MHLW Japan, GlaxoSmithKline and Genmab announced the approval of Arzerra (ofatumumab) by the MHLW for use in patients with relapsed/refractory CD20-positive chronic lymphocytic leukaemia.
mar 26, 2013
GlaxoSmithKline and Genmab announced the approval of Arzerra (ofatumumab) by the MHLW for use in patients with relapsed/refractory CD20-positive chronic lymphocytic leukaemia.
The thumbs-up triggers a milestone payment of 20 million Danish kroner to Genmab.
Ofatumumab(trade name Arzerra, also known as HuMax-CD20) is a human monoclonal antibody (for the CD20 protein) which appears to inhibit early-stage B lymphocyte activation. It is FDA approved for treating chronic lymphocytic leukemia that is refractory to fludarabine and alemtuzumab (Campath) and has also shown potential in treating Follicular non-Hodgkin’s lymphoma, Diffuse large B cell lymphoma, rheumatoid arthritis and relapsing remitting multiple sclerosis. Ofatumumab has also received conditional approval in Europe for the treatment of refractory chronic lymphocytic leukemia. This makes ofatumumab the first marketing application for an antibody produced by Genmab, as well as the first human monoclonal antibody which targets the CD20 molecule that will be available for patients with refractory CLL.
MHLW JAPAN has approved Nippon Shinyaku Co’s Regtect (acamprosate) for support maintenance of abstinence in patients with alcohol dependence

ACAMPROSATE
MAR 26 2013
MHLW JAPAN has approved Nippon Shinyaku Co’s Regtect (acamprosate) for support maintenance of abstinence in patients with alcohol dependence.
Acamprosate, also known as N-acetyl homotaurine and by the brand name Campral, is a drug used for treating alcohol dependence.
Acamprosate is thought to stabilize the chemical balance in the brain that would otherwise be disrupted by alcoholism, possibly by antagonizing glutamatergic N-methyl-D-aspartate receptors and agonizing gamma-aminobutyric acid (GABA) type A receptors. Reports indicate that acamprosate only works with a combination of attending support groups and abstinence from alcohol.Certain serious side effects include diarrhea, allergic reactions, irregular heartbeats, and low or high blood pressure, while less serious side effects include headaches, insomnia, and impotence. Acamprosate should not be taken by people with kidney problems or allergies to the drug.
Campral is manufactured and marketed in the United States by Forest Laboratories, while Merck KGaA markets it outside the US. It is sold as 333 mg white and odorless tablets of acamprosate calcium, which is the equivalent of 300 mg of acamprosate.
MAA, EU, Boehringer Ingelheim and Eli Lilly and Company announce acceptance of EMA application for investigational Type 2 Diabetes treatment empagliflozin

Empagliflozin
26 March 2013
Boehringer Ingelheim and Eli Lilly and Company today announced the European Medicines Agency (EMA) has accepted for review a marketing authorisation application (MAA) for the investigational sodium glucose cotransporter-2 (SGLT2) inhibitor, empagliflozin, for the treatment of Type 2 Diabetes (T2D) in adults. The acceptance of the MAA marks the beginning of the review process in the European Union for this potential oral diabetes treatment.
A New Drug Application (NDA) for empagliflozin was recently submitted to the Food and Drug Administration (FDA) in the United States for the treatment of Type 2 Diabetes mellitus (T2D) in adults.
Empagliflozin is part of a class of drugs being investigated for the reduction of blood glucose levels in adults with T2D. In clinical trials to date, SGLT2 inhibitors have been shown to reduce blood glucose by removing excess glucose independently of beta cell function and insulin resistance.
* Empagliflozin is an investigational compound. Its safety and efficacy have not yet been fully established
About Diabetes
An estimated 371 million people worldwide have Type 1 and Type 2 Diabetes. Type 2 Diabetes is the most common type, accounting for an estimated 90% of all diabetes cases.Diabetes is a chronic condition that occurs when the body either does not properly produce, or use, the hormone insulin.
Empagliflozin is drug which is being investigated in clinical trials for the oral treatment of type 2 diabetes by Boehringer Ingelheim and Eli Lilly and Company.[1][2] It is an inhibitor of the sodium glucose co-transporter-2 (SGLT-2), which is found almost exclusively in the proximal tubules of nephronic components in the kidneys. SGLT-2 accounts for about 90 percent of glucose reabsorption into the blood. Blocking SGLT-2 causes blood glucose to be eliminated through the urine via the urethra.[3][4]
SGLT-2 inhibitors such as empagliflozin reduce blood glucose by blocking glucose reabsorption in the kidney and thereby excreting glucose (i.e., blood sugar) via the urine.[5]
- Grempler R, Thomas L, Eckhardt M, Himmelsbach F, Sauer A, Sharp DE, Bakker RA, Mark M, Klein T, Eickelmann P (January 2012). “Empagliflozin, a novel selective sodium glucose cotransporter-2 (SGLT-2) inhibitor: characterisation and comparison with other SGLT-2 inhibitors”. Diabetes Obes Metab 14 (1): 83–90. doi:10.1111/j.1463-1326.2011.01517.x. PMID 21985634.
- “Empagliflozin”. clinicaltrials.gov. U.S. National Institutes of Health. Retrieved 22 September 2012.
- Nair S, Wilding JP (January 2010). “Sodium glucose cotransporter 2 inhibitors as a new treatment for diabetes mellitus”. J. Clin. Endocrinol. Metab. 95 (1): 34–42. doi:10.1210/jc.2009-0473. PMID 19892839.
- Bays H (March 2009). “From victim to ally: the kidney as an emerging target for the treatment of diabetes mellitus”. Curr Med Res Opin 25 (3): 671–81. doi:10.1185/03007990802710422. PMID 19232040.
- Abdul-Ghani MA, DeFronzo RA (September 2008). “Inhibition of renal glucose reabsorption: a novel strategy for achieving glucose control in type 2 diabetes mellitus”. Endocr Pract 14 (6): 782–90. PMID 18996802.
Kyowa Hakko Kirin, has received approval for NOURIAST tablets 20 mg (istradefylline), a novel antiparkinsonian agent, has been approved for manufacturing and marketing in Japan

