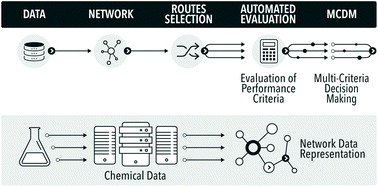
Towards automation of chemical process route selection based on data mining
A methodology for chemical routes development and evaluation on the basis of data-mining is presented. A section of the Reaxys database was converted into a network, which was used to plan hypothetical synthesis routes to convert a bio-waste feedstock, limonene, to a bulk intermediate, benzoic acid. The route evaluation considered process conditions and used multiple indicators, including exergy, E-factor, solvent score, reaction reliability and route redox efficiency, in a multi-criteria environmental sustainability evaluation. The proposed methodology is the first route evaluation based on data mining, explicitly using reaction conditions, and is amenable to full automation.
In the field of process and synthetic chemistry ‘clean synthesis’ has become one of the standard criteria for good, commercially viable synthesis routes. As a result synthetic and process chemists must be equipped with adequate methodologies for quantification of ‘cleanness’ or ‘greenness’ of alternative routes at the early phases of the development cycle. These…
View original post 1,252 more words
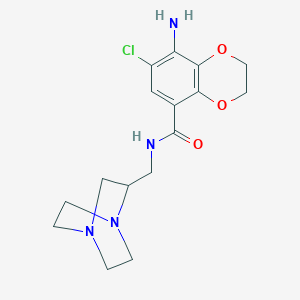
SL65.0102-10
CAS 186348-69-6
1,4-Benzodioxin-5-carboxamide, 8-amino-7-chloro-N-(1,4-diazabicyclo[2.2.2]oct-2-ylmethyl)-2,3-dihydro-, (-)-
- MW, 352.82, C16 H21 Cl N4 O3
-
US5663173 (A) – N-[(1,4-diazabicyclo[2.2.2] oct-2-yl)methyl] benzamide derivatives, their preparations and their application in therapeutics

SL65.0102-10
(-)-1,4-benzodioxin-5-carboxamide-8-amino-7-chloro-N-(1,4-diazabicyclo[2.2.2]oct-2- ylmethyl)-2,3-dihydro-, hydrochloride
CAS 186348-31-2, C16 H21 Cl N4 O3 . 2 Cl H
Melting point: 220 ° C. (decomposition). EP0748807
[α] ![]() = -16.9 ° (c = 1, H 2 O).
= -16.9 ° (c = 1, H 2 O).
[α]D = -17.9 (C = 0.75, DMSO, t = 23°C) at 589 nm. DOI: 10.1021/acs.oprd.6b00262
5-HT3 and 5-HT4 inhibitor that was potentially useful for the treatment of neurological disorders.
Innovators-sanofi

Hoechst Marion Roussel (Sanofi) my organisation 1993-1997 Process development at Mulund, Mumbai, India.
CENTRE IS DR RALPH STAPEL, HEAD PROCESS DEVELOPMENT, SANOFI
The 5-HT4 receptor is a G-protein coupled receptor (GPCR) which belongs to the serotonin receptor family. The role of the 5-HT4 receptor in the modulation of many diseases is well described in the literature.(1)
During the last decades, an impressive body of evidence suggested that selective stimulation of neuronal 5-HT4 receptor subtypes could be beneficial in the symptomatic treatment of memory disorders, including many antidepressants, antipsychotics, anorectics, antiemetics, gastroprokinetic agents, antimigraine agents, hallucinogens, and antactogens.(2)
Within effort to discover treatments of memory dysfunction, SL65.0102-10, a selective 5-HT4 partial agonist (Ki 6.6 μM), was discovered as promising agent for the treatment of cognition impairment. Serotonin receptors are the target of a variety of pharmaceutical drugs; SL65.0102-10 emerged as a promising 5-HT3 and 5-HT4 inhibitor that was potentially useful for the treatment of neurological disorders.(3)

Samir Jegham
Lead Generation Senior Advisor for Asia Pacific Research Hub at Sanofi
“DRUG APPROVALS INT” CATERS TO EDUCATION GLOBALLY, No commercial exploits are done or advertisements added by me. This article is a compilation for educational purposes only.
P.S. : The views expressed are my personal and in no-way suggest the views of the professional body or the company that I represent
SYNTHESIS
SL65.0102-10
CONTD…………..

Synthesis

PATENT
(EP0748807) Derivatives of N- (1,4-diazabicyclo (2.2.2) -oct-2-yl) methyl benzamide, their preparation and their therapeutic use


Example 5 (Compound No. 9)
Ethyl (-) – 8-Amino-7-chloro- N – [(1,4-diazabicyclo [2.2.2] oct-2-yl) methyl] -2,3-dihydro-1,4 Benzodioxin-5-carboxamide.
5.1. (+) – (2,2-dimethyl-1,3-dioxolan-4-yl) methyl methanesulfonate.
The procedure described in Example 4.1, but from (+) – 2,2-dimethyl-1,3-dioxolane-4-methanol.
5.2. (-) – 2 – [(2,2-Dimethyl-1,3-dioxolan-4-yl) methyl] -1 H -isoindole-1,3 (2 H ) -dione.
The procedure described in Example 4.2, from methane sulfonate (+) – (2,2-dimethyl-1,3-dioxolan-4-yl) methyl.
Melting point: 81.2-81.3 ° C.
[α]![]() = -34.9 ° (c = 1, CH 2 Cl 2 ).
= -34.9 ° (c = 1, CH 2 Cl 2 ).
5.3. (-) – 2- (2,3-dihydroxypropyl) -1 H -isoindole-1,3 (2 H ) -dione.
The procedure described in Example 4.3, from (-) – 2 – [(2,2-dimethyl-1,3-dioxolan-4-yl) methyl] -1 H -isoindole-1, 3 (2 H ) -dione.
Melting point: 122.8-122.9 ° C.
[α]![]() = -48.8 ° (c = 1, CH 3 OH).
= -48.8 ° (c = 1, CH 3 OH).
5.4. (-) – 2 – [(2-Phenyl-1,3-dioxolan-4-yl) methyl] -1 H -isoindole-1,3 (2 H ) -dione.
The procedure described in Example 4.4, from (-) – 2- (2,3-dihydroxypropyl) -1 H -isoindole-1,3 (2 H ) -dione.
Melting point: 84 ° C.
[α]![]() = -59 ° (c = 1, CH 2 Cl 2 ).
= -59 ° (c = 1, CH 2 Cl 2 ).
5.5. Benzoate (-) – 2-bromomethyl-1- (1,3-dihydro-1,3-dioxo-2 H -isoindol-2-yl) ethyl.
The procedure described in Example 4.5, from (-) – 2 – [(2-phenyl-1,3-dioxolan-4-yl) methyl] -1 H -isoindole-1,3 ( 2 H ) -dione.
Melting point: 118.4-118.6 ° C.
[α]![]() = -58.2 ° (c = 1, CH 2 Cl 2 ).
= -58.2 ° (c = 1, CH 2 Cl 2 ).
5.6. (+) – 2- (oxiranylmethyl) -1 H -isoindole-1,3 (2 H ) -dione. Fusion point :
The procedure described in Example 4.6, from benzoate (-) – 2-bromomethyl-1- (1,3-dihydro-1,3-dioxo-2 H -isoindol-2-yl) ethyl.
Melting point: 100.4-100.5 ° C.
[α]![]() = + 45.5 ° (c = 1, CHCl 3 ).
= + 45.5 ° (c = 1, CHCl 3 ).
5.7. Dihydrochloride (-) – 8-Amino-7-chloro- N – [(1,4-diazabicyclo [2.2.2] oct-2-yl) methyl] -2,3-dihydro-1,4-benzodioxin-5 -carboxamide.
The procedure described in Example 4.7, from (+) – 2- (oxiranylmethyl) -1 H -isoindole-1,3 (2 H ) -dione.
Melting point: 220 ° C. (decomposition).
[α] ![]() = -16.9 ° (c = 1, H 2 O).
= -16.9 ° (c = 1, H 2 O).
Paper
The process development and improvements for route selection, adapted to large scale for the pilot-scale preparation of SL65.0102-10, an N-diazabicyclo[2.2.2]-octylmethyl benzamide, a 5-HT3and 5-HT4 receptor active ligand for the treatment of neurological disorders such as cognition impairment, are described in this article. Notable steps and enhancements are compared to the original route, including the improvement of a chiral epoxide synthesis by shortening the number of chemical steps, the deprotection of a quaternary ammonium salt, and the redesign of the final amidification coupling to avoid chromatography.
Pilot Scale Process Development of SL65.0102-10, an N-Diazabicyclo[2.2.2]-octylmethyl Benzamide
(-)-1,4-benzodioxin-5-carboxamide-8-amino-7-chloro-N-(1,4-diazabicyclo[2.2.2]oct-2- ylmethyl)-2,3-dihydro-, hydrochloride (1:2), SL65.0102-10 (1).
……………….. to provide compound 1 (10.3 kg, 76.7%). Compound 1 could be recrystallized in acetone/water (12/2 volumes).
1H-NMR (DMSO-d6, 500 MHz), δ ppm: 3.38 (dd, 1H, J = 12.0 , 6.0 Hz), 3.60-3.45 (m, 7H), 3.65 (t, 1H, J =10.0 Hz), 3.72 (dt, 1H, J =6.0 , 14.0 Hz), 3.83 (m, 2H), 4.01 (m, 1H), 4.33 (m, 2H), 4.39 (m, 2H), 7.37 (s, 1H), 8.35 (t, 1H, J =6.0 Hz). Only 19 protons are observed on 1H spectrum instead of 21 expected. The two amino protons of the molecule are not visible because of chemical exchange with residual water of DMSO-d6 solvent.
13C NMR (DMSO-d6, 125 MHz): δ 38.4, 39.0, 42.8, 43.4, 45.4, 46.5, 54.4, 64.1, 65.1, 109.3, 110.0, 123.2, 130.5, 138.1, 141.8, 165.0.
HRMS: exact mass (by Xevo QToF), MH+ found: 353.1374 (MH+ calculated: 353.1380, difference: -1.7 ppm).
[α]D = -17.9 (C = 0.75, DMSO, t = 23°C) at 589 nm.
Elementary analysis: found C 43.0660%, H 5.5150%, N 12.4792%, calculated C 43.31%, H 5.68%, N 12.63%

1H AND 13C NMR PREDICT
References
-
(a) Hoyer, D.; Clarke, D. E.; Fozard, J. R.; Hartig, P. R.; Martin, G. R.; Mylecharane, E. J.; Saxena, P. R.;Humphrey, P. P. Pharmacol. Rev. 1994, 46 ( 2) 157– 203
(b) Frazer, A.; Hensler, J. G.Chapter 13: Serotonin Receptors. In Siegel, G. J.; Agranoff, B. W.; Albers, R. W.; Fisher, S. K.; Uhler, M. D., Eds.; Basic Neurochemistry: Molecular, Cellular, and Medical Aspects; Lippincott-Raven, Philadelphia,1999; pp 263– 292.
-
Frick, W.; Glombik, H.; Kramer, W.; Heuer, H.; Brummerhop, H.; Plettenburg, O. Novel fluoroglycoside heterocyclic derivatives, pharmaceutical products containing said compounds and the use thereof.
(a) WO2004/052903, 2004.
(b) WO2004/052902, 2004.
