Home » Posts tagged 'Bysanti'
Tag Archives: Bysanti
Milsaperidone
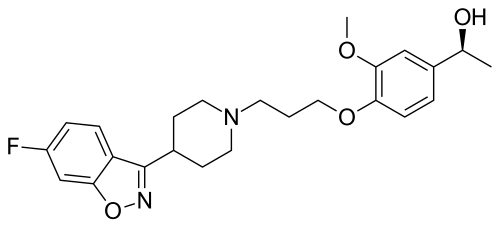
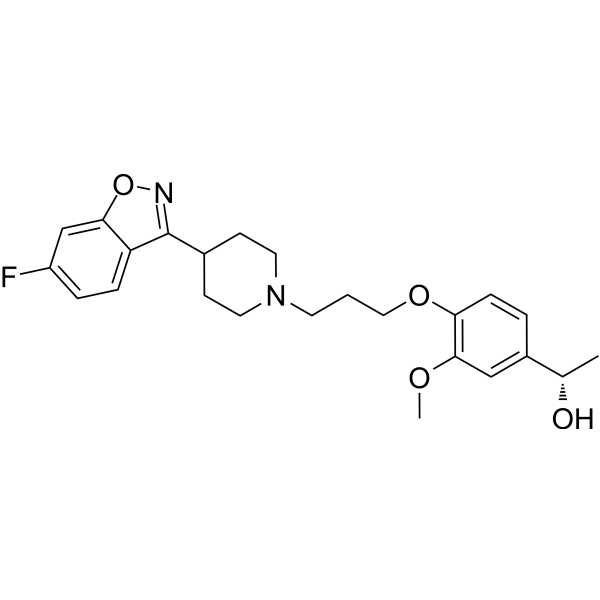
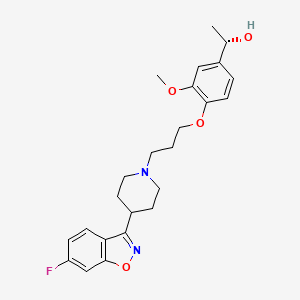
Milsaperidone
CAS 501373-88-2
C24H29FN2O4 MW 428.50
FDA APPROVED 2/20/2026, Bysanti, To treat schizophrenia and to treat manic or mixed episodes associated with bipolar I disorder
(1S)-1-[4-[3-[4-(6-fluoro-1,2-benzoxazol-3-yl)piperidin-1-yl]propoxy]-3-methoxyphenyl]ethanol
7SV1ZOG031, P-88-8991, (-)-, (S)-Hydroxy Iloperidone, P-88, VHX 869, VHX-896
Milsaperidone (INNTooltip International Nonproprietary Name, USANTooltip United States Adopted Name), also known by its developmental code name VHX-896 and its tentative brand name Bysanti, is an atypical antipsychotic which is pending approval for the treatment of schizophrenia and bipolar disorder and is in phase 3 clinical trials for treatment of major depressive disorder.[1][2][3] It is a prodrug of iloperidone (Fanapt) and acts as a dopamine D2 receptor and serotonin 5-HT2A receptor antagonist, among other actions.[1][4][5] The drug was developed by Vanda Pharmaceuticals.[1]
- OriginatorVanda Pharmaceuticals
- Class2 ring heterocyclic compounds; Alcohols; Anisoles; Antidepressants; Antipsychotics; Ethers; Fluorobenzenes; Isoxazoles; Methyl ethers; Mood stabilisers; Phenyl ethers; Piperidones; Small molecules
- Mechanism of ActionAlpha 1 adrenergic receptor antagonists; Dopamine D2 receptor antagonists; Serotonin 5-HT2 receptor antagonists
- RegisteredBipolar disorders; Schizophrenia
- 25 Feb 2026Chemical structure information added.
- 25 Feb 2026Vanda Pharmaceuticals has patent protection for an improved method of treatment with milsaperidone in USA
- 25 Feb 2026Vanda Pharmaceuticals has patents pending for an improved method of treatment with milsaperidone in China, Australia, Israel, Mexico and worldwide
PAT
Example 1
(S)-1-(4-(3-r4-(6-Fluoro-benzofd1isoxazol-3-vπ-piperidin-1-vπ-propoxy)-3-methoxy-phenvπ-ethanol
56.36 g of boran complex of (3aR, 7R)-1-methyl-3,3-diphenyl-tetrahydro-pyrrolo[1,2-c][1 ,3,2]oxazaborole (1 equivalent) is dissolved under nitrogen in methylenchloride, and the solution is cooled to 0°C. A 1M solution of 1-(4-{3-[4-(6-fluoro-benzo[d]isoxazol-3-yl)-piperidin-1-yl]-propoxy}-3-methoxy-phenyl)-ethanone (iloperidone; 1 equivalent) in methylenchloride is added via a dropping funnel over 90 minutes while the internal temperature is maintained at 0°C ± 2°C. After the addition is complete, the mixture is stirred at 0°C for 20 hours. The reaction mixture is then poured into precooled methanol (0-5°C) during 1 hour. The solution is warmed to room temperature and stirred until the H2 evolution ceases. The solution is concentrated by distillation and the residue dried in vacuum, treated with methanol and stirred for about 1 hour at 50°C and an additional hour at 0CC. The product is isolated by filtration and dried under reduced pressure for 3 hours at 50°C. The title compound is obtained (white crystals).
[α]D20– 19.3° (c=1 in chloroform)
Mp: 138.2 – 138.8°C
The boran complex used as starting material can be obtained as follows:
200 ml of a solution of (3aR, 7R)-1-methyl-3,3-diphenyl-tetrahydro-pyrrolo[1,2-c][1,3,2]oxazaborole (1M in toluene) is stirred at room temperature under nitrogen. 1.2 equivalent borane-dimethylsulfide complex is added with a syringe. The solution is stirred for 2 further hours at room temperature. The borane complex is then crystallised by addition of 4 vol dry hexane and cooling to -12°C for 1.5 hour. The product is isolated by filtration in a sintered glass funnel and dried in vacuum at 40°C. The boran complex is obtained /white crystals).
Example 2
(R)-1-(4-(3-r4-(6-Fluoro-benzord1isoxazol-3-vπ-piperidin-1-vn-propoxy)-3-methoxy-phenlvπ-ethanol
This compound is produced in analogy to Example 1, using boran complex of (3aS, 7R)-1-methyl-3,3-diphenyl-tetrahydro-pyrrolo[1,2-c][1,3,2]oxazaborole.
[α]D20 = + 18.4° (c=1 in chloroform)
Mp: 137.9 – 138.3°C
PAT
- OPTICAL ISOMERS OF AN ILOPERIDONE METABOLITE.Publication Number: ES-2370634-T3Priority Date: 2001-08-31Grant Date: 2011-12-21
- Optical isomers of an iloperidone metabolitePublication Number: US-2013296366-A1Priority Date: 2001-08-31
- Optical isomers of iloperidone metabolitesPublication Number: JP-2015214577-APriority Date: 2001-08-31
- Optical isomers of iloperidone metabolitesPublication Number: JP-2015227368-APriority Date: 2001-08-31
- Optical isomers of an Iloperidone metabolitePublication Number: US-7977356-B2Priority Date: 2001-08-31Grant Date: 2011-07-12
- Optical isomers of an iloperidone metabolitePublication Number: US-2011201646-A1Priority Date: 2001-08-31
- Optical isomers of an iloperidone metabolitePublication Number: EP-1425272-A1Priority Date: 2001-08-31
- Optical isomers of iloperidone metabolitesPublication Number: JP-2013116905-APriority Date: 2001-08-31
- Optical isomers of iloperidone metabolitesPublication Number: JP-2013227335-APriority Date: 2001-08-31
- Optical isomers of an iloperidone metabolitePublication Number: US-2009176739-A1Priority Date: 2001-08-31
- Optical isomers of an iloperidone metabolitePublication Number: US-2005020632-A1Priority Date: 2001-08-31
- Optical isomers of an iloperidone metabolitePublication Number: EP-2305656-B1Priority Date: 2001-08-31Grant Date: 2012-10-24



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
- “Vanda Pharmaceuticals”. AdisInsight. 30 July 2025. Retrieved 11 October 2025.
- IsHak WW, Hirsch D, Renteria S, Totlani J, Murphy N, Chang T, et al. (October 2025). “Depressive disorders: systematic review of approved psychiatric medications (2009-April 2025) and pipeline phase 3 medications”. BMC Psychiatry. 25 (1) 939. doi:10.1186/s12888-025-07141-3. PMC 12506068. PMID 41057811.
- Richmond LM (1 June 2025). “Med Check: FDA Accepts Bysanti Application, Cobenfy Fails as Adjunct, and More”. Psychiatric News. 60 (6) appi.pn.2025.06.6.2. doi:10.1176/appi.pn.2025.06.6.2. ISSN 0033-2704. Retrieved 11 October 2025.
- Kang J (9 May 2025). “Milsaperidone Under Review for Bipolar I Disorder and Schizophrenia”. MPR. Retrieved 11 October 2025.
- “Vanda Announces Bysanti™ NDA Filing; FDA Decision Expected in Early 2026”. BioSpace. 5 May 2025. Retrieved 11 October 2025.
| Clinical data | |
|---|---|
| Trade names | Bysanti |
| Other names | VHX896 |
| Routes of administration | Oral |
| Drug class | Atypical antipsychotic |
| Identifiers | |
| IUPAC name | |
| CAS Number | 501373-88-2 |
| PubChem CID | 10365268 |
| ChemSpider | 8540717 |
| UNII | 7SV1ZOG031 |
| KEGG | D13099 |
| Chemical and physical data | |
| Formula | C24H29FN2O4 |
| Molar mass | 428.504 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| SMILES | |
| InChI | |
////////////milsaperidone, FDA 2026, APPROVALS 2026, Bysanti, schizophrenia, 7SV1ZOG031, P-88-8991, (-)-, (S)-Hydroxy Iloperidone, P-88, VHX 869, VHX-896

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....










