| Patent | Submitted | Granted |
|---|---|---|
| Compound useful for the treatment of degenerative and inflammatory diseases [US8088764] | 2010-12-30 | 2012-01-03 |
| NOVEL COMPOUNDS USEFUL FOR THE TREATMENT OF DEGENERATIVE AND INFLAMMATORY DISEASES [US2011190260] | 2011-08-04 |
Home » Uncategorized (Page 88)
Category Archives: Uncategorized
AN IMPROVED PROCESS FOR THE PREPARATION OF DOLUTEGRAVIR

 Aurobindo Pharma MD and CEO N. Govindarajan at a company research centre. “It [the transition] is purely driven by the need to get more into areas where there is scope for better profit margins,
Aurobindo Pharma MD and CEO N. Govindarajan at a company research centre. “It [the transition] is purely driven by the need to get more into areas where there is scope for better profit margins,
Dolutegravir (I) is chemically known as (4/?,12aS)-N-[(2,4-difluorophenyl)methyl]-3,4,6,8,12,12a-hexahydro-7-hydroxy-4-methyl-6,8-dioxo-2//-pyrido[r,2′:4,5]pyrazino[2,l-b][l,3]oxazine-9-carboxamide. Dolutegravir is a human immunodeficiency virus type 1 (HIV-1) integrase strand transfer inhibitor (INSTI) indicated in combination with other antiretroviral agents for the treatment of HIV-1 infection. Dolutegravir is being marketed under the trade name Tivicay®. US 8,129,385 disclosed Dolutegravir or its pharmaceutically acceptable salts thereof. US ‘385 also discloses a process for the preparation of Dolutegravir (I). The process involves the condensation of 5-benzyloxy-4-hydroxy-6-hydroxymethyl nicotinic acid (II) with 2,4-difluorobenzylamine (III) to produce 5-benzyloxy-N-(2,4-difluorobenzyl)-4-hydroxy-6-hydroxymethyl nicotinic acid amide (IV), which is further under goes oxidation using manganese dioxide (Mn02) to produce 5-benzyloxy-N-(2,4-difluorobenzyl)-6-formyl-4-hydroxy-nicotinic acid amide (V). This amide compound (V) is reacted with sodium chlorite (NaClCh) to produce 3-benzyloxy-5-(2,4-difluorobenzylcarbamoyl)-4- hydroxy-pyridine-2-carboxylic acid (VI), which is further treated with methanol (MeOH) to produce 3-benzyloxy-5-(2,4-difluorobenzyl)-4-hydroxy-pyridine-2-carboxylic acid methyl ester (VII).

The methyl ester compound (VII) is reacted with 3-bromopropene to produce l-allyl-3-benzyloxy-5-(2,4-difluorobenzyl)-4-oxo-l,4-dihydro-pyridine-2- carboxylic acid methyl ester (VIII), which is further reacted with potassium osmate dihydrate (K2OSO4.2H2O) to produce 3-benzyloxy-5-(2,4-difluorobenzylcarbamoyl)-4-oxo-l-(2-oxo-ethyl)-l,4-dihydropyridine-2-carboxylic acid methyl ester (IX). The compound (IX) is reacted with (R)-3-amino-l-butanol (X) to produce benzyloxy Dolutegravir (XI), which is deprotected by treating with TFA to produce Dolutegravir (I). The process is as shown in scheme-I below:
The major disadvantage with the above prior-art process is that it involves large no of steps and tedious work-up procedures to isolate the required product. This results a longer period of time cycle is required to produce Dolutegravir (I), which in turn renders the process more costly and less eco friendly. Further the above processes are low yielding and with less purity. US 8,217,034 discloses variant process for the preparation of Dolutegravir.
This process involves the reaction of methyl l-(2,2-dihydroxyethyl)-4-oxo-3-[(phenylmethyl)oxy]-l,4-dihydro-2-pyridine carboxylate (XII) with (R)-3-amino-l-butanol (X) to produce (4R, 12o5)-4-methyl-7-[(phenylmethyl)oxy]-3,4,12,12a-tetrahydro-2//-pyrido[ 1 \2′,4,5] pyrazino[2,l-b][l,3]oxazine-6,8-dione (XIII), which is further undergoes bromination using NBS to produce (4R,12aS)-9-bromo-4-methyl-7-[(phenylmethyl)oxy]-3,4,12,12a-tetrahydro-2H-pyrido[r,2′:4,5]pyrazino[2,l-b][l,3]oxazine-6,8-dione (XIV). The bromo Compound (XIV) is condensed with 2,4-difluorobenzylamine (III) in the presence of Tetrakis(triphenylphosphine)palladium (Pd(PPh3)4) to produce benzyloxy Dolutegravir (XI), which is hydrogenated in the presence of Pd/C to produce Dolutegravir (I). The process is as shown in Scheme-II below:
The major disadvantage with the above prior art process of preparing Dolutegravir is the use of expensive reagent tetrakis(triphenylphosphine)palladium (Pd(PPh3)4> in coupling step. Use of this reagent on industrial scale is not preferred, which makes the process more expensive. WO 2011/119566 discloses another variant process for the preparation of Dolutegravir.
This process involves the reaction of l-(2,2-dimethoxyethyl)-5-methoxy-6-(methoxycarbonyl)-4-oxo-l,4-dihydropyridine-3-carboxylic acid (XV) with acetic acid in presence of methane sulfonic acid to produce 5-methoxy-6-(methoxycarbonyl)-4-oxo-l-(2-oxoethyl)-l,4-dihydropyridine-3-carboxylic acid (XVI), which is further condensed with (R)-3-amino-l-butanol (X) to produce (4R,12aS)-7-methoxy-4-methyl-6,8-dioxo-3,4,6,8,12,12a-hexahydro-2//-pyrido[ 1 ‘,2’:4,5]pyrazino[2,1 -b] [ 1,3]-oxazine-9-carboxylic acid (XVII). This acid Compound XVII is acylated with 2,4-difluorobenzylamine (III) in the presence of carbonyldiimidazole (CDI) to produce methoxy Dolutegravir (XVIII), which is demethylated in the presence of lithium bromide (LiBr) to produce Dolutegravir (I).
The process is as shown in Scheme-3 below:
The major disadvantage of the above prior art process of preparing Dolutegravir is the use of expensive and highly moisture sensitive reagent, 1,1-carbonyldiimidazole (CDI), during acylation. Use of this reagent on industrial scale is not preferred due to anhydrous conditions required in the process. However, there is always a need for alternative preparative routes, which for example, involve fewer steps, use reagents that are less expensive and/or easier to handle, consume smaller amounts of reagents, provide a higher yield of product, have smaller and/or more eco-friendly waste products, and/or provide a product of higher purity. Hence, there is a need to develop cost effective and commercially viable process for the preparation of Dolutegravir of formula (I). The present invention is related to a process for the preparation of pure Dolutegravir of formula (I), wherein optically active acid addition salt of (R)-3-amino-l-butanol (X) is directly condensed with 5-methoxy-6-(methoxycarbonyl)-4-oxo-l-(2-oxoethyl)-l,4-dihydropyridine-3-carboxylic acid (XVI) instead of condensing with free base of (R)-3-amino-1-butanol (X). The present invention is also related to a process for the preparation of pure Dolutegravir of formula (I), wherein, inexpensive and easily handling condensing reagents in the condensation of (4R, 12aS)-7-methoxy-4-methyl-6,8-dioxo-3,4,6,8,12,12a-hexahydro-2//-pyrido[l’,2′:4,5]pyrazino [2,l-b][l,3]oxazine-9-carboxylic acid (XVII) with 2,4-difluorobenzylamine (III).


AN IMPROVED PROCESS FOR THE PREPARATION OF DOLUTEGRAVIR
| APPLICATION NUMBER | 1361/CHE/2013 |
| APPLICANT NAME | AUROBINDO PHARMA LTD |
| DATE OF FILING | 27/03/2013 |
| PUBLICATION DATE (U/S 11A) | 16/01/2015 |
In another embodiment, 5-methoxy-6-(methoxycarbonyl)-4-oxo-l-(2-oxoethyl)-l,4- dihydropyridine-3-carboxylic acid (XVI) used in the present invention is prepared by reacting 4-methoxyacetoacetate (XIX) with N,N-dimethyl-l,l- bis(methyloxy)methanamine (DMF-DMA) (XX) to produce methyl-2- (dimethylaminomethylene)-4-methoxy-3-oxo-butanoate(methyl-3-(dimethylamino)-2 [(methyloxy)acetyl]-2-propenoate) (XXI), which is reacted with aminoacetaldehyde dimethyl acetal (XXII) to produce methyl-2-(2,2-dimethoxyethylaminomethylene)-4-methoxy-3-oxo-butanoate(methyl-3-{[2,2-bis(methyloxy)ethyl]amino}-2-[(methyloxy) acetyl]-2-propenoate) (XXIII).
The compound (XXIII) is contacted with dimethyl ethanedioate in presence of alkali metal alkoxide to produce dimethyl-1-(2,2-dimethoxyethyl)-3-methoxy-4-oxo-l ,4-dihydropyridine-2,5-dicarboxylate (XXIV), which is selectively hydrolyzed with a base to produce l-[2,2-bis(methyloxy)ethyl]-5-(methyloxy)-6-[(methyloxy)carbonyl]-4-oxo-l ,4-dihydro-3-pyridinecarboxylic acid (XV). The compound (XV) is treated with a catalytic amount of a strong protic acid in the presence of acetic acid in an organic solvent to produce a reaction mixture containing 5- methoxy-6-(methoxycarbonyl)-4-oxo-l-(2-oxoethyl)-l,4-dihydropyridine-3-carboxylic acid (XVI), The process is as shown in Scheme-IV below:
The following examples illustrate the nature of the invention and are provided for illustrative purposes only and should not be construed to limit the scope of the invention.
Example-1:
EXAMPLES: Example-1: Process for the preparation of Dolutegravir
Step-i: Preparation of (/?)-3-amino-l-butanol tartarate salt: D-(+) Tartaric acid (12.7 g, 0.085 mol) was added in to a solution of (i?,5)-3-amino-l-butnaol (7.5 g, 0.084 mol) in methanol (100 ml) at 40 °C. The reaction mixture was stirred for about 1 hour at 35-40 °C and the reaction mass was cooled to 0-5°C and maintained for 30-40 minutes. The obtained solid was filtered and washed with chilled methanol (10 ml) at 0-5 °C. The solid was dried to get (i?)-3-amino-l-butanol tartarate salt (8.0 g, 40%).
Step-ii: Preparation of (4rt,12a£)-7-methoxy-4-methyl-6,8-dioxo-3,4,6,8,12,12a-hexahydro-2H-pyrido[l’,2′;4,5]pyrazino[2,l-b][l,3]oxazine-9-carboxylic acid (XVII): l-[2,2-Bis(methyloxy)ethyl]-5-(methyloxy)-6-[(methyloxy)carbonyl]-4-oxo-l,4-dihydro-3-pyridinecarboxylic acid (XV) (lOOg; 0.3175 moles) was suspended in acetonitrile (800 ml) and heated to 80-82°C. A mixture of acetic acid (95.25 g), methanesulfonic acid (9.14 g; 0.09525 moles) and acetonitrile (200 ml) were added to the slurry at 80-82°C. The reaction mass was continued at 80-82°C to complete the reaction. After completion of the reaction, anhydrous sodium acetate (65 g) and (/?)-3-amino-l-butanol tartrate salt (79.68g; 0.3334 moles) were added at 20-25°C and stirred at 60-65°C to complete the reaction. The reaction mass was concentrated and acidified with IN aqueous hydrochloric acid (750 ml) and extracted with methylene chloride (1500 ml) at ice cold temperature. The organic layer was separated, concentrated, treated with hot methanol (350 ml) for 2 h, filtered, washed with methanol and dried to yield (4R,12aS)-7-methoxy-4-methyl-6,8-dioxo-3,4,6,8,12,12a-hexahydro-2H-pyrido[ 1′ ,2′ :4,5]pyrazino[2,1 -b] [ 1,3]oxazine-9-carboxylic acid (XVII) (72 g; HPLC purity: 99.07%).
Step-iii: Process for the preparation of Dolutegravir (I). Method A: Triethylamine (3.61 g; 0.0357 moles) was added to the suspension of (4R,12aS)-7- methoxy-4-methyl-6,8-dioxo-3,4,6,8,12,12a-hexahydro-2H-pyrido[ 1′ ,2′ :4,5]pyrazino[2,1 – b][l,3]oxazine-9-carboxylic acid (XVII) (10 g; 0.0325 moles) in methylene chloride (50 ml), and cooled to 10-15°C. Pivaloyl chloride (4.3 g; 0.0357 moles) was added to the reaction mass, and stirred at 10-15°C for 1 h. Thereafter, 2,4-difiuorobenzylamine (5.58 g; 0.0389 moles) was added at 10-15°C and then warmed to 20-25°C to complete the reaction. After completion of the reaction, IN aqueous hydrochloric acid (20 ml) was added, organic layer was separated, washed with 5% w/w aqueous sodium bicarbonate solution (10 ml) followed by 15% w/w aqueous sodium chloride solution (10 ml) and concentrated. To the concentrated mass, acetonitrile (100 ml) and Lithium bromide (5.08 g; 0.0584 moles) were added and heated to 65-70°C for 3 h to complete the reaction. After completion of the reaction, the reaction mass was acidified with 5N aqueous hydrochloric acid (40 ml), concentrated to about 50 ml and DM water was added to crystallize the product at 20-25°C. The slurry was stirred for 2 h, filtered, washed with DM water and dried to yield (4R,12aS)-N-(2,4-difluorobenzyl)-7-hydroxy-4-methyl-6,8-dioxo-3,4,6,8,12,12a,-hexahydro-2H-pyrido[ 1′ ,2′ :4,5]pyrazino[2,1 -b] [ 1,3]oxazine-9-carboxamide (I) (11.5 g, HPLC purity: 99.63%).
Method B: Isobutyl chloroformate (4.65 gm, 0.03404 moles) in methylene chloride (10 ml) was added to the solution of N-methylmorpholine (3.45 gm, 0.03410 moles) and (4R,12aS)-7-methoxy-4-methyl-6,8-dioxo-3,4,6,8,12,12a-hexahydro-2H-pyrido[ 1′ ,2′ :4,5]pyrazino-[2,1 -b][l,3]oxazine-9-carboxy!ic acid (XVII) (10.0 gm, 0.03245 moles) in methylene chloride (60 ml) at -10 to 0°C in about 1 h. 2,4-Difloro benzyl amine (4.88 gm, 0.03409 moles) in methylene chloride (10 ml) was added to the cold reaction mass, and stirred at 20-30°C for completion of reaction. After completion of reaction, the reaction mass was washed with 5%w/w aqueous sodium bicarbonate solution (20 ml), IN hydrochloric acid (20 ml), DM water (20 ml) and concentrated. Acetonitrile (120 ml) and lithium bromide (4.8 gm, 0.05516 moles) were added to the concentrated mass, and stirred at 70-80°C for 3 h to complete the reaction. After completion of reaction, the reaction mass was acidified with 5N aqueous hydrochloric acid (40 ml) and concentrated to about 50 ml. DM Water (100 ml) was added to the concentrated reaction mass and stirred for 2 h at 25-30°C to crystallize the product. The product was filtered, washed with DM Water (50 ml) and dried to yield Dolutegravir (I) (10.7 gm, HPLC purity: 99.60%).
Example-2: Process for the preparation of Dolutegravir (I) (4R, 12aS)-N-(2,4-difluorobenzyl)-7-methoxy-4-methyl-6,8-dioxo-3,4,6,8,12,12a,-hexahydro-2H-pyrido[r,2′:4,5]pyrazino[2,l-b][l,3]oxazine-9-carboxamide (XVIII) (2 g, 0.0046 moles) was suspended in isopropyl alcohol (20 ml) and lithium bromide (0.8 g, 0.00924 moles) was added and stirred at 70-80°C for 15 h to complete the reaction. After completion of reaction the reaction mass was acidified with 5N aqueous hydrochloric acid (5 ml) and concentrated. DM Water (20 ml) was added to the concentrated mass and stirred at 25-30°C to crystallize the product. The product was filtered, washed with DM Water and dried to yield Dolutegravir (I) (1.5 g, HPLC purity: 97.93%).


/////
FDA warns Mylan about cGMP violations at its Indian facilities
DRUG REGULATORY AFFAIRS INTERNATIONAL

The US FDA has warned Mylan about manufacturing concerns at three of its plants in India.
In a warning letter to the generic drug manufacturer, the FDA said it had found ‘significant violations of current good manufacturing practice’ during inspections at the plants in August and September last year and in February this year.
The inspections relate to Mylan’s Agila Specialty Formulation Facility (SFF), Sterile Product Division (SPD), and Onco Therapies Limited (OTL) sites in Bangalore.

Some of the violations cited were failure to establish and follow appropriate written procedures designed to prevent microbiological contamination of drug products, such as the use of gloves with tears and pinholes, as well as deficiencies in environmental monitoring and poor monitoring of staff……..http://www.manufacturingchemist.com/news/article_page/FDA_warns_Mylan_about_cGMP_violations_at_its_Indian_facilities/111318/cn48579?dm_i=8EU,3MBVR,9ETTTY,D0ENC,1

Recently the Food and Drug Administration (FDA) began ramping up inspections of offshore manufacturing facilities and the results are shocking. Although cGMP violations have been found worldwide, experts are…
View original post 508 more words
Filgotinib

Filgotinib
EU APPROVED 2020/9/24, JYSELECA
JAPAN APPROVED2020/9/25
- C21H23N5O3S
- MW425.504
- Elemental Analysis: C, 59.28; H, 5.45; N, 16.46; O, 11.28; S, 7.54
1206161-97-8
Cyclopropanecarboxamide, N-[5-[4-[(1,1-dioxido-4-thiomorpholinyl)methyl]phenyl][1,2,4]triazolo[1,5-a]pyridin-2-yl]-
G146034
GLPG0634
N-(5-(4-((1,1-dioxidothiomorpholino)methyl)phenyl)-[1,2,4]triazolo[1,5-a]pyridin-2-yl)cyclopropanecarboxamide
Galapagos Nv INNOVATOR
PHASE 3, Crohn’s disease, Rheumatoid arthritis, Ulcerative colitis
Filgotinib is an orally available inhibitor of JAK1/JAK2 and TYK2 in phase III clinical development at Galapagos and Gilead for the treatment of rheumatoid arthritis, moderate or severe Crohn’s disease and ulcerative colitis
IL-6 antagonist; Jak1 tyrosine kinase inhibitor; Tyk2 tyrosine kinase inhibitor; Jak3 tyrosine kinase inhibitor; Jak2 tyrosine kinase inhibitor
Autoimmune disease; Cancer; Colitis; Crohns disease; Inflammatory disease; Neoplasm; Rheumatoid arthritis; Transplant rejection
In 2017, orphan drug designation was assigned to the compound in the U.S. for the treatment of pediatric Crohn’s disease and pediatric ulcerative colitis.
GlaxoSmithKline had been developing filgotinib preclinically for the treatment of rheumatoid arthritis pursuant to a license; however, in 2010, the compound was re-acquired by Galapagos. In 2012, the product was licensed to Abbott for development and marketing. In January 2013, Abbott spun-off its research-based pharmaceutical business into a newly-formed company AbbVie. The license agreement between Galapagos and Abbott was terminated in September 2015, Galapagos regaining all rights to the product. The same year, Galapagos and Gilead entered into a global partnership and Gilead obtained the global rights of codevelopment and commercialization for the treatment of inflammatory diseases
Filgotinib (GLPG0634), by the Belgian biotech company Galápagos NV, is a drug which is currently under investigation for the treatment of rheumatoid arthritis and Crohn’s disease.
Filgotinib (GLPG0634) is an orally-available, selective inhibitor of JAK1 (Janus kinase 1) for the treatment of rheumatoid arthritis and potentially other inflammatory diseases. Filgotinib (GLPG0634) dose-dependently inhibited Th1 and Th2 differentiation and to a lesser extent the differentiation of Th17 cells in vitro. GLPG0634 was well exposed in rodents upon oral dosing, and exposure levels correlated with repression of Mx2 expression in leukocytes. The JAK1 selective inhibitor GLPG0634 (Filgotinib) is a promising novel therapeutic with potential for oral treatment of rheumatoid arthritis and possibly other immune-inflammatory diseases. Filgotinib (GLPG0634) is currently in a Phase 2 study in Crohn’s disease.

Mechanism of action
Filgotinib is a Janus kinase inhibitor with selectivity for subtype JAK1 of this enzyme. It is considered a promising agent as it inhibits JAK1 selectively. Less selective JAK inhibitors (e.g. tofacitinib) are already being marketed. They show long-term efficacy in the treatment of various inflammatory diseases. However, their lack of selectivity leads to dose-limiting side effects.[1] It is thought that inhibition of all JAK isoenzymes is beneficial in rheumatoid arthritis. However, pan-JAK inhibition might also lead to unwanted side effects that might not outweigh its benefits. This is the rationale for the development of newer and more selective inhibitors like filgotinib.
The signal transmission of large numbers of proinflammatory cytokines is dependent on JAK1. Inhibition of JAK2 may also contribute to the efficacy against RA. Nonetheless it is thought that JAK2 inhibition might lead to anemia and thrombopenia by interference witherythropoietin and thrombopoietin and granulocyte-macrophage colony-stimulating factor. Therefore one might prefer to choose a more selective JAK1 inhibitor as a primary therapeutic option. Filgotinib exerts a 30-fold selectivity for JAK1 compared to JAK2.[2] It is however still to be seen to what extent JAK2 inhibition should be avoided.
Novel crystalline forms of filgotinib salts, particularly hydrochloride salt, useful for treating JAK-mediated diseases eg inflammatory diseases, autoimmune diseases, proliferative diseases, allergy and transplant rejection. Galapagos and licensee AbbVie are developing filgotinib, a selective JAK-1 inhibitor, for treating rheumatoid arthritis (RA) and Crohn’s disease (CD). In August 2015, the drug was reported to be in phase 2 clinical development for treating RA and CD. The drug is also being investigated for the treatment of colitis and was discovered as part of the company’s arthritis alliance with GSK; however in August 2010 Galapagos reacquired the full rights. See WO2013189771, claiming use of filgotinib analog for treating inflammatory diseases. Also see WO2010010190 (co-assigned with GSK and Abbott) and WO2010149769 (assigned to Galapagos) claiming filgotinib, generically and specifically, respectively.
Clinical trials and approval
The efficacy of filgotinib is currently studied in a phase2b program (DARWIN trial 1, 2) with involvement of 886 rheumatoid arthritis patients and 180 Crohn’s disease patients.
Phase 1 study
It was shown in phase 1 studies that the pharmacokinetics of filgotinib metabolism is independent of hepatic CYP450 enzymatic degradation. The drug metabolism is however mediated by carboxylesterases. There is no interference reported with the metabolism of methotrexate nor with any of the investigated transport proteins.[3]
Phase 2 study: Proof of concept (2011)
In november 2011 Galápagos released the results of their phase 2 study (identification: NCT01384422, Eudract: 2010-022953-40) in which 36 patients were treated who showed a suboptimal clinical response to methotrexate treatment. Three groups of twelve patients were treated either with 200 mg filgotinib in a single dose, 200 mg divided in two doses or placebo. The primary end-point was the ACR20 score, which monitors improvements in the symptomatology of the patient. After the scheduled 4 weeks of treatment, 83% of the respondents showed an improved ACR20-score. Half of the treated patients showed a complete (or near complete) remission of the disease. There were no reports ofanemia nor changes in lipidemia. The company stated in their press release that filgotinib is the first selective JAK1 inhibitor that shows clinical efficacy. As a result of this study, the company stated that “GLPG0634 shows one of the highest initial response rates ever reported for rheumatoid arthritis treatments”.[4]
DARWIN 1 trial
The DARWIN 1 trial is a 24 week double blind placebo-controlled trial with 599 rheumatoid arthritis patients enrolled. All participants have moderate to severe RA and showed an insufficient response to standard methotrexate treatment. The trial compares three dosages of filgotinib as a once or twice per day regimen. During the trial all participants remain on their methotrexate treatment. According to the company, the results of this trial are expected in July 2015.[5]
DARWIN 2 trial
The DARWIN 2 trial is a double blind placebo-controlled trial with 280 rheumatoid arthritis patients enrolled who show an insufficient response to standard methotrexate treatment. This trial, in contrast to the previous DARWIN 1 trial, methotrexate is discontinued. Therefore, this trial investigates filgotinib as a monotherapy.[6] The recruitment of DARWIN trial 2b ended in november 2014.[7] Preliminary results are expected in the second quarter of 2015 and a full completion of the study is expected in the third quarter of 2015.
DARWIN 3 trial
Patients who complete DARWIN 1 and 2 will be eligible for DARWIN 3.
COSY PREDICT
Time line
- june 2011: results of first phase 2 trial
- november 2014: initiation of DARWIN 1 and 2 trials
- april 2015: expected date of DARWIN 1 trial results
- june 2015: expected date of DARWIN 2 trial results
NMR FROM NET….ABMOLE, DMSOD6
NMR MEDKOO DMSOD6
CHEMIETEK
1H NMR PREDICT
13C NMR PREDICT
……………………
MORE PREDICTS
1H NMR PREDICT
13C NMR PREDICT
PRODUCT PATENT
http://www.google.com/patents/WO2010149769A1?cl=en
| Applicants: | GALAPAGOS NV [BE/BE]; Generaal De Wittelaan L11/A3 B-2800 Mechelen (BE) (For All Designated States Except US). MENET, Christel Jeanne Marie [FR/BE]; (BE) (For US Only). SMITS, Koen Kurt [BE/BE]; (BE) (For US Only) |
| Inventors: | MENET, Christel Jeanne Marie; (BE). SMITS, Koen Kurt; (BE) |
| International Filing Date: | 25.06.2010 |
ESTIMATED EXP 2030
Condensation of 2-amino-6-bromopyridine (I) with ethoxycarbonyl isothiocyanate (II) in CH2Cl2 gives 1-(6-bromopyridin-2-yl)-3-carboethoxythiourea (III), which upon cyclization with hydroxylamine hydrochloride (IV) in the presence of DIEA in EtOH/MeOH yields 2-amino-5-bromo[1,2,4]triazolo[1,5-a]pyridine (V). N-Acylation of amine (V) with cyclopropanecarbonyl chloride (VI) using Et3N in acetonitrile, and subsequent treatment with methanolic ammonia furnishes the carboxamide (VII) (1-3), which upon Suzuki coupling with 4-(hydroxymethyl)phenylboronic acid (VIII) in the presence of PdCl2(dppf) and K2CO3 in dioxane/H2O at 90 °C, followed by bromination with PBr3 in CHCl3 affords intermediate (IX). Condensation of benzyl bromide derivative (IX) with thiomorpholine-1,1-dioxide (X) using DIEA in CH2Cl2/MeOH yields filgotinib (1,2). Alternatively, condensation of (4-bromomethylphenyl)-4,4,5,5-tetramethyl-[1,3,2]dioxaborolane (XI) with thiomorpholine 1,1-dioxide (X) in the presence of DIEA in CH2Cl2/MeOH gives intermediate (XII), which undergoes Suzuki coupling with aryl bromide (VII) in the presence of PdCl2(dppf) and K2CO3 in dioxane/H2O at 90 °C to afford the target filgotinib
The present invention is based on the discovery that the compound of the invention is able to act as an inhibitor of JAK and that it is useful for the treatment of inflammatory conditions, autoimmune diseases, proliferative diseases, transplantation rejection, diseases involving impairment of cartilage turnover, congenital cartilage malformations, and/or diseases associated with hypersecretion of IL6. In a specific aspect the compound is an inhibitor of JAKl and JAK2. The present invention also provides methods for the production of this compound, a pharmaceutical composition comprising this compound and methods for treating inflammatory conditions, autoimmune diseases, proliferative diseases, transplantation rejection, diseases involving impairment of cartilage turnover, congenital cartilage malformations, and/or diseases associated with hypersecretion of IL6 by administering the compound of the invention.
Accordingly, in a first aspect of the invention, a compound of the invention is provided having a formula (I):

[0017] The compound of the invention is a novel inhibitor of JAK that appears to exhibit a dramatically improved in vivo potency as compared to structurally similar compounds. In a particular embodiment the compound of the invention is an inhibitor of JAKl and JAK2. In particular it appears to exhibit this increase in potency at lower in vivo exposure levels compared to structurally similar compounds. The use of a compound with these improvements is expected to result in a lower dosage requirement (and therefore an improved dosing schedule).
General Synthetic Method Scheme 1

1. RCOCI, Et3N 2. NH3 / MeOH CH3CN, 20 0C 2O 0C

wherein Ar represents phenyl-Ll-heterocycloalkyl, where Ll is a bond, -CH2– or -CO- and the heterocycloalkyl group is optionally substituted.
General
1.1.1 l-(6-Bromo-pyridin-2-yl)-3-carboethoxy-thiourea (2)

(2)
[00117] To a solution of 2-amino-6-bromopyridine (1) (253.8 g, 1.467 mol) in DCM (2.5 L) cooled to 5 0C is added ethoxycarbonyl isothiocyanate (173.0 mL, 1.467 mol) dropwise over 15 min. The reaction mixture is then allowed to warm to room temp. (20 0C) and stirred for 16 h. Evaporation in vacuo gives a solid which may be collected by filtration, thoroughly washed with petrol (3×600 mL) and air-dried to afford (2). The thiourea may be used as such for the next step without any purification. 1H (400 MHz, CDCl3) δ 12.03 (IH, br s, NH), 8.81 (IH, d, J 7.8 Hz, H-3), 8.15 (IH, br s, NH), 7.60 (IH, t, J 8.0 Hz, H-4), 7.32 (IH, dd, J 7.7 and 0.6 Hz, H-5), 4.31 (2H, q, J 7.1 Hz, CH2), 1.35 (3H, t, J 7.1 Hz, CH3).
7.7.2 5-Bromo-[l, 2, 4]triazolo[l, 5-a]pyridin-2-ylamine (3)

[00118] To a suspension of hydroxylamine hydrochloride (101.8 g, 1.465 mol) in EtOH/MeOH
(1 :1, 900 mL) is added N,N-diisopropylethylamine (145.3 mL, 0.879 mol) and the mixture is stirred at room temp. (20 0C) for 1 h. l-(6-Bromo-pyridin-2-yl)-3-carboethoxy-thiourea (2) (89.0 g, 0.293 mol) is then added and the mixture slowly heated to reflux (Note: bleach scrubber is required to quench H2S evolved). After 3 h at reflux, the mixture is allowed to cool and filtered to collect the precipitated solid. Further product is collected by evaporation in vacuo of the filtrate, addition Of H2O (250 mL) and filtration. The combined solids are washed successively with H2O (250 mL), EtOH/MeOH (1 : 1, 250 mL) and Et2O (250 mL) then dried in vacuo to afford the triazolopyridine derivative (3) as a solid. The compound may be used as such for the next step without any purification. 1H (400 MHz, DMSO-t/β) δ 7.43-7.34 (2H, m, 2 x aromatic-H), 7.24 (IH, dd, J 6.8 and 1.8 Hz, aromatic-H), 6.30 (2H, br, NH2); m/z 213/215 (1 :1, M+H+, 100%).
7.7.3 General procedure for mono-acylation to afford intermediate (4):

[00119] To a solution of the 2-amino-triazolopyridine (3) (7.10 g, 33.3 mmol) in dry CH3CN
(150 mL) at 5 0C is added Et3N (11.6 mL, 83.3 mmol) followed by cyclopropanecarbonyl chloride (83.3 mmol). The reaction mixture is then allowed to warm to ambient temperature and stirred until all starting material (3) is consumed. If required, further Et3N (4.64 mL, 33.3 mmol) and cyclopropanecarbonyl chloride (33.3 mmol) is added to ensure complete reaction. Following solvent evaporation in vacuo the resultant residue is treated with 7 N methanolic ammonia solution (50 mL) and stirred at ambient temp, (for 1-16 h) to hydro lyse any bis-acylated product. Product isolation is made by removal of volatiles in vacuo followed by trituration with Et2O (50 mL). The solids are collected by filtration, washed with H2O (2x50mL), acetone (50 mL) and Et2O (50 mL), then dried in vacuo to give the required bromo intermediate (4).
Method A
Preparation of compounds of the invention via Suzuki coupling (5):
[00120] An appropriate boronic acid (2eq.) is added to a solution of bromo intermediate (4) in
1 ,4-dioxane/water (5:1). K2CO3 (2 eq.) and PdCl2dppf (5%) are added to the solution. The resulting mixture is then heated in a microwave at 140 0C for 30 min (this reaction can also be carried out by traditional heating in an oil bath at 900C for 16h under N2). Water is added and the solution is extracted with ethyl acetate. The organic layers are dried over anhyd. MgSθ4 and evaporated in vacuo. The final compound is obtained after purification by flash chromatography or preparative HPLC. HPLC: Waters
XBridge Prep Cl 8 5μm ODB 19mm ID x 100mm L (Part No.186002978). All the methods are using
MeCN/H2O gradients. H2O contains either 0.1% TFA or 0.1% NH3.
Method B

Bl. 4 4-[2-(Cyclopropanecarbonyl-amino)-[ 1 , 2, 4]triazolo[l, 5-a] pyridin-5-yl] -benzoyl chloride

[00121] 2 Drops of DMF are added to a solution of 4-[2-(cyclopropanecarbonyl-amino)- [l,2,4]triazolo[l,5-a]pyridin-5-yl]-benzoic acid (1 eq) obtained by Method A using 4-carboxyphenylboronic acid in DCM under N2 atmosphere. Then oxalyl chloride (2 eq) is added dropwise to this resulting solution (gas release). The mixture is stirred at room temperature for 2 hours. After completion of the reaction by LCMS, the solvent is removed. The crude acid chloride is used without further purification in next step.
B2. Amide formation (General Method)

[00122] An appropriate amine (1.1 eq) and Et3N (5 eq) are dissolved in DCM under N2 atmosphere and cooled at 00C. The acid chloride (Bl, 1 eq) dissolved in DCM is added dropwise to this solution. The reaction is stirred at room temperature for 16 h. After this time, reaction is complete. The compound is extracted with EtOAc and water, washed with brine and dried over anhyd. MgSO4. Organic layers are filtered and evaporated. The final compound is isolated by preparative HPLC. Preparative HPLC: Waters XBridge Prep C18 5μm ODB 19mm ID x 100mm L (Part No.186002978). All the methods are using MeCN/H2O gradients. H2O contains either 0.1% TFA or 0.1% NH3.
Method C

Wherein R3a or R3b together with the nitrogen atom to which they are attached, may form a heterocycloalkyl.
Reductive alkylation (general method)
[00123] An appropriate amine (2 eq.), cyclopropanecarboxylic acid (for example cyclopropanecarboxylic acid [5-(4-formyl-phenyl)-[l,2,4]triazolo[l,5-a]pyridine-2-yl]-amide) prepared by method A (1 eq.) and Ti(OPr)4 are mixed and stirred at room temperature for 3 hrs. The mixture is diluted in ethanol and Na(CN)BH3 (leq.) is added. The resulting solution is stirred at room temperature for 16 hrs. The mixture is diluted in water and filtered. The filtrate is washed with ethanol. The combined solvent phases are evaporated under vacuum. The final compound is isolated by preparative HPLC.
Method D 
wherein R1 and R2 together with the Nitrogen atom to which they are attached, may form a heterocycloalkyl.
Reaction ofalkylation

[00124] 2-(4-Bromomethyl-phenyl)-4,4,5,5-tetramethyl-[l,3,2]dioxaborolane (leq) and Et3N (2 eq) (or AgCO3) are dissolved in DCM/MeOH (4:1 v:v) under N2 and an amine (2 eq) is added dropwise. The resulting solution is stirred at room temperature for 16h. After this time, the reaction is complete. The solvent is evaporated. The compound is extracted with EtOAc and water, washed with brine and dried over anhyd. MgSθ4. Organic layers are filtered and evaporated. The final compound is isolated by flash chromatography.
Suzuki coupling

[00125] The obtained boronic acid (2eq.) is added to a solution of cyclopropanecarboxylic acid
(5-bromo-[l,2,4]triazolo[l,5-a]pyridin-2-yl)-amide (4) in 1 ,4-dioxane/water (5:1). K2CO3 (2 eq.) and PdCl2dppf (5%) are added to the solution. The resulting mixture is then heated in a microwave at 140 0C for 30 min (This reaction can also be carried out by traditional heating in an oil bath at 900C for 16h under N2). Water is added and the solution is extracted with ethyl acetate. The organic layers are dried over anhyd. MgSθ4 and evaporated in vacuo. The final compound is obtained after purification by flash chromatography or preparative HPLC. HPLC: Waters XBridge Prep C18 5μm ODB 19mm ID x 100mm L (Part No.186002978). All the methods are using MeCN/H2O gradients. H2O contains either 0.1% TFA or 0.1% NH3.
Synthesis of the compound of the invention and comparative examples
Compound l(the compound of the invention)
Step 1:

[00126] 2-(4-Bromomethyl-phenyl)-4,4,5,5-tetramethyl-[l,3,2]dioxaborolane (leq) and DIPEA
(2 eq) were dissolved in DCM/MeOH (5:1 v:v) under N2 and thiomorpholine 1,1 -dioxide (2 eq) was added portionwise. The resulting solution was stirred at room temperature for 16h. After this time, the reaction was complete. The solvent was evaporated. The compound was extracted with EtOAc and water, washed with brine and dried over anhyd. MgS O4. Organic layers were filtered and evaporated. The final compound was isolated without further purification.
Step 2: Suzuki coupling

[00127] 4-[4-(4,4,5,5-Tetramethyl-[l,3,2]dioxaborolan-2-yl)-benzyl]-thiomorpholine-l,l-dioxide
(l.leq.) was added to a solution of cyclopropanecarboxylic acid (5-bromo-[l,2,4]triazolo[l,5-a]pyridin-2-yl)-amide in 1 ,4-dioxane/water (4:1). K2CO3 (2 eq.) and PdCl2dppf (0.03 eq.) were added to the solution. The resulting mixture was then heated in an oil bath at 900C for 16h under N2. Water was added and the solution was extracted with ethyl acetate. The organic layers were dried over anhyd. MgSθ4 and evaporated in vacuo. The final compound was obtained after purification by flash chromatography.
[00128] Alternatively, after completion of the reaction, a palladium scavenger such as 1,2-bis(diphenylphosphino)ethane, is added, the reaction mixture is allowed to cooled down and a filtration is performed. The filter cake is reslurried in a suitable solvent (e.g. acetone), the solid is separated by filtration, washed with more acetone, and dried. The resulting solid is resuspended in water, aqueous HCl is added, and after stirring at RT, the resulting solution is filtered on celite (Celpure P300). Aqueous NaOH is then added to the filtrate, and the resulting suspension is stirred at RT, the solid is separated by filtration, washed with water and dried by suction. Finally the cake is re-solubilised in a mixture of THF/H2O, treated with a palladium scavenger (e.g. SMOPEX 234) at 500C, the suspension is filtered, the organic solvents are removed by evaporation, and the resulting slurry is washed with water and methanol, dried and sieved, to obtain the title compound as a free base.
Alternative route to Compound l(the compound of the invention):
Step 1:

[00129] 4-(Hydroxymethyl)phenylboronic acid (l.leq.) was added to a s o luti o n o f cyclopropanecarboxylic acid (5-bromo-[l,2,4]triazolo[l,5-a]pyridin-2-yl)-amide in 1 ,4-dioxane/water (4:1). K2CO3 (2 eq.) and PdCl2dppf (0.03 eq.) were added to the solution. The resulting mixture was then heated in an oil bath at 900C for 16h under N2. Water was added and the solution was extracted with ethyl acetate. The organic layers were dried over anhyd. MgSθ4 and evaporated in vacuo. The resulting mixture was used without further purification.
Step 2:

[00130] To a solution of cyclopropanecarboxylic acid [5-(4-hydroxymethyl-phenyl)- [l,2,4]triazolo[l,5-a]pyridin-2-yl]-amide (1.0 eq) in chloroform was slowly added phosphorus tribromide (1.0 equiv.). The reaction mixture was stirred at room temperature for 20 hours, quenched with ice and water (20 mL) and extracted with dichloromethane. The organic layer was dried over anhyd. MgSθ4, filtered and concentrated to dryness. The resulting white residue was triturated in dichloromethane/diethyl ether 2:1 to afford the expected product as a white solid.
Step 3:

[00131] Cyclopropanecarboxylic acid [5-(4-bromomethyl-phenyl)-[l,2,4]triazolo[l,5-a]pyridin- 2-yl]-amide (leq) and DIPEA (2 eq) were dissolved in DCM/MeOH (5:1 v:v) under N2 and thiomorpholine 1,1 -dioxide (1.1 eq) was added dropwise. The resulting solution was stirred at room temperature for 16h. After this time, the reaction was complete. The solvent was evaporated. The compound was dissolved in DCM, washed with water and dried over anhyd. MgSO^ Organic layers were filtered and evaporated. The final compound was isolated by column chromatography using EtOAc to afford the desired product.
PATENT
WO 2010010190
WO 2013173506
WO 2013189771
WO 2015117980
WO 2015117981
POLYMORPH
CN 105061420
https://encrypted.google.com/patents/CN105061420A?cl=en
JAK inhibitor N-(5-(4-(1,1-dioxothiomorpholinyl)methyl)phenyl)-[1,2,4]triazolo[1,5-a]pyridin-2-yl)cyclopropanecarboxamide, and methods for preparing the four crystal forms, wherein the four crystal forms respectively are a crystal form H1, a crystal form H2, a crystal form H3 and a crystal form H4,
POLYMORPH
E CRYSTAL
CN 105111206
D CRYSTAL
CN 105111207
H CRYSYAL
CN 105198876
G
CN 105198877
F CN 105198878
C CN 105198880
POLYMORPH
WO 2016105453
POLYMORPH
Preparation of solid forms of filgotinib free base
WO 2017012773
The present invention relates to cryst. filgotinib free base (I), a method of its prepn. and a pharmaceutical compn. comprising the same. Thus, reacting the compd. II with thiomorpholine dioxide afforded 92% I (purity of 98.6%) which was then converted into its hydrochloride salt. Pharmaceutical compn. comprising compd. I was disclosed.
POLYMORPH
CN 105669669
The present invention provides a crystal form A, B, D, G and M of N-[5-[4-[(1,1-dioxido-4-thiomorpholinyl)methyl]phenyl][1,2,4]triazolo[1,5-a]pyridin-2-yl]cyclopropanecarboxamide hydrochloride.
PAPER
Future Medicinal Chemistry (2015), 7(2), 203-235. | Language: English, Database: CAPLUSA review. The discovery of the JAK-STAT pathway was a landmark in cell biol. The identification of these pathways has changed the landscape of treatment of rheumatoid arthritis and other autoimmune diseases. The two first (unselective) JAK inhibitors have recently been approved by the US FDA for the treatment of myelofibrosis and rheumatoid arthritis and many other JAK inhibitors are currently in clin. development or at the discovery stage. Research groups have demonstrated the different roles of JAK member and the therapeutic potential of targeting them selectively. ………..
https://www.future-science.com/doi/10.4155/fmc.14.149
PAPER
Journal of Pharmaceutical Sciences (Philadelphia, PA, United States) (2018), 107(6), 1624-1632.
PATENT
US2010/331319 A1, ; Page/Page column 13-14
http://www.google.com/patents/US20100331319
Synthetic Preparation of the Compound of the Invention and Comparative Examples
The compound of the invention and the comparative examples can be produced according to the following scheme.
wherein Ar represents phenyl-L1-heterocycloalkyl, where L1 is a bond, —CH2— or —CO— and the heterocycloalkyl group is optionally substituted.
General 1.1.1 1-(6-Bromo-pyridin-2-yl)-3-carboethoxy-thiourea (2)
To a solution of 2-amino-6-bromopyridine (1) (253.8 g, 1.467 mol) in DCM (2.5 L) cooled to 5° C. is added ethoxycarbonyl isothiocyanate (173.0 mL, 1.467 mol) dropwise over 15 min. The reaction mixture is then allowed to warm to room temp. (20° C.) and stirred for 16 h. Evaporation in vacuo gives a solid which may be collected by filtration, thoroughly washed with petrol (3×600 mL) and air-dried to afford (2). The thiourea may be used as such for the next step without any purification. 1H (400 MHz, CDCl3) δ 12.03 (1H, br s, NH), 8.81 (1H, d, J=7.8 Hz, H-3), 8.15 (1H, br s, NH), 7.60 (1H, t, J=8.0 Hz, H-4), 7.32 (1H, dd, J 7.7 and 0.6 Hz, H-5), 4.31 (2H, q, J 7.1 Hz, CH2), 1.35 (3H, t, J 7.1 Hz, CH3).
1.1.2 5-Bromo-[1,2,4]triazolo[1,5-a]pyridin-2-ylamine (3)
To a suspension of hydroxylamine hydrochloride (101.8 g, 1.465 mol) in EtOH/MeOH (1:1, 900 mL) is added N,N-diisopropylethylamine (145.3 mL, 0.879 mol) and the mixture is stirred at room temp. (20° C.) for 1 h. 1-(6-Bromo-pyridin-2-yl)-3-carboethoxy-thiourea (2) (89.0 g, 0.293 mol) is then added and the mixture slowly heated to reflux (Note: bleach scrubber is required to quench H2S evolved). After 3 h at reflux, the mixture is allowed to cool and filtered to collect the precipitated solid. Further product is collected by evaporation in vacuo of the filtrate, addition of H2O (250 mL) and filtration. The combined solids are washed successively with H2O (250 mL), EtOH/MeOH (1:1, 250 mL) and Et2O (250 mL) then dried in vacuo to afford the triazolopyridine derivative (3) as a solid. The compound may be used as such for the next step without any purification. 1H (400 MHz, DMSO-d6) δ 7.43-7.34 (2H, m, 2×aromatic-H), 7.24 (1H, dd, J 6.8 and 1.8 Hz, aromatic-H), 6.30 (2H, br, NH2); m/z 213/215 (1:1, M+H+, 100%).
1.1.3 General Procedure for Mono-Acylation to Afford Intermediate (4)
To a solution of the 2-amino-triazolopyridine (3) (7.10 g, 33.3 mmol) in dry CH3CN (150 mL) at 5° C. is added Et3N (11.6 mL, 83.3 mmol) followed by cyclopropanecarbonyl chloride (83.3 mmol). The reaction mixture is then allowed to warm to ambient temperature and stirred until all starting material (3) is consumed. If required, further Et3N (4.64 mL, 33.3 mmol) and cyclopropanecarbonyl chloride (33.3 mmol) is added to ensure complete reaction. Following solvent evaporation in vacuo the resultant residue is treated with 7 N methanolic ammonia solution (50 mL) and stirred at ambient temp. (for 1-16 h) to hydrolyse any bis-acylated product. Product isolation is made by removal of volatiles in vacuo followed by trituration with Et2O (50 mL). The solids are collected by filtration, washed with H2O (2×50 mL), acetone (50 mL) and Et2O (50 mL), then dried in vacuo to give the required bromo intermediate (4).
Method A Preparation of Compounds of the Invention Via Suzuki Coupling (5):
An appropriate boronic acid (2 eq.) is added to a solution of bromo intermediate (4) in 1,4-dioxane/water (5:1). K2CO3 (2 eq.) and PdCl2dppf (5%) are added to the solution. The resulting mixture is then heated in a microwave at 140° C. for 30 min (this reaction can also be carried out by traditional heating in an oil bath at 90° C. for 16 h under N2). Water is added and the solution is extracted with ethyl acetate. The organic layers are dried over anhyd. MgSO4 and evaporated in vacuo. The final compound is obtained after purification by flash chromatography or preparative HPLC. HPLC: Waters XBridge Prep C18 5 μm ODB 19 mm ID×100 mm L (Part No. 186002978). All the methods are using MeCN/H2O gradients. H2O contains either 0.1% TFA or 0.1% NH3.
Method B
B1. 4 4-[2-(Cyclopropanecarbonyl-amino)-[1,2,4]triazolo[1,5-a]pyridin-5-yl]-benzoyl chloride
2 Drops of DMF are added to a solution of 4-[2-(cyclopropanecarbonyl-amino)-[1,2,4]triazolo[1,5-a]pyridin-5-yl]-benzoic acid (1 eq) obtained by Method A using 4-carboxyphenylboronic acid in DCM under N2 atmosphere. Then oxalyl chloride (2 eq) is added dropwise to this resulting solution (gas release). The mixture is stirred at room temperature for 2 hours. After completion of the reaction by LCMS, the solvent is removed. The crude acid chloride is used without further purification in next step.
B2. Amide Formation (General Method)
An appropriate amine (1.1 eq) and Et3N (5 eq) are dissolved in DCM under N2 atmosphere and cooled at 0° C. The acid chloride (B1, 1 eq) dissolved in DCM is added dropwise to this solution. The reaction is stirred at room temperature for 16 h. After this time, reaction is complete. The compound is extracted with EtOAc and water, washed with brine and dried over anhyd. MgSO4. Organic layers are filtered and evaporated. The final compound is isolated by preparative HPLC. Preparative HPLC: Waters XBridge Prep C18 5 μm ODB 19 mm ID×100 mm L (Part No. 186002978). All the methods are using MeCN/H2O gradients. H2O contains either 0.1% TFA or 0.1% NH3.
…
Synthesis of the Compound of the Invention and Comparative Examples Compound 1 (the Compound of the Invention) Step 1:
2-(4-Bromomethyl-phenyl)-4,4,5,5-tetramethyl-[1,3,2]dioxaborolane (1 eq) and DIPEA (2 eq) were dissolved in DCM/MeOH (5:1 v:v) under N2 and thiomorpholine 1,1-dioxide (2 eq) was added portionwise. The resulting solution was stirred at room temperature for 16 h. After this time, the reaction was complete. The solvent was evaporated. The compound was extracted with EtOAc and water, washed with brine and dried over anhyd. MgSO4. Organic layers were filtered and evaporated. The final compound was isolated without further purification.
STEP 2: Suzuki coupling
4-[4-(4,4,5,5-Tetramethyl-[1,3,2]dioxaborolan-2-yl)-benzyl]-thiomorpholine-1,1-dioxide (1.1 eq.) was added to a solution of cyclopropanecarboxylic acid (5-bromo-[1,2,4]triazolo[1,5-a]pyridin-2-yl)-amide in 1,4-dioxane/water (4:1). K2CO3 (2 eq.) and PdCl2dppf (0.03 eq.) were added to the solution. The resulting mixture was then heated in an oil bath at 90° C. for 16 h under N2. Water was added and the solution was extracted with ethyl acetate. The organic layers were dried over anhyd. MgSO4 and evaporated in vacuo. The final compound was obtained after purification by flash chromatography.
Alternatively, after completion of the reaction, a palladium scavenger such as 1,2-bis(diphenylphosphino)ethane, is added, the reaction mixture is allowed to cooled down and a filtration is performed. The filter cake is reslurried in a suitable solvent (e.g. acetone), the solid is separated by filtration, washed with more acetone, and dried. The resulting solid is resuspended in water, aqueous HCl is added, and after stirring at RT, the resulting solution is filtered on celite (Celpure P300). Aqueous NaOH is then added to the filtrate, and the resulting suspension is stirred at RT, the solid is separated by filtration, washed with water and dried by suction. Finally the cake is re-solubilised in a mixture of THF/H2O, treated with a palladium scavenger (e.g. SMOPEX 234) at 50° C., the suspension is filtered, the organic solvents are removed by evaporation, and the resulting slurry is washed with water and methanol, dried and sieved, to obtain the title compound as a free base.
Alternative Route to Compound 1 (the Compound of the Invention): Step 1:
4-(Hydroxymethyl)phenylboronic acid (1.1 eq.) was added to a solution of cyclopropanecarboxylic acid (5-bromo-[1,2,4]triazolo[1,5-a]pyridin-2-yl)-amide in 1,4-dioxane/water (4:1). K2CO3 (2 eq.) and PdCl2dppf (0.03 eq.) were added to the solution. The resulting mixture was then heated in an oil bath at 90° C. for 16 h under N2. Water was added and the solution was extracted with ethyl acetate. The organic layers were dried over anhyd. MgSO4 and evaporated in vacuo. The resulting mixture was used without further purification.
Step 2:
To a solution of cyclopropanecarboxylic acid [5-(4-hydroxymethyl-phenyl)-[1,2,4]triazolo[1,5-a]pyridin-2-yl]-amide (1.0 eq) in chloroform was slowly added phosphorus tribromide (1.0 equiv.). The reaction mixture was stirred at room temperature for 20 hours, quenched with ice and water (20 mL) and extracted with dichloromethane. The organic layer was dried over anhyd. MgSO4, filtered and concentrated to dryness. The resulting white residue was triturated in dichloromethane/diethyl ether 2:1 to afford the expected product as a white solid.
Step 3:
Cyclopropanecarboxylic acid [5-(4-bromomethyl-phenyl)-[1,2,4]triazolo[1,5-a]pyridin-2-yl]-amide (1 eq) and DIPEA (2 eq) were dissolved in DCM/MeOH (5:1 v:v) under N2 and thiomorpholine 1,1-dioxide (1.1 eq) was added dropwise. The resulting solution was stirred at room temperature for 16 h. After this time, the reaction was complete. The solvent was evaporated. The compound was dissolved in DCM, washed with water and dried over anhyd. MgSO4. Organic layers were filtered and evaporated. The final compound was isolated by column chromatography using EtOAc to afford the desired product.
…………………….
PATENT
Novel salts and pharmaceutical compositions thereof for the treatment of inflammatory disorders
Also claims a method for preparing filgotinib hydrochloride trihydrate. The present filing forms a pair with this week’s filing, WO2015117980, claiming a tablet composition comprising filgotinib hydrochloride.
The compound cyclopropanecarboxylic acid {5-[4-(l,l-dioxo-thiomorpholin-4-ylmethyl)-phenyl]-[l,2,4]triazolo[l,5-a]pyridin-2-yl -amide (Compound 1), which has the chemical structure:

is disclosed in our earlier application WO 2010/149769 (Menet C. J., 2010) as being an inhibitor of JAK and as being useful in the treatment of inflammatory conditions, autoimmune diseases, proliferative diseases, allergy, transplant rejection, diseases involving impairment of cartilage turnover, congenital cartilage malformations, and/or diseases associated with hypersecretion of IL6 or interferons. Hereafter this compound is named Compound 1. The data presented in WO 2010/149769 demonstrate that despite similar in vitro activities, Compound 1 has unexpectedly high in vivo potency compared with structurally similar compounds.
Example 1. Preparation of Compound 1
1.1. Route 1
1.1.1. 4-[4-(4,4,5,5-Tetramethyl-[l,3,2]dioxaborolan-2-yl)-benzyl]-thiomorpholine-l,l-dioxide

[00205] 2-(4-Bromomethyl-phenyl)-4,4,5,5-tetramethyl-[l,3,2]dioxaborolane (1 eq) and DIPEA (2 eq) are dissolved in DCM/MeOH (5:1 v:v) under N2 and thiomorpholine 1,1 -dioxide (2 eq) is added portionwise. The resulting solution is stirred at room temperature for 16h. After this time, the reaction is complete. The solvent is evaporated. The compound is extracted with EtOAc and water, washed with brine and dried over anhydrous MgSO i. Organic layers are filtered and evaporated. The final compound is isolated without further purification.
1.1.2. Cyclopropanecarboxylic acid (5-bromo-[l,2,4]triazolo[l,5-a]pyridin-2-yl)-amide

1.1.2.1. Step i): l-(6-Bromo-pyridin-2-yl)-3-carboethoxy-thiourea
[00206] To a solution of 2-amino-6-bromopyridine (1) (253.8 g, 1.467 mol) in DCM (2.5 L) cooled to 5°C is added ethoxycarbonyl isothiocyanate (173.0 mL, 1.467 mol) dropwise over 15 min. The reaction
mixture is then allowed to warm to room temp. (20 °C) and stirred for 16 h. Evaporation in vacuo gives a solid which may be collected by filtration, thoroughly washed with petrol (3 x 600 niL) and air-dried to afford the desired product. The thiourea may be used as such for the next step without any purification. lH (400 MHz, CDC13) δ 12.03 (1H, br s), 8.81 (1H, d), 8.15 (1H, br s), 7.60 (1H, t), 7.32 (1H, dd), 4.31 (2H, q), 1.35 (3H, t).
1.1.2.2. Step ii): 5-Bromo-[l,2,4]triazolo[l,5-a]pyridin-2-ylamine
[00207] To a suspension of hydroxylamine hydrochloride (101.8 g, 1.465 mol) in EtOH/MeOH (1 : 1, 900 mL) is added NN-diisopropylethylamine (145.3 mL, 0.879 mol) and the mixture is stirred at room temp. (20 °C) for 1 h. l-(6-Bromo-pyridin-2-yl)-3-carboethoxy-thiourea (2) (89.0 g, 0.293 mol) is then added and the mixture slowly heated to reflux (Note: bleach scrubber is required to quench H2S evolved). After 3h at reflux, the mixture is allowed to cool and filtered to collect the precipitated solid. Further product is collected by evaporation in vacuo of the filtrate, addition of H20 (250 mL) and filtration. The combined solids are washed successively with H20 (250 mL), EtOH/MeOH (1 : 1, 250 mL) and Et20 (250 mL) then dried in vacuo to afford the triazolopyridine derivative (3) as a solid. The compound may be used as such for the next step without any purification. lH (400 MHz, DMSO-i¼) δ 7.43-7.34 (2H, m, 2 x aromatic-H), 7.24 (1H, dd, J 6.8 and 1.8 Hz, aromatic-H), 6.30 (2H, br, NH2); m/z 213/215 (1 : 1, M+H+, 100%).
1.1.2.3. Step Hi): Cyclopropanecarboxylic acid (5-bromo-[l ,2,4]triazolo[l ,5-a]pyridin-2-yl)-amide
[00208] To a solution of the 2-amino-triazolopyridine obtained in the previous step (7.10 g, 33.3 mmol) in dry MeCN (150 mL) at 5°C is added Et3N (11.6 mL, 83.3 mmol) followed by cyclopropanecarbonyl chloride (83.3 mmol). The reaction mixture is then allowed to warm to ambient temperature and stirred until all starting material is consumed. If required, further Et3N (4.64 mL, 33.3 mmol) and cyclopropanecarbonyl chloride (33.3 mmol) is added to ensure complete reaction. Following solvent evaporation in vacuo the resultant residue is treated with 7 N methanolic ammonia solution (50 mL) and stirred at ambient temp, (for 1-16 h) to hydro lyse any bis-acylated product. Product isolation is made by removal of volatiles in vacuo followed by trituration with Et20 (50 mL). The solids are collected by filtration, washed with H20 (2x50mL), acetone (50 mL) and Et20 (50 mL), then dried in vacuo to give the desired compound.
1.1.3. Compound 1

[00209] 4-[4-(4,4,5,5-Tetramethyl-[l ,3,2]dioxaborolan-2-yl)-benzyl] hiomoφholine , l -dioxide (l . l eq.) is added to a solution of cyclopropanecarboxylic acid (5-bromo-[l ,2,4]triazolo[l ,5-a]pyridin-2-yl)-amide in 1 ,4-dioxane/water (4: 1). K2CO3 (2 eq.) and PdC^dppf (0.03 eq.) are added to the solution. The resulting mixture is then heated in an oil bath at 90°C for 16h under N2. Water is added and the solution is extracted with ethyl acetate. The organic layers are dried over anhydrous MgS04 and evaporated in vacuo.
[00210] The final compound is obtained after purification by flash chromatography.
[00211] Alternatively, after completion of the reaction, a palladium scavenger such as 1 ,2-bis(diphenylphosphino)ethane, is added, the reaction mixture is allowed to cool down and a filtration is performed. The filter cake is reslurried in a suitable solvent (e.g. acetone), the solid is separated by filtration, washed with more acetone, and dried. The resulting solid is resuspended in water, aqueous HC1 is added, and after stirring at room temperature, the resulting solution is filtered on celite (Celpure P300). Aqueous NaOH is then added to the filtrate, and the resulting suspension is stirred at room temperature, the solid is separated by filtration, washed with water and dried by suction. Finally the cake is re-solubilised in a mixture of THF/H20, treated with a palladium scavenger (e.g. SMOPEX 234) at 50°C, the suspension is filtered, the organic solvents are removed by evaporation, and the resulting slurry is washed with water and methanol, dried and sieved, to obtain the desired compound as a free base.
1.2. Route 2
1.2.1. Step 1: cyclopropanecarboxylic acid [5-(4-hydroxymethyl-phenyl)-[l,2, 4]triazolo[l, 5- a] pyridin-2-yl] -amide

[00212] 4-(Hydroxymethyl)phenylboronic acid (l . l eq.) is added to a solution of cyclopropanecarboxylic acid (5-bromo-[l ,2,4]triazolo[l ,5-a]pyridin-2-yl)-amide in 1 ,4-dioxane/water
(4:1). K2CO3 (2 eq.) and PdC^dppf (0.03 eq.) are added to the solution. The resulting mixture is then heated in an oil bath at 90°C for 16h under N2. Water is added and the solution is extracted with ethyl acetate. The organic layers are dried over anhydrous MgS04 and evaporated in vacuo. The resulting mixture is used without further purification.
1.2.2. Step 2: Cyclopropanecarboxylic acid [5-(4-bromomethyl-phenyl)-[l,2,4]triazolo[l,5- a Jpyridin-2-ylJ -amide

[00213] To a solution of cyclopropanecarboxylic acid [5-(4-hydroxymethyl-phenyl)-[l,2,4]triazolo[l,5-a]pyridin-2-yl] -amide (1.0 eq) in chloroform is slowly added phosphorus tribromide (1.0 eq.). The reaction mixture is stirred at room temperature for 20 h, quenched with ice and water (20 mL) and extracted with dichloromethane. The organic layer is dried over anhydrous MgSO i, filtered and concentrated to dryness. The resulting white residue is triturated in dichloromethane/diethyl ether 2:1 to afford the desired product.
1.2.3. Step 3:

[00214] Cyclopropanecarboxylic acid [5-(4-bromomethyl-phenyl)-[l,2,4]triazolo[l,5-a]pyridin-2-yl]-amide (l eq) and DIPEA (2 eq) are dissolved in DCM/MeOH (5: 1 v:v) under N2 and thiomorpho line 1,1-dioxide (1.1 eq) is added dropwise. The resulting solution is stirred at room temperature for 16h. After this time, the reaction is complete. The solvent is evaporated. The compound is dissolved in DCM, washed with water and dried over anhydrous MgSO i. Organic layers are filtered and evaporated. The final compound is isolated by column chromatography using EtOAc to afford the desired product.
…………………
PATENT
http://www.google.co.in/patents/WO2013189771A1?cl=en
Example 1. Synthesis of the compounds
1.1. Route 1
1.1.1. Synthesis of 5-Bromo-[l,2,4]triazolo[l,5-a]pyridin-2-ylamine (Intermediate 3)
led to 5 °C was added ethoxycarbonyl isothiocyanate (173.0 mL, 1.467 mol) dropwise over 15 min. The reaction mixture was then allowed to warm to room temp. (20 °C) and stirred for 16 h. Evaporation in vacuo gave a solid which was collected by filtration, thoroughly washed with petrol (3×600 mL) and air-dried to afford (2). The thiourea was used as such in the next step without any purification.
[00157] lH (400 MHz, CDC13) δ 12.03 (IH, br s, NH), 8.81 (IH, d, J 7.8 Hz, H-3), 8.15 (IH, br s, NH), 7.60 (IH, t, J 8.0 Hz, H-4), 7.32 (IH, dd, J 7.7 and 0.6 Hz, H-5), 4.31 (2H, q, J 7.1 Hz, CH2), 1.35 (3H, t, J 7.1 Hz, CH3).
1.1.1.2. 5-Bromo-f 1,2, 4]triazolo[ 1 ,5-a] pyridin-2-ylamine (3)
[00158] To a suspension of hydroxylamine hydrochloride (101.8 g, 1.465 mol) in EtOH/MeOH (1 : 1, 900 mL) was added NN-diisopropylethylamine (145.3 mL, 0.879 mol) and the mixture was stirred at room temp. (20 °C) for 1 h. l-(6-Bromo-pyridin-2-yl)-3-carboethoxy-thiourea (2) (89.0 g, 0.293 mol) was then added and the mixture slowly heated to reflux (Note: bleach scrubber was required to quench H2S evolved). After 3 h at reflux, the mixture was allowed to cool and filtered to collect the precipitated solid. Further product was collected by evaporation in vacuo of the filtrate, addition of H20 (250 mL) and filtration. The combined solids were washed successively with H20 (250 mL), EtOH/MeOH (1 : 1, 250 mL) and Et20 (250 mL) then dried in vacuo to afford the triazolopyridine derivative (3) as a solid. The compound was used as such in the next step without any purification.
[00159] lH (400 MHz, DMSO-i¼) δ 7.43-7.34 (2H, m, 2 x aromatic-H), 7.24 (1H, dd, J 6.8 and 1.8 Hz, aromatic-H), 6.30 (2H, br, NH2); m/z 213/215 (1 : 1, M+H+, 100%).
1.1.2. Synthesis of 4-[ 4-(4, 4, 5, 5-Tetramethyl-f 1, 3,2] ‘ dioxaborolan-2-yl) -benzyl] ‘- thiomor holine- 1, 1 -dioxide (Intermediate 4)
[00160] 2-(4-Bromomethyl-phenyl)-4,4,5,5-tetramethyl-[l,3,2]dioxaborolane (1 eq) and DIPEA (2 eq) were dissolved in DCM/MeOH (5:1 v:v) under N2 and thiomorpholine 1,1 -dioxide (2 eq) was added portion wise. The resulting solution was stirred at room temperature for 16h. After this time, the reaction was complete. The solvent was evaporated. The compound was extracted with EtOAc and water, washed with brine and dried over anhydrous MgSO i. Organic layers were filtered and evaporated. The final compound was isolated without further purification.
1.1.3. Synthesis of 5-[4-(l, l-Dioxothiomorpholin-4-ylmethyl)-phenyl]-[l,2,4]triazolo[l,5- a ridin-2-ylamine (Formula I)
[00161] 4-[4-(4,4,5,5-Tetramethyl-[l,3,2]dioxaborolan-2-yl)-benzyl]-thiomorpholine-l,l-dioxide (l .leq.) was added to a solution of 5-bromo-[l,2,4]triazolo[l,5-a]pyrid in-2-ylamine (4: 1). K2CO3 (2 eq.) and PdC^dppf (0.03 eq.) were added to the solution. The resulting mixture was then heated in an oil bath at 90°C for 16h under N2. Water was added and the solution was extracted with ethyl acetate. The organic layers were dried over anhydrous MgSC>4 and evaporated in vacuo. The final compound was obtained after purification by flash chromatography.
[00162] lH (400 MHz, CDC13) δ 7.94-7.92 (d, 2H), 7.52-7.48 (m, 3H), 7.37-7.34 (m, 1H), 7.02-7.00 (m, 1H), 6.00 (d, 2H), 3.76 (d, 2H), 3.15-3.13 (m, 4H), 2.93-2.91 (m, 4H).
[00163] m/z 358.2 (M+H+, 100%). 1.2. Route 2
1.2.1. Cyclopropanecarboxylic acid {5-[4-(l, l-dioxo-thiomorpholin-4-ylmethyl)-phenylJ- [l,2,4]triazolo[l,5-a]pyridin-2-yl}-amide (Formula II)
[00164] The compound according to Formula II may be synthesized according to the procedure described in WO 2010/149769.
1.2.2. Synthesis of 5-[4-(l, l-Dioxothiomorpholin-4-ylmethyl)-phenyl]-[l,2,4]triazolo[l,5- aJpyridin-2-ylamine (Formula I)
[00165] The compound according to Formula I can also be produced by hydrolysis of the compound accor ing to Formula II:
[00166] Hydrochloric acid 30% aq (12.06 kg; 3.9 rel. volumes) was added to a slurry of the compound according to Formula II (3.45 kg; 1.0 equiv.) in demineralized water (10.0 kg; 3.0 rel. volumes). Subsequently, a line rinse was performed with demineralized water (3.4 kg; 1.0 rel. volumes). The reaction mixture was heated to 80±5°C for 14.5 h. After completion of the reaction (conversion > 99%>), the reaction mixture was cooled to 20±5°C. The reaction mixture was diluted with demineralized water (6.8 kg; 2.0 rel. volumes) and sodium hydroxide 33%> aq (9.52 kg; 3.7 rel volumes) was dosed at such a rate that the temperature of the reactor contents remained below 35°C. An additional amount of sodium hydroxide 33%> aq (2.55 kg; 1.0 rel. volumes) was needed to get the pH > 10. The product was filtered off, washed twice with demineralized water (1.5 rel. volumes) and dried under vacuum for 1 h, thus yielding the crude compound according to Formula I.
[00167] The crude compound according to Formula I (5.70 kg) was re-slurried in demineralized water (23.0 kg; 8.5 rel. volumes). Hydrochloric acid 30%> aq (1.65 kg; 0.7 rel. volumes) and demineralized water (4.3 kg; 1.6 rel. volumes) were added and the reaction mixture was stirred at 20±5°C for 45 min. As the compound according to Formula I was not dissolved completely, the reaction mixture was stirred at 45±5°C for 1 h. The reaction mixture was filtered and the residue was washed with demineralized water (2.0 kg 0.75 rel. volumes). Sodium hydroxide 33%> aq (1.12 kg; 0.6 rel volumes) was added to the filtrate. An additional amount of sodium hydroxide 33%> aq (1.01 kg) was needed to get the pH > 10. The resulting reaction mixture was stirred at 20±5°C for about 3 h. The product was filtered off, washed twice with demineralized water (4.1 kg; 1.5 rel. volumes), and twice with methyl tert-butyl ether (MTBE; 3.0 kg; 1.5 rel. volumes) and dried under vacuum for 15.5 h on the filter. The product was further dried in a vacuum oven at 40±5°C for 202 h, thus affording the desired compound according to Formula I.
Update
Scheme 1: General S nthesis of Compounds of Formula I or A

Formula A
Scheme 7.

(16) (17) (18)
(18a): R3a=R3b=R2a=R (18b): R3a=R3b=D; R2a 18c): R3a=R3b=H; R2a

References
- Namour, Florence; Diderichsen, Paul Matthias; Cox, Eugène; Vayssière, Béatrice; Van der Aa, Annegret; Tasset, Chantal; Van’t Klooster, Gerben (2015-02-14). “Pharmacokinetics and Pharmacokinetic/Pharmacodynamic Modeling of Filgotinib (GLPG0634), a Selective JAK1 Inhibitor, in Support of Phase IIB Dose Selection”. Clin Pharmacokinet. Epub ahead of print.doi:10.1007/s40262-015-0240-z.
- Van Rompaey, L; Galien, R; Van der Aar, E; Clement-Lacroix, P; Van der Aar, E; Nelles, L; Smets, B; Lepescheux, L; Cristophe, T; Conrath, K; Vandeghinste, N; Vayssiere, B; De Vos, S; Fletcher, S; Brys, R; Van’t Klooster, G; Feyen, J; Menet, C (2013-10-01). “Preclinical characterization of GLPG0634, a selective inhibitor of JAK1 for the treatment of inflammatory diseases”. J Immunol. 191(7). doi:10.4049/jimmunol.1201348.
- http://acrabstracts.org/abstracts/phase-1-and-phase-2-data-confirm-that-glpg0634-a-selective-jak1-inhibitor-has-a-low-potential-for-drug-drug-interactions/
- “Galapagos’ GLPG0634 shows excellent efficacy and safety in rheumatoid arthritis Phase II study” (PDF) (Press release). Retrieved 2015-02-26.
- “Galapagos reports that the last patient in DARWIN 1 has completed 12 weeks of treatment” (PDF) (Press release). Retrieved 2015-02-26.
- “Galapagos completes recruitment for Darwin 1 study with GLPG0634 (filgotinib) in RA”. EuroInvestor. Retrieved 2015-02-26.
- NASDAQ OMX Corporate Solutions. “Galapagos completes recruitment for Darwin 2 monotherapy study with GLPG0634 (filgotinib) in RA”. Yahoo Finance. Retrieved 2015-02-26.
| US8551980 | Nov 17, 2010 | Oct 8, 2013 | Bayer Intellectual Property Gmbh | Substituted triazolopyridines |
| US8796457 | Jun 25, 2010 | Aug 5, 2014 | Galapagos Nv | Compound useful for the treatment of degenerative and inflammatory diseases |
 |
|
| Systematic (IUPAC) name | |
|---|---|
|
N-[5-[4-[(1,1-dioxo-1,4-thiazinan-4-yl)methyl]phenyl]-[1,2,4]triazolo[1,5-a]pyridin-2-yl]cyclopropanecarboxamide
|
|
| Clinical data | |
| Routes of administration |
Oral |
| Pharmacokinetic data | |
| Biological half-life | 6 hours[1] |
| Identifiers | |
| CAS Registry Number | 1206161-97-8 |
| ATC code | L01XE18 |
| IUPHAR/BPS | 7913 |
| ChemSpider | 28189566 |
| UNII | 3XVL385Q0M |
| ChEMBL | CHEMBL3301607 |
| Chemical data | |
| Formula | C21H23N5O3S |
| Molecular mass | 425.50402 g/mol |
सुकून उतना ही देना प्रभू, जितने से जिंदगी चल जाये। औकात बस इतनी देना, कि औरों का भला हो जाये।
 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO …..FOR BLOG HOME CLICK HERE
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO …..FOR BLOG HOME CLICK HERE
 LIONEL MY SON
LIONEL MY SON
He was only in first standard in school when I was hit by a deadly one in a million spine stroke called acute transverse mylitis, it made me 90% paralysed and bound to a wheel chair, Now I keep him as my source of inspiration and helping millions, thanks to millions of my readers who keep me going and help me to keep my son happy

सुकून उतना ही देना प्रभू, जितने से
जिंदगी चल जाये।
औकात बस इतनी देना,
कि औरों का भला हो जाये।
Read all about Organic Spectroscopy on ORGANIC SPECTROSCOPY INTERNATIONAL 
/////////Galapagos, GLPG0634, Filgotinib, PHASE 2, orphan drug designation, PHASE 3, Crohn’s disease, Rheumatoid arthritis, Ulceraticolitis
ve SMILES code: O=C(C1CC1)NC2=NN3C(C4=CC=C(CN5CCS(CC5)(=O)=O)C=C4)=CC=CC3=N2
Sacubitril

Sacubitril, AHU 377
NEPRILYSIN INHIBITOR
FOR HEART FAILURE
CAS 149709-62-6
CAS SODIUM SALT 149690-05-1
(2R,4S)-5-(biphenyl-4-yl)-4-((3-carboxypropionyl)amino)-2-methylpentanoic acid ethyl ester
5-(Biphenyl-4-yl)-4(S)-(3-carboxypropionamido)-2(R)-methylbutyric acid ethyl ester
N-(3-carboxy-1-oxopropyl)-(4S)-(p-phenylphenylmethyl)-4-amino-2R-methyl butanoic acid ethyl ester
[1,1′-Biphenyl]-4-pentanoic acid, γ-[(3-carboxy-1-oxopropyl)amino]-α-methyl-, α-ethyl ester, (αR,γS)-
- [1,1′-Biphenyl]-4-pentanoic acid, γ-[(3-carboxy-1-oxopropyl)amino]-α-methyl-, ethyl ester, [S-(R*,S*)]-
- (2R,4S)-4-[(3-Carboxy-1-oxopropyl)amino]-4-[(p-phenylphenyl)methyl]-2-methylbutanoic acid ethyl ester
- (2R,4S)-5-(Biphenyl-4-yl)-4-[(3-carboxypropionyl)amino]-2-methylpentanoic acid ethyl ester
-
Formula C24H29NO5 MW 411.49 g/mol
AHU377; AHU-377; Sacubitril; 149709-62-6; UNII-17ERJ0MKGI; Alpha-ethyl (alphaR,gammaS)-gamma-<(3-carboxy-1-oxopropyl)amino>-alpha-methyl<1,1′-biphenyl>-4-pentanoate
Sacubitril sodium
149690-05-1
Sacubitril is an antihypertensive drug used in combination with valsartan. The combination drug, valsartan/sacubitril, known during trials as LCZ696 and marketed under the brand name, Entresto, is a treatment for heart failure.[1] It was approved under the FDA’spriority review process for use in heart failure on July 7, 2015.


Mechanism of action
Sacubitril is a prodrug that is activated to LBQ657 by de-ethylation via esterases.[2] LBQ657 inhibits the enzyme neprilysin,[3] which is responsible for the degradation of atrial and brain natriuretic peptide, two blood pressure lowering peptides that work mainly by reducing blood volume.[4]

SYNTHESIS
CLICK ON IMAGE FOR CLEAR VIEW
SYNTHESIS

WO-2008031567
http://www.google.com/patents/WO2008031567A1?cl=en
the following steps:
and optionally the following additional steps:
Example 1 :
(E)-(R)-5-biphenyl-4-yl-4-fert-butoxycarbonylamino-2-methylpent-2-enoic acid
(E)-(R)-5-Biphenyl-4-yl-4-tert-butoxycarbonylamino-2-methylpent-2-enoic acid ethyl ester (CAS# 149709-59-1) is hydrolysed using lithium hydroxide in ethanol to yield (£)-(f?)-5-biphenyl-4-yl-4-te/t-butoxycarbonylamino-2-methylpent-2-enoic acid as a white solid. δH (400 MHz; DMSO) 1.31 (9H1 s, (CH3J3), 1.59 (3H, s, 1- CH3), 2.68 (1H, dd, J 6.8, 13.2, 5-HA), 2.86 (1H, m, 5-HB), 4.44 (1H, m, 4-H), 6.51 (1H1 d, J 9.2, 3-H), 7.16 (1H, d, J 8.0, NH), 7.26 (2H, d, J 8.0, Ar-ortho- H(Ph)), 7.31 (1H, t, J 7.6, Ar-(Ph)-para-H), 7.40 (2H, t, J 8.0, Ar-(Ph)-metø-H), 7.54 (2H, d, J 8.0, Ar-mefa-H(Ph), 7.60 (2H, d, J 7.6, Ar-(Ph)-ort/vo-H), 12.26 (1H, s, CO2H); m/z (+ESI) 404 ([MNa]+, 17%), 382 ([MHf, 2), 326 (10), 264 (100), 167 (13).
Example 2:
(2/?,4S)-5-biphenyl-4-yl-4-fert-butoxycarbonylamino-2-methylpentanoic acid in crystalline form [2(i-a)]
2(i-a) To a suspension of (E)-(f?)-5-biphenyl-4-yl-4-te/t-butoxycarbonylamino-2- methylpent-2-enoic acid [2(ii-a)] (200 g, 524.3 mmol) in degassed ethanol (900 ml) at 40 °C a solution of diiodo(p-cymene)ruthenium(ll) dimer (0.052 g, 0.0524 mmol) and (αf?,αf?)-2,2>-bis(α-Λ/,Λ/-dimethylaminophenylmethyl)-(S,S)- 1 ,1′-bis[di(3,5-dimethyl-4-methoxyphenyl)phosphine]ferrocene (= Mandyphos SL-M004-1) (0.116 g, 0.110 mmol) is added in degassed ethanol (100 ml). The solution is degassed using vacuum and a pressure of 20 bar hydrogen applied. The mixture is stirred at 40 0C for 6 h. Vessel is then purged with nitrogen. Ethanol (700 ml) is removed by distillation, lsopropyl acetate (600 ml) is added. Solvent (600 ml) is removed by distillation, lsopropyl acetate (600 ml) is added. Solvent (600 ml) is removed by distillation, lsopropyl acetate (300 ml) is added and the solution is heated to reflux. Heptane fraction (1200 ml) is added and the mixture is cooled to room temperature. The solid is collected by filtration and washed with heptane fraction-isopropyl acetate 2 : 1 mixture (360 ml). The solid is dried overnight at 50 0C under 1-50 mbar vacuum to afford the title compound as a white/off-white solid [Ratio 2(i-a) : 2(i-b) 99 : 1, as determined by HPLC analysis]. Mpt 146-147 0C; δH (500 MHz; DMSO) 1.07 (3H1 d, J 7.0, 1-CH3), 1.34 (9H, s, (CH3)3), 1-38 (1H, m, 3-HA), 1.77 (1H, m, 3-HB), 2.43 (1H, m, 2-H), 2.70 (2H, d, J 7.0, 5-H)1 3.69 (1 H, m, 4-H), 6.74 (1 H, d, J 9.0, NH)1 7.27 (2H, d, J 8.0, Ar-ortA;o-H(Ph)), 7.36 (1H, t, J 7.0, Ar-(Ph)-para-H), 7.46 (2H, t, J 7.5, Ar-(Ph)- meta-H), 7.57 (2H, d, J 8.0, Ar-mefa-H(Ph), 7.64 (2H, d, J 7.5, Ar-(Ph)-orfΛo-H), 12.01 (1H, s, CO2H); δc (500 MHz, DMSO) 18.1 (1-CH3), 28.3 [(CH3)3], 35.9 (2- C), 37.9 (3-C), 40.7 (5-C), 50.0 (4-C), 77.4 [(C(CH3)3], 126.3, 126.5, 127.2, 128.9, 129.8 (Ar-CH), 137.7 (Ar-/pso-C(Ph)), 138.3 (Ar-para-C(Ph)), 140.1 (Ar- (Ph)-/pso-C), 155.2 (NCO), 177.2 (CO2H); mlz (+ESI) 406 ([MNa]+, 6%), 384 ([MH]+, 31 ), 328 (100), 284 (19); Found: [MH]+, 384.21691. C23H30NO4 requires MH 384.21693
PATENT
http://www.google.com/patents/EP0555175A1
Example 1….THE SODIUM SALT
To a solution of N-(3-carbo(t)butoxy-1-oxopropyl)-(4S)-(p-phenylphenylmethyl)-4-amino-2R-methylbutanoic acid ethyl ester (0.80 g) in 15 ml of CH2CI2 at room temperature are added 3 ml of trifluoroacetic acid. The mixture is stirred overnight and concentrated. The residue is dissolved in tetrahydrofuran (THF), and 6.5 ml of 1 N NaOH is added. The mixture is concentrated and triturated with ether. The solid can be recrystallized from methylene chloride-hexane to give sodium N-(3-carboxy-1-oxopropyl)-(4S)-(p-phenylphenylmethyl)-4-amino-2R-methyl butanoic acid ethyl ester melting at 159-160°C; [a]D20 = – 11.4° (methanol).
The starting material is prepared as follows:
A solution of a-t-BOC-(R)-tyrosine methyl ester (5.9 g, 20 mmol) and pyridine (8 mL, 100 mmol) in methylene chloride (30 mL) is cooled to 0-5°C. Trifluoromethanesulfonic anhydride (4 mL, 23 mmol) is added at 0-5°C, and the resulting mixture is held for another 30 minutes. The reaction mixture is diluted with water (60 mL) and methylene chloride (100 mL), and washed sequentially with 0.5 N sodium hydroxide solution (1 x 50 mL), water (1 x 60 mL), 10% citric acid solution (2 x 75 mL) and water (1 x 60 mL). The organic phase is dried over MgS04 and concentrated to an oil. The oil is purified by column chromatography (silica gel, hexane/ethyl acetate, 2:1 to give methyl(R)-2-(t-butoxycarbonylamino)-3-[4-(trifluoromethylsulfonyloxy)phenyl]-propionate which crystallizes on standing; m.p. 46-48°C; [a]20 D-36.010 (c=1, CHCI3).
Nitrogen is passed through a suspension of (R)-2-(t-butoxycarbonylamino)-3-[4-(trifluoromethylsulfonyloxy)-phenyl]-propionate (1.75mmol), phenylboronic acid (3.5 mmol), anhydrous potassium carbonate (2.63 mmol) and toluene (17 mL) for 15 minutes. Tetrakis(triphenyiphosphine)paiiadium(0) is added, and the mixture is heated at 85-90° for 3 hours. The reaction mixture is cooled to 25°C, diluted with ethyl acetate (17 mL) and washed sequentially with saturated sodium bicarbonate (1 x 20 mL), water (1 x 20 mL), 10% citric acid (1 x 20 mL), water (1 x 20 mL) and saturated sodium chloride solution (1 x 20 mL). The organic phase is concentrated, and the residue is purified by column chromatography (silica gel, hexane/ethyl acetate 2:1) to yield methyl (R)-2-(t-butoxycarbonylamino)-3-(p-phenylphenyl)-propionate which can also be called N-(R)-t-butoxycarbonyl-(p-phenylphenyl)-alanine methyl ester.
To a solution of N-(R)-t-butoxycarbonyl-(p-phenylphenyl)-alanine methyl ester (6.8 g) in 60 ml of THF and 20 ml of methanol are added 20 ml of aqueous 1 N sodium hydroxide solution. The mixture is stirred for 1 h at room temperature and then acidified with 21 ml of 1 N hydrochloric acid. The aqueous solution is extracted 3x with ethyl acetate. The combined organic extracts are dried (MgS04), filtered and concentrated to give N-(R)-t-butoxycarbonyl-(p-phenylphenyl)-alanine, m.p. 98-99°C; [a]2°D -18.59° (c=1, methanol).
To a solution of N-(R)-t-butoxycarbonyl-(p-phenylphenyl)-alanine (4.8 g) in 70 ml of methylene chloride (CH2CI2) at 0°C with 1.65 g of N,O-dimethylhydroxylamine HCI, 1.7 g of triethylamine and 2.85 g of hydroxybenzotriazole are added 5.37 g of 1-[3-(dimethylamino)propyl]-3-ethylcarbodiimide hydrochloride. The mixture is stirred 17 h at room temperature. The mixture is concentrated taken up in ethyl acetate (EtOAc) and washed with saturated sodium bicarbonate, 1N HCI and brine, then dried (MgS04), filtered and concentrated to give N-(R)-t-butoxycarbonyl-(p-phenylphenyl)-alanine N,O-dimethyl hydroxylamine amide.
To a 0°C solution of N-(R)-t-butoxycarbonyl-(p-phenylphenyl)-alanine N,O-dimethyl hydroxylamine amide (5.2 g) in 250 ml of diethyl ether are added 0.64 g of lithium aluminum hydride. The reaction is stirred for 30 min. and quenched with aqueous potassium hydrogen sulfate. The mixture is stirred for additional 5 min., poured onto 1N HCI, extracted (3x) with EtOAc, dried (MgS04), filtered, and concentrated to give N-(R)-4-t-butoxycarbonyl-(p-phenylphenyl)-alanine carboxaldehyde as a colorless oil.
To a 0°C solution of N-(R)-t-butoxycarbonyl-(p-phenylphenyl)-alanine carboxaldehyde (4.4 g) in 200 ml of CH2CI2are added 10 g of carboethoxyethylidene phenyl phosphorane. The mixture is warmed to room temperature, stirred for 1 h, washed with brine, dried (MgS04), filtered and concentrated. The residue is chromatographed on silica gel eluting with (1:2) ether:hexane to give N-t-butoxycarbonyl-(4R)-(p-phenylphenylme- thyl)-4-amino-2-methyl-2-butenoic acid ethyl ester.
A solution of N-t-butoxycarbonyl-(4R)-(p-phenylphenylmethyl)-4-amino-2-methyl-2-butenoic acid ethyl ester (4.2 g) in 400 ml of ethanol is suspended with 2.0 g of 5% palladium on charcoal and then is hydrogenated at 50 psi for 6h. The catalyst is removed by filtration and the filtrate is concentrated to give N-t-butoxycarbonyl(4S)-(p-phenylphenylmethyl)-4-amino-2-methylbutanoic acid ethyl ester as a 80:20 mixture of diastereomers.
To the N-t-butoxycarbonyl(4S)-(p-phenylphenylmethyl)-4-amino-2-methylbutanoic acid ethyl ester (4.2 g) in 40 ml of CH2CI2 at 0°C is bubbled dry hydrogen chloride gas for 15 min. The mixture is stirred 2 h and concentrated to give (4S)-(p-phenylphenylmethyl)-4-amino-2-methylbutanoic acid ethyl ester hydrochloride as a 80:20 mixture of diastereomers.
To a room temperature solution of the above amine salt (3.12 g) in 15 ml of CH2CI2 and 15 ml of pyridine are added 13.5 g of succinic anhydride. The mixture is stirred for 17 h, concentrated, dissolved in ethyl acetate, washed with 1N HCI and brine, and dried (MgS04) to give N-(3-carboxy-1-oxopropyl)-(4S)-(p-phenylphenyl- methyl)-4-amino-2-methylbutanoic acid ethyl ester as a 80:20 mixture of diastereomers.
The above N-(3-carboxy-1-oxopropyl)-(4S)-(p-phenylphenylmethyl)-4-amino-2-methylbutanoic acid ethyl ester diastereomeric mixture (3.9 g) and N,N-dimethylformamide-di-t-butyl acetal (8.8 ml) are heated at 80°C in 40 ml of toluene for 2 h. The mixture is poured onto ice- 1N HCI, extracted with ether, chromatographed on silica gel eluting with (2:1) toluene:ethyl acetate to give N-(3-carbo(t)butoxy-1-oxopropyl)-(4S)-(p-phenylphe- nylmethyl)-4-amino-2R-methylbutanoic acid ethyl ester as the more polar material and the corresponding (S,S) diastereomer as the less polar material.
Example 2………THE ACID
To a solution of N-(3-carboxy-1-oxopropyl)-(4S)-(p-phenylphenylmethyl)-4-amino-(2R)-methylbutanoic acid ethyl ester (0.33 g) in 20 ml of (1:1) ethanol:tetrahydrofuran (THF) at room temperature are added 5 ml of 1 N sodium hydroxide solution (NaOH) and stirred for 17 h. The mixture is concentrated, dissolved in water and washed with ether. The aqueous layer is acidified with 1 N hydrochloric acid (HCI), extracted 3x with ethyl acetate (EtOAc), dried over magnesium sulfate (MgS04), filtered and concentrated. The residue is triturated with ether to yield N-(3-carboxy-1-oxopropyl)-(4S)-(p-phenylphenylmethyl)-4-amino-(2R)-methylbutanoic acid melting at 158-164°C, [α]D 20= -23.5° (methanol).
PATENT
CN 104557600
http://www.google.com/patents/CN104557600A?cl=zh
United States Patent US5217996 and international patent W02008031567, W02010136474 and W02012025501 reported a synthetic route follows to the chiral amino alcohols as raw materials, oxidized to the aldehyde, Victoria ladder tin reaction, chiral hydrogenation and amidation condensation reaction to obtain the objective product.
In addition, the international patent TO2008083967, TO2011088797, TO2012025502 and TO2014198195 reported that another type of preparation. The route through the 2-oxo-proline as raw material, carboxyl activating biphenyl substituted carbonyl reduction, chiral methylation, ring-opening reaction and amide condensation reaction to obtain the objective product.
Example Eight:
in the reaction flask was added (2R, 4S) -2- methyl-4-amino -5- (l, P- biphenyl-4-yl) – pentanoic acid ethyl ester (VII) (1.55g, 5mmol ), Jie of pyridine (1.2g, 15mmol) and dichloromethane burning 25mL, stirring to dissolve, butyric anhydride (1.0g, 10mmol), was heated to 4〇-45 ° C, the reaction was stirred for 6 hours. Fill Gaudin anhydride (0. 5g, 5mmol), the reaction was continued for 4 hours and the end of the reaction by TLC. Concentrated under reduced pressure, the residue was recrystallized from ethyl acetate and n-hexane to give an off-white solid sand sacubitril Kubica song (I) L 6g, a yield of 77.9%;
1H NMR (CDCl3) S 7.51 (d, 2H), 7.46 ( d, 2H), 7.36 (m, 2H), 7. 27 (m, 1H), 7. 17 (d, 2H), 5. 72 (d, 1H), 4. 19 (brs, 1H), 4. 06 (q, 2H), 2. 87-2. 72 (m, 2H), 2. 62-2. 54 (m, 2H), 2. 49 (brs, 1H), 2. 43-2. 33 ( m, 2H), I 88 (m 1H), I 54-1 43 (m, 1H), I. 18 (t, 3H), l 10 (d, 3H);…..
FAB-MSm / z : 412 [M + H] +.
PATENT
http://www.google.com/patents/WO2014198195A1?cl=en
Example 7
(2 Standby

Acetyl chloride (1 mL) 0 ° C was added ethanol (10 mL), and at room temperature for 0.5 hours, the compound (3R, 5S) -5- biphenyl-4-methyl-1- (2,2- methyl-propionyl) -3-methyl pyrrolidone (520 mg, 1.49 mmol), the reaction was refluxed for 3 days. After cooling to room temperature, and concentrated. The reaction mixture was dissolved in 8 mL of dichloromethane and pyridine 1: 1 mixed solution, and then butyryl anhydride (223 mg, 2.23 mmol). 30 ° C overnight. LC-MS detection, a small amount of starting material remaining, fill Gading anhydride (75 mg, 0.74 mmol), continue to reflect four hours. Concentrated and reverse phase column chromatography to give a white foam solid (2R, 4S) -5- biphenyl-4-yl-4- (3-carboxy – propionylamino) -2-methyl – acetic acid ester a (355 mg, 58%) and white solid (2R, 4S) -5- biphenyl-4-yl-4- (3-carboxy – propionylamino) -2-methyl – pentanoic acid b ( 13 mg, 2.3%).
a: 1H MR (400 MHz, CDCl 3 ) [delta] 7.51 (d, = 7.8 Hz, 2H), 7.46 (d, = 7.8 Hz, 2H), 7.36 (t, J = 7.6 Hz, 2H), 7.27 (t, J = 7.2 Hz, IH), 7.17 (d, J = 7.9 Hz, 2H), 5.72 (d, J = 8.1 Hz, IH), 4.19 (brs, IH), 4.06 (q, J = 7.0 Hz, 2H) , 2.87-2.72 (m, 2H), 2.62-2.54 (m, 2H), 2.49 (brs, IH), 2.43-2.33 (m, 2H), 1.88 (ddd, = 13.2, 9.5, 3.9 Hz, IH), 1.54-1.43 (m, IH), 1.18 (t, = 7.0 Hz, 3H), 1.10 (d, = 7.2 Hz, 3H).
LC-MS: t R = 3.43 min; [M + H] +: 412.0.
……………………
Paper
JOURNAL OF MEDICINAL CHEMISTRY, vol. 38, no. 10, 1995, pages 1689-1700,
http://pubs.acs.org/doi/pdf/10.1021/jm00010a014
NOTE———–DIACID
(aR,yS)-y-[ (3-Carbo-1-oxopropyl)aminol-a-methyl- [l,l’-biphenyllpentanoic Acid (21a).
To the sodium salt of 19a (0.73 g, 1.68 mM) in 20 mL of THF:EtOH was added 1 N NaOH (5.0 mL, 5.0 “01). The reaction mixture was stirred overnight and then washed with ether. The aqueous layer was acidified with 1 N HCI, re-extracted with EtOAc (3 x 10 mL), dried (MgSO& and evaporated to dryness. The solid was recrystallized from ethanol to yield 435 mg of 21a DIACID OF SACUBITRIL
melting at 165-167 “C:
[a] D25~ -28.73 (c = 10.1 in MeOH);
‘H NMR, DMSOD6
PPM 12.0, (s, 2H), 7.75 (d, 1H), 7.62 (d, 2H), 7.55 (d, 2H), 7.45 (t, 2H), 7.32 (t, lH), 7.25 (d, 2H), 4.92 (m, lH), 2.70 (d, 2H), 2.35 (t, 3H), 2.25 (m, 2H), 1.75 (m, lH), 1.32 (m, lH), 1.03 (d, 3H).
Anal. (C22H25N05) C,H,N
Note diacid is sacubitrilat (LBQ657)
NMR PREDICT
1H NMR PREDICT
13C NMR PREDICT
COSY PREDICT
…………….

NMR…..http://www.chemietek.com/Files/Line3/CHEMIETEK,%20AHU-377%20,%20Lot%2001,%20NMR-MeOD,%201.1.pdf
- Mol. Formula:C24H29NO5 ∙ C4H11NO3
- MW:532.6
- HPLC………http://www.chemietek.com/Files/Line2/CHEMIETEK,%20AHU-377%20,%20Lot%2001,%20HPLC.pdf
update………
PATENT
WO2016180275
Heart failure is a very high mortality syndrome, for patients with heart failure, so far no drug can significantly improve mortality and morbidity, and thus a new type of therapy is necessary. AHU-377 (CAS No. 149709-62-6) is an enkephalinase inhibitor, which is a prodrug ester groups can be lost through hydrolysis, converted to pharmaceutically active LBQ657, inhibit endorphin enzyme (NEP) the role of the main biological effects of NEP is to natriuretic peptides, bradykinin and other vasoactive peptide degradation failure. AHU-377 and angiotensin valsartan composition according to the molar ratio of 1 LCZ696. LCZ696 is an angiotensin receptor enkephalinase inhibitors, which can lower blood pressure, treat heart failure may become a new drug. Clinical data show, LCZ696 is more effective for the treatment of hypertension than valsartan alone.
Patents US 5,217,996 and US 5,354,892 reported the first synthesis of AHU-377, the synthetic route is as follows:
Reaction with unnatural D-tyrosine derivative as a substrate, more expensive, while the second step in the synthesis is necessary to use Pd-catalyzed Suzuki coupling reaction, whereby preparative route costs than the AHU-377 high.
Patent US 8,115,016 above routes also reported the departure from the pyroglutamate, through multi-step process for preparing a reaction AHU-377, which is more difficult methylation reaction, and the yield is not high. Patent US 8,580,974 also reported a carbonyl group of the a- introducing N, N- dimethyl enamine is converted to methyl, however, there are some problems in the route for constructing methyl chiral centers, are not suitable for scale-up synthesis route as follows:
About the latest AHU377 synthesis intermediates, Patent WO2014032627A1 reported using a Grignard reagent to react with epichlorohydrin, a quicker been important intermediates, synthetic route Compound AHU377 synthesized as follows:
However, the second step of the synthetic route use succinimide nitrogen atoms introduced by Mitsunobu reaction with hydrochloric acid hydrolysis to remove, then converted to Boc protected at the end of the synthesis process AHU377 Boc will have to take off protection, then any connection with succinic anhydride reaction product introduced into the structure of succinic acid portion, so that this method of atom economy and the economy of the steps are low.
Example 1
Synthesis of Compound 2
In inert atmosphere, a solution of three 500mL flask was added compound 1 (10g, 1eq), dissolved after 90mL THF, was added CuI (4.814g, 0.1eq), the system moves to the low temperature in the cooling bath to -20 ℃ when, biphenyl magnesium bromide dropwise addition, the internal temperature was controlled not higher than -10 ℃. Bi closed refrigeration drop, return to room temperature overnight. Completion of the reaction, the reaction solution was poured into saturated the NH 4 of Cl (10vol, 100 mL) was stirred at room temperature for 0.5h. Suction filtered, the filter cake was rinsed with a small amount of EA, and the filtrate was transferred to a separatory funnel carved, and the aqueous phase was extracted with EA (10vol × 2,100mL × 2) and the combined organic phases with saturated NaHC [theta] 3 , the NH 4 of Cl, each Brine 150mL (15vol) washed once, dried over anhydrous over MgSO 4 dried, suction filtered, and concentrated to give a white solid. Product obtained was purified by column 15.2g, yield 78%.
NMR data for the product are as follows:
1 the H NMR (400MHz, CDCl 3 ) [delta] 7.57 (D, J = 7.6Hz, 2H), 7.52 (D, J = 8.1Hz, 2H), 7.42 (T, J = 7.6Hz, 2H), 7.38-7.25 (m, 8H), 4.62-4.47 ( m, 2H), 4.09 (dd, J = 6.7,3.5Hz, 1H), 3.54 (dd, J = 9.5,3.5Hz, 1H), 3.43 (dd, J = 9.4 , 6.9Hz, 1H), 2.84 ( d, J = 6.6Hz, 2H), 2.38 (s, 1H).
Example 2
Synthesis of Compound 3
In an inert gas, at room temperature was added to the flask 500mL three Ph3P (18.54g, 2eq), 240mL DCM dissolution, butyryl diimide (of 6.44 g), compound 2 (15g), an ice-water bath cooling to 0 ℃ or so, was added dropwise DIAD (14mL) was complete, the reaction go to room temperature.Starting material the reaction was complete, the system was added to water (100 mL) quenched the reaction was stirred for 10min; liquid separation, the aqueous phase was extracted with DCM (100mL × 2), the combined organic phases with saturated Brine 100mL × 2), dried over anhydrous over MgSO 4 dried , filtration, spin dry to give a white solid; product was purified by column 15.4g, yield 82%.
NMR data for the product are as follows:
1 the H NMR (400MHz, CDCl 3 ) [delta] 7.56 (D, J = 7.4Hz, 2H), 7.49 (D, J = 8.0Hz, 2H), 7.42 (T, J = 7.6Hz, 2H), 7.37-7.30 (m, 3H), 7.27 ( d, J = 6.7Hz, 3H), 7.22 (d, J = 8.0Hz, 2H), 4.75 (s, 1H), 4.56 (d, J = 12.0Hz, 1H), 4.45 (d, J = 12.0Hz, 1H ), 4.06 (t, J = 9.6Hz, 1H), 3.70 (dd, J = 10.0,5.2Hz, 1H), 3.23 (dd, J = 13.8,10.3Hz, 1H) , 3.14-3.00 (m, 1H), 2.48 (d, J = 4.0Hz.4H).
Example 3
Synthesis of Compound 4
Protection of inert gas, at room temperature was added to the flask 1L three compound 3 (18.81g), 470mL EtOH was dissolved, was added Pd / C, replaced the H 2 three times, move heated on an oil bath at 60 ℃ reaction. Raw reaction was complete, the system was removed from the oil bath, the reaction solution was suction filtered through Celite and concentrated to give the crude product. It was purified by column pure 11.8g, a yield of 81.2%.
NMR data for the product are as follows:
1 the H NMR (400MHz, CDCl 3 ) [delta] 7.57 (D, J = 7.8Hz, 2H), 7.51 (D, J = 7.8Hz, 2H), 7.42 (T, J = 7.5Hz, 2H), 7.33 (T , J = 7.2Hz, 1H), 7.26 (d, J = 7.2Hz, 2H), 4.55 (d, J = 5.2Hz, 1H), 4.06-3.97 (m, 1H), 3.86 (dd, J = 12.0, 3.1Hz, 1H), 3.16 (dd , J = 8.1,2.9Hz, 2H), 2.58 (t, J = 7.0Hz, 4H), 1.26 (s, 2H).
Example 4
Synthesis of Compound 7
Protection of inert gas, at room temperature to a 25mL flask was added three Dess-Martin oxidant (767.7mg), 10mL DCM was dissolved, the system was cooled down to -10 deg.] C, was added 4 (500mg). Starting material the reaction was complete, to the system was added saturated NaHCO3 and Na2S2O3 each 5mL, quench the reaction stirred for 10min; aqueous phase was extracted with DCM (10mL × 3) and the combined organic phases with saturated NaHCO3, Brine 30mL each wash, dried over anhydrous MgSO4, filtration, spin dried to give the crude product used directly in the next reaction cast.
Example 5
Synthesis of Compound 8
Inert gas, at room temperature for three to 500mL flask 7 (497.5mg), 10mL DCM to dissolve an ice water bath to cool, added phosphorus ylide reagent (880.6mg), the system was removed from the ice water bath at room temperature. The reaction material completely stop the reaction, the system was added to water (5mL) to quench the reaction. Liquid separation, the aqueous phase was extracted with DCM (10mL × 2), organic phases were combined, washed with saturated Brine 20mL × 2, dried over anhydrous MgSO4, filtration, spin crude done. Product obtained was purified by column 563mg, 90% yield.
NMR data for the product are as follows:
1 the H NMR (400MHz, CDCl 3 ) δ7.60-7.53 (m, 2H), 7.51 (D, J = 8.1Hz, 2H), 7.42 (T, J = 7.6Hz, 2H), 7.33 (D, J = 7.3Hz, 1H), 7.23 (d , J = 8.1Hz, 2H), 7.13 (dd, J = 9.2,1.5Hz, 1H), 5.26 (td, J = 9.5,6.9Hz, 1H), 4.25-4.05 ( m, 2H), 3.40 (dd , J = 13.7,9.7Hz, 1H), 3.13 (dd, J = 13.8,6.7Hz, 1H), 2.53 (d, J = 2.2Hz, 4H), 1.85 (d, J = 1.4Hz, 3H), 1.30 ( t, J = 7.1Hz, 3H).
Example 6
Synthesis of Compound 9
Protection of inert gas, at room temperature to a 50mL flask was added three 8 (365mg, 1eq), 9mL of ethanol and stirred to dissolve, the system was replaced with hydrogen three times, was added Pd / C (25% w / w) at room temperature. The reaction material completely stop the reaction, the system was added to water (5mL) to quench the reaction. The reaction mixture was suction filtered through Celite and concentrated to give the crude product. Product was purified by column, yield 80.2%, purity 97.2%.
Example 7
Synthesis of Compound 10
Equipped with Compound 9 (100mg) acetic acid A reaction flask (9mL), hydrochloric acid (1mL). The reaction was heated oil bath at 80 deg.] C. The reaction material completely stop the reaction, the system was added to water (5mL) to quench the reaction. After saturated NaHCO3 and extracted with EA and concentrated to give crude product. Product obtained was purified by column 90mg, yield 84%.
NMR data for the product are as follows:
1 the H NMR (400MHz, CDCl 3 ) δ7.61-7.54 (m, 2H), 7.53-7.48 (m, 2H), 7.41 (dd, J = 10.5,4.9Hz, 2H), 7.31 (dd, J = 8.3 , 6.4Hz, 1H), 7.22 ( d, J = 8.2Hz, 2H), 5.93 (t, J = 9.7Hz, 1H), 4.34-4.00 (m, 3H), 2.91-2.71 (m, 2H), 2.68 -2.57 (m, 2H), 2.55 (ddd, J = 9.4,7.0,4.3Hz, 1H), 2.42 (dt, J = 13.3,6.8Hz, 2H), 1.97-1.74 (m, 1H), 1.64-1.46 (m, 1H), 1.23 ( td, J = 7.1,3.3Hz, 3H), 1.14 (dd, J = 7.1,3.9Hz, 3H)
Example 8
Synthesis of Compound 5
Example 8-1: The reaction flask was added compound 4 (1eq) was added water (2VOL), concentrated hydrochloric acid (2VOL), 110 ℃ reaction was heated in an oil bath overnight, complete conversion of starting material, the HPLC peak area 97%. 10% NaOH solution was added to adjust the pH to about 10, filtration products. Yield 85%.
Example 8-2: The reaction flask was added compound 4 (1eq) was added ethanol (5 vol), water (5 vol), potassium hydroxide (8 eq), was heated in an oil bath overnight at 110 ℃ reaction, complete conversion of the starting material, the HPLC peak area 99%. Water was added (5Vol), filtered to obtain the product. Yield 95%. Product was dissolved in toluene, was added ethanolic hydrochloric acid, the precipitated hydrochloride Compound 5.
NMR data for the product are as follows:
1 the H NMR (400MHz, of DMSO) [delta] 8.31 (S, 3H), 7.70-7.61 (m, 4H), 7.47 (T, J = 7.6Hz, 2H), 7.42-7.31 (m, 3H), 4.09 (the dq- , J = 42.6,7.1Hz, 1H), 3.62-3.51 (m, 1H), 3.50-3.41 (m, 1H), 3.11-3.00 (m, 1H), 2.95-2.84 (m, 1H), 1.30-1.10 (m, 1H).
EXAMPLE 9
Synthesis of Compound 6
To the reactor was added compound 5, was added absolute ethanol (3vol). Temperature of the outer set 30 ℃ heating, stirring was continued after the temperature reached 25 ℃ 20min. Was added 30% NaOH aqueous solution (1.1eq). External temperature 65 ℃ heating provided, after the internal temperature reached 60 deg.] C was slowly added (of Boc) 2 O (1.1 eq). Stirring 0.5h, reaction monitoring. After completion of the reaction, water was added slowly dropwise (8vol), turn off the heating and natural cooling. The system temperature was lowered to 25 deg.] C and continue stirring for 2h. Filter cake at 50 ℃ blast oven drying to obtain the product.
NMR data of the product are as follows:
1 the H NMR (400MHz, CDCl 3 ) δ7.61-7.50 (m, 4H), 7.61-7.50 (m, 4H), 7.46-7.39 (m, 2H), 7.48-7.38 (m, 2H), 7.38-7.23 (m, 3H), 7.37-7.26 ( m, 3H), 4.82 (d, J = 7.9Hz, 1H), 4.82 (d, J = 7.9Hz, 1H), 3.91 (s, 1H), 3.70 (d, J = 11.0Hz, 1H), 3.77-3.54 (m, 2H), 3.65-3.47 (m, 1H), 2.88 (d, J = 7.0Hz, 2H), 2.88 (d, J = 7.0Hz, 2H), 2.51 (s, 1H), 2.51 (s, 1H), 1.42 (s, 9H), 1.42 (s, 9H).
Synthesis of Intermediate Compound 6 to Compound 10, i.e., the AHU-377, a synthetic route in the background of the present invention, the cited patent application WO2014032627A1 loaded in detail, not in this repeat.
Example 10
Synthesis of Compound 2
Benzyl glycidyl ether preparation (50g) in THF (200mL) was added. Under inert gas protection, the biphenyl magnesium bromide (365mmol) was added to THF (1020mL) was added the reaction flask is placed in a low temperature bath -40 ℃ cooling. Cuprous iodide (O.leq) when the internal temperature dropped to -9 ℃. Continued to decrease the temperature of -23 ℃ dropwise addition of benzyl glycidyl ether in THF was added dropwise to control the internal temperature process of not higher than -15 deg.] C, 47 min when used, the addition was completed the cooling off the reaction was stirred overnight. The cooling system to -20 ℃ quenched with 1N HCl aqueous solution, <10 ℃ Go stirred 30min at room temperature. Liquid separation, the aqueous phase was extracted with THF, the combined THF phases. Respectively saturated ammonium chloride (250mL), saturated brine (250mL) washed. Rotary evaporation to remove THF, and water (200 mL) Continue rotary evaporation 1h, cool to precipitate a solid. Suction crude. Crude n-heptane was added 2Vol beating, suction filtration to obtain the product in a yield of 90 ~ 95%, HPLC peak area 94%. In another column purification was pure, columned yield 88.6%, HPLC 99.1%.
Example 11
Synthesis of Compound 3
Preparation Example 9, said compound taking the embodiment 2 (5g) added to the reaction flask, the reaction flask was added toluene (80mL), phthalimide (2.55 g of) and triphenylphosphine (5.35g of), the nitrogen was replaced protection. An ice-salt bath cooling to -5 deg.] C, was added dropwise DIAD (4.12g), dropwise addition was exothermic, the temperature was raised to 5 ℃. The reaction was continued 1h sampling HPLC test material substantially complete reaction. Join 12g silica spin column done to collect the product (including DIEA derivative).
Example 12
Synthesis of Compound 11
Compound 3 (3g) was added to the reaction flask embodiment taken in Preparation Example 10, was added ethanol (30 mL), with stirring. Was added hydrazine hydrate (2g) was heated in an oil bath reflux 1h, when supplemented with 20mL ethanol was stirred difficulties, the reaction was continued to 2.5h, HPLC showed the starting material the reaction was complete. Add EA / H2O 100mL each liquid separation, the EA phase was washed with water (100mL) and the combined organic phases were washed with water (100mL) and saturated brine (100mL) washed. Anhydrous magnesium sulfate and filtered spin column was done product 1.88g, yield 88%, HPLC 94%.
NMR data of the product are as follows:
1 the H NMR (400MHz, of DMSO) [delta] 7.64 (D, J = 7.2Hz, 2H), 7.57 (D, J = 8.1Hz, 2H), 7.45 (T, J = 7.6Hz, 2H), 7.39-7.32 ( m, 5H), 7.29 (d , J = 8.1Hz, 3H), 4.55-4.43 (m, 2H), 3.38-3.23 (m, 3H), 3.18-3.10 (m, 1H), 2.82-2.74 (m, 1H), 2.61-2.52 (m, 1H ).
Example 13
Synthesis of Compound 11
To the toluene solution of the compound 2 was added phthalimide (1.1 eq), triphenylphosphine (1.3 eq) with stirring. External bath set -10 ℃, to cool the system, the internal temperature dropped to 0 ~ 5 ℃, start dropping DIAD (1.3eq), control the internal temperature -5 ~ 5 ℃. Completion of the dropwise addition, the cooling bath was turned off outside the reaction was stirred at room temperature. The reaction was stirred for 1 to 4 hours. The reaction solution to give compound 3, administered directly in the next reaction. To the above reaction mixture was added hydrazine hydrate (6 eq), heated to 70 ~ 80 ℃, to complete the reaction, filtered hot, the filtrate. Aqueous sodium hydroxide solution (20vol 10%) was stirred for 0.5h, allowed to stand for liquid separation from toluene phase. Water was added (20vol) was stirred for 0.5h, allowed to stand for liquid separation from toluene phase. The toluene phase was added hydrochloric acid (20vol, 3N), stirred for 0.5h, to form a solid precipitate. Filtration and drying to obtain a product, i.e. compound 11, the hydrochloride salt, yield 60% in two steps.
NMR data of the product are as follows:
1 the H NMR (400MHz, of DMSO) [delta] 8.46 (S, 3H), 7.63 (dd, J = 16.4,7.7Hz, 4H), 7.47 (T, J = 7.6Hz, 2H), 7.42-7.22 (m, 8H ), 4.56 (d, J = 12.1Hz, 1H), 4.48 (d, J = 12.1Hz, 1H), 3.58 (d, J = 7.9Hz, 2H), 3.47 (dd, J = 10.9,6.3Hz, 1H ), 3.11 (dd, J = 13.5,4.9Hz, 1H), 2.92 (dd, J = 13.4,9.1Hz, 1H).
Example 14
Synthesis of Compound 12
Weigh Compound 11 (1.38g) was added to the reaction flask. To the reaction flask plus DCM (14ml) and Et3N (462mg, 0.73ml). Weighed (of Boc) 2O (1.23 g of) was added to DCM (5ml) was dissolved. Room temperature (8 ℃), a solution (of Boc) 2 DCM solution O was added dropwise to the reaction, (2ml) rinsed with DCM. The reaction mixture was stirred at room temperature, detected by HPLC, the reaction ends 4h. Reaction mixture was washed (15ml) 3 times with Brine (15ml) The reaction solution was washed 1 times. Inorganic sulfate, concentrated and purified by column PE:EA = 15:1 give product 560mg, yield 30.8%, HPLC 99.92%.
NMR data of the product are as follows:
1 the H NMR (400MHz, CDCl 3 ) [delta] 7.57 (D, J = 7.6Hz, 2H), 7.49 (D, J = 7.4Hz, 2H), 7.43 (T, J = 7.3Hz, 2H), 7.39-7.28 (m, 5H), 7.24 ( d, J = 9.0Hz, 3H), 5.00-4.80 (br, 1H), 4.51 (q, J = 11.8Hz, 2H), 4.08-3.85 (br, 1H), 3.43 ( d, J = 2.9Hz, 2H) , 3.02-2.77 (m, 2H), 1.42 (s, 9H).
Example 15
Synthesis of Compound 6
Weigh Compound 12 (250mg) and methanol (9ml) was added to the reaction flask. Added Pd / C (138mg, 1 / 4w / w, water content 55%). The H 2replaced 3 times, 50 ℃ stirred and heated. HPLC detection reaction, the reaction end 30h. Filtered off Pd / C, 40 ℃ concentrated under reduced pressure to remove methanol. PE:EA = 3:1 florisil column to give the product 196mg, 100% yield, 99.34% purity.
NMR data of the product are as follows:
1 the H NMR (400MHz, CDCl 3 ) δ7.61-7.50 (m, 4H), 7.61-7.50 (m, 4H), 7.46-7.39 (m, 2H), 7.48-7.38 (m, 2H), 7.38-7.23 (m, 3H), 7.37-7.26 ( m, 3H), 4.82 (d, J = 7.9Hz, 1H), 4.82 (d, J = 7.9Hz, 1H), 3.91 (s, 1H), 3.70 (d, J = 11.0Hz, 1H), 3.77-3.54 (m, 2H), 3.65-3.47 (m, 1H), 2.88 (d, J = 7.0Hz, 2H), 2.88 (d, J = 7.0Hz, 2H), 2.51 (s, 1H), 2.51 (s, 1H), 1.42 (s, 9H), 1.42 (s, 9H).
Method for preparing the AHU-377, characterized by comprising the steps of: (a) Compound (1) S- benzyl glycidyl ether and biphenyl Grignard reagent produced by the reaction of the compound (2) in an organic solvent; ( b) compound (2) with a succinimide or phthalimide Mitsunobu reaction occurs in an organic solvent to form a compound (3); (C) compound (3) in an organic solvent in the role of a catalyst under removal debenzylation protected form compound (4); (D) compound (4) with an oxidizing agent oxidation reaction occurs in an organic solvent to form a compound (7); (E) compound (7) with a phosphorus ylide reagent in an organic solvent to give the compound (8); (F.) compound (8) in an organic solvent in the selective catalytic hydrogenation of the compound (9); and (g) of the compound (9) in an organic solvent in the hydrolysis reaction of the amide compound occurs in the presence of an acid ( 10), i.e., AHU-377;
References
- John J.V. McMurray, Milton Packer, Akshay S. Desai, et al. for the PARADIGM-HF Investigators and Committees (August 30, 2014).“Angiotensin–Neprilysin Inhibition versus Enalapril in Heart Failure”. N Eng J Med 371. doi:10.1056/NEJMoa1409077.
- Solomon, SD. “HFpEF in the Future: New Diagnostic Techniques and Treatments in the Pipeline”. Boston. p. 48. Retrieved 2012-01-26.
- Gu, J.; Noe, A.; Chandra, P.; Al-Fayoumi, S.; Ligueros-Saylan, M.; Sarangapani, R.; Maahs, S.; Ksander, G.; Rigel, D. F.; Jeng, A. Y.; Lin, T. H.; Zheng, W.; Dole, W. P. (2009). “Pharmacokinetics and Pharmacodynamics of LCZ696, a Novel Dual-Acting Angiotensin Receptor-Neprilysin Inhibitor (ARNi)”. The Journal of Clinical Pharmacology 50 (4): 401–414. doi:10.1177/0091270009343932.PMID 19934029.
- Schubert-Zsilavecz, M; Wurglics, M. “Neue Arzneimittel 2010/2011.” (in German)


| WO2004085378A1 * | Mar 15, 2004 | Oct 7, 2004 | Joseph D Armstrong Iii | Process for the preparation of chiral beta amino acid derivatives by asymmetric hydrogenation |
| WO2006057904A1 * | Nov 18, 2005 | Jun 1, 2006 | Merck & Co Inc | Stereoselective preparation of 4-aryl piperidine amides by asymmetric hydrogenation of a prochiral enamide and intermediates of this process |
| WO2006069617A1 * | Dec 5, 2005 | Jul 6, 2006 | Dsm Fine Chem Austria Gmbh | Process for transition metal-catalyzed asymmetric hydrogenation of acrylic acid derivatives, and a novel catalyst system for asymmetric transition metal catalysis |
| US5217996 * | Jan 22, 1992 | Jun 8, 1993 | Ciba-Geigy Corporation | Biaryl substituted 4-amino-butyric acid amides |
NON-PATENT CITATIONS
| Reference | ||
|---|---|---|
| 1 | * | KSANDER, GARY M. ET AL: “Dicarboxylic Acid Dipeptide Neutral Endopeptidase Inhibitors” JOURNAL OF MEDICINAL CHEMISTRY, vol. 38, no. 10, 1995, pages 1689-1700, XP002340280 cited in the application |
 |
|
| Systematic (IUPAC) name | |
|---|---|
|
4-{[(2S,4R)-1-(4-Biphenylyl)-5-ethoxy-4-methyl-5-oxo-2-pentanyl]amino}-4-oxobutanoic acid
|
|
| Clinical data | |
| Legal status |
|
| Identifiers | |
| CAS Registry Number | 149709-62-6 |
| ATC code | None |
| PubChem | CID: 9811834 |
| ChemSpider | 7987587 |
| Synonyms | AHU-377; AHU377 |
| Chemical data | |
| Formula | C24H29NO5 |
| Molecular mass | 411.49 g/mol |
Relevant Clinical Literature
UK Guidance
Regulatory Literature
Other Literature
 |
|
| Systematic (IUPAC) name | |
|---|---|
|
4-{[(2S,4R)-1-(4-Biphenylyl)-5-ethoxy-4-methyl-5-oxo-2-pentanyl]amino}-4-oxobutanoic acid
|
|
| Identifiers | |
| CAS Number | 149709-62-6 |
| ATC code | None |
| PubChem | CID: 9811834 |
| ChemSpider | 7987587 |
| Synonyms | AHU-377; AHU377 |
| Chemical data | |
| Formula | C24H29NO5 |
| Molecular mass | 411.49 g/mol |
Message
Comwinchem <comwinchem@foxmail.com>
Date: 1 September 2016 at 15:16
Subject: LCZ696 (SACUBITRIL+VALSARTAN)/ Changzhou Comwin Fine Chemicals Co,. Ltd
To: amcrasto <amcrasto@gmail.com>
Dear SirHow do you do! Sincerely hope my email will bring you more LCZ696 (SACUBITRIL+VALSARTAN) possibilities.I am Wang Zhuo of ComWin from China, and in charge of LCZ696 (SACUBITRIL+VALSARTAN) global marketing.
Date: 1 September 2016 at 15:16
Subject: LCZ696 (SACUBITRIL+VALSARTAN)/ Changzhou Comwin Fine Chemicals Co,. Ltd
To: amcrasto <amcrasto@gmail.com>
Dear SirHow do you do! Sincerely hope my email will bring you more LCZ696 (SACUBITRIL+VALSARTAN) possibilities.I am Wang Zhuo of ComWin from China, and in charge of LCZ696 (SACUBITRIL+VALSARTAN) global marketing.
LCZ696 (sacubitril/Valsartan) is a combination drug for use in heart failure developed by Novartis. It consists of valsartan and sacubitril, in a 1:1 mixture by molecule count. It was approved by US FDA in July 2015.
Sacubitril (Hemicalcium) is a neprilysin inhibitor, We have developed this project since 2nd half of 2014. At present, some intermediates are in commercial scale, and some are in pilot production.
We will put our most focus on this project from 2nd half of this year. Certainly we will file DMF for Sacubitril and make submission to regulartory market. We have sold Sacbutitril to some EU customers for evaluation purpose, such as Teva, Chemo. Also, we are doing the development of LCZ696 (the final API). It’s co-crystallized valsartan and sacubitril, in a one-to one molar ratio. One LCZ696 complex consists of six valsartan anions, six sacubitril anions, 18 sodium cations, and 15 molecules of water. Now we have the sample of LCZ696 in kilogram grade.
Best Regards
Wang Zhuo
Sales Executive
Changzhou ComWin Fine Chemicals Co.,Ltd.
24th Floor, Jiaye International Commercial Plaza
99 Yanling West Road, Changzhou
Jiangsu 213003 China
Tel: 0086 519 8663 2882
Fax: 0086 519 8661 3190
सुकून उतना ही देना प्रभू, जितने से जिंदगी चल जाये। औकात बस इतनी देना, कि औरों का भला हो जाये।
 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO …..FOR BLOG HOME CLICK HERE
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO …..FOR BLOG HOME CLICK HERE
 LIONEL MY SON
LIONEL MY SONHe was only in first standard in school when I was hit by a deadly one in a million spine stroke called acute transverse mylitis, it made me 90% paralysed and bound to a wheel chair, Now I keep him as my source of inspiration and helping millions, thanks to millions of my readers who keep me going and help me to keep my son happy

सुकून उतना ही देना प्रभू, जितने से
जिंदगी चल जाये।
औकात बस इतनी देना,
कि औरों का भला हो जाये।
जिंदगी चल जाये।
औकात बस इतनी देना,
कि औरों का भला हो जाये।
////// antihypertensive drug, Heart Failure, Sacubitril, AHU 377
CCOC(=O)[C@H](C)C[C@@H](Cc1ccc(cc1)c2ccccc2)NC(=O)CCC(=O)O
CCOC(=O)[C@H](C)C[C@@H](CC1=CC=C(C=C1)C2=CC=CC=C2)NC(=O)CCC(=O)O
Updating of the HMPC-Guideline on the use of the CTD Format in the Registration of Traditional Herbal Medicinal Products
DRUG REGULATORY AFFAIRS INTERNATIONAL
 Updating of the HMPC-Guideline on the use of the CTD Format in the Registration of Traditional Herbal Medicinal Products
Updating of the HMPC-Guideline on the use of the CTD Format in the Registration of Traditional Herbal Medicinal Products
Compared to herbal medicinal products (HMPs) there is a simplified registration procedure for traditional herbal medicinal products (THMP).
EMA’s HMPC (Committee on Herbal Medicinal Products) published the draft of revision 2 on the use of the CTD format in the preparation of a registration application for traditional herbal medicinal products on 10 March 2015.
This guideline contains instructions on how to prepare a CTD for a registration application of traditional herbal medicinal products.
Now, there is a new annex 2 with a mock-up which shows by means of a concrete example where and to what extent information should be given on traditional herbal medicinal products in the dossier.
Appendix 1 is a best practice guide for module 3 on quality.
For further information please see the complete draft revision 2 of the…
View original post 33 more words
Indian and Chinese API Manufacturers in the Focus of European Authorities
DRUG REGULATORY AFFAIRS INTERNATIONAL
 |
Indian and Chinese API Manufacturers in the Focus of European Authorities
The EudraGMP database was originally launched in April 2007 and is used to exchange information on compliance with the Good Manufacturing Practices (GMP) between the relevant regulatory authorities of the EU Member States – including Iceland, Liechtenstein and Norway. Since January 2011 the data of all national authorities can be accessed. Further, since April 2013 the database also contains information about GDP, why it is referred to as Eudra GMDP database now.
The database comprising the reports about deficiencies found in inspections by the European authorities – the “non-compliance reports” or, officially, “statement of non-compliance with GMP” – was extended by three reports last week: two of these reports related to Chinese firms, one report to a company in India. The inspections were conducted by inspectors of the Italian authority.
The inspection of the Indian site (antibiotic…
View original post 224 more words
MORINIDAZOLE 吗啉硝唑

MORINIDAZOLE
吗啉硝唑
(迈灵达®
1- [3- (4-morpholinyl) -2-hydroxypropyl] -2-methyl-5- nitro -1H- imidazole
CAS 92478-27-8
Jiangsu Hansoh Pharmaceutical Co., Ltd.
Morinidazole was approved by China Food and Drug Administration (CFDA) on February 24, 2014. It was developed and marketed as a step Lingda ® by Hansoh Pharmaceutical.
A nitroimidazoles antibiotic used to treat bacterial infections including appendicitis and pelvic inflammatory disease.

Morinidazole is a nitroimidazoles antibiotic indicated for the treatment of bacterial infections including appendicitis and pelvic inflammatory disease (PID) caused by anaerobic bacteria.

PATENT
WO2006058457A1.
http://www.google.com/patents/WO2006058457A1?cl=en
……………………….
PATENT
CN1981764A.
https://www.google.com/patents/CN1981764A?cl=en
1- (2,3-epoxypropoxy yl) -2-methyl-5-nitro-imidazole (10g), morpholino (10g), 100ml of acetonitrile under reflux for 2 hours, vacuum recovery of acetonitrile, water was added 100ml, heating to the whole solution, filtered hot, let cool, filtering, washing and drying to obtain an off-white solid (11g).
Proton nuclear magnetic resonance data: 1HNMR (CD3Cl) δ2.39 ~ 2.73 (6H, m) δ2.61 (3H, s) δ3.71 ~ 3.81 (4H, m) δ4.10 ~ 4.17 (2H, m) δ4 .63 ~ 4.66 (1H, m) δ8.00 (1H, s)
CN 102199147
http://www.google.com/patents/CN102199147A?cl=en
CN 1605586
https://www.google.com/patents/CN1605586A?cl=en
Example 7 Preparation of α- (morpholino-1-yl) methyl-2-methyl-5-nitroimidazole-1-ethanol according to Example 4 the same manner as in Preparation α- (morpholino-1-yl) methyl-2-methyl-5-nitroimidazole-1-ethanol, except for using morpholine instead of 4-hydroxypiperidine, prepared by the present invention Compound 7. Proton nuclear magnetic resonance data: 1HNMR (CD3Cl) δ2.39 ~ 2.73 (6H, m) δ2.61 (3H, s) δ3.71 ~ 3.81 (4H, m) δ4.10 ~ 4.17 (2H, m) δ4
NMR PREDICT
1H NMR PREDICT
13C NMR PREDICT
COSY
| CN1810815B | Mar 8, 2006 | Mar 16, 2011 | 陕西合成药业有限公司 | Nitroimidazole derivative for treatment |
| CN1903846B | Aug 15, 2006 | Jul 13, 2011 | 杨成 | Ornidazole derivative used for therapy, its preparation method and use |
| CN100387233C | Jun 9, 2006 | May 14, 2008 | 南京圣和药业有限公司 | Use of levo morpholine nidazole for preparing medicine for antiparasitic infection |
| CN100427094C | Dec 13, 2005 | Oct 22, 2008 | 江苏豪森药业股份有限公司 | Usage of alpha-(Morpholin-1-base) methyl-2-methyl-5-azathio-1-alcohol in preparation of anti-trichomoniasis and anti-ameba medicines |
| CN100540549C | Dec 15, 2005 | Sep 16, 2009 | 南京圣和药业有限公司 | Alpha-substituted-2-methyl-5-nitro-diazole-1-alcohol derivative with optical activity |
| WO2007079653A1 * | Dec 25, 2006 | Jul 19, 2007 | Junda Cen | OPTICALLY PURE α-SUBSTITUTED 2-METHYL-5-NITROIMIDAZOLE-1-ETHANOL DERIVATIVES |

ENZALUTAMIDE
Enzalutamide, MDV-3100
MDV3100 is an orally bioavailable, organic, non-steroidal small molecule targeting the androgen receptor (AR) with potential antineoplastic activity. MDV3100 (Enzalutamide) blocks androgens from binding to the androgen receptor and prevents nuclear translocation and co-activator recruitment of the ligand-receptor complex. It also induces tumour cell apoptosis, and has no agonist activity. Early preclinical studies also suggest that MDV3100 inhibits breast cancer cell growth.
1H NMR FROM THE NET
1H NMR PREDICT AND 13 C NMR PREDICT BELOW
COSY PREDICT
Synthesis pics
……………………..
PATENT
http://www.google.com/patents/WO2015063720A1?cl=en
Enzalutamide is chemically described as 4-{3-[4-cyano-3-(trifluoromethyl)phenyl] -5 ,5 -dimethyl-4-oxo-2-sulfanylideneimidazolidin- 1 -yl } -2-fluoro-N-methylbenzamide of Formula I.

FORMULA I
Processes for the preparation of enzalutamide are described in U.S. Publication Nos. 2007/0004753 and 2007/0254933 and PCT Publication Nos. WO 2007/127010, WO 2006/124118, and WO 2011/106570.
PCT Publication No. WO 2011/106570 discloses that the processes described in U.S. Publication Nos. 2007/0004753 and 2007/0254933 result in a 25% yield of enzalutamide in the final step, which accounts for a 15% overall yield. PCT Publication No. WO 2011/106570 further discloses that the known processes for preparing enzalutamide involve the use of extremely toxic reagents, for example, acetone cyanohydrin.
Acetone cyanohydrin is toxic and therefore its use as a reagent should be avoided for industrial production of a pharmaceutical ingredient. Thus, there is a need in the art to develop a process for the preparation of enzalutamide that avoids the use of acetone cyanohydrin as a reagent
Example 6: Process for the preparation of Enzalutamide (Formula I)
Ethyl N-[3-fluoro-4-(methylcarbamoyl)-phenyl]-2-methylalaninate (Formula IV; 0.2 g) and 4-isothiocyanato 2-(triflouromethyl)-benzonitrile (Formula V; 0.33 g) were added to dimethyl sulfoxide (0.2 mL) and isopropyl acetate (0.4 mL) and heated to 90°C to 95°C. The reaction mixture was cooled to 70°C followed by the addition of methanol (0.4 mL). The reaction mixture was stirred for 2 hours. Isopropyl acetate (4 mL) was added to the reaction mixture followed by washing with water (4 mL). The organic layer was concentrated at 35°C under vacuum to obtain an oily residue which was further purified using silica gel column to obtain the title compound.
Yield: 0.2 g
…………………………
PAPER
J Med Chem 2010, 53(7): 2779
http://pubs.acs.org/doi/full/10.1021/jm901488g
A structure−activity relationship study was carried out on a series of thiohydantoins and their analogues 14 which led to the discovery of 92 (MDV3100) as the clinical candidate for the treatment of hormone refractory prostate cancer.
N-Methyl-4-[3-(4-cyano-3-trifluoromethylphenyl)-5,5-dimethyl-4-oxo-2-thioxoimidazolidin-1-yl]-2-fluorobenzamide, 92
………………………..and concentrated and the residue was purified with SiO2 column chromatography (dichloromethane/acetone, 95:5) to give92 (30 mg, 51%) as colorless crystals.
1H NMR (CDCl3, 400 MHz) δ 1.61 (s, 6H), 3.07 (d, 3H, J= 4.1 Hz), 6.71 (m, 1 H), 7.15 (dd, 1H, J = 11.7, 2.0 Hz), 7.24 (dd, 1H, J = 8.4, 2.0 Hz), 7.83 (dd, 1H, J = 8.2, 2.1 Hz), 7.95 (d, 1H, J = 2.1 Hz), 7.99 (d, 1H, J = 8.2 Hz), 8.28 (dd, 1H, J = 8.4, 8.4 Hz).
13C NMR (CDCl3, 125 MHz) δ 23.8, 26.9, 66.5, 110.3, 114.6, 117.7, 117.9, 121.7 (q, J = 272.3 Hz), 126.1, 126.9 (q, J = 4.6 Hz), 132.0, 133.3, 133.6 (q, J = 33.4 Hz), 135.2, 136.7, 138.9 (d, J = 10.8 Hz), 160.3 (d, J = 248.6 Hz), 162.6 (d, J = 3.3 Hz), 174.3, 179.6.
19F NMR (CDCl3, 100 MHz) δ −111.13, −62.58.
HRMS: found 465.1023 [M + H]+, calculated for [C21H16F4N4O2S + H]+ 465.1003.
COMPARISON OF 1H NMR KNOWN VALUES WITH PREDICTED —-KNOWN IN RED
REF
MEDIVATION PROSTATE THERAPEUTICS, INC.; JAIN, Rajendra, Parasmal; ANGELAUD, Remy; THOMPSON, Andrew; LAMBERSON, Carol; GREENFIELD, Scott Patent: WO2011/106570 A1, 2011 ; Location in patent: Page/Page column 46
Regents of the University of California Patent: US2007/254933 A1, 2007 ; Location in patent: Page/Page column 7 ;
WO2011/106570 A1,
J Med Chem 2010, 53(7): 2779
| WO2013067151A1 * | Nov 1, 2012 | May 10, 2013 | Medivation Prostate Therapeutics, Inc. | Treatment methods using diarylthiohydantoin derivatives |
| WO2014041487A2 * | Sep 11, 2013 | Mar 20, 2014 | Dr. Reddy’s Laboratories Limited | Enzalutamide polymorphic forms and its preparation |
| WO2014066799A2 * | Oct 25, 2013 | May 1, 2014 | Memorial Sloan-Kettering Cancer Center | Modulators of resistant androgen receptor |
| WO2014167428A3 * | Mar 5, 2014 | Feb 19, 2015 | Shilpa Medicare Limited | Amorphous 4-(3-(4-cyano-3-(trifluoromethyl)phenyl)-5,5-dimethyl-4-oxo-2-thioxoimidazolidin-1-yl)-2-fluoro-n-methylbenzamide |
| EP2536708A2 * | Feb 16, 2011 | Dec 26, 2012 | Aragon Pharmaceuticals, Inc. | Androgen receptor modulators and uses thereof |















From 2-fluoro-4-bromo – benzoic acid s-1, firstly the carboxylic acid is converted to acid chloride with SOCl2 s-2, and then methylamine to give the enamine compound s-3, and s-3 bromide aminoisobutyric acid Ullmann (Goldberg) in CuCl catalyzed reaction to give the compound s-4, followed by reaction of the carboxylic acid and methyl iodide to give the corresponding methyl ester compound s-5.
Aniline compound s-6 in the sulfur phosgene primary amine is converted to the isothiocyanate s-7.
Finally, the nitrogen atom of the compound s-5 attack isothiocyanate s-7, followed by transesterification ring closure to give the final Xtandi (Enzalutamide, uh miscellaneous Lu amine). Scheme: WO2011106570A1

//////////
DACLATASVIR, 达拉他韦 , Даклатасвир , داكلاتاسفير ,

Daclatasvir
BMS-790052,
EBP 883; BMS 790052
THERAPEUTIC CLAIM Treatment of hepatitis C

CHEMICAL NAMES
1. Carbamic acid, N,N’-[[1,1′-biphenyl]-4,4′-diylbis[1H-imidazole-5,2-diyl-(2S)-2,1-
pyrrolidinediyl[(1S)-1-(1-methylethyl)-2-oxo-2,1-ethanediyl]]]bis-, C,C’-dimethyl ester
2. dimethyl N,N’-(biphenyl-4,4′-diylbis{1H-imidazole-5,2-diyl-[(2S)-pyrrolidine-2,1-
diyl][(1S)-1-(1-methylethyl)-2-oxoethane-2,1-diyl]})dicarbamate
MF C40H50N8O6
MW 738.9
SPONSOR Bristol-Myers Squibb
CODE BMS-790052
CAS 1009119-64-5
SMILES:CC(C)C(C(=O)N1CCCC1C2=NC=C(N2)C3=CC=C(C=C3)C4=CC=C(C=C4)C5=CN=C(N5)C6CCCN6C(=O)C(C(C)C)NC(=O)OC)NC(=O)OC
UNII-LI2427F9CI
Activity: Treatment of Hepatitis C; HCV Drug; Treatment of HCV; Inhibitor of NS5A
Status: Launched 2014 (EU, Japan)
Originator: Bristol-Myers Squibb
Status: Launched 2014 (EU, Japan)
Originator: Bristol-Myers Squibb
NMR
FDA APPROVAL……..July 24th, 2015
Daklinza (daclatasvir) is an NS5A inhibitor indicated for use in combination with sofosbuvir for the treatment of chronic hepatitis C virus (HCV) genotype 3 infection.

Daclatasvir dihydrochloride
1. Carbamic acid, N,N’-[[1,1′-biphenyl]-4,4′-diylbis[1H-imidazole-5,2-diyl-(2S)-2,1-
pyrrolidinediyl[(1S)-1-(1-methylethyl)-2-oxo-2,1-ethanediyl]]]bis-, C,C’-dimethyl ester,
hydrochloride (1:2)
2. dimethyl N,N’-(biphenyl-4,4′-diylbis{1H-imidazole-5,2-diyl-[(2S)-pyrrolidine-2,1-
diyl][(1S)-1-(1-methylethyl)-2-oxoethane-2,1-diyl]})dicarbamate dihydrochloride
MF C40H50N8O6 . 2 HCl, MW 811.8
SPONSOR Bristol-Myers Squibb
CODE BMS-790052-05
CAS 1009119-65-6
Daclatasvir (USAN[1]) (formerly BMS-790052, trade name Daklinza) is a drug for the treatment of hepatitis C (HCV). It is was developed by Bristol-Myers Squibb and was approved in Europe on 22 August 2014.
Daclatasvir inhibits the HCV nonstructural protein NS5A.[2][3] Recent research suggests that it targets two steps of the viral replication process, enabling rapid decline of HCV RNA.[4]
Daclatasvir has been tested in combination regimens with pegylated interferon and ribavirin,[5] as well as with other direct-acting antiviral agents including asunaprevir[6][7][8][9] and sofosbuvir.[10][11]
It is on the World Health Organization’s List of Essential Medicines, a list of the most important medications needed in a basic health system.[12]
Hepatitis C virus (HCV) is a major global health problem, with an estimated 150-200 million people infected worldwide, including at least 5 million in Europe (Pawlotsky, Trends Microbiol, 2004, 12: 96-102). According to the World Health Organization, 3 to 4 million new infections occur each year. The infection is often asymptomatic; however, the majority of HCV-infected individuals develop chronic infection (Hoof agle, Hepatology, 2002, 36: S21-S29; Lauer et al, N. Engl. J. Med., 2001, 345: 41-52; Seeff, Semin. Gastrointest., 1995, 6: 20-27). Chronic infection frequently results in serious liver disease, including fibrosis and steatosis (Chisari, Nature, 2005, 435: 930-932).
About 20% of patients with chronic HCV infection develop liver cirrhosis, which progresses to hepatocellular carcinoma in 5% of the cases (Hoofnagle, Hepatology, 2002, 36: S21-S29; Blonski et al, Clin. Liver Dis., 2008, 12: 661-674; Jacobson et al, Clin. Gastroenterol. Hepatol, 2010, 8: 924-933; Castello et al., Clin. Immunol, 2010, 134: 237-250; McGivern et al., Oncogene, 2011, 30: 1969-1983).
Chronic HCV infection is the leading indication for liver transplantations (Seeff et al., Hepatology, 2002, 36: 1-2). Unfortunately, liver transplantation is not a cure for hepatitis C; viral recurrence being an invariable problem and the leading cause of graft loss (Brown, Nature, 2005, 436: 973-978; Watt et al, Am. J. Transplant, 2009, 9: 1707-1713). No vaccine protecting against HCV is yet available. Current therapies include administration of ribavirin and/or interferon-alpha (IFN-Cc), two non-specific anti-viral agents.
Using a combination treatment of pegylated IFN-CC and ribavirin, persistent clearance is achieved in about 50% of patients with genotype 1 chronic hepatitis C. However, a large number of patients have contraindications to one of the components of the combination; cannot tolerate the treatment; do not respond to interferon therapy at all; or experience a relapse when administration is stopped. In addition to limited efficacy and substantial side effects such as neutropenia, haemo lytic anemia and severe depression, current antiviral therapies are also characterized by high cost.
To improve efficacy of standard of care (SOC), a large number of direct acting antivirals (DAAs) targeting viral polyprotein processing and replication have been developed (Hofmann et al, Nat. Rev; Gastroenterol. Hepatol., 2011, 8: 257-264). These include small molecule compounds targeting HCV nonstructural proteins including the HCV protease, polymerase and NS5A protein.
Although a marked improvement of antiviral response was observed when protease inhibitors were combined with SOC (Hofmann et al, Nat. Rev; Gastroenterol. Hepatol, 2011, 8: 257-264; Bacon et al, New Engl. J. Med., 2011, 364: 1207-1217; McHutchison et al, New Engl. J. Med., 2010, 362: 1292-1303; Poordad et al, New Engl. J. Med., 201 1, 364: 1195-1206; Hezode et al, New Engl. J. Med., 2009, 360: 1839-1850; Kwo et al, Lancet, 2010, 376: 705-716), toxicity of the individual compounds and rapid development of viral resistance in a substantial fraction of patients remain major challenges (Pawlotsky, Hepatology, 2011, 53: 1742-1751; Pereira et al, Nat. Rev. Gastroenterol. Hepatol., 2009, 6: 403-411; Sarrazin et al, Gastroenterol., 2010, 138: 447-462).
New therapeutic approaches against HCV are therefore still needed. HCV entry into target cells is a promising target for antiviral preventive and therapeutic strategies since it is essential for initiation, spread, and maintenance of infection (Timpe et al, Gut, 2008, 57: 1728-1737; Zeisel et al, Hepatology, 2008, 48: 299-307). Indeed, HCV initiates infection by attaching to molecules or receptors on the surface of hepatocytes.
Current evidence suggests that HCV entry is a multistep process involving several host factors including heparan sulfate (Barth et al, J. Biol. Chem., 2003, 278: 41003-41012), the tetraspanin CD81 (Pileri et al, Science, 1998, 282: 938-941), the scavenger receptor class B type I (SR-BI) (Zeisel et al, Hepatology, 2007, 46: 1722-1731; Bartosch et al, J. Exp. Med., 2003, 197: 633-642; Grove et al, J. Virol, 2007, 81 : 3162-3169; Kapadia et al, J. Virol, 2007, 81 : 374- 383; Scarselli et al, EMBO J., 2002, 21 : 5017-5025), Occludin (Ploss et al, Nature, 2009, 457: 882-886) and Claudin-1 (CLDN1), an integral membrane protein and a component of tight-junction strands (Evans et al, Nature, 2007, 446: 801-805).
Furthermore, Niemann-Pick CI -like cholesterol absorption receptor has been identified as a new hepatitis C virus entry factor (Sainz et al, Nature Medicine, 2012, 18: 281-285).
Daclatasvir (BMS-790052; EBP 883) is a first-in-class, highly-selective oral HCV NS5A inhibitor. NS5A is an essential component for hepatitis C virus (HCV) replication complex.Daclatasvir (BMS-790052; EBP 883)has broad genotype coverage and exhibits picomolar in vitro potency against genotypes 1a (EC50 50pm) and 1b (EC50 9pm).Daclatasvir (BMS-790052; EBP 883) produces a robust decline in HCV RNA (-3.6 logs after 48 hours from a single 100 mg) dosefollowing a single dose in patients chronically infected with HCV genotype 1.
It may be many years before the symptoms of hepatitis C infection appear. However, once they do, the consequences are significant: patients may have developed fibrosis, cirrhosis or even liver cancer, with the end result being liver failure. Even if diagnosed early, there’s no guarantee of a cure.
Only around half of patients respond to the standard therapy of an interferon plus the antiviral drug ribavirin, and while two add-on antiviral therapies were approved in 2011, the treatment period is long with no guarantee of a cure, and for non-responders treatment options remain limited.
A new drug with a different mechanism is being developed by Bristol-Myers Squibb, in conjunction with Pharmasset. Daclatasvir targets non-structural protein 5A, which is an important component of the viral replication process, although its precise role in this remains unclear. The drug is active in single oral doses, and may have potential as part of a treatment regimen that avoids the use of interferon, and in patients who do not respond to standard therapy.
In an open label Phase IIa study, 10 patients with chronic hepatitis C genotype 1b infection who did not respond to standard therapy were given daclatasvir in once daily 60mg doses, plus another experimental drug, BMS-790052, which is an NSP 3 protease inhibitor, in initial twice-daily 600mg doses, later reduced to 200mg twice a day.2 Nine patients completed 24 weeks of treatment, with the 10th discontinuing after 10 weeks. In those who completed the course, HCV RNA was undetectable at week 8, and remained so until the end of the trial, with all achieving a sustained virologic response. It was also undetectable post-treatment in the patient who discontinued.
Daclatasvir has also been investigated as monotherapy in a double blind, placebo-controlled, sequential panel, multiple ascending dose study.3 Thirty patients with chronic geno-type 1 hepatitis C infection were randomised to receive a 14 day course of the drug, in once daily doses of 1, 10, 30, 60 or 100mg, 30mg twice a day, or placebo. There was no evidence of antiviral activity in the placebo group, but the mean maximum decline of 2.8 to 4.1 log IU/ml. Most experienced viral rebound on or before day 7 of treatment, which was associated with viral variants that had previously been implicated in resistance development. It was well tolerated in all dose groups.
M. Gao et al. Nature 2010, 465, 96

22/11/2013
EUROPEAN MEDICINES AGENCY ADVISES ON COMPASSIONATE USE OF DACLATASVIR
Opinion concerns use in combination with sofosbuvir in patients with chronic hepatitis C in urgent need of therapy to prevent progression of liver disease
The European Medicines Agency’s Committee for Medicinal Products for Human Use(CHMP) has given an opinion on the use of daclatasvir in combination with sofosbuvir in the treatment of chronic (long-term) hepatitis C virus (HCV) infection, in a compassionate-use programme.
Compassionate-use programmes are set up at the level of individual Member States. They are intended to give patients with a life-threatening, long-lasting or seriously disabling disease with no available treatment options access to treatments that are still under development and that have not yet received amarketing authorisation. In this specific case, Sweden has requested an opinion from the CHMP on the conditions under which early access through compassionate use could be given to daclatasvir, for the use in combination with sofosbuvir, with or without ribavirin, for a specific patient population.
The recommended compassionate use is intended for adult patients at a high risk of their liver being no longer able to function normally (decompensation) or death within 12 months if left untreated, and who have a genotype 1 infection. Further, it is recognised that the potential benefit of such combination therapy may extend to patients infected with other HCV genotypes.
Daclatasvir and sofosbuvir are both first-in-class anti-viral medicines against HCV. These medicines have been studied in combination, with or without ribavirin, in aclinical trial which included treatment-naive (previously untreated) HCV genotype-1, -2 and -3 infected patients, as well as patients with genotype 1 infection who have previously failed telaprevir or boceprevir treatment. Results from the trial indicate high efficacy, also in those who have failed treatment with these protease inhibitors. Many such patients have very advanced liver disease and are in urgent need of effective therapy in order to cease the progression of liver injury.
This is the second opinion provided by the CHMP on compassionate use of medicines in development for the treatment of hepatitis C. Overall, it isthe fourth time compassionate use has been assessed by the CHMP.
The aim of the CHMP assessment and opinion on a compassionate-use programme for new medicinal products is to ensure a common approach, whenever possible, regarding the criteria and conditions of use under Member States’ legislation. The opinion provides recommendations to the EU Member States that are considering setting up such a programme, and its implementation is not mandatory. In addition to describing which patients may benefit from the medicine, it explains how to use it and gives information on safety.
The assessment report and conditions of use of daclatasvir in combination with sofosbuvir with or without ribavirin in this setting will be published shortly on the Agency’s website.
Notes
- The first compassionate-use opinion for a hepatitis C treatment was adopted by the CHMP in October 2013.
- Sofosbuvir, which is part of this compassionate-use opinion, received a positive opinion from the CHMP recommending granting of a marketing authorisation at its November 2013 meeting.
- Daclatasvir is developed by Bristol-Myers Squibb and sofosbuvir is developed by Gilead.
|
1-6-2012
|
Anti-Viral Compounds
|
|
|
2-13-2009
|
CRYSTALLINE FORM OF METHYL ((1S)-1-(((2S)
-2-(5-(4′-(2-((2S)-1((2S)-2-((METHOXYCARBONYL)AMINO)-3-METHYLBUTANOYL)-2-PYRROLIDINYL) -1H-IMIDAZOL-5-YL)-4-BIPHENYLYL)-1H-IMIDAZOL-2-YL)-1-PYRROLIDINYL)CARBONYL) -2-METHYLPROPYL)CARBAMATE DIHYDROCHLORIDE SALT |
Synthesis
Daclatasvir dihydrochloride (Daklinza)
Daclatasvir dihydrochloride is a hepatitis C virus nonstructural 5A (NS5A) replication complex inhibitor which was first approved in Japan for the treatment of genotype 1 HCV patients who fail to respond to interferon plus ribavirin. The drug has also been approved for patients with untreated, chronic HCV who are eligible for interferon. Additionally, in Europe, daclatasvir was approved for use in combination with other products across genotype 1–4 HCV. Daclatasvir was discovered and developed by Bristol–Myers Squibb and a fascinating account describing the initiation of the program from a phenotypic screen and the medicinal chemistry strategy leading to the discovery of the compound has been recently reported.80 Daclatasvir has been prepared via two different routes81,82 and the process route is outlined in Scheme 11.83 Bromination of commercial 4,40-diacetylbiphenyl (58) gave 4,40-bis(bromoacetyl)biphenyl 59 in 82% yield. Alkylation of NBoc- L-proline (60) with 59 gave diester 61 which was treated with ammonium acetate to effect cyclization of the bis-ketoester to provide bis-imidazole 62 in 63% yield for the two steps. Acidic removal of the Boc protecting groups followed by recrystallization provided bis-pyrrolidine 63 in high yield. Acylation of 63 with N-(methoxycarbonyl)- L-valine (64) using N-(3-dimethylaminopropyl)-N0-ethylcarbodiimide(EDC) and 1-hydroxybenxotriazole hydrate (HOBT) provided declatasvir. The dihydrochloride salt was prepared and treated with Cuno Zet Carbon followed by crystallization from acetone
to give daclatasvir dihydrochloride (IX) in 74% yield.
80 Belema, M.; Meanwell, N. A. J. Med. Chem. 2014, 57, 5057.
81. Bachand, C.; Belema, M.; Deon, D. H.; Good, A. C.; Goodrich, J.; James, C. A.;
Lavoie, R.; Lopez, O. D.; Martel, A.; Meanwell, N. A.; Nguyen, V. N.; Romine, J.
L.; Ruediger, E. H.; Snyder, L. B.; St. Laurent, D. R.; Yang, F.; Langley, D. R.;
Wang, G.; Hamann, L. G. WO Patent 2008021927A2, 2008.
82. Belema, M.; Nguyen, V. N.; Bachand, C.; Deon, D. H.; Goodrich, J. T.; James, C.
A.; Lavoie, R.; Lopez, O. D.; Martel, A.; Romine, J. L.; Ruediger, E. H.; Snyder, L.
B.; St Laurent, D. R.; Yang, F.; Zhu, J.; Wong, H. S.; Langley, D. R.; Adams, S. P.;
Cantor, G. H.; Chimalakonda, A.; Fura, A.; Johnson, B. M.; Knipe, J. O.; Parker, D.
D.; Santone, K. S.; Fridell, R. A.; Lemm, J. A.; O’Boyle, D. R., 2nd; Colonno, R. J.;
Gao, M.; Meanwell, N. A.; Hamann, L. G. J. Med. Chem. 2014, 57, 2013.
83. Pack, S. K.; Geng, P.; Smith, M. J.; Hamm, J. WO Patent 2009020825A1, 2009.
PATENT
EXAMPLES
A 1 L, 3-neck round bottom flask, fitted with a nitrogen line, overhead stirrer and thermocouple, was charged with 20 g (83.9 mmol, 1 equiv) 1,1′-(biphenyl-4,4′-diyl)diethanone, 200 mL CH2Cl2 and 8.7 mL (27.1 g, 169.3 mmol, 2.02 quiv) bromine. The mixture was allowed to stir under nitrogen for about 20 hours under ambient conditions. The resulting slurry was charged with 200 mL CH2Cl2 and concentrated down to about 150 mL via vacuum distillation. The slurry was then solvent exchanged into THF to a target volume of 200 mL via vacuum distillation. The slurry was cooled to 20-25° C. over 1 hour and allowed to stir at 20-25° C. for an additional hour. The off-white crystalline solids were filtered and washed with 150 mL CH2Cl2. The product was dried under vacuum at 60° C. to yield 27.4 g (69.2 mmol, 82%) of the desired product : 1H NMR (400 MHz, CDCl3) δ 7.95-7.85 (m, 4H), 7.60-7.50 (m, 4H), 4.26 (s, 4H); 13C NMR (100 MHz, CDCl3) 6 191.0, 145.1, 133.8, 129.9, 127.9, 30.8; IR (KBr, cm−1) 3007, 2950, 1691, 1599, 1199; Anal calcd for C16H12Br2O2: C, 48.52; H, 3.05; Br, 40.34. Found: C, 48.53; H, 3.03; Br, 40.53 HRMS calcd for C16H13Br2O2 (M+H; DCI+): 394.9282. Found: 394.9292. mp 224-226° C.
A 500 mL jacketed flask, fitted with a nitrogen line, thermocouple and overhead stirrer, was charged with 20 g (50.5 mmol, 1 equiv) of Compound 2, 22.8 g (105.9 moles, 2.10 equiv) 1-(tert-butoxycarbonyl)-L-proline and 200 mL acetonitrile. The slurry was cooled to 20° C. followed by the addition of 18.2 mL (13.5 g, 104.4 mmol, 2.07 equiv) DIPEA. The slurry was warmed to 25° C. and allowed to stir for 3 hours. The resulting clear, organic solution was washed with 3×100 mL 13 wt % aqueous NaCl. The rich acetonitrile solution was solvent exchanged into toluene (target volume=215 mL) by vacuum distillation until there was less than 0.5 vol % acetonitrile.
The toluene solution of Compound 3 was charged with 78 g (1.011 moles, 20 equiv) ammonium acetate and heated to 95-100° C. The mixture was allowed to stir at 95-100° C. for 15 hours. After reaction completion, the mixture was cooled to 70-80° C. and charged with 7 mL acetic acid, 40 mL n-butanol, and 80 mL of 5 vol % aqueous acetic acid. The resulting biphasic solution was split while maintaining a temperature >50° C. The rich organic phase was charged with 80 mL of 5 vol % aqueous acetic acid, 30 mL acetic acid and 20 mL n-butanol while maintaining a temperature >50° C. The resulting biphasic solution was split while maintaining a temperature >50° C. and the rich organic phase was washed with an additional 80 mL of 5 vol % aqueous acetic acid. The rich organic phase was then solvent exchanged into toluene to a target volume of 215 mL by vacuum distillation. While maintaining a temperature >60° C., 64 mL methanol was charged. The resulting slurry was heated to 70-75° C. and aged for 1 hour. The slurry was cooled to 20-25° C. over 1 hour and aged at that temperature for an additional hour. The slurry was filtered and the cake was washed with 200 mL 10:3 toluene:methanol. The product was dried under vacuum at 70° C., resulting in 19.8 g (31.7 mmol, 63%) of the desired product: 1H NMR (400 MHz, DMSO-d6) δ 13.00-11.00 (s, 2H), 7.90-7.75 (m, 4H), 7.75-7.60 (m, 4H), 7.60-7.30 (s, 2H), 4.92-4.72 (m, 2H), 3.65-3.49 (m, 2H), 3.49-3.28 (m, 2H), 2.39-2.1 (m, 2H), 2.10-1.87 (m, 6H), 1.60-1.33 (s, 8H), 1.33-1.07 (s, 10H); 13C NMR (100 MHz, DMSO-d6) δ 154.1, 153.8, 137.5, 126.6, 125.0, 78.9, 78.5, 55.6, 55.0, 47.0, 46.7, 33.7, 32.2, 28.5, 28.2, 24.2, 23.5; IR (KBr, cm−1) 2975, 2876, 1663, 1407, 1156, 1125; HRMS calcd for C36H45N6O4 (M+H; ESI+): 625.3502. Found: 625.3502. mp 190-195° C. (decomposed).
To a 250 mL reactor equipped with a nitrogen line and overhead stirrer, 25.0 g of Compound 4 (40.01 mmol, 1 equiv) was charged followed by 250 mL methanol and 32.85 mL (400.1 mmol, 10 equiv) 6M aqueous HCl. The temperature was increased to 50° C. and agitated at 50° C. for 5 hours. The resulting slurry was cooled to 20-25° C. and held with agitation for about 18 hours. Filtration of the slurry afforded a solid which was washed successively with 100 mL 90% methanol/water (V/V) and 2×100 mL of methanol. The wet cake was dried in a vacuum oven at 50° C. overnight to give 18.12 g (31.8 mmol, 79.4%) of the desired product.
Recrystallization of Compound 5
To a 250 mL reactor equipped with a nitrogen line and an overhead stirrer, 17.8 g of Compound 5 from above was charged followed by 72 mL methanol. The resulting slurry was agitated at 50° C. for 4 hours, cooled to 20-25° C. and held with agitation at 20-25° C. for 1 hour. Filtration of the slurry afforded a crystalline solid which was washed with 60 mL methanol. The resulting wet cake was dried in a vacuum oven at 50° C. for 4 days to yield 14.7 g (25.7 mmol, 82.6%) of the purified product: 1H NMR (400 MHz, DMSO-d6) δ 10.5-10.25 (br, 2H), 10.1-9.75 (br, 2H), 8.19 (s, 2H), 7.05 (d, J=8.4, 4H), 7.92 (d, J=8.5, 4H), 5.06 (m, 2H), 3.5-3.35 (m, 4H), 2.6-2.3 (m, 4H), 2.25-2.15 (m, 2H), 2.18-1.96 (m, 2H); 13C NMR (100 MHz, DMSO-d6) δ 156.6, 142.5, 139.3, 128.1, 127.5, 126.1, 116.9, 53.2, 45.8, 29.8, 24.3; IR (KBr, cm−1) 3429, 2627, 1636, 1567, 1493, 1428, 1028. Anal calcd for C26H32N6Cl4: C, 54.75; H, 5.65; Cl, 24.86; Adjusted for 1.9% water: C, 53.71; H, 5.76; N, 14.46; Cl, 24.39. Found: C, 53.74; H, 5.72; N, 14.50; Cl, 24.49; KF=1.9. mp 240° C. (decomposed).
A 1 L jacketed flask equipped with a nitrogen line and an overhead stirrer was sequentially charged with 100 mL acetonitrile, 13.69 g (89.4 mmol, 2.5 equiv) hydroxybenzotriazole hydrate, 15.07 g (86 mmol, 2.4 equiv) N-(methoxycarbonyl)-L-valine, 16.46 g (85.9 mmol, 2.4 equiv) 1-(3-dimethyaminopropyl)-3-ethylcarbodiimide hydrochloride and an additional 100 mL acetonitrile. The resulting solution was agitated at 20° C. for 1 hour and charged with 20.4 g (35.8 mmol, 1 equiv) of purified Compound 5. The slurry was cooled to about 0° C. and 18.47 g (142.9 mmol, 4 equiv) diisopropylethylamine was added over 30 minutes while maintaining a temperature below 10° C. The solution was slowly heated to 15° C. over 3 hours and held at 15° C. for 12 hours. The resulting solution was charged with 120 mL 13 wt % aqueous NaCl and heated to 50° C. for 1 hour. After cooling to 20° C., 100 mL of isopropyl acetate was added. The biphasic solution was filtered through a 0.45 μm filter and the mixture split. The rich organic phase was washed with 2×240 mL of a 0.5 N NaOH solution containing 13 wt % NaCl followed by 120 mL 13 wt % aqueous NaCl. The mixture was then solvent exchanged into isopropyl acetate by vacuum distillation with a target volume of 400 mL. The resulting hazy solution was cooled to 20° C. and filtered through a 0.45 μm filter. The clear solution was then solvent exchanged into ethanol by vacuum distillation with a target volume of 140 mL. While maintaining a temperature of 50° C., 66.4 mL (82.3 mmol, 2.3 equiv) of 1.24M HCl in ethanol was added. The mixture was then charged with 33 mg (0.04 mmol, 0.001 equiv) of seed crystals of Compound (I) (see preparation below) and the resulting slurry was stirred at 50° C. for 3 hours. The mixture was cooled to 20° C. over 1 hour and aged at that temperature for an additional 22 hours. The slurry was filtered and the wet cake was washed with 100 mL of 2:1 acetone:ethanol. The solids were dried in a vacuum oven at 70° C. to give 22.15 g (27.3 mmol, 76.3%) of the desired product.
A solution of Compound (I) was prepared by dissolving 3.17 g of Compound (I) from above in 22 mL methanol. The solution was passed through a 47 mm Cuno Zeta Carbon® 53SP filter at ˜5 psig at a flow rate of ˜58 mL/min. The carbon filter was rinsed with 32 mL of methanol. The solution was concentrated down to 16 mL by vacuum distillation. While maintaining a temperature of 40-50° C., 15.9 mL acetone and 5 mg of seed crystals of Compound (I) (see procedure below) were added. The resulting slurry was then charged with 32 mL acetone over 30 minutes. The slurry was held at 50° C. for 2 hours, cooled to 20° C. over about 1 hour and held at 20° C. for about 20 hours. The solids were filtered, washed with 16 mL 2:1 acetone:methanol and dried in a vacuum oven at 60° C. to give 2.14 g (67.5%) of purified Compound (I):
1H NMR (400 MHz, DMSO-d6, 80° C.): 8.02 (d, J=8.34 Hz, 4 H), 7.97 (s, 2 H), 7.86 (d, J=8.34 Hz, 4 H), 6.75 (s, 2 H), 5.27 (t, J=6.44 Hz, 2 H), 4.17 (t, J=6.95 Hz, 2 H), 3.97-4.11 (m, 2 H), 3.74-3.90 (m, 2 H), 3.57 (s, 6 H), 2.32-2.46 (m, 2 H), 2.09-2.31 (m, 6 H), 1.91-2.07 (m, 2 H), 0.88 (d, J=6.57 Hz, 6 H), 0.79 (d, J=6.32 Hz, 6 H);
13C NMR (75 MHz, DMSO-d6): δ 170.9, 156.9, 149.3, 139.1, 131.7, 127.1, 126.5, 125.9, 115.0, 57.9, 52.8, 51.5, 47.2, 31.1, 28.9, 24.9, 19.6, 17.7;
IR (neat, cm−1): 3385, 2971, 2873, 2669, 1731, 1650.
Anal. Calcd for C40H52N8O6Cl2: C, 59.18; H, 6.45; N, 13.80; Cl, 8.73. Found C, 59.98; H, 6.80; N, 13.68; Cl, 8.77. mp 267° C. (decomposed).
Preparation of Seed Crystals of Compound (I)
A 250 mL round-bottom flask was charged with 6.0 g (10.5 mmol, 1 equiv) Compound 5, 3.87 g (22.1 mmol, 2.1 equiv) N-(methoxycarbonyl)-L-valine, 4.45 g (23.2 mmol, 2.2 equiv) 1-(3-dimethyaminopropyl)-3-ethylcarbodiimide hydrochloride, 0.289 g (2.14 mmol, 0.2 equiv) 1-hydroxybenzotriazole, and 30 mL acetonitrile. The resulting slurry was then charged with 7.33 mL (42.03 mmol, 4 equiv) diisopropylethylamine and allowed to stir at 24-30° C. for about 18 hours. The mixture was charged with 6 mL of water and heated to 50° C. for about 5 hours. The mixture was cooled and charged with 32 mL ethyl acetate and 30 mL water. The layers were separated and the rich organic layer was washed with 30 mL of 10 wt % aqueous NaHCO3, 30 mL water, and 20 mL of 10 wt % aqueous NaCl. The rich organic layer was then dried over MgSO4, filtered, and concentrated down to a residue. The crude material was then purified via flash chromatography (silica gel, 0-10% methanol in dichloromethane) to provide the free base of Compound (I).
The free-base of Compound (I) (0.03 g) was dissolved in 1 mL isopropanol at 20° C. Anhydrous HCl (70 μL, dissolved in ethanol, approximately 1.25M concentration) was added and the reaction mixture was stirred. To the solution was added methyl tert-butyl ether (1 mL) and the resulting slurry was stirred vigorously at 40° C. to 50° C. for 12 hours. The crystal slurry was cooled to 20° C. and filtered. The wet cake was air-dried at 20° C. A white crystalline solid (Form N-2 of Compound (I)) was obtained.
Clip
Daclatasvir synthesis: WO2009020828A1

Procedure:
Step a: A 1 L, 3 -neck round bottom flask, fitted with a nitrogen line, overhead stirrer and thermocouple, was charged with 20 g (83.9 mmol, 1 equiv) 1,1′-(biphenyl-4,4′-diyl)diethanone, 200 mL Dichloromethane and 8.7 mL (27.1g, 169.3 mmol, 2.02 equiv) bromine. The mixture was allowed to stir under nitrogen for about 20 hours under ambient conditions. The resulting slurry was charged with 200 mL Dichloromethane and concentrated down to about 150 mL via vacuum distillation. The slurry was then solvent exchanged into THF to a target volume of 200 mL via vacuum distillation. The slurry was cooled to 20-25 0C over 1 hour and allowed to stir at 20-25 0C for an additional hour. The off-white crystalline solids were filtered and washed with 150 mL Dichloromethane. The product was dried under vacuum at 60 0C to yield 27.4 g (69.2 mmol, 82%) of the desired product: 1H NMR (400 MHz, CDCl3) d 7.95-7.85 (m, 4H), 7.60-7.50 (m, 4H), 4.26 (s, 4H); 13C NMR 100 MHz, CDCl3) d 191.0, 145.1, 133.8, 129.9, 127.9, 30.8; IR (KBr, cm-1) 3007, 2950, 1691, 1599, 1199; Anal calcd for C16H12Br2O2: C, 48.52; H, 3.05; Br, 40.34. Found: C, 48.53; H, 3.03; Br, 40.53. HRMS calcd for C16H12Br2O2 (M + H; DCI+): 394.9282. Found: 394.9292. mp 224-226 0C.
Step b: A 500 mL jacketed flask, fitted with a nitrogen line, thermocouple and overhead stirrer, was charged with 20 g (50.5 mmol, 1 equiv) of Compound 2, 22.8 g (105.9 moles, 2.10 equiv) 1-(tert-butoxycarbonyl)-L-proline and 200 mL acetonitrile. The slurry was cooled to 20 0C followed by the addition of 18.2 mL (13.5 g, 104.4 mmol, 2.07 equiv) DIPEA. The slurry was warmed to 25 0C and allowed to stir for 3 hours. The resulting clear, organic solution was washed with 3 x 100 mL 13 wt% aqueous NaCl. The rich acetonitrile solution was solvent exchanged into toluene (target volume = 215 mL) by vacuum distillation until there was less than 0.5 vol% acetonitrile.
Step c: The toluene solution of Compound 3 was charged with 78 g (1.011 moles, 20 equiv) ammonium acetate and heated to 95-100 0C. The mixture was allowed to stir at 95-100 0C for 15 hours. After reaction completion, the mixture was cooled to 70- 80 0C and charged with 7 mL acetic acid, 40 mL n-butanol, and 80 mL of 5 vol% aqueous acetic acid. The resulting biphasic solution was split while maintaining a temperature > 50 0C. The rich organic phase was charged with 80 mL of 5 vol% aqueous acetic acid, 30 mL acetic acid and 20 mL n-butanol while maintaining a temperature > 50 0C. The resulting biphasic solution was split while maintaining a temperature > 50 0C and the rich organic phase was washed with an additional 80 mL of 5 vol% aqueous acetic acid. The rich organic phase was then solvent exchanged into toluene to a target volume of 215 mL by vacuum distillation. While maintaining a temperature > 60 0C, 64 mL methanol was charged. The resulting slurry was heated to 70-75 0C and aged for 1 hour. The slurry was cooled to 20-25 0C over 1 hour and aged at that temperature for an additional hour. The slurry was filtered and the cake was washed with 200 mL 10:3 toluene:methanol. The product was dried under vacuum at 70 0C, resulting in 19.8 g (31.7 mmol, 63%) of the desired product: 1H NMR (400 MHz, DMSO-^) d 13.00-11.00 (s, 2H), 7.90-7.75 (m, 4H), 7.75-7.60 (m, 4H), 7.60-7.30 (s, 2H), 4.92-4.72 (m, 2H), 3.65-3.49 (m, 2H), 3.49-3.28 (m, 2H), 2.39-2.1 (m, 2H), 2.10-1.87 (m, 6H), 1.60-1.33 (s, 8H), 1.33-1.07 (s, 10H); 13C NMR (100 MHz, DMSO-?fe) d 154.1, 153.8, 137.5, 126.6, 125.0, 78.9, 78.5, 55.6, 55.0, 47.0, 46.7, 33.7, 32.2, 28.5, 28.2, 24.2, 23.5; IR (KBr, cm-1) 2975, 2876, 1663, 1407, 1156, 1125; HRMS calcd for C36H45N6O4 (M + H; ESI+): 625.3502. Found: 625.3502. mp 190-195 0C (decomposed).
Step d: To a 250 mL reactor equipped with a nitrogen line and overhead stirrer, 25.0 g of Compound 4 (40.01 mmol, 1 equiv) was charged followed by 250 mL methanol and 32.85 mL (400.1 mmol, 10 equiv) 6M aqueous HCl. The temperature was increased to 50 0C and agitated at 50 0C for 5 hours. The resulting slurry was cooled to 20-25 0C and held with agitation for about 18 hours. Filtration of the slurry afforded a solid which was washed successively with 100 mL 90% methanoI/water (WV) and 2 x 100 mL of methanol. The wet cake was dried in a vacuum oven at 50 0C overnight to give 18.12 g (31.8 mmol, 79.4%) of the desired product.
CUT PASTE…….WO2009020825
Preparation of Compound (I)
A 1 L jacketed flask equipped with a nitrogen line and an overhead stirrer was sequentially charged with 100 mL acetonitrile, 13.69 g (89.4 mmol, 2.5 equiv) hydroxybenzotriazole hydrate, 15.07 g (86 mmol, 2.4 equiv) N-(methoxycarbonyl)- L-valine, 16.46 g (85.9 mmol, 2.4 equiv) l-(3-dimethyaminopropyl)-3- ethylcarbodiimide hydrochloride and an additional 100 mL acetonitrile. The resulting solution was agitated at 20 0C for 1 hour and charged with 20.4 g (35.8 mmol, 1 equiv) of purified Compound 7. The slurry was cooled to about 0 0C and 18.47 g (142.9 mmol, 4 equiv) diisopropylethylamine was added over 30 minutes while maintaining a temperature below 10 0C. The solution was slowly heated to 15 0C over 3 hours and held at 15 0C for 12 hours. The resulting solution was charged with 120 mL 13 wt% aqueous NaCl and heated to 50 0C for 1 hour. After cooling to 20 0C, 100 mL of isopropyl acetate was added. The biphasic solution was filtered through a 0.45 μm filter and the mixture split. The rich organic phase was washed with 2 x 240 mL of a 0.5 Ν NaOH solution containing 13 wt% NaCl followed by 120 mL 13 wt% aqueous NaCl. The mixture was then solvent exchanged into isopropyl acetate by vacuum distillation with a target volume of 400 mL. The resulting hazy solution was cooled to 20 0C and filtered through a 0.45 μm filter. The clear solution was then solvent exchanged into ethanol by vacuum distillation with a target volume of 140 mL. While maintaining a temperature of 50 0C, 66.4 mL (82.3 mmol, 2.3 equiv) of 1.24M HCl in ethanol was added. The mixture was then charged with 33 mg (0.04 mmol, 0.001 equiv) of seed crystals of Compound (I) (see preparation below) and the resulting slurry was stirred at 50 0C for 3 hours. The mixture was cooled to 20 0C over 1 hour and aged at that temperature for an additional 22 hours. The slurry was filtered and the wet cake was washed with 100 mL of 2: 1 acetone:ethanol. The solids were dried in a vacuum oven at 70 0C to give 22.15 g (27.3 mmol, 76.3%) of the desired product.
Alternative Preparation of Compound (I)
A jacketed reactor equipped with a mechanical agitator, a thermocouple and a nitrogen inlet was sequentially charged with 10 L acetonitrile, 0.671 kg (4.38 moles, 2.50 equiv) 1-hydroxybenzotriazole, 0.737 kg (4.21 moles, 2.40 equiv) N- (methoxycarbonyl)-L-valine and 0.790 kg (4.12 moles, 2.35 equiv) l-(3- dimethyaminopropyl)-3-ethylcarbodiimide hydrochloride. The mixture was agitated at 200C for 1 hour, cooled to 5 0C and charged with 1 kg (1.75 moles, 1.00 equiv) Compound 7. While maintaining a temperature < 10 0C, 0.906 kg (7.01 moles, 4 equiv) diisopropylethylamine was added. The mixture was heated to 15-20 0C over 2 hours and agitated for an additional 15 hours. After the reaction was complete, the mixture was washed once with 6.0 L 13 wt% aqueous NaCl, twice with 6.1 L (6.12 moles, 3.5 equiv) 1.0 M aqueous NaOH containing 13 wt% NaCl and once with 6.0 L 13 wt% aqueous NaCl. Water was then removed from the rich organic solution via azeotropic distillation. The mixture was cooled to 20 0C, agitated for 1 hour and filtered. The rich organic solution was then solvent exchanged into EtOH via vacuum distillation to a target volume of 5 L. While maintaining a temperature of 50 0C, 3.2 L (4.0 moles, 2.3 equiv) 1.25M HCl in EtOH was charged. The mixture was seeded with 1.6 g Compound (I) (see preparation below) and agitated at 50 0C for 3 hours. The resulting slurry was cooled to 20 0C and agitated for at least 3 hours. The product was collected by filtration and washed with 5 L 2: 1 acetone:
EtOH to give 1.29 kg (ca. 90 wt% product) of wet crude product. A reactor equipped with an overhead agitator, nitrogen inlet and thermocouple was charged with 1.11 kg of the above crude product and 7 L methanol. The resulting solution was treated with Cuno Zeta Carbon (TM) 55SP. The carbon was washed with 15 L MeOH and the combined filtrate and wash was concentrated down to 4 L via vacuum distillation. The concentrated solution was charged with 5 L acetone and seeded with 1.6 g Compound (I) (see preparation below) while maintaining a temperature of 50 0C. An additional 10 L acetone was charged and the resulting slurry was stirred at 50 0C for 3 hours. The slurry was cooled to 20 0C and allowed to agitate at 200C for 3 hours. The product was collected by filtration, washed with 5 L 2: 1 acetone: EtOH and dried under vacuum at 50-60 0C to give 0.900 kg (1.11 moles, 74% adjusted) of Compound (I)-
Carbon Treatment and Recrystallization of Compound (I) A solution of Compound (I) was prepared by dissolving 3.17 g of Compound (I) from above in 22 mL methanol. The solution was passed through a 47mm Cuno Zeta Carbon 53SP filter at ~5 psig at a flow rate of~58mL/min. The carbon filter was rinsed with 32 mL of methanol. The solution was concentrated down to 16 mL by vacuum distillation. While maintaining a temperature of 40-50 0C, 15.9 mL acetone and 5 mg of seed crystals of Compound (I) (see procedure below) were added. The resulting slurry was then charged with 32 mL acetone over 30 minutes. The slurry was held at 50 0C for 2 hours, cooled to 20 0C over about 1 hour and held at 20 0C for about 20 hours. The solids were filtered, washed with 16 mL 2: 1 acetone:methanol and dried in a vacuum oven at 60 0C to give 2.14 g (67.5%) of purified Compound (I):
1H NMR (400 MHz, DMSO-έfc, 80 0C): 8.02 (d, J=8.34 Hz, 4 H), 7.97 (s, 2 H), 7.86 (d, J=8.34 Hz, 4 H), 6.75 (s, 2 H), 5.27 (t, J=6.44 Hz, 2 H), 4.17 (t, J=6.95 Hz, 2 H), 3.97 – 4.11 (m, 2 H), 3.74 – 3.90 (m, 2 H), 3.57 (s, 6 H), 2.32 – 2.46 (m, 2 H), 2.09 – 2.31 (m, 6 H), 1.91 – 2.07 (m, 2 H), 0.88 (d, J=6.57 Hz, 6 H), 0.79 (d, J=6.32 Hz, 6 H);
13C NMR (75 MHz, DMSO-έfc): δ 170.9, 156.9, 149.3, 139.1, 131.7, 127.1, 126.5, 125.9, 115.0, 57.9, 52.8, 51.5, 47.2, 31.1, 28.9, 24.9, 19.6, 17.7;
IR (neat, cm“1): 3385, 2971, 2873, 2669, 1731, 1650.
Anal. Calcd for C40H52N8O6Cl2: C, 59.18; H, 6.45; N, 13.80; Cl, 8.73. Found C, 59.98; H, 6.80; N, 13.68; Cl, 8.77. mp 267 0C (decomposed).
Characteristic diffraction peak positions (degrees 2Θ + 0.1) @ RT, based on a high quality pattern collected with a diffractometer (CuKa) with a spinning capillary with 2Θ calibrated with a NIST other suitable standard are as follows: 10.3, 12.4, 12.8, 13.3, 13.6, 15.5, 20.3, 21.2, 22.4, 22.7, 23.7
Daclatasvir faces problems in USA
The US-FDA in 2014 issued a complete response letter for NS5A inhibitor daclatasvir saying it was unable to approve the drug because the marketing application was for its use in tandem with asunaprevir, an NS3/NS4A protease inhibitor discontinued in the US by BMS for commercial reasons. Daclatasvir is already on the market in Europe-where it is sold as Daklinza-and also in Japan where it was approved alongside asunaprevir in July as the country’s first all-oral HCV therapy. However, a delay in the large US market is clearly a major setback for BMS’ ambitions in hepatitis therapy.
To make the matter worse, US FDA has rescinded breakthrough therapy designation status from Bristol-Myers Squibb for Daclatasvir for the treatment of hepatitis C virus infection in Feb 2015.
PAPER
Makonen, B.; et. al. Hepatitis C Virus NS5A Replication Complex Inhibitors: The Discovery of Daclatasvir. J Med Chem 2014, 57(5), 2013–2032.
http://pubs.acs.org/doi/abs/10.1021/jm401836p
PATENT
http://www.google.com/patents/WO2008021927A2?cl=en
Example 24-23
methyl ((lS)-l-(((2S)-2-(5-(4′-(2-((2S)-l-((2S)-2-((methoxycarbonyl)amino)-3- methylbutanoyl)-2-pyrrolidinyl)-lH-imidazol-5-yl)-4-biphenylyl)-lH-imidazol-2-yl)-
1 -pyrrolidinyl) carbonyl) -2-methylpropyl) carbamate
A 50 mL flask equipped with a stir bar was sequentially charged with 2.5 mL acetonitrile, 0.344 g (2.25 mmol, 2.5 equiv) hydroxy benzotriazole hydrate, 0.374 g (2.13 mmol, 2.4 equiv) N-(methoxycarbonyl)-L-valine, 0.400 g (2.09 mmol, 2.4 equiv) 1 -(3 -dimethyaminopropyl)-3-ethylcarbodiimide hydrochloride and an additional 2.5 mL acetonitrile. The resulting solution was agitated at 20 0C for 1 hour and charged with 0.501 g (0.88 mmol, 1 equiv) Example A-le-4. The slurry was cooled to about 0 0C and 0.45 g (3.48 mmol, 4 equiv) diisopropylethylamine was added over 30 minutes while maintaining a temperature below 10 0C. The solution was slowly heated to 15 0C over 3 hours and held at 15 0C for 16 hours. The temperature was increased to 20 0C and stirred for 3.25 hours. The resulting solution was charged with 3.3 g of 13 wt% aqueous NaCl and heated to 50 0C for 1 hour. After cooling to 20 0C, 2.5 mL of isopropyl acetate was added. The rich organic phase was washed with 2 x 6.9 g of a 0.5 N NaOH solution containing 13 wt% NaCl followed by 3.3 g of 13 wt% aqueous NaCl. The mixture was then solvent exchanged into isopropyl acetate by vacuum distillation to a target volume of 10 mL. The resulting hazy solution was cooled to 20 0C and filtered through a 0.45 μm filter. The clear solution was then solvent exchanged into ethanol by vacuum distillation with a target volume of 3 mL. 1.67 mL (2.02 mmol, 2.3 equiv) of 1.21 M HCl in ethanol was added. The mixture was then stirred at 25 0C for 15 hours. The resulting slurry was filtered and the wet cake was washed with 2.5 mL of 2: 1 acetone:ethanol. The solids were dried in a vacuum oven at 50 0C to give 0.550 g (0.68 mmol, 77 %) of the desired product.
RecrystalHzation of Example 24-23
A solution of Example 24-23 prepared above was prepared by dissolving 0.520 g of the above product in 3.65 mL methanol. The solution was then charged with 0.078 g of type 3 Cuno Zeta loose carbon and allowed to stir for 0.25 hours. The mixture was then filtered and washed with 6 ml of methanol. The product rich solution was concentrated down to 2.6 mL by vacuum distillation. 7.8 mL acetone was added and allowed to stir at 25 0C for 15 h. The solids were filtered, washed with 2.5 mL 2: 1 acetone:ethanol and dried in a vacuum oven at 70 0C to give 0.406 g (57.0%) of the desired product as white crystals: 1H NMR (400 MHz, OMSO-d6, 80 0C): 8.02 (d, J=8.34 Hz, 4 H), 7.97 (s, 2 H), 7.86 (d, J=8.34 Hz, 4 H), 6.75 (s, 2 H), 5.27 (t, J=6.44 Hz, 2 H), 4.17 (t, J=6.95 Hz, 2 H), 3.97 – 4.11 (m, 2 H), 3.74 – 3.90 (m, 2 H), 3.57 (s, 6 H), 2.32 – 2.46 (m, 2 H), 2.09 – 2.31 (m, 6 H), 1.91 – 2.07 (m, 2 H), 0.88 (d, J=6.57 Hz, 6 H), 0.79 (d, J=6.32 Hz, 6 H); 13C NMR (75 MHz, DMSO- d6): δ 170.9, 156.9, 149.3, 139.1, 131.7, 127.1, 126.5, 125.9, 115.0, 57.9, 52.8, 51.5, 47.2, 31.1, 28.9, 24.9, 19.6, 17.7; IR (neat, cm“1): 3385, 2971, 2873, 2669, 1731, 1650. Anal. Calcd for C40H52N8O6Cl2: C, 59.18; H, 6.45; N, 13.80; Cl, 8.73. Found C, 59.98; H, 6.80; N, 13.68; Cl, 8.77. mp 267 0C (decomposed). Characteristic diffraction peak positions (degrees 2Θ ± 0.1) @ RT, based on a high quality pattern collected with a diffractometer (CuKa) with a spinning capillary with 2Θ calibrated with a NIST other suitable standard are as follows: 10.3, 12.4, 12.8, 13.3, 13.6, 15.5, 20.3, 21.2, 22.4, 22.7, 23.7
PAPER
Bioorganic & Medicinal Chemistry Letters (2015), 25(16), 3147-3150
http://www.sciencedirect.com/science/article/pii/S0960894X15005995
Scheme 1.
Synthetic route for the preparation of the target compounds 8a–8y. Reagents and conditions: (a) Br2, CH2Cl2, rt, overnight, 86%; (b) N-Boc-l-proline, MeCN, Et3N, rt, 2 h, 98%; (c) NH4OAc, toulene, 130 °C, 15 h, 85%; (d) 6 N HCl, MeOH, 50 °C, 4 h, 87%; (e) HATU, N-(methoxycarbonyl)-l-valine, DIPEA, rt, 14 h, 83%; (f) RCOCl, TEA, CH2Cl2, rt, 3 h, 64–87%.
Dimethyl((2S,2’S)-((2S,2’S)-2,2′-(5,5′-([1,1′-biphenyl]-4,4′-diyl)bis(1H-imidazole-
5,2-diyl))bis(pyrrolidine-2,1-diyl))bis(3-methyl-1-oxobutane-2,1-
diyl))dicarbamate 7……………FREE BASE
To a solution of 5 (90 mg, 0.181 mmol), N-me-thoxycarbonyl-l-valine 6 (92 mg,0.525 mmol) and DIPEA (0.18 mL, 1.03 mmol) in DMF (5 mL) was added HATU(165.5 mg, 0.434 mmol). The resulting reaction was allowed to stir at room temperature for 15 h, the reaction mixture was filtered and the residue was partitioned between EtOAc and H2O, The aqueous phase was extracted with EtOAc, and the combined organic phase was dried (MgSO4), filtered, and concentrated in vacuo. The residue was purified by flash chromatography (silica gel; 5% Methanol /CH2Cl2) to
afford 7 (0.11 g, 83 %)as white solid.
1H NMR (DMSO-d6, 500 MHz) δ: 11.56 (s, 2H), 7.69-7.48 (m, 8H), 7.26-7.03 (m, 4H), 5.24-5.05 (m, 2H), 4.09-4.04 (m, 2H), 3.85-3.75 (m, 4H), 3.58 (s, 6H), 2.24-1.98 (m, 10H), 0.87 (d, J = 3.6 Hz, 12H).
Anal. calcd. (%) for C40H50N8O6: C 65.02, H 6.82, N 15.17; found: C 65.20, H 6.79, N 15.31.
ESI-MS m/z: 739.5 (M+H)+.
NMR PREDICT
1H NMR PREDICT
13C NMR PREDICT
COSY PREDICT
Patents
http://www.who.int/phi/implementation/ip_trade/daclatasvir_report_2014_09-02.pdf
Click on images to view
Click on images to view
http://www.who.int/phi/implementation/ip_trade/daclatasvir_report_2014_09-02.pdf
Click on images to view
Click on images to view
Click on images to view
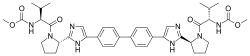 |
|
| Names | |
|---|---|
| IUPAC name
Methyl [(2S)-1-{(2S)-2-[4-(4’-{2-[(2S)-1-{(2S)-2-[(methoxycarbonyl)amino]-3-methylbutanoyl}-2-pyrrolidinyl]-1H-imidazol-4-yl}-4-biphenylyl)-1H-imidazol-2-yl]-1-pyrrolidinyl}-3-methyl-1-oxo-2-butanyl]carbamate
|
|
| Other names
BMS-790052
|
|
| Identifiers | |
| 1009119-64-5 |
|
| ATC code | J05AX14 |
| ChEBI | CHEBI:82977 |
| ChEMBL | ChEMBL2023898 ChEMBL2303621 |
| ChemSpider | 24609522 |
| Jmol-3D images | Image |
| Properties | |
| C40H50N8O6 | |
| Molar mass | 738.89 g·mol−1 |
CLIP 1
Australian Government, National Measurement Institute
REFERENCE MATERIAL ANALYSIS REPORT
HPLC: Instrument: Shimadzu Binary pump LC-20AB, SIL-20 A HT autosampler
Column: X-Bridge C-18, 5.0 m (4.6 mm x 150 mm)
Column oven: 40 °C
Mobile Phase: A = Milli-Q water buffered at pH 10 with NH4
+ -OAc; B = MeCN
Gradient 0 min 35% B; 0-15 min 35% B; 15-18 min 35-75% B; 18-23 min 75% B.
Flow rate: 1.0 mL/min
Detector: Shimadzu SPD-M20A PDA operating at 310 nm
Relative peak area response of main component:
Initial analysis: Mean = 99.2%, s = 0.01%
Thermogravimetric analysis: Non volatile residue < 0.2% mass fraction . The volatile
content (e.g. organic solvents and/or water) could not be determined by
thermogravimetric analysis.
Karl Fischer analysis: Moisture content 0.6% mass fraction
QNMR: Instrument: Bruker Avance-III-500
Field strength: 500 MHz Solvent: DMSO-d6 (2.50 ppm)
Internal standard: Potassium hydrogen maleate (98.8% mass fraction)
Initial analysis: Mean (0.86 ppm) = 98.2%, s = 0.2%
LC-MS: Instrument: Thermo Scientific Dionex UltiMate 3000 Degasser,
Column: ZORBAX RRHD SB-C8, 2.1 x 50 mm, 1.8 μm (Agilent, 857700-906)
Column temp: 30.0 °C
Solvent system: Mobile phase A: 10 mM ammonium formate, 0.01% (v/v) formic acid in Milli-Q® water.
Mobile phase B: 0.01% (v/v) formic acid in acetonitrile.
Gradient from 90% A to 100% B
Flow rate: 0.25 mL/min
Sample prep: 2 mg/mL in MeOH with trace of formic acid
Injection volume: 10 L
Ionisation mode: Electrospray positive ion
Capillary voltage: 4.5 kV
Capillary temp: 360ºC Desolvation gas temperature: 300 ºC
Cone gas flow rate: 10 (arbitrary unit) Desolvation gas flow rate: 70 (arbitrary unit)
The retention time of daclatasvir is reported along with the major peak in the mass spectrum. The latter is reported as a mass/charge ratio.
9.98 min: 739.39545 (M+H+) m/z
HS-GC-MS: Instrument: Agilent 6890/5973/G1888
Column: DB-624, 30 m x 0.25 mm I.D. x 1.4 μm
Program: 50 C (5 min), 7 C/min to 120 C, 15 °C/min to 220 °C (8.3 min)
Injector: 150 C Transfer line temp: 280 C
Carrier: Helium, 1.2 mL/min Split ratio: 50/1
Solvents detected: Ethyl acetate
TLC: Conditions: Kieselgel 60F254. Ethyl acetate : methanol (95/5)
Single spot observed, Rf = 0.18. Visualisation with UV at 254 nm
The TLC was performed on the liberated free base.
IR: Instrument: Bruker Alpha FT-IR
Range: 4000-400 cm-1, neat
Peaks: 1723, 1697, 1643, 1523, 1439, 1235, 1099, 1024 cm-1
1H NMR: Instrument: Bruker Avance III 500
Field strength: 500 MHz Solvent: DMSO-d6 (2.50 ppm)
Spectral data: 0.77 (6H, d, J = 6.7 Hz), 0.83 (6H, d, J = 6.7 Hz), 2.01 (2H, m), 2.07 (2H, m), 2.12-2.27 (4H, m), 2.38 (2H, m), 3.54 (6H, s), 3.84 (2H, m), 3.97 (2H, m), 4.12 (2H, t, J = 7.7 Hz), 5.18 (2H, t, J = 7.0 Hz), 7.31 (2 N-H, d, J = 8.5 Hz), 7.94 (4H, d, J = 8.4 Hz), 7.99 (4H, d, J = 8.4 Hz), 8.16 (2H, s) ppm
Ethyl acetate estimated at 0.6% mass fraction was observed in the 1H NMR
13C NMR: Instrument: Bruker Avance III 500
Field strength: 126 MHz Solvent: DMSO-d6 (39.5 ppm)
Spectral data: 17.8, 19.6, 25.0, 29.0, 31.2, 47.3, 51.6, 52.9, 58.0, 115.1, 125.9, 126.6, 127.3, 131.8, 139.2, 149.4, 157.0, 171.1 ppm
Melting point: > 250 oC
Microanalysis: Found: C = 59.0%; H = 6.5%; N = 13.7% (August 2015)
Calc: C = 59.2%; H = 6.5%; N = 13.8% (Calculated for C40H50N8O6.2HCl)
REFERENCE
Australian NMI NATA Certification Daclatasvir – FixHepC
https://fixhepc.com/images/coa/NMI-NATA-Daclatasvir-Certification.pdf
Oct 7, 2015 – Compound Name: Daclatasvir dihydrochloride … Note: The assigned stereochemistry of this sample of daclatasvir has not …. Melting point:.
CLIP 2
Full Text Article – European Journal of Pharmaceutical and Medical …
Nov 28, 2016 – Daclatasvir dihydrochloride (DCLD) is a new drug …. DSC thermogram of daclatasvirdihydrochloriderealed drug melting point at 273.600C as …
CLIP 3
DCV dihydrochloride (anhydrous) is a white to yellow, non hygroscopic powder which is highly soluble in water (>700mg/mL). Solubility is higher at low pH. In aqueous buffers over the physiological pH range (pH 1.2-6.8) solubility is very low (4mg/mL to 0.004 mg/mL) due to the slow formation of the less soluble hydrated form. Water content in the drug substance is adequately controlled by in process tests. The desired anhydrous crystalline form of DCV dihydrochloride (N-2) is consistently produced and has been shown to not change on storage.
[DOC]AusPAR Daclatasvir dihydrochloride – Therapeutic Goods Administration
https://www.tga.gov.au/sites/default/…/auspar-daclatasvir–dihydrochloride-151214.do…
Dec 14, 2015 – Australian Public Assessment Report for daclatasvir dihydrochloride …. Figure 1:Chemical structure of daclatasvir dihydrochloride. …… 24 weeks is based on a selected literaturereview mostly of studies in patients with GT-1.
CLIP 4
The structure of the active substance has been confirmed by UV, IR, Raman and 1 H and 13C NMR spectroscopy, MS spectrometry, and crystal X-Ray diffraction.
Daclatasvir is a white to yellow crystalline non-hygroscopic powder. It is freely soluble in water, dimethyl sulfoxide, methanol; soluble in ethanol (95%); practically insoluble in dichloromethane, tetrahydrofuran, acetonitrile, acetone and ethyl acetate.
Daclatasvir is a chiral molecule with four stereocenters (1,1’, 2, 2;) in the S configuration. The synthetic strategy and process design such as starting material and reagent selection, process parameters, and in-process controls ensure the desired configuration at each of the four chiral centers. In addition, the established control strategy minimizes epimerization and eliminates other diastereomeric impurity formation in each step.
Polymorphism has been observed for daclatasvir hydrochloride. Although two neat crystalline dihydrochloride salts, N1 and N-2 have been identified in screening studies, it has been confirmed that the form N-2 is the thermodynamically most stable polymorph and only this form produced by the proposed synthetic process.
Manufacture, characterisation and process controls
Daclatasvir dihydrochloride is synthesised in three main steps using three commercially available well defined starting materials with acceptable specifications. The synthesis involves an alkylation and formation of the imidazole ring, a coupling reaction and the formation of the hydrochloride salt.
As mentioned above, the synthetic process has been designed to ensure the correct configuration at each of the four chiral centres is achieved. In addition, it has been demonstrated that the stereogenic centres do not epimerize during normal or stressed processing conditions.
The manufacturing process has been developed using a combination of conventional univariate studies and elements of QbD such as risk assessment.
The characterisation of the active substance and its impurities are in accordance with the EU guideline on chemistry of new active substances. Potential and actual impurities were well discussed with regards to their origin and characterised. Adequate in-process controls are applied during the synthesis. The specifications and control methods for intermediate products, starting materials and reagents have been presented.
The active substance specification includes tests for: appearance, colour, identity (IR/Raman, HPLC), assay (HPLC), impurities (HPLC), residual solvents (GC), HCl content (titration), total inorganic impurities (ICP-MS), and particle size (laser light scattering). The absence of a test for chiral purity in the active substance specification has been adequately justified based on the stereochemical control during the synthetic process and demonstration that there is no epimerization during normal or stressed processing conditions. Similarly, since the N-2 form of daclatasvir hydrochloride is the thermodynamically most stable polymorph and, is consistently produced by the synthetic process and remained unchanged during storage under long-term or accelerated conditions, this parameter is not included in the specification
CLIP5
SEE
http://www.accessdata.fda.gov/drugsatfda_docs/nda/2015/206843Orig1s000ChemR.pdf
CLIP6
Daclatasvir dihydrochloride
References
- 1 Statement on a Nonproprietary Name Adopted by the USAN Council
- 2 Gao, Min; Nettles, Richard E.; Belema, Makonen; Snyder, Lawrence B.; Nguyen, Van N.; Fridell, Robert A.; Serrano-Wu, Michael H.; Langley, David R.; Sun, Jin-Hua; O’Boyle, Donald R., II; Lemm, Julie A.; Wang, Chunfu; Knipe, Jay O.; Chien, Caly; Colonno, Richard J.; Grasela, Dennis M.; Meanwell, Nicholas A.; Hamann, Lawrence G. (2010). “Chemical genetics strategy identifies an HCV NS5A inhibitor with a potent clinical effect”. Nature 465 (7294): 96–100. doi:10.1038/nature08960. PMID 20410884.
- 3 Bell, Thomas W. (2010). “Drugs for hepatitis C: unlocking a new mechanism of action”. ChemMedChem 5 (10): 1663–1665. doi:10.1002/cmdc.201000334. PMID 20821796.
- 4 Modeling shows that the NS5A inhibitor daclatasvir has two modes of action and yields a shorter estimate of the hepatitis C virus half-life. Guedj, J et al. Proceedings of the National Academy of Sciences. February 19, 2013.
- 5 AASLD: Daclatasvir with Pegylated Interferon/Ribavirin Produces High Rates of HCV Suppression. Highleyman, L. HIVandHepatitis.com. 6 December 2011.
- 6Preliminary Study of Two Antiviral Agents for Hepatitis C Genotype 1. Lok, A et al. New England Journal of Medicine. 366(3):216-224. January 19, 2012.
- 7“Bristol-Myers’ Daclatasvir, Asunaprevir Cured 77%: Study”. Bloomberg. Apr 19, 2012.
- 8AASLD: Daclatasvir plus Asunaprevir Rapidly Suppresses HCV in Prior Null Responders. Highleyman, L. HIVandHepatitis.com. 8 November 2011.
- 9High rate of response to BMS HCV drugs in harder-to-treat patients – but interferon-free prospects differ by sub-genotype. Alcorn, K. Aidsmap.com. 12 November 2012.
- 10AASLD 2012: Sofosbuvir + Daclatasvir Dual Regimen Cures Most Patients with HCV Genotypes 1, 2, or 3. Highleyman, L. HIVandHepatitis.com. 15 November 2012.
- 11Mark Sulkowski et al. (January 16, 2014). “Daclatasvir plus Sofosbuvir for Previously Treated or Untreated Chronic HCV Infection”. New England Journal of Medicine. doi:10.1056/NEJMoa1306218.
- 12“www.who.int” (PDF).
| WO2004005264A2 * | 7 Jul 2003 | 15 Jan 2004 | Axxima Pharmaceuticals Ag | Imidazole compounds for the treatment of hepatitis c virus infections |
| WO2008021927A2 * | 9 Aug 2007 | 21 Feb 2008 | Squibb Bristol Myers Co | Hepatitis c virus inhibitors |
| WO2008021928A2 * | 9 Aug 2007 | 21 Feb 2008 | Squibb Bristol Myers Co | Hepatitis c virus inhibitors |
| WO2008021936A2 * | 9 Aug 2007 | 21 Feb 2008 | Squibb Bristol Myers Co | Hepatitis c virus inhibitors |
सुकून उतना ही देना प्रभू, जितने से जिंदगी चल जाये।औकात बस इतनी देना,कि औरों का भला हो जाये।………..P.S. : The views expressed are my personal and in no-way suggest the views of the professional body or the company that I represent.
सुकून उतना ही देना प्रभू, जितने से जिंदगी चल जाये। औकात बस इतनी देना, कि औरों का भला हो जाये।
 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO …..FOR BLOG HOME CLICK HERE
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO …..FOR BLOG HOME CLICK HERE
Join me on google plus  Googleplus
Googleplus
 amcrasto@gmail.com
amcrasto@gmail.com
 LIONEL MY SON
LIONEL MY SONHe was only in first standard in school when I was hit by a deadly one in a million spine stroke called acute transverse mylitis, it made me 90% paralysed and bound to a wheel chair, Now I keep him as my source of inspiration and helping millions, thanks to millions of my readers who keep me going and help me to keep my son happy

सुकून उतना ही देना प्रभू, जितने से
जिंदगी चल जाये।
औकात बस इतनी देना,
कि औरों का भला हो जाये।
जिंदगी चल जाये।
औकात बस इतनी देना,
कि औरों का भला हो जाये।
//////////
Ozanimod, RPC1063


cas 1306760-87-1
Ozanimod, RPC1063
Receptos, Inc. INNOVATOR
IUPAC/Chemical name: (S)-5-(3-(1-((2-hydroxyethyl)amino)-2,3-dihydro-1H-inden-4-yl)-1,2,4-oxadiazol-5-yl)-2-isopropoxybenzonitrile
Benzonitrile, 5-(3-((1S)-2,3-dihydro-1-((2-hydroxyethyl)amino)-1H-inden-4-yl)-1,2,4-oxadiazol-5-yl)-2-(1-methylethoxy)-

SMILES: N#CC1=CC(C2=NC(C3=CC=CC4=C3CC[C@@H]4NCCO)=NO2)=CC=C1OC(C)C
C23H24N4O3
Molecular Weight: 404.46
Elemental Analysis: C, 68.30; H, 5.98; N, 13.85; O, 11.87
Ozanimod is a selective sphingosine 1 phosphate receptor modulators and methods which may be useful in the treatment of S1P1-associated diseases. ozanimod, a sphingosine-1-phosphate receptor 1 (S1P1) agonist in Phase III studies as a treatment for ulcerative colitis and multiple sclerosis (MS). Although Novartis’s S1P1 modulator Gilenya has been available to treat MS since 2010,
Relapsing multiple sclerosis (RMS) is a chronic autoimmune disorder of the central nervous system (CNS), characterized by recurrent acute exacerbations (relapses) of neurological dysfunction followed by variable degrees of recovery with clinical stability between relapses (remission). The CNS destruction caused by autoreactive lymphocytes can lead to the clinical symptoms, such as numbness, difficulty walking, visual loss, lack of coordination and muscle weakness, experienced by patients. The disease invariably results in progressive and permanent accumulation of disability and impairment, affecting adults during their most productive years. RMS disproportionately affects women, with its peak onset around age 30. In the past, the treatments for RMS were generally injectable agents with significant side effects. There is a substantial market opportunity for effective oral RMS therapies with improved safety and tolerability profiles.
RPC1063 is a novel, orally administered, once daily, specific and potent modulator of the sphingosine 1-phosphate 1 receptor (S1P1R) pathway. The S1P1R is expressed on white blood cells (lymphocytes), including those responsible for the development of disease. S1P1R modulation causes selective and reversible retention, or sequestration, of circulating lymphocytes in peripheral lymphoid tissue. This sequestration is achieved by modulating cell migration patterns (known as “lymphocyte trafficking”), specifically preventing migration of autoreactive lymphocytes to areas of disease inflammation, which is a major contributor to autoimmune disease. S1P1R modulation may also involve the reduction of lymphocyte migration into the central nervous system (CNS), where certain disease processes take place. This therapeutic approach diminishes the activity of autoreactive lymphocytes that are the underlying cause of many types of autoimmune disease.
WO 2015066515
Scheme 3:

Reagents: (i) (a) MsCl, pyridine; (b) TsCl, pyridine; (c) NsCl, pyridine; (d) SOCl2, DCM; (e) SOCl2, pyridine, DCM; (f) NaN3, PPh3, CBr4; (ii) (a) DIEA, DMA, HNR’R”; (b) DIEA, NaBr or Nal, DMA, HNR’R”.
Enantiomerically enriched material can be prepared in the same manner outlined in Scheme 3 using the (R)- or (5)-indanols.
Scheme 4:

Reagents: (i) Zn(CN)2, Pd(PPh3)4, NMP; (ii) (i?)-2-methylpropane-2-sulfmamide, Ti(OEt)4, toluene; (iii) NaBH4, THF; (iv) 4M HCl in dioxane, MeOH; (v) Boc20, TEA, DCM; (vi) NH2OH HCl, TEA, EtOH; (vii) HOBt, EDC, substituted benzoic acid, DMF (viii) 4M HCl in dioxane; (ix) (a) R’-LG or R”-LG, where LG represents a leaving group, K2C03, CH3CN; (b) R -C02H or R2-C02H, HOBt, EDC, DMF or R -COCl or R2-COCl, TEA, DCM; (c) R -S02C1 or R3-S02C1, TEA, DCM (d) R2-CHO, HO Ac, NaBH4 or NaCNBH3 or Na(OAc)3BH, MeOH; (e) R -OCOCl or R2-OCOCl, DIEA, DMF; (f) HN(R5R5), CDI, TEA, DCM; (g) H2NS02NH2, Δ, dioxane; (h)
(R)-tert-butyl 2-(tert-butyldimethylsilyloxy)ethyl(4-cyano-2 ,3-dihydro- lH-inden- 1-yl)carbamate INT-16)

Prepared using General Procedure 9. To a flame-dried flask under N2 was added {R)-tert- vXy\ 4-cyano-2,3-dihydro-iH-inden-l-ylcarbamate INT-8 (8.3 g, 32.1 mmol) in anhydrous DMF (240 mL). The reaction mixture was cooled to 0°C and sodium hydride (3.8 g, 60% in oil, 160.6 mmol) was added portionwise. After stirring at 0°C for 2.75 h, (2-bromoethoxy)(tert-butyl)dimethylsilane (16.9 mL, 70.7 mmol) was added. The ice bath was removed after 5 mins and the reaction mixture was allowed to warm to room temperature. After 1.5 h, the reaction mixture was quenched by the slow addition of sat. NaHC03 at 0°C. Once gas evolution was complete the reaction was extracted with EA. The organic layers were washed with water and brine, dried over MgS04 and concentrated. The product was purified by chromatography (EA / hexanes) to provide 10.76 g (80%) of {R)-tert-bvXy\ 2-(tert-butyldimethylsilyloxy)ethyl(4-cyano-2,3-dihydro-iH-inden-l-yl)carbamate INT-16 as a colorless oil. LCMS-ESI (m/z) calculated for C23H36N203Si: 416.6; found 317.2 [M-Boc]+ and 439.0 [M+Na]+, tR = 4.04 min (Method 1). 1H NMR (400 MHz, CDC13) δ 7.46 (d, J = 7.6, 1H), 7.38- 7.32 (m, 1H), 7.33 – 7.18 (m, 1H), 5.69 (s, 0.5 H), 5.19 (s, 0.5 H), 3.70 (ddd, J = 48.8, 26.6, 22.9, 1.5 H), 3.50 – 3.37 (m, 1H), 3.17 (ddd, J = 16.7, 9.4, 2.2, 2H), 2.93 (m, 1.5 H), 2.45 (s, 1H), 2.21 (dd, J = 24.5, 14.5, 1H), 1.56 – 1.37 (bs, 4.5H), 1.22 (bs, 4.5H), 0.87 – 0.74 (m, 9H), -0.04 (dd, J = 26.6, 8.2, 6H). 13C NMR (101 MHz, CDC13) δ 155.03, 146.55, 145.54, 131.16, 130.76, [128.11, 127.03], 117.58, 109.20, 79.88, [63.93, 61.88], [61.44, 60.34], [49.73, 46.76], 30.30, 29.70, 28.44, 28.12, [25.87, 25.62], -5.43. (5)-tert-butyl 2-(tert-butyldimethylsilyloxy)ethyl(4-cyano-2,3-dihydro- 1 H-inden- 1 -yl)carbamate INT- 17 is prepared in an analogous fashion using INT-9.
(R)-tert-butyl 2-(tert-butyldimethylsilyloxy)ethyl (4-(N-hydroxycarbamimidoyl)-2,3-dihydro-lH-inden-l-yl)carbamate (INT-18)

Prepared using General Procedure 3. To a solution of (R)-tert-butyl 2-(tert-butyldimethylsilyloxy)ethyl(4-cyano-2,3-dihydro-iH-inden-l-yl)carbamate INT-16 (12.0 g, 28.9 mmol) in EtOH (120 mL), under an atmosphere of N2 was added hydroxylamine-HCl (6.0 g, 86.5 mmol) and triethylamine (13.4 mL, 9.7 g, 86.5 mmol). The reaction mixture was refluxed at 80°C for 4 h. The reaction mixture was cooled to room temperature and concentrated to dryness and then diluted with DCM (500 mL). The organic layer was washed with NaHC03, water, and brine. The combined organic layers were dried over MgSC^ and concentrated to produce 11.8 g of {R)-tert- vXy\ 2-(tert-butyldimethylsilyloxy) ethyl (4-(N-hydroxycarbamimidoyl)-2,3-dihydro-iH-inden-l-yl)carbamate INT-18 as a white foamy solid, which was used without purification in the next experiment. LCMS-ESI (m/z) calculated for C23H39N304Si: 449.7; found 350.2 [M-Boc]+ and 472.2 [M+Na]+, tR = 1.79 min (Method 1). 1H NMR (400 MHz, CDC13) δ 7.32 (t, J= 7.3 Hz, 1H), 7.21 – 7.07 (m, 2H), 5.69 (s, 0.5 H), 5.19 (s, 0.5 H), 4.89 (s, 2H), 3.85 – 3.50 (m, 2H), 3.31 (ddd, J = 12.2, 9.2, 2.5 Hz, 2H), 3.28 – 3.03 (m, 2H), 3.03 – 2.70 (m, 1H), 2.29 (t, J= 23.6 Hz, 1H), 1.43 (bs, 4.5H), 1.28 (bs, 4.5H), 1.16 – 1.04 (m, 1H), 0.90 – 0.71 (m, 9H), 0.08 – -0.14 (m, 6H). 13C NMR (101 MHz, CDC13) δ 170.99, [156.20, 155.62], 152.38, [144.53, 143.57], [141.82, 141.21], 129.61, 126.78, [126.59, 126.25], [125.02, 124.77], [79.91, 79.68], 64.04, 61.88, [61.57, 61.23], [46.03, 45.76], 30.76, 30.21, [28.53, 28.28], 25.95, [25.66, 25.29], 25.13, [18.28, 17.94], 3.72, -5.34. (S)-tert-butyl 2-(tert-butyldimethylsilyloxy)ethyl (4-(N-hydroxycarbamimidoyl)-2,3-dihydro-lH-inden-l-yl)carbamate INT-19 is prepared in an analogous fashion using INT- 17.
(R)-tert-butyl 2-(tert-butyldimethylsilyloxy)ethyl(4-(5-(3-cyano-4-isopropoxyphenyl)-l,2,4-oxadiazol-3-yl)-2,3-dihydro-lH-inden-l-yl)carbamate and (R)-tert-butyl 4-(5-(3-cyano-4-isopropoxyphenyl)-l,2,4-oxadiazol-3-yl)-2,3-dihydro-lH-inden-l-yl) (2-hydroxethyl) carbamate

Prepared using General Procedure 4. To a solution of 3-cyano-4-isopropoxybenzoic acid (4.5 g, 21.9 mmol) in anhydrous DMF (100 mL) was added HOBt (5.4 g, 40.0 mmol) and EDC (5.6 g, 29.6 mmol). After 1 h, (R)-tert-butyl 2-(tert-butyldimethylsilyloxy)ethyl (4-(N-hydroxycarbamimidoyl)-2,3-dihydro-iH-inden-l-yl)carbamate INT- 18 (11.8 g, 26.3 mmol) was added and the reaction mixture was stirred at room temperature for 2 h. LCMS analysis showed complete conversion to the intermediate, (R)-tert-butyl 2-(tert-butyldimethylsilyloxy) ethyl (4-(N-(3-cyano-4-isopropoxybenzoyloxy) carbamimidoyl)-2,3-dihydro-7H-inden-l-yl)carbamate INT-20. The reaction mixture was then heated to 80°C for 12 h. The reaction mixture was cooled to room temperature and diluted with EA (250 mL). NaHC03 (250 mL) and water (350 mL) were added until all the solids dissolved. The mixture was extracted with EA and the organic layers washed successively with water and brine. The organic layers were dried over MgS04 and concentrated to produce 15.3 g of a mixture of (R)-tert-butyl 2-(tert-butyldimethylsilyloxy)ethyl(4-(5 -(3 -cyano-4-isopropoxyphenyl)-l,2,4-oxadiazol-3-yl)- 2,3-dihydro-iH-inden-l-yl) carbamate INT-21, and the corresponding material without the TBS protecting group, {R)-tert-bvXy\ 4-(5-(3-cyano-4-isopropoxyphenyl)-l,2,4-oxadiazol-3-yl)-2,3-dihydro-iH-inden-l-yl) (2-hydroxy ethyl) carbamate INT-22. The mixture was a brown oil, which could used directly without further purification or purified by chromatography (EA/hexane). INT-21: LCMS-ESI (m/z) calculated for C34H46N405Si: 618.8; found 519.2 [M-Boc]+ and 641.3 [M+Na]+, tR = 7.30 min (Method 1). 1H NMR (400 MHz, CDC13) δ 8.43 (d, J =
2.1, 1H), 8.34 (dd, J = 8.9, 2.2, 1H), 8.07 (d, J= 8.1, 1H), 7.46 – 7.26 (m, 2H), 7.12 (d, J = 9.0, 1H), 5.85 (s, 0.5H), 5.37 (s, 0.5H), 4.80 (dt, J = 12.2, 6.1, 1H), 3.92 – 3.32 (m, 3.5 H), 3.17 (s, 2H), 2.95 (s, 0.5 H), 2.62 – 2.39 (m, 1H), 2.38 – 2.05 (m, 1H), 1.53 (s, 4.5H), 1.48 (d, J = 6.1, 6H), 1.33 – 1.27 (m, 4.5H), 0.94 – 0.77 (m, 9H), 0.01 (d, J = 20.9, 6H). 13C NMR (101 MHz, DMSO) δ 173.02, 169.00, 162.75, [156.22, 155.52], [145.18, 144.12], [143.39, 142.76], 134.16, 133.89, 128.20, [128.01, 127.85], [127.04, 126.90], 126.43, 123.31, 116.93, 115.30, 113.55, 103.96, [79.95, 79.68], 72.73, 67.61, 63.42, [61.91, 61.77], 60.99, 46.11, 31.78, [30.47, 29.87], [28.55, 28.26], 25.93, 21.75, 18.30, 0.00, -5.37. INT-22: LCMS-ESI calculated for C28H32N405: 504.6; found 527.2 [M+Na]+, tR = 2.65 min (Method 1). 1H NMR (400 MHz, CDC13) δ 8.36 (d, J = 2.1, 1H), 8.27 (dd, J = 8.9, 2.2, 1H), 8.03 (d, J = 7.2, 1H), 7.35 – 7.26 (m, 2H), 7.06 (d, J = 9.0, 1H), 5.44 (s, 1H), 4.73 (dt, J= 12.2, 6.1, 1H), 3.64 (s, 2H), 3.44 (ddd, J= 17.5, 9.5,
3.2, 2H), 3.11 (dt, J = 17.4, 8.6, 3H), 2.54 – 2.38 (m, 1H), 2.04 (td, J = 17.6, 8.8, 1H), 1.50 – 1.24 (m, 15H).
(S)-tert-butyl 2-(tert-butyldimethylsilyloxy)ethyl(4-(5-(3-cyano-4-isopropoxyphenyl)-l,2,4-oxadiazol-3-yl)-2,3-dihydro-iH-inden-l-yl)carbamate INT-23 and {S)-tert- vXy\ 4-(5-(3-cyano-4-isopropoxyphenyl)-l,2,4-oxadiazol-3-yl)-2,3-dihydro-iH-inden-l-yl) (2-hydroxyethyl) carbamate INT-24 were made in an analogous fashion.
(S) IS DESIRED CONFIGURATION
……………………………………
(S)-tert-Butanesulfinamide
 CAS 343338-28-3
CAS 343338-28-3
 3-CYANO-4-ISOPROPOXYBENZOIC ACID;3-cyano-4-(propan-2-yloxy)benzoic acid;5-(1-hydroxyvinyl)-2-isopropoxybenzonitrile
3-CYANO-4-ISOPROPOXYBENZOIC ACID;3-cyano-4-(propan-2-yloxy)benzoic acid;5-(1-hydroxyvinyl)-2-isopropoxybenzonitrile
cas 258273-31-3
 (S)-1-Amino-2,3-dihydro-1H-indene-4-carbonitrile hydrochloride
(S)-1-Amino-2,3-dihydro-1H-indene-4-carbonitrile hydrochloride
cas 1306763-57-4 HCl, 1213099-69-4 FREE BASE
4-bromo-2,3-dihydro-1H-inden-1-one
cas 15115-60-3
Carbamic acid, N-[(1S)-4-cyano-2,3-dihydro-1H-inden-1-yl]-, 1,1-dimethylethyl ester, cas 1306763-31-4
(S) IS DESIRED CONFIGURATION
……………….
CAS 1306763-70-1, Carbamic acid, N-[(1S)-2,3-dihydro-4-[(hydroxyamino)iminomethyl]-1H-inden-1-yl]-, 1,1-dimethylethyl ester
…………………
CAS 1306763-71-2, Carbamic acid, N-[(1S)-4-[5-[3-cyano-4-(1-methylethoxy)phenyl]-1,2,4-oxadiazol-3-yl]-2,3-dihydro-1H-inden-1-yl]-, 1,1-dimethylethyl ester
1306760-73-5, Benzonitrile, 5-[3-[(1S)-1-amino-2,3-dihydro-1H-inden-4-yl]-1,2,4-oxadiazol-5-yl]-2-(1-methylethoxy)-
………………………..
1306763-63-2,
………………….

86864-60-0, (2-Bromoethoxy)dimethyl-tert-butylsilane
Synthesis
……………………………………
WO 2011060392
http://www.google.com/patents/WO2011060392A1?cl=en
(R)-N-(4-cyano-2,3-dihydro-lH-indene-l-ylidene)-2-methylpropane-^
(INT-4
[0304] To l-oxo-2,3-dihydro-/H-indene-4-carbonitrile INT-1 (42.5 g, 0.27 mol) and (R)-2- methylpropane-2-sulfmamide (36.0 g, 0.30 mol) in toluene (530 mL) was added titanium tetraethoxide (84.1 mL, 92.5 g, 0.40 mol) and the reaction mixture was heated at 60°C for 12 h under N2. The crude (R)-N-(4-cyano-2,3-dihydro-lH-indene-l-ylidene)-2-methylpropane- 2-sulfinamide INT-4 was used directly in the next experiment. LCMS-ESI (m/z) calculated for C14Hi6N2OS: 260.3; found 261.1 [M+H]+, tR= 3.19 min.
[0305] (R)-N'((R)-4-cyano-2,3-dihydro-lH nden-l-yl)-2-n thylprop ne-2-sulfirmmide
(INT-5)
[0306] To a flask containing the crude suspension of (R)-N-(4-cyano-2,3-dihydro-iH-indene- l-ylidene)-2-methylpropane-2-sulfrnaniide INT -4 under N2 was added THF (1.0 L) and the reaction mixture cooled to -78°C. Sodium borohydride (40.9 g, 1.08 mol) was added portion- wise over 30 mins. (The internal temperature did not rise during the addition). The reaction mixture was stirred at -78°C for 30 mins, half out of the bath for 30 mins, then warmed to 0°C over 1 h. The 0°C reaction mixture was placed in an ice bath and quenched with brine (100 mL) followed by saturated sodium potassium tartrate (420 mL) and the Ti salts precipitated. The reaction mixture was diluted with EA (1.5 L) and stirred at room temperature overnight. The organic layers were decanted and washed successively with saturated NH4CI, water, and brine. The organic layers were dried over MgS04 and filtered through a pad of MgS04. The filtrate was concentrated to produce 52.9 g of crude (R)-N-((/?)-4-cyano-2,3-dihydro-lH- inden-l-yl)-2-methylpropane-2-sulfmamide INT-5 as a brown oil, which was used directly in the next step. LCMS-ESI (m/z) calculated for C14H18 2OS: 262.3; found 263.1 [M+H]+, tR = 2.99 min. 1H NMR (400 MHz, CDC13) δ 7.89 (d, J = 7.7, 1H), 7.56 (t, J = 6.8, 1H), 7.36 (t, J = 7.7, 1H), 4.97 (q, J = 7.5, 1H), 3.50 (d, J = 7.6, 1H), 3.22 (ddd, J = 16.9, 8.8, 3.9, 1H), 3.01 (dt, J = 22.4, 6.9, 1H), 2.70 – 2.53 (m, 1H), 2.15 – 1.95 (m, 1H), 1.33 – 1.20 (m, 9H).
[0307] (R)-l-amino-2,3-dihydro-lH-indene-l-yl)-4-carbonitrile (T^T-6)
[0308] To crude (R)-N-((R)-4-cyano-2,3-dihydro-iH-inden-l-yl)-2-methylpropane-2- sulfinamide INT-5 (52.9 g, 0.20 mol) in MeOH (200 mL) was added 4N HC1 in dioxane (152.0 mL, 0.60 mol) and the resulting yellow suspension was stirred at room temperature for 1.5 h. The crude reaction mixture was diluted with MeOH (500 mL) and filtered to remove some Ti by-products. The filtrate was concentrated and the resulting solid refluxed in acetonitrile (500 mL). The resulting white solid was collected to produce 13.0 g (31% over 3 steps) of the HC1 salt of (R)-l-amino-2,3-dihydro-7H-indene-l-yl)-4-carbonitrile INT-6. LCMS-ESI (m/z) calculated for Ci0H10N2: 158.2; found 142.0 [M-NH2]+, fR = 0.84 min. Ή NMR (400 MHz, DMSO) δ 8.61 (s, 3H), 7.96 (d, J = 7.7, 1H), 7.83 (d, J = 7.5, 1H), 7.52 (t, J = 7.7, 1H), 4.80 (s, 1H), 3.23 (ddd, J = 16.6, 8.7, 5.2, 1H), 3.05 (ddd, J = 16.6, 8.6, 6.3, 1H), 2.62 – 2.51 (m, 1H), 2.15 – 2.01 (m, 1H). 13C NMR (101 MHz, DMSO) δ 148.09, 141.15, 132.48, 130.32, 127.89, 117.27, 108.05, 54.36, 39.08, 29.64. The free base can be prepared by extraction with IN NaHC03and DCM. LCMS-ESI (m/z) calculated for Ci0H10N2: 158.2; found 142.0 [M-NH2]+, tR = 0.83 min. 1H NMR (400 MHz, CDC13) δ 7.52 – 7.38 (m, 2H), 7.23 (dd, 7 = 17.4, 9.8, 1H), 4.35 (t, J = 7.6, 1H), 3.11 (ddd, 7 = 16.8, 8.7, 3.2, 1H), 2.89 (dt, J = 16.9, 8.5, 1H), 2.53 (dddd, J = 12.8, 8.1, 7.3, 3.2, 1H), 1.70 (dtd, J = 12.8, 8.8, 8.0, 1H). 13C NMR (101 MHz, DMSO) δ 150.16, 146.67, 130.19, 128.74, 127.38, 117.77, 107.42, 56.86, 38.86, 29.14. Chiral HPLC: (R)-l-amino-2,3-dihydro-7H-indene-l-yl)-4-carbonitrile was eluted using 5% EtOH in hexanes, plus 0.05% TEA: 95% ee, ¾ = 23.02 min. The (S)- enantiomer INT-7 was prepared in an analogous fashion using (5)-2-methylpropane-2- sulfinamide. tR for (S)-enantiomer = 20.17 min.
[0309] (R)-tert-butyl 4-cyano-2,3-dihydro-lH-inden-l-ylcarbamate (INT-8)
[0310] To ( ?)-l-amino-2,3-dihydro-/H-indene-l-yl)-4-carbonitrile HC1 INT-6 (11.6 g, 59.6 mmol) in DCM (100 mL) at 0°C was added TEA (12.0 mL, 131.0 mmol). To the resulting solution was added a solution of Boc anhydride (14.3 g, 65.6 mmol) in DCM (30 mL) and the reaction mixture stirred at room temperature for 1.5 h. The reaction mixture was washed with brine, and the organic layers were dried over MgS04 and filtered. Additional DCM was added to a total volume of 250 mL and Norit (4.5 g) was added. The product was refluxed for 15 mins and the hot mixture filtered through a pad of celite / silica. The filtrate was concentrated and recrystallized from EA (50 mL) and hexane (150 mL) to produce 12.93 g (84%) of (/?)-tert-butyl 4-cyano-2,3-dihydro-iH-inden-l-ylcarbamate INT-8 as an off-white solid. LCMS-ESI (m/z) calculated for C15H18N202: 258.3; found 281.1 [M+Na]+, tR = 3.45 min. Elemental Analysis determined for C^H^^O^ C calculated = 69.74%; found = 69.98%. H calculated = 7.02%; found = 7.14%. N calculated = 10.84%; found = 10.89%. 1H NMR (400 MHz, CDC13) δ 7.64 – 7.49 (m, 2H), 7.34 (dt, / = 7.7, 3.8, 1H), 5.36 – 5.20 (m, 1H), 4.78 (d, J = 6.8, 1H), 3.20 (ddd, J = 16.9, 8.9, 3.3, 1H), 3.02 (dt, J = 25.4, 8.4, 1H), 2.82 – 2.53 (m, 1H), 1.88 (dq, J = 13.2, 8.6, 1H), 1.55 – 1.44 (m, 9H). 13C NMR (101 MHz, DMSO) δ 155.52, 146.68, 146.32, 130.89, 128.70, 127.63, 117.51, 107.76, 77.98, 55.09, 31.88, 29.11, 28.19. Chiral HPLC: (R)-tert-butyl 4-cyano-2,3-dihydro-lH-inden-l- ylcarbamate was eluted using 2.5% EtOH in hexanes: >99.9% ee, tR = 19.36 min. The (5)- enantiomer INT-9 was prepared in an analogous fashion using (S)-l-amino-2,3-dihydro-7H- indene-l-yl)-4-carbonitrile HC1. tR for (5)-enantiomer = 28.98 min.
General Procedure 3. Preparation oflndane Amide Oximes
[0311] To (R)- or (5)-tert-butyl 4-cyano-2,3-dihydro-7H-inden-l-ylcarbamate (1 eq) in EtOH
(0.56 M) was added hydroxylamine hydrochloride (3 eq) and TEA (3 eq) and the reaction mixture heated at 85°C for 1-2 h. The organic soluble amide oximes were isolated by removal of the solvent and partitioning between water and DCM. The water soluble amide oximes were chromatographed or used directly in the cyclization. Pure amide oximes can be obtained by recrystallization from alcoholic solvents.
[0312] (R)-tert-butyl 4-(N -hydroxy carbamimidoyl )-2, 3-dihydro-lH-inden-l -ylcarbamate
(INT-10)
[0313] Prepared using General Procedure 3. To (R)-tert-butyl 4-cyano-2,3-dihydro-iH- inden-1 -ylcarbamate INT-8 (15.0 g, 58.2 mmol) in EtOH (100 niL) was added hydroxylamine hydrochloride (12.1 g, 174.2 mmol) and TEA (17.6 mL, 174.2 mmol) and the reaction mixture heated at 85°C for 2 h. The solvents were removed and the resulting white solid was partitioned between water and DCM. The organic layers were dried over Na2S04, concentrated, and recrystallized from isopropanol (50 mL) to afford 14.4 g (85%) of (R)-tert- butyl 4-(N-hydroxycarbaniimidoyl)-2,3-dihydro-iH-inden-l-ylcarbamate INT-10 as white crystalline solid. LCMS-ESI (m/z) calculated for C15H21N303: 291.4; found 292.1 [M+H]+, ¾ = 2.04 min. 1H NMR (400 MHz, DMSO) δ 9.53 (s, 1H), 7.38 – 7.32 (m, 1H), 7.32 – 7.12 (m, 3H), 5.68 (s, 2H), 4.97 (q, J = 8.5, 1H), 3.07 (ddd, J = 16.6, 8.7, 2.6, 1H), 2.86 (dt, J = 16.8, 8.4, 1H), 2.30 (ddd, J = 12.6, 7.6, 3.6, 1H), 1.75 (dq, J = 12.3, 9.0, 1H), 1.44 (s, 9H). General Procedure 4. Cyclization to Indane Oxadiazole Amines
[0314] A solution of the appropriate acid (1 eq), HOBt (1.3 eq), and EDC (1.3 eq) in DMF
(0.08 M in acid) was stirred at room temperature under an atmosphere of N2. After the complete formation of the HOBt- acid complex (1-3 h), the (R)- or (5)-amide oxime (1.1 eq) was added to the mixture. After complete formation of the coupled intermediate (ca. 0.5- 2 h), the mixture was heated to 75-95°C until the cyclization was complete (8-12 h). The reaction mixture was diluted with saturated NaHC03 and extracted with EA. The combined organic extracts were dried, concentrated, and either purified by chromatography (EA/hexanes) or taken on directly. The oxadiazole was treated with HC1 (5N in dioxane, 5 eq) at 50-60°C for 0.5-6 h. The reaction mixture could be extracted (DCM /NaHC03), or the resulting HC1 salt concentrated, suspended in Et20, and collected. Pure indane amines can be obtained by recrystallization from alcoholic solvents or by chromatography.
( R)-tert-butyl 4-(5-( 3-cyano-4-isopropoxyphenyl)-l,2, 4-oxadiazol-3-yl )-2,3-dihydro-lH- inden-l-ylcarbamate (INT- 12)
[0315] Prepared using General Procedure 4. To a solution of 3-cyano-4-isopropoxybenzoic acid (7.74 g, 37.7 mmol) in DMF (50 mL) was added HOBt (6.02 g, 44.6 mmol) and EDC (8.53 g, 44.6 mmol) at room temperature. The reaction was stirred for 2 h until complete formation of the HOBt-acid complex. (R)-tert-butyl 4-(N-hydroxycarbamimidoyl)-2,3- dihydro-iH-inden-l-ylcarbamate INT-10 (10.0 g, 34.3 mmol) was added and the reaction mixture stirred at room temperature for 2 h until the formation of INT-11, (R)-tert-butyl 4- (N-(3-cyano-4-isopropoxybenzolyloxy) carbamimidoyl)-2,3-dihydro-iH-inden-l- ylcarbamate. The mixture was partitioned between EA and NaHC03 and the organic layer was collected and dried over MgS04. INT-11 (16.3 g, 34.0 mmol) was re-dissolved in DMF (50 mL) and the mixture was heated to 95°C for 12 hrs. The reaction was diluted with NaHC03 (200 mL) and extracted with EA (3 X 50 mL). The organic layer was dried over Na2S04and concentrated under reduced pressure to produce 12.8 g (81%) of (R)-tert-butyl 4- (5-(3-cyano-4-isopropoxyphenyl)- 1 ,2,4-oxadiazol-3-yl)-2,3-dihydro-iH-inden- 1-ylcarbamate INT-12 as a light brown solid and used without further purification in the next step. LCMS- ESI (m/z) calculated for C26H28N404: 460.5; found 483.2 [M+Na]+, tR = 4.25 min. Ή NMR (400 MHz, CDCI3) δ 8.43 (d, J = 2.1, 1H), 8.34 (dd, J = 8.9, 2.2, 1H), 8.09 (d, J = 7.6, 1H), 7.51 (d, / = 7.5, 1H), 7.39 (t, J = 7.6, 1H), 7.12 (d, J = 9.0, 1H), 5.28 (d, J = 8.2, 1H), 4.80 (hept, J = 6.0, 1H), 3.47 (ddd, J = 17.4, 8.9, 3.5, 1H), 3.27 – 3.03 (m, 1H), 2.68 (d, J = 8.7, 1H), 1.87 (td, J = 16.7, 8.5, 1H), 1.53 – 1.43 (m, 15H). 13C NMR (101 MHz, CDC13) δ 173.00, 168.82, 162.70, 155.68, 145.31, 142.96, 134.05, 133.83, 128.25, 127.21, 126.79, 123.09, 116.78, 115.24, 113.52, 103.87, 79.52, 72.70, 55.72, 33.86, 31.47, 28.39, 21.70. Chiral HPLC: (R)-tert-butyl 4-(5-(3-cyano-4-isopropoxyphenyl)-l,2,4-oxadiazol-3-yl)-2,3- dihydro-lH-inden-l-ylcarbamate was eluted using 20% /-PrOH in hexanes: >99.9% ee, ?R = 13.33 min. The (5)-enantiomer INT-13 was prepared in an analogous fashion using (S)-tert- butyl 4-cyano-2,3-dihydro-iH-inden-l-ylcarbamate using General Procedures 3 and 4 (tR for (Syenantiomer = 16.31 min).
( R )-5-( 3-(l -amino-2,3-dihydro-lH-inden-4-yl)-l,2, 4-oxadiazol-5-yl)-2-isopropoxy- benzonitrile h drochloride (Compound 49)
[0317] To (R)-tert-butyl 4-(5-(3-cyano-4-isopropoxyphenyl)-l,2,4-oxadiazol-3-yl)-2,3- dihydro-iH-inden-l-ylcarbamate(12.8 g, 27.8 mmol) in dioxane (200 mL) was added 4N HCl in dioxane (69 mL). The solution was heated to 55°C for 1 h, and product precipitated. Dioxane was removed and the resulting solid suspended in ether and collected. The material was recrystallized from MeOH (200 mL) to produce 8.11 g (81%) of (R)-5-(3-(l-amino-2,3- dihydro-iH-inden-4-yl)-l,2,4-oxadiazol-5-yl)-2-isopropoxybenzonitrile 49 as the HCl salt. LCMS-ESI (m/z): calcd for: C21H20N4O2: 360.4; found 383.2 [M+Na]+, tR = 2.49 min. Elemental Analysis and NMR spectra determined for C21H21N402C1 * 0.5 H20; C calculated = 62.14%; found = 62.25%. H calculated = 5.46%; found = 5.30%. N calculated = 13.80%; found = 13.84%. CI calculated = 8.73%; found = 8.34%. 1H NMR (400 MHz, DMSO) δ 8.71 (s, 3H), 8.49 (d, J = 2.3, 1H), 8.39 (dd, J = 9.0, 2.3, 1H), 8.11 (d, J = 7.6, 1H), 7.91 (d, J = 7.6, 1H), 7.55 (t, J = 8.5, 2H), 4.97 (hept, J = 6.1, 1H), 4.80 (s, 1H), 3.47 (ddd, J = 17.4, 8.7, 5.3, 1H), 3.23 (ddd, 7 = 17.4, 8.6, 6.4, 1H), 2.55 (ddd, 7 = 13.7, 8.3, 3.2, 1H), 2.22 – 1.97 (m, 1H), 1.38 (d, J = 6.0, 6H). 13C NMR (101 MHz, CDC13) δ 173.28, 167.98, 162.53, 143.69, 141.29, 134.59, 133.80, 128.93, 128.11, 127.55, 122.72, 115.87, 115.24, 114.91, 102.46, 72.54, 54.38, 31.51, 29.91, 21.47. Chiral HPLC of the free base: (R)-5-(3-(l-amino-2,3- dihydro-lH-inden-4-yl)-l,2,4-oxadiazol-5-yl)-2-isopropoxy benzonitrile was eluted using 15% i‘-PrOH in hexanes plus 0.3% DEA: > 99.9% ee, tR = 30.80 min.
(S)- 5-(3-(l-amino-2,3- dihydro-lH-inden-4-yl)-l,2,4-oxadiazol-5-yl)-2-isopropoxy-benzonitrile 50 was prepared in an analogous fashion from (S)-tert-b tyl 4-cyano-2,3-dihydro-lH-inden-l-ylcarbamate: >99.9% ee, tR for (5)-enantiomer = 28.58 min.
(R)-tert-butyl 2-(tert-butyldimethylsilyloxy)ethyl(4-cyano-2,3-dihydro-lH-inden-l- yl)carbamate ( -16)
[0366] Prepared using General Procedure 9. To a flame-dried flask under N2 was added (R)- tert-butyl 4-cyano-2,3-dihydro-iH-inden-l-ylcarbamate INT-8 (8.3 g, 32.1 mmol) in anhydrous DMF (240 mL). The reaction mixture was cooled to 0°C and sodium hydride (3.8 g, 60% in oil, 160.6 mmol) was added portionwise. After stirring at 0°C for 2.75 h, (2- bromoethoxy)(½rt-butyl)dimethylsilane (16.9 mL, 70.7 mmol) was added. The ice bath was removed after 5 mins and the reaction mixture was allowed to warm to room temperature. After 1.5 h, the reaction mixture was quenched by the slow addition of sat. NaHC03at 0°C. Once gas evolution was complete the reaction was extracted with EA. The organic layers were washed with water and brine, dried over MgS04 and concentrated. The product was purified by chromatography (EA / hexanes) to provide 10.76 g (80%) of (R)-teri-butyl 2-(tert- butyldimemylsilyloxy)emyl(4-cyano-2,3-dihydro-iH-inden-l-yl)carbamate INT-16 as a colorless oil. LCMS-ESI (m/z) calculated for C23H36N203Si: 416.6; found 317.2 [M-Boc]+ and 439.0 [M+Na]+, tR = 4.04 min (Method 1). 1H NMR (400 MHz, CDC13) δ 7.46 (d, J = 7.6, 1H), 7.38- 7.32 (m, 1H), 7.33 – 7.18 (m, 1H), 5.69 (s, 0.5 H), 5.19 (s, 0.5 H), 3.70 (ddd, J = 48.8, 26.6, 22.9, 1.5 H), 3.50 – 3.37 (m, 1H), 3.17 (ddd, J = 16.7, 9.4, 2.2, 2H), 2.93 (m, 1.5 H), 2.45 (s, 1H), 2.21 (dd, J = 24.5, 14.5, 1H), 1.56 – 1.37 (bs, 4.5H), 1.22 (bs, 4.5H), 0.87 – 0.74 (m, 9H), -0.04 (dd, J = 26.6, 8.2, 6H).13C NMR (101 MHz, CDC13) δ 155.03, 146.55, 145.54, 131.16, 130.76, [128.11, 127.03], 117.58, 109.20, 79.88, [63.93, 61.88], [61.44, 60.34], [49.73, 46.76], 30.30, 29.70, 28.44, 28.12, [25.87, 25.62], -5.43. (5)-tert-butyl 2-(tert- butyldimemylsilyloxy)emyl(4-cyano-2,3-dihydro-lH-inden-l-yl)carbamate INT-17 is prepared in an analogous fashion using INT -9. [0367] (R)-tert-butyl 2-(tert-butyldimethylsilyloxy)ethyl (4-(N-hydroxycarbamimidoyl)-2,3- dihydro-1 H-inden-1 -yl)carbamate (INT-18)
[0368] Prepared using General Procedure 3. To a solution of (R)-iert-butyl 2-(tert- butyldimemylsilyloxy)ethyl(4-cyano-2,3-dmydro-/H-inden-l-yl)carbamate INT-16 (12.0 g, 28.9 mmol) in EtOH (120 mL), under an atmosphere of N2 was added hydroxylamine-HCl (6.0 g, 86.5 mmol) and triemylamine (13.4 mL, 9.7 g, 86.5 mmol). The reaction mixture was refluxed at 80°C for 4 h. The reaction mixture was cooled to room temperature and concentrated to dryness and then diluted with DCM (500 mL). The organic layer was washed with NaHC03, water, and brine. The combined organic layers were dried over MgS04 and concentrated to produce 11.8 g of (R)-tert-butyl 2-(tert-butyldimethylsilyloxy) ethyl (4-(N- hydroxycarbamimidoyl)-2,3-dihydro-iH-inden-l-yl)carbamate INT-18 as a white foamy solid, which was used without purification in the next experiment. LCMS-ESI (m/z) calculated for C23H39N304Si: 449.7; found 350.2 [M-Boc]+ and 472.2 [M+Na]+, ¾ = 1.79 min (Method 1). 1H NMR (400 MHz, CDC13) δ 7.32 (t, / = 7.3 Hz, 1H), 7.21 – 7.07 (m, 2H), 5.69 (s, 0.5 H), 5.19 (s, 0.5 H), 4.89 (s, 2H), 3.85 – 3.50 (m, 2H), 3.31 (ddd, / = 12.2, 9.2, 2.5 Hz, 2H), 3.28 – 3.03 (m, 2H), 3.03 – 2.70 (m, 1H), 2.29 (t, J = 23.6 Hz, 1H), 1.43 (bs, 4.5H), 1.28 (bs, 4.5H), 1.16 – 1.04 (m, 1H), 0.90 – 0.71 (m, 9H), 0.08 – -0.14 (m, 6H). 13C NMR (101 MHz, CDC13) 6 170.99, [156.20, 155.62], 152.38, [144.53, 143.57], [141.82, 141.21], 129.61, 126.78, [126.59, 126.25], [125.02, 124.77], [79.91, 79.68], 64.04, 61.88, [61.57, 61.23], [46.03, 45.76], 30.76, 30.21, [28.53, 28.28], 25.95, [25.66, 25.29], 25.13, [18.28, 17.94], 3.72, -5.34. ^-tert-butyl 2-(tert-butyldimethylsilyloxy)ethyl (4-(N- hydroxycarbamimidoyl)-2,3-dihydro-lH-inden-l-yl)carbamate INT-19 is prepared in an analogous fashion using INT-17. [0369] (R)-tert-butyl 2-( tert-butyldimethylsilyloxy)ethyl( 4-( 5-( 3-cyano-4-isopropoxyphenyl)- l,2,4-oxadiazol-3-yl)-2,3-dihydro-lH-inden-l-yl)carbamate and (R)-tert-butyl 4-(5-(3-cyano- 4-isopropoxyphenyl )-l,2, 4-oxadiazol-3-yl)-2,3-dihydro-lH-inden-l-yl) (2-hydroxethyl) carbamate
[0370] Prepared using General Procedure 4. To a solution of 3-cyano-4-isopropoxybenzoic acid (4.5 g, 21.9 mmol) in anhydrous DMF (100 mL) was added HOBt (5.4 g, 40.0 mmol) and EDC (5.6 g, 29.6 mmol). After 1 h, {R)-tert-buiy\ 2-(tert-butyldimethylsilyloxy)ethyl (4- (N-hydroxycarbamimidoyl)-2,3-dihydro-iH-inden-l-yl)carbamate INT-18 (11.8 g, 26.3 mmol) was added and the reaction mixture was stirred at room temperature for 2 h. LCMS analysis showed complete conversion to the intermediate, (R)-tert-b\xty\ 2-(tert- butyldimethylsilyloxy) ethyl (4-(N-(3-cyano-4-isopropoxybenzoyloxy) carbamimidoyl)-2,3- dihydro-7H-inden-l-yl)carbamate INT-20. The reaction mixture was then heated to 80°C for 12 h. The reaction mixture was cooled to room temperature and diluted with EA (250 mL). NaHC03 (250 mL) and water (350 mL) were added until all the solids dissolved. The mixture was extracted with EA and the organic layers washed successively with water and brine. The organic layers were dried over MgS04 and concentrated to produce 15.3 g of a mixture of (R)-tert-butyl 2-(tert-butyldimethylsilyloxy)ethyl(4-(5-(3-cyano-4-isopropoxyphenyl)- 1 ,2,4- oxadiazol-3-yl)- 2,3-dihydro-iH-inden-l-yl) carbamate INT-21, and the corresponding material without the TBS protecting group, (R)-tert-butyl 4-(5-(3-cyano-4- isopropoxyphenyl)-l,2,4-oxadiazol-3-yl)-2,3-dihydro-iH-inden-l-yl) (2-hydroxyethyl) carbamate INT -22. The mixture was a brown oil, which could used directly without further purification or purified by chromatography (EA hexane). INT-21: LCMS-ESI (m/z) calculated for C34H46N4O5S1: 618.8; found 519.2 [M-Boc]+ and 641.3 [M+Na]+, tR = 7.30 min (Method 1). Ή NMR (400 MHz, CDC13) δ 8.43 (d, J = 2.1, 1H), 8.34 (dd, J = 8.9, 2.2, 1H), 8.07 (d, J = 8.1, 1H), 7.46 – 7.26 (m, 2H), 7.12 (d, / = 9.0, 1H), 5.85 (s, 0.5H), 5.37 (s, 0.5H), 4.80 (dt, J = 12.2, 6.1, 1H), 3.92 – 3.32 (m, 3.5 H), 3.17 (s, 2H), 2.95 (s, 0.5 H), 2.62 – 2.39 (m, 1H), 2.38 – 2.05 (m, 1H), 1.53 (s, 4.5H), 1.48 (d, J = 6.1, 6H), 1.33 – 1.27 (m, 4.5H), 0.94 – 0.77 (m, 9H), 0.01 (d, J = 20.9, 6H). 1C NMR (101 MHz, DMSO) δ 173.02, 169.00, 162.75, [156.22, 155.52], [145.18, 144.12], [143.39, 142.76], 134.16, 133.89, 128.20, [128.01, 127.85], [127.04, 126.90], 126.43, 123.31, 116.93, 115.30, 113.55, 103.96, [79.95, 79.68], 72.73, 67.61, 63.42, [61.91, 61.77], 60.99, 46.11, 31.78, [30.47, 29.87], [28.55, 28.26], 25.93, 21.75, 18.30, 0.00, -5.37. INT-22: LCMS-ESI calculated for C28H32N4Os: 504.6; found 527.2 [M+Na]+, tR = 2.65 min (Method 1). Ή NMR (400 MHz, CDC13) δ 8.36 (d, J = 2.1, 1H), 8.27 (dd, / = 8.9, 2.2, 1H), 8.03 (d, / = 7.2, 1H), 7.35 – 7.26 (m, 2H), 7.06 (d, / = 9.0, 1H), 5.44 (s, 1H), 4.73 (dt, J = 12.2, 6.1, 1H), 3.64 (s, 2H), 3.44 (ddd, / = 17.5, 9.5, 3.2, 2H), 3.11 (dt, J = 17.4, 8.6, 3H), 2.54 – 2.38 (m, 1H), 2.04 (td, J = 17.6, 8.8, 1H), 1.50 – 1.24 (m, 15H). (5 -teri-butyl 2-(tert-butyldimethylsilyloxy)ethyl(4-(5-(3-cyano-4- isopropoxyphenyl)-l,2,4-oxadiazol-3-yl)-2,3-dihydro-iH-inden-l-yl)carbamate INT-23 and (S)-terf-butyl 4-(5-(3-cyano-4-isopropoxyphenyl)-l,2,4-oxadiazol-3-yl)-2,3-dihydro-iH- inden-l-yl) (2-hydroxyethyl) carbamate INT -24 were made in an analogous fashion.
[0371] (R)-5-(3-(l-(2-hydroxyethylamino)-2,3-dihydro-lH-inden-4-yl)-l,2,4-oxadi zol-^ 2-isopropoxybenzonitrile (Compound 85)
[0372] To a solution of (R)-tert-butyl 2-(tert-butyldimethylsilyloxy)ethyl(4-(5-(3-cyano-4- isopropoxyphenyl)-l,2,4-oxadiazol-3-yl)-2,3-dihydro-7H-inden-l-yl)carbamate INT-21 and (R)-tert-butyl 4-(5-(3-cyano-4-isopropoxyphenyl)- 1 ,2,4-oxadiazol-3-yl)-2,3-dihydro-iH- inden-l-yl) (2-hydroxethyl) carbamate INT-22 (13.9 g, 27.5 mmol) in dioxane (70 mL) at 0°C was added 4N HCl in dioxane (68.8 g, 275.4 mmol). The reaction mixture was warmed to room temperature and then heated to 50°C for 1 h. The resulting suspension was cooled to room temperature and Et20 (75 mL) was added. The precipitate was collected by filtration, washed with Et20 and dried to produce 10.5 g of an off-white solid. The HCl salt was recrystallized from MeOH (165 mL) to produce 5.98 g (56% overall yield from (R)-tert-butyl 2-(tert-butyldimethylsilyloxy)ethyl(4-cyano-2,3-dihydro-iH-inden-l-yl) carbamate) of (R)-5- (3-(l-(2-hydroxyethylamino)-2,3-dihydro-iH-inden-4-yl)-l,2,4-oxadiazol-5-yl)-2- isopropoxybenzonitrile 85 as a white solid. LCMS-ESI (m/z) calculated for C23H24N403: 404.5; found 405.4 [M+H]+, tR = 2.44 min. Ή NMR (400 MHz, DMSO) 5 9.25 (s, 2H), 8.53 (d, J = 2.3, 1H), 8.42 (dd, J = 9.0, 2.3, 1H), 8.17 (d, J = 7.7, 1H), 7.97 (d, J = 7.6, 1H), 7.63 – 7.50 (m, 2H), 5.28 (t, J = 5.0, 1H), 4.99 (hept, J = 6.1, 1H), 4.92 (s, 1H), 3.72 (q, J = 5.2, 2H), 3.57 – 3.43 (m, 1H), 3.27 (ddd, J = 17.6, 9.1, 5.0, 1H), 3.15-2.85 (m, J = 24.2, 2H), 2.53 (dtd, J = 9.0, 5.5, 5.3, 3.6, 1H), 2.30 (ddd, J = 13.4, 8.9, 4.6, 1H), 1.39 (d, J = 6.0, 6H). 13C NMR (101 MHz, DMSO) 6 173.25, 167.86, 162.47, 144.56, 139.13, 134.53, 133.77, 129.30, 128.93, 127.45, 122.83, 115.79, 115.15, 114.84, 102.40, 72.46, 61.04, 56.51, 46.38, 31.53, 27.74, 21.37. Elemental analysis for C23H25N403C1: C calc. = 62.65%; found = 62.73%; H calc. = 5.71%; found = 5.60%; N calc. = 12.71%; found = 12.64%; CI calc. = 8.04%; found = 8.16%. Chiral HRLC of the free base: (R)-5-(3-(l-(2-hydroxyemylamino)-2,3-dihydro-iH- inden-4-yl)-l,2,4-oxadiazol-5-yl)-2-isopropoxy – benzo-nitrile was eluted using 10% i‘-PrOH in hexanes plus 0.3% DEA: >99.9% ee, tR = 37.72 min.
(S)-5-(3-(l-(2-hydroxyethylamino)- 2,3-dihydro-iH-inden-4-yl)-l,2,4-oxadiazol-5-yl) -2-isopropoxy benzonitrile 86 was obtained in analogous fashion from (S)-tert-butyl 2-(tert-butyldimethylsilyloxy)ethyl(4-(5-(3- cyano-4-isopropoxyphenyl)- 1 ,2,4-oxadiazol-3-yl)-2, 3-dihydro-iH-inden- 1 -yl)carbamate INT-23 and (S)-tert-butyl 4-(5-(3-cyano-4-isopropoxyphenyl)-l,2,4-oxadiazol-3-yl)-2,3- dihydro-iH-inden-l-yl) (2-hydroxyethyl) carbamate INT-24: >99.9% ee, tR for (5)- enantiomer = 35.86 min.
(S) IS DESIRED CONFIGURATION
THE SYNTHESIS IS SUMMARISED BELOW
COSY PREDICT
1H NMR PREDICT
13C NMR PREDICT
note——-(CH3 )2CH-O-AR appears at 72 ppm

////////
















































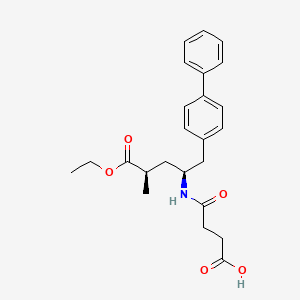




































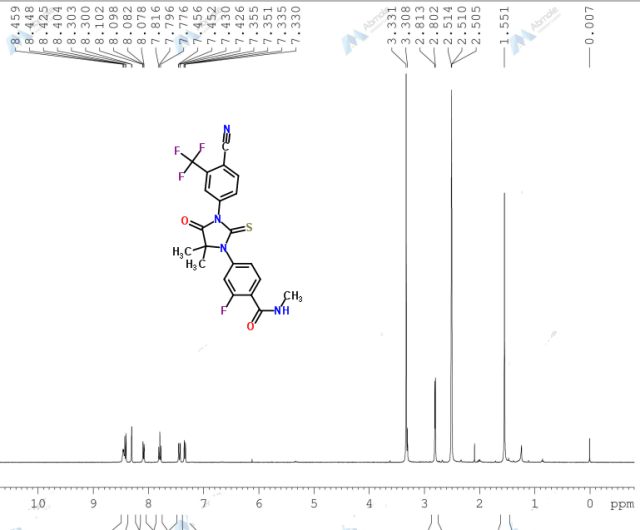
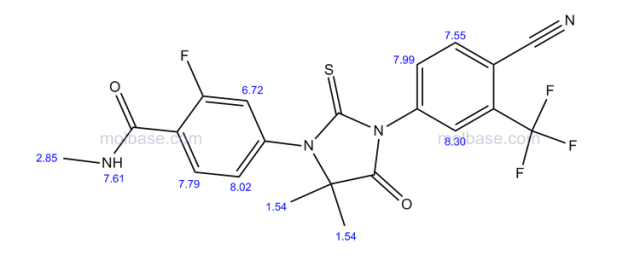


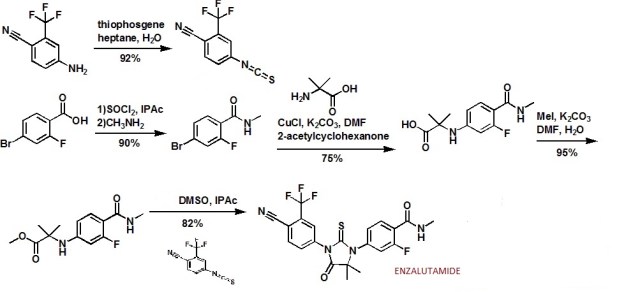


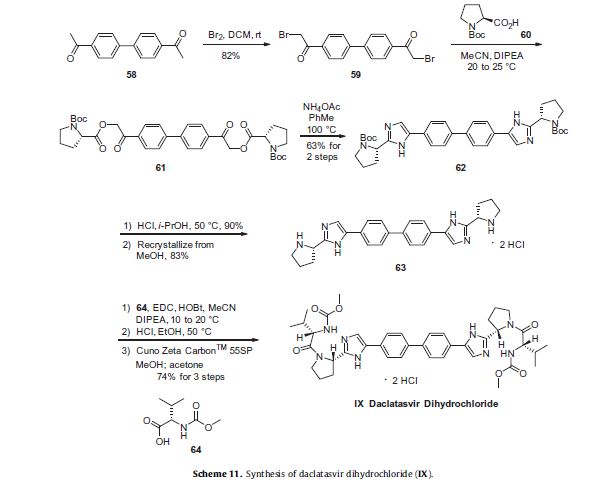


















 Click on images to view
Click on images to view










































