Lumasiran
 |
The molecular formula of lumasiran sodium is C530H669F10N173O320P43S6Na43 and the molecular weight is 17,286 Da.
lumasiran
CAS 1834610-13-7
FDA APPROVED, 11/23/2020, Oxlumo
To treat hyperoxaluria type 1
Press Release
Drug Trials Snapshot
RNA, (Gm-sp-Am-sp-Cm-Um-Um-Um-(2′-deoxy-2′-fluoro)C-Am-(2′-deoxy-2′-fluoro)U-(2′-deoxy-2′-fluoro)C-(2′-deoxy-2′-fluoro)C-Um-Gm-Gm-Am-Am-Am-Um-Am-Um-Am), 3′-[[(2S,4R)-1-[29-[[2-(acetylamino)-2-deoxy-β-D-galactopyranosyl]oxy]-14,14-bis[[3-[[3-[[5-[[2-(acetylamino)-2-deoxy-β-D-galactopyranosyl]oxy]-1-oxopentyl]amino]propyl]amino]-3-oxopropoxy]methyl]-1,12,19,25-tetraoxo-16-oxa-13,20,24-triazanonacos-1-yl]-4-hydroxy-2-pyrrolidinyl]methyl hydrogen phosphate], complex with RNA (Um-sp-(2′-deoxy-2′-fluoro)A-sp-Um-Am-Um-(2′-deoxy-2′-fluoro)U-Um-(2′-deoxy-2′-fluoro)C-(2′-deoxy-2′-fluoro)C-Am-Gm-Gm-Am-(2′-deoxy-2′-fluoro)U-Gm-(2′-deoxy-2′-fluoro)A-Am-Am-Gm-Um-Cm-sp-Cm-sp-Am) (1:1)
Nucleic Acid Sequence
Sequence Length: 44, 23, 2115 a 8 c 7 g 14 umultistranded (2); modified
OXLUMO is supplied as a sterile, preservative-free, clear, colorless-to-yellow solution for subcutaneous administration containing the equivalent of 94.5 mg of lumasiran (provided as lumasiran sodium) in 0.5 Ml of water for injection and sodium hydroxide and/or phosphoric acid to adjust the pH to ~7.0.
Lumasiran An investigational RNAi Therapeutic for Primary Hyperoxaluria Type 1 (PH1)
Overview • Lumasiran (ALN-GO1) is an investigational, subcutaneously administered (under the skin) RNA interference (RNAi) therapeutic targeting glycolate oxidase (GO) in development for the treatment of primary hyperoxaluria type 1 (PH1).
• PH1 is a rare, life-threatening disease that can cause serious damage to kidneys and progressively to other organs.1
• PH1 is characterized by the pathologic overproduction of oxalate by the liver. Oxalate is an end product of metabolism that, when in excess, is toxic and accumulates in the kidneys forming calcium oxalate crystals.1,2
• Symptoms of PH1 are often associated with recurrent kidney stones and include flank pain, urinary tract infections, painful urination, and blood in the urine.2,3
• Currently, the only curative treatment is a liver transplant, to correct the metabolic defect, combined with a kidney transplant, to replace the terminally damaged kidneys.1,3 Clinical Development
• The safety and efficacy of lumasiran are being evaluated in a randomized, double-blind, placebo-controlled, global, multicenter Phase 3 study of approximately 30 PH1 patients, called ILLUMINATE-A (NCT03681184).
• The primary endpoint is percent change in 24-hour urinary oxalate excretion from baseline to Month 6.
• Key secondary and exploratory endpoints in ILLUMINATE-A will evaluate additional measures of urinary oxalate, estimated glomerular filtration rate (eGFR), safety, and tolerability.
Regulatory Designations • Breakthrough Therapy Designation by the U.S. Food and Drug Administration (FDA) • Priority Medicines (PRIME) Designation from the European Medicines Agency (EMA) • Orphan Drug Designations in both the U.S. and the European Union

/////////lumasiran, fda 2020, 2020 approvals, Oxlumo, Breakthrough Therapy Designation, Orphan Drug, Priority Medicines (PRIME) Designation
Setmelanotide

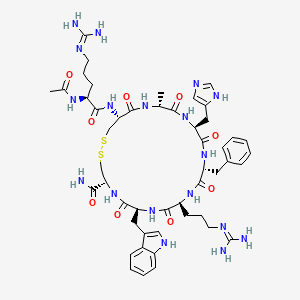

Setmelanotide
Ac-Arg-Cys(1)-D-Ala-His-D-Phe-Arg-Trp-Cys(1)-NH2
- Molecular FormulaC49H68N18O9S2
- Average mass1117.309 Da
- N-acetyl-L-arginyl-L-cysteinyl-D-alanyl-L-histidyl-D-phenylalanyl-L-arginyl-L-tryptophyl-L-cysteinamide (2->8)-disulfide
1,2-Dithia-5,8,11,14,17,20-hexaazacyclotricosane-4-carboxamide, 22-[[(2S)-2-(acetylamino)-5-[(diaminomethylene)amino]-1-oxopentyl]amino]-10-[3-[(diaminomethylene)amino]propyl]-16-(1H-imidazol-5-ylmeth yl)-7-(1H-indol-3-ylmethyl)-19-methyl-6,9,12,15,18,21-hexaoxo-13-(phenylmethyl)-, (4R,7S,10S,13R,16S,19R,22R)- [ACD/Index Name]10011920014-72-8[RN]Imcivree [Trade name]N2-acetyl-L-arginyl-L-cysteinyl-D-alanyl-L-histidyl-D-phenylalanyl-L-arginyl-Ltryptophyl- L-cysteinamide, cyclic (2-8)-disulfideN7T15V1FUYRM-493, BIM-22493UNII-N7T15V1FUYсетмеланотид [Russian] [INN]سيتميلانوتيد [Arabic] [INN]司美诺肽 [Chinese] [INN](4R,7S,10S,13R,16S,19R,22R)-22-[[(2S)-2-acetamido-5-(diaminomethylideneamino)pentanoyl]amino]-13-benzyl-10-[3-(diaminomethylideneamino)propyl]-16-(1H-imidazol-5-ylmethyl)-7-(1H-indol-3-ylmethyl)-19-methyl-6,9,12,15,18,21-hexaoxo-1,2-dithia-5,8,11,14,17,20-hexazacyclotricosane-4-carboxamide
FDA 11/25/2020, Imcivree, To treat obesity and the control of hunger associated with pro-opiomelanocortin deficiency, a rare disorder that causes severe obesity that begins at an early age
Drug Trials Snapshot, 10MG/ML, SOLUTION;SUBCUTANEOUS, Orphan


update Imcivree EMA APPROVED 2021/7/16
DESCRIPTION
IMCIVREE contains setmelanotide acetate, a melanocortin 4 (MC4) receptor agonist. Setmelanotide is an 8 amino acid cyclic peptide analog of endogenous melanocortin peptide α-MSH (alpha-melanocyte stimulating hormone).
The chemical name for setmelanotide acetate is acetyl-L-arginyl-L-cysteinyl-D-alanyl-Lhistidinyl-D-phenylalanyl-L-arginyl-L-tryptophanyl-L-cysteinamide cyclic (2→8)-disulfide acetate. Its molecular formula is C49H68N18O9S2 (anhydrous, free-base), and molecular mass is 1117.3 Daltons (anhydrous, free-base).
The chemical structure of setmelanotide is:
 |
IMCIVREE injection is a sterile clear to slightly opalescent, colorless to slightly yellow solution. Each 1 mL of IMCIVREE contains 10 mg of setmelanotide provided as setmelanotide acetate, which is a salt with 2 to 4 molar equivalents of acetate, and the following inactive ingredients: 100 mg N-(carbonyl-methoxypolyethylene glycol 2000)-1,2-distearoyl-glycero-3phosphoethanolamine sodium salt, 8 mg carboxymethylcellulose sodium (average MWt 90,500), 11 mg mannitol, 5 mg phenol, 10 mg benzyl alcohol, 1 mg edetate disodium dihydrate, and Water for Injection. The pH of IMCIVREE is 5 to 6.
Setmelanotide is a peptide drug and investigational anti-obesity medication which acts as a selective agonist of the MC4 receptor. Setmelanotide binds to and activates MC4 receptors in the paraventricular nucleus (PVN) of the hypothalamus and in the lateral hypothalamic area (LHA), areas involved in the regulation of appetite, and this action is thought to underlie its appetite suppressant effects. Setmelanotide increases resting energy expenditure in both obese animals and humans. Setmelanotide has been reported to possess the following activity profile (cAMP, EC50): MC4 (0.27 nM) > MC3 (5.3 nM) ≈ MC1 (5.8 nM) > MC5 (1600 nM) ≟ MC2 (>1000 nM).
Setmelanotide, sold under the brand name Imcivree, is a medication for the treatment of obesity.[1]
The most common side effects include injection site reactions, skin hyperpigmentation (skin patches that are darker than surrounding skin), headache and gastrointestinal side effects (such as nausea, diarrhea, and abdominal pain), among others.[1] Spontaneous penile erections in males and adverse sexual reactions in females have occurred with treatment.[1] Depression and suicidal ideation have also occurred with setmelanotide.[1]
SYN
WO 2011060355



Medical uses
Setmelanotide is indicated for chronic weight management (weight loss and weight maintenance for at least one year) in people six years and older with obesity due to three rare genetic conditions: pro-opiomelanocortin (POMC) deficiency, proprotein subtilisin/kexin type 1 (PCSK1) deficiency, and leptin receptor (LEPR) deficiency confirmed by genetic testing demonstrating variants in POMC, PCSK1, or LEPR genes considered pathogenic (causing disease), likely pathogenic, or of uncertain significance.[1] Setmelanotide is the first FDA-approved treatment for these genetic conditions.[1]
Setmelanotide is not approved for obesity due to suspected POMC, PCSK1, or LEPR deficiency with variants classified as benign (not causing disease) or likely benign or other types of obesity, including obesity associated with other genetic syndromes and general (polygenic) obesity.[1]
Setmelanotide binds to and activates MC4 receptors in the paraventricular nucleus (PVN) of the hypothalamus and in the lateral hypothalamic area (LHA), areas involved in the regulation of appetite, and this action is thought to underlie its appetite suppressant effects.[2] In addition to reducing appetite, setmelanotide increases resting energy expenditure in both obese animals and humans.[3] Importantly, unlike certain other MC4 receptor agonists, such as LY-2112688, setmelanotide has not been found to produce increases in heart rate or blood pressure.[4]
Setmelanotide has been reported to possess the following activity profile (cAMP, EC50): MC4 (0.27 nM) > MC3 (5.3 nM) ≈ MC1 (5.8 nM) > MC5 (1600 nM) ≟ MC2 (>1000 nM).[5] (19.6-fold selectivity for MC4 over MC3, the second target of highest activity.)
History
Setmelanotide was evaluated in two one-year studies.[1] The first study enrolled participants with obesity and confirmed or suspected POMC or PCSK1 deficiency while the second study enrolled participants with obesity and confirmed or suspected LEPR deficiency; all participants were six years or older.[1] The effectiveness of setmelanotide was determined by the number of participants who lost more than ten percent of their body weight after a year of treatment.[1]
The effectiveness of setmelanotide was assessed in 21 participants, ten in the first study and eleven in the second.[1] In the first study, 80 percent of participants with POMC or PCSK1 deficiency lost ten percent or more of their body weight.[1] In the second study, 46 percent of participants with LEPR deficiency lost ten percent or more of their body weight.[1]
The study also assessed the maximal (greatest) hunger in sixteen participants over the previous 24 hours using an eleven-point scale in participants twelve years and older.[1] In both studies, some, but not all, of participants’ weekly average maximal hunger scores decreased substantially from their scores at the beginning of the study.[1] The degree of change was highly variable among participants.[1]
The U.S. Food and Drug Administration (FDA) granted the application for setmelanotide orphan disease designation, breakthrough therapy designation, and priority review.[1] The FDA granted the approval of Imcivree to Rhythm Pharmaceutical, Inc.[1]
Research
Setmelanotide is a peptide drug and investigational anti-obesity medication which acts as a selective agonist of the MC4 receptor.[6][4] Its peptide sequence is Ac-Arg-Cys(1)-D-Ala-His-D-Phe-Arg-Trp-Cys(1)-NH2. It was first discovered at Ipsen and is being developed by Rhythm Pharmaceuticals for the treatment of obesity and diabetes.[6] In addition, Rhythm Pharmaceuticals is conducting trials of setmelanotide for the treatment of Prader–Willi syndrome (PWS), a genetic disorder which includes MC4 receptor deficiency and associated symptoms such as excessive appetite and obesity.[7] As of December 2014, the drug is in phase II clinical trials for obesity and PWS.[6][8][9][needs update] So far, preliminary data has shown no benefit of Setmelanotide in Prader-Willi syndrome.[10]
PATENT
WO 2007008704
WO 2011060355
WO 2011060352
US 20120225816
PAPER
Journal of Medicinal Chemistry, 61(8), 3674-3684; 2018
PATENT
https://patents.google.com/patent/US9314509
Synthesis of Example 1i.e., Ac-Arg-cyclo(Cys-D-Ala-His-D-Phe-Arg-Trp-Cys)-NH2

The title peptide having the above structure was assembled using Fmoc chemistry on an Apex peptide synthesizer (Aapptec; Louisville, Ky., USA). 220 mg of 0.91 mmol/g (0.20 mmoles) Rink Amide MBHA resin (Polymer Laboratories; Amherst, Mass., USA) was placed in a reaction well and pre-swollen in 3.0 mL of DMF prior to synthesis. For cycle 1, the resin was treated with two 3-mL portions of 25% piperidine in DMF for 5 and 10 minutes respectively, followed by 4 washes of 3-mL DMF—each wash consisting of adding 3 mL of solvent, mixing for 1 minute, and emptying for 1 minute. Amino acids stocks were prepared in NMP as 0.45M solutions containing 0.45M HOBT. HBTU was prepared as a 0.45M solution in NMP and DIPEA was prepared as a 2.73M solution in NMP. To the resin, 2 mL of the first amino acid (0 9 mmoles, Fmoc-Cys(Trt)-OH) (Novabiochem; San Diego, Calif., USA) was added along with 2 mL (0.9 mmoles) of HBTU and 1.5 mL (4.1 mmoles) of DIPEA. After one hour of constant mixing, the coupling reagents were drained from the resin and the coupling step was repeated. Following amino acid acylation, the resin was washed with two 3-mL aliquots of DMF for 1 minute. The process of assembling the peptide (deblock/wash/acylate/wash) was repeated for cycles 2-9 identical to that as described for cycle 1. The following amino acids were used: cycle 2) Fmoc-Trp(Boc)-OH (Genzyme; Cambridge, Mass., USA); cycle 3) Fmoc-Arg(Pbf)-OH (Novabiochem); cycle 4) Fmoc-DPhe-OH (Genzyme); cycle 5) Fmoc-His(Trt)-OH (Novabiochem); cycle 6) Fmoc-D-Ala-OH (Genzyme); cycle 7) Fmoc-Cys(Trt)-OH, (Novabiochem); and cycle 8) Fmoc-Arg(Pbf)-OH (Genzyme). The N-terminal Fmoc was removed with 25% piperidine in DMF as described above, followed by four 3-mL DMF washes for 1 minute. Acetylation of the N-terminus was performed by adding 0.5 mL of 3M DIPEA in NMP to the resin along with 1.45 mL of 0.45M acetic anhydride in NMP. The resin was mixed for 30 minutes and acetylation was repeated. The resin was washed with 3 mL of DMF for a total of 5 times followed with 5 washes with 5 mL of DCM each.
To cleave and deprotect the peptide, 5mL of the following reagent was added to the resin: 2% TIS/5% water/5% (w/v) DTT/88% TFA. The solution was allowed to mix for 3.5 hours. The filtrate was collected into 40 mL of cold anhydrous ethyl ether. The precipitate was pelleted for 10 minutes at 3500 rpm in a refrigerated centrifuge. The ether was decanted and the peptide was re-suspended in fresh ether. The ether workup was performed three times. Following the last ether wash, the peptide was allowed to air dry to remove residual ether.
The peptide was dissolved in 10% acetonitrile and analyzed by mass spectrometry and reverse-phase HPLC employing a 30×4.6 cm C18 column (Vydac; Hesperia, Calif., USA) with a gradient of 2-60% acetonitrile (0.1% TFA) over 30 minutes. This analysis identified a product with ˜53% purity. Mass analysis employing electrospray ionization identified a main product containing a mass of 1118.4 corresponding to the desired linear product. The crude product (˜100 mg) was diluted to a concentration of 2 mg/mL in 5% acetic acid. To this solution, 0.5M iodine/methanol was added dropwise with vigorous stirring until a pale yellow color was achieved. The solution was vigorously stirred for another 10 minutes. Excess iodine was then quenched by adding 1.0M sodium thiosulfate under continuous mixing until the mixture was rendered colorless. The peptide was re-examined by mass spectrometry analysis and HPLC. Mass spectrometry analysis identified a main species with a mass of 1116.4 which indicated successful oxidation to form the cyclic peptide. The peptide solution was purified on a preparative HPLC equipped with a C18 column using a similar elution gradient. The purified product was re-analyzed by HPLC for purity (>95%) and mass spectrometry (1116.9 which is in agreement with the expected mass of 1117.3) and subsequently lyophilized. Following lyophilization, 28 mg of purified product was obtained representing a 24% yield.
The other exemplified peptides were synthesized substantially according to the procedure described for the above-described synthetic process. Physical data for select exemplified peptides are given in Table 1.
TABLE 1 Example Mol. Wt. Mol. Wt. Purity Number (calculated) (ES-MS) (HPLC) 1 1117.3 1116.9 95.1% 2 1117.3 1116.8 99.2% 3 1280.5 1280.6 98.0% 5 1216.37 1216.20 99.9%
Preparation of Pamoate Salt of Example 1
The acetate salt of Example 1 (200 mg, 0.18 mmole) was dissolved in 10 mL of water. Sodium pamoate (155 mg, 0.36 mmole) was dissolved in 10 mL of water. The two solutions were combined and mixed well. The precipitates were collected by centrifugation at 3000 rpm for 20 minutes, washed for three times with water, and dried by lyophilization.
References
- ^ Jump up to:a b c d e f g h i j k l m n o p q r “FDA approves first treatment for weight management for people with certain rare genetic conditions”. U.S. Food and Drug Administration (FDA) (Press release). 27 November 2020. Retrieved 27 November 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain. - ^ Kim GW, Lin JE, Blomain ES, Waldman SA (January 2014). “Antiobesity pharmacotherapy: new drugs and emerging targets”. Clinical Pharmacology and Therapeutics. 95 (1): 53–66. doi:10.1038/clpt.2013.204. PMC 4054704. PMID 24105257.
- ^ Chen KY, Muniyappa R, Abel BS, Mullins KP, Staker P, Brychta RJ, et al. (April 2015). “RM-493, a melanocortin-4 receptor (MC4R) agonist, increases resting energy expenditure in obese individuals”. The Journal of Clinical Endocrinology and Metabolism. 100 (4): 1639–45. doi:10.1210/jc.2014-4024. PMC 4399297. PMID 25675384.
- ^ Jump up to:a b Kievit P, Halem H, Marks DL, Dong JZ, Glavas MM, Sinnayah P, et al. (February 2013). “Chronic treatment with a melanocortin-4 receptor agonist causes weight loss, reduces insulin resistance, and improves cardiovascular function in diet-induced obese rhesus macaques”. Diabetes. 62 (2): 490–7. doi:10.2337/db12-0598. PMC 3554387. PMID 23048186.
- ^ Muniyappa R, Chen K, Brychta R, Abel B, Mullins K, Staker P, et al. (June 2014). “A Randomized, Double-Blind, Placebo-Controlled, Crossover Study to Evaluate the Effect of a Melanocortin Receptor 4 (MC4R) Agonist, RM-493, on Resting Energy Expenditure (REE) in Obese Subjects” (PDF). Endocrine Reviews. Rhythm Pharmaceuticals. 35 (3). Retrieved 2015-05-21.
- ^ Jump up to:a b c Lee EC, Carpino PA (2015). “Melanocortin-4 receptor modulators for the treatment of obesity: a patent analysis (2008-2014)”. Pharmaceutical Patent Analyst. 4 (2): 95–107. doi:10.4155/ppa.15.1. PMID 25853469.
- ^ “Obesity and Diabetes Caused by Genetic Deficiencies in the MC4 Pathway”. Rhythm Pharmaceuticals. Retrieved 2015-05-21.
- ^ Jackson VM, Price DA, Carpino PA (August 2014). “Investigational drugs in Phase II clinical trials for the treatment of obesity: implications for future development of novel therapies”. Expert Opinion on Investigational Drugs. 23 (8): 1055–66. doi:10.1517/13543784.2014.918952. PMID 25000213. S2CID 23198484.
- ^ “RM-493: A First-in-Class, Phase 2-Ready MC4 Agonist: A New Drug Class for the Treatment of Obesity and Diabetes”. Rhythm Pharmaceuticals. Archived from the original on 2015-06-14. Retrieved 2015-05-21.
- ^ Duis J, van Wattum PJ, Scheimann A, Salehi P, Brokamp E, Fairbrother L, et al. (March 2019). “A multidisciplinary approach to the clinical management of Prader-Willi syndrome”. Molecular Genetics & Genomic Medicine. 7 (3): e514. doi:10.1002/mgg3.514. PMC 6418440. PMID 30697974.
ADDITIONAL INFORMATION
The peptide sequence is Ac-Arg-Cys(1)-D-Ala-His-D-Phe-Arg-Trp-Cys(1)-NH2. It is being researched by Rhythm Pharmaceuticals for the treatment of obesity and diabetes. In addition, Rhythm Pharmaceuticals is conducting trials of setmelanotide for the treatment of Prader–Willi syndrome (PWS), a genetic disorder which includes MC4 receptor deficiency and associated symptoms such as excessive appetite and obesity. As of December 2014, the drug is in phase II clinical trials for obesity and PWS.
L-Cysteinamide, N2-acetyl-L-arginyl-L-cysteinyl-D-alanyl-L-histidyl-D-phenylalanyl-L-arginyl-L-tryptophyl-, cyclic (2->8)-disulfide
Ac-Arg-Cys(1)-D-Ala-His-D-Phe-Arg-Trp-Cys(1)-NH2
REFERENCES
1: Lee EC, Carpino PA. Melanocortin-4 receptor modulators for the treatment of obesity: a patent analysis (2008-2014). Pharm Pat Anal. 2015;4(2):95-107. doi: 10.4155/ppa.15.1. PubMed PMID: 25853469.
2: Chen KY, Muniyappa R, Abel BS, Mullins KP, Staker P, Brychta RJ, Zhao X, Ring M, Psota TL, Cone RD, Panaro BL, Gottesdiener KM, Van der Ploeg LH, Reitman ML, Skarulis MC. RM-493, a melanocortin-4 receptor (MC4R) agonist, increases resting energy expenditure in obese individuals. J Clin Endocrinol Metab. 2015 Apr;100(4):1639-45. doi: 10.1210/jc.2014-4024. Epub 2015 Feb 12. PubMed PMID: 25675384; PubMed Central PMCID: PMC4399297.
3: Clemmensen C, Finan B, Fischer K, Tom RZ, Legutko B, Sehrer L, Heine D, Grassl N, Meyer CW, Henderson B, Hofmann SM, Tschöp MH, Van der Ploeg LH, Müller TD. Dual melanocortin-4 receptor and GLP-1 receptor agonism amplifies metabolic benefits in diet-induced obese mice. EMBO Mol Med. 2015 Feb 4;7(3):288-98. doi: 10.15252/emmm.201404508. PubMed PMID: 25652173; PubMed Central PMCID: PMC4364946.
4: Jackson VM, Price DA, Carpino PA. Investigational drugs in Phase II clinical trials for the treatment of obesity: implications for future development of novel therapies. Expert Opin Investig Drugs. 2014 Aug;23(8):1055-66. doi: 10.1517/13543784.2014.918952. Epub 2014 Jul 7. Review. PubMed PMID: 25000213.
5: Kievit P, Halem H, Marks DL, Dong JZ, Glavas MM, Sinnayah P, Pranger L, Cowley MA, Grove KL, Culler MD. Chronic treatment with a melanocortin-4 receptor agonist causes weight loss, reduces insulin resistance, and improves cardiovascular function in diet-induced obese rhesus macaques. Diabetes. 2013 Feb;62(2):490-7. doi: 10.2337/db12-0598. Epub 2012 Oct 9. PubMed PMID: 23048186; PubMed Central PMCID: PMC3554387.
6: Kumar KG, Sutton GM, Dong JZ, Roubert P, Plas P, Halem HA, Culler MD, Yang H, Dixit VD, Butler AA. Analysis of the therapeutic functions of novel melanocortin receptor agonists in MC3R- and MC4R-deficient C57BL/6J mice. Peptides. 2009 Oct;30(10):1892-900. doi: 10.1016/j.peptides.2009.07.012. Epub 2009 Jul 29. PubMed PMID: 19646498; PubMed Central PMCID: PMC2755620.
External links
- “Setmelanotide”. Drug Information Portal. U.S. National Library of Medicine.
| Clinical data | |
|---|---|
| Trade names | Imcivree |
| Other names | RM-493; BIM-22493; IRC-022493; N2-Acetyl-L-arginyl-L-cysteinyl-D-alanyl-L-histidyl-D-phenylalanyl-L-arginyl-L-tryptophyl-L-cysteinamide, cyclic (2-8)-disulfide |
| ATC code | None |
| Legal status | |
| Legal status | US: ℞-only |
| Identifiers | |
| IUPAC name[show] | |
| CAS Number | 920014-72-8 |
| PubChem CID | 11993702 |
| ChemSpider | 10166169 |
| UNII | N7T15V1FUY |
| KEGG | D11927 |
| Chemical and physical data | |
| Formula | C49H68N18O9S2 |
| Molar mass | 1117.32 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| SMILES[hide]C[C@@H]1C(=O)N[C@H](C(=O)N[C@@H](C(=O)N[C@H](C(=O)N[C@H](C(=O)N[C@@H](CSSC[C@@H](C(=O)N1)NC(=O)[C@H](CCCN=C(N)N)NC(=O)C)C(=O)N)Cc2c[nH]c3c2cccc3)CCCN=C(N)N)Cc4ccccc4)Cc5cnc[nH]5 | |
| InChI[hide]InChI=1S/C49H68N18O9S2/c1-26-41(70)63-37(20-30-22-55-25-59-30)46(75)64-35(18-28-10-4-3-5-11-28)44(73)62-34(15-9-17-57-49(53)54)43(72)65-36(19-29-21-58-32-13-7-6-12-31(29)32)45(74)66-38(40(50)69)23-77-78-24-39(47(76)60-26)67-42(71)33(61-27(2)68)14-8-16-56-48(51)52/h3-7,10-13,21-22,25-26,33-39,58H,8-9,14-20,23-24H2,1-2H3,(H2,50,69)(H,55,59)(H,60,76)(H,61,68)(H,62,73)(H,63,70)(H,64,75)(H,65,72)(H,66,74)(H,67,71)(H4,51,52,56)(H4,53,54,57)/t26-,33+,34+,35-,36+,37+,38+,39+/m1/s1Key:HDHDTKMUACZDAA-PHNIDTBTSA-N |
///////////Setmelanotide, FDA 2020, 2020 APPROVALS, Imcivree, Orphan, PEPTIDE, ANTIOBESITY, UNII-N7T15V1FUY, сетмеланотид , سيتميلانوتيد , 司美诺肽 , BIM 22493, RM 493
CC1C(=O)NC(C(=O)NC(C(=O)NC(C(=O)NC(C(=O)NC(CSSCC(C(=O)N1)NC(=O)C(CCCN=C(N)N)NC(=O)C)C(=O)N)CC2=CNC3=CC=CC=C32)CCCN=C(N)N)CC4=CC=CC=C4)CC5=CN=CN5
Melatonin

