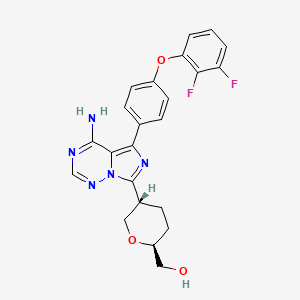
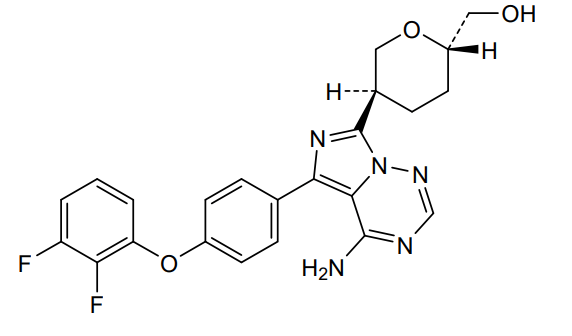
Birelentinib
CAS 2662512-15-2
MF C23H21F2N5O3 MW453.4 g/mol
[(2S,5S)-5-[4-amino-5-[4-(2,3-difluorophenoxy)phenyl]imidazo[5,1-f][1,2,4]triazin-7-yl]oxan-2-yl]methanol
[(2S,5S)-5-{4-amino-5-[4-(2,3-difluorophenoxy)phenyl]imidazo[5,1-f][1,2,4]triazin-7-yl}oxan-2-yl]methanol
tyrosine kinase inhibitor, antineoplastic, DZD8586, DZD 8586, Fast Track designation, BTK-IN-30, Z2F599L9GD
Birelentinib (also known as DZD8586) is a first-in-class, non-covalent dual inhibitor of LYN (lymphocyte-specific protein tyrosine kinase) and BTK (Bruton’s tyrosine kinase).
It is currently being developed by Dizal Pharmaceutical as an oral therapy for various B-cell malignancies.
Clinical Status and FDA Designations
As of late 2025, birelentinib has received significant attention for its potential in treating resistant blood cancers:
- Fast Track Designation: In August 2025, the U.S. FDA granted Fast Track designation to birelentinib for adult patients with relapsed or refractory (R/R) chronic lymphocytic leukemia (CLL) or small lymphocytic lymphoma (SLL).
- Target Population: It is specifically intended for those who have failed at least two prior therapies, including a covalent BTK inhibitor and a BCL-2 inhibitor.
- Key Trials: It is being evaluated in multiple studies, including the Phase 3 Tai-Shan6 trial comparing it against standard treatments like bendamustine and rituximab.
Unique Therapeutic Properties
Birelentinib is designed to overcome common drug resistance mechanisms found in existing treatments:
- Overcoming Resistance: It targets both BTK-dependent pathways (including the common C481X mutation) and BTK-independent B-cell receptor (BCR) signaling pathways.
- Blood-Brain Barrier (BBB) Penetration: A notable feature is its ability to fully penetrate the blood-brain barrier, which may offer therapeutic benefits for patients with central nervous system (CNS) involvement.
- Efficacy: Early Phase 1/2 data presented at the ASH Annual Meeting and EHA Congress in 2025 showed an Objective Response Rate (ORR) of 84.2% in heavily pretreated patients
Birelentinib is an orally bioavailable non-covalent dual inhibitor of tyrosine-protein kinases Lyn (LYN) and BTK (Bruton’s tyrosine kinase; Bruton agammaglobulinemia tyrosine kinase), with potential antineoplastic activity. Upon oral administration, birelentinib targets and inhibits both LYN and BTK, thereby blocking both BTK-dependent and BTK-independent B-cell antigen receptor (BCR) signaling pathways. This prevents the proliferation of malignant B-cells in which the BCR signaling pathway is overactivated. Birelentinib is able to cross the blood-brain barrier (BBB) and thus potentially useful in the treatment of central nervous system (CNS) metastases
SYN’
SYN



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
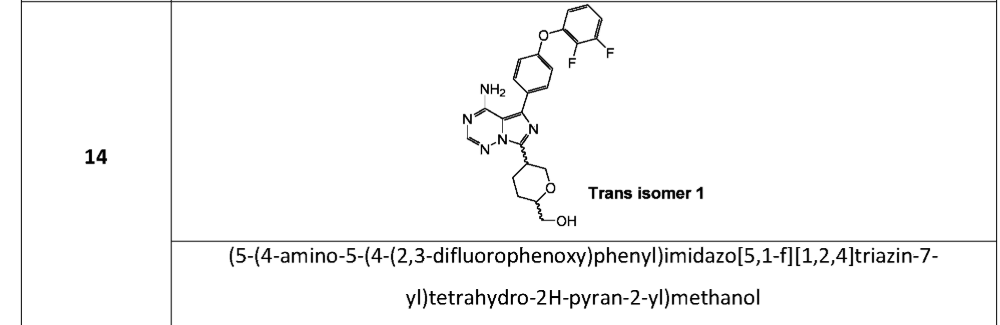
Publication Number: WO-2021136219-A1
Priority Date: 2020-01-02
/////////birelentinib, tyrosine kinase inhibitor, antineoplastic, DZD8586, DZD 8586, Fast Track designation, BTK-IN-30, Z2F599L9GD

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
