ACT-132577, Aprocitentan

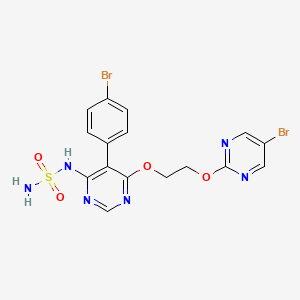
ACT-132577, Aprocitentan,
1103522-45-7
546.19, C16H14Br2N6O4S
3/19/2024 FDA APPROVED, To treat hypertension, Tryvio
N-[5-(4-bromophenyl)-6-{2-[(5-bromopyrimidin-2-yl)oxy]ethoxy}pyrimidin-4-yl]aminosulfonamide
Aprocitentan, sold under the brand name Tryvio, is a medication used to treat hypertension (high blood pressure).[1] It is developed by Idorsia.[2] It is taken by mouth.[1]
Aprocitentan is a dual endothelin-1 antagonist that targets both endothelin A and endothelin B receptors.[3][4]
Aprocitentan was approved for medical use in the United States in March 2024.[1][2][5] It is the first endothelin receptor antagonist to be approved by the US Food and Drug Administration (FDA) to treat systemic hypertension.[2]
Medical uses
Aprocitentan is indicated for the treatment of hypertension in combination with other antihypertensive drugs, to lower blood pressure in adults who are not adequately controlled on other medications.[1]
Adverse effects
Aprocitentan may cause hepatotoxicity (liver damage), edema (fluid retention), anemia (reduced hemoglobin), and decreased sperm count.[1]
Contraindications
Data from animal reproductive toxicity studies with other endothelin-receptor agonists indicate that use is contraindicated in pregnant women.[1]
Mechanism of action
Aprocitentan is an endothelin receptor agonist that inhibits the protein endothelin-1 from binding to endothelin A and endothelin B receptors.[1][4] Endothelin-1 mediates various adverse effects via its receptors, such as inflammation, cell proliferation, fibrosis, and vasoconstriction.[1]
Society and culture
Economics
Aprocitentan is developed by Idorsia, which sold it to Janssen and purchased the rights back in 2023, for US$343 million.[6]
Legal status
Aprocitentan was approved for medical use in the United States in March 2024.[1]
In April 2024, the Committee for Medicinal Products for Human Use (CHMP) of the European Medicines Agency adopted a positive opinion, recommending the granting of a marketing authorization for the medicinal product Jeraygo, intended for the treatment of resistant hypertension in adults.[7] The applicant for this medicinal product is Idorsia Pharmaceuticals Deutschland GmbH.[7]
SYN
US Patent
Trade Name
Application Number
Applicant
IDORSIA PHARMACEUTICALS LTD
IDORSIA PHARMACEUTICALS LTD
IDORSIA PHARMACEUTICALS LTD
IDORSIA PHARMACEUTICALS LTD
IDORSIA PHARMACEUTICALS LTD
PATENT
WO 02/053557
PATENT
Martin Bolli, Christoph Boss, Alexander Treiber. ” 4-pyrimidinesulfamide derivative “, US Patent US8324232B2.
EXAMPLE
Preparation A: Benzylsulfamide Potassium Salt
A.i. Benzylsulfamide
| 1H NMR (D 6-DMSO): δ 4.05 (d, J=6.4 Hz, 2H); 6.60 (s, 2H); 7.04 (s, J=6.4 Hz, 1H); 7.20-7.36 (m, 5H). |
A.ii. Benzylsulfamide Potassium Salt
Preparation B: 5-(4-bromo-phenyl)-4,6-dichloro-pyrimidine
B.i. 4-bromophenylacetic acid methyl ester
B.ii. 2-(4-bromophenyl)-malonic acid dimethyl ester
B.iii. 5-(4-bromophenyl)-pyrimidine-4,6-diol
B.iv. 5-(4-bromo-phenyl)-4,6-dichloro-pyrimidine
Example 1
{5-(4-bromo-phenyl)-6-[2-(5-bromo-pyrimidin-2-yloxy)-ethoxy]-pyrimidin-4-yl}-sulfamide
1.i. Benzyl-sulfamic acid [6-chloro-5-(4-bromophenyl)-pyrimidin-4-yl]-amide
| 1H NMR (CDCl 3): δ 4.23 (d, J=5.9 Hz, 2H); 5.94 (t br., J=6 Hz, 1H); 7.05 (d, J=8.2 Hz, 2H); 7.20-7.35 (m, 5H); 7.68 (d, J=8.2 Hz, 2H); 8.61 (s, 1H). |
1.ii. Benzyl-sulfamic acid [5-(4-bromophenyl)-6-(2-hydroxyethoxy)pyrimidin-4-yl]-amide
1.iii Benzyl-sulfamic acid [5-(4-bromophenyl)-6-{2-(5-bromo-pyrimidin-2-yloxy)-ethoxy}-pyrimidin-4-yl]-amide
1.iv. {5-(4-bromo-phenyl)-6-[2-(5-bromo-pyrimidin-2-yloxy)-ethoxy]-pyrimidin-4-yl}-sulfamide
| 1H NMR (CDCl 3): δ 4.60-4.65 (m, 2H), 4.71-4.74 (m, 2H), 5.50 (s br, 2H), 7.10 (s br, 1H), 7.13-7.17 (m, 2H), 7.55-7.59 (m, 2H), 8.49 (s, 2H), 8.50 (s, 1H). |
| Clinical data | |
|---|---|
| Trade names | Tryvio |
| Other names | ACT-132577 |
| AHFS/Drugs.com | Tryvio |
| Routes of administration | By mouth |
| Drug class | Antihypertensive |
| ATC code | C02KN01 (WHO) |
| Legal status | |
| Legal status | US: ℞-only[1] |
| Identifiers | |
| showIUPAC name | |
| CAS Number | 1103522-45-7 |
| PubChem CID | 25099191 |
| IUPHAR/BPS | 10070 |
| DrugBank | DB15059 |
| ChemSpider | 25027753 |
| UNII | MZI81HV01P |
| KEGG | D11441 |
| ChEBI | CHEBI:76609 |
| ChEMBL | ChEMBL2165326 |
| Chemical and physical data | |
| Formula | C16H14Br2N6O4S |
| Molar mass | 546.19 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| hideSMILESC1=CC(=CC=C1C2=C(N=CN=C2OCCOC3=NC=C(C=N3)Br)NS(=O)(=O)N)Br | |
References
- ^ Jump up to:a b c d e f g h i j “Tryvio- aprocitentan tablet, film coated”. DailyMed. 29 March 2024. Archived from the original on 25 April 2024. Retrieved 25 April 2024.
- ^ Jump up to:a b c “US FDA approves Idorsia’s once-daily Tryvio (aprocitentan) – the first and only endothelin receptor antagonist for the treatment of high blood pressure not adequately controlled in combination with other antihypertensives” (Press release). Idorsia. 20 March 2024. Archived from the original on 28 April 2024. Retrieved 28 April 2024 – via PR Newswire.
- ^ Ojha, Utkarsh; Ruddaraju, Sanjay; Sabapathy, Navukkarasu; Ravindran, Varun; Worapongsatitaya, Pitchaya; Haq, Jeesanul; et al. (2022). “Current and Emerging Classes of Pharmacological Agents for the Management of Hypertension”. American Journal of Cardiovascular Drugs. 22 (3): 271–285. doi:10.1007/s40256-021-00510-9. PMC 8651502. PMID 34878631.
- ^ Jump up to:a b Xu, Jingjing; Jiang, Xiaohua; Xu, Suowen (November 2023). “Aprocitentan, a dual endothelin-1 (ET-1) antagonist for treating resistant hypertension: Mechanism of action and therapeutic potential”. Drug Discovery Today. 28 (11): 103788. doi:10.1016/j.drudis.2023.103788. PMID 37742911.
- ^ “Novel Drug Approvals for 2024”. U.S. Food and Drug Administration (FDA). 29 April 2024. Archived from the original on 30 April 2024. Retrieved 30 April 2024.
- ^ Deswal, Phalguni (6 September 2023). “Idorsia reacquires aprocitentan rights from Janssen for $343m”. Pharmaceutical Technology. Archived from the original on 8 November 2023. Retrieved 8 November 2023.
- ^ Jump up to:a b “Jeraygo EPAR”. European Medicines Agency. 25 April 2024. Archived from the original on 30 April 2024. Retrieved 27 April 2024. Text was copied from this source which is copyright European Medicines Agency. Reproduction is authorized provided the source is acknowledged.
Further reading
- Mahfooz K, Najeed S, Tun HN, Khamosh M, Grewal D, Hussain A, et al. (July 2023). “New Dual Endothelin Receptor Antagonist Aprocitentan in Hypertension: A Systematic Review and Meta-Analysis”. Current Problems in Cardiology. 48 (7): 101686. doi:10.1016/j.cpcardiol.2023.101686. PMID 36893968.
/////ACT-132577, Aprocitentan, Tryvio, FDA 2024, APPROVALS 2024, N-Despropyl-macitentan, WHO 10552
Cefepime

Cefepime
88040-23-7
| INGREDIENT | UNII | CAS | INCHI KEY |
|---|---|---|---|
| Cefepime hydrochloride | I8X1O0607P | 123171-59-5 | LRAJHPGSGBRUJN-OMIVUECESA-N |
Cefepime
CAS Registry Number: 88040-23-7
CAS Name: 1-[[(6R,7R)-7-[[(2Z)-(2-Amino-4-thiazolyl)(methoxyimino)acetyl]amino]-2-carboxy-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-en-3-yl]methyl]-1-methylpyrrolidinium inner salt
Additional Names: 1-[[(6R,7R)-7-[2-(2-amino-4-thiazolyl)glyoxylamido]-2-carboxy-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-en-3-yl]methyl]-1-methylpyrrolidinium hydroxide inner salt 72-(Z)-2-(O-methyloxime); 7-[(Z)-2-(2-aminothiazol-4-yl)-2-methoxyiminoacetamido]-3-(1-methylpyrrolidinio)methyl-3-cephem-4-carboxylate
Manufacturers’ Codes: BMY-28142
Molecular Formula: C19H24N6O5S2
Molecular Weight: 480.56
Percent Composition: C 47.49%, H 5.03%, N 17.49%, O 16.65%, S 13.34%
Literature References: Semisynthetic, fourth generation cephalosporin antibiotic. Prepn: S. Aburaki et al.,DE3307550; eidem,US4406899 (both 1983 to Bristol-Myers); and antibacterial activity: T. Naito et al.,J. Antibiot.39, 1092 (1986). In vitro comparative antimicrobial spectrum: N. J. Khan et al.,Antimicrob. Agents Chemother.26, 585 (1984); and b-lactamase stability: H. C. Neu et al.,J. Antimicrob. Chemother.17, 441 (1986). HPLC determn in plasma and urine: R. H. Barbhaiya et al.,Antimicrob. Agents Chemother.31, 55 (1987). Clinical evaluations in infection: N. Clynes et al.,Diagn. Microbiol. Infect. Dis.12, 257 (1989); S. Oster et al.,Antimicrob. Agents Chemother.34, 954 (1990). Review of clinical pharmacokinetics: M. P. Okamoto et al.,Clin. Pharmacokinet.25, 88-102 (1993).
Properties: Colorless powder, mp 150° (dec). uv max (pH 7 phosphate buffer): 235, 257 nm (e 16700, 16100).
Melting point: mp 150° (dec)
Absorption maximum: uv max (pH 7 phosphate buffer): 235, 257 nm (e 16700, 16100)
Derivative Type: Sulfate
Molecular Formula: C19H24N6O5S2.H2SO4
Molecular Weight: 578.64
Percent Composition: C 39.44%, H 4.53%, N 14.52%, O 24.89%, S 16.62%
Properties: mp 210° (dec). uv max (pH 7 phosphate buffer): 236, 258 nm (e 17200, 16900).
Melting point: mp 210° (dec)
Absorption maximum: uv max (pH 7 phosphate buffer): 236, 258 nm (e 17200, 16900)
Derivative Type: Hydrochloride monohydrate
CAS Registry Number: 123171-59-5
CAS Name: 1-[[(6R,7R)-7-[[(2Z)-(2-Amino-4-thiazolyl)(methoxyimino)acetyl]amino]-2-carboxy-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-en-3-yl]methyl]-1-methylpyrrolidinium chloride monohydrochloride monohydrate
Additional Names: cefepime hydrochloride
Trademarks: Axepim (BMS); Cepimex (Mead Johnson); Maxipime (BMS)
Molecular Formula: C19H25ClN6O5S2.HCl.H2O
Molecular Weight: 571.50
Percent Composition: C 39.93%, H 4.94%, Cl 12.41%, N 14.71%, O 16.80%, S 11.22%
Therap-Cat: Antibacterial.
Keywords: Antibacterial (Antibiotics); ?Lactams; Cephalosporins.
FDA APPROVED 2/22/2024, To treat complicated urinary tract infections, Exblifep
- BMY 28142
- BMY-28142
Cefepime is a fourth-generation cephalosporin antibiotic. Cefepime has an extended spectrum of activity against Gram-positive and Gram-negative bacteria, with greater activity against both types of organism than third-generation agents. A 2007 meta-analysis suggested when data of trials were combined, mortality was increased in people treated with cefepime compared with other β-lactam antibiotics.[1] In response, the U.S. Food and Drug Administration (FDA) performed their own meta-analysis which found no mortality difference.[2]
Cefepime was patented in 1982 by Bristol-Myers Squibb and approved for medical use in 1994.[3] It is available as a generic drug and sold under a variety of trade names worldwide.[citation needed][4]
It was removed from the World Health Organization’s List of Essential Medicines in 2019.[5]
Medical use
Cefepime is usually reserved to treat moderate to severe nosocomial pneumonia, infections caused by multiple drug-resistant microorganisms (e.g. Pseudomonas aeruginosa) and empirical treatment of febrile neutropenia.[6]
Cefepime has good activity against important pathogens including Pseudomonas aeruginosa, Staphylococcus aureus, and multiple drug-resistant Streptococcus pneumoniae. A particular strength is its activity against Enterobacteriaceae. Whereas other cephalosporins are degraded by many plasmid– and chromosome-mediated beta-lactamases, cefepime is stable and is a front-line agent when infection with Enterobacteriaceae is known or suspected.[medical citation needed]
Spectrum of bacterial susceptibility
Cefepime is a broad-spectrum cephalosporin antibiotic and has been used to treat bacteria responsible for causing pneumonia and infections of the skin and urinary tract. Some of these bacteria include Pseudomonas, Escherichia, and Streptococcus species. The following represents MIC susceptibility data for a few medically significant microorganisms:[7]
- Escherichia coli: ≤0.007 – 128 μg/ml
- Pseudomonas aeruginosa: 0.06 – >256 μg/ml
- Streptococcus pneumoniae: ≤0.007 – >8 μg/ml
Chemistry
The combination of the syn-configuration of the methoxy imino moiety and the aminothiazole moiety confers extra stability to β-lactamase enzymes produced by many bacteria. The N–methyl pyrrolidine moiety increases penetration into Gram-negative bacteria. These factors increase the activity of cefepime against otherwise resistant organisms including Pseudomonas aeruginosa and Staphylococcus aureus.

Semisynthetic, fourth generation cephalosporin antibiotic. Prepn: S. Aburaki et al., DE 3307550; eidem, US 4406899 (both 1983 to Bristol-Myers); and antibacterial activity: T. Naito et al., J. Antibiot. 39, 1092 (1986).
Trade names
Following expiration of the Bristol-Myers Squibb patent,[] cefepime became available as a generic and is now] marketed by numerous companies worldwide under tradenames including Neopime (Neomed), Maxipime, Cepimax, Cepimex, and Axepim.
| Clinical data | |
|---|---|
| Pronunciation | /ˈsɛfɪpiːm/ or /ˈkɛfɪpiːm/ |
| Trade names | Maxipime, Voco |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a698021 |
| Pregnancy category | AU: B1 |
| Routes of administration | Intravenous, intramuscular |
| ATC code | J01DE01 (WHO) |
| Legal status | |
| Legal status | AU: S4 (Prescription only)CA: ℞-onlyUK: POM (Prescription only)US: ℞-only |
| Pharmacokinetic data | |
| Bioavailability | 100% (IM) |
| Metabolism | Hepatic 15% |
| Elimination half-life | 2 hours |
| Excretion | Renal 70–99% |
| Identifiers | |
| showIUPAC name | |
| CAS Number | 88040-23-7 |
| PubChem CID | 5479537 |
| DrugBank | DB01413 |
| ChemSpider | 4586395 |
| UNII | 807PW4VQE3 |
| KEGG | D02376 |
| ChEBI | CHEBI:478164 |
| ChEMBL | ChEMBL186 |
| CompTox Dashboard (EPA) | DTXSID70873208 |
| ECHA InfoCard | 100.171.025 |
| Chemical and physical data | |
| Formula | C19H24N6O5S2 |
| Molar mass | 480.56 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| Melting point | 150 °C (302 °F) (dec.) |
| showSMILES | |
| showInChI | |
| (verify) | |
References
- ^ Yahav D, Paul M, Fraser A, Sarid N, Leibovici L (May 2007). “Efficacy and safety of cefepime: a systematic review and meta-analysis”. The Lancet. Infectious Diseases. 7 (5): 338–348. doi:10.1016/S1473-3099(07)70109-3. PMID 17448937.
- ^ “FDA Alert: Cefepime (marketed as Maxipime)”. Information for Healthcare Professionals. Food and Drug Administration. Archived from the original on 2 November 2017. Retrieved 2 August 2009.
- ^ Fischer J, Ganellin CR (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 496. ISBN 9783527607495. Archived from the original on 19 June 2021. Retrieved 19 September 2020.
- ^ “Cefepime (maxipime), large spectrum 4th generation cephalosporin, resistant to beta-lactamases]”.
- ^ World Health Organization (2019). Executive summary: the selection and use of essential medicines 2019: report of the 22nd WHO Expert Committee on the selection and use of essential medicines. Geneva: World Health Organization. hdl:10665/325773. WHO/MVP/EMP/IAU/2019.05. License: CC BY-NC-SA 3.0 IGO.
- ^ Chapman TM, Perry CM (2003). “Cefepime: a review of its use in the management of hospitalized patients with pneumonia”. American Journal of Respiratory Medicine. 2 (1): 75–107. doi:10.1007/bf03256641. PMID 14720024.
- ^ “Cefepime Susceptibility and Concentration Range (μg/ml) Minimum Inhibitory Concentration (MIC) Data” (PDF). The Antimicrobial Index. toku-e.com. Archived from the original (PDF) on 1 November 2018.
External links
- “Cefepime”. Drug Information Portal. U.S. National Library of Medicine.
//////////cefepime, Exblifep, FDA 2024, APPROVALS 2024, BMY 28142, BMY-28142
Berdazimer


Berdazimer
CAS NA SALT, 1846565-00-1
FDA APPROVE 1 /5/2024, To treat molluscum contagiosum
Drug Trials Snapshot
- NVN-1000 free acid
- NVN1000 free acid
- Silsesquioxanes, 3-(2-hydroxy-1-methyl-2-nitrosohydrazinyl)propyl 3-(methylamino)propyl, polymers with silicic acid (h4sio4) tetra-et ester, hydroxy-terminated
Berdazimer sodium, sold under the brand name Zelsuvmi, is a medication used for the treatment for molluscum contagiosum.[1] Berdazimer sodium is a nitric oxide releasing agent.[1] It is a polymer formed from sodium 1-hydroxy-3-methyl-3-(3-(trimethoxysilyl)propyl)-1-triazene-2-oxide and tetraethyl silicate.[2]
Berdazimer sodium was approved for medical use in the United States in January 2024.[3][4][5]
Medical uses
Berdazimer sodium is indicated for the topical treatment of molluscum contagiosum.[1]
Pharmacology
Mechanism of action
Berdazimer sodium is a nitric oxide releasing agent.[1] The mechanism of action for the treatment of molluscum contagiosum is unknown.[1]
Pharmacodynamics
The pharmacodynamics of berdazimer sodium are unknown.[1]
Society and culture
Legal status
Berdazimer sodium was approved for medical use in the United States in January 2024.[4]
Names
Berdazimer sodium is the international nonproprietary name.[6]
Berdazimer is a polymeric substance consisting of a polysiloxane backbone (Si-O-Si bonds) with covalently bound N-diazeniumdiolate nitric oxide (NO) donors. It releases NO through exposure to proton donors like water, which will degrade the N-diazeniumdiolate entity.2 Berdazimer was previously investigated as a potential treatment for molluscum contagiosum, a viral cutaneous infection mainly affecting children, sexually active adults, and immunocompromised patients. It is one of the 5 most prevalent skin diseases in the world and the third-most common viral skin infection in children.3 Previously, the first line treatment for molluscum contagiosum was surgical excision, although it poses challenges such as repeated doctor visits, post-surgical scarring and skin discoloration, and fear and anxiety in the pediatric population.3
On Jan 05, 2024, the FDA approved berdazimer under the brand name ZELSUVMI for the treatment of adult and pediatric molluscum contagiosum, and it is the first drug to be approved for this condition. This decision is based on positive results demonstrated in 2 Phase 3 trials, B-SIMPLE 4 and B-SIMPLE 2, where reduced lesion counts were observed with once-a-day uses of berdazimer.5



 References
References
- ^ Jump up to:a b c d e f g h i “Zelsuvmi (berdazimer) topical gel” (PDF). Archived (PDF) from the original on 19 January 2024. Retrieved 9 January 2024.
- ^ “GSRS”. gsrs.ncats.nih.gov. Archived from the original on 8 January 2024. Retrieved 8 January 2024.
- ^ “Drug Approval Package: Zelsuvmi”. U.S. Food and Drug Administration (FDA). 2 February 2024. Archived from the original on 11 March 2024. Retrieved 11 March 2024.
- ^ Jump up to:a b “Novel Drug Approvals for 2024”. U.S. Food and Drug Administration (FDA). 29 April 2024. Archived from the original on 30 April 2024. Retrieved 30 April 2024.
- ^ “U.S. Food and Drug Administration Approves Zelsuvmi as a First-in-Class Medication for the Treatment of Molluscum Contagiosum”. Ligand Pharmaceuticals. 5 January 2024. Archived from the original on 8 January 2024. Retrieved 8 January 2024 – via Business Wire.
- ^ World Health Organization (2018). “International nonproprietary names for pharmaceutical substances (INN): recommended INN: list 79”. WHO Drug Information. 32 (1). hdl:10665/330941.
Further reading
- Pera Calvi I, R Marques I, Cruz SA, Mesquita YL, Padrao EM, Souza RM, et al. (2023). “Safety and efficacy of topical nitric oxide-releasing berdazimer gel for molluscum contagiosum clearance: A systematic review and meta-analysis of randomized controlled trials”. Pediatric Dermatology. 40 (6): 1060–1063. doi:10.1111/pde.15419. PMID 37721050. S2CID 262045499.
- Han H, Smythe C, Yousefian F, Berman B (February 2023). “Molluscum Contagiosum Virus Evasion of Immune Surveillance: A Review”. Journal of Drugs in Dermatology. 22 (2): 182–189. doi:10.36849/JDD.7230. PMID 36745361. S2CID 256613906.
- Lacarrubba F, Micali G, Trecarichi AC, Quattrocchi E, Monfrecola G, Verzì AE (December 2022). “New Developing Treatments for Molluscum Contagiosum”. Dermatology and Therapy. 12 (12): 2669–2678. doi:10.1007/s13555-022-00826-7. PMC 9674806. PMID 36239905.
- Ward BM, Riccio DA, Cartwright M, Maeda-Chubachi T (November 2023). “The Antiviral Effect of Berdazimer Sodium on Molluscum Contagiosum Virus Using a Novel In Vitro Methodology”. Viruses. 15 (12): 2360. doi:10.3390/v15122360. PMC 10747301. PMID 38140601.
External links
- “Berdazimer Sodium (Code C174810)”. NCI Thesaurus.
- Clinical trial number NCT04535531 for “A Phase 3 Molluscum Contagiosum Efficacy and Safety Study (B-SIMPLE4)” at ClinicalTrials.gov
- Clinical trial number NCT03927703 for “A Phase 3 Efficacy & Safety of SB206 & Vehicle Gel for the Treatment of MC (B-SIMPLE2)” at ClinicalTrials.gov
- Clinical trial number NCT03927716 for “A Phase 3 Randomized Parallel Group Study Comparing the Efficacy & Safety of SB206 & Vehicle Gel in the Treatment of MC (B-SIMPLE1)” at ClinicalTrials.gov
| Clinical data | |
|---|---|
| Trade names | Zelsuvmi |
| Other names | SB206 |
| License data | US DailyMed: Berdazimer sodium |
| Routes of administration | Topical |
| ATC code | None |
| Legal status | |
| Legal status | US: ℞-only[1] |
| Identifiers | |
| CAS Number | 1846565-00-1 |
| DrugBank | DBSALT003491DB18712 |
| UNII | ORT9SID4QYB23P7SM943 |
| KEGG | D12758 |
| ChEMBL | ChEMBL4298064 |
| Chemical and physical data | |
| Formula | Indeterminate[1] |
| Molar mass | Indeterminate[1] |
/////Berdazimer, Zelsuvmi, FDA 2024, APPROVALS 2024, NVN-1000 free acid, NVN1000 free acid
Tovorafenib

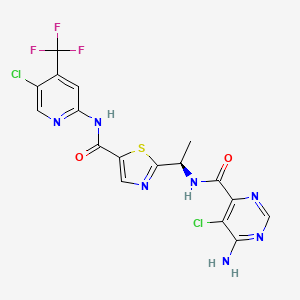
Tovorafenib
506.29
C17H12Cl2F3N7O2S
1096708-71-2
6-amino-5-chloro-N-[(1R)-1-(5-{[5-chloro-4-(trifluoromethyl)pyridin-2-yl]carbamoyl}-1,3-thiazol-2-yl)ethyl]pyrimidine-4-carboxamide
4/23/2024 FDA APROVED, To treat relapsed or refractory pediatric low-grade glioma, Ojemda
- AMG 2112819
- BIIB 024
- BIIB-024
- BIIB024
- DAY 101
- DAY-101
- DAY101
- MLN 2480
- MLN-2480
- MLN2480
- TAK 580
- TAK-580
- TAK580
Tovorafenib, sold under the brand name Ojemda, is a medication used for the treatment of glioma.[1] It is a kinase inhibitor.[1]
The most common adverse reactions include rash, hair color changes, fatigue, viral infection, vomiting, headache, hemorrhage, pyrexia, dry skin, constipation, nausea, dermatitis acneiform, and upper respiratory tract infection.[2] The most common grade 3 or 4 laboratory abnormalities include decreased phosphate, decreased hemoglobin, increased creatinine phosphokinase, increased alanine aminotransferase, decreased albumin, decreased lymphocytes, decreased leukocytes, increased aspartate aminotransferase, decreased potassium, and decreased sodium.[2]
It was approved for medical use in the United States in April 2024,[1][2][3][4] and is the first approval of a systemic therapy for the treatment of people with pediatric low-grade glioma with BRAF rearrangements, including fusions.[2]
Medical uses
Tovorafenib is indicated for the treatment of people six months of age and older with relapsed or refractory pediatric low-grade glioma harboring a BRAF fusion or rearrangement, or BRAF V600 mutation.[1][2]
History
Efficacy was evaluated in 76 participants enrolled in FIREFLY-1 (NCT04775485), a multicenter, open-label, single-arm trial in participants with relapsed or refractory pediatric low-grade glioma harboring an activating BRAF alteration detected by a local laboratory who had received at least one line of prior systemic therapy.[2] Participants were required to have documented evidence of radiographic progression and at least one measurable lesion.[2] Participants with tumors harboring additional activating molecular alterations (e.g., IDH1/2 mutations, FGFR mutations) or with a known or suspected diagnosis of neurofibromatosis type 1 were excluded.[2] Participants received tovorafenib based on body surface area (range: 290 to 476 mg/m2, up to a maximum dose of 600 mg) once weekly until they experienced disease progression or unacceptable toxicity.[2] The US Food and Drug Administration (FDA) granted the application for tovorafenib priority review, breakthrough therapy, and orphan drug designations.[2]
Society and culture
Names
Tovorafenib is the international nonproprietary name.[5]
SYN
PATENT
Huang et al., Angew. Chem. int. Ed. (2016), 55, 5309-5317
Jiang Xiao-bin et al., Org. Lett. (2003), 5, 1503
10Da
| (R)-2-(1-(6-amino-5-chloropyrimidine-4- carboxamido)ethyl)-N-(5-chloro-4- (trifluoromethyl)pyridin-2-yl)thiazole-5- carboxamide |
SYN

Patent
https://patentscope.wipo.int/search/en/detail.jsf?docId=US131345763&_cid=P22-LW02NH-45076-1
PATENT
https://patentscope.wipo.int/search/en/detail.jsf?docId=US201396258&_cid=P22-LW02NH-45076-1
PATENT
References
- ^ Jump up to:a b c d e “Archived copy” (PDF). Archived (PDF) from the original on 24 April 2024. Retrieved 24 April 2024.
- ^ Jump up to:a b c d e f g h i j “FDA grants accelerated approval to tovorafenib for patients with relapsed or refractory BRAF-altered pediatric low-grade glioma”. U.S. Food and Drug Administration (FDA). 23 April 2024. Archived from the original on 23 April 2024. Retrieved 25 April 2024.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain. - ^ “Novel Drug Approvals for 2024”. U.S. Food and Drug Administration (FDA). 29 April 2024. Archived from the original on 30 April 2024. Retrieved 30 April 2024.
- ^ “Day One’s Ojemda (tovorafenib) Receives US FDA Accelerated Approval for Relapsed or Refractory BRAF-altered Pediatric Low-Grade Glioma (pLGG), the Most Common Form of Childhood Brain Tumor”. Day One Biopharmaceuticals (Press release). 23 April 2024. Archived from the original on 23 April 2024. Retrieved 24 April 2024.
- ^ World Health Organization (2022). “International nonproprietary names for pharmaceutical substances (INN): recommended INN: list 88”. WHO Drug Information. 36 (3). hdl:10665/363551.
External links
- “Tovorafenib”. NCI Drug Dictionary.
- “Tovorafenib (Code C106254)”. NCI Thesaurus.
- Clinical trial number NCT04775485 for “A Study to Evaluate DAY101 in Pediatric and Young Adult Patients With Relapsed or Progressive Low-Grade Glioma and Advance Solid Tumors (FIREFLY-1)” at ClinicalTrials.gov
| Clinical data | |
|---|---|
| Trade names | Ojemda |
| Other names | BIIB-024, MLN2480, AMG 2112819, DAY101, TAK-580 |
| License data | US DailyMed: Tovorafenib |
| Routes of administration | By mouth |
| Drug class | Antineoplastic |
| ATC code | None |
| Legal status | |
| Legal status | US: ℞-only[1] |
| Identifiers | |
| showIUPAC name | |
| CAS Number | 1096708-71-2 |
| PubChem CID | 25161177 |
| DrugBank | DB15266 |
| ChemSpider | 28637796 |
| UNII | ZN90E4027M |
| KEGG | D12291 |
| ChEBI | CHEBI:167672 |
| ChEMBL | ChEMBL3348923 |
| PDB ligand | QOP (PDBe, RCSB PDB) |
| Chemical and physical data | |
| Formula | C17H12Cl2F3N7O2S |
| Molar mass | 506.29 g·mol−1 |
| showInChI | |
////////Tovorafenib, Ojemda, FDA 2024. APPROVALS 2024, AMG 2112819, BIIB 024, BIIB-024, BIIB024, DAY 101, DAY-101, DAY101, MLN 2480, MLN-2480, MLN2480, TAK 580, TAK-580, TAK580

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....










