Slightly soluble in water
Soluble in most organic solvents
PROUD Indian WORLD RECORD VIEWS holder on THIS BLOG, ………live, by DR ANTHONY MELVIN CRASTO, Worldpeaceambassador, Worlddrugtracker, Helping millions, 100 million hits on google, pushing boundaries,2.5 lakh plus connections worldwide, 45 lakh plus VIEWS on this blog in 227 countries, 7 CONTINENTS ……A 90 % paralysed man in action for you, I am suffering from transverse mylitis and bound to a wheel chair, [THIS BLOG HOLDS WORLD RECORD VIEWS ] A PROUD INDIAN
FDA approves new treatment for a rare genetic disorder, Fabry disease
The U.S. Food and Drug Administration today approved Galafold (migalastat), the first oral medication for the treatment of adults with Fabry disease. The drug is indicated for adults with Fabry disease who have a genetic mutation determined to be responsive (“amenable”) to treatment with Galafold based on laboratory data. Fabry disease is a rare and serious genetic disease that results from buildup of a type of fat called globotriaosylceramide (GL-3) in blood vessels, the kidneys, the heart, the nerves and other organs.
August 10, 2018
The U.S. Food and Drug Administration today approved Galafold (migalastat), the first oral medication for the treatment of adults with Fabry disease. The drug is indicated for adults with Fabry disease who have a genetic mutation determined to be responsive (“amenable”) to treatment with Galafold based on laboratory data. Fabry disease is a rare and serious genetic disease that results from buildup of a type of fat called globotriaosylceramide (GL-3) in blood vessels, the kidneys, the heart, the nerves and other organs.
“Thus far, treatment of Fabry disease has involved replacing the missing enzyme that causes the particular type of fat buildup in this disease. Galafold differs from enzyme replacement in that it increases the activity of the body’s deficient enzyme,” said Julie Beitz, M.D., director of the Office of Drug Evaluation III in FDA’s Center for Drug Evaluation and Research.
Fabry disease is an inherited disorder caused by mutations (alterations) in the alpha-galactosidase A (GLA) gene located on the X-chromosome. Fabry disease is rare and affects both males and females. It is estimated that classic Fabry disease (the most severe type) affects approximately one in 40,000 males. The later-onset type is more frequent, and in some populations, may occur in one in 1,500 to 4,000 males. Patients with Fabry disease develop slowly progressive kidney disease, cardiac hypertrophy (enlargement of the heart), arrhythmias (abnormal heart rhythm), stroke and early death.
The efficacy of Galafold was demonstrated in a six-month, placebo-controlled clinical trial in 45 adults with Fabry disease. In this trial, patients treated with Galafold over six months had a greater reduction in globotriaosylceramide (GL-3) in blood vessels of the kidneys (as measured in kidney biopsy samples) as compared to patients on placebo.The safety of Galafold was studied in four clinical trials which included a total of 139 patients with Fabry disease.
The most common adverse drug reactions in patients taking Galafold in clinical trials were headache, nasal and throat irritation (nasopharyngitis), urinary tract infection, nausea, and fever (pyrexia).
Galafold was approved using the Accelerated Approval pathway, under which the FDA may approve drugs for serious conditions where there is an unmet medical need and where a drug is shown to have certain effects that are reasonably likely to predict a clinical benefit to patients. A further study is required to verify and describe the clinical benefits of Galafold, and the sponsor will be conducting a confirmatory clinical trial of Galafold in adults with Fabry disease.
Galafold was granted Priority Review designation, under which the FDA’s goal is to take action on an application within six months of application filing where the agency determines that the drug, if approved, would provide a significant improvement in treating, diagnosing or preventing a serious condition over available therapies. Galafold also received Orphan Drug designation, which provides incentives to assist and encourage the development of drugs for rare diseases.
The FDA granted approval of Galafold to Amicus Therapeutics U.S., Inc.
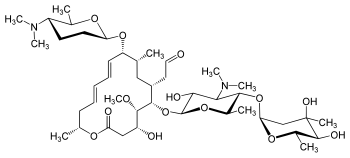
Spiramycin
スピラマイシン
CAS: 8025-81-8
Sanofi INNOVATOR
| Molecular Formula: | C43H74N2O14 |
|---|---|
| Molecular Weight: | 843.065 g/mol |
[(11E,13E)-6-({5-[(4,5-Dihydroxy-4,6-dimethyltetrahydro-2H-pyran-2-yl)oxy]-4-(dimethylamino)-3-hydroxy-6-methyltetrahydro-2H-pyran-2-yl}oxy)-10-{[5-(dimethylamino)-6-methyltetrahydro-2H-pyran-2-yl]oxy }-4-hydroxy-5-methoxy-9,16-dimethyl-2-oxooxacyclohexadeca-11,13-dien-7-yl]acetaldehyde
2-[(11E,13E)-6-[5-(4,5-dihydroxy-4,6-dimethyloxan-2-yl)oxy-4-(dimethylamino)-3-hydroxy-6-methyloxan-2-yl]oxy-10-[5-(dimethylamino)-6-methyloxan-2-yl]oxy-4-hydroxy-5-methoxy-9,16-dimethyl-2-oxo-1-oxacyclohexadeca-11,13-dien-7-yl]acetaldehyde
Leucomycin V, 9-O-[(2R,5S,6R)-5-(dimethylamino)tetrahydro-6-methyl-2H-pyran-2-yl]-
2018/7/2 japan approved, UNII: 71ODY0V87H
Slightly soluble in water
Soluble in most organic solvents
UV max (ethanol): 231nm
Specific optical rotation: -80 deg at 20 deg C/D
| スピラマイシン Spiramycin 
[検索ページへ戻る]
|
|
|
| スピラマイシン酢酸エステル JP17 Spiramycin Acetate  |
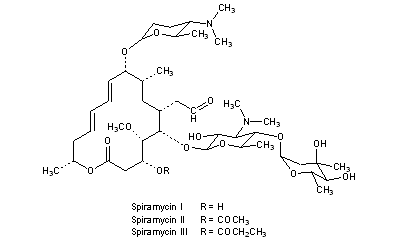
Spiramycin is a macrolide antimicrobial agent with activity against gram-positive organisms, including Streptococcus pyogenes (group A beta-hemolytic streptococci), S. viridans, Corynebacterium diphtheriae, and methicillin-sensitive Staphylococcus aureus. Spiramycin is a 16-membered ring macrolide. It was discovered in 1952 as a product of Streptomyces ambofaciens. As a preparation for oral administration it has been used since 1955, in 1987 also the parenteral form was introduced into practice. The antibacterial spectrum comprises Gram-positive cocci and rods, Gram-negative cocci and also Legionellae, mycoplasmas, chlamydiae, some types of spirochetes, Toxoplasma gondii and Cryptosporidium species. Enterobacteria, pseudomonads and pathogenic moulds are resistant. Its action is mainly bacteriostatic, on highly sensitive strains it exerts a bactericide action.
Spiramycin is a macrolide antibiotic and antiparasitic It is used to treat toxoplasmosis and various other infections of soft tissues. Although used in Europe, Canada and Mexico,[1] spiramycin is still considered an experimental drug in the United States, but can sometimes be obtained by special permission from the FDA for toxoplasmosis in the first trimester of pregnancy.[2]
Spiramycin has been used in Europe since the year 2000 under the trade name “Rovamycine”, produced by Rhone-Poulenc Rorer and Famar Lyon, France and Eczacıbaşı İlaç, Turkey. It also goes under the name Rovamycine in Canada (distributed by OdanLaboratories), where it is mostly marketed to dentists for mouth infections.
Spiramycin is a 16-membered ring macrolide. It was discovered in 1952 as a product of Streptomyces ambofaciens. As a preparation for oral administration it has been used since 1955, in 1987 also the parenteral form was introduced into practice. The antibiotic action involves inhibition of protein synthesis in the bacterial cell during translocation. Resistance to spiramycin can develop by several mechanisms and its prevalence is to a considerable extent proportional to the frequency of prescription in a given area. The antibacterial spectrum comprises Gram-positive cocci and rods, Gram-negative cocci and also Legionellae, mycoplasmas, chlamydiae, some types of spirochetes, Toxoplasma gondii and Cryptosporidium species. Enterobacteria, pseudomonads and pathogenic moulds are resistant. Its action is mainly bacteriostatic, on highly sensitive strains it exerts a bactericide action. As compared with erythromycin, it is in vitro weight for weight 5 to 20 less effective, an equipotential therapeutic dose is, however, only double. This difference between the effectiveness in vitro and in vivo is explained above all by the great affinity of spiramycin to tissues where it achieves concentrations many times higher than serum levels. An important part is played also by the slow release of the antibiotic from the tissue compartment, the marked action on microbes in sub-inhibition concentrations and the relatively long persisting post-antibiotic effect. Its great advantage is the exceptionally favourable tolerance-gastrointestinal and general. It is available for parenteral and oral administration
| Country | Trade Name | Vendor | Annotation |
|---|---|---|---|
| D | Rovamycine | Teofarma | |
| Selectomycin | Grünenthal | ||
| F | Bi Missilor | Pierre Fabre | |
| Birodogyl | Sanofi-Aventis | ||
| Missilor | Pierre Fabre | comb. | |
| Rodogyl | Pierre Fabre | ||
| Rovamycine | Grünenthal | ||
| I | Rovamicina | Sanofi-Aventis | |
| Rovamycina | Teofarma | ||
| Spiromix | Pulitzer |
 |
|
| Clinical data | |
|---|---|
| Synonyms | 2-[(4R,5S,6S,7R,9R,10R,11E,13E,16R)-6-{[(2S,3R,4R,5S,6R)-5-{[(2S,5S,6S)-4,5-dihydroxy-4,6-dimethyloxan-2-yl]oxy}-4-(dimethylamino)-3-hydroxy-6-methyloxan-2-yl]oxy}-10-{[(2R,5S,6R)-5-(dimethylamino)-6-methyloxan-2-yl]oxy}-4-hydroxy-5-methoxy-9,16-dimethyl-2-oxo-1-oxacyclohexadeca-11,13-dien-7-yl]acetaldehyde |
| Routes of administration |
oral |
| ATC code | |
| Legal status | |
| Legal status |
|
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| NIAID ChemDB | |
| E number | E710 (antibiotics) |
| ECHA InfoCard | 100.029.476 |
| Chemical and physical data | |
| Formula | C43H74N2O14 |
| Molar mass | 843.053 g/mol |
| 3D model (JSmol) | |
| Solubility in water | Insoluble in water; Very soluble in acetonitrile and methanol; Almost completely(>99.5) in ethanol. mg/mL (20 °C) |
CN 107840864
///////////Spiramycin, スピラマイシン , japan 2018, Provamycin, Rovamycin, RP 5337, Sequamycin, IL 5902, NSC-64393, ATC:J01FA02, Use:macrolide antibiotic, EINECS:232-429-6
O=CCC4C(OC2OC(C(OC1OC(C)C(O)C(O)(C)C1)C(N(C)C)C2O)C)C(OC)C(O)CC(=O)OC(C)C\C=C\C=C\C(OC3OC(C)C(N(C)C)CC3)C(C)C4
FDA approves treatment for two rare types of non-Hodgkin lymphoma
The U.S. Food and Drug Administration today approved Poteligeo (mogamulizumab-kpkc) injection for intravenous use for the treatment of adult patients with relapsed or refractory mycosis fungoides (MF) or Sézary syndrome (SS) after at least one prior systemic therapy. This approval provides a new treatment option for patients with MF and is the first FDA approval of a drug specifically for SS.
August 8, 2018
The U.S. Food and Drug Administration today approved Poteligeo (mogamulizumab-kpkc) injection for intravenous use for the treatment of adult patients with relapsed or refractory mycosis fungoides (MF) or Sézary syndrome (SS) after at least one prior systemic therapy. This approval provides a new treatment option for patients with MF and is the first FDA approval of a drug specifically for SS.
“Mycosis fungoides and Sézary syndrome are rare, hard-to-treat types of non-Hodgkin lymphoma and this approval fills an unmet medical need for these patients,” said Richard Pazdur, M.D., director of the FDA’s Oncology Center of Excellence and acting director of the Office of Hematology and Oncology Products in the FDA’s Center for Drug Evaluation and Research. “We are committed to continuing to expedite the development and review of this type of targeted therapy that offers meaningful treatments for patients.”
Non-Hodgkin lymphoma is a cancer that starts in white blood cells called lymphocytes, which are part of the body’s immune system. MF and SS are types of non-Hodgkin lymphoma in which lymphocytes become cancerous and affect the skin. MF accounts for about half of all lymphomas arising from the skin. It causes itchy red rashes and skin lesions and can spread to other parts of the body. SS is a rare form of skin lymphoma that affects the blood and lymph nodes.
Poteligeo is a monoclonal antibody that binds to a protein (called CC chemokine receptor type 4 or CCR4) found on some cancer cells.
The approval was based on a clinical trial of 372 patients with relapsed MF or SS who received either Poteligeo or a type of chemotherapy called vorinostat. Progression-free survival (the amount of time a patient stays alive without the cancer growing) was longer for patients taking Poteligeo (median 7.6 months) compared to patients taking vorinostat (median 3.1 months).
The most common side effects of treatment with Poteligeo included rash, infusion-related reactions, fatigue, diarrhea, musculoskeletal pain and upper respiratory tract infection.
Serious warnings of treatment with Poteligeo include the risk of dermatologic toxicity, infusion reactions, infections, autoimmune problems (a condition where the immune cells in the body attack other cells or organs in the body), and complications of stem cell transplantation that uses donor stem cells (allogeneic) after treatment with the drug.
The FDA granted this application Priority Review and Breakthrough Therapydesignation. Poteligeo also received Orphan Drug designation, which provides incentives to assist and encourage the development of drugs for rare diseases.
The FDA granted this approval to Kyowa Kirin, Inc.
https://www.fda.gov/Drugs/InformationOnDrugs/ApprovedDrugs/ucm615348.htm
synthesis………..https://newdrugapprovals.org/2015/08/20/lusutrombopag-oral-thrombopoietin-tpo-mimetic/
On July 31, 2018, the Food and Drug Administration approved lusutrombopag (Mulpleta, Shionogi Inc.) for thrombocytopenia in adults with chronic liver disease who are scheduled to undergo a medical or dental procedure.
Approval was based on two randomized, double-blind, placebo-controlled trials (L-PLUS 1 and L-PLUS 2, NCT02389621) involving 312 patients with chronic liver disease and severe thrombocytopenia who were undergoing an invasive procedure and had a platelet count less than 50 x 109/L. Patients were randomized 1:1 to receive 3 mg of lusutrombopag or placebo once daily for up to 7 days.
In L-PLUS 1, 78% of patients (38/49) receiving lusutrombopag required no platelet transfusion prior to the primary invasive procedure, compared with 13% (6/48) who received placebo (95% CI for treatment difference: 49%, 79%; p<0.0001). In L-PLUS 2, 65% (70/108) of patients who received lusutrombopag required no platelet transfusion prior to the primary invasive procedure or rescue therapy for bleeding from randomization through 7 days after the procedure, compared with 29% (31/107) receiving placebo (95% CI for treatment difference: 25%, 49%; p<0.0001).
The most common adverse reaction in ≥ 3% of patients was headache.
The recommended lusutrombopag dosage is 3 mg orally once daily with or without food for 7 days.
View full prescribing information for Mulpleta.
FDA granted this application priority review and fast track designation. A description of FDA expedited programs is in the Guidance for Industry: Expedited Programs for Serious Conditions-Drugs and Biologics.
Healthcare professionals should report all serious adverse events suspected to be associated with the use of any medicine and device to FDA’s MedWatch Reporting System or by calling 1-800-FDA-1088.
Follow the Oncology Center of Excellence on Twitter @FDAOncology.
Check out recent approvals at the OCE’s podcast, Drug Information Soundcast in Clinical Oncology.
Conferred CMO Asia award 2018 ![]() 🇸🇬 singapore
🇸🇬 singapore
Shobha and Aishal crasto collect my International award for Excellence in Pharma by CMO ASIA 31st July 2018 | at an award function in Le Méridien Singapore, Sentosa
Thanking one and all for support
They went thru the paralysis trauma for years and now getting recognition for the efforts
God when he shuts one door he opens many more
My family proudly hold the honor outstanding contribution to pharma society at CMO Asia ![]() 🇸🇬 singapore
🇸🇬 singapore
//////////////Anthony Crasto, International award, outstanding contribution to Pharma society, CMO ASIA, 31st July 2018 , Le Méridien, Sentosa, Singapore,
Iobenguane I 131

FDA approves first treatment for rare adrenal tumors
July 30, 2018
The U.S. Food and Drug Administration today approved Azedra (iobenguane I 131) injection for intravenous use for the treatment of adults and adolescents age 12 and older with rare tumors of the adrenal gland (pheochromocytoma or paraganglioma) that cannot be surgically removed (unresectable), have spread beyond the original tumor site and require systemic anticancer therapy. This is the first FDA-approved drug for this use.
“Many patients with these ultra-rare cancers can be treated with surgery or local therapies, but there are no effective systemic treatments for patients who experience tumor-related symptoms such as high blood pressure,” said Richard Pazdur, M.D., director of the FDA’s Oncology Center of Excellence and acting director of the Office of Hematology and Oncology Products in the FDA’s Center for Drug Evaluation and Research. “Patients will now have an approved therapy that has been shown to decrease the need for blood pressure medication and reduce tumor size in some patients.”
Pheochromocytomas are rare tumors of the adrenal glands. These glands are located right above the kidneys and make hormones including stress hormones called epinephrines and norepinephrines. Pheochromocytomas increase the production of these hormones, leading to hypertension (high blood pressure) and symptoms such as headaches, irritability, sweating, rapid heart rate, nausea, vomiting, weight loss, weakness, chest pain or anxiety. When this type of tumor occurs outside the adrenal gland, it is called a paraganglioma.
The efficacy of Azedra was shown in a single-arm, open-label, clinical trial in 68 patients that measured the number of patients who experienced a 50 percent or greater reduction of all antihypertensive medications lasting for at least six months. This endpoint was supported by the secondary endpoint, overall tumor response measured by traditional imaging criteria. The study met the primary endpoint, with 17 (25 percent) of the 68 evaluable patients experiencing a 50 percent or greater reduction of all antihypertensive medication for at least six months. Overall tumor response was achieved in 15 (22 percent) of the patients studied.
The most common severe side effects reported by patients receiving Azedra in clinical trials included low levels of white blood cells (lymphopenia), abnormally low count of a type of white blood cells (neutropenia), low blood platelet count (thrombocytopenia), fatigue, anemia, increased international normalized ratio (a laboratory test which measures blood clotting), nausea, dizziness, hypertension and vomiting.
As it is a radioactive therapeutic agent, Azedra includes a warning about radiation exposure to patients and family members, which should be minimized while the patient is receiving Azedra. The risk of radiation exposure is greater in pediatric patients. Other warnings and precautions include a risk of lower levels of blood cells (myelosuppression), underactive thyroid, elevations in blood pressure, renal failure or kidney injury and inflammation of lung tissue (pneumonitis). Myelodysplastic syndrome and acute leukemias, which are cancers of the blood and bone marrow, were observed in patients who received Azedra, and the magnitude of this risk will continue to be studied. Azedra can cause harm to a developing fetus; women should be advised of the potential risk to the fetus and to use effective contraception after receiving Azedra. Radiation exposure associated with Azedra may cause infertility in males and females.
The FDA granted this application Fast Track, Breakthrough Therapy and Priority Review designations. Azedra also received Orphan Drug designation, which provides incentives to assist and encourage the development of drugs for rare diseases.
The FDA granted the approval of Azedra to Progenics Pharmaceuticals, Inc.
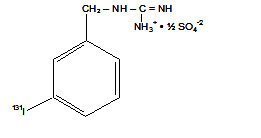
Iobenguane (131I); Iobenguane I 131; Iobeguane I 131; 3-Iodobenzylguanidine; 131I-MIBG; Azedra
77679-27-7 CAS NUMBER
PATENT US 4584187
Guanidine, [[3-(iodo-131I)phenyl]methyl]-
| Molecular Formula: | C8H10IN3 |
|---|---|
| Molecular Weight: | 279.095 g/mol |

| Molecular Formula: | C16H22I2N6O4S |
|---|---|
| Molecular Weight: | 648.259 g/mol |
AdreView
(iobenguane I 123) Injection for Intravenous Use

join me on Linkedin
join me on Researchgate

join me on Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com
SYN
CN 106187824

AdreView (iobenguane I 123 Injection) is a sterile, pyrogen-free radiopharmaceutical for intravenous injection. Each mL contains 0.08 mg iobenguane sulfate, 74 MBq (2 mCi) of I 123 (as iobenguane sulfate I 123) at calibration date and time on the label, 23 mg sodium dihydrogen phosphate dihydrate, 2.8 mg disodium hydrogen phosphate dihydrate and 10.3 mg (1% v/v) benzyl alcohol with a pH of 5.0 – 6.5. Iobenguane sulfate I 123 is also known as I 123 meta-iodobenzlyguanidine sulfate and has the following structural formula:
 |
Iodine 123 is a cyclotron-produced radionuclide that decays to Te 123 by electron capture and has a physical half-life of 13.2 hours.
Iobenguane I-131 is a guanidine analog with specific affinity for tissues of the sympathetic nervous system and related tumors. The radiolabeled forms are used as antineoplastic agents and radioactive imaging agents. (Merck Index, 12th ed) MIBG serves as a neuron-blocking agent which has a strong affinity for, and retention in, the adrenal medulla and also inhibits ADP-ribosyltransferase.
Iobenguane i-131 is a Radioactive Diagnostic Agent. The mechanism of action of iobenguane i-131 is as a Radiopharmaceutical Activity.
Iobenguane I-131 is an I 131 radioiodinated synthetic analogue of the neurotransmitter norepinephrine. Iobenguane localizes to adrenergic tissue and, in radioiodinated forms, may be used to image or eradicate tumor cells that take up and metabolize norepinephrine.
Iobenguane, also known as metaiodobenzylguanidine or mIBG, or MIBG (tradename Adreview) is a radiopharmaceutical,[1] used in a scintigraphy method called MIBG scan. Iobenguane is a radiolabeled molecule similar to noradrenaline.
The radioisotope of iodine used for the label can be iodine-123 (for imaging purposes only) or iodine-131 (which must be used when tissue destruction is desired, but is sometimes used for imaging also).
Pheochromocytoma seen as dark sphere in center of the body (it is in the left adrenal gland). Image is by MIBG scintigraphy, with radiation from radioiodine in the MIBG. Two images are seen of the same patient from front and back. Note dark image of the thyroid due to unwanted uptake of iodide radioiodine from breakdown of the pharmaceutical, by the thyroid gland in the neck. Uptake at the side of the head are from the salivary glands. Radioactivity is also seen in the bladder, from normal renal excretion of iodide.
It localizes to adrenergic tissue and thus can be used to identify the location of tumors[2] such as pheochromocytomas and neuroblastomas. With I-131 it can also be used to eradicate tumor cells that take up and metabolize norepinephrine.
Thyroid blockade with (nonradioactive) potassium iodide is indicated for nuclear medicine scintigraphy with iobenguane/mIBG. This competitively inhibits radioiodine uptake, preventing excessive radioiodine levels in the thyroid and minimizing the risk of thyroid ablation ( in the case of I-131). The minimal risk of thyroid carcinogenesis is also reduced as a result.
The FDA-approved dosing of potassium iodide for this purpose are as follows: infants less than 1 month old, 16 mg; children 1 month to 3 years, 32 mg; children 3 years to 18 years, 65 mg; adults 130 mg.[3] However, some sources recommend alternative dosing regimens.[4]
Not all sources are in agreement on the necessary duration of thyroid blockade, although agreement appears to have been reached about the necessity of blockade for both scintigraphic and therapeutic applications of iobenguane. Commercially available iobenguane is labeled with iodine-123, and product labeling recommends administration of potassium iodide 1 hour prior to administration of the radiopharmaceutical for all age groups,[5] while the European Associated of Nuclear Medicine recommends (for iobenguane labeled with either I-131 or I-123,) that potassium iodide administration begin one day prior to radiopharmaceutical administration, and continue until the day following the injection, with the exception of newborns, who do not require potassium iodide doses following radiopharmaceutical injection.[4]
Product labeling for diagnostic iodine-131 iobenguane recommends potassium iodide administration one day before injection and continuing 5 to 7 days following.[6] Iodine-131 iobenguane used for therapeutic purposes requires a different pre-medication duration, beginning 24–48 hours prior to iobenguane injection and continuing 10–15 days following injection.[7]
The FDOPA PET/CT scan has proven to be nearly 100% sensitive for detection of pheochromocytomas, vs. 90% for MIBG scans.[8][9][10] Centers which offer FDOPA PET/CT, however, are rare.
Iobenguane I 131 (as Azedra) has had a clinical trial as a treatment for malignant, recurrent or unresectable pheochromocytoma and paraganglioma, and the US FDA has granted it a Priority Review.[11]

2-(3-Iodobenzyl)guanidine
35MRW7B4AD
80663-95-2 [RN]
Guanidine, ((3-iodophenyl)methyl)-
Guanidine, N”-[(3-iodophenyl)methyl]-
iobenguane
|
|
| Clinical data | |
|---|---|
| Synonyms | meta-iodobenzylguanidine mIBG, MIBG |
| Routes of administration |
Intravenous |
| ATC code | |
| Legal status | |
| Legal status |
|
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C8H10IN3 |
| Molar mass | 275.09 g/mol |
| 3D model (JSmol) | |


NEW DRUG APPROVALS
ONE TIME
$10.00

FDA approves first treatment for rare adrenal tumors
July 30, 2018
The U.S. Food and Drug Administration today approved Azedra (iobenguane I 131) injection for intravenous use for the treatment of adults and adolescents age 12 and older with rare tumors of the adrenal gland (pheochromocytoma or paraganglioma) that cannot be surgically removed (unresectable), have spread beyond the original tumor site and require systemic anticancer therapy. This is the first FDA-approved drug for this use.
“Many patients with these ultra-rare cancers can be treated with surgery or local therapies, but there are no effective systemic treatments for patients who experience tumor-related symptoms such as high blood pressure,” said Richard Pazdur, M.D., director of the FDA’s Oncology Center of Excellence and acting director of the Office of Hematology and Oncology Products in the FDA’s Center for Drug Evaluation and Research. “Patients will now have an approved therapy that has been shown to decrease the need for blood pressure medication and reduce tumor size in some patients.”
Pheochromocytomas are rare tumors of the adrenal glands. These glands are located right above the kidneys and make hormones including stress hormones called epinephrines and norepinephrines. Pheochromocytomas increase the production of these hormones, leading to hypertension (high blood pressure) and symptoms such as headaches, irritability, sweating, rapid heart rate, nausea, vomiting, weight loss, weakness, chest pain or anxiety. When this type of tumor occurs outside the adrenal gland, it is called a paraganglioma.
The efficacy of Azedra was shown in a single-arm, open-label, clinical trial in 68 patients that measured the number of patients who experienced a 50 percent or greater reduction of all antihypertensive medications lasting for at least six months. This endpoint was supported by the secondary endpoint, overall tumor response measured by traditional imaging criteria. The study met the primary endpoint, with 17 (25 percent) of the 68 evaluable patients experiencing a 50 percent or greater reduction of all antihypertensive medication for at least six months. Overall tumor response was achieved in 15 (22 percent) of the patients studied.
The most common severe side effects reported by patients receiving Azedra in clinical trials included low levels of white blood cells (lymphopenia), abnormally low count of a type of white blood cells (neutropenia), low blood platelet count (thrombocytopenia), fatigue, anemia, increased international normalized ratio (a laboratory test which measures blood clotting), nausea, dizziness, hypertension and vomiting.
As it is a radioactive therapeutic agent, Azedra includes a warning about radiation exposure to patients and family members, which should be minimized while the patient is receiving Azedra. The risk of radiation exposure is greater in pediatric patients. Other warnings and precautions include a risk of lower levels of blood cells (myelosuppression), underactive thyroid, elevations in blood pressure, renal failure or kidney injury and inflammation of lung tissue (pneumonitis). Myelodysplastic syndrome and acute leukemias, which are cancers of the blood and bone marrow, were observed in patients who received Azedra, and the magnitude of this risk will continue to be studied. Azedra can cause harm to a developing fetus; women should be advised of the potential risk to the fetus and to use effective contraception after receiving Azedra. Radiation exposure associated with Azedra may cause infertility in males and females.
The FDA granted this application Fast Track, Breakthrough Therapy and Priority Review designations. Azedra also received Orphan Drug designation, which provides incentives to assist and encourage the development of drugs for rare diseases.
The FDA granted the approval of Azedra to Progenics Pharmaceuticals, Inc.

Fostamatinib

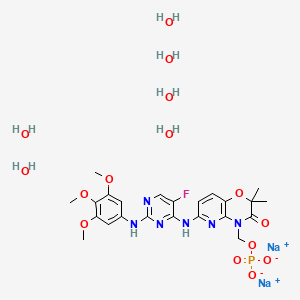
Fostamatinib disodium hexahydrate
ホスタマチニブジナトリウム水和物
| INGREDIENT | UNII | CAS | |
|---|---|---|---|
| Fostamatinib disodium | 86EEZ49YVB | 914295-16-2 |
| Molecular Formula: | C23H36FN6Na2O15P |
|---|---|
| Molecular Weight: | 732.52 g/mol |
TAVALISSE™
(fostamatinib disodium hexahydrate) Tablets, for Oral Use
DESCRIPTION
Fostamatinib is a tyrosine kinase inhibitor. TAVALISSE is formulated with the disodium hexahydrate salt of fostamatinib, a phosphate prodrug that converts to its pharmacologically active metabolite, R406, in vivo.
The chemical name for fostamatinib disodium hexahydrate is disodium (6-[[5-fluoro-2-(3,4,5trimethoxyanilino) pyrimidin-4-yl]amino]-2,2-dimethyl-3-oxo-pyrido[3,2-b][1,4]oxazin-4-yl)methyl phosphate hexahydrate. The molecular formula is C23H24FN6Na2O9P·6H2O, and the molecular weight is 732.52. The structural formula is:
 |
Fostamatinib disodium is a white to off-white powder that is practically insoluble in pH 1.2 aqueous buffer, slightly soluble in water, and soluble in methanol.
Each TAVALISSE oral tablet contains 100 mg or 150 mg fostamatinib, equivalent to 126.2 mg or 189.3 mg fostamatinib disodium hexahydrate, respectively.
The inactive ingredients in the tablet core are mannitol, sodium bicarbonate, sodium starch glycolate, povidone, and magnesium stearate. The inactive ingredients in the film coating are polyvinyl alcohol, titanium dioxide, polyethylene glycol 3350, talc, iron oxide yellow, and iron oxide red.

