IC50 value: 16 nM
Target: androgen receptor
PROUD Indian WORLD RECORD VIEWS holder on THIS BLOG, ………live, by DR ANTHONY MELVIN CRASTO, Worldpeaceambassador, Worlddrugtracker, Helping millions, 100 million hits on google, pushing boundaries,2.5 lakh plus connections worldwide, 45 lakh plus VIEWS on this blog in 227 countries, 7 CONTINENTS ……A 90 % paralysed man in action for you, I am suffering from transverse mylitis and bound to a wheel chair, [THIS BLOG HOLDS WORLD RECORD VIEWS ] A PROUD INDIAN
Home » Uncategorized (Page 73)

Dear Kind Viewer’s
WordPress is kind to me and negotiated a one time 10 year concessional subscription of 260 US dollars…….https://newdrugapprovals.org/
I need one time help to pay this one time 10 year concessional subscription to our favorite blog.
This is done to keep this blog running even after my death.
Currently I am paying 99 US Dollars per annum
email me
amcrasto@gmail.com
call +919323115463
Paypal will work for me via email request to you by me, Indian govt does not allow automatic transfer via paypal buttons on the blog
email me at amcrasto@gmail.com and tell me amount, i will request you via paypal

DR ANTHONY CRASTO
LIONEL MY SON, MY MOTIVATION
.
He was only in first standard in school when I was hit by a deadly one in a million spine stroke called acute transverse mylitis, it made me 90% paralysed and bound to a wheel chair, He cried bitterly and we had never seen him so depressed
Now I keep Lionel as my source of inspiration and helping millions, thanks to millions of my readers who keep me going and help me to keep my son and family happy.
ps
The views expressed are my personal and in no-way suggest the views of the professional body or the company that I represent,
///////////
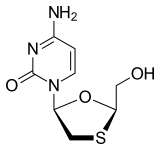
Lamivudine (2′,3′-dideoxy-3′-thiacytidine, commonly called 3TC) is an antiretroviral medication used to prevent and treat HIV/AIDS and used to treat chronic hepatitis B.[1]
It is of the nucleoside analog reverse transcriptase inhibitor (NRTI) class. It is marketed in the United States under the tradenames Epivir and Epivir-HBV.
It is on the World Health Organization’s List of Essential Medicines, a list of the most important medication needed in a basic health system.[2] As of 2015 the cost for a typical month of medication in the United States is more than 200 USD.[3]
Lamivudine has been used for treatment of chronic hepatitis B at a lower dose than for treatment of HIV/AIDS. It improves the seroconversion of e-antigen positive hepatitis B and also improves histology staging of the liver. Long term use of lamivudine leads to emergence of a resistant hepatitis B virus (YMDD) mutant. Despite this, lamivudine is still used widely as it is well tolerated.
In HIV, high level resistance is associated with the M184V/I mutation in the reverse transcriptase gene as reported by Raymond Schinazi’s group at Emory University. GlaxoSmithKline claimed that the M184V mutation reduces “viral fitness”, because of the finding that continued lamivudine treatment causes the HIV viral load to rebound but at a much lower level, and that withdrawal of lamivudine results in a higher viral load rebound with rapid loss of the M184V mutation; GSK therefore argued that there may be benefit in continuing lamivudine treatment even in the presence of high level resistance, because the resistant virus is “less fit”. The COLATE study has suggested that there is no benefit to continuing lamivudine treatment in patients with lamivudine resistance.[4] A better explanation of the data is that lamivudine continues to have a partial anti-viral effect even in the presence of the M184V mutation.
In hepatitis B, lamivudine resistance was first described in the YMDD (tyrosine–methionine–aspartate-aspartate) locus of the HBV reverse transcriptase gene. The HBV reverse transcriptase gene is 344 amino acids long and occupies codons 349 to 692 on the viral genome. The most commonly encountered resistance mutations are M204V/I/S.[5] The change in amino acid sequence from YMDD to YIDD results in a 3.2 fold reduction in the error rate of the reverse transcriptase, which correlates with a significant growth disadvantage of the virus. Other resistance mutations are L80V/I, V173L and L180M.[6]
Lamivudine is an analogue of cytidine. It can inhibit both types (1 and 2) of HIV reverse transcriptase and also the reverse transcriptase of hepatitis B virus. It is phosphorylated to active metabolites that compete for incorporation into viral DNA. They inhibit the HIV reverse transcriptase enzyme competitively and act as a chain terminator of DNA synthesis. The lack of a 3′-OH group in the incorporated nucleoside analogue prevents the formation of the 5′ to 3′ phosphodiester linkage essential for DNA chain elongation, and therefore, the viral DNA growth is terminated.
Lamivudine is administered orally, and it is rapidly absorbed with a bio-availability of over 80%. Some research suggests that lamivudine can cross the blood–brain barrier. Lamivudine is often given in combination with zidovudine, with which it is highly synergistic. Lamivudine treatment has been shown to restore zidovudine sensitivity of previously resistant HIV. Lamivudine showed no evidence of carcinogenicity or mutagenicity in in vivo studies in mice and rats at doses from 10 to 58 times those used in humans.[7]
Racemic BCH-189 (the minus form is known as Lamivudine) was invented by Dr. Bernard Belleau while at work at McGill University and Dr Paul Nguyen-Ba at the Montreal-based IAF BioChem International, Inc. laboratories in 1988 and the minus enantiomer isolated in 1989. Samples were first sent to Dr. Yung-Chi Cheng of Yale University for study of its toxicity. When used in combination with AZT, he discovered that Lamivudine’s negative form reduced side effects and increased the drug’s efficiency at inhibiting reverse transcriptase.[8] The combination of Lamivudine and AZT increased the efficiency at inhibiting an enzyme HIV uses to reproduce its genetic material. As a result, Lamivudine was identified as a less toxic agent to mitochondria DNA than other retroviral drugs.[9]
Lamivudine was approved by the Food and Drug Administration (FDA) on November 17, 1995 for use with zidovudine (AZT) and again in 2002 as a once-a-day dosed medication. The fifth antiretroviral drug on the market, it was the last NRTI for three years while the approval process switched to protease inhibitors. According to the manufacturer’s 2004 annual report, its patent will expire in the United States in 2010 and in Europe in 2011.
On September 2014, Dr. Gorbee Logan, a Liberian physician, reported positive results while treating Ebola virus disease with Lamivudine. Out of 15 patients treated with the antiviral, 13 (those treated within the third to fifth day of symptoms being manifested) survived the disease and were declared virus-free; the remaining cases (treated from the fifth day or later) died.[10][11]
Lamivudine is also available in fixed combinations with other HIV drugs:
Lamivudine (I) (CAS No. 134678-17-4) is chemically known as (-)-[2R,5S]-4T amino- 1 – [2-(hydroxymethyl)- 1 ,3 -oxathiolan-5-yl] -2( 1 H)-pyrimidin-2-one.
Formula (I)
Lamivudine is a reverse transcriptase inhibitor used alone or in combination with other classes of Anti-HIV drugs in the treatment of HIV infection. It is available commercially as a pharmaceutical composition under the brand name EPIVIR®, marketed by GlaxoSmithKline, and is covered under US 5,047,407.
This molecule has two stereo-centres, thus giving rise to four stereoisomers: (±)- Cis Lamivudine and (±)-Trans Lamivudine. The pharmaceutically active isomer however is the (-)-Cis isomer which has the absolute configuration [2R,5S] as show in Formula (I).
US 5,047,407 discloses the 1,3-oxathiolane derivatives; their geometric (cis/trans) and optical isomers. This patent describes the preparation of Lamivudine as a mixture of cis and trans isomers (shown in scheme I). The diastereomers obtained are converted into N-acetyl derivatives before separation by column chromatography using ethylacetate and methanol (99:1); however, this patent remains silent about further resolution of the cis isomer to the desired (-)- [2R,5S]-Cis-Lamivudine. Secondly, as the ethoxy group is a poor leaving group, the condensation of cytosine with compound VI gives a poor yield, i.e. 30 – 40%, of compound VII. Thirdly, chromatographic separation that has been achieved only after acetylation requires a further step of de-acetylation of the cis-(±)- isomer. Also, separation of large volumes of a compound by column chromatography makes the process undesirable on a commercial scale.
(+/-) Cis (+/-) Cis Lamivudine (VIII)
Scheme – 1 Efforts have been made in the past to overcome the shortcomings of low yield and enantiomeric enrichment, hi general, there have been two approaches to synthesize (— )-[2R,5S]-Cis-Lamivudine. One approach involves stereoselective synthesis, some examples of which are discussed below.
US 5,248,776 describes an asymmetric process for the synthesis of enantiomerically pure β-L-(-)-l,3-oxathiolone-nucleosides starting from optically pure 1,6-thioanhydro-L-gulose, which in turn can be easily prepared from L- Gulose. The condensation of the 1,3-oxathiolane derivative with the heterocyclic base is carried out in the presence of a Lewis acid, most preferably SnCl4, to give the [2R,5R] and [2R,5S] diastereomers that are then separated chromatographically.
US 5,756,706 relates a process where compound A is esterified and reduced to compound B. The hydroxy group is then converted to a leaving group (like acetyl) and the cis- and trans-2R-tetrahydrofuran derivatives are treated with a pyrimidine base, like N-acetylcytosine, in the presence trimethylsilyl triflate to give compound C in the diastereomeric ratio 4: 1 of cis and trans isomers.
A B C
Z = S5 CH
Dissolving compound C in a mixture of 3:7 ethyl acetate-hexane separates the cis isomer. The product containing predominantly the cis-2R,5S isomer and some trans-2R,5R compound is reduced with NaBH4 and subjected to column chromatography (30% MeOH-EtOAc) to yield the below compound.
US 6,175,008 describes the preparation of Lamivudine by reacting mercaptoacetaldehyde dimer with glyoxalate and further with silylated pyrimidine base to give mainly the cis-isomer by using an appropriate Lewis acid, like TMS-
I5 TMS-Tf, TiCl4 et cetera. However the stereoselectivity is not absolute and although the cis isomer is obtained in excess, this process still requires its separation from the trans isomer. The separation of the diastereomers Js done by acetylation and chromatographic separation followed by deacetylation. Further separation of the enantiomers of the cis-isomer is not mentioned.
US 6,939,965 discloses the glycosylation of 5-fluoro-cytosine with compound F (configuration: 2R and 2S)
. F
The glycosylation is carried out in the presence of TiCl3(OiPr) which is stereoselective and the cis-2R,5S-isomer is obtained in excess over the trans- 2S,5S-isomer. These diastereomers are then separated by fractional crystallization.
US 6,600,044 relates a method for converting the undesired trans-l,3-oxathiolane nucleoside to the desired cis isomer by a method of anomerizatioή or transglycosylation and the separation of the hydroxy-protected form of cis-, trans- (-)-nucleosides by fractional crystallization of their hydrochloride, hydrobromide, methanesulfonate salts. However, these cis-trans isomers already bear the [R] configuration at C2 and only differ in their configuration at C5; i.e. the isomers are [2R,5R] and [2R,5S]. Hence diastereomeric separation directly yields the desired [2R, 5S] enantiomer of Lamivudine.
In the second approach to prepare enantiomerically pure Lamivudine the resolution of racemic mixtures of nucleosides is carried out. US 5,728,575 provides one such method by using enzyme-mediated enantioselective hydrolysis of esters of the formula
wherein, ‘R’ is an acyl group and ‘Rl ‘ represents the purine or pyrimidine base.
‘R’ may be alkyl carboxylic, substituted alkyl carboxylic and preferably an acyl group that is significantly electron-withdrawing, eg. α-haloesters. After selective hydrolysis, the process involves further separation of the unhydrolyzed ester from the‘ enantiomerically pure 1,3-oxathiolane-nucleoside. Three methods are suggested in this patent, which are:
1. Separation of the more lipophilic unhydrolyzed ester by solvent extraction with one of a wide variety of nonpolar organic solvents.
2. Lyophilization followed by extraction into MeOH or EtOH. 3. Using an HPLC column designed for chiral separations.
In another of its aspects, this patent also refers to the use of the enzyme cytidine- deoxycytidine deaminase, which is enantiomer-specific, Λo catalyze the deamination of the cytosine moiety and thereby converting it to uridine. Thus, the enantiomer that remains unreacted is still basic and can be extracted by using an acidic solution.
However, the above methods suffer from the following drawbacks, (a) Enzymatic hydrolysis sets down limitations on choice of solvents: alcohol solvents cannot be used as they denature enzymes. (b) Lyophilization on an industrial scale is tedious, (c) Chiral column chromatographic separations are expensive.
WO 2006/096954 describes the separation of protected or unprotected enantiomers of the cis nucleosides of below formula by using a chiral acid to form diastereomeric salts that are isolated by filtration. Some of the acids used are R-
(-)-Camphorsulfonic acid, L-(-)-Tartaric acid, L-(-)-Malic acid, et cetera.
However, the configuration of these CIS-nucleosides are [2R,4R] and [2S,4S] as the heterocyclic base is attached at the 4 position of the oxathiolane ring and the overall stereo-structure of the molecule changes from that of the 2,5-substituted oxathiolane ring.
Thus various methods are described for the preparation of Lamivudine. However there is no mention in the prior art about the separation of an enantiomeric pair, either cis-(±) or trans-(±), from a mixture containing cis-[2R,5S], [2S,5R] and trans-[2R,5R], [2S,5S] isomers. Further, there also is a need to provide resolution of the cis-(±) isomers to yield the desired enantiomer in high optical purity.
CN 1223262 (Deng et aϊ) teaches the resolution of a certain class of compounds called Prazoles by using chiral host compounds such as dinaphthalenephenols (BINOL), diphenanthrenols or tartaric acid derivatives. The method consists of the formation of a 1:1 complex between the chiral host (BINOL) and one of the enantiomers, the guest molecule. The other enantiomer remains in solution. (S)- Omeprazole, which is pharmaceutically active as a highly potent inhibitor of gastric acid secretion, has been isolated from its racemic mixture in this manner by using S-BINOL.
BINOL is a versatile chiral ligand that has found its uses in various reactions involving asymmetric synthesis (Noyori, R. Asymmetric Catalysis in Organic
Synthesis) and optical resolution (Cram, D. J. et al J. Org. Chem. 1977, 42, 4173-
4184). Some of these reactions include BINOL-mediated oxidation and reduction reactions, C-C bond formation reactions such as Aldol reaction, Michael addition,
Mannich reaction et cetera (Brunei Chem. Rev. 2005 105, 857-897) and kinetic resolution, resolution by inclusion complexation et cetera.
BINOL, or l,l’-bi-2-Naphthol, being an atropoisomer possesses the property of chiral recognition towards appropriate compounds. One of the uses of BINOL in resolution that is known in literature is in Host-Guest complexation. In one such example, 1,1-binaphthyl derivatives have been successfully incorporated into optically active crown ethers for the enantioselective complexation of amino acid esters and chiral primary ammonium ions (Cram, D. J. Ace. Chem. Res. 1978, 11, 8-14). The chiral ‘host’ is thus able to discriminate between enantiomeric compounds by the formation of hydrogen bonds between the ether oxygen and the enantiomers. The complex formed with one of the isomers, the ‘guest’, will be less stable on steric grounds and this forms the basis for its separation.
It is evident from the literature cited that there exists a need to (a) synthesize Lamivudine by a process requiring less expensive, less hazardous and easily available reagents, and (b) achieve good yields with superior quality of product without resorting to column chromatography as a means of separation, thereby making the process of Lamivudine manufacture more acceptable industrially.
CLIP
ideally, the chemical synthesis of APIs begins from simple, inexpensive building blocks or RMs that are used for multiple purposes and are available in the fine chemicals industry, though some require uncommon RMs that contribute significantly to API manufacturing cost. RMs are converted into APIs by multi-step processes of breaking old chemical bonds and making new ones. A synthesis of 3TC is shown in . In the seven-step sequence, six steps involve breaking existing chemical bonds and creating new ones to build the molecular architecture of the API. The final recrystallization of an API is a critical step; at this stage the crystalline form of the API is determined and related substances (impurities) are removed or reduced to acceptable levels. APIs are often milled in a final step so that their particle size distribution (PSD) falls within specified limits. The crystalline form and PSD of an API must be controlled, because these properties are often critical to the formulation, dissolution, absorption and bioavailability of a drug. Bioavailability is the fraction of a drug dose that reaches systemic circulation (that is, is present in blood plasma) after administration. By definition, a drug is 100% bioavailable when administered by injection; drugs for ART are taken every day and administration by injection is not possible.
The cost of ART is absolutely critical to ensuring access in LMICs. The cost of manufacturing an API is dependent upon the cost of RMs, the cost of overheads and labour (OHL) and volume demand for the product. OHL includes the capital investment to build a manufacturing facility and operating costs, including personnel and energy, waste disposal and the eventual cost of decommissioning of the facility. Increased volume demand generally decreases the cost contribution of RM and OHL. Substantial production volumes are required to obtain full economy of scale . Producing 1–5 metric tons per year is substantially more expensive per kilogram than producing 100 metric tons of an API. There is a practical limit of approximately 50–100 metric tons/year beyond which cost reductions are modest with increased volume, but this practical limit refers to the volumes of drug manufactured in any single manufacturing plant. Exceptions to these generalizations do occur, most often when demand exceeds either the existing manufacturing capacity for a specific API or the availability of critical RMs . Exceptions that have occurred include shortages of β-thymidine for producing AZT and a squeeze on the availability and price of adenine as a starting material for TDF. Another contributor to RM and OHL costs is the efficiency of a chemical synthesis. Since operating costs for a manufacturing facility may be USD2,000/h, the number of steps or processing time for a chemical synthesis affects manufacturing cost. The efficiency of a synthesis is often quoted as an E-factor representing the kilograms of waste produced per kilogram of product manufactured. Waste management is expensive in chemical manufacturing wherever environmental guidelines are both reasonable and followed. From a slightly different perspective, increasing the overall yield of an API synthesis reduces RM use and associated cost for manufacturing.
Jinliang L, Feng LV. inventors; Shanghai Desano Pharmaceutical, assignee. A process for stereoselective synthesis of lamivudine. European Patent Application EP 2161 267 A1. 2007 June 29.

