Home » Posts tagged 'novartis' (Page 2)
Tag Archives: novartis
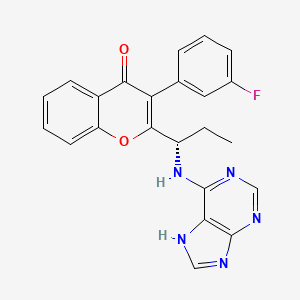
FGF 401


FGF 401
NVP-FGF-401
CAS 1708971-55-4
N-[5-Cyano-4-[(2-methoxyethyl)amino]-2-pyridinyl]-7-formyl-3,4-dihydro-6-[(4-methyl-2-oxo-1-piperazinyl)methyl]-1,8-naphthyridine-1(2H)-carboxamide
/V-(5-cyano-4-((2-methoxyethyl)amino)pyridin-2-yl)-7-formyl-6-((4-methyl-2-oxopiperazin-1 -yl)methyl)-3,4-dihydro-1 ,8-naphthyridine-1 (2H)-carboxamide
Phase I/II Hepatocellular carcinoma; Solid tumours
- Originator Novartis
- Developer Novartis Oncology
- Class Antineoplastics
- Mechanism of Action Type 4 fibroblast growth factor receptor antagonists
- 26 Jan 2016 Phase-I/II clinical trials in Solid tumours and Hepatocellular carcinoma in USA, Hong Kong, Japan, Taiwan, France, Germany and Spain (PO)
- 26 Dec 2014 Phase-I/II clinical trials in Hepatocellular carcinoma in Singapore (PO)
- 26 Dec 2014 Phase-I/II clinical trials in Solid tumours in Singapore (PO)
Activation of FGFRs (fibroblast growth factor receptors) has an essential role in regulating cell survival, proliferation, migration and differentiation.1 Dysregulation of the FGFR signaling pathway has been associated with human cancer.1 FGFRs represent an important target for cancer therapeutics because a growing body of evidence indicates that they can act in an oncogenic fashion to promote multiple steps of cancer progression, including induction of mitogenic and survival signals
FGF-401 is a FGFR4 inhibitor in phase I/II clinical studies at Novartis for the treatment of positive FGFR4 and KLB expresion solid tumors and hepatocellular carcinoma
Normal growth, as well as tissue repair and remodeling, require specific and delicate control of activating growth factors and their receptors. Fibroblast Growth Factors (FGFs) constitute a family of over twenty structurally related polypeptides that are developmental^ regulated and expressed in a wide variety of tissues. FGFs stimulate proliferation, cell migration and differentiation and play a major role in skeletal and limb development, wound healing, tissue repair, hematopoiesis, angiogenesis, and tumorigenesis (reviewed in Ornitz, Novartis Found Symp 232: 63-76; discussion 76-80, 272-82 (2001)).
The biological action of FGFs is mediated by specific cell surface receptors belonging to the Receptor Protein Tyrosine Kinase (RPTK) family of protein kinases. These proteins consist of an extracellular ligand binding domain, a single transmembrane domain and an intracellular tyrosine kinase domain which undergoes phosphorylation upon binding of FGF. Four FGFRs have been identified to date: FGFR1 (also called Fig, fms-like gene, fit- 2, bFGFR, N-bFGFR or Cek1 ), FGFR2 (also called Bek-Bacterial Expressed Kinase-, KGFR, Ksam, Ksaml and Cek3), FGFR3 (also called Cek2) and FGFR4. All mature FGFRs share a common structure consisting of an amino terminal signal peptide, three extracellular immunoglobulin-like domains (Ig domain I, Ig domain II, Ig domain III), with an acidic region between Ig domains (the “acidic box” domain), a transmembrane domain, and intracellular kinase domains (Ullrich and Schlessinger, Cell 61 : 203,1990 ; Johnson and Williams (1992) Adv. Cancer Res. 60: 1 -41). The distinct FGFR isoforms have different binding affinities for the different FGF ligands.
Alterations in FGFRs have been associated with a number of human cancers including myeloma, breast, stomach, colon, bladder, pancreatic and hepatocellular carcinomas. Recently, it was reported that FGFR4 may play an important role in liver cancer in particular (PLoS One, 2012, volume 7, 36713). Other studies have also implicated FGFR4 or its ligand FGF19 in other cancer types including breast, glioblastoma, prostate, rhabdomyosarcoma, gastric, ovarian, lung, colon (Int. J. Cancer 1993; 54:378-382; Oncogene 2010; 29:1543-1552; Cancer Res 2010; 70:802-812; Cancer Res 201 1 ; 71 :4550-4561 ; Clin Cancer Res 2004; 10:6169-6178; Cancer Res 2013;
73:2551 -2562; Clin Cancer Res 2012; 18:3780-3790; J. Clin. Invest. 2009; 1 19:3395-3407; Ann Surg Oncol 2010; 17:3354-61 ; Cancer 201 1 ; 1 17:5304-13; Clin Cancer Res 2013; 19:809-820; PNAS 2013; 1 10:12426-12431 ; Oncogene 2008; 27:85-97).
Therapies involving FGFR4 blocking antibodies have been described for instance in
WO2009/009173, WO2007/136893, WO2012/138975, WO2010/026291 , WO2008/052798 and WO2010/004204. WO2014/144737 and WO2014/01 1900 also describe low molecular weight FGFR4 inhibitors.
in spite of numerous treatment options for patients with cancer, there remains a need for effective and safe therapeutic agents and a need for new combination therapies that can be administered for the effective long-term treatment of cancer.
Liver cancer or hepatic cancer is classified as primary liver cancer (i.e. cancer that forms in the tissues of the liver) and secondary liver cancer (i.e. cancer that spreads to the liver from another part of the body). According to the National Cancer Institute at the National Institutes of Health, the number of estimated new cases and deaths from liver and intrahepatic bile duct cancer in the United States in 2014 was 33,190 and 23,000, respectively. Importantly, the percent surviving five years or more after being diagnosed with liver and intrahepatic bile duct cancer is only about 16%.
It has now been found that a combination of /V-(5-cyano-4-((2-methoxyethyl)amino)pyridin-2-yl)-7-formyl-6-((4-methyl-2-oxopiperazin-1 -yl)methyl)-3,4-dihydro-1 ,8-naphthyridine-1 (2H)-carboxamide in free form or in pharmaceutically acceptable salt form and at least one further active ingredient, as defined herein, shows synergistic combination activity in an in vitro cell proliferation assay as shown in the experimental section and may therefore be effective for the delay of progression or treatment of a proliferative disease, such as cancer, in particular liver cancer.
| Inventors | Nicole Buschmann, Robin Alec Fairhurst, Pascal Furet, Thomas Knöpfel, Catherine Leblanc, Robert Mah, Pierre NIMSGERN, Sebastien RIPOCHE, Lv LIAO, Jing XIONG, Xianglin ZHAO, Bo Han, Can Wang, |
| Applicant | Novartis Ag |

Nicole Buschmann

Drawn by worlddrugtracker, helping millions………………..
PATENT
WO 2015059668
https://www.google.com/patents/WO2015059668A1?cl=en
PATENT
A/-(5-cyano-4-((2-methoxyethyl)amino)pyridin-2-yl)-7-formyl-6-((4-methyl-2-oxopiperazin-1-yl)methyl)-3,4-dihydro-1 ,8-naphthyridine-1 (2H)-carboxamide in citric acid salt form has the following structure:

Example 1 – A/-(5-cvano-4 (2-methoxyethyl)amino)pyridin-2-yl)-7-formyl-6-((4-methyl-2-oxopiperazin-1-yl)methyl)-3,4-dihvdro-1 ,8-naphthyridine-1 (2H)-carboxamide in citric acid salt form (1 :1).

Step 1 : 2-(dimethoxymethyl)-1 ,8-naphthyridine.
The procedure described in J. Org. Chem., 2004, 69 (6), pp 1959-1966 was used. Into a 20 L 4-necked round-bottom flask was placed 2-aminopyridine-3-carbaldehyde (1000 g, 8.19 mol), 1 , 1-dimethoxypropan-2-one (1257 g, 10.64 mol), ethanol (10 L), and water (2 L). This was followed by the addition of a solution of sodium hydroxide (409.8 g, 10.24 mol) in water (1000 mL) drop wise with stirring at 0-15 °C. The solution was stirred for 3 h at 0-20 °C and then concentrated under vacuum. The resulting solution was extracted with 3×1200 mL of ethyl acetate and the organic layers were combined. The mixture was dried over sodium sulfate and concentrated under vacuum. The residue was washed with 3×300 mL of hexane and the solid was collected by filtration. This resulted in the title compound as a yellow solid. 1 H-NMR (400 MHz, DMSO-cf6) δ 9.1 1 (dd, 1 H), 8.53 (d, 1 H), 8.50 (dd, 1 H), 7.73 (d, 1 H), 7.67 (dd, 1 H), 5.44 (s, 1 H), 3.41 (s, 6H).
Step 2: 7-(dimethoxymethyl)-1 ,2,3,4-tetrahydro-1 ,8-naphthyridine.
The procedure described in J. Org. Chem. , 2004, 69 (6), pp 1959-1966 was used. Into a 5-L pressure tank reactor (5 atm) was placed 2-(dimethoxymethyl)-1 ,8-naphthyridine (200 g, 979 mmol), ethanol (3 L), Pt02 (12 g). The reactor was evacuated and flushed three times with nitrogen, followed by flushing with hydrogen. The mixture was stirred overnight at 23 °C under an
atmosphere of hydrogen. This reaction was repeated four times. The solids were filtered out and the resulting mixture was concentrated under vacuum to give the title compound as a yellow solid. 1 H-NMR (400 MHz, DMSO-d6) δ 7.14 (d, 1 H), 6.51 (d, 1 H), 6.47 – 6.41 (m, 1 H), 4.98 (s, 1 H), 3.28 -3.19 (m, 2H), 3.23 (s, 6H), 2.64 (t, 2H), 1 .73 – 1.79 (m, 2H).
Step 3: 6-bromo-7-(dimethoxymethyl)-1 ,2,3,4-tetrahydro-1 ,8-naphthyridine.
Into a 3 L 4-necked round-bottom flask was placed 7-(dimethoxymethyl)-1 ,2,3, 4-tetrahydro-1 ,8-naphthyridine (1 14.6 g, 550.3mmol) in acetonitrile (2 L). This was followed by the addition of NBS (103 g, 578 mol) in portions with stirring at 25 °C. The resulting solution was stirred for 30 min at 25 °C. The resulting mixture was concentrated under vacuum and the residue was diluted with 1000 mL of diethylether. The mixture was washed with 3×100 mL of ice/water. The aqueous phase was extracted with 2×100 mL of diethylether and the organic layers were combined. The resulting mixture was washed with 1×100 mL of brine, dried over sodium sulfate and concentrated under vacuum to give the title compound as a light yellow solid. LC-MS: (ES, m/z): 286.03 [M+H]+. 1 H-NMR: (300MHz, CDCI3) δ 1 .86 – 1 .94 (2H, m), 2.70 – 2.74 (2H, m), 3.9 – 3.43 (2H, m), 3.47 (6H, s), 5.23 (1 H, s), 5.58 (1 H, s), 7.29 (1 H, s).
Step 4: 2-(dimethoxymethyl)-5,6,7,8-tetrahydro-1 ,8-naphthyridine-3-carbaldehyde.
To a solution of 6-bromo-7-(dimethoxymethyl)-1 ,2,3, 4-tetrahydro-1 ,8-naphthyridine (15.0 g, 52.2 mmol) in THF (400 mL) at -78 °C under argon, was added MeLi (1 .6 M in Et20, 32.6 mL, 52.2 mmol), the solution was stirred for 5 min, then n-BuLi (1 .6 M in hexane, 35.9 mL, 57.5 mmol) was added slowly and the solution was stirred for 20 min. THF (100 mL) was added to the reaction at -78 °C. Subsequently, n-BuLi (1 .6 M in hexane, 49.0 mL, 78 mmol) was added and the reaction mixture was stirred for 20 min, then again n-BuLi (1 .6 M in hexane, 6.53 mL, 10.45 mmol) was added and the mixture was stirred for 10 min at – 78 °C. DMF (2.10 mL, 27.2 mmol) was added and the reaction mixture was stirred at -78 °C for 45 min, then it was allowed to warm to room temperature, poured into sat. aq. NH4CI and extracted twice with DCM. The combined organic phases were dried over Na2S04, filtered and evaporated to give the title compound as an orange oil. (UPLC-MS 3) tR 0.63 min; ESI-MS 237.2 [M+H]+.
Step 5: ethyl 2-((2-((tert-butoxycarbonyl)amino)ethyl)(methyl)amino)acetate.
Ethyl bromoacetate (1.27 mL, 1 1 .48 mmol) was added to a mixture of tert-butyl (2-(methylamino)ethyl)carbamate (2.0 g, 1 1 .48 mmol), triethylamine (4.81 mL) and THF (24 mL) at 0 °C. After stirring 24 h at room temperature the reaction mixture was partitioned between saturated aqueous NaHC03 and DCM, extracted 2x with DCM, the organic layers dried over Na2S04 and
evaporated to give the title compound as a clear pale-yellow oil. 1H NMR (400 MHz, CDCI3) δ 5.20 (s, br, 1 H), 4.18 (q, 2H), 3.24 (s, 2H), 3.22 – 3.16 (m, 2H), 2.65 – 2.61 (m, 2H), 2.38 (s, 3H), 1 .42 (s, 9H), 1 .24 (t, 3H).
Step 6: ethyl 2-((2-aminoethyl)(methyl)amino)acetate dihydrochloride.
Concentrated hydrochloric acid (10 mL) was added to a solution of ethyl 2-((2-((tert-butoxycarbonyl)amino)ethyl)(methyl)amino)acetate (3.05 g, 1 1 .13 mmol) in THF (20 mL) and EtOH (100 mL) at room temperature. After stirring 1 h at room temperature the reaction mixture was evaporated, ethanol (20 mL) added, evaporated, further ethanol (50 mL) added and then stirred at 60 °C for 70 min. The cooled reaction mixture was then evaporated to give the title compound as a pale-yellow glass. 1 H NMR (400 MHz, DMSO-d6) δ 8.58 (s, br, 3H), 4.19 (q, 2H), 4.26 – 4.15 (m, 2H), 3.44 (s, br, 2H), 3.21 (s, br, 2H), 2.88 (s, 3H), 1 .21 (t, 3H).
Step 7: 1 -((2-(dimethoxymethyl)-5,6,7,8-tetrahydro-1 ,8-naphthyridin-3-yl)methyl)-4-methylpiperazin-2-one.
Sodium triacetoxyborohydride (3.10 g, 14.61 mmol) was added to a mixture of 2-(dimethoxymethyl)-5,6,7,8-tetrahydro-1 ,8-naphthyridine-3-carbaldehyde (obtained in step 4, 2.30 g, 9.74 mmol), ethyl 2-((2-aminoethyl)(methyl)amino)acetate dihydrochloride (obtained in step 6, 2.6 g, 14.61 mmol) and triethylamine (6.75 mL, 48.7 mmol) in 1 ,2-dichloroethane (20 mL) at room temperature. The reaction mixture was stirred for 21 h at room temperature and additional sodium triacetoxyborohydride (2.6 g, 9.74 mmol) was added. After a further 4 h stirring at room temperature, again additional sodium triacetoxyborohydride (1 .3 g, 4.87 mmol) was added and the reaction maintained at 4 °C for 2.5 days. The reaction mixture was then warmed to room temperature, saturated aqueous NaHC03 solution added, the mixture extracted with DCM (3x), the combined organic layers dried over Na2S04 and evaporated. The residue was applied to a 120 g RediSep® silica column as a DCM solution and purified by normal phase chromatography, eluting with a gradient from DCM to 10% MeOH in DCM. Product containing fractions were combined and evaporated to give the title compound as an orange foam. 1 H NMR (400 MHz, CDCI3) δ 7.08 (s, 1 H), 5.30 (s, br, 1 H), 5.20 (s, 1 H), 4.69 (s, 2H), 3.44 – 3.34 (m, 2H), 3.40 (s, 6H), 3.22 – 3.15 (m, 2H), 3.24 (s, 2H), 2.71 – 2.64 (m, 2H), 2.58 – 2.50 (m, 2H), 2.31 (s, 3H), 1 .98 – 1.82 (m, 2H). (UPLC-MS 6) tR 0.33; ESI-MS 335.3 [M+H]+.
Step 8: 4-fluoro-5-iodopyridin-2-amine.
A suspension of 4-fluoropyridin-2-amine (336 g, 2.5 mol) and NIS (745 g, 2.75 mol) in MeCN (9 L) was treated with TFA (1 14 g, 1 mol). The reaction mixture was then stirred at room temperature for 8 h. The reaction mixture was diluted with EtOAc (10 L), washed with sat. aq. Na2S203 (2 x 5 L), brine (4 x 5 L). The combined organic layers were dried over Na2S04, filtered and concentrated to get the crude product. The crude product was purified by recrystallization from EtOAc/pentane (1/10) to afford the title compound as a white solid. 1H NMR (400 MHz, DMSO-cf6) δ 8.14 (d, 1 H), 6.45 (s, 2H), 6.33 (d, 1 H).
Step 9: 6-amino-4-fluoronicotinonitrile.
4-fluoro-5-iodopyridin-2-amine (obtained in step 8, 240 g, 1 mol), zinc cyanide (125 g, 1.05 mol), zinc (13 g, 0.2 mol), Pd2(dba)3 (25 g, 25 mmol) and dppf (55 g, 0.1 mol) in DMA (800 mL) were degassed and charged into the round bottom flask under nitrogen. The mixture was stirred at 100 °C for 3 h. The reaction mixture was diluted with 5% NaHC03 (2 L), extracted with EtOAc (4 x 600 mL). The combined organic layers were washed with 5% NaOH (1 L), dried over Na2S04, concentrated to 700 mL. The resulting organic phase was eluted through silica gel column with EtOAc (1.7 L). The combined organic filtrate was washed with 2 M HCI (3 x 800 mL). The pH of the aqueous phase was adjusted to 10 with saturated NaHC03. The aqueous phase was extracted whit DCM (3 x 500 mL). The combined DCM was dried over Na2S04 and concentrated. The residue was further purified by column chromatography (eluted with pentane: EtOAc 10: 1 to 3:2) followed by recrystallization from pentane/EtOAc 3/1 to give the title compound as white solid. 1 H NMR (400 MHz, DMSO-d6) δ 8.40 (d, 1 H), 7.40 (s, 2H), 6.34 (d, 1 H).
Step 10: tert-butyl (4-chloro-5-cyanopyridin-2-yl)carbamate.
A mixture of 2,4-dichloro-5-cyanopyridine (1 Og, 57.8 mmol), fe/f-butyl carbamate (8.2 g, 70.5 mmol), Pd(OAc)2 (0.26 g, 1 .1 mmol), Xantphos (1 .34 g, 2.3mmol) and K2C03 (12 g, 87 mmol) in THF (150 mL) was degassed 3x with nitrogen. The mixture was then heated at 70 °C for 4-5 h and monitored by chromatography until complete conversion. Following completion of the reaction, additional THF (100 mL) was added and heated the mixture at 70 °C for additional 1 h and then cooled to room temperature. The suspension was then filtered through a pad of celite to remove the solid. The filtrate was then concentrated and azotropically distilled with ethyl acetete before filtering to give the title compound. 1 H NMR (DMSO-d6, 400 MHz): δ 10.82 (s, 1 H), 8.79 (s, 1 H), 8.09 (s, 1 H), 1 .49 (s, 9H).
Step 1 1 : fe/f-butyl N-(5-cyano-4-((2-methoxyethyl)amino)pyridin-2-yl)carbamate.
A mixture of tert-butyl (4-chloro-5-cyanopyridin-2-yl)carbamate (obtained in step 10, 9.8 g, 38.6 mmol), 2-methoxyethylamine (5.8 g, 77.3 mmol) and DIPEA (6 g, 46.4 mmol) in DMSO (80 mL) was heated at 65-70 °C for 24 h and monitored by chromatography until complete conversion. The
solution was then cooled to room temperature and a white solid precipitated gradually. Water (20 mL) was then added slowly within 1 h. The suspension was stirred for a further 1 h, filtered and dried to give the title compound as a white solid. 1 H NMR (DMSO-d6, 400 MHz): δ 9.87 (s, 1 H), 8.18 (s, 1 H), 7.20 (s, 1 H), 6.86 (s, 9H), 3.51 (t, 2H), 3.36 (t, 2H), 3.28 (s, 3H), 1.47 (s, 9H).
Step 12: 6-amino-4-((2-methoxyethyl)amino)nicotinonitrile.
A solution of 6-amino-4-fluoronicotinonitrile (obtained in step 9, 1 .10 g, 8.02 mmol) in DMA (20 mL) was treated with 2-methoxyethylamine (2.07 mL, 24.1 mmol) and DIPEA (4.20 mL, 24.1 mmol), heated to 50 °C and stirred for 15 h. The reaction mixture was cooled to room temperature and concentrated. The crude material was purified by normal phase chromatography (24 g silica gel cartridge, heptanes/EtOAc 100:0 to 0:100). The product containing fractions were concentrated and dried under vacuum to give the title compound as an off-white solid.
An alternative synthesis of 6-amino-4-((2-methoxyethyl)amino)nicotinonitrile is outlined below:
To tert-butyl N-{5-cyano-4-[(2-methoxyethyl)amino]pyridin-2-yl}carbamate (obtained in step 1 1 , 7g) was added 30-36% aqueous HCI (40 mL), the mixture stirred at room temperature for 30 minutes and monitored by chromatography until complete conversion. The solution was then basified with 20-30% NaOH solution to pH=9-10 and filtered to give a white solid. The solid was added to ethyl acetate (15 mL) and heated to 50-55 °C to form a clear solution. The solution was then cooled to 3-6 °C, stirred for 2-3 h and filtered. The wet cake was then dried to give the title compound as a white solid. 1H NMR (400 MHz, DMSO-d6) δ 7.92 (s, 1 H), 6.39 (s, 2H), 6.15 (t, 1 H), 5.61 (s, 1 H), 3.46 (t, 2H), 3.27 (s, 3H), 3.24 (q, 2H). (UPLC-MS 3) tR 0.62; ESI-MS 193.1 [M+H]+.
Step 13: N-(5-cyano-4-((2-methoxyethyl)amino)pyridin-2-yl)-7-(dimethoxymethyl)-6-((4-methyl-2-oxopiperazin-1 -yl)methyl)-3,4-dihydro-1 ,8-naphthyridine-1 (2H)-carboxamide.
A solution of 6-amino-4-((2-methoxyethyl)amino)nicotinonitrile (obtained in step 12, 481 mg, 2.50 mmol) in anhydrous DMF (1.5 mL) was added drop wise over 10 minutes to a mixture of di(1 H-1 ,2,4-triazol-1 -yl)methanone (410 mg, 2.50 mmol) and DMF (1 .5 mL) cooled at 0 °C. After stirring for 45 minutes at 0 °C the reaction mixture was allowed to warm to room temperature and after a further 90 minutes at room temperature a solution of 1 -((2-(dimethoxymethyl)-5,6,7,8-tetrahydro-1 ,8-naphthyridin-3-yl)methyl)-4-methylpiperazin-2-one (obtained in step 7, 418 mg, 1.00 mmol) in DMF (2 mL) was added. The reaction mixture was stirred for 17.5 h at room temperature, quenched by the addition of MeOH and evaporated. The residue was applied to a 80 g RediSep® silica column as a DCM solution and purified by normal phase chromatography, eluting with a gradient from DCM to 2% MeOH in DCM. Product containing fractions were combined and evaporated to give the title compound as an orange foam. 1H NMR (400 MHz, DMSO-d6) δ 13.50 (s, 1 H), 8.27 (s,
1 H), 7.52 (s, 1 H), 7.39 (s, 1 H), 6.93 (t, 1 H), 5.45 (s, 1 H), 4.65 (s, 2H), 3.94 – 3.89 (m, 2H), 3.54 -3.50 (m, 2H), 3.40 – 3.35 (m, 2H), 3.38 (s, 6H), 3.29 (s, 3H), 3.20 – 3.16 (m, 2H), 3.05 (s, 2H), 2.86 – 2.80 (m, 2H), 2.61 – 2.55 (m, 2H), 2.22 (s, 3H), 1 .94 – 1 .88 (m, 2H). (UPLC-MS 6) tR 0.72; ESI-MS 553.3 [M+H]+.
Step 14: /V-(5-cyano-4-((2-methoxyethyl)amino)pyridin-2-yl)-7-form
yl)methyl)-3,4-dihydro-1 ,8-naphthyridine-1 (2H)-carboxamide
Concentrated hydrochloric acid (0.40 mL) was added to a solution of A/-(5-cyano-4-((2-methoxyethyl)amino)pyridin-2-yl)-7-(dimethoxymethyl)-6-((4-methyl-2-oxopiperazin-1 -yl)methyl)-3,4-dihydro-1 ,8-naphthyridine-1 (2H)-carboxamide (obtained in step 13, 470 mg, 0.808 mmol) in THF (3 mL) and water (1 mL) at room temperature. After stirring for 3 h at room temperature saturated aqueous NaHC03 was added, the mixture extracted with DCM (3x), the organic layers dried over Na2S04 and evaporated. The residue was sonicated with EtOAc (6 mL) and pentane (6 mL) and then filtered. The white solid obtained was then dissolved in DCM (6 mL), EtOAc added (3 mL), the solution warmed, sealed and allowed to stand at room temperature for 2 h. Filtration and drying gave A/-(5-cyano-4-((2-methoxyethyl)amino)pyridin-2-yl)-7-formyl-6-((4-methyl-2-oxopiperazin-1 -yl)methyl)-3,4-dihydro-1 ,8-naphthyridine-1 (2H)-carboxamide as a white solid.
1 H NMR (400 MHz, DMSO-d6) δ 13.43 (s, 1 H), 10.06 (s, 1 H), 8.24 (s, 1 H), 7.49 (s, 1 H), 7.47 (s, 1 H), 6.96 (t, br, 1 H), 4.86 (s, 2H), 3.96 – 3.90 (m, 2H), 3.52 – 3.46 (m, 2H), 3.39 – 3.33 (m, 2H), 3.30 – 3.21 (m, 2H), 3.37 (s, 3H), 3.02 (s, 2H), 2.93 – 2.86 (m, 2H), 2.61 – 2.56 (m, 2H), 2.21 (s, 3H), 1 .95 – 1.85 (m, 2H). (UPLC-MS 6) tR0.70, ESI-MS 507.2, [M+H]+.
Step 15: A/-(5-cyano-4-((2-methoxyethyl)amino)pyridin-2-yl)-7-formyl-6-((4-methyl-2-oxopiperazin-1 -yl)methyl)-3,4-dihydro-1 ,8-naphthyridine-1 (2H)-carboxamide in citric acid form (1 :1 ).
A/-(5-cyano-4-((2-methoxyethyl)amino)pyridin-2-yl)-7-formyl-6-((4-methyl-2-oxopiperazin-1 -yl)methyl)-3,4-dihydro-1 ,8-naphthyridine-1 (2H)-carboxamide (obtained in step 14, 4g, 7.896 mmol) was stirred in propionic acid (29.3 g, 29.60mL) at 70 °C until dissolution was complete (20 minutes). The solution was cooled to 55 °C and a solution of citric acid in acetone (23% w/w) was added to it. Separately, a seed suspension was prepared by adding acetone (0.2 g, 0.252mL) to A/-(5-cyano-4-((2-methoxyethyl)amino)pyridin-2-yl)-7-formyl-6-((4-methyl-2-oxopiperazin-1 -yl)methyl)-3,4-dihydro-1 ,8-naphthyridine-1 (2H)-carboxamide in citric acid form (0.0185 g, 0.026 mmol). The seed suspension was added to the solution at 50 °C and the resulting suspension was left to stir at 50 °C for 40 minutes. A further solution of citric acid in acetone (26.6g, 2.51 % w/w, 33.63 mL) was added to the reaction over 380 minutes. The resulting suspension was stirred for a further 120 minutes and cooled to 20 °C with stirring over 4 hours. The suspension was stirred for another 12 hours
before filtering the suspension under vacuum and washing the resulting solid with a propionic acid: acetone solution (1 : 1 , 7g, 7.96ml_) at room temperature. The solid was further washed with acetone (7g, 8.85ml_) at room temperature. The resulting solid was dried in an oven at 40 °C and 5mbar to give the title compound as a light orange solid (5.2g, 7.443 mmol). (mw 698.70), mp (DSC) 168.8 °C (onset).
XRPD analysis showed the same pattern as with particles obtained by a process described in PCT/I B2014/065585 (reference example 1 ) – see Figure 5.
Example 1a
Steps 1 to 14 were carried out as described in example 1 .
Step 15a: A/-(5-cyano-4-((2-methoxyethyl)amino)pyridin-2-yl)-7-formyl-6-((4-methyl-2-oxopiperazin-1 -yl)methyl)-3,4-dihydro-1 ,8-naphthyridine-1 (2H)-carboxamide in citric acid form (1 : 1 )
A/-(5-cyano-4-((2-methoxyethyl)amino)pyridin-2-yl)-7-formyl-6-((4-methyl-2-oxopiperazin-1 -yl)methyl)-3,4-dihydro-1 ,8-naphthyridine-1 (2H)-carboxamide (obtained in step 14, 5g, 9.930 mmol) was stirred in propionic acid (33.5 g, 33.84ml_) at 60 °C. Once A/-(5-cyano-4-((2-methoxyethyl)amino)pyridin-2-yl)-7-formyl-6-((4-methyl-2-oxopiperazin-1 -yl)methyl)-3,4-dihydro-1 ,8-naphthyridine-1 (2H)-carboxamide had dissolved, anhydrous citric acid powder (0.19g, 0.9889 mmol) was added. The resulting suspension was heated to 70 °C and sonicated for 5 minutes to ensure full dissolution. The resulting solution was cooled to 50 °C and a solution of citric acid in ethyl acetate (3.7 g, 1 .3% citric acid in ethyl acetate) was added over 20 minutes. Seeds of N-(5-cyano-4-((2-methoxyethyl)amino)pyridin-2-yl)-7-formyl-6-((4-methyl-2-oxopiperazin-1 -yl)methyl)-3,4-dihydro-1 ,8-naphthyridine-1 (2H)-carboxamide in citric acid form (0.02 g) were added to the solution and the suspension was aged for 15 minutes. Another aliquot of citric acid in ethyl acetate (128g, 1 .3% citric acid in ethyl acetate) was added to the suspension over 1 1 .85hours. The suspension was left to stir for over 4 hours. The suspension was then filtered under vacuum (500mbar) and the resulting solid was washed firstly with a propionic acid: ethyl acetate solution (1 : 1 , 7g, 7.44ml_) at room temperature and then with ethyl acetate (12g, 13.38ml_) at room temperature. The resulting solid was dried in an oven at 40 °C and 5mbar to give the title compound as a light orange solid (6.3 g, 9.074 mmol).
XRPD analysis showed the same pattern as with particles obtained by a process described in PCT/I B2014/065585 (reference example 1 ) – see Figure 5.
Reference example 1 (described in PCT/IB2014/065585) – V-(5-cyano-4-((2-methoxyethyl)amino)pyridin-2-yl)-7-formyl-6-((4-methyl-2-oxopiperazin-1 -yl)methyl)-3,4-dihvdro-1 ,8-naphthyridine-1 (2H)-carboxamide in citric acid form (1 :1 )
Steps 1 to 14 were carried out as described in example 1.
Reference Step 15 – /V-(5-cvano-4-((2-methoxyethyl‘)amino‘)pyridin-2-yl‘)-7-formyl-6-((4-methyl-2-oxopiperazin-1 -yl‘)methyl‘)-3,4-dihvdro-1 ,8-naphthyridine-1 (2H‘)-carboxamide in citric acid form (1 :1 )
A solution of citric acid (96.9 mg) in acetone (5 mL) was prepared at room temperature (0.1 M). A portion of the 0.1 M citric acid in acetone solution (2 mL) was then added to a suspension of Λ/-(5-cyano-4-((2-methoxyethyl)amino)pyridin-2-yl)-7-formyl-6-((4-methyl-2-oxopiperazin-1 -yl)methyl)-3,4-dihydro-1 ,8-naphthyridine-1 (2H)-carboxamide (100 mg) in acetone (4 mL) and the mixture sonicated for 1 minute then heated at 55 °C with stirring for 2 h before slowly cooling to room temperature. The white solid was then collected by filtration, washing 2x with acetone (2 mL), and dried for 18 h at 40 °C under vacuum to give the title salt.
Alternatively, N-(5-cyano-4-((2-methoxyethyl)amino)pyridin-2-yl)-7-formyl-6-((4-methyl-2-oxopiperazin-1 -yl)methyl)-3,4-dihydro-1 ,8-naphthyridine-1 (2H)-carboxamide (6.5 g, 12.83 mmol) was placed in a 500ml 4-flask reactor. 49 mL of glacial acetic acid was added and the resulting suspension was stirred at 23 °C until a clear mixture was obtained. In a separate flask, anhydrous 2-hydroxypropane-1 ,2,3-tricarboxylic acid (2.59 g, 13.47 mmol, 1 .05 equiv.) was dissolved in 49 mL of glacial acetic acid at 50 °C until a clear solution was obtained. This solution was then added at 23°C to the N-(5-cyano-4-((2-methoxyethyl)amino)pyridin-2-yl)-7-formyl-6-((4-methyl-2-oxopiperazin-1 -yl)methyl)-3,4-dihydro-1 ,8-naphthyridine-1 (2H)-carboxamide solution previously prepared. This mixture was stirred for 30 min at 23 °C and then added dropwise over 1 h to 192 mL of ethyl acetate warmed to 75 °C. The temperature remained constant over the addition. At the end of the addition, the temperature of the mixture was cooled slowly to 23 °C and let 16h at this temperature under gentle stirring. The suspension was cooled to 5-10 °C and filtered. The cake was washed with 15 mL of ethyl acetate and 15 mL of acetone. The wet cake (ca 8.5g) was transferred in a 500 mL flask containing 192 mL of dry acetone. The resulting suspension was refluxed for 24h. The suspension was filtered and the cake was washed with 2 times 15 mL of dry acetone then dried at 50 °C under vacuum for several hours to give the title salt.
PATENT
The synthesis of /V-(5-cyano-4-((2-methoxyethyl)amino)pyridin-2-yl)-7-formyl-6-((4-methyl-2-oxopiperazin-1 -yl)methyl)-3,4-dihydro-1 ,8-naphthyridine-1 (2H)-carboxamide (abbreviated herein as CPi and also named as Example 83) and salts thereof is disclosed in PCT/IB2014/065585, the content of which are incorporated by reference, as described herein below:
Example 83: /V-(5-cyano-4-((2-methoxyethyl)amino)pyridin-2-yl)-7-formyl-6-((4-methyl-2-oxopiperazin-1 -yl)methyl)-3,4-dihydro-1 ,8-naphthyridine-1 (2H)-carboxamide.