Fostamatinib, sold under the brand name Tavalisse, is a medication approved by the U.S. Food and Drug Administration since 2018 for the treatment of chronic immune thrombocytopenia (ITP). The drug is administered orally as a disodium hexahydrate salt, and is a prodrug of the active compound tamatinib (R-406),[1] which is an inhibitor of the enzyme spleen tyrosine kinase (Syk),[2] hence it is an syk inhibitor.
Fostamatinib has been investigated for the treatment and basic science of Rheumatoid Arthritis and Immune Thrombocytopenic Purpura (ITP). It was approved on April 17, 2018 under the trade name Tavalisse for use in ITP [8]. Fostamatinib has also been granted orphan drug status by the FDA [8].
Fostamatinib is indicated for use in the treatment of chronic immune thrombocytopenia (ITP) in patients who have had insufficient response to previous therapy [Label].
Syk is a protein tyrosine kinase associated with various inflammatory cells, including macrophages, which are presumed to be the cells responsible for ITP platelet clearance.[3] When FcγRs I, IIA, and IIIA bind to their ligands, the receptor complex becomes activated and triggers the phosphorylation of the immunoreceptor-activating motifs (ITAMs). This leads to various genes becoming activated, which causes a cytoskeletal rearrangement that mediates phagocytosis in cells of the monocyte/macrophage lineage. Because Syk plays an important role in FcγR-mediated signal transduction and inflammatory propagation, it is considered a good target for the inhibition of various autoimmune conditions, including rheumatoid arthritis and lymphoma.
Fostamatinib has been in clinical trials for rheumatoid arthritis, autoimmune thrombocytopenia, autoimmune hemolytic anemia, IgA nephropathy, and lymphoma.[1][4]
The investigation of fostamatinib began with studies involving the treatment of mouse models with cytopenia. Mice were used to measure the effectiveness of R788, a small molecule prodrug of the biologically active R406, a Syk inhibitor. In animal models, treatment with R406/R788 was shown to be safe and effective in reducing inflammation and joint damage in immune-mediated rheumatoid arthritis. The models responded favorably to treatment so the study progressed to Phase 2 trials involving humans. Human studies have shown that R788 has good oral bioavailability, biologic activity, is well tolerated, and does not exhibit collagen or ADP-induced platelet aggregation. In NCT00706342, 16 adults with chronic ITP were entered into an open-label, single-arm cohort dose-escalation trials beginning with 75 mg and rising to 175 mg twice a day. The dose was increased until a persistent response was evident, toxicity was reached, or 175 mg twice a day was met. 8 patients achieved persistent responses with platelet counts greater than 50,000 mm3/L on more than 67% of their visits. 3 of these patients had not persistently responded to thrombopoietic agents. 4 others had nonsustained responses. Mean peak platelet count exceeded 100,000 mm3/L in these 12 patients. Toxicity was evidenced primarily in GI-related side effects, notable diarrhea, urgency, and vomiting. 2 patients developed transaminitis.[5]
A phase II study of rheumatoid arthritis patients failing to respond to a biologic agent showed little efficacy as compared to placebo, but the drug was well tolerated. In patients with high inflammatory burden, measured by levels of C-reactive protein, ACR20 was achieved by a significantly higher portion of those in the fostamatinib group (42%) versus the placebo group (26%).[6]
Immune thrombocytopenic purpura (ITP) is an autoimmune disease where the immune system attacks and destroys platelets in the blood, causing abnormally low platelet counts. It is characterized by the antibody-mediated destruction of platelets. Patients with ITP have accelerated clearance of circulating IgG-coated platelets via Fcγ receptor-bearing macrophages in the spleen and liver, leading to different levels of thrombocytopenia and variable degrees of mucocutaneous bleeding.[7] Recent studies of ITP pathophysiology suggest decreased platelet production may also be an important component of the thrombocytopenia. Many patients exhibit responses to established therapies, including corticosteroids, IV immunoglobulin, anti-D, splenectomy, and rituximab. However, there are a significant minority of patients who retain persistently low platelet counts despite treatment. These patients are consistently at risk of intracranial hemorrhage and other bleeding complications. Several thrombopoiesis-stimulating therapies including eltrombopag and AMG 531 are being investigated to help combat low platelet counts in ITP patients. Rigel reported results from two Phase III clinical trials for fostamatinib as an ITP treatment in August and October 2016. The study is the second Phase 3, multi-center, randomized, double-blind, placebo controlled, study of fostamatinib disodium in the treatment of persistent/chronic immune thrombocytopenic purpura that Rigel has conducted. Primary outcome measures are defined as a stable platelet response by the end of the study (week 24) of at least 50,000/µL on at least 4 of the 6 visits between weeks 14-24. Participants received either a placebo, 100 mg, or 150 mg of the drug in the morning and evening for 24 full weeks. The first study, FIT 1 (047) met the primary endpoint in a statistically significant manner, with 18% of patients hitting the 50,000 platelets/µL of blood and no patients receiving the placebo meeting that criteria. As of June 2016, the open-label, long term extension study (049) is currently tracking 118 patients who opted to receive fostamatinib after completing either study 047 or 048.[8]
Approval for treatment of autoimmune hemolytic anemia (AIHA) is in Stage 1 of Phase II trials. This study is a Phase 2, multi-center, open label, Simon two-stage study to evaluate the safety and efficacy of fostamatinib disodium in the treatment of warm antibody autoimmune hemolytic anemia. Primary outcome measures examined include a hemoglobin response measured by levels higher than 10 g/dL and 2 g/dL higher than the baseline hemoglobin. Responses were studied for a period of 12 weeks and for a dose of 150 mg in the morning and evening. The study began in April 2016 and is estimated to conclude in September 2017. The study is currently recruiting participants from U.S. states including Arizona, California, D.C., Massachusetts, New York, North Carolina, and Texas. Subjects must have had a diagnosis of primary or secondary warm antibody AIHA, and must have failed at least 1 prior treatment regimen for AIHA. Subjects cannot have a platelet count less than 30,000/µL, have AIHA secondary to autoimmune disease, have uncontrolled or poorly controlled hypertension, or have cold antibody AIHA, cold agglutinin syndrome, mixed type AIHA, or paroxysmal cold hemoglobinuria.[9]
Fostamatinib as a treatment for IgA nephropathy (IgAN) is in Phase II trials, which will conclude at the end of 2016. IgAN is a chronic autoimmune disease associated with inflammation in the kidneys that reduces their ability to successfully filter blood. There are currently no disease-targeted therapies for IgAN. Participants are currently being recruited from the US, Austria, Germany, Hong Kong, Taiwan, and the UK. Patients must be between 18 and 70 years old, have renal biopsy findings consistent with IgA nephropathy, have been treated with an Angiotensin Converting Enzyme inhibitor (ACEi) and/or an Angiotensin II Receptor Blocker (ARB) for at least 90 days at the maximum approved dose, have a proteinuria > 1 gm/day at diagnosis of IgA nephropathy and a level > 0.5 gm/day at the second screening visit, and a blood pressure controlled to ≤ 1302/80 with angiotensin blockade. Eligible candidates cannot have recently used cyclophosphamide, mycophenolate mofetil, azathioprine, Rituximab, or > 15 mg/day of prednisone or any other corticosteroid equivalent. The study investigates whether fostamatinib is a safe and effective treatment for IgAN. It is a Phase 2, multi-center, randomized, double-blind, ascending-dose, placebo-controlled clinical study. Primary outcome measures include the mean change in proteinuria as measured by spot urine protein/creatinine ratio (sPCR). Effects were evaluated for 100 mg, 150 mg, and placebo formulations taken twice daily by mouth for 24 weeks. The study began in October 2014 and is expected to complete by June 2017.[10]

PATENTS
https://patents.google.com/patent/WO2008064274A1/en14
Suitable active 2,4-pyrimidinediamine compounds are described, for example, in U.S. application Serial No. 10/355,543 filed January 31 , 2003 (US2004/0029902A1), international application Serial No. PCT/US03/03022 filed January 31, 2003 (WO 03/063794), U.S. application Serial No. 10/631,029 filed July 29, 2003 (US 2005/0028212), international application Serial No. PCT/US03/24087 (WO2004/014382), U.S. application Serial No. 10/903,263 filed July 30, 2004 (US2005/0234049), and international application Serial No.
PCT/US2004/24716 (WO 2005/016893), the disclosures of which are incorporated herein by reference. In such 2,4-pyrimidinediamine compounds, the progroup(s) Rp can be attached to any available primary or secondary amine, including, for example, the N2 nitrogen atom of the 2,4-pyrimidinediamine moiety, the N4 nitrogen atom of the 2,4-pyrimidinediamine moiety, and/or a primary or secondary nitrogen atom included in a substituent on the 2,4-pyrimidinediamine compound. The use of phosphate-containing progroups Rp is especially useful for 2,4-pyrimidinediamine compounds that exhibit poor water solubility under physiological conditions (for example, solubilities of less than about 10 μg/ml). While not intending to be bound by any theory of operation, it is believed that the phosphate-containing progroups aid the solubility of the underlying active 2,4-pyrimidinediamine compound, which in turn increases its bioavailability when administered orally. It is believed that the phosphate progroups Rp are metabolized by phosphatase enzymes found in the digestive tract, permitting uptake of the underlying active drug.
[0024] It has been discovered that the water solubility and oral bioavailability of a particular biologically active 2,4-pyrimidinediamine compound, illustrated below (Compound 1), increased dramatically when formulated to include a progroup Rp of the formula -CH2-O-P(O)(OH)2 at the ring nitrogen atom highlighted with the asterisk (Compound 4):
Compound 4
EXAMPLES
1. Synthesis of Prodrug Compound 4
1.1 N4-(2,2-dimethyl-4-[(di-tert-butyl phosphonoxy)methyl]-3- oxo-5-pyrido[l,4]oxazin-6-yl)-5-fluoro-N2-(3,4,5- trimethoxyphenyl)-2,4-pyrimidinediamine (Compound 3)
4 days
[0260] N4-(2,2-dimethyl-3-oxo-4H-5-pyrido[l,4]oxazin-6-yl)-5-fluoro-N2-(3,4,5-trimethoxyphenyl)-2,4-pyrimidinediamine (1, 1.0 g, 2.12 mmol), Cs2CO3 (1.0 g, 3.07 mmol) and di-tert-butyl chloromethyl phosphate (2, 0.67 g, 2.59 mmol) in acetone (20 mL) was stirred at room temperature under nitrogen atmosphere. Progress of the reaction was monitored by LC/MS. Crude reaction mixture displayed three product peaks with close retention times with M++H 693 (minor-1), 693 (major; 3) and 477 (minor-2) besides starting material (Compound 1). Upon stirring the contents for 4 days (70% consumption), the reaction mixture was concentrated and diluted with water. The resultant pale yellow precipitate formed was collected by filtration and dried. The crude solid was purified by silica gel (pretreated with 10%NEt3/CH2Cl2 followed by eluting with hexanes) column chromatography by gradient elution with 70% EtOAc / hexanes-100% EtOAc). The fractions containing Compound 1 and M++H 693 were collected and concentrated. The resulting crude white solid was subjected to repurifϊcation in the similar manner as described previously but by eluting with 30%-50%-75%-100% EtOAc/hexanes. The major product peak with M++H 693 was collected as a white solid (270 mg, 18%) and was characterized as N4-(2,2-dimethyl-4-[(di-tert-butyl phosphonoxy)methyl]-3-oxo-5-pyrido[l,4]oxazin-6-yl)-5-fluoro-N2-(3,4,5-trimethoxyphenyl)-2,4-pyrimidinediamine (Compound 3). 1H NMR (DMSO-d6): δ 9.21 (s, IH), 9.17 (s, IH), 8.16 (d, IH, J = 2.6 Hz), 7.76 (d, IH, J = 8.5 Hz), 7.44 (d, IH, J = 8.5 Hz), 7.02 (s, 2H), 5.78 (d, IH, J3PH = 6.1 Hz), 3.64 (s, 6H), 3.58 (s, 3H), 1.45 (s, 6H), 1.33 (s, 9H). LCMS: ret. time: 14.70 min.; purity: 95%; MS (m/e): 693 (MH+). 31P NMR (DMSO-d6): -11.36.
1.2. N4-(2,2-dimethyl-4-[(dihydrogen phosphonoxy)methyl]-3- oxo-5-pyrido[l,4]oxazin-6-yl)-5-fluoro-N2-(3,4,5- trimethoxyphenyl)-2,4-pyrimidinediamine (Compound 4)
[0261] Trifluoroacetic acid (1.5 mL) was added dropwise as a neat for 5 min to N4-(2,2-dimethyl-4-[(di-tert-butyl phosphonoxy)methyl]-3-oxo-5-pyrido[l,4]oxazin-6-yl)-5-fluoro-N2-(3,4,5-trimethoxyphenyl)-2,4-pyrimidinediamine (Compound 3, 120 mg, 0.173 mmol ) dissolved in CH2Cl2 (10 mL) at 00C under nitrogen atmosphere. The contents were allowed to stir for 1.5 h. Progress of the reaction mixture was monitored by LC/MS. After complete consumption of the starting material, reaction mixture was concentrated, dried and triturated with ether. The ethereal layer was decanted and dried to provide the crude solid. LC/MS analysis of the crude displayed three peaks with M++H 581, 471 and 501. The peak corresponding to M++H 581 was collected by preparative HPLC chromatographic purification. The fractions were lyophilised and dried to provide 53 mg (52%) of off white fluffy solid and characterized as N4-(2,2-dimethyl-4-[(dihydrogen phosphonoxy)methyl]-3-oxo-5-pyrido[ 1 ,4]oxazin-6-yl)-5-fluoro-N2-(3,4,5-trimethoxyphenyl)-2,4-pyrimidinediamine (Compound 4). 1H NMR (DMSO-d6): δ 9.21 (br s, 2H), 8.16 (d, IH, J = 2.6 Hz), 7.93 (d, IH, J = 8.5 Hz), 7.39 (d, IH, J = 8.5 Hz), 7.05 (s, 2H), 5.79 (d, IH, J3PH = 6.6 Hz), 3.67 (s, 6H), 3.59 (s, 3H), 1.44 (s, 6H). LCMS: ret. time: 8.52 min.; purity: 95%; MS (m/e): 581 (MH+). 31P NMR (DMSO-d6): -2.17.
2. Alternative Synthesis of Prodrug Compound 4
[0262] An alternative method of synthesizing prodrug Compound 4 which alleviates the need for column chromatography and HPLC purification is provided below.
2.1 Synthesis of N4-(2,2-dimethyl-4- [(di-tert-butyl
phosphonoxy)methyl] -3-oxo-5-pyrido [ 1 ,4] oxazin-6-yl)-5- fluoro-N2-(3,4,5-trimethoxyphenyl)-2,4-pyrimidinediamine
(Compound 3)
rt
92% conversion
majoπminor 6.5:1
[0263] N4-(2,2-dimethyl-3-oxo-4H-5-pyrido[l,4]oxazin-6-yl)-5-fluoro-N2-(3,4,5-trimethoxyphenyl)-2,4-pyrimidinediamine (Compound 1, 19.73 g, 41.97 mmol),
Cs2CO3 (15.04 g, 46.16 mmol) and di-tert-butyl chloromethyl phosphate (13.0 g, 50.38 mmol) in DMF (100 mL) was stirred at room temperature under nitrogen atmosphere. Progress of the reaction was monitored by in process LC/MS. Crude reaction mixture displayed two product peaks (ratio 1 :6.5) with close retention times displaying M++H 693 (minor) and 693 (major) besides starting material (Compound 1). Initial yellow reaction mixture turned to olive green as the reaction progressed. Workup was carried out as follows 1). Upon stirring the contents for 30 h (92% consumption), reaction mixture was poured onto ice-water (400 mL) and stirred the contents by adding brine solution (200 mL). Fine yellow tan solid formed was filtered, washed with water and dried overnight.
2). The solid (35 g) was dissolved in MTBE (500 mL) and washed with water (40OmL). Aqueous layer was extracted with MTBE (2 X 350 mL) till the absence of UV on TLC. Combined organic layers were dried over anhydrous Na2SO4 and decanted.
Note: step 2 can be done directly, however, DMF extraction back into solution leads to difficulty in the crystallization step.
3). The dark red clear solution was subjected to 10 g of activated charcoal treatment, heated to boil and filtered.
4). The dark red clear solution was concentrated by normal heating to 400 mL of its volume and left for crystallization. The solid crystallized as granules was filtered, crushed the granules to powder, washed with MTBE (400 mL) and dried under high vacuum. See step 7 for the workup of mother liquor. Weight of the solid: 17 g; purity: 90% (Compound 3), 6.26% (Compound 1), 1.8% (minor M+ 693).
5). At this stage solid was taken in 500 ml of ethyl ether and heated to boil. Cooled and filtered to remove undissolved material. Filtrate was concentrated.
6). Above concentrate was subjected to crystallization in MTBE (300 mL).
The white solid formed was filtered, washed with MTBE (100 mL) and dried under high vacuum to provide the desired N4-(2,2-dimethyl-4-[(di-tert-butyl
phosphonoxy)methyl]-3-oxo-5-pyrido[l,4]oxazin-6-yl)-5-fluoro-N2-(3,4,5-trimethoxyphenyl)-2,4-pyrimidinediamine (Compound 3) in 97% purity. 1H NMR (DMSO-d6): δ 9.21 (s, IH), 9.17 (s, IH), 8.16 (d, IH, J = 2.6 Hz), 7.76 (d, IH, J = 8.5 Hz), 7.44 (d, IH, J = 8.5 Hz), 7.02 (s, 2H), 5.78 (d, IH, J3PH = 6.1 Hz), 3.64 (s, 6H), 3.58 (s, 3H), 1.45 (s, 6H), 1.33 (s, 9H). LCMS: ret. time: 14.70 min.; purity: 95%; MS (m/e): 693 (MH+). 31P NMR (DMSO-d6): -11.36. Weight of the solid: 15.64 g (yield: 55%); purity: 97% (Compound 3), 3% (Compound 1).
7). The mother liquor was concentrated and steps 5 and 6 were repeated to provide Compound 3.
2.2. Synthesis of N4-(2,2-dimethyl-4-[(dihydrogen
phosphonoxy)methyl] -3-oxo-5-pyrido [ 1 ,4] oxazin-6-yl)-5- fluoro-N2-(3,4,5-trimethoxyphenyl)-2,4-pyrimidinediamine
(Compound 4)
[0264] N4-(2,2-dimethyl-4-[(di-tert-butyl phosphonoxy)methyl]-3-oxo-5-pyrido[l,4]oxazin-6-yl)-5-fluoro-N2-(3,4,5-trimethoxyphenyl)-2,4-pyrimidinediamine (Compound 3); (15.0 g, 21.67 mmol) dissolved in AcOH:H20 (225 niL, 4:1) was heated at 65 0C (oil bath temp). The progress of the reaction was monitored by in process LC/MS. The reaction mixture transformed to faint tan white solid after Ih of heating. At this point most of Compound 3 converted to mono des t-butyl product. After 3h of heating, consumption of SM and complete conversion of intermediate (mono des t-butylated) to product was observed.
[0265] Reaction mixture was cooled, poured onto ice-water (200 mL), stirred for 20 min and filtered. The clear white filter cake was washed with water (600 ml) and acetone (200 mL) successively, dried for 2h followed by drying under high vacuum over P2O5 in a desiccator. Weight of the solid: 12.70 g; purity: 97% (Compound 3) and 3% (Compound 1) 1H NMR indicated acetic acid presence (1 :1)
[0266] To remove acetic acid, the solid was taken in acetonitrile (300 mL) and concentrated by rotovap vacuum. This process was repeated 2 times with acetonitrile and toluene (3 X 300 mL). The solid obtained was dried under high vacuum at 50 OC. [0267] Finally, the solid was taken in acetone (400 mL), filtered and dried to provide solid N4-(2,2-dimethyl-4-[(dihydrogen phosphonoxy)methyl]-3-oxo-5-pyrido[l,4]oxazin-6-yl)-5-fluoro-N2-(3,4,5-trimethoxyphenyl)-2,4-pyrimidinediamine (Compound 4). 1H NMR (DMSO-d6): δ 9.21 (br s, 2H), 8.16 (d, IH, J = 2.6 Hz), 7.93 (d, IH, J = 8.5 Hz), 7.39 (d, IH, J = 8.5 Hz), 7.05 (s, 2H), 5.79 (d, IH, J3PH = 6.6 Hz), 3.67 (s, 6H), 3.59 (s, 3H), 1.44 (s, 6H). LCMS: ret. time: 8.52 min.; purity: 95%; MS (m/e): 581 (MH+). 31P NMR (DMSO-d6): -2.17.
3. Synthesis of N4-(2,2-dimethyl-4-[(dihydrogen phosphonoxy)methyl]-3-oxo- 5-pyrido[l,4]oxazin-6-yl)-5-fluoro-N2-(3,4,5-trimethoxyphenyl)-2,4- pyrimidinediamine mono calcium salt (Prodrug Salt 6)
[0268] Aqueous (10 niL) NaHCO3 (0.17 g, 2.02 mmol) solution was added dropwise to a suspension of N4-(2,2-dimethyl-4-[(dihydrogen phosphonoxy)methyl]-3-oxo-5-pyrido[l,4]oxazin-6-yl)-5-fluoro-N2-(3,4,5-trimethoxyphenyl)-2,4-pyrimidinediamine (0.5 g, 0.86 mmol) in water (5 mL) at room temperature while stirring the contents. The clear solution formed was treated with aqueous (10 mL) CaCl2 (0.11 g in 10 mL water, 0.99 mmol) n a dropwise manner at room temperature. The addition resulted in the precipitation of a white solid from reaction mixture. Upon completion of addition, the contents were stirred for a period of 30 min, filtered, washed with water (40 mL) and dried. The clear white solid was taken in water (30 mL) and heated on a stir plate to boil. The solution was cooled, filtered and dried. The white solid collected and further dried under high vacuo at 80 0C for 32 h to provide 0.41 g (83%) of solid N4-(2,2-dimethyl-4-[(dihydrogen phosphonoxy)methyl]-3-oxo-5-pyrido[ 1 ,4]oxazin-6-yl)-5-fluoro-N2-(3,4,5-trimethoxyphenyl)-2,4-pyrimidinediamine mono calcium salt (Prodrug Salt 6).
[0269] Ca(OAc)2 may also used in place Of CaCl2 in this preparation.
4. Synthesis of N4-(2,2-dimethyl-4-[(dihydrogen phosphonoxy)methyl]-3-oxo- 5-pyrido[l,4]oxazin-6-yl)-5-fluoro-N2-(3,4,5-trimethoxyphenyl)-2,4- pyrimidinediamine disodium salt hexahydrate and monosodium salt
hydrate
[0270] A round-bottomed flask was charged with 10.00 g N4-(2,2-dimethyl-4-[(dihydrogen phosphonoxy)methyl]-3-oxo-5-pyrido[ 1 ,4]oxazin-6-yl)-5-fluoro-N2-(3,4,5-trimethoxyphenyl)-2,4-pyrimidinediamine (Compound 4) and 140 mL water into a round bottom flask to form a slurry having a pH between 3.6 and 3.7. The pH was adjusted to in the range of 9.3 to 10.3 by addition of 1 M aqueous NaOH, initially forming a turbid solution, which returned to a suspension upon prolonged stirring. The mixture was heated at reflux, then the turbid solution was hot filtered through filter paper. The solid collected in the filter paper was rinsed with 10 mL hot water.
Isopropanol (75 mL) was added to the filtrate, yielding a clear solution, which was allowed to cool to room temperature over about 1.5 hours with stirring, during which time a solid precipitated. The precipitate was collected by filtration, rinsed with 47 mL isopropanol, and taken up in 73 mL acetone to form a slurry, which was stirred for 1.5 hours at room temperature. The solid was again collected by filtration and rinsed with 18 mL acetone, then dried at about 40 0C under vacuum until substantially all isopropanol and acetone was removed (i.e., below 0.5 wt% each). The product was exposed to air at about 40% relative humidity and room temperature until the water content stabilized at about 15% by Karl Fisher titration, yielding 8.18 g of the title compound. 1H NMR (D2O): δ 7.67 (d, IH, J = 3.8 Hz), 7.49 (d, IH, J = 8.8 Hz), 6.87 (d, IH, J = 8.8 Hz), 6.50 (s, 2H), 5.52 (d, IH, J3PH = 2.0 Hz), 3.53 (s, 3H), 3.47 (s, 6H), 1.32 (s, 6H). 31P NMR (D2O): 2.75. The prodrug salt hydrate was obtained as a pure-white, highly crystalline material. Microscopic investigation indicated that the crystallites are plate-like with a particle size of less than 10 μm. Polarized light microscopy revealed birefringence corroborating the crystalline nature of the hydrate. [0271] The monosodium salt can be prepared from N4-(2,2-dimethyl-4-[(dihydrogen phosphonoxy)methyl]-3-oxo-5-pyrido[l,4]oxazin-6-yl)-5-fiuoro-N2-(3,4,5-trimethoxyphenyl)-2,4-pyrimidinediamine and sodium hydroxide by a proper pH control; pH of 5-5.5 results in predominantly the formation of monosodium salt.
 |
|
| Clinical data | |
|---|---|
| Trade names | Tavalisse |
| Synonyms | Fostamatinib disodium hexahydrate, tamatinib fosdium, R-788, NSC-745942, R-935788 |
| MedlinePlus | a618025 |
| Pregnancy category |
|
| Routes of administration |
by mouth |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 55% (tamatinib metabolite) |
| Protein binding | 98% (tamatinib metabolite) |
| Metabolism | Gut (ALP to tamatinib) Liver (tamatinib metabolite by CYP3A4, UGT1A9) |
| Elimination half-life | 15 hours |
| Excretion | faecal (80%), urine (20%) |
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ECHA InfoCard | 100.125.771 |
| Chemical and physical data | |
| Formula | C23H26FN6O9P |
| Molar mass | 580.47 g/mol |
| 3D model (JSmol) | |
///////////SQ8A3S5101, TAVALISSE , фостаматиниб , وستاماتينيب , 福他替尼 , FDA 2018, fostamatinib disodium hexahydrate, fostamatinib , ホスタマチニブジナトリウム水和物
COC1=CC(NC2=NC=C(F)C(NC3=NC4=C(OC(C)(C)C(=O)N4COP(O)(O)=O)C=C3)=N2)=CC(OC)=C1OC


Tafenoquine
タフェノキン
N-[2,6-dimethoxy-4-methyl-5-[3-(trifluoromethyl)phenoxy]quinolin-8-yl]pentane-1,4-diamine
Medicines for Malaria Venture
Walter Reed Army Institute (Originator)
PATENT US 4617394
Synonyms
New Drug Application (NDA): 210795
Company: GLAXOSMITHKLINE
FDA approved on July 20, 2018
FDA
Orphan
This new drug application provides for the use of KRINTAFEL (tafenoquine) tablets for the radical cure (prevention of relapse) of Plasmodium vivax malaria in patients aged 16 years and older who are receiving appropriate antimalarial therapy for acute P. vivax infection….https://www.accessdata.fda.gov/drugsatfda_docs/appletter/2018/210795Orig1s000Ltr.pdf
Tafenoquine under the commercial name of Krintafel is an 8-aminoquinoline drug manufactured by GlaxoSmithKline that is being investigated as a potential treatment for malaria, as well as for malaria prevention.[2][3]
The proposed indication for tafenoquine is for treatment of the hypnozoite stages of Plasmodium vivax and Plasmodium ovale that are responsible for relapse of these malaria species even when the blood stages are successfully cleared. This is only now achieved by administration of daily primaquine for 14 days. The main advantage of tafenoquine is that it has a long half-life (2–3 weeks) and therefore a single treatment may be sufficient to clear hypnozoites. The shorter regimen has been described as an advantage.[4]
Like primaquine, tafenoquine causes hemolysis in people with G6PD deficiency.[2] Indeed, the long half-life of tafenoquine suggests that particular care should be taken to ensure that individuals with severe G6PD deficiency do not receive the drug.
The dose of tafenoquine has not been firmly established, but for the treatment of Plasmodium vivax malaria, a dose of 800 mg over three days has been used.[5]

In 2018 United States Food and Drug Administration (FDA) approved single dose tafenoquine for the radical cure (prevention of relapse) of Plasmodium vivax malaria[6].
Tafenoquine is used for the treatment and prevention of relapse of Vivax malaria in patients 16 years and older. Tafenoquine is not indicated to treat acute vivax malaria.[1]
Malaria is a disease that remains to occur in many tropical countries. Vivax malaria, caused by Plasmodium vivax, is known to be less virulent and seldom causes death. However, it causes a substantive illness-related burden in endemic areas and it is known to present dormant forms in the hepatocytes named hypnozoites which can remain dormant for weeks or even months. This dormant form produces ongoing relapses
The US Food and Drug Administration (FDA) has approved, under Priority Review, GlaxoSmithKline (GSK)’s tafenoquine (Krintafel), which is the first single-dose medicine for the prevention of Plasmodium vivax (P vivax) malaria relapse in patients over the age of 16 years who are receiving antimalarial therapy. This is the first drug to be approved for the treatment of P vivax in over 60 years.
“[The] approval of Krintafel, the first new treatment for Plasmodium vivax malaria in over 60 years, is a significant milestone for people living with this type of relapsing malaria.” Hal Barron, MD, chief scientific officer and president of research and development of GSK, said in the announcement, “Together with our partner, Medicines for Malaria Venture (MMV), we believe Krintafel will be an important medicine for patients with malaria and contribute to the ongoing effort to eradicate this disease.”
Tafenoquine is an 8-aminoquinoline derivative with activity against all stages of the P vivax lifecycle, including hypnozoites. It was first synthesized by scientists at the Walter Reed Army Institute of Research in 1978, and in 2008, GSK entered into a collaboration with MMV, to develop tafenoquine as an anti-relapse medicine.
After an infected mosquito bite, the P vivax parasite infects the blood and causes an acute malaria episode and can also lie dormant in the liver (in a form known as hypnozoite) from where it periodically reactivates to cause relapses, which can occur weeks, months, or years after the onset of the initial infection. The dormant liver forms cannot be readily treated with most anti-malarial treatments. Primaquine, an 8-aminoquinolone, has been the only FDA-approved medicine that targeted the dormant liver stage to prevent relapse; however, effectiveness only occurs after 14 days and the treatment has shown to have poor compliance.
“The US FDA’s approval of Krintafel is a major milestone and a significant contribution towards global efforts to eradicate malaria,” commented David Reddy, PhD, chief executive officer of MMV in a recent statement, “The world has waited decades for a new medicine to counter P vivax malaria relapse. Today, we can say the wait is over. Moreover, as the first ever single-dose for this indication, Krintafel will help improve patient compliance.”
Approval for tafenoquine was granted based on the efficacy and safety data gleaned from a comprehensive global clinical development program for P vivaxprevention of relapse which has been designed by GSK and MMV in agreement with the FDA. The program consisted of 13 studies assessing the safety of a 300 mg single-dose of tafenoquine, including 3 double-blind studies referred to as DETECTIVE Parts 1 and 2 and GATHER.
With the approval of tafenoquine, GSK has also been awarded a tropical disease priority review voucher by the FDA. Additionally, GSK is waiting for a decision from Australian Therapeutics Good Administration regarding the regulatory submission for the drug.
P vivax malaria has caused around 8.5 million clinical infections each year, primarily in South Asia, South-East Asia, Latin America, and the Horn of Africa, a peninsula in East Africa. Symptoms include fever, chills, vomiting, malaise, headache and muscle pain, and can lead to death in severe cases.
Tafenoquine should not be administered to: patients who have glucose-6-phosphate dehydrogenase (G6PD) deficiency or have not been tested for G6PD deficiency, patients who are breastfeeding a child known to have G6PD deficiency or one that has not been tested for G6PD deficiency, or patients who are allergic to tafenoquine or any of the ingredients in tafenoquine or who have had an allergic reaction to similar medicines containing 8-aminoquinolines
Tafenoquine contains a stereocenter and consists of two enantiomers. This is a mixture of (R) – and the (S) – Form:
| Enantiomers of tafenoquine | |
|---|---|
 (R)-Form |
 (S)-Form |
 CLIP
CLIPUS 4431807


Nitration of 1,2-dimethoxybenzene (XXIX) with HNO3/AcOH gives 4,5-dimethoxy-1,2-dinitrobenzene (XXX), which is treated with ammonia in hot methanol to yield 4,5-dimethoxy-2-nitroaniline (XXXI). Cyclization of compound (XXXI) with buten-2-one (XXXII) by means of H3PO4 and H3AsO4 affords 5,6-dimethoxy-4-methyl-8-nitroquinoline (XXXIII), which is selectively mono-demethylated by means of HCl in ethanol to provide 5-hydroxy-6-methoxy-4-methyl-8-nitroquinoline (XXXIV). Reaction of quinoline (XXXIV) with POCl3 gives the corresponding 5-chloro derivative (XXXV), which is condensed with 3-(trifluoromethyl)phenol (IV) by means of KOH to yield the diaryl ether (XXXVI). Finally, the nitro group of (XXXVI) is reduced by means of H2 over PtO2 in THF or H2 over Raney nickel.

Nitration of 2-fluoroanisole (XXXVII) with HNO3/Ac2O gives 3-fluoro-4-methoxynitrobenzene (XXXVIII), which is reduced to the corresponding aniline (XXXIX) with SnCl2/HCl. Reaction of compound (XXXIX) with Ac2O yields the acetanilide (XL), which is nitrated with HNO3 to afford 5-fluoro-4-methoxy-2-nitroacetanilide (XLI). Hydrolysis of (XLI) with NaOH provides 5-fluoro-4-methoxy-2-nitroaniline (XLII), which is cyclized with buten-2-one (XXXII) by means of As2O5 and H3PO4 to furnish 5-fluoro-6-methoxy-4-methyl-8-nitroquinoline (XLIII). Condensation of quinoline (XLIII) with 3-(trifluoromethyl)phenol (IV) by means of K2CO3 gives the diaryl ether (XXXIV), which is finally reduced by means of H2 over PtO2 in THF.
CLIP
US 4617394

Reaction of 8-amino-6-methoxy-4-methyl-5-[3-(trifluoromethyl)phenoxy]quinoline (XIV) with phthalic anhydride (XV) affords the phthalimido derivative (XVI), which is oxidized with MCPBA to yield the quinoline N-oxide (XVII). Treatment of compound (XVII) with neutral alumina gives the quinolone derivative (XVIII), which by reaction with POCl3 in refluxing CHCl3 provides the 2-chloroquinoline derivative (XIX). Alternatively, reaction of the quinoline N-oxide (XVII) with POCl3 as before also gives the 2-chloroquinoline derivative (XIX) The removal of the phthalimido group of compound (XIX) by means of hydrazine in refluxing ethanol gives the chlorinated aminoquinoline (XX), which is finally treated with MeONa in hot DMF.
CLIP
US 6479660; WO 9713753

Chlorination of 6-methoxy-4-methylquinolin-2(1H)-one (I) with SO2Cl2 in hot acetic acid gives the 5-chloro derivative (II), which is nitrated with HNO3 in H2SO4 to yield the 8-nitroquinolinone (III). Condensation of compound (III) with 3-(trifluoromethyl)phenol (IV) by means of KOH in NMP provides the diaryl ether (V), which is treated with refluxing POCl3 to afford the 2-chloroquinoline (VI). Reaction of compound (VI) with MeONa in refluxing methanol results in the 2,6-dimethoxyquinoline derivative (VII), which is reduced with hydrazine over Pd/C to give the 8-aminoquinoline derivative (VIII). Condensation of aminoquinoline (VIII) with N-(4-iodopentyl)phthalimide (IX) by means of diisopropylamine in hot NMP yields the phthalimido precursor (X), which is finally cleaved with hydrazine in refluxing ethanol.


Reaction of 1,4-dibromopentane (XI) with potassium phthalimide (XII) gives N-(4-bromopentyl)phthalimide (XIII), which is then treated with NaI in refluxing acetone.

Reaction of 4-methoxyaniline (XXI) with ethyl acetoacetate (XXII) by means of triethanolamine in refluxing xylene gives the acetoacetanilide (XXIII), which is cyclized by means of hot triethanolamine and H2SO4 to yield 6-methoxy-4-methylquinolin-2(1H)-one (I), which is treated with refluxing POCl3 to provide 2-chloro-6-methoxy-4-methylquinoline (XXIV). Reaction of compound (XXIV) with SO2Cl2 in hot AcOH affords 2,5-dichloro-6-methoxy-4-methylquinoline (XXV), which is treated with MeONa in refluxing methanol to furnish 5-chloro-2,6-dimethoxy-4-methylquinoline (XXVI). Alternatively, the reaction of compound (XXIV) with MeONa as before gives 2,6-dimethoxy-4-methylquinoline (XXVII), which is treated with SO2Cl2 in hot AcOH to give the already described 5-chloro-2,6-dimethoxy-4-methylquinoline (XXVI). Nitration of compound (XXVI) with KNO3 and P2O5 gives the 8-nitroquinoline derivative (XXVIII), which is condensed with 3-(trifluoromethyl)phenol (IV) by means of KOH in hot NMP to yield the diaryl ether (VII). Finally, the nitro group of compound (VII) is reduced with hydrazine over Pd/C.
PAPER
http://pubs.rsc.org/en/Content/ArticleLanding/2017/RA/C7RA04867J#!divAbstract
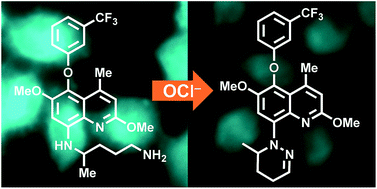
Tafenoquine (TQ), a fluorescent antimalarial drug, was used as a receptor for the fluorometric detection of hypochlorite (OCl−). TQ itself exhibits a strong fluorescence at 476 nm, but OCl−-selective cyclization of its pentan-1,4-diamine moiety creates a blue-shifted fluorescence at 361 nm. This ratiometric response facilitates rapid, selective, and sensitive detection of OCl− in aqueous media with physiological pH. This response is also applicable to a simple test kit analysis and allows fluorometric OCl− imaging in living cells.



1 H NMR (300 MHz, CDCl3, TMS) d (ppm): 7.32 (q, 1H, J ¼ 18 Hz), 7.21 (d, 1H, J ¼ 6 Hz), 7.07 (s, 1H), 6.94 (d, 1H, J ¼ 6 Hz), 6.64 (s, 1H), 6.50 (s, 1H), 5.84 (d, 1H, J ¼ 6 Hz), 4.00 (s, 3H), 3.79 (s, 3H), 3.66 (s, 1H), 2.78 (d, 2H, J ¼ 6 Hz), 2.55 (s, 3H), 1.69 (dd, 6H, J ¼ 6 Hz, J ¼ 9 Hz), 1.35 (d, 3H, J ¼ 6 Hz).
13C NMR (100 MHz, CDCl3, TMS) d (ppm): 159.64, 148.961, 146.339, 142.010, 132.085, 131.760, 131.007, 129.968, 126.917, 125.344, 122.636, 120.681, 118.006, 115.256, 112.052, 94.996, 56.989, 52.870, 48.446, 42.248, 34.439, 30.130, 23.103, 20.833.
MS (m/z): M+ calcd for C24H28F3N3O3: 463.2083; found (ESI): 464.17 (M + H)+ .
PAPER
J Med Chem 1989,32(8),1728-32
https://pubs.acs.org/doi/pdf/10.1021/jm00128a010

Synthesis of the intermediate diazepinone (IV) is accomplished by a one-pot synthesis. Condensation of 2-chloro-3-aminopyridine (I) with the anthranilic ester (II) is effected in the presence of potassium tert-butoxide as a catalyst. The resulting anthranilic amide (III) is cyclized under the influence of catalytic amounts of sulfuric acid. Treatment of (IV) with chloroacetylchloride in toluene yields the corresponding choroacetamide (V). The side chain of AQ-RA 741 is prepared starting from 4-picoline, which is alkylated by reaction with 3-(diethylamino)propylchloride in the presence of n-butyllithium. Hydrogenation of (VIII) using platinum dioxide as a catalyst furnishes the diamine (IX), which is coupled with (V) in the presence of catalytic amounts of sodium iodide in acetone leading to AQ-RA 741 as its free base.




|
8-3-2007
|
Methods and compositions for treating diseases associated with pathogenic proteins
|
|
|
12-6-2006
|
Process for the preparation of quinoline derivatives
|
|
|
3-14-2002
|
PROCESS FOR THE PREPARATION OF ANTI-MALARIAL DRUGS
|
|
|
4-2-1998
|
MULTIDENTATE METAL COMPLEXES AND METHODS OF MAKING AND USING THEREOF
|
|
|
4-18-1997
|
PROCESS FOR THE PREPARATION OF ANTI-MALARIAL DRUGS
|
|
|
12-20-1996
|
MULTIDENTATE METAL COMPLEXES AND METHODS OF MAKING AND USING THEREOF
|
|
|
12-15-1993
|
Use of interferon and a substance with an antimalarial activity for the treatment of malaria infections
|
|
|
10-15-1986
|
4-methyl-5-(unsubstituted and substituted phenoxy)-2,6-dimethoxy-8-(aminoalkylamino) quinolines
|
 |
|
| Clinical data | |
|---|---|
| Synonyms | Etaquine,[1] WR 238605,[1] SB-252263 |
| ATC code |
|
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| NIAID ChemDB | |
| Chemical and physical data | |
| Formula | C24H28F3N3O3 |
| Molar mass | 463.493 g/mol |
| 3D model (JSmol) | |
OLD CLIP
April 28, 2014
GlaxoSmithKline (GSK) and Medicines for Malaria Venture (MMV) announced the start of a Phase 3 global program to evaluate the efficacy and safety of tafenoquine, an investigational medicine which is being developed for the treatment and relapse prevention (radical cure) of Plasmodium vivax (P. vivax) malaria.
P. vivax malaria, a form of the disease caused by one of several species of Plasmodium parasites known to infect humans, occurs primarily in South and South East Asia, Latin America and the horn of Africa. Severe anemia, malnutrition and respiratory distress are among the most serious consequences described to be caused by the infection.
The Phase 3 program includes two randomized, double-blind treatment studies to investigate tafenoquine in adult patients with P. vivax malaria. The DETECTIVE study (TAF112582) aims to evaluate the efficacy, safety and tolerability of tafenoquine as a radical cure for P. vivax malaria, co-administered with chloroquine, a blood stage anti-malarial treatment. The GATHER study (TAF116564) aims to assess the incidence of hemolysis and safety and efficacy of tafenoquine compared to primaquine, the only approved treatment currently available for the radical cure of P. vivax malaria.
Tafenoquine is not yet approved or licensed for use anywhere in the world.
“P. vivax malaria can affect people of all ages and is particularly insidious because it has the potential to remain dormant within the body in excess of a year, and causes some patients to experience repeated episodes of illness after the first mosquito bite,” said Nicholas Cammack, head, Tres Cantos Medicines Development Center for Diseases of the Developing World. “Our investigation of tafenoquine for the treatment of P. vivax malaria is part of GSK’s efforts to tackle the global burden of malaria. Working with our partners, including MMV, we are determined to stop malaria in all its forms.”
“One of the big challenges we face in tackling malaria is to have new medicines to prevent relapse, caused by dormant forms of P. vivax,” said Dr. Timothy Wells, MMV’s chief scientific officer. “The Phase 3 program is designed to build upon the promising results of the Phase 2b study which showed that treatment with tafenoquine prevented relapses. If successful, tafenoquine has the potential to become a major contributor to malaria elimination. It’s a great privilege to be working with GSK on this project; they have a clear commitment to changing the face of public health in the countries in which we are working.”
Umeclidinium bromide
GSK-573719A, ウメクリジニウム臭化物
Umeclidinium bromide (trade name Incruse Ellipta) is a long-acting muscarinic antagonist approved for the maintenance treatment of chronic obstructive pulmonary disease (COPD).[1] It is also approved for this indication in combination with vilanterol (as umeclidinium bromide/vilanterol).[2][3]
In the 2014, the drug was also approved in the E.U. and in the U.S. for the maintenance treatment to relieve symptoms in adult patients with chronic obstructive pulmonary disease (COPD). It was launched in the U.K. in October 2014 and in the U.S. in January 2015. In Japan, the product candidate was approved in 2015 as monotherapy for the maintenance bronchodilator treatment to relieve symptoms in adult patients with chronic obstructive pulmonary disease (COPD) and launched on October in the same year.

Umeclidinium bromide (Ellipta)
Umeclidinium bromide is a long-acting muscarinic acetylcholine antagonist developed by GlaxoSmithKline and approved by the US FDA at the end of 2013 for use in combination with vilanterol, a b2 agonist, for the treatment of chronic obstructive pulmonary disease.269 Due to umeclidinium’s poor oral bioavailability, the drug is administrated by inhalation as dry powder.269
The most likely scale preparation of the drug is described in Scheme .270
Commercially available ethyl isonipecotate (278) was alkylated with 1-bromo-2-chloroethane in the presence of K2CO3 in acetone to give ethyl 1-(2-chloroethyl)piperidine-4-carboxylate (279). This material was then treated with lithium diisopropylamine (LDA) in THF to affect a transannular substitution reaction resulting in the cyclized quinuclidine 280 in 96% yield.270 Excess of phenyllithium was added to ester 280 in THF starting at low temperature then gradually warming to room temperature to give tertiary alcohol 281 in 61% yield. Amine 281 was finally alkylated with benzyl 2-bromoethyl ether (282) in MeCN/CHCl3 at elevated temperatures
to afford umeclidinium bromide (XXXV) in 69% yield.
269. Tal-Singer, R.; Cahn, A.; Mehta, R.; Preece, A.; Crater, G.; Kelleher, D.;Pouliquen, I. J. Eur. J. Pharmacol. 2013, 701, 40.
270. Laine, D. I.; McCleland, B.; Thomas, S.; Neipp, C.; Underwood, B.; Dufour, J.;Widdowson, K. L.; Palovich, M. R.; Blaney, F. E.; Foley, J. J.; Webb, E. F.;Luttmann, M. A.; Burman, M.; Belmonte, K.; Salmon, M. J. Med. Chem. 2009, 52, 2493.
FDA
https://www.accessdata.fda.gov/drugsatfda_docs/nda/2013/203975Orig1s000ChemR.pdf
1-[2-(benzyloxy)ethyl]-4-(hydroxydiphenylmethyl)-1-azoniabicyclo[2.2.2]octane bromide

PATENT
https://patents.google.com/patent/CN105461710A/en
umeclidinium bromide prepared patent US7439393, US RE44874, US 7488827, US 7498440, US7361787 and the like using phenyllithium prepared by reaction of intermediate 4 – [(diphenyl) hydroxymethyl] azabicyclo [2.2.2 ] octane.Specific methods: azabicyclo [2.2.2] octane-nucleophilic addition reaction with 4-carboxylate-fold amount of 2.02-2.5 phenyllithium occurs, the reaction temperature is controlled to -78 ° 0_15 ° C ο lithium Reagents expensive, difficult to store, use of harsh conditions, relatively high cost.
Example 1
Phenyl magnesium chloride: Under nitrogen atmosphere to 55g (2.3mol) of metallic magnesium sandpaper lit with 3 L of tetrahydrofuran was added dropwise 215g (1.91mol) chlorobenzene, micro-thermal reaction proceeds, controlled dropping, the reaction was kept boiling, dropwise for about 1.5 hours, after the dropping was heated slightly under reflux for 30min. Cool reserve.
[0008] Example 2
Phenyl magnesium bromide: The under argon 50.4g (2.lmol) sandpaper lit magnesium metal with 4.2 liters of anhydrous ethyl ether was added a solution of 300g (1.91mol) of bromobenzene, was added an iodine initiator, electrical hair fever reaction proceeds, controlled dropping, the reaction was kept boiling, about 1.5 hours dropwise was added dropwise to a gentle reflux heated 30min. Cool reserve.
[0009] Example 3
Preparation of crude product: azabicyclo [2.2.2] octane-4-carboxylate (135g, 0.736mo 1) was dissolved in 3L of tetrahydrofuran, under nitrogen, was cooled to -5~0 ° C, was added dropwise 300g preparation of benzyl bromide Grignard reagent. After incubation -5~0 ° C stirred for 1 hour (progress of the reaction was monitored by TLC sample). Adding 50ml of water quenching. Liquid separation, the aqueous phase was extracted twice with 500ml of tetrahydrofuran, and the combined organic phases were washed with water, dried and filtered. The solvent was partially removed under reduced pressure, the balance maintaining approximately 1L, the residue was stirred overnight at 20 ° C crystallization.Filtered, washed (petroleum ether 2 X 200 ml), the filter cake was dried at 40 ° C in vacuo to give a yellowish white crystals 121.2 g, yield 54.2%.
[0010] Example 4
Preparation of crude product: azabicyclo [2.2.2] octane-4-carboxylate (18.3g, 0.lOmo 1) was dissolved in 3L of tetrahydrofuran, under nitrogen, was cooled to 0~5 ° C, was added dropwise 0.25 mol phenyl magnesium chloride. After incubation 0~5 ° C stirred for 1 hour (progress of the reaction was monitored by TLC sample) o quenched with 10ml of water was added. Liquid separation, the aqueous phase was extracted twice with 100ml of tetrahydrofuran, and the combined organic phases were washed with water, dried and filtered. The solvent was partially removed under reduced pressure, the balance maintaining approximately 50mL, the residue was stirred overnight at 20 ° C crystallization.Filtered, washed (petroleum ether 2X20 ml), the filter cake was dried at 40 ° C in vacuo to give a yellowish white crystals 14.63 g, yield 48.1%.
[0011] Example 5
Preparation of crude product: azabicyclo [2.2.2] octane-4-carboxylate (18.38,0.1011101) ^ 31 was dissolved in tetrahydrofuran, under nitrogen, was cooled to 5~15 ° C, was added dropwise 0.30 mol of benzene bromide. After incubation 5~15 ° C stirred for 1 hour (progress of the reaction was monitored by TLC sample) o quenched with 10ml of water was added. Liquid separation, the aqueous phase was extracted twice with 100ml of tetrahydrofuran, and the combined organic phases were washed with water, dried and filtered. The solvent was partially removed under reduced pressure, the balance maintaining approximately 50mL, the residue was stirred overnight at 20 ° C crystallization.Filtered, washed (petroleum ether 2 X 20 ml), the filter cake was dried at 40 ° C in vacuo to yield 13.80 g of yellow-white crystals, yield 47.1%.
[0012] Example 6
Umeclidinium bromide purification: 100g crude product was dissolved in 320ml of water to 80 ° C a mixture of 640ml of acetone, add 5g active carbon, and filtered.The filtrate was cooled to 25 ° C, for 1 hour. Within 1 to 2 hours and cooled to 0~5 ° C for 3 hours. The filter cake with chilled 1: 2 acetone – washed twice with water (2x20ml). The filter cake was dried in vacuo at 60 ° C to give white crystalline solid (92 g, yield 92%). Purity (HPLC normalization method) 99.25%.
[0013] Example 7
Umeclidinium bromide purification: 100g crude product was dissolved in 180ml water at 50 ° C a mixture of 360ml of acetone, add 5g active carbon, and filtered.The filtrate was ~ 2 hours to 25 ° C, for 1 hour. Within 1 to 2 hours cooled to 0 ° C and left overnight protection. The filter cake with chilled 1: 2 acetone – washed twice with water (2x20ml). The filter cake was dried at 60 ° C in vacuo to give fine (98.3 g, yield 98.3%). Purity (HPLC normalization method) 97.75%.
PATENT
https://patents.google.com/patent/WO2014027045A1
International Patent Publication Number WO 2005/104745 (Glaxo Group Limited), filed 27th April 2005, discloses muscarinic acetylcholine receptor antagonists. In particular, WO 2005/104745 discloses 4- [hydroxy(diphenyl)methyl]-l-{2-[(phenylmethyl)oxy]ethyl}-l-azoniabicyclo[2.2.2]octane bromide, of formula (I), and a process for the preparation of this compound (Example 84):
4-[Hydroxy(diphenyl)methyl]-l-{2-[(phenylmethyl)oxy]ethyl}-l-azoniabicyclo[2.2.2]octane bromide may also be referred to as umeclidinium bromide.
International Patent Publication Number WO 2011/029896 (Glaxo Group Limited), filed 10th September 2010, discloses an alternative preparation for an early intermediate, ethyl-l-azabicyclo[2.2.2] octane-4-carboxylate, in the multi-step synthesis of umeclidinium bromide.
There exists a need for an alternative process for the preparation of umeclidinium bromide. In particular, a process that offers advantages over those previously disclosed in WO 2005/104745 and WO 2011/029896 is desired. Advantages may include, but are not limited to, improvements in safety, control (i.e of final product form and physical characteristics), yield, operability, handling, scalability, and efficiency.
Summary of the Invention
The present invention provides, in a first aspect, a process for the preparation of umeclidinium bromide, which comprises: a) reacting ((2-bromoethoxy)methyl)benzene, of formula (II)
in a dipolar aprotic solvent with a boiling point greater than about 90°C or an alcohol with a boiling point greater than about 80°C; and optionally
b) re-crystallising the product of step (a).
The present invention is further directed to intermediates used in the preparation of the compound of formula (III), and hence of umeclidinium bromide. The process disclosed herein provides a number of advantages over prior art processes of WO 2005/104745 and WO 2011/029896.

PATENT
EP 3248970
FORM A B AND AMORPHOUS
https://patents.google.com/patent/EP3248970A1/en
The invention relates to novel solid forms of umeclidinium bromide (I), chemically 1-[2-(benzyloxy)ethyl]-4-(hydroxydiphenylmethyl)-1-azabicyclo[2.2.2]octane bromide. In particular, to its novel crystalline forms, identified as form A and form B, as well as to an amorphous form, and to their characterization by means of analytic methods. The invention further relates to methods of their preparation and their use for the preparation of umeclidinium bromide in the API quality.
Umeclidinium bromide is indicated as an inhalation anticholinergic drug with an ultra-long-term effect in cooperating patients with the diagnosis of COPD (chronic obstructive pulmonary disease). COPD is defined as a preventable and treatable disease that is characterized by a persistent obstruction of air flow in the bronchi (bronchial obstruction), which usually progresses and is related to an intensified inflammatory response of the airways to harmful particles or gases. The main goal of the treatment of COPD is an improvement of the current control, i.e. elimination of symptoms, improvement of toleration of physical effort, improvement of the health condition and reduction of future risks, i.e. prevention and treatment of exacerbations, prevention of progression of the disease and mortality reduction
The structure of umeclidinium bromide, 1-[2-(benzyloxy)ethyl]-4-(hydroxydiphenylmethyl)-1-azabicyklo[2.2.2]octane bromide, is first mentioned in the general patent application WO2005009362 of 2003 .
Preparation of umeclidinium bromide is first disclosed in the patent EP 1 740 177B ( WO2005104745 ), where two methods (A and B) are mentioned, differing in the final processing and the product yield (method B included in Scheme 1). There, the last steps of the synthesis are described, the product being described by means of EI-MS, 1H NMR and elementary analysis. There is no information concerning the chemical purity or polymorphic form.
Preparation of the amorphous form of umeclidinium bromide
PAPER
Synthetic Communications An International Journal for Rapid Communication of Synthetic Organic Chemistry , Volume 48, 2018 – Issue 9, Convenient new synthesis of umeclidinium bromide
Umeclidinium bromide, a drug used for chronic obstructive pulmonary disease, is synthesized through a new intermediate of phenyl(quinuclidin-4-yl)methanone. This novel method with simple operation flow and cheap reagents, makes it suitable for scale up. The overall four-step process provides umeclidinium bromide in 29% yield and the purity up to 99.83%. The X-ray crystal structure of the drug molecule was first reported.



 |
|
| Clinical data | |
|---|---|
| Trade names | Incruse Ellipta |
| Synonyms | GSK573719A |
| License data | |
| Pregnancy category |
|
| Routes of administration |
Inhalation (DPI) |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Protein binding | ~89%[1] |
| Metabolism | Hepatic (CYP2D6) |
| Elimination half-life | 11 hours |
| Excretion | Feces (58%) and urine(22%) |
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| KEGG | |
| ChEBI | |
| ECHA InfoCard | 100.166.375 |
| Chemical and physical data | |
| Formula | C29H34BrNO2 |
| Molar mass | 508.49 g/mol |
| 3D model (JSmol) | |
//////////////Umeclidinium bromide, Incruse Ellipta, ウメクリジニウム臭化物 , GSK573719A, UNII-7AN603V4JV, FDA 2014
C1C[N+]2(CCC1(CC2)C(C3=CC=CC=C3)(C4=CC=CC=C4)O)CCOCC5=CC=CC=C5.[Br-]
| FDA Orange Book Patents: 1 of 15 (FDA Orange Book Patent ID) | |
|---|---|
| Patent | 9750726 |
| Expiration | Nov 29, 2030 |
| Applicant | GLAXOSMITHKLINE |
| Drug Application |
|
| FDA Orange Book Patents: 2 of 15 (FDA Orange Book Patent ID) | |
|---|---|
| Patent | 6759398 |
| Expiration | Aug 3, 2021 |
| Applicant | GLAXOSMITHKLINE |
| Drug Application |
|
| FDA Orange Book Patents: 3 of 15 (FDA Orange Book Patent ID) | |
|---|---|
| Patent | 7439393 |
| Expiration | May 21, 2025 |
| Applicant | GLAXOSMITHKLINE |
| Drug Application |
|
| FDA Orange Book Patents: 4 of 15 (FDA Orange Book Patent ID) | |
|---|---|
| Patent | 7629335 |
| Expiration | Aug 3, 2021 |
| Applicant | GLAXOSMITHKLINE |
| Drug Application |
|
| FDA Orange Book Patents: 5 of 15 (FDA Orange Book Patent ID) | |
|---|---|
| Patent | 7776895 |
| Expiration | Sep 11, 2022 |
| Applicant | GLAXOSMITHKLINE |
| Drug Application |
|
| FDA Orange Book Patents: 6 of 15 (FDA Orange Book Patent ID) | |
|---|---|
| Patent | 8161968 |
| Expiration | Feb 5, 2028 |
| Applicant | GLAXOSMITHKLINE |
| Drug Application |
|
| FDA Orange Book Patents: 7 of 15 (FDA Orange Book Patent ID) | |
|---|---|
| Patent | 8201556 |
| Expiration | Feb 5, 2029 |
| Applicant | GLAXO GRP ENGLAND |
| Drug Application | N205382 (Prescription Drug: INCRUSE ELLIPTA . Ingredients: UMECLIDINIUM BROMIDE) |
| FDA Orange Book Patents: 8 of 15 (FDA Orange Book Patent ID) | |
|---|---|
| Patent | 6537983 |
| Expiration | Aug 3, 2021 |
| Applicant | GLAXOSMITHKLINE |
| Drug Application |
|
| FDA Orange Book Patents: 9 of 15 (FDA Orange Book Patent ID) | |
|---|---|
| Patent | 7498440 |
| Expiration | Apr 27, 2025 |
| Applicant | GLAXOSMITHKLINE |
| Drug Application |
|
| FDA Orange Book Patents: 10 of 15 (FDA Orange Book Patent ID) | |
|---|---|
| Patent | 7488827 |
| Expiration | Dec 18, 2027 |
| Applicant | GLAXOSMITHKLINE |
| Drug Application |
|
| FDA Orange Book Patents: 11 of 15 (FDA Orange Book Patent ID) | |
|---|---|
| Patent | 8183257 |
| Expiration | Jul 27, 2025 |
| Applicant | GLAXOSMITHKLINE |
| Drug Application |
|
| FDA Orange Book Patents: 12 of 15 (FDA Orange Book Patent ID) | |
|---|---|
| Patent | 6878698 |
| Expiration | Aug 3, 2021 |
| Applicant | GLAXOSMITHKLINE |
| Drug Application |
|
| FDA Orange Book Patents: 13 of 15 (FDA Orange Book Patent ID) | |
|---|---|
| Patent | 8511304 |
| Expiration | Jun 14, 2027 |
| Applicant | GLAXOSMITHKLINE |
| Drug Application |
|
| FDA Orange Book Patents: 14 of 15 (FDA Orange Book Patent ID) | |
|---|---|
| Patent | RE44874 |
| Expiration | Mar 23, 2023 |
| Applicant | GLAXOSMITHKLINE |
| Drug Application |
|
| FDA Orange Book Patents: 15 of 15 (FDA Orange Book Patent ID) | |
|---|---|
| Patent | 8309572 |
| Expiration | Apr 27, 2025 |
| Applicant | GLAXOSMITHKLINE |
| Drug Application |
|