Cladribine, クラドリビン
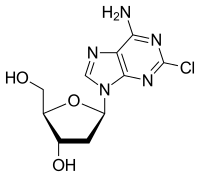
Cladribine
クラドリビン
Leustatin
RWJ 26251 / RWJ-26251
- Molecular FormulaC10H12ClN5O3
- Average mass285.687 Da
Cladribine, sold under the brand name Leustatin and Mavenclad among others, is a medication used to treat hairy cell leukemia(HCL, leukemic reticuloendotheliosis), B-cell chronic lymphocytic leukemia and relapsing-remitting multiple sclerosis.[4][5] Its chemical name is 2-chloro-2′-deoxyadenosine (2CdA).
Cladribine, a deoxyadenosine derivative developed by Ortho Biotech (currently Janssen), was first launched in the U.S. in 1993 as an intravenous treatment for hairy cell leukemia
Cladribine has been granted orphan drug designation in the U.S. in 1990 for the treatment of acute myeloid leukemia (AML) and hairy cell leukemia
As a purine analog, it is a synthetic chemotherapy agent that targets lymphocytes and selectively suppresses the immune system. Chemically, it mimics the nucleoside adenosine. However, unlike adenosine it is relatively resistant to breakdown by the enzyme adenosine deaminase, which causes it to accumulate in cells and interfere with the cell’s ability to process DNA. Cladribine is taken up cells via a transporter. Once inside a cell cladribine is activated mostly in lymphocytes, when it is triphosphorylated by the enzyme deoxyadenosine kinase (dCK). Various phosphatases dephosphorylate cladribine. Activated, triphosphorylated, cladribine is incorporated into mitochondrial and nuclear DNA, which triggers apoptosis. Non-activated cladribine is removed quickly from all other cells. This means that there is very little non-target cell loss.[4][6]
Medical uses
Cladribine is used for as a first and second-line treatment for symptomatic hairy cell leukemia and for B-cell chronic lymphocytic leukemia and is administered by intravenous or subcutaneous infusion.[5][7]
Since 2017, cladribine is approved as an oral formulation (10 mg tablet) for the treatment of RRMS in Europe, UAE, Argentina, Chile, Canada and Australia. Marketing authorization in the US was obtained in March 2019[8].
Some investigators have used the parenteral formulation orally to treat patients with HCL. It is important to note that approximately 40% of oral cladribine in bioavailable orally. It used, often in combination with other cytotoxic agents, to treat various kinds of histiocytosis, including Erdheim–Chester disease[9] and Langerhans cell histiocytosis,[10]
Cladribine can cause fetal harm when administered to a pregnant woman and is listed by the FDA as Pregnancy Category D; safety and efficacy in children has not been established.[7]
Adverse effects
Injectable cladribine suppresses the body’s ability to make new lymphocytes, natural killer cells and neutrophils (called myelosuppression); data from HCL studies showed that about 70% of people taking the drug had fewer white blood cells and about 30% developed infections and some of those progressed to septic shock; about 40% of people taking the drug had fewer red blood cells and became severely anemic; and about 10% of people had too few platelets.[7]
At the dosage used to treat HCL in two clinical trials, 16% of people had rashes and 22% had nausea, the nausea generally did not lead to vomiting.[7]
In comparison, in MS, cladribine is associated with a 6% rate of severe lymphocyte suppression (lymphopenia) (levels lower than 50% of normal). Other common side effects include headache (75%), sore throat (56%), common cold-like illness (42%) and nausea (39%)[11]
Mechanism of Action
As a purine analogue, it is taken up into rapidly proliferating cells like lymphocytes to be incorporated into DNA synthesis. Unlike adenosine, cladribine has a chlorine molecule at position 2, which renders it partially resistant to breakdown by adenosine deaminase (ADA). In cells it is phosphorylated into its toxic form, deoxyadenosine triphosphate, by the enzyme deoxycytidine kinase (DCK). This molecule is then incorporated into the DNA synthesis pathway, where it causes strand breakage. This is followed by the activation of transcription factor p53, the release of cytochrome c from mitochondria and eventual programmed cell death (apoptosis).[12] This process occurs over approximately 2 months, with a peak level of cell depletion 4–8 weeks after treatment[13]
Within the lymphocyte pool, cladribine targets B cells more than T cells. Both HCL and B-cell chronic lymphocytic leukaemia are types of B cell blood cancers. In MS, its effectiveness may be due to its ability to effectively deplete B cells, in particular memory B cells[14] In the pivotal phase 3 clinical trial of oral cladribine in MS, CLARITY, cladribine selectively depleted 80% of peripheral B cells, compared to only 40-50% of total T cells.[15] More recently, cladribine has been shown to induce long term, selective suppression of certain subtypes of B cells, especially memory B cells.[16]
Another family of enzymes, the 5´nucleotidase (5NCT) family, is also capable of dephosphorylating cladribine, making it inactive. The most important subtype of this group appears to be 5NCT1A, which is cytosolically active and specific for purine analogues. When DCK gene expression is expressed as a ratio with 5NCT1A, the cells with the highest ratios are B cells, especially germinal centre and naive B cells.[16] This again helps to explain which B cells are more vulnerable to cladribine-mediated apoptosis.
Although cladribine is selective for B cells, the long term suppression of memory B cells, which may contribute to its effect in MS, is not explained by gene or protein expression. Instead, cladribine appears to deplete the entire B cell department. However, while naive B cells rapidly move from lymphoid organs, the memory B cell pool repopulates very slowly from the bone marrow.
History
Ernest Beutler and Dennis A. Carson had studied adenosine deaminase deficiency and recognized that because the lack of adenosine deaminase led to the destruction of B cell lymphocytes, a drug designed to inhibit adenosine deaminase might be useful in lymphomas. Carson then synthesized cladribine, and through clinical research at Scripps starting in the 1980s, Beutler tested it as intravenous infusion and found it was especially useful to treat hairy cell leukemia (HCL). No pharmaceutical companies were interested in selling the drug because HCL was an orphan disease, so Beutler’s lab synthesized and packaged it and supplied it to the hospital pharmacy; the lab also developed a test to monitor blood levels. This was the first treatment that led to prolonged remission of HCL, which was previously untreatable.[17]:14–15
In February 1991 Scripps began a collaboration with Johnson & Johnson to bring intravenous cladribine to market and by December of that year J&J had filed an NDA; cladrabine was approved by the FDA in 1993 for HCL as an orphan drug,[18] and was approved in Europe later that year.[19]:2
The subcutaneous formulation was developed in Switzerland in the early 1990s and it was commercialized by Lipomed GmbH in the 2000s.[19]:2[20]
Multiple sclerosis
In the mid-1990s Beutler, in collaboration with Jack Sipe, a neurologist at Scripps, ran several clinical trials exploring the utility of cladribine in multiple sclerosis, based on the drug’s immunosuppressive effects. Sipe’s insight into MS, and Beutler’s interest in MS due to his sister’s having had it, led a very productive collaboration.[17]:17[21] Ortho-Clinical, a subsidiary of J&J, filed an NDA for cladribine for MS in 1997 but withdrew it in the late 1990s after discussion with the FDA proved that more clinical data would be needed.[22][23]
Ivax acquired the rights for oral administration of cladribine to treat MS from Scripps in 2000,[24] and partnered with Serono in 2002.[23] Ivax was acquired by Teva in 2006,[25][26] and Merck KGaA acquired control of Serono’s drug business in 2006.[27]
An oral formulation of the drug with cyclodextrin was developed[28]:16 and Ivax and Serono, and then Merck KGaA conducted several clinical studies. Merck KGaA submitted an application to the European Medicines Agency in 2009, which was rejected in 2010, and an appeal was denied in 2011.[28]:4–5 Likewise Merck KGaA’s NDA with the FDA rejected in 2011.[29] The concerns were that several cases of cancer had arisen, and the ratio of benefit to harm was not clear to regulators.[28]:54–55 The failures with the FDA and the EMA were a blow to Merck KGaA and were one of a series of events that led to a reorganization, layoffs, and closing the Swiss facility where Serono had arisen.[30][31] However, several MS clinical trials were still ongoing at the time of the rejections, and Merck KGaA committed to completing them.[29] A meta-analysis of data from clinical trials showed that cladiribine did not increase the risk of cancer at the doses used in the clinical trials.[32]
In 2015 Merck KGaA announced it would again seek regulatory approval with data from the completed clinical trials in hand,[30] and in 2016 the EMA accepted its application for review.[33] On June 22, 2017, the EMA’s Committee for Medicinal Products for Human Use (CHMP) adopted a positive opinion, recommending the granting of a marketing authorisation for the treatment of relapsing forms of multiple sclerosis.[34]
Finally, after all these problems it was approved in Europe on August 2017 for highly active RRMS.[35]
Efficacy
Cladribine is an effective treatment for relapsing remitting MS, with a reduction in the annual rate of relapses of 54.5%.[11] These effects may be sustained up to 4 years after initial treatment, even if no further doses are given.[36] Thus, cladribine is considered to be a highly effective immune reconstitution therapy in MS. Similar to alemtuzumab, cladribine is given as two courses approximately one year apart. Each course consists of 4-5 tablets given over a week in the first month, followed by a second dosing of another 4-5 tablets the following month[37] During this time and after the final dose patients are monitored for adverse effects and signs of relapse.
https://www.merckneurology.co.uk/wp-content/uploads/2017/08/mavenclad-table-1.jpg
Safety
Compared to alemtuzumab, cladribine is associated with a lower rate of severe lymphopenia. It also appears to have a lower rate of common adverse events, especially mild to moderate infections[11][36] As cladribine is not a recombinant biological therapy, it is not associated with the development of antibodies against the drug, which might reduce the effectiveness of future doses. Also, unlike alemtuzumab, cladribine is not associated with secondary autoimmunity.[38]
This is probably due to the fact cladribine more selectively targets B cells. Unlike alemtuzumab, cladribine is not associated with a rapid repopulation of the peripheral blood B cell pool, which then ´overshoots´ the original number by up to 30%.[39] Instead, B cells repopulate more slowly, reaching near normal total B cells numbers at 1 year. This phenomenon and the relative sparing of T cells, some of which might be important in regulating the system against other autoimmune reactions, is thought to explain the lack of secondary autoimmunity.
Use in clinical practice
The decision to start cladribine in MS depends on the degree of disease activity (as measured by number of relapses in the past year and T1 gadolinium-enhancing lesions on MRI), the failure of previous disease-modifying therapies, the potential risks and benefits and patient choice.
In the UK, the National Institute for Clinical Excellence (NICE) recommends cladribine for treating highly active RRMS in adults if the persons has:
rapidly evolving severe relapsing–remitting multiple sclerosis, that is, at least 2 relapses in the previous year and at least 1 T1 gadolinium-enhancing lesion at baseline MRI or
relapsing–remitting multiple sclerosis that has responded inadequately to treatment with disease-modifying therapy, defined as 1 relapse in the previous year and MRI evidence of disease activity.[40]
People with MS require counselling on the intended benefits of cladribine in reducing the risk of relapse and disease progression, versus the risk of adverse effects such as headaches, nausea and mild to moderate infections. Women of childbearing age also require counselling that they should not conceive while taking cladribine, due to the risk of harm to the fetus.
Cladribine, as the 10 mg oral preparation Mavenclad, is administered as two courses of tablets approximately one year apart. Each course consists of four to five treatment days in the first month, followed by an additional four to five treatment days in the second month. The recommended dose of Mavenclad is 3.5 mg/kg over 2 years, given in two treatment courses of 1.75 mg/kg/year. Therefore, the number of tablets administered on each treatment day depends on the person’s weight. A full guide to the dosing strategy can be found below:
https://www.merckneurology.co.uk/mavenclad/mavenclad-efficacy/
After treatment, people with MS are monitored with regular blood tests, looking specifically at the white cell count and liver function. Patients should be followed up regularly by their treating neurologist to assess efficacy, and should be able to contact their MS service in the case of adverse effects or relapse. After the first two years of active treatment no further therapy may need to be given, as cladribine has been shown to be efficacious for up to last least four years after treatment. However, if patients fail to respond, options include switching to other highly effective disease-modifying therapies such as alemtuzumab, fingolimod or natalizumab.
Research directions
Cladribine has been studied as part of a multi-drug chemotherapy regimen for drug-resistant T-cell prolymphocytic leukemia.[41]
REF
A universal biocatalyst for the preparation of base- and sugar-modified nucleosides via an enzymatic transglycosylation
Helv Chim Acta 2002, 85(7): 1901
Synthesis of 2-chloro-2′-deoxyadenosine by microbiological transglycosylation
Nucleosides Nucleotides 1993, 12(3-4): 417
Synthesis of 2-chloro-2′-deoxyadenosine by washed cells of E. coli
Biotechnol Lett 1992, 14(8): 669
Efficient syntheses of 2-chloro-2′-deoxyadenosine (cladribine) from 2′-deoxyguanosine
J Org Chem 2003, 68(3): 989
WO 2004028462
Synthesis of 2′-deoxytubercidin, 2′-deoxyadenosine, and related 2′-deoxynucleosides via a novel direct stereospecific sodium salt glycosylation procedure
J Am Chem Soc 1984, 106(21): 6379
WO 2011113476
A stereoselective process for the manufacture of a 2′-deoxy-beta-D-ribonucleoside using the vorbruggen glycosylation
Org Process Res Dev 2013, 17(11): 1419
A new synthesis of 2-chloro-2′-deoxyadenosine (Cladribine), CdA)
Nucleosides Nucleotides Nucleic Acids 2011, 30(5): 353
A dramatic concentration effect on the stereoselectivity of N-glycosylation for the synthesis of 2′-deoxy-beta-ribonucleosides
Chem Commun (London) 2012, 48(56): 7097
CN 105367616
PATENT
https://patents.google.com/patent/EP2891660A1/en
Previously Robins and Robins (Robins, M. J. and Robins, R. K., J. Am. Chem. Soc. 1965, 87, 4934-4940) reported that acid-catalyzed fusion of 1,3,5-tri-O-acety-2-deoxy-D-ribofuranose and 2,6-dichloropurine gave a 65% yield of an anomeric mixture 2,6-dichloro-9-(3′,5′-di-O-acetyl-2′-deoxy-α-,β-D-ribofuranosyl)-purines from which the α-anomer was obtained as a pure crystalline product by fractional crystallization from ethanol in 32% yield and the equivalent β-anomer remained in the mother liquor (see Scheme 1). The β-anomer, which could have been used to synthesize cladribine, wasn’t isolated further. The α-anomer was treated with methanolic ammonia which resulted in simultaneous deacetylation and amination to give 6-amino-2-chloro-9-(2′-deoxy-α-D-ribofuranosyl)-purine, which is a diastereomer of cladribine.
[0004]
Broom et al. (Christensen, L. F., Broom, A. D., Robins, M. J., and Bloch, A., J. Med. Chem. 1972, 15, 735-739) adapted Robins et al.’s method by treating the acetylated mixture (viz., 2,6-dichloro-9-(3′,5′-di-O-acety-2′-deoxy-α,β-D-ribofuranosyl)-purine) with liquid ammonia and reacylating the resulting 2′-deoxy-α-and –β-adenosines with p-toluoyl chloride (see Scheme 2). The desired 2-chloro-9-(3′,5′-di-O–p-toluoyl-2′-deoxy-β-D-ribofuranosyl)-adenine was then separated by chromatography and removal of the p-toluoyl group resulted in cladribine in 9% overall yield based on the fusion of 1,3,5-tri-O-acety-2-deoxy-D-ribofuranose and 2,6-dichloropurine.
