Home » Uncategorized (Page 59)
Category Archives: Uncategorized
SNS-032, BMS-387032 A potent and selective Cdk inhibitor
SNS 032, BMS-387032
N-[5-[[[5-(1,1-Dimethylethyl)-2-oxazolyl]methyl]thio]-2-thiazolyl]-4-piperidinecarboxamide
Cas 345627-80-7, MP 165-167° C
M.Wt:380.53, Formula:C17H24N4O2S2
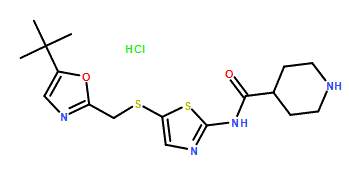
SNS 032, BMS-387032 HYDROCHLORIDE
| Formula | C17H24N4O2S2 . HCl |
|---|---|
| MW | 380.5 . 36.5 |
| CAS | 345627-90-9 |
A potent and selective Cdk inhibitor
Potent inhibitor of cyclin-dependent kinases (cdks) 9, 2 and 7 (IC50 values are 4, 38 and 62 nM respectively). Displays no activity against 190 additional kinases (IC50 >1000 nM). Arrests the cell cycle at G2/M; inhibits transcription, proliferation and colony formation, and induces apoptosis in RPMI-8226 multiple myeloma cells. Prevents tumor cell-induced VEGF secretion and in vitro angiogenesis. SNS-032 (BMS-387032) has firstly been described as a selective inhibitor of CDK2 with IC50 of 48 nM in cell-free assays and is 10- and 20-fold selective over CDK1/CDK4. It is also found to be sensitive to CDK7/9 with IC50 of 62 nM/4 nM, with little effect on CDK6. Phase 1.
Quality Control & MSDS
COA NMR HPLC Datasheet SDS/MSDS
- COA (Certificate Of Analysis)
- HPLC
- NMR (Nuclear Magnetic Resonance)
- MSDS (Material Safety Data Sheet)
SNS-032 (BMS-387032) is a potent and selective inhibitor of cyclin-dependent kinases (CDKs) 2, 7, and 9 [1], with IC50 values of 38 nM, 62 nM and 4 nM, respectively [2].
CDKs mean a family of serine/threonine kinases regulating cell cycle process. Some CDKs are related to transcription control and are often perturbed in cancer cells [3].
Decrease in the phosphorylation at Ser5 and Ser2 in the C-terminal domain (CTD) of RNA Pol II can indicate the inhibition to CDK9 and CDK7 [1]. Chronic lymphocytic leukemia (CLL) cells treated with SNS-032 for 6 or 24 hours showed a decrease in the phosphorylation of Ser2 and Ser5 of the CTD of RNA Pol II, this appeared to be both time- and concentration- dependent, and remarkably consistent among samples. For the phosphorylation of Ser2, the inhibition of SNS-032 was greater than that for the phosphorylation of Ser5, this was consistent with the fact that IC50 for the inhibition of CDK9 was lower compared with that for the inhibition of CDK7 (4 nM vs 62 nM). After 6 hours of SNS-032 exposure, protein levels of CDK7 and CDK9 were stable, but declined at 24 hours [4].
In patients with chronic lymphocytic leukemia (CLL), infusion of SNS-032 in a total dose of 75 mg/m2 resulted in a decrease in the phosphorylation at Ser5 and Ser2 in the C-terminal domain of RNA Pol II. This indicated the inhibition to Cdk9 and Cdk7 by SNS-032. This inhibition was first seen 2 hours after the beginning of the infusion with SNS-032, was pronounced after 6 hours and returned to baseline after 24 hours [1].

The cell cycle-regulated cyclin-dependent kinases (CDKs), CDK1, 2, and 4 have been extensively studied as potential therapeutic targets in cancer. Recent research has additionally underscored the potential role of several constitutively active CDKs including CDK7 and 9 as cancer targets. Phosphorylation of the c-terminal domain (CTD) of RNA Polymerase II by CDK7 and 9 are critical steps in transcriptional regulation. Inhibition of these kinases is predicted to have the greatest effect on the expression of proteins with short t½ and short-lived mRNA, including proteins involved in apoptotic regulation. CDK7 also activates cell-cycle CDKs 1, 2, 4 and 6. SNS-032 (formerly BMS-387032) has previously been described as a selective inhibitor of CDK2 with potent antitumor activity in animal models. Here we show that in addition to inhibition of CDK2, SNS-032 also inhibits CDK7/cyclinH and CDK9/cyclinT at low nanomolar concentrations in biochemical assays. The compound is highly selective for CDK inhibition; in a panel of 208 kinases, only four non-CDK proteins were inhibited by >50% at 1 μM SNS-032. The cellular pharmacology of SNS- 032 mirrors the biochemical data. Cells treated with SNS-032 show a rapid cell cycle arrest and onset of cell death that corresponds with inhibition of multiple substrates of CDK2, 7, and 9. For instance, inhibition of Rb phosphorylation, accumulation of cyclin E protein and cell-cycle arrest at GI and G2 are observed in multiple cell lines in a time and dose-dependent manner, consistent with inhibition of CDK2 and CDK7. Furthermore, SNS-032 inhibits CDK9-mediated phosphorylation of Ser2 in the CTD with an IC50 = 200 nM. Corresponding with inhibition of RNA polymerase II, the short half-life, anti-apoptotic protein Mcl-1 is rapidly depleted from cells, coincident with the phosphorylation of p53. Expression of Mcl-1 is a candidate predictor of aggressive disease and resistance to chemotherapy in CLL and is essential for survival of B-cell lymphoma and multiple myelomas, supporting the use of SNS-032 as a treatment for these diseases. SNS-032, a selective inhibitor of multiple CDKs involved in apoptosis and cell cycle regulation, has potential for antitumor activity in both solid and hematological cancers. SNS-032 is currently in phase 1 clinical studies.
SNS-032, was designed as a selective CDK2 inhibitor. Here, we show that in addition to CDK2, CDK 7 and 9 inhibitory activities also contribute to the biological activity of the molecule. The CDK2/cyclin E complex regulates entry of cells into S phase by phosphorylating Rb, a negative regulator of the transcription factor E2F. CDK2 phosphorylates a number of additional substrates, including cyclin E, signaling its degradation. Inhibiting CDK2 should therefore arrest cells in G1 and stabilize cyclin E. The cellcycle CDKs (CDK1, 2 4 and 6) are activated by phosphorylation by CDK7/cyclin H (also called CAK). Inhibition of CDK7 would therefore also result in cell-cycle arrest at multiple points in the cell cycle due to failure to activate the cell cycle CDKs. CDK 7 and 9 activate transcription by phosphorylating the CTD of RNA pol II. Inhibition of CTD phosphorylation has been shown to inhibit transcription and reduce expression of short lived proteins, including those involved in apoptosis regulation. Stalling of RNA polymerase has also been shown to activate p53, leading to apoptosis. Thus, the CDK7 and 9 inhibitory activities of SNS-032 are expected to cause cytotoxicity via induction of apoptosis.
SNS-032 is a selective CDK inhibitor, preferentially targeting CDK2, CDK7 and CDK9 in vitro. • In cell models, SNS-032 shows dual activity, targeting both cell cycle progression and apoptosis pathway proteins. • SNS-032 Inhibited CDK9 and 7-mediated phosphorylation of ser 2 and ser 5 of the CTD of RNA pol II and in turn downregulates the antiapoptotic protein Mcl-1. • SNS-032 induced a cell cycle arrest, and increased cyclin E levels are consistent with inhibition of cell cycle CDKs • Mcl-1 is a key survival factor in many B-cell malignancies. SNS-032 is being pursed as treatment for these diseases.
| Biological Activity | ||||||
|---|---|---|---|---|---|---|
| Description | SNS-032 is a novel, potent and selective CDK inhibitor of CDK2, CDK7 and CDK9 with IC50 of 38 nM, 62 nM and 4 nM, respectively. | |||||
| Targets | CDK2 | CDK7 | CDK9 | |||
| IC50 | 38 nM | 62 nM | 4 nM [1] | |||
| In Vitro | SNS-032 has low sensitivity to CDK1 and CDK4 with IC50 of 480 nM and 925 nM, respectively. SNS-032 effectively kills chronic lymphocytic leukemia cells in vitro regardless of prognostic indicators and treatment history. Compared with flavopiridol and roscovitine, SNS-032 is more potent, both in inhibition of RNA synthesis and at induction of apoptosis. SNS-032 activity is readily reversible; removal of SNS-032 reactivates RNA polymerase II, which led to resynthesis of Mcl-1 and cell survival. [1] SNS-032 inhibits three dimensional capillary network formations of endothelial cells. SNS-032 completely prevents U87MG cell–mediated capillary formation of HUVECs. In addition, SNS-032 significantly prevents the production of VEGF in both cell lines, SNS-032 prevents in vitro angiogenesis, and this action is attributable to blocking of VEGF. Preclinical studies have shown that SNS-032 induces cell cycle arrest and apoptosis across multiple cell lines. [2] SNS-032 blocks the cell cycle via inhibition of CDKs 2 and 7, and transcription via inhibition of CDKs 7 and 9. SNS-032 activity is unaffected by human serum. [3]SNS-032 induces a dose-dependent increase in annexin V staining and caspase-3 activation. At the molecular level, SNS-032 induces a marked dephosphorylation of serine 2 and 5 of RNA polymerase (RNA Pol) II and inhibits the expression of CDK2 and CDK9 and dephosphorylated CDK7. [4] | |||||
| In Vivo | SNS-032 prevents tumor cell-induced VEGF secretion in a tumor coculture model. [2] SNS-032, a new CDK inhibitor, is more selective and less cytotoxic and has been shown to prolong stable disease in solid tumors. [4] | |||||
| Clinical Trials | SNS-032 currently in phase I clinical trial for chronic lymphocytic leukemia (CLL) and multiple myeloma (MM). | |||||
| Description | SNS-032 is a selective inhibitor of CDK2 with IC50 of 48 nM. | |||||
| Targets | CDK2 | CDK7 | CDK9 | |||
| IC50 | 48 nM | 62 nM | 4 nM | |||
CLIP
http://www.mdpi.com/1420-3049/19/9/14366/htm#B39-molecules-19-14366
SNS032, previously called BMS-387032, has been developed by Sunesis. This compound, which contains a thiazole unit, selectively inhibits CDK2 (IC50: 38 nM), CDK7 (IC50: 62 nM) and CDK9 (IC50: 4 nM) [39]. Preclinical studies demonstrated that SNS032 was able to inhibit cell cycle activity along with transcription [20].
SNS032 is in phase I clinical trials for the treatment of chronic lymphoid leukemia along with multiple myeloma, and the mode of administration is intravenous [39]. The purpose is to evaluate the dose-escalation of SNS-032 along with its safety, pharmacokinetics, pharmacodynamic activity and clinical efficacy. Biomarker analyses demonstrated mechanism-based pharmacodynamic activity with inhibition of CDK7 and CDK9, although limited clinical activity in heavily pretreated patients was observed [39].
Tong, W.G.; Chen, R.; Plunkett, W.; Siegel, D.; Sinha, R.; Harvey, R.D.; Badros, A.Z.; Popplewell, L.; Coutre, S.; Fox, J.A.; et al. Phase I and pharmacologic study of SNS-032, a potent and selective CDK2, 7, and 9 inhibitor, in patients with advanced chronic lymphocytic leukemia and multiple myeloma. ASCO Annual Meeting. J. Clin. Oncol. 2010, 28, 3015–3022.

![Image result for N-(Cycloalkylamino)acyl-2-aminothiazole Inhibitors of Cyclin-Dependent Kinase 2. N-[5-[[[5-(1,1-Dimethylethyl)-2-oxazolyl]methyl]thio]-2-thiazolyl]-4- piperidinecarboxamide (BMS-387032), a Highly Efficacious and Selective Antitumor Agent,](https://journals.prous.com/journals/dof/20083311/html/df330932/images/sch01.gif)
SNS-032 (formerly BMS-387032) is a small-molecule cyclin-dependent kinase (CDK) inhibitor currently in phase I clinical trials for the treatment of B-cell malignancies and advanced solid tumors. Preclinical studies have shown that SNS-032 is a specific and potent inhibitor of CDK2, 7 and 9 which induces cell cycle arrest and apoptosis in tumor cell lines. It was shown to inhibit in vitro angiogenesis and prostaglandin E2 (PGE2) production, both strongly associated with tumorigenesis. Phase I clinical trials support the safety and tolerability of SNS-032 as evaluated in dose-escalation studies. The compound is currently administered by i.v. infusion but has shown promising potential for oral delivery.
![Image result for N-(Cycloalkylamino)acyl-2-aminothiazole Inhibitors of Cyclin-Dependent Kinase 2. N-[5-[[[5-(1,1-Dimethylethyl)-2-oxazolyl]methyl]thio]-2-thiazolyl]-4- piperidinecarboxamide (BMS-387032), a Highly Efficacious and Selective Antitumor Agent,](https://journals.prous.com/journals/dof/20083311/html/df330932/images/sch02.gif)
CLIP
![Image result for N-(Cycloalkylamino)acyl-2-aminothiazole Inhibitors of Cyclin-Dependent Kinase 2. N-[5-[[[5-(1,1-Dimethylethyl)-2-oxazolyl]methyl]thio]-2-thiazolyl]-4- piperidinecarboxamide (BMS-387032), a Highly Efficacious and Selective Antitumor Agent,](https://i0.wp.com/www.rcsb.org/pdb/images/56H_600.gif)
![Image result for N-(Cycloalkylamino)acyl-2-aminothiazole Inhibitors of Cyclin-Dependent Kinase 2. N-[5-[[[5-(1,1-Dimethylethyl)-2-oxazolyl]methyl]thio]-2-thiazolyl]-4- piperidinecarboxamide (BMS-387032), a Highly Efficacious and Selective Antitumor Agent,](https://i0.wp.com/www.mdpi.com/ijms/ijms-14-21805/article_deploy/html/images/ijms-14-21805f2-1024.png)
The structures of representative protein kinases inhibitors based on the aminopyrazole scaffold.http://www.mdpi.com/1422-0067/14/11/21805/htm
CLIP
N-(Cycloalkylamino)acyl-2-aminothiazole Inhibitors of Cyclin-Dependent Kinase 2. N-[5-[[[5-(1,1-Dimethylethyl)-2-oxazolyl]methyl]thio]-2-thiazolyl]-4- piperidinecarboxamide (BMS-387032), a Highly Efficacious and Selective Antitumor Agent,

N-Acyl-2-aminothiazoles with nonaromatic acyl side chains containing a basic amine were found to be potent, selective inhibitors of CDK2/cycE which exhibit antitumor activity in mice. In particular, compound 21 {N-[5-[[[5-(1,1-dimethylethyl)-2-oxazolyl]methyl]thio]-2-thiazolyl]-4-piperidinecarboxamide, BMS-387032}, has been identified as an ATP-competitive and CDK2-selective inhibitor which has been selected to enter Phase 1 human clinical trials as an antitumor agent. In a cell-free enzyme assay, 21 showed a CDK2/cycE IC50 = 48 nM and was 10- and 20-fold selective over CDK1/cycB and CDK4/cycD, respectively. It was also highly selective over a panel of 12 unrelated kinases. Antiproliferative activity was established in an A2780 cellular cytotoxicity assay in which 21 showed an IC50 = 95 nM. Metabolism and pharmacokinetic studies showed that 21 exhibited a plasma half-life of 5−7 h in three species and moderately low protein binding in both mouse (69%) and human (63%) serum. Dosed orally to mouse, rat, and dog, 21showed 100%, 31%, and 28% bioavailability, respectively. As an antitumor agent in mice, 21administered at its maximum-tolerated dose exhibited a clearly superior efficacy profile when compared to flavopiridol in both an ip/ip P388 murine tumor model and in a sc/ip A2780 human ovarian carcinoma xenograft model.
CLIP

http://pubs.rsc.org/en/content/articlehtml/2016/md/c6md90040b
Heat shock factor 1 (HSF1) is a transcription factor that plays key roles in cancer, including providing a mechanism for cell survival under proteotoxic stress. Therefore, inhibition of the HSF1-stress pathway represents an exciting new opportunity in cancer treatment. We employed an unbiased phenotypic screen to discover inhibitors of the HSF1-stress pathway. Using this approach we identified an initial hit (1) based on a 4,6-pyrimidine scaffold (2.00 μM). Optimisation of cellular SAR led to an inhibitor with improved potency (25, 15 nM) in the HSF1 phenotypic assay. The 4,6-pyrimidine 25 was also shown to have high potency against the CDK9 enzyme (3 nM).
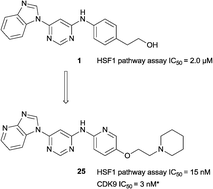
Discovery of 4,6-disubstituted pyrimidines as potent inhibitors of the heat shock factor 1 (HSF1) stress pathway and CDK9
E-mail: Paul.Workman@icr.ac.uk, Keith.Jones@icr.ac.uk
DOI: 10.1039/C6MD00159A
COMPD 25
1H NMR (500 MHz, DMSO-d6) δ 10.38 (s, 1H), 9.21 (s, 1H), 8.74 (d, J = 0.9 Hz, 1H), 8.62 (dd, J = 8.2, 1.5 Hz, 1H), 8.56 (dd, J = 4.7, 1.5 Hz, 1H), 8.16-8.13 (m, 2H), 7.64 (br d, J = 8.6 Hz, 1H), 7.52-7.47 (m, 2H), 4.14 (t, J = 5.9 Hz, 2H), 2.66 (t, J = 5.9 Hz, 2H), 2.47-2.42 (m, 4H), 1.53-1.47 (m, 4H), 1.42 – 1.33 (m, 2H). 13C NMR (126 MHz, DMSO-d6) δ 160.74, 158.32, 156.72, 154.88, 150.74, 146.47, 145.38, 143.74, 134.21, 125.02, 124.16, 122.29, 119.60, 114.32, 94.06, 66.49, 57.35, 54.35, 25.54, 23.88. HRMS (ESI+ ): calcd for C22H25N8O (M + H)+ , 417.2146; found 417.2163.
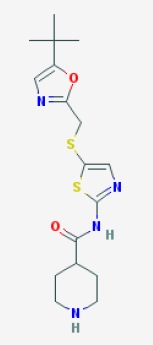
NOTE, THERE IS ERROR IN STRUCTURE ABOVE OF SNS 032
References
References:
[1]. Tong W.G., Chen R., Plunkett W., et al. Phase I and Pharmacologic Study of SNS-032, a Potent and Selective Cdk2, 7, and 9 Inhibitor, in Patients With Advanced Chronic Lymphocytic Leukemia and Multiple Myeloma. Journal of Clinical Oncology, 2010, 28(18):3015- 3022.
[2]. Chipumuro E., Marco E., Christensen C.L., et al. CDK7 Inhibition Suppresses Super-Enhancer-Linked Oncogenic Transcription in MYCN-Driven Cancer. Cell, 2014, 159:1-14.
[3]. Meng H., Jin Y.M., Liu H., et al. SNS-032 inhibits mTORC1/mTORC2 activity in acute myeloid leukemia cells and has synergistic activity with perifosine against Akt. Journal of Hematology & Oncology, 2013, 6:18.
[4]. Chen R., Wierda W.G., Chubb S., et al. Mechanism of action of SNS032, a novel cyclin-dependent kinase inhibitor, in chronic lymphocytic leukemia. Blood, 2009, 113(19):4637-4645.Chen et al (2010) Responses in mantle cell lymphoma cells to SNS-032 depend on the biological context of each cell line. Cancer Res. 70 6587. PMID: 20663900.
Conroy et al (2009) SNS-032 is a potent and selective CDK 2, 7 and 9 inhibitor that drives target modulation in patient samples. Cancer Chemother.Pharmacol. 64 723. PMID: 19169685.
Ali et al (2007) SNS-032 prevents tumor cell-induced angiogenesis by inhibiting vascular endothelial growth factor. Neoplasia 9 370. PMID: 17534442.
Misra et al (2004) N-(Cycloalkylamino)acyl-2-aminothiazole inhibitors of cyclin-dependent kinase 2. N-[5-[[[5-(1,1-Dimethylethyl)-2-oxazolyl]methyl]thio]-2-thiazolyl]-4- piperidinecarboxamide (BMS-387032), a highly efficacious and selective antitumor agent. J.Med.Chem. 47 1719. PMID: 15027863.
Abstract
SNS-032, a CDK inhibitor, exhibited modest to high anti-neuroblastoma activity against a panel of 109 neuroblastoma cell lines in the range of the therapeutic plasma levels reported for SNS-032 through a mechanism involving CDK7 and CDK9 inhibition-mediated down-regulation of XIAP, Mcl-1, BIRC2, cIAP-1 and surviving.
Abstract
The anti-AML mechanism of SNS-032, a cyclin-dependent kinase inhibitor, has been identified though characterizing in vitro effects of SNS-032 alone or in combination with perifosine.
Abstract
Although it induces apoptosis in cancer cells, SNS-032 has no significant effects on normal HSC and HPC in terms of self-renewal inhibition, differentiation suppression and apoptosis induction.
Abstract
The CDK7/9 inhibitor SNS-032-induced down-regulation of FIP1L1-PDGFRα and Bcr-Abl has the potential to be used to decrease the acquired resistant to imatinib.
Abstract
SNS-032, a CDK inhibitor, alone or in combination with Ara-C exhibited potent anti-AML activity, where down-regulation of antiapoptotic genes, cluding BCL2, XIAP amd MCL1, was associated with the synergistic anti-AML effect of the combination treatment.
CC(C)(C)C1=CN=C(O1)CSC2=CN=C(S2)NC(=O)C3CCNCC3
ミチグリニドカルシウム水和物 , Mitiglinide calcium hydrate, 快如妥/Glufast
- MF C19H25NO3
- MW 315.407 Da
Mitiglinide (INN, trade name Glufast) is a drug for the treatment of type 2 diabetes.[1]
Mitiglinide belongs to the meglitinide class of blood glucose-lowering drugs and is currently co-marketed in Japan by Kissei and Takeda. The North America rights to mitiglinide are held by Elixir Pharmaceuticals. Mitiglinide has not yet gained FDA approval.
Mitiglinide calcium hydrate was approved by Pharmaceuticals and Medical Devices Agency of Japan (PMDA) on January 29, 2004. It was co-developed and co-marketed as Glufast® by Takeda and Kissei in Japan.
Mitiglinide is a rapid-acting insulin secretion-stimulating agent. It stimulates insulin secretion by closing the ATP-sensitive K+ (ATP) channels in pancreatic beta-cells. It is indicated for the treatment of type 2 diabetes mellitus.
Glufast® is available as tablet for oral use, containing 5 mg or 10 mg of Mitiglinide calcium hydrate. The recommended dose is 10 mg three times daily just before each meal (within 5 minutes).
China , Approved 2010-04-19, 快如妥/Glufast, Kissei
ミチグリニドカルシウム水和物

C38H48CaN2O6▪2H2O : 704.92
[207844-01-7]
Pharmacology
Mitiglinide is thought to stimulate insulin secretion by closing the ATP-sensitive K(+) K(ATP) channels in pancreatic beta-cells.

Dosage
Mitiglinide is delivered in tablet form.

| Molecular Weight | 333.42 |
| Formula | C19H27NO4 |
| CAS Number | 207844-01-7 |
Mitiglinide calcium hydrate




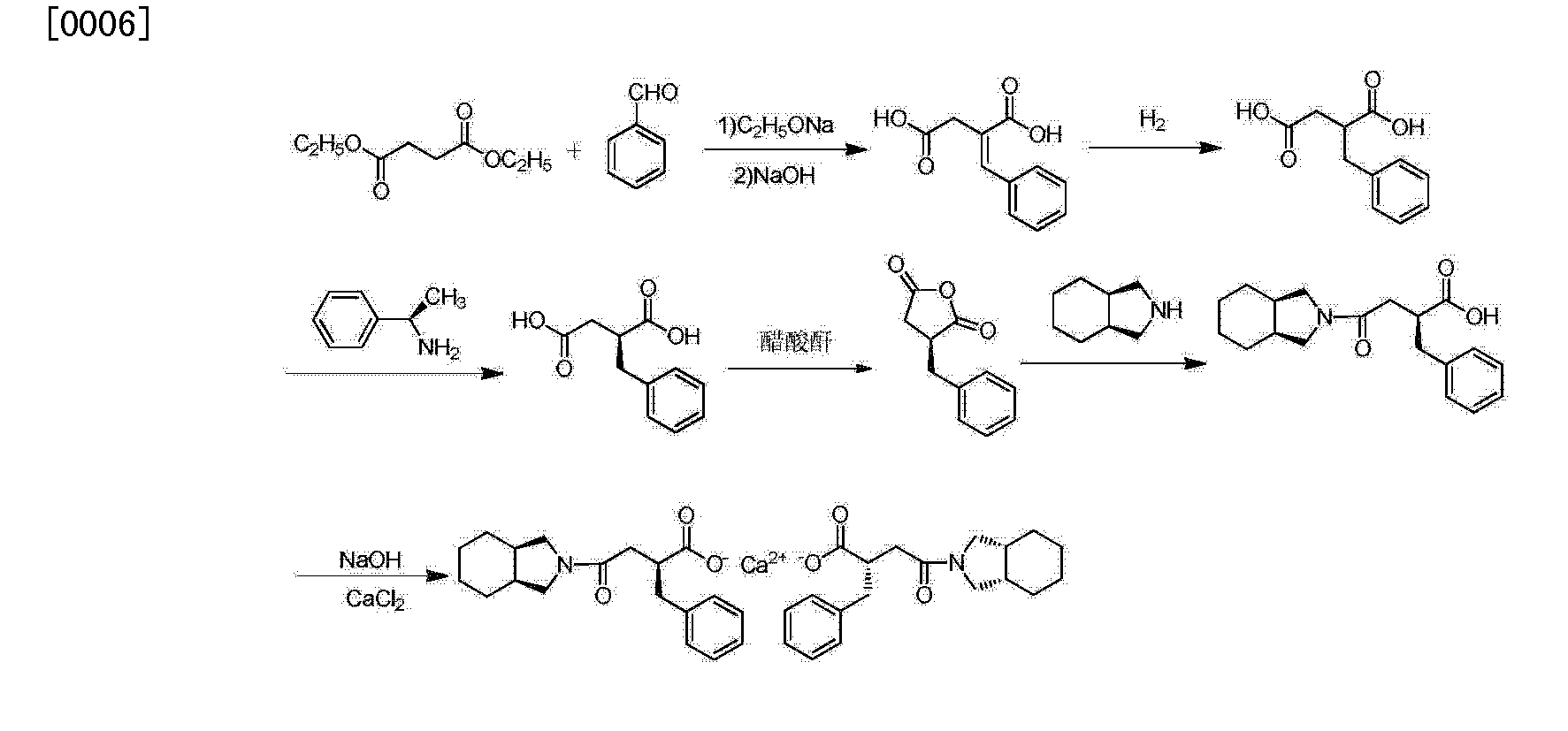
The condensation of dimethyl succinate (I) with benzaldehyde (II) by means of NaOMe in refluxing methanol followed by hydrolysis with NaOH in methanol/water gives 2-benzylidenesuccinic acid (III). Compound (III) is treated with refluxing Ac2O, yielding the corresponding anhydride (IV), which by reaction with cis-perhydroisoindole (V) in toluene affords the monoamide (VI). This amide is reduced with H2 over a chiral Rhodium catalyst and treated with (R)-1-phenylethylamine (VII) to provide the chiral salt (VIII) as a single diastereomer isolated by crystallization. Finally, this salt is treated first with aqueous NH4OH and then with aqueous CaCl2.

he optical resolution of racemic 2-benzylsuccinic acid (XV) using the chiral amines (R)-1-phenylethylamine (VII), (R)-1-(1-naphthyl)ethylamine (XIV) or (S)-1-phenyl-2-(4-tolyl)ethylamine (XVI) is carried out by fractional crystallization of the corresponding diastereomeric salts and treatment with 2N HCl, providing the desired enantiomer 2(S)-benzylsuccinic acid (XVII). Reaction of (XVII) with SOCl2 gives the corresponding acyl chloride (XVIII), which is treated with 4-nitrophenol (XIX) and TEA in dichloromethane to yield the activated diester (XX). The regioselective reaction of (XX) with cis-perhydroisoindole (V) in dichloromethane affords the monoamide (XXI), which by reaction with HCl and methanol provides the corresponding methyl ester (XXII). This ester is hydrolyzed with NaOH to the previously described chiral succinamic acid (XIII), which is finally converted into its calcium salt.

PATENT
https://www.google.com/patents/WO2009047797A2?cl=en
Perhydroisoindole derivative, (S)-mitiglinide of formula I is a potassium channel antagonist for the treatment of type 2 diabetes mellitus and is chemically known as (5)-2-benzyl-3-(cis-hexahydro-2- isoindolinylcarbonyl) propionic acid.
Formula I
It has potent oral hypoglycemic activity and is structurally different from the sulphonylureas, although it stimulates calcium influx by binding to the sulphonylurea receptor on pancreatic β-cells and closing K+ ATP channels. Perhydroisoindole derivatives including (S)-mitiglinide and salts thereof were first disclosed in US patent 5,202,335. This patent discloses preparation of (S)-mitiglinide by the reaction of (5)-3-benzyloxycarbonyl-4-phenylbutyric acid with cis-hexahydroisoindoline in the presence of N- methylmorpholine and isobutyl chloroformate followed by debenzylation with palladium on carbon in ethyl acetate to yield (5)-mitiglinide as viscous oil. (S)-Mitiglinide is isolated as its hemi calcium salt using calcium chloride in water which is further recrystallized with diisopropyl ether. Melting point of calcium salt of mitiglinide calcium dihydrate salt is herein reported as 179-185 0C. (S)-Mitiglinide prepared by the above process is obtained in low yields. Further, the synthetic method described in the patent does not enable the desired regioselectivity. Extensive purification steps are required to obtain the desired compound, which makes the process unattractive from industrial point of view. US patent 6,133,454 discloses a process for the preparation of (S)-mitiglinide by reacting dimethyl succinate with benzaldehyde in methanolic medium, to yield a diacid which is converted to corresponding anhydride and is further reacted with the perhydroisoindole to yield 2-[(cis- perhydroisomdol^-ytycarbonylmethyl^-phenylacrylic acid which is then subjected to catalytic hydrogenation using the complex rhodium/(2S,4S)-N-butoxycarbonyl-4-diphenylphosphino-2-diphenyl- phosphino-methylpyrrolidine (Rh/(S,S) BPPM) as asymmetric hydrogenation catalyst, followed by conversion to pharmaceutically acceptable salt of (S)-mitiglinide. The above patent utilizes ruthenium complex which is expensive, carcinogenic and toxicity, hence not recommended for industrial scale. European patent publication no. EP 0967204 discloses the preparation of mitiglinide by deprotecting benzyl-(S)-2-benzyl-3-(cis-hexahydro-2-isoindolinyl-carbonyl) propionate and converting the same to calcium dihydrate salt in crystalline form using calcium chloride, water and ethanol. The crystals of calcium salt are further recrystallized using ethanol and water. But the patent is silent about the crystalline form of mitiglinide calcium.
It will be appreciated by those skilled in the art that perhydroisoindole derivative, (S)-mitiglinide of formula I contains a chiral centre and therefore exists as enantiomers. Optically active compounds have increasingly gained importance since the technologies to develop optically active compounds in high purity have considerably improved. Obtaining asymmetric molecules has traditionally involved resolving the desired molecule from a racemic mixture using a chiral reagent, which is not profitable as it increases the cost and processing time. Alternatively, desired enantiomer can be obtained by selective recrystallization of one enantiomer. However such a process is considered inefficient, in that product recovery is often low, purity is uncertain and more than 50% of the material is lost. Enantiomers can also be resolved chromatographically, although the large amount of solvent required for conventional batch chromatography is cost prohibitive and results in the preparation of relatively dilute products. Limited throughput volumes also often make batch chromatography impractical for large-scale production. Even so, it is a common experience for those skilled in the art to find chiral separation of certain chiral mixtures to be inefficient or ineffective, thereby resulting in the efforts towards development of newer methodologies for asymmetric synthesis.
It would be of significant advantage to obtain (.S)-mitiglinide by development of reaction conditions necessary for productive manufacture of the required (5)-enantiomer, substantially free of the unwanted (R)-enantiomer, in large quantities that meet acceptable pharmaceutical standards. It is the property of the solid compounds to exist in different polymorphic form. By the term polymorphs mean to include different physical forms, crystal forms, crystalline/liquid crystalline/non-crystalline (amorphous) forms. This has especially become very interesting after observing that many antibiotics, antibacterials, tranquilizers etc, exhibit polymorphism and some/one of the polymorphic forms of a given drug exhibit superior bio-availability and consequently show much higher activity compared to other polymorphs. It has also been disclosed that the amorphous forms in a number of drugs exhibit different dissolution characteristics and in some cases different bioavailability patterns compared to the crystalline form [Konne T., Chem. Pharm. Bull. 38, 2003 (1990)]. The solubility of a material is also influenced by its solid-state properties, and it has been suggested that the solubility of an amorphous compound is 10 to 1600 times higher than that of its most stable crystalline structures (Bruno C. Hancock and Michael Parks, ‘What is the true solubility advantage for amorphous pharmaceuticals’, Pharmaceutical Research 2000, Apr; 17(4):397-404). Thus it can be concluded that amorphous products are in general more soluble and often show improved absorption in humans.
Thus, there is a widely recognized need for developing a stable polymorph, which would further offer advantages over crystalline forms in terms of better dissolution and the availability profiles. Also none of the prior art references disclose amorphous form of mitiglinide calcium. Thus present invention provides amorphous form of mitiglinide calcium.
It is also required that the final API like mitiglinide whether in the amorphous form or crystalline form must be free from the other impurities including the unwanted enantiomer, these can be side product and by product of the reaction, degradation products and starting materials. Impurities in final API are undesirable and in extreme cases, might even be harmful to a patient being treated with a dosage form containing the API. Therefore impurities introduced during commercial manufacturing processes must be limited to very small amounts and are preferably substantially absent. These limits are less than about 0.15 percent by weight of each identified impurity and 0.10 % by weight of unidentified and/or uncharacterized impurities. After the manufacture of APIs, the purity of the products, such as (S)- mitiglinide calcium dihydrate is required before commercialization, and in the manufacture of formulated pharmaceuticals. Therefore, pharmaceutical active compounds must be either free from these impurities or contain the impurities in acceptable limits. There is also a need for the isolation, characterization and identification of the impurities and their use as reference markers and reference standard. Thus, the present invention meets the need in the art for a novel, efficient and industrially advantageous process for providing optically pure perhydroisoindole derivatives, particularly (iS)-mitiglinide, which is unique with respect to its simplicity, scalability and involves controlling the steps of the reaction so that predominantly the desired (S)-enantiomer is produced in high yields and purity. The present invention also provides substantially pure (S)-mitiglinide and salts thereof having novel amide impurity in acceptable limit or free from this impurity.
Example 1: Preparation of (R) 4-benzyl-3-(3-phenylpropionv0-oxazolidin-2-one To a solution of (R)-4-benzyloxazolidin-2-one (50 g), 4-dimethylaminopyridine (4.85 g), 3-phenyl propionic acid (55.08 g) in dichloromethane (375 ml) under nitrogen atmosphere at 0-5 0C, dicyclohexylcarbodiimide (975.65 g) was added. The temperature was slowly raised to 25-30 0C and stirring was continued until no starting material was left as was confirmed by thin layer chromatography. Dicyclohexylurea formed during the reaction was filtered, washed with dichloromethane (200 ml) and the filtrate was washed with saturated solution of sodium bicarbonate (500 ml). The solution was dried over sodium sulphate and solvent was distilled off to obtained crude product which was purified from methanol (200 ml) at 10-15 °C and washed with methanol (50 ml) to obtain 81.0 g of the title compound. Example 2: Preparation of 3(5)-benzyl-4-(4-(J?)-benzyl-2-oxo-oxazolidin-3-yl)-4-oxo-butyrϊc acid tert-butyl ester
To a solution of (/?)-4-benzyl-3-(3-phenyl-propionyl)-oxazolidin-2-one (150 g) in anhydrous tetrahydrofuran (1.5 It) was added a solution of sodium hexamethyldisilazane (462 ml, 36-38% solution in tetrahydrofuran) with stirring at -85 to -95 0C for 60 minutes. Tert-butyl bromo acetate (137.5 g) in tetrahydrofuran (300 ml) was added to reaction mass and then stirred to 60 minutes at -85 to -95 0C. After completion of the reaction (monitored by TLC), the reaction mixture was poured into ammonium chloride solution (10%, 2.0 It) and extracted with ethyl acetate (2×750 ml). The combined organic layer was washed with demineralized water (1×750 ml) and dried over sodium sulphate. The solvent was evaporated under reduced pressure to obtain oily residue which was stirred with mixture of n-hexane (100 ml) and isopropyl alcohol (100 ml) at Oto -50C, filtered and dried under vacuum to obtain 153.12 g of title compound having chemical purity 99.41%, chiral purity 99.91% by HPLC, [α]D 20: (-)97.52° (c = 1, CHCl3) and M.P. : 117.1-118.20C.
Example 3: Preparation of 3(5)-benzyl-4-(4(i?)-benzyl-2-oxo-oxazolidin-3-yl)-4-oxobutyric acid Trifluoroacetic acid (100 g) was added to a solution of 3(5)-benzyl-4-(4-(/?)-benzyl-2-oxo-oxazolidin-3- yl)-4-oxobutyric acid tert-butyl ester (100 g) in dichloromethane (700 ml) at 25 0C and mixture was stirred further for about 12 hours ( when TLC indicated reaction to be complete). The reaction mixture was poured in to ammonium chloride solution (10%, 500 ml). The dichloromethane layer was separated and aqueous layer was extracted with dichloromethane (2 x 250 ml). The combined organic layer was dried over sodium sulphate and evaporated under reduced pressure to obtain title compound. The crude product was recrystallized from a mixture of ethyl acetate: n-hexane (1:4, 500 ml) to obtain 78.75g of the title compound having purity 99.56% by HPLC and M.P.: 145.9-146.40C.
Example 4: Preparation of (2S)-2-benzyl-l-((4R)-4-benzyl-2-oxo-oxazolidin-3-vI)-4-(hexahydro- isoindolin-2-yl)-butane-l,4-dione
To a solution of 3(5)-benzyl-4-(4-(/?)-benzyl-2-oxo-oxazolidin-3-yl)-4-oxo-butyric acid (50 g) in anhydrous dichloromethane (1.25 It) was added triethylamine (50 ml) with stirring at -20 to -30 0C and the stirred for 15 minutes. A solution of isobutylchloroformate (37.50g) in anhydrous dichloromethane (50 ml) was added at -20 to -30 0C and stirred for 60 minutes. Thereafter, a solution of cis- hexahydroisoindoline (32.50 g) in anhydrous dichloromethane (50 ml) was slowly added by maintaining temperature -20 to -300C. After the completion of the reaction (monitored by HPLC), the mixture was successively washed with 0.5N hydrochloric acid solution (500 ml), brine (300 ml) and dried over sodium sulphate. The solvent was evaporated under reduced pressure to obtain 102.0 g of the title compound having purity 94.39% by HPLC.
Example 5: Purification of r2S)-2-benzyl-l-((4R)-4-benzyl-2-oxo-oxazolidin-3-yl)-4-(hexahydro- isoindolin-2-vD-butane-l,4-dione
To the crude (2S)-2-benzyl-l-((4R)-4-benzyl-2-oxo-oxazolidin-3-yl)-4-(hexahydro-isoindolin-2-yl)- butane- 1,4-dione (51.0 g) was added methanol (150 ml) and the mixture was stirred for 5 hours at 0 to 5 0C. Solid that precipitated out was filtered, slurry washed with cold methanol (25 ml) and dried at 45 -50 0C under vacuum to obtain 28.80 g of pure title compound as a crystalline solid having purity of 99.71% by HPLC and M. P.: 104.1-105.70C.
Example 6: Preparation of calcium salt of (-SVmitiglinide. Step-1: Preparation of (-SVmitiglinide
(2S)-2-Benzyl- 1 -((4R)-4-benzyl-2-oxo-oxazolidin-3-yl)-4-(hexahydro-isoindolin-2-yl)-butane- 1 ,4-dione (28.0 g) was dissolved in tetrahydrofuran (196 ml) and a mixture of lithium hydroxide monohydrate (3.51 g) in demineralized water (56 ml) and hydrogen peroxide (40% solution, 5.5 ml) was added with stirring at 0 to 5 0C over a period of 30 minutes. The reaction mixture was further stirred at 0 to 5 0C till the completion of the reaction. After the completion of the reaction (monitored by TLC), the reaction was quenched with the addition of cooled sodium meta-bisulphate solution (25%, 168 ml) at 0 to 10 0C. The reaction mixture was extracted with ethyl acetate (2×112 ml), the layers were separated and the aqueous layer was discarded. The HPLC analysis of the aqueous layer shows 0.77% of amide impurity. The ethyl acetate layer was then extracted with aqueous ammonia solution (4%, 2×40 ml). The layers were separated and the aqueous layer was further extracted with ethyl acetate (2×280 ml). Combined ethyl acetate layer was discarded. This aqueous layer (280 ml) was used as such in the next stage. The aqueous layer display purity 96.19 % by HPLC and amide impurity 0.04% by HPLC. Step-2: Preparation of calcium salt of dSVmitiglinide
To the above stirred solution of (S)-mitiglinide in water and ammonia(280 ml), methanol (168 ml) was added, followed by calcium chloride (4.48 g) dissolved in demineralized water (56 ml) at ambient temperature and the mixture was stirred for 2 hours. The resulting precipitate was filtered, successively slurry washed with water (3 x 140 ml) and acetone (2 x 70 ml) and dried at 450C -500C under vacuum to obtain 16.1 g of title compound having purity 99.67% by HPLC and amide impurity 0.01% by HPLC. The title product was re-precipitated from a mixture of methanol and water and dried to obtain pure title compound.
Example 7: Preparation of (.SVmitiglinide
To a solution of (2S)-2-benzyl-l-((4R)-4-benzyl-2-oxo-oxazolidin-3-yl)-4-(hexahydro-isoindolin-2-yl)- butane- 1,4-dione (50 g) in tetrahydrofuran (350 ml) was added a solution of lithium hydroxide monohydrate (8.65 g) in demineralized water (100 ml) and hydrogen peroxide (30% w/w, 40 ml) with stirring at 5 to 10 0C over a period of 15 minutes. After the completion of reaction, sodium meta- bisulphate solution (40%, 500 ml) was added to the reaction mixture and the mixture was extracted with ethyl acetate (2 x 250 ml). The organic layer was dried over sodium sulphate and evaporated under vacuum to obtain 45.5 g of title compound having 35 % of R-benzyl oxozolidin-2-one as impurity. Example 8: Purification of (.S)-mitiglinide
Aqueous ammonia solution (4%, 300 ml) was added to the crude (5)-mitiglinide (30 g) and stirred. The reaction mixture was washed with ethyl acetate (3 x 300 ml). Thereafter the reaction mixture was acidified to pH 1 to 2 with IN hydrochloric acid solution (250 ml) and extracted with ethyl acetate (2 x 150 ml). The layers were separated and ethyl acetate layer was washed with demineralized water (2 x 150 ml), dried over sodium sulphate and then evaporated under reduced pressure to obtain 16.2 g of pure (5)-mitiglinide having purity 95.55% by HPLC Example 9: Preparation of calcium salt of (S)-mitiglinide
To a solution of (<S)-mitiglinide (15 g) in water (150 ml) and aqueous ammonia solution (25%, 15 ml) at 25 to 30 0C, a solution of calcium chloride (7.5 g) in demineralized water (37.5 ml) was added. The mixture was stirred for 1 hour to precipitate the calcium salt of (5)-mitiglinide dihydrate. The resulting precipitate was filtered, slurry washed with water (3 x 150ml) and dried at 45 to 50 0C to obtain 13.25 g of the title compound having purity of 98.84% by HPLC. Example 10: Purification of calcium salt of (5)-mitiglinide
(iS)-mitiglinide calcium (10 g) was dissolved in dimethylformamide (100 ml). This is followed by the addition of demineralized water (500 ml) at 25 to 30 0C. The mixture was stirred for 30 minutes. The precipitated solid was filtered, washed with water (10x 50ml) and dried at 45 to 50 0C under vacuum to obtain 8g of pure title compound as a crystalline solid having purity of 99.62% by HPLC. Example 11: Preparation of amorphous mitiglinide calcium
Crystalline mitiglinide calcium (2.0 g) was dissolved in tetrahydrofuran (20 ml) and filtered to remove undissolved and suspended particles. The solvent was then evaporated under vacuum to obtain a powder which was then dried under vacuum at 40-600C to obtain 1.70 g of the title compound. Example 12: Preparation of amorphous mitiglinide calcium
Crystalline mitiglinide calcium (2.0 g) was dissolved in dichloromethane (30 ml) and filtered to remove undissolved and suspended particles. The solvent was then evaporated under vacuum to obtain a powder which was then dried under vacuum at 40-600C to obtain 1.64 g of the title compound. Example 13: Preparation of amorphous mitiglinide calcium
Mitiglinide (2.0 g) was dissolved in methanol (20 ml) and methanolic ammonia (5.0 ml) solution was added to it. The solution was stirred at 25-30 0C and calcium chloride (1.5 g) dissolved in methanol was mixed with the solution of mitiglinide and ammonia in methanol and the solution was filtered to remove the suspended particles. The solvent was then evaporated under vacuum to obtain a powder which was then dried under vacuum at 40-600C to obtain 1.9 g of the title compound. Example 14: Preparation of amorphous mitiglinide calcium
Mitiglinide (2.0 g) was dissolved in dichloromethane (20 ml) and aqueous ammonia (3.6 ml, 25 % solution) was added to it. The solution was stirred at 25-300C and solid calcium chloride (1.5 g) was mixed with the solution of mitiglinide and ammonia in dichloromethane and the solution warmed at 30 – 35 0C. The solution was washed with water (2 xlO ml) and the clear solution was dried over sodium sulfate, filtered and evaporated under vacuum and finally dried at under vacuum at 40-60 0C to obtain 1.75 g of the title compound.
Example 15: Preparation of amorphous mitiglinide calcium
Crystalline mitiglinide calcium dihydrate (2.0 g) was dissolved in ethyl acetate (30 ml) and filtered to remove undissolved and suspended particles. Approimately. 60 % of the solvent was distilled off under vacuum to obtain a stirrable solution. The solution was then cooled to 15-2O0C, mixed with n-heptane (20 ml) and the mixture was stirred for 30 minutes. The resulting solid was filtered, washed with n-heptane and dried under vacuum at 45-600C to yield 1.72 g of the title compound. Example 16: Preparation of amorphous mitiglinide calcium
Crystalline mitiglinide calcium (2.Og) was dissolved in dichloromethane (30 ml) and filtered to remove undissolved and suspended particles. Approximately 60 % of the solvent was distilled off under vacuum to obtain a stirrable solution. The solution was then cooled to 15-200C and mixed with diisopropyl ether (20 ml). The mixture was stirred for 30 minutes and the resulting solid was filtered, washed with diisopropyl ether and dried under vacuum at 45-600C to obtain 1.70 g of the title compound. Example 17: Preparation of amorphous mitiglinide calcium
Mitiglinide (2.0 g) was dissolved in dichloromethane (20 ml) and aqueous ammonia (3.6 ml, 25 % solution) solution was added to it. The solution was stirred at 25-30 0C and mixed with solid calcium chloride (1.5 g) and the solution warmed at 30-35 0C and stirred for 30 minutes. The solution was washed with water (2 x 10 ml) and the clear solution was dried over sodium sulfate, and filtered. Approximately 60% of the solvent was distilled off under vacuum and the resulting viscous oil was cooled to 10-15 0C and mixed with diisopropyl ether (50 ml). The reaction mixture was stirred for 30-35 minutes and the resulting solid was filtered and dried at 40-600C to obtain 1.75 g of the title compound. Example 18: Conversion of amorphous mitiglinide calcium into crystalline mitiglinide calcium A suspension of amorphous mitiglinide calcium in diisopropyl ether (30 ml) was stirred for 2 hours at 25- 300C, filtered and dried under vacuum at 45-600C to obtain crystalline form of mitiglinide calcium. Example 19: Preparation of crystalline mitiglinide calcium
To a solution of mitiglinide (2.5 g) in water (2.5 ml), aqueous ammonia solution (approx 25%, 4.0 ml) and acetonitrile (2.5 ml) at 10-150C, calcium chloride (1.32 g) dissolved in demineralized water (15 ml) was added. The mixture was stirred for 2 hours. The resulting precipitate was filtered, slurry washed with water (3 x 25 ml) and acetone (2 x 5 ml) and dried at 45-500C under vacuum to obtain 2.12 g of title compound having purity: 99.72 % by HPLC.
Example 20: Preparation of crystalline mitiglinide calcium
To a solution of mitiglinide (2.5 g) in water (2.5 ml), aqueous ammonia solution (approx 25%, 4.0 ml) and tetrahydrofuran (2.5 ml) at 10-150C, calcium chloride (1.32 g) dissolved in demineralized water (15 ml) was added. The mixture was stirred for 2 hours. The resulting precipitate was filtered, slurry washed with water (3 x 25 ml) and acetone (2 x 5 ml) and dried at 45-500C under vacuum to obtain 1.95 g of title compound having purity: 99.52 % by HPLC.
Example 21; Preparation of crystalline mitiglinide calcium
To a solution of mitiglinide (30.0 g) in water (300 ml), aqueous ammonia solution (approx 25%, 48 ml) and acetone (300 ml) at 10-150C, calcium chloride (15.8 g) dissolved in demineralized water (180 ml) was added. The mixture was stirred for 2 hours. The resulting precipitate was filtered, slurry washed with water (3 x 300 ml) and acetone (2 x 60 ml) and dried at 45-500C under vacuum to obtain 24.32 g of title compound having purity: 99.42 % by HPLC.
Example 22: Preparation of crystalline mitiglinide calcium
To a solution of mitiglinide (3.0 g) in water (30 ml), aqueous ammonia solution (approx 25%, 4.8 ml) and isopropyl alcohol (300 ml) at 10-150C, calcium chloride (1.58 g) dissolved in demineralized water
(18 ml) was added. The mixture was stirred for 2 hours. The resulting precipitate was filtered, slurry washed with water (3 x 30 ml) and acetone (2 x 6 ml) and dried at 45-500C under vacuum to obtain 1.92 g of title compound having purity: 99.65 % by HPLC.
Example 23: Preparation of (2S)-2-benzyWV-((lR)-l-benzyl-2-hydroxy-ethyl)-4-(hexahvdro- isoindolin-2-yl)-4-oxo-buryramide
To a solution of (2S)-2-benzyl-l-((4R)-4-benzyl-2-oxo-oxazolidin-3-yl)-4-(hexahydro-isoindolin-2-yl)- butane-l,4-dione (20.0 g) in tetrahydrofuran (140 ml), a solution of lithium hydroxide monohydrate
(3.43 g,) in demineralized water (40 ml) was added and the reaction mixture was refluxed for 4 hours till the completion of the reactions (monitored by thin layer chromatography). After the completion of the reaction, the reaction mixture was poured into demineralized water (100 ml) and extracted with ethyl acetate (2 x 80 ml). The combined organic layer was washed with water (80 ml) and dried over sodium sulphate. The solvent was evaporated under reduced pressure to give residue which was stirred in isopropyl alcohol at 0-5 0C for 5 hours. The mixture was filtered and then dried at 40-45 0C under vacuum to obtain 12.48 g of title compound having purity 99.77 % by HPLC. Melting point = 77 – 800C.