Melatonin
| メラトニン |
| Formula | C13H16N2O2 |
|---|---|
| CAS73-31-4 | 73-31-4 |
| Mol weight | 232.2783 |
APPROVED, Melatobel, JAPAN 2020/3/25
200-659-6[EINECS]
200-797-7[EINECS]
205542[Beilstein]
73-31-4[RN]
Acetamide, N-[2-(5-methoxy-1H-indol-3-yl)ethyl]-N-[2-(5-methoxy-1H-indol-3-yl)ethyl]-Acetamide
MelatoninCAS Registry Number: 73-31-4CAS Name:N-[2-(5-Methoxy-1H-indol-3-yl)ethyl]acetamide
Additional Names:N-acetyl-5-methoxytryptamine
Trademarks: Regulin (Young)
Molecular Formula: C13H16N2O2Molecular Weight: 232.28Percent Composition: C 67.22%, H 6.94%, N 12.06%, O 13.78%
Literature References: A hormone of the pineal gland, also produced by extra-pineal tissues, that lightens skin color in amphibians by reversing the darkening effect of MSH, q.v. Melatonin has been postulated as the mediator of photic-induced antigonadotrophic activity in photoperiodic mammals and has also been shown to be involved in thermoregulation in some ectotherms and in affecting locomotor activity rhythms in sparrows. Isoln from the pineal glands of beef cattle: Lerner et al.,J. Am. Chem. Soc.80, 2587 (1958); Wurtman et al.,Science141, 277 (1963). Structure: Lerner et al.,J. Am. Chem. Soc.81, 6084 (1959). Crystal and molecular structure: A. Wakahara, Chem. Lett.1972, 1139. Synthesis from 5-methoxyindole as starting material by two different routes: Szmuszkovicz et al.,J. Org. Chem.25, 857 (1960). Biochemical role of melatonin: Chem. Eng. News45, 40 (May 1, 1967). Pharmacological studies: Barchas et al.,Nature214, 919 (1967). Identification of antigonadal action sites in mouse brain: J. D. Glass, G. R. Lynch, Science214, 821 (1981). Binding studies in human hypothalamus: S. M. Reppert et al.,Science242, 78 (1988). Efficacy in control of estrus in red deer: G. W. Asher, Anim. Reprod. Sci.22, 145 (1990). Reviews: M. K. Vaughn, Int. J. Rev. Physiol.24, 41-95 (1981); D. C.Klein et al.,Life Sci.28, 1975-1986 (1981). Book: Advan. Biosci.vol. 29, N. Birau, W. Schlott, Eds. (Pergamon Press, New York, 1981) 420 pp. Review of etiological role in clinical disease: A. Miles, D. Philbrick, Crit. Rev. Clin. Lab. Sci.25, 231-253 (1987); in psychiatric disorders: eidem,Biol. Psychiatry23, 405-425 (1988).Properties: Pale yellow leaflets from benzene, mp 116-118°. uv max: 223, 278 nm (e 27550, 6300).Melting point: mp 116-118°Absorption maximum: uv max: 223, 278 nm (e 27550, 6300)Therap-Cat-Vet: Control of estrus.
Melatonin is a hormone primarily released by the pineal gland that regulates the sleep–wake cycle.[3][4] As a dietary supplement, it is often used for the short-term treatment of insomnia, such as from jet lag or shift work, and is typically taken by mouth.[5][6][7] Evidence of its benefit for this use, however, is not strong.[8] A 2017 review found that sleep onset occurred six minutes faster with use, but found no change in total time asleep.[6] The melatonin receptor agonist medication ramelteon may work as well as melatonin supplements,[6] at greater cost but with different adverse effects, for some sleep conditions.[9]
Side effects from melatonin supplements are minimal at low doses for short durations.[3][10] They may include somnolence (sleepiness), headaches, nausea, diarrhea, abnormal dreams, irritability, nervousness, restlessness, insomnia, anxiety, migraine, lethargy, psychomotor hyperactivity, dizziness, hypertension, abdominal pain, heartburn, mouth ulcers, dry mouth, hyperbilirubinaemia, dermatitis, night sweats, pruritus, rash, dry skin, pain in the extremities, symptoms of menopause, chest pain, glycosuria (sugar in the urine), proteinuria (protein in the urine), abnormal liver function tests, increased weight, tiredness, mood swings, aggression and feeling hungover.[11][12][10][13][14] Its use is not recommended during pregnancy or breastfeeding or for those with liver disease.[7][14]
In animals (including humans), melatonin is involved in synchronizing the circadian rhythm, including sleep–wake timing, blood pressure regulation, and seasonal reproduction.[15] Many of its effects are through activation of the melatonin receptors, while others are due to its role as an antioxidant.[16][17][18] In plants, it functions to defend against oxidative stress.[19] It is also present in various foods.[10]
Melatonin was discovered in 1958.[3] It is sold over the counter in Canada and the United States;[10][13] in the United Kingdom, it is a prescription-only medication.[7] It is not approved by the US Food and Drug Administration (FDA) for any medical use.[10] In Australia and the European Union, it is indicated for difficulty sleeping in people over the age of 54.[20][11] In the European Union, it is indicated for the treatment of insomnia in children and adolescents.[12] It was approved for medical use in the European Union in 2007.[11]
SYN
https://www.ch.ic.ac.uk/local/projects/s_thipayang/synth.html
SYNTHESIS
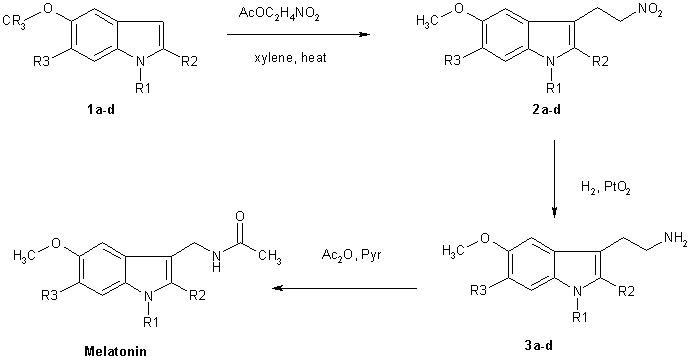
Chemical Synthesis of Melatonin
The methods for the chemical synthesis of melatonin are generally not so complicated and do not involve more than three steps of conversion. Three synthesis reactions of melatonin from primary literatures are shown below;
In 1958 melatonin was first isolated and characterised by A.B.Lerner. It was know as one of a substituted 5-hydroxyindole derivative in the pineal gland that could lighten pigment cells. It had not been know to exist in biological tissue although it had been isolated as a urinary excretion product in rats after administration of 5-hydroxytryptamine.
Melatonin or N-acetyl-5-methoxytryptamine (40 mg) was prepared by reducing 100 mg of 5-methoxyindole-3-acetonitrile with 160 mg of sodium and 2 ml of ethanol. Then the product was acetylated with 4 ml of both glacial acetic acid and acetic anhydride at 100 oC for 1 minute. Purification was achieved by countercerrent distribution and silicic acid chromatography.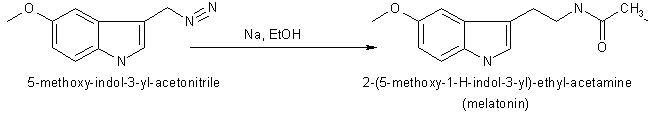
5-Methoxytryptamine hydrochloride (1g, 4.75 mmole) was dissolved in pyridine (10 ml) and acetic anhydride (10 ml) and kept overnight at 20 oC. The solution was poured onto iced, neutralised with dilute hydrochloric acid and extracted with chloroform (2×25 ml). The combined extracts were washed with water, dried in MgSO4 and evaporated to afford a liquid of N,N diacetyltryptamine derivative. The liquid was then poured into water (50 ml) and extracted with chlroform (2×25 ml). The combined organic layers were washed with water (25 ml), dried in MgSO4 and evaporated to dryness. The residual solid crystallised from benzene to afford melatonin 819 mg, 80% yield.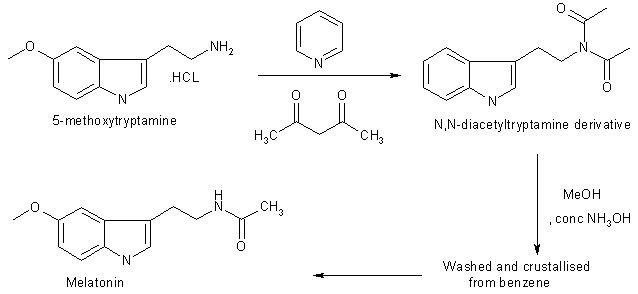
The more reactive indoles (1a-1d) were alkylated at the 3 position by reaction with nitroethene generated in situ by thermolysis of nitroethyl acetate. The nitroethyl acetate used for this purpose was prepared by acetylation of nitroethanol with acetic anhydride using NaOAc as a catalyst. These conditions constitute a substantial improvement of the overal yield of the reation. Reduction of the nitroethylated indoles (2a-d) by hydrogenation over PtO2, followed by acetylation fo the resluting tryptamines with acetic anhydride-pyridine completed the synthesis of melatonin and its derivatives (4a-d).
Biological Synthesis and Metabolism of Melatonin
The biosynthesis of melatonin (Fig.1) is initiated by the uptake of the essential amino acid tryptophan into pineal parenchymal cells. Tryptophan is the least abundant of essential amino acids in normal diets. It is converted to another amino acid, 5-hydroxytryptophan, through the action of the enzyme tryptopahn hydroxylase and then to 5-hydroxytryptamine (serotonin) by the enzyme aromatic amino acid decarboxylase. Serotonin concentrations are higher in the pineal than in any other organ or in any brain region. They exhibit a striking diurnal rhythm remaining at a maximum level during the daylight hours and falling by more than 80% soon after the onset of darkness as the serotonin is converted to melatonin, 5-hydroxytryptophol and other methoxyindoles. Serotonin’s conversion to melatonin involves two enzymes that are characteristic of the pineal : SNAT (serotonin-N-acetyltransferase) which converts the serotonin to N-acetylserotonin, and HIOMT (hydroxyindole-O-methyltrasferase) which trasfers a methyl group from S-adenosylmethionine to the 5-hydroxyl of the N-acetylserotonin. The activities of both enzymes rise soon after the onset of darkness because of the enhanced release of norepinephrine from sympathetic neurons terminating on the pineal parenchymal cells.
Another portion of the serotonin liberated from pineal cells after the onset of darkness is deaminated by the enzyme monoamine oxidase (MAO) and then either oxidized to form 5-hydroxyindole acetic acid or reduced to form 5-hydroxytryptophol (Fig.1). Both of these compounds are also substrates for HIOMT and can thus be converted in the pineal to 5-methoxyindole acetic acid 5-methoxytryptophol (Fig.1). The level of this latter indole, like that of melatonin, rises markedly in the pineal with the onset of darkness. Since 5-methoxytryptophol synthesis does not require the acetylation of serotonin, the nocturnal increase in pineal SNAT activity cannot be the trigger that causes pineal methoxyindole levels to rise. More likely, a single unexplained process- the intraparenchymal release of stored pineal serotonin, which then becomes accessible to both SNAT and MAO. This process ultimately controls the rates at which all three major pineal methoxyindoles are synthesized and generates the nocturnal increases in pineal melatonin and 5-methoxytryptophol. The proportion of available serotonin acetylated at any particular time of day or night depends on the relative activities of pineal SNAT and MAO at that time. The rates of methylation of all three 5-hydroxyindoles formed from pinela serotonin depends on HIOMT activity.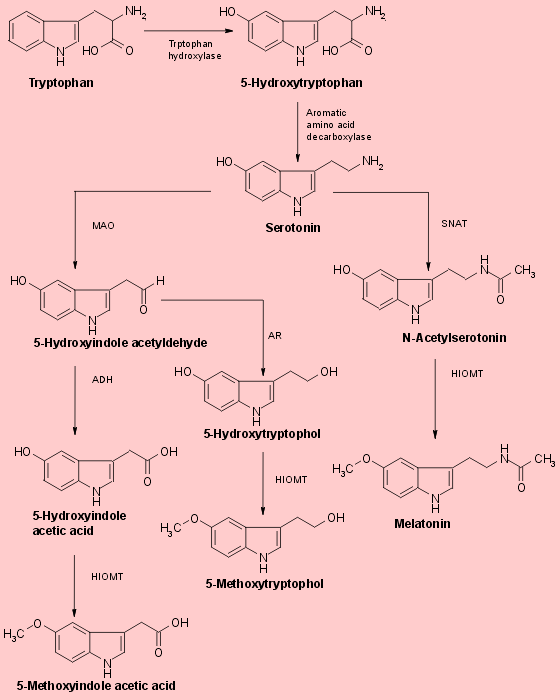 Fig.1 Biosynthesis of pineal methoxyindoles from serotonin
Fig.1 Biosynthesis of pineal methoxyindoles from serotonin
Serotonin may be either acetylated to form N-acetylserotonin through the action of the enzyme serotonin-N-acetyltransferase (SNAT), or oxidatively deaminated by monoamine oxidase (MAO) to yield an unstable aldehyde. This compound is then either oxidized to 5-hydroxyindole acetic acid by the enzyme aldehyde dehydrogenase (ADH), or reduced to from 5-hydroxytryptophol by aldehyde reductase (AR). Each of these 5-hydroxyindole derivatives of serotonin is a substrate for hydroxyindole-O-methyltrasferase (HIMOT). The enzymatic trasfer of a methyl group from S-adenosylmethionine to these hydroxyindoles yields melatonin (5-hydroxy-N-acetyltryptamine), 5-methoxyindole acetic acid and 5-methoxytryptophol respectively. Pineal serotonin is synthesized from the essential amino acid tryptophan by 5-hydroxylation folloed by decarboxylation. The first step in ths enzymic sequence is catalysed by tryptophan hydroxylase. The second step is catalysed by aromatic L-amino acid decarboxylase.
Medical uses
In the European Union it is indicated for the treatment of insomnia in children and adolescents aged 2–18 with autism spectrum disorder (ASD) and / or Smith–Magenis syndrome, where sleep hygiene measures have been insufficient[12] and for monotherapy for the short-term treatment of primary insomnia characterized by poor quality of sleep in people who are aged 55 or over.[11]
Sleep disorders
Positions on the benefits of melatonin for insomnia are mixed.[8] An Agency for Healthcare Research and Quality (AHRQ) review from 2015 stated that evidence of benefit in the general population was unclear.[8] A review from 2017, found a modest effect on time until onset of sleep.[3] Another review from 2017 put this decrease at six minutes to sleep onset but found no difference in total sleep time.[6] Melatonin may also be useful in delayed sleep phase syndrome.[3] Melatonin appears to work as well as ramelteon but costs less.[6]
Melatonin is a safer alternative than clonazepam in the treatment of REM sleep behavior disorder – a condition associated with the synucleinopathies like Parkinson’s disease and dementia with Lewy bodies.[21][22][23] In Europe it is used for short-term treatment of insomnia in people who are 55 years old or older.[24] It is deemed to be a first line agent in this group.[6]
Melatonin reduces the time until onset of sleep and increases sleep duration in children with neurodevelopmental disorders.[25]
Dementia
A 2020 Cochrane review found no evidence that melatonin helped sleep problems in people with moderate to severe dementia due to Alzheimer’s disease.[26] A 2019 review found that while melatonin may improve sleep in minimal cognitive impairment, after the onset of Alzheimer’s it has little to no effect.[27] Melatonin may, however, help with sundowning.[28]
Jet lag and shift work
Melatonin is known to reduce jet lag, especially in eastward travel. If the time it is taken is not correct, however, it can instead delay adaption.[29]
Melatonin appears to have limited use against the sleep problems of people who work shift work.[30] Tentative evidence suggests that it increases the length of time people are able to sleep.[30]
Adverse effects
Melatonin appears to cause very few side effects as tested in the short term, up to three months, at low doses.[clarification needed] Two systematic reviews found no adverse effects of exogenous melatonin in several clinical trials and comparative trials found the adverse effects headaches, dizziness, nausea, and drowsiness were reported about equally for both melatonin and placebo.[31][32] Prolonged-release melatonin is safe with long-term use of up to 12 months.[33] Although not recommended for long term use beyond this, low-dose melatonin is generally safer, and a better alternative, than many prescription and over the counter sleep aids if a sleeping medication must be used for an extended period of time. Low-doses of melatonin are usually sufficient to produce a hypnotic effect in most people. Higher doses do not appear to result in a stronger effect, but instead appear to cause drowsiness for a longer period of time.[34]
Melatonin can cause nausea, next-day grogginess, and irritability.[35] In the elderly, it can cause reduced blood flow and hypothermia.[36][needs update] In autoimmune disorders, evidence is conflicting whether melatonin supplementation may ameliorate or exacerbate symptoms due to immunomodulation.[37][38][needs update]
Melatonin can lower follicle-stimulating hormone levels.[39] Melatonin’s effects on human reproduction remain unclear.[40]
In those taking warfarin, some evidence suggests there may exist a potentiating drug interaction, increasing the anticoagulant effect of warfarin and the risk of bleeding.[41]
Functions
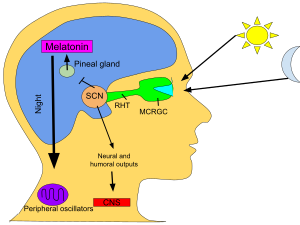
When eyes receive light from the sun, the pineal gland’s production of melatonin is inhibited and the hormones produced keep the human awake. When the eyes do not receive light, melatonin is produced in the pineal gland and the human becomes tired.
Circadian rhythm
In animals, melatonin plays an important role in the regulation of sleep–wake cycles.[42] Human infants’ melatonin levels become regular in about the third month after birth, with the highest levels measured between midnight and 8:00 am.[43] Human melatonin production decreases as a person ages.[44] Also, as children become teenagers, the nightly schedule of melatonin release is delayed, leading to later sleeping and waking times.[45]
Antioxidant
Melatonin was first reported as a potent antioxidant and free radical scavenger in 1993.[46] In vitro, melatonin acts as a direct scavenger of oxygen radicals and reactive nitrogen species including OH•, O2−•, and NO•.[47][48] In plants, melatonin works with other antioxidants to improve the overall effectiveness of each antioxidant.[48] Melatonin has been proven to be twice as active as vitamin E, believed to be the most effective lipophilic antioxidant.[49] Via signal transduction through melatonin receptors, melatonin promotes the expression of antioxidant enzymes such as superoxide dismutase, glutathione peroxidase, glutathione reductase, and catalase.[50][51]
Melatonin occurs at high concentrations within mitochondrial fluid which greatly exceed the plasma concentration of melatonin.[52][53][54] Due to its capacity for free radical scavenging, indirect effects on the expression of antioxidant enzymes, and its significant concentrations within mitochondria, a number of authors have indicated that melatonin has an important physiological function as a mitochondrial antioxidant.[50][52][53][54][55]
The melatonin metabolites produced via the reaction of melatonin with reactive oxygen species or reactive nitrogen species also react with and reduce free radicals.[51][55] Melatonin metabolites generated from redox reactions include cyclic 3-hydroxymelatonin, N1-acetyl-N2-formyl-5-methoxykynuramine (AFMK), and N1-acetyl-5-methoxykynuramine (AMK).[51][55]
Immune system
While it is known that melatonin interacts with the immune system,[56][57] the details of those interactions are unclear. An antiinflammatory effect seems to be the most relevant. There have been few trials designed to judge the effectiveness of melatonin in disease treatment. Most existing data are based on small, incomplete trials. Any positive immunological effect is thought to be the result of melatonin acting on high-affinity receptors (MT1 and MT2) expressed in immunocompetent cells. In preclinical studies, melatonin may enhance cytokine production,[58] and by doing this, counteract acquired immunodeficiences. Some studies also suggest that melatonin might be useful fighting infectious disease[59] including viral, such as HIV, and bacterial infections, and potentially in the treatment of cancer.
Biosynthesis

Overview of melatonin biosynthesis
In animals, biosynthesis of melatonin occurs through hydroxylation, decarboxylation, acetylation and a methylation starting with L-tryptophan.[60] L-tryptophan is produced in the shikimate pathway from chorismate or is acquired from protein catabolism. First L-tryptophan is hydroxylated on the indole ring by tryptophan hydroxylase to produce 5-hydroxytryptophan. This intermediate (5-HTP) is decarboxylated by pyridoxal phosphate and 5-hydroxytryptophan decarboxylase to produce serotonin.
Serotonin is itself an important neurotransmitter, but is also converted into N-acetylserotonin by serotonin N-acetyltransferase with acetyl-CoA.[61] Hydroxyindole O-methyltransferase and S-adenosyl methionine convert N-acetylserotonin into melatonin through methylation of the hydroxyl group.[61]
In bacteria, protists, fungi, and plants, melatonin is synthesized indirectly with tryptophan as an intermediate product of the shikimate pathway. In these cells, synthesis starts with D-erythrose 4-phosphate and phosphoenolpyruvate, and in photosynthetic cells with carbon dioxide. The rest of the synthesising reactions are similar, but with slight variations in the last two enzymes.[62][63]
It has been hypothesized that melatonin is made in the mitochondria and chloroplasts.[64]
Mechanism

Mechanism of melatonin biosynthesis
In order to hydroxylate L-tryptophan, the cofactor tetrahydrobiopterin (THB) must first react with oxygen and the active site iron of tryptophan hydroxylase. This mechanism is not well understood, but two mechanisms have been proposed:
1. A slow transfer of one electron from the THB to O2 could produce a superoxide which could recombine with the THB radical to give 4a-peroxypterin. 4a-peroxypterin could then react with the active site iron (II) to form an iron-peroxypterin intermediate or directly transfer an oxygen atom to the iron.
2. O2 could react with the active site iron (II) first, producing iron (III) superoxide which could then react with the THB to form an iron-peroxypterin intermediate.
Iron (IV) oxide from the iron-peroxypterin intermediate is selectively attacked by a double bond to give a carbocation at the C5 position of the indole ring. A 1,2-shift of the hydrogen and then a loss of one of the two hydrogen atoms on C5 reestablishes aromaticity to furnish 5-hydroxy-L-tryptophan.[65]
A decarboxylase with cofactor pyridoxal phosphate (PLP) removes CO2 from 5-hydroxy-L-tryptophan to produce 5-hydroxytryptamine.[66] PLP forms an imine with the amino acid derivative. The amine on the pyridine is protonated and acts as an electron sink, enabling the breaking of the C-C bond and releasing CO2. Protonation of the amine from tryptophan restores the aromaticity of the pyridine ring and then imine is hydrolyzed to produce 5-hydroxytryptamine and PLP.[67]
It has been proposed that histidine residue His122 of serotonin N-acetyl transferase is the catalytic residue that deprotonates the primary amine of 5-hydroxytryptamine, which allows the lone pair on the amine to attack acetyl-CoA, forming a tetrahedral intermediate. The thiol from coenzyme A serves as a good leaving group when attacked by a general base to give N-acetylserotonin.[68]
N-acetylserotonin is methylated at the hydroxyl position by S-adenosyl methionine (SAM) to produce S-adenosyl homocysteine (SAH) and melatonin.[67][69]
Regulation
In vertebrates, melatonin secretion is regulated by activation of the beta-1 adrenergic receptor by norepinephrine.[70] Norepinephrine elevates the intracellular cAMP concentration via beta-adrenergic receptors and activates the cAMP-dependent protein kinase A (PKA). PKA phosphorylates the penultimate enzyme, the arylalkylamine N-acetyltransferase (AANAT). On exposure to (day)light, noradrenergic stimulation stops and the protein is immediately destroyed by proteasomal proteolysis.[71] Production of melatonin is again started in the evening at the point called the dim-light melatonin onset.
Blue light, principally around 460–480 nm, suppresses melatonin biosynthesis,[72] proportional to the light intensity and length of exposure. Until recent history, humans in temperate climates were exposed to few hours of (blue) daylight in the winter; their fires gave predominantly yellow light.[citation needed] The incandescent light bulb widely used in the 20th century produced relatively little blue light.[73] Light containing only wavelengths greater than 530 nm does not suppress melatonin in bright-light conditions.[74] Wearing glasses that block blue light in the hours before bedtime may decrease melatonin loss. Use of blue-blocking goggles the last hours before bedtime has also been advised for people who need to adjust to an earlier bedtime, as melatonin promotes sleepiness.[75]
Pharmacology
Pharmacodynamics
In humans, melatonin is a full agonist of melatonin receptor 1 (picomolar binding affinity) and melatonin receptor 2 (nanomolar binding affinity), both of which belong to the class of G-protein coupled receptors (GPCRs).[51][76] Melatonin receptors 1 and 2 are both Gi/o-coupled GPCRs, although melatonin receptor 1 is also Gq-coupled.[51] Melatonin also acts as a high-capacity free radical scavenger within mitochondria which also promotes the expression of antioxidant enzymes such as superoxide dismutase, glutathione peroxidase, glutathione reductase, and catalase via signal transduction through melatonin receptors.[50][51][52][53][54][55]
Pharmacokinetics
When used several hours before sleep according to the phase response curve for melatonin in humans, small amounts (0.3 mg[77]) of melatonin shift the circadian clock earlier, thus promoting earlier sleep onset and morning awakening.[78] Melatonin is rapidly absorbed and distributed, reaching peak plasma concentrations after 60 minutes of administration, and is then eliminated.[61] Melatonin has a half life of 35–50 minutes.[79] In humans, 90% of orally administered exogenous melatonin is cleared in a single passage through the liver, a small amount is excreted in urine, and a small amount is found in saliva.[5] The bioavalibility of melatonin is between 10 and 50%.[61]
Melatonin is metabolized in the liver by cytochrome P450 enzyme CYP1A2 to 6-hydroxymelatonin. Metabolites are conjugated with sulfuric acid or glucuronic acid for excretion in the urine. 5% of melatonin is excreted in the urine as the unchanged drug.[61]
Some of the metabolites formed via the reaction of melatonin with a free radical include cyclic 3-hydroxymelatonin, N1-acetyl-N2-formyl-5-methoxykynuramine (AFMK), and N1-acetyl-5-methoxykynuramine (AMK).[51][55]
The membrane transport proteins that move melatonin across a membrane include, but are not limited to, glucose transporters, including GLUT1, and the proton-driven oligopeptide transporters PEPT1 and PEPT2.[51][55]
For research as well as clinical purposes, melatonin concentration in humans can be measured either from the saliva or blood plasma.[80]
History
Melatonin was first discovered in connection to the mechanism by which some amphibians and reptiles change the color of their skin.[81][82] As early as 1917, Carey Pratt McCord and Floyd P. Allen discovered that feeding extract of the pineal glands of cows lightened tadpole skin by contracting the dark epidermal melanophores.[83][84]
In 1958, dermatology professor Aaron B. Lerner and colleagues at Yale University, in the hope that a substance from the pineal might be useful in treating skin diseases, isolated the hormone from bovine pineal gland extracts and named it melatonin.[85] In the mid-70s Lynch et al. demonstrated that the production of melatonin exhibits a circadian rhythm in human pineal glands.[86]
The discovery that melatonin is an antioxidant was made in 1993.[87] The first patent for its use as a low-dose sleep aid was granted to Richard Wurtman at MIT in 1995.[88] Around the same time, the hormone got a lot of press as a possible treatment for many illnesses.[89] The New England Journal of Medicine editorialized in 2000: “With these recent careful and precise observations in blind persons, the true potential of melatonin is becoming evident, and the importance of the timing of treatment is becoming clear.”[90]
It was approved for medical use in the European Union in 2007.[11]
Other animals
In vertebrates, melatonin is produced in darkness, thus usually at night, by the pineal gland, a small endocrine gland[91] located in the center of the brain but outside the blood–brain barrier. Light/dark information reaches the suprachiasmatic nuclei from retinal photosensitive ganglion cells of the eyes[92][93] rather than the melatonin signal (as was once postulated). Known as “the hormone of darkness”, the onset of melatonin at dusk promotes activity in nocturnal (night-active) animals and sleep in diurnal ones including humans.
Many animals use the variation in duration of melatonin production each day as a seasonal clock.[94] In animals including humans,[95] the profile of melatonin synthesis and secretion is affected by the variable duration of night in summer as compared to winter. The change in duration of secretion thus serves as a biological signal for the organization of daylength-dependent (photoperiodic) seasonal functions such as reproduction, behavior, coat growth, and camouflage coloring in seasonal animals.[95] In seasonal breeders that do not have long gestation periods and that mate during longer daylight hours, the melatonin signal controls the seasonal variation in their sexual physiology, and similar physiological effects can be induced by exogenous melatonin in animals including mynah birds[96] and hamsters.[97] Melatonin can suppress libido by inhibiting secretion of luteinizing hormone and follicle-stimulating hormone from the anterior pituitary gland, especially in mammals that have a breeding season when daylight hours are long. The reproduction of long-day breeders is repressed by melatonin and the reproduction of short-day breeders is stimulated by melatonin.
During the night, melatonin regulates leptin, lowering its levels.
Cetaceans have lost all the genes for melatonin synthesis as well as those for melatonin receptors.[98] This is thought to be related to their unihemispheric sleep pattern (one brain hemisphere at a time). Similar trends have been found in sirenians.[98]
Plants
Until its identification in plants in 1987, melatonin was for decades thought to be primarily an animal neurohormone. When melatonin was identified in coffee extracts in the 1970s, it was believed to be a byproduct of the extraction process. Subsequently, however, melatonin has been found in all plants that have been investigated. It is present in all the different parts of plants, including leaves, stems, roots, fruits, and seeds, in varying proportions.[19][99] Melatonin concentrations differ not only among plant species, but also between varieties of the same species depending on the agronomic growing conditions, varying from picograms to several micrograms per gram.[63][100] Notably high melatonin concentrations have been measured in popular beverages such as coffee, tea, wine, and beer, and crops including corn, rice, wheat, barley, and oats.[19] In some common foods and beverages, including coffee[19] and walnuts,[101] the concentration of melatonin has been estimated or measured to be sufficiently high to raise the blood level of melatonin above daytime baseline values.
Although a role for melatonin as a plant hormone has not been clearly established, its involvement in processes such as growth and photosynthesis is well established. Only limited evidence of endogenous circadian rhythms in melatonin levels has been demonstrated in some plant species and no membrane-bound receptors analogous to those known in animals have been described. Rather, melatonin performs important roles in plants as a growth regulator, as well as environmental stress protector. It is synthesized in plants when they are exposed to both biological stresses, for example, fungal infection, and nonbiological stresses such as extremes of temperature, toxins, increased soil salinity, drought, etc.[63][102][103]
Occurrence
Dietary supplement
Melatonin is categorized by the US Food and Drug Administration (FDA) as a dietary supplement, and is sold over-the-counter in both the US and Canada.[5] FDA regulations applying to medications are not applicable to melatonin,[15] though the FDA has found false claims that it cures cancer.[104] As melatonin may cause harm in combination with certain medications or in the case of certain disorders, a doctor or pharmacist should be consulted before making a decision to take melatonin.[29] In many countries, melatonin is recognized as a neurohormone and it cannot be sold over-the-counter.[105]
Food products
Naturally-occurring melatonin has been reported in foods including tart cherries to about 0.17–13.46 ng/g,[106] bananas and grapes, rice and cereals, herbs, plums,[107] olive oil, wine[108] and beer. When birds ingest melatonin-rich plant feed, such as rice, the melatonin binds to melatonin receptors in their brains.[109] When humans consume foods rich in melatonin, such as banana, pineapple, and orange, the blood levels of melatonin increase significantly.[110]
Beverages and snacks containing melatonin were being sold in grocery stores, convenience stores, and clubs in May 2011.[111] The FDA considered whether these food products could continue to be sold with the label “dietary supplements”. On 13 January 2010, it issued a Warning Letter to Innovative Beverage, creators of several beverages marketed as drinks, stating that melatonin, while legal as a dietary supplement, was not approved as a food additive.[112] A different company selling a melatonin-containing beverage received a warning letter in 2015.[113]
Commercial availability
Immediate-release melatonin is not tightly regulated in countries where it is available as an over-the-counter medication. It is available in doses from less than half a milligram to 5 mg or more. Immediate-release formulations cause blood levels of melatonin to reach their peak in about an hour. The hormone may be administered orally, as capsules, gummies, tablets, or liquids. It is also available for use sublingually, or as transdermal patches.[medical citation needed]
Formerly, melatonin was derived from animal pineal tissue, such as bovine. It is now synthetic, which limits the risk of contamination or the means of transmitting infectious material.[15][114]
Melatonin is the most popular over-the-counter sleep remedy in the US, resulting in sales in excess of US$400 million during 2017.[115]
Research

A bottle of melatonin tablets. Melatonin is available in timed-release and in liquid forms.
Various uses and effects of melatonin have been studied. A 2015 review of studies of melatonin in tinnitus found the quality of evidence low, but not entirely without promise.[116]
Headaches
Tentative evidence shows melatonin may help reduce some types of headaches including cluster and hypnic headaches.[117][118]
Cancer
A 2013 review by the National Cancer Institutes found evidence for use to be inconclusive.[119] A 2005 review of unblinded clinical trials found a reduced rate of death, but that blinded and independently conducted randomized controlled trials are needed.[120]
Protection from radiation
Both animal[121] and human[122][123][124] studies have shown melatonin to protect against radiation-induced cellular damage. Melatonin and its metabolites protect organisms from oxidative stress by scavenging reactive oxygen species which are generated during exposure.[125] Nearly 70% of biological damage caused by ionizing radiation is estimated to be attributable to the creation of free radicals, especially the hydroxyl radical that attacks DNA, proteins, and cellular membranes. Melatonin has been described as a broadly protective, readily available, and orally self-administered antioxidant that is without known, major side effects.[126]
Epilepsy
A 2016 review found no beneficial role of melatonin in reducing seizure frequency or improving quality of life in people with epilepsy.[127]
Secondary dysmenorrhoea
A 2016 review suggested no strong evidence of melatonin compared to placebo for dysmenorrhoea secondary to endometriosis.[128]
Delirium
A 2016 review suggested no clear evidence of melatonin to reduce the incidence of delirium.[129]
Gastroesophageal reflux disease
A 2011 review said melatonin is effective in relieving epigastric pain and heartburn.[130]
Psychiatry
Melatonin might improve sleep in people with autism.[131] Children with autism have abnormal melatonin pathways and below-average physiological levels of melatonin.[132][133] Melatonin supplementation has been shown to improve sleep duration, sleep onset latency, and night-time awakenings.[132][134][135] However, many studies on melatonin and autism rely on self-reported levels of improvement and more rigorous research is needed.
While the packaging of melatonin often warns against use in people under 18 years of age, studies suggest that melatonin is an efficacious and safe treatment for insomnia in people with ADHD, including children. However, larger and longer studies are needed to establish long-term safety and optimal dosing.[136]
Melatonin in comparison to placebo is effective for reducing preoperative anxiety in adults when given as premedication. It may be just as effective as standard treatment with midazolam in reducing preoperative anxiety. Melatonin may also reduce postoperative anxiety (measured 6 hours after surgery) when compared to placebo.[137]
Some supplemental melatonin users report an increase in vivid dreaming. Extremely high doses of melatonin increased REM sleep time and dream activity in people both with and without narcolepsy.[138] Some evidence supports an antidepressant effect.[139]
References
- ^ “Melatonin – Drugs.com”. Drugs.com. Retrieved 12 October 2018.
- ^ “Melatonin”. Sleepdex. Retrieved 17 August 2011.
- ^ Jump up to:a b c d e f Auld F, Maschauer EL, Morrison I, Skene DJ, Riha RL (August 2017). “Evidence for the efficacy of melatonin in the treatment of primary adult sleep disorders” (PDF). Sleep Medicine Reviews. 34: 10–22. doi:10.1016/j.smrv.2016.06.005. PMID 28648359.
- ^ Faraone SV (2014). ADHD: Non-Pharmacologic Interventions, An Issue of Child and Adolescent Psychiatric Clinics of North America, E-Book. Elsevier Health Sciences. p. 888. ISBN 9780323326025.
- ^ Jump up to:a b c Buscemi N, Vandermeer B, Pandya R, Hooton N, Tjosvold L, Hartling L, et al. (November 2004). “Melatonin for treatment of sleep disorders” (PDF). Evidence Report/Technology Assessment No. 108. (Prepared by the University of Alberta Evidence-based Practice Center, Under Contract No. 290-02-0023.) AHRQ Publication No. 05-E002-2. Rockville, MD: Agency for Healthcare Research and Quality. Agency for Healthcare Research and Quality (AHRQ), US Department of Health and Human Services (108): 1–7. doi:10.1037/e439412005-001. PMC 4781368. PMID 15635761. Retrieved 5 June2013.
- ^ Jump up to:a b c d e f Matheson E, Hainer BL (July 2017). “Insomnia: Pharmacologic Therapy”. American Family Physician. 96 (1): 29–35. PMID 28671376.
- ^ Jump up to:a b c British national formulary : BNF 76 (76 ed.). Pharmaceutical Press. 2018. pp. 482–483. ISBN 9780857113382.
- ^ Jump up to:a b c Brasure M, MacDonald R, Fuchs E, Olson CM, Carlyle M, Diem S, et al. (2015). “Management of Insomnia Disorder[Internet]”. AHRQ Comparative Effectiveness Reviews. 15 (16): EHC027–EF. PMID 26844312.
Evidence for benzodiazepine hypnotics, melatonin agonists in the general adult population, and most pharmacologic interventions in older adults was generally insufficient
- ^ Adams, Katie S. (2014). “Melatonin agonists in the management of sleep disorders: A focus on ramelteon and tasimelteon”. Mental Health Clinician. 4 (2): 59–64. doi:10.9740/mhc.n190087. Retrieved 25 October 2020.
However, the clinical relevance of this objective and therefore the author’s conclusion that these results support the potential use of ramelteon in circadian rhythm sleep disorders is questionable. … It is unclear whether Takeda Pharmaceuticals will pursue FDA indications for ramelteon for circadian rhythm disorders given these results.
- ^ Jump up to:a b c d e “Melatonin: Side Effects, Uses, Dosage (Kids/Adults)”. Drugs.com. Retrieved 9 January 2019.
- ^ Jump up to:a b c d e “Circadin EPAR”. European Medicines Agency (EMA). Retrieved 31 May 2020.Text was copied from this source which is © European Medicines Agency. Reproduction is authorized provided the source is acknowledged.
- ^ Jump up to:a b c “Slenyto EPAR”. European Medicines Agency (EMA). Retrieved 31 May 2020. Text was copied from this source which is © European Medicines Agency. Reproduction is authorized provided the source is acknowledged.
- ^ Jump up to:a b “Summary Safety Review – MELATONIN (N-acetyl-5-methoxytryptamine) – Review of the Safety of Melatonin in Children and Adolescents”. Government of Canada. Health Canada. 10 December 2015. Retrieved 9 January 2019.
- ^ Jump up to:a b “Melatonin- Oral”. Government of Canada. Health Canada. 28 August 2018. Retrieved 9 January 2019.
- ^ Jump up to:a b c Altun A, Ugur-Altun B (May 2007). “Melatonin: therapeutic and clinical utilization”. International Journal of Clinical Practice. 61 (5): 835–45. doi:10.1111/j.1742-1241.2006.01191.x. PMID 17298593. S2CID 18050554.
- ^ Boutin JA, Audinot V, Ferry G, Delagrange P (August 2005). “Molecular tools to study melatonin pathways and actions”. Trends in Pharmacological Sciences. 26 (8): 412–9. doi:10.1016/j.tips.2005.06.006. PMID 15992934.
- ^ Hardeland R (July 2005). “Antioxidative protection by melatonin: multiplicity of mechanisms from radical detoxification to radical avoidance”. Endocrine. 27 (2): 119–30. doi:10.1385/ENDO:27:2:119. PMID 16217125.
- ^ Reiter RJ, Acuña-Castroviejo D, Tan DX, Burkhardt S (June 2001). “Free radical-mediated molecular damage. Mechanisms for the protective actions of melatonin in the central nervous system”. Annals of the New York Academy of Sciences. 939 (1): 200–15. Bibcode:2001NYASA.939..200R. doi:10.1111/j.1749-6632.2001.tb03627.x. PMID 11462772. S2CID 20404509.
- ^ Jump up to:a b c d Tan DX, Hardeland R, Manchester LC, Korkmaz A, Ma S, Rosales-Corral S, Reiter RJ (January 2012). “Functional roles of melatonin in plants, and perspectives in nutritional and agricultural science”. Journal of Experimental Botany. 63 (2): 577–97. doi:10.1093/jxb/err256. PMID 22016420.
- ^ “Australian Public Assessment Report for Melatonin” (PDF). Australian Government Department of Health and Ageing Therapeutic Goods Administration. January 2011. pp. 2, 4. Retrieved 9 January 2019.
Monotherapy for the short term treatment of primary insomnia characterised by poor quality of sleep in patients who are aged 55 or over.
- ^ McCarter SJ, Boswell CL, St Louis EK, Dueffert LG, Slocumb N, Boeve BF, et al. (March 2013). “Treatment outcomes in REM sleep behavior disorder”. Sleep Medicine (Review). 14 (3): 237–42. doi:10.1016/j.sleep.2012.09.018. PMC 3617579. PMID 23352028.
- ^ McKeith IG, Boeve BF, Dickson DW, Halliday G, Taylor JP, Weintraub D, et al. (July 2017). “Diagnosis and management of dementia with Lewy bodies: Fourth consensus report of the DLB Consortium”. Neurology (Review). 89 (1): 88–100. doi:10.1212/WNL.0000000000004058. PMC 5496518. PMID 28592453.
- ^ Boot BP (2015). “Comprehensive treatment of dementia with Lewy bodies”. Alzheimer’s Research & Therapy (Review). 7 (1): 45. doi:10.1186/s13195-015-0128-z. PMC 4448151. PMID 26029267.
- ^ “Circadin – Summary of Product Characteristics (SmPC) – (eMC)”. Electronic Medicines Compendium. 29 June 2007. Retrieved 1 March 2018.
- ^ Abdelgadir IS, Gordon MA, Akobeng AK (December 2018). “Melatonin for the management of sleep problems in children with neurodevelopmental disorders: a systematic review and meta-analysis” (PDF). Archives of Disease in Childhood. 103 (12): 1155–1162. doi:10.1136/archdischild-2017-314181. PMID 29720494. S2CID 19107081.
- ^ McCleery, Jenny; Sharpley, Ann L. (15 November 2020). “Pharmacotherapies for sleep disturbances in dementia”. The Cochrane Database of Systematic Reviews. 11: CD009178. doi:10.1002/14651858.CD009178.pub4. ISSN 1469-493X. PMID 33189083.
- ^ Spinedi E, Cardinali DP (2019). “Neuroendocrine-Metabolic Dysfunction and Sleep Disturbances in Neurodegenerative Disorders: Focus on Alzheimer’s Disease and Melatonin”. Neuroendocrinology. 108 (4): 354–364. doi:10.1159/000494889. PMID 30368508. S2CID 53101746.
- ^ Gao C, Scullin MK, Bliwise DL (2019). “Mild Cognitive Impairment and Dementia”. In Savard J, Ouellet MC (eds.). Handbook of Sleep Disorders in Medical Conditions. Academic Press. pp. 253–276. doi:10.1016/b978-0-12-813014-8.00011-1. ISBN 978-0-12-813014-8.
- ^ Jump up to:a b Herxheimer A, Petrie KJ (2002). “Melatonin for the prevention and treatment of jet lag”. The Cochrane Database of Systematic Reviews (2): CD001520. doi:10.1002/14651858.CD001520. PMID 12076414.
- ^ Jump up to:a b Liira J, Verbeek JH, Costa G, Driscoll TR, Sallinen M, Isotalo LK, Ruotsalainen JH (August 2014). “Pharmacological interventions for sleepiness and sleep disturbances caused by shift work”. The Cochrane Database of Systematic Reviews. 8 (8): CD009776. doi:10.1002/14651858.CD009776.pub2. PMID 25113164.
- ^ Buscemi N, Vandermeer B, Hooton N, Pandya R, Tjosvold L, Hartling L, et al. (December 2005). “The efficacy and safety of exogenous melatonin for primary sleep disorders. A meta-analysis”. Journal of General Internal Medicine. 20 (12): 1151–8. doi:10.1111/j.1525-1497.2005.0243.x. PMC 1490287. PMID 16423108.
- ^ Buscemi N, Vandermeer B, Hooton N, Pandya R, Tjosvold L, Hartling L, et al. (February 2006). “Efficacy and safety of exogenous melatonin for secondary sleep disorders and sleep disorders accompanying sleep restriction: meta-analysis”. BMJ. 332 (7538): 385–93. doi:10.1136/bmj.38731.532766.F6. PMC 1370968. PMID 16473858.
- ^ Lyseng-Williamson KA (November 2012). “Melatonin prolonged release: in the treatment of insomnia in patients aged ≥55 years”. Drugs & Aging. 29 (11): 911–23. doi:10.1007/s40266-012-0018-z. PMID 23044640. S2CID 1403262.
- ^ Database of Abstracts of Reviews of Effects (DARE): Quality-assessed Reviews [Internet]. York (UK): Centre for Reviews and Dissemination (UK); 1995. Optimal dosages for melatonin supplementation therapy in older adults: a systematic review of current literature. 2014.
- ^ Brent Bauer, M.D. “Melatonin side effects: What are the risks?”. Mayo Clinic. Retrieved 17 August 2011.
- ^ Zhdanova IV, Wurtman RJ, Regan MM, Taylor JA, Shi JP, Leclair OU (October 2001). “Melatonin treatment for age-related insomnia”. The Journal of Clinical Endocrinology and Metabolism. 86 (10): 4727–30. doi:10.1210/jc.86.10.4727. PMID 11600532.
- ^ Morera AL, Henry M, de La Varga M (2001). “[Safety in melatonin use]” [Safety in melatonin use]. Actas Espanolas de Psiquiatria (in Spanish). 29 (5): 334–7. PMID 11602091.
- ^ Terry PD, Villinger F, Bubenik GA, Sitaraman SV (January 2009). “Melatonin and ulcerative colitis: evidence, biological mechanisms, and future research”. Inflammatory Bowel Diseases. 15 (1): 134–40. doi:10.1002/ibd.20527. PMID 18626968.
- ^ Juszczak M, Michalska M (2006). “[The effect of melatonin on prolactin, luteinizing hormone (LH), and follicle-stimulating hormone (FSH) synthesis and secretion]” [The effect of melatonin on prolactin, luteinizing hormone (LH), and follicle-stimulating hormone (FSH) synthesis and secretion]. Postepy Higieny I Medycyny Doswiadczalnej (in Polish). 60: 431–8. PMID 16921343. Archived from the original on 2 November 2014. Retrieved 24 December 2013.
- ^ Srinivasan V, Spence WD, Pandi-Perumal SR, Zakharia R, Bhatnagar KP, Brzezinski A (December 2009). “Melatonin and human reproduction: shedding light on the darkness hormone”. Gynecological Endocrinology. 25 (12): 779–85. doi:10.3109/09513590903159649. PMID 19905996. S2CID 3442003.
- ^ Shane-McWhorter L. Melatonin. Merck Manual Professional Version. October 2018. Accessed 22 April 2019.
- ^ Emet M, Ozcan H, Ozel L, Yayla M, Halici Z, Hacimuftuoglu A (June 2016). “A Review of Melatonin, Its Receptors and Drugs”. The Eurasian Journal of Medicine. 48 (2): 135–41. doi:10.5152/eurasianjmed.2015.0267. PMC 4970552. PMID 27551178.
- ^ Ardura J, Gutierrez R, Andres J, Agapito T (2003). “Emergence and evolution of the circadian rhythm of melatonin in children”. Hormone Research. 59 (2): 66–72. doi:10.1159/000068571. PMID 12589109. S2CID 41937922.
- ^ Sack RL, Lewy AJ, Erb DL, Vollmer WM, Singer CM (1986). “Human melatonin production decreases with age”. Journal of Pineal Research. 3 (4): 379–88. doi:10.1111/j.1600-079X.1986.tb00760.x. PMID 3783419. S2CID 33664568.
- ^ Hagenauer MH, Perryman JI, Lee TM, Carskadon MA (June 2009). “Adolescent changes in the homeostatic and circadian regulation of sleep”. Developmental Neuroscience. 31(4): 276–84. doi:10.1159/000216538. PMC 2820578. PMID 19546564.
- ^ Tan DX, Chen LD, Poeggeler B, L Manchester C, Reiter RJ. Melatonin: a potent, endogenous hydroxyl radical scavenger. Endocr. J. 1993, 1, 57–60 https://docs.google.com/viewer?a=v&pid=sites&srcid=ZGVmYXVsdGRvbWFpbnxkdW54aWFudGFufGd4OjVkMjA5NGZkMzFmYjRkOTU
- ^ Poeggeler B, Saarela S, Reiter RJ, Tan DX, Chen LD, Manchester LC, Barlow-Walden LR (November 1994). “Melatonin—a highly potent endogenous radical scavenger and electron donor: new aspects of the oxidation chemistry of this indole accessed in vitro”. Annals of the New York Academy of Sciences. 738 (1): 419–20. Bibcode:1994NYASA.738..419P. doi:10.1111/j.1749-6632.1994.tb21831.x. PMID 7832450. S2CID 36383425.
- ^ Jump up to:a b Arnao MB, Hernández-Ruiz J (May 2006). “The physiological function of melatonin in plants”. Plant Signaling & Behavior. 1 (3): 89–95. doi:10.4161/psb.1.3.2640. PMC 2635004. PMID 19521488.
- ^ Pieri C, Marra M, Moroni F, Recchioni R, Marcheselli F (1994). “Melatonin: a peroxyl radical scavenger more effective than vitamin E”. Life Sciences. 55 (15): PL271-6. doi:10.1016/0024-3205(94)00666-0. PMID 7934611.
- ^ Jump up to:a b c Sharafati-Chaleshtori R, Shirzad H, Rafieian-Kopaei M, Soltani A (2017). “Melatonin and human mitochondrial diseases”. Journal of Research in Medical Sciences. 22: 2. doi:10.4103/1735-1995.199092. PMC 5361446. PMID 28400824.
- ^ Jump up to:a b c d e f g h Jockers R, Delagrange P, Dubocovich ML, Markus RP, Renault N, Tosini G, et al. (September 2016). “Update on melatonin receptors: IUPHAR Review 20”. British Journal of Pharmacology. 173 (18): 2702–25. doi:10.1111/bph.13536. PMC 4995287. PMID 27314810.
Hence, one melatonin molecule and its associated metabolites could scavenge a large number of reactive species, and thus, the overall antioxidant capacity of melatonin is believed to be greater than that of other well‐known antioxidants, such as vitamin C and vitamin E, under in vitro or in vivo conditions (Gitto et al., 2001; Sharma and Haldar, 2006; Ortiz et al., 2013).
- ^ Jump up to:a b c Reiter RJ, Rosales-Corral S, Tan DX, Jou MJ, Galano A, Xu B (November 2017). “Melatonin as a mitochondria-targeted antioxidant: one of evolution’s best ideas”. Cellular and Molecular Life Sciences. 74 (21): 3863–3881. doi:10.1007/s00018-017-2609-7. PMID 28864909. S2CID 23820389.
melatonin is specifically targeted to the mitochondria where it seems to function as an apex antioxidant … The measurement of the subcellular distribution of melatonin has shown that the concentration of this indole in the mitochondria greatly exceeds that in the blood.
- ^ Jump up to:a b c Reiter RJ, Mayo JC, Tan DX, Sainz RM, Alatorre-Jimenez M, Qin L (October 2016). “Melatonin as an antioxidant: under promises but over delivers”. Journal of Pineal Research. 61 (3): 253–78. doi:10.1111/jpi.12360. PMID 27500468. S2CID 35435683.
There is credible evidence to suggest that melatonin should be classified as a mitochondria-targeted antioxidant.
- ^ Jump up to:a b c Manchester LC, Coto-Montes A, Boga JA, Andersen LP, Zhou Z, Galano A, et al. (November 2015). “Melatonin: an ancient molecule that makes oxygen metabolically tolerable”. Journal of Pineal Research. 59 (4): 403–19. doi:10.1111/jpi.12267. PMID 26272235. S2CID 24373303.
While originally thought to be produced exclusively in and secreted from the vertebrate pineal gland [53], it is now known that the indole is present in many, perhaps all, vertebrate organs [54] and in organs of all plants that have been investigated [48, 55, 56]. That melatonin is not relegated solely to the pineal gland is also emphasized by the reports that it is present in invertebrates [57–59], which lack a pineal gland and some of which consist of only a single cell.
- ^ Jump up to:a b c d e f Mayo JC, Sainz RM, González-Menéndez P, Hevia D, Cernuda-Cernuda R (November 2017). “Melatonin transport into mitochondria”. Cellular and Molecular Life Sciences. 74 (21): 3927–3940. doi:10.1007/s00018-017-2616-8. PMID 28828619. S2CID 10920415.
- ^ Carrillo-Vico A, Guerrero JM, Lardone PJ, Reiter RJ (July 2005). “A review of the multiple actions of melatonin on the immune system”. Endocrine. 27 (2): 189–200. doi:10.1385/ENDO:27:2:189. PMID 16217132. S2CID 21133107.
- ^ Arushanian EB, Beĭer EV (2002). “[Immunotropic properties of pineal melatonin]”. Eksperimental’naia i Klinicheskaia Farmakologiia (in Russian). 65 (5): 73–80. PMID 12596522.
- ^ Carrillo-Vico A, Reiter RJ, Lardone PJ, Herrera JL, Fernández-Montesinos R, Guerrero JM, Pozo D (May 2006). “The modulatory role of melatonin on immune responsiveness”. Current Opinion in Investigational Drugs. 7 (5): 423–31. PMID 16729718.
- ^ Maestroni GJ (March 2001). “The immunotherapeutic potential of melatonin”. Expert Opinion on Investigational Drugs. 10 (3): 467–76. doi:10.1517/13543784.10.3.467. PMID 11227046. S2CID 6822594.
- ^ “MetaCyc serotonin and melatonin biosynthesis”.
- ^ Jump up to:a b c d e Tordjman S, Chokron S, Delorme R, Charrier A, Bellissant E, Jaafari N, Fougerou C (April 2017). “Melatonin: Pharmacology, Functions and Therapeutic Benefits”. Current Neuropharmacology. 15 (3): 434–443. doi:10.2174/1570159X14666161228122115. PMC 5405617. PMID 28503116.
- ^ Bochkov DV, Sysolyatin SV, Kalashnikov AI, Surmacheva IA (January 2012). “Shikimic acid: review of its analytical, isolation, and purification techniques from plant and microbial sources”. Journal of Chemical Biology. 5 (1): 5–17. doi:10.1007/s12154-011-0064-8. PMC 3251648. PMID 22826715.
- ^ Jump up to:a b c Hardeland R (February 2015). “Melatonin in plants and other phototrophs: advances and gaps concerning the diversity of functions”. Journal of Experimental Botany. 66 (3): 627–46. doi:10.1093/jxb/eru386. PMID 25240067.
- ^ Tan DX, Manchester LC, Liu X, Rosales-Corral SA, Acuna-Castroviejo D, Reiter RJ (March 2013). “Mitochondria and chloroplasts as the original sites of melatonin synthesis: a hypothesis related to melatonin’s primary function and evolution in eukaryotes”. Journal of Pineal Research. 54 (2): 127–38. doi:10.1111/jpi.12026. PMID 23137057. S2CID 206140413.
- ^ Roberts KM, Fitzpatrick PF (April 2013). “Mechanisms of tryptophan and tyrosine hydroxylase”. IUBMB Life. 65 (4): 350–7. doi:10.1002/iub.1144. PMC 4270200. PMID 23441081.
- ^ Sumi-Ichinose C, Ichinose H, Takahashi E, Hori T, Nagatsu T (March 1992). “Molecular cloning of genomic DNA and chromosomal assignment of the gene for human aromatic L-amino acid decarboxylase, the enzyme for catecholamine and serotonin biosynthesis”. Biochemistry. 31 (8): 2229–38. doi:10.1021/bi00123a004. PMID 1540578.
- ^ Jump up to:a b Dewick PM (2002). Medicinal Natural Products. A Biosynthetic Approach (2nd ed.). Wiley. ISBN 978-0-471-49640-3.
- ^ Hickman AB, Klein DC, Dyda F (January 1999). “Melatonin biosynthesis: the structure of serotonin N-acetyltransferase at 2.5 A resolution suggests a catalytic mechanism”. Molecular Cell. 3 (1): 23–32. doi:10.1016/S1097-2765(00)80171-9. PMID 10024876.
- ^ Donohue SJ, Roseboom PH, Illnerova H, Weller JL, Klein DC (October 1993). “Human hydroxyindole-O-methyltransferase: presence of LINE-1 fragment in a cDNA clone and pineal mRNA”. DNA and Cell Biology. 12 (8): 715–27. doi:10.1089/dna.1993.12.715. PMID 8397829.
- ^ Nesbitt AD, Leschziner GD, Peatfield RC (September 2014). “Headache, drugs and sleep”. Cephalalgia (Review). 34 (10): 756–66. doi:10.1177/0333102414542662. PMID 25053748. S2CID 33548757.
- ^ Schomerus C, Korf HW (December 2005). “Mechanisms regulating melatonin synthesis in the mammalian pineal organ”. Annals of the New York Academy of Sciences. 1057 (1): 372–83. Bibcode:2005NYASA1057..372S. doi:10.1196/annals.1356.028. PMID 16399907. S2CID 20517556.
- ^ Brainard GC, Hanifin JP, Greeson JM, Byrne B, Glickman G, Gerner E, Rollag MD (August 2001). “Action spectrum for melatonin regulation in humans: evidence for a novel circadian photoreceptor”. The Journal of Neuroscience. 21 (16): 6405–12. doi:10.1523/JNEUROSCI.21-16-06405.2001. PMC 6763155. PMID 11487664.
- ^ “Recent News – Program of Computer Graphics”. http://www.graphics.cornell.edu.
- ^ Kayumov L, Casper RF, Hawa RJ, Perelman B, Chung SA, Sokalsky S, Shapiro CM (May 2005). “Blocking low-wavelength light prevents nocturnal melatonin suppression with no adverse effect on performance during simulated shift work”. The Journal of Clinical Endocrinology and Metabolism. 90 (5): 2755–61. doi:10.1210/jc.2004-2062. PMID 15713707.
- ^ Burkhart K, Phelps JR (December 2009). “Amber lenses to block blue light and improve sleep: a randomized trial”. Chronobiology International. 26 (8): 1602–12. doi:10.3109/07420520903523719. PMID 20030543.
- ^ “Melatonin receptors | G protein-coupled receptors | IUPHAR/BPS Guide to Pharmacology”. http://www.guidetopharmacology.org. Retrieved 7 April 2017.
- ^ Mundey K, Benloucif S, Harsanyi K, Dubocovich ML, Zee PC (October 2005). “Phase-dependent treatment of delayed sleep phase syndrome with melatonin”. Sleep. 28 (10): 1271–8. doi:10.1093/sleep/28.10.1271. PMID 16295212.
- ^ Terman MR, Wirz-Justice A (2009). Chronotherapeutics for Affective Disorders: A Clinician’s Manual for Light and Wake Therapy. Basel: S Karger Pub. p. 71. ISBN 978-3-8055-9120-1.
- ^ “Melatonin”. http://www.drugbank.ca. Retrieved 29 January 2019.
- ^ Kennaway DJ (August 2019). “A critical review of melatonin assays: Past and present”. Journal of Pineal Research. 67 (1): e12572. doi:10.1111/jpi.12572. PMID 30919486.
- ^ Filadelfi AM, Castrucci AM (May 1996). “Comparative aspects of the pineal/melatonin system of poikilothermic vertebrates”. Journal of Pineal Research. 20 (4): 175–86. doi:10.1111/j.1600-079X.1996.tb00256.x. PMID 8836950. S2CID 41959214.
- ^ Sugden D, Davidson K, Hough KA, Teh MT (October 2004). “Melatonin, melatonin receptors and melanophores: a moving story”. Pigment Cell Research. 17 (5): 454–60. doi:10.1111/j.1600-0749.2004.00185.x. PMID 15357831.
- ^ Coates PM, Blackman MR, Cragg GM, Levine M, Moss J, White JD (2005). Encyclopedia of dietary supplements. New York, N.Y: Marcel Dekker. pp. 457–66. ISBN 978-0-8247-5504-1.
- ^ McCord CP, Allen FP (January 1917). “Evidences associating pineal gland function with alterations in pigmentation”. J Exp Zool. 23 (1): 206–24. doi:10.1002/jez.1400230108.
- ^ Lerner AB, Case JD, Takahashi Y (July 1960). “Isolation of melatonin and 5-methoxyindole-3-acetic acid from bovine pineal glands”. The Journal of Biological Chemistry. 235: 1992–7. PMID 14415935.
- ^ Lynch HJ, Wurtman RJ, Moskowitz MA, Archer MC, Ho MH (January 1975). “Daily rhythm in human urinary melatonin”. Science. 187 (4172): 169–71. Bibcode:1975Sci…187..169L. doi:10.1126/science.1167425. PMID 1167425.
- ^ Poeggeler B, Reiter RJ, Tan DX, Chen LD, Manchester LC (May 1993). “Melatonin, hydroxyl radical-mediated oxidative damage, and aging: a hypothesis”. Journal of Pineal Research. 14 (4): 151–68. doi:10.1111/j.1600-079X.1993.tb00498.x. PMID 8102180. S2CID 23460208.
- ^ US patent 5449683, Wurtman RJ, “Methods of inducing sleep using melatonin”, issued 12 September 1995, assigned to Massachusetts Institute of Technology
- ^ Arendt J (August 2005). “Melatonin: characteristics, concerns, and prospects”. Journal of Biological Rhythms. 20 (4): 291–303. doi:10.1177/0748730405277492. PMID 16077149. S2CID 19011222.
There is very little evidence in the short term for toxicity or undesirable effects in humans. The extensive promotion of the miraculous powers of melatonin in the recent past did a disservice to acceptance of its genuine benefits.
- ^ Arendt J (October 2000). “Melatonin, circadian rhythms, and sleep”. The New England Journal of Medicine. 343 (15): 1114–6. doi:10.1056/NEJM200010123431510. PMID 11027748.
- ^ Reiter RJ (May 1991). “Pineal melatonin: cell biology of its synthesis and of its physiological interactions”. Endocrine Reviews. 12 (2): 151–80. doi:10.1210/edrv-12-2-151. PMID 1649044. S2CID 3219721.
- ^ Richardson GS (2005). “The human circadian system in normal and disordered sleep”. The Journal of Clinical Psychiatry. 66 Suppl 9: 3–9, quiz 42–3. PMID 16336035.
- ^ Perreau-Lenz S, Pévet P, Buijs RM, Kalsbeek A (January 2004). “The biological clock: the bodyguard of temporal homeostasis”. Chronobiology International. 21 (1): 1–25. doi:10.1081/CBI-120027984. PMID 15129821. S2CID 42725506.
- ^ Lincoln GA, Andersson H, Loudon A (October 2003). “Clock genes in calendar cells as the basis of annual timekeeping in mammals—a unifying hypothesis”. The Journal of Endocrinology. 179 (1): 1–13. doi:10.1677/joe.0.1790001. PMID 14529560.
- ^ Jump up to:a b Arendt J, Skene DJ (February 2005). “Melatonin as a chronobiotic”. Sleep Medicine Reviews. 9 (1): 25–39. doi:10.1016/j.smrv.2004.05.002. PMID 15649736.
Exogenous melatonin has acute sleepiness-inducing and temperature-lowering effects during ‘biological daytime’, and when suitably timed (it is most effective around dusk and dawn), it will shift the phase of the human circadian clock (sleep, endogenous melatonin, core body temperature, cortisol) to earlier (advance phase shift) or later (delay phase shift) times.
- ^ Chaturvedi CM (1984). “Effect of Melatonin on the Adrenl and Gonad of the Common Mynah Acridtheres tristis”. Australian Journal of Zoology. 32 (6): 803–09. doi:10.1071/ZO9840803.
- ^ Chen HJ (July 1981). “Spontaneous and melatonin-induced testicular regression in male golden hamsters: augmented sensitivity of the old male to melatonin inhibition”. Neuroendocrinology. 33 (1): 43–6. doi:10.1159/000123198. PMID 7254478.
- ^ Jump up to:a b Huelsmann M, Hecker N, Springer MS, Gatesy J, Sharma V, Hiller M (September 2019). “Genes lost during the transition from land to water in cetaceans highlight genomic changes associated with aquatic adaptations”. Science Advances. 5 (9): eaaw6671. Bibcode:2019SciA….5.6671H. doi:10.1126/sciadv.aaw6671. PMC 6760925. PMID 31579821.
- ^ Paredes SD, Korkmaz A, Manchester LC, Tan DX, Reiter RJ (1 January 2009). “Phytomelatonin: a review”. Journal of Experimental Botany. 60 (1): 57–69. doi:10.1093/jxb/ern284. PMID 19033551. S2CID 15738948.
- ^ Bonnefont-Rousselot D, Collin F (November 2010). “Melatonin: action as antioxidant and potential applications in human disease and aging”. Toxicology. 278 (1): 55–67. doi:10.1016/j.tox.2010.04.008. PMID 20417677.
- ^ Reiter RJ, Manchester LC, Tan DX (September 2005). “Melatonin in walnuts: influence on levels of melatonin and total antioxidant capacity of blood”. Nutrition. 21 (9): 920–4. doi:10.1016/j.nut.2005.02.005. PMID 15979282.
- ^ Reiter RJ, Tan DX, Zhou Z, Cruz MH, Fuentes-Broto L, Galano A (April 2015). “Phytomelatonin: assisting plants to survive and thrive”. Molecules. 20 (4): 7396–437. doi:10.3390/molecules20047396. PMC 6272735. PMID 25911967.
- ^ Arnao MB, Hernández-Ruiz J (September 2015). “Functions of melatonin in plants: a review”. Journal of Pineal Research. 59 (2): 133–50. doi:10.1111/jpi.12253. PMID 26094813.
- ^ “187 Fake Cancer ‘Cures’ Consumers Should Avoid”. U.S. Food and Drug Administration. Archived from the original on 2 May 2017. Retrieved 20 May 2020.
- ^ Guardiola-Lemaître B (December 1997). “Toxicology of melatonin”. Journal of Biological Rhythms. 12 (6): 697–706. doi:10.1177/074873049701200627. PMID 9406047. S2CID 31090576.
- ^ Burkhardt S, Tan DX, Manchester LC, Hardeland R, Reiter RJ (October 2001). “Detection and quantification of the antioxidant melatonin in Montmorency and Balaton tart cherries (Prunus cerasus)”. Journal of Agricultural and Food Chemistry. 49 (10): 4898–902. doi:10.1021/jf010321. PMID 11600041.
- ^ González-Flores D, Velardo B, Garrido M, González-Gómez D, Lozano M, Ayuso MC, Barriga C, Paredes SD, Rodríguez AB (2011). “Ingestion of Japanese plums (Prunus salicina Lindl. cv. Crimson Globe) increases the urinary 6-sulfatoxymelatonin and total antioxidant capacity levels in young, middle-aged and elderly humans: Nutritional and functional characterization of their content”. Journal of Food and Nutrition Research. 50(4): 229–36.
- ^ Lamont KT, Somers S, Lacerda L, Opie LH, Lecour S (May 2011). “Is red wine a SAFE sip away from cardioprotection? Mechanisms involved in resveratrol- and melatonin-induced cardioprotection”. Journal of Pineal Research. 50 (4): 374–80. doi:10.1111/j.1600-079X.2010.00853.x. PMID 21342247. S2CID 8034935.
- ^ Hattori A, Migitaka H, Iigo M, Itoh M, Yamamoto K, Ohtani-Kaneko R, et al. (March 1995). “Identification of melatonin in plants and its effects on plasma melatonin levels and binding to melatonin receptors in vertebrates”. Biochemistry and Molecular Biology International. 35(3): 627–34. PMID 7773197.
- ^ Sae-Teaw M, Johns J, Johns NP, Subongkot S (August 2013). “Serum melatonin levels and antioxidant capacities after consumption of pineapple, orange, or banana by healthy male volunteers”. Journal of Pineal Research. 55 (1): 58–64. doi:10.1111/jpi.12025. PMID 23137025. S2CID 979886.
- ^ Catherine Saint Louis (14 May 2011). “Dessert, Laid-Back and Legal”. The New York Times.
- ^ Rodriguez RR (13 January 2010). “Warning Letter”. Inspections, Compliance, Enforcement, and Criminal Investigations. U.S. Food and Drug Administration. Archived from the original on 12 January 2017.
- ^ Bebida Beverage Company U.S. Food and Drug Administration 4 March 2015 (Accessed 8 December 2017)
- ^ “Melatonin”. Drugs.com. Retrieved 17 August 2011.
- ^ Loria K. “Does Melatonin Really Help You Sleep?”. Consumer Reports. Retrieved 16 March 2019.
- ^ Miroddi M, Bruno R, Galletti F, Calapai F, Navarra M, Gangemi S, Calapai G (March 2015). “Clinical pharmacology of melatonin in the treatment of tinnitus: a review”. European Journal of Clinical Pharmacology. 71 (3): 263–70. doi:10.1007/s00228-015-1805-3. PMID 25597877. S2CID 16466238.
- ^ Peres MF, Masruha MR, Zukerman E, Moreira-Filho CA, Cavalheiro EA (April 2006). “Potential therapeutic use of melatonin in migraine and other headache disorders”. Expert Opinion on Investigational Drugs. 15 (4): 367–75. doi:10.1517/13543784.15.4.367. PMID 16548786. S2CID 28114683.
- ^ Evars S, Goadsby P (2005). “Review: Hypnic headache” (PDF). Practical Neurology. 5(3): 144–49. doi:10.1111/j.1474-7766.2005.00301.x. S2CID 1196313. Retrieved 12 January 2018.
- ^ Pdq Integrative, Alternative (May 2013). “Topics in complementary and alternative therapies (PDQ)”. PDQ Cancer Information Summaries [Internet]. National Cancer Institute, National Institutes of Health. PMID 26389506.
- ^ Mills E, Wu P, Seely D, Guyatt G (November 2005). “Melatonin in the treatment of cancer: a systematic review of randomized controlled trials and meta-analysis”. Journal of Pineal Research. 39 (4): 360–6. doi:10.1111/j.1600-079X.2005.00258.x. PMID 16207291. S2CID 22225091.
- ^ Meltz ML, Reiter RJ, Herman TS, Kumar KS (March 1999). “Melatonin and protection from whole-body irradiation: survival studies in mice”. Mutation Research. 425 (1): 21–7. doi:10.1016/S0027-5107(98)00246-2. PMID 10082913.
- ^ Reiter RJ, Herman TS, Meltz ML (December 1996). “Melatonin and radioprotection from genetic damage: in vivo/in vitro studies with human volunteers”. Mutation Research. 371 (3–4): 221–8. doi:10.1016/S0165-1218(96)90110-X. PMID 9008723.
- ^ Pattanittum P, Kunyanone N, Brown J, Sangkomkamhang US, Barnes J, Seyfoddin V, Marjoribanks J (March 2016). “Dietary supplements for dysmenorrhoea”. The Cochrane Database of Systematic Reviews. 3 (3): CD002124. doi:10.1002/14651858.cd002124.pub2. PMC 7387104. PMID 27000311.
- ^ Reiter RJ, Herman TS, Meltz ML (February 1998). “Melatonin reduces gamma radiation-induced primary DNA damage in human blood lymphocytes”. Mutation Research. 397 (2): 203–8. doi:10.1016/S0027-5107(97)00211-X. PMID 9541644.
- ^ Tan DX, Manchester LC, Terron MP, Flores LJ, Reiter RJ (January 2007). “One molecule, many derivatives: a never-ending interaction of melatonin with reactive oxygen and nitrogen species?”. Journal of Pineal Research. 42 (1): 28–42. doi:10.1111/j.1600-079X.2006.00407.x. PMID 17198536. S2CID 40005308.
- ^ Shirazi A, Ghobadi G, Ghazi-Khansari M (July 2007). “A radiobiological review on melatonin: a novel radioprotector”. Journal of Radiation Research. 48 (4): 263–72. Bibcode:2007JRadR..48..263S. doi:10.1269/jrr.06070. PMID 17641465.
- ^ Brigo F, Igwe SC, Del Felice A (August 2016). “Melatonin as add-on treatment for epilepsy”. The Cochrane Database of Systematic Reviews (8): CD006967. doi:10.1002/14651858.CD006967.pub4. PMC 7386917. PMID 27513702.
- ^ Pattanittum P, Kunyanone N, Brown J, Sangkomkamhang US, Barnes J, Seyfoddin V, Marjoribanks J (March 2016). “Dietary supplements for dysmenorrhoea”. The Cochrane Database of Systematic Reviews. 3: CD002124. doi:10.1002/14651858.CD002124.pub2. PMC 7387104. PMID 27000311.
- ^ Siddiqi N, Harrison JK, Clegg A, Teale EA, Young J, Taylor J, Simpkins SA (March 2016). “Interventions for preventing delirium in hospitalised non-ICU patients” (PDF). The Cochrane Database of Systematic Reviews. 3: CD005563. doi:10.1002/14651858.CD005563.pub3. PMID 26967259.
- ^ Kandil TS, Mousa AA, El-Gendy AA, Abbas AM (January 2010). “The potential therapeutic effect of melatonin in Gastro-Esophageal Reflux Disease”. BMC Gastroenterology. 10: 7. doi:10.1186/1471-230X-10-7. PMC 2821302. PMID 20082715.
- ^ Braam W, Smits MG, Didden R, Korzilius H, Van Geijlswijk IM, Curfs LM (May 2009). “Exogenous melatonin for sleep problems in individuals with intellectual disability: a meta-analysis”. Developmental Medicine and Child Neurology (Meta-analysis). 51 (5): 340–9. doi:10.1111/j.1469-8749.2008.03244.x. PMID 19379289. S2CID 35553148.
- ^ Jump up to:a b Rossignol DA, Frye RE (September 2011). “Melatonin in autism spectrum disorders: a systematic review and meta-analysis”. Developmental Medicine and Child Neurology (Meta-analysis). 53 (9): 783–792. doi:10.1111/j.1469-8749.2011.03980.x. PMID 21518346. S2CID 9951986.
- ^ Veatch OJ, Pendergast JS, Allen MJ, Leu RM, Johnson CH, Elsea SH, Malow BA (January 2015). “Genetic variation in melatonin pathway enzymes in children with autism spectrum disorder and comorbid sleep onset delay”. Journal of Autism and Developmental Disorders. 45 (1): 100–10. doi:10.1007/s10803-014-2197-4. PMC 4289108. PMID 25059483.
- ^ “Melatonin improves sleep quality and behavior in children with asperger disorder”. Truthly. Retrieved 15 February 2015.
- ^ Giannotti F, Cortesi F, Cerquiglini A, Bernabei P (August 2006). “An open-label study of controlled-release melatonin in treatment of sleep disorders in children with autism”. Journal of Autism and Developmental Disorders. 36 (6): 741–52. doi:10.1007/s10803-006-0116-z. PMID 16897403. S2CID 19724241.
- ^ Bendz LM, Scates AC (January 2010). “Melatonin treatment for insomnia in pediatric patients with attention-deficit/hyperactivity disorder”. The Annals of Pharmacotherapy. 44(1): 185–91. doi:10.1345/aph.1M365. PMID 20028959. S2CID 207263711.
- ^ Hansen MV, Halladin NL, Rosenberg J, Gögenur I, Møller AM (April 2015). “Melatonin for pre- and postoperative anxiety in adults”. The Cochrane Database of Systematic Reviews(4): CD009861. doi:10.1002/14651858.CD009861.pub2. PMC 6464333. PMID 25856551.
- ^ Lewis A (1999). Melatonin and the Biological Clock. McGraw-Hill. p. 23. ISBN 978-0-87983-734-1.
- ^ Valdés-Tovar M, Estrada-Reyes R, Solís-Chagoyán H, Argueta J, Dorantes-Barrón AM, Quero-Chávez D, et al. (August 2018). “Circadian modulation of neuroplasticity by melatonin: a target in the treatment of depression”. British Journal of Pharmacology. 175(16): 3200–3208. doi:10.1111/bph.14197. PMC 6057892. PMID 29512136.
External links "Melatonin". Drug Information Portal. U.S. National Library of Medicine.
//////////Melatonin, Melatobel, メラトニン , JAPAN 2020, 2020 APPROVALS
Isatuximab
(A chain)
QVQLVQSGAE VAKPGTSVKL SCKASGYTFT DYWMQWVKQR PGQGLEWIGT IYPGDGDTGY
AQKFQGKATL TADKSSKTVY MHLSSLASED SAVYYCARGD YYGSNSLDYW GQGTSVTVSS
ASTKGPSVFP LAPSSKSTSG GTAALGCLVK DYFPEPVTVS WNSGALTSGV HTFPAVLQSS
GLYSLSSVVT VPSSSLGTQT YICNVNHKPS NTKVDKKVEP KSCDKTHTCP PCPAPELLGG
PSVFLFPPKP KDTLMISRTP EVTCVVVDVS HEDPEVKFNW YVDGVEVHNA KTKPREEQYN
STYRVVSVLT VLHQDWLNGK EYKCKVSNKA LPAPIEKTIS KAKGQPREPQ VYTLPPSRDE
LTKNQVSLTC LVKGFYPSDI AVEWESNGQP ENNYKTTPPV LDSDGSFFLY SKLTVDKSRW
QQGNVFSCSV MHEALHNHYT QKSLSLSPGK
(B chain)
QVQLVQSGAE VAKPGTSVKL SCKASGYTFT DYWMQWVKQR PGQGLEWIGT IYPGDGDTGY
AQKFQGKATL TADKSSKTVY MHLSSLASED SAVYYCARGD YYGSNSLDYW GQGTSVTVSS
ASTKGPSVFP LAPSSKSTSG GTAALGCLVK DYFPEPVTVS WNSGALTSGV HTFPAVLQSS
GLYSLSSVVT VPSSSLGTQT YICNVNHKPS NTKVDKKVEP KSCDKTHTCP PCPAPELLGG
PSVFLFPPKP KDTLMISRTP EVTCVVVDVS HEDPEVKFNW YVDGVEVHNA KTKPREEQYN
STYRVVSVLT VLHQDWLNGK EYKCKVSNKA LPAPIEKTIS KAKGQPREPQ VYTLPPSRDE
LTKNQVSLTC LVKGFYPSDI AVEWESNGQP ENNYKTTPPV LDSDGSFFLY SKLTVDKSRW
QQGNVFSCSV MHEALHNHYT QKSLSLSPGK
(C chain)
DIVMTQSHLS MSTSLGDPVS ITCKASQDVS TVVAWYQQKP GQSPRRLIYS ASYRYIGVPD
RFTGSGAGTD FTFTISSVQA EDLAVYYCQQ HYSPPYTFGG GTKLEIKRTV AAPSVFIFPP
SDEQLKSGTA SVVCLLNNFY PREAKVQWKV DNALQSGNSQ ESVTEQDSKD STYSLSSTLT
LSKADYEKHK VYACEVTHQG LSSPVTKSFN RGEC
(D chain)
DIVMTQSHLS MSTSLGDPVS ITCKASQDVS TVVAWYQQKP GQSPRRLIYS ASYRYIGVPD
RFTGSGAGTD FTFTISSVQA EDLAVYYCQQ HYSPPYTFGG GTKLEIKRTV AAPSVFIFPP
SDEQLKSGTA SVVCLLNNFY PREAKVQWKV DNALQSGNSQ ESVTEQDSKD STYSLSSTLT
LSKADYEKHK VYACEVTHQG LSSPVTKSFN RGEC
(Disulfide bridge: A22-A96, A147-A203, A223-C214, A229-B229, A232-B232, A264-A324, A370-A428, B22-B96, B147-B203, B223-D214, B264-B324, B370-B428, C23-C88, C134-C194, D23-D88, D134-D194)
Isatuximab
イサツキシマブ (遺伝子組換え)
APPROVED USFDA 2020/3/2, Sarclisa
EU APPROVED 2020/5/30
JAPAN APPROVED 2020/6/29
CAS 1461640-62-9
| Antineoplastic, Anti-CD38 antibody | |
| Disease | Multiple myeloma |
|---|
Isatuximab, sold under the brand name Sarclisa, is a monoclonal antibody (mAb) medication for the treatment of multiple myeloma.[4][3]
The most common side effects include neutropenia (low levels of neutrophils, a type of white blood cell), infusion reactions, pneumonia (infection of the lungs), upper respiratory tract infection (such as nose and throat infections), diarrhoea and bronchitis (inflammation of the airways in the lungs).[3]
Isatuximab is an anti-CD38 mAb intended to treat relapsed or refractory multiple myeloma.[5] It entered in Phase II trials for multiple myeloma[6] and T-cell leukemia in 2015.[7]
Medical uses
In the United States it is indicated, in combination with pomalidomide and dexamethasone, for the treatment of adults with multiple myeloma who have received at least two prior therapies including lenalidomide and a proteasome inhibitor.[8][9][10]
In the European Union it is indicated, in combination with pomalidomide and dexamethasone, for the treatment of adults with relapsed and refractory multiple myeloma (MM) who have received at least two prior therapies including lenalidomide and a proteasome inhibitor (PI) and have demonstrated disease progression on the last therapy.[3]
History
It was granted orphan drug designation for multiple myeloma by the European Medicines Agency (EMA) in April 2014, and by the U.S. Food and Drug Administration (FDA) in December 2016.[3][11]
Researchers started a Phase I study with isatuximab in combination with pomalidomide and dexamethasone for the treatment of patients with multiple myeloma (MM). The results during the Phase I trial showed that 26 out of the 45 patients discontinued the treatment due to progression of the disease. The patients had already been heavily pretreated. The latter lead to a manageable safety profile where the dose of isatuximab in combination with pomalidomide and dexamethasone would be capped to the maximum of 10 mg/kg weekly every two weeks for future studies.[12]
Based on the remarkable findings during the Phase I trial, a Phase II trial was launched where researchers investigated isatuximab as a single agent in patients with MM. The heavily pretreated patients reacted well to the single administration of isatuximab during Phase II of the trial.[13]
A Phase III combination trial for plasma cell myeloma is comparing pomalidomide and dexamethasone with and without isatuximab is in progress with an estimated completion date of 2021.[medical citation needed]
Additionally, two Phase III trials were added in 2017. The first trial highlights whether there is an added value in the combination of isatuximab with bortezomib, lenalidomide and dexamethasone. The latter will be tested in patients with newly diagnosed MM who are not qualified for a transplant (IMROZ trial). The second trial evaluates the combinations of isatuximab with carfilzomib and dexamethasone compared to carfilzomib with dexamethasone. The second trial was designed for patients who were previously treated with one to three prior lines (IKEMA). There is currently[when?] no treatment for MM, however promising improvements have been made and the study is still ongoing.[14][15]
In March 2020, it was approved for medical use in the United States.[8][9][10]
The U.S. Food and Drug Administration (FDA) approved isatuximab-irfc in March 2020, based on evidence from a clinical trial (NCT02990338) of 307 subjects with previously treated multiple myeloma.[10] The trial was conducted at 102 sites in Europe, North America, Asia, Australia and New Zealand.[10]
The trial evaluated the efficacy and side effects of isatuximab-irfc in subjects with previously treated multiple myeloma.[10] Subjects were randomly assigned to receive either isatuximab-irfc (in combination with pomalidomide and low-dose dexamethasone) or active comparator (pomalidomide and low-dose dexamethasone).[10] Treatment was administered in both groups in 28-day cycles until disease progression or unacceptable toxicity.[10] Both subjects and health care providers knew which treatment was given.[10] The trial measured the time patients lived without the cancer growing (progression-free survival or PFS).[10]
It was approved for medical use in the European Union in May 2020.[3]
Structure and reactivity
The structure of isatuximab consists of two identical immunoglobulin kappa light chains and also two equal immunoglobulin gamma heavy chains. Chemically, isatuximab is similar to the structure and reactivity of daratumumab, hence both drugs show the same CD38 targeting. However, isatuximab shows a more potent inhibition of its ectozyme function. The latter gives potential for some non-cross reactivity. Isatuximab shows action of an allosteric antagonist with the inhibition of the CD38 enzymatic activity. Additionally, isatuximab shows potential where it can induce apoptosis without cross linking.[16] Lastly, Isatuximab reveals direct killing activity when a larger increase in apoptosis is detected in CD38 expressing cancer cells. Furthermore, isatuximab demonstrated a dose dependent inhibition of CD38 enzymatic activity. However, daratumumab with the same experimental conditions shows a more limited inhibition without a dose response.[17]
Reactions
Isatuximab binds uniquely to an epitope on the CD38 receptor and is the only CD38 antibody which can start apoptosis directly.[18] Isatuximab binds to a different CD38 epitope amino-acid sequence than does the anti-CD38 monoclonal antibody daratumumab.[19] The binding with the CD38 receptor is mainly via the gamma heavy chains and are more potent than other CD38 antibodies such as daratumumab which can inhibit the enzymatic activity of CD38. Moreover, isatuximab inhibits the hydrolase activity of CD38.[medical citation needed]
The antibodies show signs of improving antitumor immunity by eliminating regulatory T cells, B cells and myeloid-derived suppressor cells. The difference in binding between isatuximab and daratumumab is in the recognition of the different amino acid groups. Isatuximab identifies 23 amino acids of CD38 to the contrary with daratumumab who has 27. The residue of Glu233 has a flexible sidechain and faces the N-terminal of Asp1 residue in the isatuximab light chain. The latter light chain of isatuximab is also flexible which makes the interaction between CD38/Glu233 and the Asp1 weaker than the other interactions between CD38 and isatuximab. The caspase-dependent apoptotic pathway and the lysosomal mediated cell death pathway in MM cells is induced by isatuximab. The MM cell death follows the downstream reactions of the lysosomal activation. The latter also activates the production of reactive oxygen species.[20]
Available forms
Isatuximab or isatuximab-irfc is available as a drug in an intravenous infusion form. Injection doses are 100 mg/5 mL (20 mg/mL) solution in single-dose vial or 500 mg/25 mL (20 mg/mL) solution in single-dose vial.[4]
Mechanism of action
Cancer of the blood that is distinguished by an overproduction of malignant plasma cells in the bone marrow is called multiple myeloma. The myeloma cells are marked with uniformed overexpression of CD38 surface glycoproteins. Although these proteins are also expressed on other myeloid and lymphoid cells, the extent is relatively minor compared to myeloma cells. The fact that CD38 glycoproteins carry out various important cellular functions, and that they are plentiful on the surface of myeloma cells, has made them an appealing target for multiple myeloma treatment.[21] CD38 was first described as an activation marker, but later the molecule displayed functions in adhesion to endothelial CD31 proteins, e.g. as an aiding component of the synapse complex, as well as an ectoenzyme implicated in the metabolism of extracellular NAD+ and cytoplasmic NADP. The tumour cells can evade the immune system, possibly due to adenosine, an immunosuppressive molecule that arises as a product of the ectoenzymatic activity of CD38.[22]
Isatuximab-irfc is an IgG1-derived monoclonal antibody that selectively binds to the CD38 that exists on the exterior of hematopoietic and multiple myeloma cells (as well as other tumor cells). This drug induces apoptosis of tumor cells and activates immune effector mechanisms such as complement dependent cytotoxicity (CDC), antibody-dependent cellular phagocytosis (ADCP), and antibody-dependent cell-mediated cytotoxicity (ADCC). Isatuximab-irfc is able to stimulate natural killer (NK) cells in the absence of CD38-positive target tumor cells and blocks CD38-positive T-regulatory cells.[4] Furthermore, the NADase activity of CD38 is adjusted by isatuximab, similarly to other CD38 antibodies. Contrarily to daratumumab however, isatuximab can incite apoptosis directly without cross-linking, and in its binding epitope.[23] According to the FDA, isatuximab-irfc alone has reduced ADCC and direct tumor cell killing activity in vitro in comparison to when it is combined with pomalidomide. As well as increased anti-tumor activity as opposed to isatuximab-irfc or pomalidomide only in a human multiple myeloma xenograft model.[4]
Metabolism and toxicity
Metabolism
Isatuximab-irfc is likely to be metabolized through catabolic pathways into smaller peptides. When isatuximab is at a constant state it is expected that the ≥99% elimination will occur approximately two months after the last dose was administered. The clearance percentage diminished when the dosages were increased over time, as well as when multiple doses were administered. However, the elimination of isatuximab-irfc did not differ when applied as a single agent or as a combination therapy.[4]
Toxicity
A dose-limiting toxicity (DLT) has characterized been characterized as the development of any of the following: grade ≥ 3 non-hematologic toxicity; grade 4 neutropenia or grade 4 thrombocytopenia lasting more than 5 days; grade ≥ 2 allergic reactions or hypersensitivity (i.e., infusion reactions); or any other toxicity considered to be dose-limiting by the investigators or sponsor. Grade ≤ 2 infusion reactions were excluded from the DLT definition, because, with suitable care, patients that suffered a grade 2 infusion reaction prior to completion of the infusion were able to finalize isatuximab administration.[23]
There is no recommended reduced dose of isatuximab-irfc. In the eventuality of hematological toxicity it may be necessary to delay administration so that the blood count may be recovered.[4] Although there is no counteracting agent for isatuximab, clinical experience with overdoses is seemingly nonexistent as well. Overdose symptoms will probably be in line with the side effects attached to isatuximab. Therefore, infusion reactions, gastrointestinal disturbances and an elevated risk of infections may occur. It is necessary to carefully monitor the patient in case of an overdose and to employ clinically indicated symptomatic and supportive procedures.[21]
No studies have been conducted with isatuximab concerning carcinogenicity, genotoxicity or fertility.[4]
Pregnancy
When given to pregnant women isatuximab-irfc can cause fetal injury, due to the mechanism of action. It can precipitate depletion of immune cells as well as decreased bone density in the fetus. Pregnant women are therefore notified of the potential risks to a fetus, and women that are able to reproduce are advised to use effective contraceptives during treatment and at least five months subsequent to the last dose of isatuximab-irfc.
Furthermore, it is not recommended to combine isatuximab-irfc with pomalidomide in women that are carrying a child, because pomalidomide may cause birth defects and death of the unborn child.[4]
Indications
Isatuximab is indicated as a CD38-directed cytolytic antibody. By inhibiting the enzymatic activity of CD38.
The binding of isatuximab to CD38 on multiple myeloma (MM) cells leads to a trigger to several mechanisms leading to direct apoptosis of target cancer cells. The triggered pathways are the caspase-dependent apoptotic and the lysosome-mediated cell death pathway in MM cells.[24]
It is used in a combination with dexamethasone and pomalidomide. The drug is thus to treat patients with multiple myeloma. Restrictions for the use of isatuximab is that the patients have to be adults who have at least received two previous treatments with lenalidomide and a proteasome inhibitor.[4]
Isatuximab is currently[when?] also being tested in a Phase II trial as a monotherapy against refractory/recurrent systemic light-chain amyloidosis.[24]
Efficacy and side effects
Efficacy
A Phase III study of patients with refractory and relapsed MM, who were resistant to lenalidomide and a proteasome inhibitor, and could not have received daratumumab, another anti-CD38 monoclonal antibody was published in 2019 (ICARIA-MM). The addition of isatuximab to pomalidomide and dexamethasone improved progression free survival to 11.5 months compared to 6.5 months, with an overall response rate of 63%.[25]
Side effects
Adverse reactions to isatuximab-irfc may include neutropenia, infusion-related reactions and/or secondary primary malignancies.[4] Of these three the most commonly occurring ones are the infusion-related reactions.[24] Examples of the most frequent symptoms of infusion-related reactions are dyspnea, cough, chills, and nausea, while the severest signs and symptoms included hypertension and dyspnea.[4]
Effects on animals
The activity of isatuximab has been researched in mouse tumor models. It has been proven that isatuximab leads to antitumor activity in MM cells. Furthermore, the combination of isatuximab and pomalidomide will lead to an extra enhanced antitumor activity in MM cells. Thus, pomalidomide in vivo and in vitro leads to an increase of the activity of isatuximab.[24]
Animal studies in reproduction toxicity have not yet been carried out. So, the risks of birth defects and miscarriage risks are unknown.[4]
Names
Isatuximab is the United States Adopted Name (USAN).[26]
It was developed by ImmunoGen and Sanofi-Aventis with the development name SAR-650984.

References
- ^ Jump up to:a b “Sarclisa Australian prescription medicine decision summary”. Therapeutic Goods Administration (TGA). 14 May 2020. Retrieved 16 August 2020.
- ^ “Isatuximab (Sarclisa) Use During Pregnancy”. Drugs.com. 25 March 2020. Retrieved 25 June 2020.
- ^ Jump up to:a b c d e f “Sarclisa EPAR”. European Medicines Agency (EMA). 24 March 2020. Retrieved 25 June 2020. Text was copied from this source which is © European Medicines Agency. Reproduction is authorized provided the source is acknowledged.
- ^ Jump up to:a b c d e f g h i j k l “Sarclisa- isatuximab injection, solution, concentrate”. DailyMed. 2 March 2020. Retrieved 26 March 2020.
- ^ ImmunoGen, Inc. Announces Data Presentations at Upcoming 57th ASH Annual Meeting and Exposition
- ^ Martin T (2015). “A Dose Finding Phase II Trial of Isatuximab (SAR650984, Anti-CD38 mAb) As a Single Agent in Relapsed/Refractory Multiple Myeloma”. Blood. 126 (23): 509. doi:10.1182/blood.V126.23.509.509.
- ^ “Safety and Efficacy of Isatuximab in Lymphoblastic Leukemia”. ClinicalTrials.gov. Retrieved 4 March 2020.
- ^ Jump up to:a b “FDA approves isatuximab-irfc for multiple myeloma”. U.S. Food and Drug Administration (FDA). 2 March 2020. Retrieved 2 March 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain. - ^ Jump up to:a b “FDA Approves New Therapy for Patients with Previously Treated Multiple Myeloma”. U.S. Food and Drug Administration (FDA) (Press release). 2 March 2020. Retrieved 4 March 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain. - ^ Jump up to:a b c d e f g h i “Drug Trials Snapshots: Sarclisa”. U.S. Food and Drug Administration(FDA). 2 March 2020. Retrieved 25 March 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain. - ^ “Isatuximab Orphan Drug Designations and Approvals”. U.S. Food and Drug Administration (FDA). 24 December 1999. Retrieved 4 March 2020.
- ^ Mikhael J, Richardson P, Usmani SZ, Raje N, Bensinger W, Karanes C, et al. (July 2019). “A phase 1b study of isatuximab plus pomalidomide/dexamethasone in relapsed/refractory multiple myeloma”. Blood. 134 (2): 123–133. doi:10.1182/blood-2019-02-895193. PMC 6659612. PMID 30862646.
- ^ Martin T (7 December 2015). “A Dose Finding Phase II Trial of Isatuximab (SAR650984, Anti-CD38 mAb) As a Single Agent in Relapsed/Refractory Multiple Myeloma”. ASH.
- ^ Orlowski RZ, Goldschmidt H, Cavo M, Martin TG, Paux G, Oprea C, Facon T (20 May 2018). “Phase III (IMROZ) study design: Isatuximab plus bortezomib (V), lenalidomide (R), and dexamethasone (d) vs VRd in transplant-ineligible patients (pts) with newly diagnosed multiple myeloma (NDMM)”. Journal of Clinical Oncology. 36 (15_suppl): TPS8055. doi:10.1200/JCO.2018.36.15_suppl.TPS8055.
- ^ Moreau P, Dimopoulos MA, Yong K, Mikhael J, Risse ML, Asset G, Martin T (January 2020). “Isatuximab plus carfilzomib/dexamethasone versus carfilzomib/dexamethasone in patients with relapsed/refractory multiple myeloma: IKEMA Phase III study design”. Future Oncology. 16 (2): 4347–4358. doi:10.2217/fon-2019-0431. PMID 31833394.
- ^ Rajan AM, Kumar S (July 2016). “New investigational drugs with single-agent activity in multiple myeloma”. Blood Cancer Journal. 6 (7): e451. doi:10.1038/bcj.2016.53. PMC 5030378. PMID 27471867.
- ^ Martin T, Baz R, Benson DM, Lendvai N, Wolf J, Munster P, et al. (June 2017). “A phase 1b study of isatuximab plus lenalidomide and dexamethasone for relapsed/refractory multiple myeloma”. Blood. 129 (25): 3294–3303. doi:10.1182/blood-2016-09-740787. PMC 5482100. PMID 28483761.
- ^ Martin TG, Corzo K, Chiron M (2019). “Therapeutic Opportunities with Pharmacological Inhibition of CD38 with Isatuximab”. Cells. 8 (12): 1522. doi:10.3390/cells8121522. PMC 6953105. PMID 31779273.
- ^ Dhillon S (2020). “Isatuximab: First Approval”. Drugs. 80 (9): 905–912. doi:10.1007/s40265-020-01311-1. PMID 32347476. S2CID 216597315.
- ^ Martin TG, Corzo K, Chiron M, Velde HV, Abbadessa G, Campana F, et al. (November 2019). “Therapeutic Opportunities with Pharmacological Inhibition of CD38 with Isatuximab”. Cells. 8 (12): 1522. doi:10.3390/cells8121522. PMC 6953105. PMID 31779273.
- ^ Jump up to:a b “Isatuximab”. Drugbank. 20 May 2019.
- ^ Morandi F, Horenstein AL, Costa F, Giuliani N, Pistoia V, Malavasi F (28 November 2018). “CD38: A Target for Immunotherapeutic Approaches in Multiple Myeloma”. Frontiers in Immunology. 9: 2722. doi:10.3389/fimmu.2018.02722. PMC 6279879. PMID 30546360.
- ^ Jump up to:a b Martin T, Strickland S, Glenn M, Charpentier E, Guillemin H, Hsu K, Mikhael J (March 2019). “Phase I trial of isatuximab monotherapy in the treatment of refractory multiple myeloma”. Blood Cancer Journal. 9 (4): 41. doi:10.1038/s41408-019-0198-4. PMC 6440961. PMID 30926770.
- ^ Jump up to:a b c d Martin TG, Corzo K, Chiron M, Velde HV, Abbadessa G, Campana F, et al. (November 2019). “Therapeutic Opportunities with Pharmacological Inhibition of CD38 with Isatuximab”. Cells. 8 (12): 1522. doi:10.3390/cells8121522. PMC 6953105. PMID 31779273.
- ^ Attal, Michel; Richardson, Paul G; Rajkumar, S Vincent; San-Miguel, Jesus; Beksac, Meral; Spicka, Ivan; Leleu, Xavier; Schjesvold, Fredrik; Moreau, Philippe; Dimopoulos, Meletios A; Huang, Jeffrey Shang-Yi (2019). “Isatuximab plus pomalidomide and low-dose dexamethasone versus pomalidomide and low-dose dexamethasone in patients with relapsed and refractory multiple myeloma (ICARIA-MM): a randomised, multicentre, open-label, phase 3 study”. The Lancet. 394 (10214): 2096–2107. doi:10.1016/s0140-6736(19)32556-5. ISSN 0140-6736. PMID 31735560. S2CID 208049235.
- ^ Statement On A Nonproprietary Name Adopted By The USAN Council – Isatuximab, American Medical Association
External links
- “Isatuximab”. Drug Information Portal. U.S. National Library of Medicine.
- Clinical trial number NCT02990338 for “Multinational Clinical Study Comparing Isatuximab, Pomalidomide, and Dexamethasone to Pomalidomide and Dexamethasone in Refractory or Relapsed and Refractory Multiple Myeloma Patients (ICARIA-MM)” at ClinicalTrials.gov
| Isatuximab (pale blue) binding CD38 (purple). PDB: 4CMH | |
| Monoclonal antibody | |
|---|---|
| Type | Whole antibody |
| Source | Chimeric (mouse/human) |
| Target | CD38 |
| Clinical data | |
| Trade names | Sarclisa |
| Other names | SAR-650984, isatuximab-irfc |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a620023 |
| License data | US DailyMed: Sarclisa |
| Pregnancy category | AU: C[1]US: N (Not classified yet)[2] |
| Routes of administration | Intravenous |
| Drug class | Antineoplastic |
| ATC code | None |
| Legal status | |
| Legal status | AU: S4 (Prescription only) [1]US: ℞-onlyEU: Rx-only [3] |
| Identifiers | |
| CAS Number | 1461640-62-9 |
| DrugBank | DB14811 |
| ChemSpider | none |
| UNII | R30772KCU0 |
| KEGG | D11050 |
| Chemical and physical data | |
| Formula | C6456H9932N1700O2026S44 |
| Molar mass | 145190.99 g·mol−1 |
////////Isatuximab, Sarclisa, 2020APPROVALS, JAPAN 2020, US 2020, EU 2020, PEPTIDE, SANOFI , イサツキシマブ (遺伝子組換え) ,
Borofalan (10B)

.png)
Borofalan (10B), ボロファラン (10B), 硼[10B]法仑
APPROVED JAPAN, 2020/3/25, Steboronine
Antineoplastic, Diagnostic aid, Radioactive agent
(2S)-2-amino-3-(4-(10B)dihydroxy(10B)phenyl)propanoic acid
| Formula | C9H12BNO4 |
|---|---|
| CAS | 80994-59-8 |
| Mol weight | 209.0069 |
- 4-(Borono-10B)-L-phenylalanine
- (10B)-4-Borono-L-phenylalanine
- Borofalan (10b)
- L-(p-[10B]Boronophenyl)alanine
- L-4-[10B]Boronophenylalanine
- p-[10B]Borono-L-phenylalanine
- L-Phenylalanine, 4-borono-10B-
Marketed Head and neck cancer - Originator Stella Pharma
- Developer Osaka University; Stella Pharma; Sumitomo Heavy Industries
- Class Antineoplastics; Borates; Propionic acids; Radiopharmaceuticals
- Mechanism of Action Ionising radiation emitters
- Phase IIGlioma
- Phase I Haemangiosarcoma; Malignant melanoma
- 20 May 2020 Launched for Head and neck cancer (Inoperable/Unresectable, Late-stage disease, Locally recurrent) in Japan (IV)
- 25 Mar 2020 Registered for Head and neck cancer (Inoperable/Unresectable, Late-stage disease, Locally recurrent) in Japan (IV) – First global approval
- 03 Mar 2020 Chemical structure information added
- Melting Point (Experimental)Value: 285-298 °C (decomp) Hattori, Yoshihide; Tetrahedron Letters 2008, VOL49(33), PG 4977-4980
- SP ROT -5.2 ° Conc: 0.50 g/100mL; Solv: hydrochloric acid 589.3 nm; Temp: 25 °C, Hattori, Yoshihide; Tetrahedron Letters 2008, VOL49(33), PG 4977-4980
Borofalan (10B)
.png)
4-[(10B)Borono]-L-phenylalanine
C9H1210BNO4 : 208.21
[80994-59-8]
With the development of atomic science, radiation therapy such as cobalt hexahydrate, linear accelerator, and electron beam has become one of the main methods of cancer treatment. However, traditional photon or electron therapy is limited by the physical conditions of the radiation itself. While killing the tumor cells, it also causes damage to a large number of normal tissues on the beam path. In addition, due to the sensitivity of tumor cells to radiation, traditional radiation therapy For the more radiation-resistant malignant tumors (such as: glioblastoma multiforme, melanoma), the treatment effect is often poor.
In order to reduce the radiation damage of normal tissues around the tumor, the concept of target treatment in chemotherapy has been applied to radiation therapy; and for tumor cells with high radiation resistance, it is currently actively developing with high relative biological effects (relative Biological effectiveness, RBE) radiation sources, such as proton therapy, heavy particle therapy, neutron capture therapy. Among them, neutron capture therapy combines the above two concepts, such as boron neutron capture therapy, by the specific agglomeration of boron-containing drugs in tumor cells, combined with precise neutron beam regulation, providing better radiation than traditional radiation. Cancer treatment options.
Boron Neutron Capture Therapy (BNCT) is a high-capture cross-section of thermal neutrons using boron-containing ( 10 B) drugs, with 10 B(n,α) 7 Li neutron capture and nuclear splitting reactions. Two heavy charged particles of 4 He and 7 Li are produced. The average energy of the two charged particles is about 2.33 MeV, which has high linear energy transfer (LET) and short range characteristics. The linear energy transfer and range of α particles are 150 keV/μm and 8 μm, respectively, while the 7 Li heavy particles are For 175 keV/μm, 5 μm, the total range of the two particles is equivalent to a cell size, so the radiation damage caused to the organism can be limited to the cell level, when the boron-containing drug is selectively aggregated in the tumor cells, with appropriate The sub-radiation source can achieve the purpose of locally killing tumor cells without causing too much damage to normal tissues.
Since the effectiveness of boron neutron capture therapy depends on the concentration of boron-containing drugs in the tumor cell position and the number of thermal neutrons, it is also called binary cancer therapy; thus, in addition to the development of neutron sources, The development of boron-containing drugs plays an important role in the study of boron neutron capture therapy.
4-( 10 B)dihydroxyboryl-L-phenylalanine (4-( 10 B)borono-L-phenylalanine, L- 10 BPA) is currently known to be able to utilize boron neutron capture therapy (boron neutron capture therapy) , BNCT) An important boron-containing drug for the treatment of cancer.
Therefore, various synthetic methods of L-BPA have been developed. As shown in the following formula (A), the prior art L-BPA synthesis method includes two methods of forming a bond (a) and a bond (b):

Among them, the method for synthesizing L-BPA by forming the bond (a) is to try to introduce a substituent containing a dihydroxylboryl group or a borono group into the skeleton of the phenylalanine, thereby the pair of the amide substituent. The position forms a carbon-boron bond to produce L-BPA.
J. Org. Chem. 1998, 63, 8019 discloses a method for the cross-coupling reaction of (S)-4-iodophenylalanine with a diboron compound by palladium-catalyzed amine end treatment. Amine-protected (S)-4-iodophenylalanine (eg (S)-N-tert-butoxycarbonyl-4-iodophenylalanine ((S)-N-Boc-4-) Iodophenylalanine)) is prepared by cross-coupling with a diboron compound such as bis(pinacolato diboron) to give (S)-N-tert-butoxycarbonyl-4-pentanoylboryl phenylalanine The amine-terminated (S)-4-boranyl ester phenylalanine of the acid ((S)-N-Boc-4-pinacolatoborono phenylalanine); afterwards, the protecting group on the amine end and the boronic end are removed. The above substituents complete the preparation of L-BPA.
However, since the selected 10 B-doped divaleryl diboron is not a commercially available compound, this method requires additional pretreatment of the preparation of the borating agent, resulting in a high process complexity and a long time consuming process. It is impossible to prepare a high yield of L-BPA. In addition, the carboxylic acid group of the protected (S)-4-iodophenylalanine at the amine end needs to be protected by a substituent to form a benzyl ester group to increase the process yield to 88%; however, The preparation of L-BPA in this manner also requires an additional step of deprotecting the carboxylic acid group, which in turn increases the process complexity of L-BPA.
Accordingly, the method provided in this document not only involves pre-treatment of the preparation of the borating agent, but also requires a large amount of process time and synthesis steps to complete the steps of protecting and deprotecting the carboxylic acid group, and is not advantageous as an industry. The main method of synthesizing L-BPA.
On the other hand, a method for synthesizing L-BPA by forming a bond (b) is a coupling reaction of an amino acid with a boron-containing benzyl fragment or a boron-containing benzaldehyde fragment. To synthesize L-BPA. Biosci. Biotech. Biochem. 1996, 60, 683 discloses an enantioselective synthesis of L-BPA which gives the hands of a cyclic ethers of boronic acid and L-proline The chiral derivatives from L-valine are subjected to a coupling reaction to produce L-BPA. However, this method requires the formation of a cyclic ether compound of boric acid from 4-boronobenzylbromide, followed by a coupling reaction with a chiral derivative of L-proline, and in the latter stage. The amino acid undergoes an undesired racemization in the synthesis step, so that the method requires an enzymatic resolution step to reduce the yield to obtain L-BPA having a certain optical purity.
Accordingly, the method provided in the literature still includes the steps of pretreatment of the preparation of the borating agent and post-treatment of the enzymatic resolution, so that the process involved in the method is complicated and takes a long time, and cannot be obtained. High yield of L-BPA.
In addition, L- 10 BPA (4-( 10 B)borono-L-phenylalanine, 4-( 10 B)dihydroxyboryl-L-phenylalanine) containing 10 boron is currently known to accumulate in tumor cells. The key factor is to use the thermal neutron beam to irradiate the boron element accumulated in the tumor cells to kill the tumor cells by capturing the high-energy particles generated by the reaction, thereby achieving the purpose of treating cancer. Therefore, 10 boron can promote the treatment of L- 10 BPA by boron neutron capture treatment.
However, the boron element present in nature contains about 19.9% of 10 boron and about 80.1% of 11 boron. Therefore, many researchers are still actively developing methods that can be applied to the synthesis of L-BPA, especially for the synthesis of 10- boron-rich L-BPA.
J.Org.Chem.1998,63,8019 additionally provides a method of synthesizing 10 boronated agents, since the method involves multiple steps, it is easy to greatly reduce the boron content of 10 10 boron enriched material in the manufacturing process. Therefore, the method provided in this document is not suitable for the synthesis of 10- boron-rich L-BPA.
Another example is the Biosci.Biotech.Biochem.1996,60,683, before the enzymatic resolution step is not performed, the method provided by the articles could not be obtained with a certain L-BPA optical purity; 10 and the method for preparing boronated agents when also relates to multi-step, resulting in conversion of boron-rich material 10 occurs during the manufacturing process. Therefore, the method provided in this document is also not suitable for the synthesis of 10- boron-rich L-BPA.
Furthermore, Bull. Chem. Soc. Jpn. 2000, 73, 231 discloses the use of palladium to catalyze 4-iodo-L-phenylalanine with 4,4,5,5-tetramethyl-1,3,2 A method in which a dioxonium pentoxide (common name: pinacolborane) is subjected to a coupling reaction. However, this document does not mention how to prepare articles 10 boron enriched L-BPA using this method, and 4,4,5,5-tetramethyl-1,3,2-dioxaborolane not a commercial 10 The compounds available in the literature are not suitable for the synthesis of 10- boron-rich L-BPA.
In addition, Synlett. 1996, 167 discloses a method for coupling a iodophenylborate with a zinc derivative of L-serine zinc derivatives, which involves first preparing phenyl iodoborate. The ester and the preparation of a zinc derivative of L-type serine acid, etc., result in a lower yield of the produced L-BPA. In addition, since the 10- boron-rich triiodide 10 boron and 1,3-diphenylpropane-1,3-diol selected for this method are not commercially available compounds, the methods provided in this document are also provided. Still not suitable for the synthesis of 10- boron-rich L-BPA.
SYN

Repub. Korean Kongkae Taeho Kongbo, 2018060319,
PAPER
Research and Development in Neutron Capture Therapy, Proceedings of the International Congress on Neutron Capture Therapy, 10th, Essen, Germany, Sept. 8-13, 2002 (2002), 1-8.
PAPER
European Journal of Pharmaceutical Sciences (2003), 18(2), 155-163
https://www.sciencedirect.com/science/article/abs/pii/S0928098702002567


PAPER
Tetrahedron Letters (2008), 49(33), 4977-4980
PATENT
WO 2004009135
PATENT
US 20130331599
PATENT
WO 2017028751
https://patents.google.com/patent/WO2017028751A1/en
Example 1
Before preparing (S)-N-tert-butoxycarbonyl-4-dihydroxyborylphenylalanine from (S)-N-tert-butoxycarbonyl-4-iodophenylalanine, it is necessary to reveal Process for preparing (S)-N-tert-butoxycarbonyl-4-iodophenylalanine by using (S)-4-iodophenylalanine as a starting material and a process for preparing 10 tributyl borate with 10 boric acid.
1. Preparation of (S)-N-tert-butoxycarbonyl-4-iodophenylalanine from (S)-4-iodophenylalanine
Please refer to the following reaction formula I, which is (S)-4-iodophenylalanine in a solvent of 1,4-dioxane (1,4-dioxane) and water (H 2 O) with hydrogen peroxide. Sodium (NaOH) and di-tert-butyl dicarbonate (Boc 2 O) are reacted to obtain a chemical reaction formula of (S)-N-tert-butoxycarbonyl-4-iodophenylalanine.

In the preparation process, two reaction vessels were selected for the reaction.
The specific operation process is as follows:
1. Set up a reaction using a 3L three-neck bottle.
2. (S)-4-iodo-L-phenylalanine (200.00 g, 687.10 mmol, 1.00 eq) was added to the reaction system.
3. Add 1,4-dioxane (1.00 L) and water (1.00 L) to the reaction system, respectively.
4. Sodium hydroxide (68.71 g, 1.72 mol, 2.50 eq) was added to the reaction system, the solution gradually became clear, and the temperature rose slightly to 19 °C.
5. When the system is cooled to 0-10 ° C, di-tert-butyl dicarbonate (254.93 g, 1.17 mol, 268.35 mL, 1.70 eq) is added to the reaction system, and the temperature of the reaction system is naturally raised to 10 to 30 ° C and Stir at room temperature (about 30 ° C) for 8 hours.
6. The reaction was detected using high performance liquid chromatography (HPLC) until the starting of the reaction.
7. The temperature of the control system is less than 40 ° C, and the 1,4-dioxane in the reaction solution is concentrated.
8. The reaction system was lowered to room temperature (about 25 ° C), 100 mL of water was added, and the pH was adjusted to 1.8-2 with hydrochloric acid (2M (ie, molarity, M)).
9. Extract three times with ethyl acetate (2 L).
10. Combine the organic phases and wash twice with saturated brine (1 L).
11. The organic phase was dried over sodium sulfate (200 g).
12. Continue drying in an oven (40-45 ° C) to give (S)-N-tert-butoxycarbonyl-4-iodo-L-phenylalanine (250.00 g, 626.28 mmol, HPLC analysis, yield 93.00 %, purity 98%).
The prepared (S) -N- tert-butoxycarbonyl-4-iodo-phenylalanine was -L- Hydrogen 1 nuclear magnetic resonance spectrum analysis (1 HNMR) as follows:
1 H NMR: (400 MHz DMSO-d 6 )
δ 7.49 (d, J = 7.8 Hz, 2H), 6.88 (d, J = 7.8 Hz, 2H), 5.80 (d, J = 5.9 Hz, 1H), 3.68 (d, J = 5.5 Hz, 1H), 3.00-2.90 (m, 1H), 2.87-2.75 (m, 1H), 1.35-1.15 (m, 9H).
Second, tributyl borate 10 was prepared from boronic acid 10
See the following reaction formulas II, 10 as boric acid (H 2 SO 4) is reacted with sulfuric acid in a solvent (butan-1-ol), and toluene (Toluene) in n-butanol, to obtain 10 tributyl borate (10 The chemical reaction formula of B(OBu) 3 ).

The specific operation process is as follows:
1. Set up a reaction device R1 using a 3L three-necked bottle, and configure a water separator on the device.
2. 10 boric acid (150.00 g, 2.46 mol, 1.00 eq) was added to the reaction R1 at room temperature (about 25 ° C).
3. Add n-butanol (1.00 L) to the reaction R1 at room temperature (about 25 ° C) and stir, and most of the boric acid cannot be dissolved.
4. Toluene (1.00 L) was added to the reaction R1 at room temperature (about 25 ° C) and stirred.
5. Concentrated sulfuric acid (4.82 g, 49.16 mmol, 2.62 mL, 0.02 eq) was added dropwise to the reaction at room temperature (about 25 ° C), at which time a large amount of solid remained undissolved.
6. The reaction system was heated to 130 ° C, and the water was continuously removed, stirred for 3.5 hours, and water (about 140 g) was formed in the water separator. The solids were all dissolved, and the solution changed from colorless to brown. .
7. TLC (DCM: MeOH = 5:1, Rf = 0.43, bromocresol green).
8. Distill off most of the toluene at atmospheric pressure.
9. After most of the toluene is distilled off, the temperature of the system is lowered to 20 to 30 ° C, and the reaction liquids of the two reactions are combined, and the apparatus is changed for distillation.
10. Oil bath external temperature 108-110 ° C pump distillation under reduced pressure, Kelvin thermometer 45 ° C, distilled n-butanol.
11. Oil bath external temperature 108-110 ° C oil pump distillation under reduced pressure, the residual butanol was distilled off.
12. Oil bath external temperature 118-120 ° C oil pump vacuum distillation, Kelvin thermometer 55 ° C, began to produce products.
13. The temperature is raised to 135-140 ° C oil pump vacuum distillation, the product is completely distilled.
14. The product is obtained as a colorless liquid 10 tributyl borate (830.00g, 3.62mol, yield 73.58%).
The results of the 1 H NMR analysis of the obtained tributyl 10 borate were as follows:
1 H NMR: (400 MHz CDCl 3 )
δ 3.82-3.68 (m, 6H), 1.57-1.42 (m, 6H), 1.34 (qd, J = 7.4, 14.9 Hz, 6H), 0.95-0.80 (m, 9H).
Three, -N- tert-butoxycarbonyl-4-iodo-phenylalanine was prepared (S) of (S) -N- tert-butoxycarbonyl-4-hydroxy-10-yl -L- phenylalanine boron
Please refer to the following reaction formula III, which is (S)-N-tert-butoxycarbonyl-4-iodophenylalanine with tributyl 10 borate, t-butyl magnesium chloride (t-BuMgCl) and bis (2-A) yl aminoethyl) ether (BDMAEE) reaction, to produce (S) -N- tert-butoxycarbonyl group -4- (10 B) dihydroxyboryl -L- phenylalanine chemical reaction.

In the preparation process, two reaction vessels were selected for the reaction.
The specific operation process is as follows:
1. Set up a reaction using a 3L three-neck bottle.
2. Tributyl 10 borate (187.60 g, 87.98 mmol, 3.20 eq) was placed in the reaction system at room temperature (about 22 ° C).
3. Sodium hydride (20.45 g, 511.24 mmol, purity 60%, 2.00 eq) was added to the reaction system at room temperature (about 22 ° C). The reaction solution was a suspension and stirred at room temperature (about 22 ° C). 5 minutes.
4. Bis(2-methylaminoethyl)ether (327.73 g, 2.04 mol, 8.00 eq) was added to the reaction at room temperature (about 22 ° C).
5. N-tert-Butoxycarbonyl-4-iodo-L-phenylalanine (100.00 g, 255.62 mmol, 1.00 eq) was added to the reaction system at room temperature (about 22 ° C), and a large amount of solid was not dissolved.
6. Lower the temperature of the reaction system to 0-5 ° C, add t-butyl magnesium chloride (1.7 M, 1.20 L, 2.04 mol, 8.00 eq) to the reaction, control the temperature between 0-10 ° C, the dropping time is about It is 1.5 hours.
7. After the completion of the charging, the temperature of the reaction system was naturally raised to room temperature (20 to 30 ° C) and stirred at this temperature for 12 hours.
8. Using high performance liquid chromatography (HPLC) to detect about 9.00% of the remaining material.
9. When the temperature of the reaction system was lowered to -5 to 0 ° C, it was quenched by dropwise addition of 500 mL of water.
10. Lower the temperature of the system to 0-5 ° C, add methyl tert-butyl ether (500 mL) to the reaction system and adjust the pH to 2.9-3.1 (using a pH meter) with 37% HCl (about 500 mL). Exothermic, the temperature of the control system is between 0-15 °C.
11. The aqueous phase obtained by liquid separation was extracted once with methyl tert-butyl ether (500 mL), and the obtained organic phases were combined to give an organic phase of about 1.1 L.
12. Slowly add a sodium hydroxide aqueous solution (1 M, 400 mL) to the obtained organic phase, exotherm during the dropwise addition, and control the system temperature between 0-15 °C.
13. After the completion of the dropwise addition, the pH of the system was about 10, and the pH was adjusted to between 12.10 and 12.6 with an aqueous sodium hydroxide solution (4M). (measured with a pH meter)
14. Dispensing.
15. The aqueous phase 1 obtained after liquid separation was extracted once with n-butanol (500 ml) to obtain aqueous phase 2.
16. Combine the aqueous phase 2 of the two reaction vessels.
17. Adjust the pH of the aqueous phase to 2.9-3.1 with 37% HCl, stir for about 40 minutes, and precipitate a large amount of solid.
18. Filtration gave a white solid which was washed once with dichloromethane (50 mL).
19. At 25 ° C, the precipitated solid was slurried with dichloromethane (150 mL) and stirred for 10 min.
20. A white solid was filtered to give (S) -N- tert-butoxycarbonyl group -4- (10 B) dihydroxyboryl -L- phenylalanine (75.00g, 240.82mmol, by HPLC analysis, a yield of 47.11% , purity 99%).
The prepared (S) -N- tert-butoxycarbonyl group -4- (10 B) results dihydroxyboryl -L- phenylalanine 1 HNMR was as follows:
1 H NMR: (400 MHz DMSO-d 6 )
Δ12.55 (br.s., 1H), 7.91 (s, 2H), 7.66 (d, J = 7.5 Hz, 2H), 7.17 (d, J = 7.5 Hz, 2H), 4.08-4.01 (m, 1H) ), 3.61-3.53 (m, 1H), 2.98 (dd, J = 4.2, 13.9 Hz, 1H), 2.79 (dd, J = 10.4, 13.5 Hz, 1H), 1.79-1.67 (m, 1H), 1.35- 1.17 (m, 9H).
Preparation of L- 10 BPA from (S)-N-tert-Butoxycarbonyl-4-dihydroxyboryl-L-phenylalanine
See the following reaction scheme IV, which is (S) -N- tert-butoxycarbonyl group -4- (10 B) of amine end dihydroxyboryl -L- phenylalanine deprotection of the chemical reaction, to obtain L- 10 BPA.

The specific operation process is as follows:
1. Set up a reaction using a 1L three-neck bottle.
2. room temperature (20-30 deg.] C) to (S) -N- tert-butoxycarbonyl group -4- (10 B) dihydroxyboryl -L- phenylalanine (67.00g, 217.31mmol, 1.00eq) was added the reaction In the system.
3. room temperature (20-30 deg.] C) water (23.75mL) and acetone (Acetone, 420.00mL) were added dropwise to the reaction flask, stirred (S) -N- tert-butoxycarbonyl group -4- (10 B) dihydroxy Boronyl-L-phenylalanine.
4. Concentrated hydrochloric acid (23.93 g, 656.28 mmol, 23.46 mL, 3.02 eq) was added dropwise to the reaction system at room temperature (20-30 ° C). After the addition was completed, the reaction system was heated to 55-60 ° C and stirred for 4.5 hours.
5. HPLC detection until the reaction of the starting material is completed.
6. The temperature is controlled below 40 ° C, and the acetone in the reaction system is concentrated.
7. Lower the concentrated system to below 15 °C, adjust the pH of the system to about 1.5 with sodium hydroxide solution (4M) (pH meter detection), stir for 40 minutes and continue to adjust the pH of the system to 6.15 using sodium hydroxide solution (4M). ~6.25, a large amount of white solid precipitated, which was filtered to give a white solid, and rinsed with acetone (200mL).
8. Obtained as a white solid L- 10 BPA (36.00 g, 171.17 mmol, HPLC, yield 78.77%, purity 99%).
The analytical results obtained by the L- 10 BPA 1 HNMR are as follows:
1 H NMR: (400 MHz D 2 O, CF 3 COOH)
δ 7.44 (d, J = 7.9 Hz, 1H), 7.03 (d, J = 7.9 Hz, 1H), 4.06 (dd, J = 5.7, 7.5 Hz, 1H), 3.11-3.01 (m, 1H), 2.98 -2.87 (m, 1H).
xample 6
Preparation of (S)-N-tert-butoxycarbonyl-4-dihydroxyboryl-L-phenylalanine from (S)-N-tert-butoxycarbonyl-4-iodophenylalanine
Please refer to the following reaction formula VII, which is a reaction of (S)-N-tert-butoxycarbonyl-4-iodophenylalanine with tributyl borate and t-butylmagnesium chloride (t-BuMgCl) to obtain (S The chemical reaction formula of -N-tert-butoxycarbonyl-4-dihydroxyboryl-L-phenylalanine.

The specific operation process is as follows:
1. Construct a reaction unit with a 250 mL three-neck bottle.
2. Tributyl borate (17.65 g, 76.68 mmol, 3.00 eq) was placed in a 250 mL reaction flask at 20-30 °C.
3. Sodium hydride (1.02 g, 25.56 mmol, 1.00 eq) was added to a 250 mL reaction vial at 20-30 °C.
4. (S)-N-tert-Butoxycarbonyl-4-iodo-L-phenylalanine (10.00 g, 25.56 mmol, 1.00 eq) was added to a 250 mL reaction vial at 20-30 °C.
5. Reduce the temperature of the reaction system to 0 ° C under nitrogen atmosphere, slowly add t-butyl magnesium chloride (1.7 M in THF, 120 mL, 8.00 eq) to the reaction, the dropping time is about 30 minutes, and the control temperature is 0. Between °C and 10 °C.
Stir at 20.20 ~ 30 ° C for 20 hours.
7. HPLC detection of the basic reaction of the raw materials, leaving only about 0.7% of the raw materials.
8. At a temperature of 0 ° C, 5 mL of water was added dropwise to the reaction to quench it. After complete quenching, stirring was continued for 10 minutes.
9. Cool down to 0 ° C, add methyl tert-butyl ether (50 mL) to the reaction and adjust the pH to 3 with 37% HCl (about 50 mL) (detected with a pH meter), adjust the pH during the process to exotherm, control the temperature at 0 Between °C and 15 °C.
12. The aqueous phase obtained by liquid separation was extracted once with methyl t-butyl ether (50 mL) and the organic phases were combined.
12. Add NaOH solution (1M, 55mL) to the obtained organic phase to adjust the pH to between 12.10-12.6. The process is exothermic and the temperature is controlled between 0 °C and 15 °C.
13. Liquid separation, the obtained aqueous phase was extracted once with n-butanol (50 mL), and most of the impurities were extracted and removed.
14. The aqueous phase obtained by liquid separation was adjusted to pH 3 with 37% HCl and stirred for about 30 minutes to precipitate a white solid.
15. Filtration gave a white solid which was washed once with dichloromethane (50 mL).
16. The precipitated solid was slurried with 25 mL of dichloromethane at 25 ° C and stirred for 10 minutes.
17. Filtration of (S)-N-tert-butoxycarbonyl-4-dihydroxyboryl-L-phenylalanine (6.8 g, HPLC, yield: 83.15%, purity 98%).
Example 7
Please continue to refer to Reaction Scheme VII. The specific operation process is as follows:
1. Construct a reaction unit with a 250 mL three-neck bottle.
2. Tributyl borate (8.82 g, 38.34 mmol, 3.00 eq) was added to a 250 mL reaction vial at 20-30 °C.
3. Sodium hydride (511.25 mg, 12.78 mmol, 1.00 eq) was added to a 250 mL reaction vial at 20-30 °C.
4. (S)-N-tert-Butoxycarbonyl-4-iodo-L-phenylalanine (5.00 g, 12.78 mmol, 1.00 eq) was added to a 250 mL reaction vial at 20-30 °C.
5. The temperature of the reaction system was lowered to 0 ° C under nitrogen atmosphere, and t-butyl magnesium chloride (1.7 M in THF, 60 mL, 8.00 eq) was added dropwise to the reaction, the dropwise addition time was about 30 minutes, and the control temperature was 0 ° C. -10 ° C between.
Stir at 6.20 ~ 30 ° C for 22 hours.
7. HPLC detection of the raw material reaction is completed.
8. At a temperature of 0 ° C, 2.5 mL of water was added dropwise to the reaction to quench it. After complete quenching, stirring was continued for 10 minutes.
9. Cool down to 0 ° C, add methyl tert-butyl ether (25 mL) to the reaction and adjust the pH to 3 with 37% HCl (about 25 mL) (detected with a pH meter), adjust the pH during the process to exotherm, control the temperature at 0 Between °C and 15 °C.
12. The aqueous phase obtained by liquid separation was extracted once with methyl t-butyl ether (25 mL) and the organic phases were combined.
12. Add NaOH solution (1M, 30mL) to the obtained organic phase to adjust the pH to between 12.10-12.6. The process is exothermic and the temperature is controlled between 0 °C and 15 °C.
13. Liquid separation, the obtained aqueous phase was extracted once with n-butanol (25 ml), and most of the impurities were extracted and removed.
14. The aqueous phase obtained by liquid separation was adjusted to pH 3 with 37% HCl and stirred for about 30 minutes to precipitate a white solid.
15. Filtration gave a white solid which was washed once with dichloromethane (25 mL).
16. The precipitated solid was slurried with 15 mL of dichloromethane at 25 ° C and stirred for 10 minutes.
17. Filtration gave (S)-N-tert-butoxycarbonyl-4-dihydroxyboryl-L-phenylalanine (3.4 g, obtained by HPLC, yield: 85.26%, purity 98%).
Bis(2-methylaminoethyl)ether is a complexing agent for Mg, which can reduce the occurrence of side reactions in the reaction. The reactions of Examples 6 and 7 were carried out without adding bis(2-methylaminoethyl)ether. The analysis showed that the iodine impurity in the reaction of Example 6 was about 17%, and the iodine impurity in the reaction of Example 7 was observed. About 28%. Therefore, it has been proved from the side that the addition of bis(2-methylaminoethyl)ether can protect the reaction from reducing iodine.
The BPA or 10 BPA obtained in the above examples were analyzed by chiral HPLC, and the ratio of the L-enantiomer to the D-enantiomer was 100:0.
The boron-containing drug L-BPA for neutron capture therapy disclosed in the present invention is not limited to the contents described in the above examples. The above-mentioned embodiments are only examples for convenience of description, and the scope of the claims should be determined by the claims.
PATENT
KR 2018060319
PATENT
WO 2019163790
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2019163790
///////////Borofalan (10B), Borofalan, Steboronine, JAPAN 2020, 2020 APPROVALS, ボロファラン (10B), ボロファラン , 硼[10B]法仑 ,
B(C1=CC=C(C=C1)CC(C(=O)O)N)(O)O
Tepotinib hydrochloride
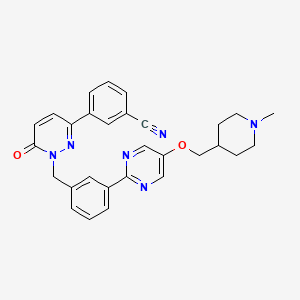
Tepotinib hydrochloride
CS-977;Tepotinib;Veledimex;MSC2156119;EMD-1214063
3-[1-[[3-[5-[(1-methylpiperidin-4-yl)methoxy]pyrimidin-2-yl]phenyl]methyl]-6-oxopyridazin-3-yl]benzonitrile;hydrate;hydrochloride
Benzonitrile, 3-(1,6-dihydro-1-((3-(5-((1-methyl-4-piperidinyl)methoxy)-2-pyrimidinyl)phenyl)methyl)-6-oxo-3-pyridazinyl)-, hydrochloride, hydrate
3- (1- {3- [5- (1-methylpiperidin-4-ylmethoxy) pyrimidine) -2-yl] -benzyl} -6-oxo-1,6-dihydro-pyridazin-3-yl) -benzonitrileтепотиниб [Russian] [INN]تيبوتينيب [Arabic] [INN]特泊替尼 [Chinese] [INN]
- 3-[1,6-Dihydro-1-[[3-[5-[(1-methyl-4-piperidinyl)methoxy]-2-pyrimidinyl]phenyl]methyl]-6-oxo-3-pyridazinyl]benzonitrile
- 3-{1-[(3-{5-[(1-methylpiperidin-4-yl)methoxy]pyrimidin2-yl}phenyl)methyl]-6-oxo-1,6-dihydropyridazin-3-yl}benzonitrile
- EMD 1214063
- MSC 2156119
| Formula | C29H28N6O2. HCl. H2OC29H28N6O2FREE |
|---|---|
| CAS | 1946826-82-9 HCL.H2OCAS No. FREE 1100598-32-0 |
| Mol weight | 547.0478492.57 FREE |
JAPAN 25/3 2020 APPROVED, Tepmetko
| Antineoplastic, Receptor tyrosine kinase inhibitor |

SYN

Bioorganic & Medicinal Chemistry Letters, 25(7), 1597-1602; 2015
PATENT
WO 2009006959
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2009006959
Example 40
The preparation of the compound 3- (1- {3- [5- (1-Methyl-piperidin-4-ylmethoxy) -pyrimidin-2-yl] -benzyl} -6-oxo-1,6-dihydro-pyridazin-3 -yl) -benzonitrile (“A257”) takes place analogously to the following scheme
40.1 17.7 g (67.8 mmol) triphenyl are added to a suspension of 13.0 g (56.5 mmol) 3- (5-hydroxypyrimidin-2-yl) -benzoic acid methyl ester and 13.4 g (62.1 mmol) N-Boc-piperidinemethanol in 115 ml THF -phosphine and cooled to 5 ° C. To the suspension kept at this temperature, 13.3 ml (67.8 mmol) of diisopropylazodicarboxylate are added dropwise with stirring within 45 minutes. The reaction mixture is stirred for 1 hour at room temperature. Then a further 22.2 g (84.7 mmol) triphenylphosphine and 16.6 ml (84.7 mmol)
Diisopropyl azodicarboxylate added. The reaction mixture turns 18
Stirred for hours at room temperature and concentrated in vacuo. The resulting solid is filtered off with suction, washed with diethyl ether and chromatographed on a silica gel column with dichloromethane / methanol as the mobile phase: 4- [2- (3-methoxycarbonyl-phenyl) -pyrimidin-5-yloxymethyl] -piperidine-1-carboxylic acid tert .-butyl ester as lemon yellow crystals;
166 ° C .; ESI 428.
40.2 To a suspension of 1.71 g (3.99 mmol) of 4- [2- (3-methoxycarbonyl-phenyl) -pyrimidin-5-yloxymethyl] -piperidine-1-carboxylic acid tert-butyl ester in 20 ml of THF are added under nitrogen 25 ml (25 mmol) of a 1 M solution of diisobutylaluminum hydride in THF were added dropwise. The reaction mixture is stirred at room temperature for 1 hour, and 1 ml of a saturated sodium sulfate solution is added. The resulting precipitate is filtered off with suction and washed with THF and hot 2-propanol. The filtrate is evaporated and recrystallized from tert-butyl methyl ether: {3- [5- (1-Methyl-piperidin-4-ylmethoxy) -pyrimidin-2-yl] -phenyl} -methanol as beige crystals; Mp 175 ° C; ESI 314.
40.3 To a solution of 313 mg (1.00 mmol) {3- [5- (1-methyl-piperidin-4-ylmethoxy) -pyrimidin-2-yl] -phenyl} -methanol in 2 ml THF are successively added 264 mg (1.30 mmol) 3- (6-oxo-1, 6-dihydro-pyridazin-3-yl) benzonitrile and 397 mg (1.5 mmol) triphenylphosphine are added. The reaction mixture is cooled in an ice bath and
294 μl (1.5 mmol) of diisopropylazodicarboxylate are added dropwise with stirring. The
The reaction mixture is stirred for 18 hours at room temperature and evaporated. The residue is chromatographed on a silica gel column using dichloromethane / methanol. The product-containing fractions are combined, evaporated, the residue digested with tert-butyl methyl ether, filtered off with suction and dried in vacuo: 3- (1- {3- [5- (1-methylpiperidin-4-ylmethoxy) pyrimidine) -2-yl] -benzyl} -6-oxo-1,6-dihydro-pyridazin-3-yl) -benzonitrile as colorless crystals; M.p. 177 ° C; ESI 493;
1 H-NMR (de-DMSO): δ [ppm] = 1.33 (m, 2H), 1.75 (m, 3H), 1.89 (m, 2H), 2.17 (S, 3H), 2.80 (m, 2H), 4.05 (d, J = 6.1 Hz 1 2H), 5.45 (s, 2H) 1 7.16 (d, J = 10 Hz, 1 H), 7.49 (m, 2H), 7.73 (t, J = 7.8 Hz, 1H ), 7.93 (d, J = 7.8 Hz, 1H) 1 8.17 (d, J = 10 Hz, 1H), 8.24 (m, 2H), 8.38 (m, 2H), 8.64 (s, 2H).
The hemisulfate, citrate, tartrate, sulfate, succinate and hydrochloride are obtained from “A257” by salt formation.
PATENT
WO 2009007074
PAPER
Bioorganic & Medicinal Chemistry Letters (2015), 25(7), 1597-1602.
https://www.sciencedirect.com/science/article/abs/pii/S0960894X15000955
PAPER
Molecules (2019), 24(6), 1173/1-1173/16.
https://www.mdpi.com/1420-3049/24/6/1173
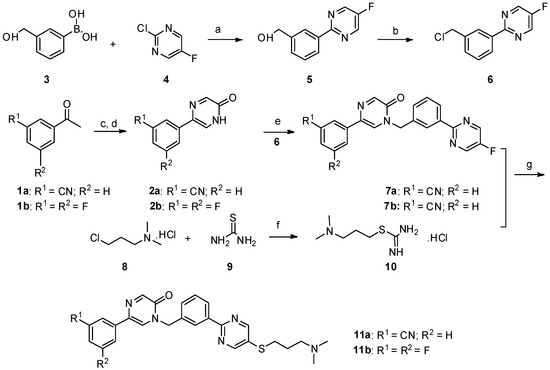
Scheme 1. Reagents and conditions: a) PdCl2(PPh3)2, Na2CO3, ethanol/toluene/water, 90 °C, 8 h; b) SOCl2, CHCl3, reflux; c) SeO2, dioxane:H2O = 10:1, reflux, 12 h; d) NaOH, −30 °C; e) NaH, DMF/THF, 0 °C—room temperature, 12 h; f) dry ethanol, reflux; g) NaOH, DMF/H2O, 60 °C, 8 h, N2.
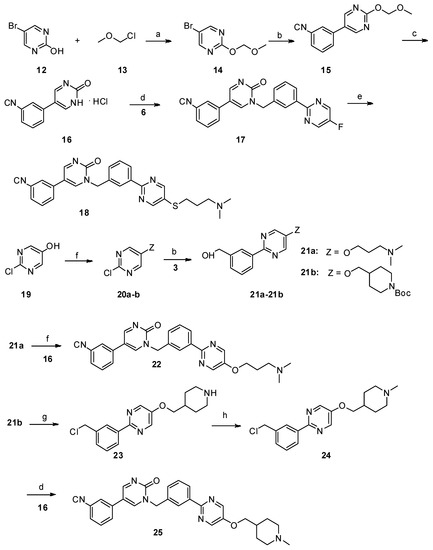
Scheme 2. Reagents and conditions: a) N,N-diisopropylethylamine, dry CH2Cl2, 0 °C—room temperature, 6 h; b) PdCl2(PPh3)2, Na2CO3, ethanol/toluene/water, 90 °C, 8 h; c) 10% aq. HCl, MeOH, reflux; d) K2CO3, dry DMF, 80 °C, 12 h; e) NaOH, DMF/H2O, 60 °C, 8 h, N2; f) PPh3, DIAD, THF, 0 °C—room temperature; g) SOCl2, CHCl3, reflux; h) 35% formaldehyde, NaBH4, MeOH.
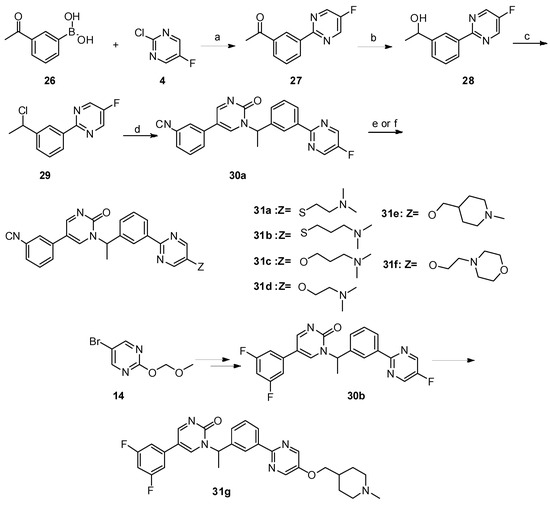
Scheme 3. Reagents and conditions: a) PdCl2(PPh3)2, Na2CO3, ethanol/toluene/water, 90 °C, 8 h; b) NaBH4, MeOH, 0 °C—room temperature, 1 h; c) SOCl2, CHCl3, reflux; d) K2CO3, dry DMF, 80 °C, 12 h; e) 31a–31b: NaOH, DMF/H2O, 60 °C, 8 h, N2; f) 31c–31g: NaH, dry DMF, 0 °C—room temperature, 5 h.
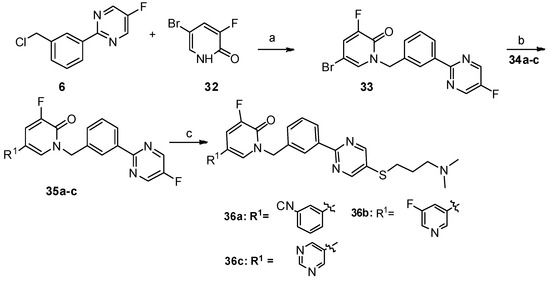
Scheme 4. Reagents and conditions: a) K2CO3, dry DMF, 80 °C, 12 h; b) PdCl2(PPh3)2, Na2CO3, DME/DMF/water, 89 °C, 12 h; c) NaOH, DMF/H2O, 60 °C, 8 h, N2.
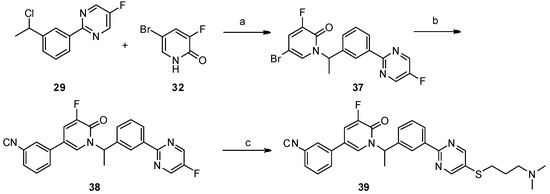
Scheme 5. Reagents and conditions: a) K2CO3, dry DMF, 80 °C, 12 h; b) PdCl2(PPh3)2, Na2CO3, DME/DMF/water, 89 °C, 12 h; c) NaOH, DMF/H2O, 60 °C, 8 h, N2.
///////////Tepotinib, Tepotinib hydrochloride, Tepmetko, JAPAN 2020, 2020 APPROVALS, тепотиниб , تيبوتينيب , 特泊替尼 , EMD 1214063, MSC 2156119
CN1CCC(CC1)COC2=CN=C(N=C2)C3=CC=CC(=C3)CN4C(=O)C=CC(=N4)C5=CC=CC(=C5)C#N.O.Cl
Tirabrutinib
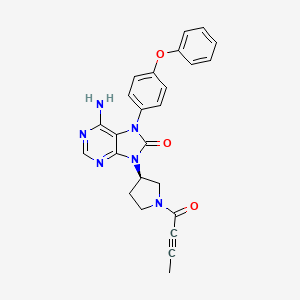
Tirabrutinib
チラブルチニブ塩酸塩
GS-4059
ONO-4059
6-amino-9-[(3R)-1-but-2-ynoylpyrrolidin-3-yl]-7-(4-phenoxyphenyl)purin-8-one
| Formula | C25H22N6O3. HCl |
|---|---|
| CAS | 1439901-97-9 HCL1351636-18-4FREE FORM |
| Mol weight | 490.9415 |
JAPAN APPROVED 2020/3/25 Velexbru
Antineoplastic, Bruton’s tyrosine kinase inhibitor
8H-Purin-8-one,6-amino-7,9-dihydro-9-((3R)-1-(1-oxo-2-butyn-1-yl)-3-pyrrolidinyl)-7-(4-phenoxyphenyl)
6-Amino-9-((3R)-1-(2-butynoyl)-3-pyrrolidinyl)-7-(4-phenoxyphenyl)-7,9-dihydro-8H-purin-8-one
Tirabrutinib (Velexbru®) is an orally administered, small molecule, Bruton’s tyrosine kinase (BTK) inhibitor being developed by Ono Pharmaceutical and its licensee Gilead Sciences for the treatment of autoimmune disorders and haematological malignancies. Tirabrutinib irreversibly and covalently binds to BTK in B cells and inhibits aberrant B cell receptor signalling in B cell-related cancers and autoimmune diseases. In March 2020, oral tirabrutinib was approved in Japan for the treatment of recurrent or refractory primary central nervous system lymphoma. Tirabrutinib is also under regulatory review in Japan for the treatment of Waldenström’s macroglobulinemia and lymphoplasmacytic lymphoma. Clinical development is underway in the USA, Europe and Japan for autoimmune disorders, chronic lymphocytic leukaemia, B cell lymphoma, Sjogren’s syndrome, pemphigus and rheumatoid arthritis. This article summarizes the milestones in the development of tirabrutinib leading to the first approval of tirabrutinib for the treatment of recurrent or refractory primary central nervous system lymphoma in Japan.