Concentrated hydrochloric acid (0.40 ml) was added to a solution of /V-(5-cyano-4-((2-methoxyethyl)amino)pyridin-2-yl)-7-(dimethoxymethyl)-6-((4-methyl-2-oxopiperazin-1 -yl)methyl)-3,4-dihydro-1 ,8-naphthyridine-1 (2H)-carboxamide (intermediate 80, 470 mg, 0.808 mmol) in THF (3 ml) and water (1 ml) at room temperature. After stirring for 3 h at room temperature saturated aqueous NaHC03 was added, the mixture extracted with DCM (3x), the organic layers dried over Na2S04 and evaporated. The residue was sonicated with EtOAc (6 ml) and pentane (6 ml) and then filtered. The white solid obtained was then dissolved in DCM (6 ml), EtOAc added (3 ml), the solution warmed, sealed and allowed to stand at room temperature for 2 h. Filtration and drying gave the title compound as a white solid.
1H NMR (400 MHz, DMSO-c/6) δ 13.43 (s, 1 H), 10.06 (s, 1 H), 8.24 (s, 1 H), 7.49 (s, 1 H), 7.47 (s, 1 H), 6.96 (t, br, 1 H), 4.86 (s, 2H), 3.96 – 3.90 (m, 2H), 3.52 – 3.46 (m, 2H), 3.39 – 3.33 (m, 2H), 3.30 – 3.21 (m, 2H), 3.37 (s, 3H), 3.02 (s, 2H), 2.93 – 2.86 (m, 2H), 2.61
– 2.56 (m, 2H), 2.21 (s, 3H), 1 .95 – 1 .85 (m, 2H).
(UPLC-MS 6) tR 0.70, ESI-MS 507.2, [M+H]+.
The following salts were prepared from the above free form form of /V-(5-cyano-4-((2-methoxyethyl)amino)pyridin-2-yl)-7-formyl-6-((4-methyl-2-oxopiperazin-1 -yl)methyl)-3,4-dihydro-1 ,8-naphthyridine-1 (2H)-carboxamide by precipitation with the appropriate counterions.
Malate with 1 :1 stoichiometry (mw 640.66), mp (DSC) 181 .1 °C (onset): Acetone (2 ml) was added to a mixture of malic acid (26.4 mg, 0.197 mmol) and /V-(5-cyano-4-((2-methoxyethyl)amino)pyridin-2-yl)-7-formyl-6-((4-methyl-2-oxopiperazin-1 -yl)methyl)-3,4-dihydro-1 ,8-naphthyridine-1 (2H)-carboxamide (100 mg, 0.197 mmol) and the mixture heated on a mini-block with heating-cooling cycles from 55 to 5 °C for 7 repeat cycles (heating rate: 1 .5 °C/min, cooling rate: 0.25 °C/min). The white solid was collected by centrifugation and dried for 18 h at 40 °C to give the title salt.
Tartrate with 1 :0.5 stoichiometry (mw 581 .72), mp (DSC) 176.7 °C (onset). A solution of tartaric acid (75.7 mg) in methanol (5 ml) was prepared at room temperature (0.1 M). A portion of the 0.1 M tartaric acid in acetone solution (2 ml) was then added to a suspension of /V-(5-cyano-4-((2-methoxyethyl)amino)pyridin-2-yl)-7-formyl-6-((4-methyl-2-oxopiperazin-1 -yl)methyl)-3,4-dihydro-1 ,8-naphthyridine-1 (2H)-carboxamide (100 mg) in methanol (4 ml) and the mixture sonicated for 1 minute then heated at 55 °C with stirring for 2 h. The white solid was then collected by filtration, washing 2x with methanol (2 ml), and dried for 18 h at 40 °C under vacuum to give the title salt.
Tartrate with 1 :1 stoichiometry (mw 656.66), mp (DSC) 169.9 °C (onset): A solution of tartaric acid (75.7 mg) in acetone (5 ml) was prepared at room temperature (0.1 M). A portion of the 0.1 M tartaric acid in acetone solution (2 ml) was then added to a suspension of /V-(5-cyano-4-((2-methoxyethyl)amino)pyridin-2-yl)-7-formyl-6-((4-methyl-2-oxopiperazin-1 -yl)methyl)-3,4-dihydro-1 ,8-naphthyridine-1 (2H)-carboxamide (100 mg) in methanol (4 ml) and the mixture sonicated for 1 minute then heated at 55 °C with stirring for 2 h. The white solid was then collected by filtration, washing 2x with acetone (2 ml), and dried for 18 h at 40 °C under vacuum to give the title salt.
Citrate with 1 :0.5 stoichiometry (mw 602.73), mp (DSC) 168.4 °C (onset): A solution of citric acid (96.9 mg) in methanol (5 ml) was prepared at room temperature (0.1 M). A portion of the 0.1 M citric acid in methanol solution (2 ml) was then added to a suspension of /V-(5-cyano-4-((2-methoxyethyl)amino)pyridin-2-yl)-7-formyl-6-((4-methyl-2-oxopiperazin-1 -yl)methyl)-3,4-dihydro-1 ,8-naphthyridine-1 (2H)-carboxamide (100 mg) in methanol (4 ml) and the mixture sonicated for 1 minute then heated at 55 °C with
stirring for 2 h. The white solid was then collected by filtration, washing 2x with acetone (2 ml), and dried for 18 h at 40 °C under vacuum to give the title salt.
Citrate with 1 :1 stoichiometry (mw 698.70), mp (DSC) 168.8 °C (onset): A solution of citric acid (96.9 mg) in acetone (5 ml) was prepared at room temperature (0.1 M). A portion of the 0.1 M citric acid in acetone solution (2 ml) was then added to a suspension of /V-(5-cyano-4-((2-methoxyethyl)amino)pyridin-2-yl)-7-formyl-6-((4-methyl-2-oxopiperazin-1 -yl)methyl)-3,4-dihydro-1 ,8-naphthyridine-1 (2H)-carboxamide (100 mg) in acetone (4 ml) and the mixture sonicated for 1 minute then heated at 55 °C with stirring for 2 h before slowly cooling to room temperature. The white solid was then collected by filtration, washing 2x with acetone (2 ml), and dried for 18 h at 40 °C under vacuum to give the title salt.
Alternatively, N-(5-cyano-4-((2-methoxyethyl)amino)pyridin-2-yl)-7-formyl-6-((4-methyl-2-oxopiperazin-1 -yl)methyl)-3,4-dihydro-1 ,8-naphthyridine-1 (2H)-carboxamide (6.5 g, 12.83 mmol) was placed in a 500ml 4-flask reactor. 49 ml of glacial acetic acid was added and the resulting suspension was stirred at 23 °C until a clear mixture was obtained. In a separate flask, anhydrous 2-hydroxypropane-1 ,2,3-tricarboxylic acid (2.59 g, 13.47 mmol, 1 .05 equiv.) was dissolved in 49 ml of glacial acetic acid at 50 °C until a clear solution was obtained. This solution was then added at 23°C to the N-(5-cyano-4-((2-methoxyethyl)amino)pyridin-2-yl)-7-formyl-6-((4-methyl-2-oxopiperazin-1 -yl)methyl)-3,4-dihydro-1 ,8-naphthyridine-1 (2H)-carboxamide solution previously prepared. This mixture was stirred for 30 min at 23 °C and then added dropwise over 1 h to 192 ml of ethyl acetate warmed to 75 °C. The temperature remained constant over the addition. At the end of the addition, the temperature of the mixture was cooled slowly to 23 °C and let 16h at this temperature under gentle stirring. The suspension was cooled to 5-10 °C and filtered. The cake was washed with 15 ml of ethyl acetate and 15 ml of acetone. The wet cake (ca 8.5g) was transferred in a 500 ml flask containing 192 ml of dry acetone. The resulting suspension was refluxed for 24h. The suspension was filtered and the cake was washed with 2 times 15 ml of dry acetone then dried at 50 °C under vacuum for several hours to give the title salt.
Intermediate 80: N-(5-cyano-4-((2-methoxyethyl)amino)pyridin-2-yl)-7- (dimethoxymethyl)-6-((4-methyl-2-oxopiperazin-1 -yl)methyl)-3,4-dihydro-1 ,8-naphthyridine-1 (2H)-carboxamide.
A solution of 6-amino-4-((2-methoxyethyl)amino)nicotinonitrile (intermediate 75, 481 mg, 2.50 mmol) in anhydrous DMF (1 .5 ml) was added drop wise over 10 minutes to a mixture of di(1 H-1 ,2,4-triazol-1 -yl)methanone (410 mg, 2.50 mmol) and DMF (1 .5 ml) cooled at 0 °C. After stirring for 45 minutes at 0 °C the reaction mixture was allowed to warm to room temperature and after a further 90 minutes at room temperature a solution of 1 -((2-(dimethoxymethyl)-5,6,7,8-tetrahydro-1 ,8-naphthyridin-3-yl)methyl)-4-methylpiperazin-2-one (intermediate 81 , 418 mg, 1 .00 mmol) in DMF (2 ml) was added. The reaction mixture was stirred for 17.5 h at room temperature, quenched by the addition of MeOH and evaporated. The residue was applied to a 80 g RediSep® silica column as a DCM solution and purified by normal phase chromatography, eluting with a gradient from DCM to 2% MeOH in DCM. Product containing fractions were combined and evaporated to give the title compound as an orange foam. 1H NMR (400 MHz, DMSO-c/6) δ 13.50 (s, 1 H), 8.27 (s, 1 H), 7.52 (s, 1 H), 7.39 (s, 1 H), 6.93 (t, 1 H), 5.45 (s, 1 H), 4.65 (s, 2H), 3.94 – 3.89 (m, 2H), 3.54 – 3.50 (m, 2H), 3.40 – 3.35 (m, 2H), 3.38 (s, 6H), 3.29 (s, 3H), 3.20 – 3.16 (m, 2H), 3.05 (s, 2H), 2.86 – 2.80 (m, 2H), 2.61 – 2.55 (m, 2H), 2.22 (s, 3H), 1 .94 – 1 .88 (m, 2H). (UPLC-MS 6) tR 0.72; ESI-MS 553.3 [M+H]+.
Intermediate 81 : 1 -((2-(dimethoxymethyl)-5,6,7,8-tetrahydro-1 ,8-naphthyridin-3-yl)methyl)-4-methylpiperazin-2-one.
Sodium triacetoxyborohydride (3.10 g, 14.61 mmol) was added to a mixture of 2-(dimethoxymethyl)-5,6,7,8-tetrahydro-1 ,8-naphthyridine-3-carbaldehyde (intermediate 41 , 2.30 g, 9.74 mmol), ethyl 2-((2-aminoethyl)(methyl)amino)acetate dihydrochloride (intermediate 82, 2.6 g, 14.61 mmol) and triethylamine (6.75 ml, 48.7 mmol) in 1 ,2-dichloroethane (20 ml) at room temperature. The reaction mixture was stirred for 21 h at room temperature and additional sodium triacetoxyborohydride (2.6 g, 9.74 mmol) was added. After a further 4 h stirring at room temperature, again additional sodium triacetoxyborohydride (1 .3 g, 4.87 mmol) was added and the reaction maintained at 4 °C for 2.5 days. The reaction mixture was then warmed to room temperature, saturated aqueous NaHC03 solution added, the mixture extracted with DCM (3x), the combined organic layers dried over Na2S04 and evaporated. The residue was applied to a 120 g RediSep® silica column as a DCM solution and purified by normal phase chromatography, eluting with a gradient from DCM to 10% MeOH in DCM. Product containing fractions were combined and evaporated to give the title compound as an orange foam. 1H NMR (400 MHz, CDCI3) δ 7.08 (s, 1 H), 5.30 (s, br, 1 H), 5.20 (s, 1 H), 4.69 (s, 2H), 3.44 – 3.34 (m, 2H), 3.40 (s, 6H), 3.22 – 3.15 (m, 2H), 3.24 (s, 2H), 2.71 -2.64 (m, 2H), 2.58 – 2.50 (m, 2H), 2.31 (s, 3H), 1 .98 – 1 .82 (m, 2H). (UPLC-MS 6) tR 0.33; ESI-MS 335.3 [M+H]+.
Intermediate 82: ethyl 2-((2-aminoethyl)(methyl)amino)acetate dihydrochloride.
Concentrated hydrochloric acid (10 ml) was added to a solution of ethyl 2-((2-((tert-butoxycarbonyl)amino)ethyl)(methyl)amino)acetate (intermediate 83, 3.05 g, 1 1 .13 mmol) in THF (20 ml) and EtOH (100 ml) at room temperature. After stirring 1 h at room temperature the reaction mixture was evaporated, ethanol (20 ml) added, evaporated, further ethanol (50 ml) added and then stirred at 60 °C for 70 min. The cooled reaction
mixture was then evaporated to give the title compound as a pale-yellow glass. 1H NMR (400 MHz, DMSO-c/6) δ 8.58 (s, br, 3H), 4.19 (q, 2H), 4.26 – 4.15 (m, 2H), 3.44 (s, br, 2H), 3.21 (s, br, 2H), 2.88 (s, 3H), 1 .21 (t, 3H).
Intermediate 83: ethyl 2-((2-((tert-butoxycarbonyl)amino)ethyl)(methyl)amino)acetate.
Ethyl bromoacetate (1 .27 ml, 1 1 .48 mmol) was added to a mixture of tert-butyl (2-(methylamino)ethyl)carbamate (2.0 g, 1 1 .48 mmol), triethylamine (4.81 ml) and THF (24 ml) at 0 °C. After stirring 24 h at room temperature the reaction mixture was partitioned between saturated aqueous NaHC03 and DCM, extracted 2x with DCM, the organic layers dried over Na2S04 and evaporated to give the title compound as a clear pale-yellow oil. 1 H NMR (400 MHz, CDCI3) δ 5.20 (s, br, 1 H), 4.18 (q, 2H), 3.24 (s, 2H), 3.22 -3.16 (m, 2H), 2.65 – 2.61 (m, 2H), 2.38 (s, 3H), 1 .42 (s, 9H), 1 .24 (t, 3H).
Intermediate 41 : 2-(dimethoxymethyl)-5,6,7,8-tetrahydro-1 ,8-naphthyridine-3-carbaldehyde.
To a solution of 6-bromo-7-(dimethoxymethyl)-1 ,2,3,4-tetrahydro-1 ,8-naphthyridine
(intermediate 12, 15.0 g, 52.2 mmol) in THF (400 ml) at -78 °C under argon, was added MeLi (1 .6 M in Et20, 32.6 ml, 52.2 mmol), the solution was stirred for 5 min, then n-BuLi (1 .6 M in hexane, 35.9 ml, 57.5 mmol) was added slowly and the solution was stirred for 20 min. THF (100 ml) was added to the reaction at – 78 °C. Subsequently, n-BuLi (1 .6 M in hexane, 49.0 ml, 78 mmol) was added and the reaction mixture was stirred for 20 min, then again n-BuLi (1 .6 M in hexane, 6.53 ml, 10.45 mmol) was added and the mixture was stirred for 10 min at – 78 °C. DMF (2.10 ml, 27.2 mmol) was added and the reaction mixture was stirred at -78 °C for 45 min, then it was allowed to warm to room
temperature, poured into sat. aq. NH4CI and extracted twice with DCM. The combined organic phases were dried over Na2S04, filtered and evaporated to give the title compound as an orange oil. (UPLC-MS 3) tR 0.63 min; ESI-MS 237.2 [M+H]+.
Intermediate 12: 6-bromo-7-(dimethoxymethyl)-1 ,2,3,4-tetrahydro-1 ,8-naphthyridine.
Into a 3 I 4-necked round-bottom flask was placed 7-(dimethoxymethyl)-1 ,2,3,4-tetrahydro-1 ,8-naphthyridine (intermediate 4, 1 14.6 g, 550.3mmol) in acetonitrile (2 I). This was followed by the addition of NBS (103 g, 578 mol) in portions with stirring at 25 °C. The resulting solution was stirred for 30 min at 25 °C. The resulting mixture was concentrated under vacuum and the residue was diluted with 1000 ml of diethylether. The mixture was washed with 3×100 ml of ice/water. The aqueous phase was extracted with 2×100 ml of diethylether and the organic layers were combined. The resulting mixture was washed with 1 x100 ml of brine, dried over sodium sulfate and concentrated under vacuum to give the title compound as a light yellow solid. LC-MS: (ES, m/z):
286.03 [M+H]+. 1H-NMR: (300MHz, CDCI3) δ 1 .86 – 1 .94 (2H, m), 2.70 – 2.74 (2H, m), 3.9 – 3.43 (2H, m), 3.47 (6H, s), 5.23 (1 H, s), 5.58 (1 H, s), 7.29 (1 H, s).
Intermediate 4: 7-(dimethoxymethyl)-1 ,2,3,4-tetrahydro-1 ,8-naphthyridine.
The procedure described in J. Org. Chem. , 2004, 69 (6), pp 1959-1966 was used. Into a 5-I pressure tank reactor (5 atm) was placed 2-(dimethoxymethyl)-1 ,8-naphthyridine (intermediate 5, 200 g, 979 mmol), ethanol (3 I), Pt02 (12 g). The reactor was evacuated and flushed three times with nitrogen, followed by flushing with hydrogen. The mixture was stirred overnight at 23 °C under an atmosphere of hydrogen. This reaction was repeated four times. The solids were filtered out and the resulting mixture was concentrated under vacuum to give the title compound as a yellow solid.
Intermediate 5: 2-(dimethoxymethyl)-1 ,8-naphthyridine.
The procedure described in J. Org. Chem. , 2004, 69 (6), pp 1959-1966 was used. Into a 20 I 4-necked round-bottom flask was placed 2-aminopyridine-3-carbaldehyde (1000 g, 8.19 mol), 1 ,1 -dimethoxypropan-2-one (1257 g, 10.64 mol), ethanol (10 I), and water (2 I). This was followed by the addition of a solution of sodium hydroxide (409.8 g, 10.24 mol) in water (1000 ml) drop wise with stirring at 0-15 °C. The solution was stirred for 3 h at 0-20 °C and then concentrated under vacuum. The resulting solution was extracted with 3×1200 ml of ethyl acetate and the organic layers were combined. The mixture was dried over sodium sulfate and concentrated under vacuum. The residue was washed with 3×300 ml of hexane and the solid was collected by filtration. This resulted in the title compound as a yellow solid. 1H-NMR (400 MHz, DMSO-c/6) δ 9.1 1 (dd, 1 H), 8.53 (d, 1 H), 8.50 (dd, 1 H), 7.73 (d, 1 H), 7.67 (dd, 1 H), 5.44 (s, 1 H), 3.41 (s, 6H).
Intermediate 75: 6-amino-4-((2-methoxyethyl)amino)nicotinonitrile.
A solution of 6-amino-4-fluoronicotinonitrile (intermediate 21 , 1 .10 g, 8.02 mmol) in DMA (20 ml) was treated with 2-methoxyethylamine (2.07 ml, 24.1 mmol) and DIPEA (4.20 ml_, 24.1 mmol), heated to 50 °C and stirred for 15 h. The reaction mixture was cooled to room temperature and concentrated. The crude material was purified by normal phase chromatography (24 g silica gel cartridge, heptanes/EtOAc 100:0 to 0:100). The product containing fractions were concentrated and dried under vacuum to give the title compound as an off-white solid.
An alternative synthesis of 6-amino-4-((2-methoxyethyl)amino)nicotinonitrile is outlined below:
To fe/ -butyl N-{5-cyano-4-[(2-methoxyethyl)amino]pyridin-2-yl}carbamate (intermediate 287, 7g) was added 30-36% aqueous HCI (40 ml), the mixture stirred at room temperature for 30 minutes and monitored by chromatography until complete conversion. The solution was then basified with 20-30% NaOH solution to pH=9-10 and filtered to give a white solid. The solid was added to ethyl acetate (15 ml) and heated to 50-55 °C to form a clear solution. The solution was then cooled to 3-6 °C, stirred for 2-3 h and filtered. The wet cake was then dried to give the title compound as a white solid. 1H NMR (400 MHz, DMSO-c/6) δ 7.92 (s, 1 H), 6.39 (s, 2H), 6.15 (t, 1 H), 5.61 (s, 1 H), 3.46 (t, 2H), 3.27 (s, 3H), 3.24 (q, 2H). (UPLC-MS 3) tR 0.62; ESI-MS 193.1 [M+H]+.
1H-NMR (400 MHz, DMSO-c/6) δ 7.14 (d, 1 H), 6.51 (d, 1 H), 6.47 – 6.41 (m, 1 H), 4.98 (s, 1 H), 3.28 – 3.19 (m, 2H), 3.23 (s, 6H), 2.64 (t, 2H), 1 .73 – 1 .79 (m, 2H).
Intermediate 21 : 6-amino-4-fluoronicotinonitrile.
4-fluoro-5-iodopyridin-2-amine (intermediate 22, 240 g, 1 mol), zinc cyanide (125 g, 1 .05 mol), zinc (13 g, 0.2 mol), Pd2(dba)3 (25 g, 25 mmol) and dppf (55 g, 0.1 mol) in DMA (800 ml) were degassed and charged into the round bottom flask under nitrogen. The mixture was stirred at 100 °C for 3 h. The reaction mixture was diluted with 5% NaHC03 (2 I), extracted with EtOAc (4 x 600 ml). The combined organic layers were washed with 5% NaOH (1 I), dried over Na2S04, concentrated to 700 ml. The resulting organic phase was eluted through silica gel column with EtOAc (1 .7 I). The combined organic filtrate was washed with 2 M HCI (3 x 800 ml). The pH of the aqueous phase was adjusted to 10 with saturated NaHC03. The aqueous phase was extracted whit DCM (3 x 500 ml). The combined DCM was dried over Na2S04 and concentrated. The residue was further purified by column chromatography (eluted with pentane: EtOAc 10:1 to 3:2) followed by recrystallization from pentane/EtOAc 3/1 to give the title compound as white solid. 1H NMR (400 MHz, DMSO-c/6) δ 8.40 (d, 1 H), 7.40 (s, 2H), 6.34 (d, 1 H).
Intermediate 22: 4-fluoro-5-iodopyridin-2-amine.
A suspension of 4-fluoropyridin-2-amine (336 g, 2.5 mol) and NIS (745 g, 2.75 mol) in MeCN (9 I) was treated with TFA (1 14 g, 1 mol). The reaction mixture was then stirred at room temperature for 8 h. The reaction mixture was diluted with EtOAc (10 I), washed with sat. aq. Na2S203 (2 x 5 I), brine (4 x 5 I). The combined organic layers were dried over Na2S04, filtered and concentrated to get the crude product. The crude product was purified by recrystallization from EtOAc/pentane (1/10) to afford the title compound as a white solid. 1H NMR (400 MHz, DMSO-c/6) δ 8.14 (d, 1 H), 6.45 (s, 2H), 6.33 (d, 1 H).
Intermediate 287: fe/ -butyl (5-cyano-4-((2-methoxyethyl)amino)pyridin-2-yl)carbamate.
A mixture of tert-butyl (4-chloro-5-cyanopyridin-2-yl)carbamate (intermediate 288, 9.8 g, 38.6 mmol), 2-methoxyethylamine (5.8 g, 77.3 mmol) and DIPEA (6 g, 46.4 mmol) in DMSO (80 ml) was heated at 65-70 °C for 24 h and monitored by chromatography until complete conversion. The solution was then cooled to room temperature and a white solid precipitated gradually. Water (20 ml) was then added slowly within 1 h. The suspension was stirred for a further 1 h, filtered and dried to give the title compound as a white solid. 1H NMR (DMSO-d6, 400 MHz): δ 9.87 (s, 1 H), 8.18 (s, 1 H), 7.20 (s, 1 H), 6.86 (s, 9H), 3.51 (t, 2H), 3.36 (t, 2H), 3.28 (s, 3H), 1 .47 (s, 9H).
Intermediate 288: tert-butyl (4-chloro-5-cyanopyridin-2-yl)carbamate.
A mixture of 2,4-dichloro-5-cyanopyridine (10g, 57.8 mmol), fe/ -butyl carbamate (8.2 g, 70.5 mmol), Pd(OAc)2 (0.26 g, 1 .1 mmol), Xantphos (1 .34 g, 2.3mmol) and K2C03 (12 g, 87 mmol) in THF (150 ml) was degassed 3x with nitrogen. The mixture was then heated at 70 °C for 4-5 h and monitored by chromatography until complete conversion. Following completion of the reaction, additional THF (100 ml) was added and heated the mixture at 70 °C for additional 1 h and then cooled to room temperature. The suspension was then filtered through a pad of celite to remove the solid. The filtrate was then concentrated and azotropically distilled with ethyl acetete before filtering to give the title compound. 1H NMR (DMSO-d6, 400 MHz): δ 10.82 (s, 1 H), 8.79 (s, 1 H), 8.09 (s, 1 H), 1 .49 (s, 9H).
/////////////FGF 401, 1708971-55-4, PHASE 1, Hepatocellular carcinoma, Solid tumours, Novartis, Novartis Oncology, Antineoplastics, Type 4 fibroblast growth factor receptor antagonists, NVP-FGF-401, Nicole Buschmann, Robin Alec Fairhurst, Pascal Furet, Thomas Knöpfel, Catherine Leblanc, Robert Mah, Pierre NIMSGERN, Sebastien RIPOCHE, Lv LIAO, Jing XIONG, Xianglin ZHAO, Bo Han, Can Wang,
Now in #MEDI 1st time disclosures Robin Fairhurst of @Novartis will also talk about an FGFR inhibitor. They are popular! #ACSSanFran
CN4CC(=O)N(Cc1cc(C=O)nc2N(CCCc12)C(=O)Nc3cc(NCCOC)c(C#N)cn3)CC4
OSILODROSTAT for Treatment of Cushing’s Syndrome
OSILODROSTAT
LCI 699, LCI 699NX
Novartis Ag INNOVATOR
UNII-5YL4IQ1078, CAS 928134-65-0
- Molecular FormulaC13H10FN3
- Average mass227.237 Da
- Originator Novartis
- Class Antihypertensives; Fluorobenzenes; Imidazoles; Nitriles; Pyridines; Small molecules
- Mechanism of Action Aldosterone synthase inhibitors
- Phase III Cushing syndrome
- Phase I Liver disorders
- Discontinued Heart failure; Hypertension; Solid tumours
Most Recent Events
- 27 Feb 2016 Novartis plans the phase III LINC-4 trial for Cushing’s syndrome in Greece, Thailand, Poland, Turkey, Russia, Brazil, Belgium, Spain, Denmark, Switzerland and USA (PO) (NCT02697734)
- 12 Jun 2015 Novartis plans a phase II trial for Cushing syndrome in Japan (NCT02468193)
- 01 Apr 2015 Phase-I clinical trials in Liver disorders in USA (PO)
Osilodrostat phosphate
CAS: 1315449-72-9
MF, C13-H10-F-N3.H3-O4-P
MW, 325.2347
- LCI 699AZA
An orally active aldosterone-synthase inhibitor.
for Treatment of Cushing’s Syndrome
4-((5R)-6,7-Dihydro-5H-pyrrolo(1,2-c)imidazol-5-yl)-3-fluorobenzonitrile dihydrogen phosphate
Aromatase inhibitor; Cytochrome P450 11B1 inhibitor
MORE SYNTHESIS COMING, WATCH THIS SPACE…………………..
SYNTHESIS
ACS Medicinal Chemistry Letters, 4(12), 1203-1207; 2013
REMIND ME, amcrasto@gmail.com, +919323115463
Osilodrostat (INN, USAN) (developmental code name LCI-699) is an orally active, nonsteroidal corticosteroid biosynthesis inhibitorwhich is under development by Novartis for the treatment of Cushing’s syndrome and pituitary ACTH hypersecretion (a specific subtype of Cushing’s syndrome).[1][2] It specifically acts as a potent and selective inhibitor of aldosterone synthase (CYP11B2) and at higher dosages of 11β-hydroxylase (CYP11B1).[2] The drug was also under development for the treatment of heart failure, hypertension, and solid tumors, but development was discontinued for these indications.[1] As of 2017, osilodrostat is in phase III and phase II clinical trialsfor treatment of pituitary ACTH hypersecretion and Cushing’s syndrome, respectively.[1]
Osilodrostat, as modulators of 11-β-hydroxylase, useful for treating a disorder ameliorated 11-β-hydroxylase inhibition eg Cushing’s disease, hypertension, congestive heart failure, metabolic syndrome, liver diseases, cerebrovascular diseases, migraine headaches, osteoporosis or prostate cancer.
Novartis is developing osilodrostat, an inhibitor of aldosterone synthase and aromatase, for treating Cushing’s disease. In July 2016, osilodrostat was reported to be in phase 3 clinical development.
The somatostatin analog pasireotide and the 11β-hydroxylase inhibitor osilodrostat (LCI699) reduce cortisol levels by distinct mechanisms of action. There exists a scientific rationale to investigate the clinical efficacy of these two agents in combination. This manuscript reports the results of a toxicology study in rats, evaluating different doses of osilodrostat and pasireotide alone and in combination. Sixty male and 60 female rats were randomized into single-sex groups to receive daily doses of pasireotide (0.3mg/kg/day, subcutaneously), osilodrostat (20mg/kg/day, orally), osilodrostat/pasireotide in combination (low dose, 1.5/0.03mg/kg/day; mid-dose, 5/0.1mg/kg/day; or high dose, 20/0.3mg/kg/day), or vehicle for 13weeks. Mean body-weight gains from baseline to Week 13 were significantly lower in the pasireotide-alone and combined-treatment groups compared to controls, and were significantly higher in female rats receiving osilodrostat monotherapy. Osilodrostat and pasireotide monotherapies were associated with significant changes in the histology and mean weights of the pituitary and adrenal glands, liver, and ovary/oviduct. Osilodrostat alone was associated with adrenocortical hypertrophy and hepatocellular hypertrophy. In combination, osilodrostat/pasireotide did not exacerbate any target organ changes and ameliorated the liver and adrenal gland changes observed with monotherapy. Cmax and AUC0-24h of osilodrostat and pasireotide increased in an approximately dose-proportional manner. In conclusion, the pasireotide and osilodrostat combination did not exacerbate changes in target organ weight or toxicity compared with either monotherapy, and had an acceptable safety profile; addition of pasireotide to the osilodrostat regimen may attenuate potential adrenal gland hyperactivation and hepatocellular hypertrophy, which are potential side effects of osilodrostat monotherapy.

