EIDD-2173, ATI-2173, Fosclevudine alafenamide


EIDD-2173; also known as ATI-2173
Fosclevudine alafenamide
Phase 2
| Molecular Weight | 529.45 |
|---|---|
| Formula | C22H29FN3O9P |
| CAS No. | 1951476-79-1 |
Hepatitis B virus (HBV) is an infectious disease that targets the liver resulting in either an acute infection, with symptoms arising in 45 to 160 days, or a chronic infection, which 350 million people worldwide are affected by. Estimates indicate that 600,000 deaths occur each year as a result of consequences related to HBV infection. HBV possesses a 3.2- kb relaxed circular DNA (rcDNA) genome that is used to form covalently closed circular DNA (cccDNA) in a host cell. The cccDNA is then transcribed by RNA polymerase II, a host DNA-dependent RNA polymerase, to produce pregenomic RNA (pgRNA). The pgRNA is then used by the virally encoded reverse transcriptase to form rcDNA. The goals of current treatments for chronic HBV infections are to reduce HBV replication and reduce liver damage.
Current treatments for chronic HBV infections include pegylated alpha interferon and nucleoside/nucleotide reverse transcriptase inhibitors (NRTIs). NRTIs are converted to their corresponding 5 ‘-triphosphate, or diphosphate in the case of phosphonate containing NRTIs, and reduce viral replication by inhibiting the HBV encoded polymerase. Clevudine is an NRTI that is no longer being developed for the treatment of chronic HBV because of drug-related skeletal myopathy that was a result of mitochondrial dysfunction in patients.
Interestingly, clevudine triphosphate has been shown to be a competitive nonsubstrate inhibitor of the HBV encoded polymerase, and due to its long intracellular half-life, is able to suppress HBV replication for an extended period of time after drug withdrawal.
The discovery and synthesis of the (S,S) and (S,R) diastereomers of clevudine phosphoramidate has been previously reported. These studies were undertaken to address the myopathy concerns associated with clevudine. The phosphoramidate moiety was utilized to deliver clevudine, as its 5 ‘-monophosphate, to the liver reducing 1) systemic exposure to clevudine and 2) the possibility of skeletal myopathy. Both phosphoramidates showed anti-HBV activity similar to clevudine with the (S,S) diastereomer being slightly more potent.
SYN
See U.S. Patent No. 10,683,319.
PATENT
WO20 17223421
PATENT




EXAMPLE 11 :
[0374] Preparation of intermediate ATI-2173 from Compound-10.
[0375] Experimental Procedure
[0376] (H-l) Crude Compound-10 in THF (see Example 10) was divided into three aliquots and stirred with 2% HC1 aq. solution at pH 5-6, 4-5, and 3-4; for 16 h at 20-25 °C; the aliquots were combined and stirred at 15-20 °C for 72 h, with no degradation of ATI-2173 observed over the second time period;
[0377] (H-2) the pH of the mixture was adjusted to 7 with 7% NaHCCh;
[0378] (H-3) phase separation was carried out using 2-MeTHF, the organic phase was washed with NA2SO4 aqueous soltion, then concentrated to 1-3 V, and MTBE (5 V) was added; this operation was repeated twice;
[0379] (H-5) ATI-2173 was precipitated gradually upon addition of seed crystal and addition of n-heptane (5 V);
[0380] (H-6) the product was filtered; and
[0381] (H-7) the wetcake was dried, resulting in ATI-2173 in 99.73a% purity and
84.4% yield.
[0382] EXAMPLE 12:
[0383] Crystallization of ATI-2173
[0384] Initial studies examined the use of single or mixed solvent systems to crystalize the amorphous product, ATI-2173. Several solvent conditions were screened, including single solvent and mixed solvent systems, in order to determine the potential for obtaining a crystalline material from the amorphous material. None of the solvents tested worked and all conditions produced an oil product. The results are shown below in Tables 8 and 9.
[0385] Table 8: Single Solvent Systems


AS ON DEC2021 3,491,869 VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@amcrasto
///////////
////////// EIDD-2173, ATI-2173, EIDD 2173, ATI 2173, Hepatitis B virus, ANTI HBV, Fosclevudine alafenamide, PHASE 2

NEW DRUG APPROVALS
ONE TIME
$10.00
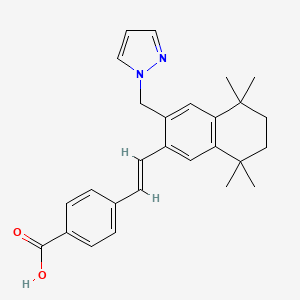
Palovarotene

Palovarotene
CAS 410528-02-8
4-[(E)-2-[5,5,8,8-tetramethyl-3-(pyrazol-1-ylmethyl)-6,7-dihydronaphthalen-2-yl]ethenyl]benzoic acid
FDA 8/16/2023
To reduce the volume of new heterotopic ossification in adults and pediatric patients (aged 8 years and older for females and 10 years and older for males) with fibrodysplasia ossificans progressiva
- RG-667
- RO-3300074
Palovarotene, sold under the brand name Sohonos, is a medication used for the treatment of heterotopic ossification and fibrodysplasia ossificans progressiva.[4][5] It is a highly selective retinoic acid receptor gamma (RARγ) agonist.[6]
It was approved for medical use in Canada in June 2022,[4] and in the United States in August 2023.[5]
Medical uses
Palovarotene is indicated for the treatment of heterotopic ossification and fibrodysplasia ossificans progressiva.[4][5]
History
Palovarotene is a retinoic acid receptor gamma (RARγ) agonist licensed to Clementia Pharmaceuticals from Roche Pharmaceuticals. At Roche, palovarotene was evaluated in more than 800 individuals including healthy volunteers and patients with chronic obstructive pulmonary disease (COPD).[7] A one-year trial did not demonstrate a significant benefit on lung density in moderate-to-severe emphysema secondary to severe α(1)-antitrypsin deficiency.[8]
In 2011, animal studies demonstrated that RARγ agonists, including palovarotene, blocked new bone formation in both an injury-induced mouse model of heterotopic ossification (HO) and a genetically modified biological mouse model of fibrodysplasia ossificans progressiva containing a continuously active ACVR1/ALK2 receptor in a dose-dependent manner.[9][10] A 2016 study demonstrated that palovarotene also inhibited spontaneous heterotopic ossification, maintained limb mobility and functioning, and restored skeletal growth in fibrodysplasia ossificans progressiva mouse models.[11]
Society and culture
Legal status
Palovarotene is being developed by Ipsen Biopharmaceuticals and was granted priority review and orphan drug designations by the United States Food and Drug Administration (FDA) for the treatment of fibrodysplasia ossificans progressiva[12][13] and orphan medicinal product designation by the European Medicines Agency (EMA) in 2014.[14][15][16][17] Phase II clinical studies failed to show a significant change in heterotopic bone volume, the main outcome measure, but prompted further investigation in a phase III clinical trial.[18] In December 2022, the FDA declined to approve palovarotene for the fibrodysplasia ossificans progressive without additional clinical trial data.[19] In January 2023, the European Medicines Agency (EMA) recommended the refusal of the marketing authorization for palovarotene for the treatment of fibrodysplasia ossificans progressiva.[20]
Research
Phase II
Clementia submitted a new drug application for palovarotene for the treatment of fibrodysplasia ossificans progressiva after observing positive phase II results.[21]
Phase III
In December 2019, Ipsen issued a partial clinical hold for people under the age of 14, due to reports of early fusion of growth plates.[22] Ipsen acquired Clementia in 2019.[23]
SYN
J. Med. Chem. 2025, 68, 2147−2182
Palovarotene (Sohonos). Palovarotene (7) is a selective retinoic acid receptor γ (RARγ) agonist that was
developed for the treatment of fibrodysplasia ossificans progressiva (FOP), a very rare autosomal dominant disorder, impacting ∼1 in2million individuals worldwide. 54,55 This orally bioavailable agonist reduces the incidence of heterotopic ossification in patients with FOP and was developed by the
French biopharmaceutical company Ipsen. 56 The small Molecule agonist was originally developed by Roche for a different indication, and was later licensed to Clementia Pharmaceuticals, which was ultimately acquired by Ipsen.
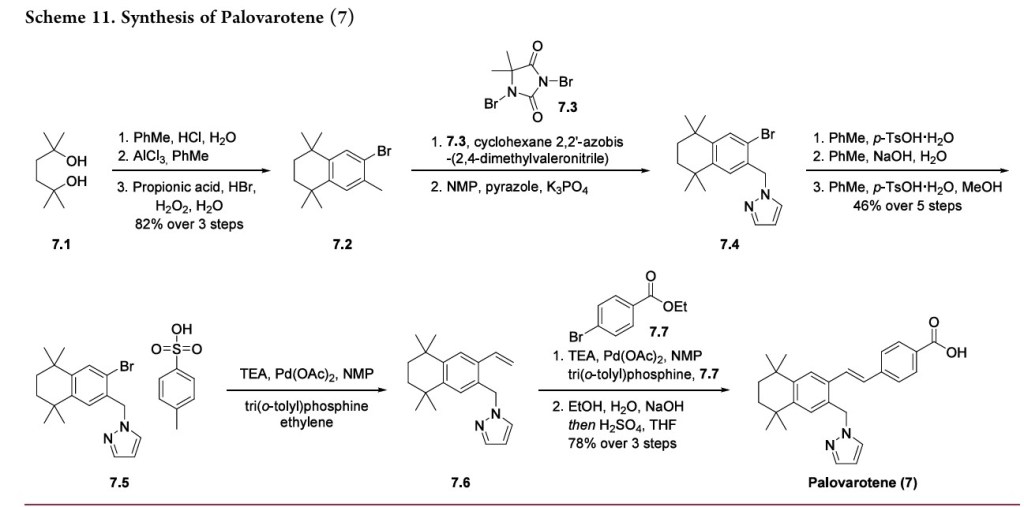
AlthoughapprovedbytheUSFDAinAugust2023,palovarotene was first approved by Health Canada in January 2022 for patients with FOP inadults andchildren aged 10 years and older for males and aged 8 years and older for females. With respect to pharmacodynamics, the agonist binds to RARγ and thus inhibits bone morphogenetic protein and Smad 1/5/8 signaling.57 This signaling inhibition permits normal muscle tissue repair and ultimately reduces the incidence of heterotopic ossification. A robust kilogram-scale synthesis of palovarotene has been disclosed in a patent by Roche and is depicted in Scheme 11.58
Starting from 2,5-dimethyl-2,5-hexanediol (7.1), the two tertiary alcohols were chlorinated with concentrated hydro chloric acid in toluene. Without isolation, the resulting

(54) Wentworth, K. L.; Masharani, U.; Hsiao, E. C. Therapeutic
advances for blocking heterotopic ossification in fibrodysplasia
ossificans progressiva. Br. J. Clin. Pharmacol. 2019, 85, 1180−1187.
(55) Semler, O.; Rehberg, M.; Mehdiani, N.; Jackels, M.; Hoyer
Kuhn, H. Current and emerging therapeutic options for the
management of rare skeletal diseases. Paediatr. Drugs 2019, 21, 95−
106.
(56) Hoy, S. M. Palovarotene: first approval. Drugs 2022, 82, 711−
716.
(57) Pignolo, R. J.; Pacifici, M. Retinoid agonists in the targeting of
heterotopic ossification. Cells 2021, 10, 3245.
(58) Martin, M. Process for preparing retinoid compounds. US
20070232810, 2007.
.
SYN
Desjardins, C., Grogan, D. R., Packman, J. N., & Harnett, M. (2017). Methods for treating heterotopic ossification (WO2017210792A1). World Intellectual Property Organization. https://patents.google.com/patent/WO2017210792A1
Chemical Communications (Cambridge, United Kingdom) (2019), 55(38), 5420-5422
WO2014105446
US20070232810
Patent
https://patents.google.com/patent/WO2002028810A3/en
WO2002028810


XAMPLE 12: PREPARATION OF 4-r(E)-2-(5,5.8.8-TETRAMETHYL-3-PYRAZOL-l-YLMETHYL -5.6.7.8-TETRAHYDRO-NAPHTHALEN-2-YL VINYLl BENZOIC ACID (6)
A mixture of 2.0 g (4.5 mmol) of (E)- methyl-4-[2-(3-bromomethyl-5,5,8,8-tetramethyl-5,6,7,8-tetrahydro-naphthalen-2-yl)vinyl]benzoate and 0.65 g (9.5 mmol) of pyrazole in 15 mL of N-methyl pyrrolidine was heated at 100°. After 2 hours, the reaction mixture was cooled to room temperature, poured into brine and extracted with ethyl acetate. The organic extracts were washed with brine, dried over sodium sulfate and concentrated under reduced pressure. The residue was stirred with hexane and the product was filtered off, washed with hexane and dried to give 1.6 g (83%) of methyl-4-[2-(5,5,8,8-Tetramethyl-3-pyrazol-l-ylmethyl-5,6,7,8-tetrahydro-naphthalen-2-yl)vinyl]benzoate (M+ = 429).
A mixture of 27.6 g (64.4 mmol) of methyl-4-[2-(5,5,8,8-tetramethyl-3-pyrazol-l-ylmethyl-5,6,7,8-tetrahydro-naphthalen-2-yl)vinyl]benzoate and 97 mL (193 mmol) of 2 N sodium hydroxide in 300 mL of ethyl alcohol was heated at reflux. After 1 hour, the reaction mixture was cooled to room temperature and diluted with 900 mL of water. The reaction mixture was acidified with 2 N HCl and the product was isolated by filtration, washed with water and pentane and dried to give 25.9 g (97%) of 4-[(E)-2-(5,5,8,8-tetramethyl-3-pyrazol-l-ylmethyl-5,6,7,8-tetrahydro-naphthalen-2-yl)vinyl]benzoic acid (m.p. = 246.5-248 °C) 6.
Proceeding as described in the example above but substituting pyrazole with pyrrole, 4-methylpyrazole, 1,2,4-triazole, moφholine, 2-pyrrohdone, 3,5-dimethylpyrzole,
δ – valerolactone, 2-methyhmidazole and 4-methylimidzole gave 4-[(E)-2-(5,5,8,8-tetramethyl-3-pyrrol-l-ylmethyl-5,6,7,8-tetrahydro-naphthalen-2-yl)vinyl]benzoic acid 7, 4-{(E)-2-[5,5,8,8-Tetramemyl-3-(4-methylpyrazol-l-ylmethyl-5,6,7,8-tetrahydro-naphthalen-2-yl)vinyl]benzoic acid 20, 4-[(E)-2-(5,5,8,8-Tetxamethyl-3-[l,2,4]triazol-l-ylmethyl-5,6,7,8Jetrahydro-naphthalen-2-yl]vinyl}benzoic acid 39, 4-[(E)-2-(5,5,8,8-tetramethyl-3-moφhohn-4-ylmethyl- 5,6,7,8-tetrahydro-naphthalen-2-yl)vinyl]benzoic acid 138, 4-[(E)-2-(5,5,8,8-tetramethyl-3- (2-oxo-pyrrohdin-l-yl-methyl-5,6,7,8-tetrahydro-naphthalen-2-yl)vinyl]benzoic acid 139, 4-{(E)-2-[5,5,8,8-Tetramet yl-3-(3,5-mmemylpyτazol-l-yhnethyl-5,6,7,8-tetrahydro-napn^ 2-yl)vinyl]benzoic acid 143, 4-[(E)-2-(5,5,8,8-tetramethyl-3-(2-oxo-piperidin-l-yl-methyl-5,6,7,8-tetrahydro-naρhthalen-2-yl)vinyl]benzoic acid 146 4-{(E)-2-[5,5,8,8-Tetramethyl-3-(2-methyhmidazol-l-ylmethyl-5,6,7,8-tetrahydro-naphthalen-2-yl)vinyl]benzoic acid 149and 4-{(E)-2-[5,5,8,8-Tetramethyl-3-(4-methyhmidazol-l-ylmethyl-5,6,7,8-tettahydro-naphthalen-2-yl)vinyl]benzoic acid 150 respectively.


AS ON DEC2021 3,491,869 VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@amcrasto
///////////
| Clinical data | |
|---|---|
| Trade names | Sohonos |
| Other names | R-667, RG-667 |
| License data | US DailyMed: Palovarotene |
| Routes of administration | By mouth |
| Drug class | Retinoic acid receptor gamma agonist |
| ATC code | M09AX11 (WHO) |
| Legal status | |
| Legal status | CA: ℞-only[1][2][3][4]US: ℞-only[5] |
| Identifiers | |
| showIUPAC name | |
| CAS Number | 410528-02-8 |
| PubChem CID | 10295295 |
| DrugBank | DB05467 |
| ChemSpider | 8470763 |
| UNII | 28K6I5M16G |
| KEGG | D09365 |
| ChEBI | CHEBI:188559 |
| Chemical and physical data | |
| Formula | C27H30N2O2 |
| Molar mass | 414.549 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| showSMILES | |
| showInChI | |
References[
- ^ “Notice: Multiple Additions to the Prescription Drug List (PDL) [2022-01-24]”. Health Canada. 24 January 2022. Archived from the original on 29 May 2022. Retrieved 28 May 2022.
- ^ “Summary Basis of Decision – Sohonos”. Health Canada. 23 October 2014. Archived from the original on 6 August 2022. Retrieved 6 August 2022.
- ^ “Sohonos product information”. Health Canada. 20 June 2022. Archived from the original on 29 January 2023. Retrieved 28 January 2023.
- ^ Jump up to:a b c d “Sohonos Product Information”. Health Canada. 22 October 2009. Archived from the original on 18 August 2023. Retrieved 17 August 2023.
- ^ Jump up to:a b c d “Archived copy” (PDF). Archived (PDF) from the original on 18 August 2023. Retrieved 18 August 2023.
- ^ “Health Canada Approves Ipsen’s Sohonos (palovarotene capsules) as the First Approved Treatment for Fibrodysplasia Ossificans Progressiva” (Press release). Ipsen. 24 January 2022. Retrieved 28 May 2022 – via Business Wire.
- ^ Hind M, Stinchcombe S (November 2009). “Palovarotene, a novel retinoic acid receptor gamma agonist for the treatment of emphysema”. Current Opinion in Investigational Drugs. 10 (11): 1243–50. PMID 19876792.
- ^ Stolk J, Stockley RA, Stoel BC, Cooper BG, Piitulainen E, Seersholm N, et al. (August 2012). “Randomised controlled trial for emphysema with a selective agonist of the γ-type retinoic acid receptor”. The European Respiratory Journal. 40 (2): 306–12. doi:10.1183/09031936.00161911. PMID 22282548.
- ^ Shimono K, Tung WE, Macolino C, Chi AH, Didizian JH, Mundy C, et al. (April 2011). “Potent inhibition of heterotopic ossification by nuclear retinoic acid receptor-γ agonists”. Nature Medicine. 17 (4): 454–60. doi:10.1038/nm.2334. PMC 3073031. PMID 21460849.
- ^ Kaplan FS, Shore EM (April 2011). “Derailing heterotopic ossification and RARing to go”. Nature Medicine. 17 (4): 420–1. doi:10.1038/nm0411-420. PMC 4913781. PMID 21475232.
- ^ Chakkalakal SA, Uchibe K, Convente MR, Zhang D, Economides AN, Kaplan FS, et al. (September 2016). “Palovarotene Inhibits Heterotopic Ossification and Maintains Limb Mobility and Growth in Mice With the Human ACVR1(R206H) Fibrodysplasia Ossificans Progressiva (FOP) Mutation”. Journal of Bone and Mineral Research. 31 (9): 1666–75. doi:10.1002/jbmr.2820. PMC 4992469. PMID 26896819.
- ^ “Ipsen announces FDA Priority Review for NDA in patients with FOP”. Ipsen (Press release). 24 August 2022. Retrieved 28 January 2023.
- ^ “Palovarotene Orphan Drug Designations and Approvals”. U.S. Food and Drug Administration (FDA). 1 January 2013. Archived from the original on 29 January 2023. Retrieved 28 January 2023.
- ^ “EU/3/14/1368”. European Medicines Agency (EMA). 17 September 2018. Archived from the original on 27 January 2023. Retrieved 28 January 2023.
- ^ “Public summary of opinion on orphan designation. Palovarotene for the treatment of fibrodysplasia ossificans progressiva” (PDF). European Medicines Agency (EMA). Archived (PDF) from the original on 22 April 2016. Retrieved 11 April 2016.
- ^ “Clementia Pharmaceuticals Receives Fast Track Designation for Palovarotene for Treatment of Fibrodysplasia Ossificans Progressiva (FOP)” (Press release). Clementia Pharmaceuticals. 1 December 2014. Retrieved 11 April 2016 – via PR Newswire.
- ^ “Clementia Pharmaceuticals Receives EMA Orphan Medicinal Product Designation for Palovarotene for the Treatment of Fibrodysplasia Ossificans Progressiva” (Press release). Clementia Pharmaceuticals. 21 November 2014. Retrieved 11 April 2016 – via PR Newswire.
- ^ Pignolo RJ, Baujat G, Hsiao EC, Keen R, Wilson A, Packman J, et al. (October 2022). “Palovarotene for Fibrodysplasia Ossificans Progressiva (FOP): Results of a Randomized, Placebo-Controlled, Double-Blind Phase 2 Trial”. Journal of Bone and Mineral Research. 37 (10): 1891–1902. doi:10.1002/jbmr.4655. PMC 9804935. PMID 35854638. S2CID 250697248.
- ^ “FDA Tells Ipsen It Won’t Approve Palovarotene for FOP”. Global Genes. 27 December 2022. Archived from the original on 29 January 2023. Retrieved 28 January 2023.
- ^ “Sohonos: Pending EC decision”. European Medicines Agency (EMA). 26 January 2023. Archived from the original on 27 January 2023. Retrieved 28 January 2023.
- ^ “Clementia Announces Plan to Submit a New Drug Application for Palovarotene for the Treatment of FOP Based on Positive Phase 2 Results”. 23 October 2018. Archived from the original on 15 December 2019. Retrieved 15 December 2019.
- ^ “Ipsen Initiates Partial Clinical Hold for Palovarotene IND120181 and IND135403 Studies”. Archived from the original on 15 December 2019. Retrieved 15 December 2019.
- ^ “Ipsen Completes Acquisition of Clementia Pharmaceuticals”. Archived from the original on 15 December 2019. Retrieved 15 December 2019.
External links
Clinical trial number NCT03312634 for “An Efficacy and Safety Study of Palovarotene for the Treatment of Fibrodysplasia Ossificans Progressiva. (MOVE)” at ClinicalTrials.gov
/////////FDA 2023, APPROVALS 2023, Palovarotene, Sohonos, RG-667, RO-3300074
syn
syn
European Journal of Medicinal Chemistry 265 (2024) 116124
Palovarotene (Sohonos)
On February 17, 2022, the FDA granted approval to Palovarotene for the treatment of heterotopic ossification (HO) linked to fibrodysplasia ossificans progressiva (FOP) [64]. FOP, or myositis ossificans pro
gressiva (MOP), is an uncommon hereditary condition marked by atypical bone growth in regions beyond the usual skeletal structure. It is commonly accompanied by recurring episodes of discomfort and abrupt
swelling of soft tissues. This disorder causes restricted mobility and fusion of joints, leading to deformities, limited movement, and premature mortality [65]. Palovarotene is an orally available retinoic acid receptor γ (RARγ) agonist [66]. Palovarotene specifically attaches to RARγ and hinders the phosphorylation process of mothers against decapentaplegic homolog (SMAD)1/5/8. This action results in the suppression of the bone morphogenetic protein (BMP)/ALK2 downstream signaling pathway, leading to a decrease in ALK2/SMAD-dependent chondrogenesis and osteoblast differentiation. Consequently, the over all effect is a reduction in endochondral ossification [67].
The preparation of Palovarotene is shown in Scheme 18 [68].Starting with 2,5-dimethylhexane-2,5-diol (PALO-001), a nucleophilic substitution reaction with HCl, followed by AlClpromoted Friedel-Crafts alkylation with 1-bromo-2-methylbenzene (PALO-003), gave PALO-004. PALO-005 was obtained by substitution with CuCN.The cyano group of PALO-005 was reduced to aldehyde by diisobutylalumium hydride (DIBAL-H) to obtain PALO-006. PALO-006 was subjected to Wittig-Horner reaction with methyl 4-((dimethoxyphosphoryl)methyl)benzoate PALO-007 to obtain olefin PALO-008.
PALO-008 was brominated with N-bromosuccinimide (NBS) to obtain PALO-009. PALO-009 was nucleophilic substituted with 1H-pyrazole (PALO-010) to obtain PALO-011, which was hydrolyzed under alkaline conditions to obtain the final product Palovarotene.
[64] S.M. Hoy, Palovarotene: first approval, Drugs 82 (2022) 711–716.
[65] R.J. Pignolo, E.M. Shore, F.S. Kaplan, Fibrodysplasia ossificans progressiva:
diagnosis, management, and therapeutic horizons, Pediatr. Endocrinol. Rev. 2
(2013) 437–448.
[66] G.J. Pavey, A.T. Qureshi, A.M. Tomasino, C.L. Honnold, D.K. Bishop, S. Agarwal,
S. Loder, B. Levi, M. Pacifici, M. Iwamoto, B.K. Potter, T.A. Davis, J.A. Forsberg,
Targeted stimulation of retinoic acid receptor-γ mitigates the formation of
heterotopic ossification in an established blast-related traumatic injury model,
Bone 90 (2016) 159–167.
[67] H. Kitoh, Clinical aspects and current therapeutic approaches for FOP,
Biomedicines 8 (2020) 325.
[68] J.-M. Lapierre, D.M. Rotstein, E.B. Sjogren, Preparation of New Retinoids for the
Treatment of Emphysema, Cancer and Dermatological Disorders, 2002.
WO2002028810.

NEW DRUG APPROVALS
ONE TIME
$10.00
Zavegepant

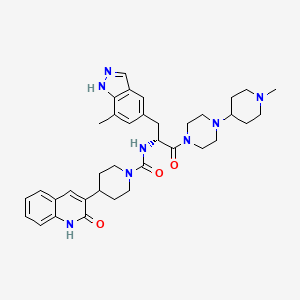
Zavegepant
ザベジェパント;
- 1337918-83-8
- as HCl: 1414976-20-7
C36H46N8O3 BASE
638.8 g/mol BASE
- Vazegepant
- BMS-742413
- BHV-3500
FDA APPR 3/9/2023Zavzpret
N-[(2R)-3-(7-methyl-1H-indazol-5-yl)-1-[4-(1-methylpiperidin-4-yl)piperazin-1-yl]-1-oxopropan-2-yl]-4-(2-oxo-1H-quinolin-3-yl)piperidine-1-carboxamide
ZAVZPRET is indicated for the acute treatment of migraine with or without aura in adults.
The recommended dose of ZAVZPRET is 10 mg given as a single spray in one nostril, as needed. The maximum dose that may be given in a 24-hour period is 10 mg (one spray). The safety of treating more than 8 migraines in a 30-day period has not been established, Nasal spray: 10 mg of zavegepant per device. Each unit-dose nasal spray device delivers a single spray containing 10 mg of zavegepant.
ZAVZPRET (zavegepant) nasal spray contains zavegepant hydrochloride, a calcitonin generelated peptide receptor antagonist. Zavegepant hydrochloride is described chemically as (R)-N- (3-(7-methyl-1H-indazol-5-yl)-1-(4-(1-methylpiperidin-4-yl) piperazin-1-yl)-1-oxopropan-2-yl)- 4-(2-oxo-1,2-dihydroquinolin-3-yl) piperidine-1-carboxamide hydrochloride and its structural formula is:

Its molecular formula is C36H46N8O3․HCl, representing a molecular weight of 675. 28 g/mol. Zavegepant free base has a molecular weight of 638.82 g/mol. Zavegepant hydrochloride is a white to off-white powder, freely soluble in water, and has pKa values of 4.8 and 8.8. Each unit-dose ZAVZPRET device for nasal administration delivers 10 mg of zavegepant (equivalent to 10.6 mg of zavegepant hydrochloride) in a buffered aqueous solution containing dextrose, hydrochloric acid, sodium hydroxide, and succinic acid in water for injection. The solution has a pH of 5.3 to 6.7.
Active ingredients in ZAVZPRET: zavegepant Inactive ingredients in ZAVZPRET: dextrose, hydrochloric acid, sodium hydroxide, and succinic acid in water for injection.
Zavegepant, sold under the brand name Zavzpret, is a medication used for the treatment of migraine.[1] Zavegepant is a calcitonin gene-related peptide receptor antagonist.[1] It is sprayed into the nose.[1] It is sold by Pfizer.[1]
The most common adverse reactions include taste disorders, nausea, nasal discomfort, and vomiting.[1]
Zavegepant was approved for medical use in the United States in March 2023.[1][2][3]
Medical usesZavegepant is a Calcitonin Gene-related Peptide Receptor Antagonist. The mechanism of action of zavegepant is as a Calcitonin Gene-related Peptide Receptor Antagonist.
Zavegepant is indicated for the acute treatment of migraine with or without aura in adults.[1]
Zavegepant is an antagonist of the calcitonin gene-related peptide (CGRP) receptor currently in phase 3 trials in an intranasal formulation for the treatment of migraine. If FDA approved, it will join other previously-approved “-gepant” drugs [rimegepant] and [ubrogepant] as an additional treatment alternative for patients with migraine, particularly those for whom traditional triptan therapy has proven ineffective. On April 15th, 2020, a phase 2 clinical trial (NCT04346615: Safety and Efficacy Trial of Vazegepant Intranasal for Hospitalized Patients With COVID-19 Requiring Supplemental Oxygen) began to investigate the use of intranasally administered zavegepant to combat the acute respiratory distress syndrome (ARDS) sometimes seen in patients with COVID-19. Acute lung injury activates the release of CGRP, which plays a role in the development of ARDS – CGRP antagonists, then, may help to blunt the significant inflammation associated with COVID-19. The clinical trial is expected to complete in September 2020.
Zavegepant is a highly soluble small molecule calcitonin gene related peptide (CGRP) receptor antagonist, with potential analgesic and immunomodulating activities. Upon administration, zavegepant targets, binds to and inhibits the activity of CGRP receptors located on mast cells in the brain. This may inhibit neurogenic inflammation caused by trigeminal nerve release of CGRP. In addition, by blocking the CGRP receptors located in smooth muscle cells within vessel walls, zavegepant inhibits the pathologic dilation of intracranial arteries. Zavegepant, by blocking the CGRP receptors, also suppresses the transmission of pain by inhibiting the central relay of pain signals from the trigeminal nerve to the caudal trigeminal nucleus. Altogether, this may relieve migraine. As CGRP receptors induce the release of pro-inflammatory mediators, such as interleukin-6 (IL-6), from inflammatory cells, zavegepant may prevent an IL-6-mediated inflammatory response. Zavegepant may also inhibit the CGRP-mediated induction of eosinophil migration and the stimulation of beta-integrin-mediated T cell adhesion to fibronectin at the site of inflammation, and may abrogate the CGRP-mediated polarization of the T cell response towards the pro-inflammatory state characterized by Th17 and IL-17. This may improve lung inflammation and oxygenation, prevent edema, and further lung injury. CGRP, a 37 amino-acid peptide expressed in and released from a subset of polymodal primary sensory neurons of the trigeminal ganglion and nerve fibers projecting to the airways and by pulmonary neuroendocrine cells, plays an important role in pain transmission, inflammation, and neurogenic vasodilatation. It is released upon acute lung injury and upregulation of transient receptor potential (TRP) channels.
SYN’
Synthesis of a CGRP Receptor Inhibitor
Publication Date: 2013
Publication Name: Synfacts
Azepino-indazoles as calcitonin gene-related peptide (CGRP) receptor antagonists
- PMID: 33096162Publication Date: 2021-01-01Journal: Bioorganic & medicinal chemistry lettersDiscovery of (R)-N-(3-(7-methyl-1H-indazol-5-yl)-1-(4-(1-methylpiperidin-4-yl)-1-oxopropan-2-yl)-4-(2-oxo-1,2-dihydroquinolin-3-yl)piperidine-1-carboxamide (BMS-742413): a potent human CGRP antagonist with superior safety profile for the treatment of migraine through intranasal delivery
PMID: 23632269Publication Date: 2013-06-01Journal: Bioorganic & medicinal chemistry letters

////////

AS ON DEC2021 3,491,869 VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@amcrasto
Patent
https://patents.google.com/patent/US20120245356A1/en
Patent
WO 2022165291
https://patents.google.com/patent/WO2022165291A1/en
Migraine is a chronic and debilitating disorder characterized by recurrent attacks lasting four to 72 hours with multiple symptoms, including typically one-sided, pulsating headaches of moderate to severe pain intensity that are associated with nausea or vomiting, and/or sensitivity to sound (phonophobia) and sensitivity to light (photophobia). Migraines are often preceded by transient neurological warning symptoms, known as auras, which typically involve visual disturbances such as flashing lights, but may also involve numbness or tingling in parts of the body. Migraine is both widespread and disabling. The Migraine Research Foundation ranks migraine as the world’s third most prevalent illness, and the Global Burden of Disease Study 2015 rates migraine as the seventh highest specific cause of disability worldwide. According to the Migraine Research Foundation, in the United States, approximately 36 million individuals suffer from migraine attacks. While most sufferers experience migraine attacks once or twice per month, more than 4 million people have chronic migraine, defined as experiencing at least 15 headache days per month, of which at least eight are migraine, for more than three months. Others have episodic migraine, which is characterized by experiencing less than 15 migraine days per month. People with episodic migraine may progress to chronic migraine over time. Migraine attacks can last four hours or up to three days. More than 90% of individuals suffering from migraine attacks are unable to work or function normally during a migraine attack, with many experiencing comorbid conditions such as depression, anxiety and insomnia. Also, those suffering from migraine often have accompanying nausea and have an aversion to consuming food or liquids during an attack.
CGRP (calcitonin gene-related peptide) is a 37 amino acid neuropeptide, which belongs to a family of peptides that includes calcitonin, adrenomedullin and amylin. In humans, two forms of CGRP (a-CGRP and 0-CGRP) exist and have similar activities. They vary by three amino acids and exhibit differential distribution. At least two CGRP receptor subtypes may also account for differential activities. The CGRP receptor is located within pain-signaling pathways, intracranial arteries and mast cells and its activation is thought to play a causal role in migraine pathophysiology. For example, research and clinical studies have shown: serum levels of CGRP are elevated during migraine attacks, infusion of intravenous CGRP produces persistent pain in migraine sufferers and non-migraine sufferers, and treatment with anti-migraine drugs normalizes CGRP activity.
Currently, clinicians use a number of pharmacologic agents for the acute treatment of migraine. A study published by the American Headache Society in 2015 concluded that the medications deemed effective for the acute treatment of migraine fell into the following classes: triptans, ergotamine derivatives, non-steroidal anti-inflammatory drugs (“NSAIDs”), opioids and combination medications. The current standard of care for the acute treatment of migraine is prescription of triptans, which are serotonin 5-HT IB/ID receptor agonists. Triptans have been developed and approved for the acute treatment of migraine over the past two decades. The initial introduction of triptans represented a shift toward drugs more selectively targeting the suspected pathophysiology of migraine. While triptans account for almost 80% of anti-migraine therapies prescribed at office visits by healthcare providers, issues such as an incomplete effect or headache recurrence remain important clinical limitations. In fact, only about 30% of patients from clinical trials are pain free at two hours after taking triptans. In addition, triptans are contraindicated in patients with cardiovascular disease, cerebrovascular disease, or significant risk factors for either because of potential systemic and cerebrovascular vasoconstriction from the 5-HT IB -mediated effects. Also, according to a January 2017 study published in the journal Headache, an estimated 2.6 million migraine sufferers in the United States have a cardiovascular event, condition or procedure that limits the potential of triptans as a treatment option.
Accordingly, there remains a significant unmet medical need for a novel migraine-specific medication that provides enhanced patient benefits compared to existing therapies.
Possible CGRP involvement in migraine has been the basis for the development and clinical testing of a number of compounds, including for example, advanced clinical candidates rimegepant (BHV-3000) and zavegepant (BHV-3500), which are developed by Biohaven Pharmaceutical Holding Company Ltd., New Haven, CT.
Zavegepant (also known as vazegepant) is a third generation, high affinity, selective and structurally unique small molecule CGRP receptor antagonist having the following formula I:

I
Zavegepant is described, for example, in WO 03/104236 published December 18, 2003 and US 8,481,546 issued July 9, 2013, which are incorporated herein in their entireties by reference.
While zavegepant is a highly soluble molecule, its bioavailability characteristics may render it challenging to prepare the drug in an oral dosage form. Enhancing the bioavailability of zavegepant and other CGRP inhibitors by different administration routes would therefore be desirable.
Calcitonin gene-related peptide (CGRP) is widely distributed in nociceptive pathways in human peripheral and central nervous system and its receptors are also expressed in pain pathways. While CGRP is involved in migraine pathophysiology, its role in non-headache pain has not been quite clear. There remains a need for new medicines to treat various pain disorders in patients in need thereof.
Scheme 1


Scheme 3

Scheme 4

tert-butyl 4-(2-methoxy-2-oxoethylidene)piperidine-l -carboxylate. Sodium hydride in mineral oil (60%, 7.92 g, 198.02 mmoles) was washed with hexanes then suspended in dimethylformamide (220 mL). The mixture was cooled to 0°C. Trimethyl phosphonoacetate (29.0 mL, 189.82 mmoles) was added dropwise to the stirred reaction mixture. After 20 min at 0°C, a solution of A-/c/7-butoxycarbonyl-4-pi peri done (30.41 g, 152.62 mmoles) in dimethylformamide (80 mL) was added to the mixture dropwise. The reaction was stirred at room temperature for 3 h and then diluted with diethyl ether (650 mL). The mixture was washed once with water and the aqueous layer was extracted once with diethyl ether. The combined organic layers were washed 4 times with water and the aqueous phase was discarded. The organic phase was washed with brine and dried over magnesium sulfate, filtered, and concentrated to dryness. The title compound was obtained as a white solid in 92% yield. 1 H- NMR (300 MHz, CDCh): 5 = 5.68 (s, 1 H), 3.66 (s, 3 H), 3.40-3.51 (m, 4 H), 2.90 (t, J= 5.49, 2 H), 2.25 (t, J= 5.49, 2 H), 1.44 (s, 9 H).

ed-butyl 4-(2-methoxy-2-oxoethyl)piperidine-l -carboxylate. A solution of tert-butyl 4- (2-methoxy-2-oxoethylidene)piperidine-l -carboxylate (35.71 g, 140 mmoles) in a mixture of 1 : 1 ethyl acetate/methanol (220 mL) was carefully treated with 50% wet 10% palladium on carbon (3.3 g). The reaction vessel was charged with 55 psi of hydrogen gas and the mixture was shaken on a Parr apparatus at room temperature for 16 h. The reaction mixture was then filtered to remove the catalyst and the filtrate concentrated in vacuo. The title compound was obtained as a clear colorless oil in 97% yield. ‘H-NMR (300 MHz, CDCh): 5 = 4.04 (d, J= 10.25, 2 H), 3.64 (s, 3 H), 2.68 (t, J= 12.44, 2 H), 2.21 (d, J= 6.95, 2 H), 1.98-1.77 (m, 1 H), 1.64 (d, J= 13.54, 2 H), 1.41 (s, 9 H), 1.25-0.99 (m, 2 H).

4-[2-Hydroxy-l-methoxycarbonyl-2-(2-nitro-phenyl)-ethyl]-piperidine-l-carboxylic acid tert-butyl ester. A A-diisopropylamine (4.40 mL, 31.3 mmoles) was dissolved in tetrahydrofuran (50 mL). The mixture was cooled to -78°C. Butyllithium (2.5 M in hexanes, 12.4 mL, 31 mmoles) was added dropwise to the stirred solution. After stirring at -78°C for 30 min, a solution of tert-butyl 4-(2-methoxy-2-oxoethyl)piperidine-l -carboxylate (6.65 g, 25.8 mmoles) in tetrahydrofuran (15 mL) was added dropwise to the mixture. Stirring was continued at -78°C for 1 h. A solution of 2-nitrobenzaldehyde (3.90 g, 25.8 mmoles) in tetrahydrofuran (20 mL) was then added to the mixture dropwise, and then stirring was continued at -78°C for a further 2.5 h. The reaction was quenched with cold aqueous ammonium chloride and then diluted with water. The mixture was extracted twice with ethyl acetate and the aqueous phase was discarded. The material was dried (magnesium sulfate) filtered, and concentrated to dryness. Silica gel chromatography afforded the desired product in 94% yield as light yellow foam. MS m/e (M- C4H8+H)+= 353.1.

4-(4-Hydroxy-2-oxo-l , 2, 3, 4-tetrahydro-quinolin-3-yl)-piperidine-l -carboxylic acid tertbutyl ester. In a 3 neck flask fitted with a nitrogen inlet, thermometer, and a mechanical stirrer, 4-[2-hydroxy-l -methoxy carbonyl-2-(2-nitro-phenyl)-ethyl]-piperidine-l -carboxylic acid tertbutyl ester (9.93 g, 24.3 mmoles) was dissolved in acetic acid (1.75 moles, 100 mL). Iron powder (8.90 g, 159 mmoles) was added to the vessel with stirring. The stirred mixture was slowly heated to 80°C for 30 min and then cooled to room temperature. It was then diluted with ethyl acetate and filtered through a pad of celite. Solids were washed with 20% methanol/ethyl acetate, and then with methanol. The filtrate was concentrated and the residue partitioned between ethyl acetate and aqueous sodium bicarbonate. The layers were separated. The resulting aqueous phase was extracted twice with ethyl acetate. The organic layers were combined. The mixture was washed twice with water and the aqueous phase was discarded. The material was dried (magnesium sulfate) filtered, and concentrated to dryness. Silica gel chromatography afforded the title compound as light yellow foam in 77% yield. MS m/e (M-H)’ = 345.1.

3-(Piperidin-4-yl)quinolin-2(lH) hydrochloride . A stirred solution of 4-(4-hydroxy-2- oxo-l,2,3,4-tetrahydro-quinolin-3-yl)-piperidine-l-carboxylic acid tert-butyl ester (5.60 g, 16.2 mmoles) in ethyl acetate (70 mL) was treated with HC1 in dioxane (4N, 40 mmoles, 10 mL). The mixture was stirred at room temperature for 45 min. More HC1 in dioxane (4N, 120 mmoles, 30 mL) was then added and stirring was continued at room temperature for 16 h. The resulting solid was collected by filtration and washed with ethyl acetate. It was then suspended in 5% water-isopropanol (100 mL) and the mixture was warmed to reflux and stirred for 20 min. The mixture was cooled to room temperature and stirred at room temperature for 16 h. The solid was collected by filtration, washed with isopropanol, and dried under high vacuum. The title compound was obtained as white solid in 75% yield. ‘H-NMR (DMSO-de) 5 11.85 (s, 1 H), 9.02 (bs, 1 H), 8.88 (bs, 1 H), 7.70 (t, J= 3.81 Hz, 2 H), 7.53 – 7.30 (d, J= 8.24 Hz, 1 H), 7.17 (t, J= 7.48 Hz, 2 H), 3.36 (d, J= 12.51 Hz, 2 H), 3.10 – 2.94 (m, 3 H), 2.01 (d, J= 13.43 Hz, 2 H), 1.87 – 1.73 (m, 2 H); MS m/e (M+H)+ = 229.0.

4-Iodo-2,6-dimethylbenzenamine hydrochloride . To a suspension of sodium bicarbonate (126 g, 1.5 moles) and 2,6-dimethylaniline (61.5 mL, 500 mmoles) in methanol (700 mL) was added iodine monochloride (1.0 M in dichloromethane, 550 mL, 550 mmoles) at room temperature over 1 h. After addition was complete, stirring was continued for 3 h. The reaction was filtered to remove excess sodium bicarbonate and the solvent removed in vacuo. The residue was re-dissolved in diethyl ether (1.5 L) and treated with hydrochloric acid (2M in ether, 375 mL, 750 mmoles). The resulting suspension was stored in the freezer (-15°C) overnight. The solid was filtered and washed with diethyl ether until it became colorless, to give 126.5 g (89%) as a grey-green powder. ‘H-NMR (DMSO-de) 5 2.33 (s, 6 H), 7.48 (s, 2 H), 9.05 (bs, 3 H); 13C-NMR (DMSO-de) 5 17.4, 91.5, 133.1, 131.2, 136.9.

Methyl 2 -(benzyloxy carbonyl) acrylate . To a flame dried three-neck round bottom flask, fitted with a mechanical stirrer, was added (S)-methyl 2-(benzyloxycarbonyl)-3- hydroxypropanoate (129 g, 509 mmoles), anhydrous dichloromethane (2 L), and methanesulfonyl chloride (49.3 mL, 636 mmoles). The mixture was cooled to -15°C, and treated with tri ethylamine (213 mL, 1527 mmoles), dropwise, to ensure the temperature of the reaction mixture did not exceed 0°C. The addition of the first equivalent of triethylamine was exothermic. After addition of tri ethylamine, the mixture was stirred at 0°C for 30 min. The cooling bath was removed and the mixture stirred at room temperature for 1.5 h. The reaction was quenched by addition of methanol (21 mL). The mixture was washed with 0.5% aqueous potassium bisulfate until the washings were pH 5, then saturated sodium bicarbonate, and brine, dried over sodium sulfate, and concentrated. Flash chromatography (silica gel, 1 :9 ethyl acetate/hexanes) gave I l l g (92%) as a viscous colorless oil, which crystallized upon standing. ’H-NMR (DMSO-de) 5 3.71 (s, 3 H), 5.10 (s, 2 H), 5.60 (s, 1 H), 5.76 (s, 1 H), 7.39-7.35 (m, 5 H), 8.96 (s, 1 H); 13C-NMR (DMSO-de) 5 52.3, 65.9, 127.8, 128.1, 128.3, 128.8, 133.3, 136.3, 153.5, 163.7.

(Z)-Methyl 3-(4-amino-3,5-dimethylphenyl)-2-(benzyloxycarbonyl) acrylate. A 2 L round bottom flask was charged 4-iodo-2,6-dimethylbenzenamine hydrochloride salt (55 g, 194 mmoles), methyl 2-(benzyloxycarbonyl)acrylate (59.2 g, 252 mmoles), tetrabutylammonium chloride (59.2 g, 213 mmoles), palladium (II) acetate (4.34 g, 19.4 mmoles), and tetrahydrofuran (1.2 L, degassed by a flow of nitrogen for 30 min). The mixture was stirred so that a suspension was formed and then degassed by a flow of nitrogen for 30 min. Triethylamine (110 mL, 789 mmoles) was added and the resulting mixture was heated at reflux for 3 h. After cooling to room temperature, the reaction mixture was filtered through a pad of celite, washed with tetrahydrofuran (2 x 100 mL), and concentrated. The residue was dissolved in di chloromethane, washed with water (3X) and brine (2X), dried over sodium sulfate, and concentrated. Flash chromatography (silica gel, using 1 :9 ethyl acetate/dichloromethane) gave a tan solid. The solid was recrystallized from warm methanol (210 mL) and water (100 mL). The mixture was held at room temperature overnight, then at 0°C for 2 h, and finally at -15°C for 2 h. The resulting solid was filtered, washed with ice cold 1 : 1 methanol/water, and dried under high vacuum overnight to give 44.7 g (65%) as a light tan solid which was a mixture of ZZE isomers (73 :27). ’H-NMR (DMSO-de) 5, 2.05 (s, 6 H), 3.61 (s, 0.8 H), 3.68 (s, 2.2 H), 5.00 (s, 0.54 H), 5.13 (s, 1.46 H), 5.24 (s, 2 H), 7.40-7.21 (m, 8 H), 8.51 (s, 0.27 H), 8.79 (s, 0.73 H); 13C-NMR (DMSO-de) 5 17.8, 51.7, 65.3, 119.4, 120.0, 120.3, 127.3, 127.7, 128.3, 130.9, 135.8, 137.2, 146.9, 154.7, 166.0.

(R)-Methyl 3-(4-amino-3,5-dimethylphenyl)-2-(benzyloxycarbonyl)propanoate. A flame- dried 2 L Parr hydrogenation bottle was charged with (Z)-methyl 3-(4-amino-3,5- dimethylphenyl)-2-(benzyloxycarbonyl)acrylate (84.5 g, 239 mmoles), di chloromethane (300 mL), and methanol (300 mL). The bottle was swirled so that a light brown suspension was formed. The mixture was degassed using a flow of nitrogen for 30 min. To this was quickly added (-)-l,2-bis((2A,5A)-2,5-diethylphospholano)-bezene(cyclooctadiene) rhodium (I) tetrafluoroborate ([(2A,5A)-Et-DuPhosRh]BF4) (2.11 g, 3.20 mmoles). The bottle was immediately attached to a Parr Hydrogenator. After 5 cycles of hydrogen (60 psi) and vacuum, the bottle was pressurized to 65 psi and the suspension was agitated at room temperature for 16 h. The reaction had become homogeneous. The reaction mixture was concentrated, and the resulting residue purified by flash chromatography (silica gel, 1 :9 ethyl acetate/dichloromethane) to give 82.9 g (98%). ‘H-NMR (DMSO-de) 5 2.04 (s, 6 H), 2.65 (dd, J= 13.4, 9.8 Hz, 1H), 2.82 (dd, J= 13.7, 5.2 Hz, 1 H), 3.62 (s, 3 H), 4.15-4.10 (m, 1H), 4.41 (s, 2 H), 5.00 (s, 2 H), 6.68 (s, 2 H), 7.37-7.28 (m, 5 H), 7.70 (d, J= 7.9 Hz, 1 H); 13C-NMR (DMSO-de) 5 17.7, 35.9, 51.7, 56.1, 65.3, 120.4, 124.0, 127.5, 127.7, 128.2, 128.3, 136.9, 142.6, 155.9, 172.5.

(R)-Methyl 2-(benzyloxycarbonyl)-3-(7-methyl-lH-indazol-5-yl)propanoate. (R)-Methyl 3-(4-amino-3,5-dimethylphenyl)-2-(benzyloxycarbonyl)propanoate (50.0 g, 140 mmoles) was weighed into a flame-dried 5 L three neck round bottom flask, followed by the addition of toluene (2.4 L) and glacial acetic acid (120 mL, 2.1 moles). The mixture was mechanically stirred to form a clear solution, and then potassium acetate (103 g, 1.05 moles) was added. To the resulting white suspension, z.w-amyl nitrite (20.7 mL, 154 mmoles) was added dropwise at room temperature, and the resulting mixture was stirred at room temperature for 16 h. Saturated sodium bicarbonate (I L) was added, followed by the careful addition of solid sodium bicarbonate to neutralize the acetic acid. The mixture was extracted with a mixture of di chloromethane (2 L) and brine (1.5 L). After separation, the aqueous layer was extracted with di chloromethane (500 mL). The combined organic layers were dried over anhydrous sodium sulfate and filtered. Solvents were removed to afford a tan solid, which was washed with hexanes (2 L) and toluene (150 mL). The solid was recrystallized from hot acetone (260 mL) and hexanes (700 mL). The slightly cloudy mixture was allowed to cool to room temperature slowly, then to 0°C for 1.5 h, and finally to -15°C for 1.5 h. The resulting solid was filtered and washed with ice-cold acetone/hexanes (1 : 1, 200 mL) to afford 39.1 g (76% yield). Analytical HPLC showed >98% UV purity. The enantiomeric excess (ee) was determined to be 99.8% (conditions: Chiralpak AD column, 4.6 x 250 mm, 10 pm; A = ethanol, B = 0.05% diethylamine/heptane; 85%B @1.0 mL/min. for 55 min. The retention times for R was 44.6 min and for S was 28.8 min). ‘H-NMR (DMSO-de) 5 2.48 (s, 3 H), 2.93 (dd, J= 13.4, 10.7 Hz, 1H), 3.10 (dd, J= 13.7, 4.9 Hz, 1H), 3.63 (s, 3H), 4.32-4.27 (m, 1 H), 4.97 (s, 2 H), 7.03 (s, 1 H), 7.24-7.22 (m, 2 H), 7.29 -7.27 (m, 3 H), 7.41 (s, 1 H), 7.83 (d, J= 8.2 Hz, 1H), 7.99 (s, 1H), 13.1 (s, 1 H); 13C-NMR (DMSO-de) 5 16.7, 36.5, 51.8, 56.0, 65.3, 117.6, 119.6, 122.7, 127.2, 127.4, 127.6, 128.2, 129.3, 133.4, 136.8, 139.2, 155.9, 172.4. Mass spec.: 368.16 (MH)+.

(R)-Methyl 2-amino-3-(7-methyl-lH-indazol-5-yl)propanoate. A Parr hydrogenation bottle was charged with (R)-methyl 2-(benzyloxycarbonyl)-3-(7-methyl-lH-indazol-5- yl)propanoate (11.0 g, 29.9 mmoles) and methanol (75 mL). The suspension was purged with nitrogen and treated with palladium (10% on charcoal, 700 mg). The bottle was shaken under hydrogen (15 psi) overnight. The mixture was filtered through a pad of celite to remove the catalyst. Concentration of the eluent gave 7.7 g (quant.) as an oil which was used without further purification. XH-NMR (CD3OD) 5 2.54 (s, 3 H), 2.98 (dd, J= 13.5, 7.0 Hz, 1 H), 3.09 (dd, J= 13.5, 5.9 Hz, 1 H), 3.68 (s, 3 H), 3.75 (dd, J= 7.0, 6.2 Hz, 1 H), 7.01 (s, 1 H), 7.39 (s, 1 H), 7.98 (s, 1 H). Mass spec.: 232.34 (M-H)’.

(R)-methyl 3-(7-methyl-lH-indazol-5-yl)-2-(4-(2-oxo-l,2-dihydroquinolin-3- yl)piperidine-l-carboxamido)propanoate. To a solution of (R)-methyl 2-amino-3-(7-methyl-lH- indazol-5-yl)propanoate hydrochloride (7.26 g, 27.0 mmoles) in dimethylformamide (50 mL) at room temperature was added N, A’-disuccinimidyl carbonate (7.60 g, 29.7 mmoles) followed by triethylamine (11.29 mL, 81 mmoles). The resulting mixture was stirred for 30 min and treated with 3-(piperidin-4-yl)quinolin-2(lH)-one (6.77 g, 29.9 mmoles) in portions. The reaction was allowed to stir for 24 h. The mixture was concentrated, dissolved in ethyl acetate, and washed sequentially with water, brine, and 0.5 N HC1 (2X). The organic phase was dried over magnesium sulfate, filtered, and concentrated. The resulting residue was purified by flash chromatography (silica gel, 20: 1 ethyl acetate/methanol) to give 11.9 g (78%). 1 H-NMR (CD3OD) 5 13.0 (s, 1 H), 11.8 (s, 1 H), 7.98 (s, 1 H), 7.63 (d, J= 7.6 Hz, 1 H), 7.57 (s, 1 H), 7.45 – 7.41 (m, 2 H), 7.27 (d, J= 8.2Hz, 1 H), 7.16 (t, J= 7.9 Hz, 1 H), 7.03 (s, 1 H), 6.85 (d, J= 7.9 Hz, 1 H), 4.31 – 4.26 (m, 1 H), 4.10 – 4.08 (m, 2 H), 3.60 (s, 3 H), 3.07 – 3.01 (m, 2 H), 2.93 – 2.88 (m, 1 H), 2.77 – 2.67 (m, 2 H), 2.48 (s, 3 H), 1.78 – 1.72 (m, 2 H), 1.34 – 1.26 (m, 2 H). Mass spec.: 488.52 (MH)+.

(R)-3-(7-methyl-lH-indazol-5-yl)-2-(4-(2-oxo-l,2-dihydroquinolin-3-yl)piperidine-l- carboxamido)propanoic acid. A solution of (R)-methyl 3-(7-methyl-lH-indazol-5-yl)-2-(4-(2- oxo-1, 2-dihydroquinolin-3-yl)piperidine-l-carboxamido)propanoate_(5.50 g, 11.3 mmoles) in tetrahydrofuran (50 mL) and methanol (10 mL) was cooled to 0°C. To this was added a cold (0°C) solution of lithium hydroxide monohydrate (0.95 g, 22.6 mmoles) in water (20 mL), dropwise over 15 min. The reaction was stirred at room temperature for additional 3 h. The mixture was concentrated to remove the organic solvents. The resulting residue was dissolved in a minimum amount of water, cooled to 0°C, and treated with cold (0°C) IN HC1 until pH 2 was attained. The resulting solid was collected by filtration, washed with cold water and ether, and then dried overnight under high vacuum to give 5.0 g (94%) as a white solid. ’H-NMR (DMSO- d6) 5 13.05 (bs, 1 H), 11.77 (s, 1 H), 7.98 (s, 1 H), 7.62 (d, J= 8.0 Hz, 1 H), 7.55 (s, 1 H), 7.44 (d, J= 8.2Hz, 1 H), 7.42 (s, 1 H), 7.27 (d, J= 8.2 Hz, 1 H), 7.16 (t, J= 7.6 Hz, 1 H), 7.05 (s, 1 H), 6.65 (d, J= 7.9 Hz, 1 H), 4.27 – 4.22 (m, 1 H), 4.10 – 4.07 (m, 2 H), 3.12 – 3.07 (m, 1 H), 3.03 – 2.99 (m, 1 H), 2.93 – 2.88 (m, 1 H), 2.77 – 2.66 (m, 2 H), 2.47 (s, 3 H), 1.77 – 1.74 (m, 2 H), 1.34 – 1.27 (m, 2 H). Mass spec.: 474.30 (MH)+.

(R)-N-(3-(7-methyl-lH-indazol-5-yl)-l-(4-(l-methylpiperidin-4-yl)piperazin-l-yl)-l- oxopropan-2-yl)-4-(2-oxo-l,2-dihydroquinolin-3-yl)piperidine-l-carboxamide (I). A flask was charged with (R)-3-(7-methyl-lH-indazol-5-yl)-2-(4-(2-oxo-l,2-dihydroquinolin-3- yl)piperidine-l-carboxamido)propanoic acid (2.9 g, 6.11 mmoles), triethylamine (3.00 mL, 21.5 mmoles), l-(l-methylpiperidin-4-yl)piperazine (1.23 g, 6.72 mmoles), and dimethylformamide (10 mL). The resulting solution was treated with 2-(lH-benzotriazole-l-yl)-l, 1,3,3- tetramethyluronium tetrafluoroborate (2.26 g, 7.03 mmoles) in portions. The reaction was allowed to stir at room temperature overnight. The mixture was concentrated under vacuum to remove dimethylformamide. The crude product was dissolved in 7% methanol in di chloromethane and purified by flash chromatography using 7% methanol in di chloromethane containing 2% of aqueous ammonium hydroxide as eluent. The pure fractions were collected and solvent was removed under vacuum. The desired product was crystallized from hot acetone to give the compound having Formula I in 77% yield. Analytical HPLC showed 99.0 % UV purity at 230 nm. The enantiomeric excess (ee) was determined to be >99.9% (conditions: Chiralpak AD column, 4.6 x 250 mm, 10 pm; eluent: 70% (0.05% diethylamine)/heptane/30%ethanol; @1.0 mL/min. for 45 min. The retention times were 18.7 min for R and 28.1 min for S). ‘H-NMR (500 MHz, DMSO-de) 5 ppm 13.01 (s, 1 H), 11.76 (s, 1 H), 7.96 (s, 1 H), 7.62 (d, J= 7.10 Hz, 1 H), 7.60 (s, 1 H), 7.42 (m, 1 H), 7.36 (s, 1 H), 7.26 (d, J = 8.25 Hz, 1 H), 7.14 (m, 1 H), 7.00 (s, 1 H), 6.69 (d, J= 8.25 Hz, 1 H), 4.78 (q, J= 7.79 Hz, 1 H), 4.14 (d, J= 12.37 Hz, 2 H), 3.54 (dd, J= 9.16, 4.58 Hz, 1 H), 3.24 (m, 1 H), 3.11 (m, 1 H), 2.97 (m, 1 H), 2.89 (m, 2 H), 2.69 (m, 4 H), 2.32 (m, 1 H), 2.21 (m, 1 H), 2.07 (m, 4 H), 1.95 (t, J= 8.25 Hz, 1 H), 1.87 (m, J= 11.28, 11.28, 3.55, 3.44 Hz, 1 H), 1.76 (t, J= 12.03 Hz, 2 H), 1.68 (t, J= 11.11 Hz, 2 H), 1.53 (t, J= 8.25 Hz, 1 H), 1.32 (m, 4 H), 1.16 (m, 2 H); 13C-NMR (DMSO-de) 5 16.80, 27.30, 30.51, 30.51, 30.67, 35.50, 38.04, 41.74, 44.00, 44.16, 45.35, 45.78, 48.14, 48.39, 51.45, 54.76, 54.76, 60.61, 114.53, 117.79, 119.29, 119.34, 121.57, 122.78, 127.46, 127.79, 129.29, 129.79, 133.31, 133.72, 136.98, 137.41, 139.12, 156.50, 161.50, 170.42.
Accurate mass analysis: m/z 639.3770, [MH]+, A = -0.2 ppm. Optical rotation: -27.36° @ 589 nm, concentration = 4.71 mg/mL in methanol. DESCRIPTION AND DOSAGE FORM
The physical and chemical properties of zavegepant (BHV-3500) drug substance mono-hydrochloride salt form are provided in Table 1.
Table 1 Physical and Chemical Properties
Biohaven number BHV-3500
Molecular formula C36H47CIN8O3
Molecular weight 675.26 (HO salt); 638.82 (free base)
Appearance White to off-white powder
Melting point ~178°C pH-solubility profile 105 mg/mL at pH = 8.2 and > 300 mg/mL at lower pH pKa 4.8 and 8.8 logD 1.21

Patent
US2022401439Bioorg Med Chem Lett
. 2021 Jan 1;31:127624.
doi: 10.1016/j.bmcl.2020.127624. Epub 2020 Oct 21.
References
- ^ Jump up to:a b c d e f g h https://www.accessdata.fda.gov/drugsatfda_docs/label/2023/216386s000lbl.pdf
- ^ https://www.accessdata.fda.gov/drugsatfda_docs/appletter/2023/216386Orig1s000ltr.pdf
- ^ “Pfizer’s Zavzpret (Zavegepant) Migraine Nasal Spray Receives FDA Approval” (Press release). 10 March 2023.
Further reading
- Croop R, Madonia J, Stock DA, Thiry A, Forshaw M, Murphy A, Coric V, Lipton RB (October 2022). “Zavegepant nasal spray for the acute treatment of migraine: A Phase 2/3 double-blind, randomized, placebo-controlled, dose-ranging trial”. Headache. 62 (9): 1153–1163. doi:10.1111/head.14389. PMC 9827820. PMID 36239038.
- Noor N, Angelette A, Lawson A, Patel A, Urits I, Viswanath O, et al. (2022). “A Comprehensive Review of Zavegepant as Abortive Treatment for Migraine”. Health Psychology Research. 10 (3): 35506. doi:10.52965/001c.35506. PMC 9239361. PMID 35774914.
- Scuteri D, Tarsitano A, Tonin P, Bagetta G, Corasaniti MT (November 2022). “Focus on zavegepant: the first intranasal third-generation gepant”. Pain Management. 12 (8): 879–885. doi:10.2217/pmt-2022-0054. PMID 36189708. S2CID 252681912.
External links
- Clinical trial number NCT04571060 for “Randomized Trial in Adult Subjects With Acute Migraines” at ClinicalTrials.gov
- Clinical trial number NCT03872453 for “Acute Treatment Trial in Adult Subjects With Migraines” at ClinicalTrials.gov
| Clinical data | |
|---|---|
| Trade names | Zavzpret |
| Other names | BHV-3500 |
| License data | US DailyMed: Zavegepant |
| Routes of administration | Nasal |
| Drug class | Calcitonin gene-related peptide receptor antagonist |
| ATC code | None |
| Legal status | |
| Legal status | US: ℞-only[1] |
| Identifiers | |
| showIUPAC name | |
| CAS Number | 1337918-83-8as HCl: 1414976-20-7 |
| PubChem CID | 53472683as HCl: 134819878 |
| DrugBank | DB15688 |
| ChemSpider | 30814207 |
| UNII | ODU3ZAZ94Jas HCl: 000QCM6HAL |
| KEGG | D11898as HCl: D11899 |
| ChEMBL | ChEMBL2397415as HCl: ChEMBL4650220 |
| Chemical and physical data | |
| Formula | C36H46N8O3 |
| Molar mass | 638.817 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| showSMILES | |
| showInChI | |
////////FDA 2023, APPROVALS 2023, Vazegepant, BMS-742413, BHV-3500, ザベジェパント , Zavegepant, ZAVZPRET, BMS
PFIZERCC1=CC(=CC2=C1NN=C2)CC(C(=O)N3CCN(CC3)C4CCN(CC4)C)NC(=O)N5CCC(CC5)C6=CC7=CC=CC=C7NC6=O

NEW DRUG APPROVALS
ONE TIME help to run this blog
$10.00
MAVORIXAFOR
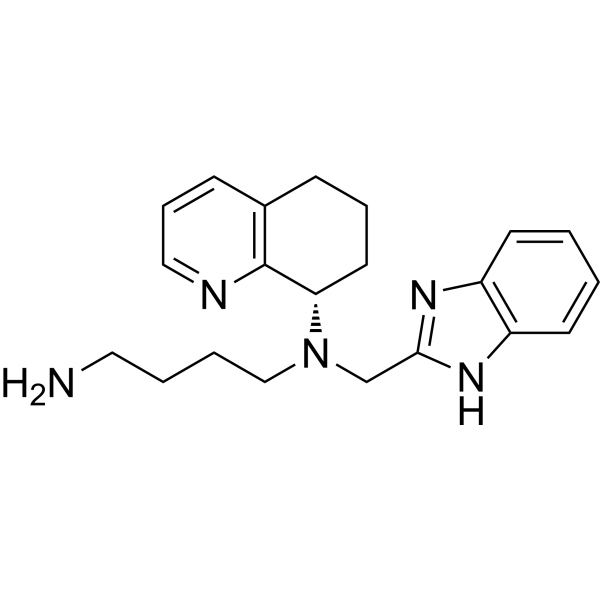
MAVORIXAFOR
AMD 070
N1-(1H-BENZIMIDAZOL-2-YLMETHYL)-N1-((S)-5,6,7,8-TETRAHYDROQUINOLIN-8-YL)-BUTANE-1,4-DIAMINE
fda approved 4/26/2024, To treat WHIM syndrome (warts, hypogammaglobulinemia, infections and myelokathexis), Xolremdi
Mavorixafor (AMD-070) is a potent, selective and orally available CXCR4 antagonist, with an IC50 value of 13 nM against CXCR4 125I-SDF binding, and also inhibits the replication of T-tropic HIV-1 (NL4.3 strain) in MT-4 cells and PBMCs with an IC50 of 1 and 9 nM, respectively.
| Molecular Weight | 349.47 |
|---|---|
| Appearance | Solid |
| Formula | C21H27N5 |
| CAS No. | 558447-26-0 |
| SMILES | NCCCCN(CC1=NC2=C(N1)C=CC=C2)[C@@H]3C4=C(CCC3)C=CC=N4 |
PHASE 2
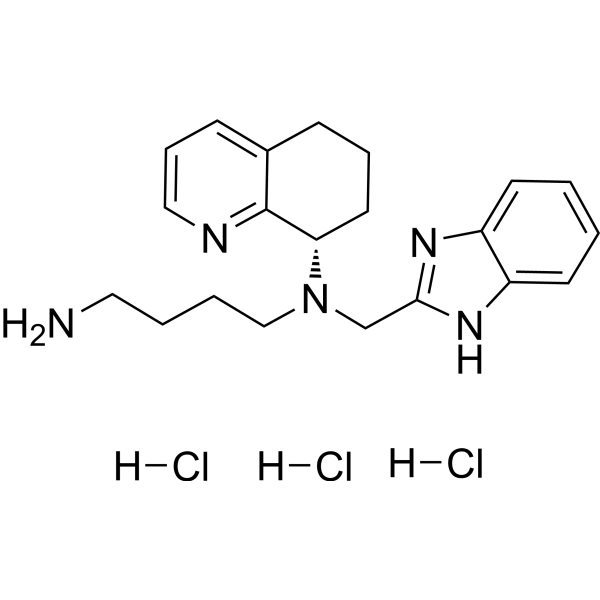
Mavorixafor trihydrochloride
| Molecular Weight | 458.86 |
|---|---|
| Appearance | Solid |
| Formula | C21H30Cl3N5 |
| CAS No. | 2309699-17-8 |
| SMILES | [H]Cl.[H]Cl.[H]Cl.NCCCCN(CC1=NC2=CC=CC=C2N1)[C@@H]3C4=NC=CC=C4CCC3 |

AMD-070 is a small molecule drug candidate that belongs to a new investigational class of anti-HIV drugs known as entry (fusion) inhibitors. Approximately 76% of HIV-patients with measurable viral load are infected with a strain of virus that is resistant to one or more classes of antiretroviral agents, thus reducing treatment options. Unlike many existing HIV drugs that target the virus after it has infected a healthy cell, AMD-070 blocks the virus from entering a healthy cell, thus preventing the replication process. AMD-070 targets the CXCR4 receptor on HIV and prevents the virus from entering and infecting healthy cells. AMD-070 is specific for the CXCR4 receptor and does not interact with any other chemokine receptors in vitro. AMD-070 strongly inhibits viral infection by all CXCR4 using virus (including virus using CXCR4 alone and/or virus using CXCR4 and CCR5) in vitro. AMD-070 is orally bioavailable in animals, it has suitable PK and toxicity profile for oral dosing. AMD-070 shows additive or synergistic effects in vitro in combination with other known anti-HIV agents. AMD-070 is active against CXCR4 using HIV strains that are resistant to existing antiretroviral therapies in vitro, reveals potent anti-HIV activity against CXCR4-using laboratory strains and clinical isolates. MD-070 had been in phase II clinical trials by Genzyme for the treatment of HIV infection. However, this research has been discontinued. AMD-070 has been studied in Phase I/II clinical trials for the treatment of Renal cell carcinoma and Phase I clinical trials for the treatment of malignant melanoma and solid tumours.
PAPER
https://pubs.acs.org/doi/10.1021/acs.oprd.2c00076
Org. Process Res. Dev. 2022, 26, 6, 1831–1836
A novel and practical synthesis of mavorixafor (1) is reported. The novelty of this synthetic route is the use of 8-chloro-5,6,7,8-tetrahydroquinoline (9) and 1,4-diaminobutane as the materials, instead of 8-amino-5,6,7,8-tetrahydroquinoline (4) and N,N-diprotected aminobutyraldehyde (6a or 6b). The preparation of (S)-8-(4-aminobutylamino)-5,6,7,8-tetrahydroquinoline (13) by resolution with N-acetyl-l-leucine was first achieved. Then the one-pot synthesis of 1 from 13 involving protection, condensation, and subsequent hydrolysis was successfully developed. In addition, the final product with a satisfactory purity (>99.5%, detected by both achiral and chiral HPLC) was obtained by a simple operation (salification) without column chromatographic purification.





NEW PAT
Scheme I


Mavorixafor


EXEMPLIFICATION
Example 1: Synthesis of Sulfonate adduct F-2d:
Scheme V:
1) AcOH, NaCI, water 1) Na 2 S 2 O 5 , THF, water
2) n-Heptane, THF 2) THF/n-heptane, acetonitrile
Step 1C Step 1 D


Step 1A: Preparation of Dl
Charge diethyl-4-aminobutyl acetal (E) (1.00 wt, 1.00 eq) to vessel A. Charge acetonitrile (10.0 vol, 7.8 wt) and adjust temperature to 20°C. Heat the mixture to 40°C. Concentrate the reaction mixture to 6.0 vol under reduced pressure at 35 to 45°C.
[0098] Acetonitrile filler (5.0 vol, 3.9wt) at 35 to 45°C. Concentrate the reaction mixture to 6.0 vol under reduced pressure 35 to 45°C. This step is repeated once as described below.
[0099] Acetonitrile filler (5.0 vol, 3.9wt) at 35 to 45°C. Concentrate the reaction mixture to 6.0 vol under reduced pressure at 35 to 45°C. Cool to 20°C.
[00100] Charge di-tert-butyl dicarbonate (1.1 eq, 1.5 wt) to a drum, followed by acetonitrile (0.4 vol, 0.3 wt) and agitate until fully dissolved. Concentrate the reaction mixture to 6.0 vol under reduced pressure at 35 to 45°C.
[00101] Charge this di-tert-butyl dicarbonate solution in acetonitrile to vessel A maintaining 20°C. Charge acetonitrile (1.5 vol, 1.1 wt) to the solution as a line rinse and stir at 20°C for 30 to 60 min..
[00102] Charge 4-dimethylaminopyridine (0.076 wt, 0.10 eq) to the vessel A at 20°C. Heat the solution to 40°C. Concentrate the reaction mixture to 5.0 vol under reduced pressure. Charge acetonitrile (5.0 vol, 3.9 wt) to the solution. Concentrate the reaction mixture to 5.0 vol under reduced pressure.
[00103] Take the resulting solution of Dl into next reaction without isolation.
Step IB: Preparation of Cl
[00104] Charge acetonitrile (2.0 vol, 1.6 wt) at 35 to 45°C to vessel A containing solution of D-1 from Step 1A.
[00105] Charge di-tert-butyl dicarbonate (1.4 eq, 1.9 wt) to a drum, followed by acetonitrile (10.0 vol, 7.8 wt) and agitate until fully dissolved. Charge this di-tert-butyl dicarbonate solution to vessel A, 2 to 6 h while distilling under vacuum at 35 to 45°C maintaining the volume of the reaction at 7.0 vol. Load acetonitrile (3.0 vol, 2.4 wt) over 20 to 40 min. as a line rinse while distilling under vacuum at 35 to 45°C, maintaining the volume of the reaction at 7.0 vol.
[00106] Charge di-tert-butyl dicarbonate, (0.14 eq, 0.19 wt) to a drum, followed by acetonitrile (1.0 vol, 0.74 wt) and agitate until fully dissolved. Charge this di-tert-butyl dicarbonate solution to vessel A over 20 to 40 min.. Charge acetonitrile (0.3 vol, 0.24 wt) over 10 to 20 min as a line rinse while distilling under vacuum at 35 to 45°C, maintaining the volume of the reaction at 7.0 vol.
[00107] Concentrate the reaction mixture to 5.0 vol distilling under vacuum at 35 to 45°C.
[00108] Charge n-heptane, (7.5 vol, 5.1 wt) to the reaction mixture, and concentrate the reaction mixture to 5.0 vol under reduced pressure at 40°C. This step is repeated once as described below.
[00109] Charge n-heptane, (7.5 vol, 5.1 wt) to the reaction mixture, and concentrate the reaction mixture to 5.0 vol under reduced pressure at 40°C.
[00110] Charge decolorizing, activated charcoal (0.2 wt) to the solution and stir for 1 to 2 h at 40°C. Filter the reaction mixture at 40°C. Charge n-heptane, (2.0 vol, 1.4 wt) to the reactor vessel and stir for 5 to 15 min. at 20°C before charging to the filter as a line rinse. Combine the filtrate and wash, and as required adjust to 20°C.
[00111] Take the resulting solution of Cl into next reaction without isolation.
Step 1C: Preparation of Bl
[00112] Charge 15% v/v acetic acid (2.0 vol) caution gas evolution, to vessel A containing solution of Cl from Step IB, maintaining the temperature at 20°C and stir for 10 min. at 20°C. Allow the phases to separate for 15 min. at 20°C. Discharge the aqueous phase to waste, retaining the organic phase in vessel A. This step is repeated once as described below.
[00113] Charge 15% v/v acetic acid (2.0 vol) maintaining 20°C and stir for 10 min. at 20°C. Allow the phases to separate for 15 min. at 20°C. Discharge the aqueous phase to waste, retaining the organic phase in vessel A.
[00114] Adjust the reaction to 30°C. Charge 4% w/w sodium chloride solution (2.1 vol) to the vessel maintaining the temperature at 30°C. Charge glacial acetic acid (4.1 vol, 4.3 wt) to the vessel maintaining 30°C. Stir the reaction mixture for 2 h maintaining the temperature at 30°C.
[00115] Charge purified water, (6.0 vol) at 30°C. Stir the contents for 5 to 10 min. at 30°C, and separate the phases, retaining the upper organic phase in vessel A. Charge the lower aqueous phase to vessel B.
[00116] Charge purified water (4.0 vol) at 30°C and stir for 5 to 10 min. maintaining the temperature at 30°C. Separate the phases at 30°C, retaining the upper organic phase in vessel A. Charge the lower aqueous phase to vessel B.
[00117] Adjust the temperature to 30°C of vessel B containing combined aqueous phases. Charge n-heptane, (2.0 vol, 1.4 wt) to vessel B and stir for 5 to 10 min. maintaining the temperature at 30°C. Separate the phases at 30°C, over 15 min.. Charge the upper organic phase to vessel A and recharge the lower aqueous phase to vessel B. This step is repeated two additional times as described below.
[00118] Charge n-heptane, (2.0 vol, 1.4 wt) to vessel B and stir for 5 to 10 min. maintaining the temperature at 30°C. Separate the phases at 30°C, over 15 min.. Charge the upper organic phase to vessel A and recharge the lower aqueous phase to vessel B.
[00119] Charge n-heptane, (2.0 vol, 1.4 wt) to vessel B and stir for 5 to 10 min. maintaining the temperature at 30°C. Separate the phases at 30°C, over 15 min., discharge the lower aqueous phase to waste and charge the upper organic layer to vessel A.
[00120] Concentrate the combined organic phases in vessel A to 3.0 vol at 10 to 20°C under reduced pressure. Offload the solution to new HDPE drum(s) and line rinse with n-heptane (0.5
vol, 0.4 wt) at 20°C. Homogenize the drum and store as “Bl solution in n-heptane,” and take into next reaction without isolation.
Step ID: Preparation of F-2d
[00121] Calculate a new 1.00 wt based on the above assay.
[00122] Charge “Bl solution in n-heptane” from Step 1C (1.00 wt, 1.00 eq, corrected for w/w assay, ca. 3.0 vol), into an appropriate vessel. THF load (3.0 vol, 2.7 wt). Heat the reaction mixture to 40°C.
[00123] Charge purified water, (0.02 vol, 0.02 wt) followed by sodium metabisulphite, (0.125 eq, 0.08 wt) as a solid via the charge hole at 40°C. Stir the resulting mixture for 30 to 35 min. at 40°C. This step was repeated four additional times to add the reagent in five portions total, as detailed below.
[00124] Charge purified water, (0.02 vol, 0.02 wt) followed by sodium metabisulphite, (0.125 eq, 0.08 wt) as a solid via the charge hole at 40°C. Stir the resulting mixture for 30 to 35 min. at 40°C.
[00125] Charge purified water, (0.02 vol, 0.02 wt) followed by sodium metabisulphite, (0.125 eq, 0.08 wt) as a solid via the charge hole at 40°C. Stir the resulting mixture for 30 to 35 min. at 40°C.
[00126] Charge purified water, (0.02 vol, 0.02 wt) followed by sodium metabisulphite, (0.125 eq, 0.08 wt) as a solid via the charge hole at 40°C. Stir the resulting mixture for 30 to 35 min. at 40°C.
[00127] Charge purified water, (0.02 vol, 0.02 wt) followed by sodium metabisulphite, (0.125 eq, 0.08 wt) as a solid via the charge hole at 40°C. Stir the resulting mixture for 36 hours at 40°C.
[00128] Cool the reaction mixture to 20°C over 3 to 4 h at a target constant rate. Filter the reaction mixture at 20°C on a 1-2 pm cloth.
[00129] Wash the solid with a pre-mixed mixture of THF (0.5 vol, 0.5 wt) and n-heptane (0.5 vol, 0.3 wt) maintaining the temperature at 20°C. This step was repeated an additional three times, as detailed below.
[00130] Wash the solid with n-heptane, (2.0 vol, 1.4 wt) as a line rinse and apply to the filtercake at 20°C.
[00131] Wash the solid with n-heptane, (2.0 vol, 1.4 wt) as a line rinse and apply to the filtercake at 20°C.
[00132] Wash the solid with acetonitrile, (2.0 vol, 1.6 wt) as a line rinse and apply to the filtercake at 20°C.
[00133] Dry the solid at 38°C under a flow of nitrogen for 12 h.
[00134] Determine residual solvent content. Pass criteria acetonitrile <2.0% w/w, n-heptane <2.0% w/w and tetrahydrofuran <2.0% w/w.
[00135] Yield of compound F-2d: 52-69%.
[00136] ‘H NMR (400 MHz, d 6 -DMSO): 8 5.22 (s, 1H), 3.77 (s, 1H), 3.45 (t, 2H), 1.70 (m, 2H), 1.44 (m, 20H) ). 13 C NMR (400 MHz, d 6 -DMSO): 8 152.6, 83.2, 82.0, 46.5, 29.6, 28.1, 26.0. FTIR (wavenumber, cm’ 1 ) 3294, 1721, 1738, 1367, 1233, 1180, 1135, 1109, 1045.
Example 2: Synthesis of F-3a:
Scheme VI:


Step 2A: Preparation of Gl
[00137] Charge J, (1.00 wt, 1.00 eq) to vessel A. Charge purified water, (1.0 vol, 1.0 wt) to vessel A and as necessary adjust the temperature to 20°C. Charge concentrated hydrochloric acid, (4.0 eq, 3.0 vol, 3.6 wt) to vessel A maintaining the temperature at 20°C. Line rinse with purified water, (0.5 vol, 0.5 wt) maintaining the contents of vessel A at 15 to 25°C.
[00138] Charge chloroacetic acid, (1.3 wt, 1.5 eq) and purified water, (1.0 vol, 1.0 wt) to vessel B and as necessary, adjust the temperature to 20°C. Stir until fully dissolved, expected 10 to 20 min.
[00139] Charge the chloroacetic acid solution to vessel A maintaining the temperature of vessel A at 20°C. Line rinse vessel A with purified water, (0.5 vol, 0.5 wt) at 15 to 25°C and charge to vessel B at 20°C. Heat the reaction mixture to 80°C. Stir the reaction mixture at 80°C for 20 h.
[00140] Cool the reaction mixture to 10°C over 1.5 h. Load 47% w/w potassium phosphate solution (6.0 vol) over 60 min. targeting a constant rate maintaining 10°C. Adjust the pH of the reaction mixture by charging 47% w/w potassium phosphate solution to pH 7.0 maintaining the reaction temperature at 10°C. Expected charge is 2.0 to 3.5 vol 47% w/w potassium phosphate solution.
[00141] Stir the slurry for >30 min. maintaining 10°C and rechecking the pH, pass criterion pH 7.0. Filter the reaction mixture through 20 pm cloth at 10°C. Wash the filter-cake with purified water, (1.0 vol, 1.0 wt) at 10°C. This step is repeated additional three times as described below.
[00142] Slurry wash the filter-cake in the reactor vessel with purified water, (10.0 vol, 10.0 wt) for 45 min. to 90 min. at 10°C. Filter the mixture through 20 pm cloth at 10°C.
[00143] Slurry wash the filter-cake in the reactor vessel with purified water, (10.0 vol, 10.0 wt) for 45 min. to 90 min. at 10°C. Filter the mixture through 20 pm cloth at 10°C.
[00144] Slurry wash the filter-cake in the reactor vessel with purified water, (10.0 vol, 10.0 wt) for 45 min. to 90 min. at 10°C. Filter the mixture through 20 pm cloth at 10°C.
[00145] Wash the filter-cake with purified water, (1.0 vol, 1.0 wt) at 10°C. The filter-cake was washed with purified water additional five times as described below.
[00146] Wash the filter-cake with purified water, (1.0 vol, 1.0 wt) at 10°C.
[00147] Wash the filter-cake with acetonitrile, (2×1.3 vol, 2×1.0 wt) at 10°C.
[00148] Dry the filter-cake on the filter under vacuum and strong nitrogen flow through the filter cake at 20°C until the water content is <15.0% w/w by Karl-Fisher analysis.
[00149] Dry the filter-cake on the filter under vacuum and strong nitrogen flow through the filter cake at 30°C until the water content is <5.0% w/w by Karl-Fisher analysis.
[00150] Dry the filter-cake on the filter under vacuum and strong nitrogen flow through the filter cake at 50°C until the water content is <1.0% w/w by Karl-Fisher analysis.
[00151] Yield of compound Gl: about 75%.
Step 2B: Preparation of F-3a
Charge di-/c/7-butyl dicarbonate, (1.85 wt, 1.4 eq) to vessel A followed by N,N-dimethylformamide, (2.6 wt, 2.7 vol) and stir at 20°C for 20 min. until dissolution achieved. Add A,A-diisopropylethylamine, (0.08 wt, 0.11 vol, 0.1 eq) to contents of vessel A at 20°C. Heat the contents of vessel A to 40°C.
[00153] Charge Gl, (1.00 wt) to vessel B followed by YW-di methyl form am ide, (5.2 wt, 5.5 vol) and adjust to 14°C.
[00154] Charge the Gl/DMF solution from vessel B to vessel A over 5 h at 40°C, at an approximately constant rate. Line rinse with Y,Y-di methyl form am ide, (0.4 wt, 0.4 vol), maintaining vessel A at 40°C. Stir the resulting reaction mixture at 40°C for 16 h.
[00155] Charge decolorizing charcoal activated, (0.20 wt). Adjust the mixture to 40°C and stir at 40°C for 60 to 90 min..
[00156] Clarify (filter) the reaction mixture into vessel B at 40°C. Charge N,N-dimethylformamide, (0.9 wt, 1.0 vol) via vessel A and filter at 40°C. Charge purified water, (3.5 vol) to the combined filtrates, over 60 min., maintaining the temperature at 40°C. As required, cool the mixture to 35°C over 30 to 60 min..
[00157] Filler F-3a, (0.02 wt) as seed material at 35°C. Stir at 34°C for 1.5 h then check for crystallization. Cool slurry to 30°C over 40 min.
[00158] Filler F-3a, (0.02 wt) as seed material at 30°C. Stir at 30°C for 1.5 h then check for crystallization.
[00159] Cool slurry at 20°C over 3.5 h at a targeted constant rate. Stir at 20°C for 3 hours. Charge purified water, (1.0 vol), maintaining the temperature at 20°C over 60 min..
Stir at 20°C for 3 hours.
[00160] Cool slurry to 2°C over 2.5 h. Stir at 2°C for 2.5 hours. Filter through 20 pm cloth and pull dry until no further filtrate passes. Wash the solid with pre-mixed Y,Y-di methyl form am ide / purified water, (2.0 vol, 1:2 v:v) at 2°C. Wash the solid with purified water, (2 x 3.0 vol) at 2°C. Dry under vacuum at 28°C until KF <0.2% w/w, and Y,Y-di methyl form am ide <0.4% w/w.
[00161] Yield of compound F-3a: 62-70%.
Example 3: Synthesis of Mavorixafor:
Scheme VI:


nce
Step 3A: Preparation of imine Q-1
[00162] To vessel A charge purified water, (8.7 vol, 8.7 wt) followed by potassium phosphate, (5.52 eq, 5.3 wt) portion-wise and cool to 15°C. Charge tetrahydrofuran, (4.3 vol, 3.8 wt) and n-heptane, (2.2 vol, 1.5 wt) to vessel A and cool the biphasic mixture to 0°C. Charge Fl, (1.00 eq, 1.00 wt) to the vessel in 2 portions maintaining 0°C.
[00163] Charge F-2d, (1.10 eq, 1.95 wt) to the vessel in 4 portions maintaining 0°C, ensuring portions are spaced by 10 min.. Stir the resulting biphasic mixture for 1.5 h at 0°C. Allow the layers to separate for 45 min. at 0°C before separating the layers. Retain the upper organic phase within vessel A.
[00164] Take the resulting solution of Ql into next reaction without isolation.
Step 3B: Preparation of amine P-1
[00165] To vessel B, charge tetrahydrofuran, (6.0 vol, 5.3 wt) and adjust to 15°C. Charge zinc chloride, (1.5 eq, 0.92 wt) to vessel B in 4 portions, maintaining 10 to 30°C. Adjust the reaction mixture in vessel B to 15°C. Stir the mixture at 15°C for 1 hour. Charge sodium borohydride,(1.0 eq, 0.17 wt) to vessel B in 2 portions maintaining 15°C. Cool the reaction mixture in vessel B to 15°C. Stir the mixture for 1 hour maintaining 15°C. Cool the reaction mixture in vessel B to -5°C.
[00166] Cool the retained organic solution of Ql in vessel A, from Step 3A, to -5°C.
[00167] Charge the organic solution in vessel A into vessel B over 1 to 2 h maintaining -5°C. Charge tetrahydrofuran, (1.0 vol, 0.9 wt) to vessel A as a line rinse and adjust to -5°C. Transfer the contents of vessel A to vessel B maintaining -5°C.
[00168] Stir the resulting reaction mixture in vessel B for 1.5 h maintaining -5°C.
[00169] Charge purified water, (4.5 vol, 4.5 wt) and glacial acetic acid, (1.0 eq, 0.27 wt, 0.26 vol) to the cleaned vessel A and cool to 0°C. Charge the contents of vessel B to vessel A over 1 to 2 h maintaining 0°C. Charge tetrahydrofuran, (1.0 vol, 0.9 wt) to vessel B as a vessel rinse, cool to 0°C and transfer to vessel A maintaining 0°C.
[00170] Warm the resulting mixture in vessel A to 30°C. Stir the resulting mixture in vessel A at 30°C for 1 h. Allow the layers to settle for 15 min. at 30°C before separating the layers. Retain the upper organic phase.
[00171] Cool the retained organic phase to 15°C. Charge to the vessel 25% w/w ammonia solution (3.0 vol) at 10 to 30°C. Cool the reaction mixture to 20°C. Charge to the vessel 25% w/w ammonium chloride solution (3.0 vol) at 20°C and stir for 1 h. Separate the layers for 15 min. at 20°C, retain the upper organic phase. Wash the retained organic phase with 10% w/w sodium chloride solution (3.0 vol) at 20°C for 10 min.. Allow the layers to settle for 10 min. at 20°C before separating and retaining the upper organic phase within the vessel.
[00172] Charge tert-butyl methyl ether, (0.5 vol, 0.4 wt) to the organic phase. Cool the mixture to 5°C. Adjust the pH of the reaction mixture to pH 5 with hydrochloric acid aqueous solution (expected ca. 9.0 vol) over 1 h at a targeted constant rate at 5°C. Stir the mixture at 5°C for 45 min.. Measure the pH of the aqueous phase to confirm the value is pH 5.
[00173] Charge sodium chloride, (2.1 wt) to the reaction mixture at 5°C and stir the mixture until everything is dissolved. Adjust the temperature of the reaction mixture to 20°C. Separate the layers at 20°C and retain the organic phase within the vessel. Tetrahydrofuran charge, (1.5 vol, 1.3 wt) maintaining 20°C.
[00174] Charge to the vessel 24% w/w sodium chloride solution (7.5 vol) at 20°C and stir for 10 min.. Separate the layers at 20°C and retain the organic phase in the vessel. This step is repeated additional one more time as described below.
[00175] Charge to the vessel 24% w/w sodium chloride solution (7.5 vol) at 20°C and stir for 10 min.. Separate the layers at 20°C and retain the organic phase in the vessel.
[00176] Heat the retained organic phase to 35°C and concentrate the mixture to 6.0 vol under reduced pressure maintaining 35°C.
[00177] Tetrahydrofuran charge, (15.0 vol, 13.2 wt) maintaining 35°C. Concentrate the mixture to 6.0 vol under reduced pressure maintaining 35°C.
[00178] Tetrahydrofuran charge, (15.0 vol, 13.2 wt) maintaining 35°C. Concentrate the mixture to 11.0 vol under reduced pressure maintaining 35°C.
[00179] Cool the mixture to -5°C. Load tert-butyl methyl ether, (10.0 vol, 7.4 wt) over 1 h maintaining -5°C. Stir the mixture at -5°C for 1.5 hours. Filter the solid on 1 to 2 pm filter cloth at -5°C. Wash the solid with pre-mixed tetrahydrofuran, (1.9 vol, 1.7 wt) and tert-butyl methyl ether, (3.1 vol, 1.9 wt) at -5°C as a displacement wash.
[00180] Wash the solid with tert-butyl methyl ether, (5.0 vol, 3.7 wt) at -5°C.
[00181] Dry the solid on the filter under a flow of nitrogen at 23°C.
[00182] Yield of compound P-1: 76-87%.
Step 3C: Preparation of compound 0-1
[00183] Charge purified water, (2.0 vol, 2.0 wt) followed by potassium phosphate, (3.3 eq, 1.54 wt), carefully portion-wise, maintaining <15°C, to vessel A. Charge toluene, (4.5 vol, 3.9 wt) to the vessel maintaining <15°C. As necessary, adjust the temperature to 10°C.
[00184] Charge P-1, (1.00 eq, 1.00 wt) to the vessel in two portions maintaining 10°C. Stir the reaction mixture at 10°C for 15 min..
[00185] Load F-3a, (1.1 eq, 0.64 wt) in 4 equal portions ensuring portions are spaced by 10 min. at 10°C.
[00186] Tetrabutylammonium iodide (TBAI) filler (0.20 eq, 0.16 wt). Heat the reaction mixture to 40°C. Stir the reaction mixture at 40°C for 30 h.
[00187] Charge pre-mixed 2-mercaptoacetic acid, (0.40 eq, 0.08 wt, 0.06 vol), and toluene, (0.5 vol, 0.4 wt) over 20 min. to Vessel A at 40°C. Line rinse with toluene, (0.5 vol, 0.4 wt) at 40°C. Adjust the temperature of the reaction mixture to 50°C. Stir the mixture at 50°C for 2.5 hours.
[00188] Adjust the temperature of Vessel A to 20°C. Charge purified water, (3.0 vol, 3.0 wt) maintaining 20°C. Stir the reaction mixture at 20°C for 15 min. and transfer to a new, clean HDPE container. Line/vessel rinse with toluene, (0.5 vol, 0.4 wt) at 20°C. Clarify (filter) the reaction mixture via a 1 pm filter at 20°C into clean Vessel A. Wash the vessel and the filter with toluene, (0.5 vol, 0.4 wt) at 20°C. Allow the layers to separate for 15 min. at 20°C, retaining the upper organic layer (organic layer 1).
[00189] Wash the aqueous layer with toluene, (2.5 vol, 2.2 wt) at 20°C for 15 min.. Allow the layers to separate for 15 min. at 20°C. Retain the upper organic layer (organic layer 2).
[00190] Combine the organic layer 1 and organic layer 2 and adjust the temperature to 20°C. Wash the combined organic layers with 10% w/w sodium chloride solution (5.0 vol) at 20°C for 15 min.. Allow the layers to settle for 15 min. at 20°C. Retain the upper organic layer.
[00191] Take the resulting solution of Ol into next reaction without isolation.
Step 3D: Preparation of compound Kl
[00192] Charge n-butanol, (2.4 wt, 3.0 vol) to vessel B and adjust to 5°C. Charge concentrated sulfuric acid, (1.1 wt, 5.0 eq, 0.6 vol) slowly to Vessel B maintaining <15°C. Line rinse with toluene, (0.4 wt, 0.5 vol) maintaining <15°C. Adjust the temperature of Vessel B to 25°C.
[00193] Heat the n-butanol/ sulfuric acid solution in Vessel B to 55°C. Charge the organic layer from Vessel A (from Step 3C) to the butanol/ sulfuric acid solution in Vessel B over 60 to 90 min. maintaining 55°C. Charge toluene, (1.3 wt, 1.5 vol) to Vessel A as a line rinse and transfer to Vessel B maintaining 55°C. Stir the contents of Vessel B at 55°C for 1.5 h.
[00194] Stir the mixture in Vessel B for 4.5 h at 55°C. Cool the contents of Vessel B to 20°C over 10 h. Filter the slurry over 1-2 pm filter cloth under nitrogen at 20°C. Wash the filter cake with pre-mixed toluene, (3.5 wt, 4.0 vol) and n-butanol, (1.0 vol, 0.8 wt) at 20°C. Wash the filter cake with toluene, (4.3 wt, 5.0 vol) at 20°C. Dry the solid at 30°C under vacuum.
[00195] Correct the output weight for assay. Expected 50-55% w/w.
[00196] Yield of compound K1: 89-92%.
Step 3E: Preparation of Mavorixafor Drug Substance
[00197] Charge Kl, (1.00 eq, 1.00 wt, corrected for HPLC assay) in vessel A followed by nitrogen-purged purified water, (2.0 wt, 2.0 vol) and if necessary, adjust the temperature to 20°C. Charge nitrogen-purged toluene, (12.0 wt, 14.0 vol) to the solution maintaining 20°C. Charge nitrogen-purged n-butanol, (0.8 wt, 1.0 vol) to the solution maintaining 20°C. Heat the biphasic mixture to 30°C. Charge nitrogen-purged 3.0 M aqueous sodium hydroxide solution (6.2 eq, 5.9 vol) maintaining 30°C. Check the pH (expected 12 to 13). Adjust the pH of the aqueous layer to pH 10.0 with nitrogen-purged 0.3 M sulfuric acid solution (expected up to 2.5 vol) maintaining 30°C. Stir the mixture at 30°C for 45 min..
[00198] Measure the pH to confirm the value is pH 10.0.
[00199] Allow the layers to settle at 30°C for 30 min. and separate the layers retaining the organic phase in the vessel, and discharge the aqueous layer into a separate container (container C).
[00200] Charge pre-mixed toluene, (4.1 wt, 4.7 vol) and n-butanol, (0.24 wt, 0.3 vol) to a separate vessel; heat the contents to 30°C and charge the aqueous layer from container C. As required adjust the temperature to 30°C and stir for 5 to 10 min. at 30°C. Allow the phases to separate for 10 to 15 min. at 30°C. Discharge the aqueous phase to waste and combine the organic phase to the organic phase in vessel A.
[00201] Charge nitrogen-purged purified water, (2.0 wt, 2.0 vol) to the organic layer maintaining the temperature at 30°C and stir for 5 to 10 min. at 30°C. Allow the phases to separate for 10 to 15 min. at 30°C. Discharge the aqueous phase to waste retaining the organic phase in the vessel. Heat the retained organic solution to 40°C. Concentrate the resulting organic phase to 7.0 vol by vacuum distillation at 40°C.
[00202] Charge nitrogen -purged toluene, (13.0 wt, 15.0 vol) to the mixture and concentrate the solution 7.0 vol by vacuum distillation at 40°C. This step is repeated additional one time as described below.
[00203] Charge nitrogen -purged toluene, (13.0 wt, 15.0 vol) to the mixture and concentrate the solution 7.0 vol by vacuum distillation at 40°C.
[00204] Charge nitrogen-purged toluene, (7.0 wt, 8.0 vol) to the mixture at 40°C, heat to 55°C and clarify the hot reaction mixture under nitrogen via a 1 pm filter.
[00205] Charge clarified nitrogen-purged toluene, (1.7 wt, 2.0 vol) to the mixture as a line and vessel rinse at 40°C. Concentrate the solution to 7.0 vol by vacuum distillation at 40°C. At the end of the distillation the product is expected to have precipitated. Heat the mixture to 63°C.
[00206] Adjust the temperature to 60.5°C. This batch will be referred to as the main batch.
[00207] Load seed material, (0.02 wt) to a new clean container. Charge clarified nitrogen-purged toluene, (0.09 wt, 0.10 vol) to this seed material and gently shake.
[00208] Seed the main batch with the slurry maintaining the temperature at 60.5 ± 2°C. Stir the reaction at the 60.5± 2°C for 1 hour.
[00209] Cool to 40°C for 2.5 h. Stir the reaction at 40°C for 1 hour.
[00210] Cool to 30°C over 2 h.. Stir the reaction at 30°C for 1 h.
[00211] Cool to 25°C 50 min. Stir the reaction at 25°C over 2 hours.
[00212] Cool to 2°C over 4 h. Stir the mixture for 12 hours at 2°C.
[00213] Filter the mixture at 2°C over 1 to 2 pm cloth. Wash the filter cake with clarified nitrogen-purged toluene, (2.0 vol, 1.7 wt) at 2°C. Dry the filter cake under vacuum and a flow of nitrogen for 1.5 h.
[00214] Dry the solid at 40°C under vacuum and a flow of nitrogen until drying specification is achieved.
[00215] Yield of the final compound mavorixafor: 72%.
[00216] When toluene is used as the recrystallization solvent, optionally with a dissolution aid such butanol or methanol, for maxorixa for recrystallization, advantages were found compared to using dichloromethane and isopropyl acetate. We have found that these solvents do not react with the API, and accordingly we believe that this change has caused the significant reduction of impurities A (imine), B (N-formyl) and C (acetamide) that we have observed.
[00217] In some embodiments, the mavorixafor composition included 7000, 6000, 5000, 4500, 4450, 4000, 3500, 3000, 2500, 2000, 1750, 1700, 1650, 1600, 1550, 1500, 1450, 1400, 1400, 1400, 1400 gold 50 ppm of toluene or less. In some embodiments, the mavorixafor composition comprises a detectable amount of toluene. In some embodiments, the mavorixafor composition comprises from a detectable amount of toluene to 1350 ppm of toluene.

////////

AS ON DEC2021 3,491,869 VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@amcrasto
| Description | Mavorixafor (AMD-070) is a potent, selective and orally available CXCR4 antagonist, with an IC50 value of 13 nM against CXCR4 125I-SDF binding, and also inhibits the replication of T-tropic HIV-1 (NL4.3 strain) in MT-4 cells and PBMCs with an IC50 of 1 and 9 nM, respectively. |
|---|---|
| IC50 & Target[1] | 125I-SDF-CXCR413 nM (IC50)HIV-1 (NL4.3 strain)1 nM (IC50, in MT-4 cells)HIV-1 (NL4.3 strain)9 nM (IC50, in PBMCs)HIV-1 (NL4.3 strain)3 nM (IC90, in MT-4 cells)HIV-1 (NL4.3 strain)26 nM (IC90, in PBMCs) |
| In Vitro | Mavorixafor (AMD-070) is a potent and orally available CXCR4 antagonist, with an IC50 value of 13 nM against CXCR4 125I-SDF binding, and also inhibits the replication of T-tropic HIV-1 (NL4.3 strain) in MT-4 cells and PBMCs with an IC50 of 1 and 9 nM, respectively. Mavorixafor (AMD-070) shows no effect on other chemokine receptors (CCR1, CCR2b, CCR4, CCR5, CXCR1, and CXCR2)[1]. Mavorixafor (AMD-070) (6.6 µM) significantly suppresses the anchorage-dependent growth, the migration and matrigel invasion of the B88-SDF-1 cells[2].MCE has not independently confirmed the accuracy of these methods. They are for reference only. |
| In Vivo | Mavorixafor (AMD-070) (2 mg/kg, p.o.) significantly reduces the number of metastatic lung nodules in mice, and lowers the expression of human Alu DNA in mice, without body weight loss[2].MCE has not independently confirmed the accuracy of these methods. They are for reference only. |
| Clinical Trial | NCT NumberSponsorConditionStart DatePhaseNCT00089466National Institute of Allergy and Infectious Diseases (NIAID)|AIDS Clinical Trials GroupHIV InfectionsNovember 2004Phase 1|Phase 2NCT02667886X4 PharmaceuticalsClear Cell Renal Cell CarcinomaJanuary 2016Phase 1|Phase 2NCT02823405X4 PharmaceuticalsMelanomaSeptember 15, 2016Phase 1NCT00361101Genzyme, a Sanofi Company|SanofiHIV Infections|X4 Tropic VirusOctober 2005Phase 1NCT03005327X4 PharmaceuticalsWHIM SyndromeDecember 2016Phase 2NCT04274738X4 PharmaceuticalsWaldenstrom´s MacroglobulinemiaApril 30, 2020Phase 1NCT04154488X4 PharmaceuticalsNeutropeniaOctober 16, 2020Phase 1NCT03995108X4 PharmaceuticalsWHIM SyndromeOctober 17, 2019Phase 3NCT05103917Abbisko Therapeutics Co, LtdTriple Negative Breast CancerJuly 21, 2021Phase 1|Phase 2NCT00063804National Institute of Allergy and Infectious Diseases (NIAID)|AIDS Clinical Trials GroupHIV Infections Phase 1NCT02923531X4 PharmaceuticalsClear Cell Renal Cell CarcinomaDecember 7, 2016Phase 1|Phase 2NCT02680782X4 Pharmaceuticals|CovanceHealthyJanuary 12, 2016Phase 1 |
REF
- [1]. Skerlj RT, et al. Discovery of novel small molecule orally bioavailable C-X-C chemokine receptor 4 antagonists that are potent inhibitors of T-tropic (X4) HIV-1 replication. J Med Chem. 2010 Apr 22;53(8):3376-88. [Content Brief][2]. Chow LN, et al. Impact of a CXCL12/CXCR4 Antagonist in Bleomycin (BLM) Induced Pulmonary Fibrosis and Carbon Tetrachloride (CCl4) Induced Hepatic Fibrosis in Mice. PLoS One. 2016 Mar 21;11(3):e0151765. [Content Brief][3]. Uchida D, et al. Effect of a novel orally bioavailable CXCR4 inhibitor, AMD070, on the metastasis of oral cancer cells. Oncol Rep. 2018 Jul;40(1):303-308. [Content Brief]
/////////////////////////////////////////////////////////////////////////////MAVORIXAFOR, AMD 070, PHASE 2

NEW DRUG APPROVALS
0NE TIME
$10.00
SMARTCHEM FROM ROW2 TECHNOLOGIES
Are you aware of any Chemical Database which offers one stop solution to the Sourcing, R&D and Business Development department? Explore Smartchem to Quickly find Suppliers (Procurement), Customers (BD) & Synthetic pathways (R&D)
Is this the information you looking for? Evaluate SmartChem, lets schedule a demo. Try us once. You will use us for life.
Anand Ramakrishnan |Vice President – Sales & Operations | +91 9821384045 | +1 973-795-1141 | ranand@row2technologies.com | http://www.row2technologies.com | WhatsApp: +91 9821384045
Anthony Crasto | Advisor | +91 9321316780 | c_acrasto@row2technologies.com | amcrasto@gmail.com | www.row2technologies.com | WhatsApp: +91 9321316780 |
Amcenestrant (SAR 439859)
Amcenestrant (SAR 439859)
アムセネストラント
| Molecular Weight | 554.48 |
|---|---|
| Formula | C31H30Cl2FNO3 |
| CAS No. | 2114339-57-8 |
6-(2,4-dichlorophenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulene-2-carboxylic acid
8-(2,4-dichlorophenyl)-9-(4-{[(3 S )-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dihydro – 5H- Benzo[7]annulene-3-carboxylic acid
8-(2,4-Dichlorophenyl)-9-(4-{[(3 S )-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dihydro-5 H -benzo [7]annulene-3-carboxylic acid
C31H30Cl2FNO3 : 554.48 [ 2114339-57-8 ] _ _ _ _ _ _
| Efficacy | Antineoplastic, Selective estrogen receptor downregulator |
|---|---|
| Comment | Selective estrogen receptor downregulator (SERD) Treatment of breast cancer |
SAR439859 (compound 43d) is an orally active, nonsteroidal and selective estrogen receptor degrader (SERD). SAR439859 is a potent ER antagonist and has ER degrading activity with an EC50 of 0.2 nM for ERα degradation. SAR439859 demonstrates robust antitumor efficacy and limited cross-resistance in ER+ breast cancer.
Amcenestrant is an orally available, nonsteroidal selective estrogen receptor degrader/downregulator (SERD), with potential antineoplastic activity. Upon oral administration, amcenestrant specifically targets and binds to the estrogen receptor (ER) and induces a conformational change that promotes ER degradation. This prevents ER-mediated signaling and inhibits both the growth and survival of ER-expressing cancer cells.
Amcenestrant is reported to be a selective estrogen receptor degrader (SERD) which has estrogen receptor antagonist properties and accelerates the proteasomal degradation of the estrogen receptor. Amcenestrant is under clinical investigation as an anticancer agent, in particular for treatment of breast cancer.
The compound and processes for preparation thereof are described in International Publication No. WO 2017/140669.
Crystalline forms are described in International Publication No. WO 2021/116074.
PAPER
Journal of Medicinal Chemistry (2020), 63(2), 512-52
https://pubs.acs.org/doi/10.1021/acs.jmedchem.9b01293
6-(2,4-Dichlorophenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3- yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulene-2-carboxylic Acid (43d).
To a solution of 6-(2,4-dichloro-phenyl)-5-[4-[1-(3-fluoropropyl)-pyrrolidin-3-yloxy]-phenyl]-8,9-dihydro-7H-benzocycloheptene-2-carboxylic acid methyl ester (42d) (80 mg, 140.72 μmol) in methanol (5 mL) was added 5 N NaOH (562.88 μL), the reaction mixture was heated to 60 °C for 5 h, and the solvent was removed under reduced pressure. The residue was taken up in water (10 mL), and aqueous HCl (5 M) was added to pH 7. The slurry was extracted with dichloromethane, dried over magnesium sulfate, filtered, and concentrated under reduced pressure. The solid was purified by column chromatography eluting with a mixture of dichloromethane, acetonitrile, and methanol (90/5/5 v/v/v) to give 60 mg (77%) of 6- (2,4-dichlorophenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]- oxyphenyl]-8,9-dihydro-7H-benzo[7]annulene-2-carboxylic acid (43d). 1 H NMR (400 MHz, DMSO-d6): 1.68 (m, 1H), 1.79 (dm, J = 25.3 Hz, 2 H), 2.07 to 2.23 (m, 5H), 2.38 (m, 1H), 2.46 (t, J = 7.2 Hz, 2H), 2.52 (m, 1H), 2.62 (m, 1H), 2.55 to 2.89 (m, 3H), 4.47 (td, J = 6.2 and 47.6 Hz, 2H), 4.72 (m, 1H), 6.63 (d, J = 8.9 Hz, 2H), 6.71 (m, 3H), 7.18 (d, J = 8.4 Hz, 1H), 8.26 (dd, J = 2.0 and 8.4 Hz, 1H), 7.58 (d, J = 2.0 Hz, 1H), 7.63 (d, J = 8.4 Hz, 1H), 7.79 (s, 1H), 12.3 (m, 1H). LCMS: 554 (M + H)+ .
PATENT
Amcenestrant can be prepared according to methods known from the literature, for example U.S. Patent No. 9,714,221.
Example 1: Preparation of amorphous Amcenestrant
[00164] Amcenestrant (20 mg, prepared according to U.S. Patent No. 9,714,221) was dissolved in ethyl acetate (0.2 mL) at room temperature (25°C). Solution was left in opened flask at RT for 16 days, until all the solvent evaporated. Obtained solid was analyzed by XRPD.
PATENT
U.S. Patent No. 9,714,221
https://patents.google.com/patent/US9714221B1/en
PATENT
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2017140669
Example 51. 6-(2,4-dichlorophenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulene-2-carboxylic acid
Methode B:
Step 1 : 6-(2,4-dichloro-phenyl)-5-{4-[1-(3-fluoro-propyl)-pyrrolidin-3-yloxy]-phenyl}-8,9-dihydro-7H-benzocycloheptene-2-arboxylic acid methyl ester.
To a solution of methyl 8-bromo-9-(4-{[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dihydro-5H-benzo[7]annulene-3-carboxylate hydrobromide (D5) (150 mg, 298.56 μιηοΙ), in dioxane (12 ml) and water (2 ml), was added 2,4-dichlorophenyl-boronic acid (62.67 mg, 328.41 μηηοΙ), Cs2C03 (204.48 mg, 626.97 μηιοΙ), and Pd(dppf)CI2 (14.63 mg, 17.91 μιηοΙ). The reaction mixture was heated at 90°C for 3 hours, and partitioned between AcOEt and water. The phases were separated and the organic phase washed with brine, dried over MgS04 and concentrated under reduced pressure. The residue was purified by column chromatography eluting with a mixture of DCM, acetonitrile and MeOH (96/2/2; V/V/V) to give 80 mg (47%) of 6-(2,4-dichloro-phenyl)-5-{4-[1-(3-fluoro-propyl)-pyrrolidin-3-yloxy]-phenyl}-8,9-dihydro-7H-benzocycloheptene-2-arboxylic acid methyl ester.
LC/MS (m/z, MH+): 568
Step 2 : 6-(2,4-dichlorophenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulene-2-carboxylic acid
To a solution of 6-(2,4-dichloro-phenyl)-5-{4-[1-(3-fluoro-propyl)-pyrrolidin-3-yloxy]-phenyl}-8,9-dihydro-7H-benzocycloheptene-2-arboxylic acid methyl ester (80 mg, 140.72μιηο!) in MeOH (5 ml) was added a solution of NaOH (562.88 μΙ, 5 M) and the reaction mixture was heated at 60°C for 5 hours and the solvent removed under reduced pressure. The residue was taken up in water (10 ml) and aqueous HCI (5 M) added to pH
7. The slurry was extracted with DCM, dried over MgS04 and concentrated under reduced pressure. The solid was purified by column chromatography eluting with a mixture of DCM, acetonitrile and MeOH (90/5/5; V/V/V) to give 60 mg (77%) of 6-(2,4-dichlorophenyl)-5-[4-[(3S)-1-(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulene-2-carboxylic acid.
PATENT
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2019020559
Intermediate (c). Tert-butyl (3S)-3-[4-(4,4!5!5-tetramethyl-1 !3,2-dioxaborolan-2yl)phenoxy]pyrrolidine-1 -carboxylate
To a solution of commercially available 4-(4,4,5,5-tetramethyl-1 ,3,2-dioxaborolan-2-yl)phenol (a) (82.7 g, 364.51 mmol) in THF (2 L) was added under argon (R)-1 -N-Boc-3-hydroxypyrrolidine (b) (84.43 g, 437.41 mmol) followed by Ν,Ν,Ν’,Ν’-tetramethylazodicarboxamide (99.1 g, 546.77 mmol). The clear reaction mixture turned orange and triphenylphosphine (143.41 g, 546.77 mmol) was added. The reaction mixture was stirred at room temperature for 24 hours, meanwhile a precipitate of triphenylphosphine oxide formed (Ph3P=0). The reaction mixture was poured in water (1 .5 L) and extracted with ethyl acetate (AcOEt) (3×1 .5 L). Gathered organic phases were dried over magnesium sulfate (MgS04), filtered and concentrated under reduced pressure. The residue was taken up into diisopropylether (1 .5 L) and the solid formed (Ph3P=0) was filtered. The solvent was concentrated under reduced pressure and the residue purified by column chromatography eluting with a mixture of heptane with AcOEt (90/10; v/v) to give 145 g (100%) of tert-butyl (3S)-3-[4-(4,4,5,5-tetramethyl-1 ,3,2-dioxaborolan-2-yl)phenoxy]pyrrolidine-1 -carboxylate (c) as a colorless oil.
1H NMR (400 MHz, DMSO-d6, δ ppm): 1 .27 (s : 12H); 1 .39 (s : 9H); 2.05 (m : 1 H); 2.14 (m : 1 H); 3.37 (3H); 3.55 (m : 1 H); 5.05 (s : 1 H); 6.94 (d, J = 8.4 Hz : 2H); 7.61 (d, J = 8.4 Hz : 2H)
Intermediate (d). (3S)-3-[4-(4,4,5,5-tetramethyl-1 ,3,2-dioxaborolan-2yl)phenoxy]pyrrolidine, hydrochloride
To a solution of (S)-tert-butyl 3-(4-(4,4,5,5-tetramethyl-1 ,3,2-dioxaborolan-2-yl)phenoxy)pyrrolidine-1 -carboxylate (c) (80 g, 195.23 mmol) in MeOH (450 ml) was added slowly HCI 4N in dioxane (250 ml).
After 1 .5 hours, the reaction mixture was concentrated under reduced pressure and the residue was taken up into Et20 with stirring to give a solid which then was filtered and dried under vacuum to give 61.8 g (95%) of (3S)-3-[4-(4,4,5,5-tetramethyl-1 ,3,2-dioxaborolan-2yl)phenoxy]pyrrolidine, hydrochloride (d) as a white powder.
1H NMR (400 MHz, DMSO-d6, δ ppm): 1.28 (s : 12H); 2.10 (m : 1 H); 2.21 (m : 1 H); 3.31 (3H); 3.48 (m : 1 H); 5.19 (m : 1 H); 6.97 (d, J = 8.4 Hz : 2H); 7.63 (d, J = 8.4 Hz : 2H); 9.48 (s : 1 H); 9.71 (s : 1 H).
LC/MS (m/z, MH+): 290
Intermediate (e). (3S)-1 -(3-fluoropropyl)-3-[4-(4,4,5,5-tetramethyl-1 ,3,2-dioxaborolan-2-yl)phenoxy]pyrrolidine
To a suspension of (S)-3-(4-(4,4,5,5-tetramethyl-1 ,3,2-dioxaborolan-2-yl)phenoxy)pyrrolidine hydrochloride (d) (20 g, 61.42 mmol) in acetonitrile (100 ml), was added K2C03 (21 .22 g, 153.54 mmol) and 1 -iodo-3-fluoropropane (12.15 g, 61.42 mmol), under argon. The reaction
mixture was stirred at 40°C for 24 hours. After cooling to room temperature, the reaction mixture was filtered and washed with acetonitrile. The filtrate was concentrated under reduced pressure and the residue was taken up in DCM and the solid formed was filtered and washed with DCM. The filtrate was concentrated to give 21.5 g (100%) of (3S)-1 -(3-fluoropropyl)-3-[4-(4,4,5,5-tetramethyl-1 ,3,2-dioxaborolan-2-yl)phenoxy]pyrrolidine (e) as a yellow foam.
1H NMR (400 MHz, DMSO-d6, δ ppm): 1.27 (s : 12H); 1 .77 (m : 2H); 1 .84 (m : 1 H); 2.27 (m : 1 H); 2.41 (m : 1 H); 2.49 (2H); 2.62 (dd, J = 2.6 and 10.4Hz : 1 H); 2.69 (m : 1 H); 2.83 (dd, J = 6.2 and 10.4Hz : 1 H); 4.47 (td, J = 6.2 and 47Hz : 2H) ; 4.99 (m : 1 H); 6.77 (d , J = 8.4 Hz : 2H); 7.58 (d, J = 8.4 Hz : 2H).
LC/MS (m/z, MH+): 350
Intermediate (B). 5-oxo-6,7,8,9-tetrahydro-5H-benzo[7]annulen-2-yl 2,2-dimethylpropanoate
To a solution of 2-hydroxy-6,7,8,9-tetrahydro-5H-benzo[7]annulen-5-one (A) (1 .52 g, 8.63 mmol), in acetone (60 ml), was added K2C03 (1 .19 g, 8.63 mmol) and pivaloyl chloride (1.06 ml, 8.63 mmol). The reaction mixture was stirred at room temperature for 16 hours, filtered and concentrated under reduced pressure. The residue was purified by flash chromatography eluting with a gradient of heptane in AcOEt (100/0 to 85/15, v/v) to give 1.55 g (69%) of 5-oxo-6,7,8,9-tetrahydro-5H-benzo[7]annulen-2-yl 2,2-dimethylpropanoate (B) as a colorless oil.
1H NMR (400 MHz, DMSO-d6, δ ppm): 7.65 (d, 1 H); 7.10-7.04 (m, 2H); 2.95 (t, 2H); 2.68 (t, 2H); 1 .85-1 .65 (m, 4H).
LC/MS (m/z, MH+): 261
Intermediate (C). 9-(trifluoromethanesulfonyloxy)-6,7-dihydro-5H-benzo[7]annulen-3-yl 2,2-dimethylpropanoate
To a solution of 5-oxo-6,7,8,9-tetrahydro-5H-benzo[7]annulen-2-yl 2,2-dimethylpropanoate (B) (15 g, 57.62 mmol) in DCM (500 ml) was added dropwise under argon pyridine (7.28 ml, 86.43 mmol) and trifluoromethanesulfonic anhydride (19.58 ml, 1 15.24 mmol). The reaction mixture was stirred at room temperature for 2 hours and ice (200 g) was added. The phases were separated, the aqueous phase was washed with DCM and the gathered organic phases were dried over MgS04, filtered and evaporated under reduced pressure to give 22 g (97%) of 9-(trifluoromethanesulfonyloxy)-6,7-dihydro-5H-benzo[7]annulen-3-yl 2,2-dimethylpropanoate (C) as a white solid.
LC/MS (m/z, MH-): 391
Intermediate (D). 9-(4-{[(3S)-1 -(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dihydro-5H-benzo[7]annulen-3-yl-2,2-dimeth lpropanoate
To a solution of 9-(trifluoromethanesulfonyloxy)-6,7-dihydro-5H-benzo[7]annulen-3-yl-2,2-dimethylpropanoate (C) (22 g, 56.07 mmol) and (3S)-1 -(3-fluoropropyl)-3-[4-(tetramethyl-1 ,3,2-dioxaborolan-2-yl)phenoxy]pyrrolidine (e) (20.56 g, 58.87 mmol) in dioxane (420 ml) and water (120 ml) was added under argon Pd(dppf)CI2 (2.75 g, 3.36 mmol) and Cs2C03 (36.57 g, 1 12.13 mmol). The reaction mixture was stirred for 1 hour at room temperature and was partitioned between water and DCM. The aqueous phase was washed with DCM and the gathered organic phases dried over MgS04, filtered and concentrated under reduced pressure. The residue was purified by column chromatography eluting with a gradient of MeOH in DCM (0 to 5%; V/V) to give 31 g (100 %) of 9-(4-{[(3S)-1 -(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dihydro-5H-benzo[7]annulen-3-yl-2,2-dimethylpropanoate (D).
LC/MS (m/z, MH+): 466
Intermediate (E). 9-(4-{[(3S)-1 -(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dihydro-5H-benzo[7]annulen-3-ol
To a solution under argon of 9-(4-{[(3S)-1 -(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dihydro-5H-benzo[7]annulen-3-yl-2,2-dimethylpropanoate (D) (24.8 g, 53.26 mmol) in MeOH (300 ml), was added NaOH 5M (23 ml, 1 15.00 mmol). The reaction mixture was stirred for 2 hours at room temperature. pH was then adjusted to 7 by addition of 6N aqueous HCI solution. The MeOH was concentrated under reduced pressure, then DCM was added. The organic phase was dried over MgS04, and concentrated under reduced pressure. The residue was purified by flash chromatography eluting with a gradient of DCM/ MeOH from 100/0 to 95/05 to give 18.8 g (93%) of 9-(4-{[(3S)-1 -(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dihydro-5H-benzo[7]annulen-3-ol (E) as a beige solid.
LC/MS (m/z, MH+): 382
Intermediate (F). 9-(4-{[(3S)-1 -(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dihydro-5H-benzo[7]annulen-3-yl trifluoromethanesulfonate
To a solution of 9-(4-{[(3S)-1 -(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dihydro-5H-benzo[7]annulen-3-ol (E) (20.6 g, 54.00 mmol) in DCM (200 ml) and pyridine (6.55 ml, 81 .00 mmol), cooled to 5°C (ice bath), was added dropwise trifluoromethanesulfonic anhydride (18.93 ml, 108.00 mmol) under argon, and the reaction temperature was maintained <15°C. The ice bath was removed, and the brown suspension was stirred at room temperature for 2 hours. Ice (200 g) and DCM (200 ml) were added and the phases separated. The organic phase was dried over MgS04, and concentrated under reduced pressure. The residue was
purified by flash chromatography eluting with a gradient of DCM/MeOH from 100/0 to 95/05 to give 24.7 g (89.1 %) of 9-(4-{[(3S)-1 -(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dihydro-5H-benzo[7]annulen-3-yl trifluoromethanesulfonate (F) as a brown oil.
LC/MS (m/z, MH+): 514
Intermediate (G). Methyl 9-(4-{[(3S)-1 -(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dihydro-5H-benzo[7]annulene-3-carboxylate
To a solution of 9-(4-{[(3S)-1 -(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dihydro-5H-benzo[7]annulen-3-yl trifluoromethanesulfonate (F) (10.1 g, 19.67 mmol) in DMF (66 ml) and MeOH (33 ml), were added Pd(dppf)CI2 (909 mg, 1.18 mmol) and diisopropylethylamine (7.21 ml). The black suspension was carbonylated in an autoclave at 70°C under 5 bars of CO for 5 hours. The reaction mixture was filtered, then the filtrate was partially concentrated under reduced pressure. The residue was partitioned between AcOEt and water. The organic phase was washed with water (2x 100 ml), dried over MgS04, and concentrated under reduced pressure. The residue was purified by flash chromatography eluting with a gradient of DCIW MeOH from 100/0 to 95/05 to give 7.13 g (86%) of methyl 9-(4-{[(3S)-1 -(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dihydro-5H-benzo[7]annulene-3-carboxylate (G) as a brown gum.
LC/MS (m/z, MH+): 424
Intermediate (A1 ). 5-oxo-6,7,8,9-tetrahydro-5H-benzo[7]annulen-2-yltrifluoromethanesulfonate
To a solution of commercially available 2-hydroxy-6,7,8,9-tetrahydro-5H-benzo[7]annulen-5-one (A) (18.5 g, 105 mmol) in DCM (185 ml) and lutidine (13.35 ml, 1 13.505 mmol), cooled at 5°C under argon, was added dropwise trifluoromethanesulfonic anhydride (20.22 ml,
123.29 mmol) while keeping temperature between 10 and 20°C. The reaction mixture was stirred for 1 hour at 5°C then at room temperature for 1 hour.
Then, ice (200 g) was added and the slurry partitioned between water and DCM. The organic phase was washed with aqueous NaHC03 solution, dried over MgS04, filtered off and concentrated under reduced pressure. The residue was purified by flash chromatography eluting with a gradient of heptane/AcOEt from 100 to 90/10 to give 28.2 g (87%) of 5-oxo-6,7,8,9-tetrahydro-5H-benzo[7]annulen-2-yl trifluoromethanesulfonate (A1 ) as an orange oil. LC/MS (m/z, MH+): 309
Intermediate (B1 ). Methyl 5-oxo-6,7,8,9-tetrahydro-5H-benzo[7]annulene-2-carboxylate
To a solution of 5-oxo-6,7,8,9-tetrahydro-5H-benzo[7]annulen-2-yl trifluoromethanesulfonate (A1 ) (5.03 g, 16.32 mmol) in DMF (24 ml) and MeOH (12 ml), were added Pd(dppf)CI2 (754 mg, 0.98 mmol) and diisopropylethylamine (6 ml). The black suspension was carbonylated in an autoclave at 70°C under 5 bars of CO for 2.5 hours. The reaction mixture was filtered, then the filtrate was partially concentrated under reduced pressure, and the residue, was partitioned between AcOEt and water. The organic phase was washed with water (2x 75 ml) and aqueous HCI 0.5 N, dried over MgS04 and concentrated under reduced pressure. The residue was purified by flash chromatography eluting with a gradient of heptane/AcOEt from 100/0 to 90/10 to give 3.4 g (95%) of methyl 5-oxo-6,7,8,9-tetrahydro-5H-benzo[7]annulene-2-carboxylate (B1 ) as a colorless oil.
LC/MS (m/z, MH+): 219
Intermediate (C1 ). Methyl 9-(trifluoromethanesulfonyloxy)-6,7-dihydro-5H-benzo[7]annulene-3-carboxylate
To a solution of methyl 5-oxo-6,7,8,9-tetrahydro-5H-benzo[7]annulene-2-carboxylate (B1 ) (18,19 g, 83,34 mmol) in DCM (500 ml) and anhydrous pyridine (1 1 ml, 130,56 mmol), cooled at 5°C under argon, was added dropwise trifluoromethanesulfonic anhydride (30 ml, 176,54 mmol). The reaction mixture, a thick suspension, was stirred at room temperature for 24 hours, then ice was added and partitioned between water and DCM. The organic phase was dried over MgS04, filtered off and concentrated under reduced pressure to give 29 g (100%) of methyl 9-(trifluoromethanesulfonyloxy)-6,7-dihydro-5H-benzo[7]annulene-3-carboxylate (C1 ) as a yellow gum.
LC/MS (m/z, MH+): 351
Intermediate (G). Methyl 9-(4-{[(3S)-1 -(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dihydro-5H-benzo[7]annulene-3-carboxylate
To a solution of methyl 9-(trifluoromethanesulfonyloxy)-6,7-dihydro-5H-benzo[7]annulene-3-carboxylate (C1 ) (29 g, 82.9 mmol), (3S)-1 -(3-fluoropropyl)-3-[4-(tetramethyl-1 ,3,2-dioxaborolan-2-yl)phenoxy]pyrrolidine (e) (28.9 g, 82.9 mmol), in dioxane (225 ml) were added Pd(dppf)CI2 under argon, complex with DCM (3.73 g, 4.57 mmol) and Cs2C03 1 .5 M aqueous solution (1 1 1.12 ml, 166.68 mmol). The reaction mixture was stirred at 60°C for 1 hour.
After cooling to room temperature, the reaction mixture was poured into a mixture of water (500 ml) and AcOEt (400ml). The organic phase was washed with brine, dried over MgS04, filtered on celite and concentrated under reduced pressure. The residue was purified by flash chromatography eluting with a gradient of DCM/MeOH from 100/0 to 95/05 to give 23 g (65%) of methyl 9-(4-{[(3S)-1 -(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dihydro-5H-benzo[7]annulene-3-carboxylate (G) as a brown gum.
LC/MS (m/z, MH+): 424
Intermediate (H). Methyl 8-bromo-9-(4-{[(3S)-1 -(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dihydro-5H-benzo[7]annulene-3-carboxylate hydrobromide
To a solution of methyl 9-(4-{[(3S)-1 -(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dihydro- 5H-benzo[7]annulene-3-carboxylate (G) (13.93 g, 32.89 mmol), in DCM (150 ml) was added under argon pyridinium tribromide (15.78 g, 44.41 mmol). The reaction mixture was stirred for 1 hour at room temperature. Water (200 ml) was added, organic phase was then dried over MgS04, and concentrated under reduced pressure. The residue was purified by flash chromatography eluting with a gradient of DCM/MeOH from 100/0 to 95/05 to give 16.4 g (85%) of methyl 8-bromo-9-(4-{[(3S)-1 -(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7-dihydro- 5H-benzo[7]annulene-3-carboxylate hydrobromide (H) as a yellow meringue.
LC/MS (m/z, MH+): 502
Intermediate (I). 6-(2,4-dichloro-phenyl)-5-{4-[1 -(3-fluoro-propyl)-pyrrolidin-3-yloxy]-phenyl}- -dihydro-7H-benzocycloheptene-2-arboxylic acid methyl ester.
To a solution of methyl 8-bromo-9-(4-{[(3S)-1 -(3-fluoropropyl)pyrrolidin-3-yl]oxy}phenyl)-6,7- dihydro-5H-benzo[7]annulene-3-carboxylate hydrobromide (H) (150 mg, 298.56 μηηοΙ), in dioxane (12 ml) and water (2 ml), was added 2,4-dichlorophenyl-boronic acid (62.67 mg, 328.41 μηιοΙ), Cs2C03 (204.48 mg, 626.97 μπιοΙ), and Pd(dppf)CI2 (14.63 mg, 17.91 mol). The reaction mixture was heated at 90°C for 3 hours, and partitioned between AcOEt and water. The phases were separated and the organic phase washed with brine, dried over MgS04 and concentrated under reduced pressure. The residue was purified by column
chromatography eluting with a mixture of DCM, acetonitrile and MeOH (96/2/2; V/V/V) to give 80 mg (47%) of 6-(2,4-dichloro-phenyl)-5-{4-[1 -(3-fluoro-propyl)-pyrrolidin-3-yloxy]-phenyl}-8,9-dihydro-7H-benzocycloheptene-2-arboxylic acid methyl ester (I).
LC/MS (m/z, MH+): 568
Compound (1 ). 6-(2,4-dichlorophenyl)-5-[4-[(3S)-1 -(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulen -2-carboxylic acid
To a solution of 6-(2,4-dichloro-phenyl)-5-{4-[1 -(3-fluoro-propyl)-pyrrolidin-3-yloxy]-phenyl}-8,9-dihydro-7H-benzocycloheptene-2-arboxylic acid methyl ester (I) (80 mg, 140.72 μηηοΙ) in MeOH (5 ml) was added a solution of NaOH (562.88 μΙ, 5 M) and the reaction mixture was heated at 60°C for 5 hours and the solvent removed under reduced pressure. The residue was taken up in water (10 ml) and aqueous HCI (5 M) added to pH 7. The slurry was extracted with DCM, dried over MgS04 and concentrated under reduced pressure. The solid was purified by column chromatography eluting with a mixture of DCM, acetonitrile and MeOH (90/5/5; V/V/V) to give 60 mg (77%) of 6-(2,4-dichlorophenyl)-5-[4-[(3S)-1 -(3-fluoropropyl)pyrrolidin-3-yl]oxyphenyl]-8,9-dihydro-7H-benzo[7]annulene-2-carboxylic acid. 1H NMR (400 MHz, DMSO-d6, δ ppm): 1 .68 (m, 1 H); 1 ,79 (dm, J=25.3 Hz, 2 H); 2.07 to 2.23 (m, 5 H); 2.38 (m, 1 H); 2.46 (t, J=7.2 Hz, 2 H); 2.52 (m, 1 H); 2.62 (m, 1 H); 2.55 to 2.89 (m, 3 H); 4.47 (td, J=6.2 and 47.6 Hz, 2 H); 4.72 (m, 1 H); 6.63 (d, J=8.9 Hz, 2 H); 6.71 (m, 3 H); 7.18 (d, J=8.4 Hz, 1 H); 8.26 (dd, J=2.0 and 8.4 Hz, 1 H); 7.58 (d, J=2,0 Hz, 1 H); 7.63 (d, J=8.4 Hz, 1 H); 7.79 (s, 1 H); 12.3 (m, 1 H)
LC/MS (m/z, MH+): 554
//////////////
Admin note for myself . I am up for Grabs
I myself Dr Anthony Melvin Crasto Looking for a post retirement assignment as Advisor API & INT, Chem.
With 36 yrs rich experience, about dozen patents, 10000plus steps covered, 200 API targets, 30 plus products commercialization in plant in full career. Hands on knowledge of Synthesis, Process, scaleup, cost reduction, DOE , softwares etc
Kindly contact me
Dr Anthony Melvin Crasto
+919321316780
amcrasto@gmail.com
About myself
Dr Anthony Crasto
click on my website to know about me
Read http://amcrasto.weebly.com/
Also http://amcrasto.weebly.com/awards.html
Also
http://amcrasto.weebly.com/felicitations.html
1000 lakh google hits, 100lakh blog views, 10 lakh viewers in USA alone, all in 7 continents, 226 countries, 30 Indian and International awards, helping millions across the world

////////

AS ON DEC2021 3,491,869 VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@amcrasto
/////////////////////////////////////////////////////////////////////////////
/////Amcenestrant, SAR439859, アムセネストラント , Antineoplastic, CANCER
C1CC2=C(C=CC(=C2)C(=O)O)C(=C(C1)C3=C(C=C(C=C3)Cl)Cl)C4=CC=C(C=C4)OC5CCN(C5)CCCF
Myself Anthony Crasto up for grabs as Advisor API & INT, Chem.
Admin note for myself . I am up for Grabs
I myself Dr Anthony Melvin Crasto Looking for a post retirement assignment as Advisor API & INT, Chem.
With 36 yrs rich experience, about dozen patents, 10000plus steps covered, 200 API targets, 30 plus products commercialization in plant in full career. Hands on knowledge of Synthesis, Process, scaleup, cost reduction, DOE , softwares etc
Kindly contact me
Dr Anthony Melvin Crasto
+919321316780
amcrasto@gmail.com
About myself
Dr Anthony Crasto
click on my website to know about me
Read http://amcrasto.weebly.com/
Also http://amcrasto.weebly.com/awards.html
Also
http://amcrasto.weebly.com/felicitations.html
1000 lakh google hits, 100lakh blog views, 10 lakh viewers in USA alone, all in 7 continents, 226 countries, 30 Indian and International awards, helping millions across the world

‘Top 10 Prominent & Great Personalities of the year 2022’ campaign by Fame Finders. ANTHONY CRASTO


‘Top 10 Prominent & Great Personalities of the year 2022’ campaign by Fame Finders.
Feeling great to be selected in the ‘Top 10 Prominent & Great Personalities of the year 2022’ campaign by Fame Finders. It is an online campaign to honour inspiring personalities and feature them in the upcoming edition of the top news sites, including – Deccan Chronicle/Asian Age/Deccan Herald, ANI, Zee5, Latestly, Lokmat Times, DailyHunt, Google News, JioNews, MSN and 70+ sites.Jan 16 2023

[youtube https://www.youtube.com/watch?v=x98E01m9anw&w=560&h=315]

SC-52458, FORASARTAN

SC-52458, FORASARTAN
- Molecular FormulaC23H28N8
- Average mass416.522 Da
PHASE 2, PFIZER, HYPERTENSION
145216-43-9[RN]
5-[(3,5-Dibutyl-1H-1,2,4-triazol-1-yl)methyl]-2-[2-(1H-tetrazol-5-yl)phenyl]pyridine
5-[(3,5-dibutyl-1H-1,2,4-triazol-1-yl)methyl]-2-[2-(2H-1,2,3,4-tetrazol-5-yl)phenyl]pyridine
форасартан[Russian][INN], فوراسارتان[Arabic][INN], 福拉沙坦[Chinese][INN]
065F7WPT0B[DBID]
7415[DBID]
UNII-065F7WPT0B[DBID]
SC 52458[DBID]
Type-1 angiotensin II receptor
Forasartan, otherwise known as the compound SC-52458, is a nonpeptide angiotensin II receptor antagonist (ARB, AT1 receptor blocker).[2][3][4][5]
Forasartan, a specific angiotensin II antagonist, is used alone or with other antihypertensive agents to treat hypertension. Forasartan competes with angiotensin II for binding at the AT1 receptor subtype. As angiotensin II is a vasoconstrictor which also stimulates the synthesis and release of aldosterone, blockage of its effects results in a decreases in systemic vascular resistance.
Indications
Forasartan is indicated for the treatment of hypertension[6] and, similar to other ARBs, it protects the kidneys from kidney blood vessel damage caused by increased kidney blood pressure by blocking renin–angiotensin system activation.[7]
Administration
Forasartan is administered in the active oral form [6] which means that it must go through first pass metabolism in the liver. The dose administered ranges between 150 mg-200 mg daily.[6] Increasing to more than 200 mg daily does not offer significantly greater AT1 receptor inhibition.[6] Forasartan is absorbed quickly in the GI, and within an hour it becomes significantly biologically active.[6] Peak plasma concentrations of the drug are reached within one hour.[6]
Contraindications
Negative side effects of Forasartan are similar to other ARBs, and include hypotension and hyperkalemia.[8] There are no drug interactions identified with forasartan.[6]
Bioorganic & Medicinal Chemistry Letters (1994), 4(1), 99-104
PATENT
EP508445
https://worldwide.espacenet.com/patent/search/family/024755845/publication/EP0508445A1?q=EP508445A1
PATENT
WO1992018092
Example 2
2-[5-[(3,5-dibutyl-1H-1,2,4-triazol-1-yI)methyl]- 2-pyridinyl]benzoic acid
Step 1 : Preparation of 2-bromo-5-picoline .
A solution of 1500 mL (14 mol) of 48%
hydrobromic acid was cooled to 10 °C and 300 g (2.8 mol) of 2-amino-5-picoline (Aldrich) was added slowly. The
solution was maintained at or below 0 °C while 450 mL (8.8 mol) of bromime was added dropwise. After the bromine addition was complete, a solution of 500 g (7.3 mol) of sodium nitrite in 1000 mL of water was added slowly over 6 h. The reaction pH was adjusted by the careful addition of 1500 mL (56 mol) of 50% sodium hydroxide at such a rate that the temperature was maintained below 30 °C. The product precipitated from the nearly colorless reaction mixture; filtration gave 450 g (94%) of 2-bromo-5-picoline as a yellow powder: mp 38-40 °C; NMR 7.27 (s, 1H), 7.28 (s, 1H), 7.12 (br s, 1H).
Step 2 : Preparation of N-methyl-N-tertbutylbenzamide.
Under nitrogen, 96.7 g (1.1 mol) of N-methyl-N-tertbutylamine and 111 g (1.1 mol) of triethylamine was dissolved in 1050 mL of anhydrous tetrahydrofuran (THF).
The solution was cooled to 0 °C and treated with 140.6 σ (1.0 mol) of benzoyl chloride. The reaction was allowed to slowly warm to ambient temperature and stir overnight.
Filtration and subsequent concentration in vacuo of the filtrate gave the crude product which was purified by sublimation (65 °, 0.2 torr) to give 184 g (96%) of
colorless N-methyl-N-tertbutybenzamide: mp 80.5-82.0 °C; NMR (CDCI3) δ1.52 (s, 9H), 2.87 (s, 3H), 7.34-7.40 (m, 3H), 7.40-7.46 (m, 2H).
Step 3 : Preparation of 2-(N-methyl-N-tertbutylcarboxamido)phenyIboronic acid.
Under nitrogen, a solution of 50.0 g (262 mmol) of N-methyl-N-tertbutylbenzamide from step 2 and 44 ml (2S2 mmol) of tetramethylethylenediamine (TMEDA) in 3350 mL of anhydrous THF was cooled to -78 °C and slowly treated with 262 mmol of sec-butyllithium in cyclohexane. After 1 h at -78 °C, the reaction was treated with 45 mL (393 mmol) of trimethyl borate and allowed to slowly warm to ambient temperature overnight with stirring. The reaction was concentrated in vacuo; the residue was dissolved in IK sodium hydroxide and extracted with methylene chloride. The pH of the aqueous phase was adjusted to six with dilute hydrochloric acid and extracted with methylene chloride; the organic layer was dried (MgSO4) and concentrated in vacuo to give 55.7 g (90%) of a 80:20 mixture of syn/anti isomers of 2-(N-methyl-N-tertbutylcarboxamido)phenyIboronic acid as a pale yellow glass: NMR (CDCI3) δ 1.30 (s, syn C(CH3)3, 7.3H), 1.54 (s, anti 0(0.3)3, 1.7H), 2.81 (s, anti CH3, 0.6H), 2.94 (s, syn CH3, 2.4H), 7.29-7.46 (m, 3H), 7.95-8.01 (m, 1H).
step 4 : Preparation of N-methyl-N-tertbwtyl-2-(5-methyl-2-pyridinyl)benzamide.
Under nitrogen, 4.44 g (25.8 mmcl) cf 2-bromo-5-picoline from step 1 in 60 mL of toluene was treated with 6.75 g (29 mmol) of 2- (N-methyl-N- tertbutylcarboxamido)phenyIboronic acid from step 3, 1.0 g of tetrakis (triphenylphosphine)palladium zero, 26 mL of ethanol, and 29 mL of 2M sodium carbonate; this mixture was heated to reflux and vigorously stirred for 24 h. The reaction was partitioned between water and ether; the organic layer was separated, dried (MgSθ4), and
concentrated in vacuo. Purification by silica gel
chromatography (Waters Prep-500A) using ethyl
acetate/hexane (1:2) gave 6.51 g (90%) of N-methyl-N- tertbutyl-2-(5-methyl-2-pyridinyl)benzamide as an oil : NMR (CDCI3) δ 1.40 (s, 9H), 2.33 (s, 3H), 2.61 (s, 3H), 7.27- 7.33 (m, 1H), 7.35-7.41 (m, 2H), 7.47-7.51 (m, 2H), 7.60- 7.66 (m, 1H), 8.43 (br s, 1H).
Step 5 : Preparation of sodium 2-(5-methyl-2- pyridinyl)benzoate.
Under nitrogen, 6.5 g (23 mmol) of N-methyl-N- tertbutyl-2-(6-methyl-3-pyridinyl)benzamide from step 4 was treated with 65 mL of anhydrous trifluoroacetic acid (TFA) at reflux for 6 h. The reaction was concentrated in vacuo and the residue dissolved in water. The pH was adjusted to 10 with aqueous sodium hydroxide and lyophilized to give the sodium salt of 2- (5-methyl-2-pyridinyl)benzoic acid as a colorless solid: NMR [CDCI3/CF3CO2H (97:3)] δ 2.62 (s, 3H), 7.42-7.48 (m, 1H), 7.67-7.80 (m, 3H), 8.18-8.24 (m, 1H), 8.28 (dd, J=8 and 2 HZ, 1H), 7.67-7.80 (m, 3H), 8.18-8.24 (m, 1H), 8.28 (dd, J=8 and 2 Hz, 1H), 8.61 (s, 1H) ; MS (FAB) m/e (rel intensity) 214 (20), 196 (100); HRMS.
Calc’d for M+H: 214.0868. Found: 214.0846.
step 6 : Preparation of ethyl 2-(5-methyl-2-pyridinyl)benzoate.
Under nitrogen, the crude sodium salt from step 5 was suspended in 50 mL of chloroform and treated with 9 mL (103 mmol) of oxalyl chloride. The reaction was stirred for 72 h, filtered under nitrogen, and concentrated in vacuo; the residue was dissolved in absolute ethanol.
Concentration in vacuo gave 2.0 g (8 mmol) of ethyl 2-(5-methyl-2-pyridinyl)benzoate as a brown oil: NMR (CDCI3) δ 1.09 (t, J=7 Hz, 3H), 2.36 (s, 3H), 4.15 (q, J=7 Hz, 2H), 7.34 (d, J=8 Hz, 1H), 7.38-7.48 (m, 1H), 7.48-7.58 (m, 3H), 7.80 (d, J=8 Hz, 1H), 8.46 (s, 1H).
Step 7 : Preparation of ethyl 2-(5-bromomethyl-2-pyridinyl)benzoate.
Under nitrogen, the crude ethyl 2-(5-methyl-2-pyridinyl)benzoate from step 6 was treated with 1.7 g (9.5 mmol) of NBS and 160 mg (0.66 mmol) of benzoyl peroxide in 145 mL of anhydrous carbon tetrachloride at reflux for 2.5 h. The reaction was filtered under nitrogen and
concentrated in vacuo to give crude ethyl 2-(5-bromomethyl-2-pyridinyl)benzoate; no purification was attempted.
step 8 : Preparation of ethyl 2-[5-[(3,5-dibutyl-1H- 1 , 2 , 4-triazol-1 -yl )methy] 1 -2-pyridinyl ] benzoate .
Under nitrogen, 630 mg (3.5 mmol) of 3,5-dibutyl-1H-1,2,4-triazole from step 3 of Example 1 was added in small portions to 5.4 mmol of sodium hydride in 8 mL of DMF; stirring was continued until hydrogen evolution had ceased. The anion solution was cooled to 0 °C and treated with a solution of the crude ethyl 2-(5-bromomethyl-2-pyridinyl)benzoate from step 7 in 10 mL of DMF. The reaction was stirred at ambient temperature overnight, quench with 1 mL of absolute ethanol, and concentrated in vacuo; the resulting residue was redisolved in methylene chloride, filtered, and reconcentrated in vacuo to give crude ethyl 2-[5-[(3,5-dibutyl-1H-1,2,4-triazol-1-yl)methyl]-2-pyridinyl]benzoate.
step 9 : Preparation of 2- [5- [ (3, 5-dibutyl-1H-1 , 2, 4 -triazol-1-yl)methyl]-2-pyridinyllbenzoic acid.
A 1.0 g sample of the crude ethyl 2-[5-[(3,5-dibutyl-1H-1,2,4-triazol-1-yl)methyl]-2-pyridinyl]benzoate from step 8 in 10 mL of water was treated with 3 mL of 101 aqueous sodium hydroxide and stirred at ambient temperature overnight. The reaction mixture was washed with 30 mL of ether and the pH adjusted to six with dilute hydrochloric acid. Purification by reverse phase chromatography (Waters Deltaprep-3000) using isocratic acetonitrile/water (28:72) (0.05% TFA) gave 5 mg of 2-[5-[(3,5-dibutyl-1H-1,2,4-triazol-1-yl)methyl]-2-pyridinyl]benzoic acid: NMR (D2O + NaO3S(CH2)3 Si(CH3)3] δ 0.80 (t, J=7 Hz, 3H), 0.86 (t, J=7 Hz, 3H), 1.19-1.33 (m, 4H), 1.54-1.68 (m, 4H), 2.65 (t, J=7 Hz, 2H), 2.82 (t, _ϊ=7 Hz, 2H), 5.43 (s, 2H), 7.45-7.59 (m, 5H), 7.64 (dd, J=8 and 2 Hz, 1H), 8.37-8.45 (m, 1H); MS (FAB) m/e (rel intensity) 393 (80), 375 (30), 212 (40), 182 (100); HRMS. Calc’d for M+Li: 399.2373. Found:
399.2374.
Example 3
5-[2-[5-[(3,5-dibutyl-1H-1,2,4-triazol-1-yl)methyl]- 2-pyridinyl]phenyl]-1H-tetrazole
Step 1 : Preparation of 2-bromo-5-bromomethylpyridine.
A solution of 296.3 g (1.72 mol) of 2-bromo-5-picoline from step 1 of Example 2 in 6000 mL of carbon tetrachloride was treated with 306.5 g (1.72 mol) of N-bromosuccinimide (NBS) and 28.3 g (173 mmol) of
azobisisobutyronitrile (AIBN). The reaction was stirred at reflux under nitrogen for 3 h, filtered, and concentrated in vacuo providing 476 g of crude 2-bromo-5-bromomethylpyridine as a brownish yellow solid (NMR indicates that this material is only 60% monobromomethyl product): NMR (CDCI3 δ 4.42 (s, 2H), 7.48 (d, .J=9 Hz, 1H), 7.60 (dd, J=9 and 3 Hz, 1H), 8.37 (d, J=3 Hz, 1H).
Step 2: Preparation of 2-bromo-5-[(3.5-dibutyl-1H-1,2,4-triazol-1-yl)methyl]pyridine.
Under nitrogen, 3.15 g (17 mmol) of solid 3,5-dibutyl-1H-1,2,4-triazole from step 3 of Example 1 was added in small portions to 33 mmol of sodium hydride in 31 ml of dimethylformamide (DMF); stirring was continued until hydrogen evolution had ceased. The anion solution was cooled to 0 °C and treated with a solution of 7.9 g (19 mmol) of crude 2-bromo-5-bromomethylpyridine from step 1 in 10 ml of dry DMF. The reaction was allowed to warm to ambient temperature and stir overnight. Methanol (10 ml) was added to destroy any unreacted sodium hydride and the
DMF was removed in vacuo. The residue was dissolved in ethyl acetate, washed with water, and dried (MgSO4).
Silica gel chromatography (Waters Prep-500A) using ethyl acetate/hexane (60:40) gave 4.8 g (47%) of 2-bromo-5-[(3,5- dibutyl-1H-1,2,4-triazol-1-yl)methyl]pyridine as an oil: NMR (CDCI3) δ 0.88 (t, J=7 Hz, 1H), 0.92 (t, J=7 Hz, 1H), 1.27-1.44 (m, 4H), 1.59-1.76 (m, 4H), 2.60-2.71 (m, 4H), 5.18 (s, 2H), 7.35 (dd, J=8 and 3 Hz), 7.46 (d, J=8 Hz, 1H), 8.23 (d, .1=3 Hz, 1H).
Step 3: Preparation of 5-[2-[5-[(3,5-dibutyl-1H-1,2,4- triazol-1-yl)methyl]-2-pyridinyl]phenyl]-1H-tetrazole.
Under nitrogen, 1.03 g (2.9 mmol) of 2-bromo-5- [(3,5-dibutyl-1H-1,2,4-triazol-1-yl)methyl]pyridine from step 2 and 2.46 g (5.7 mmol) of 2-(N-triphenyImethyltetrazol-5-yl)phenyIboronic acid from step 5 of Example 1 were treated with 1.0 g (0.86 mmol) of tetrakis (triphenyl-phosphine)palladium zero, 15 mL of toluene, 10 mL of ethanol, and 6.3 mL of 2M aqueous sodium carbonate. The reaction mixture was heated to reflux and vigorously stirred overnight. The product was purified by reverse phase chromatography (Waters Deltaprep-3000) using acetonitrile/water (20-40:80-60) (0.05% TFA). The pure fractions (by analytical HPLC) were combined, the
acetonitrile removed in vacuo, the pH adjusted to four with dilute sodium hydroxide, and the resulting suspension extracted 4 times with ether. The extracts were combined, dried (MgSθ4), and concentrated in vacuo to give 340 mg (28%) of 5-[2-[5-[(3,5-dibutyl-1H-1,2,4-triazol-1-yl)methyl]-2-pyridinyl]phenyl-1H-tetrazole as a colorless solid: mp 139-141 °C; NMR (CD3OD) δ 0.90 (t, J=7 Hz, 3H), 0.93 (t, J=7 Hz, 3H), 1.29-1.44 (m, 4H), 1.58-1.75 (m, 4H), 2.65 (t, J=7 Hz, 2H), 2.81 (t, J=7 Hz, 2H), 5.40 (s, 2H), 7.47 (d, J=8 Hz, 1H), 7.61-7.77 (m, 5H), 8.33 (d, J=2 Hz, 1H); MS (FAB) m/e (rel intensity) 417 (100), 208 (30); HRMS. Calc’d for M+H: 417.2515. Found: 417.2527.
PATENT
WO2001076573
////////

AS ON DEC2021 3,491,869 VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@amcrasto
/////////////////////////////////////////////////////////////////////////////
Pharmacology
The angiotensin II receptor, type 1
Angiotensin II binds to AT1 receptors, increases contraction of vascular smooth muscle, and stimulates aldosterone resulting in sodium reabsorption and increase in blood volume.[9] Smooth muscle contraction occurs due to increased calcium influx through the L-type calcium channels in smooth muscle cells during the plateau component, increasing the intracellular calcium and membrane potential which sustain depolarization and contraction.[10]
Effects
Forasartan is a competitive and reversible ARB that competes with the angiotensin II binding site on AT1[11] and relaxes vascular smooth muscle,[10] resulting in decreased blood pressure. Forasartan has a high affinity for the AT1 receptor (IC50=2.9 +/- 0.1nM).[12] In dogs, it was found to block the pressor response of Angiotensin II with maximal inhibition, 91%.[10] Forasartan administration selectively inhibits L-type calcium channels in the plateau component of the smooth muscle cells, favoring relaxation of the smooth muscle.[10] Forasartan also decreases heart rate by inhibiting the positive chronotropic effect of high frequency preganglionic stimuli.[13]
Scarce use
Even though experiments have been conducted on rabbits,[6] guinea pigs,[10] dogs [14] and humans,[6][13] forasartan is not a popular drug of choice for hypertension due to its short duration of action; forasartan is less effective than losartan.[6] Research demonstrates that forasartan is also significantly less potent than losartan.[6]
See also
References
- ^ Bräse, Stefan; Banert, Klaus (2010). Organic Azides: Syntheses and Applications. New York: Wiley. p. 38. ISBN 978-0-470-51998-1.
- ^ Knox C, Law V, Jewison T, Liu P, Ly S, Frolkis A, et al. (January 2011). “DrugBank 3.0: a comprehensive resource for ‘omics’ research on drugs”. Nucleic Acids Research. DrugBank. 39 (Database issue): D1035-41. doi:10.1093/nar/gkq1126. PMC 3013709. PMID 21059682.
- ^ Wishart DS, Knox C, Guo AC, Cheng D, Shrivastava S, Tzur D, et al. (January 2008). “DrugBank: a knowledgebase for drugs, drug actions and drug targets”. Nucleic Acids Research. 36 (Database issue): D901-6. doi:10.1093/nar/gkm958. PMC 2238889. PMID 18048412.
- ^ Wishart DS, Knox C, Guo AC, Shrivastava S, Hassanali M, Stothard P, et al. (January 2006). “DrugBank: a comprehensive resource for in silico drug discovery and exploration”. Nucleic Acids Research. 34 (Database issue): D668-72. doi:10.1093/nar/gkj067. PMC 1347430. PMID 16381955.
- ^ Olins GM, Corpus VM, Chen ST, McMahon EG, Palomo MA, McGraw DE, et al. (October 1993). “Pharmacology of SC-52458, an orally active, nonpeptide angiotensin AT1 receptor antagonist”. Journal of Cardiovascular Pharmacology. 22 (4): 617–25. doi:10.1097/00005344-199310000-00016. PMID 7505365. S2CID 93468.
- ^ Jump up to:a b c d e f g h i j k Hagmann M, Nussberger J, Naudin RB, Burns TS, Karim A, Waeber B, Brunner HR (April 1997). “SC-52458, an orally active angiotensin II-receptor antagonist: inhibition of blood pressure response to angiotensin II challenges and pharmacokinetics in normal volunteers”. Journal of Cardiovascular Pharmacology. 29 (4): 444–50. doi:10.1097/00005344-199704000-00003. PMID 9156352.
- ^ Naik P, Murumkar P, Giridhar R, Yadav MR (December 2010). “Angiotensin II receptor type 1 (AT1) selective nonpeptidic antagonists–a perspective”. Bioorganic & Medicinal Chemistry. 18 (24): 8418–56. doi:10.1016/j.bmc.2010.10.043. PMID 21071232.
- ^ Ram CV (August 2008). “Angiotensin receptor blockers: current status and future prospects”. The American Journal of Medicine. 121 (8): 656–63. doi:10.1016/j.amjmed.2008.02.038. PMID 18691475.
- ^ Higuchi S, Ohtsu H, Suzuki H, Shirai H, Frank GD, Eguchi S (April 2007). “Angiotensin II signal transduction through the AT1 receptor: novel insights into mechanisms and pathophysiology”. Clinical Science. 112 (8): 417–28. doi:10.1042/cs20060342. PMID 17346243.
- ^ Jump up to:a b c d e Usune S, Furukawa T (October 1996). “Effects of SC-52458, a new nonpeptide angiotensin II receptor antagonist, on increase in cytoplasmic Ca2+ concentrations and contraction induced by angiotensin II and K(+)-depolarization in guinea-pig taenia coli”. General Pharmacology. 27 (7): 1179–85. doi:10.1016/s0306-3623(96)00058-4. PMID 8981065.
- ^ Olins GM, Chen ST, McMahon EG, Palomo MA, Reitz DB (January 1995). “Elucidation of the insurmountable nature of an angiotensin receptor antagonist, SC-54629”. Molecular Pharmacology. 47 (1): 115–20. PMID 7838120.
- ^ Csajka C, Buclin T, Fattinger K, Brunner HR, Biollaz J (2002). “Population pharmacokinetic-pharmacodynamic modelling of angiotensin receptor blockade in healthy volunteers”. Clinical Pharmacokinetics. 41 (2): 137–52. doi:10.2165/00003088-200241020-00005. PMID 11888333. S2CID 13185772.
- ^ Jump up to:a b Kushiku K, Yamada H, Shibata K, Tokunaga R, Katsuragi T, Furukawa T (January 2001). “Upregulation of immunoreactive angiotensin II release and angiotensinogen mRNA expression by high-frequency preganglionic stimulation at the canine cardiac sympathetic ganglia”. Circulation Research. 88 (1): 110–6. doi:10.1161/01.res.88.1.110. PMID 11139482.
- ^ McMahon EG, Yang PC, Babler MA, Suleymanov OD, Palomo MA, Olins GM, Cook CS (June 1997). “Effects of SC-52458, an angiotensin AT1 receptor antagonist, in the dog”. American Journal of Hypertension. 10 (6): 671–7. doi:10.1016/s0895-7061(96)00500-6. PMID 9194514.
| Clinical data | |
|---|---|
| Other names | SC-52458 |
| Pregnancy category | Not assigned |
| Routes of administration | Oral |
| ATC code | C09CA (WHO) |
| Legal status | |
| Legal status | Development halted, never marketed[1] |
| Pharmacokinetic data | |
| Elimination half-life | 1–2 hours |
| Identifiers | |
| showIUPAC name | |
| CAS Number | 145216-43-9 |
| PubChem CID | 132706 |
| DrugBank | DB01342 |
| ChemSpider | 117146 |
| UNII | 065F7WPT0B |
| KEGG | D04243 |
| ChEBI | CHEBI:141552 |
| ChEMBL | ChEMBL315021 |
| CompTox Dashboard (EPA) | DTXSID70162942 |
| Chemical and physical data | |
| Formula | C23H28N8 |
| Molar mass | 416.533 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| showSMILES | |
| showInChI | |
////////SC-52458, FORASARTAN, форасартан , فوراسارتان , 福拉沙坦 , PHASE 2, PFIZER, HYPERTENSION
CCCCC1=NN(CC2=CN=C(C=C2)C2=CC=CC=C2C2=NNN=N2)C(CCCC)=N1

NEW DRUG APPROVALS
one time
$10.00
Adagrasib
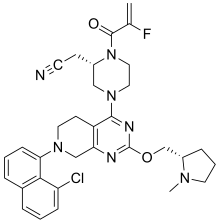
Adagrasib
| Formula | C32H35ClFN7O2 |
|---|---|
| cas | 2326521-71-3 |
| Mol weight | 604.1174 |
| Antineoplastic | |
| Disease | Non-small cell lung cancer |
|---|
| 2022/12/12 |
FDA APPROVED, KRAZATI (Mirati Therapeutics)
- MRTX-849
- MRTX849
- KRAS G12C inhibitor MRTX849
Adagrasib, sold under the brand name Krazati, is an anticancer medication used to treat non-small cell lung cancer.[1][2] Adagrasib is an inhibitor of the RAS GTPase family.[1] It is taken by mouth.[1] It is being developed by Mirati Therapeutics.[1][3]
The most common adverse reactions include diarrhea, nausea, fatigue, vomiting, musculoskeletal pain, hepatotoxicity, renal impairment, dyspnea, edema, decreased appetite, cough, pneumonia, dizziness, constipation, abdominal pain, and QTc interval prolongation.[2] The most common laboratory abnormalities include decreased lymphocytes, increased aspartate aminotransferase, decreased sodium, decreased hemoglobin, increased creatinine, decreased albumin, increased alanine aminotransferase, increased lipase, decreased platelets, decreased magnesium, and decreased potassium.[2]
It was approved for medical use in the United States in December 2022.[1][3]
Synthesis Reference
Fell, Jay B et al. “Identification of the Clinical Development Candidate MRTX849, a Covalent KRASG12C Inhibitor for the Treatment of Cancer.” Journal of medicinal chemistry vol. 63,13 (2020): 6679-6693. doi:10.1021/acs.jmedchem.9b02052
Journal of Medicinal Chemistry (2020), 63(13), 6679-6693
PATENT
WO2020101736 https://patents.google.com/patent/WO2020101736A1/en
EXAMPLE 7

2-[(2S)-4-[7-(8-chloro-1-naphthyl)-2-[[(2S)-1-methylpyrrolidin-2-yl]methoxy]-6,8-dihydro-5H- pyrido[3,4-d]pyrimidin-4-yl]-1-(2-fluoroprop-2-enoyl)piperazin-2-yl]acetonitrile
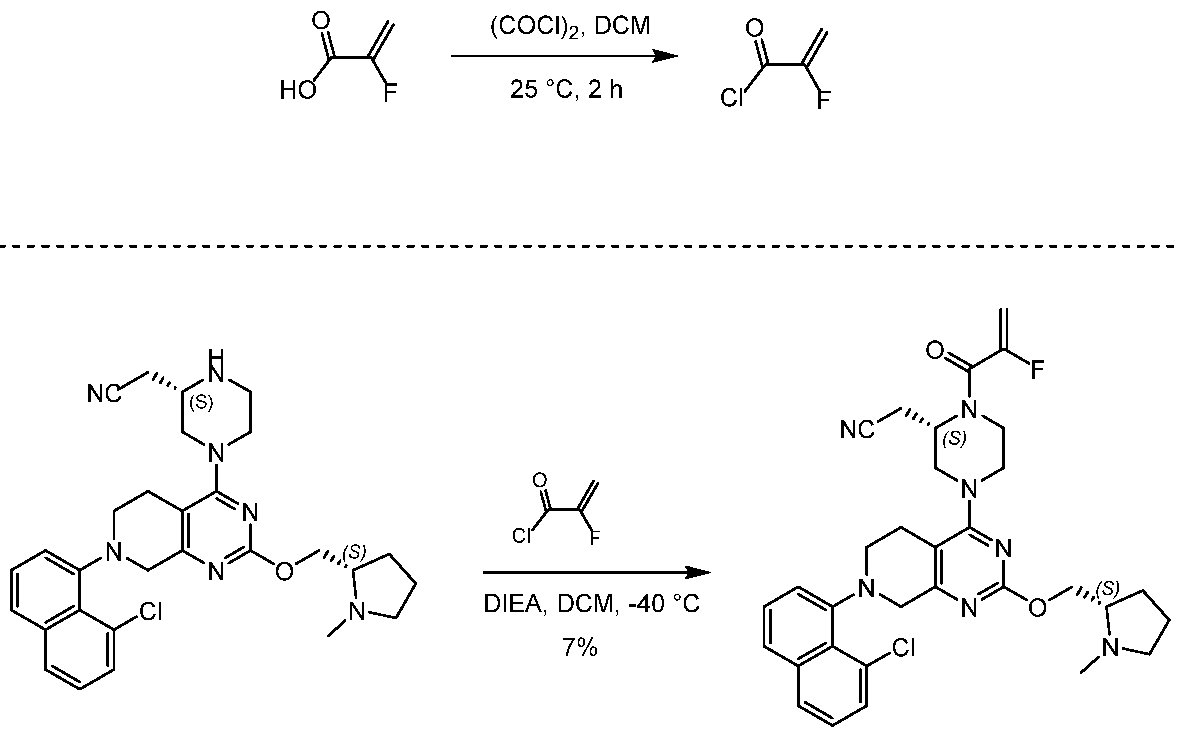
[0432] 2-fluoroprop-2-enoyl chloride. To a solution of 2-fluoroprop-2-enoic acid (400 mg, 4.44 mmol, 1 eq) in DCM (4 mL) was added (COCl)2 (846 mg, 6.66 mmol, 583 µL, 1.5 eq) and DMF (32.5 mg, 444 umol, 34.2 µL, 0.1 eq). The mixture was stirred at 25 °C for 2 hrs. The reaction mixture was concentrated under reduced pressure to remove a part of solvent and give a residue in DCM. Compound 2-fluoroprop-2-enoyl chloride (400 mg, crude) was obtained as a yellow liquid and used into the next step without further purification. [0433] Step A: 2-[(2S)-4-[7-(8-chloro-1-naphthyl)-2-[[(2S)-1- methylpyrrolidin-2-yl]methoxy]- 6,8-dihydro-5H-pyrido[3,4-d]pyrimidin-4-yl]-1-(2-fluoroprop-2-enoyl)piperazin-2- yl]acetonitrile. To a solution of 2-[(2S)-4-[7-(8-chloro-1-naphthyl)-2-[[(2S)- 1-methylpyrrolidin- 2-yl]methoxy]-6,8-dihydro-5H-pyrido[3,4-d]pyrimidin-4-yl]piperazin-2-yl]acetonitrile (300 mg, 528 umol, 1 eq, HCl) in DCM (5 mL) was added DIEA (1.73 g, 13.4 mmol, 2.33 mL, 25.4 eq) and 2-fluoroprop-2-enoyl chloride (286 mg, 2.64 mmol, 5 eq) in DCM (5 mL). The mixture was stirred at 0 °C for 1 hour. The reaction mixture was concentrated under reduced pressure to give a residue. The residue was purified by column chromatography (Al2O3, Dichloromethane/Methanol = 10/1 to 10/1). The residue was purified by prep-HPLC (column: Gemini 150 * 25 5u; mobile phase: [water (0.05% ammonia hydroxide v / v) – ACN]; B%: 55% – 85%, 12min). The residue was purified by prep-HPLC (column: Phenomenex Synergi C18 150 * 30mm * 4um; mobile phase: [water (0.225% FA) – ACN]; B%: 20% – 50%, 10.5min). The residue was concentrated under reduced pressure to remove ACN, and then lyophlization. Title compound 2-[(2S)-4-[7-(8-chloro- 1-naphthyl)-2-[[(2S)-1- methylpyrrolidin-2-yl]methoxy]-6,8-dihydro-5H-pyrido[3,4-d]pyrimidin- 4-yl]-1-(2-fluoroprop-2-enoyl)piperazin-2-yl]acetonitrile (EXAMPLE 7, 24.1 mg, 36.7 umol, 7% yield, 99.1% purity, FA) was obtained as a brown solid. [0434] SFC condition: “AD – 3S_3_5_40_3ML Column: Chiralpak AD – 3 100 × 4.6mm I.D., 3um Mobile phase: methanol (0.05% DEA) in CO2 from 5% to 40% Flow rate: 3mL/min Wavelength: 220nm”. [0435] 1H NMR (400 MHz, Acetic) d = 7.82 (d, J = 8.0 Hz, 1H), 7.69 (d, J = 8.0 Hz, 1H), 7.56 (d, J = 7.6 Hz, 1H), 7.49 (t, J = 7.6 Hz, 1H), 7.41 – 7.30 (m, 2H), 5.58 – 5.25 (m, 2H), 5.17 – 4.59 (m, 4H), 4.57 – 4.28 (m, 3H), 4.24 – 3.78 (m, 4H), 3.67 – 3.13 (m, 7H), 3.08 (br d, J = 2.4 Hz, 3H), 2.98 (br d, J = 6.4 Hz, 1H), 2.83 – 2.61 (m, 1H), 2.45 – 2.29 (m, 1H), 2.24 – 2.08 (m, 3H).
PATENT
US20190144444 https://patents.google.com/patent/US20190144444A1/en
////////

AS ON DEC2021 3,491,869 VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@amcrasto
/////////////////////////////////////////////////////////////////////////////
Adagrasib (MRTX849) is an oral, small-molecule KRAS inhibitor developed by Mirati Therapeutics. KRAS mutations are highly common in cancer and account for approximately 85% of all RAS family mutations.5 However, the development of KRAS inhibitors has been challenging due to their high affinity for guanosine triphosphate (GTP) and guanosine diphosphate (GDP), as well as the lack of a clear binding pocket.1 Adagrasib targets KRASG12C, one of the most common KRAS mutations, at the cysteine 12 residue and inhibits KRAS-dependent signalling.2 In a phase I/IB clinical study that included patients with KRASG12C-mutated advanced solid tumors (NCT03785249), adagrasib exhibited anti-tumor activity. The phase II of the same study showed that in patients with KRASG12C-mutated non-small-cell lung cancer (NSCLC), adagrasib was efficient without new safety signals.2,3,6
In February 2022, the FDA accepted a new drug application (NDA) for adagrasib for the treatment of patients with previously treated KRASG12C–positive NSCLC.7 In December 2022, the FDA granted accelerated approval to adagrasib for the treatment of KRASG12C-mutated locally advanced or metastatic NSCLC who have received at least one prior systemic therapy.8,9 Adagrasib joins sotorasib as another KRASG12C inhibitor approved by the FDA.4
Medical uses
Adagrasib is indicated for the treatment of adults with KRAS G12C-mutated locally advanced or metastatic non-small cell lung cancer (NSCLC), as determined by an FDA approved test, who have received at least one prior systemic therapy.[1][2][4]
History
Approval by the US Food and Drug Administration (FDA) was based on KRYSTAL-1, a multicenter, single-arm, open-label clinical trial (NCT03785249) which included participants with locally advanced or metastatic non-small cell lung cancer with KRAS G12C mutations.[2] Efficacy was evaluated in 112 participants whose disease has progressed on or after platinum-based chemotherapy and an immune checkpoint inhibitor, given either concurrently or sequentially.[2]
The FDA granted the application for adagrasib fast-track, breakthrough therapy, and orphan drug designations.[2]
Research
It is undergoing clinical trials.[5][6][7][8][9][10]
References
- ^ Jump up to:a b c d e f g https://www.accessdata.fda.gov/drugsatfda_docs/label/2022/216340s000lbl.pdf
- ^ Jump up to:a b c d e f g h “FDA grants accelerated approval to adagrasib for KRAS G12C-mutated NSC”. U.S. Food and Drug Administration (FDA). 12 December 2022. Retrieved 14 December 2022.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain. - ^ Jump up to:a b “Mirati Therapeutics Announces U.S. FDA Accelerated Approval of Krazati (adagrasib) as a Targeted Treatment Option for Patients with Locally Advanced or Metastatic Non-Small Cell Lung Cancer (NSCLC) with a KRASG12C Mutation” (Press release). Mirati Therapeutics Inc. 12 December 2022. Retrieved 13 December 2022 – via MultiVu.
- ^ https://www.accessdata.fda.gov/drugsatfda_docs/appletter/2022/216340Orig1s000ltr.pdf
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain. - ^ Hallin J, Engstrom LD, Hargis L, Calinisan A, Aranda R, Briere DM, et al. (January 2020). “The KRASG12C Inhibitor MRTX849 Provides Insight toward Therapeutic Susceptibility of KRAS-Mutant Cancers in Mouse Models and Patients”. Cancer Discovery. 10 (1): 54–71. doi:10.1158/2159-8290.CD-19-1167. PMC 6954325. PMID 31658955.
- ^ Fell JB, Fischer JP, Baer BR, Blake JF, Bouhana K, Briere DM, et al. (July 2020). “Identification of the Clinical Development Candidate MRTX849, a Covalent KRASG12C Inhibitor for the Treatment of Cancer”. Journal of Medicinal Chemistry. 63 (13): 6679–6693. doi:10.1021/acs.jmedchem.9b02052. PMID 32250617.
- ^ Thein KZ, Biter AB, Hong DS (January 2021). “Therapeutics Targeting Mutant KRAS”. Annual Review of Medicine. 72: 349–364. doi:10.1146/annurev-med-080819-033145. PMID 33138715. S2CID 226242453.
- ^ Christensen JG, Olson P, Briere T, Wiel C, Bergo MO (August 2020). “Targeting Krasg12c -mutant cancer with a mutation-specific inhibitor”. Journal of Internal Medicine. 288 (2): 183–191. doi:10.1111/joim.13057. PMID 32176377.
- ^ Dunnett-Kane V, Nicola P, Blackhall F, Lindsay C (January 2021). “Mechanisms of Resistance to KRASG12C Inhibitors”. Cancers. 13 (1): 151. doi:10.3390/cancers13010151. PMC 7795113. PMID 33466360.
- ^ Jänne PA, Riely GJ, Gadgeel SM, Heist RS, Ou SI, Pacheco JM, et al. (July 2022). “Adagrasib in Non–Small-Cell Lung Cancer Harboring a KRASG12C Mutation”. New England Journal of Medicine. 387 (2): 120–131. doi:10.1056/NEJMoa2204619. PMID 35658005. S2CID 249352736.
External links
- “Adagrasib”. Drug Information Portal. U.S. National Library of Medicine.
- Clinical trial number NCT03785249 for “Phase 1/2 Study of MRTX849 in Patients With Cancer Having a KRAS G12C Mutation KRYSTAL-1” at ClinicalTrials.gov
///////Adagrasib, KRAZATI, FDA 2022, APPROVALS 2022, MRTX-849, MRTX849, Mirati Therapeutics
[H][C@@]1(COC2=NC3=C(CCN(C3)C3=CC=CC4=C3C(Cl)=CC=C4)C(=N2)N2CCN(C(=O)C(F)=C)[C@@]([H])(CC#N)C2)CCCN1C

NEW DRUG APPROVALS
ONE TIME
$10.00

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO



