- Young Chemists who have just started work in industry as development chemists
- Organic Chemists/Medicinal Chemists in Research and Development who would like to gain an appreciation of development and scale up and who are perhaps contemplating moving into chemical development.
- Development and Production Chemists in industry who would like to improve their efficiency and gain an insight into alternative approaches to chemical development.
- Chemical Engineers who wish to understand a chemist’s approach to chemical development of batch processes. (Engineers would, however, need a good grounding in organic chemistry)
- Students who are about to enter the industry and can obtain company sponsorship.
- Experienced Chemists looking to refresh and/or augment their knowledge of chemical development
- Analytical Chemists who wish to gain a broader appreciation of process chemistry
- Managers who might benefit from a comprehensive and up to date overview of chemical development
Home » Posts tagged 'hyderabad'
Tag Archives: hyderabad
Dr. D Srinivasa Reddy appointed Director CSIR-IICT Hyderabad India on 7th June 2022. A new assignment
Dr. D Srinivasa Reddy appointed Director CSIR-IICT Hyderabad India on 7th June 2022. A new assignment
This is on recommendation from search cum selection committee which met Prime minister who is president CSIR on 2nd may 2022
currently he is Director CSIR-IIIM jammu
we wish him all the best in New assignment
D. Srinivasa Reddy (DSReddy)


………….Srinivasa Reddy, Director, CSIR-IICT, Hyderabad, india
PTX-COVID19-B
PTX-COVID19-B
| mRNA-based vaccine |
Providence Therapeutics; Canadian government
bioRxiv (2021), 1-50.
https://www.biorxiv.org/content/10.1101/2021.05.11.443286v1
Safe and effective vaccines are needed to end the COVID-19 pandemic caused by SARS-CoV-2. Here we report the preclinical development of a lipid nanoparticle (LNP) formulated SARS-CoV-2 mRNA vaccine, PTX-COVID19-B. PTX-COVID19-B was chosen among three candidates after the initial mouse vaccination results showed that it elicited the strongest neutralizing antibody response against SARS-CoV-2. Further tests in mice and hamsters indicated that PTX-COVID19-B induced robust humoral and cellular immune responses and completely protected the vaccinated animals from SARS-CoV-2 infection in the lung. Studies in hamsters also showed that PTX-COVID19-B protected the upper respiratory tract from SARS-CoV-2 infection. Mouse immune sera elicited by PTX-COVID19-B vaccination were able to neutralize SARS-CoV-2 variants of concern (VOCs), including the B.1.1.7, B.1.351 and P.1 lineages. No adverse effects were induced by PTX-COVID19-B in both mice and hamsters. These preclinical results indicate that PTX-COVID19-B is safe and effective. Based on these results, PTX-COVID19-B was authorized by Health Canada to enter clinical trials in December 2020 with a phase 1 clinical trial ongoing (ClinicalTrials.gov number: NCT04765436).
PTX-COVID19-B is a messenger RNA (mRNA)-based COVID-19 vaccine, a vaccine for the prevention of the COVID-19 disease caused by an infection of the SARS-CoV-2 coronavirus, created by Providence Therapeutics—a private Canadian drug company co-founded by Calgary, Alberta-based businessman Brad T. Sorenson and San Francisco-based Eric Marcusson.[1] in 2013. A team of eighteen working out of Sunnybrook Research Institute in Toronto, Ontario developed PTX-COVID19-B[2] in less than four weeks, according to the Calgary Herald.[3] Human trials with sixty volunteers began on January 26, 2021 in Toronto.[4][5][6]
Providence, which has no manufacturing facilities, partnered with Calgary-based Northern mRNA—the “anchor tenant” in their future manufacturing facilities pending financing.[2]
On 30 April 2021, Sorenson announced that Providence Therapeutics would be leaving Canada and any vaccine that it developed would not be manufactured in Canada.[2]
Overview
Providence Therapeutics Holdings Inc. was co-founded in Toronto, Ontario[7][8] by Calgary, Alberta-based businessman Brad T. Sorenson and San Francisco-based Eric Marcusson Ph.D, who was also the Chief Scientific Officer.[9][3]
PTX-COVID19-B is a messenger RNA (mRNA)-based COVID-19 vaccine. In an interview with CTV news, Sorenson said they were “building some of the important building blocks for the messenger RNA … that provides instructions to cells … to build proteins that may treat or prevent disease”.
As of January 2021, Northern RNA’s Calgary lab was proposed as the site where manufacturing of PTX-COVID19-B would take place.[10] Providence Therapeutics’ partner, Northern RNA, which located at 421 7 Avenue SW in Calgary, has been described as Providence Therapeutics northern division.[7][8]
A February 2021 Manitoba government press release said that the Winnipeg-based Emergent BioSolutions would be manufacturing the vaccine.[11]
Human trials
Phase 1
Human trials began on January 26, 2021 with 60 volunteers between the ages of 18 to 65 in Toronto.[12][13][3] Of these, 15 would receive a placebo and 3 groups of 15 would receive different doses of the vaccine.[10] The volunteers will be monitored for 13 months. The company said that enough data would be available in May which could result in a Phase 2 clinical testing beginning soon after that, pending regulatory approval. If the results of a subsequent larger human trial are positive, the vaccine could enter a commercialization phase in 2022.[14] The Phase 1 clinical trial lead was Piyush Patel. At the 29 April meeting with the House of Commons, Sorenson estimated that PTX-COVID19-B could be approved by Health Canada by “January or February 2022”.[15]:8
Provincial funding
Shortly after the first human trials on PTX-COVID19-B began in late January, on 11 February 2021, Manitoba Premier Brian Pallister announced a “term sheet” between the province and Providence Therapeutics through which Manitoba would receive 2 million doses of PTX-COVID19-B pending its approval by Health Canada.[11] The term sheet includes “best-price guarantee” PTX-COVID19-B.[13] According to a provincial statement released by the Manitoba government, pending approval of the vaccine, the actual manufacturing would take place in Winnipeg by Emergent BioSolutions.[11] Pallister said that, “Building a secure, made-in-Canada vaccine supply will put Canadians at the head of the line to get a COVID vaccine, where we belong.”[11] The down payment would be 20% with a subsequent 40% to be paid when the vaccine was approved by Health Canada; the balance would be paid on delivery of the doses.[13] Specifics about the contract were released in April 2021: the total cost was estimated as CAD $36 million and the agreement included a clause for a non-refundable advance payment of CAD $7.2 million.[2] Sorenson made this comment to Global News: “Under no circumstances is Manitoba going to be on the hook for $7.2 million unless they get real value out of it”.
Federal funding
Canada’s National Research Council (NRC) provided Providence Therapeutics with CAD $5 million for the launch of January 2021 first phase of PTX-COVID19-B clinical trials.[2]
As part of the federal government’s “next generation manufacturing supercluster” program, Providence and Northern mRNA had also been “cleared to access up to $5 million” towards the manufacturing start up process, according to a federal government spokesperson.[2]
The CBC report in late April 2021 also stated that “it could be eligible for a slice of $113 million in additional funding from the National Research Council of Canada Industrial Research Assistance Program”. The federal government had provided funding to some other companies in Canada that were also working to develop a COVID-19 vaccine.[2]
Sorenson as Providence Therapeutics CEO posted an open letter to Prime Minister Justin Trudeau, in which he requested $CDN 150 million upfront to be used to pay for clinical trial and material costs.[16][9]
On 29 April 2021, Sorenson appeared before the House of Commons standing committee on international trade, to ask the Minister of Procurement, Anita Anand, to consider PTX-COVID19-B as an alternative to Moderna and Pfizer for the “2022 booster vaccines”.[15] Sorenson said that the NRC had approached Providence Therapeutics in 2020 after the company had announced their Phase I trial PTX-COVID19-B. Sorenson told the Standing Committee that, “We’ve had really good dialogue ever since phase I started. That process has gone on. That started probably [in February], as we geared up to conclude our phase I trial and release data. Although the NRC is capped at $10 million, which is certainly not sufficient to carry out phase II and phase III trials, the NRC has, through the bureaucracy, elevated us back up to the strategic innovation fund. That occurred about three weeks ago. We’re now working with the strategic innovation fund.”[15]:7
He later said that no reply had been received from the government.[17]
In a meeting with the federal COVID-19 vaccine task force and Sorenson, task force members expressed concerns that “Providence might not be able to scale up production fast enough”.[2]
Clinical trials
PTX-COVID19-B, an mRNA Humoral Vaccine, is Intended for Prevention of COVID-19 in a General Population. This Study is Designed to Evaluate Safety, Tolerability, and Immunogenicity of PTX-COVID19-B Vaccine in Healthy Seronegative Adults Aged 18-64… https://clinicaltrials.gov/ct2/show/NCT04765436
Hyderabad Drugmaker To Make Canada Firm’s mRNA Covid Vaccine In India.. https://www.ndtv.com/india-news/hyderabad-drugmaker-biological-e-to-make-canada-firms-mrna-covid-vaccine-in-india-2454000
Biological E., will run a clinical trial of Providence’s vaccine in India and seek emergency use approval for it, the company said in a statement
Hyderabad-based Biological E said on Tuesday it has entered into a licensing agreement with Providence Therapeutics Holdings to manufacture the Canadian company’s mRNA COVID-19 vaccine in India.
Biological E., which also has a separate deal to produce about 600 million doses of Johnson & Johnson’s COVID-19 shot annually, will run a clinical trial of Providence’s vaccine in India and seek emergency use approval for it, the company said in a statement.
Providence will sell up to 30 million doses of its mRNA vaccine, PTX-COVID19-B, to Biological E, and will also provide the necessary technology transfer of the shot, with a minimum production capacity of 600 million doses in 2022 and a target capacity of 1 billion doses.
Financial details of the transaction were not disclosed.
India has been struggling with a devastating second wave of the pandemic and has managed to fully vaccinate only about 3% of its population. On Monday, the Serum Institute of India said it will increase production of AstraZeneca’s shot by nearly 40% in June, a step towards bridging the shortfall in the country.
“The mRNA platform has emerged as the front runner in delivering the first vaccines for emergency use to combat the COVID-19 pandemic,” said Mahima Datla, Biological E.’s managing director.
Messenger ribonucleic acid (mRNA) vaccines prompt the body to make a protein that is part of the virus, triggering an immune response. US companies Pfizer and Moderna use mRNA technology in their COVID-19 shots.
The drug regulator has approved clinical trials of another mRNA vaccine developed by local firm Gennova Biopharmaceuticals, and the government has said it will fund the studies.
Providence Therapeutics Announces Very Favorable Interim Phase 1 Trial Data for PTX-COVID19-B, its mRNA Vaccine Against COVID-19
CALGARY, AB, May 12, 2021 / – Providence Therapeutics Holdings Inc. (“Providence”) announced today very favorable interim clinical data of PTX-COVID19-B, its vaccine candidate against SARS-CoV-2 (“COVID-19”), from its Phase 1 study entitled “PRO-CL-001, A Phase 1, First-in-Human, Observer-Blinded, Randomized, Placebo Controlled, Ascending Dose Study to Evaluate the Safety, Tolerability, and Immunogenicity of PTX-COVID19-B Vaccine in Healthy Seronegative Adults Aged 18-64” (the “Phase 1 Study”), which found that PTX-COVID19-B met Providence’s target results for safety, tolerability, and immunogenicity in the participants of the Phase 1 Study.
Highlights from Providence Therapeutics’ “Phase 1 Study”:
- PTX-COVID19-B was generally safe and well tolerated
- PTX-COVID19-B exhibited strong virus neutralization capability across the 16µg, 40µg and 100µg dose cohorts
- PTX-COVID19-B 40µg dose was selected for Phase 2 study
- PTX-COVID19-B will be evaluated in additional Phase 1 population cohorts
The Phase 1 Study was designed with dose-escalations and was performed in seronegative adult subjects without evidence of recent exposure to COVID-19. The subjects were randomized to receive either the PTX-COVID19-B vaccine or a placebo in a 3:1 ratio. A total of 60 subjects participated in the Phase 1 Study.
The overall results of the Phase 1 Study are that PTX-COVID19-B was safe and well tolerated at the three dose levels of 16µg, 40µg and 100µg. Adverse events identified in the Phase 1 Study were generally mild to moderate in severity, self-resolving and transient. There were no serious adverse events reported in the Phase 1 Study. The most common adverse event reported in the Phase 1 Study was redness and pain at the injection site. Systemic reactions reported in the Phase 1 Study were generally mild to moderate and well tolerated with headache being the most common reaction reported. The reported adverse events of the Phase 1 Study were in line with the expectations of management of Providence as they compare very favorably to the adverse events data published on other mRNA vaccines for COVID-19 that have been approved for use by various health authorities around the world.
Based on the results of the Phase 1 Study, Providence intends to use a 40µg dose for the Phase 2 study of PTX-COVID19-B that is anticipated to be initiated in June 2021. Additional Phase 1 studies in adolescent and elderly populations are also planned to be undertaken by Providence.
PTX-COVID19-B vaccination induced high anti-S IgG antibodies:
Participants in the Phase 1 Study were vaccinated on day zero and day twenty-eight. Plasma samples were collected on day 1, day 8, day 28 (prior to the participant receiving the second dose), and day 42 to determine levels of IgG anti-S protein using electrochemiluminescence (“ECL”) assays from Meso Scale Discovery (“MSD”). Study participants in all three vaccine dose cohorts of the Phase 1 Study developed a strong IgG antibody response against Spike protein that was detected by day 28 and enhanced by day 42. No antibodies against S protein were detected in participants in the Phase 1 Study injected with placebo. The highest levels of antibodies were found in the 40 and 100 µg doses. By day 42, PTX-COVID19-B vaccinated participants had more than one log higher antibody levels than convalescent subjects-plasma (indicated in the dotted line) which was evaluated in the same assay.
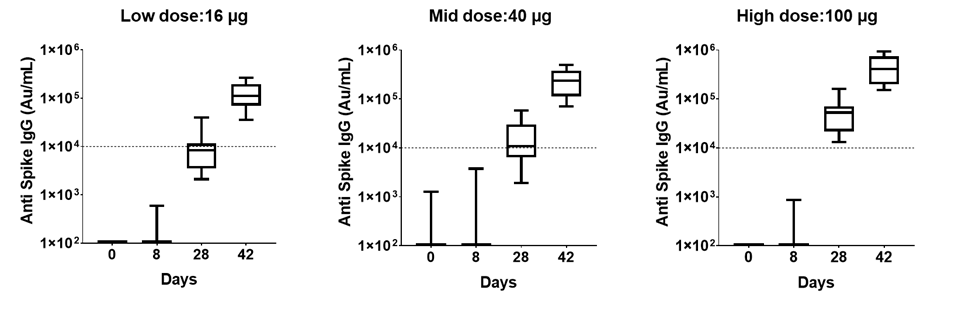
Based on the interim data of the Phase 1 Study, the level of antibodies produced in participants by PTX-COVID19-B compare favorably to the levels of antibodies produced by other mRNA vaccines that have been approved for use against COVID-19 based on the recently published report from Stanford University, where IgG responses in individuals vaccinated with the COVID-19 mRNA vaccine compared to COVID-19 infected patients were evaluated[1].
PTX-COVID19-B vaccination induced high neutralizing antibody levels:
Neutralizing activity from the Phase 1 Study participants’ plasma was evaluated by S-ACE2 MSD assay. The results indicate that the antibodies block the interaction between S protein with the ACE2 receptor and the decrease in ECL signal is used to calculate percentage inhibition of the plasma at the same dilution. All participants in the Phase 1 Study from the 16, 40 and 100 µg dose levels showed blocking activity by day 28 and all of them reached 100% blocking activity by day 42 with samples diluted 1:100 or greater. Moreover, the quantification of the antibody levels in ng/mL with a reference standard showed that all participants in the Phase 1 Study produced neutralizing antibodies by day 28 with the first immunization and increase ten-fold by day 42, two weeks after the administration of the second dose. These results indicate that PTX-COVID19-B induced a strong neutralizing antibody response which compares very favorably to the published results of other mRNA vaccines. Further studies are being conducted by Providence to determine neutralization activity using a pseudo-virus assay.
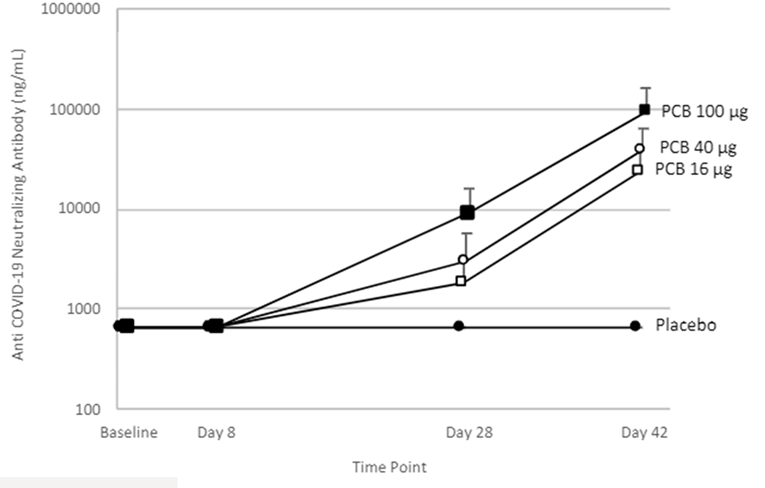
Providence intends to advance a Phase 2 clinical trial in early June 2021, with multiple trial sites in Canada. The Phase 2 clinical trial is anticipated to be structured as a comparator trial using Pfizer/BioNTech vaccine as the positive control.
About Providence Therapeutics
Providence is a leading Canadian clinical stage biotechnology company pioneering mRNA therapeutics and vaccines with operations in Calgary, Alberta and Toronto, Ontario. In response to a worldwide need for a COVID-19 vaccine, Providence expanded its focus beyond oncology therapies and devoted its energy and resources to develop a world-class mRNA vaccine for COVID-19. Providence is focused on serving the needs of Canada, and other countries that may be underserved by large pharmaceutical programs. For more information, please visit providencetherapeutics.com.
References
- ^ “Canadian company urges human trials after COVID-19 vaccine results in mice”. Lethbridge News Now. 5 August 2020. Retrieved 19 March 2021.
- ^ Jump up to:a b c d e f g h Tasker, John Paul (30 April 2021). “COVID-19 vaccine maker Providence says it’s leaving Canada after calls for more federal support go unanswered”. CBC News. Retrieved 1 May 2021.
- ^ Jump up to:a b c Stephenson, Amanda (26 January 2021). “Made-in-Canada COVID vaccine to be manufactured in Calgary”. Calgary Herald. Retrieved 22 March 2021.
- ^ Clinical trial number NCT04765436 for “PTX-COVID19-B, an mRNA Humoral Vaccine, is Intended for Prevention of COVID-19 in a General Population. This Study is Designed to Evaluate Safety, Tolerability, and Immunogenicity of PTX-COVID19-B Vaccine in Healthy Seronegative Adults Aged 18-64” at ClinicalTrials.gov
- ^ “Providence Therapeutics Holdings Inc: PTX-COVID19-B”. Montreal: McGill University. Retrieved 19 March 2021.
- ^ “Made-in-Canada coronavirus vaccine starts human clinical trials”. Canadian Broadcasting Corporation. 26 January 2021.
- ^ Jump up to:a b “Company Profile”. PitchBook.
- ^ Jump up to:a b “Company Profile”. DNB.
- ^ Jump up to:a b Code, Jillian (5 February 2021). “‘Do something’ Made-In-Canada vaccine CEO pleads for federal government to respond”. CTV News. Calgary, Alberta. Retrieved 22 March 2021.
- ^ Jump up to:a b Fieldberg, Alesia (26 January 2021). “Providence Therapeutics begins first clinical trials of Canadian-made COVID-19 vaccine”. CTV. Retrieved 2 May 2021.
- ^ Jump up to:a b c d “Manitoba Supports Made-In-Canada COVID-19 Vaccine to Protect Manitobans” (Press release). 11 February 2021. Retrieved 3 May 2021.
- ^ Providence Therapeutics Holdings Inc.: a Phase I, First-in-Human, Observer-Blinded, Randomized, Placebo Controlled, Ascending Dose Study to Evaluate the Safety, Tolerability, and Immunogenicity of PTX-COVID19-B Vaccine in Healthy Seronegative Adults Aged 18-64 (Report). Clinical Trials via U.S. National Library of Medicine. 19 February 2021. Retrieved 1 May2021.
- ^ Jump up to:a b c Gibson, Shane (11 February 2021). “Manitoba agrees to purchase 2M doses of Providence Therapeutics coronavirus vaccine”. Global News. Retrieved 2 May 2021.
- ^ “Providence Therapeutics begins first clinical trials of Canadian-made COVID-19 vaccine”. CTV. Retrieved 2 May 2021.
- ^ Jump up to:a b c Evidence (PDF), 43rd Parliament, 2nd Session. Standing Committee on International Trade, 29 April 2021, retrieved 2 May2021
- ^ Sorenson, Brad (5 February 2021). “An Open Letter to the Government of Canada”. Retrieved 3 May 2021.
- ^ Dyer, Steven. “‘Canada had an opportunity’, Calgary company explores taking vaccine development out of Canada”. CTV. Retrieved 2 May 2021.
| Vaccine description | |
|---|---|
| Target | SARS-CoV-2 |
| Vaccine type | mRNA |
| Clinical data | |
| Routes of administration | Intramuscular |
| Part of a series on the |
| COVID-19 pandemic |
|---|
| COVID-19 (disease)SARS-CoV-2 (virus) |
| showTimeline |
| showLocations |
| showInternational response |
| showMedical response |
| showImpact |
| COVID-19 portal |
////////PTX-COVID19-B, canada, hyderabad, providence, Gennova Biopharmaceuticals, biological e, COVID-19, SARS-CoV-2 , corona virus, covid 19, phase 1
NEW DRUG APPROVALS
ONE TIME
$10.00
Hyderabad. India to Host Industrial Organic Chemistry Workshops in February 2018
Dr Will Watson, an expert in Chemical Development and related fields, from Scientific Update will be visiting India in February to deliver two important workshops for Industrial Process Chemists:
Chemical Development and Scale Up in the Fine Chemical and Pharmaceutical Industries, February 5th – 7th 2018, Hyderabad, India
Practical Crystallisation & Polymorphism, February 8th & 9th 2018, Hyderabad, India
Discounts are available for groups – please contact sciup@scientificupdate.com for more information.
Lifetime achievement award, WHC17, in Hyderabad, Telangana, India 22 Aug 2017
Lifetime achievement award ……..WORLD HEALTH CONGRESS 2017 in Hyderabad, 22 aug 2017 at JNTUH KUKATPALLY. HYDERABAD, TELANGANA, INDIA, Award given by Dr. M Sunitha Reddy Head of the Department, Centre for Pharmaceutical Sciences, Institute of Science &Technology, JNTU-H, Kukatpally, Hyderabad, India
Speaking at World health congress 2017….JNTUH Hyderabad 22 aug 2017
Register Today for the ACS Symposium in India on Recent Advances in Drug Development, 11-12 November 2016 in Hyderabad, India
Inaugural ACS Industry Symposium, 11-12 November 2016 in Hyderabad, India
Recent Advances in Drug Development
Register Today for the ACS Symposium in India on Recent Advances in Drug Development
To view this email as a web page, go here.
Register now for the inaugural ACS Industry Symposium, 11-12 November 2016 in Hyderabad, India. Be sure to secure your seat today as rates will increase on 27 October!
http://acssymposium.org.in/
The theme of the Symposium is Recent Advances in Drug Development. The event will feature lectures by the world’s leading researchers and experts in the pharma industry, including:
- Dr. Peter Senter of Seattle Genetics
- Dr. Jagath Reddy Junutula of Cellerant Therapeutics, Inc.
- Dr. Ming-Wei Wang of the Shanghai Institute of Materia Medica, Chinese Academy of Sciences
This is an exclusive event being organized in partnership with Dr. Reddy’s Laboratories for pharma professionals throughout India. Space is limited so register today!
Please visit our website to learn more about the speakers and the program.
Register today to ensure your access to the ACS Industry Symposium. We look forward to seeing you in Hyderabad in November.
CAS
2540 Olentangy River Rd Columbus, OH 43202 US
CAS
2540 Olentangy River Rd Columbus, OH 43202 US
Inaugural ACS Industry Symposium, 11-12 November 2016 in Hyderabad, India
Recent Advances in Drug Development
/////// ACS Symposium, Recent Advances in Drug Development, 11-12 November 2016, Hyderabad, India, dr reddys, cas
Sreeni Labs Private Limited, Hyderabad, India ready to deliver New, Economical, Scalable Routes to your advanced intermediates & API’s in early Clinical Drug Development Stages
Sreeni Labs Private Limited, Hyderabad, India is ready to take up challenging synthesis projects from your preclinical and clinical development and supply from few grams to multi-kilo quantities. Sreeni Labs has proven route scouting ability to design and develop innovative, cost effective, scalable routes by using readily available and inexpensive starting materials. The selected route will be further developed into a robust process and demonstrate on kilo gram scale and produce 100’s of kilos of in a relatively short time.
Accelerate your early development at competitive price by taking your route selection, process development and material supply challenges (gram scale to kilogram scale) to Sreeni Labs…………
INTRODUCTION
Sreeni Labs based in Hyderabad, India is working with various global customers and solving variety of challenging synthesis problems. Their customer base ranges from USA, Canada, India and Europe. Sreeni labs Managing Director, Dr. Sreenivasa Reddy Mundla has worked at Procter & Gamble Pharmaceuticals and Eli Lilly based in USA.
The main strength of Sreeni Labs is in the design, development of innovative and highly economical synthetic routes and development of a selected route into a robust process followed by production of quality product from 100 grams to 100s of kg scale. Sreeni Labs main motto is adding value in everything they do.
They have helped number of customers from virtual biotech, big pharma, specialty chemicals, catalog companies, and academic researchers and drug developers, solar energy researchers at universities and institutions by successfully developing highly economical and simple chemistry routes to number of products that were made either by very lengthy synthetic routes or by using highly dangerous reagents and Suzuki coupling steps. They are able to supply materials from gram scale to multi kilo scale in a relatively short time by developing very short and efficient synthetic routes to a number of advanced intermediates, specialty chemicals, APIs and reference compounds. They also helped customers by drastically reducing number of steps, telescoping few steps into a single pot. For some projects, Sreeni Labs was able to develop simple chemistry and avoided use of palladium & expensive ligands. They always begin the project with end in the mind and design simple chemistry and also use readily available or easy to prepare starting materials in their design of synthetic routes
Over the years, Sreeni labs has successfully made a variety of products ranging from few mg to several kilogram scale. Sreeni labs has plenty of experience in making small select libraries of compounds, carbocyclic compounds like complex terpenoids, retinal derivatives, alkaloids, and heterocyclic compounds like multi substituted beta carbolines, pyridines, quinolines, quinolones, imidazoles, aminoimidazoles, quinoxalines, indoles, benzimidazoles, thiazoles, oxazoles, isoxazoles, carbazoles, benzothiazoles, azapines, benzazpines, natural and unnatural aminoacids, tetrapeptides, substituted oligomers of thiophenes and fused thiophenes, RAFT reagents, isocyanates, variety of ligands, heteroaryl, biaryl, triaryl compounds, process impurities and metabolites.
Sreeni Labs is Looking for any potential opportunities where people need development of cost effective scalable routes followed by quick scale up to produce quality products in the pharmaceutical & specialty chemicals area. They can also take up custom synthesis and scale up of medchem analogues and building blocks. They have flexible business model that will be in sink with customers. One can test their abilities & capabilities by giving couple of PO based (fee for service) projects.
Some of the compounds prepared by Sreeni labs;
See presentation below
LINK ON SLIDESHARE
Sreeni Labs Profile from Sreenivasa Reddy
Managing Director at Sreeni Labs Private Limited\
Few Case Studies : Source SEEENI LABS
QUOTE………….
One virtual biotech company customer from USA, through a common friend approached Sreeni Labs and told that they are buying a tetrapeptide from Bachem on mg scale at a very high price and requested us to see if we can make 5g. We accepted the challenge and developed solution phase chemistry and delivered 6g and also the process procedures in 10 weeks time. The customer told that they are using same procedures with very minor modifications and produced the tetrapeptide ip to 100kg scale as the molecule is in Phase III.
One East coast customer in our first meeting told that they are working with 4 CROs of which two are in India and two are in China and politely asked why they should work with Sreeni Labs. We told that give us a project where your CROs failed to deliver and we will give a quote and work on it. You pay us only if we deliver and you satisfy with the data. They immediately gave us a project to make 1.5g and we delivered 2g product in 9 weeks. After receiving product and the data, the customer was extremely happy as their previous CRO couldn’t deliver even a milligram in four months with 3 FTEs.
One Midwest biotech company was struggling to remove palladium from final API as they were doing a Suzuki coupling with a very expensive aryl pinacol borane and bromo pyridine derivative with an expensive ligand and relatively large amount of palldium acetate. The cost of final step catalyst, ligand and the palladium scavenging resin were making the project not viable even though the product is generating excellent data in the clinic. At this point we signed an FTE agreement with them and in four months time, we were able to design and develop a non suzuki route based on acid base chemistry and made 15g of API and compared the analytical data and purity with the Suzuki route API. This solved all three problems and the customer was very pleased with the outcome.
One big pharma customer from east coast, wrote a structure of chemical intermediate on a paper napkin in our first meeting and asked us to see if we can make it. We told that we can make it and in less than 3 weeks time we made a gram sample and shared the analytical data. The customer was very pleased and asked us to make 500g. We delivered in 4 weeks and in the next three months we supplied 25kg of the same product.
Through a common friend reference, a European customer from a an academic institute, sent us an email requesting us to quote for 20mg of a compound with compound number mentioned in J. med. chem. paper. It is a polycyclic compound with four contiguous stereogenic centers. We gave a quote and delivered 35 mg of product with full analytical data which was more pure than the published in literature. Later on we made 8g and 6g of the same product.
One West coast customer approached us through a common friend’s reference and told that they need to improve the chemistry of an advanced intermediate for their next campaign. At that time they are planning to make 15kg of that intermediate and purchased 50kg of starting raw material for $250,000. They also put five FTEs at a CRO for 5 months to optimize the remaining 5 steps wherein they are using LAH, Sodium azide, palladium catalyst and a column chromatography. We requested the customer not to purchase the 50kg raw material, and offered that we will make the 15kg for the price of raw material through a new route in less than three months time. You pay us only after we deliver 15 kg material. The customer didn’t want to take a chance with their timeline as they didn’t work with us before but requested us to develop the chemistry. In 7 weeks time, we developed a very simple four step route for their advanced intermediate and made 50g. We used very inexpensive and readily available starting material. Our route gave three solid intermediates and completely eliminated chromatographic purifications.
One of my former colleague introduced an academic group in midwest and brought us a medchem project requiring synthesis of 65 challenging polyene compounds on 100mg scale. We designed synthetic routes and successfully prepared 60 compounds in a 15 month time.
UNQUOTE…………
The man behind Seeni labs is Dr. Sreenivasa Reddy Mundla

Dr. Sreenivasa Reddy Mundla.
Managing Director at Sreeni Labs Private Limited
Sreeni Labs Private Limited
Road No:12, Plot No:24,25,26
- IDA, Nacharam
Hyderabad, 500076
Telangana State, India
Links
LINKEDIN https://in.linkedin.com/in/sreenivasa-reddy-10b5876
FACEBOOK https://www.facebook.com/sreenivasa.mundla
RESEARCHGATE https://www.researchgate.net/profile/Sreenivasa_Mundla/info
EMAIL mundlasr@hotmail.com, Info@sreenilabs.com, Sreeni@sreenilabs.com
Dr. Sreenivasa Reddy Mundla
Dr. M. Sreenivasa Reddy obtained Ph.D from University of Hyderabad under the direction Prof Professor Goverdhan Mehta in 1992. From 1992-1994, he was a post doctoral fellow at University of Wisconsin in Professor Jame Cook’s lab. From 1994 to 2000, worked at Chemical process R&D at Procter & Gamble Pharmaceuticals (P&G). From 2001 to 2007 worked at Global Chemical Process R&D at Eli Lilly and Company in Indianapolis.
In 2007 resigned to his job and founded Sreeni Labs based in Hyderabad, Telangana, India and started working with various global customers and solving various challenging synthesis problems.
The main strength of Sreeni Labs is in the design, development of a novel chemical route and its development into a robust process followed by production of quality product from 100 grams to 100’s of kg scale.
They have helped number of customers by successfully developing highly economical simple chemistry routes to number of products that were made by Suzuki coupling. they are able to shorten the route by drastically reducing number of steps, avoiding use of palladium & expensive ligands. they always use readily available or easy to prepare starting materials in their design of synthetic routes.
Sreeni Labs is Looking for any potential opportunities where people need development of cost effective scalable routes followed by quick scale up to produce quality products in the pharmaceutical & specialty chemicals area. They have flexible business model that will be in sink with customers. One can test their abilities & capabilities by giving PO based projects
Experience
Founder & Managing Director
Sreeni Labs Private Limited
August 2007 – Present (8 years 11 months)
Sreeni Labs Profile
Principal Research Scientist
Eli Lilly and Company
March 2001 – August 2007 (6 years 6 months)
Senior Research Scientist
Procter & Gamble
July 1994 – February 2001 (6 years 8 months)
Education
University of Hyderabad
Doctor of Philosophy (Ph.D.),
1986 – 1992

With Sreenivasa Mundla, Narahara sastry, Ram Kishan Rao, Jagadeesh Bharatam, Jagadish Gunjur and Jagadish Bharatham.
PUBLICATIONS
Jianye Zhang · Zhiqian Dong · Sreenivasa Reddy Mundla · X Eric Hu · William Seibel ·Ruben Papoian · Krzysztof Palczewski · Marcin Golczak
Article: ChemInform Abstract: Regioselective Synthesis of 4Halo ortho-Dinitrobenzene Derivative
Aug 2010 · ChemInform
Hong-yu Li · William T. McMillen · Charles R. Heap · Denis J. McCann · Lei Yan · Robert M. Campbell · Sreenivasa R. Mundla · Chi-Hsin R. King · Elizabeth A. Dierks · Bryan D. Anderson · Karen S. Britt · Karen L. Huss
Apr 2008 · Journal of Medicinal Chemistry
Hong-yu Li · Yan Wang · William T. McMillen · Arindam Chatterjee · John E. Toth ·Sreenivasa R. Mundla · Matthew Voss · Robert D. Boyer · J. Scott Sawyer
Feb 2008 · ChemInform
Hong-yu Li · Yan Wang · William T. McMillen · Arindam Chatterjee · John E. Toth ·Sreenivasa R. Mundla · Matthew Voss · Robert D. Boyer · J. Scott Sawyer
Nov 2007 · Tetrahedron
Hong-yu Li · Yan Wang · Charles R Heap · Chi-Hsin R King · Sreenivasa R Mundla · Matthew Voss · David K Clawson · Lei Yan · Robert M Campbell · Bryan D Anderson · Jill R Wagner ·Karen Britt · Ku X Lu · William T McMillen · Jonathan M Yingling
Apr 2006 · Journal of Medicinal Chemistry
Hui Cao · Sreenivasa R. Mundla · James M. Cook
Aug 2003 · Tetrahedron Letters
Article: ChemInform Abstract: A New Method for the Synthesis of 2,6-Dinitro and 2Halo6-nitrostyrenes
Nov 2000 · ChemInform
Article: ChemInform Abstract: A Novel Method for the Efficient Synthesis of 2-Arylamino-2-imidazolines
TGF-β inhibitors
The present invention provides 2-(6-methyl-pyridin-2-yl)-3-[6-amido-quinolin-4-yl) -5,6-dihydro-4H-pyrrolo[1,2-b]pyrazole monohydrate, i.e., Formula I.
EXAMPLE 1 Preparation of 2-(6-methyl-pyridin-2-yl)-3-[6-amido-quinolin-4-yl-5,6-dihydro-4H -pyrrolo[1,2-b]pyrazole monohydrate
Galunisertib
1H NMR (CDCl3): δ=9.0 ppm (d, 4.4 Hz, 1H); 8.23-8.19 ppm (m, 2H); 8.315 ppm (dd, 1.9 Hz, 8.9 Hz, 1H); 7.455 ppm (d, 4.4 Hz, 1H); 7.364 ppm (t, 7.7 Hz, 1H); 7.086 ppm (d, 8.0 Hz, 1H); 6.969 ppm (d, 7.7 Hz, 1H); 6.022 ppm (m, 1H); 5.497 ppm (m, 1H); 4.419 ppm (t, 7.3 Hz, 2H); 2.999 ppm (m, 2H); 2.770 ppm (p, 7.2 Hz, 7.4 Hz, 2H); 2.306 ppm (s, 3H); 1.817 ppm (m, 2H). MS ES+: 370.2; Exact: 369.16
ABOVE MOLECULE IS
https://newdrugapprovals.org/2016/05/04/galunisertib/
Galunisertib
Phase III
A TGF-beta receptor type-1 inhibitor potentially for the treatment of myelodysplastic syndrome (MDS) and solid tumours.
LY-2157299
CAS No.700874-72-2
READ MY PRESENTATION ON
KEYWORDS Sreenivasa Mundla Reddy, Managing Director, Sreeni Labs Private Limited, Hyderabad, Telangana, India, new, economical, scalable routes, early clinical drug development stages, Custom synthesis, custom manufacturing, drug discovery, PHASE 1, PHASE 2, PHASE 3, API, drugs, medicines
Flow Chemistry Symposium & Workshop 16-17 June at IICT, Hyderabad, India
MESSAGE FROM VIJAY KIRPALANI
A 2-day FLOW CHEMISTRY Symposium + Workshop has been organized on 16-17 June 2016 at
IICT Hyderabad, India by Flow Chemistry Society – India Chapter (in collaboration with IICT-Hyderabad & IIT-B)
with speakers from India, UK, Netherlands and Hungary.
Both days have intensive interactive sessions on the theory and industrial applications of Flow Chemistry followed by live demonstrations using 7 different Flow Reactor platforms — from microliters to 10,000 L/day industrial scale.
The Fees are Rs. 5,000 for Industry Delegates and Rs. 2,500 for Academic Delegates (+15% Service Tax) : contact : vk@pi-inc.co or msingh@cipla.com
I have attached a detailed program and look forward to meeting you at the event..

Best regards
Vijay Kirpalani
President
Flow Chemistry Society – India Chapter
email : vk@pi-inc.co
Tel: +91-9321342022 // +91-9821342022
ABOUT
IICT, Hyderabad, India



Dr. S. Chandrasekhar,
Director
CSIR-Indian Institute of Chemical Technology (IICT)
Hyderabad, India
SPEAKERS

Mr Vijay Kirpalani
President
Flow Chemistry Society – India Chapter, INDIA
Dr Charlotte Wiles , CHEMTRIX
UK &THE NETHERLANDS,UNIV OF HULL
Prof Anil Kumar( IIT-B), INDIA






/////Flow Chemistry, Symposium, Workshop, 16-17 June, IICT, Hyderabad, India
WO 2016012938, New patent, LINACLOTIDE, DR. REDDY’S LABORATORIES LIMITED,
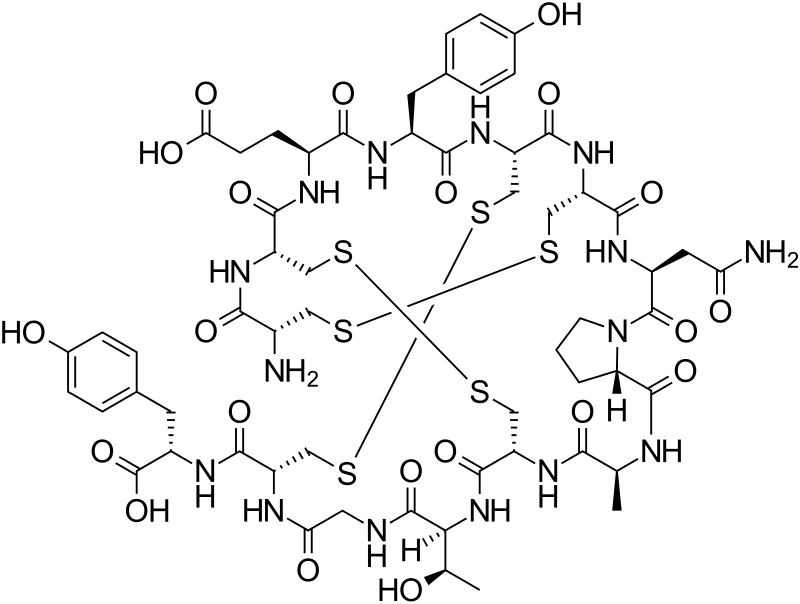
WO2016012938, IMPROVED PROCESS FOR PREPARATION OF AMORPHOUS LINACLOTIDE
DR. REDDY’S LABORATORIES LIMITED [IN/IN]; 8-2-337, Road No 3, Banjara Hills, Telangana, INDIA Hyderabad 500034 (IN)
KALITA, Dipak; (IN).
NIVRUTTI, Ramrao Jogdand; (IN).
BALAKUMARAN, Kesavan; (IN).
DESHMUKH, Shivshankar; (IN).
VUTUKURU, Naga Chandra Sekhar; (IN).
KASINA, Vara Prasad; (IN).
NALAMOTHU, Sivannarayana; (IN).
VILVA, Mohan Sundaram; (IN).
KHAN, Rashid Abdul Rehman; (IN).
TIRUMALAREDDY, Ramreddy; (IN).
MUSTOORI, Sairam; (IN)

The present application relates to an improved process for the formation of disulfide bonds in linaclotide. The present application also relates to an improved process for the purification of linaclotide.
The present application relates to an improved process for the preparation of amorphous linaclotide. Specifically, the present application relates to an improved process for the formation of disulfide bonds in linaclotide. The present application further relates to a purification process for the preparation of amorphous linaclotide.
INTRODUCTION
Linaclotide is a 14-residue peptide which is an agonist of the guanylate cyclase type-C receptor. Linaclotide may be used for the treatment of chronic constipation and irritable bowel syndrome. Structurally, linaclotide has three disulfide bonds and they are present between Cys1-Cys6, Cys2-Cys-10 and Cys5-Cys13. The structure of linaclotide is shown below:
1 2 3 4 5 6 7 8- 9 10 11 12 13 14

Benitez et al. Peptide Science, 2010, Vol. 96, No. 1 , 69-80 discloses a process for the preparation of linaclotide. The process involves the use of 2-chlorotrityl (CTC) resin and 9-fluorenylmethoxycarbonyl (Fmoc) chemistry. The Cys residues are protected by Trt (trityl) group. The amino acids are coupled to one another using 3 equivalents of 1 -[bis(dimethylamino)methylene]-6-chloro-1 H-benzotriazolium hexafluorophosphate 3-oxide (HCTU) as coupling agent and 6 equivalents of diisoprpylethylamine (DIEA) as base in dimethylformamide (DMF). The Fmoc group is removed using piperidine-DMF (1 :4). The Cys residues are incorporated using 3 equivalents of Ν,Ν’-diisopropylcarbodiimide (DIPCDI) as coupling agent and 3 equivalents of 1 -hydroxybenzotriazole (HOBt) as an activating agent. After the elongation of the peptide chain, the peptide was cleaved from the solid support (CTC resin) by first treating with 1 % trifluoroacetic acid (TFA) and then with a mixture of TFA, triisoprpylsilane (TIS) and water in the ratio of 95:2.5:2.5. The disulfide bonds are prepared by subjecting the linear peptide to air oxidation in sodium dihydrogen phosphate (100 mM) and guanidine hydrochloride buffer (2 mM).
US2010/261877A1 discloses a process for purification of linaclotide. The process involves first purification of crude peptide by reverse-phase chromatographic purification followed by concentrating the purified pools and dissolving the purified linaclotide in aqueous-isopropanol or aqueous-ethanol and spray-drying the solution to afford pure Linaclotide.
The synthesis of a peptide containing disulfide bridges is difficult for two main reasons; one is potential risk of racemization during the formation of linear chain and the other is mis-folding of the disulfide bridges. Hence, there is a need in the art to a cost-effective process for the preparation of pure linaclotide.
EXAMPLES
Example 1 : Preparation of Crude Linaclotide using polyvinyl polymer bound complex of sulfur trioxide-pyridine
The linear chain of peptide of formula (I) (0.1 g) and polyvinyl polymer bound complex of sulfur trioxide-pyridine (0.062 g) was charged in water (100 mL). The pH of the reaction mass was adjusted to 8.5 to 9 by addition of ammonium hydroxide. The reaction mass was stirred at 25 °C for 15 hours and trifluoroacetic acid (2 mL) was added to the reaction mass to adjust the pH up to 2-2.5. The reaction mass was stirred for 3 hours at the same temperature to afford crude linaclotide.
HPLC Purity: 59.92%
Example 2: Preparation of Crude Linaclotide using DMSO in water
The pH of water (100 ml_) was adjusted to 9.1 by the addition of aqueous ammonia. DMSO (1 ml_) and linear chain of peptide of formula (I) (100 mg) were charged. The reaction mass was stirred for 17 hours at 25 °C and acidified with trifluoroacetic acid to pH 1 .9 and stirred for 8 hours at the same temperature to afford crude linaclotide.
HPLC Purity: 57%
Example 3: Preparation of Crude Linaclotide using DMSO in water
The pH of water (1500 ml_) was adjusted to 9 by the addition of aqueous ammonia. DMSO (15 ml_) and linear chain of peptide of formula (I) (15 g) were charged. The reaction mass was stirred for 17 hours at 25 °C and acidified with acetic acid to pH 1 .9 and stirred for 8 hours at the same temperature to obtain crude linaclotide.
HPLC Purity: 46.02%
Example 4: Preparation of Crude Linaclotide in water
To a mixture of water (1900 mL) and ammonium sulfate (26.4 g), ammonium hydroxide was added drop wise to adjust the pH up to 8.5. Linear chain of peptide of formula (I) (26.4 g) was added and the reaction mass was stirred for 8 hours at 25 °C. Trifluoroacetic acid (20 mL) was added drop wise and the reaction mixture was stirred for 15 hours at 25 °C to afford crude linaclotide.
HPLC Purity: 63.38%
Example 5: Preparation of Crude Linaclotide using a complex of pyridine-sulfur trioxide
Linear chain of peptide of formula (I) (0.2 g) was added to water (250 mL) and the pH of the reaction mass was adjusted to 8.91 by the drop wise addition of aqueous ammonia. A complex of pyridine-sulfur trioxide (0.124 g) was added to the reaction mass and stirred for 16 hours at 25 °C. Another lot of complex of pyridine-sulfur trioxide (0.124 g) was added to the reaction mass and stirred for 5 hours at 25 °C to afford crude linaclotide.
Example 6: Preparation of Crude Linaclotide using guanidine hydrochloride
To a solution of sodium bicarbonate (0.89 g) in water (100 mL), cysteine (0.363 g), cysteine (0.072 g) and guanidine hydrochloride (9.50 g) were charged. Acetonitrile (15 mL) and linear chain of peptide of formula (I) (0.1 g) was added to the reaction mass.
The reaction mass was stirred for 3 hours at 25 °C and trifluoroacetic acid (2 mL) was added. The reaction mass was stirred for 18 hours at the same temperature. Another lot of trifluoroacetic acid (2 mL) was added to the reaction mass and stirred for 18 hours at the same temperature to afford crude linaclotide.
Example 7: Preparation of Crude Linaclotide using Clear-OX™
Pre-conditioned Clear-Ox™ (0.5 g) was added to a solution of ammonium sulfate (1 .32 g) in water (100 mL) of pH 8.5, adjusted by addition of ammonium hydroxide. The linear chain of peptide of formula (I) (0.1 g) was added to the reaction mass and stirred for 3 hours at 25 °C. Another lot of Pre-conditioned Clear-Ox™ (0.5 g) was added to the reaction mass and stirred for 1 .30 hours. Trifluoroacetic acid (2 mL) was added to the reaction mass and stirred for 16 hours at the same temperature to afford crude linaclotide.
HPLC Purity: 67.5%
Example 8: Preparation of Crude Linaclotide using reduced Glutathione
To a mixture of ammonium sulphate (5.28 g) in water (400 mL) and isopropyl alcohol (400 mL), reduced glutathione (0.248 g) was added and the pH was adjusted to 8.5 by using aqueous ammonia. The linear chain of peptide of formula (I) (0.81 g) was added to the reaction mixture and stirred at ambient temperature for 17 hours. Isopropyl alcohol was evaporated under vacuum to afford crude linaclotide.
HPLC Purity: 69.56%%
Example 9: Preparation of Crude Linaclotide using DMSO and air bubbling
To a mixture of water (95 mL) and ammonium sulfate (1 .32 g), ammonium hydroxide was added drop wise to adjust the pH up to 8.5. Linear chain of peptide of formula (I) (0.1 g) and DMSO (5 mL) was added and the reaction mass was stirred for 20 hours at 25 °C with continuous air bubbling. Trifluoroacetic acid (2 mL) was added to the reaction mass and stirred for 19 hours with continuous air bubbling at the same temperature to afford the title product.
HPLC Purity: 59.1 1 %
Example 10: Preparation of Crude Linaclotide using solid supported TEMPO
To a mixture of water (100 mL) and silica bound TEMPO (0.01 g), linear chain of peptide of formula (I) (0.1 g) and sodium hypochlorite solution (1 mL) were added and the reaction mass was stirred 18 hours at 25 °C. Another lot of sodium hypochlorite solution (0.5 mL) was added to the reaction mass and stirred for further 7 hours at the same temperature to afford title product.
HPLC Purity: 42.70%………………see more in patent
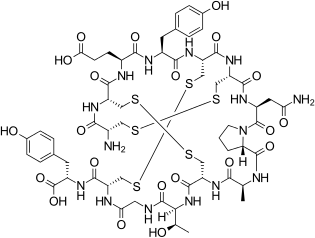 |
|
| Systematic (IUPAC) name | |
|---|---|
|
L-Cysteinyl-L-cysteinyl-L-glutamyl-L-tyrosyl-L-cysteinyl-L-cysteinyl-L-asparaginyl-L-prolyl-L-alanyl-L-cysteinyl-L-threonylglycyl-L-cysteinyl-L-tyrosine cyclo(1-6),(2-10),(5-13)-tris(disulfide)
|
|
| Clinical data | |
| Trade names | Linzess |
| Licence data | US FDA:link |
| Pregnancy category |
|
| Legal status |
|
| Routes of administration |
Oral |
| Identifiers | |
| CAS Number | 851199-59-2 |
| ATC code | A06AX04 |
| PubChem | CID 16158208 |
| IUPHAR/BPS | 5017 |
| ChemSpider | 17314504 |
| UNII | N0TXR0XR5X |
| KEGG | D09355 |
| Chemical data | |
| Formula | C59H79N15O21S6 |
| Molar mass | 1526.74 g/mol |
///////WO 2016012938, DR. REDDY’S LABORATORIES LIMITED , Telangana, INDIA , Hyderabad, LINACLOTIDE, new patent
smiles O=C(O)[C@@H](NC(=O)[C@H]4NC(=O)CNC(=O)[C@@H](NC(=O)[C@H]2NC(=O)[C@@H](NC(=O)[C@H]5N(C(=O)[C@@H](NC(=O)[C@H]1NC(=O)[C@@H](NC(=O)[C@@H](NC(=O)[C@@H](NC(=O)[C@@H](NC(=O)[C@@H](N)CSSC1)CSSC2)CCC(=O)O)Cc3ccc(O)cc3)CSSC4)CC(=O)N)CCC5)C)[C@H](O)C)Cc6ccc(O)cc6
Process Development for Low Cost Manufacturing

-
Process Development for Low Cost Manufacturing on 23-24 nov 2015 , Hyderabad, INDIA
- 23.11.2015 – 24.11.2015


- Hotel Green Park – Hyderabad, India

- View Brochure
- or click
- https://scientificupdate.co.uk/index.php/training/scheduled-training-courses/details/213-Low-Cost-Manufacturing.html?utm_source=Scientific+Update+News&utm_campaign=d31c03d4e3-India_Courses_Hyderabad7_22_2015&utm_medium=email&utm_term=0_08c5e1fb69-d31c03d4e3-78584097
Chemical process research and development is recognised as a key function during the commercialisation of a new product particularly in the generic and contract manufacturing arms of the chemical, agrochemical and pharmaceutical industries.
The synthesis and individual processes must be economic, safe and must generate product that meets the necessary quality requirements.
This 2-day course presented by highly experienced process chemists will concentrate on the development and optimisation of efficient processes to target molecules with an emphasis on raw material cost, solvent choice, yield improvement, process efficiency and work up, and waste minimisation.
Process robustness testing and reaction optimisation via stastical methods will also be covered.
A discussion of patent issues and areas where engineering and technology can help reduce operating costs.
The use of engineering and technology solutions to reduce costs will be discussed and throughout the course the emphasis will be on minimising costs and maximising returns.

///////////

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....































