-
Jegham, S.; Koenig, J. J.; Lochead, A.; Nedelec, A.; Guminski, Y.N-[(1,4-diazabicyclo[2.2.2]oct-2-yl)methyl] benzamide derivatives, their preparations and their application in therapeutics.
(a) FR 2756563 06/13/1995 9506951, 1995.
(b) US 5663173, 1997; Washington, DC: U.S. Patent and Trademark Office.
“DRUG APPROVALS INT” CATERS TO EDUCATION GLOBALLY, No commercial exploits are done or advertisements added by me. This article is a compilation for educational purposes only.
P.S. : The views expressed are my personal and in no-way suggest the views of the professional body or the company that I represent
////////SL65.0102-10, SANOFI, 5-HT3 , 5-HT4 inhibitor, neurological disorders
O=C(NCC2CN1CCN2CC1)c4cc(Cl)c(N)c3OCCOc34
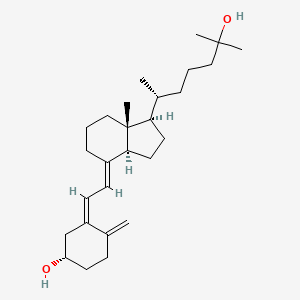
Calcifediol, カルシフェジオール

Calcifediol
カルシフェジオール
| Molecular form.: | C₂₇H₄₄O₂ |
| Appearance: | White to Off-White Solid |
| Melting Point: | 75-93ºC |
| Mol. Weight: | 400.64 |
Calcifediol (INN), also known as calcidiol, 25-hydroxycholecalciferol, or 25-hydroxyvitamin D (abbreviated 25(OH)D),[1] is a prehormone that is produced in the liver by hydroxylation of vitamin D3 (cholecalciferol) by the enzyme cholecalciferol 25-hydroxylase which was isolated by Michael F. Holick. Physicians worldwide measure this metabolite to determine a patient’s vitamin D status.[2] At a typical daily intake of vitamin D3, its full conversion to calcifediol takes approximately 7 days.[3]
Calcifediol is then converted in the kidneys (by the enzyme 25(OH)D-1α-hydroxylase) into calcitriol (1,25-(OH)2D3), a secosteroid hormone that is the active form of vitamin D. It can also be converted into 24-hydroxycalcidiol in the kidneys via 24-hydroxylation.[4][5]
Blood test
In medicine, a 25-hydroxy vitamin D (calcifediol) blood test is used to determine how much vitamin D is in the body.[6] The blood concentration of calcifediol is considered the best indicator of vitamin D status.[7]
This test can be used to diagnose vitamin D deficiency, and it is indicated in patients with high risk for vitamin D deficiency and when the results of the test would be used as supporting evidence for beginning aggressive therapies.[8] Patients with osteoporosis, chronic kidney disease, malabsorption, obesity, and some other infections may be high risk and thus have greater indication for this test.[8] Although vitamin D deficiency is common in some populations including those living at higher latitudes or with limited sun exposure, the 25(OH)D test is not indicated for entire populations.[8] Physicians may advise low risk patients to take over-the-counter vitamin D in place of having screening.[8]
It is the most sensitive measure,[9] though experts have called for improved standardization and reproducibility across different laboratories.[7] According to MedlinePlus, the normal range of calcifediol is 30.0 to 74.0 ng/mL.[6] The normal range varies widely depending on several factors, including age and geographic location. A broad reference range of 20–150 nmol/L (8-60 ng/ml) has also been suggested,[10] while other studies have defined levels below 80 nmol/L (32 ng/ml) as indicative of vitamin D deficiency.[11]
US labs generally report 25(OH)D levels as ng/mL. Other countries often use nmol/L. Multiply ng/mL by 2.5 to convert to nmol/L.
Clinical significance
Increasing calcifediol levels are associated with increasing fractional absorption of calcium from the gut up to levels of 80 nmol/L (32 ng/mL).[citation needed]Urinary calcium excretion balances intestinal calcium absorption and does not increase with calcifediol levels up to ~400 nmol/L (160 ng/mL).[12]
A study by Cedric F. Garland and Frank C. Garland of the University of California, San Diego analyzed the blood from 25,000 volunteers from Washington County, Maryland, finding that those with the highest levels of calcifediol had a risk of colon cancer that was one-fifth of typical rates.[13] However, randomized controlled trials failed to find a significant correlation between vitamin D supplementation and the risk of colon cancer.[14]
A 2012 registry study of the population of Copenhagen, Denmark, found a correlation between both low and high serum levels and increased mortality, with a level of 50–60 nmol/L being associated with the lowest mortality. The study did not show causation.[15][16]
Nmr
Regioselective Hydroxylation in the Production of 25-Hydroxyvitamin D by Coprinopsis cinerea Peroxygenase
ChemCatChem (2015), 7, (2), 283-290
1H NMR 500 MHz, CDCl3: δ= 0.55 (3 H, s, 18-H), 0.94 (1H, d, J= 6.5 Hz, 21-H), 1.06 (1H, m, 22-H), 1.22 (3 H, s, 26-H), 1.22 (3 H, s, 27-H), 1.23 (1H, m, 23-H), 1.27 (1H, m, 16-H), 1.28 (1H, m, 14-H), 1.29 (1H, m, 12-H), 1.37 (1H, m, 22-H), 1.38 (1H, m, 20-H), 1.39 (1H, m, 24-H), 1.42 (1H, m, 23-H), 1.44 (1H, m, 24-H), 1.47 (2 H, m, 11-H), 1.53 (1H, m, 15-H), 1.66 (1H, m, 15-H), 1.67 (1H, m, 2-H), 1.67 (1H, m, 9-H), 1.87 (1H, m, 16-H), 1.92 (1H, m, 2-H), 1.98 (1H, m, 17-H), 2.06 (1H, m, 12-H), 2.17 (1H, m, 1-H), 2.40 (1H, m, 1-H), 2.57 (1H, dd, J= 3.7, 13.1Hz, 4-H), 2.82 (1H, m, 9-H), 3.95 (1H, bm, 3-H), 4.82 (1H, m, 19-H), 5.05 (1H, m, 19-H), 6.03 (1H, d, J=11.2 Hz, 7-H), 6.23 ppm (1H, d, J= 11.2 Hz, 6-H).
13 C NMR 500 MHz, CDCl3: δ = 12.2 (C-18), 19.0 (C-21), 21.0 (C-23), 22.4 (C-11), 23.7 (C-15), 27.8 (C-16), 29.2 (C-9), 29.4 (C-27), 29.5 (C-26), 32.1 (C-1), 35.3 (C-2), 36.3 (C-20), 36.6 (C-22), 40.7 (C-12), 44.6 (C-24), 46.0 (C-13), 46.1 (C-4), 56.5 (C-17), 56.7 (C-14), 69.4 (C-3), 71.3 (C-25), 112.6 (C-19), 117.7 (C-7), 122.2 (C-6), 135.2 (C-5), 142.4 (C-8), 145.3 ppm (C-10).
PAPER
From Organic & Biomolecular Chemistry, 10(27), 5205-5211; 2012
http://pubs.rsc.org/en/content/articlelanding/2012/ob/c2ob25511a#!divAbstract
An efficient, two-stage, continuous-flow synthesis of 1α,25-(OH)2-vitamin D3 (activated vitamin D3) and its analogues was achieved. The developed method afforded the desired products in satisfactory yields using a high-intensity and economical light source, i.e., a high-pressure mercury lamp. In addition, our method required neither intermediate purification nor high-dilution conditions.
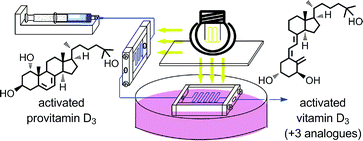
1H NMR(400 MHz, CDCl3): δ 8.13 (m, 2H), 7.68 (m, 2H), 6.64 (d, J = 8.3 Hz, 1H), 6.25 (d, J = 8.3 Hz, 1H), 5.19 (m, 2H), 3.93 (dd, J = 12.7, 8.2, 1H), 3.88 (dd, J = 14.6, 4.9 Hz, 1H), 3.58 (m, 1H), 1.02 (s, 3H), 1.02 (d, J = 6.8 Hz, 3H), 0.90 (d, J = 6.8 Hz, 3H), 0.86 (s, 9H), 0.80-0.84 (m, 9H), 0.09 (s, 3H), 0.00 (s, 3H)
13C NMR (100 MHz, CDCl3): δ 161.8, 159.6, 138.5, 135.3, 132.6, 132.5, 132.1, 130.6, 130.2, 128.7, 127.0, 126.5, 77.2, 68.5, 67.4, 67.1, 56.5, 50.6, 49.0, 44.2, 42.7, 40.4, 39.9, 39.3, 35.6, 34.7, 33.0, 30.5, 28.2, 25.9, 24.5, 21.9, 20.8, 19.9, 19.7, 18.5, 18.0, 17.4, 13.3, -4.4, -4.9
| IR (neat): 2957, 2872, 1653, 1603, 1462, 1311, 1093, 837, 762 cm-1 |
Interactive pathway map
Click on genes, proteins and metabolites below to link to respective articles. [§ 1]
Vitamin D Synthesis Pathway edit
- Jump up^ The interactive pathway map can be edited at WikiPathways: “VitaminDSynthesis_WP1531”.
References
- Jump up^ “Nomenclature of Vitamin D. Recommendations 1981. IUPAC-IUB Joint Commission on Biochemical Nomenclature (JCBN)” reproduced at the Queen Mary, University of London website. Retrieved 21 March 2010.
- Jump up^ Holick, MF; Deluca, HF; Avioli, LV (1972). “Isolation and identification of 25-hydroxycholecalciferol from human plasma”. Archives of Internal Medicine. 129 (1): 56–61. doi:10.1001/archinte.1972.00320010060005. PMID 4332591.
- Jump up^ Am J Clin Nutr 2008;87:1738–42 PMID 18541563
- Jump up^ Bender, David A.; Mayes, Peter A (2006). “Micronutrients: Vitamins & Minerals”. In Victor W. Rodwell; Murray, Robert F.; Harper, Harold W.; Granner, Darryl K.; Mayes, Peter A. Harper’s Illustrated Biochemistry. New York: Lange/McGraw-Hill. pp. 492–3. ISBN 0-07-146197-3. Retrieved December 10, 2008 through Google Book Search.
- Jump up^ Institute of Medicine (1997). “Vitamin D”. Dietary Reference Intakes for Calcium, Phosphorus, Magnesium, Vitamin D, and Fluoride. Washington, D.C: National Academy Press. p. 254. ISBN 0-309-06403-1.
- ^ Jump up to:a b “25-hydroxy vitamin D test: Medline Plus”. Retrieved 21 March 2010.
- ^ Jump up to:a b Heaney, Robert P (Dec 2004). “Functional indices of vitamin D status and ramifications of vitamin D deficiency”. American Journal of Clinical Nutrition. 80 (6): 1706S–9S. PMID 15585791.
- ^ Jump up to:a b c d American Society for Clinical Pathology, “Five Things Physicians and Patients Should Question”, Choosing Wisely: an initiative of the ABIM Foundation, American Society for Clinical Pathology, retrieved August 1, 2013, which cites
-
- Sattar, N.; Welsh, P.; Panarelli, M.; Forouhi, N. G. (2012). “Increasing requests for vitamin D measurement: Costly, confusing, and without credibility”. The Lancet. 379 (9811): 95–96. doi:10.1016/S0140-6736(11)61816-3. PMID 22243814.
- Bilinski, K. L.; Boyages, S. C. (2012). “The rising cost of vitamin D testing in Australia: Time to establish guidelines for testing”. The Medical Journal of Australia. 197 (2): 90. doi:10.5694/mja12.10561. PMID 22794049.
- Lu, Chuanyi M. (May 2012). “Pathology consultation on vitamin D testing: Clinical indications for 25(OH) vitamin D measurement [Letter to the editor]”. American Journal Clinical Pathology. American Society for Clinical Pathology (137): 831–832., which cites
- Arya, S. C.; Agarwal, N. (2012). “Pathology Consultation on Vitamin D Testing: Clinical Indications for 25(OH) Vitamin D Measurement”. American Journal of Clinical Pathology. 137 (5): 832. doi:10.1309/AJCP2GP0GHKQRCOE. PMID 22523224.
- Holick, M. F.; Binkley, N. C.; Bischoff-Ferrari, H. A.; Gordon, C. M.; Hanley, D. A.; Heaney, R. P.; Murad, M. H.; Weaver, C. M. (2011). “Evaluation, Treatment, and Prevention of Vitamin D Deficiency: An Endocrine Society Clinical Practice Guideline”. Journal of Clinical Endocrinology & Metabolism. 96 (7): 1911–1930. doi:10.1210/jc.2011-0385. PMID 21646368.
-
- Jump up^ Institute of Medicine (1997), p. 259
- Jump up^ Bender, David A. (2003). “Vitamin D”. Nutritional biochemistry of the vitamins. Cambridge: Cambridge University Press. ISBN 0-521-80388-8. Retrieved December 10, 2008 through Google Book Search.
- Jump up^ Hollis BW (February 2005). “Circulating 25-hydroxyvitamin D levels indicative of vitamin D sufficiency: implications for establishing a new effective dietary intake recommendation for vitamin D”. J Nutr. 135 (2): 317–22. PMID 15671234.
- Jump up^ Kimball; et al. (2004). “Safety of vitamin D3 in adults with multiple sclerosis”. J Clin Endocrinol Metab. 86 (3): 645–51. PMID 17823429.
- Jump up^ Maugh II, Thomas H. “Frank C. Garland dies at 60; epidemiologist helped show importance of vitamin D: Garland and his brother Cedric were the first to demonstrate that vitamin D deficiencies play a role in cancer and other diseases.”, Los Angeles Times, August 31, 2010. Accessed September 4, 2010.
- Jump up^ Wactawski-Wende, J; Kotchen, JM, Women’s Health Initiative Investigators (Mar 9, 2006). “Calcium plus vitamin D supplementation and the risk of colorectal cancer.”. N Engl J Med. 354 (7): 684–96. doi:10.1056/NEJMoa055222. PMID 16481636. Retrieved December 28, 2013.
- Jump up^ “Too much vitamin D can be as unhealthy as too little” (Press release). University of Copenhagen. May 29, 2012. Retrieved 2015-05-27.
- Jump up^ Durup, D.; Jørgensen, H. L.; Christensen, J.; Schwarz, P.; Heegaard, A. M.; Lind, B. (May 9, 2012). “A Reverse J-Shaped Association of All-Cause Mortality with Serum 25-Hydroxyvitamin D in General Practice: The CopD Study”. The Journal of Clinical Endocrinology & Metabolism. Endocrine Society. 97 (8): 2644–2652. doi:10.1210/jc.2012-1176. Retrieved 2015-05-27.
 |
|
 |
|
| Names | |
|---|---|
| IUPAC names
(6R)-6-[(1R,3aR,4E,7aR)-4-[(2Z)-2-[(5S)-5-
Hydroxy-2-methylidene-cyclohexylidene] ethylidene]-7a-methyl-2,3,3a,5,6,7-hexahydro- 1H-inden-1-yl]-2-methyl-heptan-2-ol |
|
| Other names
25-Hydroxyvitamin D3
25-Hydroxycholecalciferol Calcidiol |
|
| Identifiers | |
| 19356-17-3 |
|
| 3D model (Jmol) | Interactive image |
| ChEBI | CHEBI:17933 |
| ChEMBL | ChEMBL1222 |
| ChemSpider | 4446820 |
| DrugBank | DB00146 |
| ECHA InfoCard | 100.039.067 |
| 6921 | |
| MeSH | Calcifediol |
| PubChem | 5283731 |
| UNII | T0WXW8F54E |
| Properties | |
| C27H44O2 | |
| Molar mass | 400.64 g/mol |
| Pharmacology | |
| A11CC06 (WHO) | |
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
|
|
/////////Calcifediol, カルシフェジオール
CC(CCCC(C)(C)O)C1CCC2C1(CCCC2=CC=C3CC(CCC3=C)O)C
GMP’s for Early Stage Development of new Drug substances and products
DRUG REGULATORY AFFAIRS INTERNATIONAL
GMP’s for Early Stage Development of New Drug substances and products
View original post 1,741 more words
Balsalazide

Balsalazide
| 80573-04-2; Colazal; Balsalazide Disodium; AC1NSFNR; P80AL8J7ZP; | |
| Molecular Formula: | C17H15N3O6 |
|---|---|
| Molecular Weight: | 357.322 g/mol |
(3E)-3-[[4-(2-carboxyethylcarbamoyl)phenyl]hydrazinylidene]-6-oxocyclohexa-1,4-diene-1-carboxylic acid
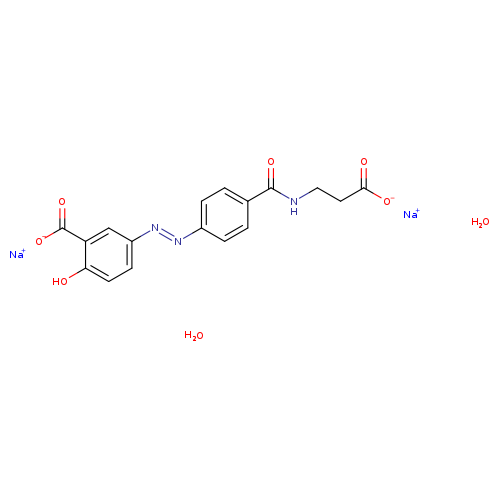 DISODIUMDIHYDRATE
DISODIUMDIHYDRATE
| CAS Number | 150399-21-6 |
|---|---|
| Weight | Average: 437.316 Monoisotopic: 437.08110308 |
| Chemical Formula | C17H17N3Na2O8 |
Balsalazide is an anti-inflammatory drug used in the treatment of inflammatory bowel disease. It is sold under the brand names Giazo, Colazal in the US and Colazide in the UK. It is also sold in generic form in the US by several generic manufacturers.
It is usually administered as the disodium salt. Balsalazide releases mesalazine, also known as 5-aminosalicylic acid, or 5-ASA,[1] in the large intestine. Its advantage over that drug in the treatment of ulcerative colitis is believed to be the delivery of the active agent past the small intestine to the large intestine, the active site of ulcerative colitis.
Balsalazide is an anti-inflammatory drug used in the treatment of Inflammatory Bowel Disease. It is sold under the name “Colazal” in the US and “Colazide” in the UK. The chemical name is (E)-5-[[-4-(2-carboxyethyl) aminocarbonyl] phenyl]azo] –2-hydroxybenzoic acid. It is usually administered as the disodium salt. Balsalazide releases mesalazine, also known as 5-aminosalicylic acid, or 5-ASA, in the large intestine. Its advantage over that drug in the treatment of Ulcerative colitis is believed to be the delivery of the active agent past the small intestine to the large intestine, the active site of ulcerative colitis.
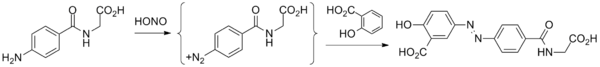
Balsalazide disodium and its complete synthesis was first disclosed by Chan[18] in 1983, assigned to Biorex Laboratories Limited, England, claiming product ‘Balsalazide’ and process of its preparation. The synthesis involves converting 4-nitrobenzoyl chloride (6) to 4- nitrobenzoyl-β-alanine (7), hydrogenating with Pd/C (5%) in ethanol and isolating by adding diethyl ether to produce 4-aminobenzoyl-β-alanine (8). Thereafter, 4-aminobenzoyl-β-alanine (8) was treated with hydrochloric acid and sodium nitrite to generate N-(4-diazoniumbenzoyl)- β-alanine hydrochloride salt (9) which was reacted at low temperature with disodium salicylate to furnish Balsalazide disodium insitu which was added to dilute hydrochloric acid at low temperature to produce Balsalazide (1) (Scheme-1.1). Thus obtained Balsalazide was recrystallized with hot ethanol and converted to pharmaceutically acceptable salt (disodium salt).

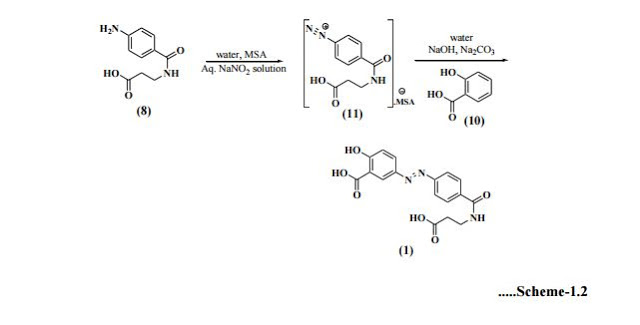
Optimization of this diazonium salt based process was performed by Huijun et al[19] and reported the preparation of the title compound in 64.6% overall yield. Zhenhau et al[20] have synthesized 1 from 4-nitrobenzoic acid (12) via chlorination, condensation, hydrogenation, diazotization, coupling and salt formation with overall yield 73%. Li et al[21] have given product in 73.9% total yield starting from 4-nitrobenzoyl chloride (6), where as Yuzhu et al[22] confirmed chemical structure of Balsalazide disodium by elemental analysis, UV, IR, 1H-NMR and ESI-MS etc. Shaojie et al[23] have also followed same process for its preparation. Yujie et al[24] synthesized 1 in this way; preparation of 4-nitrobenzoyl-β-alanine (7) under microwave irradiation of 420 W at 52oC for 10sec., reduction in ethyl acetate in the presence of Pd/C catalyst then diazotization, coupling and salt formation. Eckardt et al[25] have developed a process for the preparation of Balsalazide which comprises, conversion of 4-aminobenzoyl-β-alanine (8) to 4-ammoniumbenzoyl-β-alanine sulfonate salt using a sulfonic acid in water. This was treated with aq. sodium nitrite solution at low temperature to generate 4-diazoniumbenzoyl-β-alanine sulfonate salt (11) which was quenched with aq. disodium salicylate to furnish Balsalazide disodium solution. This was further acidified to allow isolation of 1 and then conversion to disodium salt (Scheme-1.2) in 76% yield.
http://shodhganga.inflibnet.ac.in/bitstream/10603/101297/10/10_chapter%201.pdf
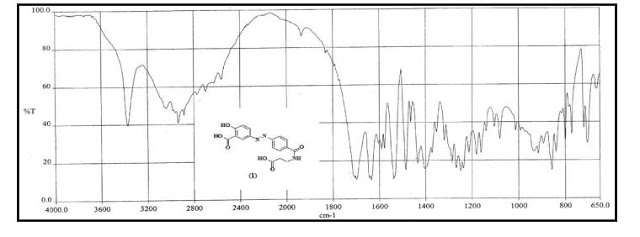
IR (KBr, cm-1 ): 3371 and 3039 (OH and NH), 1705 and 1699 (C=O), 1634 (C=O amide), 1590 and 1538 (C=C aromatic), 1464 and 1404 (aliphatic C-H), 1229 (C-N), 1073 (C-O), 773 and 738 (Ar-H out of plane bend). 1H NMR (DMSO-d6, 300 MHz, δ ppm): 2.54 (t, 2H), 3.50 (m, 2H), 6.95 (d, J = 8.8 Hz, 1H), 7.87 (d, J = 8.5 Hz, 2H), 8.02 (d, J = 8.5 Hz, 2H), 7.95 (dd, J = 8.8 Hz and 2.5 Hz, 1H), 8.34 (d, J = 2.5 Hz, 1H), 8.68 (t, J = 5.5 Hz, 1H), 12.12 (brs, 1H). MS m/z (ESI): 356 [(M-H)- ], Calculated; m/z 357.
Synthesis
Balsalazide synthesis: Biorex Laboratories, GB 2080796 (1986).
- Starting material is 4-aminohippuric acid, obtained by coupling para-aminobenzoic acid and glycine.
- That product is then treated with nitrous acid to give the diazonium salt.
- Reaction of this species with salicylic acid proceeds at the position para to the phenol to give balsalazide.
Sodium balsalazide (Balsalazide sodium)
Brief background information
Application
-
resolvent
Classes substance
-
β-alanine (3-aminopropionic acid)
-
m-aminobenzoic acid and esters and amides thereof
-
p-aminobenzoic acid and esters and amides thereof
-
azobenzene
-
salicylic acid
-
-
-
-
Synthesis Way
Trade names
| A country | Tradename | Manufacturer |
|---|---|---|
| United Kingdom | Kolazid | Shire |
| Italy | Balzid | Menarini |
| USA | Kolazal | Salix |
| Ukraine | no | no |
Formulations
-
capsules in 750 mg (as disodium salt)
PATENT
https://www.google.com/patents/US7271253
Balsalazide disodium (1) represents an effective gastrointestinal anti-inflammatory compound useful as a medicament for the treatment of diseases such as ulcerative colitis. It is delivered intact to the colon where it is cleaved by bacterial azoreduction thereby generating 5-aminosalicylic acid as the medicinally active component.
To date, relatively few patents or literature articles have dealt with the preparation of Balsalazide or the disodium salt. For instance, U.S. Pat. No. 4,412,992 (Biorex, 1983) is the first patent that we uncovered that claims the compound Balsalazide and a strategy of how to prepare it which strategy is depicted in Scheme 1.
Optimization of this diazonium-based process is detailed in Shan et al., Zhongguo Yaowu Huaxue Zazhi, 11, 110 (2001) and Shi et al., Zhongguo Yiyao Gongye Zazhi, 34, 537 (2003).
Problems arise with the above strategy and the optimization process.
It is well-documented in the literature, for instance in Thermochimica Acta, 225, 201-211 (1993), that diazonium salts can be involved in serious accidents in their use. A possible cause of some of the diazonium salt related accidents is that, for one reason or another, an intermediate material appeared in crystalline form in the vessel of the reaction. As a result, a potentially severe drawback of the above processes occurs. Since the intermediate hydrochloride salt of 4-aminobenzoyl-β-alanine has poor solubility in water, it may pose a safety-risk in the subsequent diazotation reaction.
Also, it is well-known that certain diazonium salts possess high mechanical and heat sensitivity and that their decomposition goes through the liberation of non-condensable nitrogen gas which results in the possibility of runaway reactions and explosions. Obviously this safety consideration becomes more pertinent upon further scale-up.
Therefore, for commercial production of Balsalazide disodium, there was a need to develop a scalable and intrinsically better process
Example 1 Batch Process
N-(4-Aminobenzoyl)-β-alanine (100 g) was suspended in water (1300 mL) and methanesulfonic acid (115.4 g) was added to this mixture. The mixture was cooled to 10° C. and a solution of sodium nitrite (34.46 g) in water (200 mL) was added at a rate such that the temperature stayed below 12° C. The mixture was stirred for 30 min and added to an ice-cold solution of salicylic acid (69.65 g), sodium hydroxide (40.35 g) and sodium carbonate (106.9 g) in 1 L water at 7-12° C. After 3 hours at 10° C., the mixture was heated to 60-65° C. and acidified to pH 4.0-4.5 by the addition of hydrochloric acid. After a further 3 hours at 60-65° C., the mixture was cooled to ambient temperature, filtered, washed with water and dried in vacuo to yield Balsalazide. Yield ca. 90%. Balsalazide was transformed into its disodium salt in ca. 85% yield by treatment with aqueous NaOH solution followed by crystallization from n-propanol/methanol.
1H-NMR (400 MHz; D2O): δ=8.04 ppm (s); 7.67 ppm (d; J=8.2 Hz); 7.62 ppm (d, J=9.2 Hz); 7.53 ppm (d; J=8.2 Hz); 6.84 ppm (d; J=8.9 Hz); 3.57 ppm (t, J=7.1 Hz); 2.53 ppm (t; J=7.2 Hz).
Example 2 Continuous Process
For the continuous operation, a conventional dual-head metering pump (Ratiomatic by FMI) was used to deliver the mesylate solution and the aqueous sodium nitrite solution. The schematic diagram shown in FIG. 4 represents a set-up used for the continuous process. The first pump-head was set at 13.9 g/min whereas the second was set at 2.1 g/min. These flow rates offered a residence time of 9.4 min. The yield of the coupled intermediate from this residence time was 93%. The working solutions were prepared as follow:
The mesylate solution was prepared by the addition into a 2 L 3-necked round bottom flask, of N-(4-aminobenzoyl) β-alanine (120 g) followed by of DI water (1560 g) and methanesulfonic acid (177.5 g) (Batch appearance: clear solution). The first pump-head was primed with this solution and the flow rate was adjusted to 13.9 g/min.
The sodium nitrite solution was prepared by dissolving of sodium nitrite (41.8 g) in of DI water (240 g) (Batch appearance: clear solution). The second pump-head was primed with this solution and the flow rate adjusted to 2.1 g/min.
The quenching solution (sodium salicylate) was made by adding salicylic acid (139.3 g) to DI water (900 g) followed by of sodium carbonate (106.9 g) and 50% aqueous sodium hydroxide (80 g).
The diazotation reaction was performed in a 500 ml jacketed flow reactor with a bottom drain valve. The drain valve was set at 16 g/min. For reactor start-up, the flow reactor was charged with 150 mL of DI water as a working volume and cooled to the reactions initial temperature of 0-5° C. Concomitantly, the additions of the mesylate and sodium nitrite solutions were started and the bottom valve of the flow reactor was opened. During the diazotization, the flow rate of both solutions remained fixed and the temperature was kept below 12° C. and at the end of additions the pumps were stopped while the remaining contents in the flow reactor were drained into the quenching salicylic acid solution. Analysis of the contents in the quenching reactor indicated no signs of uncoupled starting material (diazonium compound). The reactor contents were heated to 60-65° C. for 2-3 hrs before adjusting the pH to precipitate the coupling product. This provided 191.5 g of product.
| Cited Patent | Filing date | Publication date | Applicant | Title |
|---|---|---|---|---|
| US4412992 | Jul 8, 1981 | Nov 1, 1983 | Biorex Laboratories Limited | 2-Hydroxy-5-phenylazobenzoic acid derivatives and method of treating ulcerative colitis therewith |
| US6458776 * | Aug 29, 2001 | Oct 1, 2002 | Nobex Corporation | 5-ASA derivatives having anti-inflammatory and antibiotic activity and methods of treating diseases therewith |
| Reference | ||
|---|---|---|
| 1 | Chai, et al., Huaxi Yaoxue Zazhi, Jiangsu Institute of Materia Medica, Nanjing, China, 2004, 19(6), 431-433. | |
| 2 | Shan, et al., Zhongguo Yaowu Huaxue Zazhi, Institute of Materia Medica, Peking Union Medical College, Beijing China, 2001, 11(2), 110-111. | |
| 3 | Shi, et al., Zhongguo Yiyao Gongya Zazhi, Shanghai Institute of Pharmaceutical Industry, Shanghai, China, 2003, 34(11), 537-538. | |
| 4 | Su, et al., Huaxue Gongye Yu Gongcheng (Tianjin, China), College of Chemistry and Chemical Eng., Donghua Univ., Shanghai, China, 2005, 22(4), 313-315. | |
| 5 | Ullrich, et al., Decomposition of aromataic diazonium compounds, Thermochimica Acta, 1993, 225, 201-211. | |
References
-
Prakash, A; Spencer, CM: Drugs (DRUGAY) 1998 56 83- 89.
-
DE 3128819 (Biorex the Lab .; appl 07/21/1981;. GB -prior 07/21/1980, 07.07.1981.).
References
- Jump up^ Kruis, W.; Schreiber, I.; Theuer, D.; Brandes, J. W.; Schütz, E.; Howaldt, S.; Krakamp, B.; Hämling, J.; Mönnikes, H.; Koop, I.; Stolte, M.; Pallant, D.; Ewald, U. (2001). “Low dose balsalazide (1.5 g twice daily) and mesalazine (0.5 g three times daily) maintained remission of ulcerative colitis but high dose balsalazide (3.0 g twice daily) was superior in preventing relapses”. Gut. 49 (6): 783–789. doi:10.1136/gut.49.6.783. PMC 1728533
 . PMID 11709512.
. PMID 11709512.
| Patent ID | Patent Title | Submitted Date | Granted Date |
|---|---|---|---|
| US8232265 | Multi-functional ionic liquid compositions for overcoming polymorphism and imparting improved properties for active pharmaceutical, biological, nutritional, and energetic ingredients | 2007-04-26 | 2012-07-31 |
| US2011319267 | AROMATIC CARBOXYLIC ACID DERIVATIVES FOR TREATMENT AND PROPHYLAXIS OF GASTROINTESTINAL DISEASES INCLUDING COLON CANCERS | 2011-12-29 | |
| US2007213304 | Use of Aminosalicylates in Diarrhoea-Predominent Irritable Bowel Syndrome | 2007-09-13 | |
| US7119079 | Bioadhesive pharmaceutical compositions | 2004-07-22 | 2006-10-10 |
| US6699848 | Bioadhesive anti-inflammatory pharmaceutical compositions | 2004-03-02 |
 |
|
| Clinical data | |
|---|---|
| Trade names | Colazal, Giazo |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a699052 |
| Pregnancy category |
|
| ATC code | A07EC04 (WHO) |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | <1% |
| Protein binding | ≥99% |
| Biological half-life | 12hr |
| Identifiers | |
| CAS Number | 80573-04-2 |
| PubChem (CID) | 5362070 |
| DrugBank | DB01014 |
| ChemSpider | 10662422 |
| UNII | P80AL8J7ZP |
| ChEBI | CHEBI:267413 |
| ChEMBL | CHEMBL1201346 |
| ECHA InfoCard | 100.117.186 |
| Chemical and physical data | |
| Formula | C17H15N3O6 |
| Molar mass | 357.318 g/mol |
| 3D model (Jmol) | Interactive image |
 CLICK ON IMAGE
CLICK ON IMAGE
//////
O=C(O)c1cc(ccc1O)/N=N/c2ccc(cc2)C(=O)NCCC(O)=O
|
O.O.[Na+].[Na+].OC1=CC=C(C=C1C([O-])=O)\N=N\C1=CC=C(C=C1)C(=O)NCCC([O-])=O
|


 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....