To increase the stereoselectivity in favour of the β-anomer, Robins et al.(Robins, R. L. et al., J. Am. Chem. Soc. 1984, 106, 6379-6382, US4760137 , EP0173059 ) provided an improved method in which the sodium salt of 2,6-dichloropurine was coupled with 1-chloro-2-deoxy-3,5-di-O–p-toluoyl-α-D-ribofuranose in acetonitrile (MeCN) to give the protected β-nucleoside in 59% isolated yield, following chromatography and crystallisation, in addition to 13% of the undesired N-7 regioisomer (see Scheme 3). The apparently higher selectivity in this coupling reaction is attributed to it being a direct SN2 displacement of the chloride ion by the purine sodium salt. The protected N-9 2′-deoxy-β-nucleoside was treated with methanolic ammonia at 100°C to give cladribine in an overall 42% yield. The drawback of this process is that the nucleophilic 7- position nitrogen competes in the SN2 reaction against the nucleophilic 9- position, leading to a mixture of the N-7 and N-9 glycosyl isomers as well as the need for chromatography and crystallisation to obtain the pure desired isomer.
Gerszberg and Alonso (Gerszberg S. and Alonso, D. WO0064918 , and US20020052491 ) also utilised an SN2 approach with 1-chloro-2-deoxy-3,5-di-O–p-toluoyl-α-D-ribofuranose but instead coupled it with the sodium salt of 2-chloroadenine in acetone giving the desired β-anomer of the protected cladribine in 60% yield following crystallisation from ethanol (see Scheme 4). After the deprotection step using ammonia in methanol (MeOH), the β-anomer of cladribine was isolated in an overall 42% yield based on the 1-chlorosugar, and 30% if calculated based on the sodium salt since this was used in a 2.3 molar excess.
To increase the regioselectivity towards glycosylation of the N-9 position, Gupta and Munk recently ( Gupta, P. K. and Munk, S. A., US20040039190 , WO2004018490 and CA2493724 ) conducted an SN2 reaction using the anomerically pure α-anomer, 1-chloro-2-deoxy-3,5-di-O–p-toluoyl-α-D-ribofuranose but coupling it with the potassium salt of a 6-heptanoylamido modified purine (see Scheme 5). The bulky alkyl group probably imparted steric hindrance around the N-7 position, resulting in the reported improved regioselectivity. Despite this, following deprotection, the overall yield of cladribine based on the 1-chlorosugar was 43%, showing no large improvement in overall yield on related methods. Moreover 2-chloroadenine required prior acylation with heptanoic anhydride at high temperature (130°C) in 72% yield, and the coupling required cryogenic cooling (-30°C) and the use of the strong base potassium hexamethyldisilazide and was followed by column chromatography to purify the product protected cladribine.
More recently Robins et al. (Robins, M. J. et al., J. Org. Chem. 2006, 71, 7773-7779, US20080207891 ) published a procedure for synthesis of cladribine that purports to achieve almost quantitative yields in the N-9-regioselective glycosylation of 6-(substituted-imidazol-1-yl)-purine sodium salts with 1-chloro-2-deoxy-3,5-di-O–p-toluoyl-α-D-ribofuranose in MeCN/dichloromethane (DCM) mixtures to give small or no detectable amounts of the undesired α-anomer (see Scheme 6). In actuality this was only demonstrated on the multi-milligram to several grams scale, and whilst the actual coupling yield following chromatography of the desired N-9-β-anomer was high (83% to quantitative), the protected 6-(substituted-imidazol-1-yl)-products were obtained in 55% to 76% yield after recrystallisation. Following this, toxic benzyl iodide was used to activate the 6-(imidazole-1-yl) groups which were then subsequently displaced by ammonia at 60-80°C in methanolic ammonia to give cladribine in 59-70% yield following ion exchange chromatography and multiple crystallisations, or following extraction with DCM and crystallisation. Although high anomeric and regioselective glycosylation was demonstrated the procedure is longer than the prior arts, atom uneconomic and not readily applicable to industrial synthesis of cladribine such as due to the reliance on chromatography and the requirement for a pressure vessel in the substitution of the 6-(substituted-imidazole-1-yl) groups.
EXAMPLE 1 Preparation of 2-chloro-6-trimethylsilylamino-9-[3,5-di-O-(4-chlorobenzoyl)-2-deoxy-β-D-ribofuranosyl]-purine
- [0052]
2-Chloroadenine (75 g, 0.44 mol, 1.0 eq.), MeCN (900 mL, 12 P), and BSTFA (343.5 g, 1.33 mol, 3.0 eq.) were stirred and heated under reflux until the mixture was almost turned clear. The mixture was cooled to 60°C and TfOH (7.9 mL, 0.089 mol, 0.2 eq.) and then 1-O-acetyl-3,5-di-O-(4-chlorobenzoyl)-2-deoxy-D-ribofuranose (III; 200.6 g, 1.0 eq.) were added into the mixture, and then the mixture was stirred at 60°C. After 1 hour, some solid precipitated from the solution and the mixture was heated for at least a further 10 hours. The mixture was cooled to r.t. and stirred for 2 hours. The solid was filtered and dried in vacuo at 60°C to give 180.6 g in 64% yield of a mixture of 2-chloro-6-trimethylsilylamino-9-[3,5-di-O-(4-chlorobenzoyl)-2-deoxy-β-D-ribofurano syl]-purine (IVa) with 95.4% HPLC purity and its non-silylated derivative 2-chloro-6-amino-9-[3,5-di-O-(4-chlorobenzoyl)-2′-deoxy-β-D-ribofuranosyl]-purine (IVb) with 1.1 % HPLC purity.
EXAMPLE 2 Preparation of 2-chloro-6-trimethylsilylamino-9-[3,5-di-O-(4-chlorobenzoyl)-2-deoxy-β-D-ribofuranosyl]-purine by isomerisation of a mixture of 2-chloro-6-amino-9-[3,5-di-O-(4-chlorobenzoyl)-2-deoxy-α,β-D-ribofuranosyl]-purine mixture
- [0053]
50.0 g of 2-chloro-6-amino-9-[3,5-di-O-(4-chlorobenzoyl)-2-deoxy-α,β-D-ribofuranosyl]-purine as a 0.6:1.0 mixture of the β-anomer IVb and α-anomer Vb(83.16 mmol, assay of α-anomer was 58.6% (52.06 mmol) and β-anomer was 34.3% (31.10 mmol, 17.15 g)), 68.6 g BSTFA (266.5 mmol) and 180 mL of MeCN (3.6 P) were charged into a dried 4-necked flask. The mixture was heated to 60°C under N2 for about 3 h and then 2.67 g of TfOH (17.8 mmol) was added. The mixture was stirred at 60°C for 15 h and was then cooled to about 25°C and stirred for a further 2 h, and then filtered. The filter cake was washed twice with MeCN (20 mL each) and dried at 60°C in vacuo for 6 h to give 24 g of off-white solid (the assay of 2-chloro-6-amino-9-[3,5-di-O-(4-chlorobenzoyl)-2-deoxy-α-D-ribofuranosyl]-purine was 1.4% (0.60 mmol, 0.34 g),
2-chloro-6-amino-9-[3,5-di-O-(4-chlorobenzoyl)-2-deoxy-β-D-ribofuranosyl]-purine was 8.4% (3.18 mmol, 2.02 g) and
2-chloro-6-trimethylsilylamino-9-[3,5-di-O-(4-chlorobenzoyl)-2-deoxy-β-D-ribofuranosyl]-purine was 86.6% (32.73 mmol, 20.78 g)).
Analysis of the 274.8 g of the mother liquor by assay showed that it in addition to the α-anomer it contained 0.5% (1.37 g, 2.43 mmol) of
2-chloro-6-amino-9-[3,5-di-O-(4-chlorobenzoyl)-2-deoxy-β-D-ribofuranosyl]-purine and 0.01% (0.027 g, 0.05 mmol) of
2-chloro-6-trimethylsilylamino-9-[3,5-di-O-(4-chlorobenzoyl)-2-deoxy-β-D-ribofuranosyl]-purine.
EXAMPLE 3 Preparation of 2-chloro-2′-deoxy-adenosine (cladribine)
- [0054]
To the above prepared mixture of 2-chloro-6-trimethylsilylamino-9-[3,5-di-O-(4-chlorobenzoyl)-2-deoxy-β-D-ribofurano syl]- purine (IVa) and 2-chloro-6-amino-9-[3,5-di-O-(4-chlorobenzoyl)-2′-deoxy-β-D-ribofuranosyl]-purine (IVb) (179 g, >95.4% HPLC purity) in MeOH (895 mL, 5 P) was added 29% MeONa/MeOH solution (5.25 g, 0.1 eq.) at 20-30°C. The mixture was stirred at 20-30°C for 6 hours, the solid was filtered, washed with MeOH (60 mL, 0.34 P) and then dried in vacuo at 50°C for 6 hour to give 72 g white to off-white crude cladribine with 98.9% HPLC purity in ca. 93% yield.
EXAMPLE 4 Recrystallisation
- [0055]
Crude cladribine (70 g), H2O (350 mL, 5 P), MeOH (350 mL, 5 P) and 29% MeONa/MeOH solution (0.17 g) were stirred and heated under reflux until the mixture turned clear. The mixture was stirred for 3 hour and was then filtered to remove the precipitates at 74-78°C. The mixture was stirred and heated under reflux until the mixture turned clear and was then cooled. Crystals started to form at ca. 45°C. The slurry was stirred for 2 hour at the cloudy point. The slurry was cooled slowly at a rate of 5°C/0.5 hour. The slurry was stirred at 10-20°C for 4-8 hours and then filtered. The filter cake was washed three times with MeOH (50 mL each) and dried at 50°C in vacuo for 6 hours to give 62.7 g of 99.9% HPLC pure cladribine in ca. 90% yield.
EXAMPLE 5 Preparation of 2-chloro-6-trimethylsilylamino-9-[3,5-di-O-(4-chlorobenzoyl)-2-deoxy-β-D-ribofuranosyl]-purine
- [0056]
2-Chloroadenine (2.2 Kg, 13.0 mol, 1.0 eq.), MeCN (20.7 Kg, 12 P), and BSTFA (10.0 Kg, 38.9 mol, 3.0 eq.) were stirred and heated under reflux for 3 hours and then filtered through celite and was cooled to about 60°C. TfOH (0.40 Kg, 2.6 mol, 0.2 eq.) and 1-O-acetyl-3,5-di-O-(4-chlorobenzoyl)-2-deoxy-D-ribofuranose (III; 5.87 Kg, 13.0 mol, 1.0 eq.) were added into the filtrate and the mixture was stirred at about 60°C for 29.5 hours. The slurry was cooled to about 20°C and stirred for 2 hours. The solids were filtered and washed with MeCN (2.8 Kg) twice and dried in vacuo at 60°C to give 5.17 Kg with a 96.5% HPLC purity in 62% yield of a mixture of 2-chloro-6-trimethylsilylamino-9-[3,5-di-O-(4-chlorobenzoyl)-2-deoxy-β-D-ribofurano syl]-purine (IVa), and non-silylated derivative 2-chloro-6-amino-9-[3,5-di-O-(4-chlorobenzoyl)-2′-deoxy-β-D-ribofuranosyl]-purine (IVb).
EXAMPLE 6 Preparation of 2-chloro-2′-deoxy-adenosine (cladribine)
- [0057]
To a mixture of 25% sodium methoxide in MeOH (0.11 Kg, 0.5 mol, 0.1 eq.) and MeOH (14.8 Kg, 5 P) at about at 25°C was added 2-chloro-6-trimethylsilylamino-9-[3,5-di-O-(4-chlorobenzoyl)-2-deoxy-β-D-ribofurano syl]-purine (IVa) and non-silylated derivative 2-chloro-6-amino-9-[3,5-di-O-(4-chlorobenzoyl)-2′-deoxy-β-D-ribofuranosyl]-purine (IVb) (3.70 Kg, combined HPLC purity of >96.3%) and the mixture was agitated at about 25°C for 2 hours. The solids were filtered, washed with MeOH (1.11 Kg, 0.4 P) and then dried in vacuo at 60°C for 4 hours to give 1.43 Kg of a crude cladribine with 97.8% HPLC purity in ca. 87% yield.
EXAMPLE 7 Recrystallisation of crude cladribine
- [0058]
A mixture of crude cladribine (1.94 Kg, >96.0% HPLC purity), MeOH (7.77 Kg, 5 P), process purified water (9.67 Kg, 5 P) and 25% sodium methoxide in MeOH (32 g, 0.15 mol) were stirred and heated under reflux until the solids dissolved. The solution was cooled to about 70°C and treated with activated carbon (0.16 Kg) and celite for 1 hour at about 70°C, rinsed with a mixture of preheated MeOH and process purified water (W/W = 1:1.25, 1.75 Kg). The filtrate was cooled to about 45°C and maintained at this temperature for 1 hours, and then cooled to about 15°C and agitated at this temperature for 2 hours. The solids were filtered and washed with MeOH (1.0 Kg, 0.7 P) three times and were then dried in vacuo at 60°C for 4 hours giving API grade cladribine (1.5 Kg, 5.2 mol) in 80% yield with 99.84% HPLC purity.
EXAMPLE 8 Recrystallisation of crude cladribine
- [0059]
A mixture of crude cladribine (1.92 Kg, >95.7% HPLC purity), MeOH (7.76 Kg, 5 P), process purified water (9.67 Kg, 5 P) and 25% sodium methoxide in MeOH (36 g, 0.17 mol) were stirred and heated under reflux until the solids dissolved. The solution was cooled to about 70°C and treated with activated carbon (0.15 Kg) and celite for 1 hour at about 70°C, rinsed with a mixture of preheated MeOH and process purified water (1:1.25, 1.74 Kg). The filtrate was cooled to about 45°C and maintained at this temperature for 1 hour, and then cooled to about 15°C and agitated at this temperature for 2 hours. The solids were filtered and washed with MeOH (1.0 Kg, 0.7 P) three times and were giving damp cladribine (1.83 Kg). A mixture of this cladribine (1.83 Kg), MeOH (7.33 Kg, 5 P) and process purified water (9.11 Kg, 5 P) were stirred and heated under reflux until the solids dissolved and was then cooled to about 45°C and maintained at this temperature for 1 hours. The slurry was further cooled to about 15°C and agitated at this temperature for 2 hours. The solids were filtered and washed with MeOH (0.9 Kg, 0.7 P) three times and were then dried in vacuo at 60°C for 4 hours giving API grade cladribine (1.38 Kg, 4.8 mol) in 75% yield with 99.86% HPLC purity.
SYN

Cladribine can be got from 2-Deoxy-D-ribose. The detail is as follows:

SYN
https://www.tandfonline.com/doi/abs/10.1080/15257770.2015.1071848?journalCode=lncn20
March 29, 2019
Release
The U.S. Food and Drug Administration today approved Mavenclad (cladribine) tablets to treat relapsing forms of multiple sclerosis (MS) in adults, to include relapsing-remitting disease and active secondary progressive disease. Mavenclad is not recommended for MS patients with clinically isolated syndrome. Because of its safety profile, the use of Mavenclad is generally recommended for patients who have had an inadequate response to, or are unable to tolerate, an alternate drug indicated for the treatment of MS.
“We are committed to supporting the development of safe and effective treatments for patients with multiple sclerosis,” said Billy Dunn, M.D., director of the Division of Neurology Products in the FDA’s Center for Drug Evaluation and Research. “The approval of Mavenclad represents an additional option for patients who have tried another treatment without success.”
MS is a chronic, inflammatory, autoimmune disease of the central nervous system that disrupts communications between the brain and other parts of the body. Most people experience their first symptoms of MS between the ages of 20 and 40. MS is among the most common causes of neurological disability in young adults and occurs more frequently in women than in men.
For most people, MS starts with a relapsing-remitting course, in which episodes of worsening function (relapses) are followed by recovery periods (remissions). These remissions may not be complete and may leave patients with some degree of residual disability. Many, but not all, patients with MS experience some degree of persistent disability that gradually worsens over time. In some patients, disability may progress independent of relapses, a process termed secondary progressive multiple sclerosis (SPMS). In the first few years of this process, many patients continue to experience relapses, a phase of the disease described as active SPMS. Active SPMS is one of the relapsing forms of MS, and drugs approved for the treatment of relapsing forms of MS can be used to treat active SPMS.
The efficacy of Mavenclad was shown in a clinical trial in 1,326 patients with relapsing forms of MS who had least one relapse in the previous 12 months. Mavenclad significantly decreased the number of relapses experienced by these patients compared to placebo. Mavenclad also reduced the progression of disability compared to placebo.
Mavenclad must be dispensed with a patient Medication Guide that describes important information about the drug’s uses and risks. Mavenclad has a Boxed Warning for an increased risk of malignancy and fetal harm. Mavenclad is not to be used in patients with current malignancy. In patients with prior malignancy or with increased risk of malignancy, health care professionals should evaluate the benefits and risks of the use of Mavenclad on an individual patient basis. Health care professionals should follow standard cancer screening guidelines in patients treated with Mavenclad. The drug should not be used in pregnant women and in women and men of reproductive potential who do not plan to use effective contraception during treatment and for six months after the course of therapy because of the potential for fetal harm. Mavenclad should be stopped if the patient becomes pregnant.
Other warnings include the risk of decreased lymphocyte (white blood cell) counts; lymphocyte counts should be monitored before, during and after treatment. Mavenclad may increase the risk of infections; health care professionals should screen patients for infections and treatment with Mavenclad should be delayed if necessary. Mavenclad may cause hematologic toxicity and bone marrow suppression so health care professionals should measure a patient’s complete blood counts before, during and after therapy. The drug has been associated with graft-versus-host-disease following blood transfusions with non-irradiated blood. Mavenclad may cause liver injury and treatment should be interrupted or discontinued, as appropriate, if clinically significant liver injury is suspected.
The most common adverse reactions reported by patients receiving Mavenclad in the clinical trials include upper respiratory tract infections, headache and decreased lymphocyte counts.
The FDA granted approval of Mavenclad to EMD Serono, Inc.
References
- ^ Drugs.com International trade names for Cladribine Page accessed Jan 14, 2015
- ^ Jump up to:a b c d “PRODUCT INFORMATION LITAK© 2 mg/mL solution for injection” (PDF). TGA eBusiness Services. St Leonards, Australia: Orphan Australia Pty. Ltd. 10 May 2010. Retrieved 27 November 2014.
- ^ Liliemark, Jan (1997). “The Clinical Pharmacokinetics of Cladribine”. Clinical Pharmacokinetics. 32 (2): 120–131. doi:10.2165/00003088-199732020-00003. PMID 9068927.
- ^ Jump up to:a b “European Medicines Agency – – Litak”. http://www.ema.europa.eu.
- ^ Jump up to:a b “Leustat Injection. – Summary of Product Characteristics (SPC) – (eMC)”. http://www.medicines.org.uk.
- ^ Leist, TP; Weissert, R (2010). “Cladribine: mode of action and implications for treatment of multiple sclerosis”. Clinical Neuropharmacology. 34 (1): 28–35. doi:10.1097/wnf.0b013e318204cd90. PMID 21242742.
- ^ Jump up to:a b c d Cladribine label, last updated July 2012. Page accessed January 14, 2015
- ^ https://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm634837.htm
- ^ Histiocytosis Association Erdheim-Chester Disease Page accessed Aug 20, 2016
- ^ Aricò M (2016). “Langerhans cell histiocytosis in children: from the bench to bedside for an updated therapy”. Br J Haematol. 173 (5): 663–70. doi:10.1111/bjh.13955. PMID 26913480.
The combination of cytarabine and cladribine is the current standard for second-line therapy of refractory cases with vital organ dysfunction.
- ^ Jump up to:a b c Giovannoni, G; Comi, G; Cook, S; Rammohan, K; Rieckmann, P; Soelberg Sørensen, P; Vermersch, P; Chang, P; Hamlett, A; Musch, B; Greenberg, SJ; CLARITY Study, Group. (4 February 2010). “A placebo-controlled trial of oral cladribine for relapsing multiple sclerosis”. The New England Journal of Medicine. 362 (5): 416–26. doi:10.1056/NEJMoa0902533. PMID 20089960.
- ^ Johnston, JB (June 2011). “Mechanism of action of pentostatin and cladribine in hairy cell leukemia”. Leukemia & Lymphoma. 52 Suppl 2: 43–5. doi:10.3109/10428194.2011.570394. PMID 21463108.
- ^ Beutler, E; Piro, LD; Saven, A; Kay, AC; McMillan, R; Longmire, R; Carrera, CJ; Morin, P; Carson, DA (1991). “2-Chlorodeoxyadenosine (2-CdA): A Potent Chemotherapeutic and Immunosuppressive Nucleoside”. Leukemia & Lymphoma. 5 (1): 1–8. doi:10.3109/10428199109068099. PMID 27463204.
- ^ Baker, D; Marta, M; Pryce, G; Giovannoni, G; Schmierer, K (February 2017). “Memory B Cells are Major Targets for Effective Immunotherapy in Relapsing Multiple Sclerosis”. EBioMedicine. 16: 41–50. doi:10.1016/j.ebiom.2017.01.042. PMC 5474520. PMID 28161400.
- ^ Baker, D; Herrod, SS; Alvarez-Gonzalez, C; Zalewski, L; Albor, C; Schmierer, K (July 2017). “Both cladribine and alemtuzumab may effect MS via B-cell depletion”. Neurology: Neuroimmunology & Neuroinflammation. 4 (4): e360. doi:10.1212/NXI.0000000000000360. PMC 5459792. PMID 28626781.
- ^ Jump up to:a b Ceronie, B; Jacobs, BM; Baker, D; Dubuisson, N; Mao, Z; Ammoscato, F; Lock, H; Longhurst, HJ; Giovannoni, G; Schmierer, K (May 2018). “Cladribine treatment of multiple sclerosis is associated with depletion of memory B cells”. Journal of Neurology. 265 (5): 1199–1209. doi:10.1007/s00415-018-8830-y. PMC 5937883. PMID 29550884.
- ^ Jump up to:a b Marshall A. Lichtman Biographical Memoir: Ernest Beutler 1928–2008 National Academy of Sciences, 2012
- ^ Staff, The Pink Sheet Mar 8, 1993 Ortho Biotech’s Leustatin For Hairy Cell Leukemia
- ^ Jump up to:a b EMA 2004 Litak EMA package: Scientific Discussion
- ^ EMA 2004 Litak: Background Information one the Procedure
- ^ Eric Sauter and Mika Ono for Scripps News and Views. Vol 9. Issue 18. June 1, 2009 A Potential New MS Treatment’s Long and Winding Road
- ^ Tortorella C, Rovaris M, Filippi M (2001). “Cladribine. Ortho Biotech Inc”. Curr Opin Investig Drugs. 2 (12): 1751–6. PMID 11892941.
- ^ Jump up to:a b Carey Sargent for Dow Jones Newswires in the Wall Street Journal. Oct. 31, 2002 Serono Purchases Rights To Experimental MS Drug
- ^ Reuters. Dec 4, 2000. Ivax to Develop Cladribine for Multiple Sclerosis
- ^ Jennifer Bayot for the New York Times. July 26, 2005 Teva to Acquire Ivax, Another Maker of Generic Drugs
- ^ Teva Press Release, 2006. Teva Completes Acquisition of Ivax
- ^ Staff, First Word Pharma. Sept 21, 2006 Merck KGaA to acquire Serono
- ^ Jump up to:a b c EMA. 2011 Withdrawal Assessment Report for Movectro Procedure No. EMEA/H/C/001197
- ^ Jump up to:a b John Gever for MedPage Today June 22, 2011 06.22.2011 0 Merck KGaA Throws in Towel on Cladribine for MS
- ^ Jump up to:a b John Carroll for FierceBiotech Sep 11, 2015 Four years after a transatlantic slapdown, Merck KGaA will once again seek cladribine OK
- ^ Connolly, Allison (24 April 2012). “Merck KGaA to Close Merck Serono Site in Geneva, Cut Jobs”. Bloomberg.
- ^ Pakpoor, J; et al. (December 2015). “No evidence for higher risk of cancer in patients with multiple sclerosis taking cladribine”. Neurology: Neuroimmunology & Neuroinflammation. 2 (6): e158. doi:10.1212/nxi.0000000000000158. PMC 4592538. PMID 26468472.
- ^ Press release
- ^ Merck. “Cladribine Tablets Receives Positive CHMP Opinion for Treatment of Relapsing Forms of Multiple Sclerosis”. http://www.prnewswire.co.uk. Retrieved 2017-08-22.
- ^ Cladribine approved in Europe, Press Release
- ^ Jump up to:a b Giovannoni, G; Soelberg Sorensen, P; Cook, S; Rammohan, K; Rieckmann, P; Comi, G; Dangond, F; Adeniji, AK; Vermersch, P (1 August 2017). “Safety and efficacy of cladribine tablets in patients with relapsing-remitting multiple sclerosis: Results from the randomized extension trial of the CLARITY study”. Multiple Sclerosis (Houndmills, Basingstoke, England): 1352458517727603. doi:10.1177/1352458517727603. PMID 28870107.
- ^ “Sustained Efficacy – Merck Neurology”. Merck Neurology. Retrieved 28 September2018.
- ^ Guarnera, C; Bramanti, P; Mazzon, E (2017). “Alemtuzumab: a review of efficacy and risks in the treatment of relapsing remitting multiple sclerosis”. Therapeutics and Clinical Risk Management. 13: 871–879. doi:10.2147/TCRM.S134398. PMC 5522829. PMID 28761351.
- ^ Baker, D; Herrod, SS; Alvarez-Gonzalez, C; Giovannoni, G; Schmierer, K (1 August 2017). “Interpreting Lymphocyte Reconstitution Data From the Pivotal Phase 3 Trials of Alemtuzumab”. JAMA Neurology. 74 (8): 961–969. doi:10.1001/jamaneurol.2017.0676. PMC 5710323. PMID 28604916.
- ^ “Cladribine tablets for treating relapsing–remitting multiple sclerosis”. National Institute for Clinical Excellence. Retrieved 23 September 2018.
- ^ Hasanali, Zainul S.; Saroya, Bikramajit Singh; Stuart, August; Shimko, Sara; Evans, Juanita; Shah, Mithun Vinod; Sharma, Kamal; Leshchenko, Violetta V.; Parekh, Samir (24 June 2015). “Epigenetic therapy overcomes treatment resistance in T cell prolymphocytic leukemia”. Science Translational Medicine. 7 (293): 293ra102. doi:10.1126/scitranslmed.aaa5079. ISSN 1946-6234. PMC 4807901. PMID 26109102.
 |
|
| Clinical data | |
|---|---|
| Trade names | Leustatin, others[1] |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a693015 |
| License data | |
| Pregnancy category |
|
| Routes of administration |
Intravenous, subcutaneous(liquid) |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 100% (i.v.); 37 to 51% (orally)[3] |
| Protein binding | 25% (range 5-50%)[2] |
| Metabolism | Mostly via intracellularkinases; 15-18% is excreted unchanged[2] |
| Elimination half-life | Terminal elimination half-life: Approximately 10 hours after both intravenous infusion an subcutaneous bolus injection[2] |
| Excretion | Urinary[2] |
| Identifiers | |
| CAS Number | |
| PubChemCID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| ECHA InfoCard | 100.164.726 |
| Chemical and physical data | |
| Formula | C10H12ClN5O3 |
| Molar mass | 285.687 g/mol g·mol−1 |
| 3D model (JSmol) | |
E 2212
C25 H23 F3 N6 O, 480.48
CAS 1123197-68-1
(+) -2-{(E)-2-[5-methoxy-6-(4-methyl-1H-imidazol-1-yl)pyridin-3-yl]vinyl}-8-(2-trifluoromethylphenyl)-5,6,7,8-tetrahydro-[1,2,4]triazolo[1,5-a]pyridine
- (+)-5,6,7,8-Tetrahydro-2-[(1E)-2-[5-methoxy-6-(4-methyl-1H-imidazol-1-yl)-3-pyridinyl]ethenyl]-8-[2-(trifluoromethyl)phenyl][1,2,4]triazolo[1,5-a]pyridine
- (+)-2-[(E)-2-[5-Methoxy-6-(4-methyl-1H-imidazol-1-yl)pyridin-3-yl]ethenyl]-8-(2-trifluoromethylphenyl)-5,6,7,8-tetrahydro-[1,2,4]triazolo[1,5-a]pyridine

E2212
CAS 1123197-82-9
- C25 H23 F3 N6 O . 3/2 C4 H6 O6
- [1,2,4]Triazolo[1,5-a]pyridine, 5,6,7,8-tetrahydro-2-[(1E)-2-[6-methoxy-5-(4-methyl-1H-imidazol-1-yl)-2-pyridinyl]ethenyl]-8-[2-(trifluoromethyl)phenyl]-, (8S)-, (2S,3S)-2,3-dihydroxybutanedioate (2:3)
PATENT
https://patents.google.com/patent/US9453000B2/en
Examples 394 and 395 Synthesis of (+) and (−)-2-{(E)-2-[5-methoxy-6-(4-methyl-1H-imidazol-1-yl)pyridin-3-yl]vinyl}-8-(2-trifluoromethylphenyl)-5,6,7,8-tetrahydro-[1,2,4]triazolo[1,5-a]pyridine
230 mg of the racemic title compound was obtained from 1-amino-3-(2-trifluoromethylphenyl)piperidin-2-one (343 mg) and (E)-3-[5-methoxy-6-(4-methyl-1H-imidazol-1-yl)pyridin-3-yl]acrylic acid (500 mg) by the same method as in Examples 194 and 195. The racemic title compound (220 mg) was separated by CHIRALPAK™ IC manufactured by Daicel Chemical Industries, Ltd. (2 cm×25 cm; mobile phase: methanol) to obtain the title optically active compound with positive optical rotation and a retention time of 16 minutes (92 mg) and the title optically active compound with negative optical rotation and a retention time of 19 minutes (79 mg).
The property value of the title optically active compound with a retention time of 16 minutes is as follows.
ESI-MS; m/z 481 [M++H].
The property values of the title optically active compound with a retention time of 19 minutes are as follows.
ESI-MS; m/z 481 [M++H]. 1H-NMR (CDCl3) δ (ppm): 1.90-2.01 (m, 1H), 2.10-2.35 (m, 2H), 2.29 (s, 3H), 2.43-2.52 (m, 1H), 3.95 (s, 3H), 4.27-4.41 (m, 2H), 4.69 (dd, J=6.0, 8.4 Hz, 1H), 7.02 (d, J=8.0 Hz, 1H), 7.08 (d, J=16.4 Hz, 1H), 7.40 (dd, J=7.6, 7.6 Hz, 1H), 7.44-7.53 (m, 4H), 7.73 (d, J=8.0 Hz, 1H), 8.13 (d, J=1.6 Hz, 1H), 8.34 (s, 1H).
PATENT
WO2009028588
https://patentscope.wipo.int/search/en/detail.jsf;jsessionid=D6AD22B6CC7302560AE1ADCED305CDCE.wapp2nC?docId=WO2009028588&tab=FULLTEXT&queryString=%28PA%2Feisai%29%2520&recNum=93&maxRec=725
(+)および(-)-2-{(E)-2-[5-メトキシ-6-(4-メチル-1H-イミダゾール-1-イル)ピリジン-3-イル]ビニル}-8-(2-トリフルオロメチルフェニル)-5,6,7,8-テトラヒドロ-[1,2,4]トリアゾロ[1,5-a]ピリジンの合成
[化221]
実施例194および実施例195と同様の方法により、1-アミノ-3-(2-トリフルオロメチルフェニル)ピペリジン-2-オン(343mg)および(E)-3-[5-メトキシ-6-(4-メチル-1H-イミダゾール-1-イル)ピリジン-3-イル]アクリル酸(500mg)から、ラセミ体の表題化合物を230mg得た。ラセミ体の表題化合物(220mg)をダイセル製CHIRALPAK TM IC(2cm×25cm:移動相;メタノール)にて分取し、(+)の旋光性を有する保持時間16分の表題光学活性化合物(92mg)および(-)の旋光性を有する保持時間19分の表題光学活性化合物(79mg)を得た。
保持時間16分の表題光学活性体の物性値は以下の通りである。
ESI-MS;m/z 481[M ++H].
保持時間19分の表題光学活性体の物性値は以下の通りである。
ESI-MS;m/z 481[M ++H]. 1H-NMR(CDCl 3)δ(ppm):1.90-2.01(m,1H),2.10-2.35(m,2H),2.29(s,3H),2.43-2.52(m,1H),3.95(s,3H),4.27-4.41(m,2H),4.69(dd,J=6.0,8.4Hz,1H),7.02(d,J=8.0Hz,1H),7.08(d,J=16.4Hz,1H),7.40(dd,J=7.6,7.6Hz,1H),7.44-7.53(m,4H),7.73(d,J=8.0Hz,1H),8.13(d,J=1.6Hz,1H),8.34(s,1H).
Example 394 and Example 395
(+) and (−)-2-{(E) -2- [5-methoxy-6- (4-methyl-1H-imidazol-1-yl) pyridin-3-yl] Synthesis of vinyl} -8- (2-trifluoromethylphenyl) -5,6,7,8-tetrahydro- [1,2,4] triazolo [1,5-a] pyridine [Formula
221]
Example 194 and By a method similar to Example 195, 1-amino-3- (2-trifluoromethylphenyl) piperidin-2-one (343 mg) and (E) -3- [5-methoxy-6- (4-methyl-) 1 H-Imidazol-1-yl) pyridin-3-yl] acrylic acid (500 mg) gave 230 mg of the racemic title compound. Racemic title compound (220 mg) a Daicel CHIRALPAK TM IC (2 cm × 25 cm: mobile phase; methanol) was collected by min (+) title optically active compound of the retention time of 16 minutes with a optical rotation of (92 mg) The title optically active compound (79 mg) having a polarizability of (−) and a retention time of 19 minutes was obtained.
The physical property values of the title optically active substance with a retention time of 16 minutes are as follows.
ESI-MS; m / z 481 [M + + H].
The physical property values of the title optically active substance with a retention time of 19 minutes are as follows.
ESI-MS; m / z 481 [M + + H]. 1 H-NMR (CDCl 3)) Δ (ppm): 1.90 to 2.01 (m, 1 H), 2.10 to 2.35 (m, 2 H), 2.29 (s, 3 H), 2.43 to 2.52 (m) , 1 H), 3.95 (s, 3 H), 4.27-4. 41 (m, 2 H), 4.69 (dd, J = 6.0, 8.4 Hz, 1 H), 7.02 (d , J = 8.0 Hz, 1 H), 7.08 (d, J = 16.4 Hz, 1 H), 7.40 (dd, J = 7.6, 7.6 Hz, 1 H), 7.44-7. 53 (m, 4H), 7.73 (d, J = 8.0 Hz, 1 H), 8.13 (d, J = 1.6 Hz, 1 H), 8.34 (s, 1 H).
PATENT
https://patents.google.com/patent/WO2010098490A1/it

As a novel compound that has an effect of reducing the production of Aβ40 and
42 and is expected as a therapeutic or prophylactic agent for Alzheimer’s disease or the like, the present inventors have found a compound represented by the following formula (1) (compound
(D): [Formula 1]
and filed a patent application for the invention (PCT/JP08/065365).
Generally, properties of salts of compounds and those crystals that are useful as pharmaceuticals are highly important for the development of pharmaceuticals, because the properties greatly affect bioavailability of drugs, purity of drug substances, formulation of preparations, and the like. Therefore, it is necessary to research which salts and crystal forms of the compound of the formula (1) are most excellent as pharmaceuticals. Specifically, since their properties depend on the character of the individual compounds, it is generally difficult to estimate salts and crystal forms for drug substances having excellent properties and it is demanded to actually make various studies for each compound.
EXAMPLES [0023] The present invention will be described in detail below with reference to reference examples and examples; however, the present invention is not limited to these reference examples and examples. [0024]
The following abbreviations are used in the following reference examples and examples.
DMF: N,N’-dimethylformamide
THF: Tetrahydrofuran
EDC: lrEmyl-S-β-dimemylammopropytycarbodiimide hydrochloride HOBT: 1-Hydroxybenzotriazole IPEA: Diisopropylethylamine [0025]
In powder X-ray diffractometry of the crystals produced in the following examples, the resulting crystals were placed on a sample stage of a powder X-ray diffractometer and analyzed under the following conditions. [0026] Measurement conditions
Sample holder: Aluminum Target: Copper
Detector: Scintillation counter Tube voltage: 50 kV Tube current: 300 mA
Slit: DS 0.5 mm (Height limiting slit 2 mm), SS Open, RS Open Scanning rate : 5 °/min
Sampling interval: 0.02° Scan range: 5 to 35° Goniometer: Horizontal goniometer [0027] Reference Example 1
Svnmesis ofr8SV2-(fE)-246-memoxy-5-(4-memyl-lH-imidazol-l-vnpyridin-2-yllvmvU-8-(2-trifluoromethylphenyl‘)-5,6J,8-tetrahvdro-[1.2,41triazolo[l.,5-a]pyridine
[Formula 2]
Synthesis of l-amino-3-(2-trifluoromemylphenyl)piperidin-2-one Thionyl chloride (2.72 mL) was added to a solution of 2-trifluoromethylphenylacetic acid (1.9 g) in methanol (38 mL), followed by stirring at room temperature for three hours. The reaction solution was concentrated under reduced pressure. The resulting residue was diluted with DMF. Sodium hydride (containing 40% mineral oil, 410 mg) was added under ice-cooling, followed by stirring for 10 minutes. The reaction solution was further stirred for 30 minutes and then ice-cooled again. l-Chloro-3-iodopropane (1.02 mL) was added to the reaction mixture, and the reaction solution was stirred at room temperature overnight. Water and ethyl acetate were added to the reaction mixture, and the organic layer was separated. The resulting organic layer was washed with saturated aqueous sodium chloride, dried over anhydrous magnesium sulfate and then concentrated under reduced pressure. The resulting residue was diluted with ethanol (26.6 mL). Hydrazine monohydrate (7.6 mL) was added, and the reaction solution was stirred at room temperature for two hours and then at 60°C for further three hours. The reaction mixture was concentrated under reduced pressure. Saturated aqueous sodium bicarbonate and ethyl acetate and were added to the residue, and the organic layer was separated. The resulting organic layer was washed with saturated aqueous sodium chloride, dried over anhydrous magnesium sulfate and then concentrated under reduced pressure. The residue was purified by silica gel column chromatography (carrier: Chromatorex NH; elution solvent: heptane-ethyl acetate system) to obtain 1.68 g of the title compound. The property values of the compound are as follows.
ESI-MS; m/z 259 [M+H-H]. 1H-NMR (CDCl3) δ (ppm): 1.82-2.10 (m, 3H), 2.18-2.26 (m, IH), 3.58-3.76 (m, 2H), 4.07 (dd, J = 10.0, 5.6 Hz, IH), 4.60 (s, 2H), 7.24 (d, J = 7.6 Hz, IH), 7.35 (t, J = 7.6 Hz, IH), 7.51 (t, J = 7.6 Hz, IH)5 7.66 (d, J = 7.6 Hz, IH). [0028] Synthesis of (EV3-[6-methoxy-5-(4-methyl- 1 H-imidazol- 1 -yl)pyridin-2-yl]-N-f2-oxo-3 -(2-trifluoromethylphenyl)piperidin- 1 -yl]acrylamide
EDC (834 mg), HOBT (588 mg) and IPEA (2.03 mL) were added to a suspension of (E)-3-[6-methoxy-5-(4-methyl-lH-imidazol-l-yl)pyridm-2-yl]acrylic acid trifluoroacetate (1.42 g) and l-amήio-3-(2-trifluoromethylphenyl)piperidin-2-one (750 mg) in DMF (30 mL). After stirring at room temperature for 14 hours, a saturated sodium bicarbonate solution and ethyl acetate were added to the reaction solution, and the organic layer was separated. The resulting organic layer was dried over anhydrous magnesium sulfate and then concentrated under reduced pressure. The residue was purified by silica gel column chromatography (carrier: Chromatorex NH; elution solvent: ethyl acetate-methanol system) to obtain 1.23 g of the title compound. The property values of the compound are as follows. ESI-MS; m/z 500 [M1H-HJ. [0029]
Synthesis of r8S>-2-(fEV2-r6-methoxy-5-r4-methyl-lH-imidazol-l-vnpyridm’2-vnvinvU-8-(2-trifluoromethvlphenvD-5.6.7.8-tetrahvdro-ri.2.41triazoloπ.5-a1pvridine Phosphorus oxychloride (24.2 mL) was added to (E)-3~[6-methoxy-5-(4-methyl-lH-imidazol-l-yl)pyridin-2-yl]-N-[2-oxo-3-(2-trifluoromethylphenyl)piperidin-l-yl]acrylamide (1.2 g). The reaction solution was stirred at 1000C for one hour and then concentrated under reduced pressure. Subsequently, the residue was diluted with acetic acid (24.2 mL) and then ammonium acetate (1.9 g) was added, followed by stirring at 1500C for two hours. The reaction solution was left to cool to room temperature and then concentrated under reduced pressure. A saturated sodium bicarbonate solution and ethyl acetate were added to the resulting residue, and the organic layer was separated. The resulting organic layer was dried over anhydrous magnesium sulfate and then concentrated under reduced pressure. The residue was purified by silica gel column chromatography (carrier: Chromatorex NH; elution solvent: heptane-ethyl acetate system) to obtain a racemate of the title compound (750 mg). The resulting racemate (410 mg) was separated by CHIRALP AK™ IA manufactured by Daicel Chemical Industries, Ltd. (2 cm x 25 cm, mobile phase: hexane:ethanol = 8:2, flow rate: 10 mL/min) to obtain the title compound with a retention time of 33 minutes and negative optical rotation (170 mg) as crystals. The property values of the title compound are as follows.
1H-NMR (CDCl3) δ (ppm): 1.90-2.01 (m, IH), 2.10-2.35 (m, 2H), 2.29 (d, J = 1.2 Hz, 3H), 2.42-2.51 (m, IH), 4.03 (s, 3H), 4.28-4.41 (m, 2H), 4.70 (dd, J = 8.4, 6.0 Hz, IH), 6.92 (d, J = 8.0 Hz, IH), 6.95 (t, J = 1.2 Hz, IH), 7.01 (d, J = 7.6 Hz, IH), 7.39 (t, J = 7.6 Hz5 IH), 7.44 (d, J = 16.0 Hz, IH), 7.45 (d, J = 8.0 Hz, IH), 7.49 (t, J = 7.6 Hz, IH), 7.63 (d, J = 16.0 Hz5 IH), 7.72 (d, J = 7.6 Hz, IH), 7.76 (d, J = 1.2 Hz, IH). [0030]
(8S)-2-{(E)-2-[6-Methoxy-5-(4-methyl-lH-imidazol-l-yl)ρyridin-2-yl]vinyl}-8-(2-trifluoromethylphenyl)-5,6,7,8-tetrahydro-[l,2,4]triazolo[l,5-a]pyridine synthesized according to the above reference example was used for the following synthesis of salts. [0031] Example 1
Synthesis of r8SV2-{rEV2-[6-methoxy-5-(4-methyl-lH-imidazol-l-vπpyridin-2-vnvinvU-8-f2-trifluoromethylphenyl)-5.6.7.8-tetrahvdro-fl,2,4]triazolo[l.,5-a]pyridine 1.5 D-tartrate
(8S)-2-{(E)-2-[6-methoxy-5-(4-methyl-lH-imidazol-l-yl)pyridin-2-yl]vinyl}-8-(2-trifluoromethylphenyl)-5,6,7,8-tetrahydro-[l,2,4]triazolo[l,5-a]pyridine (33.70 mg) was dissolved in 285 μL of a D-tartaric acid-ethanol solution (110.92 mg/3 mL) with stirring at room temperature. The oil was precipitated when 1 mL of heptane was added. Accordingly, the oily substance was dissolved by adding 1 mL of ethanol. Further, 0.5 mL of heptane was added, and the mixture was transferred to a low temperature laboratory at about 50C (under shading) and continuously stirred for 24 hours. Thus, partial gelation occurred. Thereafter, the mixture was brought back to room temperature and continuously stirred, resulting in precipitation of a solid. The solid was collected by filtration through a glass filter and dried under reduced pressure at room temperature to obtain 21.25 mg of the title compound as white solid crystals. 1H-NMR (600 MHz, DMSOd6) δ (ppm): 1.96 (m, IH), 2.14 (s, 3H), 2.16 (m, 2H), 2.29 (m, IH), 3.98 (s, 3H), 4.28 (m, 2H), 4.29 (s, 3H), 4.51 (dd, J = 9, 6 Hz, IH), 7.22 (s, IH), 7.25 (brd, J = 8 Hz, IH), 7.27 (d, J = 8 Hz, IH), 7.32 (d, J = 16 Hz, IH)5 7.46 (d, J = 16 Hz, IH), 7.49 (brdd, J = 8 Hz, IH), 7.61 (brdd, J = 8 Hz5 IH), 7.77 (brd, J = 8 Hz, IH), 7.78 (d, J = 8 Hz, IH), 7.91 (s, IH). [0032] Example 2
Synthesis of (‘8SV2-l(Ε)-2-f6-methoxy-5-(4-methyl-lH-imidazol-l-vnpyridm-2-yllvinyl>-8-f2-trifluoromethylphenylV5,6J,8-tetrahvdro-[l ,2,4]triazolo[l ,5-a]pyridine di-D-tartrate
(8S)-2-{(E)-2-[6-methoxy-5-(4-methyl-lH-imidazol-l-yl)pyridin-2-yl]vinyl}-8-(2-trifluoromethylphenyl)-5,657,8-tetrahydro-[l ,2,4]triazolo[l ,5-a]ρyridine (810.18 mg) was dissolved in 8 mL of a D-tartaric acid-ethanol solution (751.13 mg/10 mL) with stirring at room temperature. The oil was precipitated when 2 mL of heptane was added. Accordingly, the oily substance was dissolved by ultrasonic treatment to prepare a clear solution. Several mg of crystals of the 1.5 D-tartrate prepared according to Example 1 were added, followed by stirring at room temperature. Stirring for about one hour resulted in gelation and subsequent precipitation of a solid. Further, stirring was continued while gradually adding 14 mL of heptane. A part of the suspension (2 mL) was separated and the solid was collected by filtration through a glass filter. The solid was dried under reduced pressure at room temperature to obtain 71.14 mg of the title compound as white solid crystals. 1H-NMR (400 MHz, DMSOd6) δ (ppm): 1.97 (m, IH), 2.15 (s, 3H), 2.16 (m, 2H), 2.30 (m, IH), 3.98 (s, 3H), 4.28 (m, 2H), 4.29 (s, 4H), 4.51 (dd, J = 9, 6 Hz, IH), 7.22 (brs, IH), 7.25 (brd, J = 8 Hz, IH), 7.27 (d, J = 8 Hz, IH), 7.32 (d, J = 16 Hz, IH), 7.46 (d, J = 16 Hz, IH), 7.49 (brdd, J – 8 Hz, IH), 7.61 (brdd, J = 8 Hz, IH), 7.77 (brd, J = 8 Hz, IH), 7.78 (d, J = 8 Hz, IH), 7.91 (brs, IH). [0033] Example 3
Synthesis of r8SV2-(rE)-2-r6-methoxy-5-r4-methyl-lH-imidazol-l-vnpyridin-2-yl1vinvU-8-α-trifluoromethylphenyl)-5,6J,8-tetrahydro-[1.2,4]triazolo[l,5-a]pyridine disulfate
Concentrated sulfuric acid (11.5 μL) was added to a solution of (8S)-2-{(E)-2-[6- methoxy-5-(4-methyl-lH-imidazol-l-yl)pyridin-2-yl]vinyl}-8-(2-txifluoromethylphenyl)-5,6,7,8-tetrahydro-[l52,4]triazolo[l55-a]pyridine (98.09 mg) in ethanol (1 mL), and 1 mL of ethyl acetate was added with stirring at room temperature. Since the oily portion was confirmed on the bottom of the recovery flask, the oily substance was dissolved by ultrasonic treatment. Stirring at room temperature under shading for about 30 minutes resulted in precipitation of a solid. The solid was collected by filtration through a glass filter and dried under reduced pressure at room temperature to obtain 127.94 mg of the title compound as white solid crystals. 1H-NMR (400 MHz, DMSOd6) δ (ppm): 1.97 (m, IH), 2.17 (m, 2H), 2.30 (m, IH), 2.34 (brd, J = 1 Hz, 3H), 4.01 (s, 3H), 4.29 (m, 2H), 4.52 (dd, J = 9, 6 Hz, IH)5 7.25 (brd, J = 8 Hz, IH), 7.37 (d, J = 16 Hz, IH), 7.40 (d, J = 8 Hz, IH), 7.50 (brdd, J = 8 Hz, IH), 7.55 (d, J = 16 Hz, IH), 7.61 (brdd, J = 8 Hz, IH), 7.77 (m, IH), 7.78 (m, IH), 8.00 (d, J = 8 Hz, IH), 9.36 (d, J = 2 Hz, IH). [0034] Example 4 Synthesis of (8SV2-((E)-2-[“6-methoxy-5-(4-methyl-lH-imidazol-l-ylN)ρyridin-2-yllvinvU-8-(‘2-trifluoromethylphenyl)-5,6,7,8-tetrahvdiO-[1.2,41triazolo[l,5-a]pyridine dihydrobromide
Concentrated hydrobromic acid (24.8 μL) was added to a solution of (8S)-2-{(E)-2-[6-methoxy-5-(4-methyl-lH-imidazol-l-yl)pyridin-2-yl]vinyl}-8-(2-trifluoromethylphenyl)-5,6,7,84etrahydro-[l,254]triazolo[l55-a]pyridine (51.42 mg) m ethanol (1 mL), and 1 mL of heptane was added with stirring at room temperature. After several minutes, 1 mL of heptane was further added to the solution and stirring was continued. The solution was stirred at room temperature for one hour and then further stirred at about 50C for 20 minutes. The precipitated solid was collected by filtration through a glass filter and dried under reduced pressure at room temperature to obtain 49.24 mg of the title compound as white solid crystals. 1H-NMR (400 MHz, DMSO-d6) δ (ppm): 1.99 (m, IH), 2.17 (m, 2H), 2.30 (m, IH), 2.34 (brd, J = 1 Hz5 3H), 4.01 (s, 3H), 4.30 (m, 2H), 4.52 (dd, J = 9, 6 Hz5 IH), 7.25 (brd, J = 8 Hz5 IH), 7.37 (d, J = 16 Hz, IH), 7.40 (d, J = 7 Hz, IH)57.50 (brdd, J = 8 Hz, IH), 7.55 (d, J = 16 Hz, IH), 7.61 (brdd, J = 8 Hz5 IH), 7.77 (m, IH)5 7.78 (m, IH), 8.00 (d, J = 7 Hz, IH), 9.37 (d, J = 2 Hz, IH). [0035] Example 5
Synthesis of r8SV2-((Ε)-2-r6-methoxy-5-r4-methyl-lH-imidazol-l-yl)ρyridin-2-vnvinyl}-8-r2-trifluoromethylphenyl)-5,6J,8-tetrahvdro-[1.2,41triazolo[1.5-alpyridine hydrochloride
Concentrated hydrochloric acid (3.6 μL) was added to a solution of (8S)-2-{(E)- 2-[6-methoxy-5-(4-metiiyl-lH-imidazol-l-yl)pyridin-2-yl]vinyl}-8-(2-trifluoromethylplienyl)-5,6,7,8-te1xahydro-[l,2,4]triazolo[l,5-a]pyridme (19.80 mg) in 2-propanol (1 mL), and a total of 4 mL of heptane was added in 1 mL portions with stirring at room temperature. The solution was stirred at room temperature under shading for five days. The precipitated solid was collected by filtration through a glass filter and dried under reduced pressure at room temperature to obtain 7.45 mg of the title compound as white solid crystals. 1H-NMR (400 MHz, DMSOd6) δ (ppm): 1.97 (m, IH), 2.17 (m, 2H)5 2.30 (m, IH), 2.30 (s, 3H), 4.00 (s, 3H), 4.30 (m, 2H)5 4.52 (dd, J = 9, 6 Hz5 IH), 7.25 (brd, J – 8 Hz5 IH), 7.36 (d, J = 16 Hz5 IH), 7.37 (d5 J = 8 Hz, IH), 7.50 (brt, J = 8 Hz5 IH)5 7.53 (d, J = 16 Hz5 IH)5 7.61 (brt, J = 8 Hz5 IH)5 7.66 (brs, IH), 7.77 (brd, J = 8 Hz, IH)5 7.96 (d, J = 8 Hz5 IH), 9.06 (brs, IH). [0036] Example 6
Synthesis of (8S)-2-((ΕV2-r6-methoxy-5-(‘4-methyl-lH-imidazol-l-yl)pyridin-2-yl1vinvU-8-(2-trifluoromethylphenyl)-5.6,7,8-tetrahvdro-[l,2,4]triazolo[L5-a1pyridine hydrochloride Concentrated hydrochloric acid (14.3 μL) and heptane (7 mL) were added to a solution of (8S)-2-{(E)-2-[6-methoxy-5-(4-methyl-lH-imidazol-l-yl)pyridin-2-yl]vinyl}-8-(2-trifluoromethylphenyl)-5,657,8-tetrahydro-[l,2,4]triazolo[l,5-a]pyridine (79.77 mg) in 2-propanol (3 mL). A small amount of the crystals obtained in Example 5 were added as seed crystals with stirring at room temperature. The mixture was transferred to a low temperature laboratory at about 50C and stirred for one hour. Thereafter, 1 mL of heptane was further added, followed by stirring for several minutes. When the precipitated solid was collected by filtration through a glass filter, the solid was precipitated in the filtrate. The precipitated solid was collected by filtration through a glass filter and dried under reduced pressure at room temperature to obtain 38.02 mg of the title compound as white solid crystals. 1H-NMR (400 MHz, DMSO-d6) δ (ppm): 1.97 (m, IH), 2.17 (m5 2H), 2.29 (m, IH), 2.32 (brd, J = 1 Hz, 3H), 4.00 (s, 3H), 4.30 (m, 2H), 4.52 (dd, J = 9, 6 Hz, IH), 7.25 (brd, J = 8 Hz, IH), 7.37 (d, J = 16 Hz5 IH), 7.38 (d, J = 8 Hz, IH), 7.50 (brdd, J = 8 Hz, IH)5 7.54 (d, J = 16 Hz, IH), 7.61 (brdd, J = 8 Hz, IH), 7.72 (brs, IH), 7.77 (brd, J = 8 Hz, IH), 7.98 (d, J = 8 Hz5 IH)5 9.24 (brs, IH). [0037] Example 7
SvnJhesis off8SV2-f(E>2-r6-memoxy-5-(4-mefovπ trifluoromethylt>henylV5,6,7,8-tetrahvdro-[l,2,4]triazolo[l,5-a]pyridine mesylate
Mesylic acid (0.8 μL) was added to a mixed solution of (8S)-2-{(E)-2-[6- methoxy-5-(4-methyl-lH-imidazol-l-yl)pyridin-2-yl]vinyl}-8-(2-trifluoromethylphen^ tetrahydro-[l52,4]triazolo[l,5-a]pyridine (50 mg) in t-butyl methyl ether (0.8 mL)-ethaαol (0.1 mL). The mixture was solidified as a result of stirring at room temperature for two hours. The solid was collected by filtration through a glass filter. The solid was washed with t-butyl methyl ether-ethanol (8:1) and then dried under reduced pressure at room temperature to obtain 51.9 mg of the title compound as pale yellow solid crystals.
1H-NMR (DMSO-d6) δ (ppm): 1.90-2.05 (m, IH)3 2.10-2.22 (m, 2H), 2.28-2.40 (m, IH), 2.31 (s, 3H), 2.35 (s, 3H)5 4.02 (s, 3H)5 4.25-4.39 (m, 2H), 4.50-4.55 (m, IH), 7.27 (d5 J = 8.0 Hz5 IH)5 7.38 (d, J = 16.0 Hz5 IH)5 7.41 (d, J = 8.0 Hz, IH)5 7.51 (t5 J = 8.0 Hz5 IH)5 7.55 (d, J = 16.0 Hz5 IH), 7.63 (t, J = 8.0 Hz5 IH)5 7.78 (d, J = 8.0 Hz5 IH)5 7.79 (s, IH), 8.01 (d, J = 8.0 Hz5 IH), 9.37 (s, IH). [0038] Example 8 Synthesis of (8S)-2-((ΕV2-r6-methoxy-5-(4-methyl-lH-imidazol-l-vnpyridin-2-vnvinvn-8-r2-trifluoromethylphenyl)-5.6,7,8-tetrahydro-[l.,2,4‘|triazolo[l,5-a]pyridine diphosphate
A solution of phosphoric acid (52.8 mg) in acetonitrile (0.2 mL) was added to a solution of (8S)-2-{(E)-2-[6-methoxy-5-(4-methyl-lH-mτidazol-l-yl)ρyridin-2-yl]vinyl}-8-(2-trifluoromethylphenyl)-5565758-tetrahydro-[l5254]triazolo[l,5-a]pyridine (100 mg) in acetonitrile (0.8 mL) at room temperature. The precipitated oil was solidified as a result of stirring with spatula. The solid was collected by filtration through a glass filter. The solid was washed with ice-cold acetonitrile, air-dried at room temperature for 10 minutes and then dried under reduced pressure at room temperature to obtain 120 mg of the title compound as white solid crystals. 1H-NMR (DMSO-d6) δ (ppm): 1.90-2.05 (m, IH), 2.11-2.20 (m, 2H), 2.15 (s, 3H), 2.25-2.35 (m, IH), 3.99 (s, 3H)5 4.24-4.39 (m, 2H), 4.50-4.55 (m, IH)5 7.23 (s, IH), 7.26 (d, J = 7.0 Hz, IH), 7.28 (d, J = 8.0 Hz, IH), 7.33 (d, J = 16.0 Hz5 IH), 7.47 (d, J = 16.0 Hz5 IH), 7.51 (t, J = 7.0 Hz, IH), 7.63 (t, J = 7.0 Hz, IH), 7.78 (d, J = 7.0 Hz, IH), 7.79 (d, J = 8.0 Hz, IH), 7.90 (s, IH). [0039] Example 9 Svnmesis of(8SV2-{(E)-2-[6-memoxy-5-(4-methyl-lH-irnidazol-l-yl)pyridin-2-yl1vinvU-8-(2-trifluoromethylphenyl)-5,6,7,8-tetrahvdro-[l .2.41triazolo[l .5-a]pyridine diphosphate
A solution of phosphoric acid (13.2 mg) in ethanol (0.05 mL) was added to a mixed solution of (8S)-2-{(E)-2-[6-methoxy-5-(4-methyl-lH-imidazol-l-yl)pyridin-2-yl]vinyl}-8-(2-trifluoromethylphenyl)-5,6,7,8-tetrahydro-[l,2!,4]triazolo[l,5-a]pyridme (50 mg) in heptane (0.6 mL)-ethanol (0.15 mL) at room temperature. The reaction solution was stirred at room temperature, and the precipitated solid was collected by filtration through a glass filter. The solid was washed with heptane-ethanol (3:1) and then dried under reduced pressure at room temperature to obtain 37.6 mg of the title compound as white solid crystals. 1H-NMR (DMSOd6) δ (ppm): 1.90-2.05 (m, IH), 2.11-2.20 (m, 2H), 2.15 (s, 3H), 2.25-2.35 (m, IH), 3.99 (s, 3H), 4.24-4.39 (m, 2H), 4.50-4.55 (m, IH), 7.23 (s, IH), 7.26 (d, J = 7.0 Hz, IH), 7.28 (d, J = 8.0 Hz, IH), 7.33 (d, J = 16.0 Hz, IH), 7.47 (d, J = 16.0 Hz, IH), 7.51 (t, J = 7.0 Hz, IH), 7.63 (t, J = 7.0 Hz, IH), 7.78 (d, J = 7.0 Hz, IH), 7.79 (d, J = 8.0 Hz, IH), 7.90 (s, IH).
CLIP
Development of an Efficient Manufacturing Process for E2212 toward Rapid Clinical Introduction
Process studies of E2212 (1) toward rapid clinical introduction are described. Through comprehensive route-finding studies and optimization of key condensation and cyclization steps, a racemate-based manufacturing route was established and successfully scaled-up to the hundred kilogram scale. For the rapid delivery of a drug substance containing the Z isomer for preclinical safety studies, the successful scale-up of the photoisomerization of an olefin in a flow system is also presented.
https://pubs.acs.org/doi/10.1021/acs.oprd.8b00444
E2212 (1) (18.0 kg, 92.5% yield) as a white solid. Mother liquor 3 were recycled according to the procedure described below. FTIR (cm–1, KBr) 3461, 3173, 2956, 1734, 1584, 1536, 1476, 1309, 1130, 835, 765, 752; 1H NMR (600 MHz, DMSO-d6) δ 7.91 (s, 1H), 7.78 (d, J = 8.4 Hz, 1H), 7.77 (br d, J = 8.4 Hz, 1H), 7.61 (br dd, J = 7.8, 7.8 Hz, 1H), 7.49 (br dd, J = 7.8, 7.8 Hz, 1H), 7.46 (d, J= 15.6 Hz, 1H), 7.32 (d, J = 15.6 Hz, 1H), 7.27 (d, J = 7.8 Hz, 1H), 7.25 (br d, J = 7.8 Hz, 1H), 7.22 (s, 1H), 4.51 (dd, J = 9.0, 6.0 Hz, 1H), 4.29 (s, 3H), 4.28 (m, 2H), 3.98 (s, 3H), 2.29 (m, 1H), 2.14 (s, 3H), 2.16 (m, 2H), 1.96 (m, 1H); 13C NMR (150 MHz, DMSO-d6) δ 173.3, 159.3, 155.4, 155.0, 150.1, 141.1, 137.1, 136.9, 133.6, 132.9, 131.0, 130.5, 127.6, 127.1 (q, JC–F = 30 Hz), 125.8 (q, JC–F = 5.6 Hz), 124.7 (q, JC–F = 270 Hz), 122.2, 120.7, 117.2, 116.5, 72.3, 53.7, 47.0, 37.6, 30.7, 21.3, 13.6; HRMS (ESI+) calcd for C25H23F3N6O ([M + H]+) 481.1958, found 481.1953.




///////////E2212, E 2212
Certolizumab pegol, セルトリズマブペゴル (遺伝子組換え)

>Amino acid sequence of the light chain DIQMTQSPSSLSASVGDRVTITCKASQNVGTNVAWYQQKPGKAPKALIYSASFLYSGVPY RFSGSGSGTDFTLTISSLQPEDFATYYCQQYNIYPLTFGQGTKVEIKRTVAAPSVFIFPP SDEQLKSGTASVVCLLNNFYPREAKVQWKVDNALQSGNSQESVTEQDSKDSTYSLSSTLT LSKADYEKHKVYACEVTHQGLSSPVTKSFNRGEC
>Amino acid sequence of the heavy chain EVQLVESGGGLVQPGGSLRLSCAASGYVFTDYGMNWVRQAPGKGLEWMGWINTYIGEPIY ADSVKGRFTFSLDTSKSTAYLQMNSLRAEDTAVYYCARGYRSYAMDYWGQGTLVTVSSAS TKGPSVFPLAPSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGL YSLSSVVTVPSSSLGTQTYICNVNHKPSNTKVDKKVEPKSCDKTHTCAA
Certolizumab pegol
CAS: 428863-50-7
セルトリズマブペゴル (遺伝子組換え)
CDP 870 / CDP-870 / CDP870 / PHA-738144
| Formula |
C2115H3252N556O673S16
|
|---|---|
| Cas |
428863-50-7
|
| Mol weight |
47748.8128
|
Reducing signs and symptoms of Crohn’s disease and treatment of moderately to severely active rheumatoid arthritis (RA).
Certolizumab pegol is a recombinant Fab’ antibody fragment against tumor necrosis factor alpha which is conjugated to an approximately 40kDa polyethylene glycol (PEG2MAL40K). Polyethylene glycol helps to delay the metabolism and elimination of the drugs. Chemically, the light chain is made up of 214 amino acid residues while the heavy chain is composed of 229 amino acid residues. The molecular mass of the Fab’ antibody fragment itself is 47.8 kDa. It is used for the treatment of rheumatoid arthritis and Crohn’s disease. FDA approved on April 22, 2008
Certolizumab pegol (CDP870, tradename Cimzia) is a biologic medication for the treatment of Crohn’s disease,[1][2] rheumatoid arthritis, psoriatic arthritis and ankylosing spondylitis. It is a fragment of a monoclonal antibody specific to tumor necrosis factor alpha(TNF-α) and is manufactured by UCB.[3][4][5]
Medical uses
- Crohn’s Disease
- On April 22, 2008, the U.S. FDA approved Cimzia for the treatment of Crohn’s disease in people who did not respond sufficiently or adequately to standard therapy.[4][6][7]
- Rheumatoid arthritis
- On June 26, 2009, the Committee for Medicinal Products for Human Use (CHMP) of the European Medicines Agency (EMA) issued a positive opinion recommending that the European Commission grant a marketing authorisation for Cimzia for the treatment of rheumatoid arthritis only – the CHMP refused approval for the treatment of Crohn’s disease. The marketing authorisation was granted to UCB Pharma SA on October 1, 2009.[8]
- Psoriatic arthritis
- On September 27, 2013, the U.S. FDA approved Cimzia for the treatment of adult patients with active psoriatic arthritis.[9]
Method of action
Certolizumab pegol is a monoclonal antibody directed against tumor necrosis factor alpha. More precisely, it is a PEGylated Fabfragment of a humanized TNF inhibitor monoclonal antibody.[10]
Clinical trials
- Crohn’s disease
- Positive results have been demonstrated in two phase III trials (PRECiSE 1 and 2) of certolizumab pegol versus placebo in moderate to severe active Crohn’s disease.[1][10][11][12]
- Axial spondyloarthritis
- In 2013, a phase 3 double blind randomized placebo-controlled study found significantly positive results in patient self-reported questionnaires, with rapid improvement of function and pain reduction, in patients with axial spondyloarthritis.[13]
- Rheumatoid arthritis
- Certolizumab appears beneficial in those with rheumatoid arthritis.[14]
Side effects
Significant side effects occur in 2% of people who take the medication.[14]
References
- ^ Jump up to:a b Sandborn WJ, Feagan BG, Stoinov S, et al. (July 2007). “Certolizumab pegol for the treatment of Crohn’s disease”. N. Engl. J. Med. 357 (3): 228–38. doi:10.1056/NEJMoa067594. PMC 3187683. PMID 17634458.
- ^ Goel, Niti; Sue Stephens (2010). “Certolizumab pegol”. mAbs. 2 (2): 137–147. doi:10.4161/mabs.2.2.11271. PMC 2840232. PMID 20190560.
- ^ Kaushik VV, Moots RJ (April 2005). “CDP-870 (certolizumab) in rheumatoid arthritis”. Expert Opinion on Biological Therapy. 5 (4): 601–6. doi:10.1517/14712598.5.4.601. PMID 15934837.
- ^ Jump up to:a b index.cfm?fuseaction=Search.Label_ApprovalHistory “Cimzia Label and Approval History” Check
|url=value (help). Drugs@FDA. U.S. Food and Drug Administration(FDA). Retrieved 2009-11-15. - ^ “Cimzia Prescribing Information” (PDF). US Food and Drug Administration (FDA). April 2016. Retrieved 2016-08-21.
- ^ UCB press release – Cimzia Approved in the US for the Treatment of Moderate to Severe Crohn’s Disease. Retrieved April 22, 2008.
- ^ Waknine, Yael (May 1, 2008). “FDA Approvals: Patanase, Actonel, Cimzia”. Medscape. Retrieved 2008-05-01.
- ^ “Cimzia European Public Assessment Report”. European Medicines Agency. Retrieved November 15, 2009.
- ^ “Cimzia (certolizumab pegol) approved by the U.S. FDA for treatment of adult patients with active psoriatic arthritis”. Archived from the original on October 1, 2013. Retrieved October 1, 2013.
- ^ Jump up to:a b Schreiber S. et al., Certolizumab pegol, a humanised anti-TNF pegylated FAb’ fragment, is safe and effective in the maintenance of response and remission following induction in active Crohn’s disease: a phase 3 study (precise), Gut, 2005, 54, suppl7, A82
- ^ Sandborn et al., Certolizumab pegol administered subcutaneously is effective and well tolerated in patients with active Crohn’s disease: results from a 26-week, placebo-controlled Phase 3 study (PRECiSE 1), Gastroenterology, 2006, 130, A107
- ^ “New Analysis Shows Cimzia (Certolizumab Pegol) Maintained Remission and Response in Recent Onset Crohn’s Disease” (Press release). UCB. October 23, 2006. Retrieved 2009-11-15.
- ^ Sieper J, Tubergen A, Coteur G, Woltering F, Landewe R (May 2013). “PMS50 – Rapid Improvements In Patient-Reported Outcomes With Certolizumab Pegol In Patients With Axial Spondyloarthritis, Including Ankylosing Spondylitis And Non-Radiographic Axial Spondyloarthritis: 24-Week Results Of A Phase 3 Double Blind Randomized Placebo-Controlled Study”. Value in Health. 16 (3): A227. doi:10.1016/j.jval.2013.03.1150.
- ^ Jump up to:a b Ruiz Garcia, V; Jobanputra, P; Burls, A; Vela Casasempere, P; Bort-Marti, S; Bernal, JA (Sep 8, 2017). “Certolizumab pegol (CDP870) for rheumatoid arthritis in adults”(PDF). The Cochrane Database of Systematic Reviews. 9: CD007649. doi:10.1002/14651858.CD007649.pub4. PMID 28884785.
External links
- certolizumab+pegol at the US National Library of Medicine Medical Subject Headings (MeSH)
- Cimzia Website
FDA approves treatment Cimzia (certolizumab pegol) for patients with a type of inflammatory arthritis
March 28, 2019
Release
The U.S. Food and Drug Administration today approved Cimzia (certolizumab pegol) injection for treatment of adults with a certain type of inflammatory arthritis called non-radiographic axial spondyloarthritis (nr-axSpA), with objective signs of inflammation. This is the first time that the FDA has approved a treatment for nr-axSpA.
“Today’s approval of Cimzia fulfills an unmet need for patients suffering from non-radiographic axial spondyloarthritis as there has been no FDA-approved treatments until now,” said Nikolay Nikolov, M.D., associate director for rheumatology of the Division of Pulmonary, Allergy, and Rheumatology Products in the FDA’s Center for Drug Evaluation and Research.
Nr-axSpA is a type of inflammatory arthritis that causes inflammation in the spine and other symptoms. There is no visible damage seen on x-rays, so it is referred to as non-radiographic.
The efficacy of Cimzia for the treatment of nr-axSpA was studied in a randomized clinical trial in 317 adult patients with nr-axSpA with objective signs of inflammation, indicated by elevated C-reactive protein (CRP) levels and/or sacroiliitis (inflammation of the sacroiliac joints) on MRI. The trial measured the improvement response on the Ankylosing Spondylitis Disease Activity Score, a composite scoring system that assesses disease activity including patient-reported outcomes and CRP levels. Responses were greater for patients treated with Cimzia compared to patients treated with placebo. The overall safety profile observed in the Cimzia treatment group was consistent with the known safety profile of Cimzia.
The prescribing information for Cimzia includes a Boxed Warning to advise health care professionals and patients about the increased risk of serious infections leading to hospitalization or death including tuberculosis (TB), bacterial sepsis (infection in the blood steam), invasive fungal infections (such as histoplasmosis, an infection that affects the lungs), and other infections. Cimzia should be discontinued if a patient develops a serious infection or sepsis. Health care providers are advised to perform testing for latent TB and, if positive, to start treatment for TB prior to starting Cimzia. All patients should be monitored for active TB during treatment, even if the initial latent TB test is negative. The Boxed Warning also advises that lymphoma (cancer in blood cells) and other malignancies, some fatal, have been reported in children and adolescent patients treated with tumor necrosis factor (TNF) blockers, of which Cimzia is a member. Cimzia is not indicated for use in pediatric patients. Cimzia must be dispensed with a patient Medication Guide that describes important information about the drug’s uses and risks.
Cimzia was originally approved in 2008 and is also indicated for adult patients with Crohn’s disease, moderate-to-severe rheumatoid arthritis, active ankylosing spondylitis (AS) and moderate-to-severe plaque psoriasis who are candidates for systemic therapy or phototherapy.
The FDA granted the approval of Cimzia to UCB.

Syringe with 200mg Certolizumab pegol
|
|
| Monoclonal antibody | |
|---|---|
| Type | Fab’ fragment |
| Source | Humanized (from mouse) |
| Target | TNF alpha |
| Clinical data | |
| Trade names | Cimzia |
| AHFS/Drugs.com | Consumer Drug Information |
| MedlinePlus | a608041 |
| License data | |
| Pregnancy category |
|
| Routes of administration |
Subcutaneous |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Elimination half-life | about 11 days |
| Excretion | Renal (PEG only) |
| Identifiers | |
| CAS Number | |
| ChemSpider |
|
| UNII | |
| KEGG | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C2115H3252N556O673S16 |
| Molar mass | 47,750 g/mol g·mol−1 |
///////////////FDA 2019, Cimzia, certolizumab pegol, inflammatory arthritis, UCB
FDA approves treatment Cimzia (certolizumab pegol) for patients with a type of inflammatory arthritis
FDA approves treatment Cimzia (certolizumab pegol) for patients with a type of inflammatory arthritis
March 28, 2019
Release
The U.S. Food and Drug Administration today approved Cimzia (certolizumab pegol) injection for treatment of adults with a certain type of inflammatory arthritis called non-radiographic axial spondyloarthritis (nr-axSpA), with objective signs of inflammation. This is the first time that the FDA has approved a treatment for nr-axSpA.
“Today’s approval of Cimzia fulfills an unmet need for patients suffering from non-radiographic axial spondyloarthritis as there has been no FDA-approved treatments until now,” said Nikolay Nikolov, M.D., associate director for rheumatology of the Division of Pulmonary, Allergy, and Rheumatology Products in the FDA’s Center for Drug Evaluation and Research.
Nr-axSpA is a type of inflammatory arthritis that causes inflammation in the spine and other symptoms. There is no visible damage seen on x-rays, so it is referred to as non-radiographic.
The efficacy of Cimzia for the treatment of nr-axSpA was studied in a randomized clinical trial in 317 adult patients with nr-axSpA with objective signs of inflammation, indicated by elevated C-reactive protein (CRP) levels and/or sacroiliitis (inflammation of the sacroiliac joints) on MRI. The trial measured the improvement response on the Ankylosing Spondylitis Disease Activity Score, a composite scoring system that assesses disease activity including patient-reported outcomes and CRP levels. Responses were greater for patients treated with Cimzia compared to patients treated with placebo. The overall safety profile observed in the Cimzia treatment group was consistent with the known safety profile of Cimzia.
The prescribing information for Cimzia includes a Boxed Warning to advise health care professionals and patients about the increased risk of serious infections leading to hospitalization or death including tuberculosis (TB), bacterial sepsis (infection in the blood steam), invasive fungal infections (such as histoplasmosis, an infection that affects the lungs), and other infections. Cimzia should be discontinued if a patient develops a serious infection or sepsis. Health care providers are advised to perform testing for latent TB and, if positive, to start treatment for TB prior to starting Cimzia. All patients should be monitored for active TB during treatment, even if the initial latent TB test is negative. The Boxed Warning also advises that lymphoma (cancer in blood cells) and other malignancies, some fatal, have been reported in children and adolescent patients treated with tumor necrosis factor (TNF) blockers, of which Cimzia is a member. Cimzia is not indicated for use in pediatric patients. Cimzia must be dispensed with a patient Medication Guide that describes important information about the drug’s uses and risks.
Cimzia was originally approved in 2008 and is also indicated for adult patients with Crohn’s disease, moderate-to-severe rheumatoid arthritis, active ankylosing spondylitis (AS) and moderate-to-severe plaque psoriasis who are candidates for systemic therapy or phototherapy.
The FDA granted the approval of Cimzia to UCB.
///////////////FDA 2019, Cimzia, certolizumab pegol, inflammatory arthritis, UCB
FDA approves new oral treatment for multiple sclerosis, Mavenclad (cladribine)
March 29, 2019
Release
The U.S. Food and Drug Administration today approved Mavenclad (cladribine) tablets to treat relapsing forms of multiple sclerosis (MS) in adults, to include relapsing-remitting disease and active secondary progressive disease. Mavenclad is not recommended for MS patients with clinically isolated syndrome. Because of its safety profile, the use of Mavenclad is generally recommended for patients who have had an inadequate response to, or are unable to tolerate, an alternate drug indicated for the treatment of MS.
“We are committed to supporting the development of safe and effective treatments for patients with multiple sclerosis,” said Billy Dunn, M.D., director of the Division of Neurology Products in the FDA’s Center for Drug Evaluation and Research. “The approval of Mavenclad represents an additional option for patients who have tried another treatment without success.”
MS is a chronic, inflammatory, autoimmune disease of the central nervous system that disrupts communications between the brain and other parts of the body. Most people experience their first symptoms of MS between the ages of 20 and 40. MS is among the most common causes of neurological disability in young adults and occurs more frequently in women than in men.
For most people, MS starts with a relapsing-remitting course, in which episodes of worsening function (relapses) are followed by recovery periods (remissions). These remissions may not be complete and may leave patients with some degree of residual disability. Many, but not all, patients with MS experience some degree of persistent disability that gradually worsens over time. In some patients, disability may progress independent of relapses, a process termed secondary progressive multiple sclerosis (SPMS). In the first few years of this process, many patients continue to experience relapses, a phase of the disease described as active SPMS. Active SPMS is one of the relapsing forms of MS, and drugs approved for the treatment of relapsing forms of MS can be used to treat active SPMS.
The efficacy of Mavenclad was shown in a clinical trial in 1,326 patients with relapsing forms of MS who had least one relapse in the previous 12 months. Mavenclad significantly decreased the number of relapses experienced by these patients compared to placebo. Mavenclad also reduced the progression of disability compared to placebo.
Mavenclad must be dispensed with a patient Medication Guide that describes important information about the drug’s uses and risks. Mavenclad has a Boxed Warning for an increased risk of malignancy and fetal harm. Mavenclad is not to be used in patients with current malignancy. In patients with prior malignancy or with increased risk of malignancy, health care professionals should evaluate the benefits and risks of the use of Mavenclad on an individual patient basis. Health care professionals should follow standard cancer screening guidelines in patients treated with Mavenclad. The drug should not be used in pregnant women and in women and men of reproductive potential who do not plan to use effective contraception during treatment and for six months after the course of therapy because of the potential for fetal harm. Mavenclad should be stopped if the patient becomes pregnant.
Other warnings include the risk of decreased lymphocyte (white blood cell) counts; lymphocyte counts should be monitored before, during and after treatment. Mavenclad may increase the risk of infections; health care professionals should screen patients for infections and treatment with Mavenclad should be delayed if necessary. Mavenclad may cause hematologic toxicity and bone marrow suppression so health care professionals should measure a patient’s complete blood counts before, during and after therapy. The drug has been associated with graft-versus-host-disease following blood transfusions with non-irradiated blood. Mavenclad may cause liver injury and treatment should be interrupted or discontinued, as appropriate, if clinically significant liver injury is suspected.
The most common adverse reactions reported by patients receiving Mavenclad in the clinical trials include upper respiratory tract infections, headache and decreased lymphocyte counts.
The FDA granted approval of Mavenclad to EMD Serono, Inc.
Cevimeline, セビメリン
Cevimeline
セビメリン
- Molecular FormulaC10H17NOS
- Average mass199.313 Da
Cevimeline hydrochloride
-
- Synonyms:AF-102B, SNI-2011, SNK-508, Evoxac
- ATC:N07
- Use:cognition disorder, treatment of Sjogren’s syndrome, muscarinic M3-receptor agonist
- Chemical name:(2′R,3R)-rel-2′-methylspiro[1-azabicyclo[2.2.2]octane-3,5′-[1,3]oxathiolane] hydrochloride hydrate (2:2:1)
- Formula:C10H17NOS • HCl • 1/2H2O
- MW:489.57 g/mol
- CAS-RN:153504-70-2
- InChI Key:SURWTGAXEIEOGY-GHXDPTCOSA-N
- InChI:InChI=1S/C10H17NOS.ClH/c1-8-12-10(7-13-8)6-11-4-2-9(10)3-5-11;/h8-9H,2-7H2,1H3;1H/t8-,10-;/m1./s1
Derivatives
base
- Formula:C10H17NOS
- MW:199.32 g/mol
- CAS-RN:107233-08-9
anhydrous hydrochloride
- Formula:C10H17NOS • HCl
- MW:235.78 g/mol
- CAS-RN:107220-28-0
Cevimeline is cis-2′-methylspiro {1-azabicyclo [2.2.2] octane-3, 5′ -[1,3] oxathiolane} hydro-chloride, hydrate (2:1). Its empirical formula is C10H17NOS•HCl•½ H2O, and its structural formula is:
 Cevimeline has a molecular weight of 244.79. It is a white to off white crystalline powder with a melting point range of 201 to 203°C. It is freely soluble in alcohol and chloroform, very soluble in water, and virtually insoluble in ether. The pH of a 1% solution ranges from 4.6 to 5.6. Inactive ingredients include lactose monohydrate, hydroxypropyl cellulose, and magnesium stearate.
Cevimeline has a molecular weight of 244.79. It is a white to off white crystalline powder with a melting point range of 201 to 203°C. It is freely soluble in alcohol and chloroform, very soluble in water, and virtually insoluble in ether. The pH of a 1% solution ranges from 4.6 to 5.6. Inactive ingredients include lactose monohydrate, hydroxypropyl cellulose, and magnesium stearate.
Cevimeline hydrochloride [USAN]
RN: 153504-70-2
(+-)-cis-2-Methylspiro(1,3-oxathiolane-5,3′-quinuclidine) hydrochloride, hemihydrate
Cevimeline (trade name Evoxac) is a parasympathomimetic and muscarinic agonist,[1] with particular effect on M1 and M3 receptors. It is used in the treatment of dry mouth and especially associated with Sjögren’s syndrome.
Mechanism of action
By activating the M3 receptors of the parasympathetic nervous system, cevimeline stimulates secretion by the salivary glands, thereby alleviating dry mouth.
Side effects
Known side effects include nausea, vomiting, diarrhea, excessive sweating, rash, headache, runny nose, cough, drowsiness, hot flashes, blurred vision, and difficulty sleeping.[2]
Contraindications include asthma and angle closure glaucoma.
Clip
https://www.sciencedirect.com/science/article/abs/pii/S0731708515302260



CLIP
https://www.sciencedirect.com/science/article/pii/S0040403913005042



CLIP
CLIP

- Reaction of quinuclidin-3-one (I) with trimethylsulfoxonium iodide and NaH in DMSO gives epoxide (II), which is opened with SH2 in NaOH/water, yielding 3-hydroxy-3-(sulfanylmethyl)quinuclidine (III). The cyclization of compound (III) with acetaldehyde (IV) catalyzed by boron trifluoride ethearate or by SnCl4, POCl3, H3PO4 or p-toluenesulfonic acid affords a mixture of two diastereomeric spiroracemates, the (?-trans (V) and (?-cis (cevimeline). This mixture is separated by fractional recrystallization in acetone or by TLC chromatography, and treated with hydrochloric acid. The (?-trans-compound (V) can be isomerized to cevimeline by treatment with an acidic catalyst such as an organic sulfonic acid (trifluoromethanesulfonic acid, p-toluenesulfonic acid or methanesulfonic acid), a Lewis acid (SnCl4, FeCl3, BF3 or AlCl3) or sulfuric acid in refluxing toluene, hexane or CHCl3. Cevimeline hydrochloride hemihydrate is obtained from the above mentioned hydrochloride by a complex work-up using water, isopropanol and n-hexane.
- Synthesis of Cevimeline Hydrochloride (EN:134916): Reaction of quinuclidin-3-one (I) with trimethylsulfoxonium iodide and NaH in DMSO gives epoxide (II), which is opened with SH2 in NaOH/water, yielding 3-hydroxy-3-(sulfanylmethyl)quinuclidine (III) (1,2). The cyclization of compound (III) with acetaldehyde (IV) catalyzed by boron trifluoride ethearate (1) or by SnCl4, POCl3, H3PO4 or p-toluenesulfonic acid (2) affords a mixture of two diastereomeric spiro-racemates, the (?-trans (V) and (?-cis (cevimeline). This mixture is separated by fractional recrystallization in acetone or by TLC chromatography, and treated with hydrochloric acid (1,2). The (?-trans-compound (V) can be isomerized to cevimeline by treatment with an acidic catalyst such as an organic sulfonic acid (trifluoromethanesulfonic acid, p-toluenesulfonic acid or methanesulfonic acid), a Lewis acid (SnCl4, FeCl3, BF3 or AlCl3) or sulfuric acid in refluxing toluene, hexane or CHCl3 (2,3). Cevimeline hydrochloride hemihydrate is obtained from the above mentioned hydrochloride by a complex work-up using water, isopropanol and n-hexane (4).(Scheme 13491601a) Description M.p. 203 C (4). Sources Discovered by Israel Institute for Biological Research, Ness-Ziona (IL) and licensed to Snow Brand Milk Products Co. Ltd. (JP). In the U.S., comarketed by Snow Brand Milk Products and Daiichi Pharmaceutical Co., Ltd. In Japan, codeveloped with Nippon Kayaku Co. Ltd. Ishihara Sangyo Co., Ltd. (JP) is the bulk supplier. References 1. Fisher, A., Heldman, E., Grunfeld, Y., Karton, I., Levy, A. (Israel Institute for Biological Research); Derivs. of quinuclidine; EP 0205247, JP 1986280497, US 4855290. 2. Hayashi, K., Tokumoto, S., Yoshizawa, H., Isogai, T. (Ishihara Sangyo Kaisha, Ltd.); Method for producing 2-methylspiro(1,3-oxathiolan-5,3′)quinuclidine; EP 0683168, US 5571918. 3. Haga, T., Koyanagi, T., Hara, K., Maeda, M., Shigehara, I. (Ishihara Sangyo Kaisha, Ltd.); Method for isomerization of trans-form 2-methylspiro(1,3-oxathiolane-5,3′)quinuclidine or acid addition salts thereof; EP 0298491, US 4861886. 4. Saito, K., Ono, T., Honda, N. (Snow Brand Milk Products Co., Ltd.); Preparation method of cis-2-methylspiro(1,3-oxathiolane-5,3′)quinuclidine hydrochloride.1/2 hydrate capable of disgregating easily; JP 1992108792.
PATENT
https://patents.google.com/patent/US8080663B2/en
The present invention refers to a novel, industrially advantageous process for the preparation of an intermediate useful for the preparation of Cevimeline hydrochloride (1, cis-2-methylspiro(1,3-oxathiolane-5,3′)quiniclidine, Scheme 1). This pharmaceutical is useful for the treatment of diseases of the central nervous system due to disturbances of central cholinergic function and autoimmune system (Sjörgen’s syndrome) and is marketed as Evoxac®.
U.S. Pat. No. 4,855,290 describes a process for preparation of 2-methylspiro(1,3-oxathiolane-5,3′)quiniclidine (1). The process comprises the preparation of the epoxide of 3-methylenequiniclidine, which is subsequently reacted with hydrogen sulfide to produce 3-hydroxy-3-mercaptomethylquiniclidine and condensed with acetaldehyde in the presence of a Lewis acid (boron trifluoride etherate) to provide 2-methylspiro(1,3-oxathiolane-5,3′)quiniclidine. This process is depicted in Scheme I.
This process suffers from major disadvantages when transiting to industrial scale. These include the use of the highly hazardous and difficult to handle hydrogen sulfide gas. Also, boron trifluoride etherate is employed during the condensation step with acetaldehyde. The boron trifluoride etherate reagent is an air and moisture sensitive Lewis acid which has to be used under anhydrous conditions, thus creating a serious disadvantage in industrial settings. Another drawback of this process is the use of sodium hydride. U.S. Pat. Nos. 5,571,918 and 4,861,886 relate to the isomerization of the trans- to cis-form of 2-methylspiro(1,3-oxathiolane-5,3′)quiniclidine but do not describe methods for its preparation. Thus, an industrially acceptable and cost-effective method for the preparation of Cevimeline hydrochloride which overcomes the deficiencies of the prior art is required.
Further and other objects of the invention will be realized by those skilled in the art from the following Summary of the Invention and Detailed Description of Preferred Embodiments of the Invention thereof.
According to one aspect of the invention, a novel process is provided for the preparation of 2-methylspiro(1,3-oxathiolane-5,3′)quiniclidine (1). The process is industrially practical, efficient, safe and economical, as well as being environmentally friendly. The general method is shown in the Scheme II.
wherein R is selected from C1 to C6 alkyl and aryl groups, most preferably a methyl, ethyl or propyl group; R1 is hydrogen or a C2 to C7 alkyl or aryl carbonyl group; R2 is a C1 to C6 alkyl group, preferably methyl, ethyl, propyl, or butyl group.
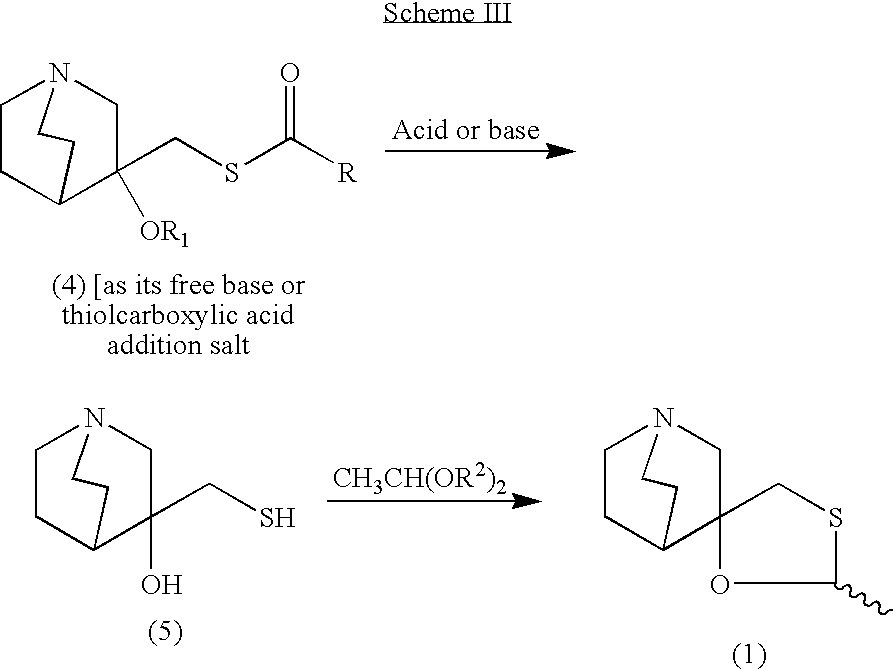
EXAMPLE I Preparation of the Epoxide of 3-methylenequiniclidine (3)
A mixture of the hydrochloric salt of 3-quiniclidinone (2, 120 g, 795.7 mmol) and trimethylsulfoxonium iodide (219 g, 993.3 mmol) in dimethylsulfoxide (91.0 g, 0.63 mol) was cooled to 0-5° C. in an ice/water bath under nitrogen atmosphere. A solution of potassium tert-butoxide (201 g, 1789.1 mmol) in dimethylsulfoxide (500 mL) was added dropwise over 45 minutes. The mixture was warmed gradually to room temperature and stirred for an additional 16 hours at room temperature. After cooling to 0-5° C. (ice/water bath) the mixture was poured into an ice/water mixture (500 g) and then sodium chloride (300 g) was added. The mixture was stirred for 30 minutes and extracted with toluene (3×400 mL). The toluene phase was dried over sodium sulfate, filtered and evaporated to furnish the epoxide of 3-methylenequiniclidine (60 g, 431.7 mmol, 54% yield) as a yellow oil. The product could be used in the next step neat or as toluene solution after the extraction without further purification.
1H NMR (400 MHz, CDCl3): δ=3.10 (d, 1H, J=14.6 Hz); 2.98-2.77 (m, 5H); 2.74 (d, 1H, J=4.8 Hz); 2.70 (d, 1H, J=4.8 Hz); 1.96-1.89 (m, 1H); 1.79-1.62 (m, 2H); 1.60-1.54 (m, 1H); 1.38-1.36 (m,1H).
LRMS (ES+): 140.0 (100, M+H+).
EXAMPLE II Preparation of the Thiolacetic Acid Salt of 3-hydroxy-3-acetoxymercaptomethylquiniclidine (4)
A solution of the epoxide of 3-methylenequiniclidine (3, 54 g, 388.5 mmol) in toluene (200 mL) was cooled to 0-5° C. (ice/water bath). Thiolacetic acid was added dropwise over 10-15 minutes. The mixture was stirred at 0-5° C. for 30 minutes and then allowed to come to room temperature. After stirring at room temperature for 2 hours the formed precipitate was filtered and washed with toluene (2×100 mL) to give the 3-hydroxy-3-acetoxymercaptomethylquiniclidine thiolacetic acid salt (4 wherein R1 is H and R is methyl, 77 g, 264.6 mmol, 68%) as a light yellow solid. The product was used in the next step without any further purification.
1H NMR (400 MHz CD3OD): δ=3.47 (d, 1H, J=14.1 Hz); 3.37-3.18 (m, 7H); 2.40 (s, 3H); 2.38 (s, 3H); 2.36-2.27 (m, 1H), 2.14-2.05 (m, 2H); 2.03-1.93 (m, 1H); 1.81-1.78 (m, 1H).
LRMS (ES+): 216.1 (100, M−[SCOCH3]−+H+).
EXAMPLE III Preparation of 2-methylspiro(1,3-oxathiolane-5,3′)quiniclidine using p-toluenesulfonic acid (1)
To a solution of 3-hydroxy-3-acetoxymercaptomethylquiniclidine thiolacetic acid salt (4 wherein R1 is H and R is methyl, 3 g, 10.3 mmol) in iso-propanol (50 mL) was added p-toluenesulfonic acid monohydrate (5.9 g, 30.9 mmol) and the mixture was heated to reflux for 3.5 hours. The mixture was cooled to room temperature and acetaldehyde diethyl acetal (6.1 g, 51.5 mmol) was added. The mixture was heated to reflux and stirred for an additional 3 hours. The solvent was evaporated and the residue was dissolved in dichloromethane (50 mL). The mixture was cooled to 0-5° C. and a 25% aqueous solution of sodium hydroxide (80 mL) was added. The mixture was stirred for 10-15 minutes and the phases were separated. The aqueous phase was extracted with dichloromethane (3×50 mL). The organic phases were combined and extracted with 5% aqueous solution of sulfuric acid (3×50 mL). The acidic aqueous phases were combined and the pH was adjusted to 12 with a 25% aqueous solution of sodium hydroxide. The aqueous phase was extracted with heptane (3×50 mL) and the organic phases were combined, dried over sodium sulfate and the solvent was evaporated to give 2-methylspiro(1,3-oxathiolane-5,3′)quiniclidine (1.8 g, 9.2 mmol, 89% yield) as a 3:1 cis/trans ratio mixture of diastereomers (determined by 1H NMR).
LRMS (ES+): 200.1 (100, M+H+).
EXAMPLE IV Preparation of 2-methylspiro(1,3-oxathiolane-5,3′)quiniclidine (1) using racemic camphorsulfonic acid
In a similar experiment as Example III, racemic camphorsulfonic acid (7.2 g, 30.9 mmol) was added to a solution of 3-hydroxy-3-acetoxymercaptomethylquiniclidine thiolacetic acid salt (4 wherein R1 is H and R is methyl, 3 g, 10.3 mmol) in iso-propanol (50 mL). The mixture was refluxed for 5 h, cooled to room temperature and acetaldehyde diethyl acetal (6.1 g, 51.5 mmol) was added. The mixture was refluxed for an additional an 8 hours and processed according to Example III to give 2-methylspiro(1,3-oxathiolane-5,3′)quiniclidine (1.32 g, 6.63 mmol, 64% yield) in a 3.5:1 cis/trans ratio mixture of diastereomers (determined by 1H NMR).
EXAMPLE V Preparation of 2-methylspiro(1,3-oxathiolane-5,3′)quiniclidine (1) using phenyl sulfonic acid
In a similar experiment as Example III, to a solution of 3-hydroxy-3-acetoxymercaptomethylquiniclidine thiolacetic acid salt (4 wherein R1 is H and R is methyl, 3 g, 10.3 mmol) in iso-propanol (50 mL) was added phenyl sulfonic acid (4.9 g, 30.9 mmol) and the mixture was refluxed 5 h, cooled to room temperature and acetaldehyde diethyl acetal (6.1 g, 51.5 mmol) was added. The mixture was refluxed for an additional 8 hours and worked up in a manner similar to Example III to furnish 2-methylspiro(1,3-oxathiolane-5,3′)quiniclidine (1.6 g, 8.2 mmol, 80% yield) as a 2.5:1 cis/trans ratio mixture of diastereomers (determined by 1H NMR).
EXAMPLE VI Preparation of 2-methylspiro(1,3-oxathiolane-5,3′)quiniclidine (1) using p-toluenesulfonic acid in butanol
To a solution of 3-hydroxy-3-acetoxymercaptomethylquiniclidine thiolacetic acid salt (4 wherein R1 is H and R is methyl, 3 g, 10.3 mmol) in butanol (100 mL) was added of p-toluenesulfonic acid monohydrate (5.9 g, 30.9 mmol) and the mixture was refluxed for 3 hours with a Dean-Stark apparatus attached to the flask. The reaction mixture was cooled to room temperature and acetaldehyde diethyl acetal (6.1 g, 51.5 mmol) was added. The mixture was heated to 80° C. for an additional 8 h and worked up according to Example III to afford 2-methylspiro(1,3-oxathiolane-5,3′)quiniclidine (1.8 g, 9.2 mmol, 89% yield) as a 3:1 cis/trans ratio mixture of diastereomers (determined by 1H NMR).
References
- ^ Ono M, Takamura E, Shinozaki K, et al. (July 2004). “Therapeutic effect of cevimeline on dry eye in patients with Sjögren’s syndrome: a randomized, double-blind clinical study”. Am. J. Ophthalmol. 138 (1): 6–17. doi:10.1016/j.ajo.2004.02.010. PMID 15234277.
- ^ [1] MedicineNet: Cevimeline. Accessed 10/12/2007
-
- US 4 855 290 (Israel Institute for Biological Research; 8.8.1989; IL-prior. 10.5.1985).
- US 4 876 260 (Israel Institute for Biological Research; 24.10.1989; USA-prior. 28.10.1987).
- EP 683 168 (Ishihara Sangyo Kaisha; appl. 19.5.1995; J-prior. 19.5.1994).
-
Method for isomerization of trans-isomer:
- US 4 861 886 (Ishihara Sangyo Kaisha; 29.8.1989; J-prior. 10.7.1987).
-
Method of separation:
- IL 81 652 (Israel Institute for Biological Research; 12.5.1991; appl. 23.2.1987).
- JP 01 290 680 (Ishihara Sangyo Kaisha; 22.11.1989; J-prior. 18.5.1988).
-
Synthesis of enantiomerically pure (S)-3-hydroxy-3-mercaptomethylquinuclidine (S)-II:
- Bos, M.; Canesso, R.: Heterocycles (HTCYAM) 38 (8), 1889 (1994).
-
Synthesis of 3-quinuclidone:
- Sternbach, L.H.; Kaiser, S.: J. Am. Chem. Soc. (JACSAT) 74, 2215 (1952).
-
External links
 |
|
 |
|
| Clinical data | |
|---|---|
| Trade names | Evoxac |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a608025 |
| Pregnancy category |
|
| Routes of administration |
By mouth (capsules) |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Protein binding | <20% |
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C10H17NOS |
| Molar mass | 199.31308 g/mol g·mol−1 |
| 3D model (JSmol) | |
/////////// Cevimeline, AF-102B, SNI-2011, SNK-508, Evoxac, セビメリン
FDA approves new oral testosterone capsule for treatment of men with certain forms of hypogonadism
FDA approves new oral testosterone capsule (testosterone undecanoate) for treatment of men with certain forms of hypogonadism
March 27, 2019
Release
The U.S. Food and Drug Administration today approved Jatenzo (testosterone undecanoate), an oral testosterone capsule to treat men with certain forms of hypogonadism. These men have low testosterone levels due to specific medical conditions, such as genetic disorders like Klinefelter syndrome or tumors that have damaged the pituitary gland. Jatenzo should not be used to treat men with “age-related hypogonadism,” in which testosterone levels decline due to aging, even if these men have symptoms that appear to be related to low testosterone. Jatenzo’s benefits do not outweigh its risks for that use.
“Jatenzo’s oral route of administration provides an important addition to current treatment options available for men with certain hypogonadal conditions who up until now have most commonly been treated with testosterone products that are applied to the skin or injected,” said Hylton V. Joffe, M.D, M.M.Sc., director of the Division of Bone, Reproductive and Urologic Products in the FDA’s Center for Drug Evaluation and Research. “But it’s important to emphasize that this drug should not, like other testosterone treatments, be used to treat older men with ‘age-related hypogonadism.’ The benefits of testosterone therapy, including Jatenzo, have not been established for this use, and Jatenzo’s effects on raising blood pressure can increase the risks of heart attack, stroke and cardiovascular death in this population.”
The efficacy of Jatenzo was demonstrated in a four-month clinical trial involving 166 men with hypogonadism. Study participants initially were given Jatenzo at a dose of 237 mg twice per day, and the dose was adjusted downward or upward to a maximum of 396 mg twice per day on the basis of testosterone levels. Eighty-seven percent of Jatenzo-treated men achieved an average testosterone level within the normal range, which was the primary study endpoint.
Jatenzo contains a boxed warning on its labeling stating that the drug can cause blood pressure to rise, increasing the risk of heart attack, stroke and cardiovascular death. Health care providers should consider a patient’s individual heart disease risks and ensure that blood pressure is adequately controlled before prescribing Jatenzo; they should also periodically monitor patient blood pressure during treatment. Jatenzo is currently one of two testosterone products that have this boxed warning. The FDA is requiring all testosterone product manufacturers to conduct blood pressure postmarketing trials to more clearly address whether these products increase blood pressure.
Common side effects, occurring in more than 2 percent of patients in the Jatenzo clinical trial, included headache, an increase in hematocrit (red blood cell count), a decrease in high-density lipoprotein cholesterol (“good” cholesterol), high blood pressure and nausea. An increase in prostate specific antigen (PSA) was also observed. Patients should have their hematocrit, cholesterol and PSA monitored regularly to check for changes. Those with benign prostate hyperplasia should be monitored for worsening of symptoms.
The FDA granted the approval of Jatenzo to Clarus Therapeutics.
//////////FDA 2019, Jatenzo, Clarus Therapeutics, (testosterone undecanoate,
Batimastat, バチマスタット

| Formula |
C23H31N3O4S2
|
|---|---|
| cas |
130370-60-4
|
| Mol weight |
477.6399
|
Batimastat (INN/USAN, codenamed BB-94) is an anticancer drug that belongs to the family of drugs called angiogenesis inhibitors. It acts as a matrix metalloproteinase inhibitor (MMPI) by mimicking natural MMPI peptides.
Batimastat was the first MMPI that went into clinical trials. First results of a Phase I trial appeared in 1994. The drug reached Phase III but was never marketed; mainly because it couldn’t be administered orally (as opposed to the newer and chemically similar MMPI marimastat), and injection into the peritoneum caused peritonitis.[1]
SYN
U.S. Patent 5,453,438
U.S. Patent 5,240,958
U.S. Patent 5,530,161

SYN
US 5240958; US 5310763; WO 9005719
The treatment of D-leucine (I) with NaNO2, H2SO4 and NaBr gives 2(R)-bromo-5-methylpentanoic acid (II), which is esterified with isobutene and H2SO4 to the corresponding tert-butyl ester (III). The condensation of (III) with dibenzyl malonate (IV) by means of potassium tert-butoxide in DMF yields the malonyl derivative (V), which is treated with trifluoroacetic acid to hydrolyze the tert-butyl ester, and without isolation is condensed with L-phenylalanine methyl amide (VI) by means of hydroxybenzotriazole (HOBT) and dicyclohexylcarbodiimide (DCC), affording 4-benzyloxy-3-(benzyloxycarbonyl)-2(R)-isobutylsuccinyl-L-phenylalanine methylamide (VII). The elimination of the benzyl groups of (VII) by hydrogenolysis over Pd/C in ethanol gives the dicarboxylic acid (VIII), which by partial decarboxylation and reaction with aqueous formaldehyde and piperidine yields 4-hydroxy-2(R)-isobutyl-3-methylenesuccinyl-L-phenylalanine methylamide (IX). The addition of thiophene-2-thiol (X) to the double bond of (IX) affords 4-hydroxy-2(R)-isobutyl-3(S)-(2-thienylsulfanylmethyl)succinyl-L-phenylalanine methylamide (XI), which is finally treated with hydroxylamine and hydroxybenzotriazole in dichloromethane/DMF.
SPEC

HPLC

References
- ^ Rothenberg, M. L.; Nelson, A. R.; Hande, K. R. (1999). “New Drugs on the Horizon: Matrix Metalloproteinase Inhibitors”. Stem Cells. 17 (4): 237–240. doi:10.1002/stem.170237. PMID 10437989.
 |
|
| Clinical data | |
|---|---|
| Pregnancy category |
|
| Routes of administration |
Injection into pleural space or abdomen |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| ECHA InfoCard | 100.222.897 |
| Chemical and physical data | |
| Formula | C23H31N3O4S2 |
| Molar mass | 477.64 g/mol g·mol−1 |
| 3D model (JSmol) | |
//////////Batimastat, BB-94, バチマスタット ,
[H][C@@](CC1=CC=CC=C1)(NC(=O)[C@]([H])(CC(C)C)[C@]([H])(CSC1=CC=CS1)C(=O)NO)C(=O)NC
Abikoviromycin
- Molecular FormulaC10H11NO
- Average mass161.200 Da
Journal of Antibiotics (2003), 56, (9), 801-804.
“ALL FOR DRUGS” CATERS TO EDUCATION GLOBALLY, No commercial exploits are done or advertisements added by me. This is a compilation for educational purposes only. P.S. : The views expressed are my personal and in no-way suggest the views of the professional body or the company that I represent
READ
 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO …..FOR BLOG HOME CLICK HERE
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO …..FOR BLOG HOME CLICK HERE amcrasto@gmail.com
amcrasto@gmail.com
Industry-Oriented Route Evaluation and Process Optimization for the Preparation of Brexpiprazole
Efforts toward route evaluation and process optimization for the preparation of brexpiprazole (1) are described. Starting from commercially available dihydroquinolinone 11, a three-step synthesis route composed of O-alkylation, oxidation, and N-alkylation was selected for industry-oriented process development aiming to reduce side reactions and achieve better impurity profiles. The reaction conditions of the three steps were investigated, and the control strategy for the process-related impurities was established. The optimized process was validated on the kilogram scale and now is viable for commercialization, with the results of not less than 99.90% purity of 1 (by HPLC) and not more than 0.05% of persistent impurities 15 and 16
Industry-Oriented Route Evaluation and Process Optimization for the Preparation of Brexpiprazole