91.3%).(2,3)
1H NMR (600 MHz, CDCl3): δ 1.43 (m, 2H), 1.56–1.60 (m, 4H), 2.36 (m, 4H), 3.34 (d, 1H, J = 14.4 Hz), 3.40 (s, 2H), 3.59 (d, 1H, J = 14.4 Hz), 4.10 (t, 2H, J = 6.6 Hz), 4.17 (d, 1H, J = 13.8 Hz), 4.31 (d, 1H, J = 13.8 Hz), 4.93 (d, 2H, J = 6.6 Hz), 5.67–5.69 (m, 1H), 5.83–5.87 (m, 1H), 6.39 (dd, 1H, J = 1.8, 3.0 Hz), 6.47 (d, 1H, J = 3.0 Hz), 6.72 (s, 1H), 6.87 (d, 1H, J = 5.4 Hz), 7.19 (s, 1H), 7.43 (d, 1H, J = 1.8 Hz), 8.03 (d, 1H, J = 5.4 Hz).
13C NMR (150 MHz, CDCl3): δ 24.2, 26.0, 26.0, 37.2, 50.2, 53.4, 54.6, 54.6, 61.4, 62.4, 110.8, 111.3, 112.2, 117.7, 128.4, 128.9, 143.3, 143.9, 146.3, 151.5, 163.6, 163.6.
IR (KBr): 3325, 2935, 1638, 1613, 1041 cm–1.
ESI-MS: m/z 431.1.
Increasing the Purity of Lafutidine Using a “Suicide Substrate”
Chengjun Wu, Zhen Li, Chunchao Wang, Yanan Zhou, and Tiemin Sun*
https://pubs.acs.org/doi/suppl/10.1021/acs.oprd.8b00070/suppl_file/op8b00070_si_001.pdf
CLIP
http://www.drugfuture.com/synth/syndata.aspx?ID=145925

EP 0282077; JP 1988225371; JP 1989230556; JP 1989230576; US 4912101
1) The reaction of 2-bromo-4-(piperidin-1-ylmethyl)pyridine (I) with 4-amino-2(Z)-buten-1-ol (II) by means of NaH in THF gives 4-[4-(piperidin-1-ylmethyl)pyridin-2-yloxy]-2(Z)-buten-1-amine (III), which is then condensed with 2-(2-furylmethylsulfinyl)acetic acid (IV) by means of 1-ethyl-3-[3-(dimethylamino)propyl]carbodiimide (EDCD) in dichloromethane.
 EP 0582304; JP 1994192195
EP 0582304; JP 1994192195
The condensation of 2-chloro-4-(piperidin-1-ylmethyl)pyridine (V) with 4-(tetrahydropyranyloxy)-2(Z)-buten-1-ol (VI) by means of NaH in THF gives 4-(piperidin-1-ylmethyl)-2-[4-(tetrahydropyranyloxy)-2(Z)-butenyloxy)pyridine (VII), which is deprotected with 4-methylbenzenesulfonic acid in methanol, yielding the free butenol (VIII). The acylation of (VIII) with methanesulfonyl chloride in toluene affords the corresponding mesylate (IX), which is finally condensed with 2-(2-furylmethylsulfonyl)acetamide (X) (obtained from the corresponding 4-nitrophenyl ester (XI) with ammonia) by means of potassium tert-butoxide in toluene.
 Chem Pharm Bull 1998,46(4),616
Chem Pharm Bull 1998,46(4),616
A new synthesis of lafutidine has been described: The condensation of 2-bromopyridine-4-carbaldehyde ethylene ketal (I) with 4-(tetrahydropyranyloxy)-2(Z)-buten-1-ol (II) by means of NaOH, K2CO3 and tetrabutylammonium bisulfate in refluxing toluene gives the corresponding substitution product (III), which by treatment with pyridinium p-toluenesulfonate (PPTS) in hot ethanol yields the 2(Z)-butenol (IV). The reaction of (IV) with SOCl2 and then with potassium phthalimide (V) affords the substituted phthalimide (VI), which by treatment with hydrazine hydrate in refluxing methanol gives the 2(Z)-butenamine (VII). The condensation of (VII) with 2-(2-furylmethylsulfinyl)acetic acid 4-nitrophenyl ester (VIII) in THF yields the expected amide (IX), which is treated with p-toluenesulfonic acid in refluxing acetone/water to eliminate the ethylene ketal protecting group yilding the aldehyde (X). Finally, this compound is reductocondensed with piperidine (XI) by means of NaBH4 in ethanol.
CLIP
Synthesis Path
References
References
- ^ Jump up to:a b UCB Japan Revised: April 2005 Stogar tablets
- ^ Jump up to:a b c d e Zuventus Healthcare Ltd. India Lafaxid tablets
-
- a EP 582 304 (Fujirebio; 5.8.1993; J-prior. 7.8.1992).
-
preparation of 2-benzenesulfonyl-4-methylpyridine:
- EP 931 790 (Kuraray; 26.1.1999; J-prior. 26.1.1998).
-
chlorination of 2-benzenesulfonyl-4-methylpyridine:
- JP 10 231 288 (Kuraray; 2.9.1998; J-prior. 21.2.1997).
- WO 9 626 188 (Sagami Res. Center; 21.2.1996; J-prior. 22.2.1995).
- b EP 282 077 (Fujirebio; 11.3.1988; J-prior. 13.3.1987).
- US 4 912 101 (Fujirebio; 27.3.1990; J-prior. 13.3.1987).
-
preparation of I:
- JP 10 231 288 (Kuraray; 2.9.1998; J-prior. 21.2.1997).
-
chlorination of 2-chloromethylpyridines forming 2-chloro-4-trichloromethylpyridine:
- EP 557 967 (Central Glass Co.; 1.9.1993; J-prior. 24.2.1993).
-
treatment of I with (Z)-4-(tetrahydro-2H-pyran-2-yloxy)-2-buten-1-ol:
- US 5 382 589 (Fujirebio; 17.1.1995; J-prior. 27.1.1992).
-
preparation of furfuryl acetate and derivatives:
- JP 8 198 844 (Fujirebio; 6.8.1996; J-prior. 23.1.1995).
- JP 8 198 843 (Fujirebio; 6.8.1996; J-prior. 23.1.1995).
- JP 07 010 860 (Central Glass Co.; 13.1.1995; J-prior. 25.6.1993).
- JP 07 010 864 (Central Glass Co.; 13.1.1995; J-prior. 25.6.1993).
-
2-(furfurylsulfinyl)acetic acid nitrophenyl ester:
- JP 07 010 862 (Central Glass Co.; 13.1.1995; J-prior. 25.6.1993).
-
4-(tetrahydro-2-pyranyloxy)-2(Z)-buten-1-ol from 2(Z)-butene-1,4-diol:
- Nishiguchi, T. et al.: J. Org. Chem. (JOCEAH) 63, 23, 8183 (1998).
- Davis, K. J. et al.: Synth. Commun. (SYNCAV) 29, 10, 1679 (1999).
- Nishiguchi, T. et al.: J. Chem. Soc., Perkin Trans. 1 (JCPRB4) 1995, 24, 2491.
 |
|
| Clinical data | |
|---|---|
| AHFS/Drugs.com | International Drug Names |
| Routes of administration |
Oral |
| ATC code | |
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ECHA InfoCard | 100.118.935 |
| Chemical and physical data | |
| Formula | C22H29N3O4S |
| Molar mass | 431.54 g/mol |
| 3D model (JSmol) | |
/////////////////LAFUTIDINE, ラフチジン , FRG-8813, ATC:A02B
C1CCN(CC1)CC2=CC(=NC=C2)OCC=CCNC(=O)CS(=O)CC3=CC=CO3

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....