Home » Posts tagged 'ANAX'
Tag Archives: ANAX
Ulacamten
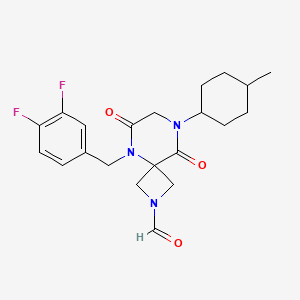
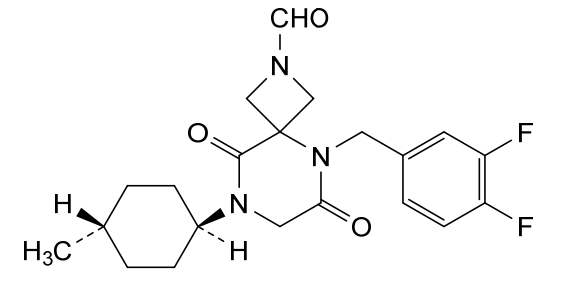
Ulacamten
CAS 2830607-59-3
MF C21H25F2N3O3 MW405.4 g/mol
5-[(3,4-difluorophenyl)methyl]-8-(4-methylcyclohexyl)-6,9-dioxo-2,5,8-triazaspiro[3.5]nonane-2-carbaldehyde
5-[(3,4-difluorophenyl)methyl]-8-[(1r,4r)-4-methylcyclohexyl]-6,9-dioxo-2,5,8-triazaspiro[3.5]nonane-2-carbaldehyde
cardiac myosin inhibitor, CK-586, CK-4021586, CK 586, CK 4021586, X325G97HZJ
Ulacamten (also known as CK-586 or CK-4021586) is an investigational drug developed by Cytokinetics that acts as a cardiac myosin inhibitor (CMI). It is currently being studied for the treatment of heart failure with preserved ejection fraction (HFpEF), a condition where the heart muscle is too stiff to fill properly
Key Characteristics and Development
- Mechanism of Action: Unlike earlier CMIs like mavacamten or aficamten, ulacamten is highly selective. It binds to the regulatory light chain (RLC) of myosin and only inhibits the “two-headed” form of cardiac myosin, potentially allowing for more precise control over heart muscle contraction.
- Clinical Status: As of March 2026, it is in Phase 2 clinical trials (specifically the AMBER-HFpEF study) to evaluate its safety, tolerability, and optimal dosage in patients with symptomatic HFpEF.
- Administration: It is designed as an orally active small molecule intended for once-daily dosing.
- Potential Benefits: Preclinical studies suggest it can reduce excessive myocardial contractility and improve left ventricular relaxation (diastolic function) without significantly compromising the heart’s overall pumping ability.
- AMBER-HFpEF: Assessment of CK-4021586 in a Multi-Center, Blinded Evaluation of Safety and Tolerability Results in HFpEFCTID: NCT06793371Phase: Phase 2Status: RecruitingDate: 2026-01-12
- A Single and Multiple Ascending Dose Study of CK-4021586 in Healthy Adult ParticipantsCTID: NCT05877053Phase: Phase 1Status: CompletedDate: 2025-05-01
SYN
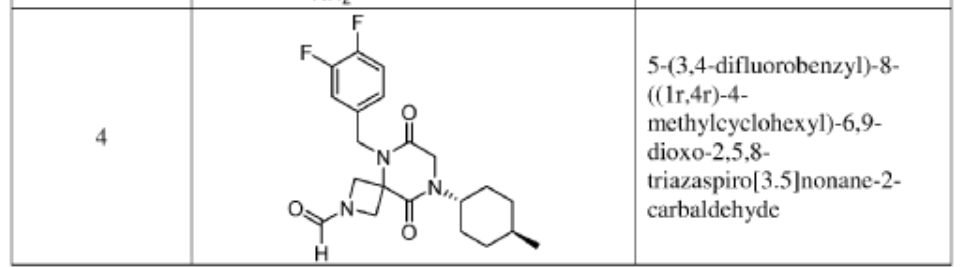
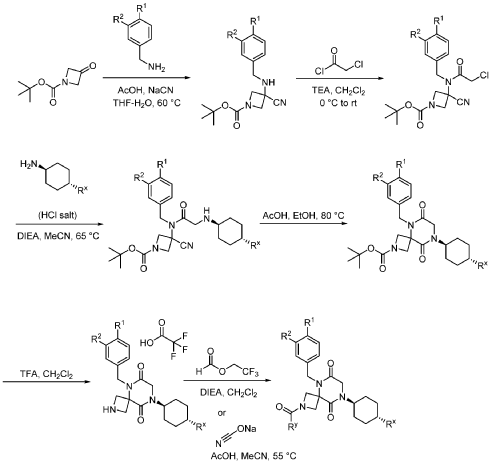
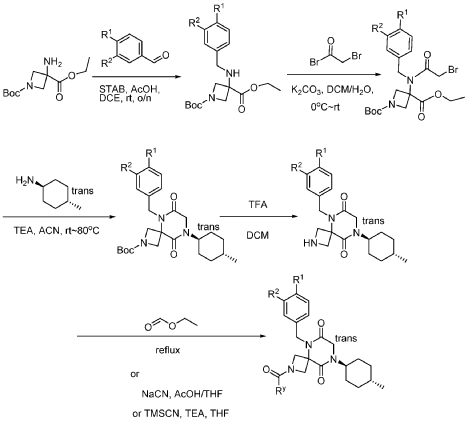
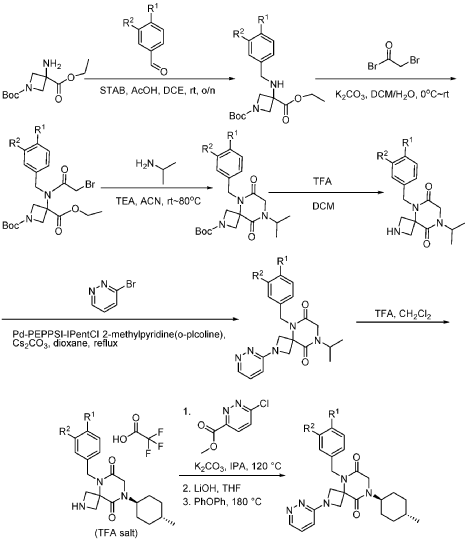
Example 1
Synthesis of 5-(3,4-difluorobenzyl)-8-((1r,4r)-4-methylcyclohexyl)-6,9-dioxo-2,5,8-triazaspiro[3.5]nonane-2-carbaldehyde
(Compound 4)
Step 1: Synthesis of 1-(tert-butyl) 3-ethyl 3-((3,4-difluorobenzyl)amino)azetidine-1,3-dicarboxylate:
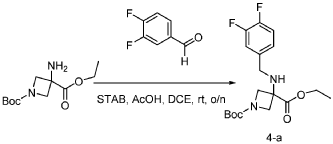
[0147] To a solution of 1-tert-butyl 3-ethyl 3-aminoazetidine-1,3-dicarboxylate (4.0 g, 16.4 mmol, 1.0 equiv) and 3,4-difluorobenzaldehyde (2.4 g, 19.6 mmol, 1.2 equiv) in DCE (40.0 mL) at 0 ˚C were added STAB (7.0 g, 32.8 mmol, 2.0 equiv) and AcOH (2.0 g, 32.8 mmol, 2.0 equiv). The resulting mixture was stirred at rt overnight, adjusted the pH to 8 with ammonium hydroxide, added water (50.0 mL) and extracted with DCM (50.0 mL) twice. The combined organic layers were washed with brine (50 mL) twice, dried over anhydrous Na 2 SO 4 , and concentrated under reduced pressure to afford 6.0 g of 1-tert-butyl 3-ethyl 3-(3,4-difluorobenzyl)amino)azetidine-1,3-dicarboxylate as a yellow oil. LRMS (ES) m/z 315 (M+H-56).
Step 2: Synthesis of 1-(tert-butyl) 3-ethyl 3-(2-bromo-N-(3,4-difluorobenzyl)acetamido)azetidine-1,3-dicarboxylate:
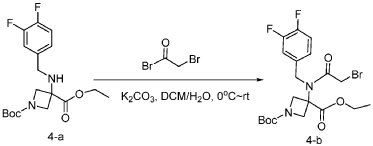
[0148] To a solution of 1-tert-butyl 3-ethyl 3-[[(3,4-difluorophenyl)methyl]amino]azetidine-1,3-dicarboxylate (6.0 g, 16.2 mmol, 1.0 equiv) in DCM (60.0 mL) at 0 ˚C were added a solution of K 2 CO 3 (3.4 g, 24.3 mmol, 1.50 equiv) in water (30 mL), and then bromoacetyl bromide (3.9 g, 19.4 mmol, 1.2 equiv) dropwise over a period of 10 min. The resulting mixture was stirred at rt overnight and extracted with DCM (50.0 mL) twice. The combined organic layers were washed with brine (100 mL) twice, dried over anhydrous Na 2 SO 4 , and concentrated under reduced pressure to afford 8.0 g of 1-(tert-butyl) 3-ethyl 3-(2-bromo-N-(3,4-difluorobenzyl)acetamido)azetidine-1,3-dicarboxylate as a yellow oil. LRMS (ES) m/z 435 (M+H-56).
Step 3: Synthesis of tert-butyl 5-(3,4-difluorobenzyl)-8-((1r,4r)-4-methylcyclohexyl)-6,9-dioxo-2,5,8-triazaspiro[3.5]nonane-2-carboxylate:
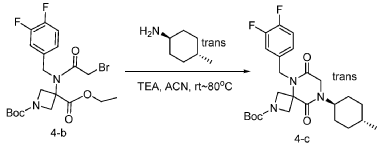
[0149] To a solution of 1-(tert-butyl) 3-ethyl 3-(2-bromo-N-(3,4-difluorobenzyl)acetamido)azetidine-1,3-dicarboxylate (8.0 g, 16.3 mmol, 1.0 equiv) in ACN (80 mL) were added TEA (4.9 g, 48.4 mmol, 3.0 equiv) and trans-(1r,4r)-4-methylcyclohexan-1-amine (2.8 g, 24.7 mmol, 1.5 equiv). The resulting mixture was stirred at rt for 1 h, gradually warmed to 80 ˚C, and stirred at 80 ˚C overnight. The mixture was cooled to rt, concentrated under reduced pressure, and triturated with a mixture of PE and EA (7/1; 80 mL) to afford 7 g (~80% purity) of tert-butyl 5-(3,4-difluorobenzyl)-8-((1r,4r)-4-methylcyclohexyl)-6,9-dioxo-2,5,8-triazaspiro[3.5]nonane-2-carboxylate as an off-white solid. LRMS (ES) m/z 422 (M+H-56).
Step 4: Synthesis of 5-(3,4-difluorobenzyl)-8-((1r,4r)-4-methylcyclohexyl)-2,5,8-triazaspiro[3.5]nonane-6,9-dione:
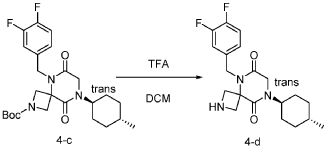
[0150] To a stirred solution of tert-butyl 5-(3,4-difluorobenzyl)-8-((1r,4r)-4-methylcyclohexyl)-6,9-dioxo-2,5,8-triazaspiro[3.5]nonane-2-carboxylate (7.0 g, 14.7 mmol, 1.0 equiv) in DCM (70.0 mL) was added TFA (18.0 mL). The resulting mixture was stirred at rt for 3h, diluted with water (100.0 mL), adjusted the pH to 13-14 with aqueous NaOH solution (2 N), and extracted with DCM (100 mL) twice. The combined organic layers were washed with brine (100.0 mL) twice, dried over anhydrous Na 2 SO 4 , and concentrated under reduced pressure to afford 4.5 g (~80% purity) of 5-(3,4-difluorobenzyl)-8-((1r,4r)-4-methylcyclohexyl)-2,5,8-triazaspiro[3.5]nonane-6,9-dione as a yellow semi-solid. LRMS (ES) m/z 378 (M+H).
Step 5: Synthesis of 5-(3,4-difluorobenzyl)-8-((1r,4r)-4-methylcyclohexyl)-6,9-dioxo-2,5,8-triazaspiro[3.5]nonane-2-carbaldehyde (Compound 4):
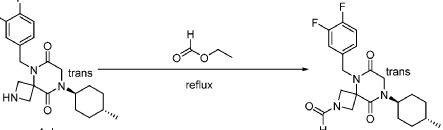
[0151] A solution of tert-butyl 5-(3,4-difluorobenzyl)-8-((1r,4r)-4-methylcyclohexyl)-6,9-dioxo-2,5,8-triazaspiro[3.5]nonane-2-carboxylate (1.5 g, 4.0 mmol, 1.0 equiv) in ethyl formate (15.0 mL) was stirred at 80
o C overnight. The mixture was cooled to rt, concentrated under reduced pressure, and purified by C18 column chromatography, eluted with a mixture of water (0.05% NH
4 HCO
3 )/CH
3 CN (3:2) to afford 1.3 g (81%) of 5-(3,4-difluorobenzyl)-8-((1r,4r)-4-methylcyclohexyl)-6,9-dioxo-2,5,8-triazaspiro[3.5]nonane-2-carbaldehyde as an amorphous white solid. An experimental X-ray powder diffraction (XRPD) pattern of this amorphous white solid is shown in FIG. 1 LRMS (ES) m/z 406 (M+H);
1 H NMR (300 MHz, DMSO-d6) δ 7.96 (s, 1H), 7.47 – 7.29 (m, 2H), 7.10 (ddd, J = 9.4, 4.4, 2.0 Hz, 1H), 4.82 (s, 2H), 4.50 (d, J = 9.6 Hz, 1H), 4.15-4.28 (m, J = 3H), 4.01 (s, 2H), 3.96 (d, J = 10.8 Hz, 1H), 1.80 – 1.69 (m, 2H), 1.65 – 1.48 (m, 4H), 1.35 (d, J = 10.9 Hz, 1H), 1.13 – 0.93 (m, 2H), 0.88 (d, J = 6.5 Hz, 3H).
PAT
5-(3,4-difluorobenzyl)-8-((1r,4r)-4-methylcyclohexyl)-6,9-dioxo-2,5,8-triazaspiro[3.5]nonane-2-carbaldehyde, also called compound 1, having the structure shown below,
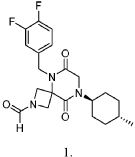
Example 1
Synthesis of 5-(3,4-difluorobenzyl)-8-((1r,4r)-4-methylcyclohexyl)-6,9-dioxo-2,5,8-triazaspiro[3.5]nonane-2-carbaldehyde (Compound 1)
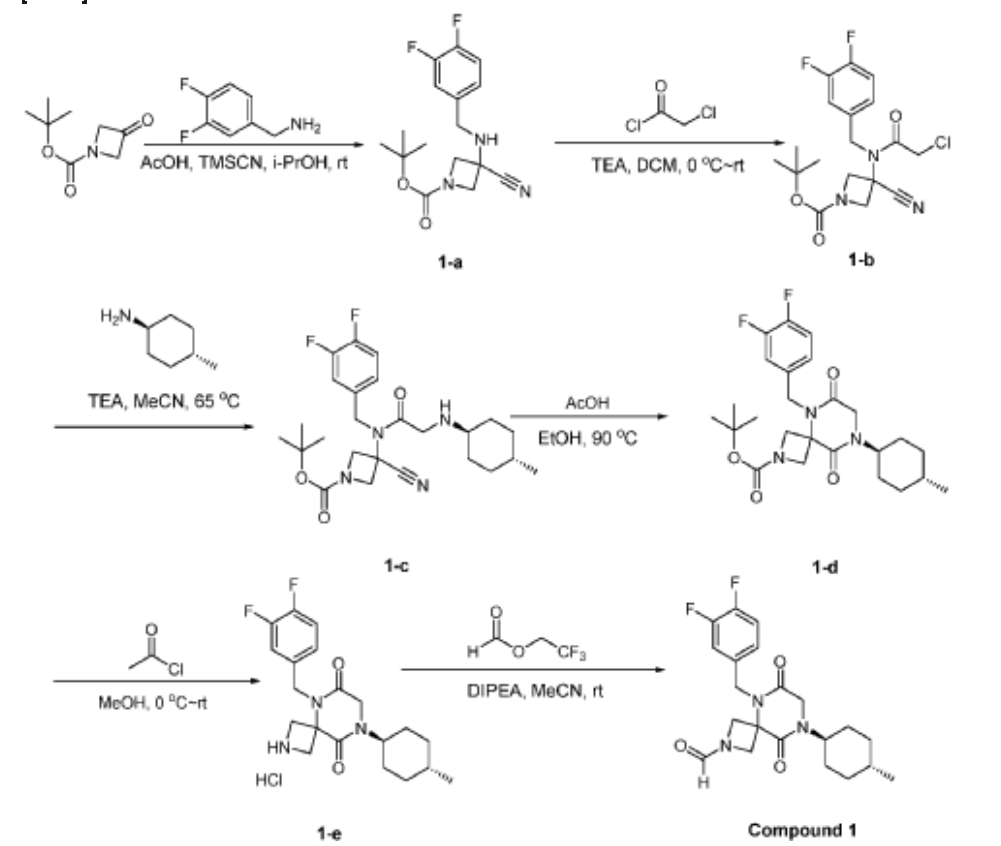
Step 6: Synthesis of 5-[(3,4-difluorophenyl)methyl]-6,9-dioxo-8-[(1r,4r)-4-methylcyclohexyl]-2,5,8-triazaspiro[3.5]nonane-2-carbaldehyde (Compound 1):
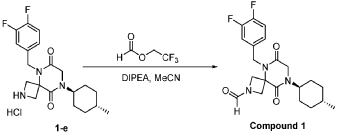
[0165] To a solution of 5-(3,4-difluorobenzyl)-8-((1r,4r)-4-methylcyclohexyl)-6,9-dioxo-2,5,8-triazaspiro[3.5]nonane-2-carbaldehyde (4.0 kg, 10.60 mol, 1 equiv) in MeCN (20 L) at r.t. were added 2,2,2-trifluoroethyl formate (1.63 kg, 12.72 mol, 1.2 equiv) and DIPEA (3.42 kg, 26.50 mol, 2.5 equiv) . The resulting mixture was stirred overnight at rt. The reaction was monitored by LCMS. The resulting mixture was concentrated under reduced pressure. The resulting mixture was diluted with EtOAc (10 L). The resulting mixture was quenched with NH 4Cl (6 L, sat.) and water (6 L), extracted with EtOAc (3×15 L). The combined organic layers were washed with NH 4Cl (aq.) (10 L) and brine (10 L), dried over anhydrous Na 2SO 4, concentrated under reduced pressure to give a crude brown oil, the crude oil was re-crystallized from cyclohexane and EtOAc (5:1, 4L, 80 °C to r.t.), filtered to afford 3 kg (1 st batch) 5-[(3,4-difluorophenyl)methyl]-6,9-dioxo-8-[(1r,4r)-4-methylcyclohexyl]-2,5,8-triazaspiro[3.5]nonane-2-carbaldehyde light yellow solid. The filtrate was concentrated under reduced pressure, re-crystallized with petroleum ether and EtOAc (10:1, 3 L, rt) to afford 800 g (2 nd batch) of light yellow solid. Two batches were combined, dried to afford 3.8 kg of Form I (m.p. at 133 °C) 5-(3,4-difluorobenzyl)-8-((1r,4r)-4-methylcyclohexyl)-6,9-dioxo-2,5,8-triazaspiro[3.5]nonane-2-carbaldehyde light yellow solid. The overall yield of this step is 97%.
PAT
- Cardiac sarcomere inhibitorsPublication Number: US-12286437-B2Priority Date: 2021-03-04Grant Date: 2025-04-29
- Cardiac sarcomere inhibitorsPublication Number: US-11919909-B2Priority Date: 2021-03-04Grant Date: 2024-03-05
- Cardiac sarcomere inhibitorsPublication Number: US-2024309011-A1Priority Date: 2021-03-04
- Cardiac sarcomere inhibitorsPublication Number: WO-2022187501-A1Priority Date: 2021-03-04
- Cardiac sarcomere inhibitorsPublication Number: US-2022306642-A1Priority Date: 2021-03-04
- cardiac sarcomere inhibitorPublication Number: CN-117083275-APriority Date: 2021-03-04
- Cardiac sarcomere inhibitorsPublication Number: EP-4301760-A1Priority Date: 2021-03-04
- Crystalline forms of 5-(3,4-difluorobenzyl)-8-((1r,4r)-4-methylcyclohexyl)-6,9-dioxo-2,5,8-triazaspiro[3.5]nonane-2-carbaldehydePublication Number: US-2024132498-A1Priority Date: 2022-09-02
- Crystalline forms of 5- (3, 4-difluorobenzyl) -8- ((1 r,4 r) -4-methylcyclohexyl) -6, 9-dioxo-2, 5, 8-triazaspiro [3.5] nonane-2-carbaldehydePublication Number: CN-119998292-APriority Date: 2022-09-02
- Crystalline forms of 5-(3,4-difluorobenzyl)-8-((1r,4r)-4-methylcyclohexyl)-6,9-dioxo-2,5,8-triazaspiro[3.5]nonane-2-carbaldehydePublication Number: WO-2024050539-A1Priority Date: 2022-09-02
- Crystalline forms of 5-(3,4-difluorobenzyl)-8-((1r,4r)-4-methylcyclohexyl)-6,9-dioxo-2,5,8-triazaspiro[3.5]nonane-2-carbaldehydePublication Number: EP-4581034-A1Priority Date: 2022-09-02
- Cardiac sarcomere inhibitorsPublication Number: TW-202302594-APriority Date: 2021-03-04
ADVERTISEMENT
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
#MedicinalChemistry, #DrugDiscovery, #OrganicSynthesis, #ChemicalLibrary, #BuildingBlocks, #SARStudies, #ChemistryInnovation, #medchem, #Drugdevelopment, #Biotech, #Biotechnology, #AnaxLaboratories, #Pharma



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

REF
Publication Name: Scientific Reports
Publication Date: 2024-05-27
PMCID: PMC11130313
PMID: 38802475
DOI: 10.1038/s41598-024-62840-3
///////////////ulacamten, ANAX, cardiac myosin inhibitor, CK-586, CK-4021586, CK 586, CK 4021586, X325G97HZJ
Suricapavir
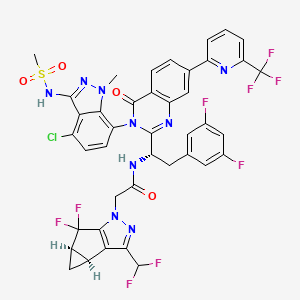
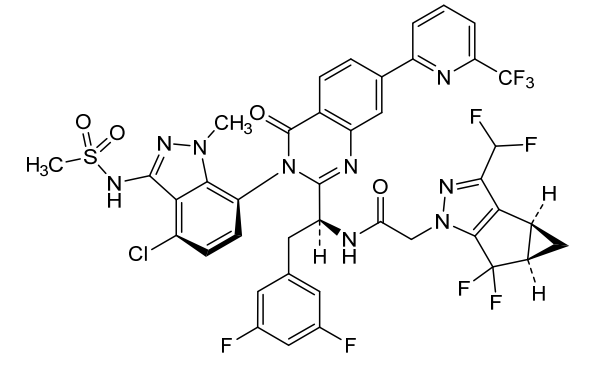
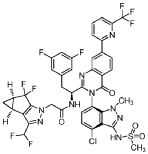
Suricapavir
CAS 2417270-21-2
MF C41H29ClF9N9O4S MF950.2 g/mol
N-[(1S)-1-[3-[4-chloro-3-(methanesulfonamido)-1-methylindazol-7-yl]-4-oxo-7-[6-(trifluoromethyl)-2-pyridinyl]quinazolin-2-yl]-2-(3,5-difluorophenyl)ethyl]-2-[(2S,4R)-9-(difluoromethyl)-5,5-difluoro-7,8-diazatricyclo[4.3.0.02,4]nona-1(6),8-dien-7-yl]acetamide
N-[(1S)-1-[3-[4-chloro-3-(methanesulfonamido)-1-methyl-indazol-7-yl]-4-oxo-7-[6-(trifluoromethyl)-2-pyridyl]quinazolin-2-yl]-2-(3,5-difluorophenyl)ethyl]-2-[(2S,4R)-9-(difluoromethyl)-5,5-difluoro-7,8-diazatricyclo[4.3.0.02,4]nona-1(6),8-dien-7-yl]acetamide
N-[(1S)-1-{(3P)-3-[4-chloro-3-(methanesulfonamido)-1-methyl-1Hindazol-7-yl]-4-oxo-7-[6-(trifluoromethyl)pyridin-2-yl]-3,4-
dihydroquinazolin-2-yl}-2-(3,5-difluorophenyl)ethyl]-2-[(3bS,4aR)-3-
(difluoromethyl)-5,5-difluoro-3b,4,4a,5-tetrahydro-1Hcyclopropa[3,4]cyclopenta[1,2-c]pyrazol-1-yl]acetamide
inhibitor of viral replication, antiviral, ZZ799EX5KN
tructurally resembles:
- Lenacapavir-type macroheterocyclic capsid inhibitors (Gilead class)
PAT
Preparation of Example 59: N-((S)-1-((3P)-3-(4-chloro-1-methyl-3-(methylsulfonamido)-1H-indazol-7-yl)-4-oxo-7-(6-(trifluoromethyl)pyridin-2-yl)-3,4-dihydroquinazolin-2-yl)-2- (3,5-difluorophenyl)ethyl)-2-((3bS,4aR)-3-(difluoromethyl)-5,5-difluoro-3b,4,4a,5- tetrahydro-1H-cyclopropa[3,4]cyclopenta[1,2-c]pyrazol-1-yl)acetamide.
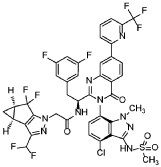
The title compound was prepared according to General Procedure D using 2-chloro-6-(trifluoromethyl)pyridine as the coupling partner. The experiment afforded the title compound, N-((S)-1-((3P)-3-(4-chloro-1-methyl-3-(methylsulfonamido)-1H-indazol-7-yl)-4-oxo-7-(6-(trifluoromethyl)pyridin-2-yl)-3,4-dihydroquinazolin-2-yl)-2-(3,5-difluorophenyl)ethyl)-2-((3bS,4aR)-3-(difluoromethyl)-5,5-difluoro-3b,4,4a,5-tetrahydro-1H-cyclopropa[3,4]cyclopenta[1,2-c]pyrazol-1-yl)acetamide. The sample was analyzed using LCMS Method F: retention time = 1.51 min.; observed ion = 948.4 (M-H).1H NMR (METHANOL-d4, 500 MHz) Shift 8.66 (s, 1H), 8.4-8.4 (m, 3H), 8.22 (t, 1H, J=7.9 Hz), 7.88 (d, 1H, J=7.7 Hz), 7.28 (br d, 1H, J=8.0 Hz), 7.20 (d, 1H, J=7.7 Hz), 6.7-6.8 (m, 1H), 6.61 (dd, 2H, J=2.2, 8.2 Hz), 6.67 (br t, 2H, J=54.7 Hz), 4.5-4.6 (m, 2H), 3.61 (s, 3H), 3.4-3.5 (m, 1H), 3.2-3.2 (m, 3H), 3.1-3.2 (m, 1H), 2.41 (br dd, 2H, J=3.7, 7.3 Hz), 1.34 (br d, 1H, J=5.4 Hz), 0.99 (br dd, 1H, J=1.9, 3.7 Hz)
PAT
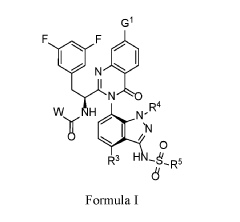
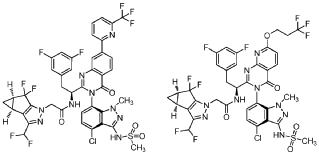
WO 2020/084492 and WO 2020/254985 disclose certain Capsid Inhibitor compounds including the two compounds shown below which will be referred to in this application as the compounds of Formula la and Formula lb.
PAT
- Inhibitors of human immunodeficiency virus replicationPublication Number: WO-2023062559-A1Priority Date: 2021-10-13
- Inhibitors of human immunodeficiency virus replicationPublication Number: US-2023149408-A1Priority Date: 2020-04-15
- Inhibitors of human immunodeficiency virus replicationPublication Number: WO-2021209900-A1Priority Date: 2020-04-15
- Pharmaceutical compositions comprising cabotegravirPublication Number: US-2023045509-A1Priority Date: 2019-12-09
- Inhibitors of human immunodeficiency virus replicationPublication Number: US-11541055-B2Priority Date: 2018-10-24Grant Date: 2023-01-03
- Inhibitors of human immunodeficiency virus replicationPublication Number: US-2020360384-A1Priority Date: 2018-10-24
- Inhibitors of human immunodeficiency virus replicationPublication Number: EP-3870577-B1Priority Date: 2018-10-24Grant Date: 2025-03-19
ADVERTISEMENT
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
#MedicinalChemistry, #DrugDiscovery, #OrganicSynthesis, #ChemicalLibrary, #BuildingBlocks, #SARStudies, #ChemistryInnovation, #medchem, #Drugdevelopment, #Biotech, #Biotechnology, #AnaxLaboratories, #Pharma



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

///////////////suricapavir, ANAX, inhibitor of viral replication, antiviral, ZZ799EX5KN
Soxataltinib
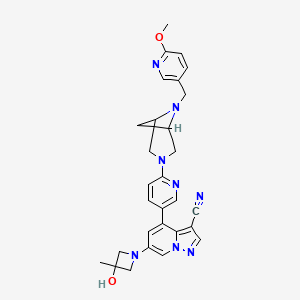
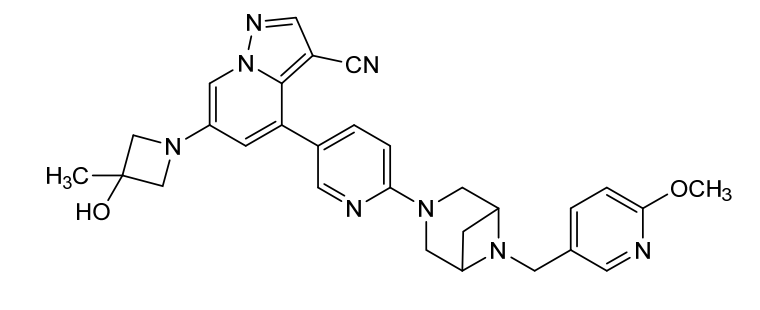
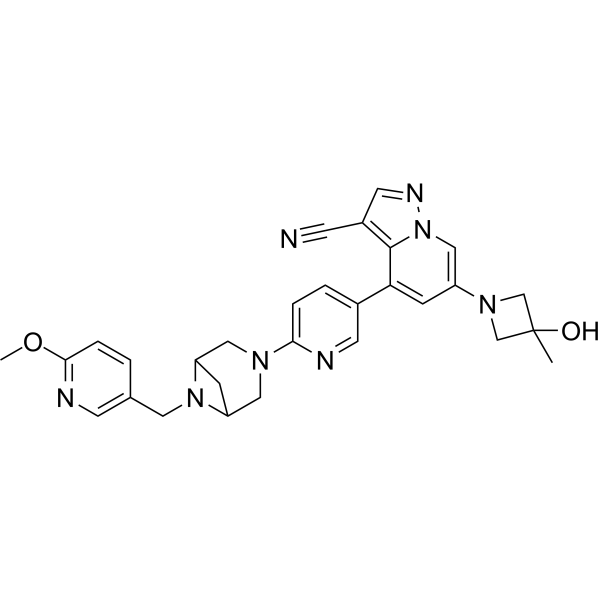
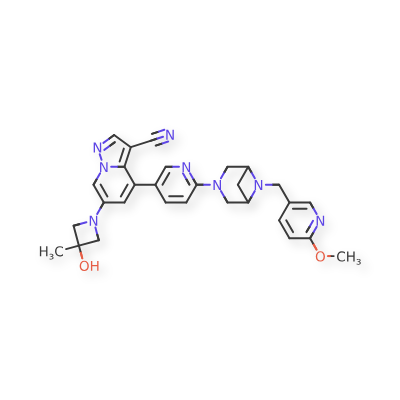
Soxataltinib
CAS 2546116-88-3
MF C29H30N8O2 MW 522.60
6-(3-hydroxy-3-methylazetidin-1-yl)-4-(6-{6-[(6-methoxypyridin-3-yl)methyl]-3,6-diazabicyclo[3.1.1]heptan-3-yl}pyridin-3-yl)pyrazolo[1,5-a]pyridine-3-carbonitrile
| Pyrazolo[1,5-a]pyridine-3-carbonitrile, 6-(3-hydroxy-3-methyl-1-azetidinyl)-4-[6-[6-[(6-methoxy-3-pyridinyl)methyl]-3,6-diazabicyclo[3.1.1]hept-3-yl]-3-pyridinyl]- |
6-(3-hydroxy-3-methylazetidin-1-yl)-4-(6-{6-[(6-methoxypyridin-3-yl)methyl]-3,6-diazabicyclo[3.1.1]heptan-3-yl}pyridin-3-yl)pyrazolo[1,5-a]pyridine-3-carbonitrile
RET-kinase inhibitor, antineoplastic, HS-10365, HS 10365, AZ4Q643U3D
Soxataltinib (example 7) is a potent inhibitor of RET-kinase , with the IC 50of 0.601 nM. Soxataltinib plays an important role in
cancer research.
Discovery and Development
- Soxataltinib corresponds to Example 114 in a patent [WO2020228756]describing pyrazolo[1,5-a]pyridine carbonitrile RET inhibitors.
- It is believed to correspond to HS-10365, a RET inhibitor developed by Hansoh Pharma (structure disclosed via patent).
Drug class comparison:
| Drug | Company | Type |
|---|---|---|
| Selpercatinib | Eli Lilly | 1st-gen selective RET inhibitor |
| Pralsetinib | Blueprint | selective RET inhibitor |
| Soxataltinib | Hansoh | next-gen RET inhibitor |
Patent Family (Major Members)
Typical family members include:
| Patent | Jurisdiction |
|---|---|
| WO2020228756 | WIPO |
| CN112209925 | China |
| US continuation filings | USA |
| EP equivalents | Europe |
One Chinese patent describes the preparation of piperazine-containing pyrazolopyridine RET inhibitors, including Soxataltinib analogues
SYN
SYN
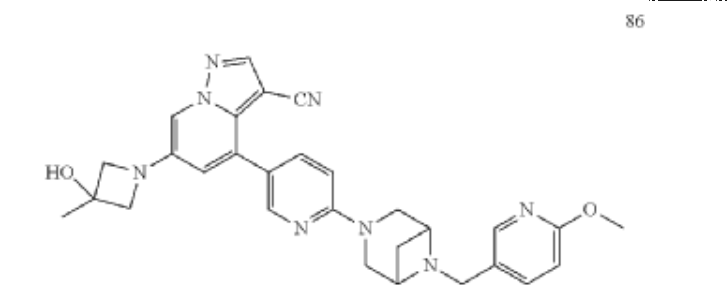
Example 32
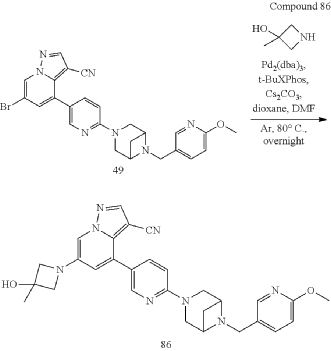
| To a 25 mL sealed tube were added successively 49 (52 mg, 0.1 mmol), Pd 2(dba) 3 (5.5 mg, 0.006 mmol), t-BuXPhos (8.4 mg, 0.02 mmol), 3-methyl-3-azetidinol (26 mg, 0.3 mmol), Cs 2CO 3 (65 mg, 0.2 mmol), 1,4-dioxane (3 mL) and DMF (1 mL). The mixture was stirred at 80° C. overnight under Ar, and TLC monitoring showed no starting material 49 remained. The mixture was cooled to room temperature, and 10 mL of water was added. The mixture was stirred for 10 min, and a yellow solid precipitated. The solid was collected by filtration, dried and purified by column chromatography to give product 86 (34 mg, 65% yield). |
SYN
[1913]6-(3-hydroxy-3-methylacetidin-1-yl)-4-(6-(6-((6-methoxypyridin-3-yl)methyl)-3,6-diazabicyclo[3.1.1]heptane-3-yl)pyridin-3-yl)pyrazolo[1,5-a]pyridin-3-carboxynitrile
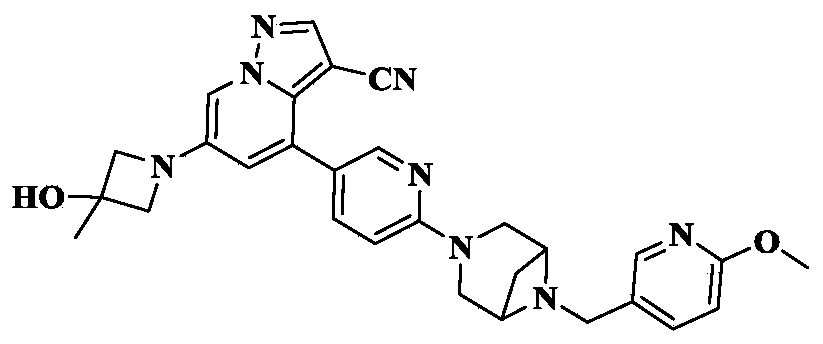
Using 3-methylacetidin-3-ol as a raw material, in the first step of Reference Example 110, 6-(3-hydroxy-3-methylacetidin-1-yl)-4-(6-(6-((6-methoxypyridin-3-yl)methyl)-3,6-diazabicyclo[3.1.1]heptane-3-yl)pyridin-3-yl)pyrazolo[1,5-a]pyridin-3-carboxynitrile was obtained.
[1916]MS m/z(ESI):523.3[M+H]
[1917]
1H NMR(400MHz,CDCl 3)δ8.40(s,1H),8.15(s,3H),7.82(d,J=7.4Hz,1H),7.74(s,1H),6.80(d,J=8.4Hz,1H),6.75(d,J=1.7Hz,1H),6.72(d,J=8.8Hz,1H),4.21(s,2H),4.01(s,2H),3.93-3.92(m,7H),3.84(d,J=7.3Hz,4H),1.68(s,3H).
PAT
- Inhibitor containing dihecyclic derivatives, its preparation method and applicationPublication Number: CN-112368283-BPriority Date: 2019-05-14Grant Date: 2023-02-17
- Inhibitor containing dihecyclic derivatives, its preparation method and applicationPublication Number: CN-115974897-APriority Date: 2019-05-14
- Inhibitor containing dihecyclic derivatives, its preparation method and applicationPublication Number: CN-116444515-APriority Date: 2019-05-14
- Inhibitor containing bicyclic derivative, preparation method therefor and use thereofPublication Number: WO-2020228756-A1Priority Date: 2019-05-14
- Inhibitor containing bicyclic derivative, preparation method therefor and use thereofPublication Number: EP-3971187-B1Priority Date: 2019-05-14Grant Date: 2025-05-07
- Ret selective inhibitor, preparation method therefor and use thereofPublication Number: WO-2021008455-A1Priority Date: 2019-07-12
- RET selective inhibitor and preparation method and use thereofPublication Number: CN-112209925-APriority Date: 2019-07-12
- RET selective inhibitors and methods for their preparation and usePublication Number: KR-102735956-B1Priority Date: 2019-07-12Grant Date: 2024-11-28
- RET selective inhibitors and preparation methods and uses thereofPublication Number: CN-114072404-BPriority Date: 2019-07-12Grant Date: 2023-09-15
- Inhibitor containing bicyclic derivative, preparation method therefor and use thereofPublication Number: US-2022259201-A1Priority Date: 2019-05-14
- Heteroaromatic ring compound as ret kinase inhibitor, and preparation and use thereofPublication Number: US-2023322769-A1Priority Date: 2020-08-20
- 3,6 diazabicyclo[3.1.1]heptane derivatives as RET kinase inhibitorsPublication Number: CN-112778337-BPriority Date: 2019-11-08Grant Date: 2023-09-26
- Ret selective inhibitor, preparation method therefor and use thereofPublication Number: EP-3998265-A1Priority Date: 2019-07-12
- RET selective inhibitor and preparation method and use thereofPublication Number: CN-117865952-APriority Date: 2019-07-12
- Ret selective inhibitor, preparation method therefor and use thereofPublication Number: US-2022289740-A1Priority Date: 2019-07-12
- Crystal form of free base of inhibitor containing bicyclic ring derivative and preparation method and application of crystal formPublication Number: US-2023406865-A1Priority Date: 2020-11-13
- Crystal form of free base of inhibitor containing bicyclic ring derivative and preparation method and application of crystal formPublication Number: EP-4245757-A1Priority Date: 2020-11-13
- Crystal form of free base of inhibitor containing bicyclic ring derivative and preparation method and application of crystal formPublication Number: WO-2022100738-A1Priority Date: 2020-11-13
- Crystal form of inhibitor free base including bicyclic derivatives, preparation method and use thereofPublication Number: KR-20230107271-APriority Date: 2020-11-13
- Heteroaromatic ring compound as ret kinase inhibitor, and preparation and use thereofPublication Number: EP-4201936-A1Priority Date: 2020-08-20
ADVERTISEMENT
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
#MedicinalChemistry, #DrugDiscovery, #OrganicSynthesis, #ChemicalLibrary, #BuildingBlocks, #SARStudies, #ChemistryInnovation, #medchem, #Drugdevelopment, #Biotech, #Biotechnology, #AnaxLaboratories, #Pharma



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

/////////soxataltinib, ANAX, RET-kinase inhibitor, antineoplastic, HS-10365, HS 10365, AZ4Q643U3D
ADVERTISEMENT
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
#MedicinalChemistry, #DrugDiscovery, #OrganicSynthesis, #ChemicalLibrary, #BuildingBlocks, #SARStudies, #ChemistryInnovation, #medchem, #Drugdevelopment, #Biotech, #Biotechnology, #AnaxLaboratories, #Pharma
Rosolutamide
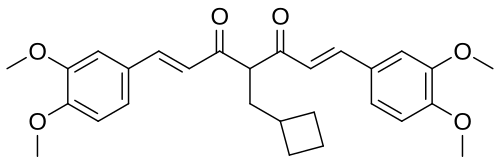
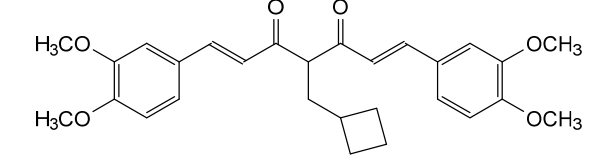
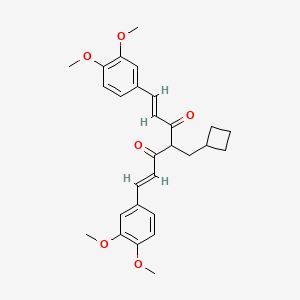
Rosolutamide
CAS 1039760-91-2
MF C28H32O6 MW464.5 g/mol
(1E,6E)-4-(cyclobutylmethyl)-1,7-bis(3,4-dimethoxyphenyl)hepta-1,6-diene-3,5-dione
(1E,6E)-4-(cyclobutylmethyl)-1,7-bis(3,4-dimethoxyphenyl)hepta-1,6-diene-3,5-dione
antiandrogen, ASC-JM-17, ASC-JM17, JM17, ALZ-003, ALZ003, 5VLL140BN9,
Rosolutamide (INNTooltip International Nonproprietary Name; developmental code name ASC-JM17, JM17, ALZ-003) is an agonist of nuclear respiratory factor 1 (NRF1), a nonsteroidal antiandrogen, and an androgen receptor degrader related to curcumin.[1][2][3][4][5] Other analogues like dimethylcurcumin (ASC-J9) are also known.[2][6]
3-hydroxy imidacloprid is an imidacloprid. It has a role as a neonicotinoid insectide and a nicotinic acetylcholine receptor agonist.
REF
- Ferroptosis-related small-molecule compounds in cancer therapy: Strategies and applicationsPublication Name: European Journal of Medicinal ChemistryPublication Date: 2022-12-15PMID: 36332549DOI: 10.1016/j.ejmech.2022.114861
- A small-molecule Nrf1 and Nrf2 activator mitigates polyglutamine toxicity in spinal and bulbar muscular atrophyPublication Name: Human Molecular GeneticsPublication Date: 2016-03-08PMCID: PMC5062587PMID: 26962150DOI: 10.1093/hmg/ddw073
SYN
SYN
SYN
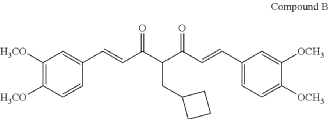
COMPD B
SYN
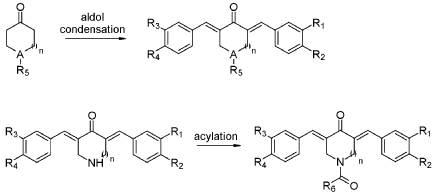
SYN
PAT
Publication Number: EP-2104659-B1
Priority Date: 2007-01-08
Grant Date: 2015-07-29
- Compounds with (1e, 6e)-1,7-bis-(3,4-dimethoxyphenyl)-4-4-disubstituted-hepta-1,6-diene-3,5-dione structural scaffold, their biological activity, and uses thereofPublication Number: US-2016264539-A1Priority Date: 2007-01-08
- Compounds with (substituted phenyl)-propenal moiety, their derivatives, biological activity, and uses thereofPublication Number: DK-2104659-T3Priority Date: 2007-01-08Grant Date: 2015-11-02
- Compounds with (1E, 6E)-1,7-Bis-(3,4-dimethoxyphenyl)-4-4-distributed-hepta-1,6-diene-3,5-dione structural scaffold, their biological activity, and uses thereofPublication Number: US-9562025-B2Priority Date: 2007-01-08Grant Date: 2017-02-07
- Compounds with (1E, 6E)-1,7-bis-(3,4-dimethoxyphenyl)-4,4-disubstituted-hepta-1,6-diene-3,5-dione structural scaffold, their biological activity, and uses thereofPublication Number: US-9000222-B2Priority Date: 2007-01-08Grant Date: 2015-04-07
- Compounds with (1 E, 6E)-1,7-bis-(3,4-dimethoxyphenyI)-4,4-disubstituted-hepta-1,6-diene-3,5-dione structural scaffold, their biological activity, and uses thereofPublication Number: US-9259402-B2Priority Date: 2007-01-08Grant Date: 2016-02-16
- Compounds with (1 E, 6E)-1,7-bis-(3,4-dimethoxyphenyl)-4,4-disubstituted-hepta-1,6-diene-3,5-dione structural scaffold, their biological activity, and uses thereofPublication Number: US-8710272-B2Priority Date: 2007-01-08Grant Date: 2014-04-29
- Compounds with (substituted phenyl)-propenal moiety, their derivatives, biological activity, and use thereofPublication Number: EP-2993165-A2Priority Date: 2007-01-08
- Compounds with (substituted phenyl)-propenal moiety, their derivatives, biological activity, and use thereofPublication Number: EP-2993165-B1Priority Date: 2007-01-08Grant Date: 2018-06-27
- Compounds with (1E, 6E)-1,7-bis-(3,4-dimethoxyphenyl)-4,4-disubstituted-hepta-1,6-diene-3,5-dione structural scaffold, their biological activity, and uses thereofPublication Number: US-2013261121-A1Priority Date: 2007-01-08
- Compounds with (substituted phenyl)-propenal moiety, their derivatives, biological activity, and uses thereofPublication Number: CA-2674780-CPriority Date: 2007-01-08Grant Date: 2014-03-11
- Compositions including androgen receptor degradation (ard) enhancers and methods of prophylactic or therapeutic treatment of skin disorders and hair lossPublication Number: CA-2694953-CPriority Date: 2007-07-31Grant Date: 2015-12-01
- Compositions containing androgen receptor degradation enhancers and methods for preventing or treating hair loss and skin diseasesPublication Number: KR-20100051808-APriority Date: 2007-07-31
- Compositions containing androgen receptor degradation (ARD) enhancers and methods for the prophylactic or therapeutic treatment of skin diseases and hair lossPublication Number: JP-2010535213-APriority Date: 2007-07-31
- Compounds with (1E, 6E)-1,7-bis-(3,4-dimethoxyphenyl)-4,4-disubstituted-hepta-1,6-diene-3,5-dione structural scaffold, their biological activity, and uses thereofPublication Number: US-2013338160-A1Priority Date: 2007-01-08
- Compounds with (1E, 6E)-1,7-bis-(3,4-dimethoxyphenyl)-4,4-disubstituted-hepta-1,6-diene-3,5-dione structural scaffold, their biological activity, and uses thereofPublication Number: US-2015190351-A1Priority Date: 2007-01-08
ADVERTISEMENT
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
#MedicinalChemistry, #DrugDiscovery, #OrganicSynthesis, #ChemicalLibrary, #BuildingBlocks, #SARStudies, #ChemistryInnovation, #medchem, #Drugdevelopment, #Biotech, #Biotechnology, #AnaxLaboratories, #Pharma



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

| Clinical data | |
|---|---|
| Other names | ASC-JM-17; ASC-JM17; JM17; ALZ-003; ALZ003 |
| Identifiers | |
| IUPAC name | |
| CAS Number | 1039760-91-2 |
| PubChem CID | 25183127 |
| DrugBank | DB16931 |
| ChemSpider | 64854816 |
| UNII | 5VLL140BN9 |
| ChEMBL | ChEMBL5266600 |
| Chemical and physical data | |
| Formula | C28H32O6 |
| Molar mass | 464.558 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| SMILES | |
| InChI | |
References
- “Proposed INN: List 131 International Nonproprietary Names for Pharmaceutical Substances (INN)” (PDF). WHO Drug Information. 38 (2): 428. 2024.
- Chang KH, Chen CM (May 2024). “The Role of NRF2 in Trinucleotide Repeat Expansion Disorders”. Antioxidants. 13 (6): 649. doi:10.3390/antiox13060649. PMC 11200942. PMID 38929088.
- Yuan J, Zhang S, Zhang Y (December 2018). “Nrf1 is paved as a new strategic avenue to prevent and treat cancer, neurodegenerative and other diseases”. Toxicology and Applied Pharmacology. 360: 273–283. Bibcode:2018ToxAP.360..273Y. doi:10.1016/j.taap.2018.09.037. PMID 30267745.
- Bott LC, Badders NM, Chen KL, Harmison GG, Bautista E, Shih CC, et al. (May 2016). “A small-molecule Nrf1 and Nrf2 activator mitigates polyglutamine toxicity in spinal and bulbar muscular atrophy”. Human Molecular Genetics. 25 (10): 1979–1989. doi:10.1093/hmg/ddw073. PMC 5062587. PMID 26962150.
- Wu YL, Chang JC, Chao YC, Chan H, Hsieh M, Liu CS (July 2022). “In Vitro Efficacy and Molecular Mechanism of Curcumin Analog in Pathological Regulation of Spinocerebellar Ataxia Type 3”. Antioxidants. 11 (7): 1389. doi:10.3390/antiox11071389. PMC 9311745. PMID 35883884.
- Sangotra A, Lieberman AP (February 2025). “Therapeutic targeting of the polyglutamine androgen receptor in Spinal and Bulbar Muscular Atrophy”. Expert Opinion on Therapeutic Targets: 1–13. doi:10.1080/14728222.2025.2464173. PMID 39915972.
/////////rosolutamide, antiandrogen, ASC-JM-17, ASC-JM17, JM17, ALZ-003, ALZ003, 5VLL140BN9, ANAX
Repinatrabit
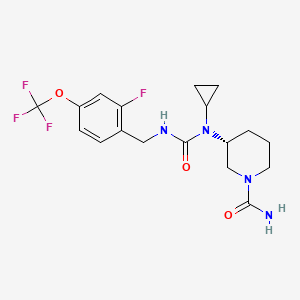
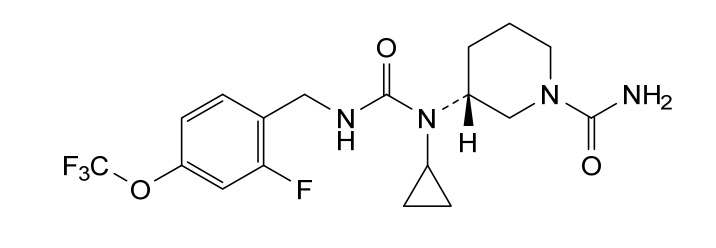
Repinatrabit
CAS 2837993-05-0
MF C18H22F4N4O3 MW 418.4 g/mol
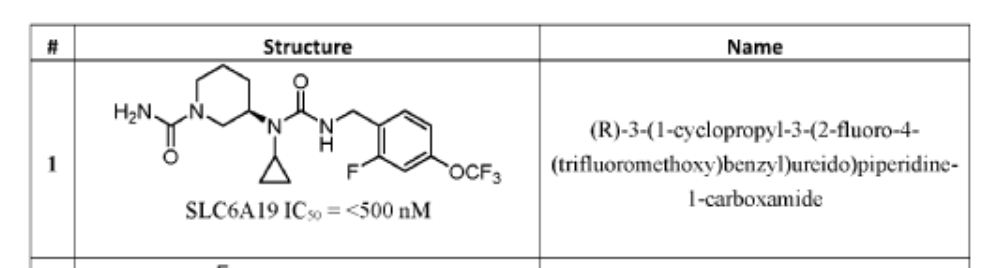
(3R)-3-[cyclopropyl-[[2-fluoro-4-(trifluoromethoxy)phenyl]methylcarbamoyl]amino]piperidine-1-carboxamide
(3R)-3-[cyclopropyl({[2-fluoro-4-(trifluoromethoxy)phenyl]methyl}carbamoyl)amino]piperidine-1-
carboxamide
solute carrier family 6 member 19 (SLC6A19) inhibitor(phenylketonuria), JNT-517, JNT 517, orphan drug, rare pediatric disease designations, Jnana Therapeutics, 5P44NDU6AC, JN 11804, JN-11804
Repinatrabit (JNT-517) is an investigational, oral, small-molecule drug developed by Jnana Therapeutics (now part of Otsuka Pharmaceutical) to treat Phenylketonuria (PKU). It acts as a selective inhibitor of the SLC6A19 transporter, reducing blood phenylalanine (Phe) levels by increasing its urinary excretion.
Key Details About Repinatrabit:
- Mechanism: It targets a novel, cryptic allosteric site to block kidney reabsorption of phenylalanine, aiming to be a first-in-class oral therapy for all PKU patients, regardless of age or genotype.
- Clinical Trials: Otsuka initiated a global Phase 3 study (NCT06971731) in December 2025 to evaluate its safety and efficacy, following positive results from earlier studies.
- Status: The FDA has granted it orphan drug and rare pediatric disease designations.
- A Study to Evaluate the Safety and Efficacy of JNT-517 in Participants With Phenylketonuria (PKU)CTID: NCT06971731Phase: Phase 3Status: RecruitingDate: 2026-02-04
- A Phase 2 Study of JNT-517 in Adolescent Participants With PhenylketonuriaCTID: NCT06637514Phase: Phase 2Status: RecruitingDate: 2025-08-19
- First-in-Human, Multiple Part Clinical Study of JNT-517 in Healthy Participants and in Participants With PhenylketonuriaCTID: NCT05781399Phase: Phase 1/Phase 2Status: Active, not recruitingDate: 2025-07-31
- A Study to Evaluate the Long-Term Safety and Efficacy of JNT-517 in Participants With PhenylketonuriaCTID: NCT06628128Phase: Phase 3Status: Not yet recruitingDate: 2025-06-03
EMA Drug Information,
Type, Orphan designations
(R)-3-(1-Cyclopropyl-3-(2-fluoro-4-(trifluoromethoxy)benzyl)ureido)piperidine-1-carboxamide
Intended Use, Treatment of hyperphenylalaninaemia, Status of Orphan Designation, Positive
First Published Date, 2024-08-22
PAT
SYN
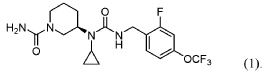
PAT
PAT
- Crystalline forms of a piperidine inhibitor of slc6a19 functionPublication Number: WO-2024118721-A1Priority Date: 2022-11-30
- Crystalline forms of a piperidine inhibitor of slc6a19 functionPublication Number: EP-4626420-A1Priority Date: 2022-11-30
- Dosing regimen for treatment of PKU with SLC6A19 functional piperidine inhibitorsPublication Number: CN-120091814-APriority Date: 2022-09-14
- Small molecule inhibitors of mammalian slc6a19 functionPublication Number: US-2024208923-A1Priority Date: 2021-03-10
- Small molecule inhibitors of mammalian slc6a19 functionPublication Number: US-2025289797-A1Priority Date: 2021-03-10
ADVERTISEMENT
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
#MedicinalChemistry, #DrugDiscovery, #OrganicSynthesis, #ChemicalLibrary, #BuildingBlocks, #SARStudies, #ChemistryInnovation, #medchem, #Drugdevelopment, #Biotech, #Biotechnology, #AnaxLaboratories, #Pharma



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

///////////repinatrabit, ANAX, JNT-517, JNT 517, orphan drug, rare pediatric disease designations, Jnana Therapeutics, 5P44NDU6AC, JN 11804, JN-11804
Remlifanserin
Remlifanserin
CAS 2289704-13-6
MF C24H29F2N3O2 MW 429.5 g/mol
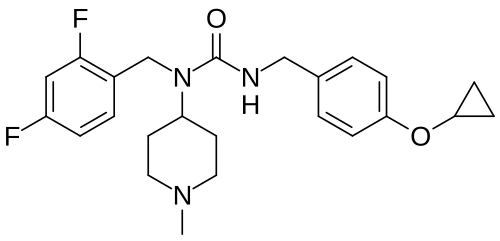
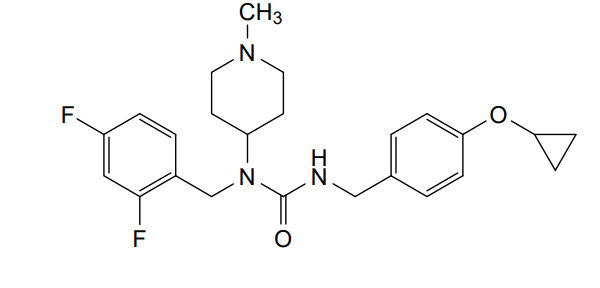
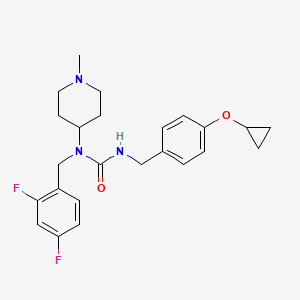
3-[(4-cyclopropyloxyphenyl)methyl]-1-[(2,4-difluorophenyl)methyl]-1-(1-methylpiperidin-4-yl)urea
N’-{[4-(cyclopropyloxy)phenyl]methyl}-N-[(2,4-difluorophenyl)methyl]-N-(1-methylpiperidin-4-yl)urea
serotonin receptor (5-HT2A) inverse agonist, ACP-204, ACP 204, H4L2AF2XB7
Remlifanserin is a small molecule drug. The usage of the INN stem ‘-anserin’ in the name indicates that Remlifanserin is a serotonin receptor antagonist. Remlifanserin has a monoisotopic molecular weight of 429.22 Da.
Remlifanserin (INNTooltip International Nonproprietary Name;[4] developmental code name ACP-204) is a selective serotonin 5-HT2A receptor inverse agonist which is under development for the treatment of Alzheimer’s disease psychosis.[1][5][6][7][8][9] It is taken by mouth.[1]
The drug is an improved follow-up compound to its developer’s earlier drug pimavanserin (Nuplaizid; ACP-103).[6] It is more potent and selective than pimavanserin as a serotonin 5-HT2A receptor inverse agonist.[10] Remlifanserin shows 32- to 123-fold selectivity for antagonism and inverse agonism of the serotonin 5-HT2A receptor over the serotonin 5-HT2C receptor depending on the bioassay.[10] For comparison, pimavanserin’s selectivity was 8- to 37-fold depending on the assay.[10] Remlifanserin shows very low affinity for the serotonin 5-HT2B receptor compared to the serotonin 5-HT2A and 5-HT2C receptors.[10] It is expected to have less QT prolongation than pimavanserin.[10] The drug blocks the head-twitch response induced by the serotonergic psychedelic DOI and the hyperlocomotion induced by the NMDA receptor antagonist dizocilpine (MK-801) in rodents.[10]
Remlifanserin is under development by Acadia Pharmaceuticals.[1][5] As of January 2025, it is in phase 3 clinical trials.[1][5] Its clinicaltrials.gov identifier (nct number) is NCT06159673.[11]
SYN
Example 17: 3-[(4-cyclopropoxyphenyl)methyl]-1-[(2,4-difluorophenyl)methyl]-1-(1-methylpiperidin-4-yl)urea; hemitartrate (17)
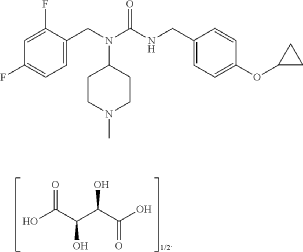
3-[(4-cyclopropoxyphenyl)methyl]-1-[(2,4-difluorophenyl)methyl]-1-(1-methylpiperidin-4-yl)urea; hemitartrate
PAT
- 3-(4-cyclo-propoxybenzyl)-1-(2,4-difluorobenzyl)-1 -(1-methylpiperidin-4-yl)urea for use in the treatment of diseases associated with the serotonin-receptor 5-htPublication Number: WO-2025029990-A1Priority Date: 2023-08-02
- Compounds, salts thereof and methods for treating diseasesPublication Number: CN-111132976-APriority Date: 2017-08-21
- COMPOUNDS, CORRESPONDING SALTS AND METHODS FOR THE TREATMENT OF DISEASESPublication Number: WO-2019040107-A1Priority Date: 2017-08-21
- Compounds, salts thereof and methods for treatment of diseasesPublication Number: EP-3672954-A1Priority Date: 2017-08-21
- Compounds, salts thereof and methods for treatment of diseasesPublication Number: US-2020270239-A1Priority Date: 2017-08-21
- Compounds, salts thereof and methods for treating diseasesPublication Number: CN-111132976-BPriority Date: 2017-08-21Grant Date: 2023-08-22
- Compounds, salts thereof and methods for treatment of diseasesPublication Number: EP-4635568-A2Priority Date: 2017-08-21
- Compounds, salts thereof and methods for treatment of diseasesPublication Number: US-12139477-B2Priority Date: 2017-08-21Grant Date: 2024-11-12
- Compounds, salts thereof and methods for treatment of diseasesPublication Number: US-2025197385-A1Priority Date: 2017-08-21
- Compounds, salts thereof and their use for the treatment of diseasesPublication Number: EP-3672954-B1Priority Date: 2017-08-21Grant Date: 2025-08-13
- Compounds, salts thereof and methods for treatment of diseasesPublication Number: US-11345693-B2Priority Date: 2017-08-21Grant Date: 2022-05-31
- Compounds, salts thereof and methods for treatment of diseasesPublication Number: US-2022298151-A1Priority Date: 2017-08-21
- Compounds, salts thereof and methods for treating diseasesPublication Number: CN-117466803-APriority Date: 2017-08-21
ADVERTISEMENT
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
#MedicinalChemistry, #DrugDiscovery, #OrganicSynthesis, #ChemicalLibrary, #BuildingBlocks, #SARStudies, #ChemistryInnovation, #medchem, #Drugdevelopment, #Biotech, #Biotechnology, #AnaxLaboratories, #Pharma



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

ADVERTISEMENT
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
#MedicinalChemistry, #DrugDiscovery, #OrganicSynthesis, #ChemicalLibrary, #BuildingBlocks, #SARStudies, #ChemistryInnovation, #medchem, #Drugdevelopment, #Biotech, #Biotechnology, #AnaxLaboratories, #Pharma
| Clinical data | |
|---|---|
| Other names | ACP-204; ACP204 |
| Routes of administration | Oral[1] |
| Drug class | Serotonin 5-HT2A receptor inverse agonist |
| Pharmacokinetic data | |
| Onset of action | 4–6 hours (6 hours fasted, 9 hours fed) (TmaxTooltip time to peak levels)[2][3] |
| Elimination half-life | 17.8–19.8 hours[2] |
| Identifiers | |
| IUPAC name | |
| CAS Number | 2289704-13-6 |
| PubChem CID | 137520242 |
| UNII | H4L2AF2XB7 |
| Chemical and physical data | |
| Formula | C24H29F2N3O2 |
| Molar mass | 429.512 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| SMILES | |
| InChI | |
References
- “ACP 204”. AdisInsight. 23 January 2025. Retrieved 22 February 2025.
- Darwish M, Feng X, Dirks B, Raether B, Pathak SS (2025). “Pharmacokinetics in Healthy Adult and Elderly Patients of ACP-204, a Novel 5-HT 2A Receptor Selective Antagonist/Inverse Agonist”. Alzheimer’s & Dementia. 21 (S5) e105732. doi:10.1002/alz70859_105732. ISSN 1552-5260. PMC 12741707.
- Darwish M, Dirks B, Feng X, Raether B, Pathak SS (2025). “Effect of Food Consumption on the Pharmacokinetics of ACP-204, a Novel 5-HT 2A Receptor Selective Antagonist/Inverse Agonist”. Alzheimer’s & Dementia. 21 (S5) e105644. doi:10.1002/alz70859_105644. ISSN 1552-5260. PMC 12741626.
- “Proposed INN: List 131 International Nonproprietary Names for Pharmaceutical Substances (INN)” (PDF). WHO Drug Information. 38 (2): 421. 2024.
remlifanserin N’-{[4-(cyclopropyloxy)phenyl]methyl}-N-[(2,4- difluorophenyl)methyl]-N-(1-methylpiperidin-4-yl)urea serotonin receptor (5-HT2A) inverse agonist […] C24H29F2N3O2 2289704-13-6 […]
- “Delving into the Latest Updates on ACP-204 with Synapse”. Synapse. 4 February 2025. Retrieved 22 February 2025.
- “ACP-204”. ALZFORUM. 5 February 2024. Retrieved 22 February 2025.
- Imbimbo C, Cotta Ramusino M, Leone S, Mazzacane F, De Franco V, Gatti A, et al. (February 2025). “Emerging Pharmacological Approaches for Psychosis and Agitation in Alzheimer’s Disease”. CNS Drugs. 39 (2): 143–160. doi:10.1007/s40263-024-01133-9. PMC 11769872. PMID 39623197.
- IsHak WW, Meyer A, Freire L, Totlani J, Murphy N, Renteria S, et al. (2024). “Overview of Psychiatric Medications in the Pipeline in Phase III Trials as of June 1, 2024: A Systematic Review”. Innovations in Clinical Neuroscience. 21 (7–9): 27–47. PMC 11424068. PMID 39329027.
- Kwon KJ, Kim HY, Han SH, Shin CY (October 2024). “Future Therapeutic Strategies for Alzheimer’s Disease: Focus on Behavioral and Psychological Symptoms”. International Journal of Molecular Sciences. 25 (21) 11338. doi:10.3390/ijms252111338. PMC 11547068. PMID 39518892.
- Burstein E, Markus Dey P, Pathak S (December 2024). “ACNP 63rd Annual Meeting: Poster Abstracts P305-P608: P497. Nonclinical Characterization of ACP-204, a Novel Second Generation 5-HT2A Inverse Agonist” (PDF). Neuropsychopharmacology. 49 (Suppl 1): 236–417 (346–347). doi:10.1038/s41386-024-02012-z. PMID 39643634.
- ACADIA Pharmaceuticals Inc. (2025-02-21). A Master Protocol for Three Independent, Seamlessly Enrolling, Double-blind, Placebo-controlled Efficacy and Safety Studies of ACP-204 in Adults with Alzheimer’s Disease Psychosis (Report). clinicaltrials.gov.
////////////remlifanserin, ANAX, serotonin receptor (5-HT2A) inverse agonist, ACP-204, ACP 204, H4L2AF2XB7
Luvometinib
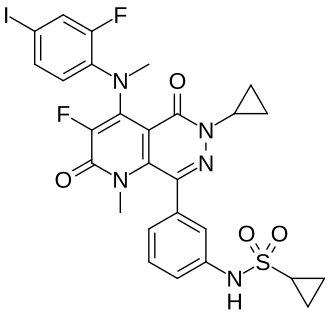
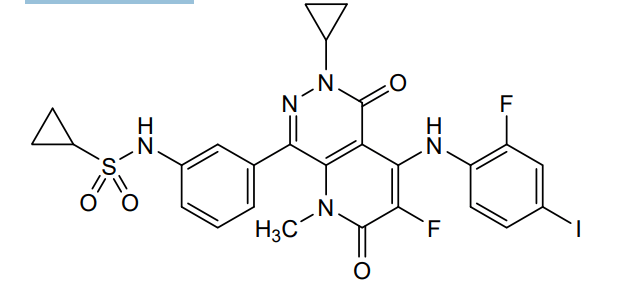
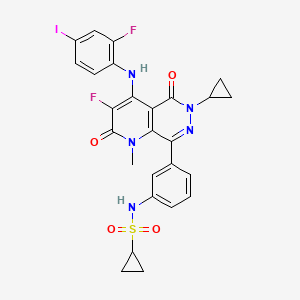
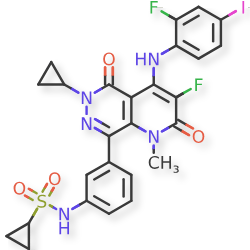
Luvometinib
CAS 2739690-43-6
MF C26H22F2IN5O4S MW665.5 g/mol
CHINA 2025, APPROVALS 2025
N-[3-[6-cyclopropyl-3-fluoro-4-(2-fluoro-4-iodoanilino)-1-methyl-2,5-dioxopyrido[2,3-d]pyridazin-8-yl]phenyl]cyclopropanesulfonamide
N-{3-[6-cyclopropyl-3-fluoro-4-(2-fluoro-4-iodoanilino)-1-methyl-2,5-dioxo-1,2,5,6-tetrahydropyrido[2,3-
d]pyridazin-8-yl]phenyl}cyclopropanesulfonamide
mitogen-activated protein kinase (MEK) inhibitor, antineoplastic, FCN 159, FCN-159, B2DYT4V89X
Luvometinib is a drug for the treatment of various types of cancer. It is a selective, orally administered inhibitor of mitogen-activated protein kinase kinases 1 and 2 (MEK1/MEK2), developed by Fosun Pharma for the treatment of rare malignancies, especially those driven by abnormal abnormal mitogen-activated protein kinase (MAPK) activation.[1][2]
In May 2025, it was approved in China for the treatment of histiocytic neoplasms such as Langerhans cell histiocytosis (LCH) and the genetic disease neurofibromatosis type 1 (NF1).[2]
Luvometinib is an orally bioavailable inhibitor of mitogen-activated protein kinase kinase (MAP2K, MAPK/ERK kinase, or MEK) 1 and 2, with potential antineoplastic activity. Upon administration, luvometinib selectively binds to and inhibits the activity of MEK1 and MEK2, preventing the activation of MEK1/2-dependent effector proteins and transcription factors, which may result in the inhibition of growth factor-mediated cell signaling and tumor cell proliferation. MEK1/2 are dual-specificity threonine/tyrosine kinases that play key roles in the activation of the RAS/RAF/MEK/ERK pathway that regulates cell growth. This pathway is often dysregulated in a variety of tumor cell types through BRAF, KRAS and NRAS mutations.
Luvometinib is a small molecule drug. The usage of the INN stem ‘-tinib’ in the name indicates that Luvometinib is a tyrosine kinase inhibitor. Luvometinib is under investigation in clinical trial NCT07004075 (FCN-159 Monotherapy Versus Chemotherapy by Investigator’s Choice in Pediatric Low-grade Glioma Patients With BRAF Alteration). Luvometinib has a monoisotopic molecular weight of 665.04 Da.
SYN
Example 8
N-(3-(6-allyl-3-ƒluoro-4-(2-ƒluoro-4-iodophenylamino)-1-methyl-2,5-dioxo-1,2,5,6- tetrahydropyrido[2,3-d]pyridazin-8-yl)phenyl)cyclopropanesulƒonamide (8)
The title compound 8 was prepared following the same procedure as described for Example 5 by substituting methanesulfonyl chloride with cyclopropanesulfonyl chloride. MS-ESI (m/z): 666 [M + 1]+.
PAT
Example 8
N-(3-(6-cyclopropyl-3-fluoro-4-(2-fluoro-4-iodophenylamino)-1-methyl-2,5-dioxo-1,2,5,6-tetrahydropyrido[2,3-d]pyridazin-8-yl)phenyl)cyclopropanesulfonamide (8)
[0136] The title compound 8 was prepared following the same procedure as described for Example 5 by substituting methanesulfonyl chloride with cyclopropanesulfonyl chloride. MS-ESI (m/z): 666 [M + 1] +.
PAT
- Protein kinase inhibitorsPublication Number: CN-106905316-BPriority Date: 2013-04-18Grant Date: 2021-06-01
- Certain protein kinase inhibitorsPublication Number: EP-2986611-B1Priority Date: 2013-04-18Grant Date: 2019-02-06
- Certain protein kinase inhibitorsPublication Number: US-10022374-B2Priority Date: 2013-04-18Grant Date: 2018-07-17



AS ON OCT2025 4.511 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
- Cheng Y, Tian H (2017). “Current Development Status of MEK Inhibitors”. Molecules. 22 (10). Basel, Switzerland: 1551. doi:10.3390/molecules22101551. PMC 6151813. PMID 28954413.
- Keam SJ (2025). “Luvometinib: First Approval”. Drugs. 85 (9): 1177–1183. doi:10.1007/s40265-025-02217-6. PMID 40751881.
| Clinical data | |
|---|---|
| Trade names | 复迈宁 (Fu Mainin) |
| Other names | FCN-159 |
| Routes of administration | Oral |
| Identifiers | |
| IUPAC name | |
| CAS Number | 2739690-43-6 |
| PubChem CID | 135210935 |
| IUPHAR/BPS | 13495 |
| UNII | B2DYT4V89X |
| Chemical and physical data | |
| Formula | C26H22F2IN5O4S |
| Molar mass | 665.45 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| SMILES | |
| InChI | |
//////////luvometinib, CHINA 2025, APPROVALS 2025, antineoplastic, FCN 159, FCN-159, B2DYT4V89X, ANAX, PTFEON, ADVECT, BLUE JET
ADVERTISEMENT
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
ADVERTISEMENT
Advect Process Systems Ltd.
ADVECT PROCESS SYSTEMS CANADA LTD
51 Beechwood Rd., Cambridge, ON Canada N1S 3S1, Call Now +1 306 850 6737, Mail Now, ask@advectprocess.com
ADVERTISEMENT
BLUE JET HEALTHCARE LTD, https://bluejethealthcare.com
Looking for a Reliable SNAC Manufacturer? Let’s Talk.
At Blue Jet Healthcare Ltd, we specialize in the scalable, high-purity production of SNAC—a critical excipient powering the next generation of oral peptide therapeutics.
With increasing demand for SNAC across global pharma pipelines, choosing the right manufacturing partner is essential. Quality, timelines, and consistency matter.

Phone No. +91 (22) 22075307 / +91 (22) 22071691
Business Development/ Contract Manufacturing: marketing1@bluejethealthcare.com, madhu.gautam71@gmail.com
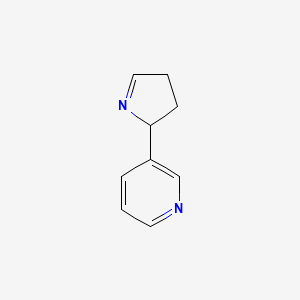
Isomiosamine
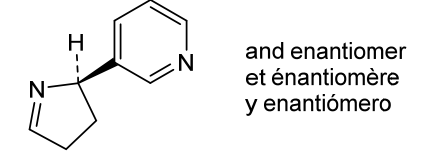
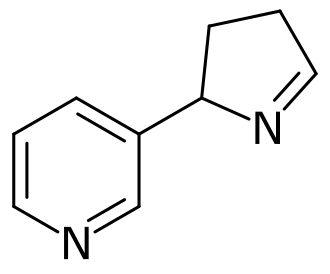
Isomiosamine
CAS 53844-46-5
MF C9H10N2 MW
146.19 g/mol
3-(3,4-dihydro-2H-pyrrol-2-yl)pyridine
rac-(3R)-3-(3,4-dihydro-2H-pyrrol-2-yl)pyridine
tumor necrosis factor alpha (TNFα) inhibitor, MyMD-1, MYMD-1, Isomyosamine, 3A50Y1J4LP, MyMD Pharmaceuticals
synthetic derivative of tobacco alkaloids
Isomyosamine, also known as MyMD-1 or MYMD-1, is a synthetic derivative of tobacco plant alkaloids being developed as a metabolic- and immunomodulator by MyMD Pharmaceuticals. To date, isomyosamine has been shown to suppress the production of IFN-γ, IL-2, IL-10, and TNF-α, and decrease the severity of experimental thyroiditis in a murine model.[1] Trials in humans are being planned, and some are underway, examining the potential benefits of isomyosamine in autoimmune diseases such as rheumatoid arthritis, and in sarcopenia and frailty.[2]
MyMD Pharmaceuticals claim that MYMD-1 is not immunosuppressive, and thus should not be associated with the dangerous side effects such as infections that are seen in currently used TNF-α inhibitors such as adalimumab.[3] While it is true that there currently is no evidence of immunosuppression in isomyosamine recipients, this has not yet been tested in large clinical trials
Safety and Efficacy of Isomyosamine in Reducing Inflammation and Treating Muscle Loss in Older Adults After Hip or Thigh Bone Fractures
CTID: NCT06942182
Phase: Phase 2
Status: Not yet recruiting
Date: 2025-04-24
SYN
Isomyosmine
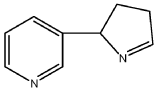
[08] Isomyosmine (3-(3,4-dihydro-2H-pyrrol-2-yl)-pyridine) is a nicotine related alkaloid present in solanecea plants containing nicotine.
PAT
| Unless otherwise clear from context, all percentages referred to herein are expressed as percent by weight based on the total weight of the composition. Percentages expressed herein as “w/v” refer to mass, in grams, of the component per 100 ml of solvent. For example, a 1% (w/v) composition of isomyosmine contains lg (1000 mg) of isomyosmine per 100 ml of solvent, which is equivalent to 10 mg/ml. |
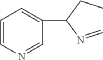
| Isomyosmine (3-(3,4-dihydro-2H-pyrrol-2-yl)-pyridine) is a nicotine related alkaloid present in solanecea plants containing nicotine. |
PAT
- Method of Treating Substance AddictionsPublication Number: US-2017333415-A1Priority Date: 2015-02-19
- Method of treating addictions to opioidsPublication Number: US-10471052-B2Priority Date: 2015-02-19Grant Date: 2019-11-12
- Method of treating cocaine addictionPublication Number: US-11331310-B2Priority Date: 2015-02-19Grant Date: 2022-05-17
- Method of treating disorders associated with chronic inflammationPublication Number: US-2021106578-A1Priority Date: 2015-03-31
- Method of Treating Substance AddictionsPublication Number: US-2020215045-A1Priority Date: 2015-02-19
- Compositions for e-cigarettesPublication Number: WO-2016133890-A1Priority Date: 2015-02-19
- Method of treating substance addictionsPublication Number: US-9884055-B2Priority Date: 2015-02-19Grant Date: 2018-02-06
- Method of Treating Substance AddictionsPublication Number: US-2018140590-A1Priority Date: 2015-02-19
- Methods of treating sarcopeniaPublication Number: US-11219620-B2Priority Date: 2015-03-31Grant Date: 2022-01-11
- Methods of Treating Apoptosis and Altering Programmed Cell DeathPublication Number: US-2018021321-A1Priority Date: 2015-03-31
- Methods of reversing normal aging process and extending lifespanPublication Number: US-11179382-B2Priority Date: 2015-03-31Grant Date: 2021-11-23
- Method of treating viral infectionsPublication Number: US-10786493-B2Priority Date: 2015-03-31Grant Date: 2020-09-29
- Methods of treating cancer, autoimmune disorders, and other conditions associated with chronic inflammationPublication Number: CN-107666907-BPriority Date: 2015-03-31Grant Date: 2022-05-13



AS ON OCT2025 4.511 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

……
Scientific studies
Preclinical studies
One preliminary murine study comparing isomyosamine to rapamycin, the best-characterised drug slowing the progression of aging, reported an increase in lifespan in the isomyosamine cohort, indicating anti-aging activity. Isomyosamine’s anti-proliferative effects were similar to those of rapamycin.[4]
Clinical trials
A phase I randomised double-blind placebo-controlled trial on healthy volunteers examining the safety and pharmacokinetic properties of different amounts of isomyosamine found no serious adverse events, but 3 cases of mild dysgeusia in the highest-dose (600 mg) cohort. A preliminary decrease in TNF-α levels was reported in the lowest-dose (150 mg) cohort, but not in the placebo cohort.[5]
| Identifiers | |
|---|---|
| CAS Number | 53844-46-5 |
| 3D model (JSmol) | Interactive image |
| ChemSpider | 9461533 |
| PubChem CID | 11286546 |
| UNII | 3A50Y1J4LP |
| CompTox Dashboard (EPA) | DTXSID80461155 |
| InChI | |
| SMILES | |
| Properties | |
| Chemical formula | C9H10N2 |
| Molar mass | 146.193 g·mol−1 |
| Related compounds | |
| Related compounds | Myosmine Nicotine |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).Infobox references | |
References
- Di Dalmazi, Giulia; Chalan, Paulina; Caturegli, Patrizio (2019-03-01). “MYMD-1, a Novel Immunometabolic Regulator, Ameliorates Autoimmune Thyroiditis via Suppression of Th1 Responses and TNF-α Release”. The Journal of Immunology. 202 (5): 1350–1362. doi:10.4049/jimmunol.1801238. ISSN 0022-1767. PMID 30674573. S2CID 59226562.
- “MyMD Pharmaceuticals® Provides Dosing Update on Phase 2 Multi-Center Clinical Trial of MYMD-1® as a Therapy for Delaying Aging and Extending Healthy Lifespan”. MyMD. Retrieved 2023-08-13.
- “MYMD-1®”. MyMD. Retrieved 2023-08-13.
- Sabini, Elena; O’Mahony, Alison; Caturegli, Patrizio (2023-02-24). Anderson, Rozalyn M (ed.). “MyMD-1 Improves Health Span and Prolongs Life Span in Old Mice: A Noninferiority Study to Rapamycin”. The Journals of Gerontology: Series A. 78 (2): 227–235. doi:10.1093/gerona/glac142. ISSN 1079-5006. PMID 35914953.
- Brager, Jenna; Chapman, Chris; Dunn, Leonard; Kaplin, Adam (2022-11-11). “A Double-blind, Placebo-controlled, Randomized, Single Ascending, and Multiple Dose Phase 1 Study to Evaluate the Safety, Tolerability, and Pharmacokinetics of Oral Dose Isomyosamine Capsules in Healthy Adult Subjects”. Drug Research. 73 (2): 95–104. doi:10.1055/a-1962-6834. ISSN 2194-9379. PMC 9902179. PMID 36368677.
/////////////isomiosamine, tumor necrosis factor alpha (TNFα) inhibitor, MyMD-1, MYMD-1, Isomyosamine, 3A50Y1J4LP, MyMD Pharmaceuticals, ANAX, ADVECT, BLUE JET
ADVERTISEMENT
ANAX LABORATORIES, WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
ADVERTISEMENT
Advect Process Systems Ltd. https://advectprocess.com/
ADVECT PROCESS SYSTEMS CANADA LTD
51 Beechwood Rd., Cambridge, ON Canada N1S 3S1, Call Now +1 306 850 6737, Mail Now, ask@advectprocess.com
ADVERTISEMENT
BLUE JET HEALTHCARE LTD, https://bluejethealthcare.com
Looking for a Reliable SNAC Manufacturer? Let’s Talk.
At Blue Jet Healthcare Ltd, we specialize in the scalable, high-purity production of SNAC—a critical excipient powering the next generation of oral peptide therapeutics.
With increasing demand for SNAC across global pharma pipelines, choosing the right manufacturing partner is essential. Quality, timelines, and consistency matter.

Phone No. +91 (22) 22075307 / +91 (22) 22071691
Business Development/ Contract Manufacturing: marketing1@bluejethealthcare.com, madhu.gautam71@gmail.com
Gridegalutamide
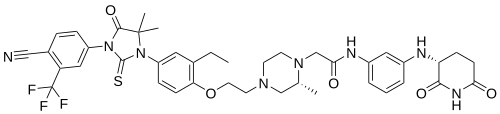
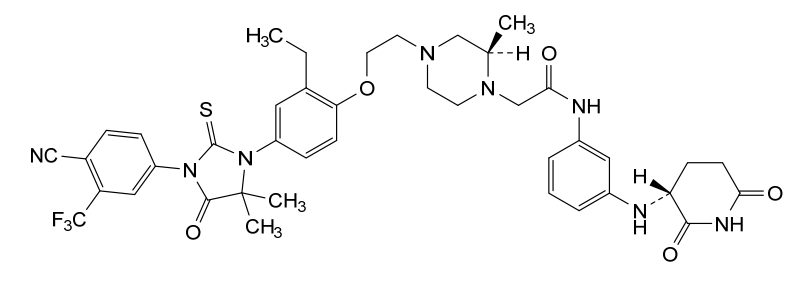
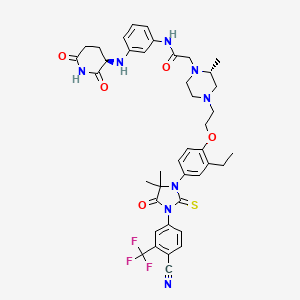
Gridegalutamide
CAS 2446929-86-6
MF C41H45F3N8O5S MW818.9 g/mol
2-[(2R)-4-[2-[4-[3-[4-cyano-3-(trifluoromethyl)phenyl]-5,5-dimethyl-4-oxo-2-sulfanylideneimidazolidin-1-yl]-2-ethylphenoxy]ethyl]-2-methylpiperazin-1-yl]-N-[3-[[(3R)-2,6-dioxopiperidin-3-yl]amino]phenyl]acetamide

antiandrogen, antineoplastic, BMS 986365, CC 94676, BMS-986365, CC-94676, CEL 010355,
VA228VR2DI,
Gridegalutamide is an investigational oral androgen receptor (AR) degrader being developed for the treatment of metastatic castration-resistant prostate cancer (mCRPC). It belongs to a class of drugs called proteolysis targeting chimeras (PROTACs), which are designed to selectively degrade specific proteins by hijacking the ubiquitin-proteasome system.[1][2] CC-94676 employs a unique dual mechanism of action, combining AR degradation with AR antagonism, potentially offering advantages over traditional AR inhibitors in overcoming resistance mechanisms.[3] Initially developed by Celgene and now under Bristol Myers Squibb, CC-94676 has demonstrated AR protein degradation and suppression of tumor growth in CRPC mouse models.[2] As of 2024, CC-94676 is being evaluated in phase I clinical trials for patients with mCRPC who have progressed on androgen deprivation therapy and at least one prior secondary hormonal therapy.[1][2]
Gridegalutamide is a small molecule drug. The usage of the INN stem ‘-lutamide’ in the name indicates that Gridegalutamide is a non-steroid antiandrogen. Gridegalutamide is under investigation in clinical trial NCT04428788 (Study to Evaluate the Safety and Tolerability of CC-94676 in Participants With Metastatic Castration-Resistant Prostate Cancer). Gridegalutamide has a monoisotopic molecular weight of 818.32 Da.
GRIDEGALUTAMIDE is a small molecule drug with a maximum clinical trial phase of II (across all indications) and has 3 investigational indications.
Gridegalutamide is an orally bioavailable androgen receptor (AR) degrader, with potential antineoplastic activity. Upon administration, gridegalutamide causes degradation of AR, prevents AR-mediated signaling and inhibits the proliferation of AR-overexpressing tumor cells. AR plays a key role in tumor cell proliferation in castration-resistant prostate cancer (CRPC).
- A Study to Evaluate the Drug Levels, Metabolism and Excretion, and Absolute Bioavailability of BMS-986365 in Healthy Male ParticipantsCTID: NCT06433505Phase: Phase 1Status: CompletedDate: 2025-03-26
- Study to Evaluate the Safety and Tolerability of CC-94676 in Participants With Metastatic Castration-Resistant Prostate CancerCTID: NCT04428788Phase: Phase 1Status: CompletedDate: 2025-12-22
SYN
DRUGHUNTER
https://drughunter.com/molecule/gridegalutamide-bms-986365-cc-94676
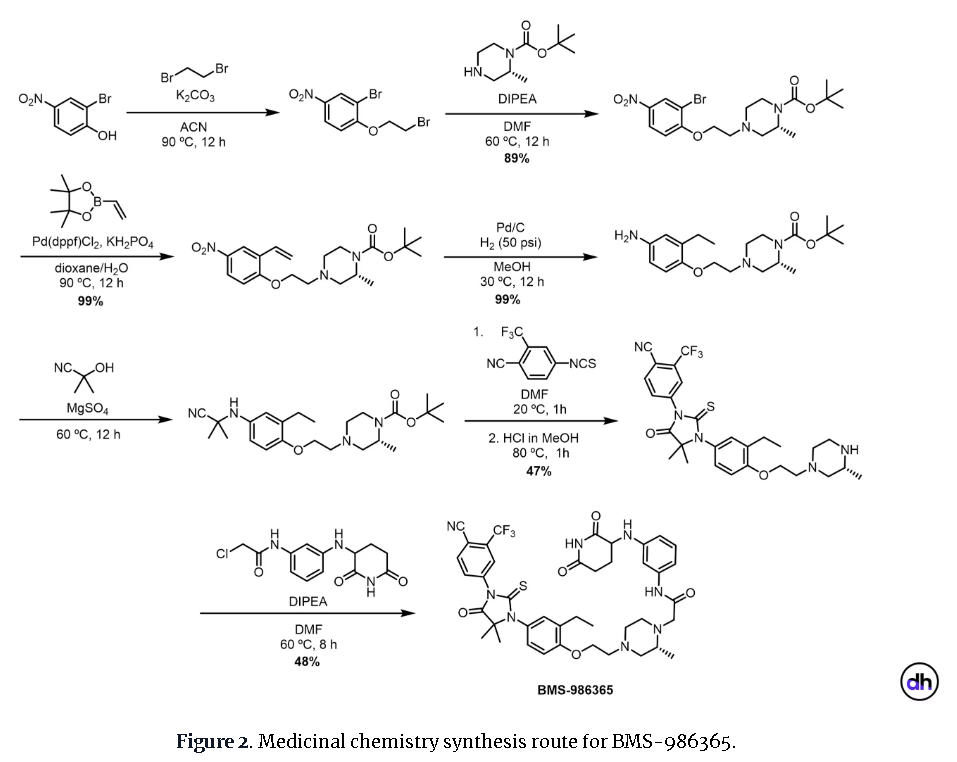
PAT
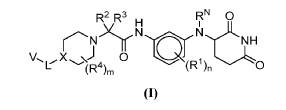
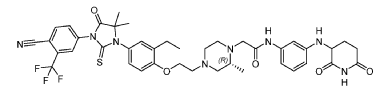
Example 17: 2-((R)-4-(2-(4-(3-(4-Cyano-3-(trifluoromethyl)phenyl)-5,5-dimethyl-4-oxo-2- thioxoimidazolidin-1-yl)-2-ethylphenoxy)ethyl)-2-methylpiperazin-1-yl)-N-(3-((2,6- dioxopiperidin-3-yl)amino)phenyl)acetamide hydrochloride



PAT
- Combination therapy with substituted 3- ((3-aminophenyl) amino) piperidine-2, 6-dione compoundsPublication Number: CN-120152718-APriority Date: 2022-11-09
- Combination therapy with substituted 3-((3-aminophenyl)amino)piperidine-2,6-dione compoundsPublication Number: WO-2024102706-A1Priority Date: 2022-11-09
- Substituted 3-((3-aminophenyl)amino)piperidine-2,6-dione compounds, compositions thereof, and methods of treatment therewithPublication Number: US-2020199073-A1Priority Date: 2018-12-19
- Substituted 3-((3-aminophenyl)amino)piperidine-2,6-dione compounds, compositions thereof, and methods of treatment therewithPublication Number: US-11149007-B2Priority Date: 2018-12-19Grant Date: 2021-10-19
- Substituted 3-((3-aminophenyl)amino)piperidine-2,6-dione compounds, compositions thereof, and methods of treatment therewithPublication Number: US-11873283-B2Priority Date: 2018-12-19Grant Date: 2024-01-16
- Substituted 3-((3-aminophenyl)amino)piperidine-2,6-dione compounds, compositions thereof, and methods of treatment therewithPublication Number: US-2024368083-A1Priority Date: 2018-12-19
- Substituted 3-((3-aminophenyl)amino)piperidine-2,6-dione compounds, compositions thereof, and methods of treatment therewithPublication Number: US-12404241-B2Priority Date: 2018-12-19Grant Date: 2025-09-02
- Substituted 3-((3-aminophenyl)amino)piperidine-2,6-dione compounds, compositions thereof, and methods of treatment therewithPublication Number: US-2023002321-A1Priority Date: 2018-12-19



AS ON OCT2025 4.511 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

……
ADVERTISEMENT
ANAX LABORATORIES, WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
ADVERTISEMENT
Advect Process Systems Ltd. https://advectprocess.com

ADVECT PROCESS SYSTEMS CANADA LTD
51 Beechwood Rd., Cambridge, ON Canada N1S 3S1, Call Now +1 306 850 6737, Mail Now, ask@advectprocess.com
References
- Salama AK, Trkulja MV, Casanova E, Uras IZ (December 2022). “Targeted Protein Degradation: Clinical Advances in the Field of Oncology”. International Journal of Molecular Sciences. 23 (23) 15440. doi:10.3390/ijms232315440. PMC 9741350. PMID 36499765.
- Xie H, Liu J, Alem Glison DM, Fleming JB (2021). “The clinical advances of proteolysis targeting chimeras in oncology”. Exploration of Targeted Anti-Tumor Therapy. 2 (6): 511–521. doi:10.37349/etat.2021.00061. PMC 9400722. PMID 36046114.
- Rathkopf DE, Patel MR, Choudhury AD, Rasco D, Lakhani N, Hawley JE, et al. (September 2024). “Safety and clinical activity of BMS-986365 (CC-94676), a dual androgen receptor ligand-directed degrader and antagonist, in heavily pretreated patients with metastatic castration-resistant prostate cancer”. Annals of Oncology. 36 (1): 76–88. doi:10.1016/j.annonc.2024.09.005. PMC 12094577. PMID 39293515.
| Clinical data | |
|---|---|
| Other names | BMS-986365; CC-94676 |
| Identifiers | |
| IUPAC name | |
| CAS Number | 2446929-86-6 |
| PubChem CID | 153513643 |
| ChemSpider | 133326102 |
| UNII | VA228VR2DI |
| KEGG | D12866 |
| ChEMBL | ChEMBL6068413 |
| Chemical and physical data | |
| Formula | C41H45F3N8O5S |
| Molar mass | 818.92 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| SMILES | |
| InChI | |
//////////gridegalutamide, ANAX, ADVECT, antiandrogen, antineoplastic, BMS 986365, CC 94676, BMS-986365, CC-94676, CEL 010355, VA228VR2DI,
Glasmacinal



Glasmacinal
CAS 2097822-02-9
MF C37H62N2O10 MW694.90
[(2S,3R,4S,6R)-4-(dimethylamino)-2-[[(2R,3S,4R,5R,8R,10R,11R,12S,13S,14R)-2-ethyl-3,4,10,13-tetrahydroxy-3,5,6,8,10,12,14-heptamethyl-15-oxo-1-oxa-6-azacyclopentadec-11-yl]oxy]-6-methyloxan-3-yl] benzoate
- (2R,3S,4R,5R,8R,10R,11R,12S,13S,14R)-11-[[2-O-Benzoyl-3,4,6-trideoxy-3-(dimethylamino)-beta-D-xylo-hexopyranosyl]oxy]-2-ethyl-3,4,10,13-tetrahydroxy-3,5,6,8,10,12,14-heptamethyl-1-oxa-6-azacyclopentadecan-15-one
- 1-Oxa-6-azacyclopentadecan-15-one, 11-[[2-O-benzoyl-3,4,6-trideoxy-3-(dimethylamino)-beta-D-xylo-hexopyranosyl]oxy]-2-ethyl-3,4,10,13-tetrahydroxy-3,5,6,8,10,12,14-heptamethyl-, (2R,3S,4R,5R,8R,10R,11R,12S,13S,14R)-
(2R,3S,4R,5R,8R,10R,11R,12S,13S,14R)-11-{[2-O-benzoyl-3,4,6-trideoxy-3-(dimethylamino) -β-D-xylo-hexopyranosyl]oxy}-2-ethyl3,4,10,13-tetrahydroxy-3,5,6,8,10,12,14-heptamethyl-1-oxa-6-
azacyclopentadecan-15-one
non-antibacterial macrolide, anti-inflammatory, EP 395, M3T8D3P634
SYN
https://patentscope.wipo.int/search/en/detail.jsf?docId=US234729681&_cid=P12-MKVZ26-57135-1
Example 2: (2S,3R,4S,6R)-4-(dimethylamino)-2-[[(2R,3S,4R,5R,8R,10R,11R,12S,13S,14R)-2-ethyl-3,4,10,13-tetrahydroxy-3,5,6,8,10,12,14-heptamethyl-15-oxo-1-oxa-6-azacyclopentadec-11-yl]oxy]-6-methyl-tetrahydropyran-3-yl] benzoate)

To a mixture of (2R,3S,4R,5R,8R,10R,11R,12S,13S,14R)-11-[(2S,3R,4S,6R)-4-(dimethylamino)-3-hydroxy-6-methyl-tetrahydropyran-2-yl]oxy-2-ethyl-3,4,10,13-tetrahydroxy-3,5,6,8,10,12,14-heptamethyl-1-oxa-6-azacyclopentadecan-15-one (Example 1) (0.5 g, 0.8500 mmol) and Triethylamine (428.2 mg, 4.23 mmol) in DCM (5 ml), cooled on ice, was added Benzoyl chloride (356.9 mg, 2.54 mmol). The reaction mixture was allowed to reach room temperature. After 3 days good conversion to the desired benzoylated product was obtained and the mixture was portioned between DCM and saturated sodium hydrogen carbonate solution. The organic phase was dried over magnesium sulphate and concentrated to a white foam. The product was purified using reversed phase chromatography (see general information)
PAT
- Azithromycin Derivatives With Epithelial Barrier Enhancement PropertiesPublication Number: US-2018354981-A1Priority Date: 2015-11-19
- Azithromycin derivatives with epithelial barrier enhancement propertiesPublication Number: US-10723752-B2Priority Date: 2015-11-19Grant Date: 2020-07-28
- Azithromycin Derivatives With Epithelial Barrier Enhancement PropertiesPublication Number: US-2020317710-A1Priority Date: 2015-11-19
- Azithromycin derivatives with epithelial barrier enhancement propertiesPublication Number: US-12049477-B2Priority Date: 2015-11-19Grant Date: 2024-07-30
- Azithromycin derivatives with epithelial barrier enhancement propertiesPublication Number: US-11236120-B2Priority Date: 2015-11-19Grant Date: 2022-02-01
- Compounds
- Publication Number: US-2022106349-A1
- Priority Date: 2015-11-19



AS ON OCT2025 4.511 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

……
ADVERTISEMENT
ANAX LABORATORIES, WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
ADVERTISEMENT
Advect Process Systems Ltd. https://advectprocess.com

ADVECT PROCESS SYSTEMS CANADA LTD
51 Beechwood Rd., Cambridge, ON Canada N1S 3S1, Call Now +1 306 850 6737, Mail Now, ask@advectprocess.com
///////glasmacinal, ANAX, ADVECT, non-antibacterial macrolide, anti-inflammatory, EP 395, M3T8D3P634

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....










