Home » Posts tagged 'corona'
Tag Archives: corona
Olgotrelvir


Olgotrelvir
STI-1558, HY-156655, CS-0887294, STI 1558, HY 156655, CS 0887294
Cas 2763596-71-8
494.6 g/mol, C22H30N4O7S, ZP3BDH359D
C22H30N4O7S 3-Pyrrolidinepropaney, α-hydroxy-β-[[(2S)-2-[(1H-indol-2-ylcarbonyl)amino]-4-methyl-1-oxopentyl]amino]-2-oxo-, (βS,3S)-
- (2S)-2-[(S)-2-(1H-Indole-2-carboxamido)-4-methylpentanamido]-1-hydroxy-3-[(S)-2-oxopyrrolidin-3-yl]propane-1-sulfonic acid
- 3-Pyrrolidinepropanesulfonic acid, alpha-hydroxy-beta-[[(2S)-2-[(1H-indol-2-ylcarbonyl)amino]-4-methyl-1-oxopentyl]amino]-2-oxo-, (betaS,3S)-
Olgotrelvir sodium, C22H30N4O7S.Na, CAS 2763596-71-8
3-Pyrrolidinepropanesulfonic acid, α-hydroxy-β-[[(2S)-2-[(1H-indol-2-ylcarbonyl)amino]-4-methyl-1-oxopentyl]amino]-2-oxo-, sodium salt (1:1), (βS,3S)-
Olgotrelvir (STI-1558) is an experimental antiviral medication being studied as a potential treatment for COVID-19. It is believed to work by inhibiting the SARS-CoV-2 main protease (Mpro), a key enzyme that SARS-CoV-2 needs to replicate,[1][2][3][4] and by blocking viral entry.[2][5]
SCHEME

Main

PATENT
US20230322668 – PROTEASE INHIBITORS AS ANTIVIRALS
Example S1: Synthesis of Compounds A-1-a, A-1-b, A-1-c and A-1-d





US20230026438 – PROTEASE INHIBITORS AS ANTIVIRALS
WO2022256434 – PROTEASE INHIBITORS AS ANTIVIRALS
Example S1: Synthesis of Compounds A-1-a, A-1-b, A-1-c and A-1-d

[00224] To a dichloromethane (2.5 L) solution of 1H-indole-2-carboxylic acid (compound 101) (200 g, 1.24 mol) and N-hydroxy succinimide (157.1 g, 1.37 mol) was added EDCI (286 g, 1.49 mmol) at 0℃. After stirring at room temperature overnight, the solvent was removed under reduced pressure. The resulting solid was triturated with deionized water, and the solid was collected and dried under reduced pressure to give the compound 102 as a light-brown solid (310 g, 96%).
1H NMR (400 MHz, CDCl3) δ 9.01 (s, 1H), 7.70 (d, J = 8.2 Hz, 1H), 7.49 – 7.35 (m, 3H), 7.19 (t, J = 7.4 Hz, 1H), 2.92 (s, 4H).
[00225] To a stirred mixture of methyl (2S)-2-{[(tert-butoxy)carbonyl]amino}-3-[(3S)-2- oxopyrrolidin-3-yl]-propanoate (compound 103) (500 g, 1748.24 mmol) in MeOH (200 mL) was
added 4M HCl in 1,4-dioxane (2000 mL) at room temperature. The mixture was stirred at rt for 2 h. LCMS indicated completion of the reaction. The reaction mixture was concentrated under reduced pressure to afford methyl (2S)-2-amino-3-[(3S)-2-oxopyrrolidin-3-yl]propanoate hydrochloride salt (compound 104) (389 g, 1721 mmol, 98%) as a light-yellow solid, which was used for next step without further purification. LCMS= [M+H]+: 187.1.
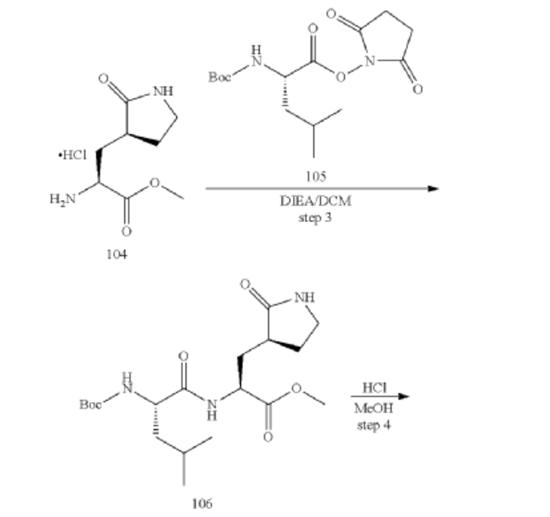
[00226] To a stirred mixture of methyl (2S)-2-amino-3-[(3S)-2-oxopyrrolidin-3-yl]propanoate hydrochloride (389 g, 1721 mmol) (compound 104) and DIEA (866.162 mL, 5240.94 mmol) in DCM (1800 mL) and EtOH (500 mL) was added 2,5-dioxopyrrolidin-1-yl (2R)-2-{[(tert-butoxy)carbonyl]amino}-4-methyl-pentanoate (compound 105) (573.66 g, 1746.98 mmol) at room temperature. The reaction mixture was stirred at room temperature for 2 h. LCMS indicated completion of the reaction. The reaction mixture was successively washed with water (1.0 L x 2), 0.5 M HCl (1.1 L), sat. NaHCO3 (1 L) and water (1 L). The organic layer was separated, dried with anhydrous Na2SO4, filtered and concentrated under reduced pressure to afford the compound 106 (700 g, 1752.23 mmol, >99%) as a light-yellow solid, which was used for next step without further purification. LCMS = [M+H]+: 400.3.
(400 MHz, DMSO-d6) δ 8.32 (d, J = 8.0 Hz, 1H), 7.62 (s, 1H), 6.88 (d, J = 8.0 Hz, 1H), 4.40 – 4.28 (m, 1H), 3.94 (dd, J = 15.1, 8.1 Hz, 1H), 3.74 – 3.52 (m, 3H), 3.15 (t, J = 8.8 Hz, 1H), 3.06 (dd, J = 16.4, 9.2 Hz, 1H), 2.33 (t, J = 9.2 Hz, 1H), 2.14 – 2.00 (m, 2H), 1.68 – 1.51 (m, 3H), 1.42 – 1.34 (m, 11H), 0.87 (dd, J = 11.4, 6.6 Hz, 6H).
[00227] A mixture of methyl (2S)-2-[(2S)-2-{[(tert-butoxy)carbonyl]amino}-4-methylpentanamido]-3-[(3S)-2-oxopyrrolidin-3-yl]propanoate (compound 106) (590 g, 1476.88 mmol) in HCl/dioxane (3 L) was stirred at room temperature for 2 h. LC-MS indicated completion of the reaction. The reaction mixture was concentrated under reduced pressure to give compound 107 as a yellow solid (490 g, 99%), which was used for next step without further purification.
LCMS = [M+H]+: 300.2.
[00228] To a stirred mixture of methyl (S)-2-((S)-2-amino-4-methylpentanamido)-3-((S)-2-oxopyrrolidin-3-yl)propanoate hydrochloride (compound 107) (418 g, 1235 mmol) and TEA (519.020 mL, 3734.03 mmol) in DMF (2500 mL) at room temperature was added 2,5-dioxopyrrolidin-1-yl 1H-indole-2-carboxylate (compound 102) (353 g, 1369.15 mmol) . The reaction mixture was stirred for 1.5 h. LCMS indicated that the reaction was complete. EtOAc (6 L) was added into the reaction mixture, which was then washed with brine (6 L x 6). The organic layers were combined, dried over anhydrous sodium sulfate, and concentrated down under reduced
pressure. Compound A-1-a was obtained as an off-white solid (414 g. Y: 76%), which was used for next step without further purification. LCMS = [M+H]+: 443.3. 1H NMR (400 MHz, DMSO-d6) δ 11.55 (s, 1H), 8.54 (t, J = 12.2 Hz, 1H), 8.40 (d, J = 8.1 Hz, 1H), 7.62 (d, J = 8.1 Hz, 2H), 7.43 (d, J = 8.2 Hz, 1H), 7.24 (t, J = 10.3 Hz, 1H), 7.18 (t, J = 7.5 Hz, 1H), 7.04 (t, J = 7.5 Hz, 1H), 4.65 – 4.50 (m, 1H), 4.44 – 4.28 (m, 1H), 3.72 – 3.55 (s, 3H), 3.19 – 3.06 (m, 2H), 2.36 (ddd, J = 13.8, 10.3, 4.0 Hz, 1H), 2.16 – 2.03 (m, 2H), 1.79 – 1.49 (m, 5H), 0.92 (dt, J = 14.4, 7.2 Hz, 6H).
[00229] To a stirred solution of methyl (S)-2-((S)-2-(1H-indole-2-carboxamido)-4-methylpentanamido)-3-((S)-2-oxopyrrolidin-3-yl)propanoate (compound A-1-a) (500 g, 1131 mmol) in THF (20 L) LiBH4 (74 g, 3393 mmol) was added portionwise at 0 ℃. The reaction mixture was stirred at 0 ℃ for 4 h. After reaction was completed (monitored by LCMS), the reaction mixture was quenched with sat. aqueous NH4Cl until no more gas formed. The mixture was washed with brine (5 L x 4), organic layer was collected, dried over anhydrous sodium sulfate, filtered, and concentrated down in vacuum. The resulting residue was purified by silica column chromatography (DCM : MeOH = 15 : 1) to give the desired product compound A-1-b (310 g, 66%) as a white solid. LCMS = [M+H]+: 415.2.
NMR (400 MHz, DMSO-d6) δ 11.57 (s, 1H), 8.39 (d, J = 8.2 Hz, 1H), 7.79 (d, J = 9.0 Hz, 1H), 7.61 (d, J = 7.9 Hz, 1H), 7.52 (s, 1H), 7.42 (d, J = 8.3 Hz, 1H), 7.26 (d, J = 1.4 Hz, 1H), 7.17 (t, J = 7.6 Hz, 1H), 7.03 (t, J = 7.5 Hz, 1H), 4.67 (t, J = 5.6 Hz, 1H), 4.50 (td, J = 9.7, 5.0 Hz, 1H), 3.80 (s, 1H), 3.40 – 3.28 (m, 1H), 3.28 – 3.20 (m, 1H), 3.15 – 2.99 (m, 2H), 2.33 – 2.20 (m, 1H), 2.12 (dt, J = 17.8, 9.4 Hz, 1H), 1.86 – 1.75 (m, 1H), 1.75 – 1.64 (m, 2H), 1.56 (ddd, J = 19.3, 9.6, 6.9 Hz, 2H), 1.45 – 1.35 (m, 1H), 0.91 (dd, J = 15.6, 6.3 Hz, 6H).
[00230] To a stirred solution of N-((S)-1-(((S)-1-hydroxy-3-((S)-2-oxopyrrolidin-3-yl)propan-2-yl)amino)-4-methyl-1-oxopentan-2-yl)-1H-indole-2-carboxamide (compound A-1-b) (8.3 g, 20 mmol) in DMSO (60 mL) was added 2-iodoxybenzoic acid (IBX) (11.2 g, 40 mmol) at room temperature. The reaction mixture was stirred at 30 ℃ for 18 h, and LCMS indicated completion of the reaction. The reaction mixture was diluted with EtOAc (300 mL) and filtered. The filtrate was washed with mixture of brine and sat. aqueous NaHCO3 (1:1 to 5:1, 200 mL x 5). The organic layer was separated, dried over anhydrous sodium sulfate, filtered, and concentrated down at rt to afford crude product. THF (40 mL) was added, and the mixture was stirred overnight at room temperature. The resulting solid was collected and dried under vacuum to yield the desired product N-((S)-4-methyl-1-oxo-1-(((S)-1-oxo-3-((S)-2-oxopyrrolidin-3-yl)-propan-2-yl)amino)pentan-2-yl)-1H-indole-2-carboxamide (compound A-1-c) as a white solid (2.5 g, 31%). LCMS = [M+H]+:
413.2. 1H NMR (400 MHz, CDCl3) δ 9.75 (s, 1H), 9.49 (s, 1H), 8.64 (s, 1H), 7.62 (d, J = 8.0 Hz, 1H), 7.40 (d, J = 8.4 Hz, 1H), 7.27 (d, J = 8.4 Hz, 1H), 7.14-7.05 (m, 2H), 7.01 (s, 1H), 6.34 (s, 1H), 4.90 (s, 1H), 4.34 (s, 1H), 3.27–3.22 (m, 2H), 2.43 (s, 1H), 2.30 (s, 1H), 2.01-1.96 (m, 1H), 1.94-1.91 (m, 1H) 1.88 – 1.65 (m, 4H), 1.00-0.98 (m, 6H).
[00231] To a stirred solution of N-((S)-4-methyl-1-oxo-1-(((S)-1-oxo-3-((S)-2-oxopyrrolidin-3-yl)propan-2-yl)amino)pentan-2-yl)-1H-indole-2-carboxamide (compound A-1-c) (31 g, 75.25 mmol) in EtOAc (300 mL) at room temperature was added a solution of NaHSO3 (27.56 mg, 72.73 mmol) in water (100 mL). The reaction mixture was heated at 50 ℃ for 3 h. After completion of reaction (monitored by LCMS), the organic layer was separated and removed. The aqueous layer was washed with EtOAc (100 mL x 5), concentrated down to remove remaining EtOAc, and then lyophilized to provide the desired product sodium (2S)-2-((S)-2-(1H-indole-2-carboxamido)-4-methylpentanamido)-1-hydroxy-3-((S)-2-oxopyrrolidin-3-yl)propane-1-sulfonate (compound A-1-d) as off-white solid (32 g, 85%). LCMS = [M-Na+2H]+: 495.2. 1H NMR (400 MHz, DMSO-d6) δ 11.57 (s, 1H), 8.45 (dd, J = 20.7, 8.2 Hz, 1H), 7.72 (dd, J = 48.9, 9.2 Hz, 1H), 7.62 (d, J = 8.1 Hz, 1H), 7.50 – 7.38 (m, 2H), 7.25 (dd, J = 5.1, 1.4 Hz, 1H), 7.18 (t, J = 7.6 Hz, 1H), 7.04 (t, J = 7.5 Hz, 1H), 5.43 (dd, J = 50.7, 5.9 Hz, 1H), 4.57 – 4.41 (m, 1H), 4.33 – 4.03 (m, 1H), 4.01 – 3.82 (m, 1H), 3.19 – 2.92 (m, 2H), 2.29 – 2.08 (m, 2H), 2.06 – 1.90 (m, 1H), 1.83 – 1.51 (m, 5H), 1.00 – 0.83 (m, 6H).
PAPER
https://www.sciencedirect.com/science/article/pii/S2666634023004026

Mechanism of action
Olgotrelvir is a prodrug that first converts to its active form, AC1115.[2][5] AC1115 is believed to work by inhibiting the SARS-CoV-2 main protease (also known as 3C-like protease). This protein is a crucial enzyme responsible for cleaving viral polyproteins into functional subunits essential for viral replication. By binding to the active site of the protease, the drug prevents this cleavage process, effectively halting viral assembly and impeding the virus’s ability to produce future virions.[1][2][3][5]
Olgotrelvir also appears to inhibit cathepsin L (CTSL),[2][5] a protein implicated in facilitating viral entry of SARS-CoV-2 into the host cell.[2][5][6]
Clinical trials
In September 2023, the drug’s developer, Sorrento Therapeutics, announced top-line data that olgotrelvir had met its primary endpoints in a phase III clinical trial that enrolled 1,212 patients with mild or moderate COVID-19. The drug appeared to shorten the recovery time of 11 COVID-19 symptoms in olgotrelvir-treated patients by 2.4 days on average compared to patients in the placebo group. The drug was also shown to reduce the viral load at day 4 in treated patients compared to the placebo group. Side effects were mostly mild and infrequent, with the most common being nausea (1.5% vs. 0.2%) and skin rash (3.3% vs. 0.3%), which occurred more often in the olgotrelvir group.[7][8][9]
References
- ^ Jump up to:a b Tong X, Keung W, Arnold LD, Stevens LJ, Pruijssers AJ, Kook S, et al. (November 2023). “Evaluation of in vitro antiviral activity of SARS-CoV-2 Mpro inhibitor pomotrelvir and cross-resistance to nirmatrelvir resistance substitutions”. Antimicrobial Agents and Chemotherapy. 67 (11): e0084023. doi:10.1128/aac.00840-23. PMC 10649086. PMID 37800975.
Other examples of Mpro inhibitors in late-stage development include STI-1558, currently in the phase 3 clinical trial in adult subjects with mild or moderate COVID-19 (NCT05716425).
- ^ Jump up to:a b c d e f Hackett DW (26 June 2023). “Second Generation Oral Mpro Inhibitor for COVID-19 Treatment Proceeds in Phase 3 Study”. Precision Vaccinations. Retrieved 27 December 2023.
- ^ Jump up to:a b “Coronavirus disease 2019 (COVID-19) emerging treatments”. BMJ Best Practice US. Archived from the original on 27 December 2023. Retrieved 27 December 2023.
- ^ Janin YL (September 2023). “On the origins of SARS-CoV-2 main protease inhibitors”. RSC Medicinal Chemistry. 15 (1): 81–118. doi:10.1039/D3MD00493G. ISSN 2632-8682. PMC 10809347. PMID 38283212. S2CID 264103864.
- ^ Jump up to:a b c d e Mao L, Shaabani N, Zhang X, Jin C, Xu W, Argent C, et al. (January 2024). “Olgotrelvir, a dual inhibitor of SARS-CoV-2 Mpro and cathepsin L, as a standalone antiviral oral intervention candidate for COVID-19”. Med (New York, N.Y.). 5 (1): 42–61.e23. doi:10.1016/j.medj.2023.12.004. PMID 38181791.
- ^ Berdowska I, Matusiewicz M (October 2021). “Cathepsin L, transmembrane peptidase/serine subfamily member 2/4, and other host proteases in COVID-19 pathogenesis – with impact on gastrointestinal tract”. World Journal of Gastroenterology. 27 (39): 6590–6600. doi:10.3748/wjg.v27.i39.6590. PMC 8554394. PMID 34754154.
- ^ Jiang R, Han B, Xu W, Zhang X, Peng C, Dang Q, et al. (June 2024). “Olgotrelvir as a Single-Agent Treatment of Nonhospitalized Patients with Covid-19”. NEJM Evidence. 3 (6): EVIDoa2400026. doi:10.1056/EVIDoa2400026. PMID 38804790.
- ^ Sherman AC, Baden LR (June 2024). “How To Measure Benefit in a Changing Pandemic – Olgotrelvir for SARS-CoV-2”. NEJM Evidence. 3 (6): EVIDe2400144. doi:10.1056/EVIDe2400144. PMID 38804789.
- ^ “Sorrento Announces Phase 3 Trial Met Primary Endpoint and Key Secondary Endpoint in Mild or Moderate COVID-19 Adult Patients Treated with Ovydso (Olgotrelvir), an Oral Mpro Inhibitor as a Standalone Treatment for COVID-19” (Press release). BioSpace. 12 September 2023. Retrieved 27 December 2023.
| Clinical data | |
|---|---|
| Trade names | Ovydso |
| Other names | STI-1558, HY-156655, CS-0887294 |
| Routes of administration | By mouth |
| Identifiers | |
| showIUPAC name | |
| CAS Number | 2763596-71-8 |
| PubChem CID | 166157331 |
| UNII | ZP3BDH359D |
| KEGG | D12777 |
| Chemical and physical data | |
| Formula | C22H30N4O7S |
| Molar mass | 494.56 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| showSMILES | |
| showInChI | |
//////Olgotrelvir, STI-1558, HY-156655, CS-0887294, STI 1558, HY 156655, CS 0887294, ZP3BDH359D
AZD1222 (ChAdOx1), Oxford–AstraZeneca COVID-19 vaccine, COVISHIELD


AZD1222 (ChAdOx1)
| Identifiers | |
|---|---|
| CAS Number | 2420395-83-9 |
ChAdOx1 nCoV- 19 Corona Virus Vaccine (Recombinant) COVISHIELD™
- DNA (recombinant simian adenovirus Ox1 ΔE1E3 vector human cytomegalovirus promoter plus human tissue plasminogen activator signal peptide fusion protein with severe acute respiratory syndrome coronavirus 2 isolate Wuhan-Hu-1 spike glycoprotein codon optimized-specifying)
The University of Oxford, AstraZeneca vaccine is a vaccine that aims to protect against COVID-19.

Manufacturer/developer: AstraZeneca, University of OxfordResearch name: AZD1222 (ChAdOx1)Vaccine type: Non-Replicating Viral VectorAdministration method: Intramuscular injection
Biological Components:
Covishield is a viral vector vaccine. It uses a weakened, non-replicating strain of Chimpanzee cold virus (adenovirus) to carry genetic material of the spike protein of SARS-CoV-2 into human cells
Vial of the Oxford–AstraZeneca vaccine manufactured by the Serum Institute of India (marketed as Covishield in India and in a few other countries).[5]
COVISHIELD INGREDIENTS
L-Histidine Ethanol
L-Histidine Hydrochloride Monohydrate,Magnesium Chloride
Hexahydrate Polysorbate 80*, Sucrose, Sodium Chloride
Disodium Edetate Dihydrate (EDTA) , Water for injection
Polysorbate 80 which is an ingredient of Covishield is known to cause anaphylactic reactions in patients as can be read here whereas Covaxin has no such component.
| NAME | DOSAGE | STRENGTH | ROUTE | LABELLER | MARKETING START | MARKETING END | ||
|---|---|---|---|---|---|---|---|---|
| Astrazeneca Covid-19 Vaccine | Injection, suspension | 50000000000 {VP}/0.5mL | Intramuscular | AstraZeneca Pharmaceuticals LP | 2020-12-22 | Not applicable |
| FORM | ROUTE | STRENGTH |
|---|---|---|
| Injection, suspension | Intramuscular | 50000000000 {VP}/0.5mL |
Storage Conditions: can be stored at 2 to 8 degrees Celsius making them convenient to store and transport.
Mechanism of Immunization: Covishield – This vaccine produces antibodies against only a specific region of the virus. It contains a portion of the DNA that codes for the spike protein (S-protein). Once inside the cells, the DNA part first needs to enter the nucleus to create its mirror image (complementary RNA). Then this RNA comes out in the cytoplasm as a messenger and starts making S-protein through a machine available for this purpose called ribosome. Since it is S-protein that provokes immunity it may not be as close to natural immunity as created by Covaxin. If there are any long-term side effects of the DNA material remaining inside the nucleus (e.g. integration in human DNA) is not yet known. So far, DNA vaccines were only being tried out for treating cancer patients and never used for preventing infections in normal subjects.
Clinical Development: Covishield has been developed by AstraZeneca with Oxford university in the UK and is being manufactured by the Serum Institute India (SII) in Pune. Covishield has completed phase 3 trials in S. Africa, Brazil and UK. 90% of the subjects in these studies were under the age of 55 making the efficacy and safety data applicable to this age group. The company has presented bridging study results in Indian population to the regulatory authorities based on which the approval was granted by DCGI. This data is not yet available in the public domain
Dosage Regimen: Covishield has been recommended to be taken in 2 doses. Observation of data from the UK shows improved protection with a gap of 12 weeks between 2 doses; though currently the expert committee set up by the Drug Controller General of India (DCGI) has recommended a gap of 4 weeks. Covaxin has been recommended to be taken in 2 doses 4 weeks apart.
Efficacy: Covishield has an average efficacy of 70% when 2 doses are administered 4 weeks apart. This data is from a meta-analysis (pooled analysis of multiple studies) of 4 Covishield trials in 11,636 patients out of which 3 trials were single blind and one double blind in 3 different countries. The efficacy of Covishield was published in The Lancet (link to the article). Observation of data has shown that the efficacy improves as the gap between the 2 doses is increased reaching a reported efficacy of 82.4% with a 12-week gap. Since, the phase-3 trials were conducted with a 4-week interval, it has become the standard.
Protection against Mutations: Preliminary research shows both vaccines are effective against the variant of the novel coronavirus first detected in the UK but there is no data on their efficacy against the mutants found in South Africa and Brazil. Data against these 2 variants is yet to be generated for both these vaccines.

. Consent: Covishield does not require any consent form as it has completed the phase-3 clinical trials
Who should not take Covishield?
Serum Institute of India’s factsheet said one should not get the Covishield vaccine if the person had a severe allergic reaction after a previous dose of this vaccine. Like Bharat Biotech, the SII factsheet also says that if a person is pregnant or plans to become pregnant or is breastfeeding she should tell the healthcare provider before taking the jab. People who have taken another anti-Covid vaccine should not take Covishield.
The ingredients of the Covishield vaccine are “L-Histidine, L-Histidine hydrochloride monohydrate, Magnesium chloride hexahydrate, Polysorbate 80, Ethanol, Sucrose, Sodium chloride, Disodium edetate dihydrate (EDTA), Water for injection,” it pointed out.
Side-effects of Covishield
Some of the very common side effects of the vaccines are tenderness, pain, warmth, redness, itching, swelling or bruising where the injection is given, generally feeling unwell, chills or feeling feverish, headache or joint aches.
Covishield is made by Serum Institute of India (SII) and Covaxin is manufactured by Bharat Biotech.
Over 50 lakh people have registered themselves on the Co-WIN portal since the window opened on Monday morning, the Centre said. Nearly 5 lakh beneficiaries above 60 or those aged 45-60 with comorbidities have received the first jab of Covid-19 vaccine till Tuesday evening.
Meanwhile, the govt has permitted all private hospitals to give Covid-19 vaccine if they adhere to the laid down norms and also asked the states and union territories to utilise the optimum capacity of private medical facilities empanelled under three categories. The states and Union Territories were also urged not to store, reserve, conserve or create a buffer stock of the COVID-19 vaccines, the Union Health Ministry said in a statement.
Sources: https://www.bbc.com/news/world-asia-india-55748124
The Oxford–AstraZeneca COVID-19 vaccine, codenamed AZD1222,[7] is a COVID-19 vaccine developed by Oxford University and AstraZeneca given by intramuscular injection, using as a vector the modified chimpanzee adenovirus ChAdOx1.[18][19][20][21] One dosing regimen showed 90% efficacy when a half-dose was followed by a full-dose after at least one month, based on mixed trials with no participants over 55 years old.[6] Another dosing regimen showed 62% efficacy when given as two full doses separated by at least one month.[6]
The research is being done by the Oxford University’s Jenner Institute and Oxford Vaccine Group with the collaboration of the Italian manufacturer Advent Srl located in Pomezia, which produced the first batch of the COVID-19 vaccine for clinical testing.[22] The team is led by Sarah Gilbert, Adrian Hill, Andrew Pollard, Teresa Lambe, Sandy Douglas and Catherine Green.[23][22]
On 30 December 2020, the vaccine was first approved for use[11][24] in the UK’s vaccination programme,[25] and the first vaccination outside of a trial was administered on 4 January 2021.[26] The vaccine has since been approved by several medicine agencies worldwide, such as the European Medicines Agency,[12][14] and the Australian Therapeutic Goods Administration (TGA),[9] and has been approved for an Emergency Use Listing (EUL) by the World Health Organization.[27]
Vaccine platform
The AZD1222 vaccine is a replication-deficient simian adenovirus vector, containing the full‐length codon‐optimised coding sequence of SARS-CoV-2 spike protein along with a tissue plasminogen activator (tPA) leader sequence.[28][29].
The adenovirus is said replication-deficient because some of its essential genes were deleted and replaced by a gene coding for the spike. Following vaccination, the adenovirus vector enters the cells, releases its genes, those are transported to the cell nucleus, thereafter the cell’s machinery does the transcription in mRNA and the translation in proteins.
The one of interest is the spike protein, an external protein that enables the SARS-type coronavirus to enter cells through the enzymatic domain of ACE2.[30] Producing it following vaccination will prompt the immune system to attack the coronavirus through antibodies and T-cells if it later infects the body.[6]
History
2020 development
In February 2020, the Jenner Institute agreed a collaboration with the Italian company Advent Srl for the production of the first batch of a vaccine candidate for clinical trials.[31]
In March 2020,[32][33] after the Gates Foundation urged the University of Oxford to find a large company partner to get its COVID-19 vaccine to market, the university backed off from its earlier pledge to donate the rights to any drugmaker.[34] Also, the UK government encouraged the University of Oxford to work with AstraZeneca instead of Merck & Co., a US based company over fears of vaccine hoarding under the Trump administration.[35]
In June 2020, the US National Institute of Allergy and Infectious Diseases (NIAID) confirmed that the third phase of testing for potential vaccines developed by Oxford University and AstraZeneca would begin in July 2020.[36]
Clinical trials
In July 2020, AstraZeneca partnered with IQVIA to speed up US clinical trials.[37]
On 31 August 2020, AstraZeneca announced that it had begun enrolling adults for a US-funded, 30,000-subject late-stage study.[38]
On 8 September 2020, AstraZeneca announced a global halt to the vaccine trial while a possible adverse reaction in a participant in the United Kingdom was investigated.[39][40][41] On 13 September, AstraZeneca and the University of Oxford resumed clinical trials in the United Kingdom after regulators concluded it was safe to do so.[42] AstraZeneca was criticised for vaccine safety after concerns from experts noting the company’s refusal to provide details about serious neurological illnesses in two participants who received the experimental vaccine in Britain.[43] While the trial resumed in the UK, Brazil, South Africa, Japan[44] and India, it remained on pause in the US till 23 October 2020[45] while the Food and Drug Administration (FDA) investigated a patient illness that triggered the clinical hold, according to the United States Department of Health and Human Services (HHS) Secretary Alex Azar.[46]
On 15 October 2020, Dr João Pedro R. Feitosa, a 28-year-old doctor from Rio de Janeiro, Brazil, who received a placebo instead of the test vaccine in a clinical trial of AZD1222, died from COVID-19 complications.[47][48][49] The Brazilian health authority Anvisa announced that the trial would continue in Brazil.[50]
Results of Phase III trial
On 23 November 2020, Oxford University and AstraZeneca announced interim results from the vaccine’s ongoing Phase III trials.[6][51] There was some criticism of the methods used in the report, which combined results of 62% and 90% from different groups of test subjects given different dosages to arrive at a 70% figure.[52][53][54] AstraZeneca said it would carry out a further multi-country trial using the lower dose which had led to a 90% claim.[55]
The full publication of the interim results from four ongoing Phase III trials on 8 December 2020 clarified these reports.[56] In the group who received the first dose of active vaccine more than 21 days earlier, there were no hospitalisations or severe disease, unlike those receiving the placebo. Serious adverse events were balanced across the active and control arms in the studies, i.e. the active vaccine did not have safety concerns. A case of transverse myelitis was reported 14 days after booster vaccination as being possibly related to vaccination, with an independent neurological committee considering the most likely diagnosis to be of an idiopathic, short segment, spinal cord demyelination. The other two cases of transverse myelitis, one in the vaccine group and the other in the control group, were considered to be unrelated to vaccination.[56]
A subsequent analysis, published on 19 February, has shown an efficacy of 76% 22 days after the first dose and increase to 81.3% when the second dose is given 12 weeks or more after the first.[57]
2021 development
In February 2021, Oxford–AstraZeneca indicated developments to adapt the vaccine to target new variants of the coronavirus,[58] with expectation of a modified vaccine being available “in a few months” as a “booster jab”.[59] A key area of concern is whether the E484K mutation could impact the immune response and, possibly, current vaccine effectiveness.[60] The E484K mutation is present in the South African (B.1.351) and Brazilian (B.1.1.28) variants, with a small number of cases of the mutation also detected in infections by the original SARS-CoV-2 virus and the UK/Kent (B.1.1.7) variant.[60]
Scottish Study
A study was carried out by universities across Scotland of the effectiveness of first dose of Pfizer–BioNTech and Oxford–AstraZeneca COVID-19 vaccines against hospital admissions in Scotland, based on a national prospective cohort study of 5.4 million people. Between 8 December 2020 to 15 February 2021, 1,137,775 patients were vaccinated in the study, 490,000 of which were with the Oxford–AstraZeneca vaccine. The first dose of the Oxford–AstraZeneca vaccine was associated with a vaccine effect of 94% for COVID-19 related hospitalisation at 28–34 days post-vaccination. Results for both vaccines combined showed a vaccine effect for prevention of COVID-19 related hospitalisation which was comparable when restricting the analysis to those aged ≥80 years (81%). The majority of the patients over the age of 65 were given the Oxford–AstraZeneca vaccine. As of 22 February 2021, the study had not been peer-reviewed.[61][62]
Approvals
On 27 November 2020, the UK government asked the Medicines and Healthcare products Regulatory Agency to assess the AZD1222 vaccine for temporary supply,[63] and it was approved for use on 30 December 2020, as their second vaccine to enter the national rollout.[64]
On 4 January 2021, Brian Pinker, 82, became the first person to receive the Oxford–AstraZeneca COVID-19 vaccine outside of clinical trials.[26]
The European Medicines Agency (EMA) received an application for a conditional marketing authorisation (CMA) for the vaccine on 12 January 2021. A press release stated that a recommendation on this could be issued by the agency by 29 January, with the European Commission then making a decision on the CMA within days.[3] The Hungarian regulator unilaterally approved the vaccine instead of waiting for EMA approval.[65]
On 29 January 2021, the EMA recommended granting a conditional marketing authorisation for AZD1222 for people 18 years of age and older,[12][13] and the recommendation was accepted by the European Commission the same day.[14][66]
On 30 January 2021, the Vietnamese Ministry of Health approved the AstraZeneca vaccine for domestic inoculation, the first to be approved in Vietnam.[67]
The vaccine has also been approved by Argentina,[68] Bangladesh,[69] Brazil,[70] the Dominican Republic,[71] El Salvador,[72] India,[73][74] Malaysia,[75] Mexico,[76] Nepal,[77] Pakistan,[78] the Philippines,[79] Sri Lanka,[80] and Taiwan[81] regulatory authorities for emergency usage in their respective countries.
On 7 February 2021, the vaccine roll out in South Africa was suspended. Researchers from the University of the Witwatersrand said in a prior-to-peer analysis that the AstraZeneca vaccine provided minimal protection against mild or moderate disease infection among young people.[82][83] The BBC reported on 8 February 2021 that Katherine O’Brien, director of immunisation at the World Health Organization, indicated she felt it was “really plausible” the AstraZeneca vaccine could have a “meaningful impact” on the South African variant particularly in preventing serious illness and death.[84] The same report also indicated the Deputy Chief Medical Officer for England Jonathan Van-Tam said the (Witwatersrand) study did not change his opinion that the AstraZeneca vaccine was “rather likely” to have an effect on severe disease from the South African variant.[84]
On 10 February 2021, South Korea granted its first approval of a COVID-19 vaccine to AstraZeneca, allowing the two-shot regimen to be administered to all adults, including the elderly. The approval came with a warning, however, that consideration is needed when administering the vaccine to individuals over 65 years of age due to limited data from that demographic in clinical trials.[85][86]
On 10 February 2021, the World Health Organization (WHO) issued interim guidance and recommended the AstraZeneca vaccine for all adults, its Strategic Advisory Group of Experts also having considered use where variants were present and concluded there was no need not to recommend it.[87]
On 16 February 2021, the Australian Therapeutic Goods Administration (TGA) granted provisional approval for COVID-19 Vaccine AstraZeneca.[9][1]
On 26 February 2021, the vaccine was authorized with terms and conditions by Health Canada.[88]
Production and supply
The vaccine is stable at refrigerator temperatures and costs around US$3 to US$4 per dose.[89] On 17 December, a tweet by the Belgian Budget State Secretary revealed the European Union (EU) would pay €1.78 (US$2.16) per dose.[90]
According to AstraZeneca’s vice-president for operations and IT, Pam Cheng, the company would have around 200 million doses ready worldwide by the end of 2020, and capacity to produce 100 million to 200 million doses per month once production is ramped up.[52]
In June 2020, further to making 100 million doses available to the UK’s NHS for their vaccination programme,[91] AstraZeneca and Emergent BioSolutions signed a US$87 million deal to manufacture doses of the vaccine specifically for the US market. The deal was part of the Trump administration’s Operation Warp Speed initiative to develop and rapidly scale production of targeted vaccines before the end of 2020.[92] Catalent will be responsible for the finishing and packaging process.[93] The majority of manufacturing work will be done in the UK.[citation needed]
On 4 June 2020, the World Health Organization‘s (WHO) COVAX facility made initial purchases of 300 million doses from the company for low- to middle-income countries.[94] Also, AstraZeneca and Serum Institute of India reached a licensing agreement to supply 1 billion doses of the Oxford University vaccine to middle- and low-income countries, including India.[95][96]
On 29 September 2020, a grant from the Bill and Melinda Gates Foundation allowed COVAX to secure an additional 100 million COVID-19 vaccine doses either from AstraZeneca or from Novavax at US$3 per dose.[97]
On 13 June 2020, AstraZeneca signed a contract with the Inclusive Vaccines Alliance, a group formed by France, Germany, Italy, and the Netherlands, to supply up to 400 million doses to all European Union member states.[98][99][100] However, the European Commission intervened to stop the deal being formalised. It took over negotiations on behalf of the whole EU, signing a deal at the end of August.[101]
In August 2020, AstraZeneca agreed to provide 300 million doses to the USA for US$1.2 billion, implying a cost of US$4 per dose. An AstraZeneca spokesman said the funding also covers development and clinical testing.[102] It also reached technology transfer agreement with Mexican and Argentinean governments and agreed to produce at least 400 million doses to be distributed throughout Latin America. The active ingredients would be produced in Argentina and sent to Mexico to be completed for distribution.[103]
In September 2020, AstraZeneca agreed to provide 20 million doses to Canada.[104][105]
In October 2020, Switzerland signed an agreement with AstraZeneca to pre-order up to 5.3 million doses.[106][107]
On 5 November 2020, a tripartite agreement was signed between the government of Bangladesh, Serum Institute of India and Beximco Pharma of Bangladesh. Under the agreement Bangladesh ordered 30 million doses of Oxford–AstraZeneca vaccine from Serum through Beximco for $4 per shot.[108]
In November 2020, Thailand ordered 26 million doses of vaccine from AstraZeneca.[109] It would cover 13 million people,[110] approximately 20% of the population, with the first lot expected to be delivered at the end of May.[111][112][113] The public health minister indicated the price paid was $5 per dose;[114] AstraZeneca (Thailand) explained in January 2021 after a controversy that the price each country paid depended on production cost and differences in supply chain, including manufacturing capacity, labour and raw material costs.[115] In January 2021, the Thai cabinet approved further talks on ordering another 35 million doses[116] and the Thai FDA approved the vaccine for emergency use for 1 year.[117][118] Siam Bioscience, a company owned by Vajiralongkorn, will received technological transfer,[119] and has the capacity to manufacture up to 200 million doses a year for export to ASEAN.[120]
Also in November, the Philippines agreed to buy 2.6 million doses,[121] reportedly worth around ₱700 million (approximately $5.6/dose).[122]
In December 2020, South Korea signed a contract with AstraZeneca to secure 20 million doses of its vaccine, reportedly worth equivalently to those signed by Thailand and the Philippines,[123] with the first shipment expected as early as January 2021. As of January 2021, the vaccine remains under review by the South Korea Disease Control and Prevention Agency.[124][125] AstraZeneca signed a deal with South Korea’s SK Bioscience to manufacture its vaccine products. The collaboration calls for the SK affiliate to manufacture AZD1222 for local and global markets.[126]
On 7 January 2021, the South African government announced that they had secured an initial 1 million doses from the Serum Institute of India, to be followed by another 500,000 doses in February.[127]
Myanmar signed a contract with Serum Institute of India to secure 30 million doses of its vaccine in December 2020. Myanmar will get doses for 15 million people from February 2021.[128]
On 22 January 2021, AstraZeneca announced that in the event the European Union approved the COVID-19 Vaccine AstraZeneca, initial supplies would be lower than expected due to production issues at Novasep in Belgium. Only 31 million of the previously predicted 80 million doses would be delivered to the European Union by March 2021.[129] In an interview with Italian newspaper La Repubblica, AstraZeneca’s CEO Pascal Soriot said the delivery schedule for the doses in the European Union was two months behind schedule. He mentioned low yield from cell cultures in one large-scale European site.[130] Analysis published in The Guardian also identified an apparently low yield from bioreactors in the Belgium plant and noted the difficulties in setting up this form of process, with variable yields often occurring.[131] As a result, the European Union imposed export controls on vaccine doses; controversy erupted as to whether doses were being diverted to the UK, and whether or not deliveries to Northern Ireland would be disrupted.[132]
On 24 February 2021, Ghana became the first country in Africa to receive the Covid-19 vaccine through the COVAX initiative, where the facility sent six hundred thousand doses of AstraZeneca/Oxford jabs to Accra.[133]
Summary
Background
A safe and efficacious vaccine against severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), if deployed with high coverage, could contribute to the control of the COVID-19 pandemic. We evaluated the safety and efficacy of the ChAdOx1 nCoV-19 vaccine in a pooled interim analysis of four trials.
Methods
This analysis includes data from four ongoing blinded, randomised, controlled trials done across the UK, Brazil, and South Africa. Participants aged 18 years and older were randomly assigned (1:1) to ChAdOx1 nCoV-19 vaccine or control (meningococcal group A, C, W, and Y conjugate vaccine or saline). Participants in the ChAdOx1 nCoV-19 group received two doses containing 5 × 1010 viral particles (standard dose; SD/SD cohort); a subset in the UK trial received a half dose as their first dose (low dose) and a standard dose as their second dose (LD/SD cohort). The primary efficacy analysis included symptomatic COVID-19 in seronegative participants with a nucleic acid amplification test-positive swab more than 14 days after a second dose of vaccine. Participants were analysed according to treatment received, with data cutoff on Nov 4, 2020. Vaccine efficacy was calculated as 1 - relative risk derived from a robust Poisson regression model adjusted for age. Studies are registered at ISRCTN89951424 and ClinicalTrials.gov, NCT04324606, NCT04400838, and NCT04444674.
Findings
Between April 23 and Nov 4, 2020, 23 848 participants were enrolled and 11 636 participants (7548 in the UK, 4088 in Brazil) were included in the interim primary efficacy analysis. In participants who received two standard doses, vaccine efficacy was 62·1% (95% CI 41·0–75·7; 27 [0·6%] of 4440 in the ChAdOx1 nCoV-19 group vs71 [1·6%] of 4455 in the control group) and in participants who received a low dose followed by a standard dose, efficacy was 90·0% (67·4–97·0; three [0·2%] of 1367 vs 30 [2·2%] of 1374; pinteraction=0·010). Overall vaccine efficacy across both groups was 70·4% (95·8% CI 54·8–80·6; 30 [0·5%] of 5807 vs 101 [1·7%] of 5829). From 21 days after the first dose, there were ten cases hospitalised for COVID-19, all in the control arm; two were classified as severe COVID-19, including one death. There were 74 341 person-months of safety follow-up (median 3·4 months, IQR 1·3–4·8): 175 severe adverse events occurred in 168 participants, 84 events in the ChAdOx1 nCoV-19 group and 91 in the control group. Three events were classified as possibly related to a vaccine: one in the ChAdOx1 nCoV-19 group, one in the control group, and one in a participant who remains masked to group allocation.
Interpretation
ChAdOx1 nCoV-19 has an acceptable safety profile and has been found to be efficacious against symptomatic COVID-19 in this interim analysis of ongoing clinical trials.
Funding
UK Research and Innovation, National Institutes for Health Research (NIHR), Coalition for Epidemic Preparedness Innovations, Bill & Melinda Gates Foundation, Lemann Foundation, Rede D’Or, Brava and Telles Foundation, NIHR Oxford Biomedical Research Centre, Thames Valley and South Midland’s NIHR Clinical Research Network, and AstraZeneca.
References
- ^ Jump up to:a b c “COVID-19 Vaccine AstraZeneca”. Therapeutic Goods Administration (TGA). 16 February 2021. Retrieved 16 February2021.
- ^ Jump up to:a b “Information for Healthcare Professionals on COVID-19 Vaccine AstraZeneca”. Medicines and Healthcare products Regulatory Agency (MHRA). 30 December 2020. Retrieved 4 January 2021.
- ^ Jump up to:a b “EMA receives application for conditional marketing authorisation of COVID-19 Vaccine AstraZeneca”. European Medicines Agency (EMA). 12 January 2021. Retrieved 12 January2021.
- ^ Jump up to:a b “Regulatory Decision Summary – AstraZeneca COVID-19 Vaccine”. Health Canada. 26 February 2021. Retrieved 26 February 2021.
- ^ Jump up to:a b “Already produced 40–50 million dosages of Covishield vaccine, says Serum Institute”. The Hindu. 28 December 2020.
- ^ Jump up to:a b c d e “AZD1222 vaccine met primary efficacy endpoint in preventing COVID-19”. Press Release (Press release). AstraZeneca. 23 November 2020. Retrieved 5 January 2021.
- ^ Jump up to:a b “AstraZeneca COVID-19 Vaccine (AZD1222)” (PDF). AstraZeneca. 27 January 2021.
- ^ “AstraZeneca and Oxford University announce landmark agreement for COVID-19 vaccine”. AstraZeneca (Press release). 30 April 2020. Retrieved 13 January 2021.
- ^ Jump up to:a b c d e “COVID-19 Vaccine AstraZeneca PI”. Therapeutic Goods Administration (TGA).
- ^ “AstraZeneca COVID-19 Vaccine monograph” (PDF). AstraZeneca. 26 February 2021.
- ^ Jump up to:a b “Conditions of Authorisation for COVID-19 Vaccine AstraZeneca”. Medicines and Healthcare products Regulatory Agency (MHRA). 30 December 2020. Retrieved 4 January 2021.
- ^ Jump up to:a b c “COVID-19 Vaccine AstraZeneca EPAR”. European Medicines Agency (EMA).
- ^ Jump up to:a b “EMA recommends COVID-19 Vaccine AstraZeneca for authorisation in the EU”. European Medicines Agency (EMA)(Press release). 29 January 2021. Retrieved 29 January 2021.
- ^ Jump up to:a b c “European Commission authorises third safe and effective vaccine against COVID-19”. European Commission (Press release). Retrieved 29 January 2021.
- ^ “아스트라제네카社 코로나19 백신 품목허가”. 식품의약품안전처(in Korean). 식품의약품안전처. 10 February 2021. Retrieved 10 February 2021.
- ^ “BPOM Terbitkan Izin Penggunaan Darurat Vaksin Covid-19 AstraZeneca”. Kompas.com. 10 March 2021. Retrieved 10 March2021.
- ^ “150,000 doses of AstraZeneca vaccine arrive in Serbia”. Government of Serbia. 21 February 2021. Retrieved 2 March 2021.
- ^ Walsh N, Shelley J, Duwe E, Bonnett W (27 July 2020). “The world’s hopes for a coronavirus vaccine may run in these health care workers’ veins”. São Paulo: CNN. Archived from the original on 3 August 2020. Retrieved 3 August 2020.
- ^ “Investigating a Vaccine Against COVID-19”. ClinicalTrials.gov(Registry). United States National Library of Medicine. 26 May 2020. NCT04400838. Archived from the original on 11 October 2020. Retrieved 14 July 2020.
- ^ “A Phase 2/3 study to determine the efficacy, safety and immunogenicity of the candidate Coronavirus Disease (COVID-19) vaccine ChAdOx1 nCoV-19”. EU Clinical Trials Register(Registry). European Union. 21 April 2020. EudraCT 2020-001228-32. Archived from the original on 5 October 2020. Retrieved 3 August 2020.
- ^ O’Reilly P (26 May 2020). “A Phase III study to investigate a vaccine against COVID-19”. ISRCTN (Registry). doi:10.1186/ISRCTN89951424. ISRCTN89951424.
- ^ Jump up to:a b “Oxford team to begin novel coronavirus vaccine research”. University of Oxford. 7 February 2020. Retrieved 28 November2020.
- ^ “COVID-19 Oxford Vaccine Trial”. COVID-19 Oxford Vaccine Trial. Retrieved 11 April 2020.
- ^ “Covid-19: Oxford-AstraZeneca coronavirus vaccine approved for use in UK”. BBC News Online. 30 December 2020. Retrieved 30 December 2020.
- ^ “Second COVID-19 vaccine authorised by medicines regulator”. GOV.UK (Press release). 30 December 2020. Retrieved 6 March2021.
- ^ Jump up to:a b “Covid: Brian Pinker, 82, first to get Oxford-AstraZeneca vaccine”. BBC News Online. 4 January 2021. Retrieved 4 January 2021.
- ^ “Coronavirus disease (COVID-19): Vaccines”. World Health Organization (WHO). Retrieved 6 March 2021.
- ^ Arashkia A, Jalilvand S, Mohajel N, Afchangi A, Azadmanesh K, Salehi-Vaziri M, et al. (2020). “Severe acute respiratory syndrome-coronavirus-2 spike (S) protein based vaccine candidates: State of the art and future prospects”. Reviews in Medical Virology. n/a(n/a): e2183. doi:10.1002/rmv.2183. PMC 7646037. PMID 33594794.
- ^ Watanabe, Y.; Mendonça, L.; Allen, E. R.; Howe, A.; Lee, M.; Allen, J. D.; Chawla, H.; Pulido, D.; Donnellan, F.; Davies, H.; Ulaszewska, M.; Belij-Rammerstorfer, S.; Morris, S.; Krebs, A. S.; Dejnirattisai, W.; Mongkolsapaya, J.; Supasa, P.; Screaton, G. R.; Green, C. M.; Lambe, T.; Zhang, P.; Gilbert, S. C.; Crispin, M. (2021), “Native-like SARS-CoV-2 spike glycoprotein expressed by ChAdOx1 nCoV-19/AZD1222 vaccine”, bioRxiv : The Preprint Server for Biology: 2021.01.15.426463, doi:10.1101/2021.01.15.426463, PMC 7836103, PMID 33501433
- ^ Wang H, Yang P, Liu K, Guo F, Zhang Y, Zhang G, Jiang C (February 2008). “SARS coronavirus entry into host cells through a novel clathrin- and caveolae-independent endocytic pathway”. Cell Research. 18 (2): 290–301. doi:10.1038/cr.2008.15. PMC 7091891. PMID 18227861.
- ^ “Oxford team to begin novel coronavirus vaccine research”. University of Oxford. Retrieved 2 January 2021.
- ^ “Covid Vaccine Front-Runner Is Months Ahead of Her Competition”. Bloomberg Businessweek. 15 July 2020.
- ^ “Bill Gates, the Virus and the Quest to Vaccinate the World”. The New York Times. 23 November 2020.
- ^ “They Pledged to Donate Rights to Their COVID Vaccine, Then Sold Them to Pharma”. Kaiser Health News. Retrieved 28 January 2021.
- ^ Strasburg J, Woo S (21 October 2020). “Oxford Developed Covid Vaccine, Then Scholars Clashed Over Money”. The Wall Street Journal.
- ^ Coleman J (10 June 2020). “Final testing stage for potential coronavirus vaccine set to begin in July”. The Hill. Retrieved 11 June 2020.
- ^ “AZN, IQV Team Up To Accelerate COVID-19 Vaccine Work, RIGL’s ITP Drug Repurposed, IMV On Watch”. RTTNews. Retrieved 15 July 2020.
- ^ “Phase 3 Clinical Testing in the US of AstraZeneca COVID-19 Vaccine Candidate Begins”. National Institutes of Health (NIH). 30 August 2020. Retrieved 1 September 2020.
- ^ “AstraZeneca Covid-19 vaccine study is put on hold”. Stat. 8 September 2020. Retrieved 10 September 2020.
- ^ “AstraZeneca Covid-19 vaccine study is put on hold”. 8 September 2020.
- ^ Wu KJ, Thomas K (8 September 2020). “AstraZeneca Pauses Vaccine Trial for Safety Review”. The New York Times. ISSN 0362-4331. Retrieved 10 September 2020.
- ^ Loftus P (13 September 2020). “AstraZeneca Covid-19 Vaccine Trials Resume in U.K.”. The Wall Street Journal. Retrieved 13 September 2020.
- ^ Grady D, Wu KJ, LaFraniere S (19 September 2020). “AstraZeneca, Under Fire for Vaccine Safety, Releases Trial Blueprints”. The New York Times. ISSN 0362-4331. Retrieved 22 September 2020.
- ^ “AstraZeneca resumes vaccine trial in talks with US”. Japan Today. 3 October 2020.
- ^ “FDA authorises restart of the COVID-19 AZD1222 vaccine US Phase III trial”. AstraZeneca (Press release). Retrieved 1 December 2020.
- ^ “U.S. health secretary says AstraZeneca trial in United States remains on hold: CNBC”. Reuters. 23 September 2020. Retrieved 24 September 2020.
- ^ “‘What’s the deal?’ Researchers in paused vaccine trial search for answers”. NBC News.
- ^ “Volunteer in AstraZeneca Covid-19 vaccine trial dies in Brazil”. NBC News.
- ^ Voluntário brasileiro que participava dos testes da vacina de Oxford e morreu com a Covid era médico e ex-aluno da UFRJ, Globo
- ^ Simões E, Burger L (22 October 2020). “AstraZeneca COVID-19 vaccine trial Brazil volunteer dies, trial to continue”. Reuters. Retrieved 22 October 2020.
- ^ “Oxford University breakthrough on global COVID-19 vaccine”(Press release). University of Oxford. 23 November 2020. Retrieved 12 January 2021.
- ^ Jump up to:a b Callaway E (23 November 2020). “Why Oxford’s positive COVID vaccine results are puzzling scientists”. Nature. 588(7836): 16–18. Bibcode:2020Natur.588…16C. doi:10.1038/d41586-020-03326-w. PMID 33230278. S2CID 227156970.
- ^ “Oxford/AstraZeneca Covid vaccine ‘dose error’ explained”. BBC News. 27 November 2020. Retrieved 27 November 2020.
- ^ Robbins R, Mueller B (25 November 2020). “After Admitting Mistake, AstraZeneca Faces Difficult Questions About Its Vaccine”. The New York Times. ISSN 0362-4331. Retrieved 27 November 2020.
- ^ Boseley S (26 November 2020). “Oxford/AstraZeneca vaccine to undergo new global trial”. The Guardian. Retrieved 27 November2020.
- ^ Jump up to:a b Voysey M, Clemens SA, Madhi SA, Weckx LY, Folegatti PM, Aley PK, et al. (January 2021). “Safety and efficacy of the ChAdOx1 nCoV-19 vaccine (AZD1222) against SARS-CoV-2: an interim analysis of four randomised controlled trials in Brazil, South Africa, and the UK”. Lancet. 397 (10269): 99–111. doi:10.1016/S0140-6736(20)32661-1. PMC 7723445. PMID 33306989.
- ^ Voysey M, Costa Clemens SA, Madhi SA, Weckx LY, Folegatti PM, Aley PK, et al. (February 2021). “Single-dose administration and the influence of the timing of the booster dose on immunogenicity and efficacy of ChAdOx1 nCoV-19 (AZD1222) vaccine: a pooled analysis of four randomised trials”. Lancet. 397(10277): 881–891. doi:10.1016/S0140-6736(21)00432-3. PMC 7894131. PMID 33617777.
- ^ Ellyatt H (8 February 2021). “AstraZeneca races to adapt Covid vaccine as South Africa suspends rollout”. CNBC. Retrieved 8 February 2021.
- ^ Triggle N (8 February 2021). “Covid: Are fears over Oxford-AstraZeneca jab justified?”. BBC. Retrieved 9 February 2021.
- ^ Jump up to:a b Wise J (February 2021). “Covid-19: The E484K mutation and the risks it poses”. BMJ. 372: n359. doi:10.1136/bmj.n359. PMID 33547053.
- ^ “Covid-19: First doses of vaccines in Scotland led to a substantial fall in hospital admissions”. the BMJ. Retrieved 25 February2021.
- ^ “scotland first vaccine data preprint” (PDF). Retrieved 25 February 2021.
- ^ “Government asks regulator to approve supply of Oxford/AstraZeneca vaccine”. Government of the United Kingdom. 27 October 2020. Retrieved 28 November 2020.
- ^ “Oxford University/AstraZeneca vaccine authorised by UK medicines regulator”. Government of the United Kingdom. 30 December 2020. Retrieved 30 December 2020.
- ^ “Everything You Need to Know About the Oxford-AstraZeneca Vaccine”. 23 January 2021.
- ^ “COVID-19 Vaccine AstraZeneca”. Union Register of medicinal products. Retrieved 18 February 2021.
- ^ Nikkei staff writers. “Coronavirus: Week of Jan. 24 to Jan. 30, Vietnam approves AstraZeneca vaccine”. Nikkei Asia.
- ^ Laing A (30 December 2020). “Argentine regulator approves AstraZeneca/Oxford COVID-19 vaccine -AstraZeneca”. Reuters.
- ^ “Oxford University-Astrazeneca vaccine: Bangladesh okays it for emergency use”. The Daily Star. 4 January 2021. Retrieved 7 January 2021.
- ^ Sabóia G, Mazieiro G, de Andrade H, Adorno L (17 January 2021). “Anvisa aprova uso emergencial das vacinas CoronaVac e AstraZeneca no Brasil” [Anvisa approves emergency use of the CoronaVac and AstraZeneca vaccines in Brazil]. UOL (in Portuguese). Retrieved 17 January 2021.
- ^ “La República Dominicana aprueba la vacuna de AstraZeneca contra la covid-19”. Agencia EFE (in Spanish). 31 December 2020.
- ^ “El Salvador greenlights AstraZeneca, Oxford University COVID-19 vaccine”. Reuters. 30 December 2020.
- ^ Gaurav K (1 January 2021). “Govt’s expert panel approves AstraZeneca/Oxford Covid-19 vaccine for emergency use”. Hindustan Times.
- ^ Prusty N, Jamkhandikar S (1 January 2021). “India drug regulator approves AstraZeneca COVID vaccine, country’s first – sources”. Reuters.
- ^ “Malaysia’s NPRA Approves AstraZeneca, Sinovac Covid-19 Vaccines”. CodeBlue. 2 March 2021. Retrieved 2 March 2021.
- ^ Comisión Federal para la Protección contra Riesgos Sanitarios. “AUTORIZACIÓN PARA USO DE EMERGENCIA A VACUNA ASTRAZENECA COVID-19”. gob.mx (in Spanish). Retrieved 7 January 2021.
- ^ “Nepal approves AstraZeneca COVID vaccine for emergency use – government statement”. Reuters. 15 January 2021.
- ^ Shahzad A (16 January 2021). “Pakistan approves AstraZeneca COVID-19 vaccine for emergency use”. Reuters. Retrieved 16 January 2021.
- ^ “Philippine regulator approves emergency use of AstraZeneca vaccine”. Reuters. 28 January 2021. Retrieved 28 January 2021.
- ^ “Sri Lanka approves vaccine amid warnings of virus spread”. AP NEWS. 22 January 2021. Retrieved 22 January 2021.
- ^ “Taiwan grants emergency authorisation for AstraZeneca COVID-19 vaccine”. MSN. Retrieved 22 February 2021.
- ^ “Latest – Oxford Covid-19 vaccine trial results – Wits University”. wits.ac.za. Retrieved 8 February 2021.
- ^ “South Africa halts AstraZeneca vaccinations after data shows little protection against mutation”. CNBC. 7 February 2021. Retrieved 8 February 2021.
- ^ Jump up to:a b “Covid: Boris Johnson ‘very confident’ in vaccines being used in UK”. BBC News. 8 February 2021. Retrieved 9 February 2021.
- ^ Kim HJ (10 February 2021). “S. Korea approves AstraZeneca’s COVID-19 vaccine for all adults”. Yonhap News Agency. Retrieved 10 February 2021.
- ^ Maresca T (10 February 2021). “South Korea approves AstraZeneca COVID-19 vaccine”. United Press International. Retrieved 10 February 2021.
- ^ “AstraZeneca-Oxford vaccine can be used for people aged over 65 – WHO”. RTÉ News and Current Affairs. 10 February 2021. Retrieved 12 February 2021.
- ^ Canada, Health. “COVID-19 vaccines and treatments portal: AstraZeneca COVID-19 Vaccine (ChAdOx1-S [recombinant])”. Health Canada. Retrieved 26 February 2021.
- ^ Belluz J (23 November 2020). “Why the AstraZeneca-Oxford Covid-19 vaccine is different”. Vox. Retrieved 26 November 2020.
- ^ Stevis-Gridneff M, Sanger-Katz M, Weiland N (18 December 2020). “A European Official Reveals a Secret: The U.S. Is Paying More for Coronavirus Vaccines”. The New York Times. Retrieved 19 December 2020.
- ^ “AstraZeneca to begin making vaccine“. BBC. 5 June 2020. Retrieved 1 July 2020.
- ^ “AstraZeneca, Emergent BioSolutions sign $87M deal to produce U.S. supply of COVID-19 vaccine”. FiercePharma. Retrieved 12 June 2020.
- ^ “AstraZeneca taps Catalent for COVID-19 vaccine finishing, packaging at Italian plant”. FiercePharma. Retrieved 16 June2020.
- ^ So AD, Woo J (December 2020). “Reserving coronavirus disease 2019 vaccines for global access: cross sectional analysis”. BMJ. 371: m4750. doi:10.1136/bmj.m4750. PMC 7735431. PMID 33323376. cited “Agreements with CEPI and Gavi and the Serum Institute of India will bring vaccine to low and middle-income countries and beyond” (Press release). AstraZeneca. 4 June 2020.
- ^ Rajagopal D (4 June 2020). “AstraZeneca & Serum Institute of India sign licensing deal for 1 billion doses of Oxford vaccine”. The Economic Times.
- ^ Kumar M (7 August 2020). “Covid-19 vaccine: Serum Institute signs up for 100 million doses of vaccines for India, low and middle-income countries”. The Financial Express.
- ^ So & Woo (2020), p. 3 cited “New collaboration makes further 100 million doses of COVID-19 vaccine available to low- and middle- income countries” (Press release). Gavi, the Vaccine Alliance. 29 September 2020.[permanent dead link]
- ^ “Covid-19: France, Italy, Germany and Netherlands sign vaccine deal for Europe”. France 24. 13 June 2020. Retrieved 15 June2020.
- ^ “AstraZeneca agrees to supply Europe with 400 mil doses of COVID-19 vaccine”. Japan Today. Retrieved 15 June 2020.
- ^ Calatayud A. “AstraZeneca to supply Europe with Covid-19 vaccine”. MarketWatch. Retrieved 15 June 2020.
- ^ Peston R (26 January 2021). “What is the dispute between the EU and AstraZeneca over Covid jabs?”. ITV News. Retrieved 27 January 2021.
- ^ Roland D (21 May 2020). “U.S. to Invest $1.2 Billion to Secure Potential Coronavirus Vaccine From AstraZeneca, Oxford University”. The Wall Street Journal. Retrieved 6 August 2020.
- ^ “AstraZeneca set to start making 400 million COVID-19 vaccines for Latam early in 2021”. Reuters. Retrieved 17 January 2021.
- ^ “With no successful vaccine candidates yet, Canada signs deal to secure 20M more COVID-19 vaccine doses”. CBC News. 25 September 2020.
- ^ Health Canada (2 October 2020). “Health Canada begins first authorization review of a COVID-19 vaccine submission”. gcnws. Retrieved 30 December 2020.
- ^ “Swiss sign next vaccine agreement with AstraZeneca”. SWI swissinfo.ch. Retrieved 16 October 2020.
- ^ “COVID-19 vaccine: Swiss federal government signs agreement with AstraZeneca”. admin.ch. Retrieved 16 October 2020.
- ^ “Dhaka to have 330 vaccination points”. The Daily Star. Retrieved 25 January 2021.
- ^ “เรื่องน่ารู้ของวัคซีนโควิด-19 ที่ไทยสั่งซื้อ”. BBC ไทย (in Thai). Retrieved 5 January 2021.
- ^ “ทำความรู้จัก ออกซ์ฟอร์ด-แอสทราเซเนกา วัคซีนที่ไทยเลือก”. มติชนออนไลน์ (in Thai). 2 January 2021. Retrieved 5 January 2021.
- ^ “ครม.ไฟเขียวงบซื้อวัคซีนโควิดเพิ่ม35ล้านโดส ฉีดให้คนไทย66ล้าน”. โพสต์ทูเดย์ (in Thai). Retrieved 5 January 2021.
- ^ “ข่าวดี ไทยเริ่มผลิตวัคซีน ‘โควิด-19’ ในประเทศ รอบที่ 2 แล้ว”. ไทยรัฐออนไลน์. 3 January 2021.
- ^ “สธ. แจง AstraZeneca เป็นผู้คัดเลือก Siam Bioscience ผลิตวัคซีนราคาทุน ขายถูกสุดในตลาด โต้ธนาธร ไม่ได้แทงม้าตัวเดียว”. THE STANDARD. 19 January 2021.
- ^ “ข่าวดี! ไทยจองซื้อวัคซีนโควิด-19 แอสตราเซเนกา “ราคาต้นทุน”” (in Thai). hfocus.org. 23 November 2020. Archived from the original on 23 November 2020.
- ^ “วัคซีนโควิด: แอสตร้าเซเนก้าชี้แจงเหตุผลเลือกสยามไบโอไซเอนซ์เป็นผู้ผลิต”. BBC News ไทย. 26 January 2021.
- ^ “โควิด-19: ทำไมรัฐบาลเลือก สยามไบโอไซเอนซ์ ผลิตวัคซีนเพื่อคนไทยและเพื่อนบ้าน”. BBC News ไทย. 15 January 2021.
- ^ “AstraZeneca vaccine approved, 50,000 doses due in February”. Bangkok Post. 21 January 2021.
- ^ “FDA approves AstraZeneca”. Bangkok Post. 22 January 2021.
- ^ “นายกฯ สำนึกในพระมหากรุณาธิคุณ ร.10 ทรงให้ “สยามไบโอไซเอนซ์” รองรับวัคซีนโควิด-19″. BBC ไทย (in Thai). 27 November 2020. Retrieved 5 January 2021.
- ^ “35m more shots to be bought in 2021”. Bangkok Post. 5 January 2021.
- ^ “Philippines, AstraZeneca Sign Deal for 2.6 Million Doses”. Bloomberg. 27 November 2020.
- ^ “Over 200 firms to ink deal for more COVID vaccines with gov’t, AstraZeneca”. Philippine Daily Inquirer. 11 January 2021.
- ^ “Korea signs agreement with AstraZeneca for COVID vaccine”. The Korea Times. 30 November 2020.
- ^ Shin H (3 December 2020). “South Korea reaches deal to buy AstraZeneca’s COVID-19 vaccine candidate: media”. Reuters. Retrieved 5 January 2021.
- ^ Cha S (4 January 2021). “S.Korea reviews AstraZeneca COVID-19 vaccine, expands ban on gatherings”. Reuters. Retrieved 5 January 2021.
- ^ Kim YC (30 November 2020). “Korea signs agreement with AstraZeneca for COVID vaccine”. The Korea Times. Retrieved 30 January 2021.
- ^ Felix J (7 January 2021). “SA will get 1 million doses of Covid-19 vaccine from India this month”. News24.com. Retrieved 7 January 2021.
- ^ “Myanmar will get doses for 15 million people this February”. 7day.news. Retrieved 8 January 2021.
- ^ Agencies (22 January 2021). “Covid: Oxford/AstraZeneca vaccine delivery to EU to be cut by 60%”. The Guardian. Retrieved 23 January 2021.
- ^ “Pascal Soriot: “There are a lot of emotions on vaccines in EU. But it’s complicated””. la Repubblica (in Italian). 26 January 2021. Retrieved 27 January 2021.
- ^ Boseley S (26 January 2021). “Why has AstraZeneca reduced promised vaccine supply to EU and is UK affected?”. The Guardian. Retrieved 27 January 2021.
- ^ “EU tightens vaccine export rules, creates post-Brexit outcry”. 30 January 2021.
- ^ “Ghana receives first historic shipment of COVID-19 vaccinations from international COVAX facility”. UN News. Retrieved 24 February 2021.
External links
| Scholia has a profile for AZD1222 (Q95042269). |
- “Medical Information site for COVID-19 Vaccine AstraZeneca”. AstraZeneca.
- “Vaccines: contract between European Commission and AstraZeneca now published”. European Commission.
- “How the Oxford-AstraZeneca Covid-19 Vaccine Works”. The New York Times.
- Background document on the AZD1222 vaccine against COVID-19 developed by Oxford University and AstraZeneca. World Health Organization (WHO) (Report).
- Australian Public Assessment Report for ChAdOx1-S (PDF) (Report).
| Box containing 100 AstraZeneca COVID-19 vaccine doses | |
| Vaccine description | |
|---|---|
| Target | SARS-CoV-2 |
| Clinical data | |
| Trade names | COVID-19 Vaccine AstraZeneca,[1][2][3] AstraZeneca COVID-19 Vaccine,[4] Covishield[5] |
| Other names | AZD1222,[6][7] ChAdOx1 nCoV-19,[8] ChAdOx1-S,[9] |
| License data | EU EMA: by INN |
| Pregnancy category | AU: B2[9][1] |
| Routes of administration | Intramuscular |
| ATC code | None |
| Legal status | |
| Legal status | AU: S4 (Prescription only) [9]CA: Schedule D; Authorized by interim order [4][10]UK: Conditional and temporary authorisation to supply [2][11]EU: Conditional marketing authorisation [12][13][14]KR – Approved[15]IND, INA[16], BD, AG, SV, DOM, MEX, NE, BR, SL, SRB[17]: Emergency Authorization only |
| Identifiers | |
| CAS Number | 2420395-83-9 |
| DrugBank | DB15656 |
| UNII | B5S3K2V0G8 |
////////AZD1222, ChAdOx1, Oxford–AstraZeneca, COVID 19 vaccine, COVISHIELD, CORONA, COVID 19, CORONA VIRUS
#AZD1222, #ChAdOx1, #Oxford–AstraZeneca, #COVID 19 vaccine, #COVISHIELD, #CORONA, #COVID 19, #CORONA VIRUS
Tozinameran, Pfizer–BioNTech COVID‑19 vaccine


SEQUENCE1
gagaauaaac uaguauucuu cuggucccca cagacucaga gagaacccgc51caccauguuc guguuccugg ugcugcugcc ucuggugucc agccagugug101ugaaccugac caccagaaca cagcugccuc cagccuacac caacagcuuu151accagaggcg uguacuaccc cgacaaggug uucagaucca gcgugcugca201cucuacccag gaccuguucc ugccuuucuu cagcaacgug accugguucc251acgccaucca cguguccggc accaauggca ccaagagauu cgacaacccc301gugcugcccu ucaacgacgg gguguacuuu gccagcaccg agaaguccaa351caucaucaga ggcuggaucu ucggcaccac acuggacagc aagacccaga401gccugcugau cgugaacaac gccaccaacg uggucaucaa agugugcgag451uuccaguucu gcaacgaccc cuuccugggc gucuacuacc acaagaacaa501caagagcugg auggaaagcg aguuccgggu guacagcagc gccaacaacu551gcaccuucga guacgugucc cagccuuucc ugauggaccu ggaaggcaag601cagggcaacu ucaagaaccu gcgcgaguuc guguuuaaga acaucgacgg651cuacuucaag aucuacagca agcacacccc uaucaaccuc gugcgggauc701ugccucaggg cuucucugcu cuggaacccc ugguggaucu gcccaucggc751aucaacauca cccgguuuca gacacugcug gcccugcaca gaagcuaccu801gacaccuggc gauagcagca gcggauggac agcuggugcc gccgcuuacu851augugggcua ccugcagccu agaaccuucc ugcugaagua caacgagaac901ggcaccauca ccgacgccgu ggauugugcu cuggauccuc ugagcgagac951aaagugcacc cugaaguccu ucaccgugga aaagggcauc uaccagacca1001gcaacuuccg ggugcagccc accgaaucca ucgugcgguu ccccaauauc1051accaaucugu gccccuucgg cgagguguuc aaugccacca gauucgccuc1101uguguacgcc uggaaccgga agcggaucag caauugcgug gccgacuacu1151ccgugcugua caacuccgcc agcuucagca ccuucaagug cuacggcgug1201uccccuacca agcugaacga ccugugcuuc acaaacgugu acgccgacag1251cuucgugauc cggggagaug aagugcggca gauugccccu ggacagacag1301gcaagaucgc cgacuacaac uacaagcugc ccgacgacuu caccggcugu1351gugauugccu ggaacagcaa caaccuggac uccaaagucg gcggcaacua1401caauuaccug uaccggcugu uccggaaguc caaucugaag cccuucgagc1451gggacaucuc caccgagauc uaucaggccg gcagcacccc uuguaacggc1501guggaaggcu ucaacugcua cuucccacug caguccuacg gcuuucagcc1551cacaaauggc gugggcuauc agcccuacag agugguggug cugagcuucg1601aacugcugca ugccccugcc acagugugcg gcccuaagaa aagcaccaau1651cucgugaaga acaaaugcgu gaacuucaac uucaacggcc ugaccggcac1701cggcgugcug acagagagca acaagaaguu ccugccauuc cagcaguuug1751gccgggauau cgccgauacc acagacgccg uuagagaucc ccagacacug1801gaaauccugg acaucacccc uugcagcuuc ggcggagugu cugugaucac1851cccuggcacc aacaccagca aucagguggc agugcuguac caggacguga1901acuguaccga agugcccgug gccauucacg ccgaucagcu gacaccuaca1951uggcgggugu acuccaccgg cagcaaugug uuucagacca gagccggcug2001ucugaucgga gccgagcacg ugaacaauag cuacgagugc gacaucccca2051ucggcgcugg aaucugcgcc agcuaccaga cacagacaaa cagcccucgg2101agagccagaa gcguggccag ccagagcauc auugccuaca caaugucucu2151gggcgccgag aacagcgugg ccuacuccaa caacucuauc gcuaucccca2201ccaacuucac caucagcgug accacagaga uccugccugu guccaugacc2251aagaccagcg uggacugcac cauguacauc ugcggcgauu ccaccgagug2301cuccaaccug cugcugcagu acggcagcuu cugcacccag cugaauagag2351cccugacagg gaucgccgug gaacaggaca agaacaccca agagguguuc2401gcccaaguga agcagaucua caagaccccu ccuaucaagg acuucggcgg2451cuucaauuuc agccagauuc ugcccgaucc uagcaagccc agcaagcgga2501gcuucaucga ggaccugcug uucaacaaag ugacacuggc cgacgccggc2551uucaucaagc aguauggcga uugucugggc gacauugccg ccagggaucu2601gauuugcgcc cagaaguuua acggacugac agugcugccu ccucugcuga2651ccgaugagau gaucgcccag uacacaucug cccugcuggc cggcacaauc2701acaagcggcu ggacauuugg agcaggcgcc gcucugcaga uccccuuugc2751uaugcagaug gccuaccggu ucaacggcau cggagugacc cagaaugugc2801uguacgagaa ccagaagcug aucgccaacc aguucaacag cgccaucggc2851aagauccagg acagccugag cagcacagca agcgcccugg gaaagcugca2901ggacgugguc aaccagaaug cccaggcacu gaacacccug gucaagcagc2951uguccuccaa cuucggcgcc aucagcucug ugcugaacga uauccugagc3001agacuggacc cuccugaggc cgaggugcag aucgacagac ugaucacagg3051cagacugcag agccuccaga cauacgugac ccagcagcug aucagagccg3101ccgagauuag agccucugcc aaucuggccg ccaccaagau gucugagugu3151gugcugggcc agagcaagag aguggacuuu ugcggcaagg gcuaccaccu3201gaugagcuuc ccucagucug ccccucacgg cgugguguuu cugcacguga3251cauaugugcc cgcucaagag aagaauuuca ccaccgcucc agccaucugc3301cacgacggca aagcccacuu uccuagagaa ggcguguucg uguccaacgg3351cacccauugg uucgugacac agcggaacuu cuacgagccc cagaucauca3401ccaccgacaa caccuucgug ucuggcaacu gcgacgucgu gaucggcauu3451gugaacaaua ccguguacga cccucugcag cccgagcugg acagcuucaa3501agaggaacug gacaaguacu uuaagaacca cacaagcccc gacguggacc3551ugggcgauau cagcggaauc aaugccagcg ucgugaacau ccagaaagag3601aucgaccggc ugaacgaggu ggccaagaau cugaacgaga gccugaucga3651ccugcaagaa cuggggaagu acgagcagua caucaagugg cccugguaca3701ucuggcuggg cuuuaucgcc ggacugauug ccaucgugau ggucacaauc3751augcuguguu gcaugaccag cugcuguagc ugccugaagg gcuguuguag3801cuguggcagc ugcugcaagu ucgacgagga cgauucugag cccgugcuga3851agggcgugaa acugcacuac acaugaugac ucgagcuggu acugcaugca3901cgcaaugcua gcugccccuu ucccguccug gguaccccga gucucccccg3951accucggguc ccagguaugc ucccaccucc accugcccca cucaccaccu4001cugcuaguuc cagacaccuc ccaagcacgc agcaaugcag cucaaaacgc4051uuagccuagc cacaccccca cgggaaacag cagugauuaa ccuuuagcaa4101uaaacgaaag uuuaacuaag cuauacuaac cccaggguug gucaauuucg4151ugccagccac acccuggagc uagcaaaaaa aaaaaaaaaa aaaaaaaaaa4201aaaagcauau gacuaaaaaa aaaaaaaaaa aaaaaaaaaa aaaaaaaaaa4251aaaaaaaaaa aaaaaaaaaa aaaaaaaaaa aaaa
Sequence Modifications
| Type | Location | Description |
|---|---|---|
| modified base | g-1 | m7g |
| modified base | g-1 | 3′-me |
| modified base | a-2 | am |
| uncommon link | g-1 – a-2 | 5′->5′ triphosphate |
Tozinameran
Pfizer–BioNTech COVID-19 vaccine
トジナメラン (JAN);
コロナウイルス修飾ウリジンRNAワクチン;
RNA (recombinant 5′-[1,2-[(3′-O-methyl)m7G-(5’→5′)-ppp-Am]]-capped all uridine→N1-methylpseudouridine-substituted severe acute respiratory syndrome coronavirus 2 secretory signal peptide contg. spike glycoprotein S1S2-specifying plus 5′- and 3′-untranslated flanking region-contg. poly(A)-tailed messenger BNT162b2), inner salt
Nucleic Acid Sequence
Sequence Length: 42841106 a 1315 c 1062 g 801 umodified
APPROVED JAPAN Comirnaty, 2021/2/14
CAS 2417899-77-3
| Active immunization (SARS-CoV-2) |
Tozinameran is mRNA encoding full length of spike protein analog of SARS-CoV-2
Target Severe acute respiratory syndrome coronavirus 2 spike glycoprotein
Coronavirus disease – COVID-19
| FORM | ROUTE | STRENGTH |
|---|---|---|
| Injection, suspension | Intramuscular | 0.23 mg/1.8mL |
| Suspension | Intramuscular | 30 mcg |
| NAME | INGREDIENTS | DOSAGE | ROUTE | LABELLER | MARKETING START | MARKETING END | ||
|---|---|---|---|---|---|---|---|---|
| Pfizer-BioNTech Covid-19 Vaccine | Pfizer-BioNTech Covid-19 Vaccine (0.23 mg/1.8mL) | Injection, suspension | Intramuscular | Pfizer Manufacturing Belgium NV | 2020-12-12 | Not applicable |
| NAME | DOSAGE | STRENGTH | ROUTE | LABELLER | MARKETING START | MARKETING END | ||
|---|---|---|---|---|---|---|---|---|
| Comirnaty | 30 mcg | Intramuscular | Bio N Tech Manufacturing Gmb H | 2021-01-06 | Not applicable | |||
| Pfizer-BioNTech Covid-19 Vaccine | Suspension | 30 mcg | Intramuscular | Biontech Manufacturing Gmbh | 2020-12-14 | Not applicable | ||
| Pfizer-BioNTech Covid-19 Vaccine | Injection, suspension | 0.23 mg/1.8mL | Intramuscular | Pfizer Manufacturing Belgium NV | 2020-12-12 | Not applicable |
The Pfizer–BioNTech COVID‑19 vaccine (pINN: tozinameran), sold under the brand name Comirnaty,[13] is a COVID-19 vaccine developed by the German company BioNTech in cooperation with Pfizer. It is both the first COVID-19 vaccine to be authorized by a stringent regulatory authority for emergency use[14][15] and the first cleared for regular use.[16]
It is given by intramuscular injection. It is an RNA vaccine composed of nucleoside-modified mRNA (modRNA) encoding a mutated form of the spike protein of SARS-CoV-2, which is encapsulated in lipid nanoparticles.[17] The vaccination requires two doses given three weeks apart.[18][19][20] Its ability to prevent severe infection in children, pregnant women, or immunocompromised people is unknown, as is the duration of the immune effect it confers.[20][21][22] As of February 2021, it is one of two RNA vaccines being deployed against COVID‑19, the other being the Moderna COVID‑19 vaccine. A third mRNA-based COVID-19 vaccine, CVnCoV, is in late-stage testing.[23]
Trials began in April 2020; by November, the vaccine had been tested on more than 40,000 people.[24] An interim analysis of study data showed a potential efficacy of over 90% in preventing infection within seven days of a second dose.[19][20] The most common side effects include mild to moderate pain at the injection site, fatigue, and headache.[25][26] As of December 2020, reports of serious side effects, such as allergic reactions, have been very rare,[a] and no long-term complications have been reported.[28] Phase III clinical trials are ongoing: monitoring of the primary outcomes will continue until August 2021, while monitoring of the secondary outcomes will continue until January 2023.[18]
In December 2020, the United Kingdom was the first country to authorize the vaccine on an emergency basis,[28] soon followed by the United States, the European Union and several other countries globally.[29][30][6][31][32]
BioNTech is the initial developer of the vaccine, and partnered with Pfizer for development, clinical research, overseeing the clinical trials, logistics, finances and for manufacturing worldwide with the exception of China.[33] The license to distribute and manufacture in China was purchased by Fosun, alongside its investment in BioNTech.[34][35] Distribution in Germany and Turkey is by BioNTech itself.[36] Pfizer indicated in November 2020, that 50 million doses could be available globally by the end of 2020, with about 1.3 billion doses in 2021.[20]
Pfizer has advanced purchase agreements of about US$3 billion for providing a licensed vaccine in the United States, the European Union, the United Kingdom, Japan, Canada, Peru, Singapore, and Mexico.[37][38] Distribution and storage of the vaccine is a logistics challenge because it needs to be stored at temperatures between −80 and −60 °C (−112 and −76 °F),[39] until five days before vaccination[38][39] when it can be stored at 2 to 8 °C (36 to 46 °F), and up to two hours at temperatures up to 25 °C (77 °F)[40][11] or 30 °C (86 °F).[41][42] In February 2021, Pfizer and BioNTech asked the U.S. Food and Drug Administration (FDA) to update the emergency use authorization (EUA) to permit the vaccine to be stored at between −25 and −15 °C (−13 and 5 °F) for up to two weeks before use.[43]
Development and funding
Before COVID-19 vaccines, a vaccine for an infectious disease had never before been produced in less than several years, and no vaccine existed for preventing a coronavirus infection in humans.[44] After the COVID-19 virus was detected in December 2019,[45] the development of BNT162b2 was initiated on 10 January 2020, when the SARS-CoV-2 genetic sequences were released by the Chinese Center for Disease Control and Prevention via GISAID,[46][47][48] triggering an urgent international response to prepare for an outbreak and hasten development of preventive vaccines.[49][50]
In January 2020, German biotech-company BioNTech started its program ‘Project Lightspeed’ to develop a vaccine against the new COVID‑19 virus based on its already established mRNA-technology.[24] Several variants of the vaccine were created in their laboratories in Mainz, and 20 of those were presented to experts of the Paul-Ehrlich-Institute in Langen.[51] Phase I / II Trials were started in Germany on 23 April 2020, and in the U.S. on 4 May 2020, with four vaccine candidates entering clinical testing. The Initial Pivotal Phase II / III Trial with the lead vaccine candidate ‘BNT162b2’ began in July. The Phase III results indicating a 95% effectiveness of the developed vaccine were published on 18 November 2020.[24]
BioNTech received a US$135 million investment from Fosun in March 2020, in exchange for 1.58 million shares in BioNTech and the future development and marketing rights of BNT162b2 in China,[35] Hong Kong, Macau and Taiwan.[52]
In June 2020, BioNTech received €100 million (US$119 million) in financing from the European Commission and European Investment Bank.[53] In September 2020, the German government granted BioNTech €375 million (US$445 million) for its COVID‑19 vaccine development program.[54]
Pfizer CEO Albert Bourla stated that he decided against taking funding from the US government’s Operation Warp Speed for the development of the vaccine “because I wanted to liberate our scientists [from] any bureaucracy that comes with having to give reports and agree how we are going to spend the money in parallel or together, etc.” Pfizer did enter into an agreement with the US for the eventual distribution of the vaccine, as with other countries.[55]
Clinical trials
See also: COVID-19 vaccine § Clinical trials started in 2020
Preliminary results from Phase I–II clinical trials on BNT162b2, published in October 2020, indicated potential for its efficacy and safety.[17][56] During the same month, the European Medicines Agency (EMA) began a periodic review of BNT162b2.[57]
The study of BNT162b2 is a continuous-phase trial in Phase III as of November 2020.[18] It is a “randomized, placebo-controlled, observer-blind, dose-finding, vaccine candidate-selection, and efficacy study in healthy individuals”.[18] The early-stage research determined the safety and dose level for two vaccine candidates, with the trial expanding during mid-2020 to assess efficacy and safety of BNT162b2 in greater numbers of participants, reaching tens of thousands of people receiving test vaccinations in multiple countries in collaboration with Pfizer and Fosun.[20][35]
The Phase III trial assesses the safety, efficacy, tolerability, and immunogenicity of BNT162b2 at a mid-dose level (two injections separated by 21 days) in three age groups: 12–15 years, 16–55 years or above 55 years.[18] For approval in the EU, an overall vaccine efficacy of 95% was confirmed by the EMA.[58] The EMA clarified that the second dose should be administered three weeks after the first dose.[59]
| Efficacy endpoint | Vaccine efficacy (95% confidence interval) [%] |
|---|---|
| After dose 1 to before dose 2 | 52.4 (29.5, 68.4) |
| ≥10 days after dose 1 to before dose 2 | 86.7 (68.6, 95.4) |
| Dose 2 to 7 days after dose 2 | 90.5 (61.0, 98.9) |
| ≥7 days after dose 2 (subjects without evidence of infection prior to 7 days after dose 2) | |
| Overall | 95.0 (90.0, 97.9) |
| 16–55 years | 95.6 (89.4, 98.6) |
| ≥55 years | 93.7 (80.6, 98.8) |
| ≥65 years | 94.7 (66.7, 99.9) |
The ongoing Phase III trial, which is scheduled to run from 2020 to 2022, is designed to assess the ability of BNT162b2 to prevent severe infection, as well as the duration of immune effect.[20][21][22]
Pfizer and BioNTech started a Phase II/III randomized control trial in healthy pregnant women 18 years of age and older (NCT04754594).[60] The study will evaluate 30 µg of BNT162b2 or placebo administered via intramuscular injection in 2 doses, 21 days apart. The Phase II portion of the study will include approximately 350 pregnant women randomized 1:1 to receive BNT162b2 or placebo at 27 to 34 weeks’ gestation. The Phase III portion of this study will assess the safety, tolerability, and immunogenicity of BNT162b2 or placebo among pregnant women enrolled at 24 to 34 weeks’ gestation. Pfizer and BioNTech announced on 18 February 2021 that the first participants received their first dose in this trial.[61]
Vaccine technology
See also: RNA vaccine and COVID-19 vaccine § Technology platforms
The BioNTech technology for the BNT162b2 vaccine is based on use of nucleoside-modified mRNA (modRNA) which encodes part of the spike protein found on the surface of the SARS-CoV-2 coronavirus (COVID‑19), triggering an immune response against infection by the virus protein.[62]
The vaccine candidate BNT162b2 was chosen as the most promising among three others with similar technology developed by BioNTech.[18][62][56] Prior to choosing BNT162b2, BioNTech and Pfizer had conducted Phase I trials on BNT162b1 in Germany and the United States, while Fosun performed a Phase I trial in China.[17][63] In these Phase I studies, BNT162b2 was shown to have a better safety profile than the other three BioNTech candidates.[63]
Sequence
The modRNA sequence of the vaccine is 4,284 nucleotides long.[64] It consists of a five-prime cap; a five prime untranslated region derived from the sequence of human alpha globin; a signal peptide (bases 55–102) and two proline substitutions (K986P and V987P, designated “2P”) that cause the spike to adopt a prefusion-stabilized conformation reducing the membrane fusion ability, increasing expression and stimulating neutralizing antibodies;[17][65] a codon-optimized gene of the full-length spike protein of SARS-CoV-2 (bases 103–3879); followed by a three prime untranslated region (bases 3880–4174) combined from AES and mtRNR1 selected for increased protein expression and mRNA stability[66] and a poly(A) tail comprising 30 adenosine residues, a 10-nucleotide linker sequence, and 70 other adenosine residues (bases 4175–4284).[64] The sequence contains no uridine residues; they are replaced by 1-methyl-3′-pseudouridylyl.[64]
Composition
In addition to the mRNA molecule, the vaccine contains the following inactive ingredients (excipients):[67][68][8]
- ALC-0315, ((4-hydroxybutyl)azanediyl)bis(hexane-6,1-diyl)bis(2-hexyldecanoate)
- ALC-0159, 2-[(polyethylene glycol)-2000]-N,N-ditetradecylacetamide
- 1,2-distearoyl-sn-glycero-3-phosphocholine (DSPC)
- cholesterol
- dibasic sodium phosphate dihydrate
- monobasic potassium phosphate
- potassium chloride
- sodium chloride
- sucrose
- water for injection
The first four of these are lipids. The lipids and modRNA together form nanoparticles. ALC-0159 is a polyethylene glycol conjugate (that is, a PEGylated lipid).[69]
The vaccine is supplied in a multidose vial as “a white to off-white, sterile, preservative-free, frozen suspension for intramuscular injection“.[11][12] It must be thawed to room temperature and diluted with normal saline before administration.[12]
Authorizations
Expedited
The United Kingdom’s Medicines and Healthcare products Regulatory Agency (MHRA) gave the vaccine “rapid temporary regulatory approval to address significant public health issues such as a pandemic” on 2 December 2020, which it is permitted to do under the Medicines Act 1968.[70] It was the first COVID‑19 vaccine to be approved for national use after undergoing large scale trials,[71] and the first mRNA vaccine to be authorized for use in humans.[14][72] The United Kingdom thus became the first Western country to approve a COVID‑19 vaccine for national use,[73] although the decision to fast-track the vaccine was criticised by some experts.[74]
On 8 December 2020, Margaret “Maggie” Keenan, 90, from Fermanagh, became the first person to receive the vaccine.[75] In a notable example of museums documenting the pandemic, the vial and syringe used for that first dose were saved acquired by The Science Museum in London for its permanent collection.[76] By 20 December, 521,594 UK residents had received the vaccine as part of the national vaccination programme. 70% had been to people aged 80 or over.[77]
After the United Kingdom, the following countries expedited processes to approve the Pfizer–BioNTech COVID‑19 vaccine for use: Argentina,[78] Australia,[79] Bahrain,[80] Canada,[7][81] Chile,[82] Costa Rica,[83] Ecuador,[82] Hong Kong,[84] Iraq,[85] Israel,[86] Jordan,[87] Kuwait,[88] Mexico,[89] Oman,[90] Panama,[91] the Philippines,[92] Qatar,[93] Saudi Arabia,[32][94] Singapore,[95][96] the United Arab Emirates,[97] and the United States.[10]
The World Health Organization (WHO) authorized it for emergency use.[98]
In the United States, an emergency use authorization (EUA) is “a mechanism to facilitate the availability and use of medical countermeasures, including vaccines, during public health emergencies, such as the current COVID‑19 pandemic”, according to the FDA.[99] Following an EUA issuance, BioNTech and Pfizer are expected to continue the Phase III clinical trial to finalize safety and efficacy data, leading to application for licensure (approval) of the vaccine in the United States.[99][100][101] The United States Centers for Disease Control and Prevention (CDC) Advisory Committee on Immunization Practices (ACIP) approved recommendations for vaccination of those aged 16 years or older.[102][103]
Standard
On 19 December 2020, the Swiss Agency for Therapeutic Products (Swissmedic) approved the Pfizer–BioNTech COVID‑19 vaccine for regular use, two months after receiving the application, stating that the vaccine fully complied with the requirements of safety, efficacy and quality. This is the first authorization under a standard procedure.[1][104] On 23 December, a Lucerne resident, a 90-year-old woman, became the first person to receive the vaccine in Switzerland.[105] This marked the beginning of mass vaccination in continental Europe.[106]
On 21 December 2020, the Committee for Medicinal Products for Human Use (CHMP) of the European Medicines Agency (EMA) recommended granting conditional marketing authorization for the Pfizer–BioNTech COVID‑19 vaccine under the brand name Comirnaty.[2][107][108] The recommendation was accepted by the European Commission the same day.[107][109]
On February 23, 2021, the Brazilian Health Regulatory Agency approved the Pfizer–BioNTech COVID-19 vaccine under its standard marketing authorization procedure. It became the first COVID-19 vaccine to receive definitive registration rather than emergency use authorization in the country.[110]
Adverse effects
The adverse effect profile of the Pfizer–BioNTech COVID‑19 vaccine is similar to that of other adult vaccines.[20] During clinical trials, the side effects deemed very common[a] are (in order of frequency): pain and swelling at the injection site, tiredness, headache, muscle aches, chills, joint pain, and fever.[68] Fever is more common after the second dose.[68] These effects are predictable and to be expected, and it is particularly important that people be aware of this to prevent vaccine hesitancy.[111]
Severe allergic reaction has been observed in approximately 11 cases per million doses of vaccine administered.[112][113] According to a report by the US Centers for Disease Control and Prevention 71% of those allergic reactions happened within 15 minutes of vaccination and mostly (81%) among people with a documented history of allergies or allergic reactions.[112] The UK’s Medicines and Healthcare products Regulatory Agency (MHRA) advised on 9 December 2020, that people who have a history of “significant” allergic reaction should not receive the Pfizer–BioNTech COVID‑19 vaccine.[114][115][116] On 12 December, the Canadian regulator followed suit, noting that: “Both individuals in the U.K. had a history of severe allergic reactions and carried adrenaline auto injectors. They both were treated and have recovered.”[67]
On 28 January 2021, the European Union published a COVID-19 vaccine safety update which found that “the benefits of Comirnaty in preventing COVID‑19 continue to outweigh any risks, and there are no recommended changes regarding the use the vaccine.”[113][117] No new side effects were identified.[113]
Manufacturing

A doctor holding the Pfizer vaccine
Pfizer and BioNTech are manufacturing the vaccine in their own facilities in the United States and in Europe in a three-stage process. The first stage involves the molecular cloning of DNA plasmids that code for the spike protein by infusing them into Escherichia coli bacteria. In the United States, this stage is conducted at a small pilot plant in Chesterfield, Missouri[118] (near St. Louis). After four days of growth, the bacteria are killed and broken open, and the contents of their cells are purified over a week and a half to recover the desired DNA product. The DNA is stored in tiny bottles and frozen for shipment. Safely and quickly transporting the DNA at this stage is so important that Pfizer has used its company jet and helicopter to assist.[119]
The second stage is being conducted at plants in Andover, Massachusetts[120] in the United States, and in Germany. The DNA is used as a template to build the desired mRNA strands. Once the mRNA has been created and purified, it is frozen in plastic bags about the size of a large shopping bag, of which each can hold up to 5 to 10 million doses. The bags are placed on special racks on trucks which take them to the next plant.[119]
The third stage is being conducted at plants in Portage, Michigan[121] (near Kalamazoo) in the United States, and Puurs in Belgium. This stage involves combining the mRNA with lipid nanoparticles, then filling vials, boxing vials, and freezing them.[119] Croda International subsidiary Avanti Polar Lipids is providing the requisite lipids.[122] As of November 2020, the major bottleneck in the manufacturing process was combining mRNA with lipid nanoparticles.[119]
In February 2021, Pfizer revealed this entire sequence initially took about 110 days on average from start to finish, and that the company was making progress on reducing that number to 60 days.[123] Vaccine manufacturers normally take several years to optimize the process of making a particular vaccine for speed and cost-effectiveness before attempting large-scale production.[123] Due to the urgency presented by the COVID-19 pandemic, Pfizer began production immediately with the process by which the vaccine had been originally formulated in the laboratory, then started to identify ways to safely speed up and scale up that process.[123]
BioNTech announced in September 2020 that it had signed an agreement to acquire from Novartis a manufacturing facility in Marburg, Germany, to expand their vaccine production capacity.[124] Once fully operational, the facility would produce up to 750 million doses per year, or over 60 million doses per month. The site will be the third BioNTech facility in Europe which currently produces the vaccine, while Pfizer operates at least four production sites in the United States and Europe.
Advance orders and logistics
Pfizer indicated in its 9 November press release that 50 million doses could be available by the end of 2020, with about 1.3 billion doses provided globally by 2021.[20] In February 2021, BioNTech announced it would increase production by more than 50% to manufacture two billion doses in 2021.[125]
In July 2020, the vaccine development program Operation Warp Speed placed an advance order of US$1.95 billion with Pfizer to manufacture 100 million doses of a COVID‑19 vaccine for use in the United States if the vaccine was shown to be safe and effective.[34][126][127][128] By mid-December 2020, Pfizer had agreements to supply 300 million doses to the European Union,[129] 120 million doses to Japan,[130] 40 million doses (10 million before 2021) to the United Kingdom,[22] 20 million doses to Canada,[131] an unspecified number of doses to Singapore,[132] and 34.4 million doses to Mexico.[133] Fosun also has agreements to supply 10 million doses to Hong Kong and Macau.[134] The Hong Kong government said it would receive its first batch of one million doses by the first quarter of 2021.[135]
BioNTech and Fosun agreed to supply Mainland China with a batch of 100 million doses in 2021, subject to regulatory approval. The initial supply will be delivered from BioNTech’s production facilities in Germany.[136]
The vaccine is being delivered in vials that, once diluted, contain 2.25 ml of vaccine (0.45 ml frozen plus 1.8ml diluent).[101] According to the vial labels, each vial contains five 0.3 ml doses, however excess vaccine may be used for one, or possibly two, additional doses.[101][137] The use of low dead space syringes to obtain the additional doses is preferable, and partial doses within a vial should be discarded.[101][138] The Italian Medicines Agency officially authorized the use of excess doses remaining within single vials.[139] As of 8 January 2021, each vial contains six doses.[68][140][141][138] In the United States, vials will be counted as five doses when accompanied by regular syringes and as six doses when accompanied by low dead space syringes.[142]

Temperature the Pfizer vaccine must be kept at to ensure effectiveness, roughly between −80 and −60 °C (−112 and −76 °F)
Logistics in developing countries which have preorder agreements with Pfizer—such as Ecuador and Peru—remain unclear.[38] Even high-income countries have limited cold chain capacity for ultracold transport and storage of a vaccine that degrades within five days when thawed, and requires two shots three weeks apart.[38] The vaccine needs to be stored and transported at ultracold temperatures between −80 and −60 °C (−112 and −76 °F),[39][22][38][143][144] much lower than for the similar Moderna vaccine. The head of Indonesia‘s Bio Farma Honesti Basyir stated that purchasing the vaccine is out of the question for the world’s fourth-most populous country, given that it did not have the necessary cold chain capability. Similarly, India’s existing cold chain network can only handle temperatures between 2 and 8 °C (36 and 46 °F), far above the requirements of the vaccine.[145][146]
In January 2021, Pfizer and BioNTech offered to supply 50 million doses of COVID‑19 vaccine for health workers across Africa between March and the end of 2021, at a discounted price of US$10 per dose.[147]
Name
BNT162b2 was the code name during development and testing,[17][148] tozinameran is the proposed international nonproprietary name (pINN),[149] and Comirnaty is the brand name.[1][2] According to BioNTech, the name Comirnaty “represents a combination of the terms COVID‑19, mRNA, community, and immunity.”[150][151]
The vaccine also has the common name “COVID‑19 mRNA vaccine (nucleoside-modified)”[2] and may be distributed in packaging with the name Pfizer–BioNTech COVID‑19 Vaccine.”[152]
How the Pfizer-BioNTech Vaccine Works
By Jonathan Corum and Carl ZimmerUpdated Jan. 21, 2021Leer en español

The German company BioNTech partnered with Pfizer to develop and test a coronavirus vaccine known as BNT162b2, the generic name tozinameran or the brand name Comirnaty. A clinical trial demonstrated that the vaccine has an efficacy rate of 95 percent in preventing Covid-19.
A Piece of the Coronavirus
The SARS-CoV-2 virus is studded with proteins that it uses to enter human cells. These so-called spike proteins make a tempting target for potential vaccines and treatments.

Spikes
Spike
protein
gene
CORONAVIRUS
Like the Moderna vaccine, the Pfizer-BioNTech vaccine is based on the virus’s genetic instructions for building the spike protein.
mRNA Inside an Oily Shell
The vaccine uses messenger RNA, genetic material that our cells read to make proteins. The molecule — called mRNA for short — is fragile and would be chopped to pieces by our natural enzymes if it were injected directly into the body. To protect their vaccine, Pfizer and BioNTech wrap the mRNA in oily bubbles made of lipid nanoparticles.

Lipid nanoparticles
surrounding mRNA
Because of their fragility, the mRNA molecules will quickly fall apart at room temperature. Pfizer is building special containers with dry ice, thermal sensors and GPS trackers to ensure the vaccines can be transported at –94°F (–70°C) to stay viable.
Entering a Cell
After injection, the vaccine particles bump into cells and fuse to them, releasing mRNA. The cell’s molecules read its sequence and build spike proteins. The mRNA from the vaccine is eventually destroyed by the cell, leaving no permanent trace.

VACCINE
PARTICLES
VACCINATED
CELL
Spike
protein
mRNA
Translating mRNA
Three spike
proteins combine
Spike
Cell
nucleus
Spikes
and protein
fragments
Displaying
spike protein
fragments
Protruding
spikes
Some of the spike proteins form spikes that migrate to the surface of the cell and stick out their tips. The vaccinated cells also break up some of the proteins into fragments, which they present on their surface. These protruding spikes and spike protein fragments can then be recognized by the immune system.
Spotting the Intruder
When a vaccinated cell dies, the debris will contain many spike proteins and protein fragments, which can then be taken up by a type of immune cell called an antigen-presenting cell.

Debris from
a dead cell
Engulfing
a spike
ANTIGEN-
PRESENTING
CELL
Digesting
the proteins
Presenting a
spike protein
fragment
HELPER
T CELL
The cell presents fragments of the spike protein on its surface. When other cells called helper T cells detect these fragments, the helper T cells can raise the alarm and help marshal other immune cells to fight the infection.
Making Antibodies
Other immune cells, called B cells, may bump into the coronavirus spikes on the surface of vaccinated cells, or free-floating spike protein fragments. A few of the B cells may be able to lock onto the spike proteins. If these B cells are then activated by helper T cells, they will start to proliferate and pour out antibodies that target the spike protein.

HELPER
T CELL
Activating
the B cell
Matching
surface proteins
VACCINATED
CELL
B CELL
SECRETED
ANTIBODIES
Stopping the Virus
The antibodies can latch onto coronavirus spikes, mark the virus for destruction and prevent infection by blocking the spikes from attaching to other cells.

ANTIBODIES
VIRUS
Killing Infected Cells
The antigen-presenting cells can also activate another type of immune cell called a killer T cell to seek out and destroy any coronavirus-infected cells that display the spike protein fragments on their surfaces.

ANTIGEN-PRESENTING CELL Presenting a spike protein fragment ACTIVATED KILLER T CELL INFECTED CELL Beginning to kill the infected cell
Remembering the Virus
The Pfizer-BioNTech vaccine requires two injections, given 21 days apart, to prime the immune system well enough to fight off the coronavirus. But because the vaccine is so new, researchers don’t know how long its protection might last.

First dose, 0.3ml
Second dose, 21 days later
A preliminary study found that the vaccine seems to offer strong protection about 10 days after the first dose, compared with people taking a placebo:

Cumulative incidence of Covid-19 among clinical trial participants 2.5% 2.0 People taking a placebo
1.5 1.0 Second dose First dose People taking the
Pfizer-BioNTech vaccine
0.5
0
1
2
3
4
8
12
16
Weeks after the first dose
It’s possible that in the months after vaccination, the number of antibodies and killer T cells will drop. But the immune system also contains special cells called memory B cells and memory T cells that might retain information about the coronavirus for years or even decades.
For more about the vaccine, see Pfizer’s Covid Vaccine: 11 Things You Need to Know.
Preparation and Injection
Each vial of the vaccine contains 5 doses of 0.3 milliliters. The vaccine must be thawed before injection and diluted with saline. After dilution the vial must be used within six hours.

A diluted vial of the vaccine at Royal Free Hospital in London.Jack Hill/Agence France-Presse
References
- ^ Jump up to:a b According to the British National Formulary and MedDRA conventions, side effects are “very common” when they occur in more than 1 in 10 instances; “common”, 1 in 100 to 1 in 10; “uncommon”, 1 in 1,000 to 1 in 100; “rare”, 1 in 10,000 to 1 in 1,000; and “very rare” when they occur in less than 1 in 10,000 instances.[27]
- ^ Jump up to:a b c d “Swissmedic grants authorisation for the first COVID-19 vaccine in Switzerland”(Press release). Swiss Agency for Therapeutic Products (Swissmedic). 19 December 2020. Retrieved 19 December 2020.
- ^ Jump up to:a b c d e “Comirnaty EPAR”. European Medicines Agency (EMA). Retrieved 23 December 2020.
- ^ “Comirnaty”. Therapeutic Goods Administration (TGA). Retrieved 25 January 2021.
- ^ “Comirnaty (BNT162b2 [mRNA]) COVID‑19 Vaccine Product Information” (PDF). Therapeutic Goods Administration (TGA). Retrieved 25 January 2021.
- ^ Australian Public Assessment Report for BNT162b2 (mRNA) (PDF) (Report). Therapeutic Goods Administration (TGA). Retrieved 25 January 2021.
- ^ Jump up to:a b “Regulatory Decision Summary – Pfizer-BioNTech COVID-19 Vaccine”. Health Canada. 9 December 2020. Archived from the original on 9 December 2020. Retrieved 9 December 2020.
- ^ Jump up to:a b “Pfizer-BioNTech COVID-19 Vaccine (tozinameran)”. Health Canada. Retrieved 15 December 2020.
- ^ Jump up to:a b “Information for Healthcare Professionals on Pfizer/BioNTech COVID-19 vaccine”. Medicines and Healthcare products Regulatory Agency (MHRA). 10 December 2020. Retrieved 21 December 2020.
- ^ “Conditions of Authorisation for Pfizer/BioNTech COVID-19 vaccine”. Medicines and Healthcare products Regulatory Agency (MHRA). 31 December 2020. Retrieved 8 January2021.
- ^ Jump up to:a b “FDA Takes Key Action in Fight Against COVID-19 By Issuing Emergency Use Authorization for First COVID-19 Vaccine” (Press release). U.S. Food and Drug Administration (FDA). 11 December 2020. Retrieved 11 December 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain. - ^ Jump up to:a b c “Pfizer-BioNTech COVID-19 Vaccine- rna ingredient bnt-162b2 injection, suspension”. DailyMed. Retrieved 14 December 2020.
- ^ Jump up to:a b c Pfizer-BioNTech COVID-19 Vaccine Emergency Use Authorization Review Memorandum (PDF). U.S. Food and Drug Administration (FDA) (Report). 14 December 2020. Retrieved 14 December 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain. - ^ “Comirnaty EPAR”. European Medicines Agency (EMA). Retrieved 23 December 2020.
- ^ Jump up to:a b “UK medicines regulator gives approval for first UK COVID-19 vaccine” (Press release). Medicines and Healthcare products Regulatory Agency (MHRA). 2 December 2020. Retrieved 2 December 2020.
- ^ Boseley S, Halliday J (2 December 2020). “UK approves Pfizer/BioNTech Covid vaccine for rollout next week”. The Guardian. Retrieved 14 December 2020.
- ^ “Swissmedic grants authorisation for the first COVID-19 vaccine in Switzerland” (Press release). Swiss Agency for Therapeutic Products (Swissmedic). 19 December 2020. Retrieved 19 December 2020.
- ^ Jump up to:a b c d e Walsh EE, Frenck RW, Falsey AR, Kitchin N, Absalon J, Gurtman A, et al. (October 2020). “Safety and Immunogenicity of Two RNA-Based Covid-19 Vaccine Candidates”. The New England Journal of Medicine. 383 (25): 2439–50. doi:10.1056/NEJMoa2027906. PMC 7583697. PMID 33053279.
- ^ Jump up to:a b c d e f Clinical trial number NCT04368728 for “NCT04368728: Study to Describe the Safety, Tolerability, Immunogenicity, and Efficacy of RNA Vaccine Candidates Against COVID-19 in Healthy Individuals” at ClinicalTrials.gov
- ^ Jump up to:a b Palca J (9 November 2020). “Pfizer says experimental COVID-19 vaccine is more than 90% effective”. NPR. Archived from the original on 9 November 2020. Retrieved 9 November 2020.
- ^ Jump up to:a b c d e f g h Herper M (9 November 2020). “Covid-19 vaccine from Pfizer and BioNTech is strongly effective, early data from large trial indicate”. STAT. Archived from the original on 9 November 2020. Retrieved 9 November 2020.
- ^ Jump up to:a b Edwards E (9 November 2020). “Pfizer’s Covid-19 vaccine promising, but many questions remain”. NBC News. Archived from the original on 22 November 2020. Retrieved 12 November 2020.
- ^ Jump up to:a b c d Gallagher J (9 November 2020). “Covid vaccine: First ‘milestone’ vaccine offers 90% protection”. BBC News. Archived from the original on 26 November 2020. Retrieved 9 November 2020.
- ^ “CureVac Initiates Rolling Submission With European Medicines Agency for COVID-19 Vaccine Candidate, CVnCoV”. CureVac (Press release).
- ^ Jump up to:a b c “Update on our COVID-19 vaccine development program with BNT162b2” (PDF)(Press release). BioNTech. 2 December 2020. Retrieved 12 December 2020.
- ^ Polack FP, Thomas SJ, Kitchin N, Absalon J, Gurtman A, Lockhart S, et al. (December 2020). “Safety and Efficacy of the BNT162b2 mRNA Covid-19 Vaccine”. N Engl J Med. 383 (27): 2603–2615. doi:10.1056/NEJMoa2034577. PMC 7745181. PMID 33301246.
- ^ “Questions and Answers About Pfizer-BioNTech COVID-19 Vaccine”. Pfizer. Retrieved 16 December 2020.
- ^ “Adverse reactions to drugs”. British National Formulary. Retrieved 19 December 2020.
- ^ Jump up to:a b “Coronavirus vaccine”. National Health Service. 7 December 2020. Archived from the original on 7 December 2020. Retrieved 7 December 2020.
- ^ Commissioner, Office of the (3 February 2021). “Pfizer-BioNTech COVID-19 Vaccine”. FDA.
- ^ “EMA recommends first COVID-19 vaccine for authorisation in the EU”. European Medicines Agency.
- ^ “Bahrain becomes second country to approve Pfizer COVID-19 vaccine”. Al Jazeera. Archived from the original on 4 December 2020. Retrieved 5 December 2020.
* “Coronavirus: Saudi Arabia approves Pfizer COVID-19 vaccine for use”. Al Arabiya English. 10 December 2020. Archived from the original on 11 December 2020. Retrieved 10 December 2020.
* Solomon DB, Torres N (11 December 2020). “Mexico approves emergency use of Pfizer’s COVID-19 vaccine”. Reuters. Retrieved 12 December 2020.
* Thomas K (20 November 2020). “F.D.A. Clears Pfizer Vaccine, and Millions of Doses Will Be Shipped Right Away”. The New York Times. Archived from the original on 12 December 2020. Retrieved 11 December 2020.
* “First shipments of Pfizer-BioNTech vaccine in Singapore by end-Dec; enough vaccines for all by Q3 2021”. The Straits Times. 14 December 2020. Retrieved 14 December 2020. - ^ Jump up to:a b Al Mulla Y (13 December 2020). “Kuwait approves emergency use of Pfizer vaccine”. Gulf News. Retrieved 14 December 2020.
- ^ Browne R (11 November 2020). “What you need to know about BioNTech – the European company behind Pfizer’s Covid-19 vaccine”. CNBC. Retrieved 14 January 2021.
- ^ Jump up to:a b Thomas K, Gelles D, Zimmer C (9 November 2020). “Pfizer’s early data shows vaccine is more than 90% effective”. The New York Times. Archived from the original on 23 November 2020. Retrieved 9 November 2020.
- ^ Jump up to:a b c Burger L (15 March 2020). “BioNTech in China alliance with Fosun over coronavirus vaccine candidate”. Reuters. Archived from the original on 14 November 2020. Retrieved 10 November 2020.
- ^ “Pfizer and BioNTech Celebrate Historic First Authorization in the U.S. of Vaccine to Prevent COVID-19”. Pfizer Inc. and BioNTech SE.
- ^ “Securing Singapore’s access to COVID-19 vaccines”. gov.sg. Government of Singapore. 14 December 2020. Retrieved 1 February 2021.
- ^ Jump up to:a b c d e “Deep-freeze hurdle makes Pfizer’s vaccine one for the rich”. Bloomberg. 10 November 2020. Archived from the original on 22 November 2020. Retrieved 12 November 2020.
Vaccine goes bad five days after thawing, requires two shots; Many nations face costly ramp up of cold-chain infrastructure
- ^ Jump up to:a b c “Pfizer-BioNTech COVID-19 Vaccine Vaccination Storage & Dry Ice Safety Handling”. Pfizer. Retrieved 17 December 2020.
- ^ “Information for Healthcare Professionals on Pfizer/BioNTech COVID-19 vaccine”. Government of the United Kingdom. Retrieved 29 January 2021.
- ^ “Recommendation for an Emergency Use Listing of Tozinameran (Covid-19 Mrna Vaccine (Nucleoside Modified)) Submitted by Biontech Manufacturing Gmbh” (PDF). World Health Organization. 26 January 2021.
- ^ “Australian Product Information – Comirnaty (BNT162b2 [mRNA]) COVID-19 Vaccine”(PDF). Therapeutic Goods Administration. Australian Government.
- ^ “Pfizer and BioNTech Submit COVID-19 Vaccine Stability Data at Standard Freezer Temperature to the U.S. FDA”. Pfizer (Press release). 19 February 2021. Retrieved 19 February 2021.
- ^ Gates B (30 April 2020). “The vaccine race explained: What you need to know about the COVID-19 vaccine”. The Gates Notes. Archived from the original on 14 May 2020. Retrieved 2 May 2020.
- ^ “World Health Organization timeline – COVID-19”. World Health Organization. 27 April 2020. Archived from the original on 29 April 2020. Retrieved 2 May 2020.
- ^ Polack FP, Thomas SJ, Kitchin N, Absalon J, Gurtman A, Lockhart S, et al. (December 2020). “Safety and Efficacy of the BNT162b2 mRNA Covid-19 Vaccine”. The New England Journal of Medicine. 383 (27): 2603–2615. doi:10.1056/NEJMoa2034577. PMC 7745181. PMID 33301246.
development of BNT162b2 was initiated on January 10, 2020, when the SARS-CoV-2 genetic sequence was released by the Chinese Center for Disease Control and Prevention and disseminated globally by the GISAID (Global Initiative on Sharing All Influenza Data) initiative
- ^ Bohn MK, Mancini N, Loh TP, Wang CB, Grimmler M, Gramegna M, et al. (October 2020). “IFCC Interim Guidelines on Molecular Testing of SARS-CoV-2 Infection”. Clinical Chemistry and Laboratory Medicine. 58 (12): 1993–2000. doi:10.1515/cclm-2020-1412. PMID 33027042.
- ^ “CEPI’s collaborative task force to assess COVID-19 vaccines on emerging viral strains”. BioSpectrum – Asia Edition. 23 November 2020.
the first SARS-CoV-2 viral genomes were shared via GISAID on 10 January 2020
- ^ Thanh Le T, Andreadakis Z, Kumar A, Gómez Román R, Tollefsen S, Saville M, Mayhew S (May 2020). “The COVID-19 vaccine development landscape”. Nature Reviews. Drug Discovery. 19 (5): 305–306. doi:10.1038/d41573-020-00073-5. PMID 32273591.
- ^ Fauci AS, Lane HC, Redfield RR (March 2020). “Covid-19 – Navigating the Uncharted”. The New England Journal of Medicine. 382 (13): 1268–1269. doi:10.1056/nejme2002387. PMC 7121221. PMID 32109011.
- ^ Papadopoulos C (14 December 2020). “Chronologie – So entstand der Corona-Impfstoff von Biontech” [Chronology – That’s how the Covid-vaccine of Biontech was being developed] (in German). Südwestrundfunk. Retrieved 20 December 2020.
- ^ 《Fosun Pharma and BioNTech form COVID‑19 vaccine strategic alliance in China》(Fosun Phrama News Content , 15 March 2020) Archived 15 August 2020 at the Wayback Machine
- ^ “Germany: Investment Plan for Europe – EIB to provide BioNTech with up to €100 million in debt financing for COVID-19 vaccine development and manufacturing”. European Investment Bank. 11 June 2020. Archived from the original on 9 November 2020. Retrieved 10 November 2020.
- ^ “BioNTech gets $445 million in German funding for vaccine”. Bloomberg L.P. 15 September 2020. Archived from the original on 9 November 2020. Retrieved 10 November 2020.
- ^ “Pfizer CEO says he would’ve released vaccine data before election if possible”. Axios. 9 November 2020. Archived from the original on 10 November 2020. Retrieved 11 November 2020.
- ^ Jump up to:a b Mulligan MJ, Lyke KE, Kitchin N, Absalon J, Gurtman A, Lockhart S, et al. (October 2020). “Phase I/II study of COVID-19 RNA vaccine BNT162b1 in adults”. Nature. 586(7830): 589–593. Bibcode:2020Natur.586..589M. doi:10.1038/s41586-020-2639-4. PMID 32785213. S2CID 221126922.
- ^ Hannah B (7 October 2020). “EMA begins rolling review of BNT162b2 COVID-19 vaccine”. European Pharmaceutical Review. Archived from the original on 11 November 2020. Retrieved 11 November 2020.
- ^ Jump up to:a b “EMA Assessment Report” (PDF). Europa (web portal). 21 December 2020. Retrieved 29 December 2020.
- ^ “Clarification of Comirnaty dosage interval”. European Medicines Agency (EMA). 28 January 2021. Retrieved 28 January 2021.
- ^ “Study to Evaluate the Safety, Tolerability, and Immunogenicity of SARS CoV-2 RNA Vaccine Candidate (BNT162b2) Against COVID-19 in Healthy Pregnant Women 18 Years of Age and Older”. ClinicalTrials.gov. Retrieved 21 February 2021.
- ^ “Pfizer and BioNTech Commence Global Clinical Trial to Evaluate COVID-19 Vaccine in Pregnant Women”. pfizer.com (Press release). 18 February 2021. Retrieved 21 February2021.
- ^ Jump up to:a b Gaebler C, Nussenzweig MC (October 2020). “All eyes on a hurdle race for a SARS-CoV-2 vaccine”. Nature. 586 (7830): 501–2. Bibcode:2020Natur.586..501G. doi:10.1038/d41586-020-02926-w. PMID 33077943. S2CID 224808629.
- ^ Jump up to:a b “China’s Fosun to end BioNTech’s COVID-19 vaccine trial, seek approval for another”. Reuters. 3 November 2020. Archived from the original on 12 December 2020. Retrieved 21 November 2020.
- ^ Jump up to:a b c World Health Organization. “Messenger RNA encoding the full-length SARS-CoV-2 spike glycoprotein” (DOC). WHO MedNet. Retrieved 16 December 2020.
- ^ Pallesen J, Wang N, Corbett KS, Wrapp D, Kirchdoerfer RN, Turner HL, et al. (August 2017). “Immunogenicity and structures of a rationally designed prefusion MERS-CoV spike antigen”. Proceedings of the National Academy of Sciences of the United States of America. 114 (35): E7348–E7357. doi:10.1073/pnas.1707304114. PMC 5584442. PMID 28807998.
- ^ Orlandini von Niessen AG, Poleganov MA, Rechner C, Plaschke A, Kranz LM, Fesser S, et al. (April 2019). “Improving mRNA-Based Therapeutic Gene Delivery by Expression-Augmenting 3′ UTRs Identified by Cellular Library Screening”. Molecular Therapy. 27 (4): 824–836. doi:10.1016/j.ymthe.2018.12.011. PMC 6453560. PMID 30638957.
- ^ Jump up to:a b “Pfizer-BioNTech COVID-19 vaccine: Health Canada recommendations for people with serious allergies”. Health Canada. 12 December 2020.
- ^ Jump up to:a b c d Comirnaty: Product Information (PDF) (Report). European Medicines Agency(EMA). Retrieved 23 December 2020.
- ^ Public Assessment Report Authorisation for Temporary Supply COVID-19 mRNA Vaccine BNT162b2 (BNT162b2 RNA) concentrate for solution for injection (PDF). Regulation 174(Report). Medicines and Healthcare products Regulatory Agency (MHRA). 15 December 2020.
- ^ “UK medicines regulator gives approval for first UK COVID-19 vaccine”. Medicines and Healthcare products Regulatory Agency (MHRA). 2 December 2020. Archived from the original on 2 December 2020. Retrieved 2 December 2020.
- ^ Neergaard L, Kirka D (2 December 2020). “Britain OKs Pfizer vaccine and will begin shots within days”. Associated Press. Archived from the original on 6 December 2020. Retrieved 6 December 2020.
- ^ Mueller B (2 December 2020). “U.K. Approves Pfizer Coronavirus Vaccine, a First in the West”. The New York Times. Retrieved 2 December 2020.
- ^ Roberts M (2 December 2020). “Covid Pfizer vaccine approved for use next week in UK”. BBC News. Archived from the original on 2 December 2020. Retrieved 2 December 2020.
- ^ Henley J, Connolly, Jones S (3 December 2020). “European and US experts question UK’s fast-track of Covid vaccine”. The Guardian. Archived from the original on 9 December 2020. Retrieved 9 December 2020.
- ^ “First patient receives Pfizer Covid-19 vaccine”. BBC. 8 December 2020. Archivedfrom the original on 8 December 2020. Retrieved 8 December 2020.
- ^ “Vaccine vials and a virtual hug: a history of coronavirus in 15 objects”. The Guardian. 21 February 2021. Retrieved 22 February 2021.
- ^ “COVID-19 Vaccination Statistics –Week ending Sunday 20th December 2020” (PDF). NHS. 24 December 2020.
- ^ “Coronavirus en la Argentina: La ANMAT aprobo el uso de emergencia de la vacuna Pfizer”. La Nación (in Spanish). Retrieved 25 December 2020.
- ^ “TGA provisionally approves Pfizer COVID-19 vaccine”. Therapeutic Goods Administration (Press release). 25 January 2021. Retrieved 26 January 2021.
- ^ “Bahrain becomes second country to approve Pfizer COVID-19 vaccine”. Al Jazeera. Retrieved 5 December 2020.
- ^ “Drug and vaccine authorizations for COVID-19: List of applications received”. Health Canada. 9 December 2020. Retrieved 9 December 2020.
- ^ Jump up to:a b “Chile y Ecuador se adelantan en Sudamérica y autorizan la vacuna de Pfizer”. El Pais. Retrieved 17 December 2020.
- ^ “First Pfizer COVID-19 vaccines set to reach Costa Rica on Wednesday – president”. Reuters. 23 December 2020. Retrieved 24 December 2020.
- ^ “SFH authorises COVID-19 vaccine by Fosun Pharma/BioNTech for emergency use in Hong Kong”. The Government of Hong Kong (Press release). 25 January 2021. Retrieved 26 January 2021.
- ^ “Iraq grants emergency approval for Pfizer COVID-19 vaccine”. MSN. Retrieved 27 December 2020.
- ^ “Israeli Health Minister ‘pleased’ as FDA approves Pfizer COVID-19 vaccine”. The Jerusalem Post. Retrieved 28 December 2020.
- ^ “Jordan approves Pfizer-BioNTech Covid vaccine”. France 24. 15 December 2020. Retrieved 15 December 2020.
- ^ “Kuwait authorizes emergency use of Pfizer-BioNTech COVID-19 vaccine”. Arab News. 13 December 2020. Retrieved 15 December 2020.
- ^ “Mexico Approves Pfizer Vaccine for Emergency Use as Covid Surges”. Bloomberg. 12 December 2020. Retrieved 12 December 2020.
- ^ “Oman issues licence to import Pfizer BioNTech Covid vaccine – TV”. Reuters. 15 December 2020. Retrieved 16 December 2020.
- ^ “Panama approves Pfizer’s COVID-19 vaccine – health ministry”. Yahoo! Finance. Retrieved 16 December 2020.
- ^ “PH authorizes Pfizer’s COVID-19 vaccine for emergency use”. CNN Philippines. 14 January 2021.
- ^ “Qatar, Oman to receive Pfizer-BioNTech COVID-19 vaccine this week”. Reuters. Retrieved 24 December 2020.
- ^ “Saudi Arabia to Launch Its Coronavirus Vaccination Program” (in Spanish). Boomberg. Retrieved 17 December 2020.
- ^ Abdullah Z (14 December 2020). “Pfizer-BioNTech COVID-19 vaccine approved by Singapore, first shipment expected by end-December”. CNA. Retrieved 16 January 2021.
- ^ “Singapore approves use of Pfizer’s COVID-19 vaccine”. AP News. 14 December 2020. Retrieved 15 December 2020.
- ^ “Dubai approves the Pfizer-BioNTech vaccine which will be free of charge”. Emirates Woman. 23 December 2020. Retrieved 28 December 2020.
- ^ “WHO issues its first emergency use validation for a COVID-19 vaccine and emphasizes need for equitable global access”. World Health Organization (WHO) (Press release). 31 December 2020. Retrieved 6 January 2021.
- ^ Jump up to:a b “Emergency Use Authorization for vaccines explained”. U.S. Food and Drug Administration (FDA). 20 November 2020. Archived from the original on 20 November 2020. Retrieved 20 November 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain. - ^ “Pfizer-BioNTech COVID-19 Vaccine EUA Letter of Authorization” (PDF). U.S. Food and Drug Administration (FDA). 11 December 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain. - ^ Jump up to:a b c d “Pfizer-BioNTech COVID-19 Vaccine EUA Fact Sheet for Healthcare Providers”(PDF). Pfizer. 11 December 2020.
- ^ Sun LH, Stanley-Becker I. “CDC greenlights advisory group’s decision to recommend Pfizer vaccine for use”. The Washington Post. Retrieved 14 December 2020.
- ^ Oliver SE, Gargano JW, Marin M, Wallace M, Curran KG, Chamberland M, et al. (December 2020). “The Advisory Committee on Immunization Practices’ Interim Recommendation for Use of Pfizer-BioNTech COVID-19 Vaccine — United States, December 2020” (PDF). MMWR. Morbidity and Mortality Weekly Report. 69 (50): 1922–24. doi:10.15585/mmwr.mm6950e2. PMC 7745957. PMID 33332292.
- ^ “COVID-19: Switzerland can start vaccinating vulnerable groups already in December”(Press release). Federal Office of Public Health. 19 December 2020. Retrieved 19 December 2020.
- ^ Erni S (23 December 2020). “90-jährige Luzernerin als erste Person in der Schweiz gegen Corona geimpft”. Neue Luzerner Zeitung. Retrieved 23 December 2020.
- ^ Pralong J (23 December 2020). “La piqûre de l’espoir pratiquée à Lucerne”. Heidi.news. Retrieved 23 December 2020.
- ^ Jump up to:a b “EMA recommends first COVID-19 vaccine for authorisation in the EU”. European Medicines Agency (EMA) (Press release). 21 December 2020. Retrieved 21 December2020.
- ^ “Comirnaty”. Union Register of medicinal products. Retrieved 8 January 2021.
- ^ “Statement by President von der Leyen on the marketing authorisation of the BioNTech-Pfizer vaccine against COVID-19”. European Commission. Retrieved 21 December 2020.
- ^ Cancian, Natália (23 February 2021). “Anvisa aprova registro da vacina da Pfizer contra Covid”. Folha de S. Paulo (in Portuguese). Retrieved 23 February 2021.
- ^ McKenna M (17 December 2020). “Vaccines Are Here. We Have to Talk About Side Effects”. Wired. Retrieved 23 December 2020.
- ^ Jump up to:a b CDC COVID-19 Response Team, Food and Drug Administration (January 2021). “Allergic Reactions Including Anaphylaxis After Receipt of the First Dose of Pfizer-BioNTech COVID-19 Vaccine — United States, December 14–23, 2020” (PDF). MMWR. Morbidity and Mortality Weekly Report. 70 (2): 46–51. doi:10.15585/mmwr.mm7002e1. PMC 7808711. PMID 33444297.
- ^ Jump up to:a b c “COVID-19 vaccine safety update: COMIRNATY” (PDF). European Medicines Agency. 28 January 2021.
- ^ Bostock N (9 December 2020). “MHRA warning after allergic reactions in NHS staff given COVID-19 vaccine”. GP. Archived from the original on 9 December 2020. Retrieved 9 December 2020.
- ^ Booth W, Cunningham E (9 December 2020). “Britain warns against Pfizer vaccine for people with history of ‘significant’ allergic reactions”. The Washington Post. Archivedfrom the original on 9 December 2020. Retrieved 9 December 2020.
- ^ Cabanillas B, Akdis C, Novak N (December 2020). “Allergic reactions to the first COVID-19 vaccine: a potential role of Polyethylene glycol?”. Allergy. doi:10.1111/all.14711. PMID 33320974. S2CID 229284320.
- ^ “First COVID-19 vaccine safety update published”. European Medicines Agency (EMA)(Press release). 28 January 2021. Retrieved 29 January 2021.
- ^ Gray B (23 November 2020). “Pfizer’s Chesterfield workforce playing a key role in coronavirus vaccine development”. St. Louis Post-Dispatch.
- ^ Jump up to:a b c d Johnson CY (17 November 2020). “A vial, a vaccine and hopes for slowing a pandemic — how a shot comes to be”. The Washington Post. Retrieved 21 December2020.
- ^ Hughes M (20 December 2020). “Andover’s piece of the vaccine: Pfizer”. The Eagle-Tribune.
- ^ Shamus KJ (13 December 2020). “Historic journey: Pfizer prepares to deliver 6.4 million doses of COVID-19 vaccines”. Detroit Free Press.
- ^ Mullin R (25 November 2020). “Pfizer, Moderna ready vaccine manufacturing networks”. Chemical & Engineering News. Washington, D.C.: American Chemical Society. Retrieved 21 December 2020.
- ^ Jump up to:a b c Weise, Elizabeth (7 February 2021). “Pfizer expects to cut COVID-19 vaccine production time by close to 50% as production ramps up, efficiencies increase”. USA Today.
- ^ “BioNTech to Acquire GMP Manufacturing Site to Expand COVID-19 Vaccine Production Capacity in First Half 2021 | BioNTech”. investors.biontech.de. Retrieved 5 February2021.
- ^ “Statement on Manufacturing | BioNTech”. investors.biontech.de. Retrieved 5 February2021.
- ^ Erman M, Ankur B (22 July 2020). “U.S. to pay Pfizer, BioNTech $1.95 bln for millions of COVID-19 vaccine doses”. Reuters. Archived from the original on 22 July 2020. Retrieved 22 July 2020.
- ^ “U.S. Government Engages Pfizer to Produce Millions of Doses of COVID-19 Vaccine”. US Department of Health and Human Services. 22 July 2020. Archived from the original on 22 July 2020. Retrieved 23 July 2020.
- ^ Nazaryan A (9 November 2020). “So is Pfizer part of Operation Warp Speed or not? Yes and no”. Yahoo!. Archived from the original on 10 November 2020. Retrieved 9 November 2020.
- ^ Pleitgen F (11 November 2020). “EU agrees to buy 300 million doses of the Pfizer/BioNTech Covid-19 vaccine”. CNN. Archived from the original on 24 November 2020. Retrieved 26 November 2020.
- ^ “Japan and Pfizer reach COVID-19 vaccine deal to treat 60 million people”. The Japan Times. 1 August 2020. Archived from the original on 10 November 2020. Retrieved 21 November 2020.
- ^ Tasker JP (9 November 2020). “Trudeau says promising new Pfizer vaccine could be ‘light at the end of the tunnel'”. CBC News. Archived from the original on 9 November 2020. Retrieved 9 November 2020.
- ^ “Pfizer and BioNTech to Supply Singapore with their BNT162b2 mRNA-based Vaccine Candidate to Combat COVID-19”. pfizer.com.sg. Pfizer Singapore. 14 December 2020. Retrieved 1 February 2021.
- ^ de Salud S. “233. Firma secretario de Salud convenio con Pfizer para fabricación y suministro de vacuna COVID-19”. gob.mx (in Spanish). Retrieved 17 December 2020.
- ^ Ng E (27 August 2020). “Fosun Pharma to supply Covid-19 vaccine to Hong Kong, Macau once approved”. South China Morning Post. Archived from the original on 20 November 2020. Retrieved 21 November 2020.
- ^ Ting V, Lau C, Wong O (11 December 2020). “Hong Kong buys 15 million Covid-19 vaccine doses from Sinovac, Pfizer”. South China Morning Post. Retrieved 18 December2020.
- ^ “BioNTech and Fosun Pharma to Supply China with mRNA-based COVID-19 Vaccine”(Press release). BioNTech. 16 December 2020. Retrieved 16 December 2020.
- ^ “Pfizer-BioNTech COVID-19 Vaccine Frequently Asked Questions”. U.S. Food and Drug Administration. 11 December 2020. Retrieved 29 December 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain. - ^ Jump up to:a b “Extra dose from vials of Comirnaty COVID-19 vaccine”. European Medicines Agency (EMA). 8 January 2021. Retrieved 8 January 2021.
- ^ “AIFA, possibile ottenere almeno 6 dosi da ogni flaconcino del vaccino BioNTech/Pfizer”. aifa.gov.it (in Italian). Retrieved 29 December 2020.
- ^ “Global information about Comirnaty”. Comirnaty IE. 8 January 2021. Retrieved 16 January 2021.
- ^ “Comirnaty Package Insert” (PDF). BioNTech Manufacturing GmbH.
- ^ Rowland C (22 January 2021). “Biden wants to squeeze an extra shot of vaccine out of every Pfizer vial. It won’t be easy”. The Washington Post. Retrieved 29 January 2021.
- ^ Kollewe J. “Pfizer and BioNTech’s vaccine poses global logistics challenge”. The Guardian. Archived from the original on 10 November 2020. Retrieved 10 November2020.
- ^ Newey S (8 September 2020). “Daunting task of distribution exposed as it emerges some vaccines must be ‘deep frozen’ at −70C”. The Telegraph. Archived from the original on 9 November 2020. Retrieved 10 November 2020.
- ^ “How China’s COVID-19 could fill the gaps left by Pfizer, Moderna, AstraZeneca”. Fortune. 5 December 2020. Archived from the original on 12 December 2020. Retrieved 5 December 2020.
- ^ “Pfizer’s Vaccine Is Out of the Question as Indonesia Lacks Refrigerators: State Pharma Boss”. Jakarta Globe. 22 November 2020. Archived from the original on 7 December 2020. Retrieved 5 December 2020.
- ^ “Pfizer Has Offered South Africa Discounted Covid-19 Vaccines”. Bloomberg. 4 January 2021. Retrieved 5 January 2021.
- ^ Polack FP, Thomas SJ, Kitchin N, Absalon J, Gurtman A, Lockhart S, et al. (December 2020). “Safety and Efficacy of the BNT162b2 mRNA Covid-19 Vaccine”. N Engl J Med. 383 (27): 2603–2615. doi:10.1056/NEJMoa2034577. PMC 7745181. PMID 33301246.
- ^ World Health Organization (2020). “International Nonproprietary Names for Pharmaceutical Substances (INN). Proposed INN: List 124 – COVID-19 (special edition)”(PDF). WHO Drug Information. 34 (3): 666. Archived (PDF) from the original on 27 November 2020. Retrieved 23 November 2020.
- ^ “Pfizer and BioNTech Receive Authorization in the European Union for COVID-19 Vaccine” (Press release). BioNTech. 21 December 2020. Retrieved 26 December 2020 – via GlobeNewswire.
- ^ Bulik BS (23 December 2020). “The inside story behind Pfizer and BioNTech’s new vaccine brand name, Comirnaty”. FiercePharma. Retrieved 25 December 2020.
- ^ “Comirnaty COVID-19 mRNA Vaccine”. Comirnaty Global. Retrieved 31 December2020.
External links
“Tozinameran”. Drug Information Portal. U.S. National Library of Medicine.
- Global Information About Pfizer–BioNTech COVID‑19 Vaccine (also known as BNT162b2) Pfizer
- Comirnaty assessment report European Medicines Agency Committee for Medicinal Products for Human Use
- A Phase 1/2/3 Study to Evaluate the Safety, Tolerability, Immunogenicity, and Efficacy of RNA Vaccine Candidates Against COVID‑19 in Healthy Individuals Pfizer clinical protocol
- Pfizer Vaccince News, updates and tracking of Israel’s vaccinaion campaign
- “How the Pfizer-BioNTech Covid-19 Vaccine Works”. The New York Times.
| A vial of the Pfizer–BioNTech COVID‑19 vaccine | |
| Vaccine description | |
|---|---|
| Target disease | COVID‑19 |
| Type | mRNA |
| Clinical data | |
| Trade names | Comirnaty[1][2] |
| Other names | BNT162b2, COVID-19 mRNA vaccine (nucleoside-modified) |
| License data | EU EMA: by INNUS DailyMed: Pfizer-BioNTech_COVID-19_Vaccine |
| Pregnancy category | AU: B1[3] |
| Routes of administration | Intramuscular |
| ATC code | None |
| Legal status | |
| Legal status | AU: S4 (Prescription only) [4][5]CA: Authorized by interim order [6][7]UK: Conditional and temporary authorization to supply [8][9]US: Unapproved (Emergency Use Authorization)[10][11][12]EU: Conditional marketing authorization granted [2]CH: Rx-only[further explanation needed][1] |
| Identifiers | |
| CAS Number | 2417899-77-3 |
| PubChem SID | 434370509 |
| DrugBank | DB15696 |
| UNII | 5085ZFP6SJ |
| KEGG | D11971 |
| Part of a series on the |
| COVID-19 pandemic |
|---|
| SARS-CoV-2 (virus)COVID-19 (disease) |
| showTimeline |
| showLocations |
| showInternational response |
| showMedical response |
| showImpact |
| COVID-19 Portal |
/////////
#Tozinameran, #APPROVALS 2021, #JAPAN 2021, Comirnaty, #Coronavirus disease, #COVID-19, #BNT162b2 , #BNT162b2, #SARS-CoV-2 Vaccine, #RNA ingredient BNT-162B2, #corona
The Pfizer-BioNTech COVID-19 vaccine (Tozinameran, INN), also known as BNT162b2, is one of four advanced mRNA-based vaccines developed through “Project Lightspeed,” a joint program between Pfizer and BioNTech.2,3 Tozinameran is a nucleoside modified mRNA (modRNA) vaccine encoding an optimized full-length version of the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) spike (S) protein. It is designed to induce immunity against SARS-CoV-2, the virus responsible for causing COVID-19.2 The modRNA is formulated in lipid nanoparticles for administration via intramuscular injection in two doses, three weeks apart.1,3
Tozinameran is undergoing evaluation in clinical trials in both the USA (NCT04368728) and Germany (NCT04380701).4,5 Tozinameran received fast track designation by the U.S. FDA on July 13, 2020.6 On December 11, 2020, the FDA issued an Emergency Use Authorization (EUA) based on 95% efficacy in clinical trials and a similar safety profile to other viral vaccines over a span of approximately 2 months.1 Tozinameran was granted an EUA in the UK on December 2, 2020,8 and in Canada on December 9, 20207 for active immunization against SARS-CoV-2.12
Currently, sufficient data are not available to determine the longevity of protection against COVID-19, nor direct evidence that the vaccine prevents the transmission of the SARS-CoV-2 virus from one individual to another.9 Fact sheets for caregivers, recipients, and healthcare providers are now available.10,11
Tozinameran has not yet been fully approved by any country. In both the UK and Canada, Tozinameran is indicated under an interim authorization for active immunization to prevent COVID-19 caused by SARS-CoV-2 in individuals aged 16 years and older.7,8
On December 11, 2020, the U.S. Food and Drug Administration granted emergency use authorization (EUA) for Tozinameran to prevent COVID-19 caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) in patients aged 16 years and above.9 Safety and immune response information for adolescents 12-15 years of age will follow, and studies to further explore the administration of Tozinameran in pregnant women, children under 12 years of age, and those in special risk groups will be evaluated in the future.1
This vaccine should only be administered where appropriate medical treatment for immediate allergic reactions are immediately available in the case of an acute anaphylactic reaction after vaccine administration.12 Tozinameran administration should be postponed in any individual suffering from an acute febrile illness. Its use should be carefully considered in immunocompromised individuals and individuals with a bleeding disorder or on anticoagulant therapy. Appropriate medical treatment should be readily available in case of an anaphylactic reaction following vaccine administration.7,8
Tozinameran contains nucleoside modified mRNA (modRNA) encapsulated in lipid nanoparticles that deliver the modRNA into host cells. The lipid nanoparticle formulation facilitates the delivery of the RNA into human cells.12 Once inside these cells, the modRNA is translated by host machinery to produce the SARS-CoV-2 spike (S) protein antigen, which is subsequently recognized by the host immune system. Tozinameran has been shown to elicit both neutralizing antibody and cellular immune responses to the S protein, which helps protect against subsequent SARS-CoV-2 infection.7,8
Tozinameran is a nucleoside modified mRNA (modRNA) vaccine encoding an optimized full-length version of the SARS-CoV-2 spike (S) protein, translated and expressed in cells in vaccinated individuals to produce the S protein antigen against which an immune response is mounted. As with all vaccines, protection cannot be guaranteed in all recipients, and full protection may not occur until at least seven days following the second dose.7,8
In U.S. clinical trials, the vaccine was 95% effective in preventing COVID-19; eight COVID-19 cases occurred in the vaccine group and 162 cases occurred in the placebo group. Of the total 170 COVID-19 cases, one case in the vaccine group and three cases in the placebo group were considered to be severe infections.1,9
- Polack FP, Thomas SJ, Kitchin N, Absalon J, Gurtman A, Lockhart S, Perez JL, Perez Marc G, Moreira ED, Zerbini C, Bailey R, Swanson KA, Roychoudhury S, Koury K, Li P, Kalina WV, Cooper D, Frenck RW Jr, Hammitt LL, Tureci O, Nell H, Schaefer A, Unal S, Tresnan DB, Mather S, Dormitzer PR, Sahin U, Jansen KU, Gruber WC: Safety and Efficacy of the BNT162b2 mRNA Covid-19 Vaccine. N Engl J Med. 2020 Dec 10. doi: 10.1056/NEJMoa2034577. [PubMed:33301246]
- Gen Eng News: BNT162 vaccine candidates [Link]
- BioNTech BNT162 Update [Link]
- Clinical Trial NCT04368728 [Link]
- Clinical Trial NCT04380701 [Link]
- FDA fast track designation: BNT162b1 and BNT162b2 [Link]
- Health Canada Interim Product Monograph: BNT162b2 SARS-CoV-2 Vaccine [Link]
- MHRA Interim Product Monograph: BNT162b2 SARS-CoV-2 Vaccine [Link]
- FDA News Release: FDA Takes Key Action in Fight Against COVID-19 By Issuing Emergency Use Authorization for First COVID-19 Vaccine [Link]
- Pfizer: Fact Sheet for Healthcare Providers Administering Vaccine, Pfizer-BioNtech COVID-19 vaccine [Link]
- Pfizer: Fact Sheet for Recipients and Caregivers, Pfizer BioNTech COVID-19 vaccine [Link]
- FDA Emergency Use Authorization: Full EUA Prescribing information, Pfizer-BioNTech COVID-19 vaccine [Link]
-
PHASESTATUSPURPOSECONDITIONSCOUNT2Active Not RecruitingPreventionCoronavirus Disease 2019 (COVID‑19)12, 3Active Not RecruitingPreventionCoronavirus Disease 2019 (COVID‑19)11, 2Active Not RecruitingPreventionCoronavirus Disease 2019 (COVID‑19)11, 2RecruitingTreatmentCoronavirus Disease 2019 (COVID‑19) / Protection Against COVID-19 and Infections With SARS CoV 2 / Respiratory Tract Infections (RTI) / RNA Virus Infections / Vaccine Adverse Reaction / Viral Infections / Virus Diseases1

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....