PATENT
WO 2011152351
https://patents.google.com/patent/WO2011152351A1/en
Example 19 (2) : 6-amino-9-[(3R) -1- (2-butinoyl) -3-pyrrolidinyl] -7- (4-phenoxyphenyl) -7,9-dihydro-8H-purine- 8-on

TLC: Rf 0.68 (ethyl acetate: methanol = 9: 1);
1 H-NMR (CDCl 3 ): δ 1.94-2.03, 2.23-2.39, 2.80-3.01, 3.50-3.63, 3.67-3.80, 3.86-4.02, 4.03-4.18, 4.23-4.33, 4.42-4.51, 5.11-5.25, 7.04-7.23, 7.34-7.45, 8.20-8.23.
PATENT
WO 2013081016
WO 2015193740
WO 2015181633
WO 2015185998
WO 2016024228
WO 2016024231
WO 2016163531
WO 2016024227
WO 2017033113
PATENT
US 20170035881
https://patents.google.com/patent/US20170035881A1/en
PATENT WO 2017033113
https://patents.google.com/patent/WO2017033113A1/en

///////Tirabrutinib, japan 2020, 2020 approvals, Velexbru , チラブルチニブ塩酸塩 , GS 4059, ONO 4059,
CC#CC(=O)N1CCC(C1)N2C3=NC=NC(=C3N(C2=O)C4=CC=C(C=C4)OC5=CC=CC=C5)N
Berotralstat
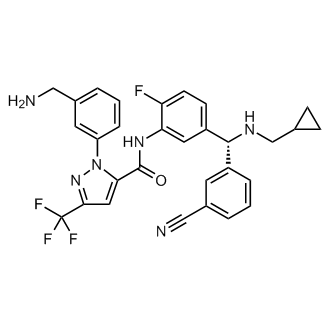

Berotralstat
CAS 1809010-50-1
DIHCl 1809010-52-3
Molecular Formula, C30-H26-F4-N6-O, Molecular Weight, 562.5684
1-(3-(Aminomethyl)phenyl)-N-(5-((R)-(3-cyanophenyl)((cyclopropylmethyl)amino)methyl)-2-fluorophenyl)-3-(trifluoromethyl)-1H-pyrazole-5-carboxamide
1H-Pyrazole-5-carboxamide, 1-(3-(aminomethyl)phenyl)-N-(5-((R)-(3-cyanophenyl)((cyclopropylmethyl)amino)methyl)-2-fluorophenyl)-3-(trifluoromethyl)-
To treat patients with hereditary angioedema
FDA APPROVED 12/4/2020, Orladeyo, 110MG CAPSULE 0RAL
New Drug Application (NDA): 214094
Company: BIOCRYST PHARMACEUTICALS INC
New Drug Application (NDA): 214094
Company: BIOCRYST PHARMACEUTICALS INC

Berotralstat Hydrochloride
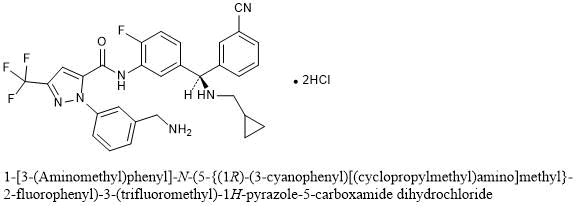
1-[3-(Aminomethyl)phenyl]-N-(5-{(1R)-(3-cyanophenyl)[(cyclopropylmethyl)amino]methyl}-2-fluorophenyl)-3-(trifluoromethyl)-1H-pyrazole-5-carboxamide dihydrochloride
C30H26F4N6O▪2HCl : 635.48
[1809010-52-3]
Berotralstat, also known as BCX-7353, is a kallikrein inhibitor. BCX7353 is a synthetic, once-daily, small molecule drug that can be taken as an oral capsule to treat HAE attacks and for prophylaxis.
Hereditary angioedema (HAE) is rare disorder caused by a SERPING1 gene mutation that triggers severe swelling of the skin and upper airway. Treatment options for HAE with deficient and dysfunctional C1-inhibitor are expanding to include small-molecule drugs that inhibit protein interactions in the kallikrein-kinin system
Serine proteases make up the largest and most extensively studied group of proteolytic enzymes. Their critical roles in physiological processes extend over such diverse areas as blood coagulation, fibrinolysis, complement activation, reproduction, digestion, and the release of physiologically active peptides. Many of these vital processes begin with cleavage of a single peptide bond or a few peptide bonds in precursor protein or peptides. Sequential limited proteolytic reactions or cascades are involved in blood clotting, fibrinolysis, and complement activation. The biological signals to start these cascades can be controlled and amplified as well. Similarly, controlled proteolysis can shut down or inactivate proteins or peptides through single bond cleavages.
Kallikreins are a subgroup of serine proteases. In humans, plasma kallikrein (KLKB1) has no known homologue, while tissue kallikrein-related peptidases (KLKs) encode a family of fifteen closely related serine proteases. Plasma kallikrein participates in a number of pathways relating to the intrinsic pathway of coagulation, inflammation, and the complement system.
Coagulation is the process by which blood forms clots, for example to stop bleeding. The physiology of coagulation is somewhat complex insofar as it includes two separate initial pathways, which converge into a final common pathway leading to clot formation. In the final common pathway, prothrombin is converted into thrombin, which in turn converts fibrinogen into fibrin, the latter being the principal building block of cross- linked fibrin polymers which form a hemostatic plug. Of the two initial pathways upstream of the final common pathway, one is known as the contact activation or intrinsic pathway, and the other is known as the tissue factor or extrinsic pathway.
The intrinsic pathway begins with formation of a primary complex on collagen by high-molecular- weight kininogen (HMWK), prekallikrein, and FXII (Factor XII; Hageman factor). Prekallikrein is converted to kallikrein, and FXII is activated to become FXIIa. FXIIa then converts Factor XI (FXI) into FXIa, and FXIa in turn activates Factor IX (FIX), which with its co-factor F Villa form the“tenase” complex, which activates Factor X (FX) to FXa. It is FXa which is responsible for the conversion of prothrombin into thrombin within the final common pathway.
Prekallikrein, the inactive precursor of plasma kallikrein, is synthesized in the liver and circulates in the plasma bound to FDVTWK or as a free zymogen. Prekallikrein is cleaved by activated factor XII(FXIIa) to release activated plasma kallikrein (PK). Activated plasma kallikrein displays endopeptidase activity towards peptide bonds after arginine (preferred) and lysine. PK then generates additional FXIIa in a feedback loop which in turn activates factor XI (FXI) to FXIa to connect to the common pathway. Although the initial activation of the intrinsic pathway is through a small amount of FXIIa activating a small amount of PK, it is the subsequent feedback activation of FXII by PK that controls the extent of activation of the intrinsic pathway and hence downstream coagulation. Hathaway, W. E., et al. (1965) Blood 26:521-32.
Activated plasma kallikrein also cleaves HMWK to release the potent vasodilator peptide bradykinin. It is also able to cleave a number of inactive precursor proteins to generate active products, such as plasmin (from plasminogen) and urokinase (from prourokinase). Plasmin, a regulator of coagulation, proteolytically cleaves fibrin into fibrin degradation products that inhibit excessive fibrin formation.
Patients who have suffered acute myocardial infarction (MI) show clinical evidence of being in a hypercoagulable (clot-promoting) state. This hypercoagulability is
paradoxically additionally aggravated in those receiving fibrinolytic therapy. Increased generation of thrombin, as measured by thrombin-antithrombin III (TAT) levels, is observed in patients undergoing such treatment compared to the already high levels observed in those receiving heparin alone. Hoffmeister, H. M. et al. (1998) Circulation 98:2527-33. The increase in thrombin has been proposed to result from plasmin-mediated activation of the intrinsic pathway by direct activation of FXII by plasmin.
Not only does the fibrinolysis-induced hypercoagulability lead to increased rates of reocclusion, but it is also probably responsible, at least in part, for failure to achieve complete fibrinolysis of the clot (thrombus), a major shortcoming of fibrinolytic therapy (Keeley, E. C. et al. (2003) Lancet 361 : 13-20). Another problem in fibrinolytic therapy is the accompanying elevated risk of intracranial hemorrhage. Menon, V. et al. (2004) (Chest l26:549S-575S; Fibrinolytic Therapy Trialists’ Collaborative Group (1994) Lancet 343 :311-22. Hence, an adjunctive anti -coagulant therapy that does not increase the risk of bleeding, but inhibits the formation of new thrombin, would be greatly beneficial. Plasma kallikrein inhibitors also have therapeutic potential for treating hereditary angioedema (HAE). HAE is is a serious and potentially life-threatening rare genetic illness, caused by mutations in the Cl -esterase inhibitor (C1INH) gene, located on chromosome 1 lq. HAE is inherited as an autosomal dominant condition, although one quarter of diagnosed cases arise from a new mutation. HAE has been classed as an orphan disease in Europe, with an estimated prevalence of 1 in 50,000. Individuals with HAE experience recurrent acute attacks of painful subcutaneous or submucosal edema of the face, larynx, gastrointestinal tract, limbs or genitalia which, if untreated, may last up to 5 days. Attacks vary in frequency, severity and location and can be life-threatening. Laryngeal attacks, with the potential for asphyxiation, pose the greatest risk. Abdominal attacks are especially painful, and often result in exploratory procedures or unnecessary surgery. Facial and peripheral attacks are disfiguring and debilitating.
HAE has a number of subtypes. HAE type I is defined by CllNH gene mutations which produce low levels of Cl -inhibitor, whereas HAE type II is defined by mutations which produce normal levels of ineffective Cl protein. HAE type III has separate pathogenesis, being caused by mutations in the F12 gene which codes for the serine protease known as Factor XII. Diagnostic criteria for distinguishing the subtypes of HAE, and distinguishing HAE from other angioedemas, can be found in Ann Allergy Asthma Immunol 2008; l00(Suppl2): S30-S40 and J Allergy Clin Immunol 2004; 114: 629-37, incorporated herein by reference.
Current treatments for HAE fall into two main types. Older non-specific treatments including androgens and antifibrinolytics are associated with significant side effects, particularly in females. Newer treatments are based on an understanding of the molecular pathology of the disease, namely that CllNH is the most important inhibitor of kallikrein in human plasma and that CllNH deficiency leads to unopposed activation of the kallikrein- bradykinin cascade, with bradykinin the most important mediator of the locally increased vascular permeability that is the hallmark of an attack. All of the currently available targeted therapies are administered by intravenous or subcutaneous injection. There is currently no specific targeted oral chronic therapy for HAE.
Therefore, a need exists to develop inhibitors of PK that can tip the balance of fibrinolysis/thrombosis at the occluding thrombus toward dissolution, thereby promoting reperfusion and also attenuating the hypercoagulable state, thus preventing thrombus from reforming and reoccluding the vessel. In particular, the creation of plasma kallikrein inhibitors that are specific and capable of being formulated for in vivo use could lead to a new class of therapeutics. Thus, what is needed are improved compositions and methods for preparing and formulating plasma kallikrein inhibitors.
For example, in patients with angioedema conditions, small polypeptide PK inhibitor DX-88 (ecallantide) alleviates edema in patients with hereditary angioedema (HAE). Williams, A. et al. (2003) Transfus. Apher. Sci. 29:255-8; Schneider, L. et al.
(2007) J Allergy Clin Immunol. 120:416-22; and Levy, J. H. et al. (2006) Expert Opin. Invest. Drugs 15: 1077-90. A bradykinin B2 receptor antagonist, Icatibant, is also effective in treating HAE. Bork, K. et al. (2007) J. Allergy Clin. Immunol. 119:1497-1503. Because plasma kallikrein generates bradykinin, inhibition of plasma kallikrein is expected to inhibit bradykinin production.
For example, in coagulation resulting from fibrinolytic treatment (e.g., treatment with tissue plasminogen activator or streptokinase), higher levels of plasma kallikrein are found in patients undergoing fibrinolysis. Hoffmeister, H. M. et al. (1998) J. Cardiovasc. Pharmacol. 31 :764-72. Plasmin-mediated activation of the intrinsic pathway has been shown to occur in plasma and blood and was markedly attenuated in plasma from individuals deficient in any of the intrinsic pathway components. Ewald, G. A. et al. (1995) Circulation 91 :28-36. Individuals who have had an acute MI were found to have elevated levels of activated plasma kallikrein and thrombin. Hoffmeister, H. M., et al. (1998) Circulation 98:2527-33.
DX-88 reduced brain edema, infarct volume, and neurological deficits in an animal model of ischemic stroke. Storini, C. et al. (2006) J Pharm. Exp. Ther. 318:849-854. Cl- inhibitor reduced infarct size in a mouse model of middle cerebral artery occlusion
(MCAO). De Simoni, M. G. et al. (2004) Am. J. Pathol. 164: 1857-1863; and Akita, N. et al. (2003) Neurosurgery 52:395-400). B2 receptor antagonists were found to reduce the infarct volume, brain swelling, and neutrophil accumulation and were neuroprotective in an MCAO animal model. Zausinger, S. et al. (2003 ) Acta Neurochir. Suppl. 86:205-7;
Lumenta, D. B. et al. (2006) Brain Res. 1069:227-34; Ding-Zhou, L. et al. (2003) Br. J Pharmacol. 139: 1539-47.
Regarding blood loss during cardiopulmonary bypass (CPB), it has been found that the kallikrein-kinin (i.e., contact) system is activated during CABG. Wachtfogel, Y. T. (1989) Blood 73:468. Activation of the contact system during CPB results in up to a 20- fold increase in plasma bradykinin. Cugno, M. et al. (2006) Chest 120:1776-82; and Campbell, D. J. et al. (2001 ) Am. J. Physiol. Reg. Integr. Comp. Physiol. 281 : 1059-70.
Plasma kallikrein inhibitors P8720 and PKSI-527 have also been found to reduce joint swelling in rat models of arthritis. De La Cadena, R. A. et al. (1995) FASEB J. 9:446- 52; Fujimori, Y. (1993) Agents Action 39:42-8. It has also been found that inflammation in animal models of arthritis was accompanied by activation of the contact system. Blais, C. Jr. et al. (1997) Arthritis Rheum. 40: 1327-33.
Additionally, plasma kallikrein inhibitor P8720 has been found to reduce inflammation in an acute and chronic rat model of inflammatory bowel disease (IBD). Stadnicki, A. et al. (1998) FASEB J. 12:325-33; Stadnicki, A. et al. (1996) Dig. Dis. Sci.
41 :9l2-20; and De La Cadena, R. A., et al. (1995) FASEB J. 9:446-52. The contact system is activated during acute and chronic intestinal inflammation. Sartor, R. B. et al. (1996) Gastroenterology 110: 1467-81. It has been found that B2 receptor antagonist, an antibody to high molecular weight kininogen, or reduction in levels of kininogen reduced clinicopathology in animal models of IBD. Ibid !; Arai, Y. et al. (1999) Dig. Dis. Sci.
44:845-51; and Keith, J. C. et al. (2005) Arthritis Res. Therapy 7 :R769-76.
H-D-Pro-Phe-Arg-chloromethylketone (CMK), an inhibitor of PK and FXII and a physiological inhibitor (Cl -inhibitor), has been found to reduce vascular permeability in multiple organs and reduce lesions in lipopolysaccharide (LPS)- or bacterial-induced sepsis in animals. Liu, D. et al. (2005) Blood 105:2350-5; Persson, K. et al. (2000) J. Exp. Med. 192: 1415-24. Clinical improvement was observed in sepsis patients treated with Cl- inhibitor. Zeerleder, S. et al. (2003) Clin. Diagnost. Lab. Immunol. 10:529-35; Caliezi, C., et al. (2002) Crit. Care Med. 30:1722-8; and Marx, G. et al. (1999) Intensive Care Med.
25: 1017-20. Fatal cases of septicemia are found to have a higher degree of contact activation. Martinez-Brotons, F. et al. (1987) Thromb. Haemost. 58:709-713; and Kalter, E. S. et al. (1985) J. Infect. Dis. 151 : 1019-27.
It has also been found that prePK levels are higher in diabetics, especially those with proliferative retinopathy, and correlate with fructosamine levels. Gao, B.-B., et al. (2007) Nature Med. 13: 181-8; and Kedzierska, K. et al. (2005) Archives Med. Res. 36:539- 43. PrePK is also found to be highest in those with a sensorimotor neuropathy. Christie,
M. et al. (1984) Thromb. Haemostas. (Stuttgart) 52:221-3. PrePK levels are elevated in diabetics and are associated with increased blood pressure. PrePK levels independently correlate with the albumin excretion rate and are elevated in diabetics with
macroalbuminuria, suggesting prePK may be a marker for progressive nephropathy. Jaffa, A. A. et al. (2003) Diabetes 52: 1215-21. Bl receptor antagonists have been found to decrease plasma leakage in rats treated with streptozotocin. Lawson, S. R. et al. (2005)
Eur. J. Pharmacol. 514:69-78. Bl receptor antagonists can also prevent streptozotocin- treated mice from developing hyperglycemia and renal dysfunction. Zuccollo, A. et al. (1996) Can. J. Physiol. Pharmacol. 74:586-9.
PATENT
WO 2015134998
https://patents.google.com/patent/WO2015134998A1/en
PATENT
WO 2020092898
https://patents.google.com/patent/WO2020092898A1/en
Example 1 : Synthetic protocol for racemic compound 54e
Reproduced from WO 2015/134998 and U.S. Patent Application Publication No. 2017/0073314 A1 (both incorporated by reference)

Preparation of 1 -(3-(aminomethyl)phenyl)-N-(5-((3-cyanophenyl)(cyclopropyl- methylamino)methyl)-2-fluorophenyl)-3-(trifluoromethyl)-lH-pyrazole-5-carboxamide
(54e)
Step-l : Preparation of 3-((3-amino-4-fluorophenyl)(hydroxy)methyl)benzonitrile (54b)
To a solution of 3-formylbenzonitrile (54a) (29 g, 217 mmol) in tetrahydrofuran (200 mL) cooled to 0 °C was added freshly prepared Grignard reagent (52c) (245 mL, 221 mmol, ~ 0.9 M in THF) stirred at 0 °C for 1 h, and room temperature for 18 h. The reaction mixture was quenched with 1 N HC1 (aq. 440 mL), stirred for 3 h, neutralized with NaOH (2 N, aq.) to pH = ~ 8. The reaction mixture was extracted with ethyl acetate (600, 300 mL). The combined extracts were washed with brine (120 mL), dried over MgS04, filtered and concentrated in vacuum. The crude product was purified by flash column
chromatography [silica gel, eluting with hexanes/ethyl acetate (1 :0 to 1 : 1) to give 3-((3- amino-4-fluorophenyl)(hydroxy)methyl)benzonitrile (54b) (36.28 g) as a brown gum which was used as such for next step; MS (ES+) 265.3 (M+23).
Step-2: Preparation of tert-butyl 3-(5-(5-((3-cyanophenyl)(hydroxy)methyl)-2- fluorophenylcarbamoyl)-3-(trifluoromethyl)-lH-pyrazol-l-yl)benzylcarbamate (54c)
To a solution of 3-((3-amino-4-fluorophenyl)(hydroxy)methyl)benzonitrile (54b) (24.682 g, 102 mmol) in DMF (480 mL) was added l-(3-((tert- butoxycarbonylamino)methyl)phenyl)-3-(trifluoromethyl)-lH-pyrazole-5-carboxylic acid (lOd) (35.0 g, 91 mmol), N-ethyl-N-isopropylpropan-2-amine (132 mL, 758 mmol), bromotripyrrolidin-l-ylphosphonium hexafluorophosphate(V) (PyBrOP, 42.8 g, 91 mmol) and stirred at room temperature for 19 h. The reaction mixture was diluted with ethyl acetate (1000 mL), washed with water (500, 400 mL), brine (400 mL), dried over MgS04, filtered and concentrated in vacuum. The crude product was purified by flash column chromatography [silica gel, eluting with hexanes/ethyl acetate (1 :0 to 1 : 1)] to afford tert- butyl 3-(5-(5-((3-cyanophenyl)(hydroxy)methyl)-2-fluorophenylcarbamoyl)-3- (trifluoromethyl)-lH-pyrazol-l-yl)benzylcarbamate (54c) (4.583 g, 5% for two steps) as a yellow solid; ¾ NMR (300 MHz, DMSO-i¾) d 10.57 (s, 1H), 7.81 (t, J= 1.7 Hz, 1H), 7.73 – 7.66 (m, 2H), 7.64 – 7.19 (m, 10H), 6.25 (d, J= 4.0 Hz, 1H), 5.78 (d, J= 4.0 Hz, 1H), 4.19 (d, J= 6.1 Hz, 2H), 1.37 (s, 9H); 19F NMR (282 MHz, DMSO-i¾) d -60.81 , -123.09; MS (ES+) 632.3 (M+23).
Step-3: Preparation of tert-butyl 3-(5-(5-((3- cyanophenyl)(cyclopropylmethylamino)methyl)-2-fluorophenylcarbamoyl)-3- (trifluoromethyl)-lH-pyrazol-l-yl)benzylcarbamate (54d)
To a solution of tert-butyl 3-(5-(5-((3-cyanophenyl)(hydroxy)methyl)-2- fluorophenylcarbamoyl)-3-(trifluoromethyl)-lH-pyrazol-l-yl)benzylcarbamate (54c) (1.333 g, 2.187 mmol) in dichloromethane (40 mL) at 0°C was added thionyl chloride (0.340 mL, 4.59 mmol) and warmed to room temperature over 2 h. The reaction mixture was quenched with triethyl amine (2.0 mL, 14.35 mmol) stirred at room temperature for 1 h. It was then treated with cyclopropylmethanamine (4.30 mL, 48.0 mmol), concentrated to remove most of dichloromethane followed by addition of acetonitrile (30 mL), stirring at 70 °C for 14 h, and concentration in vacuum to dryness. The residue was treated with chlorofrom (200 mL), washed with water (100 mL), dried over MgS04 followed by filtration and
concentration. The crude product was purified by flash column chromatography [silica gel eluting with hexanes/ethyl acetate (1 :0 to 2: 1)] to afford tert-butyl 3-(5-(5-((3- cyanophenyl)(cyclopropylmethylamino)methyl)-2-fluorophenylcarbamoyl)-3- (trifluoromethyl)-lH-pyrazol-l-yl)benzylcarbamate (54d) (184 mg, 13%) as colorless gum; ¾ NMR (300 MHz, DMSO-ά) d 10.56 (s, 1H), 7.89 (t, J= 1.7 Hz, 1H), 7.77 – 7.71 (m, 1H), 7.70 – 7.30 (m, 10H), 7.22 (dd, J= 10.3, 8.5 Hz, 1H), 4.93 (s, 1H), 4.19 (d, J= 6.2 Hz, 2H), 2.26 (d, J= 6.6 Hz, 2H), 1.37 (s, 9H), 1.00 – 0.80 (m, 1H), 0.45 – 0.28 (m, 2H), 0.12 – -0.01 (m, 2H); 19F NMR (282 MHz, DMSO-i¾) d -60.80 , -123.20; MS (ES+) 663.4 (M+l). Step-4: Preparation of l-(3-(aminomethyl)phenyl)-N-(5-((3-cyanophenyl)(cyclopropyl- methylamino)methyl)-2-fluorophenyl)-3-(trifluoromethyl)-lH-pyrazole-5-carboxamide (54e)
To a solution of tert-butyl 3-(5-(5-((3- cyanophenyl)(cyclopropylmethylamino)methyl)-2-fluorophenylcarbamoyl)-3- (trifluoromethyl)-lH-pyrazol-l-yl)benzylcarbamate (54d) (161 mg, 0.243 mmol) in 1,4- Dioxane (18 mL) was added hydrogen chloride (2.60 mL, 10.40 mmol, 4 M in l,4-dioxane) and stirred at room temperature for 16 h. the reaction mixture was treated with hexanes, decanted, washed with hexanes, and decanted again. The insoluble crude product was purified by flash column chromatography [silica gel, eluting with chloroform/CMA80 (1 :0 to 2:1)] to afford l-(3-(aminomethyl)phenyl)-N-(5-((3-cyanophenyl)(cyclopropyl- methylamino)methyl)-2-fluorophenyl)-3-(trifluoromethyl)-lH-pyrazole-5-carboxamide (54e). The pure product was dissolved in methanol (10 mL) and added 4 N HC1 (aq. 0.14 mL) followed by concentration in vacuum to dryness to give HC1 salt of l-(3- (aminomethyl)phenyl)-N-(5-((3-cyanophenyl)(cyclopropyl-methylamino)methyl)-2- fluorophenyl)-3-(trifluoromethyl)-lH-pyrazole-5-carboxamide (54e) (74 mg, 48%) white solid; ¾ NMR (300 MHz, DMSO- d, D20 ex NMR) d 8.13 (t, J = 1.7 Hz, 1H), 7.98 – 7.84 (m, 3H), 7.73 – 7.64 (m, 3H), 7.63 – 7.48 (m, 4H), 7.44 (dd, J = 10.2, 8.6 Hz, 1H),
5.75 (s, 1H), 4.12 (s, 2H), 2.76 (d, J = 7.2 Hz, 2H), 1.17 – 0.94 (m, 1H), 0.68 – 0.47 (m, 2H), 0.34-0.24 (m, 2H); 19F NMR (282 MHz, DMSO- d) d -60.82, -120.02; MS (ES+): 563.3 (M+l); Analysis calculated for C30H26F4N6O2.O HCT3.0 H2O: C, 52.26; H, 4.97; N, 12.19; Found: C, 52.26; H, 5.00; N, 11.72.
Example 2: Separation of enantiomers of racemic compound 54e
Reproduced from WO 2015/134998 and U.S. Patent Application Publication No. 2017/0073314 A1 (both incorporated by reference)

Compound I (free base) Separation of (+)-l-(3-(aminomethyl)phenyl)-N-(5-((3-cyanophenyl)(cyclopropyl- methylamino)methyl)-2-fluorophenyl)-3-(trifluoromethyl)-lFl-pyrazole-5-carboxamide (Compound I), and (-)-l-(3-(aminomethyl)phenyl)-N-(5-((3-cyanophenyl)(cyclopropyl- methylamino)methyl)-2-fluorophenyl)-3-(trifluoromethyl)-lFl-pyrazole-5-carboxamide ((-
)-enantiomer)
Isomers of Racemic l-(3-(aminomethyl)phenyl)-N-(5-((3- cyanophenyl)(cyclopropyl-methylamino)methyl)-2-fluorophenyl)-3-(trifluoromethyl)-lF[- pyrazole-5-carboxamide (54e) (0.4 g) were separated by using preparative SFC method using the following conditions to furnish:
Preparative SFC Method used:
Column 20mm x 25.0 cm ChromegaChiral CCS from
Regis Technologies (Morton Grove, IL)
CO2 Co-solvent (Solvent B) Methanol: Isopropanol (1 : 1) with 1%
Isopropylamine
Isocratic Method 20 % Co-solvent at 80 mL/min
System Pressure 200 bar
Column Temperature 25 °C
Sample Diluent Methanol: Isopropanol
Chiral Purity of peaks was determined by following Analytical SFC Method:
Column 4.6 x 100 mm ChiralPak AS from Chiral
Technologies (West Chester, PA)
CO2 Co-solvent (Solvent B) Methanol: Isopropanol (1 : 1) with 0.1%
Isopropylamine
Isocratic Method 5-65 % Co-solvent Gradient at 4 mL/min System Pressure 100 bar
Column Temperature 25 °C
Sample Diluent Methanol
Peak-l (Compound I) 2.1 min 144 mg >95% ee (UV 254)
98.6 % purity (UV 254)
Peak-2 ((-)-enantiomer) 2.4 min 172 mg 95.5 % ee (UV 254)
96.5 % purity (UV 254) 1. Peak-l assigned as (+)-l-(3-(aminomethyl)phenyl)-N-(5-((3- cyanophenyl)(cyclopropyl-methylamino)methyl)-2-fluorophenyl)-3- (trifluoromethyl)-lH-pyrazole-5-carboxamide (Compound I) (144 mg, >95%ee) free base as white solid; Optical rotation: [O]D = (+) 6.83 [CH3OH, 1.2]; ‘H NMR (300 MHz, DMSO-£¾) d 10.53 (s, 1H, D2O exchangeable), 7.88 (t, J= 1.7 Hz, 1H), 7.77 – 7.71 (m, 1H), 7.67 (dt, 7= 7.7, 1.4 Hz, 1H), 7.63 (dd, J= 7.5, 2.1 Hz, 1H), 7.56 (s, 1H), 7.54 – 7.47 (m, 2H), 7.47 – 7.38 (m, 2H), 7.34 (ddt, J= 8.6, 5.9, 2.8 Hz, 2H), 7.22 (dd, J= 10.3, 8.5 Hz, 1H), 4.93 (s, 1H), 3.77 (s, 2H), 2.31 – 2.21 (m, 2H), 0.97 – 0.80 (m, 1H), 0.42 – 0.33 (m, 2H), 0.10 – -0.02 (m, 2H); 19F NMR (282 MHz, DMSO-Ts) d -60.73 , -123.20; MS (ES+) 563.3 (M+l), 561.3 (M-l). To a solution of free base mixture of (+)-l-(3-(aminomethyl)phenyl)-N-(5-((3- cyanophenyl)(cyclopropyl-methylamino)methyl)-2-fluorophenyl)-3- (trifluoromethyl)-lH-pyrazole-5-carboxamide (Compound I) (120 mg) in methanol (15 mL) was added hydrogen chloride (0.969 mL, 1.938 mmol), stirred at room temperature for 10 min, evaporated to dryness to afford (+)-l-(3- (aminomethyl)phenyl)-N-(5-((3-cyanophenyl)(cyclopropyl-methylamino)methyl)-2- fluorophenyl)-3-(trifluoromethyl)-lH-pyrazole-5-carboxamide (Compound I) (100 mg) hydrochloride salt as white solid; ¾ NMR (300 MHz, DMSO-Ts) d 10.84 (s, 1H, D2O exchangeable), 10.44 (s, 2H, D2O exchangeable), 8.44 (s, 3H, D2O exchangeable), 8.30 (s, 1H, D2O exchangeable), 8.09 (d, J= 7.9 Hz, 1H), 7.99 (d, J = 6.8 Hz, 1H), 7.91 – 7.83 (m, 1H), 7.80 – 7.50 (m, 7H), 7.42 (dd, J= 10.3, 8.6 Hz, 1H), 5.78 (d, J= 6.9 Hz, 1H), 4.13 (d, J= 5.7 Hz, 2H), 2.88 – 2.62 (m, 2H), 1.42 – 0.99 (m, 1H), 0.73 – 0.46 (m, 2H), 0.32 (d, J= 4.4 Hz, 2H); 19F NMR (282 MHz, DMSO-i¾) d -60.81 , -119.99; MS (ES+): MS (ES+) 563.3 (M+l), MS (ES-) 561.3 (M-l), 597.3 (M+Cl); Analysis calculated for C30H26F4N6O 2HC1 l.75H20: C, 54.02; H, 4.76; Cl, 10.63; N, 12.60; Found: C, 54.12; H, 4.83; Cl, 10.10; N, 11.97. Peak-2 assigned as (-)-l-(3-(aminomethyl)phenyl)-N-(5-((3-cyanophenyl)(cyclopropyl- methylamino)methyl)-2-fluorophenyl)-3-(trifluoromethyl)-lH-pyrazole-5-carboxamide ((-)-enantiomer) (172 mg, 95.5 % ee) as free base was repurified by flash column chromatography (silica gel 12 g, eluting 0-30% MeOH in chloroform for 15 min) to afford (-)-l-(3-(aminomethyl)phenyl)-N-(5-((3-cyanophenyl)(cyclopropyl- methylamino)methyl)-2-fluorophenyl)-3-(trifluoromethyl)-lH-pyrazole-5-carboxamide ((-)-enantiomer) free base as an off-white solid; Optical rotation: [O]D = (-) 5.44
[CH3OH, 1.25]; ¾ NMR (300 MHz, DMSO-i¾) d 7.88 (t, J= 1.6 Hz, 1H), 7.74 (d, J = 8.1 Hz, 1H), 7.70 – 7.61 (m, 2H), 7.57 (s, 1H), 7.54 – 7.47 (m, 2H), 7.45 – 7.41 (m,
2H), 7.34 (ddq, J= 8.7, 6.1, 3.5, 2.8 Hz, 2H), 7.22 (dd, J= 10.3, 8.5 Hz, 1H), 4.93 (s, 1H), 3.78 (s, 2H), 2.25 (d, J= 6.9 Hz, 2H), 0.90 (ddd, J= 9.8, 8.0, 5.2 Hz, 1H), 0.47 – 0.29 (m, 2H), 0.04 (dd, J= 5.0, 1.5 Hz, 2H); 19F NMR (282 MHz, DMSO-i¾) d -60.73 , -123.19; MS (ES+) 563.3 (M+l), MS (ES-), 561.3 (M-l). To a solution of free base of (-)-l-(3-(aminomethyl)phenyl)-N-(5-((3-cyanophenyl)(cyclopropyl- methylamino)methyl)-2-fluorophenyl)-3-(trifluoromethyl)-lH-pyrazole-5-carboxamide ((-)-enantiomer) (0.124 g, 0.220 mmol) in methanol (15 mL) was added hydrogen chloride (1.102 mL, 2.204 mmol), stirred at room temperature for 10 min, evaporated to dryness to afford (-)-l-(3-(aminomethyl)phenyl)-N-(5-((3-cyanophenyl)(cyclopropyl- methylamino)methyl)-2-fluorophenyl)-3-(trifluoromethyl)-lH-pyrazole-5-carboxamide ((-)-enantiomer) (0.121 g) hydrochloride salt as an off-white solid; Ή NMEE ¾ NMR (300 MHz, DMSO-i¾) d 10.82 (s, 1H, D20 exchangeable), 10.36 (s, 2H, D2O exchangeable), 8.38 (s, 3H, D2O exchangeable), 8.27 (s, 1H), 8.06 (d, J= 7.9 Hz, 1H), 7.98 (d, J= 6.7 Hz, 1H), 7.87 (d, J= 7.7 Hz, 1H), 7.78 – 7.49 (m, 7H), 7.48 – 7.37 (m, 1H), 5.78 (s, 1H), 4.13 (d, j= 5.7 Hz, 2H), 2.72 (s, 2H), 1.14 (s, 1H), 0.56 (d, j= 7.7 Hz, 2H), 0.31 (d, J= 5.0 Hz, 2H); 19F NMR (282 MHz, DMSO-i¾) d -60.82 , -120.03; MS (ES+): MS (ES+) 563.3 (M+l), MS (ES-), 561.3 (M-l), 597.2 (M+Cl); Analysis calculated for C30H26F4N6O.2HCI. I .75H2O: C, 54.02; H, 4.76; Cl, 10.63; N, 12.60; Found: C, 54.12; H, 4.83; Cl, 10.10; N, 11.97.
Example 3 : Preparation of a Seed Crystal of Compound I*2

