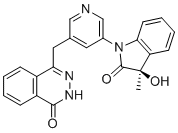
LINZAGOLIX
LINZAGOLIX
CAS 935283-04-8
C22H15F3N2O7S
- Hormone Antagonists
3-[5-[(2,3-difluoro-6-methoxyphenyl)methoxy]-2-fluoro-4-methoxyphenyl]-2,4-dioxo-1H-thieno[3,4-d]pyrimidine-5-carboxylic acid
- WHO 10711
- Treatment of Endometriosis Pain and Uterine Myoma-Associated Heavy Menstrual Bleeding
- OriginatorKissei Pharmaceutical
- DeveloperKissei Pharmaceutical; ObsEva
- Class2 ring heterocyclic compounds; Antihormones; Antineoplastics; Carboxylic acids; Fluorinated hydrocarbons; Ketones; Pyrimidines; Small molecules; Thiophenes
- Mechanism of ActionLHRH receptor antagonists
- PreregistrationUterine leiomyoma
- Phase IIIEndometriosis
- Phase IIAdenomyosis
- 22 Nov 2021FDA assigns PDUFA action date of (13/09/2022) for linzagolix for Uterine leiomyoma
- 22 Nov 2021The US FDA accepts NDA for linzagolix for Uterine leiomyoma for review
- 20 Oct 2021Efficacy and adverse events data from a phase II trial in Adenomyosis presented at the American Society for Reproductive Medicine (ASRM) 2021 Scientific Congress & Expo


Linzagolix choline
CAS#: 1321816-57-2 (choline)
Chemical Formula: C27H28F3N3O8S
Exact Mass: 611.1549
Molecular Weight: 611.58
Linzagolix is an orally bioavailable gonadotropin-releasing hormone (GnRH or LHRH) receptor antagonist, with potential hormone production inhibitory activity. Upon oral administration of linzagolix, this agent competes with GnRH for receptor binding and inhibits GnRH receptor signaling in the anterior pituitary gland, thereby inhibiting the secretion and release of luteinizing hormone (LH) and follicle stimulating hormone (FSH). In males, the inhibition of LH secretion prevents the release of testosterone. As a result, this may relieve symptoms associated with hormonally dependent disease states such as hormone-dependent prostate cancer. In women, this prevents the production of estrogen by the ovaries and may relieve symptoms from sex-hormone dependent diseases, such as pain associated with endometriosis, heavy menstrual bleeding or uterine fibroids.
Linzagolix (INN; developmental code names KLH-2109, OBE-2109; tentative brand name Yselty) is a small-molecule, non-peptide, orally active gonadotropin-releasing hormone antagonist (GnRH antagonist) which is under development by Kissei Pharmaceutical and ObsEva for the treatment of uterine fibroids, endometriosis, and adenomyosis.[1][3][2] As of December 2020, it is under review for approval for uterine fibroids, is in phase III clinical trials for endometriosis, and is in phase II clinical studies for adenomyosis.[1]
Estrogen-dependent disorders represent a challenging class of diseases that have a high incidence in the general population and are often associated with particularly severe symptomology. Uterine fibroids, for example, also referred to as leiomyomata, are among the most common benign tumors in women. Symptoms associated with uterine fibroids commonly include heavy or prolonged menstrual bleeding, pelvic pressure and pelvic organ compression, back pain, and adverse reproductive outcomes. Heavy menstrual bleeding may lead to iron deficiency anemia, a key symptom of uterine fibroids and the leading cause of surgical interventions that may include hysterectomy. Endometriosis is another estrogen-dependent gynecological condition, characterized by the presence of endometrial-like tissue outside the uterus.
Additional examples of estrogen-dependent diseases include adenomyosis and rectovaginal endometriosis, which are particularly severe endometrial growth disorders characterized by the invasion of endometrial tissue into the uterine myometrium and rectovaginal zones, respectively. The term adenomyosis or uterine adenomyosis is used to describe the presence of both endometrial glands and stroma deep within the myometrium. This condition is associated with hypertrophy and hyperplasia of the subjacent muscle cells, which may ultimately result in an altered size and globulous morphology of the uterus. Due to the severity of this disorder, one of the key symptoms is strong menstrual and even non-menstrual pelvic pain with abnormal uterine bleeding. Like adenomyosis, rectovaginal endometriosis patients present with a variety of pain symptoms including dysmenorrhea, dyspareunia, chronic pelvic pain, dysuria, and dyschezia. Treatment options for rectovaginal endometriosis are limited. Since medical therapies are either ineffective or have considerable side effects, rectovaginal endometriosis patients often undergo surgical procedures to reduce the endometrial node, and may even be subject to resection of the bowel if the node infiltrates the rectal or sigmoidal wall.

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@amcrasto
/////////////////////////////////////////////////////////////////////////////////////////////////////
Obseva Announces U.S. FDA Acceptance of New Drug Application for Linzagolix
November 22, 2021 01:05 ET | Source: ObsEva SA………. https://www.globenewswire.com/news-release/2021/11/22/2338610/0/en/Obseva-Announces-U-S-FDA-Acceptance-of-New-Drug-Application-for-Linzagolix.html
FDA Accepts NDA for Linzagolix for the Management of Heavy Menstrual Bleeding Associated with Uterine Fibroids
GENEVA, Switzerland November 22, 2021 – Obseva SA (NASDAQ: OBSV; SIX: OBSN), a biopharmaceutical company developing and commercializing novel therapies to improve women’s reproductive health, today announced that the New Drug Application (NDA) for linzagolix for the management of heavy menstrual bleeding associated with uterine fibroids in premenopausal women has been accepted for review by the United States Food and Drug Administration (FDA). The submission is based on data from the two Phase 3 PRIMROSE trials. Linzagolix has a differentiated profile and if approved, would be the first and only GnRH receptor antagonist with flexible dosing options for uterine fibroids, including a low dose option to address the needs of women who cannot or do not want to take hormones.1,4 The FDA set a target action date of September 13, 2022 for this NDA under the Prescription Drug User Fee Act (PDUFA).
“Today marks an important milestone not only in the linzagolix clinical development process, but for Obseva as a company, and most importantly, the millions of women living with uterine fibroids throughout the US. Linzagolix is a significant innovation in the field of women’s health – an area that is consistently underinvested in – and we are incredibly excited about the potential of bringing this important treatment to market” said Brian O’Callaghan, CEO of Obseva. “We are encouraged by our positive Phase 3 PRIMROSE results. If approved, we believe linzagolix will address a significant unmet need in offering a more individualized treatment option for a broader range of women.”
The Phase 3 PRIMROSE trials of linzagolix (PRIMROSE 1: US; n=574 and PRIMROSE 2: Europe and US; n=535) investigated the efficacy and safety of two dosing regimens, 100mg once daily and 200mg once daily, alone or in combination with hormonal ABT (1 mg estradiol and 0.5 mg norethisterone acetate) for the treatment of heavy menstrual bleeding associated with uterine fibroids. The NDA submission comprises positive 24-week treatment results from both studies, as well as supportive results from Week 52 and the 76-week post-treatment follow-up.
“Uterine fibroids can have a devastating impact on women’s day-to-day life. With its unique dosing options, linzagolix has the potential to significantly advance medical options for women,” stated Elizabeth Garner, MD, MPH, Chief Medical Officer of Obseva. “A dosing option without hormonal ABT would be welcomed by the significant number of women who either have contraindications to or a personal preference to avoid the use of estrogen-based therapies, while also providing a dosing option for women in whom hormonal ABT is indicated.”
The linzagolix marketing authorization application (MAA) was validated by the European Medicine Agency (EMA) with an approval recommendation from the Committee for Medicinal Products for Human Use (CHMP) expected in Q4 2021. Obseva announced previously that the company has entered into a partnership with Syneos Health to support commercialization of linzagolix in the US and EU.
About Linzagolix
Linzagolix is a novel, once daily, oral GnRH receptor antagonist with a potentially best-in-class profile1,2,3. Linzagolix is the subject of submitted marketing authorization applications for the treatment of heavy menstrual bleeding associated with uterine fibroids and is currently in late-stage clinical development for the treatment of pain associated with endometriosis. Obseva licensed linzagolix from Kissei in late 2015 and retains worldwide commercial rights, excluding Asia, for the product. Linzagolix is not currently approved anywhere in the world.
About the Phase 3 PRIMROSE Program in Uterine Fibroids
PRIMROSE 1 & 2 were prospective, randomized, parallel group, double-blind, placebo-controlled Phase 3 studies that investigated the efficacy and safety of two dosing regimens of linzagolix, 100 mg and 200 mg once daily, alone and in combination with hormonal ABT (1 mg estradiol and 0.5 mg norethisterone acetate) for the treatment of heavy menstrual bleeding associated with uterine fibroids. PRIMROSE 1 was conducted in the United States and enrolled 574 women. PRIMROSE 2 was conducted in Europe and the United States and enrolled 535 women. Both trials comprised a 52-week treatment period followed by a 6-month post treatment follow-up period. Additional information can be found here.
About Uterine Fibroids
Uterine fibroids are common benign tumors of the muscular tissue of the uterus which affect women of childbearing age and can vary in size from undetectable to large bulky masses. Few long-term medical treatments are available, and as a result, approximately 300,000 hysterectomies are performed for uterine fibroids every year in the US.
The symptoms of uterine fibroids are wide-ranging and include heavy menstrual bleeding, anemia, pelvic pressure and bloating, urinary frequency and pain that can be extremely debilitating with a significant impact on quality of life. These symptoms can also have an impact on mental health, creating the additional burden of anxiety and distress.
About Obseva
Obseva is a biopharmaceutical company built to address some of the most challenging unmet needs in women’s health – an under-researched, under-invested field of medicine. With deep expertise in clinical development, Obseva is passionate about the pursuit of advances that benefit women and their health and the importance of delivering truly meaningful innovation in this space. Through strategic in-licensing and disciplined drug development, Obseva has established a late-stage clinical pipeline with development programs focused on new therapies for the treatment of uterine fibroids, endometriosis, and preterm labor. Obseva is listed on the Nasdaq Global Select Market and is traded under the ticker symbol “OBSV” and on the SIX Swiss Exchange where it is traded under the ticker symbol “OBSN”. For more information, please visit http://www.ObsEva.com.
About Kissei
Kissei is a Japanese pharmaceutical company with approximately 70 years of history, specialized in the field of urology, kidney-dialysis and unmet medical needs. Silodosin is a Kissei product for the treatment of the signs and symptoms of benign prostatic hyperplasia which is sold worldwide through its licensees. KLH-2109/OBE2109 is a new chemical entity discovered by Kissei R&D.

……………………………

PATENT
WO 2007046392
https://patents.google.com/patent/WO2007046392A1/en
PATENT
WO 2014042176
https://patents.google.com/patent/WO2014042176A1/en

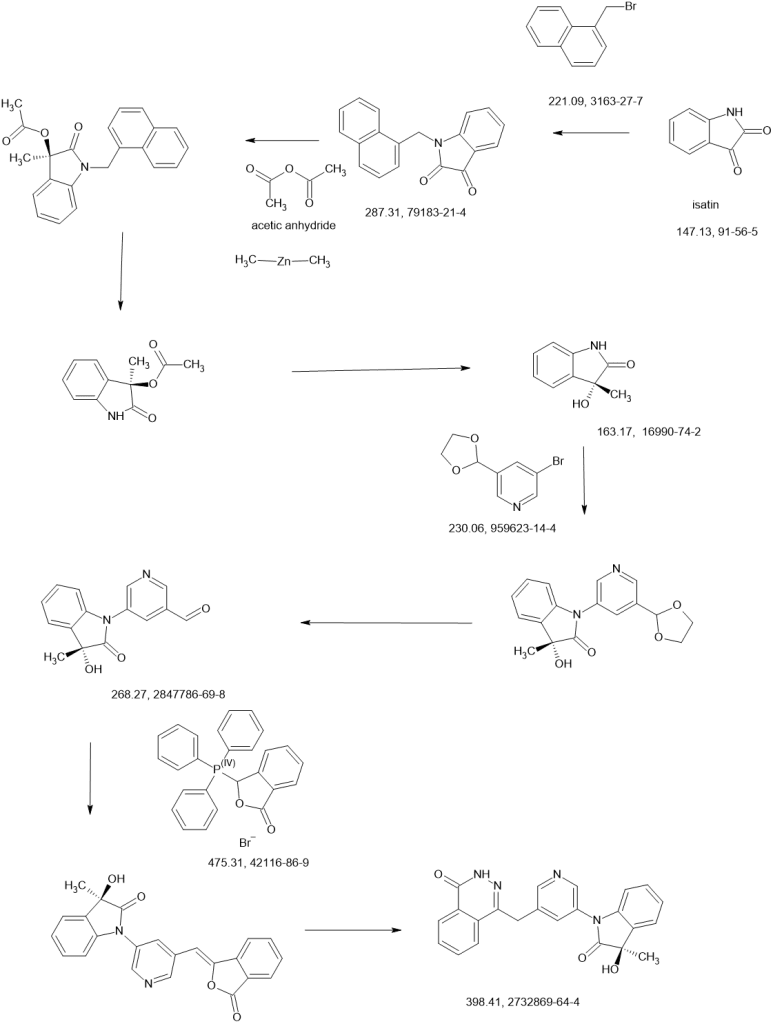
(Process 1)
Compound (D) can be produced by reacting compound (B) or a salt thereof with compound (C) in the presence of a base in a solvent. Examples of the solvent include halogen solvents such as dichloromethane, cyclic ethers such as tetrahydrofuran, 2-methyltetrahydrofuran, and tetrahydropyran, amide solvents such as N, N-dimethylformamide, aromatic hydrocarbon solvents such as toluene, A nitrile solvent such as acetonitrile, an ester solvent such as ethyl acetate, or a mixed solvent thereof and a mixed solvent thereof and water are preferable, and a mixed solvent of tetrahydrofuran and water is preferable. Examples of the base include organic bases such as triethylamine and pyridine, and inorganic bases such as sodium hydrogen carbonate, potassium hydrogen carbonate, cesium carbonate, sodium carbonate, and potassium carbonate, preferably triethylamine, sodium hydrogen carbonate, or potassium carbonate Is mentioned. The equivalent of the base may be an equivalent amount capable of neutralizing the salt and neutralizing the acid generated by the reaction. The equivalent of (C) can be used in an amount of 0.8 to 1.1 equivalents relative to (B), preferably 1.0 equivalent. The reaction temperature is usually 0 to 30 ° C., and the reaction time is usually 0.5 to 3 hours, although it varies depending on the raw material used, the solvent, the reaction temperature and the like. Examples of the salt of the compound (B) include a salt with an inorganic acid, a salt with an organic acid, a salt with an acidic amino acid, and the like. Examples of the salt with an inorganic acid include salts with hydrochloric acid, hydrobromic acid, nitric acid, sulfuric acid, phosphoric acid and the like. Examples of salts with organic acids include formic acid, acetic acid, trifluoroacetic acid, fumaric acid, oxalic acid, tartaric acid, maleic acid, citric acid, succinic acid, malic acid, methanesulfonic acid, benzenesulfonic acid, p-toluene And salts with sulfonic acid and the like. Examples of salts with acidic amino acids include salts with aspartic acid, glutamic acid and the like. Among these salts, salts with hydrochloric acid and methanesulfonic acid are preferable. Compound (C) used in Scheme 1 may be a commercially available product, or can be produced according to a known method or a method analogous thereto. Compound (D) may be isolated before the next step, but it can also be used in the next step without isolation.(Process 2)
Compound (F) can be produced by reacting compound (D) with compound (E) or a salt thereof in a solvent in the presence or absence of a base. Examples of the solvent include cyclic ethers such as tetrahydrofuran, 2-methyltetrahydrofuran, tetrahydropyran, amide solvents such as N, N-dimethylformamide, aromatic hydrocarbon solvents such as toluene, nitrile solvents such as acetonitrile, An ester solvent such as ethyl acetate or a mixed solvent thereof and a mixed solvent thereof with water, and the like are preferable, and a mixed solvent of tetrahydrofuran and water is preferable. Examples of the base include organic bases such as N, N-dimethylaminopyridine, triethylamine, N-methylpyrrolidine, N-methylmorpholine, diisopropylethylamine, and preferably N, N-dimethylaminopyridine, triethylamine and the like. . The equivalent of the base can be used in an amount of 0.1 to 2.0 equivalents relative to the compound (E), preferably 0.1 to 0.5 equivalents (provided that when a salt of the compound (E) is used, Further base necessary for neutralization is required). The reaction temperature is from room temperature to 60 ° C., and the reaction time is usually from 1 to 24 hours, although it varies depending on the raw material used, the solvent, the reaction temperature, and the like. Examples of the salt of compound (E) include a salt with an inorganic acid, a salt with an organic acid, a salt with an acidic amino acid, and the like. Examples of the salt with an inorganic acid include salts with hydrochloric acid, hydrobromic acid, nitric acid, sulfuric acid, phosphoric acid and the like. Examples of salts with organic acids include formic acid, acetic acid, trifluoroacetic acid, fumaric acid, oxalic acid, tartaric acid, maleic acid, citric acid, succinic acid, malic acid, methanesulfonic acid, benzenesulfonic acid, p-toluene And salts with sulfonic acid and the like. Examples of salts with acidic amino acids include salts with aspartic acid, glutamic acid and the like. Compound (F) may be isolated before the next step, but it can also be used in the next step without isolation.(Process 3)
The intramolecular cyclization and hydrolysis reaction in this step can be performed simultaneously or separately.
(Step 3-1)
Compound (A) can be produced by subjecting compound (F) to intramolecular cyclization and hydrolysis in the presence of a base in a solvent. Examples of the solvent include cyclic ethers such as tetrahydrofuran, 2-methyltetrahydrofuran and tetrahydropyran, lower alcohols such as methanol, ethanol and 2-propanol, amide solvents such as N, N-dimethylformamide, and nitriles such as acetonitrile. Examples thereof include a solvent and the like or a mixed solvent of a mixed solvent thereof and water, and a mixed solvent of tetrahydrofuran / methanol / water is preferable. Examples of the base include inorganic bases such as sodium hydroxide, potassium hydroxide, lithium hydroxide and sodium hydride, and metal alkoxides such as sodium methoxide and potassium tert-butoxide, preferably lithium hydroxide and sodium And methoxide. The base can be used in an amount of 3.0 to 6.0 equivalents, preferably 4.0 to 4.5 equivalents, relative to compound (F). The reaction temperature is usually from 0 to 20 ° C., and the reaction time is usually from 1 to 10 hours, although it varies depending on the raw material used, solvent, reaction temperature and the like.
(Step 3-2)
When isolating compound (G), compound (G) can be produced by subjecting compound (F) to an intramolecular cyclization reaction in a solvent in the presence of a base. Examples of the solvent include cyclic ethers such as tetrahydrofuran, 2-methyltetrahydrofuran and tetrahydropyran, lower alcohols such as methanol, ethanol and 2-propanol, amide solvents such as N, N-dimethylformamide, and nitriles such as acetonitrile. Examples thereof include a solvent and the like or a mixed solvent thereof, and a mixed solvent of tetrahydrofuran / methanol is preferable. Examples of the base include inorganic bases such as sodium hydroxide, potassium hydroxide, lithium hydroxide or sodium hydride, metal alkoxides such as sodium methoxide and potassium tert-butoxide, and lithium hydroxide, sodium methoxide and the like. preferable. The base can be used in an amount of 0.1 to 1.5 equivalents, preferably 1.0 to 1.1 equivalents, relative to compound (F). The reaction temperature is usually from 0 to 20 ° C., and the reaction time is usually from 1 to 10 hours, although it varies depending on the raw material used, solvent, reaction temperature and the like.
(Step 3-3)
The hydrolysis reaction in this step can be performed by the same method as in step 3-1 or a method analogous thereto.(Process 4)
Compound (A) can be converted to a salt thereof by a conventional method. Examples of such salts include inorganic salts such as sodium salt, potassium salt, calcium salt, magnesium salt, triethylamine, diisopropylamine, N, N′-dibenzylethylenediamine, ethanolamine, (2-hydroxyethyl) trimethylammonium. (Hereinafter referred to as choline), addition salts with organic bases such as N-methylglucamine, arginine, lysine and the like, and choline salts are preferred. Examples of the reagent used for conversion to the choline salt include choline hydroxide, choline bicarbonate, choline chloride and choline acetate.Here, the compound (B) and the salt thereof used in the above-mentioned scheme 1 are commercially available, or manufactured by the method described in a) to c), the method described in the reference examples, or a method analogous thereto. Can do.
a) JP-A 64-29373
b) Synthetic Communications, 32, 2565 (2002)
c) Synthesis, 200 (1977)Further, the compound (E) or a salt thereof used in the scheme 1 can be produced by the method described in Patent Document 1, the method described in Reference Examples, or a method analogous thereto.The compound obtained in the production process in the present specification includes hydrates or solvates thereof, and any of them can be used. Furthermore, the compound obtained in the production process in the present specification may have tautomers and / or geometric isomers, any of which can be used, and also a mixture thereof. be able to.By the production method of the present invention, the compound (A) useful as a pharmaceutical product or a salt thereof can be obtained in high yield and high purity through the compound (D) which is a production intermediate.The content of the present invention will be described in more detail by the following examples, but the present invention is not limited to the content.Reference example 1
Dimethyl 4-oxothiolane-2,3-dicarboxylate methylthioglycolate (15.0 g), tetrahydrofuran (45 g), piperidine (0.361 g) in a reaction mixture at room temperature with dimethyl maleate (21.4 g) in tetrahydrofuran (30 g) The solution was added. To the reaction mixture was added 20% sodium methoxide in methanol (43 g) at 55 ° C. under a nitrogen atmosphere. The reaction mixture was stirred at reflux for 3 hours. Diisopropyl ether (105 g) and acetic acid (0.85 g) were added to the reaction mixture at 45-50 ° C., and then cooled. The suspension was filtered to obtain wet crystals (43.3 g) of sodium salt of dimethyl 4-oxothiolane-2,3-dicarboxylate. The wet crystals were added to a mixture of 85% phosphoric acid (9.8 g), water (20 g) and ethyl acetate (150 g) at room temperature, and the aqueous layer was removed. The obtained organic layer was washed with 10% brine and then dried over anhydrous magnesium sulfate. The drying agent was removed by filtration, and the filtrate was concentrated under reduced pressure to obtain the title compound (22.7 g).Reference example 2
Dimethyl 4- (hydroxyimino) thiolane-2,3-dicarboxylate Dimethyl 4-oxothiolane-2,3-dicarboxylate (10.0 g), pyridine (5.44 g), hydroxylamine hydrochloride (3.34 g) Was stirred at 50 ° C. for 1 hour. Ethyl acetate and 7% aqueous phosphoric acid solution were added to the reaction mixture at room temperature, and the aqueous layer was removed. The obtained organic layer was washed with 5% sodium bicarbonate water and 10% brine. The organic layer was dried over anhydrous sodium sulfate. After removing the desiccant by filtration, the filtrate was concentrated under reduced pressure to obtain the title compound (10.4 g).Reference example 3
4-Aminothiophene-2,3-dicarboxylic acid dimethyl hydrochloride 4- (hydroxyimino) thiolane-2,3-dicarboxylate (10.4 g) in acetic acid (32 g) solution in 4N-hydrogen chloride / ethyl acetate solution ( 120 g) was added at room temperature. The reaction mixture was stirred at room temperature for 8 hours. After filtering the suspension, the obtained solid was dried to obtain the title compound (9.42 g).Reference example 4
4-Aminothiophene-2,3-dicarboxylic acid dimethyl methanesulfonate To a solution of methanesulfonic acid (80.0 g) in ethyl acetate (900 g), dimethyl 4- (hydroxyimino) thiolane-2,3-dicarboxylate (97. 1 g) of ethyl acetate (500 g) was added at 65-75 ° C. The reaction mixture was stirred at the same temperature for 2 hours. Methyl isobutyl ketone (100 g) was added at 45-50 ° C. and cooled to room temperature. After filtering the suspension, the obtained solid was dried to obtain the title compound (102 g).Reference Example 5
1,2-difluoro-3-[(4-fluoro-2-methoxyphenoxy) methyl] -4-methoxybenzene sodium borohydride in a solution of 2,3-difluoro-6-methoxybenzaldehyde (150 g) in toluene (900 g) (13.2 g) of 0.1N sodium hydroxide aqueous solution (180 g) was added at 35 to 39 ° C. The reaction mixture was stirred at the same temperature for 5 hours. After cooling the reaction mixture to room temperature, the aqueous layer was removed. The obtained organic layer was washed with 20% brine to obtain a toluene solution of 2,3-difluoro-6-methoxybenzyl alcohol. To this solution was added concentrated hydrochloric acid (610 g) at room temperature. The reaction mixture was stirred at 38-43 ° C. for 5 hours. After cooling the reaction mixture to room temperature, the aqueous layer was removed. The obtained organic layer was washed with water and 20% brine to obtain a toluene solution of 3- (chloromethyl) -1,2-difluoro-4-methoxybenzene. To this solution, 4-fluoro-2-methoxyphenol (125 g) and tetrabutylammonium bromide (56.2 g) were added at room temperature. A 25% aqueous sodium hydroxide solution (170 g) was added to the reaction mixture at 60 to 63 ° C., and the mixture was stirred at the same temperature for 4 hours. Water was added to the reaction mixture and the aqueous layer was removed. The obtained organic layer was washed with water and concentrated under reduced pressure. The residue was dissolved in 2-propanol and water was added. After filtering the suspension, the obtained solid was dried to obtain the title compound (232 g).Reference Example 6
1,2-difluoro-3-[(4-fluoro-2-methoxy-5-nitrophenoxy) methyl] -4-methoxybenzene 1,2-difluoro-3-[(4-fluoro-2-methoxyphenoxy) methyl ] To a solution of 4-methoxybenzene (158 g) in acetic acid (1200 g) was added 60% nitric acid (72.2 g) at 59-62 ° C., and the mixture was stirred at the same temperature for 2 hours. Water (1200 g) was added to the suspension at 15 to 19 ° C., and the mixture was stirred at the same temperature for 1 hour. After filtering the suspension, the obtained solid was washed with water to obtain wet crystals of the title compound (190 g, Net amount 168 g).Reference Example 7
2-Fluoro-5-[(2,3-difluoro-6-methoxyphenyl) methoxy] -4-methoxyaniline Raney nickel (2.5 g), ethyl acetate (180 g), 1,2-difluoro-3-[(4 -Fluoro-2-methoxy-5-nitrophenoxy) methyl] -4-methoxybenzene wet crystal (10.9 g, Net amount 10.0 g) was stirred at room temperature under a hydrogen atmosphere for 4 hours. The catalyst was removed by filtration, and the filtrate was concentrated under reduced pressure. The residue was dissolved with methanol and water was added. After filtering the suspension, the obtained solid was dried to obtain the title compound (7.97 g).Example 1
4- (phenoxycarbonylamino) thiophene-2,3-dicarboxylic acid dimethyl potassium carbonate (17.1 g), water (90 g), tetrahydrofuran (150 g) and 4-aminothiophene-2,3-dicarboxylic acid dimethyl hydrochloride (30 0.06) was added phenyl chloroformate (18.6 g) at 6-13 ° C. The reaction mixture was stirred at 12-13 ° C. for 30 minutes, and then the aqueous layer was removed. To the obtained organic layer, tert-butyl methyl ether was added and washed with 20% brine. The obtained organic layer was concentrated under reduced pressure. The residue was dissolved with diisopropyl ether and n-hexane was added. After filtering the suspension, the obtained solid was dried to obtain the title compound (37.0 g).
1 H-NMR (DMSO-d 6 ) δ ppm: 3.82 (3H, s), 3.82 (3H, s), 7.13-7.30 (3H, m), 7.40-7.46 (2H, m), 7.80 (1H, s ), 10.24 (1H, s)Example 2
4- {3- [2-Fluoro-5- (2,3-difluoro-6-methoxybenzyloxy) -4-methoxyphenyl] ureido} dimethyl thiophene-2,3-dicarboxylate 2-fluoro-5-[( 2,3-difluoro-6-methoxyphenyl) methoxy] -4-methoxyaniline (7.70 g), dimethyl 4- (phenoxycarbonylamino) thiophene-2,3-dicarboxylate (8.65 g), triethylamine (0. 37 g) and tetrahydrofuran (80 mL) were stirred at room temperature for 24 hours. The reaction mixture was concentrated under reduced pressure. Ethyl acetate and methanol were added to the residue. After filtering the suspension, the obtained solid was dried to obtain the title compound (12.0 g).
1 H-NMR (DMSO-d 6 ) δ ppm: 3.71 (3H, s), 3.82 (3H, s), 3.83 (3H, s), 3.89 (3H, s), 5.00 (2H, d, J = 1.6 Hz), 6.87-6.93 (1H, m), 7.00 (1H, d, J = 12.8Hz), 7.41-7.50 (1H, m), 7.75 (1H, d, J = 8.0Hz), 7.94 (1H, s ), 8.82 (1H, s), 8.95 (1H, s)Example 3
3- [2-Fluoro-5- (2,3-difluoro-6-methoxybenzyloxy) -4-methoxyphenyl] -2,4-dioxo-1,2,3,4-tetrahydrothieno [3,4 d] methyl pyrimidine-5-carboxylate 4- {3- [2-fluoro-5- (2,3-difluoro-6-methoxybenzyloxy) -4-methoxyphenyl] ureido} thiophene-2,3-dicarboxylic acid A methanol solution (3.48 g) of 28% sodium methoxide was added to a suspension of dimethyl (10.0 g) in tetrahydrofuran (40 g), stirred at room temperature for 3 hours, and acetic acid (1.30 g) was added. The reaction mixture was concentrated under reduced pressure. Methanol was added to the residue, and water was further added. After filtering the suspension, the obtained solid was dried to obtain the title compound (8.58 g).
1 H-NMR (DMSO-d 6 ) δ ppm: 3.79 (3H, s), 3.81 (3H, s), 3.84 (3H, s), 4.95 (2H, s), 6.88-6.94 (1H, m), 7.08 (1H, d, J = 11.6Hz), 7.19-7.23 (2H, m), 7.44-7.53 (1H, m), 11.62 (1H, s)Example 4
4- (phenoxycarbonylamino) thiophene-2,3-dicarboxylate potassium carbonate (9.38 kg), water (49 kg), tetrahydrofuran (82 kg), dimethyl 4-aminothiophene-2,3-dicarboxylate hydrochloride (16 4 kg) of the reaction mixture was stirred for 40 minutes, and then phenyl chloroformate (10.1 kg) was added at 11-21 ° C. The reaction mixture was stirred for 30 minutes, and then the aqueous layer was removed to obtain a tetrahydrofuran solution of the title compound.Example 5
4- {3- [2-Fluoro-5- (2,3-difluoro-6-methoxybenzyloxy) -4-methoxyphenyl] ureido} dimethyl thiophene-2,3-dicarboxylate 4-obtained in Example 4 To a tetrahydrofuran solution of dimethyl (phenoxycarbonylamino) thiophene-2,3-dicarboxylate, 2-fluoro-5-[(2,3-difluoro-6-methoxyphenyl) methoxy] -4-methoxyaniline (17.0 kg), Tetrahydrofuran (8.5 kg) and triethylamine (1.1 kg) were added, and the mixture was stirred at 50 ° C. for 3.5 hours to obtain a tetrahydrofuran solution of the title compound.Example 6
3- [2-Fluoro-5- (2,3-difluoro-6-methoxybenzyloxy) -4-methoxyphenyl] -2,4-dioxo-1,2,3,4-tetrahydrothieno [3,4 d] pyrimidine-5-carboxylic acid tetrahydrofuranate 4- {3- [2-fluoro-5- (2,3-difluoro-6-methoxybenzyloxy) -4-methoxyphenyl] ureido} obtained in Example 5 Methanol (41 kg) and water (47 kg) are added to a tetrahydrofuran solution of dimethyl thiophene-2,3-dicarboxylate, a 7.3% lithium hydroxide aqueous solution (80.1 kg) is added at 11 to 13 ° C., and 90 ° C. at 11 ° C. Stir for minutes. Acetic acid (11.4 kg) was added to the reaction mixture at 9 to 16 ° C., and acetic acid (13.0 kg) was further added at 29 to 31 ° C. Seed crystals were added to the reaction mixture, and the mixture was stirred at the same temperature for 30 minutes. Water (34 kg) was added to the suspension and stirred at 30 ° C. for 40 minutes. The suspension was stirred at 4-9 ° C. for 90 minutes. After the suspension was filtered, the obtained solid was washed with a mixed solution of methanol (54 kg) and water (68 kg) to give wet crystals of the title compound (31.64 kg, Net amount (compound (A) free form equivalent)) 26 0.7 kg) was obtained.
A part of the wet crystals of the title compound was dried under reduced pressure at an external temperature of 60 ° C., and 1 H-NMR, HPLC and powder X-ray diffraction were measured on the obtained dried crystals of the title compound.
1 H-NMR (DMSO-d 6 ) δ ppm: 1.68-1.82 (3H, m), 3.53-3.65 (3H, m), 3.80 (3H, s), 3.81 (3H, s), 4.94-4.98 (2H , m), 6.87-6.94 (1H, m), 7.13 (1H, d, J = 11.2Hz), 7.25 (1H, d, J = 7.2Hz), 7.39 (1H, s), 7.43-7.52 (1H, m), 11,99 (1H, s), 14.53 (1H, s)
PATENT
WO 2020089190
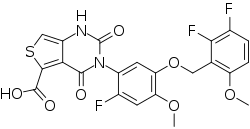
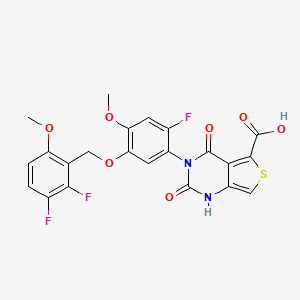
https://patents.google.com/patent/WO2020089190A2/enFor example, the GnRH antagonist may be 3-[2-fluoro-5-(2,3-difluoro-6-methoxybenzyloxy)4- methoxyphenyl]-2,4-dioxo-1 ,2,3,4- tetrahydrothieno [3,4d]pyrimidine-5-carboxylic acid, or a pharmaceutically acceptable salt thereof. The salt may be, for instance, the choline salt thereof, represented by formula (Via), below.

Compound (VI) and pharmaceutically acceptable salts thereof, such as the choline salt thereof (compound (Via)), can be synthesized, for example, using the methodology described in WO 2014/042176, the disclosure of which is incorporated herein by reference in its entirety. An exemplary synthetic scheme that may be used for the preparation of compound (VI) and the choline salt thereof is shown in Scheme 1 , below.Scheme 1 . Exemplary preparation of compound (VI) and the choline salt thereof




wherein Ri and R are each independently C alkoxy groups; LG is a nucleofugal leaving group, such as chlorine or bromine, among others; R represents an optional substituent, such as halogen, acyl group, C alkyl group, or a nitro substituent; DMAP denotes A/-dimethylaminopyridine; and TEA denotes trimethylamine.Crystalline compound (Via) has been characterized spectroscopically, for instance, in US Patent No. 9,169,266, the disclosure of which is incorporated herein by reference in its entirety. The foregoing crystalline form has been shown to exhibit characteristic X-ray powder diffraction peaks at about 7.10 2Q, about 11 .5° 2Q, about 19.4° 2Q, about 21 .5° 2Q, about 22.0° 2Q, about 22.6° 2Q, about 23.5° 2Q, and about 26.2° 2Q. Additionally, this crystalline form exhibits 13C solid-state nuclear magnetic resonance (NMR) peaks centered at about 55.5 ppm, about 57.1 ppm, about 58.7 ppm, about 69.8 ppm, about 98.1 ppm, about 110.3 ppm, about 1 1 1 .6 ppm, about 113.7 ppm, about 1 18.0 ppm, about 145.3 ppm, about 149.8 ppm, and about 155.8 ppm. This crystalline form further exhibits 19F solid-state NMR peaks centered at about -151.8 ppm, -145.2 ppm, and -131 .6 ppm.Compound (VI), as well as pharmaceutically acceptable salts thereof, such as the choline salt thereof, exhibit a high affinity for human GnRH receptor (27.4 nM). Using the compositions and methods described herein, a patient that is presenting with or has been diagnosed as having, adenomyosis or rectovaginal endometriosis may be administered a compound of formula (VI), or a pharmaceutically acceptable salt thereof, such as the choline salt thereof, to treat the disease or ameliorate one or more symptoms of the disease. Exemplary doses of compound (VI) and pharmaceutically acceptable salts thereof, such as the choline salt thereof, include doses of from 25 mg to 500 mg daily, such as doses of 100 mg per day and 200 mg per day. Additional dosing information is provided below.3-Aminoalkyl pyrimidine-2, 4(1 H,3H)-dionesAdditional GnRH antagonists that may be used in conjunction with the compositions and methods described herein include optionally substituted 3-aminoalkyl pyrimidine-2, 4(1 H,3H)-dione derivatives, such as compounds represented by formula (VII)

PATENTWO 2021023876https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2021023876&_cid=P11-KWFRM2-91270-1
In some embodiments, the compound is the choline salt of the compound represented by formula (VI), choline 3- [2-fluoro-5- (2,3-difluoro-6-methoxybenzyloxy) 4-methoxyphenyI] -2,4- dioxo-1,2,3,4-
tetrahydrothieno [3,4d] pyrimidine-5-carboxylate. It is to be understood that references herein to a compound represented by formula (VI) specifically include the choline salt of compound (VI), which is represented by formula (VIa), below.
In some embodiments, the choline 3- [2-fluoro-5- (2,3-difluoro-6-methoxybenzyloxy) 4-methoxyphenyI] -2,4-dioxo-1,2,3,4- tetrahydrothieno [3,4d ] pyrimidine-5-carboxylate is in a crystalline state.
PATENT
WO 2021023877
References
- ^ Jump up to:a b c “Linzagolix – Kissei Pharmaceutical/ObsEva – AdisInsight”.
- ^ Jump up to:a b Ezzati M, Carr BR (2015). “Elagolix, a novel, orally bioavailable GnRH antagonist under investigation for the treatment of endometriosis-related pain”. Womens Health (Lond). 11 (1): 19–28. doi:10.2217/whe.14.68. PMID 25581052.
- ^ Chodankar, Rohan; Allison, Jennifer (2018). “New Horizons in Fibroid Management”. Current Obstetrics and Gynecology Reports. 7 (2): 106–115. doi:10.1007/s13669-018-0242-6. ISSN 2161-3303.
External links
| Clinical data | |
|---|---|
| Trade names | Yselty |
| Other names | KLH-2109; OBE-2109 |
| Routes of administration | By mouth[1][2] |
| Drug class | GnRH modulator; GnRH antagonist; Antigonadotropin |
| ATC code | None |
| Identifiers | |
| showIUPAC name | |
| CAS Number | 935283-04-8 |
| PubChem CID | 16656889 |
| ChemSpider | 17590169 |
| UNII | 7CDW97HUEX |
| KEGG | D11608 |
| ChEMBL | ChEMBL3668014 |
| Chemical and physical data | |
| Formula | C22H15F3N2O7S |
| Molar mass | 508.42 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| showSMILES | |
| showInChI |
////////LINZAGOLIX, Hormone Antagonists, WHO 10711, KLH-2109, KLH 2109, OBE-2109, OBE 2109

NEW DRUG APPROVALS
ONE TIME
$10.00
Ropeginterferon alfa-2b
PCDLPQTHSL GSRRTLMLLA QMRRISLFSC LKDRHDFGFP QEEFGNQFQK AETIPVLHEM
IQQIFNLFST KDSSAAWDET LLDKFYTELY QQLNDLEACV IQGVGVTETP LMKEDSILAV
RKYFQRITLY LKEKKYSPCA WEVVRAEIMR SFSLSTNLQE SLRSKE
(Disulfide bridge: 2-99, 30-139)
Ropeginterferon alfa-2b
- AOP2014
CAS 1335098-50-4
UNII981TME683S
FDA APPROVED, 2021/11/12, BESREMI
PEPTIDE, Antineoplastic, Antiviral
Polycythemia vera (PV) is the most common Philadelphia chromosome-negative myeloproliferative neoplasm (MPN), characterized by increased hematocrit and platelet/leukocyte counts, an increased risk for hemorrhage and thromboembolic events, and a long-term propensity for myelofibrosis and leukemia.1,2 Interferon alfa-2b has been used for decades to treat PV but requires frequent dosing and is not tolerated by all patients.2 Ropeginterferon alfa-2b is a next-generation mono-pegylated type I interferon produced from proline-IFN-α-2b in Escherichia coli that has high tolerability and a long half-life.4,6 Ropeginterferon alfa-2b has shown efficacy in PV in in vitro and in vivo models and clinical trials.3,4
Ropeginterferon alfa-2b was approved by the FDA on November 12, 2021, and is currently marketed under the trademark BESREMi by PharmaEssentia Corporation.6
Ropeginterferon alfa-2b, sold under the brand name Besremi, is a medication used to treat polycythemia vera.[1][2][3][4] It is an interferon.[1][3] It is given by injection.[1][3]
The most common side effects include low levels of white blood cells and platelets (blood components that help the blood to clot), muscle and joint pain, tiredness, flu-like symptoms and increased blood levels of gamma-glutamyl transferase (a sign of liver problems).[3] Ropeginterferon alfa-2b can cause liver enzyme elevations, low levels of white blood cells, low levels of platelets, joint pain, fatigue, itching, upper airway infection, muscle pain and flu-like illness.[2] Side effects may also include urinary tract infection, depression and transient ischemic attacks (stroke-like attacks).[2]
It was approved for medical use in the European Union in February 2019,[3] and in the United States in November 2021.[2][5] Ropeginterferon alfa-2b is the first medication approved by the U.S. Food and Drug Administration (FDA) to treat polycythemia vera that people can take regardless of their treatment history, and the first interferon therapy specifically approved for polycythemia vera.[2]
https://www.fda.gov/news-events/press-announcements/fda-approves-treatment-rare-blood-disease#:~:text=FDA%20NEWS%20RELEASE-,FDA%20Approves%20Treatment%20for%20Rare%20Blood%20Disease,FDA%2DApproved%20Option%20Patients%20Can%20Take%20Regardless%20of%20Previous%20Therapies,-ShareFor Immediate Release:November 12, 2021
Today, the U.S. Food and Drug Administration approved Besremi (ropeginterferon alfa-2b-njft) injection to treat adults with polycythemia vera, a blood disease that causes the overproduction of red blood cells. The excess cells thicken the blood, slowing blood flow and increasing the chance of blood clots.
“Over 7,000 rare diseases affect more than 30 million people in the United States. Polycythemia vera affects approximately 6,200 Americans each year,” said Ann Farrell, M.D., director of the Division of Non-Malignant Hematology in the FDA’s Center for Drug Evaluation and Research. “This action highlights the FDA’s commitment to helping make new treatments available to patients with rare diseases.”
Besremi is the first FDA-approved medication for polycythemia vera that patients can take regardless of their treatment history, and the first interferon therapy specifically approved for polycythemia vera.
Treatment for polycythemia vera includes phlebotomies (a procedure that removes excess blood cells though a needle in a vein) as well as medicines to reduce the number of blood cells; Besremi is one of these medicines. Besremi is believed to work by attaching to certain receptors in the body, setting off a chain reaction that makes the bone marrow reduce blood cell production. Besremi is a long-acting drug that patients take by injection under the skin once every two weeks. If Besremi can reduce excess blood cells and maintain normal levels for at least one year, then dosing frequency may be reduced to once every four weeks.
The effectiveness and safety of Besremi were evaluated in a multicenter, single-arm trial that lasted 7.5 years. In this trial, 51 adults with polycythemia vera received Besremi for an average of about five years. Besremi’s effectiveness was assessed by looking at how many patients achieved complete hematological response, which meant that patients had a red blood cell volume of less than 45% without a recent phlebotomy, normal white cell counts and platelet counts, a normal spleen size, and no blood clots. Overall, 61% of patients had a complete hematological response.
Besremi can cause liver enzyme elevations, low levels of white blood cells, low levels of platelets, joint pain, fatigue, itching, upper airway infection, muscle pain and flu-like illness. Side effects may also include urinary tract infection, depression and transient ischemic attacks (stroke-like attacks).
Interferon alfa products like Besremi may cause or worsen neuropsychiatric, autoimmune, ischemic (not enough blood flow to a part of the body) and infectious diseases, which could lead to life-threatening or fatal complications. Patients who must not take Besremi include those who are allergic to the drug, those with a severe psychiatric disorder or a history of a severe psychiatric disorder, immunosuppressed transplant recipients, certain patients with autoimmune disease or a history of autoimmune disease, and patients with liver disease.
People who could be pregnant should be tested for pregnancy before using Besremi due to the risk of fetal harm.
Besremi received orphan drug designation for this indication. Orphan drug designation provides incentives to assist and encourage drug development for rare diseases.
The FDA granted the approval of Besremi to PharmaEssentia Corporation.
Medical uses
In the European Union, ropeginterferon alfa-2b is indicated as monotherapy in adults for the treatment of polycythemia vera without symptomatic splenomegaly.[3] In the United States it is indicated for the treatment of polycythemia vera.[1][2][5]
History
The effectiveness and safety of ropeginterferon alfa-2b were evaluated in a multicenter, single-arm trial that lasted 7.5 years.[2] In this trial, 51 adults with polycythemia vera received ropeginterferon alfa-2b for an average of about five years.[2] The effectiveness of ropeginterferon alfa-2b was assessed by looking at how many participants achieved complete hematological response, which meant that participants had a red blood cell volume of less than 45% without a recent phlebotomy, normal white cell counts and platelet counts, a normal spleen size, and no blood clots.[2] Overall, 61% of participants had a complete hematological response.[2] The U.S. Food and Drug Administration (FDA) granted the application for Ropeginterferon_alfa-2b orphan drug designation and granted the approval of Besremi to PharmaEssentia Corporation[2]
REF
- Bartalucci N, Guglielmelli P, Vannucchi AM: Polycythemia vera: the current status of preclinical models and therapeutic targets. Expert Opin Ther Targets. 2020 Jul;24(7):615-628. doi: 10.1080/14728222.2020.1762176. Epub 2020 May 18. [Article]
- How J, Hobbs G: Use of Interferon Alfa in the Treatment of Myeloproliferative Neoplasms: Perspectives and Review of the Literature. Cancers (Basel). 2020 Jul 18;12(7). pii: cancers12071954. doi: 10.3390/cancers12071954. [Article]
- Verger E, Soret-Dulphy J, Maslah N, Roy L, Rey J, Ghrieb Z, Kralovics R, Gisslinger H, Grohmann-Izay B, Klade C, Chomienne C, Giraudier S, Cassinat B, Kiladjian JJ: Ropeginterferon alpha-2b targets JAK2V617F-positive polycythemia vera cells in vitro and in vivo. Blood Cancer J. 2018 Oct 4;8(10):94. doi: 10.1038/s41408-018-0133-0. [Article]
- Gisslinger H, Zagrijtschuk O, Buxhofer-Ausch V, Thaler J, Schloegl E, Gastl GA, Wolf D, Kralovics R, Gisslinger B, Strecker K, Egle A, Melchardt T, Burgstaller S, Willenbacher E, Schalling M, Them NC, Kadlecova P, Klade C, Greil R: Ropeginterferon alfa-2b, a novel IFNalpha-2b, induces high response rates with low toxicity in patients with polycythemia vera. Blood. 2015 Oct 8;126(15):1762-9. doi: 10.1182/blood-2015-04-637280. Epub 2015 Aug 10. [Article]
- EMA Approved Products: Besremi (ropeginterferon alfa-2b ) solution for injection [Link]
- FDA Approved Drug Products: BESREMi (ropeginterferon alfa-2b-njft) injection [Link]