The somatostatin analog pasireotide and the 11β-hydroxylase inhibitor osilodrostat (LCI699) reduce cortisol levels by distinct mechanisms of action. There exists a scientific rationale to investigate the clinical efficacy of these two agents in combination. This manuscript reports the results of a toxicology study in rats, evaluating different doses of osilodrostat and pasireotide alone and in combination. Sixty male and 60 female rats were randomized into single-sex groups to receive daily doses of pasireotide (0.3 mg/kg/day, subcutaneously), osilodrostat (20 mg/kg/day, orally), osilodrostat/pasireotide in combination (low dose, 1.5/0.03 mg/kg/day; mid-dose, 5/0.1 mg/kg/day; or high dose, 20/0.3 mg/kg/day), or vehicle for 13 weeks. Mean body-weight gains from baseline to Week 13 were significantly lower in the pasireotide-alone and combined-treatment groups compared to controls, and were significantly higher in female rats receiving osilodrostat monotherapy. Osilodrostat and pasireotide monotherapies were associated with significant changes in the histology and mean weights of the pituitary and adrenal glands, liver, and ovary/oviduct. Osilodrostat alone was associated with adrenocortical hypertrophy and hepatocellular hypertrophy. In combination, osilodrostat/pasireotide did not exacerbate any target organ changes and ameliorated the liver and adrenal gland changes observed with monotherapy. Cmax and AUC0–24h of osilodrostat and pasireotide increased in an approximately dose-proportional manner.

In conclusion, the pasireotide and osilodrostat combination did not exacerbate changes in target organ weight or toxicity compared with either monotherapy, and had an acceptable safety profile; addition of pasireotide to the osilodrostat regimen may attenuate potential adrenal gland hyperactivation and hepatocellular hypertrophy, which are potential side effects of osilodrostat monotherapy.
The somatostatin class is a known class of small peptides comprising the naturally occurring somatostatin- 14 and analogues having somatostatin related activity, e.g. as disclosed by A.S. Dutta in Small Peptides, Vol.19, Elsevier (1993). By “somatostatin analogue” as used herein is meant any straight-chain or cyclic polypeptide having a structure based on that of the naturally occurring somatostatin- 14 wherein one or more amino acid units have been omitted and/or replaced by one or more other amino radical(s) and/or wherein one or more functional groups have been replaced by one or more other functional groups and/or one or more groups have been replaced by one or several other isosteric groups. In general, the term covers all modified derivatives of the native somatostatin- 14 which exhibit a somatostatin related activity, e.g. they bind to at least one of the five somatostatin receptor (SSTR), preferably in the nMolar range. Commonly known somatostatin analogs are octreotide, vapreotide, lanreotide, pasireotide.
Pasireotide, having the chemical structure as follow:
Pasireotide is called cyclo[{4-(NH2-C2H4-NH-CO-0-)Pro}-Phg-DTrp-Lys-Tyr(4-Bzl)- Phe], wherein Phg means -HN-CH(C6H5)-CO- and Bzl means benzyl, in free form, in salt or complex form or in protected form.
Cushing’s syndrome is a hormone disorder caused by high levels of Cortisol in the blood. This can be caused by taking glucocorticoid drugs, or by tumors that produce Cortisol or adrenocorticotropic hormone (ACTH) or CRH. Cushing’s disease refers to one specific cause of the syndrome: a tumor (adenoma) in the pituitary gland that produces large amounts of ACTH, which elevates Cortisol. It is the most common cause of Cushing’s syndrome, responsible for 70% of cases excluding glucocorticoid related cases. The significant decrease of Cortisol levels in Cushing’s disease patients on pasireotide support its potential use as a targeted treatment for Cushing’s disease (Colao et al. N Engl J Med 2012;366:32^12).
Compound A is potent inhibitor of the rate-limiting enzyme 1 1-beta-hydroxylase, the last step in the synthesis of Cortisol. WO 201 1/088188 suggests the potential use of compound A in treating a disease or disorder characterised by increased stress hormone levels and/or decreased androgen hormone levels, including the potential use of compound A in treating heart failure, cachexia, acute coronary syndrome, chronic stress syndrome, Cushing’s syndrome or metabolic syndrome.
Compound A, also called (R)-4-(6,7-Dihydro-5H-pyrrolo[l,2-c]imidazol-5-yl)-3-fluoro- benzonitrile, has formula (II).
Compound A can be synthesized or produced and characterized by methods as described in WO2007/024945.
PRODUCT PATENT
WO2007024945, hold protection in the EU states until August 2026, and expire in the US in March 2029 with US154 extension
PAPER
ACS Medicinal Chemistry Letters (2013), 4(12), 1203-1207.
http://pubs.acs.org/doi/abs/10.1021/ml400324c?source=chemport&journalCode=amclct
Discovery and in Vivo Evaluation of Potent Dual CYP11B2 (Aldosterone Synthase) and CYP11B1 Inhibitors

Aldosterone is a key signaling component of the renin-angiotensin-aldosterone system and as such has been shown to contribute to cardiovascular pathology such as hypertension and heart failure. Aldosterone synthase (CYP11B2) is responsible for the final three steps of aldosterone synthesis and thus is a viable therapeutic target. A series of imidazole derived inhibitors, including clinical candidate 7n, have been identified through design and structure–activity relationship studies both in vitro and in vivo. Compound 7n was also found to be a potent inhibitor of 11β-hydroxylase (CYP11B1), which is responsible for cortisol production. Inhibition of CYP11B1 is being evaluated in the clinic for potential treatment of hypercortisol diseases such as Cushing’s syndrome.
PATENT
silodrostat (LCI699; 4-[(5R)-6,7-dihydro-5H-pyrrolo[l,2-c]imidazol-5-yl]-3-fluoro-benzonitrile; CAS# 928134-65-0). Osilodrostat is a Ι Ι-β-hydroxylase inhibitor.
Osilodrostat is currently under investigation for the treatment of Cushing’s disease, primary aldosteronism, and hypertension. Osilodrostat has also shown promise in treating drug-resistant hypertension, essential hypertension, hypokalemia, hypertension, congestive heart failure, acute heart failure, heart failure, cachexia, acute coronary syndrome, chronic stress syndrome, Cushing’s syndrome, metabolic syndrome, hypercortisolemia, atrial fibrillation, renal failure, chronic renal failure, restenosis, sleep apnea, atherosclerosis, syndrome X, obesity, nephropathy, post-myocardial infarction, coronary heary disease, increased formation of collagen, cardiac or myocardiac fibrosis and/or remodeling following hypertension and endothelial dysfunction, Conn’s disease, cardiovascular diseases, renal dysfunction, liver diseases, cerebrovascular diseases, vascular diseases, retinopathy, neuropathy, insulinopathy, edema, endothelial dysfunction, baroreceptor dysfunction, migraine headaches, arrythmia, diastolic dysfunction, diastolic heart failure, impaired diastolic filling, systolic dysfunction, ischemia, hypertrophic cardiomyopathy, sudden cardia death, impaired arterial compliance, myocardial necrotic lesions, vascular damage, myocardial infarction, left ventricular hypertrophy, decreased ej ection fraction, cardiac lesions, vascular wall hypertrophy, endothelial thickening, fibrinoid, necrosis of coronary arteries, ectopic ACTH syndrome, change in adrenocortical mass, primary pigmented nodular adrenocortical disease (PPNAD), Carney complex (CNC), anorexia nervosa, chronic alcoholic poisoning, nicotine withdrawal syndrome, cocaine withdrawal syndrome, posttraumatic stress syndrome, cognitive impairment after a stroke or cortisol-induced mineral corticoid excess, ventricular arrythmia, estrogen-dependent disorders, gynecomastia, osteoporosis, prostate cancer, endometriosis, uterine fibroids, dysfunctional uterine bleeding, endometrial hyperplasia, polycyctic ovarian disease, infertility, fibrocystic breast disease, breast cancer, and fibrocystic mastopathy. WO 2013109514; WO 2007024945; and WO 2011064376.

Osilodrostat
Osilodrostat is likely subject to extensive CYP45o-mediated oxidative metabolism. These, as well as other metabolic transformations, occur in part through polymorphically-expressed enzymes, exacerbating interpatient variability. Additionally, some metabolites of osilodrostat derivatives may have undesirable side effects. In order to overcome its short half-life, the drug likely must be taken several times per day, which increases the probability of patient incompliance and discontinuance. Adverse effects associated with osilodrostat include fatigue, nausea, diarrhea, headache, hypokalemia, muscle spasms, vomiting, abdominal discomfort, abdominal pain, arthralgia, arthropod bite, dizziness, increased lipase, and pruritis.
Scheme I

EXAMPLE 1
(R)-4-(6,7-dihvdro-5H-pyrrolo[l,2-elimidazol-5-yl)-3-fluorobenzonitrile
(osilodrostat)

[00144] 4-(bromomethyl)-3-fluorobenzonitrile: 3-Fluoro-4-methylbenzonitrile (40 g, 296 mmol), NBS (63.2 g, 356 mmol) and benzoyl peroxide (3.6 g, 14.8 mmol) were taken up in carbon tetrachloride (490 mL) and refiuxed for 16 h. The mixture was allowed to cool to room temperature and filtered. The filtrate was concentrated and purified via flash column chromatography (0-5% EtOAc/hexanes) to give 4-(bromomethyl)-3-fluorobenzonitrile (35.4 g, 56%).

[00145] 2-(l-trityl-lH-imidazol-4-yl)acetic acid: Trityl chloride (40 g, 143.88 mmol, 1.2 equiv) was added to a suspension of (lH-imidazol-4-yl) acetic acid hydrochloride (20 g, 123.02 mmol, 1.0 equiv) in pyridine (200 mL). This was stirred at 50 °C for 16 h. Then the mixture was cooled and concentrated under vacuum and the crude product was purified by recrystallization from ethyl acetate (1000 ml) to afford 42 g (90%) of 2-[l-(triphenylmethyl)-lH-imidazol-4-yl] acetic acid as an off-white solid. LCMS (ESI): m/z = 369.2 [M+H]+
Step 2

2 step 2
2-( 1 -trityl- lH-imidazol-4-yl)ethanol : 2-(l-Trityl-lH-imidazol-4-yl) acetic acid (42 g, 114.00 mmol, 1.0 equiv) was suspended in THF (420 mL) and cooled to 0 °C. To this was added BH3 (1M in THF, 228.28 mL, 2.0 equiv). The clear solution obtained was stirred at 0 °C for 60 min, then warmed to room temperature until LCMS indicated completion of the reaction. The solution was cooled again to 0 °C and quenched carefully with water (300 mL). The resulting solution was extracted with ethyl acetate (3 x 100 mL) and the organic layers combined and dried over anhydrous Na2S04 and evaporated to give a sticky residue which was taken up in ethanolamine (800 mL) and heated to 90 °C for 2 h. The reaction was transferred to a separatory funnel, diluted with EtOAc (1 L) and washed with water (3 x 600 mL). The organic phase was dried over anhydrous Na2S04 and evaporated afford 35 g (87%) of 2-[l-(triphenylmethyl)-lH-imidazol-4-yl]ethanol as a white solid, which was used in the next step without further purification. LCMS (ESI) : m/z = 355.1 [M+H]+.
Step 3
![]()
3 step 3 4
4-(2-(tert-butyldimethylsilyloxy)ethyl)-l-trityl-lH-imidazole: 2-(l-Trityl-lH-imidazol-4-yl) ethanol (35 g, 98.75 mmol, 1.00 equiv) was dissolved in DCM (210 mL). To this was added imidazole (19.95 g, 293.05 mmol, 3.00 equiv) and tert-butyldimethylsilylchloride (22.40 g, 149.27 mmol, 1.50 equiv). The mixture was stirred at room temperature until LCMS indicated completion of the reaction. Then the resulting solution was diluted with 500 mL of DCM. The resulting mixture was washed with water (3 x 300 mL). The residue was purified by a silica gel column, eluted with ethyl
acetate/petroleum ether (1 :4) to afford 40 g (77%) of 4-[2-[(tert-butyldimethylsilyl)oxy]ethyl]-l-(triphenylmethyl)-lH-imidazole as a white solid. LCMS (ESI) : m/z = 469.1 [M+H]+.
Step 4

4-((5-(2-(tert-butyldimethylsilyloxy )ethylVlH-iniidazol-l -vnmethylV3-fluorobenzonitrile: 4-(2-((tert-Butyldimethylsilanyl)oxy)ethyl)-l rityl-lH-irnidazole (40 g, 85.34 mmol, 1.00 equiv) and 4-(Bromomethyl)-3-fluorobenzonitrile (27.38 g, 127.92 mmol, 1.50 equiv) obtained as a product of step 0, were dissolved in MeCN (480 mL) and DCM (80 mL), and stirred at room temperature for 48 h. Et2NH (80 mL) and MeOH (480 mL) were then added and the solution was warmed 80 °C for 3 h. The solution was evaporated to dryness and the residue was purified via flash column chromatography (EtOAc/hexanes 1 :5 to EtOAc) to afford 4-((5-(2-((tert-Butyldimethylsilanyl)oxy)ethyl)-lH-imidazol-l -yl)methyl)-3-fluorobenzonitrile (15 g, 50%). ¾ NMR (400 MHz, CDCh) δ: 7.67 (s, 1H), 7.43 (m, 2H), 6.98 (s, 1H), 6.88-6.79 (m, 1H), 5.34 (s, 2H), 3.79 (t, J= 8.0 Hz, 2H), 2.67 (t, J = 8.0 Hz, 2H), 0.88 (s, 9H), 0.02 (s, 6H). LCMS (ESI) : m/z = 360.1 [M+H]+.
Step 5

5 6
Methyl 2-(5-(2-(tert-butyldimethylsilyloxy)ethyl)-lH-imidazol-l -yl)-2-(4-cvano-2-fluorophenvDacetate: 4-((5-(2-((tert-Butyldimethylsilanyl)oxy)ethyl)-lH-imidazol-l -yl)methyl)-3-fluorobenzonitrile (15 g, 41.72 mmol, 1.00 equiv) was dissolved in anhydrous THF (150 mL) and stirred at -78 °C, then a THF solution of LiHMDS (75 mL, 1.80 equiv, 1.0 M) was added dropwise over 15 min. After 30 min, methyl cyanoformate (4.3 g, 45.50 mmol, 1.10 equiv) was added dropwise over 10 min and the solution was stirred at -78 °C for 2 h. The excess LiHMDS was quenched with aqueous saturated NH4CI and the mixture was allowed to warm to room temperature. The mixture was then diluted with EtOAc and washed
with aqueous saturated NH4CI (200 mL). The organic layers was dried over anhydrous Na2S04 and evaporated. The crude residue was purified via flash column chromatography (EtOAc/PE 3: 10 to EtOAc) to give methyl 2-(5-(2-((tert-butyldimethylsilanyl)oxy)ethyl)-lH-imidazol-l-yl)-2-(4-cyano-2-fluorophenyl) acetate (15 g, 86%) as a light yellow solid.
¾ NMR (400 MHz, CDCL3) δ: 7.66 (s, 1H), 7.54-7.43 (m, 2H), 7.15 (t, J= 8.0 Hz 1H), 6.93 (s, 1H), 6.47 (s, 1H), 3.88-3.74 (m, 5H), 2.81-2.62 (m, 2H), 0.89 (s, 9H), 0.05 (s, 6H) . LCMS (ESI) : m/z = 418.2 [M+H]+.
Step 6

Methyl 2-(4-cvano-2-fluorophenyl)-2-(5-(2-hvdroxyethyl)-lH-imidazol-l-yl) acetate: Methyl 2-(5-(2-((tert-butyldimethylsilanyl)oxy)ethyl)-lH-imidazol-l-yl)-2-(4-cyano-2-fiuorophenyl)acetate (15 g, 35.92 mmol, 1.00 equiv) was added to a solution of HCl in 1,4-dioxane (89 mL, 4.0 M, 359.2 mmol) at 0 °C and the mixture was allowed to warm to room temperature and stirred for 2 h. The solution was concentrated to dryness to give the crude alcohol, methyl 2-(4-cyano-2-fluorophenyl )-2-(5-(2 -hydroxy ethyl)-lH-imidazol-l-yl)acetate (10 g, 92%), which was used without further purification. LCMS: m/z = 304.0 [M+H]+.
Step 7

7 8
Methyl 2-(4-cvano-2-fluorophenyl)-2-(5-(2-(methylsulfonyloxy)ethyl)-lH-imidazol-l-yl) acetate: The crude methyl 2-(4-cyano-2-fluorophenyl )-2-(5-(2-hydroxyethyl)-lH-imidazol-l-yl)acetate (10 g, 32.97 mmol, 1.00 equiv) was dissolved in DCM (200 mL) and stirred at 0 °C, then Et3N (20 g, 197.65 mmol, 6.00 equiv) and
methanesulfonyl chloride (4.52 g, 39.67 mmol, 1.20 equiv) were added. After completion of the reaction, the solution was diluted with DCM and washed with aqueous saturated
NaHCC . The organic layer was dried over anhydrous Na2S04, filtered and evaporated to give the crude methyl 2-(4-cyano-2-fluorophenyl)-2-(5-(2-((methylsulfonyl)oxy)ethyl)-lH-imidazol-l-yl)acetate (11.43 g, 91%), which was used in the next step without further purification. LCMS (ESI) : m/z = 382.0 [M+H]+.
Step 8

Methyl 5-(4-cvano-2-fluorophenyl)-6.7-dihvdro-5H-pyrrolo[1.2-elimidazole-5-carboxylate: The crude methyl 2-(4-cyano-2 -fluorophenyl )-2-(5-(2- ((methylsulfonyl)oxy)ethyl)-lH-imidazol-l-yl)acetate (11.43 g, 29.97 mmol, 1.00 equiv) was dissolved in MeCN (550 mL) and then K2CO3 (12.44 g, 90.01 mmol, 3.00 equiv), Nal (13.50 g, 90.00 mmol, 3.00 equiv) and Et3N (9.09 g, 89.83 mmol, 3.00 equiv) were added. The reaction was stirred at 80 °C for 42 h. The mixture was filtered. The solids were washed with DCM. The filtrate was concentrated and purified by flash column chromatography (EtOAc) to give methyl 5-(4-cyano-2-fluorophenyl)-6,7-dihydro-5H-pyrrolo[l,2-c]imidazole-5-carboxylate (4.2 g, 49% in 3 steps).
[00153] ¾ NMR (400 MHz, CDCb) δ: 7.61 (s, 1H), 7.47-7.47 (m, 2H), 6.88 (s, 1H), 6.79-6.75 (m, 1H), 4.17-4.12 (m, 1H), 3.87 (s, 3H), 3.78-3.70 (m, 1H), 3.08-3.02 (m, 1H), 2.84-2.71 (m, 2H). LCMS (ESI) : m/z = 286.0 [M+H]+.
Step 9

10
4-(6.7-dihvdro-5H-pyrrolo[1.2-elimidazol-5-yl)-3-fluorobenzonitrile: To a 40-mL sealed tube, was placed methyl 5-(4-cyano-2-fluorophenyl)-5H,6H,7H-pyrrolo[l,2-c]imidazole-5-carboxylate (1 g, 3.51 mmol, 1.00 equiv), DMSO (10 mL), water (5 mL). The final reaction mixture was irradiated with microwave radiation for 40 min at 140 °C. The resulting solution was diluted with 100 mL of EtOAc. The resulting mixture was washed with (3 x 20 mL) brine, dried over anhydrous Na2S04, filtered and concentrated. The residue was purified by a silica gel column, eluted with ethyl acetate/petroleum ether (4: 1) to afford 420 mg (44%) of 5-(4-cyano-2-fluorophenyl)-5H,6H,7H-pyrrolo[l,2-c]irnidazole-5-carboxylic acid as a light yellow solid.
¾ NMR (400 MHz, CDCL3) δ: 7.55-7.28 (m, 3H), 6.90-6.85 (m, 2H), 5.74-5.71 (m, 1H), 3.25-3.15 (m, 1H), 3.02-2.92 (m, 2H), 2.58-2.50 (m, 1H). LCMS (ESI) : m/z = 228.2 [M+H]+.
Step 10

10
(R)-4-(6 -dihvdro-5H-pyrrolo[1.2-elirnidazol-5-yl)-3-fluorobenzonitrile:
Resolution of the enantiomers of the title compound (300 mg) was performed by chiral HPLC: Column, Chiralpak IA2, 2*25cm, 20um; mobile phase, Phase A: Hex (50%, 0.1% DEA), Phase B: EtOH (50%) ; Detector, UV 254/220 nm to afford the (S)-enantiomer (RT = 17 min) and the (R)-enantiomer (97.6 mg, desired compound) (RT = 21 min).
¾ NMR (400 MHz, DMSO-<4) δ: 7.98-7.95 (m, 1H), 7.70-7.69 (m, 1H), 7.50 (s, 1H), 6.87 (t, J= 8.0 Hz, 1H), 6.70 (s, 1H), 5.79-5.76 (m, 1H), 3.15-3.06 (m, 1H), 2.92-2.74 (m, 2H), 2.48-2.43 (m, 1H). LCMS (ESI) : m/z = 228.1 [M+H]+.
PATENT
WO2013/153129
https://www.google.com/patents/WO2013153129A1?cl=en
PATENT
WO2007/024945
http://www.google.co.in/patents/WO2007024945A1?cl=en
PATENT
Aspect (iii) of the present invention relates to phosphate salt or nitrate salt of 4-(R)-(6,7-dihydro-5H-pyrrolo[1,2-c]imidazol-5-yl)-3-fluoro-benzonitrile according to Formula (III)
abbreviated as ‘{drug3}’. In particular, the present invention relates to crystalline form of phosphate salt of 4-(R)-(6,7-dihydro-5H-pyrrolo[1,2-c]imidazol-5-yl)-3-fluoro-benzonitrile, abbreviated as ‘{drug3a}’; to crystalline Form A of phosphate salt of 4-(R)-(6,7-dihydro-5H-pyrrolo[1,2-c]imidazol-5-yl)-3-fluoro-benzonitrile, abbreviated as ‘{drug3b}’; to crystalline Form B of phosphate salt of 4-(R)-(6,7-dihydro-5H-pyrrolo[1,2-c]imidazol-5-yl)-3-fluoro-benzonitrile, abbreviated as ‘{drug3c}’; to crystalline Form C of phosphate salt of 4-(R)-(6,7-dihydro-5H-pyrrolo[1,2-c]imidazol-5-yl)-3-fluoro-benzonitrile, abbreviated as ‘{drug3d}’; to crystalline Form D of phosphate salt of 4-(R)-(6,7-dihydro-5H-pyrrolo[1,2-c]imidazol-5-yl)-3-fluoro-benzonitrile, abbreviated as ‘{drug3e}’; to crystalline Form E of phosphate salt of 4-(R)-(6,7-dihydro-5H-pyrrolo[1,2-c]imidazol-5-yl)-3-fluoro-benzonitrile, abbreviated as ‘{drug3f}’; to crystalline Form F of phosphate salt of 4-(R)-(6,7-dihydro-5H-pyrrolo[1,2-c]imidazol-5-yl)-3-fluoro-benzonitrile, abbreviated as ‘{drug3g}’; to crystalline Form G of phosphate salt of 4-(R)-(6,7-dihydro-5H-pyrrolo[1,2-c]imidazol-5-yl)-3-fluoro-benzonitrile, abbreviated as ‘{drug3h}’; to crystalline Form H of phosphate salt of 4-(R)-(6,7-dihydro-5H-pyrrolo[1,2-c]imidazol-5-yl)-3-fluoro-benzonitrile, abbreviated as ‘{drug3i}’; and to crystalline form of nitrate salt of 4-(R)-(6,7-dihydro-5H-pyrrolo[1,2-c]imidazol-5-yl)-3-fluoro-benzonitrile, abbreviated as ‘{drug3j}’. {drug3a}, {drug3b}, {drug3c}, {drug3d}, {drug3e}, {drug3f}, {drug3g}, {drug3h}, {drug3i}, and {drug3j} are specific forms falling within the definition of {drug3}. Aspect (iii) of the invention is separate from aspects (i), (ii), (iv), (v), (vi), (vii), and (viii) of the invention. Thus, all embodiments of {drug3a}, {drug3b}, {drug3c}, {drug3d}, {drug3e}, {drug3f}, {drug3g}, {drug3h}, {drug3i}, and {drug3j}, respectively, are only related to {drug3}, but neither to {drug1}, nor to {drug2}, nor to {drug4}, nor to {drug5}, nor to {drug6}, nor to {drug7}, nor to {drug8}.
PAPER
Osilodrostat (LCI699), a potent 11β-hydroxylase inhibitor, administered in combination with the multireceptor-targeted somatostatin analog pasireotide: A 13-week study in rats
- a Preclinical Safety, Novartis Institutes for BioMedical Research, East Hanover, NJ, USA
- b Drug Metabolism and Pharmacokinetics, Novartis Institutes for BioMedical Research, East Hanover, NJ, USA
- c Novartis Oncology Development, Basel, Switzerland
doi:10.1016/j.taap.2015.05.004, http://www.sciencedirect.com/science/article/pii/S0041008X15001684
CLIPS
| WO2011088188A1 * | Jan 13, 2011 | Jul 21, 2011 | Novartis Ag | Use of an adrenal hormone-modifying agent |
| Reference | ||
|---|---|---|
| 1 | * | BOSCARO M ET AL: “Treatment of Pituitary-Dependent Cushing’s Disease with the Multireceptor Ligand Somatostatin Analog Pasireotide (SOM230): A Multicenter, Phase II Trial“, JOURNAL OF CLINICAL ENDOCRINOLOGY & METABOLISM, vol. 94, no. 1, January 2009 (2009-01), pages 115-122, XP002698507, ISSN: 0021-972X |
References
- ^ Jump up to:a b c http://adisinsight.springer.com/drugs/800026342
- ^ Jump up to:a b Fleseriu M, Castinetti F (2016). “Updates on the role of adrenal steroidogenesis inhibitors in Cushing’s syndrome: a focus on novel therapies”. Pituitary. 19 (6): 643–653. doi:10.1007/s11102-016-0742-1. PMC 5080363
 . PMID 27600150.
. PMID 27600150.
External links
REFERENCES
1: Guelho D, Grossman AB. Emerging drugs for Cushing’s disease. Expert Opin Emerg Drugs. 2015 Sep;20(3):463-78. doi: 10.1517/14728214.2015.1047762. Epub 2015 Jun 2. PubMed PMID: 26021183.
2: Li L, Vashisht K, Boisclair J, Li W, Lin TH, Schmid HA, Kluwe W, Schoenfeld H, Hoffmann P. Osilodrostat (LCI699), a potent 11β-hydroxylase inhibitor, administered in combination with the multireceptor-targeted somatostatin analog pasireotide: A 13-week study in rats. Toxicol Appl Pharmacol. 2015 Aug 1;286(3):224-33. doi: 10.1016/j.taap.2015.05.004. Epub 2015 May 14. PubMed PMID: 25981165.
3: Papillon JP, Adams CM, Hu QY, Lou C, Singh AK, Zhang C, Carvalho J, Rajan S, Amaral A, Beil ME, Fu F, Gangl E, Hu CW, Jeng AY, LaSala D, Liang G, Logman M, Maniara WM, Rigel DF, Smith SA, Ksander GM. Structure-Activity Relationships, Pharmacokinetics, and in Vivo Activity of CYP11B2 and CYP11B1 Inhibitors. J Med Chem. 2015 Jun 11;58(11):4749-70. doi: 10.1021/acs.jmedchem.5b00407. Epub 2015 May 21. PubMed PMID: 25953419.
4: Fleseriu M. Medical treatment of Cushing disease: new targets, new hope. Endocrinol Metab Clin North Am. 2015 Mar;44(1):51-70. doi: 10.1016/j.ecl.2014.10.006. Epub 2014 Nov 4. Review. PubMed PMID: 25732642.
5: Wang HZ, Tian JB, Yang KH. Efficacy and safety of LCI699 for hypertension: a meta-analysis of randomized controlled trials and systematic review. Eur Rev Med Pharmacol Sci. 2015;19(2):296-304. Review. PubMed PMID: 25683946.
6: Daniel E, Newell-Price JD. Therapy of endocrine disease: steroidogenesis enzyme inhibitors in Cushing’s syndrome. Eur J Endocrinol. 2015 Jun;172(6):R263-80. doi: 10.1530/EJE-14-1014. Epub 2015 Jan 30. Review. PubMed PMID: 25637072.
7: Fleseriu M, Petersenn S. Medical therapy for Cushing’s disease: adrenal steroidogenesis inhibitors and glucocorticoid receptor blockers. Pituitary. 2015 Apr;18(2):245-52. doi: 10.1007/s11102-014-0627-0. PubMed PMID: 25560275.
8: Ménard J, Rigel DF, Watson C, Jeng AY, Fu F, Beil M, Liu J, Chen W, Hu CW, Leung-Chu J, LaSala D, Liang G, Rebello S, Zhang Y, Dole WP. Aldosterone synthase inhibition: cardiorenal protection in animal disease models and translation of hormonal effects to human subjects. J Transl Med. 2014 Dec 10;12:340. doi: 10.1186/s12967-014-0340-9. PubMed PMID: 25491597; PubMed Central PMCID: PMC4301837.
9: Oki Y. Medical management of functioning pituitary adenoma: an update. Neurol Med Chir (Tokyo). 2014;54(12):958-65. Epub 2014 Nov 29. PubMed PMID: 25446388.
10: Cai TQ, Stribling S, Tong X, Xu L, Wisniewski T, Fontenot JA, Struthers M, Akinsanya KO. Rhesus monkey model for concurrent analyses of in vivo selectivity, pharmacokinetics and pharmacodynamics of aldosterone synthase inhibitors. J Pharmacol Toxicol Methods. 2015 Jan-Feb;71:137-46. doi: 10.1016/j.vascn.2014.09.011. Epub 2014 Oct 7. PubMed PMID: 25304940.
11: Lother A, Moser M, Bode C, Feldman RD, Hein L. Mineralocorticoids in the heart and vasculature: new insights for old hormones. Annu Rev Pharmacol Toxicol. 2015;55:289-312. doi: 10.1146/annurev-pharmtox-010814-124302. Epub 2014 Sep 10. Review. PubMed PMID: 25251996.
12: Cuevas-Ramos D, Fleseriu M. Treatment of Cushing’s disease: a mechanistic update. J Endocrinol. 2014 Nov;223(2):R19-39. doi: 10.1530/JOE-14-0300. Epub 2014 Aug 18. Review. PubMed PMID: 25134660.
13: Yin L, Hu Q, Emmerich J, Lo MM, Metzger E, Ali A, Hartmann RW. Novel pyridyl- or isoquinolinyl-substituted indolines and indoles as potent and selective aldosterone synthase inhibitors. J Med Chem. 2014 Jun 26;57(12):5179-89. doi: 10.1021/jm500140c. Epub 2014 Jun 5. PubMed PMID: 24899257.
14: Li W, Luo S, Rebello S, Flarakos J, Tse FL. A semi-automated LC-MS/MS method for the determination of LCI699, a steroid 11β-hydroxylase inhibitor, in human plasma. J Chromatogr B Analyt Technol Biomed Life Sci. 2014 Jun 1;960:182-93. doi: 10.1016/j.jchromb.2014.04.012. Epub 2014 Apr 30. PubMed PMID: 24814004.
15: Trainer PJ. Next generation medical therapy for Cushing’s syndrome–can we measure a benefit? J Clin Endocrinol Metab. 2014 Apr;99(4):1157-60. doi: 10.1210/jc.2014-1054. PubMed PMID: 24702012.
16: Bertagna X, Pivonello R, Fleseriu M, Zhang Y, Robinson P, Taylor A, Watson CE, Maldonado M, Hamrahian AH, Boscaro M, Biller BM. LCI699, a potent 11β-hydroxylase inhibitor, normalizes urinary cortisol in patients with Cushing’s disease: results from a multicenter, proof-of-concept study. J Clin Endocrinol Metab. 2014 Apr;99(4):1375-83. doi: 10.1210/jc.2013-2117. Epub 2013 Dec 11. PubMed PMID: 24423285.
17: Oki Y. Medical management of functioning pituitary adenoma: an update. Neurol Med Chir (Tokyo). 2014;54 Suppl 3:958-65. PubMed PMID: 26236804.
18: Schumacher CD, Steele RE, Brunner HR. Aldosterone synthase inhibition for the treatment of hypertension and the derived mechanistic requirements for a new therapeutic strategy. J Hypertens. 2013 Oct;31(10):2085-93. doi: 10.1097/HJH.0b013e328363570c. PubMed PMID: 24107737; PubMed Central PMCID: PMC3771574.
19: Brown NJ. Contribution of aldosterone to cardiovascular and renal inflammation and fibrosis. Nat Rev Nephrol. 2013 Aug;9(8):459-69. doi: 10.1038/nrneph.2013.110. Epub 2013 Jun 18. Review. PubMed PMID: 23774812; PubMed Central PMCID: PMC3922409.
20: van der Pas R, de Herder WW, Hofland LJ, Feelders RA. Recent developments in drug therapy for Cushing’s disease. Drugs. 2013 Jun;73(9):907-18. doi: 10.1007/s40265-013-0067-6. Review. PubMed PMID: 23737437.
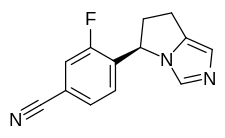 |
|
| Clinical data | |
|---|---|
| Synonyms | LCI-699 |
| Routes of administration |
Oral |
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C13H10FN3 |
| Molar mass | 227.24 g·mol−1 |
| 3D model (JSmol) | |
///////OSILODROSTAT, Novartis , osilodrostat, an inhibitor of aldosterone synthase and aromatase, treating Cushing’s disease, July 2016, phase 3 clinical development, LCI 699, 928134-65-0, 1315449-72-9, PHASE 3, LCI 699NX, LCI 699AZA, CYP11B1 CYP11B2
c1cc(c(cc1C#N)F)[C@H]2CCc3n2cnc3.OP(=O)(O)O
N#CC1=CC=C([C@H]2CCC3=CN=CN32)C(F)=C1
Novartis, Torrent drug for diabetes, NVP-LBX192, LBX-192

(R)-3-Cyclopentyl-N-(5-methoxy-thiazolo[5,4-b]pyridin-2-yl)-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionamide
3-Cyclopentyl-N-(5-methoxy-thiazolo[5,4-b]pyridin-2-yl)-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionamide
(3-Cyclopentyl-N-(5-methoxy-thiazolo[5,4-b]pyridin-2-yl)-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionamide)
(R)-3-Cyclopentyl-N-(5-methoxy-thiazolo[5,4-b]pyridin-2-yl)-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionamide
cas 866772-52-3
NVP-LBX192
LBX-192
R(−) 3-cyclopentyl-N-(5-methoxy-thiazolo[5,4-b]pyridin-2-yl)-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionamide
R(−)17c BELOW

| Inventors | Gregory Raymond Bebernitz, Ramesh Chandra Gupta, Vikrant Vijaykumar Jagtap, Appaji Baburao Mandhare, Davinder Tuli, |
| Original Assignee | Novartis Ag
|
| Molecular Formula: | C26H33N5O4S2 |
|---|---|
| Molecular Weight: | 543.70132 g/mol |


LBX192, also known as NVP-LBX192, is a Liver Targeted Glucokinase Activator. LBX192 activated the GK enzyme in vitro at low nM concentrations and significantly reduced glucose levels during an oral glucose tolerance test in normal as well as diabetic mice. A GK activator has the promise of potentially affecting both the beta-cell of the pancreas, by improving glucose sensitive insulin secretion, as well as the liver, by reducing uncontrolled glucose output and restoring post prandial glucose uptake and storage as glycogen.
SYNTHESIS BY WORLDDRUGTRACKER
54 Discovery and Evaluation of NVP-LBX192, a Liver Targeted Glucokinase Activator
https://acs.confex.com/acs/nerm09/webprogram/Paper75087.html
Sulfonamide-Thiazolpyridine Derivatives, Glucokinase Activators, Treatment Of Type 2 Diabetes
2009 52 (19) 6142 – 6152
Investigation of functionally liver selective glucokinase activators for the treatment of type 2 diabetes
Journal of Medicinal Chemistry
Bebernitz GR, Beaulieu V, Dale BA, Deacon R, Duttaroy A, Gao JP, Grondine MS, Gupta RC, Kakmak M, Kavana M, Kirman LC, Liang JS, Maniara WM, Munshi S, Nadkarni SS, Schuster HF, Stams T, Denny IS, Taslimi PM, Vash B, Caplan SL
2010 240th (August 22) Medi-198
Glucokinase activators with improved physicochemicalproperties and off target effects
American Chemical Society National Meeting and Exposition
Kirman LC, Schuster HF, Grondine MS et al
2010 240th (August 22) Medi-197
Investigation of functionally liver selective glucokinase activators
American Chemical Society National Meeting and Exposition
Schuster HF, Kirman LC, Bebernitz GC et al
PATENT
http://www.google.com/patents/US7750020
EXAMPLE 1 3-Cyclopentyl-N-(5-methoxy-thiazolo[5,4-b]pyridin-2-yl)-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionamide
A. Phenylacetic Acid Ethyl Ester
A solution of phenylacetic acid (50 g, 0.36 mol) in ethanol (150 mL) is treated with catalytic amount of sulfuric acid (4 mL). The reaction mixture is refluxed for 4 h. The reaction is then concentrated in vacuo. The residue is dissolved in diethyl ether (300 mL) and washed with saturated aqueous sodium bicarbonate solution (2×50 mL) and water (1×100 mL). The organic layer dried over sodium sulfate filtered and concentrated in vacuo to give phenylacetic acid ethyl ester as a colorless oil: 1H NMR (400 MHz, CDCl3) δ 1.2 (t, J=7.2, 3H), 3.6 (s, 2H), 4.1 (q, J=7.2, 2H), 7.3 (m, 5H); MS 165 [M+1]+.
B. (4-Chlorosulfonyl-phenyl)-acetic acid ethyl ester
To a cooled chlorosulfonic acid (83.83 g, 48 mL, 0.71 mol) under nitrogen is added the title A compound, phenylacetic acid ethyl ester (59 g, 0.35 mol) over a period of 1 h. Reaction temperature is brought to RT (28° C.), then heated to 70° C., maintaining it at this temperature for 1 h while stirring. Reaction is cooled to RT and poured over saturated aqueous sodium chloride solution (200 mL) followed by extraction with DCM (2×200 mL). The organic layer is washed with water (5×100 mL), followed by saturated aqueous sodium chloride solution (1×150 mL). The organic layer dried over sodium sulfate, filtered and concentrated in vacuo to give crude (4-chlorosulfonyl-phenyl)acetic acid ethyl ester. Further column chromatography over silica gel (60-120 mesh), using 100% hexane afforded pure (4-chlorosulfonyl-phenyl)-acetic acid ethyl ester as a colorless oil.
C. [4-(4-Methyl-piperazine-1-sulfonyl)-phenyl]-acetic acid ethyl ester
A solution of N-methylpiperazine (9.23 g, 10.21 ml, 0.092 mol), DIEA (13 g, 17.4 mL, 0.10 mol) and DCM 80 mL is cooled to 0° C., and to this is added a solution of the title B compound, (4-chlorosulfonyl-phenyl)-acetic acid ethyl ester (22 g, 0.083 mol) in 50 mL of DCM within 30 min. Reaction mixture stirred at 0° C. for 2 h, and the reaction mixture is washed with water (100 mL), followed by 0.1 N aqueous hydrochloric acid solution (1×200 mL). The organic layer dried over sodium sulfate, filtered and concentrated under vacuo to give crude [4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-acetic acid ethyl ester. Column chromatography over silicagel (60-120 mesh), using ethyl acetate afforded pure [4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-acetic acid ethyl ester as white crystalline solid: 1H NMR (400 MHz, CDCl3) δ 1.3 (t, J=7.4, 3H), 2.3 (s, 3H), 2.5 (m, 4H), 3.0 (br s, 4H), 3.7 (s, 2H), 4.2 (q, J=7.4, 2H), 7.4 (d, J=8.3, 2H), 7.7 (d, J=7.3, 2H); MS 327 [M+1]+.
D. 3-Cyclopentyl-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionic acid ethyl ester
A solution of the title C compound, [4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-acetic acid ethyl ester (15 g, 0.046 mol) in a mixture of THF (60 mL) and DMTP (10 mL) is cooled to −78° C. under nitrogen. The resulting solution is stirred at −78° C. for 45 min and to this is added LDA (25.6 mL, 6.40 g, 0.059 mol, 25% solution in THF/Hexane). A solution of iodomethylcyclopentane (11.60 g, 0.055 mol) in a mixture of DMTP (12 mL) and THF (20 mL) is added over a period of 15 min at −78° C. and reaction mixture stirred at −78° C. for 3 h further, followed by stirring at 25° C. for 12 h. The reaction mixture is then quenched by the dropwise addition of saturated aqueous ammonium chloride solution (50 mL) and is concentrated in vacuo. The residue is diluted with water (50 mL) and extracted with ethyl acetate (3×100 mL). The organic solution is washed with a saturated aqueous sodium chloride (2×150 mL), dried over sodium sulfate, filtered and concentrated in vacuo. Column chromatography over silica gel (60-120 mesh), using 50% ethyl acetate in hexane as an eluent to afford 3-cyclopentyl-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionic acid ethyl ester as a white solid: 1H NMR (400 MHz, CDCl3) δ 0.9-2.1 (m, 11H), 1.2 (t, J=7.1, 3H), 2.3 (s, 3H), 2.5 (br s, 4H), 3.0 (br s, 4H), 3.6 (m, 1H), 4.1 (q, J=7.1, 2H), 7.5 (d, J=8.3, 2H), 7.7 (d, J=8.3, 2H); MS 409 [M+1]+.
E. 3-Cyclopentyl-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionic acid
A solution of the title D compound, 3-cyclopentyl-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionic acid ethyl ester (14 g, 0.034 mol) in methanol:water (30 mL:10 mL) and sodium hydroxide (4.11 g, 0.10 mol) is stirred at 60° C. for 8 h in an oil bath. The methanol is then removed in vacuo at 45-50° C. The residue is diluted with water (25 mL) and extracted with ether (1×40 mL). The aqueous layer is acidified to pH 5 with 3 N aqueous hydrochloric acid solution. The precipitated solid is collected by vacuum filtration, washed with water (20 mL), followed by isopropyl alcohol (20 mL). Finally, solid cake is washed with 100 mL of hexane and dried under vacuum at 40° C. for 6 h to give 3-cyclopentyl-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionic acid as a white solid: 1H NMR (400 MHz, CDCl3) δ 1.1-2.0 (m, 11H), 2.4 (s, 3H), 2.7 (br s, 4H), 3.1 (br s, 4H), 3.6 (m, 1H), 7.5 (d, J=8.3, 2H), 7.6 (d, J=8.3, 2H); MS 381 [M+l]+.
F. 5-Methoxy-thiazolo[5,4-b]pyridin-2-ylamine
A solution of 6-methoxy-pyridin-3-ylamine (5.0 g, 0.0403 mol) in 10 mL of acetic acid is added slowly to a solution of potassium thiocyanate (20 g, 0.205 mol) in 100 mL of acetic acid at 0° C. followed by a solution of bromine (2.5 mL, 0.0488 mol) in 5 mL of acetic acid. The reaction is stirred for 2 h at 0° C. and then allowed to warm to RT. The resulting solid is collected by filtration and washed with acetic acid, then partitioned between ethyl acetate and saturated aqueous sodium bicarbonate. The insoluble material is removed by filtration and the organic layer is evaporated and dried to afford 5-methoxy-thiazolo[5,4-b]pyridin-2-ylamine as a tan solid.
G. 3-Cyclopentyl-N-(5-methoxy-thiazolo[5,4-b]pyridin-2-yl)-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionamide
A solution of the title E compound, 3-cyclopentyl-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionic acid (5 g, 0.013 mol) in DCM (250 mL) is cooled to 0° C. and then charged HOBt hydrate (2.66 g, 0.019 mol), followed by EDCI hydrochloride (6 g, 0.031 mol). The reaction mixture is stirred at 0° C. for 5 h. After that the solution of the title F compound, 5-methoxy-thiazolo[5,4-b]pyridin-2-ylamine (2.36 g, 0.013 mol) and D1EA (8 mL, 0.046 mol) in a mixture of DCM (60 mL) and DMF (20 mL) is added dropwise over 30 min. Reaction temperature is maintained at 0° C. for 3 h, then at RT (28° C.) for 3 days. Reaction is diluted with (60 mL) of water and the organic layer is separated and washed with saturated sodium bicarbonate solution (2×50 mL) followed by water washing (2×50 mL) and saturated sodium chloride aqueous solution (1×150 mL). Finally the organic layer is dried over sodium sulfate, filtered, and evaporated under vacuo. The crude product is purified using column chromatography over silica gel (60-120 mesh), using 40% ethyl acetate in hexane as an eluent to afford 3-cyclopentyl-N-(5-methoxy-thiazolo[5,4-b]pyridin-2-yl)-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionamide as a white solid: 1H NMR (400 MHz, CDCl3) δ 0.9-2.1 (m, 11H), 2.2 (s, 3H), 2.5 (br s, 4H), 3.1 (br s, 4H), 3.7 (m, 1H), 4.0 (s, 3H), 6.8 (d, J=8.8, 1H), 7.5 (d, J=8.3, 2H), 7.7 (d, J=8.3, 2H), 7.8 (d, J=8.8, 1H), 8.6 (s, 1H); MS 617 [M+1]+.
H. 3-Cyclopentyl-N-(5-methoxy-thiazolo[5,4-b]pyridin-2-yl)-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionamide dihydrochloride
The title G compound, 3-cyclopentyl-2-(4-methyl piperazinyl sulfonyl)phenyl-N-(5-methoxy-thiazolo[5,4-b]pyridin-2-yl)propionamide (2.8 g, 0.0051 mol) is added to a cooled solution of 10% hydrochloric acid in isopropanol (3.75 mL). The reaction mixture is stirred at 0° C. for 1 h and then at RT for 2 h. The solid is separated, triturated with 10 mL of isopropanol and collected by vacuum filtration and washed with 50 mL of hexane. The solid is dried at 70° C. for 48 h to afford 3-cyclopentyl-N-(5-methoxy-thiazolo[5,4-b]pyridin-2-yl)-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionamide dihydrochloride as an off white solid.
EXAMPLE 2 (R)-3-Cyclopentyl-N-(5-methoxy-thiazolo[5,4-b]pyridin-2-yl)-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionamide
The title compound is obtained analogously to Example 1 by employing the following additional resolution step:
The racemic title E compound of Example 1,3-cyclopentyl-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionic acid (10 g, 0.026 mol) in 1,4-dioxane (500 mL) is treated in a three necked 1 liter flask, equipped with heating mantle, water condenser, calcium chloride guard tube and mechanical stirrer with 3.18 g (0.026 mol) of (R)-(+)-1-phenylethylamine. This reaction mixture is then refluxed at 100° C. for 1 h. The clear reaction solution is cooled to RT (27° C.) and stirred for 10 h. The crystallized salt is collected by filtration under vacuum, washed with 5 mL of hexane and dried under vacuum to afford salt A.
The salt A is dissolved in 1,4-dioxane (500 mL) and heated at 100° C. for 1 h. The clear reaction solution is cooled to RT (27° C.) and stirred for 10 h. The crystallized product is collected by filtration under vacuum, washed with 50 mL of hexane, and dried under vacuum to afford salt B.
The salt B is dissolved in 1,4-dioxane (290 mL) and heated at 100° C. for 1 h. The clear reaction solution is cooled to RT (27° C.) and stirred for 10 h. The crystallized product is collected by filtration under vacuum, washed with 30 mL of hexane, and dried under vacuum to afford salt C.
The salt C is dissolved in 1,4-dioxane (100 mL) and heated at 100° C. for 1 h. The clear reaction solution is cooled to RT (27° C.) and stirred for 10 h. The crystallized product is collected by filtration under vacuum, washed with 30 ml of hexane, and dried under vacuum to afford salt D.
The salt D is treated with aqueous hydrochloric acid solution (20 mL, 1 mL of concentrated hydrochloric acid diluted with 100 mL of water) and stirred for 5 min. The white solid precipitates out and is collected by vacuum filtration, washed with 10 mL of cold water, 5 mL of isopropanol and 20 mL of hexane, and dried under vacuum to yield the hydrochloride salt of (R)-(−)-3-cyclopentyl-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionic acid, salt E.
The salt E is neutralized by stirring with aqueous sodium bicarbonate solution (10 mL, 1 g of sodium bicarbonate dissolved in 120 mL of water) for 5 min. The precipitated solid is collected by filtration, washed with 10 mL of cold water, 100 mL of hexane, and dried to afford (R)-(−)-3-cyclopentyl-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionic acid: m.p. 202.2-203.4° C.
Alternatively, the title compound may be obtained by the resolution of the racemic title compound of Example 1 using the following preparative chiral HPLC method:
- Column: Chiralcel OD-R (250×20 mm) Diacel make, Japan;
- Solvent A: water:methanol:acetonitrile (10:80:10 v/v/v);
- Solvent B: water:methanol:acetonitrile (05:90:05 v/v/v);
- Using gradient elution: gradient program (time, min/% B): 0/0, 20/0, 50/100, 55/0, 70/0;
- Flow rate: 6.0 mL/min; and
- Detection: by UV at 305 nm.
EXAMPLE 3 (S)-3-Cyclopentyl-N-(5-methoxy-thiazolo[5,4-b]pyridin-2-yl)-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionamide
The title compound is prepared analogously to Example 2.
J MED CHEM 2009, 52, 6142-52
Investigation of Functionally Liver Selective Glucokinase Activators for the Treatment of Type 2 Diabetes
http://pubs.acs.org/doi/abs/10.1021/jm900839k

Type 2 diabetes is a polygenic disease which afflicts nearly 200 million people worldwide and is expected to increase to near epidemic levels over the next 10−15 years. Glucokinase (GK) activators are currently under investigation by a number of pharmaceutical companies with only a few reaching early clinical evaluation. A GK activator has the promise of potentially affecting both the β-cells of the pancreas, by improving glucose sensitive insulin secretion, as well as the liver, by reducing uncontrolled glucose output and restoring post-prandial glucose uptake and storage as glycogen. Herein, we report our efforts on a sulfonamide chemotype with the aim to generate liver selective GK activators which culminated in the discovery of 3-cyclopentyl-N-(5-methoxy-thiazolo[5,4-b]pyridin-2-yl)-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionamide (17c). This compound activated the GK enzyme (αKa = 39 nM) in vitro at low nanomolar concentrations and significantly reduced glucose levels during an oral glucose tolerance test in normal mice.
PATENT
EP-1735322-B1
Example 2(R)-3-Cyclopentyl-N-(5-methoxy-thiazolo[5,4-b]pyridin-2-yl)-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionamide
The title compound is obtained analogously to Example 1 by employing the following additional resolution step:
The racemic title E compound of Example 1, 3-cyclopentyl-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionic acid (10 g, 0.026 mol) in 1,4-dioxane (500 mL) is treated in a three necked 1 liter flask, equipped with heating mantle, water condenser, calcium chloride guard tube and mechanical stirrer with 3.18 g (0.026 mol) of (R)-(+)-1-phenylethylamine. This reaction mixture is then refluxed at 100°C for 1 h. The clear reaction solution is cooled to RT (27°C) and stirred for 10 h. The crystallized salt is collected by filtration under vacuum, washed with 5 mL of hexane and dried under vacuum to afford salt A.
The salt A is dissolved in 1,4-dioxane (500 mL) and heated at 100°C for 1 h. The clear reaction solution is cooled to RT (27°C) and stirred for 10 h. The crystallized product is collected by filtration under vacuum, washed with 50 mL of hexane, and dried under vacuum to afford salt B.
The salt B is dissolved in 1,4-dioxane (290 mL) and heated at 100°C for 1 h. The clear reaction solution is cooled to RT (27°C) and stirred for 10 h. The crystallized product is collected by filtration under vacuum, washed with 30 mL of hexane, and dried under vacuum to afford salt C.
The salt C is dissolved in 1,4-dioxane (100 mL) and heated at 100°C for 1 h. The clear reaction solution is cooled to RT (27°C) and stirred for 10 h. The crystallized product is collected by filtration under vacuum, washed with 30ml of hexane, and dried under vacuum to afford salt D.
The salt D is treated with aqueous hydrochloric acid solution (20 mL, 1 mL of concentrated hydrochloric acid diluted with 100 mL of water) and stirred for 5 min. The white solid precipitates out and is collected by vacuum filtration, washed with 10 mL of cold water, 5 mL of isopropanol and 20 mL of hexane, and dried under vacuum to yield the hydrochloride salt of (R)-(-)-3-cyclopentyl-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionic acid, salt E.
The salt E is neutralized by stirring with aqueous sodium bicarbonate solution (10 mL, 1 g of sodium bicarbonate dissolved in 120 mL of water) for 5 min. The precipitated solid is collected by filtration, washed with 10 mL of cold water, 100 mL of hexane, and dried to afford (R)-(-)-3-cyclopentyl-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionic acid: m.p. 202.2-203.4°C.
Alternatively, the title compound may be obtained by the resolution of the racemic title compound of Example 1 using the following preparative chiral HPLC method:
- Column: Chiralcel OD-R (250 x 20 mm) Diacel make, Japan;
- Solvent A: water:methanol:acetonitrile (10:80:10 v/v/v);
- Solvent B: water:methanol:acetonitrile (05:90:05 v/v/v);
- Using gradient elution: gradient program (time, min / %B): 0/0, 20/0, 50/100, 55/0, 70/0;
- Flow rate: 6.0 mL/min; and
- Detection: by UV at 305 nm.
REFERENCES
US 7750020
WO-2005095418-A1
US-20080103167-A1
| Patent ID | Date | Patent Title |
|---|---|---|
| US2015218151 | 2015-08-06 | NOVEL PHENYLACETAMIDE COMPOUND AND PHARMACEUTICAL CONTAINING SAME |
| US7750020 | 2010-07-06 | Sulfonamide-Thiazolpyridine Derivatives As Glucokinase Activators Useful The Treatment Of Type 2 Diabetes |

Type 2 diabetes is a polygenic disease which afflicts nearly 200 million people worldwide and is expected to increase to near epidemic levels over the next 10−15 years. Glucokinase (GK) activators are currently under investigation by a number of pharmaceutical companies with only a few reaching early clinical evaluation. A GK activator has the promise of potentially affecting both the β-cells of the pancreas, by improving glucose sensitive insulin secretion, as well as the liver, by reducing uncontrolled glucose output and restoring post-prandial glucose uptake and storage as glycogen. Herein, we report our efforts on a sulfonamide chemotype with the aim to generate liver selective GK activators which culminated in the discovery of 3-cyclopentyl-N-(5-methoxy-thiazolo[5,4-b]pyridin-2-yl)-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionamide (17c). This compound activated the GK enzyme (αKa = 39 nM) in vitro at low nanomolar concentrations and significantly reduced glucose levels during an oral glucose tolerance test in normal mice.
https://www.google.com/patents/US7750020
EXAMPLE 2 (R)-3-Cyclopentyl-N-(5-methoxy-thiazolo[5,4-b]pyridin-2-yl)-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionamide
The title compound is obtained analogously to Example 1 by employing the following additional resolution step:
The racemic title E compound of Example 1,3-cyclopentyl-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionic acid (10 g, 0.026 mol) in 1,4-dioxane (500 mL) is treated in a three necked 1 liter flask, equipped with heating mantle, water condenser, calcium chloride guard tube and mechanical stirrer with 3.18 g (0.026 mol) of (R)-(+)-1-phenylethylamine. This reaction mixture is then refluxed at 100° C. for 1 h. The clear reaction solution is cooled to RT (27° C.) and stirred for 10 h. The crystallized salt is collected by filtration under vacuum, washed with 5 mL of hexane and dried under vacuum to afford salt A.
The salt A is dissolved in 1,4-dioxane (500 mL) and heated at 100° C. for 1 h. The clear reaction solution is cooled to RT (27° C.) and stirred for 10 h. The crystallized product is collected by filtration under vacuum, washed with 50 mL of hexane, and dried under vacuum to afford salt B.
The salt B is dissolved in 1,4-dioxane (290 mL) and heated at 100° C. for 1 h. The clear reaction solution is cooled to RT (27° C.) and stirred for 10 h. The crystallized product is collected by filtration under vacuum, washed with 30 mL of hexane, and dried under vacuum to afford salt C.
The salt C is dissolved in 1,4-dioxane (100 mL) and heated at 100° C. for 1 h. The clear reaction solution is cooled to RT (27° C.) and stirred for 10 h. The crystallized product is collected by filtration under vacuum, washed with 30 ml of hexane, and dried under vacuum to afford salt D.
The salt D is treated with aqueous hydrochloric acid solution (20 mL, 1 mL of concentrated hydrochloric acid diluted with 100 mL of water) and stirred for 5 min. The white solid precipitates out and is collected by vacuum filtration, washed with 10 mL of cold water, 5 mL of isopropanol and 20 mL of hexane, and dried under vacuum to yield the hydrochloride salt of (R)-(−)-3-cyclopentyl-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionic acid, salt E.
The salt E is neutralized by stirring with aqueous sodium bicarbonate solution (10 mL, 1 g of sodium bicarbonate dissolved in 120 mL of water) for 5 min. The precipitated solid is collected by filtration, washed with 10 mL of cold water, 100 mL of hexane, and dried to afford (R)-(−)-3-cyclopentyl-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionic acid: m.p. 202.2-203.4° C.
Alternatively, the title compound may be obtained by the resolution of the racemic title compound of Example 1 using the following preparative chiral HPLC method:
- Column: Chiralcel OD-R (250×20 mm) Diacel make, Japan;
- Solvent A: water:methanol:acetonitrile (10:80:10 v/v/v);
- Solvent B: water:methanol:acetonitrile (05:90:05 v/v/v);
- Using gradient elution: gradient program (time, min/% B): 0/0, 20/0, 50/100, 55/0, 70/0;
- Flow rate: 6.0 mL/min; and
- Detection: by UV at 305 nm.
| Patent ID | Date | Patent Title |
|---|---|---|
| US2015218151 | 2015-08-06 | NOVEL PHENYLACETAMIDE COMPOUND AND PHARMACEUTICAL CONTAINING SAME |
| US7750020 | 2010-07-06 | Sulfonamide-Thiazolpyridine Derivatives As Glucokinase Activators Useful The Treatment Of Type 2 Diabetes |
Torrent Research Centre, Village Bhat, Gujarat, India




Mr. Samir Mehta, 52, is the Vice Chairman of the USD 2.75 billion Torrent Group and Chairman of Torrent Pharma

Shri Sudhir Mehta – Chairman Emeritus ::
| Dr. Chaitanya Dutt – Director (Research & Development) :: |
 Born in the year 1950, Dr. Chaitanya Dutt holds an MD in Medicine. He practiced as a consulting physician before joining the company in 1982. Since then he has been associated with the Company. His rich experience spans in the areas of Pharma R&D, clinical research, manufacturing, quality assurance, etc. He is one of the key professionals in the top management team of the Company. He has been instrumental in setting up the Torrent Research Centre (TRC), the research wing of the Company. Under his prudent guidance and leadership, TRC has achieved tremendous progress in the areas of discovery research as well as development work on formulations. He does not hold any directorship in any other company. Born in the year 1950, Dr. Chaitanya Dutt holds an MD in Medicine. He practiced as a consulting physician before joining the company in 1982. Since then he has been associated with the Company. His rich experience spans in the areas of Pharma R&D, clinical research, manufacturing, quality assurance, etc. He is one of the key professionals in the top management team of the Company. He has been instrumental in setting up the Torrent Research Centre (TRC), the research wing of the Company. Under his prudent guidance and leadership, TRC has achieved tremendous progress in the areas of discovery research as well as development work on formulations. He does not hold any directorship in any other company. |
///NOVARTIS, DIABETES, Sulfonamide-Thiazolpyridine Derivatives, Glucokinase Activators, Treatment Of Type 2 Diabetes, 866772-52-3, Novartis Molecule, functionally liver selective glucokinase activators, treatment of type 2 diabetes , NVP-LBX192, LBX-192
c1(sc2nc(ccc2n1)OC)NC(C(c3ccc(cc3)S(=O)(=O)N4CCN(CC4)C)CC5CCCC5)=O
CFG 920, Novartis Scientists team up with Researchers at Aurigene, Bangalore, India,
CFG920,
Inhibitor Of Prostate Cancer With Fewer Cardiac Side Effects
Cas 1260006-20-9
Novartis
Target: CYP17/CYP11B2
Disease: Castration-resistant prostate cancer
MF C14H13ClN4O
MW: 288.0778
Elemental Analysis: C, 58.24; H, 4.54; Cl, 12.28; N, 19.40; O, 5.54
Steroid 17-alpha-hydroxylase inhibitors
CFG920 is a CYP17 inhibitor, is also an orally available inhibitor of the steroid 17-alpha-hydroxylase/C17,20 lyase (CYP17A1 or CYP17), with potential antiandrogen and antineoplastic activities. Upon oral administration, CYP17 inhibitor CFG920 inhibits the enzymatic activity of CYP17A1 in both the testes and adrenal glands, thereby inhibiting androgen production. This may decrease androgen-dependent growth signaling and may inhibit cell proliferation of androgen-dependent tumor cells.
https://clinicaltrials.gov/ct2/show/NCT01647789
NCT01647789: A Study of Oral CFG920 in Patients With Castration Resistant Prostate Cancer2012
- 09 Nov 2015Adverse events, efficacy and pharmacokinetics data from the phase I part of a phase I/II trial in Prostate cancer (Metastatic disease) presented at the 27th AACR-NCI-EORTC International Conference on Molecular Targets and Cancer Therapeutics (AACR-NCI-EORTC-2015)
- 29 Jan 2013Phase-I clinical trials in Prostate cancer in Spain (PO)
- 10 Dec 2012Phase-I clinical trials in Prostate cancer in Canada (PO)
In August 2015, preclinical data were presented at the 250th ACS meeting in Boston, MA. In monkeys, treatment with CFG-920 (3 mg/kg, po) showed good bioavailability with F value of 93%, Tmax of 0.5 h, Cmax of 1382 nM.dn and AUC of 2364 nM.h, while CFG-920 (10 mg/kg, po) showed F value of 183%, Cmax of 1179 nM.dn and Tmax of 1.04 h
Bethany Halford on Twitter: “CFG920 – @Novartis CMOS for …

Novartis is developing CFG-920 (structure shown), an oral CYP17 inhibitor, for the potential treatment of metastatic castration-resistant prostate cancer. In March 2013, a phase I/II trial was initiated and at that time, the study was expected to complete in January 2015; in August 2015, clinical data were presented
2015 250th (August 19) Abs MEDI 341
Discovery of CFG920, a dual CYP17/CYP11B2 inhibitor, for the treatment of castration resistant prostate cancer
American Chemical Society National Meeting and Exposition
Christoph Gaul, Prakash Mistry, Henrik Moebitz, Mark Perrone, Bjoern Gruenenfelder, Nelson Guerreiro, Wolfgang Hackl, Peter Wessels, Estelle Berger, Mark Bock, Saumitra Sengupta, Venkateshwar Rao, Murali Ramachandra, Thomas Antony, Kishore Narayanan, Samiulla Dodheri, Aravind Basavaraju, Shekar Chelur


| Aurigene Discovery Technologies Limited, an independent subsidiary of Dr Reddy’s, CSN Murthy is chief executive officer |

WO 2010149755

Prostate cancer is the most commonly occurring cancer in men. Doctors often treat the metastatic stage of the disease by depriving the patient of sex hormones via chemical or surgical castration. But if it progresses far enough, the cancer can survive this therapy, transforming into the castration-resistant form. “Once the cancer becomes castration-resistant, the prognosis is poor,” said Novartis’s Christoph Gaul.
In recent years, CYP17, a bifunctional 17α-hydroxylase/17,20-lyase cytochrome P450 enzyme, has emerged as a target for treating castration-resistant prostate cancer. The enzyme catalyzes the biosynthesis of sex hormones, including testosterone, and blocking it can starve prostate cancer of the androgens it needs to thrive.
Johnson & Johnson’s CYP17 inhibitor, abiraterone acetate (Zytiga), a steroid that binds irreversibly to CYP17, was approved by the Food & Drug Administration in 2011. But Novartis scientists thought they could make a better CYP17 inhibitor, Gaul told C&EN. They teamed up with researchers at Aurigene, in Bangalore, India, and came up with their clinical candidate, CFG920.
Unlike abiraterone, CFG920 isn’t a steroid, and it inhibits CYP17 reversibly. It also reversibly inhibits another cytochrome P450 enzyme, CYP11B2, which is involved in the synthesis of the mineralocorticoids, hormones that regulate cardiac function.
Treating prostate cancer patients by lowering their androgen levels turns out to have negative cardiac side effects: Patients’ lipid metabolism is thrown off and their mineralocorticoid levels jump, leading to increases in blood pressure. Those changes can be stressful for the heart. “If prostate cancer patients don’t die because of the cancer, a lot of times they die because of cardiac disease,” Gaul said.
Because CFG920 also keeps mineralocorticoid levels in check, Novartis is hoping the drug candidate will ameliorate some of the cardiac side effects of inhibiting CYP17. The compound is currently in Phase I clinical trials.
PATENT
WO 2010149755
Example 58
Prύpιn”ation ofI'(2’ChIoroψ}ri(ibi-^’\l)’3’f4’metMψ}τUin’3’yl)-imiJazoliJin’2’θne (5HA)-
Using the same reaction conditions as in Example 14. 1-(4-methyl-pyridin-3-yl)- itnida/olidin-2-onc ().-.!.4b: 600 mg. 3.3898 mmol) uas reacted with 2-chloro-4-iodo- py.idine (974 mg.4.067 mmol). 1 , 4-dioxane (60 mL). copper iodide (65 mg, 0.3398 mmol), /r<w.v-1.2-diamino cycK)hexane (0.12 ml,, 1.0169 mmol) and potassium phosphate (2.15 g, 10.1694 mmol) to afford 810 mg of the product (83% yield).
1H NMR (C1DCI3. 300 Mi l/): 6 8.5-8.4 (m. 211). 8.3 (d. IH), 7.6-7.5 (m, 2H). 7.2 (S. 111). 4.1-3.9 (ni. 4H), 2.35 <s. 3H)
LCVIS puιϊt>: 90.8%. nι-7 – 289.1 (M M)
HPl C: 97.14%
REFERENCES
1: Gomez L, Kovac JR, Lamb DJ. CYP17A1 inhibitors in castration-resistant prostate cancer. Steroids. 2015 Mar;95:80-7. doi: 10.1016/j.steroids.2014.12.021. Epub 2015 Jan 3. Review. PubMed PMID: 25560485; PubMed Central PMCID: PMC4323677.
2: Yin L, Hu Q, Hartmann RW. Recent progress in pharmaceutical therapies for castration-resistant prostate cancer. Int J Mol Sci. 2013 Jul 4;14(7):13958-78. doi: 10.3390/ijms140713958. Review. PubMed PMID: 23880851; PubMed Central PMCID: PMC3742227.
///////CFG-920, CYP17 inhibitor (prostate cancer), Novartis, CFG 920, Novartis scientists, team up , researchers , Aurigene, Bangalore, India,
Novartis Molecule for functionally liver selective glucokinase activators for the treatment of type 2 diabetes

(R)-3-Cyclopentyl-N-(5-methoxy-thiazolo[5,4-b]pyridin-2-yl)-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionamide
3-Cyclopentyl-N-(5-methoxy-thiazolo[5,4-b]pyridin-2-yl)-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionamide
(3-Cyclopentyl-N-(5-methoxy-thiazolo[5,4-b]pyridin-2-yl)-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionamide)
cas 866772-52-3
NVP-LBX192
LBX-192

54 Discovery and Evaluation of NVP-LBX192, a Liver Targeted Glucokinase Activator
https://acs.confex.com/acs/nerm09/webprogram/Paper75087.html
| Molecular Formula: | C26H33N5O4S2 |
|---|---|
| Molecular Weight: | 543.70132 g/mol |
Sulfonamide-Thiazolpyridine Derivatives, Glucokinase Activators, Treatment Of Type 2 Diabetes
2009 52 (19) 6142 – 6152
Investigation of functionally liver selective glucokinase activators for the treatment of type 2 diabetes
Journal of Medicinal Chemistry
Bebernitz GR, Beaulieu V, Dale BA, Deacon R, Duttaroy A, Gao JP, Grondine MS, Gupta RC, Kakmak M, Kavana M, Kirman LC, Liang JS, Maniara WM, Munshi S, Nadkarni SS, Schuster HF, Stams T, Denny IS, Taslimi PM, Vash B, Caplan SL
2010 240th (August 22) Medi-198
Glucokinase activators with improved physicochemicalproperties and off target effects
American Chemical Society National Meeting and Exposition
Kirman LC, Schuster HF, Grondine MS et al
2010 240th (August 22) Medi-197
Investigation of functionally liver selective glucokinase activators
American Chemical Society National Meeting and Exposition
Schuster HF, Kirman LC, Bebernitz GC et al
PATENT
http://www.google.com/patents/US7750020
EXAMPLE 1 3-Cyclopentyl-N-(5-methoxy-thiazolo[5,4-b]pyridin-2-yl)-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionamide
A. Phenylacetic Acid Ethyl Ester
A solution of phenylacetic acid (50 g, 0.36 mol) in ethanol (150 mL) is treated with catalytic amount of sulfuric acid (4 mL). The reaction mixture is refluxed for 4 h. The reaction is then concentrated in vacuo. The residue is dissolved in diethyl ether (300 mL) and washed with saturated aqueous sodium bicarbonate solution (2×50 mL) and water (1×100 mL). The organic layer dried over sodium sulfate filtered and concentrated in vacuo to give phenylacetic acid ethyl ester as a colorless oil: 1H NMR (400 MHz, CDCl3) δ 1.2 (t, J=7.2, 3H), 3.6 (s, 2H), 4.1 (q, J=7.2, 2H), 7.3 (m, 5H); MS 165 [M+1]+.
B. (4-Chlorosulfonyl-phenyl)-acetic acid ethyl ester
To a cooled chlorosulfonic acid (83.83 g, 48 mL, 0.71 mol) under nitrogen is added the title A compound, phenylacetic acid ethyl ester (59 g, 0.35 mol) over a period of 1 h. Reaction temperature is brought to RT (28° C.), then heated to 70° C., maintaining it at this temperature for 1 h while stirring. Reaction is cooled to RT and poured over saturated aqueous sodium chloride solution (200 mL) followed by extraction with DCM (2×200 mL). The organic layer is washed with water (5×100 mL), followed by saturated aqueous sodium chloride solution (1×150 mL). The organic layer dried over sodium sulfate, filtered and concentrated in vacuo to give crude (4-chlorosulfonyl-phenyl)acetic acid ethyl ester. Further column chromatography over silica gel (60-120 mesh), using 100% hexane afforded pure (4-chlorosulfonyl-phenyl)-acetic acid ethyl ester as a colorless oil.
C. [4-(4-Methyl-piperazine-1-sulfonyl)-phenyl]-acetic acid ethyl ester
A solution of N-methylpiperazine (9.23 g, 10.21 ml, 0.092 mol), DIEA (13 g, 17.4 mL, 0.10 mol) and DCM 80 mL is cooled to 0° C., and to this is added a solution of the title B compound, (4-chlorosulfonyl-phenyl)-acetic acid ethyl ester (22 g, 0.083 mol) in 50 mL of DCM within 30 min. Reaction mixture stirred at 0° C. for 2 h, and the reaction mixture is washed with water (100 mL), followed by 0.1 N aqueous hydrochloric acid solution (1×200 mL). The organic layer dried over sodium sulfate, filtered and concentrated under vacuo to give crude [4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-acetic acid ethyl ester. Column chromatography over silicagel (60-120 mesh), using ethyl acetate afforded pure [4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-acetic acid ethyl ester as white crystalline solid: 1H NMR (400 MHz, CDCl3) δ 1.3 (t, J=7.4, 3H), 2.3 (s, 3H), 2.5 (m, 4H), 3.0 (br s, 4H), 3.7 (s, 2H), 4.2 (q, J=7.4, 2H), 7.4 (d, J=8.3, 2H), 7.7 (d, J=7.3, 2H); MS 327 [M+1]+.
D. 3-Cyclopentyl-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionic acid ethyl ester
A solution of the title C compound, [4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-acetic acid ethyl ester (15 g, 0.046 mol) in a mixture of THF (60 mL) and DMTP (10 mL) is cooled to −78° C. under nitrogen. The resulting solution is stirred at −78° C. for 45 min and to this is added LDA (25.6 mL, 6.40 g, 0.059 mol, 25% solution in THF/Hexane). A solution of iodomethylcyclopentane (11.60 g, 0.055 mol) in a mixture of DMTP (12 mL) and THF (20 mL) is added over a period of 15 min at −78° C. and reaction mixture stirred at −78° C. for 3 h further, followed by stirring at 25° C. for 12 h. The reaction mixture is then quenched by the dropwise addition of saturated aqueous ammonium chloride solution (50 mL) and is concentrated in vacuo. The residue is diluted with water (50 mL) and extracted with ethyl acetate (3×100 mL). The organic solution is washed with a saturated aqueous sodium chloride (2×150 mL), dried over sodium sulfate, filtered and concentrated in vacuo. Column chromatography over silica gel (60-120 mesh), using 50% ethyl acetate in hexane as an eluent to afford 3-cyclopentyl-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionic acid ethyl ester as a white solid: 1H NMR (400 MHz, CDCl3) δ 0.9-2.1 (m, 11H), 1.2 (t, J=7.1, 3H), 2.3 (s, 3H), 2.5 (br s, 4H), 3.0 (br s, 4H), 3.6 (m, 1H), 4.1 (q, J=7.1, 2H), 7.5 (d, J=8.3, 2H), 7.7 (d, J=8.3, 2H); MS 409 [M+1]+.
E. 3-Cyclopentyl-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionic acid
A solution of the title D compound, 3-cyclopentyl-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionic acid ethyl ester (14 g, 0.034 mol) in methanol:water (30 mL:10 mL) and sodium hydroxide (4.11 g, 0.10 mol) is stirred at 60° C. for 8 h in an oil bath. The methanol is then removed in vacuo at 45-50° C. The residue is diluted with water (25 mL) and extracted with ether (1×40 mL). The aqueous layer is acidified to pH 5 with 3 N aqueous hydrochloric acid solution. The precipitated solid is collected by vacuum filtration, washed with water (20 mL), followed by isopropyl alcohol (20 mL). Finally, solid cake is washed with 100 mL of hexane and dried under vacuum at 40° C. for 6 h to give 3-cyclopentyl-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionic acid as a white solid: 1H NMR (400 MHz, CDCl3) δ 1.1-2.0 (m, 11H), 2.4 (s, 3H), 2.7 (br s, 4H), 3.1 (br s, 4H), 3.6 (m, 1H), 7.5 (d, J=8.3, 2H), 7.6 (d, J=8.3, 2H); MS 381 [M+l]+.
F. 5-Methoxy-thiazolo[5,4-b]pyridin-2-ylamine
A solution of 6-methoxy-pyridin-3-ylamine (5.0 g, 0.0403 mol) in 10 mL of acetic acid is added slowly to a solution of potassium thiocyanate (20 g, 0.205 mol) in 100 mL of acetic acid at 0° C. followed by a solution of bromine (2.5 mL, 0.0488 mol) in 5 mL of acetic acid. The reaction is stirred for 2 h at 0° C. and then allowed to warm to RT. The resulting solid is collected by filtration and washed with acetic acid, then partitioned between ethyl acetate and saturated aqueous sodium bicarbonate. The insoluble material is removed by filtration and the organic layer is evaporated and dried to afford 5-methoxy-thiazolo[5,4-b]pyridin-2-ylamine as a tan solid.
G. 3-Cyclopentyl-N-(5-methoxy-thiazolo[5,4-b]pyridin-2-yl)-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionamide
A solution of the title E compound, 3-cyclopentyl-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionic acid (5 g, 0.013 mol) in DCM (250 mL) is cooled to 0° C. and then charged HOBt hydrate (2.66 g, 0.019 mol), followed by EDCI hydrochloride (6 g, 0.031 mol). The reaction mixture is stirred at 0° C. for 5 h. After that the solution of the title F compound, 5-methoxy-thiazolo[5,4-b]pyridin-2-ylamine (2.36 g, 0.013 mol) and D1EA (8 mL, 0.046 mol) in a mixture of DCM (60 mL) and DMF (20 mL) is added dropwise over 30 min. Reaction temperature is maintained at 0° C. for 3 h, then at RT (28° C.) for 3 days. Reaction is diluted with (60 mL) of water and the organic layer is separated and washed with saturated sodium bicarbonate solution (2×50 mL) followed by water washing (2×50 mL) and saturated sodium chloride aqueous solution (1×150 mL). Finally the organic layer is dried over sodium sulfate, filtered, and evaporated under vacuo. The crude product is purified using column chromatography over silica gel (60-120 mesh), using 40% ethyl acetate in hexane as an eluent to afford 3-cyclopentyl-N-(5-methoxy-thiazolo[5,4-b]pyridin-2-yl)-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionamide as a white solid: 1H NMR (400 MHz, CDCl3) δ 0.9-2.1 (m, 11H), 2.2 (s, 3H), 2.5 (br s, 4H), 3.1 (br s, 4H), 3.7 (m, 1H), 4.0 (s, 3H), 6.8 (d, J=8.8, 1H), 7.5 (d, J=8.3, 2H), 7.7 (d, J=8.3, 2H), 7.8 (d, J=8.8, 1H), 8.6 (s, 1H); MS 617 [M+1]+.
H. 3-Cyclopentyl-N-(5-methoxy-thiazolo[5,4-b]pyridin-2-yl)-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionamide dihydrochloride
The title G compound, 3-cyclopentyl-2-(4-methyl piperazinyl sulfonyl)phenyl-N-(5-methoxy-thiazolo[5,4-b]pyridin-2-yl)propionamide (2.8 g, 0.0051 mol) is added to a cooled solution of 10% hydrochloric acid in isopropanol (3.75 mL). The reaction mixture is stirred at 0° C. for 1 h and then at RT for 2 h. The solid is separated, triturated with 10 mL of isopropanol and collected by vacuum filtration and washed with 50 mL of hexane. The solid is dried at 70° C. for 48 h to afford 3-cyclopentyl-N-(5-methoxy-thiazolo[5,4-b]pyridin-2-yl)-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionamide dihydrochloride as an off white solid.
EXAMPLE 2 (R)-3-Cyclopentyl-N-(5-methoxy-thiazolo[5,4-b]pyridin-2-yl)-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionamide
The title compound is obtained analogously to Example 1 by employing the following additional resolution step:
The racemic title E compound of Example 1,3-cyclopentyl-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionic acid (10 g, 0.026 mol) in 1,4-dioxane (500 mL) is treated in a three necked 1 liter flask, equipped with heating mantle, water condenser, calcium chloride guard tube and mechanical stirrer with 3.18 g (0.026 mol) of (R)-(+)-1-phenylethylamine. This reaction mixture is then refluxed at 100° C. for 1 h. The clear reaction solution is cooled to RT (27° C.) and stirred for 10 h. The crystallized salt is collected by filtration under vacuum, washed with 5 mL of hexane and dried under vacuum to afford salt A.
The salt A is dissolved in 1,4-dioxane (500 mL) and heated at 100° C. for 1 h. The clear reaction solution is cooled to RT (27° C.) and stirred for 10 h. The crystallized product is collected by filtration under vacuum, washed with 50 mL of hexane, and dried under vacuum to afford salt B.
The salt B is dissolved in 1,4-dioxane (290 mL) and heated at 100° C. for 1 h. The clear reaction solution is cooled to RT (27° C.) and stirred for 10 h. The crystallized product is collected by filtration under vacuum, washed with 30 mL of hexane, and dried under vacuum to afford salt C.
The salt C is dissolved in 1,4-dioxane (100 mL) and heated at 100° C. for 1 h. The clear reaction solution is cooled to RT (27° C.) and stirred for 10 h. The crystallized product is collected by filtration under vacuum, washed with 30 ml of hexane, and dried under vacuum to afford salt D.
The salt D is treated with aqueous hydrochloric acid solution (20 mL, 1 mL of concentrated hydrochloric acid diluted with 100 mL of water) and stirred for 5 min. The white solid precipitates out and is collected by vacuum filtration, washed with 10 mL of cold water, 5 mL of isopropanol and 20 mL of hexane, and dried under vacuum to yield the hydrochloride salt of (R)-(−)-3-cyclopentyl-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionic acid, salt E.
The salt E is neutralized by stirring with aqueous sodium bicarbonate solution (10 mL, 1 g of sodium bicarbonate dissolved in 120 mL of water) for 5 min. The precipitated solid is collected by filtration, washed with 10 mL of cold water, 100 mL of hexane, and dried to afford (R)-(−)-3-cyclopentyl-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionic acid: m.p. 202.2-203.4° C.
Alternatively, the title compound may be obtained by the resolution of the racemic title compound of Example 1 using the following preparative chiral HPLC method:
- Column: Chiralcel OD-R (250×20 mm) Diacel make, Japan;
- Solvent A: water:methanol:acetonitrile (10:80:10 v/v/v);
- Solvent B: water:methanol:acetonitrile (05:90:05 v/v/v);
- Using gradient elution: gradient program (time, min/% B): 0/0, 20/0, 50/100, 55/0, 70/0;
- Flow rate: 6.0 mL/min; and
- Detection: by UV at 305 nm.
EXAMPLE 3 (S)-3-Cyclopentyl-N-(5-methoxy-thiazolo[5,4-b]pyridin-2-yl)-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionamide
The title compound is prepared analogously to Example 2.
J MED CHEM 2009, 52, 6142-52
Investigation of Functionally Liver Selective Glucokinase Activators for the Treatment of Type 2 Diabetes
http://pubs.acs.org/doi/abs/10.1021/jm900839k

Type 2 diabetes is a polygenic disease which afflicts nearly 200 million people worldwide and is expected to increase to near epidemic levels over the next 10−15 years. Glucokinase (GK) activators are currently under investigation by a number of pharmaceutical companies with only a few reaching early clinical evaluation. A GK activator has the promise of potentially affecting both the β-cells of the pancreas, by improving glucose sensitive insulin secretion, as well as the liver, by reducing uncontrolled glucose output and restoring post-prandial glucose uptake and storage as glycogen. Herein, we report our efforts on a sulfonamide chemotype with the aim to generate liver selective GK activators which culminated in the discovery of 3-cyclopentyl-N-(5-methoxy-thiazolo[5,4-b]pyridin-2-yl)-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionamide (17c). This compound activated the GK enzyme (αKa = 39 nM) in vitro at low nanomolar concentrations and significantly reduced glucose levels during an oral glucose tolerance test in normal mice.
PATENT
EP-1735322-B1
Example 2(R)-3-Cyclopentyl-N-(5-methoxy-thiazolo[5,4-b]pyridin-2-yl)-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionamide
The title compound is obtained analogously to Example 1 by employing the following additional resolution step:
The racemic title E compound of Example 1, 3-cyclopentyl-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionic acid (10 g, 0.026 mol) in 1,4-dioxane (500 mL) is treated in a three necked 1 liter flask, equipped with heating mantle, water condenser, calcium chloride guard tube and mechanical stirrer with 3.18 g (0.026 mol) of (R)-(+)-1-phenylethylamine. This reaction mixture is then refluxed at 100°C for 1 h. The clear reaction solution is cooled to RT (27°C) and stirred for 10 h. The crystallized salt is collected by filtration under vacuum, washed with 5 mL of hexane and dried under vacuum to afford salt A.
The salt A is dissolved in 1,4-dioxane (500 mL) and heated at 100°C for 1 h. The clear reaction solution is cooled to RT (27°C) and stirred for 10 h. The crystallized product is collected by filtration under vacuum, washed with 50 mL of hexane, and dried under vacuum to afford salt B.
The salt B is dissolved in 1,4-dioxane (290 mL) and heated at 100°C for 1 h. The clear reaction solution is cooled to RT (27°C) and stirred for 10 h. The crystallized product is collected by filtration under vacuum, washed with 30 mL of hexane, and dried under vacuum to afford salt C.
The salt C is dissolved in 1,4-dioxane (100 mL) and heated at 100°C for 1 h. The clear reaction solution is cooled to RT (27°C) and stirred for 10 h. The crystallized product is collected by filtration under vacuum, washed with 30ml of hexane, and dried under vacuum to afford salt D.
The salt D is treated with aqueous hydrochloric acid solution (20 mL, 1 mL of concentrated hydrochloric acid diluted with 100 mL of water) and stirred for 5 min. The white solid precipitates out and is collected by vacuum filtration, washed with 10 mL of cold water, 5 mL of isopropanol and 20 mL of hexane, and dried under vacuum to yield the hydrochloride salt of (R)-(-)-3-cyclopentyl-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionic acid, salt E.
The salt E is neutralized by stirring with aqueous sodium bicarbonate solution (10 mL, 1 g of sodium bicarbonate dissolved in 120 mL of water) for 5 min. The precipitated solid is collected by filtration, washed with 10 mL of cold water, 100 mL of hexane, and dried to afford (R)-(-)-3-cyclopentyl-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionic acid: m.p. 202.2-203.4°C.
Alternatively, the title compound may be obtained by the resolution of the racemic title compound of Example 1 using the following preparative chiral HPLC method:
- Column: Chiralcel OD-R (250 x 20 mm) Diacel make, Japan;
- Solvent A: water:methanol:acetonitrile (10:80:10 v/v/v);
- Solvent B: water:methanol:acetonitrile (05:90:05 v/v/v);
- Using gradient elution: gradient program (time, min / %B): 0/0, 20/0, 50/100, 55/0, 70/0;
- Flow rate: 6.0 mL/min; and
- Detection: by UV at 305 nm.
REFERENCES
US 7750020
WO-2005095418-A1
US-20080103167-A1
| Patent ID | Date | Patent Title |
|---|---|---|
| US2015218151 | 2015-08-06 | NOVEL PHENYLACETAMIDE COMPOUND AND PHARMACEUTICAL CONTAINING SAME |
| US7750020 | 2010-07-06 | Sulfonamide-Thiazolpyridine Derivatives As Glucokinase Activators Useful The Treatment Of Type 2 Diabetes |
///NOVARTIS, DIABETES, Sulfonamide-Thiazolpyridine Derivatives, Glucokinase Activators, Treatment Of Type 2 Diabetes, 866772-52-3, Novartis Molecule, functionally liver selective glucokinase activators, treatment of type 2 diabetes , NVP-LBX192, LBX-192
c1(sc2nc(ccc2n1)OC)NC(C(c3ccc(cc3)S(=O)(=O)N4CCN(CC4)C)CC5CCCC5)=O
RP 6503, Novartis to develop and commercialize Rhizen’s inhaled dual PI3K-delta gamma inhibitor
RP 6503
RP 6503
| Molecular Formula: | C30H24F2N6O5S |
|---|---|
| Molecular Weight: | 618.610566 g/mol |
Mass: 619.1 (M++l). MP: 175-178° C Specific optical rotation (C=l in chloroform, at 25°C) : [a]D = + 147.16.

A1
RP 6503
(S)-N-(5-(4-amino-l-(l-(5-fluoro-3-(3-fluorophenyl)-4-oxo-4H-chromen-2-yl) ethyl)-lH-pyrazolo[3,4-d]pyrimidin-3-yl)-2-methoxyphenyl)methanesulfonamide
(S)-N-[5-[4-amino-1-[1-[5-fluoro-3-(3-fluorophenyl)-4-oxochromen-2-yl]ethyl]pyrazolo[3,4-d]pyrimidin-3-yl]-2-methoxyphenyl]methanesulfonamide


Novartis to develop and commercialize Rhizen’s inhaled dual PI3K-delta gamma inhibitor and related compounds worldwide
The immune pipeline includes ‘dual PI3K inhibitors for various indications’ licensed to Novartis
‘inhaled dual inhibitor’,
Phosphoinositide-3 kinase delta inhibitor; Phosphoinositide-3 kinase gamma inhibitor
WO2011055215A2 and WO2012151525A1 and U.S. Publication Nos. US20110118257 and US20120289496
Rhizen Pharmaceuticals Sa INNOVATOR
| Incozen Therapeutics Pvt. Ltd., Rhizen Pharmaceuticals Sa |
PATENT
http://www.google.com/patents/WO2011055215A2?cl=en
PATENT
http://www.google.com/patents/WO2012151525A1?cl=en
scheme 1A:
Ste -1
Step-2
Scheme 2
SCHEME 3
SCHEME4
List of Intermediates

Intermediate 27: 2-( l -(4-amino-3-iodo-lH-pyrazolo[3,4-d]pyrimidin- l – yl)ethyl)-5-fluoro-3-(3-fluorophenyl)-4H-chromen-4-one: To a solution of 3-iodo- l H- pyrazolo[3,4-d]pyrimidin-4-amine (0.800 g, 2.88 mmol) in DMF (5 ml), potassium carbonate (0.398 g, 2.88 mmol) was added and stirred at RT for 30 min. To this mixture intermediate 22 (0.500 g, 1.44 mmol) was added and stirred for 12h. The reaction mixture was diluted with water and extracted with ethyl acetate. The organic layer was dried over sodium sulphate and concentrated under reduced pressure. The crude product was purified by column chromatography with methanol: dichloromethane to afford the title compound as a off-white solid (0.300 g, 38%). Ή-NMR (5 ppm, DMSO-d63, 400 MHz): 8.02 (s, 1 H), 7.94 (s, 1 H), 7.84 (dt, J = 8.4,5.7 Hz, 1H), 7.47 (d, 7 = 8.6 Hz, 1H), 7.29 (m, 3H), 7.09 (dt, 7 = 8.8,2.3 Hz, 1 H), 6.87 (s, 2H), 5.88 (q, 7 = 7.0 Hz, 1H), 1.82 (d, 7 = 7.0 Hz, 3H).
SYNTHESIS
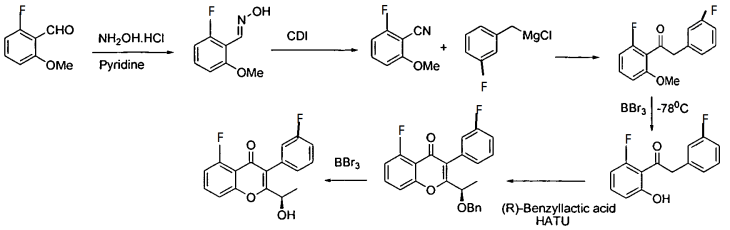
MAIN PART
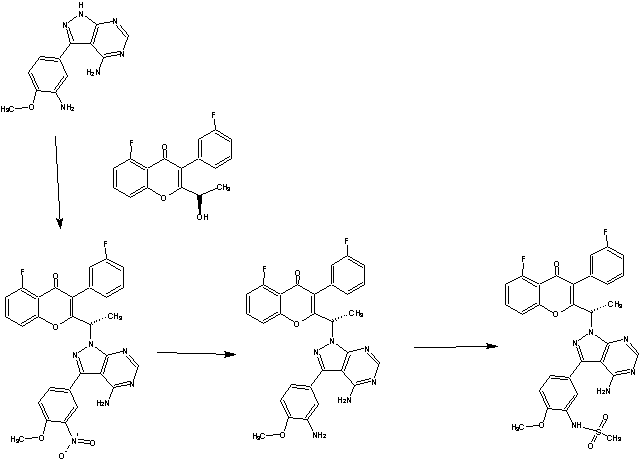
PATENT
http://www.google.com/patents/WO2015198289A1?cl=en
Prashant Kashinath Bhavar, Swaroop Kumar Venkata Satya VAKKALANKA
The present invention relates to a selective dual delta (δ) and gamma (γ) PI3K protein kinase modulator (S)-N-(5-(4-amino-1-(1-(5-fluoro-3-(3-fluorophenyl)-4-oxo-4H- chromen-2-yl)ethyl)-1H-pyrazolo[3,4-d]pyrimidin-3-yl)-2-methoxyphenyl) methane sulfonamide, methods of preparing them, pharmaceutical compositions containing them and methods of treatment, prevention and/or amelioration of PI3K kinase mediated diseases or disorders with them.
compound of formula (Al):

(Al).
The process comprises the steps of:
(a) subjecting (R)-5-fluoro-3-(3-fluorophenyl)-2-(l-hydroxyethyl)-4H-chromen-4-one:

to a Mitsunobu reaction with 3-(4-methoxy-3-nitrophenyl)-lH-pyrazolo[3,4-d]pyrimidin-4-amine:

(for example, in the presence of triphenylphosphine and diisopropylazodicarboxylate) to give (S)-2-(l-(4-amino-3-(4-methoxy-3-nitrophenyl)-lH-pyrazolo[3,4-d]pyrimidin-l-yl)ethyl)-5-fluoro-3-(3-fluorophenyl)-4H-chromen-4-one (Intermediate 3):

Intermediate 3;
(b) reducing Intermediate 3, for example with a reducing agent such as Raney Ni, to give (S)-2-(l-(4-amino-3-(3-amino-4-methoxyphenyl)-lH-pyrazolo[3,4-d]pyrimidin- l-yl)ethyl)-5-fluoro-3-( -fluorophenyl)-4H-chromen-4-one (Intermediate 4):

Intermediate 4;
The intermediates described herein may be prepared by the methods described in International Publication Nos. WO 11/055215 and WO 12/151525, both of which are hereby incorporated by reference.
Intermediate 1: N-(5-bromo-2-methoxyphenyl)methanesulfonamide:
To a solution of 5-bromo-2-methoxyaniline(1.00 g, 4.94 mmol) in dichloromethane (10 ml), pyridine (0.800 ml, 9.89 mmol) was added and cooled to 0°C. Methane sulphonyl chloride (0.40 ml, 5.19 mmol) was added and stirred for 30 min. The reaction mixture was quenched with water, extracted with ethyl acetate, dried over anhydrous sodium sulphate and concentrated under reduced pressure. The crude product was chromatographed with ethyl acetate : petroleum ether to afford the title compound as a reddish solid (1.20 g, 87%).
Intermediate 2: N-(2-methoxy-5-(4,4,5,5-tetramethyl-l,3,2-dioxaborolan-2-yl)phenyl)methanesulfonamide: Potassium acetate (0.841 g, 8.57 mmol) and bis(pinacolato)diboron (1.190 g, 4.71 mmol) were added to a solution of intermediate 1 (1.20 g, 4.28 mmol) in dioxane (17.5 ml) and the solution was degassed for 30 min.[l, -Bis(diphenylphosphino)ferrocene]dichloro palladium(II).CH2Ci2 (0.104 g, 0.128 mmol) was added under nitrogen atmosphere and heated to 80°C. After 2h the
reaction mixture was filtered through celite and concentrated. The crude product was purified by column chromatography with ethyl acetate : petroleum ether to afford the title compound as a yellow solid (1.00 g, 71%).JH-NMR (δ ppm, CDCb, 400 MHz): 7. 91 (d, / = 1.2Hz, 1H), 7. 62 (dd, / = 8.1, 1.2Hz, 1H), 6. 92 (d, / = 8.1Hz, 1H), 6.73 (s, 1H), 3.91 (s, 3H), 2.98 (s, 3H), 1.32 (s, 12H).
Intermediate 3: (S)-2-(l-(4-amino-3-(4-methoxy-3-nitrophenyl)-lH-pyrazolo[3,4-d]pyrimidin-l-yl)ethyl)-5-fluoro-3-(3-fluorophenyl)-4H-chromen-4-one: (S)-2-(l-(4-amino-3-(4-methoxy-3-nitrophenyl)-lH-pyrazolo[3,4-d]pyrimidin-l-yl)ethyl)-5-fluoro-3-(3-fluorophenyl)-4H-chromen-4-one: To a solution of (R)-5-fluoro-3-(3-fluorophenyl)-2-(l-hydroxyethyl)-4H-chromen-4-one (0.500 g, 1.64 mmol) in THF (5 ml), 3-(4-methoxy-3-nitrophenyl)-lH-pyrazolo[3,4-d]pyrimidin-4-amine (0.564 g, 1.97 mmol) and triphenylphosphine (0.649 g, 2.47 mmol) were added followed by the addition of diisopropylazodicarboxylate (0.50 ml, 2.47 mmol). ((R)-5-fluoro-3-(3-fluorophenyl)-2-(l-hydroxyethyl)-4H-chromen-4-one can be prepared as described for Intermediates 23, 25, and 26 in International Publication No. WO 2012/0151525.). After 4h at room temperature, the mixture was concentrated and the residue was purified by column chromatography with ethyl acetate : petroleum ether to afford the title compound as a brown solid (0.270 g, 29%). JH-NMR (δ ppm, DMSO-d6, 400 MHz): 8.04 (s, 1H), 7.83 (m, 1H), 7.63-7.50 (m, 3H), 7.29 (m, 2H), 7.06 (dt, J = 8.7,2.2Hz, 1H), 6.94 (m, 2H), 6.75 (dd, J = 8.1,2.1Hz, 1H), 5.95 (q, J = 7.0Hz, 1H), 4.98 (s, 2H), 3.81 (s, 3H), 1.86 (d, J = 7.0 Hz, 3H).
[109] Intermediate 4: (S)-2-(l-(4-amino-3-(3-amino-4-methoxyphenyl)-lH-pyrazolo[3,4-d]pyrimidin-l-yl)ethyl)-5-fluoro-3-(3-fluorophenyl)-4H-chromen-4-one:
(S)-2-(l-(4-amino-3-(3-amino-4-methoxyphenyl)-lH-pyrazolo[3,4-d]pyrimidin-l-yl)ethyl)-5-fluoro-3-(3-fluorophenyl)-4H-chromen-4-one : To a solution of Intermediate 3 (0.260 g, 0.455 mmol) in ethanol (5 ml), Raney Ni (0.130 g) was added and hydrogeneated at 20psi at 50°C for 24h. The reaction mixture was passed through celitepad and concentrated to afford the title compound as a brown solid (0.150 g, 60%). Mass : 540.8 (M+).
Example A
N-(5-(4-amino-l-(l-(5-fluoro-3-(3-fluorophenyl)-4-oxo-4H-chromen-2-yl)ethyl)-lH- pyrazolo[3,4-d]pyrimidin-3-yl)-2-methoxyphenyl)methanesulfonamide
To a solution of 2-(l-(4-amino-3-iodo-lH-pyrazolo[3,4-d]pyrimidin-l-yl)ethyl)-5-fluoro-3-(3-fluorophenyl)-4H-chromen-4-one (0.200 g, 0.366 mmol) in DME (2.1 ml) and water (0.67 ml), intermediate 2 (0.179 g, 0.550 mmol) and sodium carbonate (0.116 g, 1.10 mmol) were added and the system was degassed for 30 min. (2-(l-(4-amino-3-iodo-lH^yrazolo[3,4-d]pyrimidin-l-yl)ethyl)-5-fluoro-3-(3-fluorophenyl)-4H-chromen-4-one can be prepared as described for Intermediates 23, 25, and 26 in International Publication No. WO 2012/0151525). Bis(diphenylphosphino) ferrocene]dichloropalladium(II) (0.059 g, 0.075 mmol) was added and kept under microwave irradiation (microwave power = 100W, temperature = 100 °C) for 45 min. The reaction mixture was Celite filtered, concentrated and extracted with ethyl acetate. The organic layer was dried over sodium sulphate and concentrated under reduced pressure. The crude product was purified by column chromatography with methanol: dichloromethane to afford the title compound as a brown solid (0.080 g, 35%). MP: 216-218 °C. ¾-NMR (δ ppm, CDCb, 400 MHz): 8.20 (s, 1H), 7.73 (s, 1H), 7.53 (m, 2H), 7.31 (m, 2H), 7.07-6.73 (m, 6H), 6.07 (q, / = 6.2 Hz, 1H), 3.98 (s, 3H), 3.14 (s, 3H), 2.01 (d, / = 6.0Hz, 3H).
Example Al and A2
Method A
(S)-N-(5-(4-amino-l-(l-(5-fluoro-3-(3-fluorophenyl)-4-oxo-4H-chromen-2-yl)ethyl)- lH-pyrazolo[3,4-d]pyrimidin-3-yl)-2-methoxyphenyl)methanesulfonamide
and (R)-N-(5-(4-amino-l-(l-(5-fluoro-3-(3-fluorophenyl)-4-oxo-4H-chromen-2- yl)ethyl)-lH-p anesulfonamide

The two enantiomerically pure isomers were separated by preparative SFC (supercritical fluid) conditions from N-(5-(4-amino-l-(l-(5-fluoro-3-(3-fluorophenyl)-4-oxo-4H-chromen-2-yl)ethyl)-lH-pyrazolo[3,4-d]pyrimidin-3-yl)-2-methoxyphenyl)methanesulfonamide (0.500 g) on a CHIRALPAK AS-H column (250 x 30 mm; 5μπι) using methanol : CO2 (55:45) as the mobile phase at a flow rate of 80g / min.
Example Al (S-isomer): Brown solid (0.247 g). Enantiomeric excess: 97.4%. Retention time: 2.14 min. Mass: 619.1 (M++l). MP: 156-158° C.
Example A2 (R-isomer): Brown solid (0.182 g). Enantiomeric excess: 99.3%. Retention t: 3.43 min. Mass: 619.1 (M++l). MP: 168-171° C.
Method Al
(S)-N-(5-(4-amino-l-(l-(5-fluoro-3-(3-fluorophenyl)-4-oxo-4H-chromen-2-yl)ethyl)- lH-pyrazolo[3,4-d]pyrimidin-3-yl)-2-methoxyphenyl)methanesulfonamide
and (R)-N-(5-(4-amino-l-(l-(5-fluoro-3-(3-fluorophenyl)-4-oxo-4H-chromen-2- yl)ethyl)-lH-p anesulfonamide

The two enantiomerically pure isomers were separated by preparative SFC (supercritical fluid) conditions from N-(5-(4-amino-l-(l-(5-fluoro-3-(3-fluorophenyl)-4-oxo-4H-chromen-2-yl)ethyl)-lH-pyrazolo[3,4-d]pyrimidin-3-yl)-2-methoxyphenyl) methanesulfonamide (15.0 g) on a CHIRALPAK AS-H column (250 x 20 mm; 5μπι) using methanol : CO2 (45:55) as the mobile phase at a flow rate of 120g / min.
Example Al (S-isomer): Enantiomeric excess: 100 %. Retention time: 2.21 min. Mass: 619.1 (M++l). MP: 175-178° C Specific optical rotation (C=l in chloroform, at 25°C) : [a]D = + 147.16.
Example A2 (R-isomer): Enantiomeric excess: 99.3%. Retention t: 3.72 min. Mass: 619.1 (M++l). MP: 154-157° C. Specific optical rotation (C=l in chloroform, at 25°C) : [a]D = – 159.54.
Method B
Example Al
(S)-N-(5-(4-amino-l-(l-(5-fluoro-3-(3-fluorophenyl)-4-oxo-4H-chromen-2-yl)ethyl)- lH-pyrazolo[3,4-d]pyrimidin-3-yl)-2-methoxyphenyl)methanesulfonamide
To a solution of Intermediate 4 (0.500 g, 0.923 mmol) in dichloromethane (5 ml) cooled to 0°C, pyridine (0.200 ml, 1.84 mmol) was added and stirred for 10 min. Methanesulphonyl chloride (0.100 ml, 0.923 mmol) was added stirred for 30 min. The reaction mixture was quenched with water, extracted with dichloromethane and dried over sodium sulphate. The crude product was column chromatographed with methanol : dichloromethane to afford the title compound as an off-white solid (0.240 g, 42%). MP: 211-213°C. ¾-NMR (δ ppm, DMSO-d6, 400 MHz): 9.15 (s, 1H), 8.06 (s, 1H), 7.83 (m, 1H), 7.49 (m, 4H), 7.28 (m, 4H), 7.08 (dt, / = 8.6, 1.7 Hz, 1H), 6.92 (s, 2H), 5.98 (q, / = 6.9 Hz, 1H), 3.88 (s, 3H), 2.99 (s, 3H), 1.88 (d, / = 7.0 Hz, 3H). Enantiomeric excess: 85.4% as determined by HPLC on a chiralpak AS-3R column, enriched in the fast eluting isomer (retention time = 7.46 min.).
CLIPS
La Chaux-de-Fonds, Switzerland, Sept. 6, 2013 — La Chaux-de-Fonds, Switzerland (6 September 2013): Rhizen Pharmaceuticals S.A. announces a scientific poster presentation on the pre-clinical characterization of its lead calcium release activated channel (CRAC) inhibitor, RP3128, for the treatment of respiratory disorders and an oral presentation on the pharmacological profile of its novel, dual Phosphoinositide-3 kinase (PI3K) delta/gamma inhibitor, RP6503, in the pulmonary disease systems, at the European Respiratory Society Annual Congress (ERS), to be held from 7-11 September 2013, at Barcelona, Spain.
RP6503 is a novel, potent and selective inhibitor of the delta and gamma isoforms of PI3K. It is to be delivered via the inhalation route and has a long duration of action along with excellent PI3K isoform selectivity, which is expected to result in better safety. RP3128 has been optimized with high potency for CRAC channel inhibition, selectivity over the other voltage gated channels and excellent oral bioavailability. Rhizen intends to move both these compounds to the clinic in 2014.
Details of the presentations:
1. Abstract of the Poster Presentation: “Pre-clinical characterization of RP3128, a novel and potent CRAC channel inhibitor for the treatment of respiratory disorders”
Time and Location- 8 September 2013 between 14.45-16.45 in Room 3.6, at Poster Discussion: New drugs in respiratory medicine, at FIRA BARCELONA, Convention Centre de Gran Via, Barcelona, Spain
2. Abstract of Oral Presentation: “In vitro and in vivo pharmacological profile of RP6503, a novel dual PI3K delta/gamma inhibitor, in pulmonary disease systems”
Time and Location- 11 September 2013 at 8.45 in Room 3.9; Session 8.30-10.30, at the Oral Presentation: Emerging new targets for the treatment of respiratory diseases, at FIRA BARCELONA, Convention Centre de Gran Via, Barcelona, Spain
CLIPS
La Chaux-de-Fonds, Switzerland , Dec. 09, 2015 — Rhizen Pharmaceuticals S.A. announced today that they have entered into an exclusive, worldwide license agreement with Novartis for the development and commercialization of Rhizen’s, inhaled dual PI3K-delta gamma inhibitor and its closely related compounds for various indications.
Under the terms of the agreement, Rhizen will receive an upfront payment and is eligible to receive development, regulatory and sales milestones payments. In addition Rhizen is also eligible to receive tiered royalties on annual nets sales.
The lead compound is a novel, potent, and selective dual PI3K-delta gamma inhibitor with demonstrated anti-inflammatory and immuno-modulatory activity in pre-clinical systems and models representative of respiratory diseases. With a favorable ADME and PK profile and high therapeutic index in animals, the inhaled dual PI3K-delta gamma inhibitor holds promise in the treatment of human airway disorders.
About Rhizen Pharmaceuticals S.A.:
Rhizen Pharmaceuticals is an innovative, clinical-stage biopharmaceutical company focused on the discovery and development of novel therapeutics for the treatment of cancer, immune and metabolic disorders. Since its establishment in 2008, Rhizen has created a diverse pipeline of proprietary drug candidates targeting several cancers and immune associated cellular pathways. Rhizen is headquartered in La-Chaux-de-Fonds, Switzerland. For additional information, please visit Rhizen’s website, http://www.rhizen.com.
SEE
http://www.atsjournals.org/doi/abs/10.1164/ajrccm-conference.2013.187.1_MeetingAbstracts.A3880
| WO2011055215A2 | Nov 3, 2010 | May 12, 2011 | Incozen Therapeutics Pvt. Ltd. | Novel kinase modulators |
| WO2012008302A1 | Jun 28, 2011 | Jan 19, 2012 | National University Corporation Tottori University | Method for preparing novel hipsc by means of mirna introduction |
| WO2012121953A1 | Feb 29, 2012 | Sep 13, 2012 | The Trustees Of Columbia University In The City Of New York | Methods and pharmaceutical compositions for treating lymphoid malignancy |
| WO2012151525A1 | May 4, 2012 | Nov 8, 2012 | Rhizen Pharmaceuticals Sa | Novel compounds as modulators of protein kinases |
| WO2013164801A1 | May 3, 2013 | Nov 7, 2013 | Rhizen Pharmaceuticals Sa | Process for preparation of optically pure and optionally substituted 2- (1 -hydroxy- alkyl) – chromen – 4 – one derivatives and their use in preparing pharmaceuticals |
| US20110118257 | May 19, 2011 | Rhizen Pharmaceuticals Sa | Novel kinase modulators | |
| US20120289496 | May 4, 2012 | Nov 15, 2012 | Rhizen Pharmaceuticals Sa | Novel compounds as modulators of protein kinases |
///////RP 6503, Novartis, develop, commercialize, Rhizen, inhaled dual PI3K-delta gamma inhibitor, PHASE 1, RP-6503
c21c(cccc1O/C(=C(\C2=O)c3cc(ccc3)F)C(C)n4c6ncnc(c6c(n4)c5cc(c(cc5)OC)NS(=O)(=O)C)N)F
CC(C1=C(C(=O)C2=C(O1)C=CC=C2F)C3=CC(=CC=C3)F)N4C5=C(C(=N4)C6=CC(=C(C=C6)OC)NS(=O)(=O)C)C(=NC=N5)N
/////
RP 6530, Tenalisib
(S)-2-(l-(9H-purin-6-ylamino)propyl)-3-(3-fluorophenyl)-4H-chromen-4-one (Compound A1 is RP 6530).
RP 6530
RP 6530, RP6530, RP-6530
Tenalisib
RP6530-1401, NCI-2015-01804, 124584, NCT02567656
(S)-2-(l-(9H-purin-6-ylamino)propyl)-3-(3-fluorophenyl)-4H-chromen-4-one
3-(3-fluorophenyl)-2-[(1S)-1-(7H-purin-6-ylamino)propyl]chromen-4-one
MW415.4, C23H18FN5O2
CAS 1639417-53-0, 1693773-94-2
RP6530 demonstrated high potency against PI3Kδ (IC50 =24.5 nM) and γ (IC50 = 33.2 nM) enzymes with selectivity over α (>300-fold) and β (>100-fold) isoforms. Cellular potency was confirmed in target-specific assays, namely anti-FcεR1-(EC50=37.8 nM) or fMLP (EC50 = 39.0 nM) induced CD63 expression in human whole blood basophils, LPS induced CD19+ cell proliferation in human whole blood (EC50=250 nM), and LPS induced CD45R+ cell proliferation in mouse whole blood (EC50=101 nM).
A PI3K inhibitor potentially for the treatment of hematologic malignancies.
An inhibitor of phosphoinositide-3 kinase (PI3K) δ/γ isoforms and anti-cellular proliferation agent for treatment of hematol. malignancies
Rhizen Pharmaceuticals is developing RP-6530, a PI3K delta and gamma dual inhibitor, for the potential oral treatment of cancer and inflammation In November 2013, a phase I trial in patients with hematologic malignancies was initiated in Italy ]\. In September 2015, a phase I/Ib study was initiated in the US, in patients with relapsed and refractory T-cell lymphoma. At that time, the study was expected to complete in December 2016
PATENTS……..WO 11/055215 , WO 12/151525.
Inventors
| Inventors | Meyyappan Muthuppalaniappan, Srikant Viswanadha, Govindarajulu Babu, Swaroop Kumar V.S. Vakkalanka, |
| Incozen Therapeutics Pvt. Ltd., Rhizen Pharmaceuticals Sa |
- Antineoplastics; Small molecules
- Mechanism of Action Phosphatidylinositol 3 kinase delta inhibitors; Phosphatidylinositol 3 kinase gamma inhibitors
- Phase I Haematological malignancies
- Preclinical Multiple myeloma
| Swaroop K. V. S. Vakkalanka, | |
| COMPANY | Rhizen Pharmaceuticals Sa |
https://clinicaltrials.gov/ct2/show/NCT02017613
PI3K delta/gamma inhibitor RP6530 An orally active, highly selective, small molecule inhibitor of the delta and gamma isoforms of phosphoinositide-3 kinase (PI3K) with potential immunomodulating and antineoplastic activities. Upon administration, PI3K delta/gamma inhibitor RP6530 inhibits the PI3K delta and gamma isoforms and prevents the activation of the PI3K/AKT-mediated signaling pathway. This may lead to a reduction in cellular proliferation in PI3K delta/gamma-expressing tumor cells. In addition, this agent modulates inflammatory responses through various mechanisms, including the inhibition of both the release of reactive oxygen species (ROS) from neutrophils and tumor necrosis factor (TNF)-alpha activity. Unlike other isoforms of PI3K, the delta and gamma isoforms are overexpressed primarily in hematologic malignancies and in inflammatory and autoimmune diseases. By selectively targeting these isoforms, PI3K signaling in normal, non-neoplastic cells is minimally impacted or not affected at all, which minimizes the side effect profile for this agent. Check for active clinical trials using this agent. (NCI Thesaurus)
| Company | Rhizen Pharmaceuticals S.A. |
| Description | Dual phosphoinositide 3-kinase (PI3K) delta and gamma inhibitor |
| Molecular Target | Phosphoinositide 3-kinase (PI3K) delta ; Phosphoinositide 3-kinase (PI3K) gamma |
| Mechanism of Action | Phosphoinositide 3-kinase (PI3K) delta inhibitor; Phosphoinositide 3-kinase (PI3K) gamma inhibitor |
| Therapeutic Modality | Small molecule |
Dual PI3Kδ/γ Inhibition By RP6530 Induces Apoptosis and Cytotoxicity In B-Lymphoma Cells
RP6530 is a potent and selective dual PI3Kδ/γ inhibitor that inhibited growth of B-cell lymphoma cell lines with a concomitant reduction in the downstream biomarker, pAKT. Additionally, the compound showed cytotoxicity in a panel of lymphoma primary cells. Findings provide a rationale for future clinical trials in B-cell malignancies.

PI3K Dual Inhibitor (RP-6530)
| Therapeutic Area | Respiratory , Oncology – Liquid Tumors , Rheumatology | Molecule Type | Small Molecule |
|---|---|---|---|
| Indication | Peripheral T-cell lymphoma (PTCL) , Non-Hodgkins Lymphoma , Asthma , Chronic Obstructive Pulmonary Disease (COPD) , Rheumatoid Arthritis | ||
| Development Phase | Phase I | Rt. of Administration | Oral |
Description
Rhizen is developing dual PI3K gamma/delta inhibitors for liquid tumors and inflammatory conditions.
Mechanism of Action
While alpha and beta isoforms are ubiquitous in their distribution, expression of delta and gamma is restricted to circulating hematogenous cells and endothelial cells. Unlike PI3K-alpha or beta, mice lacking expression of gamma or delta do not show any adverse phenotype indicating that targeting of these specific isoforms would not result in overt toxicity. Dual delta/gamma inhibition is strongly implicated as an intervention strategy in allergic and non-allergic inflammation of the airways and other autoimmune diseases. Scientific evidence for PI3K-delta and gamma involvement in various cellular processes underlying asthma and COPD stems from inhibitor studies and gene-targeting approaches. Also, resistance to conventional therapies such as corticosteroids in several COPD patients has been attributed to an up-regulation of the PI3K delta/gamma pathway. Disruption of PI3K-delta/gamma signalling therefore provides a novel strategy aimed at counteracting the immuno-inflammatory response. Due to the pivotal role played by PI3K-delta and gamma in mediating inflammatory cell functionality such as leukocyte migration and activation, and mast cell degranulation, blocking these isoforms may also be an effective strategy for the treatment of rheumatoid arthritis as well.
Given the established criticality of these isoforms in immune surveillance, inhibitors specifically targeting the delta and gamma isoforms would be expected to attenuate the progression of immune response encountered in airway inflammation and rheumatoid arthritis.

Clinical Trials
Rhizen has identified an orally active Lead Molecule, RP-6530, that has an excellent pre-clinical profile. RP-6530 is currently in non-GLP Tox studies and is expected to enter Clinical Development in H2 2013.
In December 2013, Rhizen announced the start of a Phase I clinical trial. The study entitled A Phase-I, Dose Escalation Study to Evaluate Safety and Efficacy of RP6530, a dual PI3K delta /gamma inhibitor, in patients with Relapsed or Refractory Hematologic Malignancies is designed primarily to establish the safety and tolerability of RP6530. Secondary objectives include clinical efficacy assessment and biomarker response to allow dose determination and potential patient stratification in subsequent expansion studies.
Partners by Region
Rhizen’s pipeline consists of internally discovered (with 100% IP ownership) novel small molecule programs aimed at high value markets of Oncology, Immuno-inflammtion and Metabolic Disorders. Rhizen has been successful in securing critical IP space in these areas and efforts are on for further expansion in to several indications. Rhizen seeks partnerships to unlock the potential of these valuable assets for further development from global pharmaceutical partners. At present global rights on all programs are available and Rhizen is flexible to consider suitable business models for licensing/collaboration.
In 2012, Rhizen announced a joint venture collaboration with TG Therapeutics for global development and commercialization of Rhizen’s Novel Selective PI3K Kinase Inhibitors. The selected lead RP5264 (hereafter, to be developed as TGR-1202) is an orally available, small molecule, PI3K specific inhibitor currently being positioned for the treatment of haematological malignancies.
PATENT
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2014195888Intermediates
Intermediate 1: 3-(3-fluorophenyl)-2-(l-hydroxypropyl)-4H-chromen-4-one: To a solution of 2-(l-bromopropyl)-3-(3-fluorophenyl)-4H-chromen-4-one1 (8.80 g, 24.36 mmol ) in DMSO (85 ml), n-butanol (5 ml) was added and heated to 120° C for 3h. The reaction mixture was cooled to room temperature (RT), quenched with water and extracted with ethyl acetate. The organic layer was dried over sodium sulphate and concentrated under reduced pressure. The crude product was purified by column chromatography with ethyl acetate: petroleum ether to afford the title compound as a yellow solid (2.10 g, 29 %) which was used without further purification in next step.
Intermediate 2: 3-(3-fluorophenyl)-2-propionyl-4H-chromen-4-one: DMSO (1.90 ml, 26.82 mmol) was added to dichloromethane (70 ml) and cooled to -78°C. Oxalyl chloride (1.14 ml, 13.41 mmol) was then added. After 10 minutes, intermediate 1 (2.00 g, 6.70 mmol) in dichloromethane (20 ml) was added dropwise and stirred for 20 min. Triethylamine (7 ml) was added and stirred for lh. The reaction mixture was quenched with water and extracted with dichloromethane. The organic layer was dried over sodium sulphate and concentrated under reduced pressure. The crude product was purified by column chromatography with ethyl acetate: petroleum ether to afford the title compound as a yellow liquid (1.20 g, 60%) which was used as such in next step.
Intermediate 3: (+)/(-)-3-(3-fluorophenyl)-2-(l-hydroxypropyl)-4H-chromen-4-one :
To a solution of intermediate 2 (0.600 g, 2.02 mmol) in DMF (7.65 ml) under nitrogen purging, formic acid : trietylamine 5 : 2 azeotrope (1.80 ml) was added followed by [(S,S)tethTsDpenRuCl] (3.0 mg). The reaction mixture was heated at 80°C for 1.5 hours under continuous nitrogen purging. The reaction mixture was quenched with water, extected with ethyl acetate, dried over sodium sulphate and concentrated. The crude product was purified by column chromatography with ethyl acetate: petroleum ether to afford the title compound as a yellow solid (0.450 g, 74%). Mass: 299.0 (M+).
Enantiomeric excess: 78%, enriched in the late eluting isomer (retention time: 9.72 min.) as determined by HPLC on a chiralpak AD-H column.
Intermediate 4: (+)/(-)-3-(3-fluorophenyl)-2-(l-hydroxypropyl)-4H-chromen-4-one :
The title compound was obtained as yellow solid (0.500 g, 83%) by using a procedure similar to the one described for intermediate 3, using intermediate 2 (0.600 g, 2.02 mmol), DMF (7.65 ml), formic acid : trietylamine 5 : 2 azeotrope (1.80 ml) and [(R,R)tethTsDpenRuCl] (3.0 mg). Mass: 298.9 (M+). Enantiomeric excess: 74.8%, enriched in the fast eluting isomer (retention time: 8.52 min.) as determined by HPLC on a chiralpak AD-H column.
Intermediate 5: (R)-3-(3-fluorophenyl)-2-(l-hydroxypropyl)-4H-chromen-4-one:
Step 1 : (R)-2-(l-(benzyloxy)propyl)-3-(3-fluorophenyl)-4H-chromen-4-one: To 2-(3-fluorophenyl)-l-(2-hydroxyphenyl)ethanone (2.15 g, 9.36 mmol ), in dichloromethane ( 20 ml), HATU (4.27 g, 11.23 mmol), R-(+)2-benzyloxybutyric acid (2.00 g, 10.29 mmol) were added and stirred for lOmin, then triethylamine (14.0 ml, 101.1 mmol) was added dropwise and stirred at RT for 24h. The reaction mixture was quenched with water, extracted with dichloromethane, dried over sodium sulphate and concentrated under reduced pressure. The crude product was purified by column chromatography with ethyl acetate: petroleum ether to afford the title compound as yellow solid (1.65 g, 45%). JH-NMR (δ ppm, CDC13, 400 MHz): 8.24 (dd, / = 7.9,1.5 Hz, 1H), 7.74 (dt, / = 7.1,1.7 Hz, 1H), 7.58 (dd, / = 8.3,0.4 Hz, 1H), 7.44-7.06 (m, 10H), 4.51 (d, / = 7.8 Hz, 1H), 4.34 (d, / = 7.8 Hz, 1H), 4.25 (dd, / = 7.8,6.2 Hz, 1H), 2.17-1.90 (m, 2H), 0.95 (t, / = 7.5 Hz, 3H). Mass: 389.0 (M+).
Step 2: (R)-3-(3-fluorophenyl)-2-(l-hydroxypropyl)-4H-chromen-4-one : To (R)-2-(l-(benzyloxy)propyl)-3-(3-fluorophenyl)-4H-chromen-4-one (1.50 g, 3.86 mmol) in dichloromethane (15 ml) cooled to 0°C and aluminium chloride (1.00 g, 7.72 mmol) was added portion wise and stirred at RT for 6h. The reaction mixture was quenched with 2N HC1 solution, extracted with dichloromethane, dried over sodium sulphate and concentrated under reduced pressure. The crude product was purified by column chromatography with ethyl acetate: petroleum ether to afford the title compound as yellow solid (0.552 g, 48%).‘ JH-NMR (δ ppm, CDC13, 400 MHz): ‘ 8.24 (dd, / = 8.0,1.6 Hz, 1H), 7.72 (m, , 1H), 7.52 (dd, / = 8.4,0.5 Hz, 1H), 7.44 (m, 2H), 7.12-7.01(m,3H), 4.49 (t, / = 7.0 Hz, 1H), 1.94 (m, 2H), 0.93 (t, / = 7.5 Hz, 3H). Mass: (299.0(M+). Purity: 96.93%.
25[a] D -14.73 (c = 1, CHCI3). Enantiomeric excess: 85.92%, enriched in the fast eluting isomer (retention time: 8.57 min.) as determined by HPLC on a chiralpak AS-3R column.
Compound A
(RS)- 2-(l-(9H-purin-6-ylamino)propyl)-3-(3-fluorophenyl)-4H-chromen-4-one
To a solution of intermediate 1 (2.50 g, 8.41 mmol) in THF (25 ml), tert-butyl 9-trityl-9H-purin-6-ylcarbamate (4.81 g, 10.09 mmol) and triphenylphosphine (3.31 g, 12.62 mmol) were added and stirred at RT for 5 min. Diisopropylazodicarboxylate (2.5 ml, 12.62 mmol) was added and stirred at RT for 2h. The reaction mixture was concentrated and column chromatographed with ethyl acetate : petroleum ether to afford a yellow coloured intermediate. To the intermediate, dichloromethane (65 ml) and trifluoroacetic acid (7.9 ml) were added and the resulting mixture was stirred at RT for 12 h. The reaction mixture was then basified with aqueous sodium bicarbonate solution, extracted with dichloromethane and dried over sodium sulphate. The crude product was purified by column chromatography with methanol: dichloromethane to afford the title compound as pale-brown solid (1.05 g, 30 %). MP: 148-150°C. Mass: 415.6 (M+).
Compound Al
(S)-2-(l-(9H-purin-6-ylamino)propyl)-3-(3-fluorophenyl)-4H-chromen-4-one
Method A: To a solution of intermediate 3 (0.250 g, 0.838 mmol) in THF (5ml), tert-butyl 9-trityl-9H-purin-6-ylcarbamate (0.479 g, 1.00 mmol) and triphenylphosphine (0.329 g, 1.25 mmol) were added and the resulting mixture was stirred at RT for 5 min. Diisopropylazodicarboxylate (0.25 ml, 1.25 mmol) was then added and stirred at RT for 12 h. The reaction mixture was concentrated and column chromatographed with ethyl acetate: pet.ether to afford the yellow coloured intermediate. To the intermediate in dichloromethane (6 ml), trifluoroacetic acid (1.2 ml) was added stirred at RT for 12 h. The reaction mixture was basified with aqueous sodium bicarbonate solution, extracted with dichloromethane and dried over sodium sulphate. The crude product was purified by column chromatography with methanol: dichloromethane to afford the title compound as an off-white solid (0.015 g, 4 %). MP: 137-140°C. JH-NMR (δ ppm, DMSO- , 400 MHz): 12.94 (s, 1H), 8.12-8.10 (m, 4H), 7.84-7.80 (m, 1H), 7.61 (d, / = 8.3 Hz, 1H), 7.50-7.41 (m, 2H), 7.28-7.18 (m, 3H), 5.20-5.06 (m, 1H), 2.10-1.90 (m, 2H), 0.84 (t, / = 3.7 Hz, 3H). Enantiomeric excess: 77.4% as determined by HPLC on a chiralpak AD-H column, enriched in the fast eluting isomer (retention time = 7.90 min.).
Method B : To a solution of intermediate 5 (2.60 g, 8.68 mmol) in THF (52 ml), tert-butyl 9-trityl-9H-purin-6-ylcarbamate (4.96 g, 10.42 mmol) and triphenylphosphine (2.76 g, 13.03 mmol) were added and the resulting mixture was stirred at RT for 5 min. Dusopropylazodicarboxylate (0.25 ml, 1.25 mmol) was then added and stirred at RT for 12 h. The reaction mixture was concentrated and column chromatographed with ethyl acetate: petroleum ether to afford the yellow coloured intermediate. To the intermediate in dichloromethane (55 ml), trifluoroacetic acid (14.2 ml) was added and stirred at RT for 12 h. The reaction mixture was basified with aqueous sodium bicarbonate solution, extracted with dichloromethane and dried over sodium sulphate. The crude product was purified by column chromatography with methanol: dichloromethane to afford the title compound as pale-yellow solid (1.00 g, 27 %). MP: 168-170°C. Mass: 416.5(M++1) Enantiomeric excess: 86.5% as determined by HPLC on a chiralpak AD-H column, enriched in the fast eluting isomer (retention time = 7.90 min.).
Method C : The title compound was separated by preparative SFC conditions from Compound A (1.090 g) on a CHIRALPAK AY-H column (250 x 30 mm; 5μπι) using methanol : C(¾ (35:65) as the mobile phase at a flow rate of 80 g / min. Off-white solid (0.378 g). e.e. 100%. Rt: 2.37 min. Mass: 416.1(M++1). MP: 149-152°C.
CAUTION ethyl compd below, NOT THE PRODUCT
Example 47
(S)-2-(l-(9H-purin-6-yIamino) ethyl)-3-(3-fluorophenyl)-4H-chromen-4-one
[428] To a solution of intermediate 65 (2.0g, 8.68 mmoles) in dichloromethane (20ml), triethylamine (3.6ml, 26.06 mmoles) was added followed by N-Boc-Alanine (1.97g, 10.42 mmoles). To this mixture HATU (6.6g, 17.37 mmoles) was added and stirred at RT for 12h. The reaction mixture was quenched by the addition of water and extracted with dichloromethane. The organic layer was dried over sodium sulphate and concentrated under reduced pressure. The crude product was purified by column chromatography with ethyl acetate: petroleum ether to afford the isoflavone intermediate (1.70g). To a solution of this intermediate (1.7g) in dichloromethane (20ml), trifluoroacetic acid (3 ml) was added and stirred at RT for 2h. The reaction mixture was concentrated, basified with sodium bicarbonate solution, extracted with ethyl acetate. The organic layer was dried over sodium sulphate and concentrated under reduced pressure to afford the amine intermediate (0.641 g). To a solution of this amine intermediate (0.30g, 1.05 mmoles) in tert-butanol (6ml), N, N- diisopropylethylamine (0.36ml, 2.17 mmoles) and 6-bromopurine (0.168g, 0.847 mmoles) were added and refluxed for 24h. The reaction mixture was concentrated, diluted with water, extracted with ethyl acetate. The organic layer was dried over sodium sulphate and concentrated under reduced pressure. The crude product was purified by column chromatography with methanol: ethyl acetate to afford the title compound as off-white solid (0.041g, 10% yield). MP: 135-138 °C. Ή-NMR (δ ppm, DMSO-D6, 400 MHz): δ 12.95(s,lH), 8.15(t, / = 6.8Hz, 1H), 8.11(s, 1H), 8.08(s, 1H), 8.03(d, J = 7.8 Hz, 1H), 7.81(t ,J = 7.3Hz, 1H), 7.60 (d, J = 8.3Hz, 1H), 7.49 (t, J = 7.3Hz, 2H), 7.25(m,3H), 5.19(br m, 1H), 1.56(d, J = 6.9Hz,3H). Mass: 402.18(M+ +1).
This scheme provides a synthetic route for the preparation of compound of formula wherein all the variables are as described herein in above
15 14 10 12 12a
Abstract 2704: RP6530, a dual PI3K δ/γ inhibitor, potentiates ruxolitinib activity in the JAK2-V617F mutant erythroleukemia cell lines
– Author Affiliations
- 1Rhizen Pharmaceuticals SA, Fritz-Courvoisier 40, Switzerland;
- 2Incozen Therapeutics Pvt. Ltd., Hyderabad, India.
Abstract
Background: Myelofibrosis (MF) represents a life-threatening neoplasm that manifests particularly in the elderly population and is characterized by bone marrow fibrosis and extramedullary hematopoeisis. While ruxolitinib, a JAK1/2 inhibitor, has recently been approved by the USFDA for its disease modifying potential in MF patients, it is still not considered as a curative option. Targeting another kinase such as PI3K, downstream of JAK, could therefore be a more efficient way of treating myelofibrotic neoplasms. RP6530 is a novel, potent, and selective PI3K δ/γ inhibitor that demonstrated high potency against PI3Kδ (IC50 = 25 nM) and γ (IC50 = 33 nM) enzymes with selectivity over α (>300-fold) and β (>100-fold) isoforms. The objective of this study was to evaluate the effect of a combination of ruxolitinib and RP6530 in the JAK2-V617F mutant Human Erythroleukemia (HEL) cell line.
Methods: Passive resistance was conferred by incubating HEL cells with increasing concentrations of ruxolitinib over an 8-10-week period. Endogenous JAK2, PI3Kδ, PI3Kδ, and pAKT were estimated by Western Blotting. RP6530, ruxolitinib, and the combination of RP6530 + Ruxolitinib were tested for their effect on viability and apoptosis. Cell viability was assessed by a MTT assay. Induction of apoptosis was analyzed by Annexin V/PI staining.
Results: Resistance to ruxolitinib was confirmed by a right-ward shift in EC50 of ruxolitinib in a HEL cell proliferation assay (0.82 μM Vs. 12.2 μM). Endogeous pAKT expression was 3.7-fold higher in HEL-RR compared to HEL-RS cells indicating activation of the AKT signaling pathway. While single-agent activity of RP6530 was modest (33-46% inhibition @ 10 μM) in both HEL-RS and HEL-RR cells, addition of 10 μM RP6530 to ruxolitinib was synergistic resulting in a near-complete inhibition of proliferation (>90% for HEL-RS and >70% for HEL-RR). While the order of addition did not affect the potency of RP6530, addition of 5 μM RP6530, 4 h prior to the addition of ruxolitinib resulted in a significant reduction in EC50 of ruxolitinib (5.8 μM) in HEL-RR cells. On lines with cell proliferation data, incubation of 10 μM RP6530 with ruxolitinib for 72 h increased the percent of apoptotic cells (55% in HEL-RS and 37% in HEL-RR) compared to either agent alone (16-27% in HEL-RS and 17-21% in HEL-RR).
Conclusions: Ruxolitinib resistance in the V617F JAK-2 mutant HEL cells is accompanied by an increase in pAKT expression. Inhibition of pAKT via the addition of RP6530, a dual PI3K δ/γ inhibitor, resulted in a reversal of ruxolitinib resistance. Complementary activity was also observed in HEL-RS cells indicating that a combination of ruxolitinib and RP6530 could have a positive bearing on the clinical outcome in MF patients.
Citation Format: Swaroop Vakkalanka, Seeta Nyayapathy, Srikant Viswanadha. RP6530, a dual PI3K δ/γ inhibitor, potentiates ruxolitinib activity in the JAK2-V617F mutant erythroleukemia cell lines. [abstract]. In: Proceedings of the 106th Annual Meeting of the American Association for Cancer Research; 2015 Apr 18-22; Philadelphia, PA. Philadelphia (PA): AACR; Cancer Res 2015;75(15 Suppl):Abstract nr 2704. doi:10.1158/1538-7445.AM2015-2704
- 01 Sep 2015 Phase-I clinical trials in Haematological malignancies (Second-line therapy or greater) in USA (PO) (NCT02567656)
- 18 Nov 2014 Preclinical trials in Multiple myeloma in Switzerland (PO) prior to November 2014
- 18 Nov 2014 Early research in Multiple myeloma in Switzerland (PO) prior to November 2014
| WO2011055215A2 | Nov 3, 2010 | May 12, 2011 | Incozen Therapeutics Pvt. Ltd. | Novel kinase modulators |
| WO2012151525A1 | May 4, 2012 | Nov 8, 2012 | Rhizen Pharmaceuticals Sa | Novel compounds as modulators of protein kinases |
| WO2013164801A1 | May 3, 2013 | Nov 7, 2013 | Rhizen Pharmaceuticals Sa | Process for preparation of optically pure and optionally substituted 2- (1 -hydroxy- alkyl) – chromen – 4 – one derivatives and their use in preparing pharmaceuticals |
| US20110118257 | May 19, 2011 | Rhizen Pharmaceuticals Sa | Novel kinase modulators | |
| US20120289496 | May 4, 2012 | Nov 15, 2012 | Rhizen Pharmaceuticals Sa | Novel compounds as modulators of protein kinases |
| WO 2011055215 |
WO2015175966
WO2015051252
-
The Distillery: Therapeutics 09/04/2014
BC Innovations, TherapeuticsIndication Target/marker/pathway Summary Licensing status Publication and contact information Cardiovascular disease Intimal hyperplasia Phosphoinositide 3-kinase-g (PI3Kg) Rodent studies suggest inhibiting … -
BC Innovations, Targets & MechanismsTargets & Mechanisms: PI3K inhibition: solid immunotherapy Table 1. A peek at PI3K inhibitors. According to a study in Nature by Ali et al., inhibition of phosphoinositide 3-kinase-d (PI3Kd) or the PI3K catalytic …
-
RP6530: Phase I started 12/23/2013
Week in Review, Clinical StatusRhizen Pharmaceuticals S.A., La Chaux-de-Fonds, Switzerland Product: RP6530 Business: Cancer Molecular target: Phosphoinositide 3-kinase (PI3K) delta; Phosphoinositide 3-kinase (PI3K) gamma Description: Dual … -
Rhizen preclinical data 07/01/2013
Week in Review, Preclinical ResultsRhizen Pharmaceuticals S.A., La Chaux-de-Fonds, Switzerland Product: RP6530 Business: Cancer Indication: Treat B cell lymphoma In vitro, 2-7 M RP6530 led to a >50% dose-dependent inhibition in growth of immortalized …
/////
c1cccc4c1C(/C(=C(/[C@H](CC)Nc3c2c(ncn2)ncn3)O4)c5cc(ccc5)F)=O
CCC(C1=C(C(=O)C2=CC=CC=C2O1)C3=CC(=CC=C3)F)NC4=NC=NC5=C4NC=N5
Alembic Pharma advances 1% on Rhizen-Novartis license agreement
Swiss subsidiary Rhizen Pharmaceuticals S.A. entered into an exclusive, worldwide license agreement with Novartis for the development and commercialization of Rhizen’s, inhaled dual Pl3K-delta gamma inhibitor and its closely related compounds for various indications.
read

Rhizen Pharmaceuticals Announces Exclusive Worldwide License Agreement for the Development and Commercialization of a Dual PI3K-delta gamma Inhibitor
| Source: Rhizen Pharmaceuticals SA
La Chaux-de-Fonds, Switzerland , Dec. 09, 2015 (GLOBE NEWSWIRE) — Rhizen Pharmaceuticals S.A. announced today that they have entered into an exclusive, worldwide license agreement with Novartis for the development and commercialization of Rhizen’s, inhaled dual PI3K-delta gamma inhibitor and its closely related compounds for various indications.
Under the terms of the agreement, Rhizen will receive an upfront payment and is eligible to receive development, regulatory and sales milestones payments. In addition Rhizen is also eligible to receive tiered royalties on annual nets sales.
The lead compound is a novel, potent, and selective dual PI3K-delta gamma inhibitor with demonstrated anti-inflammatory and immuno-modulatory activity in pre-clinical systems and models representative of respiratory diseases. With a favorable ADME and PK profile and high therapeutic index in animals, the inhaled dual PI3K-delta gamma inhibitor holds promise in the treatment of human airway disorders.
About Rhizen Pharmaceuticals S.A.:
Rhizen Pharmaceuticals is an innovative, clinical-stage biopharmaceutical company focused on the discovery and development of novel therapeutics for the treatment of cancer, immune and metabolic disorders. Since its establishment in 2008, Rhizen has created a diverse pipeline of proprietary drug candidates targeting several cancers and immune associated cellular pathways. Rhizen is headquartered in La-Chaux-de-Fonds, Switzerland. For additional information, please visit Rhizen’s website, www.rhizen.com.
info@rhizen.com

RP 6503
//////
LIK 066, Licogliflozin diprolinate
Licogliflozin
LIK 066
Licogliflozin diprolinate
LIK-066, a new flozin on the horizon
C23 H28 O7 . 2 C6 H11 N O, 642.7795, 1 :2 co-crystal of Example 62 : L-proline. A melting point 176°C…WO2011048112
CAS 1291095-45-8, (1S)-1,5-anhydro-1-C-[3-[(2,3-dihydro-1,4-benzodioxin-6-yl)methyl]-4-ethylphenyl]-D-glucitol (1:1) WITH L-Proline, compd., 1:1 Proline Co-crvstal , 1:1 Proline Co-crvstal …..…WO2011048112
CAS BASE 1291094-73-9, 416.46, C23 H28 O7
(1S)-1,5-Anhydro-1-[3-(2,3-dihydro-1,4-benzodioxin-6-ylmethyl)-4-ethylphenyl]-D-glucitol bis[1-[(2S)-pyrrolidin-2-yl]ethanone]
(2S,3R,4R,5S,6R)-2-[3-(2,3-Dihydro-benzo[1,4]dioxin-6-ylmethyl)-4- ethyl-phenyl]-6-hydroxymethyl-tetrahydro-pyran-3,4,5-triol
Sodium glucose transporter-2 inhibitor
SGLT 1/2 inhibitor
Novartis Ag innovator
Clinical trial……..https://clinicaltrials.gov/ct2/show/NCT01915849
https://clinicaltrials.gov/ct2/show/NCT02470403
- 10 Jun 2015 Novartis initiates enrolment in a phase II trial for Type 2 diabetes mellitus in USA (NCT02470403)
- 02 Apr 2014 Novartis terminates a phase II trial in Type-2 diabetes mellitus in USA, Poland, Argentina, Hungary, Puerto Rico and South Africa (NCT01824264)
- 01 Jan 2014 Novartis completes a phase II trial in Type 2 diabetes mellitus in USA (NCT01915849)
Licogliflozin, a SGLT-1/2 inhibitor, is in phase II clinical development at Novartis for the treatment of metabolic disorders, for the treatment of heart failure in patients with type 2 diabetes, for the treatment of obesity and for the treatment of polycystic ovary syndrome (PCOS) in overweight and obese women. Phase II trials for the treatment of type 2 diabetes had been discontinued.
EMA/415156/2014 European Medicines Agency decision P/0183/2014 of 24 July 2014 on the agreement of a paediatric investigation plan and on the granting of a deferral and on the granting of a waiver for (S)-Pyrrolidine-2-carboxylic acid compound with (2S,3R,4R,5S,6R)-2-(3-((2,3- dihydrobenzo[b][1,4]dioxin-6-yl)methyl)-4-ethylphenyl)-6-(hydroxymethyl)tetrahydro-2H-pyran3,4,5-triol (2:1) (LIK066) (EMEA-001527-PIP01-13) in accordance with Regulation (EC) No 1901/2006 of the European Parliament and of the Council
1. Opinion of the Paediatric Committee on the agreement of a Paediatric Investigation Plan and a deferral and a waiver. 2014, EMEA-001527-PIP01-13 (here) [ Novartis revealed the IUPAC name here].
Where name is given
http://www.who.int/medicines/publications/druginformation/issues/DrugInformation2017_Vol31-4/en/


http://www.who.int/medicines/publications/druginformation/issues/PL_118.pdf?ua=1
SEE ALSO

LIK-066 is in phase II clinical studies at Novartis for the treatment of type 2 diabetes.
In June 2014, the EMA’s PDCO adopted a positive opinion on a pediatric investigation plan (PIP) for LIK-066 for type 2 diabetes
Diabetes mellitus is a metabolic disorder characterized by recurrent or persistent hyperglycemia (high blood glucose) and other signs, as distinct from a single disease or condition. Glucose level abnormalities can result in serious long-term complications, which include cardiovascular disease, chronic renal failure, retinal damage, nerve damage (of several kinds), microvascular damage and obesity.
Type 1 diabetes, also known as Insulin Dependent Diabetes Mellitus (IDDM), is characterized by loss of the insulin-producing β-cells of the islets of Langerhans of the pancreas leading to a deficiency of insulin. Type-2 diabetes previously known as adult- onset diabetes, maturity-onset diabetes, or Non-Insulin Dependent Diabetes Mellitus (NIDDM) – is due to a combination of increased hepatic glucose output, defective insulin secretion, and insulin resistance or reduced insulin sensitivity (defective responsiveness of tissues to insulin). Chronic hyperglycemia can also lead to onset or progression of glucose toxicity characterized by decrease in insulin secretion from β-cell, insulin sensitivity; as a result diabetes mellitus is self-exacerbated [Diabetes Care, 1990, 13, 610].
Chronic elevation of blood glucose level also leads to damage of blood vessels. In diabetes, the resultant problems are grouped under “microvascular disease” (due to damage of small blood vessels) and “macro vascular disease” (due to damage of the arteries). Examples of microvascular disease include diabetic retinopathy, neuropathy and nephropathy, while examples of macrovascular disease include coronary artery disease, stroke, peripheral vascular disease, and diabetic myonecrosis.
Diabetic retinopathy, characterized by the growth of weakened blood vessels in the retina as well as macular edema (swelling of the macula), can lead to severe vision loss or blindness. Retinal damage (from microangiopathy) makes it the most common cause of blindness among non-elderly adults in the US. Diabetic neuropathy is characterized by compromised nerve function in the lower extremities. When combined with damaged blood vessels, diabetic neuropathy can lead to diabetic foot. Other forms of diabetic neuropathy may present as mononeuritis or autonomic neuropathy. Diabetic nephropathy is characterized by damage to the kidney, which can lead to chronic renal failure, eventually requiring dialysis. Diabetes mellitus is the most common cause of l adult kidney failure worldwide. A high glycemic diet (i.e., a diet that consists of meals that give high postprandial blood sugar) is known to be one of the causative factors contributing to the development of obesity.
Type 2 diabetes is characterized by insulin resistance and/or inadequate insulin secretion in response to elevated glucose level. Therapies for type 2 diabetes are targeted towards increasing insulin sensitivity (such as TZDs), hepatic glucose utilization (such as biguanides), directly modifying insulin levels (such as insulin, insulin analogs, and insulin secretagogues), increasing increttn hormone action (such as exenatide and sitagliptin), or inhibiting glucose absorption from the diet (such as alpha glucosidase inhibitors) [Nature 2001 , 414, 821-827],
Glucose is unable to diffuse across the cell membrane and requires transport proteins. The transport of glucose into epithelial cells is mediated by a secondary active cotransport system, the sodium-D-glucose co-transporter (SGLT), driven by a sodium- gradient generated by the Na+/K+-ATPase. Glucose accumulated in the epithelial cell is further transported into the blood across the membrane by facilitated diffusion through GLUT transporters [Kidney International 2007, 72, S27-S35].
SGLT belongs to the sodium/glucose co-transporter family SLCA5. Two different SGLT isoforms, SGLT1 and SGLT2, have been identified to mediate renal tubular glucose reabsorption in humans [Curr. Opinon in Investigational Drugs (2007): 8(4), 285-292 and references cited herein]. Both of them are characterized by their different substrate affinity. Although both of them show 59% homology in their amino acid sequence, they are functionally different. SGLT1 transports glucose as well as galactose, and is expressed both in the kidney and in the intestine, while SGLT2 is found exclusively in the S1 and S2 segments of the renal proximal tubule.
As a consequence, glucose filtered in the glomerulus is reabsorbed into the renal proximal tubular epithelial cells by SGLT2, a low-affinity/high-capacity system, residing on the surface of epithelial cell lining in S1 and S2 tubular segments. Much smaller amounts of glucose are recovered by SGLT1 , as a high-affinity/low-capacity system, on the more distal segment of the proximal tubule. In healthy human, more than 99% of plasma glucose that is filtered in the kidney glomerulus is reabsorbed, resulting in less than 1 % of the total filtered glucose being excreted in urine. It is estimated that 90% of total renal glucose absorption is facilitated by SGLT2; remaining 10 % is likely mediated by SGLT1 [J. Parenter. Enteral Nutr. 2004, 28, 364-371].
SGLT2 was cloned as a candidate sodium glucose co-transporter, and its tissue distribution, substrate specificity, and affinities are reportedly very similar to those of the low-affinity sodium glucose co-transporter in the renal proximal tubule. A drug with a mode of action of SGLT2 inhibition will be a novel and complementary approach to existing classes of medication for diabetes and its associated diseases to meet the patient’s needs for both blood glucose control, while preserving insulin secretion. In addition, SGLT2 inhibitors which lead to loss of excess glucose (and thereby excess calories) may have additional potential for the treatment of obesity.
Indeed small molecule SGLT2 inhibitors have been discovered and the anti-diabetic therapeutic potential of such molecules has been reported in literature [T-1095 (Diabetes, 1999, 48, 1794-1800, Dapagliflozin (Diabetes, 2008, 57, 1723-1729)].
SYNTHESIS
PATENT
WO 2011048112
https://www.google.com/patents/WO2011048112A1?cl=en
Gregory Raymond Bebernitz, Mark G. Bock, Dumbala Srinivas Reddy, Atul Kashinath Hajare, Vinod Vyavahare, Sandeep Bhausaheb Bhosale, Suresh Eknath Kurhade, Videsh Salunkhe, Nadim S. Shaikh, Debnath Bhuniya, P. Venkata Palle, Lili Feng, Jessica Liang,
Example 61-62:
Ex. 61
Example 61 : Acetic acid (2R,3R,4R,5S)-3,4,5-triacetoxy-6-[3-(2,3-dihydro- benzo[1 ,4]dioxin-6-ylmethyl)-4-ethyl-phenyl]-tetrahydro-pyran-2-ylmethyl ester
Step I: To a stirred solution of acetic acid (2R,3R,4R,5S)-3,4,5-triacetoxy-6-[4-bromo-3- (2,3-dihydro-benzo[1 ,4]dioxin-6-ylmethyl)-phenyl]-tetrahydro-pyran-2-ylmethyl ester (10.0 g, 15.74 mmol) in toluene (200 mL) was added tricyclohexylphosphine (1.76 g, 6.29 mmol), a solution of potassium phosphate tribasic (13.3 g, 62.9 mmol) in water (15 mL), and ethylboronic acid (3.4 g, 47.2 mmol). The reaction mixture was degassed for 45 min then palladium (II) acetate (529 mg, 2.3 mmol) was added. After refluxing overnight, the reaction mixture was cooled to room temperature, and water was added. The resulting mixture was extracted with ethyl acetate, (2 X 200 mL), washed with water and brine, then dried over sodium sulfate, concentrated and purified by column chromatography to furnish acetic acid (2R,3R,4R,5S)-3,4,5-triacetoxy-6-[3-(2,3-dihydro- benzo[1 ,4]dioxin-6-ylmethyl)-4-ethyl-phenyl]-tetrahydro-pyran-2-ylmethyl ester (5.4 g).
Example 62: (2S,3R,4R,5S,6R)-2-[3-(2,3-Dihydro-benzo[1,4]dioxin-6-ylmethyl)-4- ethyl-phenyl]-6-hydroxymethyl-tetrahydro-pyran-3,4,5-triol
Step II: To a stirred solution of acetic acid (2R,3R,4R,5S)-3,4,5-triacetoxy-6-[3-(2,3- dihydro-benzo[1 ,4]dioxin-6-ylmethyl)-4-ethyl-phenyl]-tetrahydro-pyran-2-ylmethyl ester (9.3 g, 15.9 mmol) in methanol:THF:water 3:2:1 (170 mL) was added lithium hydroxide (764 mg, 19.1 mmol). After stirring for 2 h at room temperature, the volatiles were evaporated under reduced pressure. The resulting residue was taken up in ethyl acetate (150 mL) and washed with brine (75 mL), brine containing 5 mL of 5% aqueous KHS04 (75 mL), and brine (20 mL) again, then dried over sodium sulfate and concentrated to furnish (2S,3R,4R,5S,6R)-2-[4-Cyclopropyl-3-(2,3-dihydro- benzo[1 ,4]dioxin-6-ylmethyl)-phenyl]-6-hydroxymethyl-tetrahydro-pyran-3,4,5-triol (6.59)
H NMR (400 MHz, CD3OD): δ 1.07 (t, J = 7.6 Hz, 3H), 2.57 (q, J = 7.6 Hz, 2H), 3.34- 3.50 (m, 4H), 3.68 (dd, J = 12.0, 5.6 Hz, 1 H), 3.85-3.91 (m, 3H), 4.08 (d, J = 9.6 Hz, 1 H), 4.17 (s, 4H), 6.53-6.58 (m, 2H), 6.68 (d, J – 8.4 Hz, 1 H), 7.15-7.25 (m, 3H).
MS (ES) m z 434.2 (M+18).
PICK UP IDEAS FROM HERE
Examples 57-58:
Ex. 57 Ex. 58
Step I: To a stirred solution of 2-bromo-5-iodobenzoic acid (25.0 g, 76.48 mmol) in dichloromethane (200 mL) was added oxalyl chloride (10.3 mL, 114.74 mmol) at 0 °C followed by D F (0.9 mL). After complete addition, the reaction mixture was stirred at room temperature for 3h. Volatiles were evaporated under reduced pressure to furnish 2-bromo-5-iodo-benzoyl chloride (26.4 g). The crude product was used for the next step immediately.
Step II: To a stirred solution of 2-bromo-5-iodo-benzoyl chloride (26.4 g, 76.56 mmol) in dichloromethane (250 mL) was added benzo(1 ,4)-dioxane (10.41 g, 76.26 mmol) at 0 °C. To this reaction mixture, AICI3 (40.78 g, 305.47 mmol) was added in portions. After stirring overnight at room temperature, the reaction mixture was poured into crushed ice. The resulting mixture was extracted with dichloromethane (500 mL X 2). The dichloromethane layers were combined and washed with water (200 mL), saturated aqueous sodium bicarbonate solution (200 mL X 2), and brine (200 mL), then dried over sodium sulfate and concentrated. The solid product was triturated with hexanes, and the triturated product was dried under vacuum to furnish (2-bromo-5-iodo-phenyl)-(2,3- dihydro-benzo[1 ,4]dioxin-6-yl)-methanone (30 g).
1H N R (400 MHz, DMSO-D6): δ 4.29-4.37 (m, 4H), 7.02 (d, J = 8.4 Hz, 1 H), 7.16 (d, J = 2.4 Hz, 1 H), 7.18-7.19 (m, 1 H), 7.53 (d, J = 8.4 Hz, 1 H), 7.77-7.81 (m, 1 H), 7.82 (d, J = 2.0 Hz, 1 H).
Step III: To a stirred solution of (2-bromo-5-iodo-phenyl)-(2,3-dihydro-benzo[1 ,4]dioxin- 6-yl)-methanone (30.0 g, 67.4 mmol) in trifluoroacetic acid (100 mL) was added triethylsilane (86.2 mL, 539.3 mmol) followed by triflic acid (6.0 mL, 67.42 mmol ) at room temperature. After stirring for 25 min at room temperature, volatiles were evaporated under reduced pressure. The resulting residue was taken up in ethyl acetate and washed with saturated aqueous sodium bicarbonate solution (200 mL X 2), water (200 mL), and brine (200 mL), then dried over sodium sulfate, concentrated and purified by silica gel column chromatography to furnish 6-(2-bromo-5-iodo-benzyl)-2,3- dihydro-benzo[1 ,4]dioxine (26.5 g). H NMR (400 MHz, DMSO-D6): δ 3.90 (s, 4H), 4.2 (s, 2H), 6.65 (dd, J = 8.4 Hz, J = 2.0 Hz, H), 6.68 (d, J = 2.0 Hz, 1 H), 6.77 (d, J = 8.4 Hz, H), 7.39 (d, J = 8.4 Hz, 1 H), 7.50 (dd, J = 8.4 Hz, J = 2.4 Hz 1 H), 7.67 (d, J = 2.8 Hz, 1 H).
Step IV: To a stirred solution of 6-(2-bromo-5-iodo-benzyl)-2,3-dihydro- benzo[1 ,4]dioxine (26.5 g, 61.47 mmol) in THF:toluene 2:1 (300 mL) was added 1.6 M solution of n-BuLi in hexanes (42.3 mL, 67.62 mmol) at -78 °C. The reaction mixture was stirred for 1 h, and then transferred to a stirred solution of 2,3,4,6-tetrakis-O- (trimethylsilyl)-D-glucopyranone (28.69 g, 61.47 mmol) in toluene (100 mL) at -78 °C. After stirring for 1 h, 0.6 N methanesulfonic acid in methanol (265 mL) was added dropwise and stirred the reaction mixture for 16 h at room temperature. Reaction was quenched by the addition of aq. NaHC03 solution (~75 mL) and extracted with ethyl acetate (250 mL X 3), dried over sodium sulfate, concentrated and purified by silica gel column chromatography to furnish (3R,4S,5S,6R)-2-[4-Bromo-3-(2,3-dihydro- benzo[1 ,4]dioxin-6-ylmethyl)-phenyl]-6-hydroxymethyl-2-methoxy-tetrahydro-pyran- 3,4,5-triol (28.4 g)
Example 57: [(2R,3R,4R,5S,6S)-3,4,5-triacetoxy-6-[4-bromo-3-(2,3-dihydro-1 ,4- benzodioxin-6-ylmethyl)phenyl]tetrahydropyran-2-yl]methyl acetate
Step V: To a stirred solution of (3R,4S,5S,6R)-2-[4-bromo-3-(2,3-dihydro- benzo[1 ,4]dioxin-6-ylmethyl)-phenyl]-6-hydroxymethyl-2-methoxy-tetrahydro-pyran-3,4,5- triol (28.4 g, 57.1 mmol) in acetonitrile-dichloromethane 1 :1 (250 mL) was added triethylsilane (36.5 mL, 228.4 mmol) and boron trifluoride diethyletharate complex (14.1 mL, 114.2 mmol) at 10 °C. After stirring for 4 h at 10°C, the reaction was quenched with saturated aqueous sodium bicarbonate (~ 100 mL). The organic layer was separated, and the aqueous layer was extracted with ethyl acetate (3 X 150 mL). The organic layers were combined and dried over sodium sulfate, concentrated to furnish (3R,4R,5S,6R)-2- [4-bromo-3-(2,3-dihydro-benzo[1 ,4]dioxin-6-ylmethyl)-phenyl]-6-hydroxymethyl- tetrahydro-pyran-3,4,5-triol (28.4 g). Crude product was used for next reaction without purification. Example 58: [(2R,3R,4R,5S,6S)-3,4,5-triacetoxy-6-[4-bromo-3-(2!3-dihydro-1,4- benzodioxin-6-ylmethyl)phenyl]tetrahydropyran-2-yl]methyl acetate Step V: To a stirred solution of (3R,4R,5S,6R)-2-[4-Bromo-3-(2,3-dihydro- benzo[ 1 ,4]dioxin-6-yl methyl)-phenyl]-6-hydroxymethyl-tetrahyd ro-pyran-3,4 , 5-triol (28.4 g, 60.81 mmol) in dichloromethane (300 mL) was added pyridine (40 mL, 486.5 mmol), acetic anhydride (50 mL, 486.5 mmol) and DMAP (740 mg, 6.08 mmol) at room temperature. After stirring for 2 h, volatiles were evaporated under reduced pressure. The resulting residue was taken up in ethyl acetate (500ml) and washed with 1 N HCI (200 mL X 2) followed by brine (200ml), then dried over sodium sulfate and
concentrated. The resulting crude compound was dissolved in ethanol (320 mL) at 65 °C and allowed to cool to room temperature while stirring. Light yellow solid formed was filtered and washed with cold ethanol (150 mL) followed by hexane (200 mL) to get acetic acid (2R,3R,4R,5S)-3,4,5-triacetoxy-6-[4-bromo-3-(2,3-dihydro-benzo[1 ,4]dioxin- 6-ylmethyl)-phenyl]-tetrahydro-pyran-2-ylmethyl ester powder (22.5 g, purity 98%).
COCRYSTAL
Example 75: 1:1 Proline Co-crvstal with f2S.3R.4R.5S.6R¾-2-r3-f2.3-Dihvdro- benzori.41dioxin-6-ylmethyl)-4-ethyl-phenvn-6-hvdroxymethyl-tetrahydro-pyran- 3.4.5-triol
(2S,3R,4R,5S,6R)-2-[3-(2,3-Dihydro-benzo[1 ,4]dioxin-6-ylmethyl)-4-ethyl- phenyl]-6-hydroxymethyl-tetrahydro-pyran-3,4,5-triol (Example 62) was completely amorphous initially but formed a crystalline complex with proline. This was confirmed by powder X-ray diffraction (PXRD) analysis. The stiochiometry of Example 62 and L- proline in the co-crystal prepared by method 1 was found to be 1 :1 by NMR
spectroscopy & HPLC. Characterization data for co-crystals of Example 62 and proline prepared by method 1 is shown in Table 3. Relative intensities of the most prominent powder x-ray diffraction peaks for co-crystals of Example 62 and proline are shown in Table 3A.
Table 3

Table 3A

3.70 15.78 18.36 25.18
9.68 10.68 18.88 36.33
11.07 21.21 20.42 69.29
14.26 14.81 21.18 27.94
14.80 22.97 22.50 12.25
15.40 4 98 23.78 33.08
16.12 8.45 24.56 6.92
16.59 18.78 25.79 21.69
17.31 100.0 27.46 8.90
17.60 20.35 31.97 7.65
17.98 47.20 32.46 5.98
1:1 Proline Co-crvstal
Example 77: 1:1 Proline Co-crvstal with (2S.3R.4R.5S.6Ri-2-f3-(2.3-Dihvdro- benzoh .41dioxin-6-ylmethvh-4-ethyl-phenvn-6-hvdroxymethyl-tetrahvdro-pyran- 3.4.5-triol
Method 2:
1 :1 Co-Crvstals of Example 62 with L-Proline
(2S,3R,4R,5S,6R)-2-[3-(2,3-Dihydro-benzo[1 ,4]dioxin-6-ylmethyl)-4-ethyl-phenyl]- 6-hydroxymethyl-tetrahydro-pyran-3,4,5-triol (Example 62, 1500mg,3.6mmol), L- proline (415mg, 3.6mmol) and ethanol (23 ml_) were added to a 50 mL 3-neck round bottom flask equipped with nitrogen purging, magnetic stirring bar,
thermometer pocket & calcium chloride guard tube and the mixture was stirred at 25-30°C for 30 min., then heat to reflux. A clear solution was observed which was refluxed for 30 min., then slowly cool to 25-30°C causing percipitation. Di- isopropyl ether (DIPE, 23 mL) was added while maintaining the mixture at 25-30°C and stirring continuously for additional one to two hours at the same temperature. The precipitate was collected by filtration using vacuum (Nitrogen atmosphere), and the filter cake was washed with ethanol-DIPE mixture (1 :1 v/v, 10ml) followed by DIPE (23 mL). The product was vacuum dried at 65-70°C for 5-6 hrs.
1:1 Proline Co-crvstal (ΔΗ 53 J/g) was observed by differential scanning calorimetry (DSC) and is shown in Fig. 1. A powder X-ray diffraction (PXRD) spectrum is shown in Fig. 2.
2:1 Proline Co-crvstal
Example 78: 2:1 Proline Co-crvstal with f2S.3R.4R.5S.6R>-2-r3-f2.3-Pihvdro-benzof1.41dioxin-6-ylmethvH-4-ethyl-phenvn-6-hvdroxymethyl-tetrahvdro-pyran- 3.4.5-triol
Method 3: 1 :2 Co-Crvstals of Example 62 with L-Proline
(2S,3R,4R,5S,6R)-2-[3-(2,3-Dihydro-benzo[1 ,4]dioxin-6-ylmethyl)-4-ethyl-phenyl]-6-hydroxymethyl-tetrahydro-pyran-3,4,5-triol (Example 62, 1 kg) was added to 15 L of ethanol with agitation while maintaining the mixture at 20-25 °C. The mixture was stirred for 10 min at 20-25 °C, then L-proline (537 gm) was added while maintaining the mixture at 20-25 °C. The mixture was stirred at this temperature for 30 min., then heated to reflux and refluxed for 30 min. The mixture was slowly cooled to 25-30°C then stired for 1 hr. DIPE (15 L) was added while maintaining the temperature at 25-30 °C and the mixture was stirred at this temperature for 1 hr. The precipitated product was collected by filtration and the product was washed with DIPE (5 L). The product was air dried at 65-70 °C to yield 1.22 kg
(79%) of a 1 :2 co-crystal of Example 62 : L-proline. A melting point 176°C (ΔΗ 85 J/g) was observed by differential scanning calorimetry (DSC) and is shown in Fig.
3. A powder X-ray diffraction (PXRD) spectrum is shown in Fig. 4. Relative
intensities of the most prominent powder x-ray diffraction peaks for the 1 :2 co- crystals of Example 62 and proline are shown in Table 5.
Table 5

PATENT
WO 2012140597
http://www.google.co.in/patents/WO2012140597A1?cl=en
. TABLE 2:
Intermediate 2: (2S,3R,4R,5S,6R)-2-[3-(2,3-Dihydro-benzo[1 ,4]dioxin-6-ylmethyl)-4-
Intermediate 2
Intermediate 1
Step I: To a stirred solution of acetic acid (2R,3R,4R,5S)-3,4,5-triacetoxy-6-[4-bromo-3- (2,3-dihydro-benzo[1 ,4]dioxin-6-ylmethyl)-phenyl]-tetrahydro-pyran-2-ylmethyl ester (Intermediate 1 , 10.0 g, 15.74 mmol) in toluene (200 mL) was added
tricyclohexylphosphine (1.76 g, 6.29 mmol), a solution of potassium phosphate tribasic (13.3 g, 62.9 mmol) in water (15 mL), and ethylboronic acid (3.4 g, 47.2 mmol). The reaction mixture was degassed for 45 min then palladium (II) acetate (529 mg, 2.3 mmol) was added. After refluxing overnight, the reaction mixture was cooled to room temperature, and water was added. The resulting mixture was extracted with ethyl acetate, (2 X 200 ml_), washed with water and brine, then dried over sodium sulfate, concentrated and purified by column chromatography to furnish acetic acid
(2R,3R,4R,5S)-3,4,5-triacetoxy-6-[3-(2,3-dihydro-benzo[1 ,4]dioxin-6-ylmethyl)-4-ethyl- phenyl]-tetrahydro-pyran-2-ylmethyl ester (5.4 g).
Step II: To a stirred solution of acetic acid (2R,3R,4R,5S)-3,4,5-triacetoxy-6-[3-(2,3- dihydro-benzo[1 ,4]dioxin-6-ylmethyl)-4-ethyl-phenyl]-tetrahydro-pyran-2-ylmethyl ester (9.3 g, 15.9 mmol) in methanol:THF:water 3:2:1 (170 ml.) was added lithium hydroxide (764 mg, 19.1 mmol). After stirring for 2 h at room temperature, the volatiles were evaporated under reduced pressure. The resulting residue was taken up in ethyl acetate (150 ml.) and washed with brine (75 ml_), brine containing 5 ml. of 5% aqueous KHS04 (75 ml_), and brine (20 ml.) again, then dried over sodium sulfate and concentrated to furnish (2S,3R,4R,5S,6R)-2-[4-Cyclopropyl-3-(2,3-dihydro-benzo[1 ,4]dioxin-6-ylmethyl)- phenyl]-6-hydroxymethyl-tetrahydro-pyran-3,4,5-triol (6.5 g)
1H NMR (400 MHz, CD3OD): δ 1.07 (t, J = 7.6 Hz, 3H), 2.57 (q, J = 7.6 Hz, 2H), 3.34- 3.50 (m, 4H), 3.68 (dd, J = 12.0, 5.6 Hz, 1 H), 3.85-3.91 (m, 3H), 4.08 (d, J = 9.6 Hz, 1 H), 4.17 (s, 4H), 6.53-6.58 (m, 2H), 6.68 (d, J = 8.4 Hz, 1 H), 7.15-7.25 (m, 3H).
MS (ES) m/z 434.2 (M+18).
Example 3: Synthesis of phosphoric acid (2R,3S,4R,5R,6S)-6-[3-(2,3-dihydro- benzo[1 ,4]dioxin-6-ylmethyl)-4-ethyl-phenyl]-3,4,5-trihydroxy-tetrahydro-pyran-2- ylmethyl ester diethyl ester
To a stirred solution of (2S,3R,4R,5S,6R)-2-[3-(2,3-dihydro-benzo[1 ,4]dioxin-6-ylmethyl)- 4-ethyl-phenyl]-6-hydroxymethyl-tetrahydro-pyran-3,4,5-triol (Intermediate 2, 500 mg, 1.2 mmol) in pyridine (5 ml) was added diethylchlorophosphate (0.27 ml, 1 .9 mmol) at -40°C. After stirring for 1 h at same temperature, reaction was quenched with the addition of 1 N HCI and extracted with ethyl acetate (2 X 10 ml). Combined organic layers were washed with brine (10 ml), dried over sodium sulfate, concentrated and purified by preparative HPLC to give 220 mg of phosphoric acid (2R,3S,4R,5R,6S)-6-[3-(2,3-dihydro- benzo[1 ,4]dioxin-6-ylmethyl)-4-ethyl-phenyl]-3,4,5-trihydroxy-tetrahydro-pyran-2-ylmethyl ester diethyl ester as a white solid. 1H NMR (400 MHz, CD3OD): δ 1.07 (t, J = 7.6 Hz, 3H), 1.15 (td J = 7.2, 1.2 Hz, 3H), 1.22 (td, J = 6.8, 0.8 Hz, 3H), 2.57 (q, J = 7.6 Hz, 2H), 3.36-3.46 (m, 3H), 3.53-3.55 (m, 1 H),3.89 (s, 2H), 3.96-4.11 (m, 5H), 4.17 (s, 4H), 4.18-4.22 (m 1 H), 4.30-4.34 (m, 1 H), 6.52 (d, J = 2.0 Hz, 1 H),6.57 (dd, J = 8.4, 2.4 Hz, 1 H), 6.68 (d, J = 8.4 Hz, 1 H), 7.15- 7.22(m, 3H). MS (ES) m/z 553.3 (M+1 ).
Example 4: Synthesis of disodium salt of phosphoric acid mono- {(2R,3S,4R,5R,6S)-6-[3-(2,3-dihydro-benzo[1 ,4]dioxin-6-ylmethyl)-4-ethyl-phenyl]- 3,4,5-trihydroxy-tetrahydro-pyran-2-ylmethyl} ester
To a stirred solution of (2S,3R,4R,5S,6R)-2-[3-(2,3-Dihydro-benzo[1 ,4]dioxin-6- ylmethyl)-4-ethyl-phenyl]-6-hydroxymethyl-tetrahydro-pyran-3,4,5-triol (Intermediate 2, 1.0 g, 2.4 mmol) in THF (15 ml) was added a solution of Diethyl-phosphoramidic acid di- tert-butyl ester (780 mg, 3.12 mmol) in THF (5 ml) at 0°C followed by a solution of tetrazole (435 mg, 6.2 mmol) in DCM (12.5 ml). After stirring for 5 min at same temperature, it was stirred at room temperature for 20 min. Reaction mixture was cooled to -40 °C and added a solution of m-CPBA (830 mg, 4.8 mmol) in DCM (5 ml). The reaction mixture was stirred at same temperature for 5 min and then at room temperature for 2 h. Reaction mixture was cooled to 0°C and quenched by the addition of 10% sodium bisulfite solution (5 ml). This was extracted with ether (3 X 10 ml). Combined organic layer was washed with brine (5 ml), dried over sodium sulfate and concentrated to give 700 mg of phosphoric acid di-tert-butyl ester (2R,3S,4R,5R,6S)-6- [3-(2,3-dihydro-benzo[1 ,4]dioxin-6-ylmethyl)-4-ethyl-phenyl]-3,4,5-trihydroxy-tetrahydro- pyran-2-ylmethyl ester.
To the stirred solution of phosphoric acid di-tert-butyl ester (2R,3S,4R,5R,6S)-6-[3-(2,3- dihydro-benzo[1 ,4]dioxin-6-ylmethyl)-4-ethyl-phenyl]-3,4,5-trihydroxy-tetrahydro-pyran-2- ylmethyl ester (500 mg) in methanol (20 ml) was added amberlyst 15 ion exchange resin (250 mg) and refluxed for overnight. Reaction mixture was cooled to room temperature, filtered through celite bed and filtrate was concentrated to give 300 mg of phosphoric acid mono-{(2R,3S,4R,5R,6S)-6-[3-(2,3-dihydro-benzo[1 ,4]dioxin-6-ylmethyl)-4-ethyl- phenyl]-3,4,5-trihydroxy-tetrahydro-pyran-2-ylmethyl} ester. The crude material was taken up for next reaction.
To a solution of phosphoric acid mono-{(2R,3S,4R,5R,6S)-6-[3-(2,3-dihydro- benzo[1 ,4]dioxin-6-ylmethyl)-4-ethyl-phenyl]-3,4,5-trihydroxy-tetrahydro-pyran-2- ylmethyl} ester (300 mg, 0.6 mmol) in methanol (5 ml) was added 1 N sodium bicarbonate solution (80 mg, 0.7 mmol) in water. After stirring at room temperature for 2 h, the volatiles were evaporated under reduced pressure. The resulting solid was triturated with diethyl ether. The resulting residue was purified by preparative HPLC to give 95 mg of disodium salt of phosphoric acid mono-{(2R,3S,4R,5R,6S)-6-[3-(2,3- dihydro-benzo[1 ,4]dioxin-6-ylmethyl)-4-ethyl-phenyl]-3,4,5-trihydroxy-tetrahydro-pyran-2- ylmethyl} ester.
1H NMR (400 MHz, CD3OD): δ 1.06 (t, J = 7.4 Hz, 3H), 2.56 ( q, J = 7.3 Hz, 2H), 3.34- 3.41 (m, 2H), 3.49 (t, J = 8.8 Hz, 1 H), 3.81-3.88 (m, ,3H), 3.92-3.99 (m, 1 H), 4.05 (d, J = 9.3 Hz, 1 H), 4.16 (s, 4H), 4.20-4.25 (m, 1 H), 6.54 (m, 2H), 6.67 (d, J = 7.8 Hz, 1 H), 7.12-7.21 (m, 3H). MS (ES) m/z 497.1 (M+1 ) for phosphoric acid.

PATENT
SEE INDIAN PATENT
IN 2009DE02173
Glycoside derivatives and uses thereof
REFERENCES
Pediatric investigation plan (PIP) decision: (S)-Pyrrolidine-2-carboxylic acid compound with (2S,3R,4R,5S,6R)-2-(3-((2,3-dihydrobenzo[b][1,4]dioxin-6-yl)methyl)-4-ethylphenyl)-6-(hydroxymethyl)tetrahydro-2H-pyran-3,4,5-triol (2:1) ( LIK066) (EMEA-001527-PIP01-13)
European Medicines Agency (EMA) Web Site 2014, July 24
Safety, tolerability, pharmacokinetics (PK) and pharmacodynamics (PD) assessment of LIK066 in healthy subjects and in patients with type 2 diabetes mellitus (T2DM) (NCT01407003)
ClinicalTrials.gov Web Site 2011, August 07
IN 2009DE02173
| WO2001016147A1 | 24 Aug 2000 | 8 Mar 2001 | Kissei Pharmaceutical | Glucopyranosyloxypyrazole derivatives, medicinal compositions containing the same and intermediates in the production thereof |
| WO2001027128A1 | 2 Oct 2000 | 19 Apr 2001 | Bruce Ellsworth | C-aryl glucoside sglt2 inhibitors |
| WO2001068660A1 | 15 Mar 2001 | 20 Sep 2001 | Hideki Fujikura | Glucopyranosyloxy benzylbenzene derivatives, medicinal compositions containing the same and intermediates for the preparation of the derivatives |
| WO2001074834A1 | 29 Mar 2001 | 11 Oct 2001 | Squibb Bristol Myers Co | O-aryl glucoside sglt2 inhibitors and method |
| WO2003020737A1 | 5 Sep 2002 | 13 Mar 2003 | Squibb Bristol Myers Co | O-pyrazole glucoside sglt2 inhibitors and method of use |
| WO2003043985A1 | 20 Nov 2002 | 30 May 2003 | Andrew Thomas Bach | Heterocyclic compounds and methods of use |
| WO2004018491A1 | 21 Aug 2003 | 4 Mar 2004 | Nobuhiko Fushimi | Pyrazole derivatives, medicinal composition containing the same, medicinal use thereof, and intermediate for production thereof |
| WO2004078163A2 | 26 Feb 2004 | 16 Sep 2004 | Oern Almarsson | Pharmaceutical co-crystal compositions of drugs such as carbamazepine, celecoxib, olanzapine, itraconazole, topiramate, modafinil, 5-fluorouracil, hydrochlorothiazide, acetaminophen, aspirin, flurbiprofen, phenytoin and ibuprofen |
| WO2004080990A1 | 12 Mar 2004 | 23 Sep 2004 | Kazuhiro Ikegai | C-glycoside derivatives and salts thereof |
| WO2004099230A1 | 30 Apr 2004 | 18 Nov 2004 | Eikyu Yoshiteru | Monosaccharide compounds |
| WO2004103995A1 | 19 May 2004 | 2 Dec 2004 | Gary Michael Ksander | N-acyl nitrogen heterocycles as ligands of peroxisome proliferator-activated receptors |
| WO2005011592A2 | 29 Jul 2004 | 10 Feb 2005 | Janssen Pharmaceutica Nv | Substituted indazole-o-glucosides |
| WO2005021566A2 | 20 Aug 2004 | 10 Mar 2005 | Barsoumian Edward Leon | Glucopyranosyloxy- pirazoles, drugs containing said compounds the use and production method thereof |
| WO2005085237A1 | 3 Mar 2005 | 15 Sep 2005 | Kissei Pharmaceutical | Fused heterocycle derivative, medicinal composition containing the same, and medicinal use thereof |
| WO2005085265A1 | 3 Mar 2005 | 15 Sep 2005 | Kissei Pharmaceutical | Fused heterocycle derivative, medicinal composition containing the same, and medicinal use thereof |
| WO2006011502A1 | 27 Jul 2005 | 2 Feb 2006 | Chugai Pharmaceutical Co Ltd | Novel glucitol derivative, prodrug thereof and salt thereof, and therapeutic agent containing the same for diabetes |
| WO2006054629A1 | 17 Nov 2005 | 26 May 2006 | Kissei Pharmaceutical | 1-SUBSTITUTED-3-β-D-GLUCOPYRANOSYLATED NITROGENOUS HETERO- CYCLIC COMPOUNDS AND MEDICINES CONTAINING THE SAME |
| WO2008016132A1 | 3 Aug 2007 | 7 Feb 2008 | Daiichi Sankyo Co Ltd | Benzyl phenyl glucopyranoside derivative |
| WO2011048112A1 * | 19 Oct 2010 | 28 Apr 2011 | Novartis Ag | Glycoside derivatives and uses thereof |
| US20030114390 * | 4 Oct 2002 | 19 Jun 2003 | Washburn William N. | C-aryl glucoside SGLT2 inhibitors and method |
| US20040018998 | 21 Sep 2001 | 29 Jan 2004 | Hideki Fujikura | Glucopyranosyloxybenzylbenzene derivatives and medicinal compositions containing the same |
| US20060009400 | 28 Jun 2005 | 12 Jan 2006 | Boehringer Ingelheim International Gmbh | D-xylopyranosyl-substituted phenyl derivatives, medicaments containing such compounds, their use and process for their manufacture |
| US20060019948 | 15 Jul 2005 | 26 Jan 2006 | Boehringer Ingelheim International Gmbh | Methylidene-D-xylopyranosyl- and oxo-D-xylopyranosyl-substituted phenyl derivatives, medicaments containing such compounds, their use and process for their manufacture |
| US20060025349 | 27 Jul 2005 | 2 Feb 2006 | Boehringer Ingelheim International Gmbh | D-xylopyranosyl-phenyl-substituted cycles, medicaments containing such compounds, their use and process for their manufacture |
| US20060035841 | 9 Aug 2005 | 16 Feb 2006 | Boehringer Ingelheim International Gmbh | D-xylopyranosyl-phenyl-substituted cycles, medicaments containing such compounds, their use and process for their manufacture |
| US20060074031 | 30 Sep 2005 | 6 Apr 2006 | Boehringer Ingelheim International Gmbh | D-pyranosyl-substituted phenyl derivatives, medicaments containing such compounds, their use and process for their manufacture |
| US20060293252 | 14 Aug 2006 | 28 Dec 2006 | Sanofi-Aventis Deutschland Gmbh | Novel Thiophene Glycoside Derivatives, Processes for The Preparation, Medicaments Comprising These Compounds, and The Use Thereof |
| US20080027014 | 26 Jul 2007 | 31 Jan 2008 | Tanabe Seiyaku Co., Ltd. | Novel SGLT inhibitors |
| Citing Patent | Filing date | Publication date | Applicant | Title |
|---|---|---|---|---|
| WO2015032272A1 * | 19 Aug 2014 | 12 Mar 2015 | Jiangsu Hansoh Pharmaceutical Co., Ltd. | C-aryl glucoside derivative, preparation method for same, and medical applications thereof |
| US9034921 | 1 Jun 2012 | 19 May 2015 | Green Cross Corporation | Diphenylmethane derivatives as SGLT2 inhibitors |
INVENTORS OF LIK 066
Gregory Raymond Bebernitz, Mark G. Bock, Dumbala Srinivas Reddy, Atul Kashinath Hajare, Vinod Vyavahare, Sandeep Bhausaheb Bhosale, Suresh Eknath Kurhade, Videsh Salunkhe, Nadim S. Shaikh, Debnath Bhuniya, P. Venkata Palle, Lili Feng, Jessica Liang,
| BEBERNITZ, Gregory, Raymond; (US). BOCK, Mark, G.; (US). REDDY, Dumbala Srinivas; (IN). HAJARE, Atul Kashinath; (IN). VYAVAHARE, Vinod; (IN). BHOSALE, Sandeep Bhausaheb; (IN). KURHADE, Suresh Eknath; (IN). SALUNKHE, Videsh; (IN). SHAIKH, Nadim, S.; (IN). BHUNIYA, Debnath; (IN). PALLE, P., Venkata; (IN). FENG, Lili; (US). LIANG, Jessica; (US) |
BEBERNITZ, Gregory, Raymond….PIC NOT AVAILABLE

Dr. Srinivasa Reddy
 NADEEM SHAIKH
NADEEM SHAIKH
 Venkata Palle
Venkata Palle
ONLY FEW…………………….
//////Licogliflozin diprolinate
see……..http://medcheminternational.blogspot.in/2015/11/lik-066-novartis-for-treatment-of-type.html

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....















































 RP 6530
RP 6530