Istradefylline (KW-6002) is a selective antagonist at the A2A receptor. It has been found to be useful in the treatment of Parkinson’s disease.[1] Istradefylline reduces dyskinesia resulting from long-term treatment with classical antiparkinson drugs such as levodopa. Istradefylline is an analog of caffeine.
References
Peter A. LeWitt, MD, M. Guttman, James W. Tetrud, MD, Paul J. Tuite, MD, Akihisa Mori, PhD, Philip Chaikin, PharmD, MD, Neil M. Sussman, MD (2008). “Adenosine A2A receptor antagonist istradefylline (KW-6002) reduces off time in Parkinson’s disease: A double-blind, randomized, multicenter clinical trial (6002-US-005)”. Annals of Neurology 63 (3): 295–302. doi:10.1002/ana.21315. PMID 18306243.
TUE 26 MAR 2013
Japan, Kyorin, Pentasa, mesalazine suppositories launched
PENTASA (mesalamine) for oral administration is a controlled-release formulation of mesalamine, an amino-salicylate anti-inflammatory agent for gastrointestinal use. Chemically, mesalamine is 5-amino-2-hydroxybenzoic acid. It has a molecular weight of 153.14.
The structural formula is:

Cellular Biomedicine Group Marks the Launch of China Clinical Trial for TC-DC Therapy for Hepatocellular Carcinoma

