Home » Posts tagged 'ALXN1101)'
Tag Archives: ALXN1101)
Alexion obtains FDA breakthrough therapy status for cPMP to treat MoCD type A disorder

cyclic pyranopterin monophosphate (cPMP, ALXN1101)
Alexion Pharma International Sàrl has received a breakthrough therapy designation from the US Food and Drug Administration (FDA) for its cyclic pyranopterin monophosphate (cPMP, ALXN1101), an enzyme co-factor replacement therapy to treat patients with molybdenum cofactor deficiency (MoCD) type A.
Alexion obtains FDA breakthrough therapy status for cPMP to treat MoCD type A disorder
read all at
Cyclic pyranopterin monophosphate (cPMP) is an experimental treatment formolybdenum cofactor deficiency type A, which was developed by José Santamaría-Araujo and Schwarz at the German universities TU Braunschweig and the University of Cologne.[1][2]
cPMP is a precursor to molybdenum cofactor, which is required for the enyzme activity ofsulfite oxidase, xanthine dehydrogenase/oxidase and aldehyde oxidase.[3]
- Guenter Schwarz Laboratory, Institute for Biochemistry – University of Cologne (English, German)
- Günter Schwarz, José Angel Santamaria-Araujo, Stefan Wolf, Heon-Jin Lee, Ibrahim M. Adham, Hermann-Josef Gröne, Herbert Schwegler, Jörn Oliver Sass, Tanja Otte, Petra Hänzelmann, Ralf R. Mendel, Wolfgang Engel and Jochen Reiss (2004). “Rescue of lethal molybdenum cofactor deficiency by a biosynthetic precursor from Escherichia coli“. Human Molecular Genetics 13 (12): 1249–1255. doi:10.1093/hmg/ddh136.PMID 15115759.
- Doctors risk untried drug to stop baby’s brain dissolving, TimesOnline, November 5, 2009
- José Angel Santamaria-Araujo, Berthold Fischer, Tanja Otte, Manfred Nimtz, Ralf R. Mendel, Victor Wray and Günter Schwarz (2004). “The Tetrahydropyranopterin Structure of the Sulfur-free and Metal-free Molybdenum Cofactor Precursor”. The Journal of Biological Chemistry 279 (16): 15994–15999.doi:10.1074/jbc.M311815200. PMID 14761975.
Molybdenum cofactor (Moco) deficiency is a pleiotropic genetic disorder. Moco consists of molybdenum covalently bound to one or two dithiolates attached to a unique tricyclic pterin moiety commonly referred to as molybdopterin (MPT). Moco is synthesized by a biosynthetic pathway that can be divided into four steps, according to the biosynthetic intermediates precursor Z (cyclicpyranopterin monophosphate; cPMP), MPT, and adenylated MPT. Mutations in the Moco biosynthetase genes result in the loss of production of the molybdenum dependent enzymes sulfite-oxidase, xanthine oxidoreductase, and aldehyde oxidase. Whereas the activities of all three of these cofactor-containing enzymes are impaired by cofactor deficiency, the devastating consequences of the disease can be traced to the loss of sulfite oxidase activity. Human Moco deficiency is a rare but severe disorder accompanied by serious neurological symptoms including attenuated growth of the brain, unbeatable seizures, dislocated ocular lenses, and mental retardation. Until recently, no effective therapy was available and afflicted patients suffering from Moco deficiency died in early infancy.
It has been found that administration of the molybdopterin derivative precursor Z, a relatively stable intermediate in the Moco biosynthetic pathway, is an effective means of therapy for human Moco deficiency and associated diseases related to altered Moco synthesis {see U.S. Patent No. 7,504,095). As with most replacement therapies for illnesses, however, the treatment is limited by the availability of the therapeutic active agent.
WO 2012112922 A1
In this synthesis, the deprotection may involve, for example, either sequential or one-pot deprotection of certain amino and hydroxyl protecting groups on a compound of formula (VII) to furnish the compound of formula (I). Suitable reagents and conditions for the deprotection of a compound of formula (VII) can be readily determined by those of ordinary skill in the art. For example, compound (I) may be formed upon treatment of a compound of formula (VII) under conditions so that hydroxyl protecting groups, such as acetate, isopropylidine, and benzylidine protecting groups, are removed from the formula (VII) structure. The acetate group can be cleaved, for example, under Zemplen conditions using catalytic NaOMe as a base in methanol. The benzylidene and isopropylidene groups can be cleaved by hydrogenation or using acidic hydrolysis as reported by R.M. Harm et ah, J. Am. Chem. Soc, 72, 561 (1950). In yet another example, the deprotection can be performed so that amino protecting groups, such as 9- fluorenylmethyl carbamate (Fmoc), t-butyl carbamate (Boc), and carboxybenzyl carbamate (cbz) protecting groups are cleaved from the compound of formula (VII). 9-fluorenylmethyl carbamate (Fmoc) can be removed under mild conditions with an amine base (e.g. , piperidine) to afford the free amine and dibenzofulvene, as described by E. Atherton et al, “The
Fluorenylmethoxycarbonyl Amino Protecting Group,” in The Peptides, S. Udenfriend and J. Meienhofer, Academic Press, New York, 1987, p. 1. t-butyl carbamate (Boc) can be removed, as reported by G.L. Stahl et al., J. Org. Chem., 43, 2285 (1978), under acidic conditions (e.g., 3 M HC1 in EtOAc). Hydrogenation can be used to cleave the carboxybenzyl carbamate (cbz) protecting group as described by J. Meienhofer et al., Tetrahedron Lett., 29, 2983 (1988).
To prevent oxidation of formula (I) during the reaction, the deprotection may be performed under anaerobic conditions. The deprotection may also be performed at ambient temperature or at temperatures of from about 20 – 60 °C (e.g. , 25, 30, 35, 40, 45, 50, or 55 °C).
The compound of formula (I) may be isolated in the form of a pharmaceutically acceptable salt. For example, the compound of formula (I) may be crystallized in the presence of HC1 to form the HC1 salt form of the compound. In some embodiments, the compound of formula (I) may be crystallized as the HBr salt form of the compound. The compound of formula (I) may also be isolated, e.g., by precipitation as a sodium salt by treating with NaOH. The compound of formula (I) is labile under certain reaction and storage conditions. In some embodiments, the final solution comprising the compound of formula (I) may be acidified by methods known in the art. For example, the compound of formula (I), if stored in solution, can be stored in an acidic solution.
In some embodiments, the compound of formula (I) may be prepared, for example, by: reacting a compound of formula (II- A):
with a compound of formula (III- A):
in the presence of a hydrazine to produce a compound of formula (IV- A):
selectively protecting the compound of formula (IV-A) to prepare a compound of formula (V-A):
wherein:
Rj is a protecting group, as defined above;
phosphorylating the compound of formula (V-A) to prepare a compound of formula (VI- A):
oxidizing the compound of formula (VI-A) to prepare a compound of formula (VII- A):
; and deprotecting the compound of formula (VII-A) to prepare the compound of formula (I). For example, a compound of formula (I) can be prepared as shown in Scheme 3.
Scheme 3.
5 R = Fraoc
In another embodiment, the compound of formula (I) is prepared by:
reacting a compound of formula (II- A):
with a compound of formula (III- A):
in the presence of a hydrazine to produce a compound of formula (IV-A):
selectively protecting the compound of formula (IV-A) to prepare a compound of formula (V-B):
wherein:
each Ri is independently a protecting group, as defined above;
phosphorylating the compound of formula (V-B) to prepare a compound of formula (VI-B):
oxidizing the compound of formula (VI-B) to prepare a compound of formula (VII-B):
; and deprotecting the compound of formula (VII-B) to prepare the compound of formula (I), example, a compound of formula (I) can be prepared as shown in Scheme 4.
Scheme 4.
Alternatively, a compound of formula (I) can be formed as shown in Scheme 5. A diaminopyrimidinone compound of formula (II) can be coupled with a phosphorylated hexose sugar of formula (VIII), to give a compound of formula (IX). The piperizine ring nitrogen atoms can be protected to give a compound of formula (X) which can be oxidized to give a diol of formula (XI). The diol of formula (XI) can then be deprotected using appropriate conditions and converted to the compound of formula (I).
Scheme 5
In this embodiment, the phosphate may be introduced at the beginning of the synthesis to avoid undesirable equilibrium between the pyrano and furano isomers during subsequent steps of the synthesis. For example, a compound of formula (I) can be prepared as shown in Scheme 6.
Scheme 6.
ridine
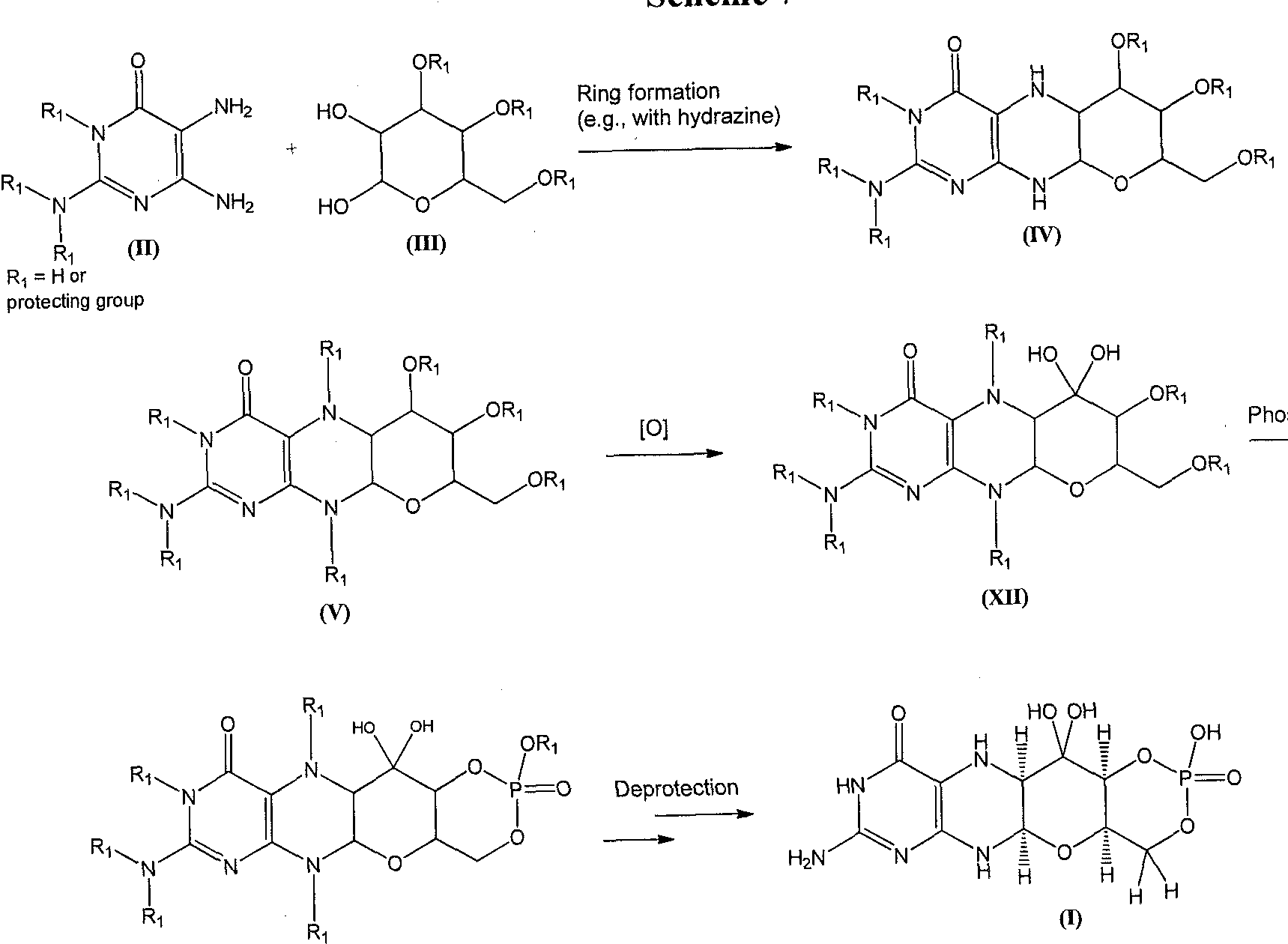
A compound of formula (I) can also be formed as shown in Scheme 7. A diaminopyrimidinone compound of formula (II) can be coupled to a compound of formula (III) to afford the piperizine derivative of formula (IV). The piperizine ring nitrogen atoms of the compound of formula (IV) can be protected under standard conditions to give a derivative of formula (V). The formula (V) structure can be oxidized to afford compounds of formula (XII). Phosphorylation of a compound of formula (XII) gives a compound of formula (VII). Global deprotection of the compound of formula (VII) can afford the compound of formula (I).
Scheme 7
Piperizine ring protection
sphorylation
(VII)
For example, a compound of formula (I) can be prepared as shown in Scheme 8.
Scheme 8.

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....