Home » Posts tagged 'organic synthesis' (Page 4)
Tag Archives: organic synthesis
FDA Grants Priority Review To New Drug Application For MNK-795 Submitted By Depomed Licensee Mallinckrodt

FDA Grants Priority Review To New Drug Application For MNK-795 Submitted By Depomed Licensee Mallinckrodt
Controlled Substance Analgesic Combination Product Uses Depomed’s Proprietary Acuform® Technology
NEWARK, Calif., July 29, 2013 /PRNewswire/ — Depomed, Inc. (NASDAQ:DEPO) announced today that the U. S. Food and Drug Administration (FDA) has accepted for filing a New Drug Application (NDA) from Mallinckrodt (NYSE: MNK) for MNK-795. MNK-795 is a controlled-release oral formulation of oxycodone and acetaminophen that has been studied for the management of moderate to severe acute pain where the use of an opioid analgesic is appropriate. MNK-795 is formulated with Depomed’s Acuform® drug delivery technology.
http://www.pharmalive.com/fda-grants-priority-review-to-new-drug-application-for-mnk-795
British drugmaker AstraZeneca and US biotechnology company FibroGen have formed a strategic partnership to develop and commercialize FG-4592 to treat anemia in patients with chronic kidney disease (CKD) and end-stage renal disease (ESRD).

cas no 808118-40-3 FG 4592
FG-4592 oral presentation at ASN 2012
FG-4592 is an orally administered small molecule inhibitor of hypoxia-inducible factor (HIF) prolyl hydroxylase activity.
As part of the agreement, the deal will focus on US, China and all major markets except the Commonwealth of Independent States, the Middle East, South Africa, Japan and Europe, while both companies can exercise the option to extend their collaboration to other anemia indications.

AstraZeneca will pay FibroGen at least $350 million as part of a strategic collaboration to develop and commercialize FG-4592, a first-in-class oral compound in late stage development for the treatment of anaemia associated with chronic kidney disease (CKD) and end-stage renal disease (ESRD).
This broad collaboration focuses on the US, China and all major markets excluding Japan, Europe, the Commonwealth of Independent States, the Middle East and South Africa, which are covered by an existing agreement between FibroGen and Astellas Pharma. The AstraZeneca-FibroGen joint effort will be focused on the development of FG-4592 to treat anaemia in CKD and ESRD, and may be extended to other anaemia indications. The companies plan to undertake an extensive FG-4592 Phase III development program for the US, and to initiate Phase III trials in China, with anticipated regulatory filings in China in 2015 and in the US in 2017.
In addition to $350 million in up front and non-contingent payments, AstraZeneca could pay FibroGen potential future development related milestone payments of up to $465 million, and potential future sales related milestone payments in addition to tiered royalty payments on future sales on FG-4592 in the low 20% range. Additional development milestones will be payable for any subsequent indications which the companies choose to pursue. AstraZeneca will be responsible for the US commercialization of FG-4592, with FibroGen undertaking specified promotional activities in the ESRD segment in this market. The companies will also co-commercialize FG-4592 in China where FibroGen will be responsible for clinical trials, regulatory matters, manufacturing and medical affairs, and AstraZeneca will oversee promotional activities and commercial distribution.
FG-4592 is a small molecule inhibitor of hypoxia-inducible factor (HIF), a protein that responds to oxygen changes in the cellular environment and meets the body’s demands for oxygen by inducing erythropoiesis, the process by which red blood cells are produced. The firms claim FG-4592 has the potential to address the considerable unmet medical need for an effective treatment for anaemia that offers the convenience of oral administration and an improved safety profile when compard with current standards of care. At present, treatment options involve a combination of injectable erythropoiesis-stimulating agents (ESAs) and iron supplements.
“Our collaboration with FibroGen on FG-4592 is an important addition to AstraZeneca’s growing late-stage portfolio in cardiovascular and metabolic disease, one of our core therapy areas,” comments Pascal Soriot, AstraZeneca CEO. “We know from our research into complications of renal disease that anaemia continues to be a challenge for patients with chronic kidney disease, due in part to the inconvenience and complexity of existing injectable and intravenous therapies and the safety concerns associated with them. The science behind this compound is compelling. Through our collaboration with FibroGen we aim to offer a first-in-class, convenient treatment option for doctors and patients.”
“FG-4592 has the potential to offer anaemia patients an oral therapy that provides coordinated erythropoiesis, that increases natural erythropoietin within the normal physiological range, and that is effective without intravenous iron supplementation and without an increased risk for hypertension,” adds Thomas B. Neff, FibroGen CEO. “We are especially pleased that AstraZeneca will share our commitment to making China the first-to-launch country for FG-4592 and join our effort to bring important innovation in anaemia therapy to CKD and ESRD patients in the US and other countries. This agreement secures proper development and commercialization resources for FG-4592, and ensures US clinical trial efforts are fully funded.”
,,,,,,,,,,
Novavax announces positive preclinical data for vaccine against influenza

Novavax announces positive preclinical data for vaccine against influenza
Novavax has announced positive preclinical results for its virus-like particle (VLP) vaccine candidate against A (H7N9) influenza.
The study examined the immunogenicity, the ability to provoke an immune response, and efficacy of two doses of its A(H7N9) VLP vaccine candidate against a lethal wild-type challenge mouse model.

There were three control groups, including Novavax’ non-homologous A(H7N3) VLP vaccine candidate, its A(H5N1) VLP vaccine candidate, and a placebo. All vaccine candidates were administered with or without Iscomatrix, a saponin-based adjuvant.
read all at
http://www.pharmaceutical-technology.com/news/newsnovavax-announces-positive-preclinical-data-for-vaccine-against-influenza?WT.mc_id=DN_News



………….
………….
Animal testing failures put drug trial volunteers in danger

© Shutterstock
Poor pre-clinical data and an absence of negative results are pushing candidate molecules into the clinic too soon
The reporting of animal studies is biased, inflating the efficacy of drug candidates and pushing them into the clinic before they are ready. This is the verdict of new research, which finds that more treatments go from pre-clinical to human trials than ought to, wasting valuable resources and potentially putting trial participants in danger.1 It adds to a growing body of evidence pointing to problems in the way animal testing is reported and managed.
READ AT
http://www.rsc.org/chemistryworld/2013/07/failures-animal-testing-put-drug-trial-volunteers-danger
………..
Analysis Of Vical’s Allovectin-7: Best Results Ever In A Melanoma Phase 3 Trial
check this video
…………….


Vical’s (VICL) Allovectin-7 is a pure immune therapy.(1) Which means it does not directly kill cancer cells, but activates the immune system to do so. Vical will soon announce A-7 phase 3 results in Melanoma, but the mechanism of action is not specific to Melanoma, and can be used in any solid tumor cancer.(2) For this reason, I expect that Allovectin-7 will become one of the best selling cancer drugs of all time.
Allovectin-7 is a substance that is being studied as a gene therapy agent in the treatment of cancer, such as malignant melanoma. It is a plasmid/lipid complex containing the DNA sequences encoding HLA-B7 and ß2 microglobulin – two components of major histocompatibility complex (MHC, class I). It increases the ability of the immune system to recognize cancer cells and kill them.
In 1999, FDA granted Allovectin-7 orphan drug designation for the treatment of invasive and metastatic melanoma.
- Allovectin-7 entry in the public domain NCI Dictionary of Cancer Terms
![]() This article incorporates public domain material from the U.S. National Cancer Institute document “Dictionary of Cancer Terms”.
This article incorporates public domain material from the U.S. National Cancer Institute document “Dictionary of Cancer Terms”.
Vical’s Allovectin-7
Allovectin is a first-class DNA-based immunotherapeutic designed to stimulate both innate and adaptive immune responses in local tumors and distal metastases. The goal is to become a first-line treatment for Stage III and IV melanoma, where it is intended to provide improved efficacy, a better safety profile, and simple outpatient administration.
As last reported, the company is approaching completion of a Phase III registration trial versus chemotherapy in patients with metastatic melanoma. The reporting of end results has had numerous delays, but the results are now expected by Q3.
Outside of Allovectin, Vical has ten clinical trials ongoing, three of those independent and the rest in collaboration. Clearly, Vical is not totally dependent on this immunotherapy though it is the most advanced independent program in the company’s pipeline.
Vical has a market cap of $257M, so clearly a homerun therapy could send the stock soaring.
UK regulator approves access to Revolade drug after three-year process
25 July 2013
The National Institute for Health and Care Excellence (NICE) in the UK has recommended immune disorder drug Revolade for use on the NHS after a process of three and a half years.
The GSK once-daily oral treatment is now available to adult patients in England and Wales living with chronic immune (idiopathic) thrombocytopenic purpura (cITP), an immune disorder associated with low-blood platelet counts.
In patients with cITP, the immune system prematurely destroys platelets or impairs their production so that platelets are lost from the circulation faster than they can be replaced from the bone marrow, where they are made.
This results in patients developing mild bruising or serious bleeding, which affects their quality of life and, in some instances, may be fatal.
It is estimated that cITP currently affects 50 in 100,000 people in the UK.
The only other licensed TPO-RA recommended by NICE is romiplostim, which is given in the form of a weekly injection.
The Royal London Hospital’s clinical director for pathology Prof Adrian Newland said: “I was very pleased to see that NICE has recognised the clinical value and cost-effectiveness of eltrombopag in their guidance.
“We now have an important addition to the treatment options for patients with severe or refractory disease.”
Revolade is an oral thrombopoietin receptor agonist (TPO-RA) that stimulates the growth and maturation of cells in the bone marrow (megakaryocytes) that produce platelets, increasing platelet production.
When added to conventional immunosuppressive therapy, Revolade, also known as eltrombopag, increases response rates compared with placebo and in some patients.
GlaxoSmithKline UK general manager Erik Van Snippenberg said: “This has been a lengthy three and a half year long appraisal process. We are pleased that NICE has recommended eltrombopag and that the small number of cITP patients in England and Wales are granted access to an alternative treatment option offering the benefit of oral convenience.
“With eltrombopag, we hope to ultimately make a meaningful difference in the quality of life of cITP patients and contribute to potential savings for the NHS.”

Compound Suffocates Tumors
Scientists have discovered a new molecule that prevents cancer cells from responding and surviving when starved of oxygen and which could be developed into new treatments for the disease, according to new research published in the Journal of the American Chemical Society.
Cancer Research UK scientists at the University of Southampton found that this molecule targets the master switch—HIF-1—that cancer cells use to adapt to low oxygen levels, a common feature in the disease.
read all at
Find out more:
- Cancer Sciences Unit
- Is it your ambition to help cure cancer? If so, visit: www.southampton.ac.uk/medicine/undergraduate/index.page
Hepatitis B

Hepatitis B
Key facts
- Hepatitis B is a viral infection that attacks the liver and can cause both acute and chronic disease.
- The virus is transmitted through contact with the blood or other body fluids of an infected person.
- About 600 000 people die every year due to the consequences of hepatitis B.
- Hepatitis B is an important occupational hazard for health workers.
- Hepatitis B is preventable with the currently available safe and effective vaccine.

Hepatitis B is a potentially life-threatening liver infection caused by the hepatitis B virus. It is a major global health problem. It can cause chronic liver disease and chronic infection and puts people at high risk of death from cirrhosis of the liver and liver cancer.
More than 240 million people have chronic (long-term) liver infections. About 600 000 people die every year due to the acute or chronic consequences of hepatitis B.
A vaccine against hepatitis B has been available since 1982. Hepatitis B vaccine is 95% effective in preventing infection and its chronic consequences, and was the first vaccine against a major human cancer.
Geographical distribution
Hepatitis B virus can cause an acute illness with symptoms that last several weeks, including yellowing of the skin and eyes (jaundice), dark urine, extreme fatigue, nausea, vomiting and abdominal pain. Hepatitis B prevalence is highest in sub-Saharan Africa and East Asia. Most people in these regions become infected with the hepatitis B virus during childhood and between 5–10% of the adult population is chronically infected.
High rates of chronic infections are also found in the Amazon and the southern parts of eastern and central Europe. In the Middle East and the Indian subcontinent, an estimated 2–5% of the general population is chronically infected. Less than 1% of the population in western Europe and North America is chronically infected.

Transmission
In highly endemic areas, HBV is most commonly spread from mother to child at birth, or from person to person in early childhood.
Perinatal or early childhood transmission may also account for more than one third of chronic infections in areas of low endemicity, although in those settings, sexual transmission and the use of contaminated needles, especially among injecting drug users, are the major routes of infection.
The hepatitis B virus can survive outside the body for at least seven days. During this time, the virus can still cause infection if it enters the body of a person who is not protected by the vaccine.
The hepatitis B virus is not spread by contaminated food or water, and cannot be spread casually in the workplace.
The incubation period of the hepatitis B virus is 75 days on average, but can vary from 30 to 180 days. The virus may be detected 30 to 60 days after infection and persists for variable periods of time.

Symptoms
Most people do not experience any symptoms during the acute infection phase. However, some people have acute illness with symptoms that last several weeks, including yellowing of the skin and eyes (jaundice), dark urine, extreme fatigue, nausea, vomiting and abdominal pain.
In some people, the hepatitis B virus can also cause a chronic liver infection that can later develop into cirrhosis of the liver or liver cancer.
More than 90% of healthy adults who are infected with the hepatitis B virus will recover and be completely rid of the virus within six months.
Who is at risk for chronic disease?
The likelihood that infection with the hepatitis B virus becomes chronic depends upon the age at which a person becomes infected. Children less than 6 years of age who become infected with the hepatitis B virus are the most likely to develop chronic infections:
- 80–90% of infants infected during the first year of life develop chronic infections;
- 30–50%% of children infected before the age of 6 years develop chronic infections.
In adults:
- <5% of otherwise healthy adults who are infected will develop chronic infection;
- 15–25% of adults who become chronically infected during childhood die from hepatitis B-related liver cancer or cirrhosis.

Diagnosis
It is not possible, on clinical grounds, to differentiate hepatitis B from hepatitis caused by other viral agents and, hence, laboratory confirmation of the diagnosis is essential. A number of blood tests are available to diagnose and monitor people with hepatitis B. They can be used to distinguish acute and chronic infections.
Laboratory diagnosis of hepatitis B infection centres on the detection of the hepatitis B surface antigen HBsAg. WHO recommends that all blood donations are tested for this marker to avoid transmission to recipients.
- Acute HBV infection is characterized by the presence of HBsAg and immunoglobulin M (IgM) antibody to the core antigen, HBcAg. During the initial phase of infection, patients are also seropositive for HBeAg.
- Chronic infection is characterized by the persistence (>6 months) of HBsAg (with or without concurrent HBeAg). Persistence of HBsAg is the principal marker of risk for developing chronic liver disease and hepatocellullar carcinoma (HCC) later in life.
- The presence of HBeAg indicates that the blood and body fluids of the infected individual are highly contagious
Treatment
There is no specific treatment for acute hepatitis B. Care is aimed at maintaining comfort and adequate nutritional balance, including replacement of fluids that are lost from vomiting and diarrhoea.
Some people with chronic hepatitis B can be treated with drugs, including interferon and antiviral agents. Treatment can slow the progression of cirrhosis, reduce incidence of HCC and improve long term survival. Treatment, however, is not readily accessible in many resource-constrained settings.
Liver cancer is almost always fatal and often develops in people at an age when they are most productive and have family responsibilities. In developing countries, most people with liver cancer die within months of diagnosis. In high-income countries, surgery and chemotherapy can prolong life for up to a few years.
People with cirrhosis are sometimes given liver transplants, with varying success.
Prevention
The hepatitis B vaccine is the mainstay of hepatitis B prevention. WHO recommends that all infants receive the hepatitis B vaccine as soon as possible after birth, preferably within 24 hours.
The birth dose should be followed by 2 or 3 doses to complete the primary series. In most cases, 1 of the following 2 options is considered appropriate:
- a 3-dose schedule of hepatitis B vaccine, with the first dose (monovalent) being given at birth and the second and third (monovalent or combined vaccine) given at the same time as the first and third doses of DTP vaccine; or
- 4 doses, where a monovalent birth dose is followed by 3 monovalent or combined vaccine doses, usually given with other routine infant vaccines.
The complete vaccine series induces protective antibody levels in more than 95% of infants, children and young adults. Protection lasts at least 20 years and is possibly lifelong.
All children and adolescents younger than 18 years old and not previously vaccinated should receive the vaccine if they live in countries where there is low or intermediate endemicity. In those settings it is possible that more people in high risk groups may acquire the infection and they should also be vaccinated. They include:
- people who frequently require blood or blood products, dialysis patients, recipients of solid organ transplantations;
- people interned in prisons;
- injecting drug users;
- household and sexual contacts of people with chronic HBV infection;
- people with multiple sexual partners, as well as health-care workers and others who may be exposed to blood and blood products through their work; and
- travellers who have not completed their hepatitis B vaccination series should be offered the vaccine before leaving for endemic areas.

The vaccine has an excellent record of safety and effectiveness. Since 1982, over one billion doses of hepatitis B vaccine have been used worldwide. In many countries, where 8–15% of children used to become chronically infected with the hepatitis B virus, vaccination has reduced the rate of chronic infection to less than 1% among immunized children.
As of July 2011, 179 Member States vaccinate infants against hepatitis B as part of their vaccination schedules. This is a major increase compared with 31 countries in 1992, the year that the World Health Assembly passed a resolution to recommend global vaccination against hepatitis B. Furthermore, as of July 2011, 93 Member States have introduced the hepatitis B birth dose.
In addition, implementation of blood safety strategies, including quality-assured screening of all donated blood and blood components used for transfusion can prevent transmission of HBV. Safe injection – unnecessary as well as unsafe injections – practices can protect against HBV transmission. Furthermore, safer sex practices, including minimizing the number of partners and using barrier protective measures (condoms), protect against transmission.
WHO response
WHO is working in the following areas to prevent and control viral hepatitis:
- raising awareness, promoting partnerships and mobilizing resources;
- formulating evidence-based policy and data for action;
- preventing of transmission; and
- executing screening, care and treatment.
WHO also organizes World Hepatitis Day on July 28 every year to increase awareness and understanding of viral hepatitis.

LUMACAFTOR an Orphan drug in clinical trial for oral the treatment of cystic fibrosis

Lumacaftor
3-[6-[1-(2,2-Difluoro-1,3-benzodioxol-5-yl)cyclopropylcarboxamido]-3-methylpyridin-2-yl]benzoic acid
3-{6-{[1-(2,2-difluoro-1,3-benzodioxol-5-yl)cyclopropanecarbonyl]amino}-3-methylpyridin-2-yl}benzoic acid
VRT-826809
VX-809
US patents: US8124781, US8461342
Indication:Cystic fibrosis
Developmental status:Phase III (US, UK, EU)
Developer:Vertex
| Vertex Pharmaceuticals |
| Company | Vertex Pharmaceuticals Inc. |
| Description | Small molecule cystic fibrosis transmembrane conductance regulator (CFTR) corrector |
| Molecular Target | Cystic fibrosis transmembrane conductance regulator (CFTR) |
| Mechanism of Action | CFTR stabilizer |
| Latest Stage of Development | Phase III |
| Indication | Cystic fibrosis (CF) |
| cas | 936727-05-8 |
http://www.ama-assn.org/resources/doc/usan/lumacaftor.pdf for all data
see……http://orgspectroscopyint.blogspot.in/2015/03/lumacaftor.html
Lumacaftor (USAN, codenamed VX-809) is an experimental drug for the treatment of cystic fibrosis being developed by Vertex Pharmaceuticals. The drug is designed to be effective in patients that have the F508del mutation in the cystic fibrosis transmembrane conductance regulator (CFTR), the defective protein that causes the disease. F508del, meaning that the amino acid phenylalanine in position 508 is missing, is found in about 60% of cystic fibrosis patients in Europe,[1] and in about 90% of persons with some mutation in the CFTR gene.
A corrector molecule, one of two new classes of ion channel modulators. The corrector modulators enhance the number of channels of the CFTR protein at the cell surface. in combination with ivacaftor in homozygous F508del pts
Results from a Phase II clinical trial indicate that patients with the most common form of genetic mutation causing cystic fibrosis—homozygous F508del—had a mean increase of 7.4% in lung function (FEV1) on a combination of lumacaftor and ivacaftor.[2]
VX-809 is an investigational corrector compound in a phase II clinical trial for oral the treatment of cystic fibrosis. The trial will evaluate single and multiple doses of VX-809 in healthy volunteers. This compound has resulted from a collaboration with the Cystic Fibrosis Foundation Therapeutics, Inc. (CFFT) . In 2010, orphan drug designation was assigned in the E.U. and the U.S. for the treatment of CF.
VX-809 may act to restore the function of the cystic fibrosis transmembrane conductance regulator (CFTR) protein, the defective cell membrane protein responsible for the progression of CF. VX-809 and other corrector compounds were designed to increase the amount of DF508-CFTR on the surface of cells lining the airway, which may result in an increase in chloride transport across the cell surface in patients with the DF508-CFTR mutation.
On January 11, 2013, the combination regimen of Lumacaftor (VX-809) and Kalydeco (Ivacaftor) was awarded by U.S. FDA with Breakthrough Therapy Designation as part of the agency’s efforts to accelerate the development and approval of drugs for serious and life-threatening disease.Breakthrough Therapy Designation for the combination regimen of VX-809 with ivacaftor was based on the Phase II combination data announced in 2012. Vertex Pharmaceuticals will report results from two Phase III trials (NCT01807949 (TRANSPORT) and NCT01807923 (TRAFFIC)) of the combination of Kalydeco + VX-809 in the middle of 2014. Positive data from TRAFFIC and TRANSPORT could open up a market with peak sales of approximately $6 billion, estimate analysts.
- 1 Merk; Schubert-Zsilavecz. Pharmazeutische Zeitung (in German) 156 (37): 24–27.
- 2 Wilschanski, M. (2013). “Novel therapeutic approaches for cystic fibrosis”. Discovery medicine 15 (81): 127–133. PMID 23449115

see……http://orgspectroscopyint.blogspot.in/2015/03/lumacaftor.htm
…………………………
PATENT
http://www.google.com/patents/EP2639222A1?cl=en
-
Compound 1 and pharmaceutically acceptable compositions thereof are useful for treating or lessening the severity of a variety of CFTR mediated diseases.
Scheme 2. Synthesis of the amine moiety.
Scheme 3. Formation of an acid salt of 3-(6-(1-(2,2-difluorobcnzo[d][1,3]dioxol-5-yl) cyclopropanecarboxamido)-3-methylpyridin-2-yl)benzoic acid.
-
Synthesis of 3-(6-(1-(2,2-difluorobenzord[d][1,3]dioxol-5-yl) cyclopropancearboxamido)-3-methylpyridin-2-yl)benzoic acid • HCl.
-
[0238]Acid Chloride Moiety
-
Commercially available 2,2-difluoro-1,3-benzodioxole-5-carboxylic acid (1.0 eq) is slurried in toluene (10 vol). Vitride® (2 eq) is added via addition funnel at a rate to maintain the temperature at 15-25 °C. At the end of addition the temperature is increased to 40 °C for 2 h then 10% (w/w) aq. NaOH (4.0 eq) is carefully added via addition funnel maintaining the temperature at 40-50 °C. After stirring for an additional 30 minutes, the layers are allowed to separate at 40 °C. The organic phase is cooled to 20 °C then washed with water (2 x 1.5 vol), dried (Na2SO4), filtered, and concentrated to afford crude Compound 18 that is used directly in the next step.
-
Compound 18 (1.0 eq) is dissolved in MTBE (5 vol). A catalytic amount of DMAP (1 mol %) is added and SOCl2 (1.2 eq) is added via addition funnel. The SOCl2 is added at a rate to maintain the temperature in the reactor at 15-25 °C. The temperature is increased to 30 °C for 1 hour then cooled to 20 °C then water (4 vol) is added via addition funnel maintaining the temperature at less than 30 °C. After stirring for an additional 30 minutes, the layers are allowed to separate. The organic layer is stirred and 10% (w/v) aq. NaOH (4.4 vol) is added. After stirring for 15 to 20 minutes, the layers are allowed to separate. The organic phase is then dried (Na2SO4), filtered, and concentrated to afford crude Compound 19 that is used directly in the next step.
-
A solution of Compound 19 (1 eq) in DMSO (1.25 vol) is added to a slurry of NaCN (1.4 eq) in DMSO (3 vol) maintaining the temperature between 30-40 °C. The mixture is stirred for 1 hour then water (6 vol) is added followed by MTBE (4 vol). After stirring for 30 min, the layers are separated. The aqueous layer is extracted with MTBE (1.8 vol). The combined organic layers are washed with water (1.8 vol), dried (Na2SO4), filtered, and concentrated to afford crude compound 20 (95%) that is used directly in the next step.
-
A mixture of compound 20 (1.0 eq), 50 wt % aqueous KOH (5.0 eq) 1-bromo-2-chloroethane (1.5 eq), and Oct4NBr (0.02 eq) is heated at 70 °C for 1 h. The reaction mixture is cooled then worked up with MTBE and water. The organic phase is washed with water and brine then the solvent is removed to afford compound 21.
-
Compound 21 is hydrolyzed using 6 M NaOH (8 equiv) in ethanol (5 vol) at 80 °C overnight. The mixture is cooled to room temperature and ethanol is evaporated under vacuum. The residue is taken into water and MTBE, 1 M HCl was added and the layers are separated. The MTBE layer was then treated with dicyclohexylamine (0.97 equiv). The slurry is cooled to 0 °C, filtered and washed with heptane to give the corresponding DCHA salt. The salt is taken into MTBE and 10% citric acid and stirred until all solids dissolve. The layers are separated and the MTBE layer was washed with water and brine. Solvent swap to heptane followed by filtration gives compound 22 after drying in a vacuum oven at 50 °C overnight.
-
Compound 22 (1.2 cq) is slurried in toluene (2.5 vol) and the mixture heated to 60 °C. SOCl2 (1.4 eq) is added via addition funnel. The toluene and SOCl2 are distilled from the reaction mixture after 30 minutes. Additional toluene (2.5 vol) is added and distilled again.
-
A solution of Compound 19 (1 eq) in DMSO (1.25 vol) is added to a slurry of Na14 CN (1.4 eq) in DMSO (3 vol) maintaining the temperature between 30-40 °C. The mixture is stirred for 1 hour then water (6 vol) is added followed by MTBE (4 vol). After stirring for 30 min, the layers are separated. The aqueous layer is extracted with MTBE (1.8 vol). The combined organic layers are washed with water (1.8 vol), dried (Na2SO4), filtered, and concentrated to afford crude compound 23 that is purified by chromatography.
-
A mixture of compound 23 (1.0 eq) and 1,2-dibromoethane (1.8 eq) in THF (3 vol) is cooled to -10 °C via external chiller. 1 M LHMDS in THF (2.5 eq) is added via an addition funnel and at a rate to maintain the temperature in the reactor below 10 °C. One hour after addition is complete, 20% w/v aq. citric acid (13 vol) is added via addition funnel maintaining the temperature in the reactor below 20 C. The external chiller is turned off and after stirring for 30 min the layers are separated. The organic layer is filtered and concentrated to afford crude compound 24 that is purified by chromatography.
-
Compound 24 is hydrolyzed using 6 M NaOH (8 equiv) in ethanol (5 vol) at 80 °C overnight. The mixture is cooled to room temperature and ethanol is evaporated under vacuum. The residue is taken into water and MTBE. 1 M HCl is added to the mixture and the organic layer is filtered and concentrated to afford compound 25.
-
A mixture of Compound 25, 4-dimethylaminopyridine, and thionyl chloride (SOCl2) in CH2Cl2 is stirred to produce compound 26, which may be further reacted with compound 6 without isolation.
-
Amine Moiety
-
2-Bromo-3-methylpyridine (1.0 eq) is dissolved in toluene (12 vol). K2CO3 (4.8 eq) is added followed by water (3.5 vol) and the mixture heated to 65 °C under a stream of N2 for 1 hour. 3-(t-Butoxycarbonyl)phenylboronic acid (1.05 eq) and Pd(dppf)Cl2-CH2Cl2 (0.015 eq) are then added and the mixture is heated to 80 °C. After 2 hours, the heat is turned off, water is added (3.5 vol) and the layers are allowed to separate. The organic phase is then washed with water (3.5 vol) and extracted with 10% aqueous methanesulfonic acid (2 eq MsOH, 7.7 vol). The aqueous phase is made basic with 50% aqueous NaOH (2 eq) and extracted with EtOAc (8 vol). The organic layer is concentrated to afford crude compound 4 (82%) that is used directly in the next step.
-
Compound 4 (1.0 eq) is dissolved in EtOAc (6 vol). Water (0. 3 vol) is added followed by urea-hydrogen peroxide (3 cq). The phthalic anhydride (3 cq) is added portion-wise as a solid to maintain the temperature in the reactor below 45 °C. After completion of phthalic anhydride addition, the mixture is heated to 45 °C. After stirring for an additional 4 hours, the heat is turned off. 10% w/w aqueous Na2SO3 (1.5 eq) is added via addition funnel. After completion of Na2SO3 addition, the mixture is stirred for an additional 30 minutes and the layers separated. The organic layer is stirred and 10% w/w aq. Na2CO3 (2 eq) is added. After stirring for 30 minutes, the layers are allowed to separate. The organic phase is washed 13% w/v aq NaCl. The organic phase is then filtered and concentrated to afford crude compound 5 (95%) that is used directly in the next step.
-
A solution of compound 5 (1 eq) and pyridine (4 eq) in MeCN (8 vol) is heated to 70 °C. A solution of methanesulfonic anhydride (1.5 eq) in MeCN (2 vol) is added over 50 min via addition funnel maintaining the temperature at less than 75 °C. The mixture is stirred for an additional 0.5 hours after complete addition. The mixture is then allowed to cool to ambient. Ethanolamine (10 eq) is added via addition funnel. After stirring for 2 hours, water (6 vol) is added and the mixture is cooled to 10 °C. After stirring for NLT 3 hours, the solid is collected by filtration and washed with water (3 vol), 2:1 MeCN/water (3 vol), and MeCN (2×1.5 vol). The solid is dried to constant weight (<1% difference) in a vacuum oven at 50 °C with a slight N2 bleed to afford compound 6 as a red-yellow solid (53% yield).
-
Compound 7 is dissolved in toluene (2.5 vol based on acid chloride) and added via addition funnel to a mixture of compound 6 (1 eq), dimethylaminopyridine (DMAP, 0.02 eq), and triethylamine (3.0 cq) in toluene (4 vol based on compound 6). After 2 hours, water (4 vol based on compound 6) is added to the reaction mixture. After stirring for 30 minutes, the layers are separated. The organic phase is then filtered and concentrated to afford a thick oil of compound 8 (quantitative crude yield). MeCN (3 vol based on crude product) is added and distilled until crystallization occurs. Water (2 vol based on crude product) is added and the mixture stirred for 2 h. The solid is collected by filtration, washed with 1:1 (by volume) MeCN/water (2 x 1 vol based on crude product), and partially dried on the filter under vacuum. The solid is dried to constant weight (<1% difference) in a vacuum oven at 60 °C with a slight N2 bleed to afford 3-(6-(1-(2,2-difluorobenzo[d] [1,3]dioxol-5-yl) cyclopropanecarboxamido)-3-methylpyridin-2-yl)-t-butylbenzoate as a brown solid.
-
To a slurry of compound 8 (1.0 eq) in MeCN (3.0 vol) is added water (0.83 vol) followed by concentrated aqueous HCl (0.83 vol). The mixture is heated to 45 ± 5 °C. After stirring for 24 to 48 hours the reaction is complete and the mixture is allowed to cool to ambient. Water (1.33 vol) is added and the mixture stirred. The solid is collected by filtration, washed with water (2 x 0.3 vol), and partially dried on the filter under vacuum. The solid is dried to constant weight (<1% difference) in a vacuum oven at 60 °C with a slight N2 bleed to afford compound 9 as an off-white solid.
-
A slurry of 3-(6-(1-(2,2-difluorobenzo[d][1,3]dioxol-5-yl) cyclopropanecarboxamido)-3-methylpyridin-2-yl)benzoic acid • HCl (1 eq) in water (10 vol) is stirred at ambient temperature. A sample is taken after stirring for 24 hours. The sample is filtered and the solid washed with water (2 x). The solid sample is submitted for DSC analysis. When DSC analysis indicates complete conversion to Compound 1, the solid is collected by filtration, washed with water (2 x 1.0 vol), and partially dried on the filter under vacuum. The solid is dried to constant weight (<1% difference) in a vacuum oven at 60 °C with a slight N2 bleed to afford Compound 1 as an off-white solid (98% yield).
-
To a slurry of 3-(6-(1-(2,2-difluorobenzo[d][1,3]dioxol-5-yl) cyclopropanecarboxamido)-3-methylpyridin-2-yl)benzoic acid • HCl (1 eq) in water (10 vol) stirred at ambient temperature is added 50% w/w aq. NaOH (2.5 eq). The mixture is stirred for NLT 15 min or until a homogeneous solution. Concentrated HCl (4 eq) is added to crystallize Compound 1. The mixture is heated to 60 °C or 90 °C if needed to reduce the level of the t-butylbenzoate ester. The mixture is heated until HPLC analysis indicates NMT 0.8% (AUC) t-butylbenzoate ester. The mixture is then cooled to ambient and the solid is collected by filtration, washed with water (3 x 3.4 vol), and partially dried on the filter under vacuum. The solid is dried to constant weight (<1% difference) in a vacuum oven at 60 °C with a slight N2 bleed to afford Compound 1 as an off-white solid (97% yield).
-
A solution of 3-(6-(1-(2,2-difluorobenzo[d][1,3]dioxol-5-yl) cyclopropanecarboxamido)-3-methylpyridin-2-yl)-t-butylbenzoate (1.0 eq) in formic acid (3.0 vol) is heated to 70 ± 10 °C. The reaction is continued until the reaction is complete (NMT 1.0% AUC 3-(6-(1-(2,2-difluorobenzo[d][1,3]dioxol-5-yl) cyclopropanecarboxamido)-3-methylpyridin-2-yl)-t-butylbenzoate) or heating for NMT 8 h. The mixture is allowed to cool to ambient. The solution is added to water (6 vol) heated at 50 °C and the mixture stirred. The mixture is then heated to 70 ± 10 °C until the level of 3-(6-(1-(2,2-difluorobenzo[d][1,3]dioxol-5-yl) cyclopropanecarboxamido)-3-methylpyridin-2-yl)-t-butylbenzoate is NMT 0.8% (AUC). The solid is collected by filtration, washed with water (2 x 3 vol), and partially dried on the filter under vacuum. The solid is dried to constant weight (<1% difference) in a vacuum oven at 60 °C with a slight N2 bleed to afford Compound 1 as an off-white solid.
-
1HNMR spectra of Compound 1 are shown in Figures 9-11 (Figures 9 and 10 depict Compound 1 in Form I in a 50 mg/mL, 0.5 methyl cellulose-polysorbate 80 suspension, and Figure 11 depicts Compound 1 as an HCl salt).
-
Table 3 below recites additional analytical data for Compound 1.
-
Table 3.
Cmpd. No. LC/MS M+1 LC/RTmin NMR 1 453.3 1.93 H NMR (400 MHz, DMSO-d6) 9.14 (s, 1H), 7.99-7.93 (m, 3H), 7.80-7.78 (m, 1H), 7.74-7.72 (m, 1H), 7.60-7.55 (m, 2H), 7.41-7.33 (m, 2H), 2.24 (s, 3H), 1.53-1.51 (m, 2H), 1.19-1.17 (m, 2H)



| WO2002096421A1 * | May 22, 2002 | Dec 5, 2002 | Neurogen Corp | 5-substituted-2-arylpyridines as crf1 modulators |
| WO2004072038A1 * | Feb 10, 2004 | Aug 26, 2004 | Vertex Pharma | Processes for the preparation of n-heteroaryl-n-aryl-amines by reacting an n-aryl carbamic acid ester with a halo-heteroaryl and analogous processes |
| WO2007056341A1 | Nov 8, 2006 | May 18, 2007 | Vertex Pharma | Heterocyclic modulators of atp-binding cassette transporters |
| see……http://orgspectroscopyint.blogspot.in/2015/03/lumacaftor.htm |
References
David Andrew Siesel;Processes for producing cycloalkylcarboxamido-pyridine benzoic acids,US patent number US8124781 B2 ;Also published as CA2707494A1, CN101910134A, EP2231606A2, EP2231606B1, EP2639222A1, EP2639223A1, EP2639224A1, US8592602, US20090176989, US20120190856, WO2009076142A2, WO2009076142A3;Filing date:Dec 4, 2008;Original Assignee:Vertex Pharmaceuticals Incorporated
David Andrew Siesel;Processes for producing cycloalkylcarboxamido-pyridine benzoic acids,US patent number US8461342 B2 ;Also published as US20100036130, US20120203006, US20130274477, WO2010138484A2, WO2010138484A3;Original Assignee:Vertex Pharmaceuticals Incorporated
Van Goor, Fredrick F. et al;Pharmaceutical compositions in the treatment of CFTR-mediated diseases such as cystic fibrosis;PCT Int. Appl., WO2011133956
Van Goor, Fredrick F. et al;Pharmaceutical compositions in the treatment of CFTR-mediated diseases such as cystic fibrosis.PCT Int. Appl., WO2011133951
Van Goor, Fredrick F. et al;Pharmaceutical compositions for treatment of CFTR-mediated diseases;PCT Int. Appl., WO2011133953
Verwijs, Marinus Jacobus et al;Preparation and pharmaceutical compositions of Lumacaftor for the treatment of cystic fibrosis and other diseases associated with CFTR mutations;PCT Int. Appl., WO2011127241
Keshavarz-Shokri, Ali et al;Preparation of Lumacaftor for therapeutical use;PCT Int. Appl., WO2011127290
Siesel, David;A process for the preparation of solid forms of (((difluorobenzodioxolyl)cyclopropanecarboxamido)methylpyridinyl)benzoic acid;U.S. Pat. Appl. Publ., US20100036130
Siesel, David;A process for the preparation of solid forms of (((difluorobenzodioxolyl)cyclopropanecarboxamido)methylpyridinyl)benzoic acid;PCT Int. Appl., WO2010138484
Young, Christopher;Dosage units of 3-(6-(1-(2,2-difluorobenzo[d][1,3]dioxol-5-yl) cyclopropanecarboxamido)-3-methylpyridin-2-yl)benzoic acid;PCT Int. Appl., WO2010037066
Hadida-Ruah, Sara et al;paration of N-pyridinyl carboxamide derivatives as modulators of ATP-binding cassette transporters;U.S. Pat. Appl. Publ., 20080019915
Siesel, David;A process for the preparation of solid forms of (((difluorobenzodioxolyl)cyclopropanecarboxamido)methylpyridinyl)benzoic acid;PCT Int. Appl., WO2009076142
Hadida Ruah, Sara et al;Preparation of N-pyridinyl carboxamide derivatives as modulators of ATP-binding cassette transporters;PCT Int. Appl., WO2007056341
video on cystic fibrosis
second video
Update on 26 mar 2015

LUMACAFTOR
VX 809
| 3-[6-[[1-(2,2-difluoro-1,3-benzodioxol-5-yl)cyclopropanecarbonyl]amino]-3-methylpyridin-2-yl]benzoic Acid | |
| CAS No.: | 936727-05-8 |
|---|---|
| Synonyms: |
|
| Formula: | C24H18F2N2O5 |
| Exact Mass: | 452.11800 |
SMILLES…. Cc1ccc(nc1c2cccc(c2)C(=O)O)NC(=O)C3(CC3)c4ccc5c(c4)OC(O5)(F)F
NMR…………….http://file.selleckchem.com/downloads/nmr/S156503-VX-809-HNMR-Selleck.pdf
-
A solution of 3-(6-(1-(2,2-difluorobenzo[d][1,3]dioxol-5-yl) cyclopropanecarboxamido)-3-methylpyridin-2-yl)-t-butylbenzoate (1.0 eq) in formic acid (3.0 vol) is heated to 70 ± 10 °C. The reaction is continued until the reaction is complete (NMT 1.0% AUC 3-(6-(1-(2,2-difluorobenzo[d][1,3]dioxol-5-yl) cyclopropanecarboxamido)-3-methylpyridin-2-yl)-t-butylbenzoate) or heating for NMT 8 h. The mixture is allowed to cool to ambient. The solution is added to water (6 vol) heated at 50 °C and the mixture stirred. The mixture is then heated to 70 ± 10 °C until the level of 3-(6-(1-(2,2-difluorobenzo[d][1,3]dioxol-5-yl) cyclopropanecarboxamido)-3-methylpyridin-2-yl)-t-butylbenzoate is NMT 0.8% (AUC). The solid is collected by filtration, washed with water (2 x 3 vol), and partially dried on the filter under vacuum. The solid is dried to constant weight (<1% difference) in a vacuum oven at 60 °C with a slight N2 bleed to afford Compound 1 as an off-white solid.
-
1HNMR spectra of Compound 1 are shown in Figures 9-11 (Figures 9 and 10 depict Compound 1 in Form I in a 50 mg/mL, 0.5 methyl cellulose-polysorbate 80 suspension, and Figure 11 depicts Compound 1 as an HCl salt).
-
Table 3 below recites additional analytical data for Compound 1.
-
Table 3.
Cmpd. No. LC/MS M+1 LC/RTmin NMR 1 453.3 1.93 H NMR (400 MHz, DMSO-d6) 9.14 (s, 1H), 7.99-7.93 (m, 3H), 7.80-7.78 (m, 1H), 7.74-7.72 (m, 1H), 7.60-7.55 (m, 2H), 7.41-7.33 (m, 2H), 2.24 (s, 3H), 1.53-1.51 (m, 2H), 1.19-1.17 (m, 2H)




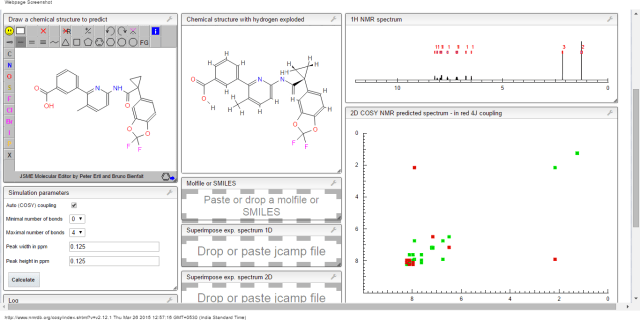
1H NMR PREDICT
![3-[6-[[1-(2,2-difluoro-1,3-benzodioxol-5-yl)cyclopropanecarbonyl]amino]-3-methylpyridin-2-yl]benzoic acid NMR spectra analysis, Chemical CAS NO. 936727-05-8 NMR spectral analysis, 3-[6-[[1-(2,2-difluoro-1,3-benzodioxol-5-yl)cyclopropanecarbonyl]amino]-3-methylpyridin-2-yl]benzoic acid H-NMR spectrum](https://i0.wp.com/pic11.molbase.net/nmr/nmr_image/2014-08-29/001/530/195/936727-05-8-1h.png)
13C NMR PREDICT
![3-[6-[[1-(2,2-difluoro-1,3-benzodioxol-5-yl)cyclopropanecarbonyl]amino]-3-methylpyridin-2-yl]benzoic acid NMR spectra analysis, Chemical CAS NO. 936727-05-8 NMR spectral analysis, 3-[6-[[1-(2,2-difluoro-1,3-benzodioxol-5-yl)cyclopropanecarbonyl]amino]-3-methylpyridin-2-yl]benzoic acid C-NMR spectrum](https://i0.wp.com/pic11.molbase.net/nmr/nmr_image/2014-08-29/001/530/195/936727-05-8-13c.png) CAS NO. 936727-05-8, 3-[6-[[1-(2,2-difluoro-1,3-benzodioxol-5-yl)cyclopropanecarbonyl]amino]-3-methylpyridin-2-yl]benzoic acid C-NMR spectral analysisCOSY PREDICT
CAS NO. 936727-05-8, 3-[6-[[1-(2,2-difluoro-1,3-benzodioxol-5-yl)cyclopropanecarbonyl]amino]-3-methylpyridin-2-yl]benzoic acid C-NMR spectral analysisCOSY PREDICT
13C NMR PREDICT
| WO2002096421A1 * | May 22, 2002 | Dec 5, 2002 | Neurogen Corp | 5-substituted-2-arylpyridines as crf1 modulators |
| WO2004072038A1 * | Feb 10, 2004 | Aug 26, 2004 | Vertex Pharma | Processes for the preparation of n-heteroaryl-n-aryl-amines by reacting an n-aryl carbamic acid ester with a halo-heteroaryl and analogous processes |
| WO2007056341A1 | Nov 8, 2006 | May 18, 2007 | Vertex Pharma | Heterocyclic modulators of atp-binding cassette transporters |
http://www.google.co.in/patents/US8124781



Synthesis of 3-(6-(1-(2,2-difluorobenzo[d][1,3]dioxol-5-yl)cyclopropanecarboxamido)-3-methylpyridin-2-yl)benzoic Acid (Compound 1)
Synthesis of 3-(6-(1-(2,2-difluorobenzo[d][1,3]dioxol-5-yl)cyclopropanecarboxamido)-3-methylpyridin-2-yl)benzoic Acid (Compound 1) Using Water and Base
Synthesis of 3-(6-(1-(2,2-difluorobenzo[d][1,3]dioxol-5-yl)cyclopropanecarboxamido)-3-methylpyridin-2-yl)benzoic Acid (Compound 1) Directly from Benzoate
Compound 1
Compound 1 is used as the starting point for the other solid state forms and can be prepared by coupling an acid chloride moiety with an amine moiety according to Schemes 1-4.
Scheme 1. Synthesis of the acid chloride moiety.
1. NaCN
2. H20
socio
Scheme 1 depicts the preparation of l-(2,2-difluorobenzo[d][l,3]dioxol-5- yl)cyclopropanecarbonyl chloride, which is used in Scheme 3 to make the amide linkage of Compound 1.
The starting material, 2,2-difluorobenzo[d][l,3]dioxole-5-carboxylic acid, is commercially available from Saltigo (an affiliate of the Lanxess Corporation). Reduction of the carboxylc acid moiety in 2,2-difluorobenzo[d][l ,3]dioxole-5-carboxylic acid to the primary alcohol, followed by conversion to the corresponding chloride using thionyl chloride (SOCl2), provides 5-(chloromethyl)-2,2-difluorobenzo[d][l,3]dioxole, which is subsequently converted to 2-(2,2-difluorobenzo[d][l,3]dioxol-5-yl)acetonitrile using sodium cyanide. Treatment of 2-(2,2- difluorobenzo[d][l,3]dioxol-5-yl)acetonitrile with base and l-bromo-2-chloroethane provides 1- (2,2-difluorobenzo[d][l,3]dioxol-5-yl)cyclopropanecarbonitrile. The nitrile moiety in l-(2,2- difluorobenzo[d][l,3]dioxol-5-yl)cyclopropanecarbonitrile is converted to a carboxylic acid using base to give l-(2,2-difluorobenzo[d][l,3]dioxol-5-yl)cyclopropanecarboxylic acid, which is converted to the desired acid chloride using thionyl chloride.
Scheme 2. Alternative synthesis of the acid chloride moiety.
Touene, H20, 70 °C3 N HC1,
DMSO,
75 °C
Scheme 2 depicts an alternative synthesis of the requisite acid chloride. 5- bromomethyl-2,2-difluoro-l,3-benzodioxole is coupled with ethyl cyanoacetate in the presence of a palladium catalyst to form the corresponding alpha cyano ethyl ester. Saponification of the ester moiety to the carboxylic acid gives the cyanoethyl compound. Alkylation of the cyanoethyl compound with l-bromo-2-chloro ethane in the presence of base gives the cyanocyclopropyl compound. Treatment of the cyanocyclopropyl compound with base gives the carboxylate salt, which is converted to the carboxylic acid by treatment with acid. Conversion of the carboxylic acid to the acid chloride is then accomplished using a chlorinating agent such as thionyl chloride or the like.
Scheme 3. Synthesis of the amine moiety.
ptBu urea-hydrogen peroxide hthalic anhydride EtOAc, water
Scheme 3 depicts the preparation of the requisite tert-butyl 3-(6-amino-3- methylpyridin-2-yl)benzoate, which is coupled with l-(2,2-difluorobenzo[d][l,3]dioxol-5- yl)cyclopropanecarbonyl chloride in Scheme 3 to give Compound 1. Palladium-catalyzed coupling of 2-bromo-3-methylpyridine with 3-(tert-butoxycarbonyl)phenylboronic acid gives tert-butyl 3-(3-methylpyridin-2-yl)benzoate, which is subsequently converted to the desired compound. Scheme 4. Formation of an acid salt of 3-(6-(l-(2,2-difluorobenzo[d] [l,3]dioxol-5-yl) cyclopropanecarboxamido)-3-methylpyridin-2-yl)benzoic acid.
Scheme 4 depicts the coupling of l-(2,2-difluorobenzo[d][l,3]dioxol-5- yl)cyclopropanecarbonyl chloride with tert-butyl 3-(6-amino-3-methylpyridin-2-yl)benzoate using triethyl amine and 4-dimethylaminopyridine to initially provide the tert-butyl ester of Compound 1.
……………………..
WO2010037066
http://www.google.im/patents/WO2010037066A2?cl=en
Syntheisis of 3-(6-(l-(2,2-difluorobenzo[d] [l,3]dioxol-5-yl) cyclopropanecarboxamido)-3-methylpyridin-2-yl)benzoic acid • HCL salt.
HCl
To a slurry of 3-(6-(l-(2,2-difluorobenzo[d][l,3]dioxol-5-yl) cyclopropanecarboxamido)-3-methylpyridin-2-yl)-t-butylbenzoate (1.0 eq) in MeCN (3.0 vol) is added water (0.83 vol) followed by concentrated aqueous HCl (0.83 vol). The mixture is heated to 45 ± 5 0C. After stirring for 24 to 48 hours the reaction is complete and the mixture is allowed to cool to ambient. Water (1.33 vol) is added and the mixture stirred. The solid is collected by filtration, washed with water (2 x 0.3 vol), and partially dried on the filter under vacuum. The solid is dried to constant weight (<1% difference) in a vacuum oven at 60 0C with a slight N2 bleed to afford 3-(6-(l-(2,2- difluorobenzo[d][l,3]dioxol-5-yl) cyclopropanecarboxamido)-3-methylpyridin-2- yl)benzoic acid • HCl as an off-white solid.
Synthesis of 3-(6-(l-(2,2-difluorobenzo[d] [l,3]dioxol-5-yl) cyclopropanecarboxamido)-3-methylpyridin-2-yl)benzoic acid (Compound 1 in Form I).
HCl
Compound 1 in Form I
A slurry of 3-(6-(l-(2,2-difluorobenzo[d][l,3]dioxol-5-yl) cyclopropanecarboxamido)-3-methylpyridin-2-yl)benzoic acid • HCl (1 eq) in water (10 vol) is stirred at ambient temperature. A sample is taken after stirring for 24 hours. The sample is filtered and the solid washed with water (2 x). The solid sample is submitted for DSC analysis. When DSC analysis indicates complete conversion to Compound 1, the solid is collected by filtration, washed with water (2 x 1.0 vol), and partially dried on the filter under vacuum. The solid is dried to constant weight (<1% difference) in a vacuum oven at 60 0C with a slight N2 bleed to afford Compound 1 as an off-white solid (98% yield).
Synthesis of 3-(6-(l-(2,2-difluorobenzo[d] [l,3]dioxol-5-yl) cyclopropanecarboxamido)-3-methylpyridin-2-yl)benzoic acid (Compound 1 in Form I) using water and base.
Compound 1 in Form I
To a slurry of 3-(6-(l-(2,2-difluorobenzo[d][l,3]dioxol-5-yl) cyclopropanecarboxamido)-3-methylpyridin-2-yl)benzoic acid • HCl (1 eq) in water (10 vol) stirred at ambient temperature is added 50% w/w aq. NaOH (2.5 eq). The mixture is stirred for NLT 15 min or until a homogeneous solution. Concentrated HCl (4 eq) is added to crystallize Compound 1. The mixture is heated to 60 0C or 90 0C if needed to reduce the level of the t-butylbenzoate ester. The mixture is heated until HPLC analysis indicates NMT 0.8% (AUC) t-butylbenzoate ester. The mixture is then cooled to ambient and the solid is collected by filtration, washed with water (3 x 3.4 vol), and partially dried on the filter under vacuum. The solid is dried to constant weight (<1% difference) in a vacuum oven at 60 0C with a slight N2 bleed to afford Compound 1 as an off-white solid (97% yield).
Synthesis of 3-(6-(l-(2,2-difluorobenzo[d] [l,3]dioxol-5-yl) cyclopropanecarboxamido)-3-methylpyridin-2-yl)benzoic acid (Compound 1 in Form I) directly from benzoate.
Compound 1 in Form I
A solution of 3-(6-(l-(2,2-difluorobenzo[d][l,3]dioxol-5-yl) cyclopropanecarboxamido)-3-methylpyridin-2-yl)-t-butylbenzoate (1.0 eq) in formic acid (3.0 vol) is heated to 70 ± 10 0C. The reaction is continued until the reaction is complete (NMT 1.0% AUC 3-(6-(l-(2,2-difluorobenzo[d][l,3]dioxol-5-yl) cyclopropanecarboxamido)-3-methylpyridin-2-yl)-t-butylbenzoate) or heating for NMT 8 h. The mixture is allowed to cool to ambient. The solution is added to water (6 vol) heated at 50 0C and the mixture stirred. The mixture is then heated to 70 ± 10 0C until the level of 3- (6-(l-(2,2-difluorobenzo[d][l,3]dioxol-5-yl) cyclopropanecarboxamido)-3-methylpyridin- 2-yl)-t-butylbenzoate is NMT 0.8% (AUC). The solid is collected by filtration, washed with water (2 x 3 vol), and partially dried on the filter under vacuum. The solid is dried to constant weight (<1% difference) in a vacuum oven at 60 0C with a slight N2 bleed to afford Compound 1 in Form I as an off-white solid.
Vifor gets FDA approval for Injectafer
Switzerland’s Vifor Pharma is celebrating after getting the thumbs-up from US regulators for Injectafer for the treatment of iron deficiency anaemia.
The US Food and Drug Administration has approved the treatment, sold in Europe as Ferinject (ferric carboxymaltose) since getting the green light in 2007. Specifically, Injectafer will be available for the treatment of IDA in adults who have had an unsatisfactory response or are intolerant to oral iron.
The approval is based on two large trials conducted by Vifor’s US partner Luitpold Pharmaceuticals which studied more than 3,500 patients, of which 1,800 were treated with Injectafer.
read all at
http://www.pharmatimes.com/Article/13-07-26/Vifor_gets_FDA_approval_for_Injectafer.aspx
Ferric carboxymaltose
Chemical structure
The active substance of FERINJECT is a complex of polynuclear iron(III)-hydroxide with 4(R)-(poly-(1→4)-
O-α-D-glucopyranosyl)-oxy-2(R),3(S),5(R),6-tetrahydroxy-hexanoate. The relative molecular weight is
approximately 150,000 Da, corresponding to the empirical formula:
[FeOx(OH)y(H2O)z]n [{(C6H10O5)m (C6H12O7)}l]k, where n ≈ 103, m ≈ 8, l ≈ 11, and k ≈ 4.
CAS-Number
9007-72-1
http://www.tga.gov.au/pdf/auspar/auspar-ferric.pdf for structure

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....