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@amcrasto
/////////////////////////////////////////////////////////////////////////////////////////////////////
References
- ^ Jump up to:a b c d e https://www.accessdata.fda.gov/drugsatfda_docs/label/2021/761166s000lbl.pdf
- ^ Jump up to:a b c d e f g h i j k l “FDA Approves Treatment for Rare Blood Disease”. U.S. Food and Drug Administration (FDA) (Press release). 12 November 2021. Retrieved 12 November 2021.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain. - ^ Jump up to:a b c d e f g “Besremi EPAR”. European Medicines Agency (EMA). Retrieved 14 November 2021. Text was copied from this source which is copyright European Medicines Agency. Reproduction is authorized provided the source is acknowledged.
- ^ Wagner SM, Melchardt T, Greil R (March 2020). “Ropeginterferon alfa-2b for the treatment of patients with polycythemia vera”. Drugs of Today. Barcelona, Spain. 56 (3): 195–202. doi:10.1358/dot.2020.56.3.3107706. PMID 32282866. S2CID 215758794.
- ^ Jump up to:a b “U.S. FDA Approves Besremi (ropeginterferon alfa-2b-njft) as the Only Interferon for Adults With Polycythemia Vera” (Press release). PharmaEssentia. 12 November 2021. Retrieved 14 November 2021 – via Business Wire.
External links
- “Ropeginterferon alfa-2b”. Drug Information Portal. U.S. National Library of Medicine.
- Clinical trial number NCT01193699 for “Safety Study of Pegylated Interferon Alpha 2b to Treat Polycythemia Vera (PEGINVERA)” at ClinicalTrials.gov
- Clinical trial number NCT02218047 for “AOP2014 vs. BAT in Patients With Polycythemia Vera Who Previously Participated in the PROUD-PV Study. (CONTI-PV)” at ClinicalTrials.gov
| Clinical data | |
|---|---|
| Trade names | Besremi |
| Other names | AOP2014, ropeginterferon alfa-2b-njft |
| License data | EU EMA: by INNUS DailyMed: Ropeginterferon_alfa |
| Pregnancy category | Contraindicated |
| Routes of administration | Subcutaneous |
| Drug class | Interferon |
| ATC code | L03AB15 (WHO) |
| Legal status | |
| Legal status | US: ℞-only [1][2]EU: Rx-only [3] |
| Identifiers | |
| CAS Number | 1335098-50-4 |
| DrugBank | DB15119 |
| UNII | 981TME683S |
| KEGG | D11027 |
/////////Ropeginterferon alfa-2b, FDA 2021, APPROVALS 2021, BESREMI, PEPTIDE, Antineoplastic, Antiviral, AOP 2014, PharmaEssentia

NEW DRUG APPROVALS
ONE TIME
$10.00
SULCONAZOLE

SULCONAZOLEсульконазол , سولكونازول , 硫康唑
- Molecular FormulaC18H15Cl3N2S
- Average mass397.749 Da
1H-Imidazole, 1-[2-[[(4-chlorophenyl)methyl]thio]-2-(2,4-dichlorophenyl)ethyl]- [ACD/Index Name]
4332
5D9HAA5Q5S
61318-90-9[RN]
(±)-1-[2,4-Dichloro-b-[(p-chlorobenzyl)thio]phenethyl]imidazole
1-[2-[[(4-Chlorophenyl)methyl]thio]-2-(2,4-dichlorophenyl)ethyl]-1H-imidazole: SulconazoleCAS Registry Number: 61318-90-9
CAS Name: 1-[2-[[(4-Chlorophenyl)methyl]thio]-2-(2,4-dichlorophenyl)ethyl]-1H-imidazole
Additional Names: (±)-1-[2,4-dichloro-b-[(p-chlorobenzyl)thio]phenethyl]imidazole
Molecular Formula: C18H15Cl3N2S
Molecular Weight: 397.75
Percent Composition: C 54.35%, H 3.80%, Cl 26.74%, N 7.04%, S 8.06%
Literature References: Prepn: K. A. M. Walker, DE2541833; idem,US4055652 (1976, 1977 both to Syntex). HPLC determn in plasma: M. Fass et al.,J. Pharm. Sci.70, 1338 (1981). Mechanism of action study: W. H. Beggs, Biochem. Arch.10, 117 (1994). Clinical trial in tinea pedis: W. A. Akers et al.,J. Am. Acad. Dermatol.21, 686 (1989). Review of pharmacology and clinical efficacy: P. Benfield, S. P. Clissold, Drugs35, 143-153 (1988).
Derivative Type: Nitrate
CAS Registry Number: 61318-91-0
Manufacturers’ Codes: RS-44872
Trademarks: Exelderm (Syntex); Myk (Cassenne); Sulcosyn (Syntex)
Molecular Formula: C18H15Cl3N2S.HNO3
Molecular Weight: 460.76
Percent Composition: C 46.92%, H 3.50%, Cl 23.08%, N 9.12%, S 6.96%, O 10.42%
Properties: Colorless crystals from acetone, mp 130.5-132°.
Melting point: mp 130.5-132°
Therap-Cat: Antifungal.
Keywords: Antifungal (Synthetic); Imidazoles.
Sulconazole (trade name Exelderm) is an antifungal medication of the imidazole class. It is available as a cream or solution to treat skin infections such as athlete’s foot, ringworm, jock itch, and sun fungus.[1][2] Although not used commercially for insect control, sulconazole nitrate exhibits a strong anti-feeding effect on the keratin-digesting Australian carpet beetle larvae Anthrenocerus australis.[3]

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@amcrasto
/////////////////////////////////////////////////////////////////////////////////////////////////////
SYN

DE 2541833 US 4038409
(Read example 5 and 9 in US patent.)
https://patents.google.com/patent/US4038409A/en
EXAMPLE 5Alternative Route to 1-[β-(R-carbonylthio)phenethyl]imidazolesA. Preparation of 1-[2,4-dichloro-β-(methylcarbonylthio)-phenethyl]imidazole, oxalate.1-(β,2,4-Trichlorophenethylimidazole (1.19g) in 5 ml of dry tetrahydrofuran was added to preformed sodium thioacetate, generated in situ from 720 mg thioacetic acid and sodium hydride (480 mg 57% dispersion in mineral oil) in 20 ml. tetrahydrofuran and the mixture stirred and refluxed under nitrogen for 18 hours. The solvent was removed under reduced pressure, water (20 ml) added and the product extracted with ether. The extracts were washed with water, dried (MgSO4), evaporated and the residue chromatographed on silica gel eluting with 10-20% acetone in dichloromethane. The pure product in ether was treated dropwise with ethereal oxalic acid until precipitation was complete, and the thus obtained oxalate salt of 1-[2,4-dichloro-β-(methylcarbonylthio)phenethyl]imidazole recrystallized from acetone/ethyl acetate with mpBy substituting other available sodium thioacids for sodium thioacetate, other compounds of this invention may be prepared.
EXAMPLE 9A. Preparation of 1-[2,4-dichloro-β-(4-chlorobenzylthio)-phenethyl]imidazoleTo a stirred solution of 330 mg sodium hydroxide in 30 ml methanol under nitrogen is added 810 mg of 1-[2,4-dichloro-β-(methylcarbonylthio)phenethyl]imidazole oxalate and the mixture is stirred at room temperature for ca. 30 minutes (until thin layer chromatography shows the disappearance of the ester). α,p-dichlorotoluene (350 mg) is then added, the solution stirred a further 15 minutes and the solvent removed under reduced pressure. Ether and water are then added to the residue and the ether extract washed with water, dried (MgSO4) and concentrated. Dropwise addition of nitric acid (d = 1.42) until precipitation is complete gives the nitrate salt of 1-[2,4-dichloro-β-(4-chlorobenzylthio)phenethyl]imidazole, recrystallized from acetone, mp 130.5°-132° C.B. By using other compounds of this invention exemplified by those set forth in Examples 2 and 4 and other suitable (substituted) hydrocarbyl halides (or mesylates, tosylates), other compounds may be prepared.
SYN
https://www.sciencedirect.com/science/article/pii/S2095177917301405
SYN
Synthesis Path
Substances Referenced in Synthesis Path
| CAS-RN | Formula | Chemical Name | CAS Index Name |
|---|---|---|---|
| 6258-66-8 | C7H7ClS | 4-chlorobenzyl mercaptan | Benzenemethanethiol, 4-chloro- |
| 24155-42-8 | C11H10Cl2N2O | 1-(2,4-dichlorophenyl)-2-(1H-imidazol-1-yl)ethanol | 1H-Imidazole-1-ethanol, α-(2,4-dichlorophenyl)- |
References
- ^ Drugs.com: sulconazole topical
- ^ Fromtling RA (April 1988). “Overview of medically important antifungal azole derivatives”. Clinical Microbiology Reviews. 1 (2): 187–217. doi:10.1128/CMR.1.2.187. PMC 358042. PMID 3069196.
- ^ Sunderland MR, Cruickshank RH, Leighs SJ (2014). “The efficacy of antifungal azole and antiprotozoal compounds in protection of wool from keratin-digesting insect larvae”. Textile Research Journal. 84 (9): 924–931. doi:10.1177/0040517513515312.
| Clinical data | |
|---|---|
| Trade names | Exelderm |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a698018 |
| Routes of administration | Topical |
| ATC code | D01AC09 (WHO) |
| Identifiers | |
| showIUPAC name | |
| CAS Number | 61318-90-9 |
| PubChem CID | 5318 |
| ChemSpider | 5127 |
| UNII | 5D9HAA5Q5S |
| KEGG | D08535 |
| ChEBI | CHEBI:9325 |
| ChEMBL | ChEMBL1221 |
| CompTox Dashboard (EPA) | DTXSID8044129 |
| Chemical and physical data | |
| Formula | C18H15Cl3N2S |
| Molar mass | 397.74 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| showSMILES | |
| showInChI | |
| (what is this?) (verify) |
/////////SULCONAZOLE, сульконазол , سولكونازول , 硫康唑 , Antifungal,

NEW DRUG APPROVALS
ONE TIME
$10.00
TNO 155
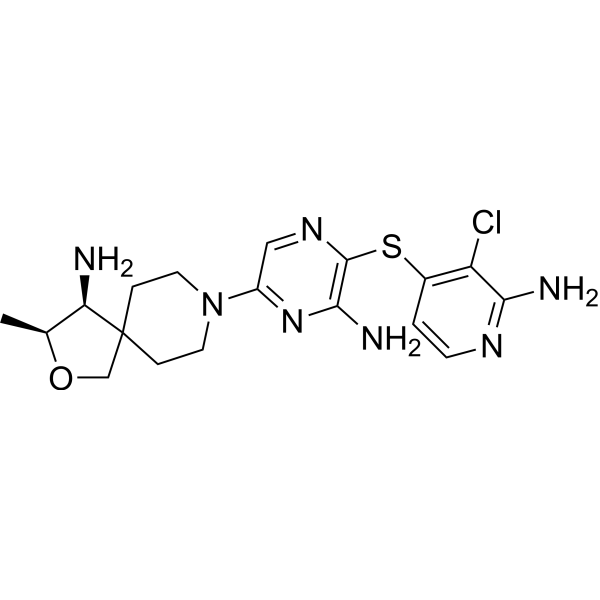
TNO 155
2-Oxa-8-azaspiro[4.5]decan-4-amine, 8-[6-amino-5-[(2-amino-3-chloro-4-pyridinyl)thio]-2-pyrazinyl]-3-methyl-, (3S,4S)-
- (3S,4S)-8-[6-Amino-5-[(2-amino-3-chloro-4-pyridinyl)thio]-2-pyrazinyl]-3-methyl-2-oxa-8-azaspiro[4.5]decan-4-amine
- (3S,4S)-8-(6-amino-5-((2-amino-3-chloropyridin-4-yl)thio)pyrazin-2-yl)-3-methyl-2-oxa-8-azaspiro[4.5]decan-4-amine
| Molecular Weight |
421.95 |
|---|---|
| Formula |
C₁₈H₂₄ClN₇OS |
| CAS No. |
- PTPN11 inhibitor TNO155
- SHP2 inhibitor TNO155
- TNO-155
- TNO155
- UNII-FPJWORQEGI
TNO155 is a potent selective and orally active allosteric inhibitor of wild-type SHP2 (IC50=0.011 µM). TNO155 has the potential for the study of RTK-dependent malignancies, especially advanced solid tumors.
- Originator Novartis
- Developer Mirati Therapeutics; Novartis
- Class Antineoplastics
- Mechanism of ActionProtein tyrosine phosphatase non receptor antagonists
- Phase I/IISolid tumours
- Phase IColorectal cancer
- 11 Jul 2021Phase I trial in Solid tumours is still ongoing in USA, Canada, Japan, South Korea, Netherlands, Singapore, Spain, Taiwan (NCT03114319)
- 04 Jun 2021Efficacy, safety and pharmacokinetics data from phase I trial in Solid tumours presented at 57th Annual Meeting of the American Society of Clinical Oncology (ASCO-2021)
- 08 Jan 2021Novartis plans a phase Ib/II trial for Solid tumours (Combination therapy, Inoperable/Unresectable, Late-stage disease, Metastatic disease, Second-line therapy or greater) in February 2021 (NCT04699188)
CLIP
Combinations with Allosteric SHP2 Inhibitor TNO155 to Block Receptor Tyrosine Kinase Signaling
//////////TNO 155, CANCER
CILENGITIDE
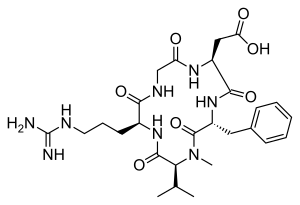
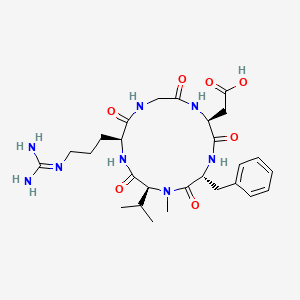
| IUPAC Condensed | cyclo[Arg-Gly-Asp-D-Phe-N(Me)Val] |
|---|---|
| HELM | PEPTIDE1{R.G.D.[dF].[meV]}$PEPTIDE1,PEPTIDE1,5:R2-1:R1$$$ |
| IUPAC | cyclo[L-arginyl-glycyl-L-alpha-aspartyl-D-phenylalanyl-N-methyl-L-valyl] |
CILENGITIDE
- Molecular FormulaC27H40N8O7
- Average mass588.656 Da
2-[(2S,5R,8S,11S)-5-benzyl-11-[3-(diaminomethylideneamino)propyl]-7-methyl-3,6,9,12,15-pentaoxo-8-propan-2-yl-1,4,7,10,13-pentazacyclopentadec-2-yl]acetic acid188968-51-6[RN]
4EDF46E4GI
7823
циленгитид
سيلانجيتيد
西仑吉肽
EMD 121974, EMD-121974, UNII-4EDF46E4GI
Cilengitide has been in phase III clinical trials by Merck Serono and NCI for the treatment of glioblastoma multiforme. However, this research has been discontinued.
Cilengitide was originally developed by Merck KGaA in collaboration with the Technical University of Munich, then received orphan drug designation from FDA for the treatment of glioma in 2005.
Cilengitide (EMD 121974) is a molecule designed and synthesized at the Technical University Munich in collaboration with Merck KGaA in Darmstadt. It is based on the cyclic peptide cyclo(-RGDfV-), which is selective for αv integrins, which are important in angiogenesis (forming new blood vessels), and other aspects of tumor biology. Hence, it is under investigation for the treatment of glioblastoma, where it may act by inhibiting angiogenesis, and influencing tumor invasion and proliferation.[1][2]
The European Medicines Agency has granted cilengitide orphan drug status.[3]
Cilengitide seems to function by inhibiting the FAK/src/AKT pathway and inducing apoptosis in endothelial cells.[4] Preclinical studies in mice of cilengitide were able to demonstrate efficacious tumor regression.[4]
In a rat xenograft model, cilengitide was able to potentiate the cytotoxic effects of radiation when cilengitide was administered prior to radiation therapy.[5] When combined with radiation, inhibition of integrin expression by cilengitide synergistically improves the cytotoxic effects of ionizing radiation for glioblastoma.[5]
Clinical trials
Phase II studies were able to demonstrate that cilengitide as a potential monotherapy in patients with recurrent glioblastoma[6] with high intratumor drug levels when 2000 mg of cilengitide is given twice weekly.[7]
Cilengitide is well tolerated, in combination with radiation and temozolomide, at a dose of 2000 mg in patients with newly diagnosed glioblastoma, regardless of MGMT promoter status.[8] In a phase I/IIa study, the addition of cilengitide to the standard of care for newly diagnosed glioblastoma (surgical resection followed by temozolomide and radiation therapy) improves progression-free survival and overall survival in patients with MGMT promoter methylation.[9]
However, in a subsequent study, cilengitide does not seem to alter the pattern of glioblastoma progression,[10]
and in an EORTC phase III randomized, controlled, multicenter clinical trial, consisting of over 500 patients in 23 countries, the addition of cilengitide to the standard of care did not improve overall survival in patients with newly diagnosed glioblastoma and methylated MGMT promoter status [11] A phase II study, the CORE trial, is currently being conducted in patients with newly diagnosed glioblastoma and unmethylated MGMT promoter status.[12]

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@amcrasto
/////////////////////////////////////////////////////////////////////////////////////////////////////
SYN
Angewandte Chemie, International Edition, 55(4), 1540-1543; 2016

SYN
Chemistry – A European Journal, 16(18), 5385-5390, S5385/1-S5385/36; 2010
Reference:1. WO0047228A1 / US7115261B1.
2. US6001961A.Route 2
Reference:1. CN102731627A.PATENTWO/2021/224234ANTIVIRAL USE OF CILENGITIDEhttps://patentscope.wipo.int/search/en/detail.jsf?docId=WO2021224234&_cid=P20-KW0M52-85135-1
PATENThttps://patents.google.com/patent/CN102731627A/enEMD121974 (Cilengitide), the Chinese another name: ring (L-arginyl glycyl-L-aspartoyl-D-phenylalanyl-N-methyl-L-valyl) is an a kind of new classification cancer therapy drug of synthetic.Merkel company discovers that EMD121974 amalgamation radiotherapy (merging to reach assists TM to add radiotherapy) possibly prolong lifetime; Simultaneously integrate plain supressor antitumor drug as first; Got into the III clinical trial phase, its important mechanism is to grow targeting that the blood supply structure of nutrition, the growth of promotion cancer cell is provided in tumour and for tumour through line artery.The EMD121974 molecular formula is: C 27H 40N 8O 7, have following structure:
The preparation method of cyclic peptide mainly contains liquid phase synthesis process, solid phase synthesis precursor peptide cyclization process, process for solid phase synthesis in liquid phase at present; Wherein preceding two kinds of synthesis techniques all are the cyclisation in liquid phase of synthetic precursor peptide, and this method needs reactant in extremely rare solvent, to react (10 -3~10 -4Mol/L), and intermolecular be prone to react generation line style or cyclic polymer, greatly reduced the cyclisation yield, bring trouble for follow-up purifying, and in large-scale production, produce a large amount of waste liquids, be unfavorable for suitability for industrialized production.In conjunction with the structure of EMD121974, utilize the false rare principle of benefit of solid phase, developed a kind of efficient cyclization reaction, the cyclisation time shortens to 20%~30% of liquid phase cyclisation, and the 2%-8% of solvent as liquid phase used in reaction.Embodiment 1The preparation of Fmoc-L-Asp (OtBu)-Wang ResinThe Wang Resin that takes by weighing the 10g substitution degree and be 0.5mmol/g joins in the reactor drum, adds an amount of DCM, and swelling 30min takes out DCM; 6.17g Fmoc-L-Asp-OtBu, DIC 2.40ml, HOBT2.1g are dissolved among the 30ml DMF; At 0-5 ℃ of activation 15min, activation solution is joined in the reactor drum that contains Wang Resin, behind the reaction 10min; Add DMAP 0.18g again, at 0~30 ℃ of reaction 1~5h.After reaction finishes, add sealing Wang Resin unreacted hydroxylation reagent diacetyl oxide 1ml and pyridine 0.5ml, behind the capping 1h, DMF, DCM, the CH of 80ml used in washing successively 3OH, DMF washing 2,1,1,2 times, each 1min.Through detecting, obtain the Fmoc-L-Asp that substitution degree is 0.47mmol/g (OtBu)-Wang Resin.Embodiment 2The EMD121974 precursor:The preparation of A-Wang Resin (Fmoc-D-Phe-N-Me-L-Val-L-Arg (Mtr)-Gly-L-Asp (OtBu)-Wang Resin)Fmoc-L-Asp (OtBu)-Wang Resin is joined in the reactor drum, behind DMF swelling 30min, take out solvent, the piperidines-DMF that adds 80ml 25% reacts 5min, and 80ml DMF washs 1 time (3min), and the piperidines-DMF that adds 80ml 25% reacts 15min; DMF, DCM, the CH of 80ml used in washing successively 3OH, DMF washing 2,1,1,2 times, each 1min; With 4.45g Fmoc-Gly-OH, 5.68g HBTU, 2.03g HOBt, be dissolved among the DMF of 30ml, dissolve the back and added DIEA 2.45ml; 0~5 ℃ of activation 15min; Activation solution is joined in the above-mentioned reactor drum, and behind reaction 1-3h under 0~30 ℃, reaction end detects with ninhydrin method.Adopt aforesaid method coupling Fmoc-L-Arg (Mtr)-OH, Fmoc-N-Me-L-Val, Fmoc-D-Phe-OH successively, finally obtain Fmoc-D-Phe-N-Me-L-Val-L-Arg (Mtr)-Gly-L-Asp (OtBu)-Wang Resin.Embodiment 3EMD121974 precursor peptide: the preparation of B-Wang Resin (D-Phe-N-Me-L-Val-L-Arg (Mtr)-Gly-L-Asp-Wang Resin)With volume ratio is that piperidines-DMF of 25% is the Fmoc deprotection agent of Fmoc-D-Phe-N-Me-L-Val-L-Arg (Mtr)-Gly-L-Asp (OtBu)-Wang Resin; Add piperidines-DMF 80ml of 25% first time; Reaction 5min, 80ml DMF washs 1 time (3min), adds piperidines-DMF 80ml of 25% for the second time; Behind the reaction 15min, DMF, DCM, the CH of 80ml used in washing successively 3OH, DMF washing 2,1,1,2 times, each 1min gets D-Phe-N-Me-L-Val-L-Arg (Mtr)-Gly-L-Asp (OtBu)-Wang Resin after washing finishes.80% the PhOH-DCM solution that adds volume ratio and be 100ml takes off OtBu with the TFA of catalytic amount, reacts 8h; DMF, DCM, the CH of 80ml used in washing successively 3OH, DMF washing 2,1,1,2 times, each 1min gets D-Phe-N-Me-L-Val-L-Arg (Mtr)-Gly-L-Asp-Wang Resin.Embodiment 4The preparation of EMD121974-Wang Resin (Cyclo (D-Phe-N-Me-L-Val-L-Arg-Gly-L-Asp)-Wang Rsin)In above-mentioned reactor drum, add cyclization reagent 3.9g DPPA, 2.5ml DIEA (reactant cyclization reagent amount of substance ratio is 1: 3), at 10~40 ℃ of reaction 3h, the multiple cyclization reagent reaction 3~5h (reaction end detects with ninhydrin method) that throws once above-mentioned equivalent; DMF, DCM, the CH of 80ml used in washing successively 3OH washing 2,1,3 times, each 3min gets Cyclo (D-Phe-N-Me-L-Val-L-Arg-Gly-L-Asp)-Wang Rsin.Embodiment 5The preparation of EMD121974 (Cyclo (D-Phe-N-Me-L-Val-L-Arg-Gly-L-Asp))In above-mentioned reactor drum, add the TFA/H of lytic reagent 120ml again 2Behind O/TlS (volume ratio is 95: 2.5: 2.5) the reaction 3h, suction filtration is removed resin, and filtrating slowly joins in the no water-ice ether; Static 2-5h, high speed centrifugation obtain thick peptide, prepare through high-pressure liquid phase; Lyophilize gets smart EMD121974; Its purity>99.5%, single impurity<0.2%, total recovery reaches 63%.Choosing substitution degree in the present embodiment is the Wang Resin of 0.5mmol/g, and can also choose substitution degree is the arbitrary Wang Resin and Fmoc-L-Asp-OtBu prepared in reaction Fmoc-L-Asp (the OtBu)-Wang Resin of 0.4~0.9mmol/g scope.All can realize technical scheme of the present invention, and obtain technique effect of the present invention.Above content is an EMD121974 and become one of best preferred version of route; And to further explain that the present invention did; But can not assert that practical implementation of the present invention is only limited to these explanations; Under the prerequisite that does not break away from the present invention’s design, can also make some simple deductions and replacement, all should be regarded as protection domain of the present invention.
CLIPhttps://www.eurekaselect.net/article/2607Cilengitide, a cyclic RGD pentapeptide, is currently in clinical phase III for treatment of glioblastomas and in phase II for several other tumors. This drug is the first anti-angiogenic small molecule targeting the integrins αvβ3, αvβ5 and α5β1. It was developed by us in the early 90s by a novel procedure, the spatial screening. This strategy resulted in c(RGDfV), the first superactive αvβ3 inhibitor (100 to 1000 times increased activity over the linear reference peptides), which in addition exhibited high selectivity against the platelet receptor αIIbβ3. This cyclic peptide was later modified by N-methylation of one peptide bond to yield an even greater antagonistic activity in c(RGDf(NMe)V). This peptide was then dubbed Cilengitide and is currently developed as drug by the company Merck-Serono (Germany). This article describes the chemical development of Cilengitide, the biochemical background of its activity and a short review about the present clinical trials. The positive anti-angiogenic effects in cancer treatment can be further increased by combination with “classical” anti-cancer therapies. Several clinical trials in this direction are under investigation.
CLIPJournal of Protein Chemistry

Schematic of the one-step chemoenzymatic synthesis of cilengitide using wild-type Mcy TE. (1) The chemically synthesised (SPPS, solid-phase peptide synthesis) mimetic substrate was condensed with benzyl mercaptane to produce pentapeptide thioester (pentapeptide-BMT). (2) Models of the substrate-O-TE acyl enzyme intermediate are marked with brackets (protein data bank, 1JMK). (3) Mechanism of TE domain catalysis: a pentapeptide -O-TE acyl-enzyme intermediate is formed by transfer of the peptidyl chain from the phosphopantethiene of the terminal peptidyl carrier protein (PCP), which was substituted by benzyl mercaptane, to the active site serine of the TE domain. For hydrolyzing TE domains, the intermediate is captured by water, generating the linear peptide; for cyclizing TE domains, an intramolecular nucleophile captures the intermediate, resulting in “cilengitide”
PATENTWO 9745447
WO 9745137
DE 19534177
WO 2000053627
WO 2000047228
US 20040063790
WO 2009124754
WO 2011079015
WO 2011069629
WO 2011144756WO 2016059622
PATENTWO 2012062777https://patents.google.com/patent/WO2012062777A1/enSynthesis of cyclic peptidesCyclo[-Arg-Gly-Asp- 6 or 7 -Phe-Val-Ala-] (1 and 2). Resin loading. 2- chlorotrityl chloride-resin ( 1 50 m g , 1 .5m m ol/g ) was p laced i n a 20 m l polypropylene syringe fitted with a polyethylene filter disk. The resin was then washed with CH2CI2 (5 χ 0.5 min), and a solution of Fmoc-L-Gly-OH (334 mg, 1 .125 mmol, 5 equiv) and DIEA (239 μΙ_, 6.25 equiv) in CH2CI2 (2.5 ml_) was added. The mixture was then stirred for 15 min. Extra DIEA (239 μΙ_, total 12.5 mmol) was added, and the mixture was stirred for an additional 45 min. The reaction was stopped by adding 3 χ DCM/ MeOH/ DIEA (85: 10:5) and stirring for 1 0 m in. The Fmoc-L-Gly-O-resin product was subjected to the following washings/treatments with CH2CI2 (3 χ 0.5 min), DMF (3 χ 0.5 min), piperidine and DMF (5 χ 0.5 min). The loading was 0.50 mmol/g, as calculated by Fmoc determination.Peptide coupling. Fmoc-L-Arg(Pbf)-OH (243 mg, 0.375 mmol, 5 equiv), Fmoc- L-Ala-OH (1 17 mg, 0.375 mmol, 5 equiv), Fmoc-L-Val-OH ( 127 mg, 0.375 mmol, 5 equiv) and Fmoc- L-Phe-OH ( 145 mg, 0.375 mmol, 5 equiv) were added sequentially to the above obtained H-L-Gly-O-resin using HCTU (155 mg, 0.375 mmol, 5 equiv), HOBt (50 mg, 0.375 mmol, 5 equiv) and DIEA (127 μΙ_, 0.75 mmol, 10 equiv) in DMF (2.5 ml_). In all cases, after 90 min of coupling, the ninhydrin test was negative. Removal of Fmoc group and washings were performed as described in general procedures. /V-Alloc-thiazole 6 or 7 (92 mg, 0.375 mmol, 5 equiv) was coupled with HATU (143 mg, 0.375 mmol, 5 equiv), HOAt (51 mg, 0.375 mmol, 5 equiv) and DIEA (127 μΙ_, 0.75 mmol, 10 equiv) for 90 min. This coupling was repeated twice in the same conditions. The Alloc group of the peptide resin was removed with Pd (PPh3)4 (9 mg, 0.0075 mmol, 0.1 equiv) in the presence of PhSiH3 (92.5 μΙ_, 0.75 mmol, 10 equiv) in DCM for 20 min. This deprotection was repeated three times in the same conditions. After washing, the resin was treated with dry THF (2ml_) for 15 min. Meanwhile, Fmoc-L-Asp(tBu)-OH (154 mg, 0.375 mmol, 5 equiv) was added to a 68 mM solution of triphosgene in dry THF (1 .15 equiv). Sym-collidine (99.5 μΙ_, 0.75 mmol, 10 equiv) was added to the clear solution, upon which a precipitate of collidinium chloride was formed. DIEA (102 μΙ_, 0.6 mmol, 8 equiv) was added to the resin, immediately followed by addition of the suspension. This coupling was repeated four times in the same conditions. The reaction mixture was stirred at 50 °C during 48 h.Peptide cleavage. Following Fmoc deprotection, the peptidyl-resin was treated with TFA-CH2CI2 (1 :99) (5 χ 30 s). The filtrate was collected on H20 (4 ml_) and the H20 was partially removed under reduced pressure. MeCN was then added to dissolve solid that formed during the removal of H20, and the solution was lyophilized to give 12 mg and 10 mg of the linear compounds 28 and 29 respectively with a purity of > 91 % as checked by HPLC (Column A, Rt 7.43 min and Rt 7.38 min respectively, linear gradient 35%-40% ACN in 15 min.)], which was used without further purification. MALDI-TOF-MS calculated for C50H71 N11 O13S2 1098.29; found mlz 1099.29 [M + H]+, 1 121 .28 [M + Na]+, 1 137.39 [M + K]+.Synthesis in solution. Cyclization. The protected linear peptides 28 and 29 were dissolved in DMF (1 L, 10“4 M), and HOAt (9.6 mg, 0.07 mmol, 5 equiv), DIPEA (24 μΙ_, 0.14 mmol, 10 equiv), and PyAOP (36.6 mg, 0.07 mmol, 5 equiv) were added. The mixture was stirred for 24 h at room temperature, and the course of the cyclization step was then checked by HPLC (Column A, Rt 1 1 -67 min and Rt 10.70 min respectively, linear gradient 45%-55% ACN in 15 min.). The solvent was removed by evaporation under reduced pressure and the protected cycle 30 and 31 were used in the next step without further purification. MALDI-TOF-MS calculated for C50H69N11 O12S2 1080.28; found mlz 1081 .28 [M + H]+, 1 103.27 [M + Na]+, 1 1 19.38 [M + K]+.Side chain deprotection. The protected cyclopeptides 30 and 31 (14.7 mg, 19.04 pmol) were treated with TFA-H20 (95: 5) during 1 h. The solvent was removed by evaporation under reduced pressure.Peptide purification. The crude product was purified by HPLC (Symmetry C8 5 μη-Ί, 30 mm x 100 mm), gradient of MeCN (30% to 75% in 15 min) MeCN (+0.05% TFA) in water (+0.05% TFA), 20 mL/min, detection at 220 nm, to give the cyclopeptides 1 and 2 (4.5 mg, 5.8 pmol and 6.5 mg, 8.37 pmol, 7.7% and 12% yield respectively). The products were characterized by HPLC (Rt 8.99 min, and Rt 8.02 min Column A, respectively, linear gradient 0%-100% ACN in 1 5 min. ) and by MALDI-TOF-MS: calculated for C33H45N11 O9S 771 .84; found mlz 772.84 [M + H]+, 794.83 [M + Na]+, 810.94 [M + K]+.Cyc/o-[Arg-Gly-Asp-Thz1X-] (3). General procedure for cyclopeptide synthesis. Solid phase synthesis: The synthesis of the linear peptide H- Asp(tBu)-XX-Arg(Pbf)-Gly-OH was performed using Fmoc-based solid phase peptide synthesis with 2-chlorotrityl chloride resin (2.0 g, 3.2 mmol).Resin loading: Fmoc-Gly-OH (594 mg, 2.0 mmol) was attached to the resin with DIPEA in DCM at room temperature for 1 .5 h. The remaining trityl groups were capped adding 0.5 mL of MeOH for 30 min. After that, the resin was filtered and washed with DCM (2x), DMF (2x). The loading of the resin was determined by titration of the Fmoc group (Chan WC and White PD. Fmoc Solid Phase Peptide Synthesis. Oxford University Press: New York, 2000). The final loading was 2.0 mmol/g. The Fmoc group was eliminated by treatment with 20% piperidine in DMF (2X10 min). The resin was washed with DMF (3x), DCM (3x). Peptide coupling: Fmoc-Arg(Pbf)-OH (5.19 g, 8.0 mmol), DIPCDI (1.23 mL, 8.0 mmol) and HOBt (1.08 g, 8.0 mmol) were dissolved in DMF and added to the resin for 1 .5 h. The end of the coupling was monitored by ninhydrin test (free amine group) (Kaiser E et al. Anal Biochem 1970, 34:595-598). The resin was filtered and washed with DMF (3X) and DCM (3X). The Fmoc group was eliminated with 20 % piperidine in DMF (2X10 min).The coupling of the thiazole module was carried out with 8 (1 .14 g, 3.0 mmol), PyAOP (1 .56 g, 3.0 mmol) and DIPEA (1 .02 mL, 6.0 mmol) in DMF for 1 .5 h. The completion of the reaction was checked with the ninhydrin test. Finally the deprotection of the amine and coupling of the Fmoc-Asp(‘Bu)-OH were carried out under the same conditions of the second amino acid.Peptide cleavage: The resin bound peptide was treated with 2% TFA in DCM (6 x 30 sec.) The resin was washed with DCM and the combined solution was evaporated under vacuum with Et20 several times, furnishing the linear peptide 32 as a white solid. The peptide was used for the next step without purification.H PLC (gradient 20 to 80% of CH3CN in 1 5 m in): tR= 8.33 min. HPLC-MS (ES(+)): m/z 795.3.Synthesis in solution. Cyclization: The product 32 (200 mg, 0.251 mmol) was dissolved in anhydrous DMF (50 mL, 5 mM), PyAOP (262 mg, 0.503 mmol) and DIPEA (213 μί, 1 .255 mmol) were added. The reaction was monitored by HPLC. Once the reaction was finished, the DMF was evaporated under vacuum. The crude was dissolved in AcOEt and the solution was washed with NH4CISat and Na2CO3 sat. The organic layer was collected, dried over Na2SO4, filtered and concentrated under vacuum. The peptide was purified by flash chromatography (CHCIs/MeOH 8:2) furnishing the protected cyclic peptide 33 as a white solid (1 56 mg, XX%). HPLC (gradient 40 to 90% of CH3CN in 1 5 min): tR= 8.86 min. HPLC-MS (ES(+)): m/z 778.2Side chain deprotection: The protected peptide 33 (125 mg, XX mmol), was treated with 25 mL of a solution of TFA H2O (95:5). After 3 h, the solvent was evaporated under vacuum and the residue was precipitated with Et2O (4X). The Et2O solution was discarded and the white solid was lyophilized to afford 3 55 mg (XX%).
Peptide purification. The end product 3 was dissolved in 5 ml MilliQ water and it was filtered through a 0.2 pm filter. The cyclic peptide was purified by semipreparative RP-HPLC using acetronitrile (0.05% TFA)/water (0.1 % TFA). The HPLC sample was vacuum concentred and transformed into the hydrochloride salt lyophilized with water with 0.05% HCI.1H-NMR (500 MHz, H20:D20-d2 9: 1 , 278 K): δ = 9.29 (t, NH Gly), 9.20 (d, J = 7.24 Hz, NH Asp), 8.90 (t, J = 5.89/5.89 Hz, NH Thz), 8.46 (d, J = 8.93 Hz, NH Arg), 7.79 (s, CH Thz), 7.22 (t, J = 5.39/5.39 Hz, ΝΗε Arg), 4.75 (m, CHa Arg), 4.63 (m, CHa Asp), 4.04 (dd, J = 3.35/14.90 Hz, CHa Gly), 3.82 (dd, J = 6.69/14.96 Hz, CHa Gly), 3.17 (m, CH25 Arg), 2.89 (m, CH2p Asp), 1 .92 (m, CH p Arg), 1 .82 (m, CHP Arg), 1 .63 (m, CH2 Arg). HPLC (gradient 0 to 20% of CH3CN in 15 min): tR= 10.52 m in. HRMS (E IS) m/z calculated 468.1540

found 469.16099 (M+H)+.Cyc/o-[Arg-Gly-Asp-Thz2X-] (4). The cyclopeptide 4 was prepared according to the process followed for 3 and using bithiazole 9 (XX mg, YY mmol) instead of 8. The linear peptide 34: HPLC (gradient 0 to 100% CH3CN in 15 min.): tR = 10.34 min, HPLC-MS (ES(+)): m/z 877.81 . The protected peptide 35: HPLC (gradient 0 to 100% CH3CN in 15 min.): tR = 13.91 min, HPLC-MS (ES(+)): m/z 860.54. The final peptide 4: 1H-NMR (500 MHz, H20:D20-d2 9: 1 , 298 K): δ = 8.93 (sbroad, NH Gly), 8.82 (d, J = 7.62 Hz, NH Asp), 8.75 (t, J = 5.69/5.69 Hz, NH Thz), 8.51 (d, J = 7.62 Hz, NH Arg), 8.05 (s, CH Thz1), 7.50 (s, CH Thz2), 7.19 (t, J = 5.38/5.38 Hz, ΝΗε Arg), 4.13 (dd, J = 5.82/14.24 Hz, CH Gly), 3.87 (dd, J = 5.96/15.69 Hz, CH Gly), 3.21 (m , CH25 Arg), 2.94 (m, CH2p Asp), 1 .95 (m , CHP Arg), 1 .87 (m , CHP Arg), 1 .68 (m , CH2y Arg). HPLC (gradient 1 0 to 25% of CH3CN in 1 5 m in): tR = 8.73 min. HRMS (EIS) m/z calculated 551 .1369 (C2oH25N906S2) found 552.14392 (2M+2H)+.Cyc/o-[Arg-Gly-Asp-Thz3X-] (5). The cyclopeptide 5 was prepared according to the process for 3 and using trithiazole 10 (XX mg, YY mmol) instead of 8. The linear peptide 36: HPLC (gradient 20 to 80% of CH3CN in 15 min.): tR = 7.60 min, HPLC-MS (ES(+)): m/z 961 .23. The protected peptide 37: HPLC (gradient 20 to 80% of CH3CN in 15 m in. ): tR = 1 3.13 min, HPLC-MS (ES(+)): m/z 944.3. The final peptide 5: HPLC (gradient 10 to 30% CH3CN in 15 m in): tR = 8.26 m in. HRMS (E IS) m/z calculated 634.1 1 99 (C23H26N10O6S3) found 635.12683 (2M+2H)+. 1H-NMR (500 MHz, DMSO-d6 298 K): δ = 9.21 (t, J = 5.4, NH Gly), 8.72 (m, NH Asp + NH Thz), 8.37 (s, CH Thz1), 7.96 (d, J = 9.2, NHa Arg), 7.77 (s, CH Thz2), 7.68 (t, J = 6.0, ΝΗε Arg), 7.23 (s, CH Thz3), 4.83 (dd, J = 14.3, 8.5, CHa Arg), 4.72 (dd, J = 16.3, 6.6, CH Thz), 4.59 (m, CH Thz + CHa Asp), 3.89 (d, J = 1 1 .5, CH Gly), 3.59 (d, J = 9.7, CH Gly), 3.13 (dd, J = 12.6, 6.3, CH25 Arg), 2.81 (dd, J = 16.3, 4.3, CHP Asp), 2.58 (dd, J = 16.5, 8.7, CHP Asp), 1 .82 (m, CHP Arg), 1 .71 (m, CHP Arg), 1 .49 (m, CH2y Arg).Cilengitide. The cilengitide was prepared according to the method described in Dechantsreiter MA et al. (J Med Chem 1999, 42:3033-3040). 1H- NMR (500 MHz, H20:D20-d2 9: 1 , 298 K): δ = 8.55 (d, J = 8.06 Hz, NH Asp), 8.37 (d, J = 7.28 Hz, NH Arg), 8.13 ( d, J = 9.19 Hz, NH Phe), 7.97 (m, NH Gly), 7.34 (m, 2H, C6H5 Phe), 7.26 (m, 3H, C6H5 Phe), 7.22 (t, J = 5.53/5.53 Hz, ΝΗε Arg), 5.19 (dd, J = 8.58/16.02 Hz, CHa Phe), 4.56 (dd, J = 7.45/- Hz, CHa Asp), 4.34 (d, J = 10.89 Hz, CHa MeVal), 4.12 (dd, J = 7.80/14.63 Hz, CH Gly), 3.95 (dd, J = 6.84/15.33 Hz, CHa Arg), 3.54 (dd, J = 3.37/14.60 Hz, CH Gly), 3.20 (m , CH25 Arg), 3.02 (m, CH2p Phe), 2.88 (s, CH3 MeVal), 2.84 (dd, J = 7.26/16.68 Hz, CHP Asp), 2.63 (dd, J = 7.60/16.54 Hz, CHP Asp), 2.06 (m, CHP Val), 1 .91 (m, CH2p Arg), 1 .57 (m, CH2 Asp), 0.88 (d, J = 6.55 Hz, CH3 Val1), 0.56 (d, J = 6.49 Hz, CH3 Val2).
PAPERJournal of medicinal chemistry (1999), 42(16), 3033-40.Peptide Science (2001), Volume Date2000, 37th, 249-250. Current opinion in investigational drugs (London, England : 2000) (2003), 4(6), 741-5. Journal of medicinal chemistry (2005), 48(24), 7675-87.Peptide Science (2006), 43rd, 215-216Angewandte Chemie, International Edition (2010), 49(15), 2732-2737, S2732/1-S2732/53.Accounts of Chemical Research (2017), 50(7), 1541-1556.
References
- ^ Burke PA, DeNardo SJ, Miers LA, Lamborn KR, Matzku S, DeNardo GL (August 2002). “Cilengitide targeting of alpha(v)beta(3) integrin receptor synergizes with radioimmunotherapy to increase efficacy and apoptosis in breast cancer xenografts”. Cancer Research. 62 (15): 4263–72. PMID 12154028.
- ^ Goodman SL, Hölzemann G, Sulyok GA, Kessler H (February 2002). “Nanomolar small molecule inhibitors for alphav(beta)6, alphav(beta)5, and alphav(beta)3 integrins”. Journal of Medicinal Chemistry. 45 (5): 1045–51. doi:10.1021/jm0102598. PMID 11855984.
- ^ Spreitzer H (October 27, 2008). “Neue Wirkstoffe – Cilengitide”. Österreichische Apothekerzeitung (in German) (22/2008): 1136–7.
- ^ Jump up to:a b Yamada S, Bu XY, Khankaldyyan V, Gonzales-Gomez I, McComb JG, Laug WE (December 2006). “Effect of the angiogenesis inhibitor Cilengitide (EMD 121974) on glioblastoma growth in nude mice”. Neurosurgery. 59 (6): 1304–12, discussion 1312. doi:10.1227/01.NEU.0000245622.70344.BE. PMID 17277694. S2CID 19861713.
- ^ Jump up to:a b Mikkelsen T, Brodie C, Finniss S, Berens ME, Rennert JL, Nelson K, Lemke N, Brown SL, Hahn D, Neuteboom B, Goodman SL (June 2009). “Radiation sensitization of glioblastoma by cilengitide has unanticipated schedule-dependency”. International Journal of Cancer. 124 (11): 2719–27. doi:10.1002/ijc.24240. PMID 19199360.
- ^ Reardon DA, Fink KL, Mikkelsen T, Cloughesy TF, O’Neill A, Plotkin S, et al. (December 2008). “Randomized phase II study of cilengitide, an integrin-targeting arginine-glycine-aspartic acid peptide, in recurrent glioblastoma multiforme”. Journal of Clinical Oncology. 26 (34): 5610–7. CiteSeerX 10.1.1.688.8987. doi:10.1200/JCO.2008.16.7510. PMID 18981465.
- ^ Gilbert MR, Kuhn J, Lamborn KR, Lieberman F, Wen PY, Mehta M, Cloughesy T, Lassman AB, Deangelis LM, Chang S, Prados M (January 2012). “Cilengitide in patients with recurrent glioblastoma: the results of NABTC 03-02, a phase II trial with measures of treatment delivery”. Journal of Neuro-Oncology. 106 (1): 147–53. doi:10.1007/s11060-011-0650-1. PMC 4351869. PMID 21739168.
- ^ Nabors LB, Mikkelsen T, Hegi ME, Ye X, Batchelor T, Lesser G, Peereboom D, Rosenfeld MR, Olsen J, Brem S, Fisher JD, Grossman SA (November 2012). “A safety run-in and randomized phase 2 study of cilengitide combined with chemoradiation for newly diagnosed glioblastoma (NABTT 0306)”. Cancer. 118 (22): 5601–7. doi:10.1002/cncr.27585. PMC 3423527. PMID 22517399.
- ^ Stupp R, Hegi ME, Neyns B, Goldbrunner R, Schlegel U, Clement PM, et al. (June 2010). “Phase I/IIa study of cilengitide and temozolomide with concomitant radiotherapy followed by cilengitide and temozolomide maintenance therapy in patients with newly diagnosed glioblastoma” (PDF). Journal of Clinical Oncology. 28(16): 2712–8. doi:10.1200/JCO.2009.26.6650. PMID 20439646.
- ^ Eisele G, Wick A, Eisele AC, Clément PM, Tonn J, Tabatabai G, et al. (March 2014). “Cilengitide treatment of newly diagnosed glioblastoma patients does not alter patterns of progression”(PDF). Journal of Neuro-Oncology. 117 (1): 141–5. doi:10.1007/s11060-014-1365-x. PMID 24442484. S2CID 21636884.
- ^ Merck Group. “Phase III Trial of Cilengitide Did Not Meet Primary Endpoint in Patients With Newly Diagnosed Glioblastoma, Date accessed: 3/24/2014.”
- ^ ASCO Meeting Library. [1] “Cilengitide combined with standard treatment for patients with newly diagnosed glioblastoma and methylated O6-methylguanine-DNA methyltransferase (MGMT) gene promoter: Key results of the multicenter, randomized, open-label, controlled, phase III CENTRIC study, Date accessed: 3/24/2014
| Names | |
|---|---|
| IUPAC name2-[(2S,5R,8S,11S)-5-benzyl-11-{3-[(diaminomethylidene)amino]propyl}-7-methyl-3,6,9,12,15-pentaoxo-8-(propan-2-yl)-1,4,7,10,13-pentaazacyclopentadecan-2-yl]acetic acid | |
| Identifiers | |
| CAS Number | 188968-51-6 |
| 3D model (JSmol) | Interactive image |
| ChEMBL | ChEMBL429876 |
| ChemSpider | 154046 |
| IUPHAR/BPS | 6597 |
| KEGG | D03497 |
| MeSH | Cilengitide |
| PubChem CID | 176873 |
| UNII | 4EDF46E4GI |
| CompTox Dashboard (EPA) | DTXSID9044035 |
| showInChI | |
| showSMILES | |
| Properties | |
| Chemical formula | C27H40N8O7 |
| Molar mass | 588.656 g/mol |
| Density | 1.417 g/mL |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references |
/////////CILENGITIDE, циленгитид , سيلانجيتيد ,西仑吉肽 , PHASE 3, EMD 121974, EMD-121974, UNII-4EDF46E4GI, orphan drug , MERCK, glioblastoma,
CC(C)C1C(=O)NC(C(=O)NCC(=O)NC(C(=O)NC(C(=O)N1C)CC2=CC=CC=C2)CC(=O)O)CCCN=C(N)N

NEW DRUG APPROVALS
ONE TIME
$10.00
Methotripremazine
Methotripremazine
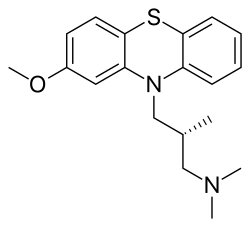
- CL 36467
- CL 39743
- N05AA02
- RP 7044
- RP-7044
- SK&F 5116
- XP-03
- XP03
Product Ingredients
| INGREDIENT | UNII | CAS | INCHI KEY |
|---|---|---|---|
| Methotrimeprazine hydrochloride | 42BB1Y2586 | 1236-99-3 | ODLGFPIWRAEFAN-PFEQFJNWSA-N |
| Methotrimeprazine maleate | 5KN5Y9V01K | 7104-38-3 | IFLZPECPTYCEBR-VIEYUMQNSA-N |
Methotrimeprazine
CAS Registry Number: 60-99-1
CAS Name: (bR)-2-Methoxy-N,N,b-trimethyl-10H-phenothiazine-10-propanamine
Additional Names: (-)-10-(3-dimethylamino-2-methylpropyl)-2-methoxyphenothiazine; levomepromazine; 2-methoxytrimeprazine; levomeprazine
Manufacturers’ Codes: RP-7044
Trademarks: Sinogan-Debil; Tisercin (EGYT); Neozine (Rh>e-Poulenc); Nirvan; Nozinan (Rh>e-Poulenc); Levoprome (Lederle)
Molecular Formula: C19H24N2OS
Molecular Weight: 328.47
Percent Composition: C 69.47%, H 7.36%, N 8.53%, O 4.87%, S 9.76%
Literature References: Prepn: Courvoisier et al.,C.R. Seances Soc. Biol. Ses Fil.151, 1378 (1957); Jacob, Robert, US2837518 (1958 to Rhône-Poulenc).Optical Rotatory Power, -17, Conc: 5 g/100mL; Solv: chloroform; Wavlen: 589.3 nm; Temp: 20 °C
Derivative Type: Maleate
CAS Registry Number: 7104-38-3
Trademarks: Minozinan; Milezin (Spofa); Neuractil; Neurocil (Bayer); Sofmin (Dainippon); Veractil
Molecular Formula: C19H24N2OS.C4H4O4
Molecular Weight: 444.54
Percent Composition: C 62.14%, H 6.35%, N 6.30%, O 18.00%, S 7.21%
Properties: Crystals, darkened by light. Dec about 190°. Sparingly sol in water (0.3% at 20°) and in ethanol (0.4%). pH of a 0.3% aq soln is 4.3. The free base is levorotatory: [a]D20 -17° (c = 5 in chloroform).
Optical Rotation: [a]D20 -17° (c = 5 in chloroform)
Therap-Cat: Analgesic.
Keywords: Analgesic (Non-Narcotic).

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@amcrasto
/////////////////////////////////////////////////////////////////////////////////////////////////////
Methotrimeprazine is a phenothiazine used in the management of psychosis, particular those of schizophrenia, and manic phases of bipolar disorder.
A phenothiazine with pharmacological activity similar to that of both chlorpromazine and promethazine. It has the histamine-antagonist properties of the antihistamines together with central nervous system effects resembling those of chlorpromazine. (From Martindale, The Extra Pharmacopoeia, 30th ed, p604)
Levomepromazine, also known as methotrimeprazine, is a phenothiazine neuroleptic drug. Brand names include Nozinan, Levoprome, Detenler, Hirnamin, Levotomin and Neurocil. It is a low-potency antipsychotic (approximately half as potent as chlorpromazine) with strong analgesic, hypnotic and antiemetic properties that are primarily used in palliative care.[1][2]
Serious side effects include tardive dyskinesia, akathisia, abnormalities in the electrical cycle of the heart, low blood pressure and the potentially fatal neuroleptic malignant syndrome.[1][2]
As is typical of phenothiazine antipsychotics, levomepromazine is a “dirty drug“, that is, it exerts its effects by blocking a variety of receptors, including adrenergic receptors, dopamine receptors, histamine receptors, muscarinic acetylcholine receptors and serotonin receptors.[1][2]
Medical uses
It can be used as an analgesic for moderate to severe pain in non-ambulant patients (the latter being because of its strong sedative effects).[3]
Levomepromazine is also used at lower doses for the treatment of nausea and insomnia.[1]
Levomepromazine is frequently prescribed and valued worldwide in palliative care medicine for its multimodal action, to treat intractable nausea or vomiting, and for severe delirium/agitation in the last days of life. Palliative care physicians will commonly prescribe it orally or via subcutaneous syringe drivers in combination with opioid analgesics such as hydromorphone.[1][2]
Levomepromazine is used for the treatment of psychosis, particularly those of schizophrenia, and manic phases of bipolar disorder. It should only be used with caution in the treatment of agitated depressions, as it can cause akathisia as a side effect, which could worsen the agitation.[1][2] A 2010 systematic review compared the efficacy of levomepromazine with atypical antipsychotic drugs:
Adverse effects
The most common side effect is akathisia.[2] Levomepromazine has prominent sedative and anticholinergic/sympatholytic effects (dry mouth, hypotension, sinus tachycardia, night sweats) and may cause weight gain.[2] These side effects normally preclude prescribing the drug in doses needed for full remission of schizophrenia, so it has to be combined with a more potent antipsychotic.[2] In any case, blood pressure and EKG should be monitored regularly.[2]
A rare but life-threatening side effect is neuroleptic malignant syndrome (NMS).[2] The symptoms of NMS include muscle stiffness, convulsions and fever.[2]
PAPER
Bulletin de la Societe de Pharmacie de Bordeaux (1964), 103(4), 224-30.
The authors define an extn. equil. const., pKe. When a basic mol., A, in an org. solvent (immiscible with water) is shaken with an aq. acid, part of A passes into the aq. phase in the equil. A + H+ .rdblhar. AH+, and Ke and pKe are defined by the equations Ke = [A]org[H+]H2O/[AH+]H2O and pKe = pKa -log ([A]org/[A]H2O), resp. Values of pKe are reported for levomepromazine, properidiazine, thioridazine, chlorpromazine, alimenazine, propiomazine, promethazine, and aminopromazine. Where 2 C atoms sep. the 2 N chain atoms, pKe is of the order of 5, and if 3, the value is near 4.3.
PATENT
JP 40009030
A soln. of 10.5 g. l-3-dimethylamino-2-methylpropanol in xylene is added a suspension of 2.5 g. Na in xylene and a soln. of 18 g. p-tosyl chloride in xylene is dropped in to give l-3-dimethylamino-2-methylpropanol tosylate (I), hydrochloride m. 98-100%. I is treated with 18 g. 2-methoxyphenothiazine and NaNH2 (prepd. from 1.85 g. Na) to give 80% l-3-(2-methoxy-10-phenothiazinyl)-2-methyl-1-dimethylaminopropane, m. 125-6° (hexane). Similarly are prepd. l-3-(3-ethyl-10-phenothiazinyl)-2-methyl-1-dimethylaminopropane (maleate m. 136°) and l-3-(10-phenothiazinyl)-2-methyl-1-dimethylaminopropane (maleate m. 174-5°). The products are tranquilizers.
PATENT
HU 152208
HU 157158
PL 66636
PAPER
Bulletin de la Societe Chimique de France (1968), (8), 3220-2.
Folia medica (1970), 12(1), 88-9
Journal of pharmaceutical sciences (1987), 76(7), 541-4.
SYN
| IN201203390 |
Deprotonation of 2-methoxyphenothiazine by means of KOH in refluxing touene/DMSO, followed by condensation of resulting pottasium salt with N-(3-chloro-2-methylpropyl)-N,N-dimethylamine in refluxing toluene leads to racemic levomepromazine , which upon finally resolution using (-)-dibenzoyl-L-tartaric acid in acetone or using di-p-toluoyl-L-tartaric acid and, optionally, HCOOH in EtOH at 60 °C affords the target levomepromazine

SYN

References
- ^ Jump up to:a b c d e f Brayfield A, ed. (13 December 2013). “Levomepromazine”. Martindale: The Complete Drug Reference. London, UK: Pharmaceutical Press. Retrieved 12 May 2014.
- ^ Jump up to:a b c d e f g h i j k Joint Formulary Committee (2013). British National Formulary (BNF) (65 ed.). London, UK: Pharmaceutical Press. ISBN 978-0-85711-084-8.
- ^ “Levomepromazine”. Farmacotherapeutisch Kompas (in Dutch). Retrieved 5 October 2016.
- ^ Jump up to:a b Sivaraman P, Rattehalli RD, Jayaram MB (October 2010). “Levomepromazine for schizophrenia”. The Cochrane Database of Systematic Reviews. 10 (10): CD007779. doi:10.1002/14651858.CD007779.pub2. PMC 3283151. PMID 20927765.
External links
- “Levomepromazine”. PubChem. National Center for Biotechnology Information.
- NOZINAN – Lévomépromazine Doctissimo Guides des Medicaments
- “Levomepromazine” (PDF). Grampians Palliative Care Team Publication. Victoria, Australia. May 2010. Archived from the original (PDF) on 2011-02-26.
- “Levomepromazine in Palliative Care” (PDF). Scotland, UK. August 2013. Archived from the original (PDF) on 2013-05-22.
| Clinical data | |
|---|---|
| AHFS/Drugs.com | Micromedex Detailed Consumer Information |
| Pregnancy category | Only if clearly needed |
| Routes of administration | Oral, seldom IM |
| Drug class | Typical antipsychotic |
| ATC code | N05AA02 (WHO) |
| Legal status | |
| Legal status | AU: S4 (Prescription only)UK: POM (Prescription only) |
| Pharmacokinetic data | |
| Bioavailability | ~50–60% |
| Metabolism | Hepatic |
| Elimination half-life | ~20 hours |
| Excretion | In feces and urine (metabolites), unchanged drug only 1% |
| Identifiers | |
| showIUPAC name | |
| CAS Number | 60-99-1 7104-38-3 (maleate), 1236-99-3 HCl) |
| PubChem CID | 72287 |
| IUPHAR/BPS | 7603 |
| DrugBank | DB01403 |
| ChemSpider | 65239 |
| UNII | 9G0LAW7ATQ |
| KEGG | D00403 |
| ChEBI | CHEBI:6838 |
| ChEMBL | ChEMBL1764 |
| CompTox Dashboard (EPA) | DTXSID1023289 |
| ECHA InfoCard | 100.000.450 |
| Chemical and physical data | |
| Formula | C19H24N2OS |
| Molar mass | 328.47 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| showSMILES | |
| showInChI | |
| (what is this?) (verify) |
///////////methotripremazine, L 36467, CL 39743, N05AA02, RP 7044, RP-7044, SK&F 5116, XP-03, XP03
O(c2cc1N(c3c(Sc1cc2)cccc3)C[C@H](C)CN(C)C)C

NEW DRUG APPROVALS
one time
$10.00
Methiomeprazine
Methiomeprazine
N,N,2-trimethyl-3-(2-methylsulfanylphenothiazin-10-yl)propan-1-amine
CAS 7009-43-0
Molecular Formula, C19-H24-N2-S2, Molecular Weight, 344.5446,
- 10H-Phenothiazine-10-propanamine, N,N,β-trimethyl-2-(methylthio)-, (±)-
- Phenothiazine, 10-[3-(dimethylamino)-2-methylpropyl]-2-(methylthio)-, (±)- (8CI)
- N,N,β-Trimethyl-2-(methylthio)-10H-phenothiazine-10-propanamine
- (±)-10-(3-Dimethylamino-2-methylpropyl)-2-(methylthio)phenothiazine
- 10584-RP
- 2-Methylthio-10-(2-methyl-3-dimethylaminopropyl)phenothiazine
- Methiomeprazine
- SKF 6270
- (+-)-10-(3-Dimethylamino-2-methylpropyl)-2-(methylthio)phenothiazine
- Phenothiazine, 10-(3-(dimethylamino)-2-methylpropyl)-2-(methylthio)-, (+-)-
- 10584 RP
- EINECS 230-285-9
- Methiomeprazinum
- Methiomeprazinum [INN-Latin]
- Metiomeprazina
- Metiomeprazina [INN-Spanish]
- RP 10584
- SKF 6270
- UNII-X2R9QTF0OL
Methiomeprazine hydrochloride
14056-64-5

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@amcrasto
///////////////////////////////////////////////////////////////////////////////////////////////////// Methiomeprazine is an antiemetic drug.
PATENTFR 2705 M 19640831.The title compd. and its derivs. are prepd. and can be used in the prepn. of antiemetic compns. A soln. of 2.280 g. 3-methylthio-10-(3-dimethylamino-2-methylpropyl)phenothiazine (I) in 12 l. EtOH is heated to 70° and added to a soln. (60°) of 969 g. d-tartaric acid in 27 l. EtOH, the soln. kept overnight and filtered, and the mother liquors from the 1st and 2nd crystns. combined and evapd. The residue (2.352 g.) is dissolved in H2O, the soln. made alk. with 700 ml. NaOH (d. 1.33) and extd. with 4 l. CH2Cl2, the org. phase sepd., the aq. phase extd. with 1 l. CH2Cl2, and the exts. combined and evapd. at ∼20 mm. The residue (1.183 g.) is taken up in 7 l. EtOH at 60°, the soln. added to 370 g. maleic acid in 1.7 l. EtOH (60°), and the mixt. kept overnight to give 1.192 g. I acid maleate (II), m. 176-7° (EtOH), [α]24D -21.2° ± 1.5° (c 2, CHCl3). II (300 g.) is added to a mixt. of 1 l. H2O and 2 l. CH2Cl2, 150 ml. NaOH (d. 1.33) added, and the org. phase sepd. and distd. to give 185 g. (-)-3-methylthio-10-(3-dimethylamino-2-methylpropyl)phenothiazine (III), m. 84-5° (iso-PrOH), [α]23D -45° ± 3° (c 2.5, C6H6).
PAPERJournal of Organic Chemistry (1960), 25, 944-7.https://pubs.acs.org/doi/abs/10.1021/jo01076a019cf. CA 54, 15391b. The prepn. of various 10-aminoalkyl derivs. of the following phenothiazines was described: 2-hydroxyphenothiazine (I), 2-methylthiophenothiazine (II), 2-methylsulfonylphenothiazine (III), 2-trifluoromethylsulfonylphenothiazine (IV), 2-trifluoromethylthiophenothiazine (V), 2-azaphenothiazine (VI), and 8-chloro-2-azaphenothiazine (VII). The direct alkylation of I was not attempted. Instead, 2-benzoyloxyphenothiazine was alkylated with NaNH2 in xylene and the ester group removed by basic hydrolysis during the workup. The alkylation of IV with 3-(4-methylpiperazinyl)propyl chloride required 48 hrs. VI (15 g.), 6.8 g. NaNH2, and 500 ml. PhMe refluxed 45 min. under N, treated with 21 g. 3-chloro-1-(1-formyl-4-piperazinyl)propane-HCl and 300 ml. PhMe, the mixt. cooled, 150 ml. H2O added, the PhMe layer extd. with dil. HCl, the acid exts. made alk., extd. with C6H6, and the solvent evapd. gave 21 g. oil. The oil dissolved in 250 ml. alc., 60 ml. H2O and 7 ml. 40% NaOH, the mixt. refluxed 2 hrs., the solvents removed, the residual oil dissolved in C6H6, the soln. extd. with HCl, made alk., extd. with C6H6, and the whole distd. gave 11 g. 10-[3-(1-piperazinyl)propyl]-2-azaphenothiazine. The distd. material was dissolved in 250 ml. MeOH and refluxed 1.5 hrs. with 1.8 g. ethylene oxide, the solvent evapd., the residue dissolved in 250 ml. C6H6, the soln. azeotropically distd. during 1 hr., cooled, and refluxed 1 hr. with 6.5 g. AcCl, the solvents evapd., the gum treated with 10% NaOH, and the C6H6 evapd. gave 4.3 g. 4-[3-(2-azaphenothiazin-10-yl)propyl]-1-piperazineëthanol; acetate dimaleate m. 147-8° (decompn.) (EtOAc). 1-Piperazinepropanol (57.6 g.) refluxed 1 hr. with 48 g. HCO2Me, the excess HCO2Me removed, and the residue distd. gave 65.3 g. oil, b1.1 174.5-7.0°, n24D 1.5072. This oil (42.8 g.) in 300 cc. CHCl3 treated with excess HCl, then 19 g. SOCl2, the mixt. refluxed 0.5 hr., 3 g. SOCl2 added, refluxing continued 2.5 hrs., and the solvents removed gave a cryst. HCl salt. Conversion of this to the free base gave 60% 1-formyl-4-(3-chloropropyl)piperazine, yellow oil, b0.4 144.5-8.5°, n25D 1.5053. By starting with I-VII the following 2,10-disubstituted phenothiazines were obtained (substituents at 2, 10, b.p./mm., and % yield given); SMe, (CH2)3NMe2, 220-3°/0.7 (HCl salt m. 149-50°), 88; SMe, CH2CHMeCH2NMe2, 218-21°/0.1 (HCl salt m. 173-4°), 93; SMe, (CH2)3N.(CH2)2.NMe.CH2.CH2, 239-42°/0.1 (di-HCl salt m. 224-5°), 92; SMe, CH2CHMeCH2N.(CH2)2. NMe.CH2.CH2, 200-20°/0.03 (dimaleate m. 174-5°), 44; SMe, (CH2)3N.(CH2)2.N[(CH2)2OAc].CH2.CH2 – (dimaleate m. 165-6°), 33; SO2Me, (CH2)3NMe2, 115-16° (HCl salt m. 112-15°), 62; SO2Me, CH2CHMeCH2NMe2, 255-60°/0.2 (HCl salt m. 234-5°), 60; SCF3, (CH2)3NMe2, 153-7°/0.1, 64; SCF3, CH2CHMeCH2NMe2, 153-7°/0.1 (picrate m. 158.5-9.5°), 54; SCF3, I (CH2)3N.(CH2)2.NMe.CH2.CH2, 220-3°/0.3 (dimaleate m. 182-3°), 63; SO2CF3, (CH2)3NMe2, 235-40°/0.04 (HCl salt m. 174-5°), 15; SO2CF3, CH2CHMeCH2NMe2, 182-4°/0.2 (picrate m. 203-4°), 19; SO2CF3, (CH2)3N.(CH2)2.NMe.CH2.CH2, – [di-HCl salt m. 249.5° (decompn.)], 16; OH, (CH2)3NMe2, 220-5°/0.05, m. 90-1° (dimaleate m. 132-3°), 49. The following 8,10-substituted 2-azaphenothiazines were similarly prepd. (8,10 substituents, m.p. or b.p., % yield given): H, (CH2)3NMe2, 165-70°/0.007 [di-HCl salt m. 240.5-4.5° (decompn.)], 63; H, CH2CHMeCH2NMe2, 190-5°/0.6 (di-HCl salt m. 234-5°), 82; H, (CH2)3N.(CH2)2.N[(CH2)2OAc].CH2.CH2, – (dimaleate m. 147-8° (decompn.), 9; Cl, (CH2)3NMe2, 215-20°/1 (di-HCl salt m. 249-50°), 66.
PATENTGB 802725N-Aminoalkyl derivs. of I, where the alkyl is a straight or branched 2-5 C atom chain and the amino may be mono- or dialkylated or may be substituted by a pyrrolidino, piperidino, morpholino, or 4-alkyl-1-piperazinyl group, are prepd. by condensing I with the appropriate halo amine or by decompg. a phenothiazine-10-carboxylate of the appropriate amino alcohol. I (4.9 g.) was heated in 50 cc. boiling anhyd. xylene with 0.88 g. sodamide 1 hr., 2.71 g. 3-dimethylamino-1-chloropropane added, the soln. boiled 6 hrs., treated with H2O, then with dil. HCl, made alk. with NaOH, extd. with ether, and the solvent was evapd. in vacuo to give 4.5 g. 3-methylthio-10-(3-dimethylaminopropyl)phenothiazine (III), b0.2 206-18°; III.2HCl m. 160° (acetone-ether); picrate m. 135° (acetone). 3-Methylthio-10-(3-dimethylamino-2-methylpropyl)phenothiazine, m. 88-9°, was prepd. from I and 3-dimethylamino-2-methyl-1-chloropropane; picrate m. 145° (EtOH). The following were similarly prepd.: 3-methylthio-10-[3-(4-methyl-1-piperazinyl)propyl]phenothiazine, b0.1 250-6° [dihydrochloride m. 220° (decompn.) (acetone-ether); dipicrate m. 252-3° (acetone-iso-PrOH); 3-methylthio-10 – (2 – dimethylaminopropyl)phenothiazine, b0.2 202-6° (hydrochloride m. 205-6°; picrate m. 190°); 3-methylthio-10- (3-pyrrolidinopropyl)phenothiazine, b0.9 261° (hydrochloride m. 161°). I was phosgenated in toluene in the presence of pyridine to the 3-methylthiophenothiazine-10-carbonyl chloride (IV), m. 125°; IV heated in toluene with 3-(4- methyl-1-piperazinyl)-2-methylpropanol gave 3-(4-methyl-1- piperazinyl)-2-methylpropyl 3-methylthiophenothiazine-10- carboxylate (V) (dihydrochloride m. 225°). A soln. of 13 7 g. V in 60 cc. ο-Cl2C6H4 was boiled for 5 hrs. till CO2 evolution ceased, the soln. cooled, 60 cc. ether added and the mixt. H2O-washed, extd. with 10% HCl, made alk. with NaOH, and extd. with ether. The ether soln. was dried over anhyd. Na2SO4 and distd. in vacuo to yield 11.25 g. crude base which gave, with an EtOH soln. of maleic acid, 12.7 g. 3-methylthio-10-[3-(4-methyl-1-piperazinyl)-2-methyl-propyl]phenothiazinecarboxylic acid dimaleate, m. 199°. 3-Methylthio-10- [2,3-bis(dimethylamino)propyl] phenothiazine neutral fumarate, m. 198°, was similarly obtained by decarboxylating 1,3-bis(dimethylamino)-2-propyl 3-methylthiophenothiazine-10-carboxylate and treating with fumaric acid. 3-Methylthio-10-(3-diethylaminopropyl)phenothiazine-HCl, m. 172°, was prepd. from 3-methylthio-10-[3-(p-toluenesulfonyloxy)propyl]phenothiazine (VI) and Et2NH; 3-methylthio-10-(3-methylaminopropyl)phenothiazine (H oxalate m. 186°), from VI and MeNH2. VI heated with excess NH3 in toluene gave 3-methylthio-10-(3-aminopropyl)phenothiazine (VII) (oxalate m. 198°). VII in dioxane was neutralized with N HCl and treated with 30% aq. HCHO and PtO2 to give III. These compds. are antiemetics and potentiators of general anasthetics or neuroleptics.
SYN

///////////Methiomeprazine , antiemetic, Metiomeprazina, RP 10584, RP-10584, RP10584, RP 10584, SKF 6270
Systematic name (3):
- 10-[3-(ジメチルアミノ)-2-メチルプロピル]-2-(メチルチオ)-10H-フェノチアジン
- N,N,β-トリメチル-2-(メチルチオ)-10H-フェノチアジン-10-プロパン-1-アミン
- N,N,β-トリメチル-2-メチルチオ-10H-フェノチアジン-10-プロパン-1-アミン
Other name (6):
- メチオメプラジン
- Methiomeprazine
- 10-[3-(Dimethylamino)-2-methylpropyl]-2-(methylthio)-10H-phenothiazine
- SKF-6270
- N,N,β-Trimethyl-2-(methylthio)-10H-phenothiazine-10-propan-1-amine
CSc1ccc2Sc3ccccc3N(CC(C)CN(C)C)c2c1

NEW DRUG APPROVALS
ONE TIME
$10.00
Dichlorquinazine

CORRECT STR OF Dichlorquinazine
7-chloro-N-[1-[4-[2-[(7-chloroquinolin-4-yl)amino]propyl]piperazin-1-yl]propan-2-yl]quinolin-4-amine;methanesulfonic acid
- 1,4-Piperazinediethanamine, N,N’-bis(7-chloro-4-quinolinyl)-α,α’-dimethyl- (9CI)
- Quinoline, 4,4-[1,4-piperazinediylbis[(1-methylethylene)imino]]bis[7-chloro- (7CI)
- Quinoline, 4,4′-[1,4-piperazinediylbis[(1-methylethylene)imino]]bis[7-chloro- (8CI)
- N1,N4-Bis(7-chloro-4-quinolinyl)-α1,α4-dimethyl-1,4-piperazinediethanamine
- 1,4-Bis[2-(7-chloro-4-quinolylamino)propyl]piperazine
- Bis[(chloro-7”-quinolyl-4”)amino-2′-propyl]-1,4-piperazine
- Dichlorquinazine
- N,N’-Bis(7-chloro-4-quinolyl)-α,α’-dimethylpiperazine-1,4-diethylamine
- NSC 129790
- RP 12278
- WR 3863
WRONG STRUCTURE
WRONG STRUCTURE
Dichlorquinazine
- BRN 0867697
- Dichlorquinazine
- EINECS 234-130-6
- NSC 129790
- RP 12278
- UNII-HT3GAD2SCM
- WR 3863
cas 10547-40-7
C28H32Cl2N6, mw
| 523.5 |
7-chloro-N-[2-[4-[2-[(7-chloroquinolin-4-yl)amino]propan-2-yl]piperazin-1-yl]propan-2-yl]quinolin-4-amine
VARIANT
RN: 23256-65-7
Molecular Formula, C28-H32-Cl2-N6.C-H4-O3-S, Molecular Weight, 619.6144
- RP-12278 mesylate
- WR-3863 mesylate
- Quinoline, 4,4′-(1,4-piperazinediylbis((1-methylethylene)imino))bis(7-chloro-, tetramethanesulfonate bis((7-chloro-4”-quinolyl)-2′-aminopropyl)-1,4-piperazine methanesulfonate