Object of the invention
Thus, one object of the present invention is to provide a process for the synthesis of_Lamivudine which is cost effective, uses less hazardous and easily available reagents, yet achieves good yields with superior quality of product without resorting to column chromatography.
A further object of the present invention is to provide an improved process for the synthesis of Lamivudine, by separating the mixture of diastereomers: Cis-[2R,5S], [2S,5R] from Trans-[2R,5R], [2S,5S] and then resolving the Cis isomers using BINOL to obtain (-)-[2R,5S]^Cis-Lamivudine with at least 99% ee.
This 1,3-oxathiolane compound VIII is further condensed with silylated cytosine in the presence of a Lewis acid such as trimethylsilyliodide to get protected 6-amino-3 – {2-hydroxymethyl- 1 ,3 -oxathiolan-5-yl} -3 -hydropyrimidine- 2-one (compound IX). OH
Cis(±)and Trans (±) racemic mixtures
Lamivudine (-)-[2
Compound (IX) is mixture of following optical isomers
SCHEME 2 The separation of the four-component diastereomeric mixture of isomers bearing the following configuration: trans-[2R,5R], [2S.5S] and cis-[2R,5S], [2S,5R] forms the next step. The separation efficiency of the benzoyl-protected compound
Example 9
Preparation of Lamivudine: (-)-[2R,5S]-4-amino-l-[2-(hydroxymethyl)-l,3- oxathiolan-5 -yl] -2(1 H)-pyrimidin-2-one
Compound I 5mL of cone. HCl was slowly added to a solution of 2Og of Lamivudine-BINOL complex in 100ml of ethylacetate and 10OmL of DM water (pH 2-2.5). The layers. were separated and a 10OmL aliquot of ethylacetate was added to the aqueous layer. The layers were separated again and the aqueous layer was neutralized using 1OmL of 10% aqueous NaOH solution. The solvent was recovered under vacuum at 40-45 0C, the product obtained was dissolved in 160 mL of methanol, filtered, the filtrate was concentrated and 32 mL of water-ethanol mixture (3:1) was added to this product, heated to get a clear solution, cooled to 5 – 10 0C and then filtered. The residue was vacuum dried at 45-50 0C. Yield: 4-5g.
Enantiomeric excess = 99.74 % m.p. = 133-135 °C [<X]D at 25°C = 98.32° (c = 5 water)
1H NMR (DMSO d6): 2.99-3.07 (dd, IH), 3.35-3.38 (dd, IH), 3.72-3.74 (m, 2H), 5.14-5.18 (t, IH), 5.32-5.38 (t, IH), 5.71-5.75 (d, IH), 6.16-6.21 (t, IH), 7.22-
7.27 (d, 2H), 7.80-7.83 (d, IH)
Moisture content: 1.67%
IR (in KBr, cm“1): 3551, 3236, 2927, 1614, 1492, 1404, 1336, 1253, 1146, 1052,
967, 786. MS: M+l =230
XRD [2Θ] (Cu – Ka1=I.54060A, Ka2=1.54443A Kβ= 1.39225A; 4OmA, 45kV):
5.08, 9.89, 10.16, 11.40, 11.65, 12.96, 13.23, 15.26, 15.82, 17.74, 18.74, 18.88,
19.67, 20.69, 22.13, 22.88, 23.71, 25.47, 26.07.
PATENT
http://www.google.com/patents/WO2013021290A1?cl=en



| EP 0382526; EP 0711771; JP 1996119967; JP 2000143662; US 5047407 |

| J Org Chem 1992,57(8),2217-9 |

PAPER

 |
|
 |
|
| Systematic (IUPAC) name | |
|---|---|
|
4-amino-1-[(2R,5S)-2-(hydroxymethyl)-1,3-oxathiolan-5-yl]-1,2-dihydropyrimidin-2-one
|
|
| Clinical data | |
| Trade names | Epivir |
| AHFS/Drugs.com | monograph |
| MedlinePlus | a696011 |
| Pregnancy category |
|
| Routes of administration |
Oral |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 86% |
| Protein binding | Less than 36% |
| Biological half-life | 5 to 7 hours |
| Excretion | Renal (circa 70%) |
| Identifiers | |
| CAS Number | 134678-17-4 |
| ATC code | J05AF05 (WHO) |
| PubChem | CID 73339 |
| DrugBank | DB00709 |
| ChemSpider | 66068 |
| UNII | 2T8Q726O95 |
| KEGG | D00353 |
| ChEMBL | CHEMBL141 |
| NIAID ChemDB | 000388 |
| Synonyms | L-2′,3′-dideoxy-3′-thiacytidine |
| PDB ligand ID | 3TC (PDBe, RCSB PDB) |
| Chemical data | |
| Formula | C8H11N3O3S |
| Molar mass | 229.26 g/mol |
DRUG REGULATORY AFFAIRS INTERNATIONAL

Since 1st October 2012, special regulations have been applying to the US Type II Drug Master Files. This year in February, the FDA published a new Guidance for Industry. Read here what the DMF holder has to consider when submitting data about the API Drug Master File.
Since the coming into force of the “Generic Drug User Fee Act” (GDUFA) on 1st October 2012, special regulations have been applying to the submission to the FDA of a Drug Master Files for a pharmaceutical API (Type II DMF). The DMF holder must pay a one-time fee when authorising the reference of his/ her DMF in an application for a generic drug (Abbreviated New Drug Application, ANDA). Moreover, the DMF will undergo a completeness assessment through the FDA.
This year in February, the FDA published a Guidance for Industry entitled “Completeness Assessments for Type II API DMFs under GDUFA”…
View original post 350 more words


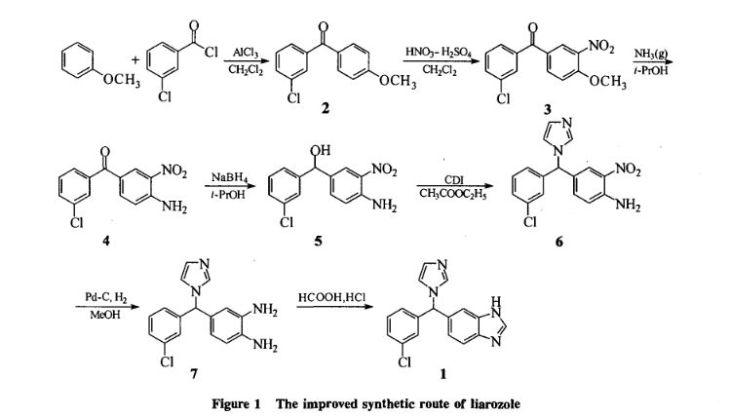
Liarozole fumarate is prepared as shown in Scheme 20970301a. Anisol is reacted with 3-chlorobenzoyl chloride (I) under Friedel-Craft conditions to give (3-chlorophenyl)(4-methoxyphenyl)methanone (II). Nitration of (II) is carried out in dichloromethane at 10 C to yield (III). The methoxy group in (III) is replaced by the amino group by means of NH3 in 2-propanol at 100 C under pressure, giving (IV). By reduction of the keto function of (IV) with sodium borohydride in 2-propanol, the corresponding alcohol (V) is obtained, which upon treatment with 1,1′-carbonyldiimidazole in refluxing dichloromethane yields the imidazolyl compound (VI). Hydrogenation of the nitro group in (VI), followed by cyclization of (VII) in a refluxing mixture of formic acid and 4N hydrochloric acid, gives the benzimidazole derivative (VIII). Finally, the treatment of (VIII) with fumaric acid in ethanol yields liarozole fumarate (IX).
http://www.google.com/patents/WO1995022540A1?cl=en
Liarozole is a racemic mixture, i.e. a mixture of its optical isomers, and is specifically mentioned as compound 28 in EP-0,371,559. Said patent application mentions the use of compounds like liarozole in the treatment of epithelial disorders. EP-0,260,744 describes the use of compounds like liarozole for inhibiting or lowering androgen formation. Whereas EP-0,371,559 and EP-0,260,744 recognize that compounds like liarozole have stereochemically isomeric forms, no example of an enantiomerically pure form is given of liarozole.
Chemically liarozole is (±)-5-[3-chlorophenyl]-lH-imidazol-l-ylmethyl]-lH-benz- imidazole, and is represented by formula (I). As can be seen from the chemical structure, liarozole has one stereogenic center (indicated with an asterisk in formula (I)).
The subject of this invention is the enantiomerically pure dextrorotatory isomer or (+)-isomer of liarozole. Said isomer will hereinafter be referred to as (+)-liarozole. Many organic compounds exist in optically active forms, i.e. they have the ability to rotate the plane of plane-polarized light. In describing an optically active compound, the prefixes D and L or R and S are used to denote the absolute configuration of the molecule about its chiral center(s). The prefixes (+) and (-) or d and 1 are employed to designate the sign of rotation of plane-polarized light by the compound, with (-) or 1 meaning that the compound is iaevorotatory and with (+) or d meaning that the compound is dextrorotatory. For a given chemical structure the optically active isomers having an opposite sign of optical rotation are called enantiomers. Said enantiomers are identical except that they are mirror images of one another. A 1: 1 -mixture of such enantiomers is called a racemic mixture.
General preparation of structures including liarozole have been extensively described in EP-0,371,559 and EP-0,260,744.
Enantiomerically pure (+)-liarozole may be prepared by reacting an enantiomerically pure intermediate diamine of formula (B)-(II) with formic acid or a functional derivative thereof.
Said functional derivative of formic acid is meant to comprise the halide, anhydride, amide and ester, including the ortho and imino ester form thereof. Also methanimidamide or an acid addition salt thereof can be used as cyclizing agent.
The general reaction conditions, work-up procedures and conventional isolation techniques for carrying out the above and following reactions are described in the prior art. When more specific conditions are required they are mentioned hereinunder. The enantiomerically pure intermediate diamine of formula (B)-(II) may be prepared by reducing an intermediate of formula (B)-(iπ) by a standard nitro-to-amine reduction reaction.
The desired enantiomer of the intermediate of formula (B)-(]H) can be prepared by fractional crystallization of a racemic mixture of the intermediate of formula (HI) with an enantiomerically pure chiral acid. Preferred chiral acid for the above fractional crystallization is 7,7-dimethyl-2-oxobicyclo[2.2.1]heptane-l-methanesulfonic acid (i.e. 10-camphorsulfonic acid).
Appropriate solvents for carrying out said fractional crystallization are water, ketones, e.g. 2-propane, 2-butanone; alcohols, e.g. methanol, ethanol, 2-propanol. Mixtures of ketones and water are very suitable for the above fractional crystallization. Preferably a mixture of 2-propanone and water is used.
The ratio of water/2-propanone by volume may vary from 1/10 to 1/2. Preferred range of said ratio is 1/5 to 1/3.
The fractional crystallizations are suitably carried out below room temperature, preferably below 5°C.
It was also found that the subsequent reaction step can be carried out without any appreciable racemization.
Alternatively the (+)-isomer of the compound of formula (I) may be prepared by cyclizing an intermediate of formula (B)-(IV) following procedures as described above for the cyclization of intermediates of formula (B)-(II) and desulfurating the thus obtained intermediate of formula (B)-(V). In formulas (B)-(TV) and (B)-(V) R represents Ci^alkyl, wherein Ci-^alkyl means a straight or branch chained saturated hydrocarbon radicals having 1 to 6 carbon atoms such as, for example, methyl, ethyl, propyl, butyl, pentyl, hexyl. Preferably R is methyl.
The intermediates of formula (B)-(IV) may be prepared by reacting an intermediate of formula (B)-(VI) with a reagent of formula (VII), alkylating the thus formed thiourea derivative of formula (B)-(VIII) subsequently cyclizing the intermediate of formula
(B)-(D ), and reducing the nitro group of the intermediate (B)-(X). In the formulas
(Vπ), (B)-(Vm), (B)-(IX) and (B)-(X) R represents Ci^alkyl as defined hereinabove.
S OR
(B)-(IV)
Experimental part
A. Preparation of the intermediates
Example 1 a) A heterogeneous mixture of (±)-4-[(3-chlorophenyl)-lH-imidazol-l-ylmethyl]-2- nitrobenzenamine (the preparation of which is described in EP-371,559) (500 g) in
2-propanone (2000 ml) and water (100 ml) was stirred at 22°C. (-)-(lR)-7,7-dimethyl- 2-oxo-bicyclo[2.2.1]heptane-l-methanesulfonic acid (353.2 g) was added and the mixture became homogeneous after 10 minutes. The mixture was first stirred for 18 hours at 20°C and then for 3 hours at 0-5°C. The precipitate was filtered off, washed with 2-propanone/water 95/5 (150 ml) and dried, yielding 308.9 g (36.2%) of product A sample (306.7 g) was partitioned between dichloromethane (500 ml) and water (750 ml). Ammonium hydroxide (100 ml) was added. This mixture was stirred for 15 minutes. The aqueous layer was separated and extracted twice with dichloromethane (250 ml each time). The separated organic layer was washed with water (250 ml), dried, filtered and the solvent was evaporated, yielding 179.7 g of (-)-(B)-4-[(3-chlorophenyl)-
20 lH-imidazol-l-ylmethyl]-2-nitrobenzenamine; mp. 89.8°C; [α]D = -19.80° (c = 0.5% in methanol) (interm. 1). b) A mixture of intermediate (1)(179.7 g) in methanol (656 ml) and a solution of ammonia in methanol (32.7 ml) was hydrogenated at 20-25 °C with platinum on activated carbon (13.1 g) as a catalyst in the presence of thiophene (0.27 g). After uptake of hydrogen (3 eq.) the catalyst was filtered off and washed with 2-propanol (30 ml). A solution of hydrochloric acid in 2-propanol (522 ml) was added to the filtrate at <30°C. The mixture was stirred for 3 hours at 20 °C, then for 3 hours at 0-5 °C. The resulting precipitate was slowly filtered off, washed with methanol (100 ml) and dried
(50 °C), yielding 185.60 g (83.2%) (+)-(B)-4-[(3-chlorophenyl)-lH-imidazol-l-yl-
20 methyl]- 1,2-benzenediamine trihydrochloride; mp. 172.5°C; [α^ = +23.73° (c = 1% in methanol) (interm. 2).
Example 2 a) A mixture of (4-amino-3-nitrophenyl) (3-chlorophenyl)methanone (50 g), formamide (375 ml) and formic acid (63 ml) was stiιτed and refluxed for 17 hours. After cooling, the mixture was poured on ice. The precipitate was filtered off and dried, yielding 55 g (99.4%) of (±)-N-[(4-amino-3-nitrophenyl) (3-chlorophenyl)methyl]formamide (interm. 3). b) A mixture of intermediate (3) (50.7 g), hydrochloric acid 6N (350 ml) and 2-propanol (70 ml) was stirred and refluxed for 17 hours. The yellow precipitate was filtered off and dried in vacuo, yielding 51 g (97.8%) of (±)-4-amino-α-(3-chloro- phenyl)-3-nitrobenzenemethanamine monohydrochloride; mp. 263°C (interm.4). c) To a solution of intermediate (4) (43 g) in tetrahydrofuran (400 ml) at room temperature was added succesively N,N-diethylethanamine (13.8 g) and (R)-(-)-α- hydroxybenzeneacetic acid (20.8 g). Then a solution of 1-hydroxybenzotriazole monohydrate (22.2 g) in tetrahydrofuran (200 ml) was added. After complete addition a solution of N,N’-dicyclohexylcarbodiimide (33.9 g) in dichloromethane (300 ml) was introduced to the mixture. After stirring for 2 hours at room temperature N,N’- dicyclohexylurea was filtered off. The filtrate was washed with a solution of potassium carbonate (10%) and the organic layer was dried to give a mixture of diastereomers (60g) (fraction 1). The same experiment with intermediate (4) (16 g) as starting material resulted in a yield of 26 g of a mixture of diastereomers (fraction 2). Fraction 1 and 2 were combined and purified by HPLC (eluent : CH2θ2/ethyl acetate 90:10), yielding 30g (32.3%) of (±)-(R,B)-N-[(4-amino-3-nitrophenyl)(3-chlorophenyl)methyl]-α- hydroxybenzeneacetamide (interm.5). d) A mixture of intermediate (5) (30 g), hydrochloric acid 12N (300 ml) and 1-propanol (100 ml) was stirred and refluxed for 17 hours and poured on ice. The mixture was extracted with ethyl acetate. The aqueous phase was basified with ammonium hydroxide and extracted with dichloromethane. The dichloromethane extracts were dried, filtered and evaporated, yielding 7.3 g (36.0%) of (+)-(B)-4-amino-α-(3-chlorophenyl)-3- nitrobenzenemethanamine (interm. 6). e) A mixture of intermediate (6) (7.3 g), 2-isothiocyanato-l,l-dimethoxyethane (4.8 g) and methanol (75 ml) was stirred and refluxed for 2 hours. The mixture was evaporated to an oily residue, yielding 11 g (100%) of (+)-(B)-N-[(4-amino-3-nitrophenyl)(3- chlorophenyl)methyl]-N’-(2,2-dimethoxyethyl)thiourea (interm.7). f) A mixture of intermediate (7) (11 g), iodomethane (2 ml) and potassium carbonate (4.97 g) was stirred at room temperature for 48 hours. The solvent was evaporated and the residue was taken off with dichloromethane and washed with water. The organic layer was dried, filtered and evaporated, yielding 11.4 g of (+)-(S)-methyl (B)-N- [(4-amino-3-nitrophenyl)(3-chlorophenyl)methyl]-N’-(2,2-dimethoxyethyl)carbam- imidothioate as an oily residue (interm. 8). g) To intermediate (8) (11.4 g) at 0°C was added sulfuric acid (100ml) (precooled to 5°C). The mixture was stirred at 5°C until complete dissolution and then was warmed to room temperature. After stirring for 2 hours, the solution was poured on ice and basified with ammonium hydroxide. The aqueous solution was extracted with ethyl acetate. The organic layer was dried, filtered and evaporated. The residue was purified by column chromatography (eluent : CH2CI2/CH3OH 98:2). The eluent of the desired fraction was evaporated, yielding 3.7 g (38.0%) of (+)-(B)-4-[(3-chlorophenyl)[2-(methylthio)-lH- imidazol-l-yl]methyl]-2-nitrobenzenamine (interm.9). h) A mixture of intermediate (9) (6.2 g), Raney nickel (6 g) and methanol (100 ml) was hydrogenated for 2 hours at 2 bar and at room temperature. After the calculated amount of hydrogen was taken up, the catalyst was filtered off. The filtrate, (+)-(B)-4-[(3- chlorophenyl)[2-(methylthio)-lH-imidazol-l-yl]methyl]-l,2-benzenediamine (interm. 10), was used for the next step. i) A mixture of intermediate (10) (5.7 g), methanimidamide monoacetate (5.2 g) and methanol (100 ml) was stirred and refluxed for 3 hours. The reaction mixture was evaporated and the residue was taken off in dichloromethane and washed with sodium hydrogen carbonate (10%). The organic layer was dried, filtered and evaporated. The oily residue was purified by column chromatography (eluent : CH2CI2/CH3OH 95:5). The eluent of the desired fraction was evaporated, yielding 4.9 g (83.7%) of (+)-(B)-5-[(3-cWorophenyl)[2-(methylthio)-lH-imidazol-l-yl]methyl]-lH-benzimidazole (interm. 11).
B. Preparation of the final compounds Example 3
A mixture of intermediate (2) (185 g) in water (512 ml) was stirred at 20 °C. Hydrochloric acid (289 ml) was added. Formic acid (85%) (61.17 ml) was added and this mixture was heated to 55°C. The reaction mixture was stirred for 3 hours at 55 °C and then cooled to 20°C. Dichloromethane (1223 ml) was added. Ammonium hydroxide (730 ml) was added dropwise at < 25°C. The separated organic layer was washed with water (500 ml), dried, filtered and the solvent was evaporated, yielding 152.88 g (108.5%) of product. A sample was dried (18 hours at 55 °C), yielding 3.18 g of (+)-(B)-5-[(3-chlorophenyl)-lH-imidazol-l-ylmethyl]-lH-benzimidazole; mp.
20 113.7°C; [αjj = +43.46° (c = 1% in methanol) (comp. 1).
Example 4
A mixture of intermediate (11) (4.9 g), Raney nickel (2 g) and ethanol (100ml) was stirred and refluxed for 5 days, while every day an additional amount of Raney nickel (2 g) was added. The catalyst was filtered off and rinsed with dichloromethane. The filtrate was evaporated and the residue was purified twice by column chromatography (silica gel; CH2CI2/CH3OH 95:5 ; CH2CI2/CH3OH NH4OH 80:20:3). The eluent of the desired fraction was evaporated and the residue was converted into the hydrochloride salt in 2-propanol and ethanol. The salt was recrystallized from 2-butanone, yielding 1.8 g (37.2%) of (+)-(B)-5-[(3-chlorophenyl)(lH-imidazol-l-yl)methyl]-lH-benzimidazole
20 monohydrochloride; mp. 212.1°C; [α]D = +42.43° (c = 1% in ethanol) (comp. 2)
Example 5
Compound (1) (149.7 g) was dissolved in 2-butanone (2424 ml). A mixture of hydrochloric acid in 2-propanol (82.6 ml) in 2-butanone (727 ml) was added over a 2 hour period at 20 °C. The reaction mixture was stirred for 16 hours at 20 °C. The precipitate was filtered off, washed with 2-butanone (242 ml) and dried (vacuum; 80°C); yielding 147.5 g (99.3%) of (+)-(B)-5-[(3-chlorophenyl)-lH-imidazol-l-ylmethyl]-lH-
20 benzimidazole monohydrochloride; mp. 214.5°C; [α] j = +36.20° (c = 1% in methanol) (comp. 2). Example 6
A mixture of compound (1) (0.72 g) in ethanol (5.1 ml; denaturated) was stirred at 20 °C until it became homogeneous. (E)-2-butenedioic acid (0.54 g) was added The mixture was stirred for 18 hours at 20 °C and then cooled 0-5 °C and precipitation resulted. More denaturated ethanol (2 ml) was added and the mixture was stirred for 2 hours at 20 °C. The precipitate was filtered off, washed with ethanol (3 ml; denaturated) and dried (vacuum; 50 °C), yielding 0.26 g (23.4%) (B)-5-[(3-chlorophenyl)-lH-imidazol-l-yl- methyl]-lH-benzimidazole (E)-2-butenedioate (2:3).ethanolate (2:1); mp. 111.2°C (comp. 3).
PAPER

Paper
1 Vahlquist, A; Blockhuys, S; Steijlen, P; Van Rossem, K; Didona, B; Blanco, D; Traupe, H (2013). “Oral liarozole in the treatment of patients with moderate/severe lamellar ichthyosis: Results of a randomized, double-blind, multinational, placebo-controlled phase II/III trial”. The British journal of dermatology 170 (1): n/a. doi:10.1111/bjd.12626. PMID 24102348.
Literature References: Inhibits cytochrome P450-dependent enzymes involved in steroid biosynthesis and retinoic acid catabolism. Prepn: A. H. M. Raeymaekers et al., EP 260744; eidem, US 4859684 (1988, 1989 both to Janssen). In vivo antitumor activity: R. Van Ginckel et al., Prostate 16, 313 (1990). Pharmacology and effect on steroid synthesis: J. Bruynseels et al., ibid., 345; and effect on retinoic acid: R. De Coster et al., J. Steroid Biochem. Mol. Biol. 43, 197 (1992). Clinical evaluation in prostate cancer: C. Mahler et al., Cancer 71, 1068 (1993); in psoriasis: P. Dockx et al., Br. J. Dermatol. 133, 426 (1995); in combination therapy for malignant brain tumors: M. E. Westarp et al., Onkologie 16, 22 (1993).
 |
|
| Names | |
|---|---|
| IUPAC name
6-[(3-Chlorophenyl)-imidazol-1-ylmethyl]-1H-benzimidazole
|
|
| Identifiers | |
| 115575-11-6 | |
| ChemSpider | 54664 |
| 5210 | |
| Jmol interactive 3D | Image |
| PubChem | 60652 |
| Properties | |
| C17H13ClN4 | |
| Molar mass | 308.77 g·mol−1 |
///////
C1=CC(=CC(=C1)Cl)C(C2=CC3=C(C=C2)N=CN3)N4C=CN=C4

ARN-509; cas 956104-40-8; ARN 509; UNII-4T36H88UA7;
ARN-509; JNJ-56021927; JNJ-927\
Phase III Prostate cancer
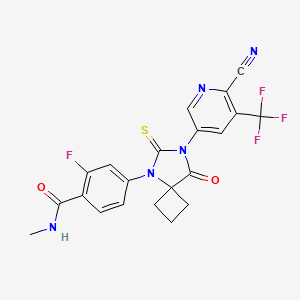
4-(7-(6-CYANO-5-(TRIFLUOROMETHYL)PYRIDIN-3-YL)-8-OXO-6-THIOXO-5,7-DIAZASPIRO[3.4]OCTAN-5-YL)-2-FLUORO-N-METHYLBENZAMIDE;
4-(7-(6-cyano-5-(trifluoroMethyl)pyridin-3-yl)-8-oxo-6-thioxo-5,7-diazaspirooctan-5-yl)-2-fluoro-N-MethylbenzaMide;
| Molecular Formula: | C21H15F4N5O2S |
|---|---|
| Molecular Weight: | 477.434713 g/mol |
| Product Name | Sponsor Only | Condition | Start Date | End Date | Phase | Last Change Date |
|---|---|---|---|---|---|---|
| ARN-509 | Aragon Pharmaceuticals Inc | Hormone refractory prostate cancer | 31-JUL-10 | 30-JUN-13 | Phase 2 | 17-SEP-13 |
| Aragon Pharmaceuticals Inc | 31-MAR-13 | 30-JUN-13 | Phase 1 | 17-SEP-13 | ||
| Aragon Pharmaceuticals Inc | Hormone refractory prostate cancer | 31-OCT-13 | 31-DEC-16 | Phase 3 | 05-NOV-13 | |
| Aragon Pharmaceuticals Inc; Johnson & Johnson | Hormone refractory prostate cancer | 28-FEB-13 | 01-FEB-14 | Phase 1 | 07-OCT-13 | |
| Aragon Pharmaceuticals Inc | Hormone dependent prostate cancer | 28-FEB-13 | 28-FEB-18 | Phase 2 | 18-OCT-13 |
Apalutamide, also known as ARN-509 and JNJ-56021927 , is an androgen receptor antagonist with potential antineoplastic activity. ARN-509 binds to AR in target tissues thereby preventing androgen-induced receptor activation and facilitating the formation of inactive complexes that cannot be translocated to the nucleus. This prevents binding to and transcription of AR-responsive genes. This ultimately inhibits the expression of genes that regulate prostate cancer cell proliferation and may lead to an inhibition of cell growth in AR-expressing tumor cells.
Apalutamide (INN) (developmental code name ARN-509, also JNJ-56021927) is a non-steroidal antiandrogen that is under development for the treatment of prostate cancer.[1] It is similar to enzalutamide both structurally and pharmacologically,[2] acting as a selective competitive antagonist of the androgen receptor (AR), but shows some advantages, including greater potency and reduced central nervous system permeation.[1][3][4] Apalutamide binds weakly to the GABAA receptor similarly to enzalutamide, but due to its relatively lower central concentrations, may have a lower risk of seizures in comparison.[1][3][5] The drug has been found to be effective and well-tolerated in clinical trials thus far,[2][4] with the most common side effects reported including fatigue, nausea, abdominal pain, and diarrhea.[6][3][5] Apalutamide is currently in phase III clinical trials for castration-resistant prostate cancer.[7]
Recently, the acquired F876L mutation of the AR identified in advanced prostate cancer cells was found to confer resistance to both enzalutamide and apalutamide.[8][9] A newer antiandrogen, ODM-201, is not affected by this mutation, nor has it been found to be affected by any other tested/well-known AR mutations.[10]
Apalutamide may be effective in a subset of prostate cancer patients with acquired resistance to abiraterone acetate.[2]
The chemical structure of ARN-509 is very similar structure to that of Enzalutamide (MDV3100) with two minor modifications: (a) two methyl groups in the 5-member ring of MDV3100 is linked by a CH2 group in ARN-509; (b) the carbon atom in the benzene ring of MDV3100 is replaced by a nitrogen atom in ARN-509. ARN-509 is considered as a Me-Too drug of Enzalutamide (MDV3100). ARN-509 was claimed to be more active than Enzalutamide (MDV3100).
ARN-509 is a novel 2nd Generation anti-androgen that is targeted to treat castration resistant prostate cancers where 1st generation anti-androgens fail. ARN-509 is unique in its action in that it inhibits both AR nuclear translocation and AR binding to androgen response elements in DNA. Importantly, and in contrast to the first-generation anti-androgen bicalutamide, it exhibits no agonist activity in prostate cancer cells that over-express AR. ARN-509 is easily synthesized, and its oral bioavailability and long half-life allow for once-daily oral dosing. In addition, its excellent preclinical safety profile makes it well suited as either a mono- or a combination therapy across the entire spectrum of prostate cancer disease states. (source: http://www.aragonpharm.com/programs/arn509.htm).
ARN-509 is a competitive AR inhibitor, which is fully antagonistic to AR overexpression, a common and important feature of CRPC. ARN-509 was optimized for inhibition of AR transcriptional activity and prostate cancer cell proliferation, pharmacokinetics and in vivo efficacy. In contrast to bicalutamide, ARN-509 lacked significant agonist activity in preclinical models of CRPC. Moreover, ARN-509 lacked inducing activity for AR nuclear localization or DNA binding. In a clinically valid murine xenograft model of human CRPC, ARN-509 showed greater efficacy than MDV3100. Maximal therapeutic response in this model was achieved at 30 mg/kg/day of ARN-509 , whereas the same response required 100 mg/kg/day of MDV3100 and higher steady-state plasma concentrations. Thus, ARN-509 exhibits characteristics predicting a higher therapeutic index with a greater potential to reach maximally efficacious doses in man than current AR antagonists. Our findings offer preclinical proof of principle for ARN-509 as a promising therapeutic in both castration-sensitive and castration-resistant forms of prostate cancer. (source: Cancer Res. 2012 Jan 20. [Epub ahead of print] )
(source: Cancer Res. 2012 Jan 20. [Epub ahead of print] )
 SYNTHESISS
SYNTHESISS

WO 2008119015
WO2011103202
WO2014190895
WO2011103202
http://www.google.com/patents/WO2011103202A2?cl=en


 Prostate cancer is one of the most common forms of cancer found in Western men and the second leading cause of cancer death in Western men. When prostate cancer is confined locally, the disease can usually be treated by surgery and/or radiation. Advanced disease is frequently treated with anti-androgen therapy, also known as androgen deprivation therapy. Administration of anti-androgens blocks androgen receptor (AR) function by competing for androgen binding; and therefore, anti-androgen therapy reduces AR activity. Frequently, such therapy fails after a time, and the cancer becomes hormone refractory, that is, the prostate cancer no longer responds to hormone therapy and the cancer does not require androgens to progress.
Prostate cancer is one of the most common forms of cancer found in Western men and the second leading cause of cancer death in Western men. When prostate cancer is confined locally, the disease can usually be treated by surgery and/or radiation. Advanced disease is frequently treated with anti-androgen therapy, also known as androgen deprivation therapy. Administration of anti-androgens blocks androgen receptor (AR) function by competing for androgen binding; and therefore, anti-androgen therapy reduces AR activity. Frequently, such therapy fails after a time, and the cancer becomes hormone refractory, that is, the prostate cancer no longer responds to hormone therapy and the cancer does not require androgens to progress.
Overexpression of AR has been identified as a cause of hormone refractory prostate cancer (Nat. Med., 10:33-39, 2004; incorporated herein by reference). Overexpression of AR is sufficient to cause progression from hormone sensitive to hormone refractory prostate cancer, suggesting that better AR antagonists than the current drugs may be able to slow the progression of prostate cancer. It has been demonstrated that overexpression of AR converts anti-androgens from antagonists to agonists in hormone refractory prostate cancer. This work explains why anti-androgen therapy fails to prevent the progression of prostate cancer.
The identification of compounds that have a high potency to anatgonize AR activity would overcome the hormone refractory prostate cancer and slowdown the progression of hormone sensitive prostate cancer. Such compounds have been identified by Sayers et al. (WO 2007/126765, published Nov. 8, 2007; which is incorporated herein by reference). One compound is known as A52, a biarylthiohydantoin, and has the chemical structure
Moilanen AM, Riikonen R, Oksala R, Ravanti L, Aho E, Wohlfahrt G, Nykänen PS, Törmäkangas OP, Palvimo JJ, Kallio PJ (2015). “Discovery of ODM-201, a new-generation androgen receptor inhibitor targeting resistance mechanisms to androgen signaling-directed prostate cancer therapies”. Sci Rep 5: 12007. doi:10.1038/srep12007. PMC 4490394. PMID 26137992
11Clegg NJ, Wongvipat J, Tran C, Ouk S, Dilhas A, Joseph J, Chen Y, Grillot K, Bischoff ED, Cai L, Aparicio A, Dorow S, Arora V, Shao G, Qian J, Zhao H, Yang G, Cao C, Sensintaffar J, Wasielewska T, Herbert MR, Bonnefous C, Darimont B, Scher HI, Smith-Jones PM, Klang M, Smith ND, de Stanchina E, Wu N, Ouerfelli O, Rix P, Heyman R, Jung ME, Sawyers CL, Hager JH. ARN-509: a novel anti-androgen for prostate cancer treatment. Cancer Res. 2012 Mar 15;72(6):1494-1503. Epub 2012 Jan 20.PubMed PMID: 22266222.
| Patent ID | Date | Patent Title |
|---|---|---|
| US2014309262 | 2014-10-16 | ANDROGEN RECEPTOR MODULATOR FOR THE TREATMENT OF PROSTATE CANCER AND ANDROGEN RECEPTOR-ASSOCIATED DISEASES |
| US2014296312 | 2014-10-02 | TREATMENT OF BREAST CANCER |
| US2014243416 | 2014-08-28 | Topical Antiandrogen Therapy for the Treatment of Becker’s Nevus |
| US8802689 | 2014-08-12 | Androgen receptor modulator for the treatment of prostate cancer and androgen receptor-associated diseases |
| US2014107085 | 2014-04-17 | Bifunctional AKR1C3 Inhibitors/Androgen Receptor Modulators and Methods of Use Thereof |
| US2014088129 | 2014-03-27 | ANTI-ANDROGENS FOR THE TREATMENT OF NON-METASTATIC CASTRATE-RESISTANT PROSTATE CANCER |
| US2013225821 | 2013-08-29 | SYNTHESIS OF THIOHYDANTOINS |
| US2013116258 | 2013-05-09 | ANDROGEN RECEPTOR MODULATORS AND USES THEREOF |
| US2011003839 | 2011-01-06 | ANDROGEN RECEPTOR MODULATOR FOR THE TREATMENT OF PROSTATE CANCER AND ANDROGEN RECEPTOR-ASSOCIATED DISEASES |
| US2010190991 | 2010-07-29 | SYNTHESIS OF THIOHYDANTOINS |
 |
|
| Systematic (IUPAC) name | |
|---|---|
|
4-[7-[6-Cyano-5-(trifluoromethyl)pyridin-3-yl]-8-oxo-6-sulfanylidene-5,7-diazaspiro[3.4]octan-5-yl]-2-fluoro-N-methylbenzamide
|
|
| Clinical data | |
| Pregnancy category |
|
| Routes of administration |
Oral |
| Identifiers | |
| CAS Number | 956104-40-8 |
| ATC code | None |
| PubChem | CID 24872560 |
| ChemSpider | 28424131 |
| Chemical data | |
| Formula | C21H15F4N5O2S |
| Molar mass | 477.434713 g/mol |
////////
CNC(=O)C1=C(C=C(C=C1)N2C(=S)N(C(=O)C23CCC3)C4=CN=C(C(=C4)C(F)(F)F)C#N)F
CNC(=O)C1=C(C=C(C=C1)N2C(=S)N(C(=O)C23CCC3)C4=CN=C(C(=C4)C(F)(F)F)C#N)F
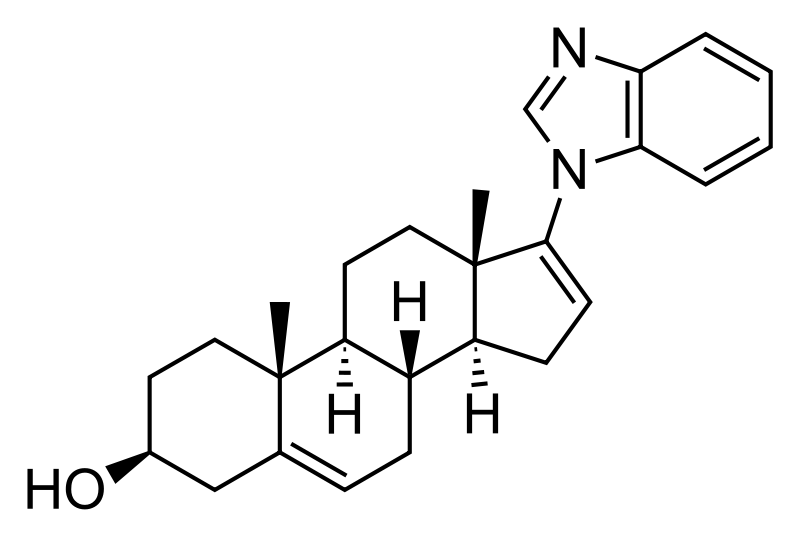
Galeterone
SYNTHESIS SEE BELOW
A SARM potentially for the treatment of prostate cancer.
Research Code, TOK-001; VN; 124; 124-1; 1241
TOK-001; Galeterone; 851983-85-2; VN/124; UNII-WA33E149SW; VN/124-1;
CAS No. 851983-85-2(Galeterone)
(3S,8R,9S,10R,13S,14S)-17-(benzimidazol-1-yl)-10,13-dimethyl-2,3,4,7,8,9,11,12,14,15-decahydro-1H-cyclopenta[a]phenanthren-3-ol
Fast track 2012 f
| Molecular Formula: | C26H32N2O |
|---|---|
| Molecular Weight: | 388.54508 g/mol |

Galeterone (TOK-001 or VN/124-1) is a novel steroidal antiandrogen under development by Tokai Pharmaceuticals for the treatment of prostate cancer. It possesses a unique dual mechanism of action, acting as both an androgen receptor antagonist and an inhibitor of CYP17A1, an enzyme required for the biosynthesis of the androgens.[1] It shows selectivity for 17,20-lyase over 17-hydroxylase.[2]
As of 2016, galeterone is being compared to enzalutamide in a phase III clinical trial (ARMOR3-SV) for AR-V7-expressing metastatic castration-resistant prostate cancer.[3][4]
Specific Androgen Receptor Modulator CYP17 Inhibitor TOK-001 is an orally bioavailable small-molecule androgen receptor modulator and CYP17 lyase inhibitor with potential antiandrogen activity. Galeterone exhibits three distinct mechanisms of action: 1) as an androgen receptor antagonist, 2) as a CYP17 lyase inhibitor and 3) by decreasing overall androgen receptor levels in prostate cancer tumors, all of which may result in a decrease in androgen-dependent growth signaling. Localized to the endoplasmic reticulum (ER), the cytochrome P450 enzyme CYP17 (P450C17 or CYP17A1) exhibits both 17alpha-hydroxylase and 17,20-lyase activities, and plays a key role in the steroidogenic pathway that produces progestins, mineralocorticoids, glucocorticoids, androgens, and estrogens.
Tokai’s lead product candidate is galeterone, a highly-selective, oral small molecule with the potential to transform the treatment of prostate cancer. We are focusing our late-stage development of galeterone on the treatment of men with metastatic, castration-resistant prostate cancer, or CRPC, whose prostate tumor cells express the AR-V7 splice variant.
We are conducting ARMOR3-SV, a Phase 3 clinical trial of galeterone evaluating whether administration of galeterone results in a statistically significant increase in radiographic progression-free survival as compared to Xtandi® (enzalutamide), an oral therapy currently approved for the treatment of CRPC, in AR-V7 positive metastatic CRPC patients. ARMOR3-SV is the first pivotal trial in prostate cancer to employ a precision medicine approach for patient selection. For more information regarding ARMOR3-SV, click here.
Galeterone has been studied in over 250 subjects in Phase 1 and Phase 2 clinical trials, including in CRPC patients with and without the AR-V7 splice variant. In these trials, galeterone demonstrated good tolerability and showed clinically meaningful reductions in levels of prostate specific antigen, or PSA, a biochemincal marker used to evaluate prostate cancer patients for signs of response to therapy.
We are currently focusing our late-stage development of galeterone on AR-V7 positive metastatic CRPC patients because it represents an unmet need in prostate cancer and our precision medicine approach provides an efficient development path. Based on the data we and our collaborators have produced to date, we also believe there is rationale for the broader clinical exploration of galeterone in the future.
Galeterone acts by disrupting the androgen receptor signaling pathway. This pathway is activated by the binding of male hormones (also known as androgens), such as testosterone and dihydrotestosterone (DHT) to androgen receptors in prostate cancer cells.
Galeterone disrupts the activation of the androgen receptor pathway in three ways:
Tokai retains global rights to galeterone. We intend to commercialize galeterone in the United States on our own, and to seek a partner to further develop and commercialize galeterone outside of the United States.
Galeterone has been granted Fast Track designation by U.S. Food and Drug Administration for the treatment of CRPC. Fast Track designation is designed to facilitate the development and expedite review of drugs intended to treat serious or life-threatening conditions and that demonstrate the potential to address unmet medical needs.

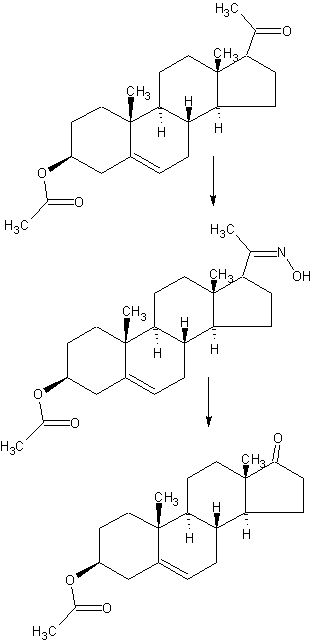
DETAILED DESCRIPTION
1J loss reaction.
(1) raw material specifications to match.
acetate pregnancy dehydropregnenolone: toluene + ethanol: Batch steep: hydrochloric acid amine light = 1: 3: 0 4: 0.213, which pregnenolone acetate pregnancy 160kg, toluene + ethanol 320kg + 160kg, approved Steep 64kg, hydrochloric acid amine light 34kg.
(2) process operation.
In the first input 1000L tank oximation with hydroxylamine hydrochloride in pyridine, and then pumped into a mixed solvent of toluene and ethanol, the reaction solution was stirred and heated to complete dissolution, pregnancy-dehydropregnenolone acetate was added and heated under reflux for 3 hours, cooling and crystallization, The Department conducted into the centrifuge centrifugal drying, apply a recovery from the mother liquor, rinse with warm water mixture to no foam, centrifugal drying, drying to a moisture at 0.2% or less, that acetic acid in pregnancy dehydropregnenolone oxime (oxime compounds) 163kg, content of 99%, a melting point of 202-204 ° C, a yield of about 102% (for pregnenolone acetate pregnancy weight ratio).
2, heavy drain hydrolysis reaction.
(1) raw material specifications to match.
acetate pregnancy dehydropregnenolone waning: Benzene: Batch steep: phosphorus oxychloride and toluene: HCl + water = 1: 6 5: 0 4: 1: 3.5, which acetate pregnancy alcohol one hand 163kg, benzene 1060kg, batch steep 64kg, phosphorus oxychloride and toluene 80kg + 80kg, hydrochloric acid + water 245kg + 325kg.
(2) process operation.
The first drying 2000L rearrangement reaction tank, then pumped to the reaction tank benzene, alcohol into acetate pregnancy oxime, pulls out into benzene, stirring heated to reflux until the reaction mixture is completely dissolved, cooling to 1 (TC When, pyridine, of the reaction liquid at temperatures down to 6 ° C, start dropping a mixed solution of previously prepared phosphorus oxychloride and toluene (1: 1 mass ratio), slowly dropping, dropping control, first After slow fast reaction when dropping liquid temperature control in 4-8 ° C, the addition was complete, the reaction solution at 9-12 ° C for 3 hours the first time under.
After incubation, a solution has been a mixed solution of hydrochloric acid and water, good preparation, while dropping the reaction liquid temperature is controlled at 15-25 ° C, the addition was complete, the reaction solution at 15-25 ° C under a second Insulation 1. 5-2 hours. After incubation, stand 40 minutes, then points to lower acidic water layer, the remaining upper layer was added 0.3 times the amount of 30-35 ° C in the brine and let stand 20 minutes, a second watershed, sub lower aqueous layer was then allowed to stand for 30 minutes, a third water diversion, to give the final weight of the upper layer reaction solution was drained.
3, the red Dingding steam distillate process.
The rearrangement reaction liquid was pumped to punch distillate tank, conduct atmospheric distillate punch, has been rushed to the reaction mixture was distilled benzene mixed solvent only, at the start of the steam valve not to open too much, so as not to rush material, distillation after cooling discharge, centrifugal drying, washing with tap water to neutral, and then into the oven dried to a moisture in the square. 5% acetic acid in dehydroepiandrosterone (rearrangement thereof) The crude product is about 142kg, content of about 97.5%, a melting point of 160 ° C _165 ° C or so, yield about 88% (for acetate pregnancy dehydropregnenolone weight ratio).
4, refining processes.
The drying in acetic acid Dehydroepiandrosterone crude into refined tin, adding 8 times the weight of the crude methanol and 0.10 times the weight of activated carbon, heat, stirring to dissolve, reflux billion. 5 hours, filtered , concentrated, cooled to about 5 ° C, the discharge


| Patent ID | Date | Patent Title |
|---|---|---|
| US2011034428 | 2011-02-10 | Treatment of Prostate Cancer |
| US7875599 | 2011-01-25 | C-17-heteroaryl steroidal CYP17 inhibitors/antiandrogens, in vitro biological activities, pharmacokinetics and antitumor activity |
| US2010137269 | 2010-06-03 | Novel C-17-Heteroaryl Steroidal Cyp17 Inhibitors/Antiandrogens: Synehesis, In Vitro Biological Activities, Pharmacokinetics and Antitumor Activity |
| US2010048914 | 2010-02-25 | Novel C-17-Heteroaryl Steroidal Cyp17 Inhibitors/Antiandrogens, In Vitro Biological Activities, Pharmacokinetics and Antitumor Activity |
| US2010048913 | 2010-02-25 | Novel C-17-Heteroaryl Steroidal CYP17 Inhibitors/Antiandrogens Synthesis In Vitro Biological Activities, Pharmacokinetics and Antitumor Activity |
| US2010048912 | 2010-02-25 | Novel C-17-Heteroaryl Steroidal CYP17 Inhibitors/Antiandrogens, In Vitro Biological Activities, Pharmacokinetics and Antitumor Activity |
| US2010048524 | 2010-02-25 | Novel C-17-Heteroaryl Steroidal CYP17 Inhibitors/Antiandrogens Synthesis In Vitro Biological Activities, Pharmacokinetics and Antitumor Activity |
| US2010047338 | 2010-02-25 | Novel C-17-Heteroaryl Steroidal CYP17 Inhibitors/Antiandrogens, In Vitro Biological Activities, Pharmacokinetics and Antitumor Activity |
| Patent ID | Date | Patent Title |
|---|---|---|
| US2013336962 | 2013-12-19 | AZIRIDINE BISPHENOL ETHERS AND RELATED COMPOUNDS AND METHODS FOR THEIR USE |
| US8569393 | 2013-10-29 | UV-LED curable compositions and inks |
| US2013203615 | 2013-08-08 | ANTIANDROGEN THERAPY MONITORING METHODS AND COMPOSITIONS |
| US2012309861 | 2012-12-06 | PHOTOINITIATORS FOR UV-LED CURABLE COMPOSITIONS AND INKS |
| US2012237502 | 2012-09-20 | METHOD FOR TREATING BREAST CANCER AND OVARIAN CANCER |
| US2011319369 | 2011-12-29 | COMBINATION OF A 17 ALPHA-HYDROXYLASE/C17, 20-LYASE INHIBITOR WITH AN ADDITIONAL THERAPEUTIC AGENT |
| US2011312924 | 2011-12-22 | NOVEL STEROIDAL CYP17 INHIBITORS/ANTIANDROGENS |
| US2011312916 | 2011-12-22 | NOVEL PRODRUGS OF STEROIDAL CYP17 INHIBITORS/ANTIANDROGENS |
| US2011118219 | 2011-05-19 | NOVEL PRODRUGS OF C-17-HETEROARYL STEROIDAL CYP17 INHIBITORS/ANTIANDROGENS: SYNTHESIS, IN VITRO BIOLOGICAL ACTIVITIES, PHARMACOKINETICS AND ANTITUMOR ACTIVITY |
| US2011105445 | 2011-05-05 | ANDROGEN RECEPTOR INACTIVATION CONTRIBUTES TO ANTITUMOR EFFICACY OF CYP17 INHIBITORS IN PROSTATE CANCER |
| Patent ID | Date | Patent Title |
|---|---|---|
| US2015051179 | 2015-02-19 | NOVEL STEROIDAL CYP17 INHIBITORS/ANTIANDROGENS |
| US2015005265 | 2015-01-01 | METHODS AND COMPOSITIONS FOR COMBINATION THERAPY USING P13K/MTOR INHIBITORES |
| US2014371261 | 2014-12-18 | INDOMETHACIN ANALOGS FOR THE TREATMENT OF CASTRATE-RESISTANT PROSTATE CANCER |
| US2014371181 | 2014-12-18 | NOVEL PRODRUGS OF STEROIDAL CYP17 INHIBITORS/ANTIANDROGENS |
| US2014343024 | 2014-11-20 | TREATMENT OF PROSTATE CANCER |
| US2014288037 | 2014-09-25 | NOVEL COMPOSITIONS AND METHODS FOR TREATING PROSTATE CANCER |
| US2014288036 | 2014-09-25 | NOVEL C-17-HETEROARYL STEROIDAL CYP17 INHIBITORS/ANTIANDROGENS, IN VITRO BIOLOGICAL ACTIVITIES, PHARMACOKINETICS AND ANTITUMOR ACTIVITY |
| US2014274983 | 2014-09-18 | NOVEL PRODRUGS OF C-17-HETEROARYL STEROIDAL CYP17 INHIBITORS/ANTIANDROGENS: SYNTHESIS, IN VITRO BIOLOGICAL ACTIVITIES, PHARMACOKINETICS AND ANTITUMOR ACTIVITY |
| US2014107085 | 2014-04-17 | Bifunctional AKR1C3 Inhibitors/Androgen Receptor Modulators and Methods of Use Thereof |
| US2013336962 | 2013-12-19 | AZIRIDINE BISPHENOL ETHERS AND RELATED COMPOUNDS AND METHODS FOR THEIR USE |


| CN101691392A * | Sep 17, 2009 | Apr 7, 2010 | 扬州市天平化工厂有限公司 | Method for preparing 3beta-acetoxyl group-5androstene-17ketone |
| CN102212099A * | Apr 2, 2011 | Oct 12, 2011 | 邵阳市科瑞化学品有限公司 | Synthesis method for dehydroepiandrosterone |
| CN102603839A * | Jan 13, 2012 | Jul 25, 2012 | 宜城市共同药业有限公司 | Preparation method of dehydroepiandrosterone |
| CN102746356A * | Jul 17, 2012 | Oct 24, 2012 | 湖北芳通药业股份有限公司 | Process for producing dehydroepiandrosterone acetate through homogeneous phase method |
| Reference | ||||
|---|---|---|---|---|
| 1 | * | 石诚等: “5-雄甾烯-3β-醇-17-酮-3-醋酸酯的工艺研究“, 《山东化工》, vol. 41, no. 1, 31 December 2012 (2012-12-31) | ||
| 2 | * | 石诚等: “醋酸妊娠双烯醇酮肟的工艺研究“, 《广州化工》, vol. 39, no. 23, 31 December 2011 (2011-12-31), pages 78 – 79 | ||
Silberstein, John L.; Taylor, Maritza N.; Antonarakis, Emmanuel S. (2016-04-01). “Novel Insights into Molecular Indicators of Response and Resistance to Modern Androgen-Axis Therapies in Prostate Cancer”. Current Urology Reports 17 (4): 29. doi:10.1007/s11934-016-0584-4. ISSN 1534-6285. PMID 26902623.
 |
|
| Systematic (IUPAC) name | |
|---|---|
|
17-(1H-benzimidazol-1-yl)androsta-5,16-dien-3β-ol
|
|
| Clinical data | |
| Routes of administration |
Oral |
| Identifiers | |
| CAS Number | 851983-85-2 |
| PubChem | CID 11188409 |
| ChemSpider | 9363493 |
| KEGG | D10125 |
| Chemical data | |
| Formula | C26H32N2O |
| Molar mass | 388.25 |
///////
C[C@]12CC[C@@H](CC1=CC[C@@H]3[C@@H]2CC[C@]4([C@H]3CC=C4N5C=NC6=CC=CC=C65)C)O
CC12CCC(CC1=CCC3C2CCC4(C3CC=C4N5C=NC6=CC=CC=C65)C)O

IC200214; ITI-214
(6aR,9aS)-2-(4-(6-fluoropyridin-2-yl)benzyl)-5-methyl-3-(phenylamino)-5,6a,7,8,9,9a-hexahydrocyclopenta[4,5]imidazo[1,2-a]pyrazolo[4,3-e]pyrimidin-4-(2H)-one phosphate
(6aR,9aS)-5-methyl-3-(phenylamino)-2-(4-(6-fluoropyridin-2-yl)-benzyl)-5,6a,7,8,9,9a-hexahydrocyclopent[4,5]imidazo[1,2-a]pyrazolo[4,3-e]pyrimidin-4(2H)-one…BASE
CAS: 1642303-38-5 (phosphate);
1160521-50-5 (free base).
Chemical Formula: C29H29FN7O5P
Molecular Weight: 605.5672
Takeda Pharmaceutical Company Limited,Intra-Cellular Therapies, Inc.
ITI-214 is an orally active, potent and Selective Inhibitors of Phosphodiesterase 1 for the Treatment of Cognitive Impairment Associated with Neurodegenerative and Neuropsychiatric Diseases. ITI-214 exhibited picomolar inhibitory potency for PDE1, demonstrated excellent selectivity against all other PDE families, and showed good efficacy in vivo. Currently, this investigational new drug is in Phase I clinical development and being considered for the treatment of several indications including cognitive deficits associated with schizophrenia and Alzheimer’s disease, movement disorders, attention deficit and hyperactivity disorders, and other CNS and non-CNS disorders.
Phosphodiesterase-1 (PDE-1) inhibitor
which is a picomolar PDE1 inhibitor with excellent selectivity against other PDE family members and against a panel of enzymes, receptors, transporters, and ion channels.
It is disclosed in WO 2009/075784 (U.S. Pub. No. 2010/0273754). This compound has been found to be a potent and selective phosphodiesterase 1 (PDE 1) inhibitor useful for the treatment or prophylaxis of disorders characterized by low levels of cAMP and/or cGMP in cells expressing PDE1, and/or reduced dopamine Dl receptor signaling activity (e.g., Parkinson’s disease, Tourette’s Syndrome, Autism, fragile X syndrome, ADHD, restless leg syndrome, depression, cognitive impairment of schizophrenia, narcolepsy); and/or any disease or condition that may be ameliorated by the enhancement of progesterone signaling. This list of disorders is exemplary and not intended to be exhaustive.

PATENT
WO 2013192556
http://www.google.com/patents/WO2013192556A2?cl=en
The method of making the Compound (ea^^a^-S^a ^^^a-hexahydro-S- methyl-3-(phenylamino)-2-((4-(6-fluoropyridin-2-yl)phenyl)methyl)- cyclopent[4,5]imidazo[l,2-fl]pyrazolo[4,3-e]pyrimidin-4(2H)-one is generally described in WO 2009/075784, the contents of which are incorporated by reference in their entirety. This compound can also be prepared as summarized or similarly summarized in the following
CMU PCU PHU PPU (SM2)
In particular, (6aR,9aS)-3-chloro-2-(4-(6-fluoropyridin-2-yl)benzyl)-5-methyl- 5,6a,7,8,9,9a-hexahydrocyclopenta[4,5]imidazo[l,2-a]pyrazolo[4,3-e]pyrimidin-4(2H)- one may be prepared as described or similarly described below.
PATENT
http://www.google.com/patents/WO2009075784A1?cl=en
EXAMPLE 14
(6aJ?,9aS)-5,6a,7,8,9,9a-hexahydro-5-methyl-3-(phenylamino)-2-((4-(6- fluoropyridin-2-yl)phenyl)methyl)-cyclopent[4,5]iinidazo[l,2-fl]pyrazolo[4,3- e]pyrimidin-4(2//)-one
This compound may be made using similar method as in example 13 wherein 2-(4-(bromomethyl)phenyl)-6-fluoropyridine may be used instead of 2-(4- (dibromomethyl)phenyl)-5-fluoropyridine.
PATENT
WO 2014205354
https://www.google.co.in/patents/WO2014205354A2?cl=en

EXAMPLES
The method of making the Compound (ea^^a^-S^a ^^^a-hexahydro-S-methyl-3-(phenylamino)-2-((4-(6-fluoropyridin-2-yl)phenyl)methyl)-cyclopent[4,5]imidazo[l,2-fl]pyrazolo[4,3-e]pyrimidin-4(2H)-one is generally described in WO 2009/075784, the contents of which are incorporated by reference in their entirety. This compound can also be prepared as summarized or similarly summarized in the following

CMU PCU PHU PPU (SM2)
![]()

In particular, (6aR,9aS)-3-chloro-2-(4-(6-fluoropyridin-2-yl)benzyl)-5-methyl-5,6a,7,8,9,9a-hexahydrocyclopenta[4,5]imidazo[l,2-a]pyrazolo[4,3-e]pyrimidin-4(2H)-one (Int-5) may be prepared as described or similarly described below. The free base crystals and the mono-phosphate salt crystals of the invention may be prepared by using the methods described or similarly described in Examples 1-14 below.
Preparation of (6aR,9aS)-3-chloro-2-(4-(6-fluoropyridin-2-yl)benzyl)-5-methyl-5,6a,7,8,9,9a-hexahydrocyclopenta[4,5]imidazo[l,2-a]pyrazolo[4,3-e]pyrimidin-4(2H)-one
(4-(6-fluoropyridin-2-yl)phenyl)methanol

The mixture of Na2C03 (121 g), water (500 mL), THF (650 mL), PdCl2(PPh3)2 (997 mg), 2-bromo-6-fluoropyridine (100 g) and 4-(hydroxymethyl)phenylboronic acid (90.7 g) is stirred at 65°C for 4 h under the nitrogen atmosphere. After cooling to room temperature, THF (200 mL) is added. The organic layer is separated and washed with 5% NaCl solution twice. The organic layer is concentrated to 400 mL. After the addition of toluene (100 mL), heptane (500 mL) is added at 55°C. The mixture is cooled to room temperature. The crystals are isolated by filtration, washed with the mixture of toluene (100 mL) and heptane (100 mL) and dried to give (4-(6-fluoropyridin-2-yl)phenyl)methanol (103 g). ]H NMR (500 MHz, CDC13) δ 1.71-1.78 (m, 1H), 4.74-4.79 (m, 2H), 6.84-6.88 (m, 1H), 7.44-7.50 (m, 2H), 7.61-7.65 (m, 1H), 7.80-7.88 (m, 1H), 7.98-8.04 (m, 2H).
2-(4-(chloromethyl)phenyl)-6-fluoropyridine

The solution of thionylchloride (43.1 mL) in AcOEt (200 mL) is added to the mixture of (4-(6-fluoropyridin-2-yl)phenyl)methanol (100 g), DMF (10 mL) and AcOEt (600 mL) at room temperature. The mixture is stirred at room temperature for 1 h. After cooling to 10°C, 15% Na2C03 solution is added. The organic layer is separated and washed with water (500 mL) and 5% NaCl solution (500 mL) twice. The organic layer is concentrated to 500 mL. After the addition of EtOH (500 mL), the mixture is concentrated to 500 mL. After addition of EtOH (500 mL), the mixture is concentrated to 500 mL. After the addition of EtOH (500 mL), the mixture is concentrated to 500 mL. After addition of EtOH (200 mL), water (700 mL) is added at 40°C. The mixture is stirred at room temperature. The crystals are isolated by filtration and dried to give 2-(4-(chloromethyl)phenyl)-6-fluoropyridine (89.5 g). ]H NMR (500 MHz, CDC13) δ 4.64 (s, 2H), 6.86-6.90 (m, 1H), 7.47-7.52 (m, 2H), 7.60-7.65 (m, 1H), 7.82-7.88 (m, 1H), 7.98-8.03 (m, 2H).
6-chloro-l-(4-methoxybenzyl)-3-methylpyrimidine-2,4(lH,3H)-dione

The mixture of 6-chloro-3-methyluracil (100 g), p-methoxybenzylchloride (107 g), K2CO3 (86.1 g) and DMAc (600 mL) is stirred at 75°C for 4 h. Water (400 mL) is added at 45°C and the mixture is cooled to room temperature. Water (800 mL) is added and the mixture is stirred at room temperature. The crystals are isolated by filtration, washed with the mixture of DMAc and water (1:2, 200mL) and dried to give 6-chloro-l-(4-methoxybenzyl)-3-methylpyrimidine-2,4(lH,3H)-dione (167 g). ]H NMR (500 MHz, CDC13) δ 3.35 (s, 3H), 3.80 (s, 3H), 5.21 (s, 2H), 5.93 (s, 1H), 6.85-6.89 (m, 2H), 7.26-7.32 (m, 2H).
izinyl-l-(4-methoxybenzyl)-3-methylpyrimidine-2,4(lH,3H)-dione

The mixture of 6-chloro-l-(4-methoxybenzyl)-3-methylpyrimidine-2,4(lH,3H)-dione (165 g), IPA (990 mL), water (124 mL) and hydrazine hydrate (62.9 mL) is stirred at room temperature for 1 h. The mixture is warmed to 60°C and stirred at the same temperature for 4 h. Isopropyl acetate (1485 mL) is added at 45°C and the mixture is stirred at the same temperature for 0.5 h. The mixture is cooled at 10°C and stirred for lh. The crystals are isolated by filtration, washed with the mixture of IPA and isopropyl acetate (1:2, 330 mL) and dried to give 6-hydrazinyl-l-(4-methoxybenzyl)-3-methylpyrimidine-2,4(lH,3H)-dione (153 g). ]H NMR (500 MHz, DMSO-i¾) δ 3.12 (s, 3H), 3.71 (s, 3H), 4.36 (s, 2H), 5.01 (s, 2H), 5.14 (s, 1H), 6.87-6.89 (m, 2H), 7.12-7.17 (m, 2H), 8.04 (s, 1H).
7-(4-methoxybenzyl)-5-methyl-2H-pyrazolo[3,4-d]pyrimidine-4,6(5H,7H)-dione

To the mixture of DMF (725 mL) and 6-hydrazinyl-l-(4-methoxybenzyl)-3-methylpyrimidine-2,4(lH,3H)-dione (145 g) is added POCI3 (58.5 mL) at 5°C. The mixture is stirred at room temperature for 1 h. Water (725 mL) is added at 50°C and the mixture is stirred at room temperature for 1 h. The crystals are isolated by filtration, washed with the mixture of DMF and water (1:1, 290 mL) and dried to give 7-(4-methoxybenzyl)-5-methyl-
2H-pyrazolo[3,4-d]pyrimidine-4,6(5H,7H)-dione (145 g). ]H NMR (500 MHz, DMSO-i¾) δ 3.23 (s, 3H), 3.71 (s, 3H), 5.05 (s, 2H), 6.82-6.90 (m, 2H), 7.28-7.36 (m, 2H), 8.48 (s, IH), 13.51 (br, IH).
2-(4-(6-fluoropyridin-2-yl)benzyl)-7-(4-methoxybenzyl)-5-methyl-2H-pyrazolo[3,4-d]pyrimidine-4,6(5H,7H)-dione

The mixture of 2-(4-(chloromethyl)phenyl)-6-fluoropyridine (100 g), 7-(4-methoxybenzyl)-5-methyl-2H-pyrazolo[3,4-d]pyrimidine-4,6(5H,7H)-dione (129 g), K2CO3(62.3 g) and DMAc (1500 mL) is stirred at 45°C for 5 h. Water (1500 mL) is added at 40°C and the mixture is stirred at room temperature for 1 h. The crystals are isolated by filtration, washed with the mixture of DMAc and water (1:1, 500 mL) and dried to give 2-(4-(6-fluoropyridin-2-yl)benzyl)-7-(4-methoxybenzyl)-5-methyl-2H-pyrazolo[3,4-d]pyrimidine-4,6(5H,7H)-dione (207 g). ]H NMR (500 MHz, DMSO- ) δ 3.21 (s, 3H), 3.66 (s, 3H), 4.98 (s, 2H), 5.45 (s, 2H), 6.77-6.82 (m, 2H), 7.13-7.16 (m, IH), 7.25-7.30 (m, 2H), 7.41-7.44 (m, 2H), 7.92-7.96 (m, IH), 8.04-8.11 (m, 3H), 8.68 (s, IH).
2-(4-(6-fluoropyridin-2-yl)benzyl)-5-methyl-2H-pyrazolo[3,4-d]pyrimidine-4,6(5H,7H)-dione

The mixture of 2-(4-(6-fluoropyridin-2-yl)benzyl)-7-(4-methoxybenzyl)-5-methyl-2H-pyrazolo[3,4-d]pyrimidine-4,6(5H,7H)-dione (105 g), CF3COOH (300 mL) and
CF3SO3H (100 g) is stirred at room temperature for 10 h. Acetonitrile (1000 mL) is added. The mixture is added to the mixture of 25% N¾ (1000 mL) and acetonitrile (500 mL) at 10°C. The mixture is stirred at room temperature for 1 h. The crystals are isolated by filtration, washed with the mixture of acetonitirile and water (1:1, 500 mL) and dried to give the crude product. The mixture of the crude product and AcOEt (1200 mL) is stirred at room temperature for 1 h. The crystals are isolated by filtration, washed with AcOEt (250 mL) and dried to give 2-(4-(6-fluoropyridin-2-yl)benzyl)-5-methyl-2H-pyrazolo[3,4-d]pyrimidine-4,6(5H,7H)-dione (75.3 g). ]H NMR (500 MHz, DMSO-rf6) δ 3.16 (s, 3H), 3.50-4.00 (br, 1H), 5.40 (s, 2H), 7.13-7.16 (m, 1H), 7.41-7.44 (m, 2H), 7.91-7.94 (m, 1H), 8.04-8.10 (m, 3H), 8.60 (s, 1H).
2-(4-(6-fluoropyridin-2-yl)benzyl)-6-(((lR,2R)-2-hydroxycyclopentyl)amino)-5-methyl-2H-pyrazolo[3,4-d]pyrimidin-4(5H)-one

The mixture of BOP reagent (126 g), 2-(4-(6-fluoropyridin-2-yl)benzyl)-5-methyl-2H-pyrazolo[3,4-d]pyrimidine-4,6(5H,7H)-dione (80 g), DBU (136 mL) and THF (1120 mL) is stirred at room temperature for 1 h. (lR,2R)-2-Aminocyclopentanol hydrochloride (37.6 g) and THF (80 mL) are added and the mixture is stirred at room temperature for 5 h. After the addition of 5% NaCl (400 mL) and AcOEt (800 mL), the organic layer is separated. The organic layer is washed with 10% NaCl (400 mL), 1M HC1 15% NaCl (400 mL), 5% NaCl (400 mL), 5% NaHC03 (400 mL) and 5%NaCl (400 mL) successively. After treatment with active charcoal, the organic layer is concentrated to 400 mL. After the addition of acetonitrile (800 mL), the mixture is concentrated to 400 mL. After the addition of acetonitrile (800 mL), seed crystals are added at 40°C. The mixture is concentrated to 400 mL. Water (800 mL) is added at room temperature and the mixture is stirred for 2 h. The crystals are isolated by filtration, washed with the mixture of acetonitrile and water (1:2, 400 mL) and dried to give 2-(4-(6-fluoropyridin-2-yl)benzyl)-6-(((lR,2R)-2-
hydroxycyclopentyl)amino)-5-methyl-2H-pyrazolo[3,4-d]pyrimidin-4(5H)-one (81.7 g). ]H NMR (500 MHz, CDC13) δ 1.47-1.59 (m, 1H), 1.68-1.93 (m, 3H), 2.02-2.12 (m, 1H), 2.24-2.34 (m, 1H), 3.42 (s, 3H), 3.98-4.12 (m, 2H), 4.68-4.70 (m, 1H), 5.37 (s, 2H), 6.86-6.90 (m, 1H), 7.36-7.42 (m, 2H), 7.58-7.63 (m, 1H), 7.81-7.88 (m, 1H), 7.89 (s, 1H), 7.97-8.01 (m, 2H).
(6aR,9aS)-2-(4-(6-fluoropyridin-2-yl)benzyl)-5-methyl-5,6a,7,8,9,9a-hexahydrocyclopenta[4,5]imidazo[l,2-a]pyrazolo[4,3-e]pyrimidin-4(2H)-one

The mixture of 2-(4-(6-fluoropyridin-2-yl)benzyl)-6-(((lR,2R)-2-hydroxycyclopentyl)amino)-5-methyl-2H-pyrazolo[3,4-d]pyrimidin-4(5H)-one (80 g), p-toluenesulfonylchloride (38.6 g), Et3N (28.2 mL), N,N-dimethylaminopyridine (24.7 g) and THF (800 mL) is stirred at 50°C for 10 h. To the mixture is added 8M NaOH (11.5 mL) at room temperature and the mixture is stirred for 2 h. After the addition of 5% NaCl (400 mL) and AcOEt (800 mL), the organic layer is separated. The organic layer is washed with 5 NaCl (400 mL) twice. The organic layer is concentrated to 240 mL. After the addition of MeOH (800 mL), the mixture is concentrated to 240 mL. After the addition of MeOH (800 mL), the mixture is concentrated to 240 mL. After the addition of MeOH (160 mL), the mixture is stirred at room temperature for 1 h and at 0°C for 1 h. The crystals are isolated by filtration, washed with cold MeOH (160 mL) and dried to give (6aR,9aS)-2-(4-(6-fluoropyridin-2-yl)benzyl)-5-methyl-5,6a,7,8,9,9a-hexahydrocyclopenta[4,5]imidazo[l,2-a]pyrazolo[4,3-e]pyrimidin-4(2H)-one (55.7 g). ]H NMR (500 MHz, CDC13) δ 1.39-1.54 (m, 1H), 1.58-1.81 (m, 3H), 1.81-1.92 (m, 1H), 2.12-2.22 (m, 1H), 3.28 (s, 3H), 4.61-4.70 (m, 2H), 5.20 (s, 2H), 6.79-6.85 (m, 1H), 7.25-7.32 (m, 2H), 7.53-7.58 (m, 1H), 7.68 (s, 1H), 7.75-7.83 (m, 1H), 7.92-7.98 (m, 2H).
(6aR,9aS)-3-chloro-2-(4-(6-fluoropyridin-2-yl)benzyl)-5-methyl-5,6a,7,8,9,9a-
hexahydrocyclopenta[4,5]imi ]pyrimidin-4(2H)-one

The mixture of (6aR,9aS)-2-(4-(6-fluoropyridin-2-yl)benzyl)-5-methyl-5,6a,7,8,9,9a-hexahydrocyclopenta[4,5]imidazo[l,2-a]pyrazolo[4,3-e]pyrimidin-4(2H)-one (50 g) and toluene (1000 mL) is concentrated to 750 mL under the nitrogen atmosphere. Toluene (250 mL) and NCS (24 g) is added. To the mixture is added LiHMDS (1M THF solution, 204 mL) at 0°C and the mixture is stirred for 0.5 h. To the mixture is added 20% NH4C1 (50 mL) at 5°C. The mixture is concentrated to 250 mL. After the addition of EtOH (250 mL), the mixture is concentrated to 150 mL. After the addition of EtOH (250 mL), the mixture is concentrated to 200 mL. After the addition of EtOH (200 mL), the mixture is warmed to 50°C. Water (300 mL) is added and the mixture is stirred at 50°C for 0.5 h. After stirring at room temperature for 1 h, the crystals are isolated by filtration, washed with the mixture of EtOH and water (1:1, 150 mL) and dried to give (6aR,9aS)-3-chloro-2-(4-(6-fluoropyridin-2-yl)benzyl)-5-methyl-5,6a,7,8,9,9a-hexahydrocyclopenta[4,5]imidazo[l,2-a]pyrazolo[4,3-e]pyrimidin-4(2H)-one (51.1 g). ]H NMR (500 MHz, CDC13) δ 1.46-1.61 (m, 1H), 1.67-1.90 (m, 3H), 1.92-2.00 (m, 1H), 2.19-2.27 (m, 1H), 3.37 (s, 3H), 4.66-4.77 (m, 2H), 5.34 (s, 2H), 6.87-6.93 (m, 1H), 7.35-7.41 (m, 2H), 7.59-7.65 (m, 1H), 7.82-7.91 (m, 1H), 7.97-8.05 (m, 2H).
EXAMPLE 1
Crystals of (6a/f,9a5)-5,6a,7,8,9,9a-hexahydro-5-methyl-3-(phenylamino)-2-((4-(6-fluoropyridin-2-yl)phenyl)methyl)-cyclopent[4,5]imidazo[l,2-a]pyrazolo[4,3-e]pyrimidin-4(2H)-one free base mono-ethanol solvate

The mixture of (6a/?,9a5′)-3-chloro-2-(4-(6-fluoropyridin-2-yl)benzyl)-5-methyl-5,6a,7,8,9,9a-hexahydrocyclopenta[4,5]imidazo[l,2-fl]pyrazolo[4,3-e]pyrimidin-4(2H)-one (2.5 g), K2C03 (1.53 g), Pd(OAc)2 (12.5 mg), Xantphos (32 mg), aniline (0.76 mL), and xylene (12.5 mL) is stirred at 125°C for 7 h under nitrogen atmosphere. After addition of water (12.5 mL), the organic layer is separated. The organic layer is washed with water (12.5 mL) twice. The organic layer is extracted with the mixture of DMAc (6.25 mL) and 0.5N HCl (12.5 mL). The organic layer is extracted with the mixture of DMAc (3.2 mL) and 0.5N HCl (6.25 mL). After addition of DMAc (6.25 mL), xylene (12.5 mL) and 25 wt % aqueous NH3 solution to the combined aqueous layer, the organic layer is separated. The aqueous layer is extracted with xylene (6.25 mL). The combined organic layer is washed with water (12.5 mL), 2.5 wt % aqueous 1 ,2-cyclohexanediamine solution (12.5 mL) twice and water (12.5 mL) successively. After treatment with active charcoal, the organic layer is concentrated. After addition of EtOH (12.5 mL), the mixture is concentrated. After addition of EtOH (12.5 mL), the mixture is concentrated. After addition of EtOH (12.5 mL), n-heptane (25 mL) is added at 70°C. The mixture is cooled to 5°C and stirred at same temperature. The crystals are isolated by filtration and dried to give (ea^^a^-S^a ^^^a-hexahydro-5-methyl-3-(phenylamino)-2-((4-(6-fluoropyridin-2-yl)phenyl)methyl)-cyclopent[4,5]imidazo[l,2-fl]pyrazolo[4,3-e]pyrimidin-4(2H)-one free base mono-ethanol solvate (2.56 g) as crystals.
]H NMR (500 MHz, DMSO-d6) δ 0.98-1.13 (m, 3H), 1.34-1.52 (m, 1H), 1.54-1.83 (m, 4H), 2.03-2.17 (m, 1H), 3.11 (s, 3H), 3.39-3.54 (m, 2H), 4.29-4.43 (m, 1H), 4.51-4.60 (m, 1H), 4.60-4.70 (m, 1H), 5.15-5.35 (m, 2H), 6.71-6.88 (m, 3H), 7.05-7.29 (m, 5H), 7.81-7.93 (m, 1H), 7.94-8.11 (m, 3H), 8.67 (s, 1H).
EXAMPLE 4
Crystals of (6a/f,9a5)-5,6a,7,8,9,9a-hexahydro-5-methyl-3-(phenylamino)-2-((4-(6-fluoropyridin-2-yl)phenyl)methyl)-cyclopent[4,5]imidazo[l,2-a]pyrazolo[4,3-e]pyrimidin-4(2H)-one free

Crystals of (6a«,9a5′)-5,6a,7,8,9,9a-hexahydro-5-methyl-3-(phenylamino)-2-((4-(6-fluoropyridin-2-yl)phenyl)methyl)-cyclopent[4,5]imidazo[l,2-fl]pyrazolo[4,3-e]pyrimidin-4(2H)-one free base mono-n-propanol solvate (2.0 g) is dissolved with ethanol (10 mL) at 70°C. Isopropyl ether (20 mL) is added and the mixture is cooled to 45°C. Isopropyl ether (10 mL) is added and the mixture is stirred at 40°C. The mixture is cooled to 5°C and stirred at same temperature. The crystals are isolated by filtration and dried to give (ea/^^a^)-5,6a,7,8,9,9a-hexahydro-5-methyl-3-(phenylamino)-2-((4-(6-fluoropyridin-2-yl)phenyl)methyl)-cyclopent[4,5]imidazo[l,2-fl]pyrazolo[4,3-e]pyrimidin-4(2H)-one free base non-solvate (1.7 g) as crystals.
[0082] ]H NMR (500 MHz, DMSO-d6) δ 1.32-1.51 (m, 1H), 1.53-1.83 (m, 4H), 1.97-2.20 (m, 1H), 3.11 (s, 3H), 4.49-4.60 (m, 1H), 4.60-4.69 (m, 1H), 5.13-5.37 (m, 2H), 6.70-6.90 (m, 3H), 7.04-7.31 (m, 5H), 7.82-7.93 (m, 1H), 7.93-8.12 (m, 3H), 8.67 (s, 1H).
EXAMPLE 5
Crystals of (6a/f,9a5)-5,6a,7,8,9,9a-hexahydro-5-methyl-3-(phenylamino)-2-((4-(6-fluoropyridin-2-yl)phenyl)methyl)-cyclopent[4,5]imidazo[l,2-a]pyrazolo[4,3-e]pyrimidin-4(2H)-one free base non-solvate

The mixture of (6a/?,9a5′)-3-chloro-2-(4-(6-fluoropyridin-2-yl)benzyl)-5-methyl-5,6a,7,8,9,9a-hexahydrocyclopenta[4,5]imidazo[l,2-fl]pyrazolo[4,3-e]pyrimidin-4(2H)-one (25 g), K2C03 (15.4 g), Pd(OAc)2 (125 mg), Xantphos (321 mg), aniline (7.6 mL), DMAc (6.25 mL) and xylene (125 mL) is stirred at 125°C for 6.5 h under nitrogen atmosphere. After addition of water (125 mL) and DMAc (50 mL), the organic layer is separated. The organic layer is washed with the mixture of DMAc (50 mL) and water (125 mL) twice. The organic layer is extracted with the mixture of DMAc (50 mL) and 0.5N HCl (125 mL). The organic layer is extracted with the mixture of DMAc (50 mL) and 0.5N HCl (62.5 mL). After addition of DMAc (50 mL), xylene (125 mL) and 25 wt % aqueous NH3 solution (25 mL) to the combined aqueous layer, the organic layer is separated. The aqueous layer is extracted with xylene (62.5 mL). The combined organic layer is washed with the mixture of DMAc (50 mL) and water (125 mL), the mixture of DMAc (50 mL) and 2.5 wt % aqueous 1,2-cyclohexanediamine solution (125 mL) twice and the mixture of DMAc (50 mL) and water (125 mL) successively. After treatment with active charcoal (1.25 g), the organic layer is concentrated to 75 mL. After addition of EtOH (125 mL), the mixture is concentrated to 75 mL. After addition of EtOH (125 mL), the mixture is concentrated to 75 mL. After addition of EtOH (125 mL), n-heptane (250 mL) is added at 70°C. After addition of seed crystals of (6a/?,9a5′)-5,6a,7,8,9,9a-hexahydro-5-methyl-3-(phenylamino)-2-((4-(6-fluoropyridin-2-yl)phenyl)methyl)-cyclopent[4,5]imidazo[l,2-fl]pyrazolo[4,3-e]pyrimidin-4(2H)-one non-solvate, the mixture is cooled to room temperature and stirred at room temperature. The crystals are isolated by filtration and dried to give (ea^^a^-S^a ^^^a-hexahydro-S-methyl-3-(phenylamino)-2-((4-(6-fluoropyridin-2-yl)phenyl)methyl)-cyclopent[4,5]imidazo-[l,2-fl]pyrazolo[4,3-e]pyrimidin-4(2H)-one free base non-solvate (23.8 g) as crystals.
EXAMPLE 8
(6a/f,9a5)-5,6a,7,8,9,9a-hexahydro-5-methyl-3-(phenylamino)-2-((4-(6-fluoropyridin-2-yl)phenyl)methyl)-cyclopent[4,5]imidazo[l,2-a]pyrazolo[4,3-e]pyrimidin-4(2H)-one mono-phosphate salt

[0094] Crystals of (6a«,9a5′)-5,6a,7,8,9,9a-hexahydro-5-methyl-3-(phenylamino)-2-((4-(6-fluoropyridin-2-yl)phenyl)methyl)-cyclopent[4,5]imidazo[l,2-fl]pyrazolo[4,3-e]pyrimidin-4(2H)-one free base non-solvate (20 g) are dissolved in acetonitrile (60 mL) at 50°C. After addition of the active charcoal (1 g), the mixture is stirred at same temperature for 0.5 h. The active charcoal is removed by filtration and washed with acetonitrile (40 mL). The filtrate and the washing are combined and warmed to 50°C. A solution of 85 wt. % phosphoric acid (2.64 mL) in acetonitrile (100 mL) is added. After addition of water (20 mL), the mixture is stirred at 50°C for lh. The crystals are isolated by filtration, washed with acetonitrile (60 mL x 3) and dried to give (6a/?,9a5′)-5,6a,7,8,9,9a-hexahydro-5-methyl-3-(phenylamino)-2-((4-(6-fluoropyridin-2-yl)phenyl)methyl)-cyclopent[4,5]imidazo[l,2-fl]pyrazolo[4,3-e]pyrimidin-4(2H)-one mono-phosphate salt (20.5 g).
EXAMPLE 9
(6a/f,9a5)-5,6a,7,8,9,9a-hexahydro-5-methyl-3-(phenylamino)-2-((4-(6-fluoropyridin-2-yl)phenyl)methyl)-cyclopent[4,5]imidazo[l,2-a]pyrazolo[4,3-e]pyrimidin-4(2H)-one mono-phosphate salt

[0095] Crystals of (6a«,9a5′)-5,6a,7,8,9,9a-hexahydro-5-methyl-3-(phenylamino)-2-((4-(6-fluoropyridin-2-yl)phenyl)methyl)-cyclopent[4,5]imidazo[l,2-fl]pyrazolo[4,3-e]pyrimidin-4(2H)-one free base mono-ethanol solvate (4 g) are dissolved in acetonitrile (12 mL) at 50°C. After addition of active charcoal (0.2 g), the mixture is stirred at same temperature for 0.5 h. Active charcoal is removed by filtration and washed with acetonitrile (8 mL). The filtrate and the washing are combined and warmed to 50°C. A solution of 85 wt. % phosphoric acid (0.528 mL) in acetonitrile (20 mL) is added. After addition of water (4 mL), the mixture is stirred at 50°C for lh. The crystals are isolated by filtration, washed with acetonitrile (12 mL x 3) and dried to give (6a/?,9a5′)-5,6a,7,8,9,9a-hexahydro-5-methyl-3-(phenylamino)-2-((4-(6-fluoropyridin-2-yl)phenyl)methyl)-cyclopent[4,5]imidazo[l,2-fl]pyrazolo[4,3-e]pyrimidin-4(2H)-one mono-phosphate salt (4.01 g).
EXAMPLE 10
(6a/f,9a5)-5,6a,7,8,9,9a-hexahydro-5-methyl-3-(phenylamino)-2-((4-(6-fluoropyridin-2-yl)phenyl)methyl)-cyclopent[4,5]imidazo[l,2-a]pyrazolo[4,3-e]pyrimidin-4(2H)-one mono-phosphate salt

Crystals of (6a«,9a5′)-5,6a,7,8,9,9a-Hexahydro-5-methyl-3-(phenylamino)-2-((4-(6-fluoropyridin-2-yl)phenyl)methyl)-cyclopent[4,5]imidazo[l,2-fl]pyrazolo[4,3-e]pyrimidin-4(2H)-one free base non-solvate (20 g) are dissolved in acetone (60 mL) at 32°C. After addition of active charcoal (1 g), the mixture is stirred at same temperature for 0.5 h. Active charcoal is removed by filtration and washed with acetone (40 mL). The filtrate and the washing are combined and warmed to 39°C. A solution of 85 wt. % phosphoric acid (2.64 mL) in acetone (100 mL) is added. After addition of water (20 mL), the mixture is stirred at 40°C for lh. The crystals are isolated by filtration, washed with acetone (60 mL x 3) and dried to give (6a/?,9a5′)-5,6a,7,8,9,9a-hexahydro-5-methyl-3-(phenylamino)-2-((4-(6-fluoropyridin-2-yl)phenyl)methyl)-cyclopent[4,5]imidazo[l,2-fl]pyrazolo[4,3-e]pyrimidin-4(2H)-one mono-phosphate salt (22.86 g).
EXAMPLE 11
(6a/f,9a5)-5,6a,7,8,9,9a-hexahydro-5-methyl-3-(phenylamino)-2-((4-(6-fluoropyridin-2-yl)phenyl)methyl)-cyclopent[4,5]imidazo[l,2-a]pyrazolo[4,3-e]pyrimidin-4(2H)-one mono-phosphate salt

Crystals of (6a«,9a5′)-5,6a,7,8,9,9a-hexahydro-5-methyl-3-(phenylamino)-2-((4-(6-fluoropyridin-2-yl)phenyl)methyl)-cyclopent[4,5]imidazo[l,2-fl]pyrazolo[4,3-e]pyrimidin-4(2H)-one free base mono-ethanol solvate (20 g) are dissolved in acetone (60 mL) at 38°C. After addition of active charcoal (1 g), the mixture is stirred at same temperature for 0.5 h. Active charcoal is removed by filtration and washed with acetone (40 mL). The filtrate and the washing are combined and warmed to 38°C. A solution of 85 wt. % phosphoric acid (2.64 mL) in acetone (100 mL) is added. After addition of water (20 mL), the mixture is stirred at 40°C for lh. The crystals are isolated by filtration, washed with acetone (60 mL x 3) and dried to give (6a/?,9a5′)-5,6a,7,8,9,9a-hexahydro-5-methyl-3-(phenylamino)-2-((4-(6-fluoropyridin-2-yl)phenyl)methyl)-cyclopent[4,5]imidazo[l,2-fl]pyrazolo[4,3-e]pyrimidin-4(2H)-one mono-phosphate salt (23.2 g).
PAPER
A diverse set of 3-aminopyrazolo[3,4-d]pyrimidinones was designed and synthesized. The structure–activity relationships of these polycyclic compounds as phosphodiesterase 1 (PDE1) inhibitors were studied along with their physicochemical and pharmacokinetic properties. Systematic optimizations of this novel scaffold culminated in the identification of a clinical candidate, (6aR,9aS)-2-(4-(6-fluoropyridin-2-yl)benzyl)-5-methyl-3-(phenylamino)-5,6a,7,8,9,9a-hexahydrocyclopenta[4,5]imidazo[1,2-a]pyrazolo[4,3-e]pyrimidin-4-(2H)-one phosphate (ITI-214), which exhibited picomolar inhibitory potency for PDE1, demonstrated excellent selectivity against all other PDE families and showed good efficacy in vivo. Currently, this investigational new drug is in Phase I clinical development and being considered for the treatment of several indications including cognitive deficits associated with schizophrenia and Alzheimer’s disease, movement disorders, attention deficit and hyperactivity disorders, and other central nervous system (CNS) and non-CNS disorders
The synthetic methods disclosed in WO 2009/075784 and WO 2013/192556 are particularly applicable, as they include the methods to prepare the compound of Formula I-B. Those skilled in the art will readily see how those methods are applicable to the synthesis of the compounds of the present invention.

Formula I-B
For example, Compounds of the Invention wherein any one or more of R1 through R8 are D, can be prepared from the corresponding aminocyclopentanol, according to the method described in WO 2009/075784 or WO 2013/192556. For example, by reacting said aminocyclopentanol, optionally as its acid salt, with Intermediate A in the presence of a coupling agent, e.g., benzotriazol-1-yloxy)tris(dimethylamino)phosphonium hexafluorophosphate (BOP reagent), and a base, e.g., l,8-diazabicyclo[5.4.0]undec-7-ene (DBU), in a solvent such as tetrahydrofuran (THF). The intermediate alcohol is then cyclized by treatment with toluenesulfonyl chloride (TsCl) in the presence of one or more bases, such as dimethylaminopyridine (DMAP) and triethylamine (TEA) in a solvent, such as THF. The reaction is summarized in the following scheme:

The required aminocyclopentanols can be prepared by methods known to those skilled in the art. For example, the aminocyclopentanol wherein R1 is D can be prepared via a reductive amination procedure that uses a reducing agent such as sodium triacetoxyborodeuteride or sodium borodeuteride as the reducing agent. For example, an optionally protected (R)-2-hydroxycyclopentanone can be reacted with 4-methoxybenzylamine in the presence of sodium triacetoxyborodeuteride to yield the desired deuterated secondary amine, wherein P is the protecting group. Reaction of the resulting amine with a strong acid such as trifluoromethanesulfonic acid (TMFSA) will result in removal of the 4-methoxybenzyl group and the protecting group to yield the desired aminocyclopentanol. Those skilled in the art will know how to choose a suitable protecting group for the secondary alcohol such that deprotection can take place during the acid treatment step (e.g., a tert-butyldimethylsilyl group or a tert-butoxycarbonyl group). Alternatively, those skilled in the art could choose a protecting group that would survive this step. If desired, the protected intermediate can be purified by chiral HPLC in order to enhance the optical purity of the final

As another example, Compounds of the Invention wherein any one or more of R9 to R15 or R21 to R22 are D can be prepared from the corresponding benzyl halide, according to the method described in WO 2009/075784 or WO 2013/192556. For example, by reacting said benzyl halide with the Intermediate B in the presence of suitable base, such as cesium carbonate or potassium carbonate, in a suitable solvent, such as dimethylformamide or dimethylacetamide. The corresponding benzyl halide can be prepared by methods well known to those skilled in the art. The reaction is summarized in the following scheme:

As another example, compounds of the invention wherein any one or more of R16 to R20 are D can be prepared from the corresponding phenyl
isothiocyanate, according to the method described in WO 2009/075784 or WO
2013/192556. For example, by reacting said phenyl isothiocyanate with Intermediate C in a suitable solvent, such as dimethylformamide. The corresponding phenyl isothiocyanate can be prepared by methods well known to those skilled in the art. The reaction is summarized in the following scheme:

Alternatively, compounds of the invention wherein any one or more of R16 to R20 are D can be prepared from the corresponding aniline, according to the method described in WO 2009/075784 or WO 2013/192556. For example, by reacting said aniline with Intermediate D and a strong base, such as lithium
hexamethyldisilylazide (LiHMDS), in a suitable solvent, such as THF at elevated temperature. Such a reaction can also be achieved by catalytic amination using a catalyst, such as tris(dibenzylideneacetone)dipalladium (Pd2(dba)3), and a ligand, such as Xantphos. The corresponding aniline can be prepared by methods well known to those skil

EXAMPLE 1. (6aR,9a5)-5-Methyl-3-(2,3,4,5,6-pentadeuterophenylamino)-2-(4-(6-fluoropyridin-2-yl)-benzyl)-5,6fl,7,8,9,9fl-hexahydrocyclopent[4,5]imidazo[l,2-a]pyrazolo[4,3-e]pyrimidin-4(2H)-one

To a solution of (6a/?,9a5′)-5,6a,7,8,9,9a-hexahydro-3-chloro-5-methyl-2-(4-(6-fluoropyridin-2-yl)-benzyl)-cyclopent[4,5]irnidazo[l,2-fl]pyrazolo[4,3-e]pyrimidin-4(2H)-one (200 mg, 0.444 mmol) and 2,3,4,5,6-pentadeuteroaniline (162 μΐ,, 1.8 mmol) in anhydrous 2-methyltetrahydrofuran (3 mL) is added LiHMDS (1.0 M in THF, 0.89 mL) dropwise at room temperature under argon atmosphere. The reaction mixture is gradually heated to 75 °C over a period of 90 min, and then heated at 75 °C for an hour. The mixture is cooled with an ice bath and then quenched by adding 0.2 mL of water. After solvent evaporation, the residue is dissolved in DMF and then filter with a 0.45 m microfilter. The collected filtrated is purified with a semi-preparative HPLC system using a gradient of 0 – 70% acetonitrile in water containing 0.1% formic acid over 16 min to give (6a/?,9a5′)-5-methyl-3-(2,3,4,5,6-pentadeuterophenylamino)-2-(4-(6-fluoropyridin-2-yl)-benzyl)-5,6fl,7,8,9,9fl-hexahydrocyclopent[4,5]imidazo[l,2-a]pyrazolo[4,3-e]pyrimidin-4(2H)-one as a formate salt, which is dissolved in ethyl acetate, basified with 12.5 mL of 5% sodium carbonate, and then extracted with ethyl acetate three times. The combined organic phase is evaporated to dryness. The residue is dissolved in 4.5 mL of THF and then filter through a 0.45 m microfilter. The filtrate is evaporated to dryness and further dried under vacuum to give (6a/?,9a5′)-5-methyl-3-(2,3,4,5,6-pentadeuterophenylamino)-2-(4-(6-fluoropyridin-2-yl)-benzyl)-5,6fl,7,8,9,9fl-hexahydrocyclopent[4,5]imidazo[l,2-a]pyrazolo[4,3-e]pyrimidin-4(2H)-one as a white solid (185.8 mg, 81.6% yield). ¾ NMR (400 MHz, CDCb) δ 7.88 (d, / = 8.4 Hz, 2H), 7.88 – 7.77 (m, 1H), 7.58 (dd, J = 7.5, 2.4 Hz, 1H), 7.05 (d, J = 8.3 Hz, 2H), 6.90 – 6.80 (m, 2H), 4.94 (s, 2H), 4.82 – 4.68 (m, 2H), 3.34 (s, 3H), 2.27 (dd, / = 12.4, 5.7 Hz, 1H), 2.09 – 1.91 (m, 1H), 1.91 – 1.67 (m, 3H), 1.67 – 1.49 (m, 1H).MS (ESI) m/z 513.3 [M+H]+.
NEW YORK and OSAKA, Japan, Nov. 3, 2014 (GLOBE NEWSWIRE) — Intra-Cellular Therapies, Inc. (Nasdaq:ITCI) and Takeda Pharmaceutical Company Limited announced today that they have entered into an agreement to mutually terminate the February 2011 license agreement covering Intra-Cellular Therapies’ proprietary compound ITI-214 and related PDE1 inhibitors and to return the rights for these compounds to Intra-Cellular Therapies.
|
||||||
Under the terms of the agreement, Intra-Cellular Therapies has regained all worldwide development and commercialization rights for the compounds previously licensed to Takeda. Takeda will be responsible for transitioning the compounds back toIntra-Cellular Therapies and will not participate in future development or commercialization activities. After transition of the program, Intra-Cellular Therapies plans to continue the clinical development of PDE1 inhibitors for the treatment of central nervous system, cardiovascular and other disorders.
“We are grateful for Takeda’s substantial efforts in advancing this program into clinical development,” said Dr. Sharon Mates, Chairman and CEO of Intra-Cellular Therapies. “This provides us with the opportunity to unify our PDE1 platform and we look forward to continuing the development of ITI-214 and our other PDE1 inhibitors.”
Intra-Cellular Therapies will discuss the PDE1 program in its previously announced earnings call on Monday, November 3, 2014 at 8:30 a.m. Eastern Time. To participate in the conference call, please dial 844-835-6563 (U.S.) or 970-315-3916 (International) five to ten minutes prior to the start of the call. The participant passcode is 25568442.
About PDE1 Inhibitors
PDE1 inhibitors are unique, orally available, investigational drug candidates being developed for the treatment of cognitive impairments accompanying schizophrenia, Alzheimer’s disease and other neuropsychiatric disorders and neurological diseases and may also treat patients with Attention Deficit Hyperactivity Disorder and Parkinson’s disease. These compounds may also have the potential to improve motor dysfunction associated with these conditions and may also have the potential to treat patients with multiple sclerosis and other autoimmune diseases and pulmonary arterial hypertension. These compounds are very selective for the PDE1 subfamily relative to other PDE subfamilies. They have no known significant off target activities at other enzymes, receptors or ion channels.
About Intra-Cellular Therapies
Intra-Cellular Therapies, Inc. (the “Company”) is developing novel drugs for the treatment of neuropsychiatric and neurodegenerative disease and other disorders of the central nervous system (“CNS”). The Company is developing its lead drug candidate, ITI-007, for the treatment of schizophrenia, behavioral disturbances in dementia, bipolar disorder and other neuropsychiatric and neurological disorders. The Company is also utilizing its phosphodiesterase platform and other proprietary chemistry platforms to develop drugs for the treatment of CNS disorders.
About Takeda Pharmaceutical Company Limited
Located in Osaka, Japan, Takeda is a research-based global company with its main focus on pharmaceuticals. As the largest pharmaceutical company in Japan and one of the global leaders of the industry, Takeda is committed to strive towards better health for people worldwide through leading innovation in medicine. Additional information about Takeda is available through its corporate website, www.Takeda.com.
Source: Intra-Cellular Therapies, Inc.; Takeda Pharmaceutical Company Limited
| US20080188492 * | Jun 6, 2006 | Aug 7, 2008 | Intra-Cellular Therapies, Inc | Organic Compounds |
| US20100273754 * | Dec 6, 2008 | Oct 28, 2010 | Peng Li | Organic compounds |
| US20110237561 * | Dec 7, 2009 | Sep 29, 2011 | Peng Li | Organic compounds |
| US20120071450 * | Dec 7, 2009 | Mar 22, 2012 | Peng Li | Organic compounds |
| US20120238589 * | Sep 20, 2012 | Peng Li | Organic compounds |
| WO2014205354A3 * | Jun 20, 2014 | May 28, 2015 | Takeda Pharmaceutical Company Limited | Free base crystals |
| WO2015196186A1 * | Jun 22, 2015 | Dec 23, 2015 | Intra-Cellular Therapies, Inc. | Organic compounds |
| US8829008 | Jun 1, 2012 | Sep 9, 2014 | Takeda Pharmaceutical Company Limited | Organic compounds |
| US9000001 | Jul 18, 2012 | Apr 7, 2015 | Intra-Cellular Therapies, Inc. | Organic compounds |
| US9006258 | Dec 5, 2007 | Apr 14, 2015 | Intra-Cellular Therapies, Inc. | Method of treating female sexual dysfunction with a PDE1 inhibitor |
| US9073936 | Mar 13, 2014 | Jul 7, 2015 | Intra-Cellular Therapies, Inc. | Organic compounds |
| WO2009075784A1 * | Dec 6, 2008 | Jun 18, 2009 | Intra Cellular Therapies Inc | Organic compounds |
| WO2010065151A1 * | Dec 7, 2009 | Jun 10, 2010 | Intra-Cellular Therapies, Inc. | Organic compounds |
| WO2013192556A2 * | Jun 21, 2013 | Dec 27, 2013 | Intra-Cellular Therapies, Inc. | Salt crystal |
//////
O=C(C1=C(NC2=CC=CC=C2)N(CC3=CC=C(C4=NC(F)=CC=C4)C=C3)N=C1N56)N(C)C5=N[C@@]7([H])[C@]6([H])CCC7.O=P(O)(O)O
OR
Fc1cccc(n1)c2ccc(cc2)Cn7nc5N3C(=N[C@@H]4CCC[C@H]34)N(C)C(=O)c5c7Nc6ccccc6
DRUG REGULATORY AFFAIRS INTERNATIONAL

The FDA has presented the draft of a revised guideline on dissolution testing for immediate release. Under certain conditions, the tests can now be standardised. Read on to get more information about FDA’s Guideline on Dissolution Testing.
http://www.gmp-compliance.org/enews_05230_FDA-Guideline-on-Dissolution-Testing_15398,Z-QCM_n.html
In August 2015, the FDA published the draft of a guideline on dissolution testing for immediate release solid oral dosage forms. It is planned that after its finalisation, a part of this guideline will replace the current guideline from August 1997.
The Biopharmaceutics Classification System (BCS) distinguishes 4 different classes of APIs depending on their solubility and permeability.
On the basis of this classification, a decision can be taken for determining when bioavailability or bioequivalence studies are required, or when a successful in vitro-in vivo correlation (IVIVC) is likely.
The BCS proposes that, for certain medicinal products which contain a high soluble API, dissolution testing can be standardised. Due to their high solubility…
View original post 121 more words

Supplier Qualification is more than auditing. Supplier qualification can be seen as a risk assessment tool. But what are the exact requirements for qualifying suppliers?
Supplier Qualification is more than auditing. Supplier qualification can be seen as a risk assessment tool. It should provide an appropriate level of confidence that suppliers, vendors and contractors are able to supply consistent quality of materials, components and services in compliance with regulatory requirements. An integrated supplier qualification process should also identify and mitigate the associated risks of materials, components and services. But what are the exact requirements?
They are wide-ranging and complex. There are different directives and regulations for medicinal drug products for human or veterinary use and for investigational medicinal drug products. Certain requirements in different directives and the EU-GMP Guidelines define expectations. Here are some examples:
Article 8 of EU-Directive 2001/83/EC
“The application [of a marketing authorization] shall be accompanied […] by […] a written confirmation that the manufacturer of the medicinal product has verified compliance of the manufacturer of active substance with principles and guidelines of good manufacturing practice by conducting audits.”
Article 46 of EU-Directive 2001/83/EC
“The holder of a manufacturing and/or import authorisation shall at least be obliged […] to use only active substances, which have been manufactured in accordance with GMP for active substances and distributed in accordance with GDP for active substances and … to ensure that the excipients are suitable for use in medicinal products by ascertaining what the appropriate GMP is.”
Article 46b of EU-Directive 2001/83/EC
“Active substances shall only be imported if they have been manufactured in accordance with standards of good manufacturing practice at least equivalent to those laid down by the European Union”. This can be shown by a written confirmation, or the exporting country is included in the so called white list, or a waiver has been granted.
EU-GMP Guidelines Chapter 5:
5.25 “The purchase of starting materials is an important operation which should involve staff who have a particular and thorough knowledge of the supplier.”
5.26 “Starting materials should only be purchased from approved suppliers …”
5.40 “…printed packaging materials shall be accorded attention similar to that given to starting materials.”
The revised Chapter 7 of the EU-GMP Guidelines describe the responsibilities of the Contract Giver when it comes to contract manufacturing and testing. He needs to assure the control of the outsourced activities, incorporating quality risk management principles and including continuous reviews of the quality of the Contract Acceptor’s performance. Audits are a helpful tool to asses the “legality, suitability and the competence of the Contract Acceptor“. The new Chapter 7 was obviously designed to intensify the control of Contract Acceptors by the Contract Giver and extend those controls to subcontractors.
The holder of the manufacturing authorisation is responsible for the supplier qualification by law but in fact the supplier qualification is one of the duties of the Qualified Person (which can be delegated) as defined in Annex 16 of the EU-GMP Guidelines. The QP of the marketing authorisation holder is responsible for certifying the drug product for the market place and is now being held accountable to ensure that all aspects of the supply chain have been made under the appropriate GMPs. However, according to Chapter 2 of the EU-GMP Guidelines, the heads of Production, Quality Control and Quality Assurance share the responsibility of approving and monitoring suppliers of materials (2.9).
So how to proceed? At the beginning of a supplier qualification process, the regulatory requirements regarding the type of material, component or service and the type of product (human/veterinary drug product or IMP) should be identified and specified. Audits, if required, should be planned and executed. The compliance of the selected supplier(s) with the requirements and user requirement specification should be demonstrated. The scope of an audit should cover this. But a successful audit is not the end of the qualification process. After finalising the contract, the compliance of the selected supplier(s) with the applicable requirements should be evaluated periodically. Changes at the supplier´s site (for example manufacturing process etc.) that pose a particular risk to the compliance with the requirements should be assessed. There needs to be a mechanism in place so that any change made by the supplier which could have an impact on the GMP status or the production or testing parameters have to be agreed to before any such changes are implemented. A supplier must also notify the contract giver immediately upon discovery of any deviation/non-conformance/complaint that may have an impact on the services provided. Those need to be assessed and respective actions need to be defined.
The use of Brokers:
Some raw materials are only available at reasonable costs if purchased through an intermediary, i.e. a Broker. If the material is critical to the process, e.g. an API or a key excipient this can give an added complexity to the process and this must be fully investigated with the Quality and Regulatory units being involved, before any orders are placed.







////////