March 21, 2013
Cellular Biomedicine Group announced that in the first week of March 2013, the company launched a clinical trial for TC-DC (Tumor Stem Cell Specific Dendritic Cell) therapy for hepatocellular carcinoma (HCC), the most common type of liver cancer. The clinical trial, which is already in progress, is the result of collaboration between CBMG, California Stem Cell (CSC) and Shanghai’s PLA 85 Hospital. It is the first immune cell clinical trial of its kind in China.
CBMG’s joint venture with CSC grants CBMG an exclusive license from CSC to develop and market CSC’s cancer (TC-DC) technology in Greater China. CBMG receives support from CSC’s California-based team of scientists and medical professionals, including CSC’s Dr. Hans Keirstead .
PLA 85 Hospital is a large general teaching hospital with 12 departments, located in Shanghai and has been granted Class A Hospital status at the Tertiary Level (the highest class). The hospital has over 600 beds with more than 200 professors and associate professors, including many well-known experts who are known for their pioneering work in the diagnosis and treatment of tumors. The principal Investigator of the trial and director of the Liver Disease Center of PLA 85 Hospital, ProfessorChengwei Chen , commented, “When I heard of the success this treatment had in clinical trials for other cancers in the U.S., I was very excited at the prospect of the hope it could bring to the millions of patients in China suffering from HCC. I am happy to lead this endeavor to help as many people as we can.” Said Dr. Steve Liu , Chairman of CBMG, “The launch of this trial is a major milestone for all of the physicians, scientists and other professionals at CBMG, CSC and PLA 85 Hospital who have contributed to this work.”
Multinational Contract Research Organization (CRO) CMIC-GCP has been contracted to manage the trial design and minimize delays.
Hepatocellular Carcinoma
Forty-five percent of the world’s HCC patients are in China, with over 300,000 new patients diagnosed every year. Currently the therapies commonly offered to most patients are surgery and local chemotherapy, with a 2-year recurrence rate of 51% and median survival time of 13 months.
CBMG’s research studies the effects of TC-DC (Tumor Stem Cell Specific Dendritic Cell) therapy. Dr. William Cao , President of CBMG said, “In simplified terms, TC-DC therapy takes a sample of the patient’s own dendritic, or immune cells and a sample of the patient’s tumor stem cells and places them together in the lab. The dendritic cell will learn the characteristics of the tumor stem cells, and is reintroduced to the patient’s body, where it can “train” the immune system to fight and destroy the tumor stem cells, which are the root cause of tumor recurrence and metastasis.”
About Cellular Biomedicine Group
Cellular Biomedicine Group, Inc. develops proprietary cell therapies for the treatment of certain degenerative diseases and cancers. Our developmental stem cell, progenitor cell, and immune cell projects are the result of research and development by scientists and doctors from China and the United States. Our flagship GMP facility, consisting of eight independent cell production lines, is designed, certified and managed according to U.S. standards. To learn more about CBMG, please visit:www.cellbiomedgroup.com
Daiichi Sankyo Receives Approval in Japan for Manufacture and Marketing of PRALIA ®
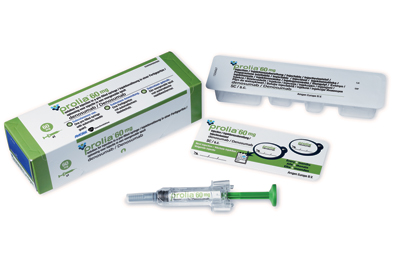
Daiichi Sankyo Receives Approval 25 mar 2013, in Japan for Manufacture and Marketing of PRALIA ® for the osteoporosis treatment PRALIA ®subcutaneous injection 60mg syringe (INN: Denosumab; genetic recombination) for the treatment of osteoporosis.
Denosumab[1] is a fully human monoclonal antibody for the treatment of osteoporosis, treatment-induced bone loss, bone metastases, rheumatoid arthritis, multiple myeloma, and giant cell tumor of bone.[2][3] It was developed by the biotechnology company Amgen.[4]
Denosumab is designed to inhibit RANKL (RANK ligand), a protein that acts as the primary signal for bone removal. In many bone loss conditions, RANKL overwhelms the body’s natural defenses against bone destruction.
In June 2010, denosumab was approved by the U.S. Food and Drug Administration (FDA) for use in postmenopausal women with risk of osteoporosis under the trade name Prolia,[5] and in November 2010, as Xgeva, for the prevention of skeleton-related events in patients with bone metastases from solid tumors.[6] Denosumab is the first RANKL inhibitor to be approved by the FDA.[7] In the summer of 2011 clinical trials were investigating denosumab in giant cell tumors, multiple myeloma with bone metastases, and hypercalcemia of malignancy, and further investigating its dosing and safety.[8]
- Pageau, Steven C. (2009). “Denosumab”. MAbs 1 (3): 210–215. doi:10.4161/mabs.1.3.8592. PMC 2726593.PMID 20065634.
- McClung, Michael R.; Lewiecki, E. Michael; Cohen, Stanley B.; Bolognese, Michael A.; Woodson, Grattan C.; Moffett, Alfred H.; Peacock, Munro; Miller, Paul D. et al. (2006). “Denosumab in Postmenopausal Women with Low Bone Mineral Density”. New England Journal of Medicine 354 (8): 821–31. doi:10.1056/NEJMoa044459. PMID 16495394.
- Ellis, G. K.; Bone, H. G.; Chlebowski, R.; Paul, D.; Spadafora, S.; Smith, J.; Fan, M.; Jun, S. (2008). “Randomized Trial of Denosumab in Patients Receiving Adjuvant Aromatase Inhibitors for Nonmetastatic Breast Cancer”. Journal of Clinical Oncology 26 (30): 4875–82. doi:10.1200/JCO.2008.16.3832.PMID 18725648.
- “Prolia (denosumab)”. Products. Amgen. Retrieved 6 May 2012.
- Matthew Perrone (June 2, 2010). “FDA clears Amgen’s bone-strengthening drug Prolia”. BioScience Technology.
- “Amgen’s Denosumab Cleared by FDA for Second Indication”. 19 Nov 2010.
- “FDA Approves Denosumab for Osteoporosis”. 2 June 2010.
- Russell S. Crawford, BPharm; Morgane C. Diven, PharmD; Laura Yarbro, PharmD (2011). “Denosumab: A Review of Its Pharmacology and Clinical Implications”. Contemporary Oncology 3 (1).
MHLW, sNDA, JAPAN, Daiichi Sankyo has received approval for Anticancer Agent irinotecan hydrochloride hydrate

Irinotecan (Camptosar, Pfizer; Campto, Yakult Honsha) is a drug used for the treatment of cancer.
Irinotecan prevents DNA from unwinding by inhibition of topoisomerase 1. In chemical terms, it is a semisynthetic analogue of the natural alkaloid camptothecin.
Its main use is in colon cancer, in particular, in combination with other chemotherapy agents. This includes the regimen FOLFIRI, which consists of infusional 5-fluorouracil,leucovorin, and irinotecan.
Irinotecan received accelerated approval by the U.S. Food and Drug Administration (FDA) in 1996[1] and full approval in 1998.[2] During development, it was known as CPT-11.
Japan, Shionogi Receives Marketing and Manufacturing Approval of a Drug for Lipodystrophy,METRELEPTIN for Subcutaneous Injection
Metreleptin, an analog of the human hormone leptin, is a unique potential therapy for certain metabolic disorders in patients with rare forms of inherited or acquired lipodystrophy. Lipodystrophy is a very rare condition characterized by loss of subcutaneous fat.
Metreleptin is being studied as a potential therapy for certain metabolic disorders in patients with inherited or acquired lipodystrophy. Metreleptin is believed to work by reducing fat accumulation in organs, caused by the disease, thereby improving insulin sensitivity. Clinical studies have been conducted by investigators at the National Institutes of Health (NIH) and other academic institutions in the US, Europe, and Japan to determine whether metreleptin can improve glycemic control and hypertriglyceridemia in patients with lipodystrophy.
In April 2012, Amylin completed its Biologics License Application (BLA) for metreleptin to treat diabetes and/or hypertriglyceridemia (high levels of triglycerides in the bloodstream) in patients with rare forms of lipodystrophy and requested Priority Review by the FDA.
If approved, metreleptin would be the first therapy indicated specifically for the treatment of diabetes and/or hypertriglyceridemia in patients with inherited or acquired lipodystrophy, and the first approved therapeutic use of a leptin analog.
About Lipodystrophy
Lipodystrophy is a life-threatening, “ultra orphan” rare disease that is estimated to impact a few thousand people worldwide, often with an early age of onset, for which there is a significant unmet medical need. There are currently no approved drugs that treat the underlying cause of the disease.
Fat tissue is a major endocrine organ producing important metabolic hormones such as leptin. People with lipodystrophy lack the required fat tissue for normal metabolic function. This can be partial, affecting select areas of the body, or generalized, affecting nearly the entire body. A lack of fat tissue can lead to relative deficiency of leptin.
Without adequate leptin function, the metabolic system, which regulates food intake and the storage and break-down of dietary fat and carbohydrates, falls out of balance. As a result, fat accumulates in the blood and organs such as liver and muscle, which can lead to life-threatening complications including insulin-resistant diabetes, hypertriglyceridemia (high levels of triglycerides in the bloodstream), acute pancreatitis, and hepatic steatosis or steatohepatitis, also known as fatty liver disease. There are no approved drugs that address the underlying relative leptin deficiency that is believed to contribute in large part to the metabolic abnormalities that occur in lipodystrophy. Currently available therapies for diabetes and hypertriglyceridemia are often rendered marginally effective due to the severity of the condition.
NOVARTIS TOBI Podhaler receives FDA approval for cystic fibrosis patients with Pseudomonas aeruginosa

Tobramycin
MAR 22.2013, FDA approves TOBI Podhaler to treat a type of bacterial lung infection in cystic fibrosispatients
The U.S. Food and Drug Administration today approved TOBI Podhaler (tobramycininhalation powder) for the management of cystic fibrosis patients with Pseudomonas aeruginosa, a bacterium that causes lung infections.
Cystic fibrosis is a genetic disease that affects about 30,000 pediatric and adult patients in the United States. Cystic fibrosis causes the body to produce thick, sticky mucus that builds up in the lungs and blocks airways. The buildup of mucus makes it easy for bacteria like P. aeruginosa to grow and cause a chronic lung infection that, over time, can severely damage the lungs. Many patients with cystic fibrosis are treated with antibiotics using a nebulizer machine.
Tobramycin is an aminoglycoside antibiotic derived from Streptomyces tenebrarius and used to treat various types of bacteria infections, particularly Gram-negative infections. It is especially effective against species of Pseudomonas.
TOBI Podhaler, a plastic, handheld inhaler device, contains a dry powder formulation of tobramycin, an antibiotic used to treat P. aeruginosa infection. The powder is inhaled twice daily using the Podhaler device for 28 days. Patients should then stop TOBI Podhaler therapy for 28 days before resuming again.
“Today’s approval broadens the available delivery mechanism options for patients withcystic fibrosis who require treatment for P. aeruginosa,” said Edward Cox, M.D., M.P.H, director of the Office of Antimicrobial Products in the FDA’s Center for Drug Evaluation and Research. “This product is the first dry powder antibacterial drug delivered with a handheld dry powder inhaler.”

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO




