Home » Posts tagged 'GMP' (Page 18)
Tag Archives: GMP
Orexo gets FDA OK for opioid dependence drug
Regulators in the USA have given the green light to Orexo of Sweden’s Zubsolv as a treatment for opioid addiction.
The US Food and Drug Administration has approved Zubsolv, a once-daily sublingual formulation of Reckitt Benckiser’s Suboxone (buprenorphine/naloxone) which currently dominates the market. It is indicated for use as maintenance treatment for people suffering from opioid dependence and should be used as part of a complete treatment plan to include counselling and psychosocial support
read all at
http://www.pharmatimes.com/Article/13-07-05/Orexo_gets_FDA_OK_for_opioid_dependence_drug.aspx
FDA clears Aurobindo’s Rizatriptan Benzoate orally disintegrating tablets
India-based Aurobindo Pharma has obtained final FDA approvals to manufacture and market Rizatriptan Benzoate orally disintegrating tablets (ODT) 5mg and 10mg (ANDA 203062)…
read more at
Rizatriptan (trade name Maxalt) is a 5-HT1 agonist triptan drug developed by Merck & Co. for the treatment of migraine headaches. It is available in strengths of 5 and 10 mg as tablets and orally disintegrating tablets (Maxalt-MLT).
Maxalt obtained approval by the United States Food and Drug Administration (FDA) on June 29, 1998. It is a second-generation triptan.
Rizatriptan is available only by prescription in Australia, the United States, Canada andNew Zealand. Similarly, it is classed as a POM (Prescription Only Medicine) in the United Kingdom, Italy (as Rizaliv), Israel (as Rizalt), The Netherlands, Croatia and Spain (asMaxalt)

Anthrax Killer from the Sea
Anthrax Killer from the Sea
 Unusual antibiotic from a marine actinomycete is effective against anthrax
Unusual antibiotic from a marine actinomycete is effective against anthrax
http://www.chemistryviews.org/details/ezine/4972631/Anthrax_Killer_from_the_Sea.html
- Anthracimycin, a Potent Anthrax Antibiotic from a Marine-Derived Actinomycete,
Kyoung Hwa Jang, Sang-Jip Nam, Jeffrey B. Locke, Christopher A. Kauffman, Deanna S. Beatty, Lauren A. Paul, William Fenical,
Angew. Chem. Int. Ed. 2013.
DOI: 10.1002/anie.201302749
From Pharmacy to the Pub — A Bark Conquers the World: Part 3
 The long road from the structure determination to the total synthesis of quinine is an exciting detective story
The long road from the structure determination to the total synthesis of quinine is an exciting detective story
FDA awards Activartis brain cancer drug Orphan Drug Designation
Austrian biotech company Activartis has received Orphan Drug Designation for its cancer immunotherapy AV0113 from the US Food and Drug Administration.
The experimental vaccine will be fast-tracked for evaluation after it was shown to improve survival rates in people with malignant glioma, an advanced form of brain cancer in a phase II clinical trial earlier this year.

Roche’s Perjeta Gets FDA Priority Review

The structure of HER2 and pertuzumab
Application follows proposed new FDA pathway designed to help bring promising medicines to people with earlier stages of breast cancer faster
Perjeta is one of the first medicines the FDA will evaluate as an option given before surgery (neoadjuvant treatment)
Perjeta, in combination with Herceptin and chemotherapy, was approved by the FDA in 2012 for HER2-positive metastatic breast cancer
Pertuzumab (also called 2C4, trade name Perjeta) is a monoclonal antibody. The first of its class in a line of agents called “HER dimerization inhibitors”. By binding to HER2, it inhibits the dimerization of HER2 with other HER receptors, which is hypothesized to result in slowed tumor growth. Pertuzumab received US FDA approval for the treatment of HER2-positive metastatic breast cancer on June 8, 2012. Pertuzumab was developed at Genentech and is now owned by Roche which acquired Genentech in 2009.
Early clinical trials of pertuzumab in prostate, breast, and ovarian cancers met with limited success.
The dosage of pertuzumab used in the pivotal phase III CLEOPATRA (Clinical Evaluation of Pertuzumab and Trastuzumab) trial was as follows: IV 840 mg loading dose followed by IV 420 mg every three weeks.
The pharmacokinetics of intravenous pertuzumab appear to be unaffected by age and no drug-drug interaction has been reported with docetaxel. The pharmacokinetics and pharmacodynamics of pertuzumab were summarized in a Feb 2012 review by Gillian Keating.
The combination of pertuzumab plus trastuzumab plus docetaxel, as compared with placebo plus trastuzumab plus docetaxel, when used as first-line treatment for HER2-positive metastatic breast cancer, significantly prolonged progression-free survival, with no increase in cardiac toxic effects in the randomized, double-blind, multinational, phase III CLEOPATRA trial.
Intravenous pertuzumab is currently being evaluated in patients with breast cancer in the following trials: MARIANNE (advanced breast cancer), NEOSPHERE (early breast cancer), TRYPHAENA (HER2-positive stage II/III breast cancer) and APHINITY (HER2-positive nonmetastatic breast cancer)
BMS, Pfizer: Eliquis Meets Phase III Goal


Eliquis® (apixaban) Demonstrated Comparable Efficacy and Significantly Lower Rates of Major Bleeding in Patients Compared to Current Standard of Care for the Treatment of Acute Venous Thromboembolism
Phase 3 AMPLIFY Results Published in New England Journal of Medicine and Presented as a Late-Breaker at the Congress of the International Society on Thrombosis and Haemostasis Show:
•Eliquis Was Noninferior to Current Standard of Care for Treatment of Both Symptomatic Deep Vein Thrombosis and Pulmonary Embolism Conditions
•69 Percent Relative Risk Reduction for Major Bleeding in Patients on Eliquis Compared to Current Standard of Care “The study results showed that apixaban, as a single-agent, has comparable efficacy with significantly fewer major bleeding events with respect to the standard of care.
These results complement the previously published results for the AMPLIFY-EXT study” PRINCETON, N.J. and NEW YORK, June 30, 2013
READ ALL AT
http://www.pharmalive.com/bms-pfizer-eliquis-meets-phase-iii-goal

Flamel Technologies Announces FDA Approval of Bloxiverz

LYON, FRANCE — (Marketwire) — 06/03/2013 — Flamel Technologies today announced that the U.S. Food and Drug Administration (FDA) has approved the company’s New Drug Application (NDA) for Bloxiverz (neostigmine methylsulfate), a drug used intravenously in the operating room for the reversal of the effects of non-depolarizing neuromuscular blocking agents after surgery. Flamel expects to launch Bloxiverz in July 2013 in 0.5 and 1.0 mg/mL strengths
read all at
.http://www.drugs.com/newdrugs/flamel-technologies-announces-fda-approval-bloxiverz-3802.html
Sunovion Pharmaceuticals Inc. Announces FDA Approval of Latuda® (lurasidone HCl) as Monotherapy and Adjunctive Therapy in Adult Patients with Bipolar Depression

lurasidone
MARLBOROUGH, Mass. – Monday, July 1st 2013 [ME NewsWire]
First Atypical Antipsychotic Indicated for the Treatment of Major Depressive Episodes Associated with Bipolar I Disorder (Bipolar Depression) Both as Monotherapy and as Adjunctive Therapy with Either Lithium or Valproate
(BUSINESS WIRE)– Sunovion Pharmaceuticals Inc. today announced that the U.S. Food and Drug Administration (FDA) approved two new indications for the use of Latuda® (lurasidone HCl) as 1) monotherapy and 2) adjunctive therapy with either lithium or valproate, both to treat adult patients with major depressive episodes associated with bipolar I disorder (bipolar depression).
read all at
http://www.me-newswire.net/news/7829/en
Lurasidone (trade name Latuda) is an atypical antipsychotic developed by Dainippon Sumitomo Pharma and marketed by Sunovion in the USA. It was approved by the U.S. Food and Drug Administration (FDA) for treatment of schizophrenia on October 28, 2010 after a review that found that two of the four Phase III clinical trials supported efficacy, while one showed only marginal efficacy and one was not interpretable because of high drop-out rates. It is currently pending approval for the treatment of bipolar disorder in the United States. It was launched in Canada for the treatment of schizophrenia in September 2012, Health Canada giving their Summary Basis of Decision (SBD) as favourable on October 15, 2012
FDA Approves Brisdelle, paroxetine mesylate- First Non-Hormonal Treatment for Hot Flashes Associated with Menopause
June 28, 2013 –The U.S. Food and Drug Administration today approved Brisdelle (paroxetine) to treat moderate to severe hot flashes (vasomotor symptoms) associated with menopause. Brisdelle, which contains the selective serotonin reuptake inhibitor paroxetine mesylate, is currently the only non-hormonal treatment for hot flashes approved by the FDA.
There are a variety of FDA-approved treatments for hot flashes, but all contain either estrogen alone or estrogen plus a progestin.
read all at
more info
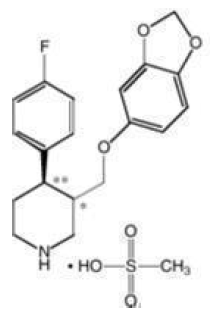
PEXEVA® (paroxetine mesylate) is an orally administered psychotropic drug with a chemical structure related to paroxetine hydrochloride (Paxil®). It is the mesylate salt of a phenylpiperidine compound identified chemically as (-)-trans-4R-(4′-fluorophenyl)-3S-[(3′,4′-methylenedioxyphenoxy) methyl] piperidine mesylate and has the empirical formula of C19H20FNO3•CH3SO3H. The molecular weight is 425.5 (329.4 as free base). The structural formula is: paroxetine mesylate
 |
Paroxetine mesylate is an odorless, off-white powder, having a melting point range of 147° to 150°C and a solubility of more than 1 g/ml in water.
Tablets
Each oval, film-coated tablet contains paroxetine mesylate equivalent to paroxetine as follows: 10 mg (white); 20 mg (scored, dark orange); 30 mg (yellow); 40 mg (rose). Inactive ingredients consist of dibasic calcium phosphate, hydroxypropyl methylcellulose, hydroxypropylcellulose, magnesium stearate, sodium starch glycolate, titanium dioxide, ferric oxide red (C.I. 77491) (20 mg and 40 mg only) and ferric oxide yellow (C.I. 77492) (20 mg, 30 mg, and 40 mg only).
EP1286965B1




 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....