A solution of Compound I ( see Example 2) in methyl tert-butyl ether (MTBE) (1 equiv) is added to a solution of HC1 (aq) (2 equiv) in methanol (cold), followed by heating to about 30°C, and keeping it at about 30°C for not longer than 5 hours while stirring at about 115 rpm. Compound I bis(HCl) is collected by filtration and dried. The crystalline material obtained can be used as a seed for the crystallization protocol described in
Example 4. Example 4: Large-Scale Synthetic & Crystallization Protocol for Compound I*2(HC1 )

Compound I (free base) Compound I bis(HCI)
37% Aqueous hydrochloric acid (38.1 kg, 32.3 L, 2.14 equiv.) was charged to a clean and empty crystallization vessel, methanol (228.9 kg, 39.5 equiv.) was added, and the contents were cooled to -7 ± 3°C. A solution of Compound I free base (approx. 101.8 kg; 180.9 moles) in MTBE (approx. 1,300 L) was filtered through a polish filter into the crystallization vessel at temperature -5 ± 5°C. After rinse with MTBE, pre-weighed Compound I»2(HCl) seed crystals (1.39 kg, 0.012 equiv.; Example 3) were charged to the crystallization vessel via the manhole. The vessel content was heated to 30-33°C, and the agitation speed was set to 25-50 rpm. After confirmed crystallization, the slurry was agitated for another three to four hours. The product slurry was transferred to centrifuge and isolated by centrifugation. The product was washed with MTBE (585 L). After dry spinning the wet product, Compound I*2(HC1), it was discharged from the centrifuge, and the product was dried at < 40°C under vacuum in a cone drier. Product Compound I»2(HCl) yield: 100 kg; 157.4 mol; approx. 85%.
‘H NMR (300 MHz, DMSO-c/i,) data is shown in the following table:

19F NMR (282 MHz, DMSO- is) data is shown in the following table:

Compound I has two basic sites. The conjugate acid of the primary amine was calculated to have a pKa value of 8.89, and the conjugate acid of the secondary amine was calculated to have a pKa value of 7.86.
The XRPD pattern of Compound I»2(HCl) is shown in Fig. 1. Compound I»2(HCl) has characteristic peaks in its XRPD pattern at values of two theta (°2Q) of 5.28, 8.96, 14.27, 16.18, 19.79, 21.16, 22.01, 23.31, 24.64, and 30.31. TG-IR analysis indicated two, distinct weight loss regions: the first was completed by 125 °C while the second began at approximately 208 °C. IR analysis of the off gasses from this experiment detected only trace amounts of water at the initial weight loss while HC1 gas was detected at the 208°C event. No other solvents were detected in the sample. Thus, it was determined that Compound I*2(HC1) initially loses water when heated and, when heated to above 200°C, the salt begins to break apart and HC1 gas is evolved. The IR signal for all these events is very weak indicating that they are occurring over a range and not at a specified temperature. An exemplary TG-IR spectrum is shown in Fig. 2.
REFERENCES
1: Sohtome Y, Sodeoka M. Development of Chaetocin and S-Adenosylmethionine Analogues as Tools for Studying Protein Methylation. Chem Rec. 2018 Dec;18(12):1660-1671. doi: 10.1002/tcr.201800118. Epub 2018 Oct 16. Review. PubMed PMID: 30324709.
2: Bensussen A, Torres-Sosa C, Gonzalez RA, Díaz J. Dynamics of the Gene Regulatory Network of HIV-1 and the Role of Viral Non-coding RNAs on Latency Reversion. Front Physiol. 2018 Sep 28;9:1364. doi: 10.3389/fphys.2018.01364. eCollection 2018. PubMed PMID: 30323768; PubMed Central PMCID: PMC6172855.
////////berotralstat, Orladeyo, BIOCRYST, APPROVALS 2020, FDA 2020, ORPHAN DRUG, CX-7353, CX 7353,
NCc1cccc(c1)n2nc(cc2C(=O)Nc3cc(ccc3F)[C@H](NCC4CC4)c5cccc(c5)C#N)C(F)(F)F
Viltolarsen
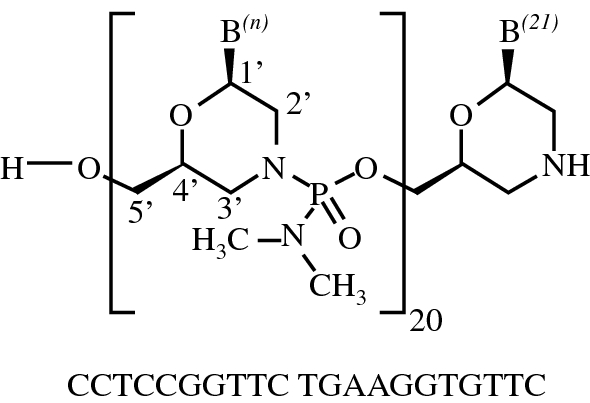
Viltolarsen
维托拉生
ビルトラルセン
| Formula | C244H381N113O88P20 |
|---|---|
| CAS | 2055732-84-6 |
| Mol weight | 6924.8155 |
APPROVED FDA 2020/8/12, Viltepso
APPROVED JAPAN PMDA 2020/3/25, VILTEPSO
- NCNP-01
- NS-065
- NS-065/NCNP-01
- WHO 10771
- WHO-10771
| NAME | DOSAGE | STRENGTH | ROUTE | LABELLER | MARKETING START | MARKETING END | ||
|---|---|---|---|---|---|---|---|---|
| Viltepso | Injection, solution | 250 mg/1 | Intravenous | Ns Pharma, Inc. | 2020-08-13 | Not applicable |
SYNWatanabe N, Nagata T, Satou Y, Masuda S, Saito T, Kitagawa H, Komaki H, Takagaki K, Takeda S: NS-065/NCNP-01: An Antisense Oligonucleotide for Potential Treatment of Exon 53 Skipping in Duchenne Muscular Dystrophy. Mol Ther Nucleic Acids. 2018 Dec 7;13:442-449. doi: 10.1016/j.omtn.2018.09.017.
| PATENT NUMBER | PEDIATRIC EXTENSION | APPROVED | EXPIRES (ESTIMATED) | |
|---|---|---|---|---|
| US9079934 | No | 2011-08-31 | 2031-08-31 |
Viltolarsen
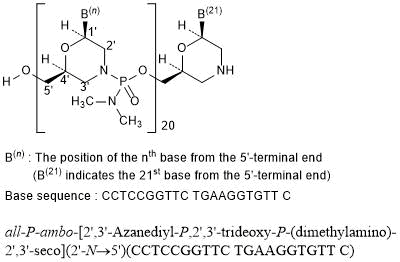
all-P-ambo-[2′,3′-Azanediyl-P,2′,3′-trideoxy-P-(dimethylamino)-2′,3′-seco](2′-N→5′)(CCTCCGGTTC TGAAGGTGTT C)
C244H381N113O88P20 : 6924.82
[2055732-84-6]
Viltolarsen, sold under the brand name Viltepso, is a medication used for the treatment of Duchenne muscular dystrophy (DMD).[3][4][2] Viltolarsen is an antisense oligonucleotide.[3][2]
The most common side effects include upper respiratory tract infection, injection site reaction, cough, and pyrexia (fever).[3][4][2]
Viltolarsen was approved for medical use in the United States in August 2020.[3][4] After golodirsen was approved in December 2019, viltolarsen is the second approved targeted treatment for people with this type of mutation in the United States.[3][5] Approximately 8% of people with DMD have a mutation that is amenable to exon 53 skipping.[3]
Medical uses
Viltolarsen is indicated for the treatment of Duchenne muscular dystrophy (DMD) in people who have a confirmed mutation of the DMD gene that is amenable to exon 53 skipping.[3][2]
DMD is a rare genetic disorder characterized by progressive muscle deterioration and weakness.[3] It is the most common type of muscular dystrophy.[3] DMD is caused by mutations in the DMD gene that results in an absence of dystrophin, a protein that helps keep muscle cells intact.[3] The first symptoms are usually seen between three and five years of age and worsen over time.[3] DMD occurs in approximately one out of every 3,600 male infants worldwide; in rare cases, it can affect females.[3]
Adverse effects
The most common side effects include upper respiratory tract infection, injection site reaction, cough, and pyrexia (fever).[3][4][2]
Although kidney toxicity was not observed in the clinical studies, the clinical experience is limited, and kidney toxicity, including potentially fatal glomerulonephritis, has been observed after administration of some antisense oligonucleotides.[3]
History
Viltolarsen was evaluated in two clinical studies with a total of 32 participants, all of whom were male and had genetically confirmed DMD.[3] The increase in dystrophin production was established in one of those two studies, a study that included sixteen DMD participants, with eight participants receiving viltolarsen at the recommended dose.[3] In the study, dystrophin levels increased, on average, from 0.6% of normal at baseline to 5.9% of normal at week 25.[3] Trial 1 provided data for evaluation of the benefits of viltolarsen.[4] The combined populations from both trials provided data for evaluation of the side effects of viltolarsen.[4] Trial 1 was conducted at six sites in the United States and Canada and Trial 2 was conducted at five sites in Japan.[4] All participants in both trials were on a stable dose of corticosteroids for at least three months before entering the trials.[4]
The U.S. Food and Drug Administration (FDA) concluded that the applicant’s data demonstrated an increase in dystrophin production that is reasonably likely to predict clinical benefit in people with DMD who have a confirmed mutation of the dystrophin gene amenable to exon 53 skipping.[3] A clinical benefit of the drug has not been established.[3] In making this decision, the FDA considered the potential risks associated with the drug, the life-threatening and debilitating nature of the disease, and the lack of available therapies.[3]
The application for viltolarsen was granted priority review designation and the FDA granted the approval to NS Pharma, Inc.[3]
References
- ^ https://www.drugs.com/pregnancy/viltolarsen.html
- ^ Jump up to:a b c d e f “Viltepso- viltolarsen injection, solution”. DailyMed. 12 August 2020. Retrieved 18 August 2020.
- ^ Jump up to:a b c d e f g h i j k l m n o p q r s t u “FDA Approves Targeted Treatment for Rare Duchenne Muscular Dystrophy Mutation”. U.S. Food and Drug Administration (FDA) (Press release). 12 August 2020. Retrieved 12 August 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain. - ^ Jump up to:a b c d e f g h “Drug Trials Snapshots: Viltepso”. U.S. Food and Drug Administration. 12 August 2020. Retrieved 18 August 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain. - ^ Anwar S, Yokota T (August 2020). “Golodirsen for Duchenne muscular dystrophy”. Drugs of Today. 56 (8): 491–504. doi:10.1358/dot.2020.56.8.3159186. PMID 33025945.
Further reading
- Dhillon S (July 2020). “Viltolarsen: First Approval”. Drugs. 80 (10): 1027–1031. doi:10.1007/s40265-020-01339-3. PMID 32519222. S2CID 219542850.
- Dzierlega K, Yokota T (June 2020). “Optimization of antisense-mediated exon skipping for Duchenne muscular dystrophy”. Gene Ther. 27 (9): 407–416. doi:10.1038/s41434-020-0156-6. PMID 32483212. S2CID 219157034.
- Hwang J, Yokota T (October 2019). “Recent advancements in exon-skipping therapies using antisense oligonucleotides and genome editing for the treatment of various muscular dystrophies”. Expert Rev Mol Med. 21: e5. doi:10.1017/erm.2019.5. PMID 31576784.
- Roshmi RR, Yokota T (October 2019). “Viltolarsen for the treatment of Duchenne muscular dystrophy”. Drugs Today. 55 (10): 627–639. doi:10.1358/dot.2019.55.10.3045038. PMID 31720560.
External links
- “Viltolarsen”. Drug Information Portal. U.S. National Library of Medicine (NLM).
- Clinical trial number NCT02740972 for “Safety and Dose Finding Study of NS-065/NCNP-01 in Boys With Duchenne Muscular Dystrophy (DMD)” at ClinicalTrials.gov
| Clinical data | |
|---|---|
| Trade names | Viltepso |
| Other names | NS-065/NCNP-01 |
| AHFS/Drugs.com | Monograph |
| License data | US DailyMed: Viltolarsen |
| Pregnancy category | US: N (Not classified yet)[1] |
| Routes of administration | Intravenous |
| Drug class | Antisense oligonucleotide |
| ATC code | None |
| Legal status | |
| Legal status | US: ℞-only [2]In general: ℞ (Prescription only) |
| Identifiers | |
| CAS Number | 2055732-84-6 |
| DrugBank | DB15005 |
| ChemSpider | 71115970 |
| UNII | SXA7YP6EKX |
| KEGG | D11528 |
| ChEMBL | ChEMBL4298062 |
| Chemical and physical data | |
| Formula | C244H381N113O88P20 |
| Molar mass | 6924.910 g·mol−1 |
//////////Viltolarsen, Viltepso, 维托拉生 , FDA 2020, EU 2020, APPROVALS 2020, NCNP-01, NS-065, NS-065/NCNP-01, WHO 10771, WHO-10771, ビルトラルセン
Pemigatinib


Pemigatinib
INCB054828
| Formula | C24H27F2N5O4 |
|---|---|
| CAS | 1513857-77-62379919-96-5 HCL |
| Mol weight | 487.4991 |
2020/4/17FDA APPROVED, PEMAZYRE
佩米替尼 [Chinese] [INN]
3-(2,6-Difluoro-3,5-dimethoxyphenyl)-1-ethyl-8-(morpholinomethyl)-1,3,4,6-tetrahydro-2H-pyrrolo[3′,2′:5,6]pyrido[4,3-d]pyrimidin-2-one
2H-Pyrrolo[3′,2′:5,6]pyrido[4,3-d]pyrimidin-2-one, 3-(2,6-difluoro-3,5-dimethoxyphenyl)-1-ethyl-1,3,4,7-tetrahydro-8-(4-morpholinylmethyl)-
3-(2,6-difluoro-3,5-dimethoxyphenyl)-1-ethyl-8-(morpholin-4-ylmethyl)-1,3,4,7-tetrahydro-2H-pyrrolo[3′,2′:5,6]pyrido[4,3-d]pyrimidin-2-one
- Originator Incyte Corporation
- Developer Incyte Corporation; Innovent Biologics
- ClassAntineoplastics; Ethers; Fluorobenzenes; Morpholines; Pyridines; Pyrimidinones; Pyrroles; Small molecules
- Mechanism of Action Type 1 fibroblast growth factor receptor antagonists; Type 3 fibroblast growth factor receptor antagonists; Type 4 fibroblast growth factor receptor antagonists; Type-2 fibroblast growth factor receptor antagonists
- Orphan Drug Status Yes – Myeloproliferative disorders; Lymphoma; Cholangiocarcinoma
- MarketedCholangiocarcinoma
- Phase IIBladder cancer; Lymphoma; Myeloproliferative disorders; Solid tumours; Urogenital cancer
- Phase I/IICancer
- 05 Nov 2020Preregistration for Cholangiocarcinoma (Late-stage disease, Metastatic disease, First line therapy, Inoperable/Unresectable) in Japan (PO) in November 2020
- 05 Nov 2020Incyte Corporation stops enrolment in the FIGHT-205 trial for Bladder cancer due to regulatory feedback
- 26 Oct 2020Preregistration for Cholangiocarcinoma (Second-line therapy or greater, Inoperable/Unresectable, Late-stage disease, Metastatic disease) in Canada (PO)
Pemigatinib, also known as INCB054828, is an orally bioavailable inhibitor of the fibroblast growth factor receptor (FGFR) types 1, 2, and 3 (FGFR1/2/3), with potential antineoplastic activity. FGFR inhibitor INCB054828 binds to and inhibits FGFR1/2/3, which may result in the inhibition of FGFR1/2/3-related signal transduction pathways. This inhibits proliferation in FGFR1/2/3-overexpressing tumor cells.
Pemigatinib (INN),[2] sold under the brand name Pemazyre, is a medication for the treatment of adults with previously treated, unresectable locally advanced or metastatic bile duct cancer (cholangiocarcinoma) with a fibroblast growth factor receptor 2 (FGFR2) fusion or other rearrangement as detected by an FDA-approved test.[3][4] Pemigatinib works by blocking FGFR2 in tumor cells to prevent them from growing and spreading.[3]
Pemigatinib belongs to a group of medicines called protein kinase inhibitors.[5] It works by blocking enzymes known as protein kinases, particularly those that are part of receptors (targets) called fibroblast growth factor receptors (FGFRs).[5] FGFRs are found on the surface of cancer cells and are involved in the growth and spread of the cancer cells.[5] By blocking the tyrosine kinases in FGFRs, pemigatinib is expected to reduce the growth and spread of the cancer.[5]
The most common adverse reactions are hyperphosphatemia and hypophosphatemia (electrolyte disorders), alopecia (spot baldness), diarrhea, nail toxicity, fatigue, dysgeusia (taste distortion), nausea, constipation, stomatitis (sore or inflammation inside the mouth), dry eye, dry mouth, decreased appetite, vomiting, joint pain, abdominal pain, back pain and dry skin.[3][4] Ocular (eye) toxicity is also a risk of pemigatinib.[3][4]
Medical uses
Cholangiocarcinoma is a rare form of cancer that forms in bile ducts, which are slender tubes that carry the digestive fluid bile from the liver to gallbladder and small intestine.[3] Pemigatinib is indicated for the treatment of adults with bile duct cancer (cholangiocarcinoma) that is locally advanced (when cancer has grown outside the organ it started in, but has not yet spread to distant parts of the body) or metastatic (when cancer cells spread to other parts of the body) and who have tumors that have a fusion or other rearrangement of a gene called fibroblast growth factor receptor 2 (FGFR2).[3] It should be used in patients who have been previously treated with chemotherapy and whose cancer has a certain type of abnormality in the FGFR2 gene.[6]
History
Pemigatinib was approved for use in the United States in April 2020 along with the FoundationOne CDX (Foundation Medicine, Inc.) as a companion diagnostic for patient selection.[3][4][7]
The approval of pemigatinib in the United States was based on the results the FIGHT-202 (NCT02924376) multicenter open-label single-arm trial that enrolled 107 participants with locally advanced or metastatic cholangiocarcinoma with an FGFR2 fusion or rearrangement who had received prior treatment.[3][4][6] The trial was conducted at 67 sites in the United States, Europe, and Asia.[6] During the clinical trial, participants received pemigatinib once a day for 14 consecutive days, followed by 7 days off, in 21-day cycles until the disease progressed or the patient experienced an unreasonable level of side effects.[3][4][6] To assess how well pemigatinib was working during the trial, participants were scanned every eight weeks.[3] The trial used established criteria to measure how many participants experienced a complete or partial shrinkage of their tumors during treatment (overall response rate).[3] The overall response rate was 36% (95% CI: 27%, 45%), with 2.8% of participants having a complete response and 33% having a partial response.[3] Among the 38 participants who had a response, 24 participants (63%) had a response lasting six months or longer and seven participants (18%) had a response lasting 12 months or longer.[3][4]
The U.S. Food and Drug Administration (FDA) granted the application for pemigatinib priority review, breakthrough therapy and orphan drug designations.[3][4][8][9] The FDA granted approval of Pemazyre to Incyte Corporation.[3]
On 24 August 2018, orphan designation (EU/3/18/2066) was granted by the European Commission to Incyte Biosciences Distribution B.V., the Netherlands, for pemigatinib for the treatment of biliary tract cancer.[5] On 17 October 2019, orphan designation EU/3/19/2216 was granted by the European Commission to Incyte Biosciences Distribution B.V., the Netherlands, for pemigatinib for the treatment of myeloid/lymphoid neoplasms with eosinophilia and rearrangement of PDGFRA, PDGFRB, or FGFR1, or with PCM1-JAK2.[10]
PATENT
US 20200281907
| The present disclosure is directed to, inter alia, methods of treating cancer in a patient in need thereof, comprising administering pemigatinib, which is 3-(2,6-difluoro-3,5-dimethoxyphenyl)-1-ethyl-8-(morpholin-4-ylmethyl)-1,3,4,7-tetrahydro-2H-pyrrolo[3′,2′: 5,6]pyrido[4,3-d]pyrimidin-2-one, having the structure shown below: |
Pemigatinib is described in U.S. Pat. No. 9,611,267, the entirety of which is incorporated herein by reference. Pemigatinib is further described in US Publication Nos.: 2019/0337948 and 2020/0002338, the entireties of which are incorporated herein by reference.
| Provided herein is a method of treating cancer comprising administering a therapy to a patient in need thereof, wherein the therapy comprises administering a therapeutically effective amount of pemigatinib to the patient while avoiding the concomitant administration of a CYP3A4 perpetrator. |
Example 1. Synthesis of Pemigatinib
Step 1: 4-(ethylamino)-1H-pyrrolo[2,3-b]pyridine-5-carbaldehyde
| A mixture of 4-chloro-1H-pyrrolo[2,3-b]pyridine-5-carbaldehyde (CAS #958230-19-8, Lakestar Tech, Lot: 124-132-29: 3.0 g, 17 mmol) and ethylamine (10M in water, 8.3 mL, 83 mmol) in 2-methoxyethanol (20 mL, 200 mmol) was heated to 130° C. and stirred overnight. The mixture was cooled to room temperature then concentrated under reduced pressure. The residue was treated with 1N HCl (30 mL) and stirred at room temperature for 1 h then neutralized with saturated NaHCO 3 aqueous solution. The precipitate was collected via filtration then washed with water and dried to provide the desired product (2.9 g, 92%). LC-MS calculated for C 10H 12N 3O [M+H] + m/z: 190.1; found: 190.1. |
Step 2: 5-{[(2,6-difluoro-3,5-dimethoxyphenyl)amino]methyl}-N-ethyl-1H-pyrrolo[2,3-b]pyridin-4-amine
| A mixture of 4-(ethylamino)-1H-pyrrolo[2,3-b]pyridine-5-carbaldehyde (7.0 g, 37 mmol), 2,6-difluoro-3,5-dimethoxyaniline (9.1 g, 48 mmol) and [(1S)-7,7-dimethyl-2-oxobicyclo[2.2.1]hept-1-yl]methanesulfonic acid (Aldrich, cat #21360: 2 g, 7 mmol) in xylenes (250 mL) was heated to reflux with azeotropic removal of water using Dean-Stark for 2 days at which time LC-MS showed the reaction was complete. The mixture was cooled to room temperature and the solvent was removed under reduced pressure. The residue was dissolved in tetrahydrofuran (500 mL) and then 2.0 M lithium tetrahydroaluminate in THF (37 mL, 74 mmol) was added slowly and the resulting mixture was stirred at 50° C. for 3 h then cooled to room temperature. The reaction was quenched by addition of water, 15% aqueous NaOH and water. The mixture was filtered and washed with THF. The filtrate was concentrated and the residue was washed with CH 2Cl 2 and then filtered to get the pure product (11 g, 82%). LC-MS calculated for C 18H 21F 2N 4O 2[M+H] + m/z: 363.2; found: 363.1. |
Step 3: 3-(2,6-Difluoro-3,5-dimethoxyphenyl)-1-ethyl-1,3,4,7-tetrahydro-2H-pyrrolo[3′,2′:5,6]pyrido[4,3-d]pyrimidin-2-one
| A solution of triphosgene (5.5 g, 18 mmol) in tetrahydrofuran (30 mL) was added slowly to a mixture of 5-{[(2,6-difluoro-3,5-dimethoxyphenyl)amino]methyl}-N-ethyl-1H-pyrrolo[2,3-b]pyridin-4-amine (5.6 g, 15 mmol) in tetrahydrofuran (100 mL) at 0° C. and then the mixture was stirred at room temperature for 6 h. The mixture was cooled to 0° C. and then 1.0 M sodium hydroxide in water (100 mL, 100 mmol) was added slowly. The reaction mixture was stirred at room temperature overnight and the formed precipitate was collected via filtration, washed with water, and then dried to provide the first batch of the purified desired product. The organic layer in the filtrate was separated and the aqueous layer was extracted with methylene chloride. The combined organic layer was concentrated and the residue was triturated with methylene chloride then filtered and dried to provide another batch of the product (total 5.5 g, 92%). LC-MS calculated for C 19H 19F 2N 4O 3[M+H] + m/z: 389.1; found: 389.1. |
Step 4: 3-(2,6-difluoro-3,5-dimethoxyphenyl)-1-ethyl-7-(phenylsulfonyl)-1,3,4,7-tetrahydro-2H-pyrrolo[3′,2′:5,6]pyrido[4,3-d]pyrimidin-2-one
| To a solution of 3-(2,6-difluoro-3,5-dimethoxyphenyl)-1-ethyl-1,3,4,7-tetrahydro-2H-pyrrolo[3′,2′:5,6]pyrido[4,3-d]pyrimidin-2-one (900 mg, 2.32 mmol) in N,N-dimethylformamide (20 mL) cooled to 0° C. was added sodium hydride (185 mg, 4.63 mmol, 60 wt % in mineral oil). The resulting mixture was stirred at 0° C. for 30 min then benzenesulfonyl chloride (0.444 mL, 3.48 mmol) was added. The reaction mixture was stirred at 0° C. for 1.5 h at which time LC-MS showed the reaction completed to the desired product. The reaction was quenched with saturated NH 4Cl solution and diluted with water. The white precipitate was collected via filtration then washed with water and hexanes, dried to afford the desired product (1.2 g, 98%) as a white solid which was used in the next step without further purification. LC-MS calculated for C 25H 23F 2N 4O 5S [M+H] + m/z: 529.1; found: 529.1. |
Step 5: 3-(2,6-difluoro-3,5-dimethoxyphenyl)-1-ethyl-2-oxo-7-(phenylsulfonyl)-2,3,4,7-tetrahydro-1H-pyrrolo[3′,2′:5,6]pyrido[4,3-d]pyrimidine-8-carbaldehyde
| To a solution of 3-(2,6-difluoro-3,5-dimethoxyphenyl)-1-ethyl-7-(phenylsulfonyl)-1,3,4,7-tetrahydro2H-pyrrolo[3′,2′: 5,6]pyrido[4,3-d]pyrimidin-2-one (1.75 g, 3.31 mmol) in tetrahydrofuran (80 mL) at −78° C. was added freshly prepared lithium diisopropylamide (1M in tetrahydrofuran (THF), 3.48 mL, 3.48 mmol). The resulting mixture was stirred at −78° C. for 30 min then N,N-dimethylformamide (1.4 mL, 18 mmol) was added slowly. The reaction mixture was stirred at −78° C. for 30 min then quenched with water and extracted with EtOAc. The organic extracts were combined then washed with water and brine. The organic layer was dried over Na 2SO 4 and concentrated. The residue was purified by flash chromatography eluted with 0 to 20% EtOAc in DCM to give the desired product as a white solid (1.68 g, 91%). LC-MS calculated for C 26H 23F 2N 4O 6S (M+H) + m/z: 557.1; found: 556.9. |
Step 6: 3-(2,6-difluoro-3,5-dimethoxyphenyl)-1-ethyl-8-(morpholin-4-ylmethyl)-7-(phenylsulfonyl)-1,3,4,7-tetrahydro-2H-pyrrolo[3′,2′:5,6]pyrido[4,3-d]pyrimidin-2-one
| To a solution 3-(2,6-difluoro-3,5-dimethoxyphenyl)-1-ethyl-2-oxo-7-(phenylsulfonyl)-2,3,4,7-tetrahydro-1H-pyrrolo[3′,2′: 5,6]pyrido[4,3-d]pyrimidine-8-carbaldehyde (1.73 g, 3.11 mmol) in dichloromethane (50 mL) was added morpholine (0.95 mL, 11 mmol), followed by acetic acid (2 mL, 30 mmol). The resulting yellow solution was stirred at room temperature overnight then sodium triacetoxyborohydride (2.3 g, 11 mmol) was added. The mixture was stirred at room temperature for 3 h at which time LC-MS showed the reaction went to completion to the desired product. The reaction was quenched with saturated NaHCO 3 then extracted with ethyl acetate (EtOAc). The organic extracts were combined then washed with water and brine. The organic layer was dried over Na 2SO 4 and concentrated. The residue was purified by flash chromatography eluted with 0 to 40% EtOAc in DCM to give the desired product as a yellow solid (1.85 g, 95%). LC-MS calculated for C 30H 32F 2N 5O 6S (M+H) + m/z: 628.2; found: 628.0. |
Step 7: 3-(2,6-difluoro-3,5-dimethoxyphenyl)-1-ethyl-8-(morpholin-4-ylmethyl)-1,3,4,7-tetrahydro-2H-pyrrolo[3′,2′: 5,6]pyrido[4,3-d]pyrimidin-2-one (pemigatinib)
| To a solution of 3-(2,6-difluoro-3,5-dimethoxyphenyl)-1-ethyl-8-(morpholin-4-ylmethyl)-7-(phenylsulfonyl)-1,3,4,7-tetrahydro-2H-pyrrolo[3′,2′: 5,6]pyrido[4,3-d]pyrimidin-2-one (1.5 g, 2.4 mmol) in tetrahydrofuran (40 mL) was added tetra-n-butylammonium fluoride (1M in THF, 7.2 mL, 7.2 mmol). The resulting solution was stirred at 50° C. for 1.5 h then cooled to room temperature and quenched with water. The mixture was extracted with dichloromethane (DCM) and the organic extracts were combined then washed with water and brine. The organic layer was dried over Na 2SO 4 and concentrated. The residue was purified by flash chromatography eluted with 0 to 10% MeOH in DCM to give the desired product as a white solid, which was further purified by prep HPLC (pH=2, acetonitrile/H 2O). LC-MS calculated for C 24H 28F 2N 5O 4 (M+H) + m/z: 488.2; found: 488.0. 1H NMR (500 MHz, DMSO) δ 12.09 (s, 1H), 8.06 (s, 1H), 7.05 (t, J=8.1 Hz, 1H), 6.87 (s, 1H), 4.78 (s, 2H), 4.50 (s, 2H), 4.17 (q, J=6.8 Hz, 2H), 3.97 (br, 2H), 3.89 (s, 6H), 3.65 (br, 2H), 3.37 (br, 2H), 3.15 (br, 2H), 1.37 (t, J=6.8 Hz, 3H). |
PATENT
WO 2019213506
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2019213506
PATENT
WO 2019213544
The present disclosure is directed to, inter alia, solid forms, including crystalline forms and amorphous forms, of 3-(2,6-difluoro-3,5-dimethoxyphenyl)-l-ethyl-8-(morpholin-4-ylmethyl)- 1 ,3,4,7 -tetrahydro-2H-pyrrolo [3 ‘,2’ : 5 ,6]pyrido [4,3 -d]pyrimidin-2-one
(Compound 1), and processes and intermediates for preparing the compound. The structure of Compound 1 is shown below.
Compound 1
Compound 1 is described in US Patent No. 9,611,267, the entirety of which is incorporated herein by reference.
Example 1
Synthesis of 3-(2,6-difluoro-3,5-dimethoxyphenyl)-l-ethyl-8-(morpholin-4-ylmethyl)-l^, 4,7-tetrahydro-2H-pyrrolo[3f,2f:5,6]pyrido[4r3-d]pyrimidin-2-one (Compound 1) Scheme 1.
Step 1: Synthesis of 4-((4-chloro-5-(l, 3-dioxolan-2-yl)-l-(phenylsulfonyl)-lH-pyrrolo[2, 3-b ] pyridin-2-yl) methyl) morpholine
To a l-L flask was added 4-chloro-5-(l,3-dioxolan-2-yl)-l-(phenylsulfonyl)-lH-pyrrolo [2,3-b] pyridine (50.0 g, 137 mmol) (see, e.g., Example 2) and tetrahydrofuran (THF, 266 g, 300 mL) under N2. To this mixture at -70 °C was added 2.0 M lithium
diisopropylamide in THF/heptane/ethyl benzene (77.4 g, 95 mL, 190 mmol, 1.4 eq.). The mixture was stirred at -70 °C for 1 h. To the mixture was added /V- formyl morpholine (29.7 g, 258 mmol, 1.9 eq.) in THF (22. 2 g, 25 mL) dropwise. The reaction was done in 30 min after addition. LC/MS showed that the desired product, 4-chloro-5-(l, 3-dioxolan-2-yl)-l-(phenylsulfonyl)- 1 //-pyrrolo [2, 3-61 pyridine-2-carbaldehyde, was formed cleanly. The reaction was quenched with acetic acid (16.4 g, 15.6 mL, 274 mmol, 2.0 eq.) and the dry ice cooling was removed. To the mixture was added morpholine (33.7 g, 33.5 mL, 387 mmol, 2.83 eq.) followed by acetic acid (74.0 g, 70 mL, 1231 mmol, and 9.0 eq.) at 0 °C (internal temperature rose from 0 °C to 18 °C) and stirred overnight. Sodium triacetoxyborohydride (52.50 g, 247.7 mmol, 1.8 eq.) was added and the reaction mixture temperature rose from 20 °C to 32 °C. The mixture was stirred at room temperature for 30 min. HPLC & LC/MS indicated the reaction was complete. Water (100 g, 100 mL) was added followed by 2.0 M sodium carbonate (Na2C03) in water (236 g, 200 mL, 400 mmol, 2.9 eq.) slowly (off gas!). The mixture was stirred for about 30 min. The organic layer was separated and water (250 g, 250 mL) and heptane (308 g, 450 mL) were added. The resulting slurry was stirred for 1 h and the solid was collected by filtration. The wet cake was washed with heptane twice (75.00 mL x 2, 51.3 g x 2) before being dried in oven at 50 °C overnight to give the desired product, 4-((4-chloro-5-( 1 3-dioxolan-2-yl)- 1 -(phenylsulfonyl)- 1 //-pyrrolo|2.3-6 |pyridin-2-yl)methyl)morpholine as a light brown solid (52.00 g, 81.8 % yield): LCMS calculated for C21H23CIN2O5S [M+H]+: 464.00; Found: 464.0; ftf NMR ^OO MHz, DMSO-de) d 8.48 (s, 1 H), 8.38 (m, 2H), 7.72 (m, 1H), 7.64 (m, 2H), 6.83 (s, 1H), 6.13 (s, 1H), 4.12 (m, 2H), 4.00 (m, 2H), 3.92 (s, 2H), 3.55 (m, 4H), 2.47 (m, 4H).
Step 2: Synthesis of 4-chloro-2-(morpholinomethyl)-l-(phenylsulfonyl)-lH-pyrrolo[2, 3-b] pyridine-5 -carbaldehyde
To a 2 L reactor with a thermocouple, an addition funnel, and a mechanical stirrer was charged 4-((4-chloro-5 -(1 ,3 -dioxolan-2-yl)- 1 -(phenylsulfonyl)- 1 //-pyrrolo [2,3 -6]pyridin-2-yl)methyl)morpholine (20.00 g, 43.1 mmol) and dichloromethane (265 g, 200 mL) at room temperature. The resulting mixture was stirred at room temperature (internal temperature
was 19.5 °C) to achieve a solution. To the resulting solution was added an aqueous hydrochloric acid solution (0.5 M, 240 g, 200.0 ml, 100 mmol, 2.32 eq.) at room temperature in 7 min. After over 23 h agitations at room temperature, the bilayer reaction mixture turned into a thick colorless suspension. When HPLC showed the reaction was complete, the slurry was cooled to 0-5 °C and aqueous sodium hydroxide solution (1 N, 104 g, 100 mL, 100 mmol, and 2.32 eq.) was added in about 10 min to adjust the pH of the reaction mixture to 10-11. «-Heptane (164 g, 240 mL) was added and the reaction mixture and the mixture were stirred at room temperature for 1 h. The solid was collected by filtration and the wet cake was washed with water (2 x 40 mL), heptane (2 x 40 ml) before being dried in oven at 50 °C under vacuum to afford the desired product, 4-chloro-2-(morpholinomethyl)-l-(phenylsulfonyl)- 1 //-pyrrolo|2.3-/i |pyridine-5-carbaldehyde as a light brown solid (16.9 g, 93% yield): LCMS calculated for C19H19CIN3O4S [M+H]+: 420.00; Found: 420.0; ¾ NMR (400 MHz, DMSO-de) d 10.33 (s, 1H), 8.76 (s, 1 H), 8.42 (m, 2H), 7.74 (m, 1H), 7.65 (m, 2H), 6.98 (s, 1H), 3.96 (m, 2H), 3.564 (m, 4H), 2.51 (m, 4H).
Step 3: Synthesis ofN-((4-chloro-2-(morpholinomethyl)-l-(phenylsulfonyl)-lH-pyrrolo [2, 3-h] pyridin-5-yl) methyl) -2, 6-difluoro-3,5-dimethoxyaniline
To a 2-L reactor equipped with a thermocouple, a nitrogen inlet and mechanical stirrer were charged AOV-dimethyl formamide (450 mL, 425 g), 4-chloro-2-(morpholinomethyl)-l-(phenylsulfonyl)- 1 //-pyrrolo|2.3-6 |pyridine-5-carbaldehyde (30.0 g, 71.45 mmol) and 2,6-difluoro-3,5-dimethoxyanihne (14.2 g, 75.0 mmol). To this suspension (internal temperature 20 °C) was added chlorotrimethylsilane (19.4 g, 22. 7 mL, 179 mmol) dropwise in 10 min at room temperature (internal temperature 20-23 °C). The suspension changed into a solution in 5 min after the chlorotrimethylsilane addition. The solution was stirred at room temperature for 1.5 h before cooled to 0-5 °C with ice-bath. Borane-THF complex in THF (1.0 M, 71.4 mL, 71.4 mmol, 64.2 g, 1.0 eq.) was added dropwise via additional funnel over 30 min while maintaining temperature at 0-5 °C. After addition, the mixture was stirred for 4 h. Water (150 g, 150 mL) was added under ice-bath cooling in 20 min, followed by slow addition of ammonium hydroxide solution (28% N¾, 15.3 g, 17 ml, 252 mmol, 3.53 eq.) to pH 9-10 while maintaining the temperature below 10 °C. More water (250 mL, 250 g) was added through the additional funnel. The slurry was stirred for 30 min and the solids were collected by filtration. The wet cake was washed with water (90 g x 2, 90 ml x 2) and heptane (61.6 g x2, 90 ml x 2). The product w as suction dried overnight to give the desired product LG-((4-chloro-2-(morphohnomethyl)-l-(phenylsulfonyl)-li/-pyrrolo[2,3-Z>]pyridin-5-yl)methyl)-2,6- difluoro-3,5-dimethoxyaniline (41.6 g, 96% yield): LCMS calculated for C27H28ClF2N405S[M+H]+: 593.10; Found: 593.1 ; ¾ NMR (400 MHz, DMSO-d6) 5 8.36 (m, 2H), 8.28 (s, 1H), 7.72 (m, 1H), 7.63 (m, 2H), 6.78 (s, 1H), 6.29 (m, 1H), 5.82 (m, 1H), 4.58 (m, 2H), 3.91 (s, 2H), 3.76 (s, 6H), 3.56 (m, 4H), 2.47 (m, 4H).
Step 4: Synthesis of l-((4-chloro-2-(morpholinomethyl)-l-(phenylsulfonyl)-lH-pyrrolo [2, 3-b ] pyridin-5-yl) methyl)-! -(2, 6-difluoro-3, 5-dimethoxyphenyl)-3-ethylurea
To a 2-L, 3-neck round bottom flask fitted with a thermocouple, a nitrogen bubbler inlet, and a magnetic stir were charged /V-((4-chloro-2-(morpholinomethyl)-l-(phenylsulfonyl)-li/-pyrrolo[2,3-b]pyridin-5-yl)methyl)-2,6-difluoro-3,5-dimethoxyaniline (67.0 g, 113 mmol) and acetonitrile (670 ml, 527 g). The suspension was cooled to 0-5 °C.
To the mixture was charged ethyl isocyanate (17.7 mL, 15.9 g, 224 mmol, 1.98 eq.) over 30 sec. The temperature stayed unchanged at 0.7 °C after the charge. Methanesulfonic acid (16.1 mL, 23.9 g, 248 mmol, 2.2 eq.) was charged dropwise over 35 min while maintaining the temperature below 2 °C. The mixture was warmed to room temperature and stirred overnight. At 24 h after addition showed that the product was 93.7%, unreacted SM was 0.73% and the major impurity (bis-isocyanate adduct) was 1.3%. The mixture was cooled with an ice-bath and quenched with sodium hydroxide (NaOH) solution (1.0M, 235 mL, 244 g, 235 mmol, 2.08 eq.) over 20 min and then saturated aqueous sodium bicarbonate
(NaHCCh) solution (1.07 M, 85 mL, 91 g, 0.091 mol, 0.80 eq.) over 10 min. Water (550 mL, 550 g) was added and the liquid became one phase. The mixture was stirred for 2 h and the solids were collected by filtration, washed with water (165 mL, 165 g) to give l-((4-chloro-2-(morpholinomethyl)- 1 -(phenylsulfonyl)- 1 //-pyrrolo| 2.3-6 |p\ ri din-5 -y l (methy l )- 1 -(2,6-difluoro-3,5-dimethoxyphenyl)-3-ethylurea ( 70.3 g, 93.7% yield).
The crude l-((4-chloro-2-(morpholinomethyl)-l -(phenylsulfonyl)- li/-pyrrolo [2, 3-61 pyridin-5-yl) methyl)- 1 -(2, 6-difluoro-3, 5-dimethoxyphenyl)-3-ethylurea (68.5 g, 103 mmol) was added in to acetonitrile (616 mL, 485 g). The mixture was heated 60-65 °C and an amber colored thin suspension was obtained. The solid was filtered off with celite and the celite was washed with acetonitrile (68.5 mL, 53.8 g). To the pale yellow filtrate was added water (685 g, 685 ml) to form a slurry. The slurry was stirred overnight at room temperature and filtered. The solid was added to water (685 mL, 685 g) and stirred at 60 °C for 2 h. The solid was filtered and re-slurred in heptane (685 mL, 469 g) overnight. The product was dried in an oven at 50 °C under vacuum for 48 h to afford l-((4-chloro-2-(morpholinomethyl)-l-(phenylsulfonyl)- 1 //-pyrrolo|2.3-6 |pyridin-5-yl)methyl)- 1 -(2.6-difluoro-3.5-
dimethoxyphenyl)-3-ethylurea as a colorless solid (62.2 g, 90.8% yield, 99.9% purity by HPLC area%). KF was 0.028%. Acetonitrile (by ‘H NMR) was about 1.56%, DCM (by ‘H NMR) 2.0%: LCMS calculated for C30H33CIF2N5O6S [M+H]+: EM: 664.17; Found: 664.2; ¾ NMR (400 MHz, DMSO-de) d 8.33 (m, 2H), 8.31 (s, 1H), 7.72 (m, 1H), 7.64 (m, 1H), 6.96 (m, 2H), 6.73 (s, 1H), 6.43 (m, 1H), 4.87 (s, 2H), 3.90 (s, 2H), 3.77 (s, 6H), 3.54 (m, 4H),
3.03 (m, 2H), 2.46 (m, 4H), 0.95 (m, 3H).
Step 5: Synthesis of 3-(2, 6-difluoro-3, 5-dimethoxyphenyl)-l-ethyl-8-(morpholin-4-ylmethyl)-7-(phenylsulfonyl)-l, 3, 4, 7-tetrahydro-2H-pyrrolo[ 3 2’:5, 6 ]pyrido[ 4, 3-d]pyrimidin-2-one
To a 2000 mL flask equipped with a thermal couple, a nitrogen inlet, and a mechanical stirrer were charged dry l-((4-chloro-2-(morpholinomethyl)-l-(phenylsulfonyl)-1 //-pyrrolo| 2.3-6 |pyridin-5-yl)methyl)- 1 -(2.6-dinuoro-3.5-dimetho\yphenyl)-3-ethylurea (30.0 g, 45.2 mmol, KF=0. l l%) and tetrahydrofuran (1200 mL, 1063 g). To this suspension at room temperature was charged 1.0 M lithium hexamethyldisilazide in THF (62.3 mL, 55.5 g, 62.3 mmol, 1.38 eq). The mixture turned into a solution after the base addition. The reaction mixture was stirred for 2 h and HPLC shows the starting material was not detectable. To this mixture was added 1.0 M hydrochloric acid (18.1 mL, -18.1 g. 18.1 mmol, 0.4 eq.). The solution was concentrated to 600 mL and water (1200 mL, 1200 g) was added. Slurry was formed after water addition. The slurry was stirred for 30 min at room temperature and the solid was collected by filtration. The wet cake was washed with water twice (60 mLx2,
60 gx2) and dried at 50 °C overnight to give 3-(2,6-difluoro-3,5-dimethoxyphenyl)-l-ethyl-8-(morpholin-4-ylmethyl)-7-(phenylsulfonyl)-l,3,4,7-tetrahydro-2H-pyrrolo[3′,2′:5,6]pyrido[4, 3-d]pyrimidin-2-one as a light brown solid (26.58 g, as-is yield 93.7%): THF by ‘H NMR 0.32%, KF 5.26%, adjusted yield was 88.5%: LCMS calculated for C30H32F2N5O6S [M+H]+: EM: 628.20; Found: 628.2; ¾ NMR (400 MHz, DMSO-de) d 8.41 (m, 2H), 8.07 (s, 1H), 7.70 (m, 1H), 7.63 (m, 2H), 7.05 (m, 1H), 6.89 (s, 1H), 4.76 (s, 2H), 4.09 (m, 2H), 3.93 (s, 2H), 3.89 (s, 6H), 3.60 (m, 4H), 2.50 (m, 4H), 1.28 (m, 3H).
Step 6: Synthesis of 3-( 2, 6-difluoro-3, 5-dimethoxyphenyl)-l-ethyl-8-(morpholin-4-ylmethyl)-1,3, 4, 7 -tetrahydro-2H-pyrrolo [ 3 ‘, 2 5, 6 ]pyrido[ 4, 3-dJpyrimidin-2-one
To a stirring suspension of 3-(2,6-difluoro-3,5-dimethoxyphenyl)-l-ethyl-8-(morpholinomethyl)-7-(phenylsulfonyl)-l,3,4,7-tetrahydro-2i/-pyrrolo[3′,2′:5,6]pyrido[4,3-d]pyrimidin-2-one (10.0 g, 15.93 mmol) in l,4-dioxane (100 ml, 103 g) in a 500 mL flask equipped with a nitrogen inlet, a condenser, a thermocouple and a heating mantle was added 1 M aqueous sodium hydroxide (63.7 ml, 66.3 g, 63.7 mmol). The reaction mixture was heated at 75 °C for 18 h. LCMS showed the reaction was complete. Water (100 mL, 100 g) was added to give a thick suspension. This slurry was stirred at room temperature for 1 h and filtered. The cake was washed with water (3 x 10 mL, 3 x 10 g) and heptane (2 x 10 mL, 2 x 6.84 g). The cake was dried overnight by pulling a vacuum through the filter cake and then dried in an oven at 50 °C under vacuum overnight to give 3-(2,6-difluoro-3,5-dimethoxyphenyl)-l-ethyl-8-(morpholin-4-ylmethyl)-l,3,4,7-tetrahydro-2H-pyrrolo[3′,2′:5, 6]pyrido[4,3-d]pyrimidin-2-one (6.8 g, 87.6% yield): LCMS calculated for C24H28F2N5O4 [M+H]+: 488.20; Found: 488.2.
PATENT
US 20130338134
https://patents.google.com/patent/US20130338134A1/en
Step 1: 3-(2,6-difluoro-3,5-dimethoxyphenyl)-1-ethyl-7-(phenylsulfonyl)-1,3,4,7-tetrahydro-2H-pyrrolo[3′,2′:5,6]pyrido[4,3-d]pyrimidin-2-one
- [0832]
- [0833]
To a solution of 3-(2,6-difluoro-3,5-dimethoxyphenyl)-1-ethyl-1,3,4,7-tetrahydro-2H-pyrrolo[3′,2′:5,6]pyrido[4,3-d]pyrimidin-2-one (Example 49, Step 3: 900 mg, 2.32 mmol) in N,N-dimethylformamide (20 mL) cooled to 0° C. was added sodium hydride (185 mg, 4.63 mmol, 60 wt % in mineral oil). The resulting mixture was stirred at 0° C. for 30 min then benzenesulfonyl chloride (0.444 mL, 3.48 mmol) was added. The reaction mixture was stirred at 0° C. for 1.5 h at which time LC-MS showed the reaction completed to the desired product. The reaction was quenched with saturated NH4Cl solution and diluted with water. The white precipitate was collected via filtration then washed with water and hexanes, dried to afford the desired product (1.2 g, 98%) as a white solid which was used in the next step without further purification. LC-MS calculated for C25H23F2N4O5S [M+H]+ m/z: 529.1; found: 529.1.
Step 2: 3-(2,6-difluoro-3,5-dimethoxyphenyl)-1-ethyl-2-oxo-7-(phenylsulfonyl)-2,3,4,7-tetrahydro-1H-pyrrolo[3′,2′:5,6]pyrido[4,3-d]pyrimidine-8-carbaldehyde
- [0834]
- [0835]
To a solution of 3-(2,6-difluoro-3,5-dimethoxyphenyl)-1-ethyl-7-(phenylsulfonyl)-1,3,4,7-tetrahydro-2H-pyrrolo[3′,2′:5,6]pyrido[4,3-d]pyrimidin-2-one (1.75 g, 3.31 mmol) in tetrahydrofuran (80 mL) at −78° C. was added freshly prepared lithium diisopropylamide (1M in tetrahydrofuran (THF), 3.48 mL, 3.48 mmol). The resulting mixture was stirred at −78° C. for 30 min then N,N-dimethylformamide (1.4 mL, 18 mmol) was added slowly. The reaction mixture was stirred at −78° C. for 30 min then quenched with water and extracted with EtOAc. The organic extracts were combined then washed with water and brine. The organic layer was dried over Na2SO4 and concentrated. The residue was purified by flash chromatography eluted with 0 to 20% EtOAc in DCM to give the desired product as a white solid (1.68 g, 91%). LC-MS calculated for C26H23F2N4O6S (M+H)+ m/z: 557.1; found: 556.9.
Step 3: 3-(2,6-difluoro-3,5-dimethoxyphenyl)-1-ethyl-8-(morpholin-4-ylmethyl)-7-(phenylsulfonyl)-1,3,4,7-tetrahydro-2H-pyrrolo[3′,2′:5,6]pyrido[4,3-d]pyrimidin-2-one
- [0836]
- [0837]
To a solution 3-(2,6-difluoro-3,5-dimethoxyphenyl)-1-ethyl-2-oxo-7-(phenylsulfonyl)-2,3,4,7-tetrahydro-1H-pyrrolo[3′,2′:5,6]pyrido[4,3-d]pyrimidine-8-carbaldehyde (1.73 g, 3.11 mmol) in dichloromethane (50 mL) was added morpholine (0.95 mL, 11 mmol), followed by acetic acid (2 mL, 30 mmol). The resulting yellow solution was stirred at room temperature overnight then sodium triacetoxyborohydride (2.3 g, 11 mmol) was added. The mixture was stirred at room temperature for 3 h at which time LC-MS showed the reaction went to completion to the desired product. The reaction was quenched with saturated NaHCO3 then extracted with ethyl acetate (EtOAc). The organic extracts were combined then washed with water and brine. The organic layer was dried over Na2SO4 and concentrated. The residue was purified by flash chromatography eluted with 0 to 40% EtOAc in DCM to give the desired product as a yellow solid (1.85 g, 95%). LC-MS calculated for C30H32F2N5O6S (M+H)+ m/z: 628.2; found: 628.0.
Step 4: 3-(2,6-difluoro-3,5-dimethoxyphenyl)-1-ethyl-8-(morpholin-4-ylmethyl)-1,3,4,7-tetrahydro-2H-pyrrolo[3′,2′:5,6]pyrido[4,3-d]pyrimidin-2-one
- [0838]
To a solution of 3-(2,6-difluoro-3,5-dimethoxyphenyl)-1-ethyl-8-(morpholin-4-ylmethyl)-7-(phenylsulfonyl)-1,3,4,7-tetrahydro-2H-pyrrolo[3′,2′:5,6]pyrido[4,3-d]pyrimidin-2-one (1.5 g, 2.4 mmol) in tetrahydrofuran (40 mL) was added tetra-n-butylammonium fluoride (1M in THF, 7.2 mL, 7.2 mmol). The resulting solution was stirred at 50° C. for 1.5 h then cooled to room temperature and quenched with water. The mixture was extracted with dichloromethane (DCM) and the organic extracts were combined then washed with water and brine. The organic layer was dried over Na2SO4 and concentrated. The residue was purified by flash chromatography eluted with 0 to 10% MeOH in DCM to give the desired product as a white solid, which was further purified by prep HPLC (pH=2, acetonitrile/H2O). LC-MS calculated for C24H28F2N5O4 (M+H)+ m/z: 488.2; found: 488.0. 1H NMR (500 MHz, DMSO) δ 12.09 (s, 1H), 8.06 (s, 1H), 7.05 (t, J=8.1 Hz, 1H), 6.87 (s, 1H), 4.78 (s, 2H), 4.50 (s, 2H), 4.17 (q, J=6.8 Hz, 2H), 3.97 (br, 2H), 3.89 (s, 6H), 3.65 (br, 2H), 3.37 (br, 2H), 3.15 (br, 2H), 1.37 (t, J=6.8 Hz, 3H).
PATENTS
| Publication Number | Title | Priority Date | Grant Date |
|---|---|---|---|
| US-2013338134-A1 | Substituted tricyclic compounds as fgfr inhibitors | 2012-06-13 | |
| US-2017137424-A1 | Substituted tricyclic compounds as fgfr inhibitors | 2012-06-13 | |
| US-2019127376-A1 | Substituted tricyclic compounds as fgfr inhibitors | 2012-06-13 | |
| US-9611267-B2 | Substituted tricyclic compounds as FGFR inhibitors | 2012-06-13 | 2017-04-04 |
| WO-2014007951-A2 | Substituted tricyclic compounds as fgfr inhibitors | 2012-06-13 |
| Publication Number | Title | Priority Date | Grant Date |
|---|---|---|---|
| JP-6336665-B2 | Substituted tricyclic compounds as FGFR inhibitors | 2012-06-13 | 2018-06-06 |
| JP-6545863-B2 | Substituted tricyclic compounds as FGFR inhibitors | 2012-06-13 | 2019-07-17 |
| JP-6711946-B2 | Substituted tricyclic compounds as FGFR inhibitors | 2012-06-13 | 2020-06-17 |
| TW-201402574-A | Substituted tricyclic compounds as FGFR inhibitors | 2012-06-13 | |
| US-10131667-B2 | Substituted tricyclic compounds as FGFR inhibitors | 2012-06-13 | 2018-11-20 |
| Publication Number | Title | Priority Date | Grant Date |
|---|---|---|---|
| JP-2015521600-A | Substituted tricyclic compounds as FGFR inhibitors | 2012-06-13 | |
| JP-2017222709-A | Substituted tricyclic compounds as FGFR inhibitors | 2012-06-13 | |
| JP-2018135377-A | Substituted tricyclic compounds as FGFR inhibitors | 2012-06-13 | |
| JP-2019178156-A | Substituted tricyclic compounds as FGFR inhibitors | 2012-06-13 | |
| JP-6301321-B2 | Substituted tricyclic compounds as FGFR inhibitors | 2012-06-13 | 2018-03-28 |
| Publication Number | Title | Priority Date | Grant Date |
|---|---|---|---|
| EP-3176170-A1 | Substituted tricyclic compounds as fgfr inhibitors | 2012-06-13 | |
| EP-3176170-B1 | Substituted tricyclic compounds as fgfr inhibitors | 2012-06-13 | 2018-11-14 |
| EP-3495367-A1 | Substituted tricyclic compounds as fgfr inhibitors | 2012-06-13 | |
| ES-2704744-T3 | Substituted tricyclic compounds as FGFR inhibitors | 2012-06-13 | 2019-03-19 |
| HU-E031916-T2 | Substituted tricyclic compounds as fgfr inhibitors | 2012-06-13 |
| Publication Number | Title | Priority Date | Grant Date |
|---|---|---|---|
| DK-2861595-T5 | Substituted tricyclic compounds as FGFR inhibitors | 2012-06-13 | 2018-01-15 |
| DK-3176170-T3 | Substituted tricyclic relations as fgfr inhibitors | 2012-06-13 | 2019-01-28 |
| EP-2861595-A2 | Substituted tricyclic compounds as fgfr inhibitors | 2012-06-13 | |
| EP-2861595-B1 | Substituted tricyclic compounds as fgfr inhibitors | 2012-06-13 | 2016-12-21 |
| EP-2861595-B9 | Substituted tricyclic compounds as fgfr inhibitors | 2012-06-13 | 2017-06-21 |
| Publication Number | Title | Priority Date | Grant Date |
|---|---|---|---|
| WO-2019191707-A1 | Heterocyclic compounds as immunomodulators | 2018-03-30 | |
| AU-2013287176-A1 | Substituted tricyclic compounds as FGFR inhibitors | 2012-06-13 | |
| CA-2876689-A1 | Substituted tricyclic compounds as fgfr inhibitors | 2012-06-13 | |
| CN-107383009-B | Substituted tricyclic compounds as FGFR inhibitors | 2012-06-13 | 2020-06-09 |
| DK-2861595-T3 | Substituted tricyclic compounds as fgfr inhibitors | 2012-06-13 | 2017-02-13 |
| Publication Number | Title | Priority Date | Grant Date |
|---|---|---|---|
| WO-2019213544-A2 | Solid forms of an fgfr inhibitor and processes for preparing the same | 2018-05-04 | |
| WO-2019213544-A3 | Solid forms of an fgfr inhibitor and processes for preparing the same | 2018-05-04 | |
| TW-202003511-A | Heterocyclic compounds as immunomodulators | 2018-03-30 | |
| US-10669271-B2 | Heterocyclic compounds as immunomodulators | 2018-03-30 | 2020-06-02 |
| US-2019300524-A1 | Heterocyclic compounds as immunomodulators | 2018-03-30 |
| Publication Number | Title | Priority Date | Grant Date |
|---|---|---|---|
| TW-201946630-A | Salts of an FGFR inhibitor | 2018-05-04 | |
| TW-202003516-A | Solid forms of an FGFR inhibitor and processes for preparing the same | 2018-05-04 | |
| US-2019337948-A1 | Solid forms of an fgfr inhibitor and processes for preparing the same | 2018-05-04 | |
| US-2020002338-A1 | Salts of an fgfr inhibitor | 2018-05-04 | |
| WO-2019213506-A1 | Salts of an fgfr inhibitor | 2018-05-04 |
| Publication Number | Title | Priority Date | Grant Date |
|---|---|---|---|
| WO-2019227007-A1 | Tricyclic heterocyclic compounds as sting activators | 2018-05-25 | |
| TW-201946626-A | Heterocyclic compounds as immunomodulators | 2018-05-11 | |
| US-10618916-B2 | Heterocyclic compounds as immunomodulators | 2018-05-11 | 2020-04-14 |
| US-2019345170-A1 | Heterocyclic compounds as immunomodulators | 2018-05-11 | |
| WO-2019217821-A1 | Tetrahydro-imidazo[4,5-c]pyridine derivatives as pd-l1 immunomodulators | 2018-05-11 |
| Publication Number | Title | Priority Date | Grant Date |
|---|---|---|---|
| US-2020040009-A1 | Tricyclic heteraryl compounds as sting activators | 2018-07-31 | |
| WO-2020028565-A1 | Tricyclic heteraryl compounds as sting activators | 2018-07-31 | |
| WO-2020028566-A1 | Heteroaryl amide compounds as sting activators | 2018-07-31 | |
| WO-2019238873-A1 | A method of precision cancer therapy | 2018-06-13 | |
| US-2019359608-A1 | Tricyclic heterocyclic compounds as sting activators | 2018-05-25 |
| Title | Priority Date | Grant Date | |
|---|---|---|---|
| WO-2020131627-A1 | Substituted pyrazolo[1,5-a]pyridine compounds as inhibitors of fgfr tyrosine kinases | 2018-12-19 | |
| WO-2020131674-A1 | 7-((3,5-dimethoxyphenyl)amino)quinoxaline derivatives as fgfr inhibitors for treating cancer | 2018-12-19 | |
| WO-2020081898-A1 | Non-invasive urinary biomarkers for the detection of urothelial carcinoma of the bladder | 2018-10-20 | |
| US-2020115378-A1 | Dihydropyrido[2,3-d]pyrimidinone compounds as cdk2 inhibitors | 2018-10-11 | |
| US-2020039994-A1 | Heteroaryl amide compounds as sting activators | 2018-07-31 |
References
- ^ “Pemigatinib (Pemazyre) Use During Pregnancy”. Drugs.com. 11 August 2020. Retrieved 24 September 2020.
- ^ World Health Organization (2018). “International nonproprietary names for pharmaceutical substances (INN): recommended INN: list 80”. WHO Drug Information. 32 (3): 479. hdl:10665/330907.
- ^ Jump up to:a b c d e f g h i j k l m n o “FDA Approves First Targeted Treatment for Patients with Cholangiocarcinoma, a Cancer of Bile Ducts”. U.S. Food and Drug Administration (FDA) (Press release). 17 April 2020. Retrieved 17 April 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain. - ^ Jump up to:a b c d e f g h “FDA grants accelerated approval to pemigatinib for cholangiocarcinoma”. U.S. Food and Drug Administration (FDA). 17 April 2020. Retrieved 20 April 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain. - ^ Jump up to:a b c d e “EU/3/18/2066”. European Medicines Agency (EMA). 19 December 2018. Retrieved 20 April 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain. - ^ Jump up to:a b c d “Drug Trials Snapshot: Pemazyre”. U.S. Food and Drug Administration (FDA). 17 April 2020. Retrieved 5 May 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain. - ^ “Pemazyre: FDA-Approved Drugs”. U.S. Food and Drug Administration (FDA). Retrieved 21 April 2020.
- ^ “Pemigatinib Orphan Drug Designation and Approval”. U.S. Food and Drug Administration (FDA). Retrieved 19 April 2020.
- ^ “Pemigatinib Orphan Drug Designation and Approval”. U.S. Food and Drug Administration (FDA). Retrieved 19 April 2020.
- ^ “EU/3/19/2216”. European Medicines Agency (EMA). 23 January 2020. Retrieved 19 April 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
Further reading
- Roskoski R (January 2020). “The role of fibroblast growth factor receptor (FGFR) protein-tyrosine kinase inhibitors in the treatment of cancers including those of the urinary bladder”. Pharmacol. Res. 151: 104567. doi:10.1016/j.phrs.2019.104567. PMID 31770593.
External links
- “Pemigatinib”. Drug Information Portal. U.S. National Library of Medicine.
- “Pemigatinib”. National Cancer Institute.
- Clinical trial number NCT02924376 for “Efficacy and Safety of Pemigatinib in Subjects With Advanced/Metastatic or Surgically Unresectable Cholangiocarcinoma Who Failed Previous Therapy – (FIGHT-202)” at ClinicalTrials.gov
| Clinical data | |
|---|---|
| Trade names | Pemazyre |
| Other names | INCB054828 |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a620028 |
| License data | US DailyMed: Pemigatinib |
| Pregnancy category | US: N (Not classified yet)[1] |
| Routes of administration | By mouth |
| ATC code | None |
| Legal status | |
| Legal status | US: ℞-only |
| Identifiers | |
| IUPAC name[show] | |
| CAS Number | 1513857-77-6 |
| PubChem CID | 86705695 |
| DrugBank | DB15102 |
| ChemSpider | 68007304 |
| UNII | Y6BX7BL23K |
| KEGG | D11417 |
| ChEMBL | ChEMBL4297522 |
| Chemical and physical data | |
| Formula | C24H27F2N5O4 |
| Molar mass | 487.508 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| SMILES[hide]CCN1C2=C3C=C(NC3=NC=C2CN(C1=O)C4=C(C(=CC(=C4F)OC)OC)F)CN5CCOCC5 | |
| InChI[hide]InChI=1S/C24H27F2N5O4/c1-4-30-21-14(11-27-23-16(21)9-15(28-23)13-29-5-7-35-8-6-29)12-31(24(30)32)22-19(25)17(33-2)10-18(34-3)20(22)26/h9-11H,4-8,12-13H2,1-3H3,(H,27,28)Key:HCDMJFOHIXMBOV-UHFFFAOYSA-N |
/////////Pemigatinib, 佩米替尼 , PEMAZYRE, FDA 2020, 2020 APPROVALS, INCB054828, INCB 054828, Orphan Drug Status, Myeloproliferative disorders, Lymphoma, Cholangiocarcinoma, INCYTE
O=C1N(CC)C2=C3C(NC(CN4CCOCC4)=C3)=NC=C2CN1C5=C(F)C(OC)=CC(OC)=C5F.[H]Cl

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....










