Home » Posts tagged 'UCB'
Tag Archives: UCB
Certolizumab pegol, セルトリズマブペゴル (遺伝子組換え)

>Amino acid sequence of the light chain DIQMTQSPSSLSASVGDRVTITCKASQNVGTNVAWYQQKPGKAPKALIYSASFLYSGVPY RFSGSGSGTDFTLTISSLQPEDFATYYCQQYNIYPLTFGQGTKVEIKRTVAAPSVFIFPP SDEQLKSGTASVVCLLNNFYPREAKVQWKVDNALQSGNSQESVTEQDSKDSTYSLSSTLT LSKADYEKHKVYACEVTHQGLSSPVTKSFNRGEC
>Amino acid sequence of the heavy chain EVQLVESGGGLVQPGGSLRLSCAASGYVFTDYGMNWVRQAPGKGLEWMGWINTYIGEPIY ADSVKGRFTFSLDTSKSTAYLQMNSLRAEDTAVYYCARGYRSYAMDYWGQGTLVTVSSAS TKGPSVFPLAPSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGL YSLSSVVTVPSSSLGTQTYICNVNHKPSNTKVDKKVEPKSCDKTHTCAA
Certolizumab pegol
CAS: 428863-50-7
セルトリズマブペゴル (遺伝子組換え)
CDP 870 / CDP-870 / CDP870 / PHA-738144
| Formula |
C2115H3252N556O673S16
|
|---|---|
| Cas |
428863-50-7
|
| Mol weight |
47748.8128
|
Reducing signs and symptoms of Crohn’s disease and treatment of moderately to severely active rheumatoid arthritis (RA).
Certolizumab pegol is a recombinant Fab’ antibody fragment against tumor necrosis factor alpha which is conjugated to an approximately 40kDa polyethylene glycol (PEG2MAL40K). Polyethylene glycol helps to delay the metabolism and elimination of the drugs. Chemically, the light chain is made up of 214 amino acid residues while the heavy chain is composed of 229 amino acid residues. The molecular mass of the Fab’ antibody fragment itself is 47.8 kDa. It is used for the treatment of rheumatoid arthritis and Crohn’s disease. FDA approved on April 22, 2008
Certolizumab pegol (CDP870, tradename Cimzia) is a biologic medication for the treatment of Crohn’s disease,[1][2] rheumatoid arthritis, psoriatic arthritis and ankylosing spondylitis. It is a fragment of a monoclonal antibody specific to tumor necrosis factor alpha(TNF-α) and is manufactured by UCB.[3][4][5]
Medical uses
- Crohn’s Disease
- On April 22, 2008, the U.S. FDA approved Cimzia for the treatment of Crohn’s disease in people who did not respond sufficiently or adequately to standard therapy.[4][6][7]
- Rheumatoid arthritis
- On June 26, 2009, the Committee for Medicinal Products for Human Use (CHMP) of the European Medicines Agency (EMA) issued a positive opinion recommending that the European Commission grant a marketing authorisation for Cimzia for the treatment of rheumatoid arthritis only – the CHMP refused approval for the treatment of Crohn’s disease. The marketing authorisation was granted to UCB Pharma SA on October 1, 2009.[8]
- Psoriatic arthritis
- On September 27, 2013, the U.S. FDA approved Cimzia for the treatment of adult patients with active psoriatic arthritis.[9]
Method of action
Certolizumab pegol is a monoclonal antibody directed against tumor necrosis factor alpha. More precisely, it is a PEGylated Fabfragment of a humanized TNF inhibitor monoclonal antibody.[10]
Clinical trials
- Crohn’s disease
- Positive results have been demonstrated in two phase III trials (PRECiSE 1 and 2) of certolizumab pegol versus placebo in moderate to severe active Crohn’s disease.[1][10][11][12]
- Axial spondyloarthritis
- In 2013, a phase 3 double blind randomized placebo-controlled study found significantly positive results in patient self-reported questionnaires, with rapid improvement of function and pain reduction, in patients with axial spondyloarthritis.[13]
- Rheumatoid arthritis
- Certolizumab appears beneficial in those with rheumatoid arthritis.[14]
Side effects
Significant side effects occur in 2% of people who take the medication.[14]
References
- ^ Jump up to:a b Sandborn WJ, Feagan BG, Stoinov S, et al. (July 2007). “Certolizumab pegol for the treatment of Crohn’s disease”. N. Engl. J. Med. 357 (3): 228–38. doi:10.1056/NEJMoa067594. PMC 3187683. PMID 17634458.
- ^ Goel, Niti; Sue Stephens (2010). “Certolizumab pegol”. mAbs. 2 (2): 137–147. doi:10.4161/mabs.2.2.11271. PMC 2840232. PMID 20190560.
- ^ Kaushik VV, Moots RJ (April 2005). “CDP-870 (certolizumab) in rheumatoid arthritis”. Expert Opinion on Biological Therapy. 5 (4): 601–6. doi:10.1517/14712598.5.4.601. PMID 15934837.
- ^ Jump up to:a b index.cfm?fuseaction=Search.Label_ApprovalHistory “Cimzia Label and Approval History” Check
|url=value (help). Drugs@FDA. U.S. Food and Drug Administration(FDA). Retrieved 2009-11-15. - ^ “Cimzia Prescribing Information” (PDF). US Food and Drug Administration (FDA). April 2016. Retrieved 2016-08-21.
- ^ UCB press release – Cimzia Approved in the US for the Treatment of Moderate to Severe Crohn’s Disease. Retrieved April 22, 2008.
- ^ Waknine, Yael (May 1, 2008). “FDA Approvals: Patanase, Actonel, Cimzia”. Medscape. Retrieved 2008-05-01.
- ^ “Cimzia European Public Assessment Report”. European Medicines Agency. Retrieved November 15, 2009.
- ^ “Cimzia (certolizumab pegol) approved by the U.S. FDA for treatment of adult patients with active psoriatic arthritis”. Archived from the original on October 1, 2013. Retrieved October 1, 2013.
- ^ Jump up to:a b Schreiber S. et al., Certolizumab pegol, a humanised anti-TNF pegylated FAb’ fragment, is safe and effective in the maintenance of response and remission following induction in active Crohn’s disease: a phase 3 study (precise), Gut, 2005, 54, suppl7, A82
- ^ Sandborn et al., Certolizumab pegol administered subcutaneously is effective and well tolerated in patients with active Crohn’s disease: results from a 26-week, placebo-controlled Phase 3 study (PRECiSE 1), Gastroenterology, 2006, 130, A107
- ^ “New Analysis Shows Cimzia (Certolizumab Pegol) Maintained Remission and Response in Recent Onset Crohn’s Disease” (Press release). UCB. October 23, 2006. Retrieved 2009-11-15.
- ^ Sieper J, Tubergen A, Coteur G, Woltering F, Landewe R (May 2013). “PMS50 – Rapid Improvements In Patient-Reported Outcomes With Certolizumab Pegol In Patients With Axial Spondyloarthritis, Including Ankylosing Spondylitis And Non-Radiographic Axial Spondyloarthritis: 24-Week Results Of A Phase 3 Double Blind Randomized Placebo-Controlled Study”. Value in Health. 16 (3): A227. doi:10.1016/j.jval.2013.03.1150.
- ^ Jump up to:a b Ruiz Garcia, V; Jobanputra, P; Burls, A; Vela Casasempere, P; Bort-Marti, S; Bernal, JA (Sep 8, 2017). “Certolizumab pegol (CDP870) for rheumatoid arthritis in adults”(PDF). The Cochrane Database of Systematic Reviews. 9: CD007649. doi:10.1002/14651858.CD007649.pub4. PMID 28884785.
External links
- certolizumab+pegol at the US National Library of Medicine Medical Subject Headings (MeSH)
- Cimzia Website
FDA approves treatment Cimzia (certolizumab pegol) for patients with a type of inflammatory arthritis
March 28, 2019
Release
The U.S. Food and Drug Administration today approved Cimzia (certolizumab pegol) injection for treatment of adults with a certain type of inflammatory arthritis called non-radiographic axial spondyloarthritis (nr-axSpA), with objective signs of inflammation. This is the first time that the FDA has approved a treatment for nr-axSpA.
“Today’s approval of Cimzia fulfills an unmet need for patients suffering from non-radiographic axial spondyloarthritis as there has been no FDA-approved treatments until now,” said Nikolay Nikolov, M.D., associate director for rheumatology of the Division of Pulmonary, Allergy, and Rheumatology Products in the FDA’s Center for Drug Evaluation and Research.
Nr-axSpA is a type of inflammatory arthritis that causes inflammation in the spine and other symptoms. There is no visible damage seen on x-rays, so it is referred to as non-radiographic.
The efficacy of Cimzia for the treatment of nr-axSpA was studied in a randomized clinical trial in 317 adult patients with nr-axSpA with objective signs of inflammation, indicated by elevated C-reactive protein (CRP) levels and/or sacroiliitis (inflammation of the sacroiliac joints) on MRI. The trial measured the improvement response on the Ankylosing Spondylitis Disease Activity Score, a composite scoring system that assesses disease activity including patient-reported outcomes and CRP levels. Responses were greater for patients treated with Cimzia compared to patients treated with placebo. The overall safety profile observed in the Cimzia treatment group was consistent with the known safety profile of Cimzia.
The prescribing information for Cimzia includes a Boxed Warning to advise health care professionals and patients about the increased risk of serious infections leading to hospitalization or death including tuberculosis (TB), bacterial sepsis (infection in the blood steam), invasive fungal infections (such as histoplasmosis, an infection that affects the lungs), and other infections. Cimzia should be discontinued if a patient develops a serious infection or sepsis. Health care providers are advised to perform testing for latent TB and, if positive, to start treatment for TB prior to starting Cimzia. All patients should be monitored for active TB during treatment, even if the initial latent TB test is negative. The Boxed Warning also advises that lymphoma (cancer in blood cells) and other malignancies, some fatal, have been reported in children and adolescent patients treated with tumor necrosis factor (TNF) blockers, of which Cimzia is a member. Cimzia is not indicated for use in pediatric patients. Cimzia must be dispensed with a patient Medication Guide that describes important information about the drug’s uses and risks.
Cimzia was originally approved in 2008 and is also indicated for adult patients with Crohn’s disease, moderate-to-severe rheumatoid arthritis, active ankylosing spondylitis (AS) and moderate-to-severe plaque psoriasis who are candidates for systemic therapy or phototherapy.
The FDA granted the approval of Cimzia to UCB.

Syringe with 200mg Certolizumab pegol
|
|
| Monoclonal antibody | |
|---|---|
| Type | Fab’ fragment |
| Source | Humanized (from mouse) |
| Target | TNF alpha |
| Clinical data | |
| Trade names | Cimzia |
| AHFS/Drugs.com | Consumer Drug Information |
| MedlinePlus | a608041 |
| License data | |
| Pregnancy category |
|
| Routes of administration |
Subcutaneous |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Elimination half-life | about 11 days |
| Excretion | Renal (PEG only) |
| Identifiers | |
| CAS Number | |
| ChemSpider |
|
| UNII | |
| KEGG | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C2115H3252N556O673S16 |
| Molar mass | 47,750 g/mol g·mol−1 |
///////////////FDA 2019, Cimzia, certolizumab pegol, inflammatory arthritis, UCB
FDA approves treatment Cimzia (certolizumab pegol) for patients with a type of inflammatory arthritis
FDA approves treatment Cimzia (certolizumab pegol) for patients with a type of inflammatory arthritis
March 28, 2019
Release
The U.S. Food and Drug Administration today approved Cimzia (certolizumab pegol) injection for treatment of adults with a certain type of inflammatory arthritis called non-radiographic axial spondyloarthritis (nr-axSpA), with objective signs of inflammation. This is the first time that the FDA has approved a treatment for nr-axSpA.
“Today’s approval of Cimzia fulfills an unmet need for patients suffering from non-radiographic axial spondyloarthritis as there has been no FDA-approved treatments until now,” said Nikolay Nikolov, M.D., associate director for rheumatology of the Division of Pulmonary, Allergy, and Rheumatology Products in the FDA’s Center for Drug Evaluation and Research.
Nr-axSpA is a type of inflammatory arthritis that causes inflammation in the spine and other symptoms. There is no visible damage seen on x-rays, so it is referred to as non-radiographic.
The efficacy of Cimzia for the treatment of nr-axSpA was studied in a randomized clinical trial in 317 adult patients with nr-axSpA with objective signs of inflammation, indicated by elevated C-reactive protein (CRP) levels and/or sacroiliitis (inflammation of the sacroiliac joints) on MRI. The trial measured the improvement response on the Ankylosing Spondylitis Disease Activity Score, a composite scoring system that assesses disease activity including patient-reported outcomes and CRP levels. Responses were greater for patients treated with Cimzia compared to patients treated with placebo. The overall safety profile observed in the Cimzia treatment group was consistent with the known safety profile of Cimzia.
The prescribing information for Cimzia includes a Boxed Warning to advise health care professionals and patients about the increased risk of serious infections leading to hospitalization or death including tuberculosis (TB), bacterial sepsis (infection in the blood steam), invasive fungal infections (such as histoplasmosis, an infection that affects the lungs), and other infections. Cimzia should be discontinued if a patient develops a serious infection or sepsis. Health care providers are advised to perform testing for latent TB and, if positive, to start treatment for TB prior to starting Cimzia. All patients should be monitored for active TB during treatment, even if the initial latent TB test is negative. The Boxed Warning also advises that lymphoma (cancer in blood cells) and other malignancies, some fatal, have been reported in children and adolescent patients treated with tumor necrosis factor (TNF) blockers, of which Cimzia is a member. Cimzia is not indicated for use in pediatric patients. Cimzia must be dispensed with a patient Medication Guide that describes important information about the drug’s uses and risks.
Cimzia was originally approved in 2008 and is also indicated for adult patients with Crohn’s disease, moderate-to-severe rheumatoid arthritis, active ankylosing spondylitis (AS) and moderate-to-severe plaque psoriasis who are candidates for systemic therapy or phototherapy.
The FDA granted the approval of Cimzia to UCB.
///////////////FDA 2019, Cimzia, certolizumab pegol, inflammatory arthritis, UCB
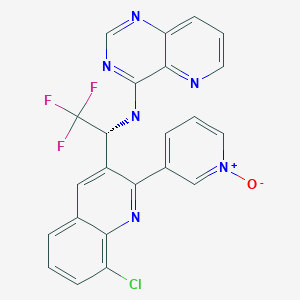
SELETALISIB, селеталисиб , سيلستاليسيب , 司来利塞 ,
SELETALISIB
CAS 1362850-20-1
UCB-5857 , Plaque psoriasis,Sjoegren’s syndrome,Immunodeficiency disorders
PHASE 3 UCB
23H14ClF3N6O , 482.85
Phosphatidylinositol 3 kinase delta (PI3Kδ) inhibitors
| Pyrido[3,2-d]pyrimidin-4-amine, N-[(1R)-1-[8-chloro-2-(1-oxido-3-pyridinyl)-3-quinolinyl]-2,2,2-trifluoroethyl]- |
3-{8-chloro-3-[(1R)-2,2,2-trifluoro-1-({pyrido[3,2-d]pyrimidin-4-yl}amino)ethyl]quinolin-2-yl}pyridin-1-ium-1-olate
Seletalisib has been used in trials studying the treatment and basic science of Primary Sjogren’s Syndrome.
- Originator UCB
- Class Anti-inflammatories; Small molecules
- Mechanism of Action Immunomodulators; Phosphatidylinositol 3 kinase delta inhibitors
- Phase III Immunodeficiency disorders
- Phase II Sjogren’s syndrome
- No development reported Plaque psoriasis
- 05 Dec 2017 UCB Celltech terminates a phase II trial in Sjogren’s syndrome in France, Spain, United Kingdom, Greece, Sweden, Italy, due to enrolment challenges (PO) (NCT02610543) (EudraCT2014-004523-51)
- 04 Nov 2017 No recent reports of development identified for phase-I development in Plaque-psoriasis in United Kingdom (PO, Capsule)
- 14 Jun 2017 Pharmacokinetics and pharmacodynamics data from Preclinical and Clinical studies in Immunodeficiency disorders presented at the 18th Annual Congress of the European League Against Rheumatism (EULAR-2017)
SYN
US 9029392
https://patents.google.com/patent/US9029392B2/en
Example 27 N—{(R)-1-[8-Chloro-2-(pyridin-3-yl)quinolin-3-yl]-2,2,2-trifluoroethyl}N-(1-oxypyrido-[3,2-d]pyrimidin-4-yl)amine
A stirred solution of Example 1 (955 mg, 2.05 mmol) in DCM (40 mL) was cooled to 0° C. MCPBA (410 mg, 1.84 mmol) was added and the mixture was allowed to warm slowly to r.t. over 3 h. The reaction mixture was partitioned between DCM and saturated aqueous NaHCO3 solution. The aqueous phase was extracted with further DCM and the combined organic fractions were washed with brine, dried Na2SO4) and evaporated in vacuo. The residue was purified by column chromatography (SiO2, 3-60% MeOH in EtOAc) to give the title compound (39 mg, 4%) as a yellow solid. δH (DMSO-d6) 9.64-9.52 (m, 1H), 9.30 (s, 1H), 9.06 (dd, J 4.2, 1.3 Hz, 1H), 8.78-8.71 (m, 2H), 8.67 (dd, J 4.9, 1.6 Hz, 1H), 8.64 (s, 1H), 8.16-8.01 (m, 4H), 7.75-7.69 (m, 1H), 7.52 (ddd, J 7.8, 4.9, 0.7 Hz, 1H), 6.65-6.52 (m, 1H). LCMS (ES+) 483 (M+H)+, RT 1.87 minutes.

