Higher-Order Structure Comparability: Case Studies of Biosimilar Monoclonal Antibodies
Figure 1a: Diagram of the antibody array enzyme-linked immunosorbent assay (ELISA)
Figure 1b: ELISA format for the antibody array technology
Great successes for monoclonal antibody (MAb)–based biologics over the past decade have provided many valuable options for patients combating some of the most serious diseases in the world, including cancer and autoimmune diseases. MAbs and antibody–drug conjugates (ADCs) are among the fastest growing biologic segments in development, with hundreds of candidates currently under clinical study.
read at
http://www.bioprocessintl.com/manufacturing/biosimilars/higher-order-structure-comparability/
Macrocycles in new drug discovery

Summary | Full Text | PDF (3354 KB) | PDF Plus (3440 KB) | Add to Favorites | Related
http://www.future-science.com/doi/full/10.4155/fmc.12.93?src=recsys
The use of drug-like macrocycles is emerging as an exciting area of medicinal chemistry, with several recent examples highlighting the favorable changes in biological and physicochemical properties that macrocyclization can afford. Natural product macrocycles and their synthetic derivatives have long been clinically useful and attention is now being focused on the wider use of macrocyclic scaffolds in medicinal chemistry in the search for new drugs for increasingly challenging targets. With the increasing awareness of concepts of drug-likeness and the dangers of ‘molecular obesity’, functionalized macrocyclic scaffolds could provide a way to generate ligand-efficient molecules with enhanced properties. In this review we will separately discuss the effects of macrocyclization upon potency, selectivity and physicochemical properties, concentrating on recent case histories in oncology drug discovery. Additionally, we will highlight selected advances in the synthesis of macrocycles and provide an outlook on the future use of macrocyclic scaffolds in medicinal chemistry.
Drug discovery: a view through the looking glass

Steven Lenhert, Franklin G Vellanti
Citation | Full Text | PDF (1063 KB) | PDF Plus (1071 KB) |
http://www.future-science.com/doi/full/10.4155/fmc.12.125?src=recsys
Drug discovery: Past and present

The field of biotechnology has revolutionized the drug discovery process. Recombinant DNA-driven drug discovery process is beginning to add new avenues for some old drugs. In its infancy, genetic engineering was considered useful only for the production of therapeutic proteins. Insulin, for example, previously prepared by isolation of pancreatic tissue of bovine or porcine species, can now be prepared identical to human insulin by biotechnology. Companies like Genentech and Biogen were founded solely with this objective. However, proteins do not make ideal drugs, being difficult to administer, rapidly cleared, and potentially immunogenic. Despite these disadvantages, a rapidly increasing number of “biopharmaceuticals” including recombinant proteins, therapeutic monoclonal antibodies, and even antisense oligonucleosides have been approved for indications ranging from metastatic breast cancer (Herceptin) to rheumatoid arthritis (Remicade, Enbrel).
READ AT
| Giridhar R. Drug discovery: Past and present. J Adv Pharm Technol Res 2012;3:2 |
| Giridhar R. Drug discovery: Past and present. J Adv Pharm Technol Res [serial online] 2012 [cited 2014 Aug 15];3:2. Available from: http://www.japtr.org/text.asp?2012/3/1/2/93554 |
HALOPERIDOL

Haloperidol /hæloʊpɛridɒl/ (INN, BAN, USAN, AAN; most common brand names: Haldol, Serenace) is an antipsychotic medication used in the treatment of schizophrenia, acute psychosis, mania, delirium, tics in Tourette syndrome, choreas, nausea and vomiting inpalliative care, intractable hiccups, agitation and severe anxiety.[3][4][5] Haloperidol is a butyrophenone derivative and functions as aninverse agonist of dopamine. It is classified as a typical antipsychotic and has pharmacological effects similar to the phenothiazines.[4]
A long-acting decanoate ester of haloperidol is used as an injection given every four weeks to people with schizophrenia or related illnesses who have poor adherence to medication regimens (most commonly due to them forgetting to take their medication, or due to poor insight into their illness) and suffer frequent relapses of illness, or to overcome the drawbacks inherent to its orally administered counterpart.[6] Such long acting injections are controversial because it can be seen as denying people their right to stop taking their medication.
It is on the World Health Organization’s List of Essential Medicines, a list of the most important medication needed in a basic health system.[7]
Skeletal formula of haloperidol decanoate: The decanoate group is highlighted in blue.
Brief background information
| Salt | ATC | Formula | MM | CAS |
|---|---|---|---|---|
| – | N05AD01 | C 21 H 23 ClFNO 2 | 375.87 g / mol | 52-86-8 |
Application
-
neuroleptic
-
antidiskinetik
-
antipsychotic
-
dopamine antagonists
Classes of substances
-
Chloro alcohols
-
p-Ftorbutirofenony 4-piperidinyl derivatives
-
Piperidinol
-
-
Synthesis pathway
Trade Names
| Country | Trade name | Manufacturer |
|---|---|---|
| Germany | Haldol-Janssen | Janssen-Cilag |
| various generic drugs | ||
| France | Haldol | Janssen-Cilag |
| United Kingdom | – “- | – “- |
| Serenak | Ivax | |
| Italy | Haldol | Janssen-Cilag |
| Serenas | Lusofarmaco | |
| Japan | Galomont | Janssen – Dainippon Sumitomo |
| Neoperidol | Janssen | |
| Serenak | Dainippon Sumitomo | |
| USA | various generic drugs | |
| Ukraine | Haloperidol | Ltd. “Gedeon Richter”, Hungary |
| various generic drugs | ||
Formulations
-
ampoules of 5 mg / 1 ml, 100 mg / ml, 50 mg / ml;
-
drops of 2 mg to 20 mg / ml, 2 mg / ml, 0.5 mg / ml;
-
garnuly 1%;
-
Powder 1%;
-
0.2% solution, 10 mg;
-
oral solution 2 mg / ml, 10 mg / ml;
-
Tablets of 0.75 mg, 1 mg, 1.5 mg, 2 mg, 3 mg, 5 mg, 10 mg, 20 mg
Links
-
Janssen, PAJ et al .: J. Med. Pharm. Chem. (JMPCAS) 1, 281 (1959).
-
DE 1289845 (Janssen; appl. 18/4/1959; GB -prior. 4.22.1958).
-
US 3,438,991 (Janssen; 4.15.1969; GB -prior. 18.11.1959).
1H NMR

13 C NMR

IR


MASS


http://www.scielo.br/scielo.php?script=sci_arttext&pid=S0100-40422012000200028
| Systematic (IUPAC) name | |
|---|---|
| 4-[4-(4-Chlorophenyl)-4-hydroxy-1-piperidyl]-1-(4-fluorophenyl)-butan-1-one | |
| Clinical data | |
| Trade names | Haldol |
| AHFS/Drugs.com | monograph |
| MedlinePlus | a682180 |
| Pregnancy cat. | C (AU) C (US) |
| Legal status | Prescription Only (S4) (AU) ℞-only (CA) POM (UK) ℞-only (US) |
| Routes | Oral, IM, IV, depot (asdecanoate ester) |
| Pharmacokinetic data | |
| Bioavailability | 60-70% (Oral)[1] |
| Protein binding | ~90%[1] |
| Metabolism | Liver-mediated[1] |
| Half-life | 14-26 hours (IV), 20.7 hours (IM), 14-37 hours (oral)[1] |
| Excretion | Biliary (hence in faeces) and in urine[1][2] |
| Identifiers | |
| CAS number | 52-86-8 |
| ATC code | N05AD01 |
| PubChem | CID 3559 |
| IUPHAR ligand | 86 |
| DrugBank | DB00502 |
| ChemSpider | 3438 |
| UNII | J6292F8L3D |
| KEGG | D00136 |
| ChEBI | CHEBI:5613 |
| ChEMBL | CHEMBL54 |
| Chemical data | |
| Formula | C21H23ClFNO2 |
| Mol. mass | 375.9 g/mol |
History
Haloperidol was discovered by Paul Janssen.[70] It was developed in 1958 at the Belgian company Janssen Pharmaceutica and submitted to the first of clinical trials in Belgiumlater that year.[71]
Haloperidol was approved by the U.S. Food and Drug Administration (FDA) on April 12, 1967; it was later marketed in the U.S. and other countries under the brand name Haldol byMcNeil Laboratories.[citation needed]
Society and culture
Coincident with civil unrest in the United States in the 1960s and 1970s, schizophrenia was racialized to match the behavior of angry/violent black men. Haldol was promoted as a way to pacify them, and was marketed to appeal to feelings of racial unease. (cf. Metzl 2010. The Protest Psychosis)
Soviet dissidents, including medical staff, have reported several times on the use of haloperidol in the Soviet Union for punitive purposes or simply to break the prisoners’ will.[72][73][74] Notable dissidents who were administered haloperidol as part of their court-ordered treatment were Sergei Kovalev and Leonid Plyushch.[75] The accounts Plyushch gave in the West, after he was allowed to leave the Soviet Union in 1976, were instrumental in triggering Western condemnation of Soviet practices at the World Psychiatric Association‘s 1977 meeting.[76] The use of haloperidol in the Soviet Union’s psychiatric system was prevalent because it was one of the few psychotropic drugs produced in quantity in the USSR.[77]
Haloperidol has been used for its sedating effects during the deportations of immigrants by the United States Immigration and Customs Enforcement (ICE). During 2002-2008, federal immigration personnel used haloperidol to sedate 356 deportees. By 2008, following court challenges over the practice, it was given to only three detainees. Following lawsuits, U.S. officials changed the procedure so the drug is administered only by the recommendation of medical personnel and under court order.[78][79]
Brand names
Haloperidol is sold under the tradenames Aloperidin, Bioperidolo, Brotopon, Dozic, Duraperidol (Germany), Einalon S, Eukystol, Haldol (common tradename in the US and UK), Halosten, Keselan, Linton, Peluces, Serenace and Sigaperidol.
Veterinary use
Haloperidol is also used on many different kinds of animals. It appears to be particularly successful when given to birds, e.g., a parrot that will otherwise continuously pluck its feathers out.[80]
References
- ^ Jump up to:a b c d e f g h i Kudo, S; Ishizaki T (December 1999). “Pharmacokinetics of haloperidol: an update”. Clinical pharmacokinetics 37 (6): 435-456. doi:10.2165/00003088-199937060-00001. PMID 10628896.
- Jump up^ “PRODUCT INFORMATION Serenace” (PDF). TGA eBusiness Services. Aspen Pharma Pty Ltd. 29 September 2011. Retrieved 29 May 2014.
- Jump up^ Joint Formulary Committee (2013). British National Formulary (BNF) (65 ed.). London, UK: Pharmaceutical Press. p. 229-230. ISBN 978-0-85711-084-8. edit
- ^ Jump up to:a b Brayfield, A, ed. (13 December 2013). “Haloperidol”. Martindale: The Complete Drug Reference. London, UK: Pharmaceutical Press. Retrieved 29 May 2014.
- Jump up^ “TGA Approved Terminology for Medicines” (PDF). Therapeutic Goods Administration. Australian Government, Department of Health and Ageing. July 1999. p. 66. Retrieved 29 May 2014.
- Jump up^ Rossi, S, ed. (2013). Australian Medicines Handbook (2013 ed.). Adelaide: The Australian Medicines Handbook Unit Trust. ISBN 978-0-9805790-9-3. edit
- Jump up^ “WHO Model List of Essential Medicines” (PDF). World Health Organization. October 2013. p. 7, 35. Retrieved 22 April 2014.
- Jump up^ “Haldol Official FDA information, side effects and uses”. Drugs.com. Retrieved 2013-10-03.
- Jump up^ Giannini, A. James; Underwood, Ned A.; Condon, Maggie (2000). “Acute Ketamine Intoxication Treated by Haloperidol”. American Journal of Therapeutics 7 (6): 389–91.doi:10.1097/00045391-200007060-00008. PMID 11304647.
- Jump up^ Giannini, A. James; Eighan, Michael S.; Loiselle, Robert H.; Giannini, Matthew C. (1984). “Comparison of Haloperidol and Chlorpromazine in the Treatment of Phencyclidine Psychosis”. The Journal of Clinical Pharmacology 24 (4): 202–4.doi:10.1002/j.1552-4604.1984.tb01831.x. PMID 6725621.
- Jump up^ Cavanaugh, SV (1986). “Psychiatric emergencies”. The Medical clinics of North America 70 (5): 1185–202. PMID 3736271.
- Jump up^ Currier, Glenn W. (2003). “The Controversy over ‘Chemical Restraint’ In Acute Care Psychiatry”. Journal of Psychiatric Practice 9 (1): 59–70. doi:10.1097/00131746-200301000-00006. PMID 15985915.
- Jump up^ Irving, Claire B; Adams, Clive E; Lawrie, Stephen (2006). “Haloperidol versus placebo for schizophrenia”. In Irving, Claire B. Cochrane Database of Systematic Reviews (4): CD003082. doi:10.1002/14651858.CD003082.pub2. PMID 17054159.
- Jump up^ Allen, MH; Currier, GW; Hughes, DH; Reyes-Harde, M; Docherty, JP; Expert Consensus Panel for Behavioral Emergencies (2001). “The Expert Consensus Guideline Series. Treatment of behavioral emergencies”. Postgraduate Medicine (Spec No): 1–88; quiz 89–90. PMID 11500996.
- Jump up^ Allen, Michael H.; Currier, Glenn W.; Hughes, Douglas H.; Docherty, John P.; Carpenter, Daniel; Ross, Ruth (2003). “Treatment of Behavioral Emergencies: A Summary of the Expert Consensus Guidelines”. Journal of Psychiatric Practice 9 (1): 16–38. doi:10.1097/00131746-200301000-00004. PMID 15985913.
- Jump up^ Allen, Michael H.; Currier, Glenn W.; Carpenter, Daniel; Ross, Ruth W.; Docherty, John P. (2005). “Introduction: Methods, Commentary, and Summary”. Journal of Psychiatric Practice 11: 5. doi:10.1097/00131746-200511001-00002.
- Jump up^ Ballard, Clive; Lana, Marisa Margallo; Theodoulou, Megan; Douglas, Simon; McShane, Rupert; Jacoby, Robin; Kossakowski, Katja; Yu, Ly-Mee; Juszczak, Edmund; on behalf of the Investigators DART AD (2008). “A Randomised, Blinded, Placebo-Controlled Trial in Dementia Patients Continuing or Stopping Neuroleptics (The DART-AD Trial)”. In Brayne, Carol. PLoS Medicine 5 (4): e76. doi:10.1371/journal.pmed.0050076.PMC 2276521. PMID 18384230. Lay summary – BBC News (April 1, 2008). “Neuroleptics provided no benefit for patients with mild behavioural problems, but were associated with a marked deterioration in verbal skills”
- ^ Jump up to:a b c d e “Haldol Official FDA information, side effects and uses”. Drugs.com. Retrieved 2013-10-03.
- Jump up^ “Haloperidol at Chemeurope”.
- ^ Jump up to:a b Work Group on Schizophrenia. “Practice Guideline for the Treatment of Patients With Schizophrenia Second Edition”. Retrieved 21 April 2014.
- Jump up^ Oosthuizen, P.; Emsley, R. A.; Turner, J.; Keyter, N. (2001). “Determining the optimal dose of haloperidol in first-episode psychosis”. Journal of Psychopharmacology 15 (4): 251–5. doi:10.1177/026988110101500403. PMID 11769818.
- Jump up^ Tauscher, Johannes; Kapur, Shitij (2001). “Choosing the Right Dose of Antipsychotics in Schizophrenia”. CNS Drugs 15 (9): 671–8. doi:10.2165/00023210-200115090-00001.PMID 11580306.
- Jump up^ Goodman and Gilman’s Pharmacological Basis of Therapeutics, 10th edition (McGraw-Hill, 2001).[page needed]
- Jump up^ American Academy of Hospice and Palliative Medicine. “Five Things Physicians and Patients Should Question”. Choosing Wisely: an initiative of the ABIM Foundation(American Academy of Hospice and Palliative Medicine). Retrieved August 1, 2013., which cites
- Smith, Thomas J.; Ritter, Joseph K.; Poklis, Justin L.; Fletcher, Devon; Coyne, Patrick J.; Dodson, Patricia; Parker, Gwendolyn (2012). “ABH Gel is Not Absorbed from the Skin of Normal Volunteers”. Journal of Pain and Symptom Management 43(5): 961–6. doi:10.1016/j.jpainsymman.2011.05.017. PMID 22560361.
- Weschules, Douglas J. (2005). “Tolerability of the Compound ABHR in Hospice Patients”. Journal of Palliative Medicine 8 (6): 1135–43.doi:10.1089/jpm.2005.8.1135. PMID 16351526.
- Jump up^ PRODUCT INFORMATION [Internet]. 2011 [cited 2013 Sep 29]. Available from:https://www.ebs.tga.gov.au/ebs/picmi/picmirepository.nsf/pdf?OpenAgent&id=CP-2011-PI-03532-3
- Jump up^ HALDOL® Injection FOR INTRAMUSCULAR INJECTION ONLY PRODUCT INFORMATION [Internet]. Janssen; 2011 [cited 2013 Sep 29]. Available from:https://www.ebs.tga.gov.au/ebs/picmi/picmirepository.nsf/pdf?OpenAgent&id=CP-2009-PI-00998-3
- Jump up^ Truven Health Analytics, Inc. DrugPoint® System (Internet) [cited 2013 Sep 29]. Greenwood Village, CO: Thomsen Healthcare; 2013.
- Jump up^ Joint Formulary Committee. British National Formulary (BNF) 65. Pharmaceutical Pr; 2013.
- ^ Jump up to:a b Leucht, Stefan; Cipriani, Andrea; Spineli, Loukia; Mavridis, Dimitris; Örey, Deniz; Richter, Franziska; Samara, Myrto; Barbui, Corrado; Engel, Rolf R; Geddes, John R; Kissling, Werner; Stapf, Marko Paul; Lässig, Bettina; Salanti, Georgia; Davis, John M (2013). “Comparative efficacy and tolerability of 15 antipsychotic drugs in schizophrenia: A multiple-treatments meta-analysis”. The Lancet 382 (9896): 951–62.doi:10.1016/S0140-6736(13)60733-3. PMID 23810019.
- ^ Jump up to:a b Silvestri, Simone; Seeman, Mary V.; Negrete, Juan-Carlos; Houle, Sylvain; Shammi, C.M.; Remington, Garry J.; Kapur, Shitij; Zipursky, Robert B.; Wilson, Alan A.; Christensen, Bruce K.; Seeman, Philip (2000). “Increased dopamine D 2 receptor binding after long-term treatment with antipsychotics in humans: A clinical PET study”.Psychopharmacology 152 (2): 174–80. doi:10.1007/s002130000532.PMID 11057521.
- Jump up^ Dorph-Petersen, Karl-Anton; Pierri, Joseph N; Perel, James M; Sun, Zhuoxin; Sampson, Allan R; Lewis, David A (2005). “The Influence of Chronic Exposure to Antipsychotic Medications on Brain Size before and after Tissue Fixation: A Comparison of Haloperidol and Olanzapine in Macaque Monkeys”. Neuropsychopharmacology 30 (9): 1649–61. doi:10.1038/sj.npp.1300710. PMID 15756305.
- Jump up^ Konopaske, Glenn T.; Dorph-Petersen, Karl-Anton; Sweet, Robert A.; Pierri, Joseph N.; Zhang, Wei; Sampson, Allan R.; Lewis, David A. (2008). “Effect of Chronic Antipsychotic Exposure on Astrocyte and Oligodendrocyte Numbers in Macaque Monkeys”. Biological Psychiatry 63 (8): 759–65.doi:10.1016/j.biopsych.2007.08.018. PMC 2386415. PMID 17945195.
- Jump up^ Vernon, Anthony C.; Natesan, Sridhar; Modo, Mike; Kapur, Shitij (2011). “Effect of Chronic Antipsychotic Treatment on Brain Structure: A Serial Magnetic Resonance Imaging Study with Ex Vivo and Postmortem Confirmation”. Biological Psychiatry 69 (10): 936–44. doi:10.1016/j.biopsych.2010.11.010. PMID 21195390.
- Jump up^ Breggin, Peter R. (2007). “Neuroleptic-Induced Neurotoxicity, Brain Damage, Persistent Cognitive Deficits, Dementia, and Psychosis”. Brain disabling treatments in psychiatry. Springer. pp. 85–114. ISBN 0-8261-2934-X.
- Jump up^ Halbreich, U; Shen, J; Panaro, V (1996). “Are chronic psychiatric patients at increased risk for developing breast cancer?”. The American Journal of Psychiatry 153 (4): 559–60. PMID 8599407.
- Jump up^ Dalton, Susanne Oksbjerg; Mellemkjaer, Lene; Thomassen, Lars; Mortensen, Preben B.; Johansen, Christoffer (2005). “Risk for cancer in a cohort of patients hospitalized for schizophrenia in Denmark, 1969–1993″. Schizophrenia Research 75 (2–3): 315–24.doi:10.1016/j.schres.2004.11.009. PMID 15885523.
- Jump up^ Grinshpoon, Alexander; Barchana, Micha; Ponizovsky, Alexander; Lipshitz, Irena; Nahon, Daniella; Tal, Orna; Weizman, Abraham; Levav, Itzhak (2005). “Cancer in schizophrenia: Is the risk higher or lower?”. Schizophrenia Research 73 (2–3): 333–41.doi:10.1016/j.schres.2004.06.016. PMID 15653279.
- Jump up^ Szarfman, Ana; Tonning, Joseph M; Levine, Jonathan G; Doraiswamy, P. Murali (2006). “Atypical Antipsychotics and Pituitary Tumors: A Pharmacovigilance Study”.Pharmacotherapy 26 (6): 748–58. doi:10.1592/phco.26.6.748. PMID 16716128.
- Jump up^ Hippisley-Cox, Julia; Vinogradova, Y; Coupland, C; Parker, C (2007). “Risk of Malignancy in Patients with Schizophrenia or Bipolar Disorder”. Archives of General Psychiatry 64 (12): 1368–76. doi:10.1001/archpsyc.64.12.1368. PMID 18056544.
- Jump up^ Levav, Itzhak; Kohn, Robert; Barchana, Micha; Lipshitz, Irena; Pugachova, Inna; Weizman, Abraham; Grinshpoon, Alexander (2009). “The risk for cancer among patients with schizoaffective disorders”. Journal of Affective Disorders 114 (1–3): 316–20.doi:10.1016/j.jad.2008.06.010. PMID 18675461.
- Jump up^ De Hert, M; Correll, CU; Bobes, J; Cetkovich-Bakmas, M; Cohen, D; Asai, I; Detraux, J; Gautam, S; Möller, HJ; Ndetei, DM; Newcomer, JW; Uwakwe, R; Leucht, S (2011).“Physical illness in patients with severe mental disorders. I. Prevalence, impact of medications and disparities in health care”. World psychiatry 10 (1): 52–77.PMC 3048500. PMID 21379357.
- Jump up^ Leentjens, Albert FG; Van Der Mast, Rose C (2005). “Delirium in elderly people: An update”. Current Opinion in Psychiatry 18 (3): 325–30.doi:10.1097/01.yco.0000165603.36671.97. PMID 16639157.
- Jump up^ Sandyk, R; Hurwitz, MD (1983). “Toxic irreversible encephalopathy induced by lithium carbonate and haloperidol. A report of 2 cases”. South African medical journal 64 (22): 875–6. PMID 6415823.
- Jump up^ Bush, S. E.; Hatton, R. C.; Winterstein, A. G.; Thomson, M. R.; Woo, G. W. (2008). “Effects of concomitant amiodarone and haloperidol on Q-Tc interval prolongation”.American Journal of Health-System Pharmacy 65 (23): 2232–6.doi:10.2146/ajhp080039. PMID 19020191.
- Jump up^ Igarashi, K.; Kasuya, F.; Fukui, M.; Usuki, E.; Castagnoli Jr, N. (1995). “Studies on the metabolism of haloperidol (HP): The role of CYP3A in the production of the neurotoxic pyridinium metabolite HPP+ found in rat brain following ip administration of HP”. Life Sciences 57 (26): 2439–46. doi:10.1016/0024-3205(95)02240-5. PMID 8847965.
- Jump up^ Usuki, Etsuko; Pearce, Robin; Parkinson, Andrew; Castagnoli, Neal (1996). “Studies on the Conversion of Haloperidol and Its Tetrahydropyridine Dehydration Product to Potentially Neurotoxic Pyridinium Metabolites by Human Liver Microsomes”. Chemical Research in Toxicology 9 (4): 800–6. doi:10.1021/tx960001y. PMID 8831826.
- Jump up^ Avent, Kathryn M.; Devoss, J. J.; Gillam, Elizabeth M. J. (2006). “Cytochrome P450-Mediated Metabolism of Haloperidol and Reduced Haloperidol to Pyridinium Metabolites”.Chemical Research in Toxicology 19 (7): 914–20. doi:10.1021/tx0600090.PMID 16841959.
- Jump up^ Avent, Kathryn M.; Riker, Richard R.; Fraser, Gilles L.; Van Der Schyf, Cornelis J.; Usuki, Etsuko; Pond, Susan M. (1997). “Metabolism of haloperidol to pyridinium species in patients receiving high doses intravenously: Is HPTP an intermediate?”. Life Sciences61 (24): 2383–90. doi:10.1016/S0024-3205(97)00955-7. PMID 9399630.
- Jump up^ Kawashima, Hidekazu; Iida, Yasuhiko; Kitamura, Youji; Saji, Hideo (2004). “Binding of 4-(4-chlorophenyl)-1-[4-(4-fluorophenyl)-4-oxobutyl]pyridinium ion (HPP+), a metabolite of haloperidol, to synthetic melanin: Implications for the dopaminergic neurotoxicity of HPP+“. Neurotoxicity Research 6 (7–8): 535–42. doi:10.1007/BF03033449.PMID 15639785.
- ^ Jump up to:a b Rollema, H; Skolnik, M; d’Engelbronner, J; Igarashi, K; Usuki, E; Castagnoli Jr, N (1994). “MPP(+)-like neurotoxicity of a pyridinium metabolite derived from haloperidol: In vivo microdialysis and in vitro mitochondrial studies”. The Journal of Pharmacology and Experimental Therapeutics 268 (1): 380–7. PMID 8301579.
- Jump up^ Mythri, Rajeswara Babu; Jagatha, Balusamy; Pradhan, Nityananda; Andersen, Julie; Bharath, M. M. Srinivas (2006). “Mitochondrial Complex I Inhibition in Parkinson’s Disease: How Can Curcumin Protect Mitochondria?”. Antioxidants & Redox Signaling 9(3): 399–408. doi:10.1089/ars.2007.9.ft-25. PMID 17184173.
- Jump up^ Beal, M. Flint (2006). “Mitochondria, Oxidative Damage, and Inflammation in Parkinson’s Disease”. Annals of the New York Academy of Sciences 991: 120–31.doi:10.1111/j.1749-6632.2003.tb07470.x. PMID 12846981.
- Jump up^ Bishnoi, Mahendra; Chopra, Kanwaljit; Kulkarni, Shrinivas K. (2008). “Activation of striatal inflammatory mediators and caspase-3 is central to haloperidol-induced orofacial dyskinesia”. European Journal of Pharmacology 590 (1–3): 241–5.doi:10.1016/j.ejphar.2008.06.033. PMID 18590723.
- Jump up^ Eyles, Darryl W.; Avent, Kathryn M.; Stedman, Terry J.; Pond, Susan M. (1997). “Two pyridinium metabolites of haloperidol are present in the brain of patients at post-mortem”.Life Sciences 60 (8): 529–34. doi:10.1016/S0024-3205(96)00656-X. PMID 9042387.
- Jump up^ Ulrich, Sven; Neuhof, Sabine; Braun, Verena; Danos, Peter; Pester, Uwe; Hoy, Ludwig (2000). “Disposition of Haloperidol Pyridinium and Reduced Haloperidol Pyridinium in Schizophrenic Patients: No Relationship with Clinical Variables During Short-Term Treatment”. Journal of Clinical Psychopharmacology 20 (2): 210–9.doi:10.1097/00004714-200004000-00014. PMID 10770460.
- Jump up^ Ulrich, S.; Sandmann, U.; Genz, A. (2005). “Serum Concentrations of Haloperidol Pyridinium Metabolites and the Relationship with Tardive Dyskinesia and Parkinsonism: A Cross-Section Study in Psychiatric Patients”. Pharmacopsychiatry 38 (4): 171–7.doi:10.1055/s-2005-871240. PMID 16025420.
- Jump up^ “Haloperidol at Drugs.com”.
- Jump up^ Seeman, P; Tallerico, T (1998). “Antipsychotic drugs which elicit little or no Parkinsonism bind more loosely than dopamine to brain D2 receptors, yet occupy high levels of these receptors”. Molecular Psychiatry 3 (2): 123–34.doi:10.1038/sj.mp.4000336. PMID 9577836.
- Jump up^ Schotte, A; Janssen PF; Megens AA; Leysen JE (1993). “Occupancy of central neurotransmitter receptors by risperidone, clozapine and haloperidol, measured ex vivo by quantitative autoradiography”. Brain Research 631 (2): 191–202. doi:10.1016/0006-8993(93)91535-z. PMID 7510574. Retrieved 21 April 2014.
- Jump up^ Leysen, JE; Janssen, PM; Gommeren, W; Wynants, J; Pauwels, PJ; Janssen, PA (1992). “In vitro and in vivo receptor binding and effects on monoamine turnover in rat brain regions of the novel antipsychotics risperidone and ocaperidone”. Molecular Pharmacology 41 (3): 494–508. PMID 1372084.
- Jump up^ Malmberg, Åsa; Mikaels, Åsa; Mohell, Nina (1998). “Agonist and Inverse Agonist Activity at the Dopamine D3 Receptor Measured by Guanosine 5′-[γ-Thio]Triphosphate-[35S] Binding”. The Journal of Pharmacology and Experimental Therapeutics 285 (1): 119–26. PMID 9536001.
- Jump up^ Leysen, JE; Janssen, PM; Megens, AA; Schotte, A (1994). “Risperidone: A novel antipsychotic with balanced serotonin-dopamine antagonism, receptor occupancy profile, and pharmacologic activity”. The Journal of Clinical Psychiatry 55 (Suppl): 5–12.PMID 7520908.
- Jump up^ Cobos, Enrique J.; Del Pozo, Esperanza; Baeyens, José M. (2007). “Irreversible blockade of sigma-1 receptors by haloperidol and its metabolites in guinea pig brain and SH-SY5Y human neuroblastoma cells”. Journal of Neurochemistry 102 (3): 812–25.doi:10.1111/j.1471-4159.2007.04533.x. PMID 17419803.
- Jump up^ Colabufo, Nicolaantonio; Berardi, Francesco; Contino, Marialessandra; Niso, Mauro; Abate, Carmen; Perrone, Roberto; Tortorella, Vincenzo (2004). “Antiproliferative and cytotoxic effects of some σ2 agonists and σ1 antagonists in tumour cell lines”. Naunyn-Schmiedeberg’s Archives of Pharmacology 370 (2): 106–13. doi:10.1007/s00210-004-0961-2. PMID 15322732.
- ^ Jump up to:a b c d e f g h i j k Kroeze, Wesley K; Hufeisen, Sandra J; Popadak, Beth A; Renock, Sean M; Steinberg, Seanna; Ernsberger, Paul; Jayathilake, Karu; Meltzer, Herbert Y; Roth, Bryan L (2003). “H1-Histamine Receptor Affinity Predicts Short-Term Weight Gain for Typical and Atypical Antipsychotic Drugs”. Neuropsychopharmacology 28 (3): 519–26.doi:10.1038/sj.npp.1300027. PMID 12629531.
- Jump up^ Ilyin, VI; Whittemore, ER; Guastella, J; Weber, E; Woodward, RM (1996). “Subtype-selective inhibition of N-methyl-D-aspartate receptors by haloperidol”. Molecular Pharmacology 50 (6): 1541–50. PMID 8967976.
- Jump up^ “drugs.com”.
- ^ Jump up to:a b Kornhuber, Johannes; Schultz, Andreas; Wiltfang, Jens; Meineke, Ingolf; Gleiter, Christoph H.; Zöchling, Robert; Boissl, Karl-Werner; Leblhuber, Friedrich; Riederer, Peter (1999). “Persistence of Haloperidol in Human Brain Tissue”. The American Journal of Psychiatry 156 (6): 885–90. PMID 10360127.
- Jump up^ Kornhuber, Johannes; Wiltfang, Jens; Riederer, Peter; Bleich, Stefan (2006). “Neuroleptic drugs in the human brain: Clinical impact of persistence and region-specific distribution”. European Archives of Psychiatry and Clinical Neuroscience 256 (5): 274–80. doi:10.1007/s00406-006-0661-7. PMID 16788768.
- Jump up^ Healy, David (1996). The psychopharmacologists 1. London: Chapman and Hall.ISBN 978-1-86036-008-4.[page needed]
- Jump up^ Granger, Bernard; Albu, Simona (2005). “The Haloperidol Story”. Annals of Clinical Psychiatry 17 (3): 137–40. doi:10.1080/10401230591002048. PMID 16433054.
- Jump up^ Podrabinek, Aleksandr (1980). Punitive Medicine. Ann Arbor Mich.: Karoma Publishers. pp. 15–20. ISBN 0-89720-022-5.
- Jump up^ Kosserev, I.; Crawshaw, R. (1994). “Medicine and the Gulag”. BMJ 309 (6970): 1726–30. doi:10.1136/bmj.309.6970.1726. PMC 2542687. PMID 7820004.
- Jump up^ de Boer, S. P.; E. J. Driessen; H. L. Verhaar (1982). Biographical Dictionary of Dissidents in the Soviet Union, 1956-1975. The Hague: Martinus Nijhoff Publishers.ISBN 90-247-2538-0.[page needed]
- Jump up^ Wade, N. (1976). “Sergei Kovalev: Biologist Denied Due Process and Medical Care”.Science 194 (4265): 585–7. doi:10.1126/science.194.4265.585. PMID 17818411.
- Jump up^ “Censuring the Soviets”. TIME (CNN). 1977-09-12. Retrieved 2009-06-21.
- Jump up^ The Children of Pavlov, TIME, Jun. 23, 1980
- Jump up^ “Fewer US deportees being sedated for removal”. Epilepsy.com. Associated Press. 2008-12-30. Retrieved 2009-06-21.
- Jump up^ Solis, Dianne (2009-01-05). “U.S. cuts back on sedating deportees with Haldol”. Seattle Times. Retrieved 2009-06-21.
- Jump up^ “Veterinary:Avian at Lloyd Center Pharmacy”.
External links
- Rx-List.com – Haloperidol
- Medline plus – Haloperidol
- Swiss scientific information on Haldol
- “WHO Model List of Essential Medicines” (PDF) (16th list (updated) ed.). World Health Organization. March 2010. Retrieved 2010-09-14.
- U.S. National Library of Medicine: Drug Information Portal – Haloperidol
FDA approves Avastin to treat patients with aggressive and late-stage cervical cancer

August 14, 2014
The U.S. Food and Drug Administration today approved a new use for Avastin (bevacizumab) to treat patients with persistent, recurrent or late-stage (metastatic) cervical cancer.
Cervical cancer grows in the tissues of the lower part of the uterus known as the cervix. It commonly occurs when human papillomaviruses (HPV), a virus that spreads through sexual contact, cause cells to become cancerous. Although there are two licensed vaccines available to prevent many types of HPV that can cause cervical cancer, the National Cancer Institute estimates that 12,360 American women will be diagnosed with cervical cancer and 4,020 will die from the disease in 2014.
Avastin works by interfering with the blood vessels that fuel the development of cancerous cells. The new indication for cervical cancer is approved for use in combination with chemotherapy drugs paclitaxel and cisplatin or in combination with paclitaxel and topotecan.
“Avastin is the first drug approved for patients with late-stage cervical cancer since the 2006 approval of topotecan with cisplatin,” said Richard Pazdur, M.D., director of the Office of Hematology and Oncology Products in the FDA’s Center for Drug Evaluation and Research. “It is also the first biologic agent approved for patients with late-stage cervical cancer and was approved in less than four months under the FDA’s priority review program, demonstrating the agency’s commitment to making promising therapies available to patients faster.”
The FDA reviewed Avastin for treatment of patients with cervical cancer under its priority review program because the drug demonstrated the potential to be a significant improvement in safety or effectiveness over available therapy in the treatment of a serious condition. Priority review provides an expedited review of a drug’s application.
The safety and effectiveness of Avastin for treatment of patients with cervical cancer was evaluated in a clinical study involving 452 participants with persistent, recurrent, or late-stage disease. Participants were randomly assigned to receive paclitaxel and cisplatin with or without Avastin or paclitaxel and topotecan with or without Avastin. Results showed an increase in overall survival to 16.8 months in participants who received chemotherapy in combination with Avastin as compared to 12.9 months for those receiving chemotherapy alone.
The most common side effects associated with use of Avastin in patients with cervical cancer include fatigue, decreased appetite, high blood pressure (hypertension), increased glucose in the blood (hyperglycemia), decreased magnesium in the blood (hypomagnesemia), urinary tract infection, headache and decreased weight. Perforations of the gastrointestinal tract and abnormal openings between the gastrointestinal tract and vagina (enterovaginal fistula) also were observed in Avastin-treated patients.
Avastin is marketed by South San Francisco, California-based Genentech, a member of the Roche Group.
| Country | Patent Number | Approved | Expires (estimated) |
|---|---|---|---|
| Canada | 2286330 | 2008-06-10 | 2018-04-03 |
| Canada | 2145985 | 2003-09-16 | 2012-10-28 |
| Property | Value | Source |
|---|---|---|
| melting point | 61 °C (FAB fragment), 71 °C (whole mAb) | Vermeer, A.W.P. & Norde, W., Biophys. J. 78:394-404 (2000) |

| Protein chemical formula | C6538H10034N1716O2033S44 |
|---|---|
| Protein average weight | 149 kDa |
A recombinant humanized monoclonal IgG1 antibody that binds to and inhibits the biologic activity of human vascular endothelial growth factor (VEGF). Bevacizumab contains human framework regions and the complementarity-determining regions of a murine antibody that binds to VEGF. Bevacizumab is produced in a Chinese Hamster Ovary mammalian cell expression system in a nutrient medium containing the antibiotic gentamicin and has a molecular weight of approximately 149 kilodaltons.
sequence
>"Bevacizumab light chain" DIQMTQSPSSLSASVGDRVTITCSASQDISNYLNWYQQKPGKAPKVLIYFTSSLHSGVPS RFSGSGSGTDFTLTISSLQPEDFATYYCQQYSTVPWTFGQGTKVEIKRTVAAPSVFIFPP SDEQLKSGTASVVCLLNNFYPREAKVQWKVDNALQSGNSQESVTEQDSKDSTYSLSSTLT LSKADYEKHKVYACEVTHQGLSSPVTKSFNRGEC
>"Bevacizumab heavy chain" EVQLVESGGGLVQPGGSLRLSCAASGYTFTNYGMNWVRQAPGKGLEWVGWINTYTGEPTY AADFKRRFTFSLDTSKSTAYLQMNSLRAEDTAVYYCAKYPHYYGSSHWYFDVWGQGTLVT VSSASTKGPSVFPLAPSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVL QSSGLYSLSSVVTVPSSSLGTQTYICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPEL LGGPSVFLFPPKPKDTLMISRTPEVTCVVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREE QYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKALPAPIEKTISKAKGQPREPQVYTLPPS REEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKTTPPVLDSDGSFFLYSKLTVDK SRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK
| Monoclonal antibody | |
|---|---|
| Type | Whole antibody |
| Source | Humanized (from mouse) |
| Target | VEGF-A |
| Clinical data | |
| Trade names | Avastin |
| AHFS/Drugs.com | monograph |
| Licence data | EMA:Link, US FDA:link |
| Pregnancy cat. | C (US) |
| Legal status | ℞ Prescription only |
| Routes | Intravenous |
| Pharmacokinetic data | |
| Bioavailability | 100% (IV only) |
| Half-life | 20 days (range: 11–50 days) |
| Identifiers | |
| CAS number | 216974-75-3 |
| ATC code | L01XC07 |
| DrugBank | DB00112 |
| UNII | 2S9ZZM9Q9V |
| KEGG | D06409 |
| ChEMBL | CHEMBL1201583 |
| Chemical data | |
| Formula | C6638H10160N1720O2108S44 |
| Mol. mass | approx. 149 kDa |
Carbohydrate Metabolism
Leaders in Pharmaceutical Business Intelligence Group, LLC, Doing Business As LPBI Group, Newton, MA
Carbohydrate Metabolism
Larry H. Bernstein, MD, FCAP, Reporter and Curator
Leaders in Pharmaceutical Intelligence
http://pharmaceuticalintelligence.com/8-9-2014/Carbohydrate_Metabolism
This is the third portion of the discussion in a series of articles that began with signaling and signaling pathways. There are fine features on the functioning of enzymes and proteins, on sequential changes in a chain reaction, and on conformational changes that we shall return to. These are critical to developing a more complete understanding of life processes. I have indicated that many of the protein-protein interactions or protein-membrane interactions and associated regulatory features have been referred to previously, but the focus of the discussion or points made were different. Even though I considered placing this after the discussion of proteins and how they play out their essential role, I needed to lay out the scope of metabolic reactions and pathways, and their complementary changes. These may not appear to be adaptive, if the…
View original post 6,261 more words
Valdecoxib


Valdecoxib is a non-steroidal anti-inflammatory drug (NSAID) used in the treatment of osteoarthritis, rheumatoid arthritis, and painfulmenstruation and menstrual symptoms. It is a cyclooxygenase-2 selective inhibitor.
Valdecoxib was manufactured and marketed under the brand name Bextra by G. D. Searle & Company. It was approved by the United States Food and Drug Administration on November 20, 2001,[1] and was available by prescription in tablet form until 2005, when it was removed from the market due to concerns about possible increased risk of heart attack and stroke. The prodrugparecoxib is available in many countries.
Uses until 2005
In the United States, the Food and Drug Administration (FDA) approved valdecoxib for the treatment of osteoarthritis, adultrheumatoid arthritis, and primary dysmenorrhea.[2]
Valdecoxib was also used off-label for controlling acute pain and various types of surgical pain.[2]
Side-effects and withdrawal from market
On April 7, 2005, Pfizer withdrew Bextra from the U.S. market on recommendation by the FDA, citing an increased risk of heart attackand stroke and also the risk of a serious, sometimes fatal, skin reaction. This was a result of recent attention to prescription NSAIDs, such as Merck’s Vioxx. Other reported side-effects were angina and Stevens–Johnson syndrome.
Pfizer first acknowledged cardiovascular risks associated with Bextra in October 2004. The American Heart Association soon after was presented with a report indicating patients using Bextra while recovering from heart surgery were 2.19 times more likely to suffer a stroke or heart attack than those taking placebos.
In a large study published in JAMA 2006, valdecoxib appeared less adverse for renal (kidney) disease and heart arrhythmia compared to Vioxx, however elevated renal risks were slightly suggested.[3]
2009 settlement for off-label uses promotions
|
|
An editor has expressed a concern that this section lends undue weight to certain ideas relative to the article as a whole. Please help to discuss and resolve the dispute before removing this message. (December 2012) |
On September 2, 2009, the United States Department of Justice fined Pfizer $2.3 billion after one of its subsidiaries, Pharmacia & UpJohn Company, pled guilty to marketing four drugs including Bextra “with the intent to defraud or mislead.”[4] Pharmacia & UpJohn admitted to criminal conduct in the promotion of Bextra, and agreed to pay the largest criminal fine ever imposed in the United States for any matter, $1.195 billion.[5] A former Pfizer district sales manager was indicted and sentenced to home confinement for destroying documents regarding the illegal promotion of Bextra.[6][7] In addition, a Regional Manager pled guilty to distribution of a mis-branded product, and was fined $75,000 and twenty-four months on probation.[8]
The remaining $1 billion of the fine was paid to resolve allegations under the civil False Claims Act case and is the largest civil fraud settlement against a pharmaceutical company. Six whistle-blowers were awarded more than $102 million for their role in the investigation.[9] Former Pfizer sales representative John Kopchinski acted as a qui tam relator and filed a complaint in 2004 outlining the illegal conduct in the marketing of Bextra.[10] Kopchinski was awarded $51.5 million for his role in the case because the improper marketing of Bextra was the largest piece of the settlement at $1.8 billion.[11]
Assay of Valdecoxib[13]
Several HPLC-UV methods have been reported for valdecoxib estimation in biological samples like human urine,[14] plasma,.[15][16] Valdecoxib has analytical methods for bioequivalence studies,[17][18] metabolite determination,[19][20][21] and estimation of formulation,[22] HPTLC method for simultaneous estimation in tablet dosage form.[23]
Brief background information
| Salt | ATC | Formula | MM | CAS |
|---|---|---|---|---|
| – | M01AH03 | C 16 H 14 N 2 O 3 S | 314.37 g / mol | 181695-72-7 |
| Systematic (IUPAC) name | |
|---|---|
| 4-(5-methyl-3-phenylisoxazol-4-yl)benzenesulfonamide | |
| Clinical data | |
| Trade names | Bextra |
| Pregnancy cat. | C (AU) May cause premature closure of the ductus arteriosus |
| Legal status | Prescription Only (S4) (AU)Withdrawn in U.S., EU, Canada& parts of Asia |
| Routes | Oral |
| Pharmacokinetic data | |
| Bioavailability | 83% |
| Protein binding | 98% |
| Metabolism | Hepatic (CYP3A4 and 2C9involved) |
| Half-life | 8 to 11 hours |
| Excretion | Renal |
| Identifiers | |
| CAS number | 181695-72-7 |
| ATC code | M01AH03 |
| PubChem | CID 119607 |
| DrugBank | DB00580 |
| ChemSpider | 106796 |
| UNII | 2919279Q3W |
| KEGG | D02709 |
| ChEBI | CHEBI:63634 |
| ChEMBL | CHEMBL865 |
| Chemical data | |
| Formula | C16H14N2O3S |
| Mol. mass | 314.364 g/mol |
Using
-
anti-inflammatory
-
antirheumatic
-
COX-2 inhibitor
Classes of substances
-
Benzenesulfonamide (s -imidy), as well as their derivatives
-
Isoxazoles
-
Synthesis pathway
| Synthesis a) |
|---|
 |
Synthesis
Source:[12]



Deoxybenzoin (I) is converted to the corresponding oxime (II) by treatment with NH2OH稨Cl under basic conditions either with sodium acetate in aqueous ethanol or in toluene in presence of potassium hydroxide in absolute ethanol. Deprotonation of the oxime under nitrogen with 2eq of butyllithium in THF followed by cyclization in ethyl acetate or acetic anhydride affords isoxazoline (III). Finally, treatment of (III) with cold chlorosulfonic acid followed by reaction of the intermediate sulfonyl chloride with aqueous ammonia affords the desired product.
J Med Chem2000,43,(5):775
Trade Names
| Country | Trade name | Manufacturer |
|---|---|---|
| Germany | Bextra | Pharmacia |
| USA | – “- | – “- |
| Ukraine | No | No |
Formulations
-
Tablets of 10 mg, 20 mg
Valdecoxib is chemically designated as 4-(5-methyl-3-phenyl-4-isoxazolyl) benzenesulfonamide and is a diaryl substituted isoxazole.
The empirical formula for valdecoxib is C16H14N2O3S, and the molecular weight is 314.36. Valdecoxib is a white crystalline powder that is relatively insoluble in water (10 µg/mL) at 25° C and pH 7.0, soluble in methanol and ethanol, and freely soluble in organic solvents and alkaline (pH=12) aqueous solutions.
BEXTRA (valdecoxib) Tablets for oral administration contain either 10 mg or 20 mg of valdecoxib. Inactive ingredients include lactose monohydrate, microcrystalline cellulose, pregelatinized starch, croscarmellose sodium,magnesium stearate, hydroxypropyl methylcellulose, polyethylene glycol, polysorbate 80, and titanium dioxide
………………………
NMR

Links
-
Talley, JJ et al .: J. Med. Chem. (JMCMAR) 43, 775-777 (2000).
-
US 5,859,257 (GD Searle; 12.1.1999; USA-prior. 13.2.1995).
Literature References:
Selective cyclooxygenase-2 (COX-2) inhibitor. Active metabolite of parecoxib, q.v. Prepn: J. J. Talley et al., WO 9625405 (1996 to Searle); eidem, US 5633272 (1997); and activity: eidem, J. Med. Chem. 43, 775 (2000).
Chromatographic determn of purity: D. A. Roston et al., J. Pharm. Biomed. Anal. 26, 339 (2001).
Gastrointestinal tolerability study: G. M. Eisen et al., Aliment. Pharmacol. Ther. 21, 591 (2005).
Clinical trial in hip arthroplasty: F. Camu et al., Am. J. Ther.9, 43 (2002).
Clinical comparison with oxycodone/acetominophen in dental pain: S. E. Daniels et al., J. Am. Dent. Assoc. 133, 611 (2002).
Clinical trial in migraine: D. Kudrow et al., Headache 45, 1151 (2005).
Review of clinical experience: M. Goldman, S. Schutzer, Formulary 37, 68-77 (2002); of clinical efficacy and safety: G. P. Joshi, Expert Rev. Neurother. 5, 11-24 (2005).
References
- Jump up^ Thomson Micromedex. “Valdecoxib. U.S. FDA Drug Approval.” Last accessed June 8, 2007.
- ^ Jump up to:a b “Pfizer to pay $2.3 billion to resolve criminal and civil health care liability relating to fraudulent marketing and the payment of kickbacks”. Stop Medicare Fraud, US Dept of Health & Human Svc, and of Justice. Retrieved 2012-07-04.
- Jump up^ “Adverse Effects of Cyclooxygenase-2 Inhibitors on Renal and Arrhythmia Events: Meta-Analysis of Randomized Trials”, (JAMA 2006, by Zhang JJ, Ding EL, Song Y.).
- Jump up^ http://news.bbc.co.uk/2/hi/business/8234533.stm Pfizer agrees record fraud fine
- Jump up^ http://www.usdoj.gov/usao/ma/Press%20Office%20-%20Press%20Release%20Files/Sept2009/PharmaciaPlea.html
- Jump up^ http://www.usdoj.gov/usao/ma/Press%20Office%20-%20Press%20Release%20Files/Mar2009/FarinaconvictionPR.html
- Jump up^ http://industry.bnet.com/pharma/10002882/pfizers-off-label-bextra-team-were-called-the-highlanders/
- Jump up^ http://www.usdoj.gov/usao/ma/Press%20Office%20-%20Press%20Release%20Files/June2009/HollowayMarySentencingPR.html
- Jump up^ http://www.fbi.gov/pressrel/pressrel09/justice_090209.htm
- Jump up^ http://www.phillipsandcohen.com/CM/NewsSettlements/NewsSettlements536.asp
- Jump up^ http://www.phillipsandcohen.com/CM/NewsSettlements/NewsSettlements531.asp
- Jump up^ Talley, J. J.; Brown, D. L.; Carter, J. S.; Graneto, M. J.; Koboldt, C. M.; Masferrer, J. L.; Perkins, W. E.; Rogers, R. S.; Shaffer, A. F.; Zhang, Y. Y.; Zweifel, B. S.; Seibert, K. (2000). “4-[5-Methyl-3-phenylisoxazol-4-yl]- benzenesulfonamide, Valdecoxib: A Potent and Selective Inhibitor of COX-2”. Journal of Medicinal Chemistry 43 (5): 775–777. doi:10.1021/jm990577v.PMID 10715145.
- Jump up^ Prafulla Kumar Sahu and M. Mathrusri Annapurna, Analytical method development by liquid chromatography, LAP Lambert Academic Publisher, Germany, 2011 ISBN 3-8443-2869-6.
- Jump up^ Zhang J Y, Fast D M and Breau A P, J Chromatogr B Analyt Technol Biomed Life Sci., 2003, 785(1), 123-134
- Jump up^ Ramakrishna N V S, Vishwottam K N; Wishu S and Koteshwara M, J Chromatogr B Analyt Technol Biomed Life Sci., 2004, 802(2), 271.
- Jump up^ Sane R T, Menon S, Deshpande A Y and Jain A, Chromatogr., 2005, 61(3-4), 137-141.
- Jump up^ Prafulla Kumar Sahu*, K. Ravi Sankar and M. Mathrusri Annapurna, Determination of Valdecoxib in human plasma using Reverse Phase HPLC”, E-Journal of Chemistry, 2011, 8(2), 875-881.
- Jump up^ Mandal U, Jayakumar M, Ganesan M, Nandi S, Pal T K, Chakraborty M K, Roy Chowdhary A. and Chattoraj T K, Indian Drugs, 2004, 41, 59.
- Zhang J.Y, Fast D.M and Breau, A.P, J Pharm Biomed Anal., 2003, 33, 61.
- Werner U, Werner D, Hinz B, Lanbrecht C and Brune K, J Biomed Chromatogr., 2004, 19, 113.
- Zhang J V, Fast D M and Breau A P, J Chromatogr B Anal Technol Biomed Life Sci., 2003, 785, 123.
- Sutariya V B, Rajashree M, Sankalia M G. and Priti P, Indian J Pharm Sci., 2004, 93, 112.
- J Gandhimathi M, Ravi T K, Shukla Nilima and Sowmiya G, Indian J Pharm Sci., 2007, 69(1), 145-147.
External links[edit]
- FDA Alert on Bextra withdrawal
- Large systematic review of adverse renal and arrhythmia risk of valdcoxib and other COX-2 inhibitors, JAMA 2006
ABACAVIR…….For the treatment of HIV-1 infection, in combination with other antiretroviral agents.
 |
|

Chemical structure of abacavir
|
{(1S,4R)-4-[2-amino-6-(cyclopropylamino)-9H-purin-9-yl]cyclopent-2-en-1-yl}methanol
(-)-cis-4-[2-Amino-6-(cyclopropylmethylamino)-9H-purin-9-yl]-2-cyclopentene-1-methanol
(1S, 4R)-4-[2-amino-6-(cyclopropylamino)-9H purin-9-yl]-2- cyclopentene-1 -methanol
Abacavir
Abacavir (ABC) is a powerful nucleoside analog reverse transcriptase inhibitor (NRTI) used to treat HIV and AIDS. [Wikipedia] Chemically, it is a synthetic carbocyclic nucleoside and is the enantiomer with 1S, 4R absolute configuration on the cyclopentene ring. In vivo, abacavir sulfate dissociates to its free base, abacavir.
Abacavir (ABC) ![]() i/ʌ.bæk.ʌ.vɪər/ is a nucleoside analog reverse transcriptase inhibitor (NRTI) used to treat HIV and AIDS. It is available under the trade name Ziagen (ViiV Healthcare) and in the combination formulations Trizivir (abacavir, zidovudine andlamivudine) and Kivexa/Epzicom (abacavir and lamivudine). It has been well tolerated: the main side effect is hypersensitivity, which can be severe, and in rare cases, fatal. Genetic testing can indicate whether an individual will be hypersensitive; over 90% of patients can safely take abacavir. However, in a separate study, the risk of heart attack increased by nearly 90%.[1]
i/ʌ.bæk.ʌ.vɪər/ is a nucleoside analog reverse transcriptase inhibitor (NRTI) used to treat HIV and AIDS. It is available under the trade name Ziagen (ViiV Healthcare) and in the combination formulations Trizivir (abacavir, zidovudine andlamivudine) and Kivexa/Epzicom (abacavir and lamivudine). It has been well tolerated: the main side effect is hypersensitivity, which can be severe, and in rare cases, fatal. Genetic testing can indicate whether an individual will be hypersensitive; over 90% of patients can safely take abacavir. However, in a separate study, the risk of heart attack increased by nearly 90%.[1]
Viral strains that are resistant to zidovudine (AZT) or lamivudine (3TC) are generally sensitive to abacavir (ABC), whereas some strains that are resistant to AZT and 3TC are not as sensitive to abacavir.
It is on the World Health Organization’s List of Essential Medicines, a list of the most important medication needed in a basic health system.[2]
Abacavir is a nucleoside reverse transcriptase inhibitor (NRTI) with activity against Human Immunodeficiency Virus Type 1 (HIV-1). Abacavir is phosphorylated to active metabolites that compete for incorporation into viral DNA. They inhibit the HIV reverse transcriptase enzyme competitively and act as a chain terminator of DNA synthesis. The concentration of drug necessary to effect viral replication by 50 percent (EC50) ranged from 3.7 to 5.8 μM (1 μM = 0.28 mcg/mL) and 0.07 to 1.0 μM against HIV-1IIIB and HIV-1BaL, respectively, and was 0.26 ± 0.18 μM against 8 clinical isolates. Abacavir had synergistic activity in cell culture in combination with the nucleoside reverse transcriptase inhibitor (NRTI) zidovudine, the non-nucleoside reverse transcriptase inhibitor (NNRTI) nevirapine, and the protease inhibitor (PI) amprenavir; and additive activity in combination with the NRTIs didanosine, emtricitabine, lamivudine, stavudine, tenofovir, and zalcitabine.
Brief background information
| Salt | ATC | Formula | MM | CAS |
|---|---|---|---|---|
| – | J05AF06 | C 14 H 18 N 6 O | 286.34 g / mol | 136470-78-5 |
| succinate | J05AF06 | C 14 H 18 N 6 O · C 4 H 6 O | 356.43 g / mol | 168146-84-7 |
| sulfate | J05AF06 | C 14 H 18 N 6 O · 1 / 2H 2 SO 4 | 670.76 g / mol | 188062-50-2 |
| Systematic (IUPAC) name | |
|---|---|
| {(1S,4R)-4-[2-amino-6-(cyclopropylamino)-9H-purin-9-yl]cyclopent-2-en-1-yl}methanol | |
| Clinical data | |
| Trade names | Ziagen |
| AHFS/Drugs.com | monograph |
| MedlinePlus | a699012 |
| Pregnancy cat. | B3 (AU) C (US) |
| Legal status | POM (UK) ℞-only (US) |
| Routes | Oral (solution or tablets) |
| Pharmacokinetic data | |
| Bioavailability | 83% |
| Metabolism | Hepatic |
| Half-life | 1.54 ± 0.63 h |
| Excretion | Renal (1.2% abacavir, 30% 5′-carboxylic acid metabolite, 36% 5′-glucuronide metabolite, 15% unidentified minor metabolites). Fecal (16%) |
| Identifiers | |
| CAS number | 136470-78-5 |
| ATC code | J05AF06 |
| PubChem | CID 441300 |
| DrugBank | DB01048 |
| ChemSpider | 390063 |
| UNII | WR2TIP26VS |
| KEGG | D07057 |
| ChEBI | CHEBI:421707 |
| ChEMBL | CHEMBL1380 |
| NIAID ChemDB | 028596 |
| Chemical data | |
| Formula | C14H18N6O |
| Mol. mass | 286.332 g/mol |
Abacavir is a carbocyclic synthetic nucleoside analogue and an antiviral agent. Intracellularly, abacavir is converted by cellular enzymes to the active metabolite carbovir triphosphate, an analogue of deoxyguanosine-5′-triphosphate (dGTP). Carbovir triphosphate inhibits the activity of HIV-1 reverse transcriptase (RT) both by competing with the natural substrate dGTP and by its incorporation into viral DNA. Viral DNA growth is terminated because the incorporated nucleotide lacks a 3′-OH group, which is needed to form the 5′ to 3′ phosphodiester linkage essential for DNA chain elongation.
Application
-
an antiviral agent, is used in the treatment of AIDS
-
ingibitor convertibility transkriptazы
Classes of substances
-
Adenine (6-aminopurines)
-
Aminoalcohols
-
Cyclopentenes and cyclopentadienes
-
Tsyklopropanы
-
-
-
| Country | Patent Number | Approved | Expires (estimated) |
|---|---|---|---|
| Canada | 2289753 | 2007-01-23 | 2018-05-14 |
| Canada | 1340589 | 1999-06-08 | 2016-06-08 |
| Canada | 2216634 | 2004-07-20 | 2016-03-28 |
| United States | 6641843 | 2000-02-04 | 2020-02-04 |
| United States | 5089500 | 1992-12-26 | 2009-12-26 |
PATENT
US5034394
Synthesis pathway
Abacavir, (-) cis-[4-[2-amino-6-cyclopropylamino)-9H-purin-9-yl]-2-cyclopenten-yl]-1 – methanol, a carbocyclic nucleoside which possesses a 2,3-dehydrocyclopentene ring, is referred to in United States Patent 5,034,394 as a reverse transcriptase inhibitor. Recently, a general synthetic strategy for the preparation of this type of compound and intermediates was reported [Crimmins, et. al., J. Org. Chem., 61 , 4192-4193 (1996) and 65, 8499-8509-4193 (2000)].
-
Abacavir is the International Nonproprietary Name (INN) of {(1S,4R)-4-[2-amino-6-(cyclopropylamino)-9H-purin-9-yl]-cyclopent-2-enyl}methanol and CAS No. 136470-78-5. Abacavir and therapeutically acceptable salts thereof, in particular the hemisulfate salt, are well-known as potent selective inhibitors of HIV-1 and HIV-2, and can be used in the treatment of human immunodeficiency virus (HIV) infection.
-
-
EP 434450-A discloses certain 9-substituted-2-aminopurines including abacavir and its salts, methods for their preparation, and pharmaceutical compositions using these compounds.
-
Different preparation processes of abacavir are known in the art. In some of them abacavir is obtained starting from an appropriate pyrimidine compound, coupling it with a sugar analogue residue, followed by a cyclisation to form the imidazole ring and a final introduction of the cyclopropylamino group at the 6 position of the purine ring.
-
According to the teachings of EP 434450-A , the abacavir base is finally isolated by trituration using acetonitrile (ACN) or by chromatography, and subsequently it can be transformed to a salt of abacavir by reaction with the corresponding acid. Such isolation methods (trituration and chromatography) usually are limited to laboratory scale because they are not appropriate for industrial use. Furthermore, the isolation of the abacavir base by trituration using acetonitrile gives a gummy solid (Example 7) and the isolation by chromatography (eluted from methanol/ethyl acetate) yields a solid foam (Example 19 or 28).
-
Other documents also describe the isolation of abacavir by trituration or chromatography, but always a gummy solid or solid foam is obtained (cf. WO9921861 and EP741710 ), which would be difficult to operate on industrial scale.
-
WO9852949 describes the preparation of abacavir which is isolated from acetone. According to this document the manufacture of the abacavir free base produces an amorphous solid which traps solvents and is, therefore, unsuitable for large scale purification, or for formulation, without additional purification procedures (cf. page 1 of WO 9852949 ). In this document, it is proposed the use of a salt of abacavir, in particular the hemisulfate salt which shows improved physical properties regarding the abacavir base known in the art. Said properties allow the manufacture of the salt on industrial scale, and in particular its use for the preparation of pharmaceutical formulations.
-
However, the preparation of a salt of abacavir involves an extra processing step of preparing the salt, increasing the cost and the time to manufacture the compound. Generally, the abacavir free base is the precursor compound for the preparation of the salt. Thus, depending on the preparation process used for the preparation of the salt, the isolation step of the abacavir free base must also be done.
The structure of abacavir corresponds to formula (I):
………………………………
http://www.google.co.in/patents/US5034394
EXAMPLE 21(-)-cis-4-[2-Amino-6-(cyclopropylmethylamino)-9H-purin-9-yl]-2-cyclopentene-1-methanol
The title compound of Example 7, (2.00 g, 6.50 mmol) was dissolved in 1,3-dimethyl-3,4,5,6-tetrahydro-2(1H)-pyrimidinone (Aldrich, 20 mL). Phosphoryl chloride (2.28 mL, 24.0 mmol) was added to the stirred, cooled (-10° C.) solution. After 3 minutes, cold water (80 mL) was added. The solution was extracted with chloroform (3×80 mL). The aqueous layer was diluted with ethanol (400 mL) and the pH adjusted to 6 with saturated aqueous NaOH. The precipitated inorganic salts were filtered off. The filtrate was further diluted with ethanol to a volume of 1 liter and the pH adjusted to 8 with additional NaOH. The resulting precipitate was filtered and dried to give the 5′-monophosphate of (±)-cis-4-[2-amino-6-(cyclopropylmethylamino)-9H-purin-9-yl]-2-cyclopentene-1-methanol as white powder (4.0 mmoles, 62% quantitated by UV absorbance); HPLC analysis as in Example 17 shows one peak. This racemic 5′ -monophosphate was dissolved in water (200 mL) and snake venom 5′-nucleotidase (EC 3.1.3.5) from Crotalus atrox (5,000 IU, Sigma) was added. After incubation at 37° C. for 10 days, HPLC analysis as in Example 17 showed that 50% of the starting nucleotide had been dephosphorylated to the nucleoside. These were separated on a 5×14 cm column of DEAE Sephadex A25 (Pharmacia) which had been preequilibrated with 50 mM ammonium bicarbonate. Title compound was eluted with 2 liters of 50 mM ammonium bicarbonate. Evaporation of water gave white powder which was dissolved in methanol, adsorbed on silica gel, and applied to a silica gel column. Title compound was eluted with methanol:chloroform/1:9 as a colorless glass. An acetonitrile solution was evaporated to give white solid foam, dried at 0.3 mm Hg over P2 O5 ; 649 mg (72% from racemate); 1 H-NMR in DMSO-d6 and mass spectrum identical with those of the racemate (title compound of Example 7); [α]20 D -48.0°, [α]20 436 -97.1°, [α]20 365 -149° (c=0.14, methanol).
Anal. Calcd. for C15 H20 N6 O.0.10CH3 CN: C, 59.96; H, 6.72; N, 28.06. Found: C, 59.93; H, 6.76; N, 28.03.
Continued elution of the Sephadex column with 2 liters of 100 mM ammonium bicarbonate and then with 2 liters of 200 mM ammonium bicarbonate gave 5′-monophosphate (see Example 22) which was stable to 5′-nucleotidase.
…………………………………………

An enantiopure β-lactam with a suitably disposed electron withdrawing group on nitrogen, participated in a π-allylpalladium mediated reaction with 2,6-dichloropurine tetrabutylammonium salt to afford an advanced cis-1,4-substituted cyclopentenoid with both high regio- and stereoselectivity. This advanced intermediate was successfully manipulated to the total synthesis of (−)-Abacavir.
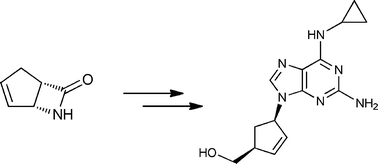
http://pubs.rsc.org/en/content/articlelanding/2012/ob/c2ob06775g#!divAbstract
………………………………….
http://www.google.com.ar/patents/EP2085397A1?cl=en
Example 1: Preparation of crystalline Form I of abacavir base using methanol as solvent
-
[0026]Abacavir (1.00 g, containing about 17% of dichloromethane) was dissolved in refluxing methanol (2.2 mL). The solution was slowly cooled to – 5 °C and, the resulting suspension, was kept at that temperature overnight under gentle stirring. The mixture was filtered off and dried under vacuum (7-10 mbar) at 40 °C for 4 hours to give a white solid (0.55 g, 66% yield, < 5000 ppm of methanol). The PXRD analysis gave the diffractogram shown in FIG. 1.
……………………………………..
http://www.google.com/patents/WO2008037760A1?cl=en
Abacavir, is the International Nonproprietary Name (INN) of {(1 S,4R)-4-[2- amino-6-(cyclopropylamino)-9H-purin-9-yl]-cyclopent-2-enyl}methanol, and CAS No. 136470-78-5. Abacavir sulfate is a potent selective inhibitor of HIV-1 and HIV-2, and can be used in the treatment of human immunodeficiency virus (HIV) infection.
The structure of abacavir hemisulfate salt corresponds to formula (I):
(I)
EP 434450-A discloses certain 9-substituted-2-aminopuhnes including abacavir and its salts, methods for their preparation, and pharmaceutical compositions using these compounds.
Different preparation processes of abacavir are known in the art. In some of them abacavir is obtained starting from an appropriate pyrimidine compound, coupling it with a sugar analogue residue, followed by a cyclisation to form the imidazole ring and a final introduction of the cyclopropylamino group at the 6 position of the purine ring. Pyrimidine compounds which have been identified as being useful as intermediates of said preparation processes include N-2-acylated abacavir intermediates such as N-{6- (cyclopropylamino)-9-[(1 R,4S)-4-(hydroxymethyl)cyclopent-2-enyl]-9H-purin- 2-yl}acetamide or N-{6-(cyclopropylamino)-9-[(1 R,4S)-4-
(hydroxymethyl)cyclopent-2-enyl]-9H-purin-2-yl}isobutyramide. The removal of the amino protective group of these compounds using acidic conditions is known in the art. According to Example 28 of EP 434450-A, the amino protective group of the N-{6-(cyclopropylamino)-9-[(1 R,4S)-4- (hydroxymethyl)cyclopent-2-enyl]-9H-purin-2-yl}isobutyramide is removed by stirring with 1 N hydrochloric acid for 2 days at room temperature. The abacavir base, after adjusting the pH to 7.0 and evaporation of the solvent, is finally isolated by trituration and chromatography. Then, it is transformed by reaction with an acid to the corresponding salt of abacavir. The main disadvantages of this method are: (i) the use of a strongly corrosive mineral acid to remove the amino protective group; (ii) the need of a high dilution rate; (iii) a long reaction time to complete the reaction; (iv) the need of isolating the free abacavir; and (v) a complicated chromatographic purification process.
Thus, despite the teaching of this prior art document, the research of new deprotection processes of a N-acylated {(1 S,4R)-4-[2-amino-6- (cyclopropylamino)-9H-purin-9-yl]-cyclopent-2-enyl}methanol is still an active field, since the industrial exploitation of the known process is difficult, as it has pointed out above. Thus, the provision of a new process for the removal of the amino protective group of a N-acylated {(1 S,4R)-4-[2-amino-6-
(cyclopropylamino)-9H-purin-9-yl]-cyclopent-2-enyl}methanol is desirable.
Example 1 : Preparation of abacavir hemisulfate
N-{6-(cyclopropylamino)-9-[(1 R,4S)-4-(hydroxymethyl)cyclopent-2-enyl]-9H- purin-2-yl}isobutyramide (6.56 g, 18.40 mmol) was slurried in a mixture of isopropanol (32.8 ml) and 10% solution of NaOH (36.1 ml, 92.0 mmol). The mixture was refluxed for 1 h. The resulting solution was cooled to 20-25 0C and tert-butyl methyl ether (32.8 ml) was added. The layers were separated and H2SO4 96% (0.61 ml, 11.03 mmol) was added dropwise to the organic layer. This mixture was cooled to 0-50C and the resulting slurry filtered off.
The solid was dried under vacuum at 40 0C. Abacavir hemisulfate (5.98 g, 97%) was obtained as a white powder.
Example 6: Preparation of abacavir
N-{6-(cyclopropylamino)-9-[(1 R,4S)-4-(hydroxymethyl)cyclopent-2-enyl]-9H- purin-2-yl}isobutyramide (1.0 g, 2.80 mmol) was slurried in a mixture of isopropanol (2 ml) and 10% solution of NaOH (1.1 ml, 2.80 mmol). The mixture was refluxed for 1 h. The resulting solution was cooled to 20-25 0C and tert-butyl methyl ether (2 ml) was added. The aqueous layer was discarded, the organic phase was cooled to 0-5 0C and the resulting slurry filtered off. The solid was dried under vacuum at 400C. Abacavir (0.62 g, 77%) was obtained as a white powder.
Example 7: Preparation of abacavir
N-{6-(cyclopropylamino)-9-[(1 R,4S)-4-(hydroxymethyl)cyclopent-2-enyl]-9H- purin-2-yl}isobutyramide (1.25 g, 3.51 mmol) was slurried in a mixture of isopropanol (2.5 ml) and 10% solution of NaOH (1.37 ml, 3.51 mmol). The mixture was refluxed for 1 h and concentrated to dryness. The residue was crystallized in acetone. Abacavir (0.47 g, 47%) was obtained as a white powder.
Example 8: Preparation of abacavir
N-{6-(cyclopropylamino)-9-[(1 R,4S)-4-(hydroxymethyl)cyclopent-2-enyl]-9H- purin-2-yl}isobutyramide (1.25 g, 3.51 mmol) was slurried in a mixture of isopropanol (2.5 ml) and 10% solution of NaOH (1.37 ml, 3.51 mmol). The mixture was refluxed for 1 h and concentrated to dryness. The residue was crystallized in acetonitrile. Abacavir (0.43 g, 43%) was obtained as a white powder.
Example 9: Preparation of abacavir
A mixture of N-{6-(cyclopropylamino)-9-[(1 R,4S)-4-(hydroxymethyl)cyclopent- 2-enyl]-9H-purin-2-yl}isobutyramide (10 g, 28 mmol), isopropanol (100 ml) and 10% solution of NaOH (16.8 ml, 42 mmol) was refluxed for 1 h. The resulting solution was cooled to 20-25 0C and washed several times with 25% solution of NaOH (10 ml). The wet organic layer was neutralized to pH 7.0-7.5 with 17% hydrochloric acid and it was concentrated to dryness under vacuum. The residue was crystallized in ethyl acetate (150 ml) to afford abacavir (7.2 g, 90%).
Example 10: Preparation of abacavir
A mixture of N-{6-(cyclopropylamino)-9-[(1 R,4S)-4-(hydroxymethyl)cyclopent- 2-enyl]-9H-purin-2-yl}isobutyramide (10 g, 28 mmol), isopropanol (100 ml) and 10% solution of NaOH (16.8 ml, 42 mmol) was refluxed for 1 h. The resulting solution was cooled to 20-25 0C and washed several times with 25% solution of NaOH (10 ml). The wet organic layer was neutralized to pH 7.0-7.5 with 17% hydrochloric acid and it was concentrated to dryness under vacuum. The residue was crystallized in acetone (300 ml) to afford abacavir (7.0 g, 88%).
…………………………………
http://www.google.com/patents/WO2004089952A1?cl=en
Abacavir of formula (1) :
or (1 S,4R)-4-[2-Amino-6-(cyclopropylamino)-9H-purin-9-yl]-2-cyclopentene-1 – methanol and its salts are nucleoside reverse transcriptase inhibitors. Abacavir sulfate is a nucleoside reverse transcriptase inhibitor and used in the treatment of human immunodeficiency virus infection. Abacavir sulfate and related compounds and their therapeutic uses are disclosed in US 5,034,394.
Crystalline forms of abacavir sulfate have not been reported in the literature. Moreover, the processes described in the literature do not produce abacavir sulfate in a stable, well-defined and reproducible crystalline form. It has now been discovered that abacavir sulfate can be prepared in three stable, well-defined and consistently reproducible crystalline forms.
Example 1
Abacavir free base (3.0 gm, obtained by the process described in example 21 of US 5,034,394) is dissolved in ethyl acetate (15 ml) and cone, sulfuric acid (0.3 ml) is added to the solution. Then the contents are stirred for 3 hours at 20°C and filtered to give 3.0 gm of form I abacavir sulfate. Example 2 Abacavir free base (3.0 gm) is dissolved in acetone (20 ml) and cone, sulfuric acid (0.3 ml) is added to the solution. Then the contents are stirred for 6 hours at 25°C and filtered to give 2.8 gm of form I abacavir sulfate.
Example 3 Abacavir free base (3.0 gm) is dissolved in acetonitrile (15 ml) and sulfuric acid (0.3 ml) is added to the solution. Then the contents are stirred for 2 hours at 25°C and the separated solid is filtered to give 3.0 gm of form II abacavir sulfate.
Example 4 Abacavir free base (3.0 gm) is dissolved in methyl tert-butyl ether (25 ml) and sulfuric acid (0.3 ml) is added to the solution. Then the contents are stirred for 1 hours at 25°C and the separated solid is filtered to give 3.0 gm of form II abacavir sulfate.
Example 5 Abacavir free base (3.0 gm) is dissolved in methanol (15 ml) and sulfuric acid (0.3 ml) is added to the solution. The contents then are cooled to 0°C and diisopropyl ether (15 ml) is added. The reaction mass is stirred for 2 hours at about 25°C and the separated solid is filtered to give 3.0 gm of form III abacavir sulfate
…………………………….
http://www.google.com.ar/patents/WO1999021861A1?cl=en
The present invention relates to a new process for the preparation of the chiral nucleoside analogue (1S, 4R)-4-[2-amino-6-(cyclopropylamino)-9H purin-9-yl]-2- cyclopentene-1 -methanol (compound of Formula (I)).
The compound of formula (I) is described as having potent activity against human immunodeficiency virus (HIV) and hepatitis B virus (HBV) in EPO34450.
Results presented at the 34th Interscience Conference on Antimicrobial Agents and Chemotherapy (October 4-7, 1994) demonstrate that the compound of formula I has significant activity against HIV comparable to, and if not better than, some current anti HIV drugs, such as zidovudine and didanosine.
Currently the compound of Formula (I) is undergoing clinical investigation to determine its safety and efficacy in humans. Therefore, there exists at the present time a need to supply large quantities of this compound for use in clinical trials.
Current routes of synthesising the compound of formula (I) involve multiple steps and are relatively expensive. It will be noted that the compound has two centres of asymmetry and it is essential that any route produces the compound of formula (I) substantially free of the corresponding enantiomer, preferably the compound of formula (I) is greater than 95% w/w free of the corresponding enantiomer.
Processes proposed for the preparation of the compound of formula (I) generally start from a pyrimidine compound, coupling with a 4-amino-2-cyclopentene-1- methanol analogue, cyciisation to form the imidazole ring and then introduction of the cyclopropylamine group into the 6 position of the purine, such routes include those suggested in EPO434450 and WO9521161. Essentially both routes disclosed in the two prior patent applications involve the following steps:-
(i) coupling (1S, 4R)-4-amino-2-cyclopentene-1 -methanol to N-(4,6-dichloro-5- formamido-2-pyrimidinyl) acetamide or a similar analogue thereof, for example N- (2-amino-4,6-dichloro-5-pyrimidinyl) formamide;
(ii) ring closure of the resultant compound to form the intermediate (1 S, 4R)-4- (2-amino-6-chloro-9H-purin-9-yl)-2-cyclopentene-1 -methanol;
(iii) substituting the halo group by a cyclopropylamino group on the 6 position of the purine ring.
The above routes are multi-step processes. By reducing the number of processing steps significant cost savings can be achieved due to the length of time to manufacture the compound being shortened and the waste streams minimised.
An alternative process suggested in the prior art involves the direct coupling of carbocyclic ribose analogues to the N atom on the 9 position of 2-amino-6-chloro purine. For example WO91/15490 discloses a single step process for the formation of the (1S, 4R)- 4-(2-amino-6-chloro-9H-purin-9-yl)-2-cyclopentene-1- methanol intermediate by reacting (1S, 4R)-4-hydroxy-2-cyclopentene-1 -methanol, in which the allylic hydroxyl group has been activated as an ester or carbonate and the other hydroxyl group has a blocking group attached (for example 1 ,4- bis- methylcarbonate) with 2-amino-6-chloropurine.
However we have found that when synthesising (1S, 4R)-4-(2-amino-6-chloro-9H- purin-9-yl)-2-cyclopentene-1- methanol by this route a significant amount of an N- 7 isomer is formed (i.e. coupling has occurred to the nitrogen at the 7- position of the purine ring) compared to the N-9 isomer desired. Further steps are therefore required to convert the N-7 product to the N-9 product, or alternatively removing the N-7 product, adding significantly to the cost. We have found that by using a transition metal catalysed process for the direct coupling of a compound of formula (II) or (III),
Example 1 (1 S. 4R)-4-[2-Amino-6-(cvclopropylamino)-9H purin-9-vπ-2-cvclopentene-1 – methanol
Triphenylphosphine (14mg) was added, under nitrogen, to a mixture of (1S.4R)- 4-hydroxy-2-cyclopentene -1 -methanol bis(methylcarbonate) (91 mg), 2-amino-6- (cyclopropylamino) purine (90mg), tris(dibenzylideneacetone)dipalladium (12mg) and dry DMF (2ml) and the resulting solution stirred at room temperature for 40 min.
The DMF was removed at 60° in vacuo and the residue partitioned between ethyl acetate (25ml.) and 20% sodium chloride solution (10ml.). The ethyl acetate solution was washed with 20% sodium chloride (2x12ml.) and with saturated sodium chloride solution, then dried (MgSO4) and the solvent removed in vacuo.
The residue was dissolved in methanol (10ml.), potassium carbonate (17mg) added and the mixture stirred under nitrogen for 15h.
The solvent was removed in vacuo and the residue chromatographed on silica gel
(Merck 9385), eluting with dichloromethane-methanol [(95:5) increasing to (90:10)] to give the title compound (53mg) as a cream foam.
δ(DMSO-d6): 7.60 (s.1 H); 7.27 (s,1 H); 6.10 (dt,1 H); 5.86 (dt, 1 H); 5.81 (s,2H); 5.39 (m,1H); 4.75 (t,1H); 3.44 (t,2H); 3.03 (m, 1H): 2.86 (m,1H);2.60 (m,1H); 1.58 (dt, 1 H); 0.65 (m, 2H); 0.57 (m,2H).
TLC SiO2/CHCI3-MeOH (4:1 ) Rf 0.38; det. UN., KMnO4
Trade Names
| Page | Trade name | Manufacturer |
|---|---|---|
| Germany | Kiveksa | GlaxoSmithKline |
| Trizivir | -»- | |
| Ziagen | -»- | |
| France | Kiveksa | -»- |
| Trizivir | -»- | |
| Ziagen | -»- | |
| United Kingdom | Kiveksa | -»- |
| Trizivir | -»- | |
| Ziagen | -»- | |
| Italy | Trizivir | -»- |
| Ziagen | -»- | |
| Japan | Épzikom | -»- |
| Ziagen | -»- | |
| USA | Épzikom | -»- |
| Trizivir | -»- | |
| Ziagen | -»- | |
| Ukraine | Virol | Ranbaksi Laboratories Limited, India |
| Ziagen | GlaksoSmitKlyayn Inc.., Canada | |
| Abamun | Tsipla Ltd, India | |
| Abacavir sulfate | Aurobindo Pharma Limited, India |
Formulations
-
Oral solution 20 mg / ml;
-
Tablets of 300 mg (as the sulfate);
-
Trizivir tablets 300 mg – abacavir in fixed combination with 150 mg of lamivudine and 300 mg zidovudine
ZIAGEN is the brand name for abacavir sulfate, a synthetic carbocyclic nucleoside analogue with inhibitory activity against HIV-1. The chemical name of abacavir sulfate is (1S,cis)-4-[2-amino-6-(cyclopropylamino)-9H-purin-9-yl]-2-cyclopentene-1-methanol sulfate (salt) (2:1). Abacavir sulfate is the enantiomer with 1S, 4R absolute configuration on the cyclopentene ring. It has a molecular formula of (C14H18N6O)2•H2SO4 and a molecular weight of 670.76 daltons. It has the following structural formula:
 |
Abacavir sulfate is a white to off-white solid with a solubility of approximately 77 mg/mL in distilled water at 25°C. It has an octanol/water (pH 7.1 to 7.3) partition coefficient (log P) of approximately 1.20 at 25°C.
ZIAGEN Tablets are for oral administration. Each tablet contains abacavir sulfate equivalent to 300 mg of abacavir as active ingredient and the following inactive ingredients: colloidal silicon dioxide, magnesium stearate, microcrystalline cellulose, and sodium starch glycolate. The tablets are coated with a film that is made of hypromellose, polysorbate 80, synthetic yellow iron oxide, titanium dioxide, and triacetin.
ZIAGEN Oral Solution is for oral administration. Each milliliter (1 mL) of ZIAGEN Oral Solution contains abacavir sulfate equivalent to 20 mg of abacavir (i.e., 20 mg/mL) as active ingredient and the following inactive ingredients: artificial strawberry and banana flavors, citric acid (anhydrous), methylparaben and propylparaben (added as preservatives), propylene glycol, saccharin sodium, sodium citrate (dihydrate), sorbitol solution, and water.
In vivo, abacavir sulfate dissociates to its free base, abacavir. All dosages for ZIAGEN are expressed in terms of abacavir.
History
Abacavir was approved by the Food and Drug Administration (FDA) on December 18, 1998 and is thus the fifteenth approved antiretroviral drug in the United States. Its patent expired in the United States on 2009-12-26.
Links
-
US 5 089 500 (Burroughs Wellcome; 18.2.1992; GB-prior. 27.6.1988).
-
Synthesis a)
-
EP 434 450 (Wellcome Found .; 26.6.1991; appl. 21.12.1990; prior-USA. 22.12.1989).
-
Crimmins, MT et al .: J. Org. Chem. (JOCEAH) 61 4192 (1996).
-
EP 1 857 458 (Solmag; appl. 5.5.2006).
-
EP 424 064 (Enzymatix; appl. 24.4.1991; GB -prior. 16.10.1989).
-
U.S. 6 340 587 (Beecham SMITHKLINE; 22.1.2002; appl. 20.8.1998; GB -prior. 22.8.1997).
-
-
Синтез b)
-
Olivo, HF et al .: J. Chem. Soc., Perkin Trans. 1 (JCPRB4) 1998, 391.
-
-
Preparation c)
-
U.S. 5 034 394 (Wellcome Found .; 23.7.1991; appl. 22.12.1989; GB -prior. 27.6.1988).
-
-
Synthesis d)
-
WO 9 924 431 (Glaxo; appl. 12.11.1998; WO-prior. 12.11.1997).
-
| WO2008037760A1 * | Sep 27, 2007 | Apr 3, 2008 | Esteve Quimica Sa | Process for the preparation of abacavir |
| EP1905772A1 * | Sep 28, 2006 | Apr 2, 2008 | Esteve Quimica, S.A. | Process for the preparation of abacavir |
| US8183370 | Sep 27, 2007 | May 22, 2012 | Esteve Quimica, Sa | Process for the preparation of abacavir |
| EP0434450A2 | 21 Dec 1990 | 26 Jun 1991 | The Wellcome Foundation Limited | Therapeutic nucleosides |
| EP0741710A1 | 3 Feb 1995 | 13 Nov 1996 | The Wellcome Foundation Limited | Chloropyrimide intermediates |
| WO1998052949A1 | 14 May 1998 | 26 Nov 1998 | Glaxo Group Ltd | Carbocyclic nucleoside hemisulfate and its use in treating viral infections |
| WO1999021861A1 | 24 Oct 1997 | 6 May 1999 | Glaxo Group Ltd | Process for preparing a chiral nucleoside analogue |
| WO1999039691A2 * | 4 Feb 1999 | 12 Aug 1999 | Brooks Nikki Thoennes | Pharmaceutical compositions |
| WO2008037760A1 * | 27 Sep 2007 | 3 Apr 2008 | Esteve Quimica Sa | Process for the preparation of abacavir |
References
- Jump up^ SFGate.com
- Jump up^ “WHO Model List of EssentialMedicines”. World Health Organization. October 2013. Retrieved 22 April 2014.
- Jump up^ https://online.epocrates.com/noFrame/showPage.do?method=drugs&MonographId=2043&ActiveSectionId=5
- Jump up^ Mallal, S., Phillips, E., Carosi, G. et al. (2008). “HLA-B*5701 screening for hypersensitivity to abacavir”. New England Journal of Medicine 358: 568–579.doi:10.1056/nejmoa0706135.
- Jump up^ Rauch, A., Nolan, D., Martin, A. et al. (2006). “Prospective genetic screening decreases the incidence of abacavir hypersensitivity reactions in the Western Australian HIV cohort study”. Clinical Infectious Diseases 43: 99–102. doi:10.1086/504874.
- Jump up^ Heatherington et al. (2002). “Genetic variations in HLA-B region and hypersensitivity reactions to abacavir”. Lancet 359: 1121–1122.
- Jump up^ Mallal et al. (2002). “Association between presence of HLA*B5701, HLA-DR7, and HLA-DQ3 and hypersensitivity to HIV-1 reverse-transcriptase inhibitor abacavir”. Lancet359: 727–732. doi:10.1016/s0140-6736(02)07873-x.
- Jump up^ Rotimi, C.N.; Jorde, L.B. (2010). “Ancestry and disease in the age of genomic medicine”. New England Journal of Medicine 363: 1551–1558.
- Jump up^ Phillips, E., Mallal, S. (2009). “Successful translation of pharmacogenetics into the clinic”. Molecular Diagnosis & Therapy 13: 1–9. doi:10.1007/bf03256308.
- Jump up^ Phillips, E., Mallal S. (2007). “Drug hypersensitivity in HIV”. Current Opinion in Allergy and Clinical Immunology 7: 324–330. doi:10.1097/aci.0b013e32825ea68a.
- Jump up^http://www.fda.gov/drugs/drugsafety/postmarketdrugsafetyinformationforpatientsandproviders/ucm123927.htmAccessed November 29, 2013.
- Jump up^ http://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=ca73b519-015a-436d-aa3c-af53492825a1
- Jump up^ Martin MA, Hoffman JM, Freimuth RR et al. (May 2014). “Clinical Pharmacogenetics Implementation Consortium Guidelines for HLA-B Genotype and Abacavir Dosing: 2014 update”. Clin Pharmacol Ther. 95 (5): 499–500. doi:10.1038/clpt.2014.38.PMC 3994233. PMID 24561393.
- Jump up^ Swen JJ, Nijenhuis M, de Boer A et al. (May 2011). “Pharmacogenetics: from bench to byte–an update of guidelines”. Clin Pharmacol Ther. 89 (5): 662–73.doi:10.1038/clpt.2011.34. PMID 21412232.
- Jump up^ Shear, N.H., Milpied, B., Bruynzeel, D.P. et al. (2008). “A review of drug patch testing and implications for HIV clinicians”. AIDS 22: 999–1007.doi:10.1097/qad.0b013e3282f7cb60.
- Jump up^ http://www.drugs.com/fda/abacavir-ongoing-safety-review-possible-increased-risk-heart-attack-12914.html Accessed November 29, 2013.
- Jump up^ Ding X, Andraca-Carrera E, Cooper C et al. (December 2012). “No association of abacavir use with myocardial infarction: findings of an FDA meta-analysis”. J Acquir Immune Defic Syndr. 61 (4): 441–7. doi:10.1097/QAI.0b013e31826f993c.PMID 22932321.
- Illing PT et al. 2012, Nature, doi:10.1038/nature11147
External links
- Full Prescribing Information
- Abacavir pathway on PharmGKB
- Abacavir dosing guidelines from the Clinical Pharmacogenetics Implementation Consortium (CPIC)
- Abacavir dosing guidelines from the Dutch Pharmacogenetics Working Group (DPWG)
EXTRA INFO
How to obtain carbocyclic nucleosides?
Carbocyclic nucleosides are synthetically the most challenging class of nucleosides, requiring multi-step and often elaborate synthetic pathways to introduce the necessary stereochemistry. There are two main strategies for the preparation of carbocyclic nucleosides. In the linear approach a cyclopentylamine is used as starting material and the heterocycle is built in a stepwise manner (see Scheme 1).
Scheme 1: Linear approach for the synthesis of abacavir.[5]
The more flexible strategy is a convergent approach: a functionalized carbocyclic moiety is condensed with a heterocycle rapidly leading to a variety of carbocyclic nucleosides. Initially, we started our syntheses from cyclopentadiene 1 that is deprotonated and alkylated with benzyloxymethyl chloride to give the diene 2. This material is converted by a hydroboration into cyclopentenol 3 or isomerized into two thermodynamically more stable cyclopentadienes 4a,b. With the protection and another hydroboration step to 5 we gain access to an enantiomerically pure precursor for the synthesis of a variety of carbocyclic 2’-deoxynucleosides e.g.:carba-dT, carba-dA or carba-BVDU.[6] The isomeric dienes 4a,b were hydroborated to the racemic carbocyclic moiety 6.

Scheme 2: Convergent approach for the synthesis of carba-dT.
The asymmetric synthesis route and the racemic route above are short and efficient ways to diverse carbocyclic D- or L-nucleosides (Scheme 2). Different heterocycles can be condensed to these precursors leading to carbocyclic purine- and pyrimidine-nucleosides. Beside α- and β-nucleosides, carbocyclic epi– andiso-nucleosides in the 2’-deoxyxylose form were accessable.[7]
What else is possible? The racemic cyclopentenol 6 can be coupled by a modified Mitsunobu-reaction.Moreover, this strategy offers the possibility of synthesizing new carbocyclic nucleosides by functionalizing the double bond before or after introduction of the nucleobase (scheme 3).[8]

Scheme 3: Functionalized carbocyclic nucleosides based on cyclopentenol 6.
Other interesting carbocyclic precursors like cyclopentenol 7 can be used to synthesize several classes of carbocyclic nucleoside analogues, e.g.: 2’,3’-dideoxy-2’,3’-didehydro nucleosides (d4-nucleosides), 2’,3’-dideoxynucleosides (ddNs), ribonucleosides, bicyclic nucleosides or even 2’-fluoro-nucleosides.

Scheme 4: Functionalized carbocyclic thymidine analogues based on cyclopentenol 7.
[1] V. E. Marquez, T. Ben-Kasus, J. J. Barchi, K. M. Green, M .C. Nicklaus, R. Agbaria, J. Am. Chem. Soc.2004,126, 543.
[2] A. D. Borthwick, K. Biggadike, Tetrahedron 1992, 48, 571.
[3] H. Bricaud, P. Herdewijn, E. De Clercq, Biochem. Pharmacol. 1983, 3583.
[4] P. L. Boyer, B. C. Vu, Z. Ambrose, J. G. Julias, S. Warnecke, C. Liao, C. Meier, V. E. Marquez, S. H. Hughes, J. Med. Chem. 2009, 52, 5356.
[5] S. M. Daluge, M. T. Martin, B. R. Sickles, D. A. Livingston, Nucleosides, Nucleotides Nucleic Acids 2000,19, 297.
[6] O. R. Ludek, C. Meier, Synthesis 2003, 2101.
[7] O. R. Ludek, T. Kraemer, J. Balzarini, C. Meier, Synthesis 2006, 1313.
[8] M. Mahler, B. Reichardt, P. Hartjen, J. van Lunzen, C. Meier, Chem. Eur. J. 2012, 18, 11046-11062.

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO


































