Nirsevimab
(Heavy chain)
QVQLVQSGAE VKKPGSSVMV SCQASGGLLE DYIINWVRQA PGQGPEWMGG IIPVLGTVHY
GPKFQGRVTI TADESTDTAY MELSSLRSED TAMYYCATET ALVVSETYLP HYFDNWGQGT
LVTVSSASTK GPSVFPLAPS SKSTSGGTAA LGCLVKDYFP EPVTVSWNSG ALTSGVHTFP
AVLQSSGLYS LSSVVTVPSS SLGTQTYICN VNHKPSNTKV DKRVEPKSCD KTHTCPPCPA
PELLGGPSVF LFPPKPKDTL YITREPEVTC VVVDVSHEDP EVKFNWYVDG VEVHNAKTKP
REEQYNSTYR VVSVLTVLHQ DWLNGKEYKC KVSNKALPAP IEKTISKAKG QPREPQVYTL
PPSREEMTKN QVSLTCLVKG FYPSDIAVEW ESNGQPENNY KTTPPVLDSD GSFFLYSKLT
VDKSRWQQGN VFSCSVMHEA LHNHYTQKSL SLSPGK
(Light chain)
DIQMTQSPSS LSAAVGDRVT ITCQASQDIV NYLNWYQQKP GKAPKLLIYV ASNLETGVPS
RFSGSGSGTD FSLTISSLQP EDVATYYCQQ YDNLPLTFGG GTKVEIKRTV AAPSVFIFPP
SDEQLKSGTA SVVCLLNNFY PREAKVQWKV DNALQSGNSQ ESVTEQDSKD STYSLSSTLT
LSKADYEKHK VYACEVTHQG LSSPVTKSFN RGEC
(Disulfide bridge: H22-H96, H153-H209, H229-L214, H235-H’235, H238-H’238, H270-H330, H376-H434, H’22-H’96, H’153-H’209, H’229-L’214, H’270-H’330, H’376-H’434, L23-L88, L’23-L’88, L134-L194, L’134-L’194)
>Heavy_chain QVQLVQSGAEVKKPGSSVMVSCQASGGLLEDYIINWVRQAPGQGPEWMGGIIPVLGTVHY GPKFQGRVTITADESTDTAYMELSSLRSEDTAMYYCATETALVVSETYLPHYFDNWGQGT LVTVSSASTKGPSVFPLAPSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFP AVLQSSGLYSLSSVVTVPSSSLGTQTYICNVNHKPSNTKVDKRVEPKSCDKTHTCPPCPA PELLGGPSVFLFPPKPKDTLYITREPEVTCVVVDVSHEDPEVKFNWYVDGVEVHNAKTKP REEQYNSTYRVVSVLTVLHQDWLEGKEYKCKVSNKALPAPIEKTISKAKGQPREPQVYTL PPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKTTPPVLDSDGSFFLYSKLT VDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK
>Light_chain DIQMTQSPSSLSAAVGDRVTITCQASQDIVNYLNWYQQKPGKAPKLLIYVASNLETGVPS RFSGSGSGTDFSLTISSLQPEDVATYYCQQYDNLPLTFGGGTKVEIKRTVAAPSVFIFPP SDEQLKSGTASVVCLLNNFYPREAKVQWKVDNALQSGNSQESVTEQDSKDSTYSLSSTLT LSKADYEKHKVYACEVTHQGLSSPVTKSFNRGEC
Nirsevimab
EMS APPROVED 2022/10/31, Beyfortus, AstraZeneca AB
| Formula | C6494H10060N1708O2050S46 |
|---|---|
| CAS | 1989556-22-0 |
| Mol weight | 146334.5658 |
Monoclonal antibody
Prevention of respiratory syncytial virus infection
- Immunoglobulin g1-kappa, anti-(human respiratory syncytial virus fusion glycoprotein f0 (protein f))human monoclonal antibody.gamma.1 heavy chain (1-456) (human vh (homo sapiens ighv1-69*01(ighd)-ighj4*01 (90.1%)) (8.8.19) (1-126) -homo sapiens ighg1*03
- Immunoglobulin g1, anti-(human respiratory syncytial virus fusion protein)(human monoclonal med18897 .gamma.1-chain), disulfide with monoclonal med18897 .kappa.-chain, dimer
Synthesis Reference
Khan, AA et al. (2020) Dosage regimens for and compositions including anti-rsv antibodies. (U.S. Patent No. 2020/0347120 A1). U.S. Patent and Trademark Office. https://patentimages.storage.googleapis.com/6b/d2/10/a841b66e0c90cf/US20200347120A1.pdf
Nirsevimab, sold under the brand name Beyfortus, is a human recombinant monoclonal antibody with activity against respiratory syncytial virus, or RSV for infants.[2][3] It is under development by AstraZeneca and Sanofi.[2][3] Nirsevimab is designed to bind to the fusion protein on the surface of the RSV virus.[4][5]
The most common side effects reported for nirsevimab are rash, pyrexia (fever) and injection site reactions (such as redness, swelling and pain where the injection is given).[6]
Nirsevimab was approved for medical use in the European Union in November 2022.[1][7]
Nirsevimab (MEDI8897) is a recombinant human immunoglobulin G1 kappa (IgG1ĸ) monoclonal antibody used to prevent respiratory syncytial virus (RSV) lower respiratory tract disease in neonates and infants.6 It binds to the prefusion conformation of the RSV F protein, a glycoprotein involved in the membrane fusion step of the viral entry process, and neutralizes several RSV A and B strains.6,1 Compared to palivizumab, another anti-RSV antibody, nirsevimab shows greater potency at reducing pulmonary viral loads in animal models. In addition, nirsevimab was developed as a single-dose treatment for all infants experiencing their first RSV season, whereas palivizumab requires five monthly doses to cover an RSV season.5 This is due to a modification in the Fc region of nirsevimab that grants it a longer half-time compared to typical monoclonal antibodies.1,6
On November 2022, nirsevimab was approved by the EMA for the prevention of RSV lower respiratory tract disease in newborns and infants during their first RSV season.6
////////

AS ON DEC2021 3,491,869 VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@amcrasto
/////////////////////////////////////////////////////////////////////////////
| Monoclonal antibody | |
|---|---|
| Type | Whole antibody |
| Source | Human |
| Target | F protein of RSV |
| Clinical data | |
| Trade names | Beyfortus |
| Other names | MED-18897, MEDI8897 |
| Routes of administration | Intramuscular |
| ATC code | None |
| Legal status | |
| Legal status | EU: Rx-only [1] |
| Identifiers | |
| CAS Number | 1989556-22-0 |
| PubChem SID | 384585358 |
| DrugBank | DB16258 |
| UNII | VRN8S9CW5V |
| KEGG | D11380 |
| ChEMBL | ChEMBL4297575 |
| Chemical and physical data | |
| Formula | C6494H10060N1708O2050S46 |
| Molar mass | 146336.58 g·mol−1 |
Adverse effects
No major hypersensitivity reactions have been reported, and adverse events of grade 3 or higher were only reported in 8% (77 of 968) of participants in clinical trial NCT02878330.[8][4]
Pharmacology
Mechanism of action
Nirsevimab binds to the prefusion conformation of the RSV fusion protein, i.e. it binds to the site at which the virus would attach to a cell; effectively rendering it useless. It has a modified Fc region, extending the half-life of the drug in order for it to last the whole RSV season.[4]
History
The opinion by the Committee for Medicinal Products for Human Use (CHMP) of the European Medicines Agency (EMA) is based on data from two randomized, double-blind, placebo-controlled multicenter clinical trials that investigated the efficacy and safety of nirsevimab in healthy preterm (premature) and full-term infants entering their first respiratory syncytial virus (RSV) season.[6] These studies demonstrated that nirsevimab prevents lower respiratory tract infection caused by RSV requiring medical attention (such as bronchiolitis and pneumonia) in term and preterm infants during their first RSV season.[6]
The safety of nirsevimab was also evaluated in a phase II/III, randomized, double‑blind, multicenter trial in infants who were born five or more weeks prematurely (less than 35 weeks gestation) at higher risk for severe RSV disease and infants with chronic lung disease of prematurity (i.e. long-term respiratory problems faced by babies born prematurely) or congenital heart disease.[6] The results of this study showed that nirsevimab had a similar safety profile compared to palivizumab (Synagis).[6]
Society and culture
Legal status
On 15 September 2022, the Committee for Medicinal Products for Human Use (CHMP) of the European Medicines Agency (EMA) adopted a positive opinion, recommending the granting of a marketing authorization for the medicinal product Beyfortus, intended for the prevention of respiratory syncytial virus (RSV) lower respiratory tract disease in newborns and infants.[9][6] Beyfortus was reviewed under EMA’s accelerated assessment program.[9] The applicant for this medicinal product is AstraZeneca AB.[9] Nirsevimab was approved for medical use in the European Union in November 2022.[1][7]
Research
Nirsevimab is being investigated as an experimental vaccine against respiratory syncytial virus, RSV, in the general infant population.[2][3] The MELODY study is an ongoing, randomized, double-blind, placebo-controlled to evaluate the safety and efficacy of nirsevimab in late preterm and term infants. Initial results have been promising, with nirsevimab reducing LRTI (lower respiratory tract infections) by 74.5% compared to placebo in infants born at term or late preterm.[5][10][11]
Ongoing trials for nirsevimab are:
- “Evaluate the Safety and Efficacy of Nirsevimab in Healthy Preterm and Term Infants in China (CHIMES)”.
- “A Study to Evaluate the Safety and Efficacy of MEDI8897 for the Prevention of Medically Attended Lower Respiratory Tract Infection Due to Respiratory Syncytial Virus in Healthy Late Preterm and Term Infants (MELODY)”.
- “Evaluate the Safety and Tolerability, for Nirsevimab in Immunocompromised Children (MUSIC)”.
References
- ^ Jump up to:a b c “Beyfortus”. Union Register of medicinal products. 3 November 2022. Retrieved 6 November 2022.
- ^ Jump up to:a b c “Nirsevimab demonstrated protection against respiratory syncytial virus disease in healthy infants in Phase 3 trial” (Press release). Sanofi. 26 April 2021. Archived from the original on 27 December 2021. Retrieved 27 December 2021.
- ^ Jump up to:a b c “Nirsevimab MELODY Phase III trial met primary endpoint of reducing RSV lower respiratory tract infections in healthy infants” (Press release). AstraZeneca. 26 April 2021. Archived from the original on 26 December 2021. Retrieved 27 December 2021.
- ^ Jump up to:a b c Griffin MP, Yuan Y, Takas T, Domachowske JB, Madhi SA, Manzoni P, et al. (Nirsevimab Study Group) (July 2020). “Single-Dose Nirsevimab for Prevention of RSV in Preterm Infants”. The New England Journal of Medicine. 383 (5): 415–425. doi:10.1056/NEJMoa1913556. PMID 32726528. S2CID 220876651.
- ^ Jump up to:a b Hammitt LL, Dagan R, Yuan Y, Baca Cots M, Bosheva M, Madhi SA, et al. (March 2022). “Nirsevimab for Prevention of RSV in Healthy Late-Preterm and Term Infants”. The New England Journal of Medicine. 386 (9): 837–846. doi:10.1056/NEJMoa2110275. PMID 35235726. S2CID 247220023.
- ^ Jump up to:a b c d e f “New medicine to protect babies and infants from respiratory syncytial virus (RSV) infection”. European Medicines Agency (EMA) (Press release). 16 September 2022. Archived from the original on 19 September 2022. Retrieved 18 September 2022. Text was copied from this source which is copyright European Medicines Agency. Reproduction is authorized provided the source is acknowledged.
- ^ Jump up to:a b “Beyfortus approved in the EU for the prevention of RSV lower respiratory tract disease in infants”. AstraZeneca (Press release). 4 November 2022. Retrieved 6 November 2022.
- ^ Clinical trial number NCT02878330 at ClinicalTrials.gov
- ^ Jump up to:a b c “Beyfortus: Pending EC decision”. European Medicines Agency (EMA). 15 September 2022. Archived from the original on 19 September 2022. Retrieved 18 September 2022. Text was copied from this source which is copyright European Medicines Agency. Reproduction is authorized provided the source is acknowledged.
- ^ Zacks Equity Research (25 March 2022). “Pfizer’s (PFE) RSV Jab Gets Another Breakthrough Therapy Tag”. Nasdaq. Archived from the original on 8 April 2022. Retrieved 8 April 2022.
- ^ “Nirsevimab significantly protected infants against RSV disease in Phase III MELODY trial”. AstraZeneca (Press release). 3 March 2022. Retrieved 6 November 2022.
////////////Nirsevimab, EU 2022, APPROVALS 2022, PEPTIDE, Monoclonal antibody, respiratory syncytial virus infection, ANTIVIRAL, 1989556-22-0, MED-18897, MEDI8897, AstraZeneca AB

NEW DRUG APPROVALS
ONE TIME
$10.00
Olutasidenib

Olutasidenib
- FT-2102
- FT2102
C18H15ClN4O2
354.79
CAS1887014-12-1
Rezlidhia (Forma Therapeutics)
SYN Caravella JA, et al. Structure-Based Design and Identification of FT-2102 (Olutasidenib), a Potent Mutant-Selective IDH1 Inhibitor. J Med Chem. 2020 Feb 27;63(4):1612-1623. doi: 10.1021/acs.jmedchem.9b01423. Epub 2020 Feb 12.
FDA 12/1/2022, To treat adults with relapsed or refractory acute myeloid leukemia with a susceptible isocitrate dehydrogenase-1 (IDH1) mutation, Rezlidhia
Olutasidenib, sold under the brand name Rezlidhia, is an anticancer medication used to treat relapsed or refractory acute myeloid leukemia.[1][2] Olutasidenib is an isocitrate dehydrogenase-1 (IDH1) inhibitor.[1] It is taken by mouth.[1]
Olutasidenib was approved for medical use in the United States in December 2022.[1][2][3][4]
Medical uses
Olutasidenib is indicated for the treatment of adults with relapsed or refractory acute myeloid leukemia with a susceptible isocitrate dehydrogenase-1 (IDH1) mutation as detected by an FDA-approved test.[1][2]
Society and culture
Names
Olutasidenib is the international nonproprietary name.[5]
Olutasidenib is an isocitrate dehydrogenase-1 (IDH1) inhibitor indicated for the treatment of patients with relapsed or refractory acute myeloid leukemia with a susceptible IDH1 mutation as detected by an FDA-approved test.
Olutasidenib (FT-2102) is a selective and potent isocitrate dehydrogenase-1 (IDH1) inhibitor approved by the FDA in December 2022.5,6 It is indicated for the treatment of relapsed or refractory acute myeloid leukemia (AML) in patients with a susceptible IDH1 mutation as determined by an FDA-approved test.5 IDH1 mutations are common in different types of cancer, such as gliomas, AML, intrahepatic cholangiocarcinoma, chondrosarcoma, and myelodysplastic syndromes (MDS), and they lead to an increase in 2-hydroxyglutarate (2-HG), a metabolite that participates in tumerogenesis.1,2 Olutasidenib inhibits the mutated IDH1 specifically, and provides a therapeutic benefit in IDH1-mutated cancers.1,5
Other IDH1 inhibitors, such as ivosidenib, have also been approved for the treatment of relapsed or refractory AML.3,4 Olutasidenib is orally bioavailable and capable of penetrating the blood-brain barrier, and is also being evaluated for the treatment of myelodysplastic syndrome (MDS), as well as solid tumors and gliomas (NCT03684811).4
SYN
https://pubs.acs.org/doi/10.1021/acs.jmedchem.9b01423

a Reagents and conditions: (a) DIEA, DMSO, 80−110 °C, 16 h, 67%; (b) (R)-2-methylpropane-2-sulfinamide, CuSO4, 55 °C, DCE, 16 h, 81%; (c) MeMgBr, DCM, −50 to −60 °C, 3 h, 63%; (d) 1 N HCl, dioxane, reflux, 16 h, >98%, 98.4% ee; (e) m-CPBA, CHCl3, reflux, 4 days, 52%; (f) Ac2O, reflux, 3 days, 60%; (g) K2CO3, MeOH, 4 h, 92%; (h) MeI, K2CO3, DMF, 45 min, 67%.
1H NMR (300 MHz,
DMSO-d6) δ 12.07 (s, 1 H), 7.71−7.76 (m, 2 H), 7.51 (dd, J = 8.79,
2.35 Hz, 1 H), 7.31 (d, J = 8.79 Hz, 1 H), 6.97 (d, J = 7.92 Hz, 1 H),
6.93 (d, J = 7.92 Hz, 1 H), 5.95 (d, J = 7.92 Hz, 1 H), 4.62−4.75 (m,
1 H), 3.58 (s, 3 H), 1.50 (d, J = 6.74 Hz, 3 H); 13C NMR (75 MHz,
DMSO-d6) δ 161.0, 155.9, 141.4, 136.6, 135.0, 133.4, 129.8, 126.7,
125.8, 120.1, 119.4, 116.7, 115.1, 104.5, 103.7, 47.4, 34.0, 20.3; LCMS
(method 2) >95% purity; tR 10.18 min; m/z 355, 357 [M + H]+
;
HRMS (ESI) calcd for C18H16ClN4O2 [M + H]+ 355.0962 found
356.0956.
////////

AS ON DEC2021 3,491,869 VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@amcrasto
/////////////////////////////////////////////////////////////////////////////
| Clinical data | |
|---|---|
| Trade names | Rezlidhia |
| Other names | FT-2102 |
| License data | US DailyMed: Olutasidenib |
| Routes of administration | By mouth |
| ATC code | None |
| Legal status | |
| Legal status | US: ℞-only [1][2] |
| Identifiers | |
| CAS Number | 1887014-12-1 |
| PubChem CID | 118955396 |
| IUPHAR/BPS | 10319 |
| DrugBank | DB16267 |
| ChemSpider | 72380144 |
| UNII | 0T4IMT8S5Z |
| KEGG | D12483 |
| ChEMBL | ChEMBL4297610 |
| PDB ligand | PWV (PDBe, RCSB PDB) |
| Chemical and physical data | |
| Formula | C18H15ClN4O2 |
| Molar mass | 354.79 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| showSMILES | |
References
- ^ Jump up to:a b c d e f https://www.accessdata.fda.gov/drugsatfda_docs/label/2022/215814s000lbl.pdf
- ^ Jump up to:a b c d https://www.accessdata.fda.gov/drugsatfda_docs/appletter/2022/215814Orig1s000ltr.pdf
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain. - ^ “Rigel Announces U.S. FDA Approval of Rezlidhia (olutasidenib) for the Treatment of Adult Patients with Relapsed or Refractory Acute Myeloid Leukemia with a Susceptible IDH1 Mutation”. Rigel Pharmaceuticals, Inc. (Press release). 1 December 2022. Retrieved 2 December 2022.
- ^ “Rigel Announces U.S. FDA Approval of Rezlidhia (olutasidenib) for the Treatment of Adult Patients with Relapsed or Refractory Acute Myeloid Leukemia with a Susceptible IDH1 Mutation” (Press release). Rigel Pharmaceuticals. 1 December 2022. Retrieved 2 December 2022 – via PR Newswire.
- ^ World Health Organization (2019). “International nonproprietary names for pharmaceutical substances (INN): recommended INN: list 82”. WHO Drug Information. 33 (3). hdl:10665/330879.
Further reading
- Liu X, Gong Y (2019). “Isocitrate dehydrogenase inhibitors in acute myeloid leukemia”. Biomarker Research. 7: 22. doi:10.1186/s40364-019-0173-z. PMC 6806510. PMID 31660152.
- Watts JM, Baer MR, Yang J, Prebet T, Lee S, Schiller GJ, et al. (November 2022). “Olutasidenib alone or with azacitidine in IDH1-mutated acute myeloid leukaemia and myelodysplastic syndrome: phase 1 results of a phase 1/2 trial”. The Lancet Haematology. doi:10.1016/S2352-3026(22)00292-7. PMID 36370742. S2CID 253471380.
External links
- “Olutasidenib”. Drug Information Portal. U.S. National Library of Medicine.
- Clinical trial number NCT02719574 for “Open-label Study of FT-2102 With or Without Azacitidine or Cytarabine in Patients With AML or MDS With an IDH1 Mutation” at ClinicalTrials.gov
/////////////Olutasidenib, FDA 2022, APPROVALS 2022, Rezlidhia, FT-2102, FT 2102

NEW DRUG APPROVALS
ONE TIME
$10.00
ABY 737
ABT-737
| Molecular Weight | 813.43 |
|---|---|
| Formula | C42H45ClN6O5S2 |
| CAS No. | 852808-04-9 |
ABT-737 is a small molecule drug that inhibits Bcl-2 and Bcl-xL, two members of the Bcl-2 family of evolutionarily-conserved proteins that share Bcl-2 Homology (BH) domains. First developed as a potential cancer chemotherapy,[1] it was subsequently identified as a senolytic (a drug that selectively induces cell death in senescent cells).[2]
The Bcl-2 family is most notable for their regulation of apoptosis, a form of programmed cell death, at the mitochondrion; Bcl-2 and Bcl-xL are anti-apoptotic proteins. Because many cancers have mutations in these genes that allow them to survive, scientists began working to develop drugs that would inhibit this pathway in the 1990s.[1] ABT-737 was one of the earliest of a series of drugs developed by Abbott Laboratories (now Abbvie) to target this pathway, based on their resolution of the 3D structure of Bcl-xL and studies using high-field solution nuclear magnetic resonance (NMR) that revealed how the BH domains of these proteins interacted with their targets.[1]
ABT-737 was superior to previous BCL-2 inhibitors given its higher affinity for Bcl-2, Bcl-xL and Bcl-w. In vitro studies showed that primary cells from patients with B-cell malignancies are sensitive to ABT-737.[3] In animal models, it improved survival, caused tumor regression, and cured a high percentage of mice.[4] In preclinical studies utilizing patient xenografts, ABT-737 showed efficacy for treating lymphoma and other blood cancers.[5]
Unfortunately, ABT-737 is not bioavailable after oral administration, leading to the development of navitoclax (ABT-263) as an orally-available derivative with similar activity on small cell lung cancer (SCLC) cell lines.[1][6] Navitoclax entered clinical trials,[1][6] and showed promise in haematologic cancers, but was stalled when it was found to cause thrombocytopenia (severe loss of platelets), which was discovered to be caused by the platelets’ requirement for Bcl-xL for survival.[1]
Subsequently, it was reported that ABT-737 specifically induces apoptosis in senescent cells in vitro and in mouse models.[2]
ABT-737, a BH3 mimetic, is a potent Bcl-2, Bcl-xL and Bcl-w inhibitor with EC50s of 30.3 nM, 78.7 nM, and 197.8 nM, respectively. ABT-737 induces the disruption of the BCL-2/BAX complex and BAK-dependent but BIM-independent activation of the intrinsic apoptotic pathway. ABT-737 induces autophagy and has the potential for acute myeloid leukemia (AML) research.
PATENT
PATENT
CN113248415
PATENT
US20070015787
Journal of Medicinal Chemistry (2007), 50(4), 641-662
https://pubs.acs.org/doi/10.1021/jm061152t

////////

AS ON DEC2021 3,491,869 VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@amcrasto
/////////////////////////////////////////////////////////////////////////////
| Names | |
|---|---|
| Preferred IUPAC name4-{4-[(4′-Chloro[1,1′-biphenyl]-2-yl)methyl]piperazin-1-yl}-N-(4-{[(2R)-4-(dimethylamino)-1-(phenylsulfanyl)butan-2-yl]amino}-3-nitrobenzene-1-sulfonyl)benzamide | |
| Identifiers | |
| CAS Number | 852808-04-9 |
| 3D model (JSmol) | Interactive image |
| ChEBI | CHEBI:47575 |
| ChemSpider | 9403232 |
| PubChemCID | 11228183 |
| UNII | Z5NFR173NV |
| CompTox Dashboard (EPA) | DTXSID7042641 |
| showInChI | |
| showSMILES | |
| Properties | |
| Chemical formula | C42H45ClN6O5S2 |
| Molar mass | 813.43 g·mol−1 |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). |
References
- ^ Jump up to:a b c d e f Croce, Carlo M; Reed, John C (October 2016). “Finally, An Apoptosis-Targeting Therapeutic for Cancer”. Cancer Research. 76 (20): 5914–5920. doi:10.1158/0008-5472.CAN-16-1248. PMC 5117672. PMID 27694602.
- ^ Jump up to:a b Yosef, Reut; Pilpel, Noam; Tokarsky-Amiel, Ronit; Biran, Anat; Ovadya, Yossi; Cohen, Snir; Vadai, Ezra; Dassa, Liat; Shahar, Elisheva; Condiotti, Reba; Ben-Porath, Ittai; Krizhanovsky, Valery (2016). “Directed elimination of senescent cells by inhibition of BCL-W and BCL-XL”. Nature Communications. 7: 11190. Bibcode:2016NatCo…711190Y. doi:10.1038/ncomms11190. PMC 4823827. PMID 27048913.
- ^ Vogler, Meike, et al. “Bcl-2 inhibitors: small molecules with a big impact on cancer therapy.” Cell Death & Differentiation 16.3 (2008): 360–367.
- ^ Oltersdorf, Tilman; Elmore, Steven W.; Shoemaker, Alexander R.; Armstrong, Robert C.; Augeri, David J.; Belli, Barbara A.; Bruncko, Milan; Deckwerth, Thomas L.; Dinges, Jurgen; Hajduk, Philip J.; Joseph, Mary K.; Kitada, Shinichi; Korsmeyer, Stanley J.; Kunzer, Aaron R.; Letai, Anthony; Li, Chi; Mitten, Michael J.; Nettesheim, David G.; Ng, ShiChung; Nimmer, Paul M.; O’Connor, Jacqueline M.; Oleksijew, Anatol; Petros, Andrew M.; Reed, John C.; Shen, Wang; Tahir, Stephen K.; Thompson, Craig B.; Tomaselli, Kevin J.; Wang, Baole; Wendt, Michael D.; Zhang, Haichao; Fesik, Stephen W.; Rosenberg, Saul H. (2005). “An inhibitor of Bcl-2 family proteins induces regression of solid tumours”. Nature. 435 (7042): 677–81. Bibcode:2005Natur.435..677O. doi:10.1038/nature03579. PMID 15902208. S2CID 4335635.
- ^ Hann CL, Daniel VC, Sugar EA, Dobromilskaya I, Murphy SC, Cope L, Lin X, Hierman JS, Wilburn DL, Watkins DN, Rudin CM (April 2008). “Therapeutic efficacy of ABT-737, a selective inhibitor of BCL-2, in small cell lung cancer”. Cancer Research. 68 (7): 2321–8. doi:10.1158/0008-5472.can-07-5031. PMC 3159963. PMID 18381439.
- ^ Jump up to:a b Hauck P, Chao BH, Litz J, Krystal GW (April 2009). “Alterations in the Noxa/Mcl-1 axis determine sensitivity of small cell lung cancer to the BH3 mimetic ABT-737”. Mol Cancer Ther. 8 (4): 883–92. doi:10.1158/1535-7163.MCT-08-1118. PMID 19372561. Retrieved 9 September 2019.
///////////ABT-737, ABT 737
CN(CC[C@@H](NC1=CC=C(C=C1[N+]([O-])=O)S(NC(C2=CC=C(C=C2)N3CCN(CC3)CC4=CC=CC=C4C5=CC=C(C=C5)Cl)=O)(=O)=O)CSC6=CC=CC=C6)C

NEW DRUG APPROVALS
ONE TIME
$9.00
Tolebrutinib, SAR 442168
Tolebrutinib
SAR442168
- Treatment of Multiple Sclerosis (MS)
CAS 1971920-73-6
PRN 2246, example 3 [WO2016196840A1]
C26H25N5O3,
| 455.5 |
4-amino-3-(4-phenoxyphenyl)-1-[(3R)-1-prop-2-enoylpiperidin-3-yl]imidazo[4,5-c]pyridin-2-one
4-amino-3-(4-phenoxyphenyl)-1-[(3R)-1-prop-2-enoylpiperidin-3-yl]imidazo[4,5-c]pyridin-2-one
(R)-1-(1-Acryloylpiperidin-3-yl)-4-amino-3-(4-phenoxyphenyl)-1H-imidazo[4,5-c]pyridin-2(3H)-one
4-amino-3-(4-phenoxyphenyl)-1-[(3R)-1-(prop-2-
enoyl)piperidin-3-yl]-1,3-dihydro-2H-imidazo[4,5-
Tolebrutinib (R&D code SAR442168), developed by Principia and later acquired by Sanofi and included in its product line, Tolebrutinib is a BTK inhibitor used to treat cancer, autoimmune diseases such as multiple sclerosis and myasthenia gravis, inflammatory diseases and thromboembolic diseases, etc.,
Tolebrutinib is an orally bioavailable, brain-penetrant, selective, small molecule inhibitor of Bruton’s tyrosine kinase (BTK), with potential immunomodulatory and anti-inflammatory activities. Upon oral administration, tolebrutinib is able to cross the blood-brain barrier and inhibits the activity of BTK both peripherally and in the central nervous system (CNS). This prevents the activation of the B-cell antigen receptor (BCR) signaling pathway, and the resulting immune activation and inflammation. The inhibition of BTK activity also prevents microglial inflammatory signaling in the CNS, and the resulting immune activation, neuroinflammation and neurodegeneration. BTK, a cytoplasmic tyrosine kinase and member of the Tec family of kinases, plays an important role in B lymphocyte development, activation, signaling, proliferation and survival. In addition to B cells, BTK is also expressed in innate immune cells, including macrophages and microglia, and plays an important role in the regulation of microglial inflammatory signaling.
BTK, a member of the Tec family non-receptor tyrosine kinases, is essential for B cell signaling downstream from the B-cell receptor. It is expressed in B cells and other hematopoietic cells such as monocytes, macrophages and mast cells. It functions in various aspects of B cell function that maintain the B cell repertoire (see Gauld S. B. et al., B cell antigen receptor signaling: roles in cell development and disease. Science,
296: 1641 -2. 2002.) B cells pay a role in rheumatoid arthritis (see Perosa F., et ai, CD20-depleting therapy in autoimmune diseases: from basic research to the clinic. / Intern Med. 267:260-77. 2010 and Dorner T, et at. Targeting B cells in immune-mediated
inflammatory disease: a comprehensive review of mechanisms of action and identification of biomarkers. Pharmacol The 125:464-75. 2010 and Honigberg, L., et. ai, The selective BTK inhibitor PCI-32765 blocks B cell and mast cell activation and prevents mouse collagen indiced arthritis. Clin. Immunol. 127 SI :S 111. 2008) and in other autoimmune diseases such as systemic lupus erythematosus and cancers (see Shlomchik M. J., et. ai, The role of B cells in lpr/lpr-induced autoimmunity. /. Exp Med. 180:1295-1306. 1994; Honigberg L. A., The Braton tyrosine kinase inhibitor PCI-32765 blocks B-cell activation and is efficacious in models of autoimmune disease and B-cell malignancy. Proc. Natl. Acad. Sci. 107: 13075-80. 2010; and Mina-Osorio P, et al., Suppression of
glomerulonephritis in lupus-prone NZB x NZW mice by RN486, a selective inhibitor of Bruton’s tyrosine kinase. Arthritis Rheum. 65: 2380-91. 2013).
There is also potential for BTK inhibitors for treating allergic diseases (see Honigberg, L., et. al., The selective BTK inhibitor PCI-32765 blocks B cell and mast cell activation and prevents mouse collagen indiced arthritis. Clin. Immunol. 127 SI :S111. 2008). It was noted that the irreversible inhibitor suppresses passive cutaneous anaphylaxis (PCA) induced by IgE antigen complex in mice. These findings are in agreement with those noted with BTK-mutant mast cells and knockout mice and suggest that BTK inhibitors may be useful for the treatment of asthma, an IgE-dependent allergic disease of the airway.
Accordingly, compounds that inhibit BTK would be useful in treatment for diseases such as autoimmune diseases, inflammatory diseases, and cancer.
PATENT
PATENT
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2016196840
Example 3
Synthesis of (R)-l-(l-acryloylpiperidin-3-yl)-4-amino-3-(4-phenoxyphenyl)-lH- imidazo[4,5-c]pyridin-2(3H)-one
Into a 100-mL round-bottom flask, was placed (R)-4-amino-3-(4-phenoxyphenyl)-l-(piperidin-3-yl)-lH-imidazo[4,5-c]pyridin-2(3H)-one (150 mg, 0.37 mmol, 1.00 equiv), DCM-CH30H (6 mL), TEA (113 mg, 1.12 mmol, 3.00 equiv). This was followed by the addition of prop-2-enoyl chloride (40.1 mg, 0.44 mmol, 1.20 equiv) dropwise with stirring at OoC in 5 min. The resulting solution was stirred for 2 h at 0 °C. The resulting mixture was concentrated under vacuum. The residue was applied onto a silica gel column with dichloromethane/methanol (30: 1). The crude product (100 mg) was purified by Prep-HPLC with the following conditions (Column, XBridge Prep CI 8 OBD
Column,5um, 19*150mm; mobile phase, water with 0.05%TFA and ACN (25.0% ACN up to 45.0% in 8 min). 54.5 mg product of (R)-l-(l -acryloylpiperidin-3-yl)-4-amino-3-(4-phenoxyphenyl)-lH-imidazo[4,5-c]pyridin-2(3H)-one was obtained as a white solid. LC-MS m/z: 465.2 (M+l)
Step 2
Into a 25-mL round-bottom flask was placed tert-butyl (3R)-3-[4-[(E)-[(dimethy]amino)-methylidene]-amino]-2-oxo-3-(4-phenoxyphenyl)-lH,2H,3H-imidazo[4,5-c]pyridin-l -yl]piperidine- l-carboxylate (150 mg, 0.27 mmol, 1.00 equiv), 1,4-dioxane (6 mL), and hydrogen chloride (3 mL). The resulting solution was stirred overnight at 50° C. The reaction mixture was quenched with water. The pH of the solution was adjusted to 9 with sodium bicarbonate. The resulting solution was extracted with dichloromethane:CH3OH=10: 1 and the organic layers were combined. The resulting mixture was washed with sodium chloride and the organic layers were combined, dried over anhydrous sodium sulfate and concentrated under vacuum. The residue was applied onto a silica gel column and eluted with dichloromethane/methanol (30: 1) to give 80 mg (74%) of 4-amino-3-(4-phenoxyphenyl)-l -[(3R)-piperidin-3-yl]-lH,2H,3H-imidazo[4,5-c]pyridin-2-one as a light yellow solid.
////////

AS ON DEC2021 3,491,869 VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@amcrasto
/////////////////////////////////////////////////////////////////////////////
/////////Tolebrutinib, SAR 442168, PRN 2246, GTPL10625, BTK’168, EX-A4699, BDBM50557487, WHO 11268, Multiple Sclerosis, (MS),

NEW DRUG APPROVALS
ONE TIME
$10.00
Mirvetuximab soravtansine-gynx

Mirvetuximab soravtansine-gynx
FDA 11/14/2022,To treat patients with recurrent ovarian cancer that is resistant to platinum therapy
| Elahere |
FDA Approves Mirvetuximab Soravtansine-gynx for FRα+ Platinum-resistant Ovarian Cancer
https://www.biochempeg.com/article/315.html
4846-85a8-48171ab38275
FDA Approves Mirvetuximab Soravtansine-gynx for FRα+ Platinum-resistant Ovarian Cancer
November 15, 2022
The FDA has granted accelerated approval to mirvetuximab soravtansine-gynx (Elahere) for the treatment of select patients with folate receptor α–positive, platinum-resistant epithelial ovarian, fallopian tube, or primary peritoneal cancer.
The FDA has granted accelerated approval to mirvetuximab soravtansine-gynx (Elahere) for the treatment of adult patients with folate receptor α (Frα)–positive, platinum-resistant epithelial ovarian, fallopian tube, or primary peritoneal cancer, who have received 1 to 3 prior systemic treatment regimens.1-3
The regulatory agency also gave the green light to the VENTANA FOLR1 (FOLR-2.1) RxDx Assay for use as a companion diagnostic device to identify patients who are eligible to receive the agent. Testing can be done on fresh or archived tissue. Newly diagnosed patients can be tested at diagnosis to determine whether this agent will be an option for them at the time of progression to platinum resistance.
The decision was supported by findings from the phase 3 SORAYA trial (NCT04296890), in which mirvetuximab soravtansine elicited a confirmed investigator-assessed objective response rate (ORR) of 31.7% (95% CI, 22.9%-41.6%); this included a complete response rate of 4.8% and a partial response rate of 26.9%. Moreover, the median duration of response (DOR) was 6.9 months (95% CI, 5.6-9.7) per investigator assessment.
“The approval of Elahere is significant for patients with FRα-positive platinum-resistant ovarian cancer, which is characterized by limited treatment options and poor outcomes,” Ursula Matulonis, MD, chief of the Division of Gynecologic Oncology at the Dana-Farber Cancer Institute, professor of medicine at the Harvard Medical School, and SORAYA co-principal investigator, stated in a press release. “Elahere impressive anti-tumor activity, durability of response, and overall tolerability observed in SORAYA demonstrate the benefit of this new therapeutic option, and I look forward to treating patients with Elahere.”
The global, single-arm SORAYA trial enrolled a total of 106 patients with platinum-resistant ovarian cancer whose tumors expressed high levels of FRα. Patients were allowed to have received up to 3 prior lines of systemic treatment, and all were required to have received bevacizumab (Avastin).
If patients had corneal disorders, ocular conditions in need of ongoing treatment, peripheral neuropathy that was greater than grade 1 in severity, or noninfectious interstitial lung disease, they were excluded.
Study participants received intravenous mirvetuximab soravtansine at 6 mg/kg once every 3 weeks until progressive disease or unacceptable toxicity. Investigators conducted tumor response assessments every 6 weeks for the first 36 weeks, and every 12 weeks thereafter.
Confirmed investigator-assessed ORR served as the primary end point for the research, and the key secondary end point was DOR by RECIST v1.1 criteria.
In the efficacy-evaluable population (n = 104), the median age was 62 years (range, 35-85). Ninety-six percent of patients were White, 2% were Asian, and 2% did not have their race information reported; 2% of patients were Hispanic or Latino. Regarding ECOG performance status, 57% of patients had a status of 0 and the remaining 43% had a status of 1.
Ten percent of patients received 1 prior line of systemic treatment, 39% received 2 prior lines, and 50% received 3 or more prior lines. All patients previously received bevacizumab, as required, and 47% previously received a PARP inhibitor.
The safety of mirvetuximab soravtansine was evaluated in all 106 patients. The median duration of treatment with the agent was 4.2 months (range, 0.7-13.3).
The all-grade toxicities most commonly experienced with mirvetuximab soravtansine included vision impairment (50%), fatigue (49%), increased aspartate aminotransferase (50%), nausea (40%), increased alanine aminotransferase (39%), keratopathy (37%), abdominal pain (36%), decreased lymphocytes (35%), peripheral neuropathy (33%), diarrhea (31%), decreased albumin (31%), constipation (30%), increased alkaline phosphatase (30%), dry eye (27%), decreased magnesium (27%), decreased leukocytes (26%), decreased neutrophils (26%), and decreased hemoglobin (25%).
Thirty-one percent of patients experienced serious adverse reactions with the agent, which included intestinal obstruction (8%), ascites (4%), infection (3%), and pleural effusion (3%). Toxicities proved to be fatalfor 2% of patients, and these included small intestinal obstruction (1%) and pneumonitis (1%).
Twenty percent of patients required dose reductions due to toxicities. Eleven percent of patients discontinued treatment with mirvetuximab soravtansine because of adverse reactions. Toxicities that resulted in more than 2% of patients discontinuing treatment included intestinal obstruction (2%) and thrombocytopenia (2%). One patient discontinued because of visual impairment.
References
- ImmunoGen announces FDA accelered approval of Elahere (mirvetuximab soravtansine-gynx) for the treatment of platinum-resistant ovarian cancer. News release. ImmunoGen Inc. November 14, 2022. Accessed November 14, 2022. http://bit.ly/3GgrCwL
- FDA grants accelerated approval to mirvetuximab soravtansine-gynx for FRα positive, platinum-resistant epithelial ovarian, fallopian tube, or peritoneal cancer. News release. FDA. November 14, 2022. Accessed November 14, 2022. http://bit.ly/3UP742w
- Elahere (mirvetuximab soravtansine-gynx). Prescribing information; ImmunoGen Inc; 2022. Accessed November 14, 2022. https://www.accessdata.fda.gov/drugsatfda_docs/label/2022/761310s000lbl.pdf

NEW DRUG APPROVALS
ONE TIME
$10.00
////////

AS ON DEC2021 3,491,869 VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@amcrasto
/////////////////////////////////////////////////////////////////////////////
//////////Mirvetuximab soravtansine-gynx, FDA 2022, APPROVALS 2022, recurrent ovarian cancer,
| Elahere |
TEREVALEFIM

TEREVALEFIM
Molecular Formula
- C9-H8-N2-S
Molecular Weight
- 176.2382
RN: 1070881-42-3
UNII: GG91UXK2M5
- 5-((E)-2-Thiophen-2-yl-vinyl)-lh-pyrazole
- 1H-Pyrazole, 3-((1E)-2-(2-thienyl)ethenyl)-
- ANG-3777
- SNV-003
- OriginatorAngion Biomedica
- ClassAnti-ischaemics; Antifibrotics; Heart failure therapies; Pyrazoles; Small molecules; Thiophenes; Urologics; Vascular disorder therapies
- Mechanism of ActionProto oncogene protein c met stimulants
- Orphan Drug StatusYes – Renal failure
- Phase IIIDelayed graft function
- Phase IIAcute kidney injury; Acute lung injury; Renal failure
- PreclinicalBrain injuries
- No development reportedHeart failure
- DiscontinuedHepatic fibrosis; Myocardial infarction; Stroke
- 02 Aug 2022Vifor Pharma has been acquired by CSL and renamed to CSL Vifor
- 14 Dec 2021Efficacy and adverse events data of a phase II GUARD trial in Acute kidney injury released by the company
- 26 Oct 2021Top-line efficacy and adverse events data from the phase III trial GIFT (Graft Improvement Following Transplant) trial in Delayed graft function released by Angion Biomedica and Vifor Pharma
Terevalefim, an hepatocyte growth factor (HGF) mimetic, selectively activates the c-Met receptor.
PATENT
WO 2004/058721
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2004058721
PATENT
PCT Application No. PCT/US2003/040917, filed December 19, 2003 and published as WO2004/058721 on July 15, 2004, the entirety of which is hereby incorporated by reference, describes certain compounds that act as HGF/SF mimetics . Such compounds include terevalefim:
Terevalefim has been demonstrated to be remarkably useful for treatment of a variety of conditions including, for example, fibrotic liver disease, ischemia-reperfusion injury, cerebral infarction, ischemic heart disease, renal disease, lung fibrosis, damaged and/or ischemic organs, transplants or grafts, stroke, cerebrovascular disease, and renal fibrosis, among others (see, for example, WO 2004/058721, WO 2010/005580, US 2011/0230407, US 7879898, and WO 2009/064422, each of which is hereby incorporated by reference.) Exemplary methods of using terevalefim for, eg, treating delayed graft function after kidney transplantation and acute lung injury, are described in WO 2021/087392 and WO 2021/183774, each of which is hereby incorporated by reference. In particular, Terevalefim is or has been the subject of clinical trials for delayed graft function in recipients of a deceased donor kidney (Clinicaltrials.gov identifier: NCT02474667), acute kidney injury after cardiac surgery involving cardiopulmonary bypass (Clinicaltrials.gov identifier: NCT02771509), and COVID -19 pneumonia (Clinicaltrials.gov identifier: NCT04459676). Without wishing to be bound by any particular theory, it is believed that terevalefim’s HGF mimetic capability imparts a variety of beneficial attributes and activities.
[0035] Terevalefim has a CAS Registry No. of 1070881-42-3 and is also known by at least the following names:
● 3-[(1E)-2-(thiophen-2-yl)ethen-1-yl]-1H-pyrazole; and
● (E)-3-[2-(2-thienyl)vinyl]-1H-pyrazole.
Synthesis of Terevalefim
[0057] In some embodiments, the present disclosure provides methods for preparing compounds useful as HGF/SF mimetics, such as terevalefim. A synthesis of terevalefim is described in detail in Example 7 of WO 2004/058721 (“the ‘721 Synthesis”). The ‘721 Synthesis is depicted in Scheme 1:
The ‘721 Synthesis includes certain features which are not desirable for preparation of terevalefim at scale and/or with consistency and/or with suitable purity for use in humans. For example, the ‘721 Synthesis includes preparation of aldehyde compound 1.2, a viscous oil that is difficult to purify with standard techniques. Additionally, the ‘721 Synthesis uses a diethoxyphosphorylacetaldehyde tosylhydrazone reagent in step 1-2. As such, step 1-2 has poor atom economy and results in multiple byproducts that must be purified away from the final product of terevalefim. Step 1-2 also uses sodium hydride, a highly reactive base that can be difficult to control and often results in byproducts that must be purified away from the final product of terevalefim. Such purification steps can be costly and time-consuming. In some embodiments, the present disclosure encompasses the recognition that one or more features of the ‘721 Synthesis can be improved to increase yield and/or increase reliability and/or increase scale and/or reduce byproducts. In some embodiments, the present disclosure provides such a synthesis, as detailed herein.
[0059] In some embodiments, the present disclosure provides a synthesis of terevalefim as depicted in Scheme 2:
Scheme 2
wherein X and R 1 are defined below and in classes and subclasses as described herein.
[0060] It will be appreciated that compounds described herein, eg, compounds in Scheme 2, may be provided and/or utilized in a salt form. For example, compounds which contain a basic nitrogen atom may form a salt with a suitable acid. Alternatively and/or additionally, compounds which contain an acidic moiety, such as a carboxylic acid group, may form a salt with a suitable base. Suitable counterions are well known in the art, eg, see generally, March ‘s Advanced Organic Chemistry: Reactions, Mechanisms, and Structure, MB Smith and J.
March, 5 th Edition, John Wiley & Sons, 2001. All forms of the compounds in Scheme 2 are contemplated by and within the scope of the present disclosure.
Step 2-1 of Scheme 2
[0061] Step 2-1 includes a condensation-elimination reaction between commercially available thiophene-2-carboxaldehyde (1.1) and acetone to provide an α,β-unsaturated ketone compound (2.1).
[0062] In some embodiments, the present disclosure provides a method comprising steps of:
(i) providing compound 1.1:
(ii) contacting compound 1.1 with acetone in the presence of a suitable base,
to compound provide 2.1:
////////

AS ON DEC2021 3,491,869 VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@amcrasto
/////////////////////////////////////////////////////////////////////////////
///////TEREVALEFIM, ANG-3777, SNV-003, Phase 3, Delayed graft function
C(=C\c1cccs1)/c2cc[nH]n2

NEW DRUG APPROVALS
ONE TIME
$10.00
RELACORILANT
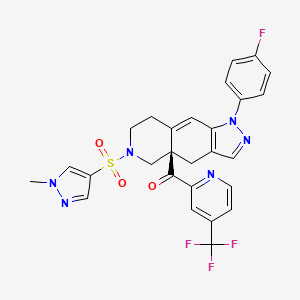
Relacorilant
- Molecular FormulaC27H22F4N6O3S
- Average mass586.561 Da
CAS 1496510-51-0
Fda approved 3/25/2026, Lifyorli
To treat platinum-resistant epithelial ovarian, fallopian tube, or primary peritoneal cancer after one to three prior systemic treatment regimens, at least one of which included bevacizumab |
Phase III, UNII-2158753C7E, 2158753C7E, CORT125134, CORT 125134
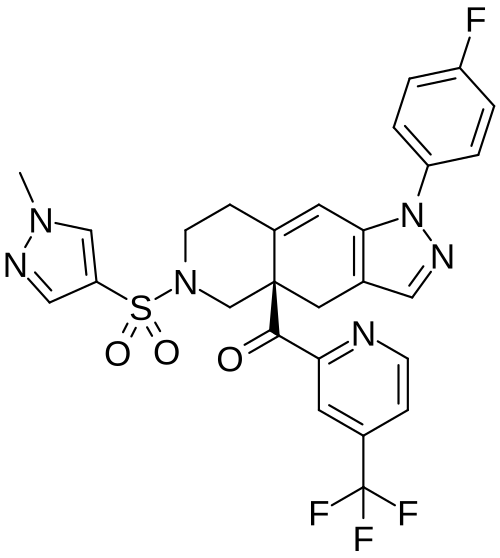
[(4aR)-1-(4-fluorophenyl)-6-(1-methylpyrazol-4-yl)sulfonyl-4,5,7,8-tetrahydropyrazolo[3,4-g]isoquinolin-4a-yl]-[4-(trifluoromethyl)pyridin-2-yl]methanone
релакорилант[Russian][INN]
ريلاكوريلانت[Arabic][INN]
瑞拉可兰[Chinese][INN]
- OriginatorCorcept Therapeutics
- ClassAntineoplastics; Fluorine compounds; Isoquinolines; Ketones; Organic sulfur compounds; Pyrazoles; Pyridines; Small molecules
- Mechanism of ActionGlucocorticoid receptor antagonists
- Orphan Drug StatusYes – Pancreatic cancer; Cushing syndrome
- Phase IIICushing syndrome; Ovarian cancer; Pancreatic cancer
- Phase IIFallopian tube cancer; Peritoneal cancer; Prostate cancer
- Phase I/IISolid tumours
- Phase IAdrenocortical carcinoma
Most Recent Events
- 09 Sep 2022Subgroup analysis efficacy data from a phase-II trial in Ovarian cancer presented at the 47th European Society for Medical Oncology Congress (ESMO-2022)
- 29 Jun 2022Phase-III clinical trials in Ovarian cancer (Combination therapy, Recurrent, Second-line therapy or greater) in USA (PO)
- 06 Jun 2022Corcept Therapeutics announces intentions to submit a NDA for Ovarian cancer
Relacorilant (developmental code name CORT-125134), sold under the brand name Lifyorli, is an antiglucocorticoid which is under development by Corcept Therapeutics for the treatment of Cushing’s syndrome.[1] It is also under development for the treatment of solid tumors and alcoholism.[1][2] The drug is a nonsteroidal compound and acts as an antagonist of the glucocorticoid receptor.[1] As of December 2017, it is in phase II clinical trials for Cushing’s syndrome and phase I/II clinical studies for solid tumors, while the clinical phase for alcoholism is unknown.[1]
The drug was approved by the USFDA in 2026 for the treatment of platinum-resistant ovarian cancer.[3]
Relacorilant is an orally available antagonist of the glucocorticoid receptor (GR), with potential antineoplastic activity. Upon administration, relacorilant competitively binds to and blocks GRs. This inhibits the activity of GRs, and prevents both the translocation of the ligand-GR complexes to the nucleus and gene expression of GR-associated genes. This decreases the negative effects that result from excess levels of endogenous glucocorticoids, like those seen when tumors overproduce glucocorticoids. In addition, by binding to GRs and preventing their activity, inhibition with CORT125134 also inhibits the proliferation of GR-overexpressing cancer cells. GRs are overexpressed in certain tumor cell types and promote tumor cell proliferation.
- OriginatorCorcept Therapeutics
- DeveloperCorcept Therapeutics; University of Chicago
- ClassAntineoplastics; Fluorine compounds; Isoquinolines; Ketones; Organic sulfur compounds; Pyrazoles; Pyridines; Small molecules
- Mechanism of ActionGlucocorticoid receptor antagonists
- Orphan Drug StatusYes – Pancreatic cancer; Ovarian cancer; Cushing syndrome
- RegisteredFallopian tube cancer; Ovarian cancer; Peritoneal cancer
- PreregistrationCushing syndrome
- Phase IIIAdenocarcinoma
- Phase IIProstate cancer
- DiscontinuedAdrenocortical carcinoma
- 27 Mar 2026Discontinued – Phase-I for Adrenocortical carcinoma (Inoperable/Unresectable, Late-stage disease, Metastatic disease, Combination therapy) in USA (PO), before March 2026 (Corcept Therapeutics pipeline, March 2026)
- 27 Mar 2026Corcept Therapeutics plans the phase II STELLA trial for Cervical cancer (Combination therapy, Second-line therapy or greater) in first quarter of 2026
- 25 Mar 2026Registered for Fallopian tube cancer (Combination therapy, Second-line therapy or greater) in USA (PO) – First global approval
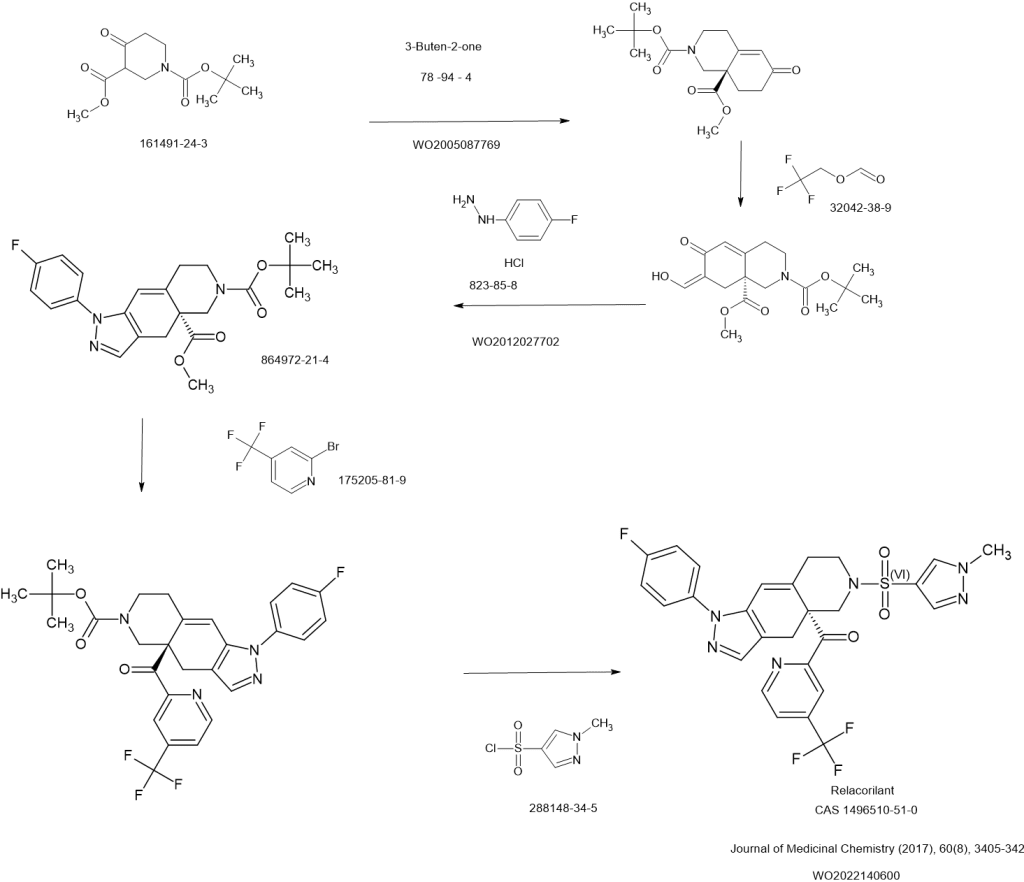
SCHEME
CLIP
https://europepmc.org/article/pmc/pmc8175224
Relacorilant (CORT125134)118) is being developed by Corcept Therapeutics, Inc. It is an orally active, high-affinity, selective antagonist of the glucocorticoid receptor that may benefit from the modulation of cortisol activity. In structural optimization, the introduction of a trifluoromethyl group to the 4-position on the pyridyl moiety was found to increase HepG2 tyrosine amino transferase assay potency by a factor of four. Relacorilant is currently being evaluated in a phase II clinical study in patients with Cushing’s syndrome.119)
2-Bromo-4-(trifluoromethyl)pyridine (17) prepared from (E)-4-ethoxy-1,1,1-trifluorobut-3-en-2-one is employed as a key intermediate for the preparation of relacorilant as shown in Scheme 31.120)
Scheme31. Synthesis of relacorilant.118)
118) H. Hunt, T. Johnson, N. Ray and I. Walters (Corcept Therapeutics, Inc.): PCT Int. Appl. WO2013/177559 (2013).
119) H. J. Hunt, J. K. Belanoff, I. Walters, B. Gourdet, J. Thomas, N. Barton, J. Unitt, T. Phillips, D. Swift and E. Eaton: Identification of the Clinical Candidate (R)-(1-(4-Fluorophenyl)-6-((1-methyl-1H-pyrazol-4-yl)sulfonyl)-4,4a,5,6,7,8-hexahydro-1H-pyrazolo[3,4-g]isoquinolin-4a-yl)(4-(trifluoromethyl)pyridin-2-yl)methanone (CORT125134): A Selective Glucocorticoid Receptor (GR) Antagonist. J. Med. Chem. 60, 3405–3421 (2017). [Abstract] [Google Scholar]
120) B. Lehnemann, J. Jung and A. Meudt (Archimica GmbH): PCT Int. Appl. WO 2007/000249 (2007).
PAPER
https://pubs.acs.org/doi/abs/10.1021/acs.jmedchem.7b00162
The nonselective glucocorticoid receptor (GR) antagonist mifepristone has been approved in the U.S. for the treatment of selected patients with Cushing’s syndrome. While this drug is highly effective, lack of selectivity for GR leads to unwanted side effects in some patients. Optimization of the previously described fused azadecalin series of selective GR antagonists led to the identification of CORT125134, which is currently being evaluated in a phase 2 clinical study in patients with Cushing’s syndrome.
PATENT
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2013177559


SYN

Cushing’s syndrome (CS) is a metabolic disorder caused by chronic hypercortisolism. CS is associated with cardiovascular, metabolic, skeletal and psychological dysfunctions and can be fatal if left untreated. The first-line treatment for all forms of CS is a surgery. However, medical therapy has to be chosen if surgical resection is not an option or is deemed ineffective. Currently available therapeutics are either not selective and have side effects or are only available as an injection (pasireotide).
ADVERTISEMENT
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
#MedicinalChemistry, #DrugDiscovery, #OrganicSynthesis, #ChemicalLibrary, #BuildingBlocks, #SARStudies, #ChemistryInnovation, #medchem, #Drugdevelopment, #Biotech, #Biotechnology, #AnaxLaboratories, #Pharma



