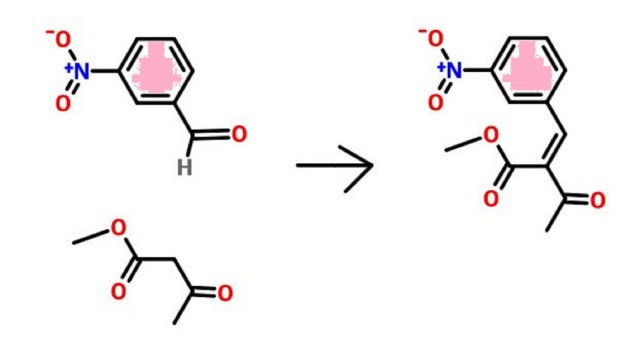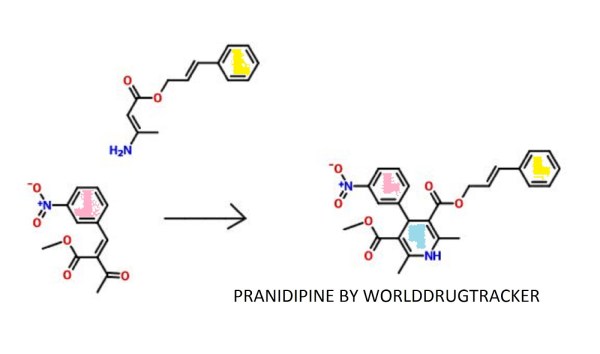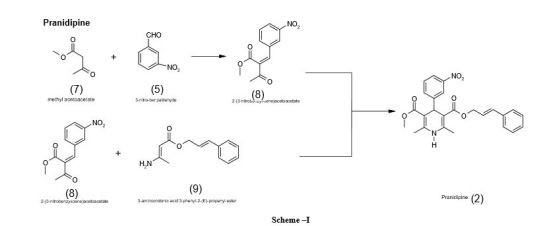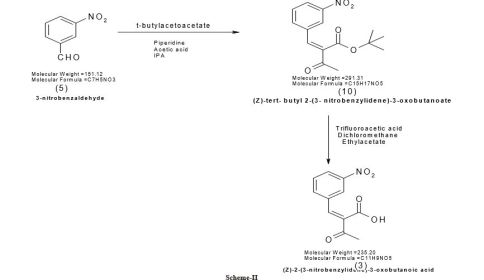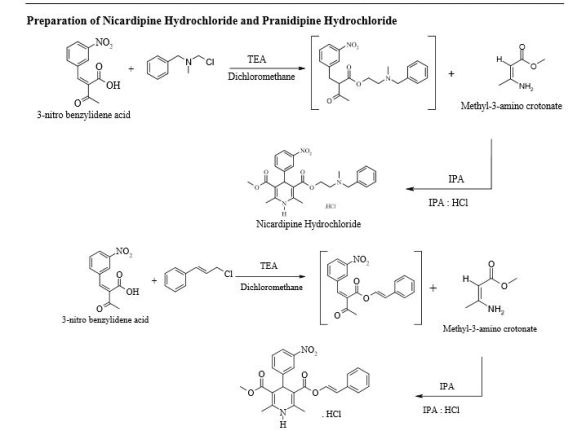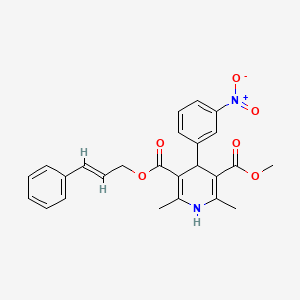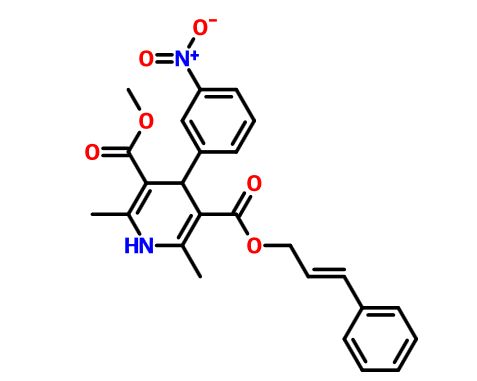

Pranidipine , OPC-13340, FRC 8411
Acalas®
NDA Filing in Japan
A calcium channel blocker potentially for the treatment of angina pectoris and hypertension.

CAS No. 99522-79-9
- Molecular FormulaC25H24N2O6
- Average mass448.468
Pranidipine is a calcium channel blocker. It is a long acting calcium channel antagonist of the dihydropyridine group.[1]

PATENT
EP 0173126
http://www.google.com/patents/EP0173126A1?cl=en
PAPER
Der Pharmacia Sinica, 2014, 5(1):11-17
pelagiaresearchlibrary.com/der-pharmacia-sinica/vol5-iss1/DPS-2014-5-1-11-17.pdf
CLICK ON IMAGE FOR CLEAR VIEW
| Patent | Submitted | Granted |
|---|---|---|
| Process for the preparation of 1,4 – dihydropyridines and novel 1,4-dihydropyridines useful as therapeutic agents [US2003230478] | 2003-12-18 | |
| Advanced Formulations and Therapies for Treating Hard-to-Heal Wounds [US2014357645] | 2014-08-19 | 2014-12-04 |
| METHODS OF TREATING CARDIOVASCULAR AND METABOLIC DISEASES [US2014322199] | 2012-08-06 | 2014-10-30 |
| Protein Carrier-Linked Prodrugs [US2014323402] | 2012-08-10 | 2014-10-30 |
| sGC STIMULATORS [US2014323448] | 2014-04-29 | 2014-10-30 |
| TREATMENT OF ARTERIAL WALL BY COMBINATION OF RAAS INHIBITOR AND HMG-CoA REDUCTASE INHIBITOR [US2014323536] | 2012-12-07 | 2014-10-30 |
| Agonists of Guanylate Cyclase Useful For the Treatment of Gastrointestinal Disorders, Inflammation, Cancer and Other Disorders [US2014329738] | 2014-03-28 | 2014-11-06 |
| METHODS, COMPOSITIONS, AND KITS FOR THE TREATMENT OF CANCER [US2014335050] | 2012-05-25 | 2014-11-13 |
| ROR GAMMA MODULATORS [US2014343023] | 2012-09-18 | 2014-11-20 |
| High-Loading Water-Soluable Carrier-Linked Prodrugs [US2014296257] | 2012-08-10 | 2014-10-02
|
| Publication Number | Publication Date | IPCR Assignee/Applicant | Structure hits | Tools | |
|---|---|---|---|---|---|
|
1.
US-20150342954-A1 |
2015-12-03 |
|
|||
|
2.
EP-2558474-B1 |
2015-11-25 |
EN
|
|
||
|
3.
US-20150307580-A1 |
2015-10-29 |
|
|||
|
4.
US-20150305974-A1 |
2015-10-29 |
|
|||
|
5.
WO-2015164658-A1 |
2015-10-29 |
EN
|
|
||
|
6.
EP-2527360-B1 |
2015-10-28 |
EN
|
|
||
|
7.
WO-2015157471-A1 |
2015-10-15 |
EN
|
|
||
|
8.
US-20150284411-A1 |
2015-10-08 |
|
|||
|
9.
US-20150283202-A1 |
2015-10-08 |
|
|||
|
10.
US-9150512-B2 |
2015-10-06 |
|
References
Jin Yang, Keisuke Fukuo, Shigeto Morimoto, Tadaaki Niinobu, Toshimitsu Suhara, Toshio Ogihara (2000). “Pranidipine Enhances the Action of Nitric Oxide Released From Endothelial Cells”. Hypertension 35: 82–85. doi:10.1161/01.hyp.35.1.82.
 |
|
| Names | |
|---|---|
| IUPAC name
methyl (2E)-phenylprop-2-en-1-yl 2,6-dimethyl-4-(3-nitrophenyl)-1,4-dihydropyridine-3,5-dicarboxylate
|
|
| Other names
2,6-dimethyl-4-(3-nitrophenyl)-1,4-dihydropyridine-3,5-dicarboxylic acid O5-methyl O3-[(E)-3-phenylprop-2-enyl] ester
|
|
| Identifiers | |
| 99522-79-9 |
|
| ChEMBL | ChEMBL1096842 |
| ChemSpider | 4940726 |
| Jmol interactive 3D | Image |
| MeSH | C048161 |
| PubChem | 6436048 |
| UNII | 9DES9QVH58 |
| Properties | |
| C25H24N2O6 | |
| Molar mass | 448.46786 |
//////////
CC1=C(C(C(=C(N1)C)C(=O)OCC=CC2=CC=CC=C2)C3=CC(=CC=C3)[N+](=O)[O-])C(=O)OC
see dipine series………..http://organicsynthesisinternational.blogspot.in/p/dipine-series.html

Nilvadipine – Wikipedia, the free encyclopedia

 manidipine
manidipine

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....