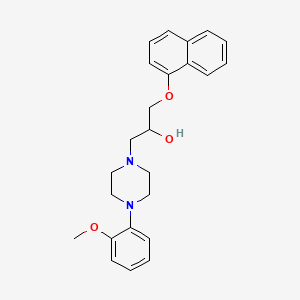
Naftopidil
1-[4-(2-methoxyphenyl)piperazin-1-yl]-3-naphthalen-1-yloxypropan-2-ol
C24H28N2O3, 392.49
CAS 57149-07-2
1-(4-(2-methoxyphenyl)piperazin-1-yl)-3-(naphthalen-1-yloxy)propan-2-ol
Naftopidil (Flivas), BM-15275NaftopidilCAS Registry Number: 57149-07-2
CAS Name: 4-(2-Methoxyphenyl)-a-[(1-naphthalenyloxy)methyl]-1-piperazineethanolAdditional Names:RS-1-[4-(2-methoxyphenyl)-1-piperazinyl]-3-(1-naphthoxy)-2-propanol; 1-(2-methoxyphenyl)-4-[3-(naphth-1-yloxy)-2-hydroxypropyl]-piperazine
Manufacturers’ Codes: KT-611Trademarks: Avishot (Kanebo); Flivas (Asahi)Molecular Formula: C24H28N2O3Molecular Weight: 392.49Percent Composition: C 73.44%, H 7.19%, N 7.14%, O 12.23%Literature References: a1-Adrenergic blocker and serotonin (5HT1A) receptor agonist. Prepn: E. C. Witte et al.,DE2408804; eidem,US3997666 (1975, 1976 both to Boehringer Mann.). Clinical pharmacodynamics: R. Kirsten et al.,Eur. J. Clin. Pharmacol.46, 271 (1994). Clinical pharmacokinetics: M. J. G. Farthing et al.,Postgrad. Med. J.70, 363 (1994). HPLC determn in human plasma: G. Niebch et al.,J. Chromatogr.534, 247 (1990). Clinical evaluation in BPH: K. Yasuda et al.,Prostate25, 46 (1994). Review of pharmacology and clinical experience: H. M. Himmel, Cardiovasc. Drug Rev.12, 32-47 (1994).
Properties: Crystals from isopropanol, mp 125-126°; also reported as colorless crystals, mp 125-129°. Insol in water. Partition coefficient (octanol/water): 75. LD50 in mice, rats (g/kg): 1.3, 6.4 orally (Himmel).Melting point: mp 125-126°; mp 125-129°Log P: Partition coefficient (octanol/water): 75Toxicity data: LD50 in mice, rats (g/kg): 1.3, 6.4 orally (Himmel)
Derivative Type: DihydrochlorideCAS Registry Number: 57149-08-3Molecular Formula: C24H28N2O3.2HClMolecular Weight: 465.41Percent Composition: C 61.94%, H 6.50%, N 6.02%, O 10.31%, Cl 15.24%Properties: Crystals from methanol/ethanol (1:2), mp 212-213°.Melting point: mp 212-213°
Therap-Cat: Antihypertensive; a-blocker in treatment of symptomatic benign prostate hypertrophy.Keywords: a-Adrenergic Blocker; Antihypertensive.
Naftopidil (INN, marketed under the brand name Flivas) is a drug used in benign prostatic hypertrophy which acts as a selective α1-adrenergic receptor antagonist or alpha blocker.[1]
PATENT
DE 2408804
CN 101671317
CN 102816136
JP 2013023467
JP 2014118360
IN 2011CH00466
US 20150353473
CN 104744405
IN 2013CH06042
IN 2012DE02071
JP 2016044182
PAPER
ChemMedChem (2009), 4(3), 393-9.
The Journal of organic chemistry (2013), 78(18), 9076-84.
e-EROS Encyclopedia of Reagents for Organic Synthesis (2014), 1-5
European journal of medicinal chemistry (2015), 96, 83-91.
Bioorganic & medicinal chemistry letters (2018), 28(9), 1534-1539.
ChemistrySelect (2019), 4(26), 7745-7750.
Green Chemistry (2019), 21(16), 4422-4433. |
PAPER
https://www.scielo.br/j/jbchs/a/q5qDxfT9mSwtL9hhQYxyhgs/?lang=en#

(S)-1-(4-(2-Methoxyphenyl)piperazin-1-yl)-3-(naphthalene1-yloxy)propan-2-ol (2b) To a solution of epoxide 8b (0.1 g, 0.5 mmol) in anhydrous 2-propanol (10 mL) was added 1-(2-methoxyphenyl) piperazine (0.096 g, 0.5 mmol) and the reaction mixture was refluxed for 32 h. After completion of reaction, the solvent was removed under reduced pressure and purification was carried out by flash column chromatography (230-400 mesh silica). The EtOAc:petroleum ether (60:40) was used as solvent system for elution, it afforded the (S)-(+)-naftopidil 2b as a yellow solid (0.156 g, 80%); mp 126-127°C; [α]D 25 +4.3o (c 1.55, MeOH);3 [α]D 25 +4.5o (c 1.5, MeOH); IR (CHCl3) νmax/cm-1 3403, 3031, 2977, 2907, 1261, 1225; 1 H NMR (300 MHz, CDCl3) d 2.58-2.70 (m, 4H, N-CH2), 2.80-2.85 (m, 2H, CH2N), 3.03-3.51 (m, 4H, NCH2), 3.51 (bs, 1H, OH), 3.75 (s, 3H, OCH3), 4.02-4.10 (m, 2H, OCH2), 4.19-4.23 (m, 1H, CH), 6.72-6.85 (m, 2H, Ar-H), 6.83-6.85 (d, 2H, J 3.9 Hz, Ar-H), 6.87-6.95 (1H, m, Ar-H), 7.14-7.29 (1H, m, Ar-H), 7.33-7.42 (3H, m, Ar-H), 7.69-7.72 (m, 1H, Ar-H), 8.19-8.22 (m, 1H, Ar-H); 13C NMR (75 MHz, CDCl3) d 50.44 (NCH2), 53.43 (NCH2), 55.17 (OCH3), 60.85 (CH2N), 65.47 (CH), 70.36 (OCH2), 104.73 (Ar), 111.03 (Ar), 118.05 (Ar), 120.39 (Ar), 120.83 (Ar), 121.78 (Ar), 122.91 (Ar), 125.07 (Ar), 125.41 (Ar), 125.67 (Ar), 126.26 (Ar), 127.32 (Ar), 134.31 (Ar), 140.87 (Ar), 152.04 (Ar), 154.21 (Ar); LC-MS m/z 393.36 (M+ + 1), 415.36 (M+ + Na); For compound 2a: [α]D 25 -10.6o (c 1, MeOH,);6 [α]D 25 -11.7o (c 1, MeOH).
PATENT
CN 1473820
PATENT
WO 2018026371
https://patents.google.com/patent/WO2018026371A1/en
PATENT
JP-2021104982
Naftopidil monohydrochloride dihydrate and its use for the preparation of naftopidil , which is known as an ameliorating agent for dysuria associated with benign prostatic hyperplasia.Naftopidil is known as an ameliorating agent for dysuria associated with benign prostatic hyperplasia. Since naftopidil is administered as a free form, there is a need for a method for preparing the free form that can be obtained efficiently and with high purity.
Japanese Unexamined Patent Publication No. 50-12186 (Patent Document 1) discloses a method for preparing naftopidil, and states that naftopidil was obtained in a yield of 29% to 79% in the examples thereof. In particular, in Example 3, naftopidil is obtained via naftopidil hydrochloride anhydride, but the yield is 49%, and the purity is not described.
Japanese Patent Application Laid-Open No. 2013-23467 (Patent Document 2) reacts 1- (2-methoxyphenyl) piperazin with 2-[(1-naphthyloxy) methyl] oxylane to obtain crude naftopidil, which is obtained as toluene. Discloses a method for obtaining purified naftopidil from water and water, as well as a mixed solvent of toluene and methanol. In this method, the yield of crude naftopidil did not reach 80%, and the purity after two purification operations using toluene and water, and then toluene and methanol was said to be 99.62% at the highest. ing. In this method, crude naftopidil is not chlorinated with hydrochloric acid.
In Indian patent application 466 / CHE / 2011 (Patent Document 3), crude naftopidil was recrystallized from ethyl acetate to obtain naftopidil in a yield of 79% and a purity of 99.90%, and further recrystallized from methanol to obtain purity. It discloses a method of obtaining 99.99% naftopidil. Even with this method, crude naftopidil is not chlorinated with hydrochloric acid.
Indian Patent Application 2071 / DEL / 2012 (Patent Document 4) discloses a method for producing green chemical naftopidil using metal nanoparticles. Here, naftopidil is purified by column chromatography using silica gel to obtain naftopidil in a yield of 63%, but the purity is not disclosed.patcit 1: Japanese Patent Application
Laid-Open No. 50-12186 patcit 2: Japanese Patent Application Laid-Open No.
2013-23467 patcit 3: Indian Patent Application 466 / CHE / 2011
patcit 4: Indian Patent Application 2071 / DEL / 2012Production
of Naftopidil Monohydrochloride Dihydrate The naftopidil monohydrochloride dihydrate according to the present invention is preferably prepared according to the following scheme.
[Chem. 2]
That is, it can be obtained by reacting 2-[(1-naphthyloxy) methyl] oxylane with 1- (2-methoxyphenyl) piperazine by adding a solvent such as toluene, and then adding / presenting hydrochloric acid. ..The present invention will be described in more detail with reference to the following examples. The reactions in the examples below, and the numbers given to the compounds, are as shown in the scheme below.
[Chem. 3]
Example 1
100 g of 1 -naphthol [1] was dissolved in chloromethyloxylan [2], and a sodium methoxide methanol solution was added dropwise. After completion of the reaction, the reaction was washed with water and the organic layer was concentrated to obtain 2-[(1-naphthyloxy) methyl] oxylan [3] (yield 89%).
Example 2
A toluene solution of 1- (2-methoxyphenyl) piperazin [4] was added dropwise to a toluene solution of 5.0 g of 2-[(1-naphthyloxy) methyl] oxylan [3]. After completion of the reaction, the mixture was washed with water and cooled by adding hydrochloric acid. After the suspension is filtered off, it is dried and (2RS) -1- [4- (2-methoxyphenyl) piperazin-1-yl] -3- (naphthalene-1-yloxy) propan-2-ol. Hydrochloride dihydrate [5] was obtained (yield 95%).
Example 3
A toluene solution of 1- (2-methoxyphenyl) piperazin [4] was added dropwise to a toluene solution of 5.0 g of 2-[(1-naphthyloxy) methyl] oxylan [3]. After completion of the reaction, the mixture was washed with water, methanol and hydrochloric acid were added to separate the liquids, and the mixture was cooled. After the suspension is filtered off, it is dried and (2RS) -1- [4- (2-methoxyphenyl) piperazin-1-yl] -3- (naphthalene-1-yloxy) propan-2-ol. Hydrochloride dihydrate [5] was obtained (yield 81%).
Example 4
A toluene solution of 1- (2-methoxyphenyl) piperazin [4] was added dropwise to a toluene solution of 5.0 g of 2-[(1-naphthyloxy) methyl] oxylan [3]. After completion of the reaction, the mixture was washed with water, methanol and hydrochloric acid were added, and the mixture was cooled. After the suspension is filtered off, it is dried and (2RS) -1- [4- (2-methoxyphenyl) piperazin-1-yl] -3- (naphthalene-1-yloxy) propan-2-ol. Hydrochloride dihydrate [5] was obtained (yield 86%).
Example 5
A toluene solution of 1- (2-methoxyphenyl) piperazin [4] was added dropwise to a toluene solution of 100 g of 2-[(1-naphthyloxy) methyl] oxylan [3]. After completion of the reaction, the mixture was washed with water, methanol and hydrochloric acid were added, and the mixture was cooled. After the suspension is filtered off, it is dried and (2RS) -1- [4- (2-methoxyphenyl) piperazin-1-yl] -3- (naphthalene-1-yloxy) propan-2-ol. Hydrochloride dihydrate [5] was obtained (yield 92%).
Example 6
(2RS) -1- [4- (2-methoxyphenyl) piperazin-1-yl] -3- (naphthalene-1-yloxy) propan-2-ol monohydrochloride dihydrate [5 ] Toluene and sodium hydroxide aqueous solution were added to 7.0 g. The organic layer was washed with water and concentrated, and then metall and acetonitrile were added and cooled. After the suspension is filtered off, it is dried and (2RS) -1- [4- (2-methoxyphenyl) piperazin-1-yl] -3- (naphthalene-1-yloxy) propan-2-ol [ 6] was obtained (yield 82%, chemical purity 99.98%).
Example 7
(2RS) -1- [4- (2-methoxyphenyl) piperazin-1-yl] -3- (naphthalene-1-yloxy) propan-2-ol monohydrochloride dihydrate [5 ] Toluene and an aqueous sodium hydroxide solution were added to 12.0 g. The organic layer was washed with water and concentrated, then metall was added and cooled. After the suspension is filtered off, it is dried and (2RS) -1- [4- (2-methoxyphenyl) piperazin-1-yl] -3- (naphthalene-1-yloxy) propan-2-ol [ 6] was obtained (yield 90%, chemical purity 99.99%).
Example 8
(2RS) -1- [4- (2-Methoxyphenyl) piperazin-1-yl] -3- (naphthalene-1-yloxy) propan-2-ol monohydrochloride dihydrate [5 ] Toluene, methanol, and potassium hydroxide aqueous solution were added to 116 g. The organic layer was washed with water and concentrated, then 2-propanol was added and cooled. After the suspension is filtered off, it is dried and (2RS) -1- [4- (2-methoxyphenyl) piperazin-1-yl] -3- (naphthalene-1-yloxy) propan-2-ol [ 6] was obtained (yield 90%, chemical purity 99.98%).
Comparative Example 1
A toluene solution of 1- (2-methoxyphenyl) piperazin [4] was added dropwise to a 10.0 g toluene solution of 2-[(1-naphthyloxy) methyl] oxylan [3]. After completion of the reaction, the mixture was washed with water and cooled. After the suspension is filtered off, it is dried and (2RS) -1- [4- (2-methoxyphenyl) piperazin-1-yl] -3- (naphthalene-1-yloxy) propan-2-ol [ 6] Crude crystals were obtained (yield 89%).
Comparative Example 2
(2RS) -1- [4- (2-methoxyphenyl) piperazin-1-yl] -3- (naphthalene-1-yloxy) propan-2-ol [6] obtained in Comparative Example 1. Methoxyol and acetonitrile were added to 6.0 g of the crude crystals of the above, and the mixture was cooled. After the suspension is filtered off, it is dried and (2RS) -1- [4- (2-methoxyphenyl) piperazin-1-yl] -3- (naphthalene-1-yloxy) propan-2-ol [ 6] was obtained (yield 85%, chemical purity 99.96%).
Naftopidil one identification hydrochloride dihydrate
(1) water and HCl content
mosquito – Le Fischer – water content value measured by the law was 7.3% to 7.5%. The amount of HCl measured by neutralization titration was 8.0% to 8.1%. Determined from these naftopidil: HCl: H 2 When calculating these molar ratios from O weight ratio of approximately 1: 1: 2. From this, it was judged that naftopidil monohydrochloride dihydrate was obtained.
(2) Powder X-ray Diffraction
The chart of the results of powder X-ray diffraction (Cu-Kα) of naftopidil monohydrochloride dihydrate was as shown in FIG. For reference, a chart of naftopidil is shown as FIG.
(3) Differential Thermal Analysis / Thermogravimetric Analysis
(TG / DTA) The chart of the results of differential thermal analysis / thermogravimetric analysis (TG / DTA) of naphthopidyl monohydrochloride dihydrate is as shown in FIG. rice field. Here, the measurement conditions were such that the heating rate was 5 ° C./min. For reference, a chart of naftopidil is shown as FIG.
PAPERShivani; Journal of Organic Chemistry 2007, V72(10), P3713-3722 https://pubs.acs.org/doi/10.1021/jo062674j
References
- ^ Sakai H, Igawa T, Onita T, Furukawa M, Hakariya T, Hayashi M, Matsuya F, Shida Y, Nishimura N, Yogi Y, Tsurusaki T, Takehara K, Nomata K, Shiraishi K, Shono T, Aoki D, Kanetake H (2011). “Efficacy of naftopidil in patients with overactive bladder associated with benign prostatic hyperplasia: prospective randomized controlled study to compare differences in efficacy between morning and evening medication”. Hinyokika Kiyo. 57 (1): 7–13. PMID 21304253.
| Clinical data | |
|---|---|
| Trade names | ertv |
| AHFS/Drugs.com | International Drug Names |
| Routes of administration | Oral |
| ATC code | none |
| Legal status | |
| Legal status | In general: ℞ (Prescription only) |
| Identifiers | |
| showIUPAC name | |
| CAS Number | 57149-07-2 |
| PubChem CID | 4418 |
| ChemSpider | 4265 |
| UNII | R9PHW59SFN |
| CompTox Dashboard (EPA) | DTXSID5045176 |
| ECHA InfoCard | 100.220.557 |
| Chemical and physical data | |
| Formula | C24H28N2O3 |
| Molar mass | 392.499 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| showSMILES | |
| showInChI | |
| (verify) |
/////////////////Naftopidil, KT 611, a-Adrenergic Blocker, Antihypertensive.
COC1=CC=CC=C1N2CCN(CC2)CC(COC3=CC=CC4=CC=CC=C43)O
NEW DRUG APPROVALS
ONE TIME
$10.00

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
