Home » Posts tagged 'Antilipemic'
Tag Archives: Antilipemic
Gemfibrozil

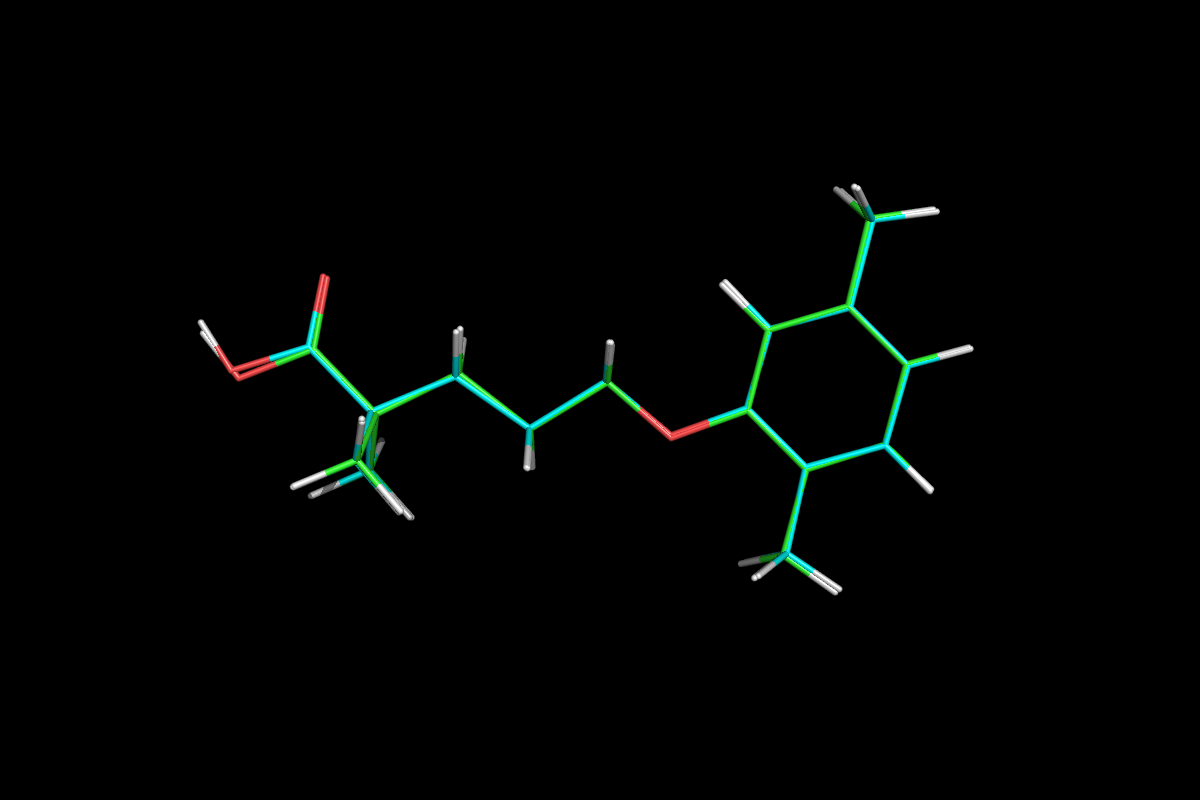
Gemfibrozil
5-(2,5-Dimethylphenoxy)-2,2-dimethylpentanoic Acid
IR (KBr, cm–1): 2959.03, 2919.78, 2877.65, 1709.42, 1613.44, 1586.60, 1511.07, 1473.81, 1414.01, 1387.89, 1317.61, 1286.34, 1271.91, 1214.39, 1159.26, 1048.83, 996.57, 803.75;
1H NMR (DMSO, 500 MHz, δ ppm): 1.12 (s, 6H), 1.60 and 1.67 (m, 4H), 2.08 (s, 3H), 2.24 (s, 3H), 3.90 (t, 2H), 6.62 (d, 1H), 6.70 (s, 1H), 6.97 (d, 1H);
13C NMR and DEPT (DMSO, 500 MHz, δ ppm): 15.39 (CH3), 20.94 (CH3), 24.67 (CH2), 24.87 (CH3, CH3), 36.43 (CH2), 40.91 (C), 67.57 (CH2), 112.07 (CH), 120.45 (CH), 122.44 (C), 129.96 (CH), 135.93 (C), 156.43 (C), 178.56 (C);
MS M/Z (ESI): 251.16 [(MH)+].
Solvent:CDCl3Instrument Type:JEOLNucleus:1HFrequency:400 MHzChemical Shift Reference:TMS
1H NMR spectrum of C15H22O3 in CDCL3 at 400 MHz
Gemfibrozil is the generic name for an oral drug used to lower lipid levels. It belongs to a group of drugs known as fibrates. It is most commonly sold as the brand name, Lopid. Other brand names include Jezil and Gen-Fibro.
history
Gemfibrozil was selected from a series of related compounds synthesized in the laboratories of the American company Parke Davisin the late 1970s. It came from research for compounds that lower plasma lipid levels in humans and in animals.[1]
Actions
- Is an activator of peroxisome proliferator-activated receptor-alpha (PPARα), a nuclear receptor that is involved in metabolism ofcarbohydrates and fats, as well as adipose tissue differentiation. This increase in the synthesis of lipoprotein lipase thereby increases the clearance of triglycerides.[citation needed]
Therapeutic effects
- Reduce triglyceride levels [2]
- Reduce very low density lipoprotein (VLDL) levels
- Modest reduction of low density lipoprotein (LDL) levels
- Moderate increase in high density lipoprotein (HDL) levels
Nontherapeutic effects and toxicities
- GI distress
- Musculoskeletal pain
- Increased incidence of gallstone
- Hypokalemia (low blood potassium)
- Increased risk of cancer
Indications
- Hyperlipidemia (Type III): Gemfibrozil is the drug of choice for therapy.
- Hypertriglyceridemia (Type IV): Gemfibrozil, though not as effective as niacin, is better tolerated.[citation needed]
Contraindications and precautions
- Gemfibrozil should not be given to these patients:
- Hepatic dysfunction
- Gemfibrozil should be used with caution in these higher risk categories:
- Biliary tract disease
- Renal dysfunction
- Pregnant women
- Obese patients
Drug interactions
- Anticoagulants: Gemfibrozil potentiates the action of warfarin and indanedione anticoagulants.[citation needed]
- Statin drugs: Concomitant administration of fibrates (including gemfibrozil) with statin drugs increases the risk of muscle cramping, myopathy, andrhabdomyolysis.
- Gemfibrozil inhibits the activation of the liver’s Cytochrome P450 system, reducing hepatic metabolism of many drugs, and prolonging their half lives and duration of action.
- Drugs metabolized by the Cytochrome P450 system include:
- Many antidepressants
- Many antipsychotics
- Many antiepileptics
- Theophylline and other methylxanthine drugs
- Several anesthetic agents
- Oral contraceptive pills
- Statins
- Warfarin
- Drugs metabolized by the Cytochrome P450 system include:
Environmental data
Gemfibrozil has been detected in biosolids (the solids remaining after wastewater treatment) at concentrations up to 2650 ng/g wet weight.[3] This indicates that it survives the wastewater treatment process.
SYNTHESIS


The sodium isobutyrate (I) is metallated with lithium diisopropylamide, and the resulting compound is alkylated with 3- (2,5-dimethylphenoxy) propyl bromide.
PATENT
Paul, L. C. 2,2-Dimethyl-ω-aryloxy alkanoic acids and salts and ester thereof. U.S. 3,674,836, 1972.
http://www.google.co.in/patents/US3674836
CLIP
Production of Gemfibrozil
(1)2,5-Dimethylphenol and 1-Bromo-3-chloropropane reaction of 1-(2,5-dimethylphenoxy)-3-chloropropane. The reaction is carried out in toluene, adding new clean off reflux 5h. Just as follows:

(2)N/A can be used to manufacture Gemfibrozil.

PAPER
Improved Process for Preparation of Gemfibrozil, an Antihypolipidemic
An improved process for the preparation of gemfibrozil, an antihypolipodimic drug substance, with an overall yield of 80% and ∼99.9% purity (including three chemical reactions) is reported. Formation and control of possible impurities are also described. Finally, gemfibrozil is isolated from water without any additional solvent purification.
Literature References:
Serum lipid regulating agent. Prepn: P. L. Creger, DE 1925423; eidem, US 3674836 (1969, 1972, both to Parke, Davis).
Production: O. P. Goel, US 4126637 (1978 to Warner-Lambert).
Pharmacology: A. H. Kissebach et al.,Atherosclerosis 24, 199 (1976); M. T. Kahonen et al., ibid. 32, 47 (1979).
Series of articles on metabolism, clinical pharmacology, kinetics and toxicology: Proc. R. Soc. Med. 69, Suppl 2, 1-120 (1976).
Toxicity data: S. M. Kurtz et al., ibid. 15.
Clinical trial in hyperlipidemia: J. E. Lewis et al., Pract. Cardiol. 9, 99 (1983).
Clinical reduction of cardiovascular risk in patients with low HDL levels: H. B. Rubins et al., N. Engl. J. Med. 341, 410 (1999).
References
- Rodney, G; et al. (1976). “The Hypolipidemic Effect of Gemfibrozil (CI-719) in Laboratory Animals”. Proc. roy. Soc. Med. 69 (Supplement 2): 6–9. PMC 1864017
 . PMID 828263.
. PMID 828263. - “Gemfibrozil.” WebMD.com Accessed 14 June 2014. http://www.webmd.com/drugs/drug-11423-gemfibrozil+oral.aspx
- http://water.epa.gov/scitech/wastetech/biosolids/tnsss-overview.cfm
External links
- DrugBank Gemfibrozil
- NIH Gemfibrozil Drug Info
- Lopid International Study
- Author (2004). “Safety of Statins”. Circulation. 109: III–50–III–57. doi:10.1161/01.cir.0000131519.15067.1f.
- http://www.ihs.gov/nptc/documents/NPTC%20Lipid%20Review.pdf
 |
|
| Systematic (IUPAC) name | |
|---|---|
|
5-(2,5-dimethylphenoxy)-2,2-dimethyl-pentanoic acid
|
|
| Clinical data | |
| Trade names | Lopid |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a686002 |
| Pregnancy category |
|
| Routes of administration |
Oral |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | Close to 100% |
| Protein binding | 95% |
| Metabolism | Hepatic (CYP3A4) |
| Biological half-life | 1.5 hours |
| Excretion | Renal 94% Feces 6% |
| Identifiers | |
| CAS Number | 25812-30-0 |
| ATC code | C10AB04 (WHO) |
| PubChem | CID 3463 |
| IUPHAR/BPS | 3439 |
| DrugBank | DB01241 |
| ChemSpider | 3345 |
| UNII | Q8X02027X3 |
| KEGG | D00334 |
| ChEBI | CHEBI:5296 |
| ChEMBL | CHEMBL457 |
| Chemical data | |
| Formula | C15H22O3 |
| Molar mass | 250.333 g/mol |
LOPID® (gemfibrozil tablets, USP) is a lipid regulating agent. It is available as tablets for oral administration. Each tablet contains 600 mg gemfibrozil. Each tablet also contains calcium stearate, NF; candelilla wax, FCC; microcrystalline cellulose, NF; hydroxypropyl cellulose, NF; hypromellose, USP; methylparaben, NF; Opaspray white; polyethylene glycol, NF; polysorbate 80, NF; propylparaben, NF; colloidal silicon dioxide, NF; pregelatinized starch, NF. The chemical name is 5-(2,5-dimethylphenoxy)2,2-dimethylpentanoic acid, with the following structural formula:
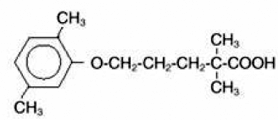 |
The empirical formula is C15H22O3 and the molecular weight is 250.35; the solubility in water and acid is 0.0019% and in dilute base it is greater than 1%. The melting point is 58° –61°C. Gemfibrozil is a white solid which is stable under ordinary conditions.
/////////Gemfibrozil, Antilipemic, Fibrates, 25812-30-0,
CC1=CC(OCCCC(C)(C)C(O)=O)=C(C)C=C1
TORCETRAPIB Revisted



Torcetrapib (CP-529,414, Pfizer) was a drug being developed to treat hypercholesterolemia (elevated cholesterol levels) and prevent cardiovascular disease. Its development was halted in 2006 when phase III studies showed excessive all-cause mortality in the treatment group receiving a combination of atorvastatin (Lipitor) and torcetrapib.

Medical uses
Torcetrapib has not been found to affect either cardiovascular disease or risk of death in those already taking a statin.[1]
Mechanism
Torcetrapib acts (as a CETP inhibitor) by inhibiting cholesterylester transfer protein (CETP), which normally transfers cholesterol from HDL cholesterol to very low density or low density lipoproteins (VLDL or LDL). Inhibition of this process results in higher HDL levels (the “good” cholesterol-containing particle) and reduces LDL levels (the “bad” cholesterol).[vague][citation needed]
Development
The first step in the synthesis was a recently created reaction of amination to p-chlorotrifluoryltoluene, a reaction that was created by Dr. Stephen Buchwald at MIT.[2]
Development of the drug began around 1990; it was first administered in humans in 1999, and manufacturing at production scale began in Ireland in 2005.[3]
Pfizer had previously announced that torcetrapib would be sold in combination with Pfizer’s statin, atorvastatin (Lipitor); however, following media and physician criticism, Pfizer had subsequently planned for torcetrapib to be sold independently of Lipitor.[4]
Clinical trials
A 2004 trial (19 subjects, non-randomised) showed that torcetrapib could increase HDL and lower LDL with and without an added statin.[5]
Nine phase III studies were completed.[6][7][8][9][10][11][12][13][14][15]
Early termination of study
On December 2, 2006 Pfizer cut off torcetrapib’s phase III trial because of “an imbalance of mortality and cardiovascular events” associated with its use.[16] This was a sudden and unexpected event and as late as November 30, 2006 Jeff Kindler, Pfizer’s chief executive, was quoted, “This will be one of the most important compounds of our generation.”[16] In the terminated trial, a 60% increase in deaths was observed among patients taking torcetrapib and atorvastatin versus taking atorvastatin alone.[17] Pfizer recommended that all patients stop taking the drug immediately.[18]
Six studies were terminated early.[6] One of the completed studies found it raised systolic blood pressure and concluded “Torcetrapib showed no clinical benefit in this or other studies, and will not be developed further.”[19]
The drug cost $800m+ to bring into Phase III development.[20]




| A Concise Asymmetric Synthesis of Torcetrapib�, M. Guino, P. H. Phua, J-C. Caille and K. K. Hii, J. Org. Chem., 2007, 72, 6290-6293.doi:10.1021/jo071031gAbstract: Optically active torcetrapib was synthesized in seven steps from achiral precursors without the need for protecting groups, utilizing an enantioselective aza-Michael reaction to achieve asymmetry. |

Example 9 Anhydrous, (-)-(2R,4S)-4-[(3,5-Bιs-trιfluoromethyl-benzyl)-methoxycarbonyl-amιnol-2- ethyl-6-trιfluoromethyl-3,4-dιhydro-2H-quιnolιne-1 -carboxylic acid ethyl ester.
A 2.6g portion of 4(S)-[(3,5-bιs-tπfluoromethyl-benzyl)-methoxycarbonyl-amιno]-2(R)- ethyl-6-tπfluoromethyl-3,4-dιhydro-2H-quιnolιne-1 -carboxylic acid ethyl ester (a mixture of predominantly amorphous material with traces of ethanolate crystalline form; the title compound was also prepared in an analogous manner starting from pure amorphous or pure ethanolate material) was charged to 13 milliliters of hexanes and heated to effect a solution at about 60°C The heat was removed and the reaction was allowed to cool to ambient over a one hour period The reaction was seeded with anhydrous (-)-(2R,4S)-4-[(3,5-bιs-tπfluoromethyl-benzyl)- methoxycarbonyl-amιno]-2-ethyl-6-trιfluoromethyl-3,4-dιhydro-2H-quιnolιne-1 – carboxylic acid ethyl ester and granulated for eighteen hours under ambient conditions. Alternately, the anhydrous crystals may be prepared from hexanes without seeding. The product was collected by filtration and air dried. The isolated product X-ray pattern matched the calculated powder pattern. Density: 1.406 Crystal System: Trigonal
Microscopy: Well formed rods and equant (fractured rods) crystals demonstrating high birefringence when viewed across the C axis. Being in the Trigonal crystal system the crystals do not demonstrate birefringence when viewed down the C axis. The crystals demonstrate a cleavage plane perpendicular to the C axis Fusion Microsocopy In Type A oil dissolution at 50°C.
Dry — clear melt at 86°C.
NMR: No trace of ethanolate
Degree of crystallmity: Highly crystalline Hygroscopicity. Non-hygroscopic at 100% relative humidity over 48 hours.
Appearance: Free flowing white powder

References
- Clark, RW; Sutfin TA, Ruggeri RB, Willauer AT, Sugarman ED, Magnus-Aryitey G, Cosgrove PG, Sand TM, Wester RT, Williams JA, Perlman ME, Bamberger MJ (January 22, 2004). “Raising high-density lipoprotein in humans through inhibition of cholesteryl ester transfer protein: an initial multidose study of torcetrapib”. Arteriosclerosis, Thrombosis, and Vascular Biology 24 (3): 490–497. doi:10.1161/01.ATV.0000118278.21719.17. PMID 14739125. Retrieved 2006-12-03.
- Clark, RW; Ruggeri RB; Cunningham D; Bamberger MJ (March 2006). “Description of the torcetrapib series of cholesteryl ester transfer protein inhibitors, including mechanism of action”. Journal of Lipid Research 47 (3): 537–552. doi:10.1194/jlr.M500349-JLR200. PMID 16326978. Retrieved 2006-12-03.
- Davidson, MH; McKenny JM; Shear CL; Revkin JH (November 7, 2006). “Efficacy and safety of torcetrapib, a novel cholesteryl ester transfer protein inhibitor, in individuals with below-average high-density lipoprotein cholesterol levels”. Journal of the American College of Cardiology 48 (9): 1774–1781. doi:10.1016/j.jacc.2006.06.067.PMID 17084249. Retrieved 2006-12-03.
- McKenny, JM; Davidson MH; Shear CL; Revkin JH (November 7, 2006). “Efficacy and safety of torcetrapib, a novel cholesteryl ester transfer protein inhibitor, in individuals with below-average high-density lipoprotein cholesterol levels on a background of atorvastatin”. Journal of the American College of Cardiology 48 (9): 1782–1790.doi:10.1016/j.jacc.2006.06.066. PMID 17084250. Retrieved 2006-12-03.
Notes
- Keene, D; Price, C; Shun-Shin, MJ; Francis, DP (Jul 18, 2014). “Effect on cardiovascular risk of high density lipoprotein targeted drug treatments niacin, fibrates, and CETP inhibitors: meta-analysis of randomised controlled trials including 117,411 patients.”. BMJ (Clinical research ed.) 349: g4379. PMID 25038074.
- Buchwald, Stephen (July 23, 2004). “Research Projects”. Retrieved 2007-10-04.
- “Pfizer Begins Production at Torcetrapib/Atorvastatin Manufacturing Facility” (Press release). Pfizer. June 22, 2005. Retrieved 2006-12-03.
- Berenson, Alex (July 26, 2006). “Heart Pill to Be Sold by Itself”. Business (The New York Times). Retrieved 2006-12-03.
- Brousseau, ME; Schaefer EJ; Wolfe ML; Bloedon LT; Digenio AG; Clark RW; Mancuso JP; Rader DJ (April 8, 2004). “Effects of an inhibitor of cholesteryl ester transfer protein on HDL cholesterol” (abstract). New England Journal of Medicine 350 (15): 1505–1515. doi:10.1056/NEJMoa031766. PMID 15071125. Retrieved 2006-12-03.
- http://clinicaltrials.gov/ct2/results?term=torcetrapib
- http://clinicaltrials.gov/ct2/show/NCT00139061 Phase III Assess HDL-C Increase And Non-HDL Lowering Effect Of Torcetrapib/Atorvastatin Vs. Fenofibrate
- http://clinicaltrials.gov/ct2/show/NCT00134511 Phase III Study To Evaluate The Effect Of Torcetrapib/Atorvastatin In Patients With Genetic High Cholesterol Disorder
- http://clinicaltrials.gov/ct2/show/NCT00134485 Phase III Study To Evaluate The Safety And Efficacy Of Torcetrapib/Atorvastatin In Subjects With Familial Hypercholerolemia
- http://clinicaltrials.gov/ct2/show/NCT00134498 Phase III Study Comparing The Efficacy & Safety Of Torcetrapib/Atorvastatin And Atorvastatin In Subjects With High Triglycerides
- http://clinicaltrials.gov/ct2/show/NCT00267254 Phase III Clinical Trial Comparing Torcetrapib/Atorvastatin To Simvastatin In Subjects With High Cholesterol
- http://clinicaltrials.gov/ct2/show/NCT00138762 Phase III Study of Torcetrapib/Atorvastatin vs Atorvastatin Alone or Placebo in Patients With High Cholesterol
- http://clinicaltrials.gov/ct2/show/NCT00134173 Phase III Coronary IVUS Study to Compare Torcetrapib/Atorvastatin to Atorvastatin Alone in Subjects With Coronary Heart Disease (ILLUSTRATE)
- http://clinicaltrials.gov/ct2/show/NCT00137462 Phase III Lipitor Trial To Study The Effect Of Torcetrpib/Atorvastatin To Atorvastatin Alone.
- http://clinicaltrials.gov/ct2/show/NCT00136981 Phase III Carotid B-Mode Ultrasound Study to Compare Anti-Atherosclerotic Effect of Torcetrpib/Atorvastatin to Atorvastatin Alone. (RADIANCE 1)
- Berenson, Alex (December 3, 2006). “Pfizer Ends Studies on Drug for Heart Disease”. The New York Times. Retrieved 2006-12-03. (registration required)
- Theresa Agovino (Associated Press) (December 3, 2006). “Pfizer ends cholesterol drug development”. Yahoo! News. Retrieved 2006-12-03.[dead link] Each study arm (torcetrapib + atorvastatin vs. atorvastatin alone) had 7500 patients enrolled; 51 deaths were observed in the atorvastatin alone arm, while 82 deaths occurred in the torcetrapib + atorvastatin arm. (Link dead as of 15 January 2007)
- Associated Press (December 2, 2006). “Pfizer cuts off cholesterol drug trials”. Yahoo! News (Yahoo.com). Retrieved 2006-12-03.[dead link] (Link dead as of 15 January 2007)
- Bots et al.; Visseren, Frank L; Evans, Gregory W; Riley, Ward A; Revkin, James H; Tegeler, Charles H; Shear, Charles L; Duggan, William T et al. (July 2007). “Torcetrapib and carotid intima-media thickness in mixed dyslipidaemia (RADIANCE 2 study): a randomised, double-blind trial”. The Lancet 370 (9582): 153–160. doi:10.1016/S0140-6736(07)61088-5.
- Cutler, D. M. (2007-03-29). “The Demise of the Blockbuster?”. The New England Journal of Medicine (Massachusetts Medical Society) 356 (13): 1292–1293. doi:10.1056/NEJMp078020.ISSN 1533-4406. PMID 17392299. Retrieved 2007-04-23.

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....