PAPER
An Effective and Convenient Method for the Preparation of KAD-1229
Helvetica Chimica ActaVolume 87, Issue 8, Version of Record online: 27 AUG 2004

PAPER
asian journal of chemistry asian journal of chemistry
(S)-Mitiglinide calcium dihydrate is designated chemically … Identification, Synthesis and Characterization of Impurities of (S)-Mitiglinide Calcium Dihydrate………http://www.asianjournalofchemistry.co.in/(X(1))/User/ViewFreeArticle.aspx?ArticleID=26_9_51
PATENT
CN 102382033

PATENT
https://www.google.com/patents/CN104311471A?cl=en
Mitiglinide calcium (mitiglinide calcium), the chemical name (2S) -2_ benzyl-3- (cis – hexahydro-2-isoindoline-carbonyl) propionic acid calcium salt dihydrate , for the treatment of type II diabetes. Kissei by Japanese pharmaceutical company research and development, and for the first time on sale in Japan in May 2004. Mitiglinide calcium is the second repaglinide, nateglinide after the first three columns MAG urea drugs, are ATP-dependent potassium channel blocker, is a derivative of phenylalanine, and its mechanism Similar sulfonylureas, but a faster onset of action and short half-life, is conducive to reducing postprandial blood glucose in diabetic patients, and avoid continuous glucose-induced low blood sugar, with the “in vitro pancreas” reputation.
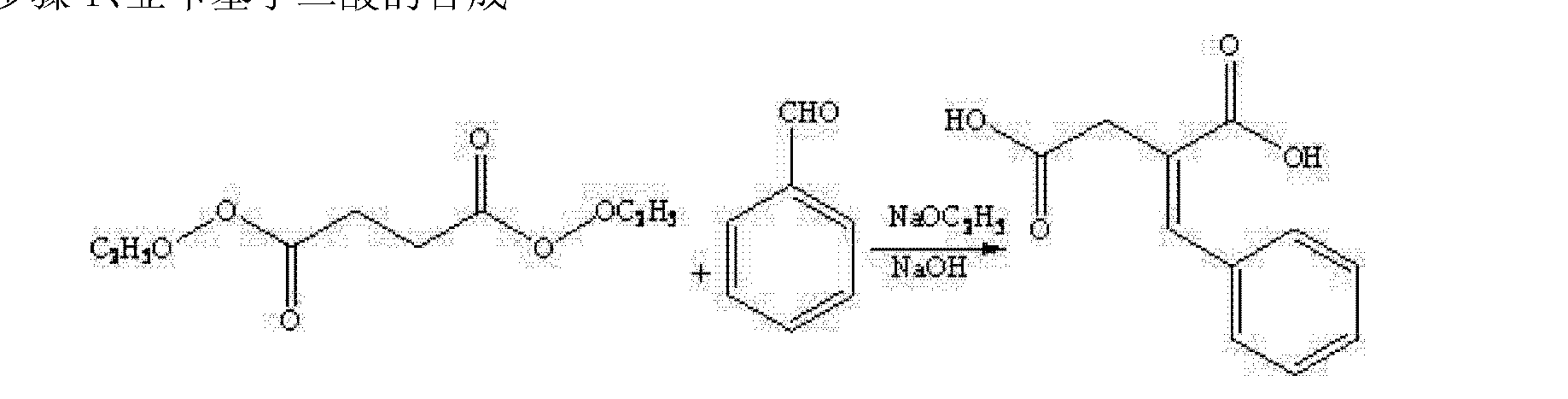
郑德强 etc. on “Food and Drug” magazine was first disclosed the synthesis of calcium Mitiglinide, this method dimethyl succinate and benzaldehyde for raw materials, Stobble condensation, hydrolysis, dehydration anhydride, cis – perhydro isoindole reduced to give racemic acid after condensation, and then split, and salt get Mitiglinide calcium. Specific synthetic route the following equation. The method is relatively complex, in the preparation process to generate half of the unwanted enantiomer, which will waste a lot of cis – perhydro isoindole, and in the preparation of cis – to use science as a whole hydride hydrogen isoindole time reducing agent, the operation is more complicated, the cost is relatively high, and the chiral amine as a resolving agent split, the yield is low.
The patent discloses a CN201010573666 diethyl succinate and benzaldehyde, condensation occurs Stobble sodium ethoxide in ethanol and then hydrolyzed benzylidene succinic acid, succinic acid benzylidene get by catalytic hydrogenation DL-2-benzyl succinic acid, DL-2-benzyl succinic acid by (R) – a chiral amine resolving to give (S) -2- benzyl succinic acid, (S) -2- benzyl succinic acid anhydride to generate its role in the acetic anhydride, and the resulting acid anhydride and cis – hexahydro isoindole reaction of Mitiglinide acid, calcium chloride and ammonia most 后米格列奈 acid reacts with calcium Mitiglinide dihydrate. The synthesis route following formula. This method effectively avoids the expensive intermediate cis – perhydro isoindole waste, reduce costs, but still amounted to a six-step synthesis route much so that the reagent type, long cycle, low yield, and direct use in the synthesis process Sodium block protonated reagent preparation sodium methylate, generate a lot of flammable hydrogen gas, limiting the industrial application of the method.
The present invention solves is to overcome the existing routes that exist in step lengthy reagent variety, low yield, long cycle, high cost, not suitable for industrial production shortcomings. The present invention provides the following formula preparation process route mitiglinide calcium, organic solvent for this preparation method uses less synthesis process is simple, high yield, good purity, suitable for industrial production.
An improved Mitiglinide calcium industrialized preparation method comprises the following steps: Step 1: Preparation of 2-benzylidene succinic acid; 2 steps: (S) prepared _2_ section succinic acid; Step 3: 2- (S) – section group _4_ oxo – (cis – perhydro isoindol-2-yl) butyric acid; Step 4: Preparation Mitiglinide calcium. Characterized in that: in step 1, using commercially available reagents protonated organic bases, protonation process using an organic alkali solution was slowly feeding methods. Step 2 chiral asymmetric reduction. Step 3 fails anhydride using direct selective amidation. Step 4 beating impurities using an aqueous solvent, prepared mitiglinide calcium dihydrate purification method.
The preparation step 1, using a commercially available organic bases as sodium methoxide or sodium ethoxide protonation agent. As optimization program, feeding method using sodium methoxide or sodium ethoxide solution formulated as the corresponding alcohol and the corresponding dialkyl succinate protonating a nucleophilic substitution reaction.
The preparation method described in Step 2, the use of Ru with BINAP homogeneous catalyst Ru (OAc) 2 [(S) -BINAP] as a chiral asymmetric synthesis of chiral reducing reagent.
The steps of the preparation method 3, using ethyl acetate as a reaction solvent, acid binding agent triethylamine do, imidazole and thionyl chloride selective amidation reagent, for cis – perhydro isoindole conduct Selective condensation title intermediate.
The step of preparing said 4, mitiglinide calcium crude product was slurried in 95% ethanol by suction, after simple preparation of high purity mitiglinide calcium dihydrate.
More specifically, the industrialized Mitiglinide calcium preparation, the following steps: Step 1: Preparation of succinate 2_ Benzylidene
Sodium methoxide (sodium ethoxide) was dissolved in methanol (ethanol), was added dropwise to dimethyl succinate (ethyl) ester, was heated at reflux for 30min, benzaldehyde was added dropwise under reflux, stirring at reflux completed the dropwise 3~5h, drops adding an aqueous solution of 4N NaOH dropwise Bi refluxed 4~6h, cooled to room temperature, adjusted with 6N HCl San PH 2, a solid precipitated, centrifuged, and dried to give the title intermediate 1. Step 2: Preparation of (S) -2- acid, benzyl butyl
Intermediate 1, methanol, and Ru (OAc) 2 [(S) -BINAP] into the reactor, the reactor with N2 the replacement air after heating to 50 ° C, a hydrogen pressure through 10h, cooled, filtered, The filtrate was concentrated to dryness to give the title intermediate 2. Step 3: 2- (S) – benzyl-4-oxo – (cis – perhydro isoindol-2-yl) butyric acid
Ethyl acetate was added to the reactor, triethylamine, imidazole and Intermediate 2, was stirred and cooled to -15~-5 ° C, was added dropwise thionyl chloride addition was complete, the -15 ° C~_5 ° C Under continued stirring 6h, a solution of cis – perhydro isoindole, drip completed, stirred at room temperature overnight, the reaction mixture was added IN hydrochloric acid, stirred Ih, separation, and the organic layer was washed with sodium hydroxide solution to extract IN The combined aqueous layer was washed with a small amount of ethyl acetate, the aqueous layer was adjusted with IN hydrochloric acid and the PH = 3, the aqueous layer was extracted with ethyl acetate, the organic layers combined, washed with water and saturated brine, and the organic layer was dried over anhydrous Na2SO4, filtered and the filtrate concentrated under reduced pressure to obtain the objective compound 3 billion Step 4: Preparation of calcium Mitiglinide
The 3 was dissolved in ethanol, was added 2N sodium hydroxide solution, after mixing the solution was added dropwise a 10% aqueous solution of calcium chloride, the reaction mixture was stirred vigorously 3~5h, ice-cooled, filtered, the filter cake with 95% ethanol beating crystallization, filtration, and dried in vacuo to give the title compound I.
Accordingly, the present invention is a method for preparing mitiglinide calcium has the following advantages:
1, Step 1, using commercially available sodium methylate (sodium ethanol) instead of sodium block as a proton agent, effectively avoid the risk of sodium block formed during the reaction a lot of flammable hydrogen gas, industrial production safer. Another use dropping protonated reagent feeding method can effectively avoid succinic acid alkyl ester of two methylene groups are protonated and reduce the incidence of side effects, so that the yield increased by nearly 20%.
2, Step 2, the selective reduction of chiral reagent (S) -BINAP instead of the original route after the first split reduction method, not only simplifies the reaction step, but low yield while avoiding split It leads to the risk of an increase in cost.
3, Step 3, the fixed selective amidation reaction conditions instead of the original first into anhydride after amidation reaction that simplifies the reaction steps to reduce the unit operations, shortening the production cycle, improve production efficiency.
4, Step 4, by using an aqueous solution of calcium Mitiglinide ethanol refining crude beating, then dried under reduced pressure to control the moisture content and reduce the difficulty of the operation, more conducive to industrial production.DETAILED DESCRIPTION The following examples further illustrate the invention, but the present invention is not limited thereto. Example One Step I: Preparation 2_ benzylidene succinic acid Sodium methoxide (9kg) and methanol (48L) into the 100L reactor, stirring to dissolve, into the high slot 50L. The dimethyl succinate (20kg) into the 200L reaction vessel, heated to reflux, methanol was added dropwise a solution of fast high tank of sodium methoxide, refluxed for reaction completion dropwise 30min, was added dropwise under reflux benzaldehyde (10. 9kg) dropwise with stirring at reflux completed 3~5h, HPLC detection benzaldehyde completion of the reaction, a solution of aqueous 4N NaOH (38L), Bi dropwise refluxed 4~6h, cooled to room temperature, 2, adjusted with 6N HCl and the precipitated solid was San PH, centrifugation, and dried in vacuo to give a pale yellow solid 19kg, i.e. an intermediate, yield 90%. Step 2: Preparation of (S) -2- butyric acid benzyl 200L detecting a high pressure hydrogenation reactor airtight, Intermediate I (19kg), methanol (95L) containing 5% Ru (0Ac) 2 [(S ) -BINAP] molecular sieve (SBA-15) supported catalyst (0. 95kg, homemade) into the reactor, purge the inside of the reactor with N2 atmosphere, followed by heating to 50 ° C, atmospheric pressure hydrogen-10h, cooled, filtered and the filtrate was concentrated to dryness under reduced pressure, the resulting solid was recrystallized from ethyl acetate and dried in vacuo to give an off-white solid 15. 5kg, i.e. intermediate 2, yield 81%, chiral purity 90. 5% θ. θ .. Step 3: 2- (S) – benzyl-4-oxo – (cis – perhydro isoindol-2-yl) butyric acid in 500L reaction vessel was charged with ethyl acetate (225L), triethylamine (1.8kg), imidazole (9. 8kg) and Intermediate 2 (15kg), stirred and cooled to -KTC, was added dropwise thionyl chloride (17. 2kg), the addition was complete, the -KTC~-5 ° C under Stirring was continued for 6h, a solution of cis – perhydro isoindole (9kg), drip completed, the reaction was stirred at room temperature for 18h, the reaction mixture was added IN HCl (150L) was stirred Ih, liquid separation, the organic layer was washed with IN sodium hydroxide solution (100LX3) extracted aqueous layers were combined, washed with ethyl acetate (50L) with, water layer was washed with IN of hydrochloric acid adjusted to PH = 3, the aqueous layer was extracted with ethyl acetate (IOOmLX 3), the combined organic layers , saturated brine (50LX 3) was washed, and the organic layer was dried over anhydrous Na2SO4, filtered, and the filtrate was concentrated under reduced pressure to give an oil 19. 8kg, i.e. Intermediate 3 Yield: 87%. Step 4: Preparation of mitiglinide calcium Intermediate 3 (. 19 8kg) and absolute ethanol (99L) into the 200L reactor, and stirred to dissolve, was added 2N sodium hydroxide solution (35L), minutes after mixing Batch into the high slot. The 500L reaction vessel was added 5% aqueous calcium chloride solution (155L), stirring was added dropwise a solution of the high slot, dropwise with vigorous stirring the reaction completion 3~5h, centrifuged, the cake was washed with 95% ethanol (99L) was recrystallized beating, centrifugation and dried in vacuo (50 ° C / 0. 09MPa), to give the title compound I 16. lkg, yield 73%.
PATENT
https://www.google.com/patents/CN102424664A?cl=en
Mitiglinide calcium Phenylalanine belong chiral compound synthesis routes according to different methods of constructing chiral center has the following three synthetic process:
① split method 😦 Document: CN 102101838A, CN 1844096, etc.)
In this method, diethyl succinate and benzaldehyde by Mobbe condensation, hydrolysis, dehydration anhydride, and after cis-hydrogenated isoindole condensation is reduced to give racemic acid, and then split, and salt to give Mitiglinide calcium. The first method step condensation reaction impurities, product separation and purification difficult, finally resolving the yield is low. This method is also a lack atom economy.
② asymmetric hydrogenation 😦 Document tetrahedron Letters, 1987,28 (17), 1905-1908; Tetrahedron Letters, 1989,30 (6), 735-738)
[0027] This method requires expensive rhodium complexes (Rh, (2S, 4Q-N_-butoxycarbonyl-4-diphenylphosphino _2_ diphenylphosphino-2-diphenylphosphino methylpyrrolidine alkyl), making the production cost is greatly improved, and the need for high-pressure hydrogenation reaction, is not conducive to industrial production.
③ chiral method 😦 Document: CN 1680321A)

The method uses phenylalanine as chiral starting materials, after diazotization, nucleophilic substitution, high temperature decarboxylation and condensation reaction product. Wherein the decarboxylation temperature is too low yield, making the overall process costs.
DISCLOSURE
The object of the present invention is to provide a simple, effective and easy-to-operate preparation Mitiglinide calcium.
The present invention provides a process for the preparation of calcium Mitiglinide, the synthesis route is as follows:
Step 1: D- phenylalanine in the acid hydrolysis of formula (¾ 2- hydroxy acid;
Step 2: formula (¾ 2- hydroxy acid under basic conditions to give protected hydroxyl sulfonate of formula (¾-hydroxyphenyl propionic acid ester;
Step 3: The formula (¾-hydroxyphenyl propionic acid ester in the acid-catalyzed carboxyl ester-protected formula (4) phenylalanine methyl sulfonate carboxylate;
Step 4: cis-hydrogen isoindole synthesis formula (6) perhydro isoindole halide;
Step 5: Under alkaline conditions, the formula ⑷ formula (6) nucleophilic substitution reaction formula (5) Mitiglinide acid
Step 6: Under alkaline conditions, the formula (¾ Mitiglinide ester hydrolysis to the calcium salt of formula (1) Mitiglinide calcium.
Preferably, the specific steps include:
Step 1: (D) – phenylalanine hydrolysis in a strong acid of formula (2) 2-hydroxyphenyl propionic acid
In (D) – phenylalanine as a starting material, in the presence of a strong acid such as sulfuric acid, _5 ° C _5 ° C hydrolysis, to give Formula (2) 2-hydroxyphenyl propionic acid White solid.
Step 2: The formula (¾ 2- hydroxy acid under basic conditions to protect the hydroxyl group sulfonic acid ester of formula (¾-hydroxyphenyl propionic acid ester
2-hydroxyphenyl propionic acid in an organic base such as triethylamine or pyridine, or an inorganic base such as sodium bicarbonate, sodium carbonate or potassium carbonate effect, p-hydroxybenzoic acid ester protecting performed, the protecting group used is an aliphatic or aromatic sulfonic acid group such as mesylate, tosylate or p-toluenesulfonic acid group, a sulfonic acid group is preferably methyl group or p-toluenesulfonic acid.
Step 3: Protect formula formula (¾-hydroxyphenyl propionic acid ester in the acid-catalyzed carboxyl ester group (4) benzenepropanoic
MitigIinide1 (I) carboxylic acid ester sulfonate
In the catalytic acid carboxyl benzenepropanoic acid ester group protection, the use of alcohol may be fatty alcohols or aromatic alcohols, preferably ethanol, t-butanol or benzyl alcohol.
Step 4: cis-hydrogen isoindole synthesis formula (6) perhydro isoindole halide
In the synthesis of perhydro isoindole halide in the haloacetyl halide can be used chloroacetyl chloride, bromoacetyl chloride or bromoacetyl bromide, chloroacetyl chloride is preferred.
Step 5: Under alkaline conditions, (4) and (6) a nucleophilic substitution reaction formula (¾ Mitiglinide acid
Under the conditions of a strong base, such as sodium alkoxide such as sodium ethoxide or sodium methylate, perhydro isoindole halide and phenylalanine sulfonate nucleophilic substitution reaction Mitiglinide ethyl reaction temperature of -10 ° C -25 ° c, preferably 0 ° C.
Step 6: Under alkaline conditions, the formula (¾ Mitiglinide ester hydrolysis to the calcium salt of formula (1) calcium Mitiglinide
Ethyl mitiglinide under basic conditions such as sodium hydroxide, potassium hydroxide, or an amine (ammonia) in the presence of an aqueous solution of calcium chloride, and hydrolyzed as calcium salt, in aqueous solution under conditions of heavy alcohol crystallization, high purity mitiglinide calcium.
The present invention and the prior art comparison, has the following advantages:
1, to find an innovative high-yield process for preparing calcium Mitiglinide route, a total yield of 47%;
2, with respect to the routing methods reported in the literature, the optical yield doubled, ee greater than 99%;
3. The process route of the raw materials are cheap, readily available, avoiding costly chiral resolving agents or the use of a catalyst;
4. The process route mild conditions, high temperature decarboxylation overcome the harsh reaction conditions.
In the present invention, (D) – phenylalanine as a starting material, after diazotization, a hydroxyl group and a carboxyl group protected, nucleophilic substitution, hydrolysis and other reactions prepared mitiglinide calcium, high yield. The present invention provides a process used by a wide range of raw materials, low prices, the total yield of 47%, optical purity greater than 99%, and mild reaction conditions, the reaction process is simple, avoid the literature, such as split, high-pressure hydrogenation method low yield, long reaction steps and other shortcomings, but also to overcome the harsh conditions of high temperature reaction deacidification, etc. for preparation and production of calcium Mitiglinide provides a new choice.
The process route mild conditions, high temperature decarboxylation overcome the harsh reaction conditions.
In the present invention, (D) – phenylalanine as a starting material, after diazotization, a hydroxyl group and a carboxyl group protected, nucleophilic substitution, hydrolysis and other reactions prepared mitiglinide calcium, high yield. The present invention provides a process used by a wide range of raw materials, low prices, the total yield of 47%, optical purity greater than 99%, and mild reaction conditions, the reaction process is simple, avoid the literature, such as split, high-pressure hydrogenation method low yield, long reaction steps and other shortcomings for Mitiglinide calcium preparation and production of a new choice.
Preferably, in the above embodiment, each step may be the following alternative, the embodiment can achieve the same advantageous effects to a third embodiment of embodiment:
Step 1: (D) – phenylalanine in the acid hydrolysis of formula (¾ 2- hydroxy acid
In (D) – phenylalanine as a starting material, in the presence of sulfuric acid, -50C _5 ° C hydrolysis, to give Formula O) 2-hydroxyphenyl propionic acid White solid.
Step 2: formula (¾ 2- hydroxy acid under basic conditions to give protected hydroxyl sulfonate of formula C3) hydroxyphenyl propionic acid ester
2-hydroxyphenyl propionic acid in an organic base such as triethylamine or pyridine, or an inorganic base such as sodium bicarbonate, sodium carbonate or potassium carbonate effect, p-hydroxybenzoic acid ester protecting performed, the protecting group used is an aliphatic or aromatic sulfonic acid group such as mesylate, tosylate or p-toluenesulfonic acid group, a sulfonic acid group is preferably methyl group or p-toluenesulfonic acid.
Step 3: Formula C3) hydroxyphenyl propionic acid ester in the acid-catalyzed carboxyl ester-protected formula (4) phenylalanine methyl sulfonate carboxylate [0118] In the acid-catalyzed, styrene-acrylic acid ester-protected carboxy, the use of alcohol may be fatty alcohols or aromatic alcohols, preferably ethanol, t-butanol or benzyl alcohol.
Step 4: cis-hydrogen isoindole synthesis formula (6) perhydro isoindole halide
In the synthesis of perhydro isoindole halide in the haloacetyl halide can be used chloroacetyl chloride, bromoacetyl chloride or bromoacetyl bromide, chloroacetyl chloride is preferred.
Step 5: Under alkaline conditions, the formula ⑷ formula (6) nucleophilic substitution reaction formula (5) Mitiglinide acid
Under the conditions of a strong base, such as sodium alkoxide such as sodium ethoxide or sodium methylate, perhydro isoindole halide and phenylalanine sulfonate nucleophilic substitution reaction Mitiglinide ethyl reaction temperature of -10 ° C -25 ° c, preferably 0 ° C.
Step 6: Under alkaline conditions, the formula (¾ Mitiglinide ester hydrolysis to the calcium salt of formula (1) calcium Mitiglinide
Ethyl mitiglinide under basic conditions such as sodium hydroxide, potassium hydroxide, or an amine (ammonia) in the presence of an aqueous solution of calcium chloride, and hydrolyzed as calcium salt, in aqueous solution under conditions of heavy alcohol crystallization, high purity mitiglinide calcium.

Patent
https://www.google.com/patents/CN103724253A?cl=en
bis [(2s) -2- benzyl-3- (cis – hexahydro isoindole-2-carbonyl) propionic acid] monocalcium dihydrate (mitiglinide calcium), the formula C38H48CaN206.2Η20 English called Mitiglinide Calcium Hydrate, structural formula (I) as
Mitiglinide Calcium is synthesized by Japan Orange Health Pharmaceutical Co., Ltd., in April 2004 in Japan, for through diet and exercise therapy can effectively control high blood sugar in type II diabetes patients.Mitiglinide calcium is the second repaglinide, nateglinide third after the United States and Glenn urea drugs belong phenylalanine derivatives. By closing ΑΤΡ Mitiglinide calcium-dependent pancreatic β cell membrane Κ channel, resulting in the Ca flow, increase intracellular Ca concentration of extracellular vesicles containing threshing leaving insulin, thereby stimulating the secretion of insulin.And only when the meal will be rapid and transient stimulates the pancreas to secrete insulin, sulphonylureas with the traditional Compared to the rapid onset and short duration of action, inhibition of postprandial hyperglycemia characteristic of type II diabetes, to avoid low blood sugar react, early first- and mild diabetes treatment, and well tolerated.
According to the literature and patent reports, prepared Mitiglinide calcium are the following methods.
Method I: 2_ (S) _ benzyl succinic acid as raw material, amides, reduction, calcium salt formation Mitiglinide this method, although fewer steps, but the chiral compound materials, expensive , the production cost is high, not suitable for industrial production. References: Sorbera LA, Leeson PA, Castaner RM, et al.Mitiglinidecalcium (KAD-1229) [J] .Drugs Future, 2000,25 (10):. 1034-1042 [0007] Method Two: succinate methyl ester with benzaldehyde for raw materials, Stobble condensation, hydrolysis, dehydration anhydride, cis – perhydro isoindole after condensation is reduced to give racemic acid, and then split into calcium salts and the like have Mitiglinide. This method is relatively complex and condensation reaction impurities, product separation and purification difficult, costly, and chiral separation time yield is low.[Reference: Zheng Dejiang, Liu Wentao, Wu Lihua synthetic calcium Mitiglinide [J] Food and Drug, 2007,9 (11): 13-15]
Method three: dimethyl succinate and benzaldehyde for raw materials, Stobbe condensation, reduction, split, with p-nitrophenol and dicyclohexyl carbodiimide activated calcium salt formation Mitiglinide This production cost is relatively high, and used column chromatography, suitable for industrial production. References: Synthesis Technology Zhang Hongmei Chen meritorious, Cao Xiaohui Mitiglinide of [J], modern chemicals, 2008,28 (8): 56-59.]
Example 1:
The cis – hexahydro-isoquinoline (250.4g, 2mol), anhydrous potassium carbonate (304.0g, 2.2mol), methylene burn (1000ml) was added to the reaction flask, keeping the temperature 0-5 ° C with vigorous stirring, dropwise acetyl chloride (271.0g, 2.4mol) in dichloromethane (500ml) solution, drip completed, room temperature 2.5h, point board monitoring, reaction complete, additional water 1000ml, organic layer was separated, water (1000ml), saturated brine (1000ml), dried over anhydrous sodium sulfate overnight, dichloromethane was distilled off under reduced pressure to give cis -N- chloroacetyl hexahydro isoindole (2) 357.4g oil close Rate: 88.6%.
The cis -N- chloroacetyl hexahydro isoindole (302.5g, 1.5mol), N_ within phenylpropionyl camphor sulfonamide (573.0g, 1.65mol), 70% sodium hydride (56.6g, 1.65 mol), Ν, Ν- dimethylformamide (900ml) was added to the reaction flask, at 50 ° C, the reaction was stirred vigorously 12h, to give the alkylated product, placed to room temperature before use.
100ml of water was slowly dropped to the above-mentioned system, drip complete, lithium hydroxide (39.5g, 1.65mol), tetrahydrofuran (600ml), at 0-5 ° C under a 30% solution of hydrogen peroxide solution 680ml, drop Albert, was transferred to the reaction was continued at room temperature for 18h, point board monitoring, reaction complete, additional water 1200ml, adjusting the pH to about 2_3, extracted with dichloromethane (900ml X 3), the combined organic phases with saturated brine (1500ml) wash, overnight over anhydrous sodium sulfate, the solvent was distilled off under reduced pressure to give a viscous liquid, to which was added ethyl acetate 250ml, stirred at room temperature, suction filtered, the filter cake with ethyl acetate (150ml) and dried to give (2s) – 2-benzyl-3- (cis – hexahydro isoindole-2-carbonyl) – propionic acid (6) as a white solid 231.8g, two steps yield: 49%. Compound 6 (230g, 0.73mol), water 1150ml, added to the reaction flask. After the whole solution, was added 2mol / L sodium hydroxide solution, 400ml, stirred at rt for 30min, was slowly added dropwise with vigorous stirring chloride (162.0g, 1.46mol) in water (320ml) solution dropwise was completed, the reaction was continued for 1.5h, filtration, water (200ml X 2) washing the filter cake to give a white solid, 60 ° C and dried under reduced pressure to 3h, the filter cake with 95% ethanol (2300ml) recrystallized Mitiglinide calcium (I) 430g, yield: 83.6%, mp: 178 ~ 183 ° C, FAB-MS: m / z316 [M + l] +; [α] D20 = + 5.45 ° (C = 1, methanol) [Document: m.ρ.: 179 ~ 185Ό, [α] d20 = + 5.64 ° (C = L 0, methanol)]; purity: 99.8% [HPLC normalization method : Column C18, mobile phase L OOmol / L potassium dihydrogen phosphate buffered saline – acetonitrile-water (20:35: 30) (adjusted pH = 2.10); detection wavelength 210nm]; iH-NMlUCDCldOOM), δ: 1.1 ~ 1.5 (16Η, m), 1.8 ~ 2.4 (6Η, m), 2.5 ~ 3.1 (14Η, m) 3.3 ~
3.8 (6H, m) 7.4 ~ 7.6 (10H, m); Elemental analysis (%):. C64.68, Η7.35, Ν3.94, Theory: C64.75, Η7.44, Ν3.97 yield : 36.05%, a purity of 99.8%.

PAPER
WEI HUANG,等: “Novel Convenient Synthesis of Mitiglinide“, 《SYNTHETIC COMMUNICATIONS》, vol. 37, no. 13, 3 July 2007 (2007-07-03), pages 2153 – 2157, XP055079498, DOI: doi:10.1080/00397910701392590
http://www.tandfonline.com/doi/abs/10.1080/00397910701392590
Abstract: A novel convenient synthesis of the hypoglycemic agent mitiglinide was developed. (2S)-4-[(3aR,7aS)-Octahydro-2H-isoindol-2-yl]-4-oxo-2-benzyl-butanoic acid (6) was prepared by selective hydrolysis of ethyl 4-[(3aR,7aS)-octahydro-2Hisoindol-2-yl]-4-oxo-2-benzyl-butanoate (5) using a-chymotrypsin; the latter was prepared by a novel facile route from (3aR,7aS)-octahydro-2H-isoindole. The overall yield was 25.6%.
Keywords: a-chymotrypsin, mitiglinide, synthesis
Mitiglinide (calcium bis[(2S)-4-[(3aR,7aS)-octahydro-2H–isoindol-2-yl]-4oxo-2-benzylbutanoate]dihydrate) is a novel oral hypoglycemic agent. It inhibits the adenosine triphosphate (ATP)-sensitive potassium channels in pancreatic b-cells and stimulates insulin release like sulfonylureas,[1] but has a rapid onset and short-lasting hypoglycemic effect as compared with the latter.
Mitiglinide has been synthesized by several related methods that involve optical resolution,[2] asymmetric synthesis,[2a,3] and diasteroselective alkylation using chiral auxiliary.[4]
In a previous article,[2] two optical resolution methods of the key compound racemic acid 4 were reported. One of them involves esterification with optically active alcohols, which are separated into the diastereomers by column chromatogeaphy and hydrolyzed. Only the diastereomeric (S)-Nbenzyl mandelamide ester could be separated; the overall yield was 28%,
The alternative method was optical resolution by optically active bases. The best result was 30.8% yield and 97% ee when using (R)-1-(1-naphthyl)-ethylamine as a base. In this article, we have developed a new optical resolution method of racemic ester 5 by a-chymotrypsin in 45.3% yield; the optical purity of (S)-acid (6) determined by chiral-phase high performance liquid chromatography (HPLC) on Sumichiral
OA3300 was 99.2% ee, and, the method can be used for scale-up preparation.
The synthesis of free acid 6 is shown in Scheme 1. (3aR,7aS)-Octahydro2H-isoindole was chloroacetylated in the presence of Et3N to afford (3aR, 7aS)-2-(chloro-acetyl)-octahydro-2H-isoindole (2), which was condensed with diethyl benzylmalonate followed by hydrolysis and decarbonylation to obtain 4-[(3aR,7aS)-octahydro-2H-isoindol-2-yl]-4-oxo-2-benzyl-butanoic acid (4). The overall yield of the three-step synthesis was 62.9%. The racemic acid (4) was esterified with SOCl2/EtOH to give the corresponding racemic ester (5). The (R)-ester was selectively hydrolyzed by a-chymotrypsin to separate out the (S)-ester, which was subjected to hydrolysis, giving 6.
The overall yield was 28.5% [based on (3aR,7aS)-octahydro-2H-isoindole].
Compound 6 was treated with calcium chloride and 25% ammonium hydroxide to give mitiglinide; after recrystallization from 95% EtOH, the pure product was obtained in 90% yield.
Patent
https://www.google.com/patents/WO2009047797A2?cl=en
EXAMPLES
Example 1: Preparation of (R) 4-benzyl-3-(3-phenylpropionv0-oxazolidin-2-one To a solution of (R)-4-benzyloxazolidin-2-one (50 g), 4-dimethylaminopyridine (4.85 g), 3-phenyl propionic acid (55.08 g) in dichloromethane (375 ml) under nitrogen atmosphere at 0-5 0C, dicyclohexylcarbodiimide (975.65 g) was added. The temperature was slowly raised to 25-30 0C and stirring was continued until no starting material was left as was confirmed by thin layer chromatography. Dicyclohexylurea formed during the reaction was filtered, washed with dichloromethane (200 ml) and the filtrate was washed with saturated solution of sodium bicarbonate (500 ml). The solution was dried over sodium sulphate and solvent was distilled off to obtained crude product which was purified from methanol (200 ml) at 10-15 °C and washed with methanol (50 ml) to obtain 81.0 g of the title compound. Example 2: Preparation of 3(5)-benzyl-4-(4-(J?)-benzyl-2-oxo-oxazolidin-3-yl)-4-oxo-butyrϊc acid tert-butyl ester
To a solution of (/?)-4-benzyl-3-(3-phenyl-propionyl)-oxazolidin-2-one (150 g) in anhydrous tetrahydrofuran (1.5 It) was added a solution of sodium hexamethyldisilazane (462 ml, 36-38% solution in tetrahydrofuran) with stirring at -85 to -95 0C for 60 minutes. Tert-butyl bromo acetate (137.5 g) in tetrahydrofuran (300 ml) was added to reaction mass and then stirred to 60 minutes at -85 to -95 0C. After completion of the reaction (monitored by TLC), the reaction mixture was poured into ammonium chloride solution (10%, 2.0 It) and extracted with ethyl acetate (2×750 ml). The combined organic layer was washed with demineralized water (1×750 ml) and dried over sodium sulphate. The solvent was evaporated under reduced pressure to obtain oily residue which was stirred with mixture of n-hexane (100 ml) and isopropyl alcohol (100 ml) at Oto -50C, filtered and dried under vacuum to obtain 153.12 g of title compound having chemical purity 99.41%, chiral purity 99.91% by HPLC, [α]D 20: (-)97.52° (c = 1, CHCl3) and M.P. : 117.1-118.20C.
Example 3: Preparation of 3(5)-benzyl-4-(4(i?)-benzyl-2-oxo-oxazolidin-3-yl)-4-oxobutyric acid Trifluoroacetic acid (100 g) was added to a solution of 3(5)-benzyl-4-(4-(/?)-benzyl-2-oxo-oxazolidin-3- yl)-4-oxobutyric acid tert-butyl ester (100 g) in dichloromethane (700 ml) at 25 0C and mixture was stirred further for about 12 hours ( when TLC indicated reaction to be complete). The reaction mixture was poured in to ammonium chloride solution (10%, 500 ml). The dichloromethane layer was separated and aqueous layer was extracted with dichloromethane (2 x 250 ml). The combined organic layer was dried over sodium sulphate and evaporated under reduced pressure to obtain title compound. The crude product was recrystallized from a mixture of ethyl acetate: n-hexane (1:4, 500 ml) to obtain 78.75g of the title compound having purity 99.56% by HPLC and M.P.: 145.9-146.40C.
Example 4: Preparation of (2S)-2-benzyl-l-((4R)-4-benzyl-2-oxo-oxazolidin-3-vI)-4-(hexahydro- isoindolin-2-yl)-butane-l,4-dione
To a solution of 3(5)-benzyl-4-(4-(/?)-benzyl-2-oxo-oxazolidin-3-yl)-4-oxo-butyric acid (50 g) in anhydrous dichloromethane (1.25 It) was added triethylamine (50 ml) with stirring at -20 to -30 0C and the stirred for 15 minutes. A solution of isobutylchloroformate (37.50g) in anhydrous dichloromethane (50 ml) was added at -20 to -30 0C and stirred for 60 minutes. Thereafter, a solution of cis- hexahydroisoindoline (32.50 g) in anhydrous dichloromethane (50 ml) was slowly added by maintaining temperature -20 to -300C. After the completion of the reaction (monitored by HPLC), the mixture was successively washed with 0.5N hydrochloric acid solution (500 ml), brine (300 ml) and dried over sodium sulphate. The solvent was evaporated under reduced pressure to obtain 102.0 g of the title compound having purity 94.39% by HPLC.
Example 5: Purification of r2S)-2-benzyl-l-((4R)-4-benzyl-2-oxo-oxazolidin-3-yl)-4-(hexahydro- isoindolin-2-vD-butane-l,4-dione
To the crude (2S)-2-benzyl-l-((4R)-4-benzyl-2-oxo-oxazolidin-3-yl)-4-(hexahydro-isoindolin-2-yl)- butane- 1,4-dione (51.0 g) was added methanol (150 ml) and the mixture was stirred for 5 hours at 0 to 5 0C. Solid that precipitated out was filtered, slurry washed with cold methanol (25 ml) and dried at 45 -50 0C under vacuum to obtain 28.80 g of pure title compound as a crystalline solid having purity of 99.71% by HPLC and M. P.: 104.1-105.70C.
Example 6: Preparation of calcium salt of (-SVmitiglinide. Step-1: Preparation of (-SVmitiglinide
(2S)-2-Benzyl- 1 -((4R)-4-benzyl-2-oxo-oxazolidin-3-yl)-4-(hexahydro-isoindolin-2-yl)-butane- 1 ,4-dione (28.0 g) was dissolved in tetrahydrofuran (196 ml) and a mixture of lithium hydroxide monohydrate (3.51 g) in demineralized water (56 ml) and hydrogen peroxide (40% solution, 5.5 ml) was added with stirring at 0 to 5 0C over a period of 30 minutes. The reaction mixture was further stirred at 0 to 5 0C till the completion of the reaction. After the completion of the reaction (monitored by TLC), the reaction was quenched with the addition of cooled sodium meta-bisulphate solution (25%, 168 ml) at 0 to 10 0C. The reaction mixture was extracted with ethyl acetate (2×112 ml), the layers were separated and the aqueous layer was discarded. The HPLC analysis of the aqueous layer shows 0.77% of amide impurity. The ethyl acetate layer was then extracted with aqueous ammonia solution (4%, 2×40 ml). The layers were separated and the aqueous layer was further extracted with ethyl acetate (2×280 ml). Combined ethyl acetate layer was discarded. This aqueous layer (280 ml) was used as such in the next stage. The aqueous layer display purity 96.19 % by HPLC and amide impurity 0.04% by HPLC. Step-2: Preparation of calcium salt of dSVmitiglinide
To the above stirred solution of (S)-mitiglinide in water and ammonia(280 ml), methanol (168 ml) was added, followed by calcium chloride (4.48 g) dissolved in demineralized water (56 ml) at ambient temperature and the mixture was stirred for 2 hours. The resulting precipitate was filtered, successively slurry washed with water (3 x 140 ml) and acetone (2 x 70 ml) and dried at 450C -500C under vacuum to obtain 16.1 g of title compound having purity 99.67% by HPLC and amide impurity 0.01% by HPLC. The title product was re-precipitated from a mixture of methanol and water and dried to obtain pure title compound.
Example 7: Preparation of (.SVmitiglinide
To a solution of (2S)-2-benzyl-l-((4R)-4-benzyl-2-oxo-oxazolidin-3-yl)-4-(hexahydro-isoindolin-2-yl)- butane- 1,4-dione (50 g) in tetrahydrofuran (350 ml) was added a solution of lithium hydroxide monohydrate (8.65 g) in demineralized water (100 ml) and hydrogen peroxide (30% w/w, 40 ml) with stirring at 5 to 10 0C over a period of 15 minutes. After the completion of reaction, sodium meta- bisulphate solution (40%, 500 ml) was added to the reaction mixture and the mixture was extracted with ethyl acetate (2 x 250 ml). The organic layer was dried over sodium sulphate and evaporated under vacuum to obtain 45.5 g of title compound having 35 % of R-benzyl oxozolidin-2-one as impurity. Example 8: Purification of (.S)-mitiglinide
Aqueous ammonia solution (4%, 300 ml) was added to the crude (5)-mitiglinide (30 g) and stirred. The reaction mixture was washed with ethyl acetate (3 x 300 ml). Thereafter the reaction mixture was acidified to pH 1 to 2 with IN hydrochloric acid solution (250 ml) and extracted with ethyl acetate (2 x 150 ml). The layers were separated and ethyl acetate layer was washed with demineralized water (2 x 150 ml), dried over sodium sulphate and then evaporated under reduced pressure to obtain 16.2 g of pure (5)-mitiglinide having purity 95.55% by HPLC Example 9: Preparation of calcium salt of (S)-mitiglinide
To a solution of (<S)-mitiglinide (15 g) in water (150 ml) and aqueous ammonia solution (25%, 15 ml) at 25 to 30 0C, a solution of calcium chloride (7.5 g) in demineralized water (37.5 ml) was added. The mixture was stirred for 1 hour to precipitate the calcium salt of (5)-mitiglinide dihydrate. The resulting precipitate was filtered, slurry washed with water (3 x 150ml) and dried at 45 to 50 0C to obtain 13.25 g of the title compound having purity of 98.84% by HPLC. Example 10: Purification of calcium salt of (5)-mitiglinide
(iS)-mitiglinide calcium (10 g) was dissolved in dimethylformamide (100 ml). This is followed by the addition of demineralized water (500 ml) at 25 to 30 0C. The mixture was stirred for 30 minutes. The precipitated solid was filtered, washed with water (10x 50ml) and dried at 45 to 50 0C under vacuum to obtain 8g of pure title compound as a crystalline solid having purity of 99.62% by HPLC. Example 11: Preparation of amorphous mitiglinide calcium
Crystalline mitiglinide calcium (2.0 g) was dissolved in tetrahydrofuran (20 ml) and filtered to remove undissolved and suspended particles. The solvent was then evaporated under vacuum to obtain a powder which was then dried under vacuum at 40-600C to obtain 1.70 g of the title compound. Example 12: Preparation of amorphous mitiglinide calcium
Crystalline mitiglinide calcium (2.0 g) was dissolved in dichloromethane (30 ml) and filtered to remove undissolved and suspended particles. The solvent was then evaporated under vacuum to obtain a powder which was then dried under vacuum at 40-600C to obtain 1.64 g of the title compound. Example 13: Preparation of amorphous mitiglinide calcium
Mitiglinide (2.0 g) was dissolved in methanol (20 ml) and methanolic ammonia (5.0 ml) solution was added to it. The solution was stirred at 25-30 0C and calcium chloride (1.5 g) dissolved in methanol was mixed with the solution of mitiglinide and ammonia in methanol and the solution was filtered to remove the suspended particles. The solvent was then evaporated under vacuum to obtain a powder which was then dried under vacuum at 40-600C to obtain 1.9 g of the title compound. Example 14: Preparation of amorphous mitiglinide calcium
Mitiglinide (2.0 g) was dissolved in dichloromethane (20 ml) and aqueous ammonia (3.6 ml, 25 % solution) was added to it. The solution was stirred at 25-300C and solid calcium chloride (1.5 g) was mixed with the solution of mitiglinide and ammonia in dichloromethane and the solution warmed at 30 – 35 0C. The solution was washed with water (2 xlO ml) and the clear solution was dried over sodium sulfate, filtered and evaporated under vacuum and finally dried at under vacuum at 40-60 0C to obtain 1.75 g of the title compound.
Example 15: Preparation of amorphous mitiglinide calcium
Crystalline mitiglinide calcium dihydrate (2.0 g) was dissolved in ethyl acetate (30 ml) and filtered to remove undissolved and suspended particles. Approimately. 60 % of the solvent was distilled off under vacuum to obtain a stirrable solution. The solution was then cooled to 15-2O0C, mixed with n-heptane (20 ml) and the mixture was stirred for 30 minutes. The resulting solid was filtered, washed with n-heptane and dried under vacuum at 45-600C to yield 1.72 g of the title compound. Example 16: Preparation of amorphous mitiglinide calcium
Crystalline mitiglinide calcium (2.Og) was dissolved in dichloromethane (30 ml) and filtered to remove undissolved and suspended particles. Approximately 60 % of the solvent was distilled off under vacuum to obtain a stirrable solution. The solution was then cooled to 15-200C and mixed with diisopropyl ether (20 ml). The mixture was stirred for 30 minutes and the resulting solid was filtered, washed with diisopropyl ether and dried under vacuum at 45-600C to obtain 1.70 g of the title compound. Example 17: Preparation of amorphous mitiglinide calcium
Mitiglinide (2.0 g) was dissolved in dichloromethane (20 ml) and aqueous ammonia (3.6 ml, 25 % solution) solution was added to it. The solution was stirred at 25-30 0C and mixed with solid calcium chloride (1.5 g) and the solution warmed at 30-35 0C and stirred for 30 minutes. The solution was washed with water (2 x 10 ml) and the clear solution was dried over sodium sulfate, and filtered. Approximately 60% of the solvent was distilled off under vacuum and the resulting viscous oil was cooled to 10-15 0C and mixed with diisopropyl ether (50 ml). The reaction mixture was stirred for 30-35 minutes and the resulting solid was filtered and dried at 40-600C to obtain 1.75 g of the title compound. Example 18: Conversion of amorphous mitiglinide calcium into crystalline mitiglinide calcium A suspension of amorphous mitiglinide calcium in diisopropyl ether (30 ml) was stirred for 2 hours at 25- 300C, filtered and dried under vacuum at 45-600C to obtain crystalline form of mitiglinide calcium. Example 19: Preparation of crystalline mitiglinide calcium
To a solution of mitiglinide (2.5 g) in water (2.5 ml), aqueous ammonia solution (approx 25%, 4.0 ml) and acetonitrile (2.5 ml) at 10-150C, calcium chloride (1.32 g) dissolved in demineralized water (15 ml) was added. The mixture was stirred for 2 hours. The resulting precipitate was filtered, slurry washed with water (3 x 25 ml) and acetone (2 x 5 ml) and dried at 45-500C under vacuum to obtain 2.12 g of title compound having purity: 99.72 % by HPLC.
Example 20: Preparation of crystalline mitiglinide calcium
To a solution of mitiglinide (2.5 g) in water (2.5 ml), aqueous ammonia solution (approx 25%, 4.0 ml) and tetrahydrofuran (2.5 ml) at 10-150C, calcium chloride (1.32 g) dissolved in demineralized water (15 ml) was added. The mixture was stirred for 2 hours. The resulting precipitate was filtered, slurry washed with water (3 x 25 ml) and acetone (2 x 5 ml) and dried at 45-500C under vacuum to obtain 1.95 g of title compound having purity: 99.52 % by HPLC.
Example 21; Preparation of crystalline mitiglinide calcium
To a solution of mitiglinide (30.0 g) in water (300 ml), aqueous ammonia solution (approx 25%, 48 ml) and acetone (300 ml) at 10-150C, calcium chloride (15.8 g) dissolved in demineralized water (180 ml) was added. The mixture was stirred for 2 hours. The resulting precipitate was filtered, slurry washed with water (3 x 300 ml) and acetone (2 x 60 ml) and dried at 45-500C under vacuum to obtain 24.32 g of title compound having purity: 99.42 % by HPLC.
Example 22: Preparation of crystalline mitiglinide calcium
To a solution of mitiglinide (3.0 g) in water (30 ml), aqueous ammonia solution (approx 25%, 4.8 ml) and isopropyl alcohol (300 ml) at 10-150C, calcium chloride (1.58 g) dissolved in demineralized water
(18 ml) was added. The mixture was stirred for 2 hours. The resulting precipitate was filtered, slurry washed with water (3 x 30 ml) and acetone (2 x 6 ml) and dried at 45-500C under vacuum to obtain 1.92 g of title compound having purity: 99.65 % by HPLC.
Example 23: Preparation of (2S)-2-benzyWV-((lR)-l-benzyl-2-hydroxy-ethyl)-4-(hexahvdro- isoindolin-2-yl)-4-oxo-buryramide
To a solution of (2S)-2-benzyl-l-((4R)-4-benzyl-2-oxo-oxazolidin-3-yl)-4-(hexahydro-isoindolin-2-yl)- butane-l,4-dione (20.0 g) in tetrahydrofuran (140 ml), a solution of lithium hydroxide monohydrate
(3.43 g,) in demineralized water (40 ml) was added and the reaction mixture was refluxed for 4 hours till the completion of the reactions (monitored by thin layer chromatography). After the completion of the reaction, the reaction mixture was poured into demineralized water (100 ml) and extracted with ethyl acetate (2 x 80 ml). The combined organic layer was washed with water (80 ml) and dried over sodium sulphate. The solvent was evaporated under reduced pressure to give residue which was stirred in isopropyl alcohol at 0-5 0C for 5 hours. The mixture was filtered and then dried at 40-45 0C under vacuum to obtain 12.48 g of title compound having purity 99.77 % by HPLC. Melting point = 77 – 800C.
PATENT
https://www.google.com/patents/CN102101838A?cl=en
Mitiglinide calcium (mitiglinide calcium), the chemical name (2S) _2_ benzyl _3_ (cis – hexahydro _2_ isoindolinyl-carbonyl) propionate dihydrate by Japanese pharmaceutical company developed Kissei ATP-dependent potassium channel blockers, 2004 for the first time in Japan for the treatment of type II diabetes.
Mitiglinide calcium is the second repaglinide, nateglinide after the first three columns MAG urea drugs, is a derivative of phenylalanine, which acts like mechanism sulfonylurea, but faster onset and the short half-life, is conducive to reducing postprandial blood glucose in diabetic patients, but also to avoid low blood sugar caused by continuous glucose, with “in vitro pancreas” reputation.
In recent years, synthetic methods as described in patent application number: Patent 200510200127 9, the synthesis process first synthesized racemic (±) 2_-benzyl-3- (cis – hexahydro iso-indole-2. carbonyl) propionic acid, and then split to give (2S) -2- benzyl-3- (cis – hexahydro isoindole-2-carbonyl) propionic acid, not a lot of waste material along _ hexahydro isoindole, and Chiral separation is not high.
DISCLOSURE
The technical problem to be solved by the present invention is to provide a material savings along _ hexahydro isoindole, and preparation of a high degree of chiral separation.
To solve the above technical problem, the technical solution of the present invention is employed as a method for preparing mitiglinide calcium, comprising the steps of:
Step 1 Synthesis, benzylidene succinic acid
With stirring, was added sodium metal in absolute ethanol, under an inert gas, the solution was heated to reflux with stirring, reflux for 45 fly 0 minutes, under reflux before the dropwise addition of benzaldehyde, and then added dropwise diethyl succinate esters, reaction stirring was continued for 2 to 3 hours, slowly reducing the LC-Ms detection, the ratio of formaldehyde starting material benzene, cooled to room temperature, after use 5 (T55wt% aqueous solution of NaOH to adjust the PH San 13.0, and then heated at reflux;. Γ4 hours, cooled to at room temperature, keeping the reaction solution temperature <25 ° C, pH adjusted with concentrated hydrochloric San 2.0, filtration, recrystallization cold tetrahydrofuran, wherein the molar ratio of sodium metal with benzaldehyde and diethyl succinate is: 0.3 … ~ 0 5: 1 2~1 5: 1; Step 2 synthesis, benzyl butyl acid
The benzylidene succinic acid into the reactor, 10% Pd / C and ethanol, evacuated, and then replaced with hydrogen three times, introducing hydrogen, atmospheric hydrogenation reaction 12~15 hours, the reaction solution suction After the filtrate was evaporated to dryness under reduced pressure, the resulting solid was recrystallized from ethyl acetate, wherein the mass ratio of benzylidene succinic acid with 10% Pd / C is 1: 0 0 15 ^ 20.
3 Synthesis [0006] step, (S) -2- acid, benzyl butyl
Benzyl succinic acid dissolved in methanol was added dropwise with stirring (R) – a chiral amine, stirred at room temperature 2 hours wide, and the precipitated solid was filtered and the solid dispersed in water, under stirring 6 mol / mL hydrochloric acid adjusted ρΗ = 1 (Γ2.0, stirred for 30 minutes, the solid by suction filtration, dried, and wherein the benzyl succinic acid (R) – chiral amine molar ratio of 1: 0~2 5 2;…
Said (R) – a chiral amine (R) -I- phenylethylamine, (R) -I- naphthylethylamine or (R) -I- phenyl-2-p-amine;
4 (S) synthesis step, -2-benzyl succinic anhydride
Reactor, has added (S) -2- benzyl succinic acid and acetic anhydride, at 7 5,0 ° C reaction 1 to 2 hours, isopropyl ether low temperature crystallization after cooling, heavy with ethyl acetate crystallization, wherein (S) -2- molar ratio of benzyl succinic acid and acetic anhydride: 1: 7 · 0 to 7 · 5;
Step 5, (2S) -2- benzyl-3- (cis – hexahydro isoindole-2-carbonyl) propionic acid Synthesis
Stirring, S- benzyl succinic anhydride is dissolved in dichloromethane, control the internal temperature <0 V, a solution of cis _ hexahydro isoindole, dropping it, keeping the internal temperature at <0! : Continue stirring for 2 to 3 hours, the reaction in 2 (T25 ° C 10~15 hours, concentrated to give a pale yellow viscous material, wherein the (S) -2- benzyl succinic anhydride and cis – hexahydro isoindole molar ratio of 1: 2 (Γ2 5; step 6, mitiglinide calcium synthesis.
To the reactor was added (2S) -2- benzyl-3- (cis – hexahydro isoindole-2-carbonyl) propionic acid, water and concentrated ammonia, stir until completely dissolved, a solution of anhydrous calcium chloride aqueous solution, gradually precipitated white solid was added dropwise and then stirred at room temperature 12~ after 15 hours, suction filtered, the filter cake washed with water, dried to give a white solid, i.e. Mitiglinide calcium crude, obtained crude product with methanol and water (volume Than 0.5 4~0 5: 1) and recrystallized as a white solid Mitiglinide calcium;
Beneficial effects: The invention provides a method for preparing calcium Mitiglinide not only saves raw material cis – hexahydro isoindole, and chiral separation is high.
Embodiment 1
Step 1 Synthesis, benzylidene succinic acid
Under stirring, sodium metal (1.7 g, 0. 072 mol) was added absolute ethanol (50 mL), and under argon, the solution was heated to reflux with stirring, reflux for 50 minutes under reflux before the dropwise addition of benzene Formaldehyde (23 mL, 0. 183 mol), and then added dropwise diethyl succinate (50 mL, 0. 275 mol), stirring was continued for 2.5 hours the reaction, reducing the slow LC-Ms detection, the ratio of formaldehyde starting material benzene , was cooled to room temperature, with 55wt.% aqueous NaOH solution adjusting pH ≥ 13.0, and then heated at reflux for 3 hours, cooled to room temperature, the reaction solution temperature maintained <25 ° C, with concentrated hydrochloric pH≤2.0, leaching, cryogenic tetrahydrofuran recrystallization, yield = 81.3%;
Step 2 synthesis, benzyl butyl acid
The benzylidene succinic acid (23. 7 g, 0. 114 mol) into the reactor, then add 10% Pd / C (4. 7g) and anhydrous ethanol (300 mL), evacuated, then Hydrogen replacement three times, introducing hydrogen, hydrogenated at atmospheric pressure for 14 hours, the reaction solution after filtration, evaporated to dryness under reduced pressure, the resulting solid was recrystallized from ethyl acetate, yield: 98% 9; step 3, (S). -2-butyric acid benzyl
Benzyl succinic acid (31. 2 g, 0. 156 mol) was dissolved in methanol (500 mL), and added dropwise with stirring (R) -I- phenylethylamine (41.2 g, 0. 343 mol), room temperature stirred for 1.5 hours, the precipitated solid was filtered, the solid dispersion to water (100 mL) and stirred at with 6 mol / mL hydrochloric acid adjusted ρΗ = 1. (Γ2. 0, stirred for 30 minutes, the solid was suction filtered, and dried Yield 87. 3%; 4 (S) synthesis step, -2-benzyl succinic anhydride
Reactor, has added (S) -2- benzylbutyl acid (27. 8 g, 0. 132 mol) and acetic anhydride (88 mL, 0. 964 mol), at 7 5,0 ° C for 1 hours, cooled and added to isopropyl ether (150 mL) low temperature crystallization, after recrystallization from ethyl acetate, yield: 73% 9;.
Under – (hexahydro-isoindole-2-carbonyl cis) acid synthesis stirring S- benzyl succinic anhydride (12. 7 g, 0 Step 5, (2S) -2- benzyl-3. 067 mol) was dissolved in dichloromethane (250 mL), to control the internal temperature <0 ° C, a solution of cis – hexahydro isoindole (18. 5 g, 0 154 mol), the addition was complete, maintaining the internal temperature in <0! : Continue stirring for 2.5 hours, the reaction in 2 (T25 ° C 12 hours, concentrated to give a pale yellow viscous material, yield: 83 1%; Step 6 Synthesis Mitiglinide calcium.
To the reactor was added (2S) -2- benzyl-3- (cis – hexahydro isoindole-2-carbonyl) propionic acid (. 28. 7 g, 0 091 mol), water (150 mL), and concentrated aqueous ammonia (12 mL), stirring until completely dissolved, a solution of anhydrous calcium chloride (12. 1 g, 0.109 mol) water (100 mL) solution was gradually precipitated white solid was added dropwise at room temperature and then stirred for 13 End hours, filtration, washing the filter cake, and drying to give a white solid, crude Mitiglinide calcium, derived from crude methanol and water (volume ratio 0.5 4~0 5: 1) and recrystallized as a white solid MIG Chennai column calcium, yield: 87.3%.
Second Embodiment
Example A similar experimental method steps 1 through 6 was carried out except in step 3, using (R) -1- naphthyl-amine (61. 4 g, 0. 359 mol) substituted (R) -I- phenylethylamine Other operating homogeneous reaction similar to this step of the synthesis yield: 87.3%.
Third Embodiment
Example A similar experimental method steps 1 through 6 was carried out except in step 3, using (R) -I- phenyl-2-p-tolyl-ethylamine (90. 2 g, 0. 374 mol) substituent (R ) -I- phenethylamine, other homogeneous reaction procedure similar to the synthesis yield of this step:. 83 4% ο
PATENT
WO 199832736

CLIP
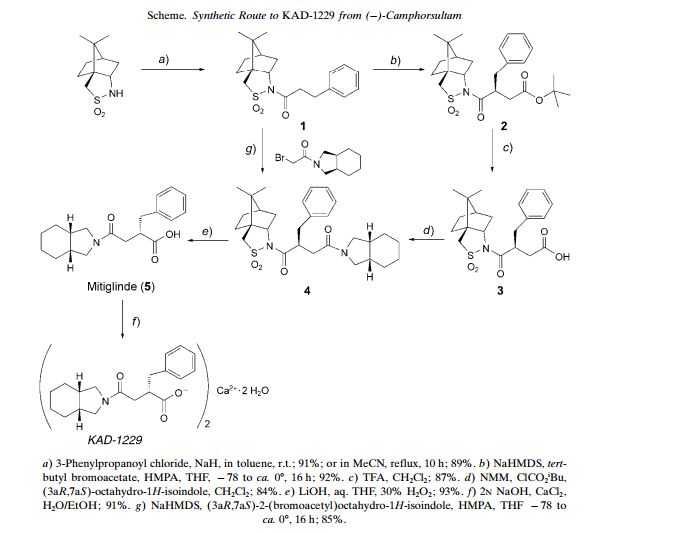
The process for the preparation of KAD-1229 starts from ()-camphorsultam ((3aS)-8,8-dimethylhexahydro-3a,6-methano-2,1-benzisothiazole 2,2-dioxide), readily available in 85% yield from ()- -camphor [4]. Treatment of ()-camphorsultam with excess 3-phenylpropionyl chloride in the presence of NaH in toluene at room temperature gave 1 in 91% yield (Scheme) [5]. An alternative procedure is to reflux camphorsultam with 1.1 to ca. 1.5 equiv. of 3-phenylpropionyl chloride in MeCN for 8 ± 10 h [6]. The crude product, acylsultam 1, purified by recrystallization from EtOH/H2O in 89% yield, was reacted with an equimolar amount of base to form the chiral enolate in dry ice/EtOH bath, followed by C()-re-alkylation [7] with tert-butyl bromoacetate to give 2. The choice of the organic base was very important: the reaction with BuLi, lithium diisopropylamide (LDA), or NaHMD (sodium hexamethyldisilazane) gave 2 in 30 ± 40%, 60%, or 90% yield, respectively, after recrystallization from MeOH. Alkylation promoted by these bases tends to give products with high diastereoselectivity, and the diastereoisomeric purity of crude product 2 was determined to be 93%. However, the reaction with NaHMDS as the base proceeded smoothly in high yield. The tert-butyl ester 2 was cleaved with TFA (CF3COOH) in CH2Cl2 to give the free acid 3 in 87% yield [8]. Acylation of (3aR,7aS)-octahydro-1H-isoindole with 3 by a mixed anhydride method afforded 4 in 84% yield [9]. Compound 4 can be also obtained in 85% yield via direct alkylation of 1 with (3aR,7aS)-2-(bromoacetyl)octahydro-1H-isoindole; however, the yield of the (2-bromoacetyl)octahydro-1H-isoindole prepared from 2-bromoacetyl bromide and cis-octahydro-1H-isoindole was only 40%. Nondestructive cleavage of 4 by hydroperoxide-assisted saponification (LiOH, aq. H2O2 , THF, r.t.) regenerated the camphorsultam (96% recovered yield) and gave mitiglinide (5) in 93% yield and high enantiomeric excess ( 99% by HPLC analysis of the corresponding methyl ester) [7]. Product 5 was treated with 2 NaOH, followed by treatment with CaCl2 . Recrystallization from aqueous EtOH gave KAD-1229 in 91% yield, with a melting point and specific rotation data identical to those in the literature [2b]. Co
(2S)-4-[(3aR,7aS)-Octahydro-2H-isoindol-2-yl]-4-oxo-2-(phenylmethyl)butanoic Acid ( Mitiglinide, 5). base
mitiglinide as a colorless viscous oil. The ee was determined to be 99.4% by HPLC analysis of the corresponding Me ester on a Chiralcel AS column (250 4.6 mm, flow rate 0.7 ml/min, UV 214 nm, n-hexane/i-PrOH 80 : 20 as the eluent).
20 Dalpha= -3.5 (c 1.0, MeOH).
1 H-NMR: 1.23 ± 1.63 (m, 8 H); 2.13 ± 2.22 (m, 2 H); 2.42 ± 2.52 (m, 2 H); 2.73 ± 3.32 (m, 7 H); 7.18 ± 7.32 (m, 5 H).
ESI-MS: 316.15 ( [M H]). Anal. calc. for C19H25NO3 (315.41): C 72.35, H 7.99, N 4.44; found: C 72.51, H 8.03, N 4.31.
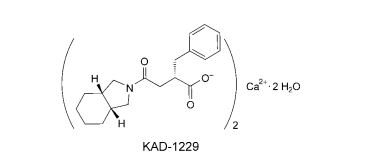
Calcium Bis{(2S)-4-[(3aR,7aS)-octahydro-2H-isoindol-2-yl]-4-oxo-2-(phenylmethyl)butanoate} Dihydrate (KAD-1229).
KAD-1229 as colorless crystals (0.82 g, 91%).
M.p. 179 ± 185 (lit. 179 ± 185 [2b]). 20 D 5.4 (c 0.6, MeOH) (lit. 20 D 5.7, c 1.0, MeOH [2b]).
1 H-NMR: 1.13 ± 1.39 (m, 16 H); 2.0 ± 2.3 (m, 6 H); 2.54 ± 2.83 (m, 14 H); 3.20 ± 3.22 (m, 6 H); 7.11 ± 7.28 (m, 10 H).
ESI-MS: 669.32 ( [M 2 H2O H]). Anal. calc. for C38H48CaN2O6 ¥2H2O (704.91): C 64.75, H 7.44, N 3.94; found: C 64.46, H 7.35, N 3.73.
REFERENCES for aboveclip
[1] H. Ohnota, T. Koizumi, N. Tsutsumi, M. Kobayashi, S. Inoue, S. J. Sato, Pharmacol. Exp. Ther. 1994, 269, 489; H. Ohnota, M. Kobayashi, T. Kiozumi, K. Katsuno, F. Sato, T. Azisawa, Biochem. Pharmacol. 1995, 49, 165; M. Kinukawa, H. Ohnota, T. Azisawa, Br. J. Pharmacol. 1996, 117, 17021.
[2] a) T. Yamaguchi, T. Yanagi, H. Hokari, Y. Mukaiyama, T. Kamijo, I. Yamamoto, Chem. Pharm. Bull. 1997, 45, 1518; b) T. Yamaguchi, T. Yanagi, H. Hokari, Y. Mukaiyama, T. Kamijo, I. Yamamoto, Chem. Pharm. Bull. 1998, 46, 337.
[3] J. P. Lecouve, C. Fugier, J. C. Souvie, Pat. WO9901430, 1999 (Chem. Abstr. 1999, 130, 110156r).
[4] M. Vandewalle, J. Van der Eycken, W. Oppolzer, C. Vullioud, Tetrahedron 1986, 42, 4035; F. A. Davis, J. C. Towson, M. C. Weismiller, S. Lal, P. J. Carroll, J. Am. Chem. Soc. 1988, 110, 8477.
[5] W. Oppolzer, O. Tamura, J. Deerberg, Helv. Chim. Acta 1992, 75, 1965.
[6] M. C. William, B. Corey, J. Org. Chem. 1998, 63, 6732.
[7] W. Oppolzer, R. Moretti, S. Thomi, Tetrahedron Lett. 1989, 30, 5603.
[8] H. Heitsch, R. Henning, H. W. Kleemann, W. Linz, W. U. Nicke, D. Ruppert, H. Urbach, A. Wagner, J. Med. Chem. 1993, 36, 2788.
[9] J. J. Plattner, P. A. Marcotte, H. D. Kleinert, H. H. Stein, J. Greer, G. Bolis, A. K. L. Fung, B. A. Bopp, J. R. Luly, J. Med. Chem. 1988, 31, 2277.
References
- Malaisse WJ (October 2008). “Mitiglinide: a rapid- and short-acting non-sulfonylurea insulinotropic agent for the treatment of type 2 diabetic patients”. Expert Opin Pharmacother. 9 (15): 2691–8. doi:10.1517/14656566.9.15.2691. PMID 18803455.
External links
- Elixir Pharmaceuticals – website of the U.S. rights holder for mitiglinide.
| Cited Patent | Filing date | Publication date | Applicant | Title |
|---|---|---|---|---|
| EP0507534A1 * | Mar 30, 1992 | Oct 7, 1992 | Kissei Pharmaceutical Co., Ltd. | Succinic acid compounds |
| EP0967204A1 * | Jan 22, 1998 | Dec 29, 1999 | Kissei Pharmaceutical Co Ltd | Process for producing benzylsuccinic acid derivatives |
| US6133454 * | Jul 1, 1998 | Oct 17, 2000 | Adir Et Compagnie | Method for preparing a substituted perhydroisoindole |
| Citing Patent | Filing date | Publication date | Applicant | Title |
|---|---|---|---|---|
| CN102898348A * | Sep 8, 2012 | Jan 30, 2013 | 迪沙药业集团有限公司 | Preparation method for Mitiglinide calcium |
| CN102898348B * | Sep 8, 2012 | Sep 2, 2015 | 迪沙药业集团有限公司 | 一种米格列奈钙的制备方法 |
| CN103450069A * | Jun 24, 2013 | Dec 18, 2013 | 山西大同大学 | Preparation method of mitiglinide calcium |
| CN103724253A * | Dec 11, 2013 | Apr 16, 2014 | 苑振亭 | Preparation method for Mitiglinide calcium hydrate |
| CN103724253B * | Dec 11, 2013 | Jun 15, 2016 | 苑振亭 | 一种米格列奈钙的制备方法 |
| CN102659562A * | May 9, 2012 | Sep 12, 2012 | 山东铂源药业有限公司 | Synthesis method of mitiglinide calcium intermediate |
| CN102898348A * | Sep 8, 2012 | Jan 30, 2013 | 迪沙药业集团有限公司 | Preparation method for Mitiglinide calcium |
| CN102898348B * | Sep 8, 2012 | Sep 2, 2015 | 迪沙药业集团有限公司 | 一种米格列奈钙的制备方法 |
| CN103450069A * | Jun 24, 2013 | Dec 18, 2013 | 山西大同大学 | Preparation method of mitiglinide calcium |
| CN103709092A * | Nov 4, 2013 | Apr 9, 2014 | 河北科技大学 | High purity mitiglinide calcium preparation method |
| CN103709092B * | Nov 4, 2013 | Jul 6, 2016 | 河北科技大学 | 米格列奈钙的制备方法 |
| CN104311471A * | Sep 23, 2014 | Jan 28, 2015 | 山东省药学科学院 | Improved mitiglinide calcium industrialized preparation method |
| CN1616427A * | Nov 13, 2003 | May 18, 2005 | 中国科学院上海药物研究所 | New method for preparing medicine mitiglinide for treating diabetes |
| CN101270074A * | Mar 21, 2007 | Sep 24, 2008 | 北京德众万全药物技术开发有限公司 | Method for preparing high purity mitiglinide calcium |
| CN101492411A * | Jan 22, 2008 | Jul 29, 2009 | 北京华禧联合科技发展有限公司 | Improved method for preparation of mitiglinide |
| WO2009047797A2 * | Oct 7, 2008 | Apr 16, 2009 | Ind-Swift Laboratories Limited | Process for the preparation of perhydroisoindole derivative |
| Reference | ||
|---|---|---|
| 1 | * | 张永亮,等: “米格列奈合成方法研究“, 《化工中间体》, no. 1, 31 December 2009 (2009-12-31), pages 16 – 22 |
| Cited Patent | Filing date | Publication date | Applicant | Title |
|---|---|---|---|---|
| CN101492411A * | Jan 22, 2008 | Jul 29, 2009 | 北京华禧联合科技发展有限公司 | Improved method for preparation of mitiglinide |
| WO2005030719A1 * | Sep 24, 2004 | Apr 7, 2005 | Les Laboratoires Servier | Novel method for preparing cis-octahydro-isoindole |
| Reference | ||
|---|---|---|
| 1 | * | WEI HUANG,等: “Novel Convenient Synthesis of Mitiglinide“, 《SYNTHETIC COMMUNICATIONS》, vol. 37, no. 13, 3 July 2007 (2007-07-03), pages 2153 – 2157, XP055079498, DOI: doi:10.1080/00397910701392590 |
| 2 | * | 张永亮,等: “米格列奈合成方法研究“, 《化工中间体》, no. 1, 31 January 2009 (2009-01-31), pages 16 – 22 |
| Citing Patent | Filing date | Publication date | Applicant | Title |
|---|---|---|---|---|
| CN103709092A * | Nov 4, 2013 | Apr 9, 2014 | 河北科技大学 | High purity mitiglinide calcium preparation method |
| CN103709092B * | Nov 4, 2013 | Jul 6, 2016 | 河北科技大学 | 米格列奈钙的制备方法 |
| EP0507534A1 * | Mar 30, 1992 | Oct 7, 1992 | Kissei Pharmaceutical Co., Ltd. | Succinic acid compounds |
| EP0967204A1 * | Jan 22, 1998 | Dec 29, 1999 | Kissei Pharmaceutical Co Ltd | Process for producing benzylsuccinic acid derivatives |
| US6133454 * | Jul 1, 1998 | Oct 17, 2000 | Adir Et Compagnie | Method for preparing a substituted perhydroisoindole |
| Citing Patent | Filing date | Publication date | Applicant | Title |
|---|---|---|---|---|
| CN102898348A * | Sep 8, 2012 | Jan 30, 2013 | 迪沙药业集团有限公司 | Preparation method for Mitiglinide calcium |
| CN102898348B * | Sep 8, 2012 | Sep 2, 2015 | 迪沙药业集团有限公司 | 一种米格列奈钙的制备方法 |
| CN103450069A * | Jun 24, 2013 | Dec 18, 2013 | 山西大同大学 | Preparation method of mitiglinide calcium |
| CN103724253A * | Dec 11, 2013 | Apr 16, 2014 | 苑振亭 | Preparation method for Mitiglinide calcium hydrate |
| CN103724253B * | Dec 11, 2013 | Jun 15, 2016 | 苑振亭 | 一种米格列奈钙的制备方法 |
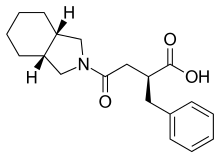 |
|
| Systematic (IUPAC) name | |
|---|---|
|
(2S)-2-benzyl-4-[(3aR,7aS)-octahydro-2H-isoindol- 2-yl]-4-oxobutanoic acid
|
|
| Clinical data | |
| AHFS/Drugs.com | International Drug Names |
| Routes of administration |
oral |
| Identifiers | |
| CAS Number | 145375-43-5 |
| ATC code | A10BX08 (WHO) |
| PubChem | CID 121891 |
| DrugBank | DB01252 |
| ChemSpider | 108739 |
| UNII | D86I0XLB13 |
| KEGG | D01854 |
| ChEMBL | CHEMBL471498 |
| Chemical data | |
| Formula | C19H25NO3 |
| Molar mass | 315.41 g/mol |
/////////207844-01-7, 145525-41-3, KAD-1229, S-21403, MITIGLINIDE, Glufast, Kissei, 145375-43-5
Quality Control & MSDS
O=C(O)[C@@H](Cc1ccccc1)CC(=O)N3C[C@H]2CCCC[C@H]2C3
New aspects of developing a dry powder inhalation formulation applying the quality-by-design approach
DRUG REGULATORY AFFAIRS INTERNATIONAL

The current work outlines the application of an up-to-date and regulatory-based pharmaceutical quality management method, applied as a new development concept in the process of formulating dry powder inhalation systems (DPIs). According to the Quality by Design (QbD) methodology and Risk Assessment (RA) thinking, a mannitol based co-spray dried formula was produced as a model dosage form with meloxicam as the model active agent.
The concept and the elements of the QbD approach (regarding its systemic, scientific, risk-based, holistic, and proactive nature with defined steps for pharmaceutical development), as well as the experimental drug formulation (including the technological parameters assessed and the methods and processes applied) are described in the current paper.
Findings of the QbD based theoretical prediction and the results of the experimental development are compared and presented. Characteristics of the developed end-product were in correlation with the predictions, and all data were confirmed by the relevant results…
View original post 223 more words
ECA Task Force will publish Draft Data Integrity Guideline at Conference in October
DRUG REGULATORY AFFAIRS INTERNATIONAL

Data Integrity has become one of the most frequently observed GMP deviations at FDA and EU Inspections. For that reason the ECA Foundation decided to set up a Task Force on Data Integrity in December 2015 – with the goal to provide Guidance for the implementation in practice. Read more about the ECA Guidance on Data Integrity.
http://www.gmp-compliance.org/eca_mitt_05545_15488_n.html
Data Integrity has become one of the most frequently observed GMP deviations at FDA and EU Inspections. This is why the topic is currently in the centre of attention of both regulators and industry. And for that reason the ECA Foundation decided to set up a Task Force on Data Integrity in December 2015 – with the goal to provide Guidance for the implementation in practice.
The ECA Task Force will be comprised of members from both the IT Compliance Group and the Analytical QC Group. Current Members are:
– Dr. Wolfgang Schumacher…
View original post 177 more words
D2/5-HT2A receptor dual antagonist, (±)-SIPI 6360

(±)-SIPI 6360
D2/5-HT2A receptor dual antagonist
7-[3-[4-(6-fluoro-1,2-benzoxazol-3-yl)piperidin-1-yl]propoxy]-3-methyl-3,4-dihydro-1H-quinolin-2-one
2(1H)-Quinolinone, 7-[3-[4-(6-fluoro-1,2-benzisoxazol-3-yl)-1-piperidinyl]propoxy]-3,4-dihydro-3-methyl-
| Molecular Formula: | C25H28FN3O3 |
|---|---|
| Molecular Weight: | 437.506523 g/mol |
Schizophrenia is a common severe mental patients, mental illness is the most serious of all, the most dangerous kind, the worldwide incidence of about I%, with the accelerate pace of social life, The incidence was significantly increased. Most schizophrenic patients due to the long treatment period, high cost, side effects and give up the treatment, often lead to more serious social consequences.
Numerous studies show that the brain monoamine neurotransmitters, especially dopamine and 5-hydroxytryptamine system is closely related to the body’s normal mental activity, these two types of system disorder can lead to a variety of neuropsychiatric diseases such as schizophrenia , neuropathic pain, mania, anxiety disorders, all kinds of depression, Parkinson’s disease and the like.
The drugs currently used clinically primarily for conventional antipsychotics (such as dopamine D2 receptor antagonists) and atypical antipsychotics (such as D2 / 5-HT2a dual antagonist), where conventional antipsychotics because it is easy leads to extrapyramidal symptoms (EPS) and gradually phased out, atypical antipsychotics variety, but no one medication to improve the overall spectrum of schizophrenia has the absolute advantage, most of the positive or negative symptoms of a a symptom improvement, or reduced side effects. So look for low toxicity, rapid onset, treatment spectral width of new anti-schizophrenia drug has been a hot topic in the world pharmaceutical industry.
In recent years, scientists have found that the dopamine D2 partial agonist can over time reduce dopamine activity in the transfer of dopamine, but not all block; the other hand, when the low dopaminergic activity is caused by stimulating effect on both positive and negative symptoms of mental illness have a significant effect. 5-HT2a receptor antagonists can improve negative symptoms, while synergies D2 EPS side effects can be reduced to about 1% level (classical antipsychotic drugs EPS incidence is about 30%), part of the 5-HTla agonism and 5-HT2a and synergy can make in therapeutic doses under observation EPS decreased to undetectable levels, therefore, has D2 ,5-HT2a, 5HTla synergy targets three new anti-drugs are currently developed Jingshenfenlie focus and an important development direction.
The present invention relates to a quinoline derivative can stabilize the brain dopaminergic, serotonergic energy system, may for a variety of neurological and psychiatric diseases have improved and treatment can be used for neuropathic pain, mania, schizophrenia, anxiety disorders, all kinds of depression, Parkinson’s disease, especially in the treatment of schizophrenia.
DETAILS COMING……….
PATENT

PATENT

Example 1
1-1
7- (3- (4- (6-fluorophenyl and [d] different dumb-3-yl) piperidin-1-yl) propoxy) -3,4-dihydro-3-carboxylic acid -one – yl quinolin -2 (1H)
1) N- (3- methoxyphenyl) propionamide
3-methoxy-aniline (0.1mol), methylene chloride (30 mL), triethylamine (0.2mol), was added to the flask lOOmL three, propionyl chloride was added dropwise under ice (0.12mol) in methylene chloride 30 mL, temperature does not exceed 5 ° C, the addition was complete, the ice bath was removed and stirred at room temperature 0.5h, the system was washed with water, dilute hydrochloric acid, saturated brine, dried over anhydrous magnesium sulfate, and evaporated to dryness to give a white powdery solid 17.01g yield 95%.
2) 2-chloro-7-methoxy-3-methylquinoline
The DMF (20mL) was added to the three 250mL flask, was added dropwise under ice-salt bath of POCl 3 (100 mL), temperature does not exceed 0 ° C, the addition was completed stirring 0.5h, was added portionwise N- (3- methoxyphenoxy yl) propanamide powder (31.0g), was slowly warmed to 50 ° C, violent reaction, to be exothermic easing slowly warmed to reflux, the reaction was kept 2h, cooled to room temperature, the system was poured into 800 g of crushed ice to sodium carbonate to adjust the pH to 7 to precipitate a yellow solid with petroleum ether – ethyl acetate to give pure product 20.86g, yield 58%.
3) 3-methyl-7-methoxy-quinolin -2 (1H) – one
2-Chloro-7-methoxy-3-methyl-quinoline (20.76g), acetic acid (150 mL) placed in 250mL one-neck flask, heated at reflux for 24h, acetic acid recovery, and the residue was recrystallized from ethanol to 95%, white needle crystalline 16.08g, yield 85%.
4) 7-methoxy-3,4-dihydro-3-methyl-quinolin -2 (1H) – one
7-Methoxy-3-methyl-quinolin -2 (1H) – one (18.92g), acetic acid (150mL), 10% Pd / C (lg) was added to the three 250mL flask, the system was replaced with nitrogen air, and then the nitrogen was replaced with hydrogen, and then the reaction was heated to 80 ° C overnight, cooled to room temperature, filtered and the filtrate evaporated to dryness to give a white powder, washed with water once, 50 ° C and dried in vacuo 4h, as a white powdery solid 18.91g yield of 98.95%.
5) 7-hydroxy-3,4-dihydro-3-methyl-quinolin -2 (1H) – one
7-Methoxy-3,4-dihydro-3-methyl-quinolin -2 (1H) – one (19.12g), 40% hydrobromic acid (150 mL) placed in 250mL one-neck flask was heated at reflux for 12h cooled to room temperature, the precipitated solid was filtered, the filter cake successively with hydrobromic acid, washed with water, 50 ° C and dried in vacuo 4h, 14.60 g as a white powdery solid, yield 82.4%.
6) 3- (1- (3-chloropropyl) piperidin-4-yl) -6-fluorophenyl and [d] oxazole different dumb
6-fluoro-3- (piperidin-4-yl) benzo [d] isoxazol dummy oxazole (22.00g), 1- bromo-3-chloropropane (40mL), anhydrous potassium carbonate (40g), acetone ( 250mL) was added to a 500mL one-neck flask was refluxed overnight, cooled to room temperature, filtered, the filter cake was washed twice with hot acetone and the combined filtrate was added dropwise a solution of anhydrous hydrogen chloride in ethanol, the precipitated white solid was filtered, the filter cake washed with acetone after washing once, it was dissolved in 200mL of water, adjusted with sodium carbonate to pH 9, and filtered to obtain a white powdery solid 15.94 g, yield 48.0%
7) 7- (3- (4- (6-fluorobenzo [d] isoxazol-3-yl dummy) piperidin-1-yl) propoxy) -3,4-dihydro-3-methyl quinolin -2 (1H) – one
3- (1- (3-chloropropyl) piperidin-4-yl) -6-fluorophenyl and [d] oxazole different dumb (lmmol), 7- hydroxy-3,4-dihydro-3-carboxylic acid yl quinolin -2 (1H) – one (1.0 mmol), anhydrous potassium carbonate (3.0mmol) were added to the lOmLDMF, 60 ° C overnight the reaction, potassium carbonate was filtered off, the mother liquor evaporated to dryness to give a pale yellow solid, the filter cake recrystallized with 95% ethanol, 50 ° C and dried in vacuo 4h, as a white powdery solid 0.30g, 69% yield.
NMR IH (of DMSO-D . 6 ): L up to .27 (D, 3H, J = 9.2Hz), 2.06-2.32 (m, 9H), 2.67-2.69 (T, 2H), 2.95 (D * D, lH, J = 3.2Hz, 12.8Hz), 3.15-3.17 ( m, 2H), 4.05 (t, 2H, J = 6Hz), 6. 37 (d, lH, J = 2.4Hz), 6.56 (d * d, lH, J = 2.4Hz, 8.0Hz), 7.05-7.11 (m, 2H), 7.25-7.29 (m, lH), 7.73-7.76 (m, lH), 7.98 (s, lH), 11.43 (brs, lH)
ESI-MS: 438 (M + 1)
Example 2
Preparation 1-1 hydrochloride
7- (3- (4- (6-fluorophenyl and [d] different dumb-3-yl) piperidin-1-yl) propoxy) -3,4-dihydro-3-methyl-quinoline morpholine -2 (1H) – one (lmmol) was dissolved with ethyl acetate (50 mL) was slowly added dropwise a solution of anhydrous hydrogen chloride in ethyl acetate (lmol / L, 5mL), stirred for 2h, the precipitated solid was filtered, the filter cake washed with ethyl acetate, 50 ° C and dried in vacuo 4h, as a white powdery solid 0.436g, yield 92%.
ESI-MS: 438 (M + 1)
Elemental analysis results:
Calcd: C, 63.35%; H, 6.17%; Cl, 7.48%; F, 4.01%; N, 8.87%; O, 10.13%
Found: C, 63.29%; H, 6.24%; CI, 7.43%; F, 4.05%; N, 8.82%; O, 10.17%
Example 3
Preparation 1-1 methanesulfonate
The 1-1 (lmmol) was dissolved with ethyl acetate (50 mL) was slowly added dropwise a solution of methanesulfonic acid in ethyl acetate (lmol / L, 5mL), stirred for 2h, the precipitated solid was filtered, the filter cake with ethyl acetate wash, 50 ° C and dried in vacuo 4h, as a white powdery solid 0.487g, yield 91.3%.
ESI-MS: 438 (M + 1, positive mode), 95 (CH 3 the SO 3 -, negative mode) Elemental analysis:
Calcd: C, 58.52%; H, 6.04%; F, 3.56%; N, 7.87%; 0, 17.99%; S, 6.01%
Found: C, 58.49%; H, 6.09%; F, 3.50%; N, 7.81%; 0, 18.02%; S, 6.09%
PATENT
Paper
Development and Kilogram-Scale Synthesis of a D2/5-HT2A Receptor Dual Antagonist (±)-SIPI 6360

The kilogram-scale synthesis of a D2/5-HT2A receptor dual antagonist (±)-SIPI 6360 was developed as an alternative treatment for schizophrenia. Specifically, three conditions were modified and optimized, including the Vilsmeier conditions, to prepare quinoline 3. In addition, the palladium-catalyzed hydrogenation was modified to synthesize dihydroquinolin-2(1H)-one 5, and the reduction of β-chloroamide was altered to form 3-chloropropanamine 8. Ultimately these improvements led to the preparation of a 1.5 kg of (±)-SIPI 6360 batch in eight steps with an overall yield of 34% and purity of 99.8%.
//////// D2/5-HT2A receptor dual antagonist (±)-SIPI 6360, 1401333-14-9
c21CC(C(Nc1cc(cc2)OCCCN3CCC(CC3)c4c5ccc(cc5on4)F)=O)C
Fortune India presents award to Ajanta pharma
Fortune India has published list of 500 mid-size companies who ranked them on various parameters based on the results of 2013-2014. Ajanta pharma features very prominently in the lists. Ajanta’s ranking on various pararmeters is given below:
Ranked 3’d largest Wealth Creator on 5 year CAGR 93.14%
Ranked 10th on Capital Employed (ROCE)
Ranked 21st in Net profit .
Ranked 182nd in Sales
On 17’th August 2015, Fortune India organized an award function to present the awards to Top 10 largest weatth creator companies and Ajanta is one of those elite companies.
The awards were presented by Mr. Piyush Goyal, Minister of State-Power,Coal& New and Renewable Energy, Govt. of India to Mr. yogesh Agrawal, Managing Director and Mr.Rajesh Agrawal,Jt.Managing Director of the company.


Ajanta Pharma, “One of the Giants of Tomorrow” – Fortune India
We are pleased to share with you that Ajanta Pharma has been honoured as “ONE OF THE GIANTS OF TOMORROW” by prestigious Fortune India magazine on 19th August 2016 at New Delhi.
The honour was conferred to our Managing Director, Mr. Yogesh Agrawal and our Jt. Managing Director, Mr. Rajesh Agrawal at the hands of Hon. Mr. Nitin Gadkari, Union Minister for Road Transport and Highways and Shipping, Govt of India. This is the 2nd year in row where Ajanta has received the recognition from Fortune India.
Fortune India (June 2016 Issue) published the list of mid-size companies based on the financial year 2014-15 results and we are pleased to share with you that Ajanta has been ranked 3rd Top Wealth creator over last 5 years.


Ajanta Pharma Limited (APL) is a pharmaceutical company headquartered in Mumbai, India. It has strong presence in Branded Generic business in India & Emerging markets; and Generic business in USA. In India, company operates in selected therapeutic areas of Cardiology, Dermatology, Ophthalmology and Pain management. Its brands in each of sub-therapeutic areas or molecules hold leadership positions. In Emerging Markets, company has presences in Africa, Asia, Middle East, and CIS on broader therapeutic segments such as anti-malarial, gastro, antibiotics, cardiology, dermatology, pain management, etc. In USA, company has already no. of approved ANDA’s which are either commercialized or in process of being commercialized and large no. of ANDA’s are awaiting US FDA approval. We have state-of-the-art research facilities for formulation (finished product) and API development located at Mumbai, India. Our R&D capabilities are evident from number of products launched 1st to market by the company providing patients most needed compliance and convenience. A dedicated and focused team of more than 750 Ajantaites work for R&D, which is growing continuously. Ajanta has four formulations manufacturing facilities located in India and 1 in Mauritius. Besides that, we also have an API manufacturing facility located at Waluj, India. Ajanta’s flagship formulation facility at Paithan (Maharashtra, India) has approval of USFDA, WHO- Geneva (prequalification), UNICEF and many regulatory authorities from different parts of the world. We continuously invest in enhancing our existing manufacturing facilities to meet current cGMP requirements and also construct new facilities to meet the company’s growth requirements. We are in process of setting up 1 more formulations manufacturing facility for domestic and emerging markets at Guwahati, Assam. Please visit http://ajantapharma.com/ for more information. Contact: careers@ajantapharma.com
Specialties
Speciality Branded Generics, Generics, Complex Formulations
-
Website
-
Industry
Pharmaceuticals
-
Type
Public Company
-
Headquarters
98 Ajanta House Charkop, Kandivili West Mumbai,Maharashtra 400067 India
-
Company Size
5001-10,000 employees
-
Founded
1973
Rajesh Agrawal (left), Ajanta Pharma’s joint managing director, with brother Yogesh, who is also managing director of the company, at their Kandivli facility
Ajanta Pharma needed a shot of its own medicine, an energiser like 30-Plus. It found its antidote in the new generation of Agrawals: Mannalal’s sons, Yogesh and Rajesh.

“When I joined Ajanta (in 2000), and realised what was going on, I wanted to run away. I thought to myself, ‘Why did I return from the US? I could have had a job there,’” says Rajesh, 39, Ajanta’s joint managing director, who has a management degree from Bentley College, Massachusetts. “It was tough in the beginning, especially the situation with creditors and debtors.”
Together, Rajesh and his older brother Yogesh, 43, who is managing director, changed Ajanta’s trajectory by focusing on the ‘specialty’ generic drug market and putting an end to the company’s legacy businesses, which included OTC drug sales and supplying drugs to government health agencies in India and other countries.
This was a risky move, but it has paid off. Ajanta Pharma closed FY15 with a consolidated net sales of Rs 1,481 crore and a net profit of Rs 310 crore (this is a compound annual growth rate, or CAGR, of 57 percent for four years since 2011). In terms of net sales, it recorded a CAGR of 31 percent for the same period. This growth has come on a low base, but the signs are encouraging. Its market value currently stands at around Rs 13,500 crore; this is a 65-fold growth in 15 years.
References
https://www.linkedin.com/company/263285
/////////Ajanta Pharma, “One of the Giants of Tomorrow” , Fortune India, AWARD, Fortune India, RAJESH AGRAWAL
ROMIDEPSIN
![Skeletal formula of (1S,4S,7Z,10S,16E,21R)-7-ethylidene-4,21-diisopropyl-2-oxa-12,13-dithia-5,8,20,23-tetrazabicyclo[8.7.6]tricos-16-ene-3,6,9,19,22-pentone](https://upload.wikimedia.org/wikipedia/commons/thumb/7/71/Romidepsin_structure_%282%29.svg/200px-Romidepsin_structure_%282%29.svg.png)
| Romidepsin; Depsipeptide; FK228; Chromadax; FR901228; Istodax; | |
| Molecular Formula: | C24H36N4O6S2 |
|---|---|
| Molecular Weight: | 540.69584 g/mol |
CAS 128517-07-7
(1S,4S,7Z,10S,16E,21R)-7-ethylidene-4,21-di(propan-2-yl)-2-oxa-12,13-dithia-5,8,20,23-tetrazabicyclo[8.7.6]tricos-16-ene-3,6,9,19,22-pentone
(E)-N-(2-amino-4-fluorophenyl)-4-((3-(pyridin-3-yl)acrylamido)methyl)benzamide
Romidepsin, also known as Istodax, is an anticancer agent used in cutaneous T-cell lymphoma (CTCL) and other peripheral T-cell lymphomas (PTCLs). Romidepsin is a natural product obtained from the bacteria Chromobacterium violaceum, and works by blocking enzymes known as histone deacetylases, thus inducing apoptosis.[1] It is sometimes referred to as depsipeptide, after the class of molecules to which it belongs. Romidepsin is branded and owned by Gloucester Pharmaceuticals, now a part of Celgene.[2]
Romidepsin, a histone deacetylase inhibitor, originally developed by Fujisawa (now Astellas Pharma), causes cell cycle arrest,
differentiation, and apoptosis in various cancer cells.
In 2004, the FDA granted fast-track designation for romidepsin as monotherapy for the treatment of cutaneous T-cell lymphoma (CTCL) in patients who have relapsed following, or become refractory to, other systemic therapies. The FDA designated romidepsin as an orphan drug and it was approved in 2009 for this indication and it was commercialized in 2010. In 2007, another fast-track designation was granted for the product as monotherapy of previously treated peripheral T-cell lymphoma.
Romidepsin (FR901228) was originally discovered and isolated from the fermentation broth of Chromobacterium violaceum No. 968. It was identified through efforts in the search for novel agents which selectively reverse the morphological phenotype of Ras oncogene-transformed cells since the Ras signaling pathway plays a critical role in cancer development. Therefore, the drug could also have multiple molecular targets for its anticancer activity besides HDAC.
FR901228 is a bicyclic depsipeptide which is structurally unrelated to any known class of cyclic peptides with an unusual disulfide bond connecting a thiol and D-cysteine.
This drug is commercially produced by fermentation; however its interesting and novel structure warrants examination of its synthesis within the context of this review
Romidepsin is a histone deacetylase (HDAC) inhibitor.HDACs catalyze the removal of acetyl groups from acetylated lysine residues in histone and non-histone proteins, resulting in the modulation of gene expression.
Romidepsin is indicated for treatment of cutaneous T-cell lymphoma (CTCL) in patients who have received at least
one prior systemic therapy; treatment of peripheral T-cell lymphoma (PTCL) in patients who have received at least
one prior therapy.
Available as an injection, containing 10 mg of romidepsin and recommended dose is 14 mg/m2 administered intravenously over a 4-hour period on days 1, 8, and 15 of a 28-day cycle until disease progression or unacceptable toxicity.

History
Romidepsin was first reported in the scientific literature in 1994, by a team of researchers from Fujisawa Pharmaceutical Company (now Astellas Pharma) in Tsukuba, Japan, who isolated it in a culture of Chromobacterium violaceum from a soil sample obtained inYamagata Prefecture.[3] It was found to have little to no antibacterial activity, but was potently cytotoxic against several human cancercell lines, with no effect on normal cells; studies on mice later found it to have antitumor activity in vivo as well.[3]
The first total synthesis of romidepsin was accomplished by Harvard researchers and published in 1996.[4] Its mechanism of actionwas elucidated in 1998, when researchers from Fujisawa and the University of Tokyo found it to be a histone deacetylase inhibitorwith effects similar to those of trichostatin A.[5]

Clinical trials
Phase I studies of romidepsin, initially codenamed FK228 and FR901228, began in 1997.[6] Phase II and phase III trials were conducted for a variety of indications. The most significant results were found in the treatment of cutaneous T-cell lymphoma (CTCL) and other peripheral T-cell lymphomas (PTCLs).[6]
In 2004, romidepsin received Fast Track designation from the FDA for the treatment of cutaneous T-cell lymphoma, and orphan drugstatus from the FDA and the European Medicines Agency for the same indication.[6] The FDA approved romidepsin for CTCL in November 2009[7] and approved romidepsin for other peripheral T-cell lymphomas (PTCLs) in June 2011.[8]
Mechanism of action
Romidepsin acts as a prodrug with the disulfide bond undergoing reduction within the cell to release a zinc-binding thiol.[3][9][10] The thiol reversibly interacts with a zinc atom in the binding pocket of Zn-dependent histone deacetylase to block its activity. Thus it is anHDAC inhibitor. Many HDAC inhibitors are potential treatments for cancer through the ability to epigenetically restore normal expression of tumor suppressor genes, which may result in cell cycle arrest, differentiation, and apoptosis.[11]

Adverse effects
The use of romidepsin is uniformly associated with adverse effects.[12] In clinical trials, the most common were nausea and vomiting,fatigue, infection, loss of appetite, and blood disorders (including anemia, thrombocytopenia, and leukopenia). It has also been associated with infections, and with metabolic disturbances (such as abnormal electrolyte levels), skin reactions, altered taste perception, and changes in cardiac electrical conduction.[12]
CLIP
http://www.scielo.br/scielo.php?script=sci_arttext&pid=S0103-50532012001200003

CLIP
Romidepsin was first isolated from the fermentation broth of Chromobacterium Violaceum WB968 in a nutrient
medium. Sterilized of 1% glucose and 1% bouillon solution were incubated with Chromobacterium Violaceum WB968, followed by further incubation with 1% glucose solution, 1% bouillon solution and adekanol gave the target romidepsin after extraction, silica gel chromatography and recrystallization.[Okuhara, M.; Goto, T.; Hori, Y. et al. US4977138A, 1990.]
The synthetic route was initiated by the deprotection L-(Fmoc)Thr-L-Val-OMe 1, subsequently coupled with
N-Alloc-S-Trt-D-Cys, followed by tosylation and then elimination to produce tripeptide 3 in the yield of 63.7% over four steps. The N-Alloc deprotection of 3 and then coupling with N-Fmoc-D-Valine were proceeded to provide tetrapeptide 4, which was subsequently removed Fmoc group to afford relative tetrapeptide 5 in 83.0% yield from compound 3. Condensation of 5 with β-hydroxy mercapto acid 6 was carried out by treating with benzotriazol-1-yl-oxy-tris-(dimethylamino)-phosphonium hexafluorphosphate (BOP) to give relative amide 7, and sequential hydrolysis yielded corresponding acid, which was performed by Mitsunobu macrolactonization
to produce depsipeptide 8 in 17.5% yield over three steps. Finally, romidepsin was obtained in the presence of iodine
in 81.0% yield and the overall yield of 7.5%.
The synthesis of intermediate β-hydroxy mercapto acid 6 commenced with the commercially available methyl 3,3-dimethoxypropionate 9. Nucleophilic addition of 9 with N,O-dimethylhydroxylamine provided Weinreb amide 10, followed by addition with lithium acetylide to give propargylic ketone 12 in the yield of 50.2% over two steps. Noyori’s asymmetric hydrogenation of ketone 12 provided (E)-alkene 14, which was removed the silyl group and then substituted with paratoluensulfonyl chloride to yield tosylate 15 in 40.6% yield across three steps. The dimethyl acetal of 15 was hydrolyzed to corresponding aldehyde by using lithium tetrafluoroborate,
which was immediately oxidized to relative carboxylic acid by applying Pinnick oxidation conditions. The trityl mercaptan was introduced by tosylate displacement to provide 6 in 65.0% yield over three steps and the overall yield of 13.3%.[2]
REF Greshock, T. J.; Johns, D. M.; Noguchi, Y., et al. Org. Lett. 2008, 10 (4), 613-616.
CLIP
Romidepsin (Istodax)
Romidepsin, a histone deacetylase inhibitor, originally developed by Fujisawa (now Astellas Pharma), causes cell cycle arrest,
differentiation, and apoptosis in various cancer cells.111 In 2004, the FDA granted fast-track designation for romidepsin as monotherapy for the treatment of cutaneous T-cell lymphoma (CTCL) in patients who have relapsed following, or become refractory
to, other systemic therapies. The FDA designated romidepsin as an orphan drug and it was approved in 2009 for this indication
and it was commercialized in 2010. In 2007, another fast-track designation was granted for the product as monotherapy of
previously treated peripheral T-cell lymphoma. Romidepsin (FR901228) was originally discovered and isolated from the fermentation
broth of Chromobacterium violaceum No. 968. It was identified through efforts in the search for novel agents which
selectively reverse the morphological phenotype of Ras oncogene-transformed cells since the Ras signaling pathway plays a
critical role in cancer development. Therefore, the drug could also have multiple molecular targets for its anticancer activity besides
HDAC.112 FR901228 is a bicyclic depsipeptide which is structurally unrelated to any known class of cyclic peptides with an unusual
disulfide bond connecting a thiol and D-cysteine. This drug is commercially produced by fermentation; however its interesting
and novel structure warrants examination of its synthesis within the context of this review.113,114 The synthesis of romidepsin
described is based on the total synthesis reported by the Williams115 and Simon groups (Scheme 20).116
L-Valine methyl ester (134) was coupled to N-Fmoc-L-threonine in the presence of the BOP reagent in 95% yield. The N-Fmoc protecting group was removed with Et2NH and the corresponding free amine was coupled to N-alloc-(S-triphenylmethyl)-D-cysteine with 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide (EDCI) and HOBT in DMF and CH2Cl2 to yield the tripeptide 135 in good yield. The threonine residue of tripeptide 135 was then subjected to dehydrating conditions to give alkene 136 in 95% yield. The N-alloc protecting group of the dehydrated tripeptide 136 was removed with palladium and tin reagents and the corresponding free amine was subsequently coupled with N-Fmoc-D-valine to give tetrapeptide 137 in 83% yield. After removal of the N-Fmoc protecting group of compound 137 with Et2NH amine 138 was obtained in quantitative yield. The acid coupling partner 145 for
amine 138 was prepared as follows: methyl 3,3-dimethoxypropionate (139) was converted to its corresponding Weinreb amide by standard conditions and reacted with lithium acetylide 140 to give propargylic ketone 141 in 75% yield. Noyori’s asymmetric reduction of ketone 141 using ruthenium catalyst 142 gave the (R)-propargylic alcohol in 98% ee. This was followed by Red-Al reduction of the alkyne to selectively yield (E)-alkene 143 in 58% yield for the two steps. Liberation of the primary alcohol
with tetrabutylammonium fluoride (TBAF) followed by selective tosylation gave 144 in 70% yield in two steps. Hydrolysis of the dimethyl acetal of 144 with LiBF4 was followed by a Pinnick oxidation to give the corresponding carboxylic acid. The tosylate was displaced with trityl mercaptan in the presence of tert-butyl alcohol to give allylic alcohol 145 in 65% yield for the three steps.
Aminoamide 138 was then coupled to acid 145 using BOP to give peptide 146 in quantitative yield. The methyl ester of compound 146 was hydrolyzed with lithium hydroxide to provide the free carboxylic acid which underwent macrolactonization under Mitsunobu conditions in the presence of diisopropyl azodicarboxylate (DIAD) and triphenylphosine to give macrocycle 147 in 24% yield.
Finally, the disulfide linkage was formed by treating bis-tritylsulfane 147 with iodine in methanol at room temperature to give romidepsin (XIII) in 81% yield.
111 Bertino, E. M.; Otterson, G. A. Expert Opin. Invest. Drugs 2011, 20, 1151.
112. Furumai, R.; Matsuyama, A.; Kobashi, N.; Lee, K.-H.; Nishiyama, M.; Nakajima,
H.; Tanaka, A.; Komatsu, Y.; Nishino, N.; Yoshida, M.; Horinouchi, S. Cancer
Res. 2002, 62, 4916.
113. Verdine, G. L.; Vrolijk, N. H.; Bertel, S. WO 2008083288 A2, 2008.
114. Verdine, G. L.; Vrolijk, N. H. WO 2008083290 A1, 2008.
115. Greshock, T. J.; Johns, D. M.; Noguchi, Y.; Williams, R. M. Org. Lett. 2008, 10,
613.
116. Li, K. W.; Wu, J.; Xing, W.; Simon, J. A. J. Am. Chem. Soc. 1996, 118, 7237.
CLIP
http://pubs.rsc.org/en/content/articlelanding/2009/np/b817886k#!divAbstract

Williams’ improved synthesis of FK228.


Williams’ synthesis of the FK228 amide isostere (74).
References
- Jump up^ “Romidepsin”. National Cancer Institute. Retrieved2009-09-11.
- Jump up^ “Romidepsin”. Gloucester Pharmaceuticals. Retrieved2009-09-11.
- ^ Jump up to:a b c Ueda H, Nakajima H, Hori Y, et al. (March 1994). “FR901228, a novel antitumor bicyclic depsipeptide produced byChromobacterium violaceum No. 968. I. Taxonomy, fermentation, isolation, physico-chemical and biological properties, and antitumor activity”. Journal of Antibiotics. 47 (3): 301–10.doi:10.7164/antibiotics.47.301. PMID 7513682.
- Jump up^ Li KW, Wu J, Xing W, Simon JA (July 1996). “Total synthesis of the antitumor depsipeptide FR-901,228”. Journal of the American Chemical Society. 118 (30): 7237–8. doi:10.1021/ja9613724.
- Jump up^ Nakajima H, Kim YB, Terano H, Yoshida M, Horinouchi S (May 1998). “FR901228, a potent antitumor antibiotic, is a novel histone deacetylase inhibitor”. Experimental Cell Research. 241(1): 126–33. doi:10.1006/excr.1998.4027. PMID 9633520.
- ^ Jump up to:a b c Masuoka Y, Shindoh N, Inamura N (2008). “Histone deacetylase inhibitors from microorganisms: the Astellas experience”. In Petersen F, Amstutz R. Natural compounds as drugs. 2. Basel: Birkhäuser. pp. 335–59. ISBN 978-3-7643-8594-1. Retrieved on November 8, 2009 through Google Book Search.
- Jump up^ http://chembl.blogspot.com/2009/11/new-drug-approvals-pt-xxiii-romidepsin.html
- Jump up^http://www.accessdata.fda.gov/scripts/cder/drugsatfda/index.cfm?fuseaction=Reports.MonthlyApprovalsAll
- Jump up^ Shigematsu, N.; Ueda, H.; Takase, S.; Tanaka, H.; Yamamoto, K.; Tada, T. (1994). “FR901228, a novel antitumor bicyclic depsipeptide produced by Chromobacterium violaceum No. 968. II. Structure determination.”. J. Antibiot. 47 (3): 311–314.doi:10.7164/antibiotics.47.311. PMID 8175483.
- Jump up^ Ueda, H.; Manda, T.; Matsumoto, S.; Mukumoto, S.; Nishigaki, F.; Kawamura, I.; Shimomura, K. (1994). “FR901228, a novel antitumor bicyclic depsipeptide produced by Chromobacterium violaceum No. 968. III. Antitumor activities on experimental tumors in mice.”. J. Antibiot. 47 (3): 315–323.doi:10.7164/antibiotics.47.315. PMID 8175484.
- Jump up^ Greshock, Thomas J.; Johns, Deidre M.; Noguchi, Yasuo; Williams, Robert M. (2008). “Improved Total Synthesis of the Potent HDAC Inhibitor FK228 (FR-901228)”. Organic Letters.10 (4): 613–616. doi:10.1021/ol702957z. PMC 3097137
 .PMID 18205373.
.PMID 18205373. - ^ Jump up to:a b [No authors listed] (October 2014). “ISTODEX Label Information (updated to October 2014)” (PDF). U.S. Food and Drug Administration.
External links
![Skeletal formula of (1S,4S,7Z,10S,16E,21R)-7-ethylidene-4,21-diisopropyl-2-oxa-12,13-dithia-5,8,20,23-tetrazabicyclo[8.7.6]tricos-16-ene-3,6,9,19,22-pentone](https://upload.wikimedia.org/wikipedia/commons/thumb/7/71/Romidepsin_structure_%282%29.svg/200px-Romidepsin_structure_%282%29.svg.png) |
|
 |
|
| Systematic (IUPAC) name | |
|---|---|
|
(1S,4S,7Z,10S,16E,21R)-7-ethylidene-4,21-diisopropyl-2-oxa-12,13-dithia-5,8,20,23-tetrazabicyclo[8.7.6]tricos-16-ene-3,6,9,19,22-pentone
|
|
| Clinical data | |
| Trade names | Istodax |
| MedlinePlus | a610005 |
| License data |
|
| Pregnancy category |
|
| Routes of administration |
Intravenous infusion |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | Not applicable (IV only) |
| Protein binding | 92–94% |
| Metabolism | Hepatic (mostly CYP3A4-mediated) |
| Biological half-life | 3 hours |
| Identifiers | |
| CAS Number | 128517-07-7 |
| ATC code | none |
| PubChem | CID 5352062 |
| IUPHAR/BPS | 7006 |
| UNII | CX3T89XQBK |
| ChEBI | CHEBI:61080 |
| ChEMBL | CHEMBL1213490 |
| Synonyms | FK228; FR901228; Istodax |
| Chemical data | |
| Formula | C24H36N4O6S2 |
| Molar mass | 540.695 g/mol |
//////////fast-track designation, Romidepsin, Depsipeptide, FK228, Chromadax, FR901228, Istodax, FDA 2009, Fujisawa, Astellas Pharma, 128517-07-7
CC=C1C(=O)NC(C(=O)OC2CC(=O)NC(C(=O)NC(CSSCCC=C2)C(=O)N1)C(C)C)C(C)C
ACT-334441, Cenerimod an S1P receptor 1 agonist

ACT-334441
Cenerimod
UNII-Y333RS1786; Y333RS1786
S1P receptor 1 agonist
CAS 1262414-04-9
Chemical Formula: C25H31N3O5
Exact Mass: 453.22637
Martin Bolli, Cyrille Lescop, Boris Mathys,Keith Morrison, Claus Mueller, Oliver Nayler,Beat Steiner,
(S)-3-(4-(5-(2-cyclopentyl-6-methoxypyridin-4-yl)-1,2,4-oxadiazol-3-yl)-2-ethyl-6-methylphenoxy)propane-1,2-diol
(2S)-3-[4-[5-(2-cyclopentyl-6-methoxypyridin-4-yl)-1,2,4-oxadiazol-3-yl]-2-ethyl-6-methylphenoxy]propane-1,2-diol
(S)-3-{4-[5-(2-Cyclopentyl-6-methoxy-pyridin-4-yl)-[1,2,4]oxadiazol-3-yl]-2-ethyl-6-methyl-phenoxy}-propane-1,2-diol
| Mechanism Of Action | Sphingosine 1 phosphate receptor modulator |
|---|---|
| Who Atc Codes | L03A-X (Other immunostimulants) |
| Ephmra Codes | L3A (Immunostimulating Agents Excluding Interferons) |
| Indication | Systemic Lupus Erythematosus |
Cenerimod is a potent and orally active immunomodulator, exhibited EC50 value of 2.7 nM. Cenerimod is an agonist for the G protein-coupled receptor S1 P1/EDG1 and has a powerful and long-lasting immunomodulating effect which is achieved by reducing the number of circulating and infiltrating T- and B-lymphocytes, without affecting their maturation, memory, or expansion. Cenerimod may be useful for prevention or treatment of diseases associated with an activated immune system

CENERIMOD
ACT-334441; lysosphingolipid receptor agonist – Actelion; S1P1 receptor modulator – Actelion; Second selective S1P1 receptor agonist – Actelion; Sphingosine 1 phosphate receptor modulators – Actelion; Sphingosine 1-phosphate receptor 1 agonists – Actelion
- Mechanism of Action Lysosphingolipid receptor agonists
- Highest Development Phases
- Phase I/II Systemic lupus erythematosus
Most Recent Events
- 09 Jun 2016 Actelion terminates a phase I drug interaction trial for Systemic lupus erythematosus (In volunteers) in France (NCT02479204)
- 22 Dec 2015 Phase-I/II clinical trials in Systemic lupus erythematosus in Ukraine, Belarus (PO) (NCT02472795)
- 24 Sep 2015 Phase-I/II clinical trials in Systemic lupus erythematosus in USA (PO) (NCT02472795)
| # | Nct Number | Title | Recruitment | Conditions | Interventions | Phase | |
|---|---|---|---|---|---|---|---|
| 1 | NCT02472795 | Clinical Study to Investigate the Biological Activity, Safety, Tolerability, and Pharmacokinetics of ACT-334441 in Subjects With Systemic Lupus Erythematosus | Recruiting | Systemic Lupus Erythematosus | Drug: ACT-334441|Drug: Placebo | Phase 2 Actelion | |
| 2 | NCT02479204 | Drug Interaction Study of ACT-334441 With Cardiovascular Medications in Healthy Subjects | Suspended | Healthy Subjects | Drug: ACT-334441 2 mg|Drug: ACT-334441 4 mg|Drug: placebo|Drug: atenolol|Drug: diltiazem ER | Phase 1 Actelion |
The human immune system is designed to defend the body against foreign micro-organisms and substances that cause infection or disease. Complex regulatory mechanisms ensure that the immune response is targeted against the intruding substance or organism and not against the host. In some cases, these control mechanisms are unregulated and autoimmune responses can develop. A consequence of the uncontrolled inflammatory response is severe organ, cell, tissue or joint damage. With current treatment, the whole immune system is usually suppressed and the body’s ability to react to infections is also severely compromised. Typical drugs in this class include azathioprine, chlorambucil, cyclophosphamide, cyclosporin, or methotrexate. Corticosteroids which reduce inflammation and suppress the immune response, may cause side effects when used in long term treatment. Nonsteroidal anti-inflammatory drugs (NSAIDs) can reduce pain and inflammation, however, they exhibit considerable side effects. Alternative treatments include agents that activate or block cytokine signaling.
Orally active compounds with immunomodulating properties, without compromising immune responses and with reduced side effects would significantly improve current treatments of uncontrolled inflammatory diseases.
In the field of organ transplantation the host immune response must be suppressed to prevent organ rejection. Organ transplant recipients can experience some rejection even when they are taking immunosuppressive drugs. Rejection occurs most frequently in the first few weeks after transplantation, but rejection episodes can also happen months or even years after transplantation. Combinations of up to three or four medications are commonly used to give maximum protection against rejection while minimizing side effects. Current standard drugs used to treat the rejection of transplanted organs interfere with discrete intracellular pathways in the activation of T-type or B-type white blood cells. Examples of such drugs are cyclosporin, daclizumab, basiliximab, everolimus, or FK506, which interfere with cytokine release or signaling; azathioprine or leflunomide, which inhibit nucleotide synthesis; or 15-deoxyspergualin, an inhibitor of leukocyte differentiation.
The beneficial effects of broad immunosuppressive therapies relate to their effects; however, the generalized immunosuppression which these drugs produce diminishes the immune system’s defense against infection and malignancies. Furthermore, standard immunosuppressive drugs are often used at high dosages and can cause or accelerate organ damage.
SYNTHESIS
PATENT
https://www.google.com/patents/WO2011007324A1?cl=zh
The human immune system is designed to defend the body against foreign microorganisms and substances that cause infection or disease. Complex regulatory mechanisms ensure that the immune response is targeted against the intruding substance or organism and not against the host. In some cases, these control mechanisms are unregulated and autoimmune responses can develop. A consequence of the uncontrolled inflammatory response is severe organ, cell, tissue or joint damage. With current treatment, the whole immune system is usually suppressed and the body’s ability to react to infections is also severely compromised. Typical drugs in this class include azathioprine, chlorambucil, cyclophosphamide, cyclosporin, or methotrexate. Corticosteroids which reduce inflammation and suppress the immune response, may cause side effects when used in long term treatment. Nonsteroidal anti-inflammatory drugs (NSAIDs) can reduce pain and inflammation, however, they exhibit considerable side effects. Alternative treatments include agents that activate or block cytokine signaling.
Orally active compounds with immunomodulating properties, without compromising immune responses and with reduced side effects would significantly improve current treatments of uncontrolled inflammatory diseases.
In the field of organ transplantation the host immune response must be suppressed to prevent organ rejection. Organ transplant recipients can experience some rejection even when they are taking immunosuppressive drugs. Rejection occurs most frequently in the first few weeks after transplantation, but rejection episodes can also happen months or even years after transplantation. Combinations of up to three or four medications are commonly used to give maximum protection against rejection while minimizing side effects. Current standard drugs used to treat the rejection of transplanted organs interfere with discrete intracellular pathways in the activation of T-type or B-type white blood cells. Examples of such drugs are cyclosporin, daclizumab, basiliximab, everolimus, or FK506, which interfere with cytokine release or signaling; azathioprine or leflunomide, which inhibit nucleotide synthesis; or 15-deoxyspergualin, an inhibitor of leukocyte differentiation.
The beneficial effects of broad immunosuppressive therapies relate to their effects; however, the generalized immunosuppression which these drugs produce diminishes the immune system’s defense against infection and malignancies. Furthermore, standard immunosuppressive drugs are often used at high dosages and can cause or accelerate organ damage.
Description of the invention
The present invention provides novel compounds of Formula (I) that are agonists for the G protein-coupled receptor S1 P1/EDG1 and have a powerful and long-lasting immunomodulating effect which is achieved by reducing the number of circulating and infiltrating T- and B-lymphocytes, without affecting their maturation, memory, or expansion. The reduction of circulating T- / B-lymphocytes as a result of S1 P1/EDG1 agonism, possibly in combination with the observed improvement of endothelial cell layer function associated with S1 P1/EDG1 activation, makes such compounds useful to treat uncontrolled inflammatory diseases and to improve vascular functionality. Prior art document WO 2008/029371 discloses compounds that act as S1 P1/EDG1 receptor agonists and show an immunomodulating effect as described above. Unexpectedly, it has been found that the compounds of the present invention have a reduced potential to constrict airway tissue/vessels when compared to compounds of the prior art document WO 2008/029371. The compounds of the present invention therefore demonstrate superiority with respect to their safety profile, e.g. a lower risk of bronchoconstriction.
Examples of WO 2008/029371 , which are considered closest prior art analogues are shown in Figure 1.
Figure 1 : Structure of Examples of prior art document WO 2008/029371 , which are considered closest analogues to the compounds of the present invention.
The data on the constriction of rat trachea rings compiled in Table 1 illustrate the superiority of the compounds of the present invention as compared to compounds of prior art document WO 2008/029371.
For instance, the compounds of Example 1 and 6 of the present invention show a significantly reduced potential to constrict rat trachea rings when compared to the compounds of prior art Examples 222 and 226 of WO 2008/029371 , respectively. Furthermore, the compounds of Example 1 and 6 of the present invention also show a reduced potential to constrict rat trachea rings when compared to the compounds of prior art Examples 196 and 204 of WO 2008/029371 , respectively. These data demonstrate that compounds wherein R1 represents 3-pentyl and R2 represents methoxy are superior compared to the closest prior art compounds of WO 2008/029371 , i.e. the compounds wherein R1 represents an isobutyl and R2 represents methoxy or wherein R1represents methyl and R2 represents 3-pentyl. Moreover, also the compound of Example 16 of the present invention, wherein R1 is 3-methyl-but-1-yl and R2 is methoxy, exhibits a markedly reduced potential to constrict rat trachea rings when compared to its closest analogue prior art Example 226 of WO 2008/029371 wherein R1 is isobutyl and R2 is methoxy.
The unexpected superiority of the compounds of the present invention is also evident from the observation that the compounds of Example 2 and 7 of the present invention show a markedly reduced potential to constrict rat trachea rings when compared to the compounds of prior art Examples 229 and 233 of WO 2008/029371 , respectively. This proves that compounds wherein R1represents cyclopentyl and R2 represents methoxy are superior compared to the closest prior art compounds of WO 2008/029371 , i.e. the compounds wherein R1 represents methyl and R2 represents cyclopentyl.
Also, the compound of Example 3 of the present invention exhibits the same low potential to constrict rat trachea rings as its S-enantiomer, i.e. the compound of Example 2 of the present invention, indicating that the configuration at this position has no significant effect on trachea constriction. Furthermore, also Example 21 of the present invention exhibits the same low potential to constrict rat trachea rings as present Example 2, which differs from Example 21 only by the linker A (forming a 5-pyridin-4-yl-[1 ,2,4]oxadiazole instead of a 3- pyridin-4-yl-[1 ,2,4]oxadiazole). This indicates that also the nature of the oxadiazole is not critical regarding trachea constriction.
Table 1 : Rat trachea constriction in % of the constriction induced by 50 mM KCI. n.d. = not determined. For experimental details and further data see Example 33.
result obtained at a compound concentration of 300 nM.
The compounds of the present invention can be utilized alone or in combination with standard drugs inhibiting T-cell activation, to provide a new immunomodulating therapy with a reduced propensity for infections when compared to standard immunosuppressive therapy. Furthermore, the compounds of the present invention can be used in combination with reduced dosages of traditional immunosuppressant therapies, to provide on the one hand effective immunomodulating activity, while on the other hand reducing end organ damage associated with higher doses of standard immunosuppressive drugs. The observation of improved endothelial cell layer function associated with S1 P1/EDG1 activation provides additional benefits of compounds to improve vascular function.
The nucleotide sequence and the amino acid sequence for the human S1 P1/EDG1 receptor are known in the art and are published in e.g.: HIa, T., and Maciag, T., J. Biol
Chem. 265 (1990), 9308-9313; WO 91/15583 published 17 October 1991 ; WO 99/46277 published 16 September 1999. The potency and efficacy of the compounds of Formula (I) are assessed using a GTPγS assay to determine EC5O values and by measuring the circulating lymphocytes in the rat after oral administration, respectively (see in experimental part). i) In a first embodiment, the invention relates to pyridine compounds of the Formula (I),
Formula (I)
PATENT
WO 2013175397
https://www.google.com/patents/WO2013175397A1?cl=en
Pyridine-4-yl derivatives of formula (PD),
Formula (PD) A represents
(the asterisks indicate the bond that is linked to the pyridine group of Formula (PD));
Ra represents 3-pentyl, 3-methyl-but-1-yl, cyclopentyl, or cyclohexyl;
Rb represents methoxy;
Rc represents 2,3-dihydroxypropoxy, -OCH2-CH(OH)-CH2-NHCO-CH2OH,
-OCH2-CH(OH)-CH2N(CH3)-CO-CH2OH, -NHS02CH3, or -NHS02CH2CH3; and
Rd represents ethyl or chloro.)
disclosed in WO201 1007324, have immunomodulating activity through their S1 P1/EDG1 receptor agonistic activity. Therefore, those pyridine-4-yl derivatives are useful for prevention and / or treatment of diseases or disorders associated with an activated immune system, including rejection of transplanted organs such as kidney, liver, heart, lung, pancreas, cornea, and skin; graft-versus-host diseases brought about by stem cell transplantation; autoimmune syndromes including rheumatoid arthritis, multiple sclerosis, inflammatory bowel diseases such as Crohn’s disease and ulcerative colitis, psoriasis, psoriatic arthritis, thyroiditis such as Hashimoto’s thyroiditis, uveo-retinitis; atopic diseases such as rhinitis, conjunctivitis, dermatitis; asthma; type I diabetes; post-infectious autoimmune diseases including rheumatic fever and post-infectious glomerulonephritis; solid cancers and tumor metastasis. 2-Cyclopentyl-6-methoxy-isonicotinic acid, which is also disclosed in WO201 1007324, is a useful intermediate for the synthesis of the pyridine-4-yl derivatives of formula (PD), wherein Ra is a cyclopentyl group.
In the process described in WO201 1007324, 2-cyclopentyl-6-methoxy-isonicotinic acid was prepared according to the following reaction scheme 1 :
Compound D Compound E
Rieke Zinc: cyclopentylzinc bromide;
PdCI2(dppf)dcm: 1 ,1 ‘-Bis(diphenylphosphino)ferrocene-palladium(ll)dichloride
dichloromethane complex
However, the abovementioned process has drawbacks for larger scale, i.e. industrial scale synthesis of 2-cyclopentyl-6-methoxy-isonicotinic acid, for the following reasons:
a) The commercially available starting material, 2,6-dichloro-isonicotinic acid (Compound A) is expensive.
b) The conversion of Compound C to Compound D is cost-intensive. The reaction has to be performed under protective atmosphere with expensive palladium catalysts and highly reactive and expensive Rieke zinc complex. Such synthesis steps are expensive to scale up and it was therefore highly desired to find alternative synthesis methods.
Even though Goldsworthy, J. Chem. Soc. 1934, 377-378 discloses the preparation of 1 -cyclopentylethanone, which is a key building block in the new process of the present invention, by using ethyl 1 -acetoacetate as a starting material, this synthesis was far from being suitable in an industrial process. The reported yield was low (see also under “Referential Examples” below). Scheme 2
ethyl 1 -acetylcyclo- 1-cyclopentyl- pentanecarboxylate ethanone
Besides the early work by Goldsworthy there are several recent examples for the preparation of 1 -cyclopentylethanone described in the literature. Such examples include:
1 ) Addition of methyl lithium to a N-cyclopentanecarbonyl-N,0-dimethylhydroxylamine at -78°C in a yield of 77%. US2006/199853 A1 , 2006 and US2006/223884 A1 , 2006.
2) Addition of methyl lithium to a cyclopentyl carboxylic acid in diethylether at -78°C in a yield of 81 %. J. Am. Chem. Soc, 1983, 105, 4008-4017.
3) Addition of methylmagnesiumbromide to cyclopentanecarbonitrile.
Bull. Soc. Chim. Fr., 1967, 3722-3729.
4) Oxidation of 1 -cyclopentylethanol with chromtrioxide. US5001 140 A1 , 1991.
WO2009/71707 A1 , 2009.
5) Addition of cyclopentylmagnesium bromide to acetic anhydride at -78 °C with a yield of 54%. WO2004/74270 A2, 2004.
6) Synthesis of 1-cyclopentylethanone in 5 steps from cyclopentanone. Zhang, Pang; Li, Lian-chu, Synth. Commun., 1986, 16, 957-966.
However, the processes described in the above-listed publications are not efficient for scale-up since they require cryogenic temperatures, expensive starting materials, toxic reagents or many steps. The lack of an efficient process to manufacture 1 -cyclopentylethanone is further also mirrored by the difficulty in sourcing this compound on kilogram scale for a reasonable price and delivery time. Therefore, the purpose of the present invention is to provide a new, efficient and cost effective process for the preparation of 2-cyclopentyl-6-methoxy-isonicotinic acid, which is suitable for industrial scale synthesis.
Patent
Disclosed in WO2011007324, have immunomodulating activity through their S1P1/EDG1 receptor agonistic activity. Therefore, those pyridine-4-yl derivatives are useful for prevention and/or treatment of diseases or disorders associated with an activated immune system, including rejection of transplanted organs such as kidney, liver, heart, lung, pancreas, cornea, and skin; graft-versus-host diseases brought about by stem cell transplantation; autoimmune syndromes including rheumatoid arthritis, multiple sclerosis, inflammatory bowel diseases such as Crohn’s disease and ulcerative colitis, psoriasis, psoriatic arthritis, thyroiditis such as Hashimoto’s thyroiditis, uveo-retinitis; atopic diseases such as rhinitis, conjunctivitis, dermatitis; asthma; type I diabetes; post-infectious autoimmune diseases including rheumatic fever and post-infectious glomerulonephritis; solid cancers and tumor metastasis. 2-Cyclopentyl-6-methoxy-isonicotinic acid, which is also disclosed in WO2011007324, is a useful intermediate for the synthesis of the pyridine-4-yl derivatives of formula (PD), wherein Ra is a cyclopentyl group.
 Rieke Zinc: cyclopentylzinc bromide;
Rieke Zinc: cyclopentylzinc bromide;
PdCl2(dppf)dcm: 1,1′-Bis(diphenylphosphino)ferrocene-palladium(II)dichloride dichloromethane complex


EXAMPLES
Example 1a
1-Cyclopentylethanone
Example 1 b
1-Cyclopentylethanone
Example 1c
1-Cyclopentylethanone
Example 1d
1-Cyclopentylethanone
Example 1e
1-Cyclopentylethanone
Example 1f
Tert-butyl 1-acetylcyclopentanecarboxylate
Example 1 g
Tert-butyl 1-acetylcyclopentanecarboxylate
Example 2
2-Cyclopentyl-6-hydroxyisonicotinic acid
Example 3
Methyl 2-cyclopentyl-6-hydroxyisonicotinate
Example 4a
Methyl 2-chloro-6-cyclopentylisonicotinate
Example 4b
Methyl 2-chloro-6-cyclopentylisonicotinate
Example 5
2-Cyclopentyl-6-methoxyisonicotinic acid
Example 6
Ethyl 4-cyclopentyl-2,4-dioxobutanoate
Example 7
Ethyl 3-cyano-6-cyclopentyl-2-hydroxyisonicotinate
REFERENTIAL EXAMPLES
Referential Example 1
Referential Example 2
PATENT
https://www.google.com/patents/US8658675
Martin Bolli, Cyrille Lescop, Boris Mathys,Keith Morrison, Claus Mueller, Oliver Nayler,Beat Steiner,
novel compounds of Formula (I) that are agonists for the G protein-coupled receptor S1P1/EDG1 and have a powerful and long-lasting immunomodulating effect which is achieved by reducing the number of circulating and infiltrating T- and B-lymphocytes, without affecting their maturation, memory, or expansion. The reduction of circulating T-/B-lymphocytes as a result of S1P1/EDG1 agonism, possibly in combination with the observed improvement of endothelial cell layer function associated with S1P1/EDG1 activation, makes such compounds useful to treat uncontrolled inflammatory diseases and to improve vascular functionality. Prior art document WO 2008/029371 discloses compounds that act as S1P1/EDG1 receptor agonists and show an immunomodulating effect as described above. Unexpectedly, it has been found that the compounds of the present invention have a reduced potential to constrict airway tissue/vessels when compared to compounds of the prior art document WO 2008/029371. The compounds of the present invention therefore demonstrate superiority with respect to their safety profile, e.g. a lower risk of bronchoconstriction.
Examples of WO 2008/029371, which are considered closest prior art analogues are shown in FIG. 1.
The data on the constriction of rat trachea rings compiled in Table 1 illustrate the superiority of the compounds of the present invention as compared to compounds of prior art document WO 2008/029371.
For instance, the compounds of Example 1 and 6 of the present invention show a significantly reduced potential to constrict rat trachea rings when compared to the compounds of prior art Examples 222 and 226 of WO 2008/029371, respectively. Furthermore, the compounds of Example 1 and 6 of the present invention also show a reduced potential to constrict rat trachea rings when compared to the compounds of prior art Examples 196 and 204 of WO 2008/029371, respectively. These data demonstrate that compounds wherein R1 represents 3-pentyl and R2represents methoxy are superior compared to the closest prior art compounds of WO 2008/029371, i.e. the compounds wherein R1 represents an isobutyl and R2represents methoxy or wherein R1 represents methyl and R2 represents 3-pentyl. Moreover, also the compound of Example 16 of the present invention, wherein R1is 3-methyl-but-1-yl and R2 is methoxy, exhibits a markedly reduced potential to constrict rat trachea rings when compared to its closest analogue prior art Example 226 of WO 2008/029371 wherein R1 is isobutyl and R2 is methoxy.
The unexpected superiority of the compounds of the present invention is also evident from the observation that the compounds of Example 2 and 7 of the present invention show a markedly reduced potential to constrict rat trachea rings when compared to the compounds of prior art Examples 229 and 233 of WO 2008/029371, respectively. This proves that compounds wherein R1 represents cyclopentyl and R2 represents methoxy are superior compared to the closest prior art compounds of WO 2008/029371, i.e. the compounds wherein R1represents methyl and R2 represents cyclopentyl.
Preparation of Intermediates2-Chloro-6-methyl-isonicotinic acid
The title compound and its ethyl ester are commercially available.
2-(1-Ethyl-propyl)-6-methoxy-isonicotinic acid
a) To a solution of 2,6-dichloroisonicotinic acid (200 g, 1.04 mol) in methanol (3 L), 32% aq. NaOH (770 mL) is added. The stirred mixture becomes warm (34° C.) and is then heated to 70° C. for 4 h before it is cooled to rt. The mixture is neutralised by adding 32% aq. HCl (100 mL) and 25% aq. HCl (700 mL). The mixture is stirred at rt overnight. The white precipitate that forms is collected, washed with methanol and dried. The filtrate is evaporated and the residue is suspended in water (200 mL). The resulting mixture is heated to 60° C. Solid material is collected, washed with water and dried. The combined crops give 2-chloro-6-methoxy-isonicotinic acid (183 g) as a white solid; LC-MS: tR=0.80 min, [M+1]+=187.93.
b) To a suspension of 2-chloro-6-methoxy-isonicotinic acid (244 g, 1.30 mol) in methanol (2.5 L), H2SO4 (20 mL) is added. The mixture is stirred at reflux for 24 h before it is cooled to 0° C. The solid material is collected, washed with methanol (200 mL) and water (500 mL) and dried under HV to give 2-chloro-6-methoxy-isonicotinic acid methyl ester (165 g) as a white solid; LC-MS: tR=0.94 min, [M+1]+=201.89.
c) Under argon, Pd(dppf) (3.04 g, 4 mmol) is added to a solution of 2-chloro-6-methoxy-isonicotinic acid methyl ester (50 g, 0.248 mol) in THF (100 mL). A 0.5 M solution of 3-pentylzincbromide in THF (550 mL) is added via dropping funnel. Upon complete addition, the mixture is heated to 85° C. for 18 h before it is cooled to rt. Water (5 mL) is added and the mixture is concentrated. The crude product is purified by filtration over silica gel (350 g) using heptane:EA 7:3 to give 2-(1-ethyl-propyl)-6-methoxy-isonicotinic acid methyl ester (53 g) as a pale yellow oil; 1H NMR (CDCl3): δ0.79 (t, J=7.5 Hz, 6H), 1.63-1.81 (m, 4H), 2.47-2.56 (m, 1H), 3.94 (s, 3H), 3.96 (s, 3H), 7.12 (d, J=1.0 Hz, 1H), 7.23 (d, J=1.0 Hz, 1H).
d) A solution of 2-(1-ethyl-propyl)-6-methoxy-isonicotinic acid methyl ester (50 g, 0.211 mol) in ethanol (250 mL), water (50 mL) and 32% aq. NaOH (50 mL) is stirred at 80° C. for 1 h. The mixture is concentrated and the residue is dissolved in water (200 mL) and extracted with TBME. The org. phase is separated and washed once with water (200 mL). The TBME phase is discarded. The combined aq. phases are acidified by adding 25% aq. HCl and then extracted with EA (400+200 mL). The combined org. extracts are concentrated. Water (550 mL) is added to the remaining residue. The mixture is heated to 70° C., cooled to rt and the precipitate that forms is collected and dried to give the title compound (40.2 g) as a white solid; LC-MS: tR=0.95 min, [M+1]+=224.04; 1H NMR (D6-DMSO): δ 0.73 (t, J=7.3 Hz, 6H), 1.59-1.72 (m, 4H), 2.52-2.58 (m, 1H), 3.88 (s, 3H), 7.00 (d, J=1.0 Hz, 1H), 7.20 (d, J=1.0 Hz, 1H).
2-Methoxy-6-(3-methyl-butyl)-isonicotinic acid
The title compound is prepared in analogy to 2-(1-ethyl-propyl)-6-methoxy-isonicotinic acid; LC-MS: tR=0.94 min, [M+1]+=224.05; 1H NMR (D6-DMSO): δ 0.92 (d, J=5.8 Hz, 6H), 1.54-1.62 (m, 3H), 2.70-2.76 (m, 2H), 3.88 (s, 3H), 6.99 (s, 1H), 7.25 (s, 1H), 13.52 (s).
2-Cyclopentyl-6-methoxy-isonicotinic acid
The title compound is prepared in analogy to 2-(1-ethyl-propyl)-6-methoxy-isonicotinic acid; LC-MS: tR=0.93 min, [M+1]+=222.02; 1H NMR (CDCl3): δ 1.68-1.77 (m, 2H), 1.81-1.90 (m, 4H), 2.03-2.12 (m, 2H), 3.15-3.25 (m, 1H), 3.99 (s, 3H), 7.18 (d, J=1.0 Hz, 1H), 7.35 (d, J=0.8 Hz, 1H).
2-Cyclohexyl-6-methoxy-isonicotinic acid
The title compound is prepared in analogy to 2-(1-ethyl-propyl)-6-methoxy-isonicotinic acid; LC-MS: tR=0.98 min, [M+1]+=236.01; 1H NMR (D6-DMSO): δ 1.17-1.29 (m, 1H), 1.31-1.43 (m, 2H), 1.44-1.55 (m, 2H), 1.67-1.73 (m, 1H), 1.76-1.83 (m, 2H), 1.84-1.92 (m, 2H), 2.66 (tt, J=11.3, 3.3 Hz, 1H), 3.88 (s, 3H), 7.00 (d, J=1.0 Hz, 1H), 7.23 (d, J=1.0 Hz, 1H).
2-Cyclopentyl-N-hydroxy-6-methoxy-isonicotinamidine
a) A solution of 2-cyclopentyl-6-methoxy-isonicotinic acid methyl ester (3.19 g, 13.6 mmol) in 7 N NH3 in methanol (50 mL) is stirred at 60° C. for 18 h. The solvent is removed in vacuo and the residue is dried under HV to give crude 2-cyclopentyl-6-methoxy-isonicotinamide (3.35 g) as a pale yellow solid; LC-MS**: tR=0.57 min, [M+1]+=221.38.
b) Pyridine (8.86 g, 91.3 mmol) is added to a solution of 2-cyclopentyl-6-methoxy-isonicotinamide (3.35 g, 15.2 mmol) in DCM (100 mL). The mixture is cooled to 0° C. before trifluoroacetic acid anhydride (9.58 g, 45.6 mmol) is added portionwise. The mixture is stirred at 0° C. for 1 h before it is diluted with DCM (100 mL) and washed with sat. aq. NaHCO3 solution (100 mL) and brine (100 mL). The separated org. phase is dried over MgSO4, filtered and concentrated. The crude product is purified by CC on silica gel eluting with heptane:EA 9:1 to give 2-cyclopentyl-6-methoxy-isonicotinonitrile (2.09 g) as pale yellow oil; LC-MS**: tR=0.80 min, [M+1]+=not detectable; 1H NMR (D6-DMSO): δ 1.61-1.82 (m, 6H), 1.94-2.03 (m, 2H), 3.16 (quint, J=7.8 Hz, 1H), 3.89 (s, 3H), 7.15 (s, 1H), 7.28 (s, 1H).
c) To a solution of 2-cyclopentyl-6-methoxy-isonicotinonitrile (2.09 g, 10.3 mmol) in methanol (100 mL), hydroxylamine hydrochloride (2.15 g, 31.0 mmol) and NaHCO3 (3.04 g, 36.2 mmol) are added. The mixture is stirred at 60° C. for 18 h before it is filtered and the filtrate is concentrated. The residue is dissolved in EA (300 mL) and washed with water (30 mL). The washings are extracted back with EA (4×100 mL) and DCM (4×100 mL). The combined org. extracts are dried over MgSO4, filtered, concentrated and dried under HV to give the title compound (2.74 g) as a white solid; LC-MS**: tR=0.47 min, [M+1]+=236.24; 1H NMR (D6-DMSO): δ 1.61-1.82 (m, 6H), 1.92-2.01 (m, 2H), 3.04-3.13 (m, 1H), 3.84 (s, 3H), 5.90 (s, 2H), 6.86 (s, 1H), 7.13 (s, 1H), 9.91 (s, 1H).
2-Cyclopentyl-6-methoxy-isonicotinic acid hydrazide
a) To a solution of 2-cyclopentyl-6-methoxy-isonicotinic acid (2.00 g, 9.04 mmol), hydrazinecarboxylic acid benzyl ester (1.50 g, 9.04 mmol) and DIPEA (2.34 g, 18.1 mmol) in DCM (40 mL), TBTU (3.19 g, 9.94 mmol) is added. The mixture is stirred at rt for 2 h before it is diluted with EA (250 mL), washed twice with sat. aq. NaHCO3 solution (150 mL) followed by brine (100 mL), dried over MgSO4, filtered and concentrated. The crude product is purified by CC on silica gel eluting with heptane:EA 4:1 to give N′-(2-cyclopentyl-6-methoxy-pyridine-4-carbonyl)-hydrazinecarboxylic acid benzyl ester (2.74 g) as pale yellow oil; LC-MS**: tR=0.74 min, [M+1]+=369.69; 1H NMR (D6-DMSO): δ 1.62-1.83 (m, 6H), 1.95-2.05 (m, 2H), 3.10-3.21 (m, 1H), 3.88 (s, 3H), 5.13 (s, 2H), 6.97 (s, 1H), 7.23 (s, 1H), 7.28-7.40 (m, 5H), 9.45 (s, 1H), 10.52 (s, 1H).
b) Pd/C (500 mg, 10% Pd) is added to a solution of N′-(2-cyclopentyl-6-methoxy-pyridine-4-carbonyl)-hydrazinecarboxylic acid benzyl ester (2.74 g, 7.42 mmol) in THF (50 mL) and methanol (50 mL). The mixture is stirred at rt under 1 bar of H2 for 25 h. The catalyst is removed by filtration and the filtrate is concentrated and dried under HV to give the title compound (1.58 g) as an off-white solid; LC-MS**: tR=0.51 min, [M+1]+=236.20; 1H NMR (D6-DMSO): δ 1.60-1.82 (m, 6H), 1.94-2.03 (m, 2H), 3.08-3.19 (m, 1H), 3.86 (s, 3H), 4.56 (s br, 2H), 6.93 (d, J=1.0 Hz, 1H), 7.20 (d, J=1.0 Hz, 1H), 9.94 (s, 1H).
3-Ethyl-4-hydroxy-5-methyl-benzonitrile
The title compound is prepared from 3-ethyl-4-hydroxy-5-methyl-benzaldehyde following literature procedures (A. K. Chakraborti, G. Kaur, Tetrahedron 55 (1999) 13265-13268); LC-MS: tR=0.90 min; 1H NMR (CDCl3): δ1.24 (t, J=7.6 Hz, 3H), 2.26 (s, 3H), 2.63 (q, J=7.6 Hz, 2H), 5.19 (s, 1H), 7.30 (s, 2H).
3-Chloro-4-hydroxy-5-methyl-benzonitrile
The title compound is prepared from commercially available 2-chloro-6-methyl-phenol in analogy to literature procedures (see 3-ethyl-4-hydroxy-5-methyl-benzonitrile); LC-MS: tR=0.85 min. 1H NMR (CDCl3): δ2.33 (s, 3H), 6.10 (s, 1H), 7.38 (s, 1H), 7.53 (d, J=1.8 Hz, 1H).
3-Ethyl-4,N-dihydroxy-5-methyl-benzamidine
The title compound is prepared from 3-ethyl-4-hydroxy-5-methyl-benzonitrile or from commercially available 2-ethyl-6-methyl-phenol following literature procedures (G. Trapani, A. Latrofa, M. Franco, C. Altomare, E. Sanna, M. Usala, G. Biggio, G. Liso, J. Med. Chem. 41 (1998) 1846-1854; A. K. Chakraborti, G. Kaur, Tetrahedron 55 (1999) 13265-13268; E. Meyer, A. C. Joussef, H. Gallardo, Synthesis 2003, 899-905); LC-MS: tR=0.55 min; 1H NMR (D6-DMSO): δ 9.25 (s br, 1H), 7.21 (s, 2H), 5.56 (s, 2H), 2.55 (q, J=7.6 Hz, 2H), 2.15 (s, 3H), 1.10 (t, J=7.6 Hz, 3H).
3-Chloro-4,N-dihydroxy-5-methyl-benzamidine
The title compound is prepared from commercially available 2-chloro-6-methyl-phenol in analogy to literature procedures (e.g. B. Roth et al. J. Med. Chem. 31 (1988) 122-129; and literature cited for 3-ethyl-4,N-dihydroxy-5-methyl-benzamidine); 3-chloro-4-hydroxy-5-methyl-benzaldehyde: LC-MS: tR=0.49 min, [M+1]+=201.00; 1H NMR 82.24 (s, 2H), 2.35 (s, 4H), 5.98 (s br, 1H), 7.59 (d, J=1.8 Hz, 1H), 7.73 (d, J=1.8 Hz, 1H), 9.80 (s, 1H); 3-chloro-4,N-dihydroxy-5-methyl-benzamidine: 1H NMR (D6-DMSO): δ 2.21 (s, 3H), 5.72 (s br, 2H), 7.40 (s, 1H), 7.48 (s, 1H), 9.29 (s br, 1H), 9.48 (s br, 1H).
(R)-4-(2,2-Dimethyl-[1,3]dioxolan-4-ylmethoxy)-3-ethyl-N-hydroxy-5-methyl-benzamidine
a) To a solution of 3-ethyl-4-hydroxy-5-methyl-benzonitrile (2.89 g, 17.9 mmol) in THF (80 mL), (R)-(2,2-dimethyl-[1,3]dioxolan-4-yl)methanol (2.84 g, 21.5 mmol) followed by triphenylphosphine (5.81 g, 21.5 mmol) is added. The mixture is cooled with an ice-bath before DEAD (9.36 g, 21.5 mmol) is added dropwise. The mixture is stirred at rt for 1 h, the solvent is removed in vacuo and the residue is purified by CC on silica gel eluting with heptane:EA 85:15 to give (R)-4-(2,2-dimethyl-[1,3]dioxolan-4-ylmethoxy)-3-ethyl-5-methyl-benzonitrile (4.45 g) as a pale yellow oil; LC-MS**: tR=0.75 min, [M+1]+=not detected; 1H NMR (CDCl3): δ1.25 (t, J=7.5 Hz, 3H), 1.44 (s, 3H), 1.49 (s, 3H), 2.34 (s, 3H), 2.65-2.77 (m, 2H), 3.80-3.90 (m, 2H), 3.94-4.00 (m, 1H), 4.21 (t, J=7.3 Hz, 1H), 4.52 (quint, J=5.8 Hz, 1H), 7.35 (s, 1H), 7.38 (s, 1H).
b) To a mixture of (R)-4-(2,2-dimethyl-[1,3]dioxolan-4-ylmethoxy)-3-ethyl-5-methyl-benzonitrile (4.45 g, 16.2 mmol) and NaHCO3 (4.75 g, 56.6 mmol) in methanol (30 mL), hydroxylamine hydrochloride (3.37 g, 48.5 mmol) is added. The mixture is stirred at 60° C. for 18 h before it is filtered and the solvent of the filtrate is removed in vacuo. The residue is dissolved in EA and washed with a small amount of water and brine. The org. phase is separated, dried over MgSO4, filtered, concentrated and dried to give the title compound (5.38 g) as a white solid; LC-MS**: tR=0.46 min, [M+1]+=309.23; 1H NMR (D6-DMSO): δ 1.17 (t, J=7.5 Hz, 3H), 1.33 (s, 3H), 1.38 (s, 3H), 2.25 (s, 3H), 2.57-2.69 (m, 2H), 3.73-3.84 (m, 3H), 4.12 (t, J=7.0 Hz, 1H), 4.39-4.45 (m, 1H), 5.76 (s br, 2H), 7.34 (s, 1H), 7.36 (s, 1H), 9.47 (s, 1H).
(R)-3-Chloro-4-(2,2-dimethyl-[1,3]dioxolan-4-ylmethoxy)-N-hydroxy-5-methyl-benzamidine
The title compound is obtained as a colorless oil (1.39 g) in analogy to (R)-4-(2,2-dimethyl-[1,3]dioxolan-4-ylmethoxy)-3-ethyl-N-hydroxy-5-methyl-benzamidine starting from 3-chloro-4-hydroxy-5-methyl-benzonitrile and L-α,β-isopropyliden glycerol; LC-MS: tR=0.66 min, [M+H]+=314.96.
(S)-4-(3-Amino-2-hydroxypropoxy)-3-ethyl-5-methylbenzonitrile
a) To a solution of 3-ethyl-4-hydroxy-5-methyl-benzonitrile (5.06 g, 31.4 mmol) in THF (80 mL), PPh3 (9.06 g, 34.5 mmol) and (R)-glycidol (2.29 mL, 34.5 mmol) are added. The mixture is cooled to 0° C. before DEAD in toluene (15.8 mL, 34.5 mmol) is added. The mixture is stirred for 18 h while warming up to rt. The solvent is evaporated and the crude product is purified by CC on silica gel eluting with heptane:EA 7:3 to give 3-ethyl-5-methyl-4-oxiranylmethoxy-benzonitrile (5.85 g) as a yellow oil; LC-MS: tR=0.96 min; [M+42]+=259.08.
b) The above epoxide is dissolved in 7 N NH3 in methanol (250 mL) and the solution is stirred at 65° C. for 18 h. The solvent is evaporated to give crude (S)-4-(3-amino-2-hydroxypropoxy)-3-ethyl-5-methylbenzonitrile (6.23 g) as a yellow oil; LC-MS: tR=0.66 min; [M+1]+=235.11.
N—((S)-3-[2-Ethyl-4-(N-hydroxycarbamimidoyl)-6-methyl-phenoxy]-2-hydroxy-propyl)-2-hydroxy-acetamide
a) To a solution of (S)-4-(3-amino-2-hydroxypropoxy)-3-ethyl-5-methylbenzonitrile (6.23 g, 26.59 mmol) in THF (150 mL), glycolic acid (2.43 g, 31.9 mmol), HOBt (4.31 g, 31.9 mmol), and EDC hydrochloride (6.12 g, 31.9 mmol) are added. The mixture is stirred at rt for 18 h before it is diluted with sat. aq. NaHCO3 and extracted twice with EA. The combined org. extracts are dried over MgSO4, filtered and concentrated. The crude product is purified by CC with DCM containing 8% of methanol to give (S)—N-[3-(4-cyano-2-ethyl-6-methyl-phenoxy)-2-hydroxy-propyl]-2-hydroxy-acetamide (7.03 g) as a yellow oil; LC-MS: tR=0.74 min, [M+1]+=293.10; 1H NMR (CDCl3): δ 1.25 (t, J=7.5 Hz, 3H), 2.32 (s, 3H), 2.69 (q, J=7.5 Hz, 2H), 3.48-3.56 (m, 3H), 3.70-3.90 (m, 3H), 4.19 (s, br, 3H), 7.06 (m, 1H), 7.36 (s, 1H), 7.38 (s, 1H).
b) The above nitrile is converted to the N-hydroxy-benzamidine according to literature procedures (e.g. E. Meyer, A. C. Joussef, H. Gallardo, Synthesis 2003, 899-905); LC-MS: tR=0.51 min, [M+1]+=326.13; 1H NMR (D6-DMSO): δ 1.17 (t, J=7.4 Hz, 3H), 2.24 (s, 3H), 2.62 (q, J=7.4 Hz, 2H), 3.23 (m, 1H), 3.43 (m, 1H), 3.67 (m, 2H), 3.83 (s, 2H), 3.93 (m, 1H), 5.27 (s br, 1H), 5.58 (s br, 1H), 5.70 (s, 2H), 7.34 (s, 1H), 7.36 (s, 1H), 7.67 (m, 1H), 9.46 (s br, 1H).
(S)—N-(3-[2-Chloro-4-(N-hydroxycarbamimidoyl)-6-methyl-phenoxy]-2-hydroxy-propyl)-2-hydroxy-acetamide
The title compound is obtained as a beige wax (1.1 g) in analogy to N—((S)-3-[2-ethyl-4-(N-hydroxycarbamimidoyl)-6-methyl-phenoxy]-2-hydroxy-propyl)-2-hydroxy-acetamide starting from 3-chloro-4-hydroxy-5-methyl-benzonitrile; LC-MS: tR=0.48 min, [M+H]+=331.94.
3-Chloro-N-hydroxy-4-methanesulfonylamino-5-methyl-benzamidine
a) A mixture of 4-amino-3-chloro-5-methylbenzonitrile (155 mg, 930 μmol) and methanesulfonylchloride (2.13 g, 18.6 mmol, 1.44 mL) is heated under microwave conditions to 150° C. for 7 h. The mixture is cooled to rt, diluted with water and extracted with EA. The org. extract is dried over MgSO4, filtered and concentrated. The crude product is purified on prep. TLC using heptane:EA 1:1 to give N-(2-chloro-4-cyano-6-methyl-phenyl)-methanesulfonamide (105 mg) as an orange solid; LC-MS**: tR=0.48 min; 1H NMR (CDCl3): δ2.59 (s, 3H), 3.18 (s, 3H), 6.27 (s, 1H), 7.55 (d, J=1.3 Hz, 1H), 7.65 (d, J=1.5 Hz, 1H).
b) Hydroxylamine hydrochloride (60 mg, 858 μmol) and NaHCO3 (72 mg, 858 μmol) is added to a solution of N-(2-chloro-4-cyano-6-methyl-phenyl)-methanesulfonamide (105 mg, 429 μmol) in methanol (10 mL). The mixture is stirred at 65° C. for 18 h. The solvent is removed in vacuo and the residue is dissolved in a small volume of water (2 mL) and extracted three times with EA (15 mL). The combined org. extracts are dried over MgSO4, filtered, concentrated and dried to give the title compound (118 mg) as a white solid; LC-MS**: tR=0.19 min, [M+1]+=277.94; 1H NMR (CDCl3): δ2.57 (s, 3H), 3.13 (s, 3H), 6.21 (s, 1H), 7.49 (d, J=1.5 Hz, 1H), 7.63 (d, J=1.5 Hz).
3-Ethyl-N-hydroxy-4-methanesulfonylamino-5-methyl-benzamidine
a) In a 2.5 L three-necked round-bottom flask 2-ethyl-6-methyl aniline (250 g, 1.85 mol) is dissolved in DCM (900 mL) and cooled to 5-10° C. Bromine (310.3 g, 1.94 mol) is added over a period of 105 min such as to keep the temperature at 5-15° C. An aq. 32% NaOH solution (275 mL) is added over a period of 10 min to the greenish-grey suspension while keeping the temperature of the reaction mixture below 25° C. DCM (70 mL) and water (100 mL) are added and the phases are separated. The aq. phase is extracted with DCM (250 mL). The combined org. phases are washed with water (300 mL) and concentrated at 50° C. to afford the 4-bromo-2-ethyl-6-methyl-aniline (389 g) as a brown oil; 1H NMR (CDCl3): δ 1.27 (t, J=7.3 Hz, 3H), 2.18 (s, 3H), 2.51 (q, J=7.3 Hz, 2H), 3.61 (s br, 1H), 7.09 (s, 2H).
b) A double-jacketed 4 L-flask is charged with 4-bromo-2-ethyl-6-methyl-aniline (324 g, 1.51 mol), sodium cyanide (100.3 g, 1.97 mol), potassium iodide (50.2 g, 0.302 mol) and copper(I)iodide (28.7 g, 0.151 mol). The flask is evacuated three times and refilled with nitrogen. A solution of N,N′-dimethylethylenediamine (191.5 mL, 1.51 mol) in toluene (750 mL) is added. The mixture is heated to 118° C. and stirred at this temperature for 21 h. The mixture is cooled to 93° C. and water (1250 mL) is added to obtain a solution. Ethyl acetate (1250 mL) is added at 22-45° C. and the layers are separated. The org. phase is washed with 10% aq. citric acid (2×500 mL) and water (500 mL). The separated org. phase is evaporated to dryness to afford 4-amino-3-ethyl-5-methyl-benzonitrile (240 g) as a metallic black solid; 1H NMR (CDCl3): δ1.29 (t, J=7.5 Hz, 3H), 2.19 (s, 3H), 2.52 (q, J=7.3 Hz, 2H), 4.10 (s br, 1H), 7.25 (s, 2H).
c) The title compound is then prepared from the above 4-amino-3-ethyl-5-methyl-benzonitrile in analogy to 3-chloro-N-hydroxy-4-methanesulfonylamino-5-methyl-benzamidine; LC-MS**: tR=0.26 min, [M+1]+=272.32.
3-Chloro-4-ethanesulfonylamino N-hydroxy-5-methyl-benzamidine
The title compound is prepared in analogy to 3-chloro-N-hydroxy-4-methanesulfonylamino-5-methyl-benzamidine using ethanesulfonylchloride; LC-MS**: tR=0.27 min, [M+1]+=292.13; 1H NMR (D6-DMSO): δ 1.36 (t, J=7.5 Hz, 3H), 2.40 (s, 3H), 3.22 (q, J=7.5 Hz), 5.88 (s, 2H), 7.57 (d, J=1.5 Hz, 1H), 7.63 (d, J=1.5 Hz, 1H), 9.18 (s, 1H), 9.78 (s, 1H).
4-Benzyloxy-3-ethyl-5-methyl-benzoic acid
a) To a solution of 3-ethyl-4-hydroxy-5-methyl-benzaldehyde (34.9 g, 0.213 mol, prepared from 2-ethyl-6-methyl-phenol according to the literature cited for 3-ethyl-4,N-dihydroxy-5-methyl-benzamidine) in MeCN (350 mL), K2CO3 (58.7 g, 0.425 mol) and benzylbromide (36.4 g, 0.213 mol) are added. The mixture is stirred at 60° C. for 2 h before it is cooled to rt, diluted with water and extracted twice with EA. The org. extracts are washed with water and concentrated to give crude 4-benzyloxy-3-ethyl-5-methyl-benzaldehyde (45 g) as an orange oil. 1H NMR (CDCl3): δ1.29 (t, J=7.5 Hz, 3H), 2.40 (s, 3H), 2.77 (q, J=7.8 Hz, 2H), 4.90 (s, 2H), 7.31-7.52 (m, 5H), 7.62 (d, J=1.5 Hz, 1H), 7.66 (d, J=1.8 Hz, 1H), 9.94 (s, 1H).
b) To a mixture of 4-benzyloxy-3-ethyl-5-methyl-benzaldehyde (132 g, 0.519 mol) and 2-methyl-2-butene (364 g, 5.19 mol) in tert.-butanol (1500 mL), a solution of NaH2PO4 dihydrate (249 g, 2.08 mol) in water (1500 mL) is added. To this mixture, NaClO2 (187.8 g, 2.08 mol) is added in portions. The temperature of the reaction mixture is kept below 30° C., and evolution of gas is observed. Upon completion of the addition, the orange bi-phasic mixture is stirred well for 3 h before it is diluted with TBME (1500 mL). The org. layer is separated and washed with 20% aq. NaHS solution (1500 mL) and water (500 mL). The org. phase is then extracted three times with 0.5 N aq. NaOH (1000 mL), the aq. phase is acidified with 25% aq. HCl (500 mL) and extracted twice with TBME (1000 mL). These org. extracts are combined and evaporated to dryness to give the title compound; 1H NMR (D6-DMSO): δ 1.17 (t, J=7.5 Hz, 3H), 2.31 (s, 3H), 2.67 (q, J=7.5 Hz, 2H), 4.86 (s, 2H), 7.34-7.53 (m, 5H), 7.68 (s, 2H), 12.70 (s, 1H).
Example 1 (S)-3-(2-Ethyl-4-{5-[2-(1-ethyl-propyl)-6-methoxy-pyridin-4-yl]-[1,2,4]oxadiazol-3-yl}-6-methyl-phenoxy)-propane-1,2-diol
a) To a solution of 2-(1-ethyl-propyl)-6-methoxy-isonicotinic acid (190 mg, 732 μmol) in THF (10 mL) and DMF (2 mL), DIPEA (190 mg, 1.46 mmol) followed by TBTU (235 mg, 732 μmol) is added. The mixture is stirred at rt for 10 min before (R)-4-(2,2-dimethyl-[1,3]dioxolan-4-ylmethoxy)-3-ethyl-N-hydroxy-5-methyl-benzamidine 226 mg, 732 μmol) is added. The mixture is stirred at rt for 1 h before it is diluted with EA and washed with water. The org. phase is separated and concentrated. The remaining residue is dissolved in dioxane (10 mL) and heated to 105° C. for 18 h. The mixture is cooled to rt, concentrated and the crude product is purified on prep. TLC plates using DCM containing 10% of methanol to give 4-{3-[4-((R)-2,2-dimethyl-[1,3]dioxolan-4-ylmethoxy)-3-ethyl-5-methyl-phenyl]-[1,2,4]oxadiazol-5-yl}-2-(1-ethyl-propyl)-6-methoxy-pyridine (256 mg) as a yellow oil; LC-MS: tR=1.28 min, [M+H]+=496.23.
b) A solution of 4-{3-[4-((R)-2,2-dimethyl-[1,3]dioxolan-4-ylmethoxy)-3-ethyl-5-methyl-phenyl]-[1,2,4]oxadiazol-5-yl}-2-(1-ethyl-propyl)-6-methoxy-pyridine (250 mg, 504 μmol) in 4 M HCl in dioxane (10 mL) is stirred at rt for 90 min before it is concentrated. The crude product is purified on prep. TLC plates using DCM containing 10% of methanol to give the title compound (76 mg) as a pale brownish solid; LC-MS: tR=1.12 min, [M+H]+=456.12; 1H NMR (CDCl3): δ0.85 (t, J=7.0 Hz, 6H), 1.33 (t, J=7.0 Hz, 3H), 1.70-1.89 (m, 4H), 2.42 (s, 3H), 2.61-2.71 (m, 1H), 2.78 (q, J=7.3 Hz, 2H), 3.82-4.00 (m, 4H), 4.04 (s, 3H), 4.14-4.21 (m, 1H), 7.34 (s, 1H), 7.46 (s, 1H), 7.86-7.91 (m, 2H).
Example 2 (S)-3-{4-[5-(2-Cyclopentyl-6-methoxy-pyridin-4-yl)-[1,2,4]oxadiazol-3-yl]-2-ethyl-6-methyl-phenoxy}-propane-1,2-diol
The title compound is prepared in analogy to Example 1 starting from 2-cyclopentyl-6-methoxy-isonicotinic acid; LC-MS: tR=1.14 min, [M+H]+=454.16; 1H NMR (CDCl3): δ1.33 (t, J=7.5 Hz, 3H), 1.72-1.78 (m, 2H), 1.85-1.94 (m, 4H), 2.03-2.15 (m, 2H), 2.41 (s, 3H), 2.72 (d, J=5.3 Hz, 1H), 2.77 (q, J=7.5 Hz, 2H), 3.19-3.28 (m, 1H), 3.81-3.94 (m, 2 H), 3.95-3.98 (m, 2H), 4.02 (s, 3H), 4.14-4.21 (m, 1H), 7.31 (d, J=1.3 Hz, 1H), 7.51 (d, J=1.0 Hz, 1H), 7.88 (d, J=1.8 Hz), 7.89 (d, J=2.0 Hz, 1H).
PAPER

A practical synthesis of S1P receptor 1 agonist ACT-334441 (1) through late-stage convergent coupling of two key intermediates is described. The first intermediate is 2-cyclopentyl-6-methoxyisonicotinic acid whose skeleton was built from 1-cyclopentylethanone, ethyl oxalate, and cyanoacetate in a Guareschi–Thorpe reaction in 42% yield over five steps. The second, chiral intermediate, is a phenol ether derived from enantiomerically pure (R)-isopropylidene glycerol ((R)-solketal) and 3-ethyl-4-hydroxy-5-methylbenzonitrile in 71% yield in a one-pot reaction. The overall sequence entails 18 chemical steps with 10 isolated intermediates. All raw materials are cheap and readily available in bulk quantities, the reaction conditions match with standard pilot plant equipment, and the route reproducibly afforded 3–20 kg of 1 in excellent purity and yield for clinical studies.
Practical Synthesis of a S1P Receptor 1 Agonist via a Guareschi–Thorpe Reaction
| Patent ID | Date | Patent Title |
|---|---|---|
| US2015133669 | 2015-05-14 | NEW PROCESS FOR THE PREPARATION OF 2-CYCLOPENTYL-6-METHOXY-ISONICOTINIC ACID |
| US8658675 | 2014-02-25 | Pyridin-4-yl derivatives |
Is AQL Testing required within the 100% Visual Inspection?
DRUG REGULATORY AFFAIRS INTERNATIONAL

Is AQL Testing required within the 100% Visual Inspection?
One of the most frequently asked questions is whether an additional testing based on samples is required after the 100% visual inspection of parenterals. The answer is: basically, “yes”.
One of the most frequently asked questions is whether an additional AQL testing based on samples is required after the 100% visual inspection of parenterals. The background for that question is the probabilistic nature of visual inspection. It is known that the discovery of defects (like for example particulates) is a matter of detection probability. In other words, visual inspection cannot exclude that defective containers may still be in the batch which hasn’t been sorted out. This applies to manual, semi-automatic and also automatic visual inspection.
The American Pharmacopoeia has reacted to that and has integrated AQL testing in the monograph Visible Particulates in Injections. Here, the value 0.65 has been…
View original post 203 more words
Two new FDA Warning Letters for API Manufacturers in China
DRUG REGULATORY AFFAIRS INTERNATIONAL


Two new FDA Warning Letters for API Manufacturers in China
In June 2016, two API manufacturers in China received a Warning Letter from the FDA. Both companies had major deficiencies regarding data integrity. For instance, manipulations were found in HPLC analyses as well as in GC analyses. You will find more information on the current FDA Warning Letters for Chongqing Lummy and Shanghai Desano here. http://www.gmp-compliance.org/enews_05496_Two-new-FDA-Warning-Letters-for-API-Manufacturers-in-China_15488,15484,Z-QCM_n.html
The Chinese Company Chongqing Lummy Pharmaceutical Co., Ltd. received a Warning Letter from the FDA on June 21, 2016. This Warning Letter referred to both the FDA inspection from March 14-16, 2016 and the response which the API manufacturer had sent to the FDA on March 31, 2016.
It was claimed that Chongqing Lummy Pharmaceuticals had no adequate control in place to prevent data manipulation or deletion. The FDA investigator’s review of the audit trail revealed that an analyst had manipulated the computerized gas…
View original post 425 more words

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....