Ezatiostat……….designed to stimulate the production of blood cells in the bone marrow

Ezatiostat
168682-53-9 (Ezatiostat); 286942-97-0 (Ezatiostat HCl salt)
gamma-Glu-S-BzCys-PhGly diethyl ester
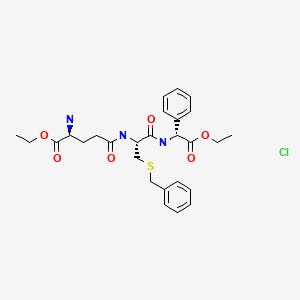
Ezatiostat hydrochloride
Target: glutathione S-transferase P1-1 (GSTP1-1) inhibitor
Pathway: hsa00480 Glutathione metabolism
Activity: Treatment of disorders of bone marrow cellular growth and differentiation
see http://www.ncbi.nlm.nih.gov/pubmed?term=TLK-199&cmd=search
| Telik, Inc. |
innovator
TLK199; TLK-199; TLK 199; Brand name: TELINTRA®。ethyl (2R)-[(4S)-4-amino-5-ethoxy-5-oxopentanoyl]-S-benzyl-L-cysteinyl-2- phenylglycinate.
ethyl (2S)-2-amino-5-[[(2R)-3-benzylsulfanyl-1-[[(1R)-2-ethoxy-2-oxo-1-phenylethyl]amino]-1-oxopropan-2-yl]amino]-5-oxopentanoate.
IUPAC/Chemical name:
(S)-ethyl 2-amino-5-(((R)-3-(benzylthio)-1-(((S)-2-ethoxy-2-oxo-1-phenylethyl)amino)-1-oxopropan-2-yl)amino)-5-oxopentanoate
C27H35N3O6S
Exact Mass: 529.2246
nmr.http://www.medkoo.com/Product-Data/Ezatiostat/ezatiostat-QC-CRB40225web.pdf
Telintra is a small molecule product candidate designed to stimulate the production of blood cells in the bone marrow. Many conditions are characterized by depleted bone marrow, including myelodysplastic syndrome, a form of pre-leukemia in which the bone marrow produces insufficient levels of one or more of the 3 major blood elements (white blood cells, red blood cells and platelets). A reduction in blood cell levels is also a common, toxic effect of many standard chemotherapeutic drugs.
Ezatiostat is a liposomal small-molecule glutathione analog inhibitor of glutathione S-transferase (GST) P1-1 with hematopoiesis-stimulating activity. After intracellular de-esterification, the active form of ezatiostat binds to and inhibits GST P1-1, thereby restoring Jun kinase and MAPK pathway activities and promoting MAPK-mediated cellular proliferation and differentiation pathways. This agent promotes the proliferation and maturation of hematopoietic precursor cells, granulocytes, monocytes, erythrocytes and platelets
Phase II trial myelodysplastic syndrome (MDS): Cancer. 2012 Apr 15;118(8):2138-47.
Phase I trial myelodysplastic syndrome (MDS): J Hematol Oncol. 2012 Apr 30;5:18. doi: 10.1186/1756-8722-5-18; Blood. 2009 Jun 25;113(26):6533-40; J Hematol Oncol. 2009 May 13;2:20.
Ezatiostat hydrochloride is the hydrochloride acid addition salt of ezatiostat. Ezatiostat, also known as TLK199 or TER 199, is a compound of the formula:
Ezatiostat has been shown to induce the differentiation of HL-60 promyelocyte leukemia cells in vitro, to potentiate the activity of cytotoxic agents both in vitro and in vivo, and to stimulate colony formation of all three lineages of hematopoietic progenitor cells in normal human peripheral blood.
In preclinical testing, ezatiostat has been shown to increase white blood cell production in normal animals, as well as in animals in which white blood cells were depleted by treatment with cisplatin or fluorouracil. Similar effects may provide a new approach to treating myelodysplastic syndrome (MDS).
Many conditions, including MDS, a form of pre-leukemia in which the bone marrow produces insufficient levels of one or more of the three major blood elements (white blood cells, red blood cells, and platelets), are characterized by depleted bone marrow. Myelosuppression, which is characterized by a reduction in blood cell levels and in a reduction of new blood cell generation in the bone marrow, is also a common, toxic effect of many standard chemotherapeutic drugs.
Ezatiostat hydrochloride in a liposomal injectable formulation was studied in a clinical trial for the treatment of MDS, and results from this trial, reported by Raza et al., J Hem. One, 2:20 (published online 13 May 2009), demonstrated that administration of TLK199 was well tolerated and resulted in multi-lineage hematologic improvement.
Ezatiostat hydrochloride in a tablet formulation has been evaluated in a clinical trial for the treatment of MDS, as reported by Raza et al., Blood, 113:6533-6540 (prepublished online 27 April 2009) and a single-patient report by Quddus et al., J Hem. One, 3:16 (published online 23 April 2010), and is currently being evaluated in clinical trials for the treatment of MDS and for severe chronic idiopathic neutropenia.
When used for treating humans, it is important that a crystalline therapeutic agent like ezatiostat hydrochloride retains its polymorphic and chemical stability, solubility, and other physicochemical properties over time and among various manufactured batches of the agent. If the physicochemical properties vary with time and among batches, the administration of a therapeutically effective dose becomes problematic and may lead to toxic side effects or to ineffective therapy, particularly if a given polymorph decomposes prior to use, to a less active, inactive, or toxic compound.
Therefore, it is important to choose a form of the crystalline agent that is stable, is manufactured reproducibly, and has physicochemical properties favorable for its use as a therapeutic agent.
Ezatiostat hydrochloride (USAN) has the molecular weight of 566.1, the trademark of Telintra®, and the CAS registry number of 286942-97-0. Ezatiostat hydrochloride has been evaluated for the treatment of myelodysplastic syndrome (MDS), in a Phase I-IIa study using a liposomal formulation (U.S. Pat. No. 7,029,695), as reported at the 2005 Annual Meeting of the American Society for Hematology (Abstract #2250) and by Raza et al. in Journal of Hematology & Oncology, 2:20 (published online on 13 May 2009); and in a Phase I study using a tablet formulation, as reported at the 2007 Annual Meeting of the American Society for Hematology (Abstract #1454) and by Raza et al. in Blood, 113:6533-6540 (prepublished online on 27 Apr. 2009), and in a single patient case report by Quddus et al. in Journal of Hematology & Oncology, 3:16 (published online on 23 Apr. 2010).
…………………………………………………………………
http://www.google.com/patents/US20110301376
Preparation of Ezatiostat Hydrochloride
In another aspect, this invention provides a process comprising the steps of contacting a compound of formula:
or a salt thereof with a compound of formula:
or a salt thereof and an activating agent under conditions which provide a compound of formula:
In one embodiment, the process further comprises deprotecting the compound of formula:
under conditions which provide a compound of formula:
or a salt thereof. In another embodiment, the compound provided is ezatiostat hydrochloride.
In another aspect, this invention provides a process comprising contacting a compound of formula:
or a salt thereof with an ethylating agent under conditions which provide a compound of formula:
In another embodiment, the process further comprises debenzylating the compound of formula:
under conditions which provide a compound of formula:
or a salt thereof.
In another aspect, this invention provides a process comprising the steps of contacting a compound of formula:
or a salt thereof having a t-butoxycarbonyl group with an activating agent and a compound of formula:
or a salt thereof under conditions which provide a compound of formula:
In another embodiment, the process further comprises deprotecting the tertiarybutyloxycarboyl (Boc) group under conditions to provide a compound of formula:
or a salt thereof.
Certain preferred embodiments of this invention are illustrated in the reaction scheme and described below. In the peptide coupling the amino acid reagents are used generally at a 1:1 molar ratio, and the activating reagent (isobutyl chloroformate) and the base (NMM) are used in slight excess over the amino acid reagents; while in the esterification of the N-BOC-L-glutamic acid γ-benzyl ester the esterifying agent (diethyl sulfate) and base are used in about 1.4-fold excess.
EXAMPLESAs relevant and unless otherwise noted, all operations were conducted under nitrogen purge and with stirring. Water was osmosis purified, and solvents were filtered. Unless otherwise stated, all temperatures are in degrees Celcius (° C.) and the following abbreviations have the following definitions:Et EthylHCl(g) HCl gasN-BOC or N-Boc N-tertiarybutyloxycarbonylL LiterKg KilogramNMM N-methylmorpholineMol Molew/w weight by weightExample 1Preparation of S-benzyl-L-cysteinyl-D-phenylglycine ethyl ester hydrochloride (3)Without stirring, 45.1 Kg N-BOC-S-benzyl-L-cysteine (1) was added to a 600 L jacketed glass-lined reactor, followed by 45 L ethyl acetate. Stirring was started and the temperature was reduced to 13° C. NMM, 15.3 Kg, was added over 50 minutes, and rinsed in with 6 L ethyl acetate, and stirring stopped. Ethyl acetate, 315 L, was added to an 800 L cooled jacketed glass-lined reactor, followed by 20.7 Kg isobutyl chloroformate, rinsed in with 11 L ethyl acetate, and the mixture cooled to −10° C. The N-BOC-S-benzyl-L-cysteine NMM salt solution was added to the 800 L reactor over 5 hours, its reactor rinsed with 11 L ethyl acetate, and the rinse solution added to the 800 L reactor, while maintaining the temperature at (−10˜−7)° C. D-Phenylglycine ethyl ester hydrochloride, 31.2 Kg, was added in 8 portions over 50 minutes, followed by 15.3 Kg NMM in 8 portions over 1.3 hours, rinsed in with 2×5 L portions of ethyl acetate, allowing the mixture to warm to −1° C. by the end of the addition. The mixture was gradually warmed to 1° C. for 30 minutes, then to 20° C. over 2 hours, and maintained at (20˜25)° C. for 5 hours. The reaction mixture was washed twice with water: the first time adding 66 L water, stirring at room temperature for 40 minutes, allowing the phases to separate for 30 minutes, then removing the aqueous phase; the second time adding 68 L water, bringing the pH to 1.9 with the addition of 0.45 L 36% hydrochloric acid, stirring at room temperature for 35 minutes, allowing the phases to separate for 1 hour, then removing the aqueous phase. The organic phase was then heated to 38° C., and the pressure reduced to about 0.25 bar until no further gas was released, then to about (0.07-0.1) bar and solvents removed by distillation until 266 L of distillate had been removed. Four cycles of addition of 45 L ethyl acetate and removal of 45 L solvent by distillation were performed, and the water content of the remaining mixture was checked to ensure that it was below 0.1%. With the mixture at 36° C., 194 L heptanes was added, maintaining the temperature about 36° C., and held at that temperature for 2.3 hours. A further 194 L heptanes was added, allowing the temperature to cool to 30° C., and the temperature then reduced to −1° C. over 2.3 hours and then to −5° C. over 1 hour, and N-BOC-S-benzyl-L-cysteinyl-D-phenylglycine ethyl ester recovered by filtration, washing twice with 30 L each of heptanes at −5° C., giving 85 Kg (63 Kg dry basis) N-BOC-S-benzyl-L-cysteinyl-D-phenylglycine ethyl ester. Without stirring, the damp N-BOC-S-benzyl-L-cysteinyl-D-phenylglycine ethyl ester was loaded into an 800 L jacketed glass-lined reactor, followed by 257 L ethyl acetate. Stirring was started and the temperature brought to 22° C., then the nitrogen purge stopped and 12.2 Kg hydrogen chloride gas was added through an immersion tube over 1.8 hours, allowing the temperature to increase to 38° C. The temperature was increased to 41° C., and the mixture held at that temperature for 9 hours. About 280 L of solvents were removed by distillation at that temperature and a pressure of (0.2˜0.1) bar over about 2 hours. Two cycles of addition of ethyl acetate and removal of solvent by distillation were performed, using 52 L in the first cycle and 77 L in the second cycle, and the viscous solution of S-benzyl-L-cysteinyl-D-phenylglycine ethyl ester hydrochloride (3) in ethyl acetate, 148 Kg, was cooled to room temperature and filtered into a storage drum.Example 2Preparation of N-BOC-L-glutamic acid α-ethyl ester (6)Without stirring, 41 Kg N-BOC-L-glutamic acid γ-benzyl ester (4) was added to an 800 L jacketed glass-lined reactor, followed by 2.5 L water and 123 L ethyl acetate. The mixture was then stirred until the N-BOC-L-glutamic acid γ-benzyl ester completely dissolved, keeping the temperature below 15° C. Potassium carbonate fine powder, 23.4 Kg, was added in five batches, and the mixture then heated to 55° C. and maintained at that temperature for 40 minutes, giving a heterogeneous and completely fluid mixture. Diethyl sulfate, 26.2 Kg, was added over 2 hours, and rinsed in with 5 L ethyl acetate, with the temperature remaining at about 52° C. The nitrogen purge was stopped and a solution of 20 Kg ammonium chloride in 73 L water at room temperature added over 2 hours to the mixture, maintaining the temperature near 50° C., then rinsing in with 10 L water. Nitrogen purging was resumed, and the mixture was maintained at about 50° C. for 3 hours, then lowered to about 45° C., the stirring stopped, the phases allowed to separate for 30 minutes, and the lower, aqueous, phase removed. The organic phase, containing N-BOC-L-glutamic acid γ-benzyl α-ethyl ester (5), was washed three times with water, each time adding 41 L water, stirring at room temperature for 30 minutes, allowing the phases to separate for 30 minutes, then removing the aqueous phase. The organic phase was heated to 35° C., and the pressure reduced, starting at about 0.2 bar and reducing as necessary until 82 Kg solvent had been removed by distillation, leaving about (70˜80) L of slightly opalescent solution. This solution was heated to 53° C., and 102 L heptanes was added, maintaining the same temperature. The solution was then filtered, rinsing with a further 13 L heptanes, then cooled to 32° C. to cause crystallization and maintained at that temperature for 1 hour. A further 66 L heptanes was added, and the mixture cooled to 22° C. and held for 1 hour, then cooled to −5° C. and held for another 1 hour. The mixture was then filtered to isolate the N-BOC-L-glutamic acid γ-benzyl α-ethyl ester (5), which was washed twice, each time with 25 L heptanes cooled to (−5˜0)° C., and dried under vacuum at 40° C., giving 39.3 Kg N-BOC-L-glutamic acid γ-benzyl α-ethyl ester (5).A 4000 L hydrogenator was purged with nitrogen, then under nitrogen sweep and no stirring loaded with 39.2 Kg N-BOC-L-glutamic acid γ-benzyl α-ethyl ester (5), 2.0 Kg 5% palladium on carbon, and 432 L ethyl acetate, and purged (3 bar) and decompressed (0.2 bar) twice with nitrogen and twice with hydrogen. Stirring was begun and the mixture heated to (37±2)° C., hydrogenated at that temperature under 2.8 bar hydrogen pressure until no further hydrogen absorption occurred, then held under 2.8 bar hydrogen pressure for 12 hours. Completion of hydrogenation was confirmed by thin-layer chromatography of a sample. The mixture was cooled to 28° C., the hydrogen purged from the hydrogenator, and the hydrogenator purged (2 bar) and decompressed (0.2 bar) twice with nitrogen. The mixture was filtered through a filter precoated with 10 Kg powdered cellulose in 200 L ethyl acetate, then the filter washed with the ethyl acetate used to form the precoat, giving a total of 626 Kg of a dilute ethyl acetate solution containing 29.5 Kg N-BOC-L-glutamic acid α-ethyl ester (6). This was distilled at (35˜40)° C. and (0.16˜0.18) bar to give 67 L of concentrated solution, then 29 L of ethyl acetate added and the solution redistilled to again give 67 L of concentrated solution.Example 3Preparation of Ezatiostat HydrochlorideThe concentrated solution of N-BOC-L-glutamic acid α-ethyl ester (6), 61.2 Kg (containing 27.8 Kg N-BOC-L-glutamic acid α-ethyl ester), was added to a 600 L jacketed glass-lined reactor, rinsed in with 5 L ethyl acetate, then cooled to 14° C. NMM, 10.8 Kg, was added over 50 minutes and rinsed in with 5 L ethyl acetate, then stirring stopped, giving an ethyl acetate solution of N-BOC-L-glutamic acid α-ethyl ester NMM salt. Ethyl acetate, 475 L, was added to a 1300 L cooled jacketed glass-lined reactor, followed by 14.5 Kg isobutyl chloroformate, rinsed in with 2×10 L ethyl acetate, and the mixture cooled to −11° C. The N-BOC-L-glutamic acid α-ethyl ester NMM salt solution was added to the 1300 L reactor over 1.3 hours, its reactor rinsed with 10 L ethyl acetate, and the rinse solution added to the 1300 L reactor, then stirred for an additional 30 minutes, while maintaining the temperature at about −13° C.S-benzyl-L-cysteinyl-D-phenylglycine ethyl ester hydrochloride (3) in ethyl acetate, 112 Kg (containing 41.3 Kg S-benzyl-L-cysteinyl-D-phenylglycine ethyl ester hydrochloride) was added in 4 portions over 45 minutes, and rinsed in with 5 L ethyl acetate, followed by 10.8 Kg NMM in 8 portions over 1.3 hours, rinsed in with 2×5 L portions of ethyl acetate, allowing the mixture to warm to −4° C. by the end of the addition. The mixture was gradually warmed to 30° C. over 2 hours, and maintained at (30˜35)° C. for 2 hours. The reaction mixture was washed twice with water: the first time adding 100 L water, heating to 41° C., allowing the phases to separate for 30 minutes, then removing the aqueous phase; the second time adding 100 L water, bringing the pH to 2.0 with the addition of 0.8 L 36% hydrochloric acid, stirring at 43° C. for 30 minutes, allowing the phases to separate for 1 hour, then removing the aqueous phase. The organic phase was then heated to 42° C., and the pressure reduced to about 0.25 bar until no further gas was released and solvents removed by distillation until 495 L of distillate had been removed. Four cycles of addition of 120 L ethyl acetate and removal of 120 L solvent by distillation were performed, and the water content of the remaining mixture was checked to ensure that it was below 0.1%. With the mixture at 42° C., 610 L of ethyl acetate was added, maintaining the temperature about 41° C., then heating to 58° C. to ensure dissolution. The solution was filtered, rinsing the filter with 18 L ethyl acetate, and the solution allowed to cool to 22° C. The nitrogen purge was stopped and 22.2 Kg hydrogen chloride gas was added through an immersion tube over 2 hours, then the mixture held at that temperature for 2 hours. The mixture was heated to 31° C. over 1.5 hours, and held at about that temperature for 15.5 hours. Solvents were removed by distillation at 33° C. and a pressure of about 0.13 bar over about 1.5 hours to give a volume of concentrated solution of about 630 L. Ethyl acetate, 100 L, was added, and the mixture cooled to 25° C. and held at that temperature for 30 minutes. The crude ezatiostat hydrochloride was recovered by filtration and washed with 30 L ethyl acetate, giving 113 Kg damp crude ezatiostat hydrochloride, which was dried at 40° C. under vacuum for 24 hours to give 52.8 Kg dry crude ezatiostat hydrochloride.Example 4Crystallization of Ezatiostat Hydrochloride to Form Pure Crystalline Ezatiostat Hydrochloride Ansolvate Form D61.5 Kg crude ezatiostat hydrochloride was added to a reactor at room temperature, followed by 399 liter (L) ethanol, and this mixture was heated to 68° C. to completely dissolve the ezatiostat hydrochloride, filtered, then allowed to cool to 65° C. and checked for clarity and the absence of crystallization. About 1.3 Kg of ezatiostat hydrochloride ansolvate form D was suspended in 9 L of ethyl acetate, and about one-half of this suspension was added to the ethanol solution. The mixture was cooled to 63° C. and the second half of the suspension added to the mixture. The resulting mixture was cooled gradually to 45° C., 928 L ethyl acetate was added, and the mixture was cooled to 26° C. and held at about that temperature for about 5 hours, then cooled to −2° C. The mixture, containing crystalline ezatiostat hydrochloride ansolvate, was filtered, and the residue washed twice with 65 L of chilled (0-5° C.) ethyl acetate. The crystalline ezatiostat hydrochloride ansolvate was dried at 30° C. for 48 hours, then cooled to room temperature and sieved. Analysis of the material by DSC and XRPD confirmed its identity as crystalline ezatiostat hydrochloride ansolvate, and Karl Fischer analysis showed a water content of 0.1%.Example 5Purifying Ezatiostat Hydrochloride Crystals to Form Pure Crystalline Ezatiostat Hydrochloride Ansolvate Form DCrude ezatiostat hydrochloride, 51.4 Kg, was added to a 600 L jacketed glass-lined reactor at room temperature, followed by 334 L of ethanol. The mixture was heated to 68° C. to completely dissolve the ezatiostat hydrochloride. The resulting solution was filtered into a 1300 L jacketed glass-lined reactor, and an additional 27 L ethanol warmed to 66° C. used to rinse the first reactor into the second reactor through the filter. The resulting solution in the second reactor was cooled to 63° C. and checked for complete dissolution; then 4 L of a seeding suspension of crystalline ezatiostat hydrochloride ansolvate in ethyl acetate was added, and the mixture cooled to 60° C. The remaining 4 L of the seeding suspension was added, and the mixture cooled to 47° C. over 2 hours. The solids in the mixture were shown by DSC to contain more than one form of ezatiostat hydrochloride, so the stages of heating to dissolution, cooling, and adding seeding suspension (this time 2×2 L), were repeated, then the mixture cooled to 41° C. This time the solids in the mixture were confirmed by DSC to be crystalline ezatiostat hydrochloride ansolvate. Ethyl acetate, 776 L, was added, and the mixture was cooled to 25° C. over 1.3 hours and further to 20° C. over an additional 5 hours, then cooled to −3° C. The mixture, containing crystalline ezatiostat hydrochloride ansolvate, was filtered and the solids washed twice with 54 L each of chilled (−5˜0)° C. ethyl acetate. The damp solids of crystalline ezatiostat hydrochloride ansolvate, 70 Kg, were dried in a vacuum oven at 25° C. for 16 hours, 35° C. for 7 hours, then at room temperature for 1 hour, then sieved. The crystalline ezatiostat hydrochloride ansolvate, 44.2 Kg, had a loss on drying at 40° C. under vacuum for 2 hours of 0.09%, and a water content by Karl Fischer analysis of 0.09%.
……………………………………………………………
U.S. Pat. No. 5,763,570
https://www.google.com/patents/US5763570
……………………………..
http://www.google.com/patents/WO2011156025A1?cl=en
Example 1. Preparation Of Ezatiostat Hydrochloride Ansolvate By Slurrying
[0082] Ezatiostat hydrochloride monohydrate was added to methyl tert-butyl ether at room temperature in excess, so that undissolved solids were present. The mixture was then agitated in a sealed vial at room temperature for 4 days, and the solids were then isolated by suction filtration. XRPD analysis of the solids established that the isolated solids were ezatiostat hydrochloride ansolvate.
[0083] Ezatiostat hydrochloride monohydrate was added to hexanes at 60 °C in excess, so that undissolved solids were present. The mixture was then agitated in a sealed vial at 60 °C for 4 days, and the solids were then isolated by suction filtration. XRPD analysis of the solids established that the isolated solids were ezatiostat hydrochloride ansolvate.
Example 2. Preparation Of Crystalline Ezatiostat Hydrochloride Ansolvate By Heating
[0084] DSC of crystalline ezatiostat hydrochloride monohydrate showed the pattern in FIG. 1, as discussed in paragraph above. Hot stage microscopy showed an initial melt followed by a recrystallization at 153 °C and a final melt at 166 °C. VT-XRPD, where XRPD patterns were obtained at 28 °C, 90 °C, and 160 °C during heating, and 28 °C after cooling of the formerly heated material, showed the presence of ezatiostat hydrochloride monohydrate at 28 °C and 90 °C during heating and of crystalline ezatiostat hydrochloride ansolvate at 160 °C and 28 °C after cooling of the formerly heated material. This confirmed that the transition at around 153/156 °C was a conversion of ezatiostat hydrochloride monohydrate form A to crystalline ezatiostat hydrochloride ansolvate form D and that the final DSC endothermic peak at about 177 °C (166 °C in the hot stage microscopy) was due to the melting of crystalline ezatiostat hydrochloride ansolvate. This was further confirmed by XRPD of the TG-IR material, where XRPD patterns obtained at room temperature both before and after heating to about 160 °C showed that the material before heating was form A and that the material after heating was form D ansolvate. DSC of crystalline ezatiostat hydrochloride ansolvate prepared by recrystallization showed the pattern in FIG. 5, with only the endothermic peak at about
177 °C followed by a broad endotherm at about (205 – 215) °C. Accordingly, the presence of the DSC endothermic peak at about 177 °C, for example at (177±2) °C, when measured under the conditions described above, is considered characteristic of crystalline ezatiostat hydrochloride ansolvate, and the substantial absence of thermal events at temperatures below this is considered indicative of the absence of other forms of ezatiostat hydrochloride
………………………………..
http://www.google.com/patents/WO2013082462A1?cl=en
Ezatiostat, also known as TLK199 or TER 199, is a compound of the formula:
[0003] Ezatiostat has been shown to induce the differentiation of HL-60 promyelocytic leukemia cells in vitro, to potentiate the activity of cytotoxic agents both in vitro and in vivo, and to stimulate colony formation of all three lineages of hematopoietic progenitor cells in normal human peripheral blood. In preclinical testing, ezatiostat has been shown to increase white blood cell production in normal animals, as well as in animals in which white blood cells were depleted by treatment with cisplatin or fluorouracil. Similar effects may provide a new approach to treating myelodysplasia syndrome (MDS).
[0004] Many conditions, including MDS, a form of pre-leukemia in which the bone marrow produces insufficient levels of one or more of the three major blood elements (white blood cells, red blood cells, and platelets), are characterized by depleted bone marrow.
Myelosuppression, which is characterized by a reduction in blood cell levels and in a reduction of new blood cell generation in the bone marrow, is also a common, toxic effect of many standard chemotherapeutic drugs.
[0005] Ezatiostat hydrochloride is the hydrochloride acid addition salt of ezatiostat.
Ezatiostat hydrochloride in a liposomal injectable formulation was studied in a clinical trial for the treatment of MDS, and results from this trial, reported by Raza et al., J. Hem. One, 2:20 (published online 13 May 2009), demonstrated that administration of TLK199 was well tolerated and resulted in multi-lineage hematologic improvement. Ezatiostat hydrochloride in a tablet formulation has been evaluated in a clinical trial for the treatment of MDS, as reported by Raza et al, Blood, 113:6533-6540 (prepublished online 27 April 2009) and a single-patient report by Quddus et al, J. Hem. One, 3:16 (published online 23 April 2010), and is currently being evaluated in clinical trials for the treatment of MDS and for severe chronic idiopathic neutropenia.
…………………………………..
http://www.google.com/patents/WO2013082462A1?cl=en
Example 1
[0048] 80 mg of crystalline ezatiostat hydrochloride was placed in a round bottom flask and dissolved in 25 mL of methanol. The solvent was then evaporated on a rotary evaporation apparatus under reduced pressure at 30 °C. After 30 minutes, the solid sample was removed from the round bottom flask and stored in a sealed vial at 2 °C in a refrigerator. Analysis of this sample was carried out within 24 hours of removing it from the rotary evaporation apparatus.
[0049] The resulting amorphous material was analyzed by 1H NMR, 13C NMR, DSC, and X-Ray powder diffraction experiments. The DSC conditions were 30 to 300 °C at 10 °C/ min using 7 mg of the amorphous material. The X-Ray powder diffraction was taken at 0-60 of 2theta. The crystalline ezatiostat hydrochloride was also analyzed.
Phase II trials: Ezatiostat is the first GSTP1-1 inhibitor shown to cause clinically significant and sustained reduction in RBC transfusions, transfusion independence, and multilineage responses in MDS patients. The tolerability and activity profile of ezatiostat may offer a new treatment option for patients with MDS. (source: Cancer. 2012 Apr 15;118(8):2138-47.)
|
References |
1: Galili N, Tamayo P, Botvinnik OB, Mesirov JP, Brooks MR, Brown G, Raza A. Prediction of response to therapy with ezatiostat in lower risk myelodysplastic syndrome. J Hematol Oncol. 2012 May 6;5:20. doi: 10.1186/1756-8722-5-20. PubMed PMID: 22559819; PubMed Central PMCID: PMC3407785.
2: Raza A, Galili N, Mulford D, Smith SE, Brown GL, Steensma DP, Lyons RM, Boccia R, Sekeres MA, Garcia-Manero G, Mesa RA. Phase 1 dose-ranging study of ezatiostat hydrochloride in combination with lenalidomide in patients with non-deletion (5q) low to intermediate-1 risk myelodysplastic syndrome (MDS). J Hematol Oncol. 2012 Apr 30;5:18. doi: 10.1186/1756-8722-5-18. PubMed PMID: 22546242; PubMed Central PMCID: PMC3416694.
3: Lyons RM, Wilks ST, Young S, Brown GL. Oral ezatiostat HCl (Telintra®, TLK199) and idiopathic chronic neutropenia (ICN): a case report of complete response of a patient with G-CSF resistant ICN following treatment with ezatiostat, a glutathione S-transferase P1-1 (GSTP1-1) inhibitor. J Hematol Oncol. 2011 Nov 2;4:43. doi: 10.1186/1756-8722-4-43. PubMed PMID: 22047626; PubMed Central PMCID: PMC3235963.
4: Raza A, Galili N, Smith SE, Godwin J, Boccia RV, Myint H, Mahadevan D, Mulford D, Rarick M, Brown GL, Schaar D, Faderl S, Komrokji RS, List AF, Sekeres M. A phase 2 randomized multicenter study of 2 extended dosing schedules of oral ezatiostat in low to intermediate-1 risk myelodysplastic syndrome. Cancer. 2012 Apr 15;118(8):2138-47. doi: 10.1002/cncr.26469. Epub 2011 Sep 1. PubMed PMID: 21887679.
5: Quddus F, Clima J, Seedham H, Sajjad G, Galili N, Raza A. Oral Ezatiostat HCl (TLK199) and Myelodysplastic syndrome: a case report of sustained hematologic response following an abbreviated exposure. J Hematol Oncol. 2010 Apr 23;3:16. doi: 10.1186/1756-8722-3-16. PubMed PMID: 20416051; PubMed Central PMCID: PMC2873355.
6: Steensma DP. Novel therapies for myelodysplastic syndromes. Hematol Oncol Clin North Am. 2010 Apr;24(2):423-41. doi: 10.1016/j.hoc.2010.02.010. Review. PubMed PMID: 20359635.
7: D’Alò F, Greco M, Criscuolo M, Voso MT. New treatments for myelodysplastic syndromes. Mediterr J Hematol Infect Dis. 2010 Aug 11;2(2):e2010021. doi: 10.4084/MJHID.2010.021. PubMed PMID: 21415972; PubMed Central PMCID: PMC3033133.
8: Raza A, Galili N, Callander N, Ochoa L, Piro L, Emanuel P, Williams S, Burris H 3rd, Faderl S, Estrov Z, Curtin P, Larson RA, Keck JG, Jones M, Meng L, Brown GL. Phase 1-2a multicenter dose-escalation study of ezatiostat hydrochloride liposomes for injection (Telintra, TLK199), a novel glutathione analog prodrug in patients with myelodysplastic syndrome. J Hematol Oncol. 2009 May 13;2:20. doi: 10.1186/1756-8722-2-20. PubMed PMID: 19439093; PubMed Central PMCID: PMC2694211.
9: Raza A, Galili N, Smith S, Godwin J, Lancet J, Melchert M, Jones M, Keck JG, Meng L, Brown GL, List A. Phase 1 multicenter dose-escalation study of ezatiostat hydrochloride (TLK199 tablets), a novel glutathione analog prodrug, in patients with myelodysplastic syndrome. Blood. 2009 Jun 25;113(26):6533-40. doi: 10.1182/blood-2009-01-176032. Epub 2009 Apr 27. PubMed PMID: 19398716.
| WO2013082462A1 * | Nov 30, 2012 | Jun 6, 2013 | Telik, Inc. | Amorphous ezatiostat ansolvate |
| US20120251496 * | Mar 20, 2012 | Oct 4, 2012 | Telik, Inc. | Ezatiostat for treating multiple myeloma |
……….
Known Yes1 kinase inhibitors, dasatinib and saracatinib.
| Compound name and NCGC ID | Structure | Clinical phase | Known targets | Yes1 IC50 (nM) |
|---|---|---|---|---|
| Dasatinib (1) NCGC00181129 |
 |
Approved | Lyn, PDGFR, KIT, Lck, BTK, Bcr–Abl, Fyn, Yes1, c-Src |
0.5 (<1.0)a |
| Saracatinib (2) NCGC00241099 |
 |
Phase II/III | c-Src, Bcr–Abl, Yes1, Lck | 6.2 (0.70)a |
| AEE-788 (3) NCGC00263149 |
 |
Phase I/II | EGFR, HER-2, VEGFR-2 | 17.5 (13.1)a |
| Dovitinib (4) NCGC00249685 |
 |
Phase III | FGFR, EGFR, PDGFR, VEGFR-1,2 | 31 (1.4)a |
| DCC-2036 (5) NCGC00263172 |
 |
Phase I/II | Bcr–Abl, Tie-2, Lyn, FLT3, VEGFR-2 | 2.5 (1.5)a |
| SGI-1776 (6) NCGC00263186 |
 |
Discontinued | Pim-1, FLT3 | 2670 (240)a |
| AMG-Tie-2-1 (7) NCGC00263199 |
 |
Preclinical | Tie-2 | 8.7 (22.0)a |
| AZ-23 (8) NCGC00250381 |
 |
Preclinical | Trk | 39.1 (3.0)a |
| Dorsomorphin (9) NCGC00165869 |
 |
Preclinical | AMPK, BMPR, TGFβ Receptor | 195.9 (29.8)a |
| AZ-628 (10) NCGC00250380 |
 |
Preclinical | Raf Kinase B,C | 348.3 (51.2)a |
- http://www.sciencedirect.com/science/article/pii/S0960894X13006677
- Data in parentheses were gathered by Reaction Biology Corp. using a [γ-33P]-ATP radiolabeled enzyme activity assay at an ATP concentration of 10 μM (www.reactionbiology.com).
SEE COMPILATION ON SIMILAR COMPOUNDS AT …………..http://drugsynthesisint.blogspot.in/p/nostat-series.html
Pharma Trends: Global Medicine Spending to Pass $1 Trillion in 2014
http://msg-latam-sfb.blogspot.in/2013/12/pharma-trends-global-medicine-spending.html
Very First Human Trials Using Cannbis To Treat Brain Cancer Are Under Way

The picture to your left is showing immunofluorescence of the human glioma cell line. (View more pictures here)
A European based pharmaceutical company called GW Pharmaceuticals is set to commence its first phase of clinical trials for the treatment of Glioblastoma Multiforme (GBM). It’s a bio-pharmaceutical company focused on discovering, developing and commercializing novel therapeutics from its proprietary cannabinoid product platform.
According to the New England Journal of Medicine, GBM accounts for approximately 50% of the 22,500 new cases of brain cancer diagnosed in the United States alone each year.(1) Treatment with regards to brain cancer are very limited which makes the study of cannabis and its effect on brain tumors crucial.
La cantera de los biológicos
Los medicamentos biotecnológicos están en plena ebullición, de forma continuada aparecen nuevas moléculas. Muchas de ellas se encuentran en fases preliminares de desarrollo y se espera que en los próximos años se vayan aprobando. ¿Se mantendrá este ritmo en el tiempo?
View original post 320 more words
Amgen-AstraZeneca Psoriasis Drug Brodalumab (AMG 827) Hits Phase 3 Endpoints

AstraZeneca and Amgen announced that the Phase 3 AMAGINE-1TM study evaluating brodalumab in patients with moderate-to-severe plaque psoriasis met all primary and secondary endpoints for both evaluated doses.
Brodalumab is a human monoclonal antibody designed for the treatment of inflammatory diseases.[1] It is being tested for the treatment of moderate to severe psoriasis[2] in Phase III clinical trials as of November 2013.[3][4]
Brodalumab was developed by Amgen, Inc.

Mechanism of action
Brodalumab binds to the interleukin-17 receptor and so prevents interleukin 17 (IL-17) from activating the receptor. This mechanism is similar to that of another anti-psoriasis antibody, ixekizumab, which however binds to IL-17 itself.[2]
At present, brodalumab is the only experimental drug in development that inhibits the IL-17 receptor, thus inhibiting several of the IL-17 ligands at once from transmitting signals to the body. Other agents currently in development seek to target the individual IL-17 ligands. By inhibiting the attachment of these ligands with the receptor, brodalumab stops the body from receiving signals that may otherwise cause inflammation and other ailments.
Researchers are currently investigating brodalumab for the treatment of psoriasis (Phase II and planned Phase III), asthma (Phase II), and psoriatic arthritis (Phase II).
Psoriasis is a chronic disease of the immune system that causes the skin cells to grow at a faster rate. Worldwide, the condition affects around 125 million individuals. Even though several types of psoriasis exist, around 80% of sufferers have plaque psoriasis. Plaque psoriasis can cause painful and itchy red, scaly patches to appear on the skin.
| Monoclonal antibody | |
|---|---|
| Type | Whole antibody |
| Source | Human |
| Target | Interleukin 17 receptor A |
| Clinical data | |
| Legal status | Investigational |
| Identifiers | |
| CAS number | 1174395-19-7 |
| ATC code | None |
| KEGG | D10061 |
| Chemical data | |
| Formula | C6372H9840N1712O1988S52 |
| Mol. mass | 144.06 kDa |
About Brodalumab (AMG 827)
Brodalumab is a novel human monoclonal antibody that binds to the interleukin-17 (IL-17) receptor and inhibits inflammatory signaling by blocking the binding of several IL-17 ligands to the receptor. By stopping IL-17 ligands from activating the receptor, brodalumab prevents the body from receiving signals that may lead to inflammation. The IL-17 pathway plays a central role in inducing and promoting inflammatory disease processes. In addition to moderate-to-severe plaque psoriasis (Phase 3), brodalumab is currently being investigated for the treatment of psoriatic arthritis (Phase 3) and asthma (Phase 2).
About the Amgen and AstraZeneca Collaboration
In April 2012, Amgen and AstraZeneca formed a collaboration to jointly develop and commercialize five monoclonal antibodies from Amgen’s clinical inflammation portfolio. With oversight from joint governing bodies, Amgen leads clinical development and commercialization for brodalumab (Phase 3 for moderate-to-severe plaque psoriasis and psoriatic arthritis, Phase 2 for asthma) and AMG 557/MEDI5872 (Phase 1b for autoimmune diseases such as systemic lupus erythematosus). AstraZeneca, through its biologics arm MedImmune, leads clinical development and commercialization for MEDI7183/AMG 181 (Phase 2 for ulcerative colitis and Crohn’s disease), MEDI2070/AMG 139 (Phase 2 for Crohn’s disease) and MEDI9929/AMG 157 (Phase 2 for asthma).
About Amgen
Amgen is committed to unlocking the potential of biology for patients suffering from serious illnesses by discovering, developing, manufacturing and delivering innovative human therapeutics. This approach begins by using tools like advanced human genetics to unravel the complexities of disease and understand the fundamentals of human biology.
Amgen focuses on areas of high unmet medical need and leverages its biologics manufacturing expertise to strive for solutions that improve health outcomes and dramatically improve people’s lives. A biotechnology pioneer since 1980, Amgen has grown to be the world’s largest independent biotechnology company, has reached millions of patients around the world and is developing a pipeline of medicines with breakaway potential.
For more information, visit www.amgen.com and follow us on www.twitter.com/amgen.

About AstraZeneca
AstraZeneca is a global, innovation-driven biopharmaceutical business that focuses on the discovery, development and commercialisation of prescription medicines, primarily for the treatment of cardiovascular, metabolic, respiratory, inflammation, autoimmune, oncology, infection and neuroscience diseases. AstraZeneca operates in over 100 countries and its innovative medicines are used by millions of patients worldwide. For more information please visit: www.astrazeneca.com.
References
- “Statement On A Nonproprietary Name Adopted By The USAN Council: Brodalumab”. American Medical Association.
- “Neue Antikörper in der Pipeline”. Pharmazeutische Zeitung (in German) (12). 2012.
- ClinicalTrials.gov NCT01708590 Study of Efficacy, Safety, and Withdrawal and Retreatment With Brodalumab in Moderate to Severe Plaque Psoriasis Subjects (AMAGINE-1)
- ClinicalTrials.gov NCT01708629 Study of Efficacy and Safety of Brodalumab Compared With Placebo and Ustekinumab in Moderate to Severe Plaque Psoriasis Subjects (AMAGINE-3)
http://ksclinic.exblog.jp/18270693/
学術面で最初の講演は、米国のJames Krueger教授による「Th1細胞,Th17細胞,Th22細胞が複雑なサイトカインネットワークによって、細胞レベル、分子レベルで乾癬を引き起こす」でした。その要約を示すスライドを幾枚か失敬します(Krueger先生、ごめんなさい)。
 乾 癬の原因究明、病態(病気の起こり方)解明の主役となった免疫学的研究の最先端を行くKrueger先生の、最新情報がコンパクトにまとまった素晴らしい 講演でした。生物学的製剤の治療根拠となるサイトカインネットワークは、現在TipDC – Th17経路によって、きわめて明快に説明されるようになり、Th17細胞が放出するIL17が表皮細胞(ケラチノサイト)の乾癬化を起こします。現在使 用されている抗TNFα製剤、抗IL12/23製剤が、より上流(免疫反応の根っこ)で免疫反応を抑制するのに比べ、IL17はより末梢における乾癬の原 因サイトカインであることから、IL17の抑制は、より乾癬をピンポイントで、そして副作用もミニマムにすることが期待される。
乾 癬の原因究明、病態(病気の起こり方)解明の主役となった免疫学的研究の最先端を行くKrueger先生の、最新情報がコンパクトにまとまった素晴らしい 講演でした。生物学的製剤の治療根拠となるサイトカインネットワークは、現在TipDC – Th17経路によって、きわめて明快に説明されるようになり、Th17細胞が放出するIL17が表皮細胞(ケラチノサイト)の乾癬化を起こします。現在使 用されている抗TNFα製剤、抗IL12/23製剤が、より上流(免疫反応の根っこ)で免疫反応を抑制するのに比べ、IL17はより末梢における乾癬の原 因サイトカインであることから、IL17の抑制は、より乾癬をピンポイントで、そして副作用もミニマムにすることが期待される。
 現在、3種類のIL17抑制薬剤が開発され、治療研究が進められている。
現在、3種類のIL17抑制薬剤が開発され、治療研究が進められている。
①IL17A抗体(Secukinumab Novartis社)
②IL17A抗体(Ixekizumab Lilly社)
③IL17A受容体抗体(Brodalumab Amgen社)
その一つ、Secukinumabの効果(PASI75)=すごく乾癬がよくなる)では、たった3回の注射で90%以上の患者がPASI75を達成する。
PASI90(=乾癬がほとんどなくなる)でみても、60%の患者で達成されている。

Secukinumabの臨床効果。上の段は「プラセボ(偽薬)」、下の段がSecukinumab。

Ixekizumabの効果(PASI90)。約80%の患者で達成されている。驚異的である。

Brodalumabの臨床効果
印象深かった講演をもう一つ、詳細に紹介いたします。
米 国のAnne Bowcock教授の”The genetics of psoriasis: Old risks, novel loci (乾癬の遺伝子研究:昔から言われていた異常、新しく見つかった場所)です。Bowcock教授は、乾癬の原因遺伝子について世界で最初に報告した研究者 です。ここでも少し講演スライドを拝借(Bowcock先生、ごめんなさい)。
 Bowcock 教授は1999年、乾癬家系の詳細な遺伝子調査から第17染色体に乾癬と関わり深い遺伝子異常があることをみつけ、科学雑誌Scienceに報告した。 21世紀を迎える直前のことであり、遠からず乾癬の原因遺伝子が確定し、完治治療を開発することも夢ではないと、当時期待したものでした。
Bowcock 教授は1999年、乾癬家系の詳細な遺伝子調査から第17染色体に乾癬と関わり深い遺伝子異常があることをみつけ、科学雑誌Scienceに報告した。 21世紀を迎える直前のことであり、遠からず乾癬の原因遺伝子が確定し、完治治療を開発することも夢ではないと、当時期待したものでした。
ところが、次々と関連遺伝子はみつけられるものの(現在は30種類以上)、肝心の原因遺伝子、特定のタンパク、メカニズムは不明のままでした。
 Bowcock 教授の息の長い研究は、第17染色体上にあるCARD14と呼ばれるタンパクの、その異常が直接乾癬を起こすことを説き明かしました。CARD14は細胞 膜上にあるタンパクで、細胞外で起こる炎症から生じる様々な刺激物質を、細胞の膜から細胞の中へ伝える役割を果たしています。その伝達経路はNFκBを介 しています(乾癬ではこの経路が活発に動いていることが、高知大学の佐野教授により解明されました)。
Bowcock 教授の息の長い研究は、第17染色体上にあるCARD14と呼ばれるタンパクの、その異常が直接乾癬を起こすことを説き明かしました。CARD14は細胞 膜上にあるタンパクで、細胞外で起こる炎症から生じる様々な刺激物質を、細胞の膜から細胞の中へ伝える役割を果たしています。その伝達経路はNFκBを介 しています(乾癬ではこの経路が活発に動いていることが、高知大学の佐野教授により解明されました)。

遺伝性膿疱性乾癬患者では、このCARD14遺伝子に点突然変異が起こっていることを発見しました。この点突然変異だけで、特殊タイプではありますが、乾癬の原因が特定されたのです。

点突然変異だけではなく、CARD14遺伝子に起こりやすい変異も、ほかの遺伝子異常(PSORS1、MHC遺伝子)、あるいは環境変化が加わると乾癬を引き起こすことも証明しました。
大変感銘深い講演でした。
会議の模様、IFPA代表者会議の報告は、また後日掲載いたします(『2012年9月教室抄録』をご覧ください)。
ブログ「PHOTO & ESSAY」もご覧ください。
For National Women’s Health Week, FDA Resources Help Women Make Informed Health Choices

For National Women’s Health Week, FDA Resources Help Women Make Informed Health Choices
By: Marsha B. Henderson, M.C.R.P. “Ask your mother.” In households throughout the country, women often make decisions about foods and medical products for themselves and their loved ones. As we celebrate National Women’s Health Week (May 11-17), I want to … Continue reading →http://blogs.fda.gov/fdavoice/index.php/2014/05/for-national-womens-health-week-fda-resources-help-women-make-informed-health-choices/?source=govdelivery&utm_medium=email&utm_source=govdelivery
New method sneaks drugs into cancer cells before triggering release

Biomedical engineering researchers have developed an anti-cancer drug delivery method that essentially smuggles the drug into a cancer cell before triggering its release. Credit: Ran Mo
Biomedical engineering…
View original post 456 more words
New technique uses ATP as trigger for targeted anti-cancer drug delivery

Biomedical engineering researchers from North Carolina State University and the University of North Carolina have developed a new technique that uses adenosine-5′-triphosphate (the so-called ‘energy molecule’) to trigger the release of anti-cancer drugs directly into cancer cells. The spherical nanoparticles are coated with a shell that incorporates hyaluronic acid, which interacts with proteins found on the surface of some cancer cells. The nanoparticle is filled with DNA molecules that release anti-cancer drug doxorubicin when they come into contact with the adenosine-5′-triphosphate inside a cancer cell. Credit: Ran Mo
Biomedical engineering researchers have developed a new technique that uses adenosine-5′-triphosphate (ATP), the so-called “energy molecule,” to trigger the release of anti-cancer drugs directly into cancer cells. Early laboratory tests show it increases the effectiveness of drugs targeting breast cancer. The technique was developed by researchers at North Carolina State University and the University of North Carolina at Chapel Hill.
“This is…
View original post 339 more words
Regadenoson……..Adenosine A2a receptor agonist, for Coronary artery disease; Sickle cell anemia

2-{4-[(methylamino)carbonyl]- 1H-pyrazol-1-yl}adenosine
(1-{9-[(2R,3R,4S,5R)-3,4-dihydroxy-5-(hydroxymethyl)oxolan-2-yl]-6-aminopurin-2-yl}pyrazol-4-yl)-N-methylcarboxamide.
Regadenoson (INN, code named CVT-3146) is an A2A adenosine receptor agonist that is a coronary vasodilator. It produces hyperemia quickly and maintains it for a duration that is useful for radionuclide myocardial perfusion imaging.[1]
It was approved by the United States Food and Drug Administration on April 10, 2008 and is marketed by Astellas Pharma under the tradename Lexiscan.[2] It is approved for use in the European Union and under the name of Rapiscan. It is currently being marketed by GE Healthcare and is being sold in both the United Kingdom and Germany.
Regadenoson has a 2- to 3-minute biological half-life, as compared with adenosine‘s 30-second half-life. Regadenoson stress protocols using a single bolus have been developed, obviating the need for an intravenous line. Regadenoson stress tests are not affected by the presence of beta blockers, as regadenoson vasodilates but does not stimulate beta adrenergic receptors.

Regadenoson is an A2A adenosine receptor agonist that is a coronary vasodilator [see CLINICAL PHARMACOLOGY]. Regadenoson is chemically described as adenosine, 2-[4-[(methylamino)carbonyl]-1H-pyrazol-1-yl]-, monohydrate. Its structural formula is:
 |
The molecular formula for regadenoson is C15H18N8O5 • H2O and its molecular weight is 408.37. Lexiscan is a sterile, nonpyrogenic solution for intravenous injection. The solution is clear and colorless. Each 1 mL in the 5 mL pre-filled syringe contains 0.084 mg of regadenoson monohydrate, corresponding to 0.08 mg regadenoson on an anhydrous basis, 10.9 mg dibasic sodium phosphate dihydrate or 8.7 mg dibasic sodium phosphate anhydrous, 5.4 mg monobasic sodium phosphate monohydrate, 150 mg propylene glycol, 1 mg edetate disodium dihydrate, and Water for Injection, with pH between 6.3 and 7.7.
Regadenoson is also referred to in the literature as CVT- 3146 or (1-{9-[(4S,2R,3R,5R)-3,4-dihydroxy-5-(hydroxymethyl)oxolan-2-yl]-6- aminopurin-2-yl}pyrazol-4-yl)-N-methylcarboxamide and has the formula:
Methods for synthesizing regadenoson and related compounds are set forth in U.S. Patent No. 6,403,567, the specification of which is incorporated herein by reference in its entirety.
Regadenoson may be administered by pharmaceutical administration methods that are known in the art. It is preferred that regadenoson is dosed i.v. It is more preferred that regadenoson is administered in a single dose i.v. The term “single dose” refers generally to a single quickly administered dose of a therapeutic amount of regadenoson. The term “single dose” does not encompass a dose or doses administered over an extended period of time by, for example continuous i.v. infusion.
Regadenoson will typically be incorporated into a pharmaceutical composition prior to use. The term “pharmaceutical composition” refers to the combination of regadenoson with at least one liquid carrier that together form a solution or a suspension. Lyophilized powders including compositions of this invention fall within the scope of “pharmaceutical compositions” so long as the powders are intended to be reconstituted by the addition of a suitable liquid carrier prior to use. Examples of suitable liquid carriers include, but are not limited to water, distilled water, de-ionized water, saline, buffer solutions, normal isotonic saline solution, dextrose in water, and combinations thereof.
Regadenoson [(l-{9-[(4S, 2R, 3R, 5R)-3,4-dihydroxy-5-(hydroxymethyl)oxalan-2-yl]-6- aminopurin-2-yl}pyrazol-4-yl)-N-methylcarboxamine] is a selective A2A-adenosine receptor agonist that is a coronary vasodilator. It is currently marketed in the form of a monohydrate as a pharmacologic stress agent indicated for radionuclide myocardial perfusion imaging (MPI) in patients unable to undergo adequate exercise stress.
U.S. Patent No. 8,106,183 describes amorphous regadenoson, and three forms of regadenoson, referred to as Form A (a monohydrate), Form B and Form C.
The synthesis of regadenoson is described, for example, in U.S. Patent Nos. 6,403,567 and 7,183,264. The syntheses disclosed are multi-step processes that proceed via 2- hydrazinoadenosine, which is prepared from the corresponding iodo-derivative (2- iodoadenosine).


……………………………
http://www.google.com/patents/WO2012149196A1?cl=en
EXAMPLE 1
Synthesis of N-Methyl-4-carboxamide
20 g (143 mmol, 1 equiv) of ethyl pyrazole-4-carboxylate and 200 mL (2310 mmol, 16.2 equiv) of a 40 % aqueous solution of methylamine were added to a three-necked flask equipped with a condenser and a heating mantle. The mixture was stirred to aid dissolution, and heated to 65 °C for 2 hours. The reaction was monitored using HPLC at 220 nm with a C18 column. The reaction mixture was then concentrated in vacuo to obtain a syrup / solid. The crude product was co-evaporated with acetonitrile (3 x 200 mL). 100 mL of acetonitrile was then added to the solids and the mixture was stirred for several hours until the solids were well suspended. The solids were then isolated by filtration, washed with 100 mL acetonitrile, and dried in an oven at 40°C to afford 14.4 g (80 % yield) of N-methyl-4-carboxamide with a purity of 93.5% by HPLC.
EXAMPLE 2
Synthesis of IDAAR-Cu+2
This preparation has reported in the literature. See, e.g., Chinese Chemical Letters, (21(1), 51-54, 2010.
An Erlenmeyer flask was charged with 350 mL of water and 75 g of Chelex 100 resin. With stirring, an aqueous solution of copper sulfate pentahydrate (59 g in 350 mL of water) was slowly added over a period of 15 minutes. The resulting slurry was stirred for 2 hours, then filtered. The resulting solids were washed with 100 – 200 mL of water and dried in a vacuum oven at 50 °C for 16 hours to afford 18 g of IDAAR-Cu+2. The copper content of the product was determined to be 11 wt % using Inductively Coupled Plasma Optical Emission Spectroscopy (ICP-OES).
EXAMPLE 3
Synthesis of Regadenoson Monohydrate
5 g (17.5 mmol, 1 equiv) of 2-fluoroadenosine, 3.07g (24.5 mmol, 1.4 equiv) of N- methylpyrazole-4-carboxamide, and 32 mL of dimethylsulfoxide were added under a nitrogen atmosphere to a dry 3-necked reaction flask equipped with a condenser and a heating mantle.. The mixture was stirred to afford a solution. 100 mL of acetonitrile was then added followed by the addition of 2.2 g of IDAAR-Cu2+ and 5.34 g (5.24 mL, 35.1 mmol, 2 equiv) of
diazabicycloundecene (DBU). The reaction mixture was heated to 70 – 80 °C overnight and monitored by HPLC at 260 nm with a C18 column until the reaction was complete. Then, the reaction mixture was evaporated in vacuo to remove most of the acetonitrile. The remaining dimethylsufoxide solution was purified by reverse phase chromatography using methanol and water. The product was dried in vacuo at a temperature that did not exceed 40° C to afford 3 g (44% yield) of regadenoson monohydrate.
EXAMPLE 4
Synthesis of 2-Hydazineadenosine
2-fluoroadenosine (4g, 14 mmol) was dissolved in 100 mL ethanol in a 300 mL three- necked flask. Hydrazine hydrate (4.1 mL, 6 equivalents, 84 mmol) was added and the mixture was heated to reflux for 1 hour. The reaction mixture was allowed to cool to room temperature and stirred overnight (16 hours). The resulting white precipitate was isolated by filtration and dried in oven at 40°C overnight to afford 2-hydrazinoadenosine (yield: 94%, 3.5g, 96% purity).
EXAMPLE 5
Synthesis of Regadenoson Form D
2-Fluoroadenosine (45 g, 0.158 moL, 1 eq.), 4-(N-methylcarboxamido)pyrazole (27.64 g, 0.221 moL, 1.4 eq.), dimethylsulfoxide (DMSO) (320 mL) and acetonitrile (960 mL) were added to a dry 3000 ml 3-neck reaction flask equipped with a condenser and heating mantle. After stirring for 10 minutes, IDAAR-Cu (20.07 g, 0.032 moL, 0.2 eq.) and DBU (48.0 g, 0.316 moL, 2 eq.) were added. The resulting mixture was then heated to 65°C overnight (18 hours).
The reaction mixture was then filtered and the filtrate was evaporated followed by 2 x 500 mL co-evaporation with xylene. The residue was diluted with 5 L acetonitrile, transferred to a 10 L flask and kept in a cold room (4°C) overnight. The resulting white precipitate was isolated by filtration and stirred in 1.8 L of water. The mixture was heated to 80° C for 2 hours, then allowed to cool in a cold room (4°C) overnight.
The white precipitate was isolated by filtration, then dissolved in 200 ml of 1 : 1 mixture of DMSO and methanol. The clear and slightly yellow solution was loaded to a reverse phase column (10 L) and eluted with water/methanol (gradient with a 5% increase of MeOH every 10 L).
The fractions with HPLC purity of more than 99.9% were combined and concentrated to a paste. The supernatant liquid was decanted and the flask heated in an oil-bath at 150° C under reduced pressure of 20mmHg for 6 hours to afford 6.2 g of Regadenoson Form D as white solid (99.94% HPLC, KF analysis 0.8%).
The fractions with HPLC purity between 50 and 99.8% (~ 23g of product as indicated by HPLC) were combined and subjected to a second purification stage.
………………………………
WO 0078779
https://www.google.com/patents/WO2000078779A2?cl=en
Example 5
(l-{9-[(4S,2R,3R,5R)-3,4-dihydroxy-5-(hydroxymethyl)oxolan-2-yl]-6-aminopurin-2- yl}pyrazol-4-yl)-N-methylcarboxamide (16)
Compound 12 (0.05 mg, 0.12 mmol) was added to 4 mL methylamine (40% sol. In water). The mixture heated at 65 °C in for 24 h. After concentration in vacuo, the residue was purified using prep. TLC (10% MeOH:DCM). ‘HNMR (CD3OD) 62.90 (s, 3 H), 3.78 (m, 1
H), 3.91 (m, 1 H), 4.13 (d, 1 H), 4.34 (d, 1 H), 4.64 (m, 1 H), 6.06 (d, 1 H), 8.11 (s, 1 H), 8.38
(s, 1 H), 9.05 (s, 1 H).
…………………..
https://www.google.com/patents/US6403567
U.S. Patent Nos. 6,403,567
Scheme 1.
Compound I can be prepared by reacting compound 1 with appropriately substituted 1,3 -dicarbonyl in a mixture of AcOH and MeOH at 80° C. (Holzer et al., J. Heterocycl. Chem. (1993) 30, 865). Compound II, which can be obtained by reacting compound I with 2,2-dimethoxypropane in the presence of an acid, can be oxidized to the carboxylic acid III, based on structurally similar compounds using potassium permanganate or pyridinium chlorochromate (M. Hudlicky, (1990) Oxidations in Organic Chemistry, ACS Monographs, American Chemical Society, Washington D.C.). Reaction of a primary or secondary amine having the formula HNR6R7, and compound III using DCC (M. Fujino et al., Chem. Pharm. Bull. (1974), 22, 1857), PyBOP (J. Martinez et al., J. Med. Chem. (1988) 28, 1874) or PyBrop (J. Caste et al. Tetrahedron, (1991), 32, 1967) coupling conditions can afford compound IV.
Compound V can be prepared as shown in Scheme 2. The Tri TBDMS derivative 4 can be obtained by treating compound 2 with TBDMSCl and imidazole in DMF followed by hydrolysis of the ethyl ester using NaOH. Reaction of a primary or secondary amine with the formula HNR6R7, and compound 4 using DCC (M. Fujino et al., Chem. Pharm. Bull. (1974), 22, 1857), PyBOP (J. Martinez et al., J. Med. Chem. (1988) 28, 1874) or PyBrop (J. Caste et al. Tetrahedron, (1991), 32, 1967) coupling conditions can afford compound V.
A specific synthesis of compound 11 is illustrated in Scheme 3. Commercially available guanosine 5 was converted to the triacetate 6 as previously described (M. J. Robins and B. Uznanski, Can. J. Chem. (1981), 59, 2601-2607). Compound 7, prepared by following the literature procedure of Cerster et al. (J. F. Cerster, A. F. Lewis, and R. K. Robins, Org. Synthesis, 242-243), was converted to compound 9 in two steps as previously described (V. Nair et al., J. Org. Chem., (1988), 53, 3051-3057). Compound 1 was obtained by reacting hydrazine hydrate with compound 9 in ethanol at 80° C. Condensation of compound 1 with ethoxycarbonylmalondialdehyde in a mixture of AcOH and MeOH at 80° C. produced compound 10. Heating compound 10 in excess methylamine afforded compound 11.
The synthesis of 1,3-dialdehyde VII is described in Scheme 4. Reaction of 3,3-diethoxypropionate or 3,3-diethoxypropionitrile or 1,1-diethoxy-2-nitroethane VI (R3=CO2R, CN or NO2) with ethyl or methyl formate in the presence of NaH can afford the dialdehyde VII (Y. Yamamoto et al., J. Org. Chem. (1989) 54, 4734).
EXAMPLE 5
(1-{9-[(4S,2R,3R,5R)-3,4-dihydroxy-5-(hydroxymethyl)oxolan-2-yl]-6-aminopurin-2 -yl}pyrazol-4N-methylcarboxamide which can also be identified as 2-(4-methylaminocarbonylpyrazol-1-yl)adenosine (16)
The mixture heated at 65° C. in for 24 h. After concentration in vacuo, the residue was purified using prep. TLC (10% MeOH:DCM). 1HNMR (CD3OD) δ2.90 (s, 3 H), 3.78 (m, 1 H), 3.91 (m, 1 H), 4.13 (d, 1 H), 4.34 (d, 1 H), 4.64 (m, 1 H), 6.06 (d, 1 H), 8.11 (s, 1 H), 8.38 (s, 1 H), 9.05 (s, 1 H).
………………………….
US 7,183,264
http://www.google.com/patents/US7183264
EXAMPLE 5
(1-{9-[(4S,2R,3R,5R)-3,4-dihydroxy-5-(hydroxymethyl)oxolan-2-yl]-6-aminopurin-2-yl}pyrazol-4-yl)-N-methylcarboxamide (16)
Compound 12 (0.05 mg, 0.12 mmol) was added to 4 mL methylamine (40% sol. In water). The mixture heated at 65° C. in for 24 h. After concentration in vacuo, the residue was purified using prep. TLC (10% MeOH:DCM). 1HNMR (CD3OD) δ2.90 (s, 3 H), 3.78 (m, 1 H), 3.91 (m, 1 H), 4.13 (d, 1 H), 4.34 (d, 1 H), 4.64 (m, 1 H), 6.06 (d, 1 H), 8.11 (s, 1 H), 8.38 (s, 1 H), 9.05 (s, 1 H).
References
- Cerqueira MD (July 2004). “The future of pharmacologic stress: selective A2A adenosine receptor agonists”. Am. J. Cardiol. 94 (2A): 33D–40D; discussion 40D–42D. doi:10.1016/j.amjcard.2004.04.017. PMID 15261132.
- CV Therapeutics and Astellas Announce FDA Approval for Lexiscan(TM)
|
12-28-2007
|
Use of A2A Adenosine Receptor Agonists in the Treatment of Ischemia
|
|
|
2-28-2007
|
N-pyrazole A2A receptor agonists
|
|
|
1-10-2007
|
Polymer coating for medical devices
|
|
|
5-5-2006
|
Polymer coating for medical devices
|
|
1-32-2012
|
USE OF A2A ADENOSINE RECEPTOR AGONISTS
|
|
|
1-32-2012
|
PROCESS FOR PREPARING AN A2A-ADENOSINE RECEPTOR AGONIST AND ITS POLYMORPHS
|
|
|
10-21-2011
|
PROCESS FOR PREPARING AN A2A-ADENOSINE RECEPTOR AGONIST AND ITS POLYMORPHS
|
|
|
6-8-2011
|
PROCESS FOR PREPARING AN A2A-ADENOSINE RECEPTOR AGONIST AND ITS POLYMORPHS
|
|
|
6-9-2010
|
Process for preparing an A2A-adenosine receptor agonist and its polymorphs
|
|
|
3-3-2010
|
PROCESS FOR PREPARING AN A2A-ADENOSINE RECEPTOR AGONIST AND ITS POLYMORPHS
|
|
|
2-3-2010
|
Use of A2A adenosine receptor agonists
|
|
|
9-3-2008
|
Polymer coating for medical devices
|
|
|
7-4-2008
|
POLYMER COATING FOR MEDICAL DEVICES
|
|
|
4-30-2008
|
Polymer coating for medical devices
|
| US6403567 | Jun 22, 1999 | Jun 11, 2002 | Cv Therapeutics, Inc. | To stimulate mammalian coronary vasodilatation and for imaging the heart; regadenoson |
| US7183264 | Aug 29, 2003 | Feb 27, 2007 | Cv Therapeutics, Inc. | Such as ethyl-1-(9-((4S,2R,3R,5R)-3,4-dihydroxy-5-(hydroxy-methyl)oxolan-2-yl)-6 -aminopurin-2-yl)pyrazole-4-carboxylate; adenosine receptors (A2A); for stimulating mammalian coronary vasodilatation for therapy and imaging the heart |
| US7732595 | Feb 2, 2007 | Jun 8, 2010 | Gilead Palo Alto, Inc. | Process for preparing an A2A-adenosine receptor agonist and its polymorphs |
| US8106183 | Apr 22, 2010 | Jan 31, 2012 | Gilead Sciences, Inc. | Process for preparing an A2A-adenosine receptor agonist and its polymorphs |
NEW PATENT
Novel process for the preparation of (1-{9-[(4S,2R,3R,5R)-3,4-dihydroxy-5-(hydroxymethyl)oxolan-2-yl)-6-aminopurin-2-yl}pyrazole-4-yl)-N-methylcarboxamide
Biophore India Pharmaceuticals Pvt Ltd

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO





