AND

PATENT
WO 2012032334
PATENT
WO 2015181053
WO 2015181055
WO 2016170014
PATENT
A SPECIFIC TRIFLUOROETHYL QUINOLINE ANALOGUE FOR USE IN THE TREATMENT OF APDS
The present invention relates to the new therapeutic use of a known chemical compound. More particularly, the present invention concerns the use of a specific substituted quinoline derivative comprising a fluorinated ethyl side-chain in the treatment of activated phosphoinositide 3 -kinase delta syndrome (APDS).
N- {(R)- 1 -[8-Chloro-2-(l -oxypyridin-3-yl)quinolin-3-yl]-2,2,2-trifiuoroethyl} -pyrido[3,2-JJpyrimidin-4-ylamine is specifically disclosed in WO 2012/032334. The compounds described in that publication are stated to be of benefit as pharmaceutical agents, especially in the treatment of adverse inflammatory, autoimmune, cardiovascular, neurodegenerative, metabolic, oncological, nociceptive and ophthalmic conditions.
There is no specific disclosure or suggestion in WO 2012/032334, however, that the compounds described therein might be beneficial in the treatment of APDS.
Activated phosphoinositide 3-kinase delta syndrome (APDS), also known as
PASLI (pi ΙΟδ-activating mutation causing senescent T cells, lymphadenopathy and immunodeficiency), is a serious medical condition that impairs the immune system.
APDS patients generally have reduced numbers of white blood cells (lymphopenia), especially B cells and T cells, compromising their propensity to recognise and attack invading microorganisms, such as viruses and bacteria, and thereby prevent infection. Individuals affected with APDS develop recurrent infections, particularly in the lungs, sinuses and ears. Recurrent respiratory tract infections may gradually lead to bronchiectasis, a condition which damages the passages leading from the windpipe to the lungs (bronchi) and can cause breathing problems. APDS patients may also suffer from chronic active viral infections, including Epstein-Barr virus infections and cytomegalovirus infections.
APDS has also been associated with abnormal clumping of white blood cells, which can lead to enlarged lymph nodes (lymphadenopathy). Alternatively, the white blood cells can build up to form solid masses (nodular lymphoid hyperplasia), usually in the moist lining of the airways or intestines. Whilst lymphadenopathy and nodular lymphoid hyperplasia are benign (noncancerous), APDS also increases the risk of developing a form of cancer called B cell lymphoma.
APDS is a disorder of childhood, typically arising soon after birth. However, the precise prevalence of APDS is currently unknown.
Phosphoinositide 3-kinase delta (ΡΒΚδ) is a lipid kinase which catalyses the generation of phosphatidylinositol 3,4,5-trisphosphate (PIP3) from phosphatidylinositol 4,5-bisphosphate (PIP2). PI3K5 activates signalling pathways within cells, and is specifically found in white blood cells, including B cells and T cells. PI3K5 signalling is involved in the growth and division (proliferation) of white blood cells, and it helps direct B cells and T cells to mature (differentiate) into different types, each of which has a distinct function in the immune system.
APDS is known to occur in two variants, categorised as APDSl and APDS2.
APDSl is associated with a heterozygous gain-of- function mutation in the PIK3CD gene encoding the PI3K5 protein; whereas APDS2 is associated with loss-of-function frameshift mutations in the regulatory PIK3R1 gene encoding the p85a regulatory subunit of class I phosphoinositide 3-kinase (PI3K) peptides. Both mutations lead to hyperactivated PI3K signalling. See I. Angulo et ah, Science, 2013, 342, 866-871; C.L. Lucas et ah, Nature Immunol, 2014, 15, 88-97; and M-C. Deau et al, J. Clin. Invest., 2014, 124, 3923-3928.
There is currently no effective treatment available for APDS. Because of the seriousness of the condition, and the fact that it arises in infancy, the provision of an effective treatment for APDS would plainly be a highly desirable objective.
It has now been found that N-{(R)-l-[8-chloro-2-(l-oxypyridin-3-yl)quinolin-3-yl]- 2,2,2-trifluoroethyl}pyrido[3,2-(i]pyrimidin-4-ylamine is capable of inhibiting the elevation of PI3K signalling in T cells (lymphocytes) from both APDSl and APDS2 patients in the presence or absence of T cell receptor activation.
The present invention accordingly provides N-{(R)-l-[8-chloro-2-(l-oxypyridin-3-yl)quinolinB-yl]-2,2,2-trifluoroethyl}pyrido[3,2-JJpyrimidin-4-ylamine of formula (A):
or a pharmaceutically acceptable salt thereof, for use in the treatment and/or prevention of APDS.
The present invention also provides a method for the treatment and/or prevention of APDS, which method comprises administering to a patient in need of such treatment an effective amount of N-{(R)-l-[8-chloro-2-(l-oxypyridin-3-yl)quinolin-3-yl]-2,2,2-trifluoro-ethyl}pyrido[3,2-(i]pyrimidin-4-ylamine of formula (A) as depicted above, or a pharmaceutically acceptable salt thereof. The present invention also provides the use of N-{(R)-l-[8-chloro-2-(l-oxypyridin-3-yl)quinolin-3-yl]-2,2,2-trifluoroethyl}pyrido[3,2-JJpyrimidin-4-ylamine of formula (A) as depicted above, or a pharmaceutically acceptable salt thereof, for the manufacture of a medicament for the treatment and/or prevention of APDS.
PAPER
Journal of Pharmacology and Experimental Therapeutics (2017), 361(3), 429-440.
http://jpet.aspetjournals.org/content/361/3/429
///////////////SELETALISIB, PHASE 3, UCB, селеталисиб , سيلستاليسيب , 司来利塞 ,
[O-][N+]1=CC(=CC=C1)C1=NC2=C(Cl)C=CC=C2C=C1[C@@H](NC1=NC=NC2=CC=CN=C12)C(F)(F)F
READ
ANTHONY MELVIN CRASTO
 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO …..FOR BLOG HOME CLICK HERE
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO …..FOR BLOG HOME CLICK HERE
 amcrasto@gmail.com
amcrasto@gmail.com
CALL +919323115463 INDIA
BRIVARACETAM
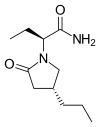
BRIVARACETAM, UCB-34714
(2S)-2-[(4R)-2-oxo-4-propylpyrrolidin-1-yl]butanamide
| Molecular Formula: | C11H20N2O2 |
|---|---|
| Molecular Weight: | 212.2887 g/mol |
UNII-U863JGG2IA
UCB; For the treatment of partial onset seizures related to epilepsy, Approved February 2016
Brivaracetam, the 4-n-propyl analog of levetiracetam, is a racetam derivative with anticonvulsant properties.[1][2] Brivaracetam is believed to act by binding to the ubiquitous synaptic vesicle glycoprotein 2A (SV2A).[3] Phase II clinical trials in adult patients with refractory partial seizures were promising. Positive preliminary results from stage III trials have been recorded,[4][5] along with evidence that it is around 10 times more potent[6] for the prevention of certain types of seizure in mouse models than levetiracetam, of which it is an analogue.
On 14 January 2016, the European Commission,[7] and on 18 February 2016, the USFDA[8] approved brivaracetam under the trade name Briviact (by UCB). The launch of this anti-epileptic is scheduled for the first quarter of that year. Currently, brivaracetam is still not approved in other countries like Australia, Canada and Switzerland.
Brivaracetam was approved by European Medicine Agency (EMA) on Jan 14, 2016 and approved by the U.S. Food and Drug Administration (FDA) on Feb 18, 2016. It was developed and marketed as Briviact® by UCB in EU/US.
Brivaracetam is a selective high-affinity synaptic vesicle protein 2A ligand, as an adjunctive therapy in the treatment of partial-onset seizures with or without secondary generalization in adult and adolescent patients from 16 years of age with epilepsy.
Briviact® is available in three formulations, including film-coated tablets, oral solution and solution for injection/infusion. And it will be available as 10 mg, 25 mg, 50 mg, 75 mg and 100 mg film-coated tablets, a 10 mg/ml oral solution, and a 10 mg/ml solution for injection/infusion. The recommended starting dose is either 25 mg twice a day or 50 mg twice a day, depending on the patient’s condition. The dose can then be adjusted according to the patient’s needs up to a maximum of 100 mg twice a day. Briviact can be given by injection or by infusion (drip) into a vein if it cannot be given by mouth.
European Patent No. 0 162 036 Bl discloses compound (S)-α-ethyl-2-oxo-l- pyrrolidine acetamide, which is known under the International Non-proprietary Name of Levetiracetam.
Levetiracetam
Levetiracetam is disclosed as a protective agent for the treatment and prevention of hypoxic and ischemic type aggressions of the central nervous system in European patent EP 0 162 036 Bl. This compound is also effective in the treatment of epilepsy.
The preparation of Levetiracetam has been disclosed in European Patent No. 0 162 036 and in British Patent No. 2 225 322.
International patent application having publication number WO 01/62726 discloses 2-oxo-l -pyrrolidine derivatives and methods for their preparation. It particularly discloses compound (2S)-2-[(4R)-2-oxo-4-propyl-pyrrolidin-l-yl] butanamide known under the international non propriety name of brivaracetam.
Brivaracetam
International patent application having publication number WO 2005/121082 describes a process of preparation of 2-oxo-l -pyrrolidine derivatives and particularly discloses a process of preparation of (2S)-2-[(4S)-4-(2,2-difluorovinyl)-2-oxo-pyrrolidin-l- yl]butanamide known under the international non propriety name of seletracetam.
Seletracetam
Kenda et al., in J. Med. Chem. 2004, 47, 530-549, describe processes of preparation of 2-oxo-l -pyrrolidine derivatives and particularly discloses compound 1-((1S)-I- carbamoyl-propyl)-2-oxo-pyrrolidone-3-carboxylic acid as a synthetic intermediate.
WO2005028435
CLIPS
Find better ways to make old and new epilepsy drugs. J. Surtees and co-inventors disclose alternative processes for making active pharmaceutical ingredients (APIs) that are used to treat epilepsy and seizures. One compound that can be prepared by their processes is the established drug levetiracetam (1, Figure 1), marketed under the trade name Keppra. Because 1 is now off-patent, there is obvious interest in new drugs.

The inventors also claim that seletracetam (2) and brivaracetam (3) (Figure 2) can be prepared by their processes. These drugs are apparently much more active than 1.
All of the drugs are used as single isomers, so a stereoselective synthesis is desirable. The inventors describe two routes for preparing the molecules; the first, shown in Figure 1, is the synthesis of 1 by the reaction between pyrrolidone (4) and chiral bromo amide 5 in the presence of a base. GC analysis showed that the conversion is 40.3% and that the product contains 51% of the (S)-enantiomer and 49% of the (R)-isomer. No details of their separation are given, although the use of chiral HPLC is discussed.
The same reaction is used to prepare derivative 6 of 1. Compound 7 is prepared from the corresponding hydroxy ester and then condensed with 4 to give 6. Chiral HPLC showed that the product is a mixture of 89.3% (S)-enantiomer 6 and 10.7% of its (R)-isomer.
The inventors do not describe the detailed preparation of 2, but they report that acid 8 is prepared in 41% yield from pyrrolidone 9 and acid 10 in the presence of NaH (Figure 2). Ammonolysis of 8 produces 2; no reaction details are provided.

In a reaction similar to the preparation of 8, acid 11 is prepared from 10 and pyrrolidone 12. The product is isolated in 77% yield and can be converted to 3 by ammonolysis. Again, no details are provided for this reaction.
The second route for preparing the substituted pyrrolidones does not start with simple pyrrolidones and is the subject of additional claims. The route involves a cyclization reaction, shown in Figure 3. The preparation of enantiomer 13 begins with the reaction of racemic salt 14 and optically pure bromo ester 15. This step produces intermediate 16, isolated as a yellow oil. The crude material is treated with 2-hydroxypyridine (2-HP) to cyclize it to 17. This ester is hydrolyzed to give acid 18. Conversion to 13 is carried out by adding ClCO2Et, followed by reaction with liquid NH3 in the presence of K2CO3. The overall yield of 13 is 32%.

This route is also used to prepare levetiracetam (1) by treating 5 with the HCl salt of amino ester 19 to give 20, recovered as its HCl salt in 49% yield. The salt is basified with Et3N and treated with 2-HP to cyclize it to 1, initially isolated as an oil. GC analysis showed 100% conversion, and chiral HPLC showed that the product contains 98.6% (S)-isomer and 1.4% (R)-isomer.
The inventors also prepared 1 and its (R)-enantiomer 21 by using a similar reaction scheme with alternative substrates to 5. Figure 4 outlines the route, which starts from protected hydroxy amide 22 and amino ester 23. When the reaction is carried out in the presence of Cs2CO3, the product is (R)-enantiomer24, which is used without purification to prepare 21 by treating it with 2-HP. Chiral HPLC showed that the product is 94% (R) and 6% (S).

When the reaction between 22 and 23 is run with K2CO3, the product is (S)-enantiomer 25. This is used to prepare 1, but the product contains only 79% (S)-isomer.
The inventors do not comment on the apparent stereoselectivity of the carbonate salts in the reaction of 22 with 23. This is an intriguing finding and worthy of investigation. (UCB S.A. [Brussels]. US Patent 8,338,621

SYNTHESIS
PATENT
WO2005028435
Example 1: Synthesis of (2S)-2-((4R)-2-oxo-4-n-propyl-l-pyrrolidinyl)butanamide 1.1 Synthesis of (2S)-2-aminobutyramide free base
1800 ml of isopropanol are introduced in a 5L reactor. 1800 g of (2S)-2- aminobutyramide tartrate are added under stirring at room temperature. 700 ml of a 25% aqueous solution of ammonium hydroxide are slowly added while maintaining the temperature below 25°C. The mixture is stirred for an additional 3 hours and then the reaction is allowed to complete at 18°C for 1 hour. The ammonium tartrate is filtered. Yield : 86%.
1.2 Synthesis of 5-hydroxy-4-n-propyl-furan-2-one
Heptane (394 ml) and morpholine (127.5 ml) are introduced in a reactor. The mixture is cooled to 0°C and glyoxylic acid (195 g, 150 ml, 50w% in water) is added. The mixture is heated at 20°C during 1 hour, and then valeraldehyde (148.8 ml) is added . The reaction mixture is heated at 43°C during 20 hours. After cooling down to 20CC, a 37 % aqueous solution of HCl (196.9 ml) is slowly added to the mixture, which is then stirred during 2 hours.
After removal of the heptane phase, the aqueous phase is washed three times with heptane. Diisopropyl ether is added to the aqueous phase. The organic phase is removed, and the aqueous phase further extracted with diisopropyl ether (2x). The diisopropyl ether phases are combined, washed with brine and then dried by azeotropic distillation. After filtration and evaporation of the solvent, 170g of 5- hydroxy-4-n-propyl-furan-2-one are obtained as a brown oil. Yield: 90.8 %
1.3 Synthesis of (2S)-2-((4R)-2-oxo-4-n-propyl-l-pyrrolidinyl)butanamide and (2S)-2-((4S)-2-oxo-4-n-propyl-l-pyrrolidinyl)butanamide
(S, R) (S, S) The (2S)-2-aιninobutyrarnide solution in isopropanol containing 250 g obtained as described here above is dried by azeotropic distillation under vacuum. To the dried (2S)-2-am obutyraιnide solution is added 5-hydroxy-4-n-propyl-furan-2-one (290 g) between 15°C and 25 °C; the mixture is heated to 30 °C and kept for at least 2 hours at that temperature. Acetic acid (1, 18 eq.), Pd/C catalyst (5 w/w%; Johnson Matthey 5% Pd on carbon – type 87L) are then added and hydrogen introduced into the system under pressure. The temperature is kept at 40 °C maximum and the H2 pressure maintained between 0,2 bar and 0,5 bar followed by stirring for at least 20 hours following the initial reaction. The solution is then cooled to between 15 °C and 25 °C and filtered to remove the catalyst. The solution of product in isopropanol is solvent switched to a solution of product in isopropyl acetate by azeotropic distillation with isopropyl acetate. The organic solution is washed with aqueous sodium bicarbonate followed by a brine wash and then filtered. After recristallisation, 349 g of (2S)-2-((4R)-2- oxo-4-n-propyl-l-pyrrolidinyl)butanamide and (2S)-2-((4S)-2-oxo-4-n-propyl-l- pyιτolidinyl)butanamide are obtained (Yield: 82.5%).
1.4 Preparation of (2S)-2-((4R)-2-oxo-4-n-propyl-l-pyrrolidinyl)butanamide The chromatographic separation of the two diastereoisomers obtained in 1.3 is performed using of (CHIRALPAK AD 20 um) chiral stationary phase and a 45/55 (volume /volume) mixture of n-heptane and ethanol as eluent at a temperature of 25 + 2°C. The crude (2S)-2-((4R)-2-oxo-4-n-propyl-l-pyrrolidinyl)butanamide thus obtained is recristallised in isopropylacetate, yielding pure (2S)-2-((4R)-2-oxo-4-n-propyl-l- pyrrolidinyl)butanamide (Overall yield: 80%) .
Example 2: Synthesis of (2S)-2-((4R)-2-oxo-4-n-propyl-l-pyrrolidinyl)butanamide
Example 1 is repeated except that in step 1.1 a solution of (2S)-2- aminoburyramide.HCl in isopropanol is used (27.72 g, 1.2 equivalent), which is neutralised with a NHs/isopropanol solution (3,4-3,7 mol/L). The resulting ainmonium chloride is removed from this solution by filtration and the solution is directly used for reaction -with 5-hydroxy-4-n-propyl-furan-2-one (23.62 g, 1.0 equivalent) without intermediate drying of the (2S)-2-aminobutyramide solution. Yield after separation of the two diastereoisomers and recristallisation: approximately 84%.

Ref ROUTE1
1. WO0162726A2.
2. WO2005028435A1 / US2007100150A1.
3. J. Med. Chem. 1988, 31, 893-897.
4. J. Org. Chem. 1981, 46, 4889-4894.
PATENT
https://www.google.com/patents/WO2007031263A1?cl=en
Example 3-Synthesis of brivaracetam (I)
3.a. Synthesis of (S) and (R) 2-((R)-2-oxo-4-propyl-pyrrolidin-l-yl)-butyric acid methyl ester fVIaa*) and (Wlab)
(VIaa) (VIab) A slurry of 60% sodium hydride suspension in mineral oil (0.94g, 23.4 mmol) in tetrahydrofuran (30 mL) is cooled at 0°C under a nitrogen atmosphere. A solution of substantially optically pure (R)-4-propyl-pyrrolidin-2-one (Ilia) (2g, 15.7 mmol) in tetrahydrofuran (2 mL) is added over a 15 minutes period. The reaction mixture is stirred 10 min at 0°C then a solution of methyl-2-bromo-butyric acid methyl ester (V) (3.69g, 20.4 mmol) in tetrahydrofuran (2mL) was added over a 20 minutes period. The reaction mixture is stirred at O0C until maximum conversion of starting material and the reaction mixture is then allowed to warm to room temperature and diluted with water (20 mL). Tetrahydrofuran is removed by evaporation and the residue is extracted with isopropyl acetate (20 ml + 10 mL). The combined organic layers are dried on anhydrous magnesium sulfate and evaporated to afford 3g (13.2 mmol, 86 %) of a mixture of epimers of compound (Via), as a mixture respectively of epimer (VIaa) and epimer (VIab). 1H NMR(400 MHz, CDCI3) of the mixture of epimers (VIaa) and (VIab) : δ = 4.68
(dd, J= 10.8, J= 5.1, 2×1 H) ; 3.71 (s, 2x3H); 3.60 (t app, J= 8.2, IH); 3.42 (t app, J= 8.7, IH); 313 (dd, J= 9.2, J = 6.8, IH); 2.95 (dd, J= 9.2, J= 6.8, IH); 2.56 (dd, J= 16.6, J = 8.7, 2xlH); 2.37 (dm, 2xlH); 2.10 (m, 2xlH); 2.00 (m, 2xlH); 1.68 (m, 2xlH); 1.46 (m, 2x2H); 1.36 (m, 2x2H); 0.92 (m, 2x6H).
13C NMR (400 MHz, CDCl3) of the mixture of epimers (VIaa) and (VIab) : δ =
175.9; 175.2; 171.9; 55.3; 52.4; 49.8; 49.5; 38.0; 37.8; 37.3; 36.9; 32.5; 32.2; 22.6; 22.4; 21.0; 14.4; 11.2; 11.1
HPLC (GRAD 90/10) of the mixture of epimers (VIaa) and (VIab): retention time= 9.84 minutes (100 %)
GC of the mixture of epimers (VIaa) and (VIab): retention time = 13.33 minutes (98.9 %)
MS of the mixture of epimers (VIaa) and (VIab) (ESI) : 228 MH+
3.b. Ammonolysis of compound of the mixture of (VIaa) and (VIab)
(VIaa) (VIab) (I) (VII)
A solution of (VIaa) and (VIab) obtained in previous reaction step (1.46g, 6.4 mmol) in aqueous ammonia 50 % w/w (18 mL) at 00C is stirred at room temperature for 5.5hours. A white precipitate that appears during the reaction, is filtered off, is washed with water and is dried to give 0.77g (3.6 mmol, yield = 56 %) of white solid which is a mixture of brivaracetam (I) and of compound (VII) in a 1 :1 ratio.
1H NMR of the mixture (I) and (VII) (400 MHz, CDCI3) : δ = 6.36 (s, broad, IH); 5.66 (s, broad, IH); 4.45 (m, IH); 3.53 (ddd, J= 28.8, J= 9.7, J= 8.1, IH); 3.02 (m, IH); 2.55 (m, IH); 2.35 (m, IH); 2.11 (m, IH); 1.96 (m, IH); 1.68 (m, IH); 1.38 (dm, 4H); 0.92 (m, 6H). 13c NMR of the mixture (I) and (VII) (400 MHz, CDCl3) : δ = 176.0; 175.9; 172.8;
172.5; 56.4; 56.3; 50.0; 49.9; 38.3; 38.1; 37.3; 37.0; 32.3; 32.2; 21.4; 21.3; 21.0; 20.9; 14.4; 10.9; 10.8
HPLC (GRAD 90/10) of the mixture of (I) and (VII) retention time= 7.67 minutes (100 %)
Melting point of the mixture of (I) and (VII) = 104.90C (heat from 400C to 1200C at 10°C/min)
Compounds (I) and (VII) are separated according to conventional techniques known to the skilled person in the art. A typical preparative separation is performed on a 11.7g scale of a 1 :1 mixture of compounds (I) and (VII) : DAICEL CHIRALPAK® AD 20 μm, 100*500 mm column at 300C with a 300 mL/minutes debit, 50 % EtOH – 50 % Heptane. The separation affords 5.28g (45 %) of compound (VII), retention time = 14 minutes and 5.2Og (44 %) of compounds (I), retention time = 23 minutes.
1H NMR of compound (I) (400 MHz, CDCl3): δ = 6.17 (s, broad, IH); 5.32 (s, broad, IH); 4.43 (dd, J= 8.6, J= 7.1, IH); 3.49 (dd, J= 9.8, J= 8.1, IH); 3.01 (dd, J= 9.8, J= 7.1, IH); 2.59 (dd, J= 16.8, J= 8.7, IH); 2.34 (m, IH); 2.08 (dd, J= 16.8, J= 7.9, IH); 1.95 (m, IH); 1.70 (m, IH); 1.47-1.28 (m, 4H); 0.91 (dt, J= 7.2, J= 2.1, 6H)
HPLC (GRAD 90/10) of compound (I) : retention time = 7.78 minutes
1H NMR of compound (VII) (400 MHz, CDCl3): δ = 6.14 (s, broad, IH); 5.27 (s, broad, IH); 4.43 (t app, J = 8.1, IH); 3.53 (t app, J = 9.1, IH); 3.01 (t app, J = 7.8, IH); 2.53 (dd, J = 16.5, J = 8.8, IH); 2.36 (m, IH); 2.14 (dd, J = 16.5, J = 8.1, IH); 1.97 (m, IH); 1.68 (m, IH); 1.43 (m, 2H); 1.34 (m, 2H); 0.92 (m, 6H)
3c. Epimerisation of compound of (2RV2-((R)-2-oxo-4-propyl-pyπOlidin-l-ylV butyramide (VID
Compound (VII) (200 mg, 0.94 mmol) is added to a solution of sodium tert- butoxide (20 mg, 10 % w/w) in isopropanol (2 mL) at room temperature. The reaction mixture is stirred at room temperature for 18h. The solvent is evaporated to afford 200 mg
(0.94 mmol, 100 %) of a white solid. Said white solid is a mixture of brivaracetam (I) and of (VII) in a ratio 49.3 / 50.7.
HPLC (ISO80): retention time= 7.45 min (49.3%) brivaracetam (I); retention time= 8.02 minutes (50.7%) compound (VII).

Reference:ROUTE 2
1. WO2007031263A1 / US2009318708A1.
PATENT
http://www.google.com/patents/WO2007065634A1?cl=en
(scheme 3).
Scheme 3
scheme 4.
5h. Synthesis of brivaracetam and (V) A suspension of (Id) and (Ie) (0.6 g, 2.3 mmol) in MIBK (10 mL) is heated at
120°C for 6 hours. The resulting solution is concentrated and separated on chromatography column (Silicagel 600.068-0.200 mm, cyclohexane/EtOAc : 10/90) to give 0.13 g of brivaracetam (0.6 mmol, 26 %, ee = 94 %) and (V).
1H NMR (400 MHz, CDCl3): δ = 6.17 (s, broad, IH); 5.32 (s, broad, IH); 4.43 (dd, J= 8.6, J= 7.1, IH); 3.49 (dd, J= 9.8, J= 8.1, IH); 3.01 (dd, J= 9.8, J= 7.1, IH); 2.59 (dd, J= 16.8, J= 8.7, IH); 2.34 (m, IH); 2.08 (dd, J= 16.8, J= 7.9, IH); 1.95 (m, IH); 1.70 (m, IH); 1.47-1.28 (m, 4H); 0.91 (dt, J= 7.2,J= 2.1, 6H).
HPLC (method 90/10) : Retention time = 7.78 minutes Chiral HPLC : Retention time = 9.66 minutes (97%) MS (ESI): 213 MH+
Route 3
Reference:1. WO2007065634A1 / US2009012313A1.
PAPER
http://pubs.acs.org/doi/abs/10.1021/acs.oprd.6b00094
A Biocatalytic Route to the Novel Antiepileptic Drug Brivaracetam


AS ON DEC2021 3,491,869 VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@amcrasto
/////////////////////////////////////////////////////////////////////////////
References
- von Rosenstiel P (Jan 2007). “Brivaracetam (UCB 34714)”. Neurotherapeutics 4 (1): 84–7. doi:10.1016/j.nurt.2006.11.004.PMID 17199019.
- Malawska B, Kulig K (Jul 2005). “Brivaracetam UCB”. Current Opinion in Investigational Drugs 6 (7): 740–746. PMID 16044671.
- Rogawski MA, Bazil CW (Jul 2008). “New molecular targets for antiepileptic drugs: alpha(2)delta, SV2A, and K(v)7/KCNQ/M potassium channels”. Current Neurology and Neuroscience Reports 8 (4): 345–352. doi:10.1007/s11910-008-0053-7. PMC 2587091.PMID 18590620.
- Clinical trial number NCT00464269 for “Double-blind, Randomized Study Evaluating the Efficacy and Safety of Brivaracetam in Adults With Partial Onset Seizures” at ClinicalTrials.gov
- Rogawski MA (Aug 2008). “Brivaracetam: a rational drug discovery success story”. British Journal of Pharmacology 154 (8): 1555–7.doi:10.1038/bjp.2008.221. PMC 2518467. PMID 18552880.
- Matagne A, Margineanu DG, Kenda B, Michel P, Klitgaard H (Aug 2008). “Anti-convulsive and anti-epileptic properties of brivaracetam (ucb 34714), a high-affinity ligand for the synaptic vesicle protein, SV2A”. British Journal of Pharmacology 154 (8): 1662.doi:10.1038/bjp.2008.198. PMID 18500360.
- http://www.ema.europa.eu/ema/index.jsp?curl=pages/medicines/human/medicines/003898/human_med_001945.jsp&mid=WC0b01ac058001d124
- http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm486827.htm
|
|
|||
| Names | |||
|---|---|---|---|
| IUPAC name
(2S)-2-[(4R)-2-oxo- 4-propylpyrrolidin-1-yl] butanamide
|
|||
| Identifiers | |||
| 357336-20-0 |
|||
| ChEMBL | ChEMBL607400 |
||
| ChemSpider | 8012964 |
||
| Jmol interactive 3D | Image | ||
| PubChem | 9837243 | ||
| UNII | U863JGG2IA |
||
| Properties | |||
| C11H20N2O2 | |||
| Molar mass | 212.15 g/mol | ||
| Pharmacology | |||
| ATC code | N03 | ||
| Legal status |
|
||
| Oral | |||
| Pharmacokinetics: | |||
| Nearly 100% | |||
| <20% | |||
| Hydrolysis, CYP2C8-mediated hydroxylation | |||
| 8 hrs | |||
| >75% renal | |||
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
|
|||
//////BRIVARACETAM, UCB, 2016 FDA, UCB-34714
CCCC1CC(=O)N(C1)C(CC)C(=O)N
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2021260721&_cid=P12-KXX1JU-33531-1
Brivaracetam is chemically known as (2S)-2-[(4R)-2-oxo-4-propyltetrahydro-1H-pyrrol-1-yl] butanamide, having the chemical structure of formula 1 as below:
Brivaracetam is basically a chemical analogue of Levetiracetam, marketed under the brand name of BRIVIACT for the treatment as adjunctive therapy in the treatment of partial-onset seizures in patients at 16 years of age and older with epilepsy. Brivaracetam has an advantage over Levetiracetam in that it gets into the brain “much more quickly,” which means that “it could be used for status epilepticus, or acute seizures than cluster, or prolonged seizures”. From the Phase III trials, the self-reported rate of irritability with Brivaracetam was 2% for both drug doses (100 mg and 200 mg) vs 1% for placebo, which compares to as much as 10% for Levetiracetam in some post-marketing studies.
With the improved safety profile and possibility to be used for wider range of epilepsy, Brivaracetam is considered as one of the most promising 3rd generation antiepileptic drugs.
Brivaracetam molecule is first disclosed in patent publication WO2001062726, which describes 2-oxo-1 -pyrrolidine derivatives and methods for their preparation. This patent publication further discloses compound (2S)-2-[(4R)-2-oxo-4-propyl-pyrrolidin-1-yl] butanamide which is known under the international non propriety name as Brivaracetam. As per Biopharmaceutics Classification System, Brivaracetam is a class I drug (high solubility and permeability).
Some prior arts US6784197 and US7629474 disclose a process for synthesizing a diastereomeric mixture of (2S)-2-[(4R)-2-oxo-4-propylpyrrolidin-1-yl]-butanamide and (2S)-2-[(4S)-2-oxo-4-propylpyrrolidin-1-yl]-butanamide (Brivaracetam) which is purified by chiral HPLC (Scheme-I & Scheme-II respectively, as provided below). This process used for chiral resolution makes it difficult for bulk manufacturing as well as it affects the overall yield making the process uneconomical.
Scheme-I
Synthesis of (2S)-2-(2-oxo-4-propyl-1-pyrrolidinyl)butanamide
[As disclosed in columns 37-38 of US 6784197 B2]
Scheme-II
1.1 Synthesis of (2S)-2-aminobutyramide-Free base
1.2 Synthesis of 5-hydroxy-4-n-propylfuran-2-one
1.3 Synthesis of (2S)-2-((4R)-2-oxo-4-n-propyl-1-pyrrolidinyl)butanamide and
(2S)-2-((4S)-2-oxo-4-n-propyl-1-pyrrolidinyl)butanamide
[As disclosed in columns 6-7 of US 7629474 B2]
Moreover, some prior arts such as US7122682B2, US8076493B2, US8338621B2 and US8957226B2 also describe processes for preparing Brivaracetam, wherein, the purifications are reportedly done by chiral HPLC methods resulting into similar shortcomings.
Kenda et al.: Journal of Medicinal Chemistry, 2004, 47, 530-549 further proposes selection of (2S)-2-[(4R)-2-oxo-4-propylpyrrolidin-1-yl]butanamide 83α (ucb 34714; Brivaracetam) as the most interesting candidate showing 10 times more potency than Levetiracetam as an antiseizure agent in audiogenic seizure-prone mice. This article further discloses methods for synthesizing the said compound Brivaracetam. However, here too these compounds are synthesized as mixtures of stereoisomers (racemic or diastereoisomeric mixtures), separated by preparative HPLC on silica gel and/or chiral phases.
All these processes for the preparation of Brivaracetam as described in the above- mentioned prior arts suffer from many disadvantages which includes difficulty in achieving desired chiral purity, tedious and cumbersome work up procedures, high temperature and longer time reaction, multiple crystallizations or isolation steps, use of excess reagents and solvents, column chromatographic separations & purifications etc. All these disadvantages affect the overall yield as well as the quality of the final product Brivaracetam and intermediates produced thereof; further, rendering such processes to be uneconomical and unsuitable for industrial scale-ups.
As a result, enantioselective synthesis of Brivaracetam was perceived to be a possible way of overcoming such problems in view of the large-scale synthesis. However, very few prior arts have been found to report successful reduction of such concept into practice.
WO2016191435A1 (also as IN201717005820A) relates to a process for a scalable synthesis of enantiomerically pure Brivaracetam from an intermediate (4R)-4- Propyldihydrofuran-2(3H)-one (compound IV):
, wherein, R is saturated or unsaturated C1-20 alkyl or C1 alkyl-unsubstituted or substituted aryl, comprising the steps of decarboxylation of
the compound of formula IV
to produce the compound of formula VI
ring-opening of the compound of formula VI to produce the compound of formula VII
, wherein Rl is saturated or unsaturated Cl-20 alkyl or Cl alkyl-unsubstituted or substituted aryl; and X is CI Br I OMs, OTs, ONs; or
the compound of formula X
reacting the compound of formula VI with (S)-2- aminobutanamide or its salt to produce the compound of formula XII in one step; or reacting the compound of formula VI with alkyl (S)-2- aminobutanoate to produce Xll-a
, wherein R in the compound of formula Xll-a is a saturated or unsaturated C1-C20 alkoxyl; and then converting Xll-a to XII that is Brivaracetam by aminolysis and amide formation reaction.
In above mentioned prior arts, the synthesis of chiral lactone which is the key starting material for making Brivaracetam involved Grignard addition, column chromatography and Krapcho decarboxylation techniques at high temperature, all of which are not at all recommendable in view of process perspective at industrial levels. Further the final step of the said reaction often involved cryogenic condition –30°C which is also difficult with respect to industrial scale up activities.
Furthermore, prior art IN201641030239A disclosed a process for the preparation of Brivaracetam of Formula (I) by means of converting enantiomerically pure compound of Formula VII to obtain enantiomerically pure compound of Formula XI:
, wherein X is each independently selected from halogen; alkyl or aryl sulfonyloxy; OR2; R2 is optionally substituted C1-C12 alkyl, aryl, alkyl aryl, aryl alkyl;
such that the said process further comprises steps of:
1) cyclizing compound of formula VII to give enantiomerically pure compound of formula IX:
, wherein R2 is optionally substituted C1-C12 alkyl, aryl, alkyl aryl or aryl alkyl;
2) converting the compound of formula IX to give a enantiomerically pure compound of formula X:
, wherein X is halogen;
3) converting compound of formula X to give a enantiomerically pure compound of formula XI:
wherein X is each independently selected from halogen; alkyl or aryl sulfonyloxy; OR2; R2 as defined above; followed by 4) treating the enantiomerically pure compound of formula XI with (S)- aminobutyramide of formula XII or its salt thereof to obtain Brivaracetam of Formula I.
However, this process suffered from drawbacks of handling acid chloride. Acid chlorides are unstable and storing a large amount of acid chloride is also not recommendable in view of safety and stability in industries. Moreover, this prior art process involves use of HBr in acetic acid, where HBr liberates Br that is hazardous and not recommendable for industrial scale-up activities in view of safety and handling.
Some other prior arts such as CN108503573A, CN105646319A, CN106588740A, CN106588831A, CN108689903B and CN108929289A report various processes of synthesizing Brivaracetam from its lactone intermediate by various ring opening techniques. Among these, in particular CN108929289A discloses a process of reacting a compound represented by the formula IV with (S) -2-aminobutyramide in order to obtain Brivaracetam. The synthetic route is as follows:
Also, CN108689903B relates to a new preparation method of Brivaracetam that comprises steps of: a) subjecting a compound of formula III and (S) -2-aminobutanamide or salt thereof to condensation reaction, in the presence of a condensing agent, in order to obtain a compound shown in a formula IV, wherein the compound has two chiral centres; b) removal of the hydroxy-protecting group R1 to obtain a compound of formula V; and c) carrying out chlorination reaction on the compound shown in the formula V using a chlorination reagent to obtain a compound shown in the formula VI; and d) carrying out substitution reaction on the compound shown in the formula VI in the presence of an
alkaline reagent, and closing a ring to obtain Brivaracetam of formula I having two chiral centres.
It has further been noted that although the above reaction goes through formation of intermediates V and/or VI; however, these intermediates are not essentially formed from the key lactone intermediate of Brivaracetam that is (4R)-4-propyldihydrofuran-2(3H)-one [or (R)-lactone].
Furthermore, CN111196771A relates to a preparation method of Brivaracetam which comprises the steps of: 1) carrying out ring-opening reaction on a compound R-4-propyldihydrofuran-2-ketone in a formula II and a compound S-2-aminobutanamide in a formula III to obtain an intermediate compound in a formula I; 2) condensing the said intermediate compound of formula I is followed by cyclization to produce Brivaracetam
However, the ring-opening reaction in step 1 of this process essentially occurs under acidic conditions, specifically in presence of Lewis acids like tetra-isopropyl titanate, anhydrous aluminium trichloride, anhydrous zinc chloride, boron trifluoride diethyl etherate etc.; and also in presence of organic solvents chosen from one or more of anhydrous tetrahydrofuran, 2-methyltetrahydrofuran, acetone, dimethyl sulfoxide and N, N-dimethylformamide; which makes this process both industrially non-scalable and environmentally unfriendly.
The prior art Org. Process Res. Dev.2016. v 20. no 9. p 1566-1575 in its scheme 4, on page 17 also discloses a scheme for synthesizing Brivaracetam from its lactone intermediate:
Nevertheless, it has been noted that the process reported in this prior art provides Brivaracetam (API) with a very poor yield of ~30% and also having an inferior chiral purity of 95.9% ee, which does not even meet the ICH-specification for the Finished Product (API).
Furthermore, a recently filed patent application WO2020148787A1 (also as IN201931002041) recites a new, improved and economical process for enantioselective synthesis and purification of a key intermediate of Brivaracetam that is the R-lactone, essentially utilizing a low chiral loading and without involving any chiral chromatographic resolution technique. Even though this prior art also discloses a process for the preparation of a chirally pure Brivaracetam of formula I utilizing the said intermediate; however, that process is mostly a conventional one.
Accordingly, there is still a need in the art for a more economical and improved process for the synthesis of Brivaracetam with better purity and yield which overcomes the drawbacks of above prior arts.
Therefore, the present inventors have developed a cost effective, novel and efficient process for the preparation of Brivaracetam which essentially avoids all the drawbacks involved in prior art as mentioned above. The currently developed process is advantageously capable of producing the key lactone intermediate with more than 80% ee applying transfer hydrogenation with a very simple operation in view of process perspective. Further, by means of using such chiral lactone with more than 80% ee, the currently developed process is also capable of delivering >99.9% chirally pure Brivaracetam with excellent yield.
EXAMPLES:
EXAMPLE 1: Synthesis of (3R)-N-[(1S)-1-carbamoylpropyl]-3-(hydroxymethyl) hexanamide [Intermediate 7 of scheme A of the present invention]
Example 1 illustrates one pot process for preparing purified (3R)-N-[(1S)-1- carbamoylpropyl]-3-(hydroxymethyl) hexanamide [Intermediate 7] from Intermediate 3 (80% ee) as developed in step 1 of scheme A of the present invention.
Procedure:
In the first step of scheme A of the present invention, a mixture of (R)/(S)-4-propyldihydrofuran-2(3H)-one (Intermediate 3, R: S isomer = 80: 20) (1 eq), (S)-2-aminobutanamide (1.1 eq), triethylamine (1.5 eq) is refluxed at a temperature of 95±5 °C for 24h. The mixture is then cooled to 60-65 °C, washed with a mixture of dichloromethane and diisopropyl ether (2.5 vol) in order to get Intermediate-7 [(3R)-N-[(1S)-1-carbamoyl-propyl]-3-(hydroxymethyl) hexanamide] (80% yield).
Results:
Formation of Intermediate 7 is confirmed further by following analytical studies: a) The 1H NMR analysis is conducted and the data as illustrated in accompanying figure 1 depicts: (400 MHz, DMSO-d6): δ 0.6 (t, J= Hz, 6H), 1.07-1.18 (m, 1H), 1.21-1.35 (m, 3H), 1.45-1.43 (m, 1H), 1.61-1.72 (m, 1H), 1.75-1.90 (m, 1H), 2.03 (dd, J=6.64 & 14.08 Hz, 1H), 2.18 (dd, J=7.0 & 14.08 Hz, 1H), 3.28 (t, J=5.36 Hz, 2H), 4.07-4.18 (m, 1H), 4.43 (t, J=5.2 Hz, 1H), 6.95 (s, 1H), 7.28 (s, 1H), 7.76 (d, J=8.08 Hz, 1H).; thus, confirming formation of Intermediate 7 of the present invention.
b) The LCMS analysis is further conducted and the data as graphically illustrated in accompanying figure 2 provides a (M+H+) value of 231.0; thus, confirming formation of Intermediate 7 of the present invention.
c) The HPLC study is also conducted and the data as graphically illustrated in accompanying figure 3 confirms formation of Intermediate 7 of the present invention with chiral purity of 97.38%
EXAMPLE 2: Preparation of (3R)-N-(1S)-1-amino-1-oxobutan-2-yl)-3-(chloromethyl) hexanamide [Intermediate 8A of scheme A of the present invention]: Example 2 illustrates a process for preparing (3R)-N-(1S)-1-Amino-1-oxobutan-2-yl)-3-(chloromethyl) hexanamide [Intermediate 8A] from Intermediate 7 of example 1 above as developed in the present invention.
Procedure:
In second step of scheme A of the present invention, the said Intermediate 7 of example 1 above that is (3R)-N-(1S)-1-Amino-1-oxobutan-2-yl)-3-(hydroxymethyl) hexanamide (~98% Chemical purity and ~97% Chiral purity) (1736.86 mmol) is dissolved in DCM (1.2 L) at RT into a RBF under N2 atm. Then the solution is cooled to 10-20 C and Oxaloyl chloride (2605.29 mmol) is added to this cooled solution at 10-20 °C. The mixture is stirred for 24 h at 25-40 °C under N2 atm. Completion of the reaction is monitored by TLC. After completion of reaction, the solvent is distilled off and the residual mass is diluted with water (6 L), stirred at 30-50 °C for 4 h. Slurry mass is then filtered and washed with water (2×400 mL) followed by MTBE (800 mL). The solid is dried under vacuum at 50-55 °C for 4-5 h to afford Intermediate 8A that is (3R)-N-(1S)-1-Amino-1-oxobutan-2-yl)-3-(chloromethyl) hexanamide as a white solid (92% yield).
Results:
Formation of Intermediate 8A is confirmed further by following analytical studies: a) The 1H NMR analysis is conducted and the data as illustrated in accompanying figure 4 depicts: 1H NMR (400 MHz, DMSO-d6) : δ 0.83 (t, J=7.44 Hz, 3H), 0.85 (t, J=6.72 Hz, 3H), 1.20-1.40 (m, 4H), 1.43-1.56 (m, 1H), 2.08-2.18 (m, 1H), 2.20-2.28 (m, 2H), 3.61 (dd, J=4.6 & 10.8 Hz, 1 H), 3.67 (dd, J=4.6 & 10.8 Hz, 1H), 4.07-4.18 (m, 1H), 6.95 (s, 1H), 7.29 (s, 1H), 7.89 (d, J=8.12 Hz, 1H); thus confirming formation of Intermediate 8A of the present invention.
b) The LCMS analysis is further conducted and the data as graphically illustrated in accompanying figure 5 (a, b) provides a (M+H+) value of 249.20; thus, confirming formation of Intermediate 8A of the present invention.
EXAMPLE 3: Process for purification of Intermediate 8A forming Intermediate 8B Example 3 illustrates a process for purifying the said Intermediate 8A of example 2 above of the present invention.
Procedure:
The Intermediate 8A as obtained in example 2 above [that is (3R)-N-(1S)-1-Amino-1-oxobutan-2-yl)-3-(chloromethyl) hexanamide] is first dissolved in a polar solvent like Acetonitrile raising the temperature to 50 to 60 °C; followed by stirring and then addition of another solvent methyl tert-butyl ether (MTBE) which is less polar in nature. The mixture is then cooled down to 0°C, the filtered mass thus obtained is dried in order to obtain a white solid of Intermediate 8B. The material thus obtained is further dissolved in THF (5 vol) at 60 °C, cooled to 20-30°C, followed by addition of heptane (5 vol), stirred at 10°C to 30 °C for 1 h. The mass obtained is filtered and washed with heptane (2×1 vol), dried under vacuum at 50-55°C in order to afford formation of purer form of Intermediate 8A that is Intermediate 8B that is (3R)-N-(1S)-1-amino-1-oxobutan-2-yl)-3-(chloromethyl) hexanamide as a white solid having a chemical purity of 99.9% along with a chiral purity of 100% (yield: 390 g).
Results:
The purification of Intermediate 8A is further confirmed by the following analytical test results:
a) Chiral HPLC: A Chiral HPLC as illustrated in accompanying figure 6 confirmed formation of purest form of Intermediate 8B having 100% chiral purity [Peak 1; RT (min) = 6.244; %Area=100%].
b) GLP-HPLC: A GLP-HPLC as illustrated in accompanying figure 7 further confirmed formation of Intermediate 8B having 99.9% chemical purity [Peak 3; BRIV8; RT = 29.278; % Area=99.90%].
EXAMPLE 4: Synthesis of (3R)-N-[(1S)-1-carbamoylpropyl]-3-(hydroxymethyl) hexanamide [Intermediate 7’ of scheme B of the present invention]
Example 4 illustrates one pot process for preparing purified (3R)-N-[(1S)-1-carbamoylpropyl]-3-(hydroxymethyl) hexanamide [Intermediate 7’] from Intermediate 6 (99.99% ee) as developed in step-1 scheme B of the present invention.
Procedure:
In another method, in the first step of scheme B of the present invention, a mixture of (R)/(S)-4-propyldihydrofuran-2(3H)-one (Intermediate 6: S isomer = 99.99% : 0.1%) (1 eq), (S)-2-aminobutanamide (1.7 eq), triethylamine (5 eq) is refluxed at a temperature between 95±5 °C for 24 h. Then, the crude reaction mass is cooled and washed with dichloromethane and diisopropyl ether mixture (2.5 vol) in order to achieve Intermediate-7’ of scheme B of the present invention [(3R)-N-[(1S)-1-carbamoylpropyl]-3-(hydroxymethyl) hexanamide] (90% yield).
Results:
The chiral purity of the formed Intermediate 7’ is analyzed by HPLC method and the data as graphically illustrated in accompanying figure 8 confirms formation of Intermediate 7’ of the present invention with chiral purity of 99.11%
EXAMPLE 5: Preparation of (3R)-N-(1S)-1-amino-1-oxobutan-2-yl)-3-(chloromethyl) hexanamide [Intermediate 8’ of scheme B of present invention]: Example 5 illustrates a process for preparing purest form of (3R)-N-(1S)-1-amino-1-oxobutan-2-yl)-3-(chloromethyl) hexanamide [Intermediate 8’] from Intermediate 7’ of example 4 as developed in scheme B (step 2) of the present invention.
Procedure:
In the second step of scheme B of the present invention, the intermediate 7’ of the above example 4 that is (3R)-N-(1S)-1-amino-1-oxobutan-2-yl)-3-(hydroxymethyl) hexanamide (98% Chemical purity and >99%Chiral purity) (1736.86 mmol) is dissolved in DCM (1.2 L) at RT in a round bottomed flask under N2 atm. Then the solution is cooled to 10-30 °C and 1-Chloro-N,N,2-trimethyl-1-propenylamine (2605.29 mmol) is added to this cooled solution at 10-30 °C. The mixture is stirred for 24 h at 25-40 °C under N2 atm. Completion of the reaction is monitored by TLC. After completion of the reaction, the solvent is distilled off and the residual mass is diluted with water (6 L), stirring at 30-50 °C for 4 h. The slurry mass thus obtained is then filtered and washed with water (2×400 mL) followed by methyl tert-buty ether (MTBE) (800 mL). The solid thus obtained is dried under vacuum at 50-55 °C for 4-5 h in order to afford formation of Intermediate 8’ that is (3R)-N-(1S)-1-amino-1-oxobutan-2-yl)-3-(chloromethyl) hexanamide as a white solid (92% yield).
Results:
Formation of Intermediate 8’ is confirmed further by following analytical studies: a) The 1H NMR analysis is conducted and the data as illustrated in accompanying figure 9 depicts: (400 MHz, DMSO-d6): 1H NMR (400 MHz, DMSO-d6) : δ 0.83 (t, J=7.44 Hz, 3H), 0.85 (t, J=6.72 Hz, 3H), 1.20-1.40 (m, 4H), 1.43-1.56 (m, 1H), 2.08-2.18 (m, 1H), 2.20-2.28 (m, 2H), 3.61 (dd, J=4.6 & 10.8 Hz, 1 H), 3.67 (dd, J=4.6 & 10.8 Hz, 1H), 4.07-4.18 (m, 1H), 6.95 (s, 1H), 7.29 (s, 1H), 7.89 (d, J=8.12 Hz, 1H); thus, confirming formation of Intermediate 8’ of the present invention.
b) The LCMS analysis is further conducted and the data as graphically illustrated in accompanying figure 10 provides a (M+H+) value of 249.1; thus, confirming formation of Intermediate 8’ of the present invention.
c) The HPLC data as illustrated in accompanying figure 11 confirms 100% chiral purity of Intermediate 8’.
EXAMPLE 6: Preparation of (2S)-2-[(4R)-2-oxo-4-propyl-pyrrolidin-1-yl] butanamide [Brivaracetam-API]:
Example 6 illustrates a process for preparing (2S)-2-[(4R)-2-oxo-4-propyl-pyrrolidin-1-yl] butanamide [Brivaracetam API] from Intermediate 8B of example 3 or from Intermediate 8’ of example 5 as developed in step 3 of scheme A or scheme B of the present invention respectively.
Procedure:
In the final step of scheme A or scheme B of the present invention, the intermediate 8B of example 3 or intermediate 8’ of example 5 above that is (3R)-N-((1S)-1-Amino-1-oxobutan-2-yl)-3-(chloromethyl) hexanamide (1608.04 mmol) is dissolved in dimethyl acetamide (0.5 vol) and isopropylacetate (2 L) into a RBF at 25-30 °C under N2 atm. Then 18-Crown-6 (160.79 mmol) is added into the solution and stirred at RT for 30 min. Reaction mixture is then cooled to 0-10 °C and t-BuOK (1.5 eq) is added portion wise to the cooled solution over 1 h maintaining the temperature from – 0-10 °C to 25 °C under N2 atm. Stirring is then continued for 2 h at -10 °C to 0 °C and then for 12 h at 15-25 °C under N2 atm. Completion of reaction is monitored by TLC. After completion of reaction, the reaction mixture is quenched with addition of 1M HCl solution (pH~6.5-7.0). The resulting mixture is extracted with i-PrOAc (2 L) and MTBE (1 L). Water (0.5 L) is added to the combined organic extract and then filtered through celite bed, washed the bed with MTBE-i-PrOAc (1:1) (400 mL). The organic part is separated and the aqueous part is re-extracted with i-PrOAc-MTBE (1:1) (2 ×0.8 L). The combined organic phases are washed with brine solution (100 mL), dried over anhydrous Na2SO4, filtered and concentrated in vacuum under a rotary evaporator to afford crude API. Distillation of dimethylacetamide solvent from the crude is then done at high vacuum pressure (0.05 mm Hg) at 70 °C. Crude product is then dissolved in isopropyl acetate (1.6 L) and treated with activated charcoal (7% w/w) to afford a tech-grade crude of Brivaracetam API as a white solid (yield: 90%) with 97.82% chemical purity.
Results:
Formation of Brivaracetam API is confirmed further by following analytical studies: a) The 1H NMR analysis is conducted and the data as illustrated in accompanying figure 12 depicts: 1H NMR (400 MHz, DMSO-d6) : δ 0.77 (t, J=7.32 Hz, 3H), 0.87 (t, J=7.2 Hz, 3H), 1.21-1.31 (m, 2H), 1.33-1.43 (m, 2H), 1.50-1.62 (m, 1H), 1.73-1.84 (m, 1H), 1.97 (dd, J=8.0 & 16.12 Hz, 1H), 2.18-2.28 (m, 1H), 2.37 (dd, J=8.4 & 16.14 Hz, 1H), 3.11 (dd, J=7.16 & 9.44 Hz, 1H), 3.36 (dd, J=9.2 & 17.5 Hz, 1H), 4.30 dd, J=5.44 & 10.28 Hz, 1H), 6.98 (s, 1H), 7.32 (s, 1H); thus, confirming formation of Brivaracetam API of the present invention.
b) The LCMS analysis is further conducted and the data as graphically illustrated in accompanying figure 13 provides a (M+H+) value of 213.0; thus, confirming formation of Brivaracetam API of the present invention.
^ Purification of Brivaracetam API:
The Brivaracetam thus formed above is further purified by means of dissolving the said material (307 g) in 30% i-PrOAc -MTBE (1 vol) at 55-60 °C, cool to 20-30°C. A mixture of Heptane and MTBE and DIPE (2:2:1) is added, stirred at 10 °C to 30°C for 1 h. The obtained mass is filtered and washed with heptane, which is subsequently dried under vacuum at 40-45 °C to afford (3R)-N-((1S)-1-amino-1-oxobutan-2-yl)-3-(chloromethyl) hexanamide as a white solid (yield: 80%, chiral purity 99.93% and chemical purity 99.94%).
Results:
a) Chiral HPLC: A Chiral HPLC as illustrated in accompanying figure 14 confirmed formation of purest form of Brivaracetam API having 99.93% chiral purity [Peak 2; RT (min) = 9.45; %Area=99.93%].
b) GLP-HPLC: A GLP-HPLC as illustrated in accompanying figure 15 further confirmed formation of Brivaracetam API having 99.9% chemical purity [Peak 2; RT = 21.138; % Area=99.94%].
DRUG SPOTLIGHT…LEVETIRACETAM

LEVETIRACETAM, etiracetam
(-)-(S)-α-ethyl-2-oxo-1-pyrrolidine acetamide
(−)-(S)-alpha-ethyl-2-oxo-1-pyrrolidineacetamide
CAS…102767-28-2
Crystals from ethyl acetate, mp 117°. [a]25D -90.0° (c = 1 in acetone). Soly (g/100 ml): water 104.0; chloroform 65.3; methanol 53.6; ethanol 16.5; acetonitrile 5.7. Practically insol in n-hexane. LD50 in male mice, male rats (mg/kg): 1081, 1038 i.v. (Gobert, 1990).
Mp: mp 117°C
| Active Ingredient: | LEVETIRACETAM |
| Dosage Form;Route: | INJECTABLE;IV (INFUSION) |
| Proprietary Name: | KEPPRA |
| Applicant: | UCB INC |
| Strength: | 500MG/5ML (100MG/ML) |
| Application Number: | N021872 |
| Product Number: | 001 |
| Approval Date: | Jul 31, 2006 |
| Reference Listed Drug | Yes |
| RX/OTC/DISCN: | RX |
Levetiracetam is an anticonvulsant medication used to treat epilepsy. Levetiracetam may selectively prevent hypersynchronization of epileptiform burst firing and propagation of seizure activity. Levetiracetam binds to the synaptic vesicle protein SV2A, which is thought to be involved in the regulation of vesicle exocytosis. Although the molecular significance of levetiracetam binding to synaptic vesicle protein SV2A is not understood, levetiracetam and related analogs showed a rank order of affinity for SV2A which correlated with the potency of their antiseizure activity in audiogenic seizure-prone mice.
Epilepsy is a chronic neurological disorder that consists of repeated occurrences of spontaneous seizures. Levetiracetam, [(S)-a-ethyl-2-oxopyrrolidine acetamide], has recently been approved as an add-on therapy for the treatment of refractory epilepsy . The (S)-enantiomer of etiracetam (levetiracetam) has shown remarkable pharmacokinetic and pharmacological activity which has led to the quick approval of this antiepileptic drug by the FDA.
Levetiracetam offers several advantages over traditional therapy, including twice-daily dosing, a wide margin of safety with no requirements for serum drug concentration monitoring and no interactions with other anticonvulsants, and less adverse effects than traditional treatments
Levetiracetam (INN) /lɛvɨtɪˈræsɨtæm/ is an anticonvulsant medication used to treatepilepsy. It is the S-enantiomer of etiracetam, structurally similar to the prototypicalnootropic drug piracetam.
Levetiracetam is marketed under the trade name Keppra. Keppra is manufactured by UCB Pharmaceuticals Inc. Since November 2008 the drug has been available as a genericbrand in the United States.
Levetiracetam has been approved in the European Union as a monotherapy treatment for epilepsy in the case of partial seizures, or as an adjunctive therapy for partial, myoclonicand tonic-clonic seizures. It is also used in veterinary medicine for similar purposes.
Levetiracetam has potential benefits for other psychiatric and neurologic conditions such as Tourette syndrome, autism, bipolar disorder and anxiety disorder, as well asAlzheimer’s disease. However, its most serious adverse effects are behavioral, and its benefit-risk ratio in these conditions is not well understood.
Along with other anticonvulsants like gabapentin, it is also sometimes used to treatneuropathic pain. It has not been found to be useful for essential tremors.
Levetiracetam (LEV) is a novel antiepileptic drug (AED) which was discovered in early 1980s and soon, in 1999 FDA approved LEV for the management of partial onset seizure. In India, LEV tablet was approved in April 2005. It acts by binding to the synaptic vesicle protein SV2A, which is present on synaptic vesicles and some neuroendocrine cells. Pharmacokinetics of LEV such as, less protein binding and lack of hepatic metabolism makes LEV less susceptible to drug interactions with other anticonvulsants. Evidence also suggests that LEV is much better than other AEDs in the way of broad therapeutic window, convenient dosing and less adverse effect. Besides the pharmacological effects, pharmacoeconomically also, LEV is a beneficial drug. All these valuable pharmacological and pharmacoeconomic aspect makes LEV an important option in management of various types of epilepsy.

- PubMed Health A division of the National Library of Medicine at the National Institutes of Health.
- Keppra (levetiracetam) Final Printed Label April 2009. Center for Drug Evaluation and Research, U.S. Food and Drug Administration. Accessed 29 July 2011.
- Keppra UCB (manufacturer’s website)
- NIH MedLine drug information
KEPPRA injection is an antiepileptic drug available as a clear, colorless, sterile solution (100 mg/mL) for intravenous administration.
The chemical name of levetiracetam, a single enantiomer, is (-)-(S)-α-ethyl-2-oxo-1-pyrrolidine acetamide, its molecular formula is C8H14N2O2 and its molecular weight is 170.21. Levetiracetam is chemically unrelated to existing antiepileptic drugs (AEDs). It has the following structural formula:
 |
Levetiracetam is a white to off-white crystalline powder with a faint odor and a bitter taste. It is very soluble in water (104.0 g/100 mL). It is freely soluble inchloroform (65.3g/100 mL) and in methanol (53.6 g/100 mL), soluble in ethanol (16.5 g/100 mL), sparingly soluble in acetonitrile (5.7 g/100 mL) and practically insoluble in n-hexane. (Solubility limits are expressed as g/100 mL solvent.)
KEPPRA injection contains 100 mg of levetiracetam per mL. It is supplied in single-use 5 mL vials containing 500 mg levetiracetam, water for injection, 45 mg sodium chloride, and buffered at approximately pH 5.5 with glacial acetic acid and 8.2 mg sodium acetate trihydrate. KEPPRA injection must be diluted prior to intravenous infusion

(S)-(−)-α-ethyl-2-oxo-1-pyrrolidine acetamide, which is referred under the International Nonproprietary Name of Levetiracetam, its dextrorotatory enantiomer and related compounds. Levetiracetam is shown as having the following structure:
Levetiracetam, a laevorotary compound is disclosed as a protective agent for the treatment and the prevention of hypoxic and ischemic type aggressions of the central nervous system in the European patent No. 162036. This compound is also effective in the treatment of epilepsy, a therapeutic indication for which it has been demonstrated that its dextrorotatory enantiomer (R)-(+)-α-ethyl-2-oxo-1-pyrrolidine acetamide completely lacks activity (A. J. GOWER et al., Eur. J. Pharmacol., 222, (1992), 193-203). Finally, in the European patent application No. 0 645 139 this compound has been disclosed for its anxiolytic activity.
The asymmetric carbon atom carries a hydrogen atom (not shown) positioned above the plane of the paper. The preparation of Levetiracetam has been described in the European patent No. 0162 036 and in the British patent No. 2 225 322, both of which are assigned to the assignee of the present invention. The preparation of the dextrorotatory enantiomer (R)-(+)-α-ethyl-2-oxo-1-pyrrolidine acetamide has been described in the European patent No. 0165 919.
-
Several processes for obtaining levetiracetam have been disclosed. One promising approach is the reaction of (S)-2-aminobutyramide (5) with an alkyl 4-halobutyrate or with a 4-halobutyryl halide followed by cyclization as outlined in EP 162036 . Clearly, said (S)-2-aminobutyramide (5) is a key intermediate in the preparation of levetiracetam and given the importance of the correct stereochemistry of levetiracetam also the correct stereochemistry in the key intermediates is of importance.
-
The separation of stereoisomers is considered to be one of the difficult tasks in chemistry since chiral compounds exhibit identical physical properties in non-chiral environments. Although several approaches for the preparation of optically pure (S)-2-aminobutyramide (5) have been reported, many of these are related to resolution of racemic (R,S)-2-aminobutyramide (e.g. WO 2006/103696 ), optionally using catalytic amounts of an aldehyde such as described in JP 2007/191470 . However, an approach directly starting from the Schiff base of racemic (R,S)-2-aminobutyramide (i.e. compound (1)) is unavailable whereas there is a need for this as said Schiff bases are highly suitable from a preparative point of view as these compounds may be conveniently isolated from the aqueous media that they are usually prepared in. This is in contrast with the parent 2-aminobutyramide which is highly soluble in water and consequently difficult to obtain in sufficient purity.
British Pat. No. 1,309,692 describes the compound α-ethyl-2-oxo-l- pyrrolidineactamide (melting point 122 degrees C.) and states that compounds of this type can be used for therapeutic purposes, for example for the treatment of motion sickness, hyperkinesia, hypertonia and epilepsy.
-
The same document discloses obtaining levetiracetam by reacting (S)-2-aminobutanamide with an alkyl 4-halobutyrate or with a 4-halobutyryl halide, and subsequent cyclization of alkyl (S)-4-[[1-(aminocarbonyl)propyl]amino]butyrate or of (S)-N-[1-(aminocarbonyl)propyl]-4-halobutanamide thus obtained, as summarized in the attached scheme:
-
The two previous processes have the drawback of operating at temperatures between -10°C and -60°C and the drawback of using intermediates for cyclization that are not readily obtained.
-
A drawback of this industrial-scale process is that it requires special equipment and special precautions for handling the products.
-
Other processes are known (for example US patents No 6,107,492 and6,124,473 ) in which levetiracetam is obtained by means of optical resolution of racemic etiracetam of formula (I). InUS patent No 6,107,492 resolution is performed by means of preparative high performance liquid chromatography or by means of a continuous simulated fluid bed chromatographic system with a chiral stationary phase. US patent No 6,124,473 discloses a continuous simulated fluid bed chromatographic system consisting of at least three chiral stationary phase columns. These industrial-scale resolution processes are affected by drawbacks related to using chromatography.
-
The industrial-scale difficulties and hazard of hydrogenation can be mentioned in relation to these processes.
-
Finally, patent application ES 447,346 describes a process for the preparation of a pyrrolidone derivative, in particular the 2-oxo-1-pyrrolidinylacetamide, which comprises first reacting pyrrolidone with formaldehyde and a secondary amine, then reacting the compound obtained with an alkylating agent such as dimethyl sulfate, followed by treating the compound obtained with sodium or potassium cyanide, and finally reacting the compound obtained with hydrogen peroxide in basic medium.
Moreover, it also mentions that these compounds can be applied in the field of memory disorders in normal or pathological conditions.
It is also known that α-ethyl-2-oxo-l-pyrrolidineacetamide possesses a protective activity against aggressions of the central nervous system caused by hypoxias, cerebral ischemia, etc. (Pharmazie, 37/11, (1982), 753-765).
U.S. patent 4,969,943 discloses the levorotatory isomer of α-ethyl-2-oxo-l- pyrrolidineacetamide, which has the absolute S configuration, a method for making the isomer and pharmaceutical compositions containing the same. U.S. patent 4,696,943 discloses that the levorotatory isomer has a 10 times higher protective activity against hypoxia and a 4 times higher protective activity against ischemia compared to the known racemic form.

 http://oasys2.confex.com/acs/229nm/techprogram/P831117.HTM
http://oasys2.confex.com/acs/229nm/techprogram/P831117.HTM
Parallel synthesis of Levetiracetam (Keppra®) and its analogs via an Ugi-RCM strategy
…………………………

http://www.google.com/patents/US7939676
Levetiracetam, (−)-(S)-alpha-ethyl-2-oxo-1-pyrrolidineacetamide, is a drug useful as a protective agent for treating and preventing hypoxic and ischemic type aggressions of the central nervous system. It is the active ingredient of KEPPRA®, tablets and flavored liquid, indicated as adjunctive therapy in the treatment of partial onset seizures in adults and children four years of age and older with epilepsy.
Levetiracetam was first described in U.S. Pat. No. 4,837,223 (UCB Societe Anonyme) where it is stated that it has particular therapeutic properties compared to the known racemic form (non proprietary name etiracetam). The S-enantiomer, for example, has a ten times higher protective activity against hypoxia and a four times higher protective activity against cerebral ischemia than the racemic mixture.
The U.S. Pat. No. 4,837,223 describes a method for the preparation of levetiracetam which comprises reacting (−)-(S)-alpha-ethyl-2-oxo-1-pyrrolidineacetic acid successively with alkylhaloformate and with ammonia. Said acid intermediate is, in turn, obtained from racemic (±)-alpha-ethyl-2-oxo-1-pyrrolidine acetic acid by a classic optical resolution according to known methods. In example 1, ethyl (±)-alpha-ethyl-2-oxo-1-pyrrolidine acetate is hydrolyzed to give the corresponding racemic acid in the presence of sodium hydroxide; said acid is subjected to chemical resolution by reaction with an optically active base, (+)-(R)-(1-phenyl ethyl)-amine, selective crystallization of diastereoisomeric salts thereof and isolation of the desired enantiomeric form; finally, the resultant (−)-(S)-alpha-ethyl-2-oxo-1-pyrrolidineacetic acid is converted into the corresponding amide via activation of the carboxyl residue with ethyl chloroformate.
Several alternative processes for the preparation of levetiracetam have been disclosed in the art.
GB 1,309,692 (UCB S.A.) describes the preparation of several N-substituted lactams, including, inter alia, 2-(2-oxo-pyrrolidino)-butyramide, i.e. the racemic form of levetiracetam, by converting the corresponding ester, obtained by reacting the appropriate pyrrolidin-2-one with an appropriate alkyl haloalkylcarboxylate, with gaseous ammonia in methanol (example 2) or by converting the corresponding acid chloride, obtained by reacting the corresponding acid with thionyl chloride, with gaseous ammonia (example 3).
WO 01/64637 (UCB Farchim) describes the preparation of levetiracetam by asymmetric hydrogenation in the presence of a chiral catalyst of (Z) or (E)-2-(2-oxotetrahydro-1H-1-pyrrolyl)-2-butenamide, which in turn is obtained by reacting the corresponding acid with PCl5 to give the corresponding acid chloride, and then with gaseous ammonia.
WO 03/014080 (UCB S.A.) describes a process for the preparation of levetiracetam and analogues thereof comprising the synthesis of the corresponding ester derivative, methyl-(S)-alpha-ethyl-2-oxo-1-pyrrolidine-acetate, and the subsequent ammonolysis reaction in the presence of water.
EP 1,566,376 (REDDYS LAB LTD DR) discloses a process for the preparation of levetiracetam by reacting 4-chlorobutyl chloride with (S)-2-Aminobutyramide hydrochloride, this latter being obtained by first reacting (5)-2-aminobutyric acid hydrochloride with thionyl chloride in methanol to give the corresponding ester hydrochloride, and then reacting the corresponding ester with ammonia in isopropanol.
Several other patents and patent applications describe other approaches to the synthesis of levetiracetam, such as, for example, U.S. Pat. Nos. 6,107,492 and 6,124,473 which describe the preparation of levetiracetam by optical resolution of etiracetam by means of preparative high performance liquid chromatography or continuous simulated moving bed chromatographic system, GB 2,225,322, which describes a process for the preparation of levetiracetam by hydrogenolysis of (S)-alpha-[2-(methylthio)-ethyl]-(2-oxo-1-pyrrolidine)-acetamide in the presence of a desulfurizing agent, and WO 2004/069796, which describes a process for preparing levetiracetam which comprises reacting (S)-2-aminobutyrramide hydrochloride and 4-chlorobutyl chloride in a solvent selected from acetonitrile and methyl tertbutyl ether in the presence of a strong base and recovering the crude product.
EXAMPLE 1Invention
Step 1
(−)-(S)-alpha-ethyl-2-oxo-1-pyrrolidine acetic acid (150 g, 0.87 mol) was dissolved in methanol (235 g, 300 ml) at 45° C. and thionyl chloride (56 g, 0.47 mol) was added dropwise over 30 min.
The reaction mixture was stirred at 45° C. for additional 15-30 min until complete conversion of (−)-(S)-alpha-ethyl-2-oxo-1-pyrrolidine acetic acid was observed via HPLC (unreacted (−)-(S)-alpha-ethyl-2-oxo-1-pyrrolidine acetic acid ≦2%, by HPLC % area).
At reaction completed, the volatiles were distilled off at moderate temperature and reduced pressure (35°-40° C., 150-200 mbar) until 10% of the whole volume was eliminated, then the mixture was reintegrated with fresh methanol up to initial volume.
After that, the reaction mixture was neutralized by bubbling ammonia gas at 20° C. up to a pH value equal to about 5, and stirred at 20° C. for 1 h. A limited amount of salts (about 44 g) precipitated and was filtered off. The resulting methanol solution was directly transferred to the autoclave.
Step 2
The reaction mixture was pressurized up to about 3 bar with ammonia gas at 20° C., and stirred until complete conversion to (−)-(S)-alpha-ethyl-2-oxo-1-pyrrolidineacetamide was observed via HPLC.
Then, once the reaction mixture was taken out of the autoclave, the residual salts formed (about 20 grams) were filtered off and the methanol solution was distilled up to a minimum volume at moderate temperature and reduced pressure (35°-40° C., 150-200 mbar).
Acetone (115 ml) was added and the mixture was distilled again at moderate temperature and reduced pressure (35°-40° C., 150-200 mbar) to minimum volume. After that acetone (300 ml) was charged over the residue and the mixture was heated and refluxed for 30 minutes. Finally, the solution was cooled down slowly to 0° C. and crude levetiracetam was isolated by filtration.
Crude levetiracetam (molar yield 73.1%, (R)-enantiomer: 1.171%) was then submitted to a final purification process in one step to give pure levetiracetam.
Acetone (750 ml) was charged over crude levetiracetam and the mixture was again stirred and heated to reflux. Once refluxed for about 30 minutes the hot mixture was filtered to remove residual salts and cooled slowly to 0° C.
Pure levetiracetam ((R)-enantiomer: 0.01%) was obtained by filtration and drying under vacuum at 40° C. Overall molar yield was 60.0% by mole of the starting amount of (−)-(S)-alpha-ethyl-2-oxo-1-pyrrolidine acetic acid (ponderal yield 78.4% by weight).
EXAMPLE 2
Example 1 was repeated, but the neutralization step with ammonia at the end of step 1 was omitted. At the end of step 2, crude levetiracetam was isolated (molar yield 73.1%, (R)-enantiomer: 2.21%). After purification step, pure levetiracetam (molar yield 64.4%, (R)-enantiomer: 0.58%) was obtained.
EXAMPLE 3Comparison
Step 1 of Example 1 was repeated using an excess of thionyl chloride (114 g, 0.96 mol) with respect to (−)-(S)-alpha-ethyl-2-oxo-1-pyrrolidine acetic acid. Further, when the complete conversion of (−)-(S)-alpha-ethyl-2-oxo-1-pyrrolidine acetic acid was observed, the reaction mixture was distilled off at moderate temperature and reduced pressure (35°-40° C., 150-200 mbar) until dryness. Decomposition of about 13% by weight of the intermediate product to starting product (−)-(S)-alpha-ethyl-2-oxo-1-pyrrolidine acetic acid was observed.
EXAMPLE 4Effect of Activating Agent Amount
Example 1 was repeated using different amount of thionyl chloride as reported in the following Table 1. The amount of thionyl chloride is expressed in terms of equivalent with respect to (−)-(S)-alpha-ethyl-2-oxo-1-pyrrolidine acetic acid.
| TABLE 1 | ||||
| Conversion | Unreacted (% | Converted (% | ||
| Sample | SOCl2 | Time (hours) | w/w) | w/w) |
| 1 | 1.10 | 1 | 0.1 | 99.9 |
| 2 | 0.78 | 0.5 | 0.4 | 99.7 |
| 3 | 0.60 | 1 | 1.1 | 99.3 |
| 4 | 0.54 | 0.5 | 1.1 | 99.3 |
| 5 | 0.29 | 2 | 2.2 | 98.4 |
| 6 | 0.09 | 5 | 4.9 | 96.7 |
| 7 | 0.05 | 24 | 1.7 | 99.0 |
The data of Table 1 clearly show that the use of a substoichiometric amount of thionyl chloride (samples 2 to 5) does not substantially affect the conversion time and conversion yield of (−)-(S)-alpha-ethyl-2-oxo-1-pyrrolidine acetic acid. On the contrary, the use of catalytic amount of thionyl chloride (samples 6 and 7) substantially increases the conversion time and/or the conversion yield.
………………………..

Compound (I) can also be condensed with 4-chlorobutyryl chloride (IV) either directly in the presence of tetrabutylammonium bromide (TBAB) in dichloromethane, followed by in situ treatment with potassium hydroxide, or via the isolation of intermediate (S)-N-[1-(carbamol)propyl]-4-chlorobutyramide (V).
……………..

An alternative procedure involves hydrolysis of racemic ethyl 2-(2-oxopyrrolidin-1-yl)burytate (VI) with sodium hydroxide to produce racemic 2-(2-oxopyrrolidin-1-yl)butyric acid (VII), which is resolved by fractional crystallization with (R)-(+)-alpha-methylbenzylamine in benzene, followed by acid-base treatment to give (S)-2-(2-oxopyrrolidin-1-yl)butyric acid (VIII). Compound (VIII) is finally treated with ethyl chloroformiate and ammonia in dichloromethane
………………………….
US Patent 8,338,621
J. Surtees and co-inventors disclose alternative processes for making active pharmaceutical ingredients (APIs) that are used to treat epilepsy and seizures. One compound that can be prepared by their processes is the established drug levetiracetam (1, Figure 1), marketed under the trade name Keppra. Because 1 is now off-patent, there is obvious interest in new drugs.

The inventors also claim that seletracetam (2) and brivaracetam (3) (Figure 2) can be prepared by their processes. These drugs are apparently much more active than 1.
All of the drugs are used as single isomers, so a stereoselective synthesis is desirable. The inventors describe two routes for preparing the molecules; the first, shown in Figure 1, is the synthesis of 1 by the reaction between pyrrolidone (4) and chiral bromo amide 5 in the presence of a base. GC analysis showed that the conversion is 40.3% and that the product contains 51% of the (S)-enantiomer and 49% of the (R)-isomer. No details of their separation are given, although the use of chiral HPLC is discussed.
The same reaction is used to prepare derivative 6 of 1. Compound 7 is prepared from the corresponding hydroxy ester and then condensed with 4 to give 6. Chiral HPLC showed that the product is a mixture of 89.3% (S)-enantiomer 6and 10.7% of its (R)-isomer.
The inventors do not describe the detailed preparation of 2, but they report that acid 8 is prepared in 41% yield from pyrrolidone 9 and acid 10 in the presence of NaH (Figure 2). Ammonolysis of 8 produces 2; no reaction details are provided.

In a reaction similar to the preparation of 8, acid 11 is prepared from 10 and pyrrolidone 12. The product is isolated in 77% yield and can be converted to 3by ammonolysis. Again, no details are provided for this reaction.
The second route for preparing the substituted pyrrolidones does not start with simple pyrrolidones and is the subject of additional claims. The route involves a cyclization reaction, shown in Figure 3. The preparation of enantiomer 13 begins with the reaction of racemic salt 14 and optically pure bromo ester 15. This step produces intermediate 16, isolated as a yellow oil. The crude material is treated with 2-hydroxypyridine (2-HP) to cyclize it to 17. This ester is hydrolyzed to give acid 18. Conversion to 13 is carried out by adding ClCO2Et, followed by reaction with liquid NH3 in the presence of K2CO3. The overall yield of 13 is 32%.

This route is also used to prepare levetiracetam (1) by treating 5 with the HCl salt of amino ester 19 to give 20, recovered as its HCl salt in 49% yield. The salt is basified with Et3N and treated with 2-HP to cyclize it to 1, initially isolated as an oil. GC analysis showed 100% conversion, and chiral HPLC showed that the product contains 98.6% (S)-isomer and 1.4% (R)-isomer.
The inventors also prepared 1 and its (R)-enantiomer 21 by using a similar reaction scheme with alternative substrates to 5. Figure 4 outlines the route, which starts from protected hydroxy amide 22 and amino ester 23. When the reaction is carried out in the presence of Cs2CO3, the product is (R)-enantiomer24, which is used without purification to prepare 21 by treating it with 2-HP. Chiral HPLC showed that the product is 94% (R) and 6% (S).

When the reaction between 22 and 23 is run with K2CO3, the product is (S)-enantiomer 25. This is used to prepare 1, but the product contains only 79% (S)-isomer.
The inventors do not comment on the apparent stereoselectivity of the carbonate salts in the reaction of 22 with 23. This is an intriguing finding and worthy of investigation. (UCB S.A. [Brussels]. US Patent 8,338,621,
…………………
Production of Levetiracetam
(1)H-MET-NH2 can be used to manufacture Levetiracetam. The detail is as follows:

(2)A reaction flask was added 500ml of methanol and deionized water 33ml, cooled to 0 ° C. Then add with stirring 50.0g (0.27mol), pass ammonia and dissolve to saturation, and seale reaction flask 0 to 5 º C reaction was stirred 96h TLC tracking,eluent, ethyl acetate / acetone (3:1) product Rf = 0.28, raw material Rf = 0.6]. feedstock point disappears, and the end of the reaction. Finanly, it was distilled under reduced pressure to obtain a yellow solid levetiracetam crude product 41.5g and the yield is 90.2%.

SYNTHESIS
SYN 1
UCB PHARMA, S.A. Patent: WO2007/65634 A1, 2007 ; Location in patent: Page/Page column 16-17 ;
941289-97-0![]()
LEVETIRACETAM
SYN 2
TEVA PHARMACEUTICAL INDUSTRIES LTD.; TEVA PHARMACEUTICALS USA, INC. Patent: WO2004/69796 A2, 2004 ; Location in patent: Page 9 ;
 AND
AND  GIVES PDT
GIVES PDT
SYN 3
 GIVES PDT
GIVES PDT
ZACH SYSTEM S.P.A. Patent: US2011/65932 A1, 2011 ; Location in patent: Page/Page column 3 ;
SYN 4

ZaCh System S.p.A. Patent: EP2147911 A1, 2010 ; Location in patent: Page/Page column 5 ;
SYN 5
 AND
AND  GIVES PDT
GIVES PDT
U C B Societe Anonyme Patent: US4696943 A1, 1987 ;
SYN 6
 AND
AND
UCB, S.A. Patent: WO2005/28435 A1, 2005 ; Location in patent: Page/Page column 10 ;
SYN 7

Tetrahedron Letters, , vol. 47, # 38 p. 6813 – 6815
SYN 8

WO2004/69796 A2, ; Page 11 ;
SEE ALSO
Tetrahedron Asymmetry, , vol. 16, # 22 p. 3739 – 3745
WO2008/77035 A2, ; Page/Page column 15 ;
EP1806339 A1, ; Page/Page column 22 ;
……………

Green Chemistry Letters and Reviews Vol. 3, No. 3, September 2010, 225230
http://www.tandfonline.com/doi/pdf/10.1080/17518251003716568
The desired compound 1 was re-crystallized in hot ethyl acetate (2106 ml) at 60degC, subsequently cooled to 25-30degC, filtered and dried at 35-40degC to obtain product in 65% yield (13 g) and 99.9% purity (by chiral HPLC) as a white solid: mp 116degC (lit3c 117degC); Rf: 0.34 [3:7 (EtOAc: Hexane)]; IR (KBr) nmax 3362, 3200, 2991, 2911, 1676, 1491, 1457, and 1383 cm1 ; 1 H NMR (400 MHz, CDCl3) d 0.91 (t, 3H, J7.5 Hz), 1.601.75 (m, 1H), 1.902.09 (m, 3H), 2.382.47 (m, 2H), 3.343.55 (m, 2H), 4.44 (dd, 1H, J6.7, 8.6 Hz) 5.74 (br, 1H s), 6.45 (br, 1H, s).
3 ) Kotkar, S.P.; Sudalai, A. Tetrahed. Lett. 2006, 47, 68136815.
……..

http://www.google.com/patents/WO2008012268A1?cl=en
Levetiracetam, (-)-(S)-alpha-ethyl-2-oxo- 1 -pyrrolidineacetamide, is a drug useful as a protective agent for treating and preventing hypoxic and ischemic type aggressions of the central nervous system. It is the active ingredient of KEPPRA®, tablets and flavored liquid, indicated as adjunctive therapy in the treatment of partial onset seizures in adults and children four years of age and older with epilepsy. Levetiracetam was first described in US 4,837,223 (UCB Societe Anonyme) where it is stated that it has particular therapeutic properties compared to the known racemic form (non proprietary name etiracetam). The S-enantiomer, for example, has a ten times higher protective activity against hypoxia and a four times higher protective activity against cerebral ischemia than the racemic mixture US ‘223 describes a method for the preparation of levetiracetam which comprises reacting (-)-(S)-alpha-ethyl-2-oxo-l-pyrrolidineacetic acid successively with alkylhaloformate and with ammonia. Said acid intermediate is, in turn, obtained from racemic (±)-alpha-ethyl-2-oxo-l -pyrrolidine acetic acid by a classic optical resolution according to known methods. In example 1 of the above US patent, ethyl (±)-alpha-ethyl-2-oxo-l -pyrrolidine acetate is hydrolyzed to give the corresponding racemic acid in the presence of sodium hydroxide; said acid is subjected to chemical resolution by reaction with an optically active base, (+)-(R)-(l -phenyl ethyl)-amine, selective crystallization of diastereoisomeric salts thereof and isolation of the desired enantiomeric form; finally, the resultant (-)-(S)-alpha-ethyl-2-oxo-l-pyrrolidineacetic acid is converted into the corresponding amide via activation of the carboxyl residue with ethyl chloroformate, in accordance with the following reaction scheme:
Several alternative processes for the preparation of levetiracetam have been disclosed in the art. WO 03/014080 (UCB S.A.) describes an improved process for the preparation of levetiracetam and analogues thereof comprising the ammonolysis reaction of the corresponding ester derivatives in the presence of water.
US 6,107,492 (Daicel Chem; UCB) and US 6,124,473 (UCB) describe the preparation of levetiracetam by optical resolution of etiracetam by means of preparative high performance liquid chromatography or continuous simulated moving bed chromatographic system.
GB 2,225,322 (UCB) describes a process for the preparation of levetiracetam by hydrogenolysis of (S)-alpha-[2-(methylthio)-ethyl]-(2-oxo-l-pyrrolidine)-acetamide in the presence of a desulfurizing agent such as NaBH4/NiC12 6 H2O, nickel Raney W-2 or nickel Raney T- 1.
WO 01/64637 (UCB Farchim) describes the preparation of levetiracetam by asymmetric hydrogenation of (Z) or (E)-2-(2-oxotetrahydro-lH-l-pyrrolyl)-2- butenamide by using a chiral catalyst. EP 162,036 (UCB) describes the preparation of levetiracetam by reacting (S)-2- aminobutanamide with an alkyl 4-halobutyrate or with a 4-halobutyryl halide, and subsequent cyclization of alkyl (S)-4-[[l-(aminocarbonyl)-propyl]-amino-butyrate or of (S)-N-[l-(aminocarbonyl)-propyl]-4-halobutanamide thus obtained. WO 2004/069796 (Teva Pharmaceutical Industries) describes a process for preparing levetiracetam which comprises reacting (S)-2-aminobutyrramide hydrochloride and 4-chlorobutyl chloride in a solvent selected from acetonitrile and methyl tertbutyl ether in the presence of a strong base and recovering the crude product. US 2005/0182262 (Dr. Reddy’s Laboratories) describes the preparation of (S)-2- aminobutyrramide hydrochloride, intermediate useful for the manufacture of levetiracetam via reaction with 4-chlorobutyl chloride.
WO 2004/076416 (Farma Lepori S.A.) describes a process to levetiracetam by means of deaminomethylation of a sufficiently pure enantiomer S-intermediate of formula
or a salt thereof.
In accordance with US ‘223, (-)-(S)-alpha-ethyl-2-oxo-l-pyrrolidineacetamide can not be obtained directly from the racemic mixture by separating the desired enantiomer.
Thus, as underlined above, in US ‘223 the resolution step is carried out on the intermediate (±)-alpha-ethyl-2-oxo-l-pyrrolidineacetic acid.
Said procedure has an intrinsic drawback due to separation of the S-enantiomer from the corresponding racemic mixture by classic optical resolution which, necessarily, leads to a loss of 50% of the acid substrate used.
Example 6 (-)-(S)-alpha-ethyl-2-oxo-l-pyrrolidineacetamide (levetiracetam).
In a 25 ml flask equipped with thermometer, mechanical stirring and bubble condenser, 3.344 g of (-)-(S)-alpha-ethyl-2-oxo-l-pyrrolidineacetic acid (19.58 mmol, e.e.= 95.0%), 0.11 ml of concentrated sulfuric acid (95.6% m/m, 1.97 mmol) and 17 ml of methanol were charged under nitrogen atmosphere at room temperature. Reaction mixture was heated up to 65°C temperature by oil bath and maintained at reflux temperature up to complete disappearing of starting material (about 2.5 h; checked by TLC, Rf = 0.58 CH2Cl2:Me0H:Ac0H 80:20: 1/silica gel). Reaction mixture was concentrated under vacuum up to a residue was formed then water (2.0 ml) was added. In a 25 ml flask equipped with magnetic stirring and condenser, 7.5 ml of 30% aqueous ammonia solution was charged and cooled to 00C temperature and, keeping under stirring, the aqueous solution of crude (-)-(S)-alpha-ethyl-2-oxo- 1-pyrrolidineacetic acid methyl ester was charged dropwise. When addition was completed, reaction mixture was thermostabilized at 200C and said conditions were maintained overnight.
At complete conversion (about 10 h) excess of ammonia was eliminated by vacuum evaporator. Reaction mixture was extracted with dichloromethane (2 x 3.5 ml), transferred into a continuous liquid-liquid extractor and then refluxed with 7 ml of dichloromethane for 6 hours. Collected organic phases were concentrated under vacuum up to a residue was formed. 2.666 g of a yellow solid was obtained which was suspended in 15.0 ml of acetone. Reaction mixture was heated up to 600C temperature so that complete dissolution of the solid was reached. Then, mixture was slowly cooled. White solid was isolated by filtration, washed with mother liquors and then with 3 ml of cold acetone and, finally, dried in oven under vacuum at 400C temperature for 4 hours to give 2.259 g of levetiracetam (13.274 mmol, 67.8% yield, e.e. 99.9%).
Example 7
(-)-(S)-alpha-ethyl-2-oxo-l-pyrrolidineacetic acid methyl ester. In a 250 ml reactor equipped with mechanical stirring, thermometer and condenser, 2.5 g of (-)-(S)-alpha-ethyl-2-oxo-l-pyrrolidineacet-N-(+)-(R)-(l-phenylethyl)-amide (9.112 mmol, d.e.= 99.3%), 24.85 g (6 eq.) of p-toluensulfonic acid supported by polymeric matrix (30.00-60.00 mesh, 2.2 mmol/g) and 75 ml of toluene were charged. To the reaction mixture was added 0.660 ml (36.64 mmol) of water under stirring and mixture was heated up to reflux temperature. Reaction was monitored by HPLC and at complete conversion of starting material (about 6 h), mixture was cooled to 600C temperature and 75 ml of methanol added. Reaction mixture was maintained at that temperature for 3 h up to complete formation of (-)-(S)-alpha- ethyl-2-oxo-l-pyrrolidineacetic acid methyl ester. Reaction mixture was permitted to cool and then it was filtered on gootch in order to separate the product from the resin. Resin was washed with methanol (2 x 75 ml) and organic phases were collected to give 365.1 g of a 0.462% organic solution of (-)-(S)-alpha-ethyl-2-oxo-l- pyrrolidineacetic acid methyl ester (1.69 g, 9.110 mmol, 100.0% yield) which was used in the following synthetic step. In order to recover (+)-(R)-(l-phenylethyl)-amine, resin was treated with 100 ml of 30% aqueous ammonia solution, 100 ml of methanol, 100 ml of 30% aqueous soda and again with 100 ml of methanol. Resin was then regenerated by washing with HCl 6 M (100 ml) and water up to neuter pH of the eluted phase. Finally, resin was washed with 100 ml of methanol and dried in oven at 500C temperature under vacuum overnight.
Example 8
(-)-(S)-alpha-ethyl-2-oxo-l-pyrrolidineacetamide (levetiracetam) (alternative 1). 365.1 g of the solution of (-)-(S)-alpha-ethyl-2-oxo-l-pyrrolidineacetic acid methyl ester (0.462%, 1.69 g, 9.110 mmol) obtained in Example 7 was charged in a flask and concentrated up to a residue was formed. 2.482 g of a brown oil was obtained. Residue was charged in a 10 ml flask equipped with magnetic stirring and condenser. Reaction mixture was cooled to 00C temperature and, keeping under stirring, 0.8 ml of water and 3.2 ml of 30% aqueous ammonia solution were charged dropwise in about 10 minutes. When addition was completed, reaction mixture was thermostabilized at 200C and said conditions were maintained overnight.
At complete conversion (about 14 h) excess of ammonia was eliminated by vacuum evaporator. Reaction mixture was then extracted with dichloromethane (10 x 5 ml). Collected organic phases were dried on Na2SO4, and concentrated under vacuum up to a residue was formed. 1.999 g of a yellow solid was obtained which was suspended in 5 ml of acetone. Reaction mixture was heated up to 600C temperature so that complete dissolution of the solid was reached. Then, mixture was slowly cooled. White solid was isolated by filtration, washed with mother liquors and then with 1 ml of cold acetone and, finally, dried in oven under vacuum at 25°C temperature for 1 night to give 0.965 g of levetiracetam (5.669 mmol, 62.2% yield, e.e. 94.2%). Example 9
(-)-(S)-alpha-ethyl-2-oxo-l-pyrrolidineacetamide (levetiracetam) (alternative 2). In a 50 ml reactor equipped with mechanical stirring, thermometer and condenser, 0.275 g of (-)-(S)-alpha-ethyl-2-oxo-l-pyrrolidineacet-N-(+)-(R)-(l-phenylethyl)- amide (1.0 mmol, d.e.= 99.3%), 10.0 g of ethyl-thiophenyl-sulfonic acid supported on silica (0.6 mmol/g, supplied by Phosphonics ®) and 15 ml of toluene were charged. To the reaction mixture was added 0.075 ml (4.0 mmol) of water under stirring and mixture was heated up to reflux temperature. Reaction is monitored by HPLC and at complete conversion of starting material (about 5 h), reaction mixture was cooled to 600C temperature and 10 ml of methanol added. Reaction mixture was maintained at that temperature for 3 h up to complete formation of (-)-(S)-alpha-ethyl-2-oxo-l- pyrrolidineacetic acid methyl ester. Reaction mixture was permitted to cool and then worked up according to the procedure described in example 7. 57.9 g of a 0.280% organic solution of (-)-(S)-alpha-ethyl-2-oxo-l-pyrrolidineacetic acid methyl ester (0.162 g, 0.875 mmol, 87.5% yield) was thus obtained. Such solution was charged in a flask and concentrated up to a residue was formed. 0.486 g of a brown oil was obtained. Residue was charged in a 5 ml flask equipped with magnetic stirring and condenser. Reaction mixture was cooled to 00C temperature and, keeping under stirring, 1.5 ml of 30% aqueous ammonia solution were charged dropwise. When addition was completed, reaction mixture was thermostabilized at 200C and said conditions were maintained overnight.
At complete conversion (about 15 h) excess of ammonia was eliminated by vacuum evaporator. Reaction mixture was then extracted with dichloromethane as described in example 8. Recrystallization of the crude product from refluxing acetone afforded 0.076 g of levetiracetam (0.447 mmol, 44.6% yield compared to the starting amide, e.e. 99.9%).
……………

PAPER FROM HINDAWI
Journal of Chemistry Volume 2013 (2013), Article ID 176512, 5 pages http://dx.doi.org/10.1155/2013/176512
Enantioselective Synthesis of Antiepileptic Agent, (−)-Levetiracetam, through Evans Asymmetric Strategy
1Department of Research and Development, Inogent Laboratories Private Limited, 28A, IDA Nacharam, Hyderabad 500 076, India
2Centre for Pharmaceutical Sciences, Institute of Science and Technology, Jawaharlal Nehru Technological University, Kukatpally, Hyderabad 500 072, India
3R&D Centre, Orchid Chemicals and Pharmaceuticals Ltd., 476/14, Sholinganallur, Chennai 600 119,
http://www.hindawi.com/journals/jchem/2013/176512/
A practical and efficient enantioselective synthesis of antiepileptic drug, (−)-Levetiracetam, has been described in five steps (33.0% overall yield) and high optical purity (99.0% ee), using Evans asymmetric strategy for -alkylation of carbonyl functionality as the key step. The simplicity of the experimental procedures and high stereochemical outcome make this method synthetically attractive for preparing the target compound on multigram scales.

white solid. Mp: 113–114°C.
S ROT= −95.0 [c = l.0, acetone].
1H NMR , 400 MHz):
δ 6.50 (br s, 1H),
5.70 (br s, 1H),
4.50 (t, J = 8.7, 6.8 Hz, 1H),
3.48 (m, 2H), 2.50 (m, 2H),
1.98–2.20 (m, 3H),
1.70 (m, 1H),
0.98 (t, J = 7.7 Hz, 3H) ppm; CH2–CH3
13C NMR , 75 MHz): δ175.9, 172.7, 55.9, 43.7, 31.0, 21.2, 18.0, 10.4 ppm;
IR : 3200, 1731, 1620 cm−1;
ESI-MS: m/z 171.0 [M++1].
Anal. calcd. for C8H14N2O2: C, 56.45; H, 8.29; N, 16.46; O, 18.80. Found: C, 56.76; H, 8.52; N, 16.87; O, 19.26.
Chiral HPLC purity 99% ee. The enantiomeric excess was determined by HPLC analysis in comparison with authentic racemic material and HPLC conditions: Chiral OD-H column; hexane: i-PrOH (90 : 10 v/v); flow rate 1.0 mL/min; UV −210 nm; column temperature 25°C; CHIRAL HPLC purity: = 14.4 min (S)-isomer (major enantiomer) and 9.3 min (R)-isomer (minor enantiomer).
…………………

Indian Journal of Chemistry -Section B (IJC-B) >
IJC-B Vol.53B [2014] >
IJC-B Vol.53B(09) [September 2014] >
http://nopr.niscair.res.in/handle/123456789/29370

1H nmr predict

 13 C NMR PREDICT
13 C NMR PREDICT
…

| US4837223 | Mar 12, 1987 | Jun 6, 1989 | Ucb Societe Anonyme | (S)-alpha-ethyl-2-oxo-1-pyrrolidineacetamide compositions |
| US6107492 | May 7, 1999 | Aug 22, 2000 | Ucb, S.A. | By optical resolution of a racemic mixture of alpha-ethyl-2-oxo-1-pyrrolidine acetamide by chromatography using silica gel supporting amylose tris(3,5-dimethylphenylcarbamate) as a packing material |
| US6124473 | May 7, 1999 | Sep 26, 2000 | Ucb, S.A. | Process for preparing (s)- and (R)-α-ethyl-2-oxo-1-pyrrolidineacetamide |
| US7531673 * | Feb 16, 2005 | May 12, 2009 | Dr. Reddy’s Laboratories Limited | Preparation of amino acid amides |
| US20050182262 * | Feb 16, 2005 | Aug 18, 2005 | Acharyulu Palle V.R. | Reacting an amino acid or acid salt with a halogenating agent (thionyl chloride, phosphorous pentachloride or oxalyl chloride) , to form an intermediate, reacting the intermediate with ammonia; amidation; chemical intermediate to form Levetiracetam |
| EP1566376A1 | Feb 17, 2005 | Aug 24, 2005 | Dr. Reddy’s Laboratories Limited | Preparation of amino acid amides |
| GB1309692A | Title not available | |||
| GB2225322A | Title not available | |||
| WO2001064637A1 | Feb 21, 2001 | Sep 7, 2001 | Edmond Differding | 2-oxo-1-pyrrolidine derivatives, process for preparing them and their uses |
| WO2003014080A2 | Aug 5, 2002 | Feb 20, 2003 | Celal Ates | Oxopyrrolidine compounds, preparation of said compounds and their use in the manufacturing of levetiracetam and analogues |
| WO2004069796A2 | Feb 3, 2004 | Aug 19, 2004 | Ben-Zion Dolitzky | Process for producing levetiracetam |
| WO2006095362A1 * | Jan 20, 2006 | Sep 14, 2006 | Rubamin Ltd | Process for preparing levetiracetam |
| WO2008012268A1 | Jul 20, 2007 | Jan 31, 2008 | Zach System Spa | Process for the preparation of levetiracetam |
……

http://orgspectroscopyint.blogspot.in/2015/03/rs-alpha-ethyl-2-oxo-l-pyrrolidineacet.html
PREPARATION OF KEY INETERMEDIATE
(±)-(R,S)-alpha-ethyl-2- oxo-l-pyrrolidineacet-N-(+)-(R)-(l-phenylethyl)-amide
methyl (±)-(R,S)-alpha-ethyl-2-oxo-l -pyrrolidine acetate with (+)-(R)-(l-phenylethyl)- amine in toluene in the presence of a base such as sodium hydride or methoxide; crystallization- induced dynamic resolution of the resultant (±)-(R,S)-alpha-ethyl-2- oxo-l-pyrrolidineacet-N-(+)-(R)-(l-phenylethyl)-amide
(R)-(+)-1-Phenylethylamine
33978-83-5
1-Pyrrolidineacetic acid, α-ethyl-2-oxo-, methyl ester

1004767-60-5
1-Pyrrolidineacetamide, α-ethyl-2-oxo-N-[(1R)-1-phenylethyl]-
(±)-(R.S)-alpha-ethyl-2-oxo-l-pyrrolidineacet-N-(+)-(R)-(l-phenylethyl)-amide
Example 1
(±)-(R,S)-alpha-ethyl-2-oxo-l-pyrrolidineacet-N-(+)-(R)-(l-phenylethyl)-amide.
In a 100 ml reactor equipped with mechanical stirring, thermometer and bubble condenser, 13.4 g of (±)-(R,S)-alpha-ethyl-2-oxo-l-pyrrolidineacetic acid methyl ester (71.6 mmol), 8.8 g of (+)-(R)-(l-phenylethyl)-amine (72.5 mmol) and 45 ml of tetrahydrofuran were charged. 3.4 g of NaH (60% dispersion in mineral oil, 85.6 mmol) was added in small portions under nitrogen atmosphere. Reaction mixture was maintained at room temperature for about 2 h. Then, it was heated up to 350C and kept under stirring overnight. Reaction was controlled by TLC (Rf = 0.5, AcOEt/silica gel).
At reaction completed, one night at 35°C temperature, reaction mixture was cooled to room temperature and 30 ml of water was slowly charged. It was transferred into a separatory funnel and was diluted with 30 ml of water and 80 ml of dichloromethane. Phases were separated and the aqueous one was washed with 50 ml of dichloromethane. Collected organic phases were washed with an aqueous acid solution, dried on Na2SO4, filtered and concentrated under vacuum. 19.5 g of an oil residue was obtained which slowly solidified. Solid was suspended in 20 ml of a hexane/dichloromethane 9/1 v/v mixture. It was then filtered, washed with 10 ml of the same solvent mixture and dried at 400C to give 12.1 g of the title compound (44.1 mmol, 61.6% yield) as dry solid.
1H NMR (400.13 MHz, CDCl3, 25 0C): δ (ppm, TMS)
7.35-7.19 (1OH, m),
6.49 (2H, br s),
5.09-5.00 (2H, m),
4.41 (IH, dd, J = 8.3, 7.4 Hz),
4.36 (IH, dd, J = 8.6, 7.1 Hz),
3.49 (IH, ddd, J = 9.8, 7.7, 6.6 Hz),
3.41 (IH, ddd, J = 9.8, 7.7, 6.2 Hz),
3.30 (IH, ddd, J = 9.6, 8.3, 5.5 Hz),
3.13 (IH, ddd, 9.7, 8.5, 6.1 Hz), 2.47-2.38 (2H, m), 2.41 (IH, ddd, J = 17.0, 9.6, 6.3 Hz), 2.26 (IH, ddd, 17.0, 9.5, 6.6 Hz), 2.10-1.98 (2H, m), 2.01-1.89 (IH, m), 1.99-1.88 (IH, m), 1.98-1.85 (IH, m), 1.88-1.78 (IH, m), 1.75- 1.62 (IH, m), 1.72-1.59 (IH, m), 1.45 (3H, d, J = 7.1 Hz), 1.44 (3H, d, J = 7.1 Hz), 0.90 (3H, t, J = 7.4 Hz), 0.86 (3H, t, J = 7.4 Hz).
13C NMR (100.62 MHz, CDCl3, 25 0C): δ (ppm, TMS)
176.05 (CO), 176.00 (CO), 169.08 (CO),
168.81 (CO), 143.59 (Cquat),
143.02 (Cquat), 128.66 (2 x CH), 128.55 (2 x CH),
127.33 (CH), 127.19 (CH), 126.05 (2 x CH),
125.80 (2 x CH), 56.98 (CH), 56.61 (CH),
48.90 (CH), 48.84 (CH), 44.08 (CH2),
43.71 (CH2), 31.19 (CH2), 31.07 (CH2), 22.08 (CH3),
22.04 (CH3), 21.21 (CH2), 20.68 (CH2),
18.28 (CH2), 18.08 (CH2), 10.50 (CH3), 10.45 (CH3).
Example 2 (±)-(R.S)-alpha-ethyl-2-oxo-l-pyrrolidineacet-N-(+)-(R)-(l-phenylethyl)-amide (alternative 1).
In a 500 ml reactor equipped with mechanical stirring, thermometer and condenser, 24.2 g of (+)-(R)-(l-phenylethyl)-amine (199.51 mmol) and 40 ml of toluene were charged. By keeping the reaction mixture at 00C temperature under nitrogen atmosphere, 9.5 g of NaH (60% mineral oil suspension, 237.50 mmol) was added in small portions. At the same temperature, 190.0 g of a toluene solution of (±)-(R,S)- alpha-ethyl-2-oxo-l-pyrrolidineacetic acid methyl ester (19.28% equal to 36.63 g, 197.77 mmol) was charged. Reaction mixture was then heated up to 35°C and maintained in that condition till complete disappearing of methyl ester reagent (about 14 h; checked by HPLC).
At reaction completed, reaction mixture was cooled and when room temperature was reached, 100 ml of water was slowly charged. Aqueous phases were separated and extracted with toluene (2 x 75 ml). Collected organic phases were treated with acid water till neuter pH. Solvent was evaporated and residue was suspended in about 100 ml of heptane for about 30 minutes. Product was isolated by filtration and dried in oven at 400C temperature under vacuum overnight to give 45.2 g of the title compound (164.54 mmol, 83.2% yield, d.e. 0.0%) as white dusty solid.
Example 3
(±)-(R,S)-alpha-ethyl-2-oxo-l-pyrrolidineacet-N-(+)-(R)-(l-phenylethyl)-amide (alternative 2).
In a 500 ml reactor equipped with mechanical stirring, thermometer and Dean-Stark distiller, 24.2 g of (+)-(R)-(l-phenylethyl)-amine (199.51 mmol) and 40 ml of toluene were charged. By keeping the reaction mixture at 00C temperature, 42.7 g of sodium methoxide (30% solution in methanol, 237.14 mmol) was added under nitrogen atmosphere. At the same temperature, 190.0 g of a toluene solution of (±)- (R,S)-alpha-ethyl-2-oxo-l-pyrrolidineacetic acid methyl ester (19.28% equal to 36.63 g, 197.77 mmol) was charged. Reaction mixture was then heated up to 65- 700C and maintained in that condition till complete disappearing of methyl ester reagent (about 4 h; checked by HPLC). After a work-up carried out according to the procedure described in example 2, 40.2 g of the title compound (146.53 mmol, 74.1% yield, d.e. 0.0%) as white dusty solid was obtained.
…….