Home » Uncategorized (Page 16)
Category Archives: Uncategorized
Sotuletinib HCl
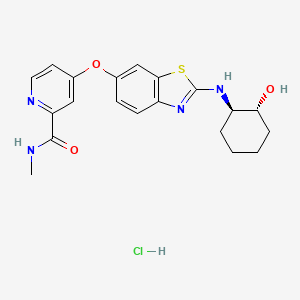

Sotuletinib HCl
CAS: 2222138-31-8 (HCl)
Chemical Formula: C20H23ClN4O3S
Molecular Weight: 434.939
7W3V82OQ0P
Synonym: Sotuletinib HCl; Sotuletinib hydrochloride, Sotuletinib monohydrochloride, BLZ945; BLZ 945; BLZ-945;
IUPAC/Chemical Name: 4-((2-(((1R,2R)-2-hydroxycyclohexyl)amino)benzo[d]thiazol-6-yl)oxy)-N-methylpicolinamide hydrochloride
| 2-PYRIDINECARBOXAMIDE, 4-((2-(((1R,2R)-2-HYDROXYCYCLOHEXYL)AMINO)-6-BENZOTHIAZOLYL)OXY)-N-METHYL- |
Sotuletinib, also known as BLZ945, is a potent and selective CSF-1R kinase inhibitor. BLZ945 showed effects of CSF1R inhibition on other tumor-infiltrating immune cells. BLZ945 attenuates the turnover rate of TAMs while increasing the number of CD8+ T cells that infiltrate cervical and breast carcinomas. BLZ945 decreases the growth of malignant cells in the mouse mammary tumor virus-driven polyomavirus middle T antigen (MMTV-PyMT) model of mammary carcinogenesis. BLZ945 prevents tumor progression in the keratin 14-expressing human papillomavirus type 16 (K14-HPV-16) transgenic model of cervical carcinogenesis.
Sotuletinib (BLZ945) is an experimental drug in development for the treatment of amyotrophic lateral sclerosis (ALS). It works as a colony-stimulating factor 1 (CSF1) receptor inhibitor.[1][2][3]
- OriginatorCelgene Corporation; Novartis
- ClassAmides; Amines; Antineoplastics; Benzothiazoles; Cyclohexanols; Ethers; Pyridines; Small molecules
- Mechanism of ActionMacrophage colony stimulating factor receptor antagonists
- Phase IIAmyotrophic lateral sclerosis
- Phase I/IISolid tumours
- 05 Dec 2022Novartis Pharmaceuticals terminates a phase I/II trials in Solid tumours (Combination therapy, Late-stage disease, Metastatic disease) in Taiwan, Japan, Israel (PO) in US, Israel, Italy, Japan, Singapore, Spain, Taiwan and Switzerland (EudraCT2015-005806-12) (NCT02829723)
- 14 Feb 2022Adverse events and pharmacodynamics data from preclinical macaque model study in brain disorders presented at the 29th Conference on Retroviruses and Opportunistic Infections
- 03 Dec 2020Chemical structure information added
An orally bioavailable inhibitor of colony stimulating factor 1 receptor (CSF-1R; CSF1R), with potential antineoplastic activity. CSF1R inhibitor BLZ945 selectively binds to CSF1R expressed on tumor-associated macrophages (TAMs), blocks the activity of CSF1R, and inhibits CSF1R-mediated signal transduction pathways. This inhibits the activity and proliferation of TAMs, and reprograms the immunosuppressive nature of existing TAMs. Altogether, this reduces TAM-mediated immune suppression in the tumor microenvironment, re-activates the immune system, and improves anti-tumor cell responses mediated by T-cells. CSF1R, also known as macrophage colony-stimulating factor receptor (M-CSFR) and CD115 (cluster of differentiation 115), is a cell-surface receptor for its ligand, colony stimulating factor 1 (CSF1); this receptor is overexpressed by TAMs in the tumor microenvironment, and plays a major role in both immune suppression and the induction of tumor cell proliferation.
Syn

WO2007/121484
WO2018069892
WO2017137958
PATENT
The free base and salts of the compound of formula (I) may be prepared for example, according to the procedures given in International Patent Application No. PCT/US2007/066898 filed on Apr. 18, 2007 and published as WO2007/121484 on Oct. 25, 2007. The compound of formula (I) has the chemical name: 4-(2-((1R,2R)-2-hydroxycyclohexylamino)benzothiazol-6-yloxy)-N-methylpicolinamide and is also known as BLZ945.








Step 1. Preparation of 4-(2-((lR,2R)-2-aminocyclohexylamino)benzo[d]thiazol-6-yloxy)-N-methylpicolinamide
To the solution of N-methyl-4-(2-(methylsulfinyl)benzo[d]thiazol-6-yloxy)picolinamide (15 mg, 43 μmole) in 400 μL of NMP was added (lR,2R)-cyclohexane-1,2-diamine (17 mg, 150 μmole). The reaction solution was stirred at 105°c for 24 hours. The crude reaction solution was purified on prep HPLC and evaporated in vaccuo to give 4-(2-((lR,2R)-2-aminocyclohexylamino)benzo[d]thiazol-6-yloxy)-N-methylpicolinamide (12 mg, 30 μmole) as white powder. ES/MS m/z 398.1(MH+).
PATENT
https://patents.google.com/patent/US20200093801A1
PATENT
CN116139135
PATENT
US20200190057
PATENT
CN110475555


AS ON DEC2021 3,491,869 VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@amcrasto
//////////
///////////
References
- ^ Pognan, François; Buono, Chiara; Couttet, Philippe; Galarneau, Jean-René; Timsit, Yoav; Wolf, Armin (29 October 2022). “Liver enzyme delayed clearance in rat treated by CSF1 receptor specific antagonist Sotuletinib”. Current Research in Toxicology. 3: 100091. doi:10.1016/j.crtox.2022.100091. ISSN 2666-027X.
- ^ Thongchot, Suyanee; Duangkaew, Supani; Yotchai, Wasan; Maungsomboon, Sorranart; Phimolsarnti, Rapin; Asavamongkolkul, Apichat; Thuwajit, Peti; Thuwajit, Chanitra; Chandhanayingyong, Chandhanarat (2 December 2022). “Novel CSF1R-positive tenosynovial giant cell tumor cell lines and their pexidartinib (PLX3397) and sotuletinib (BLZ945)-induced apoptosis”. Human Cell. 36 (1): 456–467. doi:10.1007/s13577-022-00823-0.
- ^ Martinez-Gonzalez, Loreto; Martinez, Ana (1 February 2023). “Emerging clinical investigational drugs for the treatment of amyotrophic lateral sclerosis”. Expert Opinion on Investigational Drugs. 32 (2): 141–160. doi:10.1080/13543784.2023.2178416.
////////Sotuletinib HCl, BLZ945, BLZ 945, BLZ-945,
O=C(NC)C1=NC=CC(OC2=CC=C3N=C(N[C@H]4[C@H](O)CCCC4)SC3=C2)=C1.[H]Cl
TACACICLIB, AUR-102, AURIGENE
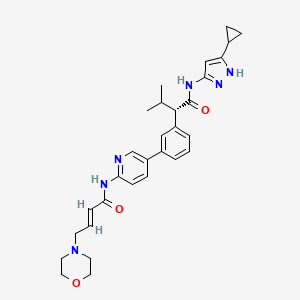
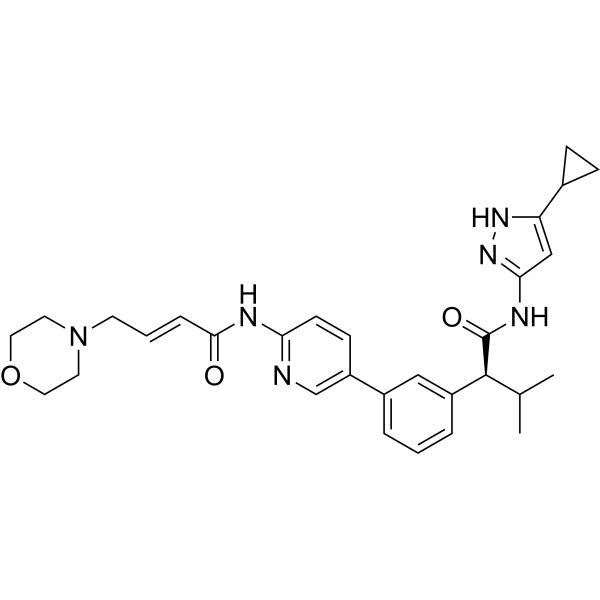
Tacaciclib
2768774-66-7
AUR-102
- Tacaciclib
- SCHEMBL24548621
- GTPL12880
- 528.6 g/mol
- C30H36N6O3
INN 12755
UNI D3G4JKK1MA
(2S)-N-(5-cyclopropyl-1H-pyrazol-3-yl)-3-methyl-2-[3-[6-[[(E)-4-morpholin-4-ylbut-2-enoyl]amino]pyridin-3-yl]phenyl]butanamide
(αS)-N-(5-Cyclopropyl-1H-pyrazol-3-yl)-α-(1-methylethyl)-3-[6-[[(2E)-4-(4-morpholinyl)-1-oxo-2-buten-1-yl]amino]-3-pyridinyl]benzeneacetamide
Benzeneacetamide, N-(5-cyclopropyl-1H-pyrazol-3-yl)-α-(1-methylethyl)-3-[6-[[(2E)-4-(4-morpholinyl)-1-oxo-2-buten-1-yl]amino]-3-pyridinyl]-, (αS)-
Tacaciclib is a CDK inhibitor, antineoplastic effect.
The present invention is directed to methods of preparation of compound of formula (I) that is useful for inhibiting Cyclin-dependent kinase 7 (CDK7) and for treating diseases or disorders mediated thereby.
CDK7, which complexes with cyclin H and RING-finger protein MAT1, phosphorylates the cell cycle CDKs in the activation of T-loop, to promote their activities (Fisher et al., Cell., Aug 26;78(4):713- 24, 1994). As such, it has been proposed that inhibiting CDK7 would provide a potent means of inhibiting cell cycle progression, which may be especially relevant given that there is compelling evidence from gene knockout studies in mice for lack of an absolute requirement for CDK2, CDK4 and CDK6 for the cell cycle at least in most cell types (M alumbres et al., Nature Cell Biology, 11, 1275 – 1276, 2009), whilst different tumors appear to require some, but they are independent of other interphase CDKs (CDK2, CDK4 , CDK6). Recent genetic and biochemical studies have confirmed the importance of CDK7 for cell cycle progression (Larochelle. et al., Mol Cell., Mar 23;25(6):839-50. 2007; Ganuza et al., EM BO J., May 30; 31(11): 2498-510, 2012).
Cyclin-dependent kinase 7 (CDK7) activates cell cycle CDKs and is a member of the general Transcription factor II Human (TFIIH). CDK7 also plays a role in transcription and possibly in DNA repair. The trimeric Cak complex CDK7/CyclinH/MATl is also a component of TFIIH, the general transcription/DNA repair factor IIH (Morgan, DO., Annu.Rev. Cell Dev. Biol. 13, 261-91, 1997). As a TFIIH subunit, CDK7 phosphorylates the CTD (Carboxy-Terminal-Domain) of the largest subunit of RNA polymerase II (pol II). The CTD of mammalian pol (II) consists of 52 heptad repeats with the consensus sequence 1 YSPTSPS 7 and the phosphorylation status of the Ser residues at positions 2 and 5 has been shown to be
important in the activation of RNAP-II indicating that it is likely to have a crucial role in the function of the CTD. CDK7, which primarily phosphorylates Ser-5 (PSS) of RNAP-II at the promoter as part of transcriptional initiation (Gomes et ah, Genes Dev. 2006 Mar 1; 20(5):601-12, 2006), in contrast with CDK9, which phosphorylates both Ser-2 and Ser-5 of the CTD heptad (Pinhero et al., Eur. J. Biochem., 271, pp. 1004-1014, 2004).
In addition to CDK7, other CDKs have been reported to phosphorylate and regulate RNA pol (II) CTD. The other CDKs include, Cdk9/ Cyclin T1 or T2 that constitute the active form of the positive transcription elongation factor (P-TEFb) (Peterlin and Price, Mol Cell., Aug 4; 23(3): 297-305,2006) and Cdkl2/Cyclin K and Cdkl3/Cyclin K as the latest members of RNAPII CTD kinases (Bartkowiak et al., Genes Dev., Oct 1 5;24(20):2303-16, 2010; Blazek et al., Genes Dev .Oct 15;25(20):2158-72, 2011).
Disruption of RNAP II CTD phosphorylation has been shown to preferentially effect proteins with short half-lives, including those of the anti-apoptotic BCL-2 family. (Konig et al., Blood, 1, 4307-4312, 1997; The transcriptional non-selective cyclin-dependent kinase inhibitor flavopiridol induces apoptosis in multiple myeloma cells through transcriptional repression and down-regulation of Mcl-1; (Gojoet al., Clin. Cancer Res. 8, 3527-3538, 2002).
This suggests that the CDK7 enzyme complexes are involved in multiple functions in the cell: cell cycle control, transcription regulation and DNA repair. It is surprising to find one kinase involved in such diverse cellular processes, some of which are even mutually exclusive. It also is puzzling that multiple attempts to find cell cycle dependent changes in CDK7 kinase activity remained unsuccessful. This is unexpected since activity and phosphorylation state of its substrate, CDC2, fluctuate during the cell cycle. In fact, it is shown that cdk7 activity is required for the activation of both Cdc2/Cyclin A and Cdc2/Cyclin B complexes, and for cell division. (Larochelle, S. et al. Genes Dev 12,370-81, 1998). Indeed, flavopiridol, a non-selective pan-CDK inhibitor that targets CTD kinases, has demonstrated efficacy for the treatment of chronic lymphocytic leukemia (CLL), but suffers from a poor toxicity profile (Lin et al.,). 27, 6012-6018, 2009; Christian et al., Clin. Lymphoma Myeloma, 9, Suppl.
3, S179-S185, 2009).
International publication WO2016193939, which is incorporated herein by reference for all purposes describes CDK7 inhibitors and processes for the preparation thereof. Inhibitors of CDK7 are currently being developed for the treatment of cancer. For drug development, it is typically advantageous to employ individual stereoisomers as they exhibit marked differences in pharmacodynamic, pharmacokinetic, and toxicological properties.
SYN
COUPLER

MAIN

Aurigene Discovery Technologies Ltd.
WO2022249141
WO2022130304
WO2022084930
WO2023224961
WO2023107861
WO2022249141
WO2022229835
WO2022130304
WO2022084930
PATENT
WO 2016/193939 COMPD 44
https://patents.google.com/patent/WO2016193939A1/en
InventorSusanta SamajdarRamulu PoddutooriChetan PanditSubhendu MUKHERJEERajeev Goswami
AURIGENE DISCOVERY TECHNOLOGIES LIMITED [IN]/[IN]
Inventors
- SAMAJDAR, Susanta
- PODDUTOORI, Ramulu
- PANDIT, Chetan
- MUKHERJEE, Subhendu
- GOSWAMI, Rajeev
PATENT
Applicants
- AURIGENE ONCOLOGY LIMITED [IN]/[IN]
Inventors
- PODDUTOORI, Ramulu
- VIJAYKUMAR BHAT, Uday
- THIMMASANDRA SEETHAPPA, Devaraja
WO2022229835
Example- 1: Preparation of compound of formula (I)
Scheme-1: Preparation of KRM-A


Step-4
KRM-A 4
Step-1: Preparation of 2-(3-bromophenyl)-3-methylbutanoic acid (1)
2M LDA (698 mL, 1.38mol) was added to a solution of 2-(3-bromophenyl) acetic acid (XA, 150 g, 0.69 mol) in THF (700mL) at -78 °C over a period of 30 min. The reaction mixture was stirred for 2h at -78 °C followed by a drop wise addition of isopropyl bromide (X B , 255 g, 2.07 mol) over a period of 30 min. The reaction mixture was stirred at room temperature overnight. Then, the reaction mixture was quenched with IN HC1 (pH 2) and the obtained product was extracted to ethyl acetate (500 mL x 3). The combined organic layer was washed with water followed by brine solution. The organic layer was dried over anhydrous sodium sulfate and concentrated under reduced pressure to afford the crude compound which was purified by silica column by eluting with 0-10% ethyl acetate-hexane system to afford the title compound (150 g, 83% yield) , HPLC purity-96%. The compound of formula (1) can also be prepared by the procedure described in CN 110590747.
Step-2: Preparation of Compound 3
2-(3-bromophenyl)-3-methylbutanoic acid (1, 510 g, 1.98 mol) was dissolved in 30% of IP A in water (10.2 L; 3.06 L of IPA-7.14 L of water) and ( 1L\ 2i ?)-cyclohexane-1,2-diamine (2, 113 g, 0.9 mol) was added. The reaction mixture was stirred at room temperature for 10 min until the precipitation was observed, then was heated to 100 °C until the solution became clear and stirred at the same temperature for another 30 min. The reaction mixture was allowed to slowly reach room temperature for 8-12h. The obtained solid was filtered and washed with 500 mL of 30% IPA-water mixture and dried under vacuum to afford the compound 3 (620 g, wet).
Work up (for Chiral purity): Small portion (100 mg) of compound 3 was taken in DCM (2-3 mL) and was added IN HC1 (pH 2) at 0 °C until the clear solution was observed. The compound was extracted into DCM, dried over NaiSCL and the solvent was evaporated to afford the title compound as white solid (20 mg). Chiral HPLC was recorded for this sample and 20.6% of undesired isomer was observed in chiral HPLC.
In order to improve the chiral purity of the title compound, the recrystallization method was performed as described below.
Step-3: Recrystallization
The compound 3 (619.90 g) was taken in 30% of IP A in water (12.4 L), then the mixture was heated to 100 °C until the solution became clear and was stirred at the same temperature for another 30min. The reaction mixture was allowed to reach room temperature slowly for 8-12h.
The obtained solid was filtered and washed with 500mL 30% IPA-water and dried under vacuum to afford a desired compound (360g, wet).
Work up for analysis (for Chiral purity): Small portion (100 mg) from above compound was taken in DCM (2-3mL), was added IN HC1 (pH 2) at 0 °C until the clear solution was observed and the compound was extracted to DCM, dried over NaiSCL and the solvent was evaporated to afford title compound as white solid (35 mg). Chiral HPLC was recorded for this sample and 10.3% of undesired isomer was observed in chiral HPLC.
The recrystallization method was repeated for three more times by using 30% of IPA in water as per the aforesaid procedure to obtain the purity of greater than 98.50% ee along with 0.27% other isomer to afford 286 g of compound 4.
Step-4: Preparation of (S)-2-(3-bromophenyl)-3-methylbutanoic acid (KRM-A)
The compound 4 (286 g) was taken in DCM (1.3 L), then was added IN HC1 at 0 °C until the clear solution was observed, and the compound was extracted to DCM (500 mL x 2). The organic layer was separated, washed with brine solution (500 mL) and dried over NaiSCL. The solvent was evaporated from the reaction mixture to afford title compound as white solid (148 g, 60% yield). Chiral HPLC: 98.50%
*H NMR (400MHz, DMSO-de): d 12.5 (s, 1H), 7.50-7.44 (m, 2H), 7.34-7.26 (m, 2H), 3.16 (d, 1H), 2.23-2.11 (m, 1H), 0.98 (d, 3H), 0.63 (d, 3H); Chiral HPLC: 98.50% retention time: 4,588 min.
Scheme-2: Preparation of compound of formula (I)


Step-1: Synthesis of (S)-2-(3-bromophenyl)-N-(5-cyclopropyl-1H-pyrazol-3-yl)-3-methylbutanamide
Step-la: Preparation ofKRM-D
To a stirred solution of KRM-A (lOOg, O.388mol) in dry DCM (600 mL, 6 vol), a catalytic amount of DMF (10 mL) was added followed by oxalyl chloride (45 mL, 0.525 mol) dropwise at 0°C over a period of 30 min. After completion of addition, the reaction mixture was stirred for 15 min at the same temperature. The reaction mixture was allowed to reach room temperature and stirred for 2 to 4h. After completion of the reaction (reaction was monitored by TLC, acid chloride formation was checked by quenching an aliquot of reaction mixture with MeOH), the reaction mixture was concentrated under vacuum at 40°C-45°C to afford crude (S)- 2-(3-bromophenyl)-3-methylbutanoyl chloride (KRM-D). The crude KRM-D was dissolved in toluene (500mL) and used for next step.
Step-lb: Preparation of compound of formula (II)
(5)-2-(3-bromophcnyl)-3-mcthylbutanoyl chloride in toluene was added slowly to a pre-cooled solution (0 to 5 °C) of ieri-butyl 3-amino-5-cyclopropyl-1H-pyrazole- l-carboxylate (KRM-B, 95.5g, 0.427 mol) and N, N-diisopropylethyl amine (100 mL, 0.583 mol) in toluene (1.2 L) at 0 °C for the period of l-2h. The reaction mixture was allowed to reach RT and stirred overnight. The reaction mixture was then cooled to 0-5°C and washed with ice-cold 1.5N HC1 (3 x 500 mL). The organic layer was washed with sodium bicarbonate solution (500 mL), brine solution (500 mL), dried over anhydrous NaiSCL , filtered and concentrated under vacuum at 45-50°C to afford crude tert-butyl (S)-5-( 2-(3-bromophenyl)-3-methylbutanamido)-3-
cyclopropyl- lH-pyrazole- 1-carboxylate (compound of formula (IG)) as light brown oil (~180g, LCMS: m/z= 461.9 (M+H) + , HPLC: 80.80%, retention time:15.89 min) . The crude product was taken as such for next step without further purification.
Step-1 c: Preparation of compound of formula (I)
To a suspension of tert-butyl (S)-5-(2-(3-bromophenyl)-3-methylbutanamido)-3-cyclopropyl-1H-pyrazole-1-carboxylate (180 g, 1,731 mol) in dioxane (360 mL ) was added 2N aqueous HC1 (360 mL) at 0 °C. The reaction mixture was stirred overnight at room temperature. After completion of the reaction, dioxane was concentrated, and the reaction mixture was diluted with water (500 mL) and basified with solid sodium bicarbonate (until pH-8). The obtained compound was extracted with DCM (700 mL x 3). The combined organic layers were washed with water (300 mL), brine solution (300 mL), and dried over anhydrous NaiSCL . The organic layer was concentrated to obtain a crude (S)-2-(3-bromophenyl)-N-(5-cyclopropyl-lH-pyrazol-3-yl)-3-methylbutanamide (Compound of formula (G)) as a semi-solid. The crude was dissolved in toluene (500 mL) and the solution was stirred for 18 h. The obtained solid was filtered and washed with toluene (100 mL) and n-heptane (200 mL). The solid was further dried under vacuum at 45-50°C for 6 h to afford a title compound (1 lOg, Yield: 78% over two steps). LCMS: m/z= 362 (M+H) + , HPLC: 97.66%, retention time: 24.10 min
Step-2: Preparation of (S, E)-N-(5-(3-(l-((5-cyclopropyl-lH-pyrazol-3-yl) amino)-3-methyl-l-oxobutan-2- yl) phenyl) pyridin-2-yl)-4-morpholinobut-2-enamide (Compound of formula (I))
To a degassed solution of (5)-2-(3-bromophcnyl)-N-(5-cyclopropyl-1 H-pyrazol-3-yl)-3-methylbutanamide (50 g, 0.138 mol) and (E)-4 -morpholino-N-(5-(4,4,5,5-tetramethyl-l,3,2-dioxaborolan-2-yl)pyridin-2-yl)but-2-enamide (KRM-C, 56.6 g, 0.151 mol, 1.1 eq) (prepared according to the procedure described in W02020202001) in 1,4-dioxane (500 mL, 10 vol) and water (100 mL, 2 vol) was added K3PO4 tribasic (73.2 g, 0.345 mol, 2.5 eq) at room temperature The reaction mass was stirred for 20 min with argon purging (degassing). Pd(dppf)Ch.DCM (3.38 g, 0.0042 mol, and 0.03eq) was added to the reaction mixture and the reaction mixture was heated to 90°C for 1-2 h (The reaction was monitored by TLC using 10% methanol in DCM as solvent system).
After completion of the reaction, the reaction mass was cooled to room temperature and filtered through Celite ® bed. The bed was washed with 1, 4-dioxane (200 mL) and the filtrate was concentrated to get crude compound. The crude compound was dissolved in 5% methanol in DCM (400 mL) and washed with water (200 mL x 2). The aqueous layer was separated and
extracted with DCM (100 mL x 2). The combined organic layer was washed with brine solution, filtered and dried over sodium sulfate. The organic layer was concentrated under vacuum at 35-40°C to obtain crude title compound (~80g).
The crude compound of formula (I), (80 g) was dissolved in 700 mL of ethyl acetate. The reaction mixture was cooled to 15°C and 2N HC1 was slowly added (until pH ~1). The reaction mixture was then stirred at room temperature for 20 min and the layers were separated. The aqueous layer (containing the product) was washed with ethyl acetate (300 mL x 3). The aqueous layer was cooled to 0°C and adjusted the pH to ~8 using 20% aqueous NaiCCL solution. The product was extracted with 10% methanol in DCM (300 mL x 3). The combined organic layer was washed with water (300 mL), dried over sodium sulfate and filtered. The filtrate was treated with activated charcoal (16 g, 20% w/w with respect to crude input of 80 g), then the reaction mixture was stirred overnight at room temperature and filtered through Celite ® bed. The bed was washed with 5% methanol in DCM (~ 20 vol, until absence of product by TLC). The filtrate was concentrated under vacuum at 35°C – 40°C to afford compound of formula (I) (70g, HPLC purity: 92.70%, retention time: 15.65 min).
Work-up for improved chiral purity: The above compound of formula (I) was dissolved in ethylacetate (~30 vol, 2L) and washed with aqueous citric acid (2 times, 400 mL x 1 and 200mL x 1), aqueous NaHCCL solution (2%, 500 mL x 1) and aqueous NaCl solution (10%, 500 mL x 1). The combined organic layer was dried over sodium sulfate and filtered. The filtrate was concentrated under vacuum at 35°C – 40°C to afford compound of formula (I) (~60g).
*H NMR (400MHz, DMSO-rfe): d: 10.79 (s, 1H), 10.46 (s, 1H), 8.61 (d, 1H), 8.28 (d, 1H), 8.07-8.05 (m, 1H), 7.69 (s, 1H), 7.56 (d, 1H), 7.39 (m, 2H), 6.84-6.77 (m, 1H), 6.62 (s, 2H), 6.51 (d, 1H), 6.13 (s, 1H) , 3.62-3.59 (m, 4H), 3.35 (d, 1H), 3.15-3.13 (m, 2H), 2.42-2.39 (m, 5H), 1.80-1.77 (m, 1H), 0.98 (d, 3H) , 0.88-0.85 (m, 2H), 0.67 (d, 3H), 0.62-0.60 (m, 2H); LCMS: m/z= 529.25-free base (M+H) + , HPLC: 98.98%, retention time: 15.40 min.
Patent
PATENT
In some embodiments, the compound of formula (I) is (E)-N-(5-(3-(l-((5-cyclopropyl-lH-pyrazol-3-yl)amino)-3-methyl-l-oxobutan-2-yl)phenyl)pyridin-2-yl)-4-morpholinobut-2-enamide or a pharmaceutically acceptable salt or a stereoisomer thereof (Compound 44).
Compound 44 is disclosed in WO 2016/193939 Al, published December 8, 2016, entitled “Substituted heterocyclyl derivatives as cdk inhibitors,” the entire contents of which are incorporated herein by reference. Compound 44A can be in the form of a fumaric acid salt or cocrystal as described in WO 2022/130304 Al, published June 23, 2022, entitled “Cocrystal of a cdk inhibitor,” the entire contents of which are incorporated herein by reference.
Example 3: Synthesis of Compounds 44A & 44B via Chiral Separation
Scheme-1


Step-1: Synthesis of 2-(3-bromophenyl)-3-methylbutanoic acid
[0352] 2M LDA (698 mL, 1.38mol) was added to a solution of 2-(3 -bromophenyl) acetic acid (reagent-1, 150g, 0.69mol) in THF (700mL) at -78 °C over a period of 30 min. The reaction mass was stirred for 2h at -78 °C followed by the drop wise addition of Isopropyl bromide (255 g, 2.07mol) over a period of 30 min at -78 °C. The reaction mass was stirred at room temperature for overnight. The reaction mass was quenched with IN HC1 (pH 2) and product extracted to ethyl acetate (500mL x 3). The combined organic layer washed with water followed by brine, dried and concentrated under reduced pressure to afford the title crude compound which was purified by silica column by eluting with 0-10% ethyl acetate -hexane system to afford the title compound 2 (150g, 83% yield). LCMS: m/z = 254.80 (M-2H)’
Step-2: Synthesis of tert-butyl 3-(2-(3-bromophenyl)-3-methylbutanamido)-5-cyclopropyl-lH-pyrazole-1 -carboxylate
[0353] 2-(3-bromophenyl)-3-methylbutanoic acid (intermediate-2, 70g, 0.0.27mol) was dissolved in dry DCM (500 mL) and added oxalyl chloride (68 mL, 0.78mol) dropwise at 0 °C followed by addition of catalytic amount of DMF (0.8mL) and maintained reaction mass at same temperature for 30min. The reaction mass was allowed to room temperature and stirred for 4h, distilled off the solvent and excess oxalyl chloride under vacuum. Re-dissolved the residue in DCM (250 mL) and added slowly to the cooled solution of tert-butyl 3 -amino-5 -cyclopropyl- 1H-pyrazole-1 -carboxylate (intermediate-3, 49g, 0.218mol) and TEA (55 mL, 0.546mol) in THF (250 mL) at 0 °C for 30min, The reaction was stirred at room temperature for 12h then the reaction mass was concentrated under reduced pressure and the residue was dissolved in DCM, washed with saturated NaHCO3 solution and brine. The organic layer was dried over anhydrous sodium sulphate and concentrated under reduced pressure, the crude was purified by silica gel column chromatography by eluting with 15% ethyl acetate-hexane to afford the title compound 4 (90g, 71% ) LCMS: m/z = 363.80 (M-Boc+2).
Step-3: Synthesis of tert-butyl 5-cyclopropyl-3-(3-methyl-2-(3-(4,4,5,5-tetramethyl-l,3,2-dioxaborolan-2-yl)phenyl)butanamido)-l H -pyr azole- 1 -carboxylate
[0354] To a degassed solution of tert-butyl 3-(2-(3-bromophenyl)-3-methylbutanamido)-5-cyclopropyl-lH-pyrazole-1 -carboxylate (intermediate-4, 90g, 0.193mol) and 4, 4, 4′, 4′, 5, 5, 5′, 5′-octamethyl-2,2′-bi(l,3,2-dioxaborolane) (62g, 0.25 Imol) in 1,4-Dioxane (500 mL) was added potassium acetate (37.80g, 0.386mol). The reaction mass was allowed to stir for 10 min with degassing at RT and added PdC12(dppf).DCM complex (12.5g, 0.015mol). The reaction mass was heated for 3-4 h at 100 °C. Reaction mixture cooled to RT and filtered on celite bed, filtrate evaporated to get dark brown liquid. The crude material was purified by silica column chromatography by eluting with 20% ethyl acetate in hexane to afford the compound 5 (90g, 86%). LCMS: m/z = 410 (M-Boc+1)+.
Step-4: Synthesis of (E)-N-(5-(3-(l-((5-cyclopropyl-lH-pyrazol-3-yl)amino)-3-methyl-l-oxobutan-2-yl)phenyl)pyridin-2-yl)-4-morpholinobut-2-enamide
[0355] To a degassed solution of tert-butyl 5-cyclopropyl-3-(3-methyl-2-(3-(4, 4,5,5-tetramethyl- 1 ,3 ,2-dioxaborolan-2-yl)phenyl)butanamido)- 1 H-pyrazole- 1 -carboxylate, 5 (10g, 0.019mol) and (E)-N-(5-bromopyridin-2-yl)-4-morpholinobut-2-enamide (7.7g, 0.023mol) in
1,4-Dioxane (lOOmL) and water (40mL) followed by Cs2CO3 (14.5g, 0.045mol) were added. The reaction mass was allowed to stir for 10 min with degassing and added Pd(PPh3)4 (1.1g, 0.00095mol), heated the reaction mass for 4 h at 100 °C in a sealed tube. The reaction mass was cooled and diluted with brine solution. The aqueous layer was separated and re-extracted with ethyl acetate. The combined organic layer was evaporated to dryness and crude material was purified by silica column chromatography by eluting with 10%-l 5 % methanol in DCM to get desired pure compound 44 (4.5g, 44%). LCMS: m/z = 529.15 (M+H)+; HPLC: 95.17%, rt: 6.34 min.
[0356] Racemic (E)-N-(5 -(3 -( 1 -((5 -cyclopropyl- 1 H-pyrazol-3 -yl)amino)-3 -methyl- 1 -oxobutan-2-yl)phenyl)pyridin-2-yl)-4-morpholinobut-2-enamide was separated by using chiral preparative HPLC column (Method: Column: Chiral Pak IA (20mm X 250 mm, 5 micron), Elution: isocratic (50:50), A=ACN, B= MeOH, Flow: 20mL/min ) to afford the pure Isomer- 1 and Isomer-2.
Isomer-1 (Compound 44-A):
[0357] 1HNMR (DMSO-d6, 400MHz): 5 12.02 (s, 1H), 10.78 (s, 1H), 10.44 (s, 1H), 8.61 (s, 1H), 8.28 (d, 1H), 8.07-8.05 (m, 1H), 7.68 (s, 1H), 7.57 (d, 1H), 7.41-7.37 (m, 2H), 6.81-6.78 (m, 1H), 6.49 (d, 1H), 6.13 (s, 1H), 3.61-3.58 (m, 4H), 3.36-3.34 (m, 1H), 3.12 (d, 2H), 2.41-2.32 (m, 5H), 1.82-1.76 (m, 1H), 0.97 (d, 3H), 0.88-0.85 (m, 2H), 0.67 (d, 3H), 0.62-0.59 (m, 2H); LCMS: m/z = 529.15 (M+H)+; HPLC: 96.72%, rt: 6.39 min; Chiral HPLC: 97.68%, rt: 14.47.
Isomer-2 (Compound 44B):
[0358] 1HNMR (DMSO-d6, 400MHz): 5 12.02 (s, 1H), 10.78 (s, 1H), 10.44 (s, 1H), 8.61 (s, 1H), 8.28 (d, 1H), 8.07-8.04 (m, 1H), 7.68 (s, 1H), 7.57 (d, 1H), 7.41-7.37 (m, 2H), 6.81-6.78 (m, 1H), 6.50 (d, 1H), 6.14 (s, 1H), 3.61-3.58 (m, 4H), 3.36-3.34 (m, 1H), 3.12 (d, 2H), 2.40-2.39 (m, 5H), 1.82-1.76 (m, 1H), 0.97 (d, 3H), 0.88-0.85 (m, 2H), 0.67 (d, 3H), 0.62-0.60 (m, 2H); LCMS: m/z = 529.15 (M+H)+; HPLC: 96.24%, rt: 6.39 min; Chiral HPLC: 97.92%, rt: 8.80.
Example 4: Preparation of Compound 44-A via Chiral Synthesis
Preparation of KRM-A (chemical precursor to Compound 44-A)


Step-4
KRM-A
Step-1: Preparation of 2-(3-bromophenyl)-3-methylbutanoic acid (1)
[0359] 2M LDA (698 mL, 1.38mol) was added to a solution of 2-(3 -bromophenyl) acetic acid (150 g, 0.69 mol) in THF (700mL) at -78 °C over a period of 30 min. The reaction mixture was stirred for 2h at -78 °C followed by drop wise addition of isopropyl bromide (XB, 255 g, 2.07 mol) over a period of 30 min at -78 °C. The reaction mass was stirred at room temperature overnight. The reaction mass was quenched with IN HC1 (pH 2) and the obtained product was extracted to ethyl acetate (500 mL x 3). The combined organic layer was washed with water followed by brine, dried over anhydrous sodium sulfate and concentrated under reduced pressure to afford the title crude compound which was purified by silica column by eluting with 0-10% ethyl acetate -hexane system to afford the title compound (150 g, 83% yield), HPLC purity-96%. The compound of formula (1) can also be prepared by the procedure described in CN110590747.
Step-2: Preparation of Compound 3
[0360] 2-(3-bromophenyl)-3-methylbutanoic acid (1, 510 g, 1.98 mol) was dissolved in 30% of IPA in water (10.2 L; 3.06 L of IPA-7.14 L of water) and (1R, 27?)-cyclohexane-l,2-diamine (2, 113 g, 0.9 mol) was added. The reaction mixture was stirred at room temperature for 10 min until the precipitation was observed, then heated to 100 °C till the solution becomes clear and was stirred at same temperature for another 30 min. The reaction mixture was allowed to attain room temperature slowly for 8-12h. The obtained solid was filtered and washed with 500 mL of 30% IPA-water mixture and dried under vacuum to afford the compound 3 (620 g, wet).
[0361] Work up for analysis (for Chiral purity): Small portion (100 mg) of compound 3 was taken in DCM (2-3 mL) and was added IN HC1 (pH 2) at 0 °C till the clear solution was observed. The compound was extracted into DCM, dried over Na2SC>4 and the solvent was evaporated to afford the title compound as white solid (20 mg). Chiral HPLC was recorded for this sample and 20.6% of undesired isomer was observed in chiral HPLC.
[0362] In order to improve the chiral purity of the title compound, the recrystallization method was performed as described below.
Step-3: Recrystallization
[0363] The compound 3 (619.90 g) was taken in 30% of IPA in water (12.4 L), then the mixture was heated to 100 °C till the solution becomes clear and stirred at same temperature for another 30min. The reaction mixture was allowed to attain room temperature slowly for 8-12h. The obtained solid was filtered and washed with 500mL 30% IP A- water and dried under vacuum to afford a desired compound (360g, wet).
[0364] Work up for analysis (for Chiral purity): Small portion (100 mg) from above compound was taken in DCM (2-3mL), was added IN HC1 (pH 2) at 0 °C till the clear solution was observed and the compound was extracted to DCM, dried over Na2SCL and the solvent was evaporated to afford title compound as white solid (35 mg). Chiral HPLC was recorded for this sample and 10.3% of undesired isomer was observed in chiral HPLC.
[0365] The recrystallization method was repeated for three more times by using 30% of IPA in water as described above to get the purity >98.50% ee along with 0.27% other isomer to afford 286 g of compound 4.
Step-4: Preparation of (S)-2-(3-bromophenyl)-3-methylbutanoic acid (KRM-A)
[0366] The compound 4 (286 g) was taken in DCM (1.3 L), then was added IN HC1 at 0 °C until the clear solution was observed, and the compound was extracted to DCM (500 mL x 2). The organic layer was separated and washed brine solution (500 mL) and dried over Na2SO4, the solvent was evaporated to afford title compound as white solid (148 g, 60% yield). Chiral HPLC: 98.50%
[0367] ‘H NMR (400MHz, DMSO-d6): 8 12.5 (s, 1H), 7.50-7.44 (m, 2H), 7.34-7.26 (m, 2H), 3.16 (d, 1H), 2.23-2.11 (m, 1H), 0.98 (d, 3H), 0.63 (d, 3H); Chiral HPLC: 98.50% retention time: 4.588 min.
Preparation of Compound 44-A


Step-1: Synthesis of (S)-2-(3-bromophenyl)-N-(5-cyclopropyl-lH-pyrazol-3-yl)-3-methylhutanamide
Step- la: Preparation of KRM-D
[0368] To a stirred solution of KRM-A (100g, 0.388mol) in dry DCM (600 mL, 6 vol), a catalytic amount of DMF (10 mL) was added followed by oxalyl chloride (45 mL, 0.525 mol) dropwise at 0 °C over a period of 30 min. After completion of addition, the reaction mixture was stirred for 15 min at the same temperature. The reaction mixture was allowed to reach room temperature and stirred for 2 to 4h. After completion of the reaction (reaction was monitored by TLC, acid chloride formation was checked by quenching an aliquot of reaction mixture with MeOH), the reaction mixture was concentrated under vacuum at 40°C-45°C to afford crude 2-(3-bromophenyl)-3 -methylbutanoyl chloride (KRM-D). The crude KRM-D was dissolved in toluene (500mL) and used for next step.
Step- lb: Preparation of compound of formula Z
[0369] (S)-2-(3-bromophenyl)-3 -methylbutanoyl chloride in toluene was added slowly to a pre-cooled solution (0 to 5 °C) of te/7-butyl 3 -amino-5 -cyclopropyl- IH-pyrazole-l -carboxylate (KRM-B, 95.5g, 0.427 mol) and N, N-diisopropylethyl amine (100 mL, 0.583 mol) in toluene (1.2 L) at 0 °C for the period of l-2h. The reaction mixture was allowed to attain RT and stirred for overnight. The reaction mixture was then cooled to 0-5°C and washed with ice-cold 1.5N HCI (3 x 500 mL). The organic layer was washed with sodium bicarbonate solution (500 mL),
brine solution (500 mL), dried over anhydrous Na2SO4, filtered and concentrated under vacuum at 45-50°C to afford crude tert-butyl (5)-5-(2-(3-bromophenyl)-3-methylbutanamido)-3-cyclopropyl-lH-pyrazole-1 -carboxylate (compound of formula Z) as light brown oil (~180g, LCMS: m/z= 461.9 (M+H)+, HPLC: 80.80%, retention time: 15.89 min). The crude product was taken as such for next step without further purification.
Step-lc: Preparation of compound of formula Y
[0370] To a suspension of tert-butyl (S)-5-(2-(3-bromophenyl)-3-methylbutanamido)-3-cyclopropyl-lH-pyrazole-1 -carboxylate (180 g, 1.731 mol) in dioxane (360 mL) was added 2N aqueous HC1 (360 mL) at 0 °C. The reaction mixture was stirred overnight at room temperature.
[0371] After completion of the reaction, dioxane was concentrated, and the reaction mixture was diluted with water (500 mL) and basified with solid sodium bicarbonate (until pH-8). The resulted compound was extracted with DCM (700 mL x 3). The combined organic layers were washed with water (300 mL) and brine solution (300 mL), and dried over anhydrous Na2SO4. The organic layer was concentrated to get a crude (<S)-2-(3-bromophenyl)-N-(5-cyclopropyl-lH-pyrazol-3-yl)-3-methylbutanamide (Compound of formula Y) as a semi solid. The crude was dissolved in toluene (500 mL) and the solution was stirred for 18 h. The solid formed was filtered and washed with toluene (100 mL) and n-heptane (200 mL). The solid was further dried under vacuum at 45-50°C for 6 h to afford a title compound (110g, Yield: 78% over two steps). LCMS: m/z= 362 (M+H)+, HPLC: 97.66%, retention time: 24.10 min
[0372] Step-2: Preparation of (S, E)-N-(5-(3-(l-((5-cyclopropyl-lH-pyrazol-3-yl) amino)-3-methyl-l-oxobutan-2-yl) phenyl) pyridin-2-yl)-4-morpholinobut-2-enamide (Compound 44A)
[0373] To a degassed solution of (<S)-2-(3-bromophenyl)-N-(5-cyclopropyl-lH-pyrazol-3-yl)-3-methylbutanamide (50 g, 0.138 mol) and (£)-4-morpholino-N-(5-(4,4,5,5-tetramethyl-l,3,2-dioxaborolan-2-yl)pyridin-2-yl)but-2-enamide (KRM-C, 56.6 g, 0.151 mol, 1.1 eq) (prepared according to the procedure described in W02020202001) in 1,4-dioxane (500 mL, 10 vol) and water (100 mL, 2 vol) was added K3PO4 tribasic (73.2 g, 0.345 mol, 2.5 eq) at room temperature The reaction mass was stirred for 20 min with argon purging (degassing). Pd(dppf)C12.DCM [l,l’-Bis(diphenylphosphino)ferrocene]dichloropalladium(II) complex with dichloromethane] (3.38 g, 0.0042 mol, and 0.03eq) was added and the reaction mixture was heated to 90°C for 1-2 h (The reaction was monitored by TLC using 10% methanol in DCM as solvent system).
[0374] After completion of the reaction, the reaction mass was cooled to room temperature and filtered through Celite® bed. The bed was washed with 1, 4-dioxane (200 mL) and the filtrate was concentrated to get crude compound. The crude compound was dissolved in 5% methanol in DCM (400 mL) and washed with water (200 mL x 2). The aqueous layer was separated and extracted with DCM (100 mL x 2). The combined organic layer was washed with brine solution, filtered and dried over sodium sulphate. The organic layer was concentrated under vacuum at 35-40°C to get crude title compound (~80g).
[0375] The crude compound 44A, (80 g) was dissolved in 700 mL of ethyl acetate. The reaction mixture was cooled to 15°C and 2N HC1 was slowly added (until pH ~1). The reaction mixture was then stirred at room temperature for 20 min and the layers were separated. The aqueous layer (containing the product) was washed with ethyl acetate (300 mL x 3). The aqueous layer was cooled to 0°C and adjusted the pH to ~8 using 20 % aqueous Na2COs solution. The product was extracted with 10% methanol in DCM (300 mL x 3). The combined organic layer was washed with water (300 mL), dried over sodium sulphate and filtered. The filtrate was treated with activated charcoal (16 g, 20% w/w with respect to crude input of 80 g), stirred overnight at room temperature and filtered through Celite® bed. The bed was washed with 5% methanol in DCM (~ 20 vol, till absence of product by TLC). The filtrate was concentrated under vacuum at 35°C – 40°C to afford compound 44A (70g, HPLC purity: 92.70%, retention time: 15.65 min).
[0376] ‘ H NMR (400MHz, DMSO-^): <5: 10.79 (s, 1H), 10.46 (s, 1H), 8.61 (d, 1H), 8.28 (d, 1H), 8.07-8.05 (m, 1H), 7.69 (s, 1H), 7.56 (d, 1H), 7.39 (m, 2H), 6.84-6.77 (m, 1H), 6.62 (s, 2H), 6.51 (d, 1H), 6.13 (s, 1H), 3.62-3.59 (m, 4H), 3.35 (d, 1H), 3.15-3.13 (m, 2H), 2.42-2.39 (m, 5H), 1.80-1.77 (m, 1H), 0.98 (d, 3H), 0.88-0.85 (m, 2H), 0.67 (d, 3H), 0.62-0.60 (m, 2H);
LCMS: m/z= 529.25-free base (M+H)+, HPLC: 98.98%, retention time: 15.40 min.


AS ON DEC2021 3,491,869 VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com
//////////
REF
POSTER SESSION: Molecular Targeted Agents| Volume 138, SUPPLEMENT 2, S47, October 01, 2020
//////Tacaciclib, GTPL12880, AUR-102
CC(C)C(C1=CC=CC(=C1)C2=CN=C(C=C2)NC(=O)C=CCN3CCOCC3)C(=O)NC4=NNC(=C4)C5CC5

NEW DRUG APPROVALS
ONE TIME
$10.00
CAREBASTINE
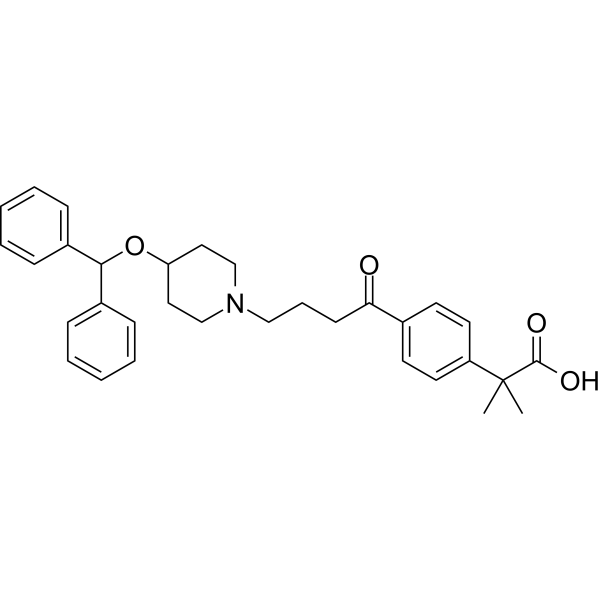
CAREBASTINE
| Molecular Weight | 499.64 |
|---|---|
| Appearance | Solid |
| Formula | C32H37NO4 |
| CAS No. | 90729-42-3 |
Carebastine is the active metabolite of Ebastine. Carebastine is a histamine H1 receptor antagonist. Carebastine inhibits VEGF-induced HUVEC and HPAEC proliferation, migration and angiogenesis in a dose-dependent manner. Carebastine suppresses the expression of macrophage migration inhibitory factor.
Carebastine is the active metabolite of Ebastine. Carebastine is a histamine H1 receptor antagonist. Carebastine inhibits VEGF-induced HUVEC and HPAEC proliferation, migration and angiogenesis in a dose-dependent manner[1]. Carebastine suppresses the expression of macrophage migration inhibitory factor[2].
Literature References: Nonsedating type histamine H1-receptor antagonist. Prepn: J. M. P. Soto et al., EP 134124; eidem, US 4550116 (both 1985 to Fordonal). Metabolized in vivo to carebastine, its active carboxylic acid metabolite.

PATENT
https://patents.google.com/patent/US8067604B2/en
These schemes also illustrate the interrelatedness of the processes and intermediates.










EXAMPLE 1
One gram of 9 was dissolved in 20 mL of DMF and 18 mg of P(tBu)3, 41 mg of Pd(dba)2, 230 mg of ZnF2 and 1.2 g of 5 were added. A mixture was stirred at 80° for 18 hours, cooled to room temperature, diluted with ether and washed with water. The organic layer was dried over sodium sulfate, filtered and stripped in vacuo. The resulting product was flash chromatographed on silica gel using 4:1 hexane ethyl acetate to yield 1.0 g (91%) of 10. A repeat of the reaction on larger scale using 15 g of 9 provided 15.2 g (93%) of 10.
EXAMPLE 2
Five grams of 9 was dissolved in 50 mL of methylene chloride and cooled to 0° C. To the solution was added 5.78 g of trimethylsilyl iodide. The mixture was stirred for 30 minutes and excess sodium bisulfite solution was added with vigorous stirring at room temperature. The layers were separated and the aqueous layer extracted twice with methylene chloride. Combined organic layers were dried, filtered and stripped in vacuo to provide 7.7 g (98%) of 1. The reaction was repeated on a larger scale using 15 g of 9 to produce 22.5 g of 1 (96%) yield.
EXAMPLE 3
Six grams of potassium carbonate, 5.8 g of piperidine 2 and 7.6 g of 1 are combined in 100 mL of DMF. The suspension is stirred at room temperature until TLC in 4:1 hexane-ethyl acetate indicates a complete reaction. The reaction mixture is poured into 400 mL of water and extracted three times with methylene chloride. The combined organic extracts are dried, filtered and reduced in vacuo. The resulting product is flash chromatographed on silica gel using ethyl acetate containing 10% triethylamine to yield 3.
EXAMPLE 4
Seven grams of 3 is dissolved in 100 mL of methanol, cooled to 0° C. and 1.1 g of sodium borohydride is added. The mixture is stirred 1 hour, concentrated and partitioned between ethyl acetate and saturated aqueous sodium bicarbonate. The bicarbonate layer is extracted twice with ethyl acetate, the combined organic layers are dried over sodium sulfate and the solution is reduced in vacuo to provide 4.
EXAMPLE 5
Two grams of 4 is dissolved in 30 mL of DMF. To this are added 16.2 mg of P(tBu)3, 36.6 mg of Pd(dba)2, 209 mg of ZnF2 and 1.056 g of 5. The mixture is heated at 80° C., cooled, diluted with ether and worked up as in example 1. The resulting product is flash chromatographed on silica gel using 9:1 ethyl acetate-triethylamine to provide 7.
EXAMPLE 6
One hundred fifty milligrams of 6 is slurried in 5 mL of water and 10 mL of methanol. To the slurry is added 175 mg of sodium hydroxide. The slurry is refluxed for one hour, cooled to room temperature and the methanol removed in vacuo. The resulting aqueous solution is distributed between water and chloroform, the chloroform layer is discarded, the aqueous layer is adjusted to pH 2.3 and extracted with chloroform. The organic layer is dried, filtered and reduced in vacuo to provide carebastine.
EXAMPLE 7
Five grams of 1 was combined with 2.64 g of 2 and 2.0 g of potassium carbonate and 80 mL of DMF. The mixture was stirred at room temperature for two hours, poured into 400 mL of water and extracted three times into methylene chloride. The combined organic layers were dried, filtered and reduced in vacuo. The resulting product was flash chromatographed on silica gel using 9:1 ethyl acetate-triethylamine to provide 2.0 g (54%) of 3.
EXAMPLE 8
One and seven-tenths grams of 3, 90 mg of P(tBu)3, 300 mg of Pd(dba)2, 250 mg of ZnF2 and 1.1 g of 5 were dissolved in 330 mL of DMF under argon. The mixture was heated to 80° for two hours, cooled to room temperature, diluted with ether and worked up as described in example 1. The resulting product was filtered through silica to provide 1.2 g (67.8%) of 6.
EXAMPLE 9
Two grams of 20, 170 mg of P(tBu)3, 560 mg of Pd(acac)2, 474 mg of ZnF2 and 2.0 g of 5 were combined in 50 mL of DMF under argon. The mixture was heated to 80° C. and monitored by HPLC. When reaction was complete, the mixture was cooled to room temperature and 250 mL of water was added. The mixture was extracted three times with ether, dried, filtered and reduced in vacuo. The resulting product was flash chromatographed in 4:1 hexane-ethyl acetate to provide 1.89 g (85%) of 8.
EXAMPLE 10
Two grams of the triflate analog of 20 were reacted as in the foregoing example with 134 mg P(tBu)3, 433 mg of Pd(acac)2, 375 mg of ZnF2 and 1.58 g of 5 to provide 1.56 g (90% yield) of 8.
Example 11
Piperidinol 25 is reacted with chlorodiphenylmethane as described in Fujii et al. Arzneim.-Forsch. 44, 527-538 (1994) to provide 6.
PATENT
WO/2023/213182CAREBASTINE SALT AND USE THEREOF
WIPO – Search International and National Patent Collections
Example 1: Potassium 2-(4-(4-(4-(diphenylmethoxy)piperidin-1-yl)butyryl)phenyl)-2-methylpropionate (carristin potassium salt ) preparation
[0060]
[0061]
Step 1: Preparation of methyl 2-(4-(4-(4-(diphenylmethoxy)piperidin-1-yl)butyryl)phenyl)-2-methylpropionate
[0062]
[0063]
Add 4-(diphenylmethoxy)piperidine hydrochloride (473mg, 1.77mmol), DMAC (4.5ml), K 3 PO 4 (1.13g, 5.3mmol), KI (29mg, 0.177mmol) to a 25ml single-neck bottle. , stir and heat to 100°C. Weigh 2-[4-(4-chloro-1-butyryl)phenyl]-2-methylpropionate methyl ester (600mg, 2.12mmol) and dissolve it in 1ml of DMAC. Add the reaction solution slowly and dropwise, and keep the reaction for 4~ 6h, TLC detects that the raw material reaction is complete. Cool to room temperature, add isopropyl acetate and water, and stir to separate layers. The aqueous phase was then extracted with isopropyl acetate, the organic phases were combined, washed twice with water, dried over anhydrous sodium sulfate, filtered, concentrated, and passed through a silica gel column to obtain 500 mg of the title product, yield 45%, purity: 97.3%.
[0064]
ESI-MS: m/z = 514.3(M+H) +。
[0065]
1H NMR (400 MHz, CDCl 3) δ: 7.93 (d, J=8.3Hz, 2H), 7.47 (m, 4H), 7.42 (d, J=8.3Hz, 2H), 7.30 (m, 4H), 7.18 (m, 2H), 3.64 (s, 3H),2.98 (m, 4H), 2.42 – 2.40 (m, 4H), 1.96 (m, 4H), 1.62 (s, 6H), 1.42 (m, 4H)。
[0066]
Step 2: Preparation of 2-(4-(4-(4-(Diphenylmethoxy)piperidin-1-yl)butyryl)phenyl)-2-methylpropionic acid (carristin)
[0067]
[0068]
Add (5-methyl-2-oxo-1,3-dioxo-4-yl)methyl-2-(4-(4-(4-(diphenylmethoxy))piperidine-1 to a 25ml three-necked flask) -Methyl)-butyryl)phenyl)-2-methylpropionate (320 mg, 0.62 mmol), 1.5 ml of methanol, 2 ml of 10% NaOH, heated to 60°C for 2 hours, and the TLC raw material reaction was completed. After the reaction is completed, cool to room temperature, concentrate to dryness, add EA, add hydrochloric acid to adjust the pH to 2~3, layer the layers, wash once with water, dry the organic phase, and concentrate to dryness to obtain 300 mg of the title product. Yield: 95%, purity 95.0%.
[0069]
ESI-MS: m/z = 500.3(M+H) +。
[0070]
1H NMR (400 MHz, CDCl 3) δ:7.75-7.63 (m, 2H), 7.57–7.24 (m,12H), 5.48 (s,1H),3.73 (m, 1H), 3.05–3.02 (m, 2H), 2.77–2.66 (m, 6H), 2.20–2.07 (m, 2H), 2.00–1.81 (m,4H), 1.58 (s, 6H)。
[0071]
Step 3: Potassium 2-(4-(4-(4-(Diphenylmethoxy)piperidin-1-yl)butyryl)phenyl)-2-methylpropionate (Carristine Potassium Salt) Preparation
[0072]
[0073]
Add 2-(4-(4-(4-(diphenylmethoxy)piperidin-1-yl)butyryl)phenyl)-2-methylpropionic acid (499mg, 1mmol) and acetonitrile 3.5 to a 25ml three-necked flask. ml, heated to 60°C, added potassium hydroxide (56 mg, 1 mmol), stirred, cooled down, a white solid precipitated, filtered, and dried to obtain 500 mg of carristine potassium salt, with a yield of 90% and a purity of 98.67%.
[0074]
ESI-MS: m/z = 500.3(M+H) +。
[0075]
1H NMR (400 MHz, CDCl 3) δ:7.75-7.63 (m, 2H), 7.57–7.24 (m,12H), 5.48 (s,1H),3.73 (m, 1H), 3.05–3.02 (m, 2H), 2.77–2.66 (m, 6H), 2.20–2.07 (m, 2H), 2.00–1.81 (m,4H), 1.58 (s, 6H)。
[0076]
Example 2: Sodium 2-(4-(4-(4-(diphenylmethoxy)piperidin-1-yl)butyryl)phenyl)-2-methylpropionate (carristine sodium salt ) preparation
[0077]
[0078]
In this example, the preparation method of 2-(4-(4-(4-(diphenylmethoxy)piperidin-1-yl)butyryl)phenyl)-2-methylpropionic acid is the same as in Example 1.
[0079]
Add 2-(4-(4-(4-(diphenylmethoxy)piperidin-1-yl)butyryl)phenyl)-2-methylpropionic acid (499mg, 1mmol) and acetonitrile 3.5 to a 25ml three-necked flask. ml, heated to 60°C, added sodium hydroxide (40 mg, 1 mmol) and stirred for 1 hour, concentrated to dryness, added methyl tert-butyl ether and stirred, filtered, and dried to obtain 458 mg of carristin sodium salt, yield 85%, purity 96.98 %.
[0080]
ESI-MS: m/z = 500.3(M+H) +。
[0081]
1H NMR (400 MHz, CDCl 3) δ:7.75-7.63 (m, 2H), 7.57–7.24 (m,12H), 5.48 (s,1H),3.73 (m, 1H), 3.05–3.02 (m, 2H), 2.77–2.66 (m, 6H), 2.20–2.07 (m, 2H), 2.00–1.81 (m,4H), 1.58 (s, 6H)。
//////////////


AS ON DEC2021 3,491,869 VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@amcrasto
///////////

NEW DRUG APPROVALS
ONE TIME
$10.00
VODOBATINIB
VODOBATINIB
1388803-90-4
| Molecular Weight | 453.92 |
|---|---|
| Appearance | Solid |
| Formula | C27H20ClN3O2 |
- SCO-088
- K0706
- K-0706
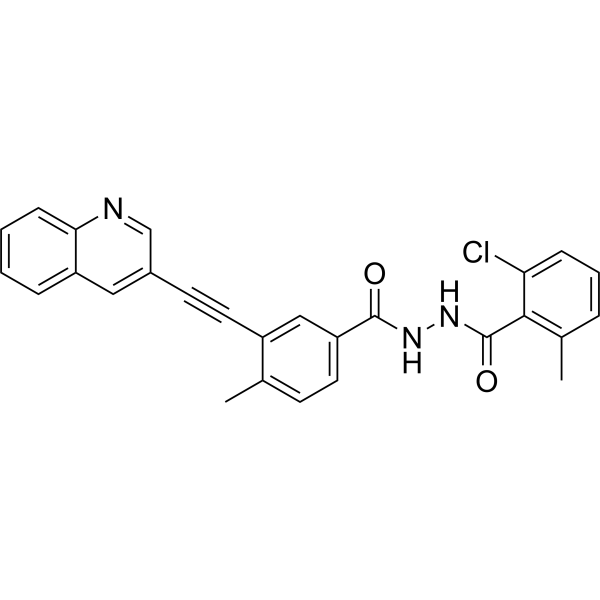
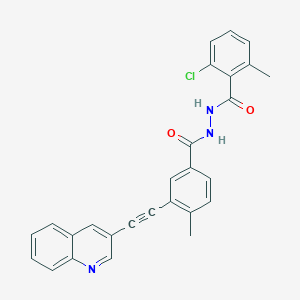
2-chloro-6-methyl-N‘-[4-methyl-3-(2-quinolin-3-ylethynyl)benzoyl]benzohydrazide
Vodobatinib (K0706) is a potent, third generation and orally active Bcr-Abl1 tyrosine kinase inhibitor with an IC50 of 7 nM. Vodobatinib exhibits activity against most BCR-ABL1 point mutants, and has no activity against BCR-ABL1T315I. Vodobatinib can be used for chronic myeloid leukemia (CML) research. Vodobatinib is a click chemistry reagent, itcontains an Alkyne group and can undergo copper-catalyzed azide-alkyne cycloaddition (CuAAc) with molecules containing Azide groups.
| Vodobatinib (K0706) is a potent, third generation and orally active Bcr-Abl1 tyrosine kinase inhibitor with an IC50 of 7 nM. Vodobatinib exhibits activity against most BCR-ABL1 point mutants, and has no activity against BCR-ABL1T315I. Vodobatinib can be used for chronic myeloid leukemia (CML) research[1][2]. Vodobatinib is a click chemistry reagent, itcontains an Alkyne group and can undergo copper-catalyzed azide-alkyne cycloaddition (CuAAc) with molecules containing Azide groups. |
Brain penetrant kinase inhibitors: Learning from kinase neuroscience discovery
Publication Name: Bioorganic & Medicinal Chemistry Letters
Publication Date: 2018-06-15
PMID: 29752185
DOI: 10.1016/j.bmcl.2018.05.007
PATENT
WO2012098416
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2012098416
EXAMPLES
Reƒerence Example 1
Methyl 3-ethynyl-4-methylbenzoate


A mixture of methyl 3-iodo-4-methylbenzoate (2.0g, 7mmol), trimethylsilylacetylene (1.2ml, 8mmol), Pd(PPh3)4 (0.42g, 0.3mmol), CuI (0.137g, 0.7mmol) and diisopropylethylamine (2.5ml, 11.4mmol) in THF (20ml) was heated at 50°C for 12hrs under nitrogen atmosphere. The reaction mixture was cooled to ambient temperature and filtered through a Celite® bed. The clear filtrate was concentrated and the residue purified by flash chromatography on silica gel (elution with 2% ethyl acetate in n-hexane) to provide methyl 4-methyl-3-[(trimethylsilyl)ethynyl]benzoate.
To the solution of methyl 4-methyl-3-[(trimethylsilyl)ethynyl]benzoate (2.3g) in THF (40ml) was added tetrabutylammonium fluoride (1.0M in THF, 3.2ml, 1 1mmol) at ambient
temperature and stirred for 15 minutes, concentrated and the residue purified by flash chromatography on silica gel (elution with 2% ethyl acetate in n-hexane) to provide methyl 3 – ethynyl- 4-methylbenzo at e .
1H NMR (500 MHz in DMSO-d6), δ 2.50 (s, 3H), 3.90 (s, 3H), 4.57 (s, 1H), 7.51 (d, J = 8.0 Hz, 1H), 7.91 (d, J = 8.0 Hz, 1H), 7.99 (s, 1H).
Similarly were prepared the following ester compounds from their corresponding iodo esters:
Methyl 3-ethynyl-4-fluorobenzoate
Methyl 3-ethynyl-4-methoxybenzoate
Reƒerence Example 2
4-Methyl-3-[(quinolin-3-yl)ethynyl]benzoic acid


A mixture of methyl 3-ethynyl-4-methylbenzoate (0.341 g, 2mmol), 3-iodoquinoline (0.5g, 2mmol), Pd(PPh3)4 (0.1 1g, 0.01mmol), CuI (0.179g, 0.1mmol) and diisopropylethylamine (0.5ml, 3mmol) in DMF (15ml) was stirred at ambient temperature for 12hrs under an atmosphere of nitrogen. The reaction mixture was concentrated and the crude product was purified by flash chromatography on silica gel (elution with 10% ethyl acetate in n-hexane) to provide methyl 4-methyl-3-[(quinolin-3-yl)ethynyl]benzoate.
Sodium hydroxide (0.15g, 3.71mmol) was added to a solution of the above methyl ester in methanol (20ml) and water (3ml) and stirred at 50°C for 3hrs and then concentrated in vacuo. Water (10ml) was added to the residue, adjusted pH to 4.0-4.5 with citric acid. The solid obtained was filtered, washed successively with water and diethyl ether and dried at ambient temperature to obtain 4-methyl-3-[(quinolin-3-yl)ethynyl]benzoic acid. 1H NMR (500 MHz in DMSO-d6), δ 2.66 (s, 3H), 7.56 (d, J = 8.0 Hz, 1H), 7.75 (t, J; = 15.1 Hz, J2 = 8.2 Hz, 1H), 7.89 (t, J} = 13.7 Hz, J2 = 8.5 Hz, 1H), 7.95 (d, J = 8.0 Hz, 1H), 8.09 (d, J = 8.2 Hz, 1H), 8.12 (d, J = 8.1 Hz, 1H), 8.17 (s, 1H), 8.75 (s, 1H), 9.1 1 (s, 1H), 12.84 (s, 1H).
Reƒerence Example 3
4-Methyl-3-[2-(3-quinolyl)ethynyl]benzohydrazide


A mixture of 4-methyl-3-[(quinolin-3-yl)ethynyl]benzoic acid (0.15g, 0.5mmol), N-(3-dimethylaminopropyl)-N’-ethylcarbodiimide hydrochloride (0.15g, 0.7mmol) and 1-hydroxybenzotriazole (0.1g, 0.7mmol) in N,N-dimethylformamide (15ml) was stirred at room temperature for 1hr. Hydrazine hydrate (1.52ml, 0.5mmol) was then added and the mixture stirred for another 3hrs. Concentration and trituration of the residue with water produced a solid which was filtered, washed successively with water and diethyl ether, and finally dried in vacuo to get the hydrazide as a pale yellow solid.
1H NMR (400 MHz in DMSO-d6), δ 2.63 (s, 3H), 4.79 (s, 2H), 7.51 (d, J = 8.0 Hz, 1H), 7.75 (t, J1 = 14.7 Hz, J2 = 7.6 Hz, 1H), 7.85-7.96 (m, 2H), 8.09-8.13 (m, 3H), 8.73 (s, 1H), 9.09 (s, 1H), 9.91 (s, 1H).
Reƒerence Example 4
N’-(3-iodo-4-methylbenzoyl)-2,4,6-trichlorobenzohydrazide


N’-(3-iodo-4-methylbenzoyl)-2,4,6-trichlorobenzohydrazide was prepared by the reaction of 3-iodo-4-methylbenzoic acid with 2,4,6-trichlorobenzohydrazide. The coupling was performed in a manner similar to that described in Reference Example 3.
Example 1.1
2,4,6-Trichloro-N’-[4-methyl-3-[2-(3-quinolyl)ethynyl]benzoyl]benzohydrazide
Method A:


A mixture of 4-methyl-3-[(quinolin-3-yl)ethynyl]benzoic acid (0.15g, 0.5mmol), N-(3-dimethylaminopropyl)-N’-ethylcarbodiimide hydrochloride (0.15g, 0.7mmol) and 1-hydroxybenzotriazole (0.1g, 0.7mmol) in N,N-dimethylformamide (15ml) was stirred at ambient temperature for 1hr. 2,4,6-Trichlorobenzohydrazide (0.125g, 0.5mmol) was added and the mixture stirred for 12hrs at ambient temperature. Concentration and trituration of the residue with water produced a solid which was filtered, washed with water and the crude product was purified by flash chromatography on silica gel (elution with 10% methanol in dichloromethane) to get 2,4,6-trichloro-N-[4-methyl-3-[2-(3-quinolyl)ethynyl]benzoyl] benzohydrazide as a white solid.
Method B:
2,4,6-Trichloro-N’-[4-methyl-3-[2-(3-quinolyl)ethynyl]benzoyl] benzohydrazide was also prepared by the reaction of 4-methyl-3-[(quinolin-3-yl)ethynyl]benzoic acid with 2,4,6-trichlorobenzohydrazide in diethyl cyanophosphonate. The condensation reaction was performed in a manner similar to that described in Method A.
Method C:


2,4,6-Trichloro-N-[4-methyl-3-[2-(3-quinolyl)ethynyl]benzoyl]benzohydrazide was also prepared by the reaction of 4-methyl-3-[(quinolin-3-yl)ethynyl]benzohydrazide with 2,4,6- trichlorobenzoyl chloride. The condensation reaction was performed in a manner similar to that described in Method A.
The compounds 1.2 to 1.14, 1.21 to 1.34, 1.36 to 1.40, and 1.43 to 1.59 were prepared in a manner similar to Example I.1, by following either of the methods A, B or C, using the appropriate substrates.
PATENT
WO2023214314 VODOBATINIB FOR REDUCING PROGRESSION OF PARKINSON’S DISEASE (wipo.int)
Vodobatinib (N’-(2-chloro-6-methylbenzoyl)-4-methyl-3-[2-(3-quinolyl) ethynyl]-benzohydrazide), a c-Abl inhibitor is represented by Formula I (referred hereinafter interchangeably as vodobatinib or compound of Formula
International Publication Nos. WO 2017/208267A1, WO 2020/250133 Al and WO 2022/024072A1, which are hereby incorporated by reference, disclose methods of use of the compound of Formula I for the treatment of Parkinson’s disease, synucleinopathies and Alzheimer’s disease (AD) respectively.
There is a continuing need for effective and safe methods for the treatment of, and delaying the progression of, neurodegenerative diseases, including in the early-stage of the diseases.
- N’-(2-chloro-6-methylbenzoyl)-4-methyl-3-[2-(3-quinolyl) ethynyl]-benzohydrazide for treatment of alzheimer’s diseasePublication Number: WO-2022024072-A1Priority Date: 2020-07-31
- Compounds for the treatment of covid-19Publication Number: EP-3875078-A1Priority Date: 2020-03-06
- Treatment for synucleinopathiesPublication Number: US-2022257582-A1Priority Date: 2019-06-11
- Novel amorphous dispersion of 4-Methyl-3-quinolin-3-ylethynyl-benzoic acid N’-(2-chloro-6-methyl-benzoyl) hydrazidePublication Number: AU-2018235446-A1Priority Date: 2017-03-15
- Novel amorphous dispersion of 4-methyl-3-quinolin-3-ylethynyl-benzoic acid n’-(2-chloro-6-methyl-benzoyl) hydrazidePublication Number: EP-3596050-A1Priority Date: 2017-03-15
- Novel amorphous dispersion of 4-methyl-3-quinolin-3-ylethynyl-benzoic acid n’-(2-chloro-6-methyl-benzoyl) hydrazidePublication Number: US-2020085751-A1Priority Date: 2017-03-15
- Novel amorphous dispersion of 4-methyl-3-quinolin-3-ylethynyl-benzoic acid n’-(2-chloro-6-methyl-benzoyl) hydrazidePublication Number: WO-2018167802-A1Priority Date: 2017-03-15
- NOVEL AMORPHOUS DISPERSION OF 4-METHYL-3-QUINOLIN-3-YETHYNYL-BENZOIC ACID HYDRAZIDE N- (2-CHLORO-6-METHYL-BENZOYL)Publication Number: WO-2018167802-A9Priority Date: 2017-03-15
- Novel amorphous dispersion of 4-methyl-3-quinolin-3-ylethynyl-benzoic acid n’-(2-chloro-6-methyl-benzoyl) hydrazidePublication Number: US-2021267906-A1Priority Date: 2017-03-15
- Novel amorphous dispersion of 4-Methyl-3-quinolin-3-ylethynyl-benzoic acid N’-(2-chloro-6-methyl-benzoyl) hydrazidePublication Number: AU-2018235446-B2Priority Date: 2017-03-15Grant Date: 2022-04-07
- Amorphous dispersion of 4-methyl-3-quinolin-3-ylethynyl-benzoic acid n’-(2-chloro-6-methyl-benzoyl) hydrazidePublication Number: US-11351123-B2Priority Date: 2017-03-15Grant Date: 2022-06-07
- Treatment of parkinson’s diseasePublication Number: US-2019275017-A1Priority Date: 2016-06-02
- Treatment of Parkinson’s diseasePublication Number: US-10849887-B2Priority Date: 2016-06-02Grant Date: 2020-12-01
- Treatment of Parkinson’s diseasePublication Number: IL-263188-APriority Date: 2016-06-02
- Treatment of Parkinson’s diseasePublication Number: CN-109475539-BPriority Date: 2016-06-02Grant Date: 2021-12-28
- Treatment of Parkinson’s diseasePublication Number: JP-6974357-B2Priority Date: 2016-06-02Grant Date: 2021-12-01
- Treatment for parkinson’s diseasePublication Number: US-2022273632-A1Priority Date: 2016-06-02
- Diarylacetylene hydrazide containing tyrosine kinase inhibitorsPublication Number: AU-2012208388-A1Priority Date: 2011-01-21
- Diarylacetylene hydrazide containing tyrosine kinase inhibitorsPublication Number: AU-2012208388-A2Priority Date: 2011-01-21
- Diarylacetylene hydrazide containing tyrosine kinase inhibitorsPublication Number: EP-2665709-B1Priority Date: 2011-01-21Grant Date: 2016-12-07
- Tyrosine kinase inhibitors containing diarylacetylene hydrazidePublication Number: ES-2608829-T3Priority Date: 2011-01-21Grant Date: 2017-04-17
- Diarylacetylene hydrazide containing tyrosine kinase inhibitorsPublication Number: US-2013296557-A1Priority Date: 2011-01-21
- Diarylacetylene hydrazide containing tyrosine kinase inhibitorsPublication Number: US-9024021-B2Priority Date: 2011-01-21Grant Date: 2015-05-05
//////////////


AS ON DEC2021 3,491,869 VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@amcrasto
///////////
Ref
- [1]. Orlando Antelope, et al. BCR-ABL1 tyrosine kinase inhibitor K0706 exhibits preclinical activity in Philadelphia chromosome-positive leukemia. Exp Hematol. 2019 Sep;77:36-40.e2. [Content Brief][2]. Phase 1 Trial of Vodobatinib, a Novel Oral BCR-ABL1 Tyrosine Kinase Inhibitor (TKI): Activity in CML Chronic Phase Patients Failing TKI Therapies Including Ponatinib. Session: 632: Chronic Myeloid Leukemia: Therapy: CML: New and Beyond.
///////VODOBATINIB, SCO-088, K0706, K-0706
CC1=C(C(=CC=C1)Cl)C(=O)NNC(=O)C2=CC(=C(C=C2)C)C#CC3=CC4=CC=CC=C4N=C3
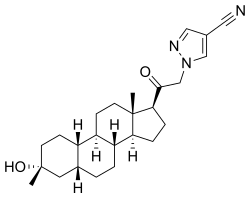
Zuranolone
Zuranolone
CAS 1632051-40-1
FDA APPROVED 8/4/2023, To treat postpartum depression
Press Release
WeightAverage: 409.574
Monoisotopic: 409.272927379Chemical FormulaC25H35N3O2
- SAGE 217
- SAGE-217
- SAGE217
Zuranolone, sold under the brand name Zurzuvae, is a medication used for the treatment of postpartum depression.[1][2] It is taken by mouth.[1]
The most common side effects include drowsiness, dizziness, diarrhea, fatigue, nasopharyngitis, and urinary tract infection.[1][2] An orally active inhibitory pregnane neurosteroid, zuranolone acts as a positive allosteric modulator of the GABAA receptor.[6][7][8]
Zuranolone was approved for medical use in the United States for the treatment of postpartum depression in August 2023.[2] It was developed by Sage Therapeutics and Biogen.[9]
Medical uses
Zuranolone is indicated for the treatment of postpartum depression.[1][2]
Adverse effects
The most common side effects include drowsiness, dizziness, diarrhea, fatigue, nasopharyngitis (cold-like symptoms), and urinary tract infection.[2]
The US FDA label contains a boxed warning noting that zuranolone can impact a person’s ability to drive and perform other potentially hazardous activities.[2] Use of zuranolone may cause suicidal thoughts and behavior.[2] Zuranolone may cause fetal harm.[2]
History
Zuranolone was developed as an improvement on the intravenously administered neurosteroid brexanolone, with high oral bioavailability and a biological half-life suitable for once-daily administration.[7][10] Its half-life is around 16 to 23 hours, compared to approximately 9 hours for brexanolone.[4][5]
The efficacy of zuranolone for the treatment of postpartum depression in adults was demonstrated in two randomized, double-blind, placebo-controlled, multicenter studies.[2] The trial participants were women with postpartum depression who met the Diagnostic and Statistical Manual of Mental Disorders criteria for a major depressive episode and whose symptoms began in the third trimester or within four weeks of delivery.[2] In study 1, participants received 50 mg of zuranolone or placebo once daily in the evening for 14 days.[2] In study 2, participants received another zuranolone product that was approximately equal to 40 mg of zuranolone or placebo, also for 14 days.[2] Participants in both studies were monitored for at least four weeks after the 14-day treatment.[2] The primary endpoint of both studies was the change in depressive symptoms using the total score from the 17-item Hamilton depression rating scale (HAMD-17), measured at day 15.[2] Participants in the zuranolone groups showed significantly more improvement in their symptoms compared to those in the placebo groups.[2] The treatment effect was maintained at day 42—four weeks after the last dose of zuranolone.[2]
Society and culture
Zuranolone is the international nonproprietary name.[11]
Legal status
Zuranolone was approved by the US Food and Drug Administration (FDA) for the treatment of postpartum depression in August 2023.[2][12] The FDA granted the application for zuranolone priority review and fast track designations.[2] Approval of Zurzuvae was granted to Sage Therapeutics, Inc.[2]
Zuranolone has also been under development for the treatment of major depressive disorder, but the application for this use was given a Complete Response Letter (CRL) by the FDA due to insufficient evidence of effectiveness.[13]
Research
In a randomized, placebo-controlled phase III trial to assess its efficacy and safety for the treatment of major depressive disorder, subjects in the zuranolone group (50 mg oral zuranolone once daily for 14 days) experienced statistically significant and sustained improvements in depressive symptoms (as measured by HAM-D score) throughout the treatment and follow-up periods of the study.[14]
Other investigational applications include insomnia, bipolar depression, essential tremor, and Parkinson’s disease.[15][6][16]
syn

PATENT
WO2022020363
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2022020363&_cid=P11-LLRZ9A-38538-1
Example 1. Synthesis of 1-(2-((3R,5R,8R,9R,10S,13S,14S,17S)-3-hydroxy-3,13-dimethylhexadecahydro-1H-cyclopenta[a]phenanthren-17-yl)-2-oxoethyl)-1H-pyrazole-4-carbonitrile (Compound 1).
[00488] To a suspension of K2CO3 (50 mg, 0.36 mmol) in THF (5 mL) was added 1H-pyrazole-4-carbonitrile (100 mg, 0.97 mmol) and 2-bromo-1-((3R,5R,8R,9R,10S,13S,14S,17S)-3-hydroxy-3,13-dimethylhexadecahydro-1H-cyclopenta[ ^]phenanthren-17-yl)ethan-1-one (50 mg, 0.12 mmol). The mixture was stirred at room temperature for 15 hours. The reaction mixture was poured into 5 mL H2O and extracted with ethyl acetate (2×10 mL). The combined organic layers were washed with brine, dried over sodium sulfate, filtered and concentrated. The residue mixture was purified by reverse-phase preparative HPLC to afford Compound 1 as a white solid (9 mg, 17.4% yield).1H NMR (500 MHZ, CDCl3) δ (ppm) 7.87 (1H, s), 7.82 (1H, s), 5.02 (1H, AB), 4.2 (1H, AB), 2.61 (1H, t), 2.16-2.24 (1H, m), 2.05 (1H, dxt), 1.70-1.88 (6H, m), 1.61-1.69 (2H, m), 1.38-1.52 (6H, m), 1.23-1.38 (5H, m), 1.28 (3H, s), 1.06-1.17 (3H, m), 0.67 (3H, s). LCMS: rt=2.24 min, m/z=410.1 [M+H]+.
PAPER
Journal of Medicinal Chemistry (2017), 60(18), 7810-7819
https://pubs.acs.org/doi/10.1021/acs.jmedchem.7b00846
Certain classes of neuroactive steroids (NASs) are positive allosteric modulators (PAM) of synaptic and extrasynaptic GABAA receptors. Herein, we report new SAR insights in a series of 5β-nor-19-pregnan-20-one analogues bearing substituted pyrazoles and triazoles at C-21, culminating in the discovery of 3α-hydroxy-3β-methyl-21-(4-cyano-1H-pyrazol-1′-yl)-19-nor-5β-pregnan-20-one (SAGE-217, 3), a potent GABAA receptor modulator at both synaptic and extrasynaptic receptor subtypes, with excellent oral DMPK properties. Compound 3 has completed a phase 1 single ascending dose (SAD) and multiple ascending dose (MAD) clinical trial and is currently being studied in parallel phase 2 clinical trials for the treatment of postpartum depression (PPD), major depressive disorder (MDD), and essential tremor (ET).


3α-Hydroxy-3β-methyl-21-(4-cyano-1H-pyrazol-1′-yl)-19- nor-5β-pregnan-20-one (3). Yield: 28 g (49%) as an off-white solid. LC-MS: tR = 1.00 min, m/z = 410 (M + 1). 1 H NMR (400 MHz, CDCl3): δ 7.86 (s, 1H), 7.80 (s, 1H), 5.08−4.84 (m, 2H), 2.70−2.55 (m, 1H), 2.25−2.15 (m, 1H), 2.10−2.00 (m, 1H), 1.88−1.59 (m, 7H), 1.53−1.30 (m, 15H), 1.25−1.00 (m, 3H), 0.67 (s, 3H). 13C NMR (100 MHz, CDCl3): δ 13.92 (CH3), 23.20, 24.44, 25.54, 25.78, 26.15 (5 × CH2), 26.69 (CH3), 31.43, 34.61 (2 × CH2), 34.77, 37.71 (2 × CH), 39.26 (CH2), 40.35 (CH), 41.21 (CH2), 41.75 (CH), 45.56 (C), 56.04, 61.24 (2 × CH), 61.78 (CH2), 72.14 (C), 93.25 (C), 113.35 (CN), 136.16, 142.49 (2 × CH), 202.23 (CO). HRMS m/z 410.2803 calcd for C25H36N3O2 + 410.2802
PATENT
WO2014169833
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2014169833&_cid=P11-LLRZJ9-40598-1
Synthetic Procedures
The compounds of the invention can be prepared in accordance with methods described in the art (Upasmi et al., J. Med. Chem. 1997, 40:73-84; and Hogenkamp et al., J. Med. Chem. 1997, 40:61- 72) and using the appropriate reagents, starting materials, and purification methods known to those skilled in the art. In some embodiments, compounds described herein can be prepared using methods shown in general Schemes 1-4, comprising a nucleophilic substitution of 19-nor pregnane bromide with a neucleophile. In certain embodiments, the nucleophile reacts with the 19-nor pregnane bromide in the presence of K2CO3 in THF.


Synthesis of compound SA-B. Compound SA (50 g, 184 mmol) and palladium black (2.5 g) in tetrahydrofuran (300 mL) and concentrated hydrobromic acid (1.0 mL) was hydrogenated with 10 atm hydrogen. After stirring at room temperature for 24h, the mixture was filtered through a pad of celite and the filtrate was concentrated in vacuo to afford the crude compound. Recrystallization from acetone gave compound SA-B (42.0 g, yield: 83.4%) as white powder.
1H NMR: (400 MHz, CDCl3) δ 2.45-2.41 (m, 1H), 2.11-3.44 (m, 2H), 3.24 (s, 3H), 2.18-2.15 (m, 1H), 2.01-1.95 (m, 1H), 1.81-1.57 (m, 7H), 1.53-1.37 (m, 7H), 1.29-1.13 (m, 3H), 1.13-0.90 (m, 2H), 0.89 (s, 3H).
Synthesis of compound SA-C. A solution of SA-B (42.0 g, 153.06 mmol) in 600 mL anhydrous toluene was added dropwise to the methyl aluminum bis(2,6-di-tert-butyl-4-methylphenoxide (MAD) (459.19 mmol, 3.0 eq, freshly prepared) solution under N2 at -78°C. After the addition was completed, the reaction mixture was stirred for 1 hr at -78°C. Then 3.0 M MeMgBr (153.06 mL, 459.19 mmol) was slowly added dropwise to the above mixture under N2 at -78°C. Then the reaction mixture was stirred for 3 hr at this temperature. TLC (Petroleum ether/ethyl acetate = 3:1) showed the reaction was completed. Then saturated aqueous NH4Cl was slowly added dropwise
to the above mixture at -78°C. After the addition was completed, the mixture was filtered, the filter cake was washed with EtOAc, the organic layer was washed with water and brine, dried over anhydrous Na2SO4, filtered and concentrated, purified by flash Chromatography on silica gel (Petroleum ether/ ethyl acetate20:1 to 3:1) to afford compound SA-C (40.2 g, yield: 90.4%) as white powder. 1H NMR: (400 MHz, CDCl3) δ 2.47-2.41 (m, 1H), 2.13-2.03 (m, 1H), 1.96-1.74 (m, 6H), 1.70-1.62 (m, 1H), 1.54-1.47 (m, 3H), 1.45-1.37 (m, 4H), 1.35-1.23 (m, 8H), 1.22-1.10 (m, 2H), 1.10-1.01 (m, 1H), 0.87 (s, 3H).
Synthesis of compound SA-D. To a solution of PPh3EtBr (204.52 g, 550.89 mmol) in THF (500 mL) was added a solution of t-BuOK (61.82 g, 550.89 mmol) in THF (300 mL) at 0°C. After the addition was completed, the reaction mixture was stirred for 1 h 60 °C, then SA-C (40.0 g, 137.72 mmol) dissolved in THF (300 mL) was added dropwise at 60°C. The reaction mixture was heated to 60 °C for 18 h. The reaction mixture was cooled to room temperature and quenched with Sat. NH4Cl, extracted with EtOAc (3*500 mL). The combined organic layers were washed with brine, dried and concentrated to give the crude product, which was purified by a flash column chromatography (Petroleum ether/ ethyl acetate50:1 to 10:1) to afford compound SA-D (38.4 g, yield:92%) as a white powder. 1H NMR: (400 MHz, CDCl3) δ 5.17-5.06 (m, 1H), 2.42-2.30 (m, 1H), 2.27-2.13 (m, 2H), 1.89-1.80 (m, 3H), 1.76-1.61 (m, 6H), 1.55-1.43 (m, 4H), 1.42-1.34 (m, 3H), 1.33-1.26 (m, 6H), 1.22-1.05 (m, 5H), 0.87 (s, 3H).
Synthesis of compound SA-E. To a solution of SA-D (38.0 g, 125.62 mmol) in dry THF (800 mL) was added dropwise a solution of BH3.Me2S (126 mL, 1.26 mol) under ice-bath. After the addition was completed, the reaction mixture was stirred for 3 h at room temperature (14-20 °C). TLC (Petroleum ether/ ethyl acetate3:1) showed the reaction was completed. The mixture was cooled to 0 °C and 3.0 M aqueous NaOH solution (400 mL) followed by 30% aqueous H2O2 (30%, 300 mL) was added. The mixture was stirred for 2 h at room temperature (14-20 °C), and then filtered, extracted with EtOAc (3*500 mL). The combined organic layers were washed with saturated aqueous Na2S2O3, brine, dried over Na2SO4 and concentrated in vacuum to give the crude product (43 g , crude) as colorless oil. The crude product was used in the next step without further purification.
Synthesis of compound SA-F. To a solution of SA-E (43.0 g, 134.16 mmol) in dichloromethane (800 mL) at 0 °C and PCC (53.8 g, 268.32 mmol) was added portion wise. Then the reaction mixture was stirred at room temperature (16-22 °C) for 3 h. TLC (Petroleum ether/ ethyl acetate3:1) showed the reaction was completed, then the reaction mixture was filtered, washed with DCM. The organic phase was washed with saturated aqueous Na2S2O3, brine, dried over Na2SO4 and concentrated in vacuum to give the crude product. The crude product was purified by a flash column chromatography (Petroleum ether/ ethyl acetate50:1 to 8:1) to afford compound SA-F (25.0 g, yield:62.5%, over two steps) as a white powder. 1H NMR (SA-F): (400 MHz, CDCl3) δ 2.57-2.50 (m, 1H), 2.19-2.11 (m, 4H), 2.03-1.97 (m, 1H), 1.89-1.80 (m, 3H), 1.76-1.58 (m, 5H), 1.47-1.42 (m, 3H), 1.35-1.19 (m, 10H), 1.13-1.04 (m, 3H), 0.88-0.84 (m, 1H), 0.61 (s, 3H).
Synthesis of compound SA. To a solution of SA-F (10 g, 31.4 mmol) and aq. HBr (5 drops, 48% in water) in 200 mL of MeOH was added dropwise bromine (5.52 g, 34.54 mmol). The reaction mixture was stirred at 17 °C for 1.5 h. The resulting solution was quenched with saturated aqueous NaHCO3 at 0°C and extracted with EtOAc (150 mLx2). The combined organic layers were dried and concentrated. The residue was purified by column chromatography on silica gel eluted with (PE: EA=15:1 to 6:1) to afford compound SA (9.5 g, yield: 76.14%) as a white solid. LC/MS: rt 5.4 mm ; m/z 379.0, 381.1, 396.1.

To a suspension of K2CO3 (50 mg, 0.36mmol) in THF (5 mL) was added ethyl 1H-pyrazole-4-carbonitrile (100 mg, 0.97 mmol ) and SA (50 mg,0.12 mmol). The mixture was stirred at rt for 15h. The reaction mixture was poured in to 5 mL H2O and extracted with EtOAc (2 x 10 mL). The combined organic layers were washed with brine, dried over sodium sulfate, filtered and concentrated. The residue mixture was purified with by reverse-phase prep-HPLC to afford the title compound as a white solid (9mg, 17.4%). 1H NMR (500 MHz, CDCl3), δ (ppm) 7.87 (1H, s),
7.82 (1H, s), 5.02 (1H, AB), 4.92 (1H, AB), 2.61 (1H, t), 2.16-2.24 (1H, m), 2.05 (1H, dXt), 1.70-1.88 (6H, m), 1.61-1.69 (2H, m), 1.38-1.52 (6H, m), 1.23-1.38 (5H, m), 1.28 (3H, s), 1.06-1.17 (3H, m), 0.67 (3H, s). LCMS: rt = 2.24 mm, m/z = 410.1 [M+H]+.
PATENT
WO2020150210


AS ON DEC2021 3,491,869 VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@amcrasto
///////////
References
- ^ Jump up to:a b c d e “Zurzuvae (zuranolone) capsules, for oral use, [controlled substance schedule pending]” (PDF). Archived (PDF) from the original on 5 August 2023. Retrieved 5 August 2023.
- ^ Jump up to:a b c d e f g h i j k l m n o p q r s t “FDA Approves First Oral Treatment for Postpartum Depression”. U.S. Food and Drug Administration (FDA) (Press release). 4 August 2023. Retrieved 4 August 2023.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain. - ^ Jump up to:a b “Zuranolone”. DrugBank Online.
- ^ Jump up to:a b Cerne R, Lippa A, Poe MM, Smith JL, Jin X, Ping X, et al. (2022). “GABAkines – Advances in the discovery, development, and commercialization of positive allosteric modulators of GABAA receptors”. Pharmacology & Therapeutics. 234: 108035. doi:10.1016/j.pharmthera.2021.108035. PMC 9787737. PMID 34793859. S2CID 244280839.
- ^ Jump up to:a b Faden J, Citrome L (2020). “Intravenous brexanolone for postpartum depression: what it is, how well does it work, and will it be used?”. Therapeutic Advances in Psychopharmacology. 10: 2045125320968658. doi:10.1177/2045125320968658. PMC 7656877. PMID 33224470.
- ^ Jump up to:a b “SAGE 217”. AdisInsight. Archived from the original on 29 March 2019. Retrieved 10 February 2018.
- ^ Jump up to:a b Blanco MJ, La D, Coughlin Q, Newman CA, Griffin AM, Harrison BL, et al. (2018). “Breakthroughs in neuroactive steroid drug discovery”. Bioorganic & Medicinal Chemistry Letters. 28 (2): 61–70. doi:10.1016/j.bmcl.2017.11.043. PMID 29223589.
- ^ Martinez Botella G, Salituro FG, Harrison BL, Beresis RT, Bai Z, Blanco MJ, et al. (2017). “Neuroactive Steroids. 2. 3α-Hydroxy-3β-methyl-21-(4-cyano-1H-pyrazol-1′-yl)-19-nor-5β-pregnan-20-one (SAGE-217): A Clinical Next Generation Neuroactive Steroid Positive Allosteric Modulator of the (γ-Aminobutyric Acid)A Receptor”. Journal of Medicinal Chemistry. 60 (18): 7810–7819. doi:10.1021/acs.jmedchem.7b00846. PMID 28753313.
- ^ Saltzman J (4 August 2023). “FDA approves postpartum depression pill from two Cambridge drug firms”. The Boston Globe. Archived from the original on 6 August 2023. Retrieved 5 August 2023.
- ^ Althaus AL, Ackley MA, Belfort GM, Gee SM, Dai J, Nguyen DP, et al. (2020). “Preclinical characterization of zuranolone (SAGE-217), a selective neuroactive steroid GABAA receptor positive allosteric modulator”. Neuropharmacology. 181: 108333. doi:10.1016/j.neuropharm.2020.108333. PMC 8265595. PMID 32976892.
- ^ World Health Organization (2019). “International nonproprietary names for pharmaceutical substances (INN): recommended INN: list 82”. WHO Drug Information. 33 (3). hdl:10665/330879.
- ^ “FDA Approves Zurzuvae (zuranolone), the First and Only Oral Treatment Approved for Women with Postpartum Depression, and Issues a Complete Response Letter for Major Depressive Disorder” (Press release). Biogen Inc. 4 August 2023. Retrieved 4 August 2023 – via GlobeNewswire.
- ^ McKenzie H. “Sage Hints at Difficult Decisions After Zuranolone’s Rejection in MDD”.
- ^ Clayton AH, Lasser R, Parikh SV, Iosifescu DV, Jung J, Kotecha M, et al. (May 2023). “Zuranolone for the Treatment of Adults With Major Depressive Disorder: A Randomized, Placebo-Controlled Phase 3 Trial”. The American Journal of Psychiatry: appiajp20220459. doi:10.1176/appi.ajp.20220459. PMID 37132201. S2CID 258461851.
- ^ Deligiannidis KM, Meltzer-Brody S, Gunduz-Bruce H, Doherty J, Jonas J, Li S, et al. (2021). “Effect of Zuranolone vs Placebo in Postpartum Depression: A Randomized Clinical Trial”. JAMA Psychiatry. 78 (9): 951–959. doi:10.1001/jamapsychiatry.2021.1559. PMC 8246337. PMID 34190962.
- ^ Bullock A, Kaul I, Li S, Silber C, Doherty J, Kanes SJ (2021). “Zuranolone as an oral adjunct to treatment of Parkinsonian tremor: A phase 2, open-label study”. Journal of the Neurological Sciences. 421: 117277. doi:10.1016/j.jns.2020.117277. PMID 33387701. S2CID 229333842.
External links
- Clinical trial number NCT04442503 for “A Study to Evaluate the Efficacy and Safety of SAGE-217 in Participants With Severe Postpartum Depression (PPD)” at ClinicalTrials.gov
- Clinical trial number NCT02978326 for “A Study to Evaluate SAGE-217 in Participants With Severe Postpartum Depression” at ClinicalTrials.gov
/////////Zuranolone, FDA 2023, APPROVALS 2023, Zurzuvae, postpartum depression , SAGE 217, SAGE-217, SAGE217
[H][C@@]1(CC[C@@]2([H])[C@]3([H])CC[C@]4([H])C[C@](C)(O)CC[C@]4([H])[C@@]3([H])CC[C@]12C)C(=O)CN1C=C(C=N1)C#N
SMARTCHEM FROM ROW2 TECHNOLOGIES
Are you aware of any Chemical Database which offers one stop solution to the Sourcing, R&D and Business Development department? Explore Smartchem to Quickly find Suppliers (Procurement), Customers (BD) & Synthetic pathways (R&D)
Is this the information you looking for? Evaluate SmartChem, lets schedule a demo. Try us once. You will use us for life.
Anand Ramakrishnan |Vice President – Sales & Operations | +91 9821384045 | +1 973-795-1141 | ranand@row2technologies.com | http://www.row2technologies.com | WhatsApp: +91 9821384045
Anthony Crasto | Advisor | +91 9321316780 | c_acrasto@row2technologies.com | amcrasto@gmail.com | www.row2technologies.com | WhatsApp: +91 9321316780 |
ABY 737
ABT-737
| Molecular Weight | 813.43 |
|---|---|
| Formula | C42H45ClN6O5S2 |
| CAS No. | 852808-04-9 |
ABT-737 is a small molecule drug that inhibits Bcl-2 and Bcl-xL, two members of the Bcl-2 family of evolutionarily-conserved proteins that share Bcl-2 Homology (BH) domains. First developed as a potential cancer chemotherapy,[1] it was subsequently identified as a senolytic (a drug that selectively induces cell death in senescent cells).[2]
The Bcl-2 family is most notable for their regulation of apoptosis, a form of programmed cell death, at the mitochondrion; Bcl-2 and Bcl-xL are anti-apoptotic proteins. Because many cancers have mutations in these genes that allow them to survive, scientists began working to develop drugs that would inhibit this pathway in the 1990s.[1] ABT-737 was one of the earliest of a series of drugs developed by Abbott Laboratories (now Abbvie) to target this pathway, based on their resolution of the 3D structure of Bcl-xL and studies using high-field solution nuclear magnetic resonance (NMR) that revealed how the BH domains of these proteins interacted with their targets.[1]
ABT-737 was superior to previous BCL-2 inhibitors given its higher affinity for Bcl-2, Bcl-xL and Bcl-w. In vitro studies showed that primary cells from patients with B-cell malignancies are sensitive to ABT-737.[3] In animal models, it improved survival, caused tumor regression, and cured a high percentage of mice.[4] In preclinical studies utilizing patient xenografts, ABT-737 showed efficacy for treating lymphoma and other blood cancers.[5]
Unfortunately, ABT-737 is not bioavailable after oral administration, leading to the development of navitoclax (ABT-263) as an orally-available derivative with similar activity on small cell lung cancer (SCLC) cell lines.[1][6] Navitoclax entered clinical trials,[1][6] and showed promise in haematologic cancers, but was stalled when it was found to cause thrombocytopenia (severe loss of platelets), which was discovered to be caused by the platelets’ requirement for Bcl-xL for survival.[1]
Subsequently, it was reported that ABT-737 specifically induces apoptosis in senescent cells in vitro and in mouse models.[2]
ABT-737, a BH3 mimetic, is a potent Bcl-2, Bcl-xL and Bcl-w inhibitor with EC50s of 30.3 nM, 78.7 nM, and 197.8 nM, respectively. ABT-737 induces the disruption of the BCL-2/BAX complex and BAK-dependent but BIM-independent activation of the intrinsic apoptotic pathway. ABT-737 induces autophagy and has the potential for acute myeloid leukemia (AML) research.
PATENT
PATENT
CN113248415
PATENT
US20070015787
Journal of Medicinal Chemistry (2007), 50(4), 641-662
https://pubs.acs.org/doi/10.1021/jm061152t

////////

AS ON DEC2021 3,491,869 VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@amcrasto
/////////////////////////////////////////////////////////////////////////////
| Names | |
|---|---|
| Preferred IUPAC name4-{4-[(4′-Chloro[1,1′-biphenyl]-2-yl)methyl]piperazin-1-yl}-N-(4-{[(2R)-4-(dimethylamino)-1-(phenylsulfanyl)butan-2-yl]amino}-3-nitrobenzene-1-sulfonyl)benzamide | |
| Identifiers | |
| CAS Number | 852808-04-9 |
| 3D model (JSmol) | Interactive image |
| ChEBI | CHEBI:47575 |
| ChemSpider | 9403232 |
| PubChemCID | 11228183 |
| UNII | Z5NFR173NV |
| CompTox Dashboard (EPA) | DTXSID7042641 |
| showInChI | |
| showSMILES | |
| Properties | |
| Chemical formula | C42H45ClN6O5S2 |
| Molar mass | 813.43 g·mol−1 |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). |
References
- ^ Jump up to:a b c d e f Croce, Carlo M; Reed, John C (October 2016). “Finally, An Apoptosis-Targeting Therapeutic for Cancer”. Cancer Research. 76 (20): 5914–5920. doi:10.1158/0008-5472.CAN-16-1248. PMC 5117672. PMID 27694602.
- ^ Jump up to:a b Yosef, Reut; Pilpel, Noam; Tokarsky-Amiel, Ronit; Biran, Anat; Ovadya, Yossi; Cohen, Snir; Vadai, Ezra; Dassa, Liat; Shahar, Elisheva; Condiotti, Reba; Ben-Porath, Ittai; Krizhanovsky, Valery (2016). “Directed elimination of senescent cells by inhibition of BCL-W and BCL-XL”. Nature Communications. 7: 11190. Bibcode:2016NatCo…711190Y. doi:10.1038/ncomms11190. PMC 4823827. PMID 27048913.
- ^ Vogler, Meike, et al. “Bcl-2 inhibitors: small molecules with a big impact on cancer therapy.” Cell Death & Differentiation 16.3 (2008): 360–367.
- ^ Oltersdorf, Tilman; Elmore, Steven W.; Shoemaker, Alexander R.; Armstrong, Robert C.; Augeri, David J.; Belli, Barbara A.; Bruncko, Milan; Deckwerth, Thomas L.; Dinges, Jurgen; Hajduk, Philip J.; Joseph, Mary K.; Kitada, Shinichi; Korsmeyer, Stanley J.; Kunzer, Aaron R.; Letai, Anthony; Li, Chi; Mitten, Michael J.; Nettesheim, David G.; Ng, ShiChung; Nimmer, Paul M.; O’Connor, Jacqueline M.; Oleksijew, Anatol; Petros, Andrew M.; Reed, John C.; Shen, Wang; Tahir, Stephen K.; Thompson, Craig B.; Tomaselli, Kevin J.; Wang, Baole; Wendt, Michael D.; Zhang, Haichao; Fesik, Stephen W.; Rosenberg, Saul H. (2005). “An inhibitor of Bcl-2 family proteins induces regression of solid tumours”. Nature. 435 (7042): 677–81. Bibcode:2005Natur.435..677O. doi:10.1038/nature03579. PMID 15902208. S2CID 4335635.
- ^ Hann CL, Daniel VC, Sugar EA, Dobromilskaya I, Murphy SC, Cope L, Lin X, Hierman JS, Wilburn DL, Watkins DN, Rudin CM (April 2008). “Therapeutic efficacy of ABT-737, a selective inhibitor of BCL-2, in small cell lung cancer”. Cancer Research. 68 (7): 2321–8. doi:10.1158/0008-5472.can-07-5031. PMC 3159963. PMID 18381439.
- ^ Jump up to:a b Hauck P, Chao BH, Litz J, Krystal GW (April 2009). “Alterations in the Noxa/Mcl-1 axis determine sensitivity of small cell lung cancer to the BH3 mimetic ABT-737”. Mol Cancer Ther. 8 (4): 883–92. doi:10.1158/1535-7163.MCT-08-1118. PMID 19372561. Retrieved 9 September 2019.
///////////ABT-737, ABT 737
CN(CC[C@@H](NC1=CC=C(C=C1[N+]([O-])=O)S(NC(C2=CC=C(C=C2)N3CCN(CC3)CC4=CC=CC=C4C5=CC=C(C=C5)Cl)=O)(=O)=O)CSC6=CC=CC=C6)C

NEW DRUG APPROVALS
ONE TIME
$9.00
Tolebrutinib, SAR 442168
Tolebrutinib
SAR442168
- Treatment of Multiple Sclerosis (MS)
CAS 1971920-73-6
PRN 2246, example 3 [WO2016196840A1]
C26H25N5O3,
| 455.5 |
4-amino-3-(4-phenoxyphenyl)-1-[(3R)-1-prop-2-enoylpiperidin-3-yl]imidazo[4,5-c]pyridin-2-one
4-amino-3-(4-phenoxyphenyl)-1-[(3R)-1-prop-2-enoylpiperidin-3-yl]imidazo[4,5-c]pyridin-2-one
(R)-1-(1-Acryloylpiperidin-3-yl)-4-amino-3-(4-phenoxyphenyl)-1H-imidazo[4,5-c]pyridin-2(3H)-one
4-amino-3-(4-phenoxyphenyl)-1-[(3R)-1-(prop-2-
enoyl)piperidin-3-yl]-1,3-dihydro-2H-imidazo[4,5-
Tolebrutinib (R&D code SAR442168), developed by Principia and later acquired by Sanofi and included in its product line, Tolebrutinib is a BTK inhibitor used to treat cancer, autoimmune diseases such as multiple sclerosis and myasthenia gravis, inflammatory diseases and thromboembolic diseases, etc.,
Tolebrutinib is an orally bioavailable, brain-penetrant, selective, small molecule inhibitor of Bruton’s tyrosine kinase (BTK), with potential immunomodulatory and anti-inflammatory activities. Upon oral administration, tolebrutinib is able to cross the blood-brain barrier and inhibits the activity of BTK both peripherally and in the central nervous system (CNS). This prevents the activation of the B-cell antigen receptor (BCR) signaling pathway, and the resulting immune activation and inflammation. The inhibition of BTK activity also prevents microglial inflammatory signaling in the CNS, and the resulting immune activation, neuroinflammation and neurodegeneration. BTK, a cytoplasmic tyrosine kinase and member of the Tec family of kinases, plays an important role in B lymphocyte development, activation, signaling, proliferation and survival. In addition to B cells, BTK is also expressed in innate immune cells, including macrophages and microglia, and plays an important role in the regulation of microglial inflammatory signaling.
BTK, a member of the Tec family non-receptor tyrosine kinases, is essential for B cell signaling downstream from the B-cell receptor. It is expressed in B cells and other hematopoietic cells such as monocytes, macrophages and mast cells. It functions in various aspects of B cell function that maintain the B cell repertoire (see Gauld S. B. et al., B cell antigen receptor signaling: roles in cell development and disease. Science,
296: 1641 -2. 2002.) B cells pay a role in rheumatoid arthritis (see Perosa F., et ai, CD20-depleting therapy in autoimmune diseases: from basic research to the clinic. / Intern Med. 267:260-77. 2010 and Dorner T, et at. Targeting B cells in immune-mediated
inflammatory disease: a comprehensive review of mechanisms of action and identification of biomarkers. Pharmacol The 125:464-75. 2010 and Honigberg, L., et. ai, The selective BTK inhibitor PCI-32765 blocks B cell and mast cell activation and prevents mouse collagen indiced arthritis. Clin. Immunol. 127 SI :S 111. 2008) and in other autoimmune diseases such as systemic lupus erythematosus and cancers (see Shlomchik M. J., et. ai, The role of B cells in lpr/lpr-induced autoimmunity. /. Exp Med. 180:1295-1306. 1994; Honigberg L. A., The Braton tyrosine kinase inhibitor PCI-32765 blocks B-cell activation and is efficacious in models of autoimmune disease and B-cell malignancy. Proc. Natl. Acad. Sci. 107: 13075-80. 2010; and Mina-Osorio P, et al., Suppression of
glomerulonephritis in lupus-prone NZB x NZW mice by RN486, a selective inhibitor of Bruton’s tyrosine kinase. Arthritis Rheum. 65: 2380-91. 2013).
There is also potential for BTK inhibitors for treating allergic diseases (see Honigberg, L., et. al., The selective BTK inhibitor PCI-32765 blocks B cell and mast cell activation and prevents mouse collagen indiced arthritis. Clin. Immunol. 127 SI :S111. 2008). It was noted that the irreversible inhibitor suppresses passive cutaneous anaphylaxis (PCA) induced by IgE antigen complex in mice. These findings are in agreement with those noted with BTK-mutant mast cells and knockout mice and suggest that BTK inhibitors may be useful for the treatment of asthma, an IgE-dependent allergic disease of the airway.
Accordingly, compounds that inhibit BTK would be useful in treatment for diseases such as autoimmune diseases, inflammatory diseases, and cancer.
PATENT
PATENT
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2016196840
Example 3
Synthesis of (R)-l-(l-acryloylpiperidin-3-yl)-4-amino-3-(4-phenoxyphenyl)-lH- imidazo[4,5-c]pyridin-2(3H)-one
Into a 100-mL round-bottom flask, was placed (R)-4-amino-3-(4-phenoxyphenyl)-l-(piperidin-3-yl)-lH-imidazo[4,5-c]pyridin-2(3H)-one (150 mg, 0.37 mmol, 1.00 equiv), DCM-CH30H (6 mL), TEA (113 mg, 1.12 mmol, 3.00 equiv). This was followed by the addition of prop-2-enoyl chloride (40.1 mg, 0.44 mmol, 1.20 equiv) dropwise with stirring at OoC in 5 min. The resulting solution was stirred for 2 h at 0 °C. The resulting mixture was concentrated under vacuum. The residue was applied onto a silica gel column with dichloromethane/methanol (30: 1). The crude product (100 mg) was purified by Prep-HPLC with the following conditions (Column, XBridge Prep CI 8 OBD
Column,5um, 19*150mm; mobile phase, water with 0.05%TFA and ACN (25.0% ACN up to 45.0% in 8 min). 54.5 mg product of (R)-l-(l -acryloylpiperidin-3-yl)-4-amino-3-(4-phenoxyphenyl)-lH-imidazo[4,5-c]pyridin-2(3H)-one was obtained as a white solid. LC-MS m/z: 465.2 (M+l)
Step 2
Into a 25-mL round-bottom flask was placed tert-butyl (3R)-3-[4-[(E)-[(dimethy]amino)-methylidene]-amino]-2-oxo-3-(4-phenoxyphenyl)-lH,2H,3H-imidazo[4,5-c]pyridin-l -yl]piperidine- l-carboxylate (150 mg, 0.27 mmol, 1.00 equiv), 1,4-dioxane (6 mL), and hydrogen chloride (3 mL). The resulting solution was stirred overnight at 50° C. The reaction mixture was quenched with water. The pH of the solution was adjusted to 9 with sodium bicarbonate. The resulting solution was extracted with dichloromethane:CH3OH=10: 1 and the organic layers were combined. The resulting mixture was washed with sodium chloride and the organic layers were combined, dried over anhydrous sodium sulfate and concentrated under vacuum. The residue was applied onto a silica gel column and eluted with dichloromethane/methanol (30: 1) to give 80 mg (74%) of 4-amino-3-(4-phenoxyphenyl)-l -[(3R)-piperidin-3-yl]-lH,2H,3H-imidazo[4,5-c]pyridin-2-one as a light yellow solid.
////////

AS ON DEC2021 3,491,869 VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@amcrasto
/////////////////////////////////////////////////////////////////////////////
/////////Tolebrutinib, SAR 442168, PRN 2246, GTPL10625, BTK’168, EX-A4699, BDBM50557487, WHO 11268, Multiple Sclerosis, (MS),

NEW DRUG APPROVALS
ONE TIME
$10.00
RELACORILANT
Relacorilant
- Molecular FormulaC27H22F4N6O3S
- Average mass586.561 Da
CAS 1496510-51-0
Phase III
[(4aR)-1-(4-fluorophenyl)-6-(1-methylpyrazol-4-yl)sulfonyl-4,5,7,8-tetrahydropyrazolo[3,4-g]isoquinolin-4a-yl]-[4-(trifluoromethyl)pyridin-2-yl]methanone
релакорилант[Russian][INN]
ريلاكوريلانت[Arabic][INN]
瑞拉可兰[Chinese][INN]
- OriginatorCorcept Therapeutics
- ClassAntineoplastics; Fluorine compounds; Isoquinolines; Ketones; Organic sulfur compounds; Pyrazoles; Pyridines; Small molecules
- Mechanism of ActionGlucocorticoid receptor antagonists
- Orphan Drug StatusYes – Pancreatic cancer; Cushing syndrome
- Phase IIICushing syndrome; Ovarian cancer; Pancreatic cancer
- Phase IIFallopian tube cancer; Peritoneal cancer; Prostate cancer
- Phase I/IISolid tumours
- Phase IAdrenocortical carcinoma
Most Recent Events
- 09 Sep 2022Subgroup analysis efficacy data from a phase-II trial in Ovarian cancer presented at the 47th European Society for Medical Oncology Congress (ESMO-2022)
- 29 Jun 2022Phase-III clinical trials in Ovarian cancer (Combination therapy, Recurrent, Second-line therapy or greater) in USA (PO)
- 06 Jun 2022Corcept Therapeutics announces intentions to submit a NDA for Ovarian cancer
Relacorilant (developmental code name CORT-125134) is an antiglucocorticoid which is under development by Corcept Therapeutics for the treatment of Cushing’s syndrome.[1] It is also under development for the treatment of solid tumors and alcoholism.[1][2] The drug is a nonsteroidal compound and acts as an antagonist of the glucocorticoid receptor.[1] As of December 2017, it is in phase II clinical trials for Cushing’s syndrome and phase I/II clinical studies for solid tumors, while the clinical phase for alcoholism is unknown.[1]
Relacorilant is an orally available antagonist of the glucocorticoid receptor (GR), with potential antineoplastic activity. Upon administration, relacorilant competitively binds to and blocks GRs. This inhibits the activity of GRs, and prevents both the translocation of the ligand-GR complexes to the nucleus and gene expression of GR-associated genes. This decreases the negative effects that result from excess levels of endogenous glucocorticoids, like those seen when tumors overproduce glucocorticoids. In addition, by binding to GRs and preventing their activity, inhibition with CORT125134 also inhibits the proliferation of GR-overexpressing cancer cells. GRs are overexpressed in certain tumor cell types and promote tumor cell proliferation.
SCHEME
CLIP
https://europepmc.org/article/pmc/pmc8175224
Relacorilant (CORT125134)118) is being developed by Corcept Therapeutics, Inc. It is an orally active, high-affinity, selective antagonist of the glucocorticoid receptor that may benefit from the modulation of cortisol activity. In structural optimization, the introduction of a trifluoromethyl group to the 4-position on the pyridyl moiety was found to increase HepG2 tyrosine amino transferase assay potency by a factor of four. Relacorilant is currently being evaluated in a phase II clinical study in patients with Cushing’s syndrome.119)
2-Bromo-4-(trifluoromethyl)pyridine (17) prepared from (E)-4-ethoxy-1,1,1-trifluorobut-3-en-2-one is employed as a key intermediate for the preparation of relacorilant as shown in Scheme 31.120)

Scheme31. Synthesis of relacorilant.118)
118) H. Hunt, T. Johnson, N. Ray and I. Walters (Corcept Therapeutics, Inc.): PCT Int. Appl. WO2013/177559 (2013).
119) H. J. Hunt, J. K. Belanoff, I. Walters, B. Gourdet, J. Thomas, N. Barton, J. Unitt, T. Phillips, D. Swift and E. Eaton: Identification of the Clinical Candidate (R)-(1-(4-Fluorophenyl)-6-((1-methyl-1H-pyrazol-4-yl)sulfonyl)-4,4a,5,6,7,8-hexahydro-1H-pyrazolo[3,4-g]isoquinolin-4a-yl)(4-(trifluoromethyl)pyridin-2-yl)methanone (CORT125134): A Selective Glucocorticoid Receptor (GR) Antagonist. J. Med. Chem. 60, 3405–3421 (2017). [Abstract] [Google Scholar]
120) B. Lehnemann, J. Jung and A. Meudt (Archimica GmbH): PCT Int. Appl. WO 2007/000249 (2007).
PAPER
https://pubs.acs.org/doi/abs/10.1021/acs.jmedchem.7b00162
The nonselective glucocorticoid receptor (GR) antagonist mifepristone has been approved in the U.S. for the treatment of selected patients with Cushing’s syndrome. While this drug is highly effective, lack of selectivity for GR leads to unwanted side effects in some patients. Optimization of the previously described fused azadecalin series of selective GR antagonists led to the identification of CORT125134, which is currently being evaluated in a phase 2 clinical study in patients with Cushing’s syndrome.

PATENT
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2013177559


SYN

////////

AS ON DEC2021 3,491,869 VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@amcrasto
/////////////////////////////////////////////////////////////////////////////
Cushing’s syndrome (CS) is a metabolic disorder caused by chronic hypercortisolism. CS is associated with cardiovascular, metabolic, skeletal and psychological dysfunctions and can be fatal if left untreated. The first-line treatment for all forms of CS is a surgery. However, medical therapy has to be chosen if surgical resection is not an option or is deemed ineffective. Currently available therapeutics are either not selective and have side effects or are only available as an injection (pasireotide).
References
- ^ Jump up to:a b c d “Relacorilant – Corcept Therapeutics – AdisInsight”.
- ^ Veneris JT, Darcy KM, Mhawech-Fauceglia P, Tian C, Lengyel E, Lastra RR, Pejovic T, Conzen SD, Fleming GF (2017). “High glucocorticoid receptor expression predicts short progression-free survival in ovarian cancer”. Gynecol. Oncol. 146 (1): 153–160. doi:10.1016/j.ygyno.2017.04.012. PMC 5955699. PMID 28456378.
External links
| Clinical data | |
|---|---|
| Other names | CORT-125134 |
| Routes of administration | By mouth |
| Drug class | Antiglucocorticoid |
| Identifiers | |
| showIUPAC name | |
| CAS Number | 1496510-51-0 |
| PubChem CID | 73051463 |
| ChemSpider | 57617720 |
| UNII | 2158753C7E |
| KEGG | D11336 |
| Chemical and physical data | |
| Formula | C27H22F4N6O3S |
| Molar mass | 586.57 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| showSMILES | |
| showInChI |
//////////////Relacorilant, Phase III , Orphan Drug, Cushing syndrome, Ovarian cancer, Pancreatic cancer, релакорилант , ريلاكوريلانت , 瑞拉可兰 ,
CN1C=C(C=N1)S(=O)(=O)N2CCC3=CC4=C(CC3(C2)C(=O)C5=NC=CC(=C5)C(F)(F)F)C=NN4C6=CC=C(C=C6)F

NEW DRUG APPROVALS
ONE TIME
$10.00
MILADEMETAN
Milademetan
| Molecular Weight | 618.53 |
|---|---|
| Formula | C30H34Cl2FN5O4 |
| CAS No. | 1398568-47-2 |
Milademetan. hcl
Chemical Formula: C30H35Cl3FN5O4
Exact Mass: 617.1972
Molecular Weight: 654.99
Elemental Analysis: C, 55.01; H, 5.39; Cl, 16.24; F, 2.90; N, 10.69; O, 9.77
1398568-47-2 (free base) 1398569-75-9 (tosylate) 2095625-97-9 (tosylate hydrate) Milademetan HCl
DS3032b; DS-3032b; DS 3032b; DS3032; DS-3032; DS 3032; DS-3032b tosylate; Milademetan tosylate; Milademetan HCl
(3’R,4’S,5’R)-N-[(3R,6S)-6-carbamoyloxan-3-yl]-6”-chloro-4′-(2-chloro-3-fluoropyridin-4-yl)-4,4-dimethyl-2”-oxo-1”,2”-dihydrodispiro[cyclohexane-1,2′-pyrrolidine-3′,3”-indole]-5′-carboxamide hydrochloride
orphan drug, UNII:R3I80TLN7S, миладеметан , ميلاديميتان , 米拉美坦
(3’R,4’S,5’R)-N-((3R,6S)-6-Carbamoyltetrahydro-2H-pyran-3-yl)-6”-chloro-4′-(2-chloro-3-fluoro-4-pyridinyl)-4,4-dimethyl-2”-oxo-1”,2”-dihydrodispiro(cyclohexane-1,2′-pyrrolidine-3′,3”-indole)-5′-carboxamide
milademetan
rolontis
SPI-2012
Milademetan, also known as DS-3032b or DS-3032, is a potent and selective MDM2 inhibitor with potential antineoplastic activity. Upon oral administration, MDM2 inhibitor DS-3032b binds to, and prevents the binding of MDM2 protein to the transcriptional activation domain of the tumor suppressor protein p53. By preventing this MDM2-p53 interaction, the proteosome-mediated enzymatic degradation of p53 is inhibited and the transcriptional activity of p53 is restored. This results in the restoration of p53 signaling and leads to the p53-mediated induction of tumor cell apoptosis.
DS-3032 (Milademetan) is an orally available, potent and selective inhibitor of the p53-MDM2 (murine double minute 2) interaction. Milademetan binds to, and prevents the binding of MDM2 protein to the transcriptional activation domain of the tumor suppressor protein p53. Milademetan is 10-fold more potent than the first-generation inhibitor nutlin-3a. By preventing this MDM2-p53 interaction, the proteasome-mediated enzymatic degradation of p53 is inhibited and the transcriptional activity of p53 is restored. This results in the restoration of p53 signaling and leads to the p53-mediated induction of tumor cell apoptosis. DS-3032 is currently being evaluated in three phase 1 clinical trials for solid and hematological malignancies, including acute myeloid leukemia (AML), acute lymphocytic leukemia (ALL), chronic myeloid leukemia (CML) in blast phase, lymphoma and myelodysplastic syndrome (MDS).
- OriginatorRigel Pharmaceuticals
- DeveloperDaiichi Sankyo Inc; National Cancer Center Hospital East; Rain Therapeutics; University of Texas M. D. Anderson Cancer Center
- ClassAntineoplastics; Cyclohexanes; Indoles; Pyrrolidines; Small molecules
- Mechanism of ActionProto-oncogene protein c mdm2 inhibitors
- Orphan Drug StatusYes – Liposarcoma
- Phase IIILiposarcoma
- Phase IISarcoma; Solid tumours
- Phase I/IIAcute myeloid leukaemia
- Phase ILymphoma; Myelodysplastic syndromes
- PreclinicalMesothelioma
- No development reportedMultiple myeloma
- 10 Aug 2022Rain Therapeutics completes enrolment in phase-III clinical trials in Liposarcoma in (Inoperable/Unresectable, Metastatic disease, Second-line therapy or greater) in United Kingdom, Taiwan, Spain, Poland, South Korea, Italy, Hong Kong, Germany, Georgia, France, Canada, Belgium, Austria (PO) (NCT04979442)
- 09 Jun 2022Efficacy, adverse events and pharmacodynamics data from phase I/II trial in Acute myeloid leukemia presented at the 27th Congress of the European Haematology Association(EHA-2022)
- 04 May 2022Rain Therapeutics plans a phase I/II MANTRA-4 trial in Solid tumours (Combination therapy, Late-stage disease) in Second half of 2022
PATENT
WO2015033974

[Example 2]
Ethyl (3’R,4’S,5’R)-6”-chloro-4′-(3-chloro-2-fluorophenyl)-4,4-dimethyl-2”-oxo 1″,2″-dihydrodispiro[cyclohexane-1,2′-pyrrolidine-3′,3″-indole]-5′-carboxylate
[0202]
[Chem. 58]
[0203]
(3E/Z)-6-chloro-3-(3-chloro-2-fluorobenzylidene)-1,3-dihydro-2H-indol-2-one ( WO 2006/091646) (98.7) under nitrogen atmosphere mg), (R)-BINAP (12.1 mg, 0.019 mmol), CuOAc (2.0 mg, 0.016 mmol), 4,4-dimethylcyclohexanone (61.4 mg, 0.48 mmol), glycine ethyl ester. (39.5 μL, 0.39 mmol) and a solution of triethylamine (6.8 μL, 0.049 mmol) in N,N-dimethylacetamide (2.0 mL) were added and stirred at room temperature for 22 hours. Ethyl acetate (2 mL), water (1 mL), and 20% aqueous ammonium chloride solution (1 mL) were added to the reaction mixture, and the mixture was vigorously stirred to separate the organic layer. The aqueous layer was extracted twice with ethyl acetate (2 mL each) and all the organic layers were combined and then washed with water three times (5 mL each). The obtained organic layer was concentrated under reduced pressure, ethyl acetate (6 mL) and silica gel (500 mg) were added to the residue, and the silica gel was separated by filtration. The filtrate was concentrated under reduced pressure, ethanol (1.0 mL) was added to the residue, water (1 mL) was added dropwise, and the mixture was stirred overnight at room temperature. The precipitated solid was filtered and dried under reduced pressure at 40° C. to obtain the title compound (137 mg, yield 82%, 94% ee) as a solid.
1H NMR (500 MHz, CDCl3): δ = 0.67 (s, 3H), 0.91 (s, 3H), 1.10-1.19 (m, 2H), 1.17 (t, J=7.3 Hz, 3H), 1.25-1.33 (m, 1H), 1.44- 1.72 (m, 3H), 1.87-2.01 (m, 1H), 3.16 (s, 1H), 4.07-4.21 (m, 2H), 4.52 (d, J = 8.5 Hz, 1H), 4.83 (d, J = 8.5 Hz, 1H), 6.74 (d, J = 1.5Hz, 1H), 6.81-6.86 (m, 1H), 7.06 (dd, J = 8.3, 2.8 Hz, 1H), 7.10-7.16 (m, 1H), 7.37 (dd, J = 8.3, 1.8 Hz, 1H), 7.48-7.54 (m, 1H), 7.81 (s, 1H).
(HPLC conditions for optical purity determination)
カラム: CHIRALPAK OD-3R 4.6 × 150 mm, 3μm
Moving layer: 10mM Rinic acid buffer: MeCN = 40:60
Flow rate: 1.0 min/min
カラム Temperature: 40°C
Exhaust wavelength: 254 nm
Injection volume: 5 μL
Hold time: Labeling compound = 13.8 min, エナンチオマー= 12.9 min
[Example 11]
11-1) Effects of various asymmetric catalysts
[0230]
[Chem. 67]
[0231]
(3E/Z)-6-chloro-3-[(2-chloro-3-fluoropyridin-4-yl)methylene]-1,3-dihydro-2H-indol-2-one ( WO 2012 / 121361), 4,4-dimethylcyclohexanone (1.5 eq.), glycine ethyl ester (1.2 eq.), triethylamine (15 mol%) in THF solution (10 times the volume), separately, Lewis acid (5 mol%) , an asymmetric ligand (6 mol %) and THF (10 times the amount) were stirred for 1 hour under a nitrogen atmosphere, a catalyst solution prepared was added, and the mixture was stirred at room temperature for 12 to 16 hours. After that, the resulting trans1 compound ((ethyl (3′S,4′R,5′S)-6″-chloro-4′-(2-chloro-3-fluoropyridin-4-yl) -4,4-dimethyl-2”-oxo-1”,2”-dihydrodispiro[cyclohexane-1,2′-pyrrolidine-3′,3”-indole]-5′-carboxylate) Optical purity and HPLC yield were measured.
(HPLC conditions for measuring optical purity)
Column: CHIRALPAK OD-3R 4.6 × 150 mm, 3 µm
Mobile phase: 10 mM phosphoric acid buffer: MeCN = 40:60
Flow rate: 1.0 min/min
column Temperature: 40°C
Detection wavelength: 254 nm
Injection volume: 5 µL
Retention time: Title compound = 13.8 min, enantiomer = 12.9 min
Main results are shown in Table 1.
[0232]
[Table 1-1]
[Table 1-2]
[0233]
11-2) Effects of various solvents
[0234]
[Chem. 68]
[0235]
(3E/Z)-6-chloro-3-[(2-chloro-3-fluoropyridin-4-yl)methylene]-1,3-dihydro-2H-indol-2-one ( WO 2012 / 121361), 4,4-dimethylcyclohexanone (1.5 eq.), glycine ethyl ester (1.2 eq.), triethylamine (15 mol%), a solvent (10 times the amount), CuOAc (5 mol%), ( A catalyst solution prepared by stirring S)-BINAP (6 mol %) and a solvent (10 times the amount) under a nitrogen atmosphere for 1 hour was added, followed by stirring at room temperature for 21.5 hours. After that, by HPLC, the resulting trans2 compound (ethyl (3’S,4’R,5’S)-6”-chloro-4′-(2-chloro-3-fluoropyridin-4-yl)- HPLC of 4,4-dimethyl-2″-oxo-1″,2″-dihydrodispiro[cyclohexane-1,2′-pyrrolidine-3′,3″-indole]-5′-carboxylate) Yields and optical purities were determined.
Table 2 shows the main results.
[0236]
[Table 2]
11-3) Examination of Cu(I) Lewis acid
PATENT
WO2014038606
WO2014038606 CLICK HERE
Example 1
[0062]
[Chem.3]
[0063]
(3′R,4′S,5′R)-N-[(3R,6S)-6-carbamoyltetrahydro-2H-pyran-3-yl]-6″-chloro-4′-(2-chloro- 3-fluoropyridin-4-yl)-4,4-dimethyl-2″-oxo-1″,2″-dihydrodispiro[cyclohexane-1,2′-pyrrolidine-3′,3″-indole]-5 ‘
-Carboxamide The compound (35 mg, 0.24 mmol) obtained in Reference Example 2, Step 3 was added to a solution of the compound (100 mg, 0.20 mmol) obtained in Step 3 of Reference Example 1 in N,N-dimethylformamide (4 ml). , triethylamine (0.04 ml, 0.30 mmol), 1-hydroxybenzotriazole (27 mg, 0.20 mmol) and 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide hydrochloride (46 mg, 0.24 mmol) were added. , and stirred for 1 hour at 50° C. After allowing to cool, the reaction solution was diluted with ethyl acetate, washed successively with water, saturated aqueous sodium hydrogencarbonate solution and saturated brine, and dried over anhydrous sodium sulfate. After evaporating the solvent under reduced pressure, the residue was purified by NH-silica gel column chromatography [chloroform:methanol=50:1 (v/v)]. After stirring for 24 hours at rt, the solvent was distilled off under reduced pressure to obtain 94 mg (76%) of the title compound as a solid.1H
– NMR (400 MHz, CDCl3 ) .) δ: 0.68 (3H, s), 0.95 (3H, s), 1.11-1.27 (2H, m), 1.35-1.81 (8H, m), 2.10-2.17 (1H, m), 2.25-2.32 (1H, m), 3.15(1H,t,J=10.5Hz), 3.27(1H,br s), 3.80(1H,dd,J=11.0,2.3Hz), 3.85-3.95(1H,m), 4.13(1H, ddd,J=10.8,4.5,1.3Hz),4.44(1H,d,J=9.2Hz),4.64(1H,d,J=9.2Hz),5.46(1H,d,J=3.7Hz),6.49( 1H,d,J=3.7Hz), 6.74(1H,d,J=1.8Hz), 7.07(1H,dd,J=8.2,1.8Hz), 7.31(1H,dd,J=8.2,2.3Hz), 7.48-7.52(2H,m),7.62(1H,s),8.05(1H,d,J=5.5Hz).MS
(ESI)m/z:618(M+H) +
Reference example 1
[0087]
[Chem.4]
[0088]
[Step 1] (3E/Z)-6-chloro-3-[(2-chloro-3-fluoropyridin-4-yl)methylene]-1,3-dihydro-2H-indol-2-one
6-chloro -1,3-dihydro-2H-indol-2-one (2.20 g, 13.11 mmol) and 2-chloro-3-fluoroisonicotinaldehyde (2.20 g, 13.8 mmol) in methanol (130 ml). , N,N-diisopropylethylamine (0.46 ml, 2.63 mmol) was added, and the mixture was heated under reflux for 16 hours. After standing to cool, the precipitate was collected by filtration, washed with cold methanol and dried to obtain 3.37 g (83%) of the title compound as a solid.
MS(APCI) m/z: 309(M+H) + .
[0089]
[Step 2] (3′S,4′R,7′S,8′S,8a′R)-6″-chloro-8′-(2-chloro-3-fluoropyridin-4-yl)-4 ,4-dimethyl-3′,4′-diphenyl-3′,4′,8′,8a′-tetrahydro-1′H-dispiro[cyclohexane-1,6′-pyrrolo[2,1-c][1 ,4]oxazine-7′,3″-indole]-1′,2″(1″H)
-dione Under a nitrogen atmosphere, the compound obtained in Step 1 (1.86 g, 6.00 mmol), (5R,6S )-5,6-diphenylmorpholin-2-one (1.67 g, 6.60 mmol) and 4,4-dimethylcyclohexanone (0.83 g, 6.60 mmol) in tetrahydrofuran (30 ml) was added with diethyl boron trifluoride. An ether complex (0.15 ml, 1.20 mmol) and molecular sieve 4A (powder) (3 g) were added, and the mixture was heated and stirred at 70° C. for 7 days. After allowing to cool, insoluble matter was removed by filtration through celite, and the filtrate was washed with saturated brine and dried over anhydrous sodium sulfate. The solvent was distilled off under reduced pressure and purified by silica gel column chromatography [n-hexane:ethyl acetate=4:1→1:1 (v/v)] to obtain 3.39 g (84%) of the title compound as a solid. rice field.
1 H-NMR (400 MHz, CDCl3) δ: 0.21 (3H, s), 0.53 (3H, s), 0.89-1.08 (3H, m), 1.28-1.43 (3H, m), 1.73-1.81 (1H, m), 2.23-2.33 (1H, m), 4.58 (1H, d, J = 11.0Hz), 4.86 (1H, d, J = 3.2Hz), 5.31 (1H, d, J = 11.0Hz), 6.25 (1H, d, J = 8.3Hz) ,6.67(1H,dd,J=8.3,1.8Hz),6.72-6.77(2H,m),6.93(1H,d,J=1.8Hz),7.04-7.17(6H,m),7.18-7.25(3H ,m),7.79(1H,t,J=4.6Hz),7.99(1H,s),8.29(1H,d,J=5.0Hz).MS
(APCI)m/z:670(M+H) + .
[0090]
[Step 3] (4′S,5′R)-6″-chloro-4′-(2-chloro-3-fluoropyridin-4-yl)-4,4-dimethyl-2″-oxo-1″ ,2″-dihydrodispiro[cyclohexane-1,2′-pyrrolidine-3′,3″-indole]-5′-carboxylic acid
The compound obtained in step 2 (630 mg, 0.94 mmol) was treated with acetonitrile (10 ml). Dissolve in water (4 ml), add potassium carbonate (130 mg, 0.94 mmol) and heat under reflux for 16 hours at 85° C. After allowing to cool, add anhydrous magnesium sulfate (113 mg, 0.94 mmol) and stir at room temperature for 15 minutes. After extraction with ethyl acetate, the organic layer was washed with saturated brine and dried over anhydrous magnesium sulfate. (2-chloro-3-fluoropyridin-4-yl)-1′-[(1R,2S)-2-hydroxy-1,2-diphenylethyl]-4,4-dimethyl-2″-oxo-1″ ,2″-dihydrodispiro[cyclohexane-1,2′-pyrrolidine-3′,3″-indole]-5′-carboxylic acid (650 mg, 100%) was obtained as a solid [MS (ESI) m/z :688(M+H) +]. The resulting carboxylic acid (650 mg, 0.94 mmol) was dissolved in methanol (30 ml) and water (8 ml), and diammonium cerium (IV) nitrate (1.55 g, 2.82 mmol) was added under ice-cooling. Stir at room temperature for 30 minutes. Potassium carbonate (780 mg, 5.64 mmol) was added under ice-cooling, and the mixture was stirred at the same temperature for 1 hour. After removing the insoluble matter by filtration through celite, the filtrate was concentrated under reduced pressure, water was added to the resulting residue, and the mixture was extracted with ethyl acetate. The organic layer was washed with saturated brine and dried over anhydrous sodium sulfate. The solvent was distilled off under reduced pressure, and the resulting residue was purified by silica gel column chromatography [chloroform:methanol=20:1→4:1 (v/v)] to obtain 152 mg (33%) of the title compound as a solid. .
1 H-NMR (500 MHz, CD 3 OD) δ: 0.74 (3H, s), 0.9 (3H, s), 1.29-1.44 (2H, m), 1.48-1.58 (2H, m), 1.64-1.76 (1H ,m),1.94-2.02(1H,m),2.11(1H,ddd,J=14.0,14.0,4.0Hz),2.43-2.53(1H,m),5.07(1H,d,J=10.3Hz), 5.32(1H,d,J=10.3Hz),6.84(1H,d,J=1.7Hz),7.16(1H,dd,J=8.3,2.0Hz),7.63(1H,dd,J=8.0,2.3Hz) ),7.75(1H,t,J=5.2Hz),8.15(1H,d,J=5.2Hz).
MS(ESI)m/z:492(M+H) + .
[0091]
Reference example 2
[0092]
[Chem.5]
[0093]
[Step 1] Methyl 2,6-anhydro-3,4,5-trideoxy-5-(dibenzylamino)-L-erythro
-hexonate 2,6-anhydro-3,4,5-trideoxy-5-( dibenzylamino)-L-erythro-hexonate methyl 2,6-anhydro-3,4,5-trideoxy-5-(dibenzylamino)-L-erythro-hexonate (1.60 g, 4.70 mmol) was The mixture was dissolved in methanol (30 ml), 1N aqueous sodium hydroxide solution (10 ml) was gradually added under ice-cooling, and the mixture was stirred at room temperature for 3 hours. Dowex 50W-X8 was added to the reaction mixture to adjust the pH to 5 to 6, insoluble materials were removed by filtration, and the filtrate was concentrated under reduced pressure to obtain 1.7 g (100%) of the title compound as a solid.
1 H-NMR (400 MHz, CDCl 3 ) δ: 1.18-1.26(1H,m), 1.36-1.48(1H,m), 1.79-1.97(2H,m), 2.62(1H,t,J=11.0Hz) ,3.18(1H,t,J=10.4Hz),3.40(1H,d,J=11.5Hz),3.51-3.61(4H,m),3.90-3.99(1H,m),7.12-7.38(10H,m ).
MS(ESI)m/z:326(M+H) + .
[0094]
[Step 2] (2S,5R)-5-(dibenzylamino)tetrahydro-2H-pyran-2-carboxamide
The compound (870 mg, 2.67 mmol) obtained in Step 1 above was dissolved in N,N-dimethylformamide (30 ml). 1-hydroxybenzotriazole (361 mg, 2.67 mmol) and 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide hydrochloride (614 mg, 3.20 mmol) were added and stirred at room temperature for 15 minutes. Ammonium chloride (285 mg, 5.44 mmol) and N,N-diisopropylethylamine (1.86 ml, 10.7 mmol) were added and stirred at room temperature for 8 hours. After diluting with ethyl acetate, the organic layer was washed with saturated aqueous sodium hydrogencarbonate solution and saturated brine in that order, and dried over anhydrous sodium sulfate. The solvent was removed under reduced pressure to give 495 mg (57%) of the title compound as a solid.
1 H-NMR (400 MHz, CDCl 3 ) δ: 1.35-1.45 (1H, m), 1.60-1.70 (1 H, m), 2.10-2.18 (1 H, m), 2.21-2.28 (1 H, m), 2.76 ( 1H,tt,J=11.4,4.0Hz),3.44(1H,t,J=10.9Hz),3.67(4H,q,J=14.2Hz),3.71-3.73(1H,m),4.04(1H,dq ,J=11.0,2.1Hz),5.35(1H,s),6.40(1H,s),7.21-7.36(10H,m).MS
(ESI)m/z:325(M+H) + .
[0095]
[Step 3] (2S,5R)-5-aminotetrahydro-2H-pyran-2-carboxamide
The compound (490 mg, 1.51 mmol) obtained in Step 2 above was dissolved in ethanol (10 ml) and treated with 20% palladium hydroxide. (100 mg) was added, and the mixture was stirred at room temperature for 16 hours under a hydrogen atmosphere. After removing the catalyst by filtration through celite, the filtrate was distilled off under reduced pressure and dried to obtain 215 mg (99%) of the title compound as a solid.
1 H-NMR (400 MHz, DMSO-d 6 ) δ: 1.11-1.22(1H,m), 1.25-1.35(1H,m), 1.83-1.91(2H,m), 2.51-2.60(1H,m), 2.90(1H,t,J=10.5Hz),3.52(1H,d,J=11.9Hz),
3.78-3.84 (1H,m),6.99(1H,br s),7.09(1H,br s). (ESI) m/z: 145(M+H) + .
PATENT
WO2012121361
PATENT
WO2015033974
PAPER
https://pubs.acs.org/doi/10.1021/acs.oprd.2c00192
Abstract

Herein, we report the structure and synthesis of the potent MDM2-p53 inhibitor BI-0282. The complex spirooxindole scaffold bearing four stereocenters embedded in a rigid polycyclic ring-system was effectively prepared on a multi-gram scale in only five synthesis steps employing a three-component 1,3-dipolar cycloaddition and a late-stage Davis–Beirut reaction as key steps.
Compound 1
Intermediate 10 (28.8 g, 44.8 mmol) is dissolved in isopropanol (300 mL) and a solution of potassium hydroxide (39.0 g, 694.9 mmol) in water (95 mL) is slowly added. After stirring for 16 h at ambient temperature, the solvents are partially removed under reduced pressure. The residue is diluted with ethyl acetate and treated with a diluted aqueous solution of citric acid. After extraction of the aqueous layer with ethyl acetate, the organic layers are combined, dried with sodium sulfate, and the solvent is removed under reduced pressure. Purification by normal phase column chromatography using dichloromethane and methanol as solvents yields rac-1 (25.8 g, 43.5 mmol) in 70% yield as an amorphous white solid.
Chiral SFC and subsequent purification by reversed phase column chromatography using acetonitrile and methanol as solvents furnishes 1 (BI-0282).
Rac-1 (60 g, 93,3 mmol) was separated by chiral SFC and reversed phase column chromatography to obtain 1 (24.4 g, 40,0 mmol, 43%) as an amorphous white solid.
Chiral HPLC (CHIRALPAK, heptane/isopropanol/trifluoroacetic acid = 70/30/0.1, flow rate 1.0 mL/min, I = 240 mM) tR = 7.8 min (1), and 11.1 min (ent-1). Preparative SFC (CHIRALPAK, carbon dioxide/(isopropanol + 1% diethylamine) = 70/30, flow rate 300 g/min, I = 290 nM).
1H NMR (500 MHz, DMSO-d6): δ 12.64 (br s, 1H), 10.29 (s, 1H), 7.67 (s, 1H), 7.47 (d, J = 8.83 Hz, 2H), 7.29–7.36 (m, 1H), 7.26 (d, J = 7.88 Hz, 1H), 7.21 (dd, J = 1.26, 8.83 Hz, 1H), 7.12 (t, J = 8.04 Hz, 1H), 6.92 (dd, J = 1.89, 7.88 Hz, 1H), 6.48 (d, J = 1.89 Hz, 1H), 5.86 (t, J = 9.14 Hz, 1H), 4.59–4.68 (m, 1H), 4.52 (dd, J = 7.88, 11.35 Hz, 1H), 4.23–4.32 (m, 1H), 4.20 (d, J = 10.09 Hz, 1H), 2.27 (dd, J = 7.57, 13.08 Hz, 1H), 2.13 (dd, J = 5.83, 13.08 Hz, 1H), 0.47–0.62 (m, 1H), 0.26–0.37 (m, 1H), 0.11–0.20 (m, 1H), −0.04 to 0.04 (m, 1H), −0.25 (s, 1H).
13C{1H} NMR (125 MHz, DMSO-d6): δ 177.5, 168.1, 156.1 (d, 1JC,F = 248.7 Hz), 146.3, 145.3, 144.0, 134.1, 130.3, 129.7, 129.5, 126.8, 126.7, 125.4 (d, 3JC,F = 4.4 Hz), 123.5 (d, 2JC,F = 13.2 Hz), 122.5, 120.0, 119.9, 119.7 (d, 2JC,F = 18.3 Hz), 118.7, 110.0, 107.3, 76.4, 69.2, 57.5, 56.8, 54.2, 51.2, 11.6, 5.5, 4.1.
HRMS (ESI) m/z: [M + H]+ calcd for C30H24Cl2FN4O4, 593.1153; found, 593.1165.
////////

AS ON DEC2021 3,491,869 VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@amcrasto
/////////////////////////////////////////////////////////////////////////////
Milademetan is under investigation in clinical trial NCT02319369 (Safety, Tolerability and Pharmacokinetics of Milademetan Alone and With 5-Azacitidine (AZA) in Acute Myelogenous Leukemia (AML) or High-Risk Myelodysplastic Syndrome (MDS)).
- [1]. ARYL SULFONOHYDRAZIDES. WO 2017069289 A1.[2]. M.M. Gounder, et al. Milademetan, an oral MDM2 inhibitor, in well-differentiated/dedifferentiated liposarcoma: results from a phase 1 study in patients with solid tumors or lymphomas. European Journal of Cancer 138S2 (2020) S1–S62.[3]. Li, Yangbing, et al. Development of novel PROTAC Small-Molecule Degraders of MDM2 Protein and Peptidomimetic Inhibitors Targeting WDR5-MLL1 Protein-Protein Interaction.[4]. Viktor Arnhold, et al. Reactivating TP53 signaling by the novel MDM2 inhibitor DS-3032b as a therapeutic option for high-risk neuroblastoma. ncotarget. 2018 Jan 5; 9(2): 2304–2319.
/////////Milademetan, DS3032b, DS-3032b, DS 3032b, DS3032, DS-3032, DS 3032, DS-3032b tosylate, Milademetan tosylate, Milademetan HCl, orphan drug, UNII:R3I80TLN7S, миладеметан , ميلاديميتان , 米拉美坦
CC1(C)CCC2(CC1)N[C@H]([C@H](C1=C(F)C(Cl)=NC=C1)[C@]21C(=O)NC2=CC(Cl)=CC=C12)C(=O)N[C@@H]1CC[C@H](OC1)C(N)=O

NEW DRUG APPROVALS
ONE TIME
$10.00

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....