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@amcrasto
/////////////////////////////////////////////////////////////////////////////////////////////////////
PATENTS
BE 626239
4-(Chloro or alkoxy)quinolines are treated with a 1,4-bis(aminoalkyl)piperazine to give the title compds. which can be used as antiinflammatory agents and as amebicides. Thus, a mixt. of 16.3 g. 4-chloroquinoline, 10 g. 1,4-bis(3-aminopropyl)piperazine, 55 g. PhOH, and 0.2 g. NH4Cl is heated 5 hrs. at 175°, poured into a mixt. of 500 ml. H2O and 100 ml. NaOH (d. 1.33), filtered, the ppt. is treated with a mixt. of 80 ml. H2O and 20 ml. NaOH, the mixt. filtered, and the ppt. washed with 500 ml. H2O and dried to give 15.9 g. 1,4-bis[3-(4-quinolyl)aminopropyl]piperazine, m. 210°(MeOH-H2O). Similarly prepd. are the following I: n, R, R1, R2, X, Y, m.p.; 2, H, H, H, MeO, H, 245° (HCONMe2); 2, H, H, H, H, SO2NMe2, 271° (HCONMe2); 2, H, H, H, H, CF3, 293° (HCONMe2); 3, Me, H, H, H, H, ∼100°; 3, Me, Ac, H, H, H, -(1); 3, Me, H, H, MeOH, 180° and 190°; 3, Me, Ac, H, MeO, H, -(2); 1, Me, H, Me, H, Cl, 264°; 2, H, H, H, Cl, H, 264° (BuOH); 1, Me, H, H, H, CF3, 240° (MeCOEt); 2, H, H, H, H, MeO, 200° (EtOH); 2, H, H, Me, H, MeO, 216° (EtOH); 3, Me, H, H, H, MeO, 218° (CH2Cl2); (1) bis(acid maleate) m. 155° (iso-PrOH), (2) bis(acid maleate) m. 155° The following II were also prepd.: n, R, R1, R2, m.p.; 1, Me, A(R = R1 = X = Y = H,Z =Cl), A(R = R1 = X = Z = H,Y = Cl), 208-10° (HCONMe2); 1, Me, A(R = R1 = X = Y= H, Z = Cl), A(R = R1 = X = Y = H,Z = MeO), 206-8° (HCONMe2); 1, Me, A(R1 = X = Y = H, R = 4-ClC6H4, Z = Cl), A(R = R1 = X = Y = H,Z = Cl, 230-2° (HCONMe2) The following III were prepd.: n, R, m, R1, R2, m.p.; 3, Me, 1, H, A(R = R1 = X = Y = H, Z= Cl), 190-1° and 213-15°; 2, H, 2, H, A(R = X = Y = H, R1 = Me, Z =Cl), 198° (PrOH); 3, Me, 2, H, A(R = R1 = X = Y = H,Z = Cl), 160-2°; 1, Me, 1, H, A(R = R1 = X = Y = H,Z = Cl), 178°; 1, Me, 1, Me, A(R1 = X = Z = H,R = Me, Y =AcNH), 330° (decompn.) (EtOH); 2, H, 2, H, A(R1 = X = Y = H,R = 4-ClC6H4,Z = Cl), 320-1° (HCONMe2); 2, H, 2, H, A(R = Y = Z = H, R1 = Me, X = Cl) 96° (iso-PrOH); 1, Me, 1, Me, A(R = R1 = X = Z = H, Y = Cl), 220° and 246-8°; 1, Me, 1, Me, A(R1 = X = Z = H, R = Me, Y = NH2), 305° (EtOH-H2O); 1, Me, 1, Me, A(R1 = X = Z = H, R = Me, Y = MeO, 244° (EtOH) Also prepd. were (m.p. given): 1,4-bis[2-(7-chloro-4-quinolylamino)propyl]hexahydro-1,4-diazepine, 169°; 1-[5-(7-chloro-4-quinolylamino)-2-pentyl]-4-[2-(7-chloro-4-quinolylamino)propyl] piperazine, 210-12°(HCONMe2); 1,4-bis[3-(7-chloro- 4-quinolylamino)propyl] hexahydro-1,4-diazepine, 186° (HCONMe2). The following were prepd. (m.p. and optical rotation given):L(+)-1,4-bis[2-(7-chloro-4-quinolylamino)propyl]piperazine, 250-1°, [α]23.5D 382° ± 1° (c 4, 50:50 MeOH-H2O); D(-)-1,4- bis[2-(7-chloro-4-quinolylamino)propyl] piperazine, 250-1°, [α]25D -382.5° ± 1° (c 4, 50:50 MeOH-H2O); DL-1,4-bis[2-(7-chloro-4-quinolylamino)propyl]piperazine (IV), 266-8°, -; meso-1,4-bis [2-(7-chloro-4-quinolylamino)propyl] piperazine (V), 270-1° (HCONMe2), -; equimol. mixt. of IV and V, 250-2°, -; 1,4-bis[2-(6-chloro-4-quinolylamino)propyl]piperazine-form A (VI-form A), 227° -; VI-form B, 110° and 245°, -. Also prepd. are the following intermediates of the general formula VII (R = H) (X, Y, Z, and m.p. given): OH, H, SO2NMe2, ∼288°; Cl, H,SO2NMe2, 170°; HO(CH2)3CHMeNH, H, H, 158° (EtOH); AcO(CH2)3CHMeNAc, H, H, -; HO(CH2)3CHMeNAc, H, H, -; MeSO3(CH2)3CHMeNAc, H, H, -; N-(5-piperazino-2-pentyl)acetamido, H, H, -; HO(CH2)3CHMeNH, MeO, H, -; AcO(CH2)3CHMeNAc, MeO, H, -; HO(CH2)3CHMeNAc, MeO, H, -; MeSO3(CH2)3CHMeNAc, MeO, H, -; N-(5-piperazino-2-pentyl)acetamido, MeO, H, -; Me(HOCH2)CH, H, Cl, 210°; Me(ClCH2)CH, H, Cl, 148-50°; Me(HOCH2)CH, Cl, H, 192°; Me(ClCH2)CH, Cl, H, 142°; Me(HOCH2)CH, H, MeO, 170°; Me(ClCH2)CH, H, MeO, 160°. Also prepd. were (m.p. given): VII (R = CO2Et, X = OH, Y = H, Z = SO2NMe2), ∼335°; VII (R = CO2H, X = OH, Y = H, Z = SO2HMe2), 310° (decompn.); 1,4-bis(2-oxopropyl)hexahydro-1,4-diazepine, -; 1,4-bis(2-oximinopropyl)hexahydro-1,4-diazepine, 180-1°; 1,4-bis(2-aminopropyl)hexahydro-1,4-diazepine, -; 1,4-bis(2-cyanoethyl)-hexahydro-1,4-diazepine, -. The following were prepd. (m.p. and optical rotation given): L(+)-4-(3-hydroxy-2-propylamino)-7-chloroquinoline, 223-4°, [α]24D 28.5° ± 2° (c 1, EtOH); L(+)-4-(3-chloro-2-propylamino)-7-chloroquinoline, 146-7°, [α]24D 103 ± 1° (c 2, EtOH); L(+)-4-(3-piperazino-2-propylamino)-7-chloroquinoline, 128-30°, [α]23D 139 ± 1° (c 2, EtOH); D(-)-4-(3-hydroxy-2-propylamino)-7-chloroquinoline, 223-4°, [α]25D – 31 ± 2° (c 1, EtOH); D(-)-4-(3-chloro-2-propylamino)-7-chloroquinoline, 147-8°, [α]24D -101 ± 1° (c 2, EtOH); D(-)-4-(3-piperazino-2-propylamino)-7-chloroquinoline, 131-2°, [α]23D -137 ± 1° (c 2, EtOH)
PATENT
FR CAM42 19631007.
Piperazines (I) are antiinflammatory and anthelmintic agents. A mixt. of 8.25 g. MeCH(NH2)CH2OH, 19.8 g. 4,6-dichloroquinoline, and 55 g. PhOH is heated to give 16.0 g. 6-chloro-4-[(3-hydroxy-2-propyl)-amino]quinoline (II), m. 192°. II (14.0 g.) is treated with a soln. of 10.6 g. SOCl2 in 40 ml. CHCl3 to give 12.5 g. 6-chloro-4-[(3-chloro-2-propyl)amino]quinoline (III), m. 142°. A mixt. of 13.2 g. 1-[2-(7-chloro-4-quinolylamino)propyl]piperazine, 11.0 g. III, 6.4 g. NaI, 2.3 g. anhyd. Et3N, and 200 ml. AcEt is refluxed 18 hrs., the solvent is distd. in vacuo, and the residue is taken up in 100 ml. MeOH. The mixt. is made alk. with 110 ml. NaOH (d. 1.33), poured into 1000 ml. H2O, and the ppt. that forms is filtered off, washed with H2O, and recrystd. in HCONMe2 to give 11.0 g. 1-[2-(7-chloro-4-quinolylamino)propyl]-4-[2-(6-chloro-4-quinolylamino)propyl]piperazine, m. 208-10°. Similarly prepd. are the following I (R, m, R1, n, R2, R3, R4, and m.p. given): H, 2, H, 2, H, MeO, H, 245°; H, 2, H, 2, H, H, SO2NMe2, 271°; H, 2, H, 2, H, H, CF3, 293°; Me, 3, Me, 3, H, MeO, H, 180° and 190°; Me, 3, H, 1, H, H, Cl, 190-1° and 213-15°; H, 2, H, 2, H, Cl, H, 264°; Me, 1, Me, 1, H, H, CF3, 240°; H, 2, H, 2, H, H, MeO, 200°; Me, 3, H, 2, H, H, Cl, 160-2°; Me, 1, H, 1, H, H, Cl, 178°; Me, 1, Me, 1, Me, AcNH, H, 330°; H, 2, H, 2, p-ClC6H4, H, Cl, 320-1°; Me, 1, Me, 1, H, Cl, H, 227° (form A); Me, 1, Me, 1, H, Cl, H, 110° and 245° (form B); H, 3, H, 3, H, H, Cl, 239-41°; Me, 1, Me, 1, Me, NH2, H, 305°; Me, 1, Me, 1, Me, MeO, H, 244°; Me, 3, Me, 3, Me, 3, H, H, MeO, 218°; H, 3, H, 3, H, H, Cl, 240-2°. Also prepd. are (m.p. given): 1,4-bis[2-(7-chloro-4-quinolylamino)propyl]hexahydrodiazepine, 169°; 2,5-dimethyl-1,4-bis[2-(7-chloro-4-quinolylamino)propyl)piperazine, 264°; 1-[5-(7-chloro-4-quinolylamino [-2-pentyl]-4-[2-(7-chloro -4-quinolylamino)propyl]piperazine, 210-12°; 2,5-dimethyl-1,4-bis[3-(7-methoxy-4-quinolylamino)propyl]piperazine, 216°; 1,4-bis[3-(3-methyl-7-chloro-4-quinolylamino)propyl] piperazine, 198°; 1,4-bis[3-(7-chloro-4-quinolylamino)propyl]hexahydrodiazepine, 186°; 1-[2(7-chloro-4-quinolylamino)propyl]-4-[2-(7-methoxy-4-quinolylamino)propyl]piperazine, 206-8°; 1,4-bis[3-(3-methyl-5-chloro-4- quinolylamino)propyl]piperazine, 96°; 1 – [2 -[2 -(p – chlorophenyl)- 7- chloro- 4- quinolylamino]propyl] -4 – [2 – (7 – chloro – 4-quinolylamino)propyl]piperazine, 230-2°; L(+) 1,4-bis[2-(7-chloro-4-quinolylamino)propyl]piperazine, 250-1°, [α]23.5D + 382° ± 1° (c 4, 50/50 MeOH-H2O); L(+)-7-chloro-4-(3-hydroxy-2-propylamino)quinoline, 223-4°, [α]24D 28.5° ± 2° (c 1, EtOH); L(+)-7-chloro-4-(3-chloro-2-propylamino)quinoline, 146-7°, [α]24D 103° + 1° (c 2, EtOH); L(+)-7-chloro-4-(3-piperazino-2-propylamino)quinoline 128-30°, [α]23D 139° ± 1° (c 2, EtOH); D(–)-1,4-bis[2-(7-chloro-4-quinolylamino)propyl]piperazine, 250-1°, [α]25D -382° ± 1° (c 4, 50:50 MeOH-H2O); meso- 1,4 – bis [2 – (7 – chloro – 4 – quinolylamino)propyl] piperazine, 270-1°.
Patent Information
BE 612207

| Publication Number | Title | Priority Date | Grant Date |
|---|---|---|---|
| US-2016045487-A1 | Compositions and methods for treating neuropathy | 2013-03-27 | |
| WO-2014160811-A1 | Compositions and methods for treating neuropathy | 2013-03-27 | |
| AU-2014234258-A1 | Piperaquine microcapsules and compositions containing them | 2013-03-22 | |
| AU-2014234258-B2 | Piperaquine microcapsules and compositions containing them | 2013-03-22 | 2019-02-14 |
| CA-2907628-A1 | Piperaquine microcapsules and compositions containing them | 2013-03-22 |
| Publication Number | Title | Priority Date | Grant Date |
|---|---|---|---|
| EP-2976069-A1 | Piperaquine microcapsules and compositions containing them | 2013-03-22 | |
| EP-2976069-B1 | Piperaquine microcapsules and compositions containing them | 2013-03-22 | 2020-05-06 |
| US-2014322296-A1 | Piperaquine microcapsules and compositions containing them | 2013-03-22 | |
| US-2016045447-A1 | Piperaquine microcapsules and compositions containing them | 2013-03-22 | |
| US-9668979-B2 | Piperaquine microcapsules and compositions containing them | 2013-03-22 | 2017-06-06 |
| Publication Number | Title | Priority Date | Grant Date |
|---|---|---|---|
| WO-2014147242-A1 | Piperaquine microcapsules and compositions containing them | 2013-03-22 | |
| AU-2009215107-A1 | Treatments for neuropathy | 2008-02-12 | |
| AU-2009215107-B2 | Treatments for neuropathy | 2008-02-12 | 2013-05-09 |
| AU-2013203934-A1 | Treatments for neuropathy | 2008-02-12 | |
| CA-2714676-A1 | Treatments for neuropathy | 2008-02-12 |
| Publication Number | Title | Priority Date | Grant Date |
|---|---|---|---|
| CA-2714676-C | Treatments for neuropathy | 2008-02-12 | 2015-04-14 |
| EP-2240177-A2 | Treatments for neuropathy | 2008-02-12 | |
| US-2009203735-A1 | Treatments for neuropathy | 2008-02-12 | |
| US-2011086878-A1 | Treatments for Neuropathy | 2008-02-12 | |
| US-2016058749-A1 | Treatments for neuropathy | 2008-02-12 |
////////////////Dichlorquinazine, BRN 0867697, Dichlorquinazine, EINECS 234-130-6, NSC 129790, RP 12278, UNII-HT3GAD2SCM, WR 3863
CC(C)(NC1=C2C=CC(=CC2=NC=C1)Cl)N3CCN(CC3)C(C)(C)NC4=C5C=CC(=CC5=NC=C4)Cl
WRONG
CC(CN1CCN(CC(C)Nc2ccnc3cc(Cl)ccc23)CC1)Nc4ccnc5cc(Cl)ccc45.CS(=O)(=O)O
AND
Clc1ccc2c(c1)nccc2NC(C)CN1CCN(CC(C)Nc2ccnc3cc(Cl)ccc32)CC1
CORRECT

NEW DRUG APPROVALS
ONETIME
$10.00
LERIGLITAZONE
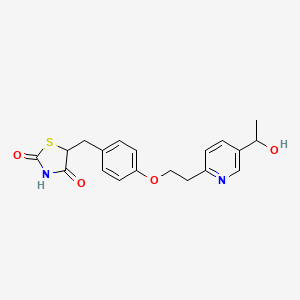
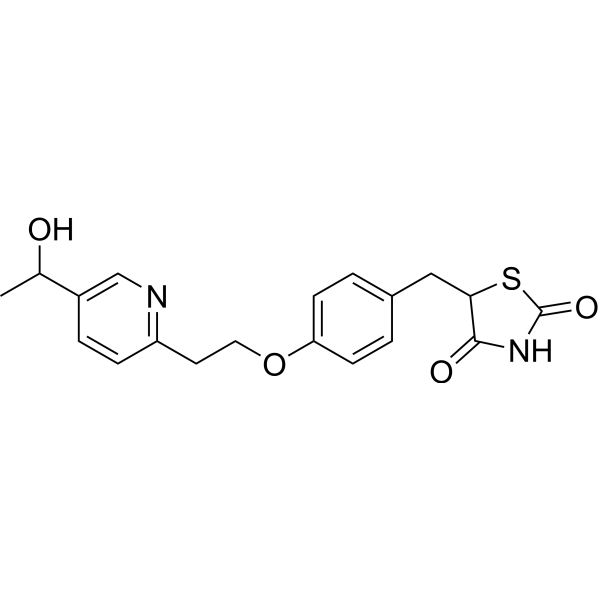
LERIGLITAZONE
C19H20N2O4S,
| MW 372.4 |
Hydroxypioglitazone, CAS 146062-44-4
MIN 102, Hydroxy Pioglitazone (M-IV)лериглитазон [Russian] [INN]ليريغليتازون [Arabic] [INN]乐立格列酮 [Chinese] [INN]
5-[[4-[2-[5-(1-hydroxyethyl)pyridin-2-yl]ethoxy]phenyl]methyl]-1,3-thiazolidine-2,4-dione
Hydroxypioglitazone is a member of the class of thiazolidenediones that is the hydroxy derivative of pioglitazone. It has a role as a human xenobiotic metabolite. It is a member of thiazolidinediones, a member of pyridines and an aromatic ether. It derives from a pioglitazone.
- OriginatorIDIBELL
- DeveloperMinoryx Therapeutics
- ClassNeuroprotectants; Phenyl ethers; Pyridines; Small molecules; Thiazolidinediones
- Mechanism of ActionPeroxisome proliferator-activated receptor gamma agonists
- Orphan Drug StatusYes – Adrenoleucodystrophy; Friedreich’s ataxia
- Phase II/IIIAdrenoleucodystrophy
- Phase IIFriedreich’s ataxia
- PreclinicalCNS disorders
- 23 Sep 2020Leriglitazone receives Rare Pediatric Disease designation from the US FDA for X-linked adrenoleukodystrophy before September 2020
- 23 Sep 2020Minoryx Therapeutics licenses leriglitazone to Sperogenix Therapeutics in China, Hong Kong and Macau for X-linked adrenoleukodystrophy (X-ALD)
- 14 Sep 2020Minoryx Therapeutics completes the phase II FRAMES trial in Friedreich’s ataxia (In adolescents, In adults) in Spain, Germany, France and Belgium (PO) (NCT03917225)

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@amcrasto
/////////////////////////////////////////////////////////////////////////////////////////////////////
Leriglitazone (Hydroxypioglitazone), a metabolite of pioglitazone. Leriglitazone (Hydroxypioglitazone) PioOH is a PPARγ agonist, stabilizes the PPARγ activation function-2 (AF-2) co-activator binding surface and enhances co-activator binding, affording slightly better transcriptional efficacy. Leriglitazone (Hydroxypioglitazone) binds to the PPARγ C-terminal ligand-binding domain (LBD) with Ki of 1.2 μM,induces transcriptional efficacy of the PPARγ (LBD) with EC50 of 680 nM.
Leriglitazone is under investigation in clinical trial NCT03917225 (A Clinical Study to Evaluate the Effect of MIN-102 on the Progression of Friedreich’s Ataxia in Male and Female Patients).
Treatment of X-Linked Adrenoleukodystrophy
PATENT
WO 9218501
WO 9322445
PAPER
Chemical & Pharmaceutical Bulletin (1995), 43(12), 2168-72
https://www.jstage.jst.go.jp/article/cpb1958/43/12/43_12_2168/_article
The metabolites of (±)-5-[p-[2-(5-ethyl-2-pyridyl)ethoxy]benzyl]-2, 4-thiazolidinedione (1, pioglitazone), which is a representative insulin-sensitizing agent, were synthesized to confirm their structures and for studies of their pharmacological properties. Of the metabolites identified, a compound hydroxylated at the 2-position of the ethoxy chain (3) and compounds oxygenated at the ethyl side chain attached to the pyridine ring (4, 5) were found to be active, although the potency was slightly lower than that of the parent compound.




PAPER
Journal of Medicinal Chemistry (1996), 39(26), 5053-5063.
https://pubs.acs.org/doi/10.1021/jm9605694
Pioglitazone (5-(4-(2-(5-ethyl-2-pyridyl)ethoxy)benzyl)-2,4-thiazolidinedione, 2) is a prototypical antidiabetic thiazolidinedione that had been evaluated for possible clinical development. Metabolites 6−9 have been identified after dosing of rats and dogs. Ketone 10 has not yet been identified as a metabolite but has been added to the list as a putative metabolite by analogy to alcohol 6 and ketone 7. We have developed improved syntheses of pioglitazone (2) metabolites 6−9 and the putative metabolite ketone 10. These entities have been compared in the KKAy mouse model of human type-II diabetes to pioglitazone (2). Ketone 10 has proven to be the most potent of these thiazolidinediones in this in vivo assay. When 6−10 were compared in vitro in the 3T3-L1 cell line to 2, for their ability to augment insulin-stimulated lipogenesis, 10 was again the most potent compound with 6, 7, and 9 roughly equivalent to 2. These data suggest that metabolites 6, 7, and 9 are likely to contribute to the pharmacological activity of pioglitazone (2), as had been previously reported for ciglitazone (1).
PATENT
WO 2015150476
Compound 5-[4-[2-(5-(1 -hydroxyethyl)-2-pyridinyl)ethoxy]benzyl]-2,4-thiazolidinedione of formula (1 ) can be prepared according to Scheme 1 (see e.g. J.Med.Chem. 1996, 39(26),5053).
Scheme 1
Scheme 2
Yet another method to prepare mixtures (c) – comprising compound (2) and (4) – and (d) – comprising compounds (3) and (5) – (scheme 3), includes the resolution of the racemic mixture VIII using the already described methods (chiral HPLC separation, enzymatic resolution, chiral resolution, etc) followed by double bond reduction in each of the enantiomers Villa and Vlllb.
Scheme 4
Compounds of formula (2), (3), (4) and (5) may be obtained from mixtures (c) and (d) (Scheme 45) by chiral HPLC separation. Alternatively, the desired enantiomerically pure compounds can be prepared by chiral synthetic procedures known to those skilled in the art (for example: asymmetric hydrogenolysis of the corresponding single isomer of compound VI).
HPLC Method
Column: Symmetry Shield RP-18, 5 μηη (4.6 x 250 mm); wavelength: 210 nm; flow: 1 mL/min; run time: 28 min; mobile phase-gradient: (t/%B): 0/10, 8/10, 12/60, 16/80, 20/80, 24/10, 28/10 [A: Water (potassium dihydrogen o-phosphate (pH~3)), B: Acetonitrile]
A mixture of compounds (2) and (4) (mixture (c)) and a mixture of compounds (3) and (5) (mixture (d)) were prepared according to Scheme 7.
Example 6: Preparation of diastereomeric mixtures D-1 and D-2 of M-IV:
Scheme 1 :
Ent-1 (VIII) Ent-2 (VIII)
Step 3 Step 3
MIV D-1 MIV D-2
Step 1 : Synthesis of compound VIII: HCI (48 ml, 2N) was added to a solution of compound VI (10 g, 0.024 mol) in methanol (200 ml) and the mixture was heated to reflux. After 4 h of reflux, the reaction mixture was cooled to r.t. and concentrated under reduced pressure to afford a yellow solid. The solid was suspended in water (70 ml) and neutralized using a saturated NaHC03 solution. The resulting pale yellow precipitate was collected by filtration and vacuum dried to afford compound VIII (7.5 g; 84% yield).
ES-MS [M+1]+: 371.0.
Step 2: Chiral prep. HPLC
Compound VIII (1 .0 g) was dissolved in a mixture containing equal volumes of acetonitrile, methanol and dichloromethane; injected (150 μΙ injections) in chiral prep-HPLC column (Chiralpak-IA 250 x 20 mm, 5 micron) and separated [Mobile phase- n-Hexane/0.05% Et3N in EtOH (50:50); flow Rate: 18ml/min; run time: 60 min]. The fractions containing the enantiomers Villa and Vlllb were separately concentrated under reduced pressure to minimum volume and the respective residues were diluted with EtOAc (100 ml), followed by water (50 ml). The resultant organic phases were
dried over anhydrous Na2S04 and concentrated to afford compounds Villa and Vlllb as off-white solids. Enantiomers Villa and Vlllb were isolated but the absolute configuration of each enantiomer has not been determined.
Compound Ent-1 (VIII): 250 mg (Yield: 50%); tR (Chiral HPLC) = 14.8 min; ES-MS [M+1]+: 371 .0; 1H NMR (400 MHz, DMSO-d6): δ 12.5 (br S, 1 H), 8.47 (s, 1 H), 7.71 (s, 1 H), 7.67 (dd, J = 8.0, 2.0 Hz, 1 H), 7.53 (d , J = 9.2 Hz, 2H), 7.31 (d, J = 7.6 Hz, 1 H), 7.08 (d, J = 8.8 Hz, 2H), 5.25 (d, J = 4.0 Hz, 1 H), 4.74-4.76 (m, 1 H), 4.43 (dd, J = 6.8, 6.4 Hz, 2H), 3.18 (t, J = 6.4 Hz, 2H), 1.34 (d, J = 6.4 Hz, 3H).
Compound Ent-2 (VIII): 237 mg (Yield: 47%); tR (Chiral HPLC) = 16.7 min; ES-MS [M+1]+: 371 .0; 1H NMR (400 MHz, DMSO-d6): δ 12.5 (br S, 1 H), 8.47 (s, 1 H), 7.71 (s, 1 H), 7.67 (dd, J = 8.0, 2.0 Hz, 1 H), 7.53 (d , J = 8.8 Hz, 2H), 7.31 (d, J = 8.0 Hz, 1 H), 7.08 (d, J = 9.2 Hz, 2H), 5.23 (d, J = 3.6Hz, 1 H), 4.75 (m, 1 H), 4.43 (dd, J = 6.8, 6.4 Hz, 2H), 3.18 (dd, J = 6.8, 6.4 Hz, 2H), 1 .34 (d, J = 6.4 Hz, 3H).
Synthesis of diastereomeric mixtures of M-IV
Synthesis of D-1 MIV
Step 3: A solution of NaBH4 (77 mg, 2.02 mmol) in 0.1 N NaOH (2 ml) was added slowly to a stirred solution of compound Ent-1 (VIII) (250 mg, 0.675 mmol), dimethylglyoxime (32 mg, 0.27 mmol) and CoCI2.6H20 (16 mg, 0.067 mmol) in a mixture of water (10 ml), THF (10 ml) and 1 M NaOH (0.5ml) solution at 10 °C, and the reaction mixture was stirred at r.t. for 1 h. After color of the reaction medium faded, additional quantity of NaBH4 (26 mg, 0.675 mmol) and CoCI2.6H20 (16 mg, 0.067 mmol) were added and stirring was continued at r.t. [additional quantities of CoC|2 and NaBH4 were added at 12 h intervals till the starting material was consumed, as monitored by LCMS]. After 90-96 h, the reaction mixture was neutralized with AcOH (pH~7); diluted with water (10 ml) and extracted in EtOAc (3 χ 50 ml). The combined organic extract was dried over anhydrous Na2S04 and concentrated to afford crude compound which was purified by flash column chromatography (Si02; 4% methanol in CH2CI2) to afford diastereomeric mixture of MIV D-1 (125 mg) as off-white solid.
Synthesis of D-2 MIV
Step 3: A solution of NaBH4 (72 mg, 1 .921 mmol) in 0.1 N NaOH (2 ml) was added slowly to a stirred solution of compound Ent-2 (VIII) (237 mg, 0.64 mmol), dimethylglyoxime (30 mg, 0.256 mmol) and CoCI2.6H20 (15 mg, 0.064 mmol) in a mixture of water (10 ml), THF (10 ml), and 1 M NaOH (0.5ml) solution at 10 °C, and the
reaction mixture was stirred at r.t. for 1 h. After color of the reaction medium faded, additional quantity of NaBH4 (24 mg, 0.64 mmol) and CoCI2.6H20 (15 mg, 0.064 mmol) were added and stirring was continued at r.t. [additional quantities of CoCI2.6H20 and NaBH4 were added at 12 h intervals till the starting material was consumed, as monitored by LCMS]. After 96 h, the reaction mixture was neutralized with AcOH (pH~7); diluted with water (10 ml) and extracted in EtOAc (3 χ 50 ml). The combined organic extract was dried over anhydrous Na2S04 and concentrated to afford crude compound, which was purified by flash column chromatography (Si02; 4% methanol in CH2CI2) to afford diastereomeric mixture of MIV D-2 (100 mg) as off-white solid.
MIV D-1 : yield: 125 mg (50%); tR (Chiral HPLC) = 17.8, 14.7 min; ES-MS [M+1]+: 373.0, 1H NMR (400 MHz, DMSO-d6): δ 12.00 (br s, NH), 8.46 (d, J = 2.0 Hz, 1 H), 7.67 (dd, J = 8.0, 2.4 Hz, 1 H), 7.30 (d, J = 8.0 Hz, 1 H), 7.13 (d, J = 8.8Hz, 2H), 6.86 (d, J = 8.4 Hz, 2H), 5.27 (d, J = 4.0 Hz, 1 H), 4.88-4.85 (m, 1 H), 4.76-4.74 (m, 1 H), 4.30 (t, J = 6.8 Hz, 2H), 3.30 (m, 1 H), 3.14 (dd, J = 6.8, 6.4 Hz, 2H), 3.08-3.02 (m, 1 H), 1 .34 (d, J = 6.4 Hz, 3H).
MIV D-2: yield: 100 mg (42%); tR (Chiral HPLC) = 19.4, 16.5 min; ES-MS [M+1]+: 373.0; 1H NMR (400 MHz, DMSO-d6): δ 12.01 (br s, -NH), (d, J = 2.0 Hz, 1 H), 7.67 (dd, J = 8.0, 2.0 Hz, 1 H), 7.31 (d, J = 8.0 Hz, 1 H), 7.13 (d, J = 8.8 Hz, 2H), 6.86 (d, J = 8.8 Hz, 2H), 5.27 (d, J = 4.0 Hz, 1 H), 4.88-4.85 (m, 1 H), 4.76-4.74 (m, 1 H), 4.30 (dd, J = 6.8, 6.4 Hz, 2H), 3.30 (m, 1 H), 3.14 (dd, J = 6.8, 6.4 Hz, 2H), 3.08-3.02 (m, 1 H), 1.34 (d, J = 6.8 Hz, 3H).
Diastereomeric mixtures D-1 and D-2 of MIV correspond to mixtures (c) and (d) described above, but the specific diastereomers present in each diastereomeric mixture have not been assigned.
Example 7: in vitro ADME and toxicological characterization
Protocol: The assays performed include cytochrome P450 inhibition with the different isoforms, microsomal and hepatocyte stability, neurotoxicity in neural cells and hERG safety assays using a patch clamp electrophysiology measurement (FDA Draft Guidance for Industry. Drug Interaction Studies – Study Design, Data Analysis, Implications for Dosing, and Labelling Recommendations 2012, The European Medicines Agency (EMA) Guideline on the Investigation of Drug Interactions Adopted in 2012, Schroeder K et al. 2003 J Biomol Screen 8 (1 ); 50-64, Barter ZE et al. 2007
Curr Drug Metab 8 (1 ); 33-45, LeCluyse EL and Alexandre E 2010 Methods Mol Biol 640; 57-82). The results indicate a safe and favourable ADME profile for the compounds of the invention.
Example 8: The brain plasma ratios of Pioglitazone, MIV, Mill and Mil following oral dosing of a single administration of Pioglitazone at 4.5 mg/kg in male C57BL/6 mice.
The brain-plasma ratio was calculated based on levels of Pioglitazone, MIV, Mill and Mllin plasma and brain quantified at C max (maximal concentration) following oral dosing of a single administration of Pioglitazone at 4.5 mg/kg in male C57BL/6 mice. The percentage brain plasma ratio was 9, 13, 7 and 1 %, respectively, for Pioglitazone, Mil and Mill as shown in the Figure 4. Thus, active metabolites Mill and Mil crossed the BBB at much lower extent than Pioglitazone as it was predicted based on the physicochemical properties of the compounds (see Tablel ). In contrast, unexpectedly metabolite MIV crossed the BBB in a higher percentage than the parent compound Piolgitazone
The calculations of the both indexes (ClogP and QPIogBB) for Pioglitazone and its metabolites Mil and Mill are shown in Table 1 . For both indexes the 2 metabolites are lower than for pioglitazone, suggesting for Mil, and Mill a less favored penetration and distribution within CNS.
TABLE 1
PATENT
WO 2018116281
https://patents.google.com/patent/WO2018116281A1/enPioglitazone is a “dirty” drug which is converted to many metabolites in vivo. The metabolic pathway of pioglitazone after oral administration has been studied in several animal species and in humans and the metabolites have been described in the literature (see e.g. Sohda et al, Chem. Pharm. Bull., 1995, 43(12), 2168-2172) and Maeshiba et al, Arzneim.-Forsch/Drug Res, 1997, 47 (I), 29-35). At least six metabolites have been identified, named M-I to M-VI. Amongst these metabolites, M-II, M-III and M-IV show some pharmacological activity but are less active than Pioglitazone in diabetic preclinical models.
[0005] 5-[[4-[2-[5-(l-hydroxyethyl)-2-pyridinyl]ethoxy]phenyl]methyl]-2,4- thiazolidinedione has the following structure:

[0006] Tanis et al. (J. Med. Chem. 39(26 ):5053-5063 (1996)) describe the synthesis of 5-[[4-[2-[5-( 1 -hydroxyethyl)-2-pyridinyl]ethoxy]phenyl]methyl]-2,4-thiazolidinedione as follows:Scheme 1

[0007] Tanis et al. describe that the intermediate 14 was obtained in a 27% yield by reacting compound 13 in an aqueous 37% formaldehyde at 170°C for 6 hours. In this process, 5-[[4- [2-[5-( 1 -hydroxyethyl)-2-pyridinyl]ethoxy]phenyl]methyl]-2,4-thiazolidinedione (compound 6 in Scheme 1) was obtained in a 2.47% overall yield.[0008] WO 2015/150476 Al describes the use of 5-[[4-[2-[5-(l-hydroxyethyl)-2- pyridinyl]ethoxy]phenyl]methyl]-2,4-thiazolidinedione, and its pharmaceutically acceptable salts, in the treatment of central nervous system (CNS) disorders. WO 2015/150476 Al describes that 5-[[4-[2-[5-(l-hydroxyethyl)-2-pyridinyl]ethoxy]phenyl]methyl]-2,4- thiazolidinedione was prepared according to the process of Tanis et al. (supra) where the intermediate corresponding to compound 14 of Tanis et al. was prepared similarly at 160°C for 5 hours providing a 17% yield. The overall yield of 5-[[4-[2-[5-(l-hydroxyethyl)-2- pyridinyl]ethoxy]phenyl]methyl]-2,4-thiazolidinedione was about 1.5%.[0009] Due to the low yield of the intermediate 2-[5-(l-methoxymethoxy-ethyl)pyridine-2- yl]ethanol, the process step for preparing this intermediate is critical for the overall yield of the product, 5-[[4-[2-[5-(l-hydroxyethyl)-2-pyridinyl]ethoxy]phenyl]methyl]-2,4- thiazolidinedione. In addition, the prior art process to obtain compound 14 is difficult to scale because the reaction is carried out in a pressure vessel at a very high temperature and it is a very dirty reaction.[0010] Accordingly, the processes described in the art afford the product 5-[[4-[2-[5-(l- hydroxyethyl)-2-pyridinyl]ethoxy]phenyl]methyl]-2,4-thiazolidinedione only in a very low overall yield and, therefore, they are not suitable for large scale synthesis. In addition, the prior art process employs CH3OCH2CI, a known carcinogen, for protecting the hydroxyl group in the key intermediate. There is a need for an improved process for synthesizing 5- [[4-[2-[5-(l-hydroxyethyl)-2-pyridinyl]ethoxy]phenyl]methyl]-2,4-thiazolidinedione, and its pharmaceutically acceptable salts.Formula I illustrated by Scheme 2:Scheme 2 r
B


deprotectionoptional saltformation

I (HCI salt)[0255] In another embodiment, the disclosure provides a process for preparing the compound of Formula I illustrated by Scheme 3 : Scheme 3C
Br. e

step ‘< step b step c

step step g

[0256] In another embodiment in Scheme 3, step c, the order of mixing of the reagents can be as follows: 1. n-BuLi, 2. ethylene oxide, and 3. Cul. This order of mixing is described in Example 2.[0257] In the step a, 2,5-dibromopyridine (1) is reacted with i-PrMgCl in THF and then further with acetaldehyde to obtain compound 2. The reaction mixture is preferably filtered over Celite® after the reaction to remove most of the salts. In one embodiment, the addition of acetaldehyde is conducted at a temperature between -15°C and -10°C to control the exothermic reaction. [0258] In the step b, compound 2 is reacted with TBDMS-C1 in the presence of imidazole having DMF as a solvent. The crude product 3 is advantageously purified by a short plug filtration.[0259] In the step c, the hydroxyl protected compound 3 is reacted with ethylene oxide in the presence of n-BuLi and Cu(I)iodide while maintaining the reaction temperature, i.e., the reaction mixture temperature, below -20°C. In one embodiment, the reaction temperature is maintained below -55°C while adding n-BuLi and Cu(I)iodide into the reaction mixture. In another embodiment, the temperature of the reaction mixture is maintained below -55°C while adding n-BuLi, followed by ethylene oxide and then Cu(I)iodide into the reaction mixture. In another embodiment, the temperature of the reaction mixture is maintained below -55°C while adding n-BuLi into the reaction mixture, followed by ethylene oxide. In this embodiment, Cu(I)iodide is added then into the reaction mixture while the reaction mixture temperature is maintained below -20°C, and preferably below -55 °C. The reaction mixture is then allowed to slowly warm to room temperature after the addition of the reagents and stirred at room temperature, e.g., 20-25°C, overnight. This process is described in detail in Example 2. After the reaction, the complexed copper is advantageously removed by washing with 10% ammonia. The crude compound 4 can be purified by column chromatography to give >99% pure product with a yield of about 52%.[0260] The following examples are illustrative, but not limiting, of the methods of the present invention. Suitable modifications and adaptations of the variety of conditions and parameters normally encountered in clinical therapy and which are obvious to those skilled in the art in view of this disclosure are within the spirit and scope of the invention.ExamplesCOMPARATIVE EXAMPLE 1Synthesis of 5-[[4-[2-[5-(l-hydroxyethyl)-2-pyridinyl]ethoxy]phenyl]methyl]- 2,4-thiazolidinedione (9a) according to the process described in WO 2015/150476 Al Scheme 4

8a 9a[0261] (a) Synthesis of l-(6-methyl-pyridin-3-yl)-ethanol (3a)[0262] LiHMDS (1.0 M in tetrahydrofuran, 463 ml, 0.463 mol) was added drop wise to a cooled solution of methyl 6-methylnicotinate (la) (20 g, 0.132 mol) and ethyl acetate (82 g, 0.927 mol) in dimethylformamide at -50°C; gradually raised the temperature to room temperature and stirred at the same temperature. After 1 h, the reaction mixture was cooled to 0°C; slowly diluted with 20% sulphuric acid and heated to reflux. After 4 h, the reaction mixture was cooled to room temperature, and further to 0°C and basified with potassium carbonate. The reaction medium was diluted with water and extracted in ethyl acetate (3×50 mL). Combined organic extract was dried over sodium sulphate and concentrated to afford crude l-(6-methylpyridin-3-yl)ethan-l-one (2a) (20.0 g) which was taken to the next step without any purification. ES-MS [M+l]+: 136.1.Sodium borohydride (2.3 g, 0.06 mol) was added in small portions over 30 min, to a solution of compound 2a (16.4 g, 0.121 mol) in ethanol (160 mL) at 0°C and the reaction mixture was stirred at same temperature. After 1 h, the reaction mixture was diluted with sodium bicarbonate solution (sat) (2×200 mL) and extracted with dichloromethane (2×500 mL). The combined organic extract was dried over anhydrous sodium sulphate and concentrated to afford a pale yellow oil, which was purified by flash column chromatography (5% methanol/dichloromethane) to afford compound 3a (17.0 g; 93% yield over 2 steps) as a pale yellow oil. ES-MS [M+l]+: 138.1. 1H NMR (400 MHz, CDC13): δ 8.35 (d, J = 2.0 Hz, 1H), 7.63 (dd, J = 8.0, 2.4 Hz, 1H), 7.12 (d, J = 8.0 Hz, 1H), 4.89 (q, J = 6.5 Hz, 1H), 3.30 (br s, 1H), 2.50 (s, 3H), 1.48 (d, J = 6.5 Hz, 3H).[0263] (b) Synthesis of 5-(l-methoxymethoxy-ethyl)-2-methyl-pyridine (4a):Compound 3a (15 g, 0.109 mol) was added, drop wise, to a cooled suspension of sodium hydride (6.56 g, 0.164 mol) in tetrahydrofurane (150 mL) and stirred at 0°C. After 30 min, chloromethyl methyl ether (13.2 g, 0.164 mol) was added drop wise while stirring and keeping the internal temperature around 0°C. After addition is over, the reaction mixture was stirred at the same temperature for 1 h. The reaction was quenched with ice cold water (80 mL) and extracted with ethyl acetate (3×50 mL). The combined organic extract was dried over anhydrous sodium sulphate and concentrated to afford an orange color oil, which was purified by flash column chromatography (1% methanol/dichloromethane) to afford compound 4a (10.0 g; 51% yield) as a pale yellow oil. ES-MS [M+l]+: 182.2. 1H NMR (400 MHz, CDC13): δ 8.45 (d, J = 2.0 Hz, 1H), 7.56 (dd, J = 8.0, 2.0 Hz, 1H), 7.14 (d, J = 8.0 Hz, 1H), 4.75 (q, J = 6.4 Hz, 1H), 4.57 (ABq, 2H), 3.36 (s, 3H), 2.53 (s, 3H), 1.48 (d, J = 6.6 Hz, 3H).[0264] (c) Synthesis of 2-[5-(l-methoxymethoxy-ethyl)-pyridin-2-yl]-ethanol (5a):A mixture of compound 4a (7.0 g, 0.0386 mol) and 37% formaldehyde solution (5.8 g, 0.077 mol) was heated to 160°C in a sealed glass tube for 5 h. The reaction mixture was cooled to room temperature and concentrated under reduced pressure to afford a crude compound which was purified by flash column chromatography (1% methanol/dichloromethane) to afford compound 5 (1.2 g; 17% yield) as pale yellow oil. ES-MS [M+l]+: 212.1. 1H NMR (400 MHz, CDC13): δ 8.42 (d, J = 2.0 Hz, 1H), 7.65 (dd, J = 8.0, 2.4 Hz, 1H), 7.25 (d, J = 8.0 Hz, 1H), 4.72 (q, J = 6.6 Hz, 1H), 4.65 (t, J = 5.6 Hz, 1H), 4.52 (ABq, 2H), 3.73 (m, 2H), 3.24 (s, 3H), 2.86 (t, J = 7.2 Hz, 2H), 1.49 (d, J = 6.4 Hz, 3H).[0265] The total yield for compound 5a from compound la was 8% molar.[0266] (d) Synthesis of 4-{2-[5-(l-methoxymethoxy-ethyl)-pyridin-2-yl]-ethoxy}- benzaldehyde (6a): Methanesulphonylchloride (1.19 g, 0.01 mol) was added, drop wise, to a cooled suspension of compound 5a (1.7 g, 0.008 mol) and triethylamine (1.79 ml, 0.013 mol) in dichloromefhane (20 mL) at 0°C and stirred at same temperature for 1 h. The reaction mixture was diluted with water (50 mL) and extracted with dichloromethane (3×50 mL). The combined organic extract was dried over anhydrous sodium sulphate and concentrated to afford 2-(5-(l-(methoxymethoxy)ethyl)pyridin-2-yl)ethyl methanesulfonate (2.04 g; 88% yield) as a yellow oil, which was taken to next step without purification. ES-MS [M+l]+: 290.[0267] 2-(5-(l-(methoxymethoxy)ethyl)pyridin-2-yl)ethyl methanesulfonate was added (2.3 g, 0.008 mol) to a stirred suspension of 4-hydroxybenzaldehyde (1.65 g, 0.0137 mol) and potassium carbonate (1.86 g, 0.0137 mol) in mixture of toluene (25 mL) and ethanol (25 mL); stirred at 85°C for 5 h. After consumption of the starting materials, the reaction mixture was diluted with water (30 mL) and extracted with ethyl acetate (2×100 mL). The combined organic extract was washed with water; dried over anhydrous sodium sulphate and concentrated to afford a crude dark yellow liquid. The crude was purified by flash column chromatography (1% methanol/dichloromethane) to afford compound 6a (1.5 g; 60% yield) as pale yellow liquid. ES-MS [M+l]+: 316.1.[0268] (e) Synthesis of 5-(4-{2-[5-(l-methoxymethoxy-ethyl)-pyridin-2-yl]-ethoxy}- benzylidene)-thiazolidine-2,4-dione (7a):Piperidine (80 mg, 0.95 mmol) was added to a solution of compound 6a (0.6 g, 1.9 mmol) and thiazolidine-2,4-dione (0.22 g, 1.9 mmol) in ethanol (15 mL) and the mixture was heated to reflux overnight. After 15 h, the reaction mixture was cooled to room temperature and concentrated under reduced pressure to afford crude mixture, which was purified by flash column chromatography (2% methanol/dichloromethane) to afford compound 7 (500 mg; 64% yield) as a yellow solid. ES-MS [M+l]+: 415.1. 1H NMR (400 MHz, DMSO-d6): δ 12.25 (br s, 1H), 8.47 (d, J = 2.0 Hz, 1H), 7.70 (dd, J = 8.0, 2.0 Hz, 1H), 7.54 (d, J = 8.8 Hz, 2H), 7.36 (d, J = 8.0 Hz, 1H), 7.21 (d, J = 8.8 Hz, 2H), 4.73 (m, 1H), 4.60-4.40 (m, 4H), 4.22 (t, J = 6.2 Hz, 1H), 3.24 (s, 3H), 3.20 (t, J = 6.8 Hz, 2H), 1.41 (d, J = 6.0 Hz, 3H).[0269] (f) Synthesis of 5-(4-{2-[5-(l-hydroxy-ethyl)-pyridin-2-yl]-ethoxy}-benzyl)- thiazolidine-2,4-dione (9a): [0270] A solution of sodium borohydride (115 mg, 3.017 mmol) in 0.2N sodium hydroxide(1.2 mL) was added slowly to a stirred solution of compound 7 (0.5 g, 1.207 mmol), dimethylglyoxime (42 mg, 0.36 mmol) and C0CI2.6H2O (23 mg, 0.096 mmol) in a mixture of water (6 mL): tetrahydrofurane (6 mL) and 1M sodium hydroxide (1 mL) solution at 10°C and after addition, the reaction mixture was stirred at room temperature. After 1 h, the reaction color lightened and additional quantities of sodium borohydride (46 mg, 1.207 mmol) and C0CI2.6H2O (22 mg, 0.096 mmol) were added and stirring was continued at room temperature. After 12 h, the reaction was neutralized with acetic acid (pH~7); diluted with water (10 mL) and extracted in ethyl acetate (3×50 mL). The combined organic extract was dried over anhydrous sodium sulphate and concentrated to afford crude compound 8a, 5-(4- (2-(5-(l-(methoxymethoxy)ethyl)pyridin-2-yl)ethoxy)benzyl)thiazolidine-2,4-dione, (0.4 g) as pale yellow semi solid, which was taken to next step without purification. ES-MS [M+l]+: 417.5.[0271] 2N HC1 (2 mL) was added to a solution of compound 8a (0.4 g, 0.96 mmol) in methanol (20 ml) and the mixture was heated to reflux. After 4 h, the reaction mixture was cooled to room temperature and then concentrated under reduced pressure to afford a residue which was dissolved in water and the solution was neutralized using sodium bicarbonate solution (sat). The resulting white precipitate was collected by filtration to afford compound 9a (250 mg; 56% yield over 2 steps) as an off-white solid. ES-MS [M+l]+: 373.4. 1H NMR (400 MHz, DMSO-de): δ 12.00 (br s, -NH), 8.46 (d, J = 2.0 Hz, 1H), 7.66 (dd, J = 8.0, 2.4 Hz, 1H), 7.30 (d, J = 8.0 Hz, 1H), 7.13 (d, J = 8.4 Hz, 2H), 6.86 (d, J = 8.4 Hz, 2H), 5.25 (d, J = 4.4 Hz, 1H), 4.86 (m, 1H), 4.75 (m, 1H), 4.30 (t, J = 6.8 Hz, 2H), 3.30 (m, 1H), 3.14 (t, J = 6.4 Hz, 2H), 3.04 (m, 1H), 1.34 (d, J = 6.4 Hz, 3H).[0272] The overall yield of compound 9a was 1.5% molar.EXAMPLE 2Synthesis of 2-(5-(l-((tert-butyldimethylsilyl)oxy)ethyl)pyridin-2-yl)ethan-l-ol[0273] The synthesis of 2-(5-(l-((tert-butyldimethylsilyl)oxy)ethyl)pyridin-2-yl)ethan-l-ol was conducted according to the Scheme 5 using the reagents and solvents listed in Table 1 below: Scheme 5TBDMS-CI OTBDMS 1 . n-BuLi, <-55°C OTBDMSImidazole
DMF

[0274] The 1H-NMR spectra were recorded with Agilent MercuryPlus 300 NMR spectrometer.[0275] LC-MS data were obtained on an Agilent 1290 series with UV detector and HP 6130MSD mass detector using as column Waters XB ridge BEH XP (2.1 x 50 mm; 2.5 μιτι) and as eluent Ammonium acetate (10 mM); Water/ Methanol/ Acetonitrile.[0276] (a) l-(6-bromopyridin-3-yl)ethan-l-ol (2)[0277] A 20 L vessel was placed under nitrogen atmosphere and charged with tetrahydrofuran (5.5 L) and 2,5-dibromopyridine (1) (2000 g, 8.44 mol, 1.0 eq) (OxChem Corporation). The mixture was cooled to -10°C and isopropyl magnesium chloride (20% in THF, 6.02 L, 11.82 mol, 1.4 eq) (Rockwood Lithium) was added slowly over 1 h, keeping the reaction temperature below 5°C. After addition, the cooling bath was removed and the temperature was kept below 30°C (some additional cooling was needed to achieve this) and the reaction mixture was stirred overnight. After 16 h, a sample was taken; quenched with saturated aqueous ammonium chloride and extracted with methyl tert-buty\ ether (TBME). The TBME was evaporated under vacuum. 1H-NMR in deuterated chloroform showed complete conversion.[0278] The reaction mixture was cooled to -15°C and a solution of acetaldehyde (472 g,10.72 mol, 1.27 eq) (Acros) in tetrahydrofuran (200 mL) was added dropwise, while keeping temperature below -10°C. After the addition was complete, the cooling bath was removed and the temperature was allowed to rise to maximum of 5-8°C. After 1.5 h, a sample was taken and the reaction was quenched with aqueous ammonium chloride as described above. 1H-NMR showed the reaction was complete.[0279] Two batches were combined for work up.[0280] The reaction mixture was quenched by pouring the mixture into a solution of aqueous ammonium chloride (1 kg in 5 L water) and stirred for 15 min, filtered over Celite and rinsed thoroughly with toluene. The filtrate was transferred to a separation funnel and the obtained two layers system was separated. The aqueous layer was extracted with toluene (2 L). The combined organic layers were dried over sodium sulfate and filtered. Evaporation of the filtrate to dryness under vacuum yielded 3.49 kg (99%) of the desired crude material. XH NMR (300 MHz, CDC13): δ 8.30 (d, J = 2.5 Hz, 1H), 7.59 (dd, J = 8.0, 2.5 Hz, 1H), 7.44 (d, J = 8.0 Hz, 1H), 4,91 (q, J = 6.5 Hz, 1H), 1.49 (d, J = 6.5 Hz, 3H).[0281] (b) 2-bromo-5-(l-((tert-butyldimethylsilyl)oxy)ethyl)pyridine (3)[0282] A 50 L reactor under nitrogen atmosphere was charged with compound 2 (10.0 kg, around 49.5 mol) and DMF (16 L). The mixture was cooled to 10°C and imidazole (6.74 kg, 99 mol, 2.0 eq) (Apollo Scientific Ltd.) was added portion wise within 30 min. The mixture was cooled to 0°C and TBDMS-Cl (7.46 kg, 49.5 mol, 1.0 eq) (Fluorochem) was added portion wise within 5 h, keeping the temperature below 3°C. The mixture reaction temperature was allowed to reach room temperature and stirred overnig ht. H NMR of a sample showed complete conversion.[0283] The reaction mixture was transferred to a 100 L extraction-vessel and the product was extracted with heptane (2×7.5 L, 10 L). The combined heptane-layers were washed with water (2×6 L, 3 L) to remove small amounts of DMF, dried over sodium sulfate and evaporated under vacuum to give crude compound 3 (15.5 kg, 49.0 mol) in a 99.0% yield. This crude product was purified by a short plug filtration, using 10 kg silica/heptane and eluted with heptane (approx. 50 L). The product-fractions were combined and evaporated under vacuum to give 12.0 kg of purified compound 3 (38 mol) as a brown oil in a 76.8% molar yield. (Average yield for 3 experiments was 78%). HPLC-MS: Rt= 2.6 min, M+l=316.1 and 318.1; 1H NMR (300 MHz, CDC13): δ 8.55 (d, J = 2.2 Hz, 1H), 7.54 (dd, J = 8.2, 2.2 Hz, 1H), 7.42 (d, J = 8.2 Hz, 1H), 4,86 (q, J = 6.5 Hz, 1H), 1.40 (d, J = 6.5 Hz, 3H), 0.88 (s, 9H), 0.02 (d, J = 26 Hz, 2x3H).[0284] (c) 2-(5-(l-((tert-butyldimethylsilyl)oxy)ethyl)pyridin-2-yl)ethan-l-ol (4)[0285] The ethylene oxide solution in diethylether was prepared in advance. Diethylether(1.2 L) in a 3 L three-necked flask was cooled at -65 °C and ethylene oxide (462.3 g, 10.5 mol, 1.06 eq) (Linde) was added and stirred at -70°C. Alternatively, the ethylene oxide solution can be made at about -20°C and then added gradually to the reaction mixture having a temperature at about -60°C. [0286] To a solution of 2-bromo-5-(l-((ieri-butyldimethylsilyl)oxy)ethyl)pyridine (3) (3.13 kg, 9.90 mol, 1.0 eq) in diethylether (7.5 L) cooled at -59°C, n-butyllithium (4 L, 10.0 mol, 2.5M in hexanes, 1.01 eq) (Aldrich Chemistry) was added while keeping temperature between -58°C and -62°C. After addition, the mixture was stirred for 1 h while keeping temperature between -60°C and -68°C. The upfront prepared ethylene oxide solution was added at once to the reaction mixture, while temperature was around -62°C. Subsequently, copper(I) iodide (962.3 g, 5.05 mol, 0.51 eq) (Acros Organics) was added in portions of 120 g, every 10 min, keeping the temperature between -61°C and -63°C. Stirring was continued for 1 h after addition keeping temperature between -61°C and -63°C. The cooling bath was removed and allowing the temperature to rise to about 15°C and further to 25 °C with a water bath overnight.[0287] Workup: The reaction-mixture was poured into a solution of 1 kg ammonium- chloride in 5 L water and stirred for 30 min, then the layers were separated. The organic layer was washed with aqueous ammonium hydroxide (10%, 2.5 L, 4x) to remove Cu-complex (blue color disappeared). The combined organic layers were dried over sodium sulfate and evaporated to give 3.12 kg (max. 9.90 mol) crude compound 4 as a brown oil. The crude compound was purified over 20 kg silica (heptane/EtOAc) by eluting with 80 L heptane/EtOAc, 20 L EtOAc, 25 L EtOAc/MeOH 95/5, 25 L EtOAc/MeOH 9/1 and 10 L EtOAc/MeOH 8/2, to give 1.47 kg of purified compound 4 (5.22 mol) as a brown oil (with tendency to solidify) in a 52.7% average molar yield (HPLC-purity of 99.5%). (Average yield over 12 experiments 52%). HPLC-MS: Rt= 2.3 min, M+l=282.1; 1H NMR (300 MHz, CDC13): δ 8.42 (d, J = 2.1 Hz, 1H), 7.61 (dd, J = 8.3, 2.1 Hz, 1H), 7.11 (d, J = 8.3 Hz, 1H), 4,88 (q, J = 7.0 Hz, 1H), 4.01 (t, J=6.0 Hz, 2 H), 3.00 (t, J=6.0 Hz, 2 H), 1.41 (d, J =7.0 Hz, 3H), 0.90 (s, 9H), 0.02 (d, J = 26 Hz, 2x3H).[0288] Another 2.5% of the product was isolated by re -purifying impure product fraction.The total yield of compound 4 from compound 1 was 39.6% molar.EXAMPLE 3Synthesis of 5-[[4-[2-[5-(l-hydroxyethyl)-2-pyridinyl]ethoxy]phenyl]methyl]- 2,4-thiazolidinedione hydrochloride (9) 2. Sodium bisulfiteethanol/water mixture
3. Addition 10% aqueous sodium hydroxide solution

until pH 12

from step e dimethylglyoxime7step g step f

step h[0289] The 1H-NMR spectra were recorded with a 400 MHz Avance Bruker NMR spectrometer. LC-MS data were obtained on a Agilent Technologies 6130 Quadrapole LC/MS using as column Agilent XDB-C18 and as eluent 0.1% formic acid (aq) and 0.05% formic acid in acetonitrile.[0290] Steps d and e: Synthesis of 4-[2-[5-[[[(l,l-dimethylethyl)dimethylsilyl]oxy]ethyl]-2- pyridinyl] ethoxy] -benzaldehyde (6)[0291] To a well stirred solution of 5-[[[(l,l-dimethylethyl)dimethylsilyl]-oxy]ethyl]-2- pyridineethanol (4) (obtained as described in Example 2) (1.91 kg) in toluene (8.6 L) at 5°C were added sodium hydroxide (30% aqueous, 2.79 L) and tetrabutylammonium bromide (7.2 g). p-Toluenesulfonyl chloride (1.62 kg) was next added in portions during 5 min. After the addition, the reaction mixture was allowed to reach room temperature in 0.5 h and stirred at this temperature for 18 h. Water (7.3 L) was then added and the mixture was mixed well. Once the solids were dissolved, the layers were allowed to settle and the organic layer was separated. This organic phase was washed with water (5.7 L, 2x), followed by washing with a solution of sodium chloride (57 g) in water (5.7 L). The solvents were concentrated at reduced pressure to an amount of 2.5 kg of a brown oil (compound 5).[0292] To this well stirred brown oil were added subsequently ethanol (7.8 L), water (0.86L), 4-hydroxybenzaldehyde (0.88 kg) and potassium carbonate (1.17 kg) and then the mixture was heated at 75 °C for 18 h. Then, the solvent was evaporated while adding toluene (7.7 L) during 6 h and then the reaction mixture was allowed to cool. At 30°C, water (7.6 L) was added, stirred until all solids were dissolved and the mixture was cooled to room temperature. The layers were allowed to settle and separated. The organic layer was washed with water (7.6 L). The first aqueous extract was extracted with toluene (2.8 L) and this organic extract was used to also extract the aqueous washing. The organic extracts were combined and concentrated under vacuum to give 3.49 kg of a black oil (crude title compound 6).[0293] 1.73 kg of this black oil was dissolved in ethanol (0.74 L) and added to a well stirred solution of sodium bisulfite (1.36 kg) in a mixture of water (3.27 L) and ethanol (0.74 L). The container of the black oil was rinsed with ethanol (0.37 L) twice and these two rinses were also added to the bisulfite reaction mixture. After 75 min, heptane (5.3 L) was added, well mixed for 5 min, and the layers were allowed to settle and separated. To the organic layer was added a solution of sodium bisulfite (0.55 kg) in water (2.65 L), and ethanol (1.06 L). After stirring for 30 min, the layers were allowed to settle and separated. The two bisulfide aqueous extracts were combined and flasks rinsed with water (2.12 L). Next, toluene (4.5 L) and heptane (4.5 L) were added, the mixture was well stirred and the pH was adjusted to 12 using sodium hydroxide (10% aq) (temperature became 32°C). After stirring for an additional 5 min, the layers were allowed to settle and separated at 30°C. The aqueous layer was extracted with a mixture of toluene (1.5 L) and heptane (3.0 L). The layers were separated and the organic layers were combined. The combined organic layers were washed with water (5 L, 2x) and concentrated under vacuum to give the purified title compound 6. This procedure was repeated with another 1.73 kg of the black oil (crude title compound 6) to give in total 2.77 kg of 4-[2-[5-[[[(l,l-dimethylethyl)dimethylsilyl]oxy]ethyl]-2- pyridinyl]ethoxy]-benzaldehyde (6) as brown oil which contained 24% m/m of toluene according to 1H NMR (yield = 80%, calculated from compound 4 and corrected for residual toluene). [0294] 1H NMR (CDC13) δ: 0.00 (s, 3H), 0.09 (s, 3H), 0.91 (s, 9H), 1.44 (d, = 6 Hz, 3H),3.30 (t, = 7 Hz, 2H), 4.47 (t, = 7 Hz, 2H), 4.92 (q, = 6 Hz, 1H), 6.99 – 7.30 (m, 3H), 7.62- 7.67 (m, 1H), 7.80 – 7.85 (m, 2H), 8.5- 8.54 (m, 1H) and 9.88 (s, 1H).[0295] LC-MS; rt 7.5 min: ES: M+ 387, 386.[0296] Step f: Synthesis of (5Z)-5-[[4-[2-[5-[[[(l,l-dimethylethyl)dimethylsilyl]oxy]ethyl]-2-pyridinyl]ethoxy]phenyl]methylene]-2,4-thiazolidinedione (7)[0297] A solution of 4-[2-[5-[[[(l,l-dimethylethyl)dimethylsilyl]oxy]ethyl]-2-pyridinyl]- ethoxy]-benzaldehyde (6) (2.75 kg, containing 24% m/m of toluene) and piperidine (6.0 g) in methanol (3.16 L) was concentrated at 40°C under reduced pressure. The residue was dissolved in methanol (10.4 L) and 2,4-thiazolidinedione (759 g) and piperidine (230 g) were added. The mixture was heated at 47°C. After 25 h, the reaction mixture was allowed to cool to room temperature. The mixture was kept at pH 5-6 by adjusting it with acetic acid, if necessary. After a night at room temperature, water (1.56 L) was added and the suspension was stirred at room temperature for additional 2 h. The solids were isolated by filtration, washed with methanol (1 L, 2x) and dried under vacuum to give crude compound 7 (1.65 kg). The crude compound was mixed with methanol (10 L) and dichloromethane (8.6 L) and heated at 32°C until all solids dissolved. Then, the solvents were removed by distillation until the temperature of the mixture reached 34°C at a pressure of 333 mbar. Then, it was allowed to cool to room temperature overnight and stirred at 2°C for additional 2 h. The solids were isolated by filtration, washed with methanol (0.5 L, 2x) and dried under vacuum to give title compound 7 (1.50 kg) (yield = 61%).[0298] 1H NMR (CDCI3) δ 0.00 (s, 3H), 0.08 (s, 3H), 0.90 (s, 9H), 1.43 (d, = 6 Hz, 3H),3.32 (t, = 7 Hz, 2H), 4.48 (t, = 7 Hz, 2H), 4.92 (q, = 6 Hz, 1H), 6.95 – 7.00 (m, 2H), 7.24 – 7,28 (m, 1H), 7.38 – 7.42 (m, 2H), 7.67 (s, 1H), 7.69 – 7.73 (m, 1H) and 8.48 (d, = 3 Hz, 1H).[0299] LC-MS; rt 7.5 min: ES: M+ 487, 486, 485.[0300] Step g: Synthesis of 5-[[4-[2-[5-[[[(l,l-dimethylethyl)dimethylsilyl]oxy]ethyl]-2- pyridinyl]ethoxy]phenyl]methyl]-2,4-thiazolidinedione (8)[0301] To a stirred suspension of (5Z)-5-[[4-[2-[5-[[[(l,l-dimethylethyl)dimethylsilyl]oxy]- ethyl]-2-pyridinyl]ethoxy]phenyl]methylene]-2,4-thiazolidinedione (7) (10 g) in THF (10 mL) and sodium hydroxide (IN aq, 21 mL) was added of a solution of cobalt chloride (26 mg) and of dimethylglyoxime (930 mg) in THF (2.3 mL) and water (1.0 mL). Then the suspension was put under a nitrogen atmosphere by applying the sequence of vacuum and flushing with nitrogen (4x). Thereafter, the suspension was heated to 30°C. Then, a stock solution of sodium borohydride was prepared by dissolving sodium borohydride (2.7 g) in a mixture of water (15.8 mL) and a solution of sodium hydroxide (1 N aq, 3.5 mL), which was put under a nitrogen atmosphere by applying a sequence of vacuum and flushing with nitrogen (3x). This was added to the suspension of compound 7 at a rate of 4.5 mL/h. Simultaneously, nitrogen gas-saturated acetic acid was added to the suspension at a rate of 0.7 mL/h to maintain a pH of 10.0-10.5. After 1 h 30 min the rate of addition of the sodium borohydride solution and acetic acid were both reduced by half. Next, 3 h 45 min after start of addition, the addition of sodium borohydride and acetic acid were stopped. The mixture was allowed to cool down to room temperature and acetone (2.5 mL) was added over a period of 1 minute. After stirring the reaction mixture for 15 min acetic acid was added until the pH was 5.5-6.0 (about 3 mL required). Next, a mixture of ethyl acetate/toluene (1/3 v/v, 30 mL) was added, well mixed and layers were allowed to settle. The aqueous layer was separated and washed with ethyl acetate/toluene (1/3 v/v, 10 mL). Both organic extracts were pooled and water (40 mL) was added, well mixed and layers were allowed to settle. The pH of the aqueous layer was adjusted to 5.5-6 using saturated sodium hydrogen carbonate solution (aq) and again mixed with the organic layer. Layers were allowed to settle and the organic layer was separated and concentrated under vacuum to give 11.09 g of yellow oil (crude mixture containing title compound 8 and its borane complex). Several batches were combined for work up.33.1 g of the crude mixture containing title compound 8 and its borane complex (not corrected for residual solvents) was dissolved in toluene (30 mL) and filtered. The filtrate was submitted to column chromatography (silica gel, gradient of toluene to toluene/ethyl acetate 1/1) to give 30.0 g of mixture of 5-[[4-[2-[5-[[[(l,l- dimethylethyl)dimethylsilyl]oxy]ethyl]-2-pyridinyl]ethoxy]phenyl]methyl]-2,4- thiazolidinedione (8) and its borane complex as a slightly yellow oil (yield = 100% from compound 4, not corrected for residual solvents). [0303] 1H NMR (CDC13) δ: -0.03 – 0.10 (m, 6H), 0.87 – 0.93 (m, 9H), 1.42 (d, / = 6 Hz, 3H),3.05-3.71 (m, 4H), 4.30 – 4.51 (m, 3H), 4.87 – 4.94 (m, 1H), 6.82 – 6.88 (m, 2H), 7.10-7.92 (m, 5H), 8.49 (d, / = 3 Hz, 0.6H) and 8.72 (brs, 0.4H).[0304] LC-MS; rt 6.8 min: ES: M+ 489, 488, 487, M“ 487, 486, 485; rt 8.1 min: ES M“ 501,500, 499, 498, 485.[0305] Step h: Synthesis of 5-[[4-[2-[5-(l-hydroxyethyl)-2-pyridinyl]ethoxy]phenyl]- methyl]-2,4-thiazolidinedione hydrochloride (9)[0306] To a stirred solution of the mixture of (5-[[4-[2-[5-[[[(l,l-dimethylethyl)- dimethylsilyl]oxy]ethyl]-2-pyridinyl]ethoxy]phenyl]methyl]-2,4-thiazolidinedione and its borane complex (8) (5.17 g) in methanol (25.2 mL) at 22°C was added hydrochloric acid (30%, 2.75 mL) in about 5 min to give a temperature rise to 28°C. This solution was heated to 40 °C. Three hours after addition, the 11 g of volatiles were removed under reduced pressure. Then, acetonitrile (40.3 mL) was added and the mixture was heated at reflux for 0.5 h. Next, the suspension was allowed to cool down to room temperature and stirred for 1 h at room temperature. Solids were isolated by filtration, washed with a mixture of acetonitrile/water (20/1 v/v, 10 mL) and with acetonitrile (10 mL) and dried under vacuum at 40 °C to give 4.00 g of white solids (crude 9) (yield = 77%, not corrected for residual solvents).[0307] Purification of 5-[[4-[2-[5-(l-hydroxyethyl)-2-pyridinyl]ethoxy]phenyl]methyl]-2,4- thiazolidinedione hydrochloride (9):[0308] The crude mixture of 5-[[4-[2-[5-(l-hydroxyethyl)-2-pyridinyl]ethoxy]phenyl]- methyl]-2,4-thiazolidinedione hydrochloride (3.95 g, crude 9) was dissolved in methanol/water (7/2 v/v, 80 mL) by heating it to 49°C. To this solution was added washed norit (obtained by heating a suspension of norit (6 g) in methanol/water (7/2 v/v, 90 mL) at 45°C for 1 h, then isolating the norit by filtration and washing it twice with methanol/water (7/2 v/v, 30 mL) and drying it under vacuum at 40°C). Equipment was rinsed with methanol/water (7/2 v/v, 18 mL). After 0.5 h of stirring at 46°C, the warm suspension was filtered to remove the norit and filter was washed twice with methanol/water (7/2 v/v, 18 mL). The filtrate was concentrated under vacuum at a bath temperature of 60°C to a mass of 11.8 g (1 v of compound and 2 v of water). To the suspension was added butanone (19.7 mL, 5 v) and the mixture was heated at a bath temperature of 95°C. Under distillation at a constant volume, butanone (95 mL) was added. Next, heating was stopped and the suspension was allowed to reach room temperature in about 0.5 h. Subsequently it was stirred for 0.75 h at room temperature. The solids were isolated by filtration, washed with a mixture of butanone/water (95/5 v/v, 18 mL) and butanone (18 mL) and dried under vacuum at 40°C to give 3.57 g of compound 9 as white solids (yield = 91%).[0309] 1H NMR (DMSO-de): δ 12.00 (br s, -NH), 8.71 (d, = 2.0 Hz, 1H), 8.45 (dd, = 8.3,1.7 Hz, 1H), 7.98 (d, = 8.3 Hz, 1H), 7.15 (d, = 8.7 Hz, 2H), 6.88 (d, = 8.7 Hz, 2H), 5.57 (s, OH), 4.95 (q, = 6.5 Hz, 1H), 4.86 (dd, = 8.9, 4.4 Hz, 1H), 4.40 (t, = 6.3 Hz, 2H), 3.49 (t, = 6.2 Hz, 2H), 3.29 (dd, = 14.2, 4.4 Hz, 1H), 3.06 (dd, = 14.2, 9.0 Hz, 1H), 1.41 (d, = 6.5 Hz, 3H).[0310] LC-MS; rt 3.5 min: ES: M+ 374, 373, M“ 372, 371.EXAMPLE 4Conditions tested in the preparation of compound 5 in the Step d[0311] The conditions described in Table 2 below were tested in the step d in the preparation of compound 5 from compound 4 providing a good yield of compound 5:Table 2Entry Reaction Conditions Amount of p-Ts-Cl / Eq1 Toluene/water/Bu4NBr/NaOH 1.052 1.083 1.074 1.07+0.035 1.076 Et3N / DCM 1.187 1.408 Pyridine / DCM 1.40 EXAMPLE 5Conditions tested in the preparation of compound 6 in the Step e[0312] The conditions described in Table 3 below were tested in the step e in the preparation of compound 6 from compound 5 providing a good yield of compound 6:Table 3

PATENT
Compound 1 is administered to the subject. The structure of 5-[[4-[2-[5-(l -hydroxy ethyljpyri din-2 – yl]ethoxy]phenyl]methyl]-l,3-thiazolidine-2,4-dione is:
[0047] The present disclosure encompasses the use of stereoisomers of 5-[[4-[2-[5-(l- hydroxyethyl)pyridin-2-yl]ethoxy]phenyl]methyl]-l,3-thiazolidine-2,4-dione. 5-[[4-[2-[5- (l-hydroxyethyl)pyridin-2-yl]ethoxy]phenyl]methyl]-l,3-thiazolidine-2,4-dione has two asymmetric centers and thus four stereoisomers are possible as follows:
//////////LERIGLITAZONE, MIN 102 , лериглитазон , ليريغليتازون , 乐立格列酮 , Hydroxy Pioglitazone, M-IV, PHASE 2
CC(C1=CN=C(C=C1)CCOC2=CC=C(C=C2)CC3C(=O)NC(=O)S3)O

NEW DRUG APPROVALS
ONE TIME
$10.00

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO