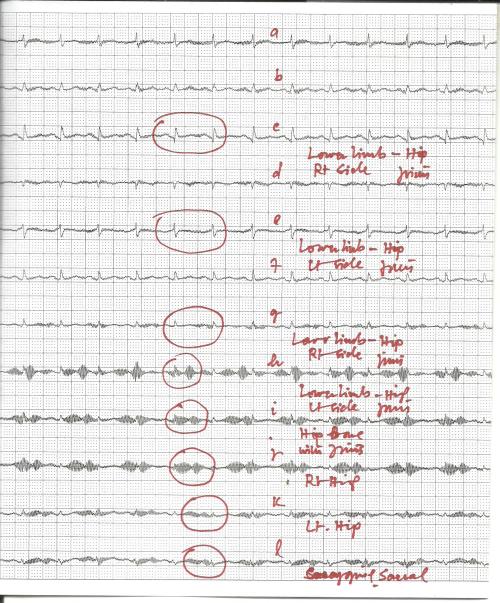
Grape Seeds Fight Bowel Cancer
University of Adelaide research has shown for the first time that grape seed can aid the effectiveness of chemotherapy in killing colon cancer cells as well as reducing the chemotherapy’s side effects.
Published in the prestigious journal PLOS ONE, the researchers say that combining grape seed extracts with chemotherapy has potential as a new approach for bowel cancer treatment – to both reduce intestinal damage commonly caused by cancer chemotherapy and to enhance its effect.
Lead author Dr Amy Cheah says there is a growing body of evidence about the antioxidant health benefits of grape seed tannins or polyphenols as anti-inflammatory agents and, more recently, for their anti-cancer properties.
“This is the first study showing that grape seed can enhance the potency of one of the major chemotherapy drugs in its action against colon cancer cells,” says Dr Cheah, researcher in the School of Agriculture, Food and Wine.
“Our research…
View original post 308 more words
Tadalafil (cialis)

Tadalafil
GF-196960, IC-351, Cialis
6R–trans)-6-(1,3-benzodioxol-5-yl)- 2,3,6,7,12,12a-hexahydro-2-methyl-pyrazino [1′, 2′:1,6] pyrido[3,4-b]indole-1,4-dione
Pyrazino[1′,2′:1,6]pyrido[3,4-b]indole-1,4-dione,6-(1,3-benzodioxol-5-yl)-2,3,6,7,12,12a-hexahydro-2-methyl-, (6R-trans)-; (6R,12aR)-6-(1,3-benzodioxol-5-yl)-2,3,6,7,12,12a-hexahydro-2-ethylpyrazino[1′,2′:1,6]pyrido[3,4-b]indole-1,4-dione; GF 196960; Adcirca;
171596-29-5 casno
Molecular Weight:
389.40
Molecular Formula:C22H19N3O4
GlaxoSmithKline (Originator), Lilly Icos (Marketer), Lilly (Licensee), Lilly Icos (Licensee)
Launched-2003
Tadalafil is currently marketed as Cialis. Cialis was developed by Eli Lilly as a treatment for impotence. In this capacity, it is reported that tadalafil functions by inhibiting the formation of cyclic guanosine monophosphate (cGMP)-specific phosphodiesterase type 5 (PDE5). The inhibition of PDE5 presumably lessens impotence by increasing the amount ot c(iMP, resulting in smooth muscle relaxation and increased blood flow.

Tadalafil is a PDE5 inhibitor marketed in pill form for treating erectile dysfunction (ED) under the name Cialis, and under the name Adcirca for the treatment of pulmonary arterial hypertension. In October 2011 the U.S. Food and Drug Administration (FDA) approved Cialis for treating the signs and symptoms of benign prostatic hyperplasia (BPH) as well as a combination of BPH and erectile dysfunction (ED) when the conditions coincide. It initially was developed by the biotechnology company ICOS, and then again developed and marketed world-wide by Lilly ICOS, LLC, the joint venture of ICOS Corporation and Eli Lilly and Company. Cialis tablets, in 2.5 mg, 5 mg, 10 mg, and 20 mg doses, are yellow, film-coated, and almond-shaped. The approved dose for pulmonary arterial hypertension is 40 mg (two 20-mg tablets) once daily.
Tadalafil can be prepared via a series of intermediates. One synthesis scheme is illustrated in Scheme 1: Scheme 1
U.S. Patent No. 5,859,006 describes the synthesis of the tadalafil intermediate (Compound III) from D-tryptophan methyl ester (Compound II) and piperonal (Compound I) using trifluoroacetic acid and dichloromethane, a halogenated solvent. Compound III is then reacted with chloroacetyl chloride (Compound IV) and chloroform, providing another intermediate of tadalafil (Compound V). WO 04/011463 describes a process of preparing tadalafil intermediates from D-tryptophan methyl ester HCl salt and piperonal by refluxing the reagents in isopropyl alcohol; the obtained intermediate is reacted with chloroacetyl chloride and THF, resulting in another intermediate of tadalafil.
Tadalafil is also manufactured and sold under the name of Tadacip by the Indian pharmaceutical company Cipla in doses of 10 mg and 20 mg.
On November 21, 2003 the FDA approved tadalafil (as Cialis) for sale in the United States as the third ED prescription drug pill (after sildenafil citrate(Viagra) and vardenafil (Levitra)). Like sildenafil and vardenafil, tadalafil is recommended as an ‘as needed’ medication. Cialis is the only one of the three that is also offered as a once-daily medication.
Moreover, tadalafil was approved in May 2009 in the United States for the treatment of pulmonary arterial hypertension and is under regulatory review in other regions for this condition. In late November 2008, Eli Lilly sold the exclusive rights to commercialize tadalafil for pulmonary arterial hypertension in the United States to United Therapeutics for an upfront payment of $150 million.

The FDA’s approval of Viagra (Sildenafil) on March 27, 1998 was a ground-breaking commercial event for the treatment of ED, with sales exceedingUS$1 billion. Subsequently, the FDA approved Levitra (vardenafil) on August 19, 2003, and Cialis (tadalafil) on November 21, 2003.
Cialis was discovered by Glaxo Wellcome (now GlaxoSmithKline) under a partnership between Glaxo and ICOS to develop new drugs that began in August 1991. [1][2] In 1993, the Bothell, Washington biotechnology company ICOS Corporation began studying compound IC351, a phosphodiesterase type 5 (PDE5) enzyme inhibitor. In 1994, Pfizer scientists discovered that sildenafil, which also inhibits the PDE5 enzyme, caused penile erection in men participating in a clinical study of a heart medicine. Although ICOS scientists were not testing compound IC351 for treating ED, they recognized its potential usefulness for treating that disorder. Soon, in 1994, ICOS received a patent for compound IC351 (structurally unlike sildenafil and vardenafil), and Phase 1 clinical trials began in 1995. In 1997, the Phase 2 clinical studies were initiated for men experiencing ED, then progressed to the Phase 3 trials that supported the drug’s FDA approval. Although Glaxo had an agreement with ICOS to share profits 50/50 for drugs resulting from the partnership, Glaxo let the agreement lapse in 1996 as the drugs developed were not in the company’s core markets.[3]

In 1998, ICOS Corporation and Eli Lilly and Company formed the Lilly ICOS, LLC, joint venture company to further develop and commercialize tadalafil as a treatment for ED. Two years later, Lilly ICOS, LLC, filed a new drug application with the FDA for compound IC351 (under the tadalafil generic name, and the Cialis brand name). In May 2002, Lilly ICOS reported to the American Urological Association that clinical trial testing demonstrated that tadalafil was effective for up to 36 hours, and one year later, the FDA approved tadalafil. One advantage Cialis has over Viagra and Levitra is its 17.5-hour half-life (thus Cialis is advertised to work for up to 36 hours, after which time there remains approximately 25 percent of the absorbed dose in the body) when compared to the four-hour half–life of sildenafil (Viagra).
In 2007, Eli Lilly and Company bought the ICOS Corporation for $2.3 billion. As a result, Eli Lilly owned Cialis and then closed the ICOS operations, ending the joint venture and firing most of ICOS’s approximately 500 employees, except for 127 employees of the ICOS biologics facility, which subsequently was bought by CMC Biopharmaceuticals A/S (CMC).
Persons surnamed “Cialis” objected to Eli Lilly and Company’s so naming the drug, but the company has maintained that the drug’s trade name is unrelated to the surname.[4]
On October 6, 2011, the U.S. FDA approved tadalafil [5] to treat the signs and symptoms of benign prostatic hyperplasia (BPH). BPH is a condition in males in which the prostate gland becomes enlarged, obstructing the free flow of urine. Symptoms may include sudden urges to urinate (urgency), difficulty in starting urination (hesitancy), a weak urine stream, and more frequent urination- especially at night. The FDA has also approved tadalafil for treatment of both BPH and erectile dysfunction (ED) where the two conditions co-exist.
Although available since 2003 in 5, 10, 20 mg dosage, in late 2008/early 2009, the U.S. FDA approved the commercial sale of Cialis in 2.5 mg dosage as a one-a-day treatment for ED. The 2.5 mg dose avoids earlier dispensing restrictions on higher dosages. The price of the 5 mg and 2.5 mg are often similar, so some people score and split the pill.[6] The manufacturer does not recommend splitting.
Moreover, tadalafil (Adcirca) 40 mg was approved in 2009 in the United States and Europe (and 2010 in Canada and Japan) as a once-daily therapy to improve exercise ability in patients withpulmonary arterial hypertension. In patients with pulmonary arterial hypertension, the pulmonary vascular lumen is decreased as a result of vasoconstriction and vascular remodeling, resulting in increased pulmonary artery pressure and pulmonary vascular resistance. Tadalafil is believed to increase pulmonary artery vasodilation, and inhibit vascular remodeling, thus lowering pulmonary arterial pressure and pulmonary vascular resistance. Right heart failure is the principal consequence of pulmonary arterial hypertension.
On October 6, 2011, the U.S. FDA approved tadalafil [6] to treat the signs and symptoms of benign prostatic hyperplasia (BPH). BPH is a condition in males in which the prostate gland becomes enlarged, obstructing the free flow of urine. Symptoms may include sudden urges to urinate (urgency), difficulty in starting urination (hesitancy), a weak urine stream, and more frequent urination- especially at night. The FDA has also approved tadalafil for treatment of both BPH and erectile dysfunction (ED) where the two conditions co-exist.
Tadalafil has been used in approximately 15,000 men participating in clinical trials, and over eight million men worldwide (primarily in the post-approval/post-marketing setting). The most commonside effects when using tadalafil are headache, stomach discomfort or pain, indigestion, burping, acid reflux. back pain, muscle aches, flushing, and stuffy or runny nose. These side effects reflect the ability of PDE5 inhibition to cause vasodilation (cause blood vessels to widen), and usually go away after a few hours. Back pain and muscle aches can occur 12 to 24 hours after taking the drug, and the symptom usually disappears after 48 hours.
In May 2005, the U.S. Food and Drug Administration found that tadalafil (along with other PDE5 inhibitors) was associated with vision impairment related to NAION (nonarteritic anterior ischemic optic neuropathy) in certain patients taking these drugs in the post-marketing (outside of clinical trials) setting. Most, but not all, of these patients had underlying anatomic or vascular risk factors for development of NAION unrelated to PDE5 use, including: low cup to disc ratio (“crowded disc”), age over 50, diabetes, hypertension, coronary artery disease, hyperlipidemia and smoking. Given the small number of NAION events with PDE5 use (fewer than one in one million), the large number of users of PDE5 inhibitors (millions) and the fact that this event occurs in a similar population to those who do not take these medicines, the FDA concluded that they were not able to draw a cause and effect relationship, given these patients underlying vascular risk factors or anatomical defects. However, the label of all three PDE5 inhibitors was changed to alert clinicians to a possible association.
In October 2007, the FDA announced that the labeling for all PDE5 inhibitors, including tadalafil, requires a more prominent warning of the potential risk of sudden hearing loss as the result of postmarketing reports of deafness associated with use of PDE5 inhibitors.[7]
Selectivity compared with other PDE5 inhibitors
Tadalafil, sildenafil, and vardenafil all act by inhibiting the PDE5 enzyme. These drugs also inhibit other PDE enzymes. Sildenafil and vardenafil inhibit PDE6, an enzyme found in the eye, more than tadalafil.[9] Some sildenafil users see a bluish tinge and have a heightened sensitivity to light because of PDE6 inhibition.[3] Sildenafil and vardenafil also inhibit PDE1 more than tadalafil.[9]PDE1 is found in the brain, heart, and vascular smooth muscle.[9] It is thought that the inhibition of PDE1 by sildenafil and vardenafil leads to vasodilation, flushing, and tachycardia.[9] Tadalafil inhibits PDE11 more than sildenafil or vardenafil.[9] PDE11 is expressed in skeletal muscle, the prostate, the liver, the kidney, the pituitary gland, and the testes.[9] The effects on the body of inhibiting PDE11 are not known.[9]
In the United States, the FDA relaxed rules on prescription drug marketing in 1997, allowing advertisements targeted directly to consumers.[10] Lilly-ICOS hired the Grey Worldwide Agency in New York, part of the Grey Global Group, to run the Cialis advertising campaign.[11] Marketers for Cialis has taken advantage of its greater duration compared to its competitors in advertisements for the drug; Stuart Elliot of The New York Times opined: “The continuous presence of women in Cialis ads is a subtle signal that the drug makes it easier for them to set the pace with their men, in contrast to the primarily male-driven imagery for Levitra and Viagra.”[11] Iconic themes in Cialis ads include couples in bathtubs and the slogan “When the moment is right, will you be ready?”[11] Cialis ads were unique among the ED drugs in mentioning specifics of the drug.[12] As a result, Cialis ads were also the first to describe the side effects in an advertisement, as the FDA requires advertisements with specifics to mention side effects. One of the first Cialis ads aired at the 2004 Super Bowl.[12] Just weeks before the Super Bowl, the FDA required more possible side effects to be listed in the advertisement, including priapism.[12] Although many parents objected to the Cialis ad being aired during the Super Bowl, Janet Jackson‘s halftime “wardrobe malfunction” overshadowed Cialis.[12] In January 2006, the Cialis ads were tweaked, adding a doctor on screen to describe side effects and only running ads where more than 90 percent of the audience are adults, effectively ending Super Bowl ads.[10] In 2004, Lilly-ICOS, Pfizer, and GlaxoSmithKline spent a combined $373.1 million to advertise Cialis, Viagra, and Levitra respectively.[12] Cialis has sponsored many golf events, including the America’s Cup and the PGA Tour, once being title sponsor of the PGA Tour Western Open tournament.[13]
CIALIS (tadalafil) is a selective inhibitor of cyclic guanosine monophosphate (cGMP)-specific phosphodiesterase type 5 (PDE5). Tadalafil has the empirical formula C22H19N3O4 representing a molecular weight of 389.41. The structural formula is:
 |
The chemical designation is pyrazino[1′,2′:1,6]pyrido[3,4-b]indole-1,4-dione, 6-(1,3-benzodioxol-5-yl)2,3,6,7,12,12a-hexahydro-2-methyl-, (6R,12aR)-. It is a crystalline solid that is practically insoluble in water and very slightly soluble in ethanol.
CIALIS is available as almond-shaped tablets for oral administration. Each tablet contains 2.5, 5, 10, or 20 mg of tadalafil and the following inactive ingredients: croscarmellose sodium, hydroxypropyl cellulose, hypromellose, iron oxide, lactose monohydrate, magnesium stearate, microcrystalline cellulose, sodium lauryl sulfate, talc, titanium dioxide, and triacetin.
Tadalafil, (6R-trans)-6-(l,3-benzodioxol-5-yl)-2,3,6,7,12,12a-hexahydro-2- methyl-pyrazino[r,2′:l,6]pyrido[3,4-b]indole-l,4-dione, with the structural formula shown below, is a white crystalline powder. (CAS# 171596-29-5). Tadalafil is a potent and selective inhibitor of the cyclic guanosine monophosphate (cGMP) – specific phosphodiesterase enzyme, PDE5. The inhibition of PDE5 increases the amount of cGMP, resulting in smooth muscle relaxation and increased blood flow. Tadalafil is therefore currently used in the treatment of male erectile dysfunction, and is commercially available as CIALIS ®.
Tadalafil U.S. Patent No. 5,859,006 describes the synthesis of tadalafil via the cyclization of TDCL (i.e., cis-methyl l,2,3,4-tetrahydro-2-chloroacetyl-l-(3,4- methylenedioxyphenyl)-9H-pyrido[3,4-b]mdole-3-carboxylate) using methylamine by purification by flash chromatography, followed by subsequent crystallization from methanol. Crude tadalafil typically requires additional purification steps, such as multiple extractions, crystallization, and/or flash chromatography, to remove the impurities present in the compound after synthesis is complete. Such purification processes increase the cost of producing tadalafil. Also, when repeating the US ‘006 process, about 250 volumes of methanol were necessary for the crystallization step
Tadalafil can be prepared via a series of intermediates. One synthesis for preparing tadalafil is illustrated below in Scheme I:
SCHEME I
U.S. Patent No. 5,859,006 discloses the synthesis of a tadalafil intermediate
(Compound III) from D-tryptophan methyl ester (Compound II) and piperonal (Compound
I) using trifluoroacetic acid and dichloromethane, a halogenated solvent. Compound III is then reacted with chloroacetyl chloride (Compound IV) and chloroform to provide another intermediate of tadalafil (Compound V).
WO 2004/011463 discloses a process of preparing tadalafil intermediates from D-tryptophan methyl ester HCl salt and piperonal by refluxing the reagents in isopropyl alcohol, reacting the intermediate thus obtained with chloroacetyl chloride and tetrahydrofuran (THF) to provide another intermediate of tadalafil.
WO 2006/110893 discloses a process for the preparation of methyl ester intermediate (Compound III), and tadalafil using the methyl ester intermediate (CompoundII).
U.S. Patent Application Publication No. 2006/0258865 Al discloses a synthesis of the tadalafil intermediate (Compound III) from D-tryptophan methyl ester
(Compound II) and piperonal (Compound I) using a dehydrating agent selected from Na2SO4, K2SO4, MgSO4, CaSO4, CaCl2, molecular sieve or mixtures thereof and a high boiling solvent such as N,N-Dimethyl acetamide. Compound III is then reacted with chloroacetyl chloride (Compound IV) in the presence of a base such as NaHCO3 and an organic solvent such as dichloromethane, providing another intermediate of tadalafil (Compound V), which is further reacted with aqueous methyl amine solution to provide tadalafil.
………………………………………….
Scheme II and III.
……………………………………………………………………………………………………a compound of .Formula I
SCHEME III
SCHEME IV
EXAMPLE l
The reaction scheme of this example is generally shown below in SchemeIV.
SCHEME IV
Compound – 1 Compound – II
Into a clean dry glass flask charged with ethanol (250 ml) under a nitrogen atmosphere was added Compound 1 (25 g) under stirring. The reaction mass was cooled to 0 to 50C and monomethylamine gas was purged into the reaction mixture for about 2 hours while maintaining the temperature between 0 to 50C. The temperature was raised to 75 to 😯0C and the reaction mixture was stirred under reflux for 2 hours. The reaction mixture was then cooled to 0 to 5°C and monomethylamine gas was again purged into the reaction mixture at 0 to 5°C. The temperature was again raised to 75 to 800C and stirred for about 1 hour. The reaction mixture was concentrated under vacuum to about 1/3 its original volume, cooled to 5 to 1O0C and stirred for 1 hour at this temperature. The solids were filtered and washed with chilled ethanol (50 ml). The wet solids were dried under vacuum for 6 hours.
Yield: 25g; Mp: 202-206.70C
Specific rotation (25°C) :+44.0 ( C=l% in DMSO)
13C NMR, DMSO-D6 : 25.78, 25.92, 57.89, 57.98, 101.17, 108.09, 108.32,
109.08, 111.48, 117.82, 118.62, 122.23, 122.97, 126.97, 135.97, 136.22, 136.55, 146.99,
147.48, 173.13
1H NMR, DMSO-D6, 300 MHz, Delta values: 2.6(m,lH), 2.7(m,3H),
2.8(d,lH), 3.0(d,lH), 3.6(bs,lH), 5.1(m,lH), 6.0(s,3H), 6.9-7.1(m, 5H), 7.2(d,lH),
7.4(d,lH), 7.8(bs, IH), 10.3(s, IH)
EXAMPLE 2
The reaction scheme of this example is generally shown below in SchemeV.
SCHEME V
Formula III Formula II
Into a clean dry flask charged with dichloromethane (200 ml) was added
Compound II (25 g) obtained in Example 1 under stirring at 25 to 300C. Next, triethylamine (16.11 g) was added to the reaction mixture and stirred for 30 minutes at 20 to 300C. The reaction mixture was cooled to 0 to 5°C and a solution of chloroacetyl chloride (12.93 g) in chloroform (50 ml) was added to the reaction mixture while maintaining temperature between -5 to 50C. The reaction mixture was stirred at -5 to 5°C for about 2 hours. Saturated aqueous sodium bicarbonate solution (50 ml) was added to the reaction mass slowly and the temperature of the reaction mixture was raised to 25 to 300C. The lower organic layer was separated and washed twice with water (75 ml). The chloroform extract was dried over anhydrous sodium sulfate. The organic layer was concentrated under vacuum until a thick yellow slurry was obtained. The slurry was cooled to 0 to5°C. The solids obtained were filtered and washed with 50 ml chilled chloroform. The wet product was dried at 750C under vacuum for 6 hours.
Yield: 22.5 g; HPLC Purity: 97%; Mp: 180-1820C
Specific rotation(25°C): -154.3(C=1% in DMSO)
13C. NMR(DMSO-Do, 300 MHZ)= 21.11, 25.88, 44.207, 51.60, 53.95,
101.16,107.66 109.56, 111.38, 118.36, 118.75, 121.58,122.74, 126.30, 130.31, 134.13,
136.57, 146.66, 147.03,167.43, 168.45
1H. NMR (CDC13, 300 MHZ):2.4(bs,3H), 3.1(m,lH), 3.8(m,lH),
4.3(bs,2H), 4.9(m,lH), 5.4(m,lH), 5.9(s,2H), 6.6-6.8(m,3H), 6.9(bs,lH), 7.1-7.3(m,3H),
7.6(d, IH), 7.7(bs,lH)
1H. NMR (DMSO-D6, 300 MHZ): 2.0 (bs,3H), 2.9(m,lH), 3.4(m,lH),
4.5(m,lH), 4.8(m,lH), 4.9(m,lH), 6.0(m,2H), 6.4-6.8(m,4H), 6.9-7.2(m,2H), 7.3(d, IH),
7.4(bs,lH), 7.5(d,lH), 10.8(s,lH)
EXAMPLE 3
The reaction scheme of this example is generally shown below in SchemeVI.
SCHEME VI
Formula II Formula I
Into a clean dry round bottom (RB) flask was charged tetrahydrofuran
(THF) (175 ml) under a nitrogen blanket and then cooled to -35 to -400C. Next 92 ml n- butyllithium (1.6 m solution in hexane) was added while maintaining the temperature between -35 to -400C. After the addition was complete, the reaction mixture was stirred at -35 to -400C for 15 minutes. A solution of compound of formula II (22.5 g) obtained in Example 2 in THF (75 ml) was prepared and slowly added to the reaction mixture while maintaining the temperature between -35 to -400C. After the addition was complete, the reaction mixture was stirred at -35 to -400C for 2.5 hours. Saturated aqueous ammonium chloride solution (25 ml) and 50 ml ethyl acetate was added to the reaction mixture at -35 to -400C. The temperature was raised to 25 to 300C and the two layers formed were separated. The upper organic layer was collected. The lower aqueous layer was thrice extracted with ethyl acetate (25 ml). The organic layers were combined together and washed with water (50 ml). The organic extract was dried over anhydrous sodium sulfate and concentrated under vacuum to obtain crude tadalafil as a dark brown solid. [0058] Yield: 22 g; HPLC Purity: 50%.
EXAMPLE 4
Purification of crude tadalafil
The crude tadalafil (22 g) obtained in Example 3 was suspended in 110 ml methanol and stirred for 1 hour at 25 to 300C. The solids obtained were filtered and washed with 25 ml chilled methanol. The wet product was dried at 600C under vacuum for 6 hours. This was further purified by using isopropyl alcohol. Yield: 9 g; HPLC Purity: >99.5%.
EXAMPLE 5
The reaction scheme of this example is generally shown below in SchemeVII.
Scheme VII
Formula VI where R = -OCH3 Formula VIA [0062] Into a clean dry RB flask charged with methanol (1900 ml) was added D- tryptophan methyl ester (190 g) under stirring at 25 to 300C. The reaction mixture was cooled to 0 to 50C. Monomethylamine gas was purged into the reaction mixture at 0 to 5°C for about 5-7 hours under stirring. The temperature of the reaction mixture was slowly raised to about 25 to 3O0C and stirred at this temperature for 5-7 hours. The reaction mixture was concentrated under vacuum to distill out the solvent. Diisopropyl ether (950 ml) was added and cooled to 25 to 3O0C under stirring for 1-2 hrs. The solids obtained were filtered, washed with Diisopropyl ether and dried under vacuum. [0063] MP: 122.4-1240C; Yield: 150 g (78.9 % w/w).
Specific rotation(25°C): +12.5 (C=I % in DMSO)
13 C NMR (300 MHZ,DMSO-D6): 25.71, 31.40, 55.67, 110.93, 111.55,
118.42, 118.73, 121.09, 123.95, 127.66,136.44, 175.39.
1H NMR (300 MHZ,DMSO-D6): 1.6(bs,2H), 2.5(m,3H), 2.8(m,lH),
3.1(m,lH), 3.4(m, IH), 6.9-7.2(m,3H), 7.3(d,lH), 7.5(d,lH), 7.8(bs,lH), 10.8(bs,lH)
EXAMPLE 6
The reaction scheme of this example is generally shown below in Scheme VIII.
SCHEME VIII
Formula VIA Formula VII
Into a clean, dry flask charged with methylene dichloride (MDC) (1000 ml) was added D-tryptophan methyl amide, the compound of Formula VIA (50 g), and piperonal, the compound of Formula VII (31.09 g), under stirring at 25 to 300C. The reaction mixture was cooled to 0 to 5°Cunder nitrogen atmosphere. Trifluoroacetic acid (85.3 g) was dissolved in MDC (250 ml) and the solution was slowly added to the reaction mixture at 0 to 5°C. The temperature of the reaction mixture was raised to 20 to 300C and stirred at this temperature for 14-16 hours. The reaction was monitored by TLC, workup was done as follows, the pH of the reaction mixture was adjusted to 8-9 using sodium carbonate solution under stirring, the two layers were settled, separated and the lower MDC layer was washed with water. The MDC layer was then dried over anhydrous sodium sulfate. The reaction mass was concentrated under vacuum at 40 to 5O0C to remove the solvent. The compound was precipitated using ethyl acetate, the solids were filtered, washed with ethyl acetate and dried.
Yield: 52.5 g; Yield: 105% w/w, HPLC Purity: 71% cis and 27% trans isomer (HPLC).
EXAMPLE 7
The reaction scheme of this example is generally shown below in Scheme IX.
SCHEME IX
1]CICOCH2C1 2]crystn
Formula H
Into a clean dry flask charge with dichloromethane (400 ml) under a nitrogen atmosphere was added the compound of Formula III obtained in Example 6 and triethylamine (28.96 g) under stirring at 20 to 3O0C. The reaction mixture was then cooled to 0 to 50C. A mixture of chloroacetyl chloride (25.85 g) in dichloromethane (100 ml) was prepared and slowly added to the reaction mixture while maintaining the temperature between -5 to 50C in 1-2 hrs. The reaction mixture was stirred at 0 to 50C for 30 min and then saturated sodium bicarbonate solution (100 ml) was added at 5 to 100C under stirring. The temperature of the reaction mixture was raised to 25 to 300C and stirred at this temperature for 15 minutes. The layers were then separated. The lower MDC layer was collected, washed twice with 100 ml water and dried over anhydrous sodium sulfate. The
MDC layer was concentrated to distill out MDC until a stirrable mass was left behind. The mass was cooled to 25-3O0C and filtered, washed, to yield off-white to light yellow colored solids. The resulted product was the cis isomer, the trans isomer left behind in the mother liquor.
Yield = 25.5 g (50%w/w); HPLC Purity: > 97%.
The physical and spectral data was similar to that obtained in Example 2.
EXAMPLE 8
The reaction scheme of this example is generally shown below in SchemeX.
SCHEME X
Formula II
Into a clean dry round bottom (RB) flask was charged THF (1625 ml) under a nitrogen blanket and then cooled to -35 to -400C. Next, 505 ml n-butyllithium (1.6 m solution in hexane) was added while maintaining the temperature between -35 to -4O0C. After the addition was complete, the reaction mixture was stirred at -35 to -4O0C for 15 minutes. 72 ml diisopropyl amine was then added at -35 to -400C and then stirred at 0-50C for 1 hr. A solution of Compound of formula II (125 g) obtained in Example 7 in THF (625 ml) was prepared and slowly added to the reaction mixture while maintaining the temperature between -40 to -5O0C. After the addition was complete, the reaction mixture was stirred at -35 to -400C for 2-6 hours. Saturated aqueous ammonium chloride solution (250 ml) and ethyl acetate (125 ml) was added to the reaction mixture at -35 to -400C. The temperature was raised to 25 to 300C and the two layers formed were separated. The upper organic layer was collected. The lower aqueous layer was extracted with ethyl acetate (65 ml). The organic layers were combined together and distilled. Isopropyl alcohol (1250 ml) was added and the distillation was continued. A mixture of methanol (250 ml) and isopropanol (375 ml) were added and crude tadalafϊl was obtained upon cooling. The crude product was filtered, washed with water and dried. [0076] Yield: 60 g; (48% w/w); HPLC Purity: >99%.
EXAMPLE 9
Purification of crude Tadalafil
The crude tadalafil obtained in Example 8 was suspended in methanol (600 ml) and stirred for 1 hour at reflux. The mixture was cooled and the solids obtained were filtered and washed with chilled methanol (60 ml). The wet product was dried at under vacuum.
Yield: 56 g; HPLC Purity: 99.8%.
……………………………………………………………………………………
Beilstein J. Org. Chem. 2011, 7, 442–495.
http://www.beilstein-journals.org/bjoc/single/articleFullText.htm?publicId=1860-5397-7-57#S28
A different approach was used in the synthesis of the phosphodiesterase inhibitor tadalafil (132, Cialis) starting from commercially available (D)-tryptophan methyl ester to form the indolopiperidine motif 135 via a Pictet–Spengler reaction followed by a double condensation to install the additional diketopiperazine ring (Scheme 28) [38,39].
To achieve the high levels of cis selectivity required from the Pictet–Spengler reaction, an extensive investigation of solvents and the influence of additives was undertaken [40]. It was identified that the use of a specific 23 mol % of benzoic acid significantly increased the cis/trans ratio from a base level of 55:45 to 92:8 (16 h reaction time at ambient temperature) in an overall yield of 86%. It was also determined that more polar solvents such as acetonitrile and nitromethane preferentially solvated the trans product and thereby allowed the isolation of the ciscompound by precipitation. It was also shown that by heating the reaction mixture under reflux the product distribution could be driven to the thermodynamically more favoured cis isomer having both the ester and the piperonyl moiety in equatorial positions. Hence, after heating under reflux for 8 h the cis/trans ratio was found to be 99:1 and the product could be isolated in an overall yield of 91%. This work represents an impressive example of a well considered and executed process optimisation study.
………………………………
The process disclosed in the patent US 5 859 006 (Scheme 1) involves condensation of D-tryptophan methyl ester with a piperonal derivative to yield a compound of formula (II). After conversion into a thioamide derivative of formula (III), cyclization occurs in presence of both an alkylating and reducing agents to provide a tetrahydro-β-carboline derivative of formula (IV), which on treatment with chloroacetyl chloride and methyl amine, gives Tadalafil. The compound of formula (IV) can also be obtained in one step, after separation of the other diastereoisomer, by a Pictet Spengler reaction between D-tryptophan methyl ester and piperonal in presence of an acid, such as trifluoroacetic acid.
-
The patent application WO2007/10038 discloses the reaction of D-tryptophan with piperonal to provide a tetrahydro-β-carboline acid that was cyclised to Tadalafil in presence of a sarcosine derivative.
The patent application WO2007/1107 discloses the reaction of D-tryptophan methyl amide with piperonal, to provide an intermediate that after reaction with chloroacetyl chloride cyclises to Tadalafil in presence of butyllithium.
Thus, the active substance prepared by the processes known up till now can only be obtained in a satisfactory quality after running through a large number of process steps. Moreover a toxic alkylating agent, such as methylamine, is often used.
Example 1
-
A 50 mL three-necked flask fitted with thermometer and reflux condenser was charged with (1R,3R)-methyl 1,2,3,4-tetrahydro-2-chloroacetyl-1-(3,4-methylenedioxyphenyl)-9H-pyrido [3,4-b] indole-3-carboxylate (VI) (1.39 g, 3.26 mmol), DMA (5.33 mL), K2CO3 (0.5 g, 3.6 mmol) and N-benzylmethylamine (0.41 mL, 3.26 mmol). The resultant solution was stirred at room temperature. After 2 hours, the mixture was poured in brine (20 mL) and extracted with isopropyl acetate. The combined organic phases were washed with brine (3 x 5 mL), dried over sodium sulfate and concentrated to a residue under reduced pressure, affording 1.5 g of the desired product (VII), as a white solid. Yield: 70%.
1H NMR (d6-DMSO 300 MHz, 298K) 2.24 (s, 3H), 2.94-3.00 (m, 5H), 3.44-3.68 (m, 3H), 5.56 (bd, J = 6.4, 1H), 5.95 (s, 1H), 5.96 (s, 1H), 6.55 (bd, J = 7.4, 1H), 6.75 (bs, 1H), 6.77 (d, J = 8.0, 1H), 6.84 (bs, 1H), 7.05 (td, J = 7.4, 0.9, 1H), 7.12 (td, J = 7.5, 1.2, 1H), 7.17-7.32 (m, 6H), 7.56 (d, J = 7.7, 1H), 10.76 (bs, 1H)
13C NMR (d6-DMSO 75.4 MHz, 298K) 21.9, 42.5, 51.3, 51.9, 52.4, 61.0, 61.7, 101.5, 107.0, 108.0, 109.8, 111.8, 118.5, 119.2, 122.0, 123.0, 126.7, 127.7, 128.7, 129.6, 131.1, 134.7, 137.1, 138.6, 147.1, 147.5, 170.6, 171.5
Example 2
-
(6R-trans)-6-(1,3-benzodioxol-5-yl)-2,3,6,7,12,12a-hexahydro-2-methyl-pyrazino [1′,2′:1,6] pyrido [3,4-b] indole-1,4-dione (Tadalafil) (I)
Under H2 atmosphere (3 atm) and magnetic stirring, Raney® Ni (2800 slurry in water, 0.0276 g, 0.47 mmol), previously washed with methanol (3 times), was added to a solution of (1R,3R)-methyl-1,2,3,4-tetrahydro-2-(2-(benzyl(methyl)amino)acetyl)-1-(3,4-methylenedioxyphenyl)-9H-pyrido[3,4-b]indole-3-carboxylate (VII) (3.00 g, 4.70 mmol) in DMA (21.3 mL). The mixture was heated at 90°C for 17 hours and then cooled to room temperature. The suspension was filtered over a pad of Celite® and the resulting solutionand the resulting solution was concentrated until 6 mL. Methanol (12 mL) was added and the solid which was so obtained was filtered over Buchner, washed with methanol (4 mL) and oven-dried under reduced pressure for 2 hours, affording 1.3 g of the title compound, as a white solid. Yield: 70%
1H NMR (d6-DMSO 300 MHz, 298K): 2.91-3.00 (m, 4H), 3.32 (s, 1H), 3.47-3.54 (dd, J = 4.6, 11.3, 1H), 3.93 (d, J = 17.1, 1H), 4.17 (d, J = 17.1, 1H), 4.35-4.40 (dd, J = 4.27, 11.6, 1H), 5.91 (s, 2H), 6.11 (s, 1H), 6.76 (s, 2H), 6.85 (s, 1H), 6.98-7.06 (m, 2H), 7.28 (d, J = 7.9, 1H), 7.52 (d, J = 7.3, 1H), 11.0 (s, 1H)
13C NMR (d6-DMSO 75.4 MHz, 298K) 23.8, 33.4, 52.0, 55.9, 56.1, 101.5, 105.3, 107.6, 108.6, 111.9, 118.7, 119.5, 119.9, 121.8, 126.4, 134.5, 136.8, 137.6, 146.7, 147.6, 167.1, 167.5………………………………………………………….
Synthesis pathway
Trade Names
Country Trade name Manufacturer Germany Cialis Lilly France Cialis – “- United Kingdom – “- – “- Italy – “- Eli Lilly USA – “- Lilly ICOS Ukraine Cialis Lilly del Caribe, Inc.., Puerto Rico (USA) Lilly SA (Packing), Spain Tadalafil Aurohem Laboratories Pvt. Ltd.., India Formulations
-
Tablets 10 mg, 20 mg
Links
-
EP 740 668 (Lab. Glaxo SA, Fr .; GB -prior. 21.01.1994).
-
US 6,140,329 (Lab. Glaxo SA, Fr .; GB -prior. 14/07/1995).
-
US 6,143,746 (Icos Corp .; 07/11/2001; GB -prior. 01.21.1994).
-
US 6,821,975 (Lilly ICOS; 23.11.2004; appl. 19.7.2002; USA-prior. 3.8.1999).
-
US 6,943,166 (Lilly ICOS; 13.9.2005; appl. 19.10.2001; USA-prior. 30.4.1999).
-
US 7,182,958 (Lilly ICOS; 27.2.2007; appl. 26.4.2000; USA-prior. 3.8.1999).
-
References
- Daugan, A; Grondin P, Ruault C, Le Monnier de Gouville AC, Coste H, Kirilovsky J, Hyafil F, Labaudinière R (October 9, 2003). “The discovery of tadalafil: a novel and highly selective PDE5 inhibitor. 1: 5,6,11,11a-tetrahydro-1H-imidazo[1′,5′:1,6]pyrido[3,4-b]indole-1,3(2H)-dione analogues”. Journal of Medicinal Chemistry 46 (21): 4525–32. doi:10.1021/jm030056e . PMID 14521414.
- Richards, Rhonda (September 17, 1991). “ICOS At A Crest On Roller Coaster”. USA Today. p. 3B.
- Ervin, Keith (June 21, 1998). “Deep Pockets + Intense Research + Total Control = The Formula — Bothell Biotech Icos Keeps The Pipeline Full Of Promise”. The Seattle Times. p. F1. Retrieved January 10, 2009.
- Revill, Jo (February 2, 2003). “Drugs giant says its new pill will pack more punch than rival Viagra”. The Observer. Retrieved 2007-04-06.
- http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm274642.htm
- https://www.consumerreports.org/health/resources/pdf/best-buy-drugs/money-saving-guides/english/PillSplitting-FINAL.pdf
- “FDA Announces Revisions to Labels for Cialis, Levitra and Viagra”. Food and Drug Administration. 2007-10-18. Retrieved 2009-09-28.
- “Cialis: Warnings, Precautions, Pregnancy, Nursing, Abuse”. RxList. 2007. Retrieved 2007-04-06.
- Bischoff, E (June 2004). “Potency, selectivity, and consequences of nonselectivity of PDE inhibition”. International Journal of Impotence Research 16: S11–4.doi:10.1038/sj.ijir.3901208 . PMID 15224129. Retrieved January 19, 2009.
- Elliott, Stuart (January 10, 2006). “For Impotence Drugs, Less Wink-Wink”. The New York Times. p. C2. Retrieved January 15, 2009.
- Elliott, Stuart (April 25, 2004). “Viagra and the Battle of the Awkward Ads”. The New York Times. p. 1. Retrieved January 15, 2009.
- McCarthy, Shawn (March 5, 2005). “First they tried to play it safe; Ads for erectile dysfunction drug Cialis bared all – including a scary potential side effect. It was risky but it has paid off”. The Globe and Mail. p. B4.
- Loyd, Linda (July 6, 2003). “Two Pills Look to Topple Viagra’s Reign in Market; Levitra Expects Approval Next Month, Cialis Later This Year”. The Philadelphia Inquirer. p. E01.
- 38 is 1 below
- 39 is 2 below
- 40 is 3 below
-
- daugan, A. C.-M. Tetracyclic Derivatives; Process of Preparation and Use. U.S. Patent 5,859,006, Jan 12, 1999.
- Daugan, A. C.-M. Tetracyclic Derivatives, Process of Preparation and Use. U.S. Patent 6,025,494, Feb 15, 2000.
- Shi, X.-X.; Liu, S.-L.; Xu, W.; Xu, Y.-L. Tetrahedron: Asymmetry 2008, 19, 435–442.doi:10.1016/j.tetasy.2007.12.017
DAUGAN A ET AL: “THE DISCOVERY OF TADALAFIL: A NOVEL AND HIGHLY SELECTIVE PDE5 INHIBITOR. 2: 2,3,6,7,12,12A-HEXAHYDROPYRAZINO[1′,2′:1,6 ÜPYRIDO[3,4-B ÜINDOLE-1,4-DIONE” JOURNAL OF MEDICINAL CHEMISTRY, AMERICAN CHEMICAL SOCIETY. WASHINGTON, US, vol. 46, no. 21, 2003, pages 4533-4542, XP008052656 ISSN: 0022-2623
- Tadalafil bound to proteins in the PDB
- National Institutes of Health – Medlineplus
- Material Safety Data Sheet PDF file
- Official Cialis (Tadalafil) Website
- U.S. National Library of Medicine: Drug Information Portal – Tadalafil
| WO2009004557A2 * | Jun 28, 2008 | Jan 8, 2009 | Ranbaxy Lab Ltd | A process for the preparation of intermediates of tetracyclic compounds |
| WO2009148341A1 | Jun 3, 2009 | Dec 10, 2009 | Zaklady Farmaceutyczne Polpharma Sa | Process for preparation of tadalafil |
| WO2012107549A1 | Feb 10, 2012 | Aug 16, 2012 | Interquim, S.A. | PROCESS FOR OBTAINING COMPOUNDS DERIVED FROM TETRAHYDRO-ß-CARBOLINE |
| EP2107059A1 | Mar 31, 2008 | Oct 7, 2009 | LEK Pharmaceuticals D.D. | Conversion of tryptophan into ß-carboline derivatives |
| US8445698 | Jun 28, 2008 | May 21, 2013 | Ranbaxy Laboratories Limited | Process for the preparation of an intermediate of tadalafil |
Flow chemistry approaches directed at improving chemical synthesis

The true potential of flow chemistry as an enabling technology can really only be fully appreciated when seen in the context of a target driven multi-step synthesis, aimed at the delivery of advanced chemical structures such as active pharmaceutical ingredients (APIs) .
As most pharmaceutical syntheses typically require between 8 and 10 chemical transformations (this is often somewhat reduced to 5/6 steps when analogue/library syntheses are being conducted), excluding protecting group manipulations, to realize the target molecule, this is a good foundation from which to explore the advantages of flow chemistry. We have generated a flow protocol for the synthesis of imatinib, the API of the Novartis block buster anticancer therapeutic Gleevec (imatinib mesylate), including a series of analogues (Scheme 11)
Furthermore, we aimed to create a route which would allow each of the three main fragments to be exchanged to address maximum variation in subsequent analogue synthesis. This requires additional planning to build flexibility into the sequence where this desired diversity can be easily introduced. Again, prior consideration of the generated intermediates, and any potential by-products that may arise, is critical and should be addressed prior to embarking on the synthesis.
Consequently, the extensive profiling of the reaction in terms of its purity profile is more closely analogous to process chemistry than traditional Medicinal Chemistry, even at the development stage. So, although more time consuming in the planning stage, having a greater understanding of the chemistry, does then enable a smoother up scaling and more rapid optimization of the route.

read all this at
http://www.degruyter.com/view/j/gps.2013.2.issue-3/gps-2013-0029/gps-2013-0029.xml
Flow chemistry approaches directed at improving chemical synthesis
1Department of Chemistry, Durham University, South Road, Durham, DH1 3LE, UK
Corresponding author: Ian R. Baxendale, Department of Chemistry, Durham University, South Road, Durham, DH1 3LE, UK
Citation Information: Green Processing and Synthesis. Volume 2, Issue 3, Pages 211–230, ISSN (Online) 2191-9550, ISSN (Print) 2191-9542, DOI: 10.1515/gps-2013-0029, May 2013

Fiduxosin ….An α1-Adrenoceptor antagonist
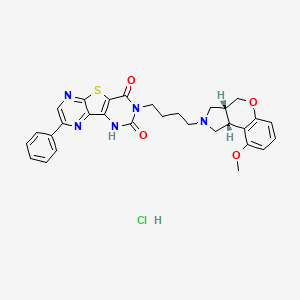
Fiduxosin hydrochloride, 208992-74-9, NCGC00162178-02, AC1L58WW,
208993-54-8 (free base)
 Fiduxosin
Fiduxosin-
Example 108
- 3-[4-((3aR,9bR)-cis -9-Methoxy-1,2,3,3a,4,9b-hexahydro-[1]-benzopyrano[3,4-c]pyrrol-2-yl)butyl]-8-(4-hydroxyphenyl)-pyrazino[2′,3′:4,5]thieno[3,2-d]pyrimidine-2,4(1H,3H)-dione
-
The product of Example 16 (0.07 g,0.105 mmol) and 4-(methoxymethyloxy) phenyl boronic acid (0.02 g, 0.11 mmol) prepared by the procedure described in Tetr.Lett., 31, 27, (1990) were treated as described in Example 106 to yield 0.029g(45%) of MOM-protected product. To the solution of this product (0.11g, 0.17 mmol) in CH3OH/THF was added 2N HCl (0.2ml) and the reaction mixture was refluxed for 1 hour. The reaction was evaporated and partitioned in NaHCO3 sol. and CH2Cl2/CH3OH to yield 0.005 g (51%) of the title compound.
-
1H NMR (500 MHz, CDCl3) d 1.81 (m, 2H), 1.98 (m, 2H), 2.25 (m, 1H), 2.65 (m, 1H), 2.88 (m, 1H), 3.08 (m, 2H), 3.22(m, 2H), 3.65 (m, 1H), 3.73 (m, 1H), 3.82 (s, 3H), 3.9 (m, 1H), 4.25 (m, 1H), 4.42 (m, 1H), 6.52 (m, 2H), 7.38 (m, 2H),7.49(m, 1H), 7.9 (t, 1H), 8.09 (d, 1H),9.12 (s, 1H);
-
MS(ESI)m/e 572 (M+H)+.
-
3-[4-((3aR,9bR)- cis -9-Methoxy-1,2,3,3a,4,9b-hexahydro-[1]-benzopyrano[3,4-c]pyrrol-2-yl)butyl]-8-chloro-pyrazino[2′,3′:4,5]thieno[3,2-d]pyrimidine-2,4(1H,3H)-dione hydrochloride
-
The product from Example 10 C (0.27 g, 1.0 mmol) and the product from Example 1E (0.20 g, 0.73 mmol) were treated as described in Example 1F to yield 0.29 g (77%) of the title compound: m.p. 220-222°;
-
1H NMR (300 MHz, CDCl3(free base)) δ 8.68 (s, 1H), 7.0 (t, 1H), 6.48 (d, 1H), 6.45 (d, 1H), 4.28 (m, 1H), 4.12 (m, 3H), 4.0 (m, 2H), 3.75 (s, 3H), 3.6 (m, 1H), 3.08 (m, 3H), 2.9 (m, 2H), 1.75 (m, 4H); MS (DCI/NH3) m/e 514(M+H)+;
-
Analysis calc’d for C24H24ClN5O4S·HCl·0.75H2O: C, 51.11; H, 4.74; N, 12.42; found: C, 51.09; H, 4.75; N, 12.43.
Fiduxosin (ABT-980), α1a-adrenoreceptor antagonist, a development compound at Abbot for the treatment of benign prostate hyperplasia, is disclosed in Organic Process Research & Development 2004, 8, 897-902 and references cited therein.
The synthetic route for preparation of Fiduxosin is as follows:

Fiduxosin (1) has been under development at Abbott Laboratories for the treatment of benign prostatic hyperplasia. A convergent strategy required methodologies for preparation of an enantiomerically pure 3,4-cis-disubstituted pyrrolidine and a 2,3,5-trisubstituted thienopyrazine in a regiospecific manner.
A [3+2] cycloaddition of an enantiopure azomethine ylide followed by a diastereoselective crystallization was employed to prepare the benzopyranopyrrolidine in high diastereomeric and enantiomeric purity. Conditions for reduction of an O-aryl lactone susceptible to epimerization were developed, and cyclization of the alcohol/phenol to the ether was accomplished in high yield.
The thienopyrazine was prepared by condensation of methyl thioglycolate and a regiospecifically prepared 2-bromo-3-cyano-5-phenylpyrazine. Conditions for effective halogen substitutive deamination to prepare regiospecific trisubstituted pyrazines will be described.
The mixture of 5 – and 6-phenyl regioisomers of 2-hydroxy-3-carboxamidopyrazine (IX) and (X), prepared by a known method, was treated with POCl3 and Et3N to produce the corresponding chloro nitriles (XI) and (XII ). Condensation of this mixture with methyl thioglycolate in the presence of NaOMe, followed by chromatographic separation of isomers furnished the desired thienopyrazine intermediate (XIII).
http://pubs.acs.org/doi/suppl/10.1021%2Fop049889k
…………………………………………………..
 Fiduxosin
Fiduxosin
……………………………………………………….
SYNTHESIS

Cycloaddition of the azomethine ylide resulting from N-trimethylsilylmethyl-N-methoxymethyl-(R)-alpha-methylbenzylamine (II) to 5-methoxycoumarin (I) produced the chiral cis-benzopyranopyrrole system (III). Lactone reduction by means of LiAlH4 or LiBH4 afforded diol (IV). After conversion of the primary alcohol of (IV) to either the corresponding chloride or the mesylate, cyclization in the presence of potassium tert-butoxide generated the tricyclic compound (V).
The alpha-methylbenzyl group of ( V) was removed by catalytic hydrogenation to give amine (VI), which was alkylated with 4-bromobutyronitrile yielding (VII). Reduction of the cyano group of (VII) using LiAlH4 in the presence AlCl3 or by catalytic hydrogenation in the presence of Raney -Ni produced the primary amine (VIII).
…………………………………………………

The mixture of 5 – and 6-phenyl regioisomers of 2-hydroxy-3-carboxamidopyrazine (IX) and (X), prepared by a known method, was treated with POCl3 and Et3N to produce the corresponding chloro nitriles (XI) and (XII ). Condensation of this mixture with methyl thioglycolate in the presence of NaOMe, followed by chromatographic separation of isomers furnished the desired thienopyrazine intermediate (XIII).
………………………………………………………….

In a regioselective synthetic method, phenyl glyoxime (XIV) was condensed with aminomalononitrile to produce the pyrazine N-oxide (XV). Reduction of the N-oxide of (XV) with triethyl phosphite yielded (XVI). Diazotization of the amino group of (XVI), followed by diazo displacement with CuBr2, furnished bromo pyrazine (XVII). This was then cyclized with methyl thioglycolate as above to yield the desired thienopyrazine intermediate (XIII).
………………………………………………….

In an alternative synthesis, phenylacetaldehyde (XVIII) was condensed with pyrrolidine (XIX) to give enamine (XX). Nitrosation of malononitrile (XXI), followed by treatment with tosyl chloride, produced the O-tosyl oxime (XXII). This was condensed with enamine (XX), and to the intermediate adduct (XXIII) was added thiophenol producing the phenylthiopyrazine (XXIV). Subsequent oxidation of the sulfide group of (XXIV) to sulfone (XXV), followed by condensation with methyl thioglycolate, gave the desired thienopyrazine (XIII).
……………………………………………………………..

The amino ester intermediate (XIII) was treated with phosgene and Et3N, and to the resulting isocyanate (XXVI) was added the primary amine (VIII), producing urea (XXVII). Then, cyclization of (XXVII) in refluxing toluene generated the desired compound,
fiduxosin
|
2-1-2002
|
Effect of fiduxosin, an antagonist selective for alpha(1A)- and alpha(1D)-adrenoceptors, on intraurethral and arterial pressure responses in conscious dogs.
|
The Journal of pharmacology and experimental therapeutics
|
|
|
2-1-2002
|
Modeling of relationships between pharmacokinetics and blockade of agonist-induced elevation of intraurethral pressure and mean arterial pressure in conscious dogs treated with alpha(1)-adrenoceptor antagonists.
|
The Journal of pharmacology and experimental therapeutics
|
|
|
1-1-2002
|
Effect of food on the pharmacokinetics of fiduxosin in healthy male subjects.
|
European journal of drug metabolism and pharmacokinetics
|
|
9-1-2012
|
Identification and analysis of hepatitis C virus NS3 helicase inhibitors using nucleic acid binding assays.
|
Nucleic acids research
|
|
|
3-1-2012
|
Small molecule screening identifies targetable zebrafish pigmentation pathways.
|
Pigment cell & melanoma research
|
|
|
7-1-2010
|
A small molecule inverse agonist for the human thyroid-stimulating hormone receptor.
|
Endocrinology
|
|
|
11-1-2009
|
A new homogeneous high-throughput screening assay for profiling compound activity on the human ether-a-go-go-related gene channel.
|
Analytical biochemistry
|
|
|
10-1-2009
|
Genetic mapping of targets mediating differential chemical phenotypes in Plasmodium falciparum.
|
Nature chemical biology
|
|
|
5-1-2007
|
Chemical genetics reveals a complex functional ground state of neural stem cells.
|
Nature chemical biology
|
|
|
5-1-2006
|
Microsphere-based protease assays and screening application for lethal factor and factor Xa.
|
Cytometry. Part A : the journal of the International Society for Analytical Cytology
|
|
|
5-1-2002
|
Single- and multiple-dose pharmacokinetics of fiduxosin under nonfasting conditions in healthy male subjects.
|
Journal of clinical pharmacology
|
|
|
5-1-2002
|
Multiple dose pharmacokinetics of fiduxosin under fasting conditions in healthy elderly male subjects.
|
The Journal of pharmacy and pharmacology
|
|
|
2-1-2002
|
Preclinical pharmacology of fiduxosin, a novel alpha(1)-adrenoceptor antagonist with uroselective properties.
|
The Journal of pharmacology and experimental therapeutics
|
Scientists develop potential new drug treatment to tackle viruses

An international team of scientists have successfully developed a novel compound which early signs suggest might prevent a range of viruses from infecting humans. Researchers from Oxford, Beijing, Leeds and Innsbruck collaborated on the new inhibitor. It targets a group of viruses responsible for hand, foot and mouth disease, especially the EV71 virus. This viral group causes numerous epidemics in children, mainly in Asia, with roughly 10 million cases reported every year in China alone. Symptoms are usually mild but in some cases the disease can prove fatal – the Chinese government reported over 900 deaths in 2010. The disease is currently untreatable and is a major global threat to public health.
This discovery, published in Nature Structural and Molecular Biology, may also have important implications for combating other diseases. Hand, foot and mouth disease is caused by several closely related viruses, and the new compound is effective…
View original post 510 more words
Temozolomide 替莫唑胺

Temozolomide 替莫唑胺
Temozolomide is a DNA damage inducer.
4-methyl-5-oxo-2,3,4,6,8-pentazabicyclo[4.3.0]nona-2,7,9-triene-9-carboxamide
3,4-dihydro-3-methyl-4-oxoimidazo(5,1-d)-1,2,3,5-tetrazine-8-carboxamide
Methazolastone, Temodar, Temodal
CAS NO 85622-93-1
Molecular Weight: 194.15
MF C6H6N6O2
Cancer Research UK (Originator), Schering-Plough (Licensee), National Cancer Institute (Codevelopment)
NMR..http://file.selleckchem.com/downloads/nmr/S123702-Methazolastone-NMR-Selleck.pdf
HPLC.http://file.selleckchem.com/downloads/hplc/S123702-Methazolastone-HPLC-Selleck.pdf
Temozolomide is an antitumor agent indicated for treating patients with malignant glioma such as cancer, breast cancer, refractory anaplastic astrocytoma, i.e., patients at first relapse who have experienced disease progression in malignant glioma, glioblastoma multiform and anaplastic astrocytoma, on a drug regimen containing a nitrosourea and procarbazine.
Temozolomide preparations are sold on the US market as hard capsules containing 5 mg, 20 mg, 100 mg or 250 mg Temozolomide (marketed as Temodar® by Schering Corporation, Kenilworth, N.J., USA). In other markets it is sold as Temodal®.
Temozolomide (brand names Temodar and Temodal and Temcad) is an oral chemotherapy drug. It is an alkylating agent used for the treatment of Grade IV astrocytoma — an aggressive brain tumor, also known as glioblastoma multiforme — as well as for treating melanoma, a form of skin cancer.
Temozolomide is also indicated for relapsed Grade III anaplastic astrocytoma and not indicated for, but as of 2011 used to treatoligodendroglioma brain tumors in some countries, replacing the older (and less well tolerated) PCV (Procarbazine–Lomustine–Vincristine) regimen.
Temozolomide, 3-methyl-8-aminocarbonyl-imidazo[5,1-d]-1,2,3,5-tetrazin-4(3H)-one, is a known antitumor drug; see for example Stevens et al., J. Med. Chem. 1984, 27, 196-201, and Wang et al., J. Chem. Soc., Chem. Commun.,1994,1687-1688. Temozolomide, the compound of formula 1:
is described in U.S. Pat. No. 5,260,291 (Lunt et al.).
The synthesis of 1 by the process described in J. Med. Chem. 1984, 27, 196-201 is depicted in the scheme I below.
In this process, 5-amino-1H-imidazole-4-carboxamide (A) is converted into 5-diazo-1H-imidazole-4-carboxamide (B), which is then cyclized with methylisocyanate in dichloromethane to provide a high yield of temozolomide. However, this process requires isolation of the unstable and potentially dangerous 5-diazo-1H-imidazole-4-carboxamide (B). Moreover, methylisocyanate is a difficult reagent to handle and ship, especially on the industrial scale, and indeed is better avoided in industrial manufacture. Furthermore, the cycloaddition of methylisocyanate requires a very long reaction time: Table I in J. Med Chem.1984, 27,196-201, suggests 20 days. Additionally, Stevens et al mention that the cycloaddition of the methylisocyanate to the compound of the formula (B) can proceed through two different intermediates:
The production of I by the two processes described in J. Chem. Soc., Chem. Commun., 1994, 1687-1688 provides a low overall yield from 5-amino-1H-imidazole-4-carboxamide (A): less than 20% (unoptimized—about 17% through 5-diazo-1H-imidazole-4-carboxamide (B) and about 15% through 5-amino-N1-(ethoxycarbonylmethyl)-1H-imidazole-1,4-dicarboxamide (C)); Scheme II below
The agent was developed by Malcolm Stevens[1] and his team at Aston University in Birmingham,[2][3] Temozolomide is a prodrug and animidazotetrazine derivative of the alkylating agent dacarbazine. It has been available in the US since August 1999, and in other countries since the early 2000s.
The therapeutic benefit of temozolomide depends on its ability to alkylate/methylate DNA, which most often occurs at the N-7 or O-6 positions ofguanine residues. This methylation damages the DNA and triggers the death of tumor cells. However, some tumor cells are able to repair this type of DNA damage, and therefore diminish the therapeutic efficacy of temozolomide, by expressing a protein O6-alkylguanine DNA alkyltransferase (AGT) encoded in humans by the O-6-methylguanine-DNA methyltransferase (MGMT) gene.[4] In some tumors, epigenetic silencing of the MGMT gene prevents the synthesis of this enzyme, and as a consequence such tumors are more sensitive to killing by temozolomide.[5] Conversely, the presence of AGT protein in brain tumors predicts poor response to temozolomide and these patients receive little benefit from chemotherapy with temozolomide.[6]
- Nitrosourea- and procarbazine-refractory anaplastic astrocytoma
- Newly diagnosed glioblastoma multiforme
- Malignant prolactinoma
Temozolomide (sometimes referred to as TMZ) is an imidazotetrazine derivative of the alkylating agent dacarbazine. It undergoes rapid chemical conversion in the systemic circulation at physiological pH to the active compound, 3-methyl-(triazen-1-yl)imidazole-4-carboxamide (MTIC). Temozolomide exhibits schedule-dependent antineoplastic activity by interfering with DNA replication. Temozolomide has demonstrated activity against recurrent glioma. In a recent randomized trial, concomitant and adjuvant temozolomide chemotherapy with radiation significantly improves, from 12.1 months to 14.6 months, progression free survival and overall survival in glioblastoma multiforme patients.
Formulations
Temozolomide is available in the United States in 5 mg, 20 mg, 100 mg, 140 mg, 180 mg & 250 mg capsules. Now also available in an IV form for people who can not swallow capsules or who have insurance that does not cover oral cancer agents.
A generic version is available in the UK.
Further improvement of anticancer potency
Laboratory studies and clinical trials are investigating whether it might be possible to further increase the anticancer potency of temozolomide by combining it with other pharmacologic agents. For example, clinical trials have indicated that the addition of chloroquine might be beneficial for the treatment of glioma patients.[8] In laboratory studies, it was found that temozolomide killed brain tumor cells more efficiently when epigallocatechin gallate (EGCG), a component of green tea, was added; however, the efficacy of this effect has not yet been confirmed in brain tumor patients.[9]More recently, use of the novel oxygen diffusion-enhancing compound trans sodium crocetinate (TSC) when combined with temozolomide and radiation therapy has been investigated in preclinical studies [10] and a clinical trial is currently underway.[11]
Because tumor cells that express the MGMT gene are more resistant to killing by temozolomide, it was investigated[according to whom?] whether the inclusion of [[O6-benzylguanine]] (O6-BG), an AGT inhibitor, would be able to overcome this resistance and improve the drug’s therapeutic effectiveness. In the laboratory, this combination indeed showed increased temozolomide activity in tumor cell culture in vitro and in animal models in vivo.[12] However, a recently completed phase-II clinical trial with brain tumor patients yielded mixed outcomes; while there was some improved therapeutic activity when O6-BG and temozolomide were given to patients with temozolomide-resistant anaplastic glioma, there seemed to be no significant restoration of temozolomide sensitivity in patients with temozolomide-resistant glioblastoma multiforme.[13]
There are also efforts to engineer hematopoietic stem cells expressing the MGMT gene prior to transplanting them into brain tumor patients. This would allow for the patients to receive stronger doses of temozolomide, since the patient’s hematopoietic cells would be resistant to the drug.[14]
High doses of temozolomide in high grade gliomas have low toxicity, but the results are comparable to the standard doses.[15]
A case report suggests that temozolomide may be of use in relapsed primary CNS lymphoma.[16] Confirmation of this possible use seems indicated.
Temozolomide, 3-methyl-8-aminocarbonyl-imidazo[5,1-d]- 1 ,2,3,5-tetrazin- 4(3H)-one, is a known antitumor drug; see for example Stevens et al., J. Med. Chem. 1984, 27, 196-201 , and Wang et al., J. Chem. Soc, Chem. Commυn., 1994, 1687-1688. Temozolomide, the compound of formula 1 :
1 is described in U.S. Patent No. 5,260,291 (Lunt et al.).
The synthesis of 1 by the process described in J. Med. Chem. 1984, 27, 196- 201 is depicted in the scheme I below. Scheme I:
In this process, 5-amino-1 H-imidazole-4-carboxamide (A) is converted into 5- diazo-1 H-imidazole-4-carboxamide (B), which is then cyclized with methylisocyanate in dichloromethane to provide a high yield of temozolomide.
However, this process requires isolation of the unstable and potentially dangerous 5-diazo-1 H-imidazole-4-carboxamide (B). Moreover, methylisocyanate is a difficult reagent to handle and ship, especially on the industrial scale, and indeed is better avoided in industrial manufacture.
Furthermore, the cycloaddition of methylisocyanate requires a very long reaction time: Table I in J. Med Chem. 1984, 27,196-201 , suggests 20 days. Additionally, Stevens et al mention that the cycloaddition of the methylisocyanate to the compound of the formula (B) can proceed through two different intermediates:
The production of I by the two processes described in J. Chem. Soc, Chem.
Commun., 1994, 1687-1688 provides a low overall yield from 5-amino-1 H- imidazole-4-carboxamide (A): less than 20% (unoptimized – about 17% through 5- diazo-1 H-imidazole-4-carboxamide (B) and about 15% through 5-amino-N1– (ethoxycarbonylmethyl)- 1 H-imidazole- 1 ,4-dicarboxamide (C)); Scheme II below
Scheme II:
Moreover, the unstable 5-diazo-1 H-imidazole-4-carboxamide (B) still has to be isolated in the branch of this process that uses it as an intermediate. Clearly, therefore, there is a need for synthetic methods that: a) are more convenient and higher yielding, especially on commercial scale; b) approach the synthesis of the temozolomide nucleus in novel ways; or c) improve the preparation or use of intermediates for the processes.
Temozolomide of formula I, is an antitumor drag and is chemically known as 3-methyl-8- aminocarbonyl-imidazole[5,l-d]-l,2,3,5-tetrazin-4(3H)-one.
Formula I
It is indicated for treating patients with malignant glioma such as cancer, breast cancer, refractory anaplastic, astrocytoma, i.e. patient at first relapse who have experienced disease progression in malignant glioma, glioblastoma multiform and anaplastic astrocytoma, on a drug containing a nitrosourea and procarbazine. It is sold in the US market as hard capsules containing 5 mg, 20 mg, 100 mg or 250 mg as Temodar® by Schering corporation.
Temozolomide and compounds having similar activity (higher alkyl analogues at the 3 -position) were first disclosed in US patent 5,260,291. According to said patent, temozolomide is prepared by the reaction of 5-diazoimidazole-4-carboxamide with methyl isocyanate in the presence of N- methylpyrrolid-2-one in dichloromethane at room temperature for three to four weeks. Melting point of temozolomide reported in above patent is 200 0C (recrystallized from acetonitrile); 21O0C with effervescence (recrystallized from acetone and water), and 2150C with effervescence and darkening (recrystallized from hot water). Major drawback of process is the longer reaction duration of three to four weeks for completion of reaction.
Further, the process described in the patent involves use of low boiling and extremely toxic, methyl isocyanate, which is very difficult to handle, especially on industrial scale, as its use should be avoided in the industrial synthesis. Further, cycloaddition reaction requires a very long period of 21 to 28 days, which makes the process unattractive for industrial scale.
US patent 5,003,099 discloses a process for preparation of aminocyanoacetamide, a key intermediate for the synthesis of temozolomide. According to the patent, aminocyanoacetamide is synthesized in two steps by the reaction of cyanoacetic acid alkyl ester using sodium nitrite in the presence of glacial acetic acid to form a hydroxyimino intermediate, which is then reduced in the presence of platinum on carbon to yield aminocyanoacetic acid alkyl ester, which is unstable.
The alkyl ester intermediate is then in situ reacted with aqueous ammonia to give the desired product. The main drawback of the above mentioned process is the use of aqueous ammonia, since aminocyanoacetamide, generated in reaction, is soluble in aqueous solution and hence difficult to extract from the reaction mass which results in lower yields. The patent is silent about the purity of intermediate and process needs extraction of the above mentioned intermediate from filtrate.
US patent 6,844,434 describes synthesis of temozolomide by cyclization of 5-amino-l-(N-rnethyl- hydrazinocarbonyl)-lH-imidazole-4-carboxylic acid in the presence of tetrabutyl nickel and periodic acid to form a reaction mixture which is concentrated under reduce pressure and resulting residue was treated with acetonitrile and filtered. The filtrate was concentrated and chromatographed on a column of silica gel to give temozolomide.
Use of time consuming and cumbersome technique i.e. column chromatography for isolation of product makes the process not suitable to employ at industrial level. US patent 7,087,751 discloses a process for the preparation of temozolomide from protected imidazole intermediate.
The process involves reaction of l-methyl-3-carbamoyliminomethyl-urea with JV- protected aminocyanoacetamide in the presence of acetic acid in a suitable solvent to form an JV- protected imidazole intermediate which is then cyclized in the presence of lithium chloride to minimize undesired cyclisation product. After cyclisation, the protected group has to be removed which makes the process more laborious with more number of steps.
As exemplified in example 1 of the above patent, yield of the JV-protected imidazole intermediate obtained is very low, almost half of the product goes in the filtrate which further needs extraction from the filtrate. After extraction of inteπnediate from the filtrate, the combined yield is only 67 %. The intermediate obtained is only 93 to 94% pure and requires additional purifications, crystallization using ethyl acetate and slurry wash with mixture of methyl tertiary butyl ether and isopropanol. These additional purification further takes away around 20 % yield of the inteπnediate thus yield of the pure intermediate, which is suitable for the further reaction, remains around 53 % which is very low from commercial point of view.
The patent also describes condensation of l-methyl-3-carbamoyliminomethyl-urea with unprotected aminocyanoacetamide in presence of acetic acid to give an imidazole intermediate. This patent fails to disclose the process of conversion of above imidazole intermediate to temozolomide, but only up to hydrolysis to prepare 5-amino-lH-imidazole-4-carboxamide hydrochloride is reported.
Another US patent no. 6,844,434 of same applicant (Schering) discloses a process for the conversion of 5-amino- lH-imidazole-4-carboxamide hydrochloride, which is prepared by the hydrolysis of above imidazole intermediate, to temozolomide. By combining the above two processes, this adds further four additional steps to the synthesis of temozolomide. The process of preparation of temozolomide is described by the following scheme:
It has been observed that for the preparation of unprotected imidazole intermediate as exemplified in US 7,087,751, use of excess amount of the acetic acid (around 21 times with respect to aminocyanoacetamide) is reported. Thereafter acetic acid is removed by distillation.
The inventors of the present invention have repeated example 2 as described in US 7,087,751 for the preparation of unprotected imidazole intermediate. As per the process, after the completion of the reaction, acetic acid has to be removed from the reaction mixture. It is noticed that removal of acetic acid is a very tedious move so as on commercial scale and leads to decomposition.
In a publication namely, Journal of Organic Chemistry, volume 62, no. 21, 7288-7294, a process is disclosed for the preparation of temozolomide by the hydrolysis of 8-cyano-3-methyl-[3H]-imidazole~ [5,l-d]-tetrazin-4-one in the presence of hydrochloric acid to give hydrochloride salt of temozolomide, which has to be neutralized to obtain temozolomide. In the same Journal, another process for the preparation of temozolomide is also described. Temozolomide is prepared by the nitrosative cyclization of imidazole intermediate using aqueous solution of sodium nitrite and tartaric acid to give temozolomide in 45 % yield in solution.
US patent publication 2007/0225496 exemplified a process for preparation of temozolomide by pyrolising N’-methyl-N,N-diphenyl urea to form vapor of methyl isocyanate which is then reacted with 5-diazo-5H-imidazole-4-carboxylic acid amide to form temozolomide.
The above described process involves use of methyl isocyanate, which is highly flammable and makes the process unsuitable for industrial synthesis, hi addition to this, isolation of temozolomide from the reaction mixture requires addition of large amount of ethyl acetate followed by addition of hexane and again ethyl acetate to isolate compound.
US patent publication 2009/0326028 describes a process for preparation of temozolomide by diazotization of imidazole intermediate in the presence of at least one metal halide, a source of nitrous acid and an acid to form acidic solution of temozolomide, wherein temozolomide forms a salt with acid. The desired product i.e. temozolomide is then isolated from the acidic solution by extraction with a solvent.
The process requires very strict reaction parameters including the addition of metal halide during diazotization as well as addition of pre-cooled reaction mixture to sodium nitrite solution to achieve desired level of selective cyclization. Patent application also describes two methods for the extraction of temozolomide.
US patent publication 2010/0036121 discloses a process for the preparation of temozolomide by reaction of 5-aminoimidazole-4-carboxamide with N-succinimidyl-N’-methylcarbamate to form carbamoyl 5~aminoimidazole-4-carboxamide which is then reacted with alkali or alkaline earth nitrile to give reaction mass containing temozolomide
-
It is described in US 5,260,291 together with compounds of broadly similar activity such as higher alkyl analogs at the 3-position.
-
J.Med.Chem. 1984, 27, 196-201 describes a process wherein 5-amino-1H-imidazole-4-carboxamide is converted into 5-diazo-1H-imidazole-4-carboxamide, which is then cyclised with methylisocyanate in dichloromethane to provide a high yield of temozolomide.
-
This process requires isolation of the unstable and potentially dangerous 5-diazo-1H-imidazole-4-carboxamide, methyl isocyanate is a difficult reagent to handle and ship, especially on the industrial scale. Furthermore, the cycloaddition of methylisocyanate requires a long reaction time (Table I in J.Med.Chem. 1984, 27, 196-201, suggests 20 days).
-
The product obtained by this process contains, high residual dichloromethane. It is essential to limit dichloromethane content in the final API below 600 ppm as per ICH guideline. Dichloromethane content can be reduced if one follows technique of US 5,260,291 .
-
US 5,260,291 discloses acetone-water recrystallisation of temozolomide, which results in low yield (60% recovery) due to decomposition of temozolomide to impurities like 5-(3-methyltriazen-1-yl)imidazole-4-carboxamide, compound of formula V
and 5-amino-1H-imidazole-4-carboxamide.
-
The production of compound of formula I by the two processes described in J.Chem.Soc., Chem.Commun., 1994, 1687-1688 provides a low overall yield from 5-amino-1H-imidazole-4-carboxamide: less than 20% (about 17% through 5-diazo-1H-imidazole-4-carboxamide and about 15% through 5-amino-N1-(ethoxy carbonylmethyl)-1H-imidazole-1,4-dicarboxamide).
-
The unstable 5-diazo-1H-imidazole-4-carboxamide has to be isolated in the branch of this process that uses it as an intermediate.
-
US 2002/0133006 discloses a process for the preparation of compound of formula I using methyl hydrazine which is a toxic and flammable liquid, hence not feasible on industrial scale and the final isolation involves tedious workup including column chromatography.
-
J.Org.Chem. 1997, 62, 7288-7294 describes a process wherein the final step of diazotization provides equi-formation of aza-hypoxanthine and temozolomide, resulting in low yield. This literature does not provide the experimental procedure for work up.
-
US 2005/0131227 describes a process involving the use of a bulky protecting group on nitrogen of the primary amide for cyclisation in presence of LiCl to minimize the undesired cyclization product. After cyclization the protecting group has to be removed which makes the process more laborious with more number of steps (Scheme I).
U.S. Pat. No. 6,844,434 describes the preparation of Temozolomide, alkyl analogs and intermediates thereof. The process, which is depicted in Scheme 3 below, comprises reacting 5-amino-1H-imidazole-4-carboxamide hydrochloride (II) with 4-nitrophenyl chloroformate to afford compound (III), which is subsequently reacted with methyl hydrazine to obtain the corresponding compound (IV), which is cyclized to yield Temozolomide.
Another process for preparing Temozolomide is described in U.S. patent application having the Publication No. 2002/0095036 (see Scheme 4 below). In this process, the imine (V) is converted to 2-cyano-N-(1,1-dimethylethyl)-2-[(diphenyl-methylene)amino]-acetamide, which is converted to 2-amino-2-cyano-N-(1,1-dimethyl-ethyl)-acetamide hydrochloride.
The latter is reacted with compound (VI) to obtain 5-amino-N4-(1,1-dimethylethyl)-N1-methyl-1H-imidazole-1,4-dicarboxamide, which is converted to 3,4-dihydro-N-(1,1-dimethylethyl)-3-methyl-imidazo-[5,1-d]-1,2,3,5-tetrazine-8-carboxamide (tert-butyl-Temozolomide), which yields Temozolomide under acidic treatment with concentrated sulfuric acid.
Yet another synthesis of Temozolomide is described by Stevens et al. in J. Org. Chem., Vol. 62, No. 21, 7288-7294, 1997, wherein Temozolomide hydrochloride salt is obtained in 65% yield by the hydrolysis of 8-cyano-3-methyl-[3H]-imidazo-[5,1-d]-tetrazin-4-one with hydrochloric acid, as shown in Scheme 5.
The main disadvantage of this process is the low yield in which Temozolomide hydrochloride is obtained (65%). It is assumed that the relatively elevated temperature of 60° C. used in the process increases the content of decomposition products.
…………………………
Synthesis
US Patent 8,232,392
Temozolomide (1) is a drug that was discovered more than 30 years ago. In the past 10 years, it has been used to treat aggressive brain tumors. S. Turchetta and co-inventors summarize several processes for preparing temozolomide, all of which use toxic reagents such as MeNCO or MeNHNH2or generate large amounts of chemical waste. They describe a safer route to 1.
The inventors’ method starts with the preparation of carbamoyl compound 4 from amide 2 by treating it with succinimidyl reagent 3 in the presence of a base. The product is isolated in 88% yield and 96.9% purity by HPLC. Reagent 3 is a nonexplosive, crystalline solid with comparatively low toxicity and is much safer than MeNCO for this reaction.

In the next stage, the amine group in 4 is converted to diazonium salt 5 via a diazotization reaction. The details of this reaction are not described, but reference is made to a method reported in 1997 (Wang, Y., et al. J. Org. Chem. 1997, 62, 7288–7294). Compound 5 is not isolated; when acid is added, it cyclizes by the reaction of the diazonium group with one of the two amide groups to give products 1 and 6 in approximately equal amounts. The desired product 1 is formed by the reaction of the secondary amide group; when the primary amide reacts, the product is its isomer, 6.
Products 1 and 6 are separated by passing the acidified reaction mixture from the diazotization reaction over a column of a polymeric adsorbent resin. The material used in the example is XAD 1600 from Rohm & Haas; other resins are covered in the claims. Compound 6 elutes from the column first; then 1 is eluted with acidified aq EtOH. After separation, 1 is recrystallized from acidified acetone and isolated in 30% yield with 99.9% purity.
The process provides an alternative, safer route to temozolomide, but half of intermediate 4 is lost as unwanted product 6. [Chemi S.p.A. [Cinisello Balsamo, Italy]. US Patent 8,232,392, July 31, 2012; )
………………..
SYNTHESIS
http://www.google.com/patents/WO2002057268A1?cl=en

EXAMPLE 1
Preparation of Temozolomide (1 ) Step A Preparation compound (3)
5-Amino-1 H-imidazole-4-carboxamide*HCI (4) (25 g, 0.154 mol) (Aldrich 16,496-8), CH2CI2 (0.6 L) and Et3N (45 mL) (Aldrich, 13,206-3) were placed into a dry 2-liter, three-necked flask equipped with dropping funnel, a gas inlet tube, a gas outlet tube, reflux condenser and mechanical stirrer, and maintained under a positive pressure of nitrogen at ambient temperature. The mixture was stirred, and a solution of 400 mL of 4-nitrophenyl chloroformate (34 g, 0.169 mol) (Aldrich, 16,021-0) in CH2CI2was added dropwise.
The reaction mixture was stirred vigorously for 4 hours and then left to stand for 18 hours at room temperature. The precipitate was collected by vacuum filtration and washed with H20 (1.5 L) to afford the product (3) as a pale yellow solid (42 g, 0.144 mol). 1H NMR (400MHz, DMSO-d6, δ): 8.40 (d, 2H), 7.83 (s, 1 H), 7.74 (d, 2H), 7.08 (bs, 1 H), 6.95 (bs, 1 H), 6.52 (s, 2H). Step B Preparation of compound (2)
Compound (3) (42 g, 0.144 mol) and DMF (0.27 L) were placed into a dry
1 -liter, three-necked flask equipped with dropping funnel, a gas inlet tube, a gas outlet tube, reflux condenser and mechanical stirrer, and maintained under a positive pressure of nitrogen. The reaction mixture was cooled to 0°C, and methylhydrazine (10 mL, 0.188 mol) (Aldrich, M5.000-1 ) was added dropwise.
The reaction mixture was stirred vigorously for 1 hour at 0°C and was then poured into EtOAc (2.1 L). The precipitate was collected by vacuum filtration and was dried under vacuum (20 mm Hg, room temperature, 18 hours) to afford (2) as a tan solid (27.1 g, 0.137 mol). 1H NMR (400MHz, DMSO-d6, δ): 7.62 (s, 1 H), 6.85 (bs, 1 H), 6.75 (bs,1 H), 6.00 (s, 2H), 5.10 (s, 2H), 3.15, s, 3H).mp: 188°C (dec).
Analysis: Calcd for C6H10N6O2: C, 36.36; H, 5.09; N, 42.41.
Found: C, 36.46; H, 4.99; N, 42.12.
Step C Preparation of Temozolomide (1 )
2 1 (Temozolomide)
Compound (2) (500 mg, 2.5 mmol), Bu4NI (95 mg, 0.25 mmol), THF (250 mL) and CH3CN (250 mL) were placed into a dry 1 -liter, three-necked flask equipped with dropping funnel, a gas inlet tube, a gas outlet tube, reflux condenser and mechanical stirrer, and maintained under a positive pressure of nitrogen.
The reaction mixture was heated at 60°C for 20 mm and then cooled to room temperature. H5lθ6 (1.14 g, 5 mmol) was added and the reaction mixture was stirred vigorously at room temperature for 1 hour. The resulting solution was treated with saturated aqueous Na2S2O3 (5 mL) and was then concentrated under reduced pressure to dryness. The residue was treated with CH3CN (200 mL) and was filtered. The filtrate was concentrated and chromatographed on a column of silica gel (1.5% to 2% AcOH/EtOAc) to afford temozolomide (1 ) (280 mg). 1H NMR (400MHz, DMSO-d6, δ): 8.80 (s, 1 H), 7.80 (bs, 1 H), 7.66 (bs, 1 H), 3.43 (s,3H).
………………
SYNTHESIS

…………………
SYNTHESIS
http://www.google.com/patents/WO2010140168A1?cl=en
Accordingly, the present invention provides an improved process for the preparation of temozolomide of formula I,
Formula I which proves to be efficient and industrially advantageous.
The process comprises the step of: a), condensing compound of formula II,
Formula II with compound of formula III,
CH3 H CH3 Formula III in the presence of an acid in an alcoholic solvent to form a compound of formula IV;
Formula IV b). isolating the compound of formula IV from the reaction mixture by filtration; c). diazotizing and cyclizing the compound of formula IV in the presence of source of nitrous acid and a suitable acid; d). isolating temozolomide therefrom; and e). optionally purifying temozolomide of formula I.
Accordingly, the present invention provides an improved process for the preparation of temozolomide of formula I, process comprises the steps of: a), diazotizing and cyclizing the compound of formula IV in the presence of a source of nitrous acid and a suitable acid; b). optionally, cooling the reaction mixture; c). isolating precipitate of temozolomide from the reaction mixture; and d). purifying temozolomide of formula I with a suitable solvent
REFERENCE EXAMPLE:
Preparation* of S-Aøiino-N’-methyl-lH-imidazole-ljΦdicarboxamide (US 7,087,751) 2-Amino-2-cyanoacetamide (10 g), l-methyl-3-methylcarbamoyliminomethyl urea (19 g) and acetic acid (120 ml) were stirred together at ambient temperature under the positive pressure of nitrogen for 2 hours. Excess acetic acid was removed under reduced pressure and methyl tertiary butyl ether (25 ml) was added to the concentrated reaction mass, cooled to obtained crude solid.
The mixture was stirred for 30 minutes and the precipitate was collected by vacuum filtration. The solid was dried under vacuum at 20-250C for 18 hours to obtain 13 g of title compound as grayish solid. The crude product was stirred with water (66 ml) for 1 hour at 20-250C, filtered, suck dried and dried under vacuum at2O0C for 18 hours to obtain 11.2 g of title compound as greyish solid.
EXAMPLES
Example 1: Preparation of hydroxylirainocyano acetic acid ethyl ester
To a suspension of ethyl cyanoacetate (1.0 Kg, 8.84 mol) and sodium nitrite (0.735 kg, 10.65 mol) in water (0.80 L), acetic acid (0.70 kg, 11.66 mol) was added at 0-50C over a period of one hour.
Temperature was slowly raised to 23-270C and the reaction mixture was stirred for one hour at that temperature. After the complete consumption of ethyl cyanoacetate (monitored by TLC/GC), the reaction mixture was extracted with ethyl acetate (5 x 1.5 L). The combined organic layer was successively washed with 10% sodium bicarbonate (2 x 1.25 L) and brine solution (1.25 L), dried over sodium sulfate and filtered through hyflow bed. Solvent was removed under reduced pressure at 40-
450C. The resulting solid was stirred with cyclohexane (3.0 L) for 30 minutes at 25-300C, filtered and dried at 40-450C under vacuum to afford 1.14 kg (91.2 %) of title compound having purity 99.82% by
HPLC.
Example 2: Preparation of aminocyanoacetic acid ethyl ester
To a solution hydroxyliminocyano acetic acid ethyl ester (1.14 Kg, 8.02 mol) in methanol (11.4 L) was added 5% platinum on carbon (91.2 g, 50 % wet) and the mixture was hydrogenated at hydrogen gas pressure of 6.2-6.4 kg/cm2 over a period of 12 hours and the completion of reaction was checked by
TLC. The reaction mixture was filtered under nitrogen atmosphere to recover the catalyst. The filtrate was used as such for the next stage.
Example 3: Preparation of amimøcyanoacetamide
The solution of aminocyanoacetic acid ethyl ester (as prepared above) in methanol was cooled to 0-5
0C and ammonia gas was purged into it approximately for 1 hour. After the completion of the reaction
(monitored by TLC), the reaction mass was concentrated to 2.5-3.0 L under reduced pressure at 40-
45°C, cooled to 0-50C and stirred for 1 hour. The precipitated solid was filtered, washed with chilled methanol (200 ml) and dried at 35-400C under vacuum for 6 hours to obtain 572 g of title compound.
The resulting product was added to methanol (4.57 L) and heated to reflux till the solution become clear. Activated charcoal (25g) was added to the reaction mixture and refluxed for 15 minutes. The solution was filtered through hyflow bed, the bed was washed with methanol (500 ml) and the filtrate was concentrated to half of its original volume (approx 2.0 L). The mixture was cooled to 0-50C and stirred for 45 minutes. The resulting solid was filtered, washed with chilled methanol (250 ml) and dried at 40-450C under vacuum to obtain 425g (53.6%) of pure title compound having purity 99.46% by HPLC. Example 4: Preparation of l-methyl-3-methylcarbamoyliminomethyl urea
A suspension of monomethyl urea (1.5 kg, 20.27 mol) in triethyl orthoformate (4.5 L, 30.40 mol) was heated to reflux at 150-1600C for 12 hours. The reaction mixture was cooled to 5-100C, and stirred for 1 hour to ensure complete precipitation, of the product. The resulting solid was filtered, washed with ethyl acetate (350ml) and dried under vacuum at 45-5O0C to yield 1.08 kg (67.9%) of title compound having purity 93.82% by HPLC.
Exainple-5: Preparation of S-amino-N^methyl-lH-imidazole-l^-dicarboxamide Acetic acid (200 ml, 3.53 mol) was added to a suspension of aminocyanoacetamide (40Og, 4.04 mol) and l-methyl-3-methylcarbamoyliminomethyl urea (76Og, 4.8 mol) in methanol (2.0 L) at 20-250C and the mixture was stirred at 20-250C for 18 hours till completion of the reaction (monitored by HPLC). The reaction mixture was cooled to 0-50C, stirred for 1 hour and the resulting solid was filtered, washed with chilled methanol (450 ml), suck dried and finally dried under vacuum at 30-350C to afford 648 g (88.04%) of title compound as an off white colored solid having purity 99.21 % by HPLC. Example 6: Preparation of temozolomide
Acetic acid (450 ml, 7.95 mol) was added to a suspension of S-amino-N^methyl-lH-imidazole-l^- dicarboxamide (500g, 2.73mol) and sodium nitrite (25Og, 3.62mol) in water (5.0 L) at -5 to 00C at such a rate so that temperature does not rise above 5°C. The reaction mixture was stirred at 0 to 5°C for one hour and absence of starting material was checked by HPLC analysis. Ice bath was removed and powdered calcium chloride (1.25Kg) was added in small lots to the reaction mass and stirred at 25- 300C for 2 hours. The reaction mass was extracted with a 2.5% solution of dimethylsulfoxide in dichloromethane (5 X 50 L). Combined organic layer was dried over sodium sulfate and filtered through a hyflow bed. Solvent was removed under reduced pressure below 4O0C and residual dimethylsulfoxide layer was degassed completely. The dimethylsulfoxide layer was cooled to 0 to – 100C and stirred for 1 hour. The resulting solid was filtered, washed with ethyl acetate (25OmL), and suck dried for 2 hours to afford 32Og of the title compound having purity 78.5% by HPLC. Example 7: Preparation of temozolomide
Acetic acid (9ml, 0.159mol) was added to a suspension of 5-ammo-N1 -methyl- lH-imidazole- 1,4- dicarboxamide (1Og, 0.054mol) and sodium nitrite (5g, 0.072mol) in water (100ml) at -5 to 00C at a rate so that temperature does not rise above 0-50C. The reaction mixture was stirred at 0-50C for one and half hour. Brine (30g) was added to the reaction mixture and stirred at room temperature for two hours to saturate the reaction mixture. The reaction mass was extracted with a 2.5% solution of dimethylsulfoxide in dichloromethane (5 X 1 L). Combined organic layer was dried over sodium sulfate and filtered through a hyflow bed. Solvent was removed under reduced pressure and residual dimethylsulfoxide layer was degassed completely. The dimethylsulfoxide layer was cooled to 0 to -5°C and stirred for 1 hour. The resulting solid was filtered, washed with ethyl acetate (2x 5 ml), and suck dried for 2 hours to afford 5.0 g of the title compound having purity 81.6% by HPLC. Example 8: Preparation of temozolomide
Acetic acid (450ml) was added to a suspension of 5 -amino-N1 -methyl- lH-imidazole- 1,4- dicarboxamide (500g) and sodium nitrite (25Og) in water (5.0 L) at -5 to O0C at a rate so that temperature does not rise above 0-50C. The reaction mixture was stirred at 0-50C for one and half hour and the absence of starting material was checked by HPLC analysis. Ice bath was removed and powdered calcium chloride (1.25 kg) was added to the reaction mixture and stirred at room temperature for two hours. The reaction mass was extracted with a 2.5% solution of dimethylsulfoxide in dichloromethane (5 X 50 L). Combined organic layer was dried over sodium sulfate and filtered through a hyflo bed. Solvent was removed under reduced pressure at below 400C and residue at 35- 400C was filtered through a candle filter to remove suspended particles and the filtrate was then degassed completely. The residual dimethylsulfoxide layer was cooled to 0±2°C and stirred for one hours. The resulting solid was filtered and sucked dried. The solid was then washed with ethyl acetate (2x 250 ml), and suck dried for 1 hours to afford 240 g of the title compound.
………………………………….
SYNTHESIS
http://www.google.com/patents/US20020133006
Example 1
Preparation of Temozolomide (1)
5-Amino-1H-imidazole-4-carboxamide.HCl (4) (25 g, 0.154 mol) (Aldrich 16,496-8), CH2Cl2(0.6 L) and Et3N (45 mL) (Aldrich, 13,206-3) were placed into a dry 2-liter, three-necked flask equipped with dropping funnel, a gas inlet tube, a gas outlet tube, reflux condenser and mechanical stirrer, and maintained under a positive pressure of nitrogen at ambient temperature. The mixture was stirred, and a solution of 400 mL of 4-nitrophenyl chloroformate (34 g, 0.169 mol) (Aldrich, 16,021-0) in CH2Cl2 was added dropwise. The reaction mixture was stirred vigorously for 4 hours and then left to stand for 18 hours at room temperature. The precipitate was collected by vacuum filtration and washed with H2O (1.5 L) to afford the product (3) as a pale yellow solid (42 g, 0.144 mol).
1H NMR (400 MHz, DMSO-d6, δ): 8.40 (d, 2H), 7.83 (s, 1H), 7.74 (d, 2H), 7.08 (bs, 1H), 6.95 (bs, 1H), 6.52 (s, 2H).
Compound (3) (42 g, 0.144 mol) and DMF (0.27 L) were placed into a dry 1-liter, three-necked flask equipped with dropping funnel, a gas inlet tube, a gas outlet tube, reflux condenser and mechanical stirrer, and maintained under a positive pressure of nitrogen. The reaction mixture was cooled to 0° C., and methylhydrazine (10 mL, 0.188 mol) (Aldrich, M5,000-1) was added dropwise. The reaction mixture was stirred vigorously for 1 hour at 0° C. and was then poured into EtOAc (2.1 L). The precipitate was collected by vacuum filtration and was dried under vacuum (20 mm Hg, room temperature, 18 hours) to afford (2) as a tan solid (27.1 g, 0.137 mol).
1H NMR (400 MHz, DMSO-d6, δ): 7.62 (s, 1H), 6.85 (bs, 1H), 6.75 (bs,1H), 6.00 (s, 2H), 5.10 (s, 2H), 3.15, s, 3H).mp: 188° C. (dec.).
Analysis: Calcd for C6H10N6O2: C, 36.36; H, 5.09; N, 42.41.
Found: C, 36.46; H, 4.99; N, 42.12.
Compound (2) (500 mg, 2.5 mmol), Bu4NI (95 mg, 0.25 mmol), THF (250 mL) and CH3CN (250 mL) were placed into a dry 1-liter, three-necked flask equipped with dropping funnel, a gas inlet tube, a gas outlet tube, reflux condenser and mechanical stirrer, and maintained under a positive pressure of nitrogen. The reaction mixture was heated at 60° C. for 20 mm and then cooled to room temperature. H5I06 (1.14 g, 5 mmol) was added and the reaction mixture was stirred vigorously at room temperature for 1 hour. The resulting solution was treated with saturated aqueous Na2S2O3 (5 mL) and was then concentrated under reduced pressure to dryness. The residue was treated with CH3CN (200 mL) and was filtered. The filtrate was concentrated and chromatographed on a column of silica gel (1.5% to 2% AcOH/EtOAc) to afford temozolomide (1) (280 mg).
1H NMR (400 MHz, DMSO-d6, δ): 8.80 (s, 1H), 7.80 (bs, 1H), 7.66 (bs, 1H), 3.43 (s, 3H).
…………………….
EXAMPLES
Example 1:
- Preparation of 3-Methyl-8-aminocarbonyl-imidazo[5,1-d]-1,2,3,5-tetrazin-4(3H)-one (Temozolomide).
-
Glacial acetic acid (25 ml), water (250 ml) and LiCl (225 g) were charged and the contents were stirred for 30 minutes and cooled to room temperature. 5-Amino-1-(N-methylcarbamoyl) imidazole-4-carboxamide (II) (25 g) was added and stirred the contents for further 30 minutes. The reaction mixture was cooled to 0°C and then added drop wise to NaNO2 solution (12.5 g in 50 ml water) at -10 to 5 °C. The reaction mass was stirred for 1 hr at 0-5 °C and then at room temperature for 5 hrs. To this reaction mixture, sodium thiosulphate solution (25 g in 250 ml of water) was added slowly and stirred for 20 minutes (solution A). This process yielded an acidic solution containing temozolomide.
……………………..
SYNTHESIS
EXAMPLES Example 1
A 250 ml reaction vessel equipped with a magnetic stirrer and a reflux condenser was charged with 8-cyano-3-methyl-[3H]-imidazo-[5,1-d]-tetrazin-4-one (10 grams, 0.0568 mol) and hydrochloric acid (36.5-38%, 50 ml). The reaction mixture was heated to 32-35° C. and stirring was maintained at this temperature for about 3 hours. A sample was withdrawn and analyzed by HPLC to verify that the high conversion was received. (If the content of the starting material 8-cyano-3-methyl-[3H]-imidazo-[5,1-d]-tetrazin-4-one is more than 2.5% by area according to HPLC, the stirring may be continued for additional one hour).
The reaction mixture was then cooled to 20° C. and 50 ml of acetone were added drop-wise while maintaining the temperature at 20° C. Stirring was continued for 15-30 minutes. The precipitated white crystals were washed with cold acetone (20 ml) and dried at 40° C. in vacuum to obtain 11.7 grams (0.0507 mol) of Temozolomide hydrochloride (89.3% yield). Purity (by HPLC): 99.6%.
…………………………
SYNTHESIS
EXAMPLES
The following Examples illustrate but do not in any way limit the present invention. Chemicals obtained from Aldrich Chemical Company (Milwaukee, Wis.) are identified by their catalog number. It should be noted that nomenclature may differ slightly between this specification and the Aldrich catalog.
Example 1 Preparation of Temozolomide (1)
Step A Preparation Compound (3)
5-Amino-1H-imidazole-4-carboxamide.HCl (4) (25 g, 0.154 mol) (Aldrich 16,496-8), CH2Cl2(0.6 L) and Et3N (45 mL) (Aldrich, 13,206-3) were placed into a dry 2-liter, three-necked flask equipped with dropping funnel, a gas inlet tube, a gas outlet tube, reflux condenser and mechanical stirrer, and maintained under a positive pressure of nitrogen at ambient temperature. The mixture was stirred, and a solution of 400 mL of 4-nitrophenyl chloroformate (34 g, 0.169 mol) (Aldrich, 16,021-0) in CH2Cl2was added dropwise. The reaction mixture was stirred vigorously for 4 hours and then left to stand for 18 hours at room temperature. The precipitate was collected by vacuum filtration and washed with H2O (1.5 L) to afford the product (3) as a pale yellow solid (42 g, 0.144 mol).
1H NMR (400 MHz, DMSO-d6, δ): 8.40 (d, 2H), 7.83 (s, 1H), 7.74 (d, 2H), 7.08 (bs, 1H), 6.95 (bs, 1H), 6.52 (s, 2H).
Step B Preparation of Compound (2)
Compound (3) (42 g, 0.144 mol) and DMF (0.27 L) were placed into a dry 1-liter, three-necked flask equipped with dropping funnel, a gas inlet tube, a gas outlet tube, reflux condenser and mechanical stirrer, and maintained under a positive pressure of nitrogen. The reaction mixture was cooled to 0° C., and methylhydrazine (10 mL, 0.188 mol) (Aldrich, M5,000-1) was added dropwise. The reaction mixture was stirred vigorously for 1 hour at 0° C. and was then poured into EtOAc (2.1 L). The precipitate was collected by vacuum filtration and was dried under vacuum (20 mm Hg, room temperature, 18 hours) to afford (2) as a tan solid (27.1 g, 0.137 mol).
1H NMR (400 MHz, DMSO-d6, δ): 7.62 (s, 1H), 6.85 (bs, 1H), 6.75 (bs,1H), 6.00 (s, 2H), 5.10 (s, 2H), 3.15, s, 3H).mp: 188° C. (dec.). Analysis: Calcd for C6H10N6O2: C, 36.36; H, 5.09; N, 42.41. Found: C, 36.46; H, 4.99; N, 42.12.
Step C Preparation of Temozolomide (1)
Compound (2) (500 mg, 2.5 mmol), Bu4NI (95 mg, 0.25 mmol), THF (250 mL) and CH3CN (250 mL) were placed into a dry 1-liter, three-necked flask equipped with dropping funnel, a gas inlet tube, a gas outlet tube, reflux condenser and mechanical stirrer, and maintained under a positive pressure of nitrogen. The reaction mixture was heated at 60° C. for 20 mm and then cooled to room temperature. H5IO6 (1.14 g, 5 mmol) was added and the reaction mixture was stirred vigorously at room temperature for 1 hour. The resulting solution was treated with saturated aqueous Na2S2O3 (5 mL) and was then concentrated under reduced pressure to dryness. The residue was treated with CH3CN (200 mL) and was filtered. The filtrate was concentrated and chromatographed on a column of silica gel (1.5% to 2% AcOH/EtOAc) to afford temozolomide (1) (280 mg).
1H NMR (400 MHz, DMSO-d6, δ): 8.80 (s, 1H), 7.80 (bs, 1H), 7.66 (bs, 1H), 3.43 (s, 3H).

TEMOZOLOMIDE
References
- Malcolm Stevens – interview, Cancer Research UK impact & achievements page
- Newlands ES, Stevens MF, Wedge SR, Wheelhouse RT, Brock C (January 1997). “Temozolomide: a review of its discovery, chemical properties, pre-clinical development and clinical trials”. Cancer Treat. Rev. 23 (1): 35–61. doi:10.1016/S0305-7372(97)90019-0. PMID 9189180.
- Stevens MF, Hickman JA, Langdon SP, Chubb D, Vickers L, Stone R, Baig G, Goddard C, Gibson NW, Slack JA et al. (November 1987). “Antitumor activity and pharmacokinetics in mice of 8-carbamoyl-3-methyl-imidazo[5,1-d]-1,2,3,5-tetrazin-4(3H)-one (CCRG 81045; M & B 39831), a novel drug with potential as an alternative to dacarbazine”. Cancer Res. 47 (22): 5846–52.PMID 3664486.
- Jacinto, FV; Esteller, M (August 2007). “MGMT hypermethylation: a prognostic foe, a predictive friend.”. DNA Repair 6 (8): 1155–60. doi:10.1016/j.dnarep.2007.03.013. PMID 17482895.
- Hegi ME, R, Hau, Mirimanoff et al. (March 2005). “MGMT gene silencing and benefit from temozolomide in glioblastoma”. N. Engl. J. Med. 352 (10): 997–1003. doi:10.1056/NEJMoa043331.PMID 15758010. More than one of
|last1=and|author=specified (help) - National Cancer Institute Of Canada Clinical Trials, Group; Hegi, ME; Mason, WP; Van Den Bent, MJ; Taphoorn, MJ; Janzer, RC; Ludwin, SK; Allgeier, A et al. (May 2009). “Effects of radiotherapy with concomitant and adjuvant temozolomide versus radiotherapy alone on survival in glioblastoma in a randomised phase III study: 5-year analysis of the EORTC-NCIC trial”. Lancet Oncology 10 (5): 459–466. doi:10.1016/S1470-2045(09)70025-7. PMID 19269895.
- Sitbon Sitruk, L.; Sanson, M.; Prades, M.; Lefebvre, G.; Schubert, B.; Poirot, C. (2010). “Chimiothérapie à gonadotoxicité inconnue et préservation de la fertilité : Exemple du témozolomide☆”.Gynécologie Obstétrique & Fertilité 38 (11): 660–662. doi:10.1016/j.gyobfe.2010.09.002. PMID 21030284. edit
- Gilbert MR (March 2006). “New treatments for malignant gliomas: careful evaluation and cautious optimism required”. Ann. Intern. Med. 144 (5): 371–3. PMID 16520480.
- Pyrko P, Schönthal AH, Hofman FM, Chen TC, Lee AS (October 2007). “The unfolded protein response regulator GRP78/BiP as a novel target for increasing chemosensitivity in malignant gliomas”.Cancer Res. 67 (20): 9809–16. doi:10.1158/0008-5472.CAN-07-0625. PMID 17942911.
- Sheehan J, Cifarelli C, Dassoulas K, Olson C, Rainey J, Han S (2010). “Trans-sodium crocetinate enhancing survival and glioma response on magnetic resonance imaging to radiation and temozolomide”. Journal of Neurosurgery 113 (2): 234–239. doi:10.3171/2009.11.JNS091314. PMID 20001586.
- “Safety and Efficacy Study of Trans Sodium Crocetinate (TSC) With Concomitant Radiation Therapy and Temozolomide in Newly Diagnosed Glioblastoma (GBM)”. ClinicalTrials.gov. November 2011.
- Ueno T, Ko SH, Grubbs E et al. (March 2006). “Modulation of chemotherapy resistance in regional therapy: a novel therapeutic approach to advanced extremity melanoma using intra-arterial temozolomide in combination with systemic O6-benzylguanine”. Mol. Cancer Ther. 5 (3): 732–8. doi:10.1158/1535-7163.MCT-05-0098. PMID 16546988.
- Friedman, HS; Jiang, SX; Reardon, DA; Desjardins, A; Vredenburgh, JJ; Rich, JN; Gururangan, S; Friedman, AH et al. (March 2009). “Phase II trial of temozolomide plus o6-benzylguanine in adults with recurrent, temozolomide-resistant malignant glioma”. J. Clin. Oncol. 27 (8): 1262–7. doi:10.1200/JCO.2008.18.8417. PMC 2667825. PMID 19204199.
- http://labs.fhcrc.org/kiem/Hans-Peter_Kiem.html
- Dall’oglio S, D’Amico A, Pioli F, Gabbani M, Pasini F, Passarin MG, Talacchi A, Turazzi S, Maluta S (December 2008). “Dose-intensity temozolomide after concurrent chemoradiotherapy in operated high-grade gliomas”. J Neurooncol 90 (3): 315–9. doi:10.1007/s11060-008-9663-9. PMID 18688571.
- Osmani AH, Masood N; Masood (2012). “Temozolomide for relapsed primary CNS lymphoma”. J Coll Physicians Surg Pak 22 (9): 594–595. PMID 22980617.
- Chemotherapy Drug Shrinks Brain Tumors American Academy of Neurology, May 21, 2007
- Information for people undergoing treatment with temozolomide Cancer Research UK (CancerHelp UK)
Wang, et al., “Alternative Syntheses of the antitumor drug temozolomide avoiding the use of methyl isocyanates”, Journal of Chemical Society, Chemical Communication, Chemical Society, Letchworth, GB, p. 1687-1688 (1994).
Wang, et al., “Antitumor imidazotetrazines. Part 33. new syntheses of the antitumor drug temozolomide using ‘masked’ methyl isocyanates”, J. Chem. Soc., Perkin Trans. 1(21):2783-2787 (1995).
Wang, et al., “Synthetic studies of 8-carbamoylimidzo-‘5, 1-D!-1, 2, 3, 5-tetrazi n-4(3H)- one: a key derivative of antitumor drug temozolomide”, Bioorg. Med Chem. Lett., 6(2):185-188 (1996).
Yongfeng Wang, “A new route to the antitumor drug temozolomide, but not thiotemozolomide”, Chem. Commun., 4:363-364 (1997).
Wang, et al., “Antitumor Imidazotetrazines. 35. New Synthetic Routes to the Antitumor Drug Temozolomide”, J. org. Chem. 62(21):7228-7294 (1997).
Newlands, E.S., et al., “Temozolomide: a review of its discovery, chemical properties, pre-clinica development and clinical trials”, Cancer Treat. Rev. , 23(1):35-61 (1997).
Wang, et al., Antitumor Imidazotetrazines. Part 36. Conversion of 5-Amino-Imidazole-4-Carboxamide to . . . Journal of the Chemical Society, Perkin Transactions 1, Chemical Society, Letchworth, GB, 10:1669-1675 (1998).
1 Catapano CV, et al. Cancer Res. 1987, 47(18), 4884-4889.
[2] Sun S, et al. J Neurooncol. 2012.
[3] Bauer M, et al. PLoS One. 2012, 7(6):e39956.
[4] Wong ST, et al. Anticancer Res. 2012, 32(7), 2835-2841.
[5] Lin CJ, et al. PLoS One. 2012, 7(6), e38706.
[6] Gori JL, et al. Cancer Gene Ther. 2012.
| US5260291 | Oct 18, 1991 | Nov 9, 1993 | Cancer Research Campaign Technology Limited | Tetrazine derivatives |
| US20020133006 | Jan 16, 2002 | Sep 19, 2002 | Schering Corporation | Synthesis of temozolomide and analogs |
| US20050131227 | Jan 21, 2005 | Jun 16, 2005 | Schering Corporation | Synthesis of temozolomide and analogs |
| US20060183898 * | Feb 16, 2006 | Aug 17, 2006 | Olga Etlin | Process for preparing temozolomide |
| CN1487941A * | Jan 16, 2002 | Apr 7, 2004 | 先灵公司 | Synthesis of temozolomide and analogs |
| CN1706843A * | Apr 8, 2005 | Dec 14, 2005 | 江苏天士力帝益药业有限公司 | Temozolomide refining process |
| US20060183898 * | Feb 16, 2006 | Aug 17, 2006 | Olga Etlin | Process for preparing temozolomide |
| US20070225496 * | Mar 23, 2007 | Sep 27, 2007 | Palle Raghavendracharyulu Venk | rocess for preparing temozolomide |
| US8258294 * | Sep 28, 2007 | Sep 4, 2012 | Cipla Limited | Process for the preparation of temozolomide and analogs |
| EP2151442A2 | Jul 22, 2009 | Feb 10, 2010 | Chemi SPA | Process for preparing temozolomide |
| EP2374807A2 * | Sep 28, 2007 | Oct 12, 2011 | Cipla Limited | An improved process for the isolation of temozolomide |
| WO2008038031A1 | Sep 28, 2007 | Apr 3, 2008 | Cipla Ltd | An improved process for the preparation of temozolomide and analogs |
| WO2010140168A1 * | Jun 2, 2010 | Dec 9, 2010 | Ind-Swift Laboratories Limited | Improved process for preparing temozolomide |
| WO2011036676A2 | Sep 14, 2010 | Mar 31, 2011 | Ashwini Nangia | Stable cocrystals of temozolomide |
A REVIEW AND METHODS TO HANDLE PHOSGENE, TRIPHOSGENE SAFELY DURING DRUG SYNTHESIS


Phosgene
Phosgene is the chemical compound with the formula COCl2. This colorless gas gained infamy as a chemical weapon during World War I. It is also a valued industrial reagent and building block in synthesis of pharmaceuticals and other organic compounds. In low concentrations, its odor resembles freshly cut hay or grass.[3] In addition to its industrial production, small amounts occur naturally from the breakdown and the combustion oforganochlorine compounds, such as those used in refrigeration systems.[4] The chemical was named by combining the Greek words ‘phos’ (meaning light) and genesis (birth); it does not mean it contains any phosphorus (cf. phosphine).
 TRIPHOSGENE
TRIPHOSGENE
Triphosgene (bis(trichloromethyl) carbonate (BTC), C3Cl6O3) is a chemical compound that is used as a safer substitute for phosgene, because at room temperature it is a solid crystal, as opposed to phosgene which is a gas.Triphosgene crystals decompose above 200 °C READ …….http://www.buss-ct.com/e/company/publications/reaction_technology/eckert_reprint_CO6_2011-hr2.pdf
This compound is commercially available. It is prepared by exhaustive free radical chlorination of dimethyl carbonate:
- CH3OCO2CH3 + 3 Cl2 → CCl3OCO2CCl3 + 6 HCl
Triphosgene can be easily recrystallized from boiling hexanes to yield pure white crystals.
Triphosgene is used as a reagent in organic synthesis for a variety of chemical transformations including to bond one carbonyl group to two alcohols, and to convert an amine group into isocyanate.
The toxicity of triphosgene is the same as phosgene since it decomposes to phosgene on heating and upon reaction with nucleophiles. Even trace moisture leads to formation of phosgene. Therefore this reagent can be safely handled if one takes all the precautions as for phosgene.
Structure and basic properties
Phosgene is a planar molecule as predicted by VSEPR theory. The C=O distance is 1.18 Å, the C—Cl distance is 1.74 Å and the Cl—C—Cl angle is 111.8°.[5] It is one of the simplest acid chlorides, being formally derived from carbonic acid.
Industrially, phosgene is produced by passing purified carbon monoxide and chlorine gas through a bed of porous activated carbon, which serves as acatalyst:[4]
- CO + Cl2 → COCl2 (ΔHrxn = −107.6kJ/mol)
The reaction is exothermic, therefore the reactor must be cooled. Typically, the reaction is conducted between 50 and 150 °C. Above 200 °C, phosgene reverts to carbon monoxide and chlorine, Keq (300K) = 0.05. World production of this compound was estimated to be 2.74 million tonnes in 1989.[4]
Because of safety issues, phosgene is often produced and consumed within the same plant, and extraordinary measures are made to contain this toxic gas. It is listed on schedule 3 of the Chemical Weapons Convention: All production sites manufacturing more than 30 tonnes per year must be declared to the OPCW.[6] Although less dangerous than many other chemical weapons, such as sarin, phosgene is still regarded as a viablechemical warfare agent because it is so easy to manufacture when compared to the production requirements of more technically advanced chemical weapons such as the first-generation nerve agent tabun.[7]
Upon ultraviolet (UV) radiation in the presence of oxygen, chloroform slowly converts into phosgene by a radical reaction. To suppress thisphotodegradation, chloroform is often stored in brown-tinted glass containers. Chlorinated compounds used to remove oil from metals, such as automotive brake cleaners, are converted to phosgene by the UV rays of arc welding processes.[8]
Phosgene may also be produced during testing for leaks of older-style refrigerant gases. Chloromethanes (R12, R22 and others) were formerly leak-tested in situ by employing a small gas torch (propane, butane or propylene gas) with a sniffer tube and a copper reaction plate in the flame nozzle of the torch. If any refrigerant gas was leaking from a pipe or joint, the gas would be sucked into the flame via the sniffer tube and would cause a colour change of the gas flame to a bright greenish blue. In the process, phosgene gas would be created due to the thermal reaction. No valid statistics are available, but anecdotal reports suggest that numerous refrigeration technicians suffered the effects of phosgene poisoning due to their ignorance of the toxicity of phosgene, produced during such leak testing.[citation needed] Electronic sensing of refrigerant gases phased out the use of flame testing for leaks in the 1980s. Similarly, phosgene poisoning is a consideration for people fighting fires that are occurring in the vicinity of freon refrigeration equipment, smoking in the vicinity of a freon leak, or fighting fires using halon or halotron.
The great majority of phosgene is used in the production of isocyanates, the most important being toluene diisocyanate (TDI) and methylene diphenyl diisocyanate (MDI). These two isocyanates are precursors to polyurethanes.
Synthesis of carbonates
Significant amounts are also used in the production of polycarbonates by its reaction with bisphenol A.[4] Polycarbonates are an important class of engineering thermoplastic found, for example, in lenses in eye glasses. Diols react with phosgene to give either linear or cyclic carbonates (R = H, alkyl, aryl):
- HOCR2-X-CR2OH + COCl2 → 1/n [OCR2-X-CR2OC(O)-]n + 2 HCl
Synthesis of isocyanates
The synthesis of isocyanates from amines illustrates the electrophilic character of this reagent and its use in introducing the equivalent of “CO2+“:[9]
Such reactions are conducted in the presence of a base such as pyridine that absorbs the hydrogen chloride.
Laboratory uses
In the research laboratory phosgene still finds limited use in organic synthesis. A variety of substitutes have been developed, notably trichloromethyl chloroformate (“diphosgene“), a liquid at room temperature, and bis(trichloromethyl) carbonate (“triphosgene“), a crystalline substance.[10] Aside from the above reactions that are widely practiced industrially, phosgene is also used to produceacid chlorides and carbon dioxide from carboxylic acids:
- RCO2H + COCl2 → RC(O)Cl + HCl + CO2
Such acid chlorides react with amines and alcohols to give, respectively, amides and esters, which are commonly used intermediates. Thionyl chloride is more commonly and more safely employed for this application. A specific application for phosgene is the production of chloroformic esters:
- ROH + COCl2 → ROC(O)Cl + HCl
Although it is somewhat hydrophobic, phosgene reacts with water to release hydrogen chloride and carbon dioxide:
- COCl2 + H2O → CO2 + 2 HCl
Analogously, with ammonia, one obtains urea:
- COCl2 + 4 NH3 → CO(NH2)2 + 2 NH4Cl
Halide exchange with nitrogen trifluoride and aluminium tribromide gives COF2 and COBr2, respectively.[4]
History
Phosgene was synthesized by the British chemist John Davy (1790–1868) in 1812 by exposing a mixture of carbon monoxide and chlorine to sunlight. He named it “phosgene” in reference of the use of light to promote the reaction; from Greek, phos (light) and gene (born).[11] It gradually became important in the chemical industry as the 19th century progressed, particularly in dye manufacturing.
Further information: Use of poison gas in World War I and Second Italo-Abyssinian War
Following the extensive use of phosgene gas in combat during World War I, it was stockpiled by various countries as part of their secret chemical weapons programs.[12][13][14]
In May 1928, eleven tons of phosgene escaped from a war surplus store in central Hamburg.[15] 300 people were poisoned of whom 10 died.[15]
.
US Army phosgene identification poster from World War II
Phosgene was then only frequently used by the Imperial Japanese Army against the Chinese during the Second Sino-Japanese War.[16] Gas weapons, such as phosgene, were produced by Unit 731 and authorized by specific orders given by Hirohito (Emperor Showa) himself, transmitted by the chief of staff of the army. For example, the Emperor authorized the use of toxic gas on 375 separate occasions during the battle of Wuhan from August to October 1938.[17]
Phosgene is an insidious poison as the odor may not be noticed and symptoms may be slow to appear.[18] The odor detection threshold for phosgene is 0.4 ppm, four times the threshold limit value. Its high toxicity arises from the action of the phosgene on the proteins in the pulmonary alveoli, the site of gas exchange: their damage disrupts the blood-air barrier, causing suffocation. It reacts with the amines of the proteins, causing crosslinking by formation of urea-like linkages, in accord with the reactions discussed above. Phosgene detection badges are worn by those at risk of exposure.[4]
Sodium bicarbonate may be used to neutralise liquid spills of phosgene. Gaseous spills may be mitigated with ammonia.[19]
.
TRIPHOSGENE HANDLING
.

Left, reaction vessel with amino acid and triphosgene dissolved in THF; middle, appearance of the reaction mixture after addition of 2,4,6-collidine; and right, appearance of the reaction mixture after microwave irradiation.

Typical glassware standard equipment for the safety phosgenation with phosgene supply from triphosgene: (A) phosgene generator (V = 1 L, T = 85 °C) loaded with 600 g of triphosgene; (B) refluxer (water cooled, T = 15 °C); (C) phosgene line (Viton hose); (D) phosgenation reactor (V = 10 L, T = 110 °C); (E) refluxer (cryostat cooled, T = −30 °C); (F) off-gas line (Viton hose) from the top of the refluxer (E); (G) cooling trap (dry ice cooled, T = −60 °C); (H) off-gas line; (I) cryostat. The assembly of the equipment is somewhat reduced to effect more clarity of the ensemble.
.

.
Phosgene is quantitatively formed from solid triphosgene in a solvent-free and safe process without any reaction heat, catalyzed by planar N-heterocycles with deactivated imino functions.
The rate of phosgene generation is adjustable to the rate of phosgene consumption in the subsequent phosgenation reaction by thermal control, catalyst concentration, and in some cases, specific properties of selected metal phthalocyanines. A thermal runaway reaction of this process is impossible.
.
Use a safer process for generating phosgene.

Decomposition of triphosgene (1a) into carbon tetrachloride, carbon dioxide, and 1 equiv of phosgene (3)
Phosgene (COCl2) is useful in organic synthesis for chlorination, chlorocarbonylation, carbonylation, and dehydration; but its high toxicity discourages its use. Until now, the best substitute for COCl2 has been triphosgene [(CCl3O)2CO], a stable solid that has low vapor pressure. Although (CCl3O)2CO can be used in phosgenation reactions, removing the unreacted reagent from reaction mixtures is difficult because of its high boiling point. In contrast, COCl2 is easily removed by evaporating it.
(CCl3O)2CO reacts with silica gel, metal salts, or Lewis acids to generate 1 equiv of phosgene by an electrocyclic reaction. H. Eckert* and J. Auerweck at the University of Technology, Munich (Germany) report that pyridine and phthalocyanine derivatives catalyze the decomposition of (CCl3O)2CO to generate 3 equiv of COCl2.
The catalysts, phenanthridine , poly(2-vinylpyridine) , and phthalocyanines , convert liquid (CCl3O)2CO to the desired COCl2. The size and structure of the catalysts allow (CCl3O)2CO to react by the mechanism shown. The reaction was run at the 100-g scale to generate 22 L of gaseous COCl2 with an oil bath or an IR heater as the heat source. Because the catalysts are not soluble in (CCl3O)2CO, the process is considered to be heterogeneous catalysis.
.

Controlled transformation of triphosgene (1) into 3 equiv of phosgene (3) catalyzed by 4
Compounds 1 and 4a−4 h are commercially available products from Sigma-Aldrich, with the following purities: 1, 98% (IR νC═O 1820 cm−1, 13C NMR δ 108.0, 140.9); 4a, 98%; 4c, n.a.; 4d, 99%;4e, 97%; 4f, 97%; 4g, 90%; 4h, 85%.
Because the reaction is controlled by temperature, turning off the heat source causes the liquid (CCl3O)2CO to crystallize and stops the reaction, making the process safe. The reaction can be used to generate COCl2 externally or to produce it in situ. According to the authors, this method fulfills the goal of “safety phosgenation on demand of consumer”.
ORGANIC PROCESS RESEARCH AND DEVELOPMENT
A FRET approach towards potential detection of phosgene is presented, which is based on a selective chemical reaction between phosgene (or triphosgene as a simulant) and donor and acceptor fluorophores.
FRET has been applied in an experimental method for the detection of phosgene. In it, phosgene or rather triphosgene as a safe substitute serves as a linker between an acceptor and a donor coumarine (forming urea groups).[3] The presence of phosgene is detected at 5×10-5M with a typical FRET emission at 464 nm.


EXAMPLES OF USE OF TRIPHOSGENE
Chlorination of Aliphatic Primary Alcohols via Triphosgene-Triethylamine Activation
Caitlan E. Ayala, Andres Villalpando, Alex L. Nguyen, Gregory T. McCandless and Rendy Kartika*
*Department of Chemistry, 232 Choppin Hall, Louisiana State University, Baton Rouge, Louisiana 70803, United States, Email: rkartika lsu.edu
lsu.edu
C. E. Ayala, A. Villalpando, A. L. Nguyen, G. T. McCandless, R. Kartika, Org. Lett., 2012, 14, 3676-3679.
DOI: 10.1021/ol301520d (free Supporting Information)

Activation of primary aliphatic alcohols with triphosgene and triethylamine mixtures afforded either alkyl chloride or diethylcarbamate products, and the switch in selectivity appeared to be driven by sterics. The reaction conditions to achieve this highly useful transformation were unexceptionally mild and readily tolerated by a wide range of sensitive functionalities.

…………………………
.

.
ABACAVIR SULPHATE

VESTIPITANT
The following synthetic route was reported by Giuseppe Guercio et al from GlaxoSmithKline:
The initial chemical development synthetic route, derived from the one used by medicinal chemistry, involved several hazardous reagents, gave low yields and produced high levels of waste. Through a targeted process of research and development, application of novel techniques and extensive route scouting, a new synthetic route for GW597599 was developed. This paper reports the optimisation work of the third and last stage in the chemical synthesis of GW597599 and the development of a pilot-plant-suitable process for the manufacturing of optically pure arylpiperazine derivative 1. In particular, the process eliminated the use of triphosgene in the synthesis of an intermediate carbamoyl chloride, substantially enhancing safety, overall yield, and throughput.

source:
Org. Process Res. Dev., 2009, 13 (6), pp 1100–1110.
Org. Process Res. Dev., 2009, 13 (3), pp 489–493.
Org. Process Res. Dev., 2008, 12 (6), pp 1188–1194.
.
TIVOZANIB

VIAGRA

EFAVIRENZ ………EP2454244A1


Enantiomerically pure hydantoins are prepared from optically pure α-amino amides utilizing triphosgene. A mechanism for the racemization observed with 1,1′-carbonyldiimidazole (CDI) for this type of reaction is proposed.
D. Zhang, X. Xing, G. D. Cuny, J. Org. Chem., 2006, 71, 1750-1753.
Double acylation of a titanaselenide by triphosgene;
4,5-ethylenedithio-1,3-diselenol-2-one

References
- JMerck Index, 11th Edition, 7310.
- http://www.inchem.org/documents/icsc/icsc/eics0007.htm
- CBRNE – Lung-Damaging Agents, Phosgene May 27, 2009
- Wolfgang Schneider; Werner Diller (2005), “Phosgene”, Ullmann’s Encyclopedia of Industrial Chemistry, Weinheim: Wiley-VCH, doi:10.1002/14356007.a19_411
- Nakata, M.; Kohata, K.; Fukuyama, T.; Kuchitsu, K. (1980). “Molecular Structure of Phosgene as Studied by Gas Electron Diffraction and Microwave Spectroscopy. The rz Structure and Isotope Effect”.Journal of Molecular Spectroscopy 83: 105–117. doi:10.1016/0022-2852(80)90314-8.
- Annex on Implementation and Verification (“Verification Annex”)
- https://itportal.decc.gov.uk/cwc_files/S2AAD_guidance.pdf
- “Common Cleaners Can Turn Into Poison Gas”. American Iron Magazine. TAM Communications. Retrieved 14 October 2011.
- R. L. Shriner, W. H. Horne, and R. F. B. Cox (1943), “p-Nitrophenyl Isocyanate”, Org. Synth.; Coll. Vol. 2: 453
- Hamley, P. “Phosgene” Encyclopedia of Reagents for Organic Synthesis, 2001 John Wiley, New York. doi: 10.1002/047084289X.rp149
- John Davy (1812). “On a Gaseous Compound of Carbonic Oxide and Chlorine”. Philosophical Transactions of the Royal Society of London 102: 144–151. doi:10.1098/rstl.1812.0008.JSTOR 107310.
- Base’s phantom war reveals its secrets, Lithgow Mercury, 7/08/2008
- Chemical warfare left its legacy, Lithgow Mercury, 9/09/2008
- Chemical bombs sit metres from Lithgow families for 60 years, The Daily Telegraph, September 22, 2008
- Ryan, T.Anthony (1996). Phosgene and Related Carbonyl Halides. Elsevier. pp. 154–155. ISBN 0444824456.
- Yuki Tanaka, “Poison Gas, the Story Japan Would Like to Forget”, Bulletin of the Atomic Scientists, October 1988, p. 16–17
- Y. Yoshimi and S. Matsuno, Dokugasusen Kankei Shiryô II, Kaisetsu, Jugonen Sensô Gokuhi Shiryoshu, 1997, p. 27–29
- Borak J., Diller W. F. (2001). “Phosgene exposure: mechanisms of injury and treatment strategies”. Journal of Occupational and Environmental Medicine 43 (2): 110–9. doi:10.1097/00043764-200102000-00008. PMID 11227628.
- “Phosgene: Health and Safety Guide”. International Programme on Chemical Safety. 1998.
-
(a) Cotarca, L. and Eckert, H. Phosgenations − A Handbook; Wiley-VCH: Weinheim, 2003.
(b) Cotarca, L. and Eckert, H. Phosgenations − A Handbook; Wiley-VCH:Weinheim, 2003; pp 20− 21.
(c) Cotarca, L. and Eckert, H. Phosgenations − A Handbook;Wiley-VCH: Weinheim, 2003; pp 44− 520.
(d) Cotarca, L. and Eckert, H. Phosgenations − A Handbook; Wiley-VCH: Weinheim, 2003; p 41.(e) Cotarca, L. and Eckert, H.Phosgenations − A Handbook; Wiley-VCH: Weinheim, 2003; pp 14− 16, 613− 615.
-
Recent online information: www.ch.tum.de/oc1/HEckert/research.htm.
-
(a) Senet, J. P. The Recent Advance in Phosgene Chemistry; SNPE: Paris, 1997; Vol. 1.
(b) Pasquato, L.; Modena, G.; Cotarca, L.; Delogu, P.; Mantovani, S. J. Org. Chem. 2000, 65,8224– 8228
(d) Dunlap, K. L. In Kirk-Othmer Encyclopedia of Chemical Technology, 5 ed.;Wiley: New York, 2006; Vol. 18, pp 802− 814.(e) Nielsen, D. H.; Burke, T. G.; Woltz, P. J. H.; Jones, E. A. J. Chem. Phys. 1952, 20, 596– 604
(f) Gordon, E. P.;Enakaeva, V. G.; Korotchenko, A. V.; Mitrokhin, A. M. Russian Patent RU 2299852, 2007.
-
(a) Eckert, H.; Forster, B. Angew. Chem. 1987, 99, 922– 923 ; Angew. Chem., Int. Ed.,1987, 26, 894–895
(b) Eckert, H. TUM-Mitteilungen (Technische Universitaet Muenchen) 2006, 3, 68– 69(d) Triphosgene; Ubichem: U.K., 1999; CD-ROM.
-
(a) Eckert, H.; Drefs, N. Chemanager 2006, 3) 10
-
Eckert, H.; Dirsch, N.; Gruber, B. (former Dr. Eckert GmbH, now Buss Chem Tech AG) German Offen. DE 19740577, 1999 (Sep. 15, 1997), Chem. Abstr. 1999, 130, 211406.;
WO 9914159, 1999; Eur. Pat. EP 1017623, 2002; U.S. Patent US 6399822, 2002; Japanese Patent JP 2001516692, 2001.
-
Mole percent 4 referring to 3 phosgene equivalents of 1 .
-
(a) Leznoff, C. C.; Lever, A. B. P. Phthalocyanines, Properties and Applications; VCH:Weinheim, NY, 1989.
(b) Lever, A. B. P. Adv. Inorg. Chem. Radiochem. 1965, 7, 28– 114
(c) Ebert, N. A.; Gottlich, H. B. J. Am. Chem. Soc. 1952, 74, 2806
[ACS Full Text ], [CAS]
], [CAS] -
The weighing error of this procedure mainly comes from icy condensed humidity at the cool glassware of the cooling trap and is less than 0.5 g, determined by a series of weighings under the same conditions, the same equipment, temperature (T = −78 °C), and handling time <10 s, but without 3. Under these conditions evaporation of 3 (bp 8 °C) hardly ever happens and can be ignored.
-
Monitox plus gas monitor (COCl2) and phosgene badges from Compurhttp://www.compur.com/gasmessgeraete/front_content.php?idcat=7&changelang=3.
- Davy’s account of his discovery of phosgene
- International Chemical Safety Card 0007
- CDC – Phosgene – NIOSH Workplace Safety and Health Topic
- NIOSH Pocket Guide to Chemical Hazards
- U.S. CDC Emergency Preparedness & Response
- U.S. EPA Acute Exposure Guideline Levels
- Regime For Schedule 3 Chemicals And Facilities Related To Such Chemicals, OPCW website
- CBWInfo website
- Use of Phosgene in WWII and in modern-day warfare (Refer to Section 4.C of the article)
- An experience with accidental poisoning by heated tetrachlorethylene solvent
‘Female Viagra’ Flibanserin now on track for Q3 filing in USA

Flibanserin, girosa
167933-07-5 cas no
147359-76-0 (monoHCl)
- Bimt 17
- BIMT 17 BS
- Bimt-17
- Flibanserin
- Girosa
- UNII-37JK4STR6Z
Women with low libido in the US will have to wait even longer for approval of the first ever treatment for the condition after regulators requested more data on the forerunner flibanserin, delaying its submission until later this year.
The US Food and Drug Administration has asked manufacturer Sprout Pharmaceuticals for data on how flibanserin interacts with other medicines and also how it affects driving ability, after around 10% of patients experienced sleepiness while on the drug
Read more at: http://www.pharmatimes.com/Article/14-02-11/Female_Viagra_now_on_track_for_Q3_filing_in_USA.aspx#ixzz2tAWxwzRD
December 11, 2013 – Sprout Pharmaceuticals today announced that it has received and appealed the Food and Drug Administration’s (FDA) Complete Response Letter (CRL) for flibanserin through the Formal Dispute Resolution process.
Flibanserin is an investigational, once-daily treatment for Hypoactive Sexual Desire Disorder, or HSDD, in premenopausal women. HSDD is the most commonly reported form of female sexual dysfunction
read all here
A new drug being developed by Boehringer Ingelheim could give a boost to the sex drive of women with low libido. The drug, known as flibanserin, has been shown in clinical trials to increase their sexual desire when taken once a day at bedtime.
The results from four pivotal Phase III clinical trials on women with hypoactive sexual desire disorder (HSDD) were presented this week at the European Society for Sexual Medicine’s congress in Lyon, France. The trials showed that participants taking flibanserin had a significant improvement in their sexual desire compared to those given a placebo. They also experienced less of the distress associated with sexual dysfunction.
The drug was initially being investigated as a treatment for depression, and acts on the serotonin receptors in the brain – it is both a 5-HT1A receptor agonist and a 5-HT2A receptor antagonist. It is also a partial agonist at the dopamine D4 receptor.

Neurotransmitters such as serotonin are believed to be involved in sexual function, and antidepressants are commonly associated with a loss of libido, so this was an obvious side-effect to look out for during clinical trials in depression. But far from suppressing the libido in women, it appeared to have the opposite effect, so trials in women with HSDD were initiated.
Hormone replacement can improve the libido of women who have had their ovaries removed, but there is no available drug to treat those who have not. There have been accusations that pharma companies invent new diseases like HSDD in order to sell more medicines, but according to Kathleen Segraves, an assistant professor at Case Western Reserve University in the US who has worked in the field of sexual functioning for many years, this is not the case here. HSDD is a very real disorder, she says, and the potential for a treatment for these women is very exciting.

Flibanserin (code name BIMT-17; proposed trade name Girosa) is a drug that was investigated by Boehringer Ingelheim as a novel, non-hormonal treatment for pre-menopausal women with Hypoactive Sexual Desire Disorder (HSDD).[1][2] Development was terminated in October 2010 following a negative report by the U.S. Food and Drug Administration.[3]
HSDD is the most commonly reported female sexual complaint and characterized by a decrease in sexual desire that causes marked personal distress and/or personal difficulties. According to prevalence studies about 1 in 10 women reported low sexual desire with associated distress, which may be HSDD.[4] The neurobiological pathway of female sexual desire involves interactions among multiple neurotransmitters, sex hormones and various psychosocial factors. Sexual desire is modulated in distinct brain areas by a balance between inhibitory and excitatory neurotransmitters, serotonin acting as an inhibitor while dopamine and norepinephrine act as a stimulator of sexual desire.[5][6]Flibanserin is a 5-HT1A receptor agonist and 5-HT2A receptor antagonist that had initially been investigated as an antidepressant. Preclinical evidence suggested that flibanserin targets these receptors preferentially in selective brain areas and helps to restore a balance between these inhibitory and excitatory effects.[6] HSDD has been recognized as a distinct sexual function disorder for more than 30 years.
The proposed mechanism of action refers back to the Kinsey dual control model. Several sex steroids, neurotransmitters, and hormones have important excitatory or inhibitory effects on the sexual response. Among the neurotransmitters, the excitatory activity is driven by dopamine and norepinephrine, while the inhibitory activity is driven by serotonin. The balance between these systems is relevant for a healthy sexual response. By modulating these neurotransmitters in selective brain areas, flibanserin, a 5-HT1A receptoragonist and 5-HT2A receptor antagonist, is likely to restore the balance between these neurotransmitter systems.[6]
Several large pivotal Phase III studies with Flibanserin were conducted in the USA, Canada and Europe. They involved more than 5,000 pre-menopausal women with generalized acquired Hypoactive Sexual Desire Disorder (HSDD). The results of the Phase III North American Trials demonstrated that
Although the two North American trials that used the flibanserin 100 mg qhs dose showed a statistically significant difference between flibanserin and placebo for the endpoint of [satisfying sexual events], they both failed to demonstrate a statistically significant improvement on the co-primary endpoint of sexual desire. Therefore, neither study met the agreed-upon criteria for success in establishing the efficacy of flibanserin for the treatment of [Hypoactive Sexual Desire Disorder].
These data were first presented on November 16, 2009 at the congress of the European Society for Sexual Medicine in Lyon, France. The women receiving Flibanserin reported that the average number of times they had “satisfying sexual events” rose from 2.8 to 4.5 times a month. However, women receiving placebo reported also an increase of “satisfying sexual events” from 2.7 to 3.7 times a month.
Evaluation of the overall improvement of their condition and whether the benefit was meaningful to the women, showed a significantly higher rate of a meaningful benefit in the flibanserin-treated patient group versus the placebo group.The onset of the Flibanserin effect was seen from the first timepoint measured after 4 weeks of treatment and maintained throughout the treatment period.
The overall incidence of adverse events among women taking flibanserin was low, the majority of adverse events being mild to moderate and resolved during the treatment. The most commonly reported adverse events included dizziness, nausea, fatigue, somnolence and insomnia.
On June 18, 2010, a federal advisory panel to the U.S. Food and Drug Administration (FDA) unanimously voted against recommending approval of Flibanserin.
Earlier in the week, a FDA staff report also recommended non-approval of the drug. While the FDA still might approve Flibanserin, in the past, negative panel votes tended to cause the FDA not to approve.
On October 8, 2010, Boehringer Ingelheim announced that it would discontinue its development of flibanserin in light of the FDA advisory panel’s recommendation.
On June 27, 2013, Sprout Pharmaceuticals confirmed they had resubmitted flibanserin for FDA approval.
Flibanserin, chemically 1 -[2-(4-(3-trifluoromethylphenyl)piperazin-1 – yl)ethyl]-2,3-dihydro-1 H-benzimidazole-2-one was disclosed in form of its hydrochloride in European Patent No. 526,434 (‘434) and has the following chemical structure:
Process for preparation of flibanserin were disclosed in European Patent No. ‘434, U.S. Application Publication No. 2007/0032655 and Drugs of the future 1998, 23(1): 9-16.
According to European Patent No. ‘434 flibanserin is prepared by condensing 1-(2-chloroethyl)-2,3-dihydro-1 H-benzimidazol-one with m- trifluoromethyl phenyl piperazine. According to U.S. Application Publication No. 2007/0032655 flibanserin is prepared by condensing 1-[(3-trifluoromethyl)phenyl]-4-(2- chloroethyl)piperazine with 1 -(2-propenyl)-1 ,3-dihydro-benzimidazol-2H-one.
According to Drugs of the future 1998, 23(1): 9-16 flibanserin is prepared by reacting 1-(2-chloroethyl)-2,3-dihydro-1 H-benzimidazol-one with m- trifluoromethylphenylpiperazine.
PATENT
1-[2-(4-(3-trifluoromethyl-phenyl)piperazin-1-yl)ethyl]-2,3-dihydro-1H-benzimidazol-2-one
Compound 3
Hydrochloride salt (isopropanol) M.p. 230-231°C
Analysis
¹H NMR (DMSO-d₆/CDCL₃ 5:2) 11.09 (b, 1H), 11.04 (s, 1H), 7.5-6.9 (8H), 4.36 (t, 2H), 4.1-3.1 (10H)
CLIP
The compound 1-[2-(4-(3-trifluoromethyl-phenyl)piperazin-1-yl)ethyl]-2,3-dihydro-1 H- benzimidazol-2-one (flibanserin) is disclosed in form of its hydrochlorid in European Patent Application EP-A-526434 and has the following chemical structure:
Flibanserin shows affinity for the 5-HTιA and 5-HT2-receptor. It is therefore a promising therapeutic agent for the treatment of a variety of diseases, for instance depression, schizophrenia, Parkinson, anxiety, sleep disturbances, sexual and mental disorders and age associated memory impairment.

EXAMPLE……… EP1518858A1
375 kg of 1-[(3-trifluoromethyl)phenyl]-4-(2-cloroethyl)piperazin are charged in a reactor with 2500 kg of water and 200 kg of aqueous Sodium Hydroxide 45%. Under stirring 169.2 kg of 1-(2-propenyl)-1,3-dihydro-benzimidazol-2H-one, 780 kg of isopropanol, 2000 kg of water and 220 kg of aqueous Sodium Hydroxide 45% are added. The reaction mixture is heated to 75-85° C. and 160 kg of concentrated hydrochloric acid and 200 kg of water are added.
The reaction mixture is stirred at constant temperature for about 45 minutes. After distillation of a mixture of water and Isopropanol (about 3000 kg) the remaining residue is cooled to about 65-75° C. and the pH is adjusted to 6.5-7.5 by addition of 125 kg of aqueous Sodium Hydroxide 45%. After cooling to a temperature of 45-50° C., the pH value is adjusted to 8-9 by addition of about 4 kg of aqueous Sodium Hydroxide 45%. Subsequently the mixture is cooled to 30-35° C. and centrifuged. The residue thus obtained is washed with 340 l of water and 126 l of isopropanol and then with water until chlorides elimination.
The wet product is dried under vacuum at a temperature of about 45-55° C. which leads to 358 kg of crude flibanserin polymorph A. The crude product thus obtained is loaded in a reactor with 1750 kg of Acetone and the resulting mixture is heated under stirring until reflux. The obtained solution is filtered and the filtrate is concentrated by distillation. The temperature is maintained for about 1 hour 0-5° C., then the precipitate solid is isolated by filtration and dried at 55° C. for at least 12 hours.
The final yield is 280 kg of pure flibanserin polymorph A.
CLIP
Flibanserin may be prepared by reacting 1-(phenylvinyl)-2,3-dihydro-1H-benzimidazol-2-one (I) with 1,2-dichloroethane (II) in the presence of NaH in warm dimethylformamide. The resulting 1-(2-chloroethyl)-2,3-dihydro-1H-benzimidazol-one (III) is in turn coupled with commercially available m-trifluoromethylphenylpiperazine hydrochloride (IV) in the presence of sodium carbonate and catalytic potassium iodide in refluxing ethanol. The crude flibanserin hydrochloride (V) is then dissolved in aqueous ethanol and the pure base is precipitated upon addition of sodium hydroxide.
PICK UP INTERMEDIATES FROM CHEM24H.COM
1-(1-phenylvinyl)-1,3-dihydro-2H-benzimidazol-2-one (I)
1,2-dichloroethane (II)
1-(2-chloroethyl)-1,3-dihydro-2H-benzimidazol-2-one (III)
1-[3-(trifluoromethyl)phenyl]piperazine; N-[3-(trifluoromethyl)phenyl]piperazine (IV)
1-(2-[4-[3-(trifluoromethyl)phenyl]piperazino]ethyl)-1,3-dihydro-2H-benzimidazol-2-one (V)
PATENT
According to another aspect of the present invention there is provided a novel compound or a salt thereof selected from the compounds of formula I, IV and VII:
Wherein R is hydrogen or an amino protecting group.
Preferable the amino protecting groups are selected from butyl, 1 ,1- diphenylmethyl, methoxymethyl, benzyloxymethyl, trichloroethoxymethyl, pyrrolidinomethyl, cyanomethyl, pivaloyloxymethyl, allyl, 2-propenyl, t- butyldimethylsilyl, methoxy, thiomethyl, phenylvinyl, 4-methoxyphenyl, benzyl, A- methoxybenzyl, 2,4-dimethoxybenzyl, 2-nitrobenzyl, t-butoxycarbonyl, benzyloxycarbonyl, phenoxycarbonyl, 4-chlorophenoxycarbonyl, A- nitrophenoxycarbonyl, methoxycarbonyl and ethoxycarbonyl. Still more preferable protecting groups are selected from t- butoxycarbonyl, ethoxycarbonyl, methoxycarbonyl, benzyloxycarbonyl, phenoxycarbonyl, phenylvinyl and 2-propenyl.
R1 is independently selected from chlorine, bromine, iodine, methanesulphonate, trifluoromethanesulphonate, paratoluenesulphonate or benzenesulphonate. Preferable R1 is independently selected from chlorine, bromine or iodine and more preferable R1 is chlorine.
Wherein R2 is hydrogen or an amino protecting group.
The amino protecting group may be any of the groups commonly used to protect the amino function such as alkyl, substituted alkyl, hetero substituted alkyl, substituted or unsubstituted unsaturated alkyl, alkyl substituted hetero atoms, substituted or unsubstituted phenyl, substituted or unsubstituted benzyl, alkyoxy carbonyl groups and aryloxy carbonyl groups.
Preferable the amino protecting groups are selected from butyl, 1 ,1 – diphenylmethyl, methoxymethyl, benzyloxymethyl, trichloroethoxymethyl, pyrrolidinomethyl, cyanomethyl, pivaloyloxymethyl, allyl, 2-propenyl, t- butyldimethylsilyl, methoxy, thiomethyl, phenylvinyl, 4-methoxyphenyl, benzyl, A- methoxybenzyl, 2,4-dimethoxybenzyl, 2-nitrobenzyl, t-butoxycarbonyl, benzyloxycarbonyl, phenoxycarbonyl, 4-chlorophenoxycarbonyl, A- nitrophenoxycarbonyl, methoxycarbonyl and ethoxycarbonyl. Still more preferable protecting groups are selected from t- butoxycarbonyl, ethoxycarbonyl, methoxycarbonyl, benzyloxycarbonyl, phenoxycarbonyl, phenylvinyl and 2-propenyl. The following examples are given for the purpose of illustrating the present invention and should not be considered as limitations on the scope and spirit of the invention.
EXAMPLES Example 1
A mixture of sodium hydroxide (47 gm) and i-(α-methylvinyl) benzimidazol-2-one (100 gm) in dimethylformamide (400 ml) was .stirred for 1 hour at room temperature. Dibromoethane (217 gm) was slowly added to the mixture and stirred at 1 hour 30 minutes. The resulting solution after addition water (500 ml) was extracted with ethyl acetate. The combined ethyl acetate extract washed with water. After drying the solvent was removed under vacuum to yield 132 gm of 1 ,3-dihydro-1-(2-bromoethyl)-3-isopropenyl-2H-benzimidazol- 2-one as a yellow oily liquid.
Example 2 A mixture of 1 ,3-dihydro-1-(2-bromoethyl)-3-isopropenyl-2H- benzimidazol-2-one (100 gm), diethanolamine (175 ml), sodium carbonate (40 gm) and potassium iodide (10 gm) was heated to 90 to 95 deg C and stirred for 2 hours. The reaction mass was cooled to room temperature and added water (500 ml). The resulting mixture extracted into ethyl acetate and the organic layer washed with water. After drying the solvent was removed under vacuum to yield 105 gm of 1 ,3-dihydro-1-[2-[N-bis-(2-hydroxyethyl)amino]ethyl]-3-isopropenyl- 2H-benzimidazol-2-one as a thick yellow oily liquid.
Example 3
To the mixture of 1 ,3-dihydro-1-[2-[N-bis-(2-hydroxyethyl)amino]ethyl]-3- isopropenyl-2H-benzimidazol-2-one (100 gm) obtained as in example 2 and chloroform (300 ml), thionyl chloride (95 ml) was slowly added. The mixture was heated to reflux and stirred for 2 hours. The excess thionyl chloride and chloroform was distilled off to yield 98 gm of 1 ,3-dihydro-1-[2-[N-[bis-(2- chloroethyl)amino]ethyl]-3-isopropenyl-2H-benzimidazol-2-one as a brown coloured sticky residue.
Example 4
1 ,3-dihydro-1-[2-[N-[bis-(2-chloroethyl)amino]ethyl]-3-isopropenyl-2H- benzimidazol-2-one (98 gm) obtained as in example 3 was added to water (500 ml) and concentrated hydrochloric acid (200 ml) mixture. The mixture was heated to 60 to 65 deg C and stirred for 1 hour. The contents of the flask cooled to room temperature and pH of the solution adjusted to 9 – 10 with 10% sodium hydroxide solution. The resulting solution extracted with ethyl acetate and washed the organic layer with water. Evaporate the solvent under reduced pressure to yield 82 gm of 1 ,3-dihydro-1-[2-[N-bis-(2-chloroethyl)amino]ethyl]- 2H-benzimidazol-2-one as a dark brown coloured oily liquid
Example 5
A mixture of 1 ,3-dihydro-1-[2-[N-bis-(2-chloroethyl)amino]ethyl]-1,2-H- benzimidazol-2-one (82 gm) obtained as in example 4, xylene (300 ml) and m- trifluoromethyl aniline (58 gm) was refluxed for 64 hours. The reaction mass was cooled to room temperature and filtered to obtain 1-[2-(4-(3- thfluoromethylphenyl)piperazin-1-yl)ethyl]-2,3-dihydro-1 H-benzimidazole-2-one hydrochloride (Flibanserin hydrochloride) as a light brown coloured solid.
The crude flibanserin hydrochloride was purified in isopropyl alcohol to give 85 gm of pure flibanserin hydrochloride as off white solid.
Example 6
Piperazine (12 gm), toluene(60 ml) and tetra butyl ammonium bromide (1 gm) mixture was heated to 60 deg C, added 1 ,3-dihydro-1-(2-bromoethyl)-3- isopropenyl-2H-benzimidazol-2-one (10 gm) and stirred for 4 hours at 90 to 95 deg C. The mixture was cooled to 60 deg C and added water (50 ml). The separated toluene layer distilled under vacuum to give 8.5 gm of 1 ,3-dihydro-1- (2-piperazinyl)ethyl-3-isopropenyl-2H-benzimidazol-2-one as a white solid.
Example 7
To the mixture of concentrated hydrochloric acid (20 ml) and water (100 ml) was added 1 ,3-dihydro-1-(2-piperazinylethyl)-3-isopropenyl-2H- benzimidazol-2-one (10 gm) obtained as in example 6 and heated to 60 to 65 deg C 1 hour. The mixture was cooled to room temperature and pH of the solution was adjusted to 9 – 10 with 10% sodium hydroxide solution, extracted with ethyl acetate and the organic layer was washed with water. After drying the solvent was removed under vacuum to yield 8.5 gm of 1 ,3-dihydro-1-(2- piperazinyl ethyl)-2H-benzimidazol-2-one as a white solid.
Example 8
3-trifluoromethylaniline (40 gm) and hydrobromic acid (85 ml; 48- 50%w/w) mixture was cooled to 0 to 5 deg C. To this mixture added sodium nitrite solution (18.5 gm in 25 ml of water) at 5 to 10 deg C and copper powder (1 gm). The temperature was slowly raised to 50 to 55 deg C and stirred for 30 minutes. Added water (200 ml) to reaction mass and applied steam distillation, collected m-trifluoromethylbromobenzene as oily liquid. The oily liquid washed with sulfuric acid for two times (2 X 10 ml) followed by washed with water (2 X 20 ml) and dried the liquid with sodium sulphate to give 22 gm of m- trifluoromethylbromobenzene.
Example 9
To a mixture of 1 ,3-dihydro-1-(2-piperazinyl ethyl)-2H-benzimidazol-2- one (10 gm) obtained as in example 7, m-trifluoromethylbromobenzene (9 gm) obtained as in example 8, sodium tert-butoxide (5.5 gm), palladium acetate (4.5 mg) and xylene (80 ml) was added tri-tert.-butylphosphine (0.2 ml). The mixture was heated to 120 deg C and stirred for 3 hours. The reaction mass was cooled, added water (100 ml) and extracted with ethyl acetate and the organic layer was washed with water. After drying the solvent was removed under vacuum to yield
10 gm of 1-[2-(4-(3-trifluoromethylphenyl)piperazin-1-yl)ethyl]-2,3-dihydro-1 H- benzimidazole-2-one (Flibanserin).
Example 10
To a mixture of 1 ,3-dihydro-1-[2-[N-bis-(2-hydroxyethyl)amino]ethyl]-3- isopropenyl-2H-benzimidazol-2-one (100 gm) obtained as in example 3, cyclohexane (400 ml) and sodium carbonate (35 gm) was added benzene sulfonyl chloride (116 gm) at room temperature. The mixture was heated to 80 to
85 deg C and stirred for 8 hours . The contents were cooled to room temperature and added water (500 ml). Distilled the organic layer to give 182 gm of 1 ,3-dihydro-1-[2-[N-[bis-(2-benzenesulfonyloxy)- ethyl]amino]ethyl]-3- isopropenyl- 2H-benzimidazol-2-one.
Example 11
1 ,3-dihydro-1 -[2-[N-[bis-(2-benzenesulfonyloxy)- ethyl]amino]ethyl]-3- isopropenyl- 2H-benzitηidazol-2-one (100 gm) obtained as in example 10, dimethylformamide (500 ml) and sodium corbonate (18 gm) was mixed and heated to 70 deg C. To the mixture was added m-trifluoromethyl aniline (27 gm) and heated to 80 to 85 deg C, stirred for 5 hours. The reaction mass was cooled and added water (2000 ml), filtered the solid to yield 1 ,3-dihydro-1-[2-[4-(3- trifluoromethylphenyl)piperazinyl]ethyl]-3-isopropenyl-2H benzimidazol-2-one. Example 12
1 ,3-dihydro-1-[2-[N-[bis-(2-benzenesulfonyloxy)- ethyl]amino]ethyl]-3- isopropenyl- 2H-benzimidazol-2-one (100 gm) obtained as in example 11 added to the mixture of water (500 ml) and concentrated hydrochloric acid (200 ml), heated to 65 deg C and stirred for 1 hour. The reaction mass was cooled to room temperature and pH adjusted to 10 to 10-5 with 10% sodium hydroxide solution. The resulting mixture was extracted with ethyl acetate and the organic
■ layer was washed with water. After drying the solvent was removed under vacuum to yield 87 gm of 1-[2-(4-(3-trifluoromethylphenyl)piperazin-1-yl)ethyl]- 2,3-dihydro-1 H-benzimidazole -2-one (Flibanserin).
Paper
Journal of Pharmaceutical and Biomedical Analysis, v.57, 2012 Jan 5, p.104(5)
Isolation and structural elucidation of flibanserin as an adulterant in a health supplement used for female sexual performance enhancement
Low, Min-Yong et al
http://www.sciencedirect.com/science/article/pii/S0731708511004833
This proposed formula and structure was further confirmed by 1H and 13C NMR data which indicated the presence of 20 carbon atoms and 21 protons.
1H NMR
13C NMR
1D and 2DNMR data were used to assign the protons and carbon atoms.
In the1H NMR spectrum , a sharp singlet at 10.00 ppm integrating for one
proton is a typical proton attached to nitrogen. HMBC correlated this proton to C-2, C-4, and C-9 suggesting that it was H-3.
Complex signals were observedbetween 7.00 to 7.31 ppm, integrating for eight protons. A triplet at 7.31 ppm,integrating for a proton has a coupling constant of 8.0 Hz. HMBC correlated thisproton with C-16, C-19, and C-21 suggesting that it was H-20.
A double-doubletsplitting pattern at chemical shift 7.11 ppm, integrating for a proton, has couplingconstants of 6.3 Hz and 1.6 Hz.
HMBC correlated this proton to C-6, C-7, and C-9 showing that it was H-8. Overlapped signals were observed from 7.04 ppm to7.10 ppm, integrating for five protons. A double-doublet splitting pattern at 7.01ppm with coupling constant 8.0 Hz and 2.0 Hz, integrating for a proton was
observed.
HMBC correlated this proton to C-17 suggesting that it was either H-19or H-21. Four triplet signals were also observed from 2.73 ppm to 4.08 ppm,integrating for a total of twelve protons.
Two of these triplet signals at 2.74 ppmand 3.22 ppm integrated for four protons each, suggesting overlapping signals ofmethylene protons. This was further confirmed by 13C and DEPT NMR.
13C and DEPT NMR data showed the signals of four methylene, eight methineand six quaternary carbon atoms. The DEPT signals at 53.1 ppm and 48.6 ppmhave intensities which were double of those from the rest of the methylene carbonsignals, suggesting two methylene carbon atoms each contributing to the signal at 53.1 ppm and 48.6 ppm.
DEPT
HMQC results further indicated that these two methylene carbon signals at 53.1 ppm and 48.6 ppm were correlated to the protons signal at 2.73 ppm and 4.08 ppm respectively, which corresponded to four protons each. The finding confirmed overlapping methylene carbon signals (at 53.1 ppm and 48.6 ppm) and methylene proton signals (at 2.73 ppm and 4.08 ppm). Hence, the unknown compound has six methylene carbon atoms with a total of twelve methylene protons.
The chemical shifts of the twelve methylene protons suggested that they were attached to relatively electronegative atoms. It was speculated that the six methylene groups were attached to the nitrogen atoms and the electron withdrawing effect of these electronegative nitrogen atoms resulted in the deshielding of the protons. HMBC and COSY correlations were used to assign the rest of the protons
HMBC
HMQC
COSY
The 13C NMR data showed that there were two quaternary carbon at
155.6 ppm and 151.3 ppm. The carbon with chemical shift 155.6 ppm was C-2. Inthe structure of imidazolone, carbonyl carbon C-2 was attached to two nitrogenatoms which helped to withdraw electrons from oxygen to C-2. Hence, C-2 wasless deshielded as compared to a normal carbonyl carbon which has chemical shiftabove 170 ppm.
Eight methine carbons and two quaternary carbons with chemicalshifts above 108 ppm suggested the presence of two aromatic rings. Thequaternary carbon with chemical shift 125.4 ppm was C-22 which was attached tothree fluorine atoms. Due to the strong electron withdrawing effect of the fluorineatoms, C-22 was highly deshielded and had a high chemical shift.
The IR spectrum of the isolated compound showed absorption bands of amide (νC=O 1685 cm-1, νN-H (stretch) 3180 cm-1, νN-H (bending) 1610 cm-1), alkyl fluoride (νC-F1077 cm-1, 1112 cm-1, 1158 cm-1), aromatic ring (ν Ar-H 3028 cm-1, 3078 cm-1 andνC=C 1401 cm-1, 1446 cm-1, 1453 cm-1, 1468 cm-1, 1487 cm-1) and alkane (νC-H2891 cm-1, 2930 cm-1 2948 cm-).
FOR MASS, HMBC ETC SEE………http://orgspectroscopyint.blogspot.in/2015/06/flibanserin.html
NMR PREDICT
13C NMR PREDICT
COSY PREDICT
NMR PREDICT FROM MOLBASE
PATENT
US5576318, 1996
1 H NMR (DMSO-d6 /CDCL3 5:2) 11.09 (b, 1H), 11.04 (s, 1H), 7.5-6.9 (SH), 4.36 (t, 2H), 4.1-3.1 (10 H)
UPDATES………..
A Facile Route of Synthesis for Making Flibanserin

REFERENCES
- Borsini F, Evans K, Jason K, Rohde F, Alexander B, Pollentier S (summer 2002). “Pharmacology of flibanserin”. CNS Drug Rev. 8 (2): 117–142. doi:10.1111/j.1527-3458.2002.tb00219.x. PMID 12177684.
- Jolly E, Clayton A, Thorp J, Lewis-D’Agostino D, Wunderlich G, Lesko L (April 2008). “Design of Phase III pivotal trials of flibanserin in female Hypoactive Sexual Desire Disorder (HSDD)”. Sexologies 17 (Suppl 1): S133–4. doi:10.1016/S1158-1360(08)72886-X.
- Spiegel online: Pharmakonzern stoppt Lustpille für die Frau, 8 October 2010 (in German)
- Nygaard I (November 2008). “Sexual dysfunction prevalence rates: marketing or real?”. Obstet Gynecol 112 (5): 968–9.doi:10.1097/01.AOG.0000335775.68187.b2. PMID 18978094.
- Clayton AH (July 2010). “The pathophysiology of hypoactive sexual desire disorder in women”. Int J Gynaecol Obstet 110 (1): 7–11.doi:10.1016/j.ijgo.2010.02.014. PMID 20434725.
- Pfaus JG (June 2009). “Pathways of sexual desire”. J Sex Med 6 (6): 1506–33. doi:10.1111/j.1743-6109.2009.01309.x.PMID 19453889.
- Yves Aubert, Thesis, Leiden University. (Dec 11, 2012) Sex, aggression and pair-bond: a study on the serotonergic regulation of female sexual function in the marmoset monkey
- Viagra for women?
- Marazziti D, Palego L, Giromella A, et al. (June 2002). “Region-dependent effects of flibanserin and buspirone on adenylyl cyclase activity in the human brain”. Int. J. Neuropsychopharmacol. 5 (2): 131–40. doi:10.1017/S1461145702002869.PMID 12135537.
- Podhorna J, Brown RE (June 2000). “Flibanserin has anxiolytic effects without locomotor side effects in the infant rat ultrasonic vocalization model of anxiety”. Br J Pharmacol 130 (4): 739–746. doi:10.1038/sj.bjp.0703364. PMC 1572126.PMID 10864879.
- Brambilla A, Baschirotto A, Grippa N, Borsini F (December 1999). “Effect of flibanserin (BIMT 17), fluoxetine, 8-OH-DPAT and buspirone on serotonin synthesis in rat brain”. Eur Neuropsychopharmacol 10 (1): 63–7. doi:10.1016/S0924-977X(99)00056-5.PMID 10647099.
| EP0200322A1 * | Mar 18, 1986 | Nov 5, 1986 | H. Lundbeck A/S | Heterocyclic compounds |
| BE904945A1 * | Title not available | |||
| GB2023594A * | Title not available | |||
| US3472854 * | May 29, 1967 | Oct 14, 1969 | Sterling Drug Inc | 1-((benzimidazolyl)-lower-alkyl)-4-substituted-piperazines |
| US4954503 * | Sep 11, 1989 | Sep 4, 1990 | Hoechst-Roussel Pharmaceuticals, Inc. | 3-(1-substituted-4-piperazinyl)-1H-indazoles |
update………..
1-(2-(4-(3-(Trifluoromethyl)phenyl)piperazin-1-yl)ethyl)-1H-benzo[d]imidazol-2(3H)-one (1)

A novel and efficient route of synthesis for making flibanserin via 2-ethoxy-1H-benzo[d]imidazole (12) was described with excellent yield. This protocol provided a more facile approach toflibanserin.
A Facile Route of Synthesis for Making Flibanserin
http://pubs.acs.org/doi/abs/10.1021/acs.oprd.6b00108

aReagents and conditions: (a) ethyl benzoylacetate, 200 °C; (b) dichloroethane, NaH, DMF; (c) conc HCl (aq); (d) 1-(3-(trifluoromethyl)phenyl)piperazine hydrochloride, Na2CO3, KI, EtOH; (e)
- 3.Bietti, G.; Borsini, F.; Turconi, M.; Giraldo, E.; Bignotti, M. For treatment of central nervous system disorders. U.S. Patent 5,576,318, 1996.
- 4.Mohan Rao, D.; Krishna Reddy, P.; Venkat Reddy, B. Preparing benzoimidazol-2-one compound, useful to prepare flibanserin, comprises reacting benzoimidazol-2-one compound with 2-(2-hydroxy-ethylamino)-ethanol to give (bis-(hydroxy-ethyl)-amino)-ethyl-benzoimidazol-2-one compound. PCT. Int.WO2,010,128,516, 2010.5.
- 5.Vernin, G.; Domlog, H.; Siv, C.; Metzger, J.; El-Shafei, A. K.Synthesis of 1-alkyl and 1, 3-dialkyl-2-benzimidazolones from 1-alkenyl-2-benzimidazolones using phase-transfer catalysis technique J. Heterocycl. Chem. 1981, 18, 85– 89, DOI: 10.1002/jhet.5570180118
-
A patent application for the new synthetic route has been filed in China (CN201610527244.4).

aReagents and conditions: (a) ethyl acetoacetate, KOH, EtOH, xylene, reflux, 56%; (b) 1,2-dibromoethane, K2CO3, DMF, 50 °C, 50%; (c) K2CO3, CH3CN, 70 °C, 80%; (d) conc. HCl (aq), isopropanol, 70 °C; (e) NaOH (aq), rt, 72% over two steps.

aReagents and conditions: (a) tetraethyl orthocarbonate, AcOH, 70 °C, 94%; (b) 1-bromo-2-chloroethane, K2CO3, acetone, reflux, 75%; (c) K2CO3, NaI, H2O, reflux, 92%; (d) conc. HCl (aq), isopropanol, 70 °C; (e) NaOH (aq), 68% over two steps.
//////////////
Route to Benzimidazol-2-ones via Decarbonylative Ring Contraction of Quinoxalinediones: Application to the Synthesis of Flibanserin, A Drug for Treating Hypoactive Sexual Desire Disorder in Women and Marine Natural Product Hunanamycin Analogue


Route to Benzimidazol-2-ones via Decarbonylative Ring Contraction of Quinoxalinediones: Application to the Synthesis of Flibanserin, A Drug for Treating Hypoactive Sexual Desire Disorder in Women and Marine Natural Product Hunanamycin Analogue
INTRODUCTION
Benzimidazol-2-ones 1 are an important class of heterocycles and a privileged scaffold in medicinal chemistry. They consist of cyclic urea fused with the aromatic backbone, which can potentially interact in a biological system by various noncovalent interactions such as hydrogen bonding and π stacking. Benzimidazolone derivatives exhibit a wide range of biological activities, and they are useful in treating various diseases including cancer, type II diabetes, central nervous system disorders, pain management, and infectious disease.1 Selected compounds embedded with a benzimidazol-2-one moiety along with their use are captured in Figure 1. It is worth mentioning that oxatomide drug with a benzimidazol-2-one core was approved for marketing a few years ago.2a Very recently, US Food and Drug Administration approved a new drug called flibanserin for the treatment of hypoactive sexual desire disorder (HSDD) in females, which contains benzimidazol-2- one motif.2b
CONCLUSIONS
We have developed a mild and new protocol for the synthesis of benzimidazol-2-ones from quinoxalinediones through decarbonylation. The present methodology can be an addition to the toolbox to prepare benzimidazolones, and it will be useful in medicinal chemistry, particularly, late-stage functionalization of natural products, drug scaffolds, or an intermediate containing quinoxaline-2,3-diones. As direct application of this method, we have successfully developed a new route for the synthesis of recently approved drug flibanserin and a urea analogue of antibiotic natural product hunanamycin A. Later application demonstrates the utility of the present method in late-stage functionalization
Synthesis of 1-(2-(4-(3-(trifluoromethyl)phenyl)piperazin-1-yl)ethyl)-1,3-dihydro-2Hbenzo[d]imidazol-2-one (Flibanserin)
Flibanserin hydrochloride as white solid.
1H NMR (400MHz ,DMSO-d6) 11.06 (s, 1 H), 10.93 (br. s., 1 H), 7.54 – 7.41 (t, J = 7.9 Hz, 1 H), 7.36 – 7.22 (m, 3 H), 7.15 (d, J = 7.6 Hz, 1 H), 7.09 – 7.01 (m, 3 H), 4.30 (t, J = 6.7 Hz, 2 H), 4.01 (d, J = 11.6 Hz, 2 H), 3.75 (d, J = 10.4 Hz, 2 H), 3.54 – 3.43 (d, J = 4.2 Hz 2 H), 3.31 – 3.10 (m, 4 H);
HRMS (ESI): m/z calculated for C20H22ON4F3[M+H]+ 391.1740 found 391.1743;

Scheme 4. Synthesis of Flibanserin through Ring Contraction
The same methodology was applied for the synthesis of flibanserin, also known as “female viagra”, which is the first approved medication for treating HSDD in women and is classified as a multifunctional serotonin agonist antagonist.(14, 15) Our synthesis of flibanserin commenced with 1-benzyl-1,4-dihydroquinoxaline-2,3-dione 36,(16) which was reacted with known chloride 37(17) under the basic condition in DMF to give the desired product 38 in good yield. Compound 38 was subjected for the decarbonylative cyclization under the optimized condition to afford the product 39 in 59% yield. Finally, the benzyl group was deprotected using trifluoromethanesulfonic acid in toluene under microwave irradiation,(8b, 18) which gave flibanserin in excellent yield (Scheme 4). The final product was isolated as HCl salt, and all of the spectral data are in agreement with the published data.(15c)

Rahul D. Shingare completed his M.Sc (Chemistry) from Fergusson College, Pune in 2008. He worked as a research associate in Ranbaxy and Lupin New drug discovery center, Gurgaon and Pune respectively until 2012 and currently pursuing his doctoral research in NCL – Pune from 2012.
Current Research Interests: Antibacterial Natural Product Hunanamycin A: Total Synthesis, SAR and Related Chemistry.
e-mail: rd.shingare@ncl.res.in
Akshay Kulkarni completed his M.Sc. from Ferguson College, Pune University in the year 2015 and joined our group as a Project Assistant in the month of October, 2015.
Current research interest: Synthesis of silicon incorporated biologically active antimalerial compounds.
e-mail : as.kulkarni@ncl.res.in
Dr.D. Srinivasa Reddy
Organic Chemistry Division
CSIR-National Chemical Laboratory
-
See, previous synthesis of Flibanserin:
(a) Bietti, G.; Borsini, F.; Turconi, M.; Giraldo, E.; Bignotti, M. For treatment of central nervous system disorders. U.S. Patent 5,576,318, 1996.
(b) Mohan, R. D.; Reddy, P. K.;Reddy, B. V. Process for the preparation of Flibanserin involving novel intermediates. WO2010128516 A2,2010.
(c) Yang, F.; Wu, C.; Li, Z.; Tian, G.; Wu, J.; Zhu, F.; Zhang, J.; He, Y.; Shen, J. A Facile route of synthesis for making Flibanserin Org. Process Res. Dev. 2016, 20, 1576 DOI: 10.1021/acs.oprd.6b00108
[ACS Full Text ], [CAS]
], [CAS] -
Xueong, X. Preparation method of Flibanserin. CN104926734 A, 2015.
//////////
AVASCULAR NECROSIS ; POST OPERATIVE AND POST SURGICAL COMPLICATIONS CASE ; AYURVEDA E.T.G AYURVEDASCAN DIAGNOSIS AND APPROACH
Recently a case of AVASCULAR NECROSIS , bilateral operated before one year, developed major complications in his both HIP-Joints severely. Surgeon, who have taken the case under his supervision, advised him to for HIP REPLACEMENT.
In this crisis satge, patient approched me and asked for the help for AYURVEDA TREATMENT.
HIS ETG AyurvedaScan traces are given below and some essential details are given below.
[TO BE LOADED SOON]
FDA Guidance on Polymorphic Compounds in Generic Drugs

The guidance issued by the US Food and Drug Administration advises companies on how to treat polymorphic drug compounds—those that exhibit multiple structural forms—in filing abbreviated new drug applications (ANDAs). The bottom line, according to the guidance, is that generic drug products containing the polymorphs be the “same” as the reference listed drug (RLD) in active ingredients, bioavailability, and bioequivalence.
The guidance pertains to orally available drugs that are either solid- or suspension-dosage products.
Polymorphisms arise when compounds are identical chemically, but not structurally. This can happen when two solids take on different crystalline forms—such as graphite and diamond; when molecules are disordered and fail to produce a repeatable crystal lattice, as is the case for the molecules in glass; or when solvent is trapped inside the crystal structure—as in hydrates, where water molecules are found within crystals.
The guidance notes that different polymorphisms may alter physical properties of compounds and affect their solubility, which in turn can alter their bioavailability or bioequivalence. In addition, polymorphic forms of a compound may alter the way the compound behaves during production, which again, may alter the finished drug’s biological activities.
On this latter point, the guidance specifically states, “Since an ANDA applicant should demonstrate that the generic drug product can be manufactured reliably using a validated process, we recommend that you pay close attention to polymorphism as it relates to pharmaceutical processing.”
The guidance also emphasizes the effect polymorphisms may have on drug stability, which again, may alter the drug’s biological activity. But the guidance goes on to say that “it is the stability of the drug product and not stability of the drug substance polymorphic form that should be the most relevant measure of drug equality.” Otherwise, a generic drug can be considered the “same” as the active ingredient in an RLD if the generic compound conforms to the standards set out in a United States Pharmacopeia (USP) monograph, if one exists for that particular drug substance.
These standards generally include the chemical name, empirical formula, and molecular structure of the compound. However, the “FDA may prescribe additional standards that are material to the sameness of a drug substance.” But as concerns polymorphisms, the guidance goes on to say “…differences in drug substance polymorphic forms do not render drug substances different active ingredients for the purposes of ANDA approvals….”
Finally, the guidance reminds ANDA applicants that the biological performance characteristics of a drug are also dependent on the drug’s formulation and advises applicants to consider the properties of both the drug substance and formulation excipients, when assessing “sameness.”
A sponsor of an Abbreviated New Drug Application (ANDA) must have information to show that the proposed generic product and the innovator product are both pharmaceutically equivalent and bioequivalent, and therefore, therapeutically equivalent.
Many pharmaceutical solids exist in several crystalline forms and thus exhibit polymorphism. Polymorphism may result in differences in the physico-chemical properties of the active ingredient and variations in these properties may render a generic drug product to be bioinequivalent to the innovator brand. For this reason, in ANDAs, careful attention is paid to the effect of polymorphism in the context of generic drug product equivalency.
This review ..Adv Drug Deliv Rev. 2004 Feb 23;56(3):397-414……discusses the impact of polymorphism on drug product manufacturability, quality, and performance. Conclusions from this analysis demonstrate that pharmaceutical solid polymorphism has no relevance to the determination of drug substance “sameness” in ANDAs.
Three decision trees for solid oral dosage forms or liquid suspensions are provided for evaluating when and how polymorphs of drug substances should be monitored and controlled in ANDA submissions. Case studies from ANDAs are provided which demonstrate the irrelevance of polymorphism to the determination of drug substance “sameness”. These case studies also illustrate the conceptual framework from these decision trees and illustrate how their general principles are sufficient to assure both the quality and the therapeutic equivalence of marketed generic drug products.
read
ANDAs: Pharmaceutical Solid Polymorphism – Food and Drug … click here
also
Issues of Polymorphism and Abbreviated New Drug Applications click here
and
POLYMORPHISM OF DRUGS – Seventh Street Development Group click here
An Overview of Solid Form Screening During Drug … – ICDD..http://www.icdd.com/ppxrd/10/presentations/PPXRD-10_Ann_Newman.pdf
http://www.ivtnetwork.com/sites/default/files/Polymorphism_01.pdf
Although polymorph/salt screening should ideally be performed to select the optimum solid form upon selection of the lead compound prior to animal pharmacokinetic (PK) studies, these screening study can be costly and time consuming. But the consequences of late discovery of a thermodynamic form are grave, so there must be a strategy to minimize the risk without spending a large amount of resources.
We find this right strategy based on early BCS classification of new compounds. We tailor the upfront polymorph/salt studies based on the risk in bioavailability, stability and manufacture-ability. Since regulatory agencies worldwide require the use of the same salt across preclinical and clinical studies, for insoluble or unstable compounds, salt screening is done early to enable further compound development.
Once salt is selected, the polymorph screening of the selected salt if soluble may be done a little later after animal study. However it is paramount to confirm 1) the polymorph in use is stable in the toxicological vehicle, 2) no changes of solid forms during shipping and storage, 3) no significant degradation upon storage.
Should there be polymorphic changes such as formation of a hydrate in the animal vehicle resulting in lowered solubility and precipitation of the hydrate, or formation of a hydrate when exposed to humidity during shipping and storage, early discovery of the stable forms will enable consistent animal exposure and avoid study repeats and delays in timelines.
Therefore, although most companies do not perform comprehensive polymorph screening until late in the development cycle, we recommend identification of a thermodynamic stable form within the confine of not only the API manufacture processes but also in the designated animal and human formulations.
For instance, for a drug product manufactured by direct compression, the solidstate properties of the active ingredient will likely be critical to the manufacture of the drug product, particularly when it constitutes the bulk of the tablet mass.
On the other hand, for a drug product manufactured by wet granulation, the solidstate properties of the active ingredient may no longer be important but the potential for polymorphic conversion is high in the presence of high moisture contents. In the context of the effect of polymorphism on pharmaceutical processing, what is most relevant is the ability to consistently manufacture a drug product that conforms to applicable in-process controls and release specifications.
This upfront work is especially critical to insoluble compounds prone to varied oral bioavailability in animal and human.

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO



















![[1860-5397-7-57-i28]](https://i0.wp.com/www.beilstein-journals.org/bjoc/content/inline/1860-5397-7-57-i28.png)