Acting 4 months ahead of schedule, the FDA has granted an accelerated approval to ceritinib (Zykadia; LDK378) as a treatment for patients with ALK-positive metastatic non-small cell lung cancer (NSCLC) following treatment with crizotinib (Xalkori), based on a single-arm clinical trial demonstrating a durable improvement in overall response rates (ORR).
The approval for the second-generation ALK inhibitor was supported by results from an analysis of 163 patients treated with single-agent ceritinib at 750 mg daily following progression on crizotinib. In these select patients, the ORR was 54.6% with a 7.4-month median duration of response, according to data submitted to the FDA by Novartis, the company developing the drug. Based on these findings, the FDA granted ceritinib a Breakthrough Therapy designation, Priority Review, and orphan product designation.
“Today’s approval illustrates how a greater understanding of the underlying molecular pathways of a disease can lead to the development of specific therapies aimed at these pathways,” Richard Pazdur, MD, director of the Office of Hematology and Oncology Products in the FDA’s Center for Drug Evaluation and Research, said in a statement. “It also demonstrates the FDA’s commitment to working cooperatively with companies to expedite a drug’s development, review and approval, reflecting the promise of the breakthrough therapy designation program.”
In the study that was the basis for the approval, the primary endpoint was ORR by RECIST criteria with a secondary outcome measure of duration of response. Treatment with ceritinib resulted in an ORR of 54.6% by investigator assessment with a median duration of response of 7.4 months. By blinded independent central review, the ORR was 43.6% and the duration of response was 7.1 months.
Earlier this year, results from a dose escalation study that examined ceritinib in 130 patients who were untreated or refractory to crizotinib were published in the New England Journal of Medicine. In this analysis for patients who received doses of at least 400 mg (n = 114), the ORR was 58%. Patients who had progressed on crizotinib (n = 80) experienced an ORR of 56% and those who were crizotinib-naïve (n =34) had an ORR of 62%.
The median progression-free survival was 7.0 months and the median duration of response was 8.2 months (95% CI; 6.9-11.4). Additionally, responses were seen in patients with untreated metastatic brain lesions who progressed on prior therapy with crizotinib, the authors of the study noted.
The most frequent adverse events were nausea (82%), diarrhea (75%), vomiting (65%), fatigue (47%) and increased alanine aminotransferase levels (35%). These adverse events were generally mild and resolved when treatment stopped or the dose was reduced.
The most common grade 3 or 4 drug-related adverse events were increased alanine aminotransferase levels (21%), increased aspartate aminotransferase levels (11%), diarrhea (7%) and increased lipase levels (7%), all of which were reversible upon treatment discontinuation.
“Zykadia represents an important treatment option for ALK-positive NSCLC patients who relapse after starting initial therapy with crizotinib,” Alice Shaw, MD, PhD, of the Massachusetts General Hospital (MGH) Cancer Center, lead author of the report, said in a statement. “This approval will affect the way we manage and monitor patients with this type of lung cancer, as we will now be able to offer them the opportunity for continued treatment response with a new ALK inhibitor.”
Two phase III studies are enrolling patients to further explore the efficacy and safety of ceritinib in patients with ALK-positive NSCLC. These studies will likely act as confirmation for the accelerated approval. In the first, ceritinib will be compared with chemotherapy in untreated patients with ALK-rearranged NSCLC (NCT01828099). The second will compare ceritinib to chemotherapy in ALK-positive patients with NSCLC following progression on chemotherapy and crizotinib (NCT01828112).
“The approval of Zykadia less than three and a half years after the first patient entered our clinical trial exemplifies what is possible with a highly focused approach to drug development and strong collaboration,” Alessandro Riva, MD, president of Novartis Oncology ad interim and global head of Oncology Development and Medical Affairs, said in a statement. “The dedication of clinical investigators, patients, the FDA and others has enabled us to bring this medicine to patients in need as swiftly as possible.”


Nitration of 2-chloro-4-fluoro-1-methylbenzene with KNO3 in the presence of H2SO4 gives 1-chloro-5-fluoro-2-methyl-4-nitrobenzene , which upon condensation with isopropyl alcohol in the presence of Cs2CO3 in 2-PrOH at 60 °C yields 5-isopropoxy-2-methyl-4-nitrobenzene .
Suzuki coupling of chloride with 4-pyridineboronic acid in the presence of Pd2dba3, K3PO4 and SPhos in dioxane/water at 150 °C (microwave irradiation) provides 4-(5-isopropoxy-2-methyl-4-nitrophenyl)pyridine , which is then subjected to global reduction using H2 over PtO2 in the presence of TFA in AcOH to afford 2-isopropoxy-5-methyl-4-piperidin-4-ylaniline .
N-Protection of piperidine with Boc2O in the presence of Et3N in CH2Cl2 furnishes the corresponding carbamate (VIII), which upon Buchwald-Hartwig cross coupling with 2,5-dichloropyrimidine derivative (prepared by condensation of 2-(isopropylsulfonyl)aniline and 2,4,5-trichloropyrimidine in the presence of NaH in DMSO/DMF) in the presence of Pd(OAc)2, Xantphos and Cs2CO3 in THF affords Boc-protected ceritinib . Finally, removal of Boc-group in compound using TFA in CH2Cl2 furnishes the target compound ceritinib
/////////////////

Anaplastic lymphoma kinase (ALK), a member of the insulin receptor superfamily of receptor tyrosine kinases, has been implicated in oncogenesis in hematopoietic and non- hematopoietic tumors. The aberrant expression of full-length ALK receptor proteins has been reported in neuroblastomas and glioblastomas; and ALK fusion proteins have occurred in anaplastic large cell lymphoma. The study of ALK fusion proteins has also raised the possibility of new therapeutic treatments for patients with ALK-positive malignancies. (Pulford et al., Cell. MoI. Life Sci. 61:2939-2953 (2004)).
Focal Adhesion Kinase (FAK) is a key enzyme in the integrin-mediated outside-in signal cascade (D. Schlaepfer et al., Prog Biophys MoI Biol 1999, 71, 43578). The trigger in the signal transduction cascade is the autophosphorylation of Y397. Phosphorylated Y397 is a SH2 docking site for Src family tyrosine kinases; the bound c-Src kinase phosphorylates other tyrosine residues in FAK. Among them, phsophorylated Y925 becomes a binding site for the SH2 site of Grb2 small adaptor protein. This direct binding of Grb2 to FAK is one of the key steps for the activation of down stream targets such as the Ras-ERK2/MAP kinase cascade.
Zeta-chain-associated protein kinase 70 (ZAP-70), a member of the protein tyrosine kinase family, is of potential prognostic importance in chronic lymphocytic leukemia (CLL). ZAP-70, known to be of importance in T and NK cell signaling but absent in normal peripheral B cells, is expressed in the majority of the poorer prognosis unmutated CLL and absent in most cases with mutated IgVH genes. ZAP-70 is also expressed in a minority of other B cell tumors. (Orchard et al., Leuk. Lymphoma 46:1689-98 (2005)). [0006] Insulin- like growth factor (IGF-I) signaling is highly implicated in cancer, with the IGF-I receptor (IGF-IR) as the predominating factor. IGR-IR is important for tumor transformation and survival of malignant cells, but is only partially involved in normal cell growth. Targeting of IGF-IR has been suggested to be a promising option for cancer therapy. (Larsson et al., Br. J. Cancer 92:2097-2101 (2005)).
Because of the emerging disease-related roles of ALK, FAK, ZAP-70 and IGF-IR, there is a continuing need for compounds which may be useful for treating and preventing a disease which responds to inhibition of ALK, FAK, ZAP-70 and/or IGF-IR
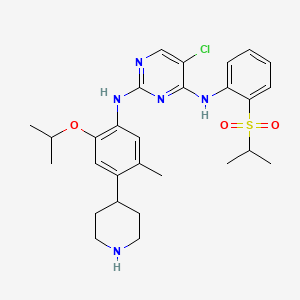
The compound 5-Chloro-N2-(2-isopropoxy-5-methyl-4-piperidin-4-yl-phenyl)-N4-[2- (propane-2-sulfonyl)-phenyl]-pyrimidine-2, 4-diamine, in the form of a free base, of formula
(I)
is an anaplastic lymphoma kinase (ALK) inhibitor, a member of the insulin receptor super family of receptor tyrosine kinases. Compound I was originally described in WO 2008/073687 Al as Example 7, compound 66. WO 2008/073687 Al , however, provides no information about crystalline forms of 5-
Chloro-N2-(2-isopropoxy-5-methyl-4-piperidin-4-yl-phenyl)-N4-[2-(propane-2-sulfonyl)- phenyl]-pyriniidine-2, 4-diamine or its corresponding salts. Crystalline forms of 5-Chloro-N2- (2-isopropoxy-5-methyl-4-piperidin-4-yl-phenyl)-N4-[2-(propane-2-sulfonyl)-phenyl]- pyrimidine-2, 4-diamine have been discovered, which are useful in treating diseases which respond to inhibition of anaplastic lymphoma kinase activity, focal adhesion kinase (FAK), zeta- chain-associated protein kinase 70 (ZAP-70) insulin-like growth factor (IGF-1 or a
combination thereof. The crystalline forms exhibit new physical properties that may be exploited in order to obtain new pharmacological properties, and that may be utilized in the drug product development of 5-Chloro-N2-(2-isopropoxy-5-methyl-4-piperidin-4-yl-phenyl)-N4-[2- (propane-2-sulfonyl)-phenyl]-pyrimidine-2, 4-diamine.
…………………….
WO 2008073687
http://www.google.com/patents/WO2008073687A2?cl=en
Example 7
5-Chloro-N2-(2-isopropoxy-5-methyl-4-piperidin-4-yl-phenyl)-N4-r2-(propane-2-sulfonyl)- phenvH-pyrimidine-2,4-diamine (66)
1 : 4-(5-Isopropoxv-2-methvl-4-nitro-phenyl)-pvridine
[0111] 4-Pyridineboronic acid (147 mg, 1.20 mmol, 1.1 equiv.) is dissolved in a 2:1 v/v mixture of dioxane and H2O (15 mL) and N2 is bubbled through for 5 minutes. Tris(dibenzylidene acetone)dipalladium (0) (100 mg, 0.109 mmol, 0.1 equiv.), 2- dicyclohexylphosphine-2′-6′-dimethoxy biphenyl (112 mg, 0.272 mmol, 0.25 equiv.), 1-chloro- 5-isopropoxy-2-methyl-4-nitro-benzene (Intermediate 4, 250 mg, 1.09 mmol, 1.0 equiv.) and K3PO4 (462 mg, 2.18 mmol, 2.0 equiv.) are added under a N2 blanket. The reaction vessel is sealed and heated with microwave irradiation to 150 0C for 20 min. After cooling to room temperature, the reaction is diluted with ethyl acetate and washed with 1 N aqueous NaOH (2x), the organic layer is then dried over Na2SO4 and filtered. After concentration, the crude product is purified by silica gel chromatography (gradient from hexanes to 30% ethyl acetate in hexanes) to give 4-(5-Isopropoxy-2-methyl-4-nitro-phenyl)-pyridine as a brown solid: ESMS m/z 273.1 (M + H+).
Steps 2 and 3 : 4-(4-Amino-5-isopropoxy-2-methyl-phenyl)-piperidine-l-carboxylic acid tert- butyl ester
[0112] 4-(5-Isopropoxy-2-methyl-4-nitro-phenyl)-pyridine from the previous step(438 mg, 1.61 mmol) dissolved in acetic acid (30 mL) is treated with TFA (0.24 mL, 3.22 mmol) and PtO2 (176 mg, 40% w/w). The reaction mixture is vigorously stirred under 1 atm. H2 for 36 hours. The reaction mixture is filtered and the filtrate is concentrated under vacuum. The resulting residue is diluted with ethyl acetate and washed with 1 N aqueous NaOH (2x), the organic layer is then dried over Na2SO4 and filtered. After concentration, the crude product (391 mg) is dissolved in anhydrous CH2Cl2 (30 mL). TEA is added (0.44 mL, 3.15, 2 equiv.) followed by Boc2O (344 mg, 1.57 equiv, 1 equiv.). The reaction is stirred at room temperature for 30 min. The reaction is concentrated under vacuum. The resulting residue is purified by silica gel chromatography (gradient from hexanes to 30% ethyl acetate in hexanes) to give 4-(4-amino-5- isopropoxy-2-methyl-phenyl)-piperidine-l-carboxylic acid tert-butyl ester as a sticky foam: ESMS m/z 293.1 (M-?Bu+H)+.
Steps 4 and 5
[0113] 4-(4-Amino-5-isopropoxy-2-methyl-phenyl)-piperidine-l-carboxylic acid tert-butyl ester (170 mg, 0.488 mmol) from the previous step, (2,5-Dichloro-pyrimidin-4-yl)-[2-(propane- 2-sulfonyl)-phenyl]-amine (Intermediate 2, 169 mg, 0.488 mmol, 1 equiv.), xantphos (28 mg, 0.049 mmol, 0.1 equiv.), palladium acetate (5.5 mg, 0.024 mmol, 0.05 equiv.), and Cs2CO3 (477 mg, 1.46 mmol, 3 equiv.) are dissolved in anhydrous THF (6 mL). N2 is bubbled through the reaction mixture for 5 minutes and then the reaction vessel is sealed and heated with microwave irradiation to 150 0C for 20 min. The reaction is filtered and the filtrate concentrated under vacuum. After concentration, the crude product is purified by silica gel chromatography (gradient from hexanes to 30% ethyl acetate in hexanes) to give 4-(4-{5-chloro-4-[2-(propane-2- sulfonyl)-phenylamino]-pyrimidin-2-ylamino}-5-isopropoxy-2-methyl-phenyl)-piperidine-l- carboxylic acid tert-butyl ester as a yellow film: ESMS m/z 658.3 (M + H+). This product (105 mg, 0.160 mmol) is dissolved in CH2Cl2 (3 mL) and treated with TFA (3 mL). After 45 min., the reaction is concentrated under vacuum. 1 N HCl in Et2O (5 mL x 2) is added causing the product HCl salt to precipitate. The solvent is removed by decantation. The resulting 5- Chloro-N2-(2-isopropoxy-5-methyl-4-piperidin-4-yl-phenyl)-N4-[2-(propane-2-sulfonyl)- phenyl]-pyrimidine-2,4-diamine (66) is dried under high vacuum, generating an off-white powder: 1H NMR (400 MHz, DMSO-J6+ trace D2O) δ 8.32 (s, IH), 8.27 (d, IH), 7.88 (d, IH), 7.67 (dd, IH), 7.45 (dd, IH), 7.42 (s, IH), 6.79 (s, IH), 4.56-4.48 (m, IH), 3.49-3.32 (m, 3H), 3.10-2.91 (m, 3H), 2.09 (s, 3H), 1.89-1.77 (m, 4H), 1.22 (d, 6H), 1.13 (d, 6H); ESMS m/z 558.1 (M + H+).
 66
66
……………………..
WO 2012082972
http://www.google.com/patents/WO2012082972A1
EXAMPLE 1
Preparation of Form A of 5-chloro-N-(2-isopropoxy-5-methyl-4-(piperidin-4-ylphenyl)-N-2- (isopropylsulfonyl phenyl)-2^-diamine
5-chloro-N-(2-isopropoxy-5-methyl-4-(piperidin-4-ylphenyl)-N-2-(isopropylsulfonyl)phenyl)- 2,4-diamine di-hydrochloride salt
The compound 2-isopropoxy-5-methyl-4-(piperdin-4-yl) aniline dihydrochloride (33.00 g, 85.25 mmol) and 2,5-dichloro-N-(2-(isopropyl sulfonyl )phenyl)pyrimidin-4-amine (32.53 g) was added to a 3 -necked 500-mL round-bottomed flask equipped with mechanical stirring, thermocouple, reflux condenser and N2 inlet-outlet. A solvent, 2-propanol (255.0 g, 325 mL), was added and the mixture to heated to reflux at 82±2 °C and stirred for at least 14 hours. The mixture was cooled to 22±3 °C over 1 hour and stirred at 22±3 °C for 3 hours. The resulting solids were filtered and rinsed with 3 x 40 g (3 x 51 mL) of 2-propanol. The solids were dried at 50±5 °C/10 mbar for 16 hours to yield 44.63 g of 5-chloro-N-(2-isopropoxy-5-methyl-4- (piperidin-4-ylphenyl)-N-2-(isopropylsulfonyl)phenyl)-2,4-diamine di-hydrochloride salt. Chemical Purity (as determined by HPLC): 97.3%. Corrected yield: 71.6%. LOD = 11.60%. The dihydrochloride salt was recrystallized using acetone:water (10:l,v/v). Chemical Purity (as determined by HPLC): 98.8%.
…………….
J Med Chem 2013, 56(14): 5675
http://pubs.acs.org/doi/abs/10.1021/jm400402q

Synthesis of 5-Chloro-N2-(2-isopropoxy-5-methyl-4-piperidin-4-ylphenyl)-N4-[2-(propane-2-sulfonyl)phenyl]pyrimidine-2,4-diamine 15b
|
10-21-2011
|
COMPOUNDS AND COMPOSITIONS AS PROTEIN KINASE INHIBITORS
|
|
|
10-21-2011
|
COMPOUNDS AND COMPOSITIONS AS PROTEIN KINASE INHIBITORS
|
|
|
10-19-2011
|
COMPOUNDS AND COMPOSITIONS AS PROTEIN KINASE INHIBITORS
|
|
|
8-5-2011
|
COMPOUNDS AND COMPOSITIONS AS PROTEIN KINASE INHIBITORS
|

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....