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
- Heteroaryl-ketone fused azadecalin glucocorticoid receptor modulatorsPublication Number: EP-3590517-B1Priority Date: 2012-05-25Grant Date: 2021-03-17
- Heteroaryl-ketone fused azadecalin glucocorticoid receptor modulatorsPublication Number: US-2021369701-A1Priority Date: 2012-05-25
- Heteroaryl-ketone fused azadecalin glucocorticoid receptor modulatorsPublication Number: EP-4119561-B1Priority Date: 2012-05-25Grant Date: 2024-09-11
- PRMT5 inhibitors and uses thereofPublication Number: US-12448388-B2Grant Date: 2025-10-21
- KRAS G12D modulating compoundsPublication Number: US-12448400-B2Grant Date: 2025-10-21
- Heteroaryl-ketone fused azadecalin glucocorticoid receptor modulatorsPublication Number: AU-2013266110-C1Priority Date: 2012-05-25Grant Date: 2018-07-12
- Heteroaryl-ketone fused azadecalin glucocorticoid receptor modulatorsPublication Number: US-11576907-B2Priority Date: 2012-05-25Grant Date: 2023-02-14
- Heteroaryl-ketone fused azadecalin glucocorticoid receptor modulatorsPublication Number: CA-2872260-CPriority Date: 2012-05-25Grant Date: 2020-12-22
- Heteroaryl-ketone fused azadecalin glucocorticoid receptor modulatorsPublication Number: US-12226412-B2Priority Date: 2012-05-25Grant Date: 2025-02-18
- Heteroaryl-ketone fused azadecalin glucocorticoid receptor modulatorsPublication Number: US-2025099457-A1Priority Date: 2012-05-25
- Heteroaryl-ketone fused azadecalin glucocorticoid receptor modulatorsPublication Number: US-10973813-B2Priority Date: 2012-05-25Grant Date: 2021-04-13
- Heteroaryl-ketone fused azadecalin glucocorticoid receptor modulatorsPublication Number: EP-3851107-B1Priority Date: 2012-05-25Grant Date: 2022-10-19
- Heteroaryl-ketone fused azadecalin glucocorticoid receptor modulatorsPublication Number: PH-12014502584-B1Priority Date: 2012-05-25
- Heteroaryl-ketone fused azadecalin glucocorticoid receptor modulatorsPublication Number: EP-4434584-A2Priority Date: 2012-05-25
- Heteroaryl-ketone fused azadecalin glucocorticoid receptor modulatorsPublication Number: EP-4119561-A1Priority Date: 2012-05-25
References
- ^ Jump up to:a b c d “Relacorilant – Corcept Therapeutics – AdisInsight”.
- ^ Veneris JT, Darcy KM, Mhawech-Fauceglia P, Tian C, Lengyel E, Lastra RR, Pejovic T, Conzen SD, Fleming GF (2017). “High glucocorticoid receptor expression predicts short progression-free survival in ovarian cancer”. Gynecol. Oncol. 146 (1): 153–160. doi:10.1016/j.ygyno.2017.04.012. PMC 5955699. PMID 28456378.
External links
| Clinical data | |
|---|---|
| Other names | CORT-125134 |
| Routes of administration | By mouth |
| Drug class | Antiglucocorticoid |
| Identifiers | |
| showIUPAC name | |
| CAS Number | 1496510-51-0 |
| PubChem CID | 73051463 |
| ChemSpider | 57617720 |
| UNII | 2158753C7E |
| KEGG | D11336 |
| Chemical and physical data | |
| Formula | C27H22F4N6O3S |
| Molar mass | 586.57 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| showSMILES | |
| showInChI |
//////////////Relacorilant, Phase III , Orphan Drug, Cushing syndrome, Ovarian cancer, Pancreatic cancer, релакорилант , ريلاكوريلانت , 瑞拉可兰 , approvals 2026, fda 2026, CORT125134, CORT 125134
CN1C=C(C=N1)S(=O)(=O)N2CCC3=CC4=C(CC3(C2)C(=O)C5=NC=CC(=C5)C(F)(F)F)C=NN4C6=CC=C(C=C6)F
Tremelimumab
(Light chain)
DIQMTQSPSS LSASVGDRVT ITCRASQSIN SYLDWYQQKP GKAPKLLIYA ASSLQSGVPS
RFSGSGSGTD FTLTISSLQP EDFATYYCQQ YYSTPFTFGP GTKVEIKRTV AAPSVFIFPP
SDEQLKSGTA SVVCLLNNFY PREAKVQWKV DNALQSGNSQ ESVTEQDSKD STYSLSSTLT
LSKADYEKHK VYACEVTHQG LSSPVTKSFN RGEC
(Heavy chain)
QVQLVESGGG VVQPGRSLRL SCAASGFTFS SYGMHWVRQA PGKGLEWVAV IWYDGSNKYY
ADSVKGRFTI SRDNSKNTLY LQMNSLRAED TAVYYCARDP RGATLYYYYY GMDVWGQGTT
VTVSSASTKG PSVFPLAPCS RSTSESTAAL GCLVKDYFPE PVTVSWNSGA LTSGVHTFPA
VLQSSGLYSL SSVVTVPSSN FGTQTYTCNV DHKPSNTKVD KTVERKCCVE CPPCPAPPVA
GPSVFLFPPK PKDTLMISRT PEVTCVVVDV SHEDPEVQFN WYVDGVEVHN AKTKPREEQF
NSTFRVVSVL TVVHQDWLNG KEYKCKVSNK GLPAPIEKTI SKTKGQPREP QVYTLPPSRE
EMTKNQVSLT CLVKGFYPSD IAVEWESNGQ PENNYKTTPP MLDSDGSFFL YSKLTVDKSR
WQQGNVFSCS VMHEALHNHY TQKSLSLSPG K
(Disulfide bridge: L23-L88, L134-L194, L214-H139, H22-H96, H152-H208, H265-H325, H371-H429, H227-H’227, H228-H’228, H231-H’231, H234-H’234)

Fab fragment of tremelimumab (blue) binding CTLA-4 (green). From PDB entry 5GGV.
Tremelimumab
| Formula | C6500H9974N1726O2026S52 |
|---|---|
| CAS | 745013-59-6 |
| Mol weight | 146380.4722 |
FDA APPROVED2022/10/21, Imjudo
PEPTIDE, CP 675206
| Antineoplastic, Immune checkpoint inhibitor, Anti-CTLA4 antibody | |
| Disease | Hepatocellular carcinoma |
|---|
Tremelimumab (formerly ticilimumab, CP-675,206) is a fully human monoclonal antibody against CTLA-4. It is an immune checkpoint blocker. Previously in development by Pfizer,[1] it is now in investigation by MedImmune, a wholly owned subsidiary of AstraZeneca.[2] It has been undergoing human trials for the treatment of various cancers but has not attained approval for any.
Imjudo (tremelimumab) in combination with Imfinzi approved in the US for patients with unresectable liver cancer
PUBLISHED24 October 2022
24 October 2022 07:00 BST
Approval based on HIMALAYA Phase III trial results which showed single priming dose of Imjudo added to Imfinzi reduced risk of death by 22% vs. sorafenib
AstraZeneca’s Imjudo (tremelimumab) in combination with Imfinzi (durvalumab) has been approved in the US for the treatment of adult patients with unresectable hepatocellular carcinoma (HCC), the most common type of liver cancer. The novel dose and schedule of the combination, which includes a single dose of the anti-CTLA-4 antibody Imjudo 300mg added to the anti-PD-L1 antibody Imfinzi 1500mg followed by Imfinzi every four weeks, is called the STRIDE regimen (Single Tremelimumab Regular Interval Durvalumab).
The approval by the US Food and Drug Administration (FDA) was based on positive results from the HIMALAYA Phase III trial. In this trial, patients treated with the combination of Imjudo and Imfinzi experienced a 22% reduction in the risk of death versus sorafenib (based on a hazard ratio [HR] of 0.78, 95% confidence interval [CI] 0.66-0.92 p=0.0035).1 Results were also published in the New England Journal of Medicine Evidence showing that an estimated 31% of patients treated with the combination were still alive after three years, with 20% of patients treated with sorafenib still alive at the same duration of follow-up.2
Liver cancer is the third-leading cause of cancer death and the sixth most commonly diagnosed cancer worldwide.3,4 It is the fastest rising cause of cancer-related deaths in the US, with approximately 36,000 new diagnoses each year.5,6
Ghassan Abou-Alfa, MD, MBA, Attending Physician at Memorial Sloan Kettering Cancer Center (MSK), and principal investigator in the HIMALAYA Phase III trial, said: “Patients with unresectable liver cancer are in need of well-tolerated treatments that can meaningfully extend overall survival. In addition to this regimen demonstrating a favourable three-year survival rate in the HIMALAYA trial, safety data showed no increase in severe liver toxicity or bleeding risk for the combination, important factors for patients with liver cancer who also have advanced liver disease.”
Dave Fredrickson, Executive Vice President, Oncology Business Unit, AstraZeneca, said: “With this first regulatory approval for Imjudo, patients with unresectable liver cancer in the US now have an approved dual immunotherapy treatment regimen that harnesses the potential of CTLA-4 inhibition in a unique combination with a PD-L1 inhibitor to enhance the immune response against their cancer.”
Andrea Wilson Woods, President & Founder, Blue Faery: The Adrienne Wilson Liver Cancer Foundation, said: “In the past, patients living with liver cancer had few treatment options and faced poor prognoses. With today’s approval, we are grateful and optimistic for new, innovative, therapeutic options. These new treatments can improve long-term survival for those living with unresectable hepatocellular carcinoma, the most common form of liver cancer. We appreciate the patients, their families, and the broader liver cancer community who continue to fight for new treatments and advocate for others.”
The safety profiles of the combination of Imjudo added to Imfinzi and for Imfinzi alone were consistent with the known profiles of each medicine, and no new safety signals were identified.
Regulatory applications for Imjudo in combination with Imfinzi are currently under review in Europe, Japan and several other countries for the treatment of patients with advanced liver cancer based on the HIMALAYA results.
Notes
Liver cancer
About 75% of all primary liver cancers in adults are HCC.3 Between 80-90% of all patients with HCC also have cirrhosis.7 Chronic liver diseases are associated with inflammation that over time can lead to the development of HCC.7
More than half of patients are diagnosed at advanced stages of the disease, often when symptoms first appear.8 A critical unmet need exists for patients with HCC who face limited treatment options.8 The unique immune environment of liver cancer provides clear rationale for investigating medications that harness the power of the immune system to treat HCC.8
HIMALAYA
HIMALAYA was a randomised, open-label, multicentre, global Phase III trial of Imfinzi monotherapy and a regimen comprising a single priming dose of Imjudo 300mg added to Imfinzi 1500mg followed by Imfinzi every four weeks versus sorafenib, a standard-of-care multi-kinase inhibitor.
The trial included a total of 1,324 patients with unresectable, advanced HCC who had not been treated with prior systemic therapy and were not eligible for locoregional therapy (treatment localised to the liver and surrounding tissue).
The trial was conducted in 181 centres across 16 countries, including in the US, Canada, Europe, South America and Asia. The primary endpoint was overall survival (OS) for the combination versus sorafenib and key secondary endpoints included OS for Imfinzi versus sorafenib, objective response rate and progression-free survival (PFS) for the combination and for Imfinzi alone.
Imfinzi
Imfinzi (durvalumab) is a human monoclonal antibody that binds to the PD-L1 protein and blocks the interaction of PD-L1 with the PD-1 and CD80 proteins, countering the tumour’s immune-evading tactics and releasing the inhibition of immune responses.
Imfinzi was recently approved to treat patients with advanced biliary tract cancer in the US based on results from the TOPAZ-1 Phase III trial. It is the only approved immunotherapy in the curative-intent setting of unresectable, Stage III non-small cell lung cancer (NSCLC) in patients whose disease has not progressed after chemoradiotherapy and is the global standard of care in this setting based on the PACIFIC Phase III trial.
Imfinzi is also approved in the US, EU, Japan, China and many other countries around the world for the treatment of extensive-stage small cell lung cancer (ES-SCLC) based on the CASPIAN Phase III trial. In 2021, updated results from the CASPIAN trial showed Imfinzi plus chemotherapy tripled patient survival at three years versus chemotherapy alone.
Imfinzi is also approved for previously treated patients with advanced bladder cancer in several countries.
Since the first approval in May 2017, more than 100,000 patients have been treated with Imfinzi.
As part of a broad development programme, Imfinzi is being tested as a single treatment and in combinations with other anti-cancer treatments for patients with SCLC, NSCLC, bladder cancer, several gastrointestinal (GI) cancers, ovarian cancer, endometrial cancer, and other solid tumours.
Imfinzi combinations have also demonstrated clinical benefit in metastatic NSCLC in the POSEIDON Phase III trial.
Imjudo
Imjudo (tremelimumab) is a human monoclonal antibody that targets the activity of cytotoxic T-lymphocyte-associated protein 4 (CTLA-4). Imjudo blocks the activity of CTLA-4, contributing to T-cell activation, priming the immune response to cancer and fostering cancer cell death.
Beyond HIMALAYA, Imjudo is being tested in combination with Imfinzi across multiple tumour types including locoregional HCC (EMERALD-3), SCLC (ADRIATIC) and bladder cancer (VOLGA and NILE).
Imjudo is also under review by global regulatory authorities in combination with Imfinzi and chemotherapy in 1st-line metastatic NSCLC based on the results of the POSEIDON Phase III trial, which showed the addition of a short course of Imjudo to Imfinzi plus chemotherapy improved both overall and progression-free survival compared to chemotherapy alone.
AstraZeneca in GI cancers
AstraZeneca has a broad development programme for the treatment of GI cancers across several medicines spanning a variety of tumour types and stages of disease. In 2020, GI cancers collectively represented approximately 5.1 million new diagnoses leading to approximately 3.6 million deaths.9
Within this programme, the Company is committed to improving outcomes in gastric, liver, biliary tract, oesophageal, pancreatic, and colorectal cancers.
Imfinzi (durvalumab) is being assessed in combinations in oesophageal and gastric cancers in an extensive development programme spanning early to late-stage disease across settings.
The Company aims to understand the potential of Enhertu (trastuzumab deruxtecan), a HER2-directed antibody drug conjugate, in the two most common GI cancers, colorectal and gastric cancers. Enhertu is jointly developed and commercialised by AstraZeneca and Daiichi Sankyo.
Lynparza (olaparib) is a first-in-class PARP inhibitor with a broad and advanced clinical trial programme across multiple GI tumour types including pancreatic and colorectal cancers. Lynparza is developed and commercialised in collaboration with MSD (Merck & Co., Inc. inside the US and Canada).
AstraZeneca in immuno-oncology (IO)
Immunotherapy is a therapeutic approach designed to stimulate the body’s immune system to attack tumours. The Company’s immuno-oncology (IO) portfolio is anchored in immunotherapies that have been designed to overcome evasion of the anti-tumour immune response. AstraZeneca is invested in using IO approaches that deliver long-term survival for new groups of patients across tumour types.
The Company is pursuing a comprehensive clinical trial programme that includes Imfinzi as a single treatment and in combination with Imjudo (tremelimumab) and other novel antibodies in multiple tumour types, stages of disease, and lines of treatment, and where relevant using the PD-L1 biomarker as a decision-making tool to define the best potential treatment path for a patient.
In addition, the ability to combine the IO portfolio with radiation, chemotherapy, and targeted small molecules from across AstraZeneca’s oncology pipeline, and from research partners, may provide new treatment options across a broad range of tumours.
AstraZeneca in oncology
AstraZeneca is leading a revolution in oncology with the ambition to provide cures for cancer in every form, following the science to understand cancer and all its complexities to discover, develop and deliver life-changing medicines to patients.
The Company’s focus is on some of the most challenging cancers. It is through persistent innovation that AstraZeneca has built one of the most diverse portfolios and pipelines in the industry, with the potential to catalyse changes in the practice of medicine and transform the patient experience.
AstraZeneca has the vision to redefine cancer care and, one day, eliminate cancer as a cause of death.
////////

AS ON DEC2021 3,491,869 VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@amcrasto
/////////////////////////////////////////////////////////////////////////////
Mechanism of action
Tremelimumab aims to stimulate an immune system attack on tumors. Cytotoxic T lymphocytes (CTLs) can recognize and destroy cancer cells. However, there is also an inhibitory mechanism (immune checkpoint) that interrupts this destruction. Tremelimumab turns off this inhibitory mechanism and allows CTLs to continue to destroy the cancer cells.[3] This is immune checkpoint blockade.
Tremelimumab binds to the protein CTLA-4, which is expressed on the surface of activated T lymphocytes and inhibits the killing of cancer cells. Tremelimumab blocks the binding of the antigen-presenting cell ligands B7.1 and B7.2 to CTLA-4, resulting in inhibition of B7-CTLA-4-mediated downregulation of T-cell activation; subsequently, B7.1 or B7.2 may interact with another T-cell surface receptor protein, CD28, resulting in a B7-CD28-mediated T-cell activation unopposed by B7-CTLA-4-mediated inhibition.
Unlike Ipilimumab (another fully human anti-CTLA-4 monoclonal antibody), which is an IgG1 isotype, tremelimumab is an IgG2 isotype.[4][5]
Clinical trials
Melanoma
Phase 1 and 2 clinical studies in metastatic melanoma showed some responses.[6] However, based on early interim analysis of phase III data, Pfizer designated tremelimumab as a failure and terminated the trial in April 2008.[1][7]
However, within a year, the survival curves showed separation of the treatment and control groups.[8] The conventional Response Evaluation Criteria in Solid Tumors (RECIST) may underrepresent the merits of immunotherapies. Subsequent immunotherapy trials (e.g. ipilimumab) have used the Immune-Related Response Criteria (irRC) instead.
Mesothelioma
Although it was designated in April 2015 as orphan drug status in mesothelioma,[9] tremelimumab failed to improve lifespan in the phase IIb DETERMINE trial, which assessed the drug as a second or third-line treatment for unresectable malignant mesothelioma.[10][11]
Non-small cell lung cancer
In a phase III trial, AstraZeneca paired tremelimumab with a PD-L1 inhibitor, durvalumab, for the first-line treatment of non-small cell lung cancer.[12] The trial was conducted across 17 countries, and in July 2017, AstraZeneca announced that it had failed to meet its primary endpoint of progression-free survival.[13]
References
- ^ Jump up to:a b “Pfizer Announces Discontinuation of Phase III Clinical Trial for Patients with Advanced Melanoma”. Pfizer.com. 1 April 2008. Retrieved 5 December 2015.
- ^ Mechanism of Pathway: CTLA-4 Inhibition[permanent dead link]
- ^ Antoni Ribas (28 June 2012). “Tumor immunotherapy directed at PD-1”. New England Journal of Medicine. 366 (26): 2517–9. doi:10.1056/nejme1205943. PMID 22658126.
- ^ Tomillero A, Moral MA (October 2008). “Gateways to clinical trials”. Methods Find Exp Clin Pharmacol. 30 (8): 643–72. doi:10.1358/mf.2008.30.5.1236622. PMID 19088949.
- ^ Poust J (December 2008). “Targeting metastatic melanoma”. Am J Health Syst Pharm. 65 (24 Suppl 9): S9–S15. doi:10.2146/ajhp080461. PMID 19052265.
- ^ Reuben, JM; et al. (1 Jun 2006). “Biologic and immunomodulatory events after CTLA-4 blockade with tremelimumab in patients with advanced malignant melanoma”. Cancer. 106 (11): 2437–44. doi:10.1002/cncr.21854. PMID 16615096. S2CID 751366.
- ^ A. Ribas, A. Hauschild, R. Kefford, C. J. Punt, J. B. Haanen, M. Marmol, C. Garbe, J. Gomez-Navarro, D. Pavlov and M. Marsha (May 20, 2008). “Phase III, open-label, randomized, comparative study of tremelimumab (CP-675,206) and chemotherapy (temozolomide [TMZ] or dacarbazine [DTIC]) in patients with advanced melanoma”. Journal of Clinical Oncology. 26 (15S): LBA9011. doi:10.1200/jco.2008.26.15_suppl.lba9011.[permanent dead link]
- ^ M.A. Marshall, A. Ribas, B. Huang (May 2010). “Evaluation of baseline serum C-reactive protein (CRP) and benefit from tremelimumab compared to chemotherapy in first-line melanoma”. Journal of Clinical Oncology. 28 (15S): 2609. doi:10.1200/jco.2010.28.15_suppl.2609.[permanent dead link]
- ^ FDA Grants AstraZeneca’s Tremelimumab Orphan Drug Status for Mesothelioma [1]
- ^ “Tremelimumab Fails Mesothelioma Drug Trial”. Archived from the original on 2016-03-06. Retrieved 2016-03-06.
- ^ AZ’ tremelimumab fails in mesothelioma trial
- ^ “AstraZeneca’s immuno-oncology combo fails crucial Mystic trial in lung cancer | FierceBiotech”.
- ^ “AstraZeneca reports initial results from the ongoing MYSTIC trial in Stage IV lung cancer”.
///////////Tremelimumab, Imjudo, APPROVALS 2022, FDA 2022, PEPTIDE, CP 675206, Antineoplastic, Immune checkpoint inhibitor, Anti-CTLA4 antibody

NEW DRUG APPROVALS
ONE TIME
$10.00
MILADEMETAN
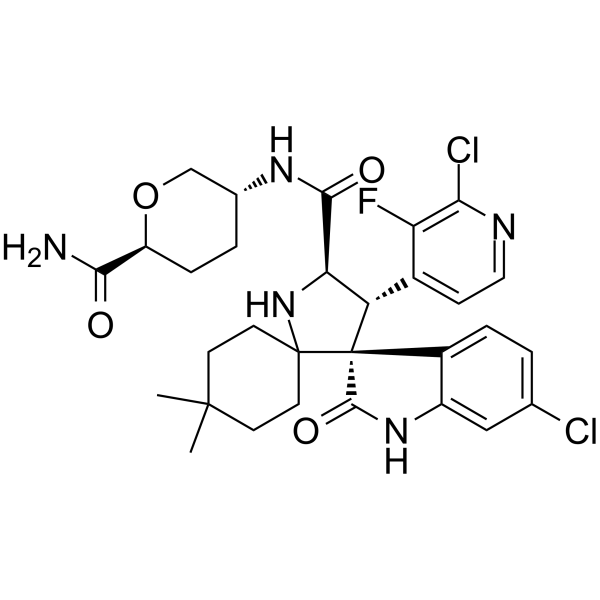
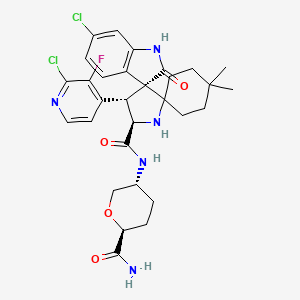
Milademetan
| Molecular Weight | 618.53 |
|---|---|
| Formula | C30H34Cl2FN5O4 |
| CAS No. | 1398568-47-2 |
Milademetan. hcl
Chemical Formula: C30H35Cl3FN5O4
Exact Mass: 617.1972
Molecular Weight: 654.99
Elemental Analysis: C, 55.01; H, 5.39; Cl, 16.24; F, 2.90; N, 10.69; O, 9.77
1398568-47-2 (free base) 1398569-75-9 (tosylate) 2095625-97-9 (tosylate hydrate) Milademetan HCl
DS3032b; DS-3032b; DS 3032b; DS3032; DS-3032; DS 3032; DS-3032b tosylate; Milademetan tosylate; Milademetan HCl
(3’R,4’S,5’R)-N-[(3R,6S)-6-carbamoyloxan-3-yl]-6”-chloro-4′-(2-chloro-3-fluoropyridin-4-yl)-4,4-dimethyl-2”-oxo-1”,2”-dihydrodispiro[cyclohexane-1,2′-pyrrolidine-3′,3”-indole]-5′-carboxamide hydrochloride
orphan drug, UNII:R3I80TLN7S, миладеметан , ميلاديميتان , 米拉美坦
(3’R,4’S,5’R)-N-((3R,6S)-6-Carbamoyltetrahydro-2H-pyran-3-yl)-6”-chloro-4′-(2-chloro-3-fluoro-4-pyridinyl)-4,4-dimethyl-2”-oxo-1”,2”-dihydrodispiro(cyclohexane-1,2′-pyrrolidine-3′,3”-indole)-5′-carboxamide
milademetan
rolontis
SPI-2012
Milademetan, also known as DS-3032b or DS-3032, is a potent and selective MDM2 inhibitor with potential antineoplastic activity. Upon oral administration, MDM2 inhibitor DS-3032b binds to, and prevents the binding of MDM2 protein to the transcriptional activation domain of the tumor suppressor protein p53. By preventing this MDM2-p53 interaction, the proteosome-mediated enzymatic degradation of p53 is inhibited and the transcriptional activity of p53 is restored. This results in the restoration of p53 signaling and leads to the p53-mediated induction of tumor cell apoptosis.
DS-3032 (Milademetan) is an orally available, potent and selective inhibitor of the p53-MDM2 (murine double minute 2) interaction. Milademetan binds to, and prevents the binding of MDM2 protein to the transcriptional activation domain of the tumor suppressor protein p53. Milademetan is 10-fold more potent than the first-generation inhibitor nutlin-3a. By preventing this MDM2-p53 interaction, the proteasome-mediated enzymatic degradation of p53 is inhibited and the transcriptional activity of p53 is restored. This results in the restoration of p53 signaling and leads to the p53-mediated induction of tumor cell apoptosis. DS-3032 is currently being evaluated in three phase 1 clinical trials for solid and hematological malignancies, including acute myeloid leukemia (AML), acute lymphocytic leukemia (ALL), chronic myeloid leukemia (CML) in blast phase, lymphoma and myelodysplastic syndrome (MDS).
- OriginatorRigel Pharmaceuticals
- DeveloperDaiichi Sankyo Inc; National Cancer Center Hospital East; Rain Therapeutics; University of Texas M. D. Anderson Cancer Center
- ClassAntineoplastics; Cyclohexanes; Indoles; Pyrrolidines; Small molecules
- Mechanism of ActionProto-oncogene protein c mdm2 inhibitors
- Orphan Drug StatusYes – Liposarcoma
- Phase IIILiposarcoma
- Phase IISarcoma; Solid tumours
- Phase I/IIAcute myeloid leukaemia
- Phase ILymphoma; Myelodysplastic syndromes
- PreclinicalMesothelioma
- No development reportedMultiple myeloma
- 10 Aug 2022Rain Therapeutics completes enrolment in phase-III clinical trials in Liposarcoma in (Inoperable/Unresectable, Metastatic disease, Second-line therapy or greater) in United Kingdom, Taiwan, Spain, Poland, South Korea, Italy, Hong Kong, Germany, Georgia, France, Canada, Belgium, Austria (PO) (NCT04979442)
- 09 Jun 2022Efficacy, adverse events and pharmacodynamics data from phase I/II trial in Acute myeloid leukemia presented at the 27th Congress of the European Haematology Association(EHA-2022)
- 04 May 2022Rain Therapeutics plans a phase I/II MANTRA-4 trial in Solid tumours (Combination therapy, Late-stage disease) in Second half of 2022
PATENT
WO2015033974

[Example 2]
Ethyl (3’R,4’S,5’R)-6”-chloro-4′-(3-chloro-2-fluorophenyl)-4,4-dimethyl-2”-oxo 1″,2″-dihydrodispiro[cyclohexane-1,2′-pyrrolidine-3′,3″-indole]-5′-carboxylate
[0202]
[Chem. 58]
[0203]
(3E/Z)-6-chloro-3-(3-chloro-2-fluorobenzylidene)-1,3-dihydro-2H-indol-2-one ( WO 2006/091646) (98.7) under nitrogen atmosphere mg), (R)-BINAP (12.1 mg, 0.019 mmol), CuOAc (2.0 mg, 0.016 mmol), 4,4-dimethylcyclohexanone (61.4 mg, 0.48 mmol), glycine ethyl ester. (39.5 μL, 0.39 mmol) and a solution of triethylamine (6.8 μL, 0.049 mmol) in N,N-dimethylacetamide (2.0 mL) were added and stirred at room temperature for 22 hours. Ethyl acetate (2 mL), water (1 mL), and 20% aqueous ammonium chloride solution (1 mL) were added to the reaction mixture, and the mixture was vigorously stirred to separate the organic layer. The aqueous layer was extracted twice with ethyl acetate (2 mL each) and all the organic layers were combined and then washed with water three times (5 mL each). The obtained organic layer was concentrated under reduced pressure, ethyl acetate (6 mL) and silica gel (500 mg) were added to the residue, and the silica gel was separated by filtration. The filtrate was concentrated under reduced pressure, ethanol (1.0 mL) was added to the residue, water (1 mL) was added dropwise, and the mixture was stirred overnight at room temperature. The precipitated solid was filtered and dried under reduced pressure at 40° C. to obtain the title compound (137 mg, yield 82%, 94% ee) as a solid.
1H NMR (500 MHz, CDCl3): δ = 0.67 (s, 3H), 0.91 (s, 3H), 1.10-1.19 (m, 2H), 1.17 (t, J=7.3 Hz, 3H), 1.25-1.33 (m, 1H), 1.44- 1.72 (m, 3H), 1.87-2.01 (m, 1H), 3.16 (s, 1H), 4.07-4.21 (m, 2H), 4.52 (d, J = 8.5 Hz, 1H), 4.83 (d, J = 8.5 Hz, 1H), 6.74 (d, J = 1.5Hz, 1H), 6.81-6.86 (m, 1H), 7.06 (dd, J = 8.3, 2.8 Hz, 1H), 7.10-7.16 (m, 1H), 7.37 (dd, J = 8.3, 1.8 Hz, 1H), 7.48-7.54 (m, 1H), 7.81 (s, 1H).
(HPLC conditions for optical purity determination)
カラム: CHIRALPAK OD-3R 4.6 × 150 mm, 3μm
Moving layer: 10mM Rinic acid buffer: MeCN = 40:60
Flow rate: 1.0 min/min
カラム Temperature: 40°C
Exhaust wavelength: 254 nm
Injection volume: 5 μL
Hold time: Labeling compound = 13.8 min, エナンチオマー= 12.9 min
[Example 11]
11-1) Effects of various asymmetric catalysts
[0230]
[Chem. 67]
[0231]
(3E/Z)-6-chloro-3-[(2-chloro-3-fluoropyridin-4-yl)methylene]-1,3-dihydro-2H-indol-2-one ( WO 2012 / 121361), 4,4-dimethylcyclohexanone (1.5 eq.), glycine ethyl ester (1.2 eq.), triethylamine (15 mol%) in THF solution (10 times the volume), separately, Lewis acid (5 mol%) , an asymmetric ligand (6 mol %) and THF (10 times the amount) were stirred for 1 hour under a nitrogen atmosphere, a catalyst solution prepared was added, and the mixture was stirred at room temperature for 12 to 16 hours. After that, the resulting trans1 compound ((ethyl (3′S,4′R,5′S)-6″-chloro-4′-(2-chloro-3-fluoropyridin-4-yl) -4,4-dimethyl-2”-oxo-1”,2”-dihydrodispiro[cyclohexane-1,2′-pyrrolidine-3′,3”-indole]-5′-carboxylate) Optical purity and HPLC yield were measured.
(HPLC conditions for measuring optical purity)
Column: CHIRALPAK OD-3R 4.6 × 150 mm, 3 µm
Mobile phase: 10 mM phosphoric acid buffer: MeCN = 40:60
Flow rate: 1.0 min/min
column Temperature: 40°C
Detection wavelength: 254 nm
Injection volume: 5 µL
Retention time: Title compound = 13.8 min, enantiomer = 12.9 min
Main results are shown in Table 1.
[0232]
[Table 1-1]
[Table 1-2]
[0233]
11-2) Effects of various solvents
[0234]
[Chem. 68]
[0235]
(3E/Z)-6-chloro-3-[(2-chloro-3-fluoropyridin-4-yl)methylene]-1,3-dihydro-2H-indol-2-one ( WO 2012 / 121361), 4,4-dimethylcyclohexanone (1.5 eq.), glycine ethyl ester (1.2 eq.), triethylamine (15 mol%), a solvent (10 times the amount), CuOAc (5 mol%), ( A catalyst solution prepared by stirring S)-BINAP (6 mol %) and a solvent (10 times the amount) under a nitrogen atmosphere for 1 hour was added, followed by stirring at room temperature for 21.5 hours. After that, by HPLC, the resulting trans2 compound (ethyl (3’S,4’R,5’S)-6”-chloro-4′-(2-chloro-3-fluoropyridin-4-yl)- HPLC of 4,4-dimethyl-2″-oxo-1″,2″-dihydrodispiro[cyclohexane-1,2′-pyrrolidine-3′,3″-indole]-5′-carboxylate) Yields and optical purities were determined.
Table 2 shows the main results.
[0236]
[Table 2]
11-3) Examination of Cu(I) Lewis acid
PATENT
WO2014038606
WO2014038606 CLICK HERE
Example 1
[0062]
[Chem.3]
[0063]
(3′R,4′S,5′R)-N-[(3R,6S)-6-carbamoyltetrahydro-2H-pyran-3-yl]-6″-chloro-4′-(2-chloro- 3-fluoropyridin-4-yl)-4,4-dimethyl-2″-oxo-1″,2″-dihydrodispiro[cyclohexane-1,2′-pyrrolidine-3′,3″-indole]-5 ‘
-Carboxamide The compound (35 mg, 0.24 mmol) obtained in Reference Example 2, Step 3 was added to a solution of the compound (100 mg, 0.20 mmol) obtained in Step 3 of Reference Example 1 in N,N-dimethylformamide (4 ml). , triethylamine (0.04 ml, 0.30 mmol), 1-hydroxybenzotriazole (27 mg, 0.20 mmol) and 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide hydrochloride (46 mg, 0.24 mmol) were added. , and stirred for 1 hour at 50° C. After allowing to cool, the reaction solution was diluted with ethyl acetate, washed successively with water, saturated aqueous sodium hydrogencarbonate solution and saturated brine, and dried over anhydrous sodium sulfate. After evaporating the solvent under reduced pressure, the residue was purified by NH-silica gel column chromatography [chloroform:methanol=50:1 (v/v)]. After stirring for 24 hours at rt, the solvent was distilled off under reduced pressure to obtain 94 mg (76%) of the title compound as a solid.1H
– NMR (400 MHz, CDCl3 ) .) δ: 0.68 (3H, s), 0.95 (3H, s), 1.11-1.27 (2H, m), 1.35-1.81 (8H, m), 2.10-2.17 (1H, m), 2.25-2.32 (1H, m), 3.15(1H,t,J=10.5Hz), 3.27(1H,br s), 3.80(1H,dd,J=11.0,2.3Hz), 3.85-3.95(1H,m), 4.13(1H, ddd,J=10.8,4.5,1.3Hz),4.44(1H,d,J=9.2Hz),4.64(1H,d,J=9.2Hz),5.46(1H,d,J=3.7Hz),6.49( 1H,d,J=3.7Hz), 6.74(1H,d,J=1.8Hz), 7.07(1H,dd,J=8.2,1.8Hz), 7.31(1H,dd,J=8.2,2.3Hz), 7.48-7.52(2H,m),7.62(1H,s),8.05(1H,d,J=5.5Hz).MS
(ESI)m/z:618(M+H) +
Reference example 1
[0087]
[Chem.4]
[0088]
[Step 1] (3E/Z)-6-chloro-3-[(2-chloro-3-fluoropyridin-4-yl)methylene]-1,3-dihydro-2H-indol-2-one
6-chloro -1,3-dihydro-2H-indol-2-one (2.20 g, 13.11 mmol) and 2-chloro-3-fluoroisonicotinaldehyde (2.20 g, 13.8 mmol) in methanol (130 ml). , N,N-diisopropylethylamine (0.46 ml, 2.63 mmol) was added, and the mixture was heated under reflux for 16 hours. After standing to cool, the precipitate was collected by filtration, washed with cold methanol and dried to obtain 3.37 g (83%) of the title compound as a solid.
MS(APCI) m/z: 309(M+H) + .
[0089]
[Step 2] (3′S,4′R,7′S,8′S,8a′R)-6″-chloro-8′-(2-chloro-3-fluoropyridin-4-yl)-4 ,4-dimethyl-3′,4′-diphenyl-3′,4′,8′,8a′-tetrahydro-1′H-dispiro[cyclohexane-1,6′-pyrrolo[2,1-c][1 ,4]oxazine-7′,3″-indole]-1′,2″(1″H)
-dione Under a nitrogen atmosphere, the compound obtained in Step 1 (1.86 g, 6.00 mmol), (5R,6S )-5,6-diphenylmorpholin-2-one (1.67 g, 6.60 mmol) and 4,4-dimethylcyclohexanone (0.83 g, 6.60 mmol) in tetrahydrofuran (30 ml) was added with diethyl boron trifluoride. An ether complex (0.15 ml, 1.20 mmol) and molecular sieve 4A (powder) (3 g) were added, and the mixture was heated and stirred at 70° C. for 7 days. After allowing to cool, insoluble matter was removed by filtration through celite, and the filtrate was washed with saturated brine and dried over anhydrous sodium sulfate. The solvent was distilled off under reduced pressure and purified by silica gel column chromatography [n-hexane:ethyl acetate=4:1→1:1 (v/v)] to obtain 3.39 g (84%) of the title compound as a solid. rice field.
1 H-NMR (400 MHz, CDCl3) δ: 0.21 (3H, s), 0.53 (3H, s), 0.89-1.08 (3H, m), 1.28-1.43 (3H, m), 1.73-1.81 (1H, m), 2.23-2.33 (1H, m), 4.58 (1H, d, J = 11.0Hz), 4.86 (1H, d, J = 3.2Hz), 5.31 (1H, d, J = 11.0Hz), 6.25 (1H, d, J = 8.3Hz) ,6.67(1H,dd,J=8.3,1.8Hz),6.72-6.77(2H,m),6.93(1H,d,J=1.8Hz),7.04-7.17(6H,m),7.18-7.25(3H ,m),7.79(1H,t,J=4.6Hz),7.99(1H,s),8.29(1H,d,J=5.0Hz).MS
(APCI)m/z:670(M+H) + .
[0090]
[Step 3] (4′S,5′R)-6″-chloro-4′-(2-chloro-3-fluoropyridin-4-yl)-4,4-dimethyl-2″-oxo-1″ ,2″-dihydrodispiro[cyclohexane-1,2′-pyrrolidine-3′,3″-indole]-5′-carboxylic acid
The compound obtained in step 2 (630 mg, 0.94 mmol) was treated with acetonitrile (10 ml). Dissolve in water (4 ml), add potassium carbonate (130 mg, 0.94 mmol) and heat under reflux for 16 hours at 85° C. After allowing to cool, add anhydrous magnesium sulfate (113 mg, 0.94 mmol) and stir at room temperature for 15 minutes. After extraction with ethyl acetate, the organic layer was washed with saturated brine and dried over anhydrous magnesium sulfate. (2-chloro-3-fluoropyridin-4-yl)-1′-[(1R,2S)-2-hydroxy-1,2-diphenylethyl]-4,4-dimethyl-2″-oxo-1″ ,2″-dihydrodispiro[cyclohexane-1,2′-pyrrolidine-3′,3″-indole]-5′-carboxylic acid (650 mg, 100%) was obtained as a solid [MS (ESI) m/z :688(M+H) +]. The resulting carboxylic acid (650 mg, 0.94 mmol) was dissolved in methanol (30 ml) and water (8 ml), and diammonium cerium (IV) nitrate (1.55 g, 2.82 mmol) was added under ice-cooling. Stir at room temperature for 30 minutes. Potassium carbonate (780 mg, 5.64 mmol) was added under ice-cooling, and the mixture was stirred at the same temperature for 1 hour. After removing the insoluble matter by filtration through celite, the filtrate was concentrated under reduced pressure, water was added to the resulting residue, and the mixture was extracted with ethyl acetate. The organic layer was washed with saturated brine and dried over anhydrous sodium sulfate. The solvent was distilled off under reduced pressure, and the resulting residue was purified by silica gel column chromatography [chloroform:methanol=20:1→4:1 (v/v)] to obtain 152 mg (33%) of the title compound as a solid. .
1 H-NMR (500 MHz, CD 3 OD) δ: 0.74 (3H, s), 0.9 (3H, s), 1.29-1.44 (2H, m), 1.48-1.58 (2H, m), 1.64-1.76 (1H ,m),1.94-2.02(1H,m),2.11(1H,ddd,J=14.0,14.0,4.0Hz),2.43-2.53(1H,m),5.07(1H,d,J=10.3Hz), 5.32(1H,d,J=10.3Hz),6.84(1H,d,J=1.7Hz),7.16(1H,dd,J=8.3,2.0Hz),7.63(1H,dd,J=8.0,2.3Hz) ),7.75(1H,t,J=5.2Hz),8.15(1H,d,J=5.2Hz).
MS(ESI)m/z:492(M+H) + .
[0091]
Reference example 2
[0092]
[Chem.5]
[0093]
[Step 1] Methyl 2,6-anhydro-3,4,5-trideoxy-5-(dibenzylamino)-L-erythro
-hexonate 2,6-anhydro-3,4,5-trideoxy-5-( dibenzylamino)-L-erythro-hexonate methyl 2,6-anhydro-3,4,5-trideoxy-5-(dibenzylamino)-L-erythro-hexonate (1.60 g, 4.70 mmol) was The mixture was dissolved in methanol (30 ml), 1N aqueous sodium hydroxide solution (10 ml) was gradually added under ice-cooling, and the mixture was stirred at room temperature for 3 hours. Dowex 50W-X8 was added to the reaction mixture to adjust the pH to 5 to 6, insoluble materials were removed by filtration, and the filtrate was concentrated under reduced pressure to obtain 1.7 g (100%) of the title compound as a solid.
1 H-NMR (400 MHz, CDCl 3 ) δ: 1.18-1.26(1H,m), 1.36-1.48(1H,m), 1.79-1.97(2H,m), 2.62(1H,t,J=11.0Hz) ,3.18(1H,t,J=10.4Hz),3.40(1H,d,J=11.5Hz),3.51-3.61(4H,m),3.90-3.99(1H,m),7.12-7.38(10H,m ).
MS(ESI)m/z:326(M+H) + .
[0094]
[Step 2] (2S,5R)-5-(dibenzylamino)tetrahydro-2H-pyran-2-carboxamide
The compound (870 mg, 2.67 mmol) obtained in Step 1 above was dissolved in N,N-dimethylformamide (30 ml). 1-hydroxybenzotriazole (361 mg, 2.67 mmol) and 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide hydrochloride (614 mg, 3.20 mmol) were added and stirred at room temperature for 15 minutes. Ammonium chloride (285 mg, 5.44 mmol) and N,N-diisopropylethylamine (1.86 ml, 10.7 mmol) were added and stirred at room temperature for 8 hours. After diluting with ethyl acetate, the organic layer was washed with saturated aqueous sodium hydrogencarbonate solution and saturated brine in that order, and dried over anhydrous sodium sulfate. The solvent was removed under reduced pressure to give 495 mg (57%) of the title compound as a solid.
1 H-NMR (400 MHz, CDCl 3 ) δ: 1.35-1.45 (1H, m), 1.60-1.70 (1 H, m), 2.10-2.18 (1 H, m), 2.21-2.28 (1 H, m), 2.76 ( 1H,tt,J=11.4,4.0Hz),3.44(1H,t,J=10.9Hz),3.67(4H,q,J=14.2Hz),3.71-3.73(1H,m),4.04(1H,dq ,J=11.0,2.1Hz),5.35(1H,s),6.40(1H,s),7.21-7.36(10H,m).MS
(ESI)m/z:325(M+H) + .
[0095]
[Step 3] (2S,5R)-5-aminotetrahydro-2H-pyran-2-carboxamide
The compound (490 mg, 1.51 mmol) obtained in Step 2 above was dissolved in ethanol (10 ml) and treated with 20% palladium hydroxide. (100 mg) was added, and the mixture was stirred at room temperature for 16 hours under a hydrogen atmosphere. After removing the catalyst by filtration through celite, the filtrate was distilled off under reduced pressure and dried to obtain 215 mg (99%) of the title compound as a solid.
1 H-NMR (400 MHz, DMSO-d 6 ) δ: 1.11-1.22(1H,m), 1.25-1.35(1H,m), 1.83-1.91(2H,m), 2.51-2.60(1H,m), 2.90(1H,t,J=10.5Hz),3.52(1H,d,J=11.9Hz),
3.78-3.84 (1H,m),6.99(1H,br s),7.09(1H,br s). (ESI) m/z: 145(M+H) + .
PATENT
WO2012121361
PATENT
WO2015033974
PAPER
https://pubs.acs.org/doi/10.1021/acs.oprd.2c00192
Abstract

Herein, we report the structure and synthesis of the potent MDM2-p53 inhibitor BI-0282. The complex spirooxindole scaffold bearing four stereocenters embedded in a rigid polycyclic ring-system was effectively prepared on a multi-gram scale in only five synthesis steps employing a three-component 1,3-dipolar cycloaddition and a late-stage Davis–Beirut reaction as key steps.
Compound 1
Intermediate 10 (28.8 g, 44.8 mmol) is dissolved in isopropanol (300 mL) and a solution of potassium hydroxide (39.0 g, 694.9 mmol) in water (95 mL) is slowly added. After stirring for 16 h at ambient temperature, the solvents are partially removed under reduced pressure. The residue is diluted with ethyl acetate and treated with a diluted aqueous solution of citric acid. After extraction of the aqueous layer with ethyl acetate, the organic layers are combined, dried with sodium sulfate, and the solvent is removed under reduced pressure. Purification by normal phase column chromatography using dichloromethane and methanol as solvents yields rac-1 (25.8 g, 43.5 mmol) in 70% yield as an amorphous white solid.
Chiral SFC and subsequent purification by reversed phase column chromatography using acetonitrile and methanol as solvents furnishes 1 (BI-0282).
Rac-1 (60 g, 93,3 mmol) was separated by chiral SFC and reversed phase column chromatography to obtain 1 (24.4 g, 40,0 mmol, 43%) as an amorphous white solid.
Chiral HPLC (CHIRALPAK, heptane/isopropanol/trifluoroacetic acid = 70/30/0.1, flow rate 1.0 mL/min, I = 240 mM) tR = 7.8 min (1), and 11.1 min (ent-1). Preparative SFC (CHIRALPAK, carbon dioxide/(isopropanol + 1% diethylamine) = 70/30, flow rate 300 g/min, I = 290 nM).
1H NMR (500 MHz, DMSO-d6): δ 12.64 (br s, 1H), 10.29 (s, 1H), 7.67 (s, 1H), 7.47 (d, J = 8.83 Hz, 2H), 7.29–7.36 (m, 1H), 7.26 (d, J = 7.88 Hz, 1H), 7.21 (dd, J = 1.26, 8.83 Hz, 1H), 7.12 (t, J = 8.04 Hz, 1H), 6.92 (dd, J = 1.89, 7.88 Hz, 1H), 6.48 (d, J = 1.89 Hz, 1H), 5.86 (t, J = 9.14 Hz, 1H), 4.59–4.68 (m, 1H), 4.52 (dd, J = 7.88, 11.35 Hz, 1H), 4.23–4.32 (m, 1H), 4.20 (d, J = 10.09 Hz, 1H), 2.27 (dd, J = 7.57, 13.08 Hz, 1H), 2.13 (dd, J = 5.83, 13.08 Hz, 1H), 0.47–0.62 (m, 1H), 0.26–0.37 (m, 1H), 0.11–0.20 (m, 1H), −0.04 to 0.04 (m, 1H), −0.25 (s, 1H).
13C{1H} NMR (125 MHz, DMSO-d6): δ 177.5, 168.1, 156.1 (d, 1JC,F = 248.7 Hz), 146.3, 145.3, 144.0, 134.1, 130.3, 129.7, 129.5, 126.8, 126.7, 125.4 (d, 3JC,F = 4.4 Hz), 123.5 (d, 2JC,F = 13.2 Hz), 122.5, 120.0, 119.9, 119.7 (d, 2JC,F = 18.3 Hz), 118.7, 110.0, 107.3, 76.4, 69.2, 57.5, 56.8, 54.2, 51.2, 11.6, 5.5, 4.1.
HRMS (ESI) m/z: [M + H]+ calcd for C30H24Cl2FN4O4, 593.1153; found, 593.1165.
////////

AS ON DEC2021 3,491,869 VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@amcrasto
/////////////////////////////////////////////////////////////////////////////
Milademetan is under investigation in clinical trial NCT02319369 (Safety, Tolerability and Pharmacokinetics of Milademetan Alone and With 5-Azacitidine (AZA) in Acute Myelogenous Leukemia (AML) or High-Risk Myelodysplastic Syndrome (MDS)).
- [1]. ARYL SULFONOHYDRAZIDES. WO 2017069289 A1.[2]. M.M. Gounder, et al. Milademetan, an oral MDM2 inhibitor, in well-differentiated/dedifferentiated liposarcoma: results from a phase 1 study in patients with solid tumors or lymphomas. European Journal of Cancer 138S2 (2020) S1–S62.[3]. Li, Yangbing, et al. Development of novel PROTAC Small-Molecule Degraders of MDM2 Protein and Peptidomimetic Inhibitors Targeting WDR5-MLL1 Protein-Protein Interaction.[4]. Viktor Arnhold, et al. Reactivating TP53 signaling by the novel MDM2 inhibitor DS-3032b as a therapeutic option for high-risk neuroblastoma. ncotarget. 2018 Jan 5; 9(2): 2304–2319.
/////////Milademetan, DS3032b, DS-3032b, DS 3032b, DS3032, DS-3032, DS 3032, DS-3032b tosylate, Milademetan tosylate, Milademetan HCl, orphan drug, UNII:R3I80TLN7S, миладеметан , ميلاديميتان , 米拉美坦
CC1(C)CCC2(CC1)N[C@H]([C@H](C1=C(F)C(Cl)=NC=C1)[C@]21C(=O)NC2=CC(Cl)=CC=C12)C(=O)N[C@@H]1CC[C@H](OC1)C(N)=O

NEW DRUG APPROVALS
ONE TIME
$10.00
Daxibotulinumtoxin A

Daxibotulinumtoxin A
FDA APPROVED 2022 2022/9/7, Daxxify
| Formula | C6708H10359N1729O1995S32 |
|---|---|
| CAS | 93384-43-1 |
| Mol weight | 148171.4934 |
Daxibotulinumtoxin A-lanm
Treatment of galbellar lines, cervical dystonia, lateral canthal lines, migraine headaches and hyperhidrosis
- DeveloperRevance Therapeutics; Shanghai Fosun Pharmaceutical
- ClassAnalgesics; Anti-inflammatories; Antiarrhythmics; Antidepressants; Antimigraines; Antipruritics; Antispasmodics; Bacterial proteins; Bacterial toxins; Botulinum toxins; Eye disorder therapies; Foot disorder therapies; Muscle relaxants; Skin disorder therapies; Urologics; Vascular disorder therapies
- Mechanism of ActionAcetylcholine inhibitors; Glutamate antagonists; Membrane transport protein modulators; Neuromuscular blocking agents
- Orphan Drug StatusYes – Torticollis
- RegisteredGlabellar lines
- Phase IIITorticollis
- Phase IIMuscle spasticity
- No development reportedSkin disorders
- DiscontinuedPlantar fasciitis
- 19 Sep 2022Efficacy data from a phase IIa FHL trials in Glabellar-lines (crow’s feet) released by Revance
- 19 Sep 2022Updated efficacy and safety data from the phase III SAKURA 1, SAKURA 2 and SAKURA 3 trials in Glabellar lines released by Revance Therapeutics
- 18 Sep 2022Updated efficacy and safety data from the phase III SAKURA 1, SAKURA 2 and SAKURA 3 trials in Glabellar lines released by Revance Therapeutics
////////

AS ON DEC2021 3,491,869 VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@amcrasto
/////////////////////////////////////////////////////////////////////////////
DAXI Impresses; Approaches FDA Approval
March 2, 2021
Dermatology Times, Dermatology Times, February 2021 (Vol. 42, No. 2), Volume 42, Issue 2
The investigational neuromodulator, evaluated in clinical trials as a treatment for glabellar lines and as a combined therapy for glabellar, dynamic forehead, and lateral canthal lines, is quickly nearing an approval by the FDA.
The long-awaited, longer-lasting neuromodulator drug candidate DaxibotulinumtoxinA for Injection (DAXI), a botulinum toxin type A formulated with a novel peptide excipient, may be nearing FDA approval.
In mid-December 2020, Revance Therapeutics shared results from its phase 2 upper facial lines study (NCT04259086),1 in which investigators looked at DAXI for combined treatment of glabellar, dynamic forehead, and lateral canthal lines.
The authors reported on a multicenter study of 48 patients enrolled to receive 40, 32, and 48 U of DAXI for injection in the glabellar complex, forehead, and lateral canthal areas, respectively. At week 4, nearly 92% of patients achieved an Investigator Global Assessment (IGA) score indicating no or mild wrinkle severity with maximum contraction on their lateral canthal lines. Nearly 96% achieved similar results on their forehead and glabellar lines at week 4.
Wrinkle severity returned to baseline at a median of 7.6 months post treatment, according to the phase 2 study findings.
The treatment was well tolerated in all upper facial regions. The most common adverse event (AE) was injection site erythema, which occurred in 6.3% of patients. The authors reported no eyelid or brow ptosis.
This was Revance’s first DAXI study on not just glabellar lines but also on forehead and periocular lines, or crow’s-feet, according to Jeffrey S. Dover, MD, FRCPC, a phase 2 study investigator and a dermatologist at SkinCare Physicians in Chestnut Hill, Massachusetts. “I think this is yet more evidence that the Revance neuromodulator produces an impressive effect on lines of negative facial expression and lasts longer than any of the other neuromodulators approved by the FDA thus far,” said Dover.
Dermatologic Surgery published 2 papers on the investigational neuromodulator in January 2021. In one study,2 investigators evaluated the use of up to 3 DAXI treatments for moderate or severe glabellar lines. They focused on data from SAKURA 1 and 2 (NCT03014622 and NCT03014635), two identical phase 3, open label, multicenter studies in which investigators evaluated single and repeat treatment of the glabellar lines with 40 U of DAXI.
The authors reported on safety results for nearly 2700 patients, including 882 who received a second treatment and 568 who got DAXI a third time. Treatment-related AEs, which were generally mild and resolved, occurred in 17.8% of patients. Eyelid ptosis occurred in 0.9% of treatments.
Investigators of 2 other studies3,4 focused on DAXI efficacy among nearly 2700 subjects enrolled in Revance’s preceding pivotal trials. Participants received repeat treatments when they returned to baseline on the IGA–Frown Wrinkle Severity (FWS) and IGA–Patient Frown Winkle Severity (PFWS) scales at 12 weeks and up to 36 weeks after treatment.
More than 96% of patients achieved no or mild severity in glabellar wrinkles on the IGA- FWS scale after each of the 3 treatments, with peak responses between weeks 2 to 4, and about one-third or more saw no or mild severity at week 24. Response rates reached highs of 92% or more at weeks 2 to 4 on the IGA-PFWS scale.
“The median duration for return to moderate or severe severity was 24 weeks,” the authors said. “If approved, I believe daxibotulinumtoxinA will change the landscape of neuromodulators significantly. The approved ones all last 3 months. They all give nice results and have few adverse effects,” Dover said.
He and other investigators have seen no rise in AEs, and those that did occur lasted no longer than those of Botox, he said.
Revance appears to be preparing for approval. The company announced on December 22, 2020, that it has a strategic commercial manufacturing agreement with Ajinomoto Bio- Pharma Services for the supply of DAXI.5
As of November 24, 2020, the FDA had deferred a decision on the neuromodulator because the required factory inspection could not be conducted due to travel restrictions related to coronavirus disease 2019.6 The FDA did not indicate any other issues.
References:
- Green JB, Mariwalla K, Coleman K, et al. A large, open-label, phase 3 safety study of DaxibotulinumtoxinA for Injection in glabellar lines: a focus on safety from the SAKURA 3 study. Derm Surg. 2021;47(1):42-46. doi:10.1097/DSS.0000000000002463
- Carruthers JD, Jean D, Fagien S, et al; SAKURA 1 and SAKURA 2 Investigator Group. DaxibotulinumtoxinA for Injection for the treatment of glabellar lines: results from each of two multicenter, randomized, double-blind, placebo-controlled, phase 3 studies (SAKURA 1 and SAKURA 2).Plast Reconstr Surg. 2020;1(145):45-58.doi: 10.1097/PRS.0000000000006327
- Fabi SG, Cohen JL, Green LJ, et al. DaxibotulinumtoxinA for Injection for the treatment of glabellar lines: efficacy results from SAKURA 3, a large, open-label, phase 3 safety study. Derm Surg. 2021;47(1):48-54. doi:10.1097/DSS.0000000000002531
- https://investors.revance.com/news-releases/news-release-details/ajinomoto-bio-pharma-services-and-revance-therapeutics-announce. December 22, 2020. Accessed January 15, 2021.
- FDA defers approval of DaxibotulinumtoxinA for Injection in glabellar lines due to COVID-19 related travel restrictions impacting manufacturing site inspection. News release. Revance Therapeutics, Inc. November 25, 2020. Accessed January 13, 2021. https://www.businesswire.com/news/home/20201125005462/en/FDA-Defers-Approval-DaxibotulinumtoxinA-Injection-Glabellar-Lines
///////////Daxibotulinumtoxin A, FDA 2022, APPROVALS 2022, DAXI, Daxibotulinumtoxin-A, DaxibotulinumtoxinA for Injection, daxibotulinumtoxinA-lanm, DAXXIFY, RT-002, Orphan Drug

NEW DRUG APPROVALS
ONE TIME
$10.00

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO



