Home » Posts tagged 'HR+/HER2- metastatic breast cancer'
Tag Archives: HR+/HER2- metastatic breast cancer
SEARCH THIS BLOG
 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....FOR BLOG HOME CLICK HERE
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....FOR BLOG HOME CLICK HEREBlog Stats
- 4,849,533 hits
Pages
- About me
- All about drugs
- Bioavailability
- CLINICAL TRIALS
- Combinatorial Chemistry in Drug Design
- DISCLAIMER
- Drug Basics
- DRUG COMPANIES
- DRUG DESIGN
- DRUG DISCOVERY
- Drug Information
- DRUG NEWS
- DRUG PORTALS
- Drug resistance
- DRUG SITES
- DRUG SYNTHESIS
- DRUGS NDA
- DRUGS, FDA2012
- FDA -US
- GMP
- HEALTH CANADA
- HELP ME/ ACCOLADES
- HSA SINGAPORE
- INDA
- KOREA DRUG APPROVAL PROCESS
- MHLW JAPAN, 厚生労働省
- NMPA CHINA
- PATENT EXPIRY DATES 1/3
- PATENT EXPIRY DATES 2/3
- PATENT EXPIRY DATES 3/3
- Patent portals
- Polymorphism
- Regulations and Guidance
- Scientificupdate (UK) on a roll
- sFDA CHINA
- SOUTH AFRICA, MCC
- SPECIAL DRUGS
- Traditional Medicine
- WHAT IS A DRUG
- World drug tracker
Archives
- February 2026 (22)
- January 2026 (28)
- December 2025 (31)
- November 2025 (31)
- October 2025 (22)
- September 2025 (24)
- August 2025 (27)
- July 2025 (25)
- June 2025 (23)
- May 2025 (34)
- April 2025 (32)
- March 2025 (4)
- October 2024 (4)
- September 2024 (17)
- May 2024 (4)
- April 2024 (2)
- February 2024 (8)
- January 2024 (1)
- December 2023 (3)
- November 2023 (2)
- October 2023 (1)
- September 2023 (1)
- August 2023 (4)
- July 2023 (1)
- April 2023 (1)
- March 2023 (1)
- January 2023 (5)
- December 2022 (3)
- November 2022 (3)
- October 2022 (9)
- September 2022 (9)
- August 2022 (7)
- July 2022 (2)
- June 2022 (7)
- May 2022 (5)
- April 2022 (7)
- March 2022 (7)
- February 2022 (12)
- January 2022 (15)
- December 2021 (26)
- November 2021 (17)
- October 2021 (21)
- September 2021 (21)
- August 2021 (19)
- July 2021 (13)
- June 2021 (30)
- May 2021 (7)
- April 2021 (9)
- March 2021 (25)
- February 2021 (10)
- January 2021 (17)
- December 2020 (21)
- November 2020 (5)
- October 2020 (5)
- September 2020 (12)
- August 2020 (3)
- July 2020 (5)
- June 2020 (1)
- May 2020 (1)
- April 2020 (1)
- March 2020 (19)
- February 2020 (9)
- January 2020 (12)
- December 2019 (4)
- November 2019 (4)
- October 2019 (11)
- September 2019 (8)
- August 2019 (14)
- July 2019 (23)
- June 2019 (15)
- May 2019 (16)
- April 2019 (21)
- March 2019 (14)
- February 2019 (7)
- January 2019 (17)
- December 2018 (7)
- November 2018 (14)
- October 2018 (12)
- September 2018 (18)
- August 2018 (22)
- July 2018 (15)
- June 2018 (38)
- May 2018 (14)
- April 2018 (19)
- March 2018 (25)
- February 2018 (15)
- January 2018 (17)
- December 2017 (20)
- November 2017 (15)
- October 2017 (19)
- September 2017 (8)
- August 2017 (13)
- July 2017 (11)
- June 2017 (6)
- May 2017 (10)
- April 2017 (23)
- March 2017 (18)
- February 2017 (17)
- January 2017 (15)
- December 2016 (19)
- November 2016 (31)
- October 2016 (38)
- September 2016 (33)
- August 2016 (40)
- July 2016 (53)
- June 2016 (50)
- May 2016 (50)
- April 2016 (70)
- March 2016 (45)
- February 2016 (49)
- January 2016 (41)
- December 2015 (37)
- November 2015 (37)
- October 2015 (49)
- September 2015 (35)
- August 2015 (29)
- July 2015 (25)
- June 2015 (28)
- May 2015 (21)
- April 2015 (41)
- March 2015 (51)
- February 2015 (23)
- January 2015 (33)
- December 2014 (46)
- November 2014 (31)
- October 2014 (38)
- September 2014 (74)
- August 2014 (86)
- July 2014 (101)
- June 2014 (170)
- May 2014 (98)
- April 2014 (113)
- March 2014 (80)
- February 2014 (63)
- January 2014 (70)
- December 2013 (100)
- November 2013 (91)
- October 2013 (102)
- September 2013 (104)
- August 2013 (114)
- July 2013 (129)
- June 2013 (131)
- May 2013 (155)
- April 2013 (109)
- March 2013 (105)
- February 2013 (81)
Categories
- 0rphan drug status (225)
- 2020 APPROVALS (48)
- accelerated approval (1)
- ACH 4471 (1)
- Acute Hepatic Porphyria (1)
- Acute myeloid leukaemia (1)
- African medicine (6)
- AIDS (7)
- Alzheimer's (4)
- ANAESTHETIC (1)
- anaesthetics (4)
- analgesic (3)
- ANALYTICAL (1)
- ANDA (5)
- Anthony crasto (42)
- ANTI ACNE (2)
- ANTI ANDROGEN (1)
- ANTI DEPRESSANT (1)
- ANTI DIABETES (4)
- ANTI DOPING (1)
- ANTI FUNGAL (6)
- ANTI HBV (1)
- ANTI INFLAMATORY (8)
- anti tubeculosis (1)
- antibacterial (10)
- Antibiotics (6)
- ANTIBODIES (54)
- ANTICONVULSANT (1)
- Antiemetic (2)
- antiglaucoma (1)
- Antihypertensives (3)
- antiinfluenza (2)
- antimalarials (12)
- Antineoplastic (50)
- antiobesity (1)
- Antispasmodic (1)
- ANTITUSSIVE (1)
- Antiulcerative (2)
- Antivirals (9)
- APPROVALS 2021 (86)
- APPROVALS 2022 (49)
- APPROVALS 2023 (35)
- approvals 2024 (55)
- APPROVALS 2025 (32)
- APPROVALS 2026 (1)
- ARAB MEDICINE (5)
- Atopic dermatitis (1)
- Australia (1)
- AUSTRALIA 2021 (1)
- AWARD (13)
- AYURVEDA (78)
- Biosimilar drugs (39)
- BLA (4)
- BLOGS (28)
- Breakthrough Therapy Designation (69)
- canada (4)
- CANADA 2021 (1)
- cancer (170)
- cfda (6)
- CHINA 2011 (1)
- CHINA 2012 (1)
- CHINA 2018 (1)
- china 2019 (1)
- CHINA 2021 (5)
- CHINA 2022 (5)
- CHINA 2023 (4)
- CHINA 2024 (20)
- CHINA 2025 (1)
- china pipeline (37)
- CHINESE HERBS (24)
- chronic kidney disease (1)
- CLINICAL TRIALS (10)
- COMPANIES (81)
- conference (7)
- Contrast agent (7)
- CORONAVIRUS (48)
- COVID-19 (46)
- CRL (1)
- CYSTIC FIBRIOSIS (2)
- Dengue (2)
- DEUTERATED (1)
- DIABETES (57)
- DNDi (1)
- Drug discovery (22)
- DRUG MARKETING (5)
- DRUG REVIEW (8)
- Duchenne muscular dystrophy (1)
- EMA (4)
- EU 2003 (1)
- EU 2005 (1)
- EU 2014 (5)
- EU 2015 (6)
- EU 2016 (4)
- EU 2017 (8)
- EU 2018 (5)
- EU 2019 (1)
- EU 2020 (5)
- EU 2021 (11)
- EU 2022 (4)
- EU 2023 (4)
- EU 2024 (1)
- EU PIPELINE (61)
- EU SUBMISSION (32)
- EXTENDED USE (1)
- FAST TRACK (4)
- FAST TRACK FDA (78)
- FDA 15 (4)
- FDA 2010 (2)
- FDA 2011 (1)
- FDA 2012 (7)
- FDA 2013 (6)
- FDA 2014 (53)
- FDA 2015 (39)
- FDA 2016 (23)
- FDA 2017 (58)
- FDA 2018 (53)
- FDA 2019 (44)
- FDA 2020 (34)
- fda 2021 (45)
- FDA 2022 (24)
- FDA 2023 (32)
- fda 2023, approvals 2023 (1)
- FDA 2024 (29)
- FDA 2025 (33)
- FDA 2026 (2)
- Featured product (1)
- female sexual dysfunction (1)
- FIBROMYGALIA (1)
- flow synthesis (14)
- FORMULATION (14)
- GAIN (1)
- Gastric Proton Pump Inhibitor (1)
- Generating Antibiotic Incentives Now (1)
- GENERIC DRUG (106)
- GENERICS (22)
- glenmark (18)
- GLENMARK (16)
- GLIPTIN (1)
- GMP (10)
- gout (1)
- Greek medicine (3)
- hepatitis C viral infections (1)
- Herbals (8)
- HOMEOPATHY (3)
- HORMONE (1)
- Human medicines European Public Assessment Report EPAR (2)
- Hypertension (1)
- hypnotic (1)
- idiopathic pulmonary fibrosis (3)
- IMPLANTS (2)
- IND (1)
- IND 2017 (2)
- IND 2018 (3)
- IND 2021 (8)
- IND Filed (2)
- INDIA 2020 (5)
- INDIA 2022 (3)
- Insomnia (1)
- Investigational device exemption (IDE) approval (2)
- JAPAN 2009 (1)
- JAPAN 2013 (1)
- JAPAN 2014 (1)
- JAPAN 2015 (2)
- JAPAN 2016 (1)
- japan 2017 (6)
- JAPAN 2018 (10)
- japan 2019 (8)
- japan 2020 (10)
- JAPAN 2021 (11)
- JAPAN 2022 (14)
- Japan marketing (34)
- Japan pipeline (38)
- Korea (3)
- korea 2012 (2)
- korea 2016 (1)
- korea 2019 (1)
- Lactams (1)
- MAA (3)
- Macrolides (2)
- Malaria (17)
- MANUFACTURING (8)
- Market info (3)
- Marketing authorisation application EU (4)
- MEDICAL DEVICE (9)
- MIGRAINE (1)
- molybdenum cofactor deficiency (1)
- MONKEY POX (1)
- Monoclonal antibody (89)
- Nanotechnology (5)
- NCE (2)
- NDA (51)
- NDA JAPAN (3)
- neuropathic pain (2)
- Neutraceuticals (1)
- New Drug Application Resubmission FDA (5)
- NEW DRUGS (76)
- New drugs canada (5)
- New drugs china (6)
- New drugs EU (10)
- New drugs Japan (2)
- NEW USE DRUGS (1)
- Newzealand (2)
- NICE (2)
- non-Hodgkin's lymphoma (1)
- NUCLEOTIDES (1)
- obesity (3)
- OCCULAR (2)
- ocular hypertension (1)
- organic chemistry (12)
- osteoarthritis (2)
- PAIN (2)
- Parkinson's disease (1)
- PATENT (93)
- PATENTS (82)
- Peptide drugs (56)
- PFIZER (1)
- PHASE 1 (90)
- PHASE 3 (25)
- PHASE1 (145)
- Phase2 drugs (282)
- Phase3 drugs (306)
- plaque psoriasis (1)
- POLYMORPH (10)
- Preclinical china (14)
- Preclinical drugs (121)
- Premture ejaculation (3)
- prime designation (1)
- Priority review (90)
- PROCESS (39)
- Promising clips (9)
- Psoriasis (1)
- QbD (2)
- QIDP (23)
- radio labelled (4)
- RADIOACTIVE AGENT (5)
- RADIOLABELLED (10)
- Rare disease (3)
- RARE PEDIATRIC DISEASE REVIEW (2)
- Regenerative medicine (4)
- Regulatory (74)
- Resubmission FDA (2)
- REVIEW (10)
- rheumatoid arthritis (3)
- SCHLEROSING AGENT (2)
- Scottish Medicines Consortium (1)
- sex arousal (2)
- SFDA FAST TRACK (2)
- SINGAPORE (4)
- sNDA (12)
- sNDA JAPAN (2)
- Solid tumours (2)
- spectroscopy (16)
- SPOTLIGHT (53)
- Stem cells (3)
- SYNTHESIS (10)
- TIVE (1)
- TOXINS (1)
- Translational medicine (2)
- Transverse myletis (2)
- Tropical Disease Priority Review Voucher (1)
- UK (1)
- UNANI MEDICINE (2)
- Uncategorized (2,278)
- US HERBS (7)
- USFDA SUBMISSION (1)
- vaccine (26)
- veterinary (8)
- virus (1)
- VITAMINS (3)
- x ray contrast agent (10)
Recent Posts
- Olomorasib February 25, 2026
- Ocadusertib February 24, 2026
- Nivegacetor February 23, 2026
- Nispomeben February 21, 2026
- Nedemelteon February 20, 2026
- Naxtarubicin, Annamycin February 19, 2026
- Navepdekinra February 18, 2026
- Napazimone February 17, 2026
- Nacresertib February 15, 2026
- Mosperafenib February 14, 2026
- Mocaciclib February 13, 2026
- Mobinitinib February 12, 2026
- Mevrometostat February 10, 2026
- Maritupirdine February 9, 2026
- Luvometinib February 8, 2026
ORGANIC SPECTROSCOPY

SUBSCRIBE
Enter your email address:
Delivered by FeedBurner
Subscribe to New Drug Approvals by EmailDR ANTHONY MELVIN CRASTO Ph.D
DR ANTHONY MELVIN CRASTO, Born in Mumbai in 1964 and graduated from Mumbai University, Completed his Ph.D from ICT, 1991,Matunga, Mumbai, India, in Organic Chemistry, The thesis topic was Synthesis of Novel Pyrethroid Analogues, Currently he is working with AFRICURE PHARMA, ROW2TECH, NIPER-G, Department of Pharmaceuticals, Ministry of Chemicals and Fertilizers, Govt. of India as ADVISOR, earlier assignment was with GLENMARK LIFE SCIENCES LTD, as CONSUlTANT, Retired from GLENMARK in Jan2022 Research Centre as Principal Scientist, Process Research (bulk actives) at Mahape, Navi Mumbai, India. Total Industry exp 32 plus yrs, Prior to joining Glenmark, he has worked with major multinationals like Hoechst Marion Roussel, now Sanofi, Searle India Ltd, now RPG lifesciences, etc. He has worked with notable scientists like Dr K Nagarajan, Dr Ralph Stapel, Prof S Seshadri, etc, He did custom synthesis for major multinationals in his career like BASF, Novartis, Sanofi, etc., He has worked in Discovery, Natural products, Bulk drugs, Generics, Intermediates, Fine chemicals, Neutraceuticals, GMP, Scaleups, etc, he is now helping millions, has 9 million plus hits on Google on all Organic chemistry websites. His friends call him Open superstar worlddrugtracker. His New Drug Approvals, Green Chemistry International, All about drugs, Eurekamoments, Organic spectroscopy international, etc in organic chemistry are some most read blogs He has hands on experience in initiation and developing novel routes for drug molecules and implementation them on commercial scale over a 32 PLUS year tenure till date Feb 2023, Around 35 plus products in his career. He has good knowledge of IPM, GMP, Regulatory aspects, he has several International patents published worldwide . He has good proficiency in Technology transfer, Spectroscopy, Stereochemistry, Synthesis, Polymorphism etc., He suffered a paralytic stroke/ Acute Transverse mylitis in Dec 2007 and is 90 %Paralysed, He is bound to a wheelchair, this seems to have injected feul in him to help chemists all around the world, he is more active than before and is pushing boundaries, He has 100 million plus hits on Google, 2.5 lakh plus connections on all networking sites, 100 Lakh plus views on dozen plus blogs, 227 countries, 7 continents, He makes himself available to all, contact him on +91 9323115463, email amcrasto@gmail.com, Twitter, @amcrasto , He lives and will die for his family, 90% paralysis cannot kill his soul., Notably he has 38 lakh plus views on New Drug Approvals Blog in 227 countries......https://newdrugapprovals.wordpress.com/ , He appreciates the help he gets from one and all, Friends, Family, Glenmark, Readers, Wellwishers, Doctors, Drug authorities, His Contacts, Physiotherapist, etc He has total of 32 International and Indian awards
Verified Services
Pages
- About me
- All about drugs
- Bioavailability
- CLINICAL TRIALS
- Combinatorial Chemistry in Drug Design
- DISCLAIMER
- Drug Basics
- DRUG COMPANIES
- DRUG DESIGN
- DRUG DISCOVERY
- Drug Information
- DRUG NEWS
- DRUG PORTALS
- Drug resistance
- DRUG SITES
- DRUG SYNTHESIS
- DRUGS NDA
- DRUGS, FDA2012
- FDA -US
- GMP
- HEALTH CANADA
- HELP ME/ ACCOLADES
- HSA SINGAPORE
- INDA
- KOREA DRUG APPROVAL PROCESS
- MHLW JAPAN, 厚生労働省
- NMPA CHINA
- PATENT EXPIRY DATES 1/3
- PATENT EXPIRY DATES 2/3
- PATENT EXPIRY DATES 3/3
- Patent portals
- Polymorphism
- Regulations and Guidance
- Scientificupdate (UK) on a roll
- sFDA CHINA
- SOUTH AFRICA, MCC
- SPECIAL DRUGS
- Traditional Medicine
- WHAT IS A DRUG
- World drug tracker
Archives
- February 2026 (22)
- January 2026 (28)
- December 2025 (31)
- November 2025 (31)
- October 2025 (22)
- September 2025 (24)
- August 2025 (27)
- July 2025 (25)
- June 2025 (23)
- May 2025 (34)
- April 2025 (32)
- March 2025 (4)
- October 2024 (4)
- September 2024 (17)
- May 2024 (4)
- April 2024 (2)
- February 2024 (8)
- January 2024 (1)
- December 2023 (3)
- November 2023 (2)
- October 2023 (1)
- September 2023 (1)
- August 2023 (4)
- July 2023 (1)
- April 2023 (1)
- March 2023 (1)
- January 2023 (5)
- December 2022 (3)
- November 2022 (3)
- October 2022 (9)
- September 2022 (9)
- August 2022 (7)
- July 2022 (2)
- June 2022 (7)
- May 2022 (5)
- April 2022 (7)
- March 2022 (7)
- February 2022 (12)
- January 2022 (15)
- December 2021 (26)
- November 2021 (17)
- October 2021 (21)
- September 2021 (21)
- August 2021 (19)
- July 2021 (13)
- June 2021 (30)
- May 2021 (7)
- April 2021 (9)
- March 2021 (25)
- February 2021 (10)
- January 2021 (17)
- December 2020 (21)
- November 2020 (5)
- October 2020 (5)
- September 2020 (12)
- August 2020 (3)
- July 2020 (5)
- June 2020 (1)
- May 2020 (1)
- April 2020 (1)
- March 2020 (19)
- February 2020 (9)
- January 2020 (12)
- December 2019 (4)
- November 2019 (4)
- October 2019 (11)
- September 2019 (8)
- August 2019 (14)
- July 2019 (23)
- June 2019 (15)
- May 2019 (16)
- April 2019 (21)
- March 2019 (14)
- February 2019 (7)
- January 2019 (17)
- December 2018 (7)
- November 2018 (14)
- October 2018 (12)
- September 2018 (18)
- August 2018 (22)
- July 2018 (15)
- June 2018 (38)
- May 2018 (14)
- April 2018 (19)
- March 2018 (25)
- February 2018 (15)
- January 2018 (17)
- December 2017 (20)
- November 2017 (15)
- October 2017 (19)
- September 2017 (8)
- August 2017 (13)
- July 2017 (11)
- June 2017 (6)
- May 2017 (10)
- April 2017 (23)
- March 2017 (18)
- February 2017 (17)
- January 2017 (15)
- December 2016 (19)
- November 2016 (31)
- October 2016 (38)
- September 2016 (33)
- August 2016 (40)
- July 2016 (53)
- June 2016 (50)
- May 2016 (50)
- April 2016 (70)
- March 2016 (45)
- February 2016 (49)
- January 2016 (41)
- December 2015 (37)
- November 2015 (37)
- October 2015 (49)
- September 2015 (35)
- August 2015 (29)
- July 2015 (25)
- June 2015 (28)
- May 2015 (21)
- April 2015 (41)
- March 2015 (51)
- February 2015 (23)
- January 2015 (33)
- December 2014 (46)
- November 2014 (31)
- October 2014 (38)
- September 2014 (74)
- August 2014 (86)
- July 2014 (101)
- June 2014 (170)
- May 2014 (98)
- April 2014 (113)
- March 2014 (80)
- February 2014 (63)
- January 2014 (70)
- December 2013 (100)
- November 2013 (91)
- October 2013 (102)
- September 2013 (104)
- August 2013 (114)
- July 2013 (129)
- June 2013 (131)
- May 2013 (155)
- April 2013 (109)
- March 2013 (105)
- February 2013 (81)
Categories
- 0rphan drug status (225)
- 2020 APPROVALS (48)
- accelerated approval (1)
- ACH 4471 (1)
- Acute Hepatic Porphyria (1)
- Acute myeloid leukaemia (1)
- African medicine (6)
- AIDS (7)
- Alzheimer's (4)
- ANAESTHETIC (1)
- anaesthetics (4)
- analgesic (3)
- ANALYTICAL (1)
- ANDA (5)
- Anthony crasto (42)
- ANTI ACNE (2)
- ANTI ANDROGEN (1)
- ANTI DEPRESSANT (1)
- ANTI DIABETES (4)
- ANTI DOPING (1)
- ANTI FUNGAL (6)
- ANTI HBV (1)
- ANTI INFLAMATORY (8)
- anti tubeculosis (1)
- antibacterial (10)
- Antibiotics (6)
- ANTIBODIES (54)
- ANTICONVULSANT (1)
- Antiemetic (2)
- antiglaucoma (1)
- Antihypertensives (3)
- antiinfluenza (2)
- antimalarials (12)
- Antineoplastic (50)
- antiobesity (1)
- Antispasmodic (1)
- ANTITUSSIVE (1)
- Antiulcerative (2)
- Antivirals (9)
- APPROVALS 2021 (86)
- APPROVALS 2022 (49)
- APPROVALS 2023 (35)
- approvals 2024 (55)
- APPROVALS 2025 (32)
- APPROVALS 2026 (1)
- ARAB MEDICINE (5)
- Atopic dermatitis (1)
- Australia (1)
- AUSTRALIA 2021 (1)
- AWARD (13)
- AYURVEDA (78)
- Biosimilar drugs (39)
- BLA (4)
- BLOGS (28)
- Breakthrough Therapy Designation (69)
- canada (4)
- CANADA 2021 (1)
- cancer (170)
- cfda (6)
- CHINA 2011 (1)
- CHINA 2012 (1)
- CHINA 2018 (1)
- china 2019 (1)
- CHINA 2021 (5)
- CHINA 2022 (5)
- CHINA 2023 (4)
- CHINA 2024 (20)
- CHINA 2025 (1)
- china pipeline (37)
- CHINESE HERBS (24)
- chronic kidney disease (1)
- CLINICAL TRIALS (10)
- COMPANIES (81)
- conference (7)
- Contrast agent (7)
- CORONAVIRUS (48)
- COVID-19 (46)
- CRL (1)
- CYSTIC FIBRIOSIS (2)
- Dengue (2)
- DEUTERATED (1)
- DIABETES (57)
- DNDi (1)
- Drug discovery (22)
- DRUG MARKETING (5)
- DRUG REVIEW (8)
- Duchenne muscular dystrophy (1)
- EMA (4)
- EU 2003 (1)
- EU 2005 (1)
- EU 2014 (5)
- EU 2015 (6)
- EU 2016 (4)
- EU 2017 (8)
- EU 2018 (5)
- EU 2019 (1)
- EU 2020 (5)
- EU 2021 (11)
- EU 2022 (4)
- EU 2023 (4)
- EU 2024 (1)
- EU PIPELINE (61)
- EU SUBMISSION (32)
- EXTENDED USE (1)
- FAST TRACK (4)
- FAST TRACK FDA (78)
- FDA 15 (4)
- FDA 2010 (2)
- FDA 2011 (1)
- FDA 2012 (7)
- FDA 2013 (6)
- FDA 2014 (53)
- FDA 2015 (39)
- FDA 2016 (23)
- FDA 2017 (58)
- FDA 2018 (53)
- FDA 2019 (44)
- FDA 2020 (34)
- fda 2021 (45)
- FDA 2022 (24)
- FDA 2023 (32)
- fda 2023, approvals 2023 (1)
- FDA 2024 (29)
- FDA 2025 (33)
- FDA 2026 (2)
- Featured product (1)
- female sexual dysfunction (1)
- FIBROMYGALIA (1)
- flow synthesis (14)
- FORMULATION (14)
- GAIN (1)
- Gastric Proton Pump Inhibitor (1)
- Generating Antibiotic Incentives Now (1)
- GENERIC DRUG (106)
- GENERICS (22)
- glenmark (18)
- GLENMARK (16)
- GLIPTIN (1)
- GMP (10)
- gout (1)
- Greek medicine (3)
- hepatitis C viral infections (1)
- Herbals (8)
- HOMEOPATHY (3)
- HORMONE (1)
- Human medicines European Public Assessment Report EPAR (2)
- Hypertension (1)
- hypnotic (1)
- idiopathic pulmonary fibrosis (3)
- IMPLANTS (2)
- IND (1)
- IND 2017 (2)
- IND 2018 (3)
- IND 2021 (8)
- IND Filed (2)
- INDIA 2020 (5)
- INDIA 2022 (3)
- Insomnia (1)
- Investigational device exemption (IDE) approval (2)
- JAPAN 2009 (1)
- JAPAN 2013 (1)
- JAPAN 2014 (1)
- JAPAN 2015 (2)
- JAPAN 2016 (1)
- japan 2017 (6)
- JAPAN 2018 (10)
- japan 2019 (8)
- japan 2020 (10)
- JAPAN 2021 (11)
- JAPAN 2022 (14)
- Japan marketing (34)
- Japan pipeline (38)
- Korea (3)
- korea 2012 (2)
- korea 2016 (1)
- korea 2019 (1)
- Lactams (1)
- MAA (3)
- Macrolides (2)
- Malaria (17)
- MANUFACTURING (8)
- Market info (3)
- Marketing authorisation application EU (4)
- MEDICAL DEVICE (9)
- MIGRAINE (1)
- molybdenum cofactor deficiency (1)
- MONKEY POX (1)
- Monoclonal antibody (89)
- Nanotechnology (5)
- NCE (2)
- NDA (51)
- NDA JAPAN (3)
- neuropathic pain (2)
- Neutraceuticals (1)
- New Drug Application Resubmission FDA (5)
- NEW DRUGS (76)
- New drugs canada (5)
- New drugs china (6)
- New drugs EU (10)
- New drugs Japan (2)
- NEW USE DRUGS (1)
- Newzealand (2)
- NICE (2)
- non-Hodgkin's lymphoma (1)
- NUCLEOTIDES (1)
- obesity (3)
- OCCULAR (2)
- ocular hypertension (1)
- organic chemistry (12)
- osteoarthritis (2)
- PAIN (2)
- Parkinson's disease (1)
- PATENT (93)
- PATENTS (82)
- Peptide drugs (56)
- PFIZER (1)
- PHASE 1 (90)
- PHASE 3 (25)
- PHASE1 (145)
- Phase2 drugs (282)
- Phase3 drugs (306)
- plaque psoriasis (1)
- POLYMORPH (10)
- Preclinical china (14)
- Preclinical drugs (121)
- Premture ejaculation (3)
- prime designation (1)
- Priority review (90)
- PROCESS (39)
- Promising clips (9)
- Psoriasis (1)
- QbD (2)
- QIDP (23)
- radio labelled (4)
- RADIOACTIVE AGENT (5)
- RADIOLABELLED (10)
- Rare disease (3)
- RARE PEDIATRIC DISEASE REVIEW (2)
- Regenerative medicine (4)
- Regulatory (74)
- Resubmission FDA (2)
- REVIEW (10)
- rheumatoid arthritis (3)
- SCHLEROSING AGENT (2)
- Scottish Medicines Consortium (1)
- sex arousal (2)
- SFDA FAST TRACK (2)
- SINGAPORE (4)
- sNDA (12)
- sNDA JAPAN (2)
- Solid tumours (2)
- spectroscopy (16)
- SPOTLIGHT (53)
- Stem cells (3)
- SYNTHESIS (10)
- TIVE (1)
- TOXINS (1)
- Translational medicine (2)
- Transverse myletis (2)
- Tropical Disease Priority Review Voucher (1)
- UK (1)
- UNANI MEDICINE (2)
- Uncategorized (2,278)
- US HERBS (7)
- USFDA SUBMISSION (1)
- vaccine (26)
- veterinary (8)
- virus (1)
- VITAMINS (3)
- x ray contrast agent (10)
Recent Comments
| shivkr2 on SPSR Excellence Award 2025 – S… | |
| Bonkasaurus on Infigratinib phosphate | |
| PALOVAROTENE | ORGAN… on Palovarotene | |
| Pfizer just purchase… on Temanogrel | |
| Pfizer To Cash In On… on Temanogrel |
SEARCH THIS BLOG
Novartis Kisqali® (ribociclib, LEE011) receives FDA approval as first-line treatment for HR+/HER2- metastatic breast cancer in combination with any aromatase inhibitor
March 16, 2017 10:06 am / Leave a comment


- Approved based on a first-line Phase III trial that met its primary endpoint of progression-free survival (PFS) at interim analysis due to superior efficacy compared to letrozole alone[1]
- At this interim analysis, Kisqali plus letrozole reduced risk of disease progression or death by 44% over letrozole alone, and demonstrated tumor burden reduction with a 53% overall response rate[1]
- Kisqali plus letrozole showed treatment benefit across all patient subgroups regardless of disease burden or tumor location[1]
- At a subsequent analysis with additional follow-up and progression events, a median PFS of 25.3 months for Kisqali plus letrozole and 16.0 months for letrozole alone was observed[2]
Basel, March 13, 2017 – The US Food and Drug Administration (FDA) has approved Kisqali®(ribociclib, formerly known as LEE011) in combination with an aromatase inhibitor as initial endocrine-based therapy for treatment of postmenopausal women with hormone receptor positive, human epidermal growth factor receptor-2 negative (HR+/HER2-) advanced or metastatic breast cancer.
Kisqali is a CDK4/6 inhibitor approved based on a first-line Phase III trial that met its primary endpoint early, demonstrating statistically significant improvement in progression-free survival (PFS) compared to letrozole alone at the first pre-planned interim analysis[1]. Kisqali was reviewed and approved under the FDA Breakthrough Therapy designation and Priority Review programs.
“Kisqali is emblematic of the innovation that Novartis continues to bring forward for people with HR+/HER2- metastatic breast cancer,” said Bruno Strigini, CEO, Novartis Oncology. “We at Novartis are proud of the comprehensive clinical program for Kisqali that has led to today’s approval and the new hope this medicine represents for patients and their families.”
The FDA approval is based on the superior efficacy and demonstrated safety of Kisqali plus letrozole versus letrozole alone in the pivotal Phase III MONALEESA-2 trial. The trial, which enrolled 668 postmenopausal women with HR+/HER2- advanced or metastatic breast cancer who received no prior systemic therapy for their advanced breast cancer, showed that Kisqali plus an aromatase inhibitor, letrozole, reduced the risk of progression or death by 44 percent over letrozole alone (median PFS not reached (95% CI: 19.3 months-not reached) vs. 14.7 months (95% CI: 13.0-16.5 months); HR=0.556 (95% CI: 0.429-0.720); p<0.0001)[1].
More than half of patients taking Kisqali plus letrozole remained alive and progression free at the time of interim analysis, therefore median PFS could not be determined[1]. At a subsequent analysis with additional 11-month follow-up and progression events, a median PFS of 25.3 months for Kisqali plus letrozole and 16.0 months for letrozole alone was observed[2]. Overall survival data is not yet mature and will be available at a later date.
“In the MONALEESA-2 trial, ribociclib plus letrozole reduced the risk of disease progression or death by 44 percent over letrozole alone, and more than half of patients (53%) with measurable disease taking ribociclib plus letrozole experienced a tumor burden reduction of at least 30 percent. This is a significant result for women with this serious form of breast cancer,” said Gabriel N. Hortobagyi, MD, Professor of Medicine, Department of Breast Medical Oncology, The University of Texas MD Anderson Cancer Center and MONALEESA-2 Principal Investigator. “These results affirm that combination therapy with a CDK4/6 inhibitor like ribociclib and an aromatase inhibitor should be a new standard of care for initial treatment of HR+ advanced breast cancer.”
Kisqali is taken with or without food as a once-daily oral dose of 600 mg (three 200 mg tablets) for three weeks, followed by one week off treatment. Kisqali is taken in combination with four weeks of any aromatase inhibitor[1].
Breast cancer is the second most common cancer in American women[3]. The American Cancer Society estimates more than 250,000 women will be diagnosed with invasive breast cancer in 2017[3]. Up to one-third of patients with early-stage breast cancer will subsequently develop metastatic disease[4].
Novartis is committed to providing patients with access to medicines, as well as resources and support to address a range of needs. The Kisqali patient support program is available to help guide eligible patients through the various aspects of getting started on treatment, from providing educational information to helping them understand their insurance coverage and identify potential financial assistance options. For more information, patients and healthcare professionals can call 1-800-282-7630.
The full prescribing information for Kisqali can be found at https://www.pharma.us.novartis.com/sites/www.pharma.us.novartis.com/files/kisqali.pdf(link is external).
About Kisqali® (ribociclib)
Kisqali (ribociclib) is a selective cyclin-dependent kinase inhibitor, a class of drugs that help slow the progression of cancer by inhibiting two proteins called cyclin-dependent kinase 4 and 6 (CDK4/6). These proteins, when over-activated, can enable cancer cells to grow and divide too quickly. Targeting CDK4/6 with enhanced precision may play a role in ensuring that cancer cells do not continue to replicate uncontrollably.
Kisqali was developed by the Novartis Institutes for BioMedical Research (NIBR) under a research collaboration with Astex Pharmaceuticals.
About the MONALEESA Clinical Trial Program
Novartis is continuing to assess Kisqali through the robust MONALEESA clinical trial program, which includes two additional Phase III trials, MONALEESA-3 and MONALEESA-7, that are evaluating Kisqali in multiple endocrine therapy combinations across a broad range of patients, including premenopausal women. MONALEESA-3 is evaluating Kisqali in combination with fulvestrant compared to fulvestrant alone in postmenopausal women with HR+/HER2- advanced breast cancer who have received no or a maximum of one prior endocrine therapy. MONALEESA-7 is investigating Kisqali in combination with endocrine therapy and goserelin compared to endocrine therapy and goserelin alone in premenopausal women with HR+/HER2- advanced breast cancer who have not previously received endocrine therapy.
About Novartis in Advanced Breast Cancer
For more than 25 years, Novartis has been at the forefront of driving scientific advancements for breast cancer patients and improving clinical practice in collaboration with the global community. With one of the most diverse breast cancer pipelines and the largest number of breast cancer compounds in development, Novartis leads the industry in discovery of new therapies and combinations, especially in HR+ advanced breast cancer, the most common form of the disease.
Kisqali® (ribociclib) Important Safety Information
Kisqali® (ribociclib) can cause a heart problem known as QT prolongation. This condition can cause an abnormal heartbeat and may lead to death. Patients should tell their healthcare provider right away if they have a change in their heartbeat (a fast or irregular heartbeat), or if they feel dizzy or faint. Kisqali can cause serious liver problems. Patients should tell their healthcare provider right away if they get any of the following signs and symptoms of liver problems: yellowing of the skin or the whites of the eyes (jaundice), dark or brown (tea-colored) urine, feeling very tired, loss of appetite, pain on the upper right side of the stomach area (abdomen), and bleeding or bruising more easily than normal. Low white blood cell counts are very common when taking Kisqali and may result in infections that may be severe. Patients should tell their healthcare provider right away if they have signs and symptoms of low white blood cell counts or infections such as fever and chills. Before taking Kisqali, patients should tell their healthcare provider if they are pregnant, or plan to become pregnant as Kisqali can harm an unborn baby. Females who are able to become pregnant and who take Kisqali should use effective birth control during treatment and for at least 3 weeks after the last dose of Kisqali. Do not breastfeed during treatment with Kisqali and for at least 3 weeks after the last dose of Kisqali. Patients should tell their healthcare provider about all of the medicines they take, including prescription and over-the-counter medicines, vitamins, and herbal supplements since they may interact with Kisqali. Patients should avoid pomegranate or pomegranate juice, and grapefruit or grapefruit juice while taking Kisqali. The most common side effects (incidence >=20%) of Kisqali when used with letrozole include white blood cell count decreases, nausea, tiredness, diarrhea, hair thinning or hair loss, vomiting, constipation, headache, and back pain. The most common grade 3/4 side effects in the Kisqali + letrozole arm (incidence >2%) were low neutrophils, low leukocytes, abnormal liver function tests, low lymphocytes, and vomiting. Abnormalities were observed in hematology and clinical chemistry laboratory tests.
Please see the Full Prescribing Information for Kisqali, available at https://www.pharma.us.novartis.com/sites/www.pharma.us.novartis.com/files/kisqali.pdf(link is external).
About Novartis
Novartis provides innovative healthcare solutions that address the evolving needs of patients and societies. Headquartered in Basel, Switzerland, Novartis offers a diversified portfolio to best meet these needs: innovative medicines, cost-saving generic and biosimilar pharmaceuticals and eye care. Novartis has leading positions globally in each of these areas. In 2016, the Group achieved net sales of USD 48.5 billion, while R&D throughout the Group amounted to approximately USD 9.0 billion. Novartis Group companies employ approximately 118,000 full-time-equivalent associates. Novartis products are sold in approximately 155 countries around the world. For more information, please visit http://www.novartis.com.
Novartis is on Twitter. Sign up to follow @Novartis and @NovartisCancer at http://twitter.com/novartis(link is external) and http://twitter.com/novartiscancer (link is external)
For Novartis multimedia content, please visit www.novartis.com/news/media-library
For questions about the site or required registration, please contact media.relations@novartis.com
References
[1] Kisqali (ribociclib) Prescribing information. East Hanover, New Jersey, USA: Novartis Pharmaceuticals Corporation; March 2016.
[2] Novartis Data on File
[3] American Cancer Society. How Common Is Breast Cancer? Available at https://www.cancer.org/cancer/breast-cancer/about/how-common-is-breast-cancer.html(link is external). Accessed January 23, 2017.
[4] O’Shaughnessy J. Extending survival with chemotherapy in metastatic breast cancer. The Oncologist. 2005;10(Suppl 3):20-29.
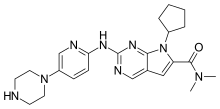
рибоциклиб , ريبوسيكليب , 瑞波西利
Ribociclib « New Drug Approvals
////////////////Novartis, Kisqali®, ribociclib, LEE011, FDA 2017, HR+/HER2- metastatic breast cancer, рибоциклиб , ريبوسيكليب , 瑞波西利
Share this:
- Print (Opens in new window) Print
- Share on X (Opens in new window) X
- Email a link to a friend (Opens in new window) Email
- Share on Pinterest (Opens in new window) Pinterest
- Share on LinkedIn (Opens in new window) LinkedIn
- Share on Mastodon (Opens in new window) Mastodon
- Share on Tumblr (Opens in new window) Tumblr
- Share on Reddit (Opens in new window) Reddit
- Share on Facebook (Opens in new window) Facebook











