| Record ID | Title | Status | Phase |
|---|---|---|---|
| NCT03041311 | Carboplatin, Etoposide, and Atezolizumab With or Without Trilaciclib (G1T28), a CDK 4/6 Inhibitor, in Extensive Stage Small Cell Lung Cancer (SCLC) | Recruiting | 2 |
| NCT02978716 | Trilaciclib (G1T28), a CDK 4/6 Inhibitor, in Combination With Gemcitabineand Carboplatin in Metastatic Triple Negative Breast Cancer (mTNBC) | Recruiting | 2 |
| NCT02514447 | Trilaciclib (G1T28), a CDK 4/6 Inhibitor, in Patients With Previously Treated Extensive Stage SCLC Receiving Topotecan Chemotherapy | Recruiting | 2 |
| NCT02499770 | Trilaciclib (G1T28), a CDK 4/6 Inhibitor, in Combination With Etoposide and Carboplatin in Extensive Stage Small Cell Lung Cancer (SCLC) | Active, not recruiting | 2 |
TRILACICLIB, G1T28
Trilaciclib
update 2021/2/12 US FDA APPROVED COSELA
- Molecular FormulaC24H30N8O
- Average mass446.548 Da
- G1T 28
- 7′,8′-Dihydro-2′-[[5-(4-methyl-1-piperazinyl)-2-pyridinyl]amino]spiro[cyclohexane-1,9′(6’H)-pyrazino[1′,2′:1,5]pyrrolo[2,3-d]pyrimidin]-6′-one
- 2′-[[5-(4-Methylpiperazin-1-yl)pyridin-2-yl]amino}-7′,8′-dihydro-6’H-spiro[cyclohexane-1,9′-pyrazino[1′,2′:1,5]pyrrolo[2,3-d]pyrimidin]-6′-one
Reduction of Chemotherapy-Induced Myelosuppression
Trilaciclib dihydrochloride
1977495-97-8
In phase II clinical development as a chemoprotectant at G1 Therapeutics for first- or second-line treatment in patients with metastatic triple negative breast cancer, in combination with gemcitabine and carboplatin

PATENT, WO 2014144326, Compound 89 (also referred to as Compound T)
| WO2014144847A3 | |
| Inventors | Norman E. Sharpless, Jay Copeland Strum, John Emerson Bisi, Patrick Joseph Roberts, Francis Xavier Tavares |
| Applicant | G1 Therapeutics, Inc. |
| Norman Sharpless | |
|---|---|
 |
|
| Born | Norman Edward Sharpless September 20, 1966 Greensboro, North Carolina |
| Nationality | American |
| Other names | Ned Sharpless |
| Occupation | Director, Lineberger Comprehensive Cancer Center Founder, G1 Therapeutics ($GTHX) |
| Notable work | Wellcome Distinguished Professor, American Society of Clinical Investigation Member, Association of American Cancer Institute board of directors, |
NCI Director Dr. Norman E. Sharpless

NCI Director Dr. Norman E. Sharpless, Credit: National Institutes of Health
Norman E. “Ned” Sharpless, M.D., was officially sworn in as the 15th director of the National Cancer Institute (NCI) on October 17, 2017. Prior to his appointment, Dr. Sharpless served as the director of the University of North Carolina (UNC) Lineberger Comprehensive Cancer Center, a position he held since January 2014.
Dr. Sharpless was a Morehead Scholar at UNC–Chapel Hill and received his undergraduate degree in mathematics. He went on to pursue his medical degree from the UNC School of Medicine, graduating with honors and distinction in 1993. He then completed his internal medicine residency at the Massachusetts General Hospital and a hematology/oncology fellowship at Dana-Farber/Partners Cancer Care, both of Harvard Medical School in Boston.
After 2 years on the faculty at Harvard Medical School, he joined the faculty of the UNC School of Medicine in the Departments of Medicine and Genetics in 2002. He became the Wellcome Professor of Cancer Research at UNC in 2012.
Dr. Sharpless is a member of the Association of American Physicians as well as the American Society for Clinical Investigation (ASCI), the nation’s oldest honor society for physician–scientists, and served on the ASCI council from 2011 to 2014. Dr. Sharpless was an associate editor of Aging Cell and deputy editor of the Journal of Clinical Investigation. He has authored more than 150 original scientific papers, reviews, and book chapters, and is an inventor on 10 patents. He cofounded two clinical-stage biotechnology companies: G1 Therapeutics and HealthSpan Diagnostics.
In addition to serving as director of NCI, Dr. Sharpless continues his research in understanding the biology of the aging process that promotes the conversion of normal self-renewing cells into dysfunctional cancer cells. Dr. Sharpless has made seminal contributions to the understanding of the relationship between aging and cancer, and in the preclinical development of novel therapeutics for melanoma, lung cancer, and breast cancer.
Synthesis
WO 2016040858


Trilaciclib (G1T28)
Trilaciclib is a potential first-in-class short-acting CDK4/6 inhibitor in development to preserve hematopoietic stem cells and enhance immune system function during chemotherapy. Trilaciclib is administered intravenously prior to chemotherapy and has the potential to significantly improve treatment outcomes.
G1 is currently evaluating trilaciclib in four Phase 2 clinical trials: three studies in patients with small-cell lung cancer (SCLC), and one study in patients with triple-negative breast cancer (TNBC). Preliminary data from the SCLC trials were presented at the American Society of Clinical Oncology 2017 Annual Meeting and at the 2016 World Conference on Lung Cancer.
Data from a Phase 1 trial in healthy volunteers were presented at the American Society of Clinical Oncology 2015 Annual Meeting and published in Science Translational Medicine. Trilacicilib has been extensively studied in animals; these preclinical data have been presented at several scientific meetings and published in Molecular Cancer Therapeutics, Science Translational Medicine, and Cancer Discovery.
Trilaciclib is a small molecule, competitive inhibitor of cyclin dependent kinases 4 and 6 (CDK4/6), with potential antineoplastic and chemoprotective activities. Upon intravenous administration, trilaciclib binds to and inhibits the activity of CDK4/6, thereby blocking the phosphorylation of the retinoblastoma protein (Rb) in early G1. This prevents G1/S phase transition, causes cell cycle arrest in the G1 phase, induces apoptosis, and inhibits the proliferation of CDK4/6-overexpressing tumor cells. In patients with CDK4/6-independent tumor cells, G1T28 may protect against multi-lineage chemotherapy-induced myelosuppression (CIM) by transiently and reversibly inducing G1 cell cycle arrest in hematopoietic stem and progenitor cells (HSPCs) and preventing transition to the S phase. This protects all hematopoietic lineages, including red blood cells, platelets, neutrophils and lymphocytes, from the DNA-damaging effects of certain chemotherapeutics and preserves the function of the bone marrow and the immune system. CDKs are serine/threonine kinases involved in the regulation of the cell cycle and may be overexpressed in certain cancer cell types. HSPCs are dependent upon CDK4/6 for proliferation.
Trilaciclib (G1T28) is a CDK4/6 inhibitor in phase II clinical development as a chemoprotectant at G1 Therapeutics for first- or second-line treatment in patients with metastatic triple negative breast cancer, in combination with gemcitabine and carboplatin. Also, phase II trials are ongoing in newly diagnosed, treatment-naive small-cell lung cancer patients, in combination with carboplatin, etoposide, and atezolizumab and phase I trials in previously treated small-cell lung cancer patients, in combination with topotecan.
U.S. Patent Nos. 8,822,683; 8,598,197; 8,598,186, 8,691,830, 8,829,102, 8,822,683, 9, 102,682, 9,499,564, 9,481,591, and 9,260,442, filed by Tavares and Strum and assigned to Gl Therapeutics describe a class of N-(heteroaryl)-pyrrolo[3,2-d]pyrimidin-2-amine cyclin dependent kinase inhibitors including those of the formula with variables as defined therein):

U.S. Patent Nos. 9,464,092, 9,487,530, and 9,527,857 which are also assigned to Gl Therapeutics describe the use of the above pyrimidine-based agents in the treatment of cancer.
These patents provide a general synthesis of the compounds that is based on a coupling reaction of a fused chloropyrimidine with a heteroaryl amine to form the central disubstituted amine. Such coupling reactions are sometimes referred to as Buchwald coupling (see WO Ί56 paragraph 127; reference WO 2010/020675). The lactam of the fused chloropyrimidine, for example, a 2-chloro-spirocyclo-pyrrolo[2,3-d]pyrimidine-one such as Intermediate K as shown below can be prepared by dehydration of the corresponding carboxylic acid. The reported process to prepare intermediate IK requires seven steps.

(Intermediate IK; page 60, paragraph 215 of WO Ί56)
WO 2013/148748 (U.S. S.N. 61/617,657) entitled “Lactam Kinase Inhibitors” filed by Tavares, and also assigned to Gl Therapeutics likewise describes the synthesis of N-(heteroaryl)-pyrrolo[3,2-d]pyrimidin-2-amines via the coupling reaction of a fused chloropyrimidine with a heteroaryl amine to form the central disubstituted amine.
WO 2013/163239 (U.S. S.N. 61/638,491) “Synthesis of Lactams” describes a method for the synthesis of this class of compounds with the variation that in the lactam preparation step, a carboxylic acid can be cyclized with a protected amine in the presence of a strong acid and a dehydrating agent, which can be together in one moiety as a strong acid anhydride. The purported improvement is that cyclization can occur without losing the protecting group on the amine before cyclization. The typical leaving group is “tBOC” (t-butoxycarbonyl). The application teaches (page 2 of WO 2013/163239) that the strong acid is, for example, trifluoroacetic acid anhydride, tribromoacetic acid anhydride, trichloroacetic acid anhydride or mixed anhydrides. An additional step may be necessary to take off the N-protecting group. The dehydrating agent can be a carbodiimide-based compound such as DCC (Ν,Ν-dicyclohexylcarbodiimide), EDC (l-ethyl-3-(3-dimethylaminopropyl)carbodiimide, or DIC (Ν,Ν-diisopropylcarbodiimide). DCC and DIC are in the same class of reagents-carbodiimides. DIC is sometimes considered better because it is a liquid at room temperature, which facilitates reactions.
WO 2015/061407 filed by Tavares and licensed to Gl Therapeutics also describes the synthesis of these compounds via the coupling of a fused chloropyrimidine with a heteroaryl amine to form the central disubstituted amine. WO ‘407 focuses on the lactam production step and in particular describes that the fused lactams of these compounds can be prepared by treating the carboxylic acid with an acid and a dehydrating agent in a manner that a leaving group on the amine is not removed during the amide-forming ring closing step.
Other publications that describe compounds of this general class include the following. WO 2014/144326 filed by Strum et al. and assigned to Gl Therapeutics describes compounds and methods for protection of normal cells during chemotherapy using pyrimidine based CDK4/6 inhibitors. WO 2014/144596 filed by Strum et al. and assigned to Gl Therapeutics describes compounds and methods for protection of hematopoietic stem and progenitor cells against ionizing radiation using pyrimidine based CDK4/6 inhibitors. WO 2014/144847 filed by Strum et al. and assigned to Gl Therapeutics describes HSPC-sparing treatments of abnormal cellular proliferation using pyrimidine based CDK4/6 inhibitors. WO2014/144740 filed by Strum et al. and assigned to Gl Therapeutics describes highly active anti -neoplastic and anti-proliferative pyrimidine based CDK 4/6 inhibitors. WO 2015/161285 filed by Strum et al. and assigned to Gl Therapeutics describes tricyclic pyrimidine based CDK inhibitors for use in radioprotection. WO 2015/161287 filed by Strum et al. and assigned to Gl Therapeutics describes analogous tricyclic pyrimidine based CDK inhibitors for the protection of cells during chemotherapy. WO 2015/161283 filed by Strum et al. and assigned to Gl Therapeutics describes analogous tricyclic pyrimidine based CDK inhibitors for use in HSPC-sparing treatments of RB-positive abnormal cellular proliferation. WO 2015/161288 filed by Strum et al. and assigned to Gl Therapeutics describes analogous tricyclic pyrimidine based CDK inhibitors for use as anti -neoplastic and anti-proliferative agents. WO 2016/040858 filed by Strum et al. and assigned to Gl Therapeutics describes the use of combinations of pyrimidine based CDK4/6 inhibitors with other anti-neoplastic agents. WO 2016/040848 filed by Strum et al. and assigned to Gl Therapeutics describes compounds and methods for treating certain Rb-negative cancers with CDK4/6 inhibitors and topoisomerase inhibitors.
Other biologically active fused spirolactams and their syntheses are described, for example, in the following publications. Griffith, D. A., et al. (2013). “Spirolactam-Based Acetyl-CoA Carboxylase Inhibitors: Toward Improved Metabolic Stability of a Chromanone Lead Structure.” Journal of Medicinal Chemistry 56(17): 7110-7119, describes metabolically stable spirolactams wherein the lactam resides on the fused ring for the inhibition of acetyl-CoA carboxylase. WO 2013/169574 filed by Bell et al. describes aliphatic spirolactams as CGRP receptor antagonists wherein the lactam resides on the spiro ring. WO 2007/061677 filed by Bell et al. describes aryl spirolactams as CGRP receptor antagonists wherein the lactam resides on the spiro ring. WO 2008/073251 filed by Bell et al. describes constrained spirolactam compounds wherein the lactam resides on the spiro ring as CGRP receptor antagonists. WO 2006/031606 filed by Bell et al. describes carboxamide spirolactam compounds wherein the spirolactam resides on the spiro ring as CGRP receptor antagonists. WO 2006/031610, WO 2006/031491, and WO 2006/029153 filed by Bell et al. describe anilide spirolactam compounds wherein the spirolactam resides on the spiro ring; WO 2008/109464 filed by Bhunai et al. describes spirolactam compounds wherein the lactam resides on the spiro ring which is optionally further fused.
Given the therapeutic activity of selected N-(heteroaryl)-pyrrolo[3,2-d]pyrimidin-2-amines, it would be useful to have additional methods for their preparation. It would also be useful to have new intermediates that can be used to prepare this class of compounds.
PATENT
WO 2014144596
PATENT
Compound 89 (also referred to as Compound T)
| WO2014144847A3 | |
| Inventors | Norman E. Sharpless, Jay Copeland Strum, John Emerson Bisi, Patrick Joseph Roberts, Francis Xavier Tavares |
| Applicant | G1 Therapeutics, Inc. |
EXAMPLES
Intermediates B, E, K, L, 1A, IF and 1CA were synthesized according to US 8,598,186 entitled CDK Inhibitors to Tavares, F.X. and Strum, J.C..
The patents WO 2013/148748 entitled Lactam Kinase Inhibitors to Tavares, F.X., WO 2013/163239 entitled Synthesis of Lactams to Tavares, F.X., and US 8,598,186 entitled CDK Inhibitors to Tavares, F.X. and Strum, J.C. are incorporated by reference herein in their entirety. Example 1
Synthesis of tert-butyl N- [2- [(5-bromo-2-chloro-pyrimidin-4yl)amino] ethyl] carbamate, Compound 1
To a solution of 5-bromo-2,4-dichloropyrimidine (3.2 g, 0.0135 mol) in ethanol (80 mL) was added Hunig’s base (3.0 mL) followed by the addition of a solution of N-(tert- butoxycarbonyl)-l,2-diaminoethane (2.5 g, 0.0156 mole) in ethanol (20 mL). The contents were stirred overnight for 20 hrs. The solvent was evaporated under vacuum. Ethyl acetate (200 mL) and water (100 mL) were added and the layers separated. The organic layer was dried with magnesium sulfate and then concentrated under vacuum. Column chromatography on silica gel using hexane/ethyl acetate (0- 60%) afforded tert-butyl N-[2-[(5-bromo-2-chloro-pyrimidin-4- yl)amino]ethyl]carbamate. 1HNMR (d6-DMSO) δ ppm 8.21 (s, 1H), 7.62 (brs, 1H), 7.27 (brs, 1H), 3.39 (m, 2H), 3.12 (m, 2H), 1.34 (s, 9H). LCMS (ESI) 351 (M + H).
Example 2
Synthesis of tert-butyl N-[2-[[2-chloro-5-(3,3-diethoxyprop-l-ynyl)pyrimidin-4- yl] amino] ethyl] carbamate, Compound 2
To tert-butyl N-[2-[(5-bromo-2-chloro-pyrimidin-4-yl)amino]ethyl]carbamate (1.265 g, 6 mmol) in THF (10 mL) was added the acetal (0.778 mL, 5.43 mmol), Pd(dppf)CH2Cl2 (148 g), and triethylamine (0.757 mL, 5.43 mmol). The contents were degassed and then purged with nitrogen. To this was then added Cul (29 mg). The reaction mixture was heated at reflux for 48 hrs. After cooling, the contents were filtered over CELITE™ and concentrated. Column chromatography of the resulting residue using hexane/ethyl acetate (0- 30%) afforded tert-butyl N- [2- [ [2-chloro-5 -(3 ,3 -diethoxyprop- 1 -ynyl)pyrimidin-4-yl]amino] ethyl] carbamate. 1HNMR (d6-DMSO) δ ppm 8.18 (s, 1H), 7.63 (brs, 1H), 7.40 (brs, 1H), 5.55 (s, 1H), 3.70 (m, 2H), 3.60 (m, 2H), 3.42 (m, 2H), 3.15 (m, 2H), 1.19 – 1.16 (m, 15H). LCMS (ESI) 399 (M + H).
Example 3
Synthesis of tert-butyl N-[2-[2-chloro-6-(diethoxymethyl)pyrrolo[2,3-d]pyrimidin-7- yl] ethyl] carbamate, Compound 3
To a solution of the coupled product (2.1 g, 0.00526 mole) in THF (30 mL) was added TBAF solid (7.0 g). The contents were heated to and maintained at 65 degrees for 2 hrs. Concentration followed by column chromatography using ethyl acetate/hexane (0-50%) afforded tert-butyl N-[2-[2-chloro-6-(diethoxymethyl)pyrrolo[2,3-d]pyrimidin-7-yl]ethyl]carbamate as a pale brown liquid (1.1 g). 1FiNMR (d6-DMSO) δ ppm 8.88 (s, 1H), 6.95 (brs, 1H), 6.69 (s, 1H), 5.79 (s, 1H), 4.29 (m, 2H), 3.59 (m, 4H), 3.34 (m, 1H), 3.18 (m, 1H), 1.19 (m, 9H), 1.17 (m, 6H). LCMS (ESI) 399 (M + H).
Example 4
Synthesis of tert-buty\ N-[2-(2-chloro-6-formyl-pyrrolo [2,3-d] pyrimidin-7- yl)ethyl] carbamate, Compound 4
To the acetal (900 mg) from the preceeding step was added AcOH (8.0 mL) and water
(1.0 mL). The reaction was stirred at room temperature for 16 hrs. Cone, and column chromatography over silica gel using ethyl acetate/hexanes (0- 60%) afforded tert-butyl N-[2-(2- chloro-6-formyl-pyrrolo[2,3-d]pyrimidin-7-yl)ethyl]carbamate as a foam (0.510 g). 1HNMR (d6-DMSO) δ ppm 9.98 (s, 1H), 9.18 (s, 1H), 7.66 (s, 1H), 6.80 (brs, 1H), 4.52 (m, 2H), 4.36 (m, 2H), 1.14 (s, 9H). LCMS (ESI) 325 (M + H).
Example 5
Synthesis of 7- [2-(teri-butoxycarbonylamino)ethyl] -2-chloro-pyrrolo [2,3-d] pyrimidine-6- carboxylic acid, Compound 5
To the aldehyde (0.940 g) from the preceeding step in DMF (4 mL) was added oxone (1.95 g, 1.1 eq). The contents were stirred at room temp for 7 hrs. Silica gel column chromatography using hexane/ethyl acetate (0- 100%) afforded l-\2-(tert- butoxycarbonylamino)ethyl]-2-chloro-pyrrolo[2,3-d]pyrimidine-6-carboxylic acid (0.545 g). 1HNMR (d6-DMSO) δ ppm 9.11 (s, 1H), 7.39 (s, 1H), 4.38 (m, 2H), 4.15 (m, 2H), 1.48 (m, 9H). LCMS (ESI) 341(M + H).
Example 6
Synthesis of methyl 7-[2-(teri-butoxycarbonylamino)ethyl]-2-chloro-pyrrolo[2,3- d]pyrimidine-6-carboxylate, Compound 6
To a solution of 2-chloro-7-propyl-pyrrolo[2,3-d]pyrimidine-6-carboxylic acid (0.545 g, 0.00156 mole) from the preceeding step in toluene (3.5 mL) and MeOH (1 mL) was added TMS- diazomethane (1.2 mL). After stirring overnight at room temperature, the excess of TMS- diazomethane was quenched with acetic acid (3 mL) and the reaction was concentrated under vacuum. The residue was purified by silica gel column chromatography with hexane/ethyl acetate (0- 70%) to afford methyl 7-[2-(tert-butoxycarbonylamino)ethyl]-2-chloro-pyrrolo[2,3- d]pyrimidine-6-carboxylate as an off white solid (0.52 g). 1HNMR (d6-DMSO) δ ppm 9.10 (s, 1H), 7.45 (s, 1H), 6.81 (brs, 1H) 4.60 (m, 2H), 3.91 (s, 3H), 3.29 (m, 2H), 1.18 (m, 9H) LCMS (ESI) 355 (M + H).
Example 7
Synthesis of Chloro tricyclic amide, Compound 7
To methyl 7- [2-(tert-butoxycarbonylamino)ethyl] -2-chloro-pyrrolo [2,3 -d]pyrimidine-6- carboxylate (0.50 g, 0.0014 mole) from the preceeding step in dichloromethane (2.0 mL) was added TFA (0.830 mL). The contents were stirred at room temperature for 1 hr. Concentration under vacuum afforded the crude amino ester which was suspended in toluene (5 mL) and Hunig’s base (0.5 mL). The contents were heated at reflux for 2 hrs. Concentration followed by silica gel column chromatography using hexane/ethyl acetate (0- 50%) afforded the desired chloro tricyclic amide (0.260 g). 1HNMR (d6-DMSO) δ ppm 9.08 (s, 1H), 8.48 (brs, 1H), 7.21 (s, 1H) 4.33 (m, 2H), 3.64 (m, 2H). LCMS (ESI) 223 (M + H).
Example 8
Synthesis of chloro-N-methyltricyclic amide, Compound 8
To a solution of the chloro tricycliclactam, Compound 7, (185 mg, 0.00083 mole) in DMF (2.0 mL) was added sodium hydride (55% dispersion in oil, 52 mg). After stirring for 15 mins, methyl iodide (62 μί, 1.2 eq). The contents were stirred at room temperature for 30 mins. After the addition of methanol (5 mL), sat NaHCOs was added followed by the addition of ethyl acetate. Separation of the organic layer followed by drying with magnesium sulfate and concentration under vacuum afforded the N-methylated amide in quantitative yield. 1FiNMR (d6-DMSO) δ ppm 9.05 (s, 1H), 7.17 (s, 1H) 4.38 (m, 2H), 3.80 (m, 2H), 3.05 (s, 3H). LCMS (ESI) 237 (M + H). Example 9
Synthesis of l-methyl-4-(6-nitro-3-pyridyl)piperazine, Compound 9
To 5-bromo-2-nitropyridine (4.93 g, 24.3 mmole) in DMF (20 mL) was added N- methylpiperazine (2.96 g, 1.1 eq) followed by the addition of DIPEA (4.65 mL, 26.7 mmole). The contents were heated at 90 degrees for 24 hrs. After addition of ethyl acetate (200 mL), water (100 mL) was added and the layers separated. Drying followed by concentration afforded the crude product which was purified by silica gel column chromatography using (0-10%) DCM/Methanol. 1HNMR (d6-DMSO) δ ppm 8.26 (s, 1H), 8.15 (1H, d, J = 9.3 Hz), 7.49 (1H, d, J = 9.4 Hz), 3.50 (m, 4H), 2.49 (m, 4H), 2.22 (s, 3H).
Example 10
Synthesis of 5-(4-methylpiperazin-l-yl)pyridin-2-amine, Compound 10
To l-methyl-4-(6-nitro-3-pyridyl)piperazine (3.4 g) in ethyl acetate (100 mL) and ethanol (100 mL) was added 10%> Pd/C (400 mg) and then the reaction was stirred under hydrogen (10 psi) overnight. After filtration through CELITE™, the solvents were evaporated and the crude product was purified by silica gel column chromatography using DCM/ 7N ammonia in MeOH (0- 5%) to afford 5-(4-methylpiperazin-l-yl)pyridin-2-amine (2.2 g). 1HNMR (d6-DMSO) δ ppm 7.56 (1H, d, J = 3 Hz), 7.13 (1H, m), 6.36 (1H, d, J = 8.8 Hz), 5.33 (brs, 2H), 2.88 (m, 4H), 2.47 (m, 4H), 2.16 (s, 3H).
Example 11
Synthesis of tert-butyl 4-(6-amino-3-pyridyl)piperazine-l-carboxylate, Compound 11
This compound was prepared as described in WO 2010/020675 Al .
Synthesis of Compound 89 (also referred to as Compound T)
Compound 89 was synthesized in a similar manner to that described for compound 78 and was converted to an HCl salt. 1HNMR (600 MHz, DMSO-d6) δ ppm 1.47 (br. s., 6 H) 1.72 (br. s., 2 H) 1.92 (br. s., 2 H) 2.77 (br. s., 3 H) 3.18 (br. s., 2 H) 3.46 (br. s., 2 H) 3.63 (br. s., 2 H) 3.66 (d, J=6.15 Hz, 2 H) 3.80 (br. s., 2 H) 7.25 (s, 1 H) 7.63 (br. s., 2 H) 7.94 (br. s., 1 H) 8.10 (br. s., 1 H) 8.39 (br. s., 1 H) 9.08 (br. s., 1 H) 11.59 (br. s., 1 H). LCMS (ESI) 447 (M + H)
PATENT
WO 2014144740
PATENT
Preparation of Active Compounds
Syntheses
The disclosed compounds can be made by the following general schemes:

Scheme 1
In Scheme 1, Ref-1 is WO 2010/020675 Al; Ref-2 is White, J. D.; et al. J. Org. Chem. 1995, 60, 3600; and Ref-3 Presser, A. and Hufher, A. Monatshefte fir Chemie 2004, 135, 1015.

Scheme 2
In Scheme 2, Ref-1 is WO 2010/020675 Al; Ref-4 is WO 2005/040166 Al; and Ref-5 is Schoenauer, K and Zbiral, E. Tetrahedron Letters 1983, 24, 573.

92


93 
3) Pd/C/H2 ![]()
Scheme 6

Scheme 7
NHfOH

Scheme 8
In Scheme 8, Ref-1 is WO 2010/020675 Al; Ref-2 is WO 2005/040166 Al; and Ref-3 is Schoenauer, K and Zbiral, E. Tetrahedron Letters 1983, 24, 573.
Alternatively, the lactam can be generated by reacting the carboxylic acid with a protected amine in the presence of a strong acid and a dehydrating agent, which can be together in one moiety as a strong acid anhydride. Examples of strong acid anhydrides include, but are not limited to, trifluoroacetic acid anhydride, tribromoacetic acid anhydride, trichloroacetic acid anhydride, or mixed anhydrides. The dehydrating agent can be a carbodiimide based compound such as but not limited to DCC (Ν,Ν-dicyclohexylcarbodiimide), EDC (l-ethyl-3-(3-
dimethylaminopropyl)carbodiimide or DIC (Ν,Ν-diisopropylcarbodiimide). An additional step may be necessary to take off the N-protecting group and the methodologies are known to those skilled in the art.
Alternatively, the halogen moiety bonded to the pyrimidine ring can be substituted with any leaving group that can be displaced by a primary amine, for example to create an intermediate for a final product such as Br, I, F, SMe, SO2Me, SOalkyl, SO2alkyl. See, for Exmaple PCT /US2013/037878 to Tavares.
Other amine intermediates and final amine compounds can be synthesized by those skilled in the art. It will be appreciated that the chemistry can employ reagents that comprise reactive functionalities that can be protected and de-protected and will be known to those skilled in the art at the time of the invention. See for example, Greene, T.W. and Wuts, P.G.M., Greene’s Protective Groups in Organic Synthesis, 4th edition, John Wiley and Sons.

Scheme 9
CDK4/6 Inhibitors of the present invention can be synthesized according to the generalized Scheme 9. Specific synthesis and characterization of the Substituted 2-aminopyrmidines can be found in, for instance, WO2012/061156.
Compounds T, Q, GG, and U were prepared as above and were characterized by mass spectrometry and NMR as shown below:
Compound T
1H NMR (600 MHz, DMSO- d6) ppm 1.47 (br. s., 6 H) 1.72 (br. s., 2 H) 1.92 (br. s., 2 H) 2.77 (br. s., 3 H) 3.18 (br. s., 2 H) 3.46 (br. s., 2 H) 3.63 (br. s., 2 H) 3.66 (d, J=6.15 Hz, 2 H) 3.80 (br. s., 2 H) 7.25 (s, 1 H) 7.63 (br. s., 2 H) 7.94 (br. s., 1 H) 8.10 (br. s., 1 H) 8.39 (br. s., 1 H) 9.08 (br. s., 1 H) 11.59 (br. s., 1 H). LCMS ESI (M + H) 447.
PATENT
Synthesis of N-(heteroaryl)-pyrrolo[3,2-d]pyrimidin-2-amines. The application appears to be particularly focused on methods for the preparation of trilaciclib and an analog of it. Trilaciclib is the company’s lead CDK4/6 inhibitor presently in phase II trials against small-cell lung cancer and triple negative breast cancer. Interestingly, the company is working on a second CDK4/6 inhibitor, G1T38 , which is in a phase II trial against breast cancer.
GENERAL METHODS
The structure of starting materials, intermediates, and final products was confirmed by standard analytical techniques, including NMR spectroscopy and mass spectrometry. Unless otherwise noted, reagents and solvents were used as received from commercial suppliers. Proton nuclear magnetic resonance spectra were obtained on a Bruker AVANCE 500 at 500 MHz in DMSO-dis. HPLC analyses were performed on a Waters HPLC using the below HPLC method.
HPLC Method
Column: Atlantis T3 (150 χ 4.6, 3 μιη)
Column Temperature: 40°C
Flow Rate: 1 mL/min
Detection: UV @ 275 nm
Analysis Time: 36 min
Mobile Phase A: Water (with 0.1% Trifluoroacetic Acid)
Mobile Phase B : Acetonitrile (with 0.1% Trifluoroacetic Acid)
Sample preparation: dissolve PC sample, wet or dry solid (~1 mg of active compound) in acetonitrile/water (1/1) to achieve complete dissolution.
HPLC Method Gradient

Example 1. General Routes of Synthesis

Scheme 1-1 : Starting from an appropriately substituted halo pyrimidine, compounds of the present invention can be prepared. In Step 1 the appropriately substituted halo pyrimidine is subjected to l,4-diazaspiro[5.5]undecan-3-one in the presence of base and heat to afford a substituted spirolactam. In Step 2 the appropriately substituted spirolactam is protected with a group selected from R2. In Step 3 the protected spirolactam is cyclized in the presence of base to afford a fused spirolactam. The fused spirolactam can be optionally oxidized to a sulfoxide or sulfone after Step 3, Step 4, Step 5, or Step 6. Oxidation prior to Step 3 results in undesired byproducts. In Step 4 the hydroxyl group of the fused spirolactam is converted to a leaving group.
In Step 5 the leaving group is dehydrated to afford a compound of Formula IV. In Step 6 the compound of Formula IV is optionally deprotected.

Scheme 1-2: Starting from an appropriately substituted halo pyrimidine compounds of the present invention can be prepared. In Step 1 the appropriately substituted halo pyrimidine is subjected to l,4-diazaspiro[5.5]undecan-3-one in the presence of base and heat to afford a substituted spirolactam. In Step 2 the appropriately substituted spirolactam is protected with a group selected from R2. In Step 3 the protected spirolactam is cyclized in the presence of base to afford a fused spirolactam of Formula IV. The fused spirolactam can be optionally oxidized to a sulfoxide or sulfone after Step 3 or Step 4. Oxidation prior to Step 3 results in undesired byproducts. In Step 4 the compound of Formula IV is optionally deprotected.

Scheme 1-3 : Starting from an appropriately substituted alkyl glycinate, compounds of the present invention can be prepared. In Step 1 the appropriately substituted alkyl glycinate is subjected to cyclohexanone and TMSCN in the presence of base to afford a cyano species. In Step 2 the appropriately substituted cyanospecies is reduced and subsequently cyclized to afford a compound of Formula I.
Scheme 1-4

Scheme 1-4: Starting from an appropriately substituted l-(aminomethyl)cyclohexan-l-amine, compounds of the present invention can be prepared. In Step 1 the appropriately substituted l-(aminomethyl)cyclohexan-l -amine is reductively aminated with an aldehyde. In Step 2 the appropriately substituted cyclohexane amine is optionally deprotected (i.e.: the group selected from R2 if not H is optionally replaced by H). In Step 3 the cyclohexane amine is cyclized to afford a compound of Formula I. In Step 4 the compound of Formula I is optionally protected.
1-5

Conversion

Scheme 1-5: Starting from an appropriately substituted halo pyrimidine, compounds of the present invention can be prepared. In Step 1 the appropriately substituted halo pyrimidine is subjected to l,4-diazaspiro[5.5]undecan-3-one in the presence of base and heat to afford a
substituted spirolactam. In Step 2 the protected spirolactam is cyclized in the presence of base to afford a fused spirolactam. The fused spirolactam can be optionally oxidized to a sulfoxide or sulfone after Step 2, Step 3, Step 4, or Step 5. Oxidation prior to Step 2 results in undesired byproducts. In Step 3 the hydroxyl group of the fused spirolactam is converted to a leaving group. In Step 4 the leaving group is dehydrated to afford a compound of Formula IV. In Step 5 the compound of Formula IV is optionally deprotected.
S

Scheme 1-6: Starting from an appropriately substituted halo pyrimidine compounds of the present invention can be prepared. In Step 1 the appropriately substituted halo pyrimidine is subjected to l,4-diazaspiro[5.5]undecan-3-one in the presence of base and heat to afford a substituted spirolactam. In Step 2 the protected spirolactam is cyclized in the presence of base to afford a fused spirolactam of Formula IV. The fused spirolactam can be optionally oxidized to a sulfoxide or sulfone after Step 2 or Step 3. Oxidation prior to Step 2 results in undesired byproducts. In Step 3 the compound of Formula IV is optionally deprotected.

Scheme 1-7: Starting from compound of Formula IV a CDK4/6 inhibitor can be prepared. In Step 1 a heteroaryl amine is subjected to a base and a compound of Formula IV is added slowly under chilled conditions to afford a nucleophilic substitution reaction. The compound of Formula IV can previously be prepared as described in the schemes herein.
Example 2. Representative Routes of Synthesis
Scheme 2-1

quant, yield 2 steps
isolated

70% yield 2 steps 75% yield 95% yield
isolated isolated isolated
Scheme 2-1 : An ester route is one embodiment, of the present invention. Ideally, the best synthesis scheme would afford crystalline intermediates to provide material of consistent purity without column chromatography, and high yielding steps while using safe and cost effective reagents when possible.
The first step in the ester route is a SNAr nucleophilic substitution of CI group in commercially available ester 3 using spirolactam 4. Due to low reactivity of 4, a reaction temperature of 85-95 °C was required. Because of the temperature requirements, DIPEA and dimethylacetamide were selected as the base and solvent, respectively. The reaction follows second-order kinetics and usually stalls after -85% conversion. Therefore, the reaction was typically stopped after 60 hours by first cooling it to room temperature at which point solid formation was observed. The mixture was then partitioned between MTBE and water and product was filtered with excellent purity with -53% yield of the desired product 5. The obtained
compound 5 was protected with a Boc group using Boc anhydride and DMAP as the catalyst and dichloromethane as the solvent. The intermediate 6 was obtained in a quantitative yield. Due to the semi-solid nature of compound 6, the material was taken to the next step without further purification. The Dieckmann condensation was initially performed with strong bases such as LiHMDS and tBuOK. A similar result to the aldehyde route (Scheme 2-2) was obtained: a partial deprotection of Boc group was observed that required column chromatography. However, the best results were obtained when DBU was used as base and THF as solvent. The reaction outcome was complete, clean conversion of 6 to 7. Moreover, the product crystallized from the reaction mixture upon seeding, and a quantitative yield was obtained for the two steps.
The hydroxyl group of 7 was removed via a two-step procedure. First, compound 7 was converted completely into triflate 8 using triflic anhydride and triethylamine in dichloromethane. The reaction was found to proceed well at 0°C. Due to the potential instability of the triflate intermediate, it was not isolated. It was immediately taken to the next step and reduced with triethylsilane and palladium tetrakis to afford the product 9 after ethyl acetate crystallization in -70% yield. The Boc group of 9 was removed using trifluoroacetic acid in dichloromethane to afford 10. Intermediate 10 was converted into the final sulfone 11 using Oxone™ in acetonitrile/water solvent system.
The obtained sulfone 11 was use-tested in the coupling step and was found to perform well. In conclusion, the route to sulfone 11 was developed which eliminated the use of column chromatography with good to excellent yields on all steps.
Scheme 2-2

Molecular Weight: 421 
Scheme 2-2: The first step of Scheme 2-2 consistently afforded product 13 contaminated with one major impurity found in substantial amount. Thorough evaluation of the reaction impurity profile by LC-MS and 2D MR was performed, which showed the impurity was structurally the condensation of two aldehyde 12 molecules and one molecule of lactam 4. Therefore, column chromatography was required to purify compound 13, which consistently resulted in a modest 30% yield. A solvent screen revealed that sec-butanol, amyl alcohol, dioxane, and tert-butanol can all be used in the reaction but a similar conversion was observed in each case. However, tert-butanol provided the cleanest reaction profile, so it was selected as a solvent for the reaction. Assessing the impact of varying the stoichiometric ratio of 4 and 12 on the reaction outcome was also investigated. The reaction was performed with 4 equivalents of amine 4 in an attempt to disrupt the 2: 1 aldehyde/amine composition of the impurity. The result was only a marginal increase in product 13 formation. The temperature impact on the reaction outcome was evaluated next. The coupling of aldehyde 12 and 4 was investigated at two different temperatures: 50 °C and 40 °C with 1 : 1 ratio of aldehyde/amine. Reactions were checked at 2 and 4 hours and then every 12 hours. The reaction progress was slow at 50°C and was accompanied by growth of other impurities. The reaction at 40°C was much cleaner; however the conversion was lower in the same time period. The mode of addition of the reagents was investigated as well at 80°C with a slow addition (over 6 hours) of either aldehyde 12 or amine 4 to the reaction mixture. The product distribution did not change and an about 1 to 1 ratio was observed between product and impurity when amine 4 was added slowly to the reaction mixture containing aldehyde 12 and
DIPEA at reflux. The product distribution did change when aldehyde 12 was added slowly to the mixture of amine 4 and DIPEA. However, the major product of the reaction was the undesired impurity. Other organic bases were tried as well as different ratios of DIPEA. No product was observed when potassium carbonate was used as a base. The results of the experiments are presented in Table 1 below.
Table 1

Compound 13 was successfully formed in three cases: triethylamine, 2,6-lutidine and DIPEA, with the DIPEA result being the best. The use of Boc protected spirolactam 4 had no effect on the impurity formation as well. Its utilization was speculated to be beneficial in performing the coupling step together with the following step, preparation of compound 14.
The major impurity formed during Step 1 of Scheme 2-2 is:

Chemical Formula:€2)Η;Μ(¾ 6( 2ί>2
Molecular Weight: 527.4903
The second step (Boc protection of the free lactam) proceeded well using DMAP as a catalyst in dichloromethane at room temperature. The product 14 is a thick oil, and, therefore, cannot be purified by crystallization. The Boc protected intermediate 14 was cyclized successfully into the desired pentacyclic structure 10 upon treatment with a strong base such as LiHMDS or tBuOK. Surprisingly, the Boc group was partially removed during the reaction. The level of deprotection was independent from the internal reaction temperature and was positively correlated with excess of base used. Therefore the mixture of the desired product 10 and 10-Boc compound was treated with acid to completely deprotect Boc group. The conversion of methyl sulfide into the final sulfone 11 was carried out with Oxone™. Initially a mixture of methanol and water was used for the reaction. As the result, a partial displacement of sulfone by methoxy group was detected. The methanol was replaced with acetonitrile and the sulfone displacement was eliminated.
In summary, the ester route (Scheme 2-1) is preferred because:
1. Formation of the impurity during the first step of Scheme 2-2 was unavoidable and resulted in yields of < 35%.
2. Column purification was required to isolate intermediate 14.
3. The aldehyde starting material was not commercially available and required two synthetic steps from the corresponding ester.

Scheme 2-3 : Starting with cyclohexanone, compounds of the present invention can be prepared. In Step 1 the methyl glycinate is subjected to cyclohexanone and TMSCN in the presence of tri ethyl amine in DCM to afford 15. In Step 2 15 hydrogenated with hydrogen gas in the presence of catalytic platinum oxide and subsequently undergoes an intramolecular cyclization to afford compound 16 which is used in the schemes above.

Scheme 2-4: Starting with compound 17, compounds of the present invention can be prepared. In Step 1 compound 17 is subjected to ethyl 2-oxoacetate in the presence platinum on carbon and hydrogen gas to afford compound 18. In Step 2 compound 18 is Boc-deprotected with hydrochloric acid. In Step 3 compound 18 is cyclized to afford compound 16 which is used in the schemes above.
Scheme 2-5

11 19
Scheme 2-5: Starting from compound 11 the CDK 4/6 inhibitor 19 can be prepared. In Step 1 5-(4-methylpiperazin-l-yl)pyridin-2-amine is subjected to LiHMDS and compound 11 is added slowly under chilled conditions to afford a nucleophilic substitution reaction and compound 19. Compound 11 can be prepared as described in the schemes herein.

Scheme 2-6: Starting from compound 11 the CDK 4/6 inhibitor 20 can be prepared. In Step 1 5-(4-isopropylpiperazin-l-yl)pyridin-2-amine is subjected to LiHMDS and compound 11 is added slowly under chilled conditions to afford a nucleophilic substitution reaction and compound 20. Compound 11 can be prepared as described in the schemes herein.
Preparation of Compound 5:
A 500 mL, three-neck flask equipped with a mechanical overhead stirrer, thermocouple, N2 inlet, and reflux condenser was charged with ethyl 4-chloro-2-(methylthio)pyrimidine-5-carboxylate 3 (49.2 g, 0.21 mol, 1.00 equiv.), spirolactam 4 (39.2 g, 0.23 mol, 1.10 equiv.), DIPEA (54.7 g, 0.42 mol, 2.00 equiv.), and DMAc (147.6 mL, 3 vol). The batch was heated to 90-95 °C, and after 60 h, IPC confirmed -14% (AUC) of ethyl 4-chloro-2-(methylthio)pyrimidine-5-carboxylate remained. The batch was cooled to RT, and precipitate formation was observed. The suspension was diluted with MTBE (100 mL, 2 vol) and water (442 mL, 9 vol) and stirred for 2 h at RT. The product was isolated by vacuum filtration and washed with MTBE (49 mL, 1 vol). The solid cake was conditioned for 1 h and dried under vacuum at 40 °C for 16 h to afford compound 5 [41.0 g, 53% yield] as an off-white solid with a purity of >99% AUC. ¾ MR (CDCh): δ 8.76 (d, J = 2.0 Hz, 1H), 6.51-6.29 (br, 1H), 4.33 (q, J = 7.0 Hz, 2H), 3.78 (s, 2H), 3.58 (s, 2H), 2.92 (s, 2H), 2.53 (s, 3H), 1.63-1.37 (m, 12H). LCMS (ESI, m/z = 365.3 [M+H]).
Preparation of Compound 6:
A 500 mL, three-neck flask equipped with a mechanical overhead stirrer, thermocouple, N2 inlet was charged with 5 [41.0 g, 0.11 mol, 1.00 equiv.], Boc-anhydride (36.8 g, 0.17 mol, 1.50 equiv.), DMAP (1.37 g, 0.01 mol, 0.10 equiv.), and dichloromethane (287 mL, 7 vol). The batch was stirred for 3 h at RT. IPC confirmed no starting material remained (AUC). The batch was concentrated into a residue under reduced pressure and taken to the next step (a quantitative yield is assumed for this step). An aliquot (200 mg) was purified by column chromatography (heptanes/ethyl acetate 0 to 100%) to afford compound 6. 1H MR (CDCh): δ 8.64 (s, 1H), 4.31 (q, J = 7.0 Hz, 2H), 4.07 (s, 2H), 3.83 (S, 2H), 3.15 (m, 2H), 2.56 (s, 3H), 172 (m, 3H), 1.59 (m, 15H), 1.42 (t, J= 7.0 Hz, 3H). LCMS (ESI, m/z = 465.2 [M+H]).
Preparation of Compound 7:
A 500 mL, three-neck flask equipped with a mechanical overhead stirrer, thermocouple, N2 inlet was charged with compound 6 [residue from a previous step, quantitative yield assumed, 52.2 g, 0.11 mol, 1.00 equiv.], and THF (261 mL, 5 vol). The batch was cooled to 0°C and 1,8-diazabicyclo[5.4.0]un-dec-7-ene (17.1 g, 0.11 mmol, 1.00 equiv.) was added keeping the internal temperature in 0-10°C range. After the addition was complete, the cooling bath was removed and the reaction mixture was allowed to warm up to RT and after 2 h, IPC confirmed no starting material remained. The batch was seeded with the product (1.0 g) and was cooled to 0°C. The slurry was stirred at 0°C for 2 h. The product was isolated by vacuum filtration and washed with cold (0°C) THF (50 mL, 1 vol). The solid cake was conditioned for 1 h and dried under vacuum at 40°C for 16 h to afford 7 [47 g, quantitative yield] as a light orange solid with a purity of >99% AUC. The color of the product changed into yellow once the batch was exposed to air for an extended period of time (~ 1 day). Material was isolated with substantial amount DBU, according to proton NMR. However, it did not interfere with the next step. 1H MR (CDCh): δ 8.71 (s, 1H), 4.03 (s, 2H), 2.57 (s, 3H), 1.85 (m, 10H), 1.51 (s, 9H). LCMS (ESI, m/z = 419.2 [M+H]).
Preparation of Compound 8:
A 500 mL, three-neck flask equipped with a mechanical overhead stirrer, thermocouple, N2 inlet was charged with 7 [40.8 g, 0.10 mol, 1.00 equiv.], triethylamine (31.5 g, 0.31 mol, 3.20 equiv.), and dichloromethane (408 mL, 10 vol). The batch was purged with N2 for 15 min and was cooled to 0°C. Triflic anhydride (44.0 g, 0.16 mol, 1.60 equiv.) was added keeping the
internal temperature in 0-10°C range. The batch was stirred at 0°C and after 3 h, IPC confirmed -7.0% (AUC) of 7 remained. [It was speculated that the product was hydrolyzing back into starting material during the analysis.] Once the reaction was deemed complete, the batch was transferred to a 1 L, separatory funnel and was washed with 50% saturated sodium bicarbonate (200 mL, 5 vol). [It was prepared by mixing saturated sodium bicarbonate (100 mL) with water (100 mL)).] The aqueous layer was separated and was extracted with DCM (2×40 mL, 1 vol). The organic layers were combined and concentrated into a residue under reduced pressure and taken to the next step. LCMS (ESI, m/z = 551.6 [M+H]).
Preparation of Compound 9:
A 500 mL, three-neck flask equipped with a mechanical overhead stirrer, thermocouple, N2 inlet was charged with compound 8 [residue from a previous step, quantitative yield assumed, 53.7 g, 0.10 mol, 1.00 equiv.], and THF (110 mL, 2 vol). The solvent was removed under vacuum distillation and the procedure was repeated two times. The flask was charged with triethylsilane (22.7 g, 0.20 mol, 2.00 equiv.), and DMF (268 mL, 5 vol). The batch was degassed by five cycles of evacuation, followed by backfilling with nitrogen. The flask was charged with tetrakis(triphenylphosphine)palladium(0) (11.3 g, 0.01 mol, 0.1 equiv.). The batch was heated to 45-50°C, and after 14 h, IPC confirmed no starting material remained. The batch was transferred to a 500 mL, separatory funnel while still warm. The reaction was partitioned between water (5 vol) and ethyl acetate (5 vol). The aqueous layer was extracted with ethyl acetate (3 x3 vol). The organic layers were combined and concentrated down to 2 volumes. The precipitate was filtered and washed with ethyl acetate (2x 1 vol). The solid cake was conditioned for 1 h and dried under vacuum at 40°C for 16 h to afford 9 [27.5 g, 70% yield] as a yellow solid with a purity of -98% AUC. Proton NMR showed some triphenylphosphine oxide present. ¾ NMR (DMSO-i¾):5 9.01 (s, 1H), 7.40 (s, 1H), 4.30 (s, 2H), 2.58 (m, 2H), 2.58 (s, 3H), 1.81 (m, 5H), 1.51 (s, 9H). LCMS (ESI, m/z = 403.4 [M+H]).
Preparation of Compound 10 from the Scheme 2-1 route:
A 500 mL, three-neck flask equipped with a mechanical overhead stirrer, thermocouple, N2 inlet was charged 9 (12.8 g, 31.8 mmol, 1.00 equiv.) and dichloromethane (64 mL, 5 vol). Trifluoroacetic acid (18.2 g, 159 mmol, 5.00 equiv.) was added over 20 min and the solution was stirred for 2 h at RT. IPC confirmed reaction was complete. The batch was transferred to a 500 mL, separatory funnel and washed with saturated sodium bicarbonate (200 mL). The aqueous layer was extracted with dichlorom ethane (3 x3 vol). The organic layers were combined and concentrated down to 1 volume. The precipitate was filtered and conditioned for 1 h and dried under vacuum at 40 °C for 16 h to afford 9 [6.72 g, 70% yield] as an off-white solid with a purity of 99.1% AUC. ¾ NMR (DMSO-dis): δ 8.95 (s, 1H), 8.32 (s, 1H), 7.15 (s, 1H), 3.68 (d, J = 1.0 Hz, 2H), 2.86 (m, 2H), 2.57 (s, 3H), 1.92 (m, 2H), 1.73 (m, 3H), 1.39 (m, 3H). LCMS, ESI, m/z = 303.2 [M+H]).
Preparation of Compound 10 from Scheme 2-2 route:
A 50 mL, three-neck flask equipped with a magnetic stirring bar, thermocouple, N2 inlet was charged 14 (680 mg, 1.62 mmol, 1.00 equiv.) and THF (6.8 mL, 10 vol). A I M solution of potassium tert-butoxide (3.2 mL, 3.24 mmol, 2.00 equiv.) in THF was added over 10 min and the solution was stirred for 2 h at RT. IPC confirmed reaction was complete. The batch was acidified with 4 N hydrogen chloride solution in dioxane (2.4 mL, 9.72 mmol, 6.00 equiv.) and stirred for additional 1 h. The batch was transferred to a 500 mL, separatory funnel and washed with saturated sodium bicarbonate (100 mL). The aqueous layer was extracted with ethyl acetate (3 x20 vol). The organic layers were combined and concentrated down to 3volumes and product precipitated. The precipitate was filtered and conditioned for 1 h and dried under vacuum at 40 °C for 16 h to afford 9 [489 mg, quantitative yield] as an off-white solid.
Preparation of Compound 11 :
A 500 mL, three-neck flask equipped with a mechanical overhead stirrer, thermocouple, N2 inlet was charged with 10 (9.00 g, 29.8 mmol, 1.00 equiv.), and acetonitrile (180 mL, 20 vol). A solution of Oxone™ (45.9 g, 0.15 mol, 5.00 equiv.) in water (180 mL, 20 vol) was added to the batch over 20 min. The batch was stirred for 2 h and IPC confirmed the reaction was complete. The batch was concentrated down to ½ of the original volume and was extracted with dichloromethane DCM (4x 10 vol). The organic layers were combined; polish filtered and concentrated down to -10 vol of DCM. The product was slowly crystallized out by addition of heptanes (-30 vol). The mixture was cooled to 0°C and the product was filtered and dried under vacuum at 40 °C for 16 h to afford 11 [9.45 g, 95% yield] as an off-white solid with a purity of >99% AUC. ¾ NMR (CDCb): 5 9.24 (s, 1H), 7.78 (s, 1H), 7.46 (s, 1H), 3.89 (d, J= 2.0 Hz, 2H), 3.43 (s, 3H), 2.98 (m, 2H), 2.10 (m, 2H), 1.86 (m, 3H), 1.50 (m, 3H). LCMS (ESI, m/z = 335.2 [M+H]).
Preparation of Compound 13:
A 250 mL, single-neck flask equipped with a mechanical overhead stirrer, thermocouple, N2 inlet, and reflux condenser was charged with 4-chloro-2-(methylthio)pyrimidine-5-carbaldehyde (2.00 g, 10.6 mmol, 1.00 equiv.), spirolactam 4 (1.96 g, 11.7 mmol, 1.10 equiv.), DIPEA (2.74 g, 21.2 mmol, 2.00 equiv.), and fert-butanol (20 mL, 10 vol). The batch was heated to 80-85 °C, and after 24 h, IPC confirmed no aldehyde 12 remained. The batch was cool to RT and concentrated into a residue, which was loaded on silica gel column. The product was eluted with ethyl acetate/heptanes (0% to 100%). The product containing fractions were pulled out and concentrated to afford 13 [0.98 g, 29% yield] as an off-white solid.
Preparation of Compound 14:
A 500 mL, three-neck flask equipped with a mechanical overhead stirrer, thermocouple, N2 inlet was charged with 13 [0.98 g, 3.00 mmol, 1.00 equiv.], Boc-anhydride (4.90 g, 21.5 mmol, 7.00 equiv.), DMAP (36 mg, 0.30 mmol, 0.10 equiv.), and dichloromethane (7 mL, 7 vol). The batch was stirred for 3 h at RT. IPC confirmed no starting material remained. The batch was cool to RT and concentrated into a residue, which was loaded on silica gel column. The product was eluted with ethyl acetate/heptanes (0% to 100%). The product containing fractions were pulled out and concentrated to afford 14 [0.98 g, 29% yield] as an off-white solid.
Preparation of Compound 15:
To a suspension of methyl glycinate (500 g, 3.98 mol, 1 eq) in DCM (10 L) was added
TEA dropwise at rt under nitrogen atmosphere, followed by the addition of cyclohexanone (781 g, 7.96 mol, 2 eq) dropwise over 15 min. To the resulting mixture was added TMSCN (591 g, 5.97 mol, 1.5 eq) dropwise over 1 hour while maintaining the internal reaction temperature below 35
°C. After stirred at rt for 2 hrs, the suspension became a clear solution. The progress of the reaction was monitored by H- MR.
When the methyl glycinate was consumed completely as indicated by H-NMR analysis, the reaction was quenched by water (5 L). The layers were separated. The aqueous layer was extracted with DCM (1 L). The combined organic phase was washed with water (5 L X 2) and
dried over Na2S04 (1.5 Kg). After filtration and concentration, 1.24 Kg of crude 15 was obtained as oil.
The crude 15 was dissolved in IPA (4 L). The solution was treated with HC1/IPA solution (4.4 mol/L, 1.1L) at RT. A large amount of solid was precipitated during the addition. The resulting suspension was stirred for 2 hrs. The solid product was collected by vacuum filtration and rinsed with MTBE (800 mL). 819 g of pure 15 was obtained as a white solid. The yield was 88.4%. ¾- MR (300 MHz, CD3OD) 4.20 (s, 2H), 3.88 (s, 3H), 2.30-2.40 (d, J = 12 Hz, 2H), 1.95-2.02 (d, J = 12 Hz, 2H), 1.55-1.85 (m, 5H), 1.20-1.40 (m, 1H).
Preparation of Compound 16:
To a solution of 15 (10 g, 43 mmol) in MeOH (100 mL) was added methanolic hydrochloride solution (2 .44 mol/L, 35.3 mL, 2 eq) and Pt02 (0.5 g, 5 wt %). The reaction suspension was stirred with hydrogen bubble at 40 °C for 6 hours. H- MR analysis showed consumption of 15. To the reaction mixture was added K2CO3 (15 g, 108 mmol, 2.5 eq) and the mixture was stirred for 3 hrs. The suspension was filtered and the filtrate was concentrated to dryness. The residual oil was diluted with DCM (100 mL) and resulting suspension was stirred for 3 hrs. After filtration, the filtrate was concentrated to provide crude 16 (6.6 g) as an oil. The crude 16 was diluted with EtOAc/hexane (1 : 1, 18 mL) at rt for 2 hrs. After filtration, 16 (4 g) was isolated. The obtained 16 was dissolved in DCM (16.7 mL) and hexane (100 mL) was added dropwise to precipitate the product. After further stirred for 1 h, 2.8 g of the pure 16 was isolated as a white solid. The yield was 39%. HPLC purity was 98.3%; MS (ESI): 169.2 (MH+); 1 H-NMR (300 MHz, D2O) 3.23 (s, 3H), 3.07 (s, 3H), 1.37-1.49 (m, 10H).
Preparation of compound 19:
5-(4-methylpiperazin-l-yl)pyridin-2-amine (803.1 g; 3.0 equivalents based on sulfone 11) was charged to a 22 L flask. The flask was blanketed with N2 and anhydrous THF added (12.4 kg). The resulting black-purple solution was cooled in an ice bath to < 5°C. 1M LiHMDS (4.7 L; 1.2 equivalents based on sulfone 11) was added via an addition funnel in three equal additions to keep the temperature below 10°C. Upon the completion of the addition, the reaction mixture was warmed to 16°C. The sulfone 11 (455.1 g; 1.00 equivalents) was added in five additions. Reaction proceeded until HPLC analysis of an IPC sample indicated less than 3% of sulfone 11 remained.
To quench the reaction, the contents of the 22L flask were transferred to a 100 L flask containing water. After stirring for 30 minutes at <30°C, the crude product was collected by filtration, washed with water and dried to afford 19 (387 g, 99.1% purity, 63.7% yield).
Preparation of compound 20:
5-(4-isopropylpiperazin-l-yl)pyridin-2-amine (1976.2 g; 3.0 equivalents based on sulfone 11) was charged to a 50 L flask. The flask was blanketed with N2 and anhydrous THF added (10.7 kg). The resulting black-purple solution was cooled in an ice bath to < 5°C. 1M LiHMDS (9.6 kg; 3.6 equivalents based on sulfone) was added via an addition funnel at a rate to keep the temperature below 10°C. Upon the completion of the addition, the reaction mixture was warmed to 16°C over 120 minutes by removing the ice bath. The sulfone (1000 g; 1.00 mol) was added in five additions. The reaction proceeded until HPLC analysis of an IPC sample indicated less than 1% of sulfone 11 remained. After completion of the reaction, ammonium chloride was added to the reaction mixture. The mixture stirred at < 32°C for at least 30 minutes and the solids collected by filtration to afford 20 (900 g, 99.1% purity, 64.2% yield).
Alternate Route to Spirolactam via cyclohexanone:
Scheme 2-7

26
In one embodiment the spirolactam is made via the synthetic scheme above. By reducing the nitrile group before addition of the glycinate group the reaction sequence proceeds in higher yield. The chemistry used in Step 1 is described in the literature (J. Org. Chem. 2005, 70,8027-8034), and was performed on a kilogram scale. The chemistry to convert Compound 24 into the
spirolactam was also demonstrated on kilogram scale. The Boc protection of Compound 23, is carried out at -70°C in order to limit formation of the di-Boc protected product. Experimental details of a 200 g pilot run are described below.
Step 1

200 g of cyclohexanone 21 was converted to 22 using Ti(Oi-Pr)4 /TMSCN/NH3. After work-up, 213 g of 22 was obtained. The H- MR was clean. The yield was 84%. The titanium salts were removed by vacuum filtration. In one embodiment, the titanium salts are removed by centrifugation or Celite filtration.
Step 2

190 g of 22 was mixed with LAH (2 eq) in MTBE for 30 minutes at 45°C. After work-up, 148 g of crude 23 was obtained.
Step 3

136 g of the crude 23 from step 2 was converted to 24 with 0.9 eq of B0C2O at -70°C. The reaction was completed and worked up. After concentration, 188 g of 24 was obtained. The yield was 86%. The H-NMR and C-NMR spectra confirmed that the compound was pure.
Step 4

188 g of 24 was subjected to methyl 2-bromoacetate and K2CO3 in acetonitrile to afford 25. 247 g of crude 25 was obtained.
Step 5

247 g of 25 was subjected to TFA in DCE heated to reflux to afford 26. After work-up, 112 g of 6 as TFA salt was obtained. H- MR was clean.
Step 6

26 27
Compound 26 was stirred in EtOH in the presence at room temperature overnight to afford 27. In one embodiment DCM is used as the solvent instead of EtOH.
Example 3. Purge of residual palladium from Step 5 Scheme 2-1:
Since palladium was used in Step 5 of Scheme 2-1, the levels of residual Pd present in the subsequent synthetic steps was determined. Table 2 below and Figure 3 show the palladium levels in the isolated solids.
Table 2

The material after Step 5 was isolated containing 1.47% (14700 ppm) of residual palladium. This data represents the highest level of palladium in the worst case scenario. The workup conditions of the latter steps purged nearly all of the palladium and the final product, 19 bis HC1 salt, contained 14 ppm of Pd, which is below the standard 20 ppm guidline. The Pd levels will likely be even lower once the catal st loading is optimized in Step 5.

19
The process developed in this route was a significant improvement over the one used for the first generation synthesis. Overall, the scheme consists of seven steps with five isolations, all by crystallization. No silica column chromatography is employed in the synthesis, which makes the process highly scalable. The process workup conditions can successfully purge the 1.47% of residual palladium after step 5 of Scheme 2-1.
///////////////TRILACICLIB, G1T28, G1T 28, SHR 6390, PHASE 2, G1 Therapeutics, Inc.
CN1CCN(CC1)C2=CN=C(C=C2)NC3=NC=C4C=C5C(=O)NCC6(N5C4=N3)CCCCC6
WO-2018001353, APREMILAST, NEW PATENT, ZHEJIANG HUAHAI PHARMACEUTICAL CO., LTD


WO-2018001353, APREMILAST, NEW PATENT, ZHEJIANG HUAHAI PHARMACEUTICAL CO., LTD
(WO2018001353) METHOD FOR PREPARING APREMILAST
ZHEJIANG HUAHAI PHARMACEUTICAL CO., LTD
DU, Xiaoqiu; (CN).
ZHOU, Lianchao; (CN).
LIU, Jiegen; (CN)
EN)Method one: (S) -1- (3-ethoxy-4-methoxyphenyl) -2- (methylsulfonyl) ethylamine N-acetyl-L-leucine salt of formula II is reacted with 3-acetylaminophthalic anhydride of formula III in an aprotic solvent to produce the compound of formula I; method two: (S) -1- (3-ethoxy-4-methoxyphenyl) -2- (methylsulfonyl) ethylamine N-acetyl-L- leucine salt of formula II is reacted with 3-acetylaminophthalic anhydride of formula III in an organic solvent in the presence of an organic alkaline or an alkali metal hydride to produce the compound of formula I. The method for preparing apremilast requires inexpensive raw materials and reagents , is suitable for industrialized production, and has great economic effects.
////////////WO 2018001353, APREMILAST, NEW PATENT, ZHEJIANG HUAHAI PHARMACEUTICAL CO., LTD
ELAGOLIX

ELAGOLIX
- Molecular FormulaC32H30F5N3O5
- Average mass631.590 Da
GNRH antagonist, Endometriosis
Endometriosis PREREGISTERED
Phase III Uterine leiomyoma
| Inventors | Yun-Fei Zhu, Chen Chen, Fabio C. Tucci, Zhiqiang Guo, Timothy D. Gross, Martin Rowbottom, R. Scott Struthers, |
| Applicant | Neurocrine Biosciences, Inc. |
WO 2005007165 PDT PATENT

| Inventors | Zhiqiang Guo, Yongsheng Chen, Dongpei Wu, Chen Chen, Warren Wade, Wesley J. Dwight, Charles Q. Huang, Fabio C. Tucci, |
| Applicant | Neurocrine Biosciences, Inc. |
- Originator Icahn School of Medicine at Mount Sinai
- Developer AbbVie; Neurocrine Biosciences
- Class Antineoplastics; Fluorinated hydrocarbons; Pyrimidines; Small molecules
- Mechanism of Action LHRH receptor antagonists
- Highest Development Phases
- Preregistration Endometriosis
- Phase III Uterine leiomyoma
- Discontinued Benign prostatic hyperplasia; Prostate cancer
- Most Recent Events
- 23 Nov 2017 AbbVie plans a phase III trial for Endometriosis (Monotherapy, Combination therapy) in USA in November 2017 (NCT03343067)
- 01 Nov 2017 Updated efficacy and adverse events data from two phase III extension trials in Endometriosis released by AbbVie
- 27 Oct 2017 Elagolix receives priority review status for Endometriosis in USA
SYN


Elagolix is a specific highly potent non-peptide, orally active antagonist of the GnRH receptor. This compound inhibits pituitary luteinizing hormone (LH) secretion directly, potentially preventing the several week delay and flare associated with peptide agonist therapy.

In 2010, elagolix sodium was licensed to Abbott by Neurocrine Biosciences for worldwide development and commercialization for the treatment of endometriosis. In January 2013, Abbott spun-off its research-based pharmaceutical business into a newly-formed company AbbVie.
AbbVie , following its spin-out from Abbott in January 2013, under license from Neurocrine , is developing elagolix, the lead from a series of non-peptide gonadotropin-releasing hormone antagonists, for treating hormone-dependent diseases, primarily endometriosis and uterine fibroids.
Elagolix sodium is an oral gonadotropin releasing hormone (GnRH) antagonist in development at Neurocrine Biosciences and Abbvie (previously Abbott). In 2017, Abbvie submitted a New Drug Application (NDA) in the U.S. for the management of endometriosis with associated pain. The candidate is being evaluated in phase III trials for the treatment of uterine fibroids.
Elagolix (INN, USAN) (former developmental code names NBI-56418, ABT-620) is a highly potent, selective, orally-active, short-duration, non-peptide antagonist of the gonadotropin-releasing hormone receptor (GnRHR) (KD = 54 pM) which is under development for clinical use by Neurocrine Biosciences and AbbVie.[2][3] As of 2017, it is in pre-registration for the treatment of endometriosis and phase III clinical trials for the treatment of uterine leiomyoma.[1][4] The drug was also under investigation for the treatment of prostate cancer and benign prostatic hyperplasia, but development for these indications was ultimately not pursued.[4] Elagolix is the first of a new class of GnRH inhibitors that have been denoted as “second-generation”, due to their non-peptide nature and oral bioavailability.[1]
Because of the relatively short elimination half-life of elagolix, the actions of gonadotropin-releasing hormone (GnRH) are not fully blocked throughout the day.[1][5] For this reason, gonadotropin and sex hormone levels are only partially suppressed, and the degree of suppression can be dose-dependently adjusted as desired.[1][5] In addition, if elagolix is discontinued, its effects are rapidly reversible.[1][5] Due to the suppression of estrogen levels by elagolix being incomplete, effects on bone mineral density are minimal, which is in contrast to first-generation GnRH inhibitors.[6][7] Moreover, the incidence and severity of menopausal side effects such as hot flashes are also reduced relative to first-generation GnRH inhibitors.[1][5]
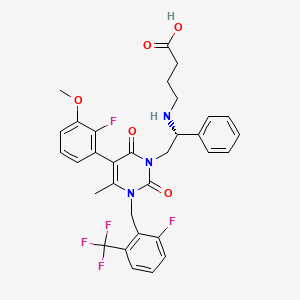
Elagolix sodium is a non-peptide antagonist of the gonadotropin-releasing hormone receptor and chemically known as sodium;4-[[(lR)-2-[5-(2-fluoro-3-methoxyphenyl)-3-[[2-fluoro-6-(trifluoromethyl)phenyl]methyl] -4-methyl-2,6-dioxopyrimidin- 1 -yl] -1 -phenylethyl] amino] butanoate as below.

The US patent number 7056927 B2 discloses, elagolix sodium salt as a white solid and process for its preparation in Example-1; Step-IH.
The US patent number 8765948 B2 discloses a process for preparation of amorphous elagolix sodium by spray drying method and solid dispersion of amorphous elagolix sodium with a polymer.
The US patent number 7056927 B2 discloses a process for preparation of elagolix sodium salt in Example -1 as given in below scheme -I.

Scheme -I
The US patent number 8765948 B2 describes a process for preparation of elagolix sodium in example- 1 and 4 as given below scheme-II:

(1c) (1e) (4a)
Scheme-II
Further, the US patent number 8765948 B2 discloses an alternate process for the preparation of compound of formula (le) as mentioned below scheme-Ill.


Scheme -III
PATENT
| WO2001055119A2 * | Jan 25, 2001 | Aug 2, 2001 | Neurocrine Biosciences, Inc. | Gonadotropin-releasing hormone receptor antagonists and methods relating thereto |
PATENT
WO 2005007165
https://encrypted.google.com/patents/WO2005007165A1?cl=en
EXAMPLE 1
3-[2(R)-{HYD OXYCARBONYLPROPYL-AMINθ} -2-PHENYLETHYL]-5-(2-FLUORO-3- METHOXYPHENYL)-l-[2-FLUORO-6-(TRIFLUOROMETHYL)BENZYL]-6-METHYL- PYRIMIDINE-2,4(lH,3H)-DIONE
Step IA: Preparation of 2-fluoro-6-(trifluoromethyl)benzylamine la To 2-fluoro-6-(trifluoromethyl)benzonitrile (45 g, 0.238 mmol) in 60 mL of TΗF was added 1 M BΗ3:TΗF slowly at 60 °C and the resulting solution was refluxed overnight. The reaction mixture was cooled to ambient temperature. Methanol (420 mL) was added slowly and stirred well. The solvents were then evaporated and the residue was partitioned between EtOAc and water. The organic layer was dried over Na2SO4. Evaporation gave la as a yellow oil (46 g, 0.238 mmol). MS (C\) m/z 194.0 (MH+).
Step IB: Preparation of N-|“2-fluoro-6-(trifluoromethyl)benzyl‘|urea lb To 2-fluoro-6-(trifluoromethyl)benzylamine la (51.5 g, 0.267 mmol) in a flask, urea (64 g, 1.07 mmol), HC1 (cone, 30.9 mmol, 0.374 mmol) and water (111 mL) were added. The mixture was refluxed for 6 hours. The mixture was cooled to ambient temperature, further cooled with ice and filtered to give a yellow solid. Recrystallization with 400 mL of EtOAc gave lb as a white solid (46.2 g, 0J96 mmol). MS (CI) m/z 237.0 (MH+).
Step 1C: Preparation of l-[2-fluoro-6-(trifluoromethyl)benzyl]-6- methylpyrimidine-2.4(lH.3H)-dione lc Nal (43.9 g, 293 mmol) was added to N-[2-fluoro-6- (trifluoromethyl)benzyl]urea lb (46.2 g, 19.6 mmol) in 365 mL of acetonitrile. The resulting mixture was cooled in an ice-water bath. Diketene (22.5 mL, 293 mmol) was added slowly via dropping funnel followed by addition of TMSCl (37.2 mL, 293 mmol) in the same manner. The resulting yellow suspension was allowed to warm to room temperature slowly and was stirred for 20 hours. LC-MS showed the disappearance of starting material. To the yellow mixture 525 mL of water was added and stirred overnight. After another 20 hours stirring, the precipitate was filtered via Buchnner funnel and the yellow solid was washed with water and EtOAc to give lc as a white solid (48.5 g, 16 mmol). 1H ΝMR (CDC13) δ 2.15 (s, 3Η), 5.37 (s, 2H), 5.60 (s, 1H), 7.23-7.56 (m, 3H), 9.02 (s, 1H); MS (CI) m/z 303.0 (MH+).
Step ID: Preparation of 5-bromo-l -[2-fluoro-6-(trifluoromethyl)benzyl“|-6- methylpyrimidine-2.4(lH.3H)-dione Id Bromine (16.5 mL, 0.32 mmol) was added to l-[2-fluoro-6-
(trifluoromethyl)benzyl]-6-methylpyrimidine-2,4(lHJH)-dione lc (48.5 g, 0J6 mol) in 145 mL of acetic acid. The resulting mixture became clear then formed precipitate within an hour. After 2 hours stirring, the yellow solid was filtered and washed with cold EtOAc to an almost white solid. The filtrate was washed with sat. ΝaΗCO3 and dried over Na2SO4. Evaporation gave a yellow solid which was washed with EtOAC to give a light yellow solid. The two solids were combined to give 59.4 g of Id (0J56 mol) total. Η NMR (CDC13) δ 2.4 (s, 3H), 5.48 (s, 2H), 7.25-7.58 (m, 3H), 8.61 (s, 1H); MS (CI) m/z 380.9 (MH+). 5-Bromo-l-[2, 6-difluorobenzyl]-6-methylpyrimidine-2,4(lHJH)-dione ld.l was made using the same procedure.
Step IE: Preparation of 5-bromo-l -r2-fluoro-6-(trifluoromethyl)benzyll-6- methyl-3-[2(R)-tert-butoxycarbonylamino-2-phenylethyll-pyrimidine-2.4(lHJH)-dione le To 5-bromo- 1 -[2-fluoro-6-(trifluoromethyl)benzyl]-6-methylpyrimidine- 2,4(lHJH)-dione Id (15 g, 39.4 mmol) in 225 mL of TΗF were added N-t-Boc-D- phenylglycinol (11.7 g, 49.2 mmol) and triphenylphosphine (15.5 g, 59J mmol), followed by addition of di-tert-butyl azodicarboxylate (13.6 g, 59J mmol). The resulting yellow solution was stirred overnight. The volatiles were evaporated and the residue was purified by silica gel with 3:7 EtOAc Ηexane to give le as a white solid (23.6 g, 39.4 mmol). MS (CI) m/z 500.0 (MΗ+-Boc).
Step IF: Preparation of 3-[2(R)-amino-2-phenylethyll-5-(2-fluoro-3- methoxyphenyl)-l-[2-fluoro-6-(trifluoromethyl)benzyll-6-methyl-pyrimidine- 2.4(lH.3H)-dione If To 5-bromo-l-[2-fluoro-6-(trifluoromethyl)benzyl]-6-methyl-3-[2(R)- tert-butoxycarbonylamino-2-phenylethyl]-pyrimidine-2,4(lH,3H)-dione le (15 g, 25 mmol) in 30 mL/90 mL of Η2O/dioxane in a pressure tube were added 2-fluoro-3- methoxyphenylboronic acid (4.25 g, 25 mmol) and sodium carbonate (15.75 g, 150 mmol). N2 gas was bubbled through for 10 min.
Tetrakis(triphenylphosphine)palladium (2.9 g, 2.5 mmol) was added, the tube was sealed and the resulting mixture was heated with stirring at 90 °C overnight. After cooling to ambient temperature, the precipitate was removed by filtration. The volatiles were removed by evaporation and the residue was partitioned between EtOAc/sat. NaHCO3. The organic solvent was evaporated and the residue was chromatographed with 2:3 EtOAc/Hexane to give 13.4 g (20.8 mmol, 83 %) yellow solid. This yellow solid (6.9 g, 10.7 mmol) was dissolved in 20 mL/20 mL CH2C12/TFA. The resulting yellow solution was stirred at room temperature for 2 hours. The volatiles were evaporated and the residue was partitioned between EtOAc/ sat. NaHCO3. The organic phase was dried over Na2SO4. Evaporation gave If as a yellow oil (4.3 g, 7.9 mmol, 74%). Η NMR (CDC13) δ 2.03 (s, 3H), 3.72-4.59 (m, 6H), 5.32-5.61 (m, 2H), 6.74-7.56 (m, 11H); MS (CI) m/z 546.0 (MH+). 3-[2(R)-amino-2-phenylethyl]-5-(2-fluoro-3-methoxyphenyl)-l-[2,6- difluorobenzyl]-6-methyl-pyrimidine-2,4(lH,3H)-dione lf.l was made using the same procedure described in this example.
Step 1G: Preparation of 3-[2(R)- {ethoxycarbonylpropyl-amino} -2-phenylethyll-5-
(2-fluoro-3 -methoxyphenyl)- 1 -[2-fluoro-6-(trifluoromethyl)benzyl“|-6-methyl- pyrimidine-2,4(lHJH)-dione lg To compound 3-[2(R)-amino-2-phenylethyl]-5-(2-fluoro-3- methoxyphenyl)-l-[2-fluoro-6-(trifluoromethyl)benzyl]-6-methyl-pyrimidine- 2,4(lH,3H)-dione If (5 g, 9.4 mmol) in 100 mL of acetonitrile were added ethyl 4- bromobutyrate (4 mL, 28.2 mmol) and Ηunig’s base (1.6 mL, 9.4 mmol). After reflux at 95 °C overnight, the reaction mixture was cooled to ambient temperature and the volatiles were removed. The residue was chromatographed with 10:10: 1 EtOAc/Ηexane/Et3N to give lg as a yellow oil (3.0 g, 4.65 mmol). MS (CI) m/z 646.2 (MH+).
Step 1H: Preparation of 3-[2(R)- {hydroxycarbonylpropyl-amino} -2-phenylethyl]- 5-(2-fluoro-3-methoxyphenyl)-l- 2-fluoro-6-(trifluoromethyl)benzyl1-6-methyl- pyrimidine-2,4(lHJH)-dione 1-1 Compound 3-[2(R)- {ethoxycarbonylpropyl-amino} -2-phenylethyl]-5-(2- fluoro-3-methoxyphenyl)-l-[2-fluoro-6-(trifluoromethyl)benzyl]-6-methyl-pyrimidine- 2,4(lH,3H)-dione lg (2.6 g, 4.0 mmol) was dissolved in 30 mL/30 mL of TΗF/water. Solid NaOΗ (1.6 g, 40 mmol) was added and the resulting mixture was heated at 50 °C overnight. The mixture was cooled to ambient temperature and the volatiles were evaporated. Citric acid was added to the aqueous solution until pΗ = 3. Extraction with EtOAc followed by evaporation of solvent gave 1.96 g of a white gel. The gel was passed through a Dowex MSC-1 macroporous strong cation-exchange column to convert to sodium salt. Lyopholization gave white solid 1-1 as the sodium salt (1.58 g, 2.47 mmol). Η NMR (CD3OD) δ 1.69-1.77 (m, 2H), 2.09 (s, 3H), 2.09-2.19 (t, J = 7.35 Hz, 2H), 2.49-2.53 (t, J = 735 H, 2H), 3.88 (s, 3H), 4.15-4.32 (m, 3H), 5.36-5.52 (m, 2H), 6.60-7.63 (m, 1 IH); HPLC-MS (CI) m/z 632.2 (MH+), tR = 26.45, (method 5)
PATENT
WO 2017221144
Process for the preparation of elagolix sodium and its polymorph forms and intermediates is claimed. Represents first filing from Dr. Reddy’s Laboratories Limited and the inventors on this API.
n a seventh aspect, the present invention provides a process for preparation of compound of formula (VII)

(VII)
wherein R is alkyl such as methyl, ethyl, propyl, isopropyl and the like,
comprising;
a) reacting the compound of formula (II) with compound of formula (III) to obtain the compound of formula (IV)

wherein t-BOC is tertiary butoxycarbonyl group; R is as described above
b) reacting the compound of formula (IV) with the compound of formula (V) to obtain the compound formula (VI), and

c) N-deprotection of the compound of formula (VI) to obtain the compound of formula
(VII)

(VI) (VII)
The reaction of compound of formula (II) with compound of formula (III) to obtain the compound of formula (IV) is carried in the presence of triarylphosphine such as triphenyl phosphine and the like and azodicarboxylates such as diethyl azodicarboxylate, diisopropyl azodicarboxylate and di-tert-butyl azodicarboxylate (DIAD) and the like.
The seventh aspect of the present invention is depicted below scheme-IV.

Scheme-IV
The eighth aspect of the present invention is depicted below scheme-IV.

R=alkyl
Scheme-IV
Example 11: Preparation of ethyl (R)-4-((2-hydroxy-l-phenylethyl)amino)butanoate (Ilia; R is ethyl)
R-(-)-2-phenylglycinol (10 g), DMAP (0.17 g) were added in THF (80 ml) at room temperature under nitrogen atmosphere. Triethylamine (30.48 ml) was added to the reaction mixture and stirred for five minutes. Ethyl-4-bromo butyrate (15.64 ml) was added and the reaction mixture heated to 80°C then stirred for 16 hours. Water (20 volumes) followed by ethyl acetate (200 ml) were added to separate the aqueous and organic layer. The organic layer was washed with IN HC1 (100 ml) followed by neutralize the resulting aqueous layer with saturated sodium carbonate solution then extract with ethyl acetate (100 ml) and the organic layer was dried over anhydrous sodium sulfate then evaporated below 50°C under reduced pressure to obtain the title compound. Yield: 14.50 g. Purity: 94.75% (by HPLC). ¾ NMR (400 MHz, DMSO-d6): δ 7.17-7.30 (m, 5H), 4.83 (m, 1H), 3.99 (q, 2H), 3.58 (dd, 1H, J = 8.8, 4.4 Hz), 3.88 (m, 1H ), 3.27 (m, 1H), 2.38 (m, 1H), 2.26 (m, 3H), 2.10 (s, 1H), 1.61 (m, 2H), 1.12 (t, 3H); m/z: 252 (MH )
Example 12: Preparation of ethyl (R)-4-((tert-butoxycarbonyl)(2-hydroxy-l-phenylethyl) amino)butanoate (III; R is ethyl)
Ethyl (R)-4-((2-hydroxy-l-phenylethyl)amino)butanoate (14 g) was added to THF (140 ml) at room temperature. The reaction mixture was cooled to 0-5 °C. Triethylamine (16.9 mL) was added to the reaction mixture followed by Di-tert-butyl dicarbonate (13.37 g) was added to reaction mixture at 0-5 °C. The reaction mixture was heated to room temperature and stirred for 16 hours. Water (300 mL) and ethyl acetate (300 mL) were added and the layers were separated. The organic layer was washed with sodium chloride then died over sodium sulfate followed by evaporation at 45°C to obtain the crude compound. The crude compound was purified by silica gel (60/120 mesh) withl5-20% EtOAc/Hexane to obtain the title compound as a pale yellow syrup. Yield: 9.5 g. Purity: 95.42% (by HPLC). ¾ NMR (400 MHz, CDC13): δ 7.24-7.34 (m, 5H), 5.08 (m, 1H), 4.09 (m, 4H), 3.10 (m, 2H), 3.00 (s, 1H), 2.21(m, 2H), 1.82 (m, 2H), 1.46 (s, 9H), 1.23 (t, 3H). m/z: 352.20 (MH )
Example 13: Preparation of ethyl (R)-4-((2-(5-bromo)-3-(2-fluoro-6-trifluoromethyl)benzyl)-4-methyl-2,6-dioxo-3,6-dihydropyrimidin-l(2H)-yl)-l-phenylethyl)(tert-butoxycarbonyl) amino)butanoate (IV; R is ethyl)
Ethyl (R)-4-((tert-butoxycarbonyl)(2-hydroxy-l -phenyl ethyl) amino)butanoate (III; R is ethyl) (1.0 g), 5-bromo-l-(2-fluoro-6-trifluoromethyl)benzyl-6-methylpyrimidine-2,4 (1H, 3H)-dione (II) (1.08 g), Triphenyl phosphine (1.49 g) were added to THF (30 mL) at room temperature under nitrogen atmosphere. DIAD (1.11 mL) was added to the reaction mixture and stirred for 16 hours at room temperature. Water (60 volume) was added to the reaction mixture followed by ethylacetate (60 mL) was added then the layers were separated. The organic layer was dried over sodium sulfate and evaporated below 50°C under reduced pressure to obtain the crude compound. The crude compound was purified by silica gel (60/120 mesh) withl5-20% EtOAc/Hexane to obtain the title compound. Yield (1.3 g). Purity: 68.87% (by HPLC); l NMR (DMSO-d6) δ 1.15-2.0 (11H), 2.43-2.48 (4H), 3.9 (2H), 4.71-4.8 (5H), 5.3 -5.4 (3H), 7.28-7.3 (8H), 8.4 (2H); m/z: 616 (M-BOC)+
Example 14: Preparation of ethyl (R)-4-((tert-butoxycarbonyl)-2-(5-(2-fluoro-3-methoxyphenyl)-3-(2-fluoro-6-trifluoromethyl)benzyl)-4-methyl-2,6-dioxo-3,6-dihydropyrimidin-l(2H)-yl)-l-phenylethyl)amino)-butanoate (VI; R is ethyl)
Ethyl (R)-4-((2-(5-bromo)-3-(2-fluoro-6-trifluoromethyl)benzyl)-4-methyl-2,6-dioxo-3,6-dihydropyrimidin-l(2H)-yl)-l-phenylethyl)(tert-butoxycarbonyl) amino)butanoate (IV; R is ethyl) (0.9 g), 2-fluoro-3-methoxy phenyl boronic acid (V) (0.214 g) and sodium carbonate (0.797 g) were added to the mixture of 1,4-dioxane (9 mL) and water (3.06 mL) at room temperature under nitrogen atmosphere. Argon gas was bubbled through for 30 minutes. Tetrakis (triphenylphosphine)palladium (0.145 g) was added to the reaction mixture at room temperature then heated to 90-95 °C and stirred for 5 hours. The reaction mixture cooled to room temperature and filtered through celite bed then the filtrate washed with ethylacetate (9 mL) and water (36 mL) was added and stirred for 30 minutes at room temperature. Ethylacetate (36 mL) was added and the separated organic layer washed with brine and dried over sodium sulfate followed by evaporation at 45°C to obtain the crude compound. The crude compound was purified by silica gel (60/120 mesh) with 20-25% EtOAc/Hexane to obtain the title compound as yellow solid. Yield: 0.5 g; Purity: 75.1% (by HPLC); m/z: 660 (M-BOC)+.
Example 15: Preparation of ethyl (R)-4-((2-(5-(2-fluoro-3-methoxyphenyl)-3-(2-fluoro-6-trifluoromethyl)benzyl)-4-methyl-2,6-dioxo-3,6-dihydropyrimidin-l(2H)-yl)-l-phenylethyl)amino)-butanoate (VII; R is ethyl)
Ethyl(R)-4-((tert-butoxycarbonyl)-2-(5-(2-fluoro-3-methoxyphenyl)-3-(2-fluoro-6-trifluoro methyl)benzyl)-4-methyl-2,6-dioxo-3,6-dihydropyrimidin-l(2H)-yl)-l-phenylethyl)amino)-butanoate (VI; R is ethyl) (0.4 g) was added to dichloromethane (4 mL) at room temperature. The reaction mixture was cooled to 0-5 °C then trifluoroacetic acid (2 mL) was added and stirred for five hours at 0-5 °C. Saturated sodium bicarbonate solution (40 mL) was added to the reaction mixture followed by dichloromethane (40 mL) was added. The organic layer was washed with brine then dried over sodium sulfate and evaporated at 35°C to obtain the crude compound. The crude compound purified by silica gel (60/120 mesh) with 30-35% EtOAc/Hexane to obtain the title compound as yellow solid. Yield: 160 mg; Purity: 88.6% (by HPLC). ‘H NMR (400 MHz, DMSO-d6): δ 7.64 (m, 1H), 7.54 (m, 2H), 7.15-7.27 (m, 6H), 6.85 (m, 2H), 5.31 (s, 2H), 3.99 (m, 3H), 3.87 (m, 2H), 3.83 (s, 3H), 2.30-2.16 (m, 4H), 2.10 (s, 3H), 1.50 (m, 2H), 1.10 (t, 3H). m/z: 660 (MH )
PAPER
Discovery of sodium R-(+)-4-(2-(5-(2-fluoro-3-methoxyphenyl)-3-(2-fluoro-6-(trifluoromethyl-)benzyl)-4-methyl-2,6-dioxo-3,6-dihydro-2H-pyrimidin-1-yl)-1-phenylethamino)butyrate (elagolix), a potent and orally available nonpeptide antagonist of the human gonadotropin-releasing hormone receptor
J Med Chem 2008, 51(23): 7478
Discovery of Sodium R-(+)-4-{2-[5-(2-Fluoro-3-methoxyphenyl)-3-(2-fluoro-6-[trifluoromethyl]benzyl)-4-methyl-2,6-dioxo-3,6-dihydro-2H-pyrimidin-1-yl]-1-phenylethylamino}butyrate (Elagolix), a Potent and Orally Available Nonpeptide Antagonist of the Human Gonadotropin-Releasing Hormone Receptor
* To whom correspondence should be addressed. Phone: 1-858-617-7600. Fax: 1-858-617-7925. E-mail: cchen@neurocrine.com, sstruthers@neurocrine.com., †
Department of Medicinal Chemistry., ‡ Department of Endocrinology., § Department of Preclinical Development.
The discovery of novel uracil phenylethylamines bearing a butyric acid as potent human gonadotropin-releasing hormone receptor (hGnRH-R) antagonists is described. A major focus of this optimization was to improve the CYP3A4 inhibition liability of these uracils while maintaining their GnRH-R potency. R-4-{2-[5-(2-Fluoro-3-methoxyphenyl)-3-(2-fluoro-6-[trifluoromethyl]benzyl)-4-methyl-2,6-dioxo-3,6-dihydro-2H-pyrimidin-1-yl]-1-phenylethylamino}butyric acid sodium salt, 10b (elagolix), was identified as a potent and selective hGnRH-R antagonist. Oral administration of 10b suppressed luteinizing hormone in castrated macaques. These efforts led to the identification of 10b as a clinical compound for the treatment of endometriosis.
NA SALT
(R)-4-{2-[5-(2-Fluoro-3-methoxyphenyl)-3-(2-fluoro-6-[trifluoromethyl]benzyl)-4-methyl-2,6-dioxo-3,6-dihydro-2H-pyrimidin-1-yl]-1-phenylethylamino}butyric Acid Sodium Salt
sodium salt as a white solid (1.58 g, 2.47 mmol, 62%). HPLC purity: 100% (220 and 254 nm). 1H NMR (CD3OD): 1.72 (m, 2H), 2.08 (s, 3H), 2.16 (t, J = 6.9 Hz, 2H), 2.50 (t, J = 6.9 Hz, 2H), 3.86 (s, 3H), 4.24 (m, 3H), 5.40 (d, J = 9.0 Hz, 1H), 5.46 (d, J = 9.0 Hz, 1H), 6.62 and 6.78 (m, 1H), 7.12 (m, 2H), 7.34 (m, 5H), 7.41 (m, 1H), 7.56 (m, 1H), 7.61 (d, J = 8.0 Hz, 1H). MS: 632 (M − Na + 2H+). Anal. (C32H29F5N3O5Na·0.75H2O): C, H, N, Na.
PATENT
CN 105218389
PATENT
“Elagolix” refers to 4-((R)-2-[5-(2-fluoro-3-methoxy-phenyl)-3-(2- fluoro-6 rifluoromethyl-benzyl)-4-methyl-2,6-dioxo-3,6-dihydro-2H-pyrimidin-l-yl]-l- phenyl-ethylamino)-butyric acid or a pharmaceutically acceptable salt thereof. Elagolix is an orally active, non-peptide GnRH antagonist and is unlike other GnRH agonists and injectable (peptide) GnRH antagonists. Elagolix produces a dose dependent suppression of pituitary and ovarian hormones in women. Methods of making Elagolix and a pharmaceutically acceptable salt thereof are described in WO 2005/007165, the contents of which are herein incorporated by reference.
References
- ^ Jump up to:a b c d e f g Ezzati, Mohammad; Carr, Bruce R (2015). “Elagolix, a novel, orally bioavailable GnRH antagonist under investigation for the treatment of endometriosis-related pain”. Women’s Health. 11(1): 19–28. doi:10.2217/whe.14.68. ISSN 1745-5057.
- Jump up^ Chen C, Wu D, Guo Z, Xie Q, Reinhart GJ, Madan A, Wen J, Chen T, Huang CQ, Chen M, Chen Y, Tucci FC, Rowbottom M, Pontillo J, Zhu YF, Wade W, Saunders J, Bozigian H, Struthers RS (2008). “Discovery of sodium R-(+)-4-{2-[5-(2-fluoro-3-methoxyphenyl)-3-(2-fluoro-6-[trifluoromethyl]benzyl)-4-methyl-2,6-dioxo-3,6-dihydro-2H-pyrimidin-1-yl]-1-phenylethylamino}butyrate (elagolix), a potent and orally available nonpeptide antagonist of the human gonadotropin-releasing hormone receptor”. J. Med. Chem. 51 (23): 7478–85. doi:10.1021/jm8006454. PMID 19006286.
- Jump up^ Thomas L. Lemke; David A. Williams (24 January 2012). Foye’s Principles of Medicinal Chemistry. Lippincott Williams & Wilkins. pp. 1411–. ISBN 978-1-60913-345-0.
- ^ Jump up to:a b AdisInsight: Elagolix.
- ^ Jump up to:a b c d Struthers RS, Nicholls AJ, Grundy J, Chen T, Jimenez R, Yen SS, Bozigian HP (2009). “Suppression of gonadotropins and estradiol in premenopausal women by oral administration of the nonpeptide gonadotropin-releasing hormone antagonist elagolix”. J. Clin. Endocrinol. Metab. 94 (2): 545–51. doi:10.1210/jc.2008-1695. PMC 2646513
 . PMID 19033369.
. PMID 19033369. - Jump up^ Diamond MP, Carr B, Dmowski WP, Koltun W, O’Brien C, Jiang P, Burke J, Jimenez R, Garner E, Chwalisz K (2014). “Elagolix treatment for endometriosis-associated pain: results from a phase 2, randomized, double-blind, placebo-controlled study”. Reprod Sci. 21 (3): 363–71. doi:10.1177/1933719113497292. PMID 23885105.
- Jump up^ Carr B, Dmowski WP, O’Brien C, Jiang P, Burke J, Jimenez R, Garner E, Chwalisz K (2014). “Elagolix, an oral GnRH antagonist, versus subcutaneous depot medroxyprogesterone acetate for the treatment of endometriosis: effects on bone mineral density”. Reprod Sci. 21 (11): 1341–51. doi:10.1177/1933719114549848. PMC 4212335
 . PMID 25249568.
. PMID 25249568.
External links
| Citing Patent | Filing date | Publication date | Applicant | Title |
|---|---|---|---|---|
| WO2014143669A1 | Mar 14, 2014 | Sep 18, 2014 | AbbVie Inc . | Compositions for use in treating heavy menstrual bleeding and uterine fibroids |
| EP2881391A1 | Dec 5, 2013 | Jun 10, 2015 | Bayer Pharma Aktiengesellschaft | Spiroindoline carbocycle derivatives and pharmaceutical compositions thereof |
| US8084614 | Apr 4, 2008 | Dec 27, 2011 | Neurocrine Biosciences, Inc. | Gonadotropin-releasing hormone receptor antagonists and methods relating thereto |
| US8263588 | Apr 4, 2008 | Sep 11, 2012 | Neurocrine Biosciences, Inc. | Gonadotropin-releasing hormone receptor antagonists and methods relating thereto |
| US8481738 | Nov 10, 2011 | Jul 9, 2013 | Neurocrine Biosciences, Inc. | Gonadotropin-releasing hormone receptor antagonists and methods relating thereto |
| US8507536 | Aug 10, 2012 | Aug 13, 2013 | Neurocrine Biosciences, Inc. | Gonadotropin-releasing hormone receptor antagonists and methods relating thereto |
| US8952161 | Jun 5, 2013 | Feb 10, 2015 | Neurocrine Biosciences, Inc. | Gonadotropin-releasing hormone receptor antagonists and methods relating thereto |
| US9034850 | Nov 19, 2010 | May 19, 2015 | Sk Chemicals Co., Ltd. | Gonadotropin releasing hormone receptor antagonist, preparation method thereof and pharmaceutical composition comprising the same |
| US9422310 | Jan 8, 2015 | Aug 23, 2016 | Neurocrine Biosciences, Inc. | Gonadotropin-releasing hormone receptor antagonists and methods relating thereto |
 |
|
| Clinical data | |
|---|---|
| Synonyms | NBI-56418; ABT-620 |
| Routes of administration |
By mouth |
| Drug class | GnRH analogue; GnRH antagonist; antigonadotropin |
| Pharmacokinetic data | |
| Biological half-life | 2.4–6.3 hours[1] |
| Identifiers | |
| CAS Number |
|
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| Chemical and physical data | |
| Formula | C32H30F5N3O5 |
| Molar mass | 631.590 g/mol |
| 3D model (JSmol) | |
///////////////ELAGOLIX, NBI 56418, UNII:5B2546MB5Z, ABT 620, priority review status, PHASE 3, AbbVie, Neurocrine Biosciences, Endometriosis
CC1=C(C(=O)N(C(=O)N1CC2=C(C=CC=C2F)C(F)(F)F)CC(C3=CC=CC=C3)NCCCC(=O)O)C4=C(C(=CC=C4)OC)F
Drug Patents International
All about Patents and Intellectual property by DR ANTHONY MELVIN CRASTO, worlddrugtracker, Ph.D ( ICT, Mumbai) , INDIA 30 Yrs Exp. in the feld of Organic Chemistry, Serving chemists around the world.
THE VIEWS EXPRESSED ARE MY PERSONAL AND IN NO-WAY SUGGEST THE VIEWS OF THE PROFESSIONAL BODY OR THE COMPANY THAT I REPRESENT, amcrasto@gmail.com, +91 9323115463 India
https://drugpatentsint.blogspot.in/
/////////////////
FDA permits marketing of device to treat diabetic foot ulcers
DRUG REGULATORY AFFAIRS INTERNATIONAL

Today, the U.S. Food and Drug Administration permitted the marketing of the Dermapace System, the first shock wave device intended to treat diabetic foot ulcers. Continue reading.
December 28, 2017
Summary
FDA permits marketing of device to treat diabetic foot ulcers
Release
Today, the U.S. Food and Drug Administration permitted the marketing of the Dermapace System, the first shock wave device intended to treat diabetic foot ulcers.
“Diabetes is the leading cause of lower limb amputations,” said Binita Ashar, M.D., director of the division of surgical devices in FDA’s Center for Devices and Radiological Health. “The FDA is dedicated to making technologies available that can help improve the quality of life for those with chronic diseases. Additional options for successfully treating and healing ulcer wounds may help prevent lower limb amputations.”
An estimated 30.3 million people in the United States have been diagnosed with diabetes, according to the Centers…
View original post 404 more words
Pexidartinib, New Patent, WO 2017215521, Crystal Pharmatech Co Ltd

Pexidartinib, New Patent, WO, 2017215521, Crystal Pharmatech Co Ltd
(WO2017215521) PLX3397 HYDROCHLORIDE CRYSTAL FORM, PREPARATION METHOD THEREFOR AND USE THEREOF
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2017215521
CRYSTAL PHARMACEUTICAL (SUZHOU) CO., LTD
INVENTORS
CHEN, Minhua; (CN).
ZHANG, Yanfeng; (CN).
ZOU, Po; (CN).
ZHANG, Xiaoyu; (CN)
Novel crystalline forms of PLX3397 hydrochloride (designated as Forms CS2 and CS3), processes for their preparation and compositions comprising them are claimed. Also claim is their use for treating giant cell tumor of the tendon sheath.
The present invention relates to a PLX3397 hydrochloride crystal form, a preparation method therefor and use thereof. The PLX3397 hydrochloride crystal form has higher solubility, larger particle size, and good stability, especially better mechanical stability, is favorable for separation of products in subsequent production, provides a better choice for preparing PLX3397-containing pharmaceutical preparations, and is very important to medicinal development.
[Figure 0009]
Fig. 1 H NMR chart of crystalline form CS3 in Example 3. Fig
//////////////Pexidartinib, New Patent, WO 2017215521, Crystal Pharmatech Co Ltd
Biocon Launches KRABEVA® in India, A Biosimilar Bevacizumab for Treating Several Types of Cancer

Biocon Launches KRABEVA® in India, A Biosimilar Bevacizumab for Treating Several Types of Cancer
On November 23, 2017, Biocon India’s premier Biopharmaceuticals Company announced that it has launched KRABEVA®, a biosimilar Bevacizumab for the treatment of patients with metastatic colorectal cancer and other types of lung, kidney, cervical, ovarian and brain cancers, in India 1.
KRABEVA®, a monoclonal antibody (mAb) developed by Biocon, will help expand access to a world-class, high quality biosimilar Bevacizumab for cancer patients in India. It is the world´s first and only Bevacizumab with a unique ´QualCheck ´ mechanism, which ensures that patients get a quality-ascertained product right up to infusion.
Bevacizumab is indicated as a first-line treatment of patients with metastatic colorectal cancer (mCRC), and is accepted as a standard treatment option in combination with chemotherapy for patients with non-small-cell lung cancer (NSLC), metastatic renal cell carcinoma or recurrent ovarian cancer.
KRABEVA® is the second key oncologic biosimilar product, from Biocon´s global biosimilars portfolio to be launched in India. It is being offered to patients at an MRP of Rs 24,000 for 100 mg / 4 ml vials and Rs 39,990 for 400 mg / 16 ml vials, making it a high quality affordable alternative to the innovator brand. In comparison, the Innovator brand for Bevacizumab marketed as Avastin® by Roche India Private Limited costs over Rs 10, 7065 for 400mg / 16ml vial.
Bevacizumab is a monoclonal antibody (mAb) targeting Vascular Endothelial Growth Factor- A (VEGF-A), a cell protein that induces growth of blood vessels that feed tumors. By blocking this protein, Bevacizumab cuts the supply of food and oxygen to the tumor, thus starving it.
Bevacizumab is prescribed in the treatment of several cancers including metastatic colorectal cancer, ovarian cancer, advanced non-small-cell lung cancer, recurrent glioblastoma, cervical cancer and renal cancer. Bevacizumab was first approved by the United States Food and
Drug Administration (USFDA), in February 2004 2.
It also features in the World Health Organization’s (WHO) list of essential medicines 3. The WHO list of essential medicines contains the medications considered to be most effective and safe to meet the most important needs in a health system. The list is frequently used by countries to help develop their own local lists of essential medicine.
1 https://www.biocon.com/biocon_press_releases_231117.asp
2 https://www.accessdata.fda.gov/scripts/cder/daf/index.cfm?event=overview.process&ApplNo=125085
3 http://www.who.int/medicines/publications/essentialmedicines/EML_2015_FINAL_amended_NOV2015.pdf?ua=1
Approval and launch of a Bevacizumab biosimilar in India would provide an affordable therapy option for patients of various types of cancer.
//////////Biocon, KRABEVA®, India, Biosimilar, Bevacizumab, Cancer
Empesertib , BAY 1161909

Empesertib , BAY 1161909, Mps1-IN-5, (-)-BAY-1161909
CAS 1443763-60-7
Chemical Formula: C29H26FN5O4S
Molecular Weight: 559.6164
[a]D20 : -78.9° (in DMSO). WO 2014198647
UNII-02Y3Z2756M
(αR)-4-Fluoro-N-[4-[2-[[2-methoxy-4-(methylsulfonyl)phenyl]amino][1,2,4]triazolo[1,5-a]pyridin-6-yl]phenyl]-α-methylbenzeneacetamide
(R)-2-(4-fluorophenyl)-N-(4-(2-((2-methoxy-4-(methylsulfonyl)phenyl)amino)-[1,2,4]triazolo[1,5-a]pyridin-6-yl)phenyl)propanamide
(2R)-2-(4-Fluorophenyl)-N-(4-(2-((4-(methanesulfonyl)-2-methoxyphenyl)amino)(1,2,4)triazolo(1,5-a)pyridin-6-yl)phenyl)propanamide
Benzeneacetamide, 4-fluoro-N-(4-(2-((2-methoxy-4-(methylsulfonyl)phenyl)amino)(1,2,4)triazolo(1,5-a)pyridin-6-yl)phenyl)-alpha-methyl-, (alphaR)-
PHASE 1, BAYER, CANCER
PRODUCT PATENT, WO2013087579, https://www.google.com/patents/WO2013087579A1

SYNTHESIS,
WO 2014198647
Analytical UPLC-MS was performed as follows:
Method A: System: UPLC Acquity (Waters) with PDA Detector und Waters ZQ mass spectrometer; Column: Acquity BEH C18 1 .7μηη 2.1 x50mm; Temperature: 60° C; Solvent A: Water + 0.1 % formic acid; Solvent B: acetonitrile; Gradient: 99 % A – 1 % A (1 .6 min) -> 1 % A (0.4 min) ; Flow: 0.8 mL/min; Injection Volume: 1 .0 μΐ (0.1 mg-1 mg/ml_ sample concentration); Detection: PDA scan range 210-400 nm – Fixed and ESI (+), scan range 170-800 m/z
LC-MS methods:
Method 1 :
Instrument: Waters ACQUITY SQD UPLC System; Column: Waters Acquity UPLC HSS T3 1 .8 μ 50 x 1 mm; Eluent A: 1 I Wasser + 0.25 ml 99%ige Formic acid, Eluent B: 1 I Acetonitril + 0.25 ml 99%ige Formic acid; Gradient: 0.0 min 90% A → 1 .2 min 5% A→ 2.0 min 5% A Ofen: 50° C; Flow: 0.40 ml/min; UV-Detection: 208 – 400 nm.


1H-NMR (300 MHz, DMSO-d6), δ [ppm] = 1 .39 (3H), 3.16 (3H), 3.83 (1 H), 3.95 (3H), 7.08-7.20 (2H), 7.34-7.45 (3H), 7.51 (1 H), 7.63-7.77 (5H), 7.92 (1 H), 8.48 (1 H), 8.64 (1 H), 9.1 1 (1 H), 10.19 (1 H).
[a]D20 : -78.9° (in DMSO).
Determination of enantiomeric purity by analytical chiral HPLC:
Column: Chiralcel OD-RH 150×4.6; Flow: 1 .00 mL/min; Solvent: A: Water with 0.1 % formic acid, B: Acetonitrile; Solvent mixture: 40% A + 60% B. Run Time: 30 min. Retention Time: 12.83 min; UV 254 nm; Enantiomeric Ratio: <1 % : > 99%.
Empesertib, also known as BAY1161909, is an orally bioavailable, selective inhibitor of the serine/threonine monopolar spindle 1 (Mps1) kinase, with potential antineoplastic activity. Upon administration, the Mps1 kinase inhibitor BAY1161909 binds to and inhibits the activity of Mps1. This causes inactivation of the spindle assembly checkpoint (SAC), accelerated mitosis, chromosomal misalignment, chromosomal missegregation, mitotic checkpoint complex destabilization, and increased aneuploidy. This leads to the induction of cell death in cancer cells overexpressing Mps1.
BAY-1161909 is an oral dual specificity protein kinase TTK inhibitor in early clinical trials at Bayer for the treatment of advanced malignancies in combination with paclitaxel.
Bayer and INSERM are developing BAY-1161909 , presumed to be the lead from monopolar spindle-1 inhibitors, including Mps-BAY-2b and Mps-BAY-2c, for the oral treatment of cancer; in July 2016, BAY-1161909 was reported to be in phase I clinical trial.
Mps-1 (Monopolar Spindle 1 ) kinase (also known as Tyrosine Threonine Kinase, TTK). Mps-1 is a dual specificity Ser/Thr kinase which plays a key role in the activation of the mitotic checkpoint (also known as spindle checkpoint, spindle assembly checkpoint) thereby ensuring proper chromosome segregation during mitosis [Abrieu A et al., Cell, 2001 , 106, 83-93]. Every dividing cell has to ensure equal separation of the replicated chromosomes into the two daughter cells. Upon entry into mitosis, chromosomes are attached at their kinetochores to the microtubules of the spindle apparatus. The mitotic checkpoint is a surveillance mechanism that is active as long as unattached kinetochores are present and prevents mitotic cells from entering anaphase and thereby completing cell division with unattached chromosomes [Suijkerbuijk SJ and Kops GJ, Biochemica et Biophysica Acta, 2008, 1786, 24- 31 ; Musacchio A and Salmon ED, Nat Rev Mol Cell Biol., 2007, 8, 379-93]. Once all kinetochores are attached in a correct amphitelic, i.e. bipolar, fashion with the mitotic spindle, the checkpoint is satisfied and the cell enters anaphase and proceeds through mitosis. The mitotic checkpoint consists of a complex network of a number of essential proteins, including members of the MAD (mitotic arrest deficient, MAD 1 -3) and Bub (Budding uninhibited by benzimidazole, Bub 1 -3) families, the motor protein CENP-E, Mps-1 kinase as well as other components, many of these being over-expressed in proliferating cells (e.g. cancer cells) and tissues [Yuan B et al., Clinical Cancer Research, 2006, 12, 405-10]. The essential role of Mps-1 kinase activity in mitotic checkpoint signalling has been shown by shRNA-silencing, chemical genetics as well as chemical inhibitors of Mps-1 kinase [Jelluma N et al., PLos ONE, 2008, 3, e2415; Jones MH et al., Current Biology, 2005, 15, 160-65; Dorer RK et al., Current Biology, 2005, 15, 1070-76; Schmidt M et al., EMBO Reports, 2005, 6, 866-72].
There is ample evidence linking reduced but incomplete mitotic checkpoint function with aneuploidy and tumorigenesis [Weaver BA and Cleveland DW, Cancer Research, 2007, 67, 10103-5; King RW, Biochimica et Biophysica Acta, 2008, 1786, 4-14]. In contrast, complete inhibition of the mitotic checkpoint has been recognised to result in severe chromosome missegregation and induction of apoptosis in tumour cells [Kops GJ et al., Nature Reviews Cancer, 2005, 5, 773-85; Schmidt M and Medema RH, Cell Cycle, 2006, 5, 159-63; Schmidt M and Bastians H, Drug Resistance Updates, 2007, 10, 162-81]. Therefore, mitotic checkpoint abrogation through pharmacological inhibition of Mps-1 kinase or other components of the mitotic checkpoint represents a new approach for the treatment of proliferative disorders including solid tumours such as carcinomas and sarcomas and leukaemias and lymphoid malignancies or other disorders associated with uncontrolled cellular proliferation.
Different compounds have been disclosed in prior art which show an inhibitory effect on Mps-1 kinase:
WO 2009/024824 A1 discloses 2-Anilinopurin-8-ones as inhibitors of Mps-1 for the treatment of proliferate disorders. WO 2010/124826 A1 discloses substituted imidazoquinoxaline compounds as inhibitors of Mps-1 kinase. WO 2011 /026579 A1 discloses substituted aminoquinoxalines as Mps-1 inhibitors.
Substituted triazolopyndine compounds have been disclosed for the treatment or prophylaxis of different diseases:
WO 2008/025821 A1 (Cellzome (UK) Ltd) relates to triazole derivatives as kinase inhibitors, especially inhibitors of ITK or PI3K, for the treatment or prophylaxis of immunological, inflammatory or allergic disorders. Said triazole derivatives are exemplified as possessing an amide, urea or aliphatic amine substituent in position 2.
WO 2009/047514 A1 (Cancer Research Technology Limited) relates to [1 ,2,4]- triazolo-[1 ,5-a]-pyridine and [1 ,2,4]-triazolo-[1 ,5-c]-pyrimidine compounds which inhibit AXL receptor tyrosine kinase function, and to the treatment of diseases and conditions that are mediated by AXL receptor tyrosine kinase, that are ameliorated by the inhibition of AXL receptor tyrosine kinase function etc., including proliferative conditions such as cancer, etc.. Said compounds are exemplified as possessing a substituent in the 5-position and a substituent in the 2-position.
WO 2009/010530 A1 discloses bicyclic heterorayl compounds and their use as phosphatidyli nositol (PI) 3-kinase. Among other compounds also substituted triazolopyridines are mentioned.
WO 2009/027283 A1 discloses triazolopyridine compounds and their use as ASK (apoptosis signal-regulating kinase) inhibitors for the treatment of autoimmune diseases and neurodegenerative diseases. WO 2010/092041 A1 (Fovea Pharmaceuticals SA) relates to [1 ,2,4]-triazolo- [1 ,5-a] -pyridines, which are said to be useful as selective kinase inhibitors, to methods for producing such compounds and methods for treating or ameliorating kinase-mediated disorder. Said triazole derivatives are exemplified as possessing a 2-chloro-5-hydroxyphenyl substituent in the 6- position of the [1 ,2,4]-triazolo-[1 ,5-a]-pyridine.
WO 2011 /064328 A1 , WO 2011 /063907 A1 , and WO 2011 /063908 A1 (Bayer Pharma AG) relate to [1 ,2,4]-triazolo-[1 ,5-a]-pyridines and their use for inhibition of Mps-1 kinase.
WO 2011 /064328 A1 discloses com ounds of fomula S2:
S2
in which
R1 is an aryl- or heteroaryl- group; wherein the aryl- or heteroaryl- group can be substituted inter alia with -N(H)C(=0)R6 or -C(=0)N(H)R6 ; in which R6represents a hydrogen or a Ci-C6-alkyl- group; the Ci-C6-alkyl- group optionally being substituted with halo-, hydroxyl-, d-C3-alkyl, R70-. WO 2011 /064328 A1 does not disclose compounds of the present invention as defined below.
WO 2011 /063907 A1 discloses compounds of fomula S1 :
S1
in which
R1 is an aryl group which is substituted at least one time; whereas the at least one substituent inter alia can be -N(H)C(=0)R6 or -C(=0)N(H)R6 ; in which R6represents a group selected from C3-C6-cycloalkyl, 3- to 10-membered heterocyclyl-, aryl-, heteroaryl-, -(CH2)q-(C3-C6-cycloalkyl), -(CH2)q-(3- to 10- membered heterocyclyl), -(CH2)q-aryl, or -(CH2)q-heteroaryl, wherein R6 is optionally substituted, and q is 0, 1 , 2 or 3;
R2 represents a substituted or unsubstituted aryl- or heteroaryl- group;
R3 and R4 inter alia can be hydrogen; and
R5 represents a substituted or unsubstituted Ci-C6-alkyl group.
WO 2011 /063908 A1 discloses com ounds of fomula S3:
S3
in which
R1 is an aryl group which is substituted at least one time; whereas the at least one substituent inter alia can be -N(H)C(=0)R6 or -C(=0)N(H)R6 ; in which R6inter alia represents a group selected from C3-C6-cycloalkyl, 3- to 10- membered heterocyclyl-, aryl-, heteroaryl-, -(CH2)q-(C3-C6-cycloalkyl), -(CH2)q– (3- to 10-membered heterocyclyl), -(CH2)q-aryl, and -(CH2)q-heteroaryl, wherein R6 is optionally substituted, and q is 0, 1 , 2 or 3;
R2 represents a substituted or unsubstituted aryl- or heteroaryl- group;
R3 and R4 inter alia can be hydrogen; and
R5 is hydrogen.
There are patent applications which are related to [1 ,2,4]-triazolo-[1 ,5-a]- pyridines and their use for inhibition of Mps-1 kinase, but which have not been published at the time of filing of this patent application: Subject matter of the EP patent applications No. 11167872.8, and No. 11167139.2 as well as of the patent application PCT/EP2011 /059806 are com ounds of fomula S4:
S4
in which
R1 represents inter alia a phenyl- group which is substituted at least one time; whereas the at least one substituent inter alia can be -N(H)C(=0)R6; in which R6inter alia can be -(CH2)q-aryl, wherein R6 is optionally substituted, and q is 0, 1 , 2 or 3;
R2 represents a substituted or unsubstituted aryl- or heteroaryl- group;
R3 and R4 inter alia can be hydrogen; and
R5 is hydrogen. However, the state of the art described above does not specifically disclose the substituted triazolopyridine compounds of general formula (I) of the present invention, or a tautomer, an N-oxide, a hydrate, a solvate, or a salt thereof, or a mixture of same, as described and defined herein, and as hereinafter referred to as “compounds of the present invention”, or their pharmacological activity.
The above mentioned patent applications which are related to [1 ,2,4]-triazolo- [1 ,5-a] -pyridines mainly focus on the effectiveness of the compounds in inhibiting Mps-1 kinase, expressed by the half maximal inhibitory concentration (IC50) of the compounds. For example, in WO 2011 /063908 A1 the effectiveness in inhibiting Mps-1 kinase was measured in an Mps-1 kinase assay with a concentration of 10 μΜ adenosine triphosphate (ATP).
The cellular concentration of ATP in mammals is in the millimolar range. Therefore it is important that a drug substance is also effective in inhibiting Mps-1 kinase in a kinase assay with a concentration of ATP in the millimolar range, e.g. 2 mM ATP, in order to potentially achieve an antiproliferative effect in a cellular assay. In addition, as one of ordinary skill in the art knows, there a many more factors determining the druglikeness of a compound. The objective of a preclinical development is to assess e.g. safety, toxicity, pharmacokinetics and metabolism parameters prior to human clinical trials. One important factor for assessing the druglikeness of a compound is the metabolic stability. The metabolic stability of a compound can be determined e.g. by incubating the compound with a suspension of liver microsomes from e.g. a rat, a dog and/or a human (for details see experimental section). Another important factor for assessing the druglikeness of a compound for the treatment of cancer is the inhibition of cell proliferation which can be determined e.g. in a HeLa cell proliferation assay (for details see experimental section). Surprisingly it was found, that the compounds of the present invention are characterized by :
– an IC50 lower than or equal to 1 nM (more potent than 1 nM) in an Mps-1 kinase assay with a concentration of 10 μΜ ATP, and
– an IC50 lower than 10 nM (more potent than 10 nM) in an Mps-1 kinase assay with a concentration of 2 mM ATP, and – a maximum oral bioavailability (Fmax) in rat that is higher than 50 % determined by means of rat liver microsomes as described below, and
– a maximum oral bioavailability (Fmax) in dog that is higher than 45 % determined by means of dog liver microsomes as described below, and
– a maximum oral bioavailability (Fmax) in human that is higher than 45 %, determined by means of human liver microsomes as described below, and
– an IC50 lower than 600 nM in a HeLa cell proliferation assay as described below. Hence, the compounds of the present invention have surprising and advantageous properties. These unexpected findings give rise to the present selection invention. The compounds of the present invention are purposively selected from the above mentioned prior art due to their superior properties. In particular, said compounds of the present invention may therefore be used for the treatment or prophylaxis of diseases of uncontrolled cell growth, proliferation and/or survival, inappropriate cellular immune responses, or inappropriate cellular inflammatory responses or diseases which are accompanied with uncontrolled cell growth, proliferation and/or survival, inappropriate cellular immune responses, or inappropriate cellular inflammatory responses, particularly in which the uncontrolled cell growth, proliferation and/or survival, inappropriate cellular immune responses, or inappropriate cellular inflammatory responses is mediated by Mps-1 kinase, such as, for example, haemotological tumours, solid tumours, and/or metastases thereof, e.g. leukaemias and myelodysplastic syndrome, malignant lymphomas, head and neck tumours including brain tumours and brain metastases, tumours of the thorax including non-small cell and small cell lung tumours, gastrointestinal tumours, endocrine tumours, mammary and other gynaecological tumours, urological tumours including renal, bladder and prostate tumours, skin tumours, and sarcomas, and/or metastases thereof.
PATENT
WO2013087579
https://www.google.com/patents/WO2013087579A1
| Inventors | Volker Schulze, Dirk Kosemund, Antje Margret Wengner, Gerhard Siemeister, Detlef STÖCKIGT, Michael Bruening |
| Applicant | Bayer Intellectual Property Gmbh, Bayer Pharma Aktiengesellschaft |
Synthesis of Examples
Compounds of the present invention
Example01.01
(2 ?)-2-(4-fluorophenyl)-N-[4-(2-{[2-methoxy-4-(methylsulfonyl)phenyl]- amino}[1,2,4]triazolo[1,5-a]pyridin-6-yl)phenyl]propanamide
To a stirred suspension of Int08.011 (6.0 g) in DMF (48 mL) and dichloromethane (96 mL) was added sodium bicarbonate (3.69 g), (2/?)-2-(4- fluorophenyl)propanoic acid (2.71 g) and HATU (8.36 g). The mixture was stirred at room temperature for 4 h. Water was added, and the mixture was stirred for 30 minutes. A half-saturated solution of sodium bicarbonate was added and the mixture was extracted with ethyl acetate. The organic phase was washed with saturated sodium chloride solution, dried (sodium sulfate) and the solvent was removed in vacuum. Silicagel chromatography gave a solid that was triturated with ethyl acetate to give 7,44 g of the title compound. 1H-NMR (400MHz, DMSO-d6): δ [ppm]= 1.40 (d, 3H), 3.16 (s, 3H), 3.84 (q, 1H), 3.96 (s, 3H), 7.09 – 7.18 (m, 2H), 7.36 – 7.44 (m, 3H), 7.51 (dd, 1H), 7.63 – 7.76 (m, 5H), 7.92 (dd, 1H), 8.48 (d, 1H), 8.60 (s, 1H), 9.10 (d, 1 H), 10.16 (s, 1H).
[a]D20 : -77.0° (in DMSO).
Column: Chiralcel OD-RH 150×4.6; Flow: 1.00 mL/min; Solvent: A: Water with 0.1 % formic acid, B: Acetonitrile; Solvent mixture: 40% A + 60% B. Run Time: 30 min. Retention Time: 12.83 min; UV 254 nm; Enantiomeric Ratio: <1% : > 99%. Racemate01.01.r
Starting with Int01.05 and Int03.02, Racemate01.01.r was prepared analogously to the procedure for the preparation of Int08.020.
Racemate01.02.r
PATENT
WO2014009219
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2014198647&recNum=63
Analytical UPLC-MS was performed as follows:
Method A: System: UPLC Acquity (Waters) with PDA Detector und Waters ZQ mass spectrometer; Column: Acquity BEH C18 1 .7μηη 2.1 x50mm; Temperature: 60° C; Solvent A: Water + 0.1 % formic acid; Solvent B: acetonitrile; Gradient: 99 % A – 1 % A (1 .6 min) -> 1 % A (0.4 min) ; Flow: 0.8 mL/min; Injection Volume: 1 .0 μΐ (0.1 mg-1 mg/ml_ sample concentration); Detection: PDA scan range 210-400 nm – Fixed and ESI (+), scan range 170-800 m/z
LC-MS methods:
Method 1 :
Instrument: Waters ACQUITY SQD UPLC System; Column: Waters Acquity UPLC HSS T3 1 .8 μ 50 x 1 mm; Eluent A: 1 I Wasser + 0.25 ml 99%ige Formic acid, Eluent B: 1 I Acetonitril + 0.25 ml 99%ige Formic acid; Gradient: 0.0 min 90% A → 1 .2 min 5% A→ 2.0 min 5% A Ofen: 50° C; Flow: 0.40 ml/min; UV-Detection: 208 – 400 nm.
Preparation of compound A1
Route I
(2/?)-2-(4-fluorophenyl)-N-[4-(2-{[2-methoxy-4-(methylsulfonyl)phenyl]-amino}[1 ,2,4]triazolo[1 ,5-a]pyridin-6-yl)phenyl]propanamide

To a stirred suspension of Int08.011 (6.0 g) in DMF (48 mL) and dichloromethane (96 mL) was added sodium bicarbonate (3.69 g), (2/?)-2-(4-fluorophenyl)propanoic acid (2.71 g) and HATU (8.36 g). The mixture was stirred at room temperature for 4 h. Water was added, and the mixture was stirred for 30 minutes. A half-saturated solution of sodium bicarbonate was added and the mixture was extracted with ethyl acetate. The organic phase was washed with saturated sodium chloride solution, dried (sodium sulfate) and the solvent was removed in vacuum. Silicagel chromatography gave a solid that was triturated with ethyl acetate to give 7.44 g of the title compound.
1H-NMR (400MHz, DMSO-d6): δ [ppm] = 1.40 (d, 3H), 3.16 (s, 3H), 3.84 (q, 1 H), 3.96 (s, 3H), 7.09 – 7.18 (m, 2H), 7.36 – 7.44 (m, 3H), 7.51 (dd, 1 H), 7.63 – 7.76 (m, 5H), 7.92 (dd, 1 H), 8.48 (d, 1 H), 8.60 (s, 1 H), 9.10 (d, 1 H), 10.16 (s, 1 H).
[a]D20 : -77.0° (in DMSO).
Determination of enantiomeric purity by analytical chiral HPLC:
Column: Chiralcel OD-RH 150×4.6; Flow: 1.00 mL/min; Solvent: A: Water with 0.1 % formic acid, B: Acetonitrile; Solvent mixture: 40% A + 60% B. Run Time: 30 min. Retention Time: 12.83 min; UV 254 nm; Enantiomeric Ratio: <1% : > 99%.
Intermediate Int08.01 1
6-(4-aminophenyl)-N-[2-methoxy-4-(methylsulfonyl)phenyl][ 1 ,2,4]-triazolo[1 ,5-a]pyridin-2-amine

To a stirred suspension of Int08.010 (12.3 g) in dichloromethane (40 mL) was added TFA (46 mL). The mixture was stirred at room temperature for 16 h. Further TFA was added (1 mL) and the mixture was stirred at room temperature for 5 h. A saturated solution of potassium carbonate was added until pH 9 was reached. The mixture was extracted with dichloromethane and methanol (10:1 mixture). The solution was dried (sodium sulfate) and the solvent was removed in vacuum. The residue was triturated with ethanol to give 9.2 g of the title compound.
1H-NMR (300MHz, DMSO-d6): δ [ppm]= 3.16 (s, 3H), 3.95 (s, 3H), 5.30 (s, 2H), 6.63 (d, 2H), 7.38 – 7.46 (m, 3H), 7.51 (dd, 1 H), 7.61 (d, 1 H), 7.84 (dd, 1 H), 8.48 (d, 1 H), 8.55 (s, 1 H), 8.93 (d, 1 H).
Intermediate Int08.010
ieri-butyl [4-(2-{[2-methoxy-4-(methylsulfonyl)phenyl]amino}[ 1 ,2,4]-triazolo[1 ,5-a]pyridin-6-yl)phenyl]carbamate

To a stirred suspension of Int01.03 (4.0 g) in toluene (250 mL) and NMP (25 mL) was added Int03.02 (8.31 g), chloro(2-dicyclohexylphosphino-2′,4′,6′-tri-isopropyl-1 ,1′-biphenyl)[2-(2-aminoethyl)phenyl] palladium(ll) methyl-tert-butylether adduct (1.08 g), X-Phos (0.64 g) and powdered potassium phosphate (16.6 g). The flask was degassed twice and backfilled with argon. The mixture was heated to reflux for 16 h.
The reaction mixture was filtered through a microfilter and the solvent was removed in vacuum. The residue was triturated with dichloromethane to give 12.3 g of the title compound.
1H-NMR (400MHz, DMSO-d6): δ [ppm] = 1.46 (s, 9H), 3.16 (s, 3H), 3.96 (s, 3H), 7.43 (d, 1 H), 7.48 – 7.59 (m, 3H), 7.63 – 7.72 (m, 3H), 7.92 (dd, 1 H), 8.48 (d, 1 H), 8.58 (s, 1 H), 9.06 – 9.12 (m, 1 H), 9.46 (s, 1 H).
Intermediate Int01.03.
ieri-butyl [4-(2-amino[1 ,2,4]triazolo[1 ,5-a]pyridin-6-yl)phenyl]carbamate

To a stirred solution of Int01.02 (5.82 g) in 1 -propanol (400 mL) was added 2M potassium carbonate solution (41 mL), {4-[(tert-butoxycarbonyl) amino] phenyl} boronic acid (8.6 g), triphenylphosphine (150 mg) and PdCl2(PPh3)2 (1.9 g). The mixture was heated to reflux for 4 h, the solvent was removed in vacuum, water (150 mL) was added and the mixture was extracted with ethyl
acetate (500 mL). The organic phase was dried (sodium sulfate), filtered through Celite and the solvent was removed in vacuum. The residue was triturated with DCM to give the title compound as a white solid. Yield: 7.2 g. 1H-NMR (400MHz, DMSO-d6): δ [ppm] = 1.37 – 1.55 (m, 9H), 5.99 (s, 2H), 7.36 (dd, 1 H), 7.48 – 7.55 (m, 2H), 7.55 – 7.62 (m, 2H), 7.69 (dd, 1 H), 8.78 (dd, 1 H), 9.44 (s, 1 H).
Intermediate Int01.02
6-Bromo[1 ,2,4]triazolo[1 ,5-a]pyridin-2-amine

Hydroxylammonium chloride (39.8 g) was suspended in methanol (200 mL) and ethanol (190 mL) and Hiinig Base (59 mL) was added at r.t. The mixture was heated to 60°C, Int01.01 (30 g) was added portionwise, and the mixture was stirred at 60 °C for 2h. The solvent was removed in vacuum and water (150 mL) was added. A solid was collected by filtration and was washed with water and dried in vacuum.
Yield: 19.3 g of the title compound.
1H-NMR (300MHz, DMSO-d6): δ [ppm] = 6.10 (s, 2H), 7.28 (dd, 1 H), 7.51 (dd, 1 H), 8.88 (dd, 1 H).
Intermediate Int01.01
Eth l [(5-bromopyridin-2-yl)carbamothioyl]carbamate

Ethoxycarbonylisothiocyanate (16.7 g) was added to a stirred solution of 2-amino-5-brompyridine (20 g) in dioxane (200 mL). The mixture was stirred for 2h at r.t. A white solid precipitated. Hexane (20 mL) was added and the white solid was collected by filtration.
Yield: 30.4 g of the title compound.
1H-NMR (300MHz, DMSO-d6): δ [ppm] = 1 .22 (t, 3H), 4.19 (q, 2H), 8.08 (dd, 1 H), 8.49 (d, 1 H), 8.57 (br. d, 1 H), 1 1 .37 – 12.35 (m, 2H).
Intermediate Int03.02
1 -bromo-2-methoxy-4-(methylsulfonyl)benzene

To a stirred solution of Int03.01 (265 mg) in chloroform (10 mL) was added 3-chlorobenzenecarboperoxoic acid (mCPBA) (890 mg). The mixture was stirred at room temperature for 1 h. A half-saturated solution of sodium bicarbonate was added and the mixture was extracted with dichloromethane. The organic phase was washed with saturated sodium chloride solution, dried (sodium sulfate) and the solvent was removed in vacuum. Silica gel chromatography gave 252 mg of the title compound.
1H-NMR (300MHz, DMSO-d6): δ [ppm] = 3.22 (s, 3H), 3.93 (s, 3H), 7.39 (dd, 1 H), 7.50 (d, 1 H), 7.84 (d, 1 H).
Intermediate Int03.01
1 -bromo-2-methoxy-4-(methylsulfanyl)benzene

To a stirred solution of 1 -bromo-4-fluoro-2-methoxybenzene (4.0 g) in DMF (40 mL) was added sodium methanethiolate (2.76 g). The mixture was stirred at room temperature for 30 minutes and at 85 °C for 2 h. Water was added and the mixture was extracted with ethyl acetate. The organic phase was washed with saturated sodium chloride solution, dried (sodium sulfate) and the solvent was removed in vacuum. Silica gel chromatography gave 280 mg of the title compound.
1H-NMR (400MHz, DMSO-d6): δ [ppm] = 2.46 (s, 3H), 3.82 (s, 3H), 6.74 (dd, 1 H), 6.91 (d, 1 H), 7.44 (d, 1 H).
Intermediate Int03.00
1 -bromo-2-methoxy-4-(methylsulfanyl)benzene (alternative procedure)

To a stirred solution of 1 -bromo-4-fluoro-2-methoxybenzene (10.0 g) in DMF (100 mL) was added sodium methanethiolate (4.44 g). The mixture was stirred at 65°C for 2 h. The mixture was cooled to 0°C and methyl iodide (4.55 mL) was added. The mixture was stirred at room temperature for 1 h and further sodium methanethiolate (4.44 g) was added. The mixture was stirred at 65 °C for 1 h. The mixture was cooled to 0°C and methyl iodide (4.55 mL) was added. The mixture was stirred at room temperature for 1 h. Water was added and the mixture was extracted with ethyl acetate. The organic phase was washed with saturated sodium chloride solution, dried (sodium sulfate) and the solvent was removed in vacuum. Silica gel chromatography gave 6.2 g of the title compound as a 2:1 mixture with the starting material. The mixture was used for the next step without purification.
Route II
(2/?)-2-(4-fluorophenyl)-N-[4-(2-{[2-methoxy-4-(methylsulfonyl)ph
amino}[1 ,2,4]triazolo[1 ,5-a]pyridin-6-yl)phenyl]propanamide

To a stirred suspension of Int21.06 (550 mg) in toluene (18 mL) was added potassium fluoride (260 mg) and powdered potassium phosphate (842 mg) and the flask was degassed twice and backfilled with argon. The mixture was stirred for 15 minutes at r.t.. Int21.03 (350 mg), dicyclohexyl(2′,6′-dimethoxybiphenyl-2-yl)phosphine (81 mg) and palladium acetate (22 mg) were added and the flask was degassed twice and backfilled with argon. The mixture was heated to 85 °C for 3 h. Water was added and the reaction mixture was extracted with ethyl acetate. The organic phase was washed with saturated sodium chloride solution, dried (sodium sulfate) and the solvent was removed in vacuum. Aminophase-silica-gel chromatography gave a solid that was triturated with a mixture of dichloromethane and hexane to give 452 mg of the title compound.
1H-NMR (300 MHz, DMSO-d6), δ [ppm] = 1 .39 (3H), 3.16 (3H), 3.83 (1 H), 3.95 (3H), 7.08-7.20 (2H), 7.34-7.45 (3H), 7.51 (1 H), 7.63-7.77 (5H), 7.92 (1 H), 8.48 (1 H), 8.64 (1 H), 9.1 1 (1 H), 10.19 (1 H).
[a]D20 : -78.9° (in DMSO).
Determination of enantiomeric purity by analytical chiral HPLC:
Column: Chiralcel OD-RH 150×4.6; Flow: 1 .00 mL/min; Solvent: A: Water with 0.1 % formic acid, B: Acetonitrile; Solvent mixture: 40% A + 60% B. Run Time: 30 min. Retention Time: 12.83 min; UV 254 nm; Enantiomeric Ratio: <1 % : > 99%.
Intermediate Int21.06
(2R)-2-(4-fluorophenyl)-N-[4-(4,4,5,5-tetramethyl-1 ,3,2-dioxaborolan-2-yl)phenyl]propanamide

To a stirred solution of 4-(4,4,5,5-tetramethyl-1 ,3,2-dioxaborolan-2-yl)aniline (1.0 g) in DMF (45 mL) and dichloromethane (90 mL) was added sodium bicarbonate (766 mg), Int09.03 (844 mg) and HATU (2.6 g). The mixture was stirred at room temperature for 4 h. Water was added, and the mixture was stirred for 30 minutes. A half-saturated solution of sodium bicarbonate was added and the mixture was extracted with ethyl acetate. The organic phase was washed with saturated sodium chloride solution, dried (sodium sulfate) and the solvent was removed in vacuum. Silica-gel chromatography gave 1.53 g of the title compound.
1H-NMR (400 MHz, DMSO-d6), δ [ppm] = 1.23 (12H), 1.37 (3H), 3.74-3.87 (1 H), 7.06-7.16 (2H), 7.31 -7.42 (2H), 7.51 -7.61 (4H), 10.12 (1 H).
Intermediate Example Int21.05
(4-{[(2R)-2-(4-fluorophenyl)propanoyl]amino}phenyl)boronic acid

To a stirred solution of (4-aminophenyl)boronic acid hydrochloride (2.00 g) in DMF (42 mL) was added sodium bicarbonate (2.9 g), (2R)-2-(4-
fluorophenyl)propanoic acid (2.04 g) and HATU (6.58 g). The mixture was stirred at room temperature for 72 h. Water (140 mL) was added, and the mixture was stirred for 2 h. The white precipitate was collected by filtration and was washed with water and was dried in vacuum to give 2.86 g of the title compound.
1H-NMR (300 MHz, DMSO-d6), δ [ppm] = 1.39 (3H), 3.84 (1 H), 7.08-7.21 (2H), 7.35-7.44 (2H), 7.52 (2H), 7.69 (2H), 7.88 (2H), 10.07 (1 H).
Intermediate Int09.03
2/?)-2-(4-fluorophenyl)propanoic acid

To a stirred solution of Int09.02 (23.6 g) in refluxing ethyl acetate (250ml_) was added a solution of (1S)-1 -phenylethanamine (17.35 g) in ethyl acetate. The mixture was allowed to cool down to room temperature within 1 h. A white solid was collected by filtration, was washed with ethyl acetate and dried in vacuum to give 27.5 g of a solid. The solid was recrystallized from 400 mL refluxing ethyl acetate. The mixture was allowed to cool down to room temperature. A white solid was collected by filtration, was washed with ethyl acetate and dried in vacuum to give 18.3 g of a solid. The solid was twice recrystallized from refluxing ethyl acetate (350 mL; 300 mL). A white solid was collected by filtration, was washed with ethyl acetate and dried in vacuum to give 10.51 g of a solid. The solid was dissolved in water, hydrochloric acid (c=2.0 M) was added until pH 5 was reached and the reaction mixture was extracted with dichloromethane. The organic phase was dried (sodium sulfate) and the solvent was removed in vacuum to give 5.6 g of the title product. The crude product was used without further purification.
1H-NMR (300MHz, DMSO-d6): δ [ppm] = 1.31 (d, 3H), 3.66 (q, 1 H), 7.05 – 7.16 (m, 2H), 7.24 – 7.33 (m, 2H), 12.28 (br. s., 1 H).
[a]D20 : -79.3° (in DMSO)
Determination of enantiomeric purity by analytical chiral HPLC:
Column: Chiralcel OJ-H 150×4.6; Flow: 1.00 mL/min; Solvent: A: Hexane, B: 2-propanol with 0.1 % formic acid; Solvent mixture: 80% A + 20% B. Run Time: 30 min. Retention Time: 3.41 min; UV 254 nm; Enantiomeric Ratio: 99.8% : 0.2%.
Intermediate Int09.02
Rac-2- 4-fluorophenyl)propanoic acid

To a stirred solution of Int09.01 (18.9 g) in ethanol (200 mL) was added a solution of potassium hydroxide (35 g), dissolved in water (200 mL). The mixture was stirred at 0 °C for 4 h. Hydrochloric acid (c=4.0 M) was added until pH 5 was reached and the reaction mixture was extracted with ethyl acetate. The organic phase was separated and the solvent was removed in vacuum to give 15.64 g of the title product. The crude product was used without further purification.
1H-NMR (300MHz, DMSO-d6): δ [ppm] = 1.31 (d, 3H), 3.66 (q, 1 H), 7.05 – 7.15 (m, 2H), 7.24 – 7.33 (m, 2H), 12.30 (s, 1 H).
Intermediate Int09.01
Rac-meth l 2-(4-fluorophenyl)propanoate

To a stirred solution of diisopropylamine (13.0 g) in tetrahydrofurane (160 mL) was added a solution of n-butyllithium in hexane (51.4 mL; c= 2.5 M) at -78 °C. The solution was stirred at 0 °C for 15 minutes. The solution was cooled to -78 °C and a solution of methyl (4-fluorophenyl)acetate (18.0 g), dissolved in tetrahydrofurane (40 mL) was added. The solution was stirred at -78 °C for 30 minutes. Methyl iodide (10.0 mL) was added at -78 °C, and the solution was allowed to warm up to 0 °C within 1 h. Water was added and the reaction mixture was extracted with ethyl acetate. The organic phase was dried (sodium sulfate) and the solvent was removed in vacuum. Silicagel chromatography gave 18.9 g of the title compound.
1H-NMR (400MHz, DMSO-d6): δ [ppm] = 1.34 (d, 3H), 3.55 (s, 3H), 3.79 (q, 1 H), 7.08 – 7.15 (m, 2H), 7.25 – 7.32 (m, 2H).
Intermediate Int21.03
6-chloro-N-[2-methoxy-4-(methylsulfonyl)phenyl][1 ,2,4]triazolo[1 ,5-a]pyridin-2-amine

To a stirred suspension of Int21.02 (0.7 g) in toluene (28 mL) was added Int03.02 (1.27 g), chloro(2-dicyclohexylphosphino-2′,4′,6′-tri-isopropyl-1 ,1′-biphenyl)[2-(2-aminoethyl)phenyl] palladium(ll) methyl-tert-butylether adduct (343 mg), X-Phos (202 mg) and powdered potassium phosphate (3.09 g). The flask was degassed twice and backfilled with argon. The mixture was heated to reflux for 3 h. Further chloro(2-dicyclohexylphosphino-2′,4′,6′-tri-isopropyl-1 ,1′-biphenyl)[2-(2-aminoethyl)phenyl] palladium(ll) methyl-tert-butylether adduct (30 mg) and X-Phos (19 mg) were added and the mixture was heated to reflux
for 15 h. The solvent was removed in vacuum. Silicagel chromatography gave a solid that was triturated ethyl acetate to give 1.0 g of the title compound. 1H-NMR (400 MHz, DMSO-d6): δ [ppm] = 3.16 (3H), 3.95 (3H), 7.42 (1 H), 7.50 (1 H), 7.62-7.69 (2H), 8.41 (1 H), 8.70 (1 H), 9.17 (1 H).
Intermediate Int21.02
6-chloro[1 ,2,4]triazolo[1 ,5-a]pyridin-2-amine
![]()
Hydroxylammonium chloride (4.4 g) was suspended in methanol (35 mL) and ethanol (35 mL) and Hiinig Base (10.2 mL) was added at r.t. The mixture was heated to 60° C, Int21.01 (4.4 g) was added portionwise, and the mixture was stirred at 60 °C for 2h. The solvent was removed in vacuum and water (150 mL) was added. A solid was collected by filtration and was washed with water and dried in vacuum.
Yield: 2.0 g of the title compound.
1H-NMR (300 MHz, DMSO-d6): δ [ppm] = 6.09 (2H), 7.28-7.37 (1 H), 7.39-7.49
(1 H), 8.84 (1 H).
Intermediate Int21.01
Eth l [(5-chloropyridin-2-yl)carbamothioyl]carbamate

Ethoxycarbonylisothiocyanate (3.37 g) was added to a stirred solution of 2-amino-5-cloropyridine (3.0 g) in dioxane (100 mL). The mixture was stirred at r.t. for 14 h. The solvent was removed in vacuum. The solid was dissolved in dichloromethane and methanol (100 : 1 ), filtered and the solvent was removed in vacuum to give a solid that was recystallized from ethyl acetate to give 4.4 g of the title compound.
1H-NMR (400 MHz, CHLOROFORM-d): δ [ppm] = 1.35 (3H), 4.31 (2H), 7.71 (1 H), 8.03 (1 H), 8.34 (1 H), 8.83 (1 H), 12.09 (1 H).
PATENT
Novel crystalline polymorphic forms of (2R)-2-(4-fluorophenyl)-N-[4-(2-{[2- methoxy-4-(methylsulfonyl)phenyl]amino}[1,2,4]triazolo[1,5-a]pyridin-6-yl)phenyl]propan-amide 4-toluenesulfonate (empesertib), and crystalline (2R)-2-(4-fluorophenyl)-N-[4-(2-{[2-methoxy-4- (methylsulfonyl)phenyl]amino}[1,2,4]triazolo[1,5-a]pyridin-6-yl)phenyl]propanamide 4- toluenesulfonate monohydrate, composition comprising them and their preparation methods are claimed. Also claims their use for treating various cancers. It is disclosed that empesertib is a potent Mps-1 kinase inhibitor.
MPS-1 INHIBITORS
The present invention covers crystalline, anhydrous (2/?)-2-(4-fluorophenyl)-/V-[4-(2-{[2-methoxy^-imethylsulfonyljphenyllaminojll^^ltriazololl^-olpyridin-e-yljphenyllpropan-amide 4-toluenesulfonate, and crystalline (2/?)-2-(4-fluorophenyl)-/V-[4-(2-{[2-methoxy-4-(methylsulfonyl)phenyl]amino}[l,2,4]triazolo[l,5-a]pyridin-6-yl)phenyl]propanamide 4-toluenesulfonate monohydrate, as compounds per se, a method of preparing said crystalline, anhydrous compound, pharmaceutical compsitions and pharmaceutical combinations comprising said crystalline, anhydrous compound, and uses of said crystalline, anhydrous compound in the treatment or prophylaxis of cancer, in particular pancreatic cancer, glioblastoma, ovarian cancer, non-small cell lung carcinoma, breast cancer, and/or gastric cancer.
BACKGROUND OF THE INVENTION
(2R)-2-(4-fluorophenyl)-/V-[4-(2-{[2-methoxy-4-(methylsulfonyl)phenyl]-amino}[l,2,4]triazolo[l,5-a]pyridin-6-yl)phenyl]propanamide is known to be a very potent inhibitor of Mps-1 kinase.
WO 2013/087579 Al discloses the compound, data showing its pharmaceutical activity, and a method for the preparation of (2/?)-2-(4-fluorophenyl)-/V-[4-(2-{[2-methoxy-4-(methylsulfonyl)phenyl]amino}[l,2,4]triazolo[l,5-a]pyridin-6-yl)phenyl]propanamide.
WO 2014/009219 Al discloses an improved method for the preparation of (2/?)-2-(4-fluorophenyl)-/V-[4-(2-{[2-methoxy-4-(methylsulfonyl)phenyl]amino}[l,2,4]triazolo[l,5-a]pyridin-6-yl)phenyl]propan-amide.
WO 2014/195408 Al discloses pharmaceutical compositions comprising (2 ?)-2-(4-fluorophenyl)-/V-[4-(2-{[2-methoxy-4-(methylsulfonyl)phenyl]amino}[l,2,4]triazolo[l,5-a]pyridin-6-yl)phenyl]propan-amide mainly in amorphous form.
Surprisingly and unexpectedly, it was observed that a crystalline form of the anhydrous 4-toluenesulfonate salt of (2/?)-2-(4-fluorophenyl)-/V-[4-(2-{[2-methoxy-4-(methylsulfonyl)-phenyl]amino}[l,2,4]triazolo[l,5-o]pyridin-6-yl)phenyl]propanamide shows superior properties in terms of its pharmacological usability compared to the free base (2/?)-2-(4-fluorophenyl)-/V-[4-(2-{[2-methoxy-4-(methylsulfonyl)phenyl]amino}[l,2,4]triazolo[l,5-a]pyridin-6-yl)phenyl]propanamide or other salts thereof.
In accordance with a first aspect, the present invention thus covers crystalline, anhydrous (2/?)-2-(4-fluorophenyl)-/V-[4-(2-{[2-methoxy-4-(methylsulfonyl)phenyl]-amino}[l,2,4]triazolo[l,5-a]pyridin-6-yl)phenyl]propanamide 4-toluenesulfonate, of formula (I) :

(I),
hereinafter also referred to as the “anhydrous tosylate salt” or “anhydrous 4-toluenesulfonate salt”,
Example 1 : Preparation of crystalline, anhydrous (2/?)-2-(4-fluorophenyl)-/V-[4-(2-{[2-methoxy-4-(methylsulfonyl)phenvnamino}fl,2,41triazolofl,5-a1pyridin-6-vDphenyllpropanamide 4-toluene-sulfonate : method 1 (without seeding)

Without seeding:
10 g (17.9 mmol) of (2/?)-2-(4-fluorophenyl)-/V-[4-(2-{[2-methoxy-4-(methylsulfonyl)phenyl]-amino}[l,2,4]triazolo[l,5-a]pyridin-6-yl)phenyl]propanamide were suspended in 2-butanone (100 ml) and heated to 65°C 4.08 g (21.4 mmol) 4-toluenesulfonic acid monohydrate in 2-butanone (20 ml) were added at 65°C. The suspension dissolved and the product precipitated from solution. The mixture was stirred for 21h at 65°C. The mixture was cooled to 20°C over 2h. After 2h stirring at 20°C the precipitate was isolated by suction filtration and washed two times with 100 ml 2-butanone (each). The product was dried in vacuum (approximately 60 mbar) at 50°C for 7h. 12.4 g (95 % of theory) were isolated.
Example 3 : Preparation of crystalline (2/?)-2-(4-fluorophenyl)-/V-[4-(2-{[2-methoxy-4- (methylsulfonyl)phenyllannino}[l,2,41triazolo[l,5-alpyriclin-6-yl)phenyllpropanamide 4; toluene-sulfonate monohydrate: method 1 : without seeding

10 g (17.9 mmol) of (2/?)-2-(4-fluorophenyl)-W-[4-(2-{[2-methoxy-4-(methylsulfonyl)phenyl]-amino}[l,2,4]triazolo[l,5-a]pyridin-6-yl)phenyl]propanamide were suspended in 2-butanone (100 ml) and water (2.4 ml) and heated to 65°C. 4.08 g (21.4 mmol) 4-toluenesulfonic acid monohydrate in 2-butanone (20 ml) were added at 65°C. The suspension dissolved and the product precipitated from solution. The mixture was stirred for 21h at 65°C and then cooled to 20°C within 2h. After 2h stirring at 20°C the precipitate was isolated by suction filtration and washed two times with 100 ml 2-butanone (each). The product was dried in vacuum (approximately 60 mbar) at 50°C for 7h. 11.4 g (85 % of theory) were isolated.
Thermogravimetry showed a wheight loss of 2.2 weight-% while heating from 32.6°C to 100°C.
lH-N MR(DMSO-d6): δ = 1.43 (3H), 2.29 (3H), 3.20 (3H), 3.87 (1H), 4.00 (3H), 4.40-4.95 (broad signal, water) 7.09-7.21 (4H), 7.41-7.50 (5H), 7.55 (1H), 7.69-7.78 (5H), 7.97 (1H), 8.51 (1H), 8.67 (1H), 9.15 (1H), 10.21 (1H) ppm.
REFERENCES
1. Combinations for the treatment of cancer comprising a Mps-1 kinase inhibitor and a mitotic inhibitor
By Wengner, Antje Margret; Siemeister, Gerhard
From PCT Int. Appl. (2014), WO 2014198645 A1 20141218.
2. Preparation of prodrug derivatives of substituted triazolopyridine monopolar spindle 1 kinase inhibitors and their use for the treatment of cancer
By Schulze, Volker; Lerchen, Hans-Georg; Bierer, Donald; Wengner, Antje Margret; Siemeister, Gerhard; Lienau, Philip; Krenz, Ursula; Kosemund, Dirk; Stoeckigt, Detlef; Bruening, Michael; et al
From PCT Int. Appl. (2014), WO 2014198647 A2 20141218.
3. Pharmaceutical compositions comprising substituted triazolopyridine compds.
By Schulze, Volker; Bruening, Michael; Stoeckigt, Detlef
From PCT Int. Appl. (2014), WO 2014195408 A1 20141211.
4. Combinations comprising inhibitors of Mps-1 kinase and anti-apoptotic protein of the Bcl-2 family for the treatment of cancer
By Siemeister, Gerhard; Bader, Benjamin; Wengner, Antje; Mumberg, Dominik; Schulze, Volker; Kroemer, Guido; Vitale, Ilio; Jemaa, Mohamed
From PCT Int. Appl. (2014), WO 2014020043 A1 20140206.
5. Method for preparing substituted triazolopyridines as Mps-1 kinase inhibitors
By Schulze, Volker; Mais, Franz-Josef
From PCT Int. Appl. (2014), WO 2014009219 A1 20140116.
6. Preparation of triazolopyridine derivatives for use as TTK inhibitors
By Schulze, Volker; Kosemund, Dirk; Wengner, Antje Margret; Siemeister, Gerhard; Stoeckigt, Detlef; Bruening, Michael
From PCT Int. Appl. (2013), WO 2013087579 A1 20130620.
///////////BAY1161909, BAY-1161909, BAY 1161909, Empesertib, Mps1-IN-5, (-)-BAY-1161909, PHASE 1
O=C(NC1=CC=C(C2=CN3C(C=C2)=NC(NC4=CC=C(S(=O)(C)=O)C=C4OC)=N3)C=C1)[C@H](C)C5=CC=C(F)C=C5
FDA clears stereotactic radiotherapy system for use in treating breast cancer
DRUG REGULATORY AFFAIRS INTERNATIONAL
 FDA clears stereotactic radiotherapy system for use in treating breast cancer
FDA clears stereotactic radiotherapy system for use in treating breast cancer
Today, the U.S. Food and Drug Administration cleared a new noninvasive stereotactic radiotherapy system intended for use in treating cancer in breast tissue. Continue reading.
December 22, 2017
Summary
FDA clears stereotactic radiotherapy system for use in treating breast cancer
Release
Today, the U.S. Food and Drug Administration cleared a new noninvasive stereotactic radiotherapy system intended for use in treating cancer in breast tissue.
“With today’s clearance, patients will have access to a treatment option that provides greater accuracy in delivering radiation therapy to breast tumors while saving surrounding breast tissue,” said Robert Ochs, Ph.D., acting deputy director for radiological health in the Office of In Vitro Diagnostics and Radiological Health in the FDA’s Center for Devices and Radiological Health.
Radiation therapy is an important treatment option for cancer patients. Approximately 60 percent of all cancer patients will…
View original post 297 